Received 1 November 2006/Returned for modification 4 January 2007/Accepted 8 February 2007
|
|
|
- Ada Robbins
- 6 years ago
- Views:
Transcription
1 CLINICAL AND VACCINE IMMUNOLOGY, May 2007, p Vol. 14, No /07/$ doi: /cvi Copyright 2007, American Society for Microbiology. All Rights Reserved. Comparative Evaluation of Enzyme-Linked Immunosorbent Assays Based on Crude and Recombinant Leishmanial Antigens for Serodiagnosis of Symptomatic and Asymptomatic Leishmania infantum Visceral Infections in Dogs Renato Porrozzi, 1 Marcos V. Santos da Costa, 1 Antonio Teva, 1 Aloísio Falqueto, 2 Adelson L. Ferreira, 2 Claudiney D. dos Santos, 2 Ana Paula Fernandes, 3 Ricardo T. Gazzinelli, 3,4 Antonio Campos-Neto, 5 and Gabriel Grimaldi, Jr. 1 * Instituto Oswaldo Cruz (Fiocruz), Av. Brasil 4365, Rio de Janeiro, RJ, Brazil 1 ; Federal University of Espírito Santo, Vitória, ES, Brazil 2 ; Federal University of Minas Gerais, Belo Horizonte, MG, Brazil 3 ; Centro de Pesquisas René Rachou (Fiocruz), Belo Horizonte, MG, Brazil 4 ; and The Forsyth Institute, Boston, Massachusetts 5 Received 1 November 2006/Returned for modification 4 January 2007/Accepted 8 February 2007 The diagnosis of visceral leishmaniasis remains difficult in rural areas where the disease is endemic, and serologic methods still need assessment, as they are not very sensitive for the detection of asymptomatic infectious dogs. Here we present data on the development of enzyme-linked immunosorbent assay (ELISA)- based methods for the detection of antibodies against recombinant leishmanial antigens (namely, the recombinant K26 [rk26] and rk39 antigens from Leishmania infantum and the ra2 protein from Leishmania donovani) in comparison to ELISAs employing crude soluble antigen (CSA). The assays utilized sera from known negative controls (n 25) and clinically asymptomatic (n 50) and symptomatic (n 50) dogs with confirmed L. infantum infections. Additional studies were also done using sera from animals harboring other infections (n 14) for the evaluation of cross-reactivity. Our study indicated that rk26 and rk39 used in ELISAs provided very high sensitivities for the detection of symptomatic dogs (94% and 100%, respectively), followed by CSA (88%) and ra2 (70%). Conversely, ra2 was more sensitive for asymptomatic dogs (88%) than rk39 and rk26 (both 66%) and CSA (30%). Some cross-reactivity in sera from dogs with other infections (Leishmania braziliensis and Leptospira interrogans) was identified, but the ra2 protein provided the greatest specificity (98%). Data further indicate that all three recombinant proteins must be used in parallel to detect essentially all infected dogs. Efforts should be made to develop a cheap and reliable serologic test based on epitope selection from these diagnostic markers for the sensitive detection of L. infantum-infected dogs. Zoonotic visceral leishmaniasis (VL) caused by Leishmania infantum (Leishmania chagasi) is an important emerging parasitic disease found in countries around the Mediterranean basin, in the Middle East, and in Latin America ( /tdr/diseases). In the Neotropics, the parasite is transmitted by the sand fly Lutzomyia longipalpis and wild canids (Cerdocyon thous and Lycalopex vetulus) serve as the major sylvatic reservoirs, and domestic dogs are the principal reservoir hosts in sites where the disease is endemic (18). Such animals are a potential source of infection for sand fly vectors (11). Hence, the prevalence and the incidence of canine visceral leishmaniasis (CVL) are important epidemiological parameters for controlling transmission, the estimation of which depends on the reliable identification of infected dogs (15, 16, 24). Control programs focus on the mass elimination of seropositive dogs (8). Mathematical modeling suggests that culling programs fail because of the high incidence of canine infection and infectiousness, the insensitivity of the diagnostic tests to detect * Corresponding author. Mailing address: Department of Immunology, Oswaldo Cruz Institute, FIOCRUZ, Pav. Leonidas Deane, 5th Floor, Room 509, Av. Brasil 4365, CEP Rio de Janeiro, RJ, Brazil. Phone: Fax: Grimaldi@ioc.fiocruz.br. Published ahead of print on 21 February infectious dogs, and time delays between diagnosis and culling used in practice by public health services (11). Due to the limitations of direct methods to detect parasites in dogs (either by microscope examination, culture, or the inoculation of hamsters with biopsy specimens) (18), the presence of antiparasite antibodies is routinely used as a marker of infection, with the production of antibody as the definition of infection or challenge (16, 24). A wide variety of possible serologic techniques and antigens have been evaluated, but serology may be less specific than the assessment of parasites in biopsy specimens due to cross-reactions with other infectious agents, and the choice of a suitable cutoff may not be obvious (16). The currently available serologic tests include the indirect fluorescence antibody test (IFAT) (20), enzyme-linked immunosorbent assays (ELISAs) (4), dot-elisa (32), the direct agglutination test (19), Western blotting (2), and the lateral immunochromatographic test (31). Overall, the reported performances of these tests indicate that in general they are suitable tools for the serodiagnosis of VL in both symptomatic and asymptomatic dogs. In addition to the serologic tests, PCRbased assays have also been used for detecting L. infantum DNA in diagnostic samples from humans and dogs (23, 25). However, results from both cross-sectional and longitudinal analyses of the PCR and serologic data suggest that PCR is most useful for detecting active infection while serology can be 544
2 VOL. 14, 2007 SERODIAGNOSIS OF CANINE VISCERAL LEISHMANIASIS 545 a more sensitive technique for the detection of all infected dogs (23). The serodiagnosis of CVL remains problematic because the current diagnostic tests lack sufficient sensitivity or specificity, require technological expertise and specialized laboratory apparatuses, and can be labor-intensive and time-consuming. However, rapid tests like the immunochromatographic-dipstick and gel tests using the recombinant K39 (rk39) and rk26 proteins of L. infantum (5, 9) as antigens seem to be most suited for point-of-care diagnosis of symptomatic cases of CVL but lack sensitivity for asymptomatic dogs (21, 22, 25, 31). Hence, efforts should be made to develop a more sensitive and specific recombinant protein-based immunoassay capable of detecting asymptomatic carriers in mass screening surveys. Here we report a study in which the diagnostic potentials of parasite-specific recombinant antigens rk39, rk26, and ra2 in comparison with that of crude soluble antigen (CSA) in ELISAs were evaluated. The findings indicate that these markers complement one another, thus increasing the overall sensitivity of the antibody detection test for symptomatic and asymptomatic dogs with confirmed L. infantum visceral infections. MATERIALS AND METHODS Dogs and infection status. Four groups of serum samples from dogs were established. Group 1 contained negative control sera from 25 healthy pets of various ages and breeds that had attended a veterinary clinic in Rio de Janeiro, Brazil. Group 2 contained cross-reaction control sera from 14 dogs with either Leishmania (Viannia) braziliensis dermal leishmaniasis (n 9; parasites isolated from the lesions were characterized by multilocus enzyme electrophoresis), leptospiroses (n 2; dogs were seropositive for Leptospira interrogans by ELISA), or toxoplasmosis (n 3; dogs were seropositive by ELISA) that had attended a veterinary clinic at the municipality of Vitoria in Espírito Santo State (a VL-free area of Brazil). Group 3 contained sera (previously classified as having IFAT antileishmanial antibody titers of 1:80) from 50 asymptomatic dogs naturally infected with L. infantum. Group 4 contained sera (with IFAT-determined titers of 1:160) from 50 L. infantum-infected dogs with active disease (clinically classified as either oligosymptomatic [n 9] or polysymptomatic [n 41]). Infections were proven by the demonstration of the presence of the parasite in Giemsa-stained smears and/or in vitro cultures of the dogs bone marrow aspirates. The necropsy findings also showed parasite-containing macrophages in the liver, spleen, and lymph nodes. All infected dogs enrolled in the study were selected based on their serologic results from IFAT (which was performed at the Federal University of Espírito Santo) during a cross-sectional serodiagnosis survey of L. infantum CVL carried out in a rural area of endemicity (northwest Espírito Santo State) in southeast Brazil. Sampling and parasitology. Permission was obtained from all householders to use their dogs. Positive control sera were samples taken from study dogs at any time when Leishmania parasites were found in the bone marrow biopsy material by culture or microscopy. In addition, postmortem culturing or histological examinations of lymphoid tissues (such as lymph node, spleen, and liver tissues) allowed for the assessment of subclinical infection. Prior to each sampling, dogs were anesthetized with 20 mg of ketamine hydrochloride (Vetalar)/kg of body weight injected intramuscularly. Then 10 ml of venous blood was taken by venipuncture. Bone marrow was aspirated from the iliac crest with a 16-by- 25-mm needle into a 20-ml syringe containing 0.5% EDTA. The sample was used to make one to four thin smears for culture in vitro and/or inoculated into hamsters. The examination of cultures and smears of bone marrow specimens was done by standard techniques (24). Paraffin sections from necropsy tissue samples (fixed in 10% neutral buffered formalin) were stained with hematoxylin and eosin. All infected dogs were euthanized with sodium pentobarbital (Euthanol), and the necropsies were performed for the assessment of parasites in the skin, bone marrow, liver, and spleen. Leishmanial isolates from studied dogs were typed as L. infantum by multilocus enzyme electrophoresis in our laboratory as described previously (12). This research has complied with all relevant Brazilian federal guidelines (Projeto de lei 3.964/97; Clinical examination. All infected dogs underwent gross physical examination by veterinary practitioners in the field. Animals were scored clinically for six typical signs of CVL (alopecia, dermatitis, chancres, conjunctivitis, lymphadenopathy, and onychogryposis) on a semiquantitative scale as described by Quinnell and coworkers (23). Dogs with total scores of 0 to 2 were classified as asymptomatic, those with scores of 3 to 6 were classified as oligosymptomatic, and those with scores of 7 to 18 were classified as polysymptomatic. Antigens. Promastigotes of L. infantum (strain MHOM/BR/2000/1669) provided the source of whole-parasite extracts. A preparation of CSA derived from promastigotes broken by ultrasonic treatment was made as described elsewhere (27). The two recombinant proteins of L. infantum, namely, rk39 (a 39-aminoacid-repetitive immunodominant B-cell epitope kinesin-related antigen) (9) and rk26 (a gene fragment product containing the repetitive sequence of K26) (5), were kindly provided by the Infectious Disease Research Institute, Seattle, WA. The L. donovani ra2 protein was prepared as described elsewhere (10). ELISA. Sera from the dogs were analyzed by standard micro-elisa (21) to detect antigen-specific antibodies (by using a whole-molecule peroxidase-conjugated rabbit anti-dog immunoglobulin G [IgG]; Sigma, St. Louis, MO). The reaction was revealed with a biotin-avidin peroxidase system. The substrate consisted of 0.04% o-phenylenediamine dihydrochloride and 0.012% hydrogen peroxidase in phosphate-citrate buffer, ph 5.0. The absorbance at 492 nm was measured in an E max microplate reader (Molecular Devices, Ramsey, MN). A group of sera with previously known titers as control values, as well as negative control sera from healthy dogs, was included in each test. The lower limit of positivity (cutoff) was determined by using the mean plus 3 standard deviations of the A 492 values for 25 normal controls (sera of group 1). Data analysis. The sensitivities for the samples from the L. infantum-infected dogs in groups 3 (asymptomatic; n 50) and 4 (symptomatic; n 50) were calculated separately by using the following equation: [number of true positives/ (number of true positives number of false negatives)] 100. Test specificities were assessed with sera from group 1 (normal controls from a region of nonendemicity; n, 25 true negatives ) and group 2 (dogs infected with L. braziliensis or other pathogens; n 14). The percent specificity was calculated by using the following equation: [number of true negatives/(number of true negatives number of cross-reactive sera, considered to be false positives)] 100. Statistical analyses of all quantitative data were performed regardless of how significant the differences looked in the table or figure. Data were appropriately analyzed as parametric (normally distributed) and nonparametric data (by using Student s t test and Fisher s exact test, respectively), and a P value of 0.05 was considered statistically significant. RESULTS All ELISA procedures were optimized with regard to antigen concentrations and the serum dilution (data not shown). Optimal antigen concentrations were 2 g/ml for CSA and 0.5 g/ml for each recombinant protein. A clear separation between the parasite-positive dogs and healthy (uninfected) controls was possible by using a single serum dilution of 1:600. Using this serum dilution, we then compared the immunoreactivities of sera from symptomatic and asymptomatic L. infantum-infected dogs, healthy controls, and dogs harboring other infections for the evaluation of cross-reactivities. Figure 1 shows the results and indicates that positive ELISA signals (means standard deviations of the A 492 values) given by IgG specific for rk39, rk26, and ra2 antigens were very variable in asymptomatic dogs ( , , and , respectively) and, as expected, significantly higher (P 0.019) in symptomatic dogs ( , , and , respectively). In addition, the optical density readings produced by sera from symptomatic dogs with all antigens but ra2 were consistently higher than those produced by sera from oligosymptomatic animals. Moreover, the overall reactivity of the sera from symptomatic dogs with ra2 was significantly lower (P 0.01) than that with either rk39 or rk26.
3 546 PORROZZI ET AL. CLIN. VACCINE IMMUNOL. FIG. 1. Comparison of Leishmania-specific IgG antibodies in different groups of dogs. Antigen-specific antibody was assessed by ELISA using either CSA derived from promastigotes (A) or the rk26 (B), rk39 (C), and ra2 (D) proteins. The recombinant antigens were evaluated for diagnostic potential by using a panel of sera from dogs with proven L. infantum infections grouped by their clinical statuses. Normal control (uninfected) diagnostic samples and cross-reaction control sera were included to achieve adequate precision for estimates of the specificities of the ELISAs. Results are expressed as the optical densities at 492 nm. Dashed lines, cutoff optical densities (mean 3 standard deviations for normal controls) for each antigen. Downloaded from Table 1 summarizes the results of the different ELISAs used in this comparative study. Sensitivities. The sensitivity of the widely used CSA-based ELISA was 88% for the detection of dogs with symptomatic disease (previously having IFAT antileishmanial antibody titers of 1:160), but this assay missed 35 of 50 of the asymptomatic dogs with CVL (having IFAT-determined titers of TABLE 1. Sensitivity of ELISAs using crude and recombinant leishmanial antigens for the diagnosis of asymptomatic and symptomatic canine visceral leishmaniasis a Antigen No. of positive sera/no. of tested sera (% sensitivity) b from: Asymptomatic dogs Symptomatic dogs Asymptomatic/ symptomatic index CSA 15/50 (30) 44/50 (88) 0.34 K26 33/50 (66) 47/50 (94) 0.70 K39 33/50 (66) 50/50 (100) 0.66 A2 42/48 (88) 35/50 (70) 1.2 Total c 50/50 (100) 50/50 (100) a Infections were proven by parasitological examination (using microscopy, in vitro culture, and/or inoculation of hamsters with biopsy material). At sampling times, dogs were examined for six typical clinical signs of VL and then classified on a semiquantitative scale as described in Materials and Methods. b The percent sensitivity was calculated as indicated in Materials and Methods. c Dog sera that tested positive with at least one of the recombinant antigens assayed by ELISA. 1:80), resulting in an extremely decreased sensitivity (30%; P ). ELISAs with rk39 and rk26 antigens gave the best results in terms of sensitivity (100 and 94%, respectively) for symptomatic dogs but detected considerably fewer asymptomatic cases (both 66%; P ). Conversely, the ra2 ELISA was 88% sensitive for asymptomatic dogs and performed less well for symptomatic cases (70% sensitivity; P 0.048), yielding the highest number of false-negative results in this group. Of note, each of the recombinant antigens used in the ELISAs detected some positive sera that others missed (data not shown). The data indicated that all three recombinant antigens must be used in parallel to obtain an assay with 100% sensitivity (Table 1). Specificities. To define potential cross-reactions with antigens for the diagnosis of CVL, we tested L. infantum-derived CSA and the recombinant antigens against sera from dogs infected with other pathogens (Table 2). As anticipated, most of the L. braziliensis-infected dogs (five of nine) were positive as determined by the CSA ELISA (77.8% cross-reactivity). Surprisingly, however, anti-rk39 and anti-rk36 antibodies were detected in three and five dogs, respectively. In addition, one dog with leptospiroses was also positive with rk39 and rk36. In contrast, specific anti-ra2 antibodies were found in only one dog with active L. braziliensis dermal infection, thus showing the excellent overall specificity of the ra2 ELISA of 98%, as assessed with healthy dogs and dogs with other infec- on April 21, 2018 by guest
4 VOL. 14, 2007 SERODIAGNOSIS OF CANINE VISCERAL LEISHMANIASIS 547 TABLE 2. Specificity of ELISAs using crude and recombinant leishmanial antigens for the diagnosis of canine visceral leishmaniasis Antigen No. of positive sera/no. of tested sera from dogs with: L. braziliensis dermal leishmaniasis a Leptospira interrogans leptospiroses a Toxoplasmosis a tions. Finally, sera from three dogs with toxoplasmosis and those from 25 healthy dogs from an area of nonendemicity were all negative with all tested antigens. DISCUSSION No infection (controls) b % Specificity c CSA 5/9 0/2 0/3 0/25 87 K26 3/9 1/2 0/3 0/25 90 K39 5/9 1/2 0/3 0/25 85 A2 1/9 0/2 0/3 0/25 96 a Infections were proven by parasitological or serologic examination. b Serum samples were obtained from healthy blood donor pets born in a VL-free area of Brazil. c The percent specificity was calculated from an equation as indicated in Materials and Methods. In general, a seroprevalence evaluation of VL may underestimate the true prevalence of infection (16), because (i) the sensitivity of antibody detection is generally lower for early or asymptomatic infections (21, 23, 25), (ii) there is a significant prepatent period before seroconversion (1), (iii) a fraction of infected dogs may never convert (16), and (iv) dogs may revert to seronegativity but remain parasite positive (24). Previous studies have reported various levels of L. infantum-specific antibodies of the IgG1 or IgG2 isotype in sera of infected dogs, sometimes with conflicting results (14, 21, 29). The genetic variability of canine immune responses may account for this controversy, but problems due to the specificities and sensitivities of serologic tests also occur (16). A reference test with suboptimal sensitivity can substantially affect the validity estimates for new tests under scrutiny. Thus, the published sensitivity and specificity estimates for serologic tests may be biased to some extent, depending on the controls and reference test used (7). On the other hand, parasite detection tests, despite being very specific, cannot be the gold standard because their sensitivities are defined by parasite burdens (16, 24, 33). In this investigation, we compared the effectiveness of using different recombinant antigens in an ELISA format to evaluate a panel of sera from dogs considered to be truly parasite positive, along with negative and cross-reaction control sera. Although the results show great variation in levels of antigenspecific IgG antibodies, titers of antibodies to the rk26 and rk39 antigens were higher in symptomatic than in asymptomatic animals (P 0.01). The results corroborated previous data showing that serologic test performance depends on many factors such as infection status (23, 25) and the type of diagnostic antigen or conjugate used (21, 27, 31). The rk39 and rk26 antigens of L. infantum have proven to be of reliable sensitivities when used in ELISAs for the diagnosis of human VL (3 5). The rk39 ELISA is highly sensitive (reported sensitivities, 93 to 100%) for symptomatic dogs (3, 26 28, 34) but fails to detect or detects fewer asymptomatic cases (sensitivities, 52.9 to 64.7%) of proven infections (21, 26). In contrast, the rk26 ELISA has very high sensitivities for both symptomatic (n 21; 100%) and asymptomatic (n 48; 98%) dogs (31). Moreover, it has been suggested that the presence of specific anti-rk39 (3, 26) and anti-a2 (10, 17) antibodies can in fact be considered as a diagnostic marker for active disease in humans and dogs. In our assays, the rk39 and rk26 antigens were the most sensitive (100% and 94%, respectively), followed by CSA (88%) and ra2 (70%), for symptomatic cases of L. infantum CVL. Conversely, the ra2 antigen detected more asymptomatic cases (88%) than rk39 and rk26 (both 66%) and CSA (30%). When we calculated an index based on the ratio of asymptomatic to symptomatic cases that were identified as positive by the respective antigen used in the ELISAs, A2 presented the highest score (1.20), followed by K39 (0.66), K26 (0.70), and CSA (0.34), suggesting an association between the response to A2 and protective immunity. However, this possibility needs further confirmation with larger populations of dogs from other areas of endemicity. In a previous ra2 ELISA assay, responsiveness was found in 93.3% of 15 symptomatic dogs (10). Taken together, our data indicate that serologic tests employing rk39 will identify the symptomatic dogs (the most infectious animals) while rk26 and ra2 antigens seem to be most effective for the serodiagnosis of both asymptomatic and symptomatic CVL. Previous studies reported high specificities (ranging from 94 to 100%) of ELISAs using the rk39, rk26, and A2 antigens (3, 10, 17, 27, 28, 31). Our study indicates lower specificities (85 to 98%) as assessed with sera from healthy blood donor dogs and cross-reactive sera obtained from dogs infected with either L. braziliensis or Leptospira interrogans (Table 2). Interestingly, previous reports have suggested that the rk39 and rk26 antigens when used for the diagnosis of human VL do not crossreact with L. braziliensis (3, 9, 13). In addition, in previous studies with human VL (10), we found that the antigen ra2 is recognized by 60% of sera from patients with mucosal leishmaniasis, which is caused by L. braziliensis. Although to a lesser extent, the present results indicate that such cross-reaction also occurs in CVL, as specific anti-ra2 antibodies were detected in one out of nine L. braziliensis-infected dogs. The reasons for the unexpected cross-reactivity of the antigens rk26 and rk39 in contrast to previous observations are unknown at the present time. The small number of animals as well as the different geographic regions of endemicity from which the dogs were enrolled in these various studies may account for this controversy. It is worth mentioning that taxonomic studies have shown genetic polymorphism in natural populations of L. braziliensis (12). Whether strain variants of this parasite modulate their antigen expression patterns differently remains to be determined. A major limitation to the study of the epidemiology of and hence control programs for CVL is the inability to identify and count asymptomatic carriers (15, 24) because classic diagnostic tests are insufficiently sensitive (16, 25). Thus, a rapid, sensitive, and specific tool for the detection of L. infantum infection in dogs would be highly desirable because it would allow for effective control interventions in areas where zoonotic VL is endemic. Recent studies have evaluated multiple-epitope chimerical antigens as diagnostic markers for the serodiagnosis of CVL (6, 30). ELISAs performed with these interesting chimerical antigens had sensitivities ranging from 79 to 96% for CVL and specificities ranging from 96 to 100%, depending on the
5 548 PORROZZI ET AL. CLIN. VACCINE IMMUNOL. negative control panel used. The main finding in our study was that the three parasite-specific recombinant antigens showed independent and complementary immunoreactivies and that assays reached an overall sensitivity of 100% when these antigens were used in parallel. These data reinforce the idea (6, 31) that the combination of these antigens in a multiple-epitope format could further improve the performance of a single-well test for the routine serodiagnosis of CVL. ACKNOWLEDGMENTS The recombinant antigens K26 and K39 were kindly supplied by the Infectious Disease Research Institute (Seattle, WA). We thank Daniel Kiefer, Fundação Nacional da Saúde, for assistance in the field studies. Financial support was received from Fiocruz, PRONEX 3 (CNPq /1998-3), and the Millennium Institute for Vaccine Development and Technology (CNPq /2005-1), National Council for Scientific and Technological Development of the Ministry of Science and Technology (Brazil). We have no conflicts of interest concerning the work reported in this paper. REFERENCES 1. Abranches, P., G. Santos-Gomes, N. Rachamim, L. Campino, L. F. Schnur, and C. L. Jaffe An experimental model for canine visceral leishmaniasis. Parasite Immunol. 13: Aisa, M. J., S. Castillejo, M. Gallego, R. Fisa, M. C. Riera, M. De Colmenares, S. Torras, X. Roura, J. Sentis, and M. Portus Diagnostic potential of Western blot analysis of sera from dogs with leishmaniasis in endemic areas and significance of the pattern. Am. J. Trop. Med. Hyg. 58: Badaró, R., D. Benson, M. C. Eulálio, M. Freire, S. Cunha, E. M. Neto, D. Pedral-Sampaio, C. Madureira, J. M. Burns, R. L. Houghton, J. R. David, and S. G. Reed rk39: a cloned antigen of Leishmania chagasi that predicts active visceral leishmaniasis. J. Infect. Dis. 173: Badaró, R., M. C. Eulálio, D. Benson, M. Freire, J. C. Miranda, D. Pedral- Sampaio, J. M. Burns, J. R. David, Jr., W. J. Johnson, and S. G. Reed Sensitivity and specificity of a recombinant Leishmania chagasi antigen in the serodiagnosis of visceral leishmaniasis. Arch. Inst. Pasteur (Tunis) 70: Bhatia, A., N. S. Daifalla, S. Jen, R. Badaro, S. G. Reed, and Y. A. Skeike Cloning, characterization and serological evaluation of K9 and K26: two related hydrophilic antigens of Leishmania chagasi. Mol. Biochem. Parasitol. 102: Boarino, A., A. Scalone, L. Gradoni, E. Ferroglio, F. Vitale, R. Zanatta, M. G. Giuffrida, and S. Rasati Development of recombinant chimeric antigen expressing immunodominant B epitopes of Leishmania infantum for serodiagnosis of visceral leishmaniasis. Clin. Diagn. Lab. Immunol. 12: Boelaert, M., S. Rijal, S. Regmi, R. Singh, B. Karki, D. Jacquet, F. Chappuis, L. Campino, P. Desjeux, D. Le Ray, S. Koirala, and P. Van der Stuyft A comparative study of the effectiveness of diagnostic tests for visceral leishmaniasis. Am. J. Trop. Med. Hyg. 70: Braga, M. D., I. C. Coelho, M. M. Pompeu, T. G. Evans, I. T. MacAullife, M. J. Teixeira, and J. W. Lima Control of canine visceral leishmaniasis: comparison of results from a rapid elimination program of serumreactive dogs using an immunoenzyme assay and slower elimination of serum-reactive dogs using filter paper elution indirect immunofluorescence. Rev. Soc. Bras. Med. Trop. 31: Burns, J. M., Jr., W. G. Shreffler, D. R. Benson, H. W. Ghalib, R. Badaro, and S. G. Reed Molecular characterization of a kinesin-related antigen of Leishmania chagasi that detects specific antibody in African and American visceral leishmaniasis. Proc. Natl. Acad. Sci. USA 90: Carvalho, F. A. A., H. Charest, C. A. P. Tavares, G. Matlashewski, E. P. Valente, A. Rabello, R. T. Gazinelli, and A. P. Fernández Diagnosis of American visceral leishmaniasis in humans and dogs using the recombinant Leishmania donovani A2 antigen. Diagn. Microbiol. Infect. Dis. 43: Courtenay, O., R. J. Quinnell, L. M. Garcez, J. J. Shaw, and C. Dye Infectiousness in a cohort of Brazilian dogs: why culling fails to control visceral leishmaniasis in areas of high transmission. J. Infect. Dis. 186: Cupolillo, E., L. R. Brahim, C. B. Toaldo, M. P. Oliveira-Neto, M. E. Felinto de Brito, A. Falqueto, M. F. Naiff, and G. Grimaldi, Jr Genetic polymorphism and molecular epidemiology of Leishmania (Viannia) braziliensis from different hosts and geographic areas in Brazil. J. Clin. Microbiol. 41: Delgado, O., M. D. Feliciangeli, V. Coraspe, S. Silva, A. Perez, and J. Arias Value of a dipstick based on recombinant rk39 antigen for differential diagnosis of American visceral leishmaniasis from other sympatric endemic diseases in Venezuela. Parasite 8: Desplazes, P., N. C. Smith, P. Arnold, H. Lutz, and J. Eckert Specific IgG1 and IgG2 antibody responses of dogs to Leishmania infantum and other parasites. Parasite Immunol. 17: Dye, C., R. Killick-Kendrick, M. M. Vitutia, R. Walton, M. Killick-Kendrick, A. E. Harith, M. W. Guy, M.-C. Cañavate, and G. Hasibeder Epidemiology of canine leishmaniasis: prevalence, incidence and basic reproduction number calculated from a cross-sectional serological survey on the island of Gozo, Malta. Parasitology 105: Dye, C., E. Vidor, and J. Deneure Serological diagnosis of leishmaniasis: on detecting infection as well as disease. Epidemiol. Infect. 103: Ghedin, E., W. W. Zhang, H. Charest, S. Sundar, R. T. Kenney, and G. Matlashewski Antibody response against Leishmania donovani amastigote-stage-specific protein in patients with visceral leishmaniasis. Clin. Diagn. Lab. Immunol. 4: Grimaldi, G., Jr., and R. B. Tesh Leishmaniasis of the New World: current concepts and implications for future research. Clin. Microbiol. Rev. 6: Harith, A., A. H. Kolk, P. A. Kager, J. Leeuwenburg, R. Muigai, S. Kiugu, and J. J. Laarman A simple and economical direct agglutination test for serodiagnosis and sero-epidemiological studies of visceral leishmaniasis. Trans. R. Soc. Trop. Med. Hyg. 80: Mancianti, F., and N. Meciani Specific serodiagnosis of canine leishmaniasis by indirect immunofluorescence, indirect hemagglutination, and counter-immunoelectrophoresis. Am. J. Vet. Res. 49: Mettler, M., F. Grimm, G. Capelli, and H. Camp Evaluation of enzyme-linked immunosorbent assays, an immunofluorescent-antibody test, and two rapid tests (immunochromatographic-dipstick and gel tests) for serological diagnosis of symptomatic and asymptomatic Leishmania infections in dogs. J. Clin. Microbiol. 43: Otranto, D., P. Paradies, M. Sasanelli, R. Spinelli, and O. Brandonisio Rapid immunochromatographic test for serodiagnosis of canine leishmaniasis. J. Clin. Microbiol. 42: Quinnell, R. J., O. Courtenay, S. Davidson, L. Garcez, B. Lambson, P. Ramos, J. J. Shaw, M. A. Shaw, and C. Dye Detection of Leishmania infantum by PCR, serology and immune response in a cohort study of Brazilian dogs. Parasitology 122: Quinnell, R. J., O. Courtenay, L. Garcez, and C. Dye The epidemiology of canine leishmaniasis: transmission rates estimated from a cohort study in Amazonian Brazil. Parasitology 115: Reithinger, R., R. J. Quinnell, B. Alexander, and C. R. Davies Rapid detection of Leishmania infantum infection in dogs: comparative study using an immunochromatographic dipstick test, enzyme-linked immunosorbent assay, and PCR. J. Clin. Microbiol. 40: Rhalem, A., H. Sahibi, N. Gueeous-Idrissi, S. Lasri, A. Natami, M. Riyad, and B. Berrag Immune response against Leishmania antigens in dogs naturally and experimentally infected with Leishmania infantum. Vet. Parasitol. 81: Rosario, E. Y., O. Genaro, J. C. França-Silva, and R. T. Da Costa Evaluation of enzyme-linked immunosorbent assay using crude Leishmania and recombinant antigens as a diagnostic marker for canine visceral leishmaniasis. Mem. Inst. Oswaldo Cruz 100: Scalone, A., R. De Luna, G. Oliva, L. Balde, G. Satta, G. Vesco, W. Mignone, C. Turilli, R. R. Mondesire, D. Simpson, A. R. Donoghue, G. R. Frank, and L. Gradoni Evaluation of the Leishmania recombinant K39 antigen as a diagnostic marker for canine leishmaniasis and validation of a standardized enzyme-linked immunosorbent assay. Vet. Parasitol. 104: Solano-Gallego, L., C. Riera, X. Roura, L. Iniesta, M. Gallego, J. E. Valladares, R. Fisa, S. Castiççejo, J. Alberola, L. Ferrer, M. Arboix, and M. Portús Leishmania infantum-specific IgG, IgG1 and IgG2 antibody responses in healthy and ill dogs from endemic areas. Evolution in the course of infection and after treatment. Vet. Parasitol. 96: Soto, M., J. M. Requena, L. Quijada, and C. Alonso Multicomponent chimeric antigen for serodiagnosis of canine visceral leishmaniasis. J. Clin. Microbiol. 36: Teodoro da Costa, R., J. C. França, W. Mayrink, E. Nascimento, O. Genaro, and A. Campos-Neto Standardization of a rapid immunochromatographic test with the recombinant antigens K39 and K26 for the diagnosis of canine visceral leishmaniasis. Trans. R. Soc. Trop. Med. Hyg. 97: Vercammen, F., D. Berkvens, J. Brandt, and W. Vansteenkiste A sensitive and specific 30-min Dot-ELISA for the detection of anti-leishmania antibodies in the dog. Vet. Parasitol. 79: Zijlstra, E. E., M. S. Ali, A. M. El Hassan, I. A. El Toum, M. Satti, H. W. Ghalib, and P. A. Kager Kala-azar: a comparative study of parasitological methods and the direct agglutination test in diagnosis. Trans. R. Soc. Trop. Med. Hyg. 86: Zijlstra, E. E., N. S. Daifalla, P. A. Pager, E. A. Khalil, A. M. El-Hassan, S. G. Reed, and H. W. Ghalib rk39 enzyme-linked immunosorbent assay for diagnosis of Leishmania donovani infection. Clin. Diagn. Lab. Immunol. 5:
Evaluation of rk28 antigen for serodiagnosis of visceral Leishmaniasis in India
 ORIGINAL ARTICLE TROPICAL AND PARASITIC DISEASES Evaluation of rk8 antigen for serodiagnosis of visceral Leishmaniasis in India M. Vaish, A. Bhatia, S. G. Reed, J. Chakravarty and S. Sundar ) Department
ORIGINAL ARTICLE TROPICAL AND PARASITIC DISEASES Evaluation of rk8 antigen for serodiagnosis of visceral Leishmaniasis in India M. Vaish, A. Bhatia, S. G. Reed, J. Chakravarty and S. Sundar ) Department
This is a repository copy of Detection of Leishmania infantum by PCR, serology and cellular immune response in a cohort study of Brazilian dogs.
 This is a repository copy of Detection of Leishmania infantum by PCR, serology and cellular immune response in a cohort study of Brazilian dogs. White Rose Research Online URL for this paper: http://eprints.whiterose.ac.uk/1262/
This is a repository copy of Detection of Leishmania infantum by PCR, serology and cellular immune response in a cohort study of Brazilian dogs. White Rose Research Online URL for this paper: http://eprints.whiterose.ac.uk/1262/
New Insights into Diagnosing Leishmaniasis
 New Insights into Diagnosing Leishmaniasis Eric Zini Snow meeting, 13 March 2009 Climate Variability and Visceral Leishmaniasis in Europe WHO/TDR, Jan. 2008 Late Eighties Maroli et al., Trop Med Int Health
New Insights into Diagnosing Leishmaniasis Eric Zini Snow meeting, 13 March 2009 Climate Variability and Visceral Leishmaniasis in Europe WHO/TDR, Jan. 2008 Late Eighties Maroli et al., Trop Med Int Health
Dual-Path Platform (DPP ) technologies, has overcome. de Oliveira et al. Parasites & Vectors (2015) 8:45 DOI /s
 de Oliveira et al. Parasites & Vectors (2015) 8:45 DOI 10.1186/s13071-015-0651-6 SHORT REPORT Open Access Multi-antigen print immunoassay (MAPIA)-based evaluation of novel recombinant Leishmania infantum
de Oliveira et al. Parasites & Vectors (2015) 8:45 DOI 10.1186/s13071-015-0651-6 SHORT REPORT Open Access Multi-antigen print immunoassay (MAPIA)-based evaluation of novel recombinant Leishmania infantum
Follow-up of Leishmania infantum naturally infected dogs treated with allopurinol: immunofluorescence antibody test, ELISA and Western blot
 Acta Tropica 84 (2002) 175/181 www.parasitology-online.com Follow-up of Leishmania infantum naturally infected dogs treated with allopurinol: immunofluorescence antibody test, ELISA and Western blot Francis
Acta Tropica 84 (2002) 175/181 www.parasitology-online.com Follow-up of Leishmania infantum naturally infected dogs treated with allopurinol: immunofluorescence antibody test, ELISA and Western blot Francis
A Novel Noninvasive Method for Diagnosis of Visceral Leishmaniasis by. rk39 Test in Sputum Samples
 JCM Accepts, published online ahead of print on 0 June 00 J. Clin. Microbiol. doi:0./jcm.00-0 Copyright 00, American Society for Microbiology and/or the Listed Authors/Institutions. All Rights Reserved.
JCM Accepts, published online ahead of print on 0 June 00 J. Clin. Microbiol. doi:0./jcm.00-0 Copyright 00, American Society for Microbiology and/or the Listed Authors/Institutions. All Rights Reserved.
Eliza Yoshie do Rosário, Odair Genaro, João C França-Silva, Roberto T da Costa, Wilson Mayrink, Alexandre Barbosa Reis*, Mariângela Carneiro/ +
 Mem Inst Oswaldo Cruz, Rio de Janeiro, Vol. 100(2): 197-203, April 2005 197 Evaluation of enzyme-linked immunosorbent assay using crude Leishmania and recombinant antigens as a diagnostic marker for canine
Mem Inst Oswaldo Cruz, Rio de Janeiro, Vol. 100(2): 197-203, April 2005 197 Evaluation of enzyme-linked immunosorbent assay using crude Leishmania and recombinant antigens as a diagnostic marker for canine
Major Article INTRODUCTION
 Major Article Revista da Sociedade Brasileira de Medicina Tropical 48(2):188-193, Mar-Apr, 2015 http://dx.doi.org/10.1590/0037-8682-0285-2014 Comparison of recombinant A2-ELISA with rke16 dipstick and
Major Article Revista da Sociedade Brasileira de Medicina Tropical 48(2):188-193, Mar-Apr, 2015 http://dx.doi.org/10.1590/0037-8682-0285-2014 Comparison of recombinant A2-ELISA with rke16 dipstick and
Iranian J Parasitol: Vol.2, No.2, 2007, pp
 Original Article Preparation of a K39sub Recombinant Antigen for the Detection of Leishmania infantum Antibodies in Human: a Comparative Study with an Immunochromatographic Test and Direct Agglutination
Original Article Preparation of a K39sub Recombinant Antigen for the Detection of Leishmania infantum Antibodies in Human: a Comparative Study with an Immunochromatographic Test and Direct Agglutination
Application of an improved ELISA method for the serological diagnosis of canine
 JCM Accepts, published online ahead of print on 17 February 2010 J. Clin. Microbiol. doi:10.1128/jcm.02402-09 Copyright 2010, American Society for Microbiology and/or the Listed Authors/Institutions. All
JCM Accepts, published online ahead of print on 17 February 2010 J. Clin. Microbiol. doi:10.1128/jcm.02402-09 Copyright 2010, American Society for Microbiology and/or the Listed Authors/Institutions. All
Comparison of serological assays for the diagnosis of canine visceral leishmaniasis in animals presenting different clinical manifestations
 Veterinary Parasitology 146 (2007) 235 241 www.elsevier.com/locate/vetpar Comparison of serological assays for the diagnosis of canine visceral leishmaniasis in animals presenting different clinical manifestations
Veterinary Parasitology 146 (2007) 235 241 www.elsevier.com/locate/vetpar Comparison of serological assays for the diagnosis of canine visceral leishmaniasis in animals presenting different clinical manifestations
DIAGNOSTIC POTENTIAL OF WESTERN BLOT ANALYSIS OF SERA FROM DOGS WITH LEISHMANIASIS IN ENDEMIC AREAS AND SIGNIFICANCE OF THE PATTERN
 Am. J. Trop. Med. Hyg., 58(2), 1998, pp. 154 159 Copyright 1998 by The American Society of Tropical Medicine and Hygiene DIAGNOSTIC POTENTIAL OF WESTERN BLOT ANALYSIS OF SERA FROM DOGS WITH LEISHMANIASIS
Am. J. Trop. Med. Hyg., 58(2), 1998, pp. 154 159 Copyright 1998 by The American Society of Tropical Medicine and Hygiene DIAGNOSTIC POTENTIAL OF WESTERN BLOT ANALYSIS OF SERA FROM DOGS WITH LEISHMANIASIS
Evaluation of Change in Canine Diagnosis Protocol Adopted by the Visceral Leishmaniasis Control Program in Brazil and a New Proposal for Diagnosis
 Evaluation of Change in Canine Diagnosis Protocol Adopted by the Visceral Leishmaniasis Control Program in Brazil and a New Proposal for Diagnosis Wendel Coura-Vital 1,2,7, Henrique Gama Ker 1,3, Bruno
Evaluation of Change in Canine Diagnosis Protocol Adopted by the Visceral Leishmaniasis Control Program in Brazil and a New Proposal for Diagnosis Wendel Coura-Vital 1,2,7, Henrique Gama Ker 1,3, Bruno
Diagnostic Accuracy of Two rk39 Antigen-Based Dipsticks and the Formol Gel Test for Rapid Diagnosis of Visceral Leishmaniasis in Northeastern Uganda
 JOURNAL OF CLINICAL MICROBIOLOGY, Dec. 2005, p. 5973 5977 Vol. 43, No. 12 0095-1137/05/$08.00 0 doi:10.1128/jcm.43.12.5973 5977.2005 Copyright 2005, American Society for Microbiology. All Rights Reserved.
JOURNAL OF CLINICAL MICROBIOLOGY, Dec. 2005, p. 5973 5977 Vol. 43, No. 12 0095-1137/05/$08.00 0 doi:10.1128/jcm.43.12.5973 5977.2005 Copyright 2005, American Society for Microbiology. All Rights Reserved.
Use of PCR RFLP to identify Leishmania species in naturally-infected dogs
 Veterinary Parasitology 140 (2006) 231 238 www.elsevier.com/locate/vetpar Use of PCR RFLP to identify Leishmania species in naturally-infected dogs Hélida Monteiro de Andrade a, *, Alexandre Barbosa Reis
Veterinary Parasitology 140 (2006) 231 238 www.elsevier.com/locate/vetpar Use of PCR RFLP to identify Leishmania species in naturally-infected dogs Hélida Monteiro de Andrade a, *, Alexandre Barbosa Reis
Proceedings of the World Small Animal Veterinary Association Sydney, Australia 2007
 Proceedings of the World Small Animal Veterinary Association Sydney, Australia 2007 Hosted by: Australian Small Animal Veterinary Association (ASAVA) Australian Small Animal Veterinary Association (ASAVA)
Proceedings of the World Small Animal Veterinary Association Sydney, Australia 2007 Hosted by: Australian Small Animal Veterinary Association (ASAVA) Australian Small Animal Veterinary Association (ASAVA)
Development of a Dipstick Assay for Detection of Leishmania-Specific Canine Antibodies
 JOURNAL OF CLINICAL MICROBIOLOGY, Jan. 2004, p. 193 197 Vol. 42, No. 1 0095-1137/04/$08.00 0 DOI: 10.1128/JCM.42.1.193 197.2004 Copyright 2004, American Society for Microbiology. All Rights Reserved. Development
JOURNAL OF CLINICAL MICROBIOLOGY, Jan. 2004, p. 193 197 Vol. 42, No. 1 0095-1137/04/$08.00 0 DOI: 10.1128/JCM.42.1.193 197.2004 Copyright 2004, American Society for Microbiology. All Rights Reserved. Development
Diagnostic Techniques To Detect Cryptic Leishmaniasis in Dogs
 CLINICAL AND DIAGNOSTIC LABORATORY IMMUNOLOGY, Sept. 2002, p. 1137 1141 Vol. 9, No. 5 1071-412X/02/$04.00 0 DOI: 10.1128/CDLI.9.5.1137 1141.2002 Copyright 2002, American Society for Microbiology. All Rights
CLINICAL AND DIAGNOSTIC LABORATORY IMMUNOLOGY, Sept. 2002, p. 1137 1141 Vol. 9, No. 5 1071-412X/02/$04.00 0 DOI: 10.1128/CDLI.9.5.1137 1141.2002 Copyright 2002, American Society for Microbiology. All Rights
Belo Horizonte, Minas Gerais, Brazil b Laboratório de Parasitologia e Histopatologia, Núcleo de Pesquisas em Ciências Biológicas/NUPEB;
 Veterinary Immunology and Immunopathology 112 (2006) 102 116 www.elsevier.com/locate/vetimm Isotype patterns of immunoglobulins: Hallmarks for clinical status and tissue parasite density in brazilian dogs
Veterinary Immunology and Immunopathology 112 (2006) 102 116 www.elsevier.com/locate/vetimm Isotype patterns of immunoglobulins: Hallmarks for clinical status and tissue parasite density in brazilian dogs
Introduction Leishmaniasis is a group of zoonotic infections caused by protozoan parasites of the genus Leishmania; an estimated persons are
 Sero-epidemological Survey on Canine Visceral Leishmaniasis and the Distribution of Sandfly Vectors in Northwestern Turkey: Prevention Strategies for Childhood Visceral Leishmaniasis by Nihal Dogan, a
Sero-epidemological Survey on Canine Visceral Leishmaniasis and the Distribution of Sandfly Vectors in Northwestern Turkey: Prevention Strategies for Childhood Visceral Leishmaniasis by Nihal Dogan, a
PUC Minas and Minasfungi do Brasil Ltda, Belo Horizonte, Minas Gerais, Brazil.
 CVI Accepts, published online ahead of print on 3 April 2013 Clin. Vaccine Immunol. doi:10.1128/cvi.00023-13 Copyright 2013, American Society for Microbiology. All Rights Reserved. 1 2 3 4 5 6 7 8 9 10
CVI Accepts, published online ahead of print on 3 April 2013 Clin. Vaccine Immunol. doi:10.1128/cvi.00023-13 Copyright 2013, American Society for Microbiology. All Rights Reserved. 1 2 3 4 5 6 7 8 9 10
APPLICATIONS OF LATENT CLASS ANALYSIS IN DIAGNOSTIC VALIDATION, THE USER PERSPECTIVE
 APPLICATIONS OF LATENT CLASS ANALYSIS IN DIAGNOSTIC VALIDATION, THE USER PERSPECTIVE Author's name(s): Boelaert M 1 *, Verloo D. 2, Van der Stuyft P 1 Affiliation(s): 1 Prince Leopold Institute of Tropical
APPLICATIONS OF LATENT CLASS ANALYSIS IN DIAGNOSTIC VALIDATION, THE USER PERSPECTIVE Author's name(s): Boelaert M 1 *, Verloo D. 2, Van der Stuyft P 1 Affiliation(s): 1 Prince Leopold Institute of Tropical
Received 29 July 2005/Returned for modification 8 September 2005/Accepted 15 September 2005
 CLINICAL AND DIAGNOSTIC LABORATORY IMMUNOLOGY, Dec. 2005, p. 1410 1415 Vol. 12, No. 12 1071-412X/05/$08.00 0 doi:10.1128/cdli.12.12.1410 1415.2005 Copyright 2005, American Society for Microbiology. All
CLINICAL AND DIAGNOSTIC LABORATORY IMMUNOLOGY, Dec. 2005, p. 1410 1415 Vol. 12, No. 12 1071-412X/05/$08.00 0 doi:10.1128/cdli.12.12.1410 1415.2005 Copyright 2005, American Society for Microbiology. All
Validation of a β-me ELISA for Detection of Anti Leishmania donovani Antibodies in Eastern Sudan
 Validation of a β-me ELISA for Detection of Anti Leishmania donovani Antibodies in Eastern Sudan Elfadil Abass, Abdelhafeiz Mahamoud, Durria Mansour, Mehdi Mohebali 2, Abdallah el Harith 3* Biomedical
Validation of a β-me ELISA for Detection of Anti Leishmania donovani Antibodies in Eastern Sudan Elfadil Abass, Abdelhafeiz Mahamoud, Durria Mansour, Mehdi Mohebali 2, Abdallah el Harith 3* Biomedical
Laboratory diagnosis of Blood and tissue flagellates
 Laboratory diagnosis of Blood and tissue flagellates (Leishmania and trypanosma) Sarah Alharbi Clinical Laboratory department Collage of Applied Medical Sciences King Saud University Leishmania and trypanosma:
Laboratory diagnosis of Blood and tissue flagellates (Leishmania and trypanosma) Sarah Alharbi Clinical Laboratory department Collage of Applied Medical Sciences King Saud University Leishmania and trypanosma:
Leishmaniasis Direct Agglutination Test: Using Pictorials as Training Materials to Reduce Inter-Reader Variability and Improve Accuracy
 Leishmaniasis Direct Agglutination Test: Using Pictorials as Training Materials to Reduce Inter-Reader Variability and Improve Accuracy Emily R. Adams 1 *, Diane Jacquet 2, Gerard Schoone 1, Kamlesh Gidwani
Leishmaniasis Direct Agglutination Test: Using Pictorials as Training Materials to Reduce Inter-Reader Variability and Improve Accuracy Emily R. Adams 1 *, Diane Jacquet 2, Gerard Schoone 1, Kamlesh Gidwani
Serological tests fail to discriminate dogs with visceral leishmaniasis that transmit Leishmania infantum to the vector Lutzomyia longipalpis
 Rev Soc Bras Med Trop 50(4):483-488, July-August, 2017 doi: 10.1590/0037-8682-0014-2017 Major Article Serological tests fail to discriminate dogs with visceral leishmaniasis that transmit Leishmania infantum
Rev Soc Bras Med Trop 50(4):483-488, July-August, 2017 doi: 10.1590/0037-8682-0014-2017 Major Article Serological tests fail to discriminate dogs with visceral leishmaniasis that transmit Leishmania infantum
Recommendations for Coping with Leishmaniasis: A Review of Control Strategies. Centro de Convenções de Reboças Red Room 17: 00h
 Recommendations for Coping with Leishmaniasis: A Review of Control Strategies Centro de Convenções de Reboças 08.04.2014 Red Room 17: 00h Leishmaniasis - a Global Problem Visceral 2012 300 000 cases 20,000
Recommendations for Coping with Leishmaniasis: A Review of Control Strategies Centro de Convenções de Reboças 08.04.2014 Red Room 17: 00h Leishmaniasis - a Global Problem Visceral 2012 300 000 cases 20,000
Molecular diagnosis of. leishmaniasis. Carlos Alberto P Tavares, Ana Paula Fernandes and Maria Norma Melo. Author Proof
 Review Molecular diagnosis of leishmaniasis Carlos Alberto P Tavares, Ana Paula Fernandes and Maria Norma Melo CONTENTS Traditional leishmaniasis diagnostic approaches Diagnosis of visceral leishmaniasis
Review Molecular diagnosis of leishmaniasis Carlos Alberto P Tavares, Ana Paula Fernandes and Maria Norma Melo CONTENTS Traditional leishmaniasis diagnostic approaches Diagnosis of visceral leishmaniasis
Amastin Peptide-Binding Antibodies as Biomarkers of Active Human Visceral Leishmaniasis
 CLINICAL AND VACCINE IMMUNOLOGY, Oct. 2006, p. 1104 1110 Vol. 13, No. 10 1556-6811/06/$08.00 0 doi:10.1128/cvi.00188-06 Copyright 2006, American Society for Microbiology. All Rights Reserved. Amastin Peptide-Binding
CLINICAL AND VACCINE IMMUNOLOGY, Oct. 2006, p. 1104 1110 Vol. 13, No. 10 1556-6811/06/$08.00 0 doi:10.1128/cvi.00188-06 Copyright 2006, American Society for Microbiology. All Rights Reserved. Amastin Peptide-Binding
Visceral leishmaniasis: an endemic disease with global impact
 Visceral leishmaniasis: an endemic disease with global impact Professor Olivier Lortholary, MD, PhD Department of Infectious and Tropical diseases Hôpital Necker-Enfants Malades Université Paris Descartes
Visceral leishmaniasis: an endemic disease with global impact Professor Olivier Lortholary, MD, PhD Department of Infectious and Tropical diseases Hôpital Necker-Enfants Malades Université Paris Descartes
Diagnosis of Leishmania infantum using Direct Agglutination Test and rke16 Dipstick Rapid Test in Domestic Dogs from Ardabil Province, Iran
 Research Journal of Parasitology, 215 ISSN 1816-4943 / DOI: 1.3923/jp.215. 215 Academic Journals Inc. Diagnosis of Leishmania infantum using Direct Agglutination Test and rke16 Dipstick Rapid Test in Domestic
Research Journal of Parasitology, 215 ISSN 1816-4943 / DOI: 1.3923/jp.215. 215 Academic Journals Inc. Diagnosis of Leishmania infantum using Direct Agglutination Test and rke16 Dipstick Rapid Test in Domestic
CONTROLE DAS LEISHMANIOSES O QUE FALTA FAZER? Centro de Convenções de Reboças Red Room 17: 00h
 CONTROLE DAS LEISHMANIOSES O QUE FALTA FAZER? Centro de Convenções de Reboças 08.04.2014 Red Room 17: 00h Leishmaniasis - a Global Problem Visceral 2012 300 000 cases 20,000 deaths (6.7%) 310 million at
CONTROLE DAS LEISHMANIOSES O QUE FALTA FAZER? Centro de Convenções de Reboças 08.04.2014 Red Room 17: 00h Leishmaniasis - a Global Problem Visceral 2012 300 000 cases 20,000 deaths (6.7%) 310 million at
A Leishmania Infantum FML-ELISA for the Detection of Symptomatic and Asymptomatic Canine Visceral Leishmaniasis in an Endemic Area of Iran
 ISSN 1735-1383 Iran. J. Immunol. December 2011, 8 (4), 244-250 Behnam Mohammadi-Ghalehbin, Gholam Reza Hatam, Bahador Sarkari, Mehdi Mohebali, Zabih Zarei, Mansoureh Jaberipour, Shahab Bohlouli A Leishmania
ISSN 1735-1383 Iran. J. Immunol. December 2011, 8 (4), 244-250 Behnam Mohammadi-Ghalehbin, Gholam Reza Hatam, Bahador Sarkari, Mehdi Mohebali, Zabih Zarei, Mansoureh Jaberipour, Shahab Bohlouli A Leishmania
Canine leishmaniosis new concepts and insights on an expanding zoonosis: part two
 Review Canine leishmaniosis new concepts and insights on an expanding zoonosis: part two Guadalupe Miró 1*, Luis Cardoso 2*, Maria Grazia Pennisi 3*, Gaetano Oliva 4* and Gad Baneth 5* 1 Departmento de
Review Canine leishmaniosis new concepts and insights on an expanding zoonosis: part two Guadalupe Miró 1*, Luis Cardoso 2*, Maria Grazia Pennisi 3*, Gaetano Oliva 4* and Gad Baneth 5* 1 Departmento de
Series Editors Samuel J. Black, University of Massachusetts, Amherst, MA, US.A. J. Richard Seed, University of North Carolina, Chapel Hill, NC, US.A.
 LEISHMANIA World Class Parasites VOLUME 4 Volumes in the World Class Parasites book series are written for researchers, students and scholars who enjoy reading about excellent research on problems of global
LEISHMANIA World Class Parasites VOLUME 4 Volumes in the World Class Parasites book series are written for researchers, students and scholars who enjoy reading about excellent research on problems of global
ORIGINAL ARTICLE. The performance of serological tests for Leishmania infantum infection screening in dogs depends on the prevalence of the disease
 ORIGINAL ARTICLE http://dx.doi.org/10.1590/s1678-9946201759039 The performance of serological tests for Leishmania infantum infection screening in dogs depends on the prevalence of the disease Ivete Lopes
ORIGINAL ARTICLE http://dx.doi.org/10.1590/s1678-9946201759039 The performance of serological tests for Leishmania infantum infection screening in dogs depends on the prevalence of the disease Ivete Lopes
Incidence of Symptomatic and Asymptomatic Leishmania donovani Infections in High-Endemic Foci in India and Nepal: A Prospective Study
 Incidence of Symptomatic and Asymptomatic Leishmania donovani Infections in High-Endemic Foci in India and Nepal: A Prospective Study Bart Ostyn 1 *, Kamlesh Gidwani 2, Basudha Khanal 3, Albert Picado
Incidence of Symptomatic and Asymptomatic Leishmania donovani Infections in High-Endemic Foci in India and Nepal: A Prospective Study Bart Ostyn 1 *, Kamlesh Gidwani 2, Basudha Khanal 3, Albert Picado
Montenegro s skin reactions and antibodies against different Leishmania species in dogs from a visceral leishmaniosis endemic area
 Veterinary Parasitology 139 (2006) 21 28 www.elsevier.com/locate/vetpar Montenegro s skin reactions and antibodies against different Leishmania species in dogs from a visceral leishmaniosis endemic area
Veterinary Parasitology 139 (2006) 21 28 www.elsevier.com/locate/vetpar Montenegro s skin reactions and antibodies against different Leishmania species in dogs from a visceral leishmaniosis endemic area
Antibody Response against a Leishmania donovani Amastigote-Stage-Specific Protein in Patients with Visceral Leishmaniasis
 CLINICAL AND DIAGNOSTIC LABORATORY IMMUNOLOGY, Sept. 1997, p. 530 535 Vol. 4, No. 5 1071-412X/97/$04.00 0 Copyright 1997, American Society for Microbiology Antibody Response against a Leishmania donovani
CLINICAL AND DIAGNOSTIC LABORATORY IMMUNOLOGY, Sept. 1997, p. 530 535 Vol. 4, No. 5 1071-412X/97/$04.00 0 Copyright 1997, American Society for Microbiology Antibody Response against a Leishmania donovani
Subclinical Form of the American Visceral Leishmaniasis
 Mem Inst Oswaldo Cruz, Rio de Janeiro, Vol. 99(8): 889-893, December 2004 889 Subclinical Form of the American Visceral Leishmaniasis Mônica Elinor Alves Gama/ +, Jackson Maurício Lopes Costa*, Cláudia
Mem Inst Oswaldo Cruz, Rio de Janeiro, Vol. 99(8): 889-893, December 2004 889 Subclinical Form of the American Visceral Leishmaniasis Mônica Elinor Alves Gama/ +, Jackson Maurício Lopes Costa*, Cláudia
Toward Diagnosing Leishmania infantum Infection in Asymptomatic Dogs in an Area Where Leishmaniasis Is Endemic
 CLINICAL AND VACCINE IMMUNOLOGY, Mar. 2009, p. 337 343 Vol. 16, No. 3 1556-6811/09/$08.00 0 doi:10.1128/cvi.00268-08 Copyright 2009, American Society for Microbiology. All Rights Reserved. Toward Diagnosing
CLINICAL AND VACCINE IMMUNOLOGY, Mar. 2009, p. 337 343 Vol. 16, No. 3 1556-6811/09/$08.00 0 doi:10.1128/cvi.00268-08 Copyright 2009, American Society for Microbiology. All Rights Reserved. Toward Diagnosing
Children with Visceral Leishmaniasis Presented to Omdurman Emergency Hospital for Children
 Original Article Children with Visceral Leishmaniasis Presented to Omdurman Emergency Hospital for Children Elfakey Walyeldinl, Ahmed Muawial, Mohamed Abdurrahman2 and Suwar M 03 Department of Paediatrics
Original Article Children with Visceral Leishmaniasis Presented to Omdurman Emergency Hospital for Children Elfakey Walyeldinl, Ahmed Muawial, Mohamed Abdurrahman2 and Suwar M 03 Department of Paediatrics
Authors Babiker, Z O E; Davidson, R N N; Mazinda, C; Kipngetich, S; Ritmeijer, K
 MSF Field Research Utility of Lymph Node Aspiration in the Diagnosis of Visceral Leishmaniasis in Sudan. Authors Babiker, Z O E; Davidson, R N N; Mazinda, C; Kipngetich, S; Ritmeijer, K Citation Publisher
MSF Field Research Utility of Lymph Node Aspiration in the Diagnosis of Visceral Leishmaniasis in Sudan. Authors Babiker, Z O E; Davidson, R N N; Mazinda, C; Kipngetich, S; Ritmeijer, K Citation Publisher
Lesion aspirate culture for the diagnosis and isolation of Leishmania spp. from patients with cutaneous leishmaniasis
 62 Mem Inst Oswaldo Cruz, Rio de Janeiro, Vol. 104(1): 62-66, February 2009 Lesion aspirate culture for the diagnosis and isolation of Leishmania spp. from patients with cutaneous leishmaniasis Zélia Maria
62 Mem Inst Oswaldo Cruz, Rio de Janeiro, Vol. 104(1): 62-66, February 2009 Lesion aspirate culture for the diagnosis and isolation of Leishmania spp. from patients with cutaneous leishmaniasis Zélia Maria
Low prevalence of Leishmania donovani infection among the blood donors in kala-azar endemic areas of Bangladesh
 Huda et al. BMC Infectious Diseases 2013, 13:62 RESEARCH ARTICLE Open Access Low prevalence of Leishmania donovani infection among the blood donors in kala-azar endemic areas of Bangladesh M Mamun Huda
Huda et al. BMC Infectious Diseases 2013, 13:62 RESEARCH ARTICLE Open Access Low prevalence of Leishmania donovani infection among the blood donors in kala-azar endemic areas of Bangladesh M Mamun Huda
Leishmania major-like Antigen for Specific and Sensitive Serodiagnosis of Human and Canine Visceral Leishmaniasis
 CLINICAL AND DIAGNOSTIC LABORATORY IMMUNOLOGY, Nov. 2002, p. 1361 1366 Vol. 9, No. 6 1071-412X/02/$04.00 0 DOI: 10.1128/CDLI.9.6.1361 1366.2002 Copyright 2002, American Society for Microbiology. All Rights
CLINICAL AND DIAGNOSTIC LABORATORY IMMUNOLOGY, Nov. 2002, p. 1361 1366 Vol. 9, No. 6 1071-412X/02/$04.00 0 DOI: 10.1128/CDLI.9.6.1361 1366.2002 Copyright 2002, American Society for Microbiology. All Rights
The Application of Latent Class Analysis for Diagnostic Test Validation of Chronic Trypanosoma cruzi Infection in Blood Donors
 BJID 2002; 6 (August) 181 The Application of Latent Class Analysis for Diagnostic Test Validation of Chronic Trypanosoma cruzi Infection in Blood Donors Dante M. Langhi Junior, José O. Bordin, Adauto Castelo,
BJID 2002; 6 (August) 181 The Application of Latent Class Analysis for Diagnostic Test Validation of Chronic Trypanosoma cruzi Infection in Blood Donors Dante M. Langhi Junior, José O. Bordin, Adauto Castelo,
Parasite density and impaired biochemical/hematological status are associated with severe clinical aspects of canine visceral leishmaniasis
 Research in Veterinary Science 81 (2006) 68 75 www.elsevier.com/locate/rvsc Parasite density and impaired biochemical/hematological status are associated with severe clinical aspects of canine visceral
Research in Veterinary Science 81 (2006) 68 75 www.elsevier.com/locate/rvsc Parasite density and impaired biochemical/hematological status are associated with severe clinical aspects of canine visceral
ter Horst, Rachel; Tefera, Tewodros; Assefa, Gessesse; Ebrahim, Abdurazik Z; Davidson, Robert N; Ritmeijer, Koert
 MSF Field Research Field evaluation of rk39 test and direct agglutination test for diagnosis of visceral leishmaniasis in a population with high prevalence of human immunodeficiency virus in Ethiopia Item
MSF Field Research Field evaluation of rk39 test and direct agglutination test for diagnosis of visceral leishmaniasis in a population with high prevalence of human immunodeficiency virus in Ethiopia Item
Comparative evaluation of several methods for Canine Visceral Leishmaniasis diagnosis
 Braz. J. Vet. Parasitol., Jaboticabal, v. 23, n. 2, p. 179-186, abr.-jun. 2014 ISSN 0103-846X (Print) / ISSN 1984-2961 (Electronic) Doi: http://dx.doi.org/10.1590/s1984-29612014033 Original Article Comparative
Braz. J. Vet. Parasitol., Jaboticabal, v. 23, n. 2, p. 179-186, abr.-jun. 2014 ISSN 0103-846X (Print) / ISSN 1984-2961 (Electronic) Doi: http://dx.doi.org/10.1590/s1984-29612014033 Original Article Comparative
Prevalence of antibodies against visceralizing Leishmania spp. in brown rats from Grenada, West Indies
 Veterinary World, EISSN: 2231-0916 Available at www.veterinaryworld.org/vol.11/september-2018/19.pdf RESEARCH ARTICLE Open Access Prevalence of antibodies against visceralizing Leishmania spp. in brown
Veterinary World, EISSN: 2231-0916 Available at www.veterinaryworld.org/vol.11/september-2018/19.pdf RESEARCH ARTICLE Open Access Prevalence of antibodies against visceralizing Leishmania spp. in brown
Laboratory diagnosis of congenital infections
 Laboratory diagnosis of congenital infections Laboratory diagnosis of HSV Direct staining Tzanck test Immunostaining HSV isolation Serology PCR Tzanck test Cell scrape from base of the lesion smear on
Laboratory diagnosis of congenital infections Laboratory diagnosis of HSV Direct staining Tzanck test Immunostaining HSV isolation Serology PCR Tzanck test Cell scrape from base of the lesion smear on
Leishmaniasis: A forgotten disease among neglected people
 ISPUB.COM The Internet Journal of Health Volume 11 Number 2 Leishmaniasis: A forgotten disease among neglected people Y Homsi, G Makdisi Citation Y Homsi, G Makdisi. Leishmaniasis: A forgotten disease
ISPUB.COM The Internet Journal of Health Volume 11 Number 2 Leishmaniasis: A forgotten disease among neglected people Y Homsi, G Makdisi Citation Y Homsi, G Makdisi. Leishmaniasis: A forgotten disease
Received 18 April 2002/Accepted 3 June 2002
 CLINICAL AND DIAGNOSTIC LABORATORY IMMUNOLOGY, Sept. 2002, p. 1119 1123 Vol. 9, No. 5 1071-412X/02/$04.00 0 DOI: 10.1128/CDLI.9.5.1119 1123.2002 Copyright 2002, American Society for Microbiology. All Rights
CLINICAL AND DIAGNOSTIC LABORATORY IMMUNOLOGY, Sept. 2002, p. 1119 1123 Vol. 9, No. 5 1071-412X/02/$04.00 0 DOI: 10.1128/CDLI.9.5.1119 1123.2002 Copyright 2002, American Society for Microbiology. All Rights
1. Department of Tropical Medicine and Infectology, Universidade Federal do Triângulo
 CVI Accepts, published online ahead of print on 4 September 2013 Clin. Vaccine Immunol. doi:10.1128/cvi.00367-13 Copyright 2013, American Society for Microbiology. All Rights Reserved. 1 Title: 2 ANTI-LEISHMANIA
CVI Accepts, published online ahead of print on 4 September 2013 Clin. Vaccine Immunol. doi:10.1128/cvi.00367-13 Copyright 2013, American Society for Microbiology. All Rights Reserved. 1 Title: 2 ANTI-LEISHMANIA
Epidemiological and Serological Study of Leishmaniasis in Najran Region, Saudi Arabia
 ISSN 57-676 7, Vol. 8, No. Epidemiological and Serological Study of Leishmaniasis in Najran Region, Saudi Arabia Mokhtar I. Khalil Department of Applied Medical Sciences, Community College, Najran University,
ISSN 57-676 7, Vol. 8, No. Epidemiological and Serological Study of Leishmaniasis in Najran Region, Saudi Arabia Mokhtar I. Khalil Department of Applied Medical Sciences, Community College, Najran University,
Criteria of Chagas Disease Cure
 Mem Inst Oswaldo Cruz, Rio de Janeiro, Vol. 94, Suppl. I: 331-335, 1999 331 Criteria of Chagas Disease Cure J Romeu Cançado Cadeira de Terapêutica Clínica, Universidade Federal de Minas Gerais, Caixa Postal
Mem Inst Oswaldo Cruz, Rio de Janeiro, Vol. 94, Suppl. I: 331-335, 1999 331 Criteria of Chagas Disease Cure J Romeu Cançado Cadeira de Terapêutica Clínica, Universidade Federal de Minas Gerais, Caixa Postal
Application of a Direct Agglutination Test for Detection of Specific Anti-Leishmania Antibodies in the Canine Reservoir
 JOURNAL OF CLINICAL MICROBIOLOGY, Oct. 1989, p. 2252-2257 0095-1137/89/102252-06$02.00/0 Copyright C 1989, American Society for Microbiology Vol. 27, No. 10 Application of a Direct Agglutination Test for
JOURNAL OF CLINICAL MICROBIOLOGY, Oct. 1989, p. 2252-2257 0095-1137/89/102252-06$02.00/0 Copyright C 1989, American Society for Microbiology Vol. 27, No. 10 Application of a Direct Agglutination Test for
Imported Visceral Leishmaniasis: Diagnostic Dilemmas and Comparative Analysis of Three Assays
 JOURNAL OF CLINICAL MICROBIOLOGY, Feb. 2002, p. 475 479 Vol. 40, No. 2 0095-1137/02/$04.00 0 DOI: 10.1128/JCM.40.2.475 479.2002 Copyright 2002, American Society for Microbiology. All Rights Reserved. Imported
JOURNAL OF CLINICAL MICROBIOLOGY, Feb. 2002, p. 475 479 Vol. 40, No. 2 0095-1137/02/$04.00 0 DOI: 10.1128/JCM.40.2.475 479.2002 Copyright 2002, American Society for Microbiology. All Rights Reserved. Imported
Leishmania and Human Immunodeficiency Virus Coinfection: the First 10 Years
 CLINICAL MICROBIOLOGY REVIEWS, Apr. 1997, p. 298 319 Vol. 10, No. 2 0893-8512/97/$04.00 0 Copyright 1997, American Society for Microbiology Leishmania and Human Immunodeficiency Virus Coinfection: the
CLINICAL MICROBIOLOGY REVIEWS, Apr. 1997, p. 298 319 Vol. 10, No. 2 0893-8512/97/$04.00 0 Copyright 1997, American Society for Microbiology Leishmania and Human Immunodeficiency Virus Coinfection: the
LD Bodies From Blood Buffy Coat: an Easy Approach for Definitive Diagnosis of Visceral Leishmaniasis
 Bangladesh J Med Microbiol 2007; 01 (02): 43-47 Bangladesh Society of Medical Microbiologists Original Article LD Bodies From Blood Buffy Coat: an Easy Approach for Definitive Diagnosis of Visceral Leishmaniasis
Bangladesh J Med Microbiol 2007; 01 (02): 43-47 Bangladesh Society of Medical Microbiologists Original Article LD Bodies From Blood Buffy Coat: an Easy Approach for Definitive Diagnosis of Visceral Leishmaniasis
LEISHMANIA IFA SLIDE STORAGE REQUIREMENTS:
 1 STORAGE REQUIREMTS: SLEDO: Slides for the diagnosis of Leishmania infantum antibodies in human serum by indirect immunofluorescent assay (IFA). INTRODUCTION: Leishmania infantum is a causal agent of
1 STORAGE REQUIREMTS: SLEDO: Slides for the diagnosis of Leishmania infantum antibodies in human serum by indirect immunofluorescent assay (IFA). INTRODUCTION: Leishmania infantum is a causal agent of
INTRODUCTION MATERIALS AND METHODS
 Am. J. Trop. Med. Hyg., 67(4), 2002, pp. 344 348 Copyright 2002 by The American Society of Tropical Medicine and Hygiene THE SENSITIVITY AND SPECIFICITY OF LEISHMANIA CHAGASI RECOMBINANT K39 ANTIGEN IN
Am. J. Trop. Med. Hyg., 67(4), 2002, pp. 344 348 Copyright 2002 by The American Society of Tropical Medicine and Hygiene THE SENSITIVITY AND SPECIFICITY OF LEISHMANIA CHAGASI RECOMBINANT K39 ANTIGEN IN
Mapping cancer, cardiovascular and malaria research in Brazil
 Brazilian Journal of Medical and Biological Research (2) 33: 853-867 Mapping cancer, cardiovascular and malaria research ISSN 1-879X Overview 853 Mapping cancer, cardiovascular and malaria research in
Brazilian Journal of Medical and Biological Research (2) 33: 853-867 Mapping cancer, cardiovascular and malaria research ISSN 1-879X Overview 853 Mapping cancer, cardiovascular and malaria research in
PARASITOLOGY CASE HISTORY 15 (HISTOLOGY) (Lynne S. Garcia)
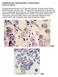 PARASITOLOGY CASE HISTORY 15 (HISTOLOGY) (Lynne S. Garcia) A biopsy was performed on a 27-year-old man with no known travel history, presenting with a perianal ulcer. The specimen was preserved in formalin
PARASITOLOGY CASE HISTORY 15 (HISTOLOGY) (Lynne S. Garcia) A biopsy was performed on a 27-year-old man with no known travel history, presenting with a perianal ulcer. The specimen was preserved in formalin
Received 24 November 1997/Returned for modification 11 February 1998/Accepted 6 April 1998
 CLINICAL AND DIAGNOSTIC LABORATORY IMMUNOLOGY, July 1998, p. 486 490 Vol. 5, No. 4 1071-412X/98/$04.00 0 Copyright 1998, American Society for Microbiology. All Rights Reserved. Seroprevalence of Antibodies
CLINICAL AND DIAGNOSTIC LABORATORY IMMUNOLOGY, July 1998, p. 486 490 Vol. 5, No. 4 1071-412X/98/$04.00 0 Copyright 1998, American Society for Microbiology. All Rights Reserved. Seroprevalence of Antibodies
Shing Faculty of Medicine, The University of Hong Kong, 21 Sassoon Road, Pokfulam, Hong Kong Special Administrative Region, China
 CVI Accepts, published online ahead of print on 4 January 2012 Clin. Vaccine Immunol. doi:10.1128/cvi.05358-11 Copyright 2012, American Society for Microbiology. All Rights Reserved. 1 2 Evaluation of
CVI Accepts, published online ahead of print on 4 January 2012 Clin. Vaccine Immunol. doi:10.1128/cvi.05358-11 Copyright 2012, American Society for Microbiology. All Rights Reserved. 1 2 Evaluation of
Post Kala-azar Dermal Leishmaniasis (PKDL) from the field to the cellular and the subcellular levels
 Post Kala-azar Dermal Leishmaniasis (PKDL) from the field to the cellular and the subcellular levels A M EL Hassan Institute of Endemic Diseases University of Khartoum Introduction PKDL is a VL related
Post Kala-azar Dermal Leishmaniasis (PKDL) from the field to the cellular and the subcellular levels A M EL Hassan Institute of Endemic Diseases University of Khartoum Introduction PKDL is a VL related
Archives of Veterinary Science ISSN X CHARACTERIZATION OF CANINE LEISHMANIASIS BY PCR-RFLP IN CUIABA, MATO GROSSO, BRAZIL
 Archives of Veterinary Science ISSN 1517-784X v.17, n.2, p.68-72, 2012 www.ser.ufpr.br/veterinary CHARACTERIZATION OF CANINE LEISHMANIASIS BY PCR-RFLP IN CUIABA, MATO GROSSO, BRAZIL Arleana do Bom Parto
Archives of Veterinary Science ISSN 1517-784X v.17, n.2, p.68-72, 2012 www.ser.ufpr.br/veterinary CHARACTERIZATION OF CANINE LEISHMANIASIS BY PCR-RFLP IN CUIABA, MATO GROSSO, BRAZIL Arleana do Bom Parto
Longitudinal Studies of Neutralizing Antibody Responses to Rotavirus in Stools and Sera of Children following Severe Rotavirus Gastroenteritis
 CLINICAL AND DIAGNOSTIC LABORATORY IMMUNOLOGY, Nov. 1998, p. 897 901 Vol. 5, No. 6 1071-412X/98/$04.00 0 Copyright 1998, American Society for Microbiology. All Rights Reserved. Longitudinal Studies of
CLINICAL AND DIAGNOSTIC LABORATORY IMMUNOLOGY, Nov. 1998, p. 897 901 Vol. 5, No. 6 1071-412X/98/$04.00 0 Copyright 1998, American Society for Microbiology. All Rights Reserved. Longitudinal Studies of
SEROLOGICAL STUDY OF VISCERAL LEISHMANIASIS IN WASSIT GOVERNOTATE
 SEROLOGICAL STUDY OF VISCERAL LEISHMANIASIS IN WASSIT GOVERNOTATE 1 Ibrahim A. Al Zubaidy, 2 Hasanain A. Al Saeedi & 3 Ban S.Al-Nasery 1,3 Zoonotic diseases researches unit/ College of Veterinary Medicine/
SEROLOGICAL STUDY OF VISCERAL LEISHMANIASIS IN WASSIT GOVERNOTATE 1 Ibrahim A. Al Zubaidy, 2 Hasanain A. Al Saeedi & 3 Ban S.Al-Nasery 1,3 Zoonotic diseases researches unit/ College of Veterinary Medicine/
Canine Visceral Leishmaniasis ( 31-Mar-2000 )
 In: Recent Advances in Canine Infectious Diseases, L. Carmichael (Ed.) Publisher: International Veterinary Information Service (www.ivis.org), Ithaca, New York, USA. Canine Visceral Leishmaniasis ( 31-Mar-2000
In: Recent Advances in Canine Infectious Diseases, L. Carmichael (Ed.) Publisher: International Veterinary Information Service (www.ivis.org), Ithaca, New York, USA. Canine Visceral Leishmaniasis ( 31-Mar-2000
Welcome to the Jungle! Dr Aileen Oon, 2017 Microbiology Registrar
 Welcome to the Jungle! Dr Aileen Oon, 2017 Microbiology Registrar AA 55M presented with sores on left olecranon and umbilical area Umbilical sores present for 3 weeks Left olecranon lesions for 1 week
Welcome to the Jungle! Dr Aileen Oon, 2017 Microbiology Registrar AA 55M presented with sores on left olecranon and umbilical area Umbilical sores present for 3 weeks Left olecranon lesions for 1 week
Leishmania Canine IgG -ELISA
 Leishmania Canine IgG -ELISA GWB-521229 www.genwaybio.com FOR RESEARCH USE ONLY Enzyme immunoassay for the qualitative detection of IgG-class antibodies against Leishmania in canine serum 1. INTRODUCTION
Leishmania Canine IgG -ELISA GWB-521229 www.genwaybio.com FOR RESEARCH USE ONLY Enzyme immunoassay for the qualitative detection of IgG-class antibodies against Leishmania in canine serum 1. INTRODUCTION
Natural infection of Didelphis aurita (Mammalia: Marsupialia) with Leishmania infantum in Brazil
 Carreira et al. Parasites & Vectors 2012, 5:111 RESEARCH Open Access Natural infection of Didelphis aurita (Mammalia: Marsupialia) with Leishmania infantum in Brazil João Carlos Araujo Carreira 1, Alba
Carreira et al. Parasites & Vectors 2012, 5:111 RESEARCH Open Access Natural infection of Didelphis aurita (Mammalia: Marsupialia) with Leishmania infantum in Brazil João Carlos Araujo Carreira 1, Alba
Prevalence of Cutaneous Leishmaniasis among HIV and Non-HIV Patients attending some Selected Hospitals in Jos Plateau State
 International Journal of Current Microbiology and Applied Sciences ISSN: 2319-7706 Volume 7 Number 06 (2018) Journal homepage: http://www.ijcmas.com Original Research Article https://doi.org/10.20546/ijcmas.2018.706.307
International Journal of Current Microbiology and Applied Sciences ISSN: 2319-7706 Volume 7 Number 06 (2018) Journal homepage: http://www.ijcmas.com Original Research Article https://doi.org/10.20546/ijcmas.2018.706.307
Validation of Two Rapid Diagnostic Tests for Visceral Leishmaniasis in Kenya
 Validation of Two Rapid Diagnostic Tests for Visceral Leishmaniasis in Kenya Jane Mbui 1, Monique Wasunna 1,2, Manica Balasegaram 3, Adrian Laussermayer 4, Rashid Juma 1, Simon Njoroge Njenga 1, George
Validation of Two Rapid Diagnostic Tests for Visceral Leishmaniasis in Kenya Jane Mbui 1, Monique Wasunna 1,2, Manica Balasegaram 3, Adrian Laussermayer 4, Rashid Juma 1, Simon Njoroge Njenga 1, George
Downloaded from:
 Campbell-Lendrum, D; Dujardin, JP; Martinez, E; Feliciangeli, MD; Perez, JE; de Silans, L; Desjeux, P (2001) Domestic and peridomestic transmission of American cutaneous leishmaniasis: Changing epidemiological
Campbell-Lendrum, D; Dujardin, JP; Martinez, E; Feliciangeli, MD; Perez, JE; de Silans, L; Desjeux, P (2001) Domestic and peridomestic transmission of American cutaneous leishmaniasis: Changing epidemiological
Antileishmanial IgG and IgE Antibodies Recognize Predominantly Carbohydrate Epitopes of Glycosylated Antigens in Visceral Leishmaniasis
 Mem Inst Oswaldo Cruz, Rio de Janeiro, Vol. 99(5): 525-530, August 2004 525 Antileishmanial IgG and IgE Antibodies Recognize Predominantly Carbohydrate Epitopes of Glycosylated Antigens in Visceral Leishmaniasis
Mem Inst Oswaldo Cruz, Rio de Janeiro, Vol. 99(5): 525-530, August 2004 525 Antileishmanial IgG and IgE Antibodies Recognize Predominantly Carbohydrate Epitopes of Glycosylated Antigens in Visceral Leishmaniasis
Leishmania (Viannia) naiffi: rare enough to be neglected?
 Mem Inst Oswaldo Cruz, Rio de Janeiro, Vol. 0(): 9-800, September 05 9 Leishmania (Viannia) naiffi: rare enough to be neglected? Giselle Aparecida Fagundes-Silva, Gustavo Adolfo Sierra Romero, Elisa Cupolillo
Mem Inst Oswaldo Cruz, Rio de Janeiro, Vol. 0(): 9-800, September 05 9 Leishmania (Viannia) naiffi: rare enough to be neglected? Giselle Aparecida Fagundes-Silva, Gustavo Adolfo Sierra Romero, Elisa Cupolillo
The Harvard community has made this article openly available. Please share how this access benefits you. Your story matters.
 Effectiveness of Insecticide Spraying and Culling of Dogs on the Incidence of Leishmania infantum Infection in Humans: A Cluster Randomized Trial in Teresina, Brazil The Harvard community has made this
Effectiveness of Insecticide Spraying and Culling of Dogs on the Incidence of Leishmania infantum Infection in Humans: A Cluster Randomized Trial in Teresina, Brazil The Harvard community has made this
Atypical cutaneous leishmaniasis cases display elevated antigen-induced interleukin-10
 Parasite Immunology, 2007, 29, 277 282 DOI: 10.1111/j.1365-3024.2007.00944.x Blackwell ORIGINAL Immune response Publishing ARTICLE in atypical Ltd cutaneous leishmaniasis Atypical cutaneous leishmaniasis
Parasite Immunology, 2007, 29, 277 282 DOI: 10.1111/j.1365-3024.2007.00944.x Blackwell ORIGINAL Immune response Publishing ARTICLE in atypical Ltd cutaneous leishmaniasis Atypical cutaneous leishmaniasis
INFLAMMATORY CELL INFILTRATION AND HIGH ANTIBODY PRODUCTION IN BALB/c MICE CAUSED BY NATURAL EXPOSURE TO LUTZOMYIA LONGIPALPIS BITES
 Am. J. Trop. Med. Hyg., 72(1), 2005, pp. 94 98 Copyright 2005 by The American Society of Tropical Medicine and Hygiene INFLAMMATORY CELL INFILTRATION AND HIGH ANTIBODY PRODUCTION IN BALB/c MICE CAUSED
Am. J. Trop. Med. Hyg., 72(1), 2005, pp. 94 98 Copyright 2005 by The American Society of Tropical Medicine and Hygiene INFLAMMATORY CELL INFILTRATION AND HIGH ANTIBODY PRODUCTION IN BALB/c MICE CAUSED
Serodiagnosis of Canine Babesiosis
 Serodiagnosis of Canine Babesiosis Xuenan XUAN, DVM, PhD National Research Center for Protozoan Diseases Obihiro University of Agriculture and Veterinary Medicine Obihiro, Hokkaido 080-8555, Japan Tel.:+81-155-495648;
Serodiagnosis of Canine Babesiosis Xuenan XUAN, DVM, PhD National Research Center for Protozoan Diseases Obihiro University of Agriculture and Veterinary Medicine Obihiro, Hokkaido 080-8555, Japan Tel.:+81-155-495648;
Visceral leishmaniasis: what are the needs for diagnosis, treatment and control?
 EVALUATING DIAGNOSTICS Visceral leishmaniasis: what are the needs for diagnosis, treatment and control? François Chappuis*, Shyam Sundar, Asrat Hailu, Hashim Ghalib, Suman Rijal #, Rosanna W. Peeling,
EVALUATING DIAGNOSTICS Visceral leishmaniasis: what are the needs for diagnosis, treatment and control? François Chappuis*, Shyam Sundar, Asrat Hailu, Hashim Ghalib, Suman Rijal #, Rosanna W. Peeling,
Diagnosis of Visceral Leishmaniasis by Enzyme-Linked Immunosorbent Assay Using Urine Samples
 CLINICAL AND DIAGNOSTIC LABORATORY IMMUNOLOGY, July 2002, p. 789 794 Vol. 9, No. 4 1071-412X/02/$04.00 0 DOI: 10.1128/CDLI.09.4.789 794.2002 Copyright 2002, American Society for Microbiology. All Rights
CLINICAL AND DIAGNOSTIC LABORATORY IMMUNOLOGY, July 2002, p. 789 794 Vol. 9, No. 4 1071-412X/02/$04.00 0 DOI: 10.1128/CDLI.09.4.789 794.2002 Copyright 2002, American Society for Microbiology. All Rights
Resolution of an Infection with Leishmania braziliensis Confers Complete Protection to a Subsequent Challenge with Leishmania major in BALB/c Mice
 Mem Inst Oswaldo Cruz, Rio de Janeiro, Vol. 94(1): 71-76, Jan./Feb. 1999 Resolution of an Infection with Leishmania braziliensis Confers Complete Protection to a Subsequent Challenge with Leishmania major
Mem Inst Oswaldo Cruz, Rio de Janeiro, Vol. 94(1): 71-76, Jan./Feb. 1999 Resolution of an Infection with Leishmania braziliensis Confers Complete Protection to a Subsequent Challenge with Leishmania major
Nanomedicine & Drug Delivery Systems Group Research Institute for Medicines and Pharmaceutical Sciences (imed.ul)
 Faculty of Pharmacy of the University of Lisbon Nanomedicine & Drug Delivery Systems Group Research Institute for Medicines and Pharmaceutical Sciences (imed.ul) Leishmania in southern Europe. (Duiardin,
Faculty of Pharmacy of the University of Lisbon Nanomedicine & Drug Delivery Systems Group Research Institute for Medicines and Pharmaceutical Sciences (imed.ul) Leishmania in southern Europe. (Duiardin,
Biomarkers for intracellular pathogens: establishing tools as vaccine and therapeutic endpoints for visceral leishmaniasis
 ORIGINAL ARTICLE PARASITOLOGICAL Biomarkers for intracellular pathogens: establishing tools as vaccine and therapeutic endpoints for visceral leishmaniasis A. C. Vallur 1, M. S. Duthie 1, C. Reinhart 1,
ORIGINAL ARTICLE PARASITOLOGICAL Biomarkers for intracellular pathogens: establishing tools as vaccine and therapeutic endpoints for visceral leishmaniasis A. C. Vallur 1, M. S. Duthie 1, C. Reinhart 1,
SEROLOGICAL STUDY OF VISCERAL LEISHMANIASIS IN WASSIT GOVERNOTATE
 SEROLOGICAL STUDY OF VISCERAL LEISHMANIASIS IN WASSIT GOVERNOTATE 1 Ibrahim A. Al Zubaidy, 2 Hasanain A. Al Saeedi & Ban S.Al-Nasiry 3 1,3 Zoonotic diseases researches unit/ College of Veterinary Medicine/
SEROLOGICAL STUDY OF VISCERAL LEISHMANIASIS IN WASSIT GOVERNOTATE 1 Ibrahim A. Al Zubaidy, 2 Hasanain A. Al Saeedi & Ban S.Al-Nasiry 3 1,3 Zoonotic diseases researches unit/ College of Veterinary Medicine/
Immunologic Indicators of Clinical Progression during Canine Leishmania infantum Infection
 CLINICAL AND VACCINE IMMUNOLOGY, Feb. 2010, p. 267 273 Vol. 17, No. 2 1556-6811/10/$12.00 doi:10.1128/cvi.00456-09 Copyright 2010, American Society for Microbiology. All Rights Reserved. Immunologic Indicators
CLINICAL AND VACCINE IMMUNOLOGY, Feb. 2010, p. 267 273 Vol. 17, No. 2 1556-6811/10/$12.00 doi:10.1128/cvi.00456-09 Copyright 2010, American Society for Microbiology. All Rights Reserved. Immunologic Indicators
Key words: malaria - Plasmodium vivax - merozoite antigens - IgG antibody response MATERIALS AND METHODS
 Mem Inst Oswaldo Cruz, Rio de Janeiro, Vol. 102(3): 335-339, June 2007 335 Comparative recognition by human IgG antibodies of recombinant proteins representing three asexual erythrocytic stage vaccine
Mem Inst Oswaldo Cruz, Rio de Janeiro, Vol. 102(3): 335-339, June 2007 335 Comparative recognition by human IgG antibodies of recombinant proteins representing three asexual erythrocytic stage vaccine
J07 Titer dynamics, complement fixation test and neutralization tests
 avllm0421c (spring 2017) J07 Titer dynamics, complement fixation test and neutralization tests Outline titer, antibody titer dynamics complement, complement fixation reaction neutralization tests 2/35
avllm0421c (spring 2017) J07 Titer dynamics, complement fixation test and neutralization tests Outline titer, antibody titer dynamics complement, complement fixation reaction neutralization tests 2/35
Evaluation of a prototype flow cytometry test for serodiagnosis of canine visceral. leishmaniasis
 CVI Accepts, published online ahead of print on 9 October 2013 Clin. Vaccine Immunol. doi:10.1128/cvi.00575-13 Copyright 2013, American Society for Microbiology. All Rights Reserved. 1 2 Evaluation of
CVI Accepts, published online ahead of print on 9 October 2013 Clin. Vaccine Immunol. doi:10.1128/cvi.00575-13 Copyright 2013, American Society for Microbiology. All Rights Reserved. 1 2 Evaluation of
FACTORS ASSOCIATED WITH VISCERAL LEISHMANIASIS IN NEPAL: BED-NET USE IS STRONGLY PROTECTIVE
 Am. J. Trop. Med. Hyg., 63(3, 4), 2000, pp. 184 188 Copyright 2000 by The American Society of Tropical Medicine and Hygiene FACTORS ASSOCIATED WITH VISCERAL LEISHMANIASIS IN NEPAL: BED-NET USE IS STRONGLY
Am. J. Trop. Med. Hyg., 63(3, 4), 2000, pp. 184 188 Copyright 2000 by The American Society of Tropical Medicine and Hygiene FACTORS ASSOCIATED WITH VISCERAL LEISHMANIASIS IN NEPAL: BED-NET USE IS STRONGLY
Intralesional meglumine antimoniate for the treatment of localised cutaneous leishmaniasis: a retrospective review of a Brazilian referral centre
 512 Mem Inst Oswaldo Cruz, Rio de Janeiro, Vol. 111(8): 512-516, August 2016 Intralesional meglumine antimoniate for the treatment of localised cutaneous leishmaniasis: a retrospective review of a Brazilian
512 Mem Inst Oswaldo Cruz, Rio de Janeiro, Vol. 111(8): 512-516, August 2016 Intralesional meglumine antimoniate for the treatment of localised cutaneous leishmaniasis: a retrospective review of a Brazilian
Neglected Diseases (NDs) Landscape in Brazil and South America
 Frontiers in Science on Neglected Diseases 13 th November 2014 Neglected Diseases (NDs) Landscape in Brazil and South America Jeffrey Shaw São Paulo University Biomedical Sciences Institute jeffreyj@usp.br
Frontiers in Science on Neglected Diseases 13 th November 2014 Neglected Diseases (NDs) Landscape in Brazil and South America Jeffrey Shaw São Paulo University Biomedical Sciences Institute jeffreyj@usp.br
