POLICIES AND PROCEDURE MANUAL
|
|
|
- Angela Horn
- 6 years ago
- Views:
Transcription
1 POLICIES AND PROCEDURE MANUAL Policy: MP058 Section: Medical Benefit Policy Subject: Negative Pressure Wound Therapy I. Policy: Negative Pressure Wound Therapy II. Purpose/Objective: To provide a policy of coverage regarding Negative Pressure Wound Therapy III. Responsibility: A. Medical Directors B. Medical Management IV. Required Definitions 1. Attachment a supporting document that is developed and maintained by the policy writer or department requiring/authoring the policy. 2. Exhibit a supporting document developed and maintained in a department other than the department requiring/authoring the policy. 3. Devised the date the policy was implemented. 4. Revised the date of every revision to the policy, including typographical and grammatical changes. 5. Reviewed the date documenting the annual review if the policy has no revisions necessary. V. Additional Definitions Medical Necessity or Medically Necessary means Covered Services rendered by a Health Care Provider that the Plan determines are: a. appropriate for the symptoms and diagnosis or treatment of the Member's condition, illness, disease or injury; b. provided for the diagnosis, and the direct care and treatment of the Member's condition, illness disease or injury; c. in accordance with current standards of good medical treatment practiced by the general medical community. d. not primarily for the convenience of the Member, or the Member's Health Care Provider; and e. the most appropriate source or level of service that can safely be provided to the Member. When applied to hospitalization, this further means that the Member requires acute care as an inpatient due to the nature of the services rendered or the Member's condition, and the Member cannot receive safe or adequate care as an outpatient. Medicaid Business Segment Medical Necessity shall mean a service or benefit that is compensable under the Medical Assistance Program and if it meets any one of the following standards: (i) (ii) (iii) The service or benefit will, or is reasonably expected to, prevent the onset of an illness, condition or disability. The service or benefit will, or is reasonably expected to, reduce or ameliorate the physical, mental or development effects of an illness, condition, injury or disability. The service or benefit will assist the Member to achieve or maintain maximum functional
2 capacity in performing daily activities, taking into account both the functional capacity of the Member and those functional capacities that are appropriate for members of the same age. ALL Durable Medical Equipment provided for home use requires advanced determination of coverage. Devices furnished at inpatient or outpatient centers are NOT SEPARATELY REIMBURSABLE. Negative Pressure Wound Therapy devices must be obtained through a participating Durable Medical Equipment Vendor(s). DESCRIPTION: Vacuum assisted wound closure is a technique used to promote healing of chronic wounds. It can be used as an adjunct to surgery or as an alternative to surgery for patients who are debilitated or are non-surgical candidates. A special foam dressing with an attached evacuation tube is inserted into the wound. The wound is sealed with an adhesive occlusive dressing. The evacuation tube leads from the wound to a canister that is attached to a negative pressure pump. The negative pressure removes the excess interstitial fluid from the wound. This results in decreased edema that allows for increased blood flow in the wound bed. It is hypothesized that the increased blood flow provides oxygen and nutrients to the wound, thus promoting the formation of granulation tissue. It also draws the edges of the wound closer together. INDICATIONS: Chronic open wounds Diabetic ulcers refractory to conventional treatment Trauma or surgically created wound complication (such as but not limited to dehisced surgical incisions or mesh graft and tissue flap) where there is documentation of medical necessity for accelerated function of granulation tissue which cannot be achieved by other available topical wound treatment Stasis ulcers Pressure ulcers CRITERIA FOR COVERAGE: The staging of pressure ulcers used in this policy is as follows: Stage I: Observable pressure related alteration of intact skin whose indicators as compared to the adjacent or opposite area on the body may include changes in one or more of the following: skin temperature (warmth or coolness), tissue consistency (firm or boggy feel) and/or sensation (pain, itching). The ulcer appears as a defined area of persistent redness in lightly pigmented skin, whereas in darker skin tones, the ulcer may appear with persistent red, blue, or purple hues. Stage II: Stage III: Stage IV Partial thickness skin loss involving epidermis and/or dermis. The ulcer is superficial and presents clinically as an abrasion, blister, or shallow crater. Full thickness skin loss involving damage or necrosis of subcutaneous tissue that may extend down to, but not through, underlying fascia. The ulcer presents clinically as a deep crater with or without undermining of adjacent tissue Full thickness skin loss with extensive destruction, tissue necrosis or damage to muscle, bone or supporting structures. Undermining and sinus tracts also may be associated with Stage IV pressure ulcers Vacuum-assisted closure of wounds may be considered medically necessary for patients with a chronic Stage III or IV pressure ulcer, neuropathic ulcer, venous or arterial insufficiency ulcer, or a chronic ulcer of mixed etiology. A complete wound therapy program described in 1 through 4 below, depending on the type of wound, should have been tried, or considered and ruled out prior to application of vacuum-assisted closure. 1. For all ulcers or chronic open wounds, the following components of a wound therapy program must include a minimum of all of the following general measures, which should be addressed prior to application of vacuumassisted closure: a. Documentation of evaluation, care, and wound measurements by a licensed professional; and b. Moist wound dressings; and c. Debridement of necrotic tissue if present, and d. Evaluation of and provision for adequate nutritional status.
3 2. For Stage III or IV pressure ulcers: a. Appropriate turning and positioning has been documented; and b. A group 2 or 3 support surface has been utilized if pressure ulcer(s) is on the posterior trunk or pelvis, and c. Body moisture and/or incontinence issues have been appropriately managed 3. For neuropathic ulcers (example, diabetic ulcers): a. The member has been participating in a comprehensive diabetic management program to improve their diabetic control; and b. Reduction in pressure on a foot ulcer has been accomplished with appropriate modalities 4. For venous insufficiency ulcers: a. Compression bandages and/or garments have been appropriately and consistently applied, and b. Leg elevation and ambulation have been included in the treatment plan. CONTINUED COVERAGE: If the member meets the criteria for coverage, authorization will be approved in four-week time increments, up to the limitation of the benefit document or applicable rider. 1. Every 30 days from the date of the initial approval a licensed healthcare professional (i.e. treating physician, PA, nurse, or physical therapist) must provide documentation: Wound characteristics including size (length, width, and depth) and drainage (color, odor). Most recent health care professional clinical notes indicating supervision or performing dressing changes. The recorded wound measurements must demonstrate progressive wound healing for continued coverage. Failure to provide this documentation will result in medical necessity denial (per Medicare guidelines). 2. Coverage will cease if any of the following occur No clinically significant objectively measured degree of wound healing (either surface area (length times width) or depth of wound) as determined by the Plan Medical Director or designee has not occurred in the previous month. The recorded wound measurements must be consistently and regularly updated every two weeks and must have demonstrated progressive wound healing. 4 months (Including the time the VAC was applied in an inpatient setting prior to discharge to home) have elapsed using a VAC pump in the treatment of any wound. Requests for coverage in excess of 4 months will be reviewed by the Plan Medical Management in conjunction with a Health Plan Medical Director. If member is not using the device, whether or not by the physician s order. CONTRAINDICATIONS: Fistulas to organs or body cavities Exposed Organs Necrotic tissue in eschar Untreated osteomyelitis Malignancy in the wound Exposed vasculature Exposed nerves Exposed anastomotic site LIMITATIONS: As documented under specific line of business. Member treated in a facility, either acute or sub-acute, will not be separately reimbursed for the device or related equipment. Reimbursement will be inclusive of the contracted per diem rates. EXCLUSIONS: Requests for coverage not meeting criteria, or application of this therapy for conditions listed under contraindications are NOT COVERED. Non-powered mechanical negative pressure wound care systems or single use non-electrically powered negative pressure wound therapy systems, including but not limited to the following, are considered experimental, investigational or unproven and therefore NOT COVERED for any indication: SNaP Wound Care System
4 PICO Single Use Negative Pressure Wound Therapy V.A.C.Via Therapy System cisnap Closed Incision System CODING ASSOCIATED WITH: Negative Pressure Wound Therapy The following codes are included below for informational purposes and may not be all inclusive. Inclusion of a procedure or device code(s) does not constitute or imply coverage nor does it imply or guarantee provider reimbursement. Coverage is determined by the member specific benefit plan document and any applicable laws regarding coverage of specific services. Please note that per Medicare coverage rules, only specific CPT/HCPCS Codes may be covered for the Medicare Business Segment. Please consult the CMS website at or the local Medicare Administrative Carrier (MAC) for more information on Medicare coverage and coding requirements E2402 Negative pressure wound therapy electrical pump, stationary or portable A9272 Wound suction, disposable, includes dressing, all accessories and components, any type, each A6550 Dressing set for negative pressure wound therapy electrical pump, stationary or portable, each A7000 Canister, disposable, used with suction pump, each A7001 canister, non-disposable, used with suction pump, each K0743 suction pump, home model, portable, for use on wounds K0744 absorptive wound dressing for use with suction pump, home model, portable, pad size 16 aq in or less K0745 absorptive wound dressing for use with suction pump, home model, portable, pad size more than 16 sq in but less than or equal to 48 sq in K0746 absorptive wound dressing for use with suction pump, home model, portable, pad size greater than 48 sq in Negative pressure wound therapy, wound assessment and instructions for ongoing care, per session; total wound surface area less than or equal to 50 square centimeters total wound surface area greater than 50 square centimeters Negative pressure wound therapy, (eg, vacuum assisted drainage collection), utilizing disposable, non-durable medical equipment including provision of exudate management collection system, topical application(s), wound assessment, and instructions for ongoing care, per session; total wound(s) surface area less than or equal to 50 square centimeters total wound surface area greater than 50 square centimeters Current Procedural Terminology (CPT ) American Medical Association: Chicago, IL LINE OF BUSINESS: Eligibility and contract specific benefit limitations and/or exclusions will apply. Coverage statements found in the line of business specific benefit document will supersede this policy. For Medicare, applicable LCD s and NCD s will supercede this policy. For PA Medicaid Business segment, this policy applies as written. REFERENCES: Banwell PE, Holten IW, Martin DL, A New Concept in Wound Healing: The Management of Wounds With Negative Pressure Therapy, The British Journal of Surgery, 85(2S) Supplement 2:149, July Mendez-Eastman S, When Wounds Won t Heal, RN, 61(1):20-24, Jan Baxandall T, Healing Cavity Wounds With Negative Pressure, Nursing Standard, 11(6):49-51, 30 Oct Chua-Patel CT, Kinsey GC, Koperski-Moen KJ, Bungum LD, Vacuum-Assisted Wound Closure, American Journal of Nursing, 100(12):45-48, Dec Argenta LC, Morykwas MJ, Vacuum-Assisted Closure: A New Method for Wound Control and Treatment: Clinical Experience, Annals of Plastic Surgery, 38(6): , June Geisinger Technology Assessment Committee, TAC Review: Wound Vac, July 12, 2000 American Academy of Orthopedic Surgeons, Scientific Program, Vacuum Assisted Wound Closure (V.A.C.Therapy) For the Management of Patients With High-Energy Soft Tissue Injuries: A Preliminary Report, March 2001, Tricenturion Negative Pressure Wound Therapy Pumps. Region A DMERC Supplier Manual Revised 4/2004.
5 Gregor S, Maegele M, Sauerland S, et al. Negative pressure wound therapy: A vacuum of evidence? Archives of Surgery 2008;143(2): Sullivan N, Snyder DL, Tipton K, et al. Negative pressure wound therapy devices. Technology Assessment Report. Prepared by the ECRI Evidence-based Practice Center for the Agency for Healthcare Research and Quality (AHRQ), Contract No Project ID: WNDT1108. Rockville, MD: AHRQ; March 30, National Institute for Health and Clinical Excellence (NICE). Negative pressure wound therapy for the open abdomen. Interventional Procedure Guidance 322. London, UK: NICE; December Food and Drug Administration, FDA Safety Communication: UPDATE on Serious Complications Associated with Negative Pressure Wound Therapy Systems, Date Issued: February 24, ECRI Product Brief, SNaP Wound Care System (Spiracur, Inc) for Delivering Negative Pressure Wound Therapy, September 2014 Fong KD, Marston W. Advances in Wound Care. February 2012, 1(1): SNaP Wound Care System: Ultraportable Mechanically Powered Negative Pressure Wound Therapy Hutton DW, Sheehan P. Comparative effectiveness of the SNaP&trade Wound Care System. Int Wound J Apr;8(2): Armstrong DG, Marston WA, Reyzelman AM et al. Comparison of negative pressure wound therapy with an ultraportable mechanically powered device vs. traditional electrically powered device for the treatment of chronic lower extremity ulcers: a multicenter randomized-controlled trial. Wound Repair Regen 2011;19(2): This policy will be revised as necessary and reviewed no less than annually. Medical Management Committee Approval Date: 02/02 Medical Management Administrative Committee Approval Date: 6/13/03 Devised: 02/02 Revised: 6/03; 7/04 criteria; 8/05; 8/06; 8/09; 10/10 (wording, refs), 10/11 (added contraindications); 5/16 (added exclusion); 10/16 (Edited Criteria, Remove P/A) Reviewed: 10/12, 10/13, 10/14, 10/15; 9/17
Negative Pressure Wound Therapy (NPWT)
 Negative Pressure Wound Therapy (NPWT) Policy Number: Original Effective Date: MM.01.005 11/19/1999 Line(s) of Business: Current Effective Date: HMO; PPO; QUEST 01/01/2015 Section: DME Place(s) of Service:
Negative Pressure Wound Therapy (NPWT) Policy Number: Original Effective Date: MM.01.005 11/19/1999 Line(s) of Business: Current Effective Date: HMO; PPO; QUEST 01/01/2015 Section: DME Place(s) of Service:
See Policy CPT/HCPCS CODE section below for any prior authorization requirements
 Effective Date: 7/1/2018 Section: DME Policy No: 377 Medical Officer 7/1/18 Date Technology Assessment Committee Approved Date: 10/10; 10/13; 9/14: 9/15; 4/16 Medical Policy Committee Approved Date: 3/03;
Effective Date: 7/1/2018 Section: DME Policy No: 377 Medical Officer 7/1/18 Date Technology Assessment Committee Approved Date: 10/10; 10/13; 9/14: 9/15; 4/16 Medical Policy Committee Approved Date: 3/03;
Premier Health Plan considers Negative Pressure Wound Therapy (NPWT) in the home setting medically necessary for the following indications:
 Premier Health Plan POLICY AND PROCEDURE MANUAL PA.009.PH Negative Pressure Wound Therapy This policy applies to the following lines of business: Premier Commercial Premier Employee Premier Health Plan
Premier Health Plan POLICY AND PROCEDURE MANUAL PA.009.PH Negative Pressure Wound Therapy This policy applies to the following lines of business: Premier Commercial Premier Employee Premier Health Plan
Negative Pressure Wound Therapy
 Origination: 6/29/04 Revised: 8/24/16 Annual Review: 11/10/16 Purpose: To provide Negative Pressure Wound Therapy (wound care treatment) guidelines for the Medical Department staff to reference when making
Origination: 6/29/04 Revised: 8/24/16 Annual Review: 11/10/16 Purpose: To provide Negative Pressure Wound Therapy (wound care treatment) guidelines for the Medical Department staff to reference when making
Topical Oxygen Wound Therapy (MEDICAID)
 Topical Oxygen Wound Therapy (MEDICAID) Last Review Date: September 8, 2017 Number: MG.MM.DM.15C8v2 Medical Guideline Disclaimer Property of EmblemHealth. All rights reserved. The treating physician or
Topical Oxygen Wound Therapy (MEDICAID) Last Review Date: September 8, 2017 Number: MG.MM.DM.15C8v2 Medical Guideline Disclaimer Property of EmblemHealth. All rights reserved. The treating physician or
Negative Pressure Wound Therapy Pumps
 Negative Pressure Wound Therapy Pumps Adopted from the National Government Services website. For any item to be covered by The Health Plan, it must: 1. Be eligible for a defined Medicare or The Health
Negative Pressure Wound Therapy Pumps Adopted from the National Government Services website. For any item to be covered by The Health Plan, it must: 1. Be eligible for a defined Medicare or The Health
VACUUM ASSISTED CLOSURE (V.A.C.) THERAPY: Mr. Ismazizi Zaharudin Jabatan pembedahan Am Hospital Kuala Lumpur
 VACUUM ASSISTED CLOSURE (V.A.C.) THERAPY: Mr. Ismazizi Zaharudin Jabatan pembedahan Am Hospital Kuala Lumpur Learning Objectives Define Negative Pressure Wound Therapy (NPWT) Discuss guidelines for the
VACUUM ASSISTED CLOSURE (V.A.C.) THERAPY: Mr. Ismazizi Zaharudin Jabatan pembedahan Am Hospital Kuala Lumpur Learning Objectives Define Negative Pressure Wound Therapy (NPWT) Discuss guidelines for the
Durable Medical Equipment Providers
 August 2009 Provider Bulletin Number 974 Durable Medical Equipment Providers Vacuum Assisted Wound Closure Therapy Negative pressure wound therapy (NPWT) must be requested and supplied by an enrolled durable
August 2009 Provider Bulletin Number 974 Durable Medical Equipment Providers Vacuum Assisted Wound Closure Therapy Negative pressure wound therapy (NPWT) must be requested and supplied by an enrolled durable
Assisted Living Resident Assessment (To be used when yes is indicated for skin issues under Section 5 of Assisted Living Resident Assessment)
 Skin Assessment Current open skin areas: Yes No Current pressure ulcer: Yes No A. Stage 1 Ulcers Report based on highest stage of existing ulcers at its worst; do not reverse stage. Number of existing
Skin Assessment Current open skin areas: Yes No Current pressure ulcer: Yes No A. Stage 1 Ulcers Report based on highest stage of existing ulcers at its worst; do not reverse stage. Number of existing
Clinical Policy Title: Vacuum assisted closure in surgical wounds
 Clinical Policy Title: Vacuum assisted closure in surgical wounds Clinical Policy Number: 17.03.00 Effective Date: September 1, 2015 Initial Review Date: June 16, 2013 Most Recent Review Date: August 17,
Clinical Policy Title: Vacuum assisted closure in surgical wounds Clinical Policy Number: 17.03.00 Effective Date: September 1, 2015 Initial Review Date: June 16, 2013 Most Recent Review Date: August 17,
POLICIES AND PROCEDURE MANUAL
 POLICIES AND PROCEDURE MANUAL Policy: MP230 Section: Medical Benefit Policy Subject: Outpatient Pulmonary Rehabilitation I. Policy: Outpatient Pulmonary Rehabilitation II. Purpose/Objective: To provide
POLICIES AND PROCEDURE MANUAL Policy: MP230 Section: Medical Benefit Policy Subject: Outpatient Pulmonary Rehabilitation I. Policy: Outpatient Pulmonary Rehabilitation II. Purpose/Objective: To provide
NEGATIVE PRESSURE WOUND THERAPY
 NEGATIVE PRESSURE WOUND THERAPY Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services, procedures, medical devices
NEGATIVE PRESSURE WOUND THERAPY Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services, procedures, medical devices
SAMPLE. Home Health Reference Tool For Nurses
 Home Health Reference Tool For Nurses Foundation Management Services, Inc. 2010. All rights reserved. Unauthorized reproduction is strictly prohibited. (10/09) Table of Contents 2 Page Medicare Eligibility
Home Health Reference Tool For Nurses Foundation Management Services, Inc. 2010. All rights reserved. Unauthorized reproduction is strictly prohibited. (10/09) Table of Contents 2 Page Medicare Eligibility
The Use of the. in Clinical Practice
 The Use of the SNAP Therapy System in Clinical Practice It s an ultraportable, mechanically-powered disposable NPWT. By Animesh Bhatia, DPM, CWS This article is written exclusively for PM and appears courtesy
The Use of the SNAP Therapy System in Clinical Practice It s an ultraportable, mechanically-powered disposable NPWT. By Animesh Bhatia, DPM, CWS This article is written exclusively for PM and appears courtesy
QUICK GUIDE SNAP THERAPY SYSTEM
 QUICK GUIDE SNAP THERAPY SYSTEM Clinical Pathway to SNAP System Full holistic assessment of patient and wound Is the wound type indicated for NPWT use without contraindications 1? SNAP System is indicated
QUICK GUIDE SNAP THERAPY SYSTEM Clinical Pathway to SNAP System Full holistic assessment of patient and wound Is the wound type indicated for NPWT use without contraindications 1? SNAP System is indicated
Negative Pressure Wound Therapy (NPWT) (Vacuum-Assisted Wound Closure)
 Negative Pressure Wound Therapy (NPWT) (Vacuum-Assisted Wound Closure) Date of Origin: 03/2018 Last Review Date: 03/28/2018 Effective Date: 03/28/2018 Dates Reviewed: 03/2018 Developed By: Medical Necessity
Negative Pressure Wound Therapy (NPWT) (Vacuum-Assisted Wound Closure) Date of Origin: 03/2018 Last Review Date: 03/28/2018 Effective Date: 03/28/2018 Dates Reviewed: 03/2018 Developed By: Medical Necessity
NEGATIVE PRESURE WOUND THERAPY PROGRAM
 NEGATIVE PRESURE WOUND THERAPY PROGRAM WWW.USTOMWOUNDCARE.COM 866-802-0006/901-619-8897 SUPPORT@USTOM.COM TABLE OF CONTENTS 1. 2. 3. 4. To place an order: 1. Please visit our website 2. Please email to
NEGATIVE PRESURE WOUND THERAPY PROGRAM WWW.USTOMWOUNDCARE.COM 866-802-0006/901-619-8897 SUPPORT@USTOM.COM TABLE OF CONTENTS 1. 2. 3. 4. To place an order: 1. Please visit our website 2. Please email to
o Venous edema o Stasis ulcers o Varicose veins (not including spider veins) o Lipodermatosclerosis
 Wound Care Equipment and Supply Benefits to Change for Texas Medicaid July 1, 2018 Effective for dates of service on or after July 1, 2018, wound care equipment and supply benefits will change for Texas
Wound Care Equipment and Supply Benefits to Change for Texas Medicaid July 1, 2018 Effective for dates of service on or after July 1, 2018, wound care equipment and supply benefits will change for Texas
WOCN Document:
 WOCN Document: www.cms.hhs.gov/medicaid/surveycert/080601.pdf OASIS Training Internet site: www.oasistraining.org M0440 Does this patient have a Skin Lesion or an Open Wound? This excludes "OSTOMIES."
WOCN Document: www.cms.hhs.gov/medicaid/surveycert/080601.pdf OASIS Training Internet site: www.oasistraining.org M0440 Does this patient have a Skin Lesion or an Open Wound? This excludes "OSTOMIES."
Corporate Medical Policy
 Corporate Medical Policy Topical Negative Pressure Therapy for Wounds File Name: Origination: Last CAP Review: Next CAP Review: Last Review: topical_negative_pressure_therapy_for_wounds 12/1998 5/2018
Corporate Medical Policy Topical Negative Pressure Therapy for Wounds File Name: Origination: Last CAP Review: Next CAP Review: Last Review: topical_negative_pressure_therapy_for_wounds 12/1998 5/2018
Wound Jeopardy: Name That Wound Session 142 Saturday, September 10 th 2011
 Initial Wound Care Consult History Physical Examination Detailed examination of the wound Photographs Cultures Procedures TCOM ABI Debridement Management Decisions A Detailed History and Physical (wound)
Initial Wound Care Consult History Physical Examination Detailed examination of the wound Photographs Cultures Procedures TCOM ABI Debridement Management Decisions A Detailed History and Physical (wound)
POLICIES AND PROCEDURE MANUAL
 POLICIES AND PROCEDURE MANUAL Policy: MP029 Section: Medical Benefit Policy Subject: Bone Growth Stimulator I. Policy: Bone Growth Stimulator II. Purpose/Objective: To provide a policy of coverage regarding
POLICIES AND PROCEDURE MANUAL Policy: MP029 Section: Medical Benefit Policy Subject: Bone Growth Stimulator I. Policy: Bone Growth Stimulator II. Purpose/Objective: To provide a policy of coverage regarding
V.A.C. Therapy Patient Guide. Are you suffering from a wound? Ask your doctor about V.A.C. Therapy and whether it may be right for you.
 V.A.C. Therapy Patient Guide Are you suffering from a wound? Ask your doctor about V.A.C. Therapy and whether it may be right for you. kci1.com 800.275.4524 Table of Contents Wound Healing is a Process...2
V.A.C. Therapy Patient Guide Are you suffering from a wound? Ask your doctor about V.A.C. Therapy and whether it may be right for you. kci1.com 800.275.4524 Table of Contents Wound Healing is a Process...2
Spinal Cord Injury Info Sheet An information series produced by the Spinal Cord Program at GF Strong Rehab Centre.
 Spinal Cord Injury Info Sheet An information series produced by the Spinal Cord Program at GF Strong Rehab Centre. What does skin do? 1. It protects you. 2. It provides sensory information. 3. It helps
Spinal Cord Injury Info Sheet An information series produced by the Spinal Cord Program at GF Strong Rehab Centre. What does skin do? 1. It protects you. 2. It provides sensory information. 3. It helps
POLICIES AND PROCEDURE MANUAL
 POLICIES AND PROCEDURE MANUAL Policy: MP034 Section: Medical Benefit Policy Subject: Foot Orthotics I. Policy: Foot Orthotics II. Purpose/Objective: To provide a policy of coverage regarding Foot Orthotics
POLICIES AND PROCEDURE MANUAL Policy: MP034 Section: Medical Benefit Policy Subject: Foot Orthotics I. Policy: Foot Orthotics II. Purpose/Objective: To provide a policy of coverage regarding Foot Orthotics
Vacuumed Assisted Closure
 Vacuumed Assisted Closure Louise Morris Lead Nurse in Tissue Viability Jackie Stephen-Haynes Consultant Nurse and senior Lecturer in Tissue Viability 2009 Aims and Objectives To develop an awareness of
Vacuumed Assisted Closure Louise Morris Lead Nurse in Tissue Viability Jackie Stephen-Haynes Consultant Nurse and senior Lecturer in Tissue Viability 2009 Aims and Objectives To develop an awareness of
Coverage Summary. Wound Treatments
 Coverage Summary Wound Treatments Policy Number: W-001 Products: UnitedHealthcare Medicare Advantage Plans Original Approval Date: 02/18/2009 Approved by: UnitedHealthcare Medicare Benefit Interpretation
Coverage Summary Wound Treatments Policy Number: W-001 Products: UnitedHealthcare Medicare Advantage Plans Original Approval Date: 02/18/2009 Approved by: UnitedHealthcare Medicare Benefit Interpretation
Acute and Chronic WOUND ASSESSMENT. Wound Assessment OBJECTIVES ITEMS TO CONSIDER
 WOUND ASSESSMENT Acute and Chronic OBJECTIVES Discuss classification systems and testing methods for pressure ulcers, venous, arterial and diabetic wounds List at least five items to be assessed and documented
WOUND ASSESSMENT Acute and Chronic OBJECTIVES Discuss classification systems and testing methods for pressure ulcers, venous, arterial and diabetic wounds List at least five items to be assessed and documented
Cahaba Medicare Policy Primer 1,2 for Apligraf
 Cahaba Medicare Policy Primer 1,2 for Apligraf MAC A: AL, GA & TN MAC B: AL, GA, & TN LCD# 31428 Indications Applied to partial- or full-thickness ulcers of the lower extremities (see individual product
Cahaba Medicare Policy Primer 1,2 for Apligraf MAC A: AL, GA & TN MAC B: AL, GA, & TN LCD# 31428 Indications Applied to partial- or full-thickness ulcers of the lower extremities (see individual product
Clinical Policy Title: Vacuum assisted closure in surgical wounds
 Clinical Policy Title: Vacuum assisted closure in surgical wounds Clinical Policy Number: 17.03.00 Effective Date: September 1, 2015 Initial Review Date: June 16, 2013 Most Recent Review Date: July 3,
Clinical Policy Title: Vacuum assisted closure in surgical wounds Clinical Policy Number: 17.03.00 Effective Date: September 1, 2015 Initial Review Date: June 16, 2013 Most Recent Review Date: July 3,
Wound Care Program for Nursing Assistants-
 Wound Care Program for Nursing Assistants- Wound Cleansing,Types & Presentation Elizabeth DeFeo, RN, WCC, OMS, CWOCN Wound, Ostomy, & Continence Specialist ldefeo@cornerstonevna.org Outline/Agenda At completion
Wound Care Program for Nursing Assistants- Wound Cleansing,Types & Presentation Elizabeth DeFeo, RN, WCC, OMS, CWOCN Wound, Ostomy, & Continence Specialist ldefeo@cornerstonevna.org Outline/Agenda At completion
Management of Complex Wounds with Vacuum Assisted Closure
 Management of Complex Wounds with Vacuum Assisted Closure Wendy McInnes Vascular / Wound Nurse Practitioner The Queen Elizabeth Hospital, Adelaide, South Australia Treasurer ANZSVN wendy.mcinnes@health.sa.gov.au
Management of Complex Wounds with Vacuum Assisted Closure Wendy McInnes Vascular / Wound Nurse Practitioner The Queen Elizabeth Hospital, Adelaide, South Australia Treasurer ANZSVN wendy.mcinnes@health.sa.gov.au
Determining Wound Diagnosis and Documentation Tips Job Aid
 Determining Wound Diagnosis and Job Aid 1 Coding Is this a traumatic injury from an accident? 800 Codes - Injury Section of the Coding Manual Code by specific site of injury. Only use for accidents or
Determining Wound Diagnosis and Job Aid 1 Coding Is this a traumatic injury from an accident? 800 Codes - Injury Section of the Coding Manual Code by specific site of injury. Only use for accidents or
If both a standardized, validated screening tool and an evaluation of clinical factors are utilized, select Response 2.
 (M1300) Pressure Ulcer Assessment: Was this patient assessed for Risk of Developing Pressure Ulcers? 0 - No assessment conducted [Go to M1306 ] 1 - Yes, based on an evaluation of clinical factors (for
(M1300) Pressure Ulcer Assessment: Was this patient assessed for Risk of Developing Pressure Ulcers? 0 - No assessment conducted [Go to M1306 ] 1 - Yes, based on an evaluation of clinical factors (for
Disclosures. Outpatient NPWT Options Free up Hospital Beds, but Do They Work? Objectives. Clinically Effective: Does it Work?
 4/16/16 Disclosures Consultant, Volcano Corporation Outpatient Options Free up Hospital Beds, but Do They Work? UCSF Vascular Symposium 16 Jonathan Labovitz, DPM Medical Director, Foot & Ankle Center Associate
4/16/16 Disclosures Consultant, Volcano Corporation Outpatient Options Free up Hospital Beds, but Do They Work? UCSF Vascular Symposium 16 Jonathan Labovitz, DPM Medical Director, Foot & Ankle Center Associate
Successful Wound Management Strategies : An Introduction. Alex Khan, APRN ACNS-BC. Organization of Wound Care Nurses
 Successful Wound Management Strategies : An Introduction Alex Khan, APRN ACNS-BC Organization of Wound Care Nurses www.woundcarenurses.org Goals & Objectives The role and importance of wound care management
Successful Wound Management Strategies : An Introduction Alex Khan, APRN ACNS-BC Organization of Wound Care Nurses www.woundcarenurses.org Goals & Objectives The role and importance of wound care management
Summary of Package Insert 1 for PuraPly Wound Matrix
 Summary of Package Insert 1 for PuraPly Wound Matrix For NGS Indications Indicated for the management of wounds including: Partial and full-thickness wounds Venous ulcers Diabetic ulcers Drainage wounds
Summary of Package Insert 1 for PuraPly Wound Matrix For NGS Indications Indicated for the management of wounds including: Partial and full-thickness wounds Venous ulcers Diabetic ulcers Drainage wounds
PNEUMATIC COMPRESSION DEVICES IN THE HOME SETTING
 Status Active Medical and Behavioral Health Policy Section: Medicine Policy Number: II-60 Effective Date: 05/19/2014 Blue Cross and Blue Shield of Minnesota medical policies do not imply that members should
Status Active Medical and Behavioral Health Policy Section: Medicine Policy Number: II-60 Effective Date: 05/19/2014 Blue Cross and Blue Shield of Minnesota medical policies do not imply that members should
Clinical Policy: Low-Frequency Ultrasound Therapy for Wound Management Reference Number: CP.MP.139 Last Review Date: 01/18
 Clinical Policy: Low-Frequency Ultrasound Therapy for Wound Management Reference Number: CP.MP.139 Last Review Date: 01/18 See Important Reminder at the end of this policy for important regulatory and
Clinical Policy: Low-Frequency Ultrasound Therapy for Wound Management Reference Number: CP.MP.139 Last Review Date: 01/18 See Important Reminder at the end of this policy for important regulatory and
Medical Policy Original Effective Date: 01/23/2019
 Page 1 of 12 Disclaimer Refer to the member s specific benefit plan and Schedule of Benefits to determine coverage. This may not be a benefit on all plans or the plan may have broader or more limited benefits
Page 1 of 12 Disclaimer Refer to the member s specific benefit plan and Schedule of Benefits to determine coverage. This may not be a benefit on all plans or the plan may have broader or more limited benefits
Treat the whole patient, not just the hole in the patient! 3/21/2017 CAN YOU CONNECT THE DOTS?? PHILOSOPHY OBJECTIVES
 CAN YOU CONNECT THE DOTS?? Boone Hospital Wound Healing Center Kimberly Jamison, MD, FACP, FAPWCA, PCWC Kim Mitchell, RN, BSN OBJECTIVES Describe the basic concepts of chronic wound care to ensure an optimal
CAN YOU CONNECT THE DOTS?? Boone Hospital Wound Healing Center Kimberly Jamison, MD, FACP, FAPWCA, PCWC Kim Mitchell, RN, BSN OBJECTIVES Describe the basic concepts of chronic wound care to ensure an optimal
First Coast Service Options (FCSO) Medicare Policy Primer
 First Coast Service Options (FCSO) Medicare Policy Primer Medicare Jurisdiction (JN) Florida, Puerto Rico and U.S. Virgin Islands Application of Skin Substitute Grafts for the treatment of DFU and VLU
First Coast Service Options (FCSO) Medicare Policy Primer Medicare Jurisdiction (JN) Florida, Puerto Rico and U.S. Virgin Islands Application of Skin Substitute Grafts for the treatment of DFU and VLU
Summary of Package Insert 1 for PuraPly Antimicrobial Wound Matrix
 Summary of Package Insert 1 for PuraPly Antimicrobial Wound Matrix For States with Non-Published Policies-Novitas Indications Indicated for the management of wounds as an effective barrier to resist microbial
Summary of Package Insert 1 for PuraPly Antimicrobial Wound Matrix For States with Non-Published Policies-Novitas Indications Indicated for the management of wounds as an effective barrier to resist microbial
Recognizing Pressure Injury
 Recognizing Pressure Injury Karen Zulkowski, DNS, RN Hawaii Recorded on March 8, 2017 1 A Little About Myself Executive editor of the Journal of the World Council of Enterostomal Therapists (JWCET) and
Recognizing Pressure Injury Karen Zulkowski, DNS, RN Hawaii Recorded on March 8, 2017 1 A Little About Myself Executive editor of the Journal of the World Council of Enterostomal Therapists (JWCET) and
Uncovering the Pressure Ulcer Coverup Rhonda Kistler RN MS CWON Wound Care Concepts Gentell
 Uncovering the Pressure Ulcer Coverup Rhonda Kistler RN MS CWON Wound Care Concepts Gentell Objectives Identify the stages of pressure ulcer according to the depth of tissue destruction. Discuss the differences
Uncovering the Pressure Ulcer Coverup Rhonda Kistler RN MS CWON Wound Care Concepts Gentell Objectives Identify the stages of pressure ulcer according to the depth of tissue destruction. Discuss the differences
WOUND CARE. By Laural Aiesi, RN, BSN Alina Kisiel RN, BSN Summit ElderCare
 WOUND CARE By Laural Aiesi, RN, BSN Alina Kisiel RN, BSN Summit ElderCare PRESSURE ULCER DIABETIC FOOT ULCER VENOUS ULCER ARTERIAL WOUND NEW OR WORSENING INCONTINENCE CHANGE IN MENTAL STATUS DECLINE IN
WOUND CARE By Laural Aiesi, RN, BSN Alina Kisiel RN, BSN Summit ElderCare PRESSURE ULCER DIABETIC FOOT ULCER VENOUS ULCER ARTERIAL WOUND NEW OR WORSENING INCONTINENCE CHANGE IN MENTAL STATUS DECLINE IN
V.A.C. Therapy Safety Information
 Bringing Safety Home V.A.C. Therapy Safety Information Bleeding Precautions Dressing Change Frequency Foam Removal Important Information 2 Prior to use of V.A.C. Therapy System it is important for the
Bringing Safety Home V.A.C. Therapy Safety Information Bleeding Precautions Dressing Change Frequency Foam Removal Important Information 2 Prior to use of V.A.C. Therapy System it is important for the
OF WOUNDS SENIOR AUDITOR CAROLINAS HEALTHCARE SYSTEM. AHIA 32 nd Annual Conference August 25-28, 2013 Chicago, Illinois
 1 THE WACKY WORLD OF WOUNDS ERIN RYDELL SENIOR AUDITOR CAROLINAS HEALTHCARE SYSTEM AHIA 32 nd Annual Conference August 25-28, 2013 Chicago, Illinois www.ahia.org Carolinas HealthCare System 2 Carolinas
1 THE WACKY WORLD OF WOUNDS ERIN RYDELL SENIOR AUDITOR CAROLINAS HEALTHCARE SYSTEM AHIA 32 nd Annual Conference August 25-28, 2013 Chicago, Illinois www.ahia.org Carolinas HealthCare System 2 Carolinas
Coding for Ulcer Debridement
 Coding for Ulcer Debridement Faculty Jeffrey D. Lehrman, DPM, FASPS, MAPWCA Adjunct Clinical Professor Temple University School of Podiatric Medicine Panel Physician Crozer Center for Wound Healing and
Coding for Ulcer Debridement Faculty Jeffrey D. Lehrman, DPM, FASPS, MAPWCA Adjunct Clinical Professor Temple University School of Podiatric Medicine Panel Physician Crozer Center for Wound Healing and
Skin Integrity and Wound Care
 Skin Integrity and Wound Care By Dr. Amer Hasanien & Dr. Ali Saleh Skin Integrity and Wound Care Skin integrity: the presence of normal Skin & Uninterrupted skin layers by wounds. Factors affecting appearance
Skin Integrity and Wound Care By Dr. Amer Hasanien & Dr. Ali Saleh Skin Integrity and Wound Care Skin integrity: the presence of normal Skin & Uninterrupted skin layers by wounds. Factors affecting appearance
Wound Care per HHVNA Wound Product Formulary
 Venous Ulcers ABI of 0.9-1.2 = normal blood flow An ABI MUST be obtained prior to inititiation of compression therapy. Compression is the Gold Standard of care to promote wound of venous ulcers. Elevation
Venous Ulcers ABI of 0.9-1.2 = normal blood flow An ABI MUST be obtained prior to inititiation of compression therapy. Compression is the Gold Standard of care to promote wound of venous ulcers. Elevation
Prepared and Presented by: Ms. Sohad Noorsaeed, RN. MSN
 Prepared and Presented by: Ms. Sohad Noorsaeed, RN. MSN Learning Outcomes Upon the completion of Skin integrity lecture, the learners will be able to: 1. Describe factors affecting skin integrity. 2. Identify
Prepared and Presented by: Ms. Sohad Noorsaeed, RN. MSN Learning Outcomes Upon the completion of Skin integrity lecture, the learners will be able to: 1. Describe factors affecting skin integrity. 2. Identify
Pressure Injury Staging Update 2016
 Pressure Injury Staging Update 2016 A Review of the New Changes for Pressure Injury Documentation and Staging Jeanne Terefenko, BSN, RN, CWOCN Ext. 5855 Pressure Ulcer Staging Updates: In April, 2016,
Pressure Injury Staging Update 2016 A Review of the New Changes for Pressure Injury Documentation and Staging Jeanne Terefenko, BSN, RN, CWOCN Ext. 5855 Pressure Ulcer Staging Updates: In April, 2016,
and Supplies Amended Date: November 1, 2016 Table of Contents
 Durable Medical Equipment and Supplies Table of Contents 1.0 Description of the Procedure, Product, or Service... 1 1.1... 1 1.2 Categories of... 1 2.0 Eligibility Requirements... 2 2.1 Provisions... 2
Durable Medical Equipment and Supplies Table of Contents 1.0 Description of the Procedure, Product, or Service... 1 1.1... 1 1.2 Categories of... 1 2.0 Eligibility Requirements... 2 2.1 Provisions... 2
2 Pressure Ulcer or Pressure Injury? (Do you have skin in the game?)
 Pressure Ulcer or Pressure Injury? (Do you have skin in the game?) Ann Rambusch, MSN, HCS D, HCS O, RN June 28, 2016 1 Pressure Ulcer or Pressure Injury? (Do you have skin in the game?) Understanding NPUAP
Pressure Ulcer or Pressure Injury? (Do you have skin in the game?) Ann Rambusch, MSN, HCS D, HCS O, RN June 28, 2016 1 Pressure Ulcer or Pressure Injury? (Do you have skin in the game?) Understanding NPUAP
Hemostasis Inflammatory Phase Proliferative/rebuilding Phase Maturation Phase
 The presenters are staff members of the CHI Health St. Elizabeth Burn and Wound Center. Many of the products discussed are used in our current practice but we have no conflict of interest to disclose.
The presenters are staff members of the CHI Health St. Elizabeth Burn and Wound Center. Many of the products discussed are used in our current practice but we have no conflict of interest to disclose.
Medicare C/D Medical Coverage Policy
 Varicose Vein Treatment Medicare C/D Medical Coverage Policy Origination Date: June 1, 1993 Review Date: February 15, 2017 Next Review: February, 2019 DESCRIPTION OF PROCEDURE OR SERVICE Varicose veins
Varicose Vein Treatment Medicare C/D Medical Coverage Policy Origination Date: June 1, 1993 Review Date: February 15, 2017 Next Review: February, 2019 DESCRIPTION OF PROCEDURE OR SERVICE Varicose veins
SKIN INTEGRITY & WOUND CARE
 SKIN INTEGRITY & WOUND CARE Chapter 34 1 skin integrity: intact skin refers to the presence of normal skin layer uninterrupted by wound 2 WOUNDS DISRUPTION IN THE INTEGRITY OF BODY TISSUE CLASSIFIED AS:
SKIN INTEGRITY & WOUND CARE Chapter 34 1 skin integrity: intact skin refers to the presence of normal skin layer uninterrupted by wound 2 WOUNDS DISRUPTION IN THE INTEGRITY OF BODY TISSUE CLASSIFIED AS:
SECTION M: SKIN CONDITIONS. M0210: Unhealed Pressure Ulcer(s) Item Rationale
 SECTION M: SKIN CONDITIONS Intent: The items in this section of the April 1, 2014 release of the LTCH CARE Data Set Version 2.01 document the presence, appearance, and change of pressure ulcers. If warranted
SECTION M: SKIN CONDITIONS Intent: The items in this section of the April 1, 2014 release of the LTCH CARE Data Set Version 2.01 document the presence, appearance, and change of pressure ulcers. If warranted
Hyperbarics in Diabetic Wound Care. Aurel Mihai, MD & Brian Kline, MD
 Hyperbarics in Diabetic Wound Care Aurel Mihai, MD & Brian Kline, MD Presentation Outline The Scope of the Problem Important Definitions Standard Wound Care Hyperbaric Oxygen as an Adjunct Diabetic Foot
Hyperbarics in Diabetic Wound Care Aurel Mihai, MD & Brian Kline, MD Presentation Outline The Scope of the Problem Important Definitions Standard Wound Care Hyperbaric Oxygen as an Adjunct Diabetic Foot
Novitas Medicare Policy Primer
 Novitas Medicare Policy Primer Medicare Jurisdiction (JL and JH) AR, CO, LA, MS, NM, OK, TX, DC, DE, MD, NJ, & PA Counties of Arlington and Fairfax in Virginia and the city of Alexandria in Virginia Application
Novitas Medicare Policy Primer Medicare Jurisdiction (JL and JH) AR, CO, LA, MS, NM, OK, TX, DC, DE, MD, NJ, & PA Counties of Arlington and Fairfax in Virginia and the city of Alexandria in Virginia Application
NPWT Case Series EXPERIENCES WITH INVIA MOTION. Precious life Progressive care. Invia Motion Negative Pressure Wound Therapy
 NPWT Case Series EXPERIENCES WITH INVIA MOTION Invia Motion Negative Pressure Wound Therapy Precious life Progressive care npwt_case_booklet_a4.indd 1 18.12.13 13:17 Chronic sacral pressure ulcer Case
NPWT Case Series EXPERIENCES WITH INVIA MOTION Invia Motion Negative Pressure Wound Therapy Precious life Progressive care npwt_case_booklet_a4.indd 1 18.12.13 13:17 Chronic sacral pressure ulcer Case
Coding for Wound Care
 Coding for Wound Care ****IMPORTANT*** Disclaimer ***Information provided is to the best of our knowledge and as current as possible. ***Please verify all policy and reimbursement information with your
Coding for Wound Care ****IMPORTANT*** Disclaimer ***Information provided is to the best of our knowledge and as current as possible. ***Please verify all policy and reimbursement information with your
Geisinger Health Plan, Geisinger Indemnity Insurance Company and Geisinger Quality Options are
 Bone Growth Stimulator Policy # : MP029 Last Review Date : 2015-10-23 Status : Active Cross References : Reviewed Geisinger Health Plan, Geisinger Indemnity Insurance Company and Geisinger Quality Options
Bone Growth Stimulator Policy # : MP029 Last Review Date : 2015-10-23 Status : Active Cross References : Reviewed Geisinger Health Plan, Geisinger Indemnity Insurance Company and Geisinger Quality Options
POLICIES AND PROCEDURE MANUAL
 POLICIES AND PROCEDURE MANUAL Policy: MP278 Section: Medical Benefit Policy Subject: Hyperthermia in Cancer Therapy (e.g., HIPEC) I. Policy: Hyperthermia in Cancer Therapy (e.g., HIPEC) II. Purpose/Objective:
POLICIES AND PROCEDURE MANUAL Policy: MP278 Section: Medical Benefit Policy Subject: Hyperthermia in Cancer Therapy (e.g., HIPEC) I. Policy: Hyperthermia in Cancer Therapy (e.g., HIPEC) II. Purpose/Objective:
Pressure Ulcer Staging. Staging of Wounds are based on the deepest level of tissue damage
 Pressure Ulcer Staging Staging of Wounds are based on the deepest level of tissue damage Pressure Ulcer Staging New Pressure Ulcer Staging Stage I Stage II Stage III Stage IV Unstageable Suspected Deep
Pressure Ulcer Staging Staging of Wounds are based on the deepest level of tissue damage Pressure Ulcer Staging New Pressure Ulcer Staging Stage I Stage II Stage III Stage IV Unstageable Suspected Deep
Teaming Together to Understand Pressure Injuries / (Ulcers): NPUAP Terminology and Staging Clarification
 Teaming Together to Understand Pressure Injuries / (Ulcers): NPUAP Terminology and Staging Clarification We encourage you to share this information with your staff and colleagues by facilitating clinician
Teaming Together to Understand Pressure Injuries / (Ulcers): NPUAP Terminology and Staging Clarification We encourage you to share this information with your staff and colleagues by facilitating clinician
New Strategies to Improve Assessment, Documentation and Prevention of Pressure Injuries
 New Strategies to Improve Assessment, Documentation and Prevention of Pressure Injuries Janet Cuddigan, PhD, RN, CWCN, FAAN Professor, UNMC College of Nursing Omaha, NE Focus of this Presentation New developments
New Strategies to Improve Assessment, Documentation and Prevention of Pressure Injuries Janet Cuddigan, PhD, RN, CWCN, FAAN Professor, UNMC College of Nursing Omaha, NE Focus of this Presentation New developments
Clinical Policy: EpiFix Wound Treatment
 Clinical Policy: Reference Number: PA.CP.MP.140 Effective Date: 03/18 Last Review Date: 04/18 Coding Implications Revision Log Description EpiFix (MiMedx Group) is dehydrated human amniotic tissue that
Clinical Policy: Reference Number: PA.CP.MP.140 Effective Date: 03/18 Last Review Date: 04/18 Coding Implications Revision Log Description EpiFix (MiMedx Group) is dehydrated human amniotic tissue that
Objectives. Major Changes to Section M. MDS 3.0 Section M Pressure Ulcers. Risk assessment Introduction of NPUAP guidelines
 MDS 3.0 Section M Pressure Ulcers Moderator: Barbara Baylis Sr. VP of Clinical and Residential Services, Kindred Healthcare Presenter: Glenda Mack, Sr. Director of Clinical Operations, Peoplefirst Rehabilitation
MDS 3.0 Section M Pressure Ulcers Moderator: Barbara Baylis Sr. VP of Clinical and Residential Services, Kindred Healthcare Presenter: Glenda Mack, Sr. Director of Clinical Operations, Peoplefirst Rehabilitation
LABETTE COMMUNITY COLLEGE BRIEF SYLLABUS. Please check with the LCC bookstore for the required texts for this class.
 LABETTE COMMUNITY COLLEGE BRIEF SYLLABUS SPECIAL NOTE: This brief syllabus is not intended to be a legal contract. A full syllabus will be distributed to students at the first class session. TEXT AND SUPPLEMENTARY
LABETTE COMMUNITY COLLEGE BRIEF SYLLABUS SPECIAL NOTE: This brief syllabus is not intended to be a legal contract. A full syllabus will be distributed to students at the first class session. TEXT AND SUPPLEMENTARY
Negative Pressure Wound Therapy in the Outpatient Setting
 Negative Pressure Wound Therapy in the Outpatient Setting Policy Number: 1.01.16 Last Review: 9/2017 Origination: 7/2001 Next Review: 9/2018 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will
Negative Pressure Wound Therapy in the Outpatient Setting Policy Number: 1.01.16 Last Review: 9/2017 Origination: 7/2001 Next Review: 9/2018 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will
Wound Care Evaluation by Kris Dalseg MS PT CWS CLT
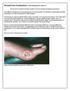 Wound Care Evaluation by Kris Dalseg MS PT CWS CLT This document is intended to describe a standard wound care evaluation for healthcare practitioners. In healthcare, all aspects of our treatment have
Wound Care Evaluation by Kris Dalseg MS PT CWS CLT This document is intended to describe a standard wound care evaluation for healthcare practitioners. In healthcare, all aspects of our treatment have
POLICIES AND PROCEDURE MANUAL
 POLICIES AND PROCEDURE MANUAL Policy: MP120 Section: Medical Benefit Policy Subject: Intracavitary Balloon Brachytherapy for Breast Cancer I. Policy: Intracavitary Balloon Brachytherapy for Breast Cancer
POLICIES AND PROCEDURE MANUAL Policy: MP120 Section: Medical Benefit Policy Subject: Intracavitary Balloon Brachytherapy for Breast Cancer I. Policy: Intracavitary Balloon Brachytherapy for Breast Cancer
Negative Pressure Wound Therapy in the Outpatient Setting. Original Policy Date 12:2013
 MP 1.01.11 Negative Pressure Wound Therapy in the Outpatient Setting Medical Policy Section Durable Medical Equipment Issue 12:2013` Original Policy Date 12:2013 Last Review Status/Date Reviewed with literature
MP 1.01.11 Negative Pressure Wound Therapy in the Outpatient Setting Medical Policy Section Durable Medical Equipment Issue 12:2013` Original Policy Date 12:2013 Last Review Status/Date Reviewed with literature
Wound Classification. Overview
 Overview Jeffrey A. Niezgoda, MD FACHM, MAPWCA, CHWS Review of Initial Wound Care Consultation Rational for Classification Wound Appearance Wound Etiology Management Algorithms Initial Wound Care Consult
Overview Jeffrey A. Niezgoda, MD FACHM, MAPWCA, CHWS Review of Initial Wound Care Consultation Rational for Classification Wound Appearance Wound Etiology Management Algorithms Initial Wound Care Consult
POLICIES AND PROCEDURE MANUAL
 POLICIES AND PROCEDURE MANUAL Policy: MP072 Section: Medical Benefit Policy Subject: Percutaneous Discectomy and Disc Decompression Nucleoplasty I. Policy: Percutaneous Discectomy and Disc Decompression
POLICIES AND PROCEDURE MANUAL Policy: MP072 Section: Medical Benefit Policy Subject: Percutaneous Discectomy and Disc Decompression Nucleoplasty I. Policy: Percutaneous Discectomy and Disc Decompression
C A R O L S I E M M S N, R N, B C, G N P M U S I N C L A I R S C H O O L O F N U R S I N G Q I P M O WHAT S IN A NAME?
 M D S A N D P R E S S U R E U L C E R S C A R O L S I E M M S N, R N, B C, G N P M U S I N C L A I R S C H O O L O F N U R S I N G Q I P M O WHAT S IN A NAME? 1777: decubitus is the oldest term used 1942
M D S A N D P R E S S U R E U L C E R S C A R O L S I E M M S N, R N, B C, G N P M U S I N C L A I R S C H O O L O F N U R S I N G Q I P M O WHAT S IN A NAME? 1777: decubitus is the oldest term used 1942
POLICIES AND PROCEDURE MANUAL
 POLICIES AND PROCEDURE MANUAL Policy: MP091 Section: Medical Benefit Policy Subject: Sacral Nerve Stimulation I. Policy: Sacral Nerve Stimulation II. Purpose/Objective: To provide a policy of coverage
POLICIES AND PROCEDURE MANUAL Policy: MP091 Section: Medical Benefit Policy Subject: Sacral Nerve Stimulation I. Policy: Sacral Nerve Stimulation II. Purpose/Objective: To provide a policy of coverage
POLICIES AND PROCEDURE MANUAL
 POLICIES AND PROCEDURE MANUAL Policy: MP046 Section: Medical Benefit Policy Subject: Progressive Stretch Devices I. Policy: Progressive Stretch Devices II. Purpose/Objective: To provide a policy of coverage
POLICIES AND PROCEDURE MANUAL Policy: MP046 Section: Medical Benefit Policy Subject: Progressive Stretch Devices I. Policy: Progressive Stretch Devices II. Purpose/Objective: To provide a policy of coverage
POLYHEAL MICRO - INSTRUCTIONS FOR USE
 POLYHEAL MICRO - INSTRUCTIONS FOR USE PRODUCT DESCRIPTION PolyHeal Micro is a medical device indicated for the treatment of wounds. It is comprised of a suspension of polystyrene negatively charged microspheres
POLYHEAL MICRO - INSTRUCTIONS FOR USE PRODUCT DESCRIPTION PolyHeal Micro is a medical device indicated for the treatment of wounds. It is comprised of a suspension of polystyrene negatively charged microspheres
Cigna Medical Coverage Policy
 Cigna Medical Coverage Policy Subject Negative Pressure Wound Therapy/Vacuum-Assisted Closure (VAC) for Nonhealing Wounds Table of Contents Coverage Policy... 1 General Background... 2 Coding/Billing Information...
Cigna Medical Coverage Policy Subject Negative Pressure Wound Therapy/Vacuum-Assisted Closure (VAC) for Nonhealing Wounds Table of Contents Coverage Policy... 1 General Background... 2 Coding/Billing Information...
POLICIES AND PROCEDURE MANUAL
 POLICIES AND PROCEDURE MANUAL Policy: MP071 Section: Medical Benefit Policy Subject: Continuous Subcutaneous Glucose Monitor CSGM I. Policy: Continuous Subcutaneous Glucose Monitor (CSGM) II. Purpose/Objective:
POLICIES AND PROCEDURE MANUAL Policy: MP071 Section: Medical Benefit Policy Subject: Continuous Subcutaneous Glucose Monitor CSGM I. Policy: Continuous Subcutaneous Glucose Monitor (CSGM) II. Purpose/Objective:
ISPUB.COM. S Saad, E Shakov, V Sebastian, A Saad INTRODUCTION METHODS CASE REPORT 2 CASE REPORT 3 CASE REPORT 1
 ISPUB.COM The Internet Journal of Surgery Volume 11 Number 1 The use of Wound Vacuum-assisted Closure (V.A.C. ) system in the treatment of Recurrent or Complex Pilonidal Cyst Disease: Experience in 4 Adolescent
ISPUB.COM The Internet Journal of Surgery Volume 11 Number 1 The use of Wound Vacuum-assisted Closure (V.A.C. ) system in the treatment of Recurrent or Complex Pilonidal Cyst Disease: Experience in 4 Adolescent
New 2010 CPT Codes (italic font represents a new or revised code/description)
 New 2010 CPT Codes (italic font represents a new or revised code/description) 14301 Adjacent tissue transfer or rearrangement, any area; defect 30.1 sq cm to 60.0 sq cm 14302 each additional 30.0 sq cm,
New 2010 CPT Codes (italic font represents a new or revised code/description) 14301 Adjacent tissue transfer or rearrangement, any area; defect 30.1 sq cm to 60.0 sq cm 14302 each additional 30.0 sq cm,
Breast debridement and closure cpt
 Breast debridement and closure cpt Close Breast debridement cpt code Medicare Billing Guidelines, Medicare payment and reimbursment, Medicare codes. Here is a list of CPT codes and Diagnoses that are.
Breast debridement and closure cpt Close Breast debridement cpt code Medicare Billing Guidelines, Medicare payment and reimbursment, Medicare codes. Here is a list of CPT codes and Diagnoses that are.
POLICIES AND PROCEDURE MANUAL
 POLICIES AND PROCEDURE MANUAL Policy: MP081 Section: Medical Benefit Policy Subject: Chelation Therapy I. Policy: Chelation Therapy II. Purpose/Objective: To provide a policy of coverage regarding Chelation
POLICIES AND PROCEDURE MANUAL Policy: MP081 Section: Medical Benefit Policy Subject: Chelation Therapy I. Policy: Chelation Therapy II. Purpose/Objective: To provide a policy of coverage regarding Chelation
DMEPOS: hospital beds, bed accessories, and pressurereducing
 ACTION: Final DATE: 07/02/2018 10:03 AM 5160-10-18 DMEPOS: hospital beds, bed accessories, and pressurereducing support surfaces. (A) Definitions and explanations. (1) "Group 1," "group 2," and "group
ACTION: Final DATE: 07/02/2018 10:03 AM 5160-10-18 DMEPOS: hospital beds, bed accessories, and pressurereducing support surfaces. (A) Definitions and explanations. (1) "Group 1," "group 2," and "group
Pressure Injury Definition and Stages
 Program Objective Pressure Injury Definition and Stages Identify the changes to the 2016 NPUAP staging system Changes to the Staging System in 2016 2 Anatomy of the Skin Anatomy of the Skin Largest organ
Program Objective Pressure Injury Definition and Stages Identify the changes to the 2016 NPUAP staging system Changes to the Staging System in 2016 2 Anatomy of the Skin Anatomy of the Skin Largest organ
Excellence in OASIS-C COS-C Prep & OASIS Training
 Excellence in OASIS-C COS-C Prep & OASIS Training Webinar Series - Session 5 April 2, 2014 2:00 3:00PM EST PRESENTER: JOAN L. USHER, BS, RHIA, COS-C, ACE JLU HEALTH RECORD SYSTEMS TEL: (781) 829-9632 FAX:
Excellence in OASIS-C COS-C Prep & OASIS Training Webinar Series - Session 5 April 2, 2014 2:00 3:00PM EST PRESENTER: JOAN L. USHER, BS, RHIA, COS-C, ACE JLU HEALTH RECORD SYSTEMS TEL: (781) 829-9632 FAX:
INFORMED-CONSENT-SKIN GRAFT SURGERY
 INFORMED-CONSENT-SKIN GRAFT SURGERY 2000 American Society of Plastic Surgeons. Purchasers of the Patient Consultation Resource Book are given a limited license to modify documents contained herein and
INFORMED-CONSENT-SKIN GRAFT SURGERY 2000 American Society of Plastic Surgeons. Purchasers of the Patient Consultation Resource Book are given a limited license to modify documents contained herein and
Palmetto Medicare Policy Primer
 Palmetto Medicare Policy Primer Medicare Jurisdiction (JM) NC, SC, WV & VA Application of Skin Substitutes LCD #L36466 Indications Presence of neuropathic diabetic foot ulcer(s) having failed to respond
Palmetto Medicare Policy Primer Medicare Jurisdiction (JM) NC, SC, WV & VA Application of Skin Substitutes LCD #L36466 Indications Presence of neuropathic diabetic foot ulcer(s) having failed to respond
ALLIANCE OF WOUND CARE STAKEHOLDERS. Palmetto GBA Public Meeting Draft LCD on Application of Skin Substitutes (DL36466) October 13, 2015
 ALLIANCE OF WOUND CARE STAKEHOLDERS Palmetto GBA Public Meeting Draft LCD on Application of Skin Substitutes (DL36466) October 13, 2015 1 ALLIANCE OF WOUND CARE Who is the Alliance? STAKEHOLDERS A non-profit
ALLIANCE OF WOUND CARE STAKEHOLDERS Palmetto GBA Public Meeting Draft LCD on Application of Skin Substitutes (DL36466) October 13, 2015 1 ALLIANCE OF WOUND CARE Who is the Alliance? STAKEHOLDERS A non-profit
Skin Deep. Agenda. Burns Wounds Debridement Evaluation and Management Services. Presented by: Mike Strong, SFM The Work Comp Experts.
 Presented by: Mike Strong, SFM The Work Comp Experts Agenda Wounds Debridement Evaluation and Management Services 2 1 Types of First Degree Second Degree Third Degree Rule of 9 Adults Infants Burn Coding
Presented by: Mike Strong, SFM The Work Comp Experts Agenda Wounds Debridement Evaluation and Management Services 2 1 Types of First Degree Second Degree Third Degree Rule of 9 Adults Infants Burn Coding
Corporate Medical Policy
 Corporate Medical Policy Pneumatic Compression Pumps for Treatment of Lymphedema and File Name: Origination: Last CAP Review: Next CAP Review: Last Review: pneumatic_compression_pumps_for _lymphedema_and_venous_ulcers
Corporate Medical Policy Pneumatic Compression Pumps for Treatment of Lymphedema and File Name: Origination: Last CAP Review: Next CAP Review: Last Review: pneumatic_compression_pumps_for _lymphedema_and_venous_ulcers
The Power of a Hydroconductive Wound Dressing with LevaFiber Technology
 The Power of a Hydroconductive Wound Dressing with LevaFiber Technology The first step in healing a chronic wound is to detoxify it by removing slough, necrotic tissue, exudate and bacteria, while keeping
The Power of a Hydroconductive Wound Dressing with LevaFiber Technology The first step in healing a chronic wound is to detoxify it by removing slough, necrotic tissue, exudate and bacteria, while keeping
