Efficacy and Safety of Carmustine Wafers in the Treatment of Glioblastoma Multiforme: A Systematic Review
|
|
|
- Colin Hopkins
- 6 years ago
- Views:
Transcription
1 Received: / Accepted: DOI: / JTN Efficacy and Safety of Carmustine Wafers in the Treatment of Glioblastoma Multiforme: A Systematic Review Review Glioblastoma Multiforme Tedavisinde Karmustin Gofretlerinin Etkinlik ve Güvenirliği: Sistematik Bir Derleme Yun-Dong Zhang 1, Ruo-Yi Daı 2, Zhuo Chen 2, Yi-Hua Zhang 1, Xu-Zhi He 1, Ji Zhou 1 1Third Military Medical University, Institute of Surgery Research, DaPing Hospital, Department of Neurosurgery, Chongqing, China 2Third Military Medical University, Institute of Surgery Research, DaPing Hospital, Department of Geriatrics, Chongqing, China Corresponding Author: Yun-Dong Zhang / yundongzhang13@yeah.net, zhanggxh68@sina.com ABSTRACT The aim of this study was to a conduct a systematic review of carmustine wafers (Gliadel wafers) for the treatment of glioblastoma multiforme (GBM) to assess the survival benefit and safety of this therapy. The inclusion criteria were 1) prospective or retrospective clinical trial; 2) patients who had undergone resection for primary GBM or first recurrence of GBM with or without carmustine wafer implantation; 3) patients with malignant gliomas that included GBM; 4) outcomes including survival analysis of the GBM population. Six trials met the inclusion criteria; four were randomized, controlled trials and two were retrospective. The trials varied with regard to the type of patients and interventions. In three of the trials, patients with GBM who received carmustine wafers had significantly longer median survival than patients who did not receive wafers. Implantation of carmustine wafers did not significantly improve progression-free survival. Carmustine wafers did not increase adverse effects. This systematic review suggests that carmustine wafers have demonstrated promise as an effective and tolerable treatment in comparison to other treatment strategies in patients with GBM. Keywords: Glioblastoma multiforme, Carmustine wafers, Gliadel wafers, BCNU wafers ÖZ Çalışmanın amacı, glioblastoma multiforme (GBM) tedavisinde karmustin gofretlerinin (Gliadel gofretleri) bu tedavinin sağkalıma faydası ve güvenirliğini değerlendirmek üzere sistematik bir gözden geçirmesini yapmaktı. Çalışmaya alma kriterleri şöyleydi: 1) prospektif veya retrospektif klinik çalışma; 2) primer GBM için rezeksiyon yapılmış hastalar veya karmustin gofret implantasyonu ile veya olmadan ilk GBM öyküsü; 3) GBM dahil malign gliomlu hastalar; 4) GBM popülasyonunda sağkalım analizi dahil sonuçlar. Çalışmaya alma kriterlerini altı çalışma karşıladı; bunların dördü randomize kontrollü çalışmalar ve diğer ikisi retrospektifti. Çalışmalar hasta tipi ve girişimlerine göre farklılık gösteriyordu. Çalışmaların üçünde karmustin gofretleri alan GBM hastalarında medyan sağkalım gofret kullanılmayan hastalardan önemli ölçüde daha uzundu. Karmustin gofretleri implantasyonu progresyonsuz sağkalımı ve yan etkileri önemli ölçüde artırmadı. Bu sistematik derleme karmustin gofretlerinin GBM hastalarında diğer tedavi stratejileriyle karşılaştırıldığında etkin ve tolere edilebilir bir tedavi olarak ümit verdiklerini göstermiştir. ANAHTAR SÖZCÜKLER: Glioblastoma multiforme, Karmustin gofretleri, Gliadel gofretleri, BCNU gofretleri Introduction Glioblastoma multiforme (GBM) has been reported to be the most frequently occurring primary central nervous system tumor (1). In the United States and European countries, about 3 in 100,000 people have newly diagnosed GBM (1). These newly diagnosed GBMs account for more than 51% of all gliomas (1). The World Health Organization (WHO) grading system for gliomas, which is based on the histological characteristics of the tumor, ranges from grade I, least malignant, to grade IV, most malignant (1). Glioblastoma multiforme is categorized as grade IV. To improve survival of patients with GBM, the standard treatment of care consists of initial surgical resection of the tumor followed by radiotherapy and subsequently chemotherapy (17). Survival rates for men and women are similar, but younger patients have better survival rates than older patients (6). Five-year survival rates in the National Cancer Institute population databases are about 13% in patients ranging in age from years but only 1% in patients aged 75 years and older (6). About 40 years ago, nitrosoureas including 1.3-bis (2chlorethyl)-1-nitrosourea (carmustine) were first used for the treatment of gliomas (10). Their efficacy was moderate Turk Neurosurg 2014, Vol: 24, No: 5,
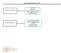
2 but doses that produced response rates up to 50% caused severe systemic side effects (10). A few years later, polymers were developed that could in theory deliver chemotherapy beyond the blood-brain barrier, and subsequently biodegradable polymers that permitted more constant drug delivery became available and such polymers were used to create the carmustine wafer (Gliadel wafer, BCNU wafer) (10). Several studies have found that carmustine wafers provide a survival benefit for patients with GBM. A meta-analysis published in 2007 found that treatment with carmustine wafers and temozolomide (TMZ) was more effective in improving survival than no chemotherapy (13). It was concluded that the clinical benefits of the treatment appeared to extend up to 24 months. A systematic review of Gliadel wafers used for the treatment of malignant glioma, which was also published in 2007 and included three randomized controlled trials (RTCs) and one prospective cohort study, found that two RTCs reported that patients with newly diagnosed malignant glioma who were treated with Gliadel wafers had a significant survival benefit compared with the control group (12). A study published in 2011 found that two s that assessed the effectiveness of Gliadel wafers for treating patients with high-grade glioma found that compared with placebo the wafer improved survival without increasing the incidence of adverse effects (5). The aim of this systematic review was to assess the survival benefit and safety of Gliadel wafers compared with placebo or alternative treatment in patients with GBM. Search Strategy Methods We performed a search using the following databases in December 2012: PubMed, Google Scholar, Biomedical Central. The search terms used were the following: glioblastoma multiforme, adjuvant, carmustine or BCNU, wafers, radiotherapy or surgery; primary or recurrent. We also searched the ASCO meeting website for relevant trials. The reference lists of the clinical trials identified were searched to identify additional trials. Using these search methods, a total of 149 records were identified (Figure 1). Next, the abstracts of the identified studies were screened for relevancy and duplicate patient databases. This resulted in 134 records being excluded, leaving 15 records (Figure 1). Selection of Studies Review studies had to meet the following criteria to be included in this systematic review: 1) prospective or retrospective clinical trial; 2) patients who had undergone resection for primary GBM or first recurrence of GBM with or without receiving Gliadel wafer implantation; 3) patients with malignant gliomas that included GBM; 4) outcomes including survival analysis of the GBM population. Studies were excluded if 1) patients had malignant gliomas but not GBM; 2) the study was not carried out to investigate the efficacy of carmustine wafer therapy; 3) the outcomes did not include survival analysis. Data Extraction The following data were extracted: study design, disease type, type of intervention, percentage of patients with GBM, percentage of male patients, Karnofsky performance score, median survival for GBM subgroup, survival rate, median progression-free survival, progression-free survival rate, duration of follow-up, and adverse events. The data were extracted from eligible studies by two independent reviewers. In instances where the two reviewers did not agree, a third reviewer was consulted to resolve the disagreement. Figure 1: Flow diagram of study selection. 640 Turk Neurosurg 2014, Vol: 24, No: 5,
3 Outcome Measures The primary outcome of the systematic review was median survival. Secondary outcomes were survival rate, median progression-free survival, and progression-free survival rate. Adverse events were reported and categorized. Selection of Trials Results Our search strategy resulted in seven papers being included in our systematic review (Figure 1) (4, 8, 9, 11, 14-16). These seven papers included six different trials; one trial was presented in two papers, one analyzing the short-term results and the other analyzing the long-term follow-up results (15, 16). Three of the studies were s (4, 8, 14-16) and two trials were retrospective (9, 11). The characteristics of the trials and types of patients are presented in Table I. Disease Type The patients studied with regard to disease type varied among the trials. All the trials only included patients with newly-diagnosed tumors except for the study by Kunwar et al. in which all patients had first recurrence of GBM (8) and the study by Brem et al. in which patients had malignant recurrent brain tumors (4). The study by McGirt et al. was the only trial that only included patients with newly diagnosed GBM (9). The trial conducted by et al. included patients with malignant glioma (15, 16), and the studies by Valtonen et al. and Noël et al. included patients with Grade III and IV glioma (11, 14). Type of Intervention In the study by Valtonen et al., one group of patients (68% GBM) was treated with Gliadel wafers and another group (100% GBM) was administered placebo wafers (14). Similarly, in the study conducted by et al. there was a Gliadel wafer group (84.2% GBM) and a placebo wafer group (88.3% GBM) (15, 16). In the study by Brem et al., patients (65.3%) were randomly assigned to surgery with Gliadel wafers or placebo wafers (4). Kunwar et al. compared patients treated with Gliadel wafers with those treated with IL13-PE38QQR (cintredekin besudotox) (8). In the study by McGirt et al., a group treated with the combination of Gliadel wafers and TMZ was compared with a group treated with TMZ alone (9). Noël et al. compared patients (71.4% GBM) treated with chemoradiotherapy followed by adjuvant chemotherapy with Gliadel wafers with patients (43.2% GBM) treated with chemoradiotherapy followed by adjuvant chemotherapy without Gliadel wafers (11). Overall Survival Six papers included data on median survival (Table II). Patients with GBM who received Gliadel wafers were found to have significantly longer median survival in the studies of Valtonen et al., et al., and McGirt et al. (9, 14, 16). Kunwar et al., and Noël et al. did not find that Gliadel wafers significantly increased median survival in patients with GBM (8, 11). In the trial by Brem et al., Gliadel wafers lowered the risk of death with an estimated hazard ratio of 0.8 (P=0.22) in the GBM subgroup analysis that included 145 patients (4). Table I: Characteristics of Trials and Patients Study Study design Disease type Type of Intervention Patient number GBM n(%) Male (%) KPS Valtonen 1997 (14) Primary Grade III and IV glioma 16 vs vs. 16 (68.8 vs. 100) 50 vs vs. 90 (Median) 2003 (16) Primary malignant glioma 120 vs vs. 106 (84.2 vs. 88.3) 63.3 vs NA 2006 (15) Primary malignant glioma 120 vs vs. 106 (84.2 vs. 88.3) 63.3 vs NA Brem 2005 (4) Kunwar 2010 (8) McGirt 2009 (9) Noël 2012 (11) Retrospective Retrospective Recurrent malignant glioma Recurrent GBM GBM Primary Grade III and IV glioma IL13- PE38QQR GW+TMZ vs. TMZ non-gw 110 vs vs vs vs vs. 73 (65.5 vs. 65.2) 93 vs. 183 (100 vs. 100) 30 vs. 45 (100 vs. 100) 20 vs. 16 (71.4 vs. 43.2) 67 vs vs vs. NA 77.0 vs (mean) 87.7 vs (Mean) 80 vs. NA (Median) 53.6 vs NA : randomized controlled trial; GW: Gliadel wafer; GBM: glioblastoma multiforme; KPS: Karnofsky performance score; TMZ: temozolomide; RT: radiotherapy; NA: not available. Turk Neurosurg 2014, Vol: 24, No: 5,
4 Table II: Summary of Efficacy Study Type of Intervention Median survival (mo) for GBM subgroup Survival rate (%) Median PFS (mo) PFS rate (%) Follow-up (mo) Valtonen 1997 (14) 53.3 vs (P= 0.008) NA NA NA (16) 13.5 vs (P=0.04) 59.2 vs (1 yr); 5.9 vs. 5.9 (P=0.9) NA (15) 13.1 vs (P=0.08) 59.2 vs (1 yr); 15.8 vs. 8.3 (2 yr); 9.2 vs. 1.7 (3 yr, P=0.01) NA NA 56 Brem 2005 (4) NA 56 vs. 36 (6 mo; P=0.02) NA NA Kunwar 2010 (8) IL13- PE38QQR 8.8 vs. 9.1 (P=0.476) NA NA NA NA McGirt 2009 (9) GW+TMZ vs. TMZ 20.7 vs (P < 0.01) NA NA NA 18±10 Noël 2012 (11) non-gw 20.8 vs vs (6 mo) a 75 vs (1 yr) a ; 38.9 vs. 0 (2 yr) a (P=0.067) 73.7 vs (6 mo)a 36.8 vs (12 mo) 9.7 vs.7.8 a a 27.6 vs (18 mo) a (P=0.4) 17.1 GBM: glioblastoma multiforme; PFS: progression free survival; RFS: relapse free survival; mo: month; yr: year; a: for GBM subgroup. Only the two papers by et al., the paper by Noël et al., and the paper by Brem et al. compared the survival rate between groups. In the initial report by et al., the survival rate was not significantly better for GBM patients who received Gliadel wafers compared with GBM patients given placebo (16). In the follow-up study by et al. the 1-, 2- and 3-year survival rates were calculated and at 3 years the patients who received Gliadel wafers had a significantly better survival rate than those who received placebo (15). Noël et al. calculated survival rates at 6 months, 1 year, and 2 years for the subgroup of patients with GBM and found no significant differences in survival rates between those who did and did not receive Gliadel wafers (11). Brem et al. found that Gliadel wafers significantly improved survival at 6 months (4). Progression-Free Survival Data on median progression-free survival were calculated in the initial study by et al. and in the study by Noël et al. (11, 16). There was no significant difference in progressionfree survival between patients given Gliadel wafers and those given placebo in the study by et al. (16). Also, Noël et al. did not find a significant difference between GBM patients who received Gliadel wafers and those who did not (11). Only in the study by Noël et al. was the progression-free survival rate reported. They found that there was no significant difference at 6, 12 and 18 months in the rates for GBM patients who did and did not receive Gliadel wafers (11). Adverse Events Only Valtonen et al., et al. in their initial paper, Kunwar et al., and Brem et al. reported the incidence of adverse events (4, 8, 14, 16). The incidence of adverse events in these studies is summarized in Table III. et al. found no significant difference in events between the groups except for intracranial hypertension being more common at 6 months after surgery in the Gliadel wafer group, a result they attributed to recurrence of the primary tumor rather than to the wafers (16). The only significant difference in adverse events between the groups in the study by Kunwar et al. was that pulmonary embolism was more common in the group that received IL13-PE38QQR (8). Discussion Our search strategy identified six trials of carmustine wafers for treating primary or first recurrence of GBM in which survival analysis was included. In two trials that also included patients with WHO Grade III glioma (11, 14), we extracted the data from subgroup analysis of GBM. The carmustine wafers were compared with placebo in three trials (4, 14, 16) and they were compared with a different treatment that did not include the wafers in another three trials (8, 9, 11). Our review revealed three trials in which patients with GBM who received carmustine wafers had statistically significant longer overall survival (9, 14, 16). Survival rate data were only reported in three trials (3, 15, 16) and the patients who received the 642 Turk Neurosurg 2014, Vol: 24, No: 5,
5 Table III: Summary of Adverse Events Adverse event (%) Valtonen 1997 ( ) 2003 ( ) Brem 2005 ( ) Kunwar 2010 ( IL13) Nervous system Abnormal gait 5 vs vs. 0.3 Amnesia 9.2 vs.10 Aphasia 17.5 vs vs. 1.2 Ataxia 5.8 vs. 4.2 Brain edema 22.5 vs vs. 0.3 Confusion 23.3 vs 20.8 Convulsion 19 vs vs Depression 15.8 vs Dizziness 5.0 vs. 9.2 Facial paralysis 6.7 vs. 4.2 Grand mal convulsion 5.0 vs. 4.2 Headache 0.3 vs. 0.4 Hemiplegia 40.8 vs vs. 0.3 Hemiparesis 38 vs vs. 0.8 Incoordination 2.5 vs vs. 0.3 Intracranial hypertension 9.2 vs. 1.7 Intracranial infection 3.6 vs. 0.9 Monoparesis 0.2 vs. 0.5 Mental status change 0.1 vs. 0.3 Neuropathy 6.7 vs Seizure 37.3 vs Speech disorder 10.8 vs. 8.3 Vascular system Anemia 7 vs. 11 Deep vein thrombosis 0.2 vs. 0.5 Thrombocytopenia 2 vs. 2 GW: Gliadel wafer. carmustine wafers had a significantly better overall survival rate in two of these trials. The incidence of events was similar in the two groups in all four trials that reported on adverse events (8, 14, 16). Overall results of these trials seem to suggest that carmustine wafer implantation demonstrates promise as an effective and tolerable treatment strategy for GBM. Several other studies that did not meet our criteria for inclusion in this systematic review reported on survival of patients with GBM who received carmustine wafers. In a single-arm study, Bock et al. enrolled 44 patients with newly diagnosed GBM. All patients received carmustine wafers and concomitant radiochemotherapy (3). The median overall survival was 12.7 months and median progression-free survival was 7.0 months. In another single-arm study, Kleinberg et al. analyzed data for 46 patients with newly diagnosed malignant glioma (87% with GBM) who received carmustine wafers followed by radiotherapy (7). The median survival for the patients with GBM was 12.8 months. Attenello et al. retrospectively reviewed the records of 1013 patients with WHO grade III or IV malignant glioma (2). A total of 288 patients received carmustine wafers. Patients with GBM who received the wafers had a median survival of 13.5 months and a 2-year survival rate of 20%. In all four studies the median survival was about 13 months for patients with GBM who had implantation of carmustine wafers. Previous systematic reviews of studies that focused on the use of carmustine wafers in patients with GBM were carried out by Perry et al. and Hart et al. (5, 12). The goal of the systematic review of Hart et al. was to assess the clinical effectiveness of the wafers and the goal of the systematic review by Perry et al. was to assess overall survival, adverse events, and quality of life. Hart et al. identified two s in which carmustine Turk Neurosurg 2014, Vol: 24, No: 5,
6 wafers were compared with placebo and patients afterwards received radiotherapy and concluded that wafer implantation significantly improved survival without causing an increase in adverse events when compared with placebo. Perry et al. included two s with patients who had newly diagnosed GBM and in both studies it was found that the wafers provided a significant survival benefit. Spiegel et al. performed a meta-analysis to assess the effect of adjuvant chemotherapy for GBM (13). They identified 16 trials in which adjuvant chemotherapy was compared with no therapy. Three of the trials included local therapy. The patients received carmustine wafers in two trials while cisplatin was used in one trial. The subgroup analysis of local therapy included all three trials. In our study, we focused on the efficacy and safety of Gliadel wafers for treating patients with GBM. We found that the percentage of adverse events in the four studies that reported these findings was similar between the two treatment groups in each study. However, there was a large variation among the studies in the percentage of adverse events and this might be attributable to the different type of tumors (e.g., newly diagnosed vs recurrent) and differences in interventions. We found differences in the definition of median survival among the five trials. Valtonen et al. and McGirt et al. defined survival as the time from surgery to death (9, 14). et al. and Kunwar et al. defined survival as the time from randomization until death or last known time the patient was alive (8, 16). In the study by Noël et al., survival was defined on the basis of the time after diagnosis by histopathological examination (11). Our review also revealed that the definitions of progression-free survival differed among the two studies that reported this outcome. et al. used progressionfree survival whereas Noël et al. used relapse-free survival (11, 16). Our systematic review of the efficacy and safety of carmustine wafers in patients with GBM was limited because the studies we identified with our search strategy varied in the type of interventions used and the types of diseases selected for study inclusion. Also, there were differences in the definitions of overall survival and progression-free survival among the studies. In addition, not all studies reported the survival rate, median progression-free survival rate, progression-free survival rate, or percentages of adverse events. In conclusion, our systematic review suggests that carmustine wafers show promise as an effective and tolerable treatment compared with other treatment strategies in patients with GBM. The use of carmustine wafers as monotherapy or in combination therapy for treating patients with GBM warrants further investigation in larger, randomized controlled trials. References 1. Adamson C, Kanu OO, Mehta AI, Di C, Lin N, Mattox AK, Bigner DD: Glioblastoma multiforme: A review of where we have been and where we are going. Expert Opin Investig Drugs 18(8): , Attenello FJ, Mukherjee D, Datoo G, McGirt MJ, Bohan E, Weingart JD, Olivi A, Quinones-Hinojosa A, Brem H: Use of Gliadel (BCNU) wafer in the surgical treatment of malignant glioma: A 10-year institutional experience. Ann Surg Oncol 15: , Bock HC, Puchner MJ, Lohmann F, Schutze M, Koll S, Ketter R, Buchalla R, Rainov N, Kantelhardt SR, Rohde V, Giese A: Firstline treatment of malignant glioma with carmustine implants followed by concomitant radiochemotherapy: A multicenter experience. Neurosurg Rev 33: , Brem H, Piantadosi S, Burger PC, Walker M, Selker R, Vick NA, Black K, Sisti M, Brem S, Mohr G, et al: -controlled trial of safety and efficacy of intraoperative controlled delivery by biodegradable polymers of chemotherapy for recurrent gliomas. The Polymer-brain Tumor Treatment Group. Lancet 345: , Hart MG, Grant R, Garside R, Rogers G, Somerville M, Stein K: Chemotherapy wafers for high grade glioma. Cochrane Database Syst Rev (3):CD007294, Kanu OO, Mehta A, Di C, Lin N, Bortoff K, Bigner DD, Yan H, Adamson DC: Glioblastoma multiforme: A review of therapeutic targets. Expert Opin Ther Targets 13: , Kleinberg LR, Weingart J, Burger P, Carson K, Grossman SA, Li K, Olivi A, Wharam MD, Brem H: Clinical course and pathologic findings after Gliadel and radiotherapy for newly diagnosed malignant glioma: Implications for patient management. Cancer Invest 22:1-9, Kunwar S, Chang S, M, Vogelbaum M, Sampson J, Barnett G, Shaffrey M, Ram Z, Piepmeier J, Prados M, Croteau D, Pedain C, Leland P, Husain SR, Joshi BH, Puri RK, Group PS: Phase III randomized trial of CED of IL13-PE38QQR vs Gliadel wafers for recurrent glioblastoma. Neuro Oncol 12: , McGirt MJ, Than KD, Weingart JD, Chaichana KL, Attenello FJ, Olivi A, Laterra J, Kleinberg LR, Grossman SA, Brem H, Quinones-Hinojosa A: Gliadel (BCNU) wafer plus concomitant temozolomide therapy after primary resection of glioblastoma multiforme. J Neurosurg 110: , Nagpal S: The role of BCNU polymer wafers (Gliadel) in the treatment of malignant glioma. Neurosurg Clin N Am 23: ix, , Noël G, Schott R, Froelich S, Gaub MP, Boyer P, Fischer-Lokou D, Dufour P, Kehrli P, Maitrot D: Retrospective comparison of chemoradiotherapy followed by adjuvant chemotherapy, with or without prior gliadel implantation (carmustine) after initial surgery in patients with newly diagnosed high-grade gliomas. Int J Radiat Oncol Biol Phys 82: , Perry J, Chambers A, Spithoff K, Laperriere N: Gliadel wafers in the treatment of malignant glioma: A systematic review. Curr Oncol 14: , Spiegel BM, Esrailian E, Laine L, Chamberlain MC: Clinical impact of adjuvant chemotherapy in glioblastoma multiforme: A meta-analysis. CNS Drugs 21: , Turk Neurosurg 2014, Vol: 24, No: 5,
7 14. Valtonen S, Timonen U, Toivanen P, Kalimo H, Kivipelto L, Heiskanen O, Unsgaard G, Kuurne T: Interstitial chemotherapy with carmustine-loaded polymers for high-grade gliomas: A randomized double-blind study. Neurosurgery 41:44-48; discussion 48-49, M, Ram Z, Riddle V, Hilt D, Bortey E: Executive Committee of the Gliadel Study Group: Gliadel wafer in initial surgery for malignant glioma: Long-term follow-up of a multicenter controlled trial. Acta Neurochir (Wien) 148: ; discussion 275, M, Hilt DC, Bortey E, Delavault P, Olivares R, Warnke PC, Whittle IR, Jaaskelainen J, Ram Z: A phase 3 trial of local chemotherapy with biodegradable carmustine (BCNU) wafers (Gliadel wafers) in patients with primary malignant glioma. Neuro Oncol 5:79-88, Zhang X, Zhang W, Cao WD, Cheng G, Zhang YQ: Glioblastoma multiforme: Molecular characterization and current treatment strategy (Review). Exp Ther Med 3:9-14, 2012 Turk Neurosurg 2014, Vol: 24, No: 5,
Clinical Management Protocol Chemotherapy [Glioblastoma Multiforme (CNS)] Protocol for Planning and Treatment
![Clinical Management Protocol Chemotherapy [Glioblastoma Multiforme (CNS)] Protocol for Planning and Treatment Clinical Management Protocol Chemotherapy [Glioblastoma Multiforme (CNS)] Protocol for Planning and Treatment](/thumbs/87/96032554.jpg) Protocol for Planning and Treatment The process to be followed when a course of chemotherapy is required to treat: GLIOBLASTOMA MULTIFORME (CNS) Patient information given at each stage following agreed
Protocol for Planning and Treatment The process to be followed when a course of chemotherapy is required to treat: GLIOBLASTOMA MULTIFORME (CNS) Patient information given at each stage following agreed
First-line treatment of malignant glioma with carmustine implants followed by concomitant radiochemotherapy: a multicenter experience
 Neurosurg Rev (2010) 33:441 449 DOI 10.1007/s10143-010-0280-7 ORIGINAL ARTICLE First-line treatment of malignant glioma with carmustine implants followed by concomitant radiochemotherapy: a multicenter
Neurosurg Rev (2010) 33:441 449 DOI 10.1007/s10143-010-0280-7 ORIGINAL ARTICLE First-line treatment of malignant glioma with carmustine implants followed by concomitant radiochemotherapy: a multicenter
Carmustine implants and Temozolomide for the treatment of newly diagnosed high grade glioma
 National Institute for Health and Clinical Excellence Health Technology Appraisal Carmustine implants and Temozolomide for the treatment of newly diagnosed high grade glioma Personal statement Conventional
National Institute for Health and Clinical Excellence Health Technology Appraisal Carmustine implants and Temozolomide for the treatment of newly diagnosed high grade glioma Personal statement Conventional
Gl i o b l a s t o m a multiforme is the most common
 J Neurosurg 110:583 588, 2009 Gliadel (BCNU) wafer plus concomitant temozolomide therapy after primary resection of glioblastoma multiforme Clinical article Ma t t h e w J. McGi r t, M.D., 1 Kh o i D.
J Neurosurg 110:583 588, 2009 Gliadel (BCNU) wafer plus concomitant temozolomide therapy after primary resection of glioblastoma multiforme Clinical article Ma t t h e w J. McGi r t, M.D., 1 Kh o i D.
Safety and efficacy of carmustine (BCNU) wafers for metastatic brain tumors
 1 di 9 28/06/2016 10.42 Surg Neurol Int. 2016; 7(Suppl 11): S295 S299. Published online 2016 May 6. doi: 10.4103/2152-7806.181987 PMCID: PMC4866053 Safety and efficacy of carmustine (BCNU) wafers for metastatic
1 di 9 28/06/2016 10.42 Surg Neurol Int. 2016; 7(Suppl 11): S295 S299. Published online 2016 May 6. doi: 10.4103/2152-7806.181987 PMCID: PMC4866053 Safety and efficacy of carmustine (BCNU) wafers for metastatic
Safety and efficacy of carmustine (BCNU) wafers for metastatic brain tumors
 SNI: Neuro-Oncology, a supplement to Surgical Neurology International OPEN ACCESS For entire Editorial Board visit : http://www.surgicalneurologyint.com Editor: Daniel Silbergeld, University of Washington
SNI: Neuro-Oncology, a supplement to Surgical Neurology International OPEN ACCESS For entire Editorial Board visit : http://www.surgicalneurologyint.com Editor: Daniel Silbergeld, University of Washington
Lynn S. Ashby 1*, Kris A. Smith 2 and Baldassarre Stea 3
 Ashby et al. World Journal of Surgical Oncology (2016) 14:225 DOI 10.1186/s12957-016-0975-5 REVIEW Gliadel wafer implantation combined with standard radiotherapy and concurrent followed by adjuvant temozolomide
Ashby et al. World Journal of Surgical Oncology (2016) 14:225 DOI 10.1186/s12957-016-0975-5 REVIEW Gliadel wafer implantation combined with standard radiotherapy and concurrent followed by adjuvant temozolomide
Of all neoplastic lesions and across all medical specialties, Eosinophilic meningitis triggered by implanted Gliadel wafers: case report
 CASE REPORT J Neurosurg 126:1783 1787, 2017 Eosinophilic meningitis triggered by implanted Gliadel wafers: case report Kiyotaka Saito, MD, 1 Kouji Yamasaki, MD, 1 Kiyotaka Yokogami, MD, PhD, 1 Asya Ivanova,
CASE REPORT J Neurosurg 126:1783 1787, 2017 Eosinophilic meningitis triggered by implanted Gliadel wafers: case report Kiyotaka Saito, MD, 1 Kouji Yamasaki, MD, 1 Kiyotaka Yokogami, MD, PhD, 1 Asya Ivanova,
Concomitant (without adjuvant) temozolomide and radiation to treat glioblastoma: A retrospective study
 Concomitant (without adjuvant) temozolomide and radiation to treat glioblastoma: A retrospective study T Sridhar 1, A Gore 1, I Boiangiu 1, D Machin 2, R P Symonds 3 1. Department of Oncology, Leicester
Concomitant (without adjuvant) temozolomide and radiation to treat glioblastoma: A retrospective study T Sridhar 1, A Gore 1, I Boiangiu 1, D Machin 2, R P Symonds 3 1. Department of Oncology, Leicester
Contemporary Management of Glioblastoma
 Contemporary Management of Glioblastoma Incidence Rates of Primary Brain Tumors Central Brain Tumor Registry of the United States, 1992-1997 100 Number of Cases per 100,000 Population 10 1 0.1 x I x I
Contemporary Management of Glioblastoma Incidence Rates of Primary Brain Tumors Central Brain Tumor Registry of the United States, 1992-1997 100 Number of Cases per 100,000 Population 10 1 0.1 x I x I
Brain tumor and Gliadel wafer treatment
 Symposium Brain tumor and Gliadel wafer treatment Panigrahi M, Das PK 1, Parikh PM 2 Department of Neurosurgery, Krishna Institute of Medical Sciences, Secunderabad, 1 Department of Medical Oncology, Indraprastha
Symposium Brain tumor and Gliadel wafer treatment Panigrahi M, Das PK 1, Parikh PM 2 Department of Neurosurgery, Krishna Institute of Medical Sciences, Secunderabad, 1 Department of Medical Oncology, Indraprastha
PROCARBAZINE, lomustine, and vincristine (PCV) is
 RAPID PUBLICATION Procarbazine, Lomustine, and Vincristine () Chemotherapy for Anaplastic Astrocytoma: A Retrospective Review of Radiation Therapy Oncology Group Protocols Comparing Survival With Carmustine
RAPID PUBLICATION Procarbazine, Lomustine, and Vincristine () Chemotherapy for Anaplastic Astrocytoma: A Retrospective Review of Radiation Therapy Oncology Group Protocols Comparing Survival With Carmustine
Supratentorial Glioblastoma Multiforme: The Role of Surgical Resection Versus Biopsy Among Older Patients
 Ann Surg Oncol DOI 10.1245/s10434-010-1242-6 ORIGINAL ARTICLE NEURO-ONCOLOGY Supratentorial Glioblastoma Multiforme: The Role of Surgical Resection Versus Biopsy Among Older Patients Kaisorn L. Chaichana,
Ann Surg Oncol DOI 10.1245/s10434-010-1242-6 ORIGINAL ARTICLE NEURO-ONCOLOGY Supratentorial Glioblastoma Multiforme: The Role of Surgical Resection Versus Biopsy Among Older Patients Kaisorn L. Chaichana,
Survival of High Grade Glioma Patients Treated by Three Radiation Schedules with Chemotherapy: A Retrospective Comparative Study
 Original Article Research in Oncology June 2017; Vol. 13, No. 1: 18-22. DOI: 10.21608/resoncol.2017.552.1022 Survival of High Grade Glioma Patients Treated by Three Radiation Schedules with Chemotherapy:
Original Article Research in Oncology June 2017; Vol. 13, No. 1: 18-22. DOI: 10.21608/resoncol.2017.552.1022 Survival of High Grade Glioma Patients Treated by Three Radiation Schedules with Chemotherapy:
Glioblastoma: Adjuvant Treatment Abdulrazag Ajlan, MD, MSc, FRCSC, UCNS(D)
 Glioblastoma: Adjuvant Treatment Abdulrazag Ajlan, MD, MSc, FRCSC, UCNS(D) *Neurosurgery Consultant, King Saud University, Riyadh, KSA *Adjunct Teaching Faculty, Neurosurgery, Stanford School Of Medicine,
Glioblastoma: Adjuvant Treatment Abdulrazag Ajlan, MD, MSc, FRCSC, UCNS(D) *Neurosurgery Consultant, King Saud University, Riyadh, KSA *Adjunct Teaching Faculty, Neurosurgery, Stanford School Of Medicine,
Scottish Medicines Consortium
 Scottish Medicines Consortium temozolomide 5, 20, 100 and 250mg capsules (Temodal ) Schering Plough UK Ltd No. (244/06) New indication: for the treatment of newly diagnosed glioblastoma multiforme concomitantly
Scottish Medicines Consortium temozolomide 5, 20, 100 and 250mg capsules (Temodal ) Schering Plough UK Ltd No. (244/06) New indication: for the treatment of newly diagnosed glioblastoma multiforme concomitantly
ORIGINAL PAPERS. The Impact of Surgery on the Efficacy of Adjuvant Therapy in Glioblastoma Multiforme
 ORIGINAL PAPERS Adv Clin Exp Med 2015, 24, 2, 279 287 DOI: 10.17219/acem/40456 Copyright by Wroclaw Medical University ISSN 1899 5276 Anna Brzozowska 1, 2, A D, Anna Toruń 3, G, Maria Mazurkiewicz1, 2,
ORIGINAL PAPERS Adv Clin Exp Med 2015, 24, 2, 279 287 DOI: 10.17219/acem/40456 Copyright by Wroclaw Medical University ISSN 1899 5276 Anna Brzozowska 1, 2, A D, Anna Toruń 3, G, Maria Mazurkiewicz1, 2,
Rapid regression of glioblastoma following carmustine wafer implantation: A case report
 MOLECULAR AND CLINICAL ONCOLOGY 5: 153-157, 2016 Rapid regression of glioblastoma following carmustine wafer implantation: A case report JUNYA FUKAI 1, HIROKI NISHIBAYASHI 1, YUJI UEMATSU 2, YONEHIRO KANEMURA
MOLECULAR AND CLINICAL ONCOLOGY 5: 153-157, 2016 Rapid regression of glioblastoma following carmustine wafer implantation: A case report JUNYA FUKAI 1, HIROKI NISHIBAYASHI 1, YUJI UEMATSU 2, YONEHIRO KANEMURA
Hypofractionated radiation therapy for glioblastoma
 Hypofractionated radiation therapy for glioblastoma Luis Souhami, MD, FASTRO Professor McGill University Department of Oncology, Division of Radiation Oncology Montreal Canada McGill University Health
Hypofractionated radiation therapy for glioblastoma Luis Souhami, MD, FASTRO Professor McGill University Department of Oncology, Division of Radiation Oncology Montreal Canada McGill University Health
Pioneering vaccines that transform lives.
 Pioneering vaccines that transform lives. Immunomic Therapeutics, Inc. LAMP-Vax for Glioblastoma: CMV-LAMP-Vax Executive Summary Executive Summary pp65-lamp-vax First Line Therapy for Glioblastoma Multiforme
Pioneering vaccines that transform lives. Immunomic Therapeutics, Inc. LAMP-Vax for Glioblastoma: CMV-LAMP-Vax Executive Summary Executive Summary pp65-lamp-vax First Line Therapy for Glioblastoma Multiforme
Corporate Medical Policy
 Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: tumor_treatment_fields_therapy 9/2013 11/2017 11/2018 6/2018 Description of Procedure or Service Tumor-treatment
Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: tumor_treatment_fields_therapy 9/2013 11/2017 11/2018 6/2018 Description of Procedure or Service Tumor-treatment
Title: Carmustine (BCNU) for recurrent glioblastoma multiforme: efficacy, toxicity and prognostic factors
 Author's response to reviews Title: Carmustine (BCNU) for recurrent glioblastoma multiforme: efficacy, toxicity and prognostic factors Authors: Thomas P Reithmeier (thomas.reithmeier@uniklinik-freiburg.de)
Author's response to reviews Title: Carmustine (BCNU) for recurrent glioblastoma multiforme: efficacy, toxicity and prognostic factors Authors: Thomas P Reithmeier (thomas.reithmeier@uniklinik-freiburg.de)
Copyright information:
 Outcome of Adult Brain Tumor Consortium (ABTC) prospective dose-finding trials of I-125 balloon brachytherapy in high-grade gliomas: challenges in clinical trial design and technology development when
Outcome of Adult Brain Tumor Consortium (ABTC) prospective dose-finding trials of I-125 balloon brachytherapy in high-grade gliomas: challenges in clinical trial design and technology development when
Collection of Recorded Radiotherapy Seminars
 IAEA Human Health Campus Collection of Recorded Radiotherapy Seminars http://humanhealth.iaea.org The Role of Radiosurgery in the Treatment of Gliomas Luis Souhami, MD Professor Department of Radiation
IAEA Human Health Campus Collection of Recorded Radiotherapy Seminars http://humanhealth.iaea.org The Role of Radiosurgery in the Treatment of Gliomas Luis Souhami, MD Professor Department of Radiation
Technology appraisal guidance Published: 27 June 2007 nice.org.uk/guidance/ta121
 Carmustine implants and temozolomide for the treatment of newly diagnosed high-grade glioma Technology appraisal guidance Published: 27 June 2007 nice.org.uk/guidance/ta121 NICE 2018. All rights reserved.
Carmustine implants and temozolomide for the treatment of newly diagnosed high-grade glioma Technology appraisal guidance Published: 27 June 2007 nice.org.uk/guidance/ta121 NICE 2018. All rights reserved.
Intracavitary Balloon Catheter Brain Brachytherapy for Malignant Gliomas or Metastasis to the Brain. Original Policy Date
 MP 8.01.23 Intracavitary Balloon Catheter Brain Brachytherapy for Malignant Gliomas or Metastasis to the Brain Medical Policy Section Therapy Issue 12/2013 Original Policy Date 12/2013 Last Review Status/Date
MP 8.01.23 Intracavitary Balloon Catheter Brain Brachytherapy for Malignant Gliomas or Metastasis to the Brain Medical Policy Section Therapy Issue 12/2013 Original Policy Date 12/2013 Last Review Status/Date
Corporate Medical Policy
 Corporate Medical Policy Analysis of MGMT Promoter Methylation in Malignant Gliomas File Name: Origination: Last CAP Review: Next CAP Review: Last Review: analysis_of_mgmt_promoter_methylation_in_malignant_gliomas
Corporate Medical Policy Analysis of MGMT Promoter Methylation in Malignant Gliomas File Name: Origination: Last CAP Review: Next CAP Review: Last Review: analysis_of_mgmt_promoter_methylation_in_malignant_gliomas
MP Intracavitary Balloon Catheter Brain Brachytherapy for Malignant Gliomas or Metastasis to the Brain. Related Policies None
 Medical Policy MP 8.01.45 BCBSA Ref. Policy: 8.01.45 Last Review: 08/20/2018 Effective Date: 08/20/2018 Section: Therapy Related Policies None DISCLAIMER Our medical policies are designed for informational
Medical Policy MP 8.01.45 BCBSA Ref. Policy: 8.01.45 Last Review: 08/20/2018 Effective Date: 08/20/2018 Section: Therapy Related Policies None DISCLAIMER Our medical policies are designed for informational
Glia-derived neoplasms of the. Í œ e Å< ˇÖ ˇÁ <9ËÜrj ]<t <ÎÜËÜä ]<ÖÁ j ] oèf ]< Clinical Development of Experimental Therapies for Malignant Glioma
![Glia-derived neoplasms of the. Í œ e Å< ˇÖ ˇÁ <9ËÜrj ]<t <ÎÜËÜä ]<ÖÁ j ] oèf ]< Clinical Development of Experimental Therapies for Malignant Glioma Glia-derived neoplasms of the. Í œ e Å< ˇÖ ˇÁ <9ËÜrj ]<t <ÎÜËÜä ]<ÖÁ j ] oèf ]< Clinical Development of Experimental Therapies for Malignant Glioma](/thumbs/87/97053330.jpg) SQU Med J, February 2011, Vol. 11, Iss. 1, pp. 5-28, Epub. 12 th Feb 11 Invited Article Submitted 4 th Aug 10 Revision ReQ. 5 th Oct 10, Revision recd. 25 th Oct 10 Accepted 8 th Nov 10 Clinical Development
SQU Med J, February 2011, Vol. 11, Iss. 1, pp. 5-28, Epub. 12 th Feb 11 Invited Article Submitted 4 th Aug 10 Revision ReQ. 5 th Oct 10, Revision recd. 25 th Oct 10 Accepted 8 th Nov 10 Clinical Development
HEMORRHAGIC GLIOBLASTOMA MULTIFORM: PREVALENCE, PREDISPOSING FACTORS AND PROGNOSIS AMONG ADULT KFMC PATIENTS.
 HEMORRHAGIC GLIOBLASTOMA MULTIFORM: PREVALENCE, PREDISPOSING FACTORS AND PROGNOSIS AMONG ADULT KFMC PATIENTS. Dr. Ahmed Lary Dr. Ali balbaid Rabea Qutub Saad Al-Maimouni Introduction Gliomas is a collection
HEMORRHAGIC GLIOBLASTOMA MULTIFORM: PREVALENCE, PREDISPOSING FACTORS AND PROGNOSIS AMONG ADULT KFMC PATIENTS. Dr. Ahmed Lary Dr. Ali balbaid Rabea Qutub Saad Al-Maimouni Introduction Gliomas is a collection
National Institute for Health and Clinical Excellence. Single Technology Appraisal (STA)
 National Institute for Health and Clinical Excellence Appendix C Comment 1: the draft scope Single Technology Appraisal (STA) Carmustine implants for the treatment of recurrent glioblastoma multiforme
National Institute for Health and Clinical Excellence Appendix C Comment 1: the draft scope Single Technology Appraisal (STA) Carmustine implants for the treatment of recurrent glioblastoma multiforme
Marizomib (MRZ): Brain Penetrant Irreversible Pan-Proteasome Inhibitor
 MARIZOMIB (MRZ) WITH BEVACIZUMAB (BEV) IN WHO GRADE IV MALIGNANT GLIOMA (G4 MG): FULL ENROLLMENT RESULTS FROM THE PHASE 1, MULTICENTER, OPEN-LABEL STUDY Daniela Bota, MD, PhD 1, Annick Desjardins, MD,
MARIZOMIB (MRZ) WITH BEVACIZUMAB (BEV) IN WHO GRADE IV MALIGNANT GLIOMA (G4 MG): FULL ENROLLMENT RESULTS FROM THE PHASE 1, MULTICENTER, OPEN-LABEL STUDY Daniela Bota, MD, PhD 1, Annick Desjardins, MD,
Corporate Medical Policy
 Corporate Medical Policy Brachytherapy, Intracavitary Balloon Catheter for Brain Cancer File Name: Origination: Last CAP Review: Next CAP Review: Last Review: brachytherapy_intracavitary_balloon_catheter_for_brain_cancer
Corporate Medical Policy Brachytherapy, Intracavitary Balloon Catheter for Brain Cancer File Name: Origination: Last CAP Review: Next CAP Review: Last Review: brachytherapy_intracavitary_balloon_catheter_for_brain_cancer
Cilengitide (Impetreve) for glioblastoma multiforme. February 2012
 Cilengitide (Impetreve) for glioblastoma multiforme February 2012 This technology summary is based on information available at the time of research and a limited literature search. It is not intended to
Cilengitide (Impetreve) for glioblastoma multiforme February 2012 This technology summary is based on information available at the time of research and a limited literature search. It is not intended to
Josh is JB s brother and caregiver.
 PUT GBM ON PAUSE PUT LIFE ON PLAY Josh is JB s brother and caregiver. JB is an Optune user. OPTUNE + TMZ HAS BEEN PROVEN TO PROVIDE LONG-TERM QUALITY SURVIVAL TO PATIENTS WITH NEWLY DIAGNOSED GBM1,2,*
PUT GBM ON PAUSE PUT LIFE ON PLAY Josh is JB s brother and caregiver. JB is an Optune user. OPTUNE + TMZ HAS BEEN PROVEN TO PROVIDE LONG-TERM QUALITY SURVIVAL TO PATIENTS WITH NEWLY DIAGNOSED GBM1,2,*
FACT SHEET. About Optune
 About Optune Optune is the Tumor Treating Fields (TTFields) delivery system that is approved by the United States (US) Food and Drug Administration (FDA) for the treatment of adult patients with glioblastoma.
About Optune Optune is the Tumor Treating Fields (TTFields) delivery system that is approved by the United States (US) Food and Drug Administration (FDA) for the treatment of adult patients with glioblastoma.
Antiangiogenic drugs in unresectable glioblastoma. Dra. Carmen Balañá. /
 Antiangiogenic drugs in unresectable glioblastoma Dra. Carmen Balañá. / Outcome for unresectable GBM Overall survival for unresectable GBM without further treatment is: 3 months at most. Radiotherapy increases
Antiangiogenic drugs in unresectable glioblastoma Dra. Carmen Balañá. / Outcome for unresectable GBM Overall survival for unresectable GBM without further treatment is: 3 months at most. Radiotherapy increases
It s s Always Something!
 It s s Always Something! New Approaches in Brain Tumor Treatment Virginia Stark-Vance, M.D. When Something Is a Brain Tumor Brain tumors aren t rare: there are over 100,000/yr Most originate as other cancers
It s s Always Something! New Approaches in Brain Tumor Treatment Virginia Stark-Vance, M.D. When Something Is a Brain Tumor Brain tumors aren t rare: there are over 100,000/yr Most originate as other cancers
Appraisal of carmustine. implants and temozolomide. for newly diagnosed high. Brain and Spine Foundation
 Appraisal of carmustine implants and temozolomide for newly diagnosed high grade glioma Brain and Spine Foundation June 2005 Submission to the National Institute for Health and Clinical Excellence Brain
Appraisal of carmustine implants and temozolomide for newly diagnosed high grade glioma Brain and Spine Foundation June 2005 Submission to the National Institute for Health and Clinical Excellence Brain
National Horizon Scanning Centre. Bevacizumab (Avastin) for glioblastoma multiforme - relapsed. August 2008
 Bevacizumab (Avastin) for glioblastoma multiforme - relapsed August 2008 This technology summary is based on information available at the time of research and a limited literature search. It is not intended
Bevacizumab (Avastin) for glioblastoma multiforme - relapsed August 2008 This technology summary is based on information available at the time of research and a limited literature search. It is not intended
Wafer migration: Monitor patients for signs of obstructive hydrocephalus (5.5). Embryo-fetal toxicity: Can cause fetal harm (5.6)
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use GLIADEL WAFER safely and effectively. See full prescribing information for GLIADEL WAFER. GLIADEL
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use GLIADEL WAFER safely and effectively. See full prescribing information for GLIADEL WAFER. GLIADEL
11/27/2017. Modern Treatment of Meningiomas. Disclosures. Modern is Better? No disclosures relevant to this presentation
 Modern Treatment of Meningiomas Michael A. Vogelbaum MD, PhD Professor of Neurosurgery Cleveland Clinic Disclosures No disclosures relevant to this presentation IP and royalties related to drug and device
Modern Treatment of Meningiomas Michael A. Vogelbaum MD, PhD Professor of Neurosurgery Cleveland Clinic Disclosures No disclosures relevant to this presentation IP and royalties related to drug and device
New Approaches in Brain Tumor Treatment. Virginia Stark-Vance, M.D.
 New Approaches in Brain Tumor Treatment Virginia Stark-Vance, M.D. The Primary Brain Tumors es for PicturesMCD for 004.JPG 00 Meningioma 30% Glioblastoma 20% Astrocytoma 10% Nerve sheath 8% Pituitary 6%
New Approaches in Brain Tumor Treatment Virginia Stark-Vance, M.D. The Primary Brain Tumors es for PicturesMCD for 004.JPG 00 Meningioma 30% Glioblastoma 20% Astrocytoma 10% Nerve sheath 8% Pituitary 6%
Glioblastoma: Current Treatment Approach 8/20/2018
 Glioblastoma: Current Treatment Approach 8/20/2018 Overview What is Glioblastoma? How is it diagnosed How is it treated? Principles of Treatment Surgery, Radiation, Chemotherapy Current Standard of care
Glioblastoma: Current Treatment Approach 8/20/2018 Overview What is Glioblastoma? How is it diagnosed How is it treated? Principles of Treatment Surgery, Radiation, Chemotherapy Current Standard of care
POLICY PRODUCT VARIATIONS DESCRIPTION/BACKGROUND RATIONALE DEFINITIONS BENEFIT VARIATIONS DISCLAIMER CODING INFORMATION REFERENCES POLICY HISTORY
 Original Issue Date (Created): November 26, 2013 Most Recent Review Date (Revised): November 26, 2013 Effective Date: April 1, 2014 POLICY PRODUCT VARIATIONS DESCRIPTION/BACKGROUND RATIONALE DEFINITIONS
Original Issue Date (Created): November 26, 2013 Most Recent Review Date (Revised): November 26, 2013 Effective Date: April 1, 2014 POLICY PRODUCT VARIATIONS DESCRIPTION/BACKGROUND RATIONALE DEFINITIONS
Parisa Mirzadehgan, MPH, CCRP
 Parisa Mirzadehgan, MPH, CCRP Approved therapy for a newly diagnosed GBM patient includes surgical resection radiation & temozolomide Upon recurrence there are few approved options surgical implantation
Parisa Mirzadehgan, MPH, CCRP Approved therapy for a newly diagnosed GBM patient includes surgical resection radiation & temozolomide Upon recurrence there are few approved options surgical implantation
The New Approaches to Brain Tumor Therapy (NABTT) CNS Consortium: Organization, Objectives, and Activities
 The New Approaches to Brain Tumor Therapy (NABTT) CNS Consortium: Organization, Objectives, and Activities Stuart A. Grossman, MD, Joy D. Fisher, MA, Steven Piantadosi, MD, PhD, and Henry Brem, MD The
The New Approaches to Brain Tumor Therapy (NABTT) CNS Consortium: Organization, Objectives, and Activities Stuart A. Grossman, MD, Joy D. Fisher, MA, Steven Piantadosi, MD, PhD, and Henry Brem, MD The
Selecting the Optimal Treatment for Brain Metastases
 Selecting the Optimal Treatment for Brain Metastases Clinical Practice Today CME Co-provided by Learning Objectives Upon completion, participants should be able to: Understand the benefits, limitations,
Selecting the Optimal Treatment for Brain Metastases Clinical Practice Today CME Co-provided by Learning Objectives Upon completion, participants should be able to: Understand the benefits, limitations,
VAL-083: Validated DNA-targeting Agent for Underserved Cancer Patients. September 2018
 VAL-083: Validated DNA-targeting Agent for Underserved Cancer Patients September 2018 Forward-Looking Statements Any statements contained in this presentation that do not describe historical facts may
VAL-083: Validated DNA-targeting Agent for Underserved Cancer Patients September 2018 Forward-Looking Statements Any statements contained in this presentation that do not describe historical facts may
Zurich Open Repository and Archive. Long-term survival of glioblastoma patients treated with radiotherapy and lomustine plus temozolomide
 University of Zurich Zurich Open Repository and Archive Winterthurerstr. 190 CH-8057 Zurich http://www.zora.uzh.ch Year: 2009 Long-term survival of glioblastoma patients treated with radiotherapy and lomustine
University of Zurich Zurich Open Repository and Archive Winterthurerstr. 190 CH-8057 Zurich http://www.zora.uzh.ch Year: 2009 Long-term survival of glioblastoma patients treated with radiotherapy and lomustine
Neuro-Oncology Program
 Neuro-Oncology Program The goals of the Neuro-oncology Committee are: 1) to improve duration and quality of life of brain tumor patients; 2) to assess disease and treatment-related effects on neurocognitive
Neuro-Oncology Program The goals of the Neuro-oncology Committee are: 1) to improve duration and quality of life of brain tumor patients; 2) to assess disease and treatment-related effects on neurocognitive
CNS Tumors: The Med Onc Perspective. Ronald J. Scheff, MD Associate Clinical Professor Weill Medical College of Cornell U.
 CNS Tumors: The Med Onc Perspective Ronald J. Scheff, MD Associate Clinical Professor Weill Medical College of Cornell U. Disclosure Speakers Bureau, Merck Basic Oncology Concepts Tissue Diagnosis Stage
CNS Tumors: The Med Onc Perspective Ronald J. Scheff, MD Associate Clinical Professor Weill Medical College of Cornell U. Disclosure Speakers Bureau, Merck Basic Oncology Concepts Tissue Diagnosis Stage
Society for Immunotherapy of Cancer (SITC) Immunotherapy for the Treatment of Brain Metastases
 Society for Immunotherapy of Cancer (SITC) Immunotherapy for the Treatment of Brain Metastases Geoffrey T. Gibney, MD Georgetown-Lombardi Comprehensive Cancer Center Medstar-Georgetown University Hospital
Society for Immunotherapy of Cancer (SITC) Immunotherapy for the Treatment of Brain Metastases Geoffrey T. Gibney, MD Georgetown-Lombardi Comprehensive Cancer Center Medstar-Georgetown University Hospital
Brain Schema March 2018
 Breast Cancer w/brain Mets 14 2283 / Lilly Abemaciclib PO Breast Ca or Melanoma mets allowed Leptomeningeal mets excluded Want brain met tissue Melanoma w/brain Mets 14 2209 / Bristol Myers Squibb Nivolumab
Breast Cancer w/brain Mets 14 2283 / Lilly Abemaciclib PO Breast Ca or Melanoma mets allowed Leptomeningeal mets excluded Want brain met tissue Melanoma w/brain Mets 14 2209 / Bristol Myers Squibb Nivolumab
21/03/2017. Disclosure. Practice Changing Articles in Neuro Oncology for 2016/17. Gliomas. Objectives. Gliomas. No conflicts to declare
 Practice Changing Articles in Neuro Oncology for 2016/17 Disclosure No conflicts to declare Frances Cusano, BScPharm, ACPR April 21, 2017 Objectives Gliomas To describe the patient selection, methodology
Practice Changing Articles in Neuro Oncology for 2016/17 Disclosure No conflicts to declare Frances Cusano, BScPharm, ACPR April 21, 2017 Objectives Gliomas To describe the patient selection, methodology
THE EFFECTIVE OF BRAIN CANCER AND XAY BETWEEN THEORY AND IMPLEMENTATION. Mustafa Rashid Issa
 THE EFFECTIVE OF BRAIN CANCER AND XAY BETWEEN THEORY AND IMPLEMENTATION Mustafa Rashid Issa ABSTRACT: Illustrate malignant tumors that form either in the brain or in the nerves originating in the brain.
THE EFFECTIVE OF BRAIN CANCER AND XAY BETWEEN THEORY AND IMPLEMENTATION Mustafa Rashid Issa ABSTRACT: Illustrate malignant tumors that form either in the brain or in the nerves originating in the brain.
Corporate Medical Policy Tumor-Treatment Fields Therapy for Glioblastoma
 Corporate Medical Policy Tumor-Treatment Fields Therapy for Glioblastoma File Name: Origination: Last CAP Review: Next CAP Review: Last Review: tumor-treatment_fields_therapy_for_glioblastoma 9/2013 11/2017
Corporate Medical Policy Tumor-Treatment Fields Therapy for Glioblastoma File Name: Origination: Last CAP Review: Next CAP Review: Last Review: tumor-treatment_fields_therapy_for_glioblastoma 9/2013 11/2017
EORTC (RTOG 0834 Endorsed) Opened: July 22, 2009
 January 2011 0834-1 EORTC 26053 22054 (RTOG 0834 Endorsed) Protocol Status: Opened: July 22, 2009 Title: Phase III Trial on Concurrent and Adjuvant Temozolomide Chemotherapy in Non-1P/19Q Deleted Anaplastic
January 2011 0834-1 EORTC 26053 22054 (RTOG 0834 Endorsed) Protocol Status: Opened: July 22, 2009 Title: Phase III Trial on Concurrent and Adjuvant Temozolomide Chemotherapy in Non-1P/19Q Deleted Anaplastic
Going Past the Data for Temozolomide. J. Lee Villano, M.D., Ph.D., Nathalie Letarte, B.Pharm, M.Sc, Linda R. Bressler, Pharm. D.
 Going Past the Data for Temozolomide J. Lee Villano, M.D., Ph.D., Nathalie Letarte, B.Pharm, M.Sc, Linda R. Bressler, Pharm. D. Departments of Medicine (JLV), Neurosurgery (JLV) and Pharmacy Practice (LRB)
Going Past the Data for Temozolomide J. Lee Villano, M.D., Ph.D., Nathalie Letarte, B.Pharm, M.Sc, Linda R. Bressler, Pharm. D. Departments of Medicine (JLV), Neurosurgery (JLV) and Pharmacy Practice (LRB)
Bevacizumab rescue therapy extends the survival in patients with recurrent malignant glioma
 Original Article Bevacizumab rescue therapy extends the survival in patients with recurrent malignant glioma Lin-Bo Cai, Juan Li, Ming-Yao Lai, Chang-Guo Shan, Zong-De Lian, Wei-Ping Hong, Jun-Jie Zhen,
Original Article Bevacizumab rescue therapy extends the survival in patients with recurrent malignant glioma Lin-Bo Cai, Juan Li, Ming-Yao Lai, Chang-Guo Shan, Zong-De Lian, Wei-Ping Hong, Jun-Jie Zhen,
Practice of Interferon Therapy
 Interferon Therapy Practice of Interferon Therapy Brain tumor JMAJ 47(1): 18 23, 2004 Toshihiko WAKABAYASHI* and Jun YOSHIDA** *Associate Professor, Center for Genetic and Regenerative Medicine, Nagoya
Interferon Therapy Practice of Interferon Therapy Brain tumor JMAJ 47(1): 18 23, 2004 Toshihiko WAKABAYASHI* and Jun YOSHIDA** *Associate Professor, Center for Genetic and Regenerative Medicine, Nagoya
Presently, malignant gliomas are treated by resection,
 Neuro-Oncology A phase 3 trial of local chemotherapy with biodegradable carmustine (BCNU) wafers (Gliadel wafers) in patients with primary malignant glioma 1,2 Manfred Westphal, 3 Dana C. Hilt, Enoch Bortey,
Neuro-Oncology A phase 3 trial of local chemotherapy with biodegradable carmustine (BCNU) wafers (Gliadel wafers) in patients with primary malignant glioma 1,2 Manfred Westphal, 3 Dana C. Hilt, Enoch Bortey,
University of Zurich. Temozolomide and MGMT forever? Zurich Open Repository and Archive. Weller, M. Year: 2010
 University of Zurich Zurich Open Repository and Archive Winterthurerstr. 190 CH-8057 Zurich Year: 2010 Temozolomide and MGMT forever? Weller, M Weller, M (2010). Temozolomide and MGMT forever? Neuro-Oncology,
University of Zurich Zurich Open Repository and Archive Winterthurerstr. 190 CH-8057 Zurich Year: 2010 Temozolomide and MGMT forever? Weller, M Weller, M (2010). Temozolomide and MGMT forever? Neuro-Oncology,
Douglas Jolly Executive VP R&D Tocagen Inc.
 REPLICATING RETROVIRUSES FOR MANIPULATION OF THE TUMOR IMMUNE ECOSYSTEM: PRECLINICAL AND CLINICAL OUTCOMES. Douglas Jolly Executive VP R&D Tocagen Inc. PEGS 2018 IT Track Boston May 1 2018 Outline Phase
REPLICATING RETROVIRUSES FOR MANIPULATION OF THE TUMOR IMMUNE ECOSYSTEM: PRECLINICAL AND CLINICAL OUTCOMES. Douglas Jolly Executive VP R&D Tocagen Inc. PEGS 2018 IT Track Boston May 1 2018 Outline Phase
Newcastle Neuro-oncology Team Audit of Outcome of Glioblastoma Multiforme Chemoradiotherapy Treatment
 Newcastle Neuro-oncology Team Audit of Outcome of Glioblastoma Multiforme Chemoradiotherapy Treatment Jennifer Wright Neurosurgery SSC Audit Team Jennifer Wright, Rachel Tresman, Cyril Dubois, Surash Surash,
Newcastle Neuro-oncology Team Audit of Outcome of Glioblastoma Multiforme Chemoradiotherapy Treatment Jennifer Wright Neurosurgery SSC Audit Team Jennifer Wright, Rachel Tresman, Cyril Dubois, Surash Surash,
Management of malignant glioma: steady progress with multimodal approaches
 Neurosurg Focus 20 (4):E3, 2006 Management of malignant glioma: steady progress with multimodal approaches LYNN S. ASHBY, M.D., AND TIMOTHY C. RYKEN, M.D. Barrow Neurological Institute, St. Joseph s Hospital
Neurosurg Focus 20 (4):E3, 2006 Management of malignant glioma: steady progress with multimodal approaches LYNN S. ASHBY, M.D., AND TIMOTHY C. RYKEN, M.D. Barrow Neurological Institute, St. Joseph s Hospital
Temozolomide in the treatment of recurrent malignant glioma in Chinese patients!"#$%&'()*+,-./0,1234
 Key words: Astrocytoma; Brain neoplasms; Disease-free survival; Glioblastoma; Neoplasm recurrence!!"!"!"#$ DTM Chan WS Poon YL Chan HK Ng Hong Kong Med J 2005;11:452-6 The Chinese University of Hong Kong,
Key words: Astrocytoma; Brain neoplasms; Disease-free survival; Glioblastoma; Neoplasm recurrence!!"!"!"#$ DTM Chan WS Poon YL Chan HK Ng Hong Kong Med J 2005;11:452-6 The Chinese University of Hong Kong,
Tristate Lung Meeting 2014 Pro-Con Debate: Surgery has no role in the management of certain subsets of N2 disease
 Tristate Lung Meeting 2014 Pro-Con Debate: Surgery has no role in the management of certain subsets of N2 disease Jennifer E. Tseng, MD UFHealth Cancer Center-Orlando Health Sep 12, 2014 Background Approximately
Tristate Lung Meeting 2014 Pro-Con Debate: Surgery has no role in the management of certain subsets of N2 disease Jennifer E. Tseng, MD UFHealth Cancer Center-Orlando Health Sep 12, 2014 Background Approximately
Management of Brain Metastases Sanjiv S. Agarwala, MD
 Management of Brain Metastases Sanjiv S. Agarwala, MD Professor of Medicine Temple University School of Medicine Chief, Oncology & Hematology St. Luke s Cancer Center, Bethlehem, PA, USA Incidence (US):
Management of Brain Metastases Sanjiv S. Agarwala, MD Professor of Medicine Temple University School of Medicine Chief, Oncology & Hematology St. Luke s Cancer Center, Bethlehem, PA, USA Incidence (US):
1. Introduction. Correspondence should be addressed to Christopher M. Lee; Received 9 July 2013; Accepted 27 August 2013
 Case Reports in Oncological Medicine Volume 2013, Article ID 431857, 5 pages http://dx.doi.org/10.1155/2013/431857 Case Report Long-Term Survival and Improved Quality of Life following Multiple Repeat
Case Reports in Oncological Medicine Volume 2013, Article ID 431857, 5 pages http://dx.doi.org/10.1155/2013/431857 Case Report Long-Term Survival and Improved Quality of Life following Multiple Repeat
CURRENT CONTROVERSIES IN THE MANAGEMENT OF HIGH GRADE GLIOMAS: AN INTERACTIVE CASE DISCUSSION *
 CURRENT CONTROVERSIES IN THE MANAGEMENT OF HIGH GRADE GLIOMAS: AN INTERACTIVE CASE DISCUSSION * Alessandro Olivi, MD, Jaishri Blakeley, MD, and Allen K. Sills, MD, FACS ABSTRACT The management of glioma
CURRENT CONTROVERSIES IN THE MANAGEMENT OF HIGH GRADE GLIOMAS: AN INTERACTIVE CASE DISCUSSION * Alessandro Olivi, MD, Jaishri Blakeley, MD, and Allen K. Sills, MD, FACS ABSTRACT The management of glioma
Optimal Management of Isolated HER2+ve Brain Metastases
 Optimal Management of Isolated HER2+ve Brain Metastases Eliot Sims November 2013 Background Her2+ve patients 15% of all breast cancer Even with adjuvant trastuzumab 10-15% relapse Trastuzumab does not
Optimal Management of Isolated HER2+ve Brain Metastases Eliot Sims November 2013 Background Her2+ve patients 15% of all breast cancer Even with adjuvant trastuzumab 10-15% relapse Trastuzumab does not
Gliomas account for 28% of the nearly 70,000 primary
 See the corresponding editorial in this issue (E2). Neurosurg Focus 37 (6):E1, 2014 AANS, 2014 Stereotactic laser ablation of high-grade gliomas Ammar H. Hawasli, M.D., Ph.D., 1 Albert H. Kim, M.D., Ph.D.,
See the corresponding editorial in this issue (E2). Neurosurg Focus 37 (6):E1, 2014 AANS, 2014 Stereotactic laser ablation of high-grade gliomas Ammar H. Hawasli, M.D., Ph.D., 1 Albert H. Kim, M.D., Ph.D.,
Cancer Cell Research 14 (2017)
 Available at http:// www.cancercellresearch.org ISSN 2161-2609 Efficacy and safety of bevacizumab for patients with advanced non-small cell lung cancer Ping Xu, Hongmei Li*, Xiaoyan Zhang Department of
Available at http:// www.cancercellresearch.org ISSN 2161-2609 Efficacy and safety of bevacizumab for patients with advanced non-small cell lung cancer Ping Xu, Hongmei Li*, Xiaoyan Zhang Department of
ORE Open Research Exeter
 ORE Open Research Exeter TITLE Temozolomide for high grade glioma AUTHORS Hart, MG; Garside, R; Rogers, G; et al. JOURNAL Cochrane Database Of Systematic Reviews DEPOSITED IN ORE 22 October 2013 This version
ORE Open Research Exeter TITLE Temozolomide for high grade glioma AUTHORS Hart, MG; Garside, R; Rogers, G; et al. JOURNAL Cochrane Database Of Systematic Reviews DEPOSITED IN ORE 22 October 2013 This version
CHMP Type II variation assessment report
 26 January 2017 EMA/CHMP/59238/2017 Invented name: Avastin International non-proprietary name: bevacizumab Procedure No. EMEA/H/C/000582/II/0093 Marketing authorisation holder (MAH): Roche Registration
26 January 2017 EMA/CHMP/59238/2017 Invented name: Avastin International non-proprietary name: bevacizumab Procedure No. EMEA/H/C/000582/II/0093 Marketing authorisation holder (MAH): Roche Registration
PRESURGICAL PLANNING. Strongly consider neuropsychological evaluation before functional imaging study Strongly consider functional imaging study
 NOTE: Consider Clinical Trials as treatment options for eligible patients. Page 1 of 6 RADIOLOGICAL PRESENTATION PRESURGICAL PLANNING TREATMENT Imaging study suggestive of glioma 1 Left hemisphere speech/motor
NOTE: Consider Clinical Trials as treatment options for eligible patients. Page 1 of 6 RADIOLOGICAL PRESENTATION PRESURGICAL PLANNING TREATMENT Imaging study suggestive of glioma 1 Left hemisphere speech/motor
Local alkylating chemotherapy applied immediately after 5-ALA guided resection of glioblastoma does not provide additional benefit
 https://doi.org/10.1007/s11060-017-2649-8 CLINICAL STUDY Local alkylating chemotherapy applied immediately after 5-ALA guided resection of glioblastoma does not provide additional benefit William Sage
https://doi.org/10.1007/s11060-017-2649-8 CLINICAL STUDY Local alkylating chemotherapy applied immediately after 5-ALA guided resection of glioblastoma does not provide additional benefit William Sage
Chemotherapy in Adults with Gliomas
 364 Chemotherapy in Adults with Gliomas Siew-Ju See and Mark R Gilbert Review Article Chemotherapy in Adults with Gliomas Siew-Ju See, 1 MBBS (S pore), MRCP (UK), Mark R Gilbert, 2 MD Abstract Treating
364 Chemotherapy in Adults with Gliomas Siew-Ju See and Mark R Gilbert Review Article Chemotherapy in Adults with Gliomas Siew-Ju See, 1 MBBS (S pore), MRCP (UK), Mark R Gilbert, 2 MD Abstract Treating
Clinical Trials for Adult Brain Tumors - the Imaging Perspective
 Clinical Trials for Adult Brain Tumors - the Imaging Perspective Whitney B. Pope, M.D., Ph.D. Department of Radiology David Geffen School of Medicine at UCLA August 22, 2015 1 Disclosure of Financial Relationships
Clinical Trials for Adult Brain Tumors - the Imaging Perspective Whitney B. Pope, M.D., Ph.D. Department of Radiology David Geffen School of Medicine at UCLA August 22, 2015 1 Disclosure of Financial Relationships
Zurich Open Repository and Archive. Long-term survival of glioblastoma patients treated with radiotherapy and lomustine plus temozolomide
 University of Zurich Zurich Open Repository and Archive Winterthurerstr. 19 CH-857 Zurich http://www.zora.uzh.ch Year: 29 Long-term survival of glioblastoma patients treated with radiotherapy and lomustine
University of Zurich Zurich Open Repository and Archive Winterthurerstr. 19 CH-857 Zurich http://www.zora.uzh.ch Year: 29 Long-term survival of glioblastoma patients treated with radiotherapy and lomustine
University of Alberta. Evaluation of Concomitant Temozolomide Treatment in Glioblastoma Multiforme Patients in Two Canadian Tertiary Care Centers
 University of Alberta Evaluation of Concomitant Temozolomide Treatment in Glioblastoma Multiforme Patients in Two Canadian Tertiary Care Centers by Ibrahim Alnaami A thesis submitted to the Faculty of
University of Alberta Evaluation of Concomitant Temozolomide Treatment in Glioblastoma Multiforme Patients in Two Canadian Tertiary Care Centers by Ibrahim Alnaami A thesis submitted to the Faculty of
Incidence of Early Pseudo-progression in a Cohort of Malignant Glioma Patients Treated With Chemoirradiation With Temozolomide
 405 Incidence of Early Pseudo-progression in a Cohort of Malignant Glioma Patients Treated With Chemoirradiation With Temozolomide Walter Taal, MD 1 Dieta Brandsma, MD, PhD 1 Hein G. de Bruin, MD, PhD
405 Incidence of Early Pseudo-progression in a Cohort of Malignant Glioma Patients Treated With Chemoirradiation With Temozolomide Walter Taal, MD 1 Dieta Brandsma, MD, PhD 1 Hein G. de Bruin, MD, PhD
Non-small Cell Lung Cancer: Multidisciplinary Role: Role of Medical Oncologist
 Non-small Cell Lung Cancer: Multidisciplinary Role: Role of Medical Oncologist Vichien Srimuninnimit, MD. Medical Oncology Division Faculty of Medicine, Siriraj Hospital Outline Resectable NSCLC stage
Non-small Cell Lung Cancer: Multidisciplinary Role: Role of Medical Oncologist Vichien Srimuninnimit, MD. Medical Oncology Division Faculty of Medicine, Siriraj Hospital Outline Resectable NSCLC stage
Bevacizumab in combination with temozolomide and regional radiation therapy for up-front treatment of patients with newly-diagnosed glioblastoma
 Bevacizumab in combination with temozolomide and regional radiation therapy for up-front treatment of patients with newly-diagnosed glioblastoma Design and analysis of single-arm Phase II clinical trial
Bevacizumab in combination with temozolomide and regional radiation therapy for up-front treatment of patients with newly-diagnosed glioblastoma Design and analysis of single-arm Phase II clinical trial
Retrospective Study of The Corticosteroids Administration in Glioblastoma Patients as A Prognostic Factor in The Disease
 The Egyptian Journal of Hospital Medicine (July 2018) Vol. 72 (5), Page 4551-4555 Retrospective Study of The Corticosteroids Administration in Glioblastoma Patients as A Prognostic Factor in The Disease
The Egyptian Journal of Hospital Medicine (July 2018) Vol. 72 (5), Page 4551-4555 Retrospective Study of The Corticosteroids Administration in Glioblastoma Patients as A Prognostic Factor in The Disease
University of Colorado Cancer Center Brain Disease Site Schema
 GBM FIRST LINE 18 0376/Kazia Phase 2a study (Part 1): A phase 2a study to evaluate the safety, pharmacokinetics and clinical activity of the PI3K/mTOR inhibitor GDC 0084 administered to patients with glioblastoma
GBM FIRST LINE 18 0376/Kazia Phase 2a study (Part 1): A phase 2a study to evaluate the safety, pharmacokinetics and clinical activity of the PI3K/mTOR inhibitor GDC 0084 administered to patients with glioblastoma
Cerebel trial Any impact on the clinical practice? Antonio Frassoldati Oncologia Clinica - Ferrara
 Cerebel trial Any impact on the clinical practice? Antonio Frassoldati Oncologia Clinica - Ferrara CNS metastases in HER2+ BC The proportion of patients with HER2+ advanced breast cancer who have CNS metastases
Cerebel trial Any impact on the clinical practice? Antonio Frassoldati Oncologia Clinica - Ferrara CNS metastases in HER2+ BC The proportion of patients with HER2+ advanced breast cancer who have CNS metastases
Temozolomide with Radiotherapy for the Treatment of Malignant Gliomas, Center Experience
 Temozolomide with Radiotherapy for the Treatment of Malignant Gliomas, Center Experience *Ehab Abdou and **Mohamed Gaafar *Department of Radiation Oncology, Faculty of Medicine, Al-Azhar University, Cairo,
Temozolomide with Radiotherapy for the Treatment of Malignant Gliomas, Center Experience *Ehab Abdou and **Mohamed Gaafar *Department of Radiation Oncology, Faculty of Medicine, Al-Azhar University, Cairo,
NON-SURGICAL STRATEGY FOR ADULT EPENDYMOMA
 NON-SURGICAL STRATEGY FOR ADULT EPENDYMOMA Roberta Rudà Department of Neuro-Oncology University and City of Health and Science Hospital of Turin, Italy EORTC EANO ESMO Conference 2015 Istanbul, March 27-28
NON-SURGICAL STRATEGY FOR ADULT EPENDYMOMA Roberta Rudà Department of Neuro-Oncology University and City of Health and Science Hospital of Turin, Italy EORTC EANO ESMO Conference 2015 Istanbul, March 27-28
Durable Response Rate in High Grade Glioma: an Emerging Endpoint for Immunotherapeutics. Timothy Cloughesy, MD University of California, Los Angeles
 Durable Response Rate in High Grade Glioma: an Emerging Endpoint for Immunotherapeutics Timothy Cloughesy, MD University of California, Los Angeles Disclosure 2 FDA Endpoints for the Approval of Cancer
Durable Response Rate in High Grade Glioma: an Emerging Endpoint for Immunotherapeutics Timothy Cloughesy, MD University of California, Los Angeles Disclosure 2 FDA Endpoints for the Approval of Cancer
Brain and Spine Tumors
 Brain and Spine Tumors Andrew J. Fabiano, MD FAANS Associate Professor of Neurosurgery Roswell Park Cancer Institute SUNY at Buffalo School of Medicine Brain Tumors Brain Tumor Basics Types of Tumors Cases
Brain and Spine Tumors Andrew J. Fabiano, MD FAANS Associate Professor of Neurosurgery Roswell Park Cancer Institute SUNY at Buffalo School of Medicine Brain Tumors Brain Tumor Basics Types of Tumors Cases
Tumor-Treatment Fields Therapy for Glioblastoma
 Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
Case 1 Metastatic Pancreatic Adenocarcinoma: What Therapy Should I Select First?
 Case 1 Metastatic Pancreatic Adenocarcinoma: What Therapy Should I Select First? Marc Peeters, MD, PhD Head of the Oncology Department Antwerp University Hospital Antwerp, Belgium marc.peeters@uza.be 71-year-old
Case 1 Metastatic Pancreatic Adenocarcinoma: What Therapy Should I Select First? Marc Peeters, MD, PhD Head of the Oncology Department Antwerp University Hospital Antwerp, Belgium marc.peeters@uza.be 71-year-old
The Role of Radiation Therapy in the Treatment of Brain Metastases. Matthew Cavey, M.D.
 The Role of Radiation Therapy in the Treatment of Brain Metastases Matthew Cavey, M.D. Objectives Provide information about the prospective trials that are driving the treatment of patients with brain
The Role of Radiation Therapy in the Treatment of Brain Metastases Matthew Cavey, M.D. Objectives Provide information about the prospective trials that are driving the treatment of patients with brain
Brain Tumors. Andrew J. Fabiano, MD FAANS. Associate Professor of Neurosurgery Roswell Park Cancer Institute SUNY at Buffalo School of Medicine
 Brain Tumors Andrew J. Fabiano, MD FAANS Associate Professor of Neurosurgery Roswell Park Cancer Institute SUNY at Buffalo School of Medicine Brain Tumors Brain Tumor Basics Types of Tumors Cases Brain
Brain Tumors Andrew J. Fabiano, MD FAANS Associate Professor of Neurosurgery Roswell Park Cancer Institute SUNY at Buffalo School of Medicine Brain Tumors Brain Tumor Basics Types of Tumors Cases Brain
Stereotactic Radiosurgery for Brain Metastasis: Changing Treatment Paradigms. Overall Clinical Significance 8/3/13
 Stereotactic Radiosurgery for Brain Metastasis: Changing Treatment Paradigms Jason Sheehan, MD, PhD Departments of Neurosurgery and Radiation Oncology University of Virginia, Charlottesville, VA USA Overall
Stereotactic Radiosurgery for Brain Metastasis: Changing Treatment Paradigms Jason Sheehan, MD, PhD Departments of Neurosurgery and Radiation Oncology University of Virginia, Charlottesville, VA USA Overall
NSCLC: immunotherapy as a first-line treatment. Paolo Bironzo Oncologia Polmonare AOU S. Luigi Gonzaga Orbassano (To)
 NSCLC: immunotherapy as a first-line treatment Paolo Bironzo Oncologia Polmonare AOU S. Luigi Gonzaga Orbassano (To) The 800-pound gorilla Platinum-based chemotherapy is the SOC for 1st-line therapy in
NSCLC: immunotherapy as a first-line treatment Paolo Bironzo Oncologia Polmonare AOU S. Luigi Gonzaga Orbassano (To) The 800-pound gorilla Platinum-based chemotherapy is the SOC for 1st-line therapy in
Gastroesophag Gastroesopha eal Junction Adenocarcinoma: What is the best adjuvant regimen? Michael G. G. H addock Haddock M.D.
 Gastroesophageal Junction Adenocarcinoma: What is the best adjuvant regimen? Michael G. Haddock M.D. Mayo Clinic Rochester, MN Locally Advanced GE Junction ACA CT S CT or CT S CT/RT Proposition Chemoradiation
Gastroesophageal Junction Adenocarcinoma: What is the best adjuvant regimen? Michael G. Haddock M.D. Mayo Clinic Rochester, MN Locally Advanced GE Junction ACA CT S CT or CT S CT/RT Proposition Chemoradiation
Management of Glioma: The Basics Glioma Update The clinical challenge. Glioma a malignant disease of the CNS
 Management of Glioma: The Basics Glioma Update 3 oger Stupp, MD Department of Oncology & Cancer Center University Hospital Zurich, Switzerland (roger.stupp@usz.ch) Bern, 3. August 3 The clinical challenge
Management of Glioma: The Basics Glioma Update 3 oger Stupp, MD Department of Oncology & Cancer Center University Hospital Zurich, Switzerland (roger.stupp@usz.ch) Bern, 3. August 3 The clinical challenge
