The Tumor Suppressor Par-4 Activates an Extrinsic Pathway for Apoptosis
|
|
|
- Arabella Wilson
- 6 years ago
- Views:
Transcription
1 The Tumor Suppressor Par-4 Activates an Extrinsic Pathway for Apoptosis Ravshan Burikhanov, 1 Yanming Zhao, 1 Anindya Goswami, 1 Shirley Qiu, 1 Steven R. Schwarze, 2 and Vivek M. Rangnekar 1,3,4,5, * 1 Department of Radiation Medicine 2 Department of Biochemistry and Molecular Biology 3 Department of Microbiology, Immunology, and Molecular Genetics 4 Graduate Center for Toxicology 5 Markey Cancer Center University of Kentucky, Lexington, KY 40536, USA *Correspondence: vmrang01@ .uky.edu DOI /j.cell SUMMARY Prostate apoptosis response-4 (Par-4) is a proapoptotic protein with intracellular functions in the cytoplasm and nucleus. Unexpectedly, we noted Par-4 protein is spontaneously secreted by normal and cancer cells in culture, and by Par-4 transgenic mice that are resistant to spontaneous tumors. Short exposure to endoplasmic reticulum (ER) stressinducing agents further increased cellular secretion of Par-4 by a brefeldin A-sensitive pathway. Secretion occurred independently of caspase activation and apoptosis. Interestingly, extracellular Par-4 induced apoptosis by binding to the stress response protein, glucose-regulated protein-78 (GRP78), expressed at the surface of cancer cells. The interaction of extracellular Par-4 and cell surface GRP78 led to apoptosis via ER stress and activation of the FADD/caspase-8/ caspase-3 pathway. Moreover, apoptosis inducible by TRAIL, which also exerts cancer cell-specific effects, is dependent on extracellular Par-4 signaling via cell surface GRP78. Thus, Par-4 activates an extrinsic pathway involving cell surface GRP78 receptor for induction of apoptosis. INTRODUCTION Prostate apoptosis response-4 (Par-4) is a leucine zipper domain protein identified in cells undergoing apoptosis in response to exogenous insults (Sells et al., 1994). Par-4 is expressed ubiquitously among the various tissue types, and resides in both the cytoplasm and the nucleus (Sells et al., 1997; El-Guendy et al., 2003). Although endogenous Par-4 is largely inactivated, and does not produce extensive apoptosis by itself, it is essential for the apoptotic function of diverse cytotoxic agents (Gurumurthy et al., 2005). Interestingly, Par-4 overexpression is sufficient to induce apoptosis in most cancer cells, but not in normal or immortalized cells, and this apoptotic action of Par-4 does not require its leucine zipper domain (El-Guendy et al., 2003). Deletion analyses have identified the core domain (aa ) of Par-4, designated SAC (selective for apoptosis in cancer cells), as the effector domain of Par-4. Like Par-4, the SAC domain is capable of nuclear entry, caspase activation, inhibition of NF-kB activity, and induction of apoptosis. The SAC domain is 100% conserved in rat, mouse, and human Par-4, and does not resemble other proapoptotic proteins (El-Guendy et al., 2003). Transgenic mice ubiquitously expressing the SAC domain of Par-4 are resistant to the growth of spontaneous and inducible tumors (Zhao et al., 2007). Consistent with its tumor suppressor function in mice (Garcia-Cao et al., 2003), Par-4 is mutated or silenced in endometrial cancer (Moreno-Bueno et al., 2007), downregulated in renal cell carcinoma (Cook et al., 1999), and in a nonfunctional state in prostate cancer due to inactivation by the cell survival kinase, Akt1 (Goswami et al., 2005). Our recent studies suggest Par-4 serves as an intracellular repressor of topoisomerase 1 (TOP1) catalytic activity, and regulates DNA topology to suppress cellular transformation by a SAC domainindependent mechanism (Goswami et al., 2008). Par-4- and SAC-Mediated Apoptosis All Par-4 studies thus far have analyzed the role of intracellular (i.e., cytoplasmic or nuclear) Par-4. Distinct partner proteins of intracellular Par-4 have been confirmed, including zpkc (Diaz- Meco et al., 1996), WT1 (Johnstone et al., 1996, ZIP kinase (Page et al., 1999), Akt1 (Goswami et al., 2005), and TOP1 (Goswami et al., 2008); each of these partner interactions requires the leucine zipper domain. Par-4 interactions with zpkc or TOP1 in the cytoplasm or nucleus, respectively, impede NF-kB activity (Diaz-Meco et al., 1996; Goswami et al., 2008), and Par-4 interactions with WT1 inhibit the Bcl-2 promoter (Cheema et al., 2003); these interactions reveal distinct, cell compartmentspecific roles for the Par-4 partners in growth regulation. Rather unexpectedly, our recent studies suggest Par-4 and SAC domain are secreted by both normal/immortalized and Cell 138, , July 24, 2009 ª2009 Elsevier Inc. 377
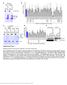
2 Figure 1. Par-4 and the SAC Protein Are Secreted and Induce Apoptosis (A) Bystander effect of Par-4 and SAC domain. PC-3 cells were transiently transfected with the indicated expression constructs, and subjected to ICC for active caspase-3 to detect apoptosis, and counterstained with DAPI (right panel). Quantification of apoptosis in transfected (T) and nontransfected (NT) cells is presented (left panel). (B) Conditioned medium (CM) from transfected cells contains secreted Par-4-GFP and SAC-GFP protein. The CM from cells transfected with the indicated expression constructs or pcb6+ vector was subjected to western blot (WB) analysis with the Par-4, ERK1/2, or actin antibody. Appropriate whole-cell extracts (WCE) were used as controls. (C) The secreted fusion proteins induce apoptosis in target PC-3 cells. Cells were treated with the CM from the indicated PC-3 transfectants (GFP, Par-4-GFP, or SAC-GFP) or with the CM that was preincubated for 30 min with neutralizing antibody (Ab) for GFP, Par-4, or PTEN. After 24 hr, the cells were scored for apoptosis by caspase-3 activation using ICC analysis. 378 Cell 138, , July 24, 2009 ª2009 Elsevier Inc.
3 cancer cells, and that these secreted proteins selectively inhibit the growth of cancer cells. We have identified a regulatory loop that involves the interaction of extracellular Par-4, via its SAC domain, with the cell surface receptor GRP78, and that is dependent on intracellular Par-4 in order to selectively induce apoptosis in cancer cells. RESULTS Par-4 Is Secreted and Induces Apoptosis of Cancer Cells via Its SAC Domain Although ectopic Par-4 and its SAC domain are well-known to exert their apoptotic effects in the nucleus of the cell in which they are produced, we have noted bystander activity in tumor regression studies wherein a single intratumoral injection of an adenoviral-par-4 construct (with a 50% 60% transduction efficiency) resulted in apoptosis in over 80% cells of the tumor (Chakraborty et al., 2001). To further characterize this bystander effect of Par-4, we transfected PC-3 cells in culture with GFP, Par-4-GFP, or SAC-GFP expression constructs, and subjected the cell populations to immunocytochemical (ICC) analysis for active caspase-3 to detect apoptosis. Intriguingly, apoptosis occurred not only in the green fluorescent Par-4-GFP or SAC- GFP transfectants, but also in neighboring cells that did not express the green-fluorescent Par-4-GFP protein (Figure 1A, right panel). Quantification of the cultures for apoptotic cells indicated a significantly large number of nontransfected cells underwent apoptosis with Par-4-GFP or SAC-GFP transfection, but not with the GFP construct (Figure 1A, left panel). Further studies revealed the conditioned medium (CM) derived from Par-4-GFP-or SAC-GFP-transfected PC-3 and BPH-1 cells contained Par-4-GFP and SAC-GFP proteins, respectively (Figure 1B). The CM did not contain detectable levels of actin or ERK1/2 (Figure 1B), implying the GFP fusion proteins are not released as a result of apoptotic cell death. Significantly, CM from the GFP (control) and SAC-GFP transfectants also contained endogenous Par-4 (Figure 1B), indicating endogenous Par-4 is secreted by PC-3 cells. Moreover, immortalized, nontransformed BPH-1 cells, which do not undergo apoptosis with ectopic Par-4, were transfected with either a pcb6+/par-4 construct (lacking the GFP-tag) or a GFP control construct. CM from these cells showed secretion of both endogenous Par-4 and ectopic Par-4 (Figure 1B), demonstrating secretion of Par-4 is neither dependent on the GFP-tag, nor on apoptosis of the transfectants. Secretion of endogenous Par-4 was not limited to prostatic cells; immortalized human embryonic lung fibroblasts HEL, human embryonic kidney epithelial cells HEK293, and mouse fibroblasts NIH 3T3 showed secretion of Par-4 (see Figure S1A available with this article online). Overall, mammalian cells secrete endogenous Par-4, as well as ectopic Par-4 and SAC protein. To determine whether secreted Par-4 was functionally active, PC-3 cells and BPH-1 cells were treated with the CM prepared from PC-3 transfectants, and their response was examined by ICC for caspase-3 activation. As seen in Figure 1C, the CM from the Par-4-GFP and SAC-GFP transfectants (but not from the GFP control transfectants) induced apoptosis in PC-3 cells, but not in the BPH-1 cells. Importantly, when the CM was preincubated with neutralizing antibodies for GFP, Par-4, or PTEN (control), the apoptotic activity in the CM was neutralized by antibodies against Par-4/SAC and the GFP-tag, but not by the control PTEN antibody. Because CM may contain other mammalian cell secreted proteins, we used recombinant Par-4 and SAC (TRX-Par-4 and TRX-SAC, respectively) as an alternate approach to determine whether extracellular Par-4 and SAC induce apoptosis. Exposure of PC-3 cells to TRX-Par-4 and TRX-SAC, but not TRX control protein, produced apoptosis in a dose- and time-dependent manner (Figure S1). When cancer cells PC-3, H460, and HeLa were treated with these proteins and scored for apoptosis, we noted that TRX-Par-4 and TRX-SAC, but not TRX, induced apoptosis in each cell line (Figure 1D). Conversely, TRX-Par-4 or TRX-SAC did not induce apoptosis in the nontransformed cells BPH-1 (Figure 1D). Indeed, TRX-Par-4 and TRX-SAC induced apoptosis in diverse other cancer cell lines, but not nontransformed cell lines (data not shown). We conclude recombinant Par-4 is competent to cause apoptosis in diverse cancer cell lines, and that the SAC domain of Par-4 is sufficient to support apoptosis. Par-4 Secretion Occurs by a BFA-Sensitive Pathway and Is Not Dependent on Apoptosis We also examined whether treatment of cells with proapoptotic proteins, such as TRAIL, potentiated the secretion of endogenous Par-4. PC-3 cells were treated with TRAIL for short time intervals ( min), which were of insufficient duration to produce apoptosis, and the harvested CM and whole-cell lysates were subjected to western blot analysis for endogenous Par-4 and actin. TRAIL did not increase the levels of intracellular Par-4 in the cell lysates, yet caused increased secretion of Par-4 in the CM (Figure 2A). To determine whether Par-4 is released via the conventional cell secretory pathway, we inhibited secretory transport with brefeldin A (BFA), which blocks anterograde ER export to the Golgi resulting in obstruction of secretion. Following pretreatment with BFA, TRAIL caused a negligible increase in Par-4 in the CM, yet the amount of intracellular Par-4 was unaltered (Figure 2A). As BFA blocks protein trafficking from the ER to the Golgi, a characteristic step in cell secretion by the classical pathway, these data indicate secretion of endogenous Par-4 occurred by the classical pathway involving the ER-Golgi network. We also determined whether Par-4 secretion in the presence of TRAIL was associated with activation of apoptosis. PC-3/DN-FADD cells stably expressing dominant-negative FADD, which inhibits TRAIL-inducible apoptosis, were treated with TRAIL for short time interval (Figure 2A). As seen in Figure 2A, TRAIL-induced Par-4 secretion in these cells. These findings confirm secretion of Par-4 is not dependent on apoptosis. (D) Recombinant Par-4 and SAC protein induce apoptosis in cancer cells. Recombinant protein was subjected to SDS-PAGE and Coomassie blue (CB) staining (left panel). Various cancer or nontransformed cells were treated with 100 nm amounts of each protein for 24 hr (right panel). The cells were scored for apoptosis by ICC for active caspase-3. Panels C & D: Mean values (+ standard deviation bars) of three separate experiments are shown. Asterisk (*) indicates the difference is statistically significant (p < 0.001) by the Student s t test. Cell 138, , July 24, 2009 ª2009 Elsevier Inc. 379
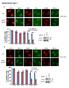
4 Figure 2. Par-4 Protein Is Secreted by an ER-Stress Inducible Pathway (A) Par-4 protein is secreted by a BFA-sensitive pathway independently of apoptosis. Cells were left untreated or pretreated with BFA (1 mm) for 30 min as indicated, then exposed to TRAIL (100 ng/ml) for 30, 60, and 180 min. Whole-cell extracts (WCE) or CM were prepared, and subjected to western blot (WB) analysis for Par-4 or actin. The CM was also subjected to SDS-PAGE followed by Coomassie blue (CB) staining to reveal the 68 kda BSA band for loading control (upper panels and lower left panel). PC-3 or PC-3/DN- FADD cells were also treated with TRAIL or left untreated (UT) for 24 hr, and scored for apoptosis by ICC for active caspase 3 (lower right panel). Mean values (+ standard deviation bars) of three separate experiments are shown. Asterisk (*) indicates the difference is statistically significant (p < 0.001) by the Student s t test. (B) Par-4 protein is secreted by an ER-stress inducible pathway. PC-3 cells were left untreated (UT) or treated with thapsigargin (TG,100 nm) or tunicamycin (TU, 5 mm) in the presence or absence of BFA (1 mm) for the indicated time intervals. CM was prepared, and subjected to SDS-PAGE followed by staining with Coomassie blue (CB; upper panel). Also, whole-cell extracts from untreated (UT) PC-3 cells or PC-3 cells treated with vehicle, TG, TU, or TRAIL for various time intervals were subjected to western blot (WB) analysis for Par- 4, GRP78, and actin (middle panel), or CHOP/ GADD153 and actin (lower panel). TRAIL increased expression of intracellular GRP78 and CHOP/GADD153 relative to untreated cells. As a whole, these findings imply secretion of Par-4 is associated with induction of ER-stress. Par-4 Secretion Is Associated with ER Stress As Par-4 induction is not dependent on apoptosis, we examined whether secretion of Par-4 is a cellular response to ER-stress. PC-3 cells were treated with prototypical inducers of ER-stress (Marchand et al., 2006), such as thapsigargin (TG; an inhibitor of sarcoplasmic/endoplasmic reticulum Ca 2+ ATPase) and tunicamycin (TU; an inhibitor of N-linked glycosylation), for short time intervals, and CM prepared from the cultures was examined for secreted Par-4 protein. As seen in Figure 2B (upper panel), TG and TU induced Par-4 secretion, which was severely attenuated by the presence of BFA. To confirm induction of ER-stress, PC-3 cells were treated with TG, TU, or TRAIL for short time intervals, and upregulation of GRP78 and CHOP/GADD153, key indicators of ER-stress was examined. As seen in Figure 2B, TG, TU, and Par-4 and SAC Bind to GRP78 To determine whether Par-4 induces apoptosis by binding to specific proteins, we performed GST-pull down assays on whole cell extracts from PC-3 cells, using either GST-Par-4 or GST as bait. GST- Par-4, but not GST, bound a number of proteins, and mass spectrometry identified the 80 kda band (Figure S2A) as GRP78 (mass spectrometry data are shown in Figure S2C and Table S1). GRP8 is a typical stress protein that is a member of the HSP70 superfamily of heat shock proteins (Lee, 2007). For verification, the proteins pulled down from the PC-3 lysates were subjected to western blot analysis for GRP78; His-SAC-GFP, but not the His-GFP control, pulled down GRP78 protein from PC-3 cells (Figure S2B). Seeking confirmation that endogenous GRP78 binds Par-4 in mammalian cells, we used PC-3 whole-cell lysates for coimmunoprecipitations with GFP, Par-4, or GRP78 antibodies, followed by immunoblotting for Par-4 or GRP78. The data demonstrate Par-4 antibody coimmunoprecipitated GRP78, and the GRP Cell 138, , July 24, 2009 ª2009 Elsevier Inc.
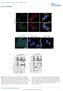
5 antibody coimmunoprecipitated Par-4 (Figure 3A), whereas the GFP antibody coimmunoprecipitated neither Par-4 nor GRP78. These findings indicate endogenous Par-4 is bound to GRP78 protein. Although the above data indicate Par-4 binds GRP78, it was unclear if the SAC domain and GRP78 interact in the same manner. We transfected PC-3 cells with GFP, Par-4-GFP, or SAC-GFP expression constructs, and subjected the cell lysates to immunoprecipitation with either GFP or Par-4 antibodies. As seen in Figure 3B, GRP78 coimmunoprecipitated with Par-4- GFP (Figure 3B) when we used the Par-4 antibody with lysates from Par-4-GFP transfectants. In coimmunoprecipitations using Par-4 antibody and lysates from cells that were either untransfected or transfected with GFP, we coimmunoprecipitated GRP78 with endogenous Par-4. Likewise, when we used GFP antibodies with lysates from the SAC-GFP transfectants, GRP78 coimmunoprecipitated with SAC-GFP, whereas coimmunoprecipitations using GFP antibodies with lysates from either GFP-transfected cells or untransfected cells (as controls) did not yield GRP78. These data indicate Par-4 antibody coimmunoprecipitates endogenous Par-4 and ectopic Par-4-GFP with endogenous GRP78, and GFP antibody coimmunoprecipitates ectopic SAC-GFP with endogenous GRP78 (Figure 3B). Accordingly, the SAC domain is sufficient for interactions with GRP78. As recent studies suggest cell surface GRP78 serves as a receptor for soluble ligands to induce either apoptosis or growth stimulation (Misra et al., 2002; Davidson et al., 2005; Gonzalez-Gronow et al., 2006), we explored whether (1) Par-4 colocalizes with GRP78 at the plasma membrane, and (2) cell surface GRP78 is essential for apoptosis by extracellular Par-4 and extracellular SAC. Treatment of PC-3 cells with TRAIL, TRX-Par-4, and TRX-SAC, but not TRX, caused upregulation of ER stress proteins GRP78 and CHOP/GADD153 (Figure S3A). FACS analysis performed on PC-3 cells, which were not chemically fixed in order to allow detection of cell surface proteins, indicated a significant increase (p < by Student t test) in GRP78 expression on the cell surface upon treatment with TRAIL, TRX-Par-4, or TRX-SAC (Figures 3C and S3B). Similarly, TRAIL treatment caused an increase in the expression of GRP78 and Par-4 protein in the membrane fraction, and GRP78 and Par- 4 were coimmunoprecipitated from this fraction (Figure S3F). To further confirm the cell surface interaction of endogenous GRP78 and Par-4, PC-3 cells were treated with TRAIL, then intact cells were biotinylated to label cell surface proteins. Whole-cell extracts were subjected to immunoprecipitation with GRP78, Par-4, or PTEN control antibody, and biotinylated proteins were detected with avidin-hrp. The GRP78 antibody, as well as the Par-4 antibody, coimmunoprecipitated biotinylated Par-4 and biotinylated GRP78 (Figure 3D). Moreover, TRX-Par-4 but not TRX control protein, pulled-down biotinylated GRP78 (Figure 3D). These findings support Par-4 binding GRP78 at the cell membrane. To examine colocalization of Par-4 and GRP78, we exposed PC-3 cells and BPH-1 cells (which do not respond to TRAIL; see Figure S3A) to TRAIL or vehicle, then performed ICC analysis for Par-4 and GRP78. As seen in Figure 3E, PC-3 and BPH-1 cells treated with vehicle showed colocalization of Par-4 and GRP78 in the ER. However, upon treatment with TRAIL, PC-3 cells showed membrane translocation of GRP78, whereas BPH-1 cells retained GRP78 in the ER (Figure 3E). Notably, in TRAIL-treated PC-3 cells, but not TRAIL-treated BPH-1 cells, Par-4 colocalized with GRP78 at the plasma membrane (Figures 3E and S3E), indicating Par-4 colocalizes with GRP78 at the plasma membrane in response to TRAIL exposure. We further confirmed colocalization of GRP78 and Par-4 with the ER marker, calnexin, and plasma membrane marker, Na + /K + -ATPase, in PC-3 cells and BPH-1 treated with either TRAIL or vehicle (Figure S3C). GRP78 Cell Surface Interaction Is Essential for Apoptosis by Extracellular Par-4 We next determined whether cell surface GRP78 is essential for apoptosis by extracellular Par-4. PC-3 cells were pretreated with GRP78 or control GST protein, subsequently treated with TRX- Par-4, and finally scored for apoptotic cells. TRX-Par-4 induced apoptosis in the presence of GST protein, but failed to induce apoptosis in the presence of GRP78 protein (Figure 4A). To further elucidate the interaction between extracellular Par-4 and cell surface GRP78, we preincubated PC-3 cells with neutralizing antibodies against various regions of GRP78, and then treated the cells with TRX-Par-4 or TRX. As seen in Figure 4B, apoptosis induced by TRX-Par-4 was blocked by the N-terminal-GRP78 antibody (NT-GRP78/N-20), but not by antibodies (H-129 for amino acids , or CT-GRP78/ C-20) targeting the C terminus of GRP78. Similarly, in lung cancer cells (H460) and cervical cancer cells (HeLa) apoptosis by TRX-Par-4 was blocked by the N-terminal GRP78 antibody, but not the C-terminal GRP78 antibodies (Figure S4A). These findings indicate the N-terminal region of cell surface GRP78 is essential for interaction with, and apoptosis by, extracellular Par-4. Apoptosis induced by TRX-Par-4 or TRX-SAC was abrogated by preincubation of PC-3 cells with NT-GRP78 neutralizing antibody, but not with several other antibodies (Figure 4C). Interestingly, when PC-3 cells were transfected with Par-4-GFP, SAC-GFP, or GFP constructs, and incubated with neutralizing antibody for GRP78 (NT-GRP78), Par-4, or PTEN (control), antibody-mediated inhibition of cell surface GRP78 prevented extracellular-par-4 or -SAC from inducing apoptosis in the primary transfectants and nontransfected cells (Figure S4C). In addition, RNAi-mediated knock-down of GRP78 in PC-3 cells resulted in diminished expression of GRP78, both in whole-cell lysates and at the plasma membrane, and these cells were resistant to apoptosis by TRX-Par-4 (Figure S5). Collectively, these results suggest extracellular-par-4 and -SAC induce apoptosis in PC- 3 cells (transfected or nontransfected) by a GRP78-dependent mechanism. Because we noted increased secretion of Par-4 by PC-3 cells in response to TRAIL (Figure 2B), and because PC-3 cells, but not BPH-1 cells, are sensitive to apoptosis induced by TRAIL (Figure S3A), extracellular-par-4, and extracellular-sac (Figure 1D), we asked whether (1) extracellular-par-4 is essential for apoptosis by TRAIL, and (2) extracellular-par-4/grp78 interaction is required to induce apoptosis by TRAIL. PC-3 cells were incubated with neutralizing antibody for GRP78, Par-4, or PTEN (for control), and next treated with either TRAIL or vehicle. As Cell 138, , July 24, 2009 ª2009 Elsevier Inc. 381
6 Figure 3. Par-4, via its SAC Domain, Binds to GRP78 and Colocalizes with GRP78 in the ER and at the Cell Membrane (A) Par-4 binds to GRP78. Whole-cell extracts from PC-3 cells were subjected to immunoprecipitation (IP) with antibodies for Par-4, GRP78, or GFP, and the immunoprecipitated complexes were resolved by SDS-PAGE and subjected to western blot (WB) analysis for GRP78 and Par-4. PC-3 whole-cell extracts were used as input. (B) Par-4 binds to GRP78 via its SAC domain. PC-3 cells were transfected with GFP, Par-4-GFP, or SAC-GFP expression constructs or left untransfected, and the cell lysates were subjected to immunoprecipitation (IP) with Par-4 or GFP antibody. The immunoprecipitated complexes were subjected to western blot (WB) analysis for GRP78 or GFP. Whole-cell extracts from the transfectants were used as input. Asterisks indicate breakdown products. (C) Recombinant Par-4, SAC, and TRAIL individually increase GRP78 expression at the cell surface. PC-3 cells were treated with TRAIL (100 ng/ml), TRX-Par-4 (100 nm), TRX-SAC (100 nm), or TRX (250 nm) for various time intervals. The cells were collected after 6 hr (unfixed, so the antibody could detect only cell surface 382 Cell 138, , July 24, 2009 ª2009 Elsevier Inc.
7 seen in Figure 4D, TRAIL-inducible apoptosis was inhibited by the Par-4 antibody, as well as by the GRP78 antibody, but not by the PTEN antibody. These data imply secreted Par-4 and cell surface GRP78 are requisite for TRAIL-induced apoptosis. Given these in vitro observations, we explored Par-4 secretion and biological activity in vivo. Our laboratory recently described SAC-transgenic mice that are resistant to the growth of spontaneous tumors, whereas GFP-transgenic mice and littermate control mice develop spontaneous tumors (Zhao et al., 2007). By extension, Par-4 transgenic mice are also resistant to the growth of spontaneous tumors (Table S2). The bone marrow cells and serum of GFP- and Par-4-transgenic animals, as well as littermate control mice, were tested for expression of GFP and Par-4 protein. As seen in Figure 4E, the bone marrow cells from the transgenic animals expressed GFP or Par-4-GFP, while the serum from the Par-4-transgenic mice, but not from the GFP and littermate control mice, contained detectable levels of Par- 4-GFP protein. Next, we tested the biological activity of the serum from Par-4 transgenic mice against normal/immortalized cells and cancer cell lines. The serum from Par-4-GFP-transgenic mice, but not from GFP-transgenic mice and littermate control mice, induced apoptosis in PC-3 cells, but not in BPH-1 cells (Figure 4E, bottom left panel). To verify the apoptotic activity of the serum from the Par-4-transgenic mice was indeed due to the Par-4-GFP protein, serum samples were pretreated with GFP or Par-4 antibody to neutralize Par-4-GFP protein, or with GRP78 antibody to impede binding of Par-4-GFP to cell surface GRP78, and applied to PC-3 cells in culture. As seen in Figure 4E (bottom right panel), GFP and Par-4 antibodies, but not the control PTEN antibody, prevented the serum from Par-4-GFP mice from inducing apoptosis in the PC-3 cells. Significantly, GRP78 antibody also negated the apoptotic effects of the serum from the Par-4-GFP mice (Figure 4E, bottom right panel). These results indicate the Par- 4 protein is secreted in mice, and effects apoptosis via cell surface GRP78 binding, thereby inhibiting the growth of tumor cells. Role of Intracellular Par-4 in Regulation of Apoptosis by Extracellular Par-4 As intracellular Par-4 is essential for apoptosis in cancer cells (Goswami et al., 2005), we investigated its role in the apoptotic action of extracellular Par-4. In this effort, we knocked-down the expression of endogenous Par-4 by RNA-interference, and then exposed the cells to recombinant Par-4, SAC, or control protein. Cells transfected with control sirna underwent apoptosis upon treatment with TRX-Par-4 or TRX-SAC, whereas cells transfected with Par-4 sirna did not (Figure 5A). Likewise, endogenous Par-4 knock-down did not alter GRP78 levels in GFP control protein treated cells, but inhibited induction of GRP78 by TRX-Par-4 or TRX-SAC (Figure 5A). We conclude endogenous Par-4 expression is essential for ER-stress dependent induction of GRP78 and for apoptosis by extracellular Par-4. As cell surface GRP78 is essential for apoptosis induced by extracellular Par-4, we sought to determine whether intracellular Par-4 regulates GRP78 expression at the cell surface. PC-3 cells were incubated with Par-4 or control sirna duplexes, treated with TRX or TRX-Par-4, and examined for cell surface GRP78 expression by FACS analysis. Knock-down of endogenous Par-4 by sirna resulted in severe reduction of cell surface GRP78 expression (Figure 5B). However, total GRP78 protein levels in the cell lysates were unaltered by Par-4 knock-down (Figure 5B), implying that Par-4 was essential for trafficking GRP78 to the plasma membrane. Next, we restored expression of cell surface GRP78 by transfecting the PC-3 cells with membrane-directed full-length GRP78 (mgrp78) or its N-terminal mutant (mdn-grp78; lacking 66 amino acids at the N-terminus), following Par-4 knock down, and tested the cells for sensitivity to extracellular Par-4. TRX-Par-4 induced apoptosis in cells transfected with mgrp78, but not mdn-grp78 (Figure 5C). Similar observations were made in H460 cells and HeLa cells (Figures S6 and S7A). We also tested immortalized BPH-1 cells, which do not respond to extracellular Par-4, for apoptosis in response to extracellular Par-4 after transfection with mgrp78 or mdn-grp78. Cell membrane-directed full-length mgrp78, but not mdn-grp78, rendered BPH-1 cells sensitive to apoptosis by TRX-Par-4 (Figure S7B). As expected, Par-4 binds to full-length mgrp78, but not mdn-grp78 (Figures S7C). Collectively, these findings indicate intracellular Par-4 is essential for trafficking of GRP78 to the plasma membrane, and that extracellular Par-4 induces apoptosis by binding to the N-terminal region of cell surface GRP78. DISCUSSION We have uncovered a novel extracellular role for Par-4 protein. Par-4 is spontaneously secreted by cultured cells in conditioned GRP78) and subjected to FACS analysis for GRP78. Mean values (+ standard deviation bars) of three separate experiments are shown. Asterisk (*) indicates the difference is statistically significant (p < 0.001) by the Student s t test. (D) GRP78 binds Par-4 at the cell surface. Cells were treated with TRAIL (100 ng/ml) or vehicle for 6 h, and then labeled with cell-impermeable sulfo-nhs-lcbiotin. Whole-cell extracts (WCE) of the TRAIL-treated/biotinylated samples were subjected to immunoprecipitation (IP) with the indicated antibodies (left panels). For the pull-down of biotinylated GRP78 from TRAIL-treated cells, the corresponding WCE (10 mg) were incubated with polyhistidine-tagged recombinant proteins TRX-Par-4 or TRX control (2 mg of each) for 2 h at 4 C. Bound complexes were purified with metal affinity resin beads (middle panels). The immunoprecipitated proteins (left panels), bound complexes (middle panels), and also the corresponding WCE (indicated as input, right panels) were subjected to SDS- PAGE, and blotted with avidin-hrp or with GRP78, Par-4, or TRX antibody. The 50 kda band indicated by the asterisk is unidentified. (E) Par-4 colocalizes with GRP78 in the ER and in the cell membrane. Cells were treated with TRAIL (100 ng/ml) or vehicle for 6 h, and subjected to ICC for Par-4 (green fluorescence), GRP78 (red fluorescence), and nuclei were revealed by DAPI (cyan fluorescence). White arrows show colocalization of Par-4 and GRP78 in merged images (yellow fluorescence). A total of 400 cells were scored for colocalization of Par-4 and GRP78 at the cell membrane in each experiment, and mean values (+ standard deviation bars) of three separate experiments are shown in the right panel. Asterisk (*) indicates the difference is statistically significant (p < ) by the Student s t test. Magnification: For colocalization images- PC-3/TRAIL (200x); PC-3/vehicle (120x); and BPH-1 (80x); for all other BPH-1 images (40x) and PC-3 images (80x). Cell 138, , July 24, 2009 ª2009 Elsevier Inc. 383
8 Figure 4. Extracellular Par-4, SAC, and TRAIL are Dependent on Cell Surface GRP78 for Induction of Apoptosis (A) Extracellular GRP78 neutralizes the apoptotic action of TRX-Par-4. PC-3 cells were pretreated with vehicle, GST, or GRP78 protein, and then treated with TRX-Par-4, TRX protein, or vehicle. After 24 hr, cells were subjected to ICC for active caspase-3 (green fluorescence, see Figure S4B), and apoptosis was quantified. The integrity of the purified proteins was verified by SDS-PAGE and Coomassie blue (CB) staining using protein molecular weight markers (upper panel). (B) Neutralizing antibody (NT-GRP78) directed toward the N-terminal region of GRP78 inhibits apoptosis by extracellular Par-4. Cells were preincubated with the indicated antibodies for 30 min, and then treated with TRX-Par-4 or TRX (100 nm of each) for 24 hr. Apoptotic cells were scored by ICC for active caspase-3. (C) NT-GRP78-antibody neutralizes the apoptotic action of exogenous Par-4 and SAC. Cells were pretreated with PTEN, NT-GRP78, DR4, or DR5 antibody for 30 min, and then treated with TRX- Par-4 (rpar-4), TRX-SAC (rsac), or TRX. After 24 h, apoptosis was quantified by ICC for active caspase-3. (D) Par-4- and NT-GRP78-antibodies individually neutralize the apoptotic action of TRAIL. PC-3 cells were pretreated with PTEN, NT-GRP78, or Par-4 antibody, then treated with TRAIL (100 ng/ ml) or vehicle. After 24 h, apoptosis was quantified by ICC for active caspase-3. (E) Par-4, secreted in the serum of Par-4-transgenic mice, induces GRP78-mediated apoptosis in PC-3 cells. Bone marrow cells from littermate control, GFP-, and Par-4-GFP-transgenic mice were analyzed by ICC for GFP (upper left panel), and the serum from these mice was examined by western blot for secreted Par-4 (upper right panel). Loading was verified by CB staining for serum albumin (upper right panel). Cells were treated with the serum samples (20% final concentration) either in the absence or presence of neutralizing antibodies, and, after 24 h, apoptosis was quantified by ICC for active caspase-3 (lower panel). In panels A-E, mean values (+ standard deviation bars) of three separate experiments are shown. Asterisk (*) indicates the difference is statistically significant (p < 0.001) by the Student s t test. medium, and Par-4 transgenic mice secrete Par-4 in their serum. Spontaneous secretion of Par-4 was noted in both normal and cancer cell cultures, and was further enhanced by exposure to ER stress-producing agents, such as TG, TU, and TRAIL. Secretion of Par-4 occurred by a BFA-sensitive pathway, and was not dependent on apoptosis of the cells. Importantly, extracellular Par-4 was functionally active, and bound to cell surface GRP78 protein to induce apoptosis in cancer cells. The SAC domain of Par-4 also interacts with GRP78, therefore implying Par-4 binds to GRP78 independently of its carboxy-terminal leucine zipper domain. Extracellular Par-4/SAC binding to cell surface GRP78 produces a robust ER stress response loop involving upregulation and trafficking of GRP78 to the cell membrane, in an intracellular Par-4-dependent manner (Figure 6). Moreover, extracellular Par-4 activates caspase-8 and caspase-3 in a FADD-dependent manner (Figures S8A S8E). Consistent with a recent report suggesting ER-stress causes activation of PERK (PKR-like ER kinase), a proximal component of the ER stress pathway, which is known to activate FADD and caspase-8 dependent apoptosis (Park et al., 2008), we noted inhibition of PERK expression by RNAi led to inhibition of caspase-8 dependent apoptosis by extracellular Par-4 and TRAIL (Figure S8F). This extracellular function of Par-4/SAC constitutes a paradigm shift in our interpretation of the mechanism of Par-4 action, and uncovers a broader therapeutic significance for this cancer cell-selective apoptotic molecule. 384 Cell 138, , July 24, 2009 ª2009 Elsevier Inc.
9 Figure 5. Cell Membrane-Directed GRP78 Overrides the Effect of Intracellular Par-4 Knockdown and Restores Sensitivity to TRX-Par-4 (A) Endogenous Par-4 expression is essential for apoptosis by extracellular Par-4. PC-3 cells were transfected with sirna duplexes for Par-4 or control sirna. The cells were treated with TRX- Par-4, TRX-SAC, or GFP protein for control for 24 hr, and apoptotic cells were scored by evaluating active caspase-3 by ICC (upper panel). Western blot analysis (lower panel) confirmed both knock-down of Par-4 expression by sirna and its effect on GRP78 expression. (B) Intracellular Par-4 regulates trafficking of cell surface GRP78. PC-3 cells were transfected with sirna duplexes for Par-4 or control sirna, treated with TRX or TRX-Par-4, and processed as intact, unfixed cells for expression of cell surface GRP78 by FACS analysis (lower panel). Knockdown of intracellular Par-4 by sirna was confirmed by western blot analysis of corresponding whole-cell lysates (upper panel). (C) Expression of membrane-targeted GRP78 restores sensitivity to TRX-Par-4 following intracellular Par-4 knock-down. PC-3 cells were transfected with sirna duplexes for Par-4 or control sirna, and knock-down of intracellular Par-4 was confirmed as indicated in Panel B. The cells were then transfected with vector, cell membrane-directed full-length GRP78 (mgrp78), or DN mutant of GRP78 (mdn-grp78), and expression of endogenous and ectopic GRP78 in whole-cell lysates was confirmed by western blot analysis (left panel inset). Finally, the transfectants were subjected to treatment with TRX or TRX-Par- 4 for 24 h, and apoptotic cells were scored by evaluating active caspase-3 by ICC (left panel). Expression of cell surface GRP78, as determined by FACS analysis, is shown for Par-4 knockdown cells transfected with the mgrp78 construct or vector and subsequently treated with TRX-Par-4 (right panel). In panels A-C, mean values (+ standard deviation bars) of three separate experiments are shown. Asterisk (*) indicates the difference is statistically significant (p < 0.001) by the Student s t test. Secretion of Par-4 Our studies indicate intracellular Par-4 colocalizes with GRP78 in the ER, and is secreted spontaneously via the classical BFAsensitive pathway involving the ER/Golgi network. Par-4 secretion is further enhanced by TG, TU, and TRAIL. Interestingly, all three agents caused upregulation of GRP78 and CHOP/GADD153, two key molecular indicators of ER stress/unfolded protein response. Although TG and TU are known to produce ER stress (Marchand et al., 2006), and 3,3 0 -diindolylmethane induces apoptosis by ER stress signaling via activation of caspase-8 and 3(Abdelrahim et al., 2006), the role of ER stress in the action of TRAIL was not recognized previously. Similar to many other proteins that are secreted into the extracellular environment, Par-4 does not have a readily discernible signal peptide sequence. Par-4 may, however, contain atypical short signal peptide sequence(s), and this possibility is currently being investigated. Apoptotic Signaling via GRP78 Cell Surface Receptor In this report we provide several lines of evidence to confirm a physical and functional interaction between Par-4 and GRP78 at the cell surface. These include: (1) Par-4 and GRP78 colocalize at the plasma membrane, as judged by ICC studies using markers for the ER and plasma membrane; (2) Par-4 and GRP78 coimmunoprecipitate from the membrane fraction using either Par-4 or GRP78 antibody; (3) Par-4 and GRP78 were biotinylated using cell-impermeable sulfo-nhs-lc-biotin, and coimmunoprecipitated by Par-4 or GRP78 antibody; (4) neutralizing antibodies against Par-4 or the N-terminus of GRP78 inhibit apoptosis by TRAIL or Par-4 in intact cells; (5) RNAi-mediated inhibition of Par-4 or GRP78 results in diminished GRP78 expression at the cell surface and resistance to apoptosis by exogenous Par-4; (6) cell membrane-directed GRP78 coimmunoprecipitates with Par-4; and (g) cell membrane-directed GRP78 can restore Cell 138, , July 24, 2009 ª2009 Elsevier Inc. 385
10 Figure 6. Model for Apoptosisby Extracellular Par-4 ER stress (a), induced by extracellular insults such as TRAIL, causes translocation of the Par-4-GRP78 complex from the ER to the plasma membrane (b). This leads to elevated expression of GRP78 and Par-4 at the plasma membrane (c), and secretion of Par-4 (d). Extracellular Par-4 binds to cell surface GRP78 (e), and induces yet more ER stress (f), thereby activating the loop for translocation of ER GRP78 to the plasma membrane, in an intracellular Par-4-dependent manner. Finally, ER stress involves upregulation of the ER component phospho-perk, and activation of caspase-8 in a PERK-dependent manner (g). Activation of caspase-8 is also dependent on activation of FADD. Collectively, these events lead to activation of caspase-3 and apoptosis. This model is not restricted to prostate cancer cells, as the key features were recapitulated in lung and cervical cancer cells (Figure S11). sensitivity to exogenous Par-4. Collectively, these data demonstrate Par-4 interacts with GRP78 at the cell surface. GRP78 exists primarily as an ER protein with intracellular chaperone functions, and is upregulated in response to ER stress in order to diminish growth inhibitory signals, and promote cell survival (Pootrakul et al., 2006; Lee, 2007). However, GRP78 is also expressed on the surface of most cancer cells and a few nontransformed cells, and serves as a receptor for extracellular ligands, such as alpha-2 macroglobulin (promotes cell proliferation) and Kringle 5 (induces apoptosis). It is also reported that VDAC may serve as a coreceptor for Kringle 5-mediated apoptosis of endothelial cells (Gonzalez-Gronow et al., 2006). Also, synthetic peptides have been specifically designed to bind cell surface GRP78, and induce apoptotic signaling (Arap et al., 2004). Although Kringle 5 has been reported to induce caspase-7 activation following GRP78 binding (Davidson et al., 2005), extrinsic Par-4 and its SAC domain induce activation of caspase-8 and 3, but not caspase-7 or 9 following cell surface GRP78 binding (Figures S8 A F and S9). This indicates ligand-specific activation of distinct caspases prompts a similar cellular outcome following binding to cell surface GRP78. Consistent with the role of ER stress in the action of TRAIL and exogenous Par-4, cancer cells (such as DU145) that are resistant to apoptosis by TRAIL or exogenous Par-4 can be rendered sensitive to the action of these proteins by bortezomib (Velcade), which induces ER stress (Figure S10). We are currently investigating the precise role of the ER components phospho-perk and phospho-eif2ea, which play a role in c-flip suppression following CD95 activation (Park et al., 2008), in activation of FADD and caspase-8 by exogenous TRAIL and Par-4. It is also noteworthy that, unlike all of the previously described proteinprotein interactions of Par-4 that are mediated by its carboxyterminal leucine zipper domain, binding of Par-4 to GRP78 is uniquely mediated by the SAC domain. Further deletion/mutational analysis will uncover the specific residues or sub-domains within the SAC sequence that promote binding and/or apoptotic signaling. Mutual Dependency of Extracellular and Intracellular Par-4 for Induction of Apoptosis In the present study, ectopic overexpression of Par-4 and SAC induced apoptosis not only in transfected cells, but also in nontransfected cells within the same cell culture. Apoptosis in the transfected and nontransfected cells was inhibited by antibodies targeting either extracellular Par-4 or GRP78, implying both paracrine and autocrine effects for secreted Par-4. Consistently, inhibition of intracellular Par-4 expression by RNAi negates caspase 3 activation and apoptosis by extracellular Par-4. These findings reveal a mutual dependency between extracellular and intracellular Par-4 in activating apoptosis. In contrast to intracellular GRP78, which may generally serve anti-apoptotic functions (Lee, 2007), cell surface GRP78 promotes apoptosis by Par-4. Furthermore, intracellular Par-4 regulates the expression of GRP78 at the cell surface, as Par-4 knock-down does not alter total GRP78 levels in whole-cell lysates, yet the amount of cell surface GRP78 is severely down-modulated. As exogenous Par-4/SAC-inducible expression of GRP78 is inhibited upon knock-down of endogenous Par-4, we infer inadequate GRP78 expression at the cell surface precludes an ER stress-dependent increase in GRP78 in response to exogenous Par-4/SAC. These results are consistent with our observation that endogenous Par-4 is essential for cell surface expression of GRP78. Although both nontransformed and cancer cells express GRP78 at the cell surface, only cancer cells exhibit ER stress and apoptosis in response to extracellular Par-4. Par-4 binds to the cell surface GRP78 molecules that are initially available on cancer cells, thereby causing ER stress that further enhances both translocation of GRP78 molecules to the cell surface and apoptosis. Nontransformed cells neither respond to extracellular Par-4 by ER stress, nor do they allow such trafficking of GRP78 to the cell surface. However, artificial overexpression of GRP78 at the cell membrane can promote apoptosis of the nontransformed cells. Therefore, differential ER stress response may contribute to the selective increase in cell surface trafficking of 386 Cell 138, , July 24, 2009 ª2009 Elsevier Inc.
11 GRP78, thus leading to Par-4-induced apoptosis in cancer cells (also see Supplemental Discussion). Similar to Par-4 and its SAC domain, TRAIL is known to selectively induce apoptosis in cancer cells only. This study reveals TRAIL causes secretion of Par-4, and that secreted Par-4 regulates apoptosis by TRAIL. Consequently, inhibition of either extracellular Par-4 or cell surface GRP78 impedes apoptosis by TRAIL. Our results further reveal intracellular Par-4 is essential for translocation of GRP78 from the ER to the cell surface in response to TRAIL, thereby triggering the binding of Par-4 and GRP78 at the cell surface to boost ER stress, and leading to activation of PERK and caspase-8. Inhibition of intracellular Par-4 by RNAi blocked all of these responses, as well as apoptosis by TRAIL (data not shown). These findings indicate extracellular and intracellular Par-4 are both essential for apoptosis by TRAIL. In summary, the present study identified a novel extrinsic pathway for apoptosis by Par-4 acting via its SAC domain. This pathway is essential for apoptosis by TRAIL, which, similar to Par-4, induces cancer-specific apoptosis. The identification of an extracellular role for Par-4 and SAC significantly broadens their therapeutic potential for primary and metastatic tumors. EXPERIMENTAL PROCEDURES Cell Culture, Plasmids, Recombinant Proteins, and Chemical Reagents Human immortalized epithelial cells BPH-1, prostate cancer cells PC-3, lung cancer cells H460, cervical cancer cells HeLa, human primary lung fibroblasts HEL, human embryonic kidney cells HEK293, and mouse fibroblasts NIH 3T3 were from American Type Culture Collection, MD. The preparation of recombinant proteins and plasmid constructs is detailed in Supplemental Experimental Procedures. The control sirna and sirna for Par-4 were from Dharmacon, Inc. and SantaCruz Biotechnology, Inc. TRAIL was purchased from R. & D. Systems. The antibodies for active caspases and ERK1/2 were from Cell Signaling, Inc. The monoclonal antibody for b-actin, purified GRP78 protein, and BFA were from Sigma Corp. All other antibodies (including those for GRP78) were from SantaCruz Biotechnology, Inc. (Santa Cruz, CA). GRP78 antibodies C-20, N-20, and H-129, were tested for inhibition of cell surface GRP78 interaction with Par-4; H-129 or 76-E6 was used for ICC and western blot analysis; and H-129 was used for immunoprecipitation and FACS analysis. Cell Surface Biotinylation Cell surface proteins were biotinylated and visualized by treatment with avidinhorseradish peroxidase (avidin-hrp) asdescribedinthesupplementalmethods. GST-Pull-Down Assay, Coimmunoprecipitation, and Western Blot Analysis Purified GST (6 mg) or GST-Par-4 (3 mg) protein was incubated with glutathione beads for 30 min, and subsequently washed and incubated with cell extracts (10 mg) for 18 h at 4 C. Protein eluted from the beads was resolved by SDS- PAGE, and subjected to Coomassie blue staining and mass spectrometry at the Protein Core Facility of the Columbia University Medical Center, NY. Proteins were coimmunoprecipitated and detected by western blot analysis as described (Goswami et al., 2005). Immunocytochemistry, Apoptosis, and FACS Analysis Immunocytochemical analysis and apoptosis procedures have been described (Goswami et al., 2005). For fluorescence-activated cell sorting (FACS) analysis, PC-3 cells were treated with recombinant proteins for 6 h and then, without fixing them to allow detection of GRP78 at the cells surface, the cells were subjected to FACS analysis using either GRP78 primary antibody (H-129) or no primary antibody as control, and R-phytoerythrin-conjugated secondary antibody (from Invitrogen, Inc.). Statistical Analysis All experiments were performed in triplicate to verify the reproducibility of the findings. Statistical analyses were carried out with Statistical Analysis System software (SAS Institute, Cary, NC) and P values were calculated using the Student t test. SUPPLEMENTAL DATA Supplemental Data include Supplemental Discussion, Supplemental Experimental Procedures, Supplemental References, eleven figures, and two tables and can be found with this article online at S (09) ACKNOWLEDGMENTS This study was supported by NIH/NCI grants CA60872, CA105453, and CA84511 (to V.M.R.). We thank Krishna Murthi Vasudevan (Dana-Farber Cancer Institute) for critical reading of the manuscript. Received: August 17, 2008 Revised: January 14, 2009 Accepted: May 1, 2009 Published: July 23, 2009 REFERENCES Abdelrahim, M., Newman, K., Vanderlaag, K., Samudio, I., and Safe, S. (2006). 3,3 0 -diindolylmethane (DIM) and its derivatives induce apoptosis in pancreatic cancer cells through endoplasmic reticulum stress-dependent upregulation of DR5. Carcinogenesis 27, Arap, M.A., Lahdenranta, J., Mintz, P.J., Hajitou, A., Sarkis, A.S., Arap, W., and Pasqualini, R. (2004). Cell surface expression of the stress response chaperone GRP78 enables tumor targeting by circulating ligands. Cancer Cell 6, Chakraborty, M., Qiu, S.G., Vasudevan, K.M., and Rangnekar, V.M. (2001). Par-4 drives trafficking and activation of Fas and Fasl to induce prostate cancer cell apoptosis and tumor regression. Cancer Res. 61, Cheema, S.K., Mishra, S.K., Rangnekar, V.M., Tari, A.M., Kumar, R., and Lopez-Berestein, G. (2003). Par-4 transcriptionally regulates Bcl-2 through a WT1-binding site on the bcl-2 promoter. J. Biol. Chem. 278, Cook, J., Krishnan, S., Ananth, S., Sells, S.F., Shi, Y., Walther, M.M., Linehan, W.M., Sukhatme, V.P., Weinstein, M.H., and Rangnekar, V.M. (1999). Decreased expression of the pro-apoptotic protein Par-4 in renal cell carcinoma. Oncogene 18, Davidson, D.J., Haskell, C., Majest, S., Kherzai, A., Egan, D.A., Walter, K.A., Schneider, A., Gubbins, E.F., Solomon, L., Chen, Z., et al. (2005). Kringle 5 of human plasminogen induces apoptosis of endothelial and tumor cells through surface-expressed glucose-regulated protein 78. Cancer Res. 65, Diaz-Meco, M.T., Municio, M.M., Frutos, S., Sanchez, P., Lozano, J., Sanz, L., and Moscat, J. (1996). The product of par-4, a gene induced during apoptosis, interacts selectively with the atypical isoforms of protein kinase C. Cell 86, El-Guendy, N., Zhao, Y., Gurumurthy, S., Gurumurthy, S., Burikhanov, R., and Rangnekar, V.M. (2003). Identification of a unique core domain of par-4 sufficient for selective apoptosis induction in cancer cells. Mol. Cell. Biol. 23, Garcia-Cao, I., Duran, A., Collado, M., Diaz-Meco, M.T., Serrano, M., and Moscat, J. (2003). Tumour-suppression activity of the proapoptotic regulator Par4. EMBO Rep. 6, Gonzalez-Gronow, M., Cuchacovich, M., Llanos, C., Urzua, C., Gawdi, G., and Pizzo, S.V. (2006). Prostate cancer cell proliferation in vitro is modulated by Cell 138, , July 24, 2009 ª2009 Elsevier Inc. 387
hexahistidine tagged GRP78 devoid of the KDEL motif (GRP78-His) on SDS-PAGE. This
 SUPPLEMENTAL FIGURE LEGEND Fig. S1. Generation and characterization of. (A) Coomassie staining of soluble hexahistidine tagged GRP78 devoid of the KDEL motif (GRP78-His) on SDS-PAGE. This protein was expressed
SUPPLEMENTAL FIGURE LEGEND Fig. S1. Generation and characterization of. (A) Coomassie staining of soluble hexahistidine tagged GRP78 devoid of the KDEL motif (GRP78-His) on SDS-PAGE. This protein was expressed
Supplementary Figure 1. Normal T lymphocyte populations in Dapk -/- mice. (a) Normal thymic development in Dapk -/- mice. Thymocytes from WT and Dapk
 Supplementary Figure 1. Normal T lymphocyte populations in Dapk -/- mice. (a) Normal thymic development in Dapk -/- mice. Thymocytes from WT and Dapk -/- mice were stained for expression of CD4 and CD8.
Supplementary Figure 1. Normal T lymphocyte populations in Dapk -/- mice. (a) Normal thymic development in Dapk -/- mice. Thymocytes from WT and Dapk -/- mice were stained for expression of CD4 and CD8.
TRAF6 ubiquitinates TGFβ type I receptor to promote its cleavage and nuclear translocation in cancer
 Supplementary Information TRAF6 ubiquitinates TGFβ type I receptor to promote its cleavage and nuclear translocation in cancer Yabing Mu, Reshma Sundar, Noopur Thakur, Maria Ekman, Shyam Kumar Gudey, Mariya
Supplementary Information TRAF6 ubiquitinates TGFβ type I receptor to promote its cleavage and nuclear translocation in cancer Yabing Mu, Reshma Sundar, Noopur Thakur, Maria Ekman, Shyam Kumar Gudey, Mariya
Supplementary Figure 1.TRIM33 binds β-catenin in the nucleus. a & b, Co-IP of endogenous TRIM33 with β-catenin in HT-29 cells (a) and HEK 293T cells
 Supplementary Figure 1.TRIM33 binds β-catenin in the nucleus. a & b, Co-IP of endogenous TRIM33 with β-catenin in HT-29 cells (a) and HEK 293T cells (b). TRIM33 was immunoprecipitated, and the amount of
Supplementary Figure 1.TRIM33 binds β-catenin in the nucleus. a & b, Co-IP of endogenous TRIM33 with β-catenin in HT-29 cells (a) and HEK 293T cells (b). TRIM33 was immunoprecipitated, and the amount of
Cells and reagents. Synaptopodin knockdown (1) and dynamin knockdown (2)
 Supplemental Methods Cells and reagents. Synaptopodin knockdown (1) and dynamin knockdown (2) podocytes were cultured as described previously. Staurosporine, angiotensin II and actinomycin D were all obtained
Supplemental Methods Cells and reagents. Synaptopodin knockdown (1) and dynamin knockdown (2) podocytes were cultured as described previously. Staurosporine, angiotensin II and actinomycin D were all obtained
Supplementary figure legends
 Supplementary figure legends Supplementary Figure 1. Exposure of CRT occurs independently from the apoptosisassociated loss of the mitochondrial membrane potential (MMP). (A) HeLa cells treated with MTX
Supplementary figure legends Supplementary Figure 1. Exposure of CRT occurs independently from the apoptosisassociated loss of the mitochondrial membrane potential (MMP). (A) HeLa cells treated with MTX
Supplementary Figure 1: si-craf but not si-braf sensitizes tumor cells to radiation.
 Supplementary Figure 1: si-craf but not si-braf sensitizes tumor cells to radiation. (a) Embryonic fibroblasts isolated from wildtype (WT), BRAF -/-, or CRAF -/- mice were irradiated (6 Gy) and DNA damage
Supplementary Figure 1: si-craf but not si-braf sensitizes tumor cells to radiation. (a) Embryonic fibroblasts isolated from wildtype (WT), BRAF -/-, or CRAF -/- mice were irradiated (6 Gy) and DNA damage
Supplementary Materials for
 www.sciencesignaling.org/cgi/content/full/6/283/ra57/dc1 Supplementary Materials for JNK3 Couples the Neuronal Stress Response to Inhibition of Secretory Trafficking Guang Yang,* Xun Zhou, Jingyan Zhu,
www.sciencesignaling.org/cgi/content/full/6/283/ra57/dc1 Supplementary Materials for JNK3 Couples the Neuronal Stress Response to Inhibition of Secretory Trafficking Guang Yang,* Xun Zhou, Jingyan Zhu,
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION doi:1.138/nature9814 a A SHARPIN FL B SHARPIN ΔNZF C SHARPIN T38L, F39V b His-SHARPIN FL -1xUb -2xUb -4xUb α-his c Linear 4xUb -SHARPIN FL -SHARPIN TF_LV -SHARPINΔNZF -SHARPIN
SUPPLEMENTARY INFORMATION doi:1.138/nature9814 a A SHARPIN FL B SHARPIN ΔNZF C SHARPIN T38L, F39V b His-SHARPIN FL -1xUb -2xUb -4xUb α-his c Linear 4xUb -SHARPIN FL -SHARPIN TF_LV -SHARPINΔNZF -SHARPIN
(a) Significant biological processes (upper panel) and disease biomarkers (lower panel)
 Supplementary Figure 1. Functional enrichment analyses of secretomic proteins. (a) Significant biological processes (upper panel) and disease biomarkers (lower panel) 2 involved by hrab37-mediated secretory
Supplementary Figure 1. Functional enrichment analyses of secretomic proteins. (a) Significant biological processes (upper panel) and disease biomarkers (lower panel) 2 involved by hrab37-mediated secretory
Supplementary Figure 1 CD4 + T cells from PKC-θ null mice are defective in NF-κB activation during T cell receptor signaling. CD4 + T cells were
 Supplementary Figure 1 CD4 + T cells from PKC-θ null mice are defective in NF-κB activation during T cell receptor signaling. CD4 + T cells were isolated from wild type (PKC-θ- WT) or PKC-θ null (PKC-θ-KO)
Supplementary Figure 1 CD4 + T cells from PKC-θ null mice are defective in NF-κB activation during T cell receptor signaling. CD4 + T cells were isolated from wild type (PKC-θ- WT) or PKC-θ null (PKC-θ-KO)
Project report October 2012 March 2013 CRF fellow: Principal Investigator: Project title:
 Project report October 2012 March 2013 CRF fellow: Gennaro Napolitano Principal Investigator: Sergio Daniel Catz Project title: Small molecule regulators of vesicular trafficking to enhance lysosomal exocytosis
Project report October 2012 March 2013 CRF fellow: Gennaro Napolitano Principal Investigator: Sergio Daniel Catz Project title: Small molecule regulators of vesicular trafficking to enhance lysosomal exocytosis
T H E J O U R N A L O F C E L L B I O L O G Y
 T H E J O U R N A L O F C E L L B I O L O G Y Supplemental material Krenn et al., http://www.jcb.org/cgi/content/full/jcb.201110013/dc1 Figure S1. Levels of expressed proteins and demonstration that C-terminal
T H E J O U R N A L O F C E L L B I O L O G Y Supplemental material Krenn et al., http://www.jcb.org/cgi/content/full/jcb.201110013/dc1 Figure S1. Levels of expressed proteins and demonstration that C-terminal
Supplementary Figure 1. PD-L1 is glycosylated in cancer cells. (a) Western blot analysis of PD-L1 in breast cancer cells. (b) Western blot analysis
 Supplementary Figure 1. PD-L1 is glycosylated in cancer cells. (a) Western blot analysis of PD-L1 in breast cancer cells. (b) Western blot analysis of PD-L1 in ovarian cancer cells. (c) Western blot analysis
Supplementary Figure 1. PD-L1 is glycosylated in cancer cells. (a) Western blot analysis of PD-L1 in breast cancer cells. (b) Western blot analysis of PD-L1 in ovarian cancer cells. (c) Western blot analysis
DO NOT EXCEED FIVE PAGES.
 NAME: Vivek M. Rangnekar, PhD OMB No. 0925-0001 and 0925-0002 (Rev. 09/17 Approved Through 03/31/2020) BIOGRAPHICAL SKETCH Provide the following information for the Senior/key personnel and other significant
NAME: Vivek M. Rangnekar, PhD OMB No. 0925-0001 and 0925-0002 (Rev. 09/17 Approved Through 03/31/2020) BIOGRAPHICAL SKETCH Provide the following information for the Senior/key personnel and other significant
SUPPLEMENTARY FIGURE LEGENDS
 SUPPLEMENTARY FIGURE LEGENDS Supplemental FIG. 1. Localization of myosin Vb in cultured neurons varies with maturation stage. A and B, localization of myosin Vb in cultured hippocampal neurons. A, in DIV
SUPPLEMENTARY FIGURE LEGENDS Supplemental FIG. 1. Localization of myosin Vb in cultured neurons varies with maturation stage. A and B, localization of myosin Vb in cultured hippocampal neurons. A, in DIV
Supplemental Material:
 Supplemental Material: MATERIALS AND METHODS RNA interference Mouse CHOP sirna (ON-TARGETplus SMARTpool Cat# L-062068-00) and control sirna (ON-TARGETplus Control) were purchased from Dharmacon. Transfection
Supplemental Material: MATERIALS AND METHODS RNA interference Mouse CHOP sirna (ON-TARGETplus SMARTpool Cat# L-062068-00) and control sirna (ON-TARGETplus Control) were purchased from Dharmacon. Transfection
SUPPLEMENTARY INFORMATION
 doi:10.1038/nature12652 Supplementary Figure 1. PRDM16 interacts with endogenous EHMT1 in brown adipocytes. Immunoprecipitation of PRDM16 complex by flag antibody (M2) followed by Western blot analysis
doi:10.1038/nature12652 Supplementary Figure 1. PRDM16 interacts with endogenous EHMT1 in brown adipocytes. Immunoprecipitation of PRDM16 complex by flag antibody (M2) followed by Western blot analysis
Tumor suppressor Spred2 interaction with LC3 promotes autophagosome maturation and induces autophagy-dependent cell death
 www.impactjournals.com/oncotarget/ Oncotarget, Supplementary Materials 2016 Tumor suppressor Spred2 interaction with LC3 promotes autophagosome maturation and induces autophagy-dependent cell death Supplementary
www.impactjournals.com/oncotarget/ Oncotarget, Supplementary Materials 2016 Tumor suppressor Spred2 interaction with LC3 promotes autophagosome maturation and induces autophagy-dependent cell death Supplementary
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION doi:10.1038/nature11429 S1a 6 7 8 9 Nlrc4 allele S1b Nlrc4 +/+ Nlrc4 +/F Nlrc4 F/F 9 Targeting construct 422 bp 273 bp FRT-neo-gb-PGK-FRT 3x.STOP S1c Nlrc4 +/+ Nlrc4 F/F casp1
SUPPLEMENTARY INFORMATION doi:10.1038/nature11429 S1a 6 7 8 9 Nlrc4 allele S1b Nlrc4 +/+ Nlrc4 +/F Nlrc4 F/F 9 Targeting construct 422 bp 273 bp FRT-neo-gb-PGK-FRT 3x.STOP S1c Nlrc4 +/+ Nlrc4 F/F casp1
Figure S1. Sorting nexin 9 (SNX9) specifically binds psmad3 and not psmad 1/5/8. Lysates from AKR-2B cells untreated (-) or stimulated (+) for 45 min
 Figure S1. Sorting nexin 9 (SNX9) specifically binds psmad3 and not psmad 1/5/8. Lysates from AKR2B cells untreated () or stimulated () for 45 min with 5 ng/ml TGFβ or 10 ng/ml BMP4 were incubated with
Figure S1. Sorting nexin 9 (SNX9) specifically binds psmad3 and not psmad 1/5/8. Lysates from AKR2B cells untreated () or stimulated () for 45 min with 5 ng/ml TGFβ or 10 ng/ml BMP4 were incubated with
Part-4. Cell cycle regulatory protein 5 (Cdk5) A novel target of ERK in Carb induced cell death
 Part-4 Cell cycle regulatory protein 5 (Cdk5) A novel target of ERK in Carb induced cell death 95 1. Introduction The process of replicating DNA and dividing cells can be described as a series of coordinated
Part-4 Cell cycle regulatory protein 5 (Cdk5) A novel target of ERK in Carb induced cell death 95 1. Introduction The process of replicating DNA and dividing cells can be described as a series of coordinated
Nature Structural and Molecular Biology: doi: /nsmb Supplementary Figure 1
 Supplementary Figure 1 Mutational analysis of the SA2-Scc1 interaction in vitro and in human cells. (a) Autoradiograph (top) and Coomassie stained gel (bottom) of 35 S-labeled Myc-SA2 proteins (input)
Supplementary Figure 1 Mutational analysis of the SA2-Scc1 interaction in vitro and in human cells. (a) Autoradiograph (top) and Coomassie stained gel (bottom) of 35 S-labeled Myc-SA2 proteins (input)
SUPPLEMENTARY INFORMATION
 Supplementary Table 1. Cell sphingolipids and S1P bound to endogenous TRAF2. Sphingolipid Cell pmol/mg TRAF2 immunoprecipitate pmol/mg Sphingomyelin 4200 ± 250 Not detected Monohexosylceramide 311 ± 18
Supplementary Table 1. Cell sphingolipids and S1P bound to endogenous TRAF2. Sphingolipid Cell pmol/mg TRAF2 immunoprecipitate pmol/mg Sphingomyelin 4200 ± 250 Not detected Monohexosylceramide 311 ± 18
SUPPLEMENTARY INFORMATION
 Supplementary Figures Supplementary Figure S1. Binding of full-length OGT and deletion mutants to PIP strips (Echelon Biosciences). Supplementary Figure S2. Binding of the OGT (919-1036) fragments with
Supplementary Figures Supplementary Figure S1. Binding of full-length OGT and deletion mutants to PIP strips (Echelon Biosciences). Supplementary Figure S2. Binding of the OGT (919-1036) fragments with
Supplementary Figure 1
 Supplementary Figure 1 Supplementary Figure 1. Neither the activation nor suppression of the MAPK pathway affects the ASK1/Vif interaction. (a, b) HEK293 cells were cotransfected with plasmids encoding
Supplementary Figure 1 Supplementary Figure 1. Neither the activation nor suppression of the MAPK pathway affects the ASK1/Vif interaction. (a, b) HEK293 cells were cotransfected with plasmids encoding
Supplementary Figure S1
 Supplementary Figure S1 Supplementary Figure S1. PARP localization patterns using GFP-PARP and PARP-specific antibody libraries GFP-PARP localization in non-fixed (A) and formaldehyde fixed (B) GFP-PARPx
Supplementary Figure S1 Supplementary Figure S1. PARP localization patterns using GFP-PARP and PARP-specific antibody libraries GFP-PARP localization in non-fixed (A) and formaldehyde fixed (B) GFP-PARPx
Supplementary Figure 1
 Supplementary Figure 1 a γ-h2ax MDC1 RNF8 FK2 BRCA1 U2OS Cells sgrna-1 ** 60 sgrna 40 20 0 % positive Cells (>5 foci per cell) b ** 80 sgrna sgrna γ-h2ax MDC1 γ-h2ax RNF8 FK2 MDC1 BRCA1 RNF8 FK2 BRCA1
Supplementary Figure 1 a γ-h2ax MDC1 RNF8 FK2 BRCA1 U2OS Cells sgrna-1 ** 60 sgrna 40 20 0 % positive Cells (>5 foci per cell) b ** 80 sgrna sgrna γ-h2ax MDC1 γ-h2ax RNF8 FK2 MDC1 BRCA1 RNF8 FK2 BRCA1
(a) Schematic diagram of the FS mutation of UVRAG in exon 8 containing the highly instable
 Supplementary Figure 1. Frameshift (FS) mutation in UVRAG. (a) Schematic diagram of the FS mutation of UVRAG in exon 8 containing the highly instable A 10 DNA repeat, generating a premature stop codon
Supplementary Figure 1. Frameshift (FS) mutation in UVRAG. (a) Schematic diagram of the FS mutation of UVRAG in exon 8 containing the highly instable A 10 DNA repeat, generating a premature stop codon
SUPPLEMENTARY INFORMATION
 DOI:.38/ncb2822 a MTC02 FAO cells EEA1 b +/+ MEFs /DAPI -/- MEFs /DAPI -/- MEFs //DAPI c HEK 293 cells WCE N M C P AKT TBC1D7 Lamin A/C EEA1 VDAC d HeLa cells WCE N M C P AKT Lamin A/C EEA1 VDAC Figure
DOI:.38/ncb2822 a MTC02 FAO cells EEA1 b +/+ MEFs /DAPI -/- MEFs /DAPI -/- MEFs //DAPI c HEK 293 cells WCE N M C P AKT TBC1D7 Lamin A/C EEA1 VDAC d HeLa cells WCE N M C P AKT Lamin A/C EEA1 VDAC Figure
Supplemental Figure 1. Western blot analysis indicated that MIF was detected in the fractions of
 Supplemental Figure Legends Supplemental Figure 1. Western blot analysis indicated that was detected in the fractions of plasma membrane and cytosol but not in nuclear fraction isolated from Pkd1 null
Supplemental Figure Legends Supplemental Figure 1. Western blot analysis indicated that was detected in the fractions of plasma membrane and cytosol but not in nuclear fraction isolated from Pkd1 null
(A) SW480, DLD1, RKO and HCT116 cells were treated with DMSO or XAV939 (5 µm)
 Supplementary Figure Legends Figure S1. Tankyrase inhibition suppresses cell proliferation in an axin/β-catenin independent manner. (A) SW480, DLD1, RKO and HCT116 cells were treated with DMSO or XAV939
Supplementary Figure Legends Figure S1. Tankyrase inhibition suppresses cell proliferation in an axin/β-catenin independent manner. (A) SW480, DLD1, RKO and HCT116 cells were treated with DMSO or XAV939
a b G75 G60 Sw-2 Sw-1 Supplementary Figure 1. Structure predictions by I-TASSER Server.
 a b G75 2 2 G60 Sw-2 Sw-1 Supplementary Figure 1. Structure predictions by I-TASSER Server. a. Overlay of top 10 models generated by I-TASSER illustrates the potential effect of 7 amino acid insertion
a b G75 2 2 G60 Sw-2 Sw-1 Supplementary Figure 1. Structure predictions by I-TASSER Server. a. Overlay of top 10 models generated by I-TASSER illustrates the potential effect of 7 amino acid insertion
SUPPLEMENTARY INFORMATION
 DOI: 10.1038/ncb2566 Figure S1 CDKL5 protein expression pattern and localization in mouse brain. (a) Multiple-tissue western blot from a postnatal day (P) 21 mouse probed with an antibody against CDKL5.
DOI: 10.1038/ncb2566 Figure S1 CDKL5 protein expression pattern and localization in mouse brain. (a) Multiple-tissue western blot from a postnatal day (P) 21 mouse probed with an antibody against CDKL5.
supplementary information
 DOI: 10.1038/ncb1875 Figure S1 (a) The 79 surgical specimens from NSCLC patients were analysed by immunohistochemistry with an anti-p53 antibody and control serum (data not shown). The normal bronchi served
DOI: 10.1038/ncb1875 Figure S1 (a) The 79 surgical specimens from NSCLC patients were analysed by immunohistochemistry with an anti-p53 antibody and control serum (data not shown). The normal bronchi served
Antibodies for Unfolded Protein Response
 Novus-lu-2945 Antibodies for Unfolded rotein Response Unfolded roteins ER lumen GR78 IRE-1 GR78 ERK Cytosol GR78 TRAF2 ASK1 JNK Activator Intron RIDD elf2α Degraded mrna XB1 mrna Translation XB1-S (p50)
Novus-lu-2945 Antibodies for Unfolded rotein Response Unfolded roteins ER lumen GR78 IRE-1 GR78 ERK Cytosol GR78 TRAF2 ASK1 JNK Activator Intron RIDD elf2α Degraded mrna XB1 mrna Translation XB1-S (p50)
Par-4 Binds to Topoisomerase 1 and Attenuates Its DNA Relaxation Activity
 Research Article Par-4 Binds to Topoisomerase 1 and Attenuates Its DNA Relaxation Activity Anindya Goswami, 1 Shirley Qiu, 1 Thomas S. Dexheimer, 3 Padhma Ranganathan, 2 Ravshan Burikhanov, 1 Yves Pommier,
Research Article Par-4 Binds to Topoisomerase 1 and Attenuates Its DNA Relaxation Activity Anindya Goswami, 1 Shirley Qiu, 1 Thomas S. Dexheimer, 3 Padhma Ranganathan, 2 Ravshan Burikhanov, 1 Yves Pommier,
Supplemental Figures:
 Supplemental Figures: Figure 1: Intracellular distribution of VWF by electron microscopy in human endothelial cells. a) Immunogold labeling of LC3 demonstrating an LC3-positive autophagosome (white arrow)
Supplemental Figures: Figure 1: Intracellular distribution of VWF by electron microscopy in human endothelial cells. a) Immunogold labeling of LC3 demonstrating an LC3-positive autophagosome (white arrow)
A Hepatocyte Growth Factor Receptor (Met) Insulin Receptor hybrid governs hepatic glucose metabolism SUPPLEMENTARY FIGURES, LEGENDS AND METHODS
 A Hepatocyte Growth Factor Receptor (Met) Insulin Receptor hybrid governs hepatic glucose metabolism Arlee Fafalios, Jihong Ma, Xinping Tan, John Stoops, Jianhua Luo, Marie C. DeFrances and Reza Zarnegar
A Hepatocyte Growth Factor Receptor (Met) Insulin Receptor hybrid governs hepatic glucose metabolism Arlee Fafalios, Jihong Ma, Xinping Tan, John Stoops, Jianhua Luo, Marie C. DeFrances and Reza Zarnegar
Myelin suppresses axon regeneration by PIR-B/SHPmediated inhibition of Trk activity
 Manuscript EMBO-2010-76298 Myelin suppresses axon regeneration by PIR-B/SHPmediated inhibition of Trk activity Yuki Fujita, Shota Endo, Toshiyuki Takai and Toshihide Yamashita Corresponding author: Toshihide
Manuscript EMBO-2010-76298 Myelin suppresses axon regeneration by PIR-B/SHPmediated inhibition of Trk activity Yuki Fujita, Shota Endo, Toshiyuki Takai and Toshihide Yamashita Corresponding author: Toshihide
Supplementary Information Supplementary Fig. 1. Elevated Usp9x in melanoma and NRAS mutant melanoma cells are dependent on NRAS for 3D growth.
 Supplementary Information Supplementary Fig. 1. Elevated Usp9x in melanoma and NRAS mutant melanoma cells are dependent on NRAS for 3D growth. a. Immunoblot for Usp9x protein in NRAS mutant melanoma cells
Supplementary Information Supplementary Fig. 1. Elevated Usp9x in melanoma and NRAS mutant melanoma cells are dependent on NRAS for 3D growth. a. Immunoblot for Usp9x protein in NRAS mutant melanoma cells
Introduction: 年 Fas signal-mediated apoptosis. PI3K/Akt
 Fas-ligand (CD95-L; Fas-L) Fas (CD95) Fas (apoptosis) 年 了 不 度 Fas Fas-L 力 不 Fas/Fas-L T IL-10Fas/Fas-L 不 年 Fas signal-mediated apoptosis 度降 不 不 力 U-118, HeLa, A549, Huh-7 MCF-7, HepG2. PI3K/Akt FasPI3K/Akt
Fas-ligand (CD95-L; Fas-L) Fas (CD95) Fas (apoptosis) 年 了 不 度 Fas Fas-L 力 不 Fas/Fas-L T IL-10Fas/Fas-L 不 年 Fas signal-mediated apoptosis 度降 不 不 力 U-118, HeLa, A549, Huh-7 MCF-7, HepG2. PI3K/Akt FasPI3K/Akt
Supplementary Information
 Supplementary Information Targeted Disruption of the EZH2/EED Complex Inhibits EZH2- dependent Cancer Woojin Kim 1,2,3, Gregory H. Bird 2,3,4, Tobias Neff 5, Guoji Guo 1,2,3, Marc A. Kerenyi 1,2,3, Loren
Supplementary Information Targeted Disruption of the EZH2/EED Complex Inhibits EZH2- dependent Cancer Woojin Kim 1,2,3, Gregory H. Bird 2,3,4, Tobias Neff 5, Guoji Guo 1,2,3, Marc A. Kerenyi 1,2,3, Loren
(A) RT-PCR for components of the Shh/Gli pathway in normal fetus cell (MRC-5) and a
 Supplementary figure legends Supplementary Figure 1. Expression of Shh signaling components in a panel of gastric cancer. (A) RT-PCR for components of the Shh/Gli pathway in normal fetus cell (MRC-5) and
Supplementary figure legends Supplementary Figure 1. Expression of Shh signaling components in a panel of gastric cancer. (A) RT-PCR for components of the Shh/Gli pathway in normal fetus cell (MRC-5) and
SUPPLEMENTARY INFORMATION
 Figure S1 Treatment with both Sema6D and Plexin-A1 sirnas induces the phenotype essentially identical to that induced by treatment with Sema6D sirna alone or Plexin-A1 sirna alone. (a,b) The cardiac tube
Figure S1 Treatment with both Sema6D and Plexin-A1 sirnas induces the phenotype essentially identical to that induced by treatment with Sema6D sirna alone or Plexin-A1 sirna alone. (a,b) The cardiac tube
William C. Comb, Jessica E. Hutti, Patricia Cogswell, Lewis C. Cantley, and Albert S. Baldwin
 Molecular Cell, Volume 45 Supplemental Information p85 SH2 Domain Phosphorylation by IKK Promotes Feedback Inhibition of PI3K and Akt in Response to Cellular Starvation William C. Comb, Jessica E. Hutti,
Molecular Cell, Volume 45 Supplemental Information p85 SH2 Domain Phosphorylation by IKK Promotes Feedback Inhibition of PI3K and Akt in Response to Cellular Starvation William C. Comb, Jessica E. Hutti,
The clathrin adaptor Numb regulates intestinal cholesterol. absorption through dynamic interaction with NPC1L1
 The clathrin adaptor Numb regulates intestinal cholesterol absorption through dynamic interaction with NPC1L1 Pei-Shan Li 1, Zhen-Yan Fu 1,2, Ying-Yu Zhang 1, Jin-Hui Zhang 1, Chen-Qi Xu 1, Yi-Tong Ma
The clathrin adaptor Numb regulates intestinal cholesterol absorption through dynamic interaction with NPC1L1 Pei-Shan Li 1, Zhen-Yan Fu 1,2, Ying-Yu Zhang 1, Jin-Hui Zhang 1, Chen-Qi Xu 1, Yi-Tong Ma
Supplementary Materials
 Supplementary Materials Figure S1. MTT Cell viability assay. To measure the cytotoxic potential of the oxidative treatment, the MTT [3-(4,5-dimethylthiazol- 2-yl)-2,5-diphenyl tetrazolium bromide] assay
Supplementary Materials Figure S1. MTT Cell viability assay. To measure the cytotoxic potential of the oxidative treatment, the MTT [3-(4,5-dimethylthiazol- 2-yl)-2,5-diphenyl tetrazolium bromide] assay
C-Phycocyanin (C-PC) is a n«sjfc&c- waefc-jduble phycobiliprotein. pigment isolated from Spirulina platensis. This water- soluble protein pigment is
 ' ^Summary C-Phycocyanin (C-PC) is a n«sjfc&c- waefc-jduble phycobiliprotein pigment isolated from Spirulina platensis. This water- soluble protein pigment is of greater importance because of its various
' ^Summary C-Phycocyanin (C-PC) is a n«sjfc&c- waefc-jduble phycobiliprotein pigment isolated from Spirulina platensis. This water- soluble protein pigment is of greater importance because of its various
Soft Agar Assay. For each cell pool, 100,000 cells were resuspended in 0.35% (w/v)
 SUPPLEMENTARY MATERIAL AND METHODS Soft Agar Assay. For each cell pool, 100,000 cells were resuspended in 0.35% (w/v) top agar (LONZA, SeaKem LE Agarose cat.5004) and plated onto 0.5% (w/v) basal agar.
SUPPLEMENTARY MATERIAL AND METHODS Soft Agar Assay. For each cell pool, 100,000 cells were resuspended in 0.35% (w/v) top agar (LONZA, SeaKem LE Agarose cat.5004) and plated onto 0.5% (w/v) basal agar.
Expanded View Figures
 Shao-Ming Shen et al Role of I in MT of cancers MO reports xpanded View igures igure V1. nalysis of the expression of I isoforms in cancer cells and their interaction with PTN. RT PR detection of Ish and
Shao-Ming Shen et al Role of I in MT of cancers MO reports xpanded View igures igure V1. nalysis of the expression of I isoforms in cancer cells and their interaction with PTN. RT PR detection of Ish and
SUPPLEMENTARY INFORMATION. Supplementary Figures S1-S9. Supplementary Methods
 SUPPLEMENTARY INFORMATION SUMO1 modification of PTEN regulates tumorigenesis by controlling its association with the plasma membrane Jian Huang 1,2#, Jie Yan 1,2#, Jian Zhang 3#, Shiguo Zhu 1, Yanli Wang
SUPPLEMENTARY INFORMATION SUMO1 modification of PTEN regulates tumorigenesis by controlling its association with the plasma membrane Jian Huang 1,2#, Jie Yan 1,2#, Jian Zhang 3#, Shiguo Zhu 1, Yanli Wang
Supplementary Figures
 Supplementary Figures Figure S1. Validation of kinase regulators of ONC201 sensitivity. Validation and screen results for changes in cell viability associated with the combination of ONC201 treatment (1
Supplementary Figures Figure S1. Validation of kinase regulators of ONC201 sensitivity. Validation and screen results for changes in cell viability associated with the combination of ONC201 treatment (1
RAW264.7 cells stably expressing control shrna (Con) or GSK3b-specific shrna (sh-
 1 a b Supplementary Figure 1. Effects of GSK3b knockdown on poly I:C-induced cytokine production. RAW264.7 cells stably expressing control shrna (Con) or GSK3b-specific shrna (sh- GSK3b) were stimulated
1 a b Supplementary Figure 1. Effects of GSK3b knockdown on poly I:C-induced cytokine production. RAW264.7 cells stably expressing control shrna (Con) or GSK3b-specific shrna (sh- GSK3b) were stimulated
Supplementary Information POLO-LIKE KINASE 1 FACILITATES LOSS OF PTEN-INDUCED PROSTATE CANCER FORMATION
 Supplementary Information POLO-LIKE KINASE 1 FACILITATES LOSS OF PTEN-INDUCED PROSTATE CANCER FORMATION X. Shawn Liu 1, 3, Bing Song 2, 3, Bennett D. Elzey 3, 4, Timothy L. Ratliff 3, 4, Stephen F. Konieczny
Supplementary Information POLO-LIKE KINASE 1 FACILITATES LOSS OF PTEN-INDUCED PROSTATE CANCER FORMATION X. Shawn Liu 1, 3, Bing Song 2, 3, Bennett D. Elzey 3, 4, Timothy L. Ratliff 3, 4, Stephen F. Konieczny
SUPPLEMENTARY INFORMATION
 DOI: 10.1038/ncb3076 Supplementary Figure 1 btrcp targets Cep68 for degradation during mitosis. a) Cep68 immunofluorescence in interphase and metaphase. U-2OS cells were transfected with control sirna
DOI: 10.1038/ncb3076 Supplementary Figure 1 btrcp targets Cep68 for degradation during mitosis. a) Cep68 immunofluorescence in interphase and metaphase. U-2OS cells were transfected with control sirna
Cell Biology Lecture 9 Notes Basic Principles of cell signaling and GPCR system
 Cell Biology Lecture 9 Notes Basic Principles of cell signaling and GPCR system Basic Elements of cell signaling: Signal or signaling molecule (ligand, first messenger) o Small molecules (epinephrine,
Cell Biology Lecture 9 Notes Basic Principles of cell signaling and GPCR system Basic Elements of cell signaling: Signal or signaling molecule (ligand, first messenger) o Small molecules (epinephrine,
nature methods Organelle-specific, rapid induction of molecular activities and membrane tethering
 nature methods Organelle-specific, rapid induction of molecular activities and membrane tethering Toru Komatsu, Igor Kukelyansky, J Michael McCaffery, Tasuku Ueno, Lidenys C Varela & Takanari Inoue Supplementary
nature methods Organelle-specific, rapid induction of molecular activities and membrane tethering Toru Komatsu, Igor Kukelyansky, J Michael McCaffery, Tasuku Ueno, Lidenys C Varela & Takanari Inoue Supplementary
Live cell imaging of trafficking of the chaperone complex vaccine to the ER. BMDCs were incubated with ER-Tracker Red (1 M) in staining solution for
 Live cell imaging of trafficking of the chaperone complex vaccine to the ER. BMDCs were incubated with ER-Tracker Red (1 M) in staining solution for 15 min at 37 C and replaced with fresh complete medium.
Live cell imaging of trafficking of the chaperone complex vaccine to the ER. BMDCs were incubated with ER-Tracker Red (1 M) in staining solution for 15 min at 37 C and replaced with fresh complete medium.
Supporting Information. FADD regulates NF-кB activation and promotes ubiquitination of cflip L to induce. apoptosis
 1 2 Supporting Information 3 4 5 FADD regulates NF-кB activation and promotes ubiquitination of cflip L to induce apoptosis 6 7 Kishu Ranjan and Chandramani Pathak* 8 9 Department of Cell Biology, School
1 2 Supporting Information 3 4 5 FADD regulates NF-кB activation and promotes ubiquitination of cflip L to induce apoptosis 6 7 Kishu Ranjan and Chandramani Pathak* 8 9 Department of Cell Biology, School
SD-1 SD-1: Cathepsin B levels in TNF treated hch
 SD-1 SD-1: Cathepsin B levels in TNF treated hch. A. RNA and B. protein extracts from TNF treated and untreated human chondrocytes (hch) were analyzed via qpcr (left) and immunoblot analyses (right) for
SD-1 SD-1: Cathepsin B levels in TNF treated hch. A. RNA and B. protein extracts from TNF treated and untreated human chondrocytes (hch) were analyzed via qpcr (left) and immunoblot analyses (right) for
Tel: ; Fax: ;
 Tel.: +98 216 696 9291; Fax: +98 216 696 9291; E-mail: mrasadeghi@pasteur.ac.ir Tel: +98 916 113 7679; Fax: +98 613 333 6380; E-mail: abakhshi_e@ajums.ac.ir A Soluble Chromatin-bound MOI 0 1 5 0 1 5 HDAC2
Tel.: +98 216 696 9291; Fax: +98 216 696 9291; E-mail: mrasadeghi@pasteur.ac.ir Tel: +98 916 113 7679; Fax: +98 613 333 6380; E-mail: abakhshi_e@ajums.ac.ir A Soluble Chromatin-bound MOI 0 1 5 0 1 5 HDAC2
Supplementary Figure S1. Venn diagram analysis of mrna microarray data and mirna target analysis. (a) Western blot analysis of T lymphoblasts (CLS)
 Supplementary Figure S1. Venn diagram analysis of mrna microarray data and mirna target analysis. (a) Western blot analysis of T lymphoblasts (CLS) and their exosomes (EXO) in resting (REST) and activated
Supplementary Figure S1. Venn diagram analysis of mrna microarray data and mirna target analysis. (a) Western blot analysis of T lymphoblasts (CLS) and their exosomes (EXO) in resting (REST) and activated
The functional investigation of the interaction between TATA-associated factor 3 (TAF3) and p53 protein
 THESIS BOOK The functional investigation of the interaction between TATA-associated factor 3 (TAF3) and p53 protein Orsolya Buzás-Bereczki Supervisors: Dr. Éva Bálint Dr. Imre Miklós Boros University of
THESIS BOOK The functional investigation of the interaction between TATA-associated factor 3 (TAF3) and p53 protein Orsolya Buzás-Bereczki Supervisors: Dr. Éva Bálint Dr. Imre Miklós Boros University of
Cyclin I activates Cdk5 and regulates expression of Bcl-2 and Bcl-XL in postmitotic mouse cells
 Research article Cyclin I activates Cdk5 and regulates expression of Bcl-2 and Bcl-XL in postmitotic mouse cells Paul T. Brinkkoetter, 1 Paul Olivier, 2 Jimmy S. Wu, 1 Scott Henderson, 1 Ronald D. Krofft,
Research article Cyclin I activates Cdk5 and regulates expression of Bcl-2 and Bcl-XL in postmitotic mouse cells Paul T. Brinkkoetter, 1 Paul Olivier, 2 Jimmy S. Wu, 1 Scott Henderson, 1 Ronald D. Krofft,
Rapid parallel measurements of macroautophagy and mitophagy in
 Supplemental Figures Rapid parallel measurements of macroautophagy and mitophagy in mammalian cells using a single fluorescent biosensor Sargsyan A, Cai J, Fandino LB, Labasky ME, Forostyan T, Colosimo
Supplemental Figures Rapid parallel measurements of macroautophagy and mitophagy in mammalian cells using a single fluorescent biosensor Sargsyan A, Cai J, Fandino LB, Labasky ME, Forostyan T, Colosimo
S1a S1b S1c. S1d. S1f S1g S1h SUPPLEMENTARY FIGURE 1. - si sc Il17rd Il17ra bp. rig/s IL-17RD (ng) -100 IL-17RD
 SUPPLEMENTARY FIGURE 1 0 20 50 80 100 IL-17RD (ng) S1a S1b S1c IL-17RD β-actin kda S1d - si sc Il17rd Il17ra rig/s15-574 - 458-361 bp S1f S1g S1h S1i S1j Supplementary Figure 1. Knockdown of IL-17RD enhances
SUPPLEMENTARY FIGURE 1 0 20 50 80 100 IL-17RD (ng) S1a S1b S1c IL-17RD β-actin kda S1d - si sc Il17rd Il17ra rig/s15-574 - 458-361 bp S1f S1g S1h S1i S1j Supplementary Figure 1. Knockdown of IL-17RD enhances
Supplementary Materials for
 www.sciencesignaling.org/cgi/content/full/10/471/eaah5085/dc1 Supplementary Materials for Phosphorylation of the exocyst protein Exo84 by TBK1 promotes insulin-stimulated GLUT4 trafficking Maeran Uhm,
www.sciencesignaling.org/cgi/content/full/10/471/eaah5085/dc1 Supplementary Materials for Phosphorylation of the exocyst protein Exo84 by TBK1 promotes insulin-stimulated GLUT4 trafficking Maeran Uhm,
Supplemental Materials. STK16 regulates actin dynamics to control Golgi organization and cell cycle
 Supplemental Materials STK16 regulates actin dynamics to control Golgi organization and cell cycle Juanjuan Liu 1,2,3, Xingxing Yang 1,3, Binhua Li 1, Junjun Wang 1,2, Wenchao Wang 1, Jing Liu 1, Qingsong
Supplemental Materials STK16 regulates actin dynamics to control Golgi organization and cell cycle Juanjuan Liu 1,2,3, Xingxing Yang 1,3, Binhua Li 1, Junjun Wang 1,2, Wenchao Wang 1, Jing Liu 1, Qingsong
Supplementary Figures
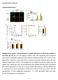 Supplementary Figures Supplementary Figure 1 Characterization of stable expression of GlucB and sshbira in the CT26 cell line (a) Live cell imaging of stable CT26 cells expressing green fluorescent protein
Supplementary Figures Supplementary Figure 1 Characterization of stable expression of GlucB and sshbira in the CT26 cell line (a) Live cell imaging of stable CT26 cells expressing green fluorescent protein
A. List of selected proteins with high SILAC (H/L) ratios identified in mass
 Supplementary material Figure S1. Interaction between UBL5 and FANCI A. List of selected proteins with high SILAC (H/L) ratios identified in mass spectrometry (MS)-based analysis of UBL5-interacting proteins,
Supplementary material Figure S1. Interaction between UBL5 and FANCI A. List of selected proteins with high SILAC (H/L) ratios identified in mass spectrometry (MS)-based analysis of UBL5-interacting proteins,
p = formed with HCI-001 p = Relative # of blood vessels that formed with HCI-002 Control Bevacizumab + 17AAG Bevacizumab 17AAG
 A.. Relative # of ECs associated with HCI-001 1.4 1.2 1.0 0.8 0.6 0.4 0.2 0.0 ol b p < 0.001 Relative # of blood vessels that formed with HCI-001 1.4 1.2 1.0 0.8 0.6 0.4 0.2 0.0 l b p = 0.002 Control IHC:
A.. Relative # of ECs associated with HCI-001 1.4 1.2 1.0 0.8 0.6 0.4 0.2 0.0 ol b p < 0.001 Relative # of blood vessels that formed with HCI-001 1.4 1.2 1.0 0.8 0.6 0.4 0.2 0.0 l b p = 0.002 Control IHC:
Supplementary Figure 1. Confocal immunofluorescence showing mitochondrial translocation of Drp1. Cardiomyocytes treated with H 2 O 2 were prestained
 Supplementary Figure 1. Confocal immunofluorescence showing mitochondrial translocation of Drp1. Cardiomyocytes treated with H 2 O 2 were prestained with MitoTracker (red), then were immunostained with
Supplementary Figure 1. Confocal immunofluorescence showing mitochondrial translocation of Drp1. Cardiomyocytes treated with H 2 O 2 were prestained with MitoTracker (red), then were immunostained with
Supplementary Figure 1. PAQR3 knockdown inhibits SREBP-2 processing in CHO-7 cells CHO-7 cells were transfected with control sirna or a sirna
 Supplementary Figure 1. PAQR3 knockdown inhibits SREBP-2 processing in CHO-7 cells CHO-7 cells were transfected with control sirna or a sirna targeted for hamster PAQR3. At 24 h after the transfection,
Supplementary Figure 1. PAQR3 knockdown inhibits SREBP-2 processing in CHO-7 cells CHO-7 cells were transfected with control sirna or a sirna targeted for hamster PAQR3. At 24 h after the transfection,
Supplementary Fig. 1. GPRC5A post-transcriptionally down-regulates EGFR expression. (a) Plot of the changes in steady state mrna levels versus
 Supplementary Fig. 1. GPRC5A post-transcriptionally down-regulates EGFR expression. (a) Plot of the changes in steady state mrna levels versus changes in corresponding proteins between wild type and Gprc5a-/-
Supplementary Fig. 1. GPRC5A post-transcriptionally down-regulates EGFR expression. (a) Plot of the changes in steady state mrna levels versus changes in corresponding proteins between wild type and Gprc5a-/-
File Name: Supplementary Information Description: Supplementary Figures and Supplementary Tables. File Name: Peer Review File Description:
 File Name: Supplementary Information Description: Supplementary Figures and Supplementary Tables File Name: Peer Review File Description: Primer Name Sequence (5'-3') AT ( C) RT-PCR USP21 F 5'-TTCCCATGGCTCCTTCCACATGAT-3'
File Name: Supplementary Information Description: Supplementary Figures and Supplementary Tables File Name: Peer Review File Description: Primer Name Sequence (5'-3') AT ( C) RT-PCR USP21 F 5'-TTCCCATGGCTCCTTCCACATGAT-3'
Intracellular MHC class II molecules promote TLR-triggered innate. immune responses by maintaining Btk activation
 Intracellular MHC class II molecules promote TLR-triggered innate immune responses by maintaining Btk activation Xingguang Liu, Zhenzhen Zhan, Dong Li, Li Xu, Feng Ma, Peng Zhang, Hangping Yao and Xuetao
Intracellular MHC class II molecules promote TLR-triggered innate immune responses by maintaining Btk activation Xingguang Liu, Zhenzhen Zhan, Dong Li, Li Xu, Feng Ma, Peng Zhang, Hangping Yao and Xuetao
Supplemental Information. NRF2 Is a Major Target of ARF. in p53-independent Tumor Suppression
 Molecular Cell, Volume 68 Supplemental Information NRF2 Is a Major Target of ARF in p53-independent Tumor Suppression Delin Chen, Omid Tavana, Bo Chu, Luke Erber, Yue Chen, Richard Baer, and Wei Gu Figure
Molecular Cell, Volume 68 Supplemental Information NRF2 Is a Major Target of ARF in p53-independent Tumor Suppression Delin Chen, Omid Tavana, Bo Chu, Luke Erber, Yue Chen, Richard Baer, and Wei Gu Figure
SUPPLEMENTARY FIGURES AND TABLE
 SUPPLEMENTARY FIGURES AND TABLE Supplementary Figure S1: Characterization of IRE1α mutants. A. U87-LUC cells were transduced with the lentiviral vector containing the GFP sequence (U87-LUC Tet-ON GFP).
SUPPLEMENTARY FIGURES AND TABLE Supplementary Figure S1: Characterization of IRE1α mutants. A. U87-LUC cells were transduced with the lentiviral vector containing the GFP sequence (U87-LUC Tet-ON GFP).
Downregulation of the small GTPase SAR1A: a key event underlying alcohol-induced Golgi fragmentation in hepatocytes
 Downregulation of the small GTPase SAR1A: a key event underlying alcohol-induced Golgi fragmentation in hepatocytes Armen Petrosyan 1*, Pi-Wan Cheng 1,3, Dahn L. Clemens 2,3 & Carol A. Casey 2,3 1 Department
Downregulation of the small GTPase SAR1A: a key event underlying alcohol-induced Golgi fragmentation in hepatocytes Armen Petrosyan 1*, Pi-Wan Cheng 1,3, Dahn L. Clemens 2,3 & Carol A. Casey 2,3 1 Department
crossmark Ca V subunits interact with the voltage-gated calcium channel
 crossmark THE JOURNAL OF BIOLOGICAL CHEMISTRY VOL. 291, NO. 39, pp. 20402 20416, September 23, 2016 Author s Choice 2016 by The American Society for Biochemistry and Molecular Biology, Inc. Published in
crossmark THE JOURNAL OF BIOLOGICAL CHEMISTRY VOL. 291, NO. 39, pp. 20402 20416, September 23, 2016 Author s Choice 2016 by The American Society for Biochemistry and Molecular Biology, Inc. Published in
(A) Dose response curves of HMLE_shGFP (blue circle), HMLE_shEcad (red square),
 Supplementary Figures and Tables Figure S1. Validation of EMT-selective small molecules (A) Dose response curves of HMLE_shGFP (blue circle), HMLE_shEcad (red square), and HMLE_Twist (black diamond) cells
Supplementary Figures and Tables Figure S1. Validation of EMT-selective small molecules (A) Dose response curves of HMLE_shGFP (blue circle), HMLE_shEcad (red square), and HMLE_Twist (black diamond) cells
Supplements. Figure S1. B Phalloidin Alexa488
 Supplements A, DMSO, PP2, PP3 Crk-myc Figure S1. (A) Src kinase activity is necessary for recruitment of Crk to Nephrin cytoplasmic domain. Human podocytes expressing /7-NephrinCD () were treated with
Supplements A, DMSO, PP2, PP3 Crk-myc Figure S1. (A) Src kinase activity is necessary for recruitment of Crk to Nephrin cytoplasmic domain. Human podocytes expressing /7-NephrinCD () were treated with
Li et al. Journal of Experimental & Clinical Cancer Research (2018) 37:108
 Li et al. Journal of Experimental & Clinical Cancer Research (2018) 37:108 https://doi.org/10.1186/s13046-018-0774-7 CORRECTION Correction to: Novel smac mimetic APG- 1387 elicits ovarian cancer cell killing
Li et al. Journal of Experimental & Clinical Cancer Research (2018) 37:108 https://doi.org/10.1186/s13046-018-0774-7 CORRECTION Correction to: Novel smac mimetic APG- 1387 elicits ovarian cancer cell killing
Supplementary Materials for
 www.sciencesignaling.org/cgi/content/full/6/305/ra106/dc1 Supplementary Materials for Controlling Long-Term Signaling: Receptor Dynamics Determine Attenuation and Refractory Behavior of the TGF-β Pathway
www.sciencesignaling.org/cgi/content/full/6/305/ra106/dc1 Supplementary Materials for Controlling Long-Term Signaling: Receptor Dynamics Determine Attenuation and Refractory Behavior of the TGF-β Pathway
Supplemental Information
 Supplemental Information Tobacco-specific Carcinogen Induces DNA Methyltransferases 1 Accumulation through AKT/GSK3β/βTrCP/hnRNP-U in Mice and Lung Cancer patients Ruo-Kai Lin, 1 Yi-Shuan Hsieh, 2 Pinpin
Supplemental Information Tobacco-specific Carcinogen Induces DNA Methyltransferases 1 Accumulation through AKT/GSK3β/βTrCP/hnRNP-U in Mice and Lung Cancer patients Ruo-Kai Lin, 1 Yi-Shuan Hsieh, 2 Pinpin
Nature Immunology: doi: /ni.3866
 Nature Immunology: doi:10.1038/ni.3866 Supplementary Figure 1 The effect of TIPE2 on chemotaxis. a, The expression of TIPE2 in dhl-60c, dhl-60t, TIPE2-expressing and 15/16Q-expressing dhl-60t neutrophils
Nature Immunology: doi:10.1038/ni.3866 Supplementary Figure 1 The effect of TIPE2 on chemotaxis. a, The expression of TIPE2 in dhl-60c, dhl-60t, TIPE2-expressing and 15/16Q-expressing dhl-60t neutrophils
Supplemental information contains 7 movies and 4 supplemental Figures
 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 Supplemental information contains 7 movies and 4 supplemental Figures Movies: Movie 1. Single virus tracking of A4-mCherry-WR MV
1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 Supplemental information contains 7 movies and 4 supplemental Figures Movies: Movie 1. Single virus tracking of A4-mCherry-WR MV
Nature Immunology: doi: /ni.3631
 Supplementary Figure 1 SPT analyses of Zap70 at the T cell plasma membrane. (a) Total internal reflection fluorescent (TIRF) excitation at 64-68 degrees limits single molecule detection to 100-150 nm above
Supplementary Figure 1 SPT analyses of Zap70 at the T cell plasma membrane. (a) Total internal reflection fluorescent (TIRF) excitation at 64-68 degrees limits single molecule detection to 100-150 nm above
Supplementary Figure 1
 Supplementary Figure 1 Constitutive EGFR signaling does not activate canonical EGFR signals (a) U251EGFRInd cells with or without tetracycline exposure (24h, 1µg/ml) were treated with EGF for 15 minutes
Supplementary Figure 1 Constitutive EGFR signaling does not activate canonical EGFR signals (a) U251EGFRInd cells with or without tetracycline exposure (24h, 1µg/ml) were treated with EGF for 15 minutes
FGL2 A new biomarker for cancer in a simple blood test
 FGL2 A new biomarker for cancer in a simple blood test WHO IS FGL2 Human gene (chromosome 7) is 7 kb long, 2 exons, monomer protein 70 KD, tetramer in solution. Fibrinogen-like protein 2 (Fgl2), a member
FGL2 A new biomarker for cancer in a simple blood test WHO IS FGL2 Human gene (chromosome 7) is 7 kb long, 2 exons, monomer protein 70 KD, tetramer in solution. Fibrinogen-like protein 2 (Fgl2), a member
Supplemental Data. Short Article. ATF4-Mediated Induction of 4E-BP1. Contributes to Pancreatic β Cell Survival. under Endoplasmic Reticulum Stress
 Cell Metabolism, Volume 7 Supplemental Data Short Article ATF4-Mediated Induction of 4E-BP1 Contributes to Pancreatic β Cell Survival under Endoplasmic Reticulum Stress Suguru Yamaguchi, Hisamitsu Ishihara,
Cell Metabolism, Volume 7 Supplemental Data Short Article ATF4-Mediated Induction of 4E-BP1 Contributes to Pancreatic β Cell Survival under Endoplasmic Reticulum Stress Suguru Yamaguchi, Hisamitsu Ishihara,
Supplementary Materials for
 advances.sciencemag.org/cgi/content/full/3/2/e1602038/dc1 Supplementary Materials for Mitochondrial metabolic regulation by GRP78 Manoj Prasad, Kevin J. Pawlak, William E. Burak, Elizabeth E. Perry, Brendan
advances.sciencemag.org/cgi/content/full/3/2/e1602038/dc1 Supplementary Materials for Mitochondrial metabolic regulation by GRP78 Manoj Prasad, Kevin J. Pawlak, William E. Burak, Elizabeth E. Perry, Brendan
Problem Set 8 Key 1 of 8
 7.06 2003 Problem Set 8 Key 1 of 8 7.06 2003 Problem Set 8 Key 1. As a bright MD/PhD, you are interested in questions about the control of cell number in the body. Recently, you've seen three patients
7.06 2003 Problem Set 8 Key 1 of 8 7.06 2003 Problem Set 8 Key 1. As a bright MD/PhD, you are interested in questions about the control of cell number in the body. Recently, you've seen three patients
Supplementary Materials for
 www.sciencesignaling.org/cgi/content/full/9/439/ra78/dc1 Supplementary Materials for Small heterodimer partner mediates liver X receptor (LXR) dependent suppression of inflammatory signaling by promoting
www.sciencesignaling.org/cgi/content/full/9/439/ra78/dc1 Supplementary Materials for Small heterodimer partner mediates liver X receptor (LXR) dependent suppression of inflammatory signaling by promoting
Supplementary Figure 1 Induction of cellular senescence and isolation of exosome. a to c, Pre-senescent primary normal human diploid fibroblasts
 Supplementary Figure 1 Induction of cellular senescence and isolation of exosome. a to c, Pre-senescent primary normal human diploid fibroblasts (TIG-3 cells) were rendered senescent by either serial passage
Supplementary Figure 1 Induction of cellular senescence and isolation of exosome. a to c, Pre-senescent primary normal human diploid fibroblasts (TIG-3 cells) were rendered senescent by either serial passage
Figure S1. (A) SDS-PAGE separation of GST-fusion proteins purified from E.coli BL21 strain is shown. An equal amount of GST-tag control, LRRK2 LRR
 Figure S1. (A) SDS-PAGE separation of GST-fusion proteins purified from E.coli BL21 strain is shown. An equal amount of GST-tag control, LRRK2 LRR and LRRK2 WD40 GST fusion proteins (5 µg) were loaded
Figure S1. (A) SDS-PAGE separation of GST-fusion proteins purified from E.coli BL21 strain is shown. An equal amount of GST-tag control, LRRK2 LRR and LRRK2 WD40 GST fusion proteins (5 µg) were loaded
TFEB-mediated increase in peripheral lysosomes regulates. Store Operated Calcium Entry
 TFEB-mediated increase in peripheral lysosomes regulates Store Operated Calcium Entry Luigi Sbano, Massimo Bonora, Saverio Marchi, Federica Baldassari, Diego L. Medina, Andrea Ballabio, Carlotta Giorgi
TFEB-mediated increase in peripheral lysosomes regulates Store Operated Calcium Entry Luigi Sbano, Massimo Bonora, Saverio Marchi, Federica Baldassari, Diego L. Medina, Andrea Ballabio, Carlotta Giorgi
genome edited transient transfection, CMV promoter
 Supplementary Figure 1. In the absence of new protein translation, overexpressed caveolin-1-gfp is degraded faster than caveolin-1-gfp expressed from the endogenous caveolin 1 locus % loss of total caveolin-1-gfp
Supplementary Figure 1. In the absence of new protein translation, overexpressed caveolin-1-gfp is degraded faster than caveolin-1-gfp expressed from the endogenous caveolin 1 locus % loss of total caveolin-1-gfp
Bioluminescence Resonance Energy Transfer (BRET)-based studies of receptor dynamics in living cells with Berthold s Mithras
 Bioluminescence Resonance Energy Transfer (BRET)-based studies of receptor dynamics in living cells with Berthold s Mithras Tarik Issad, Ralf Jockers and Stefano Marullo 1 Because they play a pivotal role
Bioluminescence Resonance Energy Transfer (BRET)-based studies of receptor dynamics in living cells with Berthold s Mithras Tarik Issad, Ralf Jockers and Stefano Marullo 1 Because they play a pivotal role
