Age-Related Conjunctival Disease in the C57BL/6.NOD- Aec1Aec2 Mouse Model of Sjögren Syndrome Develops Independent of Lacrimal Dysfunction
|
|
|
- Carmella Clarke
- 5 years ago
- Views:
Transcription
1 Cornea Age-Related Conjunctival Disease in the C57BL/6.NOD- Aec1Aec2 Mouse Model of Sjögren Syndrome Develops Independent of Lacrimal Dysfunction In-Cheon You, 1,2 Fang Bian, 2 Eugene A. Volpe, 2 Cintia S. de Paiva, 2 and Stephen C. Pflugfelder 2 1 Department of Ophthalmology, Research Institute of Clinical Medicine of Chonbuk National University Biomedical Research Institute, Chonbuk National University Hospital, Jeonju, Jeonbuk, Korea 2 Ocular Surface Center, Department of Ophthalmology, Cullen Eye Institute, Baylor College of Medicine, Houston, Texas, United States Correspondence: Stephen C. Pflugfelder, Department of Ophthalmology, Cullen Eye Institute, Baylor College of Medicine, 6565 Fannin Street, NC 505, Houston, TX 77030, USA; stevenp@bcm.edu. Submitted: September 11, 2014 Accepted: February 27, 2015 Citation: You I-C, Bian F, Volpe EA, de Paiva CS, Pflugfelder SC. Age-related conjunctival disease in the C57BL/ 6.NOD-Aec1Aec2 mouse model of Sjögren syndrome develops independent of lacrimal dysfunction. Invest Ophthalmol Vis Sci. 2015;56: DOI: /iovs PURPOSE. To investigate parameters of ocular surface disease in C57BL/6.NOD-Aec1Aec2 (Aec) mice with aging and their correlation with development of Sjögren syndrome (SS) like lacrimal gland (LG) disease. METHODS. Aec and C57BL/6 wild-type (B6) female mice were evaluated at 4, 12, and 20 weeks of age. Whole LG and eyes and adnexa were excised for histology and gene expression analysis and evaluated by flow cytometry and immunohistochemistry. Tear volume and goblet cell density was measured. Quantitative PCR evaluated T-cell related cytokine expression in cornea and conjunctiva. RESULTS. Both strains showed age-related conjunctival goblet cell loss that was more pronounced in the Aec strain and significantly greater than in B6 mice at 12 weeks. This was accompanied by CD4 þ T-cell infiltration of the conjunctiva that was greater in Aec strain at 20 weeks. Aec mice had higher levels of IL-17A, IL-17R, IL-1a, IL-1b, and TNF-a in the conjunctiva, and they significantly increase with aging. Aec mice had greater lymphocytic infiltration of the LG and conjunctiva at 20 weeks that consisted of a mixture of CD4 þ and CD8 þ cells. Flow cytometry showed a significant increase in CD4 þ T cells in Aec LG compared to B6 mice. Tear volume was significantly increased in both strains at 20 weeks. CONCLUSIONS. Aec mice developed greater conjunctival goblet cell loss associated with lymphocytic infiltration of the LG and conjunctiva with aging. Increased expression of certain T helper or inflammatory cytokines in these tissues was observed in Aec mice. The conjunctival disease appeared to be due to inflammation and not a decrease in tear volume. Keywords: C57BL/6.NOD-Aec1Aec2, inflammation, Sjögren syndrome (SS) like disease Sjögren syndrome (SS) is a chronic systemic autoimmune disease that targets primarily the salivary and lacrimal glands (LGs), resulting in severe dry mouth (stomatitis sicca) and dry eye (keratoconjunctivitis sicca), respectively. 1 Despite extensive efforts to define the genetic, environmental, and/or immunologic basis of human SS, the underlying etiology remains relatively unclear. This is due, in part, to the delay in diagnosis to an average 7 to 10 years after first symptoms, patients are seen in clinics only after onset of overt clinical disease, and comparative controls are unlikely to volunteer for exocrine gland biopsies. So, in an effort to identify the underlying pathogenesis of SS in humans, numerous mouse models in which various aspects of SS appear spontaneously or are experimentally induced are being intensively investigated. 2 Typically, these mouse models show lymphocyte infiltration of the lacrimal and salivary glands, increased expressions of proinflammatory and helper T helper (Th) cytokines, generation of autoantibodies (especially ANAs [antinuclear antibodies] and anti-m3r antibodies), and eventually decreased tear and saliva secretion. In recent years, several mouse strains, including nonobese diabetic (NOD), MRL/lpr, CD25 knockout (KO) strains, and C57BL/6.NOD-Aec1Aec2 (Aec) mouse, have been used to study the immunopathogenic mechanisms of SS. 2 9 The Aec mouse is a recently generated model in which two genetic intervals, the Idd3 region (designated Aec1, autoimmune exocrinopathy) of chromosome 3 and the Idd5 (designated Aec2) of chromosome 1, derived from the NOD mouse were bred into the SS-nonsusceptible C57BL/6 (B6) mouse. 7 These two genetic intervals have been shown to be responsible for a SS-like disease manifestation to develop in the NOD. Because Aec mice have a B6 genetic background, these mice do not exhibit signs of autoimmune diabetes and abnormal development of LGs, and LG histology is normal prior to onset of SSlike disease. 2 Aec mice exhibit several features found in human SS, including lymphocytic infiltration of salivary glands and LGs, reduced tear production, and the presence of serum autoantibodies. 6 9 However, the disease process is not fully synchronized between individual mice, 8 and the ocular surface disease has not been described. Therefore, in the present study, we compared LGs and ocular surface alterations between Aec and B6 strains at various time points. Copyright 2015 The Association for Research in Vision and Ophthalmology, Inc. j ISSN:
2 C56BL/6.NOD-Aec1Aec2 Mice IOVS j April 2015 j Vol. 56 j No. 4 j 2225 METHODS Animals This research protocol was approved by the Baylor College of Medicine Center for Comparative Medicine, and it conformed to the standards of the ARVO Statement for the Use of Animals in Ophthalmic and Vision Research. Breeding pairs of Aec mice in a B6 background were a kind gift from Ammon B. Peck (University of Florida, Gainesville, FL, USA) for establishing breeder colonies. They were housed in our vivarium in a pathogen-free environment that is managed by Baylor Center for Comparative Medicine (Houston, TX, USA). Mice were exposed to a 12-hour light cycle while in the vivarium with timers controlling the light/dark cycle. Aec female mice were evaluated at 4, 12, and 20 weeks of age, and age-matched wild-type B6 female mice were used as controls. A minimum of 33 animals per time point (4, 12, and 20 weeks) per strain were used: 3 mice for histologic sections, 8 for evaluation of corneal permeability, 10 for tear collection, 6 for flow cytometry, and 6 for gene expression studies. An additional 20 mice were used for tear collection and flow cytometry at 4 and 20 weeks in both strains. Histology, PAS Staining, and Immunohistochemistry Eyes with ocular adnexa and extraorbital LGs were surgically excised, fixed in 10% formalin, and embedded in paraffin, and 8-lm sections were cut. Sections were stained with hematoxylin and eosin for evaluation of morphology. For immunohistochemistry, extraorbital LGs and the eyes and adnexa of mice from each age per strain (n ¼ 3) were excised, embedded in optimal cutting temperature compound (OCT compound; VWR International, Suwanee, GA, USA) and flash-frozen in liquid nitrogen. Sagittal 8-lm sections were cut with a cryostat (HM 500; Micron, Waldorf, Germany) and placed on glass slides that were stored at 808C. Immunohistochemistry was performed in LGs and in the conjunctival epithelium to detect and count cells that stained positively for CD4 (clone H129.9, 10 lg/ml; BD Biosciences, San Diego, CA, USA), CD8a (clone 53e6.7, lg/ml; BD Biosciences), or CD19 (clone 6D5, 2 lg/ml; Abcam, Cambridge, MA, USA) antibodies as previously described. 10 Positively stained cells were counted in the goblet cell rich area of the palpebral conjunctiva using image analysis software (NIS Elements Software, version 3.0; Nikon, Inc., Melville, NY, USA). Results were expressed as the number of positive cells per 100 lm. For PAS staining, the eyes with surrounding eyelids were fixed in 10% buffered formalin overnight and embedded in paraffin. Serial sections 5 lm thick were cut from each sample using a microtome (Microm HM 340E, Thermoscientific, Wilmington, DE, USA). The sections were deparaffinized and stained with 0.5% PAS for identification of goblet cells. The number of positively stained goblet cells in the superior and inferior conjunctiva was counted, and the length of the basement membrane between the first and last goblet cell was measured. The data are presented as the average number of goblet cells per millimeter per eyelid. RNA Isolation and Real-Time PCR Total RNA from conjunctiva, corneal epithelium, and LG (individually collected and individually pooled) from Aec and B6 mice at 4, 12, and 20 weeks was extracted by using RNeasy Plus Mini and RNeasy Plus Micro Kit (Qiagen s RNA isolation kit; Qiagen, Inc., Valencia, CA, USA) according to the manufacturer s instructions, quantified by a spectrophotometer (NanoDrop ND-1000; Thermo Scientific, Wilmington, DE, USA), and stored at 808C. A minimum of six samples per strain and age were used, and one sample consisted of pooled samples of right and left eyes and LGs of the same animal. Samples were treated with DNase (Qiagen, Inc.) to prevent genomic DNA contamination, according to the manufacturer s instructions. First-strand complementary DNA was synthesized from 1 lg of total RNA using random hexamers and Moloney murine leukemia virus (M-MuLV) reverse transcriptase (Ready-To-Go You-Prime First-Strand Beads; GE Healthcare, Arlington Heights, NJ, USA), as previously described. 5 Quantitative real-time PCR was performed with specific minor groove binder (MGB) probes (Taqman; Applied Biosystems, Foster City, CA, USA) and PCR master mix (Taqman Gene Expression Master Mix) in a commercial thermocycling system (StepOnePlus Real-Time PCR System; Applied Biosystems). Murine MGB probes were IFN-c (Mm _m1), IL-1a (Mm _m1), IL-1b (Mm _m1), IL-6 (Mm _m1), IL-13 (Mm _m1), IL-17A (Mm _m1), TNF-a (Mm _m1), matrix metalloproteinase (MMP)-3 (Mm _m1), MMP-9 (Mm _m1), small proline-rich protein 2a (SPRR2a; Mm _m1), major histocompatibility complex (MHC) II (Mm _m1), T-box transcription factor (T-BET; Mm _m1), GATA-binding protein (GATA)-3 (Mm _m1), RAR-related orphan receptor C (RORC; Mm _m1), and interleukin-17 receptor (IL-17R; Mm _m1). The hypoxanthine phosphoribosyltransferase 1 (HPRT-1) (Mm _m1) gene was used as an endogenous reference for each reaction. The results of quantitative PCR were analyzed by the comparative C t method in which the target of change ¼ 2 DDCt was normalized by the C t value of HPRT-1 and the mean C t of relative mrna level in the B6 group at 4 weeks of age was used as the calibrator. Tear Volume Measurements Phenol red thread tests were used to measure tear volume, as previously described. 11 Briefly, a phenol red impregnated thread (ZONE-QUICK; Showa Yakuhin Kako Co., Tokyo, Japan) was held in the lateral canthus of each eye with forceps. A color change from yellow to red occurs in the thread when tears are absorbed. After 20 seconds, the thread was removed and the entire wet (red) portion was measured using imaging software (NIS Element-BR, version 3.0; Nikon, Inc.) after photographing with a microscope equipped with a digital camera (Eclipse E400 with a DS-Fi1; Nikon, Inc.). The distance was converted to volume according to a previously published standard. 11 Corneal Permeability Corneal epithelial permeability to Oregon green dextran (OGD; 70,000 MWt; Invitrogen, Eugene, OR, USA) was assessed at each time point (4, 12, and 20 weeks) in Aec and B6 mice, as previously described. 12 Briefly, 0.5 ll of 50 mg/ml OGD was instilled onto the ocular surface 1 minute before euthanasia. The severity of corneal OGD staining was graded in digital images in the 2-mm central zone of each cornea by two masked observers, using NIS Elements Software (Nikon, Inc.). The mean fluorescent intensity measured by the software inside this central zone was transferred to a database and results averaged within each group. Results are presented as means 6 SEM of gray levels.
3 C56BL/6.NOD-Aec1Aec2 Mice IOVS j April 2015 j Vol. 56 j No. 4 j 2226 Tear Washing and Epidermal Growth Factor (EGF) ELISA Tear washings were collected from each age group from Aec and B6 mice, and tear EGF concentration was measured by a previously reported method. 5 One sample consisted of tear washings pooled from both eyes of one mouse (2 ll) in PBS plus 0.1% bovine serum albumin (8 ll) and stored at 808C until the assay was performed. Tear EGF washings were collected from the same mice used for tear volume measurements on 2 consecutive days. Results are presented as means 6 SEM (in picograms per milliliter). Immunoglobulin Measurement in Tears The levels of immunoglobulins (Ig; IgA, IgG1, IgG2b, and IgM) were measured in tears using an immunoglobulin in an isotyping magnetic bead panel (Milliplex Map Kit; EMD Millipore Corp., Temecula, CA, USA). One sample consisted of tear washings pooled from both eyes of 10 mice (20 ll) in PBS plus 0.1% bovine serum albumin (5 ll) and stored at 808C until the assay was performed. Four samples from Aec and B6 mice at 4 and 20 weeks of age were used. Tear washings were collected for 3 consecutive days from mice and were later used for flow cytometry. A 25 ll sample was used according to the manufacturer s protocol. Sera were collected from B6 mice at 4 weeks of age (n ¼ 3) through cardiac puncture immediately after euthanasia, frozen at 808C until ready to use, and then used as positive controls. Labeled immunoglobulins (kappa light chain) were read by Luminex (Luminex 100 analyser with xponent 3.1 software upgrade; Luminex Corp., Austin, TX, USA). At least 50 events per bead were read, and the data were analyzed using software (Milliplex Analyst; EMD Millipore Corp.). Biological replicates were averaged, and data are presented as picograms per milliliter. Flow Cytometry Analysis of Infiltrating Cells Single-cell suspensions of LGs from Aec and B6 mice at 4, 12, and 20 weeks were prepared as previously reported. 5 In brief, right and left extraorbital LGs were excised, rinsed, and subjected to collagenase digestion for 1 hour at 378C under constant agitation. Collagenase was neutralized by adding complete RPMI with fetal bovine serum (FBS); cells were filtered by using a 70-lm cell strainer, centrifuged, and resuspended. Cells were stained with anti-cd16/32 (BD Biosciences), followed by cell-surface staining with FITCconjugated anti-cd4 (GK 1.5; BD Biosciences), PE-anti-CD8 (clone ; BD Biosciences), APC-B220 (clone RA3-6B2; BD Biosciences), APC-CXCR3 (clone ; R&D Systems, Minneapolis, MN, USA), PE-CCR6 (clone ; R&D Systems), or Pacific Blue-CD3 (clone 500A2; BD Biosciences). Single-cell preparations of splenocytes or cervical lymph nodes obtained from 4-week mice were stained with the same antibodies and served as positive controls. Propidium iodide exclusion was used to discriminate live cells. For evaluation of regulatory T cells (Treg) in both strains at 4 and 20 weeks of age, cells were prepared as described above and incubated with a stain kit (LIVE/DEAD Fixable Blue Dead Cell Stain Kit [L-23105, Invitrogen-Molecular Probes, Eugene, OR, USA]), fixed and permeabilized (CytoFix/Cytoperm; BD Biosciences), followed by staining with PE-CD25 (clone 7D-4; BD Biosciences), APC-Foxp-3 (clone FJK-16s; Ebioscience, San Diego, CA, USA), and Pacific Blue-CD45 (clone 30-F11; Biolegend, San Diego, CA, USA). Cells were kept on ice until flow cytometry analysis was performed. A BD LSRII Benchtop cytometer (was used, and data were analyzed with BD Diva software (version 2.1; BD Biosciences) and FlowJo software (version 10.1; Tree Star, Inc., Ashland, OR, USA). Statistical Analysis One-way analysis of variance (ANOVA) with Tukey s post hoc testing was used for statistical comparisons parameters within strains. Two-way ANOVA with Tukey s post hoc testing was used for statistical comparison between strains. P 0.05 was considered statistically significant. These tests were performed using GraphPad Prism 6.0 software (GraphPad Software, Inc., San Diego, CA, USA). RESULTS Ocular Surface Disease The uptake of OGD as a marker of corneal barrier function disruption was measured in Aec and B6 mice. Wild-type B6 mice had increased uptake of OGD with aging, significantly highest at 20 weeks. Aec mice had slightly, but not significantly greater OGD intensity compared with B6 mice at 4 and 12 weeks of age. However, there was no change in OGD staining with aging in Aec mice (Figs. 1A, 1B). In order to determine if CD4 þ T cells migrate to the ocular surface in Aec mice, the density of CD4 þ T cells in the conjunctival epithelium was determined by immunohistochemical staining. We chose to count infiltrating CD4 þ T cells within the conjunctival epithelium at the goblet cell rich area because number of CD4 þ T cells in this area was previously found to inversely correlated with number of goblet cells. 13 As expected, we noted a significant increase in CD4 þ T-cell infiltration in conjunctival epithelium in Aec mice compared to B6 mice at 20 weeks ( vs cells, P ¼ 0.01; Figs. 1C, 1D). There was a tendency toward increasing numbers of CD4 þ T cells infiltrating the conjunctiva with aging in both Aec and B6 mice. Filled goblet cells were identified in paraffin-embedded conjunctival sections stained with PAS. Consistent with disease progression, a significant decrease in the density of goblet cell in conjunctiva (cells per millimeter) was observed at 12 weeks and persisted at 20 weeks in both mouse strains (Figs. 1E, 1F). Goblet cell density was significantly lower in Aec mice compared to B6 mice at 12 weeks. Cytokine Profile With Aging in Ocular Surface of Aec and B6 Mice We examined expression of inflammatory cytokine and MMPs and T-cell related cytokines at the ocular surface by real-time PCR. Our results are summarized in Figure 2. We observed that Aec mice had significantly higher levels of IL-17A, IL-17R, IL-1a, and IL-1b mrna transcripts (at 4 weeks), and significantly lower levels of IFN-c, MHC II, IL-17R, and MMP-9 (at 12 and 20 weeks), and IL-17A and IL-1b mrna transcripts (at 20 weeks) in their cornea than did similarly aged B6 mice (Figs. 2A, 2B). No significant change was found in levels of IL-6, IL-13, and MMP-3 transcripts in the cornea in the two strains (data not shown). There was a significant agerelated increase in expression of IL-1a and TNF-a in Aec cornea with aging from 4 to 20 weeks. Aec mice had significantly higher levels of IL-17A and IL-1a (at 4, 12, and 20 weeks), IL-1b and IL-17R (at 12 and 20 weeks), IL-6 (at 4 weeks), and TNF-a mrna transcripts (at 12 weeks), and significantly lower levels of MHC II and SPRR2a mrna transcripts (at 4, 12, and 20 weeks) in the conjunctiva than did
4 C56BL/6.NOD-Aec1Aec2 Mice IOVS j April 2015 j Vol. 56 j No. 4 j 2227 FIGURE 1. Ocular surface phenotype in Aec mice. (A) Corneal OGD fluorescence intensity score. Bar graphs show mean 6 SEM of three independent experiments with four animals per experiment (8 eyes per experiment yielding a final sample of 24 eyes per age per strain). (B) Representative images of OGD staining of each strain. Original magnification: 310; scale bar: 100 lm. (C) CD4 þ T cells infiltrating the conjunctival epithelium. Bar graphs show mean 6 SEM of one representative experiment with three animals per age and strain. (D) Representative images of conjunctiva sections immunostained for CD4 (in red). Original magnification: 340; scale bar: 25lm. (E) Number of PAS þ conjunctival goblet cells counted in paraffin-embedded sections expressed as number per millimeter. Bar graphs show mean 6 SEM of one representative experiment with three animals per age and strain. (F) Representative images of conjunctival sections stained PAS used to generate the bar graph in (E). Original magnification: 340; scale bar: 25lm. W indicates weeks. **P < 0.01, ****P < for within-b6 strain comparison compared to age 4 weeks. ^^^^P < for within-aec strain comparison compared to age 4 weeks. P < 0.05 for age 12 vs. 20 weeks intrastrain comparison.
5 C56BL/6.NOD-Aec1Aec2 Mice IOVS j April 2015 j Vol. 56 j No. 4 j 2228 B6 mice (Figs. 2C, 2D). There was a significant age-related increase in expression of IL-17A, IL-1b, and TNF-a in Aec conjunctiva from 4 to 20 weeks. The IFN-c significantly increased with aging in Aec conjunctiva. FIGURE 2. Quantitative gene analysis of mrna transcripts in aged cornea and conjunctiva. (A, B) Relative fold of expression of T-cell related cytokines and inflammatory cytokines in cornea of Aec and B6 mice at ages 4, 12, and 20 weeks. Bar graphs show mean 6 SEM of one representative experiment with six to nine samples per strain per age. (Experiment was repeated twice with similar results.) One sample consisted of pooled right and left corneas of the same animal. (C, D) Relative fold of expression of T-cell related cytokines and inflammatory cytokines in conjunctiva of Aec and B6 mice at ages 4, 12, and 20 weeks. Bar graphs show mean 6 SEM of one representative experiment with six to nine samples per strain per age. (Experiment was repeated twice with similar results.) One sample consisted of pooled right and left conjunctivas of the same animal. *P < 0.05, **P < 0.01, ***P < for within-b6 strain comparison compared to age 4 weeks. ^P < 0.05, ^^P < 0.01, ^^^P < for within-aec strain comparison compared to age 4 weeks. P < 0.05, P < 0.01, P < for age 12 vs. 20 weeks intrastrain comparison. Evaluation of LG Function It has been shown previously that Aec mice exhibited a reduced salivary gland secretory response at 12 weeks of age. 7 However, secretory function of LG in Aec mice has not been evaluated. To determine the secretory activity of LG, tear volume was measured by phenol red cotton-thread technique. We observed that tear volume in the Aec mice was significantly greater at 20 weeks than 4 and 12 weeks (Fig. 3A). The B6 mice also exhibited an increase in tear volume with age, reaching statistical significance by 20 weeks. No difference was found between strains at any of the three age groups. Epidermal growth factor is a multifunctional cytokine that regulates proliferation, differentiation, and survival of epithelial cells. 14 EGF in tears is generally accepted to be secreted from the LGs, and tear EGF concentration is significantly lower in murine models of Sjögren syndrome. 10 We assayed EGF in tears of all mice of both strains at all three time points (Fig. 3B). We observed that Aec mice of 4 and 20 weeks had slightly lower EGF concentration in tear washings compared with same-age B6 mice. But, 12-week-old Aec mice had significantly higher EGF concentration compared with B6 mice of the same age. Immunoglobulin A is commonly found in high concentration in normal human and rodent tears, while very low levels of IgG and IgM are normally found and are thought to be present secondary to transudation from plasma and/or inflammatory processes Immunoglobulin A is produced locally within the LG, and IgA content in tears increases with age. 18 Because both strains had increased tear volume at 20 weeks compared to 4 weeks, we assayed the tears from Aec and B6 at these time points to better evaluate the quality of tears using a multiplex assay. No difference at IgA, IgG1, and IgM levels were noted between Aec and B6 mice at 4 weeks of age (Figs. 3C E). Significantly increased IgA levels were found in B6 compared to Aec (B6 > Aec; P < 0.01; Fig. 3C) and increased IgG and IgM levels were seen in Aec at 20 weeks (Aec > B6; P < 0.05 for both immunoglobulins; Figs. 3D, 3E). The ratio of IgA to IgG and IgA to IgM in B6 mice increased with aging from 4 to 20 weeks of age, indicating a greater production of IgA and low levels of IgG and IgM, while in Aec mice they did not change (Figs. 3F, 3G). Taken together, these results indicate the composition of tears is altered in Aec at 20 weeks of age, and increased tear volume may be indicative of transudation of plasma from an inflamed conjunctiva. Periductal lymphocytic infiltration has been noted in murine models of SS and the aged human LG We compared the LG histology of Aec mice at 4, 12, and 20 weeks of age with age-matched B6 mice by hematoxylin and eosin staining. The B6 mice LGs had minimal lymphocytic infiltration at all time points (Fig. 3H). Aec mice LGs showed more prominent periductal lymphocytic infiltration at all ages, and was greatest at 20 weeks of age (Fig. 3H). To better characterize the lymphocytic infiltration, LG sections from both strains were immunostained for CD4 þ, CD8 þ T cells and B cells and also by flow cytometry. Aec mice showed increased CD4 þ T-cell infiltration of the LG with aging that became especially intense at 20 weeks of age (Fig. 3I). Minimal infiltration with CD8 þ T cells and B cells (data not shown) was seen in Aec and B6 LGs. Flow cytometric analysis of freshly isolated cells from LGs of all age groups and both strains was used to quantify the immune cell subsets that infiltrated the LG, including CD4, CD8, and B cells. We observed that CD4 þ T
6 C56BL/6.NOD-Aec1Aec2 Mice IOVS j April 2015 j Vol. 56 j No. 4 j 2229 FIGURE 3. Lacrimal gland evaluation in Aec and B6 mice. (A, B) Tear volume (A) was measured by phenol red thread test, and tear EGF concentrations (B) were measured by ELISA. Tear washings from both right and left eyes from one mouse per age and strain were collected and pooled into a single tube yielding a final sample of 10 individual samples per group and age divided into two independent experiments with five samples per experiment. Bar graphs show mean 6 SEM. (C E) Tear immunoglobulin concentrations were measured by Milliplex map assay. One sample consisted of tear washings pooled from both eyes of 10 mice (20 ll) in PBS plus 0.1% bovine serum albumin (5 ll). Four samples from Aec
7 C56BL/6.NOD-Aec1Aec2 Mice IOVS j April 2015 j Vol. 56 j No. 4 j 2230 and B6 at 4 and 20 weeks of age were used. Bar graphs show mean 6 SEM. (F, G) Tear IgA-to-IgG ratio and tear IgA-to-IgM ratio. (H) Representative images of hematoxylin and eosin stained LG sections from mice aged 4 to 20 weeks. Normal LG morphology was noted in the B6 mice at all time points, in contrast to Aec LG, which showed lymphocytic infiltration (circumscribed by black dotted lines) at 20 weeks. Original magnification: 310; scale bar: 100 lm. (I) Representative images of LG frozen sections immunostained for CD4 (in red,[b]). Original magnification: 310; scale bar: 100 lm. (J) Flow cytometric analysis of CD4 þ and CD8 þ cells in LG of Aec and B6 mice at ages 4, 12, and 20 weeks. Right and left extraorbital LGs from one mouse per age and strain were excised and pooled into a single tube yielding a final sample of six individual LG samples per group and age divided into two dependent experiments with three samples per experiment. Bar graphs show mean 6 SEM. (K) A representative dot plot of the percentage of CD4 þ cells that are CCR6 þ (for Th17) or CXCR3 þ (for Th1) in the cervical lymph node (CLN) and LG. (L) A representative dot plot of the percentage of CD25 þ cells that are Foxp3 þ (for Treg) in the LG. *P < 0.05, **P < 0.01 for within-b6 strain comparison compared to age 4 weeks. ^P < 0.05, ^^P < 0.01 for within-aec strain comparison compared to age 4 weeks. P < 0.05 for age 12 vs. 20 weeks intrastrain comparison. cells significantly increased in Aec LG compared to B6 at 12 weeks ( % vs %, P < 0.01) and 20 weeks ( % vs %, P < 0.01) and was noted to increase with aging from ages 4 to 20 weeks in Aec mice (Fig. 3J). There was no significant difference in CD8 þ T cells in Aec mice LG compared to B6 mice, but a progressive increase in CD8 þ T cells with aging was noted in both strains, in roughly equal proportions with aging (Fig. 3J). To better characterize the Th phenotype of infiltrating CD4 þ T cells, expression of chemokine receptors and FOXP3 were evaluated by flow cytometry (Figs. 3K, 3L). CD4 þ CXCR3 þ and CD4 þ CCR6 þ cells identify Th1 and TH17 cells, while CD4 þ CD25 þ FOXP3 þ cells are considered regulatory T cells. 23,24 At 4 and 20 weeks of age, Aec mice had lower frequency of CD4 þ CXCR3 þ cells than B6 of the same age, but the frequency of CD4 þ CCR6 þ cells was higher. Aec mice had a 2.6-fold increase in the frequencies of both CD4 þ CXCR3 þ and CD4 þ CCR6 þ, while B6 mice had a minimal increase in Th1 cells and approximately a 6-fold increase in TH17 cells with aging from 4 to 20 weeks (Fig. 3K). Analysis of CD4 þ CD25 þ FOXP3 þ cells indicated that B6 mice have higher number of Tregs at 4 weeks (~4-fold), and it remained elevated at 20 weeks of age, where the number was over 2- fold greater than Aec (Fig. 3L). Taken together, these results show that Aec mice have increased LG lymphocytic infiltration with aging that consists mainly of CD4 þ T cells with predominance of TH17 over Th1 cells. Although there is also an increase of Tregs within the LG, their frequency is still half that of wild-type B6 mice. Inflammatory Cytokine Milieu in Aec LG We examined expression of inflammatory and T-cell related cytokines and T-cell transcription factors in the LG by qpcr (Figs. 4A, 4B). Aec mice had significantly higher levels of IFN-c, IL-17A, IL-1b, IL-6, T-BET and RORcT, but lower levels of IL-13 and GATA-3 mrna transcripts in the LG than B6 mice at 4 weeks of age (Figs. 4A, 4B). Levels of IL-17A, IL-17R, IFN-c, IL- 1b, IL-6, and TNF-a mrna transcripts tended to increase in the LG with aging in B6 mice, with a greater increase in IL-17 (and IL-17R) than IFN-c, mirroring the pattern of chemokine receptor expression seen by flow cytometry. Differential changes in some markers were observed with aging in Aec: IFN-c, T-bet, RORcT, GATA-3, and IL-6 mrna decreased, reaching statistical significance at 20 weeks for IFN-c and IL-6 compared to levels at 4 weeks. A significant increase in the levels of IL-1b, IL-17R, and TNF-a transcripts was observed in Aec mice at 12 weeks, with a subsequent decrease at 20 weeks. The levels of T-BET and GATA-3 decreased with aging in Aec mice. These results indicate that at a very early age, Aec mice have significantly increased IFN-c, IL-1 b, IL-6, IL-17A, and very low levels of IL-13 and GATA-3 compared to B6 mice. While there is a decrease in IFN-c with aging, the increase in IL- 17A does not parallel that seen in B6 mice. The inflammatory cytokine IL-1b is significantly elevated at 12 weeks compared to B6 control mice (P < 0.001). DISCUSSION Aec mice, a congenic strain of the NOD model of autoimmune exocrinopathy, develop dry eye spontaneously with aging. 2 The course of disease and characteristics of dry eye in Aec mice is similar to those of human dry eye. 2,7 A newly derived strain, known as C57BL/6.NOD-Aec1R1Aec2, has recombination in Aec1 that results in shortening of this genetic region to 19.2 cm from 48.5 cm. 9 This recombinant inbred strain has revealed that male mice maintain a full SS-like disease, whereas female mice exhibited no dacryoadenitis and no loss of LG FIGURE 4. Quantitative gene analysis of mrna transcripts in aged LG. (A, B) Relative fold of expression of T-cell related cytokines and inflammatory cytokines in LG of Aec and B6 mice at ages 4, 12, and 20 weeks. Bar graphs show mean 6 SEM expression of one representative experiment with six to nine samples per strain and age. One sample consisted of pooled right and left extraorbital LGs of the same animal. ***P < 0.001, ****P < for within-b6 strain comparison compared to age 4 weeks. ^^P < 0.01, ^^^P < 0.001, ^^^^P < for within-aec strain comparison compared to age 4 weeks. P < 0.05, P < 0.001, P < for age 12 vs. 20 weeks intrastrain comparison.
8 C56BL/6.NOD-Aec1Aec2 Mice IOVS j April 2015 j Vol. 56 j No. 4 j 2231 secretory function. 9 In the present study, we have examined parameters of the ocular surface disease in Aec female mice with aging and their correlation with development of LG disease. Both humans and mice have been noted to develop dry eye disease with aging. The B6 strain has a predilection to a TH1 inflammatory response. Age-related autoimmunity of B6 mice contributes to development of dry eye disease with aging. McClellan et al. 14 investigated the immunocytologic profile of age-related dry eye disease using three different ages of B6 mice: young (8 weeks), midlife (6 9 months), and elderly (24 months). Compared to B6, Aec mice exhibit several features found in human SS, including more severe conjunctival goblet cell loss and LG lymphocytic infiltration with aging. Age-related differences in proinflammatory and T-cell gene expression profile in the cornea and conjunctiva epithelium between the two strains were also observed. We demonstrated that like SS, the ocular surface of Aec mice developed conjunctival goblet cell loss with aging, parallel with LG lymphocytic infiltration. We also observed age-related changes in proinflammatory and T-cell gene expression profile in the cornea and conjunctiva epithelium. These findings were equal to or greater than those observed in B6 mice, demonstrating that Aec mice are more prone to developing dacryoadenitis and conjunctival goblet cell loss. Decreased density of mucin-filled goblet cells is a wellrecognized feature of aqueous deficient dry eye. 25,26 Goblet cell density has been found to correlate with severity of ocular surface disease in patients with aqueous tear deficiency. 27 Previously, we have found that the recipients of adoptively transferred CD4 þ T cells from aged B6 donors developed conjunctival inflammation with CD4 þ T-cell infiltration, increased IFN-c expression, and decreased goblet cell density. 14 In our study, older Aec and B6 mice showed an increased number of CD4 þ T cells infiltrating the conjunctival epithelium, and this T-cell infiltration was accompanied by conjunctival goblet cell loss. The reduced goblet cell density may have been due to increased IFN-c expression since both strains of mice showed increased expression of IFN-c in the conjunctiva with aging, but this was significant only in the Aec strain. The strong correlation between age and prevalence of dry eye disease has been well established. 28 In elderly people, changes in tear parameters, such as tear flow, tear meniscus height, and tear volume, are often found In several mouse strains, changes in tear volume (increase, decrease, or no change) have been noted, depending on the genetic background In particular, tear volume increased in several mouse models of dry eye, such as CD25KO, SPDEF KO, aging C57BL/6. 5,34,35 In a recent report, Tan et al. 38 reported that Schirmer I test values of Aec mice showed a significant reduction in comparison with those of the B6 control mice at 16 and 20 weeks of age. However, in our study there was a discrepancy between inflammation of LG and tear production. Tear volume in Aec mice paradoxically increased with aging similar to that observed in B6 mice. Lymphocyte infiltration of LG was more prominent in older Aec mice. Tear volume is a composite of fluid secreted by the LG and corneal and conjunctival epithelium, inclusive of IgA, lysozyme, growth factors, and antimicrobial peptides. It is possible that the increase of tear volume in Aec and B6 mice reflects the normal increase in body size and weight of mice. We observed significant increase in IgA in tears in B6 mice with aging without any changes in Aec, despite increase in size of both strains. We also observed an increase in IgG and IgM in 20- week-old Aec mice, suggesting that quality of tears in aged Aec is altered compared to B6 mice. These results also suggest increased transudation of plasma proteins from inflamed tissues, as IgG and IgM are not very abundantly found in tears from normal subjects. 17,18 Although decreases in tear volume are normally associated with both dry eye disease and aging, increases in LG secretion have been reported to occur in response to ocular surface irritation. 39 It is possible that the observed increase in tear volume in the Aec strain could be due to reflex tearing or due to transudation from the conjunctiva. Moreover, our findings suggest that increases of inflammatory mediators with aging make a greater contribution to development of ocular surface disease, mainly conjunctiva, than inflammation in the LG. Traditionally, keratoconjunctivitis sicca was attributed to tear deficiency from LG disease; however, it is not uncommon for SS patients to have clinically significant ocular surface disease in the presence of normal tear production and volume. 40 Our study provides additional evidence suggesting that LG and corneal/conjunctival inflammation are important factors in the pathogenesis of ocular surface epithelial disease and ocular discomfort in SS, even in the context of normal tear production or volume. We had previously reported that neutralization of IL-17A ameliorates corneal barrier disruption in an experimental model of dry eye. 41 IL-17A has been shown to cause barrier disruption in mice and increased expression of MMPs, which leads to increased desquamation and breakdown of tight junctions of the corneal epithelium. 41 Recently, elevated levels of IL-17A protein were found in minor salivary glands of SS patients compared with normal control subjects. 42 Interestingly, in our study, increased expression of IL-17A mrna transcripts and increased OGD intensity score as markers of corneal barrier disruption were significantly higher in older B6 mice, whereas decreased expression of both IL-17A and MMP-9 mrna transcripts and decreased OGD intensity score with aging was found in the cornea of Aec mice. In our study, there was no change in OGD staining with aging in Aec mice. In another mouse dry eye model study (Spdef / mice), the increase in fluorescein staining with age in wild-type Spdef þ/þ mice was not observed in Spdef / mice. 34 A similar pattern was observed in the autoimmune CD25KO model where corneal barrier dysfunction was only observed at 12 weeks of age despite severe dacryoadenitis at 16 weeks. 22 The proinflammatory effects of IL-1 in the LG have been extensively studied by Zoukhri and colleagues They showed that a single injection of IL-1 into the LG induced a mild decrease in LG secretion while inducing a robust, reversible inflammatory response that led to destruction of LG acinar epithelial cells. 44 Pelegrino et al. 10 reported that the peak of IL-1b (age 12 weeks) preceded the greatest increase in lymphocytic infiltration (age 16 weeks) of LG. Thus, IL-1b may be critical in inducing inflammatory dysfunction in the LG. In our study, IL-1b and TNF-a transcript levels peak in Aec LGs at age 12 weeks, while levels of IFN-c, IL-1b, and IL-6 were higher at 4 and 12 weeks of age in Aec compared to B6 mice. Our results showed the LG in both strains is infiltrated by TH1 and TH17 cells; Aec mice show a greater increase in the percentage of Th1 cells, whereas B6 mice show a greater increase in Th17 cells with aging. This was accompanied by an increase of regulatory T cells in both strains, although the B6 mice had greater frequency of Tregs both at baseline and at 20 weeks. Nguyen et al. 46 identified the differential expression of transcription factors for CD4 þ Th1, Th2, and Th17 cells in the submandibular glands of Aec mice. These included T-bet for Th1 cells, GATA-3 for Th2 cells, and RORct for Th17 cells. 46 T- bet expression was up-regulated nearly 3.5-fold in glandular tissue from 4-week-old Aec mice, but returned to normal levels by age 8 weeks. In contrast, expression of both Gata3 and RORct did not increase until after age 12 weeks, peaked at age 16 weeks, and returned to normal by age 20 weeks. 46 These data are highly indicative of the disease profile described for Aec mice, in which IFN-c expression is essential early in the
9 C56BL/6.NOD-Aec1Aec2 Mice IOVS j April 2015 j Vol. 56 j No. 4 j 2232 disease. 46 In our study, T-BET and RORC of LG were significantly increased in Aec mice compared with B6, while GATA-3 was decreased. Although SPRR2 expression increased with age in Aec, the increase was less than that observed in B6 mice. The cause for the discrepancy between strains is not clear. In contrast, Aec mice developed significant conjunctival goblet cell loss with aging that paralleled the CD4 þ T-cell infiltration in the conjunctiva and LG. Additionally, expression of the Th1 cytokine IFN-c was increased in the conjunctiva and LG and was significantly higher in the Aec strain. Therefore, Aec appears to be a relevant model for the LG inflammation and conjunctival goblet cell loss that occurs in SS. Interestingly, ocular surface disease in the Aec mice was not accompanied by decreased tear volume but rather by altered composition, suggesting that it may be related to inflammation in those tissues. Acknowledgments Supported by National Institutes of Health (NIH; Bethesda, MD, USA) Grant EY11915 (SCP), National Eye Institute/NIH (Bethesda, MD, USA) Core Grant EY RPB, Oshman Foundation (Houston, TX, USA), William Stamps Farish Fund (Houston, TX, USA), Hamill Foundation (Houston, TX, USA), and by the Cytometry and Cell Sorting Core at Baylor College of Medicine (Houston, TX, USA), which is funded by the NIH/National Institute of Allergy and Infectious Diseases Grant P30AI036211, National Cancer Institute Grant P30CA125123, and National Center for Research Resources Grant S10RR (all located in Bethesda, MD, USA). This paper was supported by research funds of Chonbuk National University, Jeonju, Jeonbuk, Korea, in The authors thank the Mahira Zaheer and Kevin Tesareski for their technical assistance with these experiments. The authors alone are responsible for the content and writing of the paper. Disclosure: I.-C. You, None; F. Bian, None; E.A. Volpe, None; C.S. de Paiva, None; S.C. Pflugfelder, None References 1. Jonsson R, Dowman SJ, Gordon TP. Sjögren Syndrome. Arthritis and Allied Conditions. 15th ed. Philadelphia, PA: Lippincott Williams & Wilkins; 2005: Nguyen CQ, Peck AB. Unravelling the pathophysiology of Sjogren syndrome-associated dry eye disease. Ocul Surf. 2009; 7: Cha S, Peck AB, Humphreys-Beher MG. Progress in understanding autoimmune exocrinopathy using the non-obese diabetic mouse: an update. Crit Rev Oral Biol Med. 2002;13: Jabs DA, Prendergast RA, Campbell AL, et al. Autoimmune Th2-mediated dacryoadenitis in MRL/MpJ mice becomes Th1- mediated in IL-4 deficient MRL/MpJ mice. Invest Ophthalmol Vis Sci. 2007;48: Rahimy E, Pitcher JD III, Pangelinan SB, et al. Spontaneous autoimmune dacryoadenitis in aged CD25KO mice. Am J Pathol. 2010;177: Bryer J, Lowry J, Cha S, et al. Alleles from chromosome 1 and 3 NOD mice combine to influence Sjögren s syndrome-like autoimmune exocrinopathy. J Rheumatol. 2000;27: Cha S, Nagashima H, Brown VB, Peck AB, Humphreys-Beher MG, Two NOD Idd-associated intervals contribute synergistically to the development of autoimmune exocrinopathy (Sjögren s syndrome) on a healthy murine background. Arthritis Rheum. 2002;46: Nguyen CQ, Sharma A, She JX, McIndoe RA, Peck AB. Differential gene expressions in the lacrimal gland during development and onset of keratoconjunctivitis sicca in Sjögren s syndrome (SJS)-like disease of the C57BL/6.NOD- Aec1Aec2 mouse. Exp Eye Res. 2009;88: Nguyen C, Singson E, Kim JY, et al. Sjögren s syndrome-like disease of C57BL/6.NOD-Aec1Aec2 mice: gender differences in keratoconjunctivitis sicca defined by a cross-over in the chromosome 3 Aec1 locus. Scand J Immunol. 2006;64: Pelegrino FS, Volpe EA, Gandhi NB, Li DQ, Pflugfelder SC, de Paiva CS. Deletion of interferon-c delays onset and severity of dacryoadenitis in CD25KO mice. Arthritis Res Ther. 2012;14: R Stewart P, Chen Z, Farley W, Olmos L, Pflugfelder SC. Effect of experimental dry eye on tear sodium concentration in the mouse. Eye Contact Lens. 2005;31: de Paiva CS, Volpe EA, Gandhi NB, et al. Disruption of TGF-b signaling improves ocular surface epithelial disease in experimental autoimmune keratoconjunctivitis sicca. PLoS One. 2011;6:e de Paiva CS, Villarreal AL, Corrales RM, et al. Dry eye-induced conjunctival epithelial squamous metaplasia is modulated by interferon-gamma. Invest Ophthalmol Vis Sci. 2007;48: McClellan AJ, Volpe EA, Zhang X, et al. Ocular surface disease and dacryoadenitis in aging C57BL/6 mice. Am J Pathol. 2014; 184: Bluestone R, Easty DL, Goldberg LS, Jones BR, Pettit TH. Lacrimal immunoglobulins and complement quantified by counter-immunoelectrophoresis. Br J Ophthalmol. 1975;59: Sen DK, Sarin GS. Immunoglobulin concentrations in human tears in ocular diseases. Br J Ophthalmol. 1979;63: Zavaro A, Samra Z, Baryishak R, Sompolinsky D. Proteins in tears from healthy and diseased eyes. Doc Ophthalmol. 1980; 50: Sen DK, Sarin GS, Mathur GP, Saha K. Variation of immunoglobulin concentrations in normal human tears related to age and sex. Acta Ophthalmol (Copenh). 1978;56: Nasu M, Matsubara O, Yamamoto H. Post-mortem prevalence of lymphocytic infiltration of the lacrymal gland: a comparative study in autoimmune and non-autoimmune diseases. J Pathol. 1984;143: Obata H, Yamamoto S, Horiuchi H, Machinami R. Histopathologic study of human lacrimal gland. Statistical analysis with special reference to aging. Ophthalmology. 1995;102: Rios JD, Horikawa Y, Chen LL, et al. Age-dependent alterations in mouse exorbital lacrimal gland structure, innervation and secretory response. Exp Eye Res. 2005;80: de Paiva CS, Hwang CS, Pitcher JD III, et al. Age-related T-cell cytokine profile parallels corneal disease severity in Sjogren s syndrome-like keratoconjuctivitis sicca in CD25KO mice. Rheumatology. 2010;49: Coursey TG, Gandhi NB, Volpe EA, Pflugfelder SC, de Paiva CS. Chemokine receptors CCR6 and CXCR3 are necessary for CD4þ T cell mediated ocular surface disease in experimental dry eye disease. PLoS One. 2013;8:e Chatenoud L. Natural and induced T CD4þCD25þFOXP3þ regulatory T cells. Methods Mol Biol. 2011;677: Stern ME, Beuerman RW, Fox RI, Gao J, Mircheff AK, Pflugfelder SC. The pathology of dry eye: the interaction between the ocular surface and lacrimal glands. Cornea. 1998; 17: Dursun D, Wang M, Monroy D, et al. A mouse model of keratoconjunctivitis sicca. Invest Ophthalmol Vis Sci. 2002; 43:
10 C56BL/6.NOD-Aec1Aec2 Mice IOVS j April 2015 j Vol. 56 j No. 4 j Pflugfelder SC, Tseng SC, Yoshino K, Monroy D, Felix C, Reis BL. Correlation of goblet cell density and mucosal epithelial membrane mucin expression with rose bengal staining in patients with ocular irritation. Ophthalmology. 1997;104: Moss SE, Klein R, Klein BE. Incidence of dry eye in an older population. Arch Ophthalmol. 2004;122: Furukawa RE, Polse KA. Changes in tear flow accompanying aging. Am J Optom Physiol Opt. 1978;55: Hamano T, Mitsunaga S, Kotani S, et al. Tear volume in relation to contact lens wear and age. CLAO J. 1990;16: Mathers WD, Lane JA, Zimmerman MB. Tear film changes associated with normal aging. Cornea. 1996;15: Patel S, Wallace I. Tear meniscus height, lower punctum lacrimale, and the tear lipid layer in normal aging. Optom Vis Sci. 2006;83: Cui L, Shen M, Wang J, et al. Age-related changes in tear menisci imaged by optical coherence tomography. Optom Vis Sci. 2011;88: Marko CK, Menon BB, Chen G, Whitsett JA, Clevers H, Gipson IK. Spdef null mice lack conjunctival goblet cells and provide a model of dry eye. Am J Pathol. 2013;183: McClellan AJ, Volpe EA, Zhang X, et al. Ocular surface disease and dacryoadenitis in aging C57BL/6 mice. Am J Pathol. 2014; 184: Kojima T, Wakamatsu TH, Dogru M, et al. Age-related dysfunction of the lacrimal gland and oxidative stress: evidence from the Cu, Zn-superoxide dismutase-1 (Sod1) knockout mice. Am J Pathol. 2012;180: Módulo CM, Machado Filho EB, Malki LT, et al. The role of dyslipidemia on ocular surface, lacrimal and meibomian gland structure and function. Curr Eye Res. 2012;37: Tan L, Xie HP. Evaluation of dry eye syndrome in C57BL/ 6.NOD-Aec1Aec2 mice. Chin J Exp Ophthalmol. 2013;31: Rolando M, Zierhut M. The ocular surface and tear film and their dysfunction in dry eye disease. Surv Ophthalmol. 2001; 45:S203 S Pflugfelder SC. What causes dryness in Sjögren s syndrome patients and how can it be targeted? Expert Rev Clin Immunol. 2014;10: de Paiva CS, Chotikavanich S, Pangelinan SB, et al. IL-17 disrupts corneal barrier following desiccating stress. Mucosal Immunol. 2009;2: Katsifis GE, Rekka S, Moutsopoulos NM, Pillemer S, Wahl SM. Systemic and local interleukin-17 and linked cytokines associated with Sjögren s syndrome immunopathogenesis. Am J Pathol. 2009;175: Zoukhri D, Hodges RR, Byon D, Kublin CL. Role of proinflammatory cytokines in the impaired lacrimation associated with autoimmune xerophthalmia. Invest Ophthalmol Vis Sci. 2002;43: Zoukhri D, Macari E, Kublin CL. A single injection of interleukin-1 induces reversible aqueous-tear deficiency, lacrimal gland inflammation, and acinar and ductal cell proliferation. Exp Eye Res. 2007;84: Zoukhri D, Fix A, Alroy J, Kublin CL. Mechanisms of murine lacrimal gland repair after experimentally induced inflammation. Invest Ophthalmol Vis Sci. 2008;49: Nguyen CQ, Hu MH, Li Y, Stewart C, Peck AB. Salivary gland tissue expression of interleukin-23 and interleukin-17 in Sjögren s syndrome: findings in humans and mice. Arthritis Rheum. 2008;58:
Role of Th17 cells in the immunopathogenesis of dry eye disease
 Role of Th17 cells in the immunopathogenesis of dry eye disease The Harvard community has made this article openly available. Please share how this access benefits you. Your story matters Citation Chauhan,
Role of Th17 cells in the immunopathogenesis of dry eye disease The Harvard community has made this article openly available. Please share how this access benefits you. Your story matters Citation Chauhan,
Supplemental Information. T Cells Enhance Autoimmunity by Restraining Regulatory T Cell Responses via an Interleukin-23-Dependent Mechanism
 Immunity, Volume 33 Supplemental Information T Cells Enhance Autoimmunity by Restraining Regulatory T Cell Responses via an Interleukin-23-Dependent Mechanism Franziska Petermann, Veit Rothhammer, Malte
Immunity, Volume 33 Supplemental Information T Cells Enhance Autoimmunity by Restraining Regulatory T Cell Responses via an Interleukin-23-Dependent Mechanism Franziska Petermann, Veit Rothhammer, Malte
The toll-like receptor 4 ligands Mrp8 and Mrp14 play a critical role in the development of autoreactive CD8 + T cells
 1 SUPPLEMENTARY INFORMATION The toll-like receptor 4 ligands Mrp8 and Mrp14 play a critical role in the development of autoreactive CD8 + T cells Karin Loser 1,2,6, Thomas Vogl 2,3, Maik Voskort 1, Aloys
1 SUPPLEMENTARY INFORMATION The toll-like receptor 4 ligands Mrp8 and Mrp14 play a critical role in the development of autoreactive CD8 + T cells Karin Loser 1,2,6, Thomas Vogl 2,3, Maik Voskort 1, Aloys
Blocking antibodies and peptides. Rat anti-mouse PD-1 (29F.1A12, rat IgG2a, k), PD-
 Supplementary Methods Blocking antibodies and peptides. Rat anti-mouse PD-1 (29F.1A12, rat IgG2a, k), PD- L1 (10F.9G2, rat IgG2b, k), and PD-L2 (3.2, mouse IgG1) have been described (24). Anti-CTLA-4 (clone
Supplementary Methods Blocking antibodies and peptides. Rat anti-mouse PD-1 (29F.1A12, rat IgG2a, k), PD- L1 (10F.9G2, rat IgG2b, k), and PD-L2 (3.2, mouse IgG1) have been described (24). Anti-CTLA-4 (clone
Supporting Information
 Supporting Information Desnues et al. 10.1073/pnas.1314121111 SI Materials and Methods Mice. Toll-like receptor (TLR)8 / and TLR9 / mice were generated as described previously (1, 2). TLR9 / mice were
Supporting Information Desnues et al. 10.1073/pnas.1314121111 SI Materials and Methods Mice. Toll-like receptor (TLR)8 / and TLR9 / mice were generated as described previously (1, 2). TLR9 / mice were
Identifying Sjögren's Syndrome. These include cholinergic blockade from autoantibodies to muscarinic acetylcholine
 Identifying Sjögren's Syndrome Melissa Barnett, OD, FAAO Melissa Barnett, OD, FAAO is a Principal Optometrist at the UC Davis Medical Center in Sacramento. She specializes in anterior segment disease and
Identifying Sjögren's Syndrome Melissa Barnett, OD, FAAO Melissa Barnett, OD, FAAO is a Principal Optometrist at the UC Davis Medical Center in Sacramento. She specializes in anterior segment disease and
1998 DESCRIPTION Evaluation of Subjective and Objective tests for diagnosing tear-film disorders known to cause ocular irritation.
 DEWS DRY EYE: DIAGNOSTIC TEST TEMPLATE RAPPORTEUR A.J.Bron 18 th Oct 2004 TEST Mixed tests TO Ocular Irritation / Dry Eye REFERENCES DIAGNOSE VERSION of TEST Multiple tests DESCRIPTION Evaluation of Subjective
DEWS DRY EYE: DIAGNOSTIC TEST TEMPLATE RAPPORTEUR A.J.Bron 18 th Oct 2004 TEST Mixed tests TO Ocular Irritation / Dry Eye REFERENCES DIAGNOSE VERSION of TEST Multiple tests DESCRIPTION Evaluation of Subjective
Supplementary Materials for
 immunology.sciencemag.org/cgi/content/full/2/16/eaan6049/dc1 Supplementary Materials for Enzymatic synthesis of core 2 O-glycans governs the tissue-trafficking potential of memory CD8 + T cells Jossef
immunology.sciencemag.org/cgi/content/full/2/16/eaan6049/dc1 Supplementary Materials for Enzymatic synthesis of core 2 O-glycans governs the tissue-trafficking potential of memory CD8 + T cells Jossef
SUPPLEMENTARY METHODS
 SUPPLEMENTARY METHODS Histological analysis. Colonic tissues were collected from 5 parts of the middle colon on day 7 after the start of DSS treatment, and then were cut into segments, fixed with 4% paraformaldehyde,
SUPPLEMENTARY METHODS Histological analysis. Colonic tissues were collected from 5 parts of the middle colon on day 7 after the start of DSS treatment, and then were cut into segments, fixed with 4% paraformaldehyde,
Cell isolation. Spleen and lymph nodes (axillary, inguinal) were removed from mice
 Supplementary Methods: Cell isolation. Spleen and lymph nodes (axillary, inguinal) were removed from mice and gently meshed in DMEM containing 10% FBS to prepare for single cell suspensions. CD4 + CD25
Supplementary Methods: Cell isolation. Spleen and lymph nodes (axillary, inguinal) were removed from mice and gently meshed in DMEM containing 10% FBS to prepare for single cell suspensions. CD4 + CD25
and follicular helper T cells is Egr2-dependent. (a) Diagrammatic representation of the
 Supplementary Figure 1. LAG3 + Treg-mediated regulation of germinal center B cells and follicular helper T cells is Egr2-dependent. (a) Diagrammatic representation of the experimental protocol for the
Supplementary Figure 1. LAG3 + Treg-mediated regulation of germinal center B cells and follicular helper T cells is Egr2-dependent. (a) Diagrammatic representation of the experimental protocol for the
Supplementary Information
 Supplementary Information Methods Lymphocyte subsets analysis was performed on samples of 7 subjects by flow cytometry on blood samples collected in ethylenediaminetetraacetic acid (EDTA)-containing tubes
Supplementary Information Methods Lymphocyte subsets analysis was performed on samples of 7 subjects by flow cytometry on blood samples collected in ethylenediaminetetraacetic acid (EDTA)-containing tubes
Immunology and Pathophysiology of Dry Eye
 Immunology and Pathophysiology of Dry Eye Associate Prof. Banu Bozkurt, MD, FEBO MSc in Immunology Selcuk University Medical Faculty, Konya, Turkey 47th PanHellenic Ophthalmology Congress 28-31 May 2014,
Immunology and Pathophysiology of Dry Eye Associate Prof. Banu Bozkurt, MD, FEBO MSc in Immunology Selcuk University Medical Faculty, Konya, Turkey 47th PanHellenic Ophthalmology Congress 28-31 May 2014,
Pair-fed % inkt cells 0.5. EtOH 0.0
 MATERIALS AND METHODS Histopathological analysis Liver tissue was collected 9 h post-gavage, and the tissue samples were fixed in 1% formalin and paraffin-embedded following a standard procedure. The embedded
MATERIALS AND METHODS Histopathological analysis Liver tissue was collected 9 h post-gavage, and the tissue samples were fixed in 1% formalin and paraffin-embedded following a standard procedure. The embedded
Dry eye syndrome in diabetic children
 European Journal of Ophthalmology / Vol. 17 no. 6, 2007 / pp. 873-878 Dry eye syndrome in diabetic children A. AKINCI 1, E. CETINKAYA 2, Z. AYCAN 2 1 Department of Pediatric Ophthalmology 2 Department
European Journal of Ophthalmology / Vol. 17 no. 6, 2007 / pp. 873-878 Dry eye syndrome in diabetic children A. AKINCI 1, E. CETINKAYA 2, Z. AYCAN 2 1 Department of Pediatric Ophthalmology 2 Department
Supplementary Information:
 Supplementary Information: Follicular regulatory T cells with Bcl6 expression suppress germinal center reactions by Yeonseok Chung, Shinya Tanaka, Fuliang Chu, Roza Nurieva, Gustavo J. Martinez, Seema
Supplementary Information: Follicular regulatory T cells with Bcl6 expression suppress germinal center reactions by Yeonseok Chung, Shinya Tanaka, Fuliang Chu, Roza Nurieva, Gustavo J. Martinez, Seema
Inflammatory Cytokine and Osmolarity Changes in the Tears of Dry Eye Patients Treated with Topical 1% Methylprednisolone
 Original Article http://dx.doi.org/10.3349/ymj.2014.55.1.203 pissn: 0513-5796, eissn: 1976-2437 Yonsei Med J 55(1):203-208, 2014 Inflammatory Cytokine and Osmolarity Changes in the Tears of Dry Eye Patients
Original Article http://dx.doi.org/10.3349/ymj.2014.55.1.203 pissn: 0513-5796, eissn: 1976-2437 Yonsei Med J 55(1):203-208, 2014 Inflammatory Cytokine and Osmolarity Changes in the Tears of Dry Eye Patients
CD14 + S100A9 + Monocytic Myeloid-Derived Suppressor Cells and Their Clinical Relevance in Non-Small Cell Lung Cancer
 CD14 + S1A9 + Monocytic Myeloid-Derived Suppressor Cells and Their Clinical Relevance in Non-Small Cell Lung Cancer Po-Hao, Feng M.D., Kang-Yun, Lee, M.D. Ph.D., Ya-Ling Chang, Yao-Fei Chan, Lu- Wei, Kuo,Ting-Yu
CD14 + S1A9 + Monocytic Myeloid-Derived Suppressor Cells and Their Clinical Relevance in Non-Small Cell Lung Cancer Po-Hao, Feng M.D., Kang-Yun, Lee, M.D. Ph.D., Ya-Ling Chang, Yao-Fei Chan, Lu- Wei, Kuo,Ting-Yu
B220 CD4 CD8. Figure 1. Confocal Image of Sensitized HLN. Representative image of a sensitized HLN
 B220 CD4 CD8 Natarajan et al., unpublished data Figure 1. Confocal Image of Sensitized HLN. Representative image of a sensitized HLN showing B cell follicles and T cell areas. 20 µm thick. Image of magnification
B220 CD4 CD8 Natarajan et al., unpublished data Figure 1. Confocal Image of Sensitized HLN. Representative image of a sensitized HLN showing B cell follicles and T cell areas. 20 µm thick. Image of magnification
Supplementary Figure 1. Characterization of basophils after reconstitution of SCID mice
 Supplementary figure legends Supplementary Figure 1. Characterization of after reconstitution of SCID mice with CD4 + CD62L + T cells. (A-C) SCID mice (n = 6 / group) were reconstituted with 2 x 1 6 CD4
Supplementary figure legends Supplementary Figure 1. Characterization of after reconstitution of SCID mice with CD4 + CD62L + T cells. (A-C) SCID mice (n = 6 / group) were reconstituted with 2 x 1 6 CD4
Dry eye disease (DED) is a common multifactorial
 Immunology and Microbiology MyD88 Deficiency Protects Against Dry Eye Induced Damage Rose Y. Reins, Carolina Lema, Justin Courson, Carolina M. E. Kunnen, and Rachel L. Redfern University of Houston, College
Immunology and Microbiology MyD88 Deficiency Protects Against Dry Eye Induced Damage Rose Y. Reins, Carolina Lema, Justin Courson, Carolina M. E. Kunnen, and Rachel L. Redfern University of Houston, College
In chronic dacryoadenitis, Sjögren s syndrome is a systemic
 Leakage of Aquaporin 5 in the Tear of Dacryoadenitis Mice Shin ichiro Hirai, Naruhiro Ishida, Katsuhiko Watanabe, and Shiro Mita PURPOSE. The objective of this study was to investigate whether leakage
Leakage of Aquaporin 5 in the Tear of Dacryoadenitis Mice Shin ichiro Hirai, Naruhiro Ishida, Katsuhiko Watanabe, and Shiro Mita PURPOSE. The objective of this study was to investigate whether leakage
Canberra, Australia). CD11c-DTR-OVA-GFP (B6.CD11c-OVA), B6.luc + and. Cancer Research Center, Germany). B6 or BALB/c.FoxP3-DTR-GFP mice were
 Supplemental Materials and Methods Mice Female C57BL/6 (B6, I-E null, H-2 b ), BALB/c (H-2 d ) + ), FVB/N (H-2 q, I-E null, CD45.1 + ), and B6D2F1 (H-2 b/d ) mice were purchased from the Animal Resources
Supplemental Materials and Methods Mice Female C57BL/6 (B6, I-E null, H-2 b ), BALB/c (H-2 d ) + ), FVB/N (H-2 q, I-E null, CD45.1 + ), and B6D2F1 (H-2 b/d ) mice were purchased from the Animal Resources
SUPPLEMENTARY INFORMATION. Involvement of IL-21 in the epidermal hyperplasia of psoriasis
 SUPPLEMENTARY INFORMATION Involvement of IL-21 in the epidermal hyperplasia of psoriasis Roberta Caruso 1, Elisabetta Botti 2, Massimiliano Sarra 1, Maria Esposito 2, Carmine Stolfi 1, Laura Diluvio 2,
SUPPLEMENTARY INFORMATION Involvement of IL-21 in the epidermal hyperplasia of psoriasis Roberta Caruso 1, Elisabetta Botti 2, Massimiliano Sarra 1, Maria Esposito 2, Carmine Stolfi 1, Laura Diluvio 2,
SUPPLEMENTARY INFORMATION
 Supplemental Figure 1. Furin is efficiently deleted in CD4 + and CD8 + T cells. a, Western blot for furin and actin proteins in CD4cre-fur f/f and fur f/f Th1 cells. Wild-type and furin-deficient CD4 +
Supplemental Figure 1. Furin is efficiently deleted in CD4 + and CD8 + T cells. a, Western blot for furin and actin proteins in CD4cre-fur f/f and fur f/f Th1 cells. Wild-type and furin-deficient CD4 +
Supplementary Information Titles Journal: Nature Medicine
 Supplementary Information Titles Journal: Nature Medicine Article Title: Corresponding Author: Supplementary Item & Number Supplementary Fig.1 Fig.2 Fig.3 Fig.4 Fig.5 Fig.6 Fig.7 Fig.8 Fig.9 Fig. Fig.11
Supplementary Information Titles Journal: Nature Medicine Article Title: Corresponding Author: Supplementary Item & Number Supplementary Fig.1 Fig.2 Fig.3 Fig.4 Fig.5 Fig.6 Fig.7 Fig.8 Fig.9 Fig. Fig.11
SUPPLEMENTARY FIGURES
 SUPPLEMENTARY FIGURES Supplementary Figure 1: Chemokine receptor expression profiles of CCR6 + and CCR6 - CD4 + IL-17A +/ex and Treg cells. Quantitative PCR analysis of chemokine receptor transcript abundance
SUPPLEMENTARY FIGURES Supplementary Figure 1: Chemokine receptor expression profiles of CCR6 + and CCR6 - CD4 + IL-17A +/ex and Treg cells. Quantitative PCR analysis of chemokine receptor transcript abundance
Islet viability assay and Glucose Stimulated Insulin Secretion assay RT-PCR and Western Blot
 Islet viability assay and Glucose Stimulated Insulin Secretion assay Islet cell viability was determined by colorimetric (3-(4,5-dimethylthiazol-2-yl)-2,5- diphenyltetrazolium bromide assay using CellTiter
Islet viability assay and Glucose Stimulated Insulin Secretion assay Islet cell viability was determined by colorimetric (3-(4,5-dimethylthiazol-2-yl)-2,5- diphenyltetrazolium bromide assay using CellTiter
Supplementary Data 1. Alanine substitutions and position variants of APNCYGNIPL. Applied in
 Supplementary Data 1. Alanine substitutions and position variants of APNCYGNIPL. Applied in Supplementary Fig. 2 Substitution Sequence Position variant Sequence original APNCYGNIPL original APNCYGNIPL
Supplementary Data 1. Alanine substitutions and position variants of APNCYGNIPL. Applied in Supplementary Fig. 2 Substitution Sequence Position variant Sequence original APNCYGNIPL original APNCYGNIPL
Supplemental Table I.
 Supplemental Table I Male / Mean ± SEM n Mean ± SEM n Body weight, g 29.2±0.4 17 29.7±0.5 17 Total cholesterol, mg/dl 534.0±30.8 17 561.6±26.1 17 HDL-cholesterol, mg/dl 9.6±0.8 17 10.1±0.7 17 Triglycerides,
Supplemental Table I Male / Mean ± SEM n Mean ± SEM n Body weight, g 29.2±0.4 17 29.7±0.5 17 Total cholesterol, mg/dl 534.0±30.8 17 561.6±26.1 17 HDL-cholesterol, mg/dl 9.6±0.8 17 10.1±0.7 17 Triglycerides,
REAL-WORLD UTILIZATION PATTERNS OF CYCLOSPORINE OPHTHALMIC EMULSION 0.05% WITHIN MANAGED CARE
 REAL-WORLD UTILIZATION PATTERNS OF CYCLOSPORINE OPHTHALMIC EMULSION 0.05% WITHIN MANAGED CARE Tina H Chiang 1, John G Walt 1, John P McMahon, Jr. 2, James E Mansfield, Jr. 2, Susan Simonyi 3 1 Allergan
REAL-WORLD UTILIZATION PATTERNS OF CYCLOSPORINE OPHTHALMIC EMULSION 0.05% WITHIN MANAGED CARE Tina H Chiang 1, John G Walt 1, John P McMahon, Jr. 2, James E Mansfield, Jr. 2, Susan Simonyi 3 1 Allergan
The first comprehensive definition of DED was published in
 Special Issue Definition and Diagnostic Criteria of Dry Eye Disease: Historical Overview and Future Directions Jun Shimazaki Department of Ophthalmology, Tokyo Dental College, Ichikawa General Hospital,
Special Issue Definition and Diagnostic Criteria of Dry Eye Disease: Historical Overview and Future Directions Jun Shimazaki Department of Ophthalmology, Tokyo Dental College, Ichikawa General Hospital,
Supplemental figure 1. PDGFRα is expressed dominantly by stromal cells surrounding mammary ducts and alveoli. A) IHC staining of PDGFRα in
 Supplemental figure 1. PDGFRα is expressed dominantly by stromal cells surrounding mammary ducts and alveoli. A) IHC staining of PDGFRα in nulliparous (left panel) and InvD6 mouse mammary glands (right
Supplemental figure 1. PDGFRα is expressed dominantly by stromal cells surrounding mammary ducts and alveoli. A) IHC staining of PDGFRα in nulliparous (left panel) and InvD6 mouse mammary glands (right
NK cell flow cytometric assay In vivo DC viability and migration assay
 NK cell flow cytometric assay 6 NK cells were purified, by negative selection with the NK Cell Isolation Kit (Miltenyi iotec), from spleen and lymph nodes of 6 RAG1KO mice, injected the day before with
NK cell flow cytometric assay 6 NK cells were purified, by negative selection with the NK Cell Isolation Kit (Miltenyi iotec), from spleen and lymph nodes of 6 RAG1KO mice, injected the day before with
Supplementary Figure 1. IL-12 serum levels and frequency of subsets in FL patients. (A) IL-12
 1 Supplementary Data Figure legends Supplementary Figure 1. IL-12 serum levels and frequency of subsets in FL patients. (A) IL-12 serum levels measured by multiplex ELISA (Luminex) in FL patients before
1 Supplementary Data Figure legends Supplementary Figure 1. IL-12 serum levels and frequency of subsets in FL patients. (A) IL-12 serum levels measured by multiplex ELISA (Luminex) in FL patients before
Role of Tyk-2 in Th9 and Th17 cells in allergic asthma
 Supplementary File Role of Tyk-2 in Th9 and Th17 cells in allergic asthma Caroline Übel 1*, Anna Graser 1*, Sonja Koch 1, Ralf J. Rieker 2, Hans A. Lehr 3, Mathias Müller 4 and Susetta Finotto 1** 1 Laboratory
Supplementary File Role of Tyk-2 in Th9 and Th17 cells in allergic asthma Caroline Übel 1*, Anna Graser 1*, Sonja Koch 1, Ralf J. Rieker 2, Hans A. Lehr 3, Mathias Müller 4 and Susetta Finotto 1** 1 Laboratory
for six pairs of mice. (b) Representative FACS analysis of absolute number of T cells (CD4 + and
 SUPPLEMENTARY DATA Supplementary Figure 1: Peripheral lymphoid organs of SMAR1 -/- mice have an effector memory phenotype. (a) Lymphocytes collected from MLNs and Peyer s patches (PPs) of WT and SMAR1
SUPPLEMENTARY DATA Supplementary Figure 1: Peripheral lymphoid organs of SMAR1 -/- mice have an effector memory phenotype. (a) Lymphocytes collected from MLNs and Peyer s patches (PPs) of WT and SMAR1
Supporting Online Material for
 www.sciencemag.org/cgi/content/full/1175194/dc1 Supporting Online Material for A Vital Role for Interleukin-21 in the Control of a Chronic Viral Infection John S. Yi, Ming Du, Allan J. Zajac* *To whom
www.sciencemag.org/cgi/content/full/1175194/dc1 Supporting Online Material for A Vital Role for Interleukin-21 in the Control of a Chronic Viral Infection John S. Yi, Ming Du, Allan J. Zajac* *To whom
Supplementary Figure 1. H-PGDS deficiency does not affect GI tract functions and anaphylactic reaction. (a) Representative pictures of H&E-stained
 1 2 3 4 5 6 7 8 9 10 11 Supplementary Figure 1. H-PGDS deficiency does not affect GI tract functions and anaphylactic reaction. (a) Representative pictures of H&E-stained jejunum sections ( 200 magnification;
1 2 3 4 5 6 7 8 9 10 11 Supplementary Figure 1. H-PGDS deficiency does not affect GI tract functions and anaphylactic reaction. (a) Representative pictures of H&E-stained jejunum sections ( 200 magnification;
Current Practice Pattern for Dry Eye Patients in South Korea: A Multicenter Study
 pissn: 11-8942 eissn: 92-9382 Korean J Ophthalmol 14;28(2):115-121 http://dx.doi.org/.3341/kjo.14.28.2.115 Original Article Current Practice Pattern for Dry Eye Patients in South Korea: A Multicenter Study
pissn: 11-8942 eissn: 92-9382 Korean J Ophthalmol 14;28(2):115-121 http://dx.doi.org/.3341/kjo.14.28.2.115 Original Article Current Practice Pattern for Dry Eye Patients in South Korea: A Multicenter Study
VEGFR2-Mediated Vascular Dilation as a Mechanism of VEGF-Induced Anemia and Bone Marrow Cell Mobilization
 Cell Reports, Volume 9 Supplemental Information VEGFR2-Mediated Vascular Dilation as a Mechanism of VEGF-Induced Anemia and Bone Marrow Cell Mobilization Sharon Lim, Yin Zhang, Danfang Zhang, Fang Chen,
Cell Reports, Volume 9 Supplemental Information VEGFR2-Mediated Vascular Dilation as a Mechanism of VEGF-Induced Anemia and Bone Marrow Cell Mobilization Sharon Lim, Yin Zhang, Danfang Zhang, Fang Chen,
PBMC from each patient were suspended in AIM V medium (Invitrogen) with 5% human
 Anti-CD19-CAR transduced T-cell preparation PBMC from each patient were suspended in AIM V medium (Invitrogen) with 5% human AB serum (Gemini) and 300 international units/ml IL-2 (Novartis). T cell proliferation
Anti-CD19-CAR transduced T-cell preparation PBMC from each patient were suspended in AIM V medium (Invitrogen) with 5% human AB serum (Gemini) and 300 international units/ml IL-2 (Novartis). T cell proliferation
METHODS Penh. Measurements and ribonuclease protection assay. Intracellular cytokine staining. ELISAs RT-PCR. Lung morphometry.
 METHODS Penh In addition to measurement of APTI, airway hyperresponsiveness to methacholine was also evaluated within 24 hours of the last allergen challenge by means of measurement of Penh (Buxco Electronics,
METHODS Penh In addition to measurement of APTI, airway hyperresponsiveness to methacholine was also evaluated within 24 hours of the last allergen challenge by means of measurement of Penh (Buxco Electronics,
of whole cell cultures in U-bottomed wells of a 96-well plate are shown. 2
 Supplementary online material Supplementary figure legends Supplementary Figure 1 Exposure to T reg cells causes loss of T resp cells in co-cultures. T resp cells were stimulated with CD3+CD28 alone or
Supplementary online material Supplementary figure legends Supplementary Figure 1 Exposure to T reg cells causes loss of T resp cells in co-cultures. T resp cells were stimulated with CD3+CD28 alone or
Autoimmune Diseases. Betsy Kirchner CNP The Cleveland Clinic
 Autoimmune Diseases Betsy Kirchner CNP The Cleveland Clinic Disclosures (financial) No relevant disclosures Learning Objectives Explain the pathophysiology of autoimmune disease Discuss safe administration
Autoimmune Diseases Betsy Kirchner CNP The Cleveland Clinic Disclosures (financial) No relevant disclosures Learning Objectives Explain the pathophysiology of autoimmune disease Discuss safe administration
I. Lines of Defense Pathogen: Table 1: Types of Immune Mechanisms. Table 2: Innate Immunity: First Lines of Defense
 I. Lines of Defense Pathogen: Table 1: Types of Immune Mechanisms Table 2: Innate Immunity: First Lines of Defense Innate Immunity involves nonspecific physical & chemical barriers that are adapted for
I. Lines of Defense Pathogen: Table 1: Types of Immune Mechanisms Table 2: Innate Immunity: First Lines of Defense Innate Immunity involves nonspecific physical & chemical barriers that are adapted for
(a) Significant biological processes (upper panel) and disease biomarkers (lower panel)
 Supplementary Figure 1. Functional enrichment analyses of secretomic proteins. (a) Significant biological processes (upper panel) and disease biomarkers (lower panel) 2 involved by hrab37-mediated secretory
Supplementary Figure 1. Functional enrichment analyses of secretomic proteins. (a) Significant biological processes (upper panel) and disease biomarkers (lower panel) 2 involved by hrab37-mediated secretory
Evidence for Technology in the Treatment of Advanced Dry Eye
 Evidence for Technology in the Treatment of Advanced Dry Eye COPE 44435-AS Chris Lievens, OD, MS, FAAO Evidence for Technology in the Treatment of Advanced Dry Eye COPE: 44435-AS Chris Lievens, OD MS FAAO
Evidence for Technology in the Treatment of Advanced Dry Eye COPE 44435-AS Chris Lievens, OD, MS, FAAO Evidence for Technology in the Treatment of Advanced Dry Eye COPE: 44435-AS Chris Lievens, OD MS FAAO
MATERIALS AND METHODS. Neutralizing antibodies specific to mouse Dll1, Dll4, J1 and J2 were prepared as described. 1,2 All
 MATERIALS AND METHODS Antibodies (Abs), flow cytometry analysis and cell lines Neutralizing antibodies specific to mouse Dll1, Dll4, J1 and J2 were prepared as described. 1,2 All other antibodies used
MATERIALS AND METHODS Antibodies (Abs), flow cytometry analysis and cell lines Neutralizing antibodies specific to mouse Dll1, Dll4, J1 and J2 were prepared as described. 1,2 All other antibodies used
JMSCR Vol 07 Issue 04 Page April 2019
 www.jmscr.igmpublication.org Index Copernicus Value: 79.54 ISSN (e)-2347-176x ISSN (p) 2455-0450 DOI: https://dx.doi.org/10.18535/jmscr/v7i4.19 Original Research Article Use of Plasma Rich in Growth Factors
www.jmscr.igmpublication.org Index Copernicus Value: 79.54 ISSN (e)-2347-176x ISSN (p) 2455-0450 DOI: https://dx.doi.org/10.18535/jmscr/v7i4.19 Original Research Article Use of Plasma Rich in Growth Factors
Bead Based Assays for Cytokine Detection
 Bead Based Assays for Cytokine Detection September 27, 2014 6 th EFIS-EJI South East European Immunology School SEEIS 2014 Timisoara, Romania The Cells of the Immune System The Immune Reaction (Th2) (Th1)
Bead Based Assays for Cytokine Detection September 27, 2014 6 th EFIS-EJI South East European Immunology School SEEIS 2014 Timisoara, Romania The Cells of the Immune System The Immune Reaction (Th2) (Th1)
Among the benign intraepithelial melanocytic proliferations, Inflamed Conjunctival Nevi. Histopathological Criteria. Resident Short Reviews
 Resident Short Reviews Inflamed conjunctival nevi (ICN) may suggest malignancy because of their rapid growth and atypical histology. The objective of this study was to characterize the diagnostic features
Resident Short Reviews Inflamed conjunctival nevi (ICN) may suggest malignancy because of their rapid growth and atypical histology. The objective of this study was to characterize the diagnostic features
Tear Mucin Assay Kit (O-Glycan Assay Method)
 Product manual (O-Glycan Assay Method) Updated on Apr. 19, 2016 Ⅰ - 1 Background Mucins are major components in tear fluid and apical cell membranes on the ocular surface epithelia. Structurally, they
Product manual (O-Glycan Assay Method) Updated on Apr. 19, 2016 Ⅰ - 1 Background Mucins are major components in tear fluid and apical cell membranes on the ocular surface epithelia. Structurally, they
Supplemental materials
 Supplemental materials 1 Supplemental Fig. 1 Immunogram This immunogram summarizes patient clinical data and immune parameters at corresponding time points for Patient IMF-32. The top panel illustrates
Supplemental materials 1 Supplemental Fig. 1 Immunogram This immunogram summarizes patient clinical data and immune parameters at corresponding time points for Patient IMF-32. The top panel illustrates
LIST OF ORGANS FOR HISTOPATHOLOGICAL ANALYSIS:!! Neural!!!!!!Respiratory:! Brain : Cerebrum,!!! Lungs and trachea! Olfactory, Cerebellum!!!!Other:!
 LIST OF ORGANS FOR HISTOPATHOLOGICAL ANALYSIS:!! Neural!!!!!!Respiratory:! Brain : Cerebrum,!!! Lungs and trachea! Olfactory, Cerebellum!!!!Other:! Spinal cord and peripheral nerves! Eyes, Inner ear, nasal
LIST OF ORGANS FOR HISTOPATHOLOGICAL ANALYSIS:!! Neural!!!!!!Respiratory:! Brain : Cerebrum,!!! Lungs and trachea! Olfactory, Cerebellum!!!!Other:! Spinal cord and peripheral nerves! Eyes, Inner ear, nasal
Meibomian Gland Dysfunction: What Does It Mean James P. McCulley, MD, FACS, FRCOph(UK)
 Meibomian Gland Dysfunction: What Does It Mean James P. McCulley, MD, FACS, FRCOph(UK) David Bruton, Jr. Professor of Ophthalmology Chairman, Department of Ophthalmology The University of Texas Southwestern
Meibomian Gland Dysfunction: What Does It Mean James P. McCulley, MD, FACS, FRCOph(UK) David Bruton, Jr. Professor of Ophthalmology Chairman, Department of Ophthalmology The University of Texas Southwestern
COPD lungs show an attached stratified mucus layer that separate. bacteria from the epithelial cells resembling the protective colonic
 COPD lungs show an attached stratified mucus layer that separate bacteria from the epithelial cells resembling the protective colonic mucus SUPPLEMENTARY TABLES AND FIGURES Tables S1 S8, page 1 and separate
COPD lungs show an attached stratified mucus layer that separate bacteria from the epithelial cells resembling the protective colonic mucus SUPPLEMENTARY TABLES AND FIGURES Tables S1 S8, page 1 and separate
Assessment of Keratitis Damage in an Age Dependent Mouse Model Using Analytical Software
 Assessment of Keratitis Damage in an Age Dependent Mouse Model Using Analytical Software Quincy C. Moore III, PhD, 1 Emmanuel B. Olorunyomi, 1 Miles E. DuBose, 2 and Cleveland O. Lane Jr, PhD 1 1 Prairie
Assessment of Keratitis Damage in an Age Dependent Mouse Model Using Analytical Software Quincy C. Moore III, PhD, 1 Emmanuel B. Olorunyomi, 1 Miles E. DuBose, 2 and Cleveland O. Lane Jr, PhD 1 1 Prairie
sequences of a styx mutant reveals a T to A transversion in the donor splice site of intron 5
 sfigure 1 Styx mutant mice recapitulate the phenotype of SHIP -/- mice. (A) Analysis of the genomic sequences of a styx mutant reveals a T to A transversion in the donor splice site of intron 5 (GTAAC
sfigure 1 Styx mutant mice recapitulate the phenotype of SHIP -/- mice. (A) Analysis of the genomic sequences of a styx mutant reveals a T to A transversion in the donor splice site of intron 5 (GTAAC
Nature Medicine doi: /nm.3957
 Supplementary Fig. 1. p38 alternative activation, IL-21 expression, and T helper cell transcription factors in PDAC tissue. (a) Tissue microarrays of pancreatic tissue from 192 patients with pancreatic
Supplementary Fig. 1. p38 alternative activation, IL-21 expression, and T helper cell transcription factors in PDAC tissue. (a) Tissue microarrays of pancreatic tissue from 192 patients with pancreatic
Figure S1. Generation of inducible PTEN deficient mice and the BMMCs (A) B6.129 Pten loxp/loxp mice were mated with B6.
 Figure S1. Generation of inducible PTEN deficient mice and the BMMCs (A) B6.129 Pten loxp/loxp mice were mated with B6.129-Gt(ROSA)26Sor tm1(cre/ert2)tyj /J mice. To induce deletion of the Pten locus,
Figure S1. Generation of inducible PTEN deficient mice and the BMMCs (A) B6.129 Pten loxp/loxp mice were mated with B6.129-Gt(ROSA)26Sor tm1(cre/ert2)tyj /J mice. To induce deletion of the Pten locus,
The Orbit. The Orbit OCULAR ANATOMY AND DISSECTION 9/25/2014. The eye is a 23 mm organ...how difficult can this be? Openings in the orbit
 The eye is a 23 mm organ...how difficult can this be? OCULAR ANATOMY AND DISSECTION JEFFREY M. GAMBLE, OD COLUMBIA EYE CONSULTANTS OPTOMETRY & UNIVERSITY OF MISSOURI DEPARTMENT OF OPHTHALMOLOGY CLINICAL
The eye is a 23 mm organ...how difficult can this be? OCULAR ANATOMY AND DISSECTION JEFFREY M. GAMBLE, OD COLUMBIA EYE CONSULTANTS OPTOMETRY & UNIVERSITY OF MISSOURI DEPARTMENT OF OPHTHALMOLOGY CLINICAL
Overview & pathophysiology of Dry Eye and the use of cyclosporine eye drops in dry eye...
 Overview & pathophysiology of Dry Eye and the use of cyclosporine eye drops in dry eye... This Allergan sponsored session was held on July 24, 2005, Hotel Satya Ashoka, Jabalpur. The session was followed
Overview & pathophysiology of Dry Eye and the use of cyclosporine eye drops in dry eye... This Allergan sponsored session was held on July 24, 2005, Hotel Satya Ashoka, Jabalpur. The session was followed
(A) PCR primers (arrows) designed to distinguish wild type (P1+P2), targeted (P1+P2) and excised (P1+P3)14-
 1 Supplemental Figure Legends Figure S1. Mammary tumors of ErbB2 KI mice with 14-3-3σ ablation have elevated ErbB2 transcript levels and cell proliferation (A) PCR primers (arrows) designed to distinguish
1 Supplemental Figure Legends Figure S1. Mammary tumors of ErbB2 KI mice with 14-3-3σ ablation have elevated ErbB2 transcript levels and cell proliferation (A) PCR primers (arrows) designed to distinguish
David Clark, MD Chief Medical Officer Aldeyra Therapeutics, Lexington, MA. ARVO: April 30th, 2018
 A Randomized, Double-Masked, Parallel-Group, Phase 2a Dry Eye Disease Clinical Trial to Evaluate the Safety and Efficacy of Topical Ocular ADX-102 (Reproxalap), a Novel Aldehyde Sequestering Agent David
A Randomized, Double-Masked, Parallel-Group, Phase 2a Dry Eye Disease Clinical Trial to Evaluate the Safety and Efficacy of Topical Ocular ADX-102 (Reproxalap), a Novel Aldehyde Sequestering Agent David
Supplementary Figure 1. Deletion of Smad3 prevents B16F10 melanoma invasion and metastasis in a mouse s.c. tumor model.
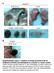 A B16F1 s.c. Lung LN Distant lymph nodes Colon B B16F1 s.c. Supplementary Figure 1. Deletion of Smad3 prevents B16F1 melanoma invasion and metastasis in a mouse s.c. tumor model. Highly invasive growth
A B16F1 s.c. Lung LN Distant lymph nodes Colon B B16F1 s.c. Supplementary Figure 1. Deletion of Smad3 prevents B16F1 melanoma invasion and metastasis in a mouse s.c. tumor model. Highly invasive growth
Supplementary Figures
 Inhibition of Pulmonary Anti Bacterial Defense by IFN γ During Recovery from Influenza Infection By Keer Sun and Dennis W. Metzger Supplementary Figures d a Ly6G Percentage survival f 1 75 5 1 25 1 5 1
Inhibition of Pulmonary Anti Bacterial Defense by IFN γ During Recovery from Influenza Infection By Keer Sun and Dennis W. Metzger Supplementary Figures d a Ly6G Percentage survival f 1 75 5 1 25 1 5 1
Dendritic cell-derived thrombospondin-1 is critical for the generation of the ocular surface Th17 response to desiccating stress
 Article Dendritic cell-derived thrombospondin-1 is critical for the generation of the ocular surface Th17 response to desiccating stress Niral B. Gandhi,* Zhitao Su,* Xiaobo Zhang,*, Eugene A. Volpe,*
Article Dendritic cell-derived thrombospondin-1 is critical for the generation of the ocular surface Th17 response to desiccating stress Niral B. Gandhi,* Zhitao Su,* Xiaobo Zhang,*, Eugene A. Volpe,*
D CD8 T cell number (x10 6 )
 IFNγ Supplemental Figure 1. CD T cell number (x1 6 ) 18 15 1 9 6 3 CD CD T cells CD6L C CD5 CD T cells CD6L D CD8 T cell number (x1 6 ) 1 8 6 E CD CD8 T cells CD6L F Log(1)CFU/g Feces 1 8 6 p
IFNγ Supplemental Figure 1. CD T cell number (x1 6 ) 18 15 1 9 6 3 CD CD T cells CD6L C CD5 CD T cells CD6L D CD8 T cell number (x1 6 ) 1 8 6 E CD CD8 T cells CD6L F Log(1)CFU/g Feces 1 8 6 p
Financial Disclosures
 March 19, 2015 Financial Disclosures Consultant: Alcon Allergan Bausch & Lomb Modernizing Medicine Ophthalmologyweb.co m Investor: Novabay Ophthotech Ocular Surface Disease Ocular surface disease is often
March 19, 2015 Financial Disclosures Consultant: Alcon Allergan Bausch & Lomb Modernizing Medicine Ophthalmologyweb.co m Investor: Novabay Ophthotech Ocular Surface Disease Ocular surface disease is often
IL-6Rα IL-6RαT-KO KO. IL-6Rα f/f bp. f/f 628 bp deleted 368 bp. 500 bp
 STD H 2 O WT KO IL-6Rα f/f IL-6Rα IL-6RαT-KO KO 1000 bp 500 bp f/f 628 bp deleted 368 bp Supplementary Figure 1 Confirmation of T-cell IL-6Rα deficiency. (a) Representative histograms and (b) quantification
STD H 2 O WT KO IL-6Rα f/f IL-6Rα IL-6RαT-KO KO 1000 bp 500 bp f/f 628 bp deleted 368 bp Supplementary Figure 1 Confirmation of T-cell IL-6Rα deficiency. (a) Representative histograms and (b) quantification
Ophthalmic Immunomodulators Prior Authorization with Quantity Limit Program Summary
 Ophthalmic Immunomodulators Prior Authorization with Quantity Limit Program Summary FDA APPROVED INDICATIONS DOSAGE 1,4 Agent Indication Dosage and Administration Restasis (cyclosporine ophthalmic emulsion)
Ophthalmic Immunomodulators Prior Authorization with Quantity Limit Program Summary FDA APPROVED INDICATIONS DOSAGE 1,4 Agent Indication Dosage and Administration Restasis (cyclosporine ophthalmic emulsion)
Dr.Sushil Kumar Tripathi
 Rabamipide:360 Benefits in Dry eye syndrome Dr.Sushil Kumar Tripathi Prevalence of Dry Eye Syndrome: vone in four patients attending ophthalmic clinics report symptoms of dry eye. vprevalence of dry eye
Rabamipide:360 Benefits in Dry eye syndrome Dr.Sushil Kumar Tripathi Prevalence of Dry Eye Syndrome: vone in four patients attending ophthalmic clinics report symptoms of dry eye. vprevalence of dry eye
Effector mechanisms of cell-mediated immunity: Properties of effector, memory and regulatory T cells
 ICI Basic Immunology course Effector mechanisms of cell-mediated immunity: Properties of effector, memory and regulatory T cells Abul K. Abbas, MD UCSF Stages in the development of T cell responses: induction
ICI Basic Immunology course Effector mechanisms of cell-mediated immunity: Properties of effector, memory and regulatory T cells Abul K. Abbas, MD UCSF Stages in the development of T cell responses: induction
White Blood Cells (WBCs)
 YOUR ACTIVE IMMUNE DEFENSES 1 ADAPTIVE IMMUNE RESPONSE 2! Innate Immunity - invariant (generalized) - early, limited specificity - the first line of defense 1. Barriers - skin, tears 2. Phagocytes - neutrophils,
YOUR ACTIVE IMMUNE DEFENSES 1 ADAPTIVE IMMUNE RESPONSE 2! Innate Immunity - invariant (generalized) - early, limited specificity - the first line of defense 1. Barriers - skin, tears 2. Phagocytes - neutrophils,
IKKα Causes Chromatin Modification on Pro-Inflammatory Genes by Cigarette Smoke in Mouse Lung
 IKKα Causes Chromatin Modification on Pro-Inflammatory Genes by Cigarette Smoke in Mouse Lung Se-Ran Yang, Samantha Valvo, Hongwei Yao, Aruna Kode, Saravanan Rajendrasozhan, Indika Edirisinghe, Samuel
IKKα Causes Chromatin Modification on Pro-Inflammatory Genes by Cigarette Smoke in Mouse Lung Se-Ran Yang, Samantha Valvo, Hongwei Yao, Aruna Kode, Saravanan Rajendrasozhan, Indika Edirisinghe, Samuel
Supplementary Figure 1: Neuregulin 1 increases the growth of mammary organoids compared to EGF. (a) Mammary epithelial cells were freshly isolated,
 1 2 3 4 5 6 7 8 9 10 Supplementary Figure 1: Neuregulin 1 increases the growth of mammary organoids compared to EGF. (a) Mammary epithelial cells were freshly isolated, embedded in matrigel and exposed
1 2 3 4 5 6 7 8 9 10 Supplementary Figure 1: Neuregulin 1 increases the growth of mammary organoids compared to EGF. (a) Mammary epithelial cells were freshly isolated, embedded in matrigel and exposed
Online Appendix Material and Methods: Pancreatic RNA isolation and quantitative real-time (q)rt-pcr. Mice were fasted overnight and killed 1 hour (h)
 Online Appendix Material and Methods: Pancreatic RNA isolation and quantitative real-time (q)rt-pcr. Mice were fasted overnight and killed 1 hour (h) after feeding. A small slice (~5-1 mm 3 ) was taken
Online Appendix Material and Methods: Pancreatic RNA isolation and quantitative real-time (q)rt-pcr. Mice were fasted overnight and killed 1 hour (h) after feeding. A small slice (~5-1 mm 3 ) was taken
JMSCR Vol 05 Issue 02 Page February 2017
 www.jmscr.igmpublication.org Impact Factor 5.84 Index Copernicus Value: 83.27 ISSN (e)-2347-176x ISSN (p) 2455-0450 DOI: https://dx.doi.org/10.18535/jmscr/v5i2.155 Prevalence of Dry Eye Diseases in the
www.jmscr.igmpublication.org Impact Factor 5.84 Index Copernicus Value: 83.27 ISSN (e)-2347-176x ISSN (p) 2455-0450 DOI: https://dx.doi.org/10.18535/jmscr/v5i2.155 Prevalence of Dry Eye Diseases in the
Testosterone-Induced Suppression of Autoimmune Diseose in Locrimoi Tissue of o Mouse Model (NZD/NZW F1) of Sjogren's Syndrome
 Investigative phthalmology & Visual Science, Vol. 32, No. 11, ctober 1991 Copyright Association for Research in Vision and phthalmology Testosterone-Induced Suppression of Autoimmune Diseose in Locrimoi
Investigative phthalmology & Visual Science, Vol. 32, No. 11, ctober 1991 Copyright Association for Research in Vision and phthalmology Testosterone-Induced Suppression of Autoimmune Diseose in Locrimoi
Supplementary Figure 1. Ex vivo IFNγ production by Tregs. Nature Medicine doi: /nm % CD127. Empty SSC 98.79% CD25 CD45RA.
 SSC CD25 1.8% CD127 Empty 98.79% FSC CD45RA CD45RA Foxp3 %IFNγ + cells 4 3 2 1 + IL-12 P =.3 IFNγ p=.9 %IL-4+ cells 3 2 1 IL-4 P =.4 c %IL-1 + cells IFNγ 4 3 2 1 Control Foxp3 IL-1 P =.41.64 4.76 MS 2.96
SSC CD25 1.8% CD127 Empty 98.79% FSC CD45RA CD45RA Foxp3 %IFNγ + cells 4 3 2 1 + IL-12 P =.3 IFNγ p=.9 %IL-4+ cells 3 2 1 IL-4 P =.4 c %IL-1 + cells IFNγ 4 3 2 1 Control Foxp3 IL-1 P =.41.64 4.76 MS 2.96
Regulatory B cells in autoimmunity. Liwei Lu University of Hong Kong, China
 Regulatory B cells in autoimmunity Liwei Lu University of Hong Kong, China Multiple functions of B cells in immunity LeBien and Tedder, Blood (2008) B cell functions in autoimmune pathogenesis The Immunopathogensis
Regulatory B cells in autoimmunity Liwei Lu University of Hong Kong, China Multiple functions of B cells in immunity LeBien and Tedder, Blood (2008) B cell functions in autoimmune pathogenesis The Immunopathogensis
Naive, memory and regulatory T lymphocytes populations analysis
 Naive, memory and regulatory T lymphocytes populations analysis Jaen Olivier, PhD ojaen@beckmancoulter.com Cellular Analysis application specialist Beckman Coulter France Introduction Flow cytometric analysis
Naive, memory and regulatory T lymphocytes populations analysis Jaen Olivier, PhD ojaen@beckmancoulter.com Cellular Analysis application specialist Beckman Coulter France Introduction Flow cytometric analysis
SUPPLEMENTARY INFORMATION. CXCR4 inhibitors could benefit to HER2 but not to Triple-Negative. breast cancer patients
 SUPPLEMENTARY INFORMATION CXCR4 inhibitors could benefit to HER2 but not to Triple-Negative breast cancer patients Lefort S. 1,2, Thuleau A. 3, Kieffer Y. 1,2, Sirven P. 1,2, Bieche I. 4, Marangoni E.
SUPPLEMENTARY INFORMATION CXCR4 inhibitors could benefit to HER2 but not to Triple-Negative breast cancer patients Lefort S. 1,2, Thuleau A. 3, Kieffer Y. 1,2, Sirven P. 1,2, Bieche I. 4, Marangoni E.
Effector T Cells and
 1 Effector T Cells and Cytokines Andrew Lichtman, MD PhD Brigham and Women's Hospital Harvard Medical School 2 Lecture outline Cytokines Subsets of CD4+ T cells: definitions, functions, development New
1 Effector T Cells and Cytokines Andrew Lichtman, MD PhD Brigham and Women's Hospital Harvard Medical School 2 Lecture outline Cytokines Subsets of CD4+ T cells: definitions, functions, development New
Suppl Video: Tumor cells (green) and monocytes (white) are seeded on a confluent endothelial
 Supplementary Information Häuselmann et al. Monocyte induction of E-selectin-mediated endothelial activation releases VE-cadherin junctions to promote tumor cell extravasation in the metastasis cascade
Supplementary Information Häuselmann et al. Monocyte induction of E-selectin-mediated endothelial activation releases VE-cadherin junctions to promote tumor cell extravasation in the metastasis cascade
Chronic dry eye disease is principally mediated by effector memory Th17 cells
 nature publishing group Chronic dry eye disease is principally mediated by effector memory Th17 cells Y Chen 1, SK Chauhan 1, H Soo Lee 1, DR Saban 1 and R Dana 1 Recent experimental and clinical data
nature publishing group Chronic dry eye disease is principally mediated by effector memory Th17 cells Y Chen 1, SK Chauhan 1, H Soo Lee 1, DR Saban 1 and R Dana 1 Recent experimental and clinical data
Supplementary Online Content
 Supplementary Online Content Uchino Y, Uchino M, Yokoi N, et al. Alteration of tear mucin 5AC in office workers using visual display terminals: the Osaka Study. JAMA Ophthalmol. Published online June 5,
Supplementary Online Content Uchino Y, Uchino M, Yokoi N, et al. Alteration of tear mucin 5AC in office workers using visual display terminals: the Osaka Study. JAMA Ophthalmol. Published online June 5,
Immune system. Aims. Immune system. Lymphatic organs. Inflammation. Natural immune system. Adaptive immune system
 Aims Immune system Lymphatic organs Inflammation Natural immune system Adaptive immune system Major histocompatibility complex (MHC) Disorders of the immune system 1 2 Immune system Lymphoid organs Immune
Aims Immune system Lymphatic organs Inflammation Natural immune system Adaptive immune system Major histocompatibility complex (MHC) Disorders of the immune system 1 2 Immune system Lymphoid organs Immune
Title: Keeping Step with DEWS2: Clinical Applications Lecturer: Scott G. Hauswirth, OD
 Title: Keeping Step with DEWS2: Clinical Applications Lecturer: Scott G. Hauswirth, OD Course Description: The Dry Eye Workshop 2 was an assemblage of experts in dry eye disease from around the world,
Title: Keeping Step with DEWS2: Clinical Applications Lecturer: Scott G. Hauswirth, OD Course Description: The Dry Eye Workshop 2 was an assemblage of experts in dry eye disease from around the world,
Supplemental Figure 1
 Supplemental Figure 1 1a 1c PD-1 MFI fold change 6 5 4 3 2 1 IL-1α IL-2 IL-4 IL-6 IL-1 IL-12 IL-13 IL-15 IL-17 IL-18 IL-21 IL-23 IFN-α Mut Human PD-1 promoter SBE-D 5 -GTCTG- -1.2kb SBE-P -CAGAC- -1.kb
Supplemental Figure 1 1a 1c PD-1 MFI fold change 6 5 4 3 2 1 IL-1α IL-2 IL-4 IL-6 IL-1 IL-12 IL-13 IL-15 IL-17 IL-18 IL-21 IL-23 IFN-α Mut Human PD-1 promoter SBE-D 5 -GTCTG- -1.2kb SBE-P -CAGAC- -1.kb
Human Immunodeficiency Virus Type-1 Myeloid Derived Suppressor Cells Inhibit Cytomegalovirus Inflammation through Interleukin-27 and B7-H4
 Human Immunodeficiency Virus Type-1 Myeloid Derived Suppressor Cells Inhibit Cytomegalovirus Inflammation through Interleukin-27 and B7-H4 Ankita Garg, Rodney Trout and Stephen A. Spector,,* Department
Human Immunodeficiency Virus Type-1 Myeloid Derived Suppressor Cells Inhibit Cytomegalovirus Inflammation through Interleukin-27 and B7-H4 Ankita Garg, Rodney Trout and Stephen A. Spector,,* Department
Supplementary Data Table of Contents:
 Supplementary Data Table of Contents: - Supplementary Methods - Supplementary Figures S1(A-B) - Supplementary Figures S2 (A-B) - Supplementary Figures S3 - Supplementary Figures S4(A-B) - Supplementary
Supplementary Data Table of Contents: - Supplementary Methods - Supplementary Figures S1(A-B) - Supplementary Figures S2 (A-B) - Supplementary Figures S3 - Supplementary Figures S4(A-B) - Supplementary
Supporting Information
 Supporting Information Valkenburg et al. 10.1073/pnas.1403684111 SI Materials and Methods ELISA and Microneutralization. Sera were treated with Receptor Destroying Enzyme II (RDE II, Accurate) before ELISA
Supporting Information Valkenburg et al. 10.1073/pnas.1403684111 SI Materials and Methods ELISA and Microneutralization. Sera were treated with Receptor Destroying Enzyme II (RDE II, Accurate) before ELISA
L-selectin Is Essential for Delivery of Activated CD8 + T Cells to Virus-Infected Organs for Protective Immunity
 Cell Reports Supplemental Information L-selectin Is Essential for Delivery of Activated CD8 + T Cells to Virus-Infected Organs for Protective Immunity Rebar N. Mohammed, H. Angharad Watson, Miriam Vigar,
Cell Reports Supplemental Information L-selectin Is Essential for Delivery of Activated CD8 + T Cells to Virus-Infected Organs for Protective Immunity Rebar N. Mohammed, H. Angharad Watson, Miriam Vigar,
Interleukin-2-Dependent Allergen-Specific Tissue-Resident Memory Cells Drive Asthma
 Immunity Supplemental Information Interleukin-2-Dependent Allergen-Specific Tissue-Resident Memory Cells Drive Asthma Brian D. Hondowicz, Dowon An, Jason M. Schenkel, Karen S. Kim, Holly R. Steach, Akshay
Immunity Supplemental Information Interleukin-2-Dependent Allergen-Specific Tissue-Resident Memory Cells Drive Asthma Brian D. Hondowicz, Dowon An, Jason M. Schenkel, Karen S. Kim, Holly R. Steach, Akshay
Examples of questions for Cellular Immunology/Cellular Biology and Immunology
 Examples of questions for Cellular Immunology/Cellular Biology and Immunology Each student gets a set of 6 questions, so that each set contains different types of questions and that the set of questions
Examples of questions for Cellular Immunology/Cellular Biology and Immunology Each student gets a set of 6 questions, so that each set contains different types of questions and that the set of questions
Ophthalmology Times Case Study Yasmin Mali, MD. Case Study
 Ophthalmology Times Case Study Yasmin Mali, MD Case Study A 57 year old female with presented with ocular irritation and discomfort in both eyes for several months. Patient was previously started on a
Ophthalmology Times Case Study Yasmin Mali, MD Case Study A 57 year old female with presented with ocular irritation and discomfort in both eyes for several months. Patient was previously started on a
The encephalitogenicity of TH17 cells is dependent on IL-1- and IL-23- induced production of the cytokine GM-CSF
 CORRECTION NOTICE Nat.Immunol. 12, 568 575 (2011) The encephalitogenicity of TH17 cells is dependent on IL-1- and IL-23- induced production of the cytokine GM-CSF Mohamed El-Behi, Bogoljub Ciric, Hong
CORRECTION NOTICE Nat.Immunol. 12, 568 575 (2011) The encephalitogenicity of TH17 cells is dependent on IL-1- and IL-23- induced production of the cytokine GM-CSF Mohamed El-Behi, Bogoljub Ciric, Hong
Interferon γ regulates idiopathic pneumonia syndrome, a. Th17 + CD4 + T-cell-mediated GvH disease
 Interferon γ regulates idiopathic pneumonia syndrome, a Th17 + CD4 + T-cell-mediated GvH disease Nora Mauermann, Julia Burian, Christophe von Garnier, Stefan Dirnhofer, Davide Germano, Christine Schuett,
Interferon γ regulates idiopathic pneumonia syndrome, a Th17 + CD4 + T-cell-mediated GvH disease Nora Mauermann, Julia Burian, Christophe von Garnier, Stefan Dirnhofer, Davide Germano, Christine Schuett,
