Cytoskeleton changes of airway smooth muscle cells in juvenile rats with airway remodeling in asthma and the RhoA/ROCK signaling pathway mechanism
|
|
|
- Kenneth Wade
- 6 years ago
- Views:
Transcription
1 Cytoskeleton changes of airway smooth muscle cells in juvenile rats with airway remodeling in asthma and the RhoA/ROCK signaling pathway mechanism B. Wei 1, Y.X. Shang 2, M. Li 2, J. Jiang 1 and H. Zhang 2 1 Department of Pediatrics, General Hospital of Shenyang Military Area Command, Shenyang, China 2 Department of Pediatrics, Shengjing Hospital of China Medical University, China Medical University, Shenyang, China Corresponding author: B. Wei bwyxcn@163.com Genet. Mol. Res. 13 (1): (2014) Received August 29, 2013 Accepted November 7, 2013 Published January 22, 2014 DOI ABSTRACT. This article aimed to investigate changes in the cytoskeleton of airway smooth muscle cells (ASMCs) in juvenile rats with airway remodeling in asthma. We further investigated the involvement of the RhoA/ROCK signaling pathway mechanism. Rat models of airway remodeling in asthma were established by antigen sensitization with ovalbumin for 2, 4, 6, and 8 weeks. The control group was treated with normal saline instead of ovalbumin. In the intervention group, after 8 weeks of culture, ASMCs were treated with the ROCKspecific inhibitor Y Immunofluorescence, real-time polymerase chain reaction, and Western blot analyses were used to observe changes in the cytoskeleton (F-actin and α-tubulin) of ASMCs and expressions of RhoA and ROCK. The asthmatic groups had significantly higher average gray values of F-actin in ASMCs compared to the control group (P < 0.01), and these values for the intervention group were
2 B. Wei et al. 560 significantly lower than those of the 8-week asthmatic group (P < 0.05). Expression levels of the α-tubulin protein in the asthmatic groups were all significantly higher than those of the control group (P < 0.01), and the levels in the intervention group were significantly reduced (P < 0.05). Expressions of RhoA and ROCK mrna and proteins in all asthmatic groups were significantly higher than those of the control group (P < 0.01). Together, these results demonstrate substantial changes of the ASMC cytoskeleton and abnormal expressions of RhoA and ROCK mrna and proteins in juvenile rats with airway remodeling in asthma. Key words: Asthma; Airway remodeling; Cytoskeleton; RhoA/ROCK; Rat INTRODUCTION The cytoskeleton is a three-dimensional grid structure composed of a variety of structural and contractile proteins in a particular pattern. It is an important and substantial foundation for maintaining specific cell morphologies and exercising systolic functions. The cytoskeleton exists in three main forms including microfilaments, microtubules, and intermediate filaments. Recent studies have revealed that the RhoA/ROCK signaling pathway in the small G protein family can regulate the reconstruction of microfilaments in smooth muscle cells by mediating protein phosphorylation, thus affecting the biological behavior of cells (Fukata et al., 2001; Stamenović, 2008). Therefore, investigating the regulatory mechanism of the RhoA/ROCK signaling pathway involved in the reconstruction of airway smooth muscle cells (ASMCs) in airway remodeling in asthma, and its pathological physiological significance, can contribute to greater understanding of the pathogenesis of this disease. However, few studies of this nature have been conducted to date. In this study, ASMCs of Sprague-Dawley (SD) rats were cultured and treated with a ROCK inhibitor. Immunofluorescence, real-time quantitative polymerase chain reaction (qpcr), and Western blot methods were used to observe expressions of F-actin, α-tubulin, RhoA, and ROCK. Changes in the cytoskeleton of ASMCs and the RhoA/ROCK signaling pathway mechanism in airway remodeling in asthma are discussed. MATERIAL AND METHODS Experimental animals and materials Specific-pathogen-free juvenile female SD rats (60-80 g) were obtained from the Experimental Animal Center of China Medical University. RNAiso TM Plus and the qpcr kit were obtained from TaKaRa Co., Ltd. (Mountain View, CA, USA). RhoA and ROCK primers and the SeeBlue TM protein marker were purchased from the Invitrogen Biotechnology Co. Ltd. (Grand Island, NY, USA). Dulbecco s modified Eagle s medium/ham s F-12 (DMEM/F12) and fetal bovine serum (FBS) were purchased from HyClone Laboratories, Inc. (Logan, UT, USA). Collagenase type IV was obtained from Gibco Inc. (USA). Phalloidin-fluorescein isothiocyante (FITC), 4',6-diamidino-2-phenylindole (DAPI) and Y were purchased from
3 Rat models of airway remodeling in asthma 561 Sigma-Aldrich, Inc. (St. Louis, MO, USA). Rabbit anti-rat α-tubulin antibody was obtained from Santa Cruz Biotechnology, Inc. (Santa Cruz, CA, USA), and the Total Protein Extraction Kit was purchased from Nanjing KGI Biological Technology Development Co., Ltd. (China). Establishment of rat models of airway remodeling in asthma Rats were injected with 1 ml antigen sensitization liquid (100 mg ovalbumin, 100 mg aluminum hydroxide, and 5 x 10 9 inactivated Bordetella pertussis) on the 1st and 8th day of the experiment. Rats injected with an equal volume of saline were used as controls. As of the 15th day of the experiment, rats were excited by ultrasonic atomizing inhalation with 1% ovalbumin for 30 min 3 times per week. Rats were divided into control group, 2-week asthmatic group, 4-week asthmatic group, 6-week asthmatic group, 8-week asthmatic group, and the intervention group. The excitation was performed for 2, 4, 6, and 8 weeks in each asthmatic group. The control group was treated with normal saline instead of ovalbumin for 2 weeks. After 8 weeks of culture, ASMCs in rats of the intervention group were treated with 10 μm ROCK-specific inhibitor, Y-27632, followed by 24 h of continued culture. Primary culture of rat ASMCs After routine disinfection and anesthesia, rats were executed by drawing left ventricular blood. Under sterile conditions, the trachea was isolated, followed by careful removal of outer and internal membranes. The tracheal smooth muscle was removed, cut into pieces, and 0.1% trypsin was added, followed by oscillation in a water bath at 37 C for 10 min. After centrifugation at 1000 rpm for 5 min, the supernatant was removed. Then, 0.1% collagenase type IV was added, followed by oscillation at 37 C for 30 min. After centrifugation at 1000 rpm for 5 min, the supernatant was removed. DMEM containing 10% FBS was added to terminate the reaction. After filtering with a 100-μm mesh cell sieve, ASMCs were inoculated into a 50-mL bottle for culture at 37 C, followed by purification according to the faster adherence characteristic of fibroblasts. After the bottle bottom was completely covered with cells, the cells were passaged (1:2) and 3rd-generation cells were collected for subsequent use. Detection of F-actin and α-tubulin by immunofluorescence staining ASMCs were fixed with 4% paraformaldehyde at room temperature for 20 min, followed by treatment with 0.1% Triton X-100 for 10 min. In dark conditions, 5 μg/ml phalloidin-fitc was added for staining at 37 C for 1 h. After sealing with 50% glycerol, slices were observed by fluorescence microscopy for F-actin and α-tubulin detection. Detection of RhoA and ROCK mrna expressions by real-time qpcr Standard curves of the target RhoA and ROCK genes, and the housekeeping gene, GAPDH, were constructed using the SYBR Green method. According to the standard curve, the three genes were quantitated. The relative expression level of target gene mrna was calculated as follows: copy number of target gene/copy number of GAPDH gene. The primers used were as follows: RhoA primer (upstream, 5'-AACTGGTGATTGTTGGTG-3ꞌ and down-
4 B. Wei et al. 562 stream, 5ꞌ-CTGCCACATAGTTTTCAA-3'), amplified fragment length, 114 bp; ROCK primer (upstream, 5ꞌ-GAGTCTGCTGGATGGCTT-3' and downstream, 5ꞌ-TCTTCCGCTTTCATCTG T-3'), amplified fragment length, 141 bp; GAPDH primer (upstream, 5'-GCACCGTCAAGGC TGAGAAC-3' and downstream, 5'-ATGGTGGTGAAGACGCCAGT-3'), amplified fragment length, 142 bp. Detection of α-tubulin, RhoA, and ROCK protein expressions by Western blot analysis The bicinchoninic acid assay was used for protein quantification. After 10% sodium dodecyl sulfate polyacrylamide gel electrophoresis was performed (stacking gel voltage, 80 V; separating gel voltage, 120 V), the gel was transferred to nitrocellulose membranes at a constant current of 280 ma for 50 min. Rabbit anti-rat antibody (1:400) and alkaline phosphatase-labeled goat anti-rabbit secondary antibody (1:2000) were used for coloration. The gray value of each gel band was recorded and analyzed quantitatively using the FluorChem V2.0 software (USA). The protein content was calculated as follows: gray value of protein/gray value of β-actin in the same sample. RESULTS Morphological observations of ASMCs ASMCs were observed under an inverted microscope. Before adherence, ASMCs were found to be round or oval, and opaque with high-density cytoplasm. After 1 day of culture, most cells adhered to the wall and began to stretch. After 3 days of culture, the adherent cells were fully stretched, presenting fusiform or polygon shapes. There were 1-2 nuclei in the center of cells, with one or more foot processes. There was a fascicular arrangement of cells in one region, which displayed a peak-like morphological feature. After 7-9 days of culture, the bottom of the bottle was completely covered by cells. The time to complete covering of cells on the bottle bottom was inversely proportional to cell viability and seeding density; time decreased with increasing cell viability and inoculation density (Figure 1). Figure 1. Morphological observation of airway smooth muscle cells (100X).
5 Rat models of airway remodeling in asthma 563 Identification of ASMCs by immunofluorescent staining α-sma immunofluorescent staining was used for identification of ASMCs. Under a fluorescence microscope with a wavelength of 488 nm, FITC-labeled α-sma appeared green, and they were uniformly distributed in the cytoplasm for the most part. DAPI-labeled nuclei exhibited blue fluorescence at a wavelength of 356 nm (Figure 2). Figure 2. Identification of airway smooth muscle cells by α-sma immunofluorescent staining (400X). Expression of F-actin in ASMCs Phalloidin could specifically bind intracellular F-actin and exhibited green fluorescence. Under the fluorescence microscope, few short and fine stress fibers were observed in ASMCs in the control group. Cells appeared stretched in asthmatic groups, and showed a relatively higher number of stress fibers. The average gray values of F-actin in ASMCs of asthmatic groups were all higher than those of the control group. The average gray value of F-actin in ASMCs of the intervention group was significantly lower than that of the 8-week asthmatic group (Table 1; Figure 3). Table 1. Comparisons of F-actin gray value and α-tubulin protein among the different groups (means ± SD). Group F-actin gray value α-tubulin protein Control group ± ± week asthmatic group ± 10.86* ± week asthmatic group ± 14.54** ± * 6-week asthmatic group ± 16.81** ± * 8-week asthmatic group ± 25.58** ± ** Intervention group ± ± *P < 0.05 compared to control group; **P < 0.01 compared to control group; P < 0.05 compared to asthmatic groups; P < 0.01 compared to 8-week asthmatic group. Distribution and morphology of α-tubulin in ASMCs α-tubulin exhibited green fluorescence after staining. The microtubule structure in the cytoplasm showed a high-density mesh-like and radiation-like distribution from the nuclear
6 B. Wei et al. 564 center to the periphery. The fluorescence signal intensity and density were significantly higher in all asthmatic groups relative to the control group. The fluorescence signal was significantly weakened in the intervention group, which showed a more pronounced mesh structure and more fragments (Figure 4). A D B E C F Figure 3. Immunofluorescence staining of F-actin in airway smooth muscle cells. A. Control group; B. 2-week asthmatic group; C. 4-week asthmatic group; D. 6-week asthmatic group; E. 8-week asthmatic group; F. intervention group.
7 Rat models of airway remodeling in asthma 565 A D B E C F Figure 4. Immunofluorescence staining of α-tubulin in airway smooth muscle cells. A. Control group; B. 2-week asthmatic group; C. 4-week asthmatic group; D. 6-week asthmatic group; E. 8-week asthmatic group; F. intervention group.
8 B. Wei et al. 566 Expression of the α-tubulin protein in ASMCs Expressions of the α-tubulin protein in ASMCs were significantly higher in all asthmatic groups than in the control group, and this difference increased with the extension of excitation time. The difference was most apparent in the 8-week asthmatic group. The expression level of α-tubulin was significantly reduced in the intervention group (Table 1; Figure 5). Figure 5. Expression of α-tubulin protein in airway smooth muscle cells. Lane 1 = control group; lane 2 = 2-week asthmatic group; lane 3 = 4-week asthmatic group; lane 4 = 6-week asthmatic group; lane 5 = 8-week asthmatic group; lane 6 = intervention group. Expressions of RhoA and ROCK mrna and proteins in ASMCs Expressions of RhoA and ROCK mrna and proteins in ASMCs were significantly higher in all asthmatic groups relative to those of the control group, and the difference increased with the extension of excitation time. Once again, this result was more significant in the 8-week asthmatic group (Table 2; Figure 6). Table 2. Expressions of RhoA and ROCK mrna and protein in airway smooth muscle cells (means ± SD). Group RhoA mrna ROCK mrna RhoA protein ROCK protein Control group ± ± ± ± week asthmatic group ± * ± * ± ± * 4-week asthmatic group ± * ± * ± ± * 6-week asthmatic group ± ** ± ** ± * ± week asthmatic group ± ** ± ** ± ** ± ** *P < 0.05 compared to control group; **P < 0.01 compared to control group. Figure 6. Expressions of RhoA and ROCK mrna and protein in airway smooth muscle cells. Lane 1 = control group; lane 2 = 2-week asthmatic group; lane 3 = 4-week asthmatic group; lane 4 = 6-week asthmatic group; lane 5 = 8-week asthmatic group.
9 Rat models of airway remodeling in asthma 567 DISCUSSION The cytoskeleton is a type of fibrous protein matrix of eukaryotic cells, and presents mesh, fascicular, and banding shapes. It is composed of microfilaments, microtubules, and intermediate filaments, which are highly coordinated with the cell nucleus, plasma membrane, and cell organs, and constitute the cellular skeleton and motion coordination system. The cytoskeleton can maintain the cell morphology and exercise motor functions, and plays an important role in signal transmission (Paavilainen et al., 2004). Microfilaments are polar fine fibers with 7 mm in diameters, which are mainly composed of actins, and they determine the intracellular spiral-shape. Microfilaments are the central structure forming stress fibers, muscle fibers, and the crypt-villus axis of the small intestine. Actin is the most abundant protein in most eukaryotic cells, and has been highly conserved in evolution, showing little variation among species. The actin family includes G- actin and F-actin, and the cytoskeleton is mainly composed of F-actin. Actin cytoskeletons are related to many cellular functions. They participate in muscle formation and cell movement. In addition, actin cytoskeletons can control transmission of mrna-related information and transportation of intracellular vesicles and receptors. In local cellular membranes, actin cytoskeletons aggregate to form fascicular stress fibers. One end of the stress fiber accepts the stimulatory signal of the extracellular matrix, while the other end is connected to cytoplasmic organelles to control adhesion and signal transmission between the cell and the extracellular matrix. At the same time, the functions of microfilaments are regulated by a signal transduction system. Actin is generally considered to be the first target protein for the initiation of extracellular signals (e.g., cytokines) and in the regulation of intracellular signals (Plastino and Sykes, 2005; Gouëffic et al., 2006; Nincheri et al., 2009; Abounit and Zurzolo, 2012; Martin et al., 2012; Karvonen et al., 2012). FITC-labeled phalloidin can specifically bind F-actin, but not G-actin, enabling observation of structural and distributional changes of F-actin. Results of the present study revealed that in the control group, a small amount of short and fine stress fibers, without filamentary pseudopod formations, were observed under a fluorescence microscope. In contrast, in asthmatic groups, cells were visibly stretched, with increased stress fibers and obvious pseudopods. This indicates actin cytoskeleton reconstruction in ASMCs in rats with airway remodeling in asthma. The microtubule is a hollow cylindrical tubular fiber with a diameter of 25 nm and is mainly composed of α- and β-tubulin (α- and β-dimer). Furthermore, it is a GTP-binding protein. Microtubules have obvious amino acid sequence homology with G proteins of the signal transduction system, and have similar functional characteristics to G proteins (Pao et al., 2009; Lee et al., 2013). They are involved in cell movement and cytoplasmic transportation, as well as chromosome movement and mitosis (Sahab et al., 2011; Sharp et al., 2011; Hinrichs et al., 2012). Therefore, microtubules are associated with cell proliferation. However, investigations of the mechanism of airway remodeling in asthma in view of the microtubule cytoskeleton are rare. In this study, ASMCs in rats with airway remodeling in asthma were cultured, and immunofluorescence staining and Western blot analysis were used to detect the expression of α-tubulin. Results revealed that α-tubulin expression levels were significantly higher in asthmatic groups than in the control group. This suggests that changes of the microtubule cytoskeleton besides microfilament reconstruction persist in rats with airway remodeling in asthma. RhoA is an important molecule for mediating cell signal transduction. Similar to G proteins, GTP enzymes of RhoA can shift between GTP binding states (activation) and GDP
10 B. Wei et al. 568 binding states (inactivated). It triggers the downstream kinase cascade reaction and exerts several biological effects (Ding et al., 2011). RhoA is also the primary pathway mediating stress fiber formation. It combines with downstream target molecules such as ROCK to directly modulate contractile proteins to participate in actin cytoskeleton reconstruction. In addition, it can change the activity of serum response factors and regulate gene expressions of different contractile proteins for long-term adjustments of the actin cytoskeleton. RhoA is also known as a regulator of heterogeneity of the actin cytoskeleton and cellular morphology (Thapar et al., 2002; dos Remedios et al., 2003; Mohseni and Chishti, 2009). Recent studies have demonstrated that the RhoA/ROCK signaling pathway is involved in many pathophysiological processes of asthma, including formation of asthmatic airway hyper-responsiveness, chemotactic aggregation of eosinophil to the airway, and airway remodeling (Bamburg and Wiggan, 2002; Takeda et al., 2006; Kume, 2008; Li et al., 2008; Goto et al., 2010). In the present study, qpcr and Western blot methods were used to detect mrna and protein expressions of RhoA and ROCK in rats with airway remodeling in asthma. Results showed that mrna and protein expressions of RhoA and ROCK were significantly upregulated in all asthmatic groups relative to the control group. After treatment with a ROCK-specific inhibitor, the expression levels of F-actin and α-tubulin decreased, suggesting that the RhoA/ ROCK signal may induce changes in the microfilament and microtubule cytoskeleton. CONCLUSIONS The cytoskeleton and RhoA/ROCK may signal transduction pathways in which external stimulations induce biological functions of ASMCs. They can be present at both the front and back ends of information transmission chains, or exist independently. These pathways are connected, and they cooperate and restrict one another, thereby forming a crisscross information transmission network. Whether other pathways are also involved requires further study. In-depth investigations of such mechanisms can broaden the prospects for studying the pathogenesis of airway remodeling as well as for development of novel strategies for its clinical prevention and treatment. REFERENCES Abounit S and Zurzolo C (2012). Wiring through tunneling nanotubes-from electrical signals to organelle transfer. J. Cell Sci. 125: Bamburg JR and Wiggan OP (2002). ADF/cofilin and actin dynamics in disease. Trends Cell Biol. 12: Ding F, Yin Z and Wang HR (2011). Ubiquitination in Rho signaling. Curr. Top. Med. Chem. 11: dos Remedios CG, Chhabra D, Kekic M, Dedova IV, et al. (2003). Actin binding proteins: regulation of cytoskeletal microfilaments. Physiol. Rev. 83: Fukata Y, Amano M and Kaibuchi K (2001). Rho-Rho-kinase pathway in smooth muscle contraction and cytoskeletal reorganization of non-muscle cells. Trends Pharmacol. Sci. 22: Goto K, Chiba Y, Sakai H and Misawa M (2010). Mechanism of inhibitory effect of prednisolone on RhoA upregulation in human bronchial smooth muscle cells. Biol. Pharm. Bull. 33: Gouëffic Y, Guilluy C, Guérin P, Patra P, et al. (2006). Hyaluronan induces vascular smooth muscle cell migration through RHAMM-mediated PI3K-dependent Rac activation. Cardiovasc. Res. 72: Hinrichs MH, Jalal A, Brenner B, Mandelkow E, et al. (2012). Tau protein diffuses along the microtubule lattice. J. Biol. Chem. 287: Karvonen HM, Lehtonen ST, Sormunen RT, Harju TH, et al. (2012). Myofibroblasts in interstitial lung diseases show diverse electron microscopic and invasive features. Lab. Invest. 92:
11 Rat models of airway remodeling in asthma 569 Kume H (2008). RhoA/Rho-kinase as a therapeutic target in asthma. Curr. Med. Chem. 15: Lee MN, Koh A, Park D, Jang JH, et al. (2013). Deacetylated αβ-tubulin acts as a positive regulator of Rheb GTPase through increasing its GTP-loading. Cell Signal. 25: Li ZX, Luo YL, Xu J, Ren DQ, et al. (2008). Rho-kinase signaling pathway participates in endothelin-1-induced human airway smooth muscle cell migration and cytoskeletal reorganization. Nan. Fang Yi Ke. Da Xue Xue Bao 28: Martin E, Dahan D, Cardouat G, Gillibert-Duplantier J, et al. (2012). Involvement of TRPV1 and TRPV4 channels in migration of rat pulmonary arterial smooth muscle cells. Pflugers Arch. 464: Mohseni M and Chishti AH (2009). Regulatory models of RhoA suppression by dematin, a cytoskeletal adaptor protein. Cell Adh. Migr. 3: Nincheri P, Luciani P, Squecco R, Donati C, et al. (2009). Sphingosine 1-phosphate induces differentiation of adipose tissue-derived mesenchymal stem cells towards smooth muscle cells. Cell. Mol. Life Sci. 66: Paavilainen VO, Bertling E, Falck S and Lappalainen P (2004). Regulation of cytoskeletal dynamics by actin-monomerbinding proteins. Trends Cell Biol. 14: Pao CS, Barker BL and Benovic JL (2009). Role of the amino terminus of G protein-coupled receptor kinase 2 in receptor phosphorylation. Biochemistry 48: Plastino J and Sykes C (2005). The actin slingshot. Curr. Opin. Cell Biol. 17: Sahab ZJ, Hall MD, Me SY, Dakshanamurthy S, et al. (2011). Tumor suppressor RARRES1 interacts with cytoplasmic carboxypeptidase AGBL2 to regulate the α-tubulin tyrosination cycle. Cancer Res. 71: Sharp JA, Plant JJ, Ohsumi TK, Borowsky M, et al. (2011). Functional analysis of the microtubule-interacting transcriptome. Mol. Biol. Cell 22: Stamenović D (2008). Cytoskeletal mechanics in airway smooth muscle cells. Respir. Physiol. Neurobiol. 163: Takeda N, Kondo M, Ito S, Ito Y, et al. (2006). Role of RhoA inactivation in reduced cell proliferation of human airway smooth muscle by simvastatin. Am. J. Respir. Cell Mol. Biol. 35: Thapar R, Karnoub AE and Campbell SL (2002). Structural and biophysical insights into the role of the insert region in Rac1 function. Biochemistry 41:
SUPPLEMENTAL MATERIAL. Supplementary Methods
 SUPPLEMENTAL MATERIAL Supplementary Methods Culture of cardiomyocytes, fibroblasts and cardiac microvascular endothelial cells The isolation and culturing of neonatal rat ventricular cardiomyocytes was
SUPPLEMENTAL MATERIAL Supplementary Methods Culture of cardiomyocytes, fibroblasts and cardiac microvascular endothelial cells The isolation and culturing of neonatal rat ventricular cardiomyocytes was
Structure and Function of Cells
 Structure and Function of Cells Learning Outcomes Explain the cell theory Explain why cell size is usually very small Describe the Fluid Mosaic Model of membranes Describe similarities and differences
Structure and Function of Cells Learning Outcomes Explain the cell theory Explain why cell size is usually very small Describe the Fluid Mosaic Model of membranes Describe similarities and differences
Name: Per/row: Cell Structure and Function Practice: Use Ch 4 in Mader Biology
 Cell Structure and Function Practice: Use Ch 4 in Mader Biology Name: Per/row: 1. Write the name of the cell part in the box next to its description/function. Cell membrane Centrioles Chloroplast Chromatin
Cell Structure and Function Practice: Use Ch 4 in Mader Biology Name: Per/row: 1. Write the name of the cell part in the box next to its description/function. Cell membrane Centrioles Chloroplast Chromatin
5/12/2015. Cell Size. Relative Rate of Reaction
 Cell Makeup Chapter 4 The Cell: The Fundamental Unit of Life We previously talked about the cell membrane The cytoplasm is everything inside the membrane, except the nucleus Includes Cytosol = liquid portion
Cell Makeup Chapter 4 The Cell: The Fundamental Unit of Life We previously talked about the cell membrane The cytoplasm is everything inside the membrane, except the nucleus Includes Cytosol = liquid portion
Top 10 Contributions on Biomedical Sciences: 2nd Edition. St Vincent s Hospital, The University of Melbourne, Australia 2
 Chapter 06 Cancer Progression: The Impact of Cytoskeletal Molecules Sam L Francis 1 and Juliana Antonipillai 2 * 1 St Vincent s Hospital, The University of Melbourne, Australia 2 School of Health and Biomedical
Chapter 06 Cancer Progression: The Impact of Cytoskeletal Molecules Sam L Francis 1 and Juliana Antonipillai 2 * 1 St Vincent s Hospital, The University of Melbourne, Australia 2 School of Health and Biomedical
Structures in Cells. Cytoplasm. Lecture 5, EH1008: Biology for Public Health, Biomolecules
 Structures in Cells Lecture 5, EH1008: Biology for Public Health, Biomolecules Limian.zheng@ucc.ie 1 Cytoplasm Nucleus Centrioles Cytoskeleton Cilia Microvilli 2 Cytoplasm Cellular material outside nucleus
Structures in Cells Lecture 5, EH1008: Biology for Public Health, Biomolecules Limian.zheng@ucc.ie 1 Cytoplasm Nucleus Centrioles Cytoskeleton Cilia Microvilli 2 Cytoplasm Cellular material outside nucleus
Human height. Length of some nerve and muscle cells. Chicken egg. Frog egg. Most plant and animal cells Nucleus Most bacteria Mitochondrion
 10 m 1 m 0.1 m 1 cm Human height Length of some nerve and muscle cells Chicken egg Unaided eye 1 mm Frog egg 100 µm 10 µm 1 µm 100 nm 10 nm Most plant and animal cells Nucleus Most bacteria Mitochondrion
10 m 1 m 0.1 m 1 cm Human height Length of some nerve and muscle cells Chicken egg Unaided eye 1 mm Frog egg 100 µm 10 µm 1 µm 100 nm 10 nm Most plant and animal cells Nucleus Most bacteria Mitochondrion
Molecular Cell Biology - Problem Drill 20: Cytoskeleton and Cellular Mobility
 Molecular Cell Biology - Problem Drill 20: Cytoskeleton and Cellular Mobility Question No. 1 of 10 1. Which of the following statements about cytoskeletal filaments is correct? Question #1 (A) The Cytoskeleton
Molecular Cell Biology - Problem Drill 20: Cytoskeleton and Cellular Mobility Question No. 1 of 10 1. Which of the following statements about cytoskeletal filaments is correct? Question #1 (A) The Cytoskeleton
Structures in Cells. Lecture 5, EH1008: Biology for Public Health, Biomolecules.
 Structures in Cells Lecture 5, EH1008: Biology for Public Health, Biomolecules Limian.zheng@ucc.ie 1 Cytoplasm Nucleus Centrioles Cytoskeleton Cilia Microvilli 2 Cytoplasm Cellular material outside nucleus
Structures in Cells Lecture 5, EH1008: Biology for Public Health, Biomolecules Limian.zheng@ucc.ie 1 Cytoplasm Nucleus Centrioles Cytoskeleton Cilia Microvilli 2 Cytoplasm Cellular material outside nucleus
CELL PARTS TYPICAL ANIMAL CELL
 AP BIOLOGY CText Reference, Campbell v.8, Chapter 6 ACTIVITY1.12 NAME DATE HOUR CELL PARTS TYPICAL ANIMAL CELL ENDOMEMBRANE SYSTEM TYPICAL PLANT CELL QUESTIONS: 1. Write the name of the cell part in the
AP BIOLOGY CText Reference, Campbell v.8, Chapter 6 ACTIVITY1.12 NAME DATE HOUR CELL PARTS TYPICAL ANIMAL CELL ENDOMEMBRANE SYSTEM TYPICAL PLANT CELL QUESTIONS: 1. Write the name of the cell part in the
Cellular Physiology (PHSI3009) Contents:
 Cellular Physiology (PHSI3009) Contents: Cell membranes and communication 2 nd messenger systems G-coupled protein signalling Calcium signalling Small G-protein signalling o RAS o MAPK o PI3K RHO GTPases
Cellular Physiology (PHSI3009) Contents: Cell membranes and communication 2 nd messenger systems G-coupled protein signalling Calcium signalling Small G-protein signalling o RAS o MAPK o PI3K RHO GTPases
Impact factor: Reporter:4A1H0019 Chen Zi Hao 4A1H0023 Huang Wan ting 4A1H0039 Sue Yi Zhu 4A1H0070 Lin Guan cheng 4A1H0077 Chen Bo xuan
 Curcumin Protects Neonatal Rat Cardiomyocytes against High Glucose-Induced Apoptosis via PI3K/Akt Signalling Pathway Wei Yu,1,2 Wenliang Zha,1 Zhiqiang Ke,1 Qing Min,2 Cairong Li,1 Huirong Sun,3 and Chao
Curcumin Protects Neonatal Rat Cardiomyocytes against High Glucose-Induced Apoptosis via PI3K/Akt Signalling Pathway Wei Yu,1,2 Wenliang Zha,1 Zhiqiang Ke,1 Qing Min,2 Cairong Li,1 Huirong Sun,3 and Chao
Cells. Variation and Function of Cells
 Cells Variation and Function of Cells Cell Theory states that: 1. All living things are made of cells 2. Cells are the basic unit of structure and function in living things 3. New cells are produced from
Cells Variation and Function of Cells Cell Theory states that: 1. All living things are made of cells 2. Cells are the basic unit of structure and function in living things 3. New cells are produced from
Neuronal plasma membrane
 ORGANELLES ORGANELLES Neuronal plasma membrane The neuronal plasma membrane contains several local domains with unique properties Presynaptic terminal Endoplasmic Reticulum In neurons the Nissl bodies
ORGANELLES ORGANELLES Neuronal plasma membrane The neuronal plasma membrane contains several local domains with unique properties Presynaptic terminal Endoplasmic Reticulum In neurons the Nissl bodies
G-Protein Signaling. Introduction to intracellular signaling. Dr. SARRAY Sameh, Ph.D
 G-Protein Signaling Introduction to intracellular signaling Dr. SARRAY Sameh, Ph.D Cell signaling Cells communicate via extracellular signaling molecules (Hormones, growth factors and neurotransmitters
G-Protein Signaling Introduction to intracellular signaling Dr. SARRAY Sameh, Ph.D Cell signaling Cells communicate via extracellular signaling molecules (Hormones, growth factors and neurotransmitters
Cell Cycle, Mitosis, and Microtubules. LS1A Final Exam Review Friday 1/12/07. Processes occurring during cell cycle
 Cell Cycle, Mitosis, and Microtubules LS1A Final Exam Review Friday 1/12/07 Processes occurring during cell cycle Replicate chromosomes Segregate chromosomes Cell divides Cell grows Cell Growth 1 The standard
Cell Cycle, Mitosis, and Microtubules LS1A Final Exam Review Friday 1/12/07 Processes occurring during cell cycle Replicate chromosomes Segregate chromosomes Cell divides Cell grows Cell Growth 1 The standard
Cell Biology. A discipline of biology: 1. Cell structure 2. Cellular processes 3. Cell division
 The Cell Cell Biology 1 A discipline of biology: 1. Cell structure 2. Cellular processes 3. Cell division Tight connection with 1. Molecular biology 2. Biochemistry Cell theory 2 1838, 1839 Theodor Schwann
The Cell Cell Biology 1 A discipline of biology: 1. Cell structure 2. Cellular processes 3. Cell division Tight connection with 1. Molecular biology 2. Biochemistry Cell theory 2 1838, 1839 Theodor Schwann
Muscle tissues. Dr. Hersh Abdul Ham-Karim BVM&S, PG Dip, MSc and PhD
 Muscle tissues Dr. Hersh Abdul Ham-Karim BVM&S, PG Dip, MSc and PhD Muscle tissue is a soft tissue that composes muscles in animal bodies, and gives rise to muscles' ability to contract. Muscle tissue
Muscle tissues Dr. Hersh Abdul Ham-Karim BVM&S, PG Dip, MSc and PhD Muscle tissue is a soft tissue that composes muscles in animal bodies, and gives rise to muscles' ability to contract. Muscle tissue
Organelles in EUKARYOTIC CELLS. Pages 4 5: cell membrane, cytoplasm/cytosol, cytoskeleton, nucleus, chromatin/chromosomes, and centrioles
 Organelles in EUKARYOTIC CELLS Pages 4 5: cell membrane, cytoplasm/cytosol, cytoskeleton, nucleus, chromatin/chromosomes, and centrioles Use the info on the following slides to complete pages 4 5 in your
Organelles in EUKARYOTIC CELLS Pages 4 5: cell membrane, cytoplasm/cytosol, cytoskeleton, nucleus, chromatin/chromosomes, and centrioles Use the info on the following slides to complete pages 4 5 in your
Comparison of Young and Old Cardiac Telocytes Using Atomic Force Microscopy
 Comparison of Young and Old Cardiac Telocytes Using Atomic Force Microscopy Jiali Luo 1, 2, 3, 4, a, Shanshan Feng 1, 2, 3, 4, b 1Key Laboratory of Regenerative Medicine, Ministry of Education, Jinan University,
Comparison of Young and Old Cardiac Telocytes Using Atomic Force Microscopy Jiali Luo 1, 2, 3, 4, a, Shanshan Feng 1, 2, 3, 4, b 1Key Laboratory of Regenerative Medicine, Ministry of Education, Jinan University,
CELLS. Cells. Basic unit of life (except virus)
 Basic unit of life (except virus) CELLS Prokaryotic, w/o nucleus, bacteria Eukaryotic, w/ nucleus Various cell types specialized for particular function. Differentiation. Over 200 human cell types 56%
Basic unit of life (except virus) CELLS Prokaryotic, w/o nucleus, bacteria Eukaryotic, w/ nucleus Various cell types specialized for particular function. Differentiation. Over 200 human cell types 56%
A Tour of the Cell Chapter 4. Outline. Early contributors to Understanding Cells. Cell Theory. Cell Size s Matt Schleiden & Ted Schann
 A Tour of the Cell Chapter 4 Outline History of the science behind cells Cell theory & its importance Why are cells small? Microscopes Cell structure and function Prokaryotic cells Eukaryotic cells Early
A Tour of the Cell Chapter 4 Outline History of the science behind cells Cell theory & its importance Why are cells small? Microscopes Cell structure and function Prokaryotic cells Eukaryotic cells Early
AP Biology Cells: Chapters 4 & 5
 AP Biology Cells: Chapters 4 & 5 Multiple Choice Identify the choice that best completes the statement or answers the question. 1. The was the first unifying principle of biology. a. spontaneous generation
AP Biology Cells: Chapters 4 & 5 Multiple Choice Identify the choice that best completes the statement or answers the question. 1. The was the first unifying principle of biology. a. spontaneous generation
Reading Packet 2- Cells Unit. Chapter 6: A Tour of the Cell 1. What is resolving power?
 AP Biology Reading Packet 2- Cells Unit Name Chapter 6: A Tour of the Cell 1. What is resolving power? 2. How is an electron microscope different from a light microscope and what is the difference between
AP Biology Reading Packet 2- Cells Unit Name Chapter 6: A Tour of the Cell 1. What is resolving power? 2. How is an electron microscope different from a light microscope and what is the difference between
3.1 Activation of human peripheral blood T lymphocytes induces reversible association of cofilin with the actin cytoskeleton
 3. RESULTS 3.1 Activation of human peripheral blood T lymphocytes induces reversible association of cofilin with the actin cytoskeleton In unstimulated resting human peripheral blood T lymphocytes (PBTs)
3. RESULTS 3.1 Activation of human peripheral blood T lymphocytes induces reversible association of cofilin with the actin cytoskeleton In unstimulated resting human peripheral blood T lymphocytes (PBTs)
Cell Communication. Chapter 11. Biology Eighth Edition Neil Campbell and Jane Reece. PowerPoint Lecture Presentations for
 Chapter 11 Cell Communication PowerPoint Lecture Presentations for Biology Eighth Edition Neil Campbell and Jane Reece Lectures by Chris Romero, updated by Erin Barley with contributions from Joan Sharp
Chapter 11 Cell Communication PowerPoint Lecture Presentations for Biology Eighth Edition Neil Campbell and Jane Reece Lectures by Chris Romero, updated by Erin Barley with contributions from Joan Sharp
SUPPLEMENTARY INFORMATION
 DOI: 10.1038/ncb3021 Supplementary figure 1 Characterisation of TIMPless fibroblasts. a) Relative gene expression of TIMPs1-4 by real time quantitative PCR (RT-qPCR) in WT or ΔTimp fibroblasts (mean ±
DOI: 10.1038/ncb3021 Supplementary figure 1 Characterisation of TIMPless fibroblasts. a) Relative gene expression of TIMPs1-4 by real time quantitative PCR (RT-qPCR) in WT or ΔTimp fibroblasts (mean ±
PHSI3009 Frontiers in Cellular Physiology 2017
 Overview of PHSI3009 L2 Cell membrane and Principles of cell communication L3 Signalling via G protein-coupled receptor L4 Calcium Signalling L5 Signalling via Growth Factors L6 Signalling via small G-protein
Overview of PHSI3009 L2 Cell membrane and Principles of cell communication L3 Signalling via G protein-coupled receptor L4 Calcium Signalling L5 Signalling via Growth Factors L6 Signalling via small G-protein
(a) TEM of a plasma. Fimbriae. Nucleoid. Ribosomes. Plasma membrane. Cell wall Capsule. Bacterial chromosome
 0 m m 0. m cm mm 00 µm 0 µm 00 nm 0 nm Human height Length of some nerve and muscle cells Chicken egg Frog egg Most plant and animal cells Most bacteria Smallest bacteria Viruses Proteins Unaided eye Light
0 m m 0. m cm mm 00 µm 0 µm 00 nm 0 nm Human height Length of some nerve and muscle cells Chicken egg Frog egg Most plant and animal cells Most bacteria Smallest bacteria Viruses Proteins Unaided eye Light
BL 424 Test pts name Multiple choice have one choice each and are worth 3 points.
 BL 424 Test 3 2010 150 pts name Multiple choice have one choice each and are worth 3 points. 1. The plasma membrane functions as a a. selective barrier to the passage of molecules. b. sensor through which
BL 424 Test 3 2010 150 pts name Multiple choice have one choice each and are worth 3 points. 1. The plasma membrane functions as a a. selective barrier to the passage of molecules. b. sensor through which
PCB 3023 Exam 4 - Form A First and Last Name
 PCB 3023 Exam 4 - Form A First and Last Name Student ID # (U Number) A Before beginning this exam, please complete the following instructions: 1) Write your name and U number on the first page of this
PCB 3023 Exam 4 - Form A First and Last Name Student ID # (U Number) A Before beginning this exam, please complete the following instructions: 1) Write your name and U number on the first page of this
Construction of a hepatocellular carcinoma cell line that stably expresses stathmin with a Ser25 phosphorylation site mutation
 Construction of a hepatocellular carcinoma cell line that stably expresses stathmin with a Ser25 phosphorylation site mutation J. Du 1, Z.H. Tao 2, J. Li 2, Y.K. Liu 3 and L. Gan 2 1 Department of Chemistry,
Construction of a hepatocellular carcinoma cell line that stably expresses stathmin with a Ser25 phosphorylation site mutation J. Du 1, Z.H. Tao 2, J. Li 2, Y.K. Liu 3 and L. Gan 2 1 Department of Chemistry,
Cell Category? Prokaryote
 CELLS Cell Category? Prokaryote Prokaryote Eukaryote Cell Category? Cell Type? Cell Category? Cell Type? Endosymbiosis eukaryotic cells were formed from simpler prokaryotes Endo within Symbiosis together
CELLS Cell Category? Prokaryote Prokaryote Eukaryote Cell Category? Cell Type? Cell Category? Cell Type? Endosymbiosis eukaryotic cells were formed from simpler prokaryotes Endo within Symbiosis together
(a) Significant biological processes (upper panel) and disease biomarkers (lower panel)
 Supplementary Figure 1. Functional enrichment analyses of secretomic proteins. (a) Significant biological processes (upper panel) and disease biomarkers (lower panel) 2 involved by hrab37-mediated secretory
Supplementary Figure 1. Functional enrichment analyses of secretomic proteins. (a) Significant biological processes (upper panel) and disease biomarkers (lower panel) 2 involved by hrab37-mediated secretory
Proteomic profiling of small-molecule inhibitors reveals dispensability of MTH1 for cancer cell survival
 Supplementary Information for Proteomic profiling of small-molecule inhibitors reveals dispensability of MTH1 for cancer cell survival Tatsuro Kawamura 1, Makoto Kawatani 1, Makoto Muroi, Yasumitsu Kondoh,
Supplementary Information for Proteomic profiling of small-molecule inhibitors reveals dispensability of MTH1 for cancer cell survival Tatsuro Kawamura 1, Makoto Kawatani 1, Makoto Muroi, Yasumitsu Kondoh,
Chapter 6: A Tour of the Cell. 1. Studying Cells 2. Intracellular Structures 3. The Cytoskeleton 4. Extracellular Structures
 Chapter 6: A Tour of the Cell 1. Studying Cells 2. Intracellular Structures 3. The Cytoskeleton 4. Extracellular Structures 1. Studying Cells Concepts of Microscopy MAGNIFICATION factor by which the image
Chapter 6: A Tour of the Cell 1. Studying Cells 2. Intracellular Structures 3. The Cytoskeleton 4. Extracellular Structures 1. Studying Cells Concepts of Microscopy MAGNIFICATION factor by which the image
1. Studying Cells. Concepts of Microscopy 11/7/2016. Chapter 6: A Tour of the Cell
 Electron microscope Light microscope Unaided eye 11/7/2016 Chapter 6: A Tour of the Cell 1. Studying Cells 2. Intracellular Structures 3. The Cytoskeleton 4. Extracellular Structures 1. Studying Cells
Electron microscope Light microscope Unaided eye 11/7/2016 Chapter 6: A Tour of the Cell 1. Studying Cells 2. Intracellular Structures 3. The Cytoskeleton 4. Extracellular Structures 1. Studying Cells
Lecture 1, Fall 2014 The structure of the eukaryotic cell, as it relates to cells chemical and biological functions.
 Lecture 1, Fall 2014 The structure of the eukaryotic cell, as it relates to cells chemical and biological functions. There are several important themes that transcends just the chemistry and bring the
Lecture 1, Fall 2014 The structure of the eukaryotic cell, as it relates to cells chemical and biological functions. There are several important themes that transcends just the chemistry and bring the
(d) are made mainly of lipids and of proteins that lie like thin sheets on the membrane surface
 Which of the following statements is no true? Biological membranes (a) are composed partly of amphipathic lipids (b) have hydrophobic and hydrophilic regions (c) are typically in a fluid state (d) are
Which of the following statements is no true? Biological membranes (a) are composed partly of amphipathic lipids (b) have hydrophobic and hydrophilic regions (c) are typically in a fluid state (d) are
CMB621: Cytoskeleton. Also known as How the cell plays with LEGOs to ensure order, not chaos, is temporally and spatially achieved
 CMB621: Cytoskeleton Also known as How the cell plays with LEGOs to ensure order, not chaos, is temporally and spatially achieved Lecture(s) Overview Lecture 1: What is the cytoskeleton? Membrane interaction
CMB621: Cytoskeleton Also known as How the cell plays with LEGOs to ensure order, not chaos, is temporally and spatially achieved Lecture(s) Overview Lecture 1: What is the cytoskeleton? Membrane interaction
Zool 3200: Cell Biology Exam 4 Part II 2/3/15
 Name:Key Trask Zool 3200: Cell Biology Exam 4 Part II 2/3/15 Answer each of the following questions in the space provided, explaining your answers when asked to do so; circle the correct answer or answers
Name:Key Trask Zool 3200: Cell Biology Exam 4 Part II 2/3/15 Answer each of the following questions in the space provided, explaining your answers when asked to do so; circle the correct answer or answers
Lecture 15. Signal Transduction Pathways - Introduction
 Lecture 15 Signal Transduction Pathways - Introduction So far.. Regulation of mrna synthesis Regulation of rrna synthesis Regulation of trna & 5S rrna synthesis Regulation of gene expression by signals
Lecture 15 Signal Transduction Pathways - Introduction So far.. Regulation of mrna synthesis Regulation of rrna synthesis Regulation of trna & 5S rrna synthesis Regulation of gene expression by signals
Neuronal plasma membrane
 ORGANELLES ORGANELLES Neuronal plasma membrane The neuronal plasma membrane contains several local domains with unique properties Presynaptic terminal Endoplasmic Reticulum In neurons the Nissl bodies
ORGANELLES ORGANELLES Neuronal plasma membrane The neuronal plasma membrane contains several local domains with unique properties Presynaptic terminal Endoplasmic Reticulum In neurons the Nissl bodies
HOMEWORK RUBRICS MECHANOTRANSDUCTION UNIT: HOMEWORK #1 (20 pts towards your grade)
 HOMEWORK RUBRICS MECHANOTRANSDUCTION UNIT: HOMEWORK #1 (20 pts towards your grade) 1. Mesenchymal stem cells (MSC) cultured on extracellular matrices with different stiffness exhibit diverse lineage commitment
HOMEWORK RUBRICS MECHANOTRANSDUCTION UNIT: HOMEWORK #1 (20 pts towards your grade) 1. Mesenchymal stem cells (MSC) cultured on extracellular matrices with different stiffness exhibit diverse lineage commitment
Topics Covered. General muscle structure non-muscle components, macro-structure, contractile elements, membrane components.
 1 Topics Covered General muscle structure non-muscle components, macro-structure, contractile elements, membrane components. Contractile function Contractile and regulatory proteins, force production.
1 Topics Covered General muscle structure non-muscle components, macro-structure, contractile elements, membrane components. Contractile function Contractile and regulatory proteins, force production.
Sphingosine-1-phosphate signaling and cardiac fibrosis. Department of Physiology, Kanazawa University School of Medicine, Kanazawa, Japan
 96 Special Issue: Cellular and Molecular Bases for Fibrotic Diseases Review Article Sphingosine-1-phosphate signaling and cardiac fibrosis Noriko Takuwa 1, 2, ), Yasuo Okamoto 1), Kazuaki Yoshioka 1) and
96 Special Issue: Cellular and Molecular Bases for Fibrotic Diseases Review Article Sphingosine-1-phosphate signaling and cardiac fibrosis Noriko Takuwa 1, 2, ), Yasuo Okamoto 1), Kazuaki Yoshioka 1) and
Neurokinin 1 receptor promotes rat airway smooth muscle cell migration in asthmatic airway remodelling by enhancing tubulin expression
 Original Article Neurokinin 1 receptor promotes rat airway smooth muscle cell migration in asthmatic airway remodelling by enhancing tubulin expression Bing Wei 1, Mingwei Sun 2, Yunxiao Shang 3, Chao
Original Article Neurokinin 1 receptor promotes rat airway smooth muscle cell migration in asthmatic airway remodelling by enhancing tubulin expression Bing Wei 1, Mingwei Sun 2, Yunxiao Shang 3, Chao
Recent Studies of Phenylketonuria By: Jennifer Gastelum Dr. Koni Stone Copyright 2014
 Recent Studies of Phenylketonuria By: Jennifer Gastelum Dr. Koni Stone Copyright 2014 Phenylketonuria (PKU) is an autosomal recessive genetic disorder that causes an accumulation of toxic metabolites in
Recent Studies of Phenylketonuria By: Jennifer Gastelum Dr. Koni Stone Copyright 2014 Phenylketonuria (PKU) is an autosomal recessive genetic disorder that causes an accumulation of toxic metabolites in
1 (a) State the maximum magnification that can be achieved by a light microscope and a transmission electron microscope.
 1 (a) State the maximum magnification that can be achieved by a light microscope and a transmission electron microscope. Select your answers from the list below. 10x 40x 100x light microscope... x transmission
1 (a) State the maximum magnification that can be achieved by a light microscope and a transmission electron microscope. Select your answers from the list below. 10x 40x 100x light microscope... x transmission
Propagation of the Signal
 OpenStax-CNX module: m44452 1 Propagation of the Signal OpenStax College This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License 3.0 By the end of this section,
OpenStax-CNX module: m44452 1 Propagation of the Signal OpenStax College This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License 3.0 By the end of this section,
Cell Signaling part 2
 15 Cell Signaling part 2 Functions of Cell Surface Receptors Other cell surface receptors are directly linked to intracellular enzymes. The largest family of these is the receptor protein tyrosine kinases,
15 Cell Signaling part 2 Functions of Cell Surface Receptors Other cell surface receptors are directly linked to intracellular enzymes. The largest family of these is the receptor protein tyrosine kinases,
A Tour of the Cell. Chapter 4. Most cells are microscopic. Cells vary in size and shape
 Chapter 4 A Tour of the Cell Most cells are microscopic Cells vary in size and shape 10 m Human height 1 m Length of some nerve and muscle cells 100 mm (10 cm) 10 mm (1 cm) Chicken egg Unaided eye 1 mm
Chapter 4 A Tour of the Cell Most cells are microscopic Cells vary in size and shape 10 m Human height 1 m Length of some nerve and muscle cells 100 mm (10 cm) 10 mm (1 cm) Chicken egg Unaided eye 1 mm
Cell Cell Communication
 IBS 8102 Cell, Molecular, and Developmental Biology Cell Cell Communication January 29, 2008 Communicate What? Why do cells communicate? To govern or modify each other for the benefit of the organism differentiate
IBS 8102 Cell, Molecular, and Developmental Biology Cell Cell Communication January 29, 2008 Communicate What? Why do cells communicate? To govern or modify each other for the benefit of the organism differentiate
Human Epithelial Cells
 The Cell Human Epithelial Cells Plant Cells Cells have an internal structure Eukaryotic cells are organized Protective membrane around them that communicates with other cells Organelles have specific jobs
The Cell Human Epithelial Cells Plant Cells Cells have an internal structure Eukaryotic cells are organized Protective membrane around them that communicates with other cells Organelles have specific jobs
Cellular compartments
 Cellular compartments 1. Cellular compartments and their function 2. Evolution of cellular compartments 3. How to make a 3D model of cellular compartment 4. Cell organelles in the fluorescent microscope
Cellular compartments 1. Cellular compartments and their function 2. Evolution of cellular compartments 3. How to make a 3D model of cellular compartment 4. Cell organelles in the fluorescent microscope
Supplemental Figure 1
 Supplemental Figure 1 A S100A4: SFLGKRTDEAAFQKLMSNLDSNRDNEVDFQEYCVFLSCIAMMCNEFFEGFPDK Overlap: SF G DE KLM LD N D VDFQEY VFL I M N FF G PD S100A2: SFVGEKVDEEGLKKLMGSLDENSDQQVDFQEYAVFLALITVMCNDFFQGCPDR
Supplemental Figure 1 A S100A4: SFLGKRTDEAAFQKLMSNLDSNRDNEVDFQEYCVFLSCIAMMCNEFFEGFPDK Overlap: SF G DE KLM LD N D VDFQEY VFL I M N FF G PD S100A2: SFVGEKVDEEGLKKLMGSLDENSDQQVDFQEYAVFLALITVMCNDFFQGCPDR
IL-13 Augments Compressive Stress-induced Tissue Factor Expression in Human Airway Epithelial Cells
 IL-13 Augments Compressive Stress-induced Tissue Factor Expression in Human Airway Epithelial Cells Jennifer A. Mitchel, Silvio Antoniak, Joo-Hyeon Lee, Sae-Hoon Kim, Maureen McGill, David I. Kasahara,
IL-13 Augments Compressive Stress-induced Tissue Factor Expression in Human Airway Epithelial Cells Jennifer A. Mitchel, Silvio Antoniak, Joo-Hyeon Lee, Sae-Hoon Kim, Maureen McGill, David I. Kasahara,
Don t Freak Out. Test on cell organelle on Friday!
 Cell Structure 1 Don t Freak Out Test on cell organelle on Friday! This test should be a buffer test and help raise your overall test score. All information will come from this week! 2 Cells Provide Compartments
Cell Structure 1 Don t Freak Out Test on cell organelle on Friday! This test should be a buffer test and help raise your overall test score. All information will come from this week! 2 Cells Provide Compartments
AP Biology Book Notes Chapter 4: Cells v Cell theory implications Ø Studying cell biology is in some sense the same as studying life Ø Life is
 AP Biology Book Notes Chapter 4: Cells v Cell theory implications Ø Studying cell biology is in some sense the same as studying life Ø Life is continuous v Small cell size is becoming more necessary as
AP Biology Book Notes Chapter 4: Cells v Cell theory implications Ø Studying cell biology is in some sense the same as studying life Ø Life is continuous v Small cell size is becoming more necessary as
Muscle and Muscle Tissue
 Muscle and Muscle Tissue Make up about half of total body mass Exerts force by converting chemical energy, ATP, to mechanical energy Muscle tissue is classified based on Shape Number and position of nuclei
Muscle and Muscle Tissue Make up about half of total body mass Exerts force by converting chemical energy, ATP, to mechanical energy Muscle tissue is classified based on Shape Number and position of nuclei
Early scientists who observed cells made detailed sketches of what they saw.
 Early scientists who observed cells made detailed sketches of what they saw. Early scientists who observed cells made detailed sketches of what they saw. CORK Early scientists who observed cells made detailed
Early scientists who observed cells made detailed sketches of what they saw. Early scientists who observed cells made detailed sketches of what they saw. CORK Early scientists who observed cells made detailed
Cell Overview. Hanan Jafar BDS.MSc.PhD
 Cell Overview Hanan Jafar BDS.MSc.PhD THE CELL is made of: 1- Nucleus 2- Cell Membrane 3- Cytoplasm THE CELL Formed of: 1. Nuclear envelope 2. Chromatin 3. Nucleolus 4. Nucleoplasm (nuclear matrix) NUCLEUS
Cell Overview Hanan Jafar BDS.MSc.PhD THE CELL is made of: 1- Nucleus 2- Cell Membrane 3- Cytoplasm THE CELL Formed of: 1. Nuclear envelope 2. Chromatin 3. Nucleolus 4. Nucleoplasm (nuclear matrix) NUCLEUS
The Cytoplasm Li Shulei Department of Histology & Embryology
 The Cytoplasm Li Shulei lishulei@tom.com Department of Histology & Embryology Cell components Cytoplasm Plasma membrane Organelles Cytoplasmic deposits Cytoskeleton Cytosol ( Matrix ) Nucleus Plasma membrane
The Cytoplasm Li Shulei lishulei@tom.com Department of Histology & Embryology Cell components Cytoplasm Plasma membrane Organelles Cytoplasmic deposits Cytoskeleton Cytosol ( Matrix ) Nucleus Plasma membrane
Muscle Tissue. General concepts. Classification of muscle. I. Functional classification is based on the type of neural control.
 Muscle Tissue LEARNING OBJECTIVES 1. Identify the three types of muscle tissue at the light microscopic level. 2. List and compare the structural and functional features of each of the three muscle fiber
Muscle Tissue LEARNING OBJECTIVES 1. Identify the three types of muscle tissue at the light microscopic level. 2. List and compare the structural and functional features of each of the three muscle fiber
cell movement and neuronal migration
 cell movement and neuronal migration Paul Letourneau letou001@umn.edu Chapter 16; The Cytoskeleton; Molecular Biology of the Cell, Alberts et al. 1 Cell migration Cell migration in 3 steps; protrusion,
cell movement and neuronal migration Paul Letourneau letou001@umn.edu Chapter 16; The Cytoskeleton; Molecular Biology of the Cell, Alberts et al. 1 Cell migration Cell migration in 3 steps; protrusion,
Epithelial interleukin-25 is a key mediator in Th2-high, corticosteroid-responsive
 Online Data Supplement: Epithelial interleukin-25 is a key mediator in Th2-high, corticosteroid-responsive asthma Dan Cheng, Zheng Xue, Lingling Yi, Huimin Shi, Kan Zhang, Xiaorong Huo, Luke R. Bonser,
Online Data Supplement: Epithelial interleukin-25 is a key mediator in Th2-high, corticosteroid-responsive asthma Dan Cheng, Zheng Xue, Lingling Yi, Huimin Shi, Kan Zhang, Xiaorong Huo, Luke R. Bonser,
10/5/2015. Cell Size. Relative Rate of Reaction
 The Cell Biology 102 Fundamental unit of life Smallest unit that displays all the basic elements of life Lecture 5: Cells Cell Theory 1. All living things are made of one or more cells Cell Theory 2. The
The Cell Biology 102 Fundamental unit of life Smallest unit that displays all the basic elements of life Lecture 5: Cells Cell Theory 1. All living things are made of one or more cells Cell Theory 2. The
Enzyme-coupled Receptors. Cell-surface receptors 1. Ion-channel-coupled receptors 2. G-protein-coupled receptors 3. Enzyme-coupled receptors
 Enzyme-coupled Receptors Cell-surface receptors 1. Ion-channel-coupled receptors 2. G-protein-coupled receptors 3. Enzyme-coupled receptors Cell-surface receptors allow a flow of ions across the plasma
Enzyme-coupled Receptors Cell-surface receptors 1. Ion-channel-coupled receptors 2. G-protein-coupled receptors 3. Enzyme-coupled receptors Cell-surface receptors allow a flow of ions across the plasma
ORGANELLES OF THE ENDOMEMBRANE SYSTEM
 Membranes compartmentalize the interior of the cell and facilitate a variety of metabolic activities. Chloroplasts and a rigid cell wall are what distinguish a plant cell from an animal cell. A typical
Membranes compartmentalize the interior of the cell and facilitate a variety of metabolic activities. Chloroplasts and a rigid cell wall are what distinguish a plant cell from an animal cell. A typical
BIO 5099: Molecular Biology for Computer Scientists (et al)
 BIO 5099: Molecular Biology for Computer Scientists (et al) Lecture 15: Being a Eukaryote: From DNA to Protein, A Tour of the Eukaryotic Cell. Christiaan van Woudenberg Being A Eukaryote Basic eukaryotes
BIO 5099: Molecular Biology for Computer Scientists (et al) Lecture 15: Being a Eukaryote: From DNA to Protein, A Tour of the Eukaryotic Cell. Christiaan van Woudenberg Being A Eukaryote Basic eukaryotes
Fluorescence Microscopy
 Fluorescence Microscopy Imaging Organelles Mitochondria Lysosomes Nuclei Endoplasmic Reticulum Plasma Membrane F-Actin AAT Bioquest Introduction: Organelle-Selective Stains Organelles are tiny, specialized
Fluorescence Microscopy Imaging Organelles Mitochondria Lysosomes Nuclei Endoplasmic Reticulum Plasma Membrane F-Actin AAT Bioquest Introduction: Organelle-Selective Stains Organelles are tiny, specialized
Superior Fluorescent Labeling Dyes Spanning the Full Visible Spectrum...1. Trademarks: HiLyte Fluor (AnaSpec, Inc.)
 Table of Contents Fluor TM Labeling Dyes Superior Fluorescent Labeling Dyes Spanning the Full Visible Spectrum....1 Fluor TM 405 Dye, an Excellent Alternative to Alexa Fluor 405 & DyLight 405....2 Fluor
Table of Contents Fluor TM Labeling Dyes Superior Fluorescent Labeling Dyes Spanning the Full Visible Spectrum....1 Fluor TM 405 Dye, an Excellent Alternative to Alexa Fluor 405 & DyLight 405....2 Fluor
Actin structure. Actin a highly conserved gene. Molecular Cell Biology
 Harvey Lodish Arnold Berk Paul Matsudaira Chris A. Kaiser Monty Krieger Matthew P. Scott Lawrence Zipursky James Darnell Molecular Cell Biology Fifth Edition Chapter 19: Cytoskeleton I: Microfilaments
Harvey Lodish Arnold Berk Paul Matsudaira Chris A. Kaiser Monty Krieger Matthew P. Scott Lawrence Zipursky James Darnell Molecular Cell Biology Fifth Edition Chapter 19: Cytoskeleton I: Microfilaments
An improved method for guinea pig airway smooth muscle cell culture and the effect of SPFF on intracellular calcium
 MOLECULAR MEDICINE REPORTS 10: 1309-1314, 2014 An improved method for guinea pig airway smooth muscle cell culture and the effect of SPFF on intracellular calcium JIAN LIU 1, YUYANG ZHANG 1, QIAN LI 1,
MOLECULAR MEDICINE REPORTS 10: 1309-1314, 2014 An improved method for guinea pig airway smooth muscle cell culture and the effect of SPFF on intracellular calcium JIAN LIU 1, YUYANG ZHANG 1, QIAN LI 1,
Exosomes/tricalcium phosphate combination scaffolds can enhance bone regeneration by activating the PI3K/Akt signalling pathway
 Exosomes/tricalcium phosphate combination scaffolds can enhance bone regeneration by activating the PI3K/Akt signalling pathway Jieyuan Zhang, Xiaolin Liu, Haiyan Li, Chunyuan Chen, Bin Hu, Xin Niu, Qing
Exosomes/tricalcium phosphate combination scaffolds can enhance bone regeneration by activating the PI3K/Akt signalling pathway Jieyuan Zhang, Xiaolin Liu, Haiyan Li, Chunyuan Chen, Bin Hu, Xin Niu, Qing
The Cell Organelles. Eukaryotic cell. The plasma membrane separates the cell from the environment. Plasma membrane: a cell s boundary
 Eukaryotic cell The Cell Organelles Enclosed by plasma membrane Subdivided into membrane bound compartments - organelles One of the organelles is membrane bound nucleus Cytoplasm contains supporting matrix
Eukaryotic cell The Cell Organelles Enclosed by plasma membrane Subdivided into membrane bound compartments - organelles One of the organelles is membrane bound nucleus Cytoplasm contains supporting matrix
Bio10 Cell Structure SRJC
 3.) Cell Structure and Function Structure of Cell Membranes Fluid mosaic model Mixed composition: Phospholipid bilayer Glycolipids Sterols Proteins Fluid Mosaic Model Phospholipids are not packed tightly
3.) Cell Structure and Function Structure of Cell Membranes Fluid mosaic model Mixed composition: Phospholipid bilayer Glycolipids Sterols Proteins Fluid Mosaic Model Phospholipids are not packed tightly
Notes Chapter 7 Cell Structure and Function Hooke looked at cork under a simple microscope and found tiny chambers he named cells.
 Notes Chapter 7 Cell Structure and Function 7.1 Cell discovery and Theory 1665 Hooke looked at cork under a simple microscope and found tiny chambers he named cells. Cells are the basic structural and
Notes Chapter 7 Cell Structure and Function 7.1 Cell discovery and Theory 1665 Hooke looked at cork under a simple microscope and found tiny chambers he named cells. Cells are the basic structural and
BIO 5099: Molecular Biology for Computer Scientists (et al) Lecture 15: Being A Eukaryote. Eukaryotic Cells. Basic eukaryotes have:
 BIO 5099: Molecular Biology for Computer Scientists (et al) Lecture 15: Being a Eukaryote: From DNA to Protein, A Tour of the Eukaryotic Cell. Christiaan van Woudenberg Being A Eukaryote Basic eukaryotes
BIO 5099: Molecular Biology for Computer Scientists (et al) Lecture 15: Being a Eukaryote: From DNA to Protein, A Tour of the Eukaryotic Cell. Christiaan van Woudenberg Being A Eukaryote Basic eukaryotes
A Tour of the Cell. Chapter 6. Biology. Edited by Shawn Lester. Inner Life of Cell. Eighth Edition Neil Campbell and Jane Reece
 Chapter 6 A Tour of the Cell Inner Life of Cell Edited by Shawn Lester PowerPoint Lecture Presentations for Biology Eighth Edition Neil Campbell and Jane Reece Lectures by Chris Romero, updated by Erin
Chapter 6 A Tour of the Cell Inner Life of Cell Edited by Shawn Lester PowerPoint Lecture Presentations for Biology Eighth Edition Neil Campbell and Jane Reece Lectures by Chris Romero, updated by Erin
NOTES: Chapter 6 STRUCTURE OF THE CELL
 Biology 120 Section 10 J. Greg Doheny NOTES: Chapter 6 STRUCTURE OF THE CELL NOTES Relative sizes of cells: If a Eukaryotic cell was the size of Columbia College, a prokaryotic cell (like a bacterium)
Biology 120 Section 10 J. Greg Doheny NOTES: Chapter 6 STRUCTURE OF THE CELL NOTES Relative sizes of cells: If a Eukaryotic cell was the size of Columbia College, a prokaryotic cell (like a bacterium)
The subcortical maternal complex controls symmetric division of mouse zygotes by
 The subcortical maternal complex controls symmetric division of mouse zygotes by regulating F-actin dynamics Xing-Jiang Yu 1,2, Zhaohong Yi 1, Zheng Gao 1,2, Dan-dan Qin 1,2, Yanhua Zhai 1, Xue Chen 1,
The subcortical maternal complex controls symmetric division of mouse zygotes by regulating F-actin dynamics Xing-Jiang Yu 1,2, Zhaohong Yi 1, Zheng Gao 1,2, Dan-dan Qin 1,2, Yanhua Zhai 1, Xue Chen 1,
Cell Communication. Biology Eighth Edition Neil Campbell and Jane Reece. PowerPoint Lecture Presentations for
 Chapter 11 Cell Communication PowerPoint Lecture Presentations for Biology Eighth Edition Neil Campbell and Jane Reece Lectures by Chris Romero, updated by Erin Barley with contributions from Joan Sharp
Chapter 11 Cell Communication PowerPoint Lecture Presentations for Biology Eighth Edition Neil Campbell and Jane Reece Lectures by Chris Romero, updated by Erin Barley with contributions from Joan Sharp
The Cell. The building blocks of life
 The Cell The building blocks of life Learning Goals I can describe the cell theory. I can differentiate between a prokaryotic and eukaryotic cell. I can describe the similarities and differences between
The Cell The building blocks of life Learning Goals I can describe the cell theory. I can differentiate between a prokaryotic and eukaryotic cell. I can describe the similarities and differences between
1.2 introduction to the cell. me239 mechanics of the cell. 1.2 introduction to the cell. 1.2 introduction to the cell.
 2. introduction to mechanics prokaryotic cells Figure 1.1 Prokaryotic cell. Cell without a nucleus. the inner life of a cell, viel & lue, harvard [2006] me239 mechanics of the cell 1 eukaryotic cells 1.2
2. introduction to mechanics prokaryotic cells Figure 1.1 Prokaryotic cell. Cell without a nucleus. the inner life of a cell, viel & lue, harvard [2006] me239 mechanics of the cell 1 eukaryotic cells 1.2
Nucleic acids. Nucleic acids are information-rich polymers of nucleotides
 Nucleic acids Nucleic acids are information-rich polymers of nucleotides DNA and RNA Serve as the blueprints for proteins and thus control the life of a cell RNA and DNA are made up of very similar nucleotides.
Nucleic acids Nucleic acids are information-rich polymers of nucleotides DNA and RNA Serve as the blueprints for proteins and thus control the life of a cell RNA and DNA are made up of very similar nucleotides.
Cellular Communication
 Cellular Communication But before we get into that What have we learned about so far? Energy and Matter Why do living things need energy? Grow Reproduce Maintain homeostasis Cellular signaling Cells communicate
Cellular Communication But before we get into that What have we learned about so far? Energy and Matter Why do living things need energy? Grow Reproduce Maintain homeostasis Cellular signaling Cells communicate
Supporting Information
 Supporting Information The Effects of Spacer Length and Composition on Aptamer-Mediated Cell-Specific Targeting with Nanoscale PEGylated Liposomal Doxorubicin Hang Xing +, [a] Ji Li +, [a] Weidong Xu,
Supporting Information The Effects of Spacer Length and Composition on Aptamer-Mediated Cell-Specific Targeting with Nanoscale PEGylated Liposomal Doxorubicin Hang Xing +, [a] Ji Li +, [a] Weidong Xu,
Chapter 3: Cells 3-1
 Chapter 3: Cells 3-1 Introduction: A. Human body consists of 75 trillion cells B. About 260 types of cells that vary in shape & size yet have much in common B. Differences in cell shape make different
Chapter 3: Cells 3-1 Introduction: A. Human body consists of 75 trillion cells B. About 260 types of cells that vary in shape & size yet have much in common B. Differences in cell shape make different
Anatomy Chapter 2 - Cells
 Cells Cells are the basic living structural, functional unit of the body Cytology is the branch of science that studies cells The human body has 100 trillion cells 200 different cell types with a variety
Cells Cells are the basic living structural, functional unit of the body Cytology is the branch of science that studies cells The human body has 100 trillion cells 200 different cell types with a variety
Renáta Schipp Gergely Berta Department of Medical Biology
 The cell III. Renáta Schipp Gergely Berta Department of Medical Biology Size and Biology Biology is a visually rich subject many of the biological events and structures are smaller than the unaided human
The cell III. Renáta Schipp Gergely Berta Department of Medical Biology Size and Biology Biology is a visually rich subject many of the biological events and structures are smaller than the unaided human
Chapter 6 Review Page 1
 hapter 6 Review Page 1 1 Which of the following statements about prokaryotic cells and eukaryotic cells is FLS? They both contain ribosomes. They both contain mitochondria. They are both bounded by a plasma
hapter 6 Review Page 1 1 Which of the following statements about prokaryotic cells and eukaryotic cells is FLS? They both contain ribosomes. They both contain mitochondria. They are both bounded by a plasma
Cell Theory Vocabulary Flashcards
 Mr. Powner Biology Cell Theory Vocabulary Flashcards Instructions: Cut out the flashcards from the following pages. Use the following words to label the backside of the flashcards. The words are not listed
Mr. Powner Biology Cell Theory Vocabulary Flashcards Instructions: Cut out the flashcards from the following pages. Use the following words to label the backside of the flashcards. The words are not listed
Cell Communication. Chapter 11. Biology Eighth Edition Neil Campbell and Jane Reece. PowerPoint Lecture Presentations for
 Chapter 11 Cell Communication PowerPoint Lecture Presentations for Biology Eighth Edition Neil Campbell and Jane Reece Lectures by Chris Romero, updated by Erin Barley with contributions from Joan Sharp
Chapter 11 Cell Communication PowerPoint Lecture Presentations for Biology Eighth Edition Neil Campbell and Jane Reece Lectures by Chris Romero, updated by Erin Barley with contributions from Joan Sharp
Advances in Computer Science Research, volume 59 7th International Conference on Education, Management, Computer and Medicine (EMCM 2016)
 7th International Conference on Education, Management, Computer and Medicine (EMCM 2016) Expression of Beta-Adrenergic Receptor in Glioma LN229 Cells and Its Effect on Cell Proliferation Ping Wang1, Qingluan
7th International Conference on Education, Management, Computer and Medicine (EMCM 2016) Expression of Beta-Adrenergic Receptor in Glioma LN229 Cells and Its Effect on Cell Proliferation Ping Wang1, Qingluan
Eukaryotic Cell Structures
 Comparing the Cell to a Factory Eukaryotic Cell Structures Structures within a eukaryotic cell that perform important cellular functions are known as organelles. Cell biologists divide the eukaryotic cell
Comparing the Cell to a Factory Eukaryotic Cell Structures Structures within a eukaryotic cell that perform important cellular functions are known as organelles. Cell biologists divide the eukaryotic cell
UNC-Duke Biology Course for Residents Fall Cell Cycle Effects of Radiation
 UNC-Duke Biology Course for Residents Fall 2018 1 Cell Cycle: Sequence of changes in a cell starting with the moment the cell is created by cell division, continuing through the doubling of the DNA and
UNC-Duke Biology Course for Residents Fall 2018 1 Cell Cycle: Sequence of changes in a cell starting with the moment the cell is created by cell division, continuing through the doubling of the DNA and
BIOSC 041. v Today s lecture. v Today s lab. v Note- Monday is a holiday good time to do some reading!
 BIOSC 041 v Today s lecture Review questions Chapter 6, Cells More review questions v Today s lab Quick review of lab safety The Scientific Method start thinking about which environments you might want
BIOSC 041 v Today s lecture Review questions Chapter 6, Cells More review questions v Today s lab Quick review of lab safety The Scientific Method start thinking about which environments you might want
Cell Biology Lecture 9 Notes Basic Principles of cell signaling and GPCR system
 Cell Biology Lecture 9 Notes Basic Principles of cell signaling and GPCR system Basic Elements of cell signaling: Signal or signaling molecule (ligand, first messenger) o Small molecules (epinephrine,
Cell Biology Lecture 9 Notes Basic Principles of cell signaling and GPCR system Basic Elements of cell signaling: Signal or signaling molecule (ligand, first messenger) o Small molecules (epinephrine,
A Tour of the Cell. reference: Chapter 6. Reference: Chapter 2
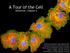 A Tour of the Cell reference: Chapter 6 Reference: Chapter 2 Monkey Fibroblast Cells stained with fluorescent dyes to show the nucleus (blue) and cytoskeleton (yellow and red fibers), image courtesy of
A Tour of the Cell reference: Chapter 6 Reference: Chapter 2 Monkey Fibroblast Cells stained with fluorescent dyes to show the nucleus (blue) and cytoskeleton (yellow and red fibers), image courtesy of
