CBER update and International Collaboration for development of HIV variant panels
|
|
|
- Antonia Norman
- 6 years ago
- Views:
Transcription
1 CBER update and International Collaboration for development of HIV variant panels Indira K. Hewlett, Ph.D Chief, Laboratory of Molecular Virology DETTD/CBER/FDA XXII SoGAT meeting
2 HIV genetic diversity: subtypes and homology 70% J A B C I H HIV-1 M G F E D 50% A HIV-2 70% B HIV-1 O 60% HIV-1 N 60% cpz SIV 75% mac/sm
3 Two major types : - HIV-1 and HIV-2; Genetic diversity of HIV - 4 HIV-1 groups : M (major), O (outlier), N (non-m, non-o), P Group M has multiple subtypes (A-H) and up to 50 Circulating Recombinant Forms (CRFs) HIV-2 has 5 major subtypes A and B are most prevalent HIV diversity is due to: - High error rate of HIV RT and absence of proof reading mechanisms - Rapid viral turnover (10 10 viral particles/day) in infected individuals - Zoonotic transmission characteristic of lentiviruses
4 Evolution of HIV-1 Genetic Variants Group O 1993 HIV Subtypes Nomenclature 1998 CRF 2001 Co-infection, dual- & super-infections URF A URFKenya Variation HIV-2 Recombination Up to 50 CRFs have been identified Tanzania Uganda URF URF A A C D 2004 SGRs 2 nd gen. recom binant
5 Worldwide distribution of predominant HIV-1 group M subtypes and CRFs Eestern Europe A CRF03_AB Western Europe North and Central America B South America B, F1, CRF12_BF B, A, C, G CRF14_BG Western Africa CRF02_AG A, G Central Africa Most CRFs, A, C, D, G, H, J, K, O. N East Africa A, D, C B CRF01_AE South Asia C China CRF07_BC, CRF08_BC Southeast Asia CRF01_AE B B South Africa C Australia B
6 Impact of HIV genetic diversity on diagnosis Schable, C., et al Lancet. (1994) Sensitivity of US licensed antibody tests for detection of HIV-1 group O infection. 344, Amendola, et al., J AIDS, (2002) Under-evaluation of HIV -1 Plasma Viral Load by a Commercially Available Assay in a Cluster of Patients Infected With HIV - 1 A/G Circulating Recombinant Form (CRF02). Colson, P., et al J. Clin. Virol., Impaired quantification of plasma HIV-1 RNA with a commercialized real-time PCR assay in a couple of HIV-1 infected individuals Zouhair, S., et al JCM, (2006), Group O HIV-1 infection that escaped detection in two immunoassays Lee, S., et al. AIDS Res and Hum Retr.( 2007) Detection of Emerging HIV variants in blood donors from urban areas of Cameroon Yao JD, et al. J. Clin. Micro., (2008). Plasma Load Discrepancies between the Roche Cobas Amplicor Human Immunodeficiency Virus Type 1 (HIV-1) Monitor Version 1.5 and Roche Cobas AmpliPrep/Cobas TaqMan HIV-1 Assays
7 CBER HIV panel development Project initiated in Cameroon, region of high HIV diversity to study virus evolution and have access to new strains for panel development. Study Goals: Collect specimens, viruses; genotyping and virus tropism Evaluate sensitivity of blood screening and diagnostic tests for ability to detect diverse strains Identify new strains for future use as reference reagents
8 CBER HIV-1 RNA genotype panel HIV-1 RNA genotype panel currently in use for testing. Panel consists of one primary isolate each of group M subtypes A-G and groups O and N cultured in PBMC and characterized by sequencing. Virus isolates are inactivated by heat treatment at 60 C for 60 mins. Panel composed of 3 members of each subtype at 10 3, 10 4 and 10 5 copies/ml spiked in negative plasma.
9 CBER HIV-2 RNA panel Seven isolates of HIV-2 subtype A were cultured in PBMC cultures and characterized by partial sequencing Viruses were inactivated by heat treatment (60 C/60 minutes and spiked into negative plasma Testing was performed in 3 laboratories Results from labs were in good agreement HIV-2 panel was formulated at 5, 10, 50,100 copies/ml and is currently in use for lot release testing of HIV-2 NAT assays
10 CBER HIV-2 Panel Isolate Testing Summary (Log 10) Isolate ID Manufacturer Manufacturer Manufacturer Mean Standard A B C Deviation B B3 B4 B B B B
11 CBER panel for CRF02_AG and CRF01_AE Emerging new HIV strains are circulating recombinant forms (CRFs) CRF02_AG and CRF01_AE are currently the most prevalent strains; need for standards Five primary virus isolates each of CRF02_AG and CRF01_AE cultured in PBMCs were characterized by full genome sequencing Heat inactivated virus isolates spiked into negative plasma were tested in five laboratories Panel consists of 3 members for each isolate spiked in negative plasma at 10 3, 10 4 and 10 5 copies/ml (log)
12 CRF01_AE and CRF02_AG Isolate Testing Summary Isolate ID Lab A Lab B Lab C Lab D Lab E Mean SD AE AE AE AE AE NYU NYU n/a NYU n/a NYU NYU
13 HIV-1 BC and BF NAT Panel development l l l l l CBER obtained viruses from NIBSC, UK and Carlos de Salud institute, Spain and in-house project in Cameroon Viruses were cultured in PBMC to high titers. Viral stocks were heat-treated to inactivate the virus, no loss of RNA copy number Aliquots were sent to multiple collaborating labs for copy number determination Data analysis ongoing; final formulation expected in the near future
14 B/C B/F Isolate Titer Testing Summary(log 10 ) Isolate ID Lab A Lab B Lab C Mean Standard Deviation P X /
15 NIAID Global HIV Viral Panels Project: Purpose To establish a set of fully characterized viruses from early acute HIV infections that are consistent with the degree of viral evolution present globally, for - Developing new assays - Validating assay platforms - Assisting regulators to evaluate test kits - Monitoring HIV drug resistance - Informing vaccine development
16 HIV Viral Panels Project Requirements Well characterized HIV reference panels encompassing epidemic Full length single genome sequencing Verified RNA concentration Fiebig staging and serological profiles for current assays/platforms including rapid POC assays Comparisons of VL from different FDA approved commercially available platforms Panels with larger volumes for use on newer diagnostic platforms
17 Phase I (Feasibility) HIV Viral Panels: Pilot study 20 pre or very early post-sc plasma units South Africa M. Vermeulen (SANBS) USA S. Stramer (ARC) Brazil E. Sabino (REDS II) Cameroon I.Hewlett (FDA) Standardize assays and validated protocols Phase II (Expansion) DAIDS EQAPOL awarded to Duke and started FY11 (7 year contract) Dynamic 60 member panels: updated and rebalanced as epidemic evolves Focus on window phase and recent seroconversions HIV reference stocks housed within four panels: 1. Major subtypes/crf (Group M) 2. Emerging strains (rare subtypes/crf_cpx, Group N, O, HIV-2) 3. Challenging strains (VL assay variation) 4. Subtype representation from diverse geographical locations
18 Tier 1 Isolates Subtype A1 B C D G CRF01_AE CRF02_AG Region of Interest Uganda, Rwanda, former Soviet Republics (IDU) North America, Western Europe, Australia, Western South America South Africa, Botswana, Zambia, Malawi, Tanzania, Ethiopia, India, Southern Brazil Uganda Nigeria, Spain (IDU), Portugal (IDU) Thailand, Vietnam, Cambodia Senegal, Nigeria, Ghana, Cote d Ivoire, Cameroon
19 Tier 2 Isolates Subtype Region of Interest F1 Brazil, Romania, Spain F2 Cameroon H, J, K DRC, Cameroon, Congo CRF04_cpx (A,G,H,K,U) Cyprus, Greece CRF05_DF DRC, Belgium CRF06_cpx (A,G,J,K) Niger, Mali, Cote d Ivoire CRF07_BC China CRF08_BC China CRF09_cpx (02,A,U) Cote d Ivoire, Mali CRF11_cpx (A,G,01,J) Cameroon, DRC, CAR CRF12_BF Argentina CRF13_cpx (A,01,11,G,J,U) Cameroon, CAR CRF14_BG Spain, Portugal CRF18_cpx (A,F,G,H,K,U) Cuba CRF20_BG, CRF23_BG, CRF24_BG Cuba CRF31 Brazil
20 HIV Viral Panels: Early Goals and Accomplishments 1. Goal to complete a pilot study before end of FY pre or very early post-sc plasma units from 4 countries (US, SA, Brazil, Cameroon) Additional European countries added mid year Obtain country support and resolve IRB issues and logistical challenges; standardize procedures 2. Accomplishments Identified needed strains and geographic locations Partnering with different groups to collaborate and pool resources Procured, isolated, propagated and characterized over 30 isolates Continued support from EQAPOL (additional 7 years)
21 Viral Isolation Summary - Samples (I. Hewlett, M. Ramaswamy, E. Brojer, K. Roth, S. Laperche) - Lab analysis by M. Manak, B. Coombs, T. Denny - Repository at SeraCare (M. Manak and P. Garrett) Success rate
22 HIV Viral Panels: Plasma summary Obtained Plasma from early stage HIV infection Plasma centers in US, S Africa, and Brazil Large vol (~100 ml) back up available Collaborations for rare subtypes Greece (NIBSC), Cameroon (FDA) Collaborations with other countries: Poland, Germany, France More (Spain, Thailand, India, Egypt, Japan) coming on board Plasma Characterization Fiebig Stage assignment Viral Load p24 Antigen EIA and Western blot Full Length Genome Sequencing Virus Infectivity/Virus Isolation
23 HIV Viral Panels: Cultured Virus Summary Optimized virus Isolation conditions from plasma Clarification by ultracentrifugation Infection of CD8-depleted PHA stimulated donor PBMC Success rate correlates with VL (65% success for VL >30,000 cp/ml) Standardized Virus Expansion Maximize viral yield at low passage number Expand in PBMC to 100 ml; Most at >10 8 copies/ml Characterization of Expanded Virus Subtype Designation based on Full Length sequencing Confirm identify to original plasma Co-receptor usage (CCR5/CXCR4) Viral Load, p24 antigen, Infectivity Consistent source of High Titer HIV isolates Comparison of quantitative NAT assays Infectivity studies for Drug and Vaccine design/evaluation
24 Acknowledgments CBER Owen Wood Sherwin Lee Ragupathy Viswanath Jiangqin Zhao Shixing Tang Siva Gaddam Stephen Kerby Supported by NHLBI IAG- Y1-HB NIAID IAA -Y1-A CBER Critical Path Funds NYU/Cameroon Phillipe Nyambi Denis Barengolts Panel Testing Participants NIAID Global Panels group Marco Schito Michael Busch Mark Manak Sodsai Stovanabutra CHAVI group
ISBT WP-TTID Annual Report for Subgroup on Virology Drs. Michael Busch, Kurt Roth and Susan Stramer
 ISBT WP-TTID Annual Report for Subgroup on Virology Drs. Michael Busch, Kurt Roth and Susan Stramer Questionnaire on NAT Screening of Blood Donations for an International Forum on 10 years of NAT Screening
ISBT WP-TTID Annual Report for Subgroup on Virology Drs. Michael Busch, Kurt Roth and Susan Stramer Questionnaire on NAT Screening of Blood Donations for an International Forum on 10 years of NAT Screening
Results of Pilot NIH Study of Global HIV Variants
 Results of Pilot NIH Study of Global HIV Variants SoGAT Blood Virology Meeting Vilnius, Lithuania - 16-17 April 2012 Mark Manak, Ph.D. MHRP, USA The opinions expressed herein are those of the authors and
Results of Pilot NIH Study of Global HIV Variants SoGAT Blood Virology Meeting Vilnius, Lithuania - 16-17 April 2012 Mark Manak, Ph.D. MHRP, USA The opinions expressed herein are those of the authors and
Diagnostic Procurement The Clinton Foundation HIV/AIDS Initiative
 Diagnostic Procurement The Clinton Foundation HIV/AIDS Initiative 28 October 2008 Geneva 1 CHAI Work in Diagnostics - Context Diagnostic Test Agreements - In 2002 when CHAI was formed, the price of many
Diagnostic Procurement The Clinton Foundation HIV/AIDS Initiative 28 October 2008 Geneva 1 CHAI Work in Diagnostics - Context Diagnostic Test Agreements - In 2002 when CHAI was formed, the price of many
HIV-1 Subtypes: An Overview. Anna Maria Geretti Royal Free Hospital
 HIV-1 Subtypes: An Overview Anna Maria Geretti Royal Free Hospital Group M Subtypes A (1, 2, 3) B C D F (1, 2) G H J K Mechanisms of HIV-1 genetic diversification Point mutations RT error rate: ~1 per
HIV-1 Subtypes: An Overview Anna Maria Geretti Royal Free Hospital Group M Subtypes A (1, 2, 3) B C D F (1, 2) G H J K Mechanisms of HIV-1 genetic diversification Point mutations RT error rate: ~1 per
Impact of changing donor demographics on HIV transmission risk: A ten year analysis of individual donation NAT screening
 Impact of changing donor demographics on HIV transmission risk: A ten year analysis of individual donation NAT screening IPFA Lisbon 216 Marion Vermeulen Disclaimer Race classifications are described as
Impact of changing donor demographics on HIV transmission risk: A ten year analysis of individual donation NAT screening IPFA Lisbon 216 Marion Vermeulen Disclaimer Race classifications are described as
Calibration and stability of WHO and secondary viral standards
 Calibration and stability of WHO and secondary viral standards Nico Lelie, Harry van Drimmelen and the International NAT Study Group Facilities DDL Diagnostic Laboratories Outline Calibration of WHO and
Calibration and stability of WHO and secondary viral standards Nico Lelie, Harry van Drimmelen and the International NAT Study Group Facilities DDL Diagnostic Laboratories Outline Calibration of WHO and
HIV and Harm Reduction in Prisons
 HIV and Harm Reduction in Prisons School of Public Health and Community Medicine Professor Kate Dolan Program of International Research and Training National Drug and Alcohol Research Centre UNSW, Sydney,
HIV and Harm Reduction in Prisons School of Public Health and Community Medicine Professor Kate Dolan Program of International Research and Training National Drug and Alcohol Research Centre UNSW, Sydney,
Infectious Disease Testing. ULTRA Product Line. Safety is not a Matter of Chance
 Infectious Disease Testing ULTRA Product Line Safety is not a Matter of Chance ULTRA Product Line The best answer for HBV, HCV and HIV screening: a global automated solution for safe results. Monolisa
Infectious Disease Testing ULTRA Product Line Safety is not a Matter of Chance ULTRA Product Line The best answer for HBV, HCV and HIV screening: a global automated solution for safe results. Monolisa
Introduction: Table/Figure Descriptions:
 Introduction: We have completed the analysis of your HIV RNA Validation Study. The validation plan was designed to verify the installation of an unmodified FDA-approved HIV RNA assay into your laboratory.
Introduction: We have completed the analysis of your HIV RNA Validation Study. The validation plan was designed to verify the installation of an unmodified FDA-approved HIV RNA assay into your laboratory.
Pandemic H1N Dr. Maria Neira Global Influenza Programme WHO, Geneva
 Pandemic H1N1 2010 Dr. Maria Neira Global Influenza Programme WHO, Geneva WHO Role during pandemic (H1N1) 2009 Under the International Health Regulations (2005) Detect event (notification by Member States
Pandemic H1N1 2010 Dr. Maria Neira Global Influenza Programme WHO, Geneva WHO Role during pandemic (H1N1) 2009 Under the International Health Regulations (2005) Detect event (notification by Member States
Copyright 2011 Joint United Nations Programme on HIV/AIDS (UNAIDS) All rights reserved ISBN
 UNAIDS DATA TABLES 2011 Copyright 2011 Joint United Nations Programme on HIV/AIDS (UNAIDS) All rights reserved ISBN 978-92-9173-945-5 UNAIDS / JC2225E The designations employed and the presentation of
UNAIDS DATA TABLES 2011 Copyright 2011 Joint United Nations Programme on HIV/AIDS (UNAIDS) All rights reserved ISBN 978-92-9173-945-5 UNAIDS / JC2225E The designations employed and the presentation of
Influenza Surveillance In the WHO African Region
 Vol. 2 N 52, Updated on 2 January 207 Contents Highlights ackground Methodology Review of 206 influenza virus circulation Seasonal patterns of influenza transmission Way forward Conclusion Editor Dr Ibramima
Vol. 2 N 52, Updated on 2 January 207 Contents Highlights ackground Methodology Review of 206 influenza virus circulation Seasonal patterns of influenza transmission Way forward Conclusion Editor Dr Ibramima
Current State of Global HIV Care Continua. Reuben Granich 1, Somya Gupta 1, Irene Hall 2, John Aberle-Grasse 2, Shannon Hader 2, Jonathan Mermin 2
 Current State of Global HIV Care Continua Reuben Granich 1, Somya Gupta 1, Irene Hall 2, John Aberle-Grasse 2, Shannon Hader 2, Jonathan Mermin 2 1) International Association of Providers of AIDS Care
Current State of Global HIV Care Continua Reuben Granich 1, Somya Gupta 1, Irene Hall 2, John Aberle-Grasse 2, Shannon Hader 2, Jonathan Mermin 2 1) International Association of Providers of AIDS Care
WHO Parvovirus B19 Genotype Panel
 WHO Parvovirus B19 Genotype Panel Mei-ying W Yu, PhD SoGAT XXII Rome, 14-15 April 2011 1 st WHO International Reference Panel for Parvovirus B19 Genotypes In Oct 2009, a plasma-derived parvovirus B19 (B19V)
WHO Parvovirus B19 Genotype Panel Mei-ying W Yu, PhD SoGAT XXII Rome, 14-15 April 2011 1 st WHO International Reference Panel for Parvovirus B19 Genotypes In Oct 2009, a plasma-derived parvovirus B19 (B19V)
ARCHITECT HIV Ag/Ab Combo: Moving HIV Diagnostics Forward in the U.S.
 ARCHITECT HIV Ag/Ab Combo: Moving HIV Diagnostics Forward in the U.S. Catherine Brennan, Ph.D. Research Fellow Infectious Diseases Research Abbott Diagnostics 1 Agenda ARCHITECT HIV Ag/Ab Combo Assay What
ARCHITECT HIV Ag/Ab Combo: Moving HIV Diagnostics Forward in the U.S. Catherine Brennan, Ph.D. Research Fellow Infectious Diseases Research Abbott Diagnostics 1 Agenda ARCHITECT HIV Ag/Ab Combo Assay What
Main global and regional trends
 I N T R O D U C T I O N Main global and regional trends Promising developments have been seen in recent years in global efforts to address the AS epidemic, including increased access to effective treatment
I N T R O D U C T I O N Main global and regional trends Promising developments have been seen in recent years in global efforts to address the AS epidemic, including increased access to effective treatment
Virus Panels for Assessing Vaccine-Elicited Neutralizing Antibodies
 Virus Panels for Assessing Vaccine-Elicited Neutralizing Antibodies Michael Seaman, Ph.D. Center for Virology and Vaccine Research Beth Israel Deaconess Medical Center Harvard Medical School J. Virol.
Virus Panels for Assessing Vaccine-Elicited Neutralizing Antibodies Michael Seaman, Ph.D. Center for Virology and Vaccine Research Beth Israel Deaconess Medical Center Harvard Medical School J. Virol.
Sex steroid hormone influences replication of major HIV subtypes
 Sex steroid hormone influences replication of major HIV subtypes Viswanath Ragupathy, Ph.D CBER/FDA Bethesda, MD st International Workshop on HIV & Women from Adolescence through Menopause Washington DC,
Sex steroid hormone influences replication of major HIV subtypes Viswanath Ragupathy, Ph.D CBER/FDA Bethesda, MD st International Workshop on HIV & Women from Adolescence through Menopause Washington DC,
New HIV Tests and Algorithm: A change we can believe in
 New HIV Tests and Algorithm: A change we can believe in Esther Babady, PhD, D (ABMM) Memorial Sloan-Kettering Cancer Center New York, New York Learning Objectives After this presentation you should be
New HIV Tests and Algorithm: A change we can believe in Esther Babady, PhD, D (ABMM) Memorial Sloan-Kettering Cancer Center New York, New York Learning Objectives After this presentation you should be
Regulatory Capacity Building and Developing Countries: CBER Perspective
 Regulatory Capacity Building and Developing Countries: CBER Perspective Karen Midthun, M.D. Director Center for Biologics Evaluation and Research US Food and Drug Administration IOM Meeting Strengthening
Regulatory Capacity Building and Developing Countries: CBER Perspective Karen Midthun, M.D. Director Center for Biologics Evaluation and Research US Food and Drug Administration IOM Meeting Strengthening
Weekly Influenza Surveillance Report. Week 11
 Weekly Influenza Surveillance Report Week 11 Report produced: 22/03/2001 Influenza activity in Ireland For the week ending the 18/03/01, week 11, influenza activity has increased. Sentinel general practices
Weekly Influenza Surveillance Report Week 11 Report produced: 22/03/2001 Influenza activity in Ireland For the week ending the 18/03/01, week 11, influenza activity has increased. Sentinel general practices
Lessons learned from the IeDEA West Africa Collaboration
 Lessons learned from the IeDEA West Africa Collaboration François DABIS with the contribution of Didier Koumavi EKOUEVI INSERM U-897, Bordeaux, France, Programme PACCI, ANRS site, Abidjan, Côte d Ivoire
Lessons learned from the IeDEA West Africa Collaboration François DABIS with the contribution of Didier Koumavi EKOUEVI INSERM U-897, Bordeaux, France, Programme PACCI, ANRS site, Abidjan, Côte d Ivoire
Influenza Update N 176
 Update N 176 04 January 2013 Summary Reporting of influenza activity has been irregular in the past two weeks due to the holiday season in many countries. As a result, overall virus detections have dropped
Update N 176 04 January 2013 Summary Reporting of influenza activity has been irregular in the past two weeks due to the holiday season in many countries. As a result, overall virus detections have dropped
THE CARE WE PROMISE FACTS AND FIGURES 2017
 THE CARE WE PROMISE FACTS AND FIGURES 2017 2 SOS CHILDREN S VILLAGES INTERNATIONAL WHERE WE WORK Facts and Figures 2017 205 58 79 families and transit 31 Foster homes 162 8 3 173 214 2 115 159 136 148
THE CARE WE PROMISE FACTS AND FIGURES 2017 2 SOS CHILDREN S VILLAGES INTERNATIONAL WHERE WE WORK Facts and Figures 2017 205 58 79 families and transit 31 Foster homes 162 8 3 173 214 2 115 159 136 148
Recommended composition of influenza virus vaccines for use in the 2007 influenza season
 Recommended composition of influenza virus vaccines for use in the 2007 influenza season September 2006 This recommendation relates to the composition of vaccines for the forthcoming winter in the southern
Recommended composition of influenza virus vaccines for use in the 2007 influenza season September 2006 This recommendation relates to the composition of vaccines for the forthcoming winter in the southern
New tools for diagnosis and surveillance
 New tools for diagnosis and surveillance Beth A Skaggs, PhD Laboratory Advisor, Division of Global HIV/AIDS CDC-Mozambique AIDS MEDICINES AND DIAGNOSTICS (AMDS) ANNUAL STAKEHOLDERS AND PARTNERS MEETING
New tools for diagnosis and surveillance Beth A Skaggs, PhD Laboratory Advisor, Division of Global HIV/AIDS CDC-Mozambique AIDS MEDICINES AND DIAGNOSTICS (AMDS) ANNUAL STAKEHOLDERS AND PARTNERS MEETING
HIV DIAGNOSTIC TESTS IN LOW- AND MIDDLE-INCOME COUNTRIES: FORECASTS OF GLOBAL DEMAND FOR
 HIV DIAGNOSTIC TESTS IN LOW- AND MIDDLE-INCOME COUNTRIES: FORECASTS OF GLOBAL DEMAND FOR 2015 2020 Presented by Vincent Habiyambere (WHO), Biyi Adesina (Avenir Health) and Meghan Wareham (CHAI) on behalf
HIV DIAGNOSTIC TESTS IN LOW- AND MIDDLE-INCOME COUNTRIES: FORECASTS OF GLOBAL DEMAND FOR 2015 2020 Presented by Vincent Habiyambere (WHO), Biyi Adesina (Avenir Health) and Meghan Wareham (CHAI) on behalf
Experience with Standardisation of Blood Virology NAT. Clare Morris Division of Retrovirology National Institute for Biological Standards and Control
 Experience with Standardisation of Blood Virology NAT Clare Morris Division of Retrovirology National Institute for Biological Standards and Control Background of Blood Virology Standardisation In the
Experience with Standardisation of Blood Virology NAT Clare Morris Division of Retrovirology National Institute for Biological Standards and Control Background of Blood Virology Standardisation In the
The MenAfrivac Experience A Successful Approach. Dr Bernard FRITZELL BFL conseils France
 The MenAfrivac Experience A Successful Approach Dr Bernard FRITZELL BFL conseils France 1 2 1996 Ministers of Health and Interior from 16 African countries recognized epidemic meningitis as a high priority
The MenAfrivac Experience A Successful Approach Dr Bernard FRITZELL BFL conseils France 1 2 1996 Ministers of Health and Interior from 16 African countries recognized epidemic meningitis as a high priority
Global Measles and Rubella Update November 2018
 Global Measles and Rubella Update November 218 Measles Number of Reported Measles by WHO Regions 218 Region Member States* Suspected cases Measles cases Clin Epi Lab Jan Feb Mar Apr May Jun Jul Aug Sep
Global Measles and Rubella Update November 218 Measles Number of Reported Measles by WHO Regions 218 Region Member States* Suspected cases Measles cases Clin Epi Lab Jan Feb Mar Apr May Jun Jul Aug Sep
Impact Dashboard - October 2014
 Impact Dashboard - 2014 By 2014, PSI and its network members averted an estimated 44.2 million DALYs globally. PSI has met 60.6% of the strategic plan (SP) target to avert 198.7 million DALYs, and 56.4%
Impact Dashboard - 2014 By 2014, PSI and its network members averted an estimated 44.2 million DALYs globally. PSI has met 60.6% of the strategic plan (SP) target to avert 198.7 million DALYs, and 56.4%
Trends in molecular diagnostics
 Trends in molecular diagnostics Detection of target genes of interest Quantification Infectious diseases HIV Hepatitis C & B TB / MAC Cytomegalovirus Herpes simplex Varicella zoster CT/GC HPV Profiling
Trends in molecular diagnostics Detection of target genes of interest Quantification Infectious diseases HIV Hepatitis C & B TB / MAC Cytomegalovirus Herpes simplex Varicella zoster CT/GC HPV Profiling
Impact Dashboard - August 2014
 Impact Dashboard - By, PSI and its network members averted an estimated 29.7 million DALYs globally. PSI has met 53.3% of the strategic plan (SP) target to avert 198.7 million DALYs, and 53.0% of the SP
Impact Dashboard - By, PSI and its network members averted an estimated 29.7 million DALYs globally. PSI has met 53.3% of the strategic plan (SP) target to avert 198.7 million DALYs, and 53.0% of the SP
SGCEP SCIE 1121 Environmental Science Spring 2012 Section Steve Thompson:
 SGCEP SCIE 1121 Environmental Science Spring 2012 Section 20531 Steve Thompson: steventhompson@sgc.edu http://www.bioinfo4u.net/ 1 First, a brief diversion... Into... how to do better on the next exam,
SGCEP SCIE 1121 Environmental Science Spring 2012 Section 20531 Steve Thompson: steventhompson@sgc.edu http://www.bioinfo4u.net/ 1 First, a brief diversion... Into... how to do better on the next exam,
Micropathology Ltd. University of Warwick Science Park, Venture Centre, Sir William Lyons Road, Coventry CV4 7EZ
 www.micropathology.com info@micropathology.com Micropathology Ltd Tel 24hrs: +44 (0) 24-76 323222 Fax / Ans: +44 (0) 24-76 - 323333 University of Warwick Science Park, Venture Centre, Sir William Lyons
www.micropathology.com info@micropathology.com Micropathology Ltd Tel 24hrs: +44 (0) 24-76 323222 Fax / Ans: +44 (0) 24-76 - 323333 University of Warwick Science Park, Venture Centre, Sir William Lyons
Update on the. Clinical Diagnostics. and. Blood Testing. Meetings of SoGAT. Micha Nübling, PEI.
 Update on the www.pei.de Clinical Diagnostics and Blood Testing Meetings of SoGAT Micha Nübling, PEI SoGAT Since 1995: 22 SoGAT meetings (organized by NIBSC) 1-2 meetings/year (connected with IPFA workshop)
Update on the www.pei.de Clinical Diagnostics and Blood Testing Meetings of SoGAT Micha Nübling, PEI SoGAT Since 1995: 22 SoGAT meetings (organized by NIBSC) 1-2 meetings/year (connected with IPFA workshop)
Procedure for Expedited Review of imported pre-qualified vaccines for use in national immunization programmes
 Procedure for Expedited Review of imported pre-qualified vaccines for use in national immunization programmes Dr Nora Dellepiane/Dr Anil Kumar Chawla WHO/HQ-Geneva, Switzerland 1 Expedited review procedure
Procedure for Expedited Review of imported pre-qualified vaccines for use in national immunization programmes Dr Nora Dellepiane/Dr Anil Kumar Chawla WHO/HQ-Geneva, Switzerland 1 Expedited review procedure
Global Measles and Rubella Update. April 2018
 Global Measles and Rubella Update April 218 Measles Number of Reported Measles by WHO Regions 218 Region Member States* Suspected cases Measles cases Clin Epi Lab Jan Feb Mar Apr May Jun Jul Aug Sep Oct
Global Measles and Rubella Update April 218 Measles Number of Reported Measles by WHO Regions 218 Region Member States* Suspected cases Measles cases Clin Epi Lab Jan Feb Mar Apr May Jun Jul Aug Sep Oct
Global Measles and Rubella Update October 2018
 Global Measles and Rubella Update October 218 Measles Number of Reported Measles Cases by WHO Regions 218 Region Member States* Suspected cases Measles cases Clin Epi Lab Jan Feb Mar Apr May Jun Jul Aug
Global Measles and Rubella Update October 218 Measles Number of Reported Measles Cases by WHO Regions 218 Region Member States* Suspected cases Measles cases Clin Epi Lab Jan Feb Mar Apr May Jun Jul Aug
Evolving Realities of HIV Treatment in Resource-limited Settings
 Evolving Realities of HIV Treatment in Resource-limited Settings Papa Salif Sow MD, MSc Department of Infectious Diseases University of Dakar, Senegal Introduction: ARV access in RLS Scale-up of ART has
Evolving Realities of HIV Treatment in Resource-limited Settings Papa Salif Sow MD, MSc Department of Infectious Diseases University of Dakar, Senegal Introduction: ARV access in RLS Scale-up of ART has
Experiences on Workforce Development in Other Regions
 Experiences on Workforce Development in Other Regions The USA Experience John MacArthur, MD, MPH (EIS 98) Director Thailand MOPH US CDC Collaboration CDC/Southeast Asia Regional Office Epidemic Intelligence
Experiences on Workforce Development in Other Regions The USA Experience John MacArthur, MD, MPH (EIS 98) Director Thailand MOPH US CDC Collaboration CDC/Southeast Asia Regional Office Epidemic Intelligence
Undetectable = Untransmittable. Mariah Wilberg Communications Specialist
 Undetectable = Untransmittable Mariah Wilberg Communications Specialist Undetectable=Untransmittable PLWH who get and stay undetectable have effectively no risk of transmitting HIV to their sex partners
Undetectable = Untransmittable Mariah Wilberg Communications Specialist Undetectable=Untransmittable PLWH who get and stay undetectable have effectively no risk of transmitting HIV to their sex partners
HIV Viral Load Testing Market Analysis. September 2012 Laboratory Services Team Clinton Health Access Initiative
 HIV Viral Load Testing Market Analysis September 2012 Laboratory Services Team Clinton Health Access Initiative Agenda Background on Viral Load Testing Growth of Global Viral Load Market Factors Impacting
HIV Viral Load Testing Market Analysis September 2012 Laboratory Services Team Clinton Health Access Initiative Agenda Background on Viral Load Testing Growth of Global Viral Load Market Factors Impacting
Complicated viral infections
 Complicated viral infections Clinical case discussion Diagnostic dilemmas NSW State Reference Laboratory for HIV St Vincent s Hospital Sydney Diagnostic dilemmas Indeterminate or discordant serology (western
Complicated viral infections Clinical case discussion Diagnostic dilemmas NSW State Reference Laboratory for HIV St Vincent s Hospital Sydney Diagnostic dilemmas Indeterminate or discordant serology (western
Objectives of Rotavirus Surveillance
 Objectives of Rotavirus Surveillance Contribute data to estimate the burden of rotavirus diarrhea in children < 5 children Document circulating rotavirus strains in the AFR region Support awareness and
Objectives of Rotavirus Surveillance Contribute data to estimate the burden of rotavirus diarrhea in children < 5 children Document circulating rotavirus strains in the AFR region Support awareness and
Dr. Natella Rakhmanina Senior Technical Advisor, Elizabeth Glaser Pediatric AIDS Foundation October 2018
 Dr. Natella Rakhmanina Senior Technical Advisor, Elizabeth Glaser Pediatric AIDS Foundation October 2018 1 Number of young people living with HIV (aged 15 19 years) by mode of HIV acquisition, 25 countries,*
Dr. Natella Rakhmanina Senior Technical Advisor, Elizabeth Glaser Pediatric AIDS Foundation October 2018 1 Number of young people living with HIV (aged 15 19 years) by mode of HIV acquisition, 25 countries,*
High Failure Rate of the ViroSeq HIV-1 Genotyping System for Drug Resistance Testing in Cameroon, a Country with Broad HIV-1 Genetic Diversity
 JOURNAL OF CLINICAL MICROBIOLOGY, Apr. 2011, p. 1635 1641 Vol. 49, No. 4 0095-1137/11/$12.00 doi:10.1128/jcm.01478-10 Copyright 2011, American Society for Microbiology. All Rights Reserved. High Failure
JOURNAL OF CLINICAL MICROBIOLOGY, Apr. 2011, p. 1635 1641 Vol. 49, No. 4 0095-1137/11/$12.00 doi:10.1128/jcm.01478-10 Copyright 2011, American Society for Microbiology. All Rights Reserved. High Failure
TRACKING THE HIV/AIDS EPIDEMIC: ISSUES, CHALLENGES and FUTURE DIRECTIONS. November 2015 CHIPTS
 TRACKING THE HIV/AIDS EPIDEMIC: ISSUES, CHALLENGES and FUTURE DIRECTIONS November 2015 CHIPTS CHINA Mortality trends HIV/AIDS mortality increase 1/100,000 4.0 3.5 3.0 2.5 2.0 1.5 1.0 0.5 0.0 0-14 15-49
TRACKING THE HIV/AIDS EPIDEMIC: ISSUES, CHALLENGES and FUTURE DIRECTIONS November 2015 CHIPTS CHINA Mortality trends HIV/AIDS mortality increase 1/100,000 4.0 3.5 3.0 2.5 2.0 1.5 1.0 0.5 0.0 0-14 15-49
New Generation of Nucleic Acid Testing. Michele Owen, Ph.D Division of HIV/AIDS Prevention Centers for Disease Control & Prevention
 New Generation of Nucleic Acid Testing Michele Owen, Ph.D Division of HIV/AIDS Prevention Centers for Disease Control & Prevention Percentage (%) Persons Living with Diagnosed or Undiagnosed HIV Infection
New Generation of Nucleic Acid Testing Michele Owen, Ph.D Division of HIV/AIDS Prevention Centers for Disease Control & Prevention Percentage (%) Persons Living with Diagnosed or Undiagnosed HIV Infection
Recommended composition of influenza virus vaccines for use in the influenza season
 Recommended composition of influenza virus vaccines for use in the 2009 2010 influenza season February 2009 This recommendation relates to the composition of vaccines for the forthcoming influenza season
Recommended composition of influenza virus vaccines for use in the 2009 2010 influenza season February 2009 This recommendation relates to the composition of vaccines for the forthcoming influenza season
Sourcing of ARVs & HIV diagnostics. Procurement for Impact P4i
 Sourcing of ARVs & HIV diagnostics Procurement for Impact P4i AMDS ANNUAL STAKEHOLDERS AND PARTNERS MEETING 29 September 2014 Global Fund: Procurement for Impact: P4i Our objective was straightforward:
Sourcing of ARVs & HIV diagnostics Procurement for Impact P4i AMDS ANNUAL STAKEHOLDERS AND PARTNERS MEETING 29 September 2014 Global Fund: Procurement for Impact: P4i Our objective was straightforward:
Role and missions of a National Reference center for blood borne agents: the French example
 Role and missions of a National Reference center for blood borne agents: the French example Virginie Sauvage, PhD Département des Agents Transmissibles par le Sang, Centre National de Référence pour les
Role and missions of a National Reference center for blood borne agents: the French example Virginie Sauvage, PhD Département des Agents Transmissibles par le Sang, Centre National de Référence pour les
Yellow fever laboratory capacity on-site assessments in Africa: preliminary findings
 Yellow fever laboratory capacity on-site assessments in Africa: preliminary findings Maurice Demanou*, Barbara W. Johnson, Gamou Fall, Jean-Luc Betoulle, Chantal Reusken, Marion Koopmans, Lee Hampton,
Yellow fever laboratory capacity on-site assessments in Africa: preliminary findings Maurice Demanou*, Barbara W. Johnson, Gamou Fall, Jean-Luc Betoulle, Chantal Reusken, Marion Koopmans, Lee Hampton,
511,000 (57% new cases) ~50,000 ~30,000
 Latest global TB estimates - 2007 (Updated Mar 2009) All forms of TB Greatest number of cases in Asia; greatest rates per capita in Africa Multidrug-resistant TB (MDR-TB) Estimated number of cases 9.27
Latest global TB estimates - 2007 (Updated Mar 2009) All forms of TB Greatest number of cases in Asia; greatest rates per capita in Africa Multidrug-resistant TB (MDR-TB) Estimated number of cases 9.27
West Nile Disease. World situation and impact on public health
 West Nile Disease World situation and impact on public health Contents Global view on WNV The Public Health consequences: the case of USA WNV circulation in Europe and Mediterranean Basin West Nile Disease
West Nile Disease World situation and impact on public health Contents Global view on WNV The Public Health consequences: the case of USA WNV circulation in Europe and Mediterranean Basin West Nile Disease
HIV Viral Load Quality Assessment Program Summary for Panel HIVVL 2018Oct26
 1 National Laboratory for HIV Reference Services National HIV and Retrovirology Laboratories National Microbiology Laboratory Public Health Agency of Canada HIV Viral Load Quality Assessment Program Summary
1 National Laboratory for HIV Reference Services National HIV and Retrovirology Laboratories National Microbiology Laboratory Public Health Agency of Canada HIV Viral Load Quality Assessment Program Summary
UNAIDS 2013 AIDS by the numbers
 UNAIDS 2013 AIDS by the numbers 33 % decrease in new HIV infections since 2001 29 % decrease in AIDS-related deaths (adults and children) since 2005 52 % decrease in new HIV infections in children since
UNAIDS 2013 AIDS by the numbers 33 % decrease in new HIV infections since 2001 29 % decrease in AIDS-related deaths (adults and children) since 2005 52 % decrease in new HIV infections in children since
AccuVert HBV Seroconversion Panel PHM941(M) ( )
 PACKAGE INSERT PHM941(M) (0605-0061) INTENDED USE PHM941(M) (0605-0061) is a group of serial bleeds from an individual plasma donor during HBV seroconversion. This panel is intended for use by diagnostics
PACKAGE INSERT PHM941(M) (0605-0061) INTENDED USE PHM941(M) (0605-0061) is a group of serial bleeds from an individual plasma donor during HBV seroconversion. This panel is intended for use by diagnostics
A smart and doable investment
 90-90-90 A smart and doable investment As of December 2013 Adults and children living with HIV Adults and children newly infected Adult & child deaths due to AIDS 35.0 million [33.2 million 37.2 million]
90-90-90 A smart and doable investment As of December 2013 Adults and children living with HIV Adults and children newly infected Adult & child deaths due to AIDS 35.0 million [33.2 million 37.2 million]
Technical Bulletin No. 161
 CPAL Central Pennsylvania Alliance Laboratory Technical Bulletin No. 161 cobas 6800 HIV-1 Viral Load Assay - New Platform - June 1, 2017 Contact: Heather Habig, MLS (ASCP) CM, MB CM, 717-851-1422 Operations
CPAL Central Pennsylvania Alliance Laboratory Technical Bulletin No. 161 cobas 6800 HIV-1 Viral Load Assay - New Platform - June 1, 2017 Contact: Heather Habig, MLS (ASCP) CM, MB CM, 717-851-1422 Operations
Challenges in HBV detec1on in blood donors
 Challenges in HBV detec1on in blood donors Jean- Pierre Allain Dept Haematology, University of Cambridge, UK Phylogene1c analysis of human and ape HBV over 1me of evolu1on (Pareskevis et al, Hepatology
Challenges in HBV detec1on in blood donors Jean- Pierre Allain Dept Haematology, University of Cambridge, UK Phylogene1c analysis of human and ape HBV over 1me of evolu1on (Pareskevis et al, Hepatology
TB Disease Prevalence Survey - Progress Report
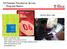 TB Disease Prevalence Survey - Progress Report 30 Oct 2011, Lille Ikushi Onozaki MD, MPH Team Leader, TB Prevalence Survey WHO Global Task Force on TB Impact Measurement Stop TB Department, WHO onozakii@who.int
TB Disease Prevalence Survey - Progress Report 30 Oct 2011, Lille Ikushi Onozaki MD, MPH Team Leader, TB Prevalence Survey WHO Global Task Force on TB Impact Measurement Stop TB Department, WHO onozakii@who.int
GLOBAL AND REGIONAL SITUATION OF AVIAN INFLUENZA
 GLOBAL AND REGIONAL SITUATION OF AVIAN INFLUENZA Dr Gounalan Pavade OIE Regional Expert Group Meeting for the Control of Avian Influenza in Asia Sapporo, Japan 3-5 October 2017 1 Avian influenza Outbreaks
GLOBAL AND REGIONAL SITUATION OF AVIAN INFLUENZA Dr Gounalan Pavade OIE Regional Expert Group Meeting for the Control of Avian Influenza in Asia Sapporo, Japan 3-5 October 2017 1 Avian influenza Outbreaks
Wild Poliovirus*, 03 Aug 2004 to 02 Aug 2005
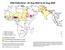 Wild Poliovirus*, 03 Aug 2004 to 02 Aug 2005 Wild virus type 1 Wild virus type 3 Wild virus type 1 & 3 Endemic countries Re-established transmission countries Case or outbreak following importation *Excludes
Wild Poliovirus*, 03 Aug 2004 to 02 Aug 2005 Wild virus type 1 Wild virus type 3 Wild virus type 1 & 3 Endemic countries Re-established transmission countries Case or outbreak following importation *Excludes
The Global Movement to Eliminate Viral Hepatitis. John W Ward, MD Task Force for Global Health CDC, Atlanta, Georgia, USA
 The Global Movement to Eliminate Viral Hepatitis John W Ward, MD Task Force for Global Health CDC, Atlanta, Georgia, USA Infection and Disease Control, Elimination and Eradication Control Below acceptable
The Global Movement to Eliminate Viral Hepatitis John W Ward, MD Task Force for Global Health CDC, Atlanta, Georgia, USA Infection and Disease Control, Elimination and Eradication Control Below acceptable
Overview of HIV Testing Practices and Technology
 Overview of HIV Testing Practices and Technology 2019 HIV Diagnostics Conference March 25 th, 2019 Michele Owen, Ph.D National Center for HIV/AIDS, Viral Hepatitis, STD and TB Prevention Centers for Disease
Overview of HIV Testing Practices and Technology 2019 HIV Diagnostics Conference March 25 th, 2019 Michele Owen, Ph.D National Center for HIV/AIDS, Viral Hepatitis, STD and TB Prevention Centers for Disease
Projected Demand for HIV Diagnostic Tests
 Projected Demand for HIV Diagnostic Tests John Stover, Yu Teng, Adebiyi Adesina, Eline Korenromp On behalf of the Diagnostics Forecasting Technical Working Group (CDC, CHAI, GFATM, PfSCM, UNAIDS, UNICEF,
Projected Demand for HIV Diagnostic Tests John Stover, Yu Teng, Adebiyi Adesina, Eline Korenromp On behalf of the Diagnostics Forecasting Technical Working Group (CDC, CHAI, GFATM, PfSCM, UNAIDS, UNICEF,
Annex 2 A. Regional profile: West Africa
 Annex 2 A. Regional profile: West Africa 355 million people at risk for malaria in 215 297 million at high risk A. Parasite prevalence, 215 Funding for malaria increased from US$ 233 million to US$ 262
Annex 2 A. Regional profile: West Africa 355 million people at risk for malaria in 215 297 million at high risk A. Parasite prevalence, 215 Funding for malaria increased from US$ 233 million to US$ 262
Emergence and Changes in swine and avian influenza viruses of Pandemic Potential
 Emergence and Changes in swine and avian influenza viruses of Pandemic Potential Fourth Annual SARInet Meeting May 23-26, 2017 Punta Cana, Dominican Republic Stephen Lindstrom, Ph.D. Diagnostics Development
Emergence and Changes in swine and avian influenza viruses of Pandemic Potential Fourth Annual SARInet Meeting May 23-26, 2017 Punta Cana, Dominican Republic Stephen Lindstrom, Ph.D. Diagnostics Development
Proposed 1 st IS for Hepatitis E Virus RNA WHO/BS/
 www.pei.de Proposed 1 st IS for Hepatitis E Virus RNA WHO/BS/09.2126 S. Baylis, Division of Virology, Paul-Ehrlich-Institut, Langen, Germany SoGAT XXII 14 th -15 th April 2011, Rome, Italy Presented at:
www.pei.de Proposed 1 st IS for Hepatitis E Virus RNA WHO/BS/09.2126 S. Baylis, Division of Virology, Paul-Ehrlich-Institut, Langen, Germany SoGAT XXII 14 th -15 th April 2011, Rome, Italy Presented at:
Contribution of avian influenza data through OFFLU network
 Dr Gounalan Pavade Chargé de Mission, OIE Headquarters Contribution of avian influenza data through OFFLU network Asia-Pacific Workshop on surveillance, prevention and control of zoonotic influenza Paro,
Dr Gounalan Pavade Chargé de Mission, OIE Headquarters Contribution of avian influenza data through OFFLU network Asia-Pacific Workshop on surveillance, prevention and control of zoonotic influenza Paro,
JOINT TB AND HIV PROGRAMMING
 JOINT TB AND HIV PROGRAMMING Haileyesus Getahun, WHO. On behalf of the Global Fund Interagency TB and HIV Working Group (Global Fund, PEPFAR, Stop TB Partnership, UNAIDS, WHO) I was admitted in a hospital
JOINT TB AND HIV PROGRAMMING Haileyesus Getahun, WHO. On behalf of the Global Fund Interagency TB and HIV Working Group (Global Fund, PEPFAR, Stop TB Partnership, UNAIDS, WHO) I was admitted in a hospital
A Summary of Clinical Evidence
 A Summary of Clinical Evidence Supporting the use of the Alere Determine HIV-1/2 Ag/Ab Combo Rapid Test to assist in the diagnosis of Human Immunodeficiency Virus (HIV) TAP HERE TO SEE THE PRODUCTS Table
A Summary of Clinical Evidence Supporting the use of the Alere Determine HIV-1/2 Ag/Ab Combo Rapid Test to assist in the diagnosis of Human Immunodeficiency Virus (HIV) TAP HERE TO SEE THE PRODUCTS Table
Aboubacar Kampo Chief of Health UNICEF Nigeria
 Aboubacar Kampo Chief of Health UNICEF Nigeria Many thanks to UNICEF colleagues in Supply Division-Copenhagen and NY for contributing to this presentation Thirty-five countries are responsible for 98%
Aboubacar Kampo Chief of Health UNICEF Nigeria Many thanks to UNICEF colleagues in Supply Division-Copenhagen and NY for contributing to this presentation Thirty-five countries are responsible for 98%
10 Years of Laboratory Capacity Building. September 6-11 th, 2015 Les Pensieres, Veyrier-du-Lac, France
 10 Years of Laboratory Capacity Building 6 th Advanced Course on Diagnostics September 6-11 th, 2015 Les Pensieres, Veyrier-du-Lac, France John N. Nkengasong, Ph.D Associate Director for Laboratory & Chief,
10 Years of Laboratory Capacity Building 6 th Advanced Course on Diagnostics September 6-11 th, 2015 Les Pensieres, Veyrier-du-Lac, France John N. Nkengasong, Ph.D Associate Director for Laboratory & Chief,
Access to treatment and disease burden
 Access to treatment and disease burden Robert Flisiak Department of Infectious Diseases and Hepatology Medical University in Białystok, Poland Moulin de Vernègues, 27-29 August 2015 Disclosures Advisor
Access to treatment and disease burden Robert Flisiak Department of Infectious Diseases and Hepatology Medical University in Białystok, Poland Moulin de Vernègues, 27-29 August 2015 Disclosures Advisor
Influenza Update N 157
 Influenza Update N 157 13 April 2012 Summary In most areas of the northern hemisphere temperate regions, influenza activity appears to have peaked and is declining. In North America, influenza indicators
Influenza Update N 157 13 April 2012 Summary In most areas of the northern hemisphere temperate regions, influenza activity appears to have peaked and is declining. In North America, influenza indicators
United Kingdom National External Quality Assessment Service for Microbiology [Established 1971]
![United Kingdom National External Quality Assessment Service for Microbiology [Established 1971] United Kingdom National External Quality Assessment Service for Microbiology [Established 1971]](/thumbs/95/123501270.jpg) United Kingdom National External Quality Assessment Service for Microbiology [Established 97] UK NEQAS covers Andrology Chemistry Genetics Haematology Histopathology Immunology Leucocyte Immunophenotyping
United Kingdom National External Quality Assessment Service for Microbiology [Established 97] UK NEQAS covers Andrology Chemistry Genetics Haematology Histopathology Immunology Leucocyte Immunophenotyping
Vaccines against Rotavirus & Norovirus. Umesh D. Parashar CDC, Atlanta, GA
 TM Vaccines against Rotavirus & Norovirus Umesh D. Parashar CDC, Atlanta, GA 1 Rotavirus is the Leading Cause Of Severe Diarrhea in Children
TM Vaccines against Rotavirus & Norovirus Umesh D. Parashar CDC, Atlanta, GA 1 Rotavirus is the Leading Cause Of Severe Diarrhea in Children
WPTTID / Virology subgroup Activities
 WPTTID / Virology subgroup 2010 2011 Activities Progress on previously launched collaborative Projects International Survey on NAT Testing of Blood Donations: Expanding Implementation and Yield from 1999
WPTTID / Virology subgroup 2010 2011 Activities Progress on previously launched collaborative Projects International Survey on NAT Testing of Blood Donations: Expanding Implementation and Yield from 1999
Global reductions in measles mortality and the risk of measles resurgence
 Global reductions in measles mortality 2000 2008 and the risk of measles resurgence Measles is one of the most contagious human diseases. In 1980 before the use of measles vaccine was widespread, there
Global reductions in measles mortality 2000 2008 and the risk of measles resurgence Measles is one of the most contagious human diseases. In 1980 before the use of measles vaccine was widespread, there
HIV transmission in the migrant population. D. Paraskevis
 HIV transmission in the migrant population D. Paraskevis Content Definitions Epidemiology of HIV in migrants Methods to estimate the origin of HIV acquisition Molecular epidemiology Bayes method based
HIV transmission in the migrant population D. Paraskevis Content Definitions Epidemiology of HIV in migrants Methods to estimate the origin of HIV acquisition Molecular epidemiology Bayes method based
Achieving undetectable: what questions remain in scaling-up HIV virologic treatment monitoring?
 !! SUPPLEMENTARY MATERIAL FOR ISSUE BRIEF: Achieving undetectable: what questions remain in scaling-up H virologic treatment? Supplementary material includes national recommendations on infant diagnosis,
!! SUPPLEMENTARY MATERIAL FOR ISSUE BRIEF: Achieving undetectable: what questions remain in scaling-up H virologic treatment? Supplementary material includes national recommendations on infant diagnosis,
Developing diagnostics for resource-limited settings
 Developing diagnostics for resource-limited settings Helen Lee University of Cambridge Trends in diagnostics: becoming a bimodal distribution! Primary health clinics! Emergency rooms! Hospital labs! Clinical
Developing diagnostics for resource-limited settings Helen Lee University of Cambridge Trends in diagnostics: becoming a bimodal distribution! Primary health clinics! Emergency rooms! Hospital labs! Clinical
Guidance for Travelers on Temporary Work Assignment Abroad
 infected person to immediately seek medical care but, prior to arrival, notify their healthcare provider that they may have been exposed to AI. For more information about avian influenza, see www.cdc.gov/flu/avian/facts.htm,
infected person to immediately seek medical care but, prior to arrival, notify their healthcare provider that they may have been exposed to AI. For more information about avian influenza, see www.cdc.gov/flu/avian/facts.htm,
Global Plan towards the elimination of new HIV infections among children by 2015 and keeping their mothers alive
 Global Plan towards the elimination of new HIV infections among children by 2015 and keeping their mothers alive DR. Nicholas Muraguri OGW, MD,MPH, MBA, PhD (c) Director, Global Plan Secretariat www.zero-hiv.org
Global Plan towards the elimination of new HIV infections among children by 2015 and keeping their mothers alive DR. Nicholas Muraguri OGW, MD,MPH, MBA, PhD (c) Director, Global Plan Secretariat www.zero-hiv.org
FAO GLOBAL INITIATIVES ON REGIONAL VETERINARY LABORATORY NETWORKS
 FAO GLOBAL INITIATIVES ON REGIONAL VETERINARY LABORATORY NETWORKS Gwenaelle Dauphin Laboratory Liaison Officer OFFLU focal point and several FAO colleagues Support to national laboratories Laboratory training
FAO GLOBAL INITIATIVES ON REGIONAL VETERINARY LABORATORY NETWORKS Gwenaelle Dauphin Laboratory Liaison Officer OFFLU focal point and several FAO colleagues Support to national laboratories Laboratory training
CHAPTER FOUR: UPDATE ON THE AIDS PANDEMIC TABLE 2 MILESTONES IN THE HISTORY OF HIV/AIDS TABLE 2 (CONTINUED) REGIONAL OVERVIEW...
 CHAPTER ONE: INTRODUCTION... 1 STUDY GOALS AND OBJECTIVES... 1 REASONS FOR DOING THE STUDY... 1 SCOPE OF REPORT... 1 MARKET ANALYSES AND FORECASTS... 2 INTENDED AUDIENCE... 2 INFORMATION SOURCES... 2 ANALYST
CHAPTER ONE: INTRODUCTION... 1 STUDY GOALS AND OBJECTIVES... 1 REASONS FOR DOING THE STUDY... 1 SCOPE OF REPORT... 1 MARKET ANALYSES AND FORECASTS... 2 INTENDED AUDIENCE... 2 INFORMATION SOURCES... 2 ANALYST
HIBA ABDALRAHIM Capsca Focal Point Public Health Authority
 HIBA ABDALRAHIM Capsca Focal Point Public Health Authority Introduction Definition Symptom Transmission Global situation Local situation Control Content Introduction Yellow fever (YF) is a mosquito-borne
HIBA ABDALRAHIM Capsca Focal Point Public Health Authority Introduction Definition Symptom Transmission Global situation Local situation Control Content Introduction Yellow fever (YF) is a mosquito-borne
David L. Suarez D.V.M., PhD. A.C.V.M.
 David L. Suarez D.V.M., PhD. A.C.V.M. Southeast Poultry Research Laboratory United States National Poultry Research Center The author has no commercial interests in any commercial products presented. U.S.
David L. Suarez D.V.M., PhD. A.C.V.M. Southeast Poultry Research Laboratory United States National Poultry Research Center The author has no commercial interests in any commercial products presented. U.S.
The first and only fully-automated, multiplexed solution for Measles, Mumps, Rubella and Varicella-zoster virus antibody testing
 Bio-Rad Laboratories BioPlex 2200 System BioPlex 2200 MMRV IgG Kit The first and only fully-automated, multiplexed solution for Measles, Mumps, Rubella and Varicella-zoster virus antibody testing Bio-Rad
Bio-Rad Laboratories BioPlex 2200 System BioPlex 2200 MMRV IgG Kit The first and only fully-automated, multiplexed solution for Measles, Mumps, Rubella and Varicella-zoster virus antibody testing Bio-Rad
OIE Situation Report for Avian Influenza
 OIE Situation Report for Avian Influenza Latest update: 24/04/2017 This report presents an overview of current disease events reported to the OIE by its Members. The objective is to describe what is happening
OIE Situation Report for Avian Influenza Latest update: 24/04/2017 This report presents an overview of current disease events reported to the OIE by its Members. The objective is to describe what is happening
Rotavirus vaccines: Issues not fully addressed in efficacy trials
 Rotavirus vaccines: Issues not fully addressed in efficacy trials TM Umesh D. Parashar Lead, Viral Gastroenteritis Team CDC, Atlanta, USA uparashar@cdc.gov 1 Two New Rotavirus Vaccines Licensed in 2006
Rotavirus vaccines: Issues not fully addressed in efficacy trials TM Umesh D. Parashar Lead, Viral Gastroenteritis Team CDC, Atlanta, USA uparashar@cdc.gov 1 Two New Rotavirus Vaccines Licensed in 2006
HIV in Women: A Global View of the HIV Epidemic
 Training Course in Reproductive Health / Sexual Health Research Geneva 2006 HIV in Women: A Global View of the HIV Epidemic George Schmid, M.D., M.Sc. Department of HIV/AIDS World Health Organization Geneva,
Training Course in Reproductive Health / Sexual Health Research Geneva 2006 HIV in Women: A Global View of the HIV Epidemic George Schmid, M.D., M.Sc. Department of HIV/AIDS World Health Organization Geneva,
An Introduction to Access to Diagnostics. 14:00-14:45 BST 17 June
 An Introduction to Access to Diagnostics 14:00-14:45 BST 17 June Chair: Dr Greg Martin, Specialist Registrar, Public Health Medicine and Editor-in- Chief of Globalization and Health @drgregmartin Speakers:
An Introduction to Access to Diagnostics 14:00-14:45 BST 17 June Chair: Dr Greg Martin, Specialist Registrar, Public Health Medicine and Editor-in- Chief of Globalization and Health @drgregmartin Speakers:
5. Over the last ten years, the proportion of HIV-infected persons who are women has: a. Increased b. Decreased c. Remained about the same 1
 Epidemiology 227 April 24, 2009 MID-TERM EXAMINATION Select the best answer for the multiple choice questions. There are 60 questions and 9 pages on the examination. Each question will count one point.
Epidemiology 227 April 24, 2009 MID-TERM EXAMINATION Select the best answer for the multiple choice questions. There are 60 questions and 9 pages on the examination. Each question will count one point.
HIV-1 AccuVert TM Seroconversion Panel
 PACKAGE INSERT PRB954 (0600-0238) INTENDED USE The is intended for use by diagnostic manufacturers, researchers, and clinical laboratories to develop, evaluate, or troubleshoot test methods. Characterized
PACKAGE INSERT PRB954 (0600-0238) INTENDED USE The is intended for use by diagnostic manufacturers, researchers, and clinical laboratories to develop, evaluate, or troubleshoot test methods. Characterized
AIDS in Africa. An Update. Basil Reekie
 AIDS in Africa An Update Basil Reekie Contents General Statistics The trend of HIV in Africa Ugandan experience UNAIDS 2006 Latest African Statistics by Country HIV Intervention Light at the end of the
AIDS in Africa An Update Basil Reekie Contents General Statistics The trend of HIV in Africa Ugandan experience UNAIDS 2006 Latest African Statistics by Country HIV Intervention Light at the end of the
HEV Assay Development Update
 HEV Assay Development Update Jeffrey M. Linnen, Ph.D. Senior Director, Research & Development Hologic Gen-Probe, San Diego, California USA IPFA/PEI 20th International Workshop on Surveillance and Screening
HEV Assay Development Update Jeffrey M. Linnen, Ph.D. Senior Director, Research & Development Hologic Gen-Probe, San Diego, California USA IPFA/PEI 20th International Workshop on Surveillance and Screening
Update from ISS on following EQA studies: ISS EQA Tri-NAT (HCV, HIV and HBV) 2012 ISS EQA NAT HBV 2012 ISS EQA NAT WNV 2012
 Update from ISS on following EQA studies: ISS EQA Tri-NAT (HCV, HIV and HBV) 2012 ISS EQA NAT HBV 2012 ISS EQA NAT WNV 2012 SOGAT XXIV Ljubljana, 8-9 May 2013 2012 ISS EQAs for NAT ISS EQA Tri-NAT (HCV,
Update from ISS on following EQA studies: ISS EQA Tri-NAT (HCV, HIV and HBV) 2012 ISS EQA NAT HBV 2012 ISS EQA NAT WNV 2012 SOGAT XXIV Ljubljana, 8-9 May 2013 2012 ISS EQAs for NAT ISS EQA Tri-NAT (HCV,
