Updates in Therapeutics 2015: The Pharmacotherapy Preparatory Review & Recertification Course
|
|
|
- Ashlyn Charles
- 5 years ago
- Views:
Transcription
1 Updates in Therapeutics 2015: The Pharmacotherapy Preparatory Review & Recertification Course Study Designs: Fundamentals and Interpretation Kevin M. Sowinski, Pharm.D., FCCP Purdue University, College of Pharmacy Indiana University, School of Medicine West Lafayette and Indianapolis, IN Page 1-469
2 Outline Introduction What and why you need to know Validity, Bias and Confounding Clinical Study Designs Observational Interventional Clinical Trials Analysis and Interpretation Summary Measures of Effect Miscellaneous
3 Issues in Study Design Study Purpose: Descriptive vs. Analytical Time Orientation Prospective Retrospective Investigator Orientation Interventional vs. Quasiexperimental Experimental Setting RCTs Observational Trials Page 1-472
4 Relative Strength of Evidence: Hierarchy of Study Designs Page 1-472
5 Validity in Study Design Internal validity Validity within the confines of the study methods Does the study design adequately and appropriately test/measure what it purports? Does the study adequately and appropriately address bias, confounding, and measurement of end points? External validity Validity related to generalizing the study results outside of the study setting Can the results be applied to other groups, patients, or systems? Addresses issues of generalizability and representativeness Page 1-473
6 Bias in Study Design Systematic, non-random variation in study methodology and conductance introducing error in interpretation Selection bias: Arise from selection of subjects Sampling bias Observation or information bias Recall bias Interviewer bias Misclassification bias Page 1-473
7 Confounding in Study Design Variable that impacts the independent/dependent variable altering the ability to determine the true effect on outcome May hide or exaggerate a true association All relevant information should be collected and evaluated Controlling for confounding during the design of a study Randomization Restriction Matching Controlling for confounding during the analysis of a study Stratification Multivariate analysis Page 1-473
8 Causality Temporality Strength Biological gradient: Dose-response Consistency Specificity Plausibility Coherence Analogy Experiment Page 1-474
9 Types of Clinical Study Design Case Reports/Case Series Document and describe experiences, novel treatments and unusual events Hypotheses generation Example: QT interval prolongation associated with FQ antibiotics Case report: One patient Case series: > 1 patient with a similar experience or multiple case reports combined Sufficient detail to recognize same/ similar cases Is IRB approval required? Page 1-474
10 Types of Clinical Study Design Observations Study Designs Case-control Cohort Cross-sectional Design does not involve investigator intervention, only observation Observational study designs investigate association, not causation Pages
11 Observations Study Designs Case-control Determine the association between exposures (risks) and disease (condition) Classic example: Aspirin use and Reye s Syndrome Often referred to as retrospective studies Useful to study exposures in rare diseases or ones that take long periods to develop Critical assumptions to minimize bias: Cases are selected to be representative of those who have the disease Controls are representative of the population without the disease and are as identical as possible to the cases (- Dz) Information is collected from cases and controls in the same way Pages
12 Observations Study Designs Case-control Advantages Inexpensive and can be conducted quickly Allows investigation of multiple possible exposures /associations Disadvantages Confounding must be controlled for Observation and recall bias: looking back to remember Selection bias: Case selection and control matching is difficult Measure of Association: Odds Ratio Pages
13 Observations Study Designs Cohort Study Determine the association between exposures/ factors and disease/condition development. Estimation of the risk of outcome (RR between the exposure groups) and study outcome of interest in those with and without an exposure Risk of an event or development of a condition relative to exposure Classic example: Framingham study Describes the incidence or natural history of a disease/condition and measures it in time sequence Pages
14 Observations Study Designs Cohort Study: Retrospective Begins and ends in the present but involves a major backward look to collect information about events that occurred in the past Advantages: Less expensive and time-consuming; no loss to follow-up, ability to investigate issues not amenable to a clinical trial or ethical or safety issues Disadvantages: Only as good as the data available, little control of confounding variables through nonstatistical approaches, recall bias Pages
15 Observations Study Designs Cohort Study: Prospective Begin in the present and progress forward, collecting data from subjects whose outcome lies in the future. Advantages: Easier to control for confounding factors, easier to plan for data collection Disadvantages: More expensive and timeintensive, loss of follow-up, difficult to study rare diseases/conditions at a reasonable cost Pages
16 Observations Study Designs Cross Sectional or Prevalence Study Identify the prevalence or characteristics of a condition in a group of individuals Snapshot in time Advantages: Easy design, data collected at one time Questionnaire, interview, or other available information Disadvantages: Does not allow a study of factors in individual subjects over time, difficult-to-study rare conditions Pages 1-477
17 Incidence and Prevalence Incidence Measure of the probability of developing a disease Incidence rate: Number of new cases of disease per population in a specified time Measured in persons/year Prevalence Measure of the number of individuals who have a condition/disease at any given time. Point prevalence (date) vs. Period prevalence (year, month) Pages 1-478
18 Interpreting Relative Risks/Odds Ratios Estimate the magnitude of association between exposure and disease. Key point:this is not cause and effect but association RR (a.k.a risk ratio): Cohort studies OR: Case control studies (estimate of the RR) The RR and OR are interpreted on the basis of their difference from 1. If the 95% CI includes 1, no statistical difference is indicated Pages 1-478
19 Interpreting Index of Risk Direction of risk Magnitude of Risk RR OR Interpretation < 1 < 1 Negative association, RR: Risk lower in exposed group, OR: Odds of exposure is lower in diseased group =1 =1 No association, RR: Risk is the same, OR: Odds of exposure is the same > 1 > 1 Positive association,, RR: Risk is greater in exposed group OR: Odds of exposure is greater on diseased group RR OR Interpretation % reduction in the risk/odds No difference in risk/odds % increase in the risk/odds fold (or 200%) increase in the risk/odds Pages 1-478
20 PPA Study Interpretation Cases (+ stroke) n=383 Controls ( stroke) n=750 Adjusted OR (95% CI) Appetite suppressant: Women ( ) Appetite suppressant: Men 0 0 Appetite suppressant: Either ( ) PPA: Women ( ) PPA: Men ( ) PPA: Either ( ) Interpret the point estimate and 95% CI in all cases? What does the point estimate mean? What does the CI mean? Which ones are statistically significant? Pages N Engl J Med 2000;343:
21 Observations Study Designs Causation REMEMBER: association not causality Considerations when evaluating causality Statistical significance observed? Strength of the association? Dose-response? Temporal relationship? Have the results been consistently shown? Biologic plausibility? Experimental (animal, in vitro, etc.) evidence? Page 1-480
22 Observational Study Design Summary of Characteristics Study Design Exposure or Outcome? Measure of Association Major Advantages Major Disadvantages Case Report/ Case Series Outcome Generate new information about natural history of Dz ID new disease/condition Usually can t measure rates of association Case-control Outcome OR Study relatively rare diseases Not practical for studying Low cost and short duration rare exposures Inability to study multiple outcomes in one study Cohort Exposure RR Study relatively rare exposures Study temporal associations Estimate direct risk estimates Not practical for studying rare diseases Increased cost and longer duration (prospective) Crosssectional N/A Prevalence Low cost and short duration Temporal associations can t be established Adapted from Pharmacotherapy 2010;30:
23 Interventional Study Design Randomized, Controlled Trials Make intervention and evaluate cause and effect Design allows assessment of causality Minimizes bias through randomization and/or stratification Parallel vs. crossover design Crossover provides practical and statistical efficiency. Crossover is not appropriate for certain types of treatment questions. Effect of treatment on a disease that worsens during the study period Pages
24 Interventional Study Design Randomized, Controlled Trials Examples: Clinical trial: Comparison of two drugs, two behavioral modifications... Educational intervention: Online course versus lecture class format Health care intervention: RPh vs. non-rphbased health care team Pages
25 Interventional Study Design Randomized, Controlled Trials Are the results of the study valid? What were the results? Can I apply the results of this study to my patient population? Will they help me care for my patients? Other issues related to RCT Subgroup analyses Primary, Composite and Surrogate Endpoints Superiority, Equivalence, Non-Inferiority Pages
26 Randomized, Controlled Trials Subgroup Analysis Important part of controlled clinical trials Often overused and over-interpreted Many potential pitfalls in identifying and interpreting: Failure to account for multiple comparisons or adjust p-values Problems with sample size, power, classification, and lack of assessment of interaction Page 1-483
27 Randomized, Controlled Trials Primary and Composite End Points Primary end point: crucial design decision What does the following statement mean? ramipril reduces the rate of death, MI, stroke, revascularization, cardiac arrest, HF, complications related to DM, and new cases of DM in high-risk patients. Treating 1000 patients with ramipril for 4 years prevents about 150 events in around 70 patients. Was there a reduction in all the end points or just some? Are all the outcomes just as likely to occur? Why would this trial have been interested in all of these outcomes? Page 1-483
28 Randomized, Controlled Trials Composite End Points Positives for using composite end points? Problems? Difficulties in interpreting composite end points Misattribution of statistically beneficial effects of composite measure to each of its component end points Dilution of effects, negative results for relatively common component of composite end point hide real differences in other end points. Undue influence exerted on composite end point by softer component end points Averaging of overall effect Should all end points weigh the same, or death weigh more? Results for each individual end point should be reported with the results for the composite Page 1-483
29 Randomized, Controlled Trials Surrogate End Points Parameters thought to be associated with clinical outcomes BP reduction and stroke prevention LDL-C reduction and CV death reduction Statins vs. hormone replacement therapy PVC suppression and mortality reduction Surrogate outcomes predict clinical outcomes Short-duration studies with surrogate end points may be too small to detect uncommon AEs Page 1-483
30 Randomized, Controlled Trials Superiority vs. Equivalence vs. Non-inferiority Superiority: Detect a difference between Txs Typical design in a clinical trial. Equivalence: Confirm the absence of meaningful difference(s) between Txs What difference is important? Non-inferiority: Investigate whether a Tx is not clinically worse (no less effective, or inferior) May be the most effective, or have a similar effect. Useful when placebo is not possible due to ethical reasons Page 1-484
31 Randomized, Controlled Trials Non-inferiority Design: ONTARGET Telmisartan, ramipril, or combination in patients with a high risk of VDz Is telmisartan non-inferior in the incidence of CV deaths? Non-inferior difference defined as < 13% Essentials of non-inferiority design Control group (ramipril) must be effective Study similar to previous study with control (HOPE) and with equal doses, clinical conditions, and design Adequate power is essential, and usually, larger sample sizes are required. Page 1-484
32 Randomized Trial of ERT-P for Secondary Prevention of CHD in Postmenopausal Women Randomized trial of ERT-P for secondary prevention of CAD in postmenopausal women Objective: Does ERT-P therapy alter the risk of CHD in postmenopausal women with established CHD? Randomized, blinded, placebo controlled CEE mg/day plus MPA 2.5 mg/day (ERT-P) and placebo n=2763 with CAD < 80; mean age = 66.7 years Follow-up averaged 4.1 years; 82% of HRT still taking at the end of 1 year; 75% 3 years Pages JAMA 1998;280:605 13
33 Randomized Trial of ERT-P for Secondary Prevention of CHD in Postmenopausal Women End points Primary: Nonfatal MI, CHD death Secondary: Many, including all-cause mortality. Are these composite outcomes appropriate? Surrogate end point: LDL-C lowered Statistical analysis: Baseline characteristics: t-test and Chi-square Power analysis and sample size calculation Kaplan-Meier with Cox proportional hazards model, intention to treat Pages JAMA 1998;280:605 13
34 Baseline Characteristics Demographics Age, mean±sd, yrs White, % Education, mean±sd, yrs ERT-P (n=1380) 67± ±3 Placebo (n=1383) 67± ±3 p-value Statistical analysis: Baseline characteristics: t-test and Chi-square Pages JAMA 1998;280:605 13
35 Surrogate Endpoints Change in Lipid Profiles after 1 year p<0.001 Statistics: No documented test for above comparison mean LDL-C decreased.p<0.001 for the difference between groups) Pages JAMA 1998;280:605 13
36 What is appropriate test?? Mean LDL (and others) decrease? A. Wilcoxon signed rank test B. Chi-square test C. Two-way ANOVA D. Two-sample t-test
37 Randomized, Controlled Trials Composite Endpoint ERT-P Placebo HR (95% CI) Primary CHD events ( ) CHD death ( ) Any thromboembolic event ( ) Gall bladder disease ( ) Statistics: Kaplan-Meier with Cox proportional hazards model, intention to treat Significant time trend: More CHD events in the treatment group than in placebo in year 1 and fewer in years 4 and 5 Which are statistically different? Yes No Conclusions Pages JAMA 1998;280:605 13
38 Common Approaches to Analyzing Clinical Trials: Intention to treat Compares outcomes based on randomization How they were intended to be treated Treatment effects under usual conditions. Analogous to routine clinical practice in which a patient receives a prescription but may not adhere to the drug Conservative estimate (may underestimate) of differences in treatment Most common approach to assessing clinical trial results Pages
39 Common Approaches to Analyzing Clinical Trials: Per-Protocol Analysis Those who do not complete/adhere to treatment are not included in the final analysis Provides additional information about treatment effectiveness and more generous estimates of differences Subject to several issues such as lower sample size and definitions of adherence Results are more difficult to interpret Page 1-486
40 Common Approaches to Analyzing Clinical Trials: As-Treated Analysis Analyzed by the actual intervention received Subjects who were in the treatment group but did not take treatment would be analyzed as if they were in the placebo group. This analysis essentially ignores the randomization process for those who did not adhere to the study design Page 1-486
41 Systematic reviews Summary that uses explicit methods to perform a comprehensive literature search, critically appraise it, and synthesize the literature Differs from a standard literature review Key is a well-documented and described systematic review. Some systematic reviews will attempt to statistically combine results from many studies Differs from other reviews, which combine evaluation with opinions Page 1-486
42 Meta-analysis Systematic review that uses statistical techniques to summarize the results of the evaluated studies These techniques may improve on: Calculation of effect size Increase statistical power Interpretation of disparate results Reduce bias Answers to questions that may not be addressable with additional study Pages
43 Issues related to meta-analysis Reliant on criteria for inclusion of previous studies and statistical methods to ensure validity. Details of included studies are essential. Page 1-487
44 Elements of trial methods Research question Identification of available studies Criteria for trial inclusion/exclusion Data collection and presentation of findings Calculation of summary estimate Page 1-487
45 Forest plot Page Pharmacotherapy 2010;30:119-26
46 Elements of trial methods Assessment of heterogeneity Statistical heterogeneity χ 2 and Cochrane Q are common tests for heterogeneity. Assessment of publication bias Sensitivity analysis Page 1-487
47 Summary Measures of Effect Absolute vs. Relative Differences Absolute differences or absolute changes Relative differences or relative changes Absolute differences are more important than relative differences Authors highlight the differences observed in their trials with relative differences because they are larger Why? Larger numbers are more convincing Most drug advertisements (both directly to patients and to health care professionals) quote relative differences Page 1-487
48 Heart Outcomes Prevention Evaluation (HOPE) Study Randomized, double blind, placebo controlled study 9297 high-risk patients received ramipril or placebo daily; average follow-up of 5 years Primary outcome: Composite of MI, stroke, or death from cardiovascular causes Page N Engl J Med 2000;342:145 53
49 Absolute vs. Relative Differences Risk reduction of Primary End Point Proportion of Patients Placebo Ramipril % RRR 4 % ARR Time (Days) Page Data extracted and adapted from NEJM 2000;342:145-53
50 Summary Measures of Effect Number Needed to Treat Another means to characterize changes or differences in absolute risk Definition: The reciprocal of the ARR NNT = 1/(ARR) Rounded to the next highest whole num Applied to clinical outcomes with dichotomous data Cautions: Assumes the baseline risk is the same for all (or that it is unrelated to RR) Extrapolation beyond studied time duration NNTs should only be provided for significant effects Number needed to harm Page 1-488
51 NNT Application HOPE study Results NNT = 1/( ) = 1/0.038 = 26.3, rounded up to 27 Interpretation NNT for each endpoint Outcome Ramipril (%) Placebo (%) Relative Risk RRR ARR NNT Combined Death from CV causes Myocardial infarction Stroke Page N Engl J Med 2000;342:145 53
52 Reporting Guidelines for Clinical Studies The Consolidated Standards of Reporting Trials (CONSORT) Updated in 2010 Improve, standardize and increase the transparency of the reporting of clinical trials The CONSORT statement and checklist The Flow Diagram Extensions to the Consort Statement Pages
53 Reporting Guidelines for Clinical Studies Strengthening the reporting of observational studies in epidemiology (STROBE) statement Initially published in 2007 Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) Initially published in 1996, renamed in 2009 Enhancing the Quality and Transparency of Health Research (EQUATOR) network Pages
54 Pharmacoeconomics Description and analysis of the costs of drug therapy to health care systems and society Identifies, measures and compares the costs/consequences of pharmaceutical products and services Outcomes research: identify, measure and evaluate the end results of health care services Types of pharmacoeconomic analyses Page 1-490
55 Pharmacoeconomic Studies Type of Analysis Outcome Objective Cost-minimization Equal Lowest cost alternative Cost-benefit Cost-effectiveness Monetary Worth the cost? Clinical Units (BP reduction, etc.) Greatest net benefit alternative Most cost-effective alternative Cost-utility Utility, QALY Greatest benefit alternative Page 1-490
56 Sensitivity/Specificity/Predictive Values Sensitivity: Proportion of true positives that are correctly identified by a test Specificity: Proportion of true negatives that are correctly identified by a test Positive Predictive Value: Proportion of patients with a positive test who are given a correct diagnosis Negative Predictive Value: Proportion of patients with a negative test who are given a correct diagnosis Page 1-490
57 Summary
Learning Objectives 9/9/2013. Hypothesis Testing. Conflicts of Interest. Descriptive statistics: Numerical methods Measures of Central Tendency
 Conflicts of Interest I have no conflict of interest to disclose Biostatistics Kevin M. Sowinski, Pharm.D., FCCP Last-Chance Ambulatory Care Webinar Thursday, September 5, 2013 Learning Objectives For
Conflicts of Interest I have no conflict of interest to disclose Biostatistics Kevin M. Sowinski, Pharm.D., FCCP Last-Chance Ambulatory Care Webinar Thursday, September 5, 2013 Learning Objectives For
9/4/2013. Decision Errors. Hypothesis Testing. Conflicts of Interest. Descriptive statistics: Numerical methods Measures of Central Tendency
 Conflicts of Interest I have no conflict of interest to disclose Biostatistics Kevin M. Sowinski, Pharm.D., FCCP Pharmacotherapy Webinar Review Course Tuesday, September 3, 2013 Descriptive statistics:
Conflicts of Interest I have no conflict of interest to disclose Biostatistics Kevin M. Sowinski, Pharm.D., FCCP Pharmacotherapy Webinar Review Course Tuesday, September 3, 2013 Descriptive statistics:
Critical Appraisal of a Meta-Analysis: Rosiglitazone and CV Death. Debra Moy Faculty of Pharmacy University of Toronto
 Critical Appraisal of a Meta-Analysis: Rosiglitazone and CV Death Debra Moy Faculty of Pharmacy University of Toronto Goal To provide practitioners with a systematic approach to evaluating a meta analysis
Critical Appraisal of a Meta-Analysis: Rosiglitazone and CV Death Debra Moy Faculty of Pharmacy University of Toronto Goal To provide practitioners with a systematic approach to evaluating a meta analysis
Evidence Based Medicine
 Course Goals Goals 1. Understand basic concepts of evidence based medicine (EBM) and how EBM facilitates optimal patient care. 2. Develop a basic understanding of how clinical research studies are designed
Course Goals Goals 1. Understand basic concepts of evidence based medicine (EBM) and how EBM facilitates optimal patient care. 2. Develop a basic understanding of how clinical research studies are designed
Clinical Evidence: Asking the Question and Understanding the Answer. Keeping Up to Date. Keeping Up to Date
 Clinical Evidence: Asking the Question and Understanding the Answer Keeping Up to Date 5,000? per day 1,500 per day 95 per day Keeping Up to Date 5,000? per day 1,500 per day 95 per day 1 Bias Bias refers
Clinical Evidence: Asking the Question and Understanding the Answer Keeping Up to Date 5,000? per day 1,500 per day 95 per day Keeping Up to Date 5,000? per day 1,500 per day 95 per day 1 Bias Bias refers
Biases in clinical research. Seungho Ryu, MD, PhD Kanguk Samsung Hospital, Sungkyunkwan University
 Biases in clinical research Seungho Ryu, MD, PhD Kanguk Samsung Hospital, Sungkyunkwan University Learning objectives Describe the threats to causal inferences in clinical studies Understand the role of
Biases in clinical research Seungho Ryu, MD, PhD Kanguk Samsung Hospital, Sungkyunkwan University Learning objectives Describe the threats to causal inferences in clinical studies Understand the role of
JAMA. 2011;305(24): Nora A. Kalagi, MSc
 JAMA. 2011;305(24):2556-2564 By Nora A. Kalagi, MSc Cardiovascular disease (CVD) is the number one cause of mortality and morbidity world wide Reducing high blood cholesterol which is a risk factor for
JAMA. 2011;305(24):2556-2564 By Nora A. Kalagi, MSc Cardiovascular disease (CVD) is the number one cause of mortality and morbidity world wide Reducing high blood cholesterol which is a risk factor for
Conflict of Interest Disclosures. Statistics: WHY do you need to know it? Outline
 Conflict of Interest Disclosures No conflicts of interest to disclose related to this presentation Updates in Therapeutics 2012: Biostatistics: A Refresher Kevin M. Sowinski, Pharm.D., FCCP Purdue University,
Conflict of Interest Disclosures No conflicts of interest to disclose related to this presentation Updates in Therapeutics 2012: Biostatistics: A Refresher Kevin M. Sowinski, Pharm.D., FCCP Purdue University,
Evidence-Based Medicine Journal Club. A Primer in Statistics, Study Design, and Epidemiology. August, 2013
 Evidence-Based Medicine Journal Club A Primer in Statistics, Study Design, and Epidemiology August, 2013 Rationale for EBM Conscientious, explicit, and judicious use Beyond clinical experience and physiologic
Evidence-Based Medicine Journal Club A Primer in Statistics, Study Design, and Epidemiology August, 2013 Rationale for EBM Conscientious, explicit, and judicious use Beyond clinical experience and physiologic
EBM: Therapy. Thunyarat Anothaisintawee, M.D., Ph.D. Department of Family Medicine, Ramathibodi Hospital, Mahidol University
 EBM: Therapy Thunyarat Anothaisintawee, M.D., Ph.D. Department of Family Medicine, Ramathibodi Hospital, Mahidol University How to appraise therapy literature Are the result valid? What are the result?
EBM: Therapy Thunyarat Anothaisintawee, M.D., Ph.D. Department of Family Medicine, Ramathibodi Hospital, Mahidol University How to appraise therapy literature Are the result valid? What are the result?
the standard deviation (SD) is a measure of how much dispersion exists from the mean SD = square root (variance)
 Normal distribution The normal distribution is also known as the Gaussian distribution or 'bell-shaped' distribution. It describes the spread of many biological and clinical measurements Properties of
Normal distribution The normal distribution is also known as the Gaussian distribution or 'bell-shaped' distribution. It describes the spread of many biological and clinical measurements Properties of
Do Women Benefit From Statins for Primary Prevention?: Controversy, Challenges and Consensus
 Do Women Benefit From Statins for Primary Prevention?: Controversy, Challenges and Consensus C. Noel Bairey Merz MD, FACC, FAHA Professor and Women s Guild Endowed Chair Director, Barbra Streisand Women
Do Women Benefit From Statins for Primary Prevention?: Controversy, Challenges and Consensus C. Noel Bairey Merz MD, FACC, FAHA Professor and Women s Guild Endowed Chair Director, Barbra Streisand Women
Outline. What is Evidence-Based Practice? EVIDENCE-BASED PRACTICE. What EBP is Not:
 Evidence Based Practice Primer Outline Evidence Based Practice (EBP) EBP overview and process Formulating clinical questions (PICO) Searching for EB answers Trial design Critical appraisal Assessing the
Evidence Based Practice Primer Outline Evidence Based Practice (EBP) EBP overview and process Formulating clinical questions (PICO) Searching for EB answers Trial design Critical appraisal Assessing the
John J.P. Kastelein MD PhD Professor of Medicine Dept. of Vascular Medicine Academic Medial Center / University of Amsterdam
 Latest Insights from the JUPITER Study John J.P. Kastelein MD PhD Professor of Medicine Dept. of Vascular Medicine Academic Medial Center / University of Amsterdam Inflammation, hscrp, and Vascular Prevention
Latest Insights from the JUPITER Study John J.P. Kastelein MD PhD Professor of Medicine Dept. of Vascular Medicine Academic Medial Center / University of Amsterdam Inflammation, hscrp, and Vascular Prevention
Medication Adherence and Outcomes in High Risk Cardiovascular Patients in the ONTARGET Trial
 Medication Adherence and Outcomes in High Risk Cardiovascular Patients in the ONTARGET Trial M. Böhm, H. Schumacher, U. Laufs, P. Sleight, R. Schmieder, T. Unger, K. Koon, S. Yusuf on behalf of the ONTARGET-Investigators
Medication Adherence and Outcomes in High Risk Cardiovascular Patients in the ONTARGET Trial M. Böhm, H. Schumacher, U. Laufs, P. Sleight, R. Schmieder, T. Unger, K. Koon, S. Yusuf on behalf of the ONTARGET-Investigators
Medication Adherence and Outcomes in High Risk Cardiovascular Patients in the ONTARGET Trial
 Medication Adherence and Outcomes in High Risk Cardiovascular Patients in the ONTARGET Trial M. Böhm, H. Schumacher, U. Laufs, P. Sleight, R. Schmieder, T. Unger, K. Koon, S. Yusuf on behalf of the ONTARGET-Investigators
Medication Adherence and Outcomes in High Risk Cardiovascular Patients in the ONTARGET Trial M. Böhm, H. Schumacher, U. Laufs, P. Sleight, R. Schmieder, T. Unger, K. Koon, S. Yusuf on behalf of the ONTARGET-Investigators
Observational Study Designs. Review. Today. Measures of disease occurrence. Cohort Studies
 Observational Study Designs Denise Boudreau, PhD Center for Health Studies Group Health Cooperative Today Review cohort studies Case-control studies Design Identifying cases and controls Measuring exposure
Observational Study Designs Denise Boudreau, PhD Center for Health Studies Group Health Cooperative Today Review cohort studies Case-control studies Design Identifying cases and controls Measuring exposure
Overview. Goals of Interpretation. Methodology. Reasons to Read and Evaluate
 Overview Critical Literature Evaluation and Biostatistics Ahl Ashley N. Lewis, PharmD, BCPS Clinical Specialist, Drug Information UNC Hospitals Background Review of basic statistics Statistical tests Clinical
Overview Critical Literature Evaluation and Biostatistics Ahl Ashley N. Lewis, PharmD, BCPS Clinical Specialist, Drug Information UNC Hospitals Background Review of basic statistics Statistical tests Clinical
INTERPRETATION OF STUDY FINDINGS: PART I. Julie E. Buring, ScD Harvard School of Public Health Boston, MA
 INTERPRETATION OF STUDY FINDINGS: PART I Julie E. Buring, ScD Harvard School of Public Health Boston, MA Drawing Conclusions TRUTH IN THE UNIVERSE Infer TRUTH IN THE STUDY Infer FINDINGS IN THE STUDY Designing
INTERPRETATION OF STUDY FINDINGS: PART I Julie E. Buring, ScD Harvard School of Public Health Boston, MA Drawing Conclusions TRUTH IN THE UNIVERSE Infer TRUTH IN THE STUDY Infer FINDINGS IN THE STUDY Designing
Experimental Design. Terminology. Chusak Okascharoen, MD, PhD September 19 th, Experimental study Clinical trial Randomized controlled trial
 Experimental Design Chusak Okascharoen, MD, PhD September 19 th, 2016 Terminology Experimental study Clinical trial Randomized controlled trial 1 PHASES OF CLINICAL TRIALS Phase I: First-time-in-man studies
Experimental Design Chusak Okascharoen, MD, PhD September 19 th, 2016 Terminology Experimental study Clinical trial Randomized controlled trial 1 PHASES OF CLINICAL TRIALS Phase I: First-time-in-man studies
Is it worth offering cardiovascular disease prevention to the elderly? Prof. Dr. Helmut Gohlke Herz-Zentrum Bad Krozingen, Germany
 Is it worth offering cardiovascular disease prevention to the elderly? Prof. Dr. Helmut Gohlke Herz-Zentrum Bad Krozingen, Germany Is it worth offering cardiovascular disease prevention to the elderly?
Is it worth offering cardiovascular disease prevention to the elderly? Prof. Dr. Helmut Gohlke Herz-Zentrum Bad Krozingen, Germany Is it worth offering cardiovascular disease prevention to the elderly?
Epidemiologic Study Designs. (RCTs)
 Epidemiologic Study Designs Epidemiologic Study Designs Experimental (RCTs) Observational Analytical Descriptive Case-Control Cohort + cross-sectional & ecologic Epidemiologic Study Designs Descriptive
Epidemiologic Study Designs Epidemiologic Study Designs Experimental (RCTs) Observational Analytical Descriptive Case-Control Cohort + cross-sectional & ecologic Epidemiologic Study Designs Descriptive
Is it ever too late for cardiovascular prevention and rehabilitation? Prof. Dr. Helmut Gohlke Herz-Zentrum Bad Krozingen, Germany
 Is it ever too late for cardiovascular prevention and rehabilitation? Prof. Dr. Helmut Gohlke Herz-Zentrum Bad Krozingen, Germany The demographic issue Life expectancy is increasing Patients are getting
Is it ever too late for cardiovascular prevention and rehabilitation? Prof. Dr. Helmut Gohlke Herz-Zentrum Bad Krozingen, Germany The demographic issue Life expectancy is increasing Patients are getting
Top 5 (Topics) Papers In GIM Rocky Mountain ACP Internal Medicine Meeting Raj Padwal November 13, 2008
 Top 5 (Topics) Papers In GIM 2008 Rocky Mountain ACP Internal Medicine Meeting Raj Padwal November 13, 2008 Methods Searched ACPJC/EBM, TOC of top medical journals, MEDSCAPE Best Evidence, consultation
Top 5 (Topics) Papers In GIM 2008 Rocky Mountain ACP Internal Medicine Meeting Raj Padwal November 13, 2008 Methods Searched ACPJC/EBM, TOC of top medical journals, MEDSCAPE Best Evidence, consultation
Is Lower Better for LDL or is there a Sweet Spot
 Is Lower Better for LDL or is there a Sweet Spot ALAN S BROWN MD, FACC FNLA FAHA FASPC DIRECTOR, DIVISION OF CARDIOLOGY ADVOCATE LUTHERAN GENERAL HOSPITAL, PARK RIDGE, ILLINOIS DIRECTOR OF CARDIOLOGY,
Is Lower Better for LDL or is there a Sweet Spot ALAN S BROWN MD, FACC FNLA FAHA FASPC DIRECTOR, DIVISION OF CARDIOLOGY ADVOCATE LUTHERAN GENERAL HOSPITAL, PARK RIDGE, ILLINOIS DIRECTOR OF CARDIOLOGY,
The Healthy User Effect: Ubiquitous and Uncontrollable S. R. Majumdar, MD MPH FRCPC FACP
 The Healthy User Effect: Ubiquitous and Uncontrollable S. R. Majumdar, MD MPH FRCPC FACP Professor of Medicine, Endowed Chair in Patient Health Management, Health Scholar of the Alberta Heritage Foundation,
The Healthy User Effect: Ubiquitous and Uncontrollable S. R. Majumdar, MD MPH FRCPC FACP Professor of Medicine, Endowed Chair in Patient Health Management, Health Scholar of the Alberta Heritage Foundation,
JUPITER NEJM Poll. Panel Discussion: Literature that Should Have an Impact on our Practice: The JUPITER Study
 Panel Discussion: Literature that Should Have an Impact on our Practice: The Study Kaiser COAST 11 th Annual Conference Maui, August 2009 Robert Blumberg, MD, FACC Ralph Brindis, MD, MPH, FACC Primary
Panel Discussion: Literature that Should Have an Impact on our Practice: The Study Kaiser COAST 11 th Annual Conference Maui, August 2009 Robert Blumberg, MD, FACC Ralph Brindis, MD, MPH, FACC Primary
Blood Pressure Targets in Diabetes
 Stockholm, 29 th August 2010 ESC Meeting Blood Pressure Targets in Diabetes Peter M Nilsson, MD, PhD Department of Clinical Sciences University Hospital, Malmö Sweden Studies on BP in DM2 ADVANCE RCT (Lancet
Stockholm, 29 th August 2010 ESC Meeting Blood Pressure Targets in Diabetes Peter M Nilsson, MD, PhD Department of Clinical Sciences University Hospital, Malmö Sweden Studies on BP in DM2 ADVANCE RCT (Lancet
GRADE. Grading of Recommendations Assessment, Development and Evaluation. British Association of Dermatologists April 2018
 GRADE Grading of Recommendations Assessment, Development and Evaluation British Association of Dermatologists April 2018 Previous grading system Level of evidence Strength of recommendation Level of evidence
GRADE Grading of Recommendations Assessment, Development and Evaluation British Association of Dermatologists April 2018 Previous grading system Level of evidence Strength of recommendation Level of evidence
Antihypertensive Trial Design ALLHAT
 1 U.S. Department of Health and Human Services Major Outcomes in High Risk Hypertensive Patients Randomized to Angiotensin-Converting Enzyme Inhibitor or Calcium Channel Blocker vs Diuretic National Institutes
1 U.S. Department of Health and Human Services Major Outcomes in High Risk Hypertensive Patients Randomized to Angiotensin-Converting Enzyme Inhibitor or Calcium Channel Blocker vs Diuretic National Institutes
Systematic Reviews and Meta- Analysis in Kidney Transplantation
 Systematic Reviews and Meta- Analysis in Kidney Transplantation Greg Knoll MD MSc Associate Professor of Medicine Medical Director, Kidney Transplantation University of Ottawa and The Ottawa Hospital KRESCENT
Systematic Reviews and Meta- Analysis in Kidney Transplantation Greg Knoll MD MSc Associate Professor of Medicine Medical Director, Kidney Transplantation University of Ottawa and The Ottawa Hospital KRESCENT
ACR OA Guideline Development Process Knee and Hip
 ACR OA Guideline Development Process Knee and Hip 1. Literature searching frame work Literature searches were developed based on the scenarios. A comprehensive search strategy was used to guide the process
ACR OA Guideline Development Process Knee and Hip 1. Literature searching frame work Literature searches were developed based on the scenarios. A comprehensive search strategy was used to guide the process
Experiences with interim trial monitoring, particularly with early stopped trials
 Experiences with interim trial monitoring, particularly with early stopped trials 1 Robert J Glynn, ScD Divisions of Preventive Medicine and Pharmacoepidemiology & Pharmacoeconomics, Brigham & Women s
Experiences with interim trial monitoring, particularly with early stopped trials 1 Robert J Glynn, ScD Divisions of Preventive Medicine and Pharmacoepidemiology & Pharmacoeconomics, Brigham & Women s
Hypertension targets: sorting out the confusion. Brian Rayner, Division of Nephrology and Hypertension, University of Cape Town
 Hypertension targets: sorting out the confusion Brian Rayner, Division of Nephrology and Hypertension, University of Cape Town Historical Perspective The most famous casualty of this approach was the
Hypertension targets: sorting out the confusion Brian Rayner, Division of Nephrology and Hypertension, University of Cape Town Historical Perspective The most famous casualty of this approach was the
Clinical Research Design and Conduction
 Clinical Research Design and Conduction Study Design M Mohsen Ibrahim, MD Cardiac Department Cairo University Algorithm for classification of types of clinical research Grimes DA and Schulz KF. Lancet
Clinical Research Design and Conduction Study Design M Mohsen Ibrahim, MD Cardiac Department Cairo University Algorithm for classification of types of clinical research Grimes DA and Schulz KF. Lancet
GLOSSARY OF GENERAL TERMS
 GLOSSARY OF GENERAL TERMS Absolute risk reduction Absolute risk reduction (ARR) is the difference between the event rate in the control group (CER) and the event rate in the treated group (EER). ARR =
GLOSSARY OF GENERAL TERMS Absolute risk reduction Absolute risk reduction (ARR) is the difference between the event rate in the control group (CER) and the event rate in the treated group (EER). ARR =
CVD risk assessment using risk scores in primary and secondary prevention
 CVD risk assessment using risk scores in primary and secondary prevention Raul D. Santos MD, PhD Heart Institute-InCor University of Sao Paulo Brazil Disclosure Honoraria for consulting and speaker activities
CVD risk assessment using risk scores in primary and secondary prevention Raul D. Santos MD, PhD Heart Institute-InCor University of Sao Paulo Brazil Disclosure Honoraria for consulting and speaker activities
Varenicline and cardiovascular and neuropsychiatric events: Do Benefits outweigh risks?
 Varenicline and cardiovascular and neuropsychiatric events: Do Benefits outweigh risks? Sonal Singh M.D., M.P.H, Johns Hopkins University Presented by: Sonal Singh, MD MPH September 19, 2012 1 CONFLICTS
Varenicline and cardiovascular and neuropsychiatric events: Do Benefits outweigh risks? Sonal Singh M.D., M.P.H, Johns Hopkins University Presented by: Sonal Singh, MD MPH September 19, 2012 1 CONFLICTS
Biases in clinical research. Seungho Ryu, MD, PhD Kanguk Samsung Hospital, Sungkyunkwan University
 Biases in clinical research Seungho Ryu, MD, PhD Kanguk Samsung Hospital, Sungkyunkwan University Learning objectives Understand goal of measurement and definition of accuracy Describe the threats to causal
Biases in clinical research Seungho Ryu, MD, PhD Kanguk Samsung Hospital, Sungkyunkwan University Learning objectives Understand goal of measurement and definition of accuracy Describe the threats to causal
Observations on US CVD Prevention Guidelines. Donald M. Lloyd-Jones, MD ScM FACC FAHA
 Observations on US CVD Prevention Guidelines Donald M. Lloyd-Jones, MD ScM FACC FAHA What are Guidelines? Evidence Base for Guidelines Tricoci, JAMA 2009 Evidence Base for Guidelines Tricoci, JAMA 2009
Observations on US CVD Prevention Guidelines Donald M. Lloyd-Jones, MD ScM FACC FAHA What are Guidelines? Evidence Base for Guidelines Tricoci, JAMA 2009 Evidence Base for Guidelines Tricoci, JAMA 2009
Recent developments for combining evidence within evidence streams: bias-adjusted meta-analysis
 EFSA/EBTC Colloquium, 25 October 2017 Recent developments for combining evidence within evidence streams: bias-adjusted meta-analysis Julian Higgins University of Bristol 1 Introduction to concepts Standard
EFSA/EBTC Colloquium, 25 October 2017 Recent developments for combining evidence within evidence streams: bias-adjusted meta-analysis Julian Higgins University of Bristol 1 Introduction to concepts Standard
Making comparisons. Previous sessions looked at how to describe a single group of subjects However, we are often interested in comparing two groups
 Making comparisons Previous sessions looked at how to describe a single group of subjects However, we are often interested in comparing two groups Data can be interpreted using the following fundamental
Making comparisons Previous sessions looked at how to describe a single group of subjects However, we are often interested in comparing two groups Data can be interpreted using the following fundamental
Diabetes Mellitus: A Cardiovascular Disease
 Diabetes Mellitus: A Cardiovascular Disease Nestoras Mathioudakis, M.D. Assistant Professor of Medicine Division of Endocrinology, Diabetes, & Metabolism September 30, 2013 1 The ABCs of cardiovascular
Diabetes Mellitus: A Cardiovascular Disease Nestoras Mathioudakis, M.D. Assistant Professor of Medicine Division of Endocrinology, Diabetes, & Metabolism September 30, 2013 1 The ABCs of cardiovascular
Glossary of Practical Epidemiology Concepts
 Glossary of Practical Epidemiology Concepts - 2009 Adapted from the McMaster EBCP Workshop 2003, McMaster University, Hamilton, Ont. Note that open access to the much of the materials used in the Epi-546
Glossary of Practical Epidemiology Concepts - 2009 Adapted from the McMaster EBCP Workshop 2003, McMaster University, Hamilton, Ont. Note that open access to the much of the materials used in the Epi-546
Clinical and Economic Value of Rivaroxaban in Coronary Artery Disease
 CHRISTOPHER B. GRANGER, MD Professor of Medicine Division of Cardiology, Department of Medicine; Director, Cardiac Care Unit Duke University Medical Center, Durham, NC Clinical and Economic Value of Rivaroxaban
CHRISTOPHER B. GRANGER, MD Professor of Medicine Division of Cardiology, Department of Medicine; Director, Cardiac Care Unit Duke University Medical Center, Durham, NC Clinical and Economic Value of Rivaroxaban
Clinical Epidemiology for the uninitiated
 Clinical epidemiologist have one foot in clinical care and the other in clinical practice research. As clinical epidemiologists we apply a wide array of scientific principles, strategies and tactics to
Clinical epidemiologist have one foot in clinical care and the other in clinical practice research. As clinical epidemiologists we apply a wide array of scientific principles, strategies and tactics to
Presentation Outline. Introduction to Biomedical Research Designs. EBM: A Practical Definition. Grade the Evidence (Example McMaster Grading System)
 Presentation Outline Introduction to Biomedical Research Designs Sean D. Sullivan, R.Ph., PhD Professor of Pharmacy, Public Health and Medicine University of Washington Why a course in Biomedical Research
Presentation Outline Introduction to Biomedical Research Designs Sean D. Sullivan, R.Ph., PhD Professor of Pharmacy, Public Health and Medicine University of Washington Why a course in Biomedical Research
Overview of Study Designs in Clinical Research
 Overview of Study Designs in Clinical Research Systematic Reviews (SR), Meta-Analysis Best Evidence / Evidence Guidelines + Evidence Summaries Randomized, controlled trials (RCT) Clinical trials, Cohort
Overview of Study Designs in Clinical Research Systematic Reviews (SR), Meta-Analysis Best Evidence / Evidence Guidelines + Evidence Summaries Randomized, controlled trials (RCT) Clinical trials, Cohort
Determinants of quality: Factors that lower or increase the quality of evidence
 Determinants of quality: Factors that lower or increase the quality of evidence GRADE Workshop CBO, NHG and Dutch Cochrane Centre CBO, April 17th, 2013 Outline The GRADE approach: step by step Factors
Determinants of quality: Factors that lower or increase the quality of evidence GRADE Workshop CBO, NHG and Dutch Cochrane Centre CBO, April 17th, 2013 Outline The GRADE approach: step by step Factors
Case-ControlControl. None
 Study Designs & Methodologies (w/ /M Measures of Association) What is the most useful and appropriate study design? Larry W. Segars, PharmD, DrPH, FCCP, FACE, BCPS, RPh Associate Dean College of Biosciences
Study Designs & Methodologies (w/ /M Measures of Association) What is the most useful and appropriate study design? Larry W. Segars, PharmD, DrPH, FCCP, FACE, BCPS, RPh Associate Dean College of Biosciences
ESC Geoffrey Rose Lecture on Population Sciences Cholesterol and risk: past, present and future
 ESC Geoffrey Rose Lecture on Population Sciences Cholesterol and risk: past, present and future Rory Collins BHF Professor of Medicine & Epidemiology Clinical Trial Service Unit & Epidemiological Studies
ESC Geoffrey Rose Lecture on Population Sciences Cholesterol and risk: past, present and future Rory Collins BHF Professor of Medicine & Epidemiology Clinical Trial Service Unit & Epidemiological Studies
Oral Hypoglycemics and Risk of Adverse Cardiac Events: A Summary of the Controversy
 Oral Hypoglycemics and Risk of Adverse Cardiac Events: A Summary of the Controversy Jeffrey Boord, MD, MPH Advances in Cardiovascular Medicine Kingston, Jamaica December 7, 2012 VanderbiltHeart.com Outline
Oral Hypoglycemics and Risk of Adverse Cardiac Events: A Summary of the Controversy Jeffrey Boord, MD, MPH Advances in Cardiovascular Medicine Kingston, Jamaica December 7, 2012 VanderbiltHeart.com Outline
The earlier BP control the better cardiovascular outcome. Jin Oh Na Cardiovascular center Korea University Medical College
 The earlier BP control the better cardiovascular outcome Jin Oh Na Cardiovascular center Korea University Medical College Index Introduction HOPE-3 Trial Sprint Study Summary Each 2 mmhg decrease in SBP
The earlier BP control the better cardiovascular outcome Jin Oh Na Cardiovascular center Korea University Medical College Index Introduction HOPE-3 Trial Sprint Study Summary Each 2 mmhg decrease in SBP
Meta Analysis. David R Urbach MD MSc Outcomes Research Course December 4, 2014
 Meta Analysis David R Urbach MD MSc Outcomes Research Course December 4, 2014 Overview Definitions Identifying studies Appraising studies Quantitative synthesis Presentation of results Examining heterogeneity
Meta Analysis David R Urbach MD MSc Outcomes Research Course December 4, 2014 Overview Definitions Identifying studies Appraising studies Quantitative synthesis Presentation of results Examining heterogeneity
CLINICAL DECISION USING AN ARTICLE ABOUT TREATMENT JOSEFINA S. ISIDRO LAPENA MD, MFM, FPAFP PROFESSOR, UPCM
 CLINICAL DECISION USING AN ARTICLE ABOUT TREATMENT JOSEFINA S. ISIDRO LAPENA MD, MFM, FPAFP PROFESSOR, UPCM Principles of Decision Making Knowing the patient s true state is often unnecessary Does my patient
CLINICAL DECISION USING AN ARTICLE ABOUT TREATMENT JOSEFINA S. ISIDRO LAPENA MD, MFM, FPAFP PROFESSOR, UPCM Principles of Decision Making Knowing the patient s true state is often unnecessary Does my patient
Systematic reviews and meta-analyses of observational studies (MOOSE): Checklist.
 Systematic reviews and meta-analyses of observational studies (MOOSE): Checklist. MOOSE Checklist Infliximab reduces hospitalizations and surgery interventions in patients with inflammatory bowel disease:
Systematic reviews and meta-analyses of observational studies (MOOSE): Checklist. MOOSE Checklist Infliximab reduces hospitalizations and surgery interventions in patients with inflammatory bowel disease:
Statistical Essentials in Interpreting Clinical Trials Stuart J. Pocock, PhD
 Statistical Essentials in Interpreting Clinical Trials Stuart J. Pocock, PhD June 03, 2016 www.medscape.com I'm Stuart Pocock, professor of medical statistics at London University. I am going to take you
Statistical Essentials in Interpreting Clinical Trials Stuart J. Pocock, PhD June 03, 2016 www.medscape.com I'm Stuart Pocock, professor of medical statistics at London University. I am going to take you
Rapid appraisal of the literature: Identifying study biases
 Rapid appraisal of the literature: Identifying study biases Rita Popat, PhD Clinical Assistant Professor Division of Epidemiology Stanford University School of Medicine August 7, 2007 What is critical
Rapid appraisal of the literature: Identifying study biases Rita Popat, PhD Clinical Assistant Professor Division of Epidemiology Stanford University School of Medicine August 7, 2007 What is critical
LDL cholesterol and cardiovascular outcomes?
 LDL cholesterol and cardiovascular outcomes? Prof Kausik Ray, BSc (hons), MBChB, FRCP, MD, MPhil (Cantab), FACC, FESC Professor of Cardiovascular Disease Prevention St Georges University of London Honorary
LDL cholesterol and cardiovascular outcomes? Prof Kausik Ray, BSc (hons), MBChB, FRCP, MD, MPhil (Cantab), FACC, FESC Professor of Cardiovascular Disease Prevention St Georges University of London Honorary
Results. NeuRA Treatments for dual diagnosis August 2016
 Introduction Many treatments have been targeted to improving symptom severity for people suffering schizophrenia in combination with substance use problems. Studies of dual diagnosis often investigate
Introduction Many treatments have been targeted to improving symptom severity for people suffering schizophrenia in combination with substance use problems. Studies of dual diagnosis often investigate
Using DOACs in CAD Patients in Sinus Ryhthm Results of the ATLAS ACS 2, COMPASS and COMMANDER-HF Trials
 Using DOACs in CAD Patients in Sinus Ryhthm Results of the ATLAS ACS 2, COMPASS and COMMANDER-HF Trials 19 th Annual San Diego Heart Failure Symposium for Primary Care Physicians January 11-12, 2019 La
Using DOACs in CAD Patients in Sinus Ryhthm Results of the ATLAS ACS 2, COMPASS and COMMANDER-HF Trials 19 th Annual San Diego Heart Failure Symposium for Primary Care Physicians January 11-12, 2019 La
Evidence Based Medicine
 Hamadan University of medical sciences School of Public Health Department of Epidemiology Evidence Based Medicine Amin Doosti-Irani, PhD in Epidemiology 10 March 2017 a_doostiirani@yahoo.com 1 Outlines
Hamadan University of medical sciences School of Public Health Department of Epidemiology Evidence Based Medicine Amin Doosti-Irani, PhD in Epidemiology 10 March 2017 a_doostiirani@yahoo.com 1 Outlines
Is There An Association?
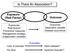 Is There An Association? Exposure (Risk Factor) Outcome Exposures Risk factors Preventive measures Management strategy Independent variables Outcomes Dependent variable Disease occurrence Lack of exercise
Is There An Association? Exposure (Risk Factor) Outcome Exposures Risk factors Preventive measures Management strategy Independent variables Outcomes Dependent variable Disease occurrence Lack of exercise
CONSORT 2010 checklist of information to include when reporting a randomised trial*
 CONSORT 2010 checklist of information to include when reporting a randomised trial* Section/Topic Title and abstract Introduction Background and objectives Item No Checklist item 1a Identification as a
CONSORT 2010 checklist of information to include when reporting a randomised trial* Section/Topic Title and abstract Introduction Background and objectives Item No Checklist item 1a Identification as a
How to Make Sense of Statistics Reported in the Medical Literature
 How to Make Sense of Statistics Reported in the Medical Literature Colin P. West, MD, PhD Division of General Internal Medicine Division of Biomedical Statistics and Informatics Denise M. Dupras, MD, PhD
How to Make Sense of Statistics Reported in the Medical Literature Colin P. West, MD, PhD Division of General Internal Medicine Division of Biomedical Statistics and Informatics Denise M. Dupras, MD, PhD
GRADE. Grading of Recommendations Assessment, Development and Evaluation. British Association of Dermatologists April 2014
 GRADE Grading of Recommendations Assessment, Development and Evaluation British Association of Dermatologists April 2014 Previous grading system Level of evidence Strength of recommendation Level of evidence
GRADE Grading of Recommendations Assessment, Development and Evaluation British Association of Dermatologists April 2014 Previous grading system Level of evidence Strength of recommendation Level of evidence
Types of Biomedical Research
 INTRODUCTION & MEASUREMENT IN CLINICAL RESEARCH Sakda Arj Ong Vallipakorn, MD MSIT, MA (Information Science) Pediatrics, Pediatric Cardiology Emergency Medicine, Ped Emergency Family Medicine Section of
INTRODUCTION & MEASUREMENT IN CLINICAL RESEARCH Sakda Arj Ong Vallipakorn, MD MSIT, MA (Information Science) Pediatrics, Pediatric Cardiology Emergency Medicine, Ped Emergency Family Medicine Section of
Statistical Methods in Medical Research
 Department of Statistics Western Michigan University Outline Surveys of Methodology 1 Survey of statistical methodology 2 Survey of contradicted studies 3 Threats to correct outcomes 1979 Methods Survey
Department of Statistics Western Michigan University Outline Surveys of Methodology 1 Survey of statistical methodology 2 Survey of contradicted studies 3 Threats to correct outcomes 1979 Methods Survey
12/26/2013. Types of Biomedical Research. Clinical Research. 7Steps to do research INTRODUCTION & MEASUREMENT IN CLINICAL RESEARCH S T A T I S T I C
 Types of Biomedical Research INTRODUCTION & MEASUREMENT IN CLINICAL RESEARCH Sakda Arj Ong Vallipakorn, MD MSIT, MA (Information Science) Pediatrics, Pediatric Cardiology Emergency Medicine, Ped Emergency
Types of Biomedical Research INTRODUCTION & MEASUREMENT IN CLINICAL RESEARCH Sakda Arj Ong Vallipakorn, MD MSIT, MA (Information Science) Pediatrics, Pediatric Cardiology Emergency Medicine, Ped Emergency
EBM, Study Design and Numbers. David Frankfurter, MD Professor OB/GYN The George Washington University
 EBM, Study Design and Numbers David Frankfurter, MD Professor OB/GYN The George Washington University 1978? 1978 Best Picture of 1978 The Deer Hunter Universal Studios The 50th Academy Awards were held
EBM, Study Design and Numbers David Frankfurter, MD Professor OB/GYN The George Washington University 1978? 1978 Best Picture of 1978 The Deer Hunter Universal Studios The 50th Academy Awards were held
HARM. Definition modified from the IHI definition of Harm by the QUEST Harm Workgroup
 Indonesia Clinical epidemiology and Evidence Based Medicine (ICE(ICE-EBM) HARM Jarir At Thobari (FK UGM) Efficacious Medical intervention Harm HARM Unintended physical injury resulting from or contributed
Indonesia Clinical epidemiology and Evidence Based Medicine (ICE(ICE-EBM) HARM Jarir At Thobari (FK UGM) Efficacious Medical intervention Harm HARM Unintended physical injury resulting from or contributed
Providing High Value Cost-Conscious Care:
 Providing High Value Cost-Conscious Care: Biostatistical Concepts You Need to Know 2012-2013 Presentation #5 0f 10 http://hvc.acponline.org/ Learning Objectives Understand that a working knowledge of basic
Providing High Value Cost-Conscious Care: Biostatistical Concepts You Need to Know 2012-2013 Presentation #5 0f 10 http://hvc.acponline.org/ Learning Objectives Understand that a working knowledge of basic
T. Suithichaiyakul Cardiomed Chula
 T. Suithichaiyakul Cardiomed Chula The cardiovascular (CV) continuum: role of risk factors Endothelial Dysfunction Atherosclerosis and left ventricular hypertrophy Myocardial infarction & stroke Endothelial
T. Suithichaiyakul Cardiomed Chula The cardiovascular (CV) continuum: role of risk factors Endothelial Dysfunction Atherosclerosis and left ventricular hypertrophy Myocardial infarction & stroke Endothelial
Treatment of Cardiovascular Risk Factors. Kevin M Hayes D.O. F.A.C.C. First Coast Heart and Vascular Center
 Treatment of Cardiovascular Risk Factors Kevin M Hayes D.O. F.A.C.C. First Coast Heart and Vascular Center Disclosures: None Objectives What do risk factors tell us What to check and when Does treatment
Treatment of Cardiovascular Risk Factors Kevin M Hayes D.O. F.A.C.C. First Coast Heart and Vascular Center Disclosures: None Objectives What do risk factors tell us What to check and when Does treatment
Dyslipidemia in women: Who should be treated and how?
 Dyslipidemia in women: Who should be treated and how? Lale Tokgozoglu, MD, FACC, FESC Professor of Cardiology Hacettepe University Faculty of Medicine Ankara, Turkey. Cause of Death in Women: European
Dyslipidemia in women: Who should be treated and how? Lale Tokgozoglu, MD, FACC, FESC Professor of Cardiology Hacettepe University Faculty of Medicine Ankara, Turkey. Cause of Death in Women: European
ALLHAT Role of Diuretics in the Prevention of Heart Failure - The Antihypertensive and Lipid- Lowering Treatment to Prevent Heart Attack Trial
 1 ALLHAT Role of Diuretics in the Prevention of Heart Failure - The Antihypertensive and Lipid- Lowering Treatment to Prevent Heart Attack Trial Davis BR, Piller LB, Cutler JA, et al. Circulation 2006.113:2201-2210.
1 ALLHAT Role of Diuretics in the Prevention of Heart Failure - The Antihypertensive and Lipid- Lowering Treatment to Prevent Heart Attack Trial Davis BR, Piller LB, Cutler JA, et al. Circulation 2006.113:2201-2210.
Generalizing the right question, which is?
 Generalizing RCT results to broader populations IOM Workshop Washington, DC, April 25, 2013 Generalizing the right question, which is? Miguel A. Hernán Harvard School of Public Health Observational studies
Generalizing RCT results to broader populations IOM Workshop Washington, DC, April 25, 2013 Generalizing the right question, which is? Miguel A. Hernán Harvard School of Public Health Observational studies
Epidemiologic study designs
 Epidemiologic study designs and critical appraisal of scientific papers Rolf H.H. Groenwold, MD, PhD Bio sketch MD, PhD in epidemiology Associate professor of Epidemiology at UMC Utrecht Research focus
Epidemiologic study designs and critical appraisal of scientific papers Rolf H.H. Groenwold, MD, PhD Bio sketch MD, PhD in epidemiology Associate professor of Epidemiology at UMC Utrecht Research focus
Evidence- and Value-based Solutions for Health Care Clinical Improvement Consults, Content Development, Training & Seminars, Tools
 Definition Key Points Key Problems Bias Choice Lack of Control Chance Observational Study Defined Epidemiological study in which observations are made, but investigators do not control the exposure or
Definition Key Points Key Problems Bias Choice Lack of Control Chance Observational Study Defined Epidemiological study in which observations are made, but investigators do not control the exposure or
Anatomy of Medical Studies: Part I. Emily D Agostino Epidemiology and Biostatistics CUNY School of Public Health at Hunter College
 Anatomy of Medical Studies: Part I!! Emily D Agostino Epidemiology and Biostatistics CUNY School of Public Health at Hunter College I. Standard format of a Medical Study Abstract (quick overview: what
Anatomy of Medical Studies: Part I!! Emily D Agostino Epidemiology and Biostatistics CUNY School of Public Health at Hunter College I. Standard format of a Medical Study Abstract (quick overview: what
9/29/2015. Primary Prevention of Heart Disease: Objectives. Objectives. What works? What doesn t?
 Primary Prevention of Heart Disease: What works? What doesn t? Samia Mora, MD, MHS Associate Professor, Harvard Medical School Associate Physician, Brigham and Women s Hospital October 2, 2015 Financial
Primary Prevention of Heart Disease: What works? What doesn t? Samia Mora, MD, MHS Associate Professor, Harvard Medical School Associate Physician, Brigham and Women s Hospital October 2, 2015 Financial
Cardiovascular Risk of Celecoxib in 6 Randomized Placebo-controlled Trials: The Cross Trial Safety Analysis
 Cardiovascular Risk of Celecoxib in 6 Randomized Placebo-controlled Trials: The Cross Trial Safety Analysis Scott D. Solomon, MD, Janet Wittes, PhD, Ernest Hawk, MD, MPH for the Celecoxib Cross Trials
Cardiovascular Risk of Celecoxib in 6 Randomized Placebo-controlled Trials: The Cross Trial Safety Analysis Scott D. Solomon, MD, Janet Wittes, PhD, Ernest Hawk, MD, MPH for the Celecoxib Cross Trials
Biases in clinical research. Seungho Ryu, MD, PhD Kanguk Samsung Hospital, Sungkyunkwan University
 Biases in clinical research Seungho Ryu, MD, PhD Kanguk Samsung Hospital, Sungkyunkwan University Learning objectives Describe the threats to causal inferences in clinical studies Understand the role of
Biases in clinical research Seungho Ryu, MD, PhD Kanguk Samsung Hospital, Sungkyunkwan University Learning objectives Describe the threats to causal inferences in clinical studies Understand the role of
No relevant financial relationships
 MANAGEMENT OF LIPID DISORDERS Balancing Benefits and harms Disclosure Robert B. Baron, MD MS Professor and Associate Dean UCSF School of Medicine No relevant financial relationships baron@medicine.ucsf.edu
MANAGEMENT OF LIPID DISORDERS Balancing Benefits and harms Disclosure Robert B. Baron, MD MS Professor and Associate Dean UCSF School of Medicine No relevant financial relationships baron@medicine.ucsf.edu
Online Supplementary Material
 Section 1. Adapted Newcastle-Ottawa Scale The adaptation consisted of allowing case-control studies to earn a star when the case definition is based on record linkage, to liken the evaluation of case-control
Section 1. Adapted Newcastle-Ottawa Scale The adaptation consisted of allowing case-control studies to earn a star when the case definition is based on record linkage, to liken the evaluation of case-control
What have We Learned in Dyslipidemia Management Since the Publication of the 2013 ACC/AHA Guideline?
 What have We Learned in Dyslipidemia Management Since the Publication of the 2013 ACC/AHA Guideline? Salim S. Virani, MD, PhD, FACC, FAHA Associate Professor, Section of Cardiovascular Research Baylor
What have We Learned in Dyslipidemia Management Since the Publication of the 2013 ACC/AHA Guideline? Salim S. Virani, MD, PhD, FACC, FAHA Associate Professor, Section of Cardiovascular Research Baylor
Confounding and Bias
 28 th International Conference on Pharmacoepidemiology and Therapeutic Risk Management Barcelona, Spain August 22, 2012 Confounding and Bias Tobias Gerhard, PhD Assistant Professor, Ernest Mario School
28 th International Conference on Pharmacoepidemiology and Therapeutic Risk Management Barcelona, Spain August 22, 2012 Confounding and Bias Tobias Gerhard, PhD Assistant Professor, Ernest Mario School
Calculating RR, ARR, NNT
 Calculating RR, ARR, NNT In a trial RR = Event rate (eg # of people with one stroke/ total people) in treatment group/event rate in the control group. ARR = Event rate in control group minus the event
Calculating RR, ARR, NNT In a trial RR = Event rate (eg # of people with one stroke/ total people) in treatment group/event rate in the control group. ARR = Event rate in control group minus the event
Health Studies 315. Clinical Epidemiology: Evidence of Risk and Harm
 Health Studies 315 Clinical Epidemiology: Evidence of Risk and Harm 1 Patients encounter (possibly) risky exposures Alcohol during pregnancy (fetal risk) Electromagnetic fields (cancer risk) Vasectomy
Health Studies 315 Clinical Epidemiology: Evidence of Risk and Harm 1 Patients encounter (possibly) risky exposures Alcohol during pregnancy (fetal risk) Electromagnetic fields (cancer risk) Vasectomy
Research Methodology Workshop. Study Type
 Research Methodology Workshop Study Type What type of question? Frequency: how common is it? Aetiology: What caused this? Intervention: Does this intervention work? Diagnosis or test evaluation: How accurate
Research Methodology Workshop Study Type What type of question? Frequency: how common is it? Aetiology: What caused this? Intervention: Does this intervention work? Diagnosis or test evaluation: How accurate
Placebo-Controlled Statin Trials
 PREVENTION OF CHD WITH LIPID MANAGEMENT AND ASPIRIN: MATCHING TREATMENT TO RISK Robert B. Baron MD MS Professor and Associate Dean UCSF School of Medicine Declaration of full disclosure: No conflict of
PREVENTION OF CHD WITH LIPID MANAGEMENT AND ASPIRIN: MATCHING TREATMENT TO RISK Robert B. Baron MD MS Professor and Associate Dean UCSF School of Medicine Declaration of full disclosure: No conflict of
Results. NeuRA Motor dysfunction April 2016
 Introduction Subtle deviations in various developmental trajectories during childhood and adolescence may foreshadow the later development of schizophrenia. Studies exploring these deviations (antecedents)
Introduction Subtle deviations in various developmental trajectories during childhood and adolescence may foreshadow the later development of schizophrenia. Studies exploring these deviations (antecedents)
Misperceptions still exist that cardiovascular disease is not a real problem for women.
 Management of Cardiovascular Risk Factors in the Cynthia A., MD University of California, San Diego ARHP 9/19/08 Disclosures Research support Wyeth, Lilly, Organon, Novo Nordisk, Pfizer Consultant fees
Management of Cardiovascular Risk Factors in the Cynthia A., MD University of California, San Diego ARHP 9/19/08 Disclosures Research support Wyeth, Lilly, Organon, Novo Nordisk, Pfizer Consultant fees
8/18/10. Disclosure. Objectives. A Pervasive Concern. Evidence Based Medicine, Family Physicians, and Knowledge Translation (KATIE)
 Evidence Based Medicine, Family Physicians, and Knowledge Translation (KATIE) David Gardner PharmD, MSc Michael Allen MD Dalhousie Refresher February 2010 Disclosure David Gardner Research/ Department
Evidence Based Medicine, Family Physicians, and Knowledge Translation (KATIE) David Gardner PharmD, MSc Michael Allen MD Dalhousie Refresher February 2010 Disclosure David Gardner Research/ Department
All medications are a double-edged sword with risks
 Menopause: The Journal of The North American Menopause Society Vol. 14, No. 5, pp. 1/14 DOI: 10.1097/gme.0b013e31802e8508 * 2007 by The North American Menopause Society REVIEW ARTICLE Postmenopausal hormone
Menopause: The Journal of The North American Menopause Society Vol. 14, No. 5, pp. 1/14 DOI: 10.1097/gme.0b013e31802e8508 * 2007 by The North American Menopause Society REVIEW ARTICLE Postmenopausal hormone
Treating Hypertension in 2018: What Makes the Most Sense Today?
 Treating Hypertension in 2018: What Makes the Most Sense Today? Daniel Blanchard, MD Professor of Medicine UC San Diego Cardiovascular Center La Jolla, California 1 2 Speaker Disclosures Consultant and/or
Treating Hypertension in 2018: What Makes the Most Sense Today? Daniel Blanchard, MD Professor of Medicine UC San Diego Cardiovascular Center La Jolla, California 1 2 Speaker Disclosures Consultant and/or
Meta-Analysis. Zifei Liu. Biological and Agricultural Engineering
 Meta-Analysis Zifei Liu What is a meta-analysis; why perform a metaanalysis? How a meta-analysis work some basic concepts and principles Steps of Meta-analysis Cautions on meta-analysis 2 What is Meta-analysis
Meta-Analysis Zifei Liu What is a meta-analysis; why perform a metaanalysis? How a meta-analysis work some basic concepts and principles Steps of Meta-analysis Cautions on meta-analysis 2 What is Meta-analysis
Purpose. Study Designs. Objectives. Observational Studies. Analytic Studies
 Purpose Study Designs H.S. Teitelbaum, DO, PhD, MPH, FAOCOPM AOCOPM Annual Meeting Introduce notions of study design Clarify common terminology used with description and interpretation of information collected
Purpose Study Designs H.S. Teitelbaum, DO, PhD, MPH, FAOCOPM AOCOPM Annual Meeting Introduce notions of study design Clarify common terminology used with description and interpretation of information collected
Statistics for Clinical Trials: Basics of Phase III Trial Design
 Statistics for Clinical Trials: Basics of Phase III Trial Design Gary M. Clark, Ph.D. Vice President Biostatistics & Data Management Array BioPharma Inc. Boulder, Colorado USA NCIC Clinical Trials Group
Statistics for Clinical Trials: Basics of Phase III Trial Design Gary M. Clark, Ph.D. Vice President Biostatistics & Data Management Array BioPharma Inc. Boulder, Colorado USA NCIC Clinical Trials Group
The Clinical Unmet need in the patient with Diabetes and ACS
 The Clinical Unmet need in the patient with Diabetes and ACS Professor Kausik Ray (UK) BSc(hons), MBChB, MD, MPhil, FRCP (lon), FRCP (ed), FACC, FESC, FAHA Diabetes is a global public health challenge
The Clinical Unmet need in the patient with Diabetes and ACS Professor Kausik Ray (UK) BSc(hons), MBChB, MD, MPhil, FRCP (lon), FRCP (ed), FACC, FESC, FAHA Diabetes is a global public health challenge
