MMS Pharmacology Lecture 2. Antirheumatic drugs. Dr Sura Al Zoubi
|
|
|
- Lucy Wilkins
- 5 years ago
- Views:
Transcription
1 MMS Pharmacology Lecture 2 Antirheumatic drugs Dr Sura Al Zoubi
2 Revision
3 Rheumatoid Arthritis Definition (RA): is the most common systemic inflammatory disease characterized by symmetrical inflammation of joints yet may involve other organ systems characterized by Symmetrical joint involvement Inflammation Pain, Swelling,Stiffness in the musculoskeletal system, and damage to joints & connective tissue. Extraarticular involvement Including rheumatoid nodules, vasculitis, eye inflammation, neurologic dysfunction, cardiopulmonary disease, lymphadenopathy, and splenomegaly, can be manifestations of the disease. Although the usual disease course is chronic, some patients will enter a remission spontaneously.
4 Epidemiology Rheumatoid arthritis is estimated to have a prevalence of 1%. does not have any racial predilections It can occur at any age, with increasing prevalence up to the seventh decade of life. Generally more common in females 3:1 First decade and older than 60 1:1 Between years 6:1
5 Pathophysiology Chronic inflammation of the synovial tissue lining the joint capsule results in the proliferation of this tissue. The factors that initiate the inflammatory process are unknown. The inflamed, proliferating synovium characteristic of rheumatoid arthritis is called pannus. The pannus invades the cartilage and eventually the bone surface, producing erosions of bone and cartilage and leading to destruction of the joint.
6
7 The immune system is a complex network of checks and balances designed to discriminate self from non-self (foreign) tissues. It helps rid the body of infectious agents, tumor cells, and products associated with the breakdown of cells. In rheumatoid arthritis, this system no longer can differentiate self from non-self tissues and attacks the synovial tissue and other connective tissues.
8
9 Clinical presentation and diagnosis Symptoms Joint pain and stiffness of more than 6 weeks' duration. May also experience fatigue, weakness, low-grade fever, loss of appetite. Muscle pain, and afternoon fatigue may also be present. Joint deformity is generally seen late in the disease.
10 Signs Tenderness with warmth and swelling over affected joints usually involving hands and feet. Distribution of joint involvement is frequently symmetrical. Rheumatoid nodules may also be present.
11 Desired outcome Primary: improve or maintain functional status Improving quality of life Ultimate achieve complete disease remission controlling disease activity and joint pain maintaining the ability to function in daily activities or work slowing destructive joint changes
12 Treatment Non-pharmacological: 1. Rest: It relieves stress on inflamed joints and prevents further joint destruction. Rest also aids in alleviation of pain. Too much rest and immobility, however, may lead to decreased range of motion and, ultimately, muscle atrophy, and contractures.
13 2. Occupational and physical therapy: provide the patient with skills and exercises necessary to increase or maintain mobility. These disciplines also may provide patients with supportive and adaptive devices such as canes, walkers, and splints. 3. Weight loss: alleviate inflamed joint stress 4. Surgery: Tenosynovectomy, tendon repair, and joint replacements are surgical options for patients with RA.(reserved for patients with severe disease).
14 Pharmacological: Treatment
15
16 NSAIDs may be used for symptomatic relief if needed. They provide relatively rapid improvement in symptoms compared with DMARDs, which may take weeks to months before benefit is seen bridge therapy NSAIDs have no impact on disease progression. NSAID use is associated with an increased risk of gastrointestinal ulcers or hemorrhage, fluid retention, exacerbation of existing hypertension, and decreased renal function in certain patient populations.
17 Corticosteroids Corticosteroids can be used in various ways: controlling symptoms before the onset of action of DMARDs. A burst of corticosteroids can be used in acute flares. Continuous low doses may be adjuncts when DMARDs do not provide adequate disease control. Corticosteroids may be injected into joints and soft tissues to control local inflammation.
18 Corticosteroids Not used as monotherapy here are data to suggest they have disease-modifying activity; however, it is preferable to avoid chronic use when possible to avoid long-term complications. NSAIDs and DMARDs have steroid-sparing properties that permit reductions of corticosteroid doses.
19 Disease Modifying Antirheumatic drugs (DMARDs) DMARDs are used in the treatment of rheumatic arthritis RA and have been shown to slow the course of the disease, induce remission prevent further destruction of the joints and involved tissues.
20 DMARDs When a patient is diagnosed with RA, DMARDs should be started within 3 months to help stop the progression of the disease at the earlier stages. NSAIDs or corticosteroids may also be used for relief of symptoms if needed.
21 Choice of drug No one DMARD is efficacious and safe in every patient, and trials of several different drugs may be necessary. 1. Low disease activity: Monotherapy with DMARDs (methotrexate, leflunomide, hydroxychloroquine, or sulfasalazine) 2. Moderate to high activity or inadequate response to monotherapy: combination DMARD therapy (usually methotrexate based) or use of anti-tnf drugs (adalimumab, certolizumab, etanercept, golimumab, and infliximab) 3. For patients with more established disease: use of other biologic therapies (for example, abatacept, rituximab) can be considered. 4. Pregnant women: Most of these agents are contraindicated for use in pregnant women
22 Methotrexate Used alone or in combination therapy The mainstay of treatment of RA MOA: folic acid antagonist that inhibits cytokine production and purine nucleotide biosynthesis, leading to immunosuppressive and antiinflammatory effects. Onset of action (response): 3-6 weeks After giving the maximum dose, a second DMARDs can be added if there is only partial or no response
23 Side effects: 1. mucosal ulceration 2. nausea. 3. Cytopenias (particularly depression of the WBC count), 4. cirrhosis of the liver 5. acute pneumonia-like syndrome. To reduce side effects, leucovorin (folinic acid) once daily (on off-days) Monitoring: Periodic liver enzyme tests, complete blood count
24 Hydroxychloroquine Used for early, mild RA, often combined with methotrexate (it is also used in the treatment of lupus and malaria). MOA: unknown in autoimmune diseases Onset of action (response): 6 weeks to 6 months. Side effects: (it has less effects on the liver and immune system than other DMARDs) 1. ocular toxicity, including irreversible retinal damage and corneal deposits. 2. CNS disturbances, 3. GI upset 4. skin discoloration and eruptions.
25 Leflunomide It is approved for the treatment of RA. It can be used as monotherapy or in combination with methotrexate MOA: (after biotransformation) Inhibitor of dihydroorotate dehydrogenase (DHODH). Side effects: headache, diarrhea, nausea, weight loss, allergic reactions (flu-like syndrome, skin rash), alopecia, and hypokalemia. It is not recommended in patients with liver disease (risk of hepatotoxicity) Monitoring: signs of infection, CBC, and liver enzymes
26 Minocycline It can be used as monotherapy or in combination with other DMARDs shown to be effective in the treatment of early RA (it is generally not utilized as first-line therapy) MOA: tetracycline antibiotic
27 Biologic agents IL-1 and TNF-α are proinflammatory cytokines involved in the pathogenesis of RA. When secreted by synovial macrophages, IL-1 and TNF-α stimulate synovial cells to proliferate and synthesize collagenase, thereby degrading cartilage, stimulating bone resorption, and inhibiting proteoglycan synthesis. Biologic agents can work on: 1. TNF-α 2. IL-1 3. T-cell 4. B-cell 5. JAK
28 The TNF-α inhibitors (adalimumab, certolizumab, etanercept, golimumab, and infliximab) have been shown to decrease signs and symptoms of RA, reduce progression of structural damage, and improve physical function. Clinical response can be seen within 2 weeks of therapy. As with DMARDs, the decision to continue or stop a biological agent can often be made within 3 months after initiation of therapy.
29 If a patient has failed therapy with one TNF-α inhibitor: 1. Use different TNF-α inhibitor 2. non-tnf biologic therapy (abatacept, rituximab, tocilizumab, tofacitinib) is appropriate. 3. Combination of TNF-α inhibitors with any of the other drugs for RA, except for the non-tnf biologic therapies (due to increased risk of infection). General adverse effects and cautions: 1. Patients receiving TNF-α inhibitors are at increased risk for infections (tuberculosis and sepsis), fungal opportunistic infections, and pancytopenia. 2. Live vaccinations should not be administered while on TNF-α inhibitor therapy. 3. These agents should be used very cautiously in those with heart failure, as they can cause and/or worsen preexisting heart failure. 4. An increased risk of lymphoma and other cancers has been observed with the use of TNF-α inhibitors.
30 Adalimumab Indicated for treatment of moderate to severe RA, either as monotherapy or in combination with methotrexate. It is also indicated for psoriatic arthritis, ankylosing spondylitis, and Crohn disease. MOA: It is a recombinant monoclonal antibody that binds to TNF-α, thereby interfering with endogenous TNF-α activity by blocking its interaction with cell surface receptors. Administration: subcutaneously weekly or every other week. Adverse effects: It may cause headache, nausea, agranulocytosis, rash, reaction at the injection site, or increased risk of infections, such as UTI, URTI, and sinusitis.
31 Certolizumab pegol It has similar indications to adalimumab. MOA: TNF-α blocker and is a potent neutralizer of TNF-α biological actions. Administration: It is combined with polyethylene glycol (pegylated) and is administered every 2 weeks via subcutaneous injection. Adverse effects: similar to other TNF-α inhibitors
32 Etanercept This agent is approved for use in patients with moderate to severe RA, either alone or in combination with methotrexate. It is also approved for use in ankylosing spondylitis and psoriasis. The combination of etanercept and methotrexate is more effective than methotrexate or etanercept alone in retarding the RA disease process, improving function, and achieving remission MOA: It binds to TNF-α, thereby blocking its interaction with cell surface TNFα receptors. Administration: Etanercept is given subcutaneously twice a week. Adverse effects: TNF-α inhibitors, it can increase the risk for infections, malignancy, and new or worsening heart failure.
33 Golimumab It is administered in combination with methotrexate or other nonbiologic DMARDs. MOA: neutralizes the biological activity of TNF-α by binding to it and blocking its interaction with cell surface receptors. Administration: subcutaneously once a month Adverse effects: may increase hepatic enzymes, reactivation of hepatitis B may occur in chronic carriers, may increase the risk of malignancies and serious infections.
34 Infliximab It is approved for use in combination with methotrexate in patients with RA who have had inadequate response to methotrexate monotherapy. This agent is not indicated for monotherapy, as this leads to the development of anti-infliximab antibodies, resulting in reduced efficacy. Additional indications include plaque psoriasis, psoriatic arthritis, ulcerative colitis, ankylosing spondylitis, and Crohn disease MOA: It is a chimeric monoclonal antibody composed of human and murine regions that binds specifically to human TNF-α and inhibits binding with its receptors. Administration: IV infusion every 8 weeks. Adverse effects: Infusion site reactions, such as fever, chills, pruritus, and urticaria,. Infections (for example, pneumonia, cellulitis, and activation of latent tuberculosis), leukopenia, and neutropenia have also been reported.
35 T cells activation T lymphocytes need two interactions to become activated: 1. The antigen-presenting cell (that is, macrophages or B cells) must interact with the receptor on the T cell 2. The CD80/CD86 protein on the antigen-presenting cell must interact with the CD28 protein on the T cell.
36 Abatacept It is indicated for patients with moderate to severe RA who have had an inadequate response to DMARDs or TNF-α inhibitors. MOA: Competes with CD28 for binding on CD80/CD86 protein, thereby preventing full T-cell activation. Administration: IV infusion every 4 weeks. Adverse effects: headache, URTI, nasopharyngitis, and nausea. Concurrent use with TNF-α inhibitors is not recommended due to increased risk of serious infections
37 B cells B lymphocytes are derived from the bone marrow and are necessary for efficient immune response. In RA, however, B cells can perpetuate the inflammatory process in the synovium by: 1. activating T lymphocytes. 2. producing autoantibodies and rheumatoid factor 3. producing proinflammatory cytokines, such as TNF-α and IL-1
38 Rituximab It is indicated for use in combination with methotrexate for patients with moderate to severe RA who have had an inadequate response to TNF-α inhibitors. MOA: monoclonal antibody directed against the CD20 antigen found on the surface of normal and malignant B lymphocytes, resulting in B-cell depletion Administration: IV infusion every 16 to 24 weeks. Adverse reactions: urticaria, hypotension, and angioedema are the most common complaints with this agent and typically occur during the first infusion. (methylprednisolone is administered 30 minutes prior to each infusion)
39 Tocilizumab It can be used as monotherapy or in combination with methotrexate or other nonbiologic DMARDs for patients with moderate to severe RA. MOA: It is a monoclonal antibody that inhibits the actions of IL-6 by blocking the IL-6 receptor. Administration: IV infusion every 4 weeks.
40 Anakinra It is used in moderate to severe RA in patients who have failed one or more DMARDs (leads to a modest reduction in the signs and symptoms) IL-1 is induced by inflammatory stimuli and mediates a variety of immunologic responses, including degradation of cartilage and stimulation of bone resorption. MOA: is an IL-1 receptor antagonist. Adverse effects: it is associated with neutropenia and is infrequently used in the treatment of RA.
41 JAK Janus kinases are intracellular enzymes that modulate immune cell activity in response to the binding of inflammatory mediators to the cellular membrane. Cytokines, growth factors, interferons, ILs, and erythropoietin can lead to an increase in Janus kinase activity and activation of the immune system.
42 Tofacitinib indicated for the treatment of moderate to severe RA in patients who have had an inadequate response or intolerance to methotrexate. MOA: It is an inhibitor of Janus kinases Administration: orally Metabolism of tofacitinib is mediated primarily by CYP3A4, and dosage adjustments may be required if the drug is taken with potent inhibitors or inducers of this isoenzyme.
43 Monitoring: Tofacitinib 1. Hemoglobin concentrations must be greater than 9 g/dl to start tofacitinib and must be monitored during therapy due to the risk for anemia. 2. lymphocyte and neutrophil counts should be checked prior to initiation of therapy and monitored during treatment. Adverse effects: increase the risk for secondary malignancy, opportunistic infections, renal, or hepatic dysfunction.
44 Thank you
45 Assessment of disease activity At each follow up visit, the physician must assess whether the disease is active or inactive a) Degree of joint pain (by visual analog scale) b) Duration of morning stiffness c) Duration of fatigue d) Presence of actively inflamed joints on examination (tender and swollen joint counts) e) Limitation of function
46 Periodically evaluate for disease activity or disease progression a) Evidence of disease progression on physical examination (loss of motion, instability, malalignment, and/or deformity) b) Erythrocyte sedimentation rate or C-reactive protein elevation c) Progression of radiographic damage of involved joints
47 Disease activity assessment
48 DAS 28 DAS stands for "Disease Activity Score" and is a measure of the activity of rheumatoid arthritis. In Europe the DAS is the recognized standard in research and clinical practice. The following parameters are included in the calculation: 1. Number of joints tender to the touch (TEN) 2. Number of swollen joints (SW) 3. Erythrocyte sedimentation rate (ESR) 4. Patient assessment of disease activity (VAS; mm) visual analogue scale
49
Antirheumatic drugs. Rheumatic Arthritis (RA)
 Antirheumatic drugs Rheumatic Arthritis (RA) Disease Modifying Antirheumatic drugs (DMARDs) DMARDs are used in the treatment of rheumatic arthritis RA and have been shown to slow the course of the disease,
Antirheumatic drugs Rheumatic Arthritis (RA) Disease Modifying Antirheumatic drugs (DMARDs) DMARDs are used in the treatment of rheumatic arthritis RA and have been shown to slow the course of the disease,
Rheumatoid arthritis
 Rheumatoid arthritis 1 Definition Rheumatoid arthritis is one of the most common inflammatory disorders affecting the population worldwide. It is a systemic inflammatory disease which affects not only
Rheumatoid arthritis 1 Definition Rheumatoid arthritis is one of the most common inflammatory disorders affecting the population worldwide. It is a systemic inflammatory disease which affects not only
Cimzia. Cimzia (certolizumab pegol) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.50.11 Section: Prescription Drugs Effective Date: April 1, 2018 Subject: Cimzia Page: 1 of 5 Last Review
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.50.11 Section: Prescription Drugs Effective Date: April 1, 2018 Subject: Cimzia Page: 1 of 5 Last Review
Rheumatoid Arthritis. By: Hadi Esmaily (PharmD., BCCP, MBA) Department of Clinical Pharmacy, Shahid Beheshti Medical University
 Rheumatoid Arthritis By: Hadi Esmaily (PharmD., BCCP, MBA) Department of Clinical Pharmacy, Shahid Beheshti Medical University Introduction RA is a Chronic, Systemic, Inflammatory disorder of unknown etiology
Rheumatoid Arthritis By: Hadi Esmaily (PharmD., BCCP, MBA) Department of Clinical Pharmacy, Shahid Beheshti Medical University Introduction RA is a Chronic, Systemic, Inflammatory disorder of unknown etiology
Corporate Medical Policy
 Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: golimumab_simponi 8/2013 2/2018 2/2019 3/2018 Description of Procedure or Service Golimumab (Simponi and
Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: golimumab_simponi 8/2013 2/2018 2/2019 3/2018 Description of Procedure or Service Golimumab (Simponi and
DRAFT. Remission rates, calculated using observed case (OC) analyses were as follows: Year 1 Year 2 Year 3 Year 4 All patients 62.
 DRAFT New Efficacy Data Shows Cimzia (certolizumab pegol) Provides Long-Term Remission of Moderate to Severe Crohn s Disease Regardless of Prior Anti-TNF Exposure, According to Data Presented at DDW Oral
DRAFT New Efficacy Data Shows Cimzia (certolizumab pegol) Provides Long-Term Remission of Moderate to Severe Crohn s Disease Regardless of Prior Anti-TNF Exposure, According to Data Presented at DDW Oral
Cimzia. Cimzia (certolizumab pegol) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.50.11 Subject: Cimzia Page: 1 of 5 Last Review Date: December 8, 2017 Cimzia Description Cimzia (certolizumab
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.50.11 Subject: Cimzia Page: 1 of 5 Last Review Date: December 8, 2017 Cimzia Description Cimzia (certolizumab
What prescribers need to know
 HUMIRA Citrate-free presentations in an Electronic Medical Record (EMR) What prescribers need to know 2 / This is your guide to identifying HUMIRA Citrate-free presentations in your Electronic Medical
HUMIRA Citrate-free presentations in an Electronic Medical Record (EMR) What prescribers need to know 2 / This is your guide to identifying HUMIRA Citrate-free presentations in your Electronic Medical
Cimzia. Cimzia (certolizumab pegol) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Cimzia Page: 1 of 5 Last Review Date: March 17, 2017 Cimzia Description Cimzia (certolizumab pegol)
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Cimzia Page: 1 of 5 Last Review Date: March 17, 2017 Cimzia Description Cimzia (certolizumab pegol)
New Cimzia data, with sites in the US, demonstrate safety and efficacy, increased participation in social activities for adult RA patients
 Long-term Cimzia (certolizumab pegol) data demonstrated rapid sustained improvements in clinical outcomes and quality of life in moderate to severe rheumatoid arthritis (RA) patients New Cimzia data, with
Long-term Cimzia (certolizumab pegol) data demonstrated rapid sustained improvements in clinical outcomes and quality of life in moderate to severe rheumatoid arthritis (RA) patients New Cimzia data, with
Medication Policy Manual. Topic: Xeljanz, tofacitinib Date of Origin: January 21, 2013
 Medication Policy Manual Policy No: dru289 Topic: Xeljanz, tofacitinib Date of Origin: January 21, 2013 Committee Approval Date: January 19, 2015 Next Review Date: January 2016 Effective Date: April 1,
Medication Policy Manual Policy No: dru289 Topic: Xeljanz, tofacitinib Date of Origin: January 21, 2013 Committee Approval Date: January 19, 2015 Next Review Date: January 2016 Effective Date: April 1,
Cimzia (certolizumab pegol) Data Showed Broad and Rapid Relief From Burden of Symptoms In Rheumatoid Arthritis Patients
 Cimzia (certolizumab pegol) Data Showed Broad and Rapid Relief From Burden of Symptoms In Rheumatoid Arthritis Patients Rapid, sustained and clinically meaningful improvement in wideranging patient-reported
Cimzia (certolizumab pegol) Data Showed Broad and Rapid Relief From Burden of Symptoms In Rheumatoid Arthritis Patients Rapid, sustained and clinically meaningful improvement in wideranging patient-reported
Regulatory Status FDA- approved indication: Simponi and Simponi ARIA are tumor necrosis factor (TNF) blockers indicated for the treatment of: (2-3)
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.51 Subject: Simponi / Simponi ARIA Page: 1 of 9 Last Review Date: March 16, 2018 Simponi / Simponi
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.51 Subject: Simponi / Simponi ARIA Page: 1 of 9 Last Review Date: March 16, 2018 Simponi / Simponi
1 P a g e. Systemic Juvenile Idiopathic Arthritis (SJIA) (1.3) Patients 2 years of age and older with active systemic juvenile idiopathic arthritis.
 LENGTH OF AUTHORIZATION: Initial: 3 months for Crohn s or Ulcerative Colitis; 1 year for all other indications. Renewal: 1 year dependent upon medical records supporting response to therapy and review
LENGTH OF AUTHORIZATION: Initial: 3 months for Crohn s or Ulcerative Colitis; 1 year for all other indications. Renewal: 1 year dependent upon medical records supporting response to therapy and review
Treating Rheumatologic Disease in Arizona: Good News, Bad News
 Treating Rheumatologic Disease in Arizona: Good News, Bad News Jeffrey R. Lisse, M.D. Ethel P. McChesney Bilby Professor of Medicine Chief, Section of Rheumatology University of Arizona School of Medicine
Treating Rheumatologic Disease in Arizona: Good News, Bad News Jeffrey R. Lisse, M.D. Ethel P. McChesney Bilby Professor of Medicine Chief, Section of Rheumatology University of Arizona School of Medicine
HELPING YOU AND YOUR PATIENTS TALK OPENLY ABOUT MODERATELY TO SEVERELY ACTIVE RA
 SIMPONI ARIA (golimumab) is indicated for the treatment of adults with moderately to severely active rheumatoid arthritis (RA) in combination with MTX, active psoriatic arthritis, and active ankylosing
SIMPONI ARIA (golimumab) is indicated for the treatment of adults with moderately to severely active rheumatoid arthritis (RA) in combination with MTX, active psoriatic arthritis, and active ankylosing
The Medical Letter. on Drugs and Therapeutics
 The Medical Letter publications are protected by US and international copyright laws. Forwarding, copying or any other distribution of this material is strictly prohibited. For further information call:
The Medical Letter publications are protected by US and international copyright laws. Forwarding, copying or any other distribution of this material is strictly prohibited. For further information call:
Medical Management of Rheumatoid Arthritis (RA)
 Medical Management of Rheumatoid Arthritis (RA) Dr Lee-Suan Teh Rheumatologist Royal Blackburn Hospital Educational objectives ABC Appreciate the epidemiology of RA Be able to diagnosis of RA Competent
Medical Management of Rheumatoid Arthritis (RA) Dr Lee-Suan Teh Rheumatologist Royal Blackburn Hospital Educational objectives ABC Appreciate the epidemiology of RA Be able to diagnosis of RA Competent
PRIOR AUTHORIZATION REQUEST GUIDE
 PRIOR AUTHORIZATION REQUEST GUIDE Drafting a Prior Authorization Request The following information is presented for informational purposes only and is not intended to provide reimbursement or legal advice.
PRIOR AUTHORIZATION REQUEST GUIDE Drafting a Prior Authorization Request The following information is presented for informational purposes only and is not intended to provide reimbursement or legal advice.
Xeljanz. Xeljanz, Xeljanz XR (tofacitinib) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.24 Subject: Xeljanz Page: 1 of 6 Last Review Date: March 16, 2018 Xeljanz Description Xeljanz, Xeljanz
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.24 Subject: Xeljanz Page: 1 of 6 Last Review Date: March 16, 2018 Xeljanz Description Xeljanz, Xeljanz
Coverage Criteria: Express Scripts, Inc. monograph dated 12/15/ months or as otherwise noted by indication
 BENEFIT DESCRIPTION AND LIMITATIONS OF COVERAGE ITEM: PRODUCT LINES: COVERED UNDER: DESCRIPTION: CPT/HCPCS Code: Company Supplying: Setting: Kineret (anakinra subcutaneous injection) Commercial HMO/PPO/CDHP
BENEFIT DESCRIPTION AND LIMITATIONS OF COVERAGE ITEM: PRODUCT LINES: COVERED UNDER: DESCRIPTION: CPT/HCPCS Code: Company Supplying: Setting: Kineret (anakinra subcutaneous injection) Commercial HMO/PPO/CDHP
Stelara. Stelara (ustekinumab) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.90.04 Subject: Stelara Page: 1 of 9 Last Review Date: September 20, 2018 Stelara Description Stelara
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.90.04 Subject: Stelara Page: 1 of 9 Last Review Date: September 20, 2018 Stelara Description Stelara
Cimzia shows duration of response up to week 26 in moderate to severe Crohn s patients who failed the intravenous infusion treatment infliximab.
 C3073-0409 CIMZIA (certolizumab pegol) PROVIDES LONG-TERM REMISSION AND RESPONSE RATES IN INFLIXIMAB-REFRACTORY CROHN S PATIENTS Cimzia shows duration of response up to week 26 in moderate to severe Crohn
C3073-0409 CIMZIA (certolizumab pegol) PROVIDES LONG-TERM REMISSION AND RESPONSE RATES IN INFLIXIMAB-REFRACTORY CROHN S PATIENTS Cimzia shows duration of response up to week 26 in moderate to severe Crohn
Regulatory Status FDA- approved indication: Simponi and Simponi ARIA are tumor necrosis factor (TNF) blockers indicated for the treatment of:
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.51 Subject: Simponi / Simponi ARIA Page: 1 of 8 Last Review Date: March 17, 2017 Simponi / Simponi
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.51 Subject: Simponi / Simponi ARIA Page: 1 of 8 Last Review Date: March 17, 2017 Simponi / Simponi
CIMZIA (certolizumab pegol)
 Pre - PA Allowance None Prior-Approval Requirements Age Diagnoses 18 years of age or older Patient must have ONE of the following: 1. Moderate to severe Crohn s Disease (CD) a. Inadequate response, intolerance
Pre - PA Allowance None Prior-Approval Requirements Age Diagnoses 18 years of age or older Patient must have ONE of the following: 1. Moderate to severe Crohn s Disease (CD) a. Inadequate response, intolerance
Seronegative Arthritis. Dr Mary Gayed 25 th April 2018
 Seronegative Arthritis Dr Mary Gayed 25 th April 2018 Overview Description of the conditions Discussion of symptoms & investigations that may be required Discussion of management and treatment Questions
Seronegative Arthritis Dr Mary Gayed 25 th April 2018 Overview Description of the conditions Discussion of symptoms & investigations that may be required Discussion of management and treatment Questions
Patient #1. Rheumatoid Arthritis. Rheumatoid Arthritis. 45 y/o female Morning stiffness in her joints >1 hour
 Patient #1 Rheumatoid Arthritis Essentials For The Family Medicine Physician 45 y/o female Morning stiffness in her joints >1 hour Hands, Wrists, Knees, Ankles, Feet Polyarticular, symmetrical swelling
Patient #1 Rheumatoid Arthritis Essentials For The Family Medicine Physician 45 y/o female Morning stiffness in her joints >1 hour Hands, Wrists, Knees, Ankles, Feet Polyarticular, symmetrical swelling
Simponi / Simponi ARIA (golimumab)
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.51 Subject: Simponi / Simponi ARIA Page: 1 of 6 Last Review Date: September 15, 2016 Simponi / Simponi
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.51 Subject: Simponi / Simponi ARIA Page: 1 of 6 Last Review Date: September 15, 2016 Simponi / Simponi
INFLIXIMAB Remicade (infliximab), Inflectra (infliximab-dyyb), Ixifi* (infliximabqbtx), Renflexis (infliximab-abda)
 Pre - PA Allowance None Prior-Approval Requirements Diagnoses Patient must have ONE of the following: 6 years of age or older 1. Moderate to severe Crohn s disease (CD) a. Patient has fistulizing disease
Pre - PA Allowance None Prior-Approval Requirements Diagnoses Patient must have ONE of the following: 6 years of age or older 1. Moderate to severe Crohn s disease (CD) a. Patient has fistulizing disease
Rheumatoid Arthritis. Module III
 Rheumatoid Arthritis Module III Management: Biological disease modifying anti-rheumatic drugs, glucocorticoids and special situations (pregnancy & lactation) Dr Ved Chaturvedi MD, DM Senior Consultant
Rheumatoid Arthritis Module III Management: Biological disease modifying anti-rheumatic drugs, glucocorticoids and special situations (pregnancy & lactation) Dr Ved Chaturvedi MD, DM Senior Consultant
Rheumatoid Arthritis
 Rheumatoid Arthritis What is rheumatoid arthritis? Rheumatoid arthritis (RA) is an autoimmune disease that causes chronic inflammation of the joints. Rheumatoid arthritis can also cause inflammation of
Rheumatoid Arthritis What is rheumatoid arthritis? Rheumatoid arthritis (RA) is an autoimmune disease that causes chronic inflammation of the joints. Rheumatoid arthritis can also cause inflammation of
Cyltezo (adalimumab-adbm) CG-DRUG-64, CG-DRUG-65
 Market DC Cyltezo (adalimumab-adbm) CG-DRUG-64, CG-DRUG-65 Override(s) Prior Authorization Quantity Limit Medications Cyltezo (adalimumab-adbm) 40 mg/0.8 ml prefilled syringe #* ^ Approval Duration 1 year
Market DC Cyltezo (adalimumab-adbm) CG-DRUG-64, CG-DRUG-65 Override(s) Prior Authorization Quantity Limit Medications Cyltezo (adalimumab-adbm) 40 mg/0.8 ml prefilled syringe #* ^ Approval Duration 1 year
Cell-Mediated Immunity and T Lymphocytes
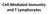 Cell-Mediated Immunity and T Lymphocytes T helper (Th) Cells Peripheral lymhoid tissue thymus Lymphoid stem cell CD8+ CD4+ CD4+ Treg CD8+ CTL + antigen Cytotoxic T lymphocyte CD4+ Th Helper T cell CD4+
Cell-Mediated Immunity and T Lymphocytes T helper (Th) Cells Peripheral lymhoid tissue thymus Lymphoid stem cell CD8+ CD4+ CD4+ Treg CD8+ CTL + antigen Cytotoxic T lymphocyte CD4+ Th Helper T cell CD4+
Rheumatoid Arthritis Update
 Rheumatoid Arthritis Update Beth Valashinas, DO, FACOI, FACR Disclosures Speaker for AbbVie Pharmaceuticals Learning Objectives Upon completion of this session, participants should be able to discuss:
Rheumatoid Arthritis Update Beth Valashinas, DO, FACOI, FACR Disclosures Speaker for AbbVie Pharmaceuticals Learning Objectives Upon completion of this session, participants should be able to discuss:
Using ENBREL to Treat Rheumatoid and Psoriatic Arthritis
 Using ENBREL to Treat Rheumatoid and Psoriatic Arthritis Writing White Papers class Bellevue Community College TABLE OF CONTENTS TABLE OF CONTENTS...2 OVERVIEW...3 RHEUMATOID ARTHRITIS... 3 JUVENILE RHEUMATOID
Using ENBREL to Treat Rheumatoid and Psoriatic Arthritis Writing White Papers class Bellevue Community College TABLE OF CONTENTS TABLE OF CONTENTS...2 OVERVIEW...3 RHEUMATOID ARTHRITIS... 3 JUVENILE RHEUMATOID
Announcing HUMIRA. Psoriasis Starter Package
 Announcing HUMIRA (adalimumab) Psoriasis Starter Package HUMIRA is indicated for the treatment of adult patients with moderate to severe chronic plaque psoriasis who are candidates for systemic therapy
Announcing HUMIRA (adalimumab) Psoriasis Starter Package HUMIRA is indicated for the treatment of adult patients with moderate to severe chronic plaque psoriasis who are candidates for systemic therapy
Corporate Medical Policy
 Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: abatacept_orencia 4/2008 2/2018 2/2019 2/2018 Description of Procedure or Service Abatacept (Orencia ), a
Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: abatacept_orencia 4/2008 2/2018 2/2019 2/2018 Description of Procedure or Service Abatacept (Orencia ), a
Treatment of Rheumatoid Arthritis: The Past, the Present and the Future
 Treatment of Rheumatoid Arthritis: The Past, the Present and the Future Lai-Ling Winchow FCP(SA) Cert Rheum(SA) Chris Hani Baragwanath Academic Hospital University of the Witwatersrand Outline of presentation
Treatment of Rheumatoid Arthritis: The Past, the Present and the Future Lai-Ling Winchow FCP(SA) Cert Rheum(SA) Chris Hani Baragwanath Academic Hospital University of the Witwatersrand Outline of presentation
Pharmacy Management Drug Policy
 SUBJECT: Cimzia (Certolizumab pegol) - for Ankylosing Spondylitis, Crohn s Disease, Psoriatic Arthritis and Rheumatoid Arthritis POLICY NUMBER: PHARMACY-07 EFFECTIVE DATE: 5/2009 LAST REVIEW DATE: 6/13/2018
SUBJECT: Cimzia (Certolizumab pegol) - for Ankylosing Spondylitis, Crohn s Disease, Psoriatic Arthritis and Rheumatoid Arthritis POLICY NUMBER: PHARMACY-07 EFFECTIVE DATE: 5/2009 LAST REVIEW DATE: 6/13/2018
RHEUMATOID ARTHRITIS DRUGS
 Rheumatology Biologics Criteria from the Exceptional Access Program RHEUMATOID ARTHRITIS DRUGS DRUG NAME BRS REIMBURSED DOSAGE FORM/ STRENGTH Adalimumab Humira 40 mg/0.8 syringe and 40mg/0.8 pen for Anakinra
Rheumatology Biologics Criteria from the Exceptional Access Program RHEUMATOID ARTHRITIS DRUGS DRUG NAME BRS REIMBURSED DOSAGE FORM/ STRENGTH Adalimumab Humira 40 mg/0.8 syringe and 40mg/0.8 pen for Anakinra
Horizon Scanning Centre November Secukinumab for active and progressive psoriatic arthritis. SUMMARY NIHR HSC ID: 5330
 Horizon Scanning Centre November 2012 Secukinumab for active and progressive psoriatic arthritis. SUMMARY NIHR HSC ID: 5330 Secukinumab is a high-affinity fully human monoclonal antibody that antagonises
Horizon Scanning Centre November 2012 Secukinumab for active and progressive psoriatic arthritis. SUMMARY NIHR HSC ID: 5330 Secukinumab is a high-affinity fully human monoclonal antibody that antagonises
1. Background: Infliximab is administered parenterally; therefore, it is not covered under retail pharmacy benefits.
 Subject: Infliximab (Remicade ) Original Original Committee Approval: October 13, 2006 Revised Last Committee Approval: December 3, 2008 Last Review: October 19, 2007 1. Background: Infliximab is a genetically
Subject: Infliximab (Remicade ) Original Original Committee Approval: October 13, 2006 Revised Last Committee Approval: December 3, 2008 Last Review: October 19, 2007 1. Background: Infliximab is a genetically
Rheumatoid Arthritis. Improving Outcomes in RA: Three Pillars. RA: Chronic Joint Destruction and Disability What We Try to Prevent
 Rheumatoid Arthritis Modern Management of Common Problems in Rheumatology: Rheumatoid Arthritis Jonathan Graf, M.D. Associate Professor of Medicine, UCSF Division of Rheumatology, SFGH Director, UCSF Rheumatoid
Rheumatoid Arthritis Modern Management of Common Problems in Rheumatology: Rheumatoid Arthritis Jonathan Graf, M.D. Associate Professor of Medicine, UCSF Division of Rheumatology, SFGH Director, UCSF Rheumatoid
Cosentyx. Cosentyx (secukinumab) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.90.11 Subject: Cosentyx Page: 1 of 7 Last Review Date: September 20, 2018 Cosentyx Description Cosentyx
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.90.11 Subject: Cosentyx Page: 1 of 7 Last Review Date: September 20, 2018 Cosentyx Description Cosentyx
Infliximab/Infliximab-dyyb DRUG.00002
 Infliximab/Infliximab-dyyb DRUG.00002 Override(s) Prior Authorization Step Therapy Medications Remicade (infliximab) Inflectra (inflectra-dyyb) Approval Duration 1 year Comment Intravenous administration
Infliximab/Infliximab-dyyb DRUG.00002 Override(s) Prior Authorization Step Therapy Medications Remicade (infliximab) Inflectra (inflectra-dyyb) Approval Duration 1 year Comment Intravenous administration
Remicade. Remicade (infliximab), Inflectra (infliximab-dyyb) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.50.02 Subsection: Gastrointestinal nts Original Policy Date: May 20, 2011 Subject: Remicade Page: 1 of
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.50.02 Subsection: Gastrointestinal nts Original Policy Date: May 20, 2011 Subject: Remicade Page: 1 of
Actemra (tocilizumab) CG-DRUG-81
 Market DC Actemra (tocilizumab) CG-DRUG-81 Override(s) Prior Authorization Approval Duration 1 year Medications Line of Business Quantity Limit Actemra (tocilizumab) vials VA MCD and All L-AGP May be subject
Market DC Actemra (tocilizumab) CG-DRUG-81 Override(s) Prior Authorization Approval Duration 1 year Medications Line of Business Quantity Limit Actemra (tocilizumab) vials VA MCD and All L-AGP May be subject
Biologics for Autoimmune Diseases
 Biologics for Autoimmune Diseases Goal(s): Restrict use of biologics to OHP funded conditions and according to OHP guidelines for use. Promote use that is consistent with national clinical practice guidelines
Biologics for Autoimmune Diseases Goal(s): Restrict use of biologics to OHP funded conditions and according to OHP guidelines for use. Promote use that is consistent with national clinical practice guidelines
Understanding Rheumatoid Arthritis
 Understanding Rheumatoid Arthritis Contents Introduction...3 What is rheumatoid arthritis?...4 Symptoms of rheumatoid arthritis... 5 What causes rheumatoid arthritis?... 10 The role of genes and family
Understanding Rheumatoid Arthritis Contents Introduction...3 What is rheumatoid arthritis?...4 Symptoms of rheumatoid arthritis... 5 What causes rheumatoid arthritis?... 10 The role of genes and family
Xeljanz (tofacitinib), Xeljanz XR (tofacitinib extended-release)
 Market DC Xeljanz (tofacitinib), Xeljanz XR (tofacitinib extended-release) Override(s) Prior Authorization Quantity Limit Medications Xeljanz (tofacitinib) Approval Duration 1 year Quantity Limit May be
Market DC Xeljanz (tofacitinib), Xeljanz XR (tofacitinib extended-release) Override(s) Prior Authorization Quantity Limit Medications Xeljanz (tofacitinib) Approval Duration 1 year Quantity Limit May be
Subject: Remicade (Page 1 of 5)
 Subject: Remicade (Page 1 of 5) Objective: I. To ensure that Health Share/Tuality Health Alliance (THA) has a process by which the appropriate utilization of Remicade (Infliximab) for members whose diagnosis
Subject: Remicade (Page 1 of 5) Objective: I. To ensure that Health Share/Tuality Health Alliance (THA) has a process by which the appropriate utilization of Remicade (Infliximab) for members whose diagnosis
Remicade. Remicade (infliximab), Inflectra (infliximab-dyyb) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Remicade Page: 1 of 9 Last Review Date: June 22, 2017 Remicade Description Remicade (infliximab),
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Remicade Page: 1 of 9 Last Review Date: June 22, 2017 Remicade Description Remicade (infliximab),
Pharmacy Prior Authorization
 Pharmacy Prior Authorization AETA BETTER HEALTH PESLVAIA & AETA BETTER HEALTH KIDS Humira (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information,
Pharmacy Prior Authorization AETA BETTER HEALTH PESLVAIA & AETA BETTER HEALTH KIDS Humira (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information,
To help you with terms and abbreviations used in this document that may be unfamiliar to you, a glossary is provided on the last pages.
 ARTHRITIS CONSUMER EXPERTS 910B RICHARDS STREET VANCOUVER BC V6B 3C1 CANADA T: 604.974-1366 F: 604.974-1377 WWW.ARTHRITISCONSUMEREXPERTS.ORG Arthritis Consumer Experts In Health Care and Research Decision-making
ARTHRITIS CONSUMER EXPERTS 910B RICHARDS STREET VANCOUVER BC V6B 3C1 CANADA T: 604.974-1366 F: 604.974-1377 WWW.ARTHRITISCONSUMEREXPERTS.ORG Arthritis Consumer Experts In Health Care and Research Decision-making
Olumiant (baricitinib) tablets
 READ THIS FOR SAFE AND EFFECTIVE USE OF YOUR MEDICINE PATIENT MEDICATION INFORMATION OLUMIANTTM (Ō-loo'-mē-ant) baricitinib tablets Read this carefully before you start taking OLUMIANT and each time you
READ THIS FOR SAFE AND EFFECTIVE USE OF YOUR MEDICINE PATIENT MEDICATION INFORMATION OLUMIANTTM (Ō-loo'-mē-ant) baricitinib tablets Read this carefully before you start taking OLUMIANT and each time you
Pharmacy Prior Authorization
 Pharmacy Prior Authorization MERC CARE PLA (MEDICAID) Humira (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information, sign and date. Fax
Pharmacy Prior Authorization MERC CARE PLA (MEDICAID) Humira (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information, sign and date. Fax
Abatacept (Orencia) for active rheumatoid arthritis. August 2009
 Abatacept (Orencia) for active rheumatoid arthritis August 2009 This technology summary is based on information available at the time of research and a limited literature search. It is not intended to
Abatacept (Orencia) for active rheumatoid arthritis August 2009 This technology summary is based on information available at the time of research and a limited literature search. It is not intended to
Otezla. Otezla (apremilast) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Otezla Page: 1 of 5 Last Review Date: March 16, 2018 Otezla Description Otezla (apremilast) Background
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Otezla Page: 1 of 5 Last Review Date: March 16, 2018 Otezla Description Otezla (apremilast) Background
Remicade (Infliximab)
 Remicade (Infliximab) Policy Number: Original Effective Date: MM.04.016 11/18/2003 Line(s) of Business: Current Effective Date: HMO; PPO; QUEST Integration 07/26/2013 Section: Prescription Drugs Place(s)
Remicade (Infliximab) Policy Number: Original Effective Date: MM.04.016 11/18/2003 Line(s) of Business: Current Effective Date: HMO; PPO; QUEST Integration 07/26/2013 Section: Prescription Drugs Place(s)
Humira (adalimumab) DRUG.00002
 Humira (adalimumab) DRUG.00002 Override(s) Prior Authorization Quantity Limit Approval Duration 1 year Medications Humira 10 mg/0.2 ml syringe Humira pediatric Crohn s Disease starter pack 40 mg/0.8 ml
Humira (adalimumab) DRUG.00002 Override(s) Prior Authorization Quantity Limit Approval Duration 1 year Medications Humira 10 mg/0.2 ml syringe Humira pediatric Crohn s Disease starter pack 40 mg/0.8 ml
Rheumatoid Arthritis Update. Whitney White, Pharm.D., BCPS Associate Professor of Pharmacy Practice Samford University McWhorter School of Pharmacy
 Rheumatoid Arthritis Update Whitney White, Pharm.D., BCPS Associate Professor of Pharmacy Practice Samford University McWhorter School of Pharmacy Disclosures I declare that neither I nor any member of
Rheumatoid Arthritis Update Whitney White, Pharm.D., BCPS Associate Professor of Pharmacy Practice Samford University McWhorter School of Pharmacy Disclosures I declare that neither I nor any member of
Dr Daniel Ching. Rheumatology Therapeutic Clinical Trials Centre Timaru
 Dr Daniel Ching Rheumatology Therapeutic Clinical Trials Centre Timaru Therapeutic Advances in Rheumatology GP CME Meeting, Dunedin, 18.08.2013 Dr Daniel Ching, MB FRCP FRACP Consultant Rheumatologist,
Dr Daniel Ching Rheumatology Therapeutic Clinical Trials Centre Timaru Therapeutic Advances in Rheumatology GP CME Meeting, Dunedin, 18.08.2013 Dr Daniel Ching, MB FRCP FRACP Consultant Rheumatologist,
London, 1 June 2006 Product name: REMICADE Procedure number: Remicade-H-240-II-73-AR SCIENTIFIC DISCUSSION 1/8
 London, 1 June 2006 Product name: REMICADE Procedure number: Remicade-H-240-II-73-AR SCIENTIFIC DISCUSSION 1/8 1. Introduction Infliximab is a chimeric human-murine IgG1κ monoclonal antibody, which binds
London, 1 June 2006 Product name: REMICADE Procedure number: Remicade-H-240-II-73-AR SCIENTIFIC DISCUSSION 1/8 1. Introduction Infliximab is a chimeric human-murine IgG1κ monoclonal antibody, which binds
Pfizer Announces Modification To Ongoing Tofacitinib FDA Post- Marketing Requirement Study In Patients With Rheumatoid Arthritis
 February 19, 2019 Media Contact: Neha Wadhwa M: +1 212-733-2835 E: Neha.Wadhwa@pfizer.com Investor Contact: Charles Triano O: +1 212-733-3901 E: Charles.E.Triano@pfizer.com Pfizer Announces Modification
February 19, 2019 Media Contact: Neha Wadhwa M: +1 212-733-2835 E: Neha.Wadhwa@pfizer.com Investor Contact: Charles Triano O: +1 212-733-3901 E: Charles.E.Triano@pfizer.com Pfizer Announces Modification
Salicylates: Interactions 10/14/2009. Salicylates DRUGS USED IN THE MANAGEMENT OF MUSCULOSKELETAL DISORDERS. Chapters 17, 18, 34 & Pages 577 &
 DRUGS USED IN THE MANAGEMENT OF MUSCULOSKELETAL DISORDERS Chapters 17, 18, 34 & Pages 577 & 579-586 Salicylates aspirin Have analgesic, antipyretic, and anti-inflammatory effects. Inhibits the production
DRUGS USED IN THE MANAGEMENT OF MUSCULOSKELETAL DISORDERS Chapters 17, 18, 34 & Pages 577 & 579-586 Salicylates aspirin Have analgesic, antipyretic, and anti-inflammatory effects. Inhibits the production
NIHR Innovation Observatory Evidence Briefing: November 2017
 NIHR Innovation Observatory Evidence Briefing: November 2017 Upadacitinib for adults with moderate to severe active rheumatoid arthritis after conventional synthetic disease-modifying anti-rheumatic drugs
NIHR Innovation Observatory Evidence Briefing: November 2017 Upadacitinib for adults with moderate to severe active rheumatoid arthritis after conventional synthetic disease-modifying anti-rheumatic drugs
Regional vs. Systemic Therapy. Corticosteroids. Regional vs. Systemic Therapy for Uveitis. Considerations
 Regional vs. Systemic Therapy for Uveitis Nisha Acharya,, M.D., M.S. Director, Uveitis Service F.I. Proctor Foundation University of California, San Francisco December 4, 2010 No financial disclosures
Regional vs. Systemic Therapy for Uveitis Nisha Acharya,, M.D., M.S. Director, Uveitis Service F.I. Proctor Foundation University of California, San Francisco December 4, 2010 No financial disclosures
Drugs and Applicable Coding: J-code: Enbrel-J1438; Humira-J0135; Remicade-J1745; Inflectra-Q5102; Cimzia-J0718; Simponi-J1602 Renflexis - pending
 Policy Subject: Anti-TNF Agents Policy Number: SHS PBD16 Category: Rheumatology & Autoimmune Policy Type: Medical Pharmacy Department: Pharmacy Product (check all that apply): Group HMO/POS Individual
Policy Subject: Anti-TNF Agents Policy Number: SHS PBD16 Category: Rheumatology & Autoimmune Policy Type: Medical Pharmacy Department: Pharmacy Product (check all that apply): Group HMO/POS Individual
Regulatory Status FDA-approved indication: Humira and Amjevita are tumor necrosis factor (TNF) blockers indicated for the treatment of: (2-3)
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.29 Subject: Humira Page: 1 of 10 Last Review Date: June 22, 2017 Humira Description Humira (adalimumab),
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.29 Subject: Humira Page: 1 of 10 Last Review Date: June 22, 2017 Humira Description Humira (adalimumab),
Center for Evidence-based Policy
 P&T Committee Brief Targeted Immune Modulators: Comparative Drug Class Review Alison Little, MD Center for Evidence-based Policy Oregon Health & Science University 3455 SW US Veterans Hospital Road, SN-4N
P&T Committee Brief Targeted Immune Modulators: Comparative Drug Class Review Alison Little, MD Center for Evidence-based Policy Oregon Health & Science University 3455 SW US Veterans Hospital Road, SN-4N
Psoriatic Arthritis- Secondary Care
 Psoriatic Arthritis- Secondary Care Our Psoriatic Arthritis: First Line Treatments information sheet gives information on the treatments that can be prescribed by a GP, or that might be prescribed if the
Psoriatic Arthritis- Secondary Care Our Psoriatic Arthritis: First Line Treatments information sheet gives information on the treatments that can be prescribed by a GP, or that might be prescribed if the
Pain or stiffness in joints after periods of inactivity or excessive use
 Arthritis Awareness* Some older adults call it Arthur ; others refer to it as their constant compassion, but most describe it as extremely painful Arthritis is a chronic joint disease It is commonly believed
Arthritis Awareness* Some older adults call it Arthur ; others refer to it as their constant compassion, but most describe it as extremely painful Arthritis is a chronic joint disease It is commonly believed
Amjevita (adalimumab-atto) CG-DRUG-64, CG-DRUG-65
 Market DC Amjevita (adalimumab-atto) CG-DRUG-64, CG-DRUG-65 Override(s) Prior Authorization Quantity Limit Medications Amjevita 20 mg/0.4 ml prefilled syringe Amjevita (adalimumab-atto) 40 mg/0.8 ml 2
Market DC Amjevita (adalimumab-atto) CG-DRUG-64, CG-DRUG-65 Override(s) Prior Authorization Quantity Limit Medications Amjevita 20 mg/0.4 ml prefilled syringe Amjevita (adalimumab-atto) 40 mg/0.8 ml 2
Remicade (infliximab) DRUG.00002
 Applicability/Effective Date *- Florida Healthy Kids Remicade (infliximab) DRUG.00002 Override(s) Prior Authorization Step Therapy Medications Remicade (infliximab) Approval Duration 1 year Comment Intravenous
Applicability/Effective Date *- Florida Healthy Kids Remicade (infliximab) DRUG.00002 Override(s) Prior Authorization Step Therapy Medications Remicade (infliximab) Approval Duration 1 year Comment Intravenous
3. Has the patient shown improvement in signs and symptoms of the disease? Y N
 Pharmacy Prior Authorization MERC CARE (MEDICAID) Renflexis (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information, sign and date. Fax
Pharmacy Prior Authorization MERC CARE (MEDICAID) Renflexis (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information, sign and date. Fax
CDEC FINAL RECOMMENDATION
 CDEC FINAL RECOMMENDATION TOFACITINIB (Xeljanz Pfizer Canada Inc.) Indication: Rheumatoid Arthritis Recommendation: The Canadian Drug Expert Committee (CDEC) recommends that tofacitinib be listed, in combination
CDEC FINAL RECOMMENDATION TOFACITINIB (Xeljanz Pfizer Canada Inc.) Indication: Rheumatoid Arthritis Recommendation: The Canadian Drug Expert Committee (CDEC) recommends that tofacitinib be listed, in combination
Amjevita (adalimumab-atto)
 *- Florida Healthy Kids Amjevita (adalimumab-atto) Override(s) Prior Authorization Quantity Limit Medications Amjevita 20 mg/0.4 ml prefilled syringe Amjevita (adalimumab-atto) 40 mg/0.8 ml 2 #* ^ prefilled
*- Florida Healthy Kids Amjevita (adalimumab-atto) Override(s) Prior Authorization Quantity Limit Medications Amjevita 20 mg/0.4 ml prefilled syringe Amjevita (adalimumab-atto) 40 mg/0.8 ml 2 #* ^ prefilled
Actemra. Actemra (tocilizumab) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.12 Subject: Actemra Page: 1 of 13 Last Review Date: September 20, 2018 Actemra Description Actemra
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.12 Subject: Actemra Page: 1 of 13 Last Review Date: September 20, 2018 Actemra Description Actemra
DERBYSHIRE JOINT AREA PRESCRIBING COMMITTEE (JAPC)
 DERBYSHIRE JOINT AREA PRERIBING COMMITTEE (JAPC) Derbyshire commissioning guidance on biologic drugs f the treatment of Rheumatoid arthritis with methotrexate This algithm is a tool to aid the implementation
DERBYSHIRE JOINT AREA PRERIBING COMMITTEE (JAPC) Derbyshire commissioning guidance on biologic drugs f the treatment of Rheumatoid arthritis with methotrexate This algithm is a tool to aid the implementation
Dr. Lyubomir Marinchev Chief of Rheumatology Department, MHAT SOFIAMED, Sofia, Bulgaria
 Dr. Lyubomir Marinchev Chief of Rheumatology Department, MHAT SOFIAMED, Sofia, Bulgaria Inter-Balkan meeting Open the frontiers and exchange of experiences, 27 th April 2013, Rhodes, Greece Patients with
Dr. Lyubomir Marinchev Chief of Rheumatology Department, MHAT SOFIAMED, Sofia, Bulgaria Inter-Balkan meeting Open the frontiers and exchange of experiences, 27 th April 2013, Rhodes, Greece Patients with
Certolizumab pegol (Cimzia) for psoriatic arthritis second line
 Certolizumab pegol (Cimzia) for psoriatic arthritis second line This technology summary is based on information available at the time of research and a limited literature search. It is not intended to
Certolizumab pegol (Cimzia) for psoriatic arthritis second line This technology summary is based on information available at the time of research and a limited literature search. It is not intended to
ADALIMUMAB Generic Brand HICL GCN Exception/Other ADALIMUMAB HUMIRA GUIDELINES FOR USE INITIAL CRITERIA (NOTE: FOR RENEWAL CRITERIA SEE BELOW)
 Generic Brand HICL GCN Exception/Other ADALIMUMAB HUMIRA 24800 GUIDELINES FOR USE INITIAL CRITERIA (NOTE: FOR RENEWAL CRITERIA SEE BELOW) 1. Does the patient have a diagnosis of moderate to severe rheumatoid
Generic Brand HICL GCN Exception/Other ADALIMUMAB HUMIRA 24800 GUIDELINES FOR USE INITIAL CRITERIA (NOTE: FOR RENEWAL CRITERIA SEE BELOW) 1. Does the patient have a diagnosis of moderate to severe rheumatoid
Inflectra (infliximab-dyyb), Remicade (infliximab), Renflexis (infliximab-abda) DRUG CG-DRUG-64
 Inflectra (infliximab-dyyb), Remicade (infliximab), Renflexis (infliximab-abda) DRUG.00002 CG-DRUG-64 Override(s) Prior Authorization *Washington Medicaid See State Specific Mandates Medications Inflectra
Inflectra (infliximab-dyyb), Remicade (infliximab), Renflexis (infliximab-abda) DRUG.00002 CG-DRUG-64 Override(s) Prior Authorization *Washington Medicaid See State Specific Mandates Medications Inflectra
Medication Policy Manual. Topic: Otezla, apremilast Date of Origin: May 9, 2014
 Medication Policy Manual Policy No: dru342 Topic: Otezla, apremilast Date of Origin: May 9, 2014 Committee Approval Date: January 19, 2015 Next Review Date: January 2016 Effective Date: April 1, 2015 IMPORTANT
Medication Policy Manual Policy No: dru342 Topic: Otezla, apremilast Date of Origin: May 9, 2014 Committee Approval Date: January 19, 2015 Next Review Date: January 2016 Effective Date: April 1, 2015 IMPORTANT
ACTEMRA (tocilizumab)
 Pre - PA Allowance None Prior-Approval Requirements Diagnoses Patient must have ONE of the following: 1. Active Polyarticular Juvenile Idiopathic Arthritis (PJIA) b. Patient has an intolerance or has experienced
Pre - PA Allowance None Prior-Approval Requirements Diagnoses Patient must have ONE of the following: 1. Active Polyarticular Juvenile Idiopathic Arthritis (PJIA) b. Patient has an intolerance or has experienced
(tofacitinib) are met.
 Xeljanz (tofacitinib) Policy Number: 5.01. 560 Origination: 3/2014 Last Review: 3/2014 Next Review: 3/2015 Policy BCBSKC will provide coverage for Xeljanz (tofacitinib) when it is determined to be medically
Xeljanz (tofacitinib) Policy Number: 5.01. 560 Origination: 3/2014 Last Review: 3/2014 Next Review: 3/2015 Policy BCBSKC will provide coverage for Xeljanz (tofacitinib) when it is determined to be medically
Anti-Tumor Necrosis Factor (Anti-TNF)
 Anti-Tumor Necrosis Factor (Anti-TNF) 110465 Anti-TNF.indd 1 9/20/16 9:01 AM s About your medicine Anti-tumor necrosis factor (anti-tnf) is a type of medicine called biologic agent that targets substance
Anti-Tumor Necrosis Factor (Anti-TNF) 110465 Anti-TNF.indd 1 9/20/16 9:01 AM s About your medicine Anti-tumor necrosis factor (anti-tnf) is a type of medicine called biologic agent that targets substance
Understanding Rheumatoid Arthritis
 Understanding Rheumatoid Arthritis Understanding Rheumatoid Arthritis What Is Rheumatoid Arthritis? 1,2 Rheumatoid arthritis (RA) is a chronic autoimmune disease. It causes joints to swell and can result
Understanding Rheumatoid Arthritis Understanding Rheumatoid Arthritis What Is Rheumatoid Arthritis? 1,2 Rheumatoid arthritis (RA) is a chronic autoimmune disease. It causes joints to swell and can result
The Rheumatoid Hand Deformities & Management. Dr. Anirudh Sharma Resident Department of Orthopedics
 + The Rheumatoid Hand Deformities & Management Dr. Anirudh Sharma Resident Department of Orthopedics + Why is Rheumatoid Arthritis important? + RA is a very debilitating disease median life expectancy
+ The Rheumatoid Hand Deformities & Management Dr. Anirudh Sharma Resident Department of Orthopedics + Why is Rheumatoid Arthritis important? + RA is a very debilitating disease median life expectancy
CIMZIA (certolizumab pegol)
 CIMZIA (certolizumab pegol) Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services, procedures, medical devices
CIMZIA (certolizumab pegol) Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services, procedures, medical devices
Inflectra infliximab-dyyb. INFLECTRA (infliximab-dyyb) for injection available in the US 1
 First FDA-approved mab biosimilar 1 INFLECTRA (infliximab-dyyb) for injection available in the US 1 INFLECTRA is biosimilar* to and has the same recommended dosage as Remicade (infliximab) 2 * Biosimilar
First FDA-approved mab biosimilar 1 INFLECTRA (infliximab-dyyb) for injection available in the US 1 INFLECTRA is biosimilar* to and has the same recommended dosage as Remicade (infliximab) 2 * Biosimilar
Regulatory Status FDA-approved indication: Orencia is a selective T cell costimulation modulator indicated for: (1)
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.18 Subject: Orencia Page: 1 of 8 Last Review Date: March 16, 2018 Orencia Description Orencia (abatacept)
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.18 Subject: Orencia Page: 1 of 8 Last Review Date: March 16, 2018 Orencia Description Orencia (abatacept)
INFLIXIMAB Remicade (infliximab), Inflectra (infliximab-dyyb), Renflexis (infliximab-abda)
 RATIONALE FOR INCLUSION IN PA PROGRAM Background Remicade, Renflexis and Inflectra are tumor necrosis factor (TNFα) blockers. Tumor necrosis factor is an endogenous protein that regulates a number of physiologic
RATIONALE FOR INCLUSION IN PA PROGRAM Background Remicade, Renflexis and Inflectra are tumor necrosis factor (TNFα) blockers. Tumor necrosis factor is an endogenous protein that regulates a number of physiologic
Rheumatoid Arthritis. Rheumatoid Arthritis. RCS 6080 Medical and Psychosocial Aspects of Rehabilitation Counseling. Rheumatic Diseases
 RCS 6080 Medical and Psychosocial Aspects of Rehabilitation Counseling Rheumatic Diseases The prevalence of rheumatoid arthritis in most Caucasian populations approaches 1% among adults 18 and over and
RCS 6080 Medical and Psychosocial Aspects of Rehabilitation Counseling Rheumatic Diseases The prevalence of rheumatoid arthritis in most Caucasian populations approaches 1% among adults 18 and over and
Immunosuppressants. Assistant Prof. Dr. Najlaa Saadi PhD Pharmacology Faculty of Pharmacy University of Philadelphia
 Immunosuppressants Assistant Prof. Dr. Najlaa Saadi PhD Pharmacology Faculty of Pharmacy University of Philadelphia Immunosuppressive Agents Very useful in minimizing the occurrence of exaggerated or inappropriate
Immunosuppressants Assistant Prof. Dr. Najlaa Saadi PhD Pharmacology Faculty of Pharmacy University of Philadelphia Immunosuppressive Agents Very useful in minimizing the occurrence of exaggerated or inappropriate
Regulatory Status FDA-approved indication: Orencia is a selective T cell co-stimulation modulator indicated for: (1)
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Orencia Page: 1 of 9 Last Review Date: September 20, 2018 Orencia Description Orencia (abatacept)
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Orencia Page: 1 of 9 Last Review Date: September 20, 2018 Orencia Description Orencia (abatacept)
Corporate Medical Policy
 Corporate Medical Policy Rituximab for the Treatment of Rheumatoid Arthritis File Name: Origination: Last CAP Review: Next CAP Review: Last Review: rituximab_for_the_treatment_of_rheumatoid_arthritis 4/2008
Corporate Medical Policy Rituximab for the Treatment of Rheumatoid Arthritis File Name: Origination: Last CAP Review: Next CAP Review: Last Review: rituximab_for_the_treatment_of_rheumatoid_arthritis 4/2008
