GINA L. QUIRARTE*, BENNO ROOZENDAAL*, AND JAMES L. MCGAUGH* MATERIALS AND METHODS
|
|
|
- Noel Miller
- 5 years ago
- Views:
Transcription
1 Proc. Natl. Acad. Sci. USA Vol. 94, pp , December 1997 Neurobiology Glucocorticoid enhancement of memory storage involves noradrenergic activation in the basolateral amygdala (dexamethasone RU adrenergic antagonists inhibitory avoidance) GINA L. QUIRARTE*, BENNO ROOZENDAAL*, AND JAMES L. MCGAUGH* *Center for the Neurobiology of Learning and Memory and Department of Psychobiology, University of California, Irvine, CA ; and Centro de Neurobiología, Universitat Nacional Autonome de Mexico, Queretaro, Qro., Mexico Contributed by James L. McGaugh, October 7, 1997 ABSTRACT Evidence indicates that the modulatory effects of the adrenergic stress hormone epinephrine as well as several other neuromodulatory systems on memory storage are mediated by activation of -adrenergic mechanisms in the amygdala. In view of our recent findings indicating that the amygdala is involved in mediating the effects of glucocorticoids on memory storage, the present study examined whether the glucocorticoid-induced effects on memory storage depend on -adrenergic activation within the amygdala. Microinfusions (0.5 g in0.2 l) of either propranolol (a nonspecific -adrenergic antagonist), atenolol (a 1 -adrenergic antagonist), or zinterol (a 2 -adrenergic antagonist) administered bilaterally into the basolateral nucleus of the amygdala (BLA) of male Sprague Dawley rats 10 min before training blocked the enhancing effect of posttraining systemic injections of dexamethasone (0.3 mg kg) on 48-h memory for inhibitory avoidance training. Infusions of these -adrenergic antagonists into the central nucleus of the amygdala did not block the dexamethasone-induced memory enhancement. Furthermore, atenolol (0.5 g) blocked the memory-enhancing effects of the specific glucocorticoid receptor (GR or type II) agonist RU infused concurrently into the BLA immediately posttraining. These results strongly suggest that -adrenergic activation is an essential step in mediating glucocorticoid effects on memory storage and that the BLA is a locus of interaction for these two systems. Extensive evidence from studies of memory of inhibitory avoidance training in rats indicates that several neuromodulatory systems interact with the noradrenergic system in the amygdala in modulating memory storage (1 8). Intraamygdala infusions of the -adrenergic antagonist propranolol or depletion of norepinephrine in the amygdala by the neurotoxin N-2-chloroethyl-N-ethyl-bromobenzylamine (DSP-4) block the memory-enhancing effect of the adrenergic stress hormone epinephrine (5, 6). Moreover, intra-amygdala infusions of propranolol as well as specific 1 -or 2 -adrenergic antagonists also block the effects, on memory, of drugs affecting opioid peptidergic and GABAergic systems (4, 7). Norepinephrine and other -adrenergic agonists administered to the amygdala after training dose-dependently enhance retention (5, 9 11). These effects are also time-dependent; they affect retention when given shortly after training but have no effect if given several hours later. These findings are consistent with evidence from recent experiments using in vivo microdialysis indicating that norepinephrine is released in the amygdala by footshock stimulation of the kind typically used in inhibitory avoidance training (12). Furthermore, adrenergic The publication costs of this article were defrayed in part by page charge payment. This article must therefore be hereby marked advertisement in accordance with 18 U.S.C solely to indicate this fact by The National Academy of Sciences $ PNAS is available online at http: and opioid peptidergic systems influence the training-induced release of norepinephrine (13, 14). Findings of a series of recent experiments from our laboratory indicate that the amygdala is involved in mediating the memory-modulating effects of glucocorticoids (15). Excitotoxically induced lesions of the basolateral (BLA), but not the central (CEA), nucleus of the amygdala blocked the enhancing effects of systemic posttraining injections of dexamethasone, a synthetic glucocorticoid, on memory for inhibitory avoidance training (16). Furthermore, infusions of the specific glucocorticoid receptor (GR or type II) agonist RU (11,17 dihydroxy-6,21-dimethyl-17 -pregna-4,6-trien-20yn-3-one) into the BLA enhanced memory storage (17). Such findings suggest that glucocorticoid effects on memory storage are mediated, at least in part, by GRs in the BLA. Although the specific mode of action of glucocorticoids differs from that of the hormones and neurotransmitters just described (i.e., glucocorticoids are highly lipophylic and bind directly to intracellular receptors in the brain), the involvement of the amygdala in mediating the effects of all these compounds appears to be strikingly similar. These findings suggest that glucocorticoid-induced effects on memory storage may also depend on intact noradrenergic neurotransmission in the amygdala. In support of this view, there is extensive evidence from biochemical and electrophysiological experiments indicating interactions between glucocorticoids and the noradrenergic system in several brain structures, including the hypothalamus, hippocampus, and cerebral cortex (18, 19). The present study examined the involvement of adrenoceptors in the amygdala in the modulating effects of glucocorticoids on memory storage. In a first experiment, rats received microinfusions of -adrenergic antagonists with either a selective affinity for 1 -or 2 -adrenoceptors or with a nonspecific affinity for both receptor types into the BLA or CEA before training in an inhibitory avoidance task. Dexamethasone was injected subcutaneously immediately after training. In a second experiment, rats received concurrent immediate posttraining infusions of the 1 -adrenergic antagonist atenolol and the specific GR agonist RU into the BLA. In both experiments the animals were tested on retention 48 h after training. MATERIALS AND METHODS Subjects. Male Sprague Dawley rats ( g at time of surgery) from Charles River Laboratories were used. They were individually housed in a temperature-controlled (22 C) colony room and maintained on a standard 12-h light 12-h dark cycle ( h lights on) with food and water Abbreviations: BLA, basolateral amygdala; CEA, central amygdala; GR, glucocorticoid receptor; RU 28362, 11,17 -dihydroxy-6,21- dimethyl-17 -pregna-4,6-trien-20yn-3-one. To whom reprint requests should be addressed
2 Neurobiology: Quirarte et al. Proc. Natl. Acad. Sci. USA 94 (1997) available ad libitum. Training and testing were performed between 1000 and 1500 h. Surgery. The animals were adapted to the vivarium for at least 1 week before surgery. They were anesthetized with sodium pentobarbital (50 mg kg of body weight, i.p.) and given atropine sulfate (0.4 mg kg, i.p.). The skull was fixed to a stereotaxic frame (Kopf Instruments, Tujunga, CA), and stainless-steel guide cannulae (15 mm; 23 gauge) were implanted bilaterally with the cannula tips 2 mm above the BLA [coordinates: anteroposterior (AP), 2.8 mm from bregma; mediolateral (ML), 5.0 mm from midline; dorsoventral (DV), 6.5 mm from skull surface] or CEA [coordinates: AP, 2.2 mm; ML, 4.3 mm; DV, 6.0 mm] according to the atlas of Paxinos and Watson (20). The cannulas and two anchoring screws were affixed to the skull with dental cement. Stylets (15 mm long 00 insect dissection pins) were inserted into the cannulae to maintain patency and were removed only for the infusion of drugs. The rats were allowed to recover 7 days before training and were handled three times for 1 min during this recovery period. Drugs and Infusion Procedures. The nonspecific adrenergic antagonist dl-propranolol (0.5 g per side; Sigma) or the specific 1 -or 2 -adrenergic antagonists atenolol (0.5 g per side; Sigma) or zinterol (0.5 g per side; Bristol-Myers), respectively, were dissolved in 0.9% saline and infused either into the BLA or the CEA 10 min before training. The doses were selected on the basis of previous experiments conducted in this laboratory (21). Bilateral intra-amygdala nuclei infusions of either of the -adrenergic antagonists or a saline control solution were made by using 30-gauge injection needles connected to a 10- l Hamilton syringe by polyethylene (PE- 20) tubing. The injection needles protruded 2 mm beyond the tips of the cannulas. A 0.2- l injection volume per hemisphere was infused over a period of 25 s by an automated syringe pump (Sage Instruments, Boston). The infusion volume was based on our findings that selective neurotoxically induced lesions of the BLA and CEA are produced with an infusion volume of 0.2 l (16). The injection needles were retained within the cannulas for an additional 30 s following drug infusion to maximize diffusion. After completion of the infusion the animals were returned to their home cages until the start of the inhibitory avoidance training. Dexamethasone (Sigma) was injected subcutaneously at a dose of 0.3 mg kg in a volume of 2.0 ml kg immediately after training. This dose was selected on the basis of our previous experiments (16). Dexamethasone was first dissolved in 100% ethanol and subsequently diluted in 0.9% saline to reach the appropriate concentration. The final concentration of ethanol was 2%. Solutions of all drugs were freshly prepared before the experiments. For the second experiment, atenolol (0.5 g) and the specific GR agonist RU (1.0, 3.0, or 10.0 ng) were infused together into the BLA immediately after training. The drugs were first dissolved in 100% ethanol and subsequently diluted with saline to reach a 2% ethanol solution. The infusion volume and procedures used were identical to those described above. Inhibitory Avoidance Apparatus and Procedure. The rats were trained in an inhibitory avoidance apparatus (8) consisting of a trough-shaped alley (91 cm long, 15 cm deep, 20 cm wide at the top, 6.4 cm wide at the floor) divided into two compartments separated by a sliding door that opened by retracting into the floor. The starting compartment (31 cm long) was illuminated and the shock compartment (60 cm long) was dark. Training was conducted in a sound and lightattenuated room. The rat was placed in the starting compartment of the apparatus, facing away from the door, and was allowed to enter the dark compartment. As the rat stepped completely into the dark compartment, the door was closed and a footshock was delivered. For the animals given pretraining infusions of drug or control solution into the BLA, the footshock level used was 0.45 ma with a duration of 1 s. Preliminary data revealed that comparable retention latencies in CEA-infused animals were obtained with a slightly lower footshock intensity. Therefore, CEA-infused animals received a footshock intensity of 0.40 ma for 1 s. The animals were removed from the shock compartment 15 s after termination of the footshock and, after drug treatment, returned to their home cages. On the retention test 48 h after training, the rat was placed in the starting compartment, as in the training session, and the latency to re-enter the dark compartment (maximum latency of 600 s) was recorded. Longer latencies were interpreted as indicating better retention. Shock was not administered on the retention test trial. Histology. The rats were anesthetized with an overdose of sodium pentobarbital (100 mg kg, i.p.) and perfused intracardially with a 0.9% saline solution followed by 10% formaldehyde (vol vol). Following decapitation, the brains were removed and placed in 10% formaldehyde. At least 24 h before sectioning, the brains were submerged in a 20% sucrose solution (wt vol) for cryoprotection. Sections of 40 m were made (using a freezing microtome) and stained with cresyl violet. The sections were examined under a light microscope. Determination of the location of the cannula tips was made according to the standardized atlas plates of Paxinos and Watson (20). Statistics. Retention data were analyzed with a two-way ANOVA with -adrenergic antagonist treatment and glucocorticoid treatment both as between-subject variables. Because different footshock intensities were used for the BLAinfused and CEA-infused animals, data of the two sets of groups were analyzed with separate ANOVAs. Further analysis used Fisher s post-hoc tests to determine the source of the detected significances in the ANOVAs. A probability level of less than 0.05 was accepted as statistical significance. The number of animals per group is indicated in the figure legends. RESULTS Fig. 1 A and B show representative brain slices showing the location of tips of cannulas aimed at the BLA and CEA, respectively. Behavioral data from 29% of the animals (108 of 371) were excluded from statistical analysis either because the cannulae in these animals were not placed bilaterally in the specific amygdala nuclei or because the infusion induced extensive damage to the targeted areas. The retention test latencies of rats given pretraining infusions of -adrenergic antagonists into either the BLA or the CEA followed by immediate posttraining systemic injections of the glucocorticoid dexamethasone are shown in Fig. 2. The retention test latencies of animals that received control treatments were relatively short, as was intended. A two-way ANOVA for the data of BLA-infused animals revealed a significant dexamethasone -adrenergic antagonist interaction [F(3,80) 10.08; P ]. Post-hoc comparisons between groups indicated that none of the -adrenergic antagonists affected retention latencies in otherwise untreated animals. Systemic posttraining injections of dexamethasone enhanced the retention of rats given saline infusions into the BLA compared with that of the corresponding vehicle-treated animals (P 0.01). However, dexamethasone did not increase the retention latencies of animals given infusions of either of the -adrenergic antagonists in the BLA. In fact, in these animals dexamethasone showed a nonsignificant impairment in retention performance. Further, retention latencies of dexamethasone-treated rats given any of the -adrenergic antagonists into the BLA were significantly shorter than retention latencies of dexamethasone-treated rats infused with saline (all P 0.01).
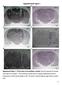
3 14050 Neurobiology: Quirarte et al. Proc. Natl. Acad. Sci. USA 94 (1997) FIG. 1. Photomicrographs illustrating representative location of cannulas and injection needle tips within the BLA (A) and CEA (B). Arrows point to the cannula tips. (The injection needles were inserted through the surgically implanted guide cannula and protruded 2 mm beyond the tip of each cannula to reach the BLA or CEA.) OT, optic tract. A two-way ANOVA for the data of CEA-infused animals revealed a significant dexamethasone effect [F(1,81) 24.55; P ], but no -adrenergic antagonist effect [F(3,81) 0.17; NS] or dexamethasone -adrenergic antagonist interaction [F(3,81) 0.56; NS]. Post-hoc comparisons indicated that none of the -adrenergic antagonists influenced retention performance of otherwise untreated animals and that dexamethasone enhanced retention in saline-treated rats (P 0.01), as well as in all of the -adrenergic antagonist-treated rats (propranolol and atenolol, P 0.05; zinterol, P 0.01). Fig. 3 shows retention test latencies for rats infused concurrently with the specific GR agonist RU and the 1 - adrenergic antagonist atenolol into the BLA immediately after training. A two-way ANOVA revealed significant glucocorticoid [F(3,80) 6.39; P 0.001] and atenolol effects [F(1,80) 14.34; P 0.001] and a significant glucocorticoid atenolol interaction [F(3,80) 3.63; P 0.05]. Post-hoc analysis revealed that posttraining infusions of the GR agonist into the BLA dose-dependently enhanced retention performance. Retention latencies of rats treated with the two lower doses of RU (i.e., 1.0 and 3.0 ng) were significantly longer than those of vehicle-treated rats (both P 0.01). Infusion of the highest dose of RU did not significantly enhance retention. When RU was infused together with atenolol the memory-enhancing effect of RU was blocked. Retention latencies of animals treated simultaneously with RU and atenolol were significantly shorter than those of animals treated with RU alone (1.0 ng, P 0.01; 3.0 ng, P 0.05). DISCUSSION These findings provide additional evidence that glucocorticoids and the noradrenergic system are involved in modulating
4 Neurobiology: Quirarte et al. Proc. Natl. Acad. Sci. USA 94 (1997) FIG. 3. Inhibitory avoidance retention latencies of animals that received posttraining concurrent administration of the GR agonist RU (1.0, 3.0, or 10.0 ng) and the 1 -adrenergic antagonist atenolol (0.5 g) into the BLA. Bars represent mean ( SEM) latency in seconds., P 0.01 as compared with the vehicle group;, P 0.05;, P 0.01 as compared with the corresponding RU group (n 9 13 group). FIG. 2. Inhibitory avoidance retention latencies of animals that received pretraining infusions of either the nonspecific -adrenergic antagonist propranolol (0.5 g), the 1 -adrenergic antagonist atenolol (0.5 g), or the 2 -adrenergic antagonist zinterol (0.5 g) into the BLA or CEA and immediate posttraining subcutaneous injections of dexamethasone (0.3 mg kg). Bars represent mean ( SEM) latency in seconds., P 0.05;, P 0.01 as compared with the corresponding vehicle group;, P 0.01 as compared with the vehicledexamethasone group (n 8 14 group). memory storage (5, 9 11, 16, 17, 22 29). Further, they indicate that the BLA is a locus of interaction between these two systems in regulating memory. Systemic injections of dexamethasone enhanced inhibitory avoidance retention when administered immediately after training. As dexamethasone has a high affinity for GRs (30), these findings are consistent with the hypothesis that long-term storage of information is strengthened by posttraining activation of GR-sensitive pathways (23, 25, 27, 28, 31). Similarly, in support of our previous findings (17), posttraining infusions of the specific GR agonist RU into the BLA enhanced memory storage. These findings, together with the finding that the BLA has a moderate density of GRs (32, 33), strongly suggest that glucocorticoid effects on memory storage are mediated, at least in part, by binding to GRs in the BLA. The finding that the highest dose of RU (10.0 ng) was ineffective in enhancing retention performance indicates that glucocorticoids, via GRs, have biphasic effects on memory storage. These findings are consistent with those of a recent study examining the effects of systemic administration of different doses of corticosterone to adrenalectomized rats on spatial memory in a Y-maze task (31). It was found that the level of GR occupancy, as measured by a binding assay, was significantly correlated with spatial memory performance following an inverted-u shape curve, whereas the level of mineralocorticoid receptor (MR or type I) occupancy was not. The effects of doses of glucocorticoids used also depend on the specific training conditions used. In the present experiment the animals received a relative lowintensity footshock which resulted in a weak memory that was enhanced by dexamethasone. However, when animals are trained under more stressful conditions that induce high circulating levels of endogenous glucocorticoids, such as in a water-maze spatial task, posttraining administration of a similar dose of dexamethasone impairs memory (34). Administration of a specific GR antagonist also impairs memory formation in the water maze (17, 25, 27). The main finding of the present experiments is that infusions of -adrenergic antagonists into the BLA blocked the memory-enhancing effects of systemically or intra-amygdally administered glucocorticoids. Moreover, the finding that higher doses of the GR agonist were ineffective in enhancing memory in animals given atenolol concurrently suggests that adrenergic receptor activation in the BLA is required in order for glucocorticoids to modulate memory storage processes. Atenolol did not simply induce a shift in the dose-response effects of glucocorticoids. No differential effects were observed following the administration of the specific 1 - or 2 -adrenergic antagonists atenolol or zinterol, respectively, or the nonspecific -adrenergic antagonist propranolol. The finding that pretraining infusions of -adrenergic antagonists administered alone did not impair memory for inhibitory avoidance training is consistent with the results of previous studies as well as with our finding that excitotoxic lesions of the BLA induced before training do not impair inhibitory avoidance learning (6, 8, 16). Previous findings indicate that a much higher dose of propranolol (i.e., 5.0 g) administered posttraining induces memory impairment (5, 9).
5 14052 Neurobiology: Quirarte et al. Proc. Natl. Acad. Sci. USA 94 (1997) Although the CEA has a higher density of GRs than does the BLA (32, 33) and the CEA is also richly innervated by noradrenergic fibers (35), antagonism of -adrenoceptors in the CEA did not block the memory-enhancing effects of systemically administered dexamethasone. Such a selective involvement of the BLA in mediating hormonal effects on memory storage is consistent with previous findings that neurotoxically induced lesions of the BLA, but not the CEA, blocked the memory enhancement induced by systemically administered dexamethasone (16) as well as with evidence that microinfusion of glucocorticoids into the BLA, but not the CEA, enhanced retention (17). The adrenal steroid receptors in the CEA are considered to be involved in feedback mechanisms of the hypothalamic pituitary adrenal cortex axis and behavioral responsiveness to stress (36). The present findings indicate that it is very unlikely that the modulatory effects of hormones on cognition involve information flowing from the BLA to the CEA as has been described for processes of fear conditioning (37). In general, the present results are highly consistent with findings of our previous studies examining the role of amygdaloid -adrenergic mechanisms in mediating the effects of other neuromodulatory systems on memory storage (7, 38). As GRs are intracellular receptors, they are not present presynaptically on the noradrenergic terminals in the BLA (39). Thus, it seems likely that glucocorticoids bind postsynaptically of the noradrenergic receptor system in the BLA, possibly in the same neurons expressing the -adrenergic receptors. Although the possibility that glucocorticoids modulate the activity and or efficacy of the noradrenergic system in the BLA via postsynaptic mechanisms has not been investigated, there is an extensive literature concerning studies of the acute effects of glucocorticoids on the noradrenergic system in hypothalamic, hippocampal, and cerebral cortical areas (18, 19, 40 43). Systemically administered glucocorticoids bind to GRs in many brain regions. It is of particular interest that GRs are abundantly present in the cell bodies of the A1 A7 noradrenergic cell groups (32, 44), and that the locus coeruleus (LC) and the nucleus of the solitary tract (NTS) densely innervate the amygdala (45, 46). Most noradrenergic nerve terminals reach the CEA, but a considerable density of fibers is found in the BLA (35). Thus, glucocorticoids might modulate memory storage by influencing the activity of the central noradrenergic system at both pre- and postsynaptic sites (18). It has been reported that glucocorticoids do not affect tyrosine hydroxylase (47) or norepinephrine synthesis rate in the LC of adult rodents (19). However, we recently found that activation of GRs in the NTS with posttraining infusions of the specific agonist RU dose-dependently enhanced memory for inhibitory avoidance training (B.R., C. L. Williams, and J.L.M., unpublished observation). Furthermore, the memory enhancement was blocked in animals given infusions of atenolol into the BLA. These findings suggest that activation of GRs in the NTS, and possibly the LC, may increase the release of norepinephrine in the BLA. We have not as yet examined this implication experimentally. Finally, there might be an indirect way for -adrenergic antagonists administered to the BLA to block the memoryenhancing effects of dexamethasone. Systemically administered dexamethasone also binds and activates hippocampal GRs (30, 48). However, the present findings indicate that a blockade of -adrenergic mechanisms in the BLA blocked the dexamethasone effect, suggesting that any contribution of GR activation in the hippocampus to memory storage is also blocked. Such an interaction between the BLA and the hippocampus in glucocorticoid-induced memory modulation is supported by recent findings from our laboratory indicating that lesions of the BLA block the memory-enhancing effects of the GR agonist RU infused directly into the dorsal hippocampus (49). These findings, together with evidence that lesions of the BLA as well as infusions of propranolol into the BLA inhibit the effects of electrical stimulation of the perforant path on the induction of long-term potentiation in the dentate gyrus granule cell synapses (50, 51), suggest that -adrenergic activation in the BLA may be essential for inducing glucocorticoid-mediated plasticity in the hippocampus. These findings are consistent with our hypothesis that activation of the BLA may modulate or comodulate memory consolidation processes in other brain regions, including the hippocampus and caudate nucleus (52). We thank Drs. Béla Bohus and Bruce S. McEwen for helpful comments on an earlier version of the manuscript, and Cindy Quach and Bichngoc Nguyen for excellent technical assistance. RU was generously provided by Rousell Uclaf (Romainville, France). This research was supported by Dirección General de Asuntos del Personal Académico-Universitat Nacional Autonome de Mexico (G.L.Q.), an R. W. and L. Gerard Trust Fellowship (B.R.), and National Institute of Mental Health National Institute on Drug Abuse Research Grant MH12526 (J.L.M.). 1. Introini-Collison, I. B., Dalmaz, C. & McGaugh, J. L. (1996) Neurobiol. Learn. Mem. 65, Introini-Collison, I. B., Ford, L. & McGaugh, J. L. (1995) Neurobiol. Learn. Mem. 63, Introini-Collison, I. B., Nagahara, A. H. & McGaugh, J. L. (1989) Brain Res. 476, Introini-Collison, I. B., Saghafi, D., Novack, G. & McGaugh, J. L. (1992) Brain Res. 572, Liang, K. C., Chen, L. L. & Huang, T.-E. (1995) Chin. J. Physiol. 38, Liang, K. C., Juler, R. G. & McGaugh, J. L. (1986) Brain Res. 368, McGaugh, J. L., Cahill, L., Parent, M. B., Mesches, M. H., Coleman-Mesches, K. & Salinas, J. A. (1995) in Plasticity in the Central Nervous System: Learning and Memory, eds. McGaugh, J. L., Bermúdez-Rattoni, F. & Prado-Alcalá, R. A. (Lawrence Erlbaum, Mahwah, NJ), pp McGaugh, J. L., Introini-Collison, I. B. & Nagahara, A. H. (1988) Brain Res. 446, Gallagher, M., Kapp, B. S., Musty, R. E. & Driscoll, P. A. (1977) Science 198, Introini-Collison, I. B., Miyazaki, B. & McGaugh, J. L. (1991) Psychopharmacology 104, Liang, K. C., McGaugh, J. L. & Yao, H.-Y. (1990) Brain Res. 508, Galvez, R., Mesches, M. H. & McGaugh, J. L. (1996) Neurobiol. Learn. Mem. 66, Galvez, R., Quirarte, G. L. & McGaugh, J. L. (1996) Soc. Neurosci. Abstr. 22, Williams, C. L., Gold, P. E. & Men, D. (1996) Soc. Neurosci. Abstr. 22, Roozendaal, B., Cahill, L. & McGaugh, J. L. (1996) in Brain Processes and Memory, eds. Ishikawa, K., McGaugh, J. L. & Sakata, H. (Elsevier, Amsterdam), pp Roozendaal, B. & McGaugh, J. L. (1996) Neurobiol. Learn. Mem. 65, Roozendaal, B. & McGaugh, J. L. (1997) Neurobiol. Learn. Mem. 67, de Kloet, E. R. (1991) Front. Neuroendocrinol. 12, McEwen, B. S. (1987) Biochem. Pharmacol. 36, Paxinos, G. & Watson, C. (1986) The Rat Brain in Stereotaxic Coordinates (Academic, San Diego). 21. McGaugh, J. L., Introini-Collison, I. B., Cahill, L., Kim, M. & Liang, K. C. (1992) in The Amygdala: Neurobiological Aspects of Emotion, Memory, and Mental Dysfunction, ed. Aggleton, J. P. (Wiley Liss, New York), pp Cottrell, G. A. & Nakajima, S. (1977) Pharmacol. Biochem. Behav. 7, de Kloet, E. R., de Kock, S., Schild, V. & Veldhuis, D. H. (1988) Neuroendocrinology 47, Flood, J. F., Vidal, D., Bennett, E. L., Orme, A. E. Rosenzweig, M. R. & Jarvik, M. E. (1978) Pharmacol. Biochem. Behav. 8,
6 Neurobiology: Quirarte et al. Proc. Natl. Acad. Sci. USA 94 (1997) Kovacs, G. L., Telegdy, G. & Lissak, K. (1977) Horm. Behav. 8, Oitzl, M. S. & de Kloet, E. R. (1992) Behav. Neurosci. 106, Pugh, C. R., Tremblay, D., Fleshner, M. & Rudy, J. W. (1997) Behav. Neurosci. 111, Roozendaal, B., Portillo-Marquez, G. & McGaugh, J. L. (1996) Behav. Neurosci. 110, Sandi, C. & Rose, S. P. R. (1994) Brain Res. 647, Reul, J. M. H. M. & de Kloet, E. R. (1985) Endocrinology 117, Lupien, S. J. & McEwen, B. S. (1997) Brain Res. Rev. 24, Honkaniemi, J., Pelto-Huikko, M., Rechardt, L., Isola, J., Lammi, A. Fuxe, K., Gustafsson, J. Å., Wikström, A.-C. & Hökfelt, T. (1992) Neuroendocrinology 55, Morimoto, M., Morita, N., Ozawa, H., Yokoyama, K. & Kawata, M. (1996) Neurosci. Res. 26, Roozendaal, B., Bohus, B. & McGaugh, J. L. (1996) Psychoneuroendocrinology 21, Fallon, J. H. & Ciofi, P. (1992) in The Amygdala: Neurobiological Aspects of Emotion, Memory, and Mental Dysfunction, ed. Aggleton, J. P. (Wiley Liss, New York), pp Schulkin, J., McEwen, B. S. & Gold, P. W. (1994) Neurosci. Biobehav. Rev. 18, Sarter, M. & Markowitsch, H. J. (1985) Behav. Neurosci. 99, McGaugh, J. L., Cahill, L. & Roozendaal, B. (1996) Proc. Natl. Acad. Sci. USA 93, Joëls, M. & de Kloet, E. R. (1994) Prog. Neurobiol. 43, Duman, R. S., Strada, S. J. & Enna, S. J. (1989) Brain Res. 477, Joëls, M. & de Kloet, E. R. (1989) Science 245, Nakagawa, K. & Kuriyama, K. (1976) J. Neurochem. 27, Stone, E. A., McEwen, B. S., Herrera, A. S. & Carr, E. D. (1987) Eur. J. Pharmacol. 141, Härfstrand, A., Fuxe, K., Cintra, A., Agnati, L. F., Zini, I., Wikström, A. C., Okret, S., Yu, Z. Y., Goldstein, M., Steinbusch, H., Verhofstad, A. & Gustafsson, J.-Å. (1986) Proc. Natl. Acad. Sci. USA 83, Fallon, J. H., Koziell, D. A. & Moore, R. Y. (1978) J. Comp. Neurol. 180, Zardetto-Smit, A. M. & Gray, T. S. (1990) Brain Res. Bull. 25, Markey, K. A., Towle, A. C. & Sze, P. Y. (1982) Endocrinology 111, McEwen, B. S., Weiss, J. M. & Schwartz, L. S. (1968) Nature (London) 220, Roozendaal, B. & McGaugh, J. L. (1997) Eur. J. Neurosci. 9, Ikegaya, Y., Saito, H. & Abe, K. (1994) Brain Res. 656, Ikegaya, Y., Nakanishi, K., Saito, H. & Abe, K. (1997) Neuro- Report 8, Packard, M. G., Cahill, L. & McGaugh, J. L. (1994) Proc. Natl. Acad. Sci. USA 91,
Glucocorticoids and the regulation of memory consolidation
 Psychoneuroendocrinology 25 (2000) 213 238 www.elsevier.com/locate/psyneuen 1999 Curt P. Richter Award Glucocorticoids and the regulation of memory consolidation Benno Roozendaal * Center for the Neurobiology
Psychoneuroendocrinology 25 (2000) 213 238 www.elsevier.com/locate/psyneuen 1999 Curt P. Richter Award Glucocorticoids and the regulation of memory consolidation Benno Roozendaal * Center for the Neurobiology
Emotional Hormones and Memory Modulation
 Emotional Hormones and Memory Modulation 933 Emotional Hormones and Memory Modulation J L McGaugh and B Roozendaal, University of California, Irvine, CA, USA ã 2009 Elsevier Ltd. All rights reserved. Introduction
Emotional Hormones and Memory Modulation 933 Emotional Hormones and Memory Modulation J L McGaugh and B Roozendaal, University of California, Irvine, CA, USA ã 2009 Elsevier Ltd. All rights reserved. Introduction
Memory enhancement of classical fear conditioning by post-training injections of corticosterone in rats
 Neurobiology of Learning and Memory 81 (2004) 67 74 www.elsevier.com/locate/ynlme Memory enhancement of classical fear conditioning by post-training injections of corticosterone in rats Gabriel K. Hui,
Neurobiology of Learning and Memory 81 (2004) 67 74 www.elsevier.com/locate/ynlme Memory enhancement of classical fear conditioning by post-training injections of corticosterone in rats Gabriel K. Hui,
Systemic administration of drugs, including adrenergic agonists
 Lesions of the nucleus basalis magnocellularis induced by 192 IgG-saporin block memory enhancement with posttraining norepinephrine in the basolateral amygdala Ann E. Power*, Leon J. Thal, and James L.
Lesions of the nucleus basalis magnocellularis induced by 192 IgG-saporin block memory enhancement with posttraining norepinephrine in the basolateral amygdala Ann E. Power*, Leon J. Thal, and James L.
Infusions of AP5 into the basolateral amygdala impair the formation, but not the expression, of step-down inhibitory avoidance
 Brazilian Journal of Medical and Biological Research (2) 33: 829-834 Amygdaloid NMDA receptors and fear memory ISSN 1-879X 829 Infusions of AP5 into the basolateral amygdala impair the formation, but not
Brazilian Journal of Medical and Biological Research (2) 33: 829-834 Amygdaloid NMDA receptors and fear memory ISSN 1-879X 829 Infusions of AP5 into the basolateral amygdala impair the formation, but not
Amygdala Modulation of Memory Consolidation: Interaction with Other Brain Systems
 Neurobiology of Learning and Memory 78, 539 552 (2002) doi:10.1006/nlme.2002.4082 Amygdala Modulation of Memory Consolidation: Interaction with Other Brain Systems James L. McGaugh, Christa K. McIntyre,
Neurobiology of Learning and Memory 78, 539 552 (2002) doi:10.1006/nlme.2002.4082 Amygdala Modulation of Memory Consolidation: Interaction with Other Brain Systems James L. McGaugh, Christa K. McIntyre,
Supplementary Methods
 1 Supplementary Methods Social Preference Test Subjects Seventy-four Long-Evans, male rats served as subjects (S-rats) in the foodpreference test, with 40 assigned to the CXT-Same (CXT-S) Condition and
1 Supplementary Methods Social Preference Test Subjects Seventy-four Long-Evans, male rats served as subjects (S-rats) in the foodpreference test, with 40 assigned to the CXT-Same (CXT-S) Condition and
Differential involvement of the hippocampus, anterior cingulate cortex, and basolateral amygdala in memory for context and footshock
 Differential involvement of the hippocampus, anterior cingulate cortex, and basolateral amygdala in memory for context and footshock Emily L. Malin and James L. McGaugh* Center for the Neurobiology of
Differential involvement of the hippocampus, anterior cingulate cortex, and basolateral amygdala in memory for context and footshock Emily L. Malin and James L. McGaugh* Center for the Neurobiology of
Contributed by James L. McGaugh, August 24, 2016 (sent for review July 5, 2016; reviewed by Mark Packard and Tracey J. Shors) Significance
 Mushroom spine dynamics in medium spiny neurons of dorsal striatum associated with memory of moderate and intense training Paola C. Bello-Medina a, Gonzalo Flores b, Gina L. Quirarte a, James L. McGaugh
Mushroom spine dynamics in medium spiny neurons of dorsal striatum associated with memory of moderate and intense training Paola C. Bello-Medina a, Gonzalo Flores b, Gina L. Quirarte a, James L. McGaugh
Frontiers in Neuroendocrinology
 Frontiers in Neuroendocrinology xxx (2009) xxx xxx Contents lists available at ScienceDirect Frontiers in Neuroendocrinology journal homepage: www.elsevier.com/locate/yfrne Review Glucocorticoids and the
Frontiers in Neuroendocrinology xxx (2009) xxx xxx Contents lists available at ScienceDirect Frontiers in Neuroendocrinology journal homepage: www.elsevier.com/locate/yfrne Review Glucocorticoids and the
Supporting Online Material for
 www.sciencemag.org/cgi/content/full/328/5983/1288/dc1 Supporting Online Material for Induction of Fear Extinction with Hippocampal-Infralimbic BDNF Jamie Peters, Laura M. Dieppa-Perea, Loyda M. Melendez,
www.sciencemag.org/cgi/content/full/328/5983/1288/dc1 Supporting Online Material for Induction of Fear Extinction with Hippocampal-Infralimbic BDNF Jamie Peters, Laura M. Dieppa-Perea, Loyda M. Melendez,
The basolateral amygdala is a cofactor in memory enhancement produced by intrahippocampal glutamate injections
 Psychobiology 1999, 27 (3), 377-385 The basolateral amygdala is a cofactor in memory enhancement produced by intrahippocampal glutamate injections MARK G. PACKARD and SCOTT A. CHEN University oj New Orleans,
Psychobiology 1999, 27 (3), 377-385 The basolateral amygdala is a cofactor in memory enhancement produced by intrahippocampal glutamate injections MARK G. PACKARD and SCOTT A. CHEN University oj New Orleans,
Neurobiology of Learning and Memory
 Neurobiology of Learning and Memory xxx (2009) xxx xxx Contents lists available at ScienceDirect Neurobiology of Learning and Memory journal homepage: www.elsevier.com/locate/ynlme Memory-enhancing corticosterone
Neurobiology of Learning and Memory xxx (2009) xxx xxx Contents lists available at ScienceDirect Neurobiology of Learning and Memory journal homepage: www.elsevier.com/locate/ynlme Memory-enhancing corticosterone
<student name> Undergraduate Research Grant Proposal
 Undergraduate Research Grant Proposal A. Project Description Objective of research: The objective of this study is to determine if hippocampal dopamine D1 receptors facilitate novel object
Undergraduate Research Grant Proposal A. Project Description Objective of research: The objective of this study is to determine if hippocampal dopamine D1 receptors facilitate novel object
NIH Public Access Author Manuscript Nat Neurosci. Author manuscript; available in PMC 2006 September 5.
 NIH Public Access Author Manuscript Published in final edited form as: Nat Neurosci. 2006 August ; 9(8): 1004 1006. Maternal presence serves as a switch between learning fear and attraction in infancy
NIH Public Access Author Manuscript Published in final edited form as: Nat Neurosci. 2006 August ; 9(8): 1004 1006. Maternal presence serves as a switch between learning fear and attraction in infancy
Abstract. R. Roesler 1, J. Quevedo 1, C. Rodrigues 1, M. Madruga 1, M.R.M. Vianna 1 and M.B.C. Ferreira 2
 Brazilian Amygdaloid Journal non-nmda of Medical receptors and Biological and memory Research expression (1999) 32: 349-353 ISSN 0100-879X Short Communication 349 Increased training prevents the impairing
Brazilian Amygdaloid Journal non-nmda of Medical receptors and Biological and memory Research expression (1999) 32: 349-353 ISSN 0100-879X Short Communication 349 Increased training prevents the impairing
Stress, memory and the amygdala
 Stress, memory and the amygdala Benno Roozendaal, Bruce S. McEwen and Sumantra Chattarji Abstract Emotionally significant experiences tend to be well remembered, and the amygdala has a pivotal role in
Stress, memory and the amygdala Benno Roozendaal, Bruce S. McEwen and Sumantra Chattarji Abstract Emotionally significant experiences tend to be well remembered, and the amygdala has a pivotal role in
LESIONS OF THE MESOLIMBIC DOPAMINE SYSTEM DISRUPT SIGNALLED ESCAPE RESPONSES IN THE RAT
 ACTA NEUROBIOL: EXP. 1988, 48: 117-121 Short communication LESIONS OF THE MESOLIMBIC DOPAMINE SYSTEM DISRUPT SIGNALLED ESCAPE RESPONSES IN THE RAT W. Jeffrey WILSON and Jennifer C. HALL Department of Psychological
ACTA NEUROBIOL: EXP. 1988, 48: 117-121 Short communication LESIONS OF THE MESOLIMBIC DOPAMINE SYSTEM DISRUPT SIGNALLED ESCAPE RESPONSES IN THE RAT W. Jeffrey WILSON and Jennifer C. HALL Department of Psychological
An Animal Model of Flashbulb Memory: Insights into the Time-Dependent Mechanisms of Memory Enhancement
 University of South Florida Scholar Commons Graduate Theses and Dissertations Graduate School 11-12-2015 An Animal Model of Flashbulb Memory: Insights into the Time-Dependent Mechanisms of Memory Enhancement
University of South Florida Scholar Commons Graduate Theses and Dissertations Graduate School 11-12-2015 An Animal Model of Flashbulb Memory: Insights into the Time-Dependent Mechanisms of Memory Enhancement
AMAN WE MET RECENTLY was only a few blocks. Mechanisms of emotional arousal and lasting declarative memory R EVIEW. Larry Cahill and James L.
 R EVIEW F.M. Ashcroft and F.M. Gribble K ATP channel structure and function 38 Ueda, K., Inagaki, N. and Seino, S. (1997) J. Biol. Chem. 272, 22983 22986 39 Carson, M.R., Travis, S.M. and Welsh, M.J. (1995)
R EVIEW F.M. Ashcroft and F.M. Gribble K ATP channel structure and function 38 Ueda, K., Inagaki, N. and Seino, S. (1997) J. Biol. Chem. 272, 22983 22986 39 Carson, M.R., Travis, S.M. and Welsh, M.J. (1995)
PHYSIOLOGY AND MAINTENANCE Vol. III - Glucocorticoids and Brain - Natalia E. Ordyan and Vera G. Shalyapina
 GLUCOCORTICOIDS AND BRAIN Natalia E. Ordyan and Vera G. Shalyapina Pavlov Institute of Physiology, Russian Academy of Sciences, St.-Petersburg, Russia. Keywords: Adaptation, hypothalamic-pituitary-adrenal
GLUCOCORTICOIDS AND BRAIN Natalia E. Ordyan and Vera G. Shalyapina Pavlov Institute of Physiology, Russian Academy of Sciences, St.-Petersburg, Russia. Keywords: Adaptation, hypothalamic-pituitary-adrenal
FACTORS THAT DETERMINE THE NON- LINEAR AMYGDALA INFLUENCE ON HIPPOCAMPUS-DEPENDENT MEMORY
 Dose-Response: An International Journal Volume 4 Issue 1 Article 4 1-2006 FACTORS THAT DETERMINE THE NON- LINEAR AMYGDALA INFLUENCE ON HIPPOCAMPUS-DEPENDENT MEMORY Irit Akirav University of Haifa, Haifa,
Dose-Response: An International Journal Volume 4 Issue 1 Article 4 1-2006 FACTORS THAT DETERMINE THE NON- LINEAR AMYGDALA INFLUENCE ON HIPPOCAMPUS-DEPENDENT MEMORY Irit Akirav University of Haifa, Haifa,
The effects of acute restraint stress and dexamethasone on retrieval of long-term memory in rats: an interaction with opiate system
 Behavioural Brain Research 154 (2004) 193 198 Research report The effects of acute restraint stress and dexamethasone on retrieval of long-term memory in rats: an interaction with opiate system Ali Rashidy-Pour
Behavioural Brain Research 154 (2004) 193 198 Research report The effects of acute restraint stress and dexamethasone on retrieval of long-term memory in rats: an interaction with opiate system Ali Rashidy-Pour
The Amygdala Modulates Memory Consolidation of Fear-Motivated Inhibitory Avoidance Learning But Not Classical Fear Conditioning
 The Journal of Neuroscience, September 15, 2000, 20(18):7059 7066 The Amygdala Modulates Memory Consolidation of Fear-Motivated Inhibitory Avoidance Learning But Not Classical Fear Conditioning Ann E.
The Journal of Neuroscience, September 15, 2000, 20(18):7059 7066 The Amygdala Modulates Memory Consolidation of Fear-Motivated Inhibitory Avoidance Learning But Not Classical Fear Conditioning Ann E.
What is the Role of the Amygdala in Long Term Memory? Jack Pemment. University of Mississippi
 LT Memory and the Amygdala 1 Running Head: Role of the amygdala in long term memory What is the Role of the Amygdala in Long Term Memory? Jack Pemment University of Mississippi LT Memory and the Amygdala
LT Memory and the Amygdala 1 Running Head: Role of the amygdala in long term memory What is the Role of the Amygdala in Long Term Memory? Jack Pemment University of Mississippi LT Memory and the Amygdala
DAILY RHYTHMS OF GLUCOCORTICOIDS CONTROL THE CIRCADIAN EXPRESSION OF THE CLOCK PROTEIN, PERIOD2, IN OVAL NUCLEUS OF
 * 3 - Manuscript DAILY RHYTHMS OF GLUCOCORTICOIDS CONTROL THE CIRCADIAN EXPRESSION OF THE CLOCK PROTEIN, PERIOD2, IN OVAL NUCLEUS OF THE BED NUCLEUS OF THE STRIA TERMINALIS AND CENTRAL NUCLEUS OF THE AMYGDALA
* 3 - Manuscript DAILY RHYTHMS OF GLUCOCORTICOIDS CONTROL THE CIRCADIAN EXPRESSION OF THE CLOCK PROTEIN, PERIOD2, IN OVAL NUCLEUS OF THE BED NUCLEUS OF THE STRIA TERMINALIS AND CENTRAL NUCLEUS OF THE AMYGDALA
Food Intake After Diazepam, Morphine or Muscimol: Microinjections in the Nucleus Accumbens Shell
 PII S0091-3057(00)00220-3 Pharmacology Biochemistry and Behavior, Vol. 66, No. 2, pp. 429 434, 2000 2000 Elsevier Science Inc. Printed in the USA. All rights reserved 0091-3057/00/$ see front matter Food
PII S0091-3057(00)00220-3 Pharmacology Biochemistry and Behavior, Vol. 66, No. 2, pp. 429 434, 2000 2000 Elsevier Science Inc. Printed in the USA. All rights reserved 0091-3057/00/$ see front matter Food
Bidirectional Influence of Epinephrine on Hippocampal LTP via β-adrenergic Receptors
 Syracuse University SURFACE Syracuse University Honors Program Capstone Projects Syracuse University Honors Program Capstone Projects Spring 5-1-2014 Bidirectional Influence of Epinephrine on Hippocampal
Syracuse University SURFACE Syracuse University Honors Program Capstone Projects Syracuse University Honors Program Capstone Projects Spring 5-1-2014 Bidirectional Influence of Epinephrine on Hippocampal
ROLE OF THE ENDOCANNABINOID SYSTEM IN REGULATING GLUCOCORTICOID EFFECTS ON MEMORY FOR EMOTIONAL EXPERIENCES
 Neuroscience 204 (2012) 104 116 ROLE OF THE ENDOCANNABINOID SYSTEM IN REGULATING GLUCOCORTICOID EFFECTS ON MEMORY FOR EMOTIONAL EXPERIENCES P. ATSAK, a * B. ROOZENDAAL a AND P. CAMPOLONGO b a Department
Neuroscience 204 (2012) 104 116 ROLE OF THE ENDOCANNABINOID SYSTEM IN REGULATING GLUCOCORTICOID EFFECTS ON MEMORY FOR EMOTIONAL EXPERIENCES P. ATSAK, a * B. ROOZENDAAL a AND P. CAMPOLONGO b a Department
Functional Inactivation of the Amygdala before But Not after Auditory Fear Conditioning Prevents Memory Formation
 The Journal of Neuroscience, 1999, Vol. 19 RC48 1of5 Functional Inactivation of the Amygdala before But Not after Auditory Fear Conditioning Prevents Memory Formation Ann E. Wilensky, Glenn E. Schafe,
The Journal of Neuroscience, 1999, Vol. 19 RC48 1of5 Functional Inactivation of the Amygdala before But Not after Auditory Fear Conditioning Prevents Memory Formation Ann E. Wilensky, Glenn E. Schafe,
Stress and Emotion. Stressors are things that challenge homeostasis -- these challenges may be real or merely anticipated
 Stress and Emotion 1 Stressors are things that challenge homeostasis -- these challenges may be real or merely anticipated Stress responses are what the body does about it 2 1 Two broad stressor categories
Stress and Emotion 1 Stressors are things that challenge homeostasis -- these challenges may be real or merely anticipated Stress responses are what the body does about it 2 1 Two broad stressor categories
CURRICULUM VITA. Juan A. Salinas Summer 2012
 CURRICULUM VITA Juan A. Salinas Summer 2012 Birth Date: August 7, 1960 Address: 7809 Woodrow Avenue 4.212 Seay Psychology Building Austin, TX 78757 Austin, TX 78712 phone: (512) 232-4643 phone: (512) 614-8211
CURRICULUM VITA Juan A. Salinas Summer 2012 Birth Date: August 7, 1960 Address: 7809 Woodrow Avenue 4.212 Seay Psychology Building Austin, TX 78757 Austin, TX 78712 phone: (512) 232-4643 phone: (512) 614-8211
Amygdala modulation of hippocampal-dependent and caudate
 Proc. Nati. Acad. Sci. USA Vol. 91, pp. 8477-8481, August 1994 Neuroiology Amygdala modulation of hippocampal-dependent and caudate nucleus-dependent memory processes (d-ampheae/hppocampus/statm/lanig)
Proc. Nati. Acad. Sci. USA Vol. 91, pp. 8477-8481, August 1994 Neuroiology Amygdala modulation of hippocampal-dependent and caudate nucleus-dependent memory processes (d-ampheae/hppocampus/statm/lanig)
Amygdala Strengthening of Cortical Memory Representations
 Chapter 7 Amygdala Strengthening of Cortical Memory Representations Candice M. Chavez, James L. McGaugh and Norman M. Weinberger Additional information is available at the end of the chapter http://dx.doi.org/10.5772/53677
Chapter 7 Amygdala Strengthening of Cortical Memory Representations Candice M. Chavez, James L. McGaugh and Norman M. Weinberger Additional information is available at the end of the chapter http://dx.doi.org/10.5772/53677
George E. Farmer and Lucien T. Thompson* INTRODUCTION HIPPOCAMPUS 22: (2012)
 Learning-Dependent Plasticity of Hippocampal CA1 Pyramidal Neuron Postburst Afterhyperpolarizations and Increased Excitability After Inhibitory Avoidance Learning Depend Upon Basolateral Amygdala Inputs
Learning-Dependent Plasticity of Hippocampal CA1 Pyramidal Neuron Postburst Afterhyperpolarizations and Increased Excitability After Inhibitory Avoidance Learning Depend Upon Basolateral Amygdala Inputs
Stress disrupts response memory retrieval
 Psychoneuroendocrinology (2013) 38, 1460 1465 Available online at www.sciencedirect.com jou rn a l home pag e : ww w. el sev ier. com/ loca te /psyn eu en SHORT COMMUNICATION Stress disrupts response memory
Psychoneuroendocrinology (2013) 38, 1460 1465 Available online at www.sciencedirect.com jou rn a l home pag e : ww w. el sev ier. com/ loca te /psyn eu en SHORT COMMUNICATION Stress disrupts response memory
Physiology & Behavior 70 (2000) Laboratório de Psicologia Comparada, Universidade Estácio de Sá, Rio de Janeiro, Brazil b
 Physiology & Behavior 70 (2000) 243 247 Microinfusion of nefazodone into the basolateral nucleus of the amygdala enhances defensive behavior induced by NMDA stimulation of the inferior colliculus S. Maisonnette
Physiology & Behavior 70 (2000) 243 247 Microinfusion of nefazodone into the basolateral nucleus of the amygdala enhances defensive behavior induced by NMDA stimulation of the inferior colliculus S. Maisonnette
Supporting Online Material for
 www.sciencemag.org/cgi/content/full/330/6009/1408/dc1 Supporting Online Material for Paradoxical False Memory for Objects After Brain Damage Stephanie M. McTighe, Rosemary A. Cowell, Boyer D. Winters,
www.sciencemag.org/cgi/content/full/330/6009/1408/dc1 Supporting Online Material for Paradoxical False Memory for Objects After Brain Damage Stephanie M. McTighe, Rosemary A. Cowell, Boyer D. Winters,
Subjects. Thirty-five adult male Lister Hooded rats (Charles River, Kent, UK),
 Supplemental Material: Subjects. Thirty-five adult male Lister Hooded rats (Charles River, Kent, UK), weighing between 280 300 g at the beginning of the experiment, were housed singly in holding rooms
Supplemental Material: Subjects. Thirty-five adult male Lister Hooded rats (Charles River, Kent, UK), weighing between 280 300 g at the beginning of the experiment, were housed singly in holding rooms
Behavioral Neuroscience: Fear thou not. Rony Paz
 Behavioral Neuroscience: Fear thou not Rony Paz Rony.paz@weizmann.ac.il Thoughts What is a reward? Learning is best motivated by threats to survival? Threats are much better reinforcers? Fear is a prime
Behavioral Neuroscience: Fear thou not Rony Paz Rony.paz@weizmann.ac.il Thoughts What is a reward? Learning is best motivated by threats to survival? Threats are much better reinforcers? Fear is a prime
Posttraining optogenetic manipulations of basolateral amygdala activity modulate consolidation of inhibitory avoidance memory in rats
 Posttraining optogenetic manipulations of basolateral amygdala activity modulate consolidation of inhibitory avoidance memory in rats Mary L. Huff a,1, Rachel L. Miller a,1, Karl Deisseroth b,c,d, David
Posttraining optogenetic manipulations of basolateral amygdala activity modulate consolidation of inhibitory avoidance memory in rats Mary L. Huff a,1, Rachel L. Miller a,1, Karl Deisseroth b,c,d, David
Enhanced Norepinephrine Release in Hypothalamus from Locus Coeruleus in SHR
 Enhanced Norepinephrine Release in Hypothalamus from Locus Coeruleus in SHR Shingo KAWASAKI, M.D., Kazuo TAKEDA, M.D., Motoo TANAKA, M.D., Hiroshi ITOH, M.D., Masahiro HIRATA, M.D., Tetsuo NAKATA, M.D.,
Enhanced Norepinephrine Release in Hypothalamus from Locus Coeruleus in SHR Shingo KAWASAKI, M.D., Kazuo TAKEDA, M.D., Motoo TANAKA, M.D., Hiroshi ITOH, M.D., Masahiro HIRATA, M.D., Tetsuo NAKATA, M.D.,
Insular Cortex-Amygdala Dialogue During Taste Recognition Memory Formation
 The highlight for April is by Dr. Federico Bermudez-Rattoni from the Institute of Cellular Physiology, National University of Mexico in Mexico City, Mexico. Dr. Bermudez-Rattoni s work over the past 25
The highlight for April is by Dr. Federico Bermudez-Rattoni from the Institute of Cellular Physiology, National University of Mexico in Mexico City, Mexico. Dr. Bermudez-Rattoni s work over the past 25
Short communication NORADRENERGIC STIMULATION OF THE LATERAL HYPOTHALAMUS AS A REINFORCEMENT IN T MAZE LEARNING IN RATS
 ACTA NEUROBIOL. EXP. 1978, 38: ROM13 Short communication NORADRENERGIC STIMULATION OF THE LATERAL HYPOTHALAMUS AS A REINFORCEMENT IN T MAZE LEARNING IN RATS Jerzy CYTAWA and Edyta JURKOWLANIEC Department
ACTA NEUROBIOL. EXP. 1978, 38: ROM13 Short communication NORADRENERGIC STIMULATION OF THE LATERAL HYPOTHALAMUS AS A REINFORCEMENT IN T MAZE LEARNING IN RATS Jerzy CYTAWA and Edyta JURKOWLANIEC Department
JLC Lee: Memory reconsolidation mediates the strengthening of memories by additional learning
 JLC Lee: Memory reconsolidation mediates the strengthening of memories by additional learning 8 6 4 Cond1 Cond2 Test VEH ANI Session Supplementary Fig. 1. Protein synthesis inhibition after a second contextual
JLC Lee: Memory reconsolidation mediates the strengthening of memories by additional learning 8 6 4 Cond1 Cond2 Test VEH ANI Session Supplementary Fig. 1. Protein synthesis inhibition after a second contextual
NIH Public Access Author Manuscript Behav Neurosci. Author manuscript; available in PMC 2011 April 1.
 NIH Public Access Author Manuscript Published in final edited form as: Behav Neurosci. 2010 April ; 124(2): 294 299. doi:10.1037/a0018911. Role of Corticosterone in Trace and Delay Conditioned Fear- Potentiated
NIH Public Access Author Manuscript Published in final edited form as: Behav Neurosci. 2010 April ; 124(2): 294 299. doi:10.1037/a0018911. Role of Corticosterone in Trace and Delay Conditioned Fear- Potentiated
Amygdala Is Critical for Stress-Induced Modulation of Hippocampal Long-Term Potentiation and Learning
 The Journal of Neuroscience, July 15, 2001, 21(14):5222 5228 Amygdala Is Critical for Stress-Induced Modulation of Hippocampal Long-Term Potentiation and Learning Jeansok J. Kim, 1 Hongjoo J. Lee, 1 Jung-Soo
The Journal of Neuroscience, July 15, 2001, 21(14):5222 5228 Amygdala Is Critical for Stress-Induced Modulation of Hippocampal Long-Term Potentiation and Learning Jeansok J. Kim, 1 Hongjoo J. Lee, 1 Jung-Soo
PLATELET-ACTIVATING FACTOR AFFECTS NOCICEPTION IN RATS AT CEREBRAL SITES OF ACTION
 PLATELET-ACTIVATING FACTOR AFFECTS NOCICEPTION IN RATS AT CEREBRAL SITES OF ACTION Lisa A. Teather 1,2 & Richard J. Wurtman 1 1 Department of Brain and Cognitive Sciences, Massachusetts Institute of Technology,
PLATELET-ACTIVATING FACTOR AFFECTS NOCICEPTION IN RATS AT CEREBRAL SITES OF ACTION Lisa A. Teather 1,2 & Richard J. Wurtman 1 1 Department of Brain and Cognitive Sciences, Massachusetts Institute of Technology,
EFFECTS OF AMYGDALOID LESIONS ON PLASMA AND PITUITARY LEVELS OF LUTEINIZING HORMONE IN THE MALE DEERMOUSE
 EFFECTS OF AMYGDALOID LESIONS ON PLASMA AND PITUITARY LEVELS OF LUTEINIZING HORMONE IN THE MALE DEERMOUSE B. E. ELEFTHERIOU, A. J. ZOLOVICK and R. L. NORMAN Department of Zoology, Kansas State University,
EFFECTS OF AMYGDALOID LESIONS ON PLASMA AND PITUITARY LEVELS OF LUTEINIZING HORMONE IN THE MALE DEERMOUSE B. E. ELEFTHERIOU, A. J. ZOLOVICK and R. L. NORMAN Department of Zoology, Kansas State University,
Rapid elevations in limbic endocannabinoid content by glucocorticoid hormones in vivo
 Psychoneuroendocrinology (2010) 35, 1333 1338 available at www.sciencedirect.com journal homepage: www.elsevier.com/locate/psyneuen Rapid elevations in limbic endocannabinoid content by glucocorticoid
Psychoneuroendocrinology (2010) 35, 1333 1338 available at www.sciencedirect.com journal homepage: www.elsevier.com/locate/psyneuen Rapid elevations in limbic endocannabinoid content by glucocorticoid
SUPPLEMENTARY INFORMATION
 doi:10.1038/nature11436 Supplementary Figure 1. CRF peptide is localized cholinergic interneurons throughout the nucleus accumbens. Immunofluorescent images demonstrating localization of CRF peptide, tyrosine
doi:10.1038/nature11436 Supplementary Figure 1. CRF peptide is localized cholinergic interneurons throughout the nucleus accumbens. Immunofluorescent images demonstrating localization of CRF peptide, tyrosine
Behavioral effects of intra septal injections of adrenergic drugs in rats
 Physiological Psychology 1976, Vol. 4 (4), 433 438 Behavioral effects of intra septal injections of adrenergic drugs in rats JOHN E. KELSEY Indiana, University, Bloomington, Indiana 47401 Injections of
Physiological Psychology 1976, Vol. 4 (4), 433 438 Behavioral effects of intra septal injections of adrenergic drugs in rats JOHN E. KELSEY Indiana, University, Bloomington, Indiana 47401 Injections of
Stress and the aging brain
 Stress and the aging brain Stress and the aging brain: What are the issues? Aging makes us less able to adjust to change Reactions of elderly to change generate stress Stress response involves acute reactions
Stress and the aging brain Stress and the aging brain: What are the issues? Aging makes us less able to adjust to change Reactions of elderly to change generate stress Stress response involves acute reactions
UvA-DARE (Digital Academic Repository) A temporal perspective on stress hormones and memory Pu, Z. Link to publication
 UvA-DARE (Digital Academic Repository) A temporal perspective on stress hormones and memory Pu, Z. Link to publication Citation for published version (APA): Pu, Z. (2009). A temporal perspective on stress
UvA-DARE (Digital Academic Repository) A temporal perspective on stress hormones and memory Pu, Z. Link to publication Citation for published version (APA): Pu, Z. (2009). A temporal perspective on stress
Stress and emotional memory: a matter of timing
 Review Stress and emotional memory: a matter of timing Marian Joëls 1, Guillen Fernandez 2 and Benno Roozendaal 3 1 Department Neuroscience and Pharmacology, Division of Neuroscience, UMC Utrecht, Rudolf
Review Stress and emotional memory: a matter of timing Marian Joëls 1, Guillen Fernandez 2 and Benno Roozendaal 3 1 Department Neuroscience and Pharmacology, Division of Neuroscience, UMC Utrecht, Rudolf
Evidence that associative interactions between synapses during the induction of long-term potentiation occur within local dendritic domains
 Proc. Natl. Acad. Sci. USA Vol. 85, pp. 2368-2372, April 1988 Neurobiology Evidence that associative interactions between synapses during the induction of long-term potentiation occur within local dendritic
Proc. Natl. Acad. Sci. USA Vol. 85, pp. 2368-2372, April 1988 Neurobiology Evidence that associative interactions between synapses during the induction of long-term potentiation occur within local dendritic
Reciprocal Hunger-Regulating Circuits Involving Alphaand
 Proceedings of the National Academy of Sciences Vol. 67, No. 2, pp. 1063-1070, October 1970 Reciprocal Hunger-Regulating Circuits Involving Alphaand Beta-Adrenergic Receptors Located, Respectively, in
Proceedings of the National Academy of Sciences Vol. 67, No. 2, pp. 1063-1070, October 1970 Reciprocal Hunger-Regulating Circuits Involving Alphaand Beta-Adrenergic Receptors Located, Respectively, in
Behavioral Neuroscience: Fear thou not. Rony Paz
 Behavioral Neuroscience: Fear thou not Rony Paz Rony.paz@weizmann.ac.il Thoughts What is a reward? Learning is best motivated by threats to survival Threats are much better reinforcers Fear is a prime
Behavioral Neuroscience: Fear thou not Rony Paz Rony.paz@weizmann.ac.il Thoughts What is a reward? Learning is best motivated by threats to survival Threats are much better reinforcers Fear is a prime
Interactions between brainstem noradrenergic neurons and the nucleus accumbens shell in modulating memory for emotionally arousing events
 Research Interactions between brainstem noradrenergic neurons and the nucleus accumbens shell in modulating memory for emotionally arousing events Erin C. Kerfoot 1,2 and Cedric L. Williams 1,3 1 Department
Research Interactions between brainstem noradrenergic neurons and the nucleus accumbens shell in modulating memory for emotionally arousing events Erin C. Kerfoot 1,2 and Cedric L. Williams 1,3 1 Department
Brain Infusion Kit mm
 Brain Infusion Kit 1 3-5 mm 0004760 STERILE MATERIALS FOR TEN BRAIN INFUSIONS REFER TO ENCLOSED SPECIFICATIONS 7147600-5 2006 DURECT Corporation Printed in U.S.A. April 2006 CONTENTS: Ten brain infusion
Brain Infusion Kit 1 3-5 mm 0004760 STERILE MATERIALS FOR TEN BRAIN INFUSIONS REFER TO ENCLOSED SPECIFICATIONS 7147600-5 2006 DURECT Corporation Printed in U.S.A. April 2006 CONTENTS: Ten brain infusion
Amnesia produced by altered release of neurotransmitters after intraamygdala injections of a protein synthesis inhibitor
 Amnesia produced by altered release of neurotransmitters after intraamygdala injections of a protein synthesis inhibitor Clinton E. Canal*, Qing Chang, and Paul E. Gold* *Neuroscience Program, Departments
Amnesia produced by altered release of neurotransmitters after intraamygdala injections of a protein synthesis inhibitor Clinton E. Canal*, Qing Chang, and Paul E. Gold* *Neuroscience Program, Departments
G. M. SULLIVAN, a * J. APERGIS, b D. E. A. BUSH, b L. R. JOHNSON, b M. HOU b AND J. E. LEDOUX b. Neuroscience 128 (2004) 7 14
 Neuroscience 128 (2004) 7 14 LESIONS IN THE BED NUCLEUS OF THE STRIA TERMINALIS DISRUPT CORTICOSTERONE AND FREEZING RESPONSES ELICITED BY A CONTEXTUAL BUT NOT BY A SPECIFIC CUE-CONDITIONED FEAR STIMULUS
Neuroscience 128 (2004) 7 14 LESIONS IN THE BED NUCLEUS OF THE STRIA TERMINALIS DISRUPT CORTICOSTERONE AND FREEZING RESPONSES ELICITED BY A CONTEXTUAL BUT NOT BY A SPECIFIC CUE-CONDITIONED FEAR STIMULUS
A non-arousing test situation abolishes the impairing effects of cortisol on delayed memory retrieval in healthy women
 Neuroscience Letters 399 (2006) 268 272 A non-arousing test situation abolishes the impairing effects of cortisol on delayed memory retrieval in healthy women Sabrina Kuhlmann, Oliver T. Wolf Department
Neuroscience Letters 399 (2006) 268 272 A non-arousing test situation abolishes the impairing effects of cortisol on delayed memory retrieval in healthy women Sabrina Kuhlmann, Oliver T. Wolf Department
Is There Savings for Pavlovian Fear Conditioning after Neurotoxic Basolateral Amygdala Lesions in Rats?
 Neurobiology of Learning and Memory 76, 268 283 (2001) doi:10.1006/nlme.2001.4042, available online at http://www.idealibrary.com on Is There Savings for Pavlovian Fear Conditioning after Neurotoxic Basolateral
Neurobiology of Learning and Memory 76, 268 283 (2001) doi:10.1006/nlme.2001.4042, available online at http://www.idealibrary.com on Is There Savings for Pavlovian Fear Conditioning after Neurotoxic Basolateral
biological psychology, p. 40 The study of the nervous system, especially the brain. neuroscience, p. 40
 biological psychology, p. 40 The specialized branch of psychology that studies the relationship between behavior and bodily processes and system; also called biopsychology or psychobiology. neuroscience,
biological psychology, p. 40 The specialized branch of psychology that studies the relationship between behavior and bodily processes and system; also called biopsychology or psychobiology. neuroscience,
SHORT COMMUNICATION Retrograde amnesia for spatial information: a dissociation between intra and extramaze cues following hippocampus lesions in rats
 European Journal of Neuroscience, Vol. 10, pp. 3295 3301, 1998 European Neuroscience Association SHORT COMMUNICATION Retrograde amnesia for spatial information: a dissociation between intra and extramaze
European Journal of Neuroscience, Vol. 10, pp. 3295 3301, 1998 European Neuroscience Association SHORT COMMUNICATION Retrograde amnesia for spatial information: a dissociation between intra and extramaze
Hippocampus and Two-Way Active Avoidance Conditioning: Contrasting Effects of Cytotoxic Lesion and Temporary Inactivation
 HIPPOCAMPUS 25:1517 1531 (2015) and Two-Way Active Avoidance Conditioning: Contrasting Effects of Cytotoxic Lesion and Temporary Inactivation Jia Wang, 1 Tobias Bast, 2 * Yu-Cong Wang, 1 and Wei-Ning Zhang
HIPPOCAMPUS 25:1517 1531 (2015) and Two-Way Active Avoidance Conditioning: Contrasting Effects of Cytotoxic Lesion and Temporary Inactivation Jia Wang, 1 Tobias Bast, 2 * Yu-Cong Wang, 1 and Wei-Ning Zhang
Deficits in amygdaloid camp-responsive element binding protein signaling play a role in genetic predisposition to anxiety and alcoholism
 Research article Related Commentary, page 2697 Deficits in amygdaloid camp-responsive element binding protein signaling play a role in genetic predisposition to anxiety and alcoholism Subhash C. Pandey,
Research article Related Commentary, page 2697 Deficits in amygdaloid camp-responsive element binding protein signaling play a role in genetic predisposition to anxiety and alcoholism Subhash C. Pandey,
Endocannabinoid Signaling within the Basolateral Amygdala Integrates Multiple Stress Hormone Effects on Memory Consolidation
 (2015) 40, 1485 1494 & 2015 American College of. All rights reserved 0893-133X/15 www.neuropsychopharmacology.org Endocannabinoid Signaling within the Basolateral Amygdala Integrates Multiple Stress Hormone
(2015) 40, 1485 1494 & 2015 American College of. All rights reserved 0893-133X/15 www.neuropsychopharmacology.org Endocannabinoid Signaling within the Basolateral Amygdala Integrates Multiple Stress Hormone
Organization of the nervous system. [See Fig. 48.1]
![Organization of the nervous system. [See Fig. 48.1] Organization of the nervous system. [See Fig. 48.1]](/thumbs/90/103926552.jpg) Nervous System [Note: This is the text version of this lecture file. To make the lecture notes downloadable over a slow connection (e.g. modem) the figures have been replaced with figure numbers as found
Nervous System [Note: This is the text version of this lecture file. To make the lecture notes downloadable over a slow connection (e.g. modem) the figures have been replaced with figure numbers as found
Cannabinoid Receptor Activation in the Basolateral Amygdala Blocks the Effects of Stress on the Conditioning and Extinction of Inhibitory Avoidance
 11078 The Journal of Neuroscience, September 9, 2009 29(36):11078 11088 Behavioral/Systems/Cognitive Cannabinoid Receptor Activation in the Basolateral Amygdala Blocks the Effects of Stress on the Conditioning
11078 The Journal of Neuroscience, September 9, 2009 29(36):11078 11088 Behavioral/Systems/Cognitive Cannabinoid Receptor Activation in the Basolateral Amygdala Blocks the Effects of Stress on the Conditioning
Emotional Memory, PTSD, and Clinical Intervention Updates
 Emotional Memory, PTSD, and Clinical Intervention Updates Wen Cai, MD, Ph.D. Chief Medical Officer--La Frontera Arizona Clinical Associate Professor--Psychiatry and Psychology University of Arizona College
Emotional Memory, PTSD, and Clinical Intervention Updates Wen Cai, MD, Ph.D. Chief Medical Officer--La Frontera Arizona Clinical Associate Professor--Psychiatry and Psychology University of Arizona College
TITLE: The STRONG STAR Multidisciplinary PTSD Research Consortium
 AD Award Number: W81XWH-08-2-0118 TITLE: The STRONG STAR Multidisciplinary PTSD Research Consortium PRINCIPAL INVESTIGATOR: Randy Strong. Ph.D. Alan Frazer, Ph.D. David Morilak, Ph.D. CONTRACTING ORGANIZATION:
AD Award Number: W81XWH-08-2-0118 TITLE: The STRONG STAR Multidisciplinary PTSD Research Consortium PRINCIPAL INVESTIGATOR: Randy Strong. Ph.D. Alan Frazer, Ph.D. David Morilak, Ph.D. CONTRACTING ORGANIZATION:
Module H NERVOUS SYSTEM
 Module H NERVOUS SYSTEM Topic from General functions of the nervous system Organization of the nervous system from both anatomical & functional perspectives Gross & microscopic anatomy of nervous tissue
Module H NERVOUS SYSTEM Topic from General functions of the nervous system Organization of the nervous system from both anatomical & functional perspectives Gross & microscopic anatomy of nervous tissue
Supplemental Figure 1
 A C E Supplemental Figure 1 AP 1mm A+C 1mm Apo 1mm B D 1mm CGS F 1mm All 1mm Supplemental Figure 1. The locations of microinfusion cannulae. Each dot represents the cannula tip of a given rat in Figure.
A C E Supplemental Figure 1 AP 1mm A+C 1mm Apo 1mm B D 1mm CGS F 1mm All 1mm Supplemental Figure 1. The locations of microinfusion cannulae. Each dot represents the cannula tip of a given rat in Figure.
SUPPLEMENTARY INFORMATION
 doi: 10.1038/nature05772 SUPPLEMENTARY INFORMATION Supplemental figure 1. Enrichment facilitates learning. a. Images showing a home cage and a cage used for environmental enrichment (EE). For EE up to
doi: 10.1038/nature05772 SUPPLEMENTARY INFORMATION Supplemental figure 1. Enrichment facilitates learning. a. Images showing a home cage and a cage used for environmental enrichment (EE). For EE up to
EFFECTS OF NITRIC OXIDE SYNTHASE INHIBITOR N G -NITRO-L-ARGININE METHYL ESTER ON SPATIAL AND CUED LEANING
 Pergamon Neuroscience Vol. 83, No. 3, pp. 837 841, 1998 Copyright 1998 IBRO. Published by Elsevier Science Ltd Printed in Great Britain. All rights reserved PII: S0306-4522(97)00457-0 0306 4522/98 $19.00+0.00
Pergamon Neuroscience Vol. 83, No. 3, pp. 837 841, 1998 Copyright 1998 IBRO. Published by Elsevier Science Ltd Printed in Great Britain. All rights reserved PII: S0306-4522(97)00457-0 0306 4522/98 $19.00+0.00
Neural Communication. Central Nervous System Peripheral Nervous System. Communication in the Nervous System. 4 Common Components of a Neuron
 Neural Communication Overview of CNS / PNS Electrical Signaling Chemical Signaling Central Nervous System Peripheral Nervous System Somatic = sensory & motor Autonomic = arousal state Parasympathetic =
Neural Communication Overview of CNS / PNS Electrical Signaling Chemical Signaling Central Nervous System Peripheral Nervous System Somatic = sensory & motor Autonomic = arousal state Parasympathetic =
DISSOCIATING SPACE AND TRACE IN DORSAL AND VENTRAL HIPPOCAMPUS. Jennifer Lee Czerniawski. A thesis submitted to the. Graduate School-New Brunswick
 DISSOCIATING SPACE AND TRACE IN DORSAL AND VENTRAL HIPPOCAMPUS By Jennifer Lee Czerniawski A thesis submitted to the Graduate School-New Brunswick Rutgers, The State University of New Jersey in partial
DISSOCIATING SPACE AND TRACE IN DORSAL AND VENTRAL HIPPOCAMPUS By Jennifer Lee Czerniawski A thesis submitted to the Graduate School-New Brunswick Rutgers, The State University of New Jersey in partial
Cholecystokinin antagonist, proglumide, stimulates growth hormone release in the rat
 J. Biosci., Vol. 15, Number 1, March 1990, pp. 17 21. Printed in India. Cholecystokinin antagonist, proglumide, stimulates growth hormone release in the rat E. VIJAYAN* and S. M. McCANN Department of Physiology,
J. Biosci., Vol. 15, Number 1, March 1990, pp. 17 21. Printed in India. Cholecystokinin antagonist, proglumide, stimulates growth hormone release in the rat E. VIJAYAN* and S. M. McCANN Department of Physiology,
A role for amygdaloid PKA and PKC in the acquisition of long-term conditional fear memories in rats
 Behavioural Brain Research 114 (2000) 145 152 www.elsevier.com/locate/bbr Research report A role for amygdaloid PKA and PKC in the acquisition of long-term conditional fear memories in rats Ki A. Goosens
Behavioural Brain Research 114 (2000) 145 152 www.elsevier.com/locate/bbr Research report A role for amygdaloid PKA and PKC in the acquisition of long-term conditional fear memories in rats Ki A. Goosens
Memory reconsolidation mediates the strengthening of memories by additional learning Lee, Jonathan
 Memory reconsolidation mediates the strengthening of memories by additional learning Lee, Jonathan DOI: 10.1038/nn.2205 Document Version Peer reviewed version Citation for published version (Harvard):
Memory reconsolidation mediates the strengthening of memories by additional learning Lee, Jonathan DOI: 10.1038/nn.2205 Document Version Peer reviewed version Citation for published version (Harvard):
LIMBIC SYSTEM. Dr. Amani A. Elfaki Associate Professor Department of Anatomy
 LIMBIC SYSTEM Dr. Amani A. Elfaki Associate Professor Department of Anatomy Learning Objectives Define the limbic system Identify the parts of the limbic system Describe the circulation of the limbic system
LIMBIC SYSTEM Dr. Amani A. Elfaki Associate Professor Department of Anatomy Learning Objectives Define the limbic system Identify the parts of the limbic system Describe the circulation of the limbic system
Tracking the Fear Engram: The Lateral Amygdala Is an Essential Locus of Fear Memory Storage
 10010 The Journal of Neuroscience, October 26, 2005 25(43):10010 10015 Brief Communication Tracking the Fear Engram: The Lateral Amygdala Is an Essential Locus of Fear Memory Storage Glenn E. Schafe, 1
10010 The Journal of Neuroscience, October 26, 2005 25(43):10010 10015 Brief Communication Tracking the Fear Engram: The Lateral Amygdala Is an Essential Locus of Fear Memory Storage Glenn E. Schafe, 1
Effects of lesions of the nucleus accumbens core and shell on response-specific Pavlovian i n s t ru mental transfer
 Effects of lesions of the nucleus accumbens core and shell on response-specific Pavlovian i n s t ru mental transfer RN Cardinal, JA Parkinson *, TW Robbins, A Dickinson, BJ Everitt Departments of Experimental
Effects of lesions of the nucleus accumbens core and shell on response-specific Pavlovian i n s t ru mental transfer RN Cardinal, JA Parkinson *, TW Robbins, A Dickinson, BJ Everitt Departments of Experimental
A post-tetanic time window for the reinforcement of long-term potentiation by appetitive and aversive stimuli
 Proc. Natl. Acad. Sci. USA Vol. 94, pp. 1494 1499, February 1997 Neurobiology A post-tetanic time window for the reinforcement of long-term potentiation by appetitive and aversive stimuli (motivation water
Proc. Natl. Acad. Sci. USA Vol. 94, pp. 1494 1499, February 1997 Neurobiology A post-tetanic time window for the reinforcement of long-term potentiation by appetitive and aversive stimuli (motivation water
The Basolateral Nucleus of the Amygdala Is Necessary to Induce the Opposing Effects of Stressful Experience on Learning in Males and Females
 5290 The Journal of Neuroscience, May 14, 2008 28(20):5290 5294 Brief Communications The Basolateral Nucleus of the Amygdala Is Necessary to Induce the Opposing Effects of Stressful Experience on Learning
5290 The Journal of Neuroscience, May 14, 2008 28(20):5290 5294 Brief Communications The Basolateral Nucleus of the Amygdala Is Necessary to Induce the Opposing Effects of Stressful Experience on Learning
Supplemental Information. A Visual-Cue-Dependent Memory Circuit. for Place Navigation
 Neuron, Volume 99 Supplemental Information A Visual-Cue-Dependent Memory Circuit for Place Navigation Han Qin, Ling Fu, Bo Hu, Xiang Liao, Jian Lu, Wenjing He, Shanshan Liang, Kuan Zhang, Ruijie Li, Jiwei
Neuron, Volume 99 Supplemental Information A Visual-Cue-Dependent Memory Circuit for Place Navigation Han Qin, Ling Fu, Bo Hu, Xiang Liao, Jian Lu, Wenjing He, Shanshan Liang, Kuan Zhang, Ruijie Li, Jiwei
Declarative memory includes semantic, episodic, and spatial memory, and
 Gallo Taste Learning and Memory in Aging Milagros Gallo, PhD Declarative memory includes semantic, episodic, and spatial memory, and in humans involves conscious recall. 1 Visual recognition memory is
Gallo Taste Learning and Memory in Aging Milagros Gallo, PhD Declarative memory includes semantic, episodic, and spatial memory, and in humans involves conscious recall. 1 Visual recognition memory is
Review Article Stress and Memory: Behavioral Effects and Neurobiological Mechanisms
 Hindawi Publishing Corporation Neural Plasticity Volume 2007, Article ID 78970, 20 pages doi:10.1155/2007/78970 Review Article Stress and Memory: Behavioral Effects and Neurobiological Mechanisms Carmen
Hindawi Publishing Corporation Neural Plasticity Volume 2007, Article ID 78970, 20 pages doi:10.1155/2007/78970 Review Article Stress and Memory: Behavioral Effects and Neurobiological Mechanisms Carmen
Pattern Memory Involves Both Elemental and Configural Processes: Evidence From the Effects of Hippocampal Lesions
 Behavioral Neuroscience 2011 American Psychological Association 2011, Vol. 125, No. 4, 567 577 0735-7044/11/$12.00 DOI: 10.1037/a0023762 Pattern Memory Involves Both Elemental and Configural Processes:
Behavioral Neuroscience 2011 American Psychological Association 2011, Vol. 125, No. 4, 567 577 0735-7044/11/$12.00 DOI: 10.1037/a0023762 Pattern Memory Involves Both Elemental and Configural Processes:
Hippocampal Corticosterone Impairs Memory Consolidation During Sleep but Improves Consolidation in the Wake State
 HIPPOCAMPUS 24:510 515 (2014) RAPID COMMUNICATION Hippocampal Corticosterone Impairs Memory Consolidation During Sleep but Improves Consolidation in the Wake State Eduard Kelemen, 1 Marie Bahrendt, 2 Jan
HIPPOCAMPUS 24:510 515 (2014) RAPID COMMUNICATION Hippocampal Corticosterone Impairs Memory Consolidation During Sleep but Improves Consolidation in the Wake State Eduard Kelemen, 1 Marie Bahrendt, 2 Jan
Systems Neuroscience November 29, Memory
 Systems Neuroscience November 29, 2016 Memory Gabriela Michel http: www.ini.unizh.ch/~kiper/system_neurosci.html Forms of memory Different types of learning & memory rely on different brain structures
Systems Neuroscience November 29, 2016 Memory Gabriela Michel http: www.ini.unizh.ch/~kiper/system_neurosci.html Forms of memory Different types of learning & memory rely on different brain structures
Time your stress if you aim for success:
 FLOOR VAN DEN BRAND Time your stress if you aim for success: different memory phases is still unclear. The aim was therefore to review existing literature on the topic and to determine whether memory is
FLOOR VAN DEN BRAND Time your stress if you aim for success: different memory phases is still unclear. The aim was therefore to review existing literature on the topic and to determine whether memory is
Separate basolateral amygdala projections to the hippocampal formation differentially modulate the consolidation of contextual and emotional learning
 University of Iowa Iowa Research Online Theses and Dissertations Fall 2016 Separate basolateral amygdala projections to the hippocampal formation differentially modulate the consolidation of contextual
University of Iowa Iowa Research Online Theses and Dissertations Fall 2016 Separate basolateral amygdala projections to the hippocampal formation differentially modulate the consolidation of contextual
Failure to defend against an environmental threat such as
 Persistence of fear memory across time requires the basolateral amygdala complex Andrew M. Poulos 1, Veronica Li, Sarah S. Sterlace, Fonda Tokushige, Ravikumar Ponnusamy, and Michael S. Fanselow Department
Persistence of fear memory across time requires the basolateral amygdala complex Andrew M. Poulos 1, Veronica Li, Sarah S. Sterlace, Fonda Tokushige, Ravikumar Ponnusamy, and Michael S. Fanselow Department
Role of the anterior cingulate cortex in the control over behaviour by Pavlovian conditioned stimuli
 Role of the anterior cingulate cortex in the control over behaviour by Pavlovian conditioned stimuli in rats RN Cardinal, JA Parkinson, H Djafari Marbini, AJ Toner, TW Robbins, BJ Everitt Departments of
Role of the anterior cingulate cortex in the control over behaviour by Pavlovian conditioned stimuli in rats RN Cardinal, JA Parkinson, H Djafari Marbini, AJ Toner, TW Robbins, BJ Everitt Departments of
Acute effect of cholecystokinin on short-term synaptic plasticity in the rat hippocampus
 Research in Pharmaceutical Sciences, October 2014; 9(5): 331-336 Received: Aug 2013 Accepted: Sep 2013 School of Pharmacy & Pharmaceutical Sciences Isfahan University of Medical Sciences Original Article
Research in Pharmaceutical Sciences, October 2014; 9(5): 331-336 Received: Aug 2013 Accepted: Sep 2013 School of Pharmacy & Pharmaceutical Sciences Isfahan University of Medical Sciences Original Article
Nature Neuroscience: doi: /nn Supplementary Figure 1. Diverse anorexigenic signals induce c-fos expression in CEl PKC-δ + neurons
 Supplementary Figure 1 Diverse anorexigenic signals induce c-fos expression in CEl PKC-δ + neurons a-c. Quantification of CEl c-fos expression in mice intraperitoneal injected with anorexigenic drugs (a),
Supplementary Figure 1 Diverse anorexigenic signals induce c-fos expression in CEl PKC-δ + neurons a-c. Quantification of CEl c-fos expression in mice intraperitoneal injected with anorexigenic drugs (a),
1971). The injection of 6-OH-dopamine results in selective degeneration. puberty. The local microinjection of 6-OH-dopamine into the locus coeruleus
 Central Research Division, Postgraduate Medical School, Budapest, Hungary EFFECT OF THE DEPLETION OF BRAIN NORADRENALINE ON THE PLASMA FSH AND GROWTH HORMONE LEVELS IN OVARIECTOMIZED RATS By E. Endr\l=o"\czi,I.
Central Research Division, Postgraduate Medical School, Budapest, Hungary EFFECT OF THE DEPLETION OF BRAIN NORADRENALINE ON THE PLASMA FSH AND GROWTH HORMONE LEVELS IN OVARIECTOMIZED RATS By E. Endr\l=o"\czi,I.
