SUPPLEMENTAL MATERIAL: CLINICAL ASPECTS OF inkt CELL BIOLOGY
|
|
|
- Hugo Bradford
- 6 years ago
- Views:
Transcription
1 SUPPLEMENTAL MATERIAL: CLINICAL ASPECTS OF inkt CELL BIOLOGY Injection of α-galcer in Phase I Clinical Trials The first in-human study was a Phase I trial using intravenous injection of α-galcer at a range of doses, from 50 4,800 mg/m 2, in patients with solid tumors (1). α-galcer was administered on days one, eight, and fifteen in a four-weekly cycle, and in the absence of contraindications, the cycle of immunization continued. As previously observed in murine models of α-galcer immunization, a rapid (within 24 h) loss of inkt cells was detected, likely due to transient TCR downregulation as a result of activation (2). Giaccone et al. (1) also described elevated serum levels of several cytokines including TNF-α and GM-CSF in a number of the patients, which correlated with the frequency of inkt cells prior to vaccination. No toxicity was observed, suggesting that activation of inkt cells through intravenous injection of α-galcer is a safe, well-tolerated treatment modality. Although no partial or complete tumor responses were observed, disease was stabilized in a proportion of the patients examined (1). It is important to note, however, that the frequency of inkt cells described in the cancer patients in this study was lower than in healthy donors, an observation that has been confirmed in other studies (3). When this initial study was undertaken, the concept of inkt cell anergy had not yet been reported (4). Repeated injection of soluble [but not cell-associated (5)] α-galcer in mice, with a short interval between administrations, led to a state in which inkt cells were no longer able to respond to a subsequent ligand challenge, a phenomenon that can last for around four weeks and was shown to depend on the PD-1/PD-L1 axis (4, 6, 7). Two Phase I/II trials with intravenous α-galcer were performed in patients with chronic Hepatitis B or Hepatitis C (8, 9). In both trials, the time between doses of α-galcer was longer than in the earlier study, with four weeks between each injection. Similar to the observations of Giaccone et al. (1), infusions of α-galcer in both groups of patients were relatively well tolerated at all the doses used, with low-grade fever and flu-like symptoms, most likely as a result of the cytokine release in response to inkt cell activation. α-galcer administration was less well tolerated in the Hepatitis C study, with a number of patients discontinuing treatment. The authors suggested that the initial baseline frequencies of inkt cells in the Hepatitis C patients may have been elevated in comparison with the previous cancer and Hepatitis B cohorts, potentially leading to increased side effects (9). Both studies reported a decrease in the blood frequency of inkt cells post α-galcer injection and an increase in cytokines, particularly TNF-α and IL-6, in some of the patients. 1
2 However, only a small number of patients in both trials showed evidence of either a transient or sustained decrease in viral DNA in response to α-galcer. These data suggested that inkt cell activation had little or no effect on viral titers and may not be a viable therapeutic strategy in such settings. α-galcer-pulsed Antigen-Presenting Cells By far the largest number of clinical trials have utilized ex vivo generated or isolated antigen-presenting cells (APCs) pulsed with α-galcer. Studies in mice demonstrated that administration of α-galcer-pulsed DCs induced a longer lasting cytokine response compared with injection of free α-galcer (10). Possible reasons for the observed differences include a more effective dose of agonist being presented by the DC, presentation of α-galcer by different CD1d-positive APCs, and the different routes of administration. Indeed, α-galcerpulsed DCs injected subcutaneously in mice did not stimulate a particularly effective inkt cell response, whereas intravenous injection induced a strong cytokine response. Furthermore, presentation of α-galcer by different APCs in vivo led to very different functional responses: DCs were found to stimulate stronger inkt cell responses than B cells, whereas B cells only stimulated Th2 cytokines and could also inhibit the DC-iNKT cell activation (11). However, targeting of inkt cell agonist directly to B cells, for BCRmediated uptake, efficiently elicits inkt cell activation and antibody responses to codelivered antigens (12). The first full-scale Phase I trial using intravenous administration of autologous immature monocyte-derived DCs pulsed with -GalCer was performed in patients with metastatic tumors (13). In a pilot study, the same group vaccinated four patients with metastatic malignancies of different origins with α-galcer-pulsed monocyte-derived DCs injected twice intravenously and twice intradermally (14). Each patient received DCs/injection, which were tolerated with no serious adverse events; an increase in the frequency of inkt cells in peripheral blood following an initial transient fall in numbers immediately postimmunization was also observed. In the larger-scale study, patients received an intravenous injection of α-galcer-pulsed immature DCs, at two weekly intervals (13). A transient decrease in the frequency of inkt, NK, T, and B cells was observed along with increased serum levels of cytokines including IFN- and IL-12 and the trans-activation of both T and NK cells. The DCs were tracked, and although initially found in the lungs, they migrated 2
3 rapidly to the liver and spleen. The authors also reported a decrease in serum tumor markers in 2 out of the 12 patients enrolled, indicating a possible antitumor effect. Two subsequent Phase I trials were published in 2005, both using monocyte-derived DCs pulsed with α-galcer. In one trial, immature DCs cultured in the presence of IL-2 were injected weekly intravenously in 11 patients with advanced or recurrent non-small-cell lung cancer (15); in the second trial, DCs matured with a cocktail of inflammatory cytokines (IL- 1β, IL-6, TNF-α, and PGE-2) and pulsed with α-galcer were injected in 5 patients with myeloma, squamous cancer, or renal cell cancer (5). As previously, DCs were generally well tolerated in both trials, with up to DCs/m 2 being injected in patients with non-smallcell cancer. The trial in non-small-cell cancer patients described an initial transient drop in blood inkt cell frequency after the first injection of DCs, followed by an expansion after the second injection. In this study, cytokine release was not measured directly, but IFN-γ RNA was quantified by qrt-pcr from sorted inkt cells and was increased after the first and second injection of DCs, but undetectable after subsequent injections, suggesting that inkt cells had become anergized (15). In the trial with matured DCs (5), patients initially received unpulsed DCs to establish a baseline response, and a reduced frequency of inkt cells was reported. α-galcer-pulsed DCs were then injected one month apart, with detailed immunomonitoring carried out. Injection of α-galcer-loaded DCs led to an increase of more than 100-fold in the frequency of inkt cells in all the patients, peaking between 7 and 30 days after injection and in some cases for more than 6 months. The authors analyzed multiple serum cytokines and demonstrated a consistent increase in the levels of IL-12p40, IP-10, and MIP-1β. The activation status of antigen-specific memory CD8 + T cells, reactive to either CMV or Influenza peptides, were assessed by HLA-tetramer staining and ELISpot. Injection of α- GalCer-pulsed DCs was associated with an increase in the frequency of IFN-γproducing CMV-specific CD8 + T cells but not a concomitant increase in the frequency of influenzaspecific T cells. These data together with the elevation in IL-12 suggest that injection of α- GalCer-pulsed DCs leads to activation of APCs in vivo, and boosting of T cell responses specific for persistent antigens. In each of the three myeloma patients enrolled in this study, the authors described a decrease in the concentration of M protein in the serum or urine, suggesting a positive effect on tumor growth. The fourth published Phase I clinical trial where α-galcer-pulsed DCs were injected enrolled nine patients with head and neck cancer (16). Patients were injected twice with 1 3
4 10 8 immature APCs directly into the nasal submucosa with a one-week interval between injections. The authors described an increase in the absolute number of inkt cells in the peripheral blood in 4 out of 9 patients, whereas a slight reduction was observed in the remaining cases. There was also clear evidence for enhanced NK cell activity in 8 out of 9 patients. The cells used for injection were a mixture of APCs derived from peripheral blood mononuclear cells, and the percentage of DCs administered in each case ranged from 30 55%. A decrease in tumor diameter in one patient was observed (from 22 to 7 mm), while there was no progression in disease in a further 5 patients. A follow-up trial was conducted in 17 patients, also with head and neck cancer injected with APCs pulsed with α-galcer (17). APCs were injected directly either into the nasal or the oral submucosa and were also labeled, allowing them to be tracked after administration. More APCs were detected in the lymph nodes after administration via the oral submucosa compared with the nasal submucosa, peaking around 48 h after injection. However, a greater increase in the absolute number of inkt cells was observed in the patients via the nasal compared with the oral route. Unfortunately, there was also evidence for an increased frequency of CD4 + /CD25 + FoxP3 + regulatory T cells after injection into the oral mucosa, indicating that despite or possibly because of the presence of activated inkt cells, there may be suppressive effects of antigen administration that are not overcome by injection of α- GalCer. More recently, four more studies were published, in which patients with different cancers were injected with APCs pulsed with α-galcer either intravenously or intradermally (18 21). Motohashi et al. (18) described the results of a large Phase I-II trial with 23 patients with non-small-cell lung cancer injected with APCs generated in the presence of GM-CSF and IL- 2, as previously described (15). Of the 23 patients enrolled, 17 completed the treatment of four intravenous injections of up to cells/m 2 of α-galcer-pulsed cells. The therapeutic regime was well tolerated and led to expansion and activation of inkt cells in some of the patients. More importantly, in the patients with increased IFN-γ production, a prolongation in survival was observed: 10 patients (more than 50% of the patients who completed the trial) who displayed an increase in the frequency of IFN-γ-producing cells of more than twofold compared with baseline levels had a median survival time of nearly 32 months. This compared favorably with the non- or poorly responding patients who had a median survival time of around 10 months, indicating that activation of inkt cells in vivo can lead to a significant enhancement in patient survival. 4
5 Nicol et al. (19) conducted a Phase I trial in patients with metastatic disease of different origins, comparing different numbers of immature monocyte-derived DCs and routes of administration (intravenous and intradermal). Minor side effects were observed, and the treatments were relatively well tolerated by the patients. A number of patients described pain and tenderness and other symptoms associated with inflammation where metastasis had been diagnosed, suggesting that inkt cell activation had led to inflammatory responses to the tumors. Interestingly, tumor flares were also experienced by a number of patients in anatomical locations harboring metastases previously unknown. Such responses were more common in patients that received intravenous DCs compared with intradermal injection. Activation of inkt cells was also more pronounced when DCs were injected intravenously. Six out of 10 of the patients experienced either a minor response (indicated by a reduction in tumor mass or a decrease in tumor-associated biomarkers) or a stabilization of disease progression, an improvement compared with the progressive disease prior to treatment initiation. In a small-scale trial with four patients with non-small-cell lung cancer, GM-CSF/IL-2 generated, α-galcer-pulsed APCs were injected intravenously 7 days prior to surgery to remove the primary tumors. At the time of surgery, an increase in both inkt numbers and IFN-γ production was described, specifically amongst tumor-infiltrating lymphocytes (TILs) (20). Recently, six patients with myeloma were injected with α-galcer-pulsed DCs and treated at the same time with the immunomodulatory drug lenalidomide, which among other effects enhances T cell activation (21). The authors suggested that by administering a lower dose of lenalidomide than is usually used, together with inkt cell agonists, the two treatment regimens may work together. An initial activation-induced decrease in inkt cell numbers was observed, together with activation of NK cells, as demonstrated by increased expression of NKG2D and CD56. The authors also observed concomitant monocyte activation and an increase in eosinophil numbers in the peripheral blood. The serum levels of soluble IL-2 receptor were also increased. Clinical responses, as indicated by a decrease in the tumorassociated monoclonal Ig levels (M spike) were observed in three of the four patients that had a detectable M spike prior to treatment. Together these data suggest that activation of inkt cells has significant potential for being combined with other immune therapies for the treatment of cancers. In Vitro Activated inkt Cells and Combination Therapies 5
6 Another monotherapy approach used in the clinic is based on ex vivo expanded, adoptively transferred inkt cells sourced from the patient. Patients with non-small-cell lung cancer received inkt cells that had been activated in vitro with α-galcer and IL-2 (22). The therapy was well tolerated, increased frequencies of inkt cells were observed, and there was evidence for downstream activation of NK cells to produce elevated levels of IFN-γ. In two other clinical trials (a Phase I trial and a subsequent Phase II trial, both in patients with head and neck squamous cell carcinoma), combination approaches were assessed (23, 24). Patients received ex vivo activated inkt cells intravenously and APCs pulsed with α- GalCer into the nasal submucosa. The frequency of inkt cells was increased in many of the patients, and there was evidence for inkt cell activation. In the Phase I study, 7 of the 8 patients demonstrated either a partial response or stabilization of disease, whereas in the Phase II trial 5 of the 10 patients had evidence for tumor regression that was associated with an increased frequency of inkt cells within the tumor (TILs). Infusion of a large number of inkt cells together with suitable APCs pulsed with α-galcer is therefore an appealing approach, as it could overcome the reduced frequency of inkt cells described in cancer patients. In mice, functionally competent inkt cells have been differentiated from induced pluripotent stem cells (ipscs), and this may represent an approach to expand inkt cells for cancer therapy in humans (25). Although ipsc-derived inkt cells were shown to release IFN-γ and to suppress tumor growth in vivo, it remains to be demonstrated whether they express PLZF and retain innate-like properties. Further studies have also been performed investigating the role of inkt cells in hematopoietic stem cell transplantation (reviewed in 26). Patients without a complete reconstitution of the inkt cell compartment have an increased rate of leukemia relapse, suggesting a role for inkt cells in the control of both the graft-versus-leukemia (GVL) and graft-versus-host disease (GVHD) responses. LITERATURE CITED 1. Giaccone G, Punt CJ, Ando Y, Ruijter R, Nishi N, et al A phase I study of the natural killer T-cell ligand α-galactosylceramide (KRN7000) in patients with solid tumors. Clin. Cancer Res. 8: Crowe NY, Uldrich AP, Kyparissoudis K, Hammond KJ, Hayakawa Y, et al Glycolipid antigen drives rapid expansion and sustained cytokine production by NK T cells. J. Immunol. 171:
7 3. Shimizu K, Hidaka M, Kadowaki N, Makita N, Konishi N, et al Evaluation of the function of human invariant NKT cells from cancer patients using α-galactosylceramideloaded murine dendritic cells. J. Immunol. 177: Parekh VV, Wilson MT, Olivares-Villagomez D, Singh AK, Wu L, et al Glycolipid antigen induces long-term natural killer T cell anergy in mice. J. Clin. Invest 115: Chang DH, Osman K, Connolly J, Kukreja A, Krasovsky J, et al Sustained expansion of NKT cells and antigen-specific T cells after injection of α-galactosylceramide loaded mature dendritic cells in cancer patients. J. Exp. Med. 201: Parekh VV, Lalani S, Kim S, Halder R, Azuma M, et al PD-1/PD-L blockade prevents anergy induction and enhances the anti-tumor activities of glycolipid-activated invariant NKT cells. J. Immunol. 182: Silk J, Salio M, Reddy BG, Shepherd D, Gileadi U, et al Cutting edge: nonglycosidic CD1d lipid ligands activate human and murine invariant NKT cells. J. Immunol. 180: Veldt BJ, van der Vliet HJ, von Blomberg BM, van Vlierberghe H, Gerken G, et al Randomized placebo controlled phase I/II trial of α-galactosylceramide for the treatment of chronic hepatitis C. J. Hepatol. 47: Woltman AM, Ter Borg MJ, Binda RS, Sprengers D, von Blomberg BM, et al α- galactosylceramide in chronic hepatitis B infection: results from a randomized placebocontrolled Phase I/II trial. Antivir. Ther. 14: Fujii S, Shimizu K, Kronenberg M, Steinman RM Prolonged IFN-γ-producing NKT response induced with α-galactosylceramide-loaded DCs. Nat. Immunol. 3: Bezbradica JS, Stanic AK, Matsuki N, Bour-Jordan H, Bluestone JA, et al Distinct roles of dendritic cells and B cells in Va14Ja18 natural T cell activation in vivo. J. Immunol. 174: Barral P, Eckl-Dorna J, Harwood NE, De Santo C, Salio M, et al B cell receptormediated uptake of CD1d-restricted antigen augments antibody responses by recruiting invariant NKT cell help in vivo. Proc. Natl. Acad. Sci. USA 105: Nieda M, Okai M, Tazbirkova A, Lin H, Yamaura A, et al Therapeutic activation of Vα24 + Vβ11 + NKT cells in human subjects results in highly coordinated secondary activation of acquired and innate immunity. Blood 103:
8 14. Okai M, Nieda M, Tazbirkova A, Horley D, Kikuchi A, et al Human peripheral blood Vα24 + Vβ11 + NKT cells expand following administration of α-galactosylceramidepulsed dendritic cells. Vox Sang. 83: Ishikawa A, Motohashi S, Ishikawa E, Fuchida H, Higashino K, et al A phase I study of α-galactosylceramide (KRN7000)-pulsed dendritic cells in patients with advanced and recurrent non-small cell lung cancer. Clin. Cancer Res. 11: Uchida T, Horiguchi S, Tanaka Y, Yamamoto H, Kunii N, et al Phase I study of α- galactosylceramide-pulsed antigen presenting cells administration to the nasal submucosa in unresectable or recurrent head and neck cancer. Cancer Immunol. Immunother. 57: Kurosaki M, Horiguchi S, Yamasaki K, Uchida Y, Motohashi S, et al Migration and immunological reaction after the administration of αgalcer-pulsed antigenpresenting cells into the submucosa of patients with head and neck cancer. Cancer Immunol. Immunother. 60: Motohashi S, Nagato K, Kunii N, Yamamoto H, Yamasaki K, et al A phase I-II study of α-galactosylceramide-pulsed IL-2/GM-CSF-cultured peripheral blood mononuclear cells in patients with advanced and recurrent non-small cell lung cancer. J. Immunol. 182: Nicol AJ, Tazbirkova A, Nieda M Comparison of clinical and immunological effects of intravenous and intradermal administration of α-galactosylceramide (KRN7000)-pulsed dendritic cells. Clin. Cancer Res. 17: Nagato K, Motohashi S, Ishibashi F, Okita K, Yamasaki K, et al Accumulation of activated invariant natural killer T cells in the tumor microenvironment after α- galactosylceramide-pulsed antigen presenting cells. J. Clin. Immunol. 32: Richter J, Neparidze N, Zhang L, Nair S, Monesmith T, et al Clinical regressions and broad immune activation following combination therapy targeting human NKT cells in myeloma. Blood 121: Motohashi S, Ishikawa A, Ishikawa E, Otsuji M, Iizasa T, et al A phase I study of in vitro expanded natural killer T cells in patients with advanced and recurrent non-small cell lung cancer. Clin. Cancer Res. 12: Kunii N, Horiguchi S, Motohashi S, Yamamoto H, Ueno N, et al Combination therapy of in vitro-expanded natural killer T cells and α-galactosylceramide-pulsed 8
9 antigen-presenting cells in patients with recurrent head and neck carcinoma. Cancer Sci. 100: Yamasaki K, Horiguchi S, Kurosaki M, Kunii N, Nagato K, et al Induction of NKT cell-specific immune responses in cancer tissues after NKT cell-targeted adoptive immunotherapy. Clin. Immunol. 138: Watarai H, Fujii S, Yamada D, Rybouchkin A, Sakata S, et al Murine induced pluripotent stem cells can be derived from and differentiate into natural killer T cells. J. Clin. Investig. 120: Dellabona P, Casorati G, de Lalla C, Montagna D, Locatelli F On the use of donorderived inkt cells for adoptive immunotherapy to prevent leukemia recurrence in pediatric recipients of HLA haploidentical HSCT for hematological malignancies. Clin. Immunol. 140:
Synergistic combinations of targeted immunotherapy to combat cancer
 Synergistic combinations of targeted immunotherapy to combat cancer Myung Ah Lee, M.D., Ph. D Division of Medical Oncology, Hepato-biliary pancreatic cancer center Seoul St. Mary s hospital, The Catholic
Synergistic combinations of targeted immunotherapy to combat cancer Myung Ah Lee, M.D., Ph. D Division of Medical Oncology, Hepato-biliary pancreatic cancer center Seoul St. Mary s hospital, The Catholic
Immune surveillance hypothesis (Macfarlane Burnet, 1950s)
 TUMOR-IMMUNITÄT A.K. Abbas, A.H. Lichtman, S. Pillai (6th edition, 2007) Cellular and Molecular Immunology Saunders Elsevier Chapter 17, immunity to tumors Immune surveillance hypothesis (Macfarlane Burnet,
TUMOR-IMMUNITÄT A.K. Abbas, A.H. Lichtman, S. Pillai (6th edition, 2007) Cellular and Molecular Immunology Saunders Elsevier Chapter 17, immunity to tumors Immune surveillance hypothesis (Macfarlane Burnet,
Personalized medicine - cancer immunotherapy
 Personalized medicine - cancer immunotherapy Özcan Met, PhD Senior Staff Scientist, Cell Therapy Director Center for Cancer Immune Therapy Department of Hematology Department of Oncology University Hospital
Personalized medicine - cancer immunotherapy Özcan Met, PhD Senior Staff Scientist, Cell Therapy Director Center for Cancer Immune Therapy Department of Hematology Department of Oncology University Hospital
2/16/2018. The Immune System and Cancer. Fatal Melanoma Transferred in a Donated Kidney 16 years after Melanoma Surgery
 C007: Immunology of Melanoma: Mechanisms of Immune Therapies Delphine J. Lee, MD, PhD Chief and Program Director, Dermatology, Harbor UCLA Medical Center Principal Investigator, Los Angeles Biomedical
C007: Immunology of Melanoma: Mechanisms of Immune Therapies Delphine J. Lee, MD, PhD Chief and Program Director, Dermatology, Harbor UCLA Medical Center Principal Investigator, Los Angeles Biomedical
I have no financial or other relationship to disclose. Invariant Natural Killer (inkt) cells. Overview. B-iNKT cell interactions
 Lipid-Antigen Presentation by CDd + B Cells is Essential for the Maintenance of inkt Cells: Aberrant s from Patients with Systemic Lupus Erythematosus Impair inkt Cell Homeostasis I have no financial or
Lipid-Antigen Presentation by CDd + B Cells is Essential for the Maintenance of inkt Cells: Aberrant s from Patients with Systemic Lupus Erythematosus Impair inkt Cell Homeostasis I have no financial or
CD40L TCR IL-12 TLR-L
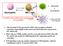 CD40L B cells plasmacells Neutrophils TCR inkt cells IL-12 Ab production Can inkt cells modulate the cytokine profile of neutrophils? TLR-L CD4+ and CD8+ T cell responses 1. The invariant TCR expressed
CD40L B cells plasmacells Neutrophils TCR inkt cells IL-12 Ab production Can inkt cells modulate the cytokine profile of neutrophils? TLR-L CD4+ and CD8+ T cell responses 1. The invariant TCR expressed
CHAPTER 1. The in-betweeners: a short introduction
 CHAPTER 1 The in-betweeners: a short introduction Adapted from: Bispecific antibody platforms for cancer immunotherapy. Lameris R, de Bruin RC, Schneiders FL, van Bergen en Henegouwen PM, Verheul HM, de
CHAPTER 1 The in-betweeners: a short introduction Adapted from: Bispecific antibody platforms for cancer immunotherapy. Lameris R, de Bruin RC, Schneiders FL, van Bergen en Henegouwen PM, Verheul HM, de
One Day BMT Course by Thai Society of Hematology. Management of Graft Failure and Relapsed Diseases
 One Day BMT Course by Thai Society of Hematology Management of Graft Failure and Relapsed Diseases Piya Rujkijyanont, MD Division of Hematology-Oncology Department of Pediatrics Phramongkutklao Hospital
One Day BMT Course by Thai Society of Hematology Management of Graft Failure and Relapsed Diseases Piya Rujkijyanont, MD Division of Hematology-Oncology Department of Pediatrics Phramongkutklao Hospital
Novel RCC Targets from Immuno-Oncology and Antibody-Drug Conjugates
 Novel RCC Targets from Immuno-Oncology and Antibody-Drug Conjugates Christopher Turner, MD Vice President, Clinical Science 04 November 2016 Uveal Melanoma Celldex Pipeline CANDIDATE INDICATION Preclinical
Novel RCC Targets from Immuno-Oncology and Antibody-Drug Conjugates Christopher Turner, MD Vice President, Clinical Science 04 November 2016 Uveal Melanoma Celldex Pipeline CANDIDATE INDICATION Preclinical
The future of HSCT. John Barrett, MD, NHBLI, NIH Bethesda MD
 The future of HSCT John Barrett, MD, NHBLI, NIH Bethesda MD Transplants today Current approaches to improve SCT outcome Optimize stem cell dose and source BMT? PBSCT? Adjusting post transplant I/S to minimize
The future of HSCT John Barrett, MD, NHBLI, NIH Bethesda MD Transplants today Current approaches to improve SCT outcome Optimize stem cell dose and source BMT? PBSCT? Adjusting post transplant I/S to minimize
ACTIVATION AND EFFECTOR FUNCTIONS OF CELL-MEDIATED IMMUNITY AND NK CELLS. Choompone Sakonwasun, MD (Hons), FRCPT
 ACTIVATION AND EFFECTOR FUNCTIONS OF CELL-MEDIATED IMMUNITY AND NK CELLS Choompone Sakonwasun, MD (Hons), FRCPT Types of Adaptive Immunity Types of T Cell-mediated Immune Reactions CTLs = cytotoxic T lymphocytes
ACTIVATION AND EFFECTOR FUNCTIONS OF CELL-MEDIATED IMMUNITY AND NK CELLS Choompone Sakonwasun, MD (Hons), FRCPT Types of Adaptive Immunity Types of T Cell-mediated Immune Reactions CTLs = cytotoxic T lymphocytes
Tumor Immunology. Wirsma Arif Harahap Surgical Oncology Consultant
 Tumor Immunology Wirsma Arif Harahap Surgical Oncology Consultant 1) Immune responses that develop to cancer cells 2) Escape of cancer cells 3) Therapies: clinical and experimental Cancer cells can be
Tumor Immunology Wirsma Arif Harahap Surgical Oncology Consultant 1) Immune responses that develop to cancer cells 2) Escape of cancer cells 3) Therapies: clinical and experimental Cancer cells can be
ASH 2011 aktualijos: MSC TPŠL gydyme. Mindaugas Stoškus VULSK HOTC MRMS
 ASH 2011 aktualijos: MSC TPŠL gydyme Mindaugas Stoškus VULSK HOTC MRMS #3042. Yukiyasu Ozawa et al. Mesenchymal Stem Cells As a Treatment for Steroid-Resistant Acute Graft Versus Host Disease (agvhd);
ASH 2011 aktualijos: MSC TPŠL gydyme Mindaugas Stoškus VULSK HOTC MRMS #3042. Yukiyasu Ozawa et al. Mesenchymal Stem Cells As a Treatment for Steroid-Resistant Acute Graft Versus Host Disease (agvhd);
IMMUNOTHERAPY FOR CANCER A NEW HORIZON. Ekaterini Boleti MD, PhD, FRCP Consultant in Medical Oncology Royal Free London NHS Foundation Trust
 IMMUNOTHERAPY FOR CANCER A NEW HORIZON Ekaterini Boleti MD, PhD, FRCP Consultant in Medical Oncology Royal Free London NHS Foundation Trust ASCO Names Advance of the Year: Cancer Immunotherapy No recent
IMMUNOTHERAPY FOR CANCER A NEW HORIZON Ekaterini Boleti MD, PhD, FRCP Consultant in Medical Oncology Royal Free London NHS Foundation Trust ASCO Names Advance of the Year: Cancer Immunotherapy No recent
Developing Novel Immunotherapeutic Cancer Treatments for Clinical Use
 Developing Novel Immunotherapeutic Cancer Treatments for Clinical Use Oncology for Scientists March 8 th, 2016 Jason Muhitch, PhD Assistant Professor Department of Urology Email: jason.muhitch@roswellpark.org
Developing Novel Immunotherapeutic Cancer Treatments for Clinical Use Oncology for Scientists March 8 th, 2016 Jason Muhitch, PhD Assistant Professor Department of Urology Email: jason.muhitch@roswellpark.org
Regulation of anti-tumor immunity through migration of immune cell subsets within the tumor microenvironment Thomas F. Gajewski, M.D., Ph.D.
 Regulation of anti-tumor immunity through migration of immune cell subsets within the tumor microenvironment Thomas F. Gajewski, M.D., Ph.D. Professor, Departments of Pathology and Medicine Program Leader,
Regulation of anti-tumor immunity through migration of immune cell subsets within the tumor microenvironment Thomas F. Gajewski, M.D., Ph.D. Professor, Departments of Pathology and Medicine Program Leader,
Natural Killer Cells: Development, Diversity, and Applications to Human Disease Dr. Michael A. Caligiuri
 Natural Killer Cells: Development, Diversity, November 26, 2008 The Ohio State University Comprehensive Cancer Center The James Cancer Hospital and Solove Research Institute Columbus, Ohio, USA 1 Human
Natural Killer Cells: Development, Diversity, November 26, 2008 The Ohio State University Comprehensive Cancer Center The James Cancer Hospital and Solove Research Institute Columbus, Ohio, USA 1 Human
T-cell activation T cells migrate to secondary lymphoid tissues where they interact with antigen, antigen-presenting cells, and other lymphocytes:
 Interactions between innate immunity & adaptive immunity What happens to T cells after they leave the thymus? Naïve T cells exit the thymus and enter the bloodstream. If they remain in the bloodstream,
Interactions between innate immunity & adaptive immunity What happens to T cells after they leave the thymus? Naïve T cells exit the thymus and enter the bloodstream. If they remain in the bloodstream,
T-cell activation T cells migrate to secondary lymphoid tissues where they interact with antigen, antigen-presenting cells, and other lymphocytes:
 Interactions between innate immunity & adaptive immunity What happens to T cells after they leave the thymus? Naïve T cells exit the thymus and enter the bloodstream. If they remain in the bloodstream,
Interactions between innate immunity & adaptive immunity What happens to T cells after they leave the thymus? Naïve T cells exit the thymus and enter the bloodstream. If they remain in the bloodstream,
COURSE: Medical Microbiology, PAMB 650/720 - Fall 2008 Lecture 16
 COURSE: Medical Microbiology, PAMB 650/720 - Fall 2008 Lecture 16 Tumor Immunology M. Nagarkatti Teaching Objectives: Introduction to Cancer Immunology Know the antigens expressed by cancer cells Understand
COURSE: Medical Microbiology, PAMB 650/720 - Fall 2008 Lecture 16 Tumor Immunology M. Nagarkatti Teaching Objectives: Introduction to Cancer Immunology Know the antigens expressed by cancer cells Understand
Supplementary Figures
 Supplementary Figures Supplementary Figure 1. NKT ligand-loaded tumour antigen-presenting B cell- and monocyte-based vaccine induces NKT, NK and CD8 T cell responses. (A) The cytokine profiles of liver
Supplementary Figures Supplementary Figure 1. NKT ligand-loaded tumour antigen-presenting B cell- and monocyte-based vaccine induces NKT, NK and CD8 T cell responses. (A) The cytokine profiles of liver
Engineered Immune Cells for Cancer Therapy : Current Status and Prospects
 When Engineering Meets Immunology Engineered Immune Cells for Cancer Therapy : Current Status and Prospects Yong Taik Lim, Ph.D. Nanomedical Systems Laboratory (http://www.nanomedicalsystems.org) SKKU
When Engineering Meets Immunology Engineered Immune Cells for Cancer Therapy : Current Status and Prospects Yong Taik Lim, Ph.D. Nanomedical Systems Laboratory (http://www.nanomedicalsystems.org) SKKU
RXi Pharmaceuticals. Immuno-Oncology World Frontiers Conference. January 23, 2018 NASDAQ: RXII. Property of RXi Pharmaceuticals
 RXi Pharmaceuticals Immuno-Oncology World Frontiers Conference January 23, 2018 NASDAQ: RXII Forward Looking Statements This presentation contains forward-looking statements within the meaning of the Private
RXi Pharmaceuticals Immuno-Oncology World Frontiers Conference January 23, 2018 NASDAQ: RXII Forward Looking Statements This presentation contains forward-looking statements within the meaning of the Private
Lecture outline. Immunological tolerance and immune regulation. Central and peripheral tolerance. Inhibitory receptors of T cells. Regulatory T cells
 1 Immunological tolerance and immune regulation Abul K. Abbas UCSF 2 Lecture outline Central and peripheral tolerance Inhibitory receptors of T cells Regulatory T cells 1 The immunological equilibrium:
1 Immunological tolerance and immune regulation Abul K. Abbas UCSF 2 Lecture outline Central and peripheral tolerance Inhibitory receptors of T cells Regulatory T cells 1 The immunological equilibrium:
TITLE: Development of Antigen Presenting Cells for adoptive immunotherapy in prostate cancer
 AD Award Number: W8-XWH-5-- TITLE: Development of Antigen Presenting Cells for adoptive immunotherapy in prostate cancer PRINCIPAL INVESTIGATOR: Mathias Oelke, Ph.D. CONTRACTING ORGANIZATION: Johns Hopkins
AD Award Number: W8-XWH-5-- TITLE: Development of Antigen Presenting Cells for adoptive immunotherapy in prostate cancer PRINCIPAL INVESTIGATOR: Mathias Oelke, Ph.D. CONTRACTING ORGANIZATION: Johns Hopkins
Tolerance 2. Regulatory T cells; why tolerance fails. Abul K. Abbas UCSF. FOCiS
 1 Tolerance 2. Regulatory T cells; why tolerance fails Abul K. Abbas UCSF FOCiS 2 Lecture outline Regulatory T cells: functions and clinical relevance Pathogenesis of autoimmunity: why selftolerance fails
1 Tolerance 2. Regulatory T cells; why tolerance fails Abul K. Abbas UCSF FOCiS 2 Lecture outline Regulatory T cells: functions and clinical relevance Pathogenesis of autoimmunity: why selftolerance fails
IMMUNOLOGICAL MEMORY. CD4 T Follicular Helper Cells. Memory CD8 T Cell Differentiation
 IMMUNOLOGICAL MEMORY CD4 T Follicular Helper Cells Memory CD8 T Cell Differentiation CD4 T Cell Differentiation Bcl-6 T-bet GATA-3 ROR t Foxp3 CD4 T follicular helper (Tfh) cells FUNCTION Provide essential
IMMUNOLOGICAL MEMORY CD4 T Follicular Helper Cells Memory CD8 T Cell Differentiation CD4 T Cell Differentiation Bcl-6 T-bet GATA-3 ROR t Foxp3 CD4 T follicular helper (Tfh) cells FUNCTION Provide essential
Bases for Immunotherapy in Multiple Myeloma
 Bases for Immunotherapy in Multiple Myeloma Paola Neri, MD, PhD Associate Professor of Medicine University of Calgary, Arnie Charbonneau Cancer Institute Disclosures Paola Neri MD, PhD Grants/research
Bases for Immunotherapy in Multiple Myeloma Paola Neri, MD, PhD Associate Professor of Medicine University of Calgary, Arnie Charbonneau Cancer Institute Disclosures Paola Neri MD, PhD Grants/research
The Immune System. Innate. Adaptive. - skin, mucosal barriers - complement - neutrophils, NK cells, mast cells, basophils, eosinophils
 Objectives - explain the rationale behind cellular adoptive immunotherapy - describe methods of improving cellular adoptive immunotherapy - identify mechanisms of tumor escape from cellular adoptive immunotherapy
Objectives - explain the rationale behind cellular adoptive immunotherapy - describe methods of improving cellular adoptive immunotherapy - identify mechanisms of tumor escape from cellular adoptive immunotherapy
Role of Innate Immunity in Hepatitis B Virus Infection Adam J. Gehring, Ph.D.
 Role of Innate Immunity in Hepatitis B Virus Infection Adam J. Gehring, Ph.D. Biology Lead Toronto Centre for Liver Disease Toronto General Hospital Research Institute University Health Network (UHN) HBV
Role of Innate Immunity in Hepatitis B Virus Infection Adam J. Gehring, Ph.D. Biology Lead Toronto Centre for Liver Disease Toronto General Hospital Research Institute University Health Network (UHN) HBV
Scott Abrams, Ph.D. Professor of Oncology, x4375 Kuby Immunology SEVENTH EDITION
 Scott Abrams, Ph.D. Professor of Oncology, x4375 scott.abrams@roswellpark.org Kuby Immunology SEVENTH EDITION CHAPTER 13 Effector Responses: Cell- and Antibody-Mediated Immunity Copyright 2013 by W. H.
Scott Abrams, Ph.D. Professor of Oncology, x4375 scott.abrams@roswellpark.org Kuby Immunology SEVENTH EDITION CHAPTER 13 Effector Responses: Cell- and Antibody-Mediated Immunity Copyright 2013 by W. H.
Dendritic Cell Based Immunotherapy for Cancer. Edgar G. Engleman, M.D.
 Dendritic Cell Based Immunotherapy for Cancer Edgar G. Engleman, M.D. Two main DC subsets Myeloid (mydc) Derived from monocytes Capture/process/present Ag to T cells Activate NK cells and B cells Plasmacytoid
Dendritic Cell Based Immunotherapy for Cancer Edgar G. Engleman, M.D. Two main DC subsets Myeloid (mydc) Derived from monocytes Capture/process/present Ag to T cells Activate NK cells and B cells Plasmacytoid
Shiv Pillai Ragon Institute, Massachusetts General Hospital Harvard Medical School
 CTLs, Natural Killers and NKTs 1 Shiv Pillai Ragon Institute, Massachusetts General Hospital Harvard Medical School CTL inducing tumor apoptosis 3 Lecture outline CD8 + Cytotoxic T lymphocytes (CTL) Activation/differentiation
CTLs, Natural Killers and NKTs 1 Shiv Pillai Ragon Institute, Massachusetts General Hospital Harvard Medical School CTL inducing tumor apoptosis 3 Lecture outline CD8 + Cytotoxic T lymphocytes (CTL) Activation/differentiation
In-vivo stimulation of macaque natural killer T cells with a-galactosylceramide
 bs_bs_banner Clinical and Experimental Immunology ORIGINAL ARTICLE doi:1.1111/cei.12132 In-vivo stimulation of macaque natural killer T cells with a-galactosylceramide C. S. Fernandez, S. Jegaskanda, D.
bs_bs_banner Clinical and Experimental Immunology ORIGINAL ARTICLE doi:1.1111/cei.12132 In-vivo stimulation of macaque natural killer T cells with a-galactosylceramide C. S. Fernandez, S. Jegaskanda, D.
Releasing the Brakes on Tumor Immunity: Immune Checkpoint Blockade Strategies
 Releasing the Brakes on Tumor Immunity: Immune Checkpoint Blockade Strategies Jason Muhitch, PhD MIR 509 October 1 st, 2014 Email: jason.muhitch@roswellpark.org 0 Holy Grail of Tumor Immunity Exquisite
Releasing the Brakes on Tumor Immunity: Immune Checkpoint Blockade Strategies Jason Muhitch, PhD MIR 509 October 1 st, 2014 Email: jason.muhitch@roswellpark.org 0 Holy Grail of Tumor Immunity Exquisite
Determinants of Immunogenicity and Tolerance. Abul K. Abbas, MD Department of Pathology University of California San Francisco
 Determinants of Immunogenicity and Tolerance Abul K. Abbas, MD Department of Pathology University of California San Francisco EIP Symposium Feb 2016 Why do some people respond to therapeutic proteins?
Determinants of Immunogenicity and Tolerance Abul K. Abbas, MD Department of Pathology University of California San Francisco EIP Symposium Feb 2016 Why do some people respond to therapeutic proteins?
Posters and Presentations
 Posters and Presentations June 2017: American Society of Clinical Oncology (ASCO) Annual - Preliminary Correlative Analysis of PD-L1 expression from the SUNRISE Study. View April 2017: American Association
Posters and Presentations June 2017: American Society of Clinical Oncology (ASCO) Annual - Preliminary Correlative Analysis of PD-L1 expression from the SUNRISE Study. View April 2017: American Association
Supplementary Figure 1. Example of gating strategy
 Supplementary Figure 1. Example of gating strategy Legend Supplementary Figure 1: First, gating is performed to include only single cells (singlets) (A) and CD3+ cells (B). After gating on the lymphocyte
Supplementary Figure 1. Example of gating strategy Legend Supplementary Figure 1: First, gating is performed to include only single cells (singlets) (A) and CD3+ cells (B). After gating on the lymphocyte
Effector mechanisms of cell-mediated immunity: Properties of effector, memory and regulatory T cells
 ICI Basic Immunology course Effector mechanisms of cell-mediated immunity: Properties of effector, memory and regulatory T cells Abul K. Abbas, MD UCSF Stages in the development of T cell responses: induction
ICI Basic Immunology course Effector mechanisms of cell-mediated immunity: Properties of effector, memory and regulatory T cells Abul K. Abbas, MD UCSF Stages in the development of T cell responses: induction
Tumors arise from accumulated genetic mutations. Tumor Immunology (Cancer)
 Tumor Immunology (Cancer) Tumors arise from accumulated genetic mutations Robert Beatty MCB150 Mutations Usually have >6 mutations in both activation/growth factors and tumor suppressor genes. Types of
Tumor Immunology (Cancer) Tumors arise from accumulated genetic mutations Robert Beatty MCB150 Mutations Usually have >6 mutations in both activation/growth factors and tumor suppressor genes. Types of
Key Points of Mistletoe Therapy. Robbin Coedy B.Sc., M.Sc.
 Key Points of Mistletoe Therapy Robbin Coedy B.Sc., M.Sc. Basic Rules of Mistletoe Therapy 1) Inject in the abdomen (preferred), thigh or upper arm 2) Most protocols are 3x/week 3) Start at a low dose
Key Points of Mistletoe Therapy Robbin Coedy B.Sc., M.Sc. Basic Rules of Mistletoe Therapy 1) Inject in the abdomen (preferred), thigh or upper arm 2) Most protocols are 3x/week 3) Start at a low dose
CONTRACTING ORGANIZATION: Johns Hopkins University School of Medicine Baltimore, MD 21205
 AD Award Number: DAMD7---7 TITLE: Development of Artificial Antigen Presenting Cells for Prostate Cancer Immunotherapy PRINCIPAL INVESTIGATOR: Jonathan P. Schneck, M.D., Ph.D. Mathias Oelke, Ph.D. CONTRACTING
AD Award Number: DAMD7---7 TITLE: Development of Artificial Antigen Presenting Cells for Prostate Cancer Immunotherapy PRINCIPAL INVESTIGATOR: Jonathan P. Schneck, M.D., Ph.D. Mathias Oelke, Ph.D. CONTRACTING
Therapeutic efficacy of MUC1- specific CTL and CD137 costimulation. mammary cancer model. Pinku Mukherjee & Sandra Gendler
 Therapeutic efficacy of MUC1- specific CTL and CD137 costimulation in a spontaneous mammary cancer model Pinku Mukherjee & Sandra Gendler Goal of Immunotherapy Boosting the low level anti-tumor immune
Therapeutic efficacy of MUC1- specific CTL and CD137 costimulation in a spontaneous mammary cancer model Pinku Mukherjee & Sandra Gendler Goal of Immunotherapy Boosting the low level anti-tumor immune
Immune Checkpoint Inhibitors: The New Breakout Stars in Cancer Treatment
 Immune Checkpoint Inhibitors: The New Breakout Stars in Cancer Treatment 1 Introductions Peter Langecker, MD, PhD Executive Medical Director, Global Oncology Clinipace Worldwide Mark Shapiro Vice President
Immune Checkpoint Inhibitors: The New Breakout Stars in Cancer Treatment 1 Introductions Peter Langecker, MD, PhD Executive Medical Director, Global Oncology Clinipace Worldwide Mark Shapiro Vice President
Madhav V. Dhodapkar, Joseph Krasovsky, Ralph M. Steinman, and Nina Bhardwaj
 Mature dendritic cells boost functionally superior CD8 + T-cell in humans without foreign helper epitopes Rapid PUBLICATION Madhav V. Dhodapkar, Joseph Krasovsky, Ralph M. Steinman, and Nina Bhardwaj Laboratory
Mature dendritic cells boost functionally superior CD8 + T-cell in humans without foreign helper epitopes Rapid PUBLICATION Madhav V. Dhodapkar, Joseph Krasovsky, Ralph M. Steinman, and Nina Bhardwaj Laboratory
S. No Topic Class No Date
 S. No Topic Class No Date 1 Introduction 2 3 4 5 6 7 Fundamentals of Immunology Definitions and basic terms Types of immunity Organs of immune system Cells of immune system Innate immunity PAMPs PRRs Phagocytosis
S. No Topic Class No Date 1 Introduction 2 3 4 5 6 7 Fundamentals of Immunology Definitions and basic terms Types of immunity Organs of immune system Cells of immune system Innate immunity PAMPs PRRs Phagocytosis
Federica Galaverna, 1 Daria Pagliara, 1 Deepa Manwani, 2 Rajni Agarwal-Hashmi, 3 Melissa Aldinger, 4 Franco Locatelli 1
 Administration of Rivogenlecleucel (Rivo-cel, BPX-501) Following αβ T- and B-Cell Depleted Haplo-HSCT in Children With Transfusion-Dependent Thalassemia Federica Galaverna, 1 Daria Pagliara, 1 Deepa Manwani,
Administration of Rivogenlecleucel (Rivo-cel, BPX-501) Following αβ T- and B-Cell Depleted Haplo-HSCT in Children With Transfusion-Dependent Thalassemia Federica Galaverna, 1 Daria Pagliara, 1 Deepa Manwani,
The Use of Allogeneic Leukocytes Infusion in Cancer Immunotherapy
 Research Article The Use of Allogeneic Leukocytes Infusion in Cancer Immunotherapy Yishu Tang 1*, Chunxia Zhou 2, Wenbo Ma 2, Dongmei Wang 2, Shuren Zhang 2 1 Department of Laboratory Medicine, The First
Research Article The Use of Allogeneic Leukocytes Infusion in Cancer Immunotherapy Yishu Tang 1*, Chunxia Zhou 2, Wenbo Ma 2, Dongmei Wang 2, Shuren Zhang 2 1 Department of Laboratory Medicine, The First
Sleeping Beauty: Current applications and future strategies. CAR-TCR Summit 2017 Partow Kebriaei, MD
 Sleeping Beauty: Current applications and future strategies CAR-TCR Summit 2017 Partow Kebriaei, MD Outline Chimeric antigen receptor (CAR) technology Viral versus nonviral vectors Results of current clinical
Sleeping Beauty: Current applications and future strategies CAR-TCR Summit 2017 Partow Kebriaei, MD Outline Chimeric antigen receptor (CAR) technology Viral versus nonviral vectors Results of current clinical
Exploring Immunotherapies: Beyond Checkpoint Inhibitors
 Exploring Immunotherapies: Beyond Checkpoint Inhibitors Authored by: Jennifer Dolan Fox, PhD VirtualScopics (Now part of BioTelemetry Research) jennifer_fox@virtualscopics.com +1 585 249 6231 Introduction
Exploring Immunotherapies: Beyond Checkpoint Inhibitors Authored by: Jennifer Dolan Fox, PhD VirtualScopics (Now part of BioTelemetry Research) jennifer_fox@virtualscopics.com +1 585 249 6231 Introduction
2/19/2018. The Immune System and Cancer. Fatal Melanoma Transferred in a Donated Kidney 16 years after Melanoma Surgery
 F141: Advanced Melanoma: Mechanisms of Immune Therapies beyond Checkpoint blockade Delphine J. Lee, MD, PhD Chief and Program Director, Dermatology, Harbor UCLA Medical Center Principal Investigator, Los
F141: Advanced Melanoma: Mechanisms of Immune Therapies beyond Checkpoint blockade Delphine J. Lee, MD, PhD Chief and Program Director, Dermatology, Harbor UCLA Medical Center Principal Investigator, Los
NKTR-255: Accessing IL-15 Therapeutic Potential through Robust and Sustained Engagement of Innate and Adaptive Immunity
 NKTR-255: Accessing IL-15 Therapeutic Potential through Robust and Sustained Engagement of Innate and Adaptive Immunity Peiwen Kuo Scientist, Research Biology Nektar Therapeutics August 31 st, 2018 Emerging
NKTR-255: Accessing IL-15 Therapeutic Potential through Robust and Sustained Engagement of Innate and Adaptive Immunity Peiwen Kuo Scientist, Research Biology Nektar Therapeutics August 31 st, 2018 Emerging
Radiation Therapy as an Immunomodulator
 Radiation Therapy as an Immunomodulator Yvonne Mowery, MD, PhD February 20, 2017 Tumor/Immune System Balance Kalbasi, JCI 2013 UNC-Duke-NC State-Wake Forest Spring 2017 2 RT Can Shift Balance Toward Elimination
Radiation Therapy as an Immunomodulator Yvonne Mowery, MD, PhD February 20, 2017 Tumor/Immune System Balance Kalbasi, JCI 2013 UNC-Duke-NC State-Wake Forest Spring 2017 2 RT Can Shift Balance Toward Elimination
08/02/59. Tumor Immunotherapy. Development of Tumor Vaccines. Types of Tumor Vaccines. Immunotherapy w/ Cytokine Gene-Transfected Tumor Cells
 Tumor Immunotherapy Autologous virus Inactivation Inactivated virus Lymphopheresis Culture? Monocyte s Dendritic cells Immunization Autologous vaccine Development of Tumor Vaccines Types of Tumor Vaccines
Tumor Immunotherapy Autologous virus Inactivation Inactivated virus Lymphopheresis Culture? Monocyte s Dendritic cells Immunization Autologous vaccine Development of Tumor Vaccines Types of Tumor Vaccines
Immune Checkpoints. PD Dr med. Alessandra Curioni-Fontecedro Department of Hematology and Oncology Cancer Center Zurich University Hospital Zurich
 Immune Checkpoints PD Dr med. Alessandra Curioni-Fontecedro Department of Hematology and Oncology Cancer Center Zurich University Hospital Zurich Activation of T cells requires co-stimulation Science 3
Immune Checkpoints PD Dr med. Alessandra Curioni-Fontecedro Department of Hematology and Oncology Cancer Center Zurich University Hospital Zurich Activation of T cells requires co-stimulation Science 3
LAMPvax DNA Vaccines as Immunotherapy for Cancer - Three Case Studies
 LAMPvax DNA Vaccines as Immunotherapy for Cancer - Three Case Studies Cancer immunotherapy has emerged as a clinically validated tool for fighting certain kinds of cancers. These therapeutic cancer vaccines
LAMPvax DNA Vaccines as Immunotherapy for Cancer - Three Case Studies Cancer immunotherapy has emerged as a clinically validated tool for fighting certain kinds of cancers. These therapeutic cancer vaccines
Diagnosis of CMV infection UPDATE ECIL
 UPDATE ECIL-4 2011 Recommendations for CMV and HHV-6 management in patients with hematological diseases Per Ljungman, Rafael de la Camara, Hermann Einsele, Dan Engelhard, Pierre Reusser, Jan Styczynski,
UPDATE ECIL-4 2011 Recommendations for CMV and HHV-6 management in patients with hematological diseases Per Ljungman, Rafael de la Camara, Hermann Einsele, Dan Engelhard, Pierre Reusser, Jan Styczynski,
NIH Public Access Author Manuscript Science. Author manuscript; available in PMC 2008 March 12.
 NIH Public Access Author Manuscript Published in final edited form as: Science. 2006 October 6; 314(5796): 126 129. Cancer Regression in Patients After Transfer of Genetically Engineered Lymphocytes Richard
NIH Public Access Author Manuscript Published in final edited form as: Science. 2006 October 6; 314(5796): 126 129. Cancer Regression in Patients After Transfer of Genetically Engineered Lymphocytes Richard
Tumor Immunology: A Primer
 Transcript Details This is a transcript of a continuing medical education (CME) activity accessible on the ReachMD network. Additional media formats for the activity and full activity details (including
Transcript Details This is a transcript of a continuing medical education (CME) activity accessible on the ReachMD network. Additional media formats for the activity and full activity details (including
LESSON 2: THE ADAPTIVE IMMUNITY
 Introduction to immunology. LESSON 2: THE ADAPTIVE IMMUNITY Today we will get to know: The adaptive immunity T- and B-cells Antigens and their recognition How T-cells work 1 The adaptive immunity Unlike
Introduction to immunology. LESSON 2: THE ADAPTIVE IMMUNITY Today we will get to know: The adaptive immunity T- and B-cells Antigens and their recognition How T-cells work 1 The adaptive immunity Unlike
Antigen Presentation and T Lymphocyte Activation. Abul K. Abbas UCSF. FOCiS
 1 Antigen Presentation and T Lymphocyte Activation Abul K. Abbas UCSF FOCiS 2 Lecture outline Dendritic cells and antigen presentation The role of the MHC T cell activation Costimulation, the B7:CD28 family
1 Antigen Presentation and T Lymphocyte Activation Abul K. Abbas UCSF FOCiS 2 Lecture outline Dendritic cells and antigen presentation The role of the MHC T cell activation Costimulation, the B7:CD28 family
0105) at the day 15, respectively1 In contrast, the percentages of CD3 + CD4 + and NK cells displayed no
 2003 12 10 83 23 Natl Med J China, December 10,2003,Vol 83, No. 23 2049 (CIK ), T, CIK 13 (PBMC),,4 7 10 13 15 ;CIK, (DC1) (CD2),CD3 + CD8 +, CD3 + CD56 +, CD25 +, 3315 % 1011 % 717 % 218 %1213 % 415 %,3616
2003 12 10 83 23 Natl Med J China, December 10,2003,Vol 83, No. 23 2049 (CIK ), T, CIK 13 (PBMC),,4 7 10 13 15 ;CIK, (DC1) (CD2),CD3 + CD8 +, CD3 + CD56 +, CD25 +, 3315 % 1011 % 717 % 218 %1213 % 415 %,3616
Ex-Vivo heat shock protein 70-peptide-activated, autologous natural killer cells adoptive therapy: from the bench to the clinic
 Ex-Vivo heat shock protein 70-peptide-activated, autologous natural killer cells adoptive therapy: from the bench to the clinic isbtc 10-13 November 2005 Valeria Milani, MD, PhD Munich Agenda 1. NK ligands
Ex-Vivo heat shock protein 70-peptide-activated, autologous natural killer cells adoptive therapy: from the bench to the clinic isbtc 10-13 November 2005 Valeria Milani, MD, PhD Munich Agenda 1. NK ligands
MECHANISMS OF CELLULAR REJECTION IN ORGAN TRANSPLANTATION AN OVERVIEW
 MECHANISMS OF CELLULAR REJECTION IN ORGAN TRANSPLANTATION AN OVERVIEW YVON LEBRANCHU Service Néphrologie et Immunologie Clinique CHU TOURS ANTIGEN PRESENTING CELL CD4 + T CELL CYTOKINE PRODUCTION CLONAL
MECHANISMS OF CELLULAR REJECTION IN ORGAN TRANSPLANTATION AN OVERVIEW YVON LEBRANCHU Service Néphrologie et Immunologie Clinique CHU TOURS ANTIGEN PRESENTING CELL CD4 + T CELL CYTOKINE PRODUCTION CLONAL
T Lymphocyte Activation and Costimulation. FOCiS. Lecture outline
 1 T Lymphocyte Activation and Costimulation Abul K. Abbas, MD UCSF FOCiS 2 Lecture outline T cell activation Costimulation, the B7:CD28 family Inhibitory receptors of T cells Targeting costimulators for
1 T Lymphocyte Activation and Costimulation Abul K. Abbas, MD UCSF FOCiS 2 Lecture outline T cell activation Costimulation, the B7:CD28 family Inhibitory receptors of T cells Targeting costimulators for
Letter to Editor Tissue micro arrays for immunohistochemical detection of inflammatory infiltrates in renal cell carcinoma
 Int J Clin Exp Med 2014;7(4):1175-1179 www.ijcem.com /ISSN:1940-5901/IJCEM0000102 Letter to Editor Tissue micro arrays for immunohistochemical detection of inflammatory infiltrates in renal cell carcinoma
Int J Clin Exp Med 2014;7(4):1175-1179 www.ijcem.com /ISSN:1940-5901/IJCEM0000102 Letter to Editor Tissue micro arrays for immunohistochemical detection of inflammatory infiltrates in renal cell carcinoma
General Overview of Immunology. Kimberly S. Schluns, Ph.D. Associate Professor Department of Immunology UT MD Anderson Cancer Center
 General Overview of Immunology Kimberly S. Schluns, Ph.D. Associate Professor Department of Immunology UT MD Anderson Cancer Center Objectives Describe differences between innate and adaptive immune responses
General Overview of Immunology Kimberly S. Schluns, Ph.D. Associate Professor Department of Immunology UT MD Anderson Cancer Center Objectives Describe differences between innate and adaptive immune responses
T cell manipulation of the graft: Yes
 T cell manipulation of the graft: Yes J.H. Frederik Falkenburg Department of Hematology L M U C Allogeneic Hematopoietic Stem Cell Transplantation (SCT) for non-malignant disorders: no need for anti-tumor
T cell manipulation of the graft: Yes J.H. Frederik Falkenburg Department of Hematology L M U C Allogeneic Hematopoietic Stem Cell Transplantation (SCT) for non-malignant disorders: no need for anti-tumor
NKTR-255: Accessing The Immunotherapeutic Potential Of IL-15 for NK Cell Therapies
 NKTR-255: Accessing The Immunotherapeutic Potential Of IL-15 for NK Cell Therapies Saul Kivimäe Senior Scientist, Research Biology Nektar Therapeutics NK Cell-Based Cancer Immunotherapy, September 26-27,
NKTR-255: Accessing The Immunotherapeutic Potential Of IL-15 for NK Cell Therapies Saul Kivimäe Senior Scientist, Research Biology Nektar Therapeutics NK Cell-Based Cancer Immunotherapy, September 26-27,
We re Reaching Ludicrous Speed: New Immunotherapy Oncology Medications
 We re Reaching Ludicrous Speed: New Immunotherapy Oncology Medications Adam Peele, PharmD, BCPS, BCOP Oncology Pharmacy Manager Cone Health Disclosures Merck Pharmaceuticals Speaker s Bureau 1 Objectives
We re Reaching Ludicrous Speed: New Immunotherapy Oncology Medications Adam Peele, PharmD, BCPS, BCOP Oncology Pharmacy Manager Cone Health Disclosures Merck Pharmaceuticals Speaker s Bureau 1 Objectives
Cancer Immunotherapy. What is it? Immunotherapy Can Work! 4/15/09. Can the immune system be harnessed to fight cancer? T CD4 T CD28.
 Cancer Immunotherapy CANCER BIOLOGY April 15, 2009 Can the immune system be harnessed to fight cancer? Can the immune system see cancer? What is the best way to turn on the immune system to fight cancer?
Cancer Immunotherapy CANCER BIOLOGY April 15, 2009 Can the immune system be harnessed to fight cancer? Can the immune system see cancer? What is the best way to turn on the immune system to fight cancer?
Immunology Lecture 4. Clinical Relevance of the Immune System
 Immunology Lecture 4 The Well Patient: How innate and adaptive immune responses maintain health - 13, pg 169-181, 191-195. Immune Deficiency - 15 Autoimmunity - 16 Transplantation - 17, pg 260-270 Tumor
Immunology Lecture 4 The Well Patient: How innate and adaptive immune responses maintain health - 13, pg 169-181, 191-195. Immune Deficiency - 15 Autoimmunity - 16 Transplantation - 17, pg 260-270 Tumor
Circulating invariant natural killer T-cell numbers predict outcome in head and neck squamous cell carcinoma: Updated analysis with 10-year follow-up
 Chapter Circulating invariant natural killer T-cell numbers predict outcome in head and neck squamous cell carcinoma: Updated analysis with 10-year follow-up Famke L. Schneiders, Renée C.G. de Bruin, Fons
Chapter Circulating invariant natural killer T-cell numbers predict outcome in head and neck squamous cell carcinoma: Updated analysis with 10-year follow-up Famke L. Schneiders, Renée C.G. de Bruin, Fons
Minor H Antigen-Specific T Cells -The Black Box of the GVL Effect
 Minor H Antigen-Specific T Cells -The Black Box of the GVL Effect All Allogeneic Stem Cell Transplants Are Blind Doctor Donor Patient Genetic polymorphism gives rise to unique peptides in donor and recipient
Minor H Antigen-Specific T Cells -The Black Box of the GVL Effect All Allogeneic Stem Cell Transplants Are Blind Doctor Donor Patient Genetic polymorphism gives rise to unique peptides in donor and recipient
Marshall T Bell Research Resident University of Colorado Grand Rounds Nov. 21, 2011
 Marshall T Bell Research Resident University of Colorado Grand Rounds Nov. 21, 2011 Most common form of cancer in adults ages 25-29 3-5% of skin cancers but 65-75% of deaths Most common metastasis to small
Marshall T Bell Research Resident University of Colorado Grand Rounds Nov. 21, 2011 Most common form of cancer in adults ages 25-29 3-5% of skin cancers but 65-75% of deaths Most common metastasis to small
Supplemental Table I.
 Supplemental Table I Male / Mean ± SEM n Mean ± SEM n Body weight, g 29.2±0.4 17 29.7±0.5 17 Total cholesterol, mg/dl 534.0±30.8 17 561.6±26.1 17 HDL-cholesterol, mg/dl 9.6±0.8 17 10.1±0.7 17 Triglycerides,
Supplemental Table I Male / Mean ± SEM n Mean ± SEM n Body weight, g 29.2±0.4 17 29.7±0.5 17 Total cholesterol, mg/dl 534.0±30.8 17 561.6±26.1 17 HDL-cholesterol, mg/dl 9.6±0.8 17 10.1±0.7 17 Triglycerides,
MATERIALS AND METHODS. Neutralizing antibodies specific to mouse Dll1, Dll4, J1 and J2 were prepared as described. 1,2 All
 MATERIALS AND METHODS Antibodies (Abs), flow cytometry analysis and cell lines Neutralizing antibodies specific to mouse Dll1, Dll4, J1 and J2 were prepared as described. 1,2 All other antibodies used
MATERIALS AND METHODS Antibodies (Abs), flow cytometry analysis and cell lines Neutralizing antibodies specific to mouse Dll1, Dll4, J1 and J2 were prepared as described. 1,2 All other antibodies used
LAG-3: Validation Of Next Generation Checkpoint Pathways
 LAG-3: Validation Of Next Generation Checkpoint Pathways Frédéric Triebel, CO/CMO Immune Checkpoint Modulation & Combination Therapies April 13, 2016 London, UK. 1 AX:PRR; NADAQ:PBMD Notice: Forward Looking
LAG-3: Validation Of Next Generation Checkpoint Pathways Frédéric Triebel, CO/CMO Immune Checkpoint Modulation & Combination Therapies April 13, 2016 London, UK. 1 AX:PRR; NADAQ:PBMD Notice: Forward Looking
Effective activity of cytokine-induced killer cells against autologous metastatic melanoma including cells with stemness features
 Effective activity of cytokine-induced killer cells against autologous metastatic melanoma including cells with stemness features Loretta Gammaitoni, Lidia Giraudo, Valeria Leuci, et al. Clin Cancer Res
Effective activity of cytokine-induced killer cells against autologous metastatic melanoma including cells with stemness features Loretta Gammaitoni, Lidia Giraudo, Valeria Leuci, et al. Clin Cancer Res
Supporting Information
 Supporting Information Chapuis et al. 10.1073/pnas.1113748109 SI Methods Selection of Patients, Targets, Isolation, and Expansion of Melanoma- Specific CTL Clones. Patients were HLA-typed, and their tumors
Supporting Information Chapuis et al. 10.1073/pnas.1113748109 SI Methods Selection of Patients, Targets, Isolation, and Expansion of Melanoma- Specific CTL Clones. Patients were HLA-typed, and their tumors
Adoptive cell therapy using genetically modified antigen-presenting cells
 Adoptive cell therapy using genetically modified antigen-presenting cells Naoto Hirano Ontario Cancer Institute Princess Margaret Cancer Centre University of Toronto UofT-USP Oncology Conference November
Adoptive cell therapy using genetically modified antigen-presenting cells Naoto Hirano Ontario Cancer Institute Princess Margaret Cancer Centre University of Toronto UofT-USP Oncology Conference November
CELLULAR AND MOLECULAR REQUIREMENTS FOR REJECTION OF B16 MELANOMA IN THE SETTING OF REGULATORY T CELL DEPLETION AND HOMEOSTATIC PROLIFERATION
 CELLULAR AND MOLECULAR REQUIREMENTS FOR REJECTION OF B16 MELANOMA IN THE SETTING OF REGULATORY T CELL DEPLETION AND HOMEOSTATIC PROLIFERATION Justin Kline 1, Long Zhang 1, and Thomas F. Gajewski 1,2 Departments
CELLULAR AND MOLECULAR REQUIREMENTS FOR REJECTION OF B16 MELANOMA IN THE SETTING OF REGULATORY T CELL DEPLETION AND HOMEOSTATIC PROLIFERATION Justin Kline 1, Long Zhang 1, and Thomas F. Gajewski 1,2 Departments
June IMMUNE DESIGN The in vivo generation of cytotoxic CD8 T cells (CTLs)
 June 2015 IMMUNE DESIGN The in vivo generation of cytotoxic CD8 T cells (CTLs) 1 Forward-looking Statements This presentation contains forward-looking statements with respect to, among other things, our
June 2015 IMMUNE DESIGN The in vivo generation of cytotoxic CD8 T cells (CTLs) 1 Forward-looking Statements This presentation contains forward-looking statements with respect to, among other things, our
CEL-SCI Corporation. NYSE American: CVM
 CEL-SCI Corporation NYSE American: CVM Geert Kersten Chief Executive Officer 8229 Boone Boulevard, Suite 802 Vienna, VA 22182, USA Phone: (703) 506-9460 Forward Looking Statements This presentation includes
CEL-SCI Corporation NYSE American: CVM Geert Kersten Chief Executive Officer 8229 Boone Boulevard, Suite 802 Vienna, VA 22182, USA Phone: (703) 506-9460 Forward Looking Statements This presentation includes
Autoimmune Diseases. Betsy Kirchner CNP The Cleveland Clinic
 Autoimmune Diseases Betsy Kirchner CNP The Cleveland Clinic Disclosures (financial) No relevant disclosures Learning Objectives Explain the pathophysiology of autoimmune disease Discuss safe administration
Autoimmune Diseases Betsy Kirchner CNP The Cleveland Clinic Disclosures (financial) No relevant disclosures Learning Objectives Explain the pathophysiology of autoimmune disease Discuss safe administration
Multi-Virus-Specific T cell Therapy for Patients after HSC and CB Transplant
 Multi-Virus-Specific T cell Therapy for Patients after HSC and CB Transplant Hanley PJ, Krance BR, Brenner MK, Leen AM, Rooney CM, Heslop HE, Shpall EJ, Bollard CM Hematopoietic Stem Cell Transplantation
Multi-Virus-Specific T cell Therapy for Patients after HSC and CB Transplant Hanley PJ, Krance BR, Brenner MK, Leen AM, Rooney CM, Heslop HE, Shpall EJ, Bollard CM Hematopoietic Stem Cell Transplantation
PUO in the Immunocompromised Host: CMV and beyond
 PUO in the Immunocompromised Host: CMV and beyond PUO in the immunocompromised host: role of viral infections Nature of host defect T cell defects Underlying disease Treatment Nature of clinical presentation
PUO in the Immunocompromised Host: CMV and beyond PUO in the immunocompromised host: role of viral infections Nature of host defect T cell defects Underlying disease Treatment Nature of clinical presentation
Translatability of cytokine data: from animals to humans. Marie-Soleil Piche, PhD Associate Scientific Director of Immunology Charles River, Montreal
 Translatability of cytokine data: from animals to humans Marie-Soleil Piche, PhD Associate Scientific Director of Immunology Charles River, Montreal Presentation outline Overview of cytokines Factors related
Translatability of cytokine data: from animals to humans Marie-Soleil Piche, PhD Associate Scientific Director of Immunology Charles River, Montreal Presentation outline Overview of cytokines Factors related
Restoring Immune Function of Tumor-Specific CD4 + T Cells during Recurrence of Melanoma
 Restoring Immune Function of Tumor-Specific CD4 + T Cells during Recurrence of Melanoma Goding SR et al. J Immunol 2013; 190:4899-4909 C. Nikolowsky Christian Doppler Laboratory for Cardiac and Thoracic
Restoring Immune Function of Tumor-Specific CD4 + T Cells during Recurrence of Melanoma Goding SR et al. J Immunol 2013; 190:4899-4909 C. Nikolowsky Christian Doppler Laboratory for Cardiac and Thoracic
Tolerance 2. Regulatory T cells; why tolerance fails. FOCiS. Lecture outline. Regulatory T cells. Regulatory T cells: functions and clinical relevance
 1 Tolerance 2. Regulatory T cells; why tolerance fails Abul K. Abbas UCSF FOCiS 2 Lecture outline Regulatory T cells: functions and clinical relevance Pathogenesis of autoimmunity: why selftolerance fails
1 Tolerance 2. Regulatory T cells; why tolerance fails Abul K. Abbas UCSF FOCiS 2 Lecture outline Regulatory T cells: functions and clinical relevance Pathogenesis of autoimmunity: why selftolerance fails
Medical Virology Immunology. Dr. Sameer Naji, MB, BCh, PhD (UK) Head of Basic Medical Sciences Dept. Faculty of Medicine The Hashemite University
 Medical Virology Immunology Dr. Sameer Naji, MB, BCh, PhD (UK) Head of Basic Medical Sciences Dept. Faculty of Medicine The Hashemite University Human blood cells Phases of immune responses Microbe Naïve
Medical Virology Immunology Dr. Sameer Naji, MB, BCh, PhD (UK) Head of Basic Medical Sciences Dept. Faculty of Medicine The Hashemite University Human blood cells Phases of immune responses Microbe Naïve
Harnessing tolerance mechanisms with monoclonal antibodies Prof. Herman Waldmann
 Harnessing tolerance mechanisms Herman Waldmann Sir William Dunn School of Pathology, Oxford University, UK 1 1. Transplantation 2 Lymphocyte depletion strategies to minimise drug immunosuppression in
Harnessing tolerance mechanisms Herman Waldmann Sir William Dunn School of Pathology, Oxford University, UK 1 1. Transplantation 2 Lymphocyte depletion strategies to minimise drug immunosuppression in
CONTRACTING ORGANIZATION: Johns Hopkins University School of Medicine Baltimore, MD 21205
 AD Award Number: DAMD7---7 TITLE: Development of Artificial Antigen Presenting Cells for Prostate Cancer Immunotherapy PRINCIPAL INVESTIGATOR: Jonathan P. Schneck, M.D., Ph.D. Mathias Oelke, Ph.D. CONTRACTING
AD Award Number: DAMD7---7 TITLE: Development of Artificial Antigen Presenting Cells for Prostate Cancer Immunotherapy PRINCIPAL INVESTIGATOR: Jonathan P. Schneck, M.D., Ph.D. Mathias Oelke, Ph.D. CONTRACTING
Checkpoint Blockade in Hematology and Stem Cell Transplantation
 Checkpoint Blockade in Hematology and Stem Cell Transplantation Saad S. Kenderian, MD Assistant Professor of Medicine and Oncology Mayo Clinic College of Medicine October 14, 2016 2015 MFMER slide-1 Disclosures
Checkpoint Blockade in Hematology and Stem Cell Transplantation Saad S. Kenderian, MD Assistant Professor of Medicine and Oncology Mayo Clinic College of Medicine October 14, 2016 2015 MFMER slide-1 Disclosures
Dendritic Cell Based Cancer Vaccine Development
 Dendritic Cell Based Cancer Vaccine Development November 10, 2005 CVCWG Meeting Rob Hershberg, MD, PhD Chief Medical Officer 1 Meeting the challenges of developing cancer vaccines-- APC8015 (Provenge TM
Dendritic Cell Based Cancer Vaccine Development November 10, 2005 CVCWG Meeting Rob Hershberg, MD, PhD Chief Medical Officer 1 Meeting the challenges of developing cancer vaccines-- APC8015 (Provenge TM
SEVENTH EDITION CHAPTER
 Judy Owen Jenni Punt Sharon Stranford Kuby Immunology SEVENTH EDITION CHAPTER 16 Tolerance, Autoimmunity, and Transplantation Copyright 2013 by W. H. Freeman and Company Immune tolerance: history * Some
Judy Owen Jenni Punt Sharon Stranford Kuby Immunology SEVENTH EDITION CHAPTER 16 Tolerance, Autoimmunity, and Transplantation Copyright 2013 by W. H. Freeman and Company Immune tolerance: history * Some
Innovation in Prevention, Early Detection & Diagnosis of Colorectal Cancer Heidelberg Workshop Session VI, Oncology Pipeline June 6, 2014
 Innovation in Prevention, Early Detection & Diagnosis of Colorectal Cancer Heidelberg Workshop Session VI, Oncology Pipeline June 6, 2014 Bernd Mueller MSD Sharp & Dohme, Germany Normal Immune Surveillance:
Innovation in Prevention, Early Detection & Diagnosis of Colorectal Cancer Heidelberg Workshop Session VI, Oncology Pipeline June 6, 2014 Bernd Mueller MSD Sharp & Dohme, Germany Normal Immune Surveillance:
Therapeutic activation of V 24 V 11 NKT cells in human subjects results in highly coordinated secondary activation of acquired and innate immunity
 Plenary paper Therapeutic activation of V 24 V 11 NKT cells in human subjects results in highly coordinated secondary activation of acquired and innate immunity Mie Nieda, Miki Okai, Andrea Tazbirkova,
Plenary paper Therapeutic activation of V 24 V 11 NKT cells in human subjects results in highly coordinated secondary activation of acquired and innate immunity Mie Nieda, Miki Okai, Andrea Tazbirkova,
T cell maturation. T-cell Maturation. What allows T cell maturation?
 T-cell Maturation What allows T cell maturation? Direct contact with thymic epithelial cells Influence of thymic hormones Growth factors (cytokines, CSF) T cell maturation T cell progenitor DN DP SP 2ry
T-cell Maturation What allows T cell maturation? Direct contact with thymic epithelial cells Influence of thymic hormones Growth factors (cytokines, CSF) T cell maturation T cell progenitor DN DP SP 2ry
The Immune System. A macrophage. ! Functions of the Immune System. ! Types of Immune Responses. ! Organization of the Immune System
 The Immune System! Functions of the Immune System! Types of Immune Responses! Organization of the Immune System! Innate Defense Mechanisms! Acquired Defense Mechanisms! Applied Immunology A macrophage
The Immune System! Functions of the Immune System! Types of Immune Responses! Organization of the Immune System! Innate Defense Mechanisms! Acquired Defense Mechanisms! Applied Immunology A macrophage
