Supplementary Tables. Psoriasis a
|
|
|
- Joseph Newman
- 6 years ago
- Views:
Transcription
1 Supplementary Tables Supplementary Table S1 Age-sex distribution of patients with psoriasis and PPP subdivided by the department specified in a claim with a diagnosis code of psoriasis and PPP in Japanese National Database Psoriasis a PPP Age Male Female Male Female (years old) N (%) N (%) N (%) N (%) All patients 0-9 3,376 (1.3) 3,177 (1.8) 396 (0.8) 382 (0.4) ,430 (1.7) 5,046 (2.9) 689 (1.5) 923 (1.0) ,800 (3.9) 9,904 (5.6) 1,821 (3.9) 3,673 (4.1) ,915 (11.0) 22,251 (12.6) 5,049 (10.7) 9,388 (10.6) ,637 (13.6) 21,588 (12.3) 7,256 (15.4) 13,237 (14.9) ,963 (17.7) 28,146 (16.0) 10,318 (21.8) 23,157 (26.0) ,510 (23.5) 33,907 (19.3) 12,384 (26.2) 23,021 (25.9) ,072 (18.9) 30,930 (17.6) 7,070 (15.0) 11,559 (13.0) 80-21,070 (8.3) 20,957 (11.9) 2,265 (4.8) 3,636 (4.1) Total 253,773 (100) 175,906 (100) 47,248 (100) 88,976 (100) Mean (SD) 57.1 (17.8) 56.1 (19.9) 55.7 (16.1) 55.5 (15.2) Patients of dermatology or rheumatology (subgroup A) b (1.0) 814 (1.6) 36 (0.4) 88 (0.3) ,137 (1.4) 1,278 (2.5) 70 (0.8) 110 (0.6) ,659 (3.3) 2,561 (5.0) 247 (2.7) 578 (3.2) ,677 (9.5) 5,582 (10.9) 828 (8.9) 1,567 (8.8) ,831 (12.1) 5,978 (11.7) 1,247 (13.4) 2,464 (13.8) ,674 (16.9) 8,607 (16.8) 1,946 (20.9) 4,777 (26.7) ,802 (25.7) 11,290 (22.0) 2,851 (30.6) 5,226 (29.2) ,355 (21.4) 9,840 (19.2) 1,638 (17.6) 2,493 (13.9) 80-6,975 (8.6) 5,319 (10.4) 442 (4.8) 623 (3.5) Total 80,920 (100) 51,269 (100) 9,305 (100) 17,890 (100) Mean (SD) 58.8 (17.1) 57.1 (19.0) 58.1 (14.8) 56.8 (14.1)
2 Supplementary Table S1 -- continued Psoriasis a PPP Age Male Female Male Female (years old) N (%) N (%) N (%) N (%) Patients of department other than dermatology or rheumatology (subgroup B) c (1.5) 232 (1.6) 53 (1.3) 30 (0.4) (1.1) 225 (1.6) 56 (1.3) 61 (0.7) (2.2) 476 (3.3) 110 (2.6) 265 (3.2) ,304 (6.8) 1,098 (7.6) 384 (9.2) 818 (9.9) ,889 (9.8) 1,380 (9.5) 600 (14.4) 1,091 (13.2) ,058 (15.9) 2,091 (14.4) 891 (21.4) 2,137 (25.9) ,797 (24.9) 3,091 (21.3) 1,165 (28.0) 2,238 (27.1) ,825 (25.1) 3,283 (22.6) 669 (16.1) 1,169 (14.2) 80-2,460 (12.8) 2,638 (18.2) 226 (5.4) 448 (5.4) Total 19,249 (100) 14,514 (100) 4,154 (100) 8,257 (100) Mean (SD) 61.7 (17.2) 61.9 (18.9) 56.8 (16.1) 56.9 (15.0) Patients of department not specified (subgroup C) d 0-9 2,278 (1.5) 2,131 (1.9) 307 (0.9) 300 (0.5) ,083 (2.0) 3,543 (3.2) 563 (1.7) 752 (1.2) ,723 (4.4) 6,867 (6.2) 1,464 (4.3) 2,830 (4.5) ,934 (12.3) 15,571 (14.1) 3,837 (11.4) 7,003 (11.1) ,917 (14.9) 14,230 (12.9) 5,409 (16.0) 9,682 (15.4) ,231 (18.4) 17,448 (15.8) 7,481 (22.1) 16,243 (25.9) ,911 (22.1) 19,526 (17.7) 8,368 (24.8) 15,557 (24.8) ,892 (16.9) 17,807 (16.2) 4,763 (14.1) 7,897 (12.6) 80-11,635 (7.6) 13,000 (11.8) 1,597 (4.7) 2,565 (4.1) Total 153,604 (100) 110,123 (100) 33,789 (100) 62,829 (100) Mean (SD) 55.6 (18.0) 54.9 (20.3) 54.9 (16.4) 54.9 (15.4)
3 Supplementary Table S1 -- continued a Patients with both of diagnosis codes of psoriasis and PPP are classified as those with psoriasis. b Patients with at least one claim with diagnosis code of psoriasis or PPP where the department was specified as dermatology or rheumatology during the 12-month observation period. c Patients with a diagnosis code of psoriasis or PPP where the department was specified as that other specialty but not as dermatology or rheumatology. d Patients with a diagnosis code of psoriasis or PPP where the department was not specified. PPP, palmoplantar pustulosis.
4 Supplementary Table S2 Diagnosis codes of psoriasis and PPP classified by the department specified in a claim in Japanese National Database Department All Specified Not patients Derma a Rheuma b Derma/ Other specified e rheuma c specialty d N f N f N f N f N f N f (%) (%) (%) (%) (%) (%) Psoriasis 130,953 2, ,189 33, , ,679 (100) (100) (100) (100) (100) (100) Plaque psoriasis 127,170 1, ,822 30, , ,705 (97.1) (69.6) (96.7) (96.5) (98.6) (97.4) Psoriasis vulgaris 103,573 1, ,002 20, , ,825 (79.1) (50.5) (78.7) (59.8) (81.4) (78.9) Psoriasis 20, ,231 11,159 44,821 77,302 (16.0) (22.9) (16.1) (33.1) (17.0) (18.0) Psoriasis vulgaris of entire body 7, , ,030 9,876 (5.5) (2.9) (5.5) (1.7) (0.8) (2.3) Psoriasis of extremities g (0.3) (0.3) (0.3) (0.2) (0.2) Psoriasis vulgaris of extremities 3, , ,417 6,002 (2.5) (1.0) (2.4) (1.1) (0.9) (1.4) Psoriasis vulgaris of lower back g (0.1) (0.1) (0.1) (0.03) (0.1) Plaque psoriasis g (0.1) (0.1) (0.03) (0.01) (0.03)
5 Supplementary Table S2 ---continued Department All Specified Not patients Derma a Rheuma b Derma/ Other specified e rheuma c specialty d N f N f N f N f N f N f (%) (%) (%) (%) (%) (%) Scalp psoriasis 1, g 1, ,395 2,832 (0.9) (0.9) (0.8) (0.5) (0.7) Seborrheic psoriasis h g (0.1) (0.1) (0.3) (0.1) (0.1) Psoriasis vulgaris of scalp 1, g 1, ,046 2,244 (0.8) (0.8) (0.4) (0.4) (0.5) Guttate psoriasis 1, g 1, ,193 2,572 (0.9) (0.9) (0.4) (0.5) (0.6) Psoriatic arthritis 3,774 1,210 4,530 2,128 1,702 8,360 (2.9) (52.6) (3.4) (6.3) (0.6) (1.9) Psoriatic arthritis 3, ,206 1,529 1,410 7,145 (2.8) (41.7) (3.2) (4.5) (0.5) (1.7) Psoriatic arthritis mutilans i ,139 (0.05) (10.6) (0.2) (1.7) (0.1) (0.3) Psoriatic spondylitis (0.1) (1.0) (0.1) (0.1) (0.01) (0.04) Pustular psoriasis 2, , ,274 4,636 (2.2) (2.9) (2.2) (1.2) (0.5) (1.1) Pustular psoriasis 2, , ,666 (2.0) (2.7) (2.0) (0.7) (0.3) (0.9) Impetigo herpetiformis j g (0.1) (0.1) (0.5) (0.1) (0.1)
6 Supplementary Table S2 ---continued Department All Specified Not patients Derma a Rheuma b Derma/ Other specified e rheuma c specialty d N f N f N f N f N f N f (%) (%) (%) (%) (%) (%) Acrodermatitis continua g (0.2) (0.2) (0.04) (0.1) (0.1) Generalized pustular psoriasis g g --- g 32 (0.02) (0.02) (0.01) Acute generalized pustular Psoriasis --- g --- g --- g --- g --- g --- g Erythrodermic psoriasis 1, , (0.8) (1.5) (0.8) (0.7) (0.1) (0.4) PPP 6, , ,620 12,625 (4.7) (3.6) (4.7) (2.4) (2.1) (2.9) PAO k (0.1) (0.9) (0.1) (0.2) (0.1) (0.1) PPP 26, ,195 12,411 96, ,224 (100) (100) (100) (100) (100) (100) PPP ,109 11,979 96, ,647 (99.8) (90.6) (99.7) (96.5) (99.9) (99.6) PAO l ,268 1,704 5,734 (2.6) (25.7) (2.8) (26.3) (1.8) (4.2)
7 Supplementary Table S2 -- continued a Patients with at least one claim with a diagnosis code of psoriasis or PPP where the department was specified as dermatology during the 12-month observation period. b Patients with at least one claim where the department was specified as rheumatology. c Patients with at least one claim where the department was specified as dermatology or rheumatology (department subgroup A). d Patients with any claim where the department was specified as other specialty but not dermatology or rheumatology (department subgroup B). e Patients with claims where the department was not specified (department subgroup C). f One patient is counted once for each code and for subclass of diagnosis and therefore sum of lower categories may exceed a total of upper category. g Less than 10 patients were found. h Internal medicine was specified for 68 (58%) of 118 patients with Seborrheic psoriasis in the department of other specialty. i Orthopedics was specified for 470 (82%) of 575 patients with Psoriatic arthritis mutilans in the department of other specialty. j Internal medicine was specified for 61 (38%) of 160 patients with Impetigo herpetiformis in the department of other specialty. k Internal medicine was specified for 54 (74%) of 73 patients with PAO in the department of other specialty under the broad disease category of psoriasis. l Internal medicine was specified for 2978 (91%) of 3268 patients with PAO in the department of other specialty under the broad disease category of PPP. PPP, palmoplantar pustulosis; PAO, pustulotic arthro-osteitis.
8 Supplementary Table S3 Use of healthcare service in each of 12 months during the study period in patients with psoriasis and PPP subdivided by the department in a claim with diagnosis code of psoriasis and PPP in Japanese National Database All Dermatology Other Department or rheumatology specialty not specified (subgroup A) (subgroup B) (subgroup C) Year and Month N b % N b % N b % N b % Patients with Psoriasis 2010 April 155, , , , May 157, , , , June 166, , , , July 168, , , , August 161, , , , September 162, , , , October 167, , , , November 168, , , , December 176, , , , January 164, , , , February 165, , , , March 177, , , , Total 429, , , ,
9 Supplementary Table S3 -- continued All Dermatology Other Department or rheumatology specialty not specified (subgroup A) (subgroup B) (subgroup C) Year and Month N b % N b % N b % N b % Patients with PPP 2010 April 41, , , , May 45, , , , June 52, , , , July 55, , , , August 52, , , , September 52, , , , October 52, , , , November 48, , , , December 49, , , , January 43, , , , February 44, , , , March 47, , , , Total 136, , , , a Patients with at least one claim with diagnosis code of psoriasis or PPP where the department was specified as either dermatology or rheumatology ( Dermatology or rheumatology ) or other specialty but not dermatology or rheumatology ( Other specialty ) during the 12-month observation period. b Number of patients with a claim with a diagnosis code of psoriasis or PPP. PPP, palmoplantar pustulosis.
10 Supplementary Table S4 Seasonal variation of use of healthcare service in patients with a diagnosis code of psoriasis and PPP in Japanese National Database Use of health-care service 2010 July 2011 January Psoriasis PPP or August or February N a % N a % All patients Yes Yes 147, , Yes No 78, , No Yes 80, , No No 123, , Total 429, , Difference (95% CI) b -0.33% (-0.51 to -0.15%) 10.0% (9.8 to 10.3%) P value c < Patients of dermatology or rheumatology (subgroup A) d Yes Yes 58, , Yes No 22, , No Yes 22, , No No 28, , Total 132, , Difference (95% CI) b 0.04% (-0.27 to 0.36%) 7.7% (7.1 to 8.4%) P value c 0.79 < Patients of department of other specialty (subgroup B) e Yes Yes 14, , Yes No 5, , No Yes 5, , No No 8, , Total 33, , Difference (95% CI) b 0.64% (0.03 to 1.25%) 6.6% (5.7 to 7.6%) P value c 0.04 <0.0001
11 Supplementary Table S4 --- continued Use of health-care service 2010 July 2011 January Psoriasis PPP or August or February N a % N a % Patients of department not specified (subgroup C) f Yes Yes 74, , Yes No 50, , No Yes 52, , No No 86, , Total 263, , Difference (95% CI) b -0.64% (-0.88 to -0.40%) 11.1% (10.8 to 11.5%) P value b < < a Number of patients with a claim with a diagnosis code of psoriasis or PPP. b Difference between the proportion of patients who used the healthcare service in the hottest season (July or August 2010) and the proportion of patients who used the service in the coldest season (January or February of 2011). c The result of paired t-test is shown. d Patients with at least one claim with diagnosis code of psoriasis or PPP where the department was specified as dermatology or rheumatology during the 12-month observation period. e Patients with a diagnosis code of psoriasis or PPP where the department was specified as other specialty but not as dermatology or rheumatology. f Patients with a diagnosis code of psoriasis or PPP where the department was not specified. PPP, palmoplantar pustulosis.
12 Supplementary Table S5 Treatments for psoriasis and PPP subdivided by the department specified in a claim with a diagnosis code of psoriasis and PPP in Japanese National Database Psoriasis PPP Psoriatic Male arthritis Male PAO N a N Age N N a N Age N (%) (%) (SD) (%) (%) (%) (SD) (%) All patients N=429,679 N=136,224 Systemic therapy Biologics 3,291 2, , (0.8) (64.8) (14.1) (33.8) (0.2) (25.7) (13.6) (7.9) Methotrexate 1,680 1, (0.4) (63.8) (15.5) (28.6) (0.1) (30.5) (13.4) (10.9) Ciclosporin 21,997 13, , (5.1) (62.8) (16.2) (7.3) (0.2) (39.0) (13.8) (8.0) Etretinate 15,481 11, , (3.6) (71.2) (13.6) (3.9) (1.4) (37.2) (11.7) (1.8) Phototherapy 21,005 12, ,408 3, (4.9) (58.9) (18.1) (1.5) (7.6) (35.1) (15.0) (1.5) Topical therapy Topical vitamin D 256, , ,698 42,903 14, (59.6) (60.9) (18.5) (1.4) (31.5) (33.3) (14.9) (1.4) Topical corticosteroid349, , , ,692 37, ,576 (81.4) (60.6) (18.1) (1.5) (80.5) (34.5) (15.3) (3.3) No treatment 38,224 19, ,851 21,209 7, ,083 (8.9) (50.7) (19.5) (4.8) (15.6) (35.5) (16.2) (9.8)
13 Supplementary Table S5 --- continued Psoriasis PPP Psoriatic Male arthritis Male PAO N a N Age N N a N Age N (%) (%) (SD) (%) (%) (%) (SD) (%) Patients of dermatology or rheumatology (subgroup A) b N=132,189 N=27,195 Systemic therapy Biologics 2,674 1, c (2.0) (68.3) (13.8) (33.5) (0.4) (25.0) (11.9) Methotrexate (0.5) (64.0) (15.6) (41.1) (0.2) (20.9) (13.0) (19.4) Ciclosporin 12,330 7, , c (9.3) (64.2) (15.8) (8.3) (0.4) (33.9) (11.9) Etretinate 9,010 6, (6.8) (70.9) (13.5) (5.6) (2.7) (36.4) (10.7) (3.5) Phototherapy 5,465 3, , (4.1) (61.3) (16.9) (3.3) (5.6) (32.6) (14.2) (2.8) Topical therapy Topical vitamin D 86,201 54, ,736 11,194 3, (65.2) (63.6) (17.7) (3.2) (41.2) (33.4) (14.0) (3.3) Topical corticosteroid112,813 70, ,580 23,415 8, (85.3) (62.6) (17.3) (3.2) (86.1) (34.2) (14.3) (2.7) No treatment 7,621 3, , (5.8) (50.0) (19.7) (5.9) (10.0) (34.4) (14.5) (4.2)
14 Supplementary Table S5 --- continued Psoriasis PPP Psoriatic Male arthritis Male PAO N a N Age N N a N Age N (%) (%) (SD) (%) (%) (%) (SD) (%) Patients of department other specialty (subgroup B) d N=33,763 N=12,411 Systemic therapy Biologics (0.9) (55.4) (15.0) (52.7) (0.4) (23.5) (11.8) (21.6) Methotrexate c (0.8) (55.1) (16.3) (50.2) (0.3) (39.5) (11.9) Ciclosporin 2,474 1, (7.3) (50.4) (16.0) (14.1) (0.3) (34.2) (16.2) (26.3) Etretinate c (1.9) (71.1) (13.5) (4.2) (0.6) (29.2) (11.8) Phototherapy (2.0) (60.2) (16.7) (5.2) (3.1) (35.0) (14.8) (19.2) Topical therapy Topical vitamin D 11,608 7, , (34.4) (63.6) (17.3) (3.4) (14.8) (31.6) (14.7) (8.4) Topical corticosteroid 19,804 12, ,037 2, ,858 (58.7) (60.6) (18.4) (4.4) (56.7) (32.7) (15.7) (26.4) No treatment 9,906 5, ,005 1, ,391 (29.3) (51.7) (17.0) (9.3) (40.3) (34.7) (14.9) (27.8)
15 Supplementary Table S5 --- continued Psoriasis PPP Psoriatic Male arthritis Male PAO N a N Age N N a N Age N (%) (%) (SD) (%) (%) (%) (SD) (%) Patients of department not specified (subgroup C) e N=263,727 N=96,618 Systemic therapy Biologics c (0.1) (44.0) (15.0) (18.9) (0.1) (27.8) (16.3) Methotrexate c (0.3) (66.8) (15.0) (9.1) (0.1) (34.8) (14.3) Ciclosporin 7,193 4, c (2.7) (64.6) (16.7) (3.2) (0.1) (46.2) (14.5) Etretinate 5,821 4, , c (2.2) (71.7) (13.6) (1.4) (1.2) (38.2) (12.4) Phototherapy 14,851 8, ,493 3, (5.6) (57.9) (18.5) (0.6) (8.8) (35.6) (15.1) (0.5) Topical therapy Topical vitamin D 158,313 93, ,877 9, (60.0) (59.3) (18.8) (0.4) (30.9) (33.3) (15.1) (0.3) Topical corticosteroid216, , ,240 27, ,095 (82.3) (59.5) (18.3) (0.4) (82.0) (34.8) (15.4) (1.4) No treatment 20,697 10, ,472 4, (7.8) (50.6) (20.3) (2.3) (13.9) (36.0) (16.9) (4.3)
16 Supplementary Table S5 -- continued a One patient is counted once for each treatment. b Patients with at least one claim with a diagnosis code of psoriasis or PPP where the department was specified as dermatology or rheumatology during the 12-month observation period. c Less than 10 patients had the treatment. d Patients with a diagnosis code of psoriasis or PPP where the department was specified as other specialty but not as dermatology or rheumatology. e Patients with a diagnosis code of psoriasis or PPP where the department was not specified. PPP, palmoplantar pustulosis; PAO, pustulotic arthro-osteitis.
17 Supplementary Table S6 Prevalence of diabetes mellitus, hyperlipidemia and hypertension in severity classes I to IV of patients with psoriasis and PPP subdivided by the department specified in the claim in Japanese National Database All patients Patients of Patients of Patients of Severity Class I (systemic therapy) dermatol or rheumatol a other department b department not specified c (subgroup A) (subgroup B) (subgroup C) Psoriasis PPP Psoriasis PPP Psoriasis PPP Psoriasis PPP Total N 39,796 2,588 22,641 1,009 3, ,635 1,385 Diabetes mellitus N d 5, , , Prevalence e (%) Difference f (%) (95%CI) (2.7,3.4) (-0.5,2.1) (1.6,2.5) (-1.4,3.9) (4.5,7.1) (-5.8,4.6) (0.9,1.9) (-1.1,1.8) Hyperlipidemia N d 8, , , , Prevalence e (%) Difference f (%) (95%CI) (2.8,3.6) (-0.6,2.6) (1.9,3.1) (-0.9,5.0) (4.8,7.7) (-8.2,2.8) (0.8,2.1) (-1.5,2.4) Hypertension N d 11, , , , Prevalence e (%) Difference f (%) (95%CI) (4.7,5.6) (-1.0,2.3) (5.3,6.6) (-3.0,3.0) (1.9,4.9) (-11.9,0.5) (0.8,2.2) (-1.0,3.2)
18 Supplementary Table S6 -- continued All patients Patients of Patients of Patients of dermatol or rheumatol a other department b department not specified c (subgroup A) (subgroup B) (subgroup C) Psoriasis PPP Psoriasis PPP Psoriasis PPP Psoriasis PPP Severity Class II (phototherapy without systemic therapy) Total N 18,833 10,153 4,332 1, ,887 8,322 Diabetes mellitus N d 1, , Prevalence e (%) Difference f (%) (95%CI) (-1.0,-0.2) (-1.4,-0.2) (1.6,2.5) (-1.4,3.9) (-4.1,0.8) (-4.9,1.3) (-0.6,0.3) (-0.9,0.5) Hyperlipidemia N d 3,125 1, ,183 1,392 Prevalence e (%) Difference f (%) (95%CI) (-1.3,-0.3) (-0.7,1.0) (1.9,3.1) (-0.9,5.0) (-3.9,1.9) (-5.0,2.8) (-1.2,0.0) (-0.6,1.2) Hypertension N d 4,369 1,885 1, ,004 1,511 Prevalence e (%) Difference f (%) (95%CI) (-1.2,-0.1) (-0.9,0.9) (5.3,6.6) (-3.0,3.0) (-6.6,-0.9) (-5.2,3.2) (-1.1,0.2) (-0.7,1.2)
19 Supplementary Table 6 -- continued All patients Patients of Patients of Patients of dermatol or rheumatol a other department b department not specified c (subgroup A) (subgroup B) (subgroup C) Psoriasis PPP Psoriasis PPP Psoriasis PPP Psoriasis PPP Severity Class III (topical therapy only) Total N 332, ,274 97,595 22,002 19,723 6, ,508 73,439 Diabetes mellitus N d 32,378 7,583 11,886 2,141 2, ,507 4,717 Prevalence e (%) Difference f (%) ref ref ref ref ref ref ref ref Hyperlipidemia N d 56,561 17,783 19,006 4,434 4,206 1,315 33,289 12,034 Prevalence e (%) Difference f (%) ref ref ref ref ref ref ref ref Hypertension N d 78,996 19,322 26,307 4,781 6,484 1,548 46,205 12,993 Prevalence e (%) Difference f (%) ref ref ref ref ref ref ref ref
20 Supplementary Table S6 -- continued All patients Patients of Patients of Patients of dermatol or rheumatol a other department b department not specified c (subgroup A) (subgroup B) (subgroup C) Psoriasis PPP Psoriasis PPP Psoriasis PPP Psoriasis PPP Severity Class IV (No treatment) Total N 38,224 21,209 7,621 2,732 9,906 5,005 20,697 13,472 Diabetes mellitus N d 4,669 2, , ,185 1,178 Prevalence e (%) Difference f (%) (95%CI) (1.6,2.2) (1.5,2.4) (-1.3,0.1) (-1.2,1.5) (0.6,2.4) (0.2,2.8) (1.0,1.8) (1.2,2.3) Hyperlipidemia N d 7,771 4,283 1, ,340 1,087 4,113 2,660 Prevalence e (%) Difference f (%) (95%CI) (1.0,1.8) (0.8,2.0) (-2.8,-1.1) (-2.0,1.1) (0.1,1.9) (-0.9,2.1) (1.4,2.4) (0.8,2.2) Hypertension N d 11,061 4,786 1, ,547 1,293 5,839 2,921 Prevalence e (%) Difference f (%) (95%CI) (1.7,2.6) (1.5,2.8) (-4.3,-2.5) (-2.6,0.9) (0.2,2.2) (-0.6,2.5) (1.7,2.8) (1.2,2.8)
21 Supplementary Table S6 -- continued a Patients with at least one claim with a diagnosis code of psoriasis or PPP where the department was specified as dermatology or rheumatology during the 12-month observation period. b Patients with a diagnosis code of psoriasis or PPP where the department was specified as other specialty but not as dermatology or rheumatology. c Patients with diagnosis code of psoriasis or PPP where the department was not specified. d Number of patients with a diagnosis code of the concurrent disease (diabetes mellitus, hyperlipidemia or hypertension) at least in one claim as well as a drug to treat the concurrent disease in the claims issued in 2 or more different months. e Prevalence was standardized to the distribution of age and sex in the entire study population of 565,903 subjects as in Table 4 in the text. f Difference of the standardized prevalence between the severity classes I, II and IV and the severity class III (topical treatment only) in each department subgroup. PPP, palmoplantar pustulosis; CI, confidence interval.
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE SCOPE
 NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE 1 Guideline title SCOPE Psoriasis: the management of psoriasis 1.1 Short title Psoriasis 2 The remit The Department of Health has asked NICE: 'to produce
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE 1 Guideline title SCOPE Psoriasis: the management of psoriasis 1.1 Short title Psoriasis 2 The remit The Department of Health has asked NICE: 'to produce
PSORIASIS PSORIASIS. Anatomic sites FACTS ABOUT PROSIASIS PSORIASIS 11/16/2017 STIGMATA OF PSORIASIS
 PSORIASIS Dr. Sami Alsuwaidan President, Saudi Society of Dermatology & Dermatologic Surgery Associate Professor and Consultant Department of Dermatology, College of Medicine King Saud University PSORIASIS
PSORIASIS Dr. Sami Alsuwaidan President, Saudi Society of Dermatology & Dermatologic Surgery Associate Professor and Consultant Department of Dermatology, College of Medicine King Saud University PSORIASIS
They are updated regularly as new NICE guidance is published. To view the latest version of this NICE Pathway see:
 bring together everything NICE says on a topic in an interactive flowchart. are interactive and designed to be used online. They are updated regularly as new NICE guidance is published. To view the latest
bring together everything NICE says on a topic in an interactive flowchart. are interactive and designed to be used online. They are updated regularly as new NICE guidance is published. To view the latest
A systematic review of treatments for severe psoriasis Griffiths C E, Clark C M, Chalmers R J, Li Wan Po A, Williams H C
 A systematic review of treatments for severe psoriasis Griffiths C E, Clark C M, Chalmers R J, Li Wan Po A, Williams H C Authors' objectives To compare the effectiveness of currently available treatments
A systematic review of treatments for severe psoriasis Griffiths C E, Clark C M, Chalmers R J, Li Wan Po A, Williams H C Authors' objectives To compare the effectiveness of currently available treatments
ETANERCEPT Generic Brand HICL GCN Exception/Other ETANERCEPT ENBREL GUIDELINES FOR USE INITIAL CRITERIA (NOTE: FOR RENEWAL CRITERIA SEE BELOW)
 Generic Brand HICL GCN Exception/Other ETANERCEPT ENBREL 18830 GUIDELINES FOR USE INITIAL CRITERIA (NOTE: FOR RENEWAL CRITERIA SEE BELOW) 1. Does the patient have a diagnosis of moderate to severe rheumatoid
Generic Brand HICL GCN Exception/Other ETANERCEPT ENBREL 18830 GUIDELINES FOR USE INITIAL CRITERIA (NOTE: FOR RENEWAL CRITERIA SEE BELOW) 1. Does the patient have a diagnosis of moderate to severe rheumatoid
KEY MESSAGES. Psoriasis patients are more prone to cardiovascular diseases, stroke, lymphoma and non-melanoma skin cancers, and increased mortality.
 KEY MESSAGES Psoriasis is a genetically determined, systemic immune-mediated chronic inflammatory disease that affects primarily the skin and joints. Psoriasis Vulgaris is characterised by well-demarcated
KEY MESSAGES Psoriasis is a genetically determined, systemic immune-mediated chronic inflammatory disease that affects primarily the skin and joints. Psoriasis Vulgaris is characterised by well-demarcated
Appendix 1: Frequently Asked Questions
 Appendix 1: Frequently Asked Questions 1. What is the funding status of Inflectra (infliximab)? Effective February 25 2016, Inflectra (infliximab) will be added to the Ontario Drug Benefit (ODB) Formulary
Appendix 1: Frequently Asked Questions 1. What is the funding status of Inflectra (infliximab)? Effective February 25 2016, Inflectra (infliximab) will be added to the Ontario Drug Benefit (ODB) Formulary
1. Background: Infliximab is administered parenterally; therefore, it is not covered under retail pharmacy benefits.
 Subject: Infliximab (Remicade ) Original Original Committee Approval: October 13, 2006 Revised Last Committee Approval: December 3, 2008 Last Review: October 19, 2007 1. Background: Infliximab is a genetically
Subject: Infliximab (Remicade ) Original Original Committee Approval: October 13, 2006 Revised Last Committee Approval: December 3, 2008 Last Review: October 19, 2007 1. Background: Infliximab is a genetically
Psoriasis. Jessica Kaffenberger, M.D. Assistant Professor of Dermatology Division of Dermatology The Ohio State University Wexner Medical Center
 Psoriasis Jessica Kaffenberger, M.D. Assistant Professor of Dermatology Division of Dermatology The Ohio State University Wexner Medical Center Learning objectives Recognize the different types of psoriasis
Psoriasis Jessica Kaffenberger, M.D. Assistant Professor of Dermatology Division of Dermatology The Ohio State University Wexner Medical Center Learning objectives Recognize the different types of psoriasis
2. Is the patient responding to Remicade therapy? Y N
 09/29/2015 Prior Authorization AETA BETTER HEALTH OF MICHIGA (MEDICAID) Remicade (MI88) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information, sign
09/29/2015 Prior Authorization AETA BETTER HEALTH OF MICHIGA (MEDICAID) Remicade (MI88) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information, sign
Prevalence of Psoriasis in Korea: A Population-Based Epidemiological Study Using the Korean National Health Insurance Database
 pissn 1013-9087ㆍeISSN 2005-3894 Ann Dermatol Vol. 29, No. 6, 2017 https://doi.org/10.5021/ad.2017.29.6.761 ORIGINAL ARTICLE Prevalence of Psoriasis in Korea: A Population-Based Epidemiological Study Using
pissn 1013-9087ㆍeISSN 2005-3894 Ann Dermatol Vol. 29, No. 6, 2017 https://doi.org/10.5021/ad.2017.29.6.761 ORIGINAL ARTICLE Prevalence of Psoriasis in Korea: A Population-Based Epidemiological Study Using
SELF-TEST QUESTIONNAIRE PSORIASIS. Plaque a raised lesion where the diameter is greater than the thickness. Number 2
 Number 2 CORE TUTORIALS IN DERMATOLOGY FOR PRIMARY CARE PDP SELF-TEST QUESTIONNAIRE AYERS ROCK, ULURU PSORIASIS NATIONAL PARK, AUSTRALIA Plaque a raised lesion where the diameter is greater than the thickness
Number 2 CORE TUTORIALS IN DERMATOLOGY FOR PRIMARY CARE PDP SELF-TEST QUESTIONNAIRE AYERS ROCK, ULURU PSORIASIS NATIONAL PARK, AUSTRALIA Plaque a raised lesion where the diameter is greater than the thickness
Predicting the Response to Phototherapy for Psoriasis Patients
 A*STAR-NHG-NTU Skin Research Grant Joint Workshop 17 October 2015 Predicting the Response to Phototherapy for Psoriasis Patients Is it possible? Dr Eugene Tan Consultant Dermatologist National Skin Centre
A*STAR-NHG-NTU Skin Research Grant Joint Workshop 17 October 2015 Predicting the Response to Phototherapy for Psoriasis Patients Is it possible? Dr Eugene Tan Consultant Dermatologist National Skin Centre
Cigna Drug and Biologic Coverage Policy
 Cigna Drug and Biologic Coverage Policy Subject Apremilast Table of Contents Coverage Policy... 1 General Background... 2 Coding/Billing Information... 4 References... 4 Effective Date... 1/1/2018 Next
Cigna Drug and Biologic Coverage Policy Subject Apremilast Table of Contents Coverage Policy... 1 General Background... 2 Coding/Billing Information... 4 References... 4 Effective Date... 1/1/2018 Next
USTEKINUMAB Generic Brand HICL GCN Exception/Other USTEKINUMAB STELARA GUIDELINES FOR USE
 Generic Brand HICL GCN Exception/Other USTEKINUMAB STELARA 36187 GUIDELINES FOR USE INITIAL CRITERIA (NOTE: FOR RENEWAL CRITERIA SEE BELOW) 1. Does the patient have a diagnosis of psoriatic arthritis (PsA)
Generic Brand HICL GCN Exception/Other USTEKINUMAB STELARA 36187 GUIDELINES FOR USE INITIAL CRITERIA (NOTE: FOR RENEWAL CRITERIA SEE BELOW) 1. Does the patient have a diagnosis of psoriatic arthritis (PsA)
Retrospective 10 years review of 100 patients with psoriasis in the Kingdom of Saudi Arabia (KSA)
 Retrospective 10 years review of 100 patients with psoriasis in the Kingdom of Saudi Arabia (KSA) Ahmed Abdullah Alhumidi King saud university, Riyadh, kingdom of Saudi Arabia Abstract Background: This
Retrospective 10 years review of 100 patients with psoriasis in the Kingdom of Saudi Arabia (KSA) Ahmed Abdullah Alhumidi King saud university, Riyadh, kingdom of Saudi Arabia Abstract Background: This
Certolizumab pegol (Cimzia) for chronic plaque psoriasis in adults
 NIHR Innovation Observatory Evidence Briefing: April 2017 Certolizumab pegol (Cimzia) for chronic plaque psoriasis in adults NIHRIO (HSRIC) ID: 2406 NICE ID: 9112 LAY SUMMARY Plaque psoriasis is the most
NIHR Innovation Observatory Evidence Briefing: April 2017 Certolizumab pegol (Cimzia) for chronic plaque psoriasis in adults NIHRIO (HSRIC) ID: 2406 NICE ID: 9112 LAY SUMMARY Plaque psoriasis is the most
Psoriasis management. A/Prof Amanda Oakley Dermatologist, Waikato
 Psoriasis management A/Prof Amanda Oakley Dermatologist, Waikato AbbVie Breakfast Session, 14 June 2014 Disclosure This breakfast session is sponsored by Abbvie Autoimmune skin disorders Psoriasis Eczema
Psoriasis management A/Prof Amanda Oakley Dermatologist, Waikato AbbVie Breakfast Session, 14 June 2014 Disclosure This breakfast session is sponsored by Abbvie Autoimmune skin disorders Psoriasis Eczema
UnitedHealthcare Pharmacy Clinical Pharmacy Programs
 UnitedHealthcare Pharmacy Clinical Pharmacy Programs Program Number 2018 P 2104-4 Program Prior Authorization/Medical Necessity Medication Taltz (ixekizumab) P&T Approval Date 8/2016, 5/2017, 2/2018 Effective
UnitedHealthcare Pharmacy Clinical Pharmacy Programs Program Number 2018 P 2104-4 Program Prior Authorization/Medical Necessity Medication Taltz (ixekizumab) P&T Approval Date 8/2016, 5/2017, 2/2018 Effective
Supplementary Online Content
 Supplementary Online Content Lynch M, Higgins E, McCormick PA, et al. The use of transient elastography and FibroTest for monitoring hepatotoxicity in patients receiving methotrexate for psoriasis. JAMA
Supplementary Online Content Lynch M, Higgins E, McCormick PA, et al. The use of transient elastography and FibroTest for monitoring hepatotoxicity in patients receiving methotrexate for psoriasis. JAMA
The retrospective evaluation of clinical and sociodemographic features of patients with psoriasis
 Annals of Medical Research DOI: 10.5455/jtomc.2018.02.032 2018;25(2):246-51 Original Article The retrospective evaluation of clinical and sociodemographic features of patients with psoriasis Mustafa Aksoy
Annals of Medical Research DOI: 10.5455/jtomc.2018.02.032 2018;25(2):246-51 Original Article The retrospective evaluation of clinical and sociodemographic features of patients with psoriasis Mustafa Aksoy
Psoriasis. Overview. Epidemiology. Epidemiology 08/08/2015. Dr Nigel Burrows Consultant Dermatologist Addenbrooke s Hospital
 Overview Psoriasis 1. Epidemiology of psoriasis 2. Histology Dr Nigel Burrows Consultant Dermatologist Addenbrooke s Hospital Aug 2015 3. Types of psoriasis 4. Assessing severity 5. Treatments Topical
Overview Psoriasis 1. Epidemiology of psoriasis 2. Histology Dr Nigel Burrows Consultant Dermatologist Addenbrooke s Hospital Aug 2015 3. Types of psoriasis 4. Assessing severity 5. Treatments Topical
QUALITY OF LIFE AMONG ADULT PATIENTS WITH PSORIASIS
 e-poster No. P2498 22 nd World Congress of Dermatology, Seoul 2011 QUALITY OF LIFE AMONG ADULT PATIENTS WITH PSORIASIS A study using data from the Malaysian Psoriasis Registry Choong-Chor Chang 1, Felix
e-poster No. P2498 22 nd World Congress of Dermatology, Seoul 2011 QUALITY OF LIFE AMONG ADULT PATIENTS WITH PSORIASIS A study using data from the Malaysian Psoriasis Registry Choong-Chor Chang 1, Felix
3. Did the patient show evidence of remission by week 8 of Humira Y N therapy?
 09/23/2015 Prior Authorization AETA BETTER HEALTH OF MICHIGA (MEDICAID) Humira (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information,
09/23/2015 Prior Authorization AETA BETTER HEALTH OF MICHIGA (MEDICAID) Humira (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information,
PSORIASIS BEST PRACTICE IN MANAGEMENT
 PSORIASIS BEST PRACTICE IN MANAGEMENT Objectives Discuss pathology of psoriasis Review types of psoriasis Review triggers and factors affecting disease severity Common comorbidity review Review first and
PSORIASIS BEST PRACTICE IN MANAGEMENT Objectives Discuss pathology of psoriasis Review types of psoriasis Review triggers and factors affecting disease severity Common comorbidity review Review first and
Psoriasis. What is Psoriasis? What causes psoriasis? Medical Topics Psoriasis
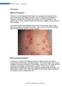 1 Psoriasis What is Psoriasis? Psoriasis is a long standing inflammatory non-contagious skin disease which waxes and wanes with triggering factors. There is a genetic predisposition in psoriasis. Internationally,
1 Psoriasis What is Psoriasis? Psoriasis is a long standing inflammatory non-contagious skin disease which waxes and wanes with triggering factors. There is a genetic predisposition in psoriasis. Internationally,
Ontario Public Drug Programs. Inflectra (infliximab) Frequently Asked Questions
 Ontario Public Drug Programs Inflectra (infliximab) Frequently Asked Questions 1. What is the funding status of Inflectra (infliximab)? Effective February 25 2016, Inflectra (infliximab) will be added
Ontario Public Drug Programs Inflectra (infliximab) Frequently Asked Questions 1. What is the funding status of Inflectra (infliximab)? Effective February 25 2016, Inflectra (infliximab) will be added
2. Does the patient have a diagnosis of Crohn s disease? Y N
 Pharmacy Prior Authorization MERC CARE PLA (MEDICAID) Stelara (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information, sign and date. Fax
Pharmacy Prior Authorization MERC CARE PLA (MEDICAID) Stelara (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information, sign and date. Fax
Inflectra Frequently Asked Questions
 Inflectra Frequently Asked Questions 1. What is the funding status of Inflectra (infliximab)? Earlier in 2016, Inflectra (infliximab) was added to the Ontario Drug Benefit (ODB) Formulary as a Limited
Inflectra Frequently Asked Questions 1. What is the funding status of Inflectra (infliximab)? Earlier in 2016, Inflectra (infliximab) was added to the Ontario Drug Benefit (ODB) Formulary as a Limited
Etanercept: a new option in paediatric plaque psoriasis
 : a new option in paediatric plaque psoriasis Steve Chaplin MSc, MRPharmS, Medical Writer, Dr David Atherton MA, MB, BChir, FRCP, Honorary Consultant in Paediatric Dermatology, Great Ormond Street Hospital
: a new option in paediatric plaque psoriasis Steve Chaplin MSc, MRPharmS, Medical Writer, Dr David Atherton MA, MB, BChir, FRCP, Honorary Consultant in Paediatric Dermatology, Great Ormond Street Hospital
Amjevita (adalimumab-atto)
 *- Florida Healthy Kids Amjevita (adalimumab-atto) Override(s) Prior Authorization Quantity Limit Medications Amjevita 20 mg/0.4 ml prefilled syringe Amjevita (adalimumab-atto) 40 mg/0.8 ml 2 #* ^ prefilled
*- Florida Healthy Kids Amjevita (adalimumab-atto) Override(s) Prior Authorization Quantity Limit Medications Amjevita 20 mg/0.4 ml prefilled syringe Amjevita (adalimumab-atto) 40 mg/0.8 ml 2 #* ^ prefilled
Request for Special Authorization Enbrel
 Certain prescription drugs call for a more detailed assessment to help ensure that they represent reasonable treatment. Special Authorization requires that you request approval from Great-West Life for
Certain prescription drugs call for a more detailed assessment to help ensure that they represent reasonable treatment. Special Authorization requires that you request approval from Great-West Life for
Psoriasis in Jordan: a single center experience
 Psoriasis in Jordan: a single center experience Shefaa Almashagbeh MD *, Deifallah Alsharari MD *, Hayat Khasawneh MD *, Diana Aljammal * MD, Hamzeh Al-housamieh MD* ABSTRACT Objectives: To evaluate the
Psoriasis in Jordan: a single center experience Shefaa Almashagbeh MD *, Deifallah Alsharari MD *, Hayat Khasawneh MD *, Diana Aljammal * MD, Hamzeh Al-housamieh MD* ABSTRACT Objectives: To evaluate the
The prevalence of psoriasis among human immunodeficiency
 CLINICAL REVIEW Psoriasis Treatment in HIV-Positive Patients: A Systematic Review of Systemic Immunosuppressive Therapies Mio Nakamura, MD; Michael Abrouk, MD; Benjamin Farahnik, MD; Tian Hao Zhu, MD;
CLINICAL REVIEW Psoriasis Treatment in HIV-Positive Patients: A Systematic Review of Systemic Immunosuppressive Therapies Mio Nakamura, MD; Michael Abrouk, MD; Benjamin Farahnik, MD; Tian Hao Zhu, MD;
Combination Topical PUVAsol with Methotrexate Versus Methotrexate in the Treatment of Palmoplantar Psoriasis Karn D, KC S
 Combination Topical PUVAsol with Methotrexate Versus Methotrexate in the Treatment of Palmoplantar Psoriasis Karn D, KC S ABSTRACT Background Department of Dermatology Dhulikhel Hospital, Kathmandu University
Combination Topical PUVAsol with Methotrexate Versus Methotrexate in the Treatment of Palmoplantar Psoriasis Karn D, KC S ABSTRACT Background Department of Dermatology Dhulikhel Hospital, Kathmandu University
Guselkumab (plaque psoriasis)
 IQWiG Reports Commission No. A18-24 Guselkumab (plaque psoriasis) Addendum to Commission A17-60 1 Addendum Commission: A18-24 Version: 1.0 Status: 27 April 2018 1 Translation of addendum A18-24 Guselkumab
IQWiG Reports Commission No. A18-24 Guselkumab (plaque psoriasis) Addendum to Commission A17-60 1 Addendum Commission: A18-24 Version: 1.0 Status: 27 April 2018 1 Translation of addendum A18-24 Guselkumab
PDP SELF-TEST QUESTIONNAIRE PSORIASIS. Plaque a raised lesion where the diameter is greater than the thickness. Number 2
 Number 2 CORE TUTORIALS IN DERMATOLOGY FOR PRIMARY CARE PDP SELF-TEST QUESTIONNAIRE AYERS ROCK, ULURU NATIONAL PARK, AUSTRALIA PSORIASIS UPDATED PDP SELF-TEST QUESTIONNAIRE 2011 Plaque a raised lesion
Number 2 CORE TUTORIALS IN DERMATOLOGY FOR PRIMARY CARE PDP SELF-TEST QUESTIONNAIRE AYERS ROCK, ULURU NATIONAL PARK, AUSTRALIA PSORIASIS UPDATED PDP SELF-TEST QUESTIONNAIRE 2011 Plaque a raised lesion
Psoriasis: Therapeutic goals
 Psoriasis: Therapeutic goals I want to die 50 45 impetiginization infliximab 600 40 35 30 400 25 20 15 200 10 5 0 22-ene 21-feb 23-mar 22-abr 22- may Efalizumab 6 doses: flare + REBOUND CSA 3 21-jun 21-jul
Psoriasis: Therapeutic goals I want to die 50 45 impetiginization infliximab 600 40 35 30 400 25 20 15 200 10 5 0 22-ene 21-feb 23-mar 22-abr 22- may Efalizumab 6 doses: flare + REBOUND CSA 3 21-jun 21-jul
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE. Proposed Health Technology Appraisal
 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Proposed Health Technology Appraisal Dimethyl fumarate for treating moderate to severe Draft scope (pre-referral) Draft remit/appraisal objective To appraise
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Proposed Health Technology Appraisal Dimethyl fumarate for treating moderate to severe Draft scope (pre-referral) Draft remit/appraisal objective To appraise
CPT Codes: The following ICD-10-CM codes support the medical necessity of CPT code 82306:
 CPT s: 82306 Vitamin D; 25 hydroxy, includes fraction(s), if performed 82652 Vitamin D; 1, 25 dihydroxy, includes fraction(s), if performed The following ICD-10-CM codes support the medical necessity of
CPT s: 82306 Vitamin D; 25 hydroxy, includes fraction(s), if performed 82652 Vitamin D; 1, 25 dihydroxy, includes fraction(s), if performed The following ICD-10-CM codes support the medical necessity of
List of Chronic Conditions under the Community Health Assist Scheme (CHAS)
 List of Chronic s under the Community Health Assist Scheme (CHAS) Chronic Diabetes Mellitus Hypertension Lipid Disorders Stroke Asthma Chronic Obstructive Pulmonary Disease (COPD) Schizophrenia Major Depression
List of Chronic s under the Community Health Assist Scheme (CHAS) Chronic Diabetes Mellitus Hypertension Lipid Disorders Stroke Asthma Chronic Obstructive Pulmonary Disease (COPD) Schizophrenia Major Depression
Vitamin D Assay Testing For services performed on or after
 2018 MEDICARE LOCAL COVERAGE DETERMINATION (LCD) - L36692 CPT CODES: 82306, 82652 Vitamin D Assay Testing For services performed on or after 2-3-2017 DLS TEST CODE AND NAME 49907 (1,25 DIHYDROXY) (CPT
2018 MEDICARE LOCAL COVERAGE DETERMINATION (LCD) - L36692 CPT CODES: 82306, 82652 Vitamin D Assay Testing For services performed on or after 2-3-2017 DLS TEST CODE AND NAME 49907 (1,25 DIHYDROXY) (CPT
50 microgram/g Calcipotriol and 500 microgram/g betamethasone (as dipropionate).
 DUPISOR Composition Gel 50 microgram/g Calcipotriol and 500 microgram/g betamethasone (as dipropionate). Action Calcipotriol is a non-steroidal antipsoriatic agent, derived from vitamin D. Calcipotriol
DUPISOR Composition Gel 50 microgram/g Calcipotriol and 500 microgram/g betamethasone (as dipropionate). Action Calcipotriol is a non-steroidal antipsoriatic agent, derived from vitamin D. Calcipotriol
ADALIMUMAB Generic Brand HICL GCN Exception/Other ADALIMUMAB HUMIRA GUIDELINES FOR USE INITIAL CRITERIA (NOTE: FOR RENEWAL CRITERIA SEE BELOW)
 Generic Brand HICL GCN Exception/Other ADALIMUMAB HUMIRA 24800 GUIDELINES FOR USE INITIAL CRITERIA (NOTE: FOR RENEWAL CRITERIA SEE BELOW) 1. Does the patient have a diagnosis of moderate to severe rheumatoid
Generic Brand HICL GCN Exception/Other ADALIMUMAB HUMIRA 24800 GUIDELINES FOR USE INITIAL CRITERIA (NOTE: FOR RENEWAL CRITERIA SEE BELOW) 1. Does the patient have a diagnosis of moderate to severe rheumatoid
STELARA (USTEKINUMAB)
 Oxford STELARA (USTEKINUMAB) UnitedHealthcare Oxford Clinical Policy Policy Number: PHARMACY 218.13 T2 Effective Date: February 1, 2017 Table of Contents Page INSTRUCTIONS FOR USE... 1 CONDITIONS OF COVERAGE...
Oxford STELARA (USTEKINUMAB) UnitedHealthcare Oxford Clinical Policy Policy Number: PHARMACY 218.13 T2 Effective Date: February 1, 2017 Table of Contents Page INSTRUCTIONS FOR USE... 1 CONDITIONS OF COVERAGE...
HARVARD PILGRIM HEALTH CARE RECOMMENDED MEDICATION REQUEST GUIDELINES
 Generic Brand HICL GCN Exception/Other CERTOLIZUMAB PEGOL CIMZIA 35554 GUIDELINES FOR USE INITIAL CRITERIA (NOTE: FOR RENEWAL CRITERIA SEE BELOW) 1. Is the request for a patient with a diagnosis of moderate
Generic Brand HICL GCN Exception/Other CERTOLIZUMAB PEGOL CIMZIA 35554 GUIDELINES FOR USE INITIAL CRITERIA (NOTE: FOR RENEWAL CRITERIA SEE BELOW) 1. Is the request for a patient with a diagnosis of moderate
EXAMINING THE CRUCIAL COALITION BETWEEN DERMATOLOGY AND RHEUMATOLOGY IN PSORIATIC ARTHRITIS
 EXAMINING THE CRUCIAL COALITION BETWEEN DERMATOLOGY AND RHEUMATOLOGY IN PSORIATIC ARTHRITIS ACTIVITY 1: EARLY COLLABORATION IN THE TREATMENT OF PSA Key Slides COMMON COMORBIDITIES OF PSORIATIC DISEASE
EXAMINING THE CRUCIAL COALITION BETWEEN DERMATOLOGY AND RHEUMATOLOGY IN PSORIATIC ARTHRITIS ACTIVITY 1: EARLY COLLABORATION IN THE TREATMENT OF PSA Key Slides COMMON COMORBIDITIES OF PSORIATIC DISEASE
Psoriasi e rischio CV
 Psoriasi e rischio CV Claudio Borghi Dipartimento di Scienze Mediche e Chirurgiche Università di Bologna David Plunkert 5 Biggest Heart Risks for Men Plos Med, 2006 Sequenza di eventi che causano malattie
Psoriasi e rischio CV Claudio Borghi Dipartimento di Scienze Mediche e Chirurgiche Università di Bologna David Plunkert 5 Biggest Heart Risks for Men Plos Med, 2006 Sequenza di eventi che causano malattie
Psoriasis. Andrei Metelitsa, MD, FRCPC, FAAD Clinical Associate Professor, Dermatology, U of C Co-Director, Institute for Skin Advancement
 Psoriasis Andrei Metelitsa, MD, FRCPC, FAAD Clinical Associate Professor, Dermatology, U of C Co-Director, Institute for Skin Advancement Copyright 2017 by Sea Courses Inc. All rights reserved. No part
Psoriasis Andrei Metelitsa, MD, FRCPC, FAAD Clinical Associate Professor, Dermatology, U of C Co-Director, Institute for Skin Advancement Copyright 2017 by Sea Courses Inc. All rights reserved. No part
Clinical Policy: Secukinumab (Cosentyx) Reference Number: CP.PHAR.261 Effective Date: 08/16 Last Review Date: 08/17
 Clinical Policy: (Cosentyx) Reference Number: CP.PHAR.261 Effective Date: 08/16 Last Review Date: 08/17 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
Clinical Policy: (Cosentyx) Reference Number: CP.PHAR.261 Effective Date: 08/16 Last Review Date: 08/17 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
Psoriasis: A Cutaneous or Systemic Disease
 Psoriasis: A Cutaneous or Systemic Disease Ian D.R. Landells, MD, FRCPC Clinical Chief Dermatology, Eastern Health Clinical Assistant Professor Disciplines of Medicine and Paediatrics Faculty of Medicine
Psoriasis: A Cutaneous or Systemic Disease Ian D.R. Landells, MD, FRCPC Clinical Chief Dermatology, Eastern Health Clinical Assistant Professor Disciplines of Medicine and Paediatrics Faculty of Medicine
Clinical and immunogenetic characteristics of psoriatic arthritis: a single-center experience from South India
 IJRCI. 2013;1(1):OA1 ORIGINAL RESEARCH Clinical and immunogenetic characteristics of psoriatic arthritis: a single-center experience from South India Mithun CB 1, Paul T Antony 2,Christina M Mariaselvam
IJRCI. 2013;1(1):OA1 ORIGINAL RESEARCH Clinical and immunogenetic characteristics of psoriatic arthritis: a single-center experience from South India Mithun CB 1, Paul T Antony 2,Christina M Mariaselvam
Clinical Policy: Apremilast (Otezla) Reference Number: CP.PHAR.245 Effective Date: 08/16 Last Review Date 08/17
 Clinical Policy: (Otezla) Reference Number: CP.PHAR.245 Effective Date: 08/16 Last Review Date 08/17 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
Clinical Policy: (Otezla) Reference Number: CP.PHAR.245 Effective Date: 08/16 Last Review Date 08/17 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
Spondyloarthropathies: Disease Perception Limits Market
 Spondyloarthropathies: Disease Perception Limits Market Psoriatic arthritis and ankylosing spondylitis form part of the group of diseases known as the spondyloarthropathies. Psoriatic arthritis is a form
Spondyloarthropathies: Disease Perception Limits Market Psoriatic arthritis and ankylosing spondylitis form part of the group of diseases known as the spondyloarthropathies. Psoriatic arthritis is a form
Effects of Acupuncture on Chinese Adult Patients with Psoriatic Arthritis: A Prospective Cohort Study
 Effects of Acupuncture on Chinese Adult Patients with Psoriatic Arthritis: A Prospective Cohort Study Peiyi Chen 1, Tiantian Xin 2* and Yingchun Zeng 3,4 1 School of Nursing, Guangzhou University of Chinese
Effects of Acupuncture on Chinese Adult Patients with Psoriatic Arthritis: A Prospective Cohort Study Peiyi Chen 1, Tiantian Xin 2* and Yingchun Zeng 3,4 1 School of Nursing, Guangzhou University of Chinese
STELARA (USTEKINUMAB)
 STELARA (USTEKINUMAB) UnitedHealthcare Commercial Medical Benefit Drug Policy Policy Number: 2017D0045J Effective Date: July 1, 2017 Table of Contents Page INSTRUCTIONS FOR USE... 1 BENEFIT CONSIDERATIONS...
STELARA (USTEKINUMAB) UnitedHealthcare Commercial Medical Benefit Drug Policy Policy Number: 2017D0045J Effective Date: July 1, 2017 Table of Contents Page INSTRUCTIONS FOR USE... 1 BENEFIT CONSIDERATIONS...
Clinical features of 53 cases with pustulotic arthroosteitis
 Annals of the Rheumatic Disease, 1981, 4G, 547-553 Clinical features of 53 cases with pustulotic arthroosteitis H. SONOZAKI1, H. MITSUI2, Y. MIYANAGA2, K. OKITSU2, M. IGARASHI2, Y. HAYASHI2, M. MATSUURA3,
Annals of the Rheumatic Disease, 1981, 4G, 547-553 Clinical features of 53 cases with pustulotic arthroosteitis H. SONOZAKI1, H. MITSUI2, Y. MIYANAGA2, K. OKITSU2, M. IGARASHI2, Y. HAYASHI2, M. MATSUURA3,
STELARA (USTEKINUMAB)
 STELARA (USTEKINUMAB) UnitedHealthcare Oxford Clinical Policy Policy Number: PHARMACY 218.15 T2 Effective Date: July 1, 2017 Table of Contents Page INSTRUCTIONS FOR USE... 1 CONDITIONS OF COVERAGE... 1
STELARA (USTEKINUMAB) UnitedHealthcare Oxford Clinical Policy Policy Number: PHARMACY 218.15 T2 Effective Date: July 1, 2017 Table of Contents Page INSTRUCTIONS FOR USE... 1 CONDITIONS OF COVERAGE... 1
WHAT CAUSES PSORIASIS TO FLARE UP WHAT CAUSES PSORIASIS TO PDF PSORIASIS PSORIATIC ARTHRITIS MEDLINEPLUS
 WHAT CAUSES PSORIASIS TO PDF PSORIASIS PSORIATIC ARTHRITIS MEDLINEPLUS GUTTATE PSORIASIS: CAUSES, SYMPTOMS, AND TREATMENT 1 / 5 2 / 5 3 / 5 what causes psoriasis to pdf Psoriasis is a skin disease that
WHAT CAUSES PSORIASIS TO PDF PSORIASIS PSORIATIC ARTHRITIS MEDLINEPLUS GUTTATE PSORIASIS: CAUSES, SYMPTOMS, AND TREATMENT 1 / 5 2 / 5 3 / 5 what causes psoriasis to pdf Psoriasis is a skin disease that
Commonly Coded Conditions in Dermatology
 Commonly Coded Conditions in Dermatology No part of this presentation may be reproduced or transmitted in any form or by any means (graphically, electronically, or mechanically, including photocopying,
Commonly Coded Conditions in Dermatology No part of this presentation may be reproduced or transmitted in any form or by any means (graphically, electronically, or mechanically, including photocopying,
chemotherapeutic agents in
 Use of biologics and chemotherapeutic agents in cutaneous emergencies: Focus on lifethreatening forms of psoriasis Alice Bendix Gottlieb MD, PhD Professor of Dermatology New York Medical College Metropolitan
Use of biologics and chemotherapeutic agents in cutaneous emergencies: Focus on lifethreatening forms of psoriasis Alice Bendix Gottlieb MD, PhD Professor of Dermatology New York Medical College Metropolitan
1. Does the patient have a diagnosis of moderate to severe polyarticular juvenile idiopathic arthritis (PJIA)?
 Humira (adalimumab) Medication Request Form (MRF) for Healthy Indiana Plan (HIP) and Hoosier Healthwise (HHW) FAX TO: (858) 790-7100 c/o MedImpact Healthcare Systems, Inc. Attn: Prior Authorization Department
Humira (adalimumab) Medication Request Form (MRF) for Healthy Indiana Plan (HIP) and Hoosier Healthwise (HHW) FAX TO: (858) 790-7100 c/o MedImpact Healthcare Systems, Inc. Attn: Prior Authorization Department
A Pilot Study. Name of investigational product:
 An Open Label, Multi Center Clinical Study to Evaluate the Efficacy and Safety of a New Topical Cosmeceutical in Relieving the Redness, Scaling and Flaking Associated with Severe Skin Conditions A Pilot
An Open Label, Multi Center Clinical Study to Evaluate the Efficacy and Safety of a New Topical Cosmeceutical in Relieving the Redness, Scaling and Flaking Associated with Severe Skin Conditions A Pilot
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Cosentyx, Cosentyx Sensoready) Reference Number: HIM.PA.SP29 Effective Date: 05/17 Last Review Date: Line of Business: Health Insurance Marketplace Coding Implications Revision Log See
Clinical Policy: (Cosentyx, Cosentyx Sensoready) Reference Number: HIM.PA.SP29 Effective Date: 05/17 Last Review Date: Line of Business: Health Insurance Marketplace Coding Implications Revision Log See
Difference Between Seborrheic Dermatitis and Psoriasis
 Difference Between Seborrheic Dermatitis and Psoriasis www.differencebetween.com Key Difference Seborrheic Dermatitis vs Psoriasis Dermatological conditions are perhaps the most worrisome diseases in the
Difference Between Seborrheic Dermatitis and Psoriasis www.differencebetween.com Key Difference Seborrheic Dermatitis vs Psoriasis Dermatological conditions are perhaps the most worrisome diseases in the
Dermatology elective for yr. 5. Natta Rajatanavin, MD. Div. of dermatology Dep. Of Medicine, Ramathibodi Hospital Mahidol University 23 rd Feb 2015
 Dermatology elective for yr. 5 Natta Rajatanavin, MD. Div. of dermatology Dep. Of Medicine, Ramathibodi Hospital Mahidol University 23 rd Feb 2015 How to diagnosis and manage eczema and psoriasis. Objectives
Dermatology elective for yr. 5 Natta Rajatanavin, MD. Div. of dermatology Dep. Of Medicine, Ramathibodi Hospital Mahidol University 23 rd Feb 2015 How to diagnosis and manage eczema and psoriasis. Objectives
2. Does the patient have a diagnosis of ulcerative colitis or Crohn s? Y N
 Pharmacy Prior Authorization AETA BETTER HEALTH LOUISIAA (MEDICAID) Remicade (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information, sign
Pharmacy Prior Authorization AETA BETTER HEALTH LOUISIAA (MEDICAID) Remicade (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information, sign
Health Status and Quality of Life in Patients with Psoriasis: An Iranian Cross-Sectional Survey
 Original Article Health Status and Quality of Life in Patients with Psoriasis: An Iranian Cross-Sectional Survey Mahshid Moradi MSc 1, Fanni Rencz MD 1,2 1, Ahmad Moradi MD 3, Orsolya Balogh PhD 1, László
Original Article Health Status and Quality of Life in Patients with Psoriasis: An Iranian Cross-Sectional Survey Mahshid Moradi MSc 1, Fanni Rencz MD 1,2 1, Ahmad Moradi MD 3, Orsolya Balogh PhD 1, László
Update on systemic therapies and emerging treatments How do I choose a systemic agent?
 Update on systemic therapies and emerging treatments How do I choose a systemic agent? Amy S. Paller, M.D. Walter J. Hamlin Professor and Chair of Dermatology Professor of Pediatrics Northwestern University
Update on systemic therapies and emerging treatments How do I choose a systemic agent? Amy S. Paller, M.D. Walter J. Hamlin Professor and Chair of Dermatology Professor of Pediatrics Northwestern University
Criteria Inclusion criteria Exclusion criteria. despite treatment with csdmards, NSAIDs, and/or previous anti-tnf therapy and/or
 Supplementary Material Table S1 Eligibility criteria (PICOS) for the SLR Criteria Inclusion criteria Exclusion criteria Population Adults (aged 18 years) with active PsA despite treatment with csdmards,
Supplementary Material Table S1 Eligibility criteria (PICOS) for the SLR Criteria Inclusion criteria Exclusion criteria Population Adults (aged 18 years) with active PsA despite treatment with csdmards,
Guttate psoriasis =ﻒدﺼﻠا ﻲﻄﻘﻨﻠا
 1 / 69 Psoriasis Psoriasis may be divided into psoriasis vulgaris, generalized pustular psoriasis, and localized pustular ps Psoriasis Vulgaris Clinical Features 2 / 69 Psoriasis vulgaris is a common chronic
1 / 69 Psoriasis Psoriasis may be divided into psoriasis vulgaris, generalized pustular psoriasis, and localized pustular ps Psoriasis Vulgaris Clinical Features 2 / 69 Psoriasis vulgaris is a common chronic
Psoraisis = ﻒدﺼﻠا 1 / 84
 1 / 84 2 / 84 3 / 84 4 / 84 5 / 84 6 / 84 Psoriasis Psoriasis may be divided into psoriasis vulgaris, generalized pustular psoriasis, and localized pustular ps Psoriasis Vulgaris Clinical Features 7 /
1 / 84 2 / 84 3 / 84 4 / 84 5 / 84 6 / 84 Psoriasis Psoriasis may be divided into psoriasis vulgaris, generalized pustular psoriasis, and localized pustular ps Psoriasis Vulgaris Clinical Features 7 /
INFLIXIMAB (REMICADE, INFLECTRA, RENFLEXIS )
 UnitedHealthcare Commercial Medical Benefit Drug Policy INFLIXIMAB (REMICADE, INFLECTRA, RENFLEXIS ) Policy Number: 2019D0004X Effective Date: January 1, 2019 Table of Contents Page INSTRUCTIONS FOR USE...
UnitedHealthcare Commercial Medical Benefit Drug Policy INFLIXIMAB (REMICADE, INFLECTRA, RENFLEXIS ) Policy Number: 2019D0004X Effective Date: January 1, 2019 Table of Contents Page INSTRUCTIONS FOR USE...
TREATMENT OPTIONS FOR PSORIASIS. Sandra Hanlon Dermatology Senior Charge Nurse NHS Ayrshire and Arran 07/03/17
 TREATMENT OPTIONS FOR PSORIASIS Sandra Hanlon Dermatology Senior Charge Nurse NHS Ayrshire and Arran 07/03/17 PSORIASIS A chronic, non-infectious inflammatory skin condition that has no cure Characterised
TREATMENT OPTIONS FOR PSORIASIS Sandra Hanlon Dermatology Senior Charge Nurse NHS Ayrshire and Arran 07/03/17 PSORIASIS A chronic, non-infectious inflammatory skin condition that has no cure Characterised
A study of treatment modalities in psoriasis in dermatology outpatient department of a tertiary care teaching hospital
 Original article A study of treatment modalities in psoriasis in dermatology outpatient department of a tertiary care teaching hospital 1Y Roja Ramani, 2 Benu Panigrahy, 3 Sailenkumar Mishra, 4 BTPS Singh
Original article A study of treatment modalities in psoriasis in dermatology outpatient department of a tertiary care teaching hospital 1Y Roja Ramani, 2 Benu Panigrahy, 3 Sailenkumar Mishra, 4 BTPS Singh
Amjevita (adalimumab-atto) CG-DRUG-64, CG-DRUG-65
 Market DC Amjevita (adalimumab-atto) CG-DRUG-64, CG-DRUG-65 Override(s) Prior Authorization Quantity Limit Medications Amjevita 20 mg/0.4 ml prefilled syringe Amjevita (adalimumab-atto) 40 mg/0.8 ml 2
Market DC Amjevita (adalimumab-atto) CG-DRUG-64, CG-DRUG-65 Override(s) Prior Authorization Quantity Limit Medications Amjevita 20 mg/0.4 ml prefilled syringe Amjevita (adalimumab-atto) 40 mg/0.8 ml 2
Supplementary Appendix
 Supplementary Appendix Increased Risk of Atrial Fibrillation and Thromboembolism in Patients with Severe Psoriasis: a Nationwide Population-based Study Tae-Min Rhee, MD 1, Ji Hyun Lee, MD 2, Eue-Keun Choi,
Supplementary Appendix Increased Risk of Atrial Fibrillation and Thromboembolism in Patients with Severe Psoriasis: a Nationwide Population-based Study Tae-Min Rhee, MD 1, Ji Hyun Lee, MD 2, Eue-Keun Choi,
This questionnaire was used both during the face-to-face interviews with the
 Additional file 1: Primary research questionnaire This questionnaire was used both during the face-to-face interviews with the dermatologists and during the expert panel 1. During the last month, how many
Additional file 1: Primary research questionnaire This questionnaire was used both during the face-to-face interviews with the dermatologists and during the expert panel 1. During the last month, how many
Developments in Psoriasis Management Celgene Corporation 07/14 USII-CELG140030
 Developments in Psoriasis Management 2014 Celgene Corporation 07/14 USII-CELG140030 Psoriasis (PsO) Is a Chronic Inflammatory Disease That Can Impact Patients Health-Related Quality of Life PsO is a chronic
Developments in Psoriasis Management 2014 Celgene Corporation 07/14 USII-CELG140030 Psoriasis (PsO) Is a Chronic Inflammatory Disease That Can Impact Patients Health-Related Quality of Life PsO is a chronic
Horizon Scanning Centre March Ixekizumab for moderate to severe chronic plaque psoriasis SUMMARY NIHR HSC ID: 5209
 Horizon Scanning Centre March 2015 Ixekizumab for moderate to severe chronic plaque psoriasis SUMMARY NIHR HSC ID: 5209 This briefing is based on information available at the time of research and a limited
Horizon Scanning Centre March 2015 Ixekizumab for moderate to severe chronic plaque psoriasis SUMMARY NIHR HSC ID: 5209 This briefing is based on information available at the time of research and a limited
Pediatric Use: Safety and effectiveness of Ustekinumab (STELARA ) in pediatric patients have not been evaluated.
 Original Issue Date (Created): January 1, 2010 Most Recent Review Date (Revised): January 28, 2014 Effective Date: April 1, 2014 I. POLICY Preauthorization Requirements for Ustekinumab (STELARA ) Note:
Original Issue Date (Created): January 1, 2010 Most Recent Review Date (Revised): January 28, 2014 Effective Date: April 1, 2014 I. POLICY Preauthorization Requirements for Ustekinumab (STELARA ) Note:
The Natural History of Psoriasis and Treatment Goals
 The Natural History of Psoriasis and Treatment Goals Psoriasis Epidemiology Prevalence Affects 2 3% of adult population (>7 million in US) Caucasians: 25% 2.5% African Americans: 1.3% (more likely to have
The Natural History of Psoriasis and Treatment Goals Psoriasis Epidemiology Prevalence Affects 2 3% of adult population (>7 million in US) Caucasians: 25% 2.5% African Americans: 1.3% (more likely to have
Approximately 3% of the US adult population,
 Disease Burden and Quality of Life in Psoriasis Patients With and Without Comorbid Psoriatic Arthritis: Results From National Psoriasis Foundation Panel Surveys Emily Edson-Heredia, MPH; Baojin Zhu, PhD;
Disease Burden and Quality of Life in Psoriasis Patients With and Without Comorbid Psoriatic Arthritis: Results From National Psoriasis Foundation Panel Surveys Emily Edson-Heredia, MPH; Baojin Zhu, PhD;
Appendix 1 J.M. Weinberg, M. Lebwohl (eds.), Advances in Psoriasis, 315 DOI / , Springer-Verlag London 2014
 Appendix 1 J.M. Weinberg, M. Lebwohl (eds.), Advances in Psoriasis, DOI 10.1007/978-1-4471-4432-8, Springer-Verlag London 2014 315 316 Appendix 1 Appendix 2 317 318 Appendix 2 Appendix 3 Sample letter
Appendix 1 J.M. Weinberg, M. Lebwohl (eds.), Advances in Psoriasis, DOI 10.1007/978-1-4471-4432-8, Springer-Verlag London 2014 315 316 Appendix 1 Appendix 2 317 318 Appendix 2 Appendix 3 Sample letter
Brodalumab for treating moderate to severe plaque psoriasis [ID878]
![Brodalumab for treating moderate to severe plaque psoriasis [ID878] Brodalumab for treating moderate to severe plaque psoriasis [ID878]](/thumbs/82/85279721.jpg) Brodalumab for treating moderate to severe plaque psoriasis [ID878] Thank you for agreeing to give us your organisation s views on this technology and its possible use in the NHS. You can provide a unique
Brodalumab for treating moderate to severe plaque psoriasis [ID878] Thank you for agreeing to give us your organisation s views on this technology and its possible use in the NHS. You can provide a unique
Combination Nonbiologic Therapy in Psoriasis. Sushil Tahiliani, MBBS, MD
 Combination Nonbiologic Therapy in Psoriasis Sushil Tahiliani, MBBS, MD Agenda Rationale Preferred and less preferred combination Morphology-specific preferred combinations Doses used in combinations Potential
Combination Nonbiologic Therapy in Psoriasis Sushil Tahiliani, MBBS, MD Agenda Rationale Preferred and less preferred combination Morphology-specific preferred combinations Doses used in combinations Potential
Ixekizumab for treating moderate to severe plaque psoriasis [ID904]
![Ixekizumab for treating moderate to severe plaque psoriasis [ID904] Ixekizumab for treating moderate to severe plaque psoriasis [ID904]](/thumbs/86/94899473.jpg) Thank you for agreeing to make a submission on your organisation s view of the technology and the way it should be used in the NHS. Healthcare professionals can provide a unique perspective on the technology
Thank you for agreeing to make a submission on your organisation s view of the technology and the way it should be used in the NHS. Healthcare professionals can provide a unique perspective on the technology
Analysis of Cardiovascular Risk Factors and Metabolic Syndrome in Korean Patients with Psoriasis
 Ann Dermatol Vol. 24, No. 1, 2012 http://dx.doi.org/10.5021/ad.2012.24.1.11 ORIGINAL ARTICLE Analysis of Cardiovascular Risk Factors and Metabolic Syndrome in Korean Patients with Psoriasis Gun-Wook Kim,
Ann Dermatol Vol. 24, No. 1, 2012 http://dx.doi.org/10.5021/ad.2012.24.1.11 ORIGINAL ARTICLE Analysis of Cardiovascular Risk Factors and Metabolic Syndrome in Korean Patients with Psoriasis Gun-Wook Kim,
Cyltezo (adalimumab-adbm) CG-DRUG-64, CG-DRUG-65
 Market DC Cyltezo (adalimumab-adbm) CG-DRUG-64, CG-DRUG-65 Override(s) Prior Authorization Quantity Limit Medications Cyltezo (adalimumab-adbm) 40 mg/0.8 ml prefilled syringe #* ^ Approval Duration 1 year
Market DC Cyltezo (adalimumab-adbm) CG-DRUG-64, CG-DRUG-65 Override(s) Prior Authorization Quantity Limit Medications Cyltezo (adalimumab-adbm) 40 mg/0.8 ml prefilled syringe #* ^ Approval Duration 1 year
Is Apremilast (Otezla) Effective in Reducing Pruritus in Adults over 18 Years Old with Plaque Psoriasis?
 Philadelphia College of Osteopathic Medicine DigitalCommons@PCOM PCOM Physician Assistant Studies Student Scholarship Student Dissertations, Theses and Papers 3-2017 Is Apremilast (Otezla) Effective in
Philadelphia College of Osteopathic Medicine DigitalCommons@PCOM PCOM Physician Assistant Studies Student Scholarship Student Dissertations, Theses and Papers 3-2017 Is Apremilast (Otezla) Effective in
This is a repository copy of Psoriasis flare with corticosteroid use in psoriatic arthritis.
 This is a repository copy of Psoriasis flare with corticosteroid use in psoriatic arthritis. White Rose Research Online URL for this paper: http://eprints.whiterose.ac.uk/98736/ Version: Accepted Version
This is a repository copy of Psoriasis flare with corticosteroid use in psoriatic arthritis. White Rose Research Online URL for this paper: http://eprints.whiterose.ac.uk/98736/ Version: Accepted Version
UnitedHealthcare Pharmacy Clinical Pharmacy Programs
 UnitedHealthcare Pharmacy Clinical Pharmacy Programs Program Number 2017 P 1041-8 Program Prior Authorization/Notification Medication Humira (adalimumab) P&T Approval Date 1/2007, 6/2008, 4/2009, 6/2009,
UnitedHealthcare Pharmacy Clinical Pharmacy Programs Program Number 2017 P 1041-8 Program Prior Authorization/Notification Medication Humira (adalimumab) P&T Approval Date 1/2007, 6/2008, 4/2009, 6/2009,
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE. Proposed Single Technology Appraisal
 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Proposed Single Technology Appraisal Apremilast for treating moderate to severe plaque psoriasis ID679 Consultee and commentator comment form Please use
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Proposed Single Technology Appraisal Apremilast for treating moderate to severe plaque psoriasis ID679 Consultee and commentator comment form Please use
3. Has the patient shown improvement in signs and symptoms of the disease? Y N
 Pharmacy Prior Authorization MERC CARE (MEDICAID) Renflexis (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information, sign and date. Fax
Pharmacy Prior Authorization MERC CARE (MEDICAID) Renflexis (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information, sign and date. Fax
Comparison of the efficacy of PUVA versus BBUVB in the treatment of psoriasis vulgaris
 IJMAMR 5 (2017) 1-6 ISSN 2053-1834 Comparison of the efficacy of PUVA versus BBUVB in the treatment of psoriasis vulgaris Tran Hau Khang* and Le Huu Doanh National Hospital of Dermatology and Venereology,
IJMAMR 5 (2017) 1-6 ISSN 2053-1834 Comparison of the efficacy of PUVA versus BBUVB in the treatment of psoriasis vulgaris Tran Hau Khang* and Le Huu Doanh National Hospital of Dermatology and Venereology,
Clinical Policy: Etanercept (Enbrel) Reference Number: PA.CP.PHAR.250 Effective Date: 01/18 Last Review Date: 08/17 Line of Business: Medicaid
 Clinical Policy: (Enbrel) Reference Number: PA.CP.PHAR.250 Effective Date: 01/18 Last Review Date: 08/17 Line of Business: Medicaid Coding Implications Revision Log Description (Enbrel ) is tumor necrosis
Clinical Policy: (Enbrel) Reference Number: PA.CP.PHAR.250 Effective Date: 01/18 Last Review Date: 08/17 Line of Business: Medicaid Coding Implications Revision Log Description (Enbrel ) is tumor necrosis
A distinct entity with a close relationship to psoriasis
 Dermatology Clinics & Research DCR, 1(1):1-7 www.scitcentral.com Original Review Article: Open Access Palmoplantar pustulosis: A distinct entity with a close relationship to psoriasis Toshiyuki Yamamoto*
Dermatology Clinics & Research DCR, 1(1):1-7 www.scitcentral.com Original Review Article: Open Access Palmoplantar pustulosis: A distinct entity with a close relationship to psoriasis Toshiyuki Yamamoto*
Phototherapy and Photochemotherapy Treatment (Ultraviolet A [PUVA] and B [UBV])
![Phototherapy and Photochemotherapy Treatment (Ultraviolet A [PUVA] and B [UBV]) Phototherapy and Photochemotherapy Treatment (Ultraviolet A [PUVA] and B [UBV])](/thumbs/89/99382632.jpg) Origination: 09/27/07 Revised: 08/2/17 Annual Review: 11/2/17 Purpose: To provide Phototherapy and Photochemotherapy Treatment (PUVA and UBV) guidelines for the Medical Department staff to reference when
Origination: 09/27/07 Revised: 08/2/17 Annual Review: 11/2/17 Purpose: To provide Phototherapy and Photochemotherapy Treatment (PUVA and UBV) guidelines for the Medical Department staff to reference when
3. Does the patient have a diagnosis of rheumatoid arthritis (RA) with moderate to high disease activity?
 Pharmacy Prior Authorization MERC CARE PLA (MEDICAID) Enbrel (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information, sign and date. Fax
Pharmacy Prior Authorization MERC CARE PLA (MEDICAID) Enbrel (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information, sign and date. Fax
Remicade (Infliximab)
 Remicade (Infliximab) Policy Number: Original Effective Date: MM.04.016 11/18/2003 Line(s) of Business: Current Effective Date: HMO; PPO; QUEST Integration 07/26/2013 Section: Prescription Drugs Place(s)
Remicade (Infliximab) Policy Number: Original Effective Date: MM.04.016 11/18/2003 Line(s) of Business: Current Effective Date: HMO; PPO; QUEST Integration 07/26/2013 Section: Prescription Drugs Place(s)
