USE OF LATEX AGGLUTINATION TEST TO DETERMINE RABIES ANTIBODIES IN PRODUCTION OF RABIES ANTISERA IN HORSES
|
|
|
- Amanda Berry
- 5 years ago
- Views:
Transcription
1 LATEX AGGLUTINATION TEST FOR DETERMINING RABIES ANTIBODIES IN HORSE SERA USE OF LATEX AGGLUTINATION TEST TO DETERMINE RABIES ANTIBODIES IN PRODUCTION OF RABIES ANTISERA IN HORSES Wachiraporn Saengseesom, Songsri Kasempimolporn, Surasak Akesowan, Suraseha Ouisuwan and Visith Sitprija Queen Saovabha Memorial Institute (WHO Collaborating Center for Research on Rabies), Thai Red Cross Society, Bangkok, Thailand Abstract. A therapeutic anti-rabies immunoglobulin for human use has been produced mainly in horses. The presently available seroneutralization test, the rapid fluorescent focus inhibition test (RFFIT), is laborious and rather difficult to carry out in horse farms. This study was undertaken to develop a simple latex agglutination test (LAT) for determining rabies antibodies in horse sera. LAT was validated by testing a total of 468 horse serum samples characterized by RFFIT. Of these, 253 of 260 samples with antibody titers of less than 100 IU/ml had agglutination score of 1+, whereas 174 of 208 samples with antibody titers equal to or greater than 100 IU/ml had agglutination scores of Results of LAT correlated with those of RFFIT (r = 0.87, p < ). LAT has the advantages of being rapid, simple to perform, easy to interpret, and applicable as an on-site testing tool for the estimation of rabies antibodies in horses. Key words: rabies antibodies, LAT, RFFIT, horse sera INTRODUCTION Correspondence: Dr Songsri Kasempimolporn, Queen Saovabha Memorial Institute, Thai Red Cross Society, 1871 Rama IV Road, Bangkok 10330, Thailand. Tel.: 66 (0) ; fax: 66 (0) songsri_k@redcross.or.th Rabies remains a public health problem in Asia. In most countries where human rabies is reported, the public health sector bears the majority of the costs related by providing services for bite treatment and the delivery of post exposure prophylaxis (PEP) to protect the victims of a suspect rabid bite (Kasempimolporn et al, 2008). Human or equine immunoglobulins, administered to severely rabiesexposed patients at the onset of PEP represent a safety net for the patient until vaccine-induced endogenous antibodies are formed (WHO, 1997). The Queen Saovabha Memorial Institute (QSMI) is the sole manufacturer of equine rabies immunoglobulin (ERIG) in Bangkok, Thailand. Purified ERIG is processed from plasma of horses, which are immunized with rabies vaccine as previously described (Luekrajang et al, 1996) and plasma is collected via plasmapheresis (Feige et al, 2003). The horse farm of QSMI is located at Hua Hin, some 200 km south of Bangkok. The serum neutralizing test is used as the bioassay method of choice for measuring the acceptability of horse antisera. The measurement of anti- Vol 41 No. 6 November
2 SOUTHEAST ASIAN J TROP MED PUBLIC HEALTH body levels in plasma and the production of ERIG are carried out at a manufacturing plant in Bangkok. Difficulty has been encountered in producing high titer antisera in horses as variations in immune response between horses may occur. It is desirable for the veterinarians responsible for the production of horse antisera to evaluate the levels of antibodies of the horses simply and quickly and to determine whether additional (booster) doses of vaccine are required. Immunized horses are not routinely monitored for seroconversion due to lack of laboratory facilities. The veterinarians at the horse farm usually submit blood samples for serological testing to the laboratory at QSMI and exsanguination of the horses is performed after the test blood samples show acceptable titers. The procedures favored by most laboratories for rabies antibody determination are a tissue culture neutralization test using live rabies virus employing an immunofluorescent-antibody staining technique, such as the rapid fluorescent focus inhibition test (RFFIT) (Smith et al, 1973), and fluorescent antibody virus neutralization test (Chiquet et al, 1998). These methods are the reference techniques for the World Health Organization (WHO, 2005) and the World Organization for Animal Health (OIE, 2004). The assays require relatively sophisticated laboratory facilities including the production of rabies virus and cell cultures, which are complicated, time consuming and costly. To overcome this disadvantage, an enzyme-linked immunosorbent assay (ELISA) has been described to quantify the level of rabies antibodies (Nicholson and Prestage, 1982; Kavaklova et al, 1984; Grassi et al, 1989). ELISA is less time consuming and less expensive than RFFIT, but even so it still cannot be carried out by the veterinarian at QSMI horse farm as facilities or resources to perform such tests are not available. Recently, a new commercial immunoenzymatic test that utilizes protein A linked with horseradish peroxidase for detecting and quantifying rabies antibodies in the sera of humans and domestic and wild carnivores has been introduced (Servat et al, 2007). Such a test kit may not be suitable for all animal sera, particularly those of herbivores (Mebatsion et al, 1989). The utility of a latex agglutination test (LAT) for the determination of rabies antibodies after human vaccination has been reported (Perrin et al, 1988; Madhusudana and Saraswati, 2003). The LAT is simple to perform and needs minimum laboratory equipment. In this study, we report the results obtained with horse sera tested with LAT and RFFIT tests. MATERIALS AND METHODS Test specimens A total of 468 horse immune serum samples were evaluated in this study. Each test serum was collected at various days after immunization with rabies vaccine. These specimens were assayed by RFFIT (Smith et al, 1973) at QSMI and were examined by LAT by examiners without previous knowledge of the results. Thirty nonimmune horse sera were included as controls. RFFIT Serum samples were heated at 56ºC for 30 minutes before testing in order to inactivate complement. Dilutions of test serum in cell culture medium were mixed with a constant amount of rabies virus in a 96-well microplate. Serum and virus were then incubated at 37ºC for 90 minutes. A concentration of 5x10 5 BHK-21/13S 1388 Vol 41 No. 6 November 2010
3 LATEX AGGLUTINATION TEST FOR DETERMINING RABIES ANTIBODIES IN HORSE SERA cells/ml was added to each well, and incubated at 37ºC for another 24 hours. The cells were then rinsed, fixed in acetone, and stained with fluorescein-conjugated rabbit anti-rabies virus globulin (TRC, Thai Red Cross Society, Thailand). A serum is considered to have antibody when 50% or more of the cells are protected from the virus. The titer of antibody in the test serum was obtained by comparison with reference serum standard (The State Serum Institute, Copenhagen, Denmark). Titers were expressed as international units per ml (IU/ml). Samples with antibody levels <100, and >200 IU/ml were grouped and subjected to LAT examination. Table 1 Comparison between antibody determination in horse immune sera using LAT and RFFIT. LAT Neutralizing antibody titer (IU/ml) < > LAT Sensitized polystyrene blue latex beads (diameter, 0.8 µm; Sigma, St Louis, M0) were prepared as previously described (Perrin et al, 1988). One ml of 10% latex beads were washed 3 times with 5 ml of 0.05 M bicarbonate buffer, ph 9.6. Beads were centrifuged at 5,000g for 30 minutes. The latex beads were suspended in 3 ml of bicarbonate buffer together with 1.75 ml of a 1: 10 dilution of rabies virus vaccine (Verorab, Merieux, France). The mixture was shaken at 37ºC for 3 hours and at 4ºC overnight. The beads were suspended in 5 ml of carbonate buffer containing 5% trehalose and 0.3% bovine serum albumin. The beads were incubated at 37ºC for 30 minutes and then washed twice with phosphate-buffered saline (PBS), ph 7.4. The sensitized latex beads were resuspended at a 3% concentration in PBS and kept at 4ºC until used. The assay was performed by spreading 25 µl aliquot of sensitized latex beads onto a double-concave glass slide, adding 25 µl aliquot of the serum to be tested, and mixing for 5 to 15 minutes at room temperature. A positive reaction for antibodies to rabies virus was agglutination visible in bright light, indicated by the formation of fine granular particles. The degree of agglutination was scored as 4+, 3+, 2+ and 1+ depending on size of agglutinated particles. A negative reaction was no agglutination, giving the serum and latex suspension with a milky, smooth consistency. Positive and negative control sera were tested with each assay. RESULTS The performance of a LAT developed in our laboratory was compared to that of the standard serological technique. Horse sera which contain rabies antibodies are capable of agglutinating rabies virus antigen-sensitized beads. The agglutination was visible with immune sera which had been characterized previously by RFFIT and no agglutination was observed with non-immune sera (data not shown), confirming the specificity of agglutination assay. Among 468 test immune sera, 260 contained virus neutralizing antibody titers of less than 100 IU/ml, 134 had titers between IU/ml and the remaining samples had a titer greater than 200 IU/ml Vol 41 No. 6 November
4 SOUTHEAST ASIAN J TROP MED PUBLIC HEALTH Titer(IU/ml) r = y = x LAT Fig 1 Correlation between LAT results and the virus-neutralizing antibody titers. Antibody titers were obtained using RFFIT. (Table 1). The agglutination observed were graded from 1+ to 4+. A score of 1+ corresponded to titers less than 100 IU/ml, 2+ corresponded to titers equivalent IU/ml, and 3+ to 4+ corresponded to titers greater than 200 IU/ml. The LAT and RFFIT outcomes correlated significantly, with a correlation coefficient of 0.87, p < (Fig 1). Determination of rabies antibodies in well-characterized seropositive serum samples by LAT was positive over a wide range of serum titers, ranging from <100 to >100 IU/ml. Interpretation of positive LAT serum samples became easier at titers >200 IU/ml. DISCUSSION Horses are found to be suitable for the economical large-volume production of therapeutic ERIG for human use. ERIG is produced by immunizing horses with a cell culture-derived rabies vaccine. Plasmapheresis in immunized horses is performed and crude plasma is submitted to the manufacturing plant if acceptable serologic titer is demonstrated. Antibody titer of less than 100 IU/ml is considered too low for use. However, we have found that there are variations in antibody levels between individual horses. The immune response variation among horses may be due to differences in horse immunogenic background, nutritional status and general health (Redwan et al, 2009). For this reason, on-site testing for demonstration of adequate seroconversion following rabies vaccination is necessary. The presently available test, such as RFFIT, is laborious and time consuming. Agglutination reactions observed with particular antigens can be extended to a wide variety of soluble antigens by attaching them to the surface of latex beads. The utility of this procedure has been demonstrated for detection of several viral antibodies, such as rabies virus (Perrin et al, 1988), HIV (Quinn et al, 1988), varicella virus (Steinberg and Gershon, 1991) and avian influenza virus (Horie et al, 2009). The LAT developed in this study is a simple procedure that is easy and rapid to perform. We used commercial cell-culture rabies vaccine of human use as a coating antigen. The antigen can be prepared easily, as it is not necessary to purify the viral proteins or virions. This test is proposed for determining the antibody status of immunized horses and for optimizing immunization schedules in an effort to reduce a waste of low-titer raw plasma. We compared LAT with standard RFFIT. Using RFFIT, all sera had titers ranging from 3 to 480 IU/ml (data not shown). When the samples were grouped according to their titers and tested by LAT, there is good correlation in degree (magnitude) of positivity, although it is not known whether results of LAT represent the detection of neutralizing antibodies. However, in this study discrepant results were obtained for 41 (9%) serum samples, with some samples showing high antibody 1390 Vol 41 No. 6 November 2010
5 LATEX AGGLUTINATION TEST FOR DETERMINING RABIES ANTIBODIES IN HORSE SERA titers exhibiting low degree of agglutination, and vice versa. These discrepant results may be due to a different degree of accuracy and/or diversity among the two test systems. In addition, these may exist components in serum that affect the specific agglutination of sensitized latex. In previous studies, LAT developed for detecting rabies antibodies in human sera showed 4+ agglutination with serum samples having a titer greater than 8 IU/ ml (Perrin et al, 1988; Madhusudana and Saraswati, 2003), while 2-4+ agglutination reactions in this study occurred widely in tested cases with titers greater than 100 IU/ ml. One explanation for such discrepancies could reside with the antigen coated on the latex beads. For human sera purified whole virus or purified rabies glycoprotein was used, whereas we used commercial human rabies vaccine preparation. In addition, different sources of serum might also be a cause. An accurate definition of antibody titers should be expressed in terms of the neutralizing titration. LAT is not proposed for determining antibody titration but rather for the rapid screening of rabies antibodies and its sensitivity seems to be adequate. If the results of LAT are directly correlated with RFFIT and if the degree of agglutination correlates with the level of rabies neutralizing antibody, then the presence of moderate agglutination of LAT rabies antibodies should equate with acceptable neutralizing antibody titers. Sera with agglutination scores greater than 2+ were considered as corresponding to neutralizing antibody higher than 100 IU/ml. The simplicity of LAT permits its performance under field conditions with a capacity for testing many sera in the same run. The test can be performed at a horse farm for routine use if sensitized beads are supplied from a central laboratory. Reagents were stable for 2-3 months at 4ºC, after which they lost stability (data not shown). LAT gives a faster result (within minutes) than RFFIT (2 days) or other laboratory tests which may not be available on a daily basis. LAT does not require the use of live virus and so does not require virus containment facilities or any sophisticated equipment. Although LAT is subjective in interpretation and performing the assay requires some degree of skill, inter-reader variability between skilled persons was low. When a panel of samples were tested by two skilled persons, the LAT results showed a relatively high degree of agreement (95%) (data not shown). In summary, data presented show that LAT can be used for in-process control to continuously monitor antibody response of horses at various stages of rabies immunization. It is reasonably robust and can be used on-site in horse farms. REFERENCES Chiquet F, Aubert M, Sagne L. Development of a fluorescent antibody virus neutralization test (FAVN test) for the quantitation of rabies-neutralizing antibody. J Immunol Methods 1998; 212: Feige K, Ehrat FB, Kastner SB, Schwarzwald CC. Automated plasmapheresis compared with other plasma collection methods in the horse. J Vet Med Physiol Pathol Clin Med 2003; 50: Grassi M, Wandeler I, Peterhaus E. Enzymelinked immunosorbent assay for determination of antibodies to the envelope glycoprotein of rabies virus. J Clin Microbiol 1989; 27: Horie M, Ogawa H, Yamada K, et al. A latex agglutination test using a recombinant nucleoprotein for detection of antibodies against avian influenza virus. J Virol Vol 41 No. 6 November
6 SOUTHEAST ASIAN J TROP MED PUBLIC HEALTH Methods 2009; 161: Kasempimolporn S, Jitapunkul S, Sitprija V. Moving towards the elimination of rabies in Thailand. J Med Assoc Thai 2008; 91: Kavaklova L, Eskenazy M, Gancheva T, Vacheva V. Enzyme-linked immunosorbent assay for detection of rabies antibodies in human sera. Acta Virol 1984; 28: Luekrajang T, Wangsai J, Phanuphak P. Production of antirabies serum of equine origin. In: Meslin FX, Kaplan MM, Koprowski H, eds. WHO laboratory techniques in rabies. Geneva: WHO, 1996: Madhusudana SN, Saraswati S. Development and evaluation of a latex agglutination test for rabies antibodies. J Clin Virol 2003; 27: Mebatsion T, Frost JW, Krauss H. Enzymelinked immunosorbent assay (ELISA) using staphylococcal protein a for the measurement of rabies antibodies in various species. J Vet Med B 1989; 36: Nicholson KG, Prestage H. Enzyme-linked immunosorbent assay: a rapid reproducible test for the measurement of rabies antibody. J Med Virol 1982; 9: World Organization for Animal Health (OIE). Rabies. In: Biological Standards Commission. Manual of diagnostic tests and vaccines for terrestrial animals. 5 th ed. Paris: OIE, 2004: Perrin P, Versmisse P, Sureau P. A rabies agglutination test (RAT) for rabies antibody detection. J Biol Stand 1988; 16: Quinn TC, Riggin CH, Kline RL, et al. Rapid latex agglutination assay using recombinant envelope polypeptide for the detection of antibody to the HIV. JAMA 1988; 260: Redwan el-em, Fahmy A, EL Hanafy A, Abd EL-Baky N, Sallam SM. Ovine anti-rabies antibody production and evaluation. Comp Immunol Microbiol Infect Dis 2009; 32: Servat A, Feyssaguet M, Blanchard I, et al. A quantitative indirect ELISA to monitor the effectiveness of rabies vaccination in domestic and wild carnivores. J Immunol Methods 2007; 318: Smith JS, Yager PA, Bear GM. A rapid reproducible test for determining rabies neutralizing antibody. Bull World Health Organ 1973; 48: Steinberg SP, Gershon AA. Measurement of antibodies to varicella virus by using latex agglutination test. J Clin Microbiol 1991; 29: World Health Organization (WHO). Recommendations on rabies post-exposure treatment and the correct technique of intradermal immunization against rabies. Geneva: WHO, World Health Organization (WHO). WHO Expert Consultation on Rabies, first report. World Health Organ Tech Rep Ser 2005; 931: Vol 41 No. 6 November 2010
REAL-TIME PCR ANALYSIS OF DOG CEREBROSPINAL FLUID AND SALIVA SAMPLES FOR ANTE-MORTEM DIAGNOSIS OF RABIES
 REAL-TIME PCR ANALYSIS OF DOG CEREBROSPINAL FLUID AND SALIVA SAMPLES FOR ANTE-MORTEM DIAGNOSIS OF RABIES Wachiraporn Saengseesom, Channarong Mitmoonpitak, Songsri Kasempimolporn and Visith Sitprija Queen
REAL-TIME PCR ANALYSIS OF DOG CEREBROSPINAL FLUID AND SALIVA SAMPLES FOR ANTE-MORTEM DIAGNOSIS OF RABIES Wachiraporn Saengseesom, Channarong Mitmoonpitak, Songsri Kasempimolporn and Visith Sitprija Queen
Rubella Latex Agglutination Test
 Rubella Latex Agglutination Test Cat. No.:DLAT1088 Pkg.Size:30T Intended use The Rubella Latex Agglutination Test is a rapid latex particle agglutination test for the qualitative and semi-quantitative
Rubella Latex Agglutination Test Cat. No.:DLAT1088 Pkg.Size:30T Intended use The Rubella Latex Agglutination Test is a rapid latex particle agglutination test for the qualitative and semi-quantitative
IVD information *Droppers for the sensitized and control cells. Not for use other than dispensing the sensitized and control cells.
 In Vitro Diagnostic Reagent Instruction Manual of Diagnostic Reagent for Determination of anti-hbs Thoroughly read this instruction manual before use of this kit Background of the development and features
In Vitro Diagnostic Reagent Instruction Manual of Diagnostic Reagent for Determination of anti-hbs Thoroughly read this instruction manual before use of this kit Background of the development and features
Identification of Microbes Lecture: 12
 Diagnostic Microbiology Identification of Microbes Lecture: 12 Electron Microscopy 106 virus particles per ml required for visualization, 50,000-60,000 magnification normally used. Viruses may be detected
Diagnostic Microbiology Identification of Microbes Lecture: 12 Electron Microscopy 106 virus particles per ml required for visualization, 50,000-60,000 magnification normally used. Viruses may be detected
CHAPTER 4 IMMUNOLOGICAL TECHNIQUES
 CHAPTER 4 IMMUNOLOGICAL TECHNIQUES Nitroblue Tetrazolium Chloride (NBT) Reduction test NBT reduction test was evaluated by employing the method described by Hudson and Hay,1989 based upon principle that
CHAPTER 4 IMMUNOLOGICAL TECHNIQUES Nitroblue Tetrazolium Chloride (NBT) Reduction test NBT reduction test was evaluated by employing the method described by Hudson and Hay,1989 based upon principle that
See external label 2 C-8 C = C-REACTIVE PROTEIN (CRP) LATEX SLIDE TEST
 CORTEZ DIAGNOSTICS, INC. 21250 Califa Street, Suite 102 and 116, Woodland Hills, CA 91367 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com
CORTEZ DIAGNOSTICS, INC. 21250 Califa Street, Suite 102 and 116, Woodland Hills, CA 91367 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com
Issued by: LABORATORY MANAGER Original Date: March 20, 2000 Approved by: Laboratory Director Revision Date: July 26, 2000 CRYPTOCOCCAL ANTIGEN
 Policy # MI\TECH\11\v01 Page 1 of 6 Issued by: LABORATORY MANAGER Original Date: March 20, 2000 Approved by: Laboratory Director Revision Date: July 26, 2000 CRYPTOCOCCAL ANTIGEN Latex particles coated
Policy # MI\TECH\11\v01 Page 1 of 6 Issued by: LABORATORY MANAGER Original Date: March 20, 2000 Approved by: Laboratory Director Revision Date: July 26, 2000 CRYPTOCOCCAL ANTIGEN Latex particles coated
Cost comparison of rabies pre-exposure vaccination with post-exposure treatment in Thai children
 Vaccine 24 (2006) 1478 1482 Cost comparison of rabies pre-exposure vaccination with post-exposure treatment in Thai children Pannipa Chulasugandha a,, Pakamatz Khawplod a, Piyalamporn Havanond b, Henry
Vaccine 24 (2006) 1478 1482 Cost comparison of rabies pre-exposure vaccination with post-exposure treatment in Thai children Pannipa Chulasugandha a,, Pakamatz Khawplod a, Piyalamporn Havanond b, Henry
Economical intradermal rabies vaccine application By F.X Meslin and M.Warrell at the occasion of the international seminar on "intradermal
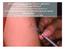 Economical intradermal rabies vaccine application By F.X Meslin and M.Warrell at the occasion of the international seminar on "intradermal immunization: an alternative route for vaccine administration"
Economical intradermal rabies vaccine application By F.X Meslin and M.Warrell at the occasion of the international seminar on "intradermal immunization: an alternative route for vaccine administration"
WHO Prequalification of In Vitro Diagnostics Programme PUBLIC REPORT. Product: Murex HIV Ag/Ab Combination Number: PQDx
 WHO Prequalification of In Vitro Diagnostics Programme PUBLIC REPORT Product: Murex HIV Ag/Ab Combination Number: PQDx 0144-043-00 Abstract Murex HIV Ag/Ab Combination with product codes 7G79-09 (GE41,
WHO Prequalification of In Vitro Diagnostics Programme PUBLIC REPORT Product: Murex HIV Ag/Ab Combination Number: PQDx 0144-043-00 Abstract Murex HIV Ag/Ab Combination with product codes 7G79-09 (GE41,
Trichinella Cat #
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
Mycoplasma pneumoniae IgG ELISA Kit
 Mycoplasma pneumoniae IgG ELISA Kit Catalog Number KA2260 96 assays Version: 01 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
Mycoplasma pneumoniae IgG ELISA Kit Catalog Number KA2260 96 assays Version: 01 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
Herpes Simplex Virus 2 IgG HSV 2 IgG
 DIAGNOSTIC AUTOMATION, INC. 21250 Califa Street, Suite 102 and 116, Woodland Hills, CA 91367 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com
DIAGNOSTIC AUTOMATION, INC. 21250 Califa Street, Suite 102 and 116, Woodland Hills, CA 91367 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com
Detection of neuraminidase-inhibiting antibodies for measurement of Influenza vaccine immunogenicity
 Borgis New Med 2015; 19(4): 147-155 DOI: 10.5604/14270994.1191796 Detection of neuraminidase-inhibiting antibodies for measurement of Influenza vaccine immunogenicity *Mónika Rózsa 1, István Jankovics
Borgis New Med 2015; 19(4): 147-155 DOI: 10.5604/14270994.1191796 Detection of neuraminidase-inhibiting antibodies for measurement of Influenza vaccine immunogenicity *Mónika Rózsa 1, István Jankovics
NF-κB p65 (Phospho-Thr254)
 Assay Biotechnology Company www.assaybiotech.com Tel: 1-877-883-7988 Fax: 1-877-610-9758 NF-κB p65 (Phospho-Thr254) Colorimetric Cell-Based ELISA Kit Catalog #: OKAG02015 Please read the provided manual
Assay Biotechnology Company www.assaybiotech.com Tel: 1-877-883-7988 Fax: 1-877-610-9758 NF-κB p65 (Phospho-Thr254) Colorimetric Cell-Based ELISA Kit Catalog #: OKAG02015 Please read the provided manual
See external label 2 C-8 C 96 tests CHEMILUMINESCENCE. CMV IgG. Cat # Step (20-25 C Room temp.) Volume
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
M I C R O B I O L O G Y
 ninth edition TORTORA FUNKE CASE M I C R O B I O L O G Y a n i n t r o d u c t i o n 18 Practical Applications of Immunology PowerPoint Lecture Slide Presentation prepared by Christine L. Case Vaccine
ninth edition TORTORA FUNKE CASE M I C R O B I O L O G Y a n i n t r o d u c t i o n 18 Practical Applications of Immunology PowerPoint Lecture Slide Presentation prepared by Christine L. Case Vaccine
E. Histolytica IgG (Amebiasis)
 DIAGNOSTIC AUTOMATION, INC. 21250 Califa Street, Suite 102 and 116, Woodland hills, CA 91367 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com
DIAGNOSTIC AUTOMATION, INC. 21250 Califa Street, Suite 102 and 116, Woodland hills, CA 91367 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com
Secondary fluorescent staining of virus antigens by rheumatoid factor and fluorescein-conjugated anti-lgm
 Ann. rheum. Dis. (1973), 32, 53 Secondary fluorescent staining of virus antigens by rheumatoid factor and fluorescein-conjugated anti-lgm P. V. SHIRODARIA, K. B. FRASER, AND F. STANFORD From the Department
Ann. rheum. Dis. (1973), 32, 53 Secondary fluorescent staining of virus antigens by rheumatoid factor and fluorescein-conjugated anti-lgm P. V. SHIRODARIA, K. B. FRASER, AND F. STANFORD From the Department
Gladstone Institutes, University of California (UCSF), San Francisco, USA
 Fluorescence-linked Antigen Quantification (FLAQ) Assay for Fast Quantification of HIV-1 p24 Gag Marianne Gesner, Mekhala Maiti, Robert Grant and Marielle Cavrois * Gladstone Institutes, University of
Fluorescence-linked Antigen Quantification (FLAQ) Assay for Fast Quantification of HIV-1 p24 Gag Marianne Gesner, Mekhala Maiti, Robert Grant and Marielle Cavrois * Gladstone Institutes, University of
SUMMARY OF PRODUCT CHARACTERISTICS
 SUMMARY OF PRODUCT CHARACTERISTICS 1. Name of the product: 1.1 Product name: EQUIRAB Rabies Antiserum - Equine 1.2 Strength: 1000 I.U. / 5 ml 1.3 Pharmaceutical Dosage form: Injection 2. Qualitative and
SUMMARY OF PRODUCT CHARACTERISTICS 1. Name of the product: 1.1 Product name: EQUIRAB Rabies Antiserum - Equine 1.2 Strength: 1000 I.U. / 5 ml 1.3 Pharmaceutical Dosage form: Injection 2. Qualitative and
Cryptococcal Antigen Latex Agglutination Test
 Cryptococcal Antigen Latex Agglutination Test Cat. No.:DLAT1109 Pkg.Size:100T Intended use The Cryptococcal Antigen Latex Agglutination Test is a simple and rapid latex agglutination test for the qualitative
Cryptococcal Antigen Latex Agglutination Test Cat. No.:DLAT1109 Pkg.Size:100T Intended use The Cryptococcal Antigen Latex Agglutination Test is a simple and rapid latex agglutination test for the qualitative
by Jean Lang, a Emmanuel Feroldi, a and Nguyen Cong Vien b a Sanofi Pasteur, Marcy L etoile, France
 Pre-exposure Purified Vero Cell Rabies Vaccine and Concomitant Routine Childhood Vaccinations: 5-year Post-vaccination Follow-up Study of an Infant Cohort in Vietnam by Jean Lang, a Emmanuel Feroldi, a
Pre-exposure Purified Vero Cell Rabies Vaccine and Concomitant Routine Childhood Vaccinations: 5-year Post-vaccination Follow-up Study of an Infant Cohort in Vietnam by Jean Lang, a Emmanuel Feroldi, a
HIV-1 p24 ELISA Kit. purified polyclonal antibody raised against the full length recombinant p24 is used.
 HIV-1 p24 ELISA Kit 80-001 1 kit 96 assays This kit can measure the amount of HIV-1 Gag p24 antigen in cell culture medium handily by a sandwich ELISA (Enzyme Linked Immunosorbent Assay) method. p24 antigen
HIV-1 p24 ELISA Kit 80-001 1 kit 96 assays This kit can measure the amount of HIV-1 Gag p24 antigen in cell culture medium handily by a sandwich ELISA (Enzyme Linked Immunosorbent Assay) method. p24 antigen
LONG-TERM PROTECTIVE RABIES ANTIBODIES IN THAI CHILDREN AFTER PRE-EXPOSURE RABIES VACCINATION
 LONG-TERM PROTECTIVE RABIES ANTIBODIES IN THAI CHILDREN AFTER PRE-EXPOSURE RABIES VACCINATION Supawat Chatchen, Shakil Ibrahim, Pataporn Wisetsing and Kriengsak Limkittikul Department of Tropical Pediatrics,
LONG-TERM PROTECTIVE RABIES ANTIBODIES IN THAI CHILDREN AFTER PRE-EXPOSURE RABIES VACCINATION Supawat Chatchen, Shakil Ibrahim, Pataporn Wisetsing and Kriengsak Limkittikul Department of Tropical Pediatrics,
SEROLOGICAL DIAGNOSIS OF VIRAL INFECTIONS:
 SEROLOGICAL DIAGNOSIS OF VIRAL INFECTIONS: POSSIBILITIES OF SEROLOGICAL DIAGNOSIS TYPES OF SEROLOGICAL REACTIONS SEROLOGICAL REACTIONS Ag-Ab reactions used for the detection of unknown Ag or Ab, in vitro
SEROLOGICAL DIAGNOSIS OF VIRAL INFECTIONS: POSSIBILITIES OF SEROLOGICAL DIAGNOSIS TYPES OF SEROLOGICAL REACTIONS SEROLOGICAL REACTIONS Ag-Ab reactions used for the detection of unknown Ag or Ab, in vitro
H.pylori IgA Cat #
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
See external label 96 tests HSV 2 IgA. Cat #
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
E. Histolytica IgG ELISA Kit
 E. Histolytica IgG ELISA Kit Catalog Number KA3193 96 assays Version: 01 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of
E. Histolytica IgG ELISA Kit Catalog Number KA3193 96 assays Version: 01 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of
CLINICAL RELEVANCE. D. A. Grosenbaugh, DVM, PhD a C. S. Backus, DVM, PhD b K. Karaca, DVM, PhD a J. M. Minke, DVM, PhD c R. M. Nordgren, DVM, PhD a
 The Anamnestic Serologic Response to Vaccination with a Canarypox Virus Vectored Recombinant West Nile Virus (WNV) Vaccine in Horses Previously Vaccinated with an Inactivated WNV Vaccine* D. A. Grosenbaugh,
The Anamnestic Serologic Response to Vaccination with a Canarypox Virus Vectored Recombinant West Nile Virus (WNV) Vaccine in Horses Previously Vaccinated with an Inactivated WNV Vaccine* D. A. Grosenbaugh,
Human Allergen Specific IgE ELISA Kits
 Intended Use Human Allergen Specific IgE ELISA Kits These kits are in vitro assays for the qualitative or quantitative detection of allergen-specific IgE antibodies in human serum. They are intended for
Intended Use Human Allergen Specific IgE ELISA Kits These kits are in vitro assays for the qualitative or quantitative detection of allergen-specific IgE antibodies in human serum. They are intended for
Rubella virus IgG ELISA Kit
 Rubella virus IgG ELISA Kit Catalog Number KA0223 96 assays Version: 03 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of
Rubella virus IgG ELISA Kit Catalog Number KA0223 96 assays Version: 03 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of
Laboratory Procedure Handout RHEUMATOID FACTORS
 KING ABDULAZIA UNIVERSITY FACULTY OF APPLIED MEDICAL SCIENCES DEPARTEMENT OF LABORATORY MEDICAL TECHNOLOGY Laboratory Procedure Handout RHEUMATOID FACTORS RF Latex agglutination for detection of RF INTRODUCTION
KING ABDULAZIA UNIVERSITY FACULTY OF APPLIED MEDICAL SCIENCES DEPARTEMENT OF LABORATORY MEDICAL TECHNOLOGY Laboratory Procedure Handout RHEUMATOID FACTORS RF Latex agglutination for detection of RF INTRODUCTION
VIRAL TITER COUNTS. The best methods of measuring infectious lentiviral titer
 VIRAL TITER COUNTS The best methods of measuring infectious lentiviral titer FLUORESCENCE CYCLES qpcr of Viral RNA SUMMARY Viral vectors are now routinely used for gene transduction in a wide variety of
VIRAL TITER COUNTS The best methods of measuring infectious lentiviral titer FLUORESCENCE CYCLES qpcr of Viral RNA SUMMARY Viral vectors are now routinely used for gene transduction in a wide variety of
Human HBcAb IgM ELISA kit
 Human HBcAb IgM ELISA kit Catalog number: NR-R10163 (96 wells) The kit is designed to qualitatively detect HBcAb IgM in human serum or plasma. FOR RESEARCH USE ONLY. NOT FOR DIAGNOSTIC OR THERAPEUTIC PURPOSES
Human HBcAb IgM ELISA kit Catalog number: NR-R10163 (96 wells) The kit is designed to qualitatively detect HBcAb IgM in human serum or plasma. FOR RESEARCH USE ONLY. NOT FOR DIAGNOSTIC OR THERAPEUTIC PURPOSES
Influenza A IgG ELISA
 Influenza A IgG ELISA For the qualitative determination of IgG-class antibodies against Influenza A virus in human serum or plasma (citrate, heparin). For Research Use Only. Not For Use In Diagnostic Procedures.
Influenza A IgG ELISA For the qualitative determination of IgG-class antibodies against Influenza A virus in human serum or plasma (citrate, heparin). For Research Use Only. Not For Use In Diagnostic Procedures.
Effective Date: 01/07 Supersedes Revision/Date: 01/02 Revision: 01/07 Date Adopted:
 Institution: Procedure No.: Page 1 of 7 Procedure: ASI RUBELLA TEST Doc#: 6004-600 NCCLS Effective Date: 01/07 Supersedes Revision/Date: 01/02 Revision: 01/07 Supersedes Procedure # Prepared by: ASI Date
Institution: Procedure No.: Page 1 of 7 Procedure: ASI RUBELLA TEST Doc#: 6004-600 NCCLS Effective Date: 01/07 Supersedes Revision/Date: 01/02 Revision: 01/07 Supersedes Procedure # Prepared by: ASI Date
Assessing safety and immunogenicity of post-exposure prophylaxis following interchangeability of rabies vaccines in humans
 Research Paper Human Vaccines & Immunotherapeutics 10:5, 1354 1358; May 2014; 2014 Landes Bioscience Assessing safety and immunogenicity of post-exposure prophylaxis following interchangeability of rabies
Research Paper Human Vaccines & Immunotherapeutics 10:5, 1354 1358; May 2014; 2014 Landes Bioscience Assessing safety and immunogenicity of post-exposure prophylaxis following interchangeability of rabies
Procine sphingomyelin ELISA Kit
 Procine sphingomyelin ELISA Kit For the quantitative in vitro determination of Procine sphingomyelin concentrations in serum - plasma - celiac fluid - tissue homogenate - body fluid FOR LABORATORY RESEARCH
Procine sphingomyelin ELISA Kit For the quantitative in vitro determination of Procine sphingomyelin concentrations in serum - plasma - celiac fluid - tissue homogenate - body fluid FOR LABORATORY RESEARCH
LIAISON Measles IgG The fully automated solution for quantitative antibody detection
 LIAISON Measles IgG The fully automated solution for quantitative antibody detection FOR OUTSIDE THE US AND CANADA ONLY LIAISON Measles IgG Number of tests 100 Key assay features Method Assay range Solid
LIAISON Measles IgG The fully automated solution for quantitative antibody detection FOR OUTSIDE THE US AND CANADA ONLY LIAISON Measles IgG Number of tests 100 Key assay features Method Assay range Solid
IgG Antibodies To Toxoplasma Gondii ELISA Kit Protocol
 IgG Antibodies To Toxoplasma Gondii ELISA Kit Protocol (Cat. No.:EK-310-85) 330 Beach Road, Burlingame CA Tel: 650-558-8898 Fax: 650-558-1686 E-Mail: info@phoenixpeptide.com www.phoenixpeptide.com INTENDED
IgG Antibodies To Toxoplasma Gondii ELISA Kit Protocol (Cat. No.:EK-310-85) 330 Beach Road, Burlingame CA Tel: 650-558-8898 Fax: 650-558-1686 E-Mail: info@phoenixpeptide.com www.phoenixpeptide.com INTENDED
IMMUNOGENICITY AND SAFETY OF PURIFIED VERO-CELL RABIES VACCINE IN SEVERELY RABIES-EXPOSED PATIENTS IN CHINA
 IMMUNOGENICITY AND SAFETY OF VERO-CELL RABIES VACCINE IMMUNOGENICITY AND SAFETY OF PURIFIED VERO-CELL RABIES VACCINE IN SEVERELY RABIES-EXPOSED PATIENTS IN CHINA XJ Wang 1, J Lang 3, XR Tao 1, JD Shu 2,
IMMUNOGENICITY AND SAFETY OF VERO-CELL RABIES VACCINE IMMUNOGENICITY AND SAFETY OF PURIFIED VERO-CELL RABIES VACCINE IN SEVERELY RABIES-EXPOSED PATIENTS IN CHINA XJ Wang 1, J Lang 3, XR Tao 1, JD Shu 2,
Validation report. First commercially-available ELISA which allows for the detection of Lumpy Skin Disease antibodies
 Validation report ID Screen Capripox Double Antigen Multi-species Multi-species Double Antigen ELISA for detection of antibodies against capripoxviruses including Lumpy skin disease virus (LSDV), sheeppox
Validation report ID Screen Capripox Double Antigen Multi-species Multi-species Double Antigen ELISA for detection of antibodies against capripoxviruses including Lumpy skin disease virus (LSDV), sheeppox
CEPHIA Consortium for the Evaluation and Performance of HIV Incidence Assays STANDARD OPERATING PROCEDURE
 CEPHIA Consortium for the Evaluation and Performance of HIV Incidence Assays STANDARD OPERATING PROCEDURE TITLE : SOP for (off board dilution) Less Sensitive Modified VITROS Enzyme Immunoassay CEPHIA DOCUMENT
CEPHIA Consortium for the Evaluation and Performance of HIV Incidence Assays STANDARD OPERATING PROCEDURE TITLE : SOP for (off board dilution) Less Sensitive Modified VITROS Enzyme Immunoassay CEPHIA DOCUMENT
H.Pylori IgG Cat # 1503Z
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
Please use only the valid version of the package insert provided with the kit.
 Please use only the valid version of the package insert provided with the kit. Intended Use For the qualitative screening of serum IgG antibodies to Trichinella using an Enzyme Linked Immunoabsorbant Assay
Please use only the valid version of the package insert provided with the kit. Intended Use For the qualitative screening of serum IgG antibodies to Trichinella using an Enzyme Linked Immunoabsorbant Assay
In the Name of God. Talat Mokhtari-Azad Director of National Influenza Center
 In the Name of God Overview of influenza laboratory diagnostic technology: advantages and disadvantages of each test available Talat Mokhtari-Azad Director of National Influenza Center Tehran- Iran 1 1)
In the Name of God Overview of influenza laboratory diagnostic technology: advantages and disadvantages of each test available Talat Mokhtari-Azad Director of National Influenza Center Tehran- Iran 1 1)
Herpes Simplex Virus 2 IgM HSV 2 IgM
 DIAGNOSTIC AUTOMATION, INC. 21250 Califa Street, Suite 102 and 116, Woodland Hills, CA 91367 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com
DIAGNOSTIC AUTOMATION, INC. 21250 Califa Street, Suite 102 and 116, Woodland Hills, CA 91367 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com
Toxocara. Cat # Toxocara ELISA. Enzyme Linked Immunosorbent Assay. ELISA-Indirect; Antigen Coated Plate
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
CONTENTS. STUDY DESIGN METHODS ELISA protocol for quantitation of mite (Dermatophagoides spp.) Der p 1 or Der f 1
 CONTENTS STUDY DESIGN METHODS ELISA protocol for quantitation of mite (Dermatophagoides spp.) Der p 1 or Der f 1 ELISA protocol for mite (Dermatophagoides spp.) Group 2 ALLERGENS RESULTS (SUMMARY) TABLE
CONTENTS STUDY DESIGN METHODS ELISA protocol for quantitation of mite (Dermatophagoides spp.) Der p 1 or Der f 1 ELISA protocol for mite (Dermatophagoides spp.) Group 2 ALLERGENS RESULTS (SUMMARY) TABLE
CHEMILUMINESCENCE ENZYME IMMUNOASSAY (CLIA) Toxoplasma IgG. Cat # (20-25 C Room temp.) Volume
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
OIE Reference Laboratory Reports Activities
 OIE Reference Laboratory Reports Activities Activities in 2014 This report has been submitted : 2015-01-16 15:04:37 Name of disease (or topic) for which you are a designated OIE Reference Laboratory: Equine
OIE Reference Laboratory Reports Activities Activities in 2014 This report has been submitted : 2015-01-16 15:04:37 Name of disease (or topic) for which you are a designated OIE Reference Laboratory: Equine
ASF DIAGNOSIS UPDATE. oie - ASF REFERENCE LABORATORY, MADRID. Prof. José M. Sánchez- Vizcaíno
 ASF DIAGNOSIS UPDATE oie - ASF REFERENCE LABORATORY, MADRID Prof. José M. Sánchez- Vizcaíno jmvizcaino@visavet.ucm.es www.sanidadanimal.info BSL-2 LABORATORIES BSL-3 LABORATORIES BSL-3 BOXES MONITORING
ASF DIAGNOSIS UPDATE oie - ASF REFERENCE LABORATORY, MADRID Prof. José M. Sánchez- Vizcaíno jmvizcaino@visavet.ucm.es www.sanidadanimal.info BSL-2 LABORATORIES BSL-3 LABORATORIES BSL-3 BOXES MONITORING
Updates in rabies vaccine protocols and diagnostic techniques used globally and nationally
 Updates in rabies vaccine protocols and diagnostic techniques used globally and nationally Susan Moore, Rabies Laboratory/KSVDL/College of Veterinary Medicine/Kansas State University, Manhattan, KS 66502,
Updates in rabies vaccine protocols and diagnostic techniques used globally and nationally Susan Moore, Rabies Laboratory/KSVDL/College of Veterinary Medicine/Kansas State University, Manhattan, KS 66502,
This kit is intended for Research Use Only. Not for use in diagnostic procedures.
 This kit is intended for Research Use Only. Not for use in diagnostic procedures. Introduction The DRG Epstein-Barr Virus (VCA) IgM Enzyme Immunoassay Kit provides materials for determination of IgM-class
This kit is intended for Research Use Only. Not for use in diagnostic procedures. Introduction The DRG Epstein-Barr Virus (VCA) IgM Enzyme Immunoassay Kit provides materials for determination of IgM-class
WHO Prequalification of In Vitro Diagnostics Programme PUBLIC REPORT. Product: First Response HIV Card Test Number: PQDx
 WHO Prequalification of In Vitro Diagnostics Programme PUBLIC REPORT Product: First Response HIV 1-2-0 Card Test Number: PQDx 0018-010-00 Abstract First Response HIV 1-2-0 Card Test with product codes
WHO Prequalification of In Vitro Diagnostics Programme PUBLIC REPORT Product: First Response HIV 1-2-0 Card Test Number: PQDx 0018-010-00 Abstract First Response HIV 1-2-0 Card Test with product codes
Fluorescent-Antibody Assay
 JOURNAL OF CUNICAL MICROBIOLOGY, Mar. 1977, p. 32-325 Copyright ( 1977 American Society for Microbiology Vol. 5, No. 3 Printed in U.S.A. Comparison of Rabies Humoral Antibody Titers in Rabbits and Humans
JOURNAL OF CUNICAL MICROBIOLOGY, Mar. 1977, p. 32-325 Copyright ( 1977 American Society for Microbiology Vol. 5, No. 3 Printed in U.S.A. Comparison of Rabies Humoral Antibody Titers in Rabbits and Humans
WHO Prequalification of In Vitro Diagnostics PUBLIC REPORT. Product: OraQuick HIV 1/2 Rapid Antibody Test WHO reference number: PQDx
 WHO Prequalification of In Vitro Diagnostics PUBLIC REPORT Product: OraQuick HIV 1/2 Rapid Antibody Test WHO reference number: PQDx 0159-055-00 OraQuick HIV 1/2 Rapid Antibody Test with product codes 5x4-0010
WHO Prequalification of In Vitro Diagnostics PUBLIC REPORT Product: OraQuick HIV 1/2 Rapid Antibody Test WHO reference number: PQDx 0159-055-00 OraQuick HIV 1/2 Rapid Antibody Test with product codes 5x4-0010
The first and only fully-automated, multiplexed solution for Measles, Mumps, Rubella and Varicella-zoster virus antibody testing
 Bio-Rad Laboratories BioPlex 2200 System BioPlex 2200 MMRV IgG Kit The first and only fully-automated, multiplexed solution for Measles, Mumps, Rubella and Varicella-zoster virus antibody testing Bio-Rad
Bio-Rad Laboratories BioPlex 2200 System BioPlex 2200 MMRV IgG Kit The first and only fully-automated, multiplexed solution for Measles, Mumps, Rubella and Varicella-zoster virus antibody testing Bio-Rad
To detect antibodies to Avian Influenza (AI) using the haemagglutination inhibition test in avian serum specimens 2.
 SADC Harmonized SOP for Avian Influenza HA and HI Serological Tests Prepared by: Dr. P.V. Makaya, Dr. Joule Kangumba and Ms Delille Wessels Reviewed by Dr. P.V. Makaya 1. Purpose and scope To detect antibodies
SADC Harmonized SOP for Avian Influenza HA and HI Serological Tests Prepared by: Dr. P.V. Makaya, Dr. Joule Kangumba and Ms Delille Wessels Reviewed by Dr. P.V. Makaya 1. Purpose and scope To detect antibodies
Republic of the Philippines. Department of Health OFFICE OF THE SECRETARY. June 8, 2009 ADMINISTRATIVE ORDER. No:
 Republic of the Philippines Department of Health OFFICE OF THE SECRETARY June 8, 2009 ADMINISTRATIVE ORDER No: 2009-0027 1 / 22 SUBJECT: Amendment to AO 2007-0029 regarding the Revised Guidelines on. I.
Republic of the Philippines Department of Health OFFICE OF THE SECRETARY June 8, 2009 ADMINISTRATIVE ORDER No: 2009-0027 1 / 22 SUBJECT: Amendment to AO 2007-0029 regarding the Revised Guidelines on. I.
HIV-1 ICx/CRx Kit Reagents for Immune Dissociation and Reactivity Confirmation
 INTENDED USE The RETRO-TEK HIV-1 IC x/cr x Kit is supplied for research purposes only. It is not intended for use in the diagnosis or prognosis of disease or for screening and may not be used as a confirmatory
INTENDED USE The RETRO-TEK HIV-1 IC x/cr x Kit is supplied for research purposes only. It is not intended for use in the diagnosis or prognosis of disease or for screening and may not be used as a confirmatory
ACTG Laboratory Technologist Committee Revised Version 2.0 ACTG Lab Man Coulter HIV-1 p24 ELISA May 21, 2004
 Coulter HIV p24 1. PRINCIPLE The Human Immunodeficiency Virus Type 1 (HIV-1) is recognized as the etiologic agent of acquired immunodeficiency syndrome (AIDS). The virus is transmitted by sexual contact,
Coulter HIV p24 1. PRINCIPLE The Human Immunodeficiency Virus Type 1 (HIV-1) is recognized as the etiologic agent of acquired immunodeficiency syndrome (AIDS). The virus is transmitted by sexual contact,
WHO Prequalification of In Vitro Diagnostics PUBLIC REPORT. Product: OraQuick HIV 1/2 Rapid Antibody Test WHO reference number: PQDx
 WHO Prequalification of In Vitro Diagnostics PUBLIC REPORT Product: OraQuick HIV 1/2 Rapid Antibody Test WHO reference number: PQDx 0159-055-00 OraQuick HIV 1/2 Rapid Antibody Test with product codes 5x4-0010
WHO Prequalification of In Vitro Diagnostics PUBLIC REPORT Product: OraQuick HIV 1/2 Rapid Antibody Test WHO reference number: PQDx 0159-055-00 OraQuick HIV 1/2 Rapid Antibody Test with product codes 5x4-0010
H.Pylori IgG
 DIAGNOSTIC AUTOMATION, INC. 21250 Califa Street, Suite 102 and116, Woodland Hills, CA 91367 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com
DIAGNOSTIC AUTOMATION, INC. 21250 Califa Street, Suite 102 and116, Woodland Hills, CA 91367 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com
Exosome ELISA Complete Kits
 Exosome ELISA Complete Kits EXOEL-CD9A-1, EXOEL-CD63A-1, EXOEL-CD81A-1 User Manual See PAC for Storage Conditions for Individual Components Version 12 4/17/2017 A limited-use label license covers this
Exosome ELISA Complete Kits EXOEL-CD9A-1, EXOEL-CD63A-1, EXOEL-CD81A-1 User Manual See PAC for Storage Conditions for Individual Components Version 12 4/17/2017 A limited-use label license covers this
Exosome ELISA Complete Kits
 Exosome ELISA Complete Kits EXOEL-CD9A-1, EXOEL-CD63A-1, EXOEL-CD81A-1 User Manual See PAC for Storage Conditions for Individual Components Version 12 4/17/2017 A limited-use label license covers this
Exosome ELISA Complete Kits EXOEL-CD9A-1, EXOEL-CD63A-1, EXOEL-CD81A-1 User Manual See PAC for Storage Conditions for Individual Components Version 12 4/17/2017 A limited-use label license covers this
FACTORS AFFECTING COMPLIANCE TO RABIES POST-EXPOSURE PROPHYLAXIS AMONG PEDIATRIC PATIENTS SEEN AT THE RESEARCH INSTITUTE FOR TROPICAL MEDICINE
 56 ORIGINAL ARTICLE FACTORS AFFECTING COMPLIANCE TO RABIES POST-EXPOSURE PROPHYLAXIS AMONG PEDIATRIC PATIENTS SEEN AT THE RESEARCH INSTITUTE FOR TROPICAL MEDICINE AUTHORS: Ruth Faye Romero-Sengson *Philippine
56 ORIGINAL ARTICLE FACTORS AFFECTING COMPLIANCE TO RABIES POST-EXPOSURE PROPHYLAXIS AMONG PEDIATRIC PATIENTS SEEN AT THE RESEARCH INSTITUTE FOR TROPICAL MEDICINE AUTHORS: Ruth Faye Romero-Sengson *Philippine
SP.718 Special Topics at Edgerton Center: D-Lab Health: Medical Technologies for the Developing World
 MIT OpenCourseWare http://ocw.mit.edu SP.718 Special Topics at Edgerton Center: D-Lab Health: Medical Technologies for the Developing World Spring 2009 For information about citing these materials or our
MIT OpenCourseWare http://ocw.mit.edu SP.718 Special Topics at Edgerton Center: D-Lab Health: Medical Technologies for the Developing World Spring 2009 For information about citing these materials or our
See external label 2 C-8 C Σ=96 tests Cat # 1505Z. MICROWELL ELISA H.Pylori IgA Cat # 1505Z
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
PNEUMOCOCCUS VALENT LATEX KIT
 PNEUMOCOCCUS 7-10-13-VALENT LATEX KIT Latex particles coated with pneumococcal antiserum raised in rabbits for in vitro diagnostic use Application The Pneumococcus 7-10-13-valent Latex Kit is a ready to
PNEUMOCOCCUS 7-10-13-VALENT LATEX KIT Latex particles coated with pneumococcal antiserum raised in rabbits for in vitro diagnostic use Application The Pneumococcus 7-10-13-valent Latex Kit is a ready to
Human Immunodeficiency Virus type 1 (HIV-1) gp120 / Glycoprotein 120 ELISA Pair Set
 Human Immunodeficiency Virus type 1 (HIV-1) gp120 / Glycoprotein 120 ELISA Pair Set Catalog Number : SEK11233 To achieve the best assay results, this manual must be read carefully before using this product
Human Immunodeficiency Virus type 1 (HIV-1) gp120 / Glycoprotein 120 ELISA Pair Set Catalog Number : SEK11233 To achieve the best assay results, this manual must be read carefully before using this product
FOR LABORATORY USE ONLY
 YK060 Insulin ELISA FOR LABORATORY USE ONLY Kasumigaseki Place, 3-6-7, Kasumigaseki, Chiyoda-ku Tokyo 100-0013 Japan URL: http://www.sceti.co.jp/export/ e-mail: medical@sceti.co.jp Contents
YK060 Insulin ELISA FOR LABORATORY USE ONLY Kasumigaseki Place, 3-6-7, Kasumigaseki, Chiyoda-ku Tokyo 100-0013 Japan URL: http://www.sceti.co.jp/export/ e-mail: medical@sceti.co.jp Contents
H. pylori IgM. Cat # H. pylori IgM ELISA. ELISA: Enzyme Linked Immunosorbent Assay. ELISA - Indirect; Antigen Coated Plate
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com H. pylori
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com H. pylori
RABIES PROTECTION PROGRAM
 Page 1 of 5 STANDARD OPERATING PROCEDURES (SOP) SCOPE Rabies is a virus causing an acute central nervous system infection, which is typically transmitted by introducing the rabies virus into open cuts
Page 1 of 5 STANDARD OPERATING PROCEDURES (SOP) SCOPE Rabies is a virus causing an acute central nervous system infection, which is typically transmitted by introducing the rabies virus into open cuts
CYTOMEGALOVIRUS (CMV) IgM ELISA Kit Protocol
 CYTOMEGALOVIRUS (CMV) IgM ELISA Kit Protocol (Cat. No.:EK-310-91) 330 Beach Road, Burlingame CA Tel: 650-558-8898 Fax: 650-558-1686 E-Mail: info@phoenixpeptide.com www.phoenixpeptide.com INTENDED USE The
CYTOMEGALOVIRUS (CMV) IgM ELISA Kit Protocol (Cat. No.:EK-310-91) 330 Beach Road, Burlingame CA Tel: 650-558-8898 Fax: 650-558-1686 E-Mail: info@phoenixpeptide.com www.phoenixpeptide.com INTENDED USE The
Instructions for Use. APO-AB Annexin V-Biotin Apoptosis Detection Kit 100 tests
 3URGXFW,QIRUPDWLRQ Sigma TACS Annexin V Apoptosis Detection Kits Instructions for Use APO-AB Annexin V-Biotin Apoptosis Detection Kit 100 tests For Research Use Only. Not for use in diagnostic procedures.
3URGXFW,QIRUPDWLRQ Sigma TACS Annexin V Apoptosis Detection Kits Instructions for Use APO-AB Annexin V-Biotin Apoptosis Detection Kit 100 tests For Research Use Only. Not for use in diagnostic procedures.
Toxoplasma gondii IgM (Toxo IgM)
 DIAGNOSTIC AUTOMATION, INC. 21250 Califa Street, Suite 102 and116, Woodland Hills, CA 91367 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com
DIAGNOSTIC AUTOMATION, INC. 21250 Califa Street, Suite 102 and116, Woodland Hills, CA 91367 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com
Application of μmacs Streptavidin MicroBeads for the analysis of HIV-1 directly from patient plasma
 Excerpt from MACS&more Vol 8 1/2004 Application of μmacs Streptavidin MicroBeads for the analysis of HIV-1 directly from patient plasma L. Davis Lupo and Salvatore T. Butera HIV and Retrovirology Branch,
Excerpt from MACS&more Vol 8 1/2004 Application of μmacs Streptavidin MicroBeads for the analysis of HIV-1 directly from patient plasma L. Davis Lupo and Salvatore T. Butera HIV and Retrovirology Branch,
REVIEW OF THE ANALYSIS RELATED TO RABIES DIAGNOSIS AND FOLLOW-UP OF ORAL VACCINATION PERFORMED IN NRLS IN 2015
 European Union European Union WHO Collaborating OIE Reference Reference Centre Reference NANCY LABORATORY FOR RABIES AND WILDLIFE Laboratory for Rabies Institute for Rabies Serology for Research and Management
European Union European Union WHO Collaborating OIE Reference Reference Centre Reference NANCY LABORATORY FOR RABIES AND WILDLIFE Laboratory for Rabies Institute for Rabies Serology for Research and Management
Human Cytomegalovirus Virus (CMV) IgG ELISA Kit
 Human Cytomegalovirus Virus Catalog No: IRAPKT1410 (CMV) IgG ELISA Kit Lot No: SAMPLE INTENDED USE The CMV IgG ELISA is intended for use in evaluating a patient s serologic status to cytomegalovirus (CMV)
Human Cytomegalovirus Virus Catalog No: IRAPKT1410 (CMV) IgG ELISA Kit Lot No: SAMPLE INTENDED USE The CMV IgG ELISA is intended for use in evaluating a patient s serologic status to cytomegalovirus (CMV)
(For In Vitro Diagnostic Use)
 Read this insert carefully before performing the assay and keep for future reference. The reliability of assay procedure other than those described in this package insert cannot be guaranteed. 207727 (For
Read this insert carefully before performing the assay and keep for future reference. The reliability of assay procedure other than those described in this package insert cannot be guaranteed. 207727 (For
Bordetella pertussis igg-pt elisa Kit
 Bordetella pertussis IgG-PT ELISA Kit Application Bordetella pertussis IgG-PT ELISA Kit is a quantitative test for the detection of IgG antibodies against pertussis toxin in human serum samples. Background
Bordetella pertussis IgG-PT ELISA Kit Application Bordetella pertussis IgG-PT ELISA Kit is a quantitative test for the detection of IgG antibodies against pertussis toxin in human serum samples. Background
Each year in the United States, approximately 6,000
 Comparison of anamnestic responses to rabies vaccination in dogs and cats with current and out-of-date vaccination status Michael C. Moore, DVM, MPH; Rolan D. Davis, MS; Qing Kang, PhD; Christopher I.
Comparison of anamnestic responses to rabies vaccination in dogs and cats with current and out-of-date vaccination status Michael C. Moore, DVM, MPH; Rolan D. Davis, MS; Qing Kang, PhD; Christopher I.
Bovine Brucellosis Control of indirect ELISA kits
 Bovine Brucellosis Control of indirect ELISA kits (Individual serum samples & pooled serum samples) Standard Operating Procedure Control of Bovine brucellosis Serum ELISA kits SOP Page 1 / 7 02 February
Bovine Brucellosis Control of indirect ELISA kits (Individual serum samples & pooled serum samples) Standard Operating Procedure Control of Bovine brucellosis Serum ELISA kits SOP Page 1 / 7 02 February
WHO Prequalification of Diagnostics Programme PUBLIC REPORT
 WHO Prequalification of Diagnostics Programme PUBLIC REPORT Product: ABON HIV 1/2/O Tri-Line Human Immunodeficiency Virus Rapid Test Device Number: PQDx 0141-051-00 Abstract ABON HIV 1/2/O Tri-Line Human
WHO Prequalification of Diagnostics Programme PUBLIC REPORT Product: ABON HIV 1/2/O Tri-Line Human Immunodeficiency Virus Rapid Test Device Number: PQDx 0141-051-00 Abstract ABON HIV 1/2/O Tri-Line Human
A LYOPHILIZED FORMULATION TO EXTEND THE SHELF-LIFE OF TUBERCULIN PPD
 SOUTHEAST ASIAN J TROP MED PUBLIC HEALTH A LYOPHILIZED FORMULATION TO EXTEND THE SHELF-LIFE OF TUBERCULIN PPD Thipchuta Bharnthong 1,2, Virapong Prachayasittikul 1, Chartchalerm Isarankura Na Ayudhya 1,
SOUTHEAST ASIAN J TROP MED PUBLIC HEALTH A LYOPHILIZED FORMULATION TO EXTEND THE SHELF-LIFE OF TUBERCULIN PPD Thipchuta Bharnthong 1,2, Virapong Prachayasittikul 1, Chartchalerm Isarankura Na Ayudhya 1,
TECHNICAL BULLETIN. Catalog Number RAB0447 Storage Temperature 20 C
 Phospho-Stat3 (ptyr 705 ) and pan-stat3 ELISA Kit for detection of human, mouse, or rat phospho-stat3 (ptyr 705 ) and pan-stat3 in cell and tissue lysates Catalog Number RAB0447 Storage Temperature 20
Phospho-Stat3 (ptyr 705 ) and pan-stat3 ELISA Kit for detection of human, mouse, or rat phospho-stat3 (ptyr 705 ) and pan-stat3 in cell and tissue lysates Catalog Number RAB0447 Storage Temperature 20
Standards and beyond: challenges of application of old methods to next generation products. Elena Semenova, Penny Post, Tim Fields, Manon Cox
 Standards and beyond: challenges of application of old methods to next generation products Elena Semenova, Penny Post, Tim Fields, Manon Cox March 25, 2014 Recombinant Flu vaccines Propagation of the influenza
Standards and beyond: challenges of application of old methods to next generation products Elena Semenova, Penny Post, Tim Fields, Manon Cox March 25, 2014 Recombinant Flu vaccines Propagation of the influenza
See external label 2 C-8 C 96 tests Chemiluminescence. CMV IgM. Cat # Diluted samples, controls & calibrator 100 µl 30 minutes
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
Safety and immunogenicity of intradermal rabies vaccination for post exposure prophylaxis
 International Journal of Community Medicine and Public Health Hardanahalli RS et al. Int J Community Med Public Health. 2016 Mar;3(3):692-696 http://www.ijcmph.com pissn 2394-6032 eissn 2394-6040 Research
International Journal of Community Medicine and Public Health Hardanahalli RS et al. Int J Community Med Public Health. 2016 Mar;3(3):692-696 http://www.ijcmph.com pissn 2394-6032 eissn 2394-6040 Research
Hosta virus X ELISA KIT
 Hosta virus X ELISA KIT Cat. No.:DEIA9256 Pkg.Size:96T Intended use The Hosta virus X ELISA KIT is used to detect HVX in both leaves and roots of hosta plants. Reagents And Materials Provided 1. Capture
Hosta virus X ELISA KIT Cat. No.:DEIA9256 Pkg.Size:96T Intended use The Hosta virus X ELISA KIT is used to detect HVX in both leaves and roots of hosta plants. Reagents And Materials Provided 1. Capture
Human Carbamylated LDL ELISA Kit (CBL-LDL Quantitation)
 Product Manual Human Carbamylated LDL ELISA Kit (CBL-LDL Quantitation) Catalog Number MET-5032 96 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Lipoproteins are submicroscopic
Product Manual Human Carbamylated LDL ELISA Kit (CBL-LDL Quantitation) Catalog Number MET-5032 96 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Lipoproteins are submicroscopic
Constitutive Reporter Lentiviral Vectors Expressing Fluorescent Proteins
 Constitutive Reporter Lentiviral Vectors Expressing Fluorescent Proteins www.vectalys.com/products/ Constitutive Reporter Lentiviral Vectors Catalog Number referring to this User Manual: 0008VCT; 0009VCT;
Constitutive Reporter Lentiviral Vectors Expressing Fluorescent Proteins www.vectalys.com/products/ Constitutive Reporter Lentiviral Vectors Catalog Number referring to this User Manual: 0008VCT; 0009VCT;
See external label 2 C-8 C Σ=96 tests Cat # 3171Z. Free Estriol. Cat # 3171Z. Enzyme Linked Immunosorbent Assay
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
Human HIV (1+2) antigen&antibody ELISA Kit
 Human HIV (1+2) antigen&antibody ELISA Kit Catalog Number. CSB-E18042h For the qualitative determination of human HIV (1+2) antibody and P24 antigen concentrations in serum, plasma. This package insert
Human HIV (1+2) antigen&antibody ELISA Kit Catalog Number. CSB-E18042h For the qualitative determination of human HIV (1+2) antibody and P24 antigen concentrations in serum, plasma. This package insert
MYCOPLASMA PNEUMONIAE IFA IgM
 1 MYCOPLASMA PNEUMONIAE IFA IgM PMYPNM: Indirect immunofluorescent assay (IFA) kit for the diagnosis of Mycoplasma pneumoniae IgM antibodies in human serum. INTRODUCTION: Atypical pneumonia produced by
1 MYCOPLASMA PNEUMONIAE IFA IgM PMYPNM: Indirect immunofluorescent assay (IFA) kit for the diagnosis of Mycoplasma pneumoniae IgM antibodies in human serum. INTRODUCTION: Atypical pneumonia produced by
