Anumber of phenotypic characteristics differentiate antigenprimed
|
|
|
- Evelyn Baker
- 6 years ago
- Views:
Transcription
1 A transgenic mouse model to analyze CD8 effector T cell differentiation in vivo N. Manjunath*, P. Shankar*, B. Stockton, P. D. Dubey*, J. Lieberman*, and U. H. von Andrian* *The Center for Blood Research and the Departments of Pediatrics and Pathology, Harvard Medical School, Boston, MA 02115; and Department of Medicine, Tufts University, Boston, MA Edited by Irving L. Weissman, Stanford University School of Medicine, Stanford, CA, and approved September 22, 1999 (received for review March 31, 1999) Antigen-specific effector T cells are prerequisite to immune protection, but because of the lack of effector cell-specific markers, their generation and differentiation has been difficult to study. We report that effector cells are highly enriched in a T cell subset that can be specifically identified in transgenic (T-GFP) mice expressing green fluorescent protein (GFP) under control of the murine CD4 promoter and proximal enhancer. Consistent with previous studies of these transcriptional control elements, GFP was strongly and specifically expressed in nearly all resting and short-term activated CD4 and CD8 T cells. However, when T-GFP mice were challenged with vaccinia virus, allogeneic tumor cells, or staphylococcal enterotoxin A, the cytotoxic and IFN- -producing T cells lost GFP expression. Upon T cell receptor (TCR) ligation by CD3, sorted GFP cells fluxed calcium and proliferated vigorously. In contrast, GFP effector cells showed a diminished calcium flux and did not proliferate. Instead, they underwent apoptosis unless supplied with exogenous IL-2. By reverse transcription PCR analysis, the GFP cells up-regulated the pro-apoptotic molecule, Fas-L, and down-regulated gene expression of the proximal TCR signaling molecule, CD3, and c-jun, a component of the AP-1 transcription factor. Thus, differential regulation of TCR signaling may explain the divergent responses of naïve and effector T cells to antigen stimulation. Anumber of phenotypic characteristics differentiate antigenprimed T cells from naïve T cells. The latter express high molecular weight isoforms of CD45 and high levels of L-selectin (CD62L), but relatively low levels of CD44, CD11a (LFA-1), and CD49d ( 4 integrin). In contrast, antigen-primed T cells downregulate L-selectin, up-regulate CD44, CD11a, and CD49d, and express low molecular isoforms of CD45 (1 3). However, most of these changes in surface markers reflect quantitative variations in levels of expression, necessitating assessment of the overall pattern of expression to distinguish between subsets. Moreover, it is not clear if any of them can discriminate between activated (preeffector) versus effector T cells. Thus, at present there is no simple method to unambiguously differentiate, at the single-cell level, between naïve, activated, and effector T cells (4). Such a method would facilitate cellular and molecular analysis of events involved in effector and memory cell generation. We have developed a transgenic mouse model (T-GFP) that identifies a T cell population that is highly enriched for effector T cells generated in vivo. The green fluorescent protein (GFP) transgene, controlled by the CD4 promoter proximal enhancer, was expressed in all naïve T cells, but was shut off in a subset of antigen-responsive cells when the animals were challenged with vaccinia virus, superantigen, or allogeneic cells. Analysis of sorted cells revealed that all of the effector cell activity resided in the GFP population. T cell receptor (TCR) stimulation elicited distinct responses in GFP naïve cells compared with GFP effector T cells. Materials and Methods Transgene Construct and Microinjection. The murine CD4 promoter and enhancer construct (p37.1) (5) containing a unique SalI cloning site was a gift from Dan Littman, New York University Medical Center. pegfp-c1 (CLONTECH) was the source of enhanced GFP cdna. SalI sites at both ends of the GFP coding sequence were introduced by PCR, and the product was cloned into the SalI site of p37.1 to derive the T-GFP construct (Fig. 1A). Vector sequences were removed by NotI digestion, and the vector-free T-GFP construct was purified on a GeneClean column (Bio-Lab, St. Paul). Pronuclear DNA injection into fertilized FVB oocytes was performed at the Brigham and Women s Hospital Core Transgenic Facility (Boston). Founder pups were identified by PCR analysis of tail DNA and confirmed by flow cytometry. T-GFP animals were crossed to C57 background. Mice were used at 6 8 weeks of age unless stated otherwise. Antibody Staining, Flow Cytometry, and Cell Sorting. To determine T cell specificity of GFP expression, splenocytes from T-GFP mice were stained with phycoerythrin (PE)-conjugated antimouse Thy1.2, CD4, CD8, B220, Mac-1, Gr-1, and NK1.1 mabs (PharMingen). For activation markers, peritoneal exudate lymphocytes (PELs) and splenocytes were stained with PE-labeled anti-mouse CD62L (L-selectin), CD11a, CD11b, CD44, CD69, and CD25 antibodies (PharMingen). To measure apoptosis, cells were stained with either propidium iodide (PI) or PE-labeled annexin V (PharMingen). All samples were analyzed on a FACScan flow cytometer (Becton Dickinson) following standard procedures. For cell sorting, cells were stained with azidefree PE-labeled antibodies to CD4 or CD8, and the positive subset was sorted on the basis of GFP expression by using an Epics Cell Sorter (Coulter). Antigen Challenge. For vaccinia virus challenge, T-GFP mice were infected with the WR strain of vaccinia virus (from American Type Culture Collection; 10 5 plaque-forming units mouse in 0.2 ml of PBS i.p.). To elicit an allo-specific response, mice were injected i.p. with P815 (H2 d ) tumor cells. For superantigen stimulation, mice were injected i.p. with 20 g mouse staphylococcal enterotoxin A (SEA) (Sigma). Cytotoxicity Assays. To test viral-specific cytotoxicity, on day 7 postinfection, PELs were harvested by flushing with 3 ml of PBS. PELs were stained with CD8, and the GFP CD8 and GFP CD8 T cells were sorted into separate vials. Sorted cells were tested for lysis of 51 Cr-labeled MC57G (H2 b ) target cells infected with vaccinia virus in a standard 4-hr chromium release assay. For allo-specific CTL, sorted GFP CD8 and GFP CD8 T cells obtained 7 days after immunization with P815 cells were tested for lysis of 51 Cr-labeled P815 targets. Cytotoxicity was This paper was submitted directly (Track II) to the PNAS office. Abbreviations: GFP, green fluorescent protein; SEA, staphylococcal enterotoxin A; TCR, T cell receptor; PE, phycoerythrin; PEL, peritoneal exudate lymphocyte; PI, propidium iodide; RT-PCR, reverse transcription PCR. To whom reprint requests should be addressed at: The Center for Blood Research, Harvard Medical School, 200 Longwood Avenue, Boston, MA uva@cbr.med. harvard.edu. The publication costs of this article were defrayed in part by page charge payment. This article must therefore be hereby marked advertisement in accordance with 18 U.S.C solely to indicate this fact PNAS November 23, 1999 vol. 96 no. 24
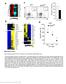
2 Fig. 1. T cells from T-GFP mice express GFP uniformly and selectively. To generate T-GFP mice, the GFP coding sequence was placed under control of the murine CD4 promoter proximal enhancer without the intronic silencer and the plasmid construct (A) was microinjected into fertilized oocytes. (B) Splenocytes from T-GFP mice were stained with mabs against murine T cells (Thy1.2), CD4, CD8, B cells (B220), macrophages (Mac-1), and NK cells (NK1.1) and examined by flow cytometry. Examination of peripheral blood leukocytes revealed that Gr-1 granulocytes were also GFP (not shown). defined as: (test release spontaneous release) (maximum release spontaneous release) 100%. Intracellular IFN- Assay. IFN- production was determined at the single cell level by flow cytometry. For vaccinia-specific IFN- production, PELs obtained on day 7 postinfection were restimulated in vitro by using vaccinia-infected adherent macrophages as described (6). The cultures were incubated for 6 8 hr in the presence of 1 M brefeldin A and stained for IFN- by using an intracellular staining kit (PharMingen). Briefly, cells were stained with cychrome-labeled CD8, fixed, permeabilized, and stained with PE-labeled anti-mouse IFN- antibody. CD8 gated cells were analyzed for IFN- production versus GFP expression by three-color flow cytometry. For allo-specific and SEA-specific IFN- production, PELs from immunized mice were restimulated with either 10 4 P815 cells or 10 g ml SEA for 6 hr in the presence of brefeldin A followed by intracellular staining for IFN-. Reverse Transcription PCR (RT-PCR) Assay. For gene expression studies, mice were immunized with vaccinia virus, and CD4 and CD8 T cells from splenocytes obtained on day 7 postinfection were sorted to separate GFP and GFP subsets. A total of sorted cells were dissolved in Trizol reagent (BRL), and total cellular RNA was extracted according to the manufacturer s recommendation. RNA was treated with RQ-1 DNase (Promega), extracted with phenol chloroform, and precipitated with ethanol. One hundred nanograms of RNA was reverse-transcribed by using a Perkin Elmer RT-PCR core kit. cdnas were amplified by using the following primer pairs derived from the Genbank database (from 5 to 3 ): GCAAGGGCGAGGAGCTGT- TCACCGGGG and TTACTTGTACAGCTCGTCCATGCCGA- GAG for enhanced GFP (nucleotides ); TGGTTCGGCAT- GACACTCTCAGTGC and CCACTTTCATTCACCACCAGGT- TCAC for CD4 (nucleotides ), CCGAGAGCGGTGCCT- ACGGCTACAG and GACCGGCTGTGCCGCGGAGGTGAC for c-jun (nucleotides ); CCGGGCACCATGAAGGCGGCC- GTCGA and GGATTCTGGGTAGGTAGGGTTGGCTC for Ets-1 (nucleotides ); GTGTCTGTTCTCGCCTGCATCCTCC and GGCAGCAGTCGCAGTCTCTGCACTCCTGCTG for CD3 (nucleotides ); TCATCTTGGGCTCCTCCAGGGTCAG and GGCTTTGGTTGGTGAACTCACGGAG for Fas-L (nucleotides ); and TGGAATCCTGTGGCATCCATGAAAC and TA- AAACGCAGCTCAGTAACAGTCCG for actin (nucleotides ). Thermal cycling conditions were: 1 min at 94 C denaturation; 1 min at 55 C annealing; and 2 min at 72 C extension. After amplification for 35 cycles, the PCR products were analyzed on 10% polyacrylamide gels. Lymphocyte Proliferation Assay. T-GFP mice were immunized with allogeneic P815 cells i.p. Splenic T cells were sorted 7 days later into GFP and GFP subsets. A total of sorted cells were cultured in the absence or presence of 1 g ml purified CD3 in triplicate in 96-well culture plates. A total of 10 5 irradiated spleen cells well were added as feeder cells. In some experiments, 100 units of recombinant IL-2 was added. Two days after stimulation, the cultures were pulsed with 3 H thymidine (0.5 Ci well) for 6 hr, harvested, and counted for 3 H incorporation by using a Packard Topcount harvester and microplate reader. Calcium Mobilization Assay. Intracellular Ca 2 concentration ([Ca 2 ] i ) was determined as described (7). Splenocytes from P815-immunized mice obtained on day 7 postimmunization were loaded with 1 M Indo-1 at 37 C for 1 hr, stained with PE-labeled CD8, washed, and resuspended to cells ml. [Ca2 ] i was determined by using a Coulter Epic V flow cytometer. After running the cells for 1 min to obtain a steady baseline, CD3 antibody (5 g ml) was added, and after 1 min, goat anti-hamster IgG (1 g ml) was added to crosslink CD3. [Ca 2 ] i was monitored during the next 10 min. Results Production of T-GFP Mice. In an effort to track individual T cells in vivo, we generated transgenic mice, termed T-GFP, which express enhanced GFP selectively and uniformly in CD4 and CD8 T cells. To generate T-GFP mice, the GFP coding sequence was placed under control of the murine CD4 promoter and proximal enhancer without the intronic silencer (Fig. 1A), which suppresses CD4 gene expression in CD8 cells (8). The T-GFP construct was microinjected into fertilized eggs, and founders were identified by PCR analysis of tail DNA (not shown). Flow cytometric analysis of T-GFP splenocytes confirmed uniform T cell-specific expression of GFP (Fig. 1B). Many Transgenic T Cells Lose GFP Expression After Antigen Challenge. We produced the T-GFP mice in part because we expected that GFP would provide an easy-to-follow marker for use in adoptive transfer experiments to study antigen-specific memory T cells. To test the ability of T-GFP mice to mount a T cell-mediated immune response, we infected T-GFP mice with vaccinia virus and examined their splenocytes and PELs on day 7 postinfection. Surprisingly, many T cells in both the CD4 and CD8 compartments had lost GFP expression (Fig. 2A). Similar results were obtained from three different founders, excluding the possibility that this finding was the result of positional effects of transgene integration (not shown). GFP T cells were not apoptotic because they did not stain with PI or annexin V (Fig. 2A). GFP T cells were more frequent among PELs than among splenocytes and in both populations the CD8 subset contained more GFP cells than the CD4 subset. Because the immune response to vaccinia infection is dominated by CD8 T cells (9), we speculated that primarily the activated virus-specific T cells had lost GFP expression. To test this possibility, we stained PELs and splenocytes for activation markers and analyzed GFP coexpression by flow cytometry after gating on CD8 T cells. Indeed, most GFP CD8 cells were activated, i.e., they were L-selectin low, LFA-1 high, and CD44 high. A subset of these cells also expressed CD69, CD11b (Mac-1), and CD25. These IMMUNOLOGY Manjunath et al. PNAS November 23, 1999 vol. 96 no
3 Fig. 2. A subset of activated T cells loses GFP expression after vaccinia infection in vivo or after CD3 stimulation in vitro. (A) T-GFP mice were infected with vaccinia virus, and on day 7 postinfection, T cells in PELs (Top) and splenocytes (Middle) were tested for GFP expression. PELs also were tested for cell viability (Bottom). Representative results from one infected mouse (of 10 mice analyzed) are shown. (B) Six days postvaccinia infection, PELs and splenocytes were analyzed for expression of GFP versus activation markers by using three-color flow cytometry. Splenocytes from uninfected mice are shown as a control. Representative results from one mouse each (of four analyzed) are shown after gating on the CD8 subset. (C) Transgenic splenocytes were stimulated in vitro with CD3, and GFP expression in the CD4 and CD8 subset was monitored over time. changes apparently were caused by the vaccinia infection because nearly all CD8 T cells from uninfected control mice remained GFP and were L-selectin high, LFA-1 low, and CD44 low (Fig. 2B). A closer examination of Fig. 2B revealed that a substantial fraction of GFP CD8 T cells in infected mice also expressed some of these activation markers. This finding suggests that loss of GFP occurred independent of or perhaps subsequent to changes associated with an activation phenotype. To distinguish between these two possibilities and to determine the kinetics of GFP downregulation, we stimulated splenocytes from naïve animals in vitro with CD3 and monitored GFP expression at various time points thereafter. Initially, proliferating T cells continued to express GFP, but starting from day 4, a GFP population became apparent, and by day 7 poststimulation, the majority of CD8 and about half of the CD4 T cells were GFP (Fig. 2C). Similar kinetics also were seen after infection with vaccinia virus in vivo (not shown). These results indicate that loss of GFP expression is associated with events occurring late after activation. CD8 T Cells That Lose GFP Expression Are Exclusive Mediators of Effector Functions. During an immune response to viral infection or allo-stimulation in vivo, phenotypic signs of T cell activation become evident within 3 4 days, but CTL activity becomes first demonstrable only after 5 6 days, suggesting that a period of maturation is required to generate functional effector CTL (10). Because loss of GFP was also a late event after activation, we examined whether GFP down-regulation was associated with the acquisition of effector functions. For these studies, CD8 T cells from vaccinia-infected mice were sorted on the basis of GFP expression and tested for cytotoxic function. Only GFP cells, but not GFP cells, were capable of virus-specific cytotoxicity (Fig. 3A, Left). Similar results also were obtained in an allospecific response after challenge with P815 (H2 d ) cells (Fig. 3A, Right), suggesting that loss of GFP may be a marker for effector T cells. To examine this hypothesis further, we also tested GFP and GFP cells from vaccinia-infected mice for antigen-induced production of the major CD8 cell effector cytokine, IFN- (Fig. 3B). Using three-color flow cytometry to study selectively the CD8 subset, we detected virus-specific IFN- production in GFP, but not in GFP, cells. These results also held true when effector cell differentiation was induced by other stimuli such as allogeneic P815 tumor cells or SEA (Fig. 3B). Several Molecular Changes Are Associated with Loss of GFP Expression in T Cells. Although T-GFP effector cells had lost CD4 promoter proximal enhancer-driven GFP expression, the GFP CD4 T cells Manjunath et al.
4 Fig. 3. Loss of GFP expression discriminates a subset of differentiated T cells that are cytotoxic and produce IFN-.(A) Seven days postvaccinia infection or postimmunization with allogeneic P815 cells, CD8 T cells from PELs were sorted into GFP and GFP cells and tested for lysis of vaccinia-infected MC57G (Left) or allogeneic P815 (Right) target cells. Results from two T-GFP mice for each method of immunization are shown. E:T ratio, effector-to-target ratio. (B) Mice were challenged i.p. with vaccinia virus, P815 cells, or SEA. After 7 days, PELs were tested for antigen (Ag)-specific intracellular IFN- production as described in Materials and Methods. Gated CD8 cells from one representative mouse (of three tested with similar results) for each method of immunization are shown. continued to express CD4 on their surface. To determine whether this finding was reflected at the transcriptional level, we sorted CD4 T cells from spleens of P815-challenged T-GFP mice and tested the GFP and GFP CD4 cells for GFP and CD4 gene expression by RT-PCR. Fig. 4 shows that while the GFP transgene was turned off, endogenous CD4 gene expression continued in the GFP cells. We interpret these results to mean that the promoter proximal enhancer used in the T-GFP construct is turned off in the GFP cells, but cellular CD4 expression continues presumably because alternate enhancer(s) can drive CD4 gene expression. In fact, in addition to the proximal enhancer contained in our transgene, one or more other CD4 enhancers may exist (11, 12). Because effector T cells in T-GFP mice lost GFP expression, we reasoned that analysis of transcription factors that are thought to control CD4 expression might reveal molecular events that are distinctly associated with effector T cells. Transcription factors implicated in the control of CD4 expression include the Ets family member Ets-1 and the AP-1 complex (12). Gene expression of these elements was down-regulated in GFP effector T cells compared with GFP cells in both CD4 and CD8 T cells. We have observed that the cytotoxic and IFN- producing CD8 T cells from human adults infected with Epstein Barr virus down-regulate CD3 (J.L., unpublished data). Thus, we assessed CD3 expression in GFP and GFP T cells and found that it, too, was significantly reduced in the GFP subset in both CD4 and CD8 T cells. One of the mechanisms of effector T cell-induced target cell killing is induction of apoptosis via the Fas pathway (13). Fas Fas-L interactions also have been thought to regulate immune homeostasis by autocrine and paracrine interactions (14). It seems likely that this important function also is associated with effector cells. Indeed, Fig. 4. Differential gene expression in GFP and GFP T cells after immunization. Seven days postvaccinia infection, splenocytes were stained with CD4 or CD8 antibodies and sorted based on GFP expression. RT-PCR was performed as detailed in Materials and Methods, and the PCR products were resolved by PAGE. The MW lane shows a 100-bp ladder; the lowermost band represents 200 bp. The larger of the two bands in lanes Ets-1 represents the predicted full-length product. gene expression of Fas-L was dramatically up-regulated in sorted GFP cells when compared with GFP cells. GFP T Cells Differ from GFP T Cells in Their Response to TCR Engagement. The results in Fig. 4 suggest that the GFP population down-regulated CD3 and the transcription factor c-jun. This finding was of particular interest because these proteins constitute the most proximal and most distal molecules involved in the induction of IL-2 synthesis after T cell activation via the TCR (15). However, because our data were obtained by RT- PCR that may not be truly quantitative, we sought to characterize directly the response of GFP T cells to TCR ligation. We tested whether calcium mobilization in GFP T cells is different from GFP T cells. CD8 splenocytes from P815-stimulated T-GFP mice were analyzed for calcium flux in response to CD3. GFP CD8 T cells mobilized calcium rapidly and at a high frequency, whereas the calcium flux appeared delayed and involved a smaller fraction of GFP CD8 T cells (Fig. 5A). This finding is surprising considering that antigen-specific cytotoxicity is thought to be calcium dependent (16). It is possible that a small or transient calcium flux in effector cells is enough for effector functions, whereas a much larger and sustained signal may be needed for naïve cells to proliferate (17). We also compared sorted GFP T cells with their GFP counterparts for proliferation after CD3 stimulation. As shown in Fig. 5B, the GFP T cells proliferated vigorously, whereas the GFP T cells did not. However, despite their inability to proliferate, many GFP, but almost no GFP, T cells responded to CD3 by producing IFN- (Fig. 5C). To determine the ultimate fate of CD3-stimulated GFP T cells, we used annexin V staining to assess whether these cells underwent apoptosis. Indeed, apoptosis was induced in GFP, but not in GFP, T cells under these conditions (Fig. 5D). Thus, in contrast to naïve and pre-effector (GFP ) T cells, TCR crosslinking on effector (GFP ) T cells resulted in reduced calcium mobilization and lack of proliferation, but sustained IMMUNOLOGY Manjunath et al. PNAS November 23, 1999 vol. 96 no
5 Fig. 5. GFP effector T cells show diminished Ca 2 mobilization, do not proliferate, and undergo apoptosis after stimulation with CD3, but they exert effector functions and can be rescued by exogenous IL-2. (A) Splenocytes from P815-immunized T-GFP mice were loaded with Indo-1 and stained with CD8. After gating on the CD8 subset, the [Ca 2 ] i flux response to CD3 crosslinking was monitored in the GFP and GFP subsets as described in Materials and Methods. Ratio of blue to violet (B V) median channel fluorescence intensity over time (Left and Middle) as well as % cells responding (Right) are shown. A decrease in the blue violet fluorescence ratio indicates calcium flux. Representative results from one mouse (of three mice tested) are shown. Arrows at 1 min and 2 min indicate addition of hamster anti-mouse CD3 and goat anti-hamster IgG, respectively. (B) Sorted GFP or GFP CD8 T cells obtained from P815-primed T-GFP mice were culturedwith CD3intheabsence(Left)orpresence (Right) of 100 units of recombinant IL-2 and tested for proliferation by 3 H thymidine incorporation after 48 hr. Results from two independent experiments are shown. (C) PELs obtained 7 days after challenge with P815 cells were stimulated with CD3 and tested for intracellular IFN- production. Results from one mouse (of three tested with similar results) are shown. (D) PELs from P815-immunized mice were tested for annexin V staining without further restimulation (Left) or 8 hr after stimulation with CD3 (Right). Results from one mouse (of three tested with similar results) are shown. effector activity and, eventually, apoptosis. However, it seems unlikely that effector T cells always undergo apoptosis upon antigen stimulation in vivo. It is more likely that their survival after antigen recognition is tightly controlled, e.g., through dependence on survival or growth factors that may be released by other cells. To examine this possibility, we tested whether GFP T cells could be rescued from apoptosis and induced to proliferate by exogenously supplied IL-2. Indeed, upon addition of IL-2 GFP T cells proliferated as well as their GFP counterparts. (Fig. 5B). Thus, dependence on exogenous IL-2 for survival appears to characterize effector T cells. Discussion The ability to better distinguish between naive, activated, and effector T cells would be of help in furthering our understanding of effector T cell differentiation and protective immunity. We have developed a transgenic mouse model in which selective and uniform expression of GFP was seen in naive and early activated (pre-effector) T cells. Surprisingly, the transgene was turned off in a T cell population that contained essentially all of the differentiated CD8 effector T cells. These mice may provide a valuable tool to isolate mature effector T cells as a discrete subpopulation that can be readily identified by loss of GFP expression. After recognizing cognate antigen, activated T cells downregulate L-selectin, up-regulate CD44, CD11a, and CD49d, and express low molecular weight isoforms of CD45 (1 3). Increased expression of CD44 frequently is used as a marker to identify and isolate murine effector T cells because CD44 up-regulation occurs uniformly after activation (18, 19). However, CD44 ligation by antibodies or natural ligands can affect T cell trafficking and function (20, 21). Thus, antibodies against CD44 may not be ideal tools to select T cells for functional studies or in adoptive transfer experiments. Moreover, because CD44 up-regulation occurs within 2 days of antigen recognition (10), it appears to be a marker of T cell activation rather than effector differentiation. In contrast to most other activation markers, loss of GFP expression is detected only at 4 5 days after activation. This time course of GFP extinction parallels the reported induction of effector cytotoxic molecules like perforin, granzymes, and FasL in CD8 T cells (22). Thus, the loss of GFP expression is closely linked to and perhaps specifically triggered by effector differentiation. However, it cannot be excluded that down-regulation of GFP transcription occurred somewhat earlier, and GFP T cells became detectable only after some delay because GFP protein might have a relatively long half-life in T cells. In either case, T-GFP mice appear to provide a tool to identify and select differentiated effector T cells that are clearly distinct from naïve and early activated (pre-effector) T cells. Antigen-specific CTL activity and IFN- production was seen exclusively in the GFP population. Moreover, although most of the GFP CD8 PEL were CD44 high, and the majority expressed little or no L-selectin (Fig. 2B), these cells were incapable of killing vaccinia-infected target cells (Fig. 3A) or secreting IFN- upon antigen challenge (Fig. 3B). This population probably represented activated pre-effector T cells. By day 7 after vaccinia infection, approximately 60% of CD4 T cells and over 80% of CD8 T cells accumulating at the site of infection (i.e., the peritoneal cavity) had lost GFP expression (Fig. 1B). In the spleen, nearly 30% of the CD4 T cells and 60% of the CD8 T cells were GFP. It is not clear whether all GFP T cells specifically recognize vaccinia-associated antigens. However, the presence of large numbers of GFP -activated T cells in our experiments agrees with recent studies using TCR transgenic mice and MHC-peptide tetramers to detect antigen-specific T cells in other models of viral infection. These studies have shown that there is a massive expansion of such T cells during immune responses in vivo (4, 23, 24). It is unlikely that the extinction of GFP represented a nonspecific inflammatory stress response, Manjunath et al.
6 because a clearly discernable subset (5 8%) of GFP CD8 T cells (memory cells) capable of viral-specific IFN- production persisted in the spleens 3 months after vaccinia infection (unpublished data). Moreover, GFP CD8 T cells showed a tendency to increase in spleens of uninfected animals as they aged (6.9 3% in 4-week-old mice versus % in mice older than 1 year (n 3, data not shown). These GFP CD8 T cells probably represent memory cells to environmental antigens, exposure to which increases over time. Although antigen-specific effector functions such as cytotoxicity and IFN- production resided only in the GFP population, not all GFP cells produced IFN- upon antigen-specific stimulation (Fig. 3B). Thus, it cannot be excluded that the GFP population contained additional T cell subsets that may not exert effector functions or perform other functions (e.g., regulatory) that were not assayed here. However, it is likely that the method used for antigen-specific stimulation was not optimal to ensure stimulation of all antigen-specific T cells within the 6-hr assay period. Alternatively, some GFP cells may have represented activated bystander (antigen-nonspecific) cells. However, even after exposure to CD3, which should stimulate all T cells, a substantial number of GFP cells failed to produce IFN- (Fig. 5C). These results raise the possibility that not all effector T cells acquired all of the effector functions tested (i.e., cytotoxicity as well as IFN- production). In fact, the existence of antigenspecific T cells identifiable by MHC-peptide tetramers in vivo, which do not secrete IFN- has been recognized recently (25). Further studies with T-GFP mice that will be crossed with TCR transgenic mice might provide insights into these issues. It has been reported that CD4 expression is up-regulated in antigen-activated CD4 T cells both in vitro and in vivo (26). However, upon activation of cells in vitro with lectins or phorbol 12-myristate 13-acetate, CD4 expression decreases (26, 27). Our results indicate that while the proximal CD4 enhancer-driven GFP expression is turned off, endogenous CD4 protein continues to be expressed. These differences suggest that the transcriptional regulation of the murine CD4 locus is even more complex than previously thought. Interestingly, many GFP cells, but not naïve GFP T cells, underwent apoptosis after CD3 stimulation in vitro (Fig. 5D). However, it is difficult to imagine that antigen recognition would induce apoptosis of e.g., viral-specific effector T cells at a site of viral infection. Accordingly, apoptosis was not evident in effector cells that were generated in vivo because GFP PELs from vaccinia-infected T-GFP mice did not stain with annexin V or PI (Fig. 2A). Moreover, the GFP T cells could be rescued from CD3-induced apoptosis in vitro by addition of IL-2 (Fig. 5B). Thus, effector T cell survival in vivo may depend on growth factors such as IL-2 that must be produced by other T cells. Indeed, it has been reported recently that effector CD8 T cells are incapable of endogenous IL-2 synthesis (28). Our data suggest a plausible explanation for this observation. Both the most proximal (CD3 ) and the most distal (AP-1) component in the antigen-triggered signaling cascade that induces IL-2 synthesis were down-regulated in GFP T cells. It should be cautioned that the RT-PCR technique that was used to compare the transcription of these molecules is at best semiquantitative. Nevertheless, our findings raise the possibility that differential transcription of key molecules involved in TCR signaling in naive versus effector T cells is responsible for the observed divergent response to antigen in terms of survival and proliferation. Our findings suggest that effector differentiation may be associated with down-modulation of signaling components that control T cell proliferation. Similar molecular and functional changes have been reported previously in situations of chronic antigen exposure and have been interpreted as a deficiency or failure of the immune system. For instance, in situations of chronic antigenic stimulation like cancer, autoimmune diseases, and HIV infection, T cells down-regulate CD3 and may be hyporesponsive to TCR engagement (29 31). Lack of proliferation upon TCR stimulation is also a hallmark of anergic T cells. Anergic T cells also down-regulate the expression of the AP-1 transcription factor (32). Our data indicate that normal effector T cells generated during an ongoing immune response in vivo also share these features. Further molecular comparison of isolated effector, anergic, and memory T cells may provide insights into the generation of these T cell subsets. T-GFP mice may provide a valuable tool to such studies. We thank Alan Flint, John Daley, and Mark Ryan for assistance in cell sorting and Dan Littman for generously providing plasmid clones. This work was supported by National Institutes of Health Grants HL54936 and HL56949 (to U.H.v.A.), HL02881 (to N.M.), and HL29583 (to P.D.D.). 1. Vietta, E. S., Berton, M. T., Burget, C., Keoron, M., Lee, W. T. & Yin, X. M. (1991) Annu. Rev. Immunol. 9, Gray, D. (1993) Annu. Rev. Immunol. 11, Swain, S. L., Croft, M., Dubey, C., Haynes, L., Rogers, P., Zhang, X. & Bradley, L. M. (1996) Immunol. Rev. 150, Zimmermann, C., Brduscha-Riem, K., Blaser, C., Zinkernagel, R. M. & Pircher, H. (1996) J. Exp. Med. 183, Sawada, S. & Littman, D. R. (1991) Mol. Cell. Biol. 11, Shankar, P., Fabry, J. & Lieberman, J. (1995) J. Immunol. Invest. 24, Chused, T. M., Wilson, H. A., Greenblatt, D., Ishida, Y., Edison, L. J., Tsien, R. Y. & Finkelman, F. D. (1987) Cytometry 8, Sawada, S., Scarborough, J. D., Kileen, N. & Littman, D. R. (1994) Cell 77, Kagi, D., Seiler, P., Pavlovic, J., Burki, K., Zinkernagel, R. M. & Hengartner, H. (1995) Eur. J. Immunol. 25, Kedl, R. M. & Mescher, M. F. (1997) J. Immunol. 159, Wurster, A. L., Siu, G., Leiden, J. M. & Hedrick, S. M. (1994) Mol. Cell. Biol. 14, Ellmeier, W., Sawada, S. & Littman, D. R. (1999) Annu. Rev. Immunol. 17, Takahasi, T., Tanaka, M., Brannan C. I., Jenkins, N. A., Copeland N. G., Suda, T. & Nagata, S. (1994) Cell 76, Russell, J. H. (1995) Curr. Opin. Immunol. 7, Berridge, M. J. (1997) Crit. Rev. Immunol. 17, Esser, M. T., Krishnamurthy, B. & Braciale, V. L. (1996) J. Exp. Med. 183, Lezzi, G., Karjalainen, K. & Lanzavecchia, A. (1998) Immunity 8, Goldstein, J. S., Chen, T., Brunswick, M., Mostowsky, H. & Kozlowski, S. (1998) J. Immunol. 160, Budd, R. C., Cerottini, J. C., Horvath, C., Bron, C., Pedrazzini, T., Howe, R. C. & MacDonald, H. R. (1987) J. Immunol. 138, DeGrendele, H. C., Estess, P. & Siegelman, M. H. (1997) Science 278, Seth, A., Gote, L., Nagarkatti, M. & Nagarkatti, P. S. (1991) Proc. Natl. Acad. USA 88, Opferman, J. T., Ober, B. T. & Ashton-Rickart, P. G. (1999) Science 283, Butz, E. A. & Bevan, M. J. (1998) Immunity 8, Murali-Krishna, K., Altman, J. D., Suresh, M., Sourdive, D. J. D., Zajec, A. J., Miller, J. D., Slansky, J. & Ahmed, R. (1998) Immunity 8, Zajac, A. J., Blattman, J. N., Murali-Krishna, K., Sourdive, D. J. D., Suresh, M., Altman, J. D. & Ahmed, R. (1998) J. Exp. Med. 188, Ridgeway, W., Fasso, M. & Fathman C. G. (1998) J. Immunol. 161, Solbach, W. (1982) J. Exp. Med. 156, Caruso, A., Licenziati, S., Morelli, D., Florentini, S., Ricotta, D., Malacarne, F., Sfondrini, L. & Balsar, A. (1998) Eur. J. Immunol. 28, Mizoguchi, H. O., O Shea, J. J., Longo, D. L., Loeffler, C. M., McVicar, D. W. & Ochea, A. C. (1992) Science 258, Maurice, M. M., Lankester, A. C., Bezemer, A. C., Geertsman, M. F., Tak, P. P., Breedveld, F. C., van Lier, R. A. W. & Verweij, C. L. (1997) J. Immunol. 159, Trimble, L. A. & Lieberman, J. (1998) Blood 91, Schwartz, R. H. (1997) Curr. Opin. Immunol. 9, IMMUNOLOGY Manjunath et al. PNAS November 23, 1999 vol. 96 no
SUPPLEMENTARY INFORMATION
 Complete but curtailed T-cell response to very-low-affinity antigen Dietmar Zehn, Sarah Y. Lee & Michael J. Bevan Supp. Fig. 1: TCR chain usage among endogenous K b /Ova reactive T cells. C57BL/6 mice
Complete but curtailed T-cell response to very-low-affinity antigen Dietmar Zehn, Sarah Y. Lee & Michael J. Bevan Supp. Fig. 1: TCR chain usage among endogenous K b /Ova reactive T cells. C57BL/6 mice
Cytotoxicity assays. Rory D. de Vries, PhD 1. Viroscience lab, Erasmus MC, Rotterdam, the Netherlands
 Cytotoxicity assays Rory D. de Vries, PhD 1 1 Viroscience lab, Erasmus MC, Rotterdam, the Netherlands Anti-influenza immunity Humoral / CD4+ / CD8+ / NK? Function of CTL Elimination of virus-infected cells?
Cytotoxicity assays Rory D. de Vries, PhD 1 1 Viroscience lab, Erasmus MC, Rotterdam, the Netherlands Anti-influenza immunity Humoral / CD4+ / CD8+ / NK? Function of CTL Elimination of virus-infected cells?
Scott Abrams, Ph.D. Professor of Oncology, x4375 Kuby Immunology SEVENTH EDITION
 Scott Abrams, Ph.D. Professor of Oncology, x4375 scott.abrams@roswellpark.org Kuby Immunology SEVENTH EDITION CHAPTER 13 Effector Responses: Cell- and Antibody-Mediated Immunity Copyright 2013 by W. H.
Scott Abrams, Ph.D. Professor of Oncology, x4375 scott.abrams@roswellpark.org Kuby Immunology SEVENTH EDITION CHAPTER 13 Effector Responses: Cell- and Antibody-Mediated Immunity Copyright 2013 by W. H.
Supplemental Table I.
 Supplemental Table I Male / Mean ± SEM n Mean ± SEM n Body weight, g 29.2±0.4 17 29.7±0.5 17 Total cholesterol, mg/dl 534.0±30.8 17 561.6±26.1 17 HDL-cholesterol, mg/dl 9.6±0.8 17 10.1±0.7 17 Triglycerides,
Supplemental Table I Male / Mean ± SEM n Mean ± SEM n Body weight, g 29.2±0.4 17 29.7±0.5 17 Total cholesterol, mg/dl 534.0±30.8 17 561.6±26.1 17 HDL-cholesterol, mg/dl 9.6±0.8 17 10.1±0.7 17 Triglycerides,
MATERIALS AND METHODS. Neutralizing antibodies specific to mouse Dll1, Dll4, J1 and J2 were prepared as described. 1,2 All
 MATERIALS AND METHODS Antibodies (Abs), flow cytometry analysis and cell lines Neutralizing antibodies specific to mouse Dll1, Dll4, J1 and J2 were prepared as described. 1,2 All other antibodies used
MATERIALS AND METHODS Antibodies (Abs), flow cytometry analysis and cell lines Neutralizing antibodies specific to mouse Dll1, Dll4, J1 and J2 were prepared as described. 1,2 All other antibodies used
Commercially available HLA Class II tetramers (Beckman Coulter) conjugated to
 Class II tetramer staining Commercially available HLA Class II tetramers (Beckman Coulter) conjugated to PE were combined with dominant HIV epitopes (DRB1*0101-DRFYKTLRAEQASQEV, DRB1*0301- PEKEVLVWKFDSRLAFHH,
Class II tetramer staining Commercially available HLA Class II tetramers (Beckman Coulter) conjugated to PE were combined with dominant HIV epitopes (DRB1*0101-DRFYKTLRAEQASQEV, DRB1*0301- PEKEVLVWKFDSRLAFHH,
Cellular Immune response. Jianzhong Chen, Ph.D Institute of immunology, ZJU
 Cellular Immune response Jianzhong Chen, Ph.D Institute of immunology, ZJU Concept of adaptive immune response T cell-mediated adaptive immune response I. Concept of immune response A collective and coordinated
Cellular Immune response Jianzhong Chen, Ph.D Institute of immunology, ZJU Concept of adaptive immune response T cell-mediated adaptive immune response I. Concept of immune response A collective and coordinated
NK cell flow cytometric assay In vivo DC viability and migration assay
 NK cell flow cytometric assay 6 NK cells were purified, by negative selection with the NK Cell Isolation Kit (Miltenyi iotec), from spleen and lymph nodes of 6 RAG1KO mice, injected the day before with
NK cell flow cytometric assay 6 NK cells were purified, by negative selection with the NK Cell Isolation Kit (Miltenyi iotec), from spleen and lymph nodes of 6 RAG1KO mice, injected the day before with
Immune response. This overview figure summarizes simply how our body responds to foreign molecules that enter to it.
 Immune response This overview figure summarizes simply how our body responds to foreign molecules that enter to it. It s highly recommended to watch Dr Najeeb s lecture that s titled T Helper cells and
Immune response This overview figure summarizes simply how our body responds to foreign molecules that enter to it. It s highly recommended to watch Dr Najeeb s lecture that s titled T Helper cells and
TCR, MHC and coreceptors
 Cooperation In Immune Responses Antigen processing how peptides get into MHC Antigen processing involves the intracellular proteolytic generation of MHC binding proteins Protein antigens may be processed
Cooperation In Immune Responses Antigen processing how peptides get into MHC Antigen processing involves the intracellular proteolytic generation of MHC binding proteins Protein antigens may be processed
Naive, memory and regulatory T lymphocytes populations analysis
 Naive, memory and regulatory T lymphocytes populations analysis Jaen Olivier, PhD ojaen@beckmancoulter.com Cellular Analysis application specialist Beckman Coulter France Introduction Flow cytometric analysis
Naive, memory and regulatory T lymphocytes populations analysis Jaen Olivier, PhD ojaen@beckmancoulter.com Cellular Analysis application specialist Beckman Coulter France Introduction Flow cytometric analysis
NKTR-255: Accessing The Immunotherapeutic Potential Of IL-15 for NK Cell Therapies
 NKTR-255: Accessing The Immunotherapeutic Potential Of IL-15 for NK Cell Therapies Saul Kivimäe Senior Scientist, Research Biology Nektar Therapeutics NK Cell-Based Cancer Immunotherapy, September 26-27,
NKTR-255: Accessing The Immunotherapeutic Potential Of IL-15 for NK Cell Therapies Saul Kivimäe Senior Scientist, Research Biology Nektar Therapeutics NK Cell-Based Cancer Immunotherapy, September 26-27,
The Adaptive Immune Responses
 The Adaptive Immune Responses The two arms of the immune responses are; 1) the cell mediated, and 2) the humoral responses. In this chapter we will discuss the two responses in detail and we will start
The Adaptive Immune Responses The two arms of the immune responses are; 1) the cell mediated, and 2) the humoral responses. In this chapter we will discuss the two responses in detail and we will start
T-cell activation T cells migrate to secondary lymphoid tissues where they interact with antigen, antigen-presenting cells, and other lymphocytes:
 Interactions between innate immunity & adaptive immunity What happens to T cells after they leave the thymus? Naïve T cells exit the thymus and enter the bloodstream. If they remain in the bloodstream,
Interactions between innate immunity & adaptive immunity What happens to T cells after they leave the thymus? Naïve T cells exit the thymus and enter the bloodstream. If they remain in the bloodstream,
T-cell activation T cells migrate to secondary lymphoid tissues where they interact with antigen, antigen-presenting cells, and other lymphocytes:
 Interactions between innate immunity & adaptive immunity What happens to T cells after they leave the thymus? Naïve T cells exit the thymus and enter the bloodstream. If they remain in the bloodstream,
Interactions between innate immunity & adaptive immunity What happens to T cells after they leave the thymus? Naïve T cells exit the thymus and enter the bloodstream. If they remain in the bloodstream,
Micro 204. Cytotoxic T Lymphocytes (CTL) Lewis Lanier
 Micro 204 Cytotoxic T Lymphocytes (CTL) Lewis Lanier Lewis.Lanier@ucsf.edu Lymphocyte-mediated Cytotoxicity CD8 + αβ-tcr + T cells CD4 + αβ-tcr + T cells γδ-tcr + T cells Natural Killer cells CD8 + αβ-tcr
Micro 204 Cytotoxic T Lymphocytes (CTL) Lewis Lanier Lewis.Lanier@ucsf.edu Lymphocyte-mediated Cytotoxicity CD8 + αβ-tcr + T cells CD4 + αβ-tcr + T cells γδ-tcr + T cells Natural Killer cells CD8 + αβ-tcr
Supplementary Figure 1 CD4 + T cells from PKC-θ null mice are defective in NF-κB activation during T cell receptor signaling. CD4 + T cells were
 Supplementary Figure 1 CD4 + T cells from PKC-θ null mice are defective in NF-κB activation during T cell receptor signaling. CD4 + T cells were isolated from wild type (PKC-θ- WT) or PKC-θ null (PKC-θ-KO)
Supplementary Figure 1 CD4 + T cells from PKC-θ null mice are defective in NF-κB activation during T cell receptor signaling. CD4 + T cells were isolated from wild type (PKC-θ- WT) or PKC-θ null (PKC-θ-KO)
ACTIVATION AND EFFECTOR FUNCTIONS OF CELL-MEDIATED IMMUNITY AND NK CELLS. Choompone Sakonwasun, MD (Hons), FRCPT
 ACTIVATION AND EFFECTOR FUNCTIONS OF CELL-MEDIATED IMMUNITY AND NK CELLS Choompone Sakonwasun, MD (Hons), FRCPT Types of Adaptive Immunity Types of T Cell-mediated Immune Reactions CTLs = cytotoxic T lymphocytes
ACTIVATION AND EFFECTOR FUNCTIONS OF CELL-MEDIATED IMMUNITY AND NK CELLS Choompone Sakonwasun, MD (Hons), FRCPT Types of Adaptive Immunity Types of T Cell-mediated Immune Reactions CTLs = cytotoxic T lymphocytes
Blocking antibodies and peptides. Rat anti-mouse PD-1 (29F.1A12, rat IgG2a, k), PD-
 Supplementary Methods Blocking antibodies and peptides. Rat anti-mouse PD-1 (29F.1A12, rat IgG2a, k), PD- L1 (10F.9G2, rat IgG2b, k), and PD-L2 (3.2, mouse IgG1) have been described (24). Anti-CTLA-4 (clone
Supplementary Methods Blocking antibodies and peptides. Rat anti-mouse PD-1 (29F.1A12, rat IgG2a, k), PD- L1 (10F.9G2, rat IgG2b, k), and PD-L2 (3.2, mouse IgG1) have been described (24). Anti-CTLA-4 (clone
Supplementary Materials for
 www.sciencesignaling.org/cgi/content/full/3/114/ra23/dc1 Supplementary Materials for Regulation of Zap70 Expression During Thymocyte Development Enables Temporal Separation of CD4 and CD8 Repertoire Selection
www.sciencesignaling.org/cgi/content/full/3/114/ra23/dc1 Supplementary Materials for Regulation of Zap70 Expression During Thymocyte Development Enables Temporal Separation of CD4 and CD8 Repertoire Selection
Supplemental Information. T Cells Enhance Autoimmunity by Restraining Regulatory T Cell Responses via an Interleukin-23-Dependent Mechanism
 Immunity, Volume 33 Supplemental Information T Cells Enhance Autoimmunity by Restraining Regulatory T Cell Responses via an Interleukin-23-Dependent Mechanism Franziska Petermann, Veit Rothhammer, Malte
Immunity, Volume 33 Supplemental Information T Cells Enhance Autoimmunity by Restraining Regulatory T Cell Responses via an Interleukin-23-Dependent Mechanism Franziska Petermann, Veit Rothhammer, Malte
Shiv Pillai Ragon Institute, Massachusetts General Hospital Harvard Medical School
 CTLs, Natural Killers and NKTs 1 Shiv Pillai Ragon Institute, Massachusetts General Hospital Harvard Medical School CTL inducing tumor apoptosis 3 Lecture outline CD8 + Cytotoxic T lymphocytes (CTL) Activation/differentiation
CTLs, Natural Killers and NKTs 1 Shiv Pillai Ragon Institute, Massachusetts General Hospital Harvard Medical School CTL inducing tumor apoptosis 3 Lecture outline CD8 + Cytotoxic T lymphocytes (CTL) Activation/differentiation
RAISON D ETRE OF THE IMMUNE SYSTEM:
 RAISON D ETRE OF THE IMMUNE SYSTEM: To Distinguish Self from Non-Self Thereby Protecting Us From Our Hostile Environment. Innate Immunity Acquired Immunity Innate immunity: (Antigen nonspecific) defense
RAISON D ETRE OF THE IMMUNE SYSTEM: To Distinguish Self from Non-Self Thereby Protecting Us From Our Hostile Environment. Innate Immunity Acquired Immunity Innate immunity: (Antigen nonspecific) defense
Supplemental Materials for. Effects of sphingosine-1-phosphate receptor 1 phosphorylation in response to. FTY720 during neuroinflammation
 Supplemental Materials for Effects of sphingosine-1-phosphate receptor 1 phosphorylation in response to FTY7 during neuroinflammation This file includes: Supplemental Table 1. EAE clinical parameters of
Supplemental Materials for Effects of sphingosine-1-phosphate receptor 1 phosphorylation in response to FTY7 during neuroinflammation This file includes: Supplemental Table 1. EAE clinical parameters of
T Cell Activation, Costimulation and Regulation
 1 T Cell Activation, Costimulation and Regulation Abul K. Abbas, MD University of California San Francisco 2 Lecture outline T cell antigen recognition and activation Costimulation, the B7:CD28 family
1 T Cell Activation, Costimulation and Regulation Abul K. Abbas, MD University of California San Francisco 2 Lecture outline T cell antigen recognition and activation Costimulation, the B7:CD28 family
Examples of questions for Cellular Immunology/Cellular Biology and Immunology
 Examples of questions for Cellular Immunology/Cellular Biology and Immunology Each student gets a set of 6 questions, so that each set contains different types of questions and that the set of questions
Examples of questions for Cellular Immunology/Cellular Biology and Immunology Each student gets a set of 6 questions, so that each set contains different types of questions and that the set of questions
COURSE: Medical Microbiology, PAMB 650/720 - Fall 2008 Lecture 16
 COURSE: Medical Microbiology, PAMB 650/720 - Fall 2008 Lecture 16 Tumor Immunology M. Nagarkatti Teaching Objectives: Introduction to Cancer Immunology Know the antigens expressed by cancer cells Understand
COURSE: Medical Microbiology, PAMB 650/720 - Fall 2008 Lecture 16 Tumor Immunology M. Nagarkatti Teaching Objectives: Introduction to Cancer Immunology Know the antigens expressed by cancer cells Understand
Antigen Presentation and T Lymphocyte Activation. Abul K. Abbas UCSF. FOCiS
 1 Antigen Presentation and T Lymphocyte Activation Abul K. Abbas UCSF FOCiS 2 Lecture outline Dendritic cells and antigen presentation The role of the MHC T cell activation Costimulation, the B7:CD28 family
1 Antigen Presentation and T Lymphocyte Activation Abul K. Abbas UCSF FOCiS 2 Lecture outline Dendritic cells and antigen presentation The role of the MHC T cell activation Costimulation, the B7:CD28 family
T cell maturation. T-cell Maturation. What allows T cell maturation?
 T-cell Maturation What allows T cell maturation? Direct contact with thymic epithelial cells Influence of thymic hormones Growth factors (cytokines, CSF) T cell maturation T cell progenitor DN DP SP 2ry
T-cell Maturation What allows T cell maturation? Direct contact with thymic epithelial cells Influence of thymic hormones Growth factors (cytokines, CSF) T cell maturation T cell progenitor DN DP SP 2ry
1. Overview of Adaptive Immunity
 Chapter 17A: Adaptive Immunity Part I 1. Overview of Adaptive Immunity 2. T and B Cell Production 3. Antigens & Antigen Presentation 4. Helper T cells 1. Overview of Adaptive Immunity The Nature of Adaptive
Chapter 17A: Adaptive Immunity Part I 1. Overview of Adaptive Immunity 2. T and B Cell Production 3. Antigens & Antigen Presentation 4. Helper T cells 1. Overview of Adaptive Immunity The Nature of Adaptive
Nature Immunology: doi: /ni Supplementary Figure 1. Gene expression profile of CD4 + T cells and CTL responses in Bcl6-deficient mice.
 Supplementary Figure 1 Gene expression profile of CD4 + T cells and CTL responses in Bcl6-deficient mice. (a) Gene expression profile in the resting CD4 + T cells were analyzed by an Affymetrix microarray
Supplementary Figure 1 Gene expression profile of CD4 + T cells and CTL responses in Bcl6-deficient mice. (a) Gene expression profile in the resting CD4 + T cells were analyzed by an Affymetrix microarray
Scott Abrams, Ph.D. Professor of Oncology, x4375 Kuby Immunology SEVENTH EDITION
 Scott Abrams, Ph.D. Professor of Oncology, x4375 scott.abrams@roswellpark.org Kuby Immunology SEVENTH EDITION CHAPTER 11 T-Cell Activation, Differentiation, and Memory Copyright 2013 by W. H. Freeman and
Scott Abrams, Ph.D. Professor of Oncology, x4375 scott.abrams@roswellpark.org Kuby Immunology SEVENTH EDITION CHAPTER 11 T-Cell Activation, Differentiation, and Memory Copyright 2013 by W. H. Freeman and
Cell Mediated Immunity CELL MEDIATED IMMUNITY. Basic Elements of Cell Mediated Immunity (CMI) Antibody-dependent cell-mediated cytotoxicity (ADCC)
 Chapter 16 CELL MEDIATED IMMUNITY Cell Mediated Immunity Also known as Cellular Immunity or CMI The effector phase T cells Specificity for immune recognition reactions TH provide cytokines CTLs do the
Chapter 16 CELL MEDIATED IMMUNITY Cell Mediated Immunity Also known as Cellular Immunity or CMI The effector phase T cells Specificity for immune recognition reactions TH provide cytokines CTLs do the
Shenghua Zhou, Rong Ou, Lei Huang, and Demetrius Moskophidis*
 JOURNAL OF VIROLOGY, Jan. 2002, p. 829 840 Vol. 76, No. 2 0022-538X/02/$04.00 0 DOI: 10.1128/JVI.76.2.829 840.2002 Copyright 2002, American Society for Microbiology. All Rights Reserved. Critical Role
JOURNAL OF VIROLOGY, Jan. 2002, p. 829 840 Vol. 76, No. 2 0022-538X/02/$04.00 0 DOI: 10.1128/JVI.76.2.829 840.2002 Copyright 2002, American Society for Microbiology. All Rights Reserved. Critical Role
Adaptive immune responses: T cell-mediated immunity
 MICR2209 Adaptive immune responses: T cell-mediated immunity Dr Allison Imrie allison.imrie@uwa.edu.au 1 Synopsis: In this lecture we will discuss the T-cell mediated immune response, how it is activated,
MICR2209 Adaptive immune responses: T cell-mediated immunity Dr Allison Imrie allison.imrie@uwa.edu.au 1 Synopsis: In this lecture we will discuss the T-cell mediated immune response, how it is activated,
Optimizing Intracellular Flow Cytometry:
 Optimizing Intracellular Flow Cytometry: Simultaneous Detection of Cytokines and Transcription Factors An encore presentation by Jurg Rohrer, PhD, BD Biosciences 10.26.10 Outline Introduction Cytokines
Optimizing Intracellular Flow Cytometry: Simultaneous Detection of Cytokines and Transcription Factors An encore presentation by Jurg Rohrer, PhD, BD Biosciences 10.26.10 Outline Introduction Cytokines
C. Incorrect! MHC class I molecules are not involved in the process of bridging in ADCC.
 Immunology - Problem Drill 13: T- Cell Mediated Immunity Question No. 1 of 10 1. During Antibody-dependent cell mediated cytotoxicity (ADCC), the antibody acts like a bridge between the specific antigen
Immunology - Problem Drill 13: T- Cell Mediated Immunity Question No. 1 of 10 1. During Antibody-dependent cell mediated cytotoxicity (ADCC), the antibody acts like a bridge between the specific antigen
Eosinophils are required. for the maintenance of plasma cells in the bone marrow
 Eosinophils are required for the maintenance of plasma cells in the bone marrow Van Trung Chu, Anja Fröhlich, Gudrun Steinhauser, Tobias Scheel, Toralf Roch, Simon Fillatreau, James J. Lee, Max Löhning
Eosinophils are required for the maintenance of plasma cells in the bone marrow Van Trung Chu, Anja Fröhlich, Gudrun Steinhauser, Tobias Scheel, Toralf Roch, Simon Fillatreau, James J. Lee, Max Löhning
Immunological alterations in mice irradiated with low doses
 Immunological alterations in mice irradiated with low doses "Frédéric Joliot-Curie" National Research Institute for Radiobiology and Radiohygiene, Budapest, Hungary The structure of the immune system INNATE
Immunological alterations in mice irradiated with low doses "Frédéric Joliot-Curie" National Research Institute for Radiobiology and Radiohygiene, Budapest, Hungary The structure of the immune system INNATE
ACTIVATION OF T LYMPHOCYTES AND CELL MEDIATED IMMUNITY
 ACTIVATION OF T LYMPHOCYTES AND CELL MEDIATED IMMUNITY The recognition of specific antigen by naïve T cell induces its own activation and effector phases. T helper cells recognize peptide antigens through
ACTIVATION OF T LYMPHOCYTES AND CELL MEDIATED IMMUNITY The recognition of specific antigen by naïve T cell induces its own activation and effector phases. T helper cells recognize peptide antigens through
NKTR-255: Accessing IL-15 Therapeutic Potential through Robust and Sustained Engagement of Innate and Adaptive Immunity
 NKTR-255: Accessing IL-15 Therapeutic Potential through Robust and Sustained Engagement of Innate and Adaptive Immunity Peiwen Kuo Scientist, Research Biology Nektar Therapeutics August 31 st, 2018 Emerging
NKTR-255: Accessing IL-15 Therapeutic Potential through Robust and Sustained Engagement of Innate and Adaptive Immunity Peiwen Kuo Scientist, Research Biology Nektar Therapeutics August 31 st, 2018 Emerging
Supplementary Figure 1. mrna expression of chitinase and chitinase-like protein in splenic immune cells. Each splenic immune cell population was
 Supplementary Figure 1. mrna expression of chitinase and chitinase-like protein in splenic immune cells. Each splenic immune cell population was sorted by FACS. Surface markers for sorting were CD11c +
Supplementary Figure 1. mrna expression of chitinase and chitinase-like protein in splenic immune cells. Each splenic immune cell population was sorted by FACS. Surface markers for sorting were CD11c +
TITLE: MODULATION OF T CELL TOLERANCE IN A MURINE MODEL FOR IMMUNOTHERAPY OF PROSTATIC ADENOCARCINOMA
 AD Award Number: DAMD17-01-1-0085 TITLE: MODULATION OF T CELL TOLERANCE IN A MURINE MODEL FOR IMMUNOTHERAPY OF PROSTATIC ADENOCARCINOMA PRINCIPAL INVESTIGATOR: ARTHUR A HURWITZ, Ph.d. CONTRACTING ORGANIZATION:
AD Award Number: DAMD17-01-1-0085 TITLE: MODULATION OF T CELL TOLERANCE IN A MURINE MODEL FOR IMMUNOTHERAPY OF PROSTATIC ADENOCARCINOMA PRINCIPAL INVESTIGATOR: ARTHUR A HURWITZ, Ph.d. CONTRACTING ORGANIZATION:
Test Bank for Basic Immunology Functions and Disorders of the Immune System 4th Edition by Abbas
 Test Bank for Basic Immunology Functions and Disorders of the Immune System 4th Edition by Abbas Chapter 04: Antigen Recognition in the Adaptive Immune System Test Bank MULTIPLE CHOICE 1. Most T lymphocytes
Test Bank for Basic Immunology Functions and Disorders of the Immune System 4th Edition by Abbas Chapter 04: Antigen Recognition in the Adaptive Immune System Test Bank MULTIPLE CHOICE 1. Most T lymphocytes
Suppl Video: Tumor cells (green) and monocytes (white) are seeded on a confluent endothelial
 Supplementary Information Häuselmann et al. Monocyte induction of E-selectin-mediated endothelial activation releases VE-cadherin junctions to promote tumor cell extravasation in the metastasis cascade
Supplementary Information Häuselmann et al. Monocyte induction of E-selectin-mediated endothelial activation releases VE-cadherin junctions to promote tumor cell extravasation in the metastasis cascade
Effector mechanisms of cell-mediated immunity: Properties of effector, memory and regulatory T cells
 ICI Basic Immunology course Effector mechanisms of cell-mediated immunity: Properties of effector, memory and regulatory T cells Abul K. Abbas, MD UCSF Stages in the development of T cell responses: induction
ICI Basic Immunology course Effector mechanisms of cell-mediated immunity: Properties of effector, memory and regulatory T cells Abul K. Abbas, MD UCSF Stages in the development of T cell responses: induction
Immunity and Cancer. Doriana Fruci. Lab di Immuno-Oncologia
 Immunity and Cancer Doriana Fruci Lab di Immuno-Oncologia Immune System is a network of cells, tissues and organs that work together to defend the body against attacks of foreign invaders (pathogens, cancer
Immunity and Cancer Doriana Fruci Lab di Immuno-Oncologia Immune System is a network of cells, tissues and organs that work together to defend the body against attacks of foreign invaders (pathogens, cancer
Cover Page. The handle holds various files of this Leiden University dissertation.
 Cover Page The handle http://hdl.handle.net/1887/23854 holds various files of this Leiden University dissertation. Author: Marel, Sander van der Title: Gene and cell therapy based treatment strategies
Cover Page The handle http://hdl.handle.net/1887/23854 holds various files of this Leiden University dissertation. Author: Marel, Sander van der Title: Gene and cell therapy based treatment strategies
Cell isolation. Spleen and lymph nodes (axillary, inguinal) were removed from mice
 Supplementary Methods: Cell isolation. Spleen and lymph nodes (axillary, inguinal) were removed from mice and gently meshed in DMEM containing 10% FBS to prepare for single cell suspensions. CD4 + CD25
Supplementary Methods: Cell isolation. Spleen and lymph nodes (axillary, inguinal) were removed from mice and gently meshed in DMEM containing 10% FBS to prepare for single cell suspensions. CD4 + CD25
of whole cell cultures in U-bottomed wells of a 96-well plate are shown. 2
 Supplementary online material Supplementary figure legends Supplementary Figure 1 Exposure to T reg cells causes loss of T resp cells in co-cultures. T resp cells were stimulated with CD3+CD28 alone or
Supplementary online material Supplementary figure legends Supplementary Figure 1 Exposure to T reg cells causes loss of T resp cells in co-cultures. T resp cells were stimulated with CD3+CD28 alone or
Supplementary Figure 1 IL-27 IL
 Tim-3 Supplementary Figure 1 Tc0 49.5 0.6 Tc1 63.5 0.84 Un 49.8 0.16 35.5 0.16 10 4 61.2 5.53 10 3 64.5 5.66 10 2 10 1 10 0 31 2.22 10 0 10 1 10 2 10 3 10 4 IL-10 28.2 1.69 IL-27 Supplementary Figure 1.
Tim-3 Supplementary Figure 1 Tc0 49.5 0.6 Tc1 63.5 0.84 Un 49.8 0.16 35.5 0.16 10 4 61.2 5.53 10 3 64.5 5.66 10 2 10 1 10 0 31 2.22 10 0 10 1 10 2 10 3 10 4 IL-10 28.2 1.69 IL-27 Supplementary Figure 1.
Optimizing Intracellular Flow Cytometry
 Optimizing Intracellular Flow Cytometry Detection of Cytokines, Transcription Factors, and Phosphoprotein by Flow Cytometry Presented by Erika O Donnell, PhD, BD Biosciences 23-14876-00 Outline Basic principles
Optimizing Intracellular Flow Cytometry Detection of Cytokines, Transcription Factors, and Phosphoprotein by Flow Cytometry Presented by Erika O Donnell, PhD, BD Biosciences 23-14876-00 Outline Basic principles
TITLE: Development of Antigen Presenting Cells for adoptive immunotherapy in prostate cancer
 AD Award Number: W8-XWH-5-- TITLE: Development of Antigen Presenting Cells for adoptive immunotherapy in prostate cancer PRINCIPAL INVESTIGATOR: Mathias Oelke, Ph.D. CONTRACTING ORGANIZATION: Johns Hopkins
AD Award Number: W8-XWH-5-- TITLE: Development of Antigen Presenting Cells for adoptive immunotherapy in prostate cancer PRINCIPAL INVESTIGATOR: Mathias Oelke, Ph.D. CONTRACTING ORGANIZATION: Johns Hopkins
Adaptive Immunity. Jeffrey K. Actor, Ph.D. MSB 2.214,
 Adaptive Immunity Jeffrey K. Actor, Ph.D. MSB 2.214, 500-5344 Lecture Objectives: Understand role of various molecules including cytokines, chemokines, costimulatory and adhesion molecules in the development
Adaptive Immunity Jeffrey K. Actor, Ph.D. MSB 2.214, 500-5344 Lecture Objectives: Understand role of various molecules including cytokines, chemokines, costimulatory and adhesion molecules in the development
Supplementary Materials for
 immunology.sciencemag.org/cgi/content/full/2/16/eaan6049/dc1 Supplementary Materials for Enzymatic synthesis of core 2 O-glycans governs the tissue-trafficking potential of memory CD8 + T cells Jossef
immunology.sciencemag.org/cgi/content/full/2/16/eaan6049/dc1 Supplementary Materials for Enzymatic synthesis of core 2 O-glycans governs the tissue-trafficking potential of memory CD8 + T cells Jossef
Detailed step-by-step operating procedures for NK cell and CTL degranulation assays
 Supplemental methods Detailed step-by-step operating procedures for NK cell and CTL degranulation assays Materials PBMC isolated from patients, relatives and healthy donors as control K562 cells (ATCC,
Supplemental methods Detailed step-by-step operating procedures for NK cell and CTL degranulation assays Materials PBMC isolated from patients, relatives and healthy donors as control K562 cells (ATCC,
CONTRACTING ORGANIZATION: Johns Hopkins University School of Medicine Baltimore, MD 21205
 AD Award Number: DAMD7---7 TITLE: Development of Artificial Antigen Presenting Cells for Prostate Cancer Immunotherapy PRINCIPAL INVESTIGATOR: Jonathan P. Schneck, M.D., Ph.D. Mathias Oelke, Ph.D. CONTRACTING
AD Award Number: DAMD7---7 TITLE: Development of Artificial Antigen Presenting Cells for Prostate Cancer Immunotherapy PRINCIPAL INVESTIGATOR: Jonathan P. Schneck, M.D., Ph.D. Mathias Oelke, Ph.D. CONTRACTING
Newly Recognized Components of the Innate Immune System
 Newly Recognized Components of the Innate Immune System NOD Proteins: Intracellular Peptidoglycan Sensors NOD-1 NOD-2 Nod Protein LRR; Ligand Recognition CARD RICK I-κB p50 p65 NF-κB Polymorphisms in Nod-2
Newly Recognized Components of the Innate Immune System NOD Proteins: Intracellular Peptidoglycan Sensors NOD-1 NOD-2 Nod Protein LRR; Ligand Recognition CARD RICK I-κB p50 p65 NF-κB Polymorphisms in Nod-2
Optimizing Intracellular Flow Cytometry:
 Optimizing Intracellular Flow Cytometry: Simultaneous Detection of Cytokines and Transcription Factors Presented by Jurg Rohrer, PhD, BD Biosciences 23-10780-00 Outline Introduction Cytokines Transcription
Optimizing Intracellular Flow Cytometry: Simultaneous Detection of Cytokines and Transcription Factors Presented by Jurg Rohrer, PhD, BD Biosciences 23-10780-00 Outline Introduction Cytokines Transcription
Resolution of a chronic viral infection after interleukin-10 receptor blockade
 ARTICLE Resolution of a chronic viral infection after interleukin-10 receptor blockade Mette Ejrnaes, 1 Christophe M. Filippi, 1 Marianne M. Martinic, 1 Eleanor M. Ling, 1 Lisa M. Togher, 1 Shane Crotty,
ARTICLE Resolution of a chronic viral infection after interleukin-10 receptor blockade Mette Ejrnaes, 1 Christophe M. Filippi, 1 Marianne M. Martinic, 1 Eleanor M. Ling, 1 Lisa M. Togher, 1 Shane Crotty,
Direct ex vivo characterization of human antigen-specific CD154 + CD4 + T cells Rapid antigen-reactive T cell enrichment (Rapid ARTE)
 Direct ex vivo characterization of human antigen-specific CD154 + CD4 + T cells Rapid antigen-reactive T cell enrichment (Rapid ARTE) Introduction Workflow Antigen (ag)-specific T cells play a central
Direct ex vivo characterization of human antigen-specific CD154 + CD4 + T cells Rapid antigen-reactive T cell enrichment (Rapid ARTE) Introduction Workflow Antigen (ag)-specific T cells play a central
Signal 3 requirement for memory CD8 1 T-cell activation is determined by the infectious pathogen
 3176 DOI 1.12/eji.21141537 Eur. J. Immunol. 211. 41: 3176 3186 Signal 3 requirement for memory CD8 1 T-cell activation is determined by the infectious pathogen Selina J. Keppler 1,2 and Peter Aichele 1
3176 DOI 1.12/eji.21141537 Eur. J. Immunol. 211. 41: 3176 3186 Signal 3 requirement for memory CD8 1 T-cell activation is determined by the infectious pathogen Selina J. Keppler 1,2 and Peter Aichele 1
Supporting Information
 Supporting Information lpek et al. 1.173/pnas.1121217 SI Materials and Methods Mice. cell knockout, inos / (Taconic arms), Rag1 /, INγR /, and IL-12p4 / mice (The Jackson Laboratory) were maintained and/or
Supporting Information lpek et al. 1.173/pnas.1121217 SI Materials and Methods Mice. cell knockout, inos / (Taconic arms), Rag1 /, INγR /, and IL-12p4 / mice (The Jackson Laboratory) were maintained and/or
Recombinant Vaccinia Virus-Induced T-Cell Immunity: Quantitation of the Response to the Virus Vector and the Foreign Epitope
 JOURNAL OF VIROLOGY, Apr. 2002, p. 3329 3337 Vol. 76, No. 7 0022-538X/02/$04.00 0 DOI: 10.1128/JVI.76.7.3329 3337.2002 Copyright 2002, American Society for Microbiology. All Rights Reserved. Recombinant
JOURNAL OF VIROLOGY, Apr. 2002, p. 3329 3337 Vol. 76, No. 7 0022-538X/02/$04.00 0 DOI: 10.1128/JVI.76.7.3329 3337.2002 Copyright 2002, American Society for Microbiology. All Rights Reserved. Recombinant
Ex vivo Human Antigen-specific T Cell Proliferation and Degranulation Willemijn Hobo 1, Wieger Norde 1 and Harry Dolstra 2*
 Ex vivo Human Antigen-specific T Cell Proliferation and Degranulation Willemijn Hobo 1, Wieger Norde 1 and Harry Dolstra 2* 1 Department of Laboratory Medicine - Laboratory of Hematology, Radboud University
Ex vivo Human Antigen-specific T Cell Proliferation and Degranulation Willemijn Hobo 1, Wieger Norde 1 and Harry Dolstra 2* 1 Department of Laboratory Medicine - Laboratory of Hematology, Radboud University
Supplementary Figures
 Supplementary Figures Supplementary Fig. 1. Surface thiol groups and reduction of activated T cells. (a) Activated CD8 + T-cells have high expression levels of free thiol groups on cell surface proteins.
Supplementary Figures Supplementary Fig. 1. Surface thiol groups and reduction of activated T cells. (a) Activated CD8 + T-cells have high expression levels of free thiol groups on cell surface proteins.
Immunology. T-Lymphocytes. 16. Oktober 2014, Ruhr-Universität Bochum Karin Peters,
 Immunology T-Lymphocytes 16. Oktober 2014, Ruhr-Universität Bochum Karin Peters, karin.peters@rub.de The role of T-effector cells in the immune response against microbes cellular immunity humoral immunity
Immunology T-Lymphocytes 16. Oktober 2014, Ruhr-Universität Bochum Karin Peters, karin.peters@rub.de The role of T-effector cells in the immune response against microbes cellular immunity humoral immunity
TITLE: Development of Antigen Presenting Cells for adoptive immunotherapy in prostate cancer
 AD Award Number: W8-XWH-5-- TITLE: Development of Antigen Presenting Cells for adoptive immunotherapy in prostate cancer PRINCIPAL INVESTIGATOR: Mathias Oelke,. CONTRACTING ORGANIZATION: Johns Hopkins
AD Award Number: W8-XWH-5-- TITLE: Development of Antigen Presenting Cells for adoptive immunotherapy in prostate cancer PRINCIPAL INVESTIGATOR: Mathias Oelke,. CONTRACTING ORGANIZATION: Johns Hopkins
Lecture 6. Burr BIO 4353/6345 HIV/AIDS. Tetramer staining of T cells (CTL s) Andrew McMichael seminar: Background
 Lecture 6 Burr BIO 4353/6345 HIV/AIDS Andrew McMichael seminar: Background Tetramer staining of T cells (CTL s) 1. Vβ 19: There are 52 T cell receptor (TCR) Vβ gene segments in germ line DNA (See following
Lecture 6 Burr BIO 4353/6345 HIV/AIDS Andrew McMichael seminar: Background Tetramer staining of T cells (CTL s) 1. Vβ 19: There are 52 T cell receptor (TCR) Vβ gene segments in germ line DNA (See following
D CD8 T cell number (x10 6 )
 IFNγ Supplemental Figure 1. CD T cell number (x1 6 ) 18 15 1 9 6 3 CD CD T cells CD6L C CD5 CD T cells CD6L D CD8 T cell number (x1 6 ) 1 8 6 E CD CD8 T cells CD6L F Log(1)CFU/g Feces 1 8 6 p
IFNγ Supplemental Figure 1. CD T cell number (x1 6 ) 18 15 1 9 6 3 CD CD T cells CD6L C CD5 CD T cells CD6L D CD8 T cell number (x1 6 ) 1 8 6 E CD CD8 T cells CD6L F Log(1)CFU/g Feces 1 8 6 p
Supplementary Figure S1. PTPN2 levels are not altered in proliferating CD8+ T cells. Lymph node (LN) CD8+ T cells from C57BL/6 mice were stained with
 Supplementary Figure S1. PTPN2 levels are not altered in proliferating CD8+ T cells. Lymph node (LN) CD8+ T cells from C57BL/6 mice were stained with CFSE and stimulated with plate-bound α-cd3ε (10µg/ml)
Supplementary Figure S1. PTPN2 levels are not altered in proliferating CD8+ T cells. Lymph node (LN) CD8+ T cells from C57BL/6 mice were stained with CFSE and stimulated with plate-bound α-cd3ε (10µg/ml)
Supplementary Figure 1. Generation of knockin mice expressing L-selectinN138G. (a) Schematics of the Sellg allele (top), the targeting vector, the
 Supplementary Figure 1. Generation of knockin mice expressing L-selectinN138G. (a) Schematics of the Sellg allele (top), the targeting vector, the targeted allele in ES cells, and the mutant allele in
Supplementary Figure 1. Generation of knockin mice expressing L-selectinN138G. (a) Schematics of the Sellg allele (top), the targeting vector, the targeted allele in ES cells, and the mutant allele in
Cell-mediated Immunity
 Cellular & Molecular Immunology Cell-mediated Immunity Nicholas M. Ponzio, Ph.D. Department of Pathology & Laboratory Medicine April 6, 2009 Today s Presentation: Overview Cellular Interactions In Humoral
Cellular & Molecular Immunology Cell-mediated Immunity Nicholas M. Ponzio, Ph.D. Department of Pathology & Laboratory Medicine April 6, 2009 Today s Presentation: Overview Cellular Interactions In Humoral
IMMUNE CELL SURFACE RECEPTORS AND THEIR FUNCTIONS
 LECTURE: 07 Title: IMMUNE CELL SURFACE RECEPTORS AND THEIR FUNCTIONS LEARNING OBJECTIVES: The student should be able to: The chemical nature of the cellular surface receptors. Define the location of the
LECTURE: 07 Title: IMMUNE CELL SURFACE RECEPTORS AND THEIR FUNCTIONS LEARNING OBJECTIVES: The student should be able to: The chemical nature of the cellular surface receptors. Define the location of the
RAISON D ETRE OF THE IMMUNE SYSTEM:
 RAISON D ETRE OF THE IMMUNE SYSTEM: To Distinguish Self from Non-Self Thereby Protecting Us From Our Hostile Environment. Innate Immunity Adaptive Immunity Innate immunity: (Antigen - nonspecific) defense
RAISON D ETRE OF THE IMMUNE SYSTEM: To Distinguish Self from Non-Self Thereby Protecting Us From Our Hostile Environment. Innate Immunity Adaptive Immunity Innate immunity: (Antigen - nonspecific) defense
Critical Role for Alpha/Beta and Gamma Interferons in Persistence of Lymphocytic Choriomeningitis Virus by Clonal Exhaustion of Cytotoxic T Cells
 JOURNAL OF VIROLOGY, Sept. 2001, p. 8407 8423 Vol. 75, No. 18 0022-538X/01/$04.00 0 DOI: 10.1128/JVI.75.18.8407 8423.2001 Copyright 2001, American Society for Microbiology. All Rights Reserved. Critical
JOURNAL OF VIROLOGY, Sept. 2001, p. 8407 8423 Vol. 75, No. 18 0022-538X/01/$04.00 0 DOI: 10.1128/JVI.75.18.8407 8423.2001 Copyright 2001, American Society for Microbiology. All Rights Reserved. Critical
Effective activity of cytokine-induced killer cells against autologous metastatic melanoma including cells with stemness features
 Effective activity of cytokine-induced killer cells against autologous metastatic melanoma including cells with stemness features Loretta Gammaitoni, Lidia Giraudo, Valeria Leuci, et al. Clin Cancer Res
Effective activity of cytokine-induced killer cells against autologous metastatic melanoma including cells with stemness features Loretta Gammaitoni, Lidia Giraudo, Valeria Leuci, et al. Clin Cancer Res
Recombinant adeno-associated virus vectors induce functionally impaired transgene product specific CD8 + T cells in mice
 Recombinant adeno-associated virus vectors induce functionally impaired transgene product specific CD8 + T cells in mice Shih-Wen Lin,, Marcio O. Lasaro, Hildegund C.J. Ertl J Clin Invest. 2007;117(12):3958-3970.
Recombinant adeno-associated virus vectors induce functionally impaired transgene product specific CD8 + T cells in mice Shih-Wen Lin,, Marcio O. Lasaro, Hildegund C.J. Ertl J Clin Invest. 2007;117(12):3958-3970.
ECM1 controls T H 2 cell egress from lymph nodes through re-expression of S1P 1
 ZH, Li et al, page 1 ECM1 controls T H 2 cell egress from lymph nodes through re-expression of S1P 1 Zhenhu Li 1,4,Yuan Zhang 1,4, Zhiduo Liu 1, Xiaodong Wu 1, Yuhan Zheng 1, Zhiyun Tao 1, Kairui Mao 1,
ZH, Li et al, page 1 ECM1 controls T H 2 cell egress from lymph nodes through re-expression of S1P 1 Zhenhu Li 1,4,Yuan Zhang 1,4, Zhiduo Liu 1, Xiaodong Wu 1, Yuhan Zheng 1, Zhiyun Tao 1, Kairui Mao 1,
Supplementary Figure 1. Efficient DC depletion in CD11c.DOG transgenic mice
 Supplementary Figure 1. Efficient DC depletion in CD11c.DOG transgenic mice (a) CD11c.DOG transgenic mice (tg) were treated with 8 ng/g body weight (b.w.) diphtheria toxin (DT) i.p. on day -1 and every
Supplementary Figure 1. Efficient DC depletion in CD11c.DOG transgenic mice (a) CD11c.DOG transgenic mice (tg) were treated with 8 ng/g body weight (b.w.) diphtheria toxin (DT) i.p. on day -1 and every
and follicular helper T cells is Egr2-dependent. (a) Diagrammatic representation of the
 Supplementary Figure 1. LAG3 + Treg-mediated regulation of germinal center B cells and follicular helper T cells is Egr2-dependent. (a) Diagrammatic representation of the experimental protocol for the
Supplementary Figure 1. LAG3 + Treg-mediated regulation of germinal center B cells and follicular helper T cells is Egr2-dependent. (a) Diagrammatic representation of the experimental protocol for the
T Cell Effector Mechanisms I: B cell Help & DTH
 T Cell Effector Mechanisms I: B cell Help & DTH Ned Braunstein, MD The Major T Cell Subsets p56 lck + T cells γ δ ε ζ ζ p56 lck CD8+ T cells γ δ ε ζ ζ Cα Cβ Vα Vβ CD3 CD8 Cα Cβ Vα Vβ CD3 MHC II peptide
T Cell Effector Mechanisms I: B cell Help & DTH Ned Braunstein, MD The Major T Cell Subsets p56 lck + T cells γ δ ε ζ ζ p56 lck CD8+ T cells γ δ ε ζ ζ Cα Cβ Vα Vβ CD3 CD8 Cα Cβ Vα Vβ CD3 MHC II peptide
Supporting Information
 Supporting Information Valkenburg et al. 10.1073/pnas.1403684111 SI Materials and Methods ELISA and Microneutralization. Sera were treated with Receptor Destroying Enzyme II (RDE II, Accurate) before ELISA
Supporting Information Valkenburg et al. 10.1073/pnas.1403684111 SI Materials and Methods ELISA and Microneutralization. Sera were treated with Receptor Destroying Enzyme II (RDE II, Accurate) before ELISA
T-Pharmacytes for the Targeted Eradication of HIV Reservoirs
 T-Pharmacytes for the Targeted Eradication of HIV Reservoirs Walker Lab Rachel O Connor Alicja Trocha Dan Karel Irvine Lab Stefanie Mueller Humanized Mouse Core Vlad Vrbanac Andy Tager Todd Allen Darrell
T-Pharmacytes for the Targeted Eradication of HIV Reservoirs Walker Lab Rachel O Connor Alicja Trocha Dan Karel Irvine Lab Stefanie Mueller Humanized Mouse Core Vlad Vrbanac Andy Tager Todd Allen Darrell
Supplemental Figure 1. Signature gene expression in in vitro differentiated Th0, Th1, Th2, Th17 and Treg cells. (A) Naïve CD4 + T cells were cultured
 Supplemental Figure 1. Signature gene expression in in vitro differentiated Th0, Th1, Th2, Th17 and Treg cells. (A) Naïve CD4 + T cells were cultured under Th0, Th1, Th2, Th17, and Treg conditions. mrna
Supplemental Figure 1. Signature gene expression in in vitro differentiated Th0, Th1, Th2, Th17 and Treg cells. (A) Naïve CD4 + T cells were cultured under Th0, Th1, Th2, Th17, and Treg conditions. mrna
The Role of CD4 T Cells in the Pathogenesis of Murine AIDS
 JOURNAL OF VIROLOGY, June 2006, p. 5777 5789 Vol. 80, No. 12 0022-538X/06/$08.00 0 doi:10.1128/jvi.02711-05 Copyright 2006, American Society for Microbiology. All Rights Reserved. The Role of CD4 T Cells
JOURNAL OF VIROLOGY, June 2006, p. 5777 5789 Vol. 80, No. 12 0022-538X/06/$08.00 0 doi:10.1128/jvi.02711-05 Copyright 2006, American Society for Microbiology. All Rights Reserved. The Role of CD4 T Cells
Supplementary Figures
 Inhibition of Pulmonary Anti Bacterial Defense by IFN γ During Recovery from Influenza Infection By Keer Sun and Dennis W. Metzger Supplementary Figures d a Ly6G Percentage survival f 1 75 5 1 25 1 5 1
Inhibition of Pulmonary Anti Bacterial Defense by IFN γ During Recovery from Influenza Infection By Keer Sun and Dennis W. Metzger Supplementary Figures d a Ly6G Percentage survival f 1 75 5 1 25 1 5 1
W/T Itgam -/- F4/80 CD115. F4/80 hi CD115 + F4/80 + CD115 +
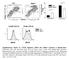 F4/8 % in the peritoneal lavage 6 4 2 p=.15 n.s p=.76 CD115 F4/8 hi CD115 + F4/8 + CD115 + F4/8 hi CD115 + F4/8 + CD115 + MHCII MHCII Supplementary Figure S1. CD11b deficiency affects the cellular responses
F4/8 % in the peritoneal lavage 6 4 2 p=.15 n.s p=.76 CD115 F4/8 hi CD115 + F4/8 + CD115 + F4/8 hi CD115 + F4/8 + CD115 + MHCII MHCII Supplementary Figure S1. CD11b deficiency affects the cellular responses
Activation-induced cell death (AICD) 1 in CD4 Th has
 Unequal Death in T Helper Cell (Th)1 and Th2 Effectors: Th1, but not Th2, Effectors Undergo Rapid Fas/FasL-mediated Apoptosis By Xiaohong Zhang,* Thomas Brunner, Laura Carter,* Richard W. Dutton,* Paul
Unequal Death in T Helper Cell (Th)1 and Th2 Effectors: Th1, but not Th2, Effectors Undergo Rapid Fas/FasL-mediated Apoptosis By Xiaohong Zhang,* Thomas Brunner, Laura Carter,* Richard W. Dutton,* Paul
Supplementary Table; Supplementary Figures and legends S1-S21; Supplementary Materials and Methods
 Silva et al. PTEN posttranslational inactivation and hyperactivation of the PI3K/Akt pathway sustain primary T cell leukemia viability Supplementary Table; Supplementary Figures and legends S1-S21; Supplementary
Silva et al. PTEN posttranslational inactivation and hyperactivation of the PI3K/Akt pathway sustain primary T cell leukemia viability Supplementary Table; Supplementary Figures and legends S1-S21; Supplementary
L-selectin Is Essential for Delivery of Activated CD8 + T Cells to Virus-Infected Organs for Protective Immunity
 Cell Reports Supplemental Information L-selectin Is Essential for Delivery of Activated CD8 + T Cells to Virus-Infected Organs for Protective Immunity Rebar N. Mohammed, H. Angharad Watson, Miriam Vigar,
Cell Reports Supplemental Information L-selectin Is Essential for Delivery of Activated CD8 + T Cells to Virus-Infected Organs for Protective Immunity Rebar N. Mohammed, H. Angharad Watson, Miriam Vigar,
Adaptive Immunity: Specific Defenses of the Host
 17 Adaptive Immunity: Specific Defenses of the Host SLOs Differentiate between innate and adaptive immunity, and humoral and cellular immunity. Define antigen, epitope, and hapten. Explain the function
17 Adaptive Immunity: Specific Defenses of the Host SLOs Differentiate between innate and adaptive immunity, and humoral and cellular immunity. Define antigen, epitope, and hapten. Explain the function
Supplementary Figures
 Supplementary Figures Supplementary Figure 1. NKT ligand-loaded tumour antigen-presenting B cell- and monocyte-based vaccine induces NKT, NK and CD8 T cell responses. (A) The cytokine profiles of liver
Supplementary Figures Supplementary Figure 1. NKT ligand-loaded tumour antigen-presenting B cell- and monocyte-based vaccine induces NKT, NK and CD8 T cell responses. (A) The cytokine profiles of liver
Rapid antigen-specific T cell enrichment (Rapid ARTE)
 Direct ex vivo characterization of human antigen-specific CD154+CD4+ T cell Rapid antigen-specific T cell enrichment (Rapid ARTE) Introduction Workflow Antigen (ag)-specific T cells play a central role
Direct ex vivo characterization of human antigen-specific CD154+CD4+ T cell Rapid antigen-specific T cell enrichment (Rapid ARTE) Introduction Workflow Antigen (ag)-specific T cells play a central role
Supporting Online Material for
 www.sciencemag.org/cgi/content/full/1175194/dc1 Supporting Online Material for A Vital Role for Interleukin-21 in the Control of a Chronic Viral Infection John S. Yi, Ming Du, Allan J. Zajac* *To whom
www.sciencemag.org/cgi/content/full/1175194/dc1 Supporting Online Material for A Vital Role for Interleukin-21 in the Control of a Chronic Viral Infection John S. Yi, Ming Du, Allan J. Zajac* *To whom
Effector T Cells and
 1 Effector T Cells and Cytokines Andrew Lichtman, MD PhD Brigham and Women's Hospital Harvard Medical School 2 Lecture outline Cytokines Subsets of CD4+ T cells: definitions, functions, development New
1 Effector T Cells and Cytokines Andrew Lichtman, MD PhD Brigham and Women's Hospital Harvard Medical School 2 Lecture outline Cytokines Subsets of CD4+ T cells: definitions, functions, development New
Supplemental Methods In vitro T cell assays Inhibition of perforin- and FasL-mediated cytotoxicity Flow Cytometry
 Supplemental Methods In vitro T cell assays Cell lines Jurkat (ATCC #TIB-152), CCRF-CEM (ATCC #CCL-119), MOLT-4 (ATCC #CRL- 1582), Hut 78 (ATCC #TIB-161), SupT1 (ATCC #CRL-1942), Raji (ATCC #CCL-86) and
Supplemental Methods In vitro T cell assays Cell lines Jurkat (ATCC #TIB-152), CCRF-CEM (ATCC #CCL-119), MOLT-4 (ATCC #CRL- 1582), Hut 78 (ATCC #TIB-161), SupT1 (ATCC #CRL-1942), Raji (ATCC #CCL-86) and
Supplementary Figure 1. Normal T lymphocyte populations in Dapk -/- mice. (a) Normal thymic development in Dapk -/- mice. Thymocytes from WT and Dapk
 Supplementary Figure 1. Normal T lymphocyte populations in Dapk -/- mice. (a) Normal thymic development in Dapk -/- mice. Thymocytes from WT and Dapk -/- mice were stained for expression of CD4 and CD8.
Supplementary Figure 1. Normal T lymphocyte populations in Dapk -/- mice. (a) Normal thymic development in Dapk -/- mice. Thymocytes from WT and Dapk -/- mice were stained for expression of CD4 and CD8.
Manuscript: OX40 signaling is involved in the autoactivation of CD4 + CD28 - T cells and contributes to pathogenesis of autoimmune arthritis
 Manuscript: OX40 signaling is involved in the autoactivation of CD4 + CD28 - T cells and contributes to pathogenesis of autoimmune arthritis Vincent Laufer Rheumatology JC October 17 Disclosures NIH 2
Manuscript: OX40 signaling is involved in the autoactivation of CD4 + CD28 - T cells and contributes to pathogenesis of autoimmune arthritis Vincent Laufer Rheumatology JC October 17 Disclosures NIH 2
7-AAD/CFSE Cell-Mediated Cytotoxicity Assay Kit
 7-AAD/CFSE Cell-Mediated Cytotoxicity Assay Kit Catalog Number KA1293 96 assays Version: 02 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Background... 3 Principle of
7-AAD/CFSE Cell-Mediated Cytotoxicity Assay Kit Catalog Number KA1293 96 assays Version: 02 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Background... 3 Principle of
