3 The abbreviations used are: Eph kinase, erythropoietin-producing hepatocellular
|
|
|
- Abigayle Holmes
- 6 years ago
- Views:
Transcription
1 THE JOURNAL OF BIOLOGICAL CHEMISTRY VOL. 286, NO. 48, pp , December 2, by The American Society for Biochemistry and Molecular Biology, Inc. Printed in the U.S.A. Efnb1 and Efnb2 Proteins Regulate Thymocyte Development, Peripheral T Cell Differentiation, and Antiviral Immune Responses and Are Essential for Interleukin-6 (IL-6) Signaling * S Received for publication, September 9, 2011, and in revised form, September 28, 2011 Published, JBC Papers in Press, October 5, 2011, DOI /jbc.M Hongyu Luo, Tania Charpentier, Xuehai Wang, Shijie Qi, Bing Han, Tao Wu 1, Rafik Terra, Alain Lamarre, and Jiangping Wu 2 From the Laboratoire Immunologie and Service Nephrologie, Centre de Recherche du Centre Hospitalier de l Université de Montréal, Notre-Dame Hospital, Montreal, Quebec H2L 4M1, Canada, Institut National de la Recherche Scientifique, INRS-Institut Armand-Frappier, Laval, Québec H7V 1B7, Canada, and the Institute of Cardiology, First Affiliated Hospital, Medical College, Zhejiang University, Hangzhou, China Background: Ephrins (Efn) are the ligands of Eph kinases. The roles of Efn in the T cell compartment are studied. Results: Efnb1 and Efnb2 double knock-out mice showed compromised thymocyte development, Th1 and Th17 function, IL-6 receptor signaling, and antivirus responses. Conclusion: Efnb1 and Efnb2 are involved in the T cell development and function. Significance: This study has revealed novel biological roles of Efns. Erythropoietin-producing hepatocellular kinases (Eph kinases) constitute the largest family of cell membrane receptor tyrosine kinases, and their ligand ephrins are also cell surface molecules. Because of promiscuous interaction between Ephs and ephrins, there is considerable redundancy in this system, reflecting the essential roles of these molecules in the biological system through evolution. In this study, both Efnb1 and Efnb2 were null-mutated in the T cell compartment of mice through loxp-mediated gene deletion. Mice with this double conditional mutation (double KO mice) showed reduced thymus and spleen size and cellularity. There was a significant decrease in the DN4, double positive, and single positive thymocyte subpopulations and mature CD4 and CD8 cells in the periphery. dko thymocytes and peripheral T cells failed to compete with their WT counterparts in irradiated recipients, and the T cells showed compromised ability of homeostatic expansion. dko naive T cells were inferior in differentiating into Th1 and Th17 effectors in vitro. The dko mice showed diminished immune response against LCMV infection. Mechanistic studies revealed that IL-6 signaling in dko T cells was compromised, in terms of abated * This work was supported in part by Canadian Institutes of Health Research Grants MOP57697 and MOP69089 (to J. W.), IMH and MOP97829 (to H. L.), and MOP89797 (to A. L.), by grants from the Heart and Stroke Foundation of Quebec, the Quebec Ministry of Economic Development, Innovation, and Exportation Grant PSR-SIIRI-069, by The J.-Louis Levesque Foundation (to J. W.), a group grant from the Fonds de la Recherche en Santé du Québec for Transfusional and Hemovigilance Medical Research (to J. W.), and a J.-Louis Levesque Foundation grant (to A. L.). S The on-line version of this article (available at contains supplemental Figs. S1 S3 and Table I, A and B. 1 Supported in part by Zhejiang Provincial Natural Science Foundation of China Grant Y and National Natural Sciences Foundation of China (Projects for Young Scientists Grant ). 2 To whom correspondence should be addressed: Laboratory of Immunology, CHUM Research Centre, Notre-Dame Hospital, Pavillion DeSève, Rm. Y-5616, 1560 Sherbrooke St. East, Montreal, Quebec H2L 4M1, Canada. Tel.: (Ext ); Fax: ; jianping.wu@ umontreal.ca. induction of STAT3 phosphorylation upon IL-6 stimulation. This defect likely contributed to the observed in vitro and in vivo phenotype in dko mice. This study revealed novel roles of Efnb1 and Efnb2 in T cell development and function. Ephs 3 are the largest family of cell surface receptor tyrosine kinases, comprising about 25% of known receptor tyrosine kinases (1). There are a total of 15 Ephs that are classified into A and B subfamilies according to their sequence homology; the former has nine members and the latter six, although not all are expressed in a given species (2, 3). The ligands of Ephs, ephrins (Efns), are also cell surface molecules (1). There are nine Efns divided into A and B subfamilies according to the way they anchor to the cell surface. The Efna subfamily has six members that are glycosylphosphatidylinositol-anchored membrane proteins; the Efnb subfamily has three members that are transmembrane proteins. Interactions between Ephs and Efns are promiscuous. One Eph can interact with multiple Efns and vice versa. In general, Epha members preferentially interact with Efna members and Ephb members with Efnb members (2 4). Such promiscuous interactions suggest that these molecules are so vital to biological systems that heavy redundancy is essential. Ephs are receptor tyrosine kinases that can initiate signal transduction upon ligand binding. However, it is known that although Efns are ligands, they can also transduce signals into cells (2, 3) in a phenomenon called reverse signaling. As a result, the interaction between Eph and Efns results in bi-directional signaling. Because Ephs and Efns are cell surface molecules, 3 The abbreviations used are: Eph kinase, erythropoietin-producing hepatocellular kinase; Efn, ephrin; dko, double KO; BMTx, bone marrow transplantation; DP, double positive; SP, single positive; qpcr, quantitative PCR; LCMV, lymphocytic choriomeningitis virus. DECEMBER 2, 2011 VOLUME 286 NUMBER 48 JOURNAL OF BIOLOGICAL CHEMISTRY 41135
2 they will normally be activated locally by their binding partners expressed on opposing cells during physical contact. As a result, the major functions of Eph and Efn are related to pattern formation; however, additional functions not related to pattern formation have been recently observed. Most reported functions of Ephs occur in the central nervous system where they are expressed in neurons and control axon and dendrite positioning (2, 3). They are essential in the development of neuronal connections, circuit plasticity, and repair. However, they are also involved in a variety of other processes. Some Ephs and Efns are expressed in endothelial cells and are vital in angiogenesis during normal embryonic development as well as in tumorigenesis (4, 5). Studies have shown that intestinal epithelial cells express different levels of some Eph and Efnb family members that modulate the movement of epithelial cells along the crypt axis to maintain epithelium self-renewal (6). It has been reported that Ephb2 and Efnb2 are expressed on the endoderm during embryonic development and that their bidirectional interaction is essential in urorectal development (7). Pancreatic -cells communicate with each other via Epha and Efna family members to synchronize their insulin secretion in response to blood glucose fluctuations (8). Several Ephb and Efnb family members are expressed on osteoclasts and osteoblasts where they regulate bone development, maintenance, and repair (9, 10). Multiple Eph and Efn members have been found to be expressed in some cancer cells, and they seem to influence cancer cell growth (11). Ephb4 and Efnb2 are expressed on hematopoietic progenitor cells and regulate red blood cell production in response to hypoxia (12). Efnb1 and Epha4 expression in platelets contributes to the clotting process (13). Efnb1 expression on kidney epithelial cells (podocytes) likely plays a role in glomerular filtration (14). The interaction between Ephb2 and Efnb2 regulates the ionic homeostasis of vestibular endolymph fluid in the inner ear (15). Our group and others have reported that Ephs and Efns, particularly their B family members as well as some A family members, are expressed in thymocytes and T cells and are capable of modulating T cell responses and survival (16 24). We have shown that Efnb1, Efnb2, and Efnb3 initiate signaling through their Eph receptors and can costimulate peripheral T cells in terms of enhancing cytokine production and proliferation in vitro (25 27). We further demonstrated that one of these Efnb receptors, Ephb6, although lacking kinase activity, can transmit signals into T cells (28) and that its null mutation leads to compromised T cells responses in vitro and in vivo (29). However, Ephb6 null mutants have normal thymus structure and thymocyte development (29), probably due to complementary functions of other Eph family members. To reveal the roles of these highly redundant Eph/Efn family members in thymocyte development and T cell immune responses in vivo and in vitro, we generated Efnb1/Efnb2 double gene knock-out (dko) mice with T cell-specific deletion of these two genes. Results obtained from these mice showed that Efnb1 and Efnb2 are involved in proper thymocyte development and peripheral T cell function. MATERIALS AND METHODS Generation of T Cell-specific Efnb1 and Efnb2 Gene Knockout Mice A PCR fragment amplified with a primer set (5 -CTGAATAAGGGCTGCGAAAG-3 and 5 -GCAAATG- GCTTAACCCAAGA-3 ) based on the Efnb1 genomic sequence was used as a probe to isolate a genomic BAC DNA clone 4M20 from the 129/sv mouse BAC genomic library RPCI- 22. A genomic BAC DNA clone 85F06PCR fragment based on the EFN2 sequence was similarly amplified with the primer set (5 -GCTGC TCTTC AGTCA GTCAG C-3 and 5 -TACCA GTCAT CACAT CGCAG-3 ). The targeting vectors were constructed by recombination and routine cloning methods using a 12-kb Efnb1 genomic fragment from clone 4M20 for Efnb1 and a 12-kb Efnb2 genomic fragment from clone 85F06 for Efnb2 (illustrated in Fig. 1, A and E). The final targeting fragments for Efnb1 and Efnb2 were excised from its cloning vector backbone by NotI and electroporated into ES cells. After G418 selection, the FRT-flanked Neo/TK cassette was eliminated by subsequent transient transfection of the ES cells with a Flippase expression vector. The targeting scheme is shown in Fig. 1A. These genetic engineering steps in ES cells resulted in two net insertions for the EFNB1 constructs as follows: a 118-bp LoxPcontaining sequence (5 -AGTACGGGCC CAAGCTGGCC GCCCTAGGGG CGCGCCTGCA GATAACTTCG TATA- ATGTAT GCTATACGAA GTTATGATAT CAAGCTTATC GATACCGTCG AAGCTTGCTA GCGGTACC-3 ) at position (based on the sequence of AL of the Gen- Bank TM ), and a 151-bp LoxP- plus FRT-containing sequence (5 -GGCCGCCCTA GGGGCGCGCC TGCAGATAAC TTCGATAATG TATGCTATAC GAAGTTATGG ATC- GAAGTTC CTATTTCTAA AAAGTATAGG AACTTCT- TAA GGCCACCGCG GCCGAACGCT AGAGCTTGTC GACGGTACCT AACTTCCTAG G-3 ) at position 28713, 756 bp upstream and 1285 bp downstream of the Efnb1 Exon 1, respectively. Similarly for Efnb2, the genetic engineering steps in ES cells resulted in two net insertions as follows: a 71-bp LoxP-containing sequence (5 -TCGACCTCGA CGGTATC- GAT AAGCTTGATA TCATAACTTC GTATAGCATA CATTATACGA AGTTATCGTT T-3 ) at position (based on the sequence of AC in the GenBank TM ), and a 102-bp LoxP plus FRT-containing sequence (5 -GCGTTA- ATTA AGAAGTTCCT ATACTTTTTA GAGAATAGGA ACTTCGATCC ATAACTTCGT ATAGCATACA TTATAC- GAAG TTATCTGCAG GCGCGCCCCT AG-3 ) at position , 923 bp upstream and 930 bp downstream of Efnb2 Exon 1, respectively. The targeted ES cell clones were injected into C57BL/6 blastocysts. Chimeric male mice were mated with C57BL/6 females to establish mutated Efnb1 and Efnb2 allele germ line transmission. Southern blotting with a probe corresponding to the 3 sequences outside the targeting region, as shown in Fig. 1A (black square), was used to screen and confirm the gene targeting and the successful removal of the Neo-TK selection marker in Efnb1 ES cells and eventually in mouse tail DNA. The targeted allele showed a 7.3-kb AseI/ScaI fragment, whereas the WT allele showed an 11.6-kb fragment (Fig. 1B). Southern blotting with a probe corresponding to the 5 sequences outside the JOURNAL OF BIOLOGICAL CHEMISTRY VOLUME 286 NUMBER 48 DECEMBER 2, 2011
3 targeting region, as illustrated in Fig. 1E (black square), was used to screen and confirm the gene targeting and the successful removal of the Neo-TK selection marker in Efnb2 ES cells and eventually in mouse tail DNA. The targeted allele showed an 8.1-kb PacI/PacI fragment in tail DNA (Fig. 1F), although a WT allele showed a 15.5-kb fragment. PCR was used for routine genotyping of the targeted allele(s). The following PCR conditions were used for Efnb1: 4 min at 95 C, followed by 34 cycles of 15 s at 94 C, 30 s at 58 C, and 60 s at 72 C, and a final incubation at 72 C for 10 min. Primers 5 -GTCTC CACTG CCCAT AGCTC-3 (forward) and 5 -TGCTC CCAGT CCAGTC ACTA-3 (reverse) amplified a 271-bp fragment from the targeted allele and a 133-bp fragment from the WT allele. PCR conditions for Efnb2 were similar except the annealing temperature was reduced from 58 to 52 C. Primers 5 -TAGCC ATCCC TTGGA ATACG-3 (forward) and 5 -TTGGC GCGCC CCTT TCGAAG-3 (reverse) amplified a 456-bp fragment from the targeted allele and a 355-bp fragment from the WT allele. Mice with targeted Efnb1 allele(s) were named Efnb1 f/f (loxp insertions in both alleles) or Efnb1 f (loxp insertion in one allele). Mice with targeted Efnb2 allele(s) were named Efnb2 f/f (loxp insertion in both alleles). These two lines were backcrossed with C57BL/6 for more than 10 generations and then mated with Lck promoter-driven Cre recombinase transgenic mice in the C57BL/6 background (strain B6.Cg-Tg (Lck-Cre) 540Jxm/ J, The Jackson Laboratory, Bar Harbor, ME) to obtain T cellspecific Efnb1 and Efnb2 gene knock-out mice (Lck-Cre- Efnb1 f/f ) and Efnb2 gene knock-out mice (Lck-Cre-Efnb2 f/f ). Because Efnb1 is an X-linked gene, Lck-Cre-Efnb1 f in males is equivalent to Lck-Cre-Efnb1 f/f in females in that the Efnb1 gene is completely null-mutated. For the convenience of description, Lck-Cre-Efnb1 f males and Lck-Cre-Efnb1 f/f females are collectively referred to as Lck-Cre-Efnb1 f/f mice. PCR was used to confirm the T cell-specific deletion of Efnb1 using two different primer pairs. Primer pair 1 (forward, 5 -GTCTC CACTG CCCAT AGCTC-3, and reverse, 5 -TGCTC CCAGT CCAGT CACTA-3 ) was used to detect a 271-bp fragment derived from an allele with undeleted Exon 1. Primer pair 2 (forward, 5 -GTCTC CACTG CCCAT AGCTC-3, and reverse, 5 -AC- CCT TACAT CGAAG AACTG GGCA-3 ) was used to detect a 492-bp fragment derived from an allele with Exon 1 deleted. Similarly, T cell-specific deletion of Efnb2 was confirmed using two different primer pairs. Primer pair 1 (forward, 5 -TAGCC ATCCC TTGGA ATACG-3, and reverse, 5 -TTGGC GCGCC CCTTT CGAAG-3 ) was used to detect a 456-bp fragment derived from an allele(s) with undeleted Exon 1. Primer pair 2 (forward, 5 -CTAAG GCTCT CAGCC TCGTG-3, and reverse, 5 -TTGGC GCGCC CCTTT CGAAG-3 ) was used to detect a 291-bp fragment derived from an allele(s) with Exon 1 deleted. The PCR conditions used were same as described above for routine genotyping. Reverse Transcription/Quantitative-PCR (RT/qPCR) Efnb1, Efnb2, T-bet, and ROR t mrna levels were measured by RT/qPCR. Total RNA from cells was extracted using TRIzol (Invitrogen) and then reverse-transcribed with Superscript II TM reverse transcriptase (Invitrogen). Primers used are listed in supplemental Table IA. qpcr conditions for the reactions was as follows: 2 min at 50 C, 2 min at 95 C followed by 45 cycles of 10 s at 94 C, 20 s at 58 C, and 20 s at 72 C. -Actin or GAPDH mrna levels were used as internal controls, and data were expressed as signal ratios of test gene mrna/control gene mrna. Flow Cytometry Single cell suspensions from the thymus, spleen, or lymph nodes were prepared and stained for flow cytometry as described in our previous publications (25 29). For intracellular antigen staining, cells were first stained with Abs against cell surface antigens, fixed with Cytofix/Cytoperm TM solution (BD Biosciences), and then stained with mabs against intracellular antigens. Antibodies for flow cytometry are shown in supplemental Table IB. The following synthetic peptides were purchased from Sigma Genosys: gp(33 41), KAVYNFATC (LCMV-GP, H-2D b ); np( ), FQPQNGQFI (LCMV-NP, H-2D b ); gp( ), SGVENPGGYCL (LCMV-GP, H-2D b ); and gp(61 80), GLNGPDIYKGVYQFKSVEFD (LCMV-GP, I-A b ). PE-gp(33 41), PE-np( ), and PE-gp( ) H-2D b tetrameric complexes were synthesized in-house and used at 1:100 dilution as described previously (30). These MHC tetramers were used to detect LCMV-specific CD8 T cells. Briefly, splenocytes were first stained with PE-gp(33 41), PE-np( ), or PE-gp( ) tetramers for 30 min at 37 C, followed by staining with FITC rat anti-mouse CD8 and APC-rat anti-mouse CD62L mabs at 4 C for another 20 min. 7-Aminoactinomycin D was used for exclusion of dead cells. After washing, cells were fixed in 0.5% paraformaldehyde, and samples were analyzed by flow cytometry. Assessment of Thymocyte and T Cell Proliferation Mice were injected intraperitoneally with 1 mg of 5-bromo-2-deoxyuridine (BrdU) in 0.2 ml of PBS and sacrificed 90 min later for thymocyte analysis. Another group of mice was injected with BrdU daily for 4 days and then sacrificed for spleen T cell analysis. Thymocytes or spleen cells were then isolated and stained with Abs against cell surface markers. They were subsequently permeabilized and stained with FITC-conjugated anti-brdu Ab using a BrdU flow kit (BD Biosciences) according to the manufacturer s instructions. Generation of Bone Marrow Chimeras Eight- to 10-weekold C57BL/6(CD45.2 ) C57B6.SJL(CD45.1 ) F1 mice were irradiated at 1100 rads. Twenty four hours later, they received T cell-depleted bone marrow cells from C57/B6.SJL and dko mice in a 1:1 ratio. Efnb1 f/f /Efnb2 f/f mouse bone marrow was used as a control. Thymocytes and spleen cells of the recipients were analyzed by flow cytometry 8 10 weeks following the bone marrow transplantation (BMTx). In Vitro Th1, Th2, Th17, and Treg Polarization Naive CD4 T cells were isolated from pooled splenocytes and lymph node cells using the naive CD4 T cells isolation kit (R&D Systems). Purity of the naive CD4 cells was routinely greater than 95%. Purified naive T cells ( cells/well) were mixed with T cell-depleted irradiated (3000 rads) C57BL/6 feeder splenocytes ( cells/well) and cultured in 96-well plates under Th1, Th2, Th17, or Treg polarization conditions, as described in our earlier publication (31), and then analyzed by flow cytometry. DECEMBER 2, 2011 VOLUME 286 NUMBER 48 JOURNAL OF BIOLOGICAL CHEMISTRY 41137
4 LCMV Infection and Clearance LCMV-WE was obtained from Dr. R. M. Zinkernagel (Zurich, Switzerland). Viral stock was propagated in vitro, and viral titers were determined by the focus-forming assay as described previously (32). Mice were infected by the i.v. route with 200 focus-forming units of LCMV-WE. Mice were sacrificed 8 days post-infection to collect spleens for primary immune response analysis or 1 month later for memory immune response analysis. To evaluate viral clearance, mice were bled at days 4, 5, 7, 9, 11, 14, 23, and 28 post-infection, and LCMV titers in whole blood were measured by the focus-forming assay. Detection of LCMV-specific IFN- and TNF- -producing T Cells One million splenocytes from LCMV-infected mice were seeded in single wells of 96-well round-bottomed plates. Cells were maintained in 5% RPMI 1640 medium supplemented with 100 units/ml mril-2, 10 g/ml brefeldin A, 10 M gp(33 41) or gp(61 80) peptide. After 5 h of incubation at 37 C, cells were stained with phycoerythrin-conjugated rat anti-mouse CD8 or CD4 mabs and 7-aminoactinomycin D. Cells were then fixed, permeabilized and stained with allophycocyanin-rat anti-mouse TNF- and FITC-rat anti-mouse IFN- mabs. Frequency of IFN- and TNF- -secreting T cells was determined by flow cytometry. 51 Cr Release Assay Eight days following LCMV infection, splenocytes were tested for cytotoxic activity in a standard 51 Cr release assay as described previously (32). Immunoblotting dko or control spleen T cells were purified with the EasySep TM T cell enrichment kit from Stemcell Technologies (Vancouver, British Columbia, Canada). These cells as well as total thymocytes were reacted with or without IL-6 (50 ng/ml) at 37 C for 5 min for the detection of STAT3 phosphorylation. The cells were lysed in RIPA buffer (25 mm Tris, ph 7.6, 150 mm NaCl, 1% Nonidet P-40, 1% sodium deoxycholate, 0.1% SDS) supplemented with protease inhibitors and phosphatase inhibitors (Complete TM protease inhibitor mixture and PhosSTOP phosphatase inhibitor mixture, Roche Diagnostics). The lysates were resolved by 8% SDS-PAGE and transferred to nitrocellulose membranes. STAT3 was detected by blotting with rabbit anti-mouse phospho-stat3 (Tyr-705) monoclonal antibody (clone D3A7, Cell Signaling Technology) followed by HRP-conjugated donkey anti-rabbit IgG (GE Healthcare). The membranes were then stripped and reprobed with rabbit anti-mouse STAT3 mab (clone 79D7, Cell Signaling Technology) for total STAT3 expression. The signals were detected with SuperSignal West Pico Chemiluminescent Substrate (Thermo Scientific, Rockford, IL). RESULTS Generation of Efnb1 and Efnb2 Conditional KO Mice Efnb1 and Efnb2 null mutation caused embryonic lethality (4, 33). To study the function of these genes in the T cell compartment, we generated conditional KO mice with LoxP sites flanking Exon 1 of Efnb1 or Efnb2 (Fig. 1, A, B, E, and F). These mice (Efnb1 f/f and Efnb f/f ) were crossed with transgenic mice expressing Cre recombinase driven by a proximal Lck promoter. As shown in the upper panels of Fig. 1, C and G, in the presence of the Lckpromoter Cre transgene, Exon 1 of Efnb1 or Efnb2 was deleted in thymocytes but was maintained in the genome of tail tissue in Lck-Cre-Efnb1 f/f and Lck-Cre-Efnb2 f/f mice. RT-qPCR confirmed T cell-specific deletion of Efnb1 and Efnb2 mrna in thymocytes and spleen T cells but not in spleen B cells (lower panels, Fig. 1, C and G). mrna deletion in the thymocytes and spleen T cells was not complete in this quantitative assay, probably because of the less than perfect effectiveness of the LoxP-Cre deletion system. Abrogated cell surface expression of Efnb1 and Efnb2 proteins in thymocytes and peripheral T cells is demonstrated by flow cytometry (Fig. 1, D and H). The transgenic Cre in transgenic mice B6.Cg-Tg (Lck- Cre) 540Jxm/J generated by Dr. J. Marth was driven by the Lck proximal promoter (34). This proximal promoter becomes most effective in thymocytes starting from the DN3 stage (34, 35). Indeed, the effective deletion of Efnb1 started from the DN3 (double negative 3) stage and onward (Fig. 1D). For Efnb2, obvious deletion was observed from the DN4 stage and onward, although the deletion could also be seen at the DN2 and DN3 stage (Fig. 1H). Because the Lck proximal promoter ceases to be active after the SP stages (34, 35), the Efnb1 and Efnb2 deletion observed in peripheral T cells (Fig. 1, D and H) was caused by Efnb1 and Efnb2 deletion in their progenitor cells, i.e. DN3 and DN4 cells. Although the deletion in the DN3 and DN4 stage was not complete, the residual undeleted DN cells did not seem to develop into a major population in the periphery, and even the incomplete deletion was sufficient to cause phenotypic changes in the T cell compartment, as will be described below. It is worth mentioning that deletion of Efnb1 alone did not cause compensative up-regulation of Efnb2 or vice versa (supplemental Fig. 1A). Thymus and Spleen Features of dko Mice Lck-Cre-Efnb1 f/f and Lck-Cre-Efnb2 f/f single gene knock-out mice showed no discernable difference from the control Efnb1 f/f and Efnb2 f/f mice in terms of lymphoid organ size and histology (data not shown). We therefore generated Lck-Cre-Efnb1 f/f /Efnb2 f/f dko mice. Efnb1 f/f /Efnb2 f/f mice were used as controls and are called WT controls in the subsequent text. The dko mice had smaller thymi and spleens (Fig. 2A). The weight and cellularity of these organs and the T cell number in the spleen were diminished compared with those of WT control mice (Fig. 2B). Efnb1 and Efnb2 Are Essential in Thymocyte Development Thymocyte subpopulation analysis by flow cytometry of Lck- Cre-Efnb1 f/f and Lck-Cre-Efnb2 f/f single gene knock-out mice showed no significant abnormality (data not shown). The dko thymi were then analyzed. Representative histograms are shown in Fig. 3A, and data from the 20 to 27 pairs of dko and controls are summarized in Table 1. The absolute number but not the percentage of CD4 and CD8 SP cells of dko thymi was decreased compared with that of controls (Table 1). For the double positive (DP) cells that constitute the bulk of the thymus, the cell number in the dko thymi was significantly reduced compared with controls (a mean of cells in the dko thymi versus a mean of in the control thymi), consistent with the reduced thymic weight and cellularity in the dko mice; the percentage of these cells in the dko thymi also showed significant decrease (Table 1). These results show that Efnb1 and Efnb2 play a critical role in DP population development, and the diminished pool of DP cells in the dko JOURNAL OF BIOLOGICAL CHEMISTRY VOLUME 286 NUMBER 48 DECEMBER 2, 2011
5 DECEMBER 2, 2011 VOLUME 286 NUMBER 48 JOURNAL OF BIOLOGICAL CHEMISTRY 41139
6 FIGURE 1 continued JOURNAL OF BIOLOGICAL CHEMISTRY VOLUME 286 NUMBER 48 DECEMBER 2, 2011
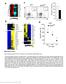
7 FIGURE 2. General features of dko lymphoid organs. A, gross morphology of the dko thymus and spleen. B, weight and cellularity of dko thymus and spleen. Mouse number (n) of each group is shown. Asterisks indicate a highly significant difference (p 0.01, paired Student s t test). thymus might contribute to smaller SP populations. There was a significant relative increase in the percentage of double negative (DN) cells in the dko thymi, although the absolute number of these cells remained unchanged compared with controls (Table 1). This suggests that the increased DN percentage is mainly a result of a decreased DP cell population. We wondered whether compromised progression from DN cells to DP cells contributed to the significant decrease in the DN population in the dko thymus. Because the proximal Lck promoter is most effective in the DN3 and DN4 stages (35), we chose to analyze subpopulations from the DN2 to DN4 stages. Representative histograms are shown in Fig. 3B, and a summary of data from 14 to 22 pairs of dko mice and controls are presented in Table 2. The percentage and absolute cell number of the DN2 subpopulation in the dko thymus were comparable with controls, whereas those of the DN3 subpopulation were increased, and those of the DN4 subpopulation were reduced (Table 2). This is consistent with the notion that the most effective Cre expression driven by the Lck proximal promoter starts from the DN3 stage. The results suggest that deletion of both Efnb1 and Efnb2 impairs the development from the DN3 to the DN4 stage and that this is at least a contributing factor leading to a reduced DP population. The existence of different thymocyte subpopulations represents a balance between their proliferation, apoptosis, and progression. We wondered whether null mutation of both Efnb1 and Efnb2 affects the proliferation of thymocytes. The proliferation of DN3, DN4, DP, and SP subpopulations was assessed in vivo. As shown in Fig. 3C, no apparent difference in the proportion of BrdU-positive CD25 ictcr or CD25 ictcr DN3 cells or CD25 ictcr DN4 cells was observed between dko and control thymi nor was there any difference at the DP, CD4SP, and CD8SP stages. We used ictcr or ictcr to gate DN3 and DN4 subpopulations for their BrdU uptake (Fig. 3C). As dko and control thymi did not differ significantly in their ictcr cell percentage at the DN3 and DN4 stages (supplemental Fig. 1B), the BrdU results from Fig. 3C were not caused by abnormal ictcr expression in dko cells. These results indicate that the reduced cellularity of certain subpopulations in the dko thymus does not seem to be the result of their decreased proliferation. FIGURE 1. Generation of mice with T cell-specific Efnb1 f/f and Efnb2 f/f null mutation. A and E, schemes of Efnb1 f/f and Efnb2 f/f mouse generation. The schemes of genetic manipulation to generate floxed Efnb1 (A) and Efnb2 (E) mice are illustrated. Bold lines represent left and right arms of genomic sequences used in gene targeting. Empty squares represent exons (E) or thymidine kinase gene/neomycin-resistant gene cassettes (TK/Neo). LoxP and FRT sites are represented by large and small arrowheads, respectively. The solid box represents a genomic region from which probes were produced for Southern blot analysis. B and F, Southern blot analysis of tail DNA of Efnb1- and Efnb2-targeted mice. For Efnb1-targeted mice (B), tail DNA was digested with AseI and ScaI and analyzed by Southern blotting. The 11.6-kb band was derived from the wild type allele, and the 7.3-kb band was from the mutated allele. For Efnb2-targeted mice (F), the tail DNA was digested with PacI and analyzed by Southern blotting. The 15.5-kb band was derived from the wild type allele, and the 8.1-kb band was from the mutated allele. C and G, T cell-specific deletion of Efnb1 and Efnb2 in Lck-Cre-Efnb1 f/f and Lck-Cre-Efnb2 f/f mice according to PCR. Efnb1 f/f mice and Efnb2 f/f mice were crossed with transgenic mice harboring Cre recombinase driven by the proximal LCK promoter. In the upper panel, the tail and thymocyte DNA of the mice was analyzed with PCR using primer pairs 1 and 2 as described in supplemental material. For Efnb1 f/f mice, primer pair 1 detected a 271-bp fragment derived from the undeleted allele, although primer pair 2 detected a 492-bp fragment derived from the allele with Efnb1 exon 1 deleted. For Efnb2 f/f mice, primer pair 1 detected a 459-bp fragment derived from the undeleted allele, whereas primer pair 2 detected a 291-bp fragment derived from the allele with the Efnb2 exon 1 deleted. In the lower panel, total RNA was extracted from Lck-Cre-Efnb1 f/f (C)orLck-Cre-Efnb2 f/f (G) thymocytes, spleen T cells, and spleen B cells. Their Efnb1 and Efnb2 mrna was analyzed by RT/qPCR using -actin as an internal control. Data are expressed as the means S.D. of the ratios of Efnb1/ -actin (C) and Efnb2/ -actin (G) signals. D and H, deletion of Efnb1 and Efnb2 expression in Lck-Cre-Efnb1 f/f and Lck-Cre-Efnb2 f/f thymocytes and spleen T cells according to flow cytometry. Thymocytes at different differentiation stages (DN2 4, DP, CD4SP, and CD8SP) and spleen CD4 and CD8 cells were gated and were analyzed for Efnb1 expression (E) or Efnb2 expression (H) by flow cytometry. The dotted line, isotypic control Ab; the dashed line, control WT cells stained with anti-efnb1 or anti-efnb2 Abs; the solid line, Lck-Cre-Efnb1 f/f and Lck-Cre-Efnb2 f/f cells stained with anti-efnb1 or anti-efnb2 Abs, respectively. The percentage of Efnb1- or Efnb2-positive cells after the deduction of the background signal (isotypic Ab control) is shown. All the experiments in this figure were repeated two or more times, and data from representative ones are shown. DECEMBER 2, 2011 VOLUME 286 NUMBER 48 JOURNAL OF BIOLOGICAL CHEMISTRY 41141
8 Efnb1 and Efnb2 Required for T Cell Development FIGURE 3. Flow cytometry analysis of dko thymocytes. A, DN, DP, CD4SP, and CD8SP subpopulations in dko thymi. Thymocytes were analyzed for DN, DP, CD4SP, and CD8SP subpopulations. Representative histograms are shown, and the percentage of each subpopulation is indicated. Data from 20 to 27 pairs of dko and control WT mice are summarized in Table 1. B, DN2, DN3, and DN4 subpopulations in dko thymi. Thymocytes were first gated on Lin cells, and the Lin cells were analyzed for DN2, DN3, and DN4 subpopulation based on CD25 and CD44 expression. Representative histograms are shown. Data from 20 to 27 pairs of dko and control WT mice are summarized in Table 2. C, DN3, DN4, DP, CD4SP, and CD8SP cell proliferation in dko thymi according to BrdU staining. Control Efnb1f/f/Efnb2f/f and dko mice were injected intraperitoneally with BrdU (1 mg/mouse), and their thymocytes were analyzed by flow cytometry 90 min later. Thymocytes were stained with lineage markers as well as anti-brdu Ab. Percentages of BrdU-positive DN3 (CD25 ictcr and CD25 ictcr ), DN4 (CD25 ictcr ), DP, CD4SP, and CD8SP cells are shown. D, dko thymocyte apoptosis. dko and WT control thymocytes were stained with annexin V and lineage markers ex vivo or cultured for 24 h in plain medium and then analyzed as indicated. The percentage of annexin V-positive cells in DN3, DN4, DP, CD4SP, and CD8SP cells is shown. In C and D, the experiments were repeated more than three times, and representative histograms are shown. We then evaluated whether thymocyte over-apoptosis was a factor causing the decrease of certain thymic subpopulations in dko mice. Freshly isolated dko thymocytes presented enhanced annexin V expression in all stages examined (DN3, DN4, DP, CD4SP, and CD8SP) compared with the controls (Fig. 3D). The higher apoptotic rate in DN3 cells does not seem to be consistent with increased DN3 cell percentage and absolute number in dko thymi; it is possible that the effect of the blockage of progress from DN3 to DN4 resulted in an accumu JOURNAL OF BIOLOGICAL CHEMISTRY lation of DN3 cells that outweighed the detrimental effect of apoptosis in this subpopulation. The results of this section indicate that in the absence of Efnb1 and Efnb2, there is increased apoptosis in all subsets of thymocytes, and this could contribute to the reduced cellularity in the T cell compartment. We assessed the CD3, CD5, CD24, and CD69 expression in dko thymocyte subpopulations, particularly at the DP stage, where negative and positive selections occur. These cell surface proteins are critical in negative (CD3 and CD25) and positive VOLUME 286 NUMBER 48 DECEMBER 2, 2011
9 TABLE 1 Percentage and absolute numbers of CD4SP, CD8SP, DP, and DN subpopulations CD4SP CD8SP DP DN % Cells ( 10 6 ) % Cells ( 10 6 ) % Cells ( 10 6 ) % Cells ( 10 6 ) WT dko a a b a a n 27 n 20 n 27 n 20 n 27 n 20 n 27 n 20 a p b p 0.01; Student s t test. TABLE 2 Percentage and absolute numbers of DN2, DN3, and DN4 thymocytes DN2 DN3 DN4 % Cells ( 10 6 ) % Cells ( 10 6 ) % Cells ( 10 6 ) WT dko a b a b n 16 n 14 n 22 n 14 n 22 n 14 a p b p 0.05; Student s t test. selection or markers (CD24 and CD69) of the selection process when TCR activation occurs (36 39). No significant difference was found between dko and WT cells (supplemental Fig. 2A) with regard to the expression of these molecules. Therefore, the significantly reduced percentage and absolute number of DP cells (Table 1) in the dko thymus is unlikely to be a result of a more aggressive negative selection or less efficient positive selection. Rather, it is more likely due to increased general apoptosis in all thymocyte subpopulations and blocked progression from DN3 to DN4. Efnb1 and Efnb2 Are Required for Proper Function of Peripheral T Cells in Vitro The peripheral T cell subpopulation and T cell function of dko mice were evaluated next. There was a significant decrease of spleen weight and cellularity (Fig. 2B) and a significant reduction in T cell percentage in the spleen and lymph nodes of dko mice compared with controls (Fig. 4A). Both the CD4 and CD8 cells of the dko spleen and lymph nodes were proportionally decreased (Fig. 4B). There was a 2-fold relative increase in the percentage of CD44 hi CD62L lo memory phenotype CD4 and CD8 T cells in the dko spleen compared with the WT spleen (Fig. 4C, histograms, left panel). This increase came at the expense of CD44 lo CD62L hi dko naive CD4 and CD8 T cells that were reduced almost 2-fold compared with controls. Considering the approximate 2-fold decrease of the absolute T cell number in the dko spleen (Fig. 2B), the absolute memory phenotype T cell number in the dko spleen remained unchanged, although both percentage and absolute number of naive T cells in the dko spleen were significantly reduced (Fig. 4C, bar graph, right panel). It seems that Efnb1 and Efnb2 affect naive CD4 and CD8 cells more than memory cells. Some CD44 hi CD62L lo T cells in the periphery are derived directly from CD44 hi CD62L lo thymocytes (40) and display innate immune function by rapidly secreting IFN- upon stimulation (41). We examined the SP thymocytes of dko and WT for their CD44 and CD62L expression and noticed a higher CD44 hi CD62L lo percentage in dko than in WT thymi (Fig. 4D). We also evaluated CD44 hi CD62L lo spleen T cells for their intracellular IFN expression 4 h after stimulation with phorbol 12-myristate 13-acetate and ionomycin. IFN- -positive WT versus dko CD4CD44 hi cells were 1.5 versus 6.2%, and WT versus dko CD8CD44 hi cells were 5.2 versus 6.0% (supplemental Fig. 2B). So, there is indeed an increase in the innate type CD44 hi CD62L lo T cell population in the periphery, and these cells are probably directly derived from the thymus as suggested by Rodgers (40). However, the increment is small in scale compared with the increase of CD44 hi CD62L lo CD4 and CD8 cells in the dko mice as shown in Fig. 4C. Because the majority of the dko peripheral CD44 hi CD62L lo cells did not behave like innate immune cells by rapidly secreting IFN upon phorbol 12-myristate 13-acetate /ionomycin stimulation, they are probably genuine memory T cells. Are there additional factors causing the skew of memory versus naive T cell ratio in dko mice? We assessed the apoptosis of these cells from the spleen. Both naive and memory type CD4 and CD8 cells from dko mice showed an increased annexin V-positive percentage compared with those from WT mice (Fig. 4E), but naive type cells did not present a higher proportion compared with memory type cells. This higher apoptotic rate of all the dko subsets likely contributes to the reduced peripheral T cell populations in dko mice, but it is not a reason for a relatively higher percentage of memory type T cells in these mice. We also demonstrated that both naive and memory type T cells from dko mice manifested a similar BrdU uptake in vivo compared with cells from WT mice (supplemental Fig. 3A). Taken together, these results suggest that the significant reduction of naive dko T cells compared with memory dko T cells is due to reduced output of naive T cells from the dko thymus, but Efnb1 and Efnb2 do not seem to affect significantly the conversion from naive to memory type T cells. We next assessed different functional T cell subpopulations either ex vivo or following in vitro differentiation. There was no change in the relative proportion of CD25 FoxP3 Treg cells in the dko spleen and lymph nodes relative to the control spleen and lymph nodes when examined ex vivo (supplemental Fig. 3B). When naive CD4 cells were cultured under Th1, Th2, Th17, and Treg conditions (Fig. 4F), the dko cells (lower row) showed compromised differentiation toward Th1 (IFN- IL4 cells; 1st panel) and Th17 (IFN- IL-17 cells; 3rd panel) but not Th2 (IL-4 IFN- cells; 2nd panel) nor Treg DECEMBER 2, 2011 VOLUME 286 NUMBER 48 JOURNAL OF BIOLOGICAL CHEMISTRY 41143
10 Efnb1 and Efnb2 Required for T Cell Development FIGURE 4. Flow cytometry analysis of dko peripheral lymphocytes and their in vitro differentiation. A, T and B cell subpopulations in dko spleens. Spleen and lymph node cells were stained with anti-cd3 and anti-b220 Abs for T and B cells, respectively. Percentage of CD3- and B220-positive cells is shown. B, CD4 and CD8 T cell subpopulations in dko spleens. Spleen or lymph node cells were stained with anti-cd4 and anti-cd8 Abs for CD4 and CD8 T cells, respectively. Percentage of CD4- and CD8-positive cells is shown. C, naive and memory T cell populations in dko spleens. CD44loCD62Lhi cells and CD44hiCD62Llo cells among CD4 and CD8 spleen cells were analyzed, and their percentage is shown in the histogram. The bar graph at left illustrates the absolute number of CD44loCD62Lhi cells and CD44hiCD62Llo cells in the spleen based on the calculation of the percentage and spleen cellularity of 10 pairs of dko and WT mice. ***, p (Student s t tests). D, CD44loCD62Lhi cells and CD44hiCD62Llo cells in the dko SP thymocytes. CD4SP and CD8SP thymocytes were gated, and CD44hiCD62Llo cells among these subpopulations were analyzed by flow cytometry. Their percentage is shown in the histograms. E, apoptosis of spleen CD44loCD62Lhi cells and CD44hiCD62Llo T cells of dko mice. dko and WT spleen CD44loCD62Lhi cells and CD44hiCD62Llo cells in the CD4 and CD8 subpopulations were analyzed ex vivo for their annexin V expression by flow cytometry. F, in vitro Th1, Th2, Th17, and Treg differentiation according to intracellular IFN-, IL-4, IL-17, and Foxp3 staining. Naive CD4 cells were cultured under Th1, Th2, Th17, or Treg conditions (3 days for Th1, Th17, and Treg and 5 days for Th2). Cells were further stimulated with phorbol 12-myristate 13-acetate and ionomycin for 4 h before being harvested. The intracellular expression of IFN-, IL-4, IL-17, and Foxp3 was determined by flow cytometry. Percentage of IFN-, IL-4, IL-17, and Foxp3-positive cells is shown. G, expression of T-bet and ROR t mrna in Th1 and Th17 cells. T-bet and ROR t mrna from in vitro differentiated Th1 and Th17 cells, as described in F, was quantified by RT-qPCR, using GAPDH as an internal control. Data from five independent experiments are expressed as the means S.D. of the ratios of T-bet/GAPDH or ROR t/gapdh signals. *, p 0.05 (Student s t test). Experiments in this figure were repeated more than three times, unless indicated otherwise, and representative data are shown JOURNAL OF BIOLOGICAL CHEMISTRY VOLUME 286 NUMBER 48 DECEMBER 2, 2011
11 Efnb1 and Efnb2 Required for T Cell Development FIGURE 4 continued (IL-17 FoxP3 cells; 4th panel), compared with controls (upper row). In testing the in vitro Th1 and Th17 differentiation, we started with the same number of naive CD4 cells. The compromised Th1 and Th17 development of dko CD4 could be due to defective expansion or differentiation, or both. To distinguish these possibilities, we examined the expression of transcription factors T-bet and ROR t, which are essential transcription factors in Th1 and Th17 cell differentiation, respectively. As shown in Fig. 4G, under Th1 culture conditions, there was a small but significant increase in T-bet expression in dko T cells compared with WT T cells, although the ROR t expression of the dko and WT cells under Th17 culture conditions was similar. The reason for this small increase is not clear, but this result suggests that the defective Th1 and Th17 cell developdecember 2, 2011 VOLUME 286 NUMBER 48 ment of dko CD4 T cells is probably mainly due to compromised expansion rather than differentiation. Pivotal Role of Efnb1 and Efnb2 Reverse Signaling in T Cell Development and Homeostatic Expansion Ephs are capable of transducing signals in both directions (2, 3). We wanted to understand which direction of signaling was critical for the observed phenotype in the dko mice. To this end, we used a model of whole body irradiation followed by BMTx, using B6.SJL bone marrow cells to compete with dko bone marrow cells. In this model, CD45.1 single positive cells were derived from competitor B6.SJL bone marrow cells; CD45.2 single positive cells were derived from the dko bone marrow cells; and CD45.1/CD45.2 double positive cells were derived from residual recipient bone marrow cells and peripheral cells. JOURNAL OF BIOLOGICAL CHEMISTRY 41145
12 Efnb1 and Efnb2 Required for T Cell Development FIGURE 5. Compromised development and expansion of dko bone marrow cells and mature T cells in irradiated recipients. A, dko bone marrow cells failed to compete with WT bone marrow cells in irradiated recipients. T cell-depleted dko and WT bone marrow cells (CD45.2 ) were mixed with T cell-depleted bone marrow cells from B6.SJL competitors (CD45.1 ) at 1:1 ratio and transplanted to lethally irradiated C57BL/6 B6.SJL F1 recipients. After 90 days, cells from the thymus, spleen, lymph node, and blood were analyzed for CD45.2 and CD45.1 staining. Percentages of CD45.2 cells (derived from dko and WT control bone marrow cells), CD45.1 cells (derived from competing B6.SJL bone marrow cells), and CD45.1 /CD45.2 cells (derived from residual cells of the recipients) are shown. B, dko bone marrow cells in the presence of competing B6.SJL bone marrow cells had significantly reduced capability to develop into T cells in the periphery. In the whole body irradiation-bmtx model described in A, B220 B cells and CD3 T cells among CD45.2 cells (derived from dko or control WT bone marrow cells) in the spleen, lymph node, and blood were determined by flow cytometry 90 days later; percentages are shown. C, dko T cells presented failed homeostatic expansion in sublethally irradiated recipients. B6.SJL mice (CD45.1 ) were sublethally irradiated at 600 rads and transplanted i.v. with CFSE-labeled spleen cells from dko or WT control mice (CD45.2 ). The histograms show profiles of carboxyfluorescein succinimidyl ester (CFSE)-positive cells gated on CD4 CD45.2 and CD8 CD45.2 cells. Experiments in this figure were repeated more than three times, and representative histograms are shown. As shown in Fig. 5A, 63.9% of the thymocytes in the control recipients were derived from WT donor bone marrow cells (CD45.2 single positive cells; upper left panel), whereas in the test recipients only 0.4% thymocytes were derived from dko donor bone marrow cells (CD45.2 single positive cells; Fig. 5A, upper right panel). Similar significant differences were also found in the spleen (Fig. 5A, 2nd row), lymph nodes (3rd row), and blood (last row). This clearly indicates that the dko JOURNAL OF BIOLOGICAL CHEMISTRY bone marrow cells were significantly inferior to the WT control bone marrow cells in their capacity to compete with B6.SJL bone marrow cells to develop and expand in the void niche created by irradiation. As expected, the proportion of CD3 T cells in the spleen, lymph nodes, and blood derived from dko bone marrow cells was significantly lower than the proportion derived from control bone marrow cells (Fig. 5B). However, the percentage of B VOLUME 286 NUMBER 48 DECEMBER 2, 2011
13 FIGURE 6. Compromised in vivo anti-lcmv immune responses of dko mice. A, spleen cell number on day 8 following LCMV infection. Means S.D. of absolute cell number of total splenocytes, CD4 cells, and CD8 cells of control WT control (n 4) and dko (n 4) mice on day 8 post-lcmv infection are shown. B and C, virus-specific CD8 cells on day 8 post-lcmv infection. On day 8 post-infection, the percentage of gp33, np396, and gp276 tetramer-positive cells among CD8 cells (B) and the absolute number of gp33, np396, and gp276 tetramer-positive CD8 cells per spleen were measured by flow cytometry. Means S.D. of data from four pairs of WT control and dko mice are shown. D, virus-specific lymphokine-producing CD4 and CD8 cells on day 8 and day 32 post-lcmv infection. Percentage of IFN- -producing virus-specific CD4 cells (gp61-positive) and CD8 cells (gp33-positive) (left column), and percentage of TNF- -producing virus-specific CD4 cells (gp61-positive) and CD8 cells (gp33-positive) (right column) were measured by flow cytometry on day 8 (upper row) and day 32 (lower row) post-lcvm infection. Means S.D. of data from 4 to 6 pairs of WT control and dko mice are shown. E, cytotoxic T lymphocyte activity on day 8 post-lcmv infection. On day 8 post LCMV infection, virus-specific cytotoxic T lymphocyte activity was measured with 51 Cr-release assays at different effectors versus target cell ratios. Percentage of specific lysis was calculated based on ratios of spleen cells versus target cells. Means S.D. of data from WT control (n 4) and dko (n 3) mice are shown. A E, data from 3 to 4 mice were pooled and analyzed by one-way analysis of variance followed by Bonferroni s multiple comparison test. p values are indicated when they reached cells (B220-positive cells) derived from dko bone marrow cells was greater than that derived from control bone marrow, indicating a T cell-specific development defect due to the Lck-Crecaused deletion of both Enb1 and Enb2. In this system, Ephs of dko thymocytes and peripheral T cells should have sufficient forward Efnb1 and Efnb2 stimulation from competing WT B6.SJL bone marrow-derived cells, whose Efnb1 and Efnb2 expression was normal. Therefore, we could attribute the defective development and expansion of dko thymocytes mainly to the absence of both Efnb1 and Efnb2 reverse signaling. To understand whether mature dko T cells were incompetent in homeostatic expansion, we transferred CFSE-labeled dko or WT spleen cells into sublethally irradiated B6.SJL mice. As shown in Fig. 5C, dko donor T cells showed significantly lower homeostatic proliferation in vivo compared with WT control donor T cells. This compromised homeostatic proliferation of mature dko T cells along with their increased apoptosis and reduced thymocyte output as described above are likely all contributing factors resulting in the significant decrease of peripheral T cells in the dko mice. Efnb1 and Efnb2 Deletion in the T Cell Compartment Results in Compromised Immune Responses to LCMV Infection The reduced peripheral T cell population and defective T helper cell differentiation in dko mice strongly suggested that these mice might have incompetent immune responses in vivo. We therefore assessed their anti-lcmv immune response. Eight days after the mice were infected with LCMV (strain WE), there was a significant increase in the absolute number of total spleen cells in both dko and control mice (Fis. 6A versus 2B). The DECEMBER 2, 2011 VOLUME 286 NUMBER 48 JOURNAL OF BIOLOGICAL CHEMISTRY 41147
Supporting Information Table of Contents
 Supporting Information Table of Contents Supporting Information Figure 1 Page 2 Supporting Information Figure 2 Page 4 Supporting Information Figure 3 Page 5 Supporting Information Figure 4 Page 6 Supporting
Supporting Information Table of Contents Supporting Information Figure 1 Page 2 Supporting Information Figure 2 Page 4 Supporting Information Figure 3 Page 5 Supporting Information Figure 4 Page 6 Supporting
The autoimmune disease-associated PTPN22 variant promotes calpain-mediated Lyp/Pep
 SUPPLEMENTARY INFORMATION The autoimmune disease-associated PTPN22 variant promotes calpain-mediated Lyp/Pep degradation associated with lymphocyte and dendritic cell hyperresponsiveness Jinyi Zhang, Naima
SUPPLEMENTARY INFORMATION The autoimmune disease-associated PTPN22 variant promotes calpain-mediated Lyp/Pep degradation associated with lymphocyte and dendritic cell hyperresponsiveness Jinyi Zhang, Naima
Figure S1. Generation of inducible PTEN deficient mice and the BMMCs (A) B6.129 Pten loxp/loxp mice were mated with B6.
 Figure S1. Generation of inducible PTEN deficient mice and the BMMCs (A) B6.129 Pten loxp/loxp mice were mated with B6.129-Gt(ROSA)26Sor tm1(cre/ert2)tyj /J mice. To induce deletion of the Pten locus,
Figure S1. Generation of inducible PTEN deficient mice and the BMMCs (A) B6.129 Pten loxp/loxp mice were mated with B6.129-Gt(ROSA)26Sor tm1(cre/ert2)tyj /J mice. To induce deletion of the Pten locus,
Nature Immunology: doi: /ni Supplementary Figure 1. Cellularity of leukocytes and their progenitors in naive wild-type and Spp1 / mice.
 Supplementary Figure 1 Cellularity of leukocytes and their progenitors in naive wild-type and Spp1 / mice. (a, b) Gating strategies for differentiated cells including PMN (CD11b + Ly6G hi and CD11b + Ly6G
Supplementary Figure 1 Cellularity of leukocytes and their progenitors in naive wild-type and Spp1 / mice. (a, b) Gating strategies for differentiated cells including PMN (CD11b + Ly6G hi and CD11b + Ly6G
Supplemental Figure 1
 Supplemental Figure 1 1a 1c PD-1 MFI fold change 6 5 4 3 2 1 IL-1α IL-2 IL-4 IL-6 IL-1 IL-12 IL-13 IL-15 IL-17 IL-18 IL-21 IL-23 IFN-α Mut Human PD-1 promoter SBE-D 5 -GTCTG- -1.2kb SBE-P -CAGAC- -1.kb
Supplemental Figure 1 1a 1c PD-1 MFI fold change 6 5 4 3 2 1 IL-1α IL-2 IL-4 IL-6 IL-1 IL-12 IL-13 IL-15 IL-17 IL-18 IL-21 IL-23 IFN-α Mut Human PD-1 promoter SBE-D 5 -GTCTG- -1.2kb SBE-P -CAGAC- -1.kb
Cell isolation. Spleen and lymph nodes (axillary, inguinal) were removed from mice
 Supplementary Methods: Cell isolation. Spleen and lymph nodes (axillary, inguinal) were removed from mice and gently meshed in DMEM containing 10% FBS to prepare for single cell suspensions. CD4 + CD25
Supplementary Methods: Cell isolation. Spleen and lymph nodes (axillary, inguinal) were removed from mice and gently meshed in DMEM containing 10% FBS to prepare for single cell suspensions. CD4 + CD25
MATERIALS AND METHODS. Neutralizing antibodies specific to mouse Dll1, Dll4, J1 and J2 were prepared as described. 1,2 All
 MATERIALS AND METHODS Antibodies (Abs), flow cytometry analysis and cell lines Neutralizing antibodies specific to mouse Dll1, Dll4, J1 and J2 were prepared as described. 1,2 All other antibodies used
MATERIALS AND METHODS Antibodies (Abs), flow cytometry analysis and cell lines Neutralizing antibodies specific to mouse Dll1, Dll4, J1 and J2 were prepared as described. 1,2 All other antibodies used
Supplemental Figure 1. Signature gene expression in in vitro differentiated Th0, Th1, Th2, Th17 and Treg cells. (A) Naïve CD4 + T cells were cultured
 Supplemental Figure 1. Signature gene expression in in vitro differentiated Th0, Th1, Th2, Th17 and Treg cells. (A) Naïve CD4 + T cells were cultured under Th0, Th1, Th2, Th17, and Treg conditions. mrna
Supplemental Figure 1. Signature gene expression in in vitro differentiated Th0, Th1, Th2, Th17 and Treg cells. (A) Naïve CD4 + T cells were cultured under Th0, Th1, Th2, Th17, and Treg conditions. mrna
Supplementary Figure 1. Generation of knockin mice expressing L-selectinN138G. (a) Schematics of the Sellg allele (top), the targeting vector, the
 Supplementary Figure 1. Generation of knockin mice expressing L-selectinN138G. (a) Schematics of the Sellg allele (top), the targeting vector, the targeted allele in ES cells, and the mutant allele in
Supplementary Figure 1. Generation of knockin mice expressing L-selectinN138G. (a) Schematics of the Sellg allele (top), the targeting vector, the targeted allele in ES cells, and the mutant allele in
Supplementary Materials for
 www.sciencesignaling.org/cgi/content/full/3/114/ra23/dc1 Supplementary Materials for Regulation of Zap70 Expression During Thymocyte Development Enables Temporal Separation of CD4 and CD8 Repertoire Selection
www.sciencesignaling.org/cgi/content/full/3/114/ra23/dc1 Supplementary Materials for Regulation of Zap70 Expression During Thymocyte Development Enables Temporal Separation of CD4 and CD8 Repertoire Selection
Supplementary Figure S1. PTPN2 levels are not altered in proliferating CD8+ T cells. Lymph node (LN) CD8+ T cells from C57BL/6 mice were stained with
 Supplementary Figure S1. PTPN2 levels are not altered in proliferating CD8+ T cells. Lymph node (LN) CD8+ T cells from C57BL/6 mice were stained with CFSE and stimulated with plate-bound α-cd3ε (10µg/ml)
Supplementary Figure S1. PTPN2 levels are not altered in proliferating CD8+ T cells. Lymph node (LN) CD8+ T cells from C57BL/6 mice were stained with CFSE and stimulated with plate-bound α-cd3ε (10µg/ml)
Nature Immunology: doi: /ni Supplementary Figure 1. Huwe1 has high expression in HSCs and is necessary for quiescence.
 Supplementary Figure 1 Huwe1 has high expression in HSCs and is necessary for quiescence. (a) Heat map visualizing expression of genes with a known function in ubiquitin-mediated proteolysis (KEGG: Ubiquitin
Supplementary Figure 1 Huwe1 has high expression in HSCs and is necessary for quiescence. (a) Heat map visualizing expression of genes with a known function in ubiquitin-mediated proteolysis (KEGG: Ubiquitin
Supplementary Table; Supplementary Figures and legends S1-S21; Supplementary Materials and Methods
 Silva et al. PTEN posttranslational inactivation and hyperactivation of the PI3K/Akt pathway sustain primary T cell leukemia viability Supplementary Table; Supplementary Figures and legends S1-S21; Supplementary
Silva et al. PTEN posttranslational inactivation and hyperactivation of the PI3K/Akt pathway sustain primary T cell leukemia viability Supplementary Table; Supplementary Figures and legends S1-S21; Supplementary
Supplemental Table I.
 Supplemental Table I Male / Mean ± SEM n Mean ± SEM n Body weight, g 29.2±0.4 17 29.7±0.5 17 Total cholesterol, mg/dl 534.0±30.8 17 561.6±26.1 17 HDL-cholesterol, mg/dl 9.6±0.8 17 10.1±0.7 17 Triglycerides,
Supplemental Table I Male / Mean ± SEM n Mean ± SEM n Body weight, g 29.2±0.4 17 29.7±0.5 17 Total cholesterol, mg/dl 534.0±30.8 17 561.6±26.1 17 HDL-cholesterol, mg/dl 9.6±0.8 17 10.1±0.7 17 Triglycerides,
and follicular helper T cells is Egr2-dependent. (a) Diagrammatic representation of the
 Supplementary Figure 1. LAG3 + Treg-mediated regulation of germinal center B cells and follicular helper T cells is Egr2-dependent. (a) Diagrammatic representation of the experimental protocol for the
Supplementary Figure 1. LAG3 + Treg-mediated regulation of germinal center B cells and follicular helper T cells is Egr2-dependent. (a) Diagrammatic representation of the experimental protocol for the
Nature Immunology: doi: /ni Supplementary Figure 1. Gene expression profile of CD4 + T cells and CTL responses in Bcl6-deficient mice.
 Supplementary Figure 1 Gene expression profile of CD4 + T cells and CTL responses in Bcl6-deficient mice. (a) Gene expression profile in the resting CD4 + T cells were analyzed by an Affymetrix microarray
Supplementary Figure 1 Gene expression profile of CD4 + T cells and CTL responses in Bcl6-deficient mice. (a) Gene expression profile in the resting CD4 + T cells were analyzed by an Affymetrix microarray
ECM1 controls T H 2 cell egress from lymph nodes through re-expression of S1P 1
 ZH, Li et al, page 1 ECM1 controls T H 2 cell egress from lymph nodes through re-expression of S1P 1 Zhenhu Li 1,4,Yuan Zhang 1,4, Zhiduo Liu 1, Xiaodong Wu 1, Yuhan Zheng 1, Zhiyun Tao 1, Kairui Mao 1,
ZH, Li et al, page 1 ECM1 controls T H 2 cell egress from lymph nodes through re-expression of S1P 1 Zhenhu Li 1,4,Yuan Zhang 1,4, Zhiduo Liu 1, Xiaodong Wu 1, Yuhan Zheng 1, Zhiyun Tao 1, Kairui Mao 1,
Supplementary Figure 1. Efficiency of Mll4 deletion and its effect on T cell populations in the periphery. Nature Immunology: doi: /ni.
 Supplementary Figure 1 Efficiency of Mll4 deletion and its effect on T cell populations in the periphery. Expression of Mll4 floxed alleles (16-19) in naive CD4 + T cells isolated from lymph nodes and
Supplementary Figure 1 Efficiency of Mll4 deletion and its effect on T cell populations in the periphery. Expression of Mll4 floxed alleles (16-19) in naive CD4 + T cells isolated from lymph nodes and
Supplemental Information. T Cells Enhance Autoimmunity by Restraining Regulatory T Cell Responses via an Interleukin-23-Dependent Mechanism
 Immunity, Volume 33 Supplemental Information T Cells Enhance Autoimmunity by Restraining Regulatory T Cell Responses via an Interleukin-23-Dependent Mechanism Franziska Petermann, Veit Rothhammer, Malte
Immunity, Volume 33 Supplemental Information T Cells Enhance Autoimmunity by Restraining Regulatory T Cell Responses via an Interleukin-23-Dependent Mechanism Franziska Petermann, Veit Rothhammer, Malte
SUPPLEMENTARY INFORMATION
 Supplemental Figure 1. Furin is efficiently deleted in CD4 + and CD8 + T cells. a, Western blot for furin and actin proteins in CD4cre-fur f/f and fur f/f Th1 cells. Wild-type and furin-deficient CD4 +
Supplemental Figure 1. Furin is efficiently deleted in CD4 + and CD8 + T cells. a, Western blot for furin and actin proteins in CD4cre-fur f/f and fur f/f Th1 cells. Wild-type and furin-deficient CD4 +
Supplementary Figure 1 CD4 + T cells from PKC-θ null mice are defective in NF-κB activation during T cell receptor signaling. CD4 + T cells were
 Supplementary Figure 1 CD4 + T cells from PKC-θ null mice are defective in NF-κB activation during T cell receptor signaling. CD4 + T cells were isolated from wild type (PKC-θ- WT) or PKC-θ null (PKC-θ-KO)
Supplementary Figure 1 CD4 + T cells from PKC-θ null mice are defective in NF-κB activation during T cell receptor signaling. CD4 + T cells were isolated from wild type (PKC-θ- WT) or PKC-θ null (PKC-θ-KO)
Supplementary Figures. T Cell Factor-1 initiates T helper 2 fate by inducing GATA-3 and repressing Interferon-γ
 Supplementary Figures T Cell Factor-1 initiates T helper 2 fate by inducing GATA-3 and repressing Interferon-γ Qing Yu, Archna Sharma, Sun Young Oh, Hyung-Geun Moon, M. Zulfiquer Hossain, Theresa M. Salay,
Supplementary Figures T Cell Factor-1 initiates T helper 2 fate by inducing GATA-3 and repressing Interferon-γ Qing Yu, Archna Sharma, Sun Young Oh, Hyung-Geun Moon, M. Zulfiquer Hossain, Theresa M. Salay,
Supplementary Figure 1. Normal T lymphocyte populations in Dapk -/- mice. (a) Normal thymic development in Dapk -/- mice. Thymocytes from WT and Dapk
 Supplementary Figure 1. Normal T lymphocyte populations in Dapk -/- mice. (a) Normal thymic development in Dapk -/- mice. Thymocytes from WT and Dapk -/- mice were stained for expression of CD4 and CD8.
Supplementary Figure 1. Normal T lymphocyte populations in Dapk -/- mice. (a) Normal thymic development in Dapk -/- mice. Thymocytes from WT and Dapk -/- mice were stained for expression of CD4 and CD8.
Blocking antibodies and peptides. Rat anti-mouse PD-1 (29F.1A12, rat IgG2a, k), PD-
 Supplementary Methods Blocking antibodies and peptides. Rat anti-mouse PD-1 (29F.1A12, rat IgG2a, k), PD- L1 (10F.9G2, rat IgG2b, k), and PD-L2 (3.2, mouse IgG1) have been described (24). Anti-CTLA-4 (clone
Supplementary Methods Blocking antibodies and peptides. Rat anti-mouse PD-1 (29F.1A12, rat IgG2a, k), PD- L1 (10F.9G2, rat IgG2b, k), and PD-L2 (3.2, mouse IgG1) have been described (24). Anti-CTLA-4 (clone
SUPPLEMENTARY INFORMATION
 doi: 1.138/nature775 4 O.D. (595-655) 3 1 -ζ no antibody isotype ctrl Plated Soluble 1F6 397 7H11 Supplementary Figure 1 Soluble and plated anti- Abs induce -! signalling. B3Z cells stably expressing!
doi: 1.138/nature775 4 O.D. (595-655) 3 1 -ζ no antibody isotype ctrl Plated Soluble 1F6 397 7H11 Supplementary Figure 1 Soluble and plated anti- Abs induce -! signalling. B3Z cells stably expressing!
Optimizing Intracellular Flow Cytometry:
 Optimizing Intracellular Flow Cytometry: Simultaneous Detection of Cytokines and Transcription Factors An encore presentation by Jurg Rohrer, PhD, BD Biosciences 10.26.10 Outline Introduction Cytokines
Optimizing Intracellular Flow Cytometry: Simultaneous Detection of Cytokines and Transcription Factors An encore presentation by Jurg Rohrer, PhD, BD Biosciences 10.26.10 Outline Introduction Cytokines
Supplementary Figure 1 IL-27 IL
 Tim-3 Supplementary Figure 1 Tc0 49.5 0.6 Tc1 63.5 0.84 Un 49.8 0.16 35.5 0.16 10 4 61.2 5.53 10 3 64.5 5.66 10 2 10 1 10 0 31 2.22 10 0 10 1 10 2 10 3 10 4 IL-10 28.2 1.69 IL-27 Supplementary Figure 1.
Tim-3 Supplementary Figure 1 Tc0 49.5 0.6 Tc1 63.5 0.84 Un 49.8 0.16 35.5 0.16 10 4 61.2 5.53 10 3 64.5 5.66 10 2 10 1 10 0 31 2.22 10 0 10 1 10 2 10 3 10 4 IL-10 28.2 1.69 IL-27 Supplementary Figure 1.
Supplementary Materials for
 immunology.sciencemag.org/cgi/content/full/2/16/eaan6049/dc1 Supplementary Materials for Enzymatic synthesis of core 2 O-glycans governs the tissue-trafficking potential of memory CD8 + T cells Jossef
immunology.sciencemag.org/cgi/content/full/2/16/eaan6049/dc1 Supplementary Materials for Enzymatic synthesis of core 2 O-glycans governs the tissue-trafficking potential of memory CD8 + T cells Jossef
Islet viability assay and Glucose Stimulated Insulin Secretion assay RT-PCR and Western Blot
 Islet viability assay and Glucose Stimulated Insulin Secretion assay Islet cell viability was determined by colorimetric (3-(4,5-dimethylthiazol-2-yl)-2,5- diphenyltetrazolium bromide assay using CellTiter
Islet viability assay and Glucose Stimulated Insulin Secretion assay Islet cell viability was determined by colorimetric (3-(4,5-dimethylthiazol-2-yl)-2,5- diphenyltetrazolium bromide assay using CellTiter
SUPPLEMENTARY INFORMATION
 doi: 1.138/nature89 IFN- (ng ml ) 5 4 3 1 Splenocytes NS IFN- (ng ml ) 6 4 Lymph node cells NS Nfkbiz / Nfkbiz / Nfkbiz / Nfkbiz / IL- (ng ml ) 3 1 Splenocytes IL- (ng ml ) 1 8 6 4 *** ** Lymph node cells
doi: 1.138/nature89 IFN- (ng ml ) 5 4 3 1 Splenocytes NS IFN- (ng ml ) 6 4 Lymph node cells NS Nfkbiz / Nfkbiz / Nfkbiz / Nfkbiz / IL- (ng ml ) 3 1 Splenocytes IL- (ng ml ) 1 8 6 4 *** ** Lymph node cells
PBMC from each patient were suspended in AIM V medium (Invitrogen) with 5% human
 Anti-CD19-CAR transduced T-cell preparation PBMC from each patient were suspended in AIM V medium (Invitrogen) with 5% human AB serum (Gemini) and 300 international units/ml IL-2 (Novartis). T cell proliferation
Anti-CD19-CAR transduced T-cell preparation PBMC from each patient were suspended in AIM V medium (Invitrogen) with 5% human AB serum (Gemini) and 300 international units/ml IL-2 (Novartis). T cell proliferation
Examples of questions for Cellular Immunology/Cellular Biology and Immunology
 Examples of questions for Cellular Immunology/Cellular Biology and Immunology Each student gets a set of 6 questions, so that each set contains different types of questions and that the set of questions
Examples of questions for Cellular Immunology/Cellular Biology and Immunology Each student gets a set of 6 questions, so that each set contains different types of questions and that the set of questions
T cell development October 28, Dan Stetson
 T cell development October 28, 2016 Dan Stetson stetson@uw.edu 441 Lecture #13 Slide 1 of 29 Three lectures on T cells (Chapters 8, 9) Part 1 (Today): T cell development in the thymus Chapter 8, pages
T cell development October 28, 2016 Dan Stetson stetson@uw.edu 441 Lecture #13 Slide 1 of 29 Three lectures on T cells (Chapters 8, 9) Part 1 (Today): T cell development in the thymus Chapter 8, pages
Supporting Information
 Supporting Information Valkenburg et al. 10.1073/pnas.1403684111 SI Materials and Methods ELISA and Microneutralization. Sera were treated with Receptor Destroying Enzyme II (RDE II, Accurate) before ELISA
Supporting Information Valkenburg et al. 10.1073/pnas.1403684111 SI Materials and Methods ELISA and Microneutralization. Sera were treated with Receptor Destroying Enzyme II (RDE II, Accurate) before ELISA
SUPPLEMENTARY FIGURES
 SUPPLEMENTARY FIGURES Supplementary Figure 1: Chemokine receptor expression profiles of CCR6 + and CCR6 - CD4 + IL-17A +/ex and Treg cells. Quantitative PCR analysis of chemokine receptor transcript abundance
SUPPLEMENTARY FIGURES Supplementary Figure 1: Chemokine receptor expression profiles of CCR6 + and CCR6 - CD4 + IL-17A +/ex and Treg cells. Quantitative PCR analysis of chemokine receptor transcript abundance
Peli1 negatively regulates T-cell activation and prevents autoimmunity
 Peli1 negatively regulates T-cell activation and prevents autoimmunity Mikyoung Chang 1,*, Wei Jin 1,5,*, Jae-Hoon Chang 1, Yi-chuan Xiao 1, George Brittain 1, Jiayi Yu 1, Xiaofei Zhou 1, Yi-Hong Wang
Peli1 negatively regulates T-cell activation and prevents autoimmunity Mikyoung Chang 1,*, Wei Jin 1,5,*, Jae-Hoon Chang 1, Yi-chuan Xiao 1, George Brittain 1, Jiayi Yu 1, Xiaofei Zhou 1, Yi-Hong Wang
PTEN inhibits IL-2 receptor mediated expansion of CD4 + CD25 + Tregs
 Research article PTEN inhibits IL-2 receptor mediated expansion of CD4 + CD25 + Tregs Patrick T. Walsh, 1 Jodi L. Buckler, 1 Jidong Zhang, 1 Andrew E. Gelman, 1 Nicole M. Dalton, 1 Devon K. Taylor, 1 Steven
Research article PTEN inhibits IL-2 receptor mediated expansion of CD4 + CD25 + Tregs Patrick T. Walsh, 1 Jodi L. Buckler, 1 Jidong Zhang, 1 Andrew E. Gelman, 1 Nicole M. Dalton, 1 Devon K. Taylor, 1 Steven
Supplemental Figure 1. Activated splenocytes upregulate Serpina3g and Serpina3f expression.
 Relative Serpin expression 25 2 15 1 5 Serpina3f 1 2 3 4 5 6 8 6 4 2 Serpina3g 1 2 3 4 5 6 C57BL/6 DBA/2 Supplemental Figure 1. Activated splenocytes upregulate Serpina3g and Serpina3f expression. Splenocytes
Relative Serpin expression 25 2 15 1 5 Serpina3f 1 2 3 4 5 6 8 6 4 2 Serpina3g 1 2 3 4 5 6 C57BL/6 DBA/2 Supplemental Figure 1. Activated splenocytes upregulate Serpina3g and Serpina3f expression. Splenocytes
Nature Immunology: doi: /ni Supplementary Figure 1. DNA-methylation machinery is essential for silencing of Cd4 in cytotoxic T cells.
 Supplementary Figure 1 DNA-methylation machinery is essential for silencing of Cd4 in cytotoxic T cells. (a) Scheme for the retroviral shrna screen. (b) Histogram showing CD4 expression (MFI) in WT cytotoxic
Supplementary Figure 1 DNA-methylation machinery is essential for silencing of Cd4 in cytotoxic T cells. (a) Scheme for the retroviral shrna screen. (b) Histogram showing CD4 expression (MFI) in WT cytotoxic
Supporting Online Material for
 www.sciencemag.org/cgi/content/full/1175194/dc1 Supporting Online Material for A Vital Role for Interleukin-21 in the Control of a Chronic Viral Infection John S. Yi, Ming Du, Allan J. Zajac* *To whom
www.sciencemag.org/cgi/content/full/1175194/dc1 Supporting Online Material for A Vital Role for Interleukin-21 in the Control of a Chronic Viral Infection John S. Yi, Ming Du, Allan J. Zajac* *To whom
Effector mechanisms of cell-mediated immunity: Properties of effector, memory and regulatory T cells
 ICI Basic Immunology course Effector mechanisms of cell-mediated immunity: Properties of effector, memory and regulatory T cells Abul K. Abbas, MD UCSF Stages in the development of T cell responses: induction
ICI Basic Immunology course Effector mechanisms of cell-mediated immunity: Properties of effector, memory and regulatory T cells Abul K. Abbas, MD UCSF Stages in the development of T cell responses: induction
T-cell activation T cells migrate to secondary lymphoid tissues where they interact with antigen, antigen-presenting cells, and other lymphocytes:
 Interactions between innate immunity & adaptive immunity What happens to T cells after they leave the thymus? Naïve T cells exit the thymus and enter the bloodstream. If they remain in the bloodstream,
Interactions between innate immunity & adaptive immunity What happens to T cells after they leave the thymus? Naïve T cells exit the thymus and enter the bloodstream. If they remain in the bloodstream,
T-cell activation T cells migrate to secondary lymphoid tissues where they interact with antigen, antigen-presenting cells, and other lymphocytes:
 Interactions between innate immunity & adaptive immunity What happens to T cells after they leave the thymus? Naïve T cells exit the thymus and enter the bloodstream. If they remain in the bloodstream,
Interactions between innate immunity & adaptive immunity What happens to T cells after they leave the thymus? Naïve T cells exit the thymus and enter the bloodstream. If they remain in the bloodstream,
SUPPLEMENTARY INFORMATION
 Complete but curtailed T-cell response to very-low-affinity antigen Dietmar Zehn, Sarah Y. Lee & Michael J. Bevan Supp. Fig. 1: TCR chain usage among endogenous K b /Ova reactive T cells. C57BL/6 mice
Complete but curtailed T-cell response to very-low-affinity antigen Dietmar Zehn, Sarah Y. Lee & Michael J. Bevan Supp. Fig. 1: TCR chain usage among endogenous K b /Ova reactive T cells. C57BL/6 mice
Supplementary Information
 Supplementary Information Supplementary Figure 1! a! b! Nfatc1!! Nfatc1"! P1! P2! pa1! pa2! ex1! ex2! exons 3-9! ex1! ex11!!" #" Nfatc1A!!" Nfatc1B! #"!" Nfatc1C! #" DN1! DN2! DN1!!A! #A!!B! #B!!C! #C!!A!
Supplementary Information Supplementary Figure 1! a! b! Nfatc1!! Nfatc1"! P1! P2! pa1! pa2! ex1! ex2! exons 3-9! ex1! ex11!!" #" Nfatc1A!!" Nfatc1B! #"!" Nfatc1C! #" DN1! DN2! DN1!!A! #A!!B! #B!!C! #C!!A!
Commercially available HLA Class II tetramers (Beckman Coulter) conjugated to
 Class II tetramer staining Commercially available HLA Class II tetramers (Beckman Coulter) conjugated to PE were combined with dominant HIV epitopes (DRB1*0101-DRFYKTLRAEQASQEV, DRB1*0301- PEKEVLVWKFDSRLAFHH,
Class II tetramer staining Commercially available HLA Class II tetramers (Beckman Coulter) conjugated to PE were combined with dominant HIV epitopes (DRB1*0101-DRFYKTLRAEQASQEV, DRB1*0301- PEKEVLVWKFDSRLAFHH,
Optimizing Intracellular Flow Cytometry:
 Optimizing Intracellular Flow Cytometry: Simultaneous Detection of Cytokines and Transcription Factors Presented by Jurg Rohrer, PhD, BD Biosciences 23-10780-00 Outline Introduction Cytokines Transcription
Optimizing Intracellular Flow Cytometry: Simultaneous Detection of Cytokines and Transcription Factors Presented by Jurg Rohrer, PhD, BD Biosciences 23-10780-00 Outline Introduction Cytokines Transcription
Eosinophils are required. for the maintenance of plasma cells in the bone marrow
 Eosinophils are required for the maintenance of plasma cells in the bone marrow Van Trung Chu, Anja Fröhlich, Gudrun Steinhauser, Tobias Scheel, Toralf Roch, Simon Fillatreau, James J. Lee, Max Löhning
Eosinophils are required for the maintenance of plasma cells in the bone marrow Van Trung Chu, Anja Fröhlich, Gudrun Steinhauser, Tobias Scheel, Toralf Roch, Simon Fillatreau, James J. Lee, Max Löhning
Supplementary Figure Legends. group) and analyzed for Siglec-G expression utilizing a monoclonal antibody to Siglec-G (clone SH2.1).
 Supplementary Figure Legends Supplemental Figure : Naïve T cells express Siglec-G. Splenocytes were isolated from WT B or Siglec-G -/- animals that have not been transplanted (n= per group) and analyzed
Supplementary Figure Legends Supplemental Figure : Naïve T cells express Siglec-G. Splenocytes were isolated from WT B or Siglec-G -/- animals that have not been transplanted (n= per group) and analyzed
The encephalitogenicity of TH17 cells is dependent on IL-1- and IL-23- induced production of the cytokine GM-CSF
 CORRECTION NOTICE Nat.Immunol. 12, 568 575 (2011) The encephalitogenicity of TH17 cells is dependent on IL-1- and IL-23- induced production of the cytokine GM-CSF Mohamed El-Behi, Bogoljub Ciric, Hong
CORRECTION NOTICE Nat.Immunol. 12, 568 575 (2011) The encephalitogenicity of TH17 cells is dependent on IL-1- and IL-23- induced production of the cytokine GM-CSF Mohamed El-Behi, Bogoljub Ciric, Hong
Supplementary Figure 1. Characterization of basophils after reconstitution of SCID mice
 Supplementary figure legends Supplementary Figure 1. Characterization of after reconstitution of SCID mice with CD4 + CD62L + T cells. (A-C) SCID mice (n = 6 / group) were reconstituted with 2 x 1 6 CD4
Supplementary figure legends Supplementary Figure 1. Characterization of after reconstitution of SCID mice with CD4 + CD62L + T cells. (A-C) SCID mice (n = 6 / group) were reconstituted with 2 x 1 6 CD4
T cell protein tyrosine phosphatase attenuates T cell signaling to maintain tolerance in mice
 SUPPLEMENTAL METHODS T cell protein tyrosine phosphatase attenuates T cell signaling to maintain tolerance in mice Florian Wiede 1, Benjamin J. Shields 1, Sock Hui Chew 1, Konstantinos Kyparissoudis 2,
SUPPLEMENTAL METHODS T cell protein tyrosine phosphatase attenuates T cell signaling to maintain tolerance in mice Florian Wiede 1, Benjamin J. Shields 1, Sock Hui Chew 1, Konstantinos Kyparissoudis 2,
Cover Page. The handle holds various files of this Leiden University dissertation.
 Cover Page The handle http://hdl.handle.net/1887/23854 holds various files of this Leiden University dissertation. Author: Marel, Sander van der Title: Gene and cell therapy based treatment strategies
Cover Page The handle http://hdl.handle.net/1887/23854 holds various files of this Leiden University dissertation. Author: Marel, Sander van der Title: Gene and cell therapy based treatment strategies
Tbk1-TKO! DN cells (%)! 15! 10!
 a! T Cells! TKO! B Cells! TKO! b! CD4! 8.9 85.2 3.4 2.88 CD8! Tbk1-TKO! 1.1 84.8 2.51 2.54 c! DN cells (%)! 4 3 2 1 DP cells (%)! 9 8 7 6 CD4 + SP cells (%)! 5 4 3 2 1 5 TKO! TKO! TKO! TKO! 15 1 5 CD8
a! T Cells! TKO! B Cells! TKO! b! CD4! 8.9 85.2 3.4 2.88 CD8! Tbk1-TKO! 1.1 84.8 2.51 2.54 c! DN cells (%)! 4 3 2 1 DP cells (%)! 9 8 7 6 CD4 + SP cells (%)! 5 4 3 2 1 5 TKO! TKO! TKO! TKO! 15 1 5 CD8
T Cell Development. Xuefang Cao, MD, PhD. November 3, 2015
 T Cell Development Xuefang Cao, MD, PhD November 3, 2015 Thymocytes in the cortex of the thymus Early thymocytes development Positive and negative selection Lineage commitment Exit from the thymus and
T Cell Development Xuefang Cao, MD, PhD November 3, 2015 Thymocytes in the cortex of the thymus Early thymocytes development Positive and negative selection Lineage commitment Exit from the thymus and
Supplemental Materials
 Supplemental Materials Programmed death one homolog maintains the pool size of regulatory T cells by promoting their differentiation and stability Qi Wang 1, Jianwei He 1, Dallas B. Flies 2, Liqun Luo
Supplemental Materials Programmed death one homolog maintains the pool size of regulatory T cells by promoting their differentiation and stability Qi Wang 1, Jianwei He 1, Dallas B. Flies 2, Liqun Luo
SUPPLEMENTARY INFORMATION
 doi:10.1038/nature11095 Supplementary Table 1. Summary of the binding between Angptls and various Igdomain containing receptors as determined by flow cytometry analysis. The results were summarized from
doi:10.1038/nature11095 Supplementary Table 1. Summary of the binding between Angptls and various Igdomain containing receptors as determined by flow cytometry analysis. The results were summarized from
Supplemental Materials for. Effects of sphingosine-1-phosphate receptor 1 phosphorylation in response to. FTY720 during neuroinflammation
 Supplemental Materials for Effects of sphingosine-1-phosphate receptor 1 phosphorylation in response to FTY7 during neuroinflammation This file includes: Supplemental Table 1. EAE clinical parameters of
Supplemental Materials for Effects of sphingosine-1-phosphate receptor 1 phosphorylation in response to FTY7 during neuroinflammation This file includes: Supplemental Table 1. EAE clinical parameters of
Development of B and T lymphocytes
 Development of B and T lymphocytes What will we discuss today? B-cell development T-cell development B- cell development overview Stem cell In periphery Pro-B cell Pre-B cell Immature B cell Mature B cell
Development of B and T lymphocytes What will we discuss today? B-cell development T-cell development B- cell development overview Stem cell In periphery Pro-B cell Pre-B cell Immature B cell Mature B cell
The number of peripheral T cells is always maintained at a
 The Journal of Immunology MEKK3 Is Essential for Lymphopenia-Induced T Cell Proliferation and Survival 1 Xiaofang Wang,* Xing Chang,* Valeria Facchinetti, Yuan Zhuang, and ing Su 2 * T cell homeostasis
The Journal of Immunology MEKK3 Is Essential for Lymphopenia-Induced T Cell Proliferation and Survival 1 Xiaofang Wang,* Xing Chang,* Valeria Facchinetti, Yuan Zhuang, and ing Su 2 * T cell homeostasis
SUPPLEMENTARY FIGURE 1
 SUPPLEMENTARY FIGURE 1 A LN Cell count (1 ) 1 3 1 CD+ 1 1 CDL lo CD hi 1 CD+FoxP3+ 1 1 1 7 3 3 3 % of cells 9 7 7 % of cells CD+ 3 1 % of cells CDL lo CD hi 1 1 % of CD+ cells CD+FoxP3+ 3 1 % of CD+ T
SUPPLEMENTARY FIGURE 1 A LN Cell count (1 ) 1 3 1 CD+ 1 1 CDL lo CD hi 1 CD+FoxP3+ 1 1 1 7 3 3 3 % of cells 9 7 7 % of cells CD+ 3 1 % of cells CDL lo CD hi 1 1 % of CD+ cells CD+FoxP3+ 3 1 % of CD+ T
Canberra, Australia). CD11c-DTR-OVA-GFP (B6.CD11c-OVA), B6.luc + and. Cancer Research Center, Germany). B6 or BALB/c.FoxP3-DTR-GFP mice were
 Supplemental Materials and Methods Mice Female C57BL/6 (B6, I-E null, H-2 b ), BALB/c (H-2 d ) + ), FVB/N (H-2 q, I-E null, CD45.1 + ), and B6D2F1 (H-2 b/d ) mice were purchased from the Animal Resources
Supplemental Materials and Methods Mice Female C57BL/6 (B6, I-E null, H-2 b ), BALB/c (H-2 d ) + ), FVB/N (H-2 q, I-E null, CD45.1 + ), and B6D2F1 (H-2 b/d ) mice were purchased from the Animal Resources
of whole cell cultures in U-bottomed wells of a 96-well plate are shown. 2
 Supplementary online material Supplementary figure legends Supplementary Figure 1 Exposure to T reg cells causes loss of T resp cells in co-cultures. T resp cells were stimulated with CD3+CD28 alone or
Supplementary online material Supplementary figure legends Supplementary Figure 1 Exposure to T reg cells causes loss of T resp cells in co-cultures. T resp cells were stimulated with CD3+CD28 alone or
Supplemental Information. Otic Mesenchyme Cells Regulate. Spiral Ganglion Axon Fasciculation. through a Pou3f4/EphA4 Signaling Pathway
 Neuron, Volume 73 Supplemental Information Otic Mesenchyme Cells Regulate Spiral Ganglion Axon Fasciculation through a Pou3f4/EphA4 Signaling Pathway Thomas M. Coate, Steven Raft, Xiumei Zhao, Aimee K.
Neuron, Volume 73 Supplemental Information Otic Mesenchyme Cells Regulate Spiral Ganglion Axon Fasciculation through a Pou3f4/EphA4 Signaling Pathway Thomas M. Coate, Steven Raft, Xiumei Zhao, Aimee K.
Optimizing Intracellular Flow Cytometry
 Optimizing Intracellular Flow Cytometry Detection of Cytokines, Transcription Factors, and Phosphoprotein by Flow Cytometry Presented by Erika O Donnell, PhD, BD Biosciences 23-14876-00 Outline Basic principles
Optimizing Intracellular Flow Cytometry Detection of Cytokines, Transcription Factors, and Phosphoprotein by Flow Cytometry Presented by Erika O Donnell, PhD, BD Biosciences 23-14876-00 Outline Basic principles
SUPPORTING INFORMATIONS
 SUPPORTING INFORMATIONS Mice MT/ret RetCD3ε KO α-cd25 treated MT/ret Age 1 month 3 mnths 6 months 1 month 3 months 6 months 1 month 3 months 6 months 2/87 Survival 87/87 incidence of 17/87 1 ary tumor
SUPPORTING INFORMATIONS Mice MT/ret RetCD3ε KO α-cd25 treated MT/ret Age 1 month 3 mnths 6 months 1 month 3 months 6 months 1 month 3 months 6 months 2/87 Survival 87/87 incidence of 17/87 1 ary tumor
Supplementary Data Table of Contents:
 Supplementary Data Table of Contents: - Supplementary Methods - Supplementary Figures S1(A-B) - Supplementary Figures S2 (A-B) - Supplementary Figures S3 - Supplementary Figures S4(A-B) - Supplementary
Supplementary Data Table of Contents: - Supplementary Methods - Supplementary Figures S1(A-B) - Supplementary Figures S2 (A-B) - Supplementary Figures S3 - Supplementary Figures S4(A-B) - Supplementary
B6/COLODR/SPL/11C/83/LAP/#2.006 B6/COLODR/SPL/11C/86/LAP/#2.016 CD11C B6/COLODR/SPL/11C/80/LAP/#2.011 CD11C
 CD3-specific antibody-induced immune tolerance and suppression of autoimmune encephalomyelitis involves TGF-β production through phagocytes digesting apoptotic T cells Sylvain Perruche 1,3, Pin Zhang 1,
CD3-specific antibody-induced immune tolerance and suppression of autoimmune encephalomyelitis involves TGF-β production through phagocytes digesting apoptotic T cells Sylvain Perruche 1,3, Pin Zhang 1,
B220 CD4 CD8. Figure 1. Confocal Image of Sensitized HLN. Representative image of a sensitized HLN
 B220 CD4 CD8 Natarajan et al., unpublished data Figure 1. Confocal Image of Sensitized HLN. Representative image of a sensitized HLN showing B cell follicles and T cell areas. 20 µm thick. Image of magnification
B220 CD4 CD8 Natarajan et al., unpublished data Figure 1. Confocal Image of Sensitized HLN. Representative image of a sensitized HLN showing B cell follicles and T cell areas. 20 µm thick. Image of magnification
Immunology - Lecture 2 Adaptive Immune System 1
 Immunology - Lecture 2 Adaptive Immune System 1 Book chapters: Molecules of the Adaptive Immunity 6 Adaptive Cells and Organs 7 Generation of Immune Diversity Lymphocyte Antigen Receptors - 8 CD markers
Immunology - Lecture 2 Adaptive Immune System 1 Book chapters: Molecules of the Adaptive Immunity 6 Adaptive Cells and Organs 7 Generation of Immune Diversity Lymphocyte Antigen Receptors - 8 CD markers
Supplementary Figures
 MiR-29 controls innate and adaptive immune responses against intracellular bacterial infection by targeting IFN-γ Feng Ma 1,2,5, Sheng Xu 1,5, Xingguang Liu 1, Qian Zhang 1, Xiongfei Xu 1, Mofang Liu 3,
MiR-29 controls innate and adaptive immune responses against intracellular bacterial infection by targeting IFN-γ Feng Ma 1,2,5, Sheng Xu 1,5, Xingguang Liu 1, Qian Zhang 1, Xiongfei Xu 1, Mofang Liu 3,
Supplementary Figures
 Inhibition of Pulmonary Anti Bacterial Defense by IFN γ During Recovery from Influenza Infection By Keer Sun and Dennis W. Metzger Supplementary Figures d a Ly6G Percentage survival f 1 75 5 1 25 1 5 1
Inhibition of Pulmonary Anti Bacterial Defense by IFN γ During Recovery from Influenza Infection By Keer Sun and Dennis W. Metzger Supplementary Figures d a Ly6G Percentage survival f 1 75 5 1 25 1 5 1
Effector T Cells and
 1 Effector T Cells and Cytokines Andrew Lichtman, MD PhD Brigham and Women's Hospital Harvard Medical School 2 Lecture outline Cytokines Subsets of CD4+ T cells: definitions, functions, development New
1 Effector T Cells and Cytokines Andrew Lichtman, MD PhD Brigham and Women's Hospital Harvard Medical School 2 Lecture outline Cytokines Subsets of CD4+ T cells: definitions, functions, development New
Supporting Information
 Supporting Information lpek et al. 1.173/pnas.1121217 SI Materials and Methods Mice. cell knockout, inos / (Taconic arms), Rag1 /, INγR /, and IL-12p4 / mice (The Jackson Laboratory) were maintained and/or
Supporting Information lpek et al. 1.173/pnas.1121217 SI Materials and Methods Mice. cell knockout, inos / (Taconic arms), Rag1 /, INγR /, and IL-12p4 / mice (The Jackson Laboratory) were maintained and/or
W/T Itgam -/- F4/80 CD115. F4/80 hi CD115 + F4/80 + CD115 +
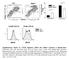 F4/8 % in the peritoneal lavage 6 4 2 p=.15 n.s p=.76 CD115 F4/8 hi CD115 + F4/8 + CD115 + F4/8 hi CD115 + F4/8 + CD115 + MHCII MHCII Supplementary Figure S1. CD11b deficiency affects the cellular responses
F4/8 % in the peritoneal lavage 6 4 2 p=.15 n.s p=.76 CD115 F4/8 hi CD115 + F4/8 + CD115 + F4/8 hi CD115 + F4/8 + CD115 + MHCII MHCII Supplementary Figure S1. CD11b deficiency affects the cellular responses
IMMUNOLOGICAL MEMORY. CD4 T Follicular Helper Cells. Memory CD8 T Cell Differentiation
 IMMUNOLOGICAL MEMORY CD4 T Follicular Helper Cells Memory CD8 T Cell Differentiation CD4 T Cell Differentiation Bcl-6 T-bet GATA-3 ROR t Foxp3 CD4 T follicular helper (Tfh) cells FUNCTION Provide essential
IMMUNOLOGICAL MEMORY CD4 T Follicular Helper Cells Memory CD8 T Cell Differentiation CD4 T Cell Differentiation Bcl-6 T-bet GATA-3 ROR t Foxp3 CD4 T follicular helper (Tfh) cells FUNCTION Provide essential
B6.SJL (Ly5.2) mice were obtained from Taconic Farms. Rag1-deficient mice were
 Supplementary Methods Mice. B6.SJL (Ly5.2) mice were obtained from Taconic Farms. Rag1-deficient mice were purchased from The Jackson Laboratory. Real-time PCR Total cellular RNA was extracted from the
Supplementary Methods Mice. B6.SJL (Ly5.2) mice were obtained from Taconic Farms. Rag1-deficient mice were purchased from The Jackson Laboratory. Real-time PCR Total cellular RNA was extracted from the
Akt and mtor pathways differentially regulate the development of natural and inducible. T H 17 cells
 Akt and mtor pathways differentially regulate the development of natural and inducible T H 17 cells Jiyeon S Kim, Tammarah Sklarz, Lauren Banks, Mercy Gohil, Adam T Waickman, Nicolas Skuli, Bryan L Krock,
Akt and mtor pathways differentially regulate the development of natural and inducible T H 17 cells Jiyeon S Kim, Tammarah Sklarz, Lauren Banks, Mercy Gohil, Adam T Waickman, Nicolas Skuli, Bryan L Krock,
Supplementary Information:
 Supplementary Information: Follicular regulatory T cells with Bcl6 expression suppress germinal center reactions by Yeonseok Chung, Shinya Tanaka, Fuliang Chu, Roza Nurieva, Gustavo J. Martinez, Seema
Supplementary Information: Follicular regulatory T cells with Bcl6 expression suppress germinal center reactions by Yeonseok Chung, Shinya Tanaka, Fuliang Chu, Roza Nurieva, Gustavo J. Martinez, Seema
Test Bank for Basic Immunology Functions and Disorders of the Immune System 4th Edition by Abbas
 Test Bank for Basic Immunology Functions and Disorders of the Immune System 4th Edition by Abbas Chapter 04: Antigen Recognition in the Adaptive Immune System Test Bank MULTIPLE CHOICE 1. Most T lymphocytes
Test Bank for Basic Immunology Functions and Disorders of the Immune System 4th Edition by Abbas Chapter 04: Antigen Recognition in the Adaptive Immune System Test Bank MULTIPLE CHOICE 1. Most T lymphocytes
Development of all CD4 T lineages requires nuclear factor TOX
 ARTICLE Development of all CD4 T lineages requires nuclear factor TOX Parinaz Aliahmad and Jonathan Kaye Department of Immunology, The Scripps Research Institute, La Jolla, CA 92037 The Journal of Experimental
ARTICLE Development of all CD4 T lineages requires nuclear factor TOX Parinaz Aliahmad and Jonathan Kaye Department of Immunology, The Scripps Research Institute, La Jolla, CA 92037 The Journal of Experimental
Supplementary. presence of the. (c) mrna expression. Error. in naive or
 Figure 1. (a) Naive CD4 + T cells were activated in the presence of the indicated cytokines for 3 days. Enpp2 mrna expression was measured by qrt-pcrhr, infected with (b, c) Naive CD4 + T cells were activated
Figure 1. (a) Naive CD4 + T cells were activated in the presence of the indicated cytokines for 3 days. Enpp2 mrna expression was measured by qrt-pcrhr, infected with (b, c) Naive CD4 + T cells were activated
T cell maturation. T-cell Maturation. What allows T cell maturation?
 T-cell Maturation What allows T cell maturation? Direct contact with thymic epithelial cells Influence of thymic hormones Growth factors (cytokines, CSF) T cell maturation T cell progenitor DN DP SP 2ry
T-cell Maturation What allows T cell maturation? Direct contact with thymic epithelial cells Influence of thymic hormones Growth factors (cytokines, CSF) T cell maturation T cell progenitor DN DP SP 2ry
(Stratagene, La Jolla, CA) (Supplemental Fig. 1A). A 5.4-kb EcoRI fragment
 SUPPLEMENTAL INFORMATION Supplemental Methods Generation of RyR2-S2808D Mice Murine genomic RyR2 clones were isolated from a 129/SvEvTacfBR λ-phage library (Stratagene, La Jolla, CA) (Supplemental Fig.
SUPPLEMENTAL INFORMATION Supplemental Methods Generation of RyR2-S2808D Mice Murine genomic RyR2 clones were isolated from a 129/SvEvTacfBR λ-phage library (Stratagene, La Jolla, CA) (Supplemental Fig.
Supplementary Figure 1 Cytokine receptors on developing thymocytes that can potentially signal Runx3d expression.
 Supplementary Figure 1 Cytokine receptors on developing thymocytes that can potentially signal Runx3d expression. (a) Characterization of c-independent SP8 cells. Stainings for maturation markers (top)
Supplementary Figure 1 Cytokine receptors on developing thymocytes that can potentially signal Runx3d expression. (a) Characterization of c-independent SP8 cells. Stainings for maturation markers (top)
The T cell receptor for MHC-associated peptide antigens
 1 The T cell receptor for MHC-associated peptide antigens T lymphocytes have a dual specificity: they recognize polymporphic residues of self MHC molecules, and they also recognize residues of peptide
1 The T cell receptor for MHC-associated peptide antigens T lymphocytes have a dual specificity: they recognize polymporphic residues of self MHC molecules, and they also recognize residues of peptide
Supplemental Methods. CD107a assay
 Supplemental Methods CD107a assay For each T cell culture that was tested, two tubes were prepared. One tube contained BCMA-K562 cells, and the other tube contained NGFR-K562 cells. Both tubes contained
Supplemental Methods CD107a assay For each T cell culture that was tested, two tubes were prepared. One tube contained BCMA-K562 cells, and the other tube contained NGFR-K562 cells. Both tubes contained
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION doi:10.1038/nature11429 S1a 6 7 8 9 Nlrc4 allele S1b Nlrc4 +/+ Nlrc4 +/F Nlrc4 F/F 9 Targeting construct 422 bp 273 bp FRT-neo-gb-PGK-FRT 3x.STOP S1c Nlrc4 +/+ Nlrc4 F/F casp1
SUPPLEMENTARY INFORMATION doi:10.1038/nature11429 S1a 6 7 8 9 Nlrc4 allele S1b Nlrc4 +/+ Nlrc4 +/F Nlrc4 F/F 9 Targeting construct 422 bp 273 bp FRT-neo-gb-PGK-FRT 3x.STOP S1c Nlrc4 +/+ Nlrc4 F/F casp1
Absence of mouse 2B4 promotes NK cell mediated killing of activated CD8 + T cells, leading to prolonged viral persistence and altered pathogenesis
 Research article Absence of mouse 2B4 promotes NK cell mediated killing of activated CD8 + T cells, leading to prolonged viral persistence and altered pathogenesis Stephen N. Waggoner, 1 Ruth T. Taniguchi,
Research article Absence of mouse 2B4 promotes NK cell mediated killing of activated CD8 + T cells, leading to prolonged viral persistence and altered pathogenesis Stephen N. Waggoner, 1 Ruth T. Taniguchi,
Reviewers' comments: Reviewer #1 Expert in EAE and IL-17a (Remarks to the Author):
 Reviewers' comments: Reviewer #1 Expert in EAE and IL-17a (Remarks to the Author): This study shows that the inducible camp early repressor (ICER) is involved in development of Th17 cells that are pathogenic
Reviewers' comments: Reviewer #1 Expert in EAE and IL-17a (Remarks to the Author): This study shows that the inducible camp early repressor (ICER) is involved in development of Th17 cells that are pathogenic
Adaptive immune responses: T cell-mediated immunity
 MICR2209 Adaptive immune responses: T cell-mediated immunity Dr Allison Imrie allison.imrie@uwa.edu.au 1 Synopsis: In this lecture we will discuss the T-cell mediated immune response, how it is activated,
MICR2209 Adaptive immune responses: T cell-mediated immunity Dr Allison Imrie allison.imrie@uwa.edu.au 1 Synopsis: In this lecture we will discuss the T-cell mediated immune response, how it is activated,
B and T lymphocyte attenuator regulates CD8 + T cell intrinsic homeostasis and memory cell generation
 27 Nature Publishing Group http://www.nature.com/natureimmunology B and T lymphocyte attenuator regulates CD8 + T cell intrinsic homeostasis and memory cell generation Carsten Krieg 1, Onur Boyman 1,2,
27 Nature Publishing Group http://www.nature.com/natureimmunology B and T lymphocyte attenuator regulates CD8 + T cell intrinsic homeostasis and memory cell generation Carsten Krieg 1, Onur Boyman 1,2,
T Cell Differentiation
 T Cell Differentiation Ned Braunstein, MD MHC control of Immune Responsiveness: Concept Whether or not an individual makes an immune response to a particular antigen depends on what MHC alleles an individual
T Cell Differentiation Ned Braunstein, MD MHC control of Immune Responsiveness: Concept Whether or not an individual makes an immune response to a particular antigen depends on what MHC alleles an individual
Probe. Hind III Q,!?R'!! /0!!!!D1"?R'! vector. Homologous recombination
 Supple-Zhang Page 1 Wild-type locus Targeting construct Targeted allele Exon Exon3 Exon Probe P1 P P3 FRT FRT loxp loxp neo vector amh I Homologous recombination neo P1 P P3 FLPe recombination Q,!?R'!!
Supple-Zhang Page 1 Wild-type locus Targeting construct Targeted allele Exon Exon3 Exon Probe P1 P P3 FRT FRT loxp loxp neo vector amh I Homologous recombination neo P1 P P3 FLPe recombination Q,!?R'!!
Supplementary Figure 1. Efficient DC depletion in CD11c.DOG transgenic mice
 Supplementary Figure 1. Efficient DC depletion in CD11c.DOG transgenic mice (a) CD11c.DOG transgenic mice (tg) were treated with 8 ng/g body weight (b.w.) diphtheria toxin (DT) i.p. on day -1 and every
Supplementary Figure 1. Efficient DC depletion in CD11c.DOG transgenic mice (a) CD11c.DOG transgenic mice (tg) were treated with 8 ng/g body weight (b.w.) diphtheria toxin (DT) i.p. on day -1 and every
IL-6Rα IL-6RαT-KO KO. IL-6Rα f/f bp. f/f 628 bp deleted 368 bp. 500 bp
 STD H 2 O WT KO IL-6Rα f/f IL-6Rα IL-6RαT-KO KO 1000 bp 500 bp f/f 628 bp deleted 368 bp Supplementary Figure 1 Confirmation of T-cell IL-6Rα deficiency. (a) Representative histograms and (b) quantification
STD H 2 O WT KO IL-6Rα f/f IL-6Rα IL-6RαT-KO KO 1000 bp 500 bp f/f 628 bp deleted 368 bp Supplementary Figure 1 Confirmation of T-cell IL-6Rα deficiency. (a) Representative histograms and (b) quantification
Supplementary Figure 1: Expression of NFAT proteins in Nfat2-deleted B cells (a+b) Protein expression of NFAT2 (a) and NFAT1 (b) in isolated splenic
 Supplementary Figure 1: Expression of NFAT proteins in Nfat2-deleted B cells (a+b) Protein expression of NFAT2 (a) and NFAT1 (b) in isolated splenic B cells from WT Nfat2 +/+, TCL1 Nfat2 +/+ and TCL1 Nfat2
Supplementary Figure 1: Expression of NFAT proteins in Nfat2-deleted B cells (a+b) Protein expression of NFAT2 (a) and NFAT1 (b) in isolated splenic B cells from WT Nfat2 +/+, TCL1 Nfat2 +/+ and TCL1 Nfat2
NK cell flow cytometric assay In vivo DC viability and migration assay
 NK cell flow cytometric assay 6 NK cells were purified, by negative selection with the NK Cell Isolation Kit (Miltenyi iotec), from spleen and lymph nodes of 6 RAG1KO mice, injected the day before with
NK cell flow cytometric assay 6 NK cells were purified, by negative selection with the NK Cell Isolation Kit (Miltenyi iotec), from spleen and lymph nodes of 6 RAG1KO mice, injected the day before with
sequences of a styx mutant reveals a T to A transversion in the donor splice site of intron 5
 sfigure 1 Styx mutant mice recapitulate the phenotype of SHIP -/- mice. (A) Analysis of the genomic sequences of a styx mutant reveals a T to A transversion in the donor splice site of intron 5 (GTAAC
sfigure 1 Styx mutant mice recapitulate the phenotype of SHIP -/- mice. (A) Analysis of the genomic sequences of a styx mutant reveals a T to A transversion in the donor splice site of intron 5 (GTAAC
Supplementary Fig. 1 p38 MAPK negatively regulates DC differentiation. (a) Western blot analysis of p38 isoform expression in BM cells, immature DCs
 Supplementary Fig. 1 p38 MAPK negatively regulates DC differentiation. (a) Western blot analysis of p38 isoform expression in BM cells, immature DCs (idcs) and mature DCs (mdcs). A myeloma cell line expressing
Supplementary Fig. 1 p38 MAPK negatively regulates DC differentiation. (a) Western blot analysis of p38 isoform expression in BM cells, immature DCs (idcs) and mature DCs (mdcs). A myeloma cell line expressing
