RESEARCH ARTICLE. Wanaporn Charoenchokthavee 1, Duangchit Panomvana Na Ayudhya 1, Virote Sriuranpong 2, Nutthada Areepium 1 * Abstract.
|
|
|
- Trevor Palmer
- 5 years ago
- Views:
Transcription
1 RESEARCH ARTICLE Effects of SULT1A1 Copy Number Variation on Estrogen Concentration and Tamoxifen-Associated Adverse Drug Reactions in Premenopausal Thai Breast Cancer Patients: A Preliminary Study Wanaporn Charoenchokthavee 1, Duangchit Panomvana Na Ayudhya 1, Virote Sriuranpong 2, Nutthada Areepium 1 * Abstract Tamoxifen is a pharmacological estrogen inhibitor that binds to the estrogen receptor (ER) in breast cells. However, it shows an estrogenic effect in other organs, which causes adverse drug reactions (ADRs). The sulfotransferase 1A1 (SULT1A1) enzyme encoded by the SULT1A1 gene is involved in estrogen metabolism. Previous research has suggested that the SULT1A1 copy number is linked with the plasma estradiol (E2) concentration. Here, a total of 34 premenopausal breast cancer patients, selected from the Thai Tamoxifen (TTAM) Project, were screened for their SULT1A1 copy number, plasma E2 concentration and ADRs. The mean age was 44.3 ± 11.1 years, and they were subtyped as ER+/ progesterone receptor (PR)+ (28 patients), ER+/ PR- (5 patients) and ER-/PR- (1 patient). Three patients reported ADRs, which were irregular menstruation (2 patients) and vaginal discharge (1 patient). Most (33) patients had two SULT1A1 copies, with one patient having three copies. The median plasma E2 concentration was 1,575.6 (IQR 865.4) pg/ml. Patients with ADRs had significantly higher plasma E2 concentrations than those patients without ADRs (p = 0.014). The plasma E2 concentration was numerically higher in the patient with three SULT1A1 copies, but this lacked statistical significance. Keywords: SULT1A1 - copy number variations - adverse drug reaction - estrogen - tamoxifen - breast cancer Asian Pac J Cancer Prev, 17 (4), Introduction Tamoxifen (TAM) has been widely used for antihormone therapy and provides a pharmacological effect in estrogen receptor (ER) positive (ER+) breast cancer patients, decreasing the recurrence of breast cancer and mortality rate by 50% and 30%, respectively (Klein et al., 2013). TAM is the trans-isomer of a triphenylethylene derivative (Dhingra, 1999) and has been used as an ER antagonist, where it competes with estrogen for binding to the ER and prevents cancer cell growth by its antiproliferative effect (Dhingra, 1999; Klein et al., 2013). In the absence of TAM treatment, estradiol (E2) exerts its activity by binding to the ERs present in the mammary gland and inducing a conformational change that then allows the link to co-activators to express cell proliferation (Del Re et al., 2012; Yang et al., 2013). In contrast, the binding of TAM to the ER increases the association with co-repressors that then actively inhibits gene transcription (Del Re et al., 2012). Furthermore, the ratio of the coactivators to co-repressors available to interact with the TAM-bound ERs determines whether TAM will behave as an antagonist or an agonist in different target organs (Dhingra, 1999). For example, TAM acts as ER-antagonist in the mammary gland but as an ER-agonist in the uterus where it can subsequently cause endometrial thickness (Dieudonne et al., 2014) or endometrial carcinoma (Maximov, 2013), a serious adverse drug reaction (ADR). However, the most common TAM-associated ADR is hot flashes (Kiyotani et al., 2012; Lorizio et al., 2012; Westbrook and Stearns, 2013), which result from the cessation of bound E2 on the ER. Estradiol is mainly produced in the ovary and shows marked changes in plasma levels during the menstrual cycle in premenopausal women, while plasma E2 levels are quite stable during a given month in postmenopausal women, which reflects the different E2 production sources. In postmenopausal women the plasma E2 is derived from transformed estrone (E1), which accounts for its low plasma concentration compared with the plasma E2 concentrations in premenopausal women (Gjerde et al., 2008). For example, premenopausal women have 1 Department of Pharmacy Practice, Faculty of Pharmaceutical Science, 2 Medical Oncology Unit, Department of Medicine, Faculty of Medicine, Chulalongkorn University, Bangkok, Thailand *For correspondence: Nutthada.a@pharm.chula.ac.th Asian Pacific Journal of Cancer Prevention, Vol 17,
2 Wanaporn Charoenchokthavee et al been reported to have a significantly higher (p < 0.001) plasma E2 concentration (median 96.5 pg/ml; range: pg/ml) than postmenopausal women (median 12.5 pg/ml; range pg/ml) (Matsui et al., 2013). For breast cancer patients, the plasma E2 concentrations were reported to range widely from ,150.0pg/ml in premenopausal breast cancer patients, depending on their menstrual patterns (Berliere et al., 2013), while in postmenopausal breast cancer patients they were less varied and lower at a median of 2.6 pg/ml and range of pg/ml (Gjerde et al., 2010). For both premenopausal and postmenopausal breast cancer patients, TAM is an estrogen antagonist of choice. However, previous studies have focused on the plasma E2 concentration in postmenopausal patients due to its stable concentration during the month (Gjerde et al., 2008) compared with the E2 concentrations in premenopausal women. Even though plasma E2 concentrations are difficult to compare among premenopausal patients due to the high degree of variability within the menstrual cycle, the study of plasma E2 concentrations and its metabolic pathway should not be neglected due to the outstanding pharmacological effect of TAM as an anti-estrogen agent in premenopausal breast cancer treatment. Estrogen metabolism also involves phase I (cytochrome P450) and phase II (sulfotransferase) metabolic enzymes, as well as the 17β-hydroxysteroid dehydrogenase type 1 (17β-HSD 1) and type 2 (17β-HSD 2) that reversibly convert E1 and E2 (Gjerde et al., 2010; Kallstrom et al., 2010). It was previously reported that CYP2D6, CYP3A5 and SULT1A1 polymorphisms did not affect the plasma E1 and E2 concentrations, whereas the SULT1A1 copy number was positively related to the plasma E1 and E2 concentrations (p=0.024 and 0.010, respectively) (Gjerde et al., 2010). The plasma E2 concentration was significantly decreased (level change -18.2%; p=0.004), while the E1 concentration was numerically but not significantly decreased (level change -6.9%; p=0.093), after TAM was administered in postmenopausal breast cancer patients (Lonning et al., 1995). In contrast, the plasma E2 concentrations were reported in a separate study to be significantly increased by 239% from 28 to 95pg/ml (p < 0.05) and the E1 concentrations to be numerically but not significantly increased by 264% from 42 pg/ml to 153 pg/ml (p=0.06) in postmenopausal patients, while the plasma E1 and E2 concentrations were numerically but not significantly increased in premenopausal patients (Lum et al., 1997). Previous studies have suggested that the presence of two copies of the SULT1A1 gene (SULT1A1x2) was the most common genotype in the Caucasian ( %) (Gjerde et al., 2010; Moyer et al., 2011) and Japanese (65.0%) (Yu et al., 2013) populations, while the copy number variation (CNV) ranged from one to five copies (Gjerde et al., 2010; Moyer et al., 2011; Yu et al., 2013). The SULT1A1 copy number has been suggested to be strongly associated with the SULT1A1 enzymatic activity (p=0.008), where patients with higher copy numbers of SULT1A1 genes showed a higher enzyme activity than those patients with just two copies (Yu et al., 2013). However, the relationship between the SULT1A1 CNV 1852 Asian Pacific Journal of Cancer Prevention, Vol 17, 2016 and the plasma E2 concentration has never been reported in Thai breast cancer patients. The objective of the present study was to determine the prevalence of SULT1A1 CNV, the plasma E2 concentration and TAM-associated ADRs, as well as their association in premenopausal Thai breast cancer patients undergoing TAM treatment. Materials and Methods Patients and sample preparation: Premenopausal breast cancer patients were randomly selected from the Thai Tamoxifen (TTAM) Project, for those patients who were taking 20 mg TAM once daily and visited the oncology outpatient clinic at King Chulalongkorn memorial hospital between February and March All patients were older than 18 years with normal hepatic and renal functions (aspartate aminotransferase and alanine aminotransferase <2 upper normal limit, serum creatinine <1.2 mg/dl) in the four weeks prior to selection for this study. Patients must have filled a prescription for TAM for at least 2 months and their medication adherence was evaluated by self-report. Their medication record was screened for drug-drug interaction by a clinical pharmacist. Patients who reported less than 80% adherence to the TAM administration regime, showed evident drug-drug interaction or were diagnosed for psychiatric illness/cognitive impairment were excluded from the study. For the remaining 34 patients, who then formed the study group, 10 ml of whole blood was drawn from each patient by a professional nurse and stored in a Vacutainer K2EDTA tube (BD, USA). The whole blood was separated into buffy coat and plasma sections. DNA extraction was prepared from the buffy coat section by using QIAamp DNA Mini kit (Qiagen, Netherlands) and used to determine the SULT1A1 copy number, while the plasma fraction was used for the E2 quantification. All DNA and plasma samples were stored at -20 o C and -80 o C, respectively, until analysis. The study protocol was approved by the Institutional Review Board of the Faculty of Medicine, Chulalongkorn University (IRB No.406/57). Determination of the SULT1A1 copy number: The DNA samples were diluted to a final concentration of 10 ng/μl with autoclaved ultra-pure water. The analysis was performed by TaqMan Universal PCR Master Mix, TaqMan Drug Metabolism Copy Number Assay Sets, Reference Assay for Copy Number determination with a Step One Plus real-time PCR system (Applied Biosystem, USA) and ViiA7 software (Applied Biosystems, USA). The thermal cycling for the qpcr in the TaqMan assay was 95 o C for 10 min followed by 40 cycles of 95 o C for 15 s and 60 o C for 1 min. Estradiol (E2) quantification: The Estradiol (E2) Bioassay TM ELISA Kit (Human) (United States Biological, USA) was employed to perform the plasma E2 concentration quantification, with all samples being analyzed within 6 months after sample collection. The detection range was 250-5,000 pg/ml and the sensitivity of the test was 1 pg/ml. The color intensity
3 was inversely proportional to the E2 concentration in the plasma sample and was measured spectrophotometrically at 450 nm in multi-label counter ( VICTOR3 ; PerkinElmer, USA). The plasma samples were brought to room temperature and mixed gently prior to the assay. Due to the low expected and found plasma E2 concentrations in the premenopausal breast cancer patients, sample dilution was not necessary in this study and blank plasma was subsequently used instead of PBS (dilution buffer). All standards (100 ul/well) and plasma samples (100 ul/ well) were added in duplicate to the wells of a 96-well plate. Standard E2 concentrations of 0, 250, 500, 1,000, 2,500 and 5,000 pg/ml were prepared for the calibration curve (E2 concentration versus the optical density) and the best fit line was derived by regression analysis. The E2 concentration of each sample was calculated by interpolating from the constructed E2 calibration curve. TAM-associated ADRs TAM-associated ADRs were individually evaluated by the hospital pharmacist based on data recorded in the patient s medical profile and from face-to-face interviews with the patients at recruitment time. Data analysis Data analysis was conducted by descriptive statistics using the SPSS (version 22) software. The Kolmogorov- Smirnov and Shapiro-Wilk tests were performed for normality tests, while the Kruskal-Wallis and Mann- Whitney U tests were performed for hypotheses testing of the non-parametric data. Results The 34 premenopausal breast cancer patients were categorized as being in stages 0-III of breast cancer (1, 13, 14 and 6 patients for stages 0 to III, respectively), with a mean age of 44.3±11.1 years and BMI of 23.4±3.6. The average duration of TAM administration in their treatment was 15.9 (IQR 34.1) months. Their hormone receptor statuses were ER+/PR+ (28 patients), ER+/ PR- (5 patients) and ER-/PR- (1 patient). Three patients (8.8%) reported having TAM-associated ADRs, which were irregular menstruation (2 patients) and vaginal discharge (1 patient) (Table 1). Most (33/34; 97.1%) patients had two copies of the SULT1A1 genes, while only one patient (2.9%) had three copies. The median plasma E2 concentration was 1,575.6 (IQR 865.4)pg/ml and ranged from ,186.5 pg/ml. The regression equation of the E2 calibration curve was y=-8.76 x 10-5 (x) ( R2 =0.864). Those three premenopausal patients who reported their TAM-associated ADRs had significantly higher E2 concentrations than those patients without reported ADRs (p=0.014, Table 2, Figure 1) but there was no significant (p=0.065) difference in the E2 concentrations among the two different TAM-associated ADRs symptoms (Table 2). The E2 concentration showed a trend to increase with more SULT1A1 copy numbers but, subject to the low sample size, this was not statistically Table 1. Demographic and clinical data of the premenopausal thai breast cancer patients Demographic and clinical data: Frequency (%) (N = 34) Age (years) No. (%) TAM administration No. (%) (2.9) < 6 months 6 (17.6) (38.2) 6 months-1 year 5 (14.7) (50.0) 1 year-3 years 12 (35.3) > 50 3 (8.8) > 3 years 11 (32.4) Hormone receptor status No. (%) TAM-associated ADRs No. (%) ER+ 33 (97.0) Irregular menstruation 2 (5.9) PR+ 28 (82.4) Vaginal discharge 1 (2.9) Table 2. Estradiol (E2) concentrations in different SULT1A1 copy numbers and TAM-associated ADRs SULT1A1 copy number N E2 concentration (pg/ml) p-value SULT1A1 x 2 copies 33 1,567.9 (IQR 829.1) a SULT1A1 x 3 copies 1 2,280.8 (IQR 0.0) TAM-associated ADRs N E2 concentration (pg/ml) p-value No ADRs presented 31 1,557.6 (IQR 740.9) 0.014* a ADRs presented 3 2,511.2 (IQR 0.0) b Irregular menstruation 2 3,067.5 (IQR 0.0) Vaginal discharge 1 2,511.2 (IQR 0.0) a Mann-Whitney U test, b Kruskal-Wallis test, *p-value<0.05 Figure 1. Estradiol (E2) concentration (pg/ml) between different TAM-associated ADRs. (The E2 concentrations of patient number 12 and 33 were 1,557.6 and 1,583.2 pg/ml, respectively.) Asian Pacific Journal of Cancer Prevention, Vol 17,
4 Wanaporn Charoenchokthavee et al significant (p=0.353) (Table 2). Discussion This is the first report on the CNV in the SULT1A1 gene in Thai premenopausal breast cancer patients. Two copies of SULT1A1 genes was the most common genotype (33/34) in premenopausal Thai breast cancer patients undergoing TAM treatment, which corresponds to that reported in other populations, except for the four copies reported in Caucasian (Gjerde et al., 2010; Moyer et al., 2011) and Japanese populations (Yu et al., 2013). In contrast to a previous study (Gjerde et al., 2010), no association between the SULT1A1 copy number and plasma E2 concentration was found. However, this might reflect the low sample size and/or the very low prevalence of other copy numbers in the Thai breast cancer population. The plasma E2 concentrations in these 34 premenopausal patients were highly variable and ranged from ,186.5 pg/ml, which was higher than the previously reported E2 concentrations (Lum et al., 1997; Gjerde et al., 2008). These differences might come from several factors. Firstly, the premenopausal breast patients recruited in this study had naturally higher plasma E2 concentrations compared with the postmenopausal breast cancer patients included in the previous study (Gjerde et al., 2010). Secondly, the E2 concentration was determined by ELISA in this study and by radioimmunoassay in the previous study (Lum et al., 1997). Thirdly, the patients had been using TAM at 20 mg once daily for at least two months in this study compared to at 10 mg twice a day for an undefined period in the previous study (Lum et al., 1997). Finally, the larger number of patients recruited in this study (N=34) compared with the previous study (N=6) (Lum et al., 1997), and this additionally may include different phases of the menstrual cycle that would yield different plasma E2 concentrations. The plasma E2 concentration increases in the follicular phase until reaching a peak and then decreases in the ovulatory phase, increases again in the luteal phase then decreases to begin the new menstrual cycle. Although the menstrual cycle was not controlled in this study, the menstrual phase during each patient s menstrual cycle was predicted by interviewing them at the start of the study. Overall the plasma E2 concentrations were not significantly (p=0.195) different among the different menstrual phases (follicular phase (10/25), ovulatory phase (6/25) and luteal phase (9/25)) from those patients who informed their menstrual starting date in this study (data not shown). An association between the plasma E2 concentration and TAM-associated ADRs was found in this study (p=0.017). The two TAM-associated ADRs in this preliminary study (irregular menstruation and vaginal discharge/dryness) might be a consequence of the estrogen antagonist effect of TAM. However, several factors need to be considered before reaching a firm conclusion. Firstly, the patient interview and medical record review were used to determine the TAM-associated ADRs in this study without using any other means of evaluation, and so the limitation of using retrospective or subjective data 1854 Asian Pacific Journal of Cancer Prevention, Vol 17, 2016 collection might have led to an over- or under-estimation of those ADRs. Other factors might need to be explored for controlling these possible confounders, such as the patient s level of follicle stimulating hormone and sex hormone-binding globulin and their individual E2 baseline concentrations. Secondly, the limited numbers of patients in this preliminary study could be problematic, especially as only three patients reported TAM-associated ADRs. Therefore, more patients need to be evaluated for this relationship before generalizing the result to the target population. Finally, those TAM-associated ADRs reported in this study did not include hot flashes, which are usually the most common TAM-associated ADRs in breast cancer patients. In conclusion, these findings provide the preliminary prevalence of SULT1A1 CNV in Thai premenopausal breast cancer patients undergoing TAM treatment, including the association between the plasma E2 concentration and TAM-associated ADRs. The high prevalence of SULT1A1x2 copies was firstly suggested from this study, which indicated that most of the Thai premenopausal breast cancer patients are carrying SULT1A1 genes with normal enzyme activities. References Berliere M, Duhoux FP, Dalenc F, et al (2013). Tamoxifen and ovarian function. PLoS ONE, 8, Del Re M, Michelucci A, Simi P, et al (2012). Pharmacogenetics of anti-estrogen treatment of breast cancer. Cancer Treat Rev, 38, Dhingra K (1999). Antiestrogens-Tamoxifen, SERMs and Beyond. Invest New Drug, 17, Dieudonne AS, Lambrechts D, Smeets D, et al (2014). The rs variant in CYP2D6 is associated with an increased double endometrial thickness in postmenopausal women on tamoxifen. Ann Oncol, 25, Gjerde J, Geisler J, Lundgren S, et al (2010). Associations between tamoxifen, estrogens, and FSH serum levels during steady state tamoxifen treatment of postmenopausal women with breast cancer. BMC Cancer, 10, 313. Gjerde J, Hauglid M, Breilid H, et al (2008). Effects of CYP2D6 and SULT1A1 genotypes including SULT1A1 gene copy number on tamoxifen metabolism. Ann Oncol, 19, Kallstrom A, Salme R, Ryden L, et al (2010). 17β-Hydroxysteroid dehydrogenase type 1 as predictor of tamoxifen response in premenopausal breast cancer. Eur J Cancer, 46, Kiyotani K, Mushiroda T, Imamura CK, et al (2012). Doseadjustment study of tamoxifen based on CYP2D6 genotypes in Japanese breast cancer patients. Breast Cancer Res Treat, 131, Klein DJ, Thorn CF, Desta Z, et al (2013). PharmGKB summary: Tamoxifen pathway, pharmacokinetics. Pharmacogenet Genomics, 23, Lonning P, Johannessen D, Lien E, et al (1995). Influence of tamoxifen on sex hormones, gonadotrophins and sex hormone binding globulin in postmenopausal breast cancer patients. J Steroid Biochem Mol Biol, 52, Lorizio W, Wu AHB, Beattie MS, et al (2012). Clinical and biomarker predictors of side effects from tamoxifen. Breast Cancer Res Treat, 132, Lum S, Woltering E, Fletcher W, et al (1997). Changes in serum estrogen levels in women during tamoxifen therapy. Am J Surg, 173,
5 Matsui S, Yasui T, Tani A, et al (2013). Associations of estrogen and testosterone with insulin resistance in pre- and postmenopausal women with and without hormone therapy. Int J Endocrinol Metab, 11, Moyer AM, Suman VJ, Weinshilboum RM, et al (2011). SULT1A1, CYP2C19 and disease-free survival in early breast cancer patients receiving tamoxifen. Pharmacogenomics, 12, Westbrook K, Stearns V (2013). Pharmacogenomics of breast cancer therapy: An update. Pharmacol Ther, 139, Yang G, Nowsheen S, Aziz K, et al (2013). Toxicity and adverse effects of Tamoxifen and other anti-estrogen drugs. Pharmacol Ther, 139, Yu X, Kubota T, Dhakal I, et al (2013). Copy number variation in sulfotransferase isoform 1A1 (SULT1A1) is significantly associated with enzymatic activity in Japanese subjects. Pharmgenomics Pers Med, 6, Asian Pacific Journal of Cancer Prevention, Vol 17,
Detection of Cytochrome P450 Polymorphisms in Breast Cancer Patients May Impact on Tamoxifen Therapy
 DOI:10.22034/APJCP.2018.19.2.343 Polymorphisms in Drug Metabolizing Genes RESEARCH ARTICLE Editorial Process: Submission:01/03/2017 Acceptance:10/27/2017 Detection of Cytochrome P450 Polymorphisms in Breast
DOI:10.22034/APJCP.2018.19.2.343 Polymorphisms in Drug Metabolizing Genes RESEARCH ARTICLE Editorial Process: Submission:01/03/2017 Acceptance:10/27/2017 Detection of Cytochrome P450 Polymorphisms in Breast
Design, Synthesis and Biological Screening of Focused Libraries of New Antiestrogens
 Design, Synthesis and Biological Screening of Focused Libraries of New Antiestrogens Presenter: Jelena Janjic, Dipl.Pharm. Graduate student Pharmaceutical Sciences Prof. Dr. Peter Wipf s group Dr. Billy
Design, Synthesis and Biological Screening of Focused Libraries of New Antiestrogens Presenter: Jelena Janjic, Dipl.Pharm. Graduate student Pharmaceutical Sciences Prof. Dr. Peter Wipf s group Dr. Billy
696 Biomed Environ Sci, 2015; 28(9):
 696 Biomed Environ Sci, 2015; 28(9): 696-700 Letter to the Editor Fluoride Exposure, Follicle Stimulating Hormone Receptor Gene Polymorphism and Hypothalamus-pituitary-ovarian Axis Hormones in Chinese
696 Biomed Environ Sci, 2015; 28(9): 696-700 Letter to the Editor Fluoride Exposure, Follicle Stimulating Hormone Receptor Gene Polymorphism and Hypothalamus-pituitary-ovarian Axis Hormones in Chinese
PERIMENOPAUSE. Objectives. Disclosure. The Perimenopause Perimenopause Menopause. Definitions of Menopausal Transition: STRAW.
 PERIMENOPAUSE Patricia J. Sulak, MD Founder, Living WELL Aware LLC Author, Should I Fire My Doctor? Author, Living WELL Aware: Eleven Essential Elements to Health and Happiness Endowed Professor Texas
PERIMENOPAUSE Patricia J. Sulak, MD Founder, Living WELL Aware LLC Author, Should I Fire My Doctor? Author, Living WELL Aware: Eleven Essential Elements to Health and Happiness Endowed Professor Texas
Melatonin and its Role in the Inhibition of Breast Cancer Ciara Nicol Ross Copyright 2014 by Ciara Ross and Koni Stone
 1 Melatonin and its Role in the Inhibition of Breast Cancer Ciara Nicol Ross Copyright 2014 by Ciara Ross and Koni Stone Cancer is a disease caused by out of control division of abnormal cells within a
1 Melatonin and its Role in the Inhibition of Breast Cancer Ciara Nicol Ross Copyright 2014 by Ciara Ross and Koni Stone Cancer is a disease caused by out of control division of abnormal cells within a
Estrone EIA kit. For the quantitative determination of estrone in dried fecal extracts, urine and tissue culture media. Cat. No.
 K-ASSAY KAMIYA BIOMEDICAL COMPANY KAMIYA BIOMEDICAL COMPANY Estrone EIA kit For the quantitative determination of estrone in dried fecal extracts, urine and tissue culture media Cat. No. KT-720 For Research
K-ASSAY KAMIYA BIOMEDICAL COMPANY KAMIYA BIOMEDICAL COMPANY Estrone EIA kit For the quantitative determination of estrone in dried fecal extracts, urine and tissue culture media Cat. No. KT-720 For Research
Summary
 Summary 118 This thesis is focused on the background of elevated levels of FSH in the early follicular phase of women with regular menstrual cycles. In the introduction (chapter 1) we describe the characteristics
Summary 118 This thesis is focused on the background of elevated levels of FSH in the early follicular phase of women with regular menstrual cycles. In the introduction (chapter 1) we describe the characteristics
CASE 41. What is the pathophysiologic cause of her amenorrhea? Which cells in the ovary secrete estrogen?
 CASE 41 A 19-year-old woman presents to her gynecologist with complaints of not having had a period for 6 months. She reports having normal periods since menarche at age 12. She denies sexual activity,
CASE 41 A 19-year-old woman presents to her gynecologist with complaints of not having had a period for 6 months. She reports having normal periods since menarche at age 12. She denies sexual activity,
Implications of Progesterone Receptor Status for the Biology and Prognosis of Breast Cancers
 日大医誌 75 (1): 10 15 (2016) 10 Original Article Implications of Progesterone Receptor Status for the Biology and Prognosis of Breast Cancers Naotaka Uchida 1), Yasuki Matsui 1), Takeshi Notsu 1) and Manabu
日大医誌 75 (1): 10 15 (2016) 10 Original Article Implications of Progesterone Receptor Status for the Biology and Prognosis of Breast Cancers Naotaka Uchida 1), Yasuki Matsui 1), Takeshi Notsu 1) and Manabu
Estrogens and progestogens
 Estrogens and progestogens Estradiol and Progesterone hormones produced by the gonads are necessary for: conception embryonic maturation development of primary and secondary sexual characteristics at puberty.
Estrogens and progestogens Estradiol and Progesterone hormones produced by the gonads are necessary for: conception embryonic maturation development of primary and secondary sexual characteristics at puberty.
Pharmacogenomics-based individualization of drug therapy
 ETH Zurich-JST Workshop on Medical Research, 15 Sep 2008 Pharmacogenomics-based individualization of drug therapy Taisei Mushiroda, Ph.D. Laboratory for Pharmacogenetics Center for Genomic Medicine, RIKEN
ETH Zurich-JST Workshop on Medical Research, 15 Sep 2008 Pharmacogenomics-based individualization of drug therapy Taisei Mushiroda, Ph.D. Laboratory for Pharmacogenetics Center for Genomic Medicine, RIKEN
Pharmacogenetics: DNA analysis. to explain / predict. response to drug therapy. Maurizio Ferrari & Ron van Schaik
 Maurizio Ferrari & Ron van Schaik Workshop IFCC Kuala Lumpur November 19, 2012 Predictive, Preventive and Personalized Medicine Part II: Pharmacogenetics l r.vanschaik@erasmusmc.nl Pharmacogenetics: DNA
Maurizio Ferrari & Ron van Schaik Workshop IFCC Kuala Lumpur November 19, 2012 Predictive, Preventive and Personalized Medicine Part II: Pharmacogenetics l r.vanschaik@erasmusmc.nl Pharmacogenetics: DNA
ab beta Estradiol ELISA Kit
 Version 4 Last updated 12 January 2018 ab108667 17 beta Estradiol ELISA Kit A competitive immunoenzymatic assay for the quantitative measurement of 17 beta Estradiol in serum and plasma. This product is
Version 4 Last updated 12 January 2018 ab108667 17 beta Estradiol ELISA Kit A competitive immunoenzymatic assay for the quantitative measurement of 17 beta Estradiol in serum and plasma. This product is
Natural Hormones Replacement An Evidence and Practice Based Approach
 Natural Hormones Replacement An Evidence and Practice Based Approach Andres Ruiz, PharmD, MSc, FACA President/Partner Stonegate Pharmacy PRESENTED BY THE AMERICAN COLLEGE OF APOTHECARIES 2830 SUMMER OAKS
Natural Hormones Replacement An Evidence and Practice Based Approach Andres Ruiz, PharmD, MSc, FACA President/Partner Stonegate Pharmacy PRESENTED BY THE AMERICAN COLLEGE OF APOTHECARIES 2830 SUMMER OAKS
Ultra-Sensitive Estradiol lumelisa Catalog No. GWB-AEB745, legacy id (96 Tests)
 For Research Use Only. Not for use in Diagnostic Procedures. INTENDED USE The GenWay, Inc. Ultra Sensitive Estradiol (E2) (Chemiluminescence Enzyme Linked Immunosorbent Assay) is used for the ultra sensitive
For Research Use Only. Not for use in Diagnostic Procedures. INTENDED USE The GenWay, Inc. Ultra Sensitive Estradiol (E2) (Chemiluminescence Enzyme Linked Immunosorbent Assay) is used for the ultra sensitive
Hormonal Control of Human Reproduction
 Hormonal Control of Human Reproduction Bởi: OpenStaxCollege The human male and female reproductive cycles are controlled by the interaction of hormones from the hypothalamus and anterior pituitary with
Hormonal Control of Human Reproduction Bởi: OpenStaxCollege The human male and female reproductive cycles are controlled by the interaction of hormones from the hypothalamus and anterior pituitary with
MICROWELL ELISA LUTEINIZING HORMONE (LH) ENZYMEIMMUNOASSAY TEST KIT LH ELISA. Cat # 4225Z
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
WEIGHT GAIN DURING MENOPAUSE EMERGING RESEARCH
 MENOPAUSE WHEN DOES IT OCCUR? The cessation of the menstrual cycle for one year. WEIGHT GAIN DURING MENOPAUSE EMERGING RESEARCH Jan Schroeder, Ph.D. Chair of The Department of Kinesiology California State
MENOPAUSE WHEN DOES IT OCCUR? The cessation of the menstrual cycle for one year. WEIGHT GAIN DURING MENOPAUSE EMERGING RESEARCH Jan Schroeder, Ph.D. Chair of The Department of Kinesiology California State
Month-Long Hormone Assessment: What goes up must come down. Disclaimer. Aims and Objectives. George Gillson MD PhD
 Month-Long Hormone Assessment: What goes up must come down George Gillson MD PhD 1 Disclaimer The information presented in this Webinar is the opinion of Dr. Gillson based on his research and experience.
Month-Long Hormone Assessment: What goes up must come down George Gillson MD PhD 1 Disclaimer The information presented in this Webinar is the opinion of Dr. Gillson based on his research and experience.
Female Reproductive System. Lesson 10
 Female Reproductive System Lesson 10 Learning Goals 1. What are the five hormones involved in the female reproductive system? 2. Understand the four phases of the menstrual cycle. Human Reproductive System
Female Reproductive System Lesson 10 Learning Goals 1. What are the five hormones involved in the female reproductive system? 2. Understand the four phases of the menstrual cycle. Human Reproductive System
The Science of your Cycle
 The Science of your Cycle Day 3: Get to know your cycle (Part I) with Jennifer Aldoretta Cofounder & CEO of Groove Today s goals Learn how your hormones work together to create the changes that happen
The Science of your Cycle Day 3: Get to know your cycle (Part I) with Jennifer Aldoretta Cofounder & CEO of Groove Today s goals Learn how your hormones work together to create the changes that happen
Hormone Balance - Female Report SAMPLE. result graph based on Luteal Phase. result graph based on Luteal Phase
 Patient Name: Patient DOB: Gender: Physician: Test Hormone Balance - Female Report SAMPLE Grote, Mary Jane Batch Number: B6437 2/16/1954 Accession Number: N52281 F Date Received: 2/3/2015 Any Lab Test
Patient Name: Patient DOB: Gender: Physician: Test Hormone Balance - Female Report SAMPLE Grote, Mary Jane Batch Number: B6437 2/16/1954 Accession Number: N52281 F Date Received: 2/3/2015 Any Lab Test
Lecture 10: Hormonal agents
 Lecture 10: Hormonal agents The survival and proliferation (growth) of some cancer cells is dependent on the action of steroid hormones. Early stage breast cancer and early stage prostate cancer are the
Lecture 10: Hormonal agents The survival and proliferation (growth) of some cancer cells is dependent on the action of steroid hormones. Early stage breast cancer and early stage prostate cancer are the
Hormone. Free Androgen Index. 2-Hydroxyestrone. Reference Range. Hormone. Estrone Ratio. Free Androgen Index
 Hormonal Health PATIENT: Sample Report TEST REF: TST-12345 Hormonal Health 0.61 0.30-1.13 ng/ml DHEA-S 91 35-430 mcg/dl tient: SAMPLE TIENT e: x: N: Sex Binding Globulin 80 18-114 nmol/l Testosterone 0.34
Hormonal Health PATIENT: Sample Report TEST REF: TST-12345 Hormonal Health 0.61 0.30-1.13 ng/ml DHEA-S 91 35-430 mcg/dl tient: SAMPLE TIENT e: x: N: Sex Binding Globulin 80 18-114 nmol/l Testosterone 0.34
Estrogen (ER) receptor biology: Implications for HRT? Dr. Christoph A. Meier Endocrine Unit University Hospital Geneva
 Estrogen (ER) receptor biology: Implications for HRT? Dr. Christoph A. Meier Endocrine Unit University Hospital Geneva Estrogens prevent postmenopausal bone-loss HRT & osteoporosis bip & estrogen bip
Estrogen (ER) receptor biology: Implications for HRT? Dr. Christoph A. Meier Endocrine Unit University Hospital Geneva Estrogens prevent postmenopausal bone-loss HRT & osteoporosis bip & estrogen bip
SAMPLE REPORT. Order Number: PATIENT. Age: 40 Sex: F MRN:
 Patient: Age: 40 Sex: F MRN: SAMPLE PATIENT Order Number: Completed: Received: Collected: SAMPLE REPORT Progesterone ng/ml 0.34 0.95 21.00 DHEA-S mcg/dl Testosterone ng/ml 48 35 0.10 0.54 0.80 430 Sex
Patient: Age: 40 Sex: F MRN: SAMPLE PATIENT Order Number: Completed: Received: Collected: SAMPLE REPORT Progesterone ng/ml 0.34 0.95 21.00 DHEA-S mcg/dl Testosterone ng/ml 48 35 0.10 0.54 0.80 430 Sex
Investigation: The Human Menstrual Cycle Research Question: How do hormones control the menstrual cycle?
 Investigation: The Human Menstrual Cycle Research Question: How do hormones control the menstrual cycle? Introduction: The menstrual cycle (changes within the uterus) is an approximately 28-day cycle that
Investigation: The Human Menstrual Cycle Research Question: How do hormones control the menstrual cycle? Introduction: The menstrual cycle (changes within the uterus) is an approximately 28-day cycle that
Ron van Schaik. Pharmacogenetics. London, Oct 8-9, Clinical implementation: a 7 year experience
 Ron van Schaik Associate Professor Pharmacogenetics Eur Clin Chem / Advisor EMA - PGWG London, Oct 8-9, 2012 Pharmacogenetics Clinical implementation: a 7 year experience Pharmacogenetics Core Laboratory*
Ron van Schaik Associate Professor Pharmacogenetics Eur Clin Chem / Advisor EMA - PGWG London, Oct 8-9, 2012 Pharmacogenetics Clinical implementation: a 7 year experience Pharmacogenetics Core Laboratory*
Interpreting follicular Progesterone: Late follicular Progesterone to Estradiol ratio is not influenced by protocols or gonadotropins used
 Interpreting follicular Progesterone: Late follicular Progesterone to Estradiol ratio is not influenced by protocols or gonadotropins used Ellenbogen A., M.D., Shalom-Paz E., M.D, Asalih N., M.D, Samara
Interpreting follicular Progesterone: Late follicular Progesterone to Estradiol ratio is not influenced by protocols or gonadotropins used Ellenbogen A., M.D., Shalom-Paz E., M.D, Asalih N., M.D, Samara
Reproductive physiology
 Reproductive physiology Sex hormones: Androgens Estrogens Gestagens Learning objectives 86 (also 90) Sex Genetic sex Gonadal sex Phenotypic sex XY - XX chromosomes testes - ovaries external features Tha
Reproductive physiology Sex hormones: Androgens Estrogens Gestagens Learning objectives 86 (also 90) Sex Genetic sex Gonadal sex Phenotypic sex XY - XX chromosomes testes - ovaries external features Tha
1. During the follicular phase of the ovarian cycle, the hypothalamus releases GnRH.
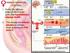 1. During the follicular phase of the ovarian cycle, the hypothalamus releases GnRH. 2. This causes the anterior pituitary to secrete small quantities of FSH and LH. 3. At this time, the follicles in the
1. During the follicular phase of the ovarian cycle, the hypothalamus releases GnRH. 2. This causes the anterior pituitary to secrete small quantities of FSH and LH. 3. At this time, the follicles in the
RESEARCH ARTICLE. Prevalence of CTR1 and ERCC1 Polymorphisms and Response of Biliary Tract Cancer to Gemcitabine-Platinum Chemotherapy
 RESEARCH ARTICLE Prevalence of CTR1 and ERCC1 Polymorphisms and Response of Biliary Tract Cancer to Gemcitabine-Platinum Chemotherapy Skolchart Pongmaneratanakul 1, Suebpong Tanasanvimon 2, Thitima Pengsuparp
RESEARCH ARTICLE Prevalence of CTR1 and ERCC1 Polymorphisms and Response of Biliary Tract Cancer to Gemcitabine-Platinum Chemotherapy Skolchart Pongmaneratanakul 1, Suebpong Tanasanvimon 2, Thitima Pengsuparp
PHARMACOGENETICS OF BREAST CANCER
 PHARMACOGENETICS OF BREAST CANCER MALGORZATA JAREMKO, PhD Mount Sinai School of Medicine, Department of Genetics and Genomic Sciences Outlines Breast cancer therapeutic situation Pharmacogenetics of antiestrogen
PHARMACOGENETICS OF BREAST CANCER MALGORZATA JAREMKO, PhD Mount Sinai School of Medicine, Department of Genetics and Genomic Sciences Outlines Breast cancer therapeutic situation Pharmacogenetics of antiestrogen
William J. Gradishar MD
 Northwestern University Feinberg School of Medicine Adjuvant Endocrine Therapy For Postmenopausal Women SOBO 2013 William J. Gradishar MD Betsy Bramsen Professor of Breast Oncology Director, Maggie Daley
Northwestern University Feinberg School of Medicine Adjuvant Endocrine Therapy For Postmenopausal Women SOBO 2013 William J. Gradishar MD Betsy Bramsen Professor of Breast Oncology Director, Maggie Daley
CYP2D6 and the oestrogen receptor
 Powered by Website address: https://www.gesundheitsindustriebw.de/en/article/news/cyp2d6-and-the-oestrogenreceptor/ CYP2D6 and the oestrogen receptor The medicinal adjuvant therapy of breast cancer is
Powered by Website address: https://www.gesundheitsindustriebw.de/en/article/news/cyp2d6-and-the-oestrogenreceptor/ CYP2D6 and the oestrogen receptor The medicinal adjuvant therapy of breast cancer is
N. Shirazian, MD. Endocrinologist
 N. Shirazian, MD Internist, Endocrinologist Inside the ovary Day 15-28: empty pyfollicle turns into corpus luteum (yellow body) Immature eggs Day 1-13: 13: egg developing inside the growing follicle Day
N. Shirazian, MD Internist, Endocrinologist Inside the ovary Day 15-28: empty pyfollicle turns into corpus luteum (yellow body) Immature eggs Day 1-13: 13: egg developing inside the growing follicle Day
Effects of ABCB1 3435C>T genotype on serum levels of cortisol and aldosterone in women with normal menstrual cycles
 Effects of ABCB1 3435C>T genotype on serum levels of cortisol and aldosterone in women with normal menstrual cycles T. Nakamura 1, N. Okamura 2, M. Yagi 1, H. Omatsu 3, M. Yamamori 3, A. Kuwahara 2, K.
Effects of ABCB1 3435C>T genotype on serum levels of cortisol and aldosterone in women with normal menstrual cycles T. Nakamura 1, N. Okamura 2, M. Yagi 1, H. Omatsu 3, M. Yamamori 3, A. Kuwahara 2, K.
Estrone. Enzyme Immunoassay Kit. 1 Plate Kit Catalog Number 1C Plate Kit Catalog Number 1C7871. Species Independent. Sample Types Validated:
 FT-1C7870 Estrone Enzyme Immunoassay Kit 1 Plate Kit Catalog Number 1C7870 5 Plate Kit Catalog Number 1C7871 Species Independent Sample Types Validated: Dried Fecal Extracts, Urine and Tissue Culture Media
FT-1C7870 Estrone Enzyme Immunoassay Kit 1 Plate Kit Catalog Number 1C7870 5 Plate Kit Catalog Number 1C7871 Species Independent Sample Types Validated: Dried Fecal Extracts, Urine and Tissue Culture Media
2016. All Rights Reserved. 1
 Pamela W. Smith, M.D., MPH, MS Copyright 2016 The suggested dosages are for educational purposes only. They are suggestions for patients with normal renal and hepatic function. They are based upon my research
Pamela W. Smith, M.D., MPH, MS Copyright 2016 The suggested dosages are for educational purposes only. They are suggestions for patients with normal renal and hepatic function. They are based upon my research
FSH pharmacogenetics
 5th World Congress on Ovulation Induction Roma, 13 15 September 2007 FSH pharmacogenetics Hermann M. Behre Center of Reproductive Medicine and Andrology, University Hospital Halle, Germany & Manuela Simoni
5th World Congress on Ovulation Induction Roma, 13 15 September 2007 FSH pharmacogenetics Hermann M. Behre Center of Reproductive Medicine and Andrology, University Hospital Halle, Germany & Manuela Simoni
FEP Medical Policy Manual
 FEP Medical Policy Manual Effective Date: October 15, 2018 Related Policies: 2.04.38 Cytochrome P450 Genotype-Guided Treatment Strategy Genotype-Guided Tamoxifen Treatment Description Tamoxifen is prescribed
FEP Medical Policy Manual Effective Date: October 15, 2018 Related Policies: 2.04.38 Cytochrome P450 Genotype-Guided Treatment Strategy Genotype-Guided Tamoxifen Treatment Description Tamoxifen is prescribed
Soyfood Consumption and Breast Cancer Survival. Xiao Ou Shu, M.D., Ph.D. Ingram Professor of Cancer Research Vanderbilt University, U.S.A.
 Soyfood Consumption and Breast Cancer Survival Xiao Ou Shu, M.D., Ph.D. Ingram Professor of Cancer Research Vanderbilt University, U.S.A. Objectives Brief summary of the health benefits of soyfood consumption
Soyfood Consumption and Breast Cancer Survival Xiao Ou Shu, M.D., Ph.D. Ingram Professor of Cancer Research Vanderbilt University, U.S.A. Objectives Brief summary of the health benefits of soyfood consumption
The Human Menstrual Cycle
 The Human Menstrual Cycle Name: The female human s menstrual cycle is broken into two phases: the Follicular Phase and the Luteal Phase. These two phases are separated by an event called ovulation. (1)
The Human Menstrual Cycle Name: The female human s menstrual cycle is broken into two phases: the Follicular Phase and the Luteal Phase. These two phases are separated by an event called ovulation. (1)
Reproductive. Estrone sulfate Analyte Information
 Reproductive Estrone sulfate Analyte Information - 1 - Estrone sulfate Introduction Estrone sulfate (E1-S) is a sulfate derivative of estrone, and is the most abundant form of circulating estrogens in
Reproductive Estrone sulfate Analyte Information - 1 - Estrone sulfate Introduction Estrone sulfate (E1-S) is a sulfate derivative of estrone, and is the most abundant form of circulating estrogens in
Synthesis of sex steroids
 Synthesis of sex steroids CH 3 NAD(P)H NAD(P)+ Dehidroepiandroszteron Androszténdion 17- -hidroxiszteroid dehidrogenáz CH 3 dehydroepiandrosterone Dehidroepiandroszteron (DHEA) 3SDH Koleszterin CH 3 aromatase
Synthesis of sex steroids CH 3 NAD(P)H NAD(P)+ Dehidroepiandroszteron Androszténdion 17- -hidroxiszteroid dehidrogenáz CH 3 dehydroepiandrosterone Dehidroepiandroszteron (DHEA) 3SDH Koleszterin CH 3 aromatase
TRHC.UEMEE Ph P y h si s o i logy l of fmen M str st u r at a i t on i
 . TRHCUEMEE Physiology of Menstruation Learning objectives: By the end of this lecture the students should be able to: Define menstruation. List the Characters of normal menstruation Enumerate Components
. TRHCUEMEE Physiology of Menstruation Learning objectives: By the end of this lecture the students should be able to: Define menstruation. List the Characters of normal menstruation Enumerate Components
5. Summary of Data Reported and Evaluation
 326 5. Summary of Data Reported and Evaluation 5.1 Exposure data Combined estrogen progestogen menopausal therapy involves the co-administration of an estrogen and a progestogen to peri- or postmenopausal
326 5. Summary of Data Reported and Evaluation 5.1 Exposure data Combined estrogen progestogen menopausal therapy involves the co-administration of an estrogen and a progestogen to peri- or postmenopausal
Labrix Clinical Services, Inc.
 Labrix Clinical Services, Inc. Advantages of Labrix Clinical Services, Inc. We Cater to the Healthcare Practitioner Labrix sample collection tubes are small; only 1 ml of saliva is required from the patient.
Labrix Clinical Services, Inc. Advantages of Labrix Clinical Services, Inc. We Cater to the Healthcare Practitioner Labrix sample collection tubes are small; only 1 ml of saliva is required from the patient.
Reproductive Health and Pituitary Disease
 Reproductive Health and Pituitary Disease Janet F. McLaren, MD Assistant Professor Division of Reproductive Endocrinology and Infertility Department of Obstetrics and Gynecology jmclaren@uabmc.edu Objectives
Reproductive Health and Pituitary Disease Janet F. McLaren, MD Assistant Professor Division of Reproductive Endocrinology and Infertility Department of Obstetrics and Gynecology jmclaren@uabmc.edu Objectives
SPRMs, androgens and breast cancer
 SPRMs, androgens and breast cancer Dr Joëlle Desreux BMS 04/06/2016 WHI : the nuclear explosion Looking for the perfect menopause treatment Hot flushes relieve Osteoporosis prevention Cardio-vascular health
SPRMs, androgens and breast cancer Dr Joëlle Desreux BMS 04/06/2016 WHI : the nuclear explosion Looking for the perfect menopause treatment Hot flushes relieve Osteoporosis prevention Cardio-vascular health
Estrogens & Antiestrogens
 Estrogens & Antiestrogens Menstrual cycle... Changes and hormonal events Natural estrogens: Estadiol >> Estrone > Estriol Ineffective orally Synthesis: From cholesterol ; role of aromatase enzyme in converting
Estrogens & Antiestrogens Menstrual cycle... Changes and hormonal events Natural estrogens: Estadiol >> Estrone > Estriol Ineffective orally Synthesis: From cholesterol ; role of aromatase enzyme in converting
SOFTly: The Long Natural History of [Trials for] [premenopausal] ER+ Breast Cancer
![SOFTly: The Long Natural History of [Trials for] [premenopausal] ER+ Breast Cancer SOFTly: The Long Natural History of [Trials for] [premenopausal] ER+ Breast Cancer](/thumbs/95/125806264.jpg) SOFTly: The Long Natural History of [Trials for] [premenopausal] ER+ Breast Cancer Charles Moertel Lecture May 12, 2017 Gini Fleming Charles Moertel Founder of NCCTG Dedication to high quality clinical
SOFTly: The Long Natural History of [Trials for] [premenopausal] ER+ Breast Cancer Charles Moertel Lecture May 12, 2017 Gini Fleming Charles Moertel Founder of NCCTG Dedication to high quality clinical
Estradiol level 70. been reported that if concentrations of estradiol in a 70- year-old man are compared to those of a 70-year-old
 Your browser does not support script The Estradiol Test measures the amount of estradiol (a female sex hormone which is basically a form of estrogen) in your blood. Any Lab Test Now Estradiol is a type
Your browser does not support script The Estradiol Test measures the amount of estradiol (a female sex hormone which is basically a form of estrogen) in your blood. Any Lab Test Now Estradiol is a type
ROLE OF HORMONAL ASSAY IN DIAGNOSING PCOD DR GAANA SREENIVAS (JSS,MYSURU)
 ROLE OF HORMONAL ASSAY IN DIAGNOSING PCOD DR GAANA SREENIVAS (JSS,MYSURU) In 1935, Stein and Leventhal described 7 women with bilateral enlarged PCO, amenorrhea or irregular menses, infertility and masculinizing
ROLE OF HORMONAL ASSAY IN DIAGNOSING PCOD DR GAANA SREENIVAS (JSS,MYSURU) In 1935, Stein and Leventhal described 7 women with bilateral enlarged PCO, amenorrhea or irregular menses, infertility and masculinizing
Serum levels of 25-OH vitamin D, folic acid and testosterone in patients with breast cancer: a case control study
 Asian Biomedicine Vol. 5 No. 5 October 2011; 663-667 Brief communication (Original) DOI: 10.5372/1905-7415.0505.097 Serum levels of 25-OH vitamin D, folic acid and testosterone in patients with breast
Asian Biomedicine Vol. 5 No. 5 October 2011; 663-667 Brief communication (Original) DOI: 10.5372/1905-7415.0505.097 Serum levels of 25-OH vitamin D, folic acid and testosterone in patients with breast
 Mdi Medical Management of Breast Cancer Morbidity and Mortality Aug 13, 2009 Irina Kovatch, PGY3 Introduction Metastatic disease is the principal cause of death from breast cancer Metastatic events often
Mdi Medical Management of Breast Cancer Morbidity and Mortality Aug 13, 2009 Irina Kovatch, PGY3 Introduction Metastatic disease is the principal cause of death from breast cancer Metastatic events often
9.4 Regulating the Reproductive System
 9.4 Regulating the Reproductive System The Reproductive System to unite a single reproductive cell from a female with a single reproductive cell from a male Both male and female reproductive systems include
9.4 Regulating the Reproductive System The Reproductive System to unite a single reproductive cell from a female with a single reproductive cell from a male Both male and female reproductive systems include
The Relation between Estrogen-positive Receptor in Breast Cancer (ER+) and Obesity in Jeddah
 https://www.lexispublisher.com/biology-todaysworld.html doi: 10.15412/J.JBTW.01070203 The Relation between Estrogen-positive Receptor in Breast Cancer (ER+) and Obesity in Jeddah Sabah Abdulaziz Linjawi1*,
https://www.lexispublisher.com/biology-todaysworld.html doi: 10.15412/J.JBTW.01070203 The Relation between Estrogen-positive Receptor in Breast Cancer (ER+) and Obesity in Jeddah Sabah Abdulaziz Linjawi1*,
Soyfood Consumption and Breast Cancer Survival. Xiao Ou Shu, M.D., Ph.D. Ingram Professor of Cancer Research Vanderbilt University, U.S.A.
 Soyfood Consumption and Breast Cancer Survival Xiao Ou Shu, M.D., Ph.D. Ingram Professor of Cancer Research Vanderbilt University, U.S.A. Objectives Brief summary of the health benefits of soyfood consumption
Soyfood Consumption and Breast Cancer Survival Xiao Ou Shu, M.D., Ph.D. Ingram Professor of Cancer Research Vanderbilt University, U.S.A. Objectives Brief summary of the health benefits of soyfood consumption
Breast Cancer Risk in Patients Using Hormonal Contraception
 Breast Cancer Risk in Patients Using Hormonal Contraception Bradley L. Smith, Pharm.D. Smith.bradley1@mayo.edu Pharmacy Ground Rounds Mayo Clinic Rochester April 3 rd, 2018 2017 MFMER slide-1 Presentation
Breast Cancer Risk in Patients Using Hormonal Contraception Bradley L. Smith, Pharm.D. Smith.bradley1@mayo.edu Pharmacy Ground Rounds Mayo Clinic Rochester April 3 rd, 2018 2017 MFMER slide-1 Presentation
Adipose tissue and related gynecological cancers George Valsamakis
 Adipose tissue and related gynecological cancers George Valsamakis Εndocrinologist Visiting Associate clinical Professor Warwick Medical School, UK European Scope Fellow in Obesity (IASO) Obesity and
Adipose tissue and related gynecological cancers George Valsamakis Εndocrinologist Visiting Associate clinical Professor Warwick Medical School, UK European Scope Fellow in Obesity (IASO) Obesity and
For the quantitative measurement of Human Estradiol 17-beta concentrations in plasma and serum
 ab108667 Estradiol 17-beta Human ELISA Kit Instructions for Use For the quantitative measurement of Human Estradiol 17-beta concentrations in plasma and serum This product is for research use only and
ab108667 Estradiol 17-beta Human ELISA Kit Instructions for Use For the quantitative measurement of Human Estradiol 17-beta concentrations in plasma and serum This product is for research use only and
Endocrinology of the Female Reproductive Axis
 Endocrinology of the Female Reproductive Axis girlontheriver.com Geralyn Lambert-Messerlian, PhD, FACB Professor Women and Infants Hospital Alpert Medical School at Brown University Women & Infants BROWN
Endocrinology of the Female Reproductive Axis girlontheriver.com Geralyn Lambert-Messerlian, PhD, FACB Professor Women and Infants Hospital Alpert Medical School at Brown University Women & Infants BROWN
Innovative Nuclear Receptor Modulation Medicine
 Innovative Nuclear Receptor Modulation Medicine Dale C Leitman, M.D., Ph.D. Isaac Cohen, O.M.D., Ph.D. October 2015 Corporate Mission Iaterion, Inc. is Developing Novel Pharmaceutical Nuclear Receptor
Innovative Nuclear Receptor Modulation Medicine Dale C Leitman, M.D., Ph.D. Isaac Cohen, O.M.D., Ph.D. October 2015 Corporate Mission Iaterion, Inc. is Developing Novel Pharmaceutical Nuclear Receptor
REFERENCE NUMBER: NH.PST.05 EFFECTIVE DATE: 10/10
 PAGE: 1 of 5 IMPORTANT REMINDER This Clinical Policy has been developed by appropriately experienced and licensed health care professionals based on a thorough review and consideration of generally accepted
PAGE: 1 of 5 IMPORTANT REMINDER This Clinical Policy has been developed by appropriately experienced and licensed health care professionals based on a thorough review and consideration of generally accepted
The Latest Research: Hormonal Therapies
 The Latest Research: Hormonal Therapies Sameer Gupta, M.D., M.P.H 9/29/2018 Attending Physician, Hematology/Oncology Bryn Mawr Hospital Clinical Assistant Professor, Jefferson Medical College Disclosures
The Latest Research: Hormonal Therapies Sameer Gupta, M.D., M.P.H 9/29/2018 Attending Physician, Hematology/Oncology Bryn Mawr Hospital Clinical Assistant Professor, Jefferson Medical College Disclosures
Clinical Oncology - Science in focus - Editorial. Understanding oestrogen receptor function in breast cancer, and its interaction with the
 Clinical Oncology - Science in focus - Editorial TITLE: Understanding oestrogen receptor function in breast cancer, and its interaction with the progesterone receptor. New preclinical findings and their
Clinical Oncology - Science in focus - Editorial TITLE: Understanding oestrogen receptor function in breast cancer, and its interaction with the progesterone receptor. New preclinical findings and their
Test Results SB Samples Arrived: 01/15/2014 Samples Collected: Saliva: 01/11/14 07:14 Date Closed: 01/18/2014
 Test Results 8605 SW Creekside Place Beaverton, OR 97008 Phone: 503-466-2445 Fax: 503-466-1636 info@zrtlab.com www.zrtlab.com 2014 01 15 001 SB Samples Arrived: 01/15/2014 Samples Collected: Saliva: 01/11/14
Test Results 8605 SW Creekside Place Beaverton, OR 97008 Phone: 503-466-2445 Fax: 503-466-1636 info@zrtlab.com www.zrtlab.com 2014 01 15 001 SB Samples Arrived: 01/15/2014 Samples Collected: Saliva: 01/11/14
Chapter 28: REPRODUCTIVE SYSTEM: MALE
 Chapter 28: REPRODUCTIVE SYSTEM: MALE I. FUNCTIONAL ANATOMY (Fig. 28.1) A. Testes: glands which produce male gametes, as well as glands producing testosterone 2. Seminiferous tubules (Fig.28.3; 28.5) a.
Chapter 28: REPRODUCTIVE SYSTEM: MALE I. FUNCTIONAL ANATOMY (Fig. 28.1) A. Testes: glands which produce male gametes, as well as glands producing testosterone 2. Seminiferous tubules (Fig.28.3; 28.5) a.
The Human Cathelicidin LL37 Peptide has High Plasma Levels in B and C Hepatitis Related to Viral Activity but not to 25-Hydroxyvitamin D Plasma Level
 The Human Cathelicidin LL37 Peptide has High Plasma Levels in B and C Hepatitis Related to Viral Activity but not to 25-Hydroxyvitamin D Plasma Level SIMONA ALEXANDRA IACOB 1, EUGENIA PANAITESCU 2, DIANA
The Human Cathelicidin LL37 Peptide has High Plasma Levels in B and C Hepatitis Related to Viral Activity but not to 25-Hydroxyvitamin D Plasma Level SIMONA ALEXANDRA IACOB 1, EUGENIA PANAITESCU 2, DIANA
Medical risk reducing strategies for breast cancer
 Medical risk reducing strategies for breast cancer PROF. DR. H. DEPYPERE Menopause Clinic, University Hospital, Ghent, Belgium Life expectancy in Belgium 46,6 y in 1880 and 83,8 y in 2014 2 Women Men Aantal
Medical risk reducing strategies for breast cancer PROF. DR. H. DEPYPERE Menopause Clinic, University Hospital, Ghent, Belgium Life expectancy in Belgium 46,6 y in 1880 and 83,8 y in 2014 2 Women Men Aantal
The serum estradiol/oocyte ratio in patients with breast cancer undergoing ovarian stimulation with letrozole and gonadotropins
 Original Article Obstet Gynecol Sci 2018;61(2):242-246 https://doi.org/10.5468/ogs.2018.61.2.242 pissn 2287-8572 eissn 2287-8580 The serum estradiol/oocyte ratio in patients with breast cancer undergoing
Original Article Obstet Gynecol Sci 2018;61(2):242-246 https://doi.org/10.5468/ogs.2018.61.2.242 pissn 2287-8572 eissn 2287-8580 The serum estradiol/oocyte ratio in patients with breast cancer undergoing
ab Estradiol E2 Human ELISA Kit
 ab108640 Estradiol E2 Human ELISA Kit Instructions for Use A competitive immunoenzymatic assay for the quantitative measurement of Estradiol E2 in Human Serum. This product is for research use only and
ab108640 Estradiol E2 Human ELISA Kit Instructions for Use A competitive immunoenzymatic assay for the quantitative measurement of Estradiol E2 in Human Serum. This product is for research use only and
RNA preparation from extracted paraffin cores:
 Supplementary methods, Nielsen et al., A comparison of PAM50 intrinsic subtyping with immunohistochemistry and clinical prognostic factors in tamoxifen-treated estrogen receptor positive breast cancer.
Supplementary methods, Nielsen et al., A comparison of PAM50 intrinsic subtyping with immunohistochemistry and clinical prognostic factors in tamoxifen-treated estrogen receptor positive breast cancer.
Rat Mullerian Inhibiting Substance/Anti-Mullerian hormone, MIS/AMH ELISA kit
 Rat Mullerian Inhibiting Substance/Anti-Mullerian hormone, MIS/AMH ELISA kit Catalog No. E0228r (96 tests) Operating instruction www.eiaab.com FOR RESEARCH USE ONLY; NOT FOR THERAPEUTIC OR DIAGNOSTIC APPLICATIONS!
Rat Mullerian Inhibiting Substance/Anti-Mullerian hormone, MIS/AMH ELISA kit Catalog No. E0228r (96 tests) Operating instruction www.eiaab.com FOR RESEARCH USE ONLY; NOT FOR THERAPEUTIC OR DIAGNOSTIC APPLICATIONS!
Prediction of HBsAg Loss by Quantitative HBsAg Kinetics during Long-Term 2015
 THAI J 16 GASTROENTEROL Treatment with Nucleos(t)ide Original Analogues Article Prediction of HBsAg Loss by Quantitative HBsAg Kinetics during Long-Term Treatment with Nucleos(t)ide Analogues Sombutsook
THAI J 16 GASTROENTEROL Treatment with Nucleos(t)ide Original Analogues Article Prediction of HBsAg Loss by Quantitative HBsAg Kinetics during Long-Term Treatment with Nucleos(t)ide Analogues Sombutsook
Learning Objectives. Oncology I: Solid Tumors Last updated: August 2010
 Learning Objectives Oncology I: Solid Tumors Last updated: August 2010 Upon completion of this program, participants will be able to: Identify specific drug therapies used in cardiology in which pharmacogenomic
Learning Objectives Oncology I: Solid Tumors Last updated: August 2010 Upon completion of this program, participants will be able to: Identify specific drug therapies used in cardiology in which pharmacogenomic
Endocrine Steroids 2. Signal transduction 3. Prostaglandins
 Endocrine - 2 1. Steroids 2. Signal transduction 3. Prostaglandins Estrogen Menopause (pause in the menes) ["change of life" at about 50] - lack of estrogen. (Some hysterectomy or ovarian cancer surgeries
Endocrine - 2 1. Steroids 2. Signal transduction 3. Prostaglandins Estrogen Menopause (pause in the menes) ["change of life" at about 50] - lack of estrogen. (Some hysterectomy or ovarian cancer surgeries
Original Article Impact of estrogen-to-oocyte ratio on live birth rate in women undergoing in vitro fertilization and embryo transfer
 Int J Clin Exp Med 2015;8(7):11327-11331 www.ijcem.com /ISSN:1940-5901/IJCEM0008838 Original Article Impact of estrogen-to-oocyte ratio on live birth rate in women undergoing in vitro fertilization and
Int J Clin Exp Med 2015;8(7):11327-11331 www.ijcem.com /ISSN:1940-5901/IJCEM0008838 Original Article Impact of estrogen-to-oocyte ratio on live birth rate in women undergoing in vitro fertilization and
Extended Hormonal Therapy
 Extended Hormonal Therapy Dr. Caroline Lohrisch, Medical Oncologist, BC Cancer Agency Vancouver Centre November 1, 2014 www.fpon.ca Optimal Endocrine Therapy for Women with Hormone Receptor Positive Early
Extended Hormonal Therapy Dr. Caroline Lohrisch, Medical Oncologist, BC Cancer Agency Vancouver Centre November 1, 2014 www.fpon.ca Optimal Endocrine Therapy for Women with Hormone Receptor Positive Early
Urinary Melatonin Levels and Risk of Postmenopausal Breast Cancer in the Women's Health Initiative Observational Study
 University of Massachusetts Amherst ScholarWorks@UMass Amherst Masters Theses 1911 - February 2014 2012 Urinary Melatonin Levels and Risk of Postmenopausal Breast Cancer in the Women's Health Initiative
University of Massachusetts Amherst ScholarWorks@UMass Amherst Masters Theses 1911 - February 2014 2012 Urinary Melatonin Levels and Risk of Postmenopausal Breast Cancer in the Women's Health Initiative
Sample Provincial exam Q s: Reproduction
 Sample Provincial exam Q s: Reproduction 11. Functions Testosterone Makes the male sex organs function normally, and also inhibits hypothalamus s release of GnRH and thus LH & FSH and thus testosterone
Sample Provincial exam Q s: Reproduction 11. Functions Testosterone Makes the male sex organs function normally, and also inhibits hypothalamus s release of GnRH and thus LH & FSH and thus testosterone
VVA : new therapeutic options? BMS october 2017 A Pintiaux
 VVA : new therapeutic options? BMS october 2017 A Pintiaux Disclosures Member of the european board of TEVA without personal gain No conflict of interest for this presentation VVA Vulvar and vaginal atrophy
VVA : new therapeutic options? BMS october 2017 A Pintiaux Disclosures Member of the european board of TEVA without personal gain No conflict of interest for this presentation VVA Vulvar and vaginal atrophy
Crosstalk between Adiponectin and IGF-IR in breast cancer. Prof. Young Jin Suh Department of Surgery The Catholic University of Korea
 Crosstalk between Adiponectin and IGF-IR in breast cancer Prof. Young Jin Suh Department of Surgery The Catholic University of Korea Obesity Chronic, multifactorial disorder Hypertrophy and hyperplasia
Crosstalk between Adiponectin and IGF-IR in breast cancer Prof. Young Jin Suh Department of Surgery The Catholic University of Korea Obesity Chronic, multifactorial disorder Hypertrophy and hyperplasia
Jim Paoletti BS Pharmacy, FAARM, FIACP, Director of Education, P2P
 presents Converting Patients from Conventional Pioneering Technologies For to Bioidentical Lifestyle Based Medicine Hormone Therapy with Jim Paoletti BS Pharmacy, FAARM, FIACP, Director of Education, P2P
presents Converting Patients from Conventional Pioneering Technologies For to Bioidentical Lifestyle Based Medicine Hormone Therapy with Jim Paoletti BS Pharmacy, FAARM, FIACP, Director of Education, P2P
Uterine Fibroids: No financial disclosures. Current Challenges, Promising Future. Off-label uses of drugs. Alison Jacoby, MD.
 Uterine Fibroids: Current Challenges, Promising Future Alison Jacoby, MD Professor, Dept of Obstetrics, Gynecology and Reproductive Sciences No financial disclosures Off-label uses of drugs The BIG Questions
Uterine Fibroids: Current Challenges, Promising Future Alison Jacoby, MD Professor, Dept of Obstetrics, Gynecology and Reproductive Sciences No financial disclosures Off-label uses of drugs The BIG Questions
The 6 th Scientific Meeting of the Asia Pacific Menopause Federation
 Predicting the menopause The menopause marks the end of ovarian follicular activity and is said to have occurred after 12 months amenorrhoea. The average age of the menopause is between 45 and 60 years
Predicting the menopause The menopause marks the end of ovarian follicular activity and is said to have occurred after 12 months amenorrhoea. The average age of the menopause is between 45 and 60 years
Testosterone ELISA. Catalog No (96 Tests) For Research Use Only. Not for use in Diagnostic Procedures.
 For Research Use Only. Not for use in Diagnostic Procedures. INTENDED USE The GenWay, Inc. Testosterone ELISA is for the quantitative determination of Testosterone concentration in human serum or plasma
For Research Use Only. Not for use in Diagnostic Procedures. INTENDED USE The GenWay, Inc. Testosterone ELISA is for the quantitative determination of Testosterone concentration in human serum or plasma
6. Clinical Use of Selective Estrogen Receptor Modulators and Aromatase Inhibitors in Prevention and Adjuvant Treatment of Breast Cancer
 6. Clinical Use of Selective Estrogen Receptor Modulators and Aromatase Inhibitors in Prevention and Adjuvant Treatment of Breast Cancer Saad J. Sirop, MD James N. Ingle, MD Matthew P. Goetz, MD Introduction
6. Clinical Use of Selective Estrogen Receptor Modulators and Aromatase Inhibitors in Prevention and Adjuvant Treatment of Breast Cancer Saad J. Sirop, MD James N. Ingle, MD Matthew P. Goetz, MD Introduction
Reproductive Testing: Less is More G. Wright Bates, Jr., M.D. Professor and Director Reproductive Endocrinology and Infertility Objectives
 Reproductive Testing: Less is More G. Wright Bates, Jr., M.D. Professor and Director Reproductive Endocrinology and Infertility Objectives 1. Review definition of infertility and impact of age 2. Stress
Reproductive Testing: Less is More G. Wright Bates, Jr., M.D. Professor and Director Reproductive Endocrinology and Infertility Objectives 1. Review definition of infertility and impact of age 2. Stress
SYSTEMIC THERAPY OPTIONS FOR BREAST CANCER IN 2014
 SYSTEMIC THERAPY OPTIONS FOR BREAST Oncology Day 2014 CANCER IN 2014 Dr. Katherine Enright, M.D., M.P.H., F.R.C.P.(C) Katherine.enright@Trilliumhealthpartners.ca OBJECTIVES 1. Outline an approach to the
SYSTEMIC THERAPY OPTIONS FOR BREAST Oncology Day 2014 CANCER IN 2014 Dr. Katherine Enright, M.D., M.P.H., F.R.C.P.(C) Katherine.enright@Trilliumhealthpartners.ca OBJECTIVES 1. Outline an approach to the
Risk of renal side effects with ADT. E. David Crawford University of Colorado, Aurora, CO, USA
 Risk of renal side effects with ADT E. David Crawford University of Colorado, Aurora, CO, USA ADT: A key treatment for advanced prostate cancer John Hunter 1780-castration 1904: First RP 1938: Acid Phos.
Risk of renal side effects with ADT E. David Crawford University of Colorado, Aurora, CO, USA ADT: A key treatment for advanced prostate cancer John Hunter 1780-castration 1904: First RP 1938: Acid Phos.
ER-P Expression in Different Thyroid Carcinomas: Immunohistochemical Study
 Med. J. Cairo Univ., Vol. 85, No. 6, September: 5-9, 17 www.medicaljournalofcairouniversity.net ER-P Expression in Different Thyroid Carcinomas: Immunohistochemical Study MIRAN A. EL-KORDY, M.Sc.; FAHIMA
Med. J. Cairo Univ., Vol. 85, No. 6, September: 5-9, 17 www.medicaljournalofcairouniversity.net ER-P Expression in Different Thyroid Carcinomas: Immunohistochemical Study MIRAN A. EL-KORDY, M.Sc.; FAHIMA
GONADAL FUNCTION: An Overview
 GONADAL FUNCTION: An Overview University of PNG School of Medicine & Health Sciences Division of Basic Medical Sciences Clinical Biochemistry BMLS III & BDS IV VJ Temple 1 What are the Steroid hormones?
GONADAL FUNCTION: An Overview University of PNG School of Medicine & Health Sciences Division of Basic Medical Sciences Clinical Biochemistry BMLS III & BDS IV VJ Temple 1 What are the Steroid hormones?
Bad to the bones: treatments for breast and prostate cancer
 12 th Annual Osteoporosis: New Insights in Research, Diagnosis, and Clinical Care 23 rd July 2015 Bad to the bones: treatments for breast and prostate cancer Richard Eastell, MD FRCP (Lond, Edin, Ireland)
12 th Annual Osteoporosis: New Insights in Research, Diagnosis, and Clinical Care 23 rd July 2015 Bad to the bones: treatments for breast and prostate cancer Richard Eastell, MD FRCP (Lond, Edin, Ireland)
Reproductive. Estradiol Analyte Information
 Reproductive Estradiol Analyte Information - 1 - Estradiol Introduction Estradiol (E2 or 17β-estradiol) is the major estrogen in humans. Although it is often called the "female" hormone, it is also present
Reproductive Estradiol Analyte Information - 1 - Estradiol Introduction Estradiol (E2 or 17β-estradiol) is the major estrogen in humans. Although it is often called the "female" hormone, it is also present
Breast Cancer and Bone Loss. One in seven women will develop breast cancer during a lifetime
 Breast Cancer and Bone Loss One in seven women will develop breast cancer during a lifetime Causes of Bone Loss in Breast Cancer Patients Aromatase inhibitors Bil Oophorectomy Hypogonadism Steroids Chemotherapy
Breast Cancer and Bone Loss One in seven women will develop breast cancer during a lifetime Causes of Bone Loss in Breast Cancer Patients Aromatase inhibitors Bil Oophorectomy Hypogonadism Steroids Chemotherapy
Fertility Diagnostics
 Fertility Diagnostics Fertility hormones measured on PATHFAST For internal use only Diagnostics PATHFAST Chemiluminescence-immuno-analyzer 1 Content: page 1. Fertility hormones - general aspects 1.1 Reproductive
Fertility Diagnostics Fertility hormones measured on PATHFAST For internal use only Diagnostics PATHFAST Chemiluminescence-immuno-analyzer 1 Content: page 1. Fertility hormones - general aspects 1.1 Reproductive
