Enantioseparation condition of D,L-tryptophan using ligand exchange chromatography
|
|
|
- Millicent Flowers
- 6 years ago
- Views:
Transcription
1 Indian Journal of Chemistry Vol. 47A, January 2008, pp Enantioseparation condition of D,L-tryptophan using ligand exchange chromatography Hongyuan Yan & Kyung Ho Row* Center for Advanced Bioseparation Technology, Department of Chemical Engineering, Inha University, Incheon, , Korea Received 7 March 2007; revised 15 November 2007 Chiral recognition mechanism and enantioseparation conditions of tryptophan enantiomers using ligand exchange chromatography are reported here. The effect of different kinds and concentration of ligands, bivalent copper ion, organic modifier, ph of mobile phase, and temperature on enantioseparation has been evaluated. The results show that the enantioselectivity is strongly affected by the ph and the ligand concentration. Under the optimum condition, baseline separation of the two enantiomers has been obtained on a C 18 column with a resolution of 3.42 in less than 30 min using methanol-water solution (20:80 v/v) as mobile phase containing 3.0 mmol L -1 L- phenylalanine and 0.5 mmol L -1 copper sulphate. Thermodynamic data (ΔΔH and ΔΔS) obtained by Van t Hoff plots reveal that the enantioseparation is an enthalpy-controlled process. The mechanism of chiral discrimination is based on the stabilities of the copper(ii) binary complexes and their ternary diasteremeric complexes with amino acids formed in solution and stationary phase. The proposed method has been successfully used for the quality evaluation of tryptophan enantiomers. IPC Code: Int Cl. 8 G01N 30/00 Amino acids are essential components of many natural molecules and frequently found in food, feeds, body fluids and tissues. Chiral analysis of D,L-amino acids is of great interest in life science and many other related fields. L-tryptophan [L-2-amino-3-(indol-3- yl)propionic acid] is a vital constituent of proteins and indispensable in human nutrition for establishing and maintaining a positive nitrogen balance. As an essential amino acid, L-tryptophan is also considered as the precursor of the neurotransmitter serotonin, which plays an important role in brain function and related regulatory mechanisms 1-4. However, D- tryptophan does not show these biological functions and is used as pharmaceutical intermediate. Enantioseparation of racemic amino acids has been extensively studied by gas chromatography and HPLC using chiral stationary phases or chiral mobile phase additives Among the chromatographic methods developed thus far, the HPLC methods using chiral stationary phase require a special chiral column, which is expensive and can only separate a couple of enantiomers in most cases 11. In contrast, HPLC methods based on chiral mobile phase additives (such as β-cyclodextrin or chiral ligand salts) are not only efficient tools for the separation of racemic drugs but also relatively cheap and feasible 12. Ligand exchange chromatography exploits the rapid and reversible formation of metal ion complexes to separate compounds, which can donate electrons and coordinate to the immobilized metal ions. The enantioselectivity is based on the formation of a kind of ternary complex which conjuncts with an achiral stationary phase to generate an equilibrium-secondary chemical equilibrium Any difference in the stability or energy of these diasteromeric complexes will affect the chemical equilibrium resulting in different chromatographic behaviour. As a result, the enantiomers can be separated on a conventional HPLC column. Retention of a given species is directly related to the stability of the mixed ligand complex it forms with the metal ion immobilized on a chromatographic support Ligand exchange with soluble metal complexes, that partition between the mobile and stationary phases, also affects enantioseparation. We report herein the chiral recognition mechanism and enantioseparation conditions of tryptophan enantiomers on a C 18 column using chiral mobile phase additives. The effect of different kinds and concentration of ligands, bivalent copper ion, organic modifier, ph of mobile phase, and temperature on enantioseparation have been evaluated. The chiral recognition mechanisms and thermodynamic characteristic are also discussed. Experimental L-phenylalanine, L-serine, L-alanine, L-leucine and L-histidine were purchased from Tokyo Kasei Kogyo Co. LTD (Japan). L-tryptophan and D- tryptophan were obtained from Sigma (ST Louis, MO, USA). Anhydrous cupric sulfate (Extra Pure grade) was purchased from TEDIA Company (USA). Acetonitrile, tetrahydrofuran, and methanol were all of HPLC grade and were obtained from Duksan Pure Chemical Co. (Ansan, Korea). Double distilled water
2 76 INDIAN J CHEM, SEC A, JANUARY 2008 was filtered with a 0.45 μm filter membrane before use. Chiral ligand-exchange RP-HPLC system HPLC analysis was performed using a liquid chromatography system containing a Waters 600s Multisolvent Delivery System and a Waters 616 pump (Waters, Milford, MA, USA), a Waters 486 Tunable Absorbance UV detector (Waters, Milford, MA, USA), and a Rheodyne injection valve (20 μl sample loop). Autochro 2000 data software (Younglin Co., Korea) was used as a data acquisition system. The analytical column was packed with C 18 stationary phase (OptimaPak 150 mm 4.6 mm I.D., particle size 5 µm, RStech, Daejeon, Korea). The solution of chiral mobile phase additive (CMPA) comprised of 3.0 mmol L -1 L-phenylalanine mixed with 0.5 mmol L -1 cupric sulfate in water. The mobile phase consisted of CMPA solution-methanol (80:20, v/v). The flow-rate of the mobile phase was set at 1.0 ml min -1. Chromatographic assay was carried out at ambient temperature. UV wavelength was set at 280 nm. The retention factor was calculated from the equation k = (t R -t 0 )/t 0, where t R and t 0 are the retention times of analyte and unretained solutes, respectively. The enantioseparation factor was calculated from the equation α=k L /k D, where k L and k D are the retention factors of L-tryptophan and D-tryptophan, respectively. Resolution (R) was calculated from the equation R=2(t L -t D )/(w D +w L ), where t L and t D are the retention times of L-tryptophan and D-tryptophan, respectively, and w D and w L are the baseline peak widths of the two enantiomers. The number of theoretical plates (N) was calculated by the equation N=16(t R /w) 2. Results and discussion Mechanism of enantioselectivity According to the three-point interaction rule of chiral recognition in ligand-exchange chromatography 18, at least two points of interaction are available for both tryptophan enantiomers: simultaneous coordination of the amine and carboxylate groups to the metal ion. A good selector that discriminates between the two enantiomers should therefore stabilize the third interaction with one of the enantiomers to the maximum possible extent. Chiral selectivity can also be achieved if the third interaction is destabilized for one of the enantiomers relative to the other. Furthermore, the difference in stability of the diastereomeric ternary complexes in aqueous phase has an influence on enantioselectivity and the chiral recognition takes place on the column where the initial complex is adsorbed. Bivalent copper cation was selected due to the rapid formation and excellent stability of its diastereomeric complexes. First, L-phenylalanine and bivalence metal ion (Cu 2+ ) formed a complex, and then incorporated with tryptophan to produce a ternary complex by the carboxylic group and its neighbouring amino group. As tryptophan has two enantiomers, D-tryptophan and L-tryptophan; so, two kinds of ternary complexes with different configurations are formed. The enantioselectivity depends on the differences in the relative stabilities, the energy and the affinity to the stationary phase of cis- and trans- complexes. The complexes formed in mobile phase could conjunct with the stationary phase to generate an equilibrium-secondary chemical equilibrium. Any difference such as stability or energy of these diasteromeric complexes will affect the chemical equilibrium, thereby resulting in their different chromatographic behaviours. As a result, the enantiomers can be separated on a C 18 column (Fig. 1). Effect of different ligands on enantioselectivity An important component of the recognition process is the structure and property of the chiral ligand. In order to investigate the effect of different ligands on the enantioselectivity, L-phenylalanine, L-serine, L-alanine, L-leucine and L-histidine were explored for the enantioseparation of D and L-tryptophan under the same experimental conditions For amino acids Intensity C D L Time (min) Fig. 1 Chromatogram of tryptophan enantioseparation by ligand exchange HPLC (Mobile phase: methanol-water (20:80, v/v, containing 3.0 mmol L -1 L-phenylalanine and 0.5 mmol L -1 cupric sulfate; flow rate: 1.0 ml/min; UV wavelength: 280 nm). 50
3 NOTES 77 Different ligands Table 1 Effect of different ligands on tryptophan enantioseparation Retention factor Selectivity Resolution Efficiency L-phenylalanine L-serine L-alanine L-histidine L-leucine α k D k L (α) (R) N D N L Methanol in mobile phase (%) 13 Effect of L-phenylalanine Effect of methanol Effect of Cu Concentration of L-phenylalanine/Cu 2+ (mmol/l) Fig. 2 Effect of L-phenylalanine, Cu 2+, and methanol concentration on tryptophan enantioseparation (a: enantioseparation factor of the tryptophan enantiomers; flow rate: 1.0 ml/min; UV wavelength: 280 nm). with smaller or more flexible side chains as chiral ligand reagent, there was little enantioselectivity. L-phenylalanine (R=3.51) showed better resolution than L-histidine (R=0.81) and L-leucine (R=1.82) (Table 1). No enantioselectivity was observed if L- alanine and L-serine were used as ligand reagents. It appeared that the ligand for enantioseparation should possess a larger group to produce space exclude function and also should possess certain lipophilia to be retained by the reverse stationary phase. A consistent trend of higher enantioselectivity with increasing size of the side group was evident, indicating that the size of amino acid side chain is an important factor in determining enantioselectivity in the ligand exchange chromatography. Effects of the organic modifier on enantioseparation The effect of organic modifier on the tryptophan enantioseparation was more significant with less polar organic modifier (acetonitrile and tetrahydrofuran in this case). Tetrahydrofuran as organic modifier hardly showed any enantioselectivity while acetonitrile gave better enantioselectivity (R=0.94) in low flow rate (0.5 ml min -1 ). The best enantioselectivity was obtained by using methanol as organic modifier. Figure 2 shows that both the retention and separation of the two enantiomers decreased with increasing methanol content. Increasing the organic modifier could decrease the polarity of the mobile phase and consequently diminish the hydrophilic interaction of analytes with the mobile phase. Moreover, the retention ability of L-enantiomers is diminished than D-enantiomers because the lipophilic interaction is more effective in the more stable ternary complex. Although the enantioselectivity increases with a decreased amount of organic modifier, the retention time is longer than 70 min when the concentration of methanol is below 10%. Considering the retention time and enantioselectivity, 20% methanol was selected as the organic modifier for further studies. Effects of ligand concentration on enantioseparation The effect of L-phenylalanine concentration on the enantioselectivity was investigated in a range of 0 to 8.0 mmol L -1 and the results are shown in Fig. 2. With the increase in concentration of L-phenylalanine, the value of k and α increased, and the value of α tended to remain constant when the concentration of ligand was above 3.0 mmol L -1. Increasing the ligand concentration in the mobile phase could result in the formation of more ligand complexes. These complexes could be distributed on the stationary phase. As a result, the retention and separation of the enantiomers could be increased. When the ligand concentration reached a certain threshold (3.0 mmol L -1 ), the above parameter tended to remain constant, and the two enantiomers could be baseline separated within 30 min. Effects of bivalent copper ion concentration Different concentrations of Cu 2+ were investigated in a range from 0 to 6.0 mmol L -1. Figure 2 shows that with the decrease in Cu 2+ concentration in the mobile phase, both retention and separation of the two enantiomers increased. Additionally, R of the two
4 78 INDIAN J CHEM, SEC A, JANUARY lnk D lnk L lna 0.25 lnk lna /T Fig. 3 Van t Hoff plots by plotting ln k versus 1/T (k L and k D are the retention factors of L-tryptophan and D-tryptophan; α is the enantioseparation factor of the tryptophan enantiomers; T: temperature, K) enantiomers increased from 1.81 to 3.53 when Cu 2+ concentration decreased from 6.0 to 0.3 mmol L -1. Although decrease the Cu 2+ concentration in mobile phase could increase the retention and separation of both D- and L-tryptophan continuously, the elution time of tryptophan enantiomers was longer than 30 min if the concentration of Cu 2+ was lower than 0.5 mmol L -1. Considering the retention and separation, 0.5 mmol L -1 was used as the optimum Cu 2+ concentration. Effects of ph of the mobile phase ph-dependence of the enantioselectivity was investigated in a ph range 4.1 to 5.2 using phosphoric acid, hydrochloric acid, and trifluoroacetic acid to adjust ph. In contrast to reports that changing the ph of the mobile phase hardly influenced the resolution 20, our results showed that the resolution of tryptophan enantiomers distinctly decreased from 3.42 to 0 when the ph of the mobile phase was lowered from 4.9 to 4.1. Also, no enantioselectivity was observed at lower ph, which was due to the increased acid in mobile phase destroying the stereo structures of the ternary diasteremeric complex. When the ph of the mobile phase exceeds 5.0, Cu 2+ precipitates and blocks the chromatographic systems. Hence, 4.9 were chosen as the optimum ph of the mobile phase. Effect of temperature on the enantioseparation The temperature-dependence of the enantioseparation was investigated in the range 20 C to 50 C. At lower temperature, the formation of the two transient diastereomeric ternary complexes is expected to be more favourable. Consequently, retention times improve at lower temperature. In addition, the formation of more stable ternary complex is expected to be more favorable than that of the less stable ternary complex at lower temperature. The difference in the stability of the two diastereomeric ternary complexes increases and the separation factors increase as the temperature decreases. However, R only slightly changes from 3.42 to 3.36 while temperature increases from 20 C to 50 C, which is due to the rate of equilibrium for the formation of ternary complexes is slow at lower temperature. Thus, the lifetime of the transient diastereomeric ternary complex is quite long at lower temperature and, consequently, the chromatographic peaks corresponding to the two enantiomers broaden. Thermodynamics of enantioseparation The retention behaviour and thermodynamic parameters determined in this study were used to estimate the enthalpy, entropy, and Gibbs free energy of association between the two enantiomers and the C 18 stationary phase. Data obtained from retention and separations at temperature ranging from 20 C to 50 C were processed using the Van t Hoff equation to estimate the thermodynamic properties of the separation: ΔG ΔH ΔS ln k = = + RT RT R
5 NOTES 79 Table 2 Thermodynamic parameters of tryptophan enantioseparation Analytes ΔH (J.mol -1 ) ΔS (J.mol -1.K -1 ) ΔΔH (J.mol -1 ) ΔΔS (J.mol -1.K -1 ) D-tryptophan L-tryptophan ΔH and ΔS were obtained from linear regression of the Van t Hoff plots by plotting ln k versus 1/T; ΔΔH and ΔΔS were obtained from plot of ln a versus 1/T. ΔΔG ΔΔH ln a = = RT RT ΔΔS + R where R and T are the gas constant and the absolute temperature (Fig. 3). Enthalpy ( H) and entropy ( S), enthalpy difference ( H) and entropy difference ( S) can be calculated form the slopes and intercepts of linear portion of above equation. The results are listed in Table 2. The absolute values of H and S of L-tryptophan were larger than D-tryptophan indicating that L- tryptophan has stronger affinity to the stationary phase during the separation. Moreover, the finding that H > T S indicates that the chiral separation on the C 18 column is an enthalpy-controlled process. In general, for the temperature range C. The enthalpic contribution to the overall substrates transfer energy of the substrates was found to be more significant than the entropic contribution. The decrease in temperature led to an increase in the separation factors, which is consistent with the results of the temperature-dependence studies. Quantitative determination In order to assure the reliability of the assay, calibration graphs were constructed by using the areas of the chromatographic peaks measured at eight increasing concentrations in the range 1.0 to 200 mg L -1 for the both tryptophan enantiomers. Good linearity throughout the concentration is obtained for both enantiomers and the linear correlation equations were: Y= 84.73X for L-tryptophan and Y=81.66X for D-tryptophan with coefficient of regression (r) more than 0.999, respectively. Precision and accuracy were assessed by performing replicate analyses of quality control samples against calibration standards and were calculated as the relative standard deviation (RSD). The procedure was repeated on the same day and four different days on the same spiked standards at concentrations in the range of standard series. The intra-assay relative standard deviations and inter-assay relative standard deviations of the proposed method, determined on the basis of peak-area for three replications, were lower than 0.67% and 1.21% for D-tryptophan and 0.59% and 1.86 % for L-tryptophan, respectively. Conclusions The proposed method can be used for the quality evaluation of tryptophan enantiomers. The results show that the enantioselectivity is strongly affected by the ph of mobile phase and the ligand concentration. The selectivity of tryptophan enantioseparation depends on the stabilities of the ternary diasteremeric complexes. Baseline separation of the two enantiomers with a resolution of 3.42 in less than 30 min can be obtained on the C 18 column. Thermodynamic data ( H and S) obtained by Van t Hoff plots reveal that the enantioseparation is an enthalpy-controlled process. Acknowledgement The authors gratefully acknowledge the financial support from the center for Advanced Bioseparation Technology, Inha University. References 1 Young S N, Neurosci Biobehav Rev, 20 (1996) Capuron L, Ravand A, Neveu P J, Miller A H, Maes M & Dantzer R, Mol Psychiatr, 7 (2002) Riemann D, Feige B, Hornyak M, Koch S, Hohagen F & Voderholzer U, Psychiatr Res, 109 (2002) Hughes J H, Gallagher P & Young A H, Eur Neuropsychopharmacol, 12 (2002) Chang S C, Charles R & Gil-Av E, J Chromatogr A, 235 (1982) Hobo T, Suzuki S, Watabe K & Gil-Av E, Anal Chem, 57 (1985) Kim K & Lee K, Biotechnol Bioprocess Eng, 5 (2000) Garnier F, Randon J & Rocca J L, Talanta, 51 (2000) Nakamura M, Kiyohara S, Salto K, Suglta K & Sugo T, J Chromatogr A, 822 (1998) Slama I, Jourdan E, Grosset C, Ravel A, Villet A & Peyrin E, J Chromatogr B, 795 (2003) Peter A, Torok G, Armstrong D W, Toth G & Tourwe D, J Chromatogr A, 904 (2000) 1
6 80 INDIAN J CHEM, SEC A, JANUARY Taylor D R & Maher K, J Chromatogr Sci, 30 (1992) Hyun M H, Han S C, Lee C W & Lee Y K, J Chromatogr A, 950 (2002) Schmid M G, Schreiner K, Reisinger D & Gubitz G, J Sep Sci, 29 (2006) Davankov V A, J Chromatogr A, 1000 (2003) Wachsmann M & Brueckner H, Chromatographia, 47 (1998) Chen Z, Lin J, Uchiyama K & Hobo T, Anal Sci, 16 (2000) Zheng Z X, Lin J M & Qu F, J Chromatogr A, 1007 (2003) Hyun M H, Han S C & Whangbo S H, J Chromatogr A, 992 (2003) Zeng S, Zhong L, Pan L & Li Y, J Chromatogr A, 728 (1999) 151.
Introduction. and aryl-α-amino ketones, 11
 Enantiomeric Resolution of γ-amino Acids Bull. Korean Chem. Soc. 2011, Vol. 32, No. 8 3017 DOI 10.5012/bkcs.2011.32.8.3017 Liquid Chromatographic Resolution of Vigabatrin and Its Analogue γ-amino Acids
Enantiomeric Resolution of γ-amino Acids Bull. Korean Chem. Soc. 2011, Vol. 32, No. 8 3017 DOI 10.5012/bkcs.2011.32.8.3017 Liquid Chromatographic Resolution of Vigabatrin and Its Analogue γ-amino Acids
International Journal of Pharma and Bio Sciences
 International Journal of Pharma and Bio Sciences RESEARCH ARTICLE PHARMACEUTICAL ANALYSIS DEVELOPMENT AND VALIDATION OF LIQUID CHROMATOGRAPHIC METHOD FOR ESTIMATION OF ESCITALOPRAM OXALATE IN TABLET DOSAGE
International Journal of Pharma and Bio Sciences RESEARCH ARTICLE PHARMACEUTICAL ANALYSIS DEVELOPMENT AND VALIDATION OF LIQUID CHROMATOGRAPHIC METHOD FOR ESTIMATION OF ESCITALOPRAM OXALATE IN TABLET DOSAGE
RP-HPLC Analysis of Temozolomide in Pharmaceutical Dosage Forms
 Asian Journal of Chemistry Vol. 22, No. 7 (2010), 5067-5071 RP-HPLC Analysis of Temozolomide in Pharmaceutical Dosage Forms A. LAKSHMANA RAO*, G. TARAKA RAMESH and J.V.L.N.S. RAO Department of Pharmaceutical
Asian Journal of Chemistry Vol. 22, No. 7 (2010), 5067-5071 RP-HPLC Analysis of Temozolomide in Pharmaceutical Dosage Forms A. LAKSHMANA RAO*, G. TARAKA RAMESH and J.V.L.N.S. RAO Department of Pharmaceutical
Asian Journal of Pharmaceutical Analysis and Medicinal Chemistry Journal home page:
 Research Article CODEN: AJPAD7 ISSN: 2321-0923 Asian Journal of Pharmaceutical Analysis and Medicinal Chemistry Journal home page: www.ajpamc.com ANALYTICAL METHOD DEVELOPMENT AND VALIDATION OF GEFITINIB
Research Article CODEN: AJPAD7 ISSN: 2321-0923 Asian Journal of Pharmaceutical Analysis and Medicinal Chemistry Journal home page: www.ajpamc.com ANALYTICAL METHOD DEVELOPMENT AND VALIDATION OF GEFITINIB
IJPAR Vol.3 Issue 4 Oct-Dec-2014 Journal Home page:
 IJPAR Vol.3 Issue 4 Oct-Dec-2014 Journal Home page: ISSN: 2320-2831 Research article Open Access Method development and validation of tenofovir disoproxil fumerate and emtricitabine in combined tablet
IJPAR Vol.3 Issue 4 Oct-Dec-2014 Journal Home page: ISSN: 2320-2831 Research article Open Access Method development and validation of tenofovir disoproxil fumerate and emtricitabine in combined tablet
F. Al-Rimawi* Faculty of Science and Technology, Al-Quds University, P.O. Box 20002, East Jerusalem. Abstract
 JJC Jordan Journal of Chemistry Vol. 4 No.4, 2009, pp. 357-365 Development and Validation of Analytical Method for Fluconazole and Fluconazole Related Compounds (A, B, and C) in Capsule Formulations by
JJC Jordan Journal of Chemistry Vol. 4 No.4, 2009, pp. 357-365 Development and Validation of Analytical Method for Fluconazole and Fluconazole Related Compounds (A, B, and C) in Capsule Formulations by
Development, Estimation and Validation of Lisinopril in Bulk and its Pharmaceutical Formulation by HPLC Method
 ISSN: 0973-4945; CODEN ECJAO E- Chemistry http://www.e-journals.net 2012, 9(1), 340-344 Development, Estimation and Validation of Lisinopril in Bulk and its Pharmaceutical Formulation by PLC Method V.
ISSN: 0973-4945; CODEN ECJAO E- Chemistry http://www.e-journals.net 2012, 9(1), 340-344 Development, Estimation and Validation of Lisinopril in Bulk and its Pharmaceutical Formulation by PLC Method V.
Available online at Scholars Research Library
 Available online at www.scholarsresearchlibrary.com Scholars Research Library Der Pharmacia Lettre, 2015, 7 (3):157-161 (http://scholarsresearchlibrary.com/archive.html) ISSN 0975-5071 USA CODEN: DPLEB4
Available online at www.scholarsresearchlibrary.com Scholars Research Library Der Pharmacia Lettre, 2015, 7 (3):157-161 (http://scholarsresearchlibrary.com/archive.html) ISSN 0975-5071 USA CODEN: DPLEB4
International Journal of Pharma and Bio Sciences DEVELOPMENT AND VALIDATION OF RP-HPLC METHOD FOR THE ESTIMATION OF STRONTIUM RANELATE IN SACHET
 International Journal of Pharma and Bio Sciences RESEARCH ARTICLE ANALYTICAL CHEMISTRY DEVELOPMENT AND VALIDATION OF RP-HPLC METHOD FOR THE ESTIMATION OF STRONTIUM RANELATE IN SACHET K.MYTHILI *, S.GAYATRI,
International Journal of Pharma and Bio Sciences RESEARCH ARTICLE ANALYTICAL CHEMISTRY DEVELOPMENT AND VALIDATION OF RP-HPLC METHOD FOR THE ESTIMATION OF STRONTIUM RANELATE IN SACHET K.MYTHILI *, S.GAYATRI,
Reverse Phase HPLC Analysis of Atomoxetine in Pharmaceutical Dosage Forms
 Asian Journal of Chemistry Vol. 21, No. 2 (2009), 829-833 Reverse Phase HPLC Analysis of Atomoxetine in Pharmaceutical Dosage Forms B.V.V.S. JAGADEESH, S. SATYANARAYANA RAJU, V.JAYATHIRTHA RAO and J.V.L.N.
Asian Journal of Chemistry Vol. 21, No. 2 (2009), 829-833 Reverse Phase HPLC Analysis of Atomoxetine in Pharmaceutical Dosage Forms B.V.V.S. JAGADEESH, S. SATYANARAYANA RAJU, V.JAYATHIRTHA RAO and J.V.L.N.
A Validated Chiral Liquid Chromatographic Method for The Enantiomeric Separation of Dapoxetine Hydrochloride
 Received on 15/05/2012; Revised on 22/05/2012; Accepted on 09/06/2012 A Validated Chiral Liquid Chromatographic thod for The Enantiomeric Separation of Dapoxetine Hydrochloride T.Rohith 1 and S. Ananda
Received on 15/05/2012; Revised on 22/05/2012; Accepted on 09/06/2012 A Validated Chiral Liquid Chromatographic thod for The Enantiomeric Separation of Dapoxetine Hydrochloride T.Rohith 1 and S. Ananda
DEVELOPMENT AND VALIDATION OF RP-HPLC METHOD FOR QUANTITATIVE ANALYSIS TOLBUTAMIDE IN PURE AND PHARMACEUTICAL FORMULATIONS
 Int. J. Chem. Sci.: 11(4), 2013, 1607-1614 ISSN 0972-768X www.sadgurupublications.com DEVELOPMENT AND VALIDATION OF RP-HPLC METHOD FOR QUANTITATIVE ANALYSIS TOLBUTAMIDE IN PURE AND PHARMACEUTICAL FORMULATIONS
Int. J. Chem. Sci.: 11(4), 2013, 1607-1614 ISSN 0972-768X www.sadgurupublications.com DEVELOPMENT AND VALIDATION OF RP-HPLC METHOD FOR QUANTITATIVE ANALYSIS TOLBUTAMIDE IN PURE AND PHARMACEUTICAL FORMULATIONS
ISSN (Print)
 Scholars Academic Journal of Pharmacy (SAJP) Sch. Acad. J. Pharm., 2014; 3(3): 240-245 Scholars Academic and Scientific Publisher (An International Publisher for Academic and Scientific Resources) www.saspublisher.com
Scholars Academic Journal of Pharmacy (SAJP) Sch. Acad. J. Pharm., 2014; 3(3): 240-245 Scholars Academic and Scientific Publisher (An International Publisher for Academic and Scientific Resources) www.saspublisher.com
SIMULTANEOUS ESTIMATION OF VALSARTAN AND HYDROCHLOROTHIAZIDE IN TABLETS BY RP-HPLC METHOD
 170 Original Article SIMULTANEOUS ESTIMATION OF VALSARTAN AND HYDROCHLOROTHIAZIDE IN TABLETS BY RP-HPLC METHOD *Lakshmana Rao A, 1 Bhaskara Raju V *V.V. Institute of Pharmaceutical Sciences, Gudlavalleru,
170 Original Article SIMULTANEOUS ESTIMATION OF VALSARTAN AND HYDROCHLOROTHIAZIDE IN TABLETS BY RP-HPLC METHOD *Lakshmana Rao A, 1 Bhaskara Raju V *V.V. Institute of Pharmaceutical Sciences, Gudlavalleru,
Hyderabad, India. Department of Pharmaceutical Chemistry, Glocal University, Saharanpur, India.
 International Journal On Engineering Technology and Sciences IJETS RP-HPLC Method development and validation for the Simultaneous Estimation of Metformin and Empagliflozine in Tablet Dosage Form Shaik
International Journal On Engineering Technology and Sciences IJETS RP-HPLC Method development and validation for the Simultaneous Estimation of Metformin and Empagliflozine in Tablet Dosage Form Shaik
METHOD DEVELOPMENT AND VALIDATION BY RP-HPLC FOR ESTIMATION OF ZOLPIDEM TARTARATE
 WORLD JOURNAL OF PHARMACY AND PHARMACEUTICAL SCIENCES Ramalakshmi et al. SJIF Impact Factor 6.647 Volume 7, Issue 2, 1010-1018 Research Article ISSN 2278 4357 METHOD DEVELOPMENT AND VALIDATION BY RP-HPLC
WORLD JOURNAL OF PHARMACY AND PHARMACEUTICAL SCIENCES Ramalakshmi et al. SJIF Impact Factor 6.647 Volume 7, Issue 2, 1010-1018 Research Article ISSN 2278 4357 METHOD DEVELOPMENT AND VALIDATION BY RP-HPLC
Scholars Research Library. Der Pharmacia Lettre, 2015, 7 (5):44-49 (
 Available online at www.scholarsresearchlibrary.com Scholars Research Library Der Pharmacia Lettre, 2015, 7 (5):44-49 (http://scholarsresearchlibrary.com/archive.html) ISSN 0975-5071 USA CODEN: DPLEB4
Available online at www.scholarsresearchlibrary.com Scholars Research Library Der Pharmacia Lettre, 2015, 7 (5):44-49 (http://scholarsresearchlibrary.com/archive.html) ISSN 0975-5071 USA CODEN: DPLEB4
Pelagia Research Library
 Available online at www.pelagiaresearchlibrary.com Der Pharmacia Sinica, 2014, 5(5):91-98 ISSN: 0976-8688 CODEN (USA): PSHIBD A novel RP-HPLC method development and validation of Perindopril Erbumine in
Available online at www.pelagiaresearchlibrary.com Der Pharmacia Sinica, 2014, 5(5):91-98 ISSN: 0976-8688 CODEN (USA): PSHIBD A novel RP-HPLC method development and validation of Perindopril Erbumine in
Pelagia Research Library
 Available online at www.pelagiaresearchlibrary.com Der Pharmacia Sinica, 2015, 6(1):6-10 ISSN: 0976-8688 CODEN (USA): PSHIBD Validated RP-HPLC method for simultaneous estimation of metformin hydrochloride
Available online at www.pelagiaresearchlibrary.com Der Pharmacia Sinica, 2015, 6(1):6-10 ISSN: 0976-8688 CODEN (USA): PSHIBD Validated RP-HPLC method for simultaneous estimation of metformin hydrochloride
Simultaneous Estimation of Gemcitabine Hydrochloride and Capecitabine Hydrochloride in Combined Tablet Dosage Form by RP-HPLC Method
 ISSN: 0973-4945; CODEN ECJHAO E- Chemistry http://www.e-journals.net 2011, 8(3), 1212-1217 Simultaneous Estimation of Gemcitabine Hydrochloride and Capecitabine Hydrochloride in Combined Tablet Dosage
ISSN: 0973-4945; CODEN ECJHAO E- Chemistry http://www.e-journals.net 2011, 8(3), 1212-1217 Simultaneous Estimation of Gemcitabine Hydrochloride and Capecitabine Hydrochloride in Combined Tablet Dosage
Volume 2 (6), 2014, Page CODEN (USA)-IJPRUR, e-issn: International Journal of Pharma Research and Health Sciences
 CODEN (USA)-IJPRUR, e-issn: 2348-6465 International Journal of Pharma Research and Health Sciences Available online at www.pharmahealthsciences.net Original Article Simultaneous Determination of Salbutamol
CODEN (USA)-IJPRUR, e-issn: 2348-6465 International Journal of Pharma Research and Health Sciences Available online at www.pharmahealthsciences.net Original Article Simultaneous Determination of Salbutamol
Journal of Chemical and Pharmaceutical Research
 Available on line www.jocpr.com Journal of Chemical and Pharmaceutical Research ISSN No: 0975-7384 CODEN(USA): JCPRC5 J. Chem. Pharm. Res., 2011, 3(1):138-144 Simultaneous RP HPLC determination of Latanoprost
Available on line www.jocpr.com Journal of Chemical and Pharmaceutical Research ISSN No: 0975-7384 CODEN(USA): JCPRC5 J. Chem. Pharm. Res., 2011, 3(1):138-144 Simultaneous RP HPLC determination of Latanoprost
Available online Research Article
 Available online www.jocpr.com Journal of Chemical and Pharmaceutical Research, 2016, 8(1):171-176 Research Article ISSN : 0975-7384 CODEN(USA) : JCPRC5 Reverse phase high performance liquid chromatography
Available online www.jocpr.com Journal of Chemical and Pharmaceutical Research, 2016, 8(1):171-176 Research Article ISSN : 0975-7384 CODEN(USA) : JCPRC5 Reverse phase high performance liquid chromatography
J Pharm Sci Bioscientific Res (4): ISSN NO
 Development and Validation of Analytical Methods for Simultaneous Estimation of Pregabalin and Amitriptyline Hydrochloride in their Combined Marketed Dosage form ABSTRACT: Nikhilkumar Patel, Gurjit Kaur,
Development and Validation of Analytical Methods for Simultaneous Estimation of Pregabalin and Amitriptyline Hydrochloride in their Combined Marketed Dosage form ABSTRACT: Nikhilkumar Patel, Gurjit Kaur,
Development and Validation of a Polysorbate 20 Assay in a Therapeutic Antibody Formulation by RP-HPLC and Charged Aerosol Detector (CAD)
 LIFE SCIENCES AR ENOUGH? HOW DO YOU KNOW RAW MATERIALS ARE PURE? HOW DO YOU EVALUATE PRODUCT PACKAGING? HOW DO YOU DO YOU KNOW WHAT ANALYTICAL TECHNIQUE TO USE? HOW DO YOU SIMULTANEOUSLY TEST FOR TWO BYPRODUCTS?
LIFE SCIENCES AR ENOUGH? HOW DO YOU KNOW RAW MATERIALS ARE PURE? HOW DO YOU EVALUATE PRODUCT PACKAGING? HOW DO YOU DO YOU KNOW WHAT ANALYTICAL TECHNIQUE TO USE? HOW DO YOU SIMULTANEOUSLY TEST FOR TWO BYPRODUCTS?
Available online Research Article
 Available online www.jocpr.com Journal of Chemical and Pharmaceutical Research, 2015, 7(9):951-960 Research Article ISSN : 0975-7384 CODEN(USA) : JCPRC5 Analytical method development and validation for
Available online www.jocpr.com Journal of Chemical and Pharmaceutical Research, 2015, 7(9):951-960 Research Article ISSN : 0975-7384 CODEN(USA) : JCPRC5 Analytical method development and validation for
A Reverse Phase HPLC Method Development and Validation for the Determination of Paliperidone in Pure and Dosage Forms
 Chem Sci Trans., 2013, 2(1), 41-46 Chemical Science Transactions DOI:10.7598/cst2013.268 ISSN/E-ISSN: 2278-3458/2278-3318 RESEARCH ARTICLE A Reverse Phase HPLC Method Development and Validation for the
Chem Sci Trans., 2013, 2(1), 41-46 Chemical Science Transactions DOI:10.7598/cst2013.268 ISSN/E-ISSN: 2278-3458/2278-3318 RESEARCH ARTICLE A Reverse Phase HPLC Method Development and Validation for the
Amudha S et al., Asian Journal of Pharmthiaceutical Technology & Innovation, 04 (21); 2016; Research Article
 Asian Journal of Pharmaceutical Technology & Innovation ISSN: 2347-8810 Research Article Received on: 09-11-2016 Accepted on: 20-11-2016 Published on: 15-12-2016 Corresponding Author: * Amudha S, Dept.
Asian Journal of Pharmaceutical Technology & Innovation ISSN: 2347-8810 Research Article Received on: 09-11-2016 Accepted on: 20-11-2016 Published on: 15-12-2016 Corresponding Author: * Amudha S, Dept.
DEVELOPMENT AND VALIDATION OF RP-HPLC METHOD FOR ESTIMATION OF LACOSAMIDE IN BULK AND ITS PHARMACEUTICAL FORMULATION
 http://www.rasayanjournal.com Vol.4, No.3 (2011), 666-672 ISSN: 0974-1496 CODEN: RJCABP DEVELOPMENT AND VALIDATION OF RP-HPLC METHOD FOR IN BULK AND ITS PHARMACEUTICAL FORMULATION V.Kalyan Chakravarthy*
http://www.rasayanjournal.com Vol.4, No.3 (2011), 666-672 ISSN: 0974-1496 CODEN: RJCABP DEVELOPMENT AND VALIDATION OF RP-HPLC METHOD FOR IN BULK AND ITS PHARMACEUTICAL FORMULATION V.Kalyan Chakravarthy*
NOVEL RP-HPLC METHOD. B.Lakshmi et a. concentration range KEY INTRODUCTION. Diltiazem is used to. . It works by of contractionn of the. (dilate).
 B.Lakshmi et a ISSN-2319-2119 al, The Experiment, January. 2013 Vol..6(4), 365-371 A NOVEL RP-HPLC METHOD FOR THE DETERMINATION OF DILTIAZEM IN PHARMACEUTICAL DRUG PRODUCTS ABSTRACT High resolution RP-HPLC
B.Lakshmi et a ISSN-2319-2119 al, The Experiment, January. 2013 Vol..6(4), 365-371 A NOVEL RP-HPLC METHOD FOR THE DETERMINATION OF DILTIAZEM IN PHARMACEUTICAL DRUG PRODUCTS ABSTRACT High resolution RP-HPLC
ISSN: ; CODEN ECJHAO E-Journal of Chemistry 2011, 8(3),
 ISSN: 0973-4945; CODEN ECJHAO E- Chemistry http://www.e-journals.net 2011, 8(3), 1275-1279 Simultaneous Determination of Paracetamol, Phenylephrine Hydrochloride, Oxolamine Citrate and Chlorpheniramine
ISSN: 0973-4945; CODEN ECJHAO E- Chemistry http://www.e-journals.net 2011, 8(3), 1275-1279 Simultaneous Determination of Paracetamol, Phenylephrine Hydrochloride, Oxolamine Citrate and Chlorpheniramine
A New Stability-Indicating and Validated RP-HPLC Method for the Estimation of Liraglutide in Bulk and Pharmaceutical Dosage Forms
 OPEN ACCESS Eurasian Journal of Analytical Chemistry ISSN: 1306-3057 2017 12(2):31-44 DOI 10.12973/ejac.2017.00152a A New Stability-Indicating and Validated RP-HPLC Method for the Estimation of Liraglutide
OPEN ACCESS Eurasian Journal of Analytical Chemistry ISSN: 1306-3057 2017 12(2):31-44 DOI 10.12973/ejac.2017.00152a A New Stability-Indicating and Validated RP-HPLC Method for the Estimation of Liraglutide
DETERMINATION OF LOTEPREDNOL ETABONATE AND TOBRAMYCIN IN COMBINED DOSAGE FORM USING RP-HPLC METHOD
 280 P a g e e-issn: 2248-9126 Vol 5 Issue 4 2015 280-284. Print ISSN: 2248-9118 Indian Journal of Pharmaceutical Science & Research www.ijpsrjournal.com DETERMINATION OF LOTEPREDNOL ETABONATE AND TOBRAMYCIN
280 P a g e e-issn: 2248-9126 Vol 5 Issue 4 2015 280-284. Print ISSN: 2248-9118 Indian Journal of Pharmaceutical Science & Research www.ijpsrjournal.com DETERMINATION OF LOTEPREDNOL ETABONATE AND TOBRAMYCIN
RP-HPLC Method Development and Validation of Abacavir Sulphate in Bulk and Tablet Dosage Form
 RP-HPLC Method Development and Validation of Abacavir Sulphate in Bulk and Tablet Dosage Form S. LAVANYA* 1, SK. MANSURA BEGUM 1, K. NAGAMALLESWARA RAO 2, K. GAYATHRI DEVI 3 Department of pharmaceutical
RP-HPLC Method Development and Validation of Abacavir Sulphate in Bulk and Tablet Dosage Form S. LAVANYA* 1, SK. MANSURA BEGUM 1, K. NAGAMALLESWARA RAO 2, K. GAYATHRI DEVI 3 Department of pharmaceutical
CHAPTER INTRODUCTION OF DOSAGE FORM AND LITERATURE REVIEW
 132 CHAPTER 6 DEVELOPMENT AND VALIDATION OF A STABILITY-INDICATING RP-HPLC METHOD FOR SIMULTANEOUS DETERMINATION OF PARACETAMOL, TRAMADOL HYDROCHLORIDE AND DOMPERIDONE IN A COMBINED DOSAGE FORM 6.1 INTRODUCTION
132 CHAPTER 6 DEVELOPMENT AND VALIDATION OF A STABILITY-INDICATING RP-HPLC METHOD FOR SIMULTANEOUS DETERMINATION OF PARACETAMOL, TRAMADOL HYDROCHLORIDE AND DOMPERIDONE IN A COMBINED DOSAGE FORM 6.1 INTRODUCTION
Development and Validation of a Simultaneous HPLC Method for Quantification of Amlodipine Besylate and Metoprolol Tartrate in Tablets
 Journal of PharmaSciTech 0; ():- Research Article Development and Validation of a Simultaneous HPLC Method for Quantification of Amlodipine Besylate and Metoprolol Tartrate in Tablets * Sayyed Hussain,
Journal of PharmaSciTech 0; ():- Research Article Development and Validation of a Simultaneous HPLC Method for Quantification of Amlodipine Besylate and Metoprolol Tartrate in Tablets * Sayyed Hussain,
UPLC/MS Monitoring of Water-Soluble Vitamin Bs in Cell Culture Media in Minutes
 UPLC/MS Monitoring of Water-Soluble Vitamin Bs in Cell Culture Media in Minutes Catalin E. Doneanu, Weibin Chen, and Jeffrey R. Mazzeo Waters Corporation, Milford, MA, U.S. A P P L I C AT ION B E N E F
UPLC/MS Monitoring of Water-Soluble Vitamin Bs in Cell Culture Media in Minutes Catalin E. Doneanu, Weibin Chen, and Jeffrey R. Mazzeo Waters Corporation, Milford, MA, U.S. A P P L I C AT ION B E N E F
Analytical Method Development for USP Related Compounds in Paclitaxel Using an Agilent Poroshell 120 PFP
 Analytical Method Development for USP Related Compounds in Paclitaxel Using an Agilent Poroshell 1 PFP Application Note Clinical Research Author Rongjie Fu Agilent Technologies (Shanghai) Co. Ltd Abstract
Analytical Method Development for USP Related Compounds in Paclitaxel Using an Agilent Poroshell 1 PFP Application Note Clinical Research Author Rongjie Fu Agilent Technologies (Shanghai) Co. Ltd Abstract
Impurity Profiling of Carbamazepine by HPLC/UV
 Application Note: 52049 Impurity Profiling of Carbamazepine by HPLC/UV Terry Zhang, Guifeng Jiang, Thermo Fisher Scientific, San Jose, CA, USA Key Words Accela Hypersil GOLD Carbamazepine Drug Analysis
Application Note: 52049 Impurity Profiling of Carbamazepine by HPLC/UV Terry Zhang, Guifeng Jiang, Thermo Fisher Scientific, San Jose, CA, USA Key Words Accela Hypersil GOLD Carbamazepine Drug Analysis
Journal of Chemical and Pharmaceutical Research, 2013, 5(1): Research Article
 Available online www.jocpr.com Journal of Chemical and Pharmaceutical Research, 2013, 5(1):180-184 Research Article ISSN : 0975-7384 CODEN(USA) : JCPRC5 A simple and sensitive RP-HPLC method for estimation
Available online www.jocpr.com Journal of Chemical and Pharmaceutical Research, 2013, 5(1):180-184 Research Article ISSN : 0975-7384 CODEN(USA) : JCPRC5 A simple and sensitive RP-HPLC method for estimation
Application Note. Agilent Application Solution Analysis of ascorbic acid, citric acid and benzoic acid in orange juice. Author. Abstract.
 Agilent Application Solution Analysis of ascorbic acid, citric acid and benzoic acid in orange juice Application Note Author Food Syed Salman Lateef Agilent Technologies, Inc. Bangalore, India 8 6 4 2
Agilent Application Solution Analysis of ascorbic acid, citric acid and benzoic acid in orange juice Application Note Author Food Syed Salman Lateef Agilent Technologies, Inc. Bangalore, India 8 6 4 2
Development and validation of RP-HPLC method for simultaneous estimation of gliclazide and metformin in pure and tablet dosage form
 ISSN: 2320-2831 IJPAR Vol.3 Issue 4 Oct-Dec-2014 Journal Home page: Research article Open Access Development and validation of RP-HPLC method for simultaneous estimation of gliclazide and metformin in
ISSN: 2320-2831 IJPAR Vol.3 Issue 4 Oct-Dec-2014 Journal Home page: Research article Open Access Development and validation of RP-HPLC method for simultaneous estimation of gliclazide and metformin in
Simultaneous estimation of Metformin HCl and Sitagliptin in drug substance and drug products by RP-HPLC method
 International Journal of Chemical and Pharmaceutical Sciences 2017, Mar., Vol. 8 (1) ISSN: 0976-9390 IJCPS Simultaneous estimation of Metformin HCl and Sitagliptin in drug substance and drug products by
International Journal of Chemical and Pharmaceutical Sciences 2017, Mar., Vol. 8 (1) ISSN: 0976-9390 IJCPS Simultaneous estimation of Metformin HCl and Sitagliptin in drug substance and drug products by
Development and Validation for Simultaneous Estimation of Sitagliptin and Metformin in Pharmaceutical Dosage Form using RP-HPLC Method
 International Journal of PharmTech Research CODEN (USA): IJPRIF ISSN : 0974-4304 Vol.5, No.4, pp 1736-1744, Oct-Dec 2013 Development and Validation for Simultaneous Estimation of Sitagliptin and Metformin
International Journal of PharmTech Research CODEN (USA): IJPRIF ISSN : 0974-4304 Vol.5, No.4, pp 1736-1744, Oct-Dec 2013 Development and Validation for Simultaneous Estimation of Sitagliptin and Metformin
Ankit et al Journal of Drug Delivery & Therapeutics; 2013, 3(2), Available online at RESEARCH ARTICLE
 Ankit et al Journal of Drug Delivery & Therapeutics; 2013, 3(2), 26-30 26 Available online at http://jddtonline.info RESEARCH ARTICLE METHOD DEVELOPMENT AND ITS VALIDATION FOR QUANTITATIVE SIMULTANEOUS
Ankit et al Journal of Drug Delivery & Therapeutics; 2013, 3(2), 26-30 26 Available online at http://jddtonline.info RESEARCH ARTICLE METHOD DEVELOPMENT AND ITS VALIDATION FOR QUANTITATIVE SIMULTANEOUS
SIMULTANEOUS DETERMINATION OF ATORVASTATIN AND EZETIMIBE BY RP-HPLC IN PURE AND PHARMACEUTICAL DOSAGE FORM
 Int. J. Chem. Sci.: 6(3), 2008, 1576-1582 SIMULTANEOUS DETERMINATION OF ATORVASTATIN AND EZETIMIBE BY RP-HPLC IN PURE AND PHARMACEUTICAL DOSAGE FORM B. NEELIMA, P. RAVI KUMAR, M. MURALI KRISHNA, V. HIMA
Int. J. Chem. Sci.: 6(3), 2008, 1576-1582 SIMULTANEOUS DETERMINATION OF ATORVASTATIN AND EZETIMIBE BY RP-HPLC IN PURE AND PHARMACEUTICAL DOSAGE FORM B. NEELIMA, P. RAVI KUMAR, M. MURALI KRISHNA, V. HIMA
DEVELOPMENT AND VALIDATION OF RP-HPLC METHOD ESTIMATION OF TOLVAPTAN IN BULK PHARMACEUTICAL FORMULATION
 http://www.rasayanjournal.com Vol.4, No.1 (2011), 165-171 ISSN: 0974-1496 CODEN: RJCABP DEVELOPMENT AND VALIDATION OF RP-HPLC METHOD FOR AND ITS PHARMACEUTICAL FORMULATION V. Kalyana Chakravarthy * and
http://www.rasayanjournal.com Vol.4, No.1 (2011), 165-171 ISSN: 0974-1496 CODEN: RJCABP DEVELOPMENT AND VALIDATION OF RP-HPLC METHOD FOR AND ITS PHARMACEUTICAL FORMULATION V. Kalyana Chakravarthy * and
Scholars Research Library. Der Pharmacia Lettre, 2016, 8 (6): (http://scholarsresearchlibrary.com/archive.html)
 Available online at www.scholarsresearchlibrary.com Scholars Research Library Der Pharmacia Lettre, 2016, 8 (6):217-223 (http://scholarsresearchlibrary.com/archive.html) ISSN 0975-5071 USA CODEN: DPLEB4
Available online at www.scholarsresearchlibrary.com Scholars Research Library Der Pharmacia Lettre, 2016, 8 (6):217-223 (http://scholarsresearchlibrary.com/archive.html) ISSN 0975-5071 USA CODEN: DPLEB4
Determination of Tetracyclines in Chicken by Solid-Phase Extraction and High-Performance Liquid Chromatography
 Determination of Tetracyclines in Chicken by Solid-Phase Extraction and High-Performance Liquid Chromatography Application ote Food Safety Authors Chen-Hao Zhai and Yun Zou Agilent Technologies Co. Ltd.
Determination of Tetracyclines in Chicken by Solid-Phase Extraction and High-Performance Liquid Chromatography Application ote Food Safety Authors Chen-Hao Zhai and Yun Zou Agilent Technologies Co. Ltd.
Journal of Chemical and Pharmaceutical Research
 Available on line www.jocpr.com Journal of Chemical and Pharmaceutical Research ISSN No: 0975-7384 CODEN(USA): JCPRC5 J. Chem. Pharm. Res., 2011, 3(2):770-775 Validation of Rapid Liquid Chromatographic
Available on line www.jocpr.com Journal of Chemical and Pharmaceutical Research ISSN No: 0975-7384 CODEN(USA): JCPRC5 J. Chem. Pharm. Res., 2011, 3(2):770-775 Validation of Rapid Liquid Chromatographic
World Journal of Pharmaceutical Research
 World Journal of Pharmaceutical ReseaRch Volume 3, Issue 3, 4527-4535. Research Article ISSN 2277 715 DEVELOPMENT AND VALIDATION OF STABILITY INDICATING HPLC METHOD FOR ESTIMATION OF RAMOSETRON Zarana
World Journal of Pharmaceutical ReseaRch Volume 3, Issue 3, 4527-4535. Research Article ISSN 2277 715 DEVELOPMENT AND VALIDATION OF STABILITY INDICATING HPLC METHOD FOR ESTIMATION OF RAMOSETRON Zarana
Stability indicating RP-HPLC method for simultaneous determination of Beclomethasone Dipropionate and Clotrimazole from dosage form
 International Journal of ChemTech Research CODEN (USA): IJCRGG, ISSN: 0974-4290, ISSN(Online):2455-9555 Vol.10 No.9, pp 437-442, 2017 Stability indicating RP-HPLC method for simultaneous determination
International Journal of ChemTech Research CODEN (USA): IJCRGG, ISSN: 0974-4290, ISSN(Online):2455-9555 Vol.10 No.9, pp 437-442, 2017 Stability indicating RP-HPLC method for simultaneous determination
36 J App Pharm Vol. 6; Issue 1: 36-42; January, 2014 Rao et al., 2014
 36 J App Pharm Vol. 6; Issue 1: 36-42; January, 2014 Rao et al., 2014 Original Research Article SIMPLE AND RAPID LIQUID CHROMATOGRAPHIC METHOD FOR REAL-TIME QUANTIFICATION OF NAPROXEN / ESOMEPRAZOLE MAGNESIUM
36 J App Pharm Vol. 6; Issue 1: 36-42; January, 2014 Rao et al., 2014 Original Research Article SIMPLE AND RAPID LIQUID CHROMATOGRAPHIC METHOD FOR REAL-TIME QUANTIFICATION OF NAPROXEN / ESOMEPRAZOLE MAGNESIUM
New RP-HPLC Method for the Determination of Fludarabine in Pharmaceutical Dosage Forms
 Online : ISSN 2349-669X Print : ISSN 0973-9874 New RP-HPLC Method for the Determination of Fludarabine in Pharmaceutical Dosage Forms LAKSHMI B 1, *, RAMA KRISHNA K 2 AND JAYAVEERA K N 3 1. Department
Online : ISSN 2349-669X Print : ISSN 0973-9874 New RP-HPLC Method for the Determination of Fludarabine in Pharmaceutical Dosage Forms LAKSHMI B 1, *, RAMA KRISHNA K 2 AND JAYAVEERA K N 3 1. Department
DEVELOPMENT AND VALIDATION OF RP-HPLC METHOD FOR QUANTITATIVE ANALYSIS OF TRAMADOL IN PURE AND PHARMACEUTICAL FORMULATIONS
 Int. J. Chem. Sci.: 10(4), 2012, 2039-2047 ISSN 0972-768X www.sadgurupublications.com DEVELOPMENT AND VALIDATION OF RP-HPLC METHOD FOR QUANTITATIVE ANALYSIS OF TRAMADOL IN PURE AND PHARMACEUTICAL FORMULATIONS
Int. J. Chem. Sci.: 10(4), 2012, 2039-2047 ISSN 0972-768X www.sadgurupublications.com DEVELOPMENT AND VALIDATION OF RP-HPLC METHOD FOR QUANTITATIVE ANALYSIS OF TRAMADOL IN PURE AND PHARMACEUTICAL FORMULATIONS
SIMPLE AND VALIDATED RP-HPLC METHOD FOR THE ESTIMATION OF CARBOPLATIN IN BULK AND FORMULATION DOSAGE FORM. Subhashini.Edla* B.
 SIMPLE AND VALIDATED RP-HPLC METHOD FOR THE ESTIMATION OF CARBOPLATIN IN BULK AND FORMULATION DOSAGE FORM Subhashini.Edla* B.Syama sundhar Abstract Dept of Chemistry, Acharya Nagarjuna University, Nagarjuna
SIMPLE AND VALIDATED RP-HPLC METHOD FOR THE ESTIMATION OF CARBOPLATIN IN BULK AND FORMULATION DOSAGE FORM Subhashini.Edla* B.Syama sundhar Abstract Dept of Chemistry, Acharya Nagarjuna University, Nagarjuna
Pankti M. Shah et al, Asian Journal of Pharmaceutical Technology & Innovation, 04 (17); 2016; 07-16
 Asian Journal of Pharmaceutical Technology & Innovation ISSN: 2347-8810 Research Article Received on: 30-03-2016 Accepted on: 01-04-2016 Published on: 15-04-2016 Corresponding Author: *Pankti M. Shah,
Asian Journal of Pharmaceutical Technology & Innovation ISSN: 2347-8810 Research Article Received on: 30-03-2016 Accepted on: 01-04-2016 Published on: 15-04-2016 Corresponding Author: *Pankti M. Shah,
Effective use of Pharmacopeia guidelines to reduce cost of chromatographic analysis for Fluticasone propionate
 Effective use of Pharmacopeia guidelines to reduce cost of chromatographic analysis for Fluticasone propionate Application Note Pharmaceutical QA/QC Author Siji Joseph Agilent Technologies, Inc. Bangalore,
Effective use of Pharmacopeia guidelines to reduce cost of chromatographic analysis for Fluticasone propionate Application Note Pharmaceutical QA/QC Author Siji Joseph Agilent Technologies, Inc. Bangalore,
Development and Validation of RP-HPLC Method for the Estimation of Gemigliptin
 Human Journals Research Article September 2018 Vol.:13, Issue:2 All rights are reserved by Hajera Khan et al. Development and Validation of RP-HPLC Method for the Estimation of Gemigliptin Keywords: Gemigliptin,
Human Journals Research Article September 2018 Vol.:13, Issue:2 All rights are reserved by Hajera Khan et al. Development and Validation of RP-HPLC Method for the Estimation of Gemigliptin Keywords: Gemigliptin,
Sanjog Ramdharane 1, Dr. Vinay Gaitonde 2
 JPSBR: Volume 5, Issue 2: 2015 (151-155) ISS. 2271-3681 A ew Gradient RP- HPLC Method for Quantitative Analysis of : (3-luoro-4- Morpholin-4-yl-Phenyl)-Carbamic Acid Methyl Ester and its Related Substances
JPSBR: Volume 5, Issue 2: 2015 (151-155) ISS. 2271-3681 A ew Gradient RP- HPLC Method for Quantitative Analysis of : (3-luoro-4- Morpholin-4-yl-Phenyl)-Carbamic Acid Methyl Ester and its Related Substances
International Journal of Medicine and Pharmaceutical Research. International Journal of Medicine and Pharmaceutical Research
 International Journal of Medicine and Pharmaceutical Research Journal Home Page: www.pharmaresearchlibrary.com/ijmpr Research Article Open Access Development and Validation of RP-HPLC Method for the Estimation
International Journal of Medicine and Pharmaceutical Research Journal Home Page: www.pharmaresearchlibrary.com/ijmpr Research Article Open Access Development and Validation of RP-HPLC Method for the Estimation
Development and Validation of a RPLC Method for the Determination of 2-Phenoxyethanol in Senselle Lubricant Formulation
 Research Paper Development and Validation of a RPLC Method for the Determination of 2-Phenoxyethanol in Senselle Lubricant Formulation G. A. SHABIR*, T. K. BRADSHAW, G. Q. SHAR 1 AND S. A. ARAIN 2 Oxford
Research Paper Development and Validation of a RPLC Method for the Determination of 2-Phenoxyethanol in Senselle Lubricant Formulation G. A. SHABIR*, T. K. BRADSHAW, G. Q. SHAR 1 AND S. A. ARAIN 2 Oxford
Determination of enantiomeric purity of Timolol Maleate by Supercritical Fluid Chromatography
 Determination of enantiomeric purity of Timolol Maleate by Supercritical Fluid Chromatography Dr. Damian Connolly, (PMBRC, WIT) & Mr. Adrian Marley (Allergan Pharmaceuticals, Westport) PMBRC 3 rd Analytical
Determination of enantiomeric purity of Timolol Maleate by Supercritical Fluid Chromatography Dr. Damian Connolly, (PMBRC, WIT) & Mr. Adrian Marley (Allergan Pharmaceuticals, Westport) PMBRC 3 rd Analytical
Simultaneous Determination of Halobetasol and Salicylic Acid Related Substances by Reversed Phase High Performance Liquid Chromatographic Method
 Asian Journal of Chemistry Vol. 22, No. 5 (2010), 3369-3375 Simultaneous Determination of Halobetasol and Salicylic Acid Related Substances by Reversed Phase High Performance Liquid Chromatographic Method
Asian Journal of Chemistry Vol. 22, No. 5 (2010), 3369-3375 Simultaneous Determination of Halobetasol and Salicylic Acid Related Substances by Reversed Phase High Performance Liquid Chromatographic Method
Maximizing chromatographic peak capacity with the Agilent 1290 Infinity LC system
 Maximizing chromatographic peak capacity with the Agilent 1290 Infinity LC system A practical guide on how to use parameters to increase peak capacity Application Note Pharmaceutical and Chemical Author
Maximizing chromatographic peak capacity with the Agilent 1290 Infinity LC system A practical guide on how to use parameters to increase peak capacity Application Note Pharmaceutical and Chemical Author
RP- HPLC and Visible Spectrophotometric methods for the Estimation of Meropenem in Pure and Pharmaceutical Formulations
 International Journal of ChemTech Research CODEN( USA): IJCRGG ISSN : 0974-4290 Vol. 3, No.2, pp 605-609, April-June 2011 RP- HPLC and Visible Spectrophotometric methods for the Estimation of Meropenem
International Journal of ChemTech Research CODEN( USA): IJCRGG ISSN : 0974-4290 Vol. 3, No.2, pp 605-609, April-June 2011 RP- HPLC and Visible Spectrophotometric methods for the Estimation of Meropenem
Research Article. Development and validation of RP-HPLC method for the determination of Zaltoprofen in bulk and pharmaceutical tablet dosage form
 Available online www.jocpr.com Journal of Chemical and Pharmaceutical Research, 2015, 7(1):5-9 Research Article ISSN : 0975-7384 CODEN(USA) : JCPRC5 Development and validation of RP-HPLC method for the
Available online www.jocpr.com Journal of Chemical and Pharmaceutical Research, 2015, 7(1):5-9 Research Article ISSN : 0975-7384 CODEN(USA) : JCPRC5 Development and validation of RP-HPLC method for the
Analysis of glipizide binding to normal and glycated human serum Albumin by high-performance affinity chromatography
 Analytical and Bioanalytical Chemistry Electronic Supplementary Material Analysis of glipizide binding to normal and glycated human serum Albumin by high-performance affinity chromatography Ryan Matsuda,
Analytical and Bioanalytical Chemistry Electronic Supplementary Material Analysis of glipizide binding to normal and glycated human serum Albumin by high-performance affinity chromatography Ryan Matsuda,
RP-HPLC METHOD DEVELOPMENT AND VALIDATION FOR THE ESTIMATION OF BACLOFEN IN BULK AND PHARMACEUTICAL DOSAGE FORMS
 Int. J. Chem. Sci.: 11(1), 2013, 390-398 ISSN 0972-768X www.sadgurupublications.com RP-HPLC METHOD DEVELOPMENT AND VALIDATION FOR THE ESTIMATION OF BACLOFEN IN BULK AND PHARMACEUTICAL DOSAGE FORMS SAROJ
Int. J. Chem. Sci.: 11(1), 2013, 390-398 ISSN 0972-768X www.sadgurupublications.com RP-HPLC METHOD DEVELOPMENT AND VALIDATION FOR THE ESTIMATION OF BACLOFEN IN BULK AND PHARMACEUTICAL DOSAGE FORMS SAROJ
Separation of Macrocyclic Lactones (Avermectins) on FLARE C18 MM & FLARE C18+ Columns
 Separation of Macrocyclic Lactones (Avermectins) on FLARE C8 MM & FLARE C8+ Columns Introduction Diamond Analytics Technical Note: T05- Avermectins are a series of 6-membered macrocyclic lactone derivatives
Separation of Macrocyclic Lactones (Avermectins) on FLARE C8 MM & FLARE C8+ Columns Introduction Diamond Analytics Technical Note: T05- Avermectins are a series of 6-membered macrocyclic lactone derivatives
Two chiral centres (diastereoisomers)
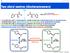 1 Two chiral centres (diastereoisomers) Mirror N S N Two enantiomers differ by absolute configuration N N 2 N 2 N A molecule with 1 stereogenic centre exists as 2 stereoisomers or enantiomers Enantiomers
1 Two chiral centres (diastereoisomers) Mirror N S N Two enantiomers differ by absolute configuration N N 2 N 2 N A molecule with 1 stereogenic centre exists as 2 stereoisomers or enantiomers Enantiomers
ASSAY AND IMPURITY METHOD FOR DURACOR TABLETS BY HPLC
 ASSAY AND IMPURITY METHOD FOR DURACOR TABLETS BY HPLC METHOD APPROVALS Norvin Pharma Inc. Author Analytical Laboratory Approver Analytical Laboratory Group Leader Approver Manager Quality Control Chemistry
ASSAY AND IMPURITY METHOD FOR DURACOR TABLETS BY HPLC METHOD APPROVALS Norvin Pharma Inc. Author Analytical Laboratory Approver Analytical Laboratory Group Leader Approver Manager Quality Control Chemistry
Analysis of L- and D-Amino Acids Using UPLC Yuta Mutaguchi 1 and Toshihisa Ohshima 2*
 Analysis of L- and D-Amino Acids Using UPLC Yuta Mutaguchi 1 and Toshihisa Ohshima 2* 1 Department of Biotechnology, Akita Prefectural University, Akita City, Japan; 2 Department of Biomedical Engineering,
Analysis of L- and D-Amino Acids Using UPLC Yuta Mutaguchi 1 and Toshihisa Ohshima 2* 1 Department of Biotechnology, Akita Prefectural University, Akita City, Japan; 2 Department of Biomedical Engineering,
Amino acids. Ing. Petrová Jaroslava. Workshop on Official Controls of Feed AGR 46230, , Ankara. Turkey ÚKZÚZ - NRL RO Praha 1
 Amino acids Ing. Petrová Jaroslava Workshop on Official Controls of Feed AGR 46230, 6. 7. 12. 2011, Ankara. Turkey 6.12.2011 ÚKZÚZ - NRL RO Praha 1 Content of this presentation 1. Function of amino acids
Amino acids Ing. Petrová Jaroslava Workshop on Official Controls of Feed AGR 46230, 6. 7. 12. 2011, Ankara. Turkey 6.12.2011 ÚKZÚZ - NRL RO Praha 1 Content of this presentation 1. Function of amino acids
HPTLC Determination of Atomoxetine Hydrochloride from its Bulk Drug and Pharmaceutical Preparations
 Asian Journal of Chemistry Vol. 20, No. 7 (2008), 5409-5413 HPTLC Determination of Atomoxetine Hydrochloride from its Bulk Drug and Pharmaceutical Preparations S.S. KAMAT, VINAYAK T. VELE, VISHAL C. CHOUDHARI
Asian Journal of Chemistry Vol. 20, No. 7 (2008), 5409-5413 HPTLC Determination of Atomoxetine Hydrochloride from its Bulk Drug and Pharmaceutical Preparations S.S. KAMAT, VINAYAK T. VELE, VISHAL C. CHOUDHARI
Isocratic Reversed Phase Liquid Chromatographic Method Validation for the Determination of Cilostazol in Pure and Formulations
 Human Journals Research Article October 2015 Vol.:4, Issue:3 All rights are reserved by Rambabu K et al. Isocratic Reversed Phase Liquid Chromatographic Method Validation for the Determination of Cilostazol
Human Journals Research Article October 2015 Vol.:4, Issue:3 All rights are reserved by Rambabu K et al. Isocratic Reversed Phase Liquid Chromatographic Method Validation for the Determination of Cilostazol
Journal of Chemical and Pharmaceutical Research, 2017, 9(9): Research Article
 Available online www.jocpr.com Journal of Chemical and Pharmaceutical Research, 2017, 9(9):70-80 Research Article ISSN : 0975-7384 CODEN(USA) : JCPRC5 Development and Validation of Stability Indicating
Available online www.jocpr.com Journal of Chemical and Pharmaceutical Research, 2017, 9(9):70-80 Research Article ISSN : 0975-7384 CODEN(USA) : JCPRC5 Development and Validation of Stability Indicating
REVERSE PHASE HPLC METHOD FOR THE ANALYSIS OF ALFUZOSIN HYDROCHLORIDE IN PHARMACEUTICAL DOSAGE FORMS
 Int. J. Chem. Sci.: 6(1), 2008, 399-404 REVERSE PHASE HPLC METHOD FOR THE ANALYSIS OF ALFUZOSIN HYDROCHLORIDE IN PHARMACEUTICAL DOSAGE FORMS S. APPALA RAJU, ARVIND B. KARADI and SHOBHA MANJUNATH HKES s
Int. J. Chem. Sci.: 6(1), 2008, 399-404 REVERSE PHASE HPLC METHOD FOR THE ANALYSIS OF ALFUZOSIN HYDROCHLORIDE IN PHARMACEUTICAL DOSAGE FORMS S. APPALA RAJU, ARVIND B. KARADI and SHOBHA MANJUNATH HKES s
Extended Application Note
 Extended Application Note Comparison of 4 μm vs. 2.o Columns in Analysis of Drospirenone/Ethinyl Estradiol Tablet APP A-339 www.mtc-usa.com 1-732-578-1777 INTRODUCTION The formulation of drospirenone and
Extended Application Note Comparison of 4 μm vs. 2.o Columns in Analysis of Drospirenone/Ethinyl Estradiol Tablet APP A-339 www.mtc-usa.com 1-732-578-1777 INTRODUCTION The formulation of drospirenone and
Determination of Glabridin in Licorice Root (Glycyrrhiza glabral.) Using High Performance Liquid Chromatography
 Determination of Glabridin in Licorice Root (Glycyrrhiza glabral.) Using High Performance Liquid Chromatography Faridah a, Siti Umrah Noor a, Rahmawati T a. Abstract Public opinionwho define that beautiful
Determination of Glabridin in Licorice Root (Glycyrrhiza glabral.) Using High Performance Liquid Chromatography Faridah a, Siti Umrah Noor a, Rahmawati T a. Abstract Public opinionwho define that beautiful
Simultaneous Quantitation of Organic Acids and Monosaccharides by High-Performance Liquid Chromatography
 International Conference on Chemical, Material and Food Engineering (CMFE-2015) Simultaneous Quantitation of Organic Acids and Monosaccharides by High-Performance Liquid Chromatography Lu Xin Institute
International Conference on Chemical, Material and Food Engineering (CMFE-2015) Simultaneous Quantitation of Organic Acids and Monosaccharides by High-Performance Liquid Chromatography Lu Xin Institute
Method Development and Validation for the Estimation of Saroglitazar in Bulk and Pharmaceutical Dosage Form by RP-HPLC
 Original Research Method Development and Validation for the Estimation of Saroglitazar in Bulk and Pharmaceutical Dosage Form by RP-HPLC Hanumantha Rao K 1, Lakshmana Rao A 2,*, Chandra Sekhar KB 3 1 Assistant
Original Research Method Development and Validation for the Estimation of Saroglitazar in Bulk and Pharmaceutical Dosage Form by RP-HPLC Hanumantha Rao K 1, Lakshmana Rao A 2,*, Chandra Sekhar KB 3 1 Assistant
DEVELOPMENT AND VALIDATION OF NEW HPLC METHOD FOR THE ESTIMATION OF PALIPERIDONE IN PHARMACEUTICAL DOSAGE FORMS
 ISSN: 0974-1496 e-issn: 0976-0083 CODEN: RJCABP http://www.rasayanjournal.com http://www.rasayanjournal.co.in DEVELOPMENT AND VALIDATION OF NEW HPLC METHOD FOR THE ESTIMATION OF PALIPERIDONE IN PHARMACEUTICAL
ISSN: 0974-1496 e-issn: 0976-0083 CODEN: RJCABP http://www.rasayanjournal.com http://www.rasayanjournal.co.in DEVELOPMENT AND VALIDATION OF NEW HPLC METHOD FOR THE ESTIMATION OF PALIPERIDONE IN PHARMACEUTICAL
Development and validation of related substances method for Varenicline and its impurities
 Available online at www.scholarsresearchlibrary.com Scholars Research Library Der Pharmacia Lettre, 2016, 8 (1):304-309 (http://scholarsresearchlibrary.com/archive.html) ISSN 0975-5071 USA CODEN: DPLEB4
Available online at www.scholarsresearchlibrary.com Scholars Research Library Der Pharmacia Lettre, 2016, 8 (1):304-309 (http://scholarsresearchlibrary.com/archive.html) ISSN 0975-5071 USA CODEN: DPLEB4
Scholars Research Library
 Available online at www.scholarsresearchlibrary.com Scholars Research Library Der Pharmacia Lettre, 2010, 2(2): 294-299 (http://scholarsresearchlibrary.com/archive.html) ISSN 0975-5071 USA CODEN: DPLEB4
Available online at www.scholarsresearchlibrary.com Scholars Research Library Der Pharmacia Lettre, 2010, 2(2): 294-299 (http://scholarsresearchlibrary.com/archive.html) ISSN 0975-5071 USA CODEN: DPLEB4
A HIGH PERFORMANCE LIQUID CHROMATOGRAPHIC ASSAY FOR LERCANIDIPINE HYDROCHLORIDE
 Int. J. Chem. Sci.: 6(1), 2008, 441-446 A HIGH PERFORMANCE LIQUID CHROMATOGRAPHIC ASSAY FOR LERCANIDIPINE HYDROCHLORIDE S. APPALA RAJU, ARVIND B. KARADI and SHOBHA MANJUNATH HKES s College of Pharmacy,
Int. J. Chem. Sci.: 6(1), 2008, 441-446 A HIGH PERFORMANCE LIQUID CHROMATOGRAPHIC ASSAY FOR LERCANIDIPINE HYDROCHLORIDE S. APPALA RAJU, ARVIND B. KARADI and SHOBHA MANJUNATH HKES s College of Pharmacy,
Chromatography on Immobilized Artificial Membrane
 Chromatography on Immobilized Artificial Membrane Principles of measuring compounds affinity to phospholipids using immobilized artificial membrane chromatography Chromatography on Immobilized Artificial
Chromatography on Immobilized Artificial Membrane Principles of measuring compounds affinity to phospholipids using immobilized artificial membrane chromatography Chromatography on Immobilized Artificial
ANALYTICAL TECHNIQUES FOR THE QUALITY OF ANTI- AIDS DRUGS
 Research Article N. Bala Krishna, IJPRBS, 2013; Volume 2(1): 63-77 ISSN: 2277-8713 IJPRBS N.BALAA KRISHNA, Dr. M.V.V NAGESWARA REDDY IJPRBS-QR CODE PAPER-QR CODE DEVELOPMENT OF NEW ANALYTICAL TECHNIQUES
Research Article N. Bala Krishna, IJPRBS, 2013; Volume 2(1): 63-77 ISSN: 2277-8713 IJPRBS N.BALAA KRISHNA, Dr. M.V.V NAGESWARA REDDY IJPRBS-QR CODE PAPER-QR CODE DEVELOPMENT OF NEW ANALYTICAL TECHNIQUES
Impact of Reversed-Phase Chiral Chromatography on the LC-MS Analysis of Drugs in Biological Fluids
 Impact of Reversed-Phase Chiral Chromatography on the LC-MS Analysis of Drugs in Biological Fluids David S. Bell, Carmen T. Santasania, Jennifer Claus, Wayne K. Way, and Craig R. Aurand Supelco, Div. of
Impact of Reversed-Phase Chiral Chromatography on the LC-MS Analysis of Drugs in Biological Fluids David S. Bell, Carmen T. Santasania, Jennifer Claus, Wayne K. Way, and Craig R. Aurand Supelco, Div. of
Asian Journal of Chemistry Vol. 21, No. 1 (2009),
 Asian Journal of Chemistry Vol. 21, o. 1 (2009), 176-182 imultaneous Estimation of Related Impurities of Tizanidine Hydrochloride in its Active Pharmaceutical Ingradient by Reversed-Phase Liquid Chromatography
Asian Journal of Chemistry Vol. 21, o. 1 (2009), 176-182 imultaneous Estimation of Related Impurities of Tizanidine Hydrochloride in its Active Pharmaceutical Ingradient by Reversed-Phase Liquid Chromatography
Separation of Perillyl Alcohol from the Peel of Citrus Unshiu by Supercritical CO 2 and Preparative High-Performance Liquid Chromatography
 Korean J. Chem. Eng., 18(3), 352-356 (2001) Separation of Perillyl Alcohol from the Peel of Citrus Unshiu by Supercritical CO 2 and Preparative High-Performance Liquid Chromatography Chong Ho Lee, Youn-Woo
Korean J. Chem. Eng., 18(3), 352-356 (2001) Separation of Perillyl Alcohol from the Peel of Citrus Unshiu by Supercritical CO 2 and Preparative High-Performance Liquid Chromatography Chong Ho Lee, Youn-Woo
New RP - HPLC Method for the Determination of Valproic acid in Human Plasma
 New RP - HPLC Method for the Determination of Valproic acid in Human Plasma C.Venkata Nagendra Prasad 1, Ch.Santhosh Kumari a, B.Srinivasa Reddy 1 and Prof. J. Sriramulu 2 1 Sree Dattha Institute of Pharmacy,
New RP - HPLC Method for the Determination of Valproic acid in Human Plasma C.Venkata Nagendra Prasad 1, Ch.Santhosh Kumari a, B.Srinivasa Reddy 1 and Prof. J. Sriramulu 2 1 Sree Dattha Institute of Pharmacy,
Rapid and sensitive UHPLC screening for water soluble vitamins in sports beverages
 APPLICATION NOTE 21671 Rapid and sensitive UHPLC screening for water soluble vitamins in sports beverages Authors Jon Bardsley, Thermo Fisher Scientific, Runcorn, UK Keywords Vanquish Flex, Acclaim PolarAdvantage
APPLICATION NOTE 21671 Rapid and sensitive UHPLC screening for water soluble vitamins in sports beverages Authors Jon Bardsley, Thermo Fisher Scientific, Runcorn, UK Keywords Vanquish Flex, Acclaim PolarAdvantage
A Validated RP-HPLC Method for the Determination of Recombinant Human Insulin in Bulk and Pharmaceutical Dosage Form
 http://www.e-journals.net ISSN: 0973-4945; CODEN ECJHAO E-Journal of Chemistry 2010, 7(S1), S449-S457 A Validated RP-HPLC Method for the Determination of Recombinant Human Insulin in Bulk and Pharmaceutical
http://www.e-journals.net ISSN: 0973-4945; CODEN ECJHAO E-Journal of Chemistry 2010, 7(S1), S449-S457 A Validated RP-HPLC Method for the Determination of Recombinant Human Insulin in Bulk and Pharmaceutical
A New HILIC/RP Mixed-Mode Column and Its Applications in Surfactant Analysis
 A New HILIC/RP Mixed-Mode Column and Its Applications in Surfactant Analysis X. Liu, C. Pohl, Dionex Corporation, Sunnyvale, CA, USA ABSTRACT Although reversed-phase (RP) silica columns (e.g., C18 and
A New HILIC/RP Mixed-Mode Column and Its Applications in Surfactant Analysis X. Liu, C. Pohl, Dionex Corporation, Sunnyvale, CA, USA ABSTRACT Although reversed-phase (RP) silica columns (e.g., C18 and
Tentu Nageswara Rao et al. / Int. Res J Pharm. App Sci., 2012; 2(4): 35-40
 International Research Journal of Pharmaceutical and Applied Sciences Available online at www.irjpas.com Int. Res J Pharm. App Sci., 2012; 2(4):35-40 Research Article Estimation of Fesoterodine fumarate
International Research Journal of Pharmaceutical and Applied Sciences Available online at www.irjpas.com Int. Res J Pharm. App Sci., 2012; 2(4):35-40 Research Article Estimation of Fesoterodine fumarate
Analysis of Amino Acids Derived Online Using an Agilent AdvanceBio AAA Column
 Application Note Pharmaceutical and Food Testing Analysis of Amino Acids Derived Online Using an Agilent AdvanceBio AAA Column Author Lu Yufei Agilent Technologies, Inc. Abstract A liquid chromatographic
Application Note Pharmaceutical and Food Testing Analysis of Amino Acids Derived Online Using an Agilent AdvanceBio AAA Column Author Lu Yufei Agilent Technologies, Inc. Abstract A liquid chromatographic
Achiral SFC: Development of an Orthogonal SFC Method for Mometasone Furoate Impurity Analysis
 Achiral SFC: Development of an Orthogonal SFC Method for Mometasone Furoate Impurity Analysis Zhenyu Wang Merck Research Laboratories NJCG 2013 Outline SFC chronicle The journey of a half century Re-birth
Achiral SFC: Development of an Orthogonal SFC Method for Mometasone Furoate Impurity Analysis Zhenyu Wang Merck Research Laboratories NJCG 2013 Outline SFC chronicle The journey of a half century Re-birth
Corresponding Author:
 Adv J Pharm Life sci Res, 2017 5;3:1-8 ISSN 2454 3535 (On-line) RP-HPLC Method for Estimation of Mupirocin in Bulk and Pharmaceutical Formulation S.K.Attar 1 *, M.S.Kalshetti 2, N. A. Jadhao 3, N. R. Patel
Adv J Pharm Life sci Res, 2017 5;3:1-8 ISSN 2454 3535 (On-line) RP-HPLC Method for Estimation of Mupirocin in Bulk and Pharmaceutical Formulation S.K.Attar 1 *, M.S.Kalshetti 2, N. A. Jadhao 3, N. R. Patel
Analytical Method Development and Validation for the Estimation of Guaifenesin and Dextromethorphan by RP-HPLC
 Human Journals Research Article September 2018 Vol.:13, Issue:2 All rights are reserved by Ramya Sree Borra et al. Analytical Method Development and Validation for the Estimation of Guaifenesin and Dextromethorphan
Human Journals Research Article September 2018 Vol.:13, Issue:2 All rights are reserved by Ramya Sree Borra et al. Analytical Method Development and Validation for the Estimation of Guaifenesin and Dextromethorphan
