Department of OUTCOMES RESEARCH
|
|
|
- Curtis Townsend
- 6 years ago
- Views:
Transcription
1 Department of OUTCOMES RESEARCH
2 Clinical Research Design Sources of Error Types of Clinical Research Randomized Trials! Daniel I. Sessler, M.D. Professor and Chair Department of OUTCOMES RESEARCH The Cleveland Clinic!
3 Sources of Error There is no perfect study All are limited by practical and ethical considerations It is impossible to control all potential confounders Multiple studies required to prove a hypothesis Good design limits risk of false results Statistics at best partially compensate for systematic error Major types of error Selection bias Measurement bias Confounding Reverse causation Chance
4 Statistical Association Chance Causal A B Causal B A Statistical Association Selection Bias Measurement Bias Confounding Bias
5 Selection Bias Non-random selection for inclusion / treatment Or selective loss Subtle forms of disease may be missed When treatment is non-random: Newer treatments assigned to patients most likely to benefit Better patients seek out latest treatments Nice patients may be given the preferred treatment Compliance may vary as a function of treatment Patients drop out for lack of efficacy or because of side effects Largely prevented by randomization
6 Confounding Association between two factors caused by third factor For example: Transfusions are associated with high mortality But larger, longer operations require more blood Increased mortality consequent to larger operations Another example: Mortality greater in Florida than Alaska But average age is much higher in Florida Increased mortality from age, rather than geography of FL Largely prevented by randomization
7 Measurement Bias Quality of measurement varies non-randomly Quality of records generally poor Not necessarily randomly so Patients given new treatments watched more closely Subjects with disease may better remember exposures When treatment is unblinded Benefit may be over-estimated Complications may be under-estimated Largely prevented by blinding
8 Example of Measurement Bias Reported parental history Arthritis (%) No arthritis (%) Neither parent One parent Both parents 15 8 P = 0.003! From Schull & Cobb, J Chronic Dis, 1969!
9 Types of Clinical Research Observational Case series Implicit historical control The pleural of anecdote is not data Single cohort (natural history) Retrospective cohort Case-control Retrospective versus prospective Prospective data usually of higher quality Randomized clinical trial Strongest design; gold standard First major example: use of streptomycin for TB in 1948
10 Case-Control Studies Identify cases & matched controls Look back in time and compare on exposure E x p o s u r e! Time! Case Group! Control Group!
11 Cohort Studies Identify exposed & matched unexposed patients Look forward in time and compare on disease Exposed! Unexposed! D i s e a s e! Time!
12 Randomized Clinical Trials (RCTs) A type of prospective cohort study Best protection again bias and confounding Randomization: reduces selection bias & confounding Blinding: reduces measurement error Not subject to reverse causation RCTs often correct observational results Types Parallel group Cross-over Factorial Cluster
13 Factorial Trials Simultaneously test 2 or more interventions Clonidine vs. Placebo Clonidine +ASA Clonidine + Placebo Placebo + ASA Placebo + Placebo ASA vs. Placebo Clonidine +ASA Clonidine + Placebo Placebo + ASA Placebo + Placebo
14 Pros & Cons Advantages More efficient than separate trials Can test for interactions Disadvantages Complexity, potential for reduced compliance Reduces fraction of eligible subjects and enrollment Rarely powered for interactions But interactions influence sample size requirements
15 Randomization and Allocation Only reliable protection against Selection bias Confounding Concealed allocation Independent of investigators Unpredictable Methods Computer-controlled Random-block Envelopes, web-accessed, telephone Stratification Rarely necessary
16 Blinding / Masking Only reliable prevention for measurement bias Essential for subjective responses Use for objective responses whenever possible Careful design required to maintain blinding Potential groups to blind Patients Providers Investigators, including data collection & adjudicators Maintain blinding throughout data analysis Even data-entry errors can be non-random Statisticians are not immune to bias! Placebo effect can be enormous
17 Selection of Outcomes Surrogate or intermediate May not actually relate to outcomes of interest Bone density for fractures Intraoperative hypotension for stroke Usually continuous: implies smaller sample size Rarely powered for complications Major outcomes Severe events (i.e., myocardial infarction, stroke) Usual dichotomous: implies larger sample size Mortality Cost effectiveness / cost utility Quality-of-life
18 Composite Outcomes Any of 2 component outcomes, for example: Cardiac death, myocardial infarction, or non-fatal arrest Wound infection, anastomotic leak, abscess, or sepsis Usually permits a smaller sample size Incidence of each should be comparable Otherwise common outcome(s) dominate composite Severity of each should be comparable Unreasonable to lump minor and major events Death often included to prevent survivor bias Beware of heterogeneous results
19 Outcomes Approaches
20 Interim Analyses & Stopping Rules Reasons trials are stopped early Ethics Money Regulatory issues Drug expiration Personnel Other opportunities Pre-defined interim analyses Spend alpha and beta power Avoid convenience sample Avoid looking between scheduled analyses Pre-defined stopping rules Efficacy versus futility
21 Conclusion: Good Clinical Trials Test a specific a priori hypothesis Evaluate clinically important outcomes Are well designed, with A priori and adequate sample size Defined stopping rules Are randomized and blinded when possible Use appropriate statistical analysis Make conclusions that follow from the data And acknowledged substantive limitations
22 Department of OUTCOMES RESEARCH
Statistics for Clinical Trials: Basics of Phase III Trial Design
 Statistics for Clinical Trials: Basics of Phase III Trial Design Gary M. Clark, Ph.D. Vice President Biostatistics & Data Management Array BioPharma Inc. Boulder, Colorado USA NCIC Clinical Trials Group
Statistics for Clinical Trials: Basics of Phase III Trial Design Gary M. Clark, Ph.D. Vice President Biostatistics & Data Management Array BioPharma Inc. Boulder, Colorado USA NCIC Clinical Trials Group
Randomized Controlled Trial
 Randomized Controlled Trial Training Course in Sexual and Reproductive Health Research Geneva 2016 Dr Khalifa Elmusharaf MBBS, PgDip, FRSPH, PHD Senior Lecturer in Public Health Graduate Entry Medical
Randomized Controlled Trial Training Course in Sexual and Reproductive Health Research Geneva 2016 Dr Khalifa Elmusharaf MBBS, PgDip, FRSPH, PHD Senior Lecturer in Public Health Graduate Entry Medical
CRITICAL APPRAISAL OF MEDICAL LITERATURE. Samuel Iff ISPM Bern
 CRITICAL APPRAISAL OF MEDICAL LITERATURE Samuel Iff ISPM Bern siff@ispm.unibe.ch Contents Study designs Asking good questions Pitfalls in clinical studies How to assess validity (RCT) Conclusion Step-by-step
CRITICAL APPRAISAL OF MEDICAL LITERATURE Samuel Iff ISPM Bern siff@ispm.unibe.ch Contents Study designs Asking good questions Pitfalls in clinical studies How to assess validity (RCT) Conclusion Step-by-step
Protocol Development: The Guiding Light of Any Clinical Study
 Protocol Development: The Guiding Light of Any Clinical Study Susan G. Fisher, Ph.D. Chair, Department of Clinical Sciences 1 Introduction Importance/ relevance/ gaps in knowledge Specific purpose of the
Protocol Development: The Guiding Light of Any Clinical Study Susan G. Fisher, Ph.D. Chair, Department of Clinical Sciences 1 Introduction Importance/ relevance/ gaps in knowledge Specific purpose of the
Experimental Design. Terminology. Chusak Okascharoen, MD, PhD September 19 th, Experimental study Clinical trial Randomized controlled trial
 Experimental Design Chusak Okascharoen, MD, PhD September 19 th, 2016 Terminology Experimental study Clinical trial Randomized controlled trial 1 PHASES OF CLINICAL TRIALS Phase I: First-time-in-man studies
Experimental Design Chusak Okascharoen, MD, PhD September 19 th, 2016 Terminology Experimental study Clinical trial Randomized controlled trial 1 PHASES OF CLINICAL TRIALS Phase I: First-time-in-man studies
Is There An Association?
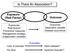 Is There An Association? Exposure (Risk Factor) Outcome Exposures Risk factors Preventive measures Management strategy Independent variables Outcomes Dependent variable Disease occurrence Lack of exercise
Is There An Association? Exposure (Risk Factor) Outcome Exposures Risk factors Preventive measures Management strategy Independent variables Outcomes Dependent variable Disease occurrence Lack of exercise
Overview of Study Designs in Clinical Research
 Overview of Study Designs in Clinical Research Systematic Reviews (SR), Meta-Analysis Best Evidence / Evidence Guidelines + Evidence Summaries Randomized, controlled trials (RCT) Clinical trials, Cohort
Overview of Study Designs in Clinical Research Systematic Reviews (SR), Meta-Analysis Best Evidence / Evidence Guidelines + Evidence Summaries Randomized, controlled trials (RCT) Clinical trials, Cohort
Intervention vs. Observational Trials:
 Intervention vs. Observational Trials: A Brief Introduction Introduction Just like an architect translates a vision for a building into blueprints, so an investigator translates their research idea into
Intervention vs. Observational Trials: A Brief Introduction Introduction Just like an architect translates a vision for a building into blueprints, so an investigator translates their research idea into
Purpose. Study Designs. Objectives. Observational Studies. Analytic Studies
 Purpose Study Designs H.S. Teitelbaum, DO, PhD, MPH, FAOCOPM AOCOPM Annual Meeting Introduce notions of study design Clarify common terminology used with description and interpretation of information collected
Purpose Study Designs H.S. Teitelbaum, DO, PhD, MPH, FAOCOPM AOCOPM Annual Meeting Introduce notions of study design Clarify common terminology used with description and interpretation of information collected
Issues to Consider in the Design of Randomized Controlled Trials
 Issues to Consider in the Design of Randomized Controlled Trials Jay Wilkinson, MD, MPH Professor of Pediatrics & Epidemiology Miller School of Medicine Seminar Purpose To facilitate an interactive discussion
Issues to Consider in the Design of Randomized Controlled Trials Jay Wilkinson, MD, MPH Professor of Pediatrics & Epidemiology Miller School of Medicine Seminar Purpose To facilitate an interactive discussion
Online Appendices Online Appendix 1 Online Appendix 2 Online Appendix 3a Online Appendix 3b Online Appendix 3c Online Appendix 4 Online Appendix 5
 Online Appendices Online Appendix 1 Search terms used to identify studies Online Appendix 2 The identification process for eligible studies Online Appendix 3a- Niacin, Risk of bias table Online Appendix
Online Appendices Online Appendix 1 Search terms used to identify studies Online Appendix 2 The identification process for eligible studies Online Appendix 3a- Niacin, Risk of bias table Online Appendix
Vocabulary. Bias. Blinding. Block. Cluster sample
 Bias Blinding Block Census Cluster sample Confounding Control group Convenience sample Designs Experiment Experimental units Factor Level Any systematic failure of a sampling method to represent its population
Bias Blinding Block Census Cluster sample Confounding Control group Convenience sample Designs Experiment Experimental units Factor Level Any systematic failure of a sampling method to represent its population
Module 5. The Epidemiological Basis of Randomised Controlled Trials. Landon Myer School of Public Health & Family Medicine, University of Cape Town
 Module 5 The Epidemiological Basis of Randomised Controlled Trials Landon Myer School of Public Health & Family Medicine, University of Cape Town Introduction The Randomised Controlled Trial (RCT) is the
Module 5 The Epidemiological Basis of Randomised Controlled Trials Landon Myer School of Public Health & Family Medicine, University of Cape Town Introduction The Randomised Controlled Trial (RCT) is the
SIH: Trials and Research Endeavors. Tim Amrhein, MD Assistant Professor of Neuroradiology Duke University Medical
 SIH: Trials and Research Endeavors Tim Amrhein, MD Assistant Professor of Neuroradiology Duke University Medical Center @TimAmrheinMD Disclosures No relevant disclosures RSNA Research Scholar Grant ASNR
SIH: Trials and Research Endeavors Tim Amrhein, MD Assistant Professor of Neuroradiology Duke University Medical Center @TimAmrheinMD Disclosures No relevant disclosures RSNA Research Scholar Grant ASNR
Chapter 13 Summary Experiments and Observational Studies
 Chapter 13 Summary Experiments and Observational Studies What have we learned? We can recognize sample surveys, observational studies, and randomized comparative experiments. o These methods collect data
Chapter 13 Summary Experiments and Observational Studies What have we learned? We can recognize sample surveys, observational studies, and randomized comparative experiments. o These methods collect data
EBM: Therapy. Thunyarat Anothaisintawee, M.D., Ph.D. Department of Family Medicine, Ramathibodi Hospital, Mahidol University
 EBM: Therapy Thunyarat Anothaisintawee, M.D., Ph.D. Department of Family Medicine, Ramathibodi Hospital, Mahidol University How to appraise therapy literature Are the result valid? What are the result?
EBM: Therapy Thunyarat Anothaisintawee, M.D., Ph.D. Department of Family Medicine, Ramathibodi Hospital, Mahidol University How to appraise therapy literature Are the result valid? What are the result?
CONSORT 2010 checklist of information to include when reporting a randomised trial*
 CONSORT 2010 checklist of information to include when reporting a randomised trial* Section/Topic Title and abstract Introduction Background and objectives Item No Checklist item 1a Identification as a
CONSORT 2010 checklist of information to include when reporting a randomised trial* Section/Topic Title and abstract Introduction Background and objectives Item No Checklist item 1a Identification as a
Thursday, April 25, 13. Intervention Studies
 Intervention Studies Outline Introduction Advantages and disadvantages Avoidance of bias Parallel group studies Cross-over studies 2 Introduction Intervention study, or clinical trial, is an experiment
Intervention Studies Outline Introduction Advantages and disadvantages Avoidance of bias Parallel group studies Cross-over studies 2 Introduction Intervention study, or clinical trial, is an experiment
In this second module in the clinical trials series, we will focus on design considerations for Phase III clinical trials. Phase III clinical trials
 In this second module in the clinical trials series, we will focus on design considerations for Phase III clinical trials. Phase III clinical trials are comparative, large scale studies that typically
In this second module in the clinical trials series, we will focus on design considerations for Phase III clinical trials. Phase III clinical trials are comparative, large scale studies that typically
CHL 5225 H Advanced Statistical Methods for Clinical Trials. CHL 5225 H The Language of Clinical Trials
 CHL 5225 H Advanced Statistical Methods for Clinical Trials Two sources for course material 1. Electronic blackboard required readings 2. www.andywillan.com/chl5225h code of conduct course outline schedule
CHL 5225 H Advanced Statistical Methods for Clinical Trials Two sources for course material 1. Electronic blackboard required readings 2. www.andywillan.com/chl5225h code of conduct course outline schedule
Chapter 13. Experiments and Observational Studies. Copyright 2012, 2008, 2005 Pearson Education, Inc.
 Chapter 13 Experiments and Observational Studies Copyright 2012, 2008, 2005 Pearson Education, Inc. Observational Studies In an observational study, researchers don t assign choices; they simply observe
Chapter 13 Experiments and Observational Studies Copyright 2012, 2008, 2005 Pearson Education, Inc. Observational Studies In an observational study, researchers don t assign choices; they simply observe
Version No. 7 Date: July Please send comments or suggestions on this glossary to
 Impact Evaluation Glossary Version No. 7 Date: July 2012 Please send comments or suggestions on this glossary to 3ie@3ieimpact.org. Recommended citation: 3ie (2012) 3ie impact evaluation glossary. International
Impact Evaluation Glossary Version No. 7 Date: July 2012 Please send comments or suggestions on this glossary to 3ie@3ieimpact.org. Recommended citation: 3ie (2012) 3ie impact evaluation glossary. International
Title 1 Descriptive title identifying the study design, population, interventions, and, if applicable, trial acronym
 SPIRIT 2013 Checklist: Recommended items to address in a clinical trial protocol and related documents* Section/item Item No Description Addressed on page number Administrative information Title 1 Descriptive
SPIRIT 2013 Checklist: Recommended items to address in a clinical trial protocol and related documents* Section/item Item No Description Addressed on page number Administrative information Title 1 Descriptive
1 Case Control Studies
 Statistics April 11, 2013 Debdeep Pati 1 Case Control Studies 1. Case-control (retrospective) study: Select cases, Select controls, Compare cases and controls 2. Cutaneous melanoma (Autier, 1996). 420
Statistics April 11, 2013 Debdeep Pati 1 Case Control Studies 1. Case-control (retrospective) study: Select cases, Select controls, Compare cases and controls 2. Cutaneous melanoma (Autier, 1996). 420
Mapping the Informed Health Choices (IHC) Key Concepts (KC) to core concepts for the main steps of Evidence-Based Health Care (EBHC).
 KC No KC Short Title KC Statement EBHC concept EBHC sub concept EBHC statement EBHC Step Reported 1.01 Treatments can harm Treatments may be harmful Explain the use of harm/aetiologies for (rare) adverse
KC No KC Short Title KC Statement EBHC concept EBHC sub concept EBHC statement EBHC Step Reported 1.01 Treatments can harm Treatments may be harmful Explain the use of harm/aetiologies for (rare) adverse
Epidemiology: Overview of Key Concepts and Study Design. Polly Marchbanks
 Epidemiology: Overview of Key Concepts and Study Design Polly Marchbanks Lecture Outline (1) Key epidemiologic concepts - Definition - What epi is not - What epi is - Process of epi research Lecture Outline
Epidemiology: Overview of Key Concepts and Study Design Polly Marchbanks Lecture Outline (1) Key epidemiologic concepts - Definition - What epi is not - What epi is - Process of epi research Lecture Outline
Study Designs in Epidemiology
 Study Designs in Epidemiology Dr. Sireen Alkhaldi, BDS, MPH, DrPH First semester 2017/ 2018 Department of Family and Community Medicine School of Medicine/ The University of Jordan Epidemiologic Study
Study Designs in Epidemiology Dr. Sireen Alkhaldi, BDS, MPH, DrPH First semester 2017/ 2018 Department of Family and Community Medicine School of Medicine/ The University of Jordan Epidemiologic Study
Critical Appraisal Series
 Definition for therapeutic study Terms Definitions Study design section Observational descriptive studies Observational analytical studies Experimental studies Pragmatic trial Cluster trial Researcher
Definition for therapeutic study Terms Definitions Study design section Observational descriptive studies Observational analytical studies Experimental studies Pragmatic trial Cluster trial Researcher
Types of Biomedical Research
 INTRODUCTION & MEASUREMENT IN CLINICAL RESEARCH Sakda Arj Ong Vallipakorn, MD MSIT, MA (Information Science) Pediatrics, Pediatric Cardiology Emergency Medicine, Ped Emergency Family Medicine Section of
INTRODUCTION & MEASUREMENT IN CLINICAL RESEARCH Sakda Arj Ong Vallipakorn, MD MSIT, MA (Information Science) Pediatrics, Pediatric Cardiology Emergency Medicine, Ped Emergency Family Medicine Section of
12/26/2013. Types of Biomedical Research. Clinical Research. 7Steps to do research INTRODUCTION & MEASUREMENT IN CLINICAL RESEARCH S T A T I S T I C
 Types of Biomedical Research INTRODUCTION & MEASUREMENT IN CLINICAL RESEARCH Sakda Arj Ong Vallipakorn, MD MSIT, MA (Information Science) Pediatrics, Pediatric Cardiology Emergency Medicine, Ped Emergency
Types of Biomedical Research INTRODUCTION & MEASUREMENT IN CLINICAL RESEARCH Sakda Arj Ong Vallipakorn, MD MSIT, MA (Information Science) Pediatrics, Pediatric Cardiology Emergency Medicine, Ped Emergency
AP Statistics Exam Review: Strand 2: Sampling and Experimentation Date:
 AP Statistics NAME: Exam Review: Strand 2: Sampling and Experimentation Date: Block: II. Sampling and Experimentation: Planning and conducting a study (10%-15%) Data must be collected according to a well-developed
AP Statistics NAME: Exam Review: Strand 2: Sampling and Experimentation Date: Block: II. Sampling and Experimentation: Planning and conducting a study (10%-15%) Data must be collected according to a well-developed
Issues in Randomization
 INTRODUCTION TO THE PRINCIPLES AND PRACTICE OF CLINICAL RESEARCH Issues in Randomization Paul Wakim, PhD Chief, Biostatistics and Clinical Epidemiology Service Clinical Center, National Institutes of Health
INTRODUCTION TO THE PRINCIPLES AND PRACTICE OF CLINICAL RESEARCH Issues in Randomization Paul Wakim, PhD Chief, Biostatistics and Clinical Epidemiology Service Clinical Center, National Institutes of Health
UNDERSTANDING EPIDEMIOLOGICAL STUDIES. Csaba P Kovesdy, MD FASN Salem VA Medical Center, Salem VA University of Virginia, Charlottesville VA
 UNDERSTANDING EPIDEMIOLOGICAL STUDIES Csaba P Kovesdy, MD FASN Salem VA Medical Center, Salem VA University of Virginia, Charlottesville VA Study design in epidemiological research: Summary Observational
UNDERSTANDING EPIDEMIOLOGICAL STUDIES Csaba P Kovesdy, MD FASN Salem VA Medical Center, Salem VA University of Virginia, Charlottesville VA Study design in epidemiological research: Summary Observational
Regulatory Hurdles for Drug Approvals
 Regulatory Hurdles for Drug Approvals William R. Hiatt, MD Professor of Medicine/Cardiology University of Colorado School of Medicine President, CPC Clinical Research 25 min Conflicts CPC Clinical Research
Regulatory Hurdles for Drug Approvals William R. Hiatt, MD Professor of Medicine/Cardiology University of Colorado School of Medicine President, CPC Clinical Research 25 min Conflicts CPC Clinical Research
TEST YOURSELF: Multiple Choice 1 QUESTIONS
 1. In the investigation of an epidemic the most appropriate measure to describe the frequency of occurrence of illness is the: A. Prevalence B. Incidence rate C. Case-fatality rate D. Attack rate E. Mortality
1. In the investigation of an epidemic the most appropriate measure to describe the frequency of occurrence of illness is the: A. Prevalence B. Incidence rate C. Case-fatality rate D. Attack rate E. Mortality
Clinical Research Scientific Writing. K. A. Koram NMIMR
 Clinical Research Scientific Writing K. A. Koram NMIMR Clinical Research Branch of medical science that determines the safety and effectiveness of medications, devices, diagnostic products and treatment
Clinical Research Scientific Writing K. A. Koram NMIMR Clinical Research Branch of medical science that determines the safety and effectiveness of medications, devices, diagnostic products and treatment
Common Statistical Issues in Biomedical Research
 Common Statistical Issues in Biomedical Research Howard Cabral, Ph.D., M.P.H. Boston University CTSI Boston University School of Public Health Department of Biostatistics May 15, 2013 1 Overview of Basic
Common Statistical Issues in Biomedical Research Howard Cabral, Ph.D., M.P.H. Boston University CTSI Boston University School of Public Health Department of Biostatistics May 15, 2013 1 Overview of Basic
Epidemiological study design. Paul Pharoah Department of Public Health and Primary Care
 Epidemiological study design Paul Pharoah Department of Public Health and Primary Care Molecules What/why? Organelles Cells Tissues Organs Clinical medicine Individuals Public health medicine Populations
Epidemiological study design Paul Pharoah Department of Public Health and Primary Care Molecules What/why? Organelles Cells Tissues Organs Clinical medicine Individuals Public health medicine Populations
Research Methodology Workshop. Study Type
 Research Methodology Workshop Study Type What type of question? Frequency: how common is it? Aetiology: What caused this? Intervention: Does this intervention work? Diagnosis or test evaluation: How accurate
Research Methodology Workshop Study Type What type of question? Frequency: how common is it? Aetiology: What caused this? Intervention: Does this intervention work? Diagnosis or test evaluation: How accurate
Clinical Evidence: Asking the Question and Understanding the Answer. Keeping Up to Date. Keeping Up to Date
 Clinical Evidence: Asking the Question and Understanding the Answer Keeping Up to Date 5,000? per day 1,500 per day 95 per day Keeping Up to Date 5,000? per day 1,500 per day 95 per day 1 Bias Bias refers
Clinical Evidence: Asking the Question and Understanding the Answer Keeping Up to Date 5,000? per day 1,500 per day 95 per day Keeping Up to Date 5,000? per day 1,500 per day 95 per day 1 Bias Bias refers
Overview of Study Designs
 Overview of Study Designs Kyoungmi Kim, Ph.D. July 13 & 20, 2016 This seminar is jointly supported by the following NIH-funded centers: We are video recording this seminar so please hold questions until
Overview of Study Designs Kyoungmi Kim, Ph.D. July 13 & 20, 2016 This seminar is jointly supported by the following NIH-funded centers: We are video recording this seminar so please hold questions until
Variable Data univariate data set bivariate data set multivariate data set categorical qualitative numerical quantitative
 The Data Analysis Process and Collecting Data Sensibly Important Terms Variable A variable is any characteristic whose value may change from one individual to another Examples: Brand of television Height
The Data Analysis Process and Collecting Data Sensibly Important Terms Variable A variable is any characteristic whose value may change from one individual to another Examples: Brand of television Height
Randomized Clinical Trials
 Randomized Clinical Trials p. 1/42 Randomized Clinical Trials Hematology/Oncology Lecture Series Elizabeth G. Hill, PhD Associate Professor of Biostatistics 17 November 2011 Randomized Clinical Trials
Randomized Clinical Trials p. 1/42 Randomized Clinical Trials Hematology/Oncology Lecture Series Elizabeth G. Hill, PhD Associate Professor of Biostatistics 17 November 2011 Randomized Clinical Trials
Biases in clinical research. Seungho Ryu, MD, PhD Kanguk Samsung Hospital, Sungkyunkwan University
 Biases in clinical research Seungho Ryu, MD, PhD Kanguk Samsung Hospital, Sungkyunkwan University Learning objectives Describe the threats to causal inferences in clinical studies Understand the role of
Biases in clinical research Seungho Ryu, MD, PhD Kanguk Samsung Hospital, Sungkyunkwan University Learning objectives Describe the threats to causal inferences in clinical studies Understand the role of
Investigator Initiated Study Proposal Form
 Please submit completed form to IISReview@KCI1.com Date Submitted Name & Title Institution Address Phone Number Email Address Principal Investigator / Institution YES NO Multi Center Study Acelity Product(s)
Please submit completed form to IISReview@KCI1.com Date Submitted Name & Title Institution Address Phone Number Email Address Principal Investigator / Institution YES NO Multi Center Study Acelity Product(s)
Overview of Clinical Study Design Laura Lee Johnson, Ph.D. Statistician National Center for Complementary and Alternative Medicine US National
 Overview of Clinical Study Design Laura Lee Johnson, Ph.D. Statistician National Center for Complementary and Alternative Medicine US National Institutes of Health 2012 How Many Taken a similar course
Overview of Clinical Study Design Laura Lee Johnson, Ph.D. Statistician National Center for Complementary and Alternative Medicine US National Institutes of Health 2012 How Many Taken a similar course
Rapid appraisal of the literature: Identifying study biases
 Rapid appraisal of the literature: Identifying study biases Rita Popat, PhD Clinical Assistant Professor Division of Epidemiology Stanford University School of Medicine August 7, 2007 What is critical
Rapid appraisal of the literature: Identifying study biases Rita Popat, PhD Clinical Assistant Professor Division of Epidemiology Stanford University School of Medicine August 7, 2007 What is critical
Clinical Research Design and Conduction
 Clinical Research Design and Conduction Study Design M Mohsen Ibrahim, MD Cardiac Department Cairo University Algorithm for classification of types of clinical research Grimes DA and Schulz KF. Lancet
Clinical Research Design and Conduction Study Design M Mohsen Ibrahim, MD Cardiac Department Cairo University Algorithm for classification of types of clinical research Grimes DA and Schulz KF. Lancet
Appraising the Literature Overview of Study Designs
 Chapter 5 Appraising the Literature Overview of Study Designs Barbara M. Sullivan, PhD Department of Research, NUHS Jerrilyn A. Cambron, PhD, DC Department of Researach, NUHS EBP@NUHS Ch 5 - Overview of
Chapter 5 Appraising the Literature Overview of Study Designs Barbara M. Sullivan, PhD Department of Research, NUHS Jerrilyn A. Cambron, PhD, DC Department of Researach, NUHS EBP@NUHS Ch 5 - Overview of
Thursday, April 25, 13. Intervention Studies
 Intervention Studies Outline Introduction Advantages and disadvantages Avoidance of bias Parallel group studies Cross-over studies 2 Introduction Intervention study, or clinical trial, is an experiment
Intervention Studies Outline Introduction Advantages and disadvantages Avoidance of bias Parallel group studies Cross-over studies 2 Introduction Intervention study, or clinical trial, is an experiment
Designed Experiments have developed their own terminology. The individuals in an experiment are often called subjects.
 When we wish to show a causal relationship between our explanatory variable and the response variable, a well designed experiment provides the best option. Here, we will discuss a few basic concepts and
When we wish to show a causal relationship between our explanatory variable and the response variable, a well designed experiment provides the best option. Here, we will discuss a few basic concepts and
Sampling. (James Madison University) January 9, / 13
 Sampling The population is the entire group of individuals about which we want information. A sample is a part of the population from which we actually collect information. A sampling design describes
Sampling The population is the entire group of individuals about which we want information. A sample is a part of the population from which we actually collect information. A sampling design describes
Tool to Assess Risk of Bias in Cohort Studies
 Tool to Assess Risk of Bias in Cohort Studies Contributed by the CLARITY Group at McMaster University 1. Was selection of exposed and non-exposed cohorts drawn from the same population? Exposed and unexposed
Tool to Assess Risk of Bias in Cohort Studies Contributed by the CLARITY Group at McMaster University 1. Was selection of exposed and non-exposed cohorts drawn from the same population? Exposed and unexposed
PhD Course in Biostatistics
 PhD Course in Biostatistics Univ.-Prof. DI Dr. Andrea Berghold Institute for Medical Informatics, Statistics and Documentation Medical University of Graz andrea.berghold@medunigraz.at Content Introduction
PhD Course in Biostatistics Univ.-Prof. DI Dr. Andrea Berghold Institute for Medical Informatics, Statistics and Documentation Medical University of Graz andrea.berghold@medunigraz.at Content Introduction
Controlled Trials. Spyros Kitsiou, PhD
 Assessing Risk of Bias in Randomized Controlled Trials Spyros Kitsiou, PhD Assistant Professor Department of Biomedical and Health Information Sciences College of Applied Health Sciences University of
Assessing Risk of Bias in Randomized Controlled Trials Spyros Kitsiou, PhD Assistant Professor Department of Biomedical and Health Information Sciences College of Applied Health Sciences University of
Introduction to Experiments
 Experimental Methods in the Social Sciences Introduction to Experiments August 5, 2013 Experiments Increasingly Important in the Social Sciences Field experiments in political science as early as 1920s
Experimental Methods in the Social Sciences Introduction to Experiments August 5, 2013 Experiments Increasingly Important in the Social Sciences Field experiments in political science as early as 1920s
UNIT I SAMPLING AND EXPERIMENTATION: PLANNING AND CONDUCTING A STUDY (Chapter 4)
 UNIT I SAMPLING AND EXPERIMENTATION: PLANNING AND CONDUCTING A STUDY (Chapter 4) A DATA COLLECTION (Overview) When researchers want to make conclusions/inferences about an entire population, they often
UNIT I SAMPLING AND EXPERIMENTATION: PLANNING AND CONDUCTING A STUDY (Chapter 4) A DATA COLLECTION (Overview) When researchers want to make conclusions/inferences about an entire population, they often
GLOSSARY OF GENERAL TERMS
 GLOSSARY OF GENERAL TERMS Absolute risk reduction Absolute risk reduction (ARR) is the difference between the event rate in the control group (CER) and the event rate in the treated group (EER). ARR =
GLOSSARY OF GENERAL TERMS Absolute risk reduction Absolute risk reduction (ARR) is the difference between the event rate in the control group (CER) and the event rate in the treated group (EER). ARR =
Study design. Chapter 64. Research Methodology S.B. MARTINS, A. ZIN, W. ZIN
 Chapter 64 Study design S.B. MARTINS, A. ZIN, W. ZIN Research Methodology Scientific methodology comprises a set of rules and procedures to investigate the phenomena of interest. These rules and procedures
Chapter 64 Study design S.B. MARTINS, A. ZIN, W. ZIN Research Methodology Scientific methodology comprises a set of rules and procedures to investigate the phenomena of interest. These rules and procedures
Conducting and managing randomised controlled trials (RCTs)
 Conducting and managing randomised controlled trials (RCTs) Introduction Study Design We often wish to investigate the efficacy of new treatments and interventions on patient outcomes In this session,
Conducting and managing randomised controlled trials (RCTs) Introduction Study Design We often wish to investigate the efficacy of new treatments and interventions on patient outcomes In this session,
Choosing the right study design
 Choosing the right study design Main types of study design Randomised controlled trial (RCT) Cohort study Case-control study Cross-sectional study Case series/case note review Expert opinion BEST QUALITY
Choosing the right study design Main types of study design Randomised controlled trial (RCT) Cohort study Case-control study Cross-sectional study Case series/case note review Expert opinion BEST QUALITY
Assessing risk of bias
 Assessing risk of bias Norwegian Research School for Global Health Atle Fretheim Research Director, Norwegian Institute of Public Health Professor II, Uiniversity of Oslo Goal for the day We all have an
Assessing risk of bias Norwegian Research School for Global Health Atle Fretheim Research Director, Norwegian Institute of Public Health Professor II, Uiniversity of Oslo Goal for the day We all have an
Special Features of Randomized Controlled Trials
 Special Features of Randomized Controlled Trials Bangkok 2006 Kenneth F. Schulz, PhD, MBA Critical Methodological Elements in RCTs Randomization Avoiding and handling exclusions after trial entry Blinding
Special Features of Randomized Controlled Trials Bangkok 2006 Kenneth F. Schulz, PhD, MBA Critical Methodological Elements in RCTs Randomization Avoiding and handling exclusions after trial entry Blinding
Pros and Cons of Clinical Trials vs. Observational Studies. Beth Devine PCORP Summer Institute July 14, 2015
 Pros and Cons of Clinical Trials vs. Observational Studies Beth Devine PCORP Summer Institute July 14, 2015 Outline Randomized Trials Definitions: Pragmatic-explanatory continuum indicator summary (PRECIS)
Pros and Cons of Clinical Trials vs. Observational Studies Beth Devine PCORP Summer Institute July 14, 2015 Outline Randomized Trials Definitions: Pragmatic-explanatory continuum indicator summary (PRECIS)
Chapter 11: Experiments and Observational Studies p 318
 Chapter 11: Experiments and Observational Studies p 318 Observation vs Experiment An observational study observes individuals and measures variables of interest but does not attempt to influence the response.
Chapter 11: Experiments and Observational Studies p 318 Observation vs Experiment An observational study observes individuals and measures variables of interest but does not attempt to influence the response.
Goal: To become familiar with the methods that researchers use to investigate aspects of causation and methods of treatment
 Goal: To become familiar with the methods that researchers use to investigate aspects of causation and methods of treatment Scientific Study of Causation and Treatment Methods for studying causation Case
Goal: To become familiar with the methods that researchers use to investigate aspects of causation and methods of treatment Scientific Study of Causation and Treatment Methods for studying causation Case
95% 2.5% 2.5% +2SD 95% of data will 95% be within of data will 1.96 be within standard deviations 1.96 of sample mean
 Efficient Clinical Trials John H. Powers, MD Senior Medical Scientist SAIC in support of Clinical Collaborative Research Branch NIAID/NIH Introduction Definitions what is a small clinical trial? Review
Efficient Clinical Trials John H. Powers, MD Senior Medical Scientist SAIC in support of Clinical Collaborative Research Branch NIAID/NIH Introduction Definitions what is a small clinical trial? Review
Anatomy of Medical Studies: Part I. Emily D Agostino Epidemiology and Biostatistics CUNY School of Public Health at Hunter College
 Anatomy of Medical Studies: Part I!! Emily D Agostino Epidemiology and Biostatistics CUNY School of Public Health at Hunter College I. Standard format of a Medical Study Abstract (quick overview: what
Anatomy of Medical Studies: Part I!! Emily D Agostino Epidemiology and Biostatistics CUNY School of Public Health at Hunter College I. Standard format of a Medical Study Abstract (quick overview: what
lab exam lab exam Experimental Design Experimental Design when: Nov 27 - Dec 1 format: length = 1 hour each lab section divided in two
 lab exam when: Nov 27 - Dec 1 length = 1 hour each lab section divided in two register for the exam in your section so there is a computer reserved for you If you write in the 1st hour, you can t leave
lab exam when: Nov 27 - Dec 1 length = 1 hour each lab section divided in two register for the exam in your section so there is a computer reserved for you If you write in the 1st hour, you can t leave
Clinical problems and choice of study designs
 Evidence Based Dentistry Clinical problems and choice of study designs Asbjørn Jokstad University of Oslo, Norway Nov 21 2001 1 Manipulation with intervention Yes Experimental study No Non-experimental
Evidence Based Dentistry Clinical problems and choice of study designs Asbjørn Jokstad University of Oslo, Norway Nov 21 2001 1 Manipulation with intervention Yes Experimental study No Non-experimental
REVIEW FOR THE PREVIOUS LECTURE
 Slide 2-1 Calculator: The same calculator policies as for the ACT hold for STT 315: http://www.actstudent.org/faq/answers/calculator.html. It is highly recommended that you have a TI-84, as this is the
Slide 2-1 Calculator: The same calculator policies as for the ACT hold for STT 315: http://www.actstudent.org/faq/answers/calculator.html. It is highly recommended that you have a TI-84, as this is the
RANDOMIZED CLINICAL TRIALS. Study Designs. Randomized Clinical Trials (RCT) ASSIGN EXPOSURE Follow up Check for OUTCOME. Experimental studies
 RANDOMIZED CLINICAL TRIALS Study Designs Observational studies Analytic studies Descriptive studies Randomized Controlled trials Case control Cohort Cross sectional Randomized Clinical Trials (RCT) Assign
RANDOMIZED CLINICAL TRIALS Study Designs Observational studies Analytic studies Descriptive studies Randomized Controlled trials Case control Cohort Cross sectional Randomized Clinical Trials (RCT) Assign
Biostatistics and Epidemiology Step 1 Sample Questions Set 1
 Biostatistics and Epidemiology Step 1 Sample Questions Set 1 1. A study wishes to assess birth characteristics in a population. Which of the following variables describes the appropriate measurement scale
Biostatistics and Epidemiology Step 1 Sample Questions Set 1 1. A study wishes to assess birth characteristics in a population. Which of the following variables describes the appropriate measurement scale
NEED A SAMPLE SIZE? How to work with your friendly biostatistician!!!
 NEED A SAMPLE SIZE? How to work with your friendly biostatistician!!! BERD Pizza & Pilots November 18, 2013 Emily Van Meter, PhD Assistant Professor Division of Cancer Biostatistics Overview Why do we
NEED A SAMPLE SIZE? How to work with your friendly biostatistician!!! BERD Pizza & Pilots November 18, 2013 Emily Van Meter, PhD Assistant Professor Division of Cancer Biostatistics Overview Why do we
研究設計理論 ( 一 ): RANDOMIZED TRIALS. 醫學研究初階課程 General Medicine, CGMH 劉素旬醫師 03/12/2013
 1 研究設計理論 ( 一 ): RANDOMIZED TRIALS 醫學研究初階課程 General Medicine, CGMH 劉素旬醫師 03/12/2013 2 Learning Objectives Definition of a trial Features of trial design Elements of a trial Purposes of randomization and
1 研究設計理論 ( 一 ): RANDOMIZED TRIALS 醫學研究初階課程 General Medicine, CGMH 劉素旬醫師 03/12/2013 2 Learning Objectives Definition of a trial Features of trial design Elements of a trial Purposes of randomization and
115 remained abstinent. 140 remained abstinent. Relapsed Remained abstinent Total
 Chapter 10 Exercises 1. Intent-to-treat analysis: Example 1 In a randomized controlled trial to determine whether the nicotine patch reduces the risk of relapse among smokers who have committed to quit,
Chapter 10 Exercises 1. Intent-to-treat analysis: Example 1 In a randomized controlled trial to determine whether the nicotine patch reduces the risk of relapse among smokers who have committed to quit,
Observational Study Designs. Review. Today. Measures of disease occurrence. Cohort Studies
 Observational Study Designs Denise Boudreau, PhD Center for Health Studies Group Health Cooperative Today Review cohort studies Case-control studies Design Identifying cases and controls Measuring exposure
Observational Study Designs Denise Boudreau, PhD Center for Health Studies Group Health Cooperative Today Review cohort studies Case-control studies Design Identifying cases and controls Measuring exposure
aps/stone U0 d14 review d2 teacher notes 9/14/17 obj: review Opener: I have- who has
 aps/stone U0 d14 review d2 teacher notes 9/14/17 obj: review Opener: I have- who has 4: You should be able to explain/discuss each of the following words/concepts below... Observational Study/Sampling
aps/stone U0 d14 review d2 teacher notes 9/14/17 obj: review Opener: I have- who has 4: You should be able to explain/discuss each of the following words/concepts below... Observational Study/Sampling
The RoB 2.0 tool (individually randomized, cross-over trials)
 The RoB 2.0 tool (individually randomized, cross-over trials) Study design Randomized parallel group trial Cluster-randomized trial Randomized cross-over or other matched design Specify which outcome is
The RoB 2.0 tool (individually randomized, cross-over trials) Study design Randomized parallel group trial Cluster-randomized trial Randomized cross-over or other matched design Specify which outcome is
Guidelines for Reporting Non-Randomised Studies
 Revised and edited by Renatus Ziegler B.C. Reeves a W. Gaus b a Department of Public Health and Policy, London School of Hygiene and Tropical Medicine, Great Britain b Biometrie und Medizinische Dokumentation,
Revised and edited by Renatus Ziegler B.C. Reeves a W. Gaus b a Department of Public Health and Policy, London School of Hygiene and Tropical Medicine, Great Britain b Biometrie und Medizinische Dokumentation,
Lecture 2. Key Concepts in Clinical Research
 Lecture 2 Key Concepts in Clinical Research Outline Key Statistical Concepts Bias and Variability Type I Error and Power Confounding and Interaction Statistical Difference vs Clinical Difference One-sided
Lecture 2 Key Concepts in Clinical Research Outline Key Statistical Concepts Bias and Variability Type I Error and Power Confounding and Interaction Statistical Difference vs Clinical Difference One-sided
Design of Experiments & Introduction to Research
 Design of Experiments & Introduction to Research 1 Design of Experiments Introduction to Research Definition and Purpose Scientific Method Research Project Paradigm Structure of a Research Project Types
Design of Experiments & Introduction to Research 1 Design of Experiments Introduction to Research Definition and Purpose Scientific Method Research Project Paradigm Structure of a Research Project Types
WHAT TO DO IN ABSENCE OF HEAD TO HEAD CLINICAL TRIAL DATA. Lead the economic evaluation group at CHERE, University of Technology, Sydney
 WHAT TO DO IN ABSENCE OF HEAD TO HEAD CLINICAL TRIAL DATA MIXED TREATMENT COMPARISON (MTC) Stephen Goodall, Associate Professor of Health Economics Centre for Health Economics Research and Evaluation,
WHAT TO DO IN ABSENCE OF HEAD TO HEAD CLINICAL TRIAL DATA MIXED TREATMENT COMPARISON (MTC) Stephen Goodall, Associate Professor of Health Economics Centre for Health Economics Research and Evaluation,
Trick or Treat. In April!
 Trick or Treat. In April! Take one small envelope but Don t open it! Teaching and Leading EBM 2018 Randomization: simple, stratified and block Juan M. Lozano, MD, MSc Describe three methods of randomization:
Trick or Treat. In April! Take one small envelope but Don t open it! Teaching and Leading EBM 2018 Randomization: simple, stratified and block Juan M. Lozano, MD, MSc Describe three methods of randomization:
CLINICAL DECISION USING AN ARTICLE ABOUT TREATMENT JOSEFINA S. ISIDRO LAPENA MD, MFM, FPAFP PROFESSOR, UPCM
 CLINICAL DECISION USING AN ARTICLE ABOUT TREATMENT JOSEFINA S. ISIDRO LAPENA MD, MFM, FPAFP PROFESSOR, UPCM Principles of Decision Making Knowing the patient s true state is often unnecessary Does my patient
CLINICAL DECISION USING AN ARTICLE ABOUT TREATMENT JOSEFINA S. ISIDRO LAPENA MD, MFM, FPAFP PROFESSOR, UPCM Principles of Decision Making Knowing the patient s true state is often unnecessary Does my patient
Study design continued: intervention studies. Outline. Repetition principal approaches. Gustaf Edgren, PhD Karolinska Institutet.
 Study design continued: intervention studies Gustaf Edgren, PhD Karolinska Institutet Background Outline Uncontrolled trials Non randomized, controlled trials Randomized controlled trials (with an overview
Study design continued: intervention studies Gustaf Edgren, PhD Karolinska Institutet Background Outline Uncontrolled trials Non randomized, controlled trials Randomized controlled trials (with an overview
STAB22 Statistics I. Lecture 12
 STAB22 Statistics I Lecture 12 1 Midterm Grades Frequency 0 20 40 60 0 10 20 30 40 Marks ( / 40 ) Min Q1 Median Q3 Max Marks / 40 17.5 65 80 90 100 Marks / 100 7 26 32 36 40 2 Example (Sample Survey) The
STAB22 Statistics I Lecture 12 1 Midterm Grades Frequency 0 20 40 60 0 10 20 30 40 Marks ( / 40 ) Min Q1 Median Q3 Max Marks / 40 17.5 65 80 90 100 Marks / 100 7 26 32 36 40 2 Example (Sample Survey) The
Epidemiologic Methods I & II Epidem 201AB Winter & Spring 2002
 DETAILED COURSE OUTLINE Epidemiologic Methods I & II Epidem 201AB Winter & Spring 2002 Hal Morgenstern, Ph.D. Department of Epidemiology UCLA School of Public Health Page 1 I. THE NATURE OF EPIDEMIOLOGIC
DETAILED COURSE OUTLINE Epidemiologic Methods I & II Epidem 201AB Winter & Spring 2002 Hal Morgenstern, Ph.D. Department of Epidemiology UCLA School of Public Health Page 1 I. THE NATURE OF EPIDEMIOLOGIC
Trial Designs. Professor Peter Cameron
 Trial Designs Professor Peter Cameron OVERVIEW Review of Observational methods Principles of experimental design applied to observational studies Population Selection Looking for bias Inference Analysis
Trial Designs Professor Peter Cameron OVERVIEW Review of Observational methods Principles of experimental design applied to observational studies Population Selection Looking for bias Inference Analysis
Critical Review Form Therapy Objectives: Methods:
 Critical Review Form Therapy Clinical Trial Comparing Primary Coronary Angioplasty with Tissue-Plasminogen Activator for Acute Myocardial Infarction (GUSTO-IIb), NEJM 1997; 336: 1621-1628 Objectives: To
Critical Review Form Therapy Clinical Trial Comparing Primary Coronary Angioplasty with Tissue-Plasminogen Activator for Acute Myocardial Infarction (GUSTO-IIb), NEJM 1997; 336: 1621-1628 Objectives: To
Randomization: Too Important to Gamble with
 Randomization: Too Important to Gamble with A Presentation for the Delaware Chapter of the ASA Oct 18, 2012 Dennis Sweitzer, Ph.D., Principal Biostatistician Medidata Randomization Center of Excellence
Randomization: Too Important to Gamble with A Presentation for the Delaware Chapter of the ASA Oct 18, 2012 Dennis Sweitzer, Ph.D., Principal Biostatistician Medidata Randomization Center of Excellence
STATINS EVALUATION IN CORONARY PROCEDURES AND REVASCULARIZATION
 STATINS EVALUATION IN CORONARY PROCEDURES AND REVASCULARIZATION Otávio Berwanger, MD, FACS - On behalf of the SECURE Steering Committee and Investigators Trial Organization Trial Steering Committee Dr.
STATINS EVALUATION IN CORONARY PROCEDURES AND REVASCULARIZATION Otávio Berwanger, MD, FACS - On behalf of the SECURE Steering Committee and Investigators Trial Organization Trial Steering Committee Dr.
Epidemiologic Methods and Counting Infections: The Basics of Surveillance
 Epidemiologic Methods and Counting Infections: The Basics of Surveillance Ebbing Lautenbach, MD, MPH, MSCE University of Pennsylvania School of Medicine Nothing to disclose PENN Outline Definitions / Historical
Epidemiologic Methods and Counting Infections: The Basics of Surveillance Ebbing Lautenbach, MD, MPH, MSCE University of Pennsylvania School of Medicine Nothing to disclose PENN Outline Definitions / Historical
Accelerating Innovation in Statistical Design
 Implementing a National Cancer Clinical Trials System for the 21 st Century, Workshop #2 Session #5: Accelerating Innovation Through Effective Partnerships Accelerating Innovation in Statistical Design
Implementing a National Cancer Clinical Trials System for the 21 st Century, Workshop #2 Session #5: Accelerating Innovation Through Effective Partnerships Accelerating Innovation in Statistical Design
Handout 1: Introduction to the Research Process and Study Design STAT 335 Fall 2016
 DESIGNING OBSERVATIONAL STUDIES As we have discussed, for the purpose of establishing cause-and-effect relationships, observational studies have a distinct disadvantage in comparison to randomized comparative
DESIGNING OBSERVATIONAL STUDIES As we have discussed, for the purpose of establishing cause-and-effect relationships, observational studies have a distinct disadvantage in comparison to randomized comparative
Focused Research Questions and Study Designs
 Focused Research Questions and Study Designs PAROS Literature Review Workshop 11-12 April 2011, Khoo Teck Puat Hospital, Singapore Dianne Bautista, PhD Singapore Clinical Research Institute 11-12 April
Focused Research Questions and Study Designs PAROS Literature Review Workshop 11-12 April 2011, Khoo Teck Puat Hospital, Singapore Dianne Bautista, PhD Singapore Clinical Research Institute 11-12 April
Challenges of Observational and Retrospective Studies
 Challenges of Observational and Retrospective Studies Kyoungmi Kim, Ph.D. March 8, 2017 This seminar is jointly supported by the following NIH-funded centers: Background There are several methods in which
Challenges of Observational and Retrospective Studies Kyoungmi Kim, Ph.D. March 8, 2017 This seminar is jointly supported by the following NIH-funded centers: Background There are several methods in which
Use of Sacubitril/Valsartan in Heart Failure
 Use of Sacubitril/Valsartan in Heart Failure & the PARADIGM-HF trial Sarah Mackenzie, PharmD student, University of Toronto Presentation Outline Overview of: Entresto PARADIGM-HF trial Critical Appraisal
Use of Sacubitril/Valsartan in Heart Failure & the PARADIGM-HF trial Sarah Mackenzie, PharmD student, University of Toronto Presentation Outline Overview of: Entresto PARADIGM-HF trial Critical Appraisal
EPIDEMIOLOGY-BIOSTATISTICS EXAM Midterm 2004 PRINT YOUR LEGAL NAME:
 EPIDEMIOLOGY-BIOSTATISTICS EXAM Midterm 2004 PRINT YOUR LEGAL NAME: Instructions: This exam is 20% of your course grade. The maximum number of points for the course is 1,000; hence, this exam is worth
EPIDEMIOLOGY-BIOSTATISTICS EXAM Midterm 2004 PRINT YOUR LEGAL NAME: Instructions: This exam is 20% of your course grade. The maximum number of points for the course is 1,000; hence, this exam is worth
Clinical Epidemiology I: Deciding on Appropriate Therapy
 Clinical Epidemiology I: Deciding on Appropriate Therapy 1 Clinical Scenario [UG2B] 65 yo man controlled HTN 6-mo Hx cardioversion-resistant afib Benefit vs risk of long-term anticoagulation:? prevent
Clinical Epidemiology I: Deciding on Appropriate Therapy 1 Clinical Scenario [UG2B] 65 yo man controlled HTN 6-mo Hx cardioversion-resistant afib Benefit vs risk of long-term anticoagulation:? prevent
