Inducible nitric oxide synthase expression before and after eradication of Helicobacter pylori in di erent forms of gastritis
|
|
|
- Ashley McGee
- 6 years ago
- Views:
Transcription
1 FEMS Immunology and Medical Microbiology 30 (2001) 127^131 Inducible nitric oxide synthase expression before and after eradication of Helicobacter pylori in di erent forms of gastritis David Antos a, Georg Enders b, Gabriele Rieder c, Manfred Stolte d, Ekkehard Bayerdo«r er e, Rudolf A. Hatz a; * a Department of Surgery, Klinikum Grosshadern, Ludwig-Maximilians-University Munich, Marchioninistr. 15, Munich, Germany b Institute for Surgical Research, Klinikum Grosshadern, Ludwig-Maximilians-University Munich, Munich, Germany c Howard Hughes Research Institute, Ann Arbor, MI, USA d Department of Pathology, Klinikum Bayreuth, Bayreuth, Germany e Medical Department I, Technical University Hospital, Dresden, Germany Accepted 1 November 2000 Abstract An increased expression of inducible nitric oxide synthase (inos) has been observed in the inflamed human gastric mucosa as well as in some tumors. This observation suggests a pathobiological role of elevated NO production. The purpose of this study was to compare the immunohistochemical inos expression in the different kinds of gastritis before and after the eradication of Helicobacter pylori. We performed inos and H. pylori immunohistochemical staining and counted inos positive cells. We detected elevated expression of inos around sites infected with H. pylori. inos expression in chemical gastritis was strongly elevated in mucosal glands. After treatment, we found a significant difference in inos expression in patients with classical H. pylori-induced antrum predominant gastritis and in patients with active autoimmune gastritis. In the special case of progressed gastritis with intestinal metaplasia we found persistence of intestinal metaplasia, and we could not find a significant difference in the number of positive inos cells before and after treatment. The persistence of IM as a possibly precancerous lesion is probably at least in the antrum a source of continuous inos induction even after H. pylori eradication. ß 2001 Federation of European Microbiological Societies. Published by Elsevier Science B.V. All rights reserved. Keywords: Intestinal metaplasia; Gastritis; inos; Helicobacter pylori 1. Introduction Helicobacter pylori is now recognized as the pathogenic factor in chronic active gastritis [1^3]. On the basis of H. pylori-induced chronic active gastritis, peptic ulcer disease, i.e. duodenal ulcer or gastric ulcer disease, may develop [3,4]. Moreover, H. pylori infection is considered to be an important stage in the development of gastric MALT (mucosa-associated lymphoid tissue) lymphoma (marginal B- cell lymphoma in the REAL classi cation) [5,6]. Successful eradication of the bacteria could avoid development of these diseases [7,8]. Although the proof that H. pylori infection alone can result in the development of human gastric carcinoma is still lacking, in 1994 the World Health * Corresponding author. Tel.: +49(89) ; Fax: +49 (89) ; hatz@gch.med.uni-muenchen.de Organization classi ed H. pylori as a de nite human carcinogen based on the convincing, mainly epidemiological evidence [9]. Recently, persistent H. pylori infection in the Mongolian gerbil model was demonstrated. It showed that the sequential histopathological changes in the gastric mucosa closely mimic the gastric mucosal changes caused by H. pylori infection in humans and that H. pylori infection alone may cause gastric cancer in an animal model [10]. Nitric oxide (NO) is a free radical gas, which is synthesized from L-arginine by three isoforms of nitric oxide synthase (NOS). Two of these, the neuronal type (nnos) and the endothelial type (enos) are constitutively expressed, and their activities depend on Ca 2 levels. The inducible type (inos) is independent on Ca 2 levels. inos was rst characterized and isolated from macrophages and contributes to the cell-mediated host defense system. It has been increasingly observed in in amed human stomach as well as in gastrointestinal, gynecological and central nervous system tumors [11^13]. This observation suggests a / 01 / $20.00 ß 2001 Federation of European Microbiological Societies. Published by Elsevier Science B.V. All rights reserved. PII: S (00)
2 128 D. Antos et al. / FEMS Immunology and Medical Microbiology 30 (2001) 127^131 pathobiological role of in ammation- and tumor-associated NO production. Gastritis, or in ammation of the gastric mucosa, is not a single disease but rather a group of disorders which all induce in ammatory changes in the gastric mucosa but di er in their clinical features, histologic characteristics, and causative mechanisms. One of the classi cations divides the in ammation into three major forms. The most common form is antrum predominant H. pylori-associated gastritis (HAG) [14]. The so-called `gastric carcinoma risk index' gastritis is one subtype of HAG with more intense severity of in ammation in the corpus and it is associated with intestinal metaplasia (IMG). It is associated with a high risk for development of gastric cancer [15]. The second form is autoimmune gastritis (AIG), associated with parietal cell antibodies. Its chronic atrophic form is also associated with gastric cancer. Some recent studies showed the possible link between H. pylori antibodies and AIG [16^18] and its association with the active form of AIG [19,20]. The third major form is chemical gastritis (CG) found mostly in patients with intermittent or long-term use of nonsteroidal anti-in ammatory drugs (NSAIDs). The purpose of this study was to nd inos expression in IMG and AIG before and after eradication of H. pylori, compare it with HAG, where data already exist [26,29] and with CG. We also stained pre-eradication biopsies for H. pylori in order to nd any space relationship with positive inos cells. The results may illuminate a possible role of H. pylori infection and inos expression in the in amed stomach and changes which occur after elimination of the gastric intruder. 2. Materials and methods 2.1. Patients and therapy Twenty-four patients undergoing upper intestinal endoscopy were studied (13 men and 11 women, age range 30^ 77 years, mean age 57 years). Eighteen of them were additionally investigated 3 months after H. pylori eradication therapy. None of the patients had received antibiotics, bismuth compounds, H 2 -antagonists, or proton pump inhibitors during the previous 4 weeks. Patients with a present or past history of malignant disease, history of gastric surgery, contra-indications for the biopsy, or a known allergy against the study medication were excluded. Inclusion criteria were the histological ndings for each group of gastritis. There were six patients with HAG, six with active AIG, six with IMG, and six with CG. All patients with CG had a history of intermittent or longterm use of NSAIDs. None of the others had taken NSAIDs within 2 months before the study. All patients gave informed consent, and the study was approved by the ethics committee of the Surgical Faculty, Klinikum GroM hadern, University of Munich. For treatment of H. pylori infection, an approved therapy regimen which contains omeprazole 2U20 mg, clarithromycin 2U250 mg and amoxicillin 2U1000 mg, each given for 7 days before meals, was used [7] Tissue sampling, histopathology, and H. pylori status A total of four biopsy specimens were obtained from de ned adjacent locations in the antrum and corpus. All specimens were immediately placed in 3.7% neutral formalin and embedded in para n for histological examination. Histopathological classi cation of gastritis was performed on H and E-stained, para n embedded sections by one pathologist (M.S.) in accordance with the Sydney system and included assessment of atrophy grade, chronicity, activity, and intestinal metaplasia on a scale of 0 (absent) to 3 (high) as reported [21]. The presence and density of H. pylori colonization was identi ed by Warthin^Starry stain. Histopathological criteria for inclusion into the study were for HAG grade of chronicity and activity in the antrum at least 2 and positive H. pylori status; for IMG grade of chronicity and activity in the corpus (at least 2 or higher) v grade of chronicity and activity in the antrum, positive H. pylori status and presence of IM [15]. We deliberately avoided to discriminate between histological subtypes of IM (complete, incomplete, enterocolic), since in particular the enterocolic type of IM which is associated with a very high gastric cancer risk, is found in only 1^2% of German patients with IM and therefore would not be feasible for routine diagnostics; for active AIG-dense, diffuse locally emphasized lymphocytic in ltration of the lamina propria between the glands in the oxyntic mucosa, focal destruction of individual glands in the corpus of the stomach by lymphocytes, reactive pseudohypertrophy of the parietal cells [19] and positive H. pylori status; for CG di use foveolar hyperplasia and apical edema with slight brosis [22] Immunohistochemical analysis Immunohistochemistry was performed on formalin xed, para n embedded and cryostat cut (3-Wm) consecutive sections. The sections were depara nized and incubated in H 2 O 2 /methanol for 30 min at room temperature to destroy endogenous peroxidase activity. Nonspeci c immunoglobulins were blocked with 10% normal swine serum at room temperature for 45 min. Excess liquid was drained and sections were incubated in a humid chamber with 1:500 dilution of rabbit polyclonal antibody to mouse macrophage inos (Transduction Laboratories, Lexington, KY, USA). Sections were incubated overnight at 4³C. Similarly, for detection of H. pylori we also used rabbit polyclonal antibody (DAKO) in the humid chamber with 1:100 dilution at room temperature for 30 min. The next step was an incubation at room temperature for
3 D. Antos et al. / FEMS Immunology and Medical Microbiology 30 (2001) 127^ min with 1:200 goat anti-rabbit biotinylated IgG (Vector Laboratories, Burlingame, CA, USA). The biotinylated conjugate was detected by incubating sections with avidin-peroxidase 1:1000 dilution (Vector Laboratories) for 45 min. Immunolabeling was detected using DAB (Vector Laboratories) as the chromogen, followed by washing and staining with Mayer's hematoxylin. Positive and negative controls were included in every procedure. To quantify the extent of inos expression we used a calibrated eye-piece graticulate on a Leitz Laborlux D microscope (Leitz, Wetzlar, Germany). We investigated ve consecutive sections from the antrum and corpus from each patient. inos positive cells were counted at a magni cation of 400U. Each grid covered mm 2 and 10 consecutive grids per section were counted, which covered the total tissue area in most cases Statistical analysis All statistics were performed using SPSS statistical package (Version 10.0; SPSS Inc., Chicago, IL, USA). Di erences between values in two groups were compared using the Mann^Whitney U test for paired samples. A P value of was considered statistically signi cant. 3. Results and discussion NO, synthesized by inos, is a potent vasodilator that has di erent e ects depending on the particular conditions under which it is produced. Normally, it is involved in the physiological regulation of the gastric microcirculation and its integrity. Exceeding physiological levels as in HAG, NO could act as a cytotoxic not only for the microorganism but also for the cells and tissues and has therefore been called a `double-edged sword' [23]. In order to study the e ects of H. pylori eradication in the di erent forms of gastritis on inos expression we examined HAG, IMG, active AIG and CG. All our patients were successfully treated, which was con rmed by histopathology using Warthin^Starry staining for H. pylori. Chronicity and activity values decreased in all treated gastritis forms. Additionally, the histological ndings in the mucosa of the active AIG group showed reversibility of changes, which were considered to be inclusion criteria. In the case of HAG it was possible to con rm the data of previous studies regarding a decrease of inos expression after successful eradication of H. pylori [26,29]. Statistically signi cant di erences in the number of counted positive inos cells before and after treatment were found (antrum: 64.2 cells before vs cells after the eradication; *P ; Fig. 1). The inos expression in CG was strongly elevated in the mucosal glands. Surprisingly, a high number of inos positive cells (19.3) was found and there was a signi cant Fig. 1. Number of positive inos cells before and after eradication in active autoimmune gastritis (active AIG; corpus biopsy: 40.6 vs. 7.6 cells/ mm 2 ; *P ); and H. pylori-associated gastritis (HAG; antrum biopsy: 64.2 vs cells/ mm 2 ; *P ). Di erences were found in both groups after treatment. Number of positive inos cells in chemical gastritis (CG; antrum biopsy: 19.3 cells/ mm 2 ). di erence with the number of inos positive cells in patients with AIG following treatment for H. pylori (*P ). Active AIG is hypothesized as a rst step in the development of chronic AIG. More recent investigations have shown that in some patients H. pylori infection can induce antigastric antibodies and lead to atrophic autoimmune gastritis of the oxyntic mucosa [18]. The question thus arises as to whether this form of autoimmune gastritis can be healed by eradicating H. pylori. Our immunohistological and histopathological ndings con rm this hypothesis. A statistically signi cant decrease of inos expression was observed (corpus: 40.6 vs. 7.6; *P ; Fig. 1) combined with a reduction of autoaggressive in ammatory in ltrates with focal destruction of corpus glands and diminished hypertrophy of parietal cells. As far as we know, this is the rst study showing inos expression in this gastritis form. There was no signi cance found between active AIG and HAG concerning inos, neither in pre- Fig. 2. Number of positive inos cells before and after eradication in corpus predominant H. pylori gastritis with intestinal metaplasia (IMG; antrum: 35.6 vs cells/ mm 2 ; P s 0.05; corpus: 1.7 vs. 3.0 cells/ mm 2 ; P s 0.05). No di erences were found after treatment, neither in antrum nor in corpus.
4 130 D. Antos et al. / FEMS Immunology and Medical Microbiology 30 (2001) 127^131 nor in post-treatment results. This suggests similar pathological development based on the infection with H. pylori. It was possible to detect inos in macrophages and neutrophils. Neutrophils are a potential source of reactive nitrogen species (RNS, such as nitric oxide and peroxynitrite) and reactive oxygen species (ROS, such as superoxide and hydrogen peroxide) which are potentially genotoxic and probably one of the factors which contribute to gastric carcinogenesis [24]. A model proposed for the etiology of stomach cancer contains a sequence of events progressing from in ammation to atrophy, to metaplasia, to dysplasia, to carcinoma in situ, and nally to gastric carcinoma [25]. Our patients with IMG are one very interesting group in this sequence, because at this stage the possible histopathological reversibility would be of high clinical interest. In our investigations, the persistence of IM in all six IMG patients was observed. Additionally, inos expression after treatment showed no signi cant di erence, neither in the antrum, nor in the corpus (antrum: 35.6 vs. 18.8; corpus: 1.7 vs. 3.0, P s 0.05; Fig. 2). Elevated expression of inos around the sites infected with H. pylori was observed, thus supporting our hypothesis that inos is induced by H. pylori-associated in ammation. There is evidence that inos induces apoptosis [26]. H. pylori infection has a positive proliferative e ect on gastric epithelium [27]. A recently published study shows that the development of IM is characterized by much lower rates of apoptosis along with relatively increased proliferation, which may represent a crucial step in gastric carcinogenesis resulting from H. pylori infection [28]. In our IMG patients, persistence of IM 3 months after treatment was found. Statistically, our results also demonstrate no change of inos expression in the antrum and corpus in this group. This could be due to a small number (six) of investigated patients. But although there were similar preeradication gastritis parameters in the antrum for the HAG and the IMG group, inos expression in the antrum was initially signi cant lower in IMG than in HAG (*P ). A lower inos expression in the IMG group may induce lower but signi cantly unchanged apoptosis even after eradication of H. pylori. These data could con- rm the nding of Scotiniotis et al. [28]. It is therefore unclear if IMG patients do bene t from eradication. Furthermore prolonged IM combined with continuous inos expression could lead to carcinogenesis even in the absence of H. pylori. These ndings should be con rmed on higher number of patients. In summary, inos expression in stomach mucosa decreased in HAG and active AIG after successful H. pylori eradication. In the case of IMG we could not nd inos decrease, additionally we observed persistence of IM. Longer follow-up of these patients and further studies are necessary to clarify the possibility of development of carcinogenesis in this group. Acknowledgements We would like to thank Dr. G. Meimarakis for statistical guidance, Dr. S Rappel and Dr. H. Mu«ller for assistance with collections of histological sections, Ch. Otahal, T. Seher and M. Rolle for technical support. This work was sponsored by a grant of the Chiles Foundation, Portland, OR, USA, and by the Weigand-Stiftung, Munich, Germany (to R.A.H.). References [1] Korman, M.G., Marks, I.N., Hunt, R.H., Axon, A., Blaser, M.J., McCarthy, D.M. and Tytgat, G.N.J. (1993) Helicobacter pylori: a workshop review. Eur. J. Gastroenterol. Hepatol. 5, 963^967. [2] Graham, D.Y., Malaty, H.M., Evans, D.G., Evans, D.J., Klein, P.D. and Adam, E. (1991) Epidemiology of Helicobacter pylori in an asymptomatic population in the United States. Gastroenterology 100, 1495^1501. [3] NIH Consensus Conference (1994) Helicobacter pylori in peptic ulcer disease. J. Am. Med. Assoc. 272, 65^69. [4] Graham, D.Y. (1993) Treatment of peptic ulcers caused by Helicobacter pylori. N. Engl. J. Med. 328, 349^350. [5] Harris, N.L., Ja e, E.S., Stein, H., Banks, P.M., Chan, J.K., Cleary, M.L., Delsol, G., De Wolf-Peeters, C. and Falini, B. (1994) A revised European-American classi cation of lymphoid neoplasms: A proposal from the International Lymphoma Study Group. Blood 84, 1361^1392. [6] Wotherspoon, A.C., Ortiz Hidalgo, C., Falzon, M.R. and Isaacson, P.G. (1991) Helicobacter pylori-associated gastritis and primary B-cell gastric lymphoma. Lancet 338, 1175^1176. [7] Lind, T., Megraud, F., Unge, P., Bayerdo«r er, E., O'morain, C., Spiller, R., Veldhuyzen Van Zanten, S., Bardhan, K.D., Hellblom, M., Wrangstadh, M., Zeijlon, L. and Cederberg, C. (1999) The MACH2 study: role of omeprazole in eradication of Helicobacter pylori with 1-week triple therapies. Gastroenterology 116, 248^253. [8] Bayerdo«r er, E., Neubauer, A., Rudolph, B., Thiede, C., Lehn, N., Eidt, S. and Stolte, M. (1995) Regression of primary gastric lymphoma of mucosa-associated lymphoid tissue type after cure of Helicobacter pylori infection. MALT Lymphoma Study Group. Lancet 24, 1591^1594. [9] International Agency for Research on Cancer (1994) Schistosomes, liver ukes and humans. IARC, Vol. 61, Geneva. [10] Honda, S., Fujioka, T., Tokieda, M., Satoh, R., Nishizono, A. and Nasu, M. (1998) Development of Helicobacter pylori-induced gastric carcinoma in Mongolian gerbils. Cancer Res. 58, 4255^4259. [11] Duenas-Gonzalez, Isales, A., del Mar Abad-Hernandez, C.M., Gonzalez-Sarmiento, M., Sangueza, R. and Rodriguez-Commes, O. (1997) Expression of inducible nitric oxide synthase in breast cancer correlates with metastatic disease. Mod. Pathol. 10, 645^649. [12] Yagihashi, N., Kasajima, H., Sugai, S., Matsumoto, K., Ebina, Y., Morita, T., Murakami, T. and Yagihashi, S. (2000) Increased in situ expression of nitric oxide synthase in human colorectal cancer. Virchows Arch. 436, 109^114. [13] Thomsen, L.L. and Miles, D.W. (1998) Role of nitric oxide in tumour progression: lessons from human tumours. Cancer Metastasis Rev. 17, 107^118. [14] Hatz, R.A., Rieder, G., Stolte, M., Bayerdo«r er, E., Meimarakis, G., Schildberg, F.W. and Enders, G. (1997) Pattern of adhesion molecule expression on vascular endothelium in Helicobacter pylori-associated antral gastritis. Gastroenterology 112, 1908^1919. [15] Meining, A., Bayerdo«r er, E., Muller, P., Miehlke, S., Lehn, N.,
5 D. Antos et al. / FEMS Immunology and Medical Microbiology 30 (2001) 127^ Ho«lzel, D., Hatz, R. and Stolte, M. (1998) Gastric carcinoma risk index in patients infected with Helicobacter pylori. Virchows Arch. 432, 311^314. [16] Appelmelk, B.J., Simoons-Smit, I., Negrini, R., Moran, A.P., Aspinall, G.O., Forte, J.G., de Vries, T., Quan, H., Verboom, T., Maaskant, J.J., Ghiara, P., Kuipers, E.J., Bloemena, E., Tadema, T.M., Townsend, R.R., Tyagarajan, K., Crothers Jr., J.M., Monteiro, M.A. and de Graa, J. (1996) Potential role of molecular mimicry between Helicobacter pylori lipopolysaccharide and host Lewis blood group antigens in autoimmunity. Infect. Immun. 64, 2031^2040. [17] Faller, G., Steininger, H., Kra«nzlein, J., Maul, H., Kerkau, T., Hensen, J., Hahn, E.G. and Kirchner, T. (1997) Antigastric autoantibodies in Helicobacter pylori infection: implications of histological and clinical parameters of gastritis. Gut 41, 619^623. [18] Negrini, R., Savio, A., Poiesi, C., Appelmelk, B., Bu oli, F., Paterlini, A., Cesari, P., Gra eo, M., Vaira, D. and Franzin, G. (1996) Antigenic mimicry between Helicobacter pylori and gastric mucosa in the pathogenesis of body atrophic gastritis. Gastroenterology 111, 655^665. [19] Stolte, M., Baumann, K., Bethke, B., Ritter, M., Lauer, E. and Eidt, H. (1992) Active autoimmune gastritis without total atrophy of the glands. Z. Gastroenterol. 30, 729^735. [20] Stolte, M., Meier, E. and Meining, A. (1998) Cure of autoimmune gastritis by Helicobacter pylori eradication in a 21-year-old male. Z. Gastroenterol. 36, 641^643. [21] Bayerdo«r er, E., Lehn, N., Hatz, R., Mannes, G.A., Oertel, H., Sauerbruch, T. and Stolte, M. (1992) Di erence in expression of Helicobacter pylori gastritis in antrum and body. Gastroenterology 102, 1575^1582. [22] Dixon, M.F., Genta, R.M., Yardley, J.H. and Correa, P. (1996) Classi cation and grading of gastritis. Am. J. Surg. Pathol. 20, 1161^1181. [23] Schmidt, H.W. and Walter, U. (1994) NO at work. Cell 78, 919^925. [24] Tamir, S. and Tannenbaum, S.R. (1996) The role of nitric oxide (NO) in the carcinogenic process. Biochim. Biophys. Acta 1288, F31^F36. [25] Correa, P. (1988) A human model of gastric carcinogenesis. Cancer Res. 48, 3554^3560. [26] Mannick, E.E., Bravo, L.E., Zarama, G., Realpe, J.L., Zhang, X.J., Ruiz, B., Fontham, E.T., Mera, R., Miller, M.J. and Correa, P. (1996) Inducible nitric oxide synthase, nitrotyrosine, and apoptosis in Helicobacter pylori gastritis: e ect of antibiotics and antioxidants. Cancer Res. 15, 3238^3243. [27] Bechi, P., Balzi, M., Becciolini, A., Maugeri, A., Raggi, C.C., Amorosi, A. and Dei, R. (1996) Helicobacter pylori and cell proliferation of the gastric mucosa: possible implications for gastric carcinogenesis. Am. J. Gastroenterol. 91, 271^276. [28] Scotiniotis, I.A., Rokkas, T., Furth, E.E., Rigas, B. and Shi, S.J. (2000) Altered gastric epithelial cell kinetics in Helicobacter pyloriassociated intestinal metaplasia: implications for gastric carcinogenesis. Int. J. Cancer 85, 192^200. [29] Hahm, K.B., Lee, K.J., Choi, S.Y., Kim, J.H., Cho, S.W., Yim, H., Park, S.J. and Chung, M.H. (1997) Possibility of chemoprevention by the eradication of Helicobacter pylori: oxidative DNA damage and apoptosis in H. pylori infection. Am. J. Gastroenterol. 92, 1853^1857.
Gastric atrophy: use of OLGA staging system in practice
 Gastroenterology and Hepatology From Bed to Bench. 2016 RIGLD, Research Institute for Gastroenterology and Liver Diseases ORIGINAL ARTICLE Gastric atrophy: use of OLGA staging system in practice Mahsa
Gastroenterology and Hepatology From Bed to Bench. 2016 RIGLD, Research Institute for Gastroenterology and Liver Diseases ORIGINAL ARTICLE Gastric atrophy: use of OLGA staging system in practice Mahsa
Helicobacter pylori and Gastric Cancer
 Helicobacter pylori and Gastric Cancer MANFRED STOLTE, ALEXANDER MEINING Department of Pathology, Klinikum Bayreuth, Bayreuth, Germany Key Words. Cancer Gastritis Helicobacter pylori Prevention ABSTRACT
Helicobacter pylori and Gastric Cancer MANFRED STOLTE, ALEXANDER MEINING Department of Pathology, Klinikum Bayreuth, Bayreuth, Germany Key Words. Cancer Gastritis Helicobacter pylori Prevention ABSTRACT
Cyclooxygenase-2 Expression in Gastric Antral Mucosa Before and After Eradication of Helicobacter pylori Infection
 THE AMERICAN JOURNAL OF GASTROENTEROLOGY Vol. 94, No. 5, 1999 1999 by Am. Coll. of Gastroenterology ISSN 0002-9270/99/$20.00 Published by Elsevier Science Inc. PII S0002-9270(99)00126-4 Cyclooxygenase-2
THE AMERICAN JOURNAL OF GASTROENTEROLOGY Vol. 94, No. 5, 1999 1999 by Am. Coll. of Gastroenterology ISSN 0002-9270/99/$20.00 Published by Elsevier Science Inc. PII S0002-9270(99)00126-4 Cyclooxygenase-2
PDF hosted at the Radboud Repository of the Radboud University Nijmegen
 PDF hosted at the Radboud Repository of the Radboud University Nijmegen The following full text is a publisher's version. For additional information about this publication click this link. http://hdl.handle.net/2066/48400
PDF hosted at the Radboud Repository of the Radboud University Nijmegen The following full text is a publisher's version. For additional information about this publication click this link. http://hdl.handle.net/2066/48400
Index. Note: Page numbers of article titles are in boldface type.
 Note: Page numbers of article titles are in boldface type. A Adherence, to bismuth quadruple therapy, 543 546 Adjuvant therapy, probiotics as, 567 569 Age factors, in gastric cancer, 611 612, 616 AID protein,
Note: Page numbers of article titles are in boldface type. A Adherence, to bismuth quadruple therapy, 543 546 Adjuvant therapy, probiotics as, 567 569 Age factors, in gastric cancer, 611 612, 616 AID protein,
Helicobacter pylori Improved Detection of Helicobacter pylori
 DOI:http://dx.doi.org/10.7314/APJCP.2016.17.4.2099 RESEARCH ARTICLE Improved Detection of Helicobacter pylori Infection and Premalignant Gastric Mucosa Using Conventional White Light Source Gastroscopy
DOI:http://dx.doi.org/10.7314/APJCP.2016.17.4.2099 RESEARCH ARTICLE Improved Detection of Helicobacter pylori Infection and Premalignant Gastric Mucosa Using Conventional White Light Source Gastroscopy
Gastric MALT-lymphoma and Helicobacter pylori infection
 Aliment Pharmacol Ther 1997; 11 (Suppl. 1): 89 94. Gastric MALT-lymphoma and Helicobacter pylori infection E. BAYERDO RFFER, S. MIEHLKE, A. NEUBAUER & M. STOLTE* Department of Gastroenterology, Hepatology
Aliment Pharmacol Ther 1997; 11 (Suppl. 1): 89 94. Gastric MALT-lymphoma and Helicobacter pylori infection E. BAYERDO RFFER, S. MIEHLKE, A. NEUBAUER & M. STOLTE* Department of Gastroenterology, Hepatology
Lymphocytic Gastritis, Isolated Type Occurring in Family Members. A Case Report.
 Lymphocytic Gastritis, Isolated Type Occurring in Family Members. A Case Report. Alan Shienbaum, DO; AndriyPavlenko, MD; Jun Liu, MD, PhD; Janusz J Godyn, MD. Pathology Department, Kennedy University Hospitals,
Lymphocytic Gastritis, Isolated Type Occurring in Family Members. A Case Report. Alan Shienbaum, DO; AndriyPavlenko, MD; Jun Liu, MD, PhD; Janusz J Godyn, MD. Pathology Department, Kennedy University Hospitals,
Association of Helicobacter pylori infection with Atrophic gastritis in patients with Dyspepsia
 ADVANCES IN BIORESEARCH Adv. Biores., Vol 8 [3] May 2017: 137-141 2017 Society of Education, India Print ISSN 0976-4585; Online ISSN 2277-1573 Journal s URL:http://www.soeagra.com/abr.html CODEN: ABRDC3
ADVANCES IN BIORESEARCH Adv. Biores., Vol 8 [3] May 2017: 137-141 2017 Society of Education, India Print ISSN 0976-4585; Online ISSN 2277-1573 Journal s URL:http://www.soeagra.com/abr.html CODEN: ABRDC3
Original Article Increased lysozyme expression in gastric biopsies with intestinal metaplasia and pseudopyloric metaplasia
 www.ijcem.com/ijcem908003 Original Article Increased lysozyme in gastric biopsies with intestinal metaplasia and pseudopyloric metaplasia Carlos A Rubio, Ragnar Befrits Gastrointestinal and Liver Pathology
www.ijcem.com/ijcem908003 Original Article Increased lysozyme in gastric biopsies with intestinal metaplasia and pseudopyloric metaplasia Carlos A Rubio, Ragnar Befrits Gastrointestinal and Liver Pathology
Update on the pathological classification of gastritis. Hala El-Zimaity, M.D. M.S. Epidemiology McMaster University Hamilton, Ontario Canada
 Update on the pathological classification of gastritis Hala El-Zimaity, M.D. M.S. Epidemiology McMaster University Hamilton, Ontario Canada CLASSIFICATION GASTRITIS GASTROPATHY 1. Acute 2. Chronic 3. Uncommon
Update on the pathological classification of gastritis Hala El-Zimaity, M.D. M.S. Epidemiology McMaster University Hamilton, Ontario Canada CLASSIFICATION GASTRITIS GASTROPATHY 1. Acute 2. Chronic 3. Uncommon
Endoscopic atrophic classification before and after H. pylori eradication is closely associated with histological atrophy and intestinal metaplasia
 E311 before and after H. pylori eradication is closely associated with histological atrophy and intestinal metaplasia Authors Institution Masaaki Kodama, Tadayoshi Okimoto, Ryo Ogawa, Kazuhiro Mizukami,
E311 before and after H. pylori eradication is closely associated with histological atrophy and intestinal metaplasia Authors Institution Masaaki Kodama, Tadayoshi Okimoto, Ryo Ogawa, Kazuhiro Mizukami,
Helicobacter and gastritis
 1 Helicobacter and gastritis Dr. Hala Al Daghistani Helicobacter pylori is a spiral-shaped gram-negative rod. H. pylori is associated with antral gastritis, duodenal (peptic) ulcer disease, gastric ulcers,
1 Helicobacter and gastritis Dr. Hala Al Daghistani Helicobacter pylori is a spiral-shaped gram-negative rod. H. pylori is associated with antral gastritis, duodenal (peptic) ulcer disease, gastric ulcers,
The New England Journal of Medicine. Patients
 HELICOBACTER PYLORI INFECTION AND THE DEVELOPMENT OF GASTRIC CANCER NAOMI UEMURA, M.D., SHIRO OKAMOTO, M.D., SOICHIRO YAMAMOTO, M.D., NOBUTOSHI MATSUMURA, M.D., SHUJI YAMAGUCHI, M.D., MICHIO YAMAKIDO,
HELICOBACTER PYLORI INFECTION AND THE DEVELOPMENT OF GASTRIC CANCER NAOMI UEMURA, M.D., SHIRO OKAMOTO, M.D., SOICHIRO YAMAMOTO, M.D., NOBUTOSHI MATSUMURA, M.D., SHUJI YAMAGUCHI, M.D., MICHIO YAMAKIDO,
The Nobel Prize in Physiology or Medicine for 2005
 The Nobel Prize in Physiology or Medicine for 2005 jointly to Barry J. Marshall and J. Robin Warren for their discovery of "the bacterium Helicobacter pylori and its role in gastritis and peptic ulcer
The Nobel Prize in Physiology or Medicine for 2005 jointly to Barry J. Marshall and J. Robin Warren for their discovery of "the bacterium Helicobacter pylori and its role in gastritis and peptic ulcer
Acid-Peptic Diseases of the Stomach and Duodenum Including Helicobacter pylori and NSAIDs Prof. Sheila Crowe
 Acid-Peptic Diseases of the Stomach and Duodenum Including Helicobacter pylori and NSAIDs 1 Division of Gastroenterology UC San Diego School of Medicine Clinical presentations of Helicobacter pylori infection
Acid-Peptic Diseases of the Stomach and Duodenum Including Helicobacter pylori and NSAIDs 1 Division of Gastroenterology UC San Diego School of Medicine Clinical presentations of Helicobacter pylori infection
Gastritis and gastric atrophy Hala El-Zimaity a,b
 Gastritis and gastric Hala El-Zimaity a,b a Department of Pathology, McMaster University, Hamilton, Ontario, Canada and b Department of Medicine, Michael E. DeBakey Veterans Affairs Medical Center, Baylor
Gastritis and gastric Hala El-Zimaity a,b a Department of Pathology, McMaster University, Hamilton, Ontario, Canada and b Department of Medicine, Michael E. DeBakey Veterans Affairs Medical Center, Baylor
Regression of Advanced Gastric MALT Lymphoma after the Eradication of Helicobacter pylori
 Gut and Liver, Vol. 6, No. 2, April 2012, pp. 270-274 CASE REPORT Regression of Advanced Gastric MALT Lymphoma after the Eradication of Helicobacter pylori Soo-Kyung Park, Hwoon-Yong Jung, Do Hoon Kim,
Gut and Liver, Vol. 6, No. 2, April 2012, pp. 270-274 CASE REPORT Regression of Advanced Gastric MALT Lymphoma after the Eradication of Helicobacter pylori Soo-Kyung Park, Hwoon-Yong Jung, Do Hoon Kim,
A COMPARATIVE STUDY BETWEEN IMMUNOHISTOCHEMISTRY, HEMATOXYLIN & EOSIN AND GEIMSA STAIN FOR HELICOBACTER PYLORI DETECTION IN CHRONIC GASTRITIS
 Original Research Article Pathology International Journal of Pharma and Bio Sciences ISSN 0975-6299 A COMPARATIVE STUDY BETWEEN IMMUNOHISTOCHEMISTRY, HEMATOXYLIN & EOSIN AND GEIMSA STAIN FOR HELICOBACTER
Original Research Article Pathology International Journal of Pharma and Bio Sciences ISSN 0975-6299 A COMPARATIVE STUDY BETWEEN IMMUNOHISTOCHEMISTRY, HEMATOXYLIN & EOSIN AND GEIMSA STAIN FOR HELICOBACTER
Clinicopathologic Analysis of Lymphocytic Gastritis
 The Korean Journal of Pathology 2007; 41: 289-95 Clinicopathologic Analysis of Lymphocytic Gastritis Jeong Eun Hwang Young Ok Hong Dong Eun Song Se Jin Jang Eunsil Yu Department of Pathology, University
The Korean Journal of Pathology 2007; 41: 289-95 Clinicopathologic Analysis of Lymphocytic Gastritis Jeong Eun Hwang Young Ok Hong Dong Eun Song Se Jin Jang Eunsil Yu Department of Pathology, University
Histopathological Characteristics of Atrophic Gastritis in Adult Population
 Journal of Pharmacy and Pharmacology 3 (2015) 133-138 doi: 10.17265/2328-2150/2015.03.004 D DAVID PUBLISHING Histopathological Characteristics of Atrophic Gastritis in Adult Population Marija Milićević,
Journal of Pharmacy and Pharmacology 3 (2015) 133-138 doi: 10.17265/2328-2150/2015.03.004 D DAVID PUBLISHING Histopathological Characteristics of Atrophic Gastritis in Adult Population Marija Milićević,
Ethnic Distribution of Atrophic Autoimmune Gastritis in the United States
 Ethnic Distribution of Atrophic Autoimmune Gastritis in the United States Robert M. Genta, Regan Allen, Massimo Rugge Miraca Life Sciences Research Institute, Miraca Life Sciences, Irving, Texas UTSW University
Ethnic Distribution of Atrophic Autoimmune Gastritis in the United States Robert M. Genta, Regan Allen, Massimo Rugge Miraca Life Sciences Research Institute, Miraca Life Sciences, Irving, Texas UTSW University
Research Article Correlation between the Intensity of Helicobacter pylori Colonization and Severity of Gastritis
 Hindawi Gastroenterology Research and Practice Volume 2017, Article ID 8320496, 5 pages https://doi.org/10.1155/2017/8320496 Research Article Correlation between the Intensity of Helicobacter pylori Colonization
Hindawi Gastroenterology Research and Practice Volume 2017, Article ID 8320496, 5 pages https://doi.org/10.1155/2017/8320496 Research Article Correlation between the Intensity of Helicobacter pylori Colonization
ÐÑÏÓÊÅÊËÇÌÅÍÅÓ ÎÅÍÏÃËÙÓÓÅÓ ÁÍÁÊÏÉÍÙÓÅÉÓ ÅËËÇÍÙÍ ÅÑÅÕÍÇÔÙÍ
 ÐÑÏÓÊÅÊËÇÌÅÍÅÓ ÎÅÍÏÃËÙÓÓÅÓ ÁÍÁÊÏÉÍÙÓÅÉÓ ÅËËÇÍÙÍ ÅÑÅÕÍÇÔÙÍ 205 206 ÐÑÏÓÊÅÊËÇÌÅÍÅÓ ÎÅÍÏÃËÙÓÓÅÓ ÁÍÁÊÏÉÍÙÓÅÉÓ ÅËËÇÍÙÍ ÅÑÅÕÍÇÔÙÍ EPITHELIAL CELL TURNOVER IN NON-DYSPLASTIC GASTRIC MUCOSA ADJACENT TO EARLY AND
ÐÑÏÓÊÅÊËÇÌÅÍÅÓ ÎÅÍÏÃËÙÓÓÅÓ ÁÍÁÊÏÉÍÙÓÅÉÓ ÅËËÇÍÙÍ ÅÑÅÕÍÇÔÙÍ 205 206 ÐÑÏÓÊÅÊËÇÌÅÍÅÓ ÎÅÍÏÃËÙÓÓÅÓ ÁÍÁÊÏÉÍÙÓÅÉÓ ÅËËÇÍÙÍ ÅÑÅÕÍÇÔÙÍ EPITHELIAL CELL TURNOVER IN NON-DYSPLASTIC GASTRIC MUCOSA ADJACENT TO EARLY AND
- Helicobacter - THE EASE AND DIFFICULTY OF A NEW DISCOVERY. Robin Warren
 - Helicobacter - THE EASE AND DIFFICULTY OF A NEW DISCOVERY Robin Warren EARLY DAYS First reports 100 years ago considered spirochaetes 1940 Freedburg saw curved organisms in the stomach 1954 Palmer: Freedburg
- Helicobacter - THE EASE AND DIFFICULTY OF A NEW DISCOVERY Robin Warren EARLY DAYS First reports 100 years ago considered spirochaetes 1940 Freedburg saw curved organisms in the stomach 1954 Palmer: Freedburg
The effect of proton pump inhibitors on the gastric mucosal microenvironment
 Original papers The effect of proton pump inhibitors on the gastric mucosal microenvironment Yen-Chun Peng,,, A F, Lan-Ru Huang, A, C, E, Hui-Ching Ho, C, Chi-Sen Chang, C, E, Shou-Wu Lee, E, Ching-Chang
Original papers The effect of proton pump inhibitors on the gastric mucosal microenvironment Yen-Chun Peng,,, A F, Lan-Ru Huang, A, C, E, Hui-Ching Ho, C, Chi-Sen Chang, C, E, Shou-Wu Lee, E, Ching-Chang
Key words : low-grade MALT lymphoma, epithelial change, empty lamina propria, B-cell clonality, H. pylori, eradication, long-term follow-up
 Department of Pathology, The University of Tokushima School of Medicine, Tokushima, Japan ; Department of Oral and Maxillofacial Surgery, The University of Tokushima School of Dentistry, Tokushima, Japan
Department of Pathology, The University of Tokushima School of Medicine, Tokushima, Japan ; Department of Oral and Maxillofacial Surgery, The University of Tokushima School of Dentistry, Tokushima, Japan
Gastroenterology Tutorial
 Gastroenterology Tutorial Gastritis Poorly defined term that refers to inflammation of the stomach. Infection with H. pylori is the most common cause of gastritis. Most patients remain asymptomatic Some
Gastroenterology Tutorial Gastritis Poorly defined term that refers to inflammation of the stomach. Infection with H. pylori is the most common cause of gastritis. Most patients remain asymptomatic Some
Helicobacter pylori:an Emerging Pathogen
 Bacteriology at UW-Madison Bacteriology 330 Home Page Helicobacter pylori:an Emerging Pathogen by Karrie Holston, Department of Bacteriology University of Wisconsin-Madison Description of Helicobacter
Bacteriology at UW-Madison Bacteriology 330 Home Page Helicobacter pylori:an Emerging Pathogen by Karrie Holston, Department of Bacteriology University of Wisconsin-Madison Description of Helicobacter
~Helicobacter pylori, gastritis, and peptic ulceration
 17 Department of Pathology J I Wyatt Department of Medicine T M Shallcross J E Crabtree RV Heatley St James University Hospital, Leeds LS9 7TF Correspondence to: Dr J I Wyatt. Accepted for publication
17 Department of Pathology J I Wyatt Department of Medicine T M Shallcross J E Crabtree RV Heatley St James University Hospital, Leeds LS9 7TF Correspondence to: Dr J I Wyatt. Accepted for publication
Histopathology: gastritis and peptic ulceration
 Histopathology: gastritis and peptic ulceration These presentations are to help you identify, and to test yourself on identifying, basic histopathological features. They do not contain the additional factual
Histopathology: gastritis and peptic ulceration These presentations are to help you identify, and to test yourself on identifying, basic histopathological features. They do not contain the additional factual
Review article: potential gastrointestinal effects of long-term acid suppression with proton pump inhibitors
 Aliment Pharmacol Ther 2000; 14: 651±668. Review article: potential gastrointestinal effects of long-term acid suppression with proton pump inhibitors L. LAINE*, D. AHNEN, C.MCCLAINà, E.SOLCIA &J.H.WALSH±
Aliment Pharmacol Ther 2000; 14: 651±668. Review article: potential gastrointestinal effects of long-term acid suppression with proton pump inhibitors L. LAINE*, D. AHNEN, C.MCCLAINà, E.SOLCIA &J.H.WALSH±
Helicobacter 2008;13:1-6. Am J Gastroent 2007;102: Am J of Med 2004;117:31-35.
 An Update on Helicobacter pylori and Its Treatment Trenika Mitchell, PharmD, BCPS Clinical Assistant Professor University of Kentucky College of Pharmacy October 18, 2008 Objectives Review the epidemiology
An Update on Helicobacter pylori and Its Treatment Trenika Mitchell, PharmD, BCPS Clinical Assistant Professor University of Kentucky College of Pharmacy October 18, 2008 Objectives Review the epidemiology
Relationship of Blood Group Determinants on Helicobacter pylori Lipopolysaccharide with Host Lewis Phenotype and Inflammatory Response
 INFECTION AND IMMUNITY, Feb. 2000, p. 937 941 Vol. 68, No. 2 0019-9567/00/$04.00 0 Copyright 2000, American Society for Microbiology. All Rights Reserved. Relationship of Blood Group Determinants on Helicobacter
INFECTION AND IMMUNITY, Feb. 2000, p. 937 941 Vol. 68, No. 2 0019-9567/00/$04.00 0 Copyright 2000, American Society for Microbiology. All Rights Reserved. Relationship of Blood Group Determinants on Helicobacter
Correlation Between Endoscopic and Histological Findings in Different Gastroduodenal Lesion and its Association with Helicobacter Pylori
 ORIGINAL ARTICLE Correlation Between Endoscopic and Histological Findings in Different Gastroduodenal Lesion and its Association with Helicobacter Pylori *A. Sultana 1, SM Badruddoza 2, F Rahman 3 1 Dr.
ORIGINAL ARTICLE Correlation Between Endoscopic and Histological Findings in Different Gastroduodenal Lesion and its Association with Helicobacter Pylori *A. Sultana 1, SM Badruddoza 2, F Rahman 3 1 Dr.
Helicobacter pylori: Diagnosis, treatment and risks of untreated infection
 Helicobacter pylori: Diagnosis, treatment and risks of untreated infection Klaus Mönkemüller Department of Gastroenterology, Hepatology und Infectius Diseases Otto-von-Guericke University, Magdeburg bb
Helicobacter pylori: Diagnosis, treatment and risks of untreated infection Klaus Mönkemüller Department of Gastroenterology, Hepatology und Infectius Diseases Otto-von-Guericke University, Magdeburg bb
Original Article. Abstract
 Original Article Association of helicobacter pylori with carcinoma of stomach Muhammad Arif, Serajuddaula Syed Department of Pathology, Sindh Medical College, Karachi Abstract Objective: To note the association
Original Article Association of helicobacter pylori with carcinoma of stomach Muhammad Arif, Serajuddaula Syed Department of Pathology, Sindh Medical College, Karachi Abstract Objective: To note the association
The role of antisecretory drugs in the treatment of Helicobacter pylori infection
 Aliment Pharmacol Ther 1997; 11 (Suppl. 1): 21 25. The role of antisecretory drugs in the treatment of Helicobacter pylori infection W. L. PETERSON Department of Internal Medicine, University of Texas
Aliment Pharmacol Ther 1997; 11 (Suppl. 1): 21 25. The role of antisecretory drugs in the treatment of Helicobacter pylori infection W. L. PETERSON Department of Internal Medicine, University of Texas
National Digestive Diseases Information Clearinghouse
 Gastritis National Digestive Diseases Information Clearinghouse U.S. Department of Health and Human Services NATIONAL INSTITUTES OF HEALTH What is gastritis? Gastritis is a condition in which the stomach
Gastritis National Digestive Diseases Information Clearinghouse U.S. Department of Health and Human Services NATIONAL INSTITUTES OF HEALTH What is gastritis? Gastritis is a condition in which the stomach
Gastrointestinal pathology 2018 lecture 4. Dr Heyam Awad FRCPath
 Gastrointestinal pathology 2018 lecture 4 Dr Heyam Awad FRCPath Topics to be covered Peptic ulcer disease Hiatal hernia Gastric neoplasms Peptic ulcer disease (PUD)= chronic gastric ulcer Causes H pylori
Gastrointestinal pathology 2018 lecture 4 Dr Heyam Awad FRCPath Topics to be covered Peptic ulcer disease Hiatal hernia Gastric neoplasms Peptic ulcer disease (PUD)= chronic gastric ulcer Causes H pylori
Introduction. Original articles. Nicolás Rocha, 1 Sandra Huertas, 2 Rosario Albis, 3 Diego Aponte, 4 Luis Carlos Sabbagh. 5
 Original articles Correlation of endoscopic and histological findings in diagnosis of gastrointestinal metaplasia in patients referred to the Clinica Colombia for upper endoscopies Nicolás Rocha, 1 Sandra
Original articles Correlation of endoscopic and histological findings in diagnosis of gastrointestinal metaplasia in patients referred to the Clinica Colombia for upper endoscopies Nicolás Rocha, 1 Sandra
COMPARISON OF ONCE-A-DAY VERSUS TWICE-A-DAY CLARITHROMYCIN IN TRIPLE THERAPY FOR HELICOBACTER PYLORI ERADICATION
 Phil J Gastroenterol 2006; 2: 25-29 COMPARISON OF ONCE-A-DAY VERSUS TWICE-A-DAY CLARITHROMYCIN IN TRIPLE THERAPY FOR HELICOBACTER PYLORI ERADICATION Marianne P Collado, Ma Fatima P Calida, Peter P Sy,
Phil J Gastroenterol 2006; 2: 25-29 COMPARISON OF ONCE-A-DAY VERSUS TWICE-A-DAY CLARITHROMYCIN IN TRIPLE THERAPY FOR HELICOBACTER PYLORI ERADICATION Marianne P Collado, Ma Fatima P Calida, Peter P Sy,
Two Cases of Primary Gastric Lymphoma, Mucosa-Associated Lymphoid Tissue (MALT)-type
 Med. J. Kagoshima Univ., Vol. 47, Suppl. 2. 93-96, November, 1995 Case Report Two Cases of Primary Gastric Lymphoma, Mucosa-Associated Lymphoid Tissue (MALT)-type Mitsuharu NOMOTO1, Hiroshi SHIRAHAMA1,
Med. J. Kagoshima Univ., Vol. 47, Suppl. 2. 93-96, November, 1995 Case Report Two Cases of Primary Gastric Lymphoma, Mucosa-Associated Lymphoid Tissue (MALT)-type Mitsuharu NOMOTO1, Hiroshi SHIRAHAMA1,
Research Article Performance of Routine Helicobacter pylori Invasive Tests in Patients with Dyspepsia
 Gastroenterology Research and Practice Volume 2013, Article ID 184806, 5 pages http://dx.doi.org/10.1155/2013/184806 Research Article Performance of Routine Helicobacter pylori Invasive Tests in Patients
Gastroenterology Research and Practice Volume 2013, Article ID 184806, 5 pages http://dx.doi.org/10.1155/2013/184806 Research Article Performance of Routine Helicobacter pylori Invasive Tests in Patients
Epithelial cell proliferation and glandular atrophy in lymphocytic gastritis: Effect of H pylori treatment
 P.O.Box 2345, Beijing 100023,China World J Gastroenterol 2003;9(12):2706-2710 Fax: +86-10-85381893 World Journal of Gastroenterology E-mail: wjg@wjgnet.com www.wjgnet.com Copyright 2003 by The WJG Press
P.O.Box 2345, Beijing 100023,China World J Gastroenterol 2003;9(12):2706-2710 Fax: +86-10-85381893 World Journal of Gastroenterology E-mail: wjg@wjgnet.com www.wjgnet.com Copyright 2003 by The WJG Press
HISTOPATHOLOGICAL STUDY OF ENDOSCOPIC BIOPSIES OF STOMACH
 HISTOPATHOLOGICAL STUDY OF ENDOSCOPIC BIOPSIES OF STOMACH Pages with reference to book, From 177 To 179 Javed Iqbal Kazi, Syed Mahmood Alam ( Departments of Pathology, Jinnah Postgraduate Medical Centre,
HISTOPATHOLOGICAL STUDY OF ENDOSCOPIC BIOPSIES OF STOMACH Pages with reference to book, From 177 To 179 Javed Iqbal Kazi, Syed Mahmood Alam ( Departments of Pathology, Jinnah Postgraduate Medical Centre,
DOCTORAL THESIS SUMMARY
 UNIVERSITY OF MEDICINE AND PHARMACY CRAIOVA DOCTORAL THESIS HISTOPATHOLOGICAL AND IMMUNOHISTOCHEMICAL STUDY OF GASTRIC CARCINOMAS SUMMARY Scientific Coordinator: Univ. Prof. Dr. SIMIONESCU CRISTIANA EUGENIA
UNIVERSITY OF MEDICINE AND PHARMACY CRAIOVA DOCTORAL THESIS HISTOPATHOLOGICAL AND IMMUNOHISTOCHEMICAL STUDY OF GASTRIC CARCINOMAS SUMMARY Scientific Coordinator: Univ. Prof. Dr. SIMIONESCU CRISTIANA EUGENIA
Correlation between Gastric Mucosal Morphologic Patterns and Histopathological Severity of
 Hindawi Publishing Corporation BioMed Research International Volume 2015, Article ID 808505, 7 pages http://dx.doi.org/10.1155/2015/808505 Research Article Correlation between Gastric Mucosal Morphologic
Hindawi Publishing Corporation BioMed Research International Volume 2015, Article ID 808505, 7 pages http://dx.doi.org/10.1155/2015/808505 Research Article Correlation between Gastric Mucosal Morphologic
Sonic hedgehog expression in gastric cancer and gastric adenoma
 ONCOLOGY REPORTS 17: 1051-1055, 2007 Sonic hedgehog expression in gastric cancer and gastric adenoma SUN-YOUNG LEE 1, HYE SEUNG HAN 2, KYUNG YUNG LEE 3, TAE SOOK HWANG 2, JEONG HWAN KIM 1, IN-KYUNG SUNG
ONCOLOGY REPORTS 17: 1051-1055, 2007 Sonic hedgehog expression in gastric cancer and gastric adenoma SUN-YOUNG LEE 1, HYE SEUNG HAN 2, KYUNG YUNG LEE 3, TAE SOOK HWANG 2, JEONG HWAN KIM 1, IN-KYUNG SUNG
Prevpac Pylera Omeclamox-Pak
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.50.21 Subject: -Pak Page: 1 of 5 Last Review Date: September 20, 2018 -Pak Description (lansoprazole,
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.50.21 Subject: -Pak Page: 1 of 5 Last Review Date: September 20, 2018 -Pak Description (lansoprazole,
Gastric adenocarcinoma of the
 Comment From the Editors Spasmolytic Polypeptide- Expressing Metaplasia and Intestinal Metaplasia: Time for Reevaluation of Metaplasias and the Origins of Gastric Cancer Gastric adenocarcinoma of the intestinal
Comment From the Editors Spasmolytic Polypeptide- Expressing Metaplasia and Intestinal Metaplasia: Time for Reevaluation of Metaplasias and the Origins of Gastric Cancer Gastric adenocarcinoma of the intestinal
Anatomic Pathology / CHRONIC INACTIVE GASTRITIS AND COCCOID H PYLORI
 Anatomic Pathology / CHRONIC INACTIVE GASTRITIS AND COCCOID H PYLORI Chronic Inactive Gastritis and Coccoid Helicobacter pylori in Patients Treated for Gastroesophageal Reflux Disease or With H pylori
Anatomic Pathology / CHRONIC INACTIVE GASTRITIS AND COCCOID H PYLORI Chronic Inactive Gastritis and Coccoid Helicobacter pylori in Patients Treated for Gastroesophageal Reflux Disease or With H pylori
HISTOPATHOLOGICAL EVALUATION OF H. PYLORI ASSOCIATED GASTRIC LESIONS IN BENIN CITY, NIGERIA
 408 East African Medical Journal December 2012 East African Medical Journal Vol. 89 No. 12 December 2012 HISTOPATHOLOGICAL EVALUATION OF H. PYLORI ASSOCIATED GASTRIC LESIONS IN BENIN CITY, NIGERIA M. O.
408 East African Medical Journal December 2012 East African Medical Journal Vol. 89 No. 12 December 2012 HISTOPATHOLOGICAL EVALUATION OF H. PYLORI ASSOCIATED GASTRIC LESIONS IN BENIN CITY, NIGERIA M. O.
Common Inflammatory Gastrointestinal Disorders: Endoscopic and Pathologic Correlations
 Common Inflammatory Gastrointestinal Disorders: Endoscopic and Pathologic Correlations Nicole C. Panarelli, M.D. Attending Pathologist Montefiore Medical Center Associate Professor of Pathology - Albert
Common Inflammatory Gastrointestinal Disorders: Endoscopic and Pathologic Correlations Nicole C. Panarelli, M.D. Attending Pathologist Montefiore Medical Center Associate Professor of Pathology - Albert
H. pylori Antigen ELISA Kit
 H. pylori Antigen ELISA Kit Catalog Number KA3142 96 assays Version: 04 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of
H. pylori Antigen ELISA Kit Catalog Number KA3142 96 assays Version: 04 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of
Review article: exploring the link between Helicobacter pylori and gastric cancer
 Aliment Pharmacol Ther 1999; 13 (Suppl. 1), 3±11 Review article: exploring the link between Helicobacter pylori and gastric cancer E. J. KUIPERS Free University Hospital, Amsterdam, The Netherlands SUMMARY
Aliment Pharmacol Ther 1999; 13 (Suppl. 1), 3±11 Review article: exploring the link between Helicobacter pylori and gastric cancer E. J. KUIPERS Free University Hospital, Amsterdam, The Netherlands SUMMARY
Are Gastrointestinal Mucosal Mast Cells Increased in Patients With Systemic Mastocytosis?
 Anatomic Pathology / GASTRODUODENAL MAST CELLS IN MASTOCYTOSIS Are Gastrointestinal Mucosal Mast Cells Increased in Patients With Systemic Mastocytosis? Sabine I. Siegert, MD, 1 Joachim Diebold, MD, PhD,
Anatomic Pathology / GASTRODUODENAL MAST CELLS IN MASTOCYTOSIS Are Gastrointestinal Mucosal Mast Cells Increased in Patients With Systemic Mastocytosis? Sabine I. Siegert, MD, 1 Joachim Diebold, MD, PhD,
PATHOLOGY OF NON NEOPLASTIC LESIONS OF THE UPPER GASTROINTESTINAL TRACT.
 PATHOLOGY OF NON NEOPLASTIC LESIONS OF THE UPPER GASTROINTESTINAL TRACT. OESOPHAGEAL LESIONS OESOPHAGITIS AND OTHER NON NEOPLASTIC DISORDERS Corrosive Gastroesophageal reflux (GERD), Pills, Acid intake,
PATHOLOGY OF NON NEOPLASTIC LESIONS OF THE UPPER GASTROINTESTINAL TRACT. OESOPHAGEAL LESIONS OESOPHAGITIS AND OTHER NON NEOPLASTIC DISORDERS Corrosive Gastroesophageal reflux (GERD), Pills, Acid intake,
ulcer healing role 118 Bicarbonate, prostaglandins in duodenal cytoprotection 235, 236
 Subject Index Actin cellular forms 48, 49 epidermal growth factor, cytoskeletal change induction in mucosal repair 22, 23 wound repair 64, 65 polyamine effects on cytoskeleton 49 51 S-Adenosylmethionine
Subject Index Actin cellular forms 48, 49 epidermal growth factor, cytoskeletal change induction in mucosal repair 22, 23 wound repair 64, 65 polyamine effects on cytoskeleton 49 51 S-Adenosylmethionine
Clinical Study Multifaceted Assessment of Chronic Gastritis: A Study of Correlations between Serological, Endoscopic, and Histological Diagnostics
 Gastroenterology Research and Practice Volume 2, Article ID 6346, 7 pages doi:.55/2/6346 Clinical Study Multifaceted Assessment of Chronic Gastritis: A Study of Correlations between Serological, Endoscopic,
Gastroenterology Research and Practice Volume 2, Article ID 6346, 7 pages doi:.55/2/6346 Clinical Study Multifaceted Assessment of Chronic Gastritis: A Study of Correlations between Serological, Endoscopic,
Case Report Features of the Atrophic Corpus Mucosa in Three Cases of Autoimmune Gastritis Revealed by Magnifying Endoscopy
 Volume 2012, Article ID 368160, 4 pages doi:10.1155/2012/368160 Case Report Features of the Atrophic Corpus Mucosa in Three Cases of Autoimmune Gastritis Revealed by Magnifying Endoscopy Kazuyoshi Yagi,
Volume 2012, Article ID 368160, 4 pages doi:10.1155/2012/368160 Case Report Features of the Atrophic Corpus Mucosa in Three Cases of Autoimmune Gastritis Revealed by Magnifying Endoscopy Kazuyoshi Yagi,
What is the status of Sequential Therapy Versus Standard Triple- Drug Therapy in peptic ulcer disease in eradicating H pylori?
 What is the status of Sequential Therapy Versus Standard Triple- Drug Therapy in peptic ulcer disease in eradicating H pylori? Sequential Therapy Versus Standard Triple- Drug Therapy for Helicobacter pylori
What is the status of Sequential Therapy Versus Standard Triple- Drug Therapy in peptic ulcer disease in eradicating H pylori? Sequential Therapy Versus Standard Triple- Drug Therapy for Helicobacter pylori
594 Lewin, Weinstein, and Riddell s Gastrointestinal Pathology and Its Clinical Implications
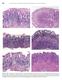 594 Lewin, Weinstein, and Riddell s Gastrointestinal Pathology and Its Clinical Implications Figure 13-20. Stages in the natural history of H. pylori. Biopsies from the antrum are on the left and the oxyntic
594 Lewin, Weinstein, and Riddell s Gastrointestinal Pathology and Its Clinical Implications Figure 13-20. Stages in the natural history of H. pylori. Biopsies from the antrum are on the left and the oxyntic
Helicobacter Pylori Testing HELICOBACTER PYLORI TESTING HS-131. Policy Number: HS-131. Original Effective Date: 9/17/2009
 Easy Choice Health Plan, Inc. Harmony Health Plan of Illinois, Inc. Missouri Care, Inc. Ohana Health Plan, a plan offered by WellCare Health Insurance of Arizona, Inc. WellCare Health Insurance of Illinois,
Easy Choice Health Plan, Inc. Harmony Health Plan of Illinois, Inc. Missouri Care, Inc. Ohana Health Plan, a plan offered by WellCare Health Insurance of Arizona, Inc. WellCare Health Insurance of Illinois,
Treatment of Helicobacter pylori Infection
 Treatment of Helicobacter pylori Infection Epidemiology of H. pylori infection (North America) Which are the high risk groups? Epidemiology of H. pylori infection (North America) Which are the high risk
Treatment of Helicobacter pylori Infection Epidemiology of H. pylori infection (North America) Which are the high risk groups? Epidemiology of H. pylori infection (North America) Which are the high risk
Gastritis Associated with Epstein-Barr Virus Infection
 CASE REPORT Gastritis Associated with Epstein-Barr Virus Infection Akari Hisamatsu 1, Takayuki Nagai 1, Hitoshi Okawara 1, Hiroshi Nakashima 1, Takako Tasaki 1, Yoshifumi Nakagawa 1, Masahiko Hashinaga
CASE REPORT Gastritis Associated with Epstein-Barr Virus Infection Akari Hisamatsu 1, Takayuki Nagai 1, Hitoshi Okawara 1, Hiroshi Nakashima 1, Takako Tasaki 1, Yoshifumi Nakagawa 1, Masahiko Hashinaga
(Index words: Clarithromycin, compliance, efficacy, tinidazole and omeprazole therapy)
 Comparison of one week and two weeks of triple therapy for the eradication of Helicobacter pylori in a Sri Lankan population: a randomised, controlled study HA de Silva 1, J Hewavisenthi 2, A Pathmeswaran
Comparison of one week and two weeks of triple therapy for the eradication of Helicobacter pylori in a Sri Lankan population: a randomised, controlled study HA de Silva 1, J Hewavisenthi 2, A Pathmeswaran
Corporate Medical Policy
 Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: helicobacter_pylori_testing 01/01/2019 N/A 01/01/2020 01/01/2019 Policy Effective April 1, 2019 Description
Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: helicobacter_pylori_testing 01/01/2019 N/A 01/01/2020 01/01/2019 Policy Effective April 1, 2019 Description
ESMO Preceptorship Gastrointestinal Tumours Valencia October 2017
 www.esmo.org ESMO Preceptorship Valencia 06-07 October 2017 Gastrointestinal Tumours I have no conflicts of interest to declare Fátima Carneiro Gastrointestinal tumours Multidisciplinary management, standards
www.esmo.org ESMO Preceptorship Valencia 06-07 October 2017 Gastrointestinal Tumours I have no conflicts of interest to declare Fátima Carneiro Gastrointestinal tumours Multidisciplinary management, standards
SUMMARY INTRODUCTION. Aliment Pharmacol Ther 2000; 14: 625±634. Accepted for publication 20 January 2000
 Aliment Pharmacol Ther 2000; 14: 625±634. Cure of Helicobacter pylori infection in atrophic body gastritis patients does not improve mucosal atrophy but reduces hypergastrinemia and its related effects
Aliment Pharmacol Ther 2000; 14: 625±634. Cure of Helicobacter pylori infection in atrophic body gastritis patients does not improve mucosal atrophy but reduces hypergastrinemia and its related effects
Comparative study of invasive methods for diagnosis of Helicobacter pylori in humans
 ISSN: 2319-7706 Volume 2 Number 7 (2013) pp. 63-68 http://www.ijcmas.com Original Research Article Comparative study of invasive methods for diagnosis of Helicobacter pylori in humans V.Subbukesavaraja
ISSN: 2319-7706 Volume 2 Number 7 (2013) pp. 63-68 http://www.ijcmas.com Original Research Article Comparative study of invasive methods for diagnosis of Helicobacter pylori in humans V.Subbukesavaraja
New developments in pathogenesis, gastric cancer. Matthias Ebert. II. Medizinische Klinik Klinikum rechts der Isar TU München
 New developments in pathogenesis, diagnosis, therapy and prevention of gastric cancer Matthias Ebert II. Medizinische Klinik Klinikum rechts der Isar TU München Gastric Cancer Pathogenesis Diagnosis Treatment
New developments in pathogenesis, diagnosis, therapy and prevention of gastric cancer Matthias Ebert II. Medizinische Klinik Klinikum rechts der Isar TU München Gastric Cancer Pathogenesis Diagnosis Treatment
Three-day lansoprazole quadruple therapy for Helicobacter pylori-positive duodenal ulcers: a randomized controlled study
 Aliment Pharmacol Ther 2001; 15: 843±849. Three-day lansoprazole quadruple therapy for Helicobacter pylori-positive duodenal ulcers: a randomized controlled study B. C. Y. WONG*, W. H. WANG*, W.M.WONG*,G.K.K.LAU*,F.M.Y.FUNG*,N.N.S.KUNGà,
Aliment Pharmacol Ther 2001; 15: 843±849. Three-day lansoprazole quadruple therapy for Helicobacter pylori-positive duodenal ulcers: a randomized controlled study B. C. Y. WONG*, W. H. WANG*, W.M.WONG*,G.K.K.LAU*,F.M.Y.FUNG*,N.N.S.KUNGà,
Survey on repeat prescribing for acid suppression drugs in primary care in Cornwall and the Isles of Scilly
 Aliment Pharmacol Ther 1999; 13: 813±817. Survey on repeat prescribing for acid suppression drugs in primary care in Cornwall and the Isles of Scilly R. BOUTET, M. WILCOCK & I. MACKENZIE 1 Department of
Aliment Pharmacol Ther 1999; 13: 813±817. Survey on repeat prescribing for acid suppression drugs in primary care in Cornwall and the Isles of Scilly R. BOUTET, M. WILCOCK & I. MACKENZIE 1 Department of
Proton pump inhibitor (proton pump inhibitor)-based triple therapies using clarithromycin in combination SUMMARY INTRODUCTION
 Aliment Pharmacol Ther 2003; 18: 799 804. doi: 10.1046/j.1365-2036.2003.01764.x Esomeprazole-based one-week triple therapy with clarithromycin and metronidazole is effective in eradicating Helicobacter
Aliment Pharmacol Ther 2003; 18: 799 804. doi: 10.1046/j.1365-2036.2003.01764.x Esomeprazole-based one-week triple therapy with clarithromycin and metronidazole is effective in eradicating Helicobacter
The long-term management of patients with bleeding duodenal ulcers
 Aliment Pharmacol Ther (1997); 11: 505±510. The long-term management of patients with bleeding duodenal ulcers M. E. MCALINDON, J. S. W. TAYLOR & S. D. RYDER Department of Medicine, University Hospital,
Aliment Pharmacol Ther (1997); 11: 505±510. The long-term management of patients with bleeding duodenal ulcers M. E. MCALINDON, J. S. W. TAYLOR & S. D. RYDER Department of Medicine, University Hospital,
Helicobacter pylori infection: several studies on epidemiology, eradication and gastric epithelial cell turnover Liu, W.
 UvA-DARE (Digital Academic Repository) Helicobacter pylori infection: several studies on epidemiology, eradication and gastric epithelial cell turnover Liu, W. Link to publication Citation for published
UvA-DARE (Digital Academic Repository) Helicobacter pylori infection: several studies on epidemiology, eradication and gastric epithelial cell turnover Liu, W. Link to publication Citation for published
Helicobacter pylori Eradication Therapy Success Regarding Different Treatment Period Based on Clarithromycin or Metronidazole Triple-Therapy Regimens
 Helicobacter ISSN 1523-5378 Filipec Blackwell Oxford, HEL 1083-4389 1523-5378 Journal XXX Original H. 2008 pylori Kanizaj compilation The UK Eradication Publishing Article Authors et al. Ltd 2008 Therapy
Helicobacter ISSN 1523-5378 Filipec Blackwell Oxford, HEL 1083-4389 1523-5378 Journal XXX Original H. 2008 pylori Kanizaj compilation The UK Eradication Publishing Article Authors et al. Ltd 2008 Therapy
ÐÑÏÓÊÅÊËÇÌÅÍÅÓ ÎÅÍÏÃËÙÓÓÅÓ ÁÍÁÊÏÉÍÙÓÅÉÓ ÅËËÇÍÙÍ ÅÑÅÕÍÇÔÙÍ ÐÑÏÓÊÅÊËÇÌÅÍÅÓ ÎÅÍÏÃËÙÓÓÅÓ ÁÍÁÊÏÉÍÙÓÅÉÓ ÅËËÇÍÙÍ ÅÑÅÕÍÇÔÙÍ
 ÐÑÏÓÊÅÊËÇÌÅÍÅÓ ÎÅÍÏÃËÙÓÓÅÓ ÁÍÁÊÏÉÍÙÓÅÉÓ ÅËËÇÍÙÍ ÅÑÅÕÍÇÔÙÍ 39 40 HELICOBACTER PYLORI COLONIZATION IN PALATINE SALIVARY GLANDS E. Labropoulou, C. Labropoulou 2, V. Mamali 2, C. Tzamalis, A. Christopoulou
ÐÑÏÓÊÅÊËÇÌÅÍÅÓ ÎÅÍÏÃËÙÓÓÅÓ ÁÍÁÊÏÉÍÙÓÅÉÓ ÅËËÇÍÙÍ ÅÑÅÕÍÇÔÙÍ 39 40 HELICOBACTER PYLORI COLONIZATION IN PALATINE SALIVARY GLANDS E. Labropoulou, C. Labropoulou 2, V. Mamali 2, C. Tzamalis, A. Christopoulou
Gastritis (and gastropathy) Dr Ian Brown Envoi Pathology Brisbane, Australia
 Gastritis (and gastropathy) Dr Ian Brown Envoi Pathology Brisbane, Australia ianbrown@envoi.com.au Topics for discussion Classification of gastritis Minimal diagnostic criteria for gastritis H.pylori negative
Gastritis (and gastropathy) Dr Ian Brown Envoi Pathology Brisbane, Australia ianbrown@envoi.com.au Topics for discussion Classification of gastritis Minimal diagnostic criteria for gastritis H.pylori negative
Long-Term Effects of Helicobacter pylori Eradication on Metachronous Gastric Cancer Development
 Gut and Liver, Vol. 12, No. 2, March 218, pp. 133-141 ORiginal Article Long-Term ffects of Helicobacter pylori radication on Metachronous Gastric Cancer Development Seung Jun Han 1, Sang Gyun Kim 1, Joo
Gut and Liver, Vol. 12, No. 2, March 218, pp. 133-141 ORiginal Article Long-Term ffects of Helicobacter pylori radication on Metachronous Gastric Cancer Development Seung Jun Han 1, Sang Gyun Kim 1, Joo
Histopathological study of Helicobacter Pylori bearing Gastric Biopsies for Gastric Lesions
 ORIGINAL ARTICLE Histopathological study of Helicobacter Pylori bearing Gastric Biopsies for Gastric Lesions MULAZIM HUSSAIN BUKHARI, SAMINA QAMAR*, SHAHBAZ AHMAD QURESHI*, MALIK MAQSOOD ANWAR** ABSTRACT
ORIGINAL ARTICLE Histopathological study of Helicobacter Pylori bearing Gastric Biopsies for Gastric Lesions MULAZIM HUSSAIN BUKHARI, SAMINA QAMAR*, SHAHBAZ AHMAD QURESHI*, MALIK MAQSOOD ANWAR** ABSTRACT
The effect of CagA status on response to Helicobacter pylori eradication therapy in Western Turkey
 Brazilian Journal of Medical and Biological Research (2001) 34: 1435-1439 CagA status affects H. pylori eradication rate ISSN 0100-879X 1435 The effect of CagA status on response to Helicobacter pylori
Brazilian Journal of Medical and Biological Research (2001) 34: 1435-1439 CagA status affects H. pylori eradication rate ISSN 0100-879X 1435 The effect of CagA status on response to Helicobacter pylori
Bcl-2 Expression in CagA Strain H. Pylori Gastritis (Immunohistochemical and Insitu Hybridization Study)
 CAGA THE IRAQI STRAIN POSTGRADUATE H. PYLORI MEDICAL GASTRITIS JOURNAL Bcl- Expression in CagA Strain H. Pylori Gastritis (Immunohistochemical and Insitu Hybridization Study) Hussam Hasson Ali *, Hassan
CAGA THE IRAQI STRAIN POSTGRADUATE H. PYLORI MEDICAL GASTRITIS JOURNAL Bcl- Expression in CagA Strain H. Pylori Gastritis (Immunohistochemical and Insitu Hybridization Study) Hussam Hasson Ali *, Hassan
Histopathological profile of gastritis in adult patients seen at a referral hospital in Kenya
 Online Submissions: wjg.wjgnet.com World J Gastroenterol 27 August 14; 13(3): 4117-4121 World Journal of Gastroenterology ISSN 17-9327 wjg@wjgnet.com 27 WJG. All rights reserved. RAPID COMMUNICATION Histopathological
Online Submissions: wjg.wjgnet.com World J Gastroenterol 27 August 14; 13(3): 4117-4121 World Journal of Gastroenterology ISSN 17-9327 wjg@wjgnet.com 27 WJG. All rights reserved. RAPID COMMUNICATION Histopathological
Current evidence indicates that the anatomical distribution and severity of H. pylori gastritis are strongly SUMMARY INTRODUCTION
 Aliment Pharmacol Ther 2001; 15: 1163±1175. Double gastric infection with Helicobacter pylori and non- Helicobacter pylori bacteria during acid-suppressive therapy: increase of pro-in ammatory cytokines
Aliment Pharmacol Ther 2001; 15: 1163±1175. Double gastric infection with Helicobacter pylori and non- Helicobacter pylori bacteria during acid-suppressive therapy: increase of pro-in ammatory cytokines
Maastricht Ⅴ /Florence
 2016 21 10 577 Maastricht Ⅴ /Florence 200001 2015 10 8 9 Maastricht V 1 / 2 3 4 / 5 Maastricht Ⅴ Interpretation of Management of Helicobacter pylori Infection the Maastricht Ⅴ / Florence Consensus Report
2016 21 10 577 Maastricht Ⅴ /Florence 200001 2015 10 8 9 Maastricht V 1 / 2 3 4 / 5 Maastricht Ⅴ Interpretation of Management of Helicobacter pylori Infection the Maastricht Ⅴ / Florence Consensus Report
What is your diagnosis? a. Lymphocytic colitis. b. Collagenous colitis. c. Common variable immunodeficiency (CVID) associated colitis
 Case History A 24 year old male presented with fatigue, fever, watery diarrhea, and a cough with sputum production for the past three weeks. His past medical history was significant for recurrent bouts
Case History A 24 year old male presented with fatigue, fever, watery diarrhea, and a cough with sputum production for the past three weeks. His past medical history was significant for recurrent bouts
Coordinate Expression of Cytokeratins 7 and 20 in Prostate Adenocarcinoma and Bladder Urothelial Carcinoma
 Anatomic Pathology / CYTOKERATINS 7 AND 20 IN PROSTATE AND BLADDER CARCINOMAS Coordinate Expression of Cytokeratins 7 and 20 in Prostate Adenocarcinoma and Bladder Urothelial Carcinoma Nader H. Bassily,
Anatomic Pathology / CYTOKERATINS 7 AND 20 IN PROSTATE AND BLADDER CARCINOMAS Coordinate Expression of Cytokeratins 7 and 20 in Prostate Adenocarcinoma and Bladder Urothelial Carcinoma Nader H. Bassily,
Increased frequency of activated T-cells in the Helicobacter pylori-infected antrum and duodenum
 FEMS Immunology and Medical Microbiology 36 (2003) 159^168 www.fems-microbiology.org Increased frequency of activated T-cells in the Helicobacter pylori-infected antrum and duodenum E. Stro«mberg a, A.
FEMS Immunology and Medical Microbiology 36 (2003) 159^168 www.fems-microbiology.org Increased frequency of activated T-cells in the Helicobacter pylori-infected antrum and duodenum E. Stro«mberg a, A.
H.pylori IgA Cat #
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
Immunoglobulin G Antibody against Helicobacter pylori: Clinical Implications of Levels Found in Serum
 CLINICAL AND DIAGNOSTIC LABORATORY IMMUNOLOGY, Sept. 2002, p. 1044 1048 Vol. 9, No. 5 1071-412X/02/$04.00 0 DOI: 10.1128/CDLI.9.5.1044 1048.2002 Copyright 2002, American Society for Microbiology. All Rights
CLINICAL AND DIAGNOSTIC LABORATORY IMMUNOLOGY, Sept. 2002, p. 1044 1048 Vol. 9, No. 5 1071-412X/02/$04.00 0 DOI: 10.1128/CDLI.9.5.1044 1048.2002 Copyright 2002, American Society for Microbiology. All Rights
The prevalence of lymphoid follicles in Helicobacter pylon associated gastritis in patients
 I Clin Pathol 1995;48:325-329 Department of Histopathology, Al Qassimi Hospital, Sharjah, United Arab mirates A M Zaitoun Correspondence to: Dr A M Zaitoun, Mayday Healthcare NHS Trust, Mayday Road Thornton
I Clin Pathol 1995;48:325-329 Department of Histopathology, Al Qassimi Hospital, Sharjah, United Arab mirates A M Zaitoun Correspondence to: Dr A M Zaitoun, Mayday Healthcare NHS Trust, Mayday Road Thornton
Jeffrey L. Winslow, MD, 1 Thomas D. Trainer, MD, 1 and Richard B. Colletti, MD 2. Abstract
 Anatomic Pathology / COLLAGENOUS GASTRITIS Collagenous Gastritis A Long-Term Follow-up With the Development of Endocrine Cell Hyperplasia, Intestinal Metaplasia, and Epithelial Changes Indeterminate for
Anatomic Pathology / COLLAGENOUS GASTRITIS Collagenous Gastritis A Long-Term Follow-up With the Development of Endocrine Cell Hyperplasia, Intestinal Metaplasia, and Epithelial Changes Indeterminate for
THE PREVALENCE OF HELICBACTER PYLORI AMONG PATIENTS COMPLAINING FROM ABDOMINAL PAIN
 THE PREVALENCE OF HELICBACTER PYLORI AMONG PATIENTS COMPLAINING FROM ABDOMINAL PAIN Ahed J. Al-Khatib Jordan University of Science and Technology, Jordan Ahmed Saber Abu-zaiton Al-albayt University Abstract
THE PREVALENCE OF HELICBACTER PYLORI AMONG PATIENTS COMPLAINING FROM ABDOMINAL PAIN Ahed J. Al-Khatib Jordan University of Science and Technology, Jordan Ahmed Saber Abu-zaiton Al-albayt University Abstract
Helicobacter heilmannii-associated Gastritis: Clinicopathologic Findings
 J Korean Med Sci 2007; 22: 63-9 ISSN 1011-8934 Copyright The Korean Academy of Medical Sciences Helicobacter heilmannii-associated Gastritis: Clinicopathologic Findings and Comparison with Helicobacter
J Korean Med Sci 2007; 22: 63-9 ISSN 1011-8934 Copyright The Korean Academy of Medical Sciences Helicobacter heilmannii-associated Gastritis: Clinicopathologic Findings and Comparison with Helicobacter
Helicobacter pylori and gastric cancer: current status of the Austrain Czech German gastric cancer prevention trial (PRISMA-Study)
 PO Box 2345, Beijing 100023, China Fax: +86-10-85381893 E-mail: wjg@wjgnet.com www.wjgnet.com World J Gastroentero, 2001;7(2):243-247 World Journal of Gastroenterology Copyright 2001 by the WJG Press ISSN
PO Box 2345, Beijing 100023, China Fax: +86-10-85381893 E-mail: wjg@wjgnet.com www.wjgnet.com World J Gastroentero, 2001;7(2):243-247 World Journal of Gastroenterology Copyright 2001 by the WJG Press ISSN
Proton Pump Inhibitors Drug Class Prior Authorization Protocol
 Proton Pump Inhibitors Drug Class Prior Authorization Protocol Line of Business: Medi-Cal P&T Approval Date: November 15, 2017 Effective Date: January 1, 2018 This policy has been developed through review
Proton Pump Inhibitors Drug Class Prior Authorization Protocol Line of Business: Medi-Cal P&T Approval Date: November 15, 2017 Effective Date: January 1, 2018 This policy has been developed through review
Gastric Polyps. Bible class
 Gastric Polyps Bible class 29.08.2018 Starting my training in gastroenterology, some decades ago, my first chief always told me that colonoscopy may seem technically more challenging but gastroscopy has
Gastric Polyps Bible class 29.08.2018 Starting my training in gastroenterology, some decades ago, my first chief always told me that colonoscopy may seem technically more challenging but gastroscopy has
Effect of Helicobacter pylori Eradication on Metachronous Recurrence After Endoscopic Resection of Gastric Neoplasm
 60 ORIGINAL CONTRIBUTIONS nature publishing group see related editorial on page x Effect of Helicobacter pylori Eradication on Metachronous Recurrence After Endoscopic Resection of Gastric Neoplasm Suh
60 ORIGINAL CONTRIBUTIONS nature publishing group see related editorial on page x Effect of Helicobacter pylori Eradication on Metachronous Recurrence After Endoscopic Resection of Gastric Neoplasm Suh
