Benlysta (belimumab) Prior Authorization Criteria Program Summary
|
|
|
- Jordan Parrish
- 6 years ago
- Views:
Transcription
1 Benlysta (belimumab) Prior Authorization Criteria Program Summary This prior authorization applies to Commercial, NetResults A series, NetResults F series and Health Insurance Marketplace formularies. This policy only targets subcutaneous Benlysta. OBJECTIVE The intent of the Benlysta (belimumab) Prior Authorization (PA) program is to ensure appropriate selection of patients for treatment according to product labeling and/or clinical studies and/or guidelines and according to dosing recommended in product labeling. The program will consider belimumab appropriate for adult patients with active, autoantibody and/or anti-dsdna positive, systemic lupus erythematosus (SLE) who are receiving standard of care therapy. The criteria will not approve belimumab for patients with the following: severe active lupus nephritis or severe active central nervous system lupus, current therapy with other biologic agents or intravenous cyclophosphamide, or current therapy for treatment of a chronic infection. The initial criteria will also allow for a patient who has any FDA approved diagnosis that is not already addressed in the criteria set with a requested dosage that is within FDA limits. Renewal criteria will have similar requirements to initial criteria with the exception that the patient will have to have improvement or stabilization in at least one SLE diagnostic criterion. The dose of belimumab will be limited to the FDA labeled dose. TARGET AGENT Benlysta (belimumab) Brand (generic) GPI Multisource Code Benlysta (belimumab) 200 mg/ml autoinjector D520 M, N, O, or Y 200 mg/ml prefilled syringe E520 M, N, O, or Y CRITERIA F APPROVAL Initial Evaluation Benlysta (belimumab) will be approved when ALL of the following are met: 1. ONE of the following: i. ALL of the following: a. The patient has a diagnosis of active systemic lupus erythematosus (SLE) disease b. ONE of the following: 1. The patient has a history of positive autoantibody test results [positive antinuclear antibody (ANA >1:80) and/or anti-dsdna (>30 IU/mL)] 2. The patient has a history of at least 4 SLE related disease manifestations (SLE related disease manifestations include, but are not limited to: malar rash, discoid rash, photosensitivity, oral ulcers, nonerosive arthritis, pleuritis/pericarditis, renal disorder [persistent proteinuria >0.5 grams/day or cellular casts], hematologic disorder [hemolytic anemia (with reticulocytosis), AL_CS_Benlysta_PA_ProgSum_0817 Page 1 of 5
2 leukopenia, lymphopenia, or positive finding of antiphospholipid antibodies or anti-sm antibodies]) c. ONE of the following: 1. The patient is currently on a standard of care SLE treatment regimen comprised of at least one of the following: corticosteroids, antimalarials (e.g. hydroxychloroquine, chloroquine), nonsteroidal anti-inflammatory drugs (e.g. NSAIDS), aspirin, and/or immunosuppressives (e.g. azathioprine, methotrexate, cyclosporine, oral cyclophosphamide, or mycophenolate) 2. The patient has a documented intolerance, FDA labeled contraindication, or hypersensitivity to ALL the standard of care drug classes listed above ii. The patient has another FDA labeled diagnosis 2. The patient does NOT have severe active lupus nephritis requiring inter-current treatment with cyclophosphamide or other biologic therapy or the initiation of other immunosuppressive therapy 3. The patient does NOT have severe active central nervous system lupus complications requiring inter-current treatment with cyclophosphamide or other biologic therapy or the initiation of other immunosuppressive therapy 4. The patient is NOT currently being treated with intravenous cyclophosphamide intravenous cyclophosphamide will be discontinued before starting therapy with Benlysta 5. The patient is NOT currently using another biologic agent the biologic agent will be discontinued before starting therapy with Benlysta 6. The patient is NOT currently being treated for a chronic infection 7. The patient does not have any FDA labeled contraindications to the requested agent 8. The dose is within the FDA labeled dose Length of Approval: 12 months Renewal Evaluation Benlysta (belimumab) will be approved when ALL of the following are met: 1. The patient has been previously approved for Benlysta through the Prime Therapeutics medical drug policy process 2. ONE of the following: a. The patient is currently on a standard of care SLE treatment regimen comprised of at least one of the following: corticosteroids, antimalarials (e.g. hydroxychloroquine, chloroquine), nonsteroidal anti-inflammatory drugs (e.g. NSAIDS), aspirin, and/or immunosuppressives (e.g. azathioprine, methotrexate, cyclosporine, oral cyclophosphamide, or mycophenolate) b. The patient has a documented intolerance, FDA labeled contraindication, or hypersensitivity to the standard of care drug classes listed above 3. The patient has had a decrease in symptoms or stabilization in at least one SLE related disease manifestation present prior to administration of Benlysta (belimumab) (SLE related disease manifestations include, but are not limited to: malar rash, discoid rash, photosensitivity, oral ulcers, nonerosive arthritis, pleuritis/pericarditis, renal disorder [persistent proteinuria >0.5 AL_CS_Benlysta_PA_ProgSum_0817 Page 2 of 5
3 grams/day or cellular casts], hematologic disorder [hemolytic anemia (with reticulocytosis), leukopenia, lymphopenia, or positive finding of antiphospholipid antibodies]) 4. The patient does NOT have severe active lupus nephritis requiring inter-current treatment with cyclophosphamide or other biologic therapy or the initiation of other immunosuppressive therapy 5. The patient does NOT have severe active central nervous system lupus complications requiring inter-current treatment with cyclophosphamide or other biologic therapy or the initiation of other immunosuppressive therapy 6. The patient is NOT currently being treated with intravenous cyclophosphamide intravenous cyclophospamide will be discontinued before starting therapy with Benlysta 7. The patient is NOT currently using another biologic agent the biologic therapy will be discontinued before starting therapy with Benlysta 8. The patient is NOT currently being treated for a chronic infection 9. The patient does not have any FDA labeled contraindications to the requested agent 10. The dose is within the FDA labeled dose Length of Approval: 12 months This pharmacy policy is not an authorization, certification, explanation of benefits or a contract. Eligibility and benefits are determined on a case-by-case basis according to the terms of the member s plan in effect as of the date services are rendered. All pharmacy policies are based on (i) information in FDA approved package inserts (and black box warning, alerts, or other information disseminated by the FDA as applicable); (ii) research of current medical and pharmacy literature; and/or (iii) review of common medical practices in the treatment and diagnosis of disease as of the date hereof. Physicians and other providers are solely responsible for all aspects of medical care and treatment, including the type, quality, and levels of care and treatment. The purpose of Blue Cross and Blue Shield of Alabama s pharmacy policies are to provide a guide to coverage. Pharmacy policies are not intended to dictate to physicians how to practice medicine. Physicians should exercise their medical judgment in providing the care they feel is most appropriate for their patients. Neither this policy, nor the successful adjudication of a pharmacy claim, is guarantee of payment. AL_CS_Benlysta_PA_ProgSum_0817 Page 3 of 5
4 FDA APPROVED INDICATIONS DOSAGE 1 Agent Indication* Dosage Benlysta (belimumab) Active autoantibody positive systemic lupus erythematosus in adults who are receiving standard therapy Intravenous Administration: 10 mg/kg administered intravenously at 2-week intervals for the first 3 doses and at 4-week intervals thereafter Subcutaneous Administration: 200 mg subcutaneous once weekly * Limitation of use: efficacy has not been evaluated in patients with severe active lupus nephritis or severe active central nervous system lupus. It has not been studied in combination with other biologics or intravenous cyclophosphamide so is not recommended in these situations. CLINICAL RATIONALE Systemic Lupus Erythematosus (SLE) 2-5 Systemic Lupus Erythematosus (SLE) is a chronic inflammatory autoimmune disease of unknown cause. 4 It has a broad range of clinical and serological manifestations and can affect any organ. Clinical symptoms of SLE include fatigue, fever, myalgia, changes in weight, skin and mucus membrane lesions and ulcers, and vascular disease. SLE can also include cardiac, renal, pulmonary, and neurologic involvement. Due to its multisystem involvement and likelihood of changes in presentation, the diagnosis of SLE may be difficult. 4 The American College of Rheumatology published criteria to aid in diagnosing patients with SLE. The criteria requires the patient has, at any time in his or her medical history, at least four of the following diagnostic criteria: malar rash, discoid rash, photosensitivity, oral ulcers, nonerosive arthritis, serositis (e.g. pleuritis/pericarditis), renal disorder [e.g. persistent proteinuria >0.5 grams/day or cellular casts], hematologic disorder [e.g. hemolytic anemia (with reticulocytosis), leukopenia, lymphopenia, or thrombocytopenia], and/or immunologic disorder (e.g. positive finding of antiphospholipid antibodies or anti-sm antibodies). 4 In the 2012 revised American College of Rheumatology (ACR) SLE classification criteria, a person is classified as having SLE in the presence of biopsy-proven lupus nephritis with antinuclear antibodies (ANA) or anti-dsdna antibodies or if 4 of the ACR diagnostic criteria, including at least 1 clinical and 1 immunologic criterion have been established. 4 When SLE is suspected standard laboratory evaluations include CBC with differential, serum creatinine, and urinalysis with microscopy. Nonspecific symptomatic treatments such as nonsteroidal anti-inflammatory drugs (NSAIDs), salicylates, and topical therapies may be used initially in SLE patients. Additional conventional SLE therapies include antimalarial drugs, such as hydroxychloroquine and quinacrine, and many nonspecific immunosuppressive medications, such as glucocorticoids, azathioprine, methotrexate, mycophenolate mofetil, and cyclophosphamide. The goal has been the suppression and rebooting of an overactive immune system and thus disease amelioration. However, because of the nonspecific nature and broad immunologic impact of these therapies, the effects have not been optimal and not without a price with regard to infections and other adverse events. In addition, many of these therapies have been empirically introduced in the clinic without the support of good-quality randomized controlled trials. However, hydroxychloroquine is considered the cornerstone of SLE management with evidence from the LUMIA (Lupus in Minorities: Nature versus Nurture) and other trials with evidence of a decrease in flares and prolonged life. 8 Guidelines The American College of Rheumatology (ACR) 4 and The European League Against Rheumatism (EULAR) 5 have published guidelines for the management of adults with SLE. The guidelines are based on available evidence-based information for the diagnosis and management of SLE with the goal of improving the quality of care for patients by primary care physicians. The recommendations are based on both evidence-based AL_CS_Benlysta_PA_ProgSum_0817 Page 4 of 5
5 research, as well as expert consensus. Neither set of guidelines provides specific recommendations for drug selection and use. Compendia Supported Indications For the purposes of the criteria, indications deemed appropriate are those that are supported in AHFS, DrugDex with a level of evidence of 2a or NCCN with a level of evidence of 2a or stronger. References 1. Benlysta Prescribing Information. GlaxoSmithKline. July Goldberg A, Katzap E. Belimumab for the Treatment of Systemic Lupus Erythematosus. International Journal of Clinical Rheumatology. 2010;5(4): FDA Arthritis advisory committee meeting briefing document BLA October Available at: Accessed April 12, American College of Rheumatology and Ad Hoc Committee on Systemic Lupus Erythematosus Guidelines. Guidelines for referral and management of systemic lupus erythematosus in adults. Arthritis Rheum. 1999;42: Bertsias G, Ioannidis J, Boletis J, et al. EULAR recommendations for the management of systemic lupus erythematosus (SLE) report of a task force of the European Standing Committee for International Clinical Studies Including Therapeutics (ESCISIT). Ann Rheum Dis. 2008;67: Navarra S, Guzmán R, Gallacher A, Hall S, et al. Efficacy and safety of belimumab in patients with active systemic lupus erythematosus: a randomised, placebo-controlled, phase 3 trial. Lancet. 377(9767): Wiglesworth A, Ennis K, Kockler, D. Belimumab: A BLyS-Specific Inhibitor for Systemic Lupus Erythematosus. Annals of Pharmacotherapy. 2010;44: Bartels, Christie. Systemic Lupus Erythematosus (SLE) Treatment & Management. Medscape. Available at Accessed 7/20/17. This pharmacy policy is not an authorization, certification, explanation of benefits or a contract. Eligibility and benefits are determined on a case-by-case basis according to the terms of the member s plan in effect as of the date services are rendered. All pharmacy policies are based on (i) information in FDA approved package inserts (and black box warning, alerts, or other information disseminated by the FDA as applicable); (ii) research of current medical and pharmacy literature; and/or (iii) review of common medical practices in the treatment and diagnosis of disease as of the date hereof. Physicians and other providers are solely responsible for all aspects of medical care and treatment, including the type, quality, and levels of care and treatment. The purpose of Blue Cross and Blue Shield of Alabama s pharmacy policies are to provide a guide to coverage. Pharmacy policies are not intended to dictate to physicians how to practice medicine. Physicians should exercise their medical judgment in providing the care they feel is most appropriate for their patients. Neither this policy, nor the successful adjudication of a pharmacy claim, is guarantee of payment. AL_CS_Benlysta_PA_ProgSum_0817 Page 5 of 5
ONE of the following:
 Medical Coverage Policy Belimumab (Benlysta) EFFECTIVE DATE: 01 01 2012 POLICY LAST UPDATED: 11 21 2017 OVERVIEW Belimumab (Benlysta ) is indicated for the treatment of adult patients with active, autoantibody-positive,
Medical Coverage Policy Belimumab (Benlysta) EFFECTIVE DATE: 01 01 2012 POLICY LAST UPDATED: 11 21 2017 OVERVIEW Belimumab (Benlysta ) is indicated for the treatment of adult patients with active, autoantibody-positive,
Committee Approval Date: May 9, 2014 Next Review Date: May 2015
 Medication Policy Manual Policy No: dru248 Topic: Benlysta, belimumab Date of Origin: May 13, 2011 Committee Approval Date: May 9, 2014 Next Review Date: May 2015 Effective Date: June 1, 2014 IMPORTANT
Medication Policy Manual Policy No: dru248 Topic: Benlysta, belimumab Date of Origin: May 13, 2011 Committee Approval Date: May 9, 2014 Next Review Date: May 2015 Effective Date: June 1, 2014 IMPORTANT
Methotrexate Injectable Step Therapy Program Summary
 Methotrexate Injectable Step Therapy Program Summary This prior authorization applies to Commercial, SourceRx and Health Insurance Marketplace formularies. OBJECTIVE The intent of the methotrexate injectable
Methotrexate Injectable Step Therapy Program Summary This prior authorization applies to Commercial, SourceRx and Health Insurance Marketplace formularies. OBJECTIVE The intent of the methotrexate injectable
Corporate Medical Policy
 Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: belimumab_benlysta 6/2011 2/2018 2/2019 3/2018 Description of Procedure or Service Belimumab (Benlysta) is
Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: belimumab_benlysta 6/2011 2/2018 2/2019 3/2018 Description of Procedure or Service Belimumab (Benlysta) is
* Autoantibody positive (i.e. antinuclear antibody or ANA titre 1:80 or anti-double stranded (ds) DNA antibody 30 IU/mL)
 Benlysta (belimumab) Policy Number: 5.02.509 Last Review: 05/2018 Origination: 06/2011 Next Review: 05/2019 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will provide coverage for Benlysta
Benlysta (belimumab) Policy Number: 5.02.509 Last Review: 05/2018 Origination: 06/2011 Next Review: 05/2019 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will provide coverage for Benlysta
Cigna Drug and Biologic Coverage Policy
 Cigna Drug and Biologic Coverage Policy Subject Belimumab Table of Contents Coverage Policy... 1 General Background... 2 Coding/Billing Information... 4 References... 4 Effective Date... 11/15/2017 Next
Cigna Drug and Biologic Coverage Policy Subject Belimumab Table of Contents Coverage Policy... 1 General Background... 2 Coding/Billing Information... 4 References... 4 Effective Date... 11/15/2017 Next
Benlysta. Benlysta (belimumab) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.99.01 Subject: Benlysta Page: 1 of 5 Last Review Date: June 22, 2018 Benlysta Description Benlysta (belimumab)
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.99.01 Subject: Benlysta Page: 1 of 5 Last Review Date: June 22, 2018 Benlysta Description Benlysta (belimumab)
Benlysta. Benlysta (belimumab) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Benlysta Page: 1 of 5 Last Review Date: December 3, 2015 Benlysta Description Benlysta (belimumab)
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Benlysta Page: 1 of 5 Last Review Date: December 3, 2015 Benlysta Description Benlysta (belimumab)
LUPUS CAN DO EVERYTHING, BUT NOT EVERYTHING IS LUPUS LUPUS 101 SLE SUBSETS AUTOIMMUNE DISEASE 11/4/2013 HOWARD HAUPTMAN, MD IDIOPATHIC DISCOID LUPUS
 LUPUS 101 LUPUS CAN DO EVERYTHING, BUT NOT EVERYTHING IS LUPUS HOWARD HAUPTMAN, MD IDIOPATHIC DISCOID LUPUS SLE SUBSETS SUBACUTE CUTANEOUS LUPUS DRUG INDUCED LUPUS NEONATAL LUPUS LATE ONSET LUPUS ANTI-PHOSPHOLIPID
LUPUS 101 LUPUS CAN DO EVERYTHING, BUT NOT EVERYTHING IS LUPUS HOWARD HAUPTMAN, MD IDIOPATHIC DISCOID LUPUS SLE SUBSETS SUBACUTE CUTANEOUS LUPUS DRUG INDUCED LUPUS NEONATAL LUPUS LATE ONSET LUPUS ANTI-PHOSPHOLIPID
Prior Authorization with Quantity Limit Program Summary
 Gocovri (amantadine) Prior Authorization with Quantity Limit Program Summary This prior authorization applies to Commercial, NetResults A series, SourceRx and Health Insurance Marketplace formularies.
Gocovri (amantadine) Prior Authorization with Quantity Limit Program Summary This prior authorization applies to Commercial, NetResults A series, SourceRx and Health Insurance Marketplace formularies.
This program applies to Commercial, GenPlus and Health Insurance Marketplace formularies.
 OBJECTIVE The intent of the GLP-1 (glucagon-like peptide-1) Agonists [Adlyxin (lixisenatide), Byetta (exenatide), Bydureon (exenatide extended-release), Tanzeum (albiglutide), Trulicity (dulaglutide),
OBJECTIVE The intent of the GLP-1 (glucagon-like peptide-1) Agonists [Adlyxin (lixisenatide), Byetta (exenatide), Bydureon (exenatide extended-release), Tanzeum (albiglutide), Trulicity (dulaglutide),
Efficacy and Safety of Belimumab in the treatment of Systemic Lupus Erythematosus: a Prospective Multicenter Study.
 1. Title Efficacy and Safety of Belimumab in the treatment of Systemic Lupus Erythematosus: a Prospective Multicenter Study. 2. Background Systemic Lupus Erythematosus (SLE) is a chronic, autoimmune and
1. Title Efficacy and Safety of Belimumab in the treatment of Systemic Lupus Erythematosus: a Prospective Multicenter Study. 2. Background Systemic Lupus Erythematosus (SLE) is a chronic, autoimmune and
9/25/2013 SYSTEMIC LUPUS ERYTHEMATOSUS (SLE)
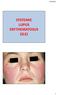 SYSTEMIC LUPUS ERYTHEMATOSUS (SLE) 1 Other Types of Lupus Discoid Lupus Erythematosus Lupus Pernio --- Sarcoidosis Lupus Vulgaris --- Tuberculosis of the face Manifestations of SLE Fever Rashes Arthritis
SYSTEMIC LUPUS ERYTHEMATOSUS (SLE) 1 Other Types of Lupus Discoid Lupus Erythematosus Lupus Pernio --- Sarcoidosis Lupus Vulgaris --- Tuberculosis of the face Manifestations of SLE Fever Rashes Arthritis
Arthritis & Rheumatology Clinics of Kansas PATIENT EDUCATION SYSTEMIC LUPUS ERYTHEMATOSUS
 Arthritis & Rheumatology Clinics of Kansas PATIENT EDUCATION SYSTEMIC LUPUS ERYTHEMATOSUS Introduction: There is perhaps no rheumatic disease that evokes so much fear and confusion among both patients
Arthritis & Rheumatology Clinics of Kansas PATIENT EDUCATION SYSTEMIC LUPUS ERYTHEMATOSUS Introduction: There is perhaps no rheumatic disease that evokes so much fear and confusion among both patients
Summary Article: Lupus (Systemic Lupus Erythematosus) from Harvard Medical School Health Topics A-Z
 Topic Page: Systemic Lupus Erythematosus Summary Article: Lupus (Systemic Lupus Erythematosus) from Harvard Medical School Health Topics A-Z What Is It? Lupus is thought to develop when the immune system
Topic Page: Systemic Lupus Erythematosus Summary Article: Lupus (Systemic Lupus Erythematosus) from Harvard Medical School Health Topics A-Z What Is It? Lupus is thought to develop when the immune system
.,Dr Ali Alkazzaz Babylon collage of medicine 2016
 .,Dr Ali Alkazzaz Babylon collage of medicine 2016 Lupus history Lupus is the Latin word for wolf 1 st used medically in the 10 th century Described clinically in the 19 th century Butterfly rash in 1845
.,Dr Ali Alkazzaz Babylon collage of medicine 2016 Lupus history Lupus is the Latin word for wolf 1 st used medically in the 10 th century Described clinically in the 19 th century Butterfly rash in 1845
Lupus. Fast facts. What is lupus? What causes lupus? Who gets lupus?
 Lupus Systemic lupus erythematosus, referred to as SLE or lupus, is sometimes called the "great imitator." Why? Because of its wide range of symptoms, people often confuse lupus with other health problems.
Lupus Systemic lupus erythematosus, referred to as SLE or lupus, is sometimes called the "great imitator." Why? Because of its wide range of symptoms, people often confuse lupus with other health problems.
Definition Chronic autoimmune disease The body s immune system starts attacking itself Can affect most organs and tissues in the body Brain, lungs, he
 LIVING WITH SYSTEMIC LUPUS ERYTHEMATOSUS Stacy Kennedy, M.D.,M.B.A. Rowan Diagnostic Clinic Salisbury, N.C. May 11, 2013 Agenda What is lupus Who is affected Causes of lupus Symptoms and organ involvement
LIVING WITH SYSTEMIC LUPUS ERYTHEMATOSUS Stacy Kennedy, M.D.,M.B.A. Rowan Diagnostic Clinic Salisbury, N.C. May 11, 2013 Agenda What is lupus Who is affected Causes of lupus Symptoms and organ involvement
Development of SLE among Possible SLE Patients Seen in Consultation: Long-Term Follow-Up. Disclosures. Background. Evidence-Based Medicine.
 Development of SLE among Patients Seen in Consultation: Long-Term Follow-Up Abstract # 1699 May Al Daabil, MD Bonnie L. Bermas, MD Alexander Fine Hsun Tsao Patricia Ho Joseph F. Merola, MD Peter H. Schur,
Development of SLE among Patients Seen in Consultation: Long-Term Follow-Up Abstract # 1699 May Al Daabil, MD Bonnie L. Bermas, MD Alexander Fine Hsun Tsao Patricia Ho Joseph F. Merola, MD Peter H. Schur,
Living with Lupus: An Insider s Perspective
 Living with Lupus: An Insider s Perspective Pamela Thorpe, MD, FACP Lupus Foundation of America, Inc. Philadelphia Tri-State Chapter Volunteer May 2014 My Own Story Is it Lupus Yet? The What What is this?
Living with Lupus: An Insider s Perspective Pamela Thorpe, MD, FACP Lupus Foundation of America, Inc. Philadelphia Tri-State Chapter Volunteer May 2014 My Own Story Is it Lupus Yet? The What What is this?
UNDERSTANDING SYSTEMIC LUPUS ERYTHEMATOSUS
 UNDERSTANDING SYSTEMIC LUPUS ERYTHEMATOSUS Stacy Kennedy, M.D.,M.B.A. October 20, 2012 Agenda What is lupus Who is affected Causes of lupus Symptoms and organ involvement Diagnosis Treatment Pregnancy
UNDERSTANDING SYSTEMIC LUPUS ERYTHEMATOSUS Stacy Kennedy, M.D.,M.B.A. October 20, 2012 Agenda What is lupus Who is affected Causes of lupus Symptoms and organ involvement Diagnosis Treatment Pregnancy
The only biologic approved to treat SLE: now with multiple delivery options
 The only biologic approved to treat SLE: now with multiple delivery options BENLYSTA (belimumab) Autoinjector SC Prefilled syringe IV Intravenous infusion Consider the options: visit Belimumab.com INDICATION
The only biologic approved to treat SLE: now with multiple delivery options BENLYSTA (belimumab) Autoinjector SC Prefilled syringe IV Intravenous infusion Consider the options: visit Belimumab.com INDICATION
Systemic Lupus Erythematosus (SLE) Epidemiology of SLE (United States)
 1:10-2:25pm Closing the Loop on Lupus: Primary Care s Key Role in the Elusive Diagnosis and Management of Patients SPEAKER Richard Sadovsky, MD Richard Furie, MD Disclosures This session is supported by
1:10-2:25pm Closing the Loop on Lupus: Primary Care s Key Role in the Elusive Diagnosis and Management of Patients SPEAKER Richard Sadovsky, MD Richard Furie, MD Disclosures This session is supported by
High Impact Rheumatology
 High Impact Rheumatology Systemic Lupus Erythematosus Bernard Rubin, DO MPH Case 1: History A 45-year-old woman presents with severe dyspnea and cough. She was in excellent health until 4 weeks ago when
High Impact Rheumatology Systemic Lupus Erythematosus Bernard Rubin, DO MPH Case 1: History A 45-year-old woman presents with severe dyspnea and cough. She was in excellent health until 4 weeks ago when
Horizon Scanning Centre May Tabalumab for systemic lupus erythematosus SUMMARY NIHR HSC ID: 5581
 Horizon Scanning Centre May 2014 Tabalumab for systemic lupus erythematosus SUMMARY NIHR HSC ID: 5581 This briefing is based on information available at the time of research and a limited literature search.
Horizon Scanning Centre May 2014 Tabalumab for systemic lupus erythematosus SUMMARY NIHR HSC ID: 5581 This briefing is based on information available at the time of research and a limited literature search.
To live with lupus, we need to know about lupus.
 To live with lupus, we need to know about lupus. Zineb Aouhab, MD Assistant Professor of Medicine Loyola University Medical Center Division of Rheumatology 1 Where did the word lupus come from? The word
To live with lupus, we need to know about lupus. Zineb Aouhab, MD Assistant Professor of Medicine Loyola University Medical Center Division of Rheumatology 1 Where did the word lupus come from? The word
Rheumatology Primer: What Labs and When
 Rheumatology Primer: What Labs and When Irina Konon, MD Department of Internal Medicine Division of Rheumatology Medical College of Wisconsin Disclosures None 1 Objective Discuss principles of laboratory
Rheumatology Primer: What Labs and When Irina Konon, MD Department of Internal Medicine Division of Rheumatology Medical College of Wisconsin Disclosures None 1 Objective Discuss principles of laboratory
Clinical Policy: Anakinra (Kineret) Reference Number: CP.PHAR.244 Effective Date: Last Review Date: Line of Business: HIM, Medicaid
 Clinical Policy: (Kineret) Reference Number: CP.PHAR.244 Effective Date: 08.16 Last Review Date: 11.18 Line of Business: HIM, Medicaid Coding Implications Revision Log See Important Reminder at the end
Clinical Policy: (Kineret) Reference Number: CP.PHAR.244 Effective Date: 08.16 Last Review Date: 11.18 Line of Business: HIM, Medicaid Coding Implications Revision Log See Important Reminder at the end
Evidence Based Treatment of SLE with Treatment Algorithm. Dr. Md. Mujibur Rahman Professor of Medicine Shaheed Suhrawardy Medical College
 Evidence Based Treatment of SLE with Treatment Algorithm Dr. Md. Mujibur Rahman Professor of Medicine Shaheed Suhrawardy Medical College Natural Histoty Inflammatory multisystem disease Onset usually between
Evidence Based Treatment of SLE with Treatment Algorithm Dr. Md. Mujibur Rahman Professor of Medicine Shaheed Suhrawardy Medical College Natural Histoty Inflammatory multisystem disease Onset usually between
Systemic Lupus Erythematosus
 Systemic Lupus Erythematosus Marc C. Hochberg, MD, MPH Professor of Medicine and Head, Division of Rheumatology University of Maryland School of Medicine CASE: HISTORY A 26-year-old woman is seen for migratory
Systemic Lupus Erythematosus Marc C. Hochberg, MD, MPH Professor of Medicine and Head, Division of Rheumatology University of Maryland School of Medicine CASE: HISTORY A 26-year-old woman is seen for migratory
Soliris Medical Policy Prior Authorization Program Summary
 Soliris Medical Policy Prior Authorization Program Summary Precertification/Prior Authorization may be required under certain plans. Please verify each member s benefits. OBJECTIVE The intent of the Soliris
Soliris Medical Policy Prior Authorization Program Summary Precertification/Prior Authorization may be required under certain plans. Please verify each member s benefits. OBJECTIVE The intent of the Soliris
Ask the Expert: Photosensitivity in Cutaneous Lupus
 Ask the Expert: Photosensitivity in Cutaneous Lupus Victoria P. Werth, MD Department of Dermatology & Medicine University of Pennsylvania ; Philadelphia VA Hospital Overview Definition Impact of photosensitivity
Ask the Expert: Photosensitivity in Cutaneous Lupus Victoria P. Werth, MD Department of Dermatology & Medicine University of Pennsylvania ; Philadelphia VA Hospital Overview Definition Impact of photosensitivity
SLE and the Antiphospholipid Syndrome
 SLE and the Antiphospholipid Syndrome Susan Y. Ritter MD, PhD Associate Physician Division of Rheumatology, Immunology and Allergy Department of Medicine Brigham and Women s Hospital Instructor in Medicine
SLE and the Antiphospholipid Syndrome Susan Y. Ritter MD, PhD Associate Physician Division of Rheumatology, Immunology and Allergy Department of Medicine Brigham and Women s Hospital Instructor in Medicine
Systemic Lupus Erythematosus (SLE) Learning Objectives. Presenter Disclosure Information
 9:30 10:45 AM Closing the Loop on Lupus: Primary Care s Key Role in the Elusive Diagnosis and Management of Patients Presenter Disclosure Information The following relationships exist related to this presentation:
9:30 10:45 AM Closing the Loop on Lupus: Primary Care s Key Role in the Elusive Diagnosis and Management of Patients Presenter Disclosure Information The following relationships exist related to this presentation:
Dr. Venkateswari. R. Dr. Janani Sankar s unit Kanchi Kamakoti CHILDS Trust Hospital
 Dr. Venkateswari. R. Dr. Janani Sankar s unit Kanchi Kamakoti CHILDS Trust Hospital Acknowledgements: KKCTH Dr. Ramkumar Consultant Dermatologist Dr. Ramprakash Consultant Ophthalmologist Dr. Prasad Manne
Dr. Venkateswari. R. Dr. Janani Sankar s unit Kanchi Kamakoti CHILDS Trust Hospital Acknowledgements: KKCTH Dr. Ramkumar Consultant Dermatologist Dr. Ramprakash Consultant Ophthalmologist Dr. Prasad Manne
Living with Lupus. LFA - Georgia Gary E Myerson MD Arthritis and Rheumatology of GA
 Living with Lupus LFA - Georgia Gary E Myerson MD Arthritis and Rheumatology of GA LUPUS A REALITY CHECK LUPUS A REALITY CHECK LUPUS A REALITY CHECK LUPUS A REALITY CHECK SLE 1.5 million Americans: some
Living with Lupus LFA - Georgia Gary E Myerson MD Arthritis and Rheumatology of GA LUPUS A REALITY CHECK LUPUS A REALITY CHECK LUPUS A REALITY CHECK LUPUS A REALITY CHECK SLE 1.5 million Americans: some
Parathyroid Hormone Analog for Osteoporosis Prior Authorization with Quantity Limit Criteria Program Summary
 Parathyroid Hormone Analog for Osteoporosis Prior Authorization with Quantity Limit Criteria Program Summary This prior authorization program applies to Commercial, NetResults A series, NetResults F series
Parathyroid Hormone Analog for Osteoporosis Prior Authorization with Quantity Limit Criteria Program Summary This prior authorization program applies to Commercial, NetResults A series, NetResults F series
Policy. Section: Medicine Effective Date: January 15, 2015 Subsection: Pathology/Laboratory Original Policy Date: December 5, 2014 Subject:
 Last Review Status/Date: December 2014 Page: 1 of 10 Summary Systemic lupus erythematosus (SLE) is an autoimmune connective tissue disease that can be difficult to diagnose because patients often present
Last Review Status/Date: December 2014 Page: 1 of 10 Summary Systemic lupus erythematosus (SLE) is an autoimmune connective tissue disease that can be difficult to diagnose because patients often present
Conflict of Interest. Systemic Lupus Erythematosus and the Antiphospholipid Syndrome Bonnie L. Bermas, MD Brigham and Women s Hospital.
 Systemic Lupus Erythematosus and the Antiphospholipid Syndrome Bonnie L. Bermas, MD Brigham and Women s Hospital Conflict of Interest Disclosures: None Overview Diagnostic Classification Criteria of SLE
Systemic Lupus Erythematosus and the Antiphospholipid Syndrome Bonnie L. Bermas, MD Brigham and Women s Hospital Conflict of Interest Disclosures: None Overview Diagnostic Classification Criteria of SLE
COMPLEX CUTANEOUS LUPUS CASES PEARLS AND PITFALLS
 COMPLEX CUTANEOUS LUPUS CASES PEARLS AND PITFALLS Benjamin F. Chong, MD, MSCS February 17, 2018 CONFLICTS OF INTEREST D I S CLOSURE OF R E LATI ONSHIPS W I T H I NDUSTRY Benjamin Chong, MD, MSCS F059 Complex
COMPLEX CUTANEOUS LUPUS CASES PEARLS AND PITFALLS Benjamin F. Chong, MD, MSCS February 17, 2018 CONFLICTS OF INTEREST D I S CLOSURE OF R E LATI ONSHIPS W I T H I NDUSTRY Benjamin Chong, MD, MSCS F059 Complex
Living with Lupus. Helping Your Patient With Systemic Lupus Erythematosus By Hussein M. Halaby, MBBS, ABIM, FRCPC; and John M. Esdaile, MD, MPH, FRCPC
 Focus on CME at the University McGill University of British Columbia Living with Lupus Helping Your Patient With Systemic Lupus Erythematosus By Hussein M. Halaby, MBBS, ABIM, FRCPC; and John M. Esdaile,
Focus on CME at the University McGill University of British Columbia Living with Lupus Helping Your Patient With Systemic Lupus Erythematosus By Hussein M. Halaby, MBBS, ABIM, FRCPC; and John M. Esdaile,
Learning about Lupus. Learning About Lupus. Lupus Society of Illinois
 Learning About Lupus Learning about Lupus Lupus Society of Illinois 525 W. Monroe Street, Suite 900 Chicago, Illinois 60661 Robert S. Katz, M.D. Professor of Medicine Rush University Medical Center Northwestern
Learning About Lupus Learning about Lupus Lupus Society of Illinois 525 W. Monroe Street, Suite 900 Chicago, Illinois 60661 Robert S. Katz, M.D. Professor of Medicine Rush University Medical Center Northwestern
Systemic Lupus Erythematosus among Jordanians: A Single Rheumatology Unit Experience
 Systemic Lupus Erythematosus among Jordanians: A Single Rheumatology Unit Experience Ala M. AlHeresh MD* ABSTRACT Objectives: To study the characteristics of Systemic Lupus Erythematosus in Jordan and
Systemic Lupus Erythematosus among Jordanians: A Single Rheumatology Unit Experience Ala M. AlHeresh MD* ABSTRACT Objectives: To study the characteristics of Systemic Lupus Erythematosus in Jordan and
Clinical Policy: Ixekizumab (Taltz) Reference Number: ERX.SPA.122 Effective Date:
 Clinical Policy: (Taltz) Reference Number: ERX.SPA.122 Effective Date: 10.01.16 Last Review Date: 11.18 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
Clinical Policy: (Taltz) Reference Number: ERX.SPA.122 Effective Date: 10.01.16 Last Review Date: 11.18 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
Systemic lupus erythematosus in a male patient
 IOP Conference Series: Earth and Environmental Science PAPER OPEN ACCESS Systemic lupus erythematosus in a male patient To cite this article: H Sibarani and Z Zubir 2018 IOP Conf. Ser.: Earth Environ.
IOP Conference Series: Earth and Environmental Science PAPER OPEN ACCESS Systemic lupus erythematosus in a male patient To cite this article: H Sibarani and Z Zubir 2018 IOP Conf. Ser.: Earth Environ.
Systemic examination
 PROLONGED FEVER IN AN ADOLESCENT BOY Dr.Praveena Lionel, DNB PG, Dr.Kannan (HOD) Railway Hospital, Perambur History 11 yrs old adolescent boy was admitted with c/o Fever -1 wk Myalgia -1 wk Arthralgia
PROLONGED FEVER IN AN ADOLESCENT BOY Dr.Praveena Lionel, DNB PG, Dr.Kannan (HOD) Railway Hospital, Perambur History 11 yrs old adolescent boy was admitted with c/o Fever -1 wk Myalgia -1 wk Arthralgia
RITUXAN (rituximab and hyaluronidase human)
 Drug Prior Authorization Guideline RITUXIMAB products J9310 RITUXAN (rituximab and hyaluronidase human) PA9847 Covered Service: Prior Authorization Required: Additional Information: Yes when meets criteria
Drug Prior Authorization Guideline RITUXIMAB products J9310 RITUXAN (rituximab and hyaluronidase human) PA9847 Covered Service: Prior Authorization Required: Additional Information: Yes when meets criteria
Clinical Policy: Tocilizumab (Actemra) Reference Number: CP.PHAR.263 Effective Date: Last Review Date: Line of Business: HIM, Medicaid
 Clinical Policy: (Actemra) Reference Number: CP.PHAR.263 Effective Date: 07.01.16 Last Review Date: 11.18 Line of Business: HIM, Medicaid Coding Implications Revision Log See Important Reminder at the
Clinical Policy: (Actemra) Reference Number: CP.PHAR.263 Effective Date: 07.01.16 Last Review Date: 11.18 Line of Business: HIM, Medicaid Coding Implications Revision Log See Important Reminder at the
Medical Policy An independent licensee of the Blue Cross Blue Shield Association
 Ocaliva (obeticholic acid) Page 1 of 6 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Ocaliva (obeticholic acid) Prime Therapeutics will review Prior Authorization
Ocaliva (obeticholic acid) Page 1 of 6 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Ocaliva (obeticholic acid) Prime Therapeutics will review Prior Authorization
Clinical Policy: Etanercept (Enbrel) Reference Number: CP.PHAR.250 Effective Date: 08/16 Last Review Date: 08/17 Line of Business: Medicaid
 Clinical Policy: (Enbrel) Reference Number: CP.PHAR.250 Effective Date: 08/16 Last Review Date: 08/17 Line of Business: Medicaid Coding Implications Revision Log See Important Reminder at the end of this
Clinical Policy: (Enbrel) Reference Number: CP.PHAR.250 Effective Date: 08/16 Last Review Date: 08/17 Line of Business: Medicaid Coding Implications Revision Log See Important Reminder at the end of this
CSTAR CASE STUDIES: BLOCK F Type 3 Hypersensitivity Reaction
 CSTAR CASE STUDIES: BLOCK F Type 3 Hypersensitivity Reaction Setting: ER Mr. Smith I ve just felt so weak for so long, and I ve lost so much weight, and now I m having trouble breathing it s affecting
CSTAR CASE STUDIES: BLOCK F Type 3 Hypersensitivity Reaction Setting: ER Mr. Smith I ve just felt so weak for so long, and I ve lost so much weight, and now I m having trouble breathing it s affecting
Neuropsychiatric Systemic Lupus Erythematosus (NPSLE) Case presentations and topic discussion The Rheumatology Unit UMMC experience
 Neuropsychiatric Systemic Lupus Erythematosus (NPSLE) Case presentations and topic discussion The Rheumatology Unit UMMC experience References Sanna G, Bertolaccini ML. Neuropsychiatric manifestations
Neuropsychiatric Systemic Lupus Erythematosus (NPSLE) Case presentations and topic discussion The Rheumatology Unit UMMC experience References Sanna G, Bertolaccini ML. Neuropsychiatric manifestations
Medical Policy An independent licensee of the Blue Cross Blue Shield Association
 CGRP Page 1 of 8 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: CGRP (calcitonin gene-related peptide) Prime Therapeutics will review Prior Authorization requests
CGRP Page 1 of 8 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: CGRP (calcitonin gene-related peptide) Prime Therapeutics will review Prior Authorization requests
Clinical Policy: Abatacept (Orencia) Reference Number: ERX.SPA.123 Effective Date:
 Clinical Policy: (Orencia) Reference Number: ERX.SPA.123 Effective Date: 10.01.16 Last Review Date: 05.18 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
Clinical Policy: (Orencia) Reference Number: ERX.SPA.123 Effective Date: 10.01.16 Last Review Date: 05.18 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
Clinical Policy: Secukinumab (Cosentyx) Reference Number: CP.PHAR.261 Effective Date: Last Review Date: Line of Business: HIM, Medicaid
 Clinical Policy: (Cosentyx) Reference Number: CP.PHAR.261 Effective Date: 08.16 Last Review Date: 11.18 Line of Business: HIM, Medicaid Coding Implications Revision Log See Important Reminder at the end
Clinical Policy: (Cosentyx) Reference Number: CP.PHAR.261 Effective Date: 08.16 Last Review Date: 11.18 Line of Business: HIM, Medicaid Coding Implications Revision Log See Important Reminder at the end
Clinical Policy: Apremilast (Otezla) Reference Number: CP.PHAR.245 Effective Date: Last Review Date: Line of Business: HIM, Medicaid
 Clinical Policy: (Otezla) Reference Number: CP.PHAR.245 Effective Date: 08.16 Last Review Date: 11.18 Line of Business: HIM, Medicaid Revision Log See Important Reminder at the end of this policy for important
Clinical Policy: (Otezla) Reference Number: CP.PHAR.245 Effective Date: 08.16 Last Review Date: 11.18 Line of Business: HIM, Medicaid Revision Log See Important Reminder at the end of this policy for important
Systemic Lupus Erythematosus
 A Patient s Guide to Systemic Lupus Erythematosus 2 kidneys. These changes make it impossible for the kidneys to function normally. The inflammation of SLE can be seen in the lining, covering, and muscles
A Patient s Guide to Systemic Lupus Erythematosus 2 kidneys. These changes make it impossible for the kidneys to function normally. The inflammation of SLE can be seen in the lining, covering, and muscles
LUPUS (SLE) MEDICAL SOURCE STATEMENT
 LUPUS (SLE) MEDICAL SOURCE STATEMENT From: Re: (Name of Patient) (Social Security No.) Please answer the following questions concerning your patient s impairments. Attach relevant treatment notes, radiologist
LUPUS (SLE) MEDICAL SOURCE STATEMENT From: Re: (Name of Patient) (Social Security No.) Please answer the following questions concerning your patient s impairments. Attach relevant treatment notes, radiologist
Section: Medicine Effective Date: January 15, 2016 Subsection: Pathology/Laboratory Original Policy Date: December 5, 2014 Subject:
 Last Review Status/Date: December 2015 Page: 1 of 11 Summary Systemic lupus erythematosus (SLE) is an autoimmune connective tissue disease that can be difficult to diagnose because patients often present
Last Review Status/Date: December 2015 Page: 1 of 11 Summary Systemic lupus erythematosus (SLE) is an autoimmune connective tissue disease that can be difficult to diagnose because patients often present
Belimumab in the treatment of systemic lupus erythematosus: high disease activity predictors of response
 ARD Online First, published on April 5, 2012 as 10.1136/annrheumdis-2011-200937 1 Unit for Clinical Therapy Research, Infl ammatory Diseases (ClinTRID), The Karolinska Institute, Stockholm, Sweden 2 Division
ARD Online First, published on April 5, 2012 as 10.1136/annrheumdis-2011-200937 1 Unit for Clinical Therapy Research, Infl ammatory Diseases (ClinTRID), The Karolinska Institute, Stockholm, Sweden 2 Division
Lupus The Clinical Perspective
 Lupus The Clinical Perspective Anca D. Askanase, M.D., M.P.H. Associate Professor of Medicine Director Lupus Center Columbia University Medical Center New York-Presbyterian Hospital Systemic Lupus Erythematosus
Lupus The Clinical Perspective Anca D. Askanase, M.D., M.P.H. Associate Professor of Medicine Director Lupus Center Columbia University Medical Center New York-Presbyterian Hospital Systemic Lupus Erythematosus
Peter H. Schur Elena M. Massarotti. Editors. Lupus Erythematosus. Clinical Evaluation and Treatment
 Lupus Erythematosus Peter H. Schur Elena M. Massarotti Editors Lupus Erythematosus Clinical Evaluation and Treatment Editors Peter H. Schur Division of Rheumatology, Immunology, Allergy Brigham and Women
Lupus Erythematosus Peter H. Schur Elena M. Massarotti Editors Lupus Erythematosus Clinical Evaluation and Treatment Editors Peter H. Schur Division of Rheumatology, Immunology, Allergy Brigham and Women
Policy. Background
 Last Review Status/Date: December 2016 Page: 1 of 11 Summary Systemic lupus erythematosus (SLE) is an autoimmune connective tissue disease that can be difficult to diagnose because patients often present
Last Review Status/Date: December 2016 Page: 1 of 11 Summary Systemic lupus erythematosus (SLE) is an autoimmune connective tissue disease that can be difficult to diagnose because patients often present
Coverage Criteria: Express Scripts, Inc. monograph dated 12/15/ months or as otherwise noted by indication
 BENEFIT DESCRIPTION AND LIMITATIONS OF COVERAGE ITEM: PRODUCT LINES: COVERED UNDER: DESCRIPTION: CPT/HCPCS Code: Company Supplying: Setting: Kineret (anakinra subcutaneous injection) Commercial HMO/PPO/CDHP
BENEFIT DESCRIPTION AND LIMITATIONS OF COVERAGE ITEM: PRODUCT LINES: COVERED UNDER: DESCRIPTION: CPT/HCPCS Code: Company Supplying: Setting: Kineret (anakinra subcutaneous injection) Commercial HMO/PPO/CDHP
Clinical Policy: Ixekizumab (Taltz) Reference Number: CP.PHAR.257 Effective Date: Last Review Date: 11.18
 Clinical Policy: (Taltz) Reference Number: CP.PHAR.257 Effective Date: 08.01.16 Last Review Date: 11.18 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
Clinical Policy: (Taltz) Reference Number: CP.PHAR.257 Effective Date: 08.01.16 Last Review Date: 11.18 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
Additional file 2: Details of cohort studies and randomised trials
 Reference Randomised trials Ye et al. 2001 Abstract 274 R=1 WD=0 Design, numbers, treatments, duration Randomised open comparison of: (45 patients) 1.5 g for 3, 1 g for 3, then 0.5 to 0.75 g IV cyclophosphamide
Reference Randomised trials Ye et al. 2001 Abstract 274 R=1 WD=0 Design, numbers, treatments, duration Randomised open comparison of: (45 patients) 1.5 g for 3, 1 g for 3, then 0.5 to 0.75 g IV cyclophosphamide
New Drug Evaluation: Belimumab Injection, Intravenous and Subcutaneous
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Clinical Policy: Anakinra (Kineret) Reference Number: ERX.SPA.135 Effective Date:
 Clinical Policy: (Kineret) Reference Number: ERX.SPA.135 Effective Date: 10.01.16 Last Review Date: 05.18 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
Clinical Policy: (Kineret) Reference Number: ERX.SPA.135 Effective Date: 10.01.16 Last Review Date: 05.18 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
Acthar Gel. H. P. Acthar Gel (corticotropin; ACTH) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.30.10 Subject: Acthar Gel Page: 1 of 7 Last Review Date: November 30, 2018 Acthar Gel Description H.
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.30.10 Subject: Acthar Gel Page: 1 of 7 Last Review Date: November 30, 2018 Acthar Gel Description H.
Name of Policy: Zoledronic Acid (Reclast ) Injection
 Name of Policy: Zoledronic Acid (Reclast ) Injection Policy #: 355 Latest Review Date: May 2011 Category: Pharmacy Policy Grade: Active Policy but no longer scheduled for regular literature reviews and
Name of Policy: Zoledronic Acid (Reclast ) Injection Policy #: 355 Latest Review Date: May 2011 Category: Pharmacy Policy Grade: Active Policy but no longer scheduled for regular literature reviews and
Clinical Policy: Secukinumab (Cosentyx) Reference Number: ERX.SPA.165 Effective Date:
 Clinical Policy: (Cosentyx) Reference Number: ERX.SPA.165 Effective Date: 10.01.16 Last Review Date: 11.18 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
Clinical Policy: (Cosentyx) Reference Number: ERX.SPA.165 Effective Date: 10.01.16 Last Review Date: 11.18 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
Elevated BLyS levels in patients with systemic lupus erythematosus: Associated factors and responses to belimumab
 (2016) 25, 346 354 http://lup.sagepub.com PAPER Elevated BLyS levels in patients with systemic lupus erythematosus: Associated factors and responses to belimumab DA Roth 1, A Thompson 2, Y Tang 2*, AE
(2016) 25, 346 354 http://lup.sagepub.com PAPER Elevated BLyS levels in patients with systemic lupus erythematosus: Associated factors and responses to belimumab DA Roth 1, A Thompson 2, Y Tang 2*, AE
Corporate Medical Policy
 Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: abatacept_orencia 4/2008 2/2018 2/2019 2/2018 Description of Procedure or Service Abatacept (Orencia ), a
Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: abatacept_orencia 4/2008 2/2018 2/2019 2/2018 Description of Procedure or Service Abatacept (Orencia ), a
Opinion 23 July 2014
 The legally binding text is the original French version TRANSPARENCY COMMITTEE Opinion 23 July 2014 IMUREL 50 mg, film-coated tablet (B/100) (CIP: 34009 364 149 0 7) IMUREL 25 mg, film-coated tablet (B/50)
The legally binding text is the original French version TRANSPARENCY COMMITTEE Opinion 23 July 2014 IMUREL 50 mg, film-coated tablet (B/100) (CIP: 34009 364 149 0 7) IMUREL 25 mg, film-coated tablet (B/50)
Persistent disease activity may have significant implications for your SLE patients life journey. 1,2
 Persistent disease activity may have significant implications for your SLE patients life journey. 1,2 Is it time to increase the focus on disease activity reduction? BENLYSTA (belimumab) is indicated as
Persistent disease activity may have significant implications for your SLE patients life journey. 1,2 Is it time to increase the focus on disease activity reduction? BENLYSTA (belimumab) is indicated as
Name of Policy: Boniva (Ibandronate Sodium) Infusion
 Name of Policy: Boniva (Ibandronate Sodium) Infusion Policy #: 266 Latest Review Date: April 2010 Category: Pharmacology Policy Grade: Active Policy but no longer scheduled for regular literature reviews
Name of Policy: Boniva (Ibandronate Sodium) Infusion Policy #: 266 Latest Review Date: April 2010 Category: Pharmacology Policy Grade: Active Policy but no longer scheduled for regular literature reviews
Corporate Medical Policy
 Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: golimumab_simponi 8/2013 2/2018 2/2019 3/2018 Description of Procedure or Service Golimumab (Simponi and
Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: golimumab_simponi 8/2013 2/2018 2/2019 3/2018 Description of Procedure or Service Golimumab (Simponi and
Drug Class Prior Authorization Criteria Immune Globulins
 Drug Class Prior Authorization Criteria Immune Globulins Line of Business: Medicaid P & T Approval Date: August 16, 2017 Effective Date: August 16, 2017 This policy has been developed through review of
Drug Class Prior Authorization Criteria Immune Globulins Line of Business: Medicaid P & T Approval Date: August 16, 2017 Effective Date: August 16, 2017 This policy has been developed through review of
Undifferentiated Connective Tissue Disease and Overlap Syndromes. Mark S. Box, MD
 Undifferentiated Connective Tissue Disease and Overlap Syndromes Mark S. Box, MD Overlap Syndromes As many as 25% of patients with rheumatic diseases with systemic symptoms cannot be definitely diagnosed
Undifferentiated Connective Tissue Disease and Overlap Syndromes Mark S. Box, MD Overlap Syndromes As many as 25% of patients with rheumatic diseases with systemic symptoms cannot be definitely diagnosed
Original Article. Abstract
 Original Article Diagnostic accuracy of antinuclear antibodies and anti-double stranded DNA antibodies in patients of systemic lupus erythematosus presenting with dermatological features Attiya Tareen*,
Original Article Diagnostic accuracy of antinuclear antibodies and anti-double stranded DNA antibodies in patients of systemic lupus erythematosus presenting with dermatological features Attiya Tareen*,
Clinical Policy: Canakinumab (Ilaris) Reference Number: ERX.SPA.04 Effective Date:
 Clinical Policy: (Ilaris) Reference Number: ERX.SPA.04 Effective Date: 04.01.17 Last Review Date: 05.18 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
Clinical Policy: (Ilaris) Reference Number: ERX.SPA.04 Effective Date: 04.01.17 Last Review Date: 05.18 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
MANAGING THE PATIENT WITH POSITIVE ANA
 MANAGING THE PATIENT WITH POSITIVE ANA Rafael F. Rivas-Chacon, M.D. Disclosures Grant/Research support for: Pfizer Study JIA A3921104 Tofacitinib not related to this presentation 1 Positive Antinuclear
MANAGING THE PATIENT WITH POSITIVE ANA Rafael F. Rivas-Chacon, M.D. Disclosures Grant/Research support for: Pfizer Study JIA A3921104 Tofacitinib not related to this presentation 1 Positive Antinuclear
EXTENDED REPORT. Clinical and epidemiological research
 1 Allegheny Singer Research Institute, West Penn Allegheny Health System, Temple University School of Medicine, Pittsburgh, Pennsylvania, USA 2 Instituto Nacional de Ciencias Medicas y Nutricion Salvador
1 Allegheny Singer Research Institute, West Penn Allegheny Health System, Temple University School of Medicine, Pittsburgh, Pennsylvania, USA 2 Instituto Nacional de Ciencias Medicas y Nutricion Salvador
Medical Policy An independent licensee of the Blue Cross Blue Shield Association
 Afrezza Page 1 of 7 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Afrezza (human insulin) Prime Therapeutics will review Prior Authorization requests Prior Authorization
Afrezza Page 1 of 7 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Afrezza (human insulin) Prime Therapeutics will review Prior Authorization requests Prior Authorization
Name of Policy: Therapeutic Apheresis, with Extracorporeal Column Immunoadsorption and Plasma Reinfusion
 Name of Policy: Therapeutic Apheresis, with Extracorporeal Column Immunoadsorption and Plasma Reinfusion Policy #: 010 Latest Review Date: December 2008 Category: Surgical Policy Grade: Effective March
Name of Policy: Therapeutic Apheresis, with Extracorporeal Column Immunoadsorption and Plasma Reinfusion Policy #: 010 Latest Review Date: December 2008 Category: Surgical Policy Grade: Effective March
Exclusion Criteria. Required Medical Documentation
 Partnership HealthPlan of California: New and Revised, th Quarter P & T 2016 Effective //2016 Generic substitution required when there is an FDA approved generic equivalent; additional criteria apply for
Partnership HealthPlan of California: New and Revised, th Quarter P & T 2016 Effective //2016 Generic substitution required when there is an FDA approved generic equivalent; additional criteria apply for
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Ilaris) Reference Number: CP.PHAR.246 Effective Date: 08.01.16 Last Review Date: 05.18 Line of Business: Commercial, Medicaid Revision Log See Important Reminder at the end of this policy
Clinical Policy: (Ilaris) Reference Number: CP.PHAR.246 Effective Date: 08.01.16 Last Review Date: 05.18 Line of Business: Commercial, Medicaid Revision Log See Important Reminder at the end of this policy
Late onset systemic lupus erythematosus in southern Chinese. Citation Annals Of The Rheumatic Diseases, 1998, v. 57 n. 7, p.
 Title Late onset systemic lupus erythematosus in southern Chinese Author(s) Ho, CTK; Mok, CC; Lau, CS; Wong, RWS Citation Annals Of The Rheumatic Diseases, 1998, v. 57 n. 7, p. 437-440 Issued Date 1998
Title Late onset systemic lupus erythematosus in southern Chinese Author(s) Ho, CTK; Mok, CC; Lau, CS; Wong, RWS Citation Annals Of The Rheumatic Diseases, 1998, v. 57 n. 7, p. 437-440 Issued Date 1998
Lupus Related Kidney Diseases. Jason Cobb MD Assistant Professor Renal Division Emory University School of Medicine October 14, 2017
 Lupus Related Kidney Diseases Jason Cobb MD Assistant Professor Renal Division Emory University School of Medicine October 14, 2017 Financial Disclosures MedImmune Lupus Nephritis Kidney Biopsy Biomarkers
Lupus Related Kidney Diseases Jason Cobb MD Assistant Professor Renal Division Emory University School of Medicine October 14, 2017 Financial Disclosures MedImmune Lupus Nephritis Kidney Biopsy Biomarkers
Technology appraisal guidance Published: 22 June 2016 nice.org.uk/guidance/ta397
 Belimumab for treating active autoantibody-positive systemic lupus erythematosus Technology appraisal guidance Published: 22 June 2016 nice.org.uk/guidance/ta397 NICE 2018. All rights reserved. Subject
Belimumab for treating active autoantibody-positive systemic lupus erythematosus Technology appraisal guidance Published: 22 June 2016 nice.org.uk/guidance/ta397 NICE 2018. All rights reserved. Subject
Clinical Policy: Colchicine (Colcrys) Reference Number: CP.PMN.123 Effective Date: Last Review Date: 05.18
 Clinical Policy: (Colcrys) Reference Number: CP.PMN.123 Effective Date: 05.01.11 Last Review Date: 05.18 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
Clinical Policy: (Colcrys) Reference Number: CP.PMN.123 Effective Date: 05.01.11 Last Review Date: 05.18 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
A cross-sectional hospital based study of clinical and. patients from central rural India
 original article A cross-sectional hospital based study of clinical and patients from central rural India Sachin Ratanlal Agrawal, Iadarilang Tiewsoh, Atulsingh Rajput, Ajitprasad Jain Access this article
original article A cross-sectional hospital based study of clinical and patients from central rural India Sachin Ratanlal Agrawal, Iadarilang Tiewsoh, Atulsingh Rajput, Ajitprasad Jain Access this article
Clinical Policy: Sarilumab (Kevzara) Reference Number: CP.PHAR.346 Effective Date: Last Review Date: 11.18
 Clinical Policy: (Kevzara) Reference Number: CP.PHAR.346 Effective Date: 07.18.17 Last Review Date: 11.18 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
Clinical Policy: (Kevzara) Reference Number: CP.PHAR.346 Effective Date: 07.18.17 Last Review Date: 11.18 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
Updates to the Alberta Drug Benefit List. Effective January 1, 2018
 Updates to the Alberta Drug Benefit List Effective January 1, 2018 Inquiries should be directed to: Pharmacy Services Alberta Blue Cross 10009 108 Street NW Edmonton AB T5J 3C5 Telephone Number: (780)
Updates to the Alberta Drug Benefit List Effective January 1, 2018 Inquiries should be directed to: Pharmacy Services Alberta Blue Cross 10009 108 Street NW Edmonton AB T5J 3C5 Telephone Number: (780)
LONGITUDINAL TREATMENT PATTERNS AND ASSOCIATED OUTCOMES IN PATIENTS WITH NEWLY DIAGNOSED SYSTEMIC LUPUS ERYTHEMATOSUS. Hong Kan 7/12/2016
 LONGITUDINAL TREATMENT PATTERNS AND ASSOCIATED OUTCOMES IN PATIENTS WITH NEWLY DIAGNOSED SYSTEMIC LUPUS ERYTHEMATOSUS Hong Kan 7/12/2016 1 Acknowledgements Research conceptualization and design, programming
LONGITUDINAL TREATMENT PATTERNS AND ASSOCIATED OUTCOMES IN PATIENTS WITH NEWLY DIAGNOSED SYSTEMIC LUPUS ERYTHEMATOSUS Hong Kan 7/12/2016 1 Acknowledgements Research conceptualization and design, programming
Thrombopoietin Receptor Agonists Prior Authorization with Quantity Limit Program Summary
 Thrombopoietin Receptor Agonists Prior Authorization with Quantity Limit Program Summary Thrombopoietin Receptor Agonists Prior Authorization with Quantity Limit OBJECTIVE The intent of the prior authorization
Thrombopoietin Receptor Agonists Prior Authorization with Quantity Limit Program Summary Thrombopoietin Receptor Agonists Prior Authorization with Quantity Limit OBJECTIVE The intent of the prior authorization
