How to write & publish a scientific paper Basic Concepts & Methods
|
|
|
- Samuel Ray
- 5 years ago
- Views:
Transcription
1 INAYA MEDICAL COLLEGE (IMC) NMT LECTURE 5 How to write & publish a scientific paper Basic Concepts & Methods DR. MOHAMMED MOSTAFA EMAM
2 I. Before start writing II.Writing the article III.Making the article to be published DR. MOHAMMED MOSTAFA EMAM 2
3 The title Once of the most important items to publish an article. It has to have catch up First impressions are strong impressions; a title ought therefore to be well studied, and to give, so far as its limits permit, a definite and concise indications of what is to come T Clifford Allbutt DR. MOHAMMED MOSTAFA EMAM 3
4 Do: Write the results of the article: Absence of an Effect of Liposuction on Insulin Action and Risk Factors for Coronary Heart Disease Write the aim of the study: Cardiovascular Effects of Continuous Positive Airway Pressure in Patients with Heart Failure and Obstructive Sleep Apnea Influenza Vaccination and Reduction in Hospitalizations for Cardiac Disease and Stroke among the Elderly Ask a question: Exposure to Lead in Children How Low Is Low Enough? DR. MOHAMMED MOSTAFA EMAM 4
5 Don t Use very long titles: On the addition to the method of microscopic research by a new way of producin colour-contrast between an object and its background or between parts of the object itself Use metaphoric sentences DR. MOHAMMED MOSTAFA EMAM 5
6 Structure IMRAD ointroduction omaterial and Method oresults oand odiscussion DR. MOHAMMED MOSTAFA EMAM 6
7 Introduction It has the aim of giving a brief vision of the stateof-art of the matter and justifying the study. The situation about the matter is A...There is a blank of the knowledge with respect to B...so...we did C All the asseverations will have to be supported by bibliographical references DR. MOHAMMED MOSTAFA EMAM 7
8 DR. MOHAMMED MOSTAFA EMAM 8 Rheumatoid arthritis is a systemic autoimmune disease that affects approximately 1 percent of the adult population. 1 It is characterized by chronic inflammation in the synovial membrane of affected joints that ultimately leads to loss of daily function due to chronic pain and fatigue. The majority of patients also have deterioration of cartilage and bone in the affected joints, which may eventually lead to permanent disability. Rheumatoid arthritis is associated with increased morbidity and mortality. 2 Although the precise pathogenesis of rheumatoid arthritis remains unclear, it has been postulated that multiple exogenous or endogenous antigenic triggers, or both, act in the presence of a background genetic predisposition to initiate a self-perpetuating series of autoimmune responses in the synovial compartment. 3,4 Many cell populations, including monocytes, macrophages, B cells, T cells, endothelial cells, and fibroblasts, participate in the ongoing inflammatory process. 3 The precise contribution of B cells to the immunopathogenesis of rheumatoid arthritis is not fully understood, although a number of mechanisms have been proposed. 4,5,6 However, strong evidence for a critical role of B cells in rheumatoid arthritis came from a small open-label study of rituximab in combination with cyclophosphamide and corticosteroids. 7 Rituximab is a genetically engineered chimeric anti-cd20 monoclonal antibody that is approved for the treatment of relapsed or refractory, low-grade or follicular, CD20+ B-cell non-hodgkin's lymphoma. CD20 is a B-cell surface antigen that is expressed only on pre-b and mature B cells. It is not present on stem cells and is lost before differentiation of B cells into plasma cells. Therefore, rituximab causes a selective transient depletion of the CD20+ B-cell subpopulation. 7 To confirm the role of B cells in rheumatoid arthritis, we evaluated the effect of rituximab in patients with active rheumatoid arthritis in a multicenter, randomized, double-blind, controlled study.
9 DR. MOHAMMED MOSTAFA EMAM 9 Says what is known about the disease Rheumatoid arthritis is a systemic autoimmune disease that affects approximately 1 percent of the adult population. 1 It is characterized by chronic inflammation in the synovial membrane of affected joints that ultimately leads to loss of daily function due to chronic pain and fatigue. The majority of patients also have deterioration of cartilage and bone in the affected joints, which may eventually lead to permanent disability. Rheumatoid arthritis is associated with increased morbidity and mortality. 2 Highlights the importance of a fact
10 Always backed by bibliographical references DR. MOHAMMED MOSTAFA EMAM 10
11 DR. MOHAMMED MOSTAFA EMAM 11 Blank in the knowledge Although the precise pathogenesis of rheumatoid arthritis remains unclear, it has been postulated that multiple exogenous or endogenous antigenic triggers, or both, act in the presence of a background genetic predisposition to initiate a self-perpetuating series of autoimmune responses in the synovial compartment. 3,4 Many cell populations, including monocytes, macrophages, B cells, T cells, endothelial cells, and fibroblasts, participate in the ongoing inflammatory process. 3 The precise contribution of B cells to the immunopathogenesis of rheumatoid arthritis is not fully understood, although a number of mechanisms have been proposed. 4,5,6 However, strong evidence for a critical role of B cells in rheumatoid arthritis came from a small open-label study of rituximab in combination with cyclophosphamide and corticosteroids. 7 Transition to the so...
12 DR. MOHAMMED MOSTAFA EMAM 12 Rituximab is a genetically engineered chimeric anti-cd20 monoclonal antibody that is approved for the treatment of relapsed or refractory, low-grade or follicular, CD20+ B-cell non-hodgkin's lymphoma. CD20 is a B-cell surface antigen that is expressed only on pre-b and mature B cells. It is not present on stem cells and is lost before differentiation of B cells into plasma cells. Therefore, rituximab causes a selective transient depletion of the CD20+ B-cell subpopulation. 7 To confirm the role of B cells in rheumatoid arthritis, we evaluated the effect of rituximab in patients with active rheumatoid arthritis in a multicenter, randomized, double-blind, controlled study So...What we did?
13 DR. MOHAMMED MOSTAFA EMAM 13 Material and Method How we did it? A short paragraph Must include: -What subjects we included, -Definitions -What parameters were assessed, -What instrumentation was used to assess -Was there a statistical study? What kind?
14 DR. MOHAMMED MOSTAFA EMAM 14 We studied the patients with the characteristics A and excluded those with the item B. We measured C, D and F. We defined F as C+D. To make the measurements we used the device ABC. To study if there were statistical difference between the patients, we did the test X...
15 Definitions Inclusion criteria Patients Patients were recruited from 26 rheumatology centers in 11 countries (Australia, Canada, Israel, and 8 European countries). Eligible patients were at least 21 years of age, fulfilled the revised 1987 American Rheumatism Association criteria, 1 and had active disease despite treatment with at least 10 mg of methotrexate per week. Active disease was defined by the presence of at least eight swollen and eight tender joints and at least two of the following: a serum C-reactive protein level of at least 15 mg per liter, an erythrocyte sedimentation rate of at least 28 mm per hour, or morning stiffness lasting longer than 45 minutes. In addition, eligible patients were seropositive for rheumatoid factor, as defined by a plasma rheumatoid factor level of at least 20 IU per milliliter. Patients were excluded if they had an autoimmune disease other than rheumatoid arthritis (except concurrent Sjögren's syndrome), American Rheumatism Association functional class IV disease,). Exclusion criteria DR. MOHAMMED MOSTAFA EMAM 15
16 What was done? Study Protocol Therapy with Patients were randomly assigned to receive one of four treatments: oral methotrexate at a dose of 10 mg or more per week plus placebos for rituximab and cyclophosphamide (control group), rituximab plus placebos for methotrexate and cyclophosphamide, rituximab plus cyclophosphamide in an intravenous infusion of 750 mg on days 3 and 17 plus placebo for methotrexate, and rituximab plus methotrexate at a dose of 10 mg or more a week plus placebo for cyclophosphamide. In all three groups that received rituximab (MabThera, Roche; Rituxan, Genentech and IDEC Pharmaceuticals), rituximab was administered as a 1000-mg intravenous infusion on days 1 and 15. Investigators and patients remained blinded to the assigned study medications. Clinical assessments were performed at baseline (day 1) and at weeks 12, 16, 20, and 24 according to the American College of DR. MOHAMMED MOSTAFA EMAM 16
17 DR. MOHAMMED MOSTAFA EMAM 17 What instrument was used? Rheumatology (ACR) core set of disease-activity measures. These consisted of a count of swollen joints (66 joints evaluated),... and laboratory evaluation of acute-phase reactants (serum C-reactive protein level and erythrocyte sedimentation rate). Laboratory assessments (including complete blood counts and serum biochemical analyses) were performed at screening (three weeks before baseline), on days 1, 3, 15, and 17, and at weeks 4, 8, 12, 16, 20, and Definitions
18 DR. MOHAMMED MOSTAFA EMAM 18 What do we study-measure? Clinical Outcome Measures The primary end point of the study was the proportion of patients with an ACR 50 response at week and the value for one acute-phase reactant (either serum C-reactive protein level or erythrocyte sedimentation rate). 9 Secondary outcomes included ACR 20 and ACR 70 responses (20 percent and 70 percent improvement, respectively, according to the ACR criteria), and the response according to the criteria of the European League against Rheumatism (EULAR response). 11
19 DR. MOHAMMED MOSTAFA EMAM 19 What tools we use to detect differences? Statistical Analysis Sample-size calculations were based on the assumption... On the basis of these assumptions and with the use of Fisher's exact test with a two-sided significance level of 0.05, we calculated that a sample of 40 patients per treatment group would provide the study with 82 percent power to detect a difference between the two proportions. How the assessed parameters were statistically treated
20 DR. MOHAMMED MOSTAFA EMAM 20 Results Short and concise paragraph Answers the questions on the section Material and Method We might write it in the same order than it was in the section Material and Method. Never try to explain the results or take any conclusion
21 N patients were studed. A were included. B were excluded for having 1, demographical features being D, E and F. With respect to the parameter A the result was 1, with respect to B, the result was 2. With respect to C, the result was D... DR. MOHAMMED MOSTAFA EMAM 21
22 DR. MOHAMMED MOSTAFA EMAM 22 Of the 4164 hospital admissions sampled from the participating hospitals, 3745 patient charts (89.9%) were eligible for a full screening by the stage 1 reviewers (Fig. 1). Of these, 1527 (40.8%) were assessed as positive for 1 or more screening criteria (Table 1)... Included & excluded
23 DR. MOHAMMED MOSTAFA EMAM 23 Enumerates results...the physician reviewers identified a total of 1133 injuries or complications in 858 charts. In 401 (46.7%) of these charts the injuries resulted in death, disability at the time of discharge or prolonged hospital stay. In 255 of the charts one or more of the AEs were rated 4 or higher on the 6-point causation scale (Box 1).
24 DR. MOHAMMED MOSTAFA EMAM 24 Statistical analysis (if necessary) There was a trend for AEs to occur more frequently in the teaching hospitals than in the large community or small hospitals (Table 2). The trend was significant for AEs across the 3 hospital types (p < 0.001) but not for preventable AEs (p = 0.8)...
25 DR. MOHAMMED MOSTAFA EMAM 25 Discusion Highlights the importance of the subject. It may start with a short review. Gives an explanation of the results Compares the results with those of other studies Result-comparation-explanation Signs limitations of the study A review of the state of the art can be done
26 DR. MOHAMMED MOSTAFA EMAM 26 Is an extended version of the introduction, followed by a summary of the results, comparison with those of other studies, and what our results mean. Starts where the introduction ended It is the place for new hypotesis
27 A is a very important parameter to determine B, as other studies have shown. Ours showed that A had the characteristics 1, 2 and 3, which is similar to other studies carried out to this respect. Nevertheless, It had the characteristic 4, which was different. We think that it may be due to... DR. MOHAMMED MOSTAFA EMAM 27
28 Incident rates of endometrial cancer vary more than 10-fold worldwide. 18 In addition to host susceptibility, dietary factors may play an important role... In our study population, the average intake of isoflavones from soya food was about 25 times that reported in a Western population Some of these previous studies were not specifically designed to investigate the role of soya food...the sample sizes of the previous studies were relatively small, which limited the statistical power...this population based casecontrol study... indicates that usual consumption of soya foods by adults,... is associated with a significantly reduced risk of endometrial cancer... Although not all associations were statistically significant in subgroup analyses, the different measurements produced similar results....studies with measured oestrogen concentrations are needed to better understand the joint effect of soya and endogenous oestrogen on endometrial cancer risk. DR. MOHAMMED MOSTAFA EMAM 28
29 DR. MOHAMMED MOSTAFA EMAM 29 Incident rates of endometrial cancer vary more than 10-fold worldwide. 18 In addition to host susceptibility, dietary factors may play an important role... Results of our study Review In our study population, the average intake of isoflavones from soya food was about 25 times that reported in a Western population Some of these previous studies were not specifically designed to investigate the role of soya food...the sample sizes of the previous studies were relatively small, which limited the statistical power...this population based case-control study... indicates that usual consumption of soya foods by adults,... is associated with a significantly reduced risk of endometrial cancer... Conclusion/Hypotesis Comparison with respect to other studies Although not all associations were statistically significant in subgroup analyses, the different measurements produced similar results. Limitations of the study...studies with measured oestrogen concentrations are needed to better understand the joint effect of soya and endogenous oestrogen on endometrial cancer risk.
30 DR. MOHAMMED MOSTAFA EMAM 30 AND IF IT IS A CLINICAL CASE? Introduction Clinical case Conclusions
31 DR. MOHAMMED MOSTAFA EMAM 31 Introduction: Usually based on rarity. Clinical case: A N yr-old (wo)man was admitted for a feature of...his/her clinical history was...in the physical examination the laboratory findings showed the radiological techniques were To exclude a the A test was asked, with the result 1. With the suspect of a B, test B, was asked.thus, we diagnosed the pathology P
32 DR. MOHAMMED MOSTAFA EMAM 32 A 28-year-old man was admitted to the hospital because of abdominal pain and fever. The patient had been well until 10 days earlier, when mild epigastric pain developed. Two days before admission... On physical examination, the patient did not appear to be in severe pain, and there was... The urine was positive (+) for ketones; the sediment contained......radiographs of the abdomen obtained both while the patient was supine and while he was upright showed... A cystic mass, 1.5 cm in diameter, was contiguous with the head and neck of the pancreas. The remainder of the pancreas was unremarkable Oral intake was stopped. The patient was given fluid and electrolytes as well as ranitidine, metronidazole, ampicillin, minidose heparin, and morphine, which was administered...
33 I. Before start writing II. Writing the article III.Making the article to be published (Next Lecture) Block III. Making the article to be published DR. MOHAMMED MOSTAFA EMAM 33
New Evidence reports on presentations given at ACR Improving Radiographic, Clinical, and Patient-Reported Outcomes with Rituximab
 New Evidence reports on presentations given at ACR 2009 Improving Radiographic, Clinical, and Patient-Reported Outcomes with Rituximab From ACR 2009: Rituximab Rituximab in combination with methotrexate
New Evidence reports on presentations given at ACR 2009 Improving Radiographic, Clinical, and Patient-Reported Outcomes with Rituximab From ACR 2009: Rituximab Rituximab in combination with methotrexate
1.0 Abstract. Title. Keywords. Rationale and Background
 1.0 Abstract Title A Prospective, Multi-Center Study in Rheumatoid Arthritis Patients on Adalimumab to Evaluate its Effect on Synovitis Using Ultrasonography in an Egyptian Population Keywords Synovitis
1.0 Abstract Title A Prospective, Multi-Center Study in Rheumatoid Arthritis Patients on Adalimumab to Evaluate its Effect on Synovitis Using Ultrasonography in an Egyptian Population Keywords Synovitis
I n the past, analgesics and nonsteroidal
 Rituximab in advanced rheumatoid arthritis Michael Guida BSc, MA Rheumatoid arthritis (RA) continues to have a major impact on public health. Costs to the individual and to the NHS are high, and treatment
Rituximab in advanced rheumatoid arthritis Michael Guida BSc, MA Rheumatoid arthritis (RA) continues to have a major impact on public health. Costs to the individual and to the NHS are high, and treatment
This clinical study synopsis is provided in line with Boehringer Ingelheim s Policy on Transparency and Publication of Clinical Study Data.
 abcd Clinical Study Synopsis for Public Disclosure This clinical study synopsis is provided in line with Boehringer Ingelheim s Policy on Transparency and Publication of Clinical Study Data. The synopsis
abcd Clinical Study Synopsis for Public Disclosure This clinical study synopsis is provided in line with Boehringer Ingelheim s Policy on Transparency and Publication of Clinical Study Data. The synopsis
Corporate Medical Policy
 Corporate Medical Policy Rituximab for the Treatment of Rheumatoid Arthritis File Name: Origination: Last CAP Review: Next CAP Review: Last Review: rituximab_for_the_treatment_of_rheumatoid_arthritis 4/2008
Corporate Medical Policy Rituximab for the Treatment of Rheumatoid Arthritis File Name: Origination: Last CAP Review: Next CAP Review: Last Review: rituximab_for_the_treatment_of_rheumatoid_arthritis 4/2008
TRANSPARENCY COMMITTEE OPINION. 8 November 2006
 The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 8 November 2006 MABTHERA 100 mg, concentrate for solution for infusion (CIP 560 600-3) Pack of 2 MABTHERA 500 mg,
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 8 November 2006 MABTHERA 100 mg, concentrate for solution for infusion (CIP 560 600-3) Pack of 2 MABTHERA 500 mg,
NEWS RELEASE Genentech Contacts: Media: Joe St. Martin (650) Investor: Karl Mahler Thomas Kudsk Larsen (973)
 NEWS RELEASE Genentech Contacts: Media: Joe St. Martin (650) 467-6800 Investor: Karl Mahler 011 41 61 687 8503 Thomas Kudsk Larsen (973) 235-3655 Biogen Idec Contacts: Media: Christina Chan (781) 464-3260
NEWS RELEASE Genentech Contacts: Media: Joe St. Martin (650) 467-6800 Investor: Karl Mahler 011 41 61 687 8503 Thomas Kudsk Larsen (973) 235-3655 Biogen Idec Contacts: Media: Christina Chan (781) 464-3260
SYNOPSIS. Clinical Study Report IM Double-blind Period
 Name of Sponsor/Company: Bristol-Myers Squibb Name of Finished Product: Abatacept () Name of Active Ingredient: Abatacept () Individual Study Table Referring to the Dossier SYNOPSIS (For National Authority
Name of Sponsor/Company: Bristol-Myers Squibb Name of Finished Product: Abatacept () Name of Active Ingredient: Abatacept () Individual Study Table Referring to the Dossier SYNOPSIS (For National Authority
New Evidence reports on presentations given at EULAR Rituximab for the Treatment of Rheumatoid Arthritis and Vasculitis
 New Evidence reports on presentations given at EULAR 2011 Rituximab for the Treatment of Rheumatoid Arthritis and Vasculitis Report on EULAR 2011 presentations Anti-TNF failure and response to rituximab
New Evidence reports on presentations given at EULAR 2011 Rituximab for the Treatment of Rheumatoid Arthritis and Vasculitis Report on EULAR 2011 presentations Anti-TNF failure and response to rituximab
Understanding Rheumatoid Arthritis
 Understanding Rheumatoid Arthritis Understanding Rheumatoid Arthritis What Is Rheumatoid Arthritis? 1,2 Rheumatoid arthritis (RA) is a chronic autoimmune disease. It causes joints to swell and can result
Understanding Rheumatoid Arthritis Understanding Rheumatoid Arthritis What Is Rheumatoid Arthritis? 1,2 Rheumatoid arthritis (RA) is a chronic autoimmune disease. It causes joints to swell and can result
Study synopsis of the global non-interventional study SWITCH-RA
 Study synopsis of the global non-interventional study SWITCH-RA Protocol number: MA22401 Title of Study: A global multi-centre observational study in RA patients who are non-responders or intolerant to
Study synopsis of the global non-interventional study SWITCH-RA Protocol number: MA22401 Title of Study: A global multi-centre observational study in RA patients who are non-responders or intolerant to
New Evidence reports on presentations given at EULAR Tocilizumab for the Treatment of Rheumatoid Arthritis
 New Evidence reports on presentations given at EULAR 2012 Tocilizumab for the Treatment of Rheumatoid Arthritis Report on EULAR 2012 presentations Tocilizumab monotherapy is superior to adalimumab monotherapy
New Evidence reports on presentations given at EULAR 2012 Tocilizumab for the Treatment of Rheumatoid Arthritis Report on EULAR 2012 presentations Tocilizumab monotherapy is superior to adalimumab monotherapy
GENENTECH AND BIOGEN IDEC RECEIVE A COMPLETE RESPONSE FROM FDA FOR EARLIER USE OF RITUXAN FOR RHEUMATOID ARTHRITIS
 NEWS RELEASE Genentech Contacts: Media / Advocacy: Megan Pace (650) 467-7334 Investor: Susan Morris (650) 225-6523 Karl Mahler 011 41 61 68785 03 Biogen Idec Contacts: Media: Amy Reilly (617) 914-6524
NEWS RELEASE Genentech Contacts: Media / Advocacy: Megan Pace (650) 467-7334 Investor: Susan Morris (650) 225-6523 Karl Mahler 011 41 61 68785 03 Biogen Idec Contacts: Media: Amy Reilly (617) 914-6524
Efficacy and Safety of Tocilizumab in the Treatment of Rheumatoid Arthritis and Juvenile Idiopathic Arthritis
 New Evidence reports on presentations given at EULAR 2010 Efficacy and Safety of Tocilizumab in the Treatment of Rheumatoid Arthritis and Juvenile Idiopathic Arthritis Report on EULAR 2010 presentations
New Evidence reports on presentations given at EULAR 2010 Efficacy and Safety of Tocilizumab in the Treatment of Rheumatoid Arthritis and Juvenile Idiopathic Arthritis Report on EULAR 2010 presentations
2.0 Synopsis. Adalimumab DE019 OLE (5-year) Clinical Study Report Amendment 1 R&D/06/095. (For National Authority Use Only)
 2.0 Synopsis Abbott Laboratories Name of Study Drug: Humira Name of Active Ingredient: Adalimumab Individual Study Table Referring to Part of Dossier: Volume: Page: (For National Authority Use Only) Title
2.0 Synopsis Abbott Laboratories Name of Study Drug: Humira Name of Active Ingredient: Adalimumab Individual Study Table Referring to Part of Dossier: Volume: Page: (For National Authority Use Only) Title
New Evidence reports on presentations given at EULAR Safety and Efficacy of Tocilizumab as Monotherapy and in Combination with Methotrexate
 New Evidence reports on presentations given at EULAR 2009 Safety and Efficacy of Tocilizumab as Monotherapy and in Combination with Methotrexate Report on EULAR 2009 presentations Tocilizumab inhibits
New Evidence reports on presentations given at EULAR 2009 Safety and Efficacy of Tocilizumab as Monotherapy and in Combination with Methotrexate Report on EULAR 2009 presentations Tocilizumab inhibits
corticosteroids. The effort to slow the progression of RA includes diseasemodifying (DMARDs), which include gold salts,
 Rituximab for Patients with Refractory Rheumatoid Arthritis Patty Ghazvini, PharmD, Angela Singh, PharmD, Phillip Treadwell, PharmD, Marlon Honeywell, PharmD, and Natosha Canty, MD Dr. Ghazvini and Dr.
Rituximab for Patients with Refractory Rheumatoid Arthritis Patty Ghazvini, PharmD, Angela Singh, PharmD, Phillip Treadwell, PharmD, Marlon Honeywell, PharmD, and Natosha Canty, MD Dr. Ghazvini and Dr.
Synopsis (C0743T10) CNTO 1275 Module 5.3 C0743T10. Associated with Module 5.3 of the Dossier
 Module 5.3 Protocol: EudraCT No.: 2005-003525-92 Title of the study: A Phase 2, Multicenter, Randomized, Double-blind, Placebo-controlled Trial of, a Fully Human Anti-IL-12 Monoclonal Antibody, Administered
Module 5.3 Protocol: EudraCT No.: 2005-003525-92 Title of the study: A Phase 2, Multicenter, Randomized, Double-blind, Placebo-controlled Trial of, a Fully Human Anti-IL-12 Monoclonal Antibody, Administered
Correspondence should be addressed to Martin J. Bergman;
 Autoimmune Diseases Volume 2013, Article ID 367190, 7 pages http://dx.doi.org/10.1155/2013/367190 Research Article Composite Indices Using 3 or 4 Components of the Core Data Set Have Similar Predictive
Autoimmune Diseases Volume 2013, Article ID 367190, 7 pages http://dx.doi.org/10.1155/2013/367190 Research Article Composite Indices Using 3 or 4 Components of the Core Data Set Have Similar Predictive
ClinicalTrials.gov Protocol Registration and Results System (PRS) Receipt Release Date: 01/19/2016. ClinicalTrials.gov ID: NCT
 ClinicalTrials.gov Protocol Registration and Results System (PRS) Receipt Release Date: 01/19/2016 ClinicalTrials.gov ID: NCT00595413 Study Identification Unique Protocol ID: 27905 Brief Title: Atacicept
ClinicalTrials.gov Protocol Registration and Results System (PRS) Receipt Release Date: 01/19/2016 ClinicalTrials.gov ID: NCT00595413 Study Identification Unique Protocol ID: 27905 Brief Title: Atacicept
Technology appraisal guidance Published: 25 August 2010 nice.org.uk/guidance/ta195
 Adalimumab, etanercept, infliximab, rituximab and abatacept for the treatment of rheumatoid arthritis after the failure of a TNF inhibitor Technology appraisal guidance Published: 25 August 2010 nice.org.uk/guidance/ta195
Adalimumab, etanercept, infliximab, rituximab and abatacept for the treatment of rheumatoid arthritis after the failure of a TNF inhibitor Technology appraisal guidance Published: 25 August 2010 nice.org.uk/guidance/ta195
1. Background: Infliximab is administered parenterally; therefore, it is not covered under retail pharmacy benefits.
 Subject: Infliximab (Remicade ) Original Original Committee Approval: October 13, 2006 Revised Last Committee Approval: December 3, 2008 Last Review: October 19, 2007 1. Background: Infliximab is a genetically
Subject: Infliximab (Remicade ) Original Original Committee Approval: October 13, 2006 Revised Last Committee Approval: December 3, 2008 Last Review: October 19, 2007 1. Background: Infliximab is a genetically
Polymyalgia rheumatica and giant cell arteritis
 Polymyalgia rheumatica and giant cell arteritis What is polymyalgia rheumatica? Polymyalgia rheumatica is a rheumatic disorder associated with moderate-to-severe musculoskeletal pain and stiffness in the
Polymyalgia rheumatica and giant cell arteritis What is polymyalgia rheumatica? Polymyalgia rheumatica is a rheumatic disorder associated with moderate-to-severe musculoskeletal pain and stiffness in the
Monoclonal Antibodies in the Management of Rheumatoid Arthritis Prof. John D. Isaacs
 John D Isaacs Professor of Clinical Rheumatology Director, Wilson Horne Immunotherapy Centre Newcastle University, UK 1 Rheumatoid arthritis Targeting T-cells Targeting B-cells Costimulation blockade Novel
John D Isaacs Professor of Clinical Rheumatology Director, Wilson Horne Immunotherapy Centre Newcastle University, UK 1 Rheumatoid arthritis Targeting T-cells Targeting B-cells Costimulation blockade Novel
Abatacept (Orencia) for active rheumatoid arthritis. August 2009
 Abatacept (Orencia) for active rheumatoid arthritis August 2009 This technology summary is based on information available at the time of research and a limited literature search. It is not intended to
Abatacept (Orencia) for active rheumatoid arthritis August 2009 This technology summary is based on information available at the time of research and a limited literature search. It is not intended to
Rheumatoid Arthritis. Marge Beckman FALU, FLMI Vice President RGA Underwriting Quarterly Underwriting Meeting March 24, 2011
 Rheumatoid Arthritis Marge Beckman FALU, FLMI Vice President RGA Underwriting Quarterly Underwriting Meeting March 24, 2011 The security of experience. The power of innovation. www.rgare.com Case Study
Rheumatoid Arthritis Marge Beckman FALU, FLMI Vice President RGA Underwriting Quarterly Underwriting Meeting March 24, 2011 The security of experience. The power of innovation. www.rgare.com Case Study
Efficacy and Safety of Rituximab in the Treatment of Rheumatoid Arthritis and ANCA-associated Vasculitis
 New Evidence reports on presentations given at ACR/ARHP 2010 Efficacy and Safety of Rituximab in the Treatment of Rheumatoid Arthritis and ANCA-associated Vasculitis Report on ACR/ARHP 2010 presentations
New Evidence reports on presentations given at ACR/ARHP 2010 Efficacy and Safety of Rituximab in the Treatment of Rheumatoid Arthritis and ANCA-associated Vasculitis Report on ACR/ARHP 2010 presentations
GIANT CELL ARTERITIS. Page 1 of 6 Reproduction of this material requires written permission of the Vasculitis Foundation. Copyright 2018.
 What is giant cell arteritis (GCA)? Giant cell arteritis (GCA) is a form of vasculitis a family of rare disorders characterized by inflammation of the blood vessels, which can restrict blood flow and damage
What is giant cell arteritis (GCA)? Giant cell arteritis (GCA) is a form of vasculitis a family of rare disorders characterized by inflammation of the blood vessels, which can restrict blood flow and damage
Rituxan Hycela. Rituxan Hycela (rituximab and hyaluronidase human) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.21.96 Subject: Rituxan Hycela Page: 1 of 5 Last Review Date: September 15, 2017 Rituxan Hycela Description
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.21.96 Subject: Rituxan Hycela Page: 1 of 5 Last Review Date: September 15, 2017 Rituxan Hycela Description
Corporate Medical Policy
 Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: abatacept_orencia 4/2008 2/2018 2/2019 2/2018 Description of Procedure or Service Abatacept (Orencia ), a
Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: abatacept_orencia 4/2008 2/2018 2/2019 2/2018 Description of Procedure or Service Abatacept (Orencia ), a
Clinical Policy: Rituximab (Rituxan) Reference Number: PA.CP.PHAR.260
 Clinical Policy: (Rituxan) Reference Number: PA.CP.PHAR.260 Effective Date: 01/18 Last Review Date: 04/18 Coding Implications Revision Log Description The intent of the criteria is to ensure that patients
Clinical Policy: (Rituxan) Reference Number: PA.CP.PHAR.260 Effective Date: 01/18 Last Review Date: 04/18 Coding Implications Revision Log Description The intent of the criteria is to ensure that patients
CLINICAL MEDICAL POLICY
 CLINICAL MEDICAL POLICY Policy Name: Rituxan (rituximab) Policy Number: MP-031-MD-DE Responsible Department(s): Medical Management; Clinical Pharmacy Provider Notice Date: 10/01/2017 Issue Date: 11/01/2017
CLINICAL MEDICAL POLICY Policy Name: Rituxan (rituximab) Policy Number: MP-031-MD-DE Responsible Department(s): Medical Management; Clinical Pharmacy Provider Notice Date: 10/01/2017 Issue Date: 11/01/2017
Etiology: Pathogenesis Clinical manifestation Investigation Treatment Prognosis
 Etiology: Pathogenesis Clinical manifestation Investigation Treatment Prognosis JIA is the most common rheumatic disease in childhood and a major cause of chronic disability. Etiology: Unknown, but may
Etiology: Pathogenesis Clinical manifestation Investigation Treatment Prognosis JIA is the most common rheumatic disease in childhood and a major cause of chronic disability. Etiology: Unknown, but may
Using ENBREL to Treat Rheumatoid and Psoriatic Arthritis
 Using ENBREL to Treat Rheumatoid and Psoriatic Arthritis Writing White Papers class Bellevue Community College TABLE OF CONTENTS TABLE OF CONTENTS...2 OVERVIEW...3 RHEUMATOID ARTHRITIS... 3 JUVENILE RHEUMATOID
Using ENBREL to Treat Rheumatoid and Psoriatic Arthritis Writing White Papers class Bellevue Community College TABLE OF CONTENTS TABLE OF CONTENTS...2 OVERVIEW...3 RHEUMATOID ARTHRITIS... 3 JUVENILE RHEUMATOID
London, 1 June 2006 Product name: REMICADE Procedure number: Remicade-H-240-II-73-AR SCIENTIFIC DISCUSSION 1/8
 London, 1 June 2006 Product name: REMICADE Procedure number: Remicade-H-240-II-73-AR SCIENTIFIC DISCUSSION 1/8 1. Introduction Infliximab is a chimeric human-murine IgG1κ monoclonal antibody, which binds
London, 1 June 2006 Product name: REMICADE Procedure number: Remicade-H-240-II-73-AR SCIENTIFIC DISCUSSION 1/8 1. Introduction Infliximab is a chimeric human-murine IgG1κ monoclonal antibody, which binds
KTE-C19 for relapsed or refractory mantle cell lymphoma
 NIHR Innovation Observatory Evidence Briefing: July 2017 KTE-C19 for relapsed or refractory mantle cell lymphoma NIHRIO (HSRIC) ID: 11846 NICE ID: 9122 LAY SUMMARY Mantle cell lymphoma is an uncommon type
NIHR Innovation Observatory Evidence Briefing: July 2017 KTE-C19 for relapsed or refractory mantle cell lymphoma NIHRIO (HSRIC) ID: 11846 NICE ID: 9122 LAY SUMMARY Mantle cell lymphoma is an uncommon type
FDA Perspective on Disease Modification in Schizophrenia
 FDA Perspective on Disease Modification in Schizophrenia Robert Levin, M.D. Clinical Team Leader Division of Psychiatry Products Food and Drug Administration Goals and Expectations Develop treatments that
FDA Perspective on Disease Modification in Schizophrenia Robert Levin, M.D. Clinical Team Leader Division of Psychiatry Products Food and Drug Administration Goals and Expectations Develop treatments that
9-10 VIBE ACTIVITIES. Auto-immune. Issue 189. Healthy Vibe Auto-immune disorders page 26 BODY. Issue 189. Page 1. disorders are HEALTHY.
 Y Name: VIBE ACTIVITIES E A R Years Healthy Vibe Auto-immune disorders page 26 BODY A uto-immune DiSoRDeRS occur When A PeRSon S usually helpful MMune SySTeM MiSTAkenly ATTACkS his or her body. TheRe ARe
Y Name: VIBE ACTIVITIES E A R Years Healthy Vibe Auto-immune disorders page 26 BODY A uto-immune DiSoRDeRS occur When A PeRSon S usually helpful MMune SySTeM MiSTAkenly ATTACkS his or her body. TheRe ARe
Summary of Risk Minimization Measures
 Table 6.1.4-1: Summary of Risk Minimization Measures Safety Concern Vaccination Hepatic and renal impairment Combination therapy Elderly Routine Risk Minimization Measures Specific subsection on vaccination
Table 6.1.4-1: Summary of Risk Minimization Measures Safety Concern Vaccination Hepatic and renal impairment Combination therapy Elderly Routine Risk Minimization Measures Specific subsection on vaccination
DRAFT FOR POC BOARD Clinical Commissioning Policy Proposition: Rituximab in Connective Tissue Disease associated Interstitial Lung Disease (adults)
 Clinical Commissioning Policy Proposition: Rituximab in Connective Tissue Disease associated Interstitial Lung Disease (adults) Reference: NHS England A/14/X01 CHECK Information Reader Box (IRB) to be
Clinical Commissioning Policy Proposition: Rituximab in Connective Tissue Disease associated Interstitial Lung Disease (adults) Reference: NHS England A/14/X01 CHECK Information Reader Box (IRB) to be
Horizon Scanning Centre November Secukinumab for active and progressive psoriatic arthritis. SUMMARY NIHR HSC ID: 5330
 Horizon Scanning Centre November 2012 Secukinumab for active and progressive psoriatic arthritis. SUMMARY NIHR HSC ID: 5330 Secukinumab is a high-affinity fully human monoclonal antibody that antagonises
Horizon Scanning Centre November 2012 Secukinumab for active and progressive psoriatic arthritis. SUMMARY NIHR HSC ID: 5330 Secukinumab is a high-affinity fully human monoclonal antibody that antagonises
1 Executive summary. Background
 1 Executive summary Background Rheumatoid Arthritis (RA) is the most common inflammatory polyarthropathy in the UK affecting between.5% and 1% of the population. The mainstay of RA treatment interventions
1 Executive summary Background Rheumatoid Arthritis (RA) is the most common inflammatory polyarthropathy in the UK affecting between.5% and 1% of the population. The mainstay of RA treatment interventions
Bringing the clinical experience with anakinra to the patient
 Rheumatology 2003;42(Suppl. 2):ii36 ii40 doi:10.1093/rheumatology/keg331, available online at www.rheumatology.oupjournals.org Bringing the clinical experience with anakinra to the patient S. B. Cohen
Rheumatology 2003;42(Suppl. 2):ii36 ii40 doi:10.1093/rheumatology/keg331, available online at www.rheumatology.oupjournals.org Bringing the clinical experience with anakinra to the patient S. B. Cohen
Certolizumab pegol (Cimzia) for psoriatic arthritis second line
 Certolizumab pegol (Cimzia) for psoriatic arthritis second line This technology summary is based on information available at the time of research and a limited literature search. It is not intended to
Certolizumab pegol (Cimzia) for psoriatic arthritis second line This technology summary is based on information available at the time of research and a limited literature search. It is not intended to
Review B cell targeted therapies Edward Keystone
 Available online http://arthritis-research.com/contents/7/s3/s13 Review B cell targeted therapies Edward Keystone Department of Medicine, University of Toronto, Ontario, Canada Corresponding author: Edward
Available online http://arthritis-research.com/contents/7/s3/s13 Review B cell targeted therapies Edward Keystone Department of Medicine, University of Toronto, Ontario, Canada Corresponding author: Edward
New Evidence reports on presentations given at EULAR Tocilizumab for the Treatment of Rheumatoid Arthritis and Juvenile Idiopathic Arthritis
 New Evidence reports on presentations given at EULAR 2011 Tocilizumab for the Treatment of Rheumatoid Arthritis and Juvenile Idiopathic Arthritis Report on EULAR 2011 presentations Benefit of continuing
New Evidence reports on presentations given at EULAR 2011 Tocilizumab for the Treatment of Rheumatoid Arthritis and Juvenile Idiopathic Arthritis Report on EULAR 2011 presentations Benefit of continuing
Principal Investigator. General Information. Conflict of Interest. Certification Published on The YODA Project (
 Principal Investigator First Name: Liana Last Name: Fraenkel Degree: MD, MPH Primary Affiliation: Yale University School of Medicine E-mail: christine.ramsey@gmail.com Phone number: 610-613-6745 Address:
Principal Investigator First Name: Liana Last Name: Fraenkel Degree: MD, MPH Primary Affiliation: Yale University School of Medicine E-mail: christine.ramsey@gmail.com Phone number: 610-613-6745 Address:
SCIENTIFIC DISCUSSION
 European Medicines Agency London, 20 September 2007 Product name: Remicade Procedure number: EMEA/H/C/240/II/95 SCIENTIFIC DISCUSSION 7 Westferry Circus, Canary Wharf, London, E14 4HB, UK Tel. (44-20)
European Medicines Agency London, 20 September 2007 Product name: Remicade Procedure number: EMEA/H/C/240/II/95 SCIENTIFIC DISCUSSION 7 Westferry Circus, Canary Wharf, London, E14 4HB, UK Tel. (44-20)
FOR PUBLIC CONSULTATION ONLY. Evidence Review: Rituximab for immunoglobulin G4-related disease (IgG4-RD)
 Evidence Review: Rituximab for immunoglobulin G4-related disease (IgG4-RD) NHS England FOR PUBLIC CONSULTATION ONLY Evidence Review: Rituximab for immunoglobulin G4-related disease (IgG4- RD) First published:
Evidence Review: Rituximab for immunoglobulin G4-related disease (IgG4-RD) NHS England FOR PUBLIC CONSULTATION ONLY Evidence Review: Rituximab for immunoglobulin G4-related disease (IgG4- RD) First published:
Understanding your diagnosis. Dr Graham Collins Consultant Haemtologist Oxford University Hospitals
 Understanding your diagnosis Dr Graham Collins Consultant Haemtologist Oxford University Hospitals Common questions I get asked What is lymphoma? What subtype do I have and what does that mean? What are
Understanding your diagnosis Dr Graham Collins Consultant Haemtologist Oxford University Hospitals Common questions I get asked What is lymphoma? What subtype do I have and what does that mean? What are
A cost effectiveness analysis of treatment options for methotrexate-naive rheumatoid arthritis Choi H K, Seeger J D, Kuntz K M
 A cost effectiveness analysis of treatment options for methotrexate-naive rheumatoid arthritis Choi H K, Seeger J D, Kuntz K M Record Status This is a critical abstract of an economic evaluation that meets
A cost effectiveness analysis of treatment options for methotrexate-naive rheumatoid arthritis Choi H K, Seeger J D, Kuntz K M Record Status This is a critical abstract of an economic evaluation that meets
Coverage Criteria: Express Scripts, Inc. monograph dated 12/15/ months or as otherwise noted by indication
 BENEFIT DESCRIPTION AND LIMITATIONS OF COVERAGE ITEM: PRODUCT LINES: COVERED UNDER: DESCRIPTION: CPT/HCPCS Code: Company Supplying: Setting: Kineret (anakinra subcutaneous injection) Commercial HMO/PPO/CDHP
BENEFIT DESCRIPTION AND LIMITATIONS OF COVERAGE ITEM: PRODUCT LINES: COVERED UNDER: DESCRIPTION: CPT/HCPCS Code: Company Supplying: Setting: Kineret (anakinra subcutaneous injection) Commercial HMO/PPO/CDHP
NEW EFFECTIVE TREATMENTS FOR PSORIATIC ARTHRITIS PATIENTS Promising data to support two new drug classes
 Annual European Congress of Rheumatology (EULAR) 2017 Madrid, Spain, 14-17 June 2017 NEW EFFECTIVE TREATMENTS FOR PSORIATIC ARTHRITIS PATIENTS Promising data to support two new drug classes Madrid, Spain,
Annual European Congress of Rheumatology (EULAR) 2017 Madrid, Spain, 14-17 June 2017 NEW EFFECTIVE TREATMENTS FOR PSORIATIC ARTHRITIS PATIENTS Promising data to support two new drug classes Madrid, Spain,
Cell-Mediated Immunity and T Lymphocytes
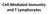 Cell-Mediated Immunity and T Lymphocytes T helper (Th) Cells Peripheral lymhoid tissue thymus Lymphoid stem cell CD8+ CD4+ CD4+ Treg CD8+ CTL + antigen Cytotoxic T lymphocyte CD4+ Th Helper T cell CD4+
Cell-Mediated Immunity and T Lymphocytes T helper (Th) Cells Peripheral lymhoid tissue thymus Lymphoid stem cell CD8+ CD4+ CD4+ Treg CD8+ CTL + antigen Cytotoxic T lymphocyte CD4+ Th Helper T cell CD4+
Synopsis (C0524T12 GO LIVE)
 Protocol: EudraCT No.: 2005-003232-21 Title of the study: A Multicenter, Randomized, Double-blind, Placebo-controlled Trial of Golimumab, a Fully Human Anti-TNFα Monoclonal Antibody, Administered Intravenously,
Protocol: EudraCT No.: 2005-003232-21 Title of the study: A Multicenter, Randomized, Double-blind, Placebo-controlled Trial of Golimumab, a Fully Human Anti-TNFα Monoclonal Antibody, Administered Intravenously,
PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert.
 PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. GENERIC DRUG NAME / COMPOUND NUMBER: Tofacitinib / CP-690,550
PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. GENERIC DRUG NAME / COMPOUND NUMBER: Tofacitinib / CP-690,550
Mechanisms of Autontibodies
 Mechanisms of Autontibodies Production in Rheumatic Diseases Eisa Salehi PhD Tehran University of Medical Sciences Immunology Department Introduction Rheumatic diseases: Cause inflammation, swelling, and
Mechanisms of Autontibodies Production in Rheumatic Diseases Eisa Salehi PhD Tehran University of Medical Sciences Immunology Department Introduction Rheumatic diseases: Cause inflammation, swelling, and
Rheumatoid Arthritis
 Rheumatoid Arthritis Rheumatoid arthritis (RA) is an autoimmune disease that causes chronic inflammation of the joints. Autoimmune diseases are illnesses that occur when the body's tissues are mistakenly
Rheumatoid Arthritis Rheumatoid arthritis (RA) is an autoimmune disease that causes chronic inflammation of the joints. Autoimmune diseases are illnesses that occur when the body's tissues are mistakenly
The Hospital for Sick Children Technology Assessment at SickKids (TASK)
 The Hospital for Sick Children Technology Assessment at SickKids (TASK) THE USE OF BIOLOGIC RESPONSE MODIFIERS IN POLYARTICULAR-COURSE JUVENILE IDIOPATHIC ARTHRITIS Report No. 2010-01 Date: January 11,
The Hospital for Sick Children Technology Assessment at SickKids (TASK) THE USE OF BIOLOGIC RESPONSE MODIFIERS IN POLYARTICULAR-COURSE JUVENILE IDIOPATHIC ARTHRITIS Report No. 2010-01 Date: January 11,
Clinical Commissioning Policy Proposition: Tocilizumab for Giant cell arteritis (adults)
 Clinical Commissioning Policy Proposition: Tocilizumab for Giant cell arteritis (adults) Version Number: NHS England A13X12/01 Information Reader Box (IRB) to be inserted on inside front cover for documents
Clinical Commissioning Policy Proposition: Tocilizumab for Giant cell arteritis (adults) Version Number: NHS England A13X12/01 Information Reader Box (IRB) to be inserted on inside front cover for documents
When is it Rheumatoid Arthritis When to Refer
 When is it Rheumatoid Arthritis When to Refer Nancy A. Brown, DO Spring 2015 When is it Rheumatoid Arthritis When to Refer Learning objectives To review the definition and epidemiology of Rheumatoid Arthritis
When is it Rheumatoid Arthritis When to Refer Nancy A. Brown, DO Spring 2015 When is it Rheumatoid Arthritis When to Refer Learning objectives To review the definition and epidemiology of Rheumatoid Arthritis
Criteria Inclusion criteria Exclusion criteria. despite treatment with csdmards, NSAIDs, and/or previous anti-tnf therapy and/or
 Supplementary Material Table S1 Eligibility criteria (PICOS) for the SLR Criteria Inclusion criteria Exclusion criteria Population Adults (aged 18 years) with active PsA despite treatment with csdmards,
Supplementary Material Table S1 Eligibility criteria (PICOS) for the SLR Criteria Inclusion criteria Exclusion criteria Population Adults (aged 18 years) with active PsA despite treatment with csdmards,
Page: 17 December 2012 (Study M13-692) 22 October 2013 (Study M13-692)
 2.0 Synopsis AbbVie Inc. Name of Study Drug: Adalimumab Name of Active Ingredient: D2E7 Individual Study Table Referring to Part of Dossier: Volume: Page: (For National Authority Use Only) Title of Studies:
2.0 Synopsis AbbVie Inc. Name of Study Drug: Adalimumab Name of Active Ingredient: D2E7 Individual Study Table Referring to Part of Dossier: Volume: Page: (For National Authority Use Only) Title of Studies:
Center for Evidence-based Policy
 P&T Committee Brief Targeted Immune Modulators: Comparative Drug Class Review Alison Little, MD Center for Evidence-based Policy Oregon Health & Science University 3455 SW US Veterans Hospital Road, SN-4N
P&T Committee Brief Targeted Immune Modulators: Comparative Drug Class Review Alison Little, MD Center for Evidence-based Policy Oregon Health & Science University 3455 SW US Veterans Hospital Road, SN-4N
Horizon Scanning Technology Summary. Abatacept (Orencia) for juvenile idiopathic arthritis. National Horizon Scanning Centre.
 Horizon Scanning Technology Summary National Horizon Scanning Centre Abatacept (Orencia) for juvenile idiopathic arthritis June 2007 This technology summary is based on information available at the time
Horizon Scanning Technology Summary National Horizon Scanning Centre Abatacept (Orencia) for juvenile idiopathic arthritis June 2007 This technology summary is based on information available at the time
Cochrane Musculoskeletal Group: Plain Language Summary (PLS) Guide for Authors
 Cochrane Musculoskeletal Group: Plain Language Summary (PLS) Guide for Authors Cochrane Musculoskeletal Group, October 2011 Contents 1. About Cochrane Plain Language Summaries (PLS)... 2 Who is this guide
Cochrane Musculoskeletal Group: Plain Language Summary (PLS) Guide for Authors Cochrane Musculoskeletal Group, October 2011 Contents 1. About Cochrane Plain Language Summaries (PLS)... 2 Who is this guide
Scintigraphic Findings and Serum Matrix Metalloproteinase 3 and Vascular Endothelial Growth Factor Levels in Patients with Polymyalgia Rheumatica
 The Open General and Internal Medicine Journal, 29, 3, 53-57 53 Open Access Scintigraphic Findings and Serum Matrix Metalloproteinase 3 and Vascular Endothelial Growth Factor Levels in Patients with Polymyalgia
The Open General and Internal Medicine Journal, 29, 3, 53-57 53 Open Access Scintigraphic Findings and Serum Matrix Metalloproteinase 3 and Vascular Endothelial Growth Factor Levels in Patients with Polymyalgia
RITUXAN (rituximab and hyaluronidase human)
 Drug Prior Authorization Guideline RITUXIMAB products J9310 RITUXAN (rituximab and hyaluronidase human) PA9847 Covered Service: Prior Authorization Required: Additional Information: Yes when meets criteria
Drug Prior Authorization Guideline RITUXIMAB products J9310 RITUXAN (rituximab and hyaluronidase human) PA9847 Covered Service: Prior Authorization Required: Additional Information: Yes when meets criteria
Canadian Society of Internal Medicine Annual Meeting 2016 Montreal, QC
 Canadian Society of Internal Medicine Annual Meeting 2016 Montreal, QC Update on the Treatment of Rheumatoid Arthritis Sabrina Fallavollita MDCM McGill University Canadian Society of Internal Medicine
Canadian Society of Internal Medicine Annual Meeting 2016 Montreal, QC Update on the Treatment of Rheumatoid Arthritis Sabrina Fallavollita MDCM McGill University Canadian Society of Internal Medicine
A. Kopchev, S.Monov, D. Kyurkchiev, I.Ivanova, T. Georgiev (UMHAT St. Ivan Rilski, Medical University - Sofia, Bulgaria)
 International Journal of Pharmaceutical Science Invention ISSN (Online): 2319 6718, ISSN (Print): 2319 670X Volume 6 Issue 7 July 2017 PP. 08-12 Vascular endothelial growth factor (VEGF), cartilage oligomeric
International Journal of Pharmaceutical Science Invention ISSN (Online): 2319 6718, ISSN (Print): 2319 670X Volume 6 Issue 7 July 2017 PP. 08-12 Vascular endothelial growth factor (VEGF), cartilage oligomeric
Rheumatoid arthritis
 Rheumatoid arthritis 1 Definition Rheumatoid arthritis is one of the most common inflammatory disorders affecting the population worldwide. It is a systemic inflammatory disease which affects not only
Rheumatoid arthritis 1 Definition Rheumatoid arthritis is one of the most common inflammatory disorders affecting the population worldwide. It is a systemic inflammatory disease which affects not only
OU Children s Physicians Pediatric Arthritis Center
 Please complete the following questionnaire for your child: Patient Name: Birth Date: Parent/Caretaker Name: Primary Care Physician (Full Name, City, & State) Mother s Occupation: Fathers Occupation: Name
Please complete the following questionnaire for your child: Patient Name: Birth Date: Parent/Caretaker Name: Primary Care Physician (Full Name, City, & State) Mother s Occupation: Fathers Occupation: Name
Efficacy of B-Cell Targeted Therapy with Rituximab in Patients with Rheumatoid Arthritis
 The new england journal of medicine original article Efficacy of B-Cell Targeted Therapy with Rituximab in Patients with Rheumatoid Arthritis Jonathan C.W. Edwards, M.D., Leszek Szczepański, M.D., Ph.D.,
The new england journal of medicine original article Efficacy of B-Cell Targeted Therapy with Rituximab in Patients with Rheumatoid Arthritis Jonathan C.W. Edwards, M.D., Leszek Szczepański, M.D., Ph.D.,
MEDICAL COVERAGE GUIDELINES ORIGINAL EFFECTIVE DATE: 08/19/14 SECTION: DRUGS LAST REVIEW DATE: LAST CRITERIA REVISION DATE: ARCHIVE DATE:
 RITUXAN (rituximab) Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Medical Coverage Guideline
RITUXAN (rituximab) Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Medical Coverage Guideline
Rheumatoid Arthritis. Manish Relan, MD FACP RhMSUS Arthritis & Rheumatology Care Center. Jacksonville, FL (904)
 Rheumatoid Arthritis Manish Relan, MD FACP RhMSUS Arthritis & Rheumatology Care Center. Jacksonville, FL (904) 503-6999. 1 Disclosures Speaker Bureau: Abbvie 2 Objectives Better understand the pathophysiology
Rheumatoid Arthritis Manish Relan, MD FACP RhMSUS Arthritis & Rheumatology Care Center. Jacksonville, FL (904) 503-6999. 1 Disclosures Speaker Bureau: Abbvie 2 Objectives Better understand the pathophysiology
Ustekinumab (Stelara) for psoriatic arthritis second line after disease modifying anti rheumatic drugs (DMARDs)
 Ustekinumab (Stelara) for psoriatic arthritis second line after disease modifying anti rheumatic drugs (DMARDs) January 2010 This technology summary is based on information available at the time of research
Ustekinumab (Stelara) for psoriatic arthritis second line after disease modifying anti rheumatic drugs (DMARDs) January 2010 This technology summary is based on information available at the time of research
Review Article Efficacy and Safety of Iguratimod for the Treatment of Rheumatoid Arthritis
 Clinical and Developmental Immunology Volume 2013, Article ID 310628, 16 pages http://dx.doi.org/10.1155/2013/310628 Review Article Efficacy and Safety of Iguratimod for the Treatment of Rheumatoid Arthritis
Clinical and Developmental Immunology Volume 2013, Article ID 310628, 16 pages http://dx.doi.org/10.1155/2013/310628 Review Article Efficacy and Safety of Iguratimod for the Treatment of Rheumatoid Arthritis
Is Tanezumab More Effective than a Placebo in Reducing Pain in Patients with Osteoarthritis?
 Philadelphia College of Osteopathic Medicine DigitalCommons@PCOM PCOM Physician Assistant Studies Student Scholarship Student Dissertations, Theses and Papers 2018 Is Tanezumab More Effective than a Placebo
Philadelphia College of Osteopathic Medicine DigitalCommons@PCOM PCOM Physician Assistant Studies Student Scholarship Student Dissertations, Theses and Papers 2018 Is Tanezumab More Effective than a Placebo
LOCALLY AVAILABLE BIOLOGIC AGENTS IN THE TREATMENT OF PSORIATIC ARTHRITIS
 Locally Available Biologic Agents in the Treatment of Psoriatic Arthritis 253 Phil. J. Internal Medicine, 47: 253-259, Nov.-Dec., 2009 LOCALLY AVAILABLE BIOLOGIC AGENTS IN THE TREATMENT OF PSORIATIC ARTHRITIS
Locally Available Biologic Agents in the Treatment of Psoriatic Arthritis 253 Phil. J. Internal Medicine, 47: 253-259, Nov.-Dec., 2009 LOCALLY AVAILABLE BIOLOGIC AGENTS IN THE TREATMENT OF PSORIATIC ARTHRITIS
National Institute for Health and Clinical Excellence Level 1A, City Tower Piccadilly Plaza Manchester M1 4BD
 xxxxx xxxxxx xxxxxx xxxxxxx - xxxxxxxxx x National Institute for Health and Clinical Excellence Level 1A, City Tower Piccadilly Plaza Manchester M1 4BD Dear xxxxx, 1st September 2011 Comments on the August
xxxxx xxxxxx xxxxxx xxxxxxx - xxxxxxxxx x National Institute for Health and Clinical Excellence Level 1A, City Tower Piccadilly Plaza Manchester M1 4BD Dear xxxxx, 1st September 2011 Comments on the August
Pain or stiffness in joints after periods of inactivity or excessive use
 Arthritis Awareness* Some older adults call it Arthur ; others refer to it as their constant compassion, but most describe it as extremely painful Arthritis is a chronic joint disease It is commonly believed
Arthritis Awareness* Some older adults call it Arthur ; others refer to it as their constant compassion, but most describe it as extremely painful Arthritis is a chronic joint disease It is commonly believed
We are IntechOpen, the world s leading publisher of Open Access books Built by scientists, for scientists. International authors and editors
 We are IntechOpen, the world s leading publisher of Open Access books Built by scientists, for scientists 3,700 108,500 1.7 M Open access books available International authors and editors Downloads Our
We are IntechOpen, the world s leading publisher of Open Access books Built by scientists, for scientists 3,700 108,500 1.7 M Open access books available International authors and editors Downloads Our
Infections and Biologics
 Overview Infections and Biologics James Galloway What is the risk of infection with biologics? Are some patients at greater risk? Are some drugs safer? Case scenario You recently commenced Judith, a 54
Overview Infections and Biologics James Galloway What is the risk of infection with biologics? Are some patients at greater risk? Are some drugs safer? Case scenario You recently commenced Judith, a 54
Annual influenza epidemics due to influenza
 Paediatrica Indonesiana VOLUME 51 January NUMER 1 Original Article Immunogenicity and safety of a trivalent inactivated influenza vaccine Eddy Fadlyana 1, Kusnandi Rusmil 1, Novilia Sjafri achtiar, Rachmat
Paediatrica Indonesiana VOLUME 51 January NUMER 1 Original Article Immunogenicity and safety of a trivalent inactivated influenza vaccine Eddy Fadlyana 1, Kusnandi Rusmil 1, Novilia Sjafri achtiar, Rachmat
National Horizon Scanning Centre. Rituximab (MabThera) for chronic lymphocytic leukaemia. September 2007
 Rituximab (MabThera) for chronic lymphocytic leukaemia This technology summary is based on information available at the time of research and a limited literature search. It is not intended to be a definitive
Rituximab (MabThera) for chronic lymphocytic leukaemia This technology summary is based on information available at the time of research and a limited literature search. It is not intended to be a definitive
Corporate Medical Policy
 Corporate Medical Policy Monoclonal Antibodies for Non-Hodgkin Lymphoma and Acute Myeloid File Name: Origination: Last CAP Review: Next CAP Review: Last Review: monoclonal_antibodies_for_non_hodgkin_lymphoma_acute_myeloid_leukemia
Corporate Medical Policy Monoclonal Antibodies for Non-Hodgkin Lymphoma and Acute Myeloid File Name: Origination: Last CAP Review: Next CAP Review: Last Review: monoclonal_antibodies_for_non_hodgkin_lymphoma_acute_myeloid_leukemia
THE CARDIOVASCULAR INFLAMMATORY CONTINUUM DR AB MAHARAJ
 THE CARDIOVASCULAR INFLAMMATORY CONTINUUM DR AB MAHARAJ Disclosures: On National Advisory Boards of: (1) Pfizer Pharmaceuticals (2) MSD (3) Roche Pharmaceuticals (4) Abbott International: AfME Rheumatology
THE CARDIOVASCULAR INFLAMMATORY CONTINUUM DR AB MAHARAJ Disclosures: On National Advisory Boards of: (1) Pfizer Pharmaceuticals (2) MSD (3) Roche Pharmaceuticals (4) Abbott International: AfME Rheumatology
Horizon Scanning Centre January Apremilast for psoriatic arthritis SUMMARY NIHR HSC ID: 3716
 Horizon Scanning Centre January 2013 Apremilast for psoriatic arthritis SUMMARY NIHR HSC ID: 3716 This briefing is based on information available at the time of research and a limited literature search.
Horizon Scanning Centre January 2013 Apremilast for psoriatic arthritis SUMMARY NIHR HSC ID: 3716 This briefing is based on information available at the time of research and a limited literature search.
TREAT-TO-TARGET IN RHEUMATOID ARTHRITIS
 TREAT-TO-TARGET IN RHEUMATOID ARTHRITIS To receive up to 10 CME credits for this activity, complete the evaluation, attestation and post-test answer sheet (minimum passing grade of 70%) and return all
TREAT-TO-TARGET IN RHEUMATOID ARTHRITIS To receive up to 10 CME credits for this activity, complete the evaluation, attestation and post-test answer sheet (minimum passing grade of 70%) and return all
Supplementary appendix
 Supplementary appendix This appendix formed part of the original submission and has been peer reviewed. We post it as supplied by the authors. Supplement to: Blum CA, Nigro N, Briel M, et al. Adjunct prednisone
Supplementary appendix This appendix formed part of the original submission and has been peer reviewed. We post it as supplied by the authors. Supplement to: Blum CA, Nigro N, Briel M, et al. Adjunct prednisone
Summary of the risk management plan (RMP) for Gazyvaro (obinutuzumab)
 EMA/319729/2014 Summary of the risk management plan (RMP) for Gazyvaro (obinutuzumab) This is a summary of the risk management plan (RMP) for Gazyvaro, which details the measures to be taken in order to
EMA/319729/2014 Summary of the risk management plan (RMP) for Gazyvaro (obinutuzumab) This is a summary of the risk management plan (RMP) for Gazyvaro, which details the measures to be taken in order to
1.0 Abstract. Title. Keywords. Adalimumab, Rheumatoid Arthritis, Effectiveness, Safety. Rationale and Background
 1.0 Abstract Title Assessment of the safety of adalimumab in rheumatoid arthritis (RA) patients showing rapid progression of structural damage of the joints, who have no prior history of treatment with
1.0 Abstract Title Assessment of the safety of adalimumab in rheumatoid arthritis (RA) patients showing rapid progression of structural damage of the joints, who have no prior history of treatment with
BESPONSA (inotuzumab ozogamicin)
 BESPONSA (inotuzumab ozogamicin) Fact Sheet BESPONSA (inotuzumab ozogamicin) is an antibody-drug conjugate (ADC) composed of a monoclonal antibody (mab) targeting CD22, a cell surface antigen expressed
BESPONSA (inotuzumab ozogamicin) Fact Sheet BESPONSA (inotuzumab ozogamicin) is an antibody-drug conjugate (ADC) composed of a monoclonal antibody (mab) targeting CD22, a cell surface antigen expressed
2.0 Synopsis. Adalimumab DE018 OLE (5 year) Clinical Study Report R&D/06/369. (For National Authority Use Only) to Part of Dossier: Volume:
 2.0 Synopsis Abbott Laboratories Name of Study Drug: Name of Active Ingredient: Individual Study Table Referring to Part of Dossier: Volume: Page: (For National Authority Use Only) Title of Study: A Multi-center
2.0 Synopsis Abbott Laboratories Name of Study Drug: Name of Active Ingredient: Individual Study Table Referring to Part of Dossier: Volume: Page: (For National Authority Use Only) Title of Study: A Multi-center
2.0 Synopsis. Adalimumab (HUMIRA ) W Clinical Study Report R&D/15/0629. Individual Study Table Referring to Part of Dossier: Volume:
 2.0 Synopsis AbbVie Inc. Name of Study Drug: Adalimumab / HUMIRA Name of Active Ingredient: Adalimumab Individual Study Table Referring to Part of Dossier: Volume: Page: (For National Authority Use Only)
2.0 Synopsis AbbVie Inc. Name of Study Drug: Adalimumab / HUMIRA Name of Active Ingredient: Adalimumab Individual Study Table Referring to Part of Dossier: Volume: Page: (For National Authority Use Only)
CIBMTR Center Number: CIBMTR Recipient ID: RETIRED. Today s Date: Date of HSCT for which this form is being completed:
 Juvenile Idiopathic Arthritis Pre-HSCT Data Sequence Number: Date Received: Registry Use Only Today s Date: Date of HSCT for which this form is being completed: HSCT type: autologous allogeneic, allogeneic,
Juvenile Idiopathic Arthritis Pre-HSCT Data Sequence Number: Date Received: Registry Use Only Today s Date: Date of HSCT for which this form is being completed: HSCT type: autologous allogeneic, allogeneic,
ERROR CORRECTION FORM
 Juvenile Idiopathic Arthritis Pre-HSCT Data Sequence Number: Registry Use Only Date of HSCT for which this form is being completed: HSCT type: autologous allogeneic, allogeneic, syngeneic unrelated related
Juvenile Idiopathic Arthritis Pre-HSCT Data Sequence Number: Registry Use Only Date of HSCT for which this form is being completed: HSCT type: autologous allogeneic, allogeneic, syngeneic unrelated related
Horizon Scanning Technology Summary. Adalimumab (Humira) for juvenile idiopathic arthritis. National Horizon Scanning Centre.
 Horizon Scanning Technology Summary National Horizon Scanning Centre Adalimumab (Humira) for juvenile idiopathic arthritis June 2007 This technology summary is based on information available at the time
Horizon Scanning Technology Summary National Horizon Scanning Centre Adalimumab (Humira) for juvenile idiopathic arthritis June 2007 This technology summary is based on information available at the time
Guide to Understanding and Managing Arthritis
 Guide to Understanding and Managing Arthritis The content in this guide is not intended to be a substitute for professional medical advice, diagnosis, or treatment. Always seek the advice of your physician
Guide to Understanding and Managing Arthritis The content in this guide is not intended to be a substitute for professional medical advice, diagnosis, or treatment. Always seek the advice of your physician
