column If you would like a subscription, or more information, please contact your nearest Waters Office.
|
|
|
- Kerry Nelson
- 5 years ago
- Views:
Transcription
1 column Waters A Reprint from Spring 995 If you would like a subscription, or more information, please contact your nearest Waters Office. Check the Waters website for up-to-date information: Published by Waters Corporation 34 Maple Street Milford, MA 0757 USA Tel: Toll-free: Fax: Editor: Uwe D. Neue, Ph.D. ISSN # Waters Corporation
2 Waters is giving a little something extra to the customer who purchases the 0,000th Symmetry column. Spring 995 Separation of Enantiomers by Micellar Electrokinetic Capillary Chromatography with Novel Chiral Surfactants Jeffrey R. Mazzeo, Edward R. Grover and Michael E. Swartz through the use of a chiral stationary phase, or through the use of a chiral mobile phase additive. Many chiral selectors have been described for the separation of enantiomeric mixtures by LC. There are more than fifty commercially available chiral LC columns, which include Pirkle-type, cyclodextrin, protein, carbohydrate, crown ether, and ligand exchange (). Table of Contents Novel Chiral Surfactants Interconverting Conformers Summary This paper describes the separation of enantiomers using a novel chiral surfactant and capillary electrophoresis (CE). Before describing the results, theoretical background is given that illustrates the advantage of using CE instead of LC in achieving the resolution of enantiomers. Enantioselectivity data obtained with a chiral stationary phase used in LC shows that the applicability of a given chiral selector is significantly increased when used in CE. Experimental results using chiral surfactants for the CE separation of enantiomers are consistent with this hypothesis. Introduction The separation and quantitation of enantiomeric mixtures is important because many drugs, agrochemicals, food additives and fragrances are chiral, and their enantiomers have been shown to exhibit different bioactivity. As a result, the FDA has published conservative guidelines to ascertain enantiopurity. A mixture of two enantiomers can be separated through the use of a chiral selector. Liquid chromatography (LC) is currently the predominant method for separating enantiomers. In LC, separation of enantiomers may be achieved Anionic Ion Pairing Literature Corner Ion Analysis by CE Application Notebook Polyolefin Separation Antisense Therapeutic Amino Acid Analysis In This Issue: Look for our NEW Pull-out Applications Notes
3 A researcher interested in separating a chiral compound into its two enantiomers and determining enantiomeric purity by LC is faced with a daunting task. Frequently, five or more chiral stationary phases must be screened before partial resolution is achieved. To optimize partitioning, the mobile phase can be altered; but in many cases, simple changes in the mobile phase can lead to complete loss of enantioselectivity. After considerable trial and error, the desired resolution may be obtained. To make matters worse, chiral stationary phases often suffer from reproducibility and/or stability issues. As a result, LC separation of enantiomers is generally an expensive (columns, mobile phases, method development time), labor intensive process. To understand why separation of enantiomers is difficult by LC, the resolution equation for chromatography must be examined: Figure a: A Graph of Pirkle Data (2) Showing Success Rate with N=5,000 % of Compounds Alpha 32% Resolved Data from reference 2 was plotted as alpha versus percent of compounds exhibiting at least this alpha. Shaded area represents the percent of the compounds examined which could be baseline resolved assuming a capacity factor of and 5,000 theoretical plates. Figure b: Graph of Pirkle Data (2) Showing Success Rate with N=00, Rs = N 2 4 ( ) α α A fundamental problem exists because enantioselectivity values (α) are not great enough to compensate for the low plate counts (N) obtained in LC. The plate counts of LC chiral columns are limited to a maximum of about 5,000. Partitioning can be optimized by increasing or decreasing the strength of the mobile phase. Unfortunately, these modifications can often influence selectivity in an unpredictable way. Given these limitations, the goal has been to develop phases with higher α. However, because pharmaceutical and agrochemical compounds are diverse in structure, a chiral phase which achieves high alphas for many structures has been elusive. Since a limit has apparently been reached in developing chiral LC phases with higher enantioselectivity, it is logical to try existing chiral selectors in a separation system which exhibits significantly higher plates (N) than LC. Capillary electrophoresis is an ideal system for chiral separations since typical plate counts for separation of k 2 k 2 + % of Compounds Alpha 98% Resolved Data from reference 2 was plotted as alpha versus percent of compounds exhibiting at least this alpha. Shaded area represents the percent of the compounds examined which could be baseline resolved assuming a capacity factor of and 00,000 theoretical plates. small molecules are 00,000 or greater. Referring to the resolution equation above, assume a capacity factor value of (k 2 = ) for a given separation. Under these conditions, an LC chiral column exhibiting 5,000 theoretical plates would require an alpha value of.20 to achieve baseline resolution (Rs =.5). A chiral selector in CE exhibiting 00,000 theoretical plates would require an alpha value of.04 to achieve baseline resolution. (An alpha value of.04 in LC would lead to a resolution of 0.3.) Therefore, a given chiral selector will resolve more enantiomeric mixtures when used in CE compared to LC. Another way to illustrate the advantages of high efficiency in the separation of enantiomers is to examine the enantioselectivity data published by Pirkle on a 2,2,2-trifluoro--(9-anthryl)- ethanol LC phase (2). A total of 52 racemates were screened with this phase. The enantioselectivity data for 2
4 these compounds can be plotted as alpha vs. % of the compounds exhibiting at least this alpha. For instance, 00% of the compounds had an alpha of.02 or greater, 98% had an alpha of.04 or greater, etc. Assuming a capacity factor value of and a desired resolution of.5, this phase could resolve 32% of the compounds screened when used in an LC mode exhibiting 5,000 theoretical plates (Figure a). The same phase, when used in a CE format exhibiting 00,000 theoretical plates, could resolve 98% of the compounds (Figure b). Exploiting the phase s enantioselectivity in CE could increase its applicability threefold. There are several desirable characteristics of a chiral selector for use in CE. First, the ideal selector should lead to high efficiencies, otherwise it will not be broadly applicable. Since the selector is dissolved in the buffer, which will constantly move through the detector, it should have low UV absorbance. It should also be usable over a wide ph range, since ph is a critical selectivity variable in CE. It should be easily implemented, and available in high chemical and optical purity. Finally, both enantiomers of the selector should be available. This will allow for exact enantiomer migration order reversal. The ability to reverse enantiomer migration order is important to improve quantitation of enantiomeric purity and to determine if a resulting separation is enantiomeric in nature. Many different chiral selectors have been described in CE, including proteins, crown ethers, cyclodextrins and bile salts. Proteins suffer from several disadvantages, including high UV absorbance, low efficiency, and finite ph range. Crown ethers have been applicable only to primary amines. Bile salts have been useful only for hydrophobic analytes and, in general, exhibit low enantioselectivity. Cyclodextrins currently enjoy the most popularity as chiral selectors for CE, but have not been widely applicable. None of these selectors allows for exact enantiomer migration order reversal. We have focused our efforts on the development of novel chiral surfactants for the separation of enantiomers by micellar electrokinetic capillary chromatography (MECC). (R)- and (S)-Ndodecoxycarbonylvaline, chiral surfactants synthesized in our laboratories, meet the required characteristics of a chiral selector for CE (3). The rest of this article will overview the performance of these novel chiral surfactants. Experimental All CE separations were performed on a Quanta 4000E Capillary Electrophoresis System (Waters, Milford, MA). Buffers were prepared with surfactant and disodium phosphate and/or disodium tetraborate. The ph was then adjusted with either sodium hydroxide or phosphoric acid ( J.T. Baker). Prior to use, AccuSep (Waters) capillaries, 50 µm I.D. x 60 cm (52.5 cm effective distance), were rinsed for five minutes with 0.5 mol/l NaOH. Between injections, the capillaries were rinsed with 0. mol/l NaOH (3 minutes) and buffer (3 minutes). Unless indicated, separations were performed with UV detection at 24 nm and a voltage of + 2 kv. Hydrostatic injection times ranged from 2 to 20 seconds. All buffers and samples were filtered through a 0.45 µm filter (Millipore, Bedford, MA). Separations were performed at ambient temperature. Samples were prepared as 0 mg/ml stock solutions in methanol and then diluted to 0. mg/ml in buffer. The micelle migration time was measured with the hydrophobic compound sulconazole. Racemates and the individual enantiomers (when possible) were purchased from Sigma (St. Louis, MO) and Aldrich (Milwaukee, WI). Figure 2: Structure of (S)-N-dodecoxycarbonylvaline Results and Discussion In MECC, anionic surfactant molecules are added to CE buffers at concentrations above their critical micelle concentration (cmc), resulting in aggregates of surfactant monomers known as micelles. These micelles consist of a hydrophobic core and a hydrophilic surface. Analytes partition between the aqueous CE buffer and the micelle, resulting in separation based on hydrophobicity, hydrogen bonding and ionic interaction. Incorporation of a chiral selector into the surfactant molecules affords micelles with chiral selectivity. The structure of (S)-N-dodecoxycarbonylvaline is shown in Figure 2. In this surfactant, the amino acid valine is the chiral selector. Under typical operating conditions, MECC operates with a finite migration window (unlike LC where there is an infinite elution window). In uncoated fused silica capillaries with cathodic electroosmotic flow, anionic micelles move slowly toward the cathode, while unpartitioned analytes move rapidly toward the cathode. This condition results in a migration window, the boundaries of which an analyte must migrate within. Because of this finite migration window, the resolution equation for MECC is different from the resolution equation for LC (4): Rs = N 2 4 ( ) α α k 2 k + 2 k 2 t aq t mc + t aq where t aq is the migration time of the analyte if it does not interact with the micelles and is the migration time if it completely partitions into the micelle. O (S) N dodecoxycarbonylvaline O H N H O OH 3
5 The equation for calculation of the capacity factor value is also different from the one used in LC (4): k = t r t aq t aq t r k optimum = t aq where t r is the observed migration time of the analyte. Note that if approaches infinity, both the resolution equation and the capacity factor equation are identical to those used in LC. The limited migration window also leads to an optimum value for the capacity factor, which is determined by the values of t aq and (5): The separation of pseudoephedrine enantiomers using (S)-N-dodecoxycarbonylvaline in Figure 3 demonstrates an MECC migration window. The baseline disturbance at minutes is the marker for the electroosmotic flow (methanol in this case, formamide may also be used), the migration time of a neutral compound which does not partition into the micelles. The peak at minutes is the micelle marker, the migration time for sulconazole, which is completely partitioned into the micelles. The peaks at and minutes are the two pseudoephedrine enantiomers, which are separated with a resolution value of 5.2. In this case, the alpha value was.24, and the average plate count was 04,000. Since in MECC the selective phase is dissolved in the buffer, k can be linearly changed through the surfactant concentration (phase ratio) (4), with no change in alpha. Figure 4 shows the separation of atenolol enantiomers at two surfactant concentrations. At 25 mm, k was 0.6, less than the optimum value of 2.8, and the resolution was 0.8. Increasing the surfactant concentration to 00 mm led to a capacity factor of 2.5 and a resolution of 2.7. In both cases, α was.04. This ability to optimize partitioning independent of selectivity is a significant advantage of MECC versus LC. 2 Figure 3: Separation of Pseudoephedrine Enantiomers The baseline disturbance at minutes is the electroosmotic flow marker (methanol) and the peak at minutes is the micelle marker (sulconazole). The resolution value of the two enantiomers, was 5.2, the alpha value.24 and the average plate count 04,000. Figure 4: Influence of Surfactant Concentration on the Separation of Atenolol Enantiomers mau mau mau Sample: 00 µg/ml racemic pseudoephedrine and 50 µg/ml sulconazole dissolved in buffer Buffer: 50 mm (S)-N-dodecoxycarbonylvaline 25 mm Na 2 HPO 4 /25 mm NaB 4 O 7 Reproduced from reference 3, with permission of Elsevier Science Publishers t os Minutes Sample: 00 µg/ml racemic atenolol dissolved in buffer Buffer: 25 mm or 00 mm (S)- N-dodecoxycarbonylvaline 25 mm Na 2 HPO 4 /25 mm NaB 4 O 7 Reproduced from reference 3, with permission of Elsevier Science Publishers. 25 mm SURFACTANT 00 mm SURFACTANT Minutes At 25 mm surfactant, the capacity factor for the two enantiomers was 0.6, less than the optimum value of 2.8, and the resolution was only 0.8. At 00 mm surfactant, k was 2.5, closer to the optimum of 2.8, and resolution was
6 (R)- and (S)-N-dodecoxycarbonylvaline can be used to exactly reverse the migration order of a separated enantiomeric pair. This ability is shown in Figure 5, where the two enantiomers of the surfactant are used to separate a 3: ratio of (S):(R) benzoin. With the (R)-surfactant, the (S)-enantiomer of benzoin was more highly retained and migrated second. With the (S)-surfactant, the (S)-enantiomer was less retained and migrated first. This ability to exactly reverse migration order is important to improve quantitation of enantiomeric purity and to determine if a resulting separation is enantiomeric. Peaks which represent the separation of enantiomers will be reversed in migration order when run with the two enantiomers of the surfactant, while achiral constituents of the sample migrate at the same time with both enantiomers of the surfactant. A general advantage of CE over LC is the ability to perform fast separations. Fast chiral separations can be obtained with (R)- and (S)-N-dodecoxycarbonylvaline when a low ionic strength buffer and high field strength are employed. Figure 6 shows the separation of N- methylpseudoephedrine enantiomers in less than 90 seconds under such conditions. Figure 5: Migration Order Reversal of Benzoin Enantiomers mau mau Sample: 3: ratio of (S)- to (R)-benzoin (200 µg/ml) dissolved in buffer Buffer: 25 mm (R)- or (S)-N-dodecoxycarbonylvaline 25 mm Na 2 HPO 4 /25 mm NaB 4 O 7 Reproduced from reference 3, with permission of Elsevier Science Publishers. (R) SURFACTANT (S) SURFACTANT 2 (R) (S) (S) (R) Minutes (R)-Benzoin migrated first with (R)-N-dodecoxycarbonylvaline, while (S)-benzoin migrated first with (S)-N-dodecoxycarbonylvaline. The migrate times of the peaks were the same in the two cases. Figure 6: Fast Separation of N-methylpseudoephedrine Enantiomers Sample: 00 µg/ml racemic N-methylpseudoephedrine dissolved in buffer Buffer: 25 mm (S)-N-dodecoxycarbonylvaline 50 mm CHES Capillary: 50 µm i.d. x 35 cm Voltage: + 30 kv Reproduced from reference 3, with permission of Elsevier Science Publishers Seconds The enantiomers of N-methylpseudoephedrine were separated in less than 90 seconds using a low ionic strength buffer, high field strength and short capillary. 5
7 (R)- and (S)-N-dodecoxycarbonylvaline have been successful in separating neutral and basic enantiomeric mixtures which contain at least two hydrogen bonding sites near the chiral center. Compounds which are acidic will separate if they are reasonably hydrophobic. Hydrophilic acids are generally not separable due to low partitioning, most likely due to charge repulsion. Performing separations at acidic ph would minimize any charge repulsion, however, (R)- and (S)-N-dodecoxycarbonylvaline are insoluble below ph 6.5. A list of chiral compounds which have been separated with (R)- and (S)-N-dodecoxycarbonylvaline to date is given in Table I. Compounds which have structures similar to those in Table I should be separable. We continue to add to this list as improved methods of implementing the chiral surfactants are developed. The novel chiral surfactants described in this article are currently under development at Waters. If you would like to be kept informed on new developments in this area, please check box number 3 on the reply card. Conclusion Capillary electrophoresis promises to play a major role in the analytical scale separation of enantiomeric mixtures. CE separations of enantiomers have the potential to replace LC methods, due to higher efficiency, faster methods development time, reduced solvent consumption, and ease of use. MECC with synthetic chiral surfactants represents the ideal method for separation of enantiomers in CE. The advantages of this approach include exact enantiomer migration order reversal, simultaneous Table I: Compounds Baseline Resolved with (R)- and (S)-N-dodecoxycarbonylvaline Acebutolol AQC-Amino Acids Aminoglutethimide Atenolol Benzoin Bupivacaine CBZ-Amino Acids Disopyramide Ephedrine Epinephrine FMOC-Amino Acids Glutethimide Homatropine Isoproterenol Ketamine Laudanosine chiral and achiral separations, straightforward methods development, and high purity. Waters synthetic chiral surfactant technology offers the ability to perform controlled surfactant design, so that those structural features which influence enantioselectivity can be studied and combined into new surfactants. Our goal is to develop 4-6 complementary chiral surfactants which will separate all chiral compounds. In the next article, method development with the surfactants will be described. Methadone Metoprolol Nadolol N-Methylpseudoephedrine Norephedrine Norphenylephrine Octopamine Oxprenolol Phenylglutethimide Pindolol Propranolol PTH-Amino Acids Terbutaline Tetramisole Tolperisone Warfarin References () M. Zief and L.J. Crane (Editors), Chromatographic Chiral Separations, Marcel Dekker, Inc., New York, 988. (2) W. Pirkle and D. House, J. Org. Chem., 44 (979) 957. (3) J. Mazzeo, E. Grover, M. Swartz and J. Petersen, J. Chromatogr., in press. (4) S. Terabe, K. Otsuka and T. Ando, Anal. Chem., 57 (985) 834. (5) J. Foley, Anal. Chem., 62 (990) 302. Special Offer If you are using Bondclone, Whatman s Equivalent or any other brand product that claims to be similar to a Waters µbondapak C 8, we have a special offer for you. Please call , ext for information on this special offer. µbondapak C 8 is a trademark of Waters Corporation. Bondclone is a trademark of Phenomenex, Inc. 6
Capillary electromigration techniques:
 CE capillary electrophoresis CZE capillary zone electrophoresis EKC electrokinetic chromatography MEKC/MECC micellar electrokinetic chromatography MEEKC microemulsion electrokinetic chromatography CEC
CE capillary electrophoresis CZE capillary zone electrophoresis EKC electrokinetic chromatography MEKC/MECC micellar electrokinetic chromatography MEEKC microemulsion electrokinetic chromatography CEC
Improved Pirkie Type 1 Amide Type: Asymmetric carbon atoms are bonded directly with CONH group
 1 sur 6 26/06/2013 14:16 Special Merits of SUMICHIRAL OA 1.The large number of theoretical plates of the columns offer high resolution. 2.The packing materials have chemical stability and the columns have
1 sur 6 26/06/2013 14:16 Special Merits of SUMICHIRAL OA 1.The large number of theoretical plates of the columns offer high resolution. 2.The packing materials have chemical stability and the columns have
UPLC/MS Monitoring of Water-Soluble Vitamin Bs in Cell Culture Media in Minutes
 UPLC/MS Monitoring of Water-Soluble Vitamin Bs in Cell Culture Media in Minutes Catalin E. Doneanu, Weibin Chen, and Jeffrey R. Mazzeo Waters Corporation, Milford, MA, U.S. A P P L I C AT ION B E N E F
UPLC/MS Monitoring of Water-Soluble Vitamin Bs in Cell Culture Media in Minutes Catalin E. Doneanu, Weibin Chen, and Jeffrey R. Mazzeo Waters Corporation, Milford, MA, U.S. A P P L I C AT ION B E N E F
IAM Chromatography. Introduction
 IAM Chromatography IAM Drug Discovery Columns Introduction References: 1. Regis 1998-99 Chromatography Catalog A simple, rapid method to predict drug transport across biological barriers has been a long
IAM Chromatography IAM Drug Discovery Columns Introduction References: 1. Regis 1998-99 Chromatography Catalog A simple, rapid method to predict drug transport across biological barriers has been a long
176 YMC Chiral Columns
 176 YMC Chiral Columns YMC Chiral Columns 177 Chiral Columns Content YMC Chiral NEA(R)(S)... 178-181 YMC Chiral CD BR... 182-185 Ordering Information... 186 HPLC Columns for Optical Isomer Separation Introduction
176 YMC Chiral Columns YMC Chiral Columns 177 Chiral Columns Content YMC Chiral NEA(R)(S)... 178-181 YMC Chiral CD BR... 182-185 Ordering Information... 186 HPLC Columns for Optical Isomer Separation Introduction
Micellar Electrokinetic
 Micellar Electrokinetic Chromatography MEKC belongs to a mode of CE but also to micro-lc. Shigeru Terabe University of Hyogo (Japan) About 20 years have passed since publication of the first paper on micellar
Micellar Electrokinetic Chromatography MEKC belongs to a mode of CE but also to micro-lc. Shigeru Terabe University of Hyogo (Japan) About 20 years have passed since publication of the first paper on micellar
Implementing a Generic Methods Development Strategy for Enantiomer Analysis
 2 Chapman and Fu-Tai Chen Implementing a Generic Methods Development Strategy for Enantiomer Analysis Jeff Chapman and Fu-Tai A. Chen, Beckman Coulter Inc., Fullerton, California, USA. Introduction Whether
2 Chapman and Fu-Tai Chen Implementing a Generic Methods Development Strategy for Enantiomer Analysis Jeff Chapman and Fu-Tai A. Chen, Beckman Coulter Inc., Fullerton, California, USA. Introduction Whether
Enantioresolution of Amphetamine, Methamphetamine, and Deprenyl (Selegiline) by LC, GC, and CE
 Enantioresolution of Amphetamine, Methamphetamine, and Deprenyl (Selegiline) by LC, GC, and CE Daniel W. Armstrong*, Kimbry L. Rundlett, and Usha B. Nair Department of Chemistry, University of Missouri-Rolla,
Enantioresolution of Amphetamine, Methamphetamine, and Deprenyl (Selegiline) by LC, GC, and CE Daniel W. Armstrong*, Kimbry L. Rundlett, and Usha B. Nair Department of Chemistry, University of Missouri-Rolla,
Capillary electrophoretic techniques toward the metabolome analysis*
 Pure Appl. Chem., Vol. 73, No. 10, pp. 1563 1572, 2001. 2001 IUPAC Capillary electrophoretic techniques toward the metabolome analysis* Shigeru Terabe 1,, Michal J. Markuszewski 1, Naoko Inoue 1, Koji
Pure Appl. Chem., Vol. 73, No. 10, pp. 1563 1572, 2001. 2001 IUPAC Capillary electrophoretic techniques toward the metabolome analysis* Shigeru Terabe 1,, Michal J. Markuszewski 1, Naoko Inoue 1, Koji
[ APPLICATION NOTE ] The Separation of 8 -THC, 9 -THC, and Their Enantiomers by UPC 2 Using Trefoil Chiral Columns INTRODUCTION APPLICATION BENEFITS
![[ APPLICATION NOTE ] The Separation of 8 -THC, 9 -THC, and Their Enantiomers by UPC 2 Using Trefoil Chiral Columns INTRODUCTION APPLICATION BENEFITS [ APPLICATION NOTE ] The Separation of 8 -THC, 9 -THC, and Their Enantiomers by UPC 2 Using Trefoil Chiral Columns INTRODUCTION APPLICATION BENEFITS](/thumbs/78/77330968.jpg) The Separation of 8 -THC, 9 -THC, and Their Enantiomers by UPC 2 Using Trefoil Chiral Columns Jacquelyn Runco, Andrew Aubin, and Catharine Layton Waters Corporation, Milford, MA, USA APPLICATION BENEFITS
The Separation of 8 -THC, 9 -THC, and Their Enantiomers by UPC 2 Using Trefoil Chiral Columns Jacquelyn Runco, Andrew Aubin, and Catharine Layton Waters Corporation, Milford, MA, USA APPLICATION BENEFITS
International Journal of Pharma and Bio Sciences
 International Journal of Pharma and Bio Sciences RESEARCH ARTICLE PHARMACEUTICAL ANALYSIS DEVELOPMENT AND VALIDATION OF LIQUID CHROMATOGRAPHIC METHOD FOR ESTIMATION OF ESCITALOPRAM OXALATE IN TABLET DOSAGE
International Journal of Pharma and Bio Sciences RESEARCH ARTICLE PHARMACEUTICAL ANALYSIS DEVELOPMENT AND VALIDATION OF LIQUID CHROMATOGRAPHIC METHOD FOR ESTIMATION OF ESCITALOPRAM OXALATE IN TABLET DOSAGE
Separation of Macrocyclic Lactones (Avermectins) on FLARE C18 MM & FLARE C18+ Columns
 Separation of Macrocyclic Lactones (Avermectins) on FLARE C8 MM & FLARE C8+ Columns Introduction Diamond Analytics Technical Note: T05- Avermectins are a series of 6-membered macrocyclic lactone derivatives
Separation of Macrocyclic Lactones (Avermectins) on FLARE C8 MM & FLARE C8+ Columns Introduction Diamond Analytics Technical Note: T05- Avermectins are a series of 6-membered macrocyclic lactone derivatives
Residue Monograph prepared by the meeting of the Joint FAO/WHO Expert Committee on Food Additives (JECFA), 82 nd meeting 2016.
 Residue Monograph prepared by the meeting of the Joint FAO/WHO Expert Committee on Food Additives (JECFA), 82 nd meeting 2016 Aspartame This monograph was also published in: Compendium of Food Additive
Residue Monograph prepared by the meeting of the Joint FAO/WHO Expert Committee on Food Additives (JECFA), 82 nd meeting 2016 Aspartame This monograph was also published in: Compendium of Food Additive
Using Hydrophilic Interaction Chromatography (HILIC) for the Retention of Highly Polar Analytes
 Using Hydrophilic Interaction Chromatography (HILIC) for the Retention of Highly Polar Analytes Eric S. Grumbach,, Diane M. Diehl and Jeffrey R. Mazzeo 2003 Waters Corp. Outline Introduction HILIC Definition
Using Hydrophilic Interaction Chromatography (HILIC) for the Retention of Highly Polar Analytes Eric S. Grumbach,, Diane M. Diehl and Jeffrey R. Mazzeo 2003 Waters Corp. Outline Introduction HILIC Definition
John P. McCauley and Rui Chen Waters Corporation, Milford, MA, USA INTRODUCTION APPLICATION BENEFITS WATERS SOLUTIONS KEY WORDS
 Enantiomeric and diastereomeric separations of fragrance and essential oil components using the ACQUITY UPC 2 System with ACQUITY UPC 2 Trefoil Columns John P. McCauley and Rui Chen Waters Corporation,
Enantiomeric and diastereomeric separations of fragrance and essential oil components using the ACQUITY UPC 2 System with ACQUITY UPC 2 Trefoil Columns John P. McCauley and Rui Chen Waters Corporation,
Tenofovir disoproxil fumarate (Tenofoviri disoproxili fumaras)
 C 19 H 30 N 5 O 10 P. C 4 H 4 O 4 Relative molecular mass. 635.5. Chemical names. bis(1-methylethyl) 5-{[(1R)-2-(6-amino-9H-purin-9-yl)-1-methylethoxy]methyl}-5-oxo-2,4,6,8-tetraoxa-5-λ 5 - phosphanonanedioate
C 19 H 30 N 5 O 10 P. C 4 H 4 O 4 Relative molecular mass. 635.5. Chemical names. bis(1-methylethyl) 5-{[(1R)-2-(6-amino-9H-purin-9-yl)-1-methylethoxy]methyl}-5-oxo-2,4,6,8-tetraoxa-5-λ 5 - phosphanonanedioate
F. Al-Rimawi* Faculty of Science and Technology, Al-Quds University, P.O. Box 20002, East Jerusalem. Abstract
 JJC Jordan Journal of Chemistry Vol. 4 No.4, 2009, pp. 357-365 Development and Validation of Analytical Method for Fluconazole and Fluconazole Related Compounds (A, B, and C) in Capsule Formulations by
JJC Jordan Journal of Chemistry Vol. 4 No.4, 2009, pp. 357-365 Development and Validation of Analytical Method for Fluconazole and Fluconazole Related Compounds (A, B, and C) in Capsule Formulations by
Comparison of a UPLC Method across Multiple UHPLC Systems
 Comparison of a UPLC Method across Multiple UHPLC Systems Tanya Jenkins Waters Corporation, Milford, MA, U.S. INTRODUCTION In 2004, Waters introduced the ACQUITY UPLC System. Since this launch, many liquid
Comparison of a UPLC Method across Multiple UHPLC Systems Tanya Jenkins Waters Corporation, Milford, MA, U.S. INTRODUCTION In 2004, Waters introduced the ACQUITY UPLC System. Since this launch, many liquid
CE Columns and Accessories
 CE Columns and Accessories Complete Selection of Stationary Phases Sepax Nanofilm Capillary-Polyacrylamide Coatings Sepax Nanofilm Capillary-Poly(ethylene glycol) Coatings.. 4 Sepax Nanofilm Capillary-PEG/PAAm
CE Columns and Accessories Complete Selection of Stationary Phases Sepax Nanofilm Capillary-Polyacrylamide Coatings Sepax Nanofilm Capillary-Poly(ethylene glycol) Coatings.. 4 Sepax Nanofilm Capillary-PEG/PAAm
TENOFOVIR TABLETS: Final text for addition to The International Pharmacopoeia (June 2010)
 June 2010 TENOFOVIR TABLETS: Final text for addition to The International Pharmacopoeia (June 2010) This monograph was adopted at the Forty-fourth WHO Expert Committee on Specifications for Pharmaceutical
June 2010 TENOFOVIR TABLETS: Final text for addition to The International Pharmacopoeia (June 2010) This monograph was adopted at the Forty-fourth WHO Expert Committee on Specifications for Pharmaceutical
CYCLOSERINI CAPSULAE - CYCLOSERINE CAPSULES (AUGUST 2015)
 August 2015 Document for comment 1 2 3 4 5 CYCLOSERINI CAPSULAE - CYCLOSERINE CAPSULES DRAFT PROPOSAL FOR THE INTERNATIONAL PHARMACOPOEIA (AUGUST 2015) DRAFT FOR COMMENT 6 Should you have any comments
August 2015 Document for comment 1 2 3 4 5 CYCLOSERINI CAPSULAE - CYCLOSERINE CAPSULES DRAFT PROPOSAL FOR THE INTERNATIONAL PHARMACOPOEIA (AUGUST 2015) DRAFT FOR COMMENT 6 Should you have any comments
Separation of Vitamin D and Vitamin D Metabolites on FLARE C18 MM (Mixed Mode) HPLC Column
 Diamond Analytics Technical Note: T131-1 Separation of Vitamin D and Vitamin D Metabolites on FLARE C1 MM (Mixed Mode) HPLC Column Introduction In this technical note, the versatility of the diamond-based
Diamond Analytics Technical Note: T131-1 Separation of Vitamin D and Vitamin D Metabolites on FLARE C1 MM (Mixed Mode) HPLC Column Introduction In this technical note, the versatility of the diamond-based
LC Columns with Liquid Separation Cell Technology
 Obelisc LC Columns with Liquid Separation Cell Technology Obelisc HPLC Columns - Liquid Separation Cell Technology Introduction Obelisc HPLC columns are the latest, innovative columns from SIELC Technologies,
Obelisc LC Columns with Liquid Separation Cell Technology Obelisc HPLC Columns - Liquid Separation Cell Technology Introduction Obelisc HPLC columns are the latest, innovative columns from SIELC Technologies,
Impact of Reversed-Phase Chiral Chromatography on the LC-MS Analysis of Drugs in Biological Fluids
 Impact of Reversed-Phase Chiral Chromatography on the LC-MS Analysis of Drugs in Biological Fluids David S. Bell, Carmen T. Santasania, Jennifer Claus, Wayne K. Way, and Craig R. Aurand Supelco, Div. of
Impact of Reversed-Phase Chiral Chromatography on the LC-MS Analysis of Drugs in Biological Fluids David S. Bell, Carmen T. Santasania, Jennifer Claus, Wayne K. Way, and Craig R. Aurand Supelco, Div. of
Chiral Multicolumn Method Development on the Agilent 1260 Infinity II SFC System
 Chiral Multicolumn Method Development on the Agilent 126 Infinity II SFC System Application Note Small Molecule Pharmaceuticals Author Edgar Naegele Agilent Technologies, Inc. Waldbronn, Germany Abstract
Chiral Multicolumn Method Development on the Agilent 126 Infinity II SFC System Application Note Small Molecule Pharmaceuticals Author Edgar Naegele Agilent Technologies, Inc. Waldbronn, Germany Abstract
Introduction. and aryl-α-amino ketones, 11
 Enantiomeric Resolution of γ-amino Acids Bull. Korean Chem. Soc. 2011, Vol. 32, No. 8 3017 DOI 10.5012/bkcs.2011.32.8.3017 Liquid Chromatographic Resolution of Vigabatrin and Its Analogue γ-amino Acids
Enantiomeric Resolution of γ-amino Acids Bull. Korean Chem. Soc. 2011, Vol. 32, No. 8 3017 DOI 10.5012/bkcs.2011.32.8.3017 Liquid Chromatographic Resolution of Vigabatrin and Its Analogue γ-amino Acids
ABACAVIR SULFATE Proposal for revision of The International Pharmacopoeia (August 2012)
 August 2012 RESTRICTED ABACAVIR SULFATE Proposal for revision of The International Pharmacopoeia (August 2012) Draft for comment This document was provided by a quality control expert. Should you have
August 2012 RESTRICTED ABACAVIR SULFATE Proposal for revision of The International Pharmacopoeia (August 2012) Draft for comment This document was provided by a quality control expert. Should you have
World Journal of Pharmaceutical Research
 World Journal of Pharmaceutical ReseaRch Volume 3, Issue 3, 4527-4535. Research Article ISSN 2277 715 DEVELOPMENT AND VALIDATION OF STABILITY INDICATING HPLC METHOD FOR ESTIMATION OF RAMOSETRON Zarana
World Journal of Pharmaceutical ReseaRch Volume 3, Issue 3, 4527-4535. Research Article ISSN 2277 715 DEVELOPMENT AND VALIDATION OF STABILITY INDICATING HPLC METHOD FOR ESTIMATION OF RAMOSETRON Zarana
Increasing resolution using longer columns while maintaining analysis time Advantages of the wide power range of the Agilent 1290 Infinity LC System
 Increasing resolution using longer columns while maintaining analysis time Advantages of the wide power range of the Agilent 129 Infinity LC System Application Note Pharmaceutical and Chemical Analysis
Increasing resolution using longer columns while maintaining analysis time Advantages of the wide power range of the Agilent 129 Infinity LC System Application Note Pharmaceutical and Chemical Analysis
ASSAY AND IMPURITY METHOD FOR DURACOR TABLETS BY HPLC
 ASSAY AND IMPURITY METHOD FOR DURACOR TABLETS BY HPLC METHOD APPROVALS Norvin Pharma Inc. Author Analytical Laboratory Approver Analytical Laboratory Group Leader Approver Manager Quality Control Chemistry
ASSAY AND IMPURITY METHOD FOR DURACOR TABLETS BY HPLC METHOD APPROVALS Norvin Pharma Inc. Author Analytical Laboratory Approver Analytical Laboratory Group Leader Approver Manager Quality Control Chemistry
Analytical and Preparative SFC Columns
 Analytical and Preparative SFC Columns Best Performance for Supercritical Fluid Chromatography Sepax SFC-Cyano... Sepax SFC-Amino... Sepax SFC-Pyridine... Sepax SFC-SCX... 5 Sepax SFC-Diol... 6 Sepax SFC-Silica...
Analytical and Preparative SFC Columns Best Performance for Supercritical Fluid Chromatography Sepax SFC-Cyano... Sepax SFC-Amino... Sepax SFC-Pyridine... Sepax SFC-SCX... 5 Sepax SFC-Diol... 6 Sepax SFC-Silica...
[APPLICATION NOTE] DETERMINATION OF AMINO ACIDS IN BEERS USING THE UPLC AMINO ACID ANALYSIS SOLUTION
![[APPLICATION NOTE] DETERMINATION OF AMINO ACIDS IN BEERS USING THE UPLC AMINO ACID ANALYSIS SOLUTION [APPLICATION NOTE] DETERMINATION OF AMINO ACIDS IN BEERS USING THE UPLC AMINO ACID ANALYSIS SOLUTION](/thumbs/85/93034719.jpg) DETERMINATION OF AMINO ACIDS IN BEERS USING THE UPLC AMINO ACID ANALYSIS SOLUTION Mark E. Benvenuti and Paula Hong Waters Corporation, Milford, MA USA INTRODUCTION: Beer is a complex matrix consisting
DETERMINATION OF AMINO ACIDS IN BEERS USING THE UPLC AMINO ACID ANALYSIS SOLUTION Mark E. Benvenuti and Paula Hong Waters Corporation, Milford, MA USA INTRODUCTION: Beer is a complex matrix consisting
Pelagia Research Library
 Available online at www.pelagiaresearchlibrary.com Der Pharmacia Sinica, 2014, 5(5):91-98 ISSN: 0976-8688 CODEN (USA): PSHIBD A novel RP-HPLC method development and validation of Perindopril Erbumine in
Available online at www.pelagiaresearchlibrary.com Der Pharmacia Sinica, 2014, 5(5):91-98 ISSN: 0976-8688 CODEN (USA): PSHIBD A novel RP-HPLC method development and validation of Perindopril Erbumine in
ISSN: ; CODEN ECJHAO E-Journal of Chemistry 2011, 8(3),
 ISSN: 0973-4945; CODEN ECJHAO E- Chemistry http://www.e-journals.net 2011, 8(3), 1275-1279 Simultaneous Determination of Paracetamol, Phenylephrine Hydrochloride, Oxolamine Citrate and Chlorpheniramine
ISSN: 0973-4945; CODEN ECJHAO E- Chemistry http://www.e-journals.net 2011, 8(3), 1275-1279 Simultaneous Determination of Paracetamol, Phenylephrine Hydrochloride, Oxolamine Citrate and Chlorpheniramine
SeQuant ZIC -HILIC For all who expect more...
 SeQuant ZIC -ILIC For all who expect more... The better choice for PLC and LC/MS of all types of polar and hydrophilic compounds EMD Millipore is a division of Merck KGaA, Darmstadt, Germany Poor retention
SeQuant ZIC -ILIC For all who expect more... The better choice for PLC and LC/MS of all types of polar and hydrophilic compounds EMD Millipore is a division of Merck KGaA, Darmstadt, Germany Poor retention
SeQuant ZIC -HILIC For all who expect more...
 SeQuant ZIC -ILIC For all who expect more... The better choice for PLC and LC/MS of all types of polar and hydrophilic compounds EMD Millipore Corp. is a subsidiary of Merck KGaA, Darmstadt, Germany Poor
SeQuant ZIC -ILIC For all who expect more... The better choice for PLC and LC/MS of all types of polar and hydrophilic compounds EMD Millipore Corp. is a subsidiary of Merck KGaA, Darmstadt, Germany Poor
Two chiral centres (diastereoisomers)
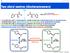 1 Two chiral centres (diastereoisomers) Mirror N S N Two enantiomers differ by absolute configuration N N 2 N 2 N A molecule with 1 stereogenic centre exists as 2 stereoisomers or enantiomers Enantiomers
1 Two chiral centres (diastereoisomers) Mirror N S N Two enantiomers differ by absolute configuration N N 2 N 2 N A molecule with 1 stereogenic centre exists as 2 stereoisomers or enantiomers Enantiomers
ACE. For increased polar retention and alternative selectivity. Alternative selectivity for method development
 ACE C8-Amide For increased polar retention and alternative selectivity Alternative selectivity for method development Improved separations with polar, acidic, basic and phenolic compounds High efficiency
ACE C8-Amide For increased polar retention and alternative selectivity Alternative selectivity for method development Improved separations with polar, acidic, basic and phenolic compounds High efficiency
IJPAR Vol.3 Issue 4 Oct-Dec-2014 Journal Home page:
 IJPAR Vol.3 Issue 4 Oct-Dec-2014 Journal Home page: ISSN: 2320-2831 Research article Open Access Method development and validation of tenofovir disoproxil fumerate and emtricitabine in combined tablet
IJPAR Vol.3 Issue 4 Oct-Dec-2014 Journal Home page: ISSN: 2320-2831 Research article Open Access Method development and validation of tenofovir disoproxil fumerate and emtricitabine in combined tablet
Analysis of Rosuvastatin in Dried Blood Spot and Plasma Using ACQUITY UPLC with 2D Technology
 Analysis of Rosuvastatin in Dried Blood Spot and Plasma Using ACQUITY UPLC with 2D Technology Claude Mallet, 1 Jennifer Simeone, 2 Paul Rainville 3 1 Workflow Integration Group, Separations Technologies,
Analysis of Rosuvastatin in Dried Blood Spot and Plasma Using ACQUITY UPLC with 2D Technology Claude Mallet, 1 Jennifer Simeone, 2 Paul Rainville 3 1 Workflow Integration Group, Separations Technologies,
RP-HPLC Analysis of Temozolomide in Pharmaceutical Dosage Forms
 Asian Journal of Chemistry Vol. 22, No. 7 (2010), 5067-5071 RP-HPLC Analysis of Temozolomide in Pharmaceutical Dosage Forms A. LAKSHMANA RAO*, G. TARAKA RAMESH and J.V.L.N.S. RAO Department of Pharmaceutical
Asian Journal of Chemistry Vol. 22, No. 7 (2010), 5067-5071 RP-HPLC Analysis of Temozolomide in Pharmaceutical Dosage Forms A. LAKSHMANA RAO*, G. TARAKA RAMESH and J.V.L.N.S. RAO Department of Pharmaceutical
RITONAVIRI COMPRESSI RITONAVIR TABLETS. Final text for addition to The International Pharmacopoeia (July 2012)
 July 2012 RITONAVIRI COMPRESSI RITONAVIR TABLETS Final text for addition to The International Pharmacopoeia (July 2012) This monograph was adopted at the Forty-sixth WHO Expert Committee on Specifications
July 2012 RITONAVIRI COMPRESSI RITONAVIR TABLETS Final text for addition to The International Pharmacopoeia (July 2012) This monograph was adopted at the Forty-sixth WHO Expert Committee on Specifications
ARTESUNATE TABLETS: Final text for revision of The International Pharmacopoeia (December 2009) ARTESUNATI COMPRESSI ARTESUNATE TABLETS
 December 2009 ARTESUNATE TABLETS: Final text for revision of The International Pharmacopoeia (December 2009) This monograph was adopted at the Forty-fourth WHO Expert Committee on Specifications for Pharmaceutical
December 2009 ARTESUNATE TABLETS: Final text for revision of The International Pharmacopoeia (December 2009) This monograph was adopted at the Forty-fourth WHO Expert Committee on Specifications for Pharmaceutical
Analysis of Counterfeit Antidiabetic Drugs by UHPLC with the Agilent 1220 Infinity Mobile LC
 Analysis of Counterfeit Antidiabetic Drugs by UPLC with the Agilent 1 Infinity Mobile LC Application Note Small Molecule Pharmaceuticals & Generics Author Sonja Schneider Agilent Technologies, Inc. Waldbronn,
Analysis of Counterfeit Antidiabetic Drugs by UPLC with the Agilent 1 Infinity Mobile LC Application Note Small Molecule Pharmaceuticals & Generics Author Sonja Schneider Agilent Technologies, Inc. Waldbronn,
A Validated Chiral Liquid Chromatographic Method for The Enantiomeric Separation of Dapoxetine Hydrochloride
 Received on 15/05/2012; Revised on 22/05/2012; Accepted on 09/06/2012 A Validated Chiral Liquid Chromatographic thod for The Enantiomeric Separation of Dapoxetine Hydrochloride T.Rohith 1 and S. Ananda
Received on 15/05/2012; Revised on 22/05/2012; Accepted on 09/06/2012 A Validated Chiral Liquid Chromatographic thod for The Enantiomeric Separation of Dapoxetine Hydrochloride T.Rohith 1 and S. Ananda
METHOD DEVELOPMENT AND VALIDATION BY RP-HPLC FOR ESTIMATION OF ZOLPIDEM TARTARATE
 WORLD JOURNAL OF PHARMACY AND PHARMACEUTICAL SCIENCES Ramalakshmi et al. SJIF Impact Factor 6.647 Volume 7, Issue 2, 1010-1018 Research Article ISSN 2278 4357 METHOD DEVELOPMENT AND VALIDATION BY RP-HPLC
WORLD JOURNAL OF PHARMACY AND PHARMACEUTICAL SCIENCES Ramalakshmi et al. SJIF Impact Factor 6.647 Volume 7, Issue 2, 1010-1018 Research Article ISSN 2278 4357 METHOD DEVELOPMENT AND VALIDATION BY RP-HPLC
Removal of Triton X-100 from Plasma Samples Using Mixed-Mode Solid Phase Extraction (SPE)
 Removal of Triton X- from Plasma Samples Using Mixed-Mode Solid Phase Extraction (SPE) Jonathan P. Danaceau, Erin Chambers, and Kenneth J. Fountain Waters Corporation, 34 Maple Street, Milford, MA USA
Removal of Triton X- from Plasma Samples Using Mixed-Mode Solid Phase Extraction (SPE) Jonathan P. Danaceau, Erin Chambers, and Kenneth J. Fountain Waters Corporation, 34 Maple Street, Milford, MA USA
Sample Preparation Techniques for Biological Matrices: Finding the right balance to achieve optimal results
 Sample Preparation Techniques for Biological Matrices: Finding the right balance to achieve optimal results Presenter: Diana Wang Applications Engineer SPP-Support@agilent.com 1 Today s Agenda - Why sample
Sample Preparation Techniques for Biological Matrices: Finding the right balance to achieve optimal results Presenter: Diana Wang Applications Engineer SPP-Support@agilent.com 1 Today s Agenda - Why sample
Rapid and sensitive UHPLC screening for water soluble vitamins in sports beverages
 APPLICATION NOTE 21671 Rapid and sensitive UHPLC screening for water soluble vitamins in sports beverages Authors Jon Bardsley, Thermo Fisher Scientific, Runcorn, UK Keywords Vanquish Flex, Acclaim PolarAdvantage
APPLICATION NOTE 21671 Rapid and sensitive UHPLC screening for water soluble vitamins in sports beverages Authors Jon Bardsley, Thermo Fisher Scientific, Runcorn, UK Keywords Vanquish Flex, Acclaim PolarAdvantage
Impurity Profiling of Carbamazepine by HPLC/UV
 Application Note: 52049 Impurity Profiling of Carbamazepine by HPLC/UV Terry Zhang, Guifeng Jiang, Thermo Fisher Scientific, San Jose, CA, USA Key Words Accela Hypersil GOLD Carbamazepine Drug Analysis
Application Note: 52049 Impurity Profiling of Carbamazepine by HPLC/UV Terry Zhang, Guifeng Jiang, Thermo Fisher Scientific, San Jose, CA, USA Key Words Accela Hypersil GOLD Carbamazepine Drug Analysis
Student Handout. This experiment allows you to explore the properties of chiral molecules. You have
 Student Handout This experiment allows you to explore the properties of chiral molecules. You have learned that some compounds exist as enantiomers non-identical mirror images, such as your left and right
Student Handout This experiment allows you to explore the properties of chiral molecules. You have learned that some compounds exist as enantiomers non-identical mirror images, such as your left and right
GABRIEL HANCU 1*, CLAUDIA SĂMĂRGHIŢAN 1, AURA RUSU 1, ELEONORA MIRCIA 2
 SOTALOL CHIRAL SEPARATION BY CAPILLARY ELECTROPHORESIS GABRIEL HANCU 1*, CLAUDIA SĂMĂRGHIŢAN 1, AURA RUSU 1, ELEONORA MIRCIA 2 1 Department of Pharmaceutical Chemistry, Faculty of Pharmacy, University
SOTALOL CHIRAL SEPARATION BY CAPILLARY ELECTROPHORESIS GABRIEL HANCU 1*, CLAUDIA SĂMĂRGHIŢAN 1, AURA RUSU 1, ELEONORA MIRCIA 2 1 Department of Pharmaceutical Chemistry, Faculty of Pharmacy, University
LEVONORGESTREL AND ETHINYLESTRADIOL TABLETS. (January 2012) DRAFT FOR COMMENT
 January 2012 RESTRICTED DRAFT PROPOSAL FOR The International Pharmacopoeia LEVONORGESTREL AND ETHINYLESTRADIOL TABLETS (January 2012) DRAFT FOR COMMENT This document was provided by a quality control expert.
January 2012 RESTRICTED DRAFT PROPOSAL FOR The International Pharmacopoeia LEVONORGESTREL AND ETHINYLESTRADIOL TABLETS (January 2012) DRAFT FOR COMMENT This document was provided by a quality control expert.
ARTENIMOLUM ARTENIMOL. Adopted revised text for addition to The International Pharmacopoeia
 February 2012 ARTENIMOLUM ARTENIMOL Adopted revised text for addition to The International Pharmacopoeia This monograph was adopted at the Forty-sixth WHO Expert Committee on Specifications for Pharmaceutical
February 2012 ARTENIMOLUM ARTENIMOL Adopted revised text for addition to The International Pharmacopoeia This monograph was adopted at the Forty-sixth WHO Expert Committee on Specifications for Pharmaceutical
CORESTA RECOMMENDED METHOD NÄ 9
 CORESTA RECOMMENDED METHOD NÄ 9 DETERMINATION OF NICOTINE IN CIGARETTE FILTERS BY GAS CHROMATOGRAPHIC ANALYSIS (April 2009) 0. INTRODUCTION In 2001 the CORESTA Routine Analytical Chemistry Sub-Group was
CORESTA RECOMMENDED METHOD NÄ 9 DETERMINATION OF NICOTINE IN CIGARETTE FILTERS BY GAS CHROMATOGRAPHIC ANALYSIS (April 2009) 0. INTRODUCTION In 2001 the CORESTA Routine Analytical Chemistry Sub-Group was
Application Note. Agilent Application Solution Analysis of ascorbic acid, citric acid and benzoic acid in orange juice. Author. Abstract.
 Agilent Application Solution Analysis of ascorbic acid, citric acid and benzoic acid in orange juice Application Note Author Food Syed Salman Lateef Agilent Technologies, Inc. Bangalore, India 8 6 4 2
Agilent Application Solution Analysis of ascorbic acid, citric acid and benzoic acid in orange juice Application Note Author Food Syed Salman Lateef Agilent Technologies, Inc. Bangalore, India 8 6 4 2
2D-LC as an Automated Desalting Tool for MSD Analysis
 2D-LC as an Automated Desalting Tool for MSD Analysis Direct Mass Selective Detection of a Pharmaceutical Peptide from an MS-Incompatible USP Method Application Note Biologics and Biosimilars Author Sonja
2D-LC as an Automated Desalting Tool for MSD Analysis Direct Mass Selective Detection of a Pharmaceutical Peptide from an MS-Incompatible USP Method Application Note Biologics and Biosimilars Author Sonja
Analysis of Common Sweeteners and Additives in Beverages with the PerkinElmer Flexar FX-15 System Equipped with a PDA Detector
 application Note Liquid Chromatography Author Njies Pedjie PerkinElmer, Inc. Shelton, CT 06484 USA Analysis of Common Sweeteners and Additives in Beverages with the PerkinElmer Flexar FX-15 System Equipped
application Note Liquid Chromatography Author Njies Pedjie PerkinElmer, Inc. Shelton, CT 06484 USA Analysis of Common Sweeteners and Additives in Beverages with the PerkinElmer Flexar FX-15 System Equipped
SIMULTANEOUS ESTIMATION OF VALSARTAN AND HYDROCHLOROTHIAZIDE IN TABLETS BY RP-HPLC METHOD
 170 Original Article SIMULTANEOUS ESTIMATION OF VALSARTAN AND HYDROCHLOROTHIAZIDE IN TABLETS BY RP-HPLC METHOD *Lakshmana Rao A, 1 Bhaskara Raju V *V.V. Institute of Pharmaceutical Sciences, Gudlavalleru,
170 Original Article SIMULTANEOUS ESTIMATION OF VALSARTAN AND HYDROCHLOROTHIAZIDE IN TABLETS BY RP-HPLC METHOD *Lakshmana Rao A, 1 Bhaskara Raju V *V.V. Institute of Pharmaceutical Sciences, Gudlavalleru,
A New HILIC/RP Mixed-Mode Column and Its Applications in Surfactant Analysis
 A New HILIC/RP Mixed-Mode Column and Its Applications in Surfactant Analysis X. Liu, C. Pohl, Dionex Corporation, Sunnyvale, CA, USA ABSTRACT Although reversed-phase (RP) silica columns (e.g., C18 and
A New HILIC/RP Mixed-Mode Column and Its Applications in Surfactant Analysis X. Liu, C. Pohl, Dionex Corporation, Sunnyvale, CA, USA ABSTRACT Although reversed-phase (RP) silica columns (e.g., C18 and
Development and Validation for Simultaneous Estimation of Sitagliptin and Metformin in Pharmaceutical Dosage Form using RP-HPLC Method
 International Journal of PharmTech Research CODEN (USA): IJPRIF ISSN : 0974-4304 Vol.5, No.4, pp 1736-1744, Oct-Dec 2013 Development and Validation for Simultaneous Estimation of Sitagliptin and Metformin
International Journal of PharmTech Research CODEN (USA): IJPRIF ISSN : 0974-4304 Vol.5, No.4, pp 1736-1744, Oct-Dec 2013 Development and Validation for Simultaneous Estimation of Sitagliptin and Metformin
FLUNITRAZEPAM Latest Revision: January 24, 2006
 FLUNITRAZEPAM Latest Revision: January 24, 2006 1. SYNONYMS CFR: Flunitrazepam CAS #: 1622-62-4 Other Names: 5-(2-Fluorophenyl)-1,3-dihydro-1-methyl-7-nitro-2H- 1,4-benzodiazepin-2-one Flunitrax Hipnosedon
FLUNITRAZEPAM Latest Revision: January 24, 2006 1. SYNONYMS CFR: Flunitrazepam CAS #: 1622-62-4 Other Names: 5-(2-Fluorophenyl)-1,3-dihydro-1-methyl-7-nitro-2H- 1,4-benzodiazepin-2-one Flunitrax Hipnosedon
Pelagia Research Library
 Available online at www.pelagiaresearchlibrary.com Der Pharmacia Sinica, 2015, 6(1):6-10 ISSN: 0976-8688 CODEN (USA): PSHIBD Validated RP-HPLC method for simultaneous estimation of metformin hydrochloride
Available online at www.pelagiaresearchlibrary.com Der Pharmacia Sinica, 2015, 6(1):6-10 ISSN: 0976-8688 CODEN (USA): PSHIBD Validated RP-HPLC method for simultaneous estimation of metformin hydrochloride
INTERNATIONAL PHARMACOPOEIA MONOGRAPH ON LAMIVUDINE TABLETS
 RESTRICTED INTERNATIONAL PHARMACOPOEIA MONOGRAPH ON LAMIVUDINE TABLETS DRAFT FOR COMMENT Please address any comments you may have on this document, by 12 July 2006, to Dr S. Kopp, Quality Assurance and
RESTRICTED INTERNATIONAL PHARMACOPOEIA MONOGRAPH ON LAMIVUDINE TABLETS DRAFT FOR COMMENT Please address any comments you may have on this document, by 12 July 2006, to Dr S. Kopp, Quality Assurance and
Am I getting the very best value from my UHPLC analyses?
 Am I getting the very best value from my UHPLC analyses? Stephen Luke LC Columns Product Manager 1 Primary reasons for UHPLC use Very fast Very high resolution Column 2.1 x 50 mm Column 2.1 x 150 mm Run
Am I getting the very best value from my UHPLC analyses? Stephen Luke LC Columns Product Manager 1 Primary reasons for UHPLC use Very fast Very high resolution Column 2.1 x 50 mm Column 2.1 x 150 mm Run
CHAPTER INTRODUCTION OF DOSAGE FORM AND LITERATURE REVIEW
 132 CHAPTER 6 DEVELOPMENT AND VALIDATION OF A STABILITY-INDICATING RP-HPLC METHOD FOR SIMULTANEOUS DETERMINATION OF PARACETAMOL, TRAMADOL HYDROCHLORIDE AND DOMPERIDONE IN A COMBINED DOSAGE FORM 6.1 INTRODUCTION
132 CHAPTER 6 DEVELOPMENT AND VALIDATION OF A STABILITY-INDICATING RP-HPLC METHOD FOR SIMULTANEOUS DETERMINATION OF PARACETAMOL, TRAMADOL HYDROCHLORIDE AND DOMPERIDONE IN A COMBINED DOSAGE FORM 6.1 INTRODUCTION
Journal of Chemical and Pharmaceutical Research, 2017, 9(9): Research Article
 Available online www.jocpr.com Journal of Chemical and Pharmaceutical Research, 2017, 9(9):70-80 Research Article ISSN : 0975-7384 CODEN(USA) : JCPRC5 Development and Validation of Stability Indicating
Available online www.jocpr.com Journal of Chemical and Pharmaceutical Research, 2017, 9(9):70-80 Research Article ISSN : 0975-7384 CODEN(USA) : JCPRC5 Development and Validation of Stability Indicating
Separation of Saccharides Using TSKgel Amide-80, a Packing Material for High-Performance Normal Phase Partition Chromatography (1) Table of Contents
 No. 055 SEPARATION REPORT Separation of Saccharides Using TSKgel Amide-80, a Packing Material for High-Performance Normal Phase Partition Chromatography (1) Table of Contents 1. Introduction 1 2. Elution
No. 055 SEPARATION REPORT Separation of Saccharides Using TSKgel Amide-80, a Packing Material for High-Performance Normal Phase Partition Chromatography (1) Table of Contents 1. Introduction 1 2. Elution
Rapid and sensitive UHPLC screening of additives in carbonated beverages with a robust organic acid column
 APPLICATION NOTE 21673 Rapid and sensitive UHPLC screening of additives in carbonated beverages with a robust organic acid column Authors Aaron Lamb and Brian King, Thermo Fisher Scientific, Runcorn, UK
APPLICATION NOTE 21673 Rapid and sensitive UHPLC screening of additives in carbonated beverages with a robust organic acid column Authors Aaron Lamb and Brian King, Thermo Fisher Scientific, Runcorn, UK
SEPARATION OF ENANTIOMERS BY CHIRALLY MODIFIED MEMBRANES
 XXIII ARS SEPARATORIA Toruń, Poland 2008 41 SEPARATION OF ENANTIOMERS BY CHIRALLY MODIFIED MEMBRANES Izabela KOTER Nicolaus Copernicus University, Faculty of Chemistry, 7 Gagarin St., 87-100 Torun, Poland
XXIII ARS SEPARATORIA Toruń, Poland 2008 41 SEPARATION OF ENANTIOMERS BY CHIRALLY MODIFIED MEMBRANES Izabela KOTER Nicolaus Copernicus University, Faculty of Chemistry, 7 Gagarin St., 87-100 Torun, Poland
vii LIST OF TABLES TABLE NO DESCRIPTION PAGE 1.1 System Suitability Parameters and Recommendations Acidic and Alkaline Hydrolysis 15
 vii LIST OF TABLES TABLE NO DESCRIPTION PAGE CHAPTER- 1 1.1 System Suitability Parameters and Recommendations 07 1.2 Acidic and Alkaline Hydrolysis 15 1.3 Oxidative Degradation Study 16 1.4 Hydrolysis
vii LIST OF TABLES TABLE NO DESCRIPTION PAGE CHAPTER- 1 1.1 System Suitability Parameters and Recommendations 07 1.2 Acidic and Alkaline Hydrolysis 15 1.3 Oxidative Degradation Study 16 1.4 Hydrolysis
SULFAMETHOXAZOLE AND TRIMETHOPRIM TABLETS Draft proposal for The International Pharmacopoeia (September 2010)
 September 2010 RESTRICTED SULFAMETHOXAZOLE AND TRIMETHOPRIM TABLETS Draft proposal for The International Pharmacopoeia (September 2010) REVISED DRAFT FOR COMMENT This document was provided by a quality
September 2010 RESTRICTED SULFAMETHOXAZOLE AND TRIMETHOPRIM TABLETS Draft proposal for The International Pharmacopoeia (September 2010) REVISED DRAFT FOR COMMENT This document was provided by a quality
DEVELOPMENT AND VALIDATION OF RP-HPLC METHOD FOR QUANTITATIVE ANALYSIS OF TRAMADOL IN PURE AND PHARMACEUTICAL FORMULATIONS
 Int. J. Chem. Sci.: 10(4), 2012, 2039-2047 ISSN 0972-768X www.sadgurupublications.com DEVELOPMENT AND VALIDATION OF RP-HPLC METHOD FOR QUANTITATIVE ANALYSIS OF TRAMADOL IN PURE AND PHARMACEUTICAL FORMULATIONS
Int. J. Chem. Sci.: 10(4), 2012, 2039-2047 ISSN 0972-768X www.sadgurupublications.com DEVELOPMENT AND VALIDATION OF RP-HPLC METHOD FOR QUANTITATIVE ANALYSIS OF TRAMADOL IN PURE AND PHARMACEUTICAL FORMULATIONS
Molecules in the mirror
 Stereoselective analysis Molecules in the mirror Enantiomer separation using capillary electrophoresis Prof. Dr. Gerhard K. E. Scriba Department of Pharmaceutical / Medical Chemistry, Friedrich-Schiller
Stereoselective analysis Molecules in the mirror Enantiomer separation using capillary electrophoresis Prof. Dr. Gerhard K. E. Scriba Department of Pharmaceutical / Medical Chemistry, Friedrich-Schiller
CHAPTER 8 HIGH PERFORMANCE LIQUID CHROMATOGRAPHY (HPLC) ANALYSIS OF PHYTOCHEMICAL CONSTITUENTS OF M. ROXBURGHIANUS AND P. FRATERNUS PLANT EXTRACTS
 CHAPTER 8 HIGH PERFORMANCE LIQUID CHROMATOGRAPHY (HPLC) ANALYSIS OF PHYTOCHEMICAL CONSTITUENTS OF M. ROXBURGHIANUS AND P. FRATERNUS PLANT EXTRACTS CHAPTER 8: HIGH PERFORMANCE LIQUID CHROMATOGRAPHY (HPLC)
CHAPTER 8 HIGH PERFORMANCE LIQUID CHROMATOGRAPHY (HPLC) ANALYSIS OF PHYTOCHEMICAL CONSTITUENTS OF M. ROXBURGHIANUS AND P. FRATERNUS PLANT EXTRACTS CHAPTER 8: HIGH PERFORMANCE LIQUID CHROMATOGRAPHY (HPLC)
Ultra High Performance Liquid Chromatograph. Nexera X2 C196-E079
 Ultra High Performance Liquid Chromatograph Nexera X2 C196-E79 Maximizing the Potential of UHPLC/HPLC Analyses Intelligence Intelligent data solutions, including a new i -PDeA peak calculation method for
Ultra High Performance Liquid Chromatograph Nexera X2 C196-E79 Maximizing the Potential of UHPLC/HPLC Analyses Intelligence Intelligent data solutions, including a new i -PDeA peak calculation method for
Application Note. Authors: C. Ledesma, M. Gibert, J.R. Gibert Ingenieria Analitica S.L. Extracts from various food products
 High-Performance Anion-Exchange Chromatography coupled with Pulsed Electrochemical Detection as a powerful tool to evaluate lactose content in lactose-free labeled products Application Note Authors: C.
High-Performance Anion-Exchange Chromatography coupled with Pulsed Electrochemical Detection as a powerful tool to evaluate lactose content in lactose-free labeled products Application Note Authors: C.
Development, Estimation and Validation of Lisinopril in Bulk and its Pharmaceutical Formulation by HPLC Method
 ISSN: 0973-4945; CODEN ECJAO E- Chemistry http://www.e-journals.net 2012, 9(1), 340-344 Development, Estimation and Validation of Lisinopril in Bulk and its Pharmaceutical Formulation by PLC Method V.
ISSN: 0973-4945; CODEN ECJAO E- Chemistry http://www.e-journals.net 2012, 9(1), 340-344 Development, Estimation and Validation of Lisinopril in Bulk and its Pharmaceutical Formulation by PLC Method V.
New RP - HPLC Method for the Determination of Valproic acid in Human Plasma
 New RP - HPLC Method for the Determination of Valproic acid in Human Plasma C.Venkata Nagendra Prasad 1, Ch.Santhosh Kumari a, B.Srinivasa Reddy 1 and Prof. J. Sriramulu 2 1 Sree Dattha Institute of Pharmacy,
New RP - HPLC Method for the Determination of Valproic acid in Human Plasma C.Venkata Nagendra Prasad 1, Ch.Santhosh Kumari a, B.Srinivasa Reddy 1 and Prof. J. Sriramulu 2 1 Sree Dattha Institute of Pharmacy,
A New Stability-Indicating and Validated RP-HPLC Method for the Estimation of Liraglutide in Bulk and Pharmaceutical Dosage Forms
 OPEN ACCESS Eurasian Journal of Analytical Chemistry ISSN: 1306-3057 2017 12(2):31-44 DOI 10.12973/ejac.2017.00152a A New Stability-Indicating and Validated RP-HPLC Method for the Estimation of Liraglutide
OPEN ACCESS Eurasian Journal of Analytical Chemistry ISSN: 1306-3057 2017 12(2):31-44 DOI 10.12973/ejac.2017.00152a A New Stability-Indicating and Validated RP-HPLC Method for the Estimation of Liraglutide
Lux. Five Distinct Polysaccharide Phases to Resolve 92 % of Your Enantiomers* Lux Cellulose-1. Lux Cellulose-3. Lux Cellulose-2.
 Five Distinct Polysaccharide Phases to Resolve 9 % of Your Enantiomers* If Lux analytical columns ( 4.6 mm ID) do not provide at least an equivalent or better separation as compared to a competing column
Five Distinct Polysaccharide Phases to Resolve 9 % of Your Enantiomers* If Lux analytical columns ( 4.6 mm ID) do not provide at least an equivalent or better separation as compared to a competing column
Sepax Technologies, Inc.
 Sepax Technologies, Inc. Sepax Technologies, Inc. develops and manufactures products in the area of chemical and biological separations, biosurfaces and proteomics. Sepax product portfolio includes ) liquid
Sepax Technologies, Inc. Sepax Technologies, Inc. develops and manufactures products in the area of chemical and biological separations, biosurfaces and proteomics. Sepax product portfolio includes ) liquid
Development and Validation of a Simultaneous HPLC Method for Quantification of Amlodipine Besylate and Metoprolol Tartrate in Tablets
 Journal of PharmaSciTech 0; ():- Research Article Development and Validation of a Simultaneous HPLC Method for Quantification of Amlodipine Besylate and Metoprolol Tartrate in Tablets * Sayyed Hussain,
Journal of PharmaSciTech 0; ():- Research Article Development and Validation of a Simultaneous HPLC Method for Quantification of Amlodipine Besylate and Metoprolol Tartrate in Tablets * Sayyed Hussain,
DEVELOPMENT AND VALIDATION OF RP-HPLC METHOD FOR QUANTITATIVE ANALYSIS TOLBUTAMIDE IN PURE AND PHARMACEUTICAL FORMULATIONS
 Int. J. Chem. Sci.: 11(4), 2013, 1607-1614 ISSN 0972-768X www.sadgurupublications.com DEVELOPMENT AND VALIDATION OF RP-HPLC METHOD FOR QUANTITATIVE ANALYSIS TOLBUTAMIDE IN PURE AND PHARMACEUTICAL FORMULATIONS
Int. J. Chem. Sci.: 11(4), 2013, 1607-1614 ISSN 0972-768X www.sadgurupublications.com DEVELOPMENT AND VALIDATION OF RP-HPLC METHOD FOR QUANTITATIVE ANALYSIS TOLBUTAMIDE IN PURE AND PHARMACEUTICAL FORMULATIONS
Analysis of Amino Acids Derived Online Using an Agilent AdvanceBio AAA Column
 Application Note Pharmaceutical and Food Testing Analysis of Amino Acids Derived Online Using an Agilent AdvanceBio AAA Column Author Lu Yufei Agilent Technologies, Inc. Abstract A liquid chromatographic
Application Note Pharmaceutical and Food Testing Analysis of Amino Acids Derived Online Using an Agilent AdvanceBio AAA Column Author Lu Yufei Agilent Technologies, Inc. Abstract A liquid chromatographic
APPLICATIONS TN A Comparison of Various Kinetex C18 Phases USP: L1
 APPLICATIONS A Comparison of Various Kinetex C18 Phases Zeshan Aqeel and Jeff Layne Phenomenex, Inc., 411 Madrid Ave., Torrance, CA 91 USA Introduction When performing reversed phase LC separations, there
APPLICATIONS A Comparison of Various Kinetex C18 Phases Zeshan Aqeel and Jeff Layne Phenomenex, Inc., 411 Madrid Ave., Torrance, CA 91 USA Introduction When performing reversed phase LC separations, there
Maximizing chromatographic peak capacity with the Agilent 1290 Infinity LC system
 Maximizing chromatographic peak capacity with the Agilent 1290 Infinity LC system A practical guide on how to use parameters to increase peak capacity Application Note Pharmaceutical and Chemical Author
Maximizing chromatographic peak capacity with the Agilent 1290 Infinity LC system A practical guide on how to use parameters to increase peak capacity Application Note Pharmaceutical and Chemical Author
ZIDOVUDINE, LAMIVUDINE AND ABACAVIR TABLETS Draft proposal for The International Pharmacopoeia (September 2006)
 September 2006 RESTRICTED ZIDOVUDINE, LAMIVUDINE AND ABACAVIR TABLETS Draft proposal for The International Pharmacopoeia (September 2006) This document was provided by a contracted quality control laboratory.
September 2006 RESTRICTED ZIDOVUDINE, LAMIVUDINE AND ABACAVIR TABLETS Draft proposal for The International Pharmacopoeia (September 2006) This document was provided by a contracted quality control laboratory.
Simultaneous Analysis of Active Pharmaceutical Ingredients and Their Counter-Ions Using a Mixed-Mode Column
 P-CN54E Simultaneous Analysis of Active Pharmaceutical Ingredients and Their Counter-Ions Using a Mixed-Mode Column Pittcon 5 9-6P Kenichiro Tanaka, William Hedgepeth, Yuki Sato Shimadzu Scientific Instruments,
P-CN54E Simultaneous Analysis of Active Pharmaceutical Ingredients and Their Counter-Ions Using a Mixed-Mode Column Pittcon 5 9-6P Kenichiro Tanaka, William Hedgepeth, Yuki Sato Shimadzu Scientific Instruments,
This revision also necessitates a change in the table numbering in the test for Organic Impurities.
 Methylphenidate Hydrochloride Extended-Release Tablets Type of Posting Notice of Intent to Revise Posting Date 27 Jul 2018 Targeted Official Date To Be Determined, Revision Bulletin Expert Committee Chemical
Methylphenidate Hydrochloride Extended-Release Tablets Type of Posting Notice of Intent to Revise Posting Date 27 Jul 2018 Targeted Official Date To Be Determined, Revision Bulletin Expert Committee Chemical
Independent Column Temperature Control Using an LTM Oven Module for Improved Multidimensional Separation of Chiral Compounds
 Independent Column Temperature Control Using an LTM Oven Module for Improved Multidimensional Separation of Chiral Compounds Application Note Food and Flavors Author Frank David Research Institute for
Independent Column Temperature Control Using an LTM Oven Module for Improved Multidimensional Separation of Chiral Compounds Application Note Food and Flavors Author Frank David Research Institute for
Unequalled durability against water elution. % tr
 Unequalled durability against water elution 2 Revolutionary aqueous durability for aminopropyl phase 1 3 4 1. fructose (2.5mg/mL) 3. sucrose (2.5mg/mL) 500 hr 2. glucose 400 hr Aqueous to non-aqueous Normal
Unequalled durability against water elution 2 Revolutionary aqueous durability for aminopropyl phase 1 3 4 1. fructose (2.5mg/mL) 3. sucrose (2.5mg/mL) 500 hr 2. glucose 400 hr Aqueous to non-aqueous Normal
*, G. Manetto, Z. Deyl, F. Tagliaro, I. Miksık ˇ
 Journal of Chromatography B, 741 (2000) 67 75 www.elsevier.com/ locate/ chromb Capillary electrophoretic separation of vitamins in sodium dodecyl sulfate containing buffers with lower aliphatic alcohols
Journal of Chromatography B, 741 (2000) 67 75 www.elsevier.com/ locate/ chromb Capillary electrophoretic separation of vitamins in sodium dodecyl sulfate containing buffers with lower aliphatic alcohols
[19] ,516,984 5/1985 Warner... 55/16. CO-CH, (N-hydroxysuccinimide) (dicyclohexylcarbodiimide) CO-CH, CH,-CHRCOON' +
![[19] ,516,984 5/1985 Warner... 55/16. CO-CH, (N-hydroxysuccinimide) (dicyclohexylcarbodiimide) CO-CH, CH,-CHRCOON' + [19] ,516,984 5/1985 Warner... 55/16. CO-CH, (N-hydroxysuccinimide) (dicyclohexylcarbodiimide) CO-CH, CH,-CHRCOON' +](/thumbs/80/81911159.jpg) United States Patent Warner et al. [19] US005770084A [11] Patent Number: 5,770,084 [45] Date of Patent: Jun. 23, 1998 [54] POLYMERIZED CHIRAL MICELLES FOR CHIRAL SEPARATIONS [75] Inventors: Isiah M. Warner,
United States Patent Warner et al. [19] US005770084A [11] Patent Number: 5,770,084 [45] Date of Patent: Jun. 23, 1998 [54] POLYMERIZED CHIRAL MICELLES FOR CHIRAL SEPARATIONS [75] Inventors: Isiah M. Warner,
Determination of Water- and Fat-Soluble Vitamins in Nutritional Supplements by HPLC with UV Detection
 Application Note 5 Determination of Water- and Fat- Vitamins in Nutritional Supplements by HPLC with UV Detection Introduction Vitamins are a well-known a group of compounds that are essential for human
Application Note 5 Determination of Water- and Fat- Vitamins in Nutritional Supplements by HPLC with UV Detection Introduction Vitamins are a well-known a group of compounds that are essential for human
Achiral SFC: Development of an Orthogonal SFC Method for Mometasone Furoate Impurity Analysis
 Achiral SFC: Development of an Orthogonal SFC Method for Mometasone Furoate Impurity Analysis Zhenyu Wang Merck Research Laboratories NJCG 2013 Outline SFC chronicle The journey of a half century Re-birth
Achiral SFC: Development of an Orthogonal SFC Method for Mometasone Furoate Impurity Analysis Zhenyu Wang Merck Research Laboratories NJCG 2013 Outline SFC chronicle The journey of a half century Re-birth
Simultaneous Estimation of Gemcitabine Hydrochloride and Capecitabine Hydrochloride in Combined Tablet Dosage Form by RP-HPLC Method
 ISSN: 0973-4945; CODEN ECJHAO E- Chemistry http://www.e-journals.net 2011, 8(3), 1212-1217 Simultaneous Estimation of Gemcitabine Hydrochloride and Capecitabine Hydrochloride in Combined Tablet Dosage
ISSN: 0973-4945; CODEN ECJHAO E- Chemistry http://www.e-journals.net 2011, 8(3), 1212-1217 Simultaneous Estimation of Gemcitabine Hydrochloride and Capecitabine Hydrochloride in Combined Tablet Dosage
Poster No. PT [ ANALYSIS OF ANION CONSTITUENTS OF URINE BY INORGANIC CAPILLARY ELECTROPHORESIS
 Poster No. PT-56 94-[ ANALYSS OF ANON CONSTTUENTS OF URNE BY NORGANC CAPLLARY ELECTROPHORESS Presented by: Bill J.Wildman*, Peter E.Jackson, William R.Jones and Peter G.Alden Waters Division of Millipore
Poster No. PT-56 94-[ ANALYSS OF ANON CONSTTUENTS OF URNE BY NORGANC CAPLLARY ELECTROPHORESS Presented by: Bill J.Wildman*, Peter E.Jackson, William R.Jones and Peter G.Alden Waters Division of Millipore
Analysis of Testosterone, Androstenedione, and Dehydroepiandrosterone Sulfate in Serum for Clinical Research
 Analysis of Testosterone, Androstenedione, and Dehydroepiandrosterone Sulfate in Serum for Clinical Research Dominic Foley, Michelle Wills, and Lisa Calton Waters Corporation, Wilmslow, UK APPLICATION
Analysis of Testosterone, Androstenedione, and Dehydroepiandrosterone Sulfate in Serum for Clinical Research Dominic Foley, Michelle Wills, and Lisa Calton Waters Corporation, Wilmslow, UK APPLICATION
COSMOSIL CHiRAL Series
 Immobilized Polysaccharide Derivative-Based Chiral Columns CSMSIL CiRAL Series Immobilized chiral selectors can withstand many different solvents Sharpen peaks with 3 μm particles Preparative separations
Immobilized Polysaccharide Derivative-Based Chiral Columns CSMSIL CiRAL Series Immobilized chiral selectors can withstand many different solvents Sharpen peaks with 3 μm particles Preparative separations
