3.1 Activation of human peripheral blood T lymphocytes induces reversible association of cofilin with the actin cytoskeleton
|
|
|
- Frederick Pope
- 5 years ago
- Views:
Transcription
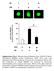
1 3. RESULTS 3.1 Activation of human peripheral blood T lymphocytes induces reversible association of cofilin with the actin cytoskeleton In unstimulated resting human peripheral blood T lymphocytes (PBTs) the small actin binding protein cofilin exists mainly in its inactive phosphorylated form. Following stimulation of accessory receptors, cofilin is dephosphorylated on phosphoserine residues (Samstag et al., 1991; Samstag et al., 1992; Samstag et al., 1994). To investigate whether the interaction of cofilin with the actin cytoskeleton is regulated through T cell activation, the amounts of cofilin in actin cytoskeletal fractions of PBTs were analysed in unstimulated PBT and at different time points after anti-cd2 stimulation. The cytoskeleton was prepared from the Triton X-100 insoluble material which contains the cytoskeletal matrix as a major component (Gregorio et al., 1992; Marano et al., 1989) and detergent insoluble membrane complexes (Schroeder et al., 1994; Sargiacomo et al., 1993). Cytoskeletal actin and its associated proteins ( actin cytoskeletal fraction ) were released from this fraction by treatment with cytochalasin B, an agent that interferes with the polymerization of actin filaments and destroys the integrity of the actin cytoskeleton (Preston et al., 1990). Western-blot analysis showed that cofilin was not detectable in actin cytoskeletal fractions prepared from resting T cells (Fig. 3A, lane 1). Following CD2 stimulation, however, cofilin transiently co-localized with the actin cytoskeleton (Fig. 3A, lanes 2-7). Cofilin was detectable in cytoskeletal fractions as early as 15 minutes after T cell stimulation. The maximum accumulation of cofilin was obtained after 1-2 hours. After 4 hours the amounts of cofilin translocated to the actin cytoskeleton gradually decreased and only minor amounts could be detected 6-8 hours after CD2 stimulation. The kinetics of the CD2 induced dephosphorylation of cofilin in NEPHGE-Western blot analysis (Fig. 3B) showed an analogous time course as its interaction with the actin 39
2 cytoskeleton (Fig. 3A). In line with previous results (Samstag et al., 1991), cytoplasmic cofilin was dephosphorylated within minutes following CD2 receptor stimulation. Maximal cofilin dephosphorylation occured 30 minutes to 2 hours after anti CD2 treatment. Subsequently, the amounts of dephosphorylated cofilin gradually decreased again (Fig. 3B lanes 2-7). A actin cofilin resting T cell 15 min 30 min α CD2 1h 2h 4h h MW (kd) SDS- PAGE B cofilin +P cofilin -P resting T cell 15 min 30 min α CD2 1h 2h 4h h 7 acidic basic NEPHGE 40
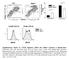
3 Figure 3 T cell activation induces dephosphorylation of cofilin and its association with the actin cytoskeleton in vivo. (A) Time course of the association of cofilin with the actin cytoskeleton following CD2 stimulation of freshly isolated human peripheral blood T cells (PBTs) for 15 min to 8 hours. The amounts of cofilin and actin in actin cytoskeletal fractions were determined by SDS- PAGE/Western blot analysis. Unstimulated PBT cells (lane 1), CD2 stimulated PBT cells (lanes 2-7). Data are representative for four independent experiments. (B) Time course of the dephosphorylation of cofilin following CD2 stimulation for 15 min to 8 hours. The phosphorylation state of cytoplasmic cofilin was examined by NEPHGE- Western blot analysis. The upper band corresponds to the phosphorylated form of cofilin (cofilin+p), the lower band represents the unphophorylated form (cofilin-p). Unstimulated PBT cells (lane 1), CD2 stimulated PBT cells (lanes 2-7). 3.2 In activated T lymphocytes the dephosphorylated form of cofilin associates with the actin cytoskeleton Both kinetics described above suggest that cofilin dephosphorylation represents a critical step in the activation induced association of cofilin with the actin cytoskeleton. To test this hypothesis, the phosphorylation state of cofilin in cytoskeletal actin fractions of CD2 stimulated PBT cells was analyzed by NEPHGE-Western blot analysis. As shown in Fig. 4 (lanes 2-4), cofilin associated with the actin cytoskeleton exclusively in its unphosphorylated basic form. These data strongly support the view, that activation induced cofilin dephosphorylation is essential for cofilin/actin cytoskeleton interactions in vivo. 41
4 cytoplasmic fraction actin cytoskeletal fractions α CD2 stimulation: cofilin +P cofilin -P 2h 30 min 1h 2h acidic basic NEPHGE Figure 4 The dephosphorylated form of cofilin associates with the actin cytoskeleton PBTs were stimulated via CD2 for the indicated times. The phosphorylation state of cofilin in the actin cytoskeletal fractions (lanes 2-4) was visualized by NEPHGE-Western blot analysis. Lane 1 shows, as a control, the phosphorylation state of cofilin in the Triton X-100 soluble cytoplasmic fraction of CD2 stimulated PBT cells (2 h). 3.3 Cofilin co-immunoprecipitates with cytoskeletal actin in activated T lymphocytes To confirm that cofilin binds to cytoskeletal actin in vivo, an actin specific antiserum was used to precipitate actin from actin cytoskeletal fractions of resting and activated T cells, respectively. Co-precipitation of cofilin was then detected employing a biotinylated cofilin specific antiserum. Significant amounts of cofilin are present in immunoprecipitates of cytoskeletal actin from CD2 activated PBT cells (Fig. 5, bar 2), but not from unstimulated PBT cells (Fig. 5, bar 1). Interestingly, in the continuously growing malignant T lymphoma 42
5 line Jurkat in which a spontaneous dephosphorylation of cofilin occurs (Samstag et al., 1996) a strong association of cofilin with the actin cytoskeleton was found in the absence of external stimuli (Fig. 5, bar 3) OD (450 nm) rt (PBT) α CD2 (PBT) Jurkat neg Figure 5 Cofilin co-precipitates with cytoskeletal actin in activated T cells. Actin was immunoprecipitated from actin cytoskeletal fractions of resting PBT cells (bar 1), CD2 stimulated PBT cells (bar 2) and Jurkat cells (bar 3). Bar 4 shows a control precipitation (no actin antiserum) from CD2 stimulated PBTs. Co-precipitation of cofilin was determined in a sandwich ELISA system employing a biotinylated cofilin specific antiserum. The same amounts of protein were applied to all samples. 43
6 3.4 The PI3-kinase inhibitor wortmannin prevents dephosphorylation of cofilin and its association with the actin cytoskeleton in peripheral blood T lymphocytes but not in Jurkat T lymphoma cells Given that PI3-kinase has been reported to influence actin cytoskeletal rearrangements following stimulation of T lymphocytes through accessory receptors such as CD2 or CD28 (Shimizu et al., 1995; Woscholski et al., 1994), it has been investigated whether PI3- kinase activity is involved in the signaling cascade from receptor stimulation to cofilin dephosphorylation and its subsequent translocation to the cytoskeleton. To this end, human peripheral blood T cells were activated by CD2 stimulation or CD3 x CD28 crosslinking, respectively, for 1 hour in the presence or absence of wortmannin, a specific and irreversible inhibitor of PI3-kinase activity (Wolscholski et al., 1994). Fig. 6 shows that, comparable to CD2 stimulation (Fig. 6A, lane 5 and Fig. 6B, lane 5), crosslinking of CD28 with the TCR/CD3 complex also results in dephosphorylation of cofilin (Fig. 6A, lane 3) and its association with the actin cytoskeleton (Fig. 6B, lane 3). Preincubation of the cells with 100 nm wortmannin for 20 minutes before cell stimulation results in an inhibition of cofilin dephosphorylation under both conditions (Fig. 6A, lanes 4 and 6). In addition, wortmannin significantly blocks the association of cofilin with the actin cytoskeleton, irrespective of the kind of receptor stimulation (CD2 or CD3 x CD28) (Fig. 6B, lanes 4 and 6). 44
7 A rt CD3/CD28 CD2 wortmannin cofilin +P cofilin -P acidic basic NEPHGE B rt CD3/CD28 CD2 wortmannin actin cofilin MW (kd) SDS- PAGE Figure 6 Dephosphorylation of cofilin and its association with the actin cytoskeleton are inhibited by wortmannin. PBTs were activated by CD3 x CD28 cross-linking (lanes 3 and 4) or CD2 stimulation (lanes 5 and 6) for 1 hour in the presence (lanes 2, 4 and 6) or absence (lanes 1, 3 and 5) of 100 nm wortmannin. rt indicates unstimulated resting PBTs (lanes 1 and 2). (A) The phosphorylation state of cytoplasmic cofilin was revealed by NEPHGE-Western blot analysis. (B) The amounts of cofilin and actin in the actin cytoskeletal fractions were determined by SDS-PAGE/Western blot analysis. 45
8 3.5 Stimulation with PMA does not influence the association of cofilin with the actin cytoskeleton Phorbolesters, such as phorbol myristate acetate (PMA) are considered to represent a stimulus which mimicks receptor mediated costimulation of T lymphocytes (Weiss et al., 1986). As shown in Fig. 7A (lanes 3 and 4) this mode of T cell activation indeed leads to a wortmannin sensitive dephosphorylation of cofilin. However, in marked contrast to antibody initiated surface receptor stimulation (Fig. 6), the association of cofilin with the actin cytoskeleton can not be induced by stimulation with PMA (Fig. 7B, lane 3) or PMA plus the calcium ionophore (A23187) (Fig. 7C, lanes 3 and 4). Figure 7 The association of cofilin with the actin cytoskeleton is not inducible through PMA. (A) PBTs were activated by CD2 receptor triggering (lanes 1 and 2) or PMA (lanes 3 and 4) in the presence (lanes 2 and 4) or absence (lanes 1 and 3) of 100 nm wortmannin. rt indicates unstimulated resting PBTs (lane 5). The phosphorylation state of cytoplasmic cofilin was analysed by NEPHGE-Western blot analysis. (B) PBTs were activated by CD2 receptor triggering (lanes 1 and 2) or PMA (lanes 3 and 4) in the presence (lanes 2 and 4) or absence (lanes 1 and 3) of 100 nm wortmannin. The amounts of cofilin and actin in the actin cytoskeletal fractions were determined by SDS-PAGE/Western blot analysis. (C) PBTs were activated with 10 nm PMA (lanes 2, 4, and 5) plus different concentrations of the ionophore A23187 (lanes 4 and 5). As a positive control, cells were stimulated with PMA plus anti-cd3 (lane 3). rt indicates unstimulated resting PBTs (lane 1). The amounts of cofilin and actin in the actin cytoskeletal fractions were determined by SDS-PAGE/Western blot analysis. 46
9 A CD2 PMA rt wortmannin cofilin +P cofilin -P acidic basic NEPHGE B CD2 PMA MW (kd) wortmannin actin 43 SDS- PAGE cofilin C PMA rt - CD3 0.5 µm Iono. 1 µm Iono. MW (kd) actin 43 SDS- PAGE cofilin
10 3.6 Wortmannin does not inhibit the constitutive association of cofilin with the actin cytoskeleton in Jurkat T lymphoma cells In Jurkat T lymphoma cells cofilin exists mainly in its dephosphorylated form (due to permanent activity of serine phosphatases) (Samstag et al., 1996). As expected, in these transformed cells cofilin associates with F-actin in the absence of external stimuli (Fig. 8, lane 1). Interestingly, here, in contrast to the situation described in untransformed T lymphocytes, wortmannin neither inhibits the spontaneous cofilin dephosphorylation (not shown) nor its constitutive association with the actin cytoskeleton (Fig. 8, lanes 2-5). These findings imply that in Jurkat cells a transforming event downstream of PI3-kinase has occured which leads to continuous cofilin activation through dephosphorylation. wortmannin (nm) actin cofilin MW (kd) SDS- PAGE Figure 8 In Jurkat T lymphoma cells cofilin constitutively associates with the actin cytoskeleton independent of PI3-Kinase activity. Unstimulated Jurkat T cells were treated with nm wortmannin as indicated lanes 2-5 for 5 hours and compared to untreated cells (lane 1). The amounts of cofilin and actin in the actin cytoskeletal fractions were determined by SDS-PAGE Western blot analysis. 48
11 3.7 The activation induced association of cofilin with the actin cytoskeleton is blocked by cofilin derived peptides With the aim of blocking the interaction of cofilin with the actin cytoskeleton in vivo, peptides corresponding to the residues 1-12 (MAS), (WAP), and (DAI) of the human cofilin sequence were synthesized. As a control, a non-actin binding control peptide (WAPQ) was designed by substitution of lysines 112 and 114 to glutamines in peptide WAP (Fig. 9A). A FLAG epitope (Hopp et al., 1988) was added to the N- terminus of each peptide to allow their detection by a monoclonal FLAG-antibody. To enable peptide entry into cells, these oligo-peptides were individually coupled to Penetratin, a 16 amino acid region of the Antennapedia homeodomain which has been shown to translocate through biological membranes in a rapid and energy-independent fashion without endosomal degradation (Perez et al., 1992; Derossi et al., 1994). Direct binding of these synthetic peptides to cytoskeletal actin was analysed in peptide binding assays in vitro. To this end, actin was immunoprecipitated from actin cytoskeletal fractions of resting T cells, in which no association of the endogenous cofilin with F-actin exists. As shown in Fig. 9B, the peptides MAS and WAP, corresponding to residues 1-12 and , significantly bound to the immunoprecipitated actin, whereas the control peptide WAPQ clearly did not. The peptide DAI, corresponding to the residues , showed only low binding to isolated cytoskeletal actin. Therefore, only the peptides MAS and WAP, as well as the control peptide WAPQ, were selected for subsequent in vivo competition experiments. 49
12 A Penetratin FLAG DAIKKKL (DAI) WAPESAPLKSKM (WAP) WAPESAPLQSQM (WAPQ) MASGVAVSDGVIK (MAS) B rel. OD DAI WAP WAPQ MAS Penetratin Figure 9 (A) Generation of Penetratin-cofilin peptides. The N-terminus of each peptide, corresponding to residues 1-12 (MAS), (WAP), and (DAI) of the human cofilin sequence was tagged by a FLAG epitope. A negative control peptide (WAPQ) was synthesized by substitution of two lysines (Lys 112 and Lys 114) to glutamines of peptide All peptides were coupled to the peptide carrier molecule Penetratin (Appligene). The Penetratin coupled peptides were purified by HPLC and mass spectrometry. (B) In vitro binding assays of Penetratin coupled peptides to cytoskeletal actin. Actin was immunoprecipitated from isolated actin cytoskeletal fractions of resting PBT cells. Peptides which bound to cytoskeletal actin were detected by a FLAG specific mab in an ELISA system. 50
13 3.8 Penetratin coupled peptides efficiently internalize into living cells To confirm that Penetratin coupled peptides are able to enter living primary T lymphocytes, PBTs were incubated with the different peptides for 2 hours at 37 C. Then, uptake of the peptides was determined by intracellular fluorescence staining employing a FLAG-specific mab and subsequent flow cytometric analysis. Fig. 10 shows that all penetratin coupled peptides (MAS, WAP, WAPQ) indeed efficiently entered T lymphocytes. Importantly, all tested peptides, including the negative control peptide (WAPQ) were taken up with comparable efficiency. No negative effects on cellular viability were exerted by any of these peptides. MAS rel. cell number WAP WAPQ Log fluorescence (FLAG) Figure 10 Internalization of penetratin coupled peptides into living PBT cells. PBTs were incubated with the Penetratin coupled peptides for 2 hours at 37 C, and the uptake of these peptides was determined by intracellular flow cytometric analysis employing a FLAG specific mab. The fluorescence intensities of cells without peptide treatment as a negative control (black area) and of the peptide treated cells (white area) are shown as an overlay. 51
14 3.9 The activation induced association of cofilin with the actin cytoskeleton in vivo is blocked by cell permeable cofilin derived peptides To investigate whether the cofilin peptides can block the association of cofilin with the actin cytoskeleton in vivo, freshly isolated human peripheral blood T cells (PBT) were preincubated with the Penetratin coupled peptides MAS and WAP (Fig. 11A, lanes 2-4), the control peptide WAPQ (Fig. 11A, lanes 5-7), or without peptides (Fig. 11A, lane 1), respectively for 2 hours. Subsequently, the T lymphocytes were stimulated via CD2 receptors in order to induce the association of cofilin with the actin cytoskeleton. The amounts of actin and cofilin in the actin cytoskeletal fractions were determined by Western blot analysis. As shown in Fig. 11A and 11B, this activation induced interaction was indeed blocked in a dose dependent manner by preincubation of cells with MAS and WAP peptides. The combination of the MAS and WAP peptides acts synergistically in inhibiting the association of cofilin with the actin cytoskeleton in vivo, as determined by densitometric quantification of cofilin band intensities (Fig. 11B). In contrast, presence of the control peptide WAPQ did not alter the amounts of cofilin in the actin cytoskeletal fractions of CD2-activated T cells (Fig. 11A lanes 5-7, and Fig. 11B). This synergistic inhibitory effect of the cofilin peptides MAS and WAP in vivo indicates that the residues 1-12 and of human cofilin function cooperatively in the interaction between cofilin and F-actin in activated T lymphocytes. 52
15 A w/o peptide MAS + WAP WAPQ (µm) actin cofilin B ratio cofilin/actin WAPQ MAS WAP WAP+MAS µm peptide Figure 11 The interaction of cofilin with the actin cytoskeleton is blocked by MAS and WAP peptides in vivo. PBT cells were preincubated with peptides MAS, WAP, WAPQ, the combination of MAS and WAP peptides or without peptide, respectively. Subsequently, PBT cells were stimulated via CD2 for 2 hours. The amounts of actin and cofilin in the actin cytoskeletal fractions were determined by Western blot analysis (A). In (B), a densitometric quantification of such Western blots is shown (expressed as the ratio of cofilin to actin band intensities). 53
16 3.10 Cofilin is involved in the process of the receptor cap formation The next series of experiments was aimed at investigating the functional consequences of the interaction of cofilin with the actin cytoskeleton during T cell activation processes. The clustering of T cell surface receptors upon receptor cross-linking ( capping ) plays an important role in the recruitment of intracellular signaling molecules to membrane receptors and the subsequent induction of cell proliferation (Fisher et al., 1998; Holsiger et al., 1998; Snapper et al., 1998). Receptor cap formation of T lymphocytes is known to depend on a functional actin cytoskeleton (De Petis, 1974). To investigate the potential functional roles of cofilin in regulating the T cell actin cytoskeleton, it was analyzed whether cofilin participates in the process of receptor cap formation in human peripheral blood T lymphocytes. Following receptor cap formation by mab mediated cross-linking of CD2 receptors in human PBTs, a co-localization of cofilin with CD2 caps is observed (>90% of the capped cells) (Fig. 12A). Employing an actin specific antiserum co-accumulation of actin at the CD2-caps was detected as well (Fig. 12B). Note that here F-actin can not be visualized by staining with phalloidin, since binding of phalloidin to F-actin is blocked by cofilin once it has bound to actin (Carlier and Pantaloni, 1997; Moon et al.,1993). Many studies have shown that PI3-kinase and the Rho family of small GTPases, including Rho, Rac and Cdc 42, regulate the reorganization of the actin cytoskeleton. Therefore, the potential co-localization of these proteins with CD2 receptor caps was also examined. As shown in Fig. 12C, the p85 subunit of PI3-kinase is detectable in CD2 caps. Moreover, one of the major PI3-kinase substrates, phosphatidylinositol 4,5-bisphosphate (PtdIns(4,5)P 2 ), also co-localizes in CD2 caps (Fig. 12D). In contrast, no co-localization of the Rho GTPases Rac1 (Fig. 12E), RhoA or Cdc42 (data not shown) was found. 54
17 A CD2 cap cofilin B CD2 cap actin C CD2 cap p85 D CD2 cap PIP 2 E CD2 cap Rac1 55
18 Figure 12 Cofilin, actin, PI3-kinase and PtdIns(4,5)P 2 (PIP 2 ), but not Rac1, co-localize with CD2 receptor caps. Receptor caps were induced in PBTs by incubation for 45 min with CD2 antibodies which were cross-linked by Texas-red labeled secondary antibodies. Cofilin (A), actin (B), the p85 subunit of PI3-kinase (C), PtdIns(4,5)P 2 (PIP 2 ) (D) and Rac1 (E) were visualized by indirect immunofluorescence staining employing Cy-2 labeled secondary antibodies. CD2 caps (red, left panels) and the counterstained proteins (green, right panels) were analyzed with a confocal laser scanning microscope. In the digital overlays of red and green fluorescence (middle panels) co-localizing proteins appear yellow. Since cofilin strongly co-localizes with CD2 receptor caps, the next experiments were aimed at investigating whether cofilin derived peptides could influence the formation of CD2 receptor caps. To this end, purified human resting T lymphocytes were preincubated with cofilin derived peptides MAS and WAP (Fig. 13B) or control peptide WAPQ (Fig. 13A), respectively. Then caps were formed by cross-linkage of CD2 receptors. In the presence of the control peptide WAPQ, CD2 receptor caps are induced and cofilin clearly co-localizes within these receptor caps (Fig. 13A). In contrast, pretreatment with the cofilin derived peptides MAS and WAP completely prevented CD2 receptor cap formation and co-accumulation of cofilin (Fig. 13B). Thus, these data provide evidence that the interaction of cofilin with the actin cytoskeleton is a critical event during the process of receptor cap formation. Figure 13 CD2 receptor cap formation is prevented by cofilin derived peptides. PBT cells were preincubated with the combination of MAS and WAP peptides (each 2.5 µm) (B) or WAPQ (5 µm) (A), respectively. Subsequently, CD2 receptor caps were induced by incubation with CD2 mabs which were cross-linked by Cy-3 labeled secondary antibodies. Cells were fixed and permeabilized, and cofilin was counterstained, employing Cy-2 labeled secondary antibodies. The CD2 caps (red, upper panels) and cofilin (green, middle panels) were visualized by indirect immunofluorescence staining and analyzed with a confocal laser scanning microscope. In the digital overlays of red and green fluorescence (lower panels) co-localizing proteins appears yellow. 56
19 A: WAPQ CD2 cap B: MAS+WAP CD2 cap cofilin cofilin 57
20 3.11 The interaction of cofilin with the actin cytoskeleton is required for T cell activation To investigate which functional T cell responses are dependent on the interaction between F-actin and cofilin, the effects of the cofilin derived peptides on cytokine production, expression of T cell surface activation markers and T cell proliferation were analyzed. In a first set of experiments, proliferation of the human peripheral blood T lymphocytes was examined. To this end, PBT cells were preincubated with the cofilin derived peptides MAS and WAP, the control peptide WAPQ, or without peptides, respectively. Subsequently, cells were stimulated via CD2 receptors (Fig. 14A) or, alternatively, by the combination of PMA plus calcium ionophore (A23187) (Fig. 14B), which bypasses proximal receptor mediated signaling events leading to direct activation of protein kinase C (PKC) and an increase of the intracellular free calcium concentration. In both modes of stimulation a strong proliferative response was observed. However, as shown in Fig. 14A, the CD2 induced PBT cell proliferation was significantly inhibited by preincubation with the peptides MAS and WAP. In contrast, upon stimulation of T lymphocytes with PMA plus ionophore, the cofilin derived pepetides MAS and WAP failed to inhibit cell proliferation (Fig. 14B). The control peptide WAPQ had no effect on T cell proliferation. These findings suggest that the cofilin/f-actin interaction is essential for early receptor mediated activation steps that are located upstream of PKC activation and Ca 2+ mobilization. 58
21 A CD2 WAPQ w/o peptide WAP+MAS cpm B cpm Anti-CD2 (µg/ml) PMA/Ionophore PMA (nμ) WAPQ w/o peptide WAP+MAS Figure 14 CD2-induced T cell proliferation is inhibited by cofilin derived peptides. Proliferative responses of resting PBT cells, preincubated with the combination of MAS and WAP peptides (2.5 µm each ), WAPQ (5 µm, ), or without peptide treatment ( ), respectively. (A) PBT cells were stimulated with various concentrations of anti-cd2. (B) Alternatively, T cells were stimulated with various concentration of PMA plus ionophore (A23187). All cultures were maintained for 48 hours adding 3 H-thymidine during the last 16 hours of culture. The experiments were repeated at least 3 times providing comparable results. 59
22 To further explore the role of cofilin in T cell activation processes, the effects of cofilin derived peptides on cytokine production was analyzed. Following CD2 stimulation of resting human T lymphocytes, the amounts of the T cell growth factor Interleukin-2 (IL-2) as well as the proinflammatory cytokine γifn in the T cell culture supernatants were determined by ELISA. As shown in Fig. 15A, preincubation with the WAP and MAS peptides strongly inhibited the CD2 inducible IL-2 production. In marked contrast, the CD2 induced production of γifn was dramatically enhanced by preincubation with the peptides WAP and MAS (Fig. 15B). The control peptide WAPQ was ineffective in both cases. These results indicate that the interaction of cofilin with F-actin is differentically involved in the production of cytokines. 60
23 A IL-2 pg/ml rt CD2 MAS+WAP WAPQ B 1200 γifn 1000 pg/ml rt CD2 MAS+WAP WAPQ Figure 15 IL-2 production is inhibited by cofilin derived peptides, whereas γifn production is enhanced. PBT cells were preincubated with the combination of MAS and WAP peptides (2.5 µm each) (A and B, bars 3) or WAPQ (5 µm) (A and B, bars 4), subsequently, T cells were activated via CD2 receptor triggering. Bar 2 in A and B shows the CD2 activated cells without peptide treatment. rt indicates unstimulated resting PBTs (A and B, bars 1). The production of IL-2 (A) and γifn (B) was determined in cell culture supernatants harvested after 48 h of T cell culture. A representative experiment of three independent experiments is shown. 61
24 Finally, the effects of cofilin derived peptides on the expression levels of different activation markers on the surface of activated T lymphocytes was investigated. To this end, human resting T lymphocytes were preincubated with the cofilin derived peptides MAS plus WAP, the control peptide WAPQ, or without peptides. Subsequently, PBT cells were activated via CD2 receptors and the expression of different activation markers on the surface of activated T cells was measured by flow cytometric analysis. Interestingly, different influences of the cofilin derived peptides on the CD2-induced expression of various activation markers were observed. Notably, the upregulation of CD69, an early T cell activation marker, was almost completely inhibited by preincubating the cells with cofilin derived peptides (WAP+MAS) (Fig. 16A). In marked contrast, a strong increase in the surface expression of MHC class II molecules in the presence of WAP and MAS peptides was observed (Fig. 16B). This might be a secondary effect due to the observed increase in γifn production, which has been described to induce upregulation of MHC expression. The CD2 induced upregulation of CD25 expression (IL-2 receptor α chain) was not significantly altered by preincubation with cofilin derived peptides (Fig. 16C). In all cases, the control peptide WAPQ did not show any effect. Taken together, the WAP and MAS peptides exert strong inhibitory effects on T cell proliferation, IL-2 production and the expression of CD69, whereas the production of γifn and the expression of MHC class II molecules are significantly enhanced. 62
25 A. CD 69 B. MHC II C. CD rt 2. CD2 3. CD2 + MAS/WAP 4. CD2 + WAPQ Figure 16 Diverse effects of the cofilin derived peptides on the expression of different activation markers. PBT cells were preincubated with the combination of MAS and WAP peptides (2.5 µm each 3), WAPQ (5 µm. 4) or without peptide (2), respectively. Subsequently, PBT cells were activated by CD2 receptor triggering (2, 3 and 4). Unstimulated resting PBTs (rt) are shown in (1). Expression of CD69 (A) was monitored after 16 hours, MHC II (HLA-DR) (B) and CD25 (C) after 48 hours of culture. 63
26 3.12 The cofilin derived peptides inhibit CD2-mediated activation induced cell death (ACID) As shown above, the interaction of cofilin with the actin cytoskeleton plays an important role for the activation of primary T lymphocytes. In previously activated T cells or hybridoma cell lines stimulation induces apoptosis, a process which has been termed activation induced cell death (AICD) (Kabelitz et al., 1993). To address the question whether cofilin also affects AICD, the influence of cofilin derived peptides on CD2- induced apoptosis of the IL-2 dependent human T cell clone D cells was investigated (Wesselborg et al., 1993). Following preincubation with cofilin derived peptides MAS and WAP, control peptide WAPQ or without peptide treatment, D cells were stimulated with three mitogenic CD2 mabs (M1, M2 and 3PT) directed against different CD2 epitopes. After 6 hours of incubation, apoptosis was determined by two different experimental methods: (1) by Annexin V and PI double staining (Fig. 17A) or, alternatively, (2) by terminal transferase (TdT)-mediated nick end labeling (TUNEL) staining (Fig. 17B). Fig. 17 demonstrates that CD2 stimulation efficiently induced apoptosis in this T cell clone. This induction of apoptosis by anti-cd2 mabs was significantly reduced by preincubation with MAS and WAP peptides (Fig 17A-3, 17B-3). As expected, no effect was observed by a treatment with the control peptide WAPQ (Fig. 17A-4, 17B-4). These data clearly demonstrate that the cofilin derived peptides MAS and WAP prevent activation induced programmed cell death in D cells. 64
27 A B 1. neg. cont PI rel. cell number 2. CD2 3. CD2 + MAS/WAP 4. CD2 + WAPQ Annexin V TUNEL Figure 17 CD-mediated apoptosis (ACID) is inhibited by cofilin derived peptides. The T cell clone D was preincubated for 2 hours with the combination of MAS and WAP peptides (2.5 µm each) (3), WAPQ (5 µm) (4) or without peptide (1 and 2), respectively. Subsequently, cells were activated for 8 hours with anti-cd2 mab. (1) shows, as a negative control the unstimulated T cell clone. (A) Uptake of PI (Y-axis) and staining with annexin V-FITC (X-axis) of D cells cultured for 6 hours. (B) DNA fragmentation was revealed Tdt-mediated nick end labeling (TUNEL assay) (incorporation of FITC labled dutp) and intracellular flow cytometric analysis. 65
28 3.13 Cofilin derived peptides do not influence CD95-induced apoptosis D cells express CD95 (Fas) receptors and are sensitive to the CD95 mediatedapoptotic signal when directly triggered via CD95 receptors. To determine whether the cofilin derived peptides MAS and WAP also affect CD95 mediated programmed cell death, apoptosis the D T cell clone was triggered with anti-cd95 mab in the presence of MAS and WAP peptides, WAPQ peptide or in the absence of peptides. Anti-CD95 induced cell death (apoptosis) was detected by Annexin V and TUNEL assays. Interestingly, as shown in Fig.18 both apoptosis assays demonstrated that in marked contrast to the CD2-induced ACID, the cofilin derived pepetides MAS and WAP did not affect apoptosis in response to CD95 triggering. This result provides evidence for the involvement of cofilin in the CD2-mediated signaling pathway of activation induced apoptosis, but not on CD95 (Fas)-mediated signaling. 66
29 A B 1. neg. cont PI rel. cell number 2. CD95 3. CD95 + MAS/WAP Annexin V TUNEL Figure 18 CD95 induced apoptosis is not inhibited by cofilin derived peptides. The T cell clone D was preincubated for 2 hours with the combination of MAS and WAP peptides (2.5 µm each) (3) or without peptide (1 and 2), respectively. Subsequently, cells were activated by CD95 receptor triggering. (1) Unstimulated T cells. (A) Uptake of PI (Yaxis) and staining with annexin V-FITC (X-axis) of D cells cultured for 5 hours. (B) TUNEL assay of fixed cells after 6 hours of stimulation with CD95 mab. DNA fragmentation was revealed by Tdt mediated nick end labeling (incorporation of FITC labeled dutp) and subsequent intracellular flow cytometric analysis. 67
30 3.14 Apoptosis induced via CD2 is inhibited by soluble anti-cd95 mab Fas mediated cell death has been suggested to be the major pathway of activation induced apoptosis of T cell hybridomas (Brunner et al., 1995; Dhein et al., 1995; Ju-S et al., 1995; Yang et al., 1995). To determine whether CD2 induced apoptosis in the D T cell clone is dependent on CD95/CD95 ligand (Fas/FasL) interaction, cells were stimulated via CD2 in the presence or absence of a blocking anti-cd95 mab (soluble anti-cd95 mab F(ab) fragments, which prevent the interaction between CD95 and its ligand without inducing apoptotic signals). Apoptotic cells were determined by the Annexin V assays. Fig. 19 shows the results of a representative experiment, demonstrating that the treatment with the blocking anti-cd95 mab inhibited CD2 induced apoptosis in a dose dependent manner. This result demonstrates that the CD95/CD95 ligand (Fas/FasL) system is involved in CD2 induced apoptosis in the D T cell clone. 68
31 1. neg. control 7-ADD 2. CD2 3. CD2 + Apo 1 (0.5 µg/ml) 4. CD2 + Apo 1 (1 µg/ml) Annexin V Figure 19 Inhibiton of activation induced apoptosis in anti-cd2 stimulated D cells by soluble anti-cd95 mab Anti-CD2 stimulated D cells were cultured in the presence of F(ab) fragments of CD95 mab in different concentrations (3, 4), or in the absence of anti-cd95 (1 and 2). The unstimulated T cell clone is shown in (1). Apoptosis was evaluated by flow cytometric analysis of Annexin V-FITC (X-axis)/7-AAD (Y-axis) stained cells after 6 hours of culture. 69
32 3.15 Cofilin derived peptides inhibit the expression of CD95 ligand As described above, CD95 signals are involved in CD2-induced apoptosis of D cells, therefore, the ability of cofilin derived peptides to affect the expression of CD95 ligand during the CD2-induced apoptotic process was examined. The CD95 ligand is a 40 kd type II membrane protein. Human CD95 ligand is released as a 26 kd protein from the T cell membrane by metalloproteinases (Kayagaki et al., 1995; Oyaizu et al., 1997). D cells were treated with CD2 mabs in the presence of cofilin derived peptides (MAS+WAP), the control peptide (WAPQ) or in the absence of peptide. The expression of CD95 ligand on the cell surface after 6 hours of culture was determined by flow cytometric analysis. Fig. 20A (2) demonstrates that CD2 stimulation induces surface expression of CD95 ligand on D cells. Detection of CD95 ligand expression following CD2 stimulation could be improved by preincubation of D cells with an inhibitor of matrix metalloproteinases, KB 8301, which blocks the cleavage of membrane bound forms of human CD95 ligand (Fig. 20B (2)). The expression of CD95 ligand was strongly inhibited by MAS and WAP peptides, both in the presence (Fig. 20B, (3)) and absence (Fig. 20A, (3)) of the matrix metalloproteinases inhibitor KB The control peptide WAPQ showed no effect on CD95 ligand expression. These results indicate that cofilin is involved in the signaling pathway downstream of CD2 receptor stimulation leading to CD95L expression but upstream of the CD95/CD95L interaction during activation induced apoptosis. Moreover, the cofilin derived peptides likely have no influence on cleavage of human membrane bound CD95 ligand. 70
33 A: - KB 8301 B: + KB neg.control M1 M2 M1 M2 2. CD2 Relative cell number M1 M2 M1 M2 3. CD2 + MAS/WAP 4. CD2 + WAPQ Log fluorescence (CD95L) Figure 20 Expression of human CD95 ligand on the surface of the T cell clone D is prevented by cofilin derived peptides. The expression of CD95 ligand on the human T cell clone D was analysed by flow cytometry in the presence (B), or absence of a metalloproteinase inhibitor, KB8301 (A). D cells were pretreated with a combination of MAS and WAP peptides (3), control peptide WAPQ (4) or without peptide (1 and 2). (1) shows, as a negative control, unstimulated cells. In (2) (3) and (4) the cells were stimulated via CD2 triggering for 6 hours to induce CD95 ligand expression. Fluorescence intensity (Y-axis) versus cell number (X-axis) is shown. 71
Supplementary Figure 1. Normal T lymphocyte populations in Dapk -/- mice. (a) Normal thymic development in Dapk -/- mice. Thymocytes from WT and Dapk
 Supplementary Figure 1. Normal T lymphocyte populations in Dapk -/- mice. (a) Normal thymic development in Dapk -/- mice. Thymocytes from WT and Dapk -/- mice were stained for expression of CD4 and CD8.
Supplementary Figure 1. Normal T lymphocyte populations in Dapk -/- mice. (a) Normal thymic development in Dapk -/- mice. Thymocytes from WT and Dapk -/- mice were stained for expression of CD4 and CD8.
3.1 Inhibition of IL-2 secretion by a CD44 receptor globulin and induction of T cell proliferation by cross-linking of CD44
 3. RESULTS 3.1 Inhibition of IL-2 secretion by a CD44 receptor globulin and induction of T cell proliferation by cross-linking of CD44 Antigen-specific activation of the influenca HA specific murine T
3. RESULTS 3.1 Inhibition of IL-2 secretion by a CD44 receptor globulin and induction of T cell proliferation by cross-linking of CD44 Antigen-specific activation of the influenca HA specific murine T
Introduction: 年 Fas signal-mediated apoptosis. PI3K/Akt
 Fas-ligand (CD95-L; Fas-L) Fas (CD95) Fas (apoptosis) 年 了 不 度 Fas Fas-L 力 不 Fas/Fas-L T IL-10Fas/Fas-L 不 年 Fas signal-mediated apoptosis 度降 不 不 力 U-118, HeLa, A549, Huh-7 MCF-7, HepG2. PI3K/Akt FasPI3K/Akt
Fas-ligand (CD95-L; Fas-L) Fas (CD95) Fas (apoptosis) 年 了 不 度 Fas Fas-L 力 不 Fas/Fas-L T IL-10Fas/Fas-L 不 年 Fas signal-mediated apoptosis 度降 不 不 力 U-118, HeLa, A549, Huh-7 MCF-7, HepG2. PI3K/Akt FasPI3K/Akt
Supplementary Figure S I: Effects of D4F on body weight and serum lipids in apoe -/- mice.
 Supplementary Figures: Supplementary Figure S I: Effects of D4F on body weight and serum lipids in apoe -/- mice. Male apoe -/- mice were fed a high-fat diet for 8 weeks, and given PBS (model group) or
Supplementary Figures: Supplementary Figure S I: Effects of D4F on body weight and serum lipids in apoe -/- mice. Male apoe -/- mice were fed a high-fat diet for 8 weeks, and given PBS (model group) or
were isolated from the freshly drawn blood of healthy donors and ACS patients using the
 Supplemental Figure 1. Quality control of CD4 + T-cell purification. CD4 + T cells were isolated from the freshly drawn blood of healthy donors and ACS patients using the RosetteSep CD4 + T Cell Enrichment
Supplemental Figure 1. Quality control of CD4 + T-cell purification. CD4 + T cells were isolated from the freshly drawn blood of healthy donors and ACS patients using the RosetteSep CD4 + T Cell Enrichment
SUPPLEMENTARY INFORMATION
 Figure S1 Treatment with both Sema6D and Plexin-A1 sirnas induces the phenotype essentially identical to that induced by treatment with Sema6D sirna alone or Plexin-A1 sirna alone. (a,b) The cardiac tube
Figure S1 Treatment with both Sema6D and Plexin-A1 sirnas induces the phenotype essentially identical to that induced by treatment with Sema6D sirna alone or Plexin-A1 sirna alone. (a,b) The cardiac tube
Intracellular MHC class II molecules promote TLR-triggered innate. immune responses by maintaining Btk activation
 Intracellular MHC class II molecules promote TLR-triggered innate immune responses by maintaining Btk activation Xingguang Liu, Zhenzhen Zhan, Dong Li, Li Xu, Feng Ma, Peng Zhang, Hangping Yao and Xuetao
Intracellular MHC class II molecules promote TLR-triggered innate immune responses by maintaining Btk activation Xingguang Liu, Zhenzhen Zhan, Dong Li, Li Xu, Feng Ma, Peng Zhang, Hangping Yao and Xuetao
Propagation of the Signal
 OpenStax-CNX module: m44452 1 Propagation of the Signal OpenStax College This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License 3.0 By the end of this section,
OpenStax-CNX module: m44452 1 Propagation of the Signal OpenStax College This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License 3.0 By the end of this section,
- 1 - Cell types Monocytes THP-1 cells Macrophages. LPS Treatment time (Hour) IL-6 level (pg/ml)
 Supplementary Table ST1: The dynamic effect of LPS on IL-6 production in monocytes and THP-1 cells after GdA treatment. Monocytes, THP-1 cells and macrophages (5x10 5 ) were incubated with 10 μg/ml of
Supplementary Table ST1: The dynamic effect of LPS on IL-6 production in monocytes and THP-1 cells after GdA treatment. Monocytes, THP-1 cells and macrophages (5x10 5 ) were incubated with 10 μg/ml of
G-Protein Signaling. Introduction to intracellular signaling. Dr. SARRAY Sameh, Ph.D
 G-Protein Signaling Introduction to intracellular signaling Dr. SARRAY Sameh, Ph.D Cell signaling Cells communicate via extracellular signaling molecules (Hormones, growth factors and neurotransmitters
G-Protein Signaling Introduction to intracellular signaling Dr. SARRAY Sameh, Ph.D Cell signaling Cells communicate via extracellular signaling molecules (Hormones, growth factors and neurotransmitters
Nature Immunology: doi: /ni.3866
 Nature Immunology: doi:10.1038/ni.3866 Supplementary Figure 1 The effect of TIPE2 on chemotaxis. a, The expression of TIPE2 in dhl-60c, dhl-60t, TIPE2-expressing and 15/16Q-expressing dhl-60t neutrophils
Nature Immunology: doi:10.1038/ni.3866 Supplementary Figure 1 The effect of TIPE2 on chemotaxis. a, The expression of TIPE2 in dhl-60c, dhl-60t, TIPE2-expressing and 15/16Q-expressing dhl-60t neutrophils
Principles of Genetics and Molecular Biology
 Cell signaling Dr. Diala Abu-Hassan, DDS, PhD School of Medicine Dr.abuhassand@gmail.com Principles of Genetics and Molecular Biology www.cs.montana.edu Modes of cell signaling Direct interaction of a
Cell signaling Dr. Diala Abu-Hassan, DDS, PhD School of Medicine Dr.abuhassand@gmail.com Principles of Genetics and Molecular Biology www.cs.montana.edu Modes of cell signaling Direct interaction of a
Supplementary Figure 1. Validation of astrocytes. Primary astrocytes were
 Supplementary Figure 1. Validation of astrocytes. Primary astrocytes were separated from the glial cultures using a mild trypsinization protocol. Anti-glial fibrillary acidic protein (GFAP) immunofluorescent
Supplementary Figure 1. Validation of astrocytes. Primary astrocytes were separated from the glial cultures using a mild trypsinization protocol. Anti-glial fibrillary acidic protein (GFAP) immunofluorescent
Figure S1. PMVs from THP-1 cells expose phosphatidylserine and carry actin. A) Flow
 SUPPLEMENTARY DATA Supplementary Figure Legends Figure S1. PMVs from THP-1 cells expose phosphatidylserine and carry actin. A) Flow cytometry analysis of PMVs labelled with annexin-v-pe (Guava technologies)
SUPPLEMENTARY DATA Supplementary Figure Legends Figure S1. PMVs from THP-1 cells expose phosphatidylserine and carry actin. A) Flow cytometry analysis of PMVs labelled with annexin-v-pe (Guava technologies)
Nature Immunology: doi: /ni.3631
 Supplementary Figure 1 SPT analyses of Zap70 at the T cell plasma membrane. (a) Total internal reflection fluorescent (TIRF) excitation at 64-68 degrees limits single molecule detection to 100-150 nm above
Supplementary Figure 1 SPT analyses of Zap70 at the T cell plasma membrane. (a) Total internal reflection fluorescent (TIRF) excitation at 64-68 degrees limits single molecule detection to 100-150 nm above
Signal Transduction Cascades
 Signal Transduction Cascades Contents of this page: Kinases & phosphatases Protein Kinase A (camp-dependent protein kinase) G-protein signal cascade Structure of G-proteins Small GTP-binding proteins,
Signal Transduction Cascades Contents of this page: Kinases & phosphatases Protein Kinase A (camp-dependent protein kinase) G-protein signal cascade Structure of G-proteins Small GTP-binding proteins,
SUPPLEMENTARY INFORMATION
 doi: 1.138/nature775 4 O.D. (595-655) 3 1 -ζ no antibody isotype ctrl Plated Soluble 1F6 397 7H11 Supplementary Figure 1 Soluble and plated anti- Abs induce -! signalling. B3Z cells stably expressing!
doi: 1.138/nature775 4 O.D. (595-655) 3 1 -ζ no antibody isotype ctrl Plated Soluble 1F6 397 7H11 Supplementary Figure 1 Soluble and plated anti- Abs induce -! signalling. B3Z cells stably expressing!
Figure S1. Western blot analysis of clathrin RNA interference in human DCs Human immature DCs were transfected with 100 nm Clathrin SMARTpool or
 Figure S1. Western blot analysis of clathrin RNA interference in human DCs Human immature DCs were transfected with 100 nm Clathrin SMARTpool or control nontargeting sirnas. At 90 hr after transfection,
Figure S1. Western blot analysis of clathrin RNA interference in human DCs Human immature DCs were transfected with 100 nm Clathrin SMARTpool or control nontargeting sirnas. At 90 hr after transfection,
Part-4. Cell cycle regulatory protein 5 (Cdk5) A novel target of ERK in Carb induced cell death
 Part-4 Cell cycle regulatory protein 5 (Cdk5) A novel target of ERK in Carb induced cell death 95 1. Introduction The process of replicating DNA and dividing cells can be described as a series of coordinated
Part-4 Cell cycle regulatory protein 5 (Cdk5) A novel target of ERK in Carb induced cell death 95 1. Introduction The process of replicating DNA and dividing cells can be described as a series of coordinated
Integrin v 3 targeted therapy for Kaposi s sarcoma with an in vitro evolved antibody 1
 Integrin v 3 targeted therapy for Kaposi s sarcoma with an in vitro evolved antibody 1 CHRISTOPH RADER, 2 MIKHAIL POPKOV, JOHN A. NEVES, AND CARLOS F. BARBAS III 2 Department of Molecular Biology and The
Integrin v 3 targeted therapy for Kaposi s sarcoma with an in vitro evolved antibody 1 CHRISTOPH RADER, 2 MIKHAIL POPKOV, JOHN A. NEVES, AND CARLOS F. BARBAS III 2 Department of Molecular Biology and The
Supplementary Figure 1. Supernatants electrophoresis from CD14+ and dendritic cells. Supernatants were resolved by SDS-PAGE and stained with
 Supplementary Figure 1. Supernatants electrophoresis from CD14+ and dendritic cells. Supernatants were resolved by SDS-PAGE and stained with Coomassie brilliant blue. One µg/ml recombinant human (rh) apo-e
Supplementary Figure 1. Supernatants electrophoresis from CD14+ and dendritic cells. Supernatants were resolved by SDS-PAGE and stained with Coomassie brilliant blue. One µg/ml recombinant human (rh) apo-e
Cell Signaling part 2
 15 Cell Signaling part 2 Functions of Cell Surface Receptors Other cell surface receptors are directly linked to intracellular enzymes. The largest family of these is the receptor protein tyrosine kinases,
15 Cell Signaling part 2 Functions of Cell Surface Receptors Other cell surface receptors are directly linked to intracellular enzymes. The largest family of these is the receptor protein tyrosine kinases,
Supplementary Figure 1.TRIM33 binds β-catenin in the nucleus. a & b, Co-IP of endogenous TRIM33 with β-catenin in HT-29 cells (a) and HEK 293T cells
 Supplementary Figure 1.TRIM33 binds β-catenin in the nucleus. a & b, Co-IP of endogenous TRIM33 with β-catenin in HT-29 cells (a) and HEK 293T cells (b). TRIM33 was immunoprecipitated, and the amount of
Supplementary Figure 1.TRIM33 binds β-catenin in the nucleus. a & b, Co-IP of endogenous TRIM33 with β-catenin in HT-29 cells (a) and HEK 293T cells (b). TRIM33 was immunoprecipitated, and the amount of
The T cell receptor for MHC-associated peptide antigens
 1 The T cell receptor for MHC-associated peptide antigens T lymphocytes have a dual specificity: they recognize polymporphic residues of self MHC molecules, and they also recognize residues of peptide
1 The T cell receptor for MHC-associated peptide antigens T lymphocytes have a dual specificity: they recognize polymporphic residues of self MHC molecules, and they also recognize residues of peptide
The role of the scaffolding protein Tks5 in EGF signaling
 The role of the scaffolding protein Tks5 in EGF signaling PhD Thesis Anna Fekete Doctoral School in Biology Head of the School: Dr. Anna Erdei Structural Biochemistry Doctoral Program Head of the Program:
The role of the scaffolding protein Tks5 in EGF signaling PhD Thesis Anna Fekete Doctoral School in Biology Head of the School: Dr. Anna Erdei Structural Biochemistry Doctoral Program Head of the Program:
Nature Immunology doi: /ni.3268
 Supplementary Figure 1 Loss of Mst1 and Mst2 increases susceptibility to bacterial sepsis. (a) H&E staining of colon and kidney sections from wild type and Mst1 -/- Mst2 fl/fl Vav-Cre mice. Scale bar,
Supplementary Figure 1 Loss of Mst1 and Mst2 increases susceptibility to bacterial sepsis. (a) H&E staining of colon and kidney sections from wild type and Mst1 -/- Mst2 fl/fl Vav-Cre mice. Scale bar,
Supplemental information
 Supplemental information PI(3)K p11δ controls the sucellular compartmentalization of TLR4 signaling and protects from endotoxic shock Ezra Aksoy, Salma Taoui, David Torres, Sandrine Delauve, Aderrahman
Supplemental information PI(3)K p11δ controls the sucellular compartmentalization of TLR4 signaling and protects from endotoxic shock Ezra Aksoy, Salma Taoui, David Torres, Sandrine Delauve, Aderrahman
Supplementary Materials
 Supplementary Materials Figure S1. MTT Cell viability assay. To measure the cytotoxic potential of the oxidative treatment, the MTT [3-(4,5-dimethylthiazol- 2-yl)-2,5-diphenyl tetrazolium bromide] assay
Supplementary Materials Figure S1. MTT Cell viability assay. To measure the cytotoxic potential of the oxidative treatment, the MTT [3-(4,5-dimethylthiazol- 2-yl)-2,5-diphenyl tetrazolium bromide] assay
hexahistidine tagged GRP78 devoid of the KDEL motif (GRP78-His) on SDS-PAGE. This
 SUPPLEMENTAL FIGURE LEGEND Fig. S1. Generation and characterization of. (A) Coomassie staining of soluble hexahistidine tagged GRP78 devoid of the KDEL motif (GRP78-His) on SDS-PAGE. This protein was expressed
SUPPLEMENTAL FIGURE LEGEND Fig. S1. Generation and characterization of. (A) Coomassie staining of soluble hexahistidine tagged GRP78 devoid of the KDEL motif (GRP78-His) on SDS-PAGE. This protein was expressed
The Annexin V Apoptosis Assay
 The Annexin V Apoptosis Assay Development of the Annexin V Apoptosis Assay: 1990 Andree at al. found that a protein, Vascular Anticoagulant α, bound to phospholipid bilayers in a calcium dependent manner.
The Annexin V Apoptosis Assay Development of the Annexin V Apoptosis Assay: 1990 Andree at al. found that a protein, Vascular Anticoagulant α, bound to phospholipid bilayers in a calcium dependent manner.
KEY CONCEPT QUESTIONS IN SIGNAL TRANSDUCTION
 Signal Transduction - Part 2 Key Concepts - Receptor tyrosine kinases control cell metabolism and proliferation Growth factor signaling through Ras Mutated cell signaling genes in cancer cells are called
Signal Transduction - Part 2 Key Concepts - Receptor tyrosine kinases control cell metabolism and proliferation Growth factor signaling through Ras Mutated cell signaling genes in cancer cells are called
THE ROLE OF ALTERED CALCIUM AND mtor SIGNALING IN THE PATHOGENESIS OF CYSTINOSIS
 Research Foundation, 18 month progress report THE ROLE OF ALTERED CALCIUM AND mtor SIGNALING IN THE PATHOGENESIS OF CYSTINOSIS Ekaterina Ivanova, doctoral student Elena Levtchenko, MD, PhD, PI Antonella
Research Foundation, 18 month progress report THE ROLE OF ALTERED CALCIUM AND mtor SIGNALING IN THE PATHOGENESIS OF CYSTINOSIS Ekaterina Ivanova, doctoral student Elena Levtchenko, MD, PhD, PI Antonella
The modulatory effect of cholesterol rich lipid rafts on the effector functions, differentiation and communication of lymphocytes.
 The modulatory effect of cholesterol rich lipid rafts on the effector functions, differentiation and communication of lymphocytes Ph.D. thesis Emese Izsépi Biology Doctoral School, Immunology Program Supervisor:
The modulatory effect of cholesterol rich lipid rafts on the effector functions, differentiation and communication of lymphocytes Ph.D. thesis Emese Izsépi Biology Doctoral School, Immunology Program Supervisor:
Supplementary Figure 1 CD4 + T cells from PKC-θ null mice are defective in NF-κB activation during T cell receptor signaling. CD4 + T cells were
 Supplementary Figure 1 CD4 + T cells from PKC-θ null mice are defective in NF-κB activation during T cell receptor signaling. CD4 + T cells were isolated from wild type (PKC-θ- WT) or PKC-θ null (PKC-θ-KO)
Supplementary Figure 1 CD4 + T cells from PKC-θ null mice are defective in NF-κB activation during T cell receptor signaling. CD4 + T cells were isolated from wild type (PKC-θ- WT) or PKC-θ null (PKC-θ-KO)
SUPPLEMENTARY INFORMATION
 Supplementary Figures Supplementary Figure S1. Binding of full-length OGT and deletion mutants to PIP strips (Echelon Biosciences). Supplementary Figure S2. Binding of the OGT (919-1036) fragments with
Supplementary Figures Supplementary Figure S1. Binding of full-length OGT and deletion mutants to PIP strips (Echelon Biosciences). Supplementary Figure S2. Binding of the OGT (919-1036) fragments with
Lecture 7: Signaling Through Lymphocyte Receptors
 Lecture 7: Signaling Through Lymphocyte Receptors Questions to Consider After recognition of its cognate MHC:peptide, how does the T cell receptor activate immune response genes? What are the structural
Lecture 7: Signaling Through Lymphocyte Receptors Questions to Consider After recognition of its cognate MHC:peptide, how does the T cell receptor activate immune response genes? What are the structural
Multi-Parameter Apoptosis Assay Kit
 Multi-Parameter Apoptosis Assay Kit Catalog Number KA1335 5 x 96 assays Version: 05 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Background... 3 Principle of the Assay...
Multi-Parameter Apoptosis Assay Kit Catalog Number KA1335 5 x 96 assays Version: 05 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Background... 3 Principle of the Assay...
Impact factor: Reporter:4A1H0019 Chen Zi Hao 4A1H0023 Huang Wan ting 4A1H0039 Sue Yi Zhu 4A1H0070 Lin Guan cheng 4A1H0077 Chen Bo xuan
 Curcumin Protects Neonatal Rat Cardiomyocytes against High Glucose-Induced Apoptosis via PI3K/Akt Signalling Pathway Wei Yu,1,2 Wenliang Zha,1 Zhiqiang Ke,1 Qing Min,2 Cairong Li,1 Huirong Sun,3 and Chao
Curcumin Protects Neonatal Rat Cardiomyocytes against High Glucose-Induced Apoptosis via PI3K/Akt Signalling Pathway Wei Yu,1,2 Wenliang Zha,1 Zhiqiang Ke,1 Qing Min,2 Cairong Li,1 Huirong Sun,3 and Chao
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION doi:1.138/nature9814 a A SHARPIN FL B SHARPIN ΔNZF C SHARPIN T38L, F39V b His-SHARPIN FL -1xUb -2xUb -4xUb α-his c Linear 4xUb -SHARPIN FL -SHARPIN TF_LV -SHARPINΔNZF -SHARPIN
SUPPLEMENTARY INFORMATION doi:1.138/nature9814 a A SHARPIN FL B SHARPIN ΔNZF C SHARPIN T38L, F39V b His-SHARPIN FL -1xUb -2xUb -4xUb α-his c Linear 4xUb -SHARPIN FL -SHARPIN TF_LV -SHARPINΔNZF -SHARPIN
Supplementary Information
 Supplementary Information Targeted Disruption of the EZH2/EED Complex Inhibits EZH2- dependent Cancer Woojin Kim 1,2,3, Gregory H. Bird 2,3,4, Tobias Neff 5, Guoji Guo 1,2,3, Marc A. Kerenyi 1,2,3, Loren
Supplementary Information Targeted Disruption of the EZH2/EED Complex Inhibits EZH2- dependent Cancer Woojin Kim 1,2,3, Gregory H. Bird 2,3,4, Tobias Neff 5, Guoji Guo 1,2,3, Marc A. Kerenyi 1,2,3, Loren
TRAF6 ubiquitinates TGFβ type I receptor to promote its cleavage and nuclear translocation in cancer
 Supplementary Information TRAF6 ubiquitinates TGFβ type I receptor to promote its cleavage and nuclear translocation in cancer Yabing Mu, Reshma Sundar, Noopur Thakur, Maria Ekman, Shyam Kumar Gudey, Mariya
Supplementary Information TRAF6 ubiquitinates TGFβ type I receptor to promote its cleavage and nuclear translocation in cancer Yabing Mu, Reshma Sundar, Noopur Thakur, Maria Ekman, Shyam Kumar Gudey, Mariya
MCB*4010 Midterm Exam / Winter 2008
 MCB*4010 Midterm Exam / Winter 2008 Name: ID: Instructions: Answer all 4 questions. The number of marks for each question indicates how many points you need to provide. Write your answers in point form,
MCB*4010 Midterm Exam / Winter 2008 Name: ID: Instructions: Answer all 4 questions. The number of marks for each question indicates how many points you need to provide. Write your answers in point form,
SUPPLEMENTARY INFORMATION
 doi:10.1038/nature11700 Figure 1: RIP3 as a potential Sirt2 interacting protein. Transfected Flag-tagged Sirt2 was immunoprecipitated from cells and eluted from the Sepharose beads using Flag peptide.
doi:10.1038/nature11700 Figure 1: RIP3 as a potential Sirt2 interacting protein. Transfected Flag-tagged Sirt2 was immunoprecipitated from cells and eluted from the Sepharose beads using Flag peptide.
Recent Studies of Phenylketonuria By: Jennifer Gastelum Dr. Koni Stone Copyright 2014
 Recent Studies of Phenylketonuria By: Jennifer Gastelum Dr. Koni Stone Copyright 2014 Phenylketonuria (PKU) is an autosomal recessive genetic disorder that causes an accumulation of toxic metabolites in
Recent Studies of Phenylketonuria By: Jennifer Gastelum Dr. Koni Stone Copyright 2014 Phenylketonuria (PKU) is an autosomal recessive genetic disorder that causes an accumulation of toxic metabolites in
Cell Cycle, Mitosis, and Microtubules. LS1A Final Exam Review Friday 1/12/07. Processes occurring during cell cycle
 Cell Cycle, Mitosis, and Microtubules LS1A Final Exam Review Friday 1/12/07 Processes occurring during cell cycle Replicate chromosomes Segregate chromosomes Cell divides Cell grows Cell Growth 1 The standard
Cell Cycle, Mitosis, and Microtubules LS1A Final Exam Review Friday 1/12/07 Processes occurring during cell cycle Replicate chromosomes Segregate chromosomes Cell divides Cell grows Cell Growth 1 The standard
a surface permeabilized
 a surface permeabilized RAW 64.7 P388D1 J774 b CD11b + Ly-6G - Blood Monocytes WT Supplementary Figure 1. Cell surface expression on macrophages and DCs. (a) RAW64.7, P388D1, and J774 cells were subjected
a surface permeabilized RAW 64.7 P388D1 J774 b CD11b + Ly-6G - Blood Monocytes WT Supplementary Figure 1. Cell surface expression on macrophages and DCs. (a) RAW64.7, P388D1, and J774 cells were subjected
Signaling. Dr. Sujata Persad Katz Group Centre for Pharmacy & Health research
 Signaling Dr. Sujata Persad 3-020 Katz Group Centre for Pharmacy & Health research E-mail:sujata.persad@ualberta.ca 1 Growth Factor Receptors and Other Signaling Pathways What we will cover today: How
Signaling Dr. Sujata Persad 3-020 Katz Group Centre for Pharmacy & Health research E-mail:sujata.persad@ualberta.ca 1 Growth Factor Receptors and Other Signaling Pathways What we will cover today: How
Chapter 20. Cell - Cell Signaling: Hormones and Receptors. Three general types of extracellular signaling. endocrine signaling. paracrine signaling
 Chapter 20 Cell - Cell Signaling: Hormones and Receptors Three general types of extracellular signaling endocrine signaling paracrine signaling autocrine signaling Endocrine Signaling - signaling molecules
Chapter 20 Cell - Cell Signaling: Hormones and Receptors Three general types of extracellular signaling endocrine signaling paracrine signaling autocrine signaling Endocrine Signaling - signaling molecules
C-Phycocyanin (C-PC) is a n«sjfc&c- waefc-jduble phycobiliprotein. pigment isolated from Spirulina platensis. This water- soluble protein pigment is
 ' ^Summary C-Phycocyanin (C-PC) is a n«sjfc&c- waefc-jduble phycobiliprotein pigment isolated from Spirulina platensis. This water- soluble protein pigment is of greater importance because of its various
' ^Summary C-Phycocyanin (C-PC) is a n«sjfc&c- waefc-jduble phycobiliprotein pigment isolated from Spirulina platensis. This water- soluble protein pigment is of greater importance because of its various
Supplementary figure legends
 Supplementary figure legends Supplementary Figure 1. Exposure of CRT occurs independently from the apoptosisassociated loss of the mitochondrial membrane potential (MMP). (A) HeLa cells treated with MTX
Supplementary figure legends Supplementary Figure 1. Exposure of CRT occurs independently from the apoptosisassociated loss of the mitochondrial membrane potential (MMP). (A) HeLa cells treated with MTX
Cells and reagents. Synaptopodin knockdown (1) and dynamin knockdown (2)
 Supplemental Methods Cells and reagents. Synaptopodin knockdown (1) and dynamin knockdown (2) podocytes were cultured as described previously. Staurosporine, angiotensin II and actinomycin D were all obtained
Supplemental Methods Cells and reagents. Synaptopodin knockdown (1) and dynamin knockdown (2) podocytes were cultured as described previously. Staurosporine, angiotensin II and actinomycin D were all obtained
p47 negatively regulates IKK activation by inducing the lysosomal degradation of polyubiquitinated NEMO
 Supplementary Information p47 negatively regulates IKK activation by inducing the lysosomal degradation of polyubiquitinated NEMO Yuri Shibata, Masaaki Oyama, Hiroko Kozuka-Hata, Xiao Han, Yuetsu Tanaka,
Supplementary Information p47 negatively regulates IKK activation by inducing the lysosomal degradation of polyubiquitinated NEMO Yuri Shibata, Masaaki Oyama, Hiroko Kozuka-Hata, Xiao Han, Yuetsu Tanaka,
Regulation of cell function by intracellular signaling
 Regulation of cell function by intracellular signaling Objectives: Regulation principle Allosteric and covalent mechanisms, Popular second messengers, Protein kinases, Kinase cascade and interaction. regulation
Regulation of cell function by intracellular signaling Objectives: Regulation principle Allosteric and covalent mechanisms, Popular second messengers, Protein kinases, Kinase cascade and interaction. regulation
SUPPLEMENT. Materials and methods
 SUPPLEMENT Materials and methods Cell culture and reagents Cell media and reagents were from Invitrogen unless otherwise indicated. Antibiotics and Tet-certified serum were from Clontech. In experiments
SUPPLEMENT Materials and methods Cell culture and reagents Cell media and reagents were from Invitrogen unless otherwise indicated. Antibiotics and Tet-certified serum were from Clontech. In experiments
LPS LPS P6 - + Supplementary Fig. 1.
 P6 LPS - - - + + + - LPS + + - - P6 + Supplementary Fig. 1. Pharmacological inhibition of the JAK/STAT blocks LPS-induced HMGB1 nuclear translocation. RAW 267.4 cells were stimulated with LPS in the absence
P6 LPS - - - + + + - LPS + + - - P6 + Supplementary Fig. 1. Pharmacological inhibition of the JAK/STAT blocks LPS-induced HMGB1 nuclear translocation. RAW 267.4 cells were stimulated with LPS in the absence
W/T Itgam -/- F4/80 CD115. F4/80 hi CD115 + F4/80 + CD115 +
 F4/8 % in the peritoneal lavage 6 4 2 p=.15 n.s p=.76 CD115 F4/8 hi CD115 + F4/8 + CD115 + F4/8 hi CD115 + F4/8 + CD115 + MHCII MHCII Supplementary Figure S1. CD11b deficiency affects the cellular responses
F4/8 % in the peritoneal lavage 6 4 2 p=.15 n.s p=.76 CD115 F4/8 hi CD115 + F4/8 + CD115 + F4/8 hi CD115 + F4/8 + CD115 + MHCII MHCII Supplementary Figure S1. CD11b deficiency affects the cellular responses
Supplementary Figure 1. PD-L1 is glycosylated in cancer cells. (a) Western blot analysis of PD-L1 in breast cancer cells. (b) Western blot analysis
 Supplementary Figure 1. PD-L1 is glycosylated in cancer cells. (a) Western blot analysis of PD-L1 in breast cancer cells. (b) Western blot analysis of PD-L1 in ovarian cancer cells. (c) Western blot analysis
Supplementary Figure 1. PD-L1 is glycosylated in cancer cells. (a) Western blot analysis of PD-L1 in breast cancer cells. (b) Western blot analysis of PD-L1 in ovarian cancer cells. (c) Western blot analysis
Supplementary information. The Light Intermediate Chain 2 Subpopulation of Dynein Regulates Mitotic. Spindle Orientation
 Supplementary information The Light Intermediate Chain 2 Subpopulation of Dynein Regulates Mitotic Spindle Orientation Running title: Dynein LICs distribute mitotic functions. Sagar Mahale a, d, *, Megha
Supplementary information The Light Intermediate Chain 2 Subpopulation of Dynein Regulates Mitotic Spindle Orientation Running title: Dynein LICs distribute mitotic functions. Sagar Mahale a, d, *, Megha
T H E J O U R N A L O F C E L L B I O L O G Y
 T H E J O U R N A L O F C E L L B I O L O G Y Supplemental material Krenn et al., http://www.jcb.org/cgi/content/full/jcb.201110013/dc1 Figure S1. Levels of expressed proteins and demonstration that C-terminal
T H E J O U R N A L O F C E L L B I O L O G Y Supplemental material Krenn et al., http://www.jcb.org/cgi/content/full/jcb.201110013/dc1 Figure S1. Levels of expressed proteins and demonstration that C-terminal
Supplementary Materials for
 www.sciencesignaling.org/cgi/content/full/2/1/ra81/dc1 Supplementary Materials for Delivery of MicroRNA-126 by Apoptotic Bodies Induces CXCL12- Dependent Vascular Protection Alma Zernecke,* Kiril Bidzhekov,
www.sciencesignaling.org/cgi/content/full/2/1/ra81/dc1 Supplementary Materials for Delivery of MicroRNA-126 by Apoptotic Bodies Induces CXCL12- Dependent Vascular Protection Alma Zernecke,* Kiril Bidzhekov,
T cell maturation. T-cell Maturation. What allows T cell maturation?
 T-cell Maturation What allows T cell maturation? Direct contact with thymic epithelial cells Influence of thymic hormones Growth factors (cytokines, CSF) T cell maturation T cell progenitor DN DP SP 2ry
T-cell Maturation What allows T cell maturation? Direct contact with thymic epithelial cells Influence of thymic hormones Growth factors (cytokines, CSF) T cell maturation T cell progenitor DN DP SP 2ry
Supplementary Materials for
 www.sciencesignaling.org/cgi/content/full/6/283/ra57/dc1 Supplementary Materials for JNK3 Couples the Neuronal Stress Response to Inhibition of Secretory Trafficking Guang Yang,* Xun Zhou, Jingyan Zhu,
www.sciencesignaling.org/cgi/content/full/6/283/ra57/dc1 Supplementary Materials for JNK3 Couples the Neuronal Stress Response to Inhibition of Secretory Trafficking Guang Yang,* Xun Zhou, Jingyan Zhu,
Plasma exposure levels from individual mice 4 hours post IP administration at the
 Supplemental Figure Legends Figure S1. Plasma exposure levels of MKC-3946 in mice. Plasma exposure levels from individual mice 4 hours post IP administration at the indicated dose mg/kg. Data represent
Supplemental Figure Legends Figure S1. Plasma exposure levels of MKC-3946 in mice. Plasma exposure levels from individual mice 4 hours post IP administration at the indicated dose mg/kg. Data represent
Stewart et al. CD36 ligands promote sterile inflammation through assembly of a TLR 4 and 6 heterodimer
 NFκB (fold induction) Stewart et al. ligands promote sterile inflammation through assembly of a TLR 4 and 6 heterodimer a. mrna (fold induction) 5 4 3 2 1 LDL oxldl Gro1a MIP-2 RANTES mrna (fold induction)
NFκB (fold induction) Stewart et al. ligands promote sterile inflammation through assembly of a TLR 4 and 6 heterodimer a. mrna (fold induction) 5 4 3 2 1 LDL oxldl Gro1a MIP-2 RANTES mrna (fold induction)
Gelsolin activity controls efficient early HIV-1 infection
 García-Expósito et al. Retrovirology 2013, 10:39 RESEARCH Open Access Gelsolin activity controls efficient early HIV-1 infection Laura García-Expósito 1, Serena Ziglio 1,2, Jonathan Barroso-González 1,
García-Expósito et al. Retrovirology 2013, 10:39 RESEARCH Open Access Gelsolin activity controls efficient early HIV-1 infection Laura García-Expósito 1, Serena Ziglio 1,2, Jonathan Barroso-González 1,
A mechanism for glycoconjugate vaccine activation of the adaptive immune system and its implications for vaccine design
 A mechanism for glycoconjugate vaccine activation of the adaptive immune system and its implications for vaccine design Fikri Y. Avci 1,2, Xiangming Li 3, Moriya Tsuji 3, Dennis L. Kasper 1,2* Supplementary
A mechanism for glycoconjugate vaccine activation of the adaptive immune system and its implications for vaccine design Fikri Y. Avci 1,2, Xiangming Li 3, Moriya Tsuji 3, Dennis L. Kasper 1,2* Supplementary
HOMEWORK RUBRICS MECHANOTRANSDUCTION UNIT: HOMEWORK #1 (20 pts towards your grade)
 HOMEWORK RUBRICS MECHANOTRANSDUCTION UNIT: HOMEWORK #1 (20 pts towards your grade) 1. Mesenchymal stem cells (MSC) cultured on extracellular matrices with different stiffness exhibit diverse lineage commitment
HOMEWORK RUBRICS MECHANOTRANSDUCTION UNIT: HOMEWORK #1 (20 pts towards your grade) 1. Mesenchymal stem cells (MSC) cultured on extracellular matrices with different stiffness exhibit diverse lineage commitment
Complexity DNA. Genome RNA. Transcriptome. Protein. Proteome. Metabolites. Metabolome
 DNA Genome Complexity RNA Transcriptome Systems Biology Linking all the components of a cell in a quantitative and temporal manner Protein Proteome Metabolites Metabolome Where are the functional elements?
DNA Genome Complexity RNA Transcriptome Systems Biology Linking all the components of a cell in a quantitative and temporal manner Protein Proteome Metabolites Metabolome Where are the functional elements?
Supplementary Figures
 Supplementary Figures Figure S1. Validation of kinase regulators of ONC201 sensitivity. Validation and screen results for changes in cell viability associated with the combination of ONC201 treatment (1
Supplementary Figures Figure S1. Validation of kinase regulators of ONC201 sensitivity. Validation and screen results for changes in cell viability associated with the combination of ONC201 treatment (1
Altered lipid raft associated signaling and ganglioside expression in T lymphocytes from patients with systemic lupus erythematosus
 Research article Altered lipid raft associated signaling and ganglioside expression in T lymphocytes from patients with systemic lupus erythematosus Elizabeth C. Jury, 1 Panagiotis S. Kabouridis, 2 Fabian
Research article Altered lipid raft associated signaling and ganglioside expression in T lymphocytes from patients with systemic lupus erythematosus Elizabeth C. Jury, 1 Panagiotis S. Kabouridis, 2 Fabian
(a) Significant biological processes (upper panel) and disease biomarkers (lower panel)
 Supplementary Figure 1. Functional enrichment analyses of secretomic proteins. (a) Significant biological processes (upper panel) and disease biomarkers (lower panel) 2 involved by hrab37-mediated secretory
Supplementary Figure 1. Functional enrichment analyses of secretomic proteins. (a) Significant biological processes (upper panel) and disease biomarkers (lower panel) 2 involved by hrab37-mediated secretory
genome edited transient transfection, CMV promoter
 Supplementary Figure 1. In the absence of new protein translation, overexpressed caveolin-1-gfp is degraded faster than caveolin-1-gfp expressed from the endogenous caveolin 1 locus % loss of total caveolin-1-gfp
Supplementary Figure 1. In the absence of new protein translation, overexpressed caveolin-1-gfp is degraded faster than caveolin-1-gfp expressed from the endogenous caveolin 1 locus % loss of total caveolin-1-gfp
SUPPLEMENTARY INFORMATION
 Supplemental Figure 1. Furin is efficiently deleted in CD4 + and CD8 + T cells. a, Western blot for furin and actin proteins in CD4cre-fur f/f and fur f/f Th1 cells. Wild-type and furin-deficient CD4 +
Supplemental Figure 1. Furin is efficiently deleted in CD4 + and CD8 + T cells. a, Western blot for furin and actin proteins in CD4cre-fur f/f and fur f/f Th1 cells. Wild-type and furin-deficient CD4 +
Supplementary Figure 1 Role of Raf-1 in TLR2-Dectin-1-mediated cytokine expression
 Supplementary Figure 1 Supplementary Figure 1 Role of Raf-1 in TLR2-Dectin-1-mediated cytokine expression. Quantitative real-time PCR of indicated mrnas in DCs stimulated with TLR2-Dectin-1 agonist zymosan
Supplementary Figure 1 Supplementary Figure 1 Role of Raf-1 in TLR2-Dectin-1-mediated cytokine expression. Quantitative real-time PCR of indicated mrnas in DCs stimulated with TLR2-Dectin-1 agonist zymosan
Supplementary Material
 Supplementary Material accompanying the manuscript Interleukin 37 is a fundamental inhibitor of innate immunity Marcel F Nold, Claudia A Nold-Petry, Jarod A Zepp, Brent E Palmer, Philip Bufler & Charles
Supplementary Material accompanying the manuscript Interleukin 37 is a fundamental inhibitor of innate immunity Marcel F Nold, Claudia A Nold-Petry, Jarod A Zepp, Brent E Palmer, Philip Bufler & Charles
Enzyme-coupled Receptors. Cell-surface receptors 1. Ion-channel-coupled receptors 2. G-protein-coupled receptors 3. Enzyme-coupled receptors
 Enzyme-coupled Receptors Cell-surface receptors 1. Ion-channel-coupled receptors 2. G-protein-coupled receptors 3. Enzyme-coupled receptors Cell-surface receptors allow a flow of ions across the plasma
Enzyme-coupled Receptors Cell-surface receptors 1. Ion-channel-coupled receptors 2. G-protein-coupled receptors 3. Enzyme-coupled receptors Cell-surface receptors allow a flow of ions across the plasma
NLRX1: 5 -GCTCCATGGCTTAGAGCATC-3 (forward) 5 -AACTCCTCCTCCGTCCTGAT-3 (reverse) β-actin
 NLRX1 β-actin 1 2 3 4 5 6 1 2 3 4 5 6 NLRX1 (667 bp) β-actin (523 bp) Supplementary Figure 1: Expression of NLRX1 in human cell lines. 1: HeLa, 2: HEK293T, 3: MCF-7, 4:Ramos, 5:Jurkat, 6: THP1. The following
NLRX1 β-actin 1 2 3 4 5 6 1 2 3 4 5 6 NLRX1 (667 bp) β-actin (523 bp) Supplementary Figure 1: Expression of NLRX1 in human cell lines. 1: HeLa, 2: HEK293T, 3: MCF-7, 4:Ramos, 5:Jurkat, 6: THP1. The following
Actin cytoskeletal dynamics in T lymphocyte activation and migration
 Actin cytoskeletal dynamics in T lymphocyte activation and migration Yvonne Samstag, Sybille M. Eibert, Martin Klemke, and Guido H. Wabnitz Institute for Immunology, Ruprecht-Karls-University, Heidelberg,
Actin cytoskeletal dynamics in T lymphocyte activation and migration Yvonne Samstag, Sybille M. Eibert, Martin Klemke, and Guido H. Wabnitz Institute for Immunology, Ruprecht-Karls-University, Heidelberg,
Supporting Information. FADD regulates NF-кB activation and promotes ubiquitination of cflip L to induce. apoptosis
 1 2 Supporting Information 3 4 5 FADD regulates NF-кB activation and promotes ubiquitination of cflip L to induce apoptosis 6 7 Kishu Ranjan and Chandramani Pathak* 8 9 Department of Cell Biology, School
1 2 Supporting Information 3 4 5 FADD regulates NF-кB activation and promotes ubiquitination of cflip L to induce apoptosis 6 7 Kishu Ranjan and Chandramani Pathak* 8 9 Department of Cell Biology, School
Incorporation of photo-caged lysine (pc-lys) at K273 of human LCK allows specific control of the enzyme activity.
 Supplementary Figure 1 Incorporation of photo-caged lysine (pc-lys) at K273 of human LCK allows specific control of the enzyme activity. (a) Modeling of the kinase domain of LCK with ATP (left) or pc-lys
Supplementary Figure 1 Incorporation of photo-caged lysine (pc-lys) at K273 of human LCK allows specific control of the enzyme activity. (a) Modeling of the kinase domain of LCK with ATP (left) or pc-lys
Supplemental Materials Molecular Biology of the Cell
 Supplemental Materials Molecular Biology of the Cell Gilberti et al. SUPPLEMENTAL FIGURE LEGENDS: Figure S1: The effect of pharmacological inhibitors on particle uptake. The data presented in Figure 1
Supplemental Materials Molecular Biology of the Cell Gilberti et al. SUPPLEMENTAL FIGURE LEGENDS: Figure S1: The effect of pharmacological inhibitors on particle uptake. The data presented in Figure 1
Nature Medicine: doi: /nm.4078
 Supplementary Figure 1. Cetuximab induces ER stress response in DiFi cells. (a) Scheme of SILAC proteome. (b) MS-base read out of SILAC experiment. The histogram of log 2 -transformed normalized H/L ratios
Supplementary Figure 1. Cetuximab induces ER stress response in DiFi cells. (a) Scheme of SILAC proteome. (b) MS-base read out of SILAC experiment. The histogram of log 2 -transformed normalized H/L ratios
Supplemental Table I.
 Supplemental Table I Male / Mean ± SEM n Mean ± SEM n Body weight, g 29.2±0.4 17 29.7±0.5 17 Total cholesterol, mg/dl 534.0±30.8 17 561.6±26.1 17 HDL-cholesterol, mg/dl 9.6±0.8 17 10.1±0.7 17 Triglycerides,
Supplemental Table I Male / Mean ± SEM n Mean ± SEM n Body weight, g 29.2±0.4 17 29.7±0.5 17 Total cholesterol, mg/dl 534.0±30.8 17 561.6±26.1 17 HDL-cholesterol, mg/dl 9.6±0.8 17 10.1±0.7 17 Triglycerides,
SUPPLEMENTARY INFORMATION
 Supplementary Discussion The cell cycle machinery and the DNA damage response network are highly interconnected and co-regulated in assuring faithful duplication and partition of genetic materials into
Supplementary Discussion The cell cycle machinery and the DNA damage response network are highly interconnected and co-regulated in assuring faithful duplication and partition of genetic materials into
Tbk1-TKO! DN cells (%)! 15! 10!
 a! T Cells! TKO! B Cells! TKO! b! CD4! 8.9 85.2 3.4 2.88 CD8! Tbk1-TKO! 1.1 84.8 2.51 2.54 c! DN cells (%)! 4 3 2 1 DP cells (%)! 9 8 7 6 CD4 + SP cells (%)! 5 4 3 2 1 5 TKO! TKO! TKO! TKO! 15 1 5 CD8
a! T Cells! TKO! B Cells! TKO! b! CD4! 8.9 85.2 3.4 2.88 CD8! Tbk1-TKO! 1.1 84.8 2.51 2.54 c! DN cells (%)! 4 3 2 1 DP cells (%)! 9 8 7 6 CD4 + SP cells (%)! 5 4 3 2 1 5 TKO! TKO! TKO! TKO! 15 1 5 CD8
El Azzouzi et al., http ://www.jcb.org /cgi /content /full /jcb /DC1
 Supplemental material JCB El Azzouzi et al., http ://www.jcb.org /cgi /content /full /jcb.201510043 /DC1 THE JOURNAL OF CELL BIOLOGY Figure S1. Acquisition of -phluorin correlates negatively with podosome
Supplemental material JCB El Azzouzi et al., http ://www.jcb.org /cgi /content /full /jcb.201510043 /DC1 THE JOURNAL OF CELL BIOLOGY Figure S1. Acquisition of -phluorin correlates negatively with podosome
7.06 Cell Biology EXAM #3 April 24, 2003
 7.06 Spring 2003 Exam 3 Name 1 of 8 7.06 Cell Biology EXAM #3 April 24, 2003 This is an open book exam, and you are allowed access to books and notes. Please write your answers to the questions in the
7.06 Spring 2003 Exam 3 Name 1 of 8 7.06 Cell Biology EXAM #3 April 24, 2003 This is an open book exam, and you are allowed access to books and notes. Please write your answers to the questions in the
Procaspase-3. Cleaved caspase-3. actin. Cytochrome C (10 M) Z-VAD-fmk. Procaspase-3. Cleaved caspase-3. actin. Z-VAD-fmk
 A HeLa actin - + + - - + Cytochrome C (1 M) Z-VAD-fmk PMN - + + - - + actin Cytochrome C (1 M) Z-VAD-fmk Figure S1. (A) Pan-caspase inhibitor z-vad-fmk inhibits cytochrome c- mediated procaspase-3 cleavage.
A HeLa actin - + + - - + Cytochrome C (1 M) Z-VAD-fmk PMN - + + - - + actin Cytochrome C (1 M) Z-VAD-fmk Figure S1. (A) Pan-caspase inhibitor z-vad-fmk inhibits cytochrome c- mediated procaspase-3 cleavage.
Validation & Assay Performance Summary
 Validation & Assay Performance Summary CellSensor DBE-bla MDA-MB-468 Cell Line Cat. no. K1814 Pathway Description The phosphatidylinositol-3-kinase (PI3K) signaling cascade is essential for cell growth
Validation & Assay Performance Summary CellSensor DBE-bla MDA-MB-468 Cell Line Cat. no. K1814 Pathway Description The phosphatidylinositol-3-kinase (PI3K) signaling cascade is essential for cell growth
Supplemental Materials. STK16 regulates actin dynamics to control Golgi organization and cell cycle
 Supplemental Materials STK16 regulates actin dynamics to control Golgi organization and cell cycle Juanjuan Liu 1,2,3, Xingxing Yang 1,3, Binhua Li 1, Junjun Wang 1,2, Wenchao Wang 1, Jing Liu 1, Qingsong
Supplemental Materials STK16 regulates actin dynamics to control Golgi organization and cell cycle Juanjuan Liu 1,2,3, Xingxing Yang 1,3, Binhua Li 1, Junjun Wang 1,2, Wenchao Wang 1, Jing Liu 1, Qingsong
Muse Assays for Cell Analysis
 Muse Assays for Cell Analysis Multiple Assay Outputs for Cell Analysis Cell Health Cell Signalling Immunology Muse Count & Viability Kit Muse Cell Cycle Kit Muse Annexin V & Dead Cell Kit Muse Caspase
Muse Assays for Cell Analysis Multiple Assay Outputs for Cell Analysis Cell Health Cell Signalling Immunology Muse Count & Viability Kit Muse Cell Cycle Kit Muse Annexin V & Dead Cell Kit Muse Caspase
Activation-induced cell death (AICD) 1 in CD4 Th has
 Unequal Death in T Helper Cell (Th)1 and Th2 Effectors: Th1, but not Th2, Effectors Undergo Rapid Fas/FasL-mediated Apoptosis By Xiaohong Zhang,* Thomas Brunner, Laura Carter,* Richard W. Dutton,* Paul
Unequal Death in T Helper Cell (Th)1 and Th2 Effectors: Th1, but not Th2, Effectors Undergo Rapid Fas/FasL-mediated Apoptosis By Xiaohong Zhang,* Thomas Brunner, Laura Carter,* Richard W. Dutton,* Paul
The Biochemistry of apoptosis
 The Biochemistry of apoptosis 1 1 The apoptosis is composed of multiple biochemical events 2 2 Biochemical, cellular, and molecular events in Apoptosis 1. Membrane blebbing; phosphatidyl serine exposure
The Biochemistry of apoptosis 1 1 The apoptosis is composed of multiple biochemical events 2 2 Biochemical, cellular, and molecular events in Apoptosis 1. Membrane blebbing; phosphatidyl serine exposure
Supplementary Data 1. Alanine substitutions and position variants of APNCYGNIPL. Applied in
 Supplementary Data 1. Alanine substitutions and position variants of APNCYGNIPL. Applied in Supplementary Fig. 2 Substitution Sequence Position variant Sequence original APNCYGNIPL original APNCYGNIPL
Supplementary Data 1. Alanine substitutions and position variants of APNCYGNIPL. Applied in Supplementary Fig. 2 Substitution Sequence Position variant Sequence original APNCYGNIPL original APNCYGNIPL
SD-1 SD-1: Cathepsin B levels in TNF treated hch
 SD-1 SD-1: Cathepsin B levels in TNF treated hch. A. RNA and B. protein extracts from TNF treated and untreated human chondrocytes (hch) were analyzed via qpcr (left) and immunoblot analyses (right) for
SD-1 SD-1: Cathepsin B levels in TNF treated hch. A. RNA and B. protein extracts from TNF treated and untreated human chondrocytes (hch) were analyzed via qpcr (left) and immunoblot analyses (right) for
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION doi:10.10/nature10195 NCBI gene: Tagged Subunit(s: HA-Vpx; FLAG-Cul4 HA-DCAF1 FLAG-Cul4 HA-FLAG-Vpx Mock Vpx (SIVmac 100 (a ; 0.159 (b ; 0.05 DCAF1 DDB1 DDA1 Cul4A 1; 0.024591
SUPPLEMENTARY INFORMATION doi:10.10/nature10195 NCBI gene: Tagged Subunit(s: HA-Vpx; FLAG-Cul4 HA-DCAF1 FLAG-Cul4 HA-FLAG-Vpx Mock Vpx (SIVmac 100 (a ; 0.159 (b ; 0.05 DCAF1 DDB1 DDA1 Cul4A 1; 0.024591
BIOLOGY 103 Spring 2001 MIDTERM LAB SECTION
 BIOLOGY 103 Spring 2001 MIDTERM NAME KEY LAB SECTION ID# (last four digits of SS#) STUDENT PLEASE READ. Do not put yourself at a disadvantage by revealing the content of this exam to your classmates. Your
BIOLOGY 103 Spring 2001 MIDTERM NAME KEY LAB SECTION ID# (last four digits of SS#) STUDENT PLEASE READ. Do not put yourself at a disadvantage by revealing the content of this exam to your classmates. Your
Receptor mediated Signal Transduction
 Receptor mediated Signal Transduction G-protein-linked receptors adenylyl cyclase camp PKA Organization of receptor protein-tyrosine kinases From G.M. Cooper, The Cell. A molecular approach, 2004, third
Receptor mediated Signal Transduction G-protein-linked receptors adenylyl cyclase camp PKA Organization of receptor protein-tyrosine kinases From G.M. Cooper, The Cell. A molecular approach, 2004, third
Supplementary Table; Supplementary Figures and legends S1-S21; Supplementary Materials and Methods
 Silva et al. PTEN posttranslational inactivation and hyperactivation of the PI3K/Akt pathway sustain primary T cell leukemia viability Supplementary Table; Supplementary Figures and legends S1-S21; Supplementary
Silva et al. PTEN posttranslational inactivation and hyperactivation of the PI3K/Akt pathway sustain primary T cell leukemia viability Supplementary Table; Supplementary Figures and legends S1-S21; Supplementary
Page 39 of 44. 8h LTA & AT h PepG & AT h LTA
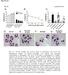 Page 39 of 44 Fig. S1 A: B: C: D: 8h LTA 8h LTA & AT7519 E: F: 8h PepG G: 8h PepG & AT7519 Fig. S1. AT7519 overrides the survival effects of lipoteichoic acid (LTA) and peptidoglycan (PepG). (A) Human
Page 39 of 44 Fig. S1 A: B: C: D: 8h LTA 8h LTA & AT7519 E: F: 8h PepG G: 8h PepG & AT7519 Fig. S1. AT7519 overrides the survival effects of lipoteichoic acid (LTA) and peptidoglycan (PepG). (A) Human
