Golimumab: a novel anti-tumor necrosis factor
|
|
|
- Stephanie Barrett
- 6 years ago
- Views:
Transcription
1 Golimumab: a novel anti-tumor necrosis factor Rossini M, De Vita S, Ferri C, et al. Biol Ther This slide deck represents the opinions of the authors, and not necessarily the opinions of the publisher or editorial board. This material has been peer reviewed. Medical writing assistance for this study and summary slide was provided by Dr. Pietro Zucchi, freelance medical writer and Dr. Itala Brancaleone freelance translator. Support for this assistance was funded by MSD Italia S.r.l. For a full list of acknowledgments and conflicts of interest for all authors of this article, please see the full text online.
2 ABBREVIATIONS ACR, American College of Rheumatology; ASAS, ASsessment in AS International Working Group criteria; ASspiMRI-a, AS spine MRI-activity; AUC, area under the curve; BASDAI, Bath Ankylosing Spondylitis Disease Activity Index; BASFI, Bath Ankylosing Spondylitis Functional Index; BASMI, Bath Ankylosing Spondylitis Metrology Index; CDAI, Clinical Disease Activity Index; CRP, C-reactive protein; DAS28, Disease Activity Score 28; ESR, erythrocyte sedimentation rate; FACIT-F, Functional Assessment of Chronic Illness Therapy Fatigue; GM, golimumab; HAQ-DI, Health Assessment Questionnaire Disability Index; ITT, intention to treat; IV, intravenous; JSEQ, Jenkins Sleep Evaluation Questionnaire; MASES, Maastricht Ankylosing Spondylitis Enthesitis Score; msasss, modified Stoke AS Spine Score; MTX, methotrexate; NAPSI, Nail Psoriasis Severity Index; PASI, Psoriasis Area and Severity Index; RF, Rheumatoid factor; SF- 36, Short-Form 36; SHS, Sharp/van der Heijde score; SC, subcutaneous; TNF, tumor necrosis factor.
3 INTRODUCTION Golimumab is a human monoclonal antibody preventing the binding of TNF to its receptors through the binding to both soluble and transmembrane human TNF forms. The manufacturing procedure with a peculiar recombinant DNA technology gives golimumab a relatively low immunogenicity and a long in vivo half-life. Golimumab is effective in modulating selective markers of inflammation and bone metabolism. Golimumab is the first anti-tnf agent with once-monthly SC administration that has been approved by the U.S. Food and Drug Administration and the European Medicines Agency for the treatment of rheumatoid arthritis, psoriatic arthritis, and ankylosing spondylitis.
4 CLINICAL EFFICACY IN MODERATE/SEVERE PSORIATIC ARTHRITIS Title and study design Main results GO-REVEAL [1 4] Multicenter randomized placebocontrolled D-B phase III trial in 405 patients with a history of moderate/severe active PsA 6 months Treatments: GM 50 mg/month (G1) GM 100 mg/month (G2) placebo (G3) ACR20 at W14: G1 51%, G2 45%, G3 9% (P <0.001) ACR20 at W24: G1 52%, G2 61%, G3 12% (P <0.001) ACR50 and ACR70 at W14: G1 30% and 12% vs. G3 2% and 1%; P <0.001) ACR50 and ACR70 at W24: G1 32% and 19% vs. G3 4% and 1%; P <0.001 PASI75 at W14: G1 40%, G2 58%, G3 3% (P <0.001) PASI75 at W24: G1 56% vs. G3 1% (P <0.001) NAPSI and PsA-modified MASES at W14 and W24: significantly greater for GM vs. placebo (P <0.001) HAQ-DI at W24 and physical component SF-36 at W14: significantly greater for GM vs. placebo (P <0.001) Follow-up at W52 (all patients switched to GM) PsA-modified SHS at W24: G (P = vs. G3), G2-0.02, G W52 maintenance of radiographic benefits in patients treated with GM W52: ACR20 G1 67%, G2 71%, G3 66%; ACR50 G1 49%, G2 51%, G3 39%; ACR70 G1 36%, G2 30%, G3 20%; DAS28 (CRP) G1 82%, G2 83%, G3 81%; PASI75 G1 62%, G2 69%, G3 48%; PsA-modified MASES G1+G2 54% Follow-up at W104 (all patients receiving GM) GM: ACR20 63% 70%, DAS28-CRP 77% 86%, PASI75 56% 72%; PsA-modified MASES: 40% 60%, HAQ: 53% 59%; mean change in PsA-modified SHS Correlation with patient-reported outcome / clinical outcome After 24 weeks, GM superior to placebo for HAQD I, SF-36 and productivity (P <0.001). Situation maintained to W52 and W Kavanaugh A et al. Arthritis Rheum 2009;60: ; 2. Kavanaugh A, Mease P. J Rheumatol Suppl. 2012;89:90 3; 3. Kavanaugh A et al. Ann Rheum Dis Nov 17. [Epub ahead of print]; 4. Kavanaugh A et al. Arthritis Care Res (Hoboken) May 10:NA. doi: /acr [Epub ahead of print].
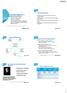
5 Proportions of patients achieving a PASI 75 response ( 75% improvement of the Psoriasis Area and Severity Index) through W24 of the GO-REVEAL study [1] 1. Kavanaugh A et al. Arthritis Rheum 2009;60:
6 CLINICAL EFFICACY IN ACTIVE ANKYLOSING SPONDYLITIS Title and study design GO-RAISE [1 6] Multicenter randomized placebocontrolled D-B phase III study in 356 adults patients with active AS despite current or previous therapy with DMARDs or NSAIDs for at least 3 months Treatments: GM 50 mg/month (G1) GM 100 mg/month (G2) placebo (G3) Main results ASAS20 at W14: G1 59.4%, G2 60%, G3 21.8% (P <0.001) ASAS40 at W24: G1 43.5%, G2 54.3%, G3 15.4% (P <0.001) Lower mean BASDAI and BASFI scores in groups receiving GM vs. placebo BASMI at W14: significantly more patients receiving GM 50 mg and 100 mg showed improvement 1 unit Improvement in physical component SF-36. W14: G1+G2 7.8 vs. G3 2.4, P <0.05; W24: G1+G2 8.1 vs. G3 2.0, P <0.05) Improvement in mental component SF-36. W14: G1+G2 2.5 vs. G3 0.1, P <0.05; W24: G1+G2 2.9 vs. G3-0.3, P <0.05) Improvement in JSEQ. W14: G1+G2-3.0 vs. G3 0.0, P <0.001; W24: G1+G2-3.0 vs. G3-1.0, P <0.001 Follow-up at W104 (all patients receiving GM) ASAS20: G1 60.1%, G2 71.4%, G3 38.5% ASAS40: G1 55.8%, G2 54.3%, G3 38.5% ASAS partial remission: G1 31.9%, G2 30.7%, G3 21.8% Improvement in ASspiMRI-a at W14 with GM maintained through W104, correlated with ASDAS and CRP Follow-up at W208 (all patients receiving GM) msasss: 2.1 placebo GM, 1.3 GM 50 mg; 2.0 GM 100 mg Follow-up at W252 (254 patients) Reduction in signs/symptoms; improvement in physical function/range of motion maintained up to 5 years 1. Inman RD et al. Arthritis Rheum 2008;58: ; 2. Deodhar A et al. Arthritis Care Res (Hoboken) 2010;62: ; 3. Deodhar A et al. EULAR 2013, Madrid June ABS THU0352; 4. Braun J et al. Ann Rheum Dis 2012;71:661 7; 5. Braun J et al. Ann Rheum Dis 2012;71:878 84; 6. Braun J et al. Ann Rheum Dis May 3. [Epub ahead of print].
7 CLINICAL EFFICACY IN MODERATE/SEVERE RHEUMATOID ARTHRITIS (1) Title and study design GO-AFTER [1 3] Multicenter randomized placebocontrolled D-B trial in 461 patients previously treated 1 doses of a anti-tnf without SAEs Treatments: GM 50 mg/month (G1) GM 100 mg/month (G2) placebo (G3) Main results ACR20 at W14: G1 35% vs.g3 18% (P = ); at W24: G1 34% vs.g3 17% (P = ) ACR20 difference greater if associated with a DMARD ACR50 and ACR70 at W14 and W24: significantly more patients in groups treated with GM vs. placebo DAS28 response (EULAR) and DAS28 (ESR) at W14 and W24: significantly more patients in groups treated with GM vs. placebo HAQ-DI reduction at W24: G1 50% (P = vs. G3), G2 54% (P = vs.g3), G3 34% FACIT-F improvement at weeks 14 and 24: significantly greater in GM vs. placebo Follow-up: W160 (all patients receiving GM) - ACR20: G1 67%, G2 56%, G3 63; HAQ-DI: G1 65%, G2 64%, G3 59% W252 (183 patients) - ACR20: 60.3%; ACR50: 42.3%; ACR70: 21.7%; DAS28-CRP EULAR: 84.3%; DAS28-CRP <.6: 29.0%; CDAI 2.8: 16.0% GO-MORE [4, 5] Multicenter open-label study in 3280 patients naive to biologic therapy treated with GM 50 mg/month for 6 months. In patients not in remission GM SC vs.gm IV+SC After 6 months: 82.1% of 3280 patients with good/moderate DAS28 (ESR) response, 23.9% disease remission; 37.4% HAQ DI 0.5. Disease remission: 27.8% if duration <2 years vs.21% if >10 years After 12 months: similar DAS28-ESR remission rates. Mean normalized AUC DAS28 ESR (month 6 12): 3.67 with SC and IV GM 3.67 with SC GM EULAR responses: 85% with GM+MTX, 81% with GM+leflunomide Positive feedback on GM autoinjector use 1. Smolen JS et al. Lancet 2009;374:210 21; 2. Smolen JS et al. Ann Rheum Dis 2012;71:1671 9; 3. Smolen JS et al. EULAR 2013, Madrid June ABS THU0202; 4. Combe B et al. Ann Rheum Dis 2013 Jun 5. [Epub ahead of print]; 5. Wollenhaupt J et al. EULAR 2013, Madrid June ABS AB0273.
8 CLINICAL EFFICACY IN MODERATE/SEVERE RHEUMATOID ARTHRITIS (2) Title and study design Main results GO-BEFORE [1, 2] Randomized placebo-controlled D-B trial in 637 patients naïve to MTX Treatments Placebo + MTX (G1); GM 100 mg + placebo (G2); GM 50 mg + MTX (G3); GM 100 mg + MTX (G4) W24 (ITT): ACR50 G3+G4 38.4% vs. 29.4% G1 (P = 0.053) Post-hoc modified ITT: ACR50 G3+G4 38.5% vs. G1 29.4%; (P = 0.049) G3 40.5% vs. G1 29.4% (P = 0.038); G4 36.5% vs. G1 29.4% (P = 0.177) Follow-up at W 256 (402 patients) 84.3% of patients with ACR20, 93.9% with EULAR DAS28-CRP, 80.6% with improvement in HAQ-DI 0.25 GO-FORWARD [3 6] Multicenter randomized placebocontrolled D-B trial in 444 patients treated with MTX Treatments: Placebo + MTX (G1) GM 100 mg/month + placebo (G2) GM 50 mg/month + MTX (G3) GM 100 mg/month + MTX (G4) At W52 all patients switched to GM ACR20 at W14: G3 55.1%, G4 56.3, G1 33.1% (P = 0.001) HAQ-DI at W24: G3 + G4 vs G1 or G2 (P <0.001) ACR50 and ACR70 at W14; ACR20, ACR50, and ACR70 at W24: significantly greater for GM+MTX vs. placebo+mtx (P <0.05 for all comparisons) DAS28 at W14: G3+G4 73%, G1 44.4% (P <0.001) DAS28 at W24: G3+G4 74.2%, G1 42.1% (P <0.001) Mean improvement in HAQ-DI, physical component of SF-36, and FACIT scores at W14 and W24: significantly greater in G3 and G4 vs. G1 MRI assessment (G3+G4 vs. G1) Wrist and metacarpo-phalangeal synovitis: W vs , P <0.001; W vs , P <0.001 Osteitis: W vs. 0.19, P = 0.003; W vs. 0.71, P = Follow-up: W52 (G1, G2, G3, G4) - ACR20: 44%, 45%, 64%, 58%; DAS28 3.2: 34%, 31%, 42%, 53% W104 - ACR20: GM 50 mg 74.7%; GM 100 mg 71.6%; HAQ-DI 0.25: 88% with GM W252 (313 patients) - ACR20: 76.0%; DAS28-CRP EULAR: 89.5%; HAQ-DI 0.25: 68.5% 1. Smolen JS et al. Lancet 2009;374:210 21; 2. Smolen JS et al. Ann Rheum Dis 2012;71:1671 9; 3. Smolen JS et al. EULAR 2013, Madrid June ABS THU0202; 4. Combe B et al. Ann Rheum Dis 2013 Jun 5. [Epub ahead of print]; 5. Wollenhaupt J et al. EULAR 2013, Madrid June ABS AB0273.
9 Percentage of patients with moderate/severe rheumatoid arthritis, naive to biologic therapy who achieved low disease activity or remission with GM 50 mg/month for 6 months in the GO-MORE study [1]. DAS28, 28-joint disease;activity score; ESR, erythrocyte sedimentation rate. 1. Combe B et al. Ann Rheum Dis 2013 Jun 5. [Epub ahead of print].
10 DISCUSSION Golimumab, used as a monotherapy or in combination with methotrexate*, is indicated for the treatment of severe/moderate active forms of rheumatoid arthritis, psoriatic arthritis and ankylosing spondylitis not responding adequately to conventional therapy. Large studies demonstrated that the drug is effective in improving signs and symptoms of these diseases and patients s physical function, even in the long term. Golimumab has proved to have a safety profile consistent with that of other anti-tnf agents. *in accordance with the therapeutic indications reported in Simponi' (golimumab) Summary of Product Characteristics.
11 CONCLUSION The use of golimumab proved to be: Cost-effective, Simple: no loading dose is required; its two delivery systems are easy to use even by patients who may have difficulty handling normal syringes given the impact of the disease on the hands, Convenient for the patient: the once-monthly SC injection could help to minimize the impact of treatment delivery on the patient s quality of life.
12 Copyright Copyright The Authors This slide deck is distributed under the terms of the Creative Commons Attribution Noncommercial License which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and the source are credited.
PsA. SIMPONI (golimumab) Rheumatoid arthritis. Psoriatic arthritis. Ankylosing spondylitis EFFICACY EFFICACY EFFICACY. QoL. QoL.
 RA Rheumatoid arthritis PsA Psoriatic arthritis AS Ankylosing spondylitis EFFICACY EFFICACY EFFICACY QoL QoL QoL SAFETY SAFETY SAFETY EXPERIENCE EXPERIENCE EXPERIENCE SUMMARY SUMMARY SUMMARY Copyright
RA Rheumatoid arthritis PsA Psoriatic arthritis AS Ankylosing spondylitis EFFICACY EFFICACY EFFICACY QoL QoL QoL SAFETY SAFETY SAFETY EXPERIENCE EXPERIENCE EXPERIENCE SUMMARY SUMMARY SUMMARY Copyright
NEW ZEALAND DATA SHEET
 NEW ZEALAND DATA SHEET SIMPONI Solution for Injection in a pre-filled syringe Solution for Injection in a pre-filled pen, SmartJect NAME OF MEDICINE SIMPONI Solution for Injection in a pre-filled syringe
NEW ZEALAND DATA SHEET SIMPONI Solution for Injection in a pre-filled syringe Solution for Injection in a pre-filled pen, SmartJect NAME OF MEDICINE SIMPONI Solution for Injection in a pre-filled syringe
Horizon Scanning Centre January Apremilast for psoriatic arthritis SUMMARY NIHR HSC ID: 3716
 Horizon Scanning Centre January 2013 Apremilast for psoriatic arthritis SUMMARY NIHR HSC ID: 3716 This briefing is based on information available at the time of research and a limited literature search.
Horizon Scanning Centre January 2013 Apremilast for psoriatic arthritis SUMMARY NIHR HSC ID: 3716 This briefing is based on information available at the time of research and a limited literature search.
Efficacy and Safety of Tocilizumab in the Treatment of Rheumatoid Arthritis and Juvenile Idiopathic Arthritis
 New Evidence reports on presentations given at EULAR 2010 Efficacy and Safety of Tocilizumab in the Treatment of Rheumatoid Arthritis and Juvenile Idiopathic Arthritis Report on EULAR 2010 presentations
New Evidence reports on presentations given at EULAR 2010 Efficacy and Safety of Tocilizumab in the Treatment of Rheumatoid Arthritis and Juvenile Idiopathic Arthritis Report on EULAR 2010 presentations
PRODUCT INFORMATION HUMIRA
 NAME OF THE MEDICINE Adalimumab (rch) DESCRIPTION PRODUCT INFORMATION HUMIRA (adalimumab) is a recombinant human immunoglobulin (IgG1) monoclonal antibody containing only human peptide sequences. was created
NAME OF THE MEDICINE Adalimumab (rch) DESCRIPTION PRODUCT INFORMATION HUMIRA (adalimumab) is a recombinant human immunoglobulin (IgG1) monoclonal antibody containing only human peptide sequences. was created
APC/DTC Briefing Document
 Page 1 London New Drugs Group APC/DTC Briefing Document GOLIMUMAB Contents Summary 1 Background 5 Guidelines 5 Dosing information 5 Drug interactions 6 Clinical studies 6 Ankylosing spondylitis 6 Psoriatic
Page 1 London New Drugs Group APC/DTC Briefing Document GOLIMUMAB Contents Summary 1 Background 5 Guidelines 5 Dosing information 5 Drug interactions 6 Clinical studies 6 Ankylosing spondylitis 6 Psoriatic
SIMPONI Solution for Injection in a pre-filled syringe Solution for Injection in a pre-filled pen, SmartJect
 SIMPONI Solution for Injection in a pre-filled syringe Solution for Injection in a pre-filled pen, SmartJect PRODUCT INFORMATION NAME OF THE MEDICINE Golimumab (rmc) CAS Registry Number: 476181-74-5 DESCRIPTION
SIMPONI Solution for Injection in a pre-filled syringe Solution for Injection in a pre-filled pen, SmartJect PRODUCT INFORMATION NAME OF THE MEDICINE Golimumab (rmc) CAS Registry Number: 476181-74-5 DESCRIPTION
KEYWORDS: golimumab, human monoclonal antibody, new anti-tnf Shweta Bhagat 1, Bhaskar Dasgupta 2 &
 DRUG EVALUATION Golimumab: a new anti-tnf agent on the horizon for inflammatory arthritis Rheumatoid arthritis (RA), psoriatic arthritis, and ankylosing spondylitis are highly prevalent inflammatory arthritides.
DRUG EVALUATION Golimumab: a new anti-tnf agent on the horizon for inflammatory arthritis Rheumatoid arthritis (RA), psoriatic arthritis, and ankylosing spondylitis are highly prevalent inflammatory arthritides.
Horizon Scanning Centre November Secukinumab for active and progressive psoriatic arthritis. SUMMARY NIHR HSC ID: 5330
 Horizon Scanning Centre November 2012 Secukinumab for active and progressive psoriatic arthritis. SUMMARY NIHR HSC ID: 5330 Secukinumab is a high-affinity fully human monoclonal antibody that antagonises
Horizon Scanning Centre November 2012 Secukinumab for active and progressive psoriatic arthritis. SUMMARY NIHR HSC ID: 5330 Secukinumab is a high-affinity fully human monoclonal antibody that antagonises
Golimumab, compared to placebo, significantly improved symptoms in adults with active nonradiographic
 golimumab 50mg/0.5mL solution for injection in pre-filled pen or syringe and 100mg/mL solution for injection in pre-filled pen (Simponi ) SMC No. (1124/16) Merck Sharp & Dohme Limited 8 January 2016 The
golimumab 50mg/0.5mL solution for injection in pre-filled pen or syringe and 100mg/mL solution for injection in pre-filled pen (Simponi ) SMC No. (1124/16) Merck Sharp & Dohme Limited 8 January 2016 The
New long-term data on Cimzia (certolizumab pegol) presented at EULAR 2014 shows sustained outcomes in patients with spondyloarthritis
 New long-term data on Cimzia (certolizumab pegol) presented at EULAR 2014 shows sustained outcomes in patients with spondyloarthritis For the attention of European journalists only New 96-week interim
New long-term data on Cimzia (certolizumab pegol) presented at EULAR 2014 shows sustained outcomes in patients with spondyloarthritis For the attention of European journalists only New 96-week interim
Pharmacy Medical Necessity Guidelines:
 Pharmacy Medical Necessity Guidelines: Effective: January 1, 2018 Prior Authorization Required Type of Review Care Management Not Covered Type of Review Clinical Review Pharmacy (RX) or Medical (MED) Benefit
Pharmacy Medical Necessity Guidelines: Effective: January 1, 2018 Prior Authorization Required Type of Review Care Management Not Covered Type of Review Clinical Review Pharmacy (RX) or Medical (MED) Benefit
SYNOPSIS. Issue Date: 17 Jan 2013
 STELARA (ustekinumab) Clinical Study Report CNTO1275PSA3002 24-Week CSR SYNOPSIS Issue Date: 17 Jan 2013 Name of Sponsor/Company Name of Finished Product Name of Active Ingredient(s) Janssen Research &
STELARA (ustekinumab) Clinical Study Report CNTO1275PSA3002 24-Week CSR SYNOPSIS Issue Date: 17 Jan 2013 Name of Sponsor/Company Name of Finished Product Name of Active Ingredient(s) Janssen Research &
SIMPONI ARIA (GOLIMUMAB) INJECTION FOR INTRAVENOUS INFUSION
 UnitedHealthcare Commercial Medical Benefit Drug Policy SIMPONI ARIA (GOLIMUMAB) INJECTION FOR INTRAVENOUS INFUSION Policy Number: PHA031 Effective Date: March 1, 2019 Table of Contents Page COVERAGE RATIONALE...
UnitedHealthcare Commercial Medical Benefit Drug Policy SIMPONI ARIA (GOLIMUMAB) INJECTION FOR INTRAVENOUS INFUSION Policy Number: PHA031 Effective Date: March 1, 2019 Table of Contents Page COVERAGE RATIONALE...
certolizumab pegol (Cimzia )
 Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
Criteria Inclusion criteria Exclusion criteria. despite treatment with csdmards, NSAIDs, and/or previous anti-tnf therapy and/or
 Supplementary Material Table S1 Eligibility criteria (PICOS) for the SLR Criteria Inclusion criteria Exclusion criteria Population Adults (aged 18 years) with active PsA despite treatment with csdmards,
Supplementary Material Table S1 Eligibility criteria (PICOS) for the SLR Criteria Inclusion criteria Exclusion criteria Population Adults (aged 18 years) with active PsA despite treatment with csdmards,
Pharmacy Medical Necessity Guidelines: Simponi and Simponi Aria (golimumab)
 Pharmacy Medical Necessity Guidelines: Simponi and Simponi Aria (golimumab) Effective: November 20, 2017 Prior Authorization Required Type of Review Care Management Not Covered Type of Review Clinical
Pharmacy Medical Necessity Guidelines: Simponi and Simponi Aria (golimumab) Effective: November 20, 2017 Prior Authorization Required Type of Review Care Management Not Covered Type of Review Clinical
Proper treatment of psoriatic arthritis (PsA) requires. Update on Treatment of Psoriatic Arthritis. Philip Mease, M.D.
 167 Update on Treatment of Psoriatic Arthritis Philip Mease, M.D. Abstract Some of this past year s key papers or abstracts on psoriatic arthritis (PsA) assessment and treatment are reviewed in this paper.
167 Update on Treatment of Psoriatic Arthritis Philip Mease, M.D. Abstract Some of this past year s key papers or abstracts on psoriatic arthritis (PsA) assessment and treatment are reviewed in this paper.
SpA non-radiografica: fase precoce di spondilite anchilosante o altro?
 Rheumatology Department of Lucania, S. Carlo Hospital of Potenza and Madonna delle Grazie Hospital of Matera SpA non-radiografica: fase precoce di spondilite anchilosante o altro? Ignazio Olivieri Disclosures
Rheumatology Department of Lucania, S. Carlo Hospital of Potenza and Madonna delle Grazie Hospital of Matera SpA non-radiografica: fase precoce di spondilite anchilosante o altro? Ignazio Olivieri Disclosures
1.0 Abstract. Title P13-562
 1.0 Abstract Title P13-562 Assessment of Rheumatoid Arthritis, Psoriatic Arthritis, Ankylosing Spondylitis, Plaque Psoriasis, Crohn s Disease and Ulcerative Colitis patients`adherence attitudes to maintenance
1.0 Abstract Title P13-562 Assessment of Rheumatoid Arthritis, Psoriatic Arthritis, Ankylosing Spondylitis, Plaque Psoriasis, Crohn s Disease and Ulcerative Colitis patients`adherence attitudes to maintenance
New Evidence reports on presentations given at EULAR Tocilizumab for the Treatment of Rheumatoid Arthritis
 New Evidence reports on presentations given at EULAR 2012 Tocilizumab for the Treatment of Rheumatoid Arthritis Report on EULAR 2012 presentations Tocilizumab monotherapy is superior to adalimumab monotherapy
New Evidence reports on presentations given at EULAR 2012 Tocilizumab for the Treatment of Rheumatoid Arthritis Report on EULAR 2012 presentations Tocilizumab monotherapy is superior to adalimumab monotherapy
Rheumatology journal club October 20, 2017 Presented by: Matthew Stoll MD,PhD,PSCS
 Efficacy and safety of abatacept, a T-cell modulator, in a randomised, double-blind, placebo-controlled, phase III study in psoriatic arthritis (Mease et al., 2017) Rheumatology journal club October 20,
Efficacy and safety of abatacept, a T-cell modulator, in a randomised, double-blind, placebo-controlled, phase III study in psoriatic arthritis (Mease et al., 2017) Rheumatology journal club October 20,
Opinion 1 October 2014
 The legally binding text is the original French version TRANSPARENCY COMMITTEE Opinion 1 October 2014 CIMZIA 200 mg, solution for subcutaneous injection 1 B/2 1 ml prefilled syringes with needle guard
The legally binding text is the original French version TRANSPARENCY COMMITTEE Opinion 1 October 2014 CIMZIA 200 mg, solution for subcutaneous injection 1 B/2 1 ml prefilled syringes with needle guard
Ustekinumab (Stelara) for psoriatic arthritis second line after disease modifying anti rheumatic drugs (DMARDs)
 Ustekinumab (Stelara) for psoriatic arthritis second line after disease modifying anti rheumatic drugs (DMARDs) January 2010 This technology summary is based on information available at the time of research
Ustekinumab (Stelara) for psoriatic arthritis second line after disease modifying anti rheumatic drugs (DMARDs) January 2010 This technology summary is based on information available at the time of research
EULAR UCB, Inc. All rights reserved. For unsolicited request only.
 1 EULAR 213 2 CZP in AxSpA Effects of certolizumab pegol (CZP) on the signs and symptoms of AxSpA at week 24 (RAPID-AxSpA) 3 RAPID-AxSpA: Ongoing 24-week trial in adult patients with active AxSpA according
1 EULAR 213 2 CZP in AxSpA Effects of certolizumab pegol (CZP) on the signs and symptoms of AxSpA at week 24 (RAPID-AxSpA) 3 RAPID-AxSpA: Ongoing 24-week trial in adult patients with active AxSpA according
ESPONDILOARTROPATÍAS. Dr. Julio Ramírez García
 ESPONDILOARTROPATÍAS Dr. Julio Ramírez García Bloque 1: Caracterización de los pacientes con SpA axial ABSTRACT NUMBER: 1509 Similarities and Differences between Non-Radiographic and Radiographic Axial
ESPONDILOARTROPATÍAS Dr. Julio Ramírez García Bloque 1: Caracterización de los pacientes con SpA axial ABSTRACT NUMBER: 1509 Similarities and Differences between Non-Radiographic and Radiographic Axial
Annual Rheumatology & Therapeutics Review for Organizations & Societies
 Annual Rheumatology & Therapeutics Review for Organizations & Societies Comparative Effectiveness Studies of Biologics Learning Objectives Understand the motivation for comparative effectiveness research
Annual Rheumatology & Therapeutics Review for Organizations & Societies Comparative Effectiveness Studies of Biologics Learning Objectives Understand the motivation for comparative effectiveness research
New Evidence reports on presentations given at ACR Improving Radiographic, Clinical, and Patient-Reported Outcomes with Rituximab
 New Evidence reports on presentations given at ACR 2009 Improving Radiographic, Clinical, and Patient-Reported Outcomes with Rituximab From ACR 2009: Rituximab Rituximab in combination with methotrexate
New Evidence reports on presentations given at ACR 2009 Improving Radiographic, Clinical, and Patient-Reported Outcomes with Rituximab From ACR 2009: Rituximab Rituximab in combination with methotrexate
The Cosentyx clinical trial programme 1-11
 The Cosentyx clinical trial programme 1-11 There are eight pivotal trials (four in psoriasis, two in psoriatic arthritis, two in ankylosing spondylitis) There are two head-to-head trials in psoriasis showing
The Cosentyx clinical trial programme 1-11 There are eight pivotal trials (four in psoriasis, two in psoriatic arthritis, two in ankylosing spondylitis) There are two head-to-head trials in psoriasis showing
STELARA DATA SHEET NAME OF THE MEDICINE DESCRIPTION V L C L V H C H 1 C H 2 C H 3. Fab. F(ab)' 2. hinge
 DATA SHEET NAME OF THE MEDICINE Ustekinumab (rmc). CAS Registry Number: 815610-63-0. DESCRIPTION (ustekinumab) is a human IgG1kappa monoclonal antibody with an approximate molecular weight of 148,600 daltons.
DATA SHEET NAME OF THE MEDICINE Ustekinumab (rmc). CAS Registry Number: 815610-63-0. DESCRIPTION (ustekinumab) is a human IgG1kappa monoclonal antibody with an approximate molecular weight of 148,600 daltons.
New Evidence reports on presentations given at EULAR Safety and Efficacy of Tocilizumab as Monotherapy and in Combination with Methotrexate
 New Evidence reports on presentations given at EULAR 2009 Safety and Efficacy of Tocilizumab as Monotherapy and in Combination with Methotrexate Report on EULAR 2009 presentations Tocilizumab inhibits
New Evidence reports on presentations given at EULAR 2009 Safety and Efficacy of Tocilizumab as Monotherapy and in Combination with Methotrexate Report on EULAR 2009 presentations Tocilizumab inhibits
REMICADE POWDER FOR INJECTION
 REMICADE POWDER FOR INJECTION Infliximab PRODUCT INFORMATION NAME OF THE MEDICINE Infliximab Powder for Injection DESCRIPTION Each vial of REMICADE contains infliximab 100 mg. REMICADE Powder for Injection
REMICADE POWDER FOR INJECTION Infliximab PRODUCT INFORMATION NAME OF THE MEDICINE Infliximab Powder for Injection DESCRIPTION Each vial of REMICADE contains infliximab 100 mg. REMICADE Powder for Injection
Synopsis (C0524T12 GO LIVE)
 Protocol: EudraCT No.: 2005-003232-21 Title of the study: A Multicenter, Randomized, Double-blind, Placebo-controlled Trial of Golimumab, a Fully Human Anti-TNFα Monoclonal Antibody, Administered Intravenously,
Protocol: EudraCT No.: 2005-003232-21 Title of the study: A Multicenter, Randomized, Double-blind, Placebo-controlled Trial of Golimumab, a Fully Human Anti-TNFα Monoclonal Antibody, Administered Intravenously,
CLINICAL MONOGRAPH FOR SIMPONI ARIA (golimumab)
 CLINICAL MONOGRAPH FOR SIMPONI ARIA (golimumab) INDICATIONS SIMPONI ARIA is indicated for the treatment of adult patients with: Moderately to severely active rheumatoid arthritis (RA), in combination with
CLINICAL MONOGRAPH FOR SIMPONI ARIA (golimumab) INDICATIONS SIMPONI ARIA is indicated for the treatment of adult patients with: Moderately to severely active rheumatoid arthritis (RA), in combination with
Abatacept (Orencia) for active rheumatoid arthritis. August 2009
 Abatacept (Orencia) for active rheumatoid arthritis August 2009 This technology summary is based on information available at the time of research and a limited literature search. It is not intended to
Abatacept (Orencia) for active rheumatoid arthritis August 2009 This technology summary is based on information available at the time of research and a limited literature search. It is not intended to
Psoriatic Arthritis: New and Emergent Therapies
 Psoriatic Arthritis: New and Emergent Therapies Alice Bendix Gottlieb MD, PhD Professor of Dermatology New York Medical College Metropolitan Hospital New York, NY, USA DISCLOSURE OF RELEVANT RELATIONSHIPS
Psoriatic Arthritis: New and Emergent Therapies Alice Bendix Gottlieb MD, PhD Professor of Dermatology New York Medical College Metropolitan Hospital New York, NY, USA DISCLOSURE OF RELEVANT RELATIONSHIPS
Cimzia (certolizumab pegol)
 DRUG POLICY BENEFIT APPLICATION Cimzia (certolizumab pegol) Benefit determinations are based on the applicable contract language in effect at the time the services were rendered. Exclusions, limitations
DRUG POLICY BENEFIT APPLICATION Cimzia (certolizumab pegol) Benefit determinations are based on the applicable contract language in effect at the time the services were rendered. Exclusions, limitations
golimumab (Simponi Aria, Simponi )
 Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
Cosentyx clinical trial program in spondyloarthritis (SpA) 1-7
 Cosentyx clinical trial program in spondyloarthritis (SpA) 1-7 There are five pivotal trials; three in psoriatic arthritis, two in ankylosing spondylitis More than 10,000 patients have been treated with
Cosentyx clinical trial program in spondyloarthritis (SpA) 1-7 There are five pivotal trials; three in psoriatic arthritis, two in ankylosing spondylitis More than 10,000 patients have been treated with
SIMPONI ARIA (GOLIMUMAB) INJECTION FOR INTRAVENOUS INFUSION
 UnitedHealthcare Oxford Clinical Policy SIMPONI ARIA (GOLIMUMAB) INJECTION FOR INTRAVENOUS INFUSION Policy Number: PHARMACY 292.4 T2 Effective Date: April 1, 2018 Table of Contents Page INSTRUCTIONS FOR
UnitedHealthcare Oxford Clinical Policy SIMPONI ARIA (GOLIMUMAB) INJECTION FOR INTRAVENOUS INFUSION Policy Number: PHARMACY 292.4 T2 Effective Date: April 1, 2018 Table of Contents Page INSTRUCTIONS FOR
NEW EFFECTIVE TREATMENTS FOR PSORIATIC ARTHRITIS PATIENTS Promising data to support two new drug classes
 Annual European Congress of Rheumatology (EULAR) 2017 Madrid, Spain, 14-17 June 2017 NEW EFFECTIVE TREATMENTS FOR PSORIATIC ARTHRITIS PATIENTS Promising data to support two new drug classes Madrid, Spain,
Annual European Congress of Rheumatology (EULAR) 2017 Madrid, Spain, 14-17 June 2017 NEW EFFECTIVE TREATMENTS FOR PSORIATIC ARTHRITIS PATIENTS Promising data to support two new drug classes Madrid, Spain,
Clinical Policy: Etanercept (Enbrel) Reference Number: PA.CP.PHAR.250 Effective Date: 01/18 Last Review Date: 08/17 Line of Business: Medicaid
 Clinical Policy: (Enbrel) Reference Number: PA.CP.PHAR.250 Effective Date: 01/18 Last Review Date: 08/17 Line of Business: Medicaid Coding Implications Revision Log Description (Enbrel ) is tumor necrosis
Clinical Policy: (Enbrel) Reference Number: PA.CP.PHAR.250 Effective Date: 01/18 Last Review Date: 08/17 Line of Business: Medicaid Coding Implications Revision Log Description (Enbrel ) is tumor necrosis
What is Cosentyx (secukinumab)?
 What is Cosentyx (secukinumab)? Cosentyx is the first of a new class of medicines called interleukin- 17A (IL- 17A) inhibitors to be approved for the treatment of moderate- to- severe plaque psoriasis,
What is Cosentyx (secukinumab)? Cosentyx is the first of a new class of medicines called interleukin- 17A (IL- 17A) inhibitors to be approved for the treatment of moderate- to- severe plaque psoriasis,
Axial Spondyloarthritis. Doug White, Rheumatologist Waikato Hospital
 Axial Spondyloarthritis Doug White, Rheumatologist Waikato Hospital Disclosures Presentations / Consulting Abbott Laboratories AbbVie MSD Novartis Roche Clinical Trials Abbott Laboratories AbbVie Actelion
Axial Spondyloarthritis Doug White, Rheumatologist Waikato Hospital Disclosures Presentations / Consulting Abbott Laboratories AbbVie MSD Novartis Roche Clinical Trials Abbott Laboratories AbbVie Actelion
Regulatory Status FDA- approved indication: Simponi and Simponi ARIA are tumor necrosis factor (TNF) blockers indicated for the treatment of:
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.51 Subject: Simponi / Simponi ARIA Page: 1 of 8 Last Review Date: March 17, 2017 Simponi / Simponi
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.51 Subject: Simponi / Simponi ARIA Page: 1 of 8 Last Review Date: March 17, 2017 Simponi / Simponi
Certolizumab pegol (Cimzia) for psoriatic arthritis second line
 Certolizumab pegol (Cimzia) for psoriatic arthritis second line This technology summary is based on information available at the time of research and a limited literature search. It is not intended to
Certolizumab pegol (Cimzia) for psoriatic arthritis second line This technology summary is based on information available at the time of research and a limited literature search. It is not intended to
Cosentyx clinical trial program in spondyloarthritis (SpA) 1-5
 Cosentyx clinical trial program in spondyloarthritis (SpA) 1-5 There are four pivotal trials; two in psoriatic arthritis, two in ankylosing spondylitis More than 10,000 patients have been treated with
Cosentyx clinical trial program in spondyloarthritis (SpA) 1-5 There are four pivotal trials; two in psoriatic arthritis, two in ankylosing spondylitis More than 10,000 patients have been treated with
SIMPONI Solution for Injection in a pre-filled syringe Solution for Injection in a pre-filled pen, SmartJect PRODUCT INFORMATION
 SIMPONI Solution for Injection in a pre-filled syringe Solution for Injection in a pre-filled pen, SmartJect PRODUCT INFORMATION NAME OF THE MEDICINE Golimumab (rmc) CAS Registry Number: 476181-74-5 DESCRIPTION
SIMPONI Solution for Injection in a pre-filled syringe Solution for Injection in a pre-filled pen, SmartJect PRODUCT INFORMATION NAME OF THE MEDICINE Golimumab (rmc) CAS Registry Number: 476181-74-5 DESCRIPTION
Scottish Medicines Consortium
 Scottish Medicines Consortium etanercept 25mg vial of powder for subcutaneous injection (Enbrel ) (No. 212/05) Wyeth New indication: severe active ankylosing spondylitis inadequately controlled by conventional
Scottish Medicines Consortium etanercept 25mg vial of powder for subcutaneous injection (Enbrel ) (No. 212/05) Wyeth New indication: severe active ankylosing spondylitis inadequately controlled by conventional
Principal Investigator. General Information. Conflict of Interest. Certification Published on The YODA Project (
 Principal Investigator First Name: Liana Last Name: Fraenkel Degree: MD, MPH Primary Affiliation: Yale University School of Medicine E-mail: christine.ramsey@gmail.com Phone number: 610-613-6745 Address:
Principal Investigator First Name: Liana Last Name: Fraenkel Degree: MD, MPH Primary Affiliation: Yale University School of Medicine E-mail: christine.ramsey@gmail.com Phone number: 610-613-6745 Address:
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS
 ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 This medicinal product is subject to additional monitoring. This will allow quick identification of new safety information. Healthcare professionals are asked
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 This medicinal product is subject to additional monitoring. This will allow quick identification of new safety information. Healthcare professionals are asked
Guideline on the Clinical Investigation of Medicinal Products for the Treatment of Axial Spondyloarthritis
 12 October 2017 EMA/CPMP/EWP/4891/03 Rev.1 Committee for Medicinal Products for Human Use (CHMP) Guideline on the Clinical Investigation of Medicinal Products for the Treatment of Axial Draft agreed by
12 October 2017 EMA/CPMP/EWP/4891/03 Rev.1 Committee for Medicinal Products for Human Use (CHMP) Guideline on the Clinical Investigation of Medicinal Products for the Treatment of Axial Draft agreed by
Clinical Policy: Certolizumab (Cimzia) Reference Number: PA.CP.PHAR.247 Effective Date: 01/18 Last Review Date: 08/17 Line of Business: Medicaid
 Clinical Policy: (Cimzia) Reference Number: PA.CP.PHAR.247 Effective Date: 01/18 Last Review Date: 08/17 Line of Business: Medicaid Coding Implications Revision Log Description (Cimzia ) is a tumor necrosis
Clinical Policy: (Cimzia) Reference Number: PA.CP.PHAR.247 Effective Date: 01/18 Last Review Date: 08/17 Line of Business: Medicaid Coding Implications Revision Log Description (Cimzia ) is a tumor necrosis
Certolizumab pegol (Cimzia) for the treatment of ankylosing spondylitis second or third line
 Certolizumab pegol (Cimzia) for the treatment of ankylosing spondylitis second or third line August 2011 This technology summary is based on information available at the time of research and a limited
Certolizumab pegol (Cimzia) for the treatment of ankylosing spondylitis second or third line August 2011 This technology summary is based on information available at the time of research and a limited
Synopsis (C0743T10) CNTO 1275 Module 5.3 C0743T10. Associated with Module 5.3 of the Dossier
 Module 5.3 Protocol: EudraCT No.: 2005-003525-92 Title of the study: A Phase 2, Multicenter, Randomized, Double-blind, Placebo-controlled Trial of, a Fully Human Anti-IL-12 Monoclonal Antibody, Administered
Module 5.3 Protocol: EudraCT No.: 2005-003525-92 Title of the study: A Phase 2, Multicenter, Randomized, Double-blind, Placebo-controlled Trial of, a Fully Human Anti-IL-12 Monoclonal Antibody, Administered
10/28/2013. Disclosure. Ustekinumab. IL-12, IL-23 and Ustekinumab. IL-23 in Facet Joints in Patients with AS
 for the Treatment of Patients with Active Ankylosing Spondylitis: Results of a 28-Week, Prospective, Open-Label, Proof-of-Concept Study (TOPAS) Disclosure The study was supported by an unrestricted research
for the Treatment of Patients with Active Ankylosing Spondylitis: Results of a 28-Week, Prospective, Open-Label, Proof-of-Concept Study (TOPAS) Disclosure The study was supported by an unrestricted research
Rheumatoid arthritis 2010: Treatment and monitoring
 October 12, 2010 By Yusuf Yazici, MD [1] The significant changes in the way rheumatoid arthritis has been managed include earlier, more aggressive treatment with combination therapy. Significant changes
October 12, 2010 By Yusuf Yazici, MD [1] The significant changes in the way rheumatoid arthritis has been managed include earlier, more aggressive treatment with combination therapy. Significant changes
adalimumab, 40mg/0.8mL, solution for injection (Humira ) SMC No. (858/13) AbbVie Ltd (previously part of Abbott)
 adalimumab, 40mg/0.8mL, solution for injection (Humira ) SMC No. (858/13) AbbVie Ltd (previously part of Abbott) 08 March 2013 The Scottish Medicines Consortium (SMC) has completed its assessment of the
adalimumab, 40mg/0.8mL, solution for injection (Humira ) SMC No. (858/13) AbbVie Ltd (previously part of Abbott) 08 March 2013 The Scottish Medicines Consortium (SMC) has completed its assessment of the
HARVARD PILGRIM HEALTH CARE RECOMMENDED MEDICATION REQUEST GUIDELINES
 Generic Brand HICL GCN Exception/Other GOLIMUMAB SIMPONI 22533, 22536, 34697, 35001 ROUTE = SUBCUTANE. GUIDELINES FOR USE INITIAL CRITERIA (NOTE: FOR RENEWAL CRITERIA SEE BELOW) 1. Is the request for a
Generic Brand HICL GCN Exception/Other GOLIMUMAB SIMPONI 22533, 22536, 34697, 35001 ROUTE = SUBCUTANE. GUIDELINES FOR USE INITIAL CRITERIA (NOTE: FOR RENEWAL CRITERIA SEE BELOW) 1. Is the request for a
Serum sclerostin as a possible biomarker in ankylosing spondylitis: a casecontrol
 Serum sclerostin as a possible biomarker in ankylosing spondylitis: a casecontrol study Fabio Massimo Perrotta 1, MD, Fulvia Ceccarelli 2, MD, PhD, Cristiana Barbati 2, PhD, Tania Colasanti 2, PhD, Antonia
Serum sclerostin as a possible biomarker in ankylosing spondylitis: a casecontrol study Fabio Massimo Perrotta 1, MD, Fulvia Ceccarelli 2, MD, PhD, Cristiana Barbati 2, PhD, Tania Colasanti 2, PhD, Antonia
Canadian Society of Internal Medicine Annual Meeting 2016 Montreal, QC
 Canadian Society of Internal Medicine Annual Meeting 2016 Montreal, QC Update on the Treatment of Rheumatoid Arthritis Sabrina Fallavollita MDCM McGill University Canadian Society of Internal Medicine
Canadian Society of Internal Medicine Annual Meeting 2016 Montreal, QC Update on the Treatment of Rheumatoid Arthritis Sabrina Fallavollita MDCM McGill University Canadian Society of Internal Medicine
Treat - to - Target Pathway Commissioning Chronic and Complex Care MIDLANDS RHEUMATOLOGY & MUSCULOSKELETAL (MSK) COMMISSIONING NETWORK
 Treat - to - Target Pathway Commissioning Chronic and Complex Care MIDLANDS RHEUMATOLOGY & MUSCULOSKELETAL (MSK) COMMISSIONING NETWORK Dr Bruce Kirkham Consultant Rheumatologist Guy s & St Thomas NHS Foundation
Treat - to - Target Pathway Commissioning Chronic and Complex Care MIDLANDS RHEUMATOLOGY & MUSCULOSKELETAL (MSK) COMMISSIONING NETWORK Dr Bruce Kirkham Consultant Rheumatologist Guy s & St Thomas NHS Foundation
ARTHRITIS ADVISORY COMMITTEE MEETING
 ARTHRITIS ADVISORY COMMITTEE MEETING July 23, 2013 sbla 125057/323: adalimumab for the treatment of Active non-radiographic axial spondyloarthritis in adults with objective signs of inflammation by elevated
ARTHRITIS ADVISORY COMMITTEE MEETING July 23, 2013 sbla 125057/323: adalimumab for the treatment of Active non-radiographic axial spondyloarthritis in adults with objective signs of inflammation by elevated
24-Week CNTO1275PSA3001 Clinical Study Report
 24-Week CNTO1275PSA3001 Clinical Study Report SYNOPSIS Issue Date: 17 Jan 2013 Name of Sponsor/Company Name of Finished Product Name of Active Ingredient(s) Janssen Research & Development, Inc Ustekinumab
24-Week CNTO1275PSA3001 Clinical Study Report SYNOPSIS Issue Date: 17 Jan 2013 Name of Sponsor/Company Name of Finished Product Name of Active Ingredient(s) Janssen Research & Development, Inc Ustekinumab
2016 update of the ASAS/EULAR recommendations for the management of axial spondyloarthritis. Online supplementary material
 2016 update of the ASAS/EULAR recommendations for the management of axial spondyloarthritis Online supplementary material 1. Introduction A systematic literature review (SLR) was performed to inform the
2016 update of the ASAS/EULAR recommendations for the management of axial spondyloarthritis Online supplementary material 1. Introduction A systematic literature review (SLR) was performed to inform the
Supplemental Table 1. Key Inclusion Criteria Inclusion Criterion OPTIMA PREMIER 18 years old with RA (per 1987 revised American College of General
 Supplemental Table 1. Key Inclusion Criteria Inclusion Criterion OPTIMA PREMIER 18 years old with RA (per 1987 revised American College of General Rheumatology classification criteria) 34 ; erythrocyte
Supplemental Table 1. Key Inclusion Criteria Inclusion Criterion OPTIMA PREMIER 18 years old with RA (per 1987 revised American College of General Rheumatology classification criteria) 34 ; erythrocyte
SCIENTIFIC DISCUSSION. London, 27 April 2006 Product name: HUMIRA/TRUDEXA Procedure number: EMEA/H/C/ /II/26
 SCIENTIFIC DISCUSSION London, 27 April 2006 Product name: HUMIRA/TRUDEXA Procedure number: EMEA/H/C/481-482/II/26 3.1. Introduction Adalimumab is a recombinant human immunoglobulin (IgG 1 ) monoclonal
SCIENTIFIC DISCUSSION London, 27 April 2006 Product name: HUMIRA/TRUDEXA Procedure number: EMEA/H/C/481-482/II/26 3.1. Introduction Adalimumab is a recombinant human immunoglobulin (IgG 1 ) monoclonal
TRANSPARENCY COMMITTEE OPINION. 26 April 2006
 TRANSPARENCY COMMITTEE OPINION 26 April 2006 REMICADE 100 mg powder for concentrate for solution for infusion Box of 1 (CIP code: 562 070.1) Applicant : laboratoires Schering Plough List I Drug for hospital
TRANSPARENCY COMMITTEE OPINION 26 April 2006 REMICADE 100 mg powder for concentrate for solution for infusion Box of 1 (CIP code: 562 070.1) Applicant : laboratoires Schering Plough List I Drug for hospital
COMMITTEE FOR MEDICINAL PRODUCTS FOR HUMAN USE (CHMP)
 European Medicines Agency Pre-Authorisation Evaluation of Medicines for Human Use London, 23 April 2009 Doc. Ref. CPMP/EWP/4891/03 COMMITTEE FOR MEDICINAL PRODUCTS FOR HUMAN USE (CHMP) GUIDELINE ON CLINICAL
European Medicines Agency Pre-Authorisation Evaluation of Medicines for Human Use London, 23 April 2009 Doc. Ref. CPMP/EWP/4891/03 COMMITTEE FOR MEDICINAL PRODUCTS FOR HUMAN USE (CHMP) GUIDELINE ON CLINICAL
Drug Therapy Guidelines
 Simponi, Simponi Aria Applicable Medical Benefit x Effective: 2/13/18 Pharmacy- Formulary 1 x Next Review: 12/18 Pharmacy- Formulary 2 x Date of Origin: 7/2010 Pharmacy- Formulary 3/Exclusive x Review
Simponi, Simponi Aria Applicable Medical Benefit x Effective: 2/13/18 Pharmacy- Formulary 1 x Next Review: 12/18 Pharmacy- Formulary 2 x Date of Origin: 7/2010 Pharmacy- Formulary 3/Exclusive x Review
University of California, San Diego, School of Medicine, La Jolla, CA, USA; 2
 2194 Long-term (104-Week) Efficacy and Safety Profile of Apremilast, an Oral Phosphodiesterase 4 Inhibitor, in Patients With Psoriatic Arthritis: Results From a Phase III, Randomized, Controlled Trial
2194 Long-term (104-Week) Efficacy and Safety Profile of Apremilast, an Oral Phosphodiesterase 4 Inhibitor, in Patients With Psoriatic Arthritis: Results From a Phase III, Randomized, Controlled Trial
Journal of Symptoms and Signs 2013; Volume 2, Number 6
 Journal of Symptoms and Signs 2013; Volume 2, Number 6 Expert Opinion Efficacy and safety of golimumab in rheumatoid arthritis, psoriatic arthritis and ankylosing spondylitis Jose R. Maneiro 1, Juan J.
Journal of Symptoms and Signs 2013; Volume 2, Number 6 Expert Opinion Efficacy and safety of golimumab in rheumatoid arthritis, psoriatic arthritis and ankylosing spondylitis Jose R. Maneiro 1, Juan J.
Horizon Scanning Technology Summary. Adalimumab (Humira) for juvenile idiopathic arthritis. National Horizon Scanning Centre.
 Horizon Scanning Technology Summary National Horizon Scanning Centre Adalimumab (Humira) for juvenile idiopathic arthritis June 2007 This technology summary is based on information available at the time
Horizon Scanning Technology Summary National Horizon Scanning Centre Adalimumab (Humira) for juvenile idiopathic arthritis June 2007 This technology summary is based on information available at the time
5/4/2018. Outcome Measures in Spondyloarthritis. Learning Objectives. Outcome Measures Clinical Outcome Assessments
 Outcome Measures in Spondyloarthritis Marina N Magrey MD Associate Professor Case Western Reserve University School of Medicine at MetroHealth Medical Center Learning Objectives What are outcome measures
Outcome Measures in Spondyloarthritis Marina N Magrey MD Associate Professor Case Western Reserve University School of Medicine at MetroHealth Medical Center Learning Objectives What are outcome measures
ABSTRACT. Keywords: Africa; Efficacy; Etanercept; Maintenance therapy; Middle East; Rheumatoid arthritis ORIGINAL RESEARCH
 Rheumatol Ther (2018) 5:149 158 https://doi.org/10.1007/s40744-018-0094-6 ORIGINAL RESEARCH Maintenance of Remission with Etanercept DMARD Combination Therapy Compared with DMARDs Alone in African and
Rheumatol Ther (2018) 5:149 158 https://doi.org/10.1007/s40744-018-0094-6 ORIGINAL RESEARCH Maintenance of Remission with Etanercept DMARD Combination Therapy Compared with DMARDs Alone in African and
Gender differences in effectiveness of treatment in rheumatic diseases
 Gender differences in effectiveness of treatment in rheumatic diseases Irene van der Horst-Bruinsma Associate Professor Rheumatology Center of Excellence of Axial Spondyloarthritis ARC/VU University Medical
Gender differences in effectiveness of treatment in rheumatic diseases Irene van der Horst-Bruinsma Associate Professor Rheumatology Center of Excellence of Axial Spondyloarthritis ARC/VU University Medical
ETANERCEPT Generic Brand HICL GCN Exception/Other ETANERCEPT ENBREL GUIDELINES FOR USE INITIAL CRITERIA (NOTE: FOR RENEWAL CRITERIA SEE BELOW)
 Generic Brand HICL GCN Exception/Other ETANERCEPT ENBREL 18830 GUIDELINES FOR USE INITIAL CRITERIA (NOTE: FOR RENEWAL CRITERIA SEE BELOW) 1. Does the patient have a diagnosis of moderate to severe rheumatoid
Generic Brand HICL GCN Exception/Other ETANERCEPT ENBREL 18830 GUIDELINES FOR USE INITIAL CRITERIA (NOTE: FOR RENEWAL CRITERIA SEE BELOW) 1. Does the patient have a diagnosis of moderate to severe rheumatoid
Strengthening our knowledge in Spondylarthritides: Focusing on Ankylosing Spondylitis and Psoriatic Arthritis
 Department of Pathophysiology Metical School National and Kapodestrian University of Athens Strengthening our knowledge in Spondylarthritides: Focusing on Ankylosing Spondylitis and Psoriatic Arthritis
Department of Pathophysiology Metical School National and Kapodestrian University of Athens Strengthening our knowledge in Spondylarthritides: Focusing on Ankylosing Spondylitis and Psoriatic Arthritis
1.0 Abstract. Title. Keywords. Rationale and Background
 1.0 Abstract Title A Prospective, Multi-Center Study in Rheumatoid Arthritis Patients on Adalimumab to Evaluate its Effect on Synovitis Using Ultrasonography in an Egyptian Population Keywords Synovitis
1.0 Abstract Title A Prospective, Multi-Center Study in Rheumatoid Arthritis Patients on Adalimumab to Evaluate its Effect on Synovitis Using Ultrasonography in an Egyptian Population Keywords Synovitis
2.0 Synopsis. Adalimumab DE019 OLE (5-year) Clinical Study Report Amendment 1 R&D/06/095. (For National Authority Use Only)
 2.0 Synopsis Abbott Laboratories Name of Study Drug: Humira Name of Active Ingredient: Adalimumab Individual Study Table Referring to Part of Dossier: Volume: Page: (For National Authority Use Only) Title
2.0 Synopsis Abbott Laboratories Name of Study Drug: Humira Name of Active Ingredient: Adalimumab Individual Study Table Referring to Part of Dossier: Volume: Page: (For National Authority Use Only) Title
Imaging of axial spondyloarthritis including ankylosing spondylitis
 Imaging of axial spondyloarthritis including ankylosing spondylitis ACR 2012 Prof. Dr. med. J. Braun Rheumazentrum Ruhrgebiet, Herne Ruhr-Universität Bochum Germany Modified New York Criteria 1984 for
Imaging of axial spondyloarthritis including ankylosing spondylitis ACR 2012 Prof. Dr. med. J. Braun Rheumazentrum Ruhrgebiet, Herne Ruhr-Universität Bochum Germany Modified New York Criteria 1984 for
PRODUCT INFORMATION. RENFLEXIS Infliximab NAME OF THE MEDICINE DESCRIPTION PHARMACOLOGY
 PRODUCT INFORMATION Infliximab NAME OF THE MEDICINE (Infliximab) Powder for Injection CAS number: 170277-31-3 DESCRIPTION RENFLEXIS (infliximab) is a biosimilar medicine to Remicade (infliximab). The comparability
PRODUCT INFORMATION Infliximab NAME OF THE MEDICINE (Infliximab) Powder for Injection CAS number: 170277-31-3 DESCRIPTION RENFLEXIS (infliximab) is a biosimilar medicine to Remicade (infliximab). The comparability
The prevalence and clinical effect of immunogenicity of TNF-α blockers in patients with axial spondyloarthritis
 The prevalence and clinical effect of immunogenicity of TNF-α blockers in patients with axial spondyloarthritis G. Bornstein 1,2, M. Lidar 3, P. Langevitz 3, A. Fardman 2, I. Ben-Zvi 4, C. Grossman 4 1
The prevalence and clinical effect of immunogenicity of TNF-α blockers in patients with axial spondyloarthritis G. Bornstein 1,2, M. Lidar 3, P. Langevitz 3, A. Fardman 2, I. Ben-Zvi 4, C. Grossman 4 1
ARTHRITIS ADVISORY COMMITTEE MEETING
 ARTHRITIS ADVISORY COMMITTEE MEETING July 23, 2013 sbla 125160/215: Cimzia (certolizumab) for the treatment of active axial spondyloarthritis, including patients with ankylosing spondylitis Disclaimer
ARTHRITIS ADVISORY COMMITTEE MEETING July 23, 2013 sbla 125160/215: Cimzia (certolizumab) for the treatment of active axial spondyloarthritis, including patients with ankylosing spondylitis Disclaimer
London, 1 June 2006 Product name: REMICADE Procedure number: Remicade-H-240-II-73-AR SCIENTIFIC DISCUSSION 1/8
 London, 1 June 2006 Product name: REMICADE Procedure number: Remicade-H-240-II-73-AR SCIENTIFIC DISCUSSION 1/8 1. Introduction Infliximab is a chimeric human-murine IgG1κ monoclonal antibody, which binds
London, 1 June 2006 Product name: REMICADE Procedure number: Remicade-H-240-II-73-AR SCIENTIFIC DISCUSSION 1/8 1. Introduction Infliximab is a chimeric human-murine IgG1κ monoclonal antibody, which binds
Cigna Drug and Biologic Coverage Policy
 Cigna Drug and Biologic Coverage Policy Subject Apremilast Table of Contents Coverage Policy... 1 General Background... 2 Coding/Billing Information... 4 References... 4 Effective Date... 1/1/2018 Next
Cigna Drug and Biologic Coverage Policy Subject Apremilast Table of Contents Coverage Policy... 1 General Background... 2 Coding/Billing Information... 4 References... 4 Effective Date... 1/1/2018 Next
ADALIMUMAB Generic Brand HICL GCN Exception/Other ADALIMUMAB HUMIRA GUIDELINES FOR USE INITIAL CRITERIA (NOTE: FOR RENEWAL CRITERIA SEE BELOW)
 Generic Brand HICL GCN Exception/Other ADALIMUMAB HUMIRA 24800 GUIDELINES FOR USE INITIAL CRITERIA (NOTE: FOR RENEWAL CRITERIA SEE BELOW) 1. Does the patient have a diagnosis of moderate to severe rheumatoid
Generic Brand HICL GCN Exception/Other ADALIMUMAB HUMIRA 24800 GUIDELINES FOR USE INITIAL CRITERIA (NOTE: FOR RENEWAL CRITERIA SEE BELOW) 1. Does the patient have a diagnosis of moderate to severe rheumatoid
LOCALLY AVAILABLE BIOLOGIC AGENTS IN THE TREATMENT OF PSORIATIC ARTHRITIS
 Locally Available Biologic Agents in the Treatment of Psoriatic Arthritis 253 Phil. J. Internal Medicine, 47: 253-259, Nov.-Dec., 2009 LOCALLY AVAILABLE BIOLOGIC AGENTS IN THE TREATMENT OF PSORIATIC ARTHRITIS
Locally Available Biologic Agents in the Treatment of Psoriatic Arthritis 253 Phil. J. Internal Medicine, 47: 253-259, Nov.-Dec., 2009 LOCALLY AVAILABLE BIOLOGIC AGENTS IN THE TREATMENT OF PSORIATIC ARTHRITIS
APPLICATION FOR SUBSIDY BY SPECIAL AUTHORITY
 APPLICANT (stamp sticker acceptable) Page 1 Fm SA1621 Adalimumab INITIAL APPLICATION - rheumatoid arthritis Applications only from a rheumatologist. Approvals valid f 6 months. The patient has had an initial
APPLICANT (stamp sticker acceptable) Page 1 Fm SA1621 Adalimumab INITIAL APPLICATION - rheumatoid arthritis Applications only from a rheumatologist. Approvals valid f 6 months. The patient has had an initial
Regulatory Status FDA- approved indication: Simponi and Simponi ARIA are tumor necrosis factor (TNF) blockers indicated for the treatment of: (2-3)
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.51 Subject: Simponi / Simponi ARIA Page: 1 of 9 Last Review Date: March 16, 2018 Simponi / Simponi
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.51 Subject: Simponi / Simponi ARIA Page: 1 of 9 Last Review Date: March 16, 2018 Simponi / Simponi
Cover Page. The handle holds various files of this Leiden University dissertation
 Cover Page The handle http://hdl.handle.net/1887/43590 holds various files of this Leiden University dissertation Author: Machado, Pedro Title: Health and imaging outcomes in axial spondyloarthritis Issue
Cover Page The handle http://hdl.handle.net/1887/43590 holds various files of this Leiden University dissertation Author: Machado, Pedro Title: Health and imaging outcomes in axial spondyloarthritis Issue
2010 Annual Meeting of the Canadian Rheumatology Association February 3 to 6, Quebec City, Quebec. Copyright. Not for Sale or Commercial Distribution
 21 Annual Meeting of the Canadian Rheumatology Association February 3 to 6, Quebec City, Quebec Copyright In February 21, Quebec City hosted the annual meeting of the Canadian Rheumatology Association
21 Annual Meeting of the Canadian Rheumatology Association February 3 to 6, Quebec City, Quebec Copyright In February 21, Quebec City hosted the annual meeting of the Canadian Rheumatology Association
Performance of the Ankylosing Spondylitis Disease Activity Score (ASDAS) in patients under biological therapies
 Performance of the Ankylosing Spondylitis Disease Activity Score (ASDAS) in patients under biological therapies 1. Introduction The Ankylosing Spondylitis Disease Activity Score (ASDAS) is a new instrument
Performance of the Ankylosing Spondylitis Disease Activity Score (ASDAS) in patients under biological therapies 1. Introduction The Ankylosing Spondylitis Disease Activity Score (ASDAS) is a new instrument
C. Assess clinical response after the first three months of treatment.
 Government Health Plan (GHP) of Puerto Rico Authorization Criteria Tumor Necrosis Factor Alpha (TNFα) Adalimumab (Humira ) Managed by MCO Section I. Prior Authorization Criteria A. Physician must submit
Government Health Plan (GHP) of Puerto Rico Authorization Criteria Tumor Necrosis Factor Alpha (TNFα) Adalimumab (Humira ) Managed by MCO Section I. Prior Authorization Criteria A. Physician must submit
ENBREL (Etanercept) 25 mg and 50 mg powder for injection and water for injections
 PRODUCT INFORMATION ENBREL Etanercept (rch) NAME OF THE MEDICINE ENBREL (Etanercept) 25 mg and 50 mg powder for injection and water for injections ENBREL (Etanercept) 25 mg and 50 mg solution for injection
PRODUCT INFORMATION ENBREL Etanercept (rch) NAME OF THE MEDICINE ENBREL (Etanercept) 25 mg and 50 mg powder for injection and water for injections ENBREL (Etanercept) 25 mg and 50 mg solution for injection
Coverage Criteria: Express Scripts, Inc. monograph dated 12/15/ months or as otherwise noted by indication
 BENEFIT DESCRIPTION AND LIMITATIONS OF COVERAGE ITEM: PRODUCT LINES: COVERED UNDER: DESCRIPTION: CPT/HCPCS Code: Company Supplying: Setting: Kineret (anakinra subcutaneous injection) Commercial HMO/PPO/CDHP
BENEFIT DESCRIPTION AND LIMITATIONS OF COVERAGE ITEM: PRODUCT LINES: COVERED UNDER: DESCRIPTION: CPT/HCPCS Code: Company Supplying: Setting: Kineret (anakinra subcutaneous injection) Commercial HMO/PPO/CDHP
2017 Blue Cross and Blue Shield of Louisiana
 Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
