The ch-tog/xmap215 protein is essentialfor spindle pole organization in human somatic cells
|
|
|
- Cori Terry
- 6 years ago
- Views:
Transcription
1 RESEARCH COMMUNICATION The ch-tog/xmap215 protein is essentialfor spindle pole organization in human somatic cells Fanni Gergely, 1 Viji M. Draviam, 2,4 and Jordan W. Raff 3,5 1 Department of Pharmacology, 2 Department of Zoology, and 3 Department of Genetics, University of Cambridge, Wellcome Trust/Cancer Research UK Institute, Cambridge CB2 1QR, UK The ch-tog/xmap215 family of proteins bind directly to microtubules and appear to play an essential role in stabilizing spindle microtubules. These proteins stabilize microtubules mainly by influencing microtubule plus-end dynamics, yet, in vivo, they are all strongly concentrated at spindle poles, where the minus ends of the microtubules are concentrated. In Drosophila embryos, the centrosomalprotein D-TACC is required to efficiently recruit ch-tog/msps to centrosomes. In humans, ch-tog and the three known TACC proteins have been implicated in cancer, but their functions are unknown. Here we extensively depleted TACC3 and ch- TOG from HeLa cells using RNA interference. In TACC3-depleted cells, spindles are well organized, but microtubules are partially destabilized and ch-tog is no longer concentrated on spindle microtubules. In ch- TOG-depleted cells, relatively robust spindles form, but the spindles are highly disorganized. Thus, in human somatic cells, ch-tog appears to play a major role in organizing spindle poles, and a more minor role in stabilizing spindle microtubules that is, at least in part, mediated via an interaction with TACC3. Supplemental material is available at Received August 7, 2002; revised version accepted November 4, The XMAP215/ch-TOG family of microtubule associated proteins (MAPs) are among the best studied microtubule-stabilizing proteins. In the presence of purified tubulin, these proteins stabilize microtubules mainly by stimulating growth rates at microtubule plus ends, whereas in Xenopus egg extracts they also increase microtubule plus-end stability by counteracting the activity of the microtubule catastrophe factor XKCM1 (Tournebize et al. 2000; Kinoshita et al. 2001). These proteins are essential for stabilizing spindle microtubules in both Xenopus egg extracts and worm embryos (Matthews et [Keywords: ch-tog; XMAP215; TACC; mitosis; mitotic spindle] 4 Present address: Department of Biology, Massachusetts Institute of Technology, Cambridge, Massachusetts 02139, USA 5 Corresponding author. j.raff@welc.cam.ac.uk; FAX Article and publication are at gad al. 1998; Tournebize et al. 2000; Popov et al. 2001). Surprisingly, although XMAP215/ch-TOG mainly influences microtubule plus-end dynamics, these proteins are strongly concentrated at centrosomes, where the minus ends of the microtubules are clustered (Nabeshima et al. 1995; Wang et al. 1997; Charrasse et al. 1998; Matthews et al. 1998; Cullen et al. 1999; Tournebize et al. 2000; Garcia et al. 2001). In Drosophila, the centrosomal protein D-TACC has been shown to be required to efficiently recruit Msps/ ch-tog to centrosomes (Cullen et al. 2001; Lee et al. 2001). The interaction between D-TACC and Msps/ch- TOG is highly conserved, and in human cells, all three known TACC proteins are concentrated at centrosomes and can interact with ch-tog (Gergely et al. 2000; Cullen et al. 2001). Interestingly, ch-tog and the three human TACC proteins have been implicated in cancer but the function of these proteins in mammalian cells is not known (Gergely 2002; Raff 2002). An attractive possibility is that these proteins function in human somatic cells as they do in frog and fly embryos, and are required to stabilize spindle microtubules. Perturbing their function could therefore lead to spindle defects and so contribute to the genetic instability that is essential for the development of cancer (Li et al. 1997; Brinkley and Goepfert 1998; Doxsey 1998; Salisbury et al. 1999). Results and Discussion To test whether TACC3 and ch-tog play an essential role in cell division in humans, we used small interfering RNAs (sirnas) to reduce the levels of each protein in HeLa cells (Elbashir et al. 2001). In cells transfected with TACC3 sirna, the overall levels of TACC3 were reduced by 80% 90% after 48 h (Fig. 1A, lane 3). A similar treatment of cells with ch-tog sirna produced an 60% overall reduction in ch-tog protein levels (Fig. 1, lane 3). Both proteins were also depleted to these respective levels in cells simultaneously treated with both sirnas (Fig. 1B, lane 4). As the efficiency of transfection with sirnas often varied from experiment to experiment and from cell to cell, we tested the levels of depletion in individual cells by immunofluorescence. We found that the levels of TACC3 and ch-tog protein normally vary during the cell cycle, and are highest during mitosis (see Supplementary Fig. 1). We therefore only compared the levels of TACC3 and ch-tog on centrosomes and spindles in mitotic cells. Approximately 70% of sirna-treated mitotic cells had strongly reduced levels of TACC3 (scored as <20% normal levels) or ch-tog (scored as <30% normal levels) on their centrosomes/ spindles, whereas the remaining 30% of mitotic cells had only weakly reduced or normal levels (Fig. 2). In TACC3-depleted HeLa cells, the mitotic spindles were usually bipolar and well organized, and the microtubules did not appear to be dramatically destabilized (Figs. 2A, 3A). In 30% of all TACC3-depleted metaphase cells, however, at least one chromosome was not aligned properly at the metaphase plate (Fig. 3A,C, graph 5). There was a strong correlation between this chromosome alignment defect and the levels of TACC3 observed in a cell by immunofluorescence. Cells with high levels of TACC3 rarely had a chromosome alignment defect (<5%), whereas most ( 70%) cells that had no, or 336 GENES & DEVELOPMENT 17: by Cold Spring Harbor Laboratory Press ISSN /03 $5.00;
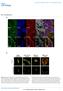
2 ch-tog and TACC3 in human cells Figure 1. TACC3 and ch-tog protein levels are reduced by sirna treatment. A Western blot showing the levels of TACC3 (A) and ch-tog (B) after 48 h of sirna treatment. Actin is shown as a loading control. (A) Mock-depleted cells (lane 1), ch-tog-depleted cells (lane 2), TACC3-depleted cells (lane 3), and TACC3- and ch-tog-depleted cells (lane 4). (B) Mock-depleted cells (lane 1), TACC3-depleted cells (lane 2), ch-togdepleted cells (lane 3), and ch-tog- and TACC3-depleted cells (lane 4). (Fig. 4; see Materials and Methods). This comparison proved relatively straightforward for TACC3-depleted cells, as the spindles in these cells were usually well organized and bipolar. The microtubule density in these spindles was reduced by 20% (Fig. 4A). In ch-tog-depleted cells, however, the spindles were so disorganized that it was impossible to objectively define the spindle area, and no meaningful quantitative comparison could be made. In an attempt to overcome this problem, we treated cells with sirnas for 24 h and then chilled them for 2 h to depolymerize the microtubules. We then warmed the cells to allow the microtubules to regrow for varying lengths of time before fixing the cells for analysis. Again, we analyzed areas of an sirna-treated coverslip that contained at least one strongly depleted cell and a cell that had normal levels of TACC3 or ch-tog. This assay produced a striking qualitative difference between the rates of microtubule regrowth in depleted and nondepleted cells. After 25 min of recovery, relatively normal bipolar spindles with the majority of chromosomes aligned at a metaphase plate had reformed in the majority of mitotic cells ( 60%) that were not depleted of either protein (Fig. 4B,C). In TACC3-depleted cells, the spindles that reformed were usually bipolar and well or- very little, detectable TACC3 showed the chromosome alignment defect. In addition, TACC3-depleted cells had an approximately threefold-higher mitotic index than control cells (Fig. 3C, graph 1), suggesting that the failure to properly align chromosomes at metaphase was triggering the spindle assembly checkpoint (Shah and Cleveland 2000). This delay in mitosis appeared to be transient, however, as a small number of TACC3-depleted cells were observed exiting mitosis relatively normally even when no TACC3 was detectable on the spindle (data not shown). In ch-tog-depleted mitotic cells, the spindle microtubules did not appear to be dramatically destabilized, but the spindles were usually highly disorganized (Figs. 2B, 3B,C, graph 6). As in TACC3-depleted cells, chromosomes often failed to align properly at the metaphase plate, but this defect was much stronger in ch-tog-depleted cells (Fig. 3B,C, graph 3), and the mitotic index in these cells was elevated by approximately fivefold (Fig. 3C, graph 1). Even these highly disorganized cells, however, were capable of eventually exiting mitosis, and we observed many cells in interphase with an aberrant DNA content (Fig. 3C, graph 2). In both TACC3- and ch-togdepleted cells, we saw no obvious defects in microtubule organization in interphase cells. These observations demonstrate that both TACC3 and ch-tog are essential for mitosis in HeLa cells. Surprisingly, however, microtubules did not appear to be dramatically destabilized by the depletion of either protein. To test whether spindle microtubules were subtly destabilized by the depletion of TACC3 or ch-tog, we attempted to quantify the intensity of spindle fluorescence in control and depleted cells. As fluorescence levels often varied between different coverslips, we selected areas of sirna-treated coverslips that contained at least one strongly depleted cell and a cell that had normal levels of TACC3 or ch-tog. We then optically sectioned each cell and calculated the average pixel intensity in a manually defined area that contained the entire spindle region Figure 2. Immunofluorescence analysis of TACC3 and ch- TOG protein levels in sirna-treated cells. (A) Mock-depleted (top panels) and TACC3-depleted (bottom panels) HeLa cells stained to reveal the distribution of TACC3, DNA, and microtubules. In this and all subsequent experiments, images were acquired from experimental and control cells that were treated and processed for immunofluorescence at the same time using identical settings on the confocal microscope. The TACC3 sirna treatment has strongly reduced TACC3 levels in two mitotic cells (arrows), but substantial levels of TACC3 remain in one mitotic cell (arrowhead). (B) Mock-depleted (top panels) and ch-tog-depleted (bottom panels) HeLa cells stained to reveal the distribution of ch-tog, DNA, and microtubules. The ch-tog sirna treatment strongly reduced ch-tog levels in most cells visible in this field, but small amounts of protein are still detectable on the centrosomes and spindles of some mitotic cells (arrowhead). Bar, 10 µm. GENES & DEVELOPMENT 337
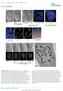
3 Gergely et al. Figure 3. Mitotic defects in TACC3- and ch-tog-depleted HeLa cells. (A,B) Mock-depleted (left panels) and either TACC3-depleted (A, right panels) or ch- TOG-depleted (B, right panels) cells were stained to reveal the distribution of microtubules (green in merged image), TACC3 or ch-tog (blue in merged image), and DNA (red in merged image). Bar, 10 µm. (C) Quantitation of the defects observed in TACC3- and ch-tog-depleted cells. In all graphs, the percentage of cells exhibiting a particular phenotype is indicated for mock-depleted (blue bars), TACC3-depleted (red bars), ch-tog-depleted (yellow bars), or TACC3- and ch- TOG-depleted (orange bars) cells. (Graph 1) The mitotic index in the population of cells as determined by phospho-histone H3 staining. (Graph 2) The percentage of interphase cells exhibiting multiple or satellite nuclei. (Graph 3) The percentage of mitotic cells in a prometaphase-like state (with no discernible metaphase plate). (Graph 4) The percentage of mitotic cells with a normal metaphase plate. (Graph 5) The percentage of mitotic cells with a clear metaphase plate, but with at least one chromosome not aligned at the equator. (Graph 6) The percentage of mitotic cells with more than two spindle poles. Two-hundred-fifty cells were scored from each of four independent experiments for both the mitotic index and the multinucleated phenotype. Fifty metaphase cells from two independent experiments were scored for the prometaphase, metaphase, and lagging chromosome phenotypes. Thirty mitotic cells were scored from each of four independent experiments for the presence of multipolar spindles. Error bars represent the standard deviation. ganized, but they had an 30% lower density of microtubules (Fig. 4A), and only 25% had successfully aligned the majority of their chromosomes at a metaphase plate (Fig. 4B). In ch-togdepleted cells, only 10% of mitotic cells had successfully aligned the majority of their chromosomes on a metaphase plate (Fig. 4B), and microtubule density was clearly qualitatively lower than normal (Fig. 4C). Again, however, this difference in microtubule density was impossible to quantify, as these spindles were too disorganized (Fig. 4C). These observations strongly suggest that microtubules are partially destabilized by the depletion of TACC3 and ch-tog. It was previously shown that D-TACC is required to efficiently recruit ch-tog/ Msps to centrosomes and spindle poles in Drosophila embryos and oocytes (Cullen et al. 2001; Lee et al. 2001). In TACC3-depleted cells, however, the level of ch-tog at centrosomes was only slightly, if at all, reduced (Fig. 5A). This may be because ch-tog can still be recruited to spindle poles by TACC1 or TACC2, which remain concentrated at centrosomes in TACC3- depleted cells (data not shown). Unexpectedly, however, the level of ch-tog on the spindle was greatly reduced in TACC3- depleted cells (Fig. 5A). This result is very surprising, as ch-tog can bind directly to purified microtubules in vitro (Spittle et al. 2000). In human somatic cells, it seems that ch-tog requires TACC3 to bind efficiently to spindle microtubules. This failure to recruit ch-tog to mitotic spindles presumably explains why spindle microtubules are partially destabilized in TACC3- depleted cells. In contrast, the depletion of ch-tog did not appear to disrupt the localization of TACC3 (Fig. 5B), TACC1, or TACC2 (data not shown) on centrosomes or spindles. As the depletion of ch-tog was never as efficient as the depletion of TACC3 (Fig. 1), it is possible that the residual ch-tog is required to efficiently localize the TACC proteins to centrosomes/spindles. The depletion of either TACC3 or ch- TOG did not seem to affect the centrosomal localization of -tubulin (see Supplementary Fig. 2), TACC1, or TACC2 (data not shown), and all of these proteins were recruited to the extra spindle poles often found in the ch-tog-depleted cells. This raised the possibility that these disorganized spindle poles might not arise from a defect in spindle pole organization, but rather from the presence of extra centrosomes accumulated from previously aberrant rounds of mitosis. To test this possibility, we followed mitosis in living HeLa cells during their first round of division after ch-tog depletion (see Supplementary Fig. 3). Initially, we attempted to do this by depleting ch-tog in synchronized cells that were injected with a GFP-tubulin construct, allowing us to fol- 338 GENES & DEVELOPMENT
4 ch-tog and TACC3 in human cells Figure 4. The depletion of TACC3 and ch-tog partially destabilizes spindle microtubules. (A) A graph showing the relative spindle fluorescence intensity in nondepleted or TACC3-depleted mitotic cells (black bars), or in nondepleted or TACC3-depleted cells that have been chilled and allowed to recover for 25 min (hatched bars). Note that the spindle fluorescence of both sets of nondepleted cells has been normalized to 1. (B) A graph showing the percentage of cells that have properly aligned the majority of their chromosomes at a metaphase plate after 25 min of recovery from cold treatment. Error bars represent the standard deviation. (C, top panels) A field of cells treated with TACC3 sirna. One mitotic cell (marked with *) has not been significantly depleted of TACC3, but two other mitotic cells (marked with arrowheads) have been significantly depleted. Fields such as these were used to compare the spindle microtubule density in the depleted and nondepleted cells (see Materials and Methods). Fields of TACC3 (middle panels) and ch-tog (bottom panels) sirna-treated cells that were chilled to depolymerize the microtubules and then allowed to recover for 25 min. In each panel, a nondepleted mitotic cell is marked with *, and depleted cells are marked with arrowheads. Bar, 10 µm. low the behavior of microtubules. Unfortunately, GFPtubulin proved to be deleterious to cells, and very few injected cells entered mitosis. We therefore followed the behavior of spindle poles indirectly, by monitoring the behavior of ch-tog-depleted cells that were coinjected with a histone-gfp construct. All 10 ch-tog-depleted cells observed in this way entered a prolonged prometaphase period in which some of the chromosomes repeatedly aligned on a metaphase plate-like structure (that often appeared to be organized by multiple poles), and then reverted to a more disorganized state (Supplementary Fig. 3). Eight of these cells eventually exited mitosis, and the chromosomes were often segregated unevenly between multiple poles, although chromosome segregation and cytokinesis appeared to occur at normal rates. None of these defects were observed in mock-injected control cells. Thus, spindle pole organization is disrupted even during the first round of mitosis after ch- TOG depletion. Our results have two important implications for mechanisms of TACC3 and ch-tog function in human cells. First, TACC3 is essential for mitosis in HeLa cells. This result is important because a recent study reported that TACC3 knockout mice die during embryogenesis due to a massive increase in apoptosis, and this phenotype is partially rescued by lowering p53 levels (Piekorz et al. 2002). Surprisingly, however, TACC3 / ;p53 / cells have only a slight increase in chromosomal abnormalities, suggesting that TACC3 is not essential for spindle function in mice. We suspect that TACC3 knockout mice, like TACC3-depleted human cells, have high levels of relatively subtle mitotic defects that cause a transient arrest in mitosis. These transiently arrested cells would then normally be eliminated by apoptosis in a p53-dependent manner during the next cell cycle, even if no gross chromosomal abnormalities were generated (Lanni and Jacks 1998; Ciciarello et al. 2001). Second, although ch-tog/xmap215 plays an essential role in stabilizing spindle microtubules in frog egg extracts and worm embryos (Matthews et al. 1998; Tour- GENES & DEVELOPMENT 339
5 Gergely et al. nebize et al. 2000; Popov et al. 2001), in human somatic cells it appears to play a more minor role in stabilizing spindle microtubules, and a more major role in organizing spindle poles. This may reflect a difference between embryonic and somatic cell types, as the phenotype in ch-tog-depleted HeLa cells is reminiscent of that observed in Drosophila msps mutant larval brain cells that are partially depleted of Msps/ch-TOG (Cullen et al. 1999). In frog egg extracts, XMAP215 stabilizes microtubules primarily by counteracting the microtubule-destabilizing activity of XKCM1/Kin1 (Desai et al. 1999). Perhaps XKCM1 is less active in somatic cells, so it is less important for XMAP215/ch-TOG to counteract its activity. It was recently shown that the changes in microtubule dynamics that occur during the cell cycle are different in mammalian tissue culture cells and Xenopus egg extracts (Rusan et al. 2001). How might ch-tog function to organize spindle poles in somatic cells? When overexpressed in HeLa cells, the TACC proteins form large polymers in the cytoplasm that also contain ch-tog (Gergely et al. 2000). Perhaps the TACCs and ch-tog can form a structural lattice at centrosomes that functions both to maintain the integrity of spindle poles and to stabilize spindle microtubules. In the TACC3-depleted cells, the spindle pole organizing function might not be disrupted because TACC1 and/or TACC2 can still recruit ch-tog to centrosomes. We are currently attempting to test this possibility by simultaneously depleting all three TACC proteins from HeLa cells. Regardless of its mechanism, the role of ch-tog/xmap215 in organizing spindle poles may explain the longstanding mystery of why this potent regulator of microtubule plus-end dynamics is so strongly concentrated at centrosomes. Materials and methods Immunofluoresence Human HeLa cells were cultured, fixed, and stained with antibodies as described (Gergely et al. 2000). Rabbit anti-ch-tog antibodies (Dionne et al. 2000; Tournebize et al. 2000), and rabbit anti-tacc3 antibodies (Gergely et al. 2000) were described previously. DM1 anti- -tubulin antibody (Sigma) was used at 1/1000 dilution, and GTU-88 anti- -tubulin antibody (Sigma) was used at 1/500 dilution. Phospho-histone H3 antibody (Upstate Biotechnology) was used at 1/1000 dilution to identify mitotic cells. Appropriate Alexa488- (Molecular Probes), Cy5-, or Cy3 (Jackson)-coupled secondary antibodies were used at 1/500 dilution. DNA was detected with either propidium iodide or TOTO. All imaging was performed using a Bio-Rad Radiance 2000 scanning confocal microscope using identical settings on the confocal microscope. The images were imported into Adobe Photoshop 6.0, and all of the images in any individual figure were processed in exactly the same way. Figure 5. The localization of ch-tog is disrupted in TACC3- depleted cells. (A) The distribution of ch-tog (left panels, red in merged image) and microtubules (middle panels, green in merged image) in mock-depleted (top panels) and TACC3-depleted (bottom panels) cells. Ch-TOG is detectable on centrosomes, but is not detectable on spindle microtubules in the TACC3-depleted cells. (B) The distribution of TACC3 (left panels, red in merged image) and microtubules (middle panels, green in merged image) in mock-depleted (top panels) and ch- TOG-depleted (bottom panels) cells. TACC3 remains concentrated on spindles in the ch-tog-depleted cells. Bar, 10 µm. RNA preparation and transfection Twenty-one-nucleotide (nt) RNAs were synthesized, deprotected, and gel-purified by Dharmacon. RNAs were annealed as described (Elbashir et al. 2001). The sirna sequences targeting ch-tog corresponded to region nt, and the sirna targeting TACC3 corresponded to region nt of the coding sequence. The control RNA duplex used in all mock sirna experiments corresponded to a 21-nt-long region of Fanconi anemia protein E. Cells were seeded on coverslips to 60% confluence in 24-well plates, 16 h before transfection. We transfected 80 pmole of RNA duplex per well using the Oligofectamine reagent (Life Technologies) according to the manufacturer s instructions. Western blotting Whole-cell extracts were made by pelleting HeLa cells and boiling the pellet in SDS sample buffer. The extracts were separated by SDS-PAGE and blotted to nitrocellulose. Blots were incubated with primary antibodies at 1 µg/ml final concentration, and antibody binding was detected using the Supersignal kit (Pierce) according to the manufacturer s instructions. For quantitation, the blots were scanned into Adobe Photoshop and then imported into NIH Image The total pixel intensity of each band was counted, and the degree of depletion was calculated after normalizing this figure to the total pixel intensity of an actin control. Microtubule regrowth assay and spindle intensity quantification Twenty-four hours after the addition of the sirna/oligofectamine solution, cells were transferred to 4 C for 2 h. Cells were then recovered at 37 C and fixed at various time points. Cells were stained with anti- tubulin and anti-tacc3 or ch-tog antibodies. Individual mitotic cells were optically sectioned using a confocal laser scanning microscope. These 3D datasets (z-series of 14-en face optical sections, 1-µm steps) were projected onto a single image plane. Each channel of the projected image was inverted in NIH Image We measured spindle microtubule intensity by defining the spindle area manually (containing the centrosomes and all visible microtubule bundles projecting towards chromosomes, but excluding astral microtubules), and calculated the mean pixel intensity per unit area of spindle. The same approach was applied to measure TACC3 antibody staining intensity in the spindle area. Having obtained both measurements for each cell, we scored them according to their TACC3 levels and divided them into two groups. The first group consisted of images of cells from the TACC3 RNA-treated coverslips that exhibited comparable levels (within 10%) of TACC3 in untreated cells, and cells in the second group had at least a threefold reduction in TACC3 340 GENES & DEVELOPMENT
6 ch-tog and TACC3 in human cells levels. Each bar in Figure 4A represents data collected from three independent experiments, in which 20 cells (10 control and 10 TACC3-depleted) were scored. Datasets were normalized to control within each experiment. Acknowledgments We thank Duane Compton, Tony Hyman, K.J. Patel, and G. Wahl for reagents. We thank Jon Pines and members of the Raff lab for comments on the manuscript. This work was supported by a Royal Society Dorothy Hodgkin Fellowship (F.G.), a Cambridge NehruScholarship (V.M.D.), and a Wellcome Trust Senior Research Fellowship (J.R.). The publication costs of this article were defrayed in part by payment of page charges. This article must therefore be hereby marked advertisement in accordance with 18 USC section 1734 solely to indicate this fact. References Brinkley, B.R. and Goepfert, T.M Supernumerary centrosomes and cancer: Boveri s hypothesis resurrected. Cell Motil. Cytoskeleton 41: Charrasse, S., Schroeder, M., Gauthier-Rouviere, C., Ango, F., Cassimeris, L., Gard, D.L., and Larroque, C The TOGp protein is a new human microtubule-associated protein homologous to the Xenopus XMAP215. J. Cell Sci. 111: Ciciarello, M., Mangiacasale, R., Casenghi, M., Zaira Limongi, M., D Angelo, M. Soddu., S., Lavia, P., and Cundari, E p53 displacement from centrosomes and p53-mediated G1 arrest following transient inhibition of the mitotic spindle. J. Biol. Chem. 276: Cullen, C.F. and Ohkura, H Msps protein is localized to acentrosomal poles to ensure bipolarity of Drosophila meiotic spindles. Nat. Cell Biol. 3: Cullen, C.F., Deak, P., Glover, D.M., and Ohkura, H Mini spindles. A gene encoding a conserved microtubule-associated protein required for the integrity of the mitotic spindle in Drosophila. J. Cell Biol. 146: Desai, A., Verma, S., Mitchison, T.J., and Walczak, C.E Kin I kinesins are microtubule-destabilizing enzymes. Cell 96: Dionne, M.A., Sanchez, A., and Compton, D.A ch-togp is required for microtubule aster formation in a mammalian mitotic extract. J. Biol. Chem. 275: Doxsey, S The centrosome A tiny organelle with big potential. Nat. Genet. 20: Elbashir, S.M., Harborth, J., Lendeckel, W., Yalcin, A., Weber, K., and Tuschl, T Duplexes of 21-nucleotide RNAs mediate RNA interference in cultured mammalian cells. Nature 411: Garcia, M.A., Vardy, L., Koonrugsa, N., and Toda, T Fission yeast ch-tog/xmap215 homologue Alp14 connects mitotic spindles with the kinetochore and is a component of the Mad2-dependent spindle checkpoint. EMBO J. 20: Gergely, F Centrosomal TACCtics. Bioessays 10: Gergely, F., Karlsson, C., Still, I., Cowell, J., Kilmartin, J., and Raff, J.W The TACC domain identifies a new family of proteins that can interact with centrosomes and microtubules. Proc. Natl. Acad. Sci. 97: Kinoshita, K., Arnal, I., Desai, A., Drechsel, D.N., and Hyman, A.A Reconstitution of physiological microtubule dynamics using purified components. Science 294: Lanni, J.S. and Jacks, T Characterization of the p53-dependent postmitotic checkpoint following spindle disruption. Mol. Cell Biol. 18: Lee, M.J., Gergely, F., Jeffers, K., Peak-Chew, S.Y., and Raff, J.W Msps/XMAP215 interacts with the centrosomal protein D-TACC to regulate microtubule behaviour. Nat. Cell Biol. 3: Li, R., Yerganian, G., Duesberg, P., Kraemer, A., Willer, A., Rausch, C., Hehlmann, R Aneuploidy correlated 100% with chemical transformation of Chinese hamster cells. Proc. Natl. Acad. Sci. 94: Matthews, L.R., Carter, P., Thierry-Mieg, D., and Kemphues, K ZYG-9, a Caenorhabditis elegans protein required for microtubule organization and function, is a component of meiotic and mitotic spindle poles. J. Cell Biol.. 141: Nabeshima, K., Kurooka, H., Takeuchi, M., Kinoshita, K., Nakaseko, Y., and Yanagida, M p93dis1, which is required for sister chromatid separation, is a novel microtubule and spindle pole body-associating protein phosphorylated at the Cdc2 target sites. Genes and Dev 9: Piekorz, R.P., Hoffmeyer, A., Duntsch, C.D., McKay, C., Nakajima, H., Sexl, V., Snyder, L., Rehg, J., and Ihle, J.N The centrosomal protein TACC3 is essential for hematopoietic stem cell function and genetically interfaces with p53-regulated apoptosis. EMBO J. 21: Popov, A.V., Pozniakovsky, A., Arnal, I., Antony, C., Ashford, A.J., Kinoshita, K., Tournebize, R., Hyman, A.A., and Karsenti, E XMAP215 regulates microtubule dynamics through two distinct domains. EMBO J. 20: Raff, J.W Centrosomes and cancer: Lessons from a TACC. Trends Cell Biol. 12: Rusan, N.M., Fagerstrom, C.J., Yvon, A.M., and Wadsworth, P Cell cycle-dependent changes in microtubule dynamics in living cells expressing green fluorescent protein- tubulin. Mol. Biol. Cell 12: Salisbury, J.L., Whitehead, C.M., Lingle, W.L., and Barrett, S.L Centrosomes and cancer. Biol. Cell 91: Shah, J.V. and Cleveland, D.W Waiting for anaphase: Mad2 and the spindle assembly checkpoint. Cell 103: Spittle, C., Charrasse, S., Larroque, C., and Cassimeris, L The interaction of TOGp with microtubules and tubulin. J. Biol. Chem. 275: Tournebize, R., Popov, A., Kinoshita, K., Ashford, A.J., Rybina, S., Pozniakovsky, A., Mayer, T.U., Walczak, C.E., Karsenti, E., and Hyman, A.A Control of microtubule dynamics by the antagonistic activities of XMAP215 and XKCM1 in Xenopus egg extracts. Nat. Cell Biol. 2: Towbin, H., Staehlin, T., and Gordon, J Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: Procedures and some applications. Proc. Natl. Acad. Sci. 80: Wang, P.J. and Huffaker, T.C Stu2p: A microtubule-binding protein that is an essential component of the yeast spindle pole body. J. Cell Biol. 139: GENES & DEVELOPMENT 341
klp-18 (RNAi) Control. supplementary information. starting strain: AV335 [emb-27(g48); GFP::histone; GFP::tubulin] bleach
![klp-18 (RNAi) Control. supplementary information. starting strain: AV335 [emb-27(g48); GFP::histone; GFP::tubulin] bleach klp-18 (RNAi) Control. supplementary information. starting strain: AV335 [emb-27(g48); GFP::histone; GFP::tubulin] bleach](/thumbs/91/104639484.jpg) DOI: 10.1038/ncb1891 A. starting strain: AV335 [emb-27(g48); GFP::histone; GFP::tubulin] bleach embryos let hatch overnight transfer to RNAi plates; incubate 5 days at 15 C RNAi food L1 worms adult worms
DOI: 10.1038/ncb1891 A. starting strain: AV335 [emb-27(g48); GFP::histone; GFP::tubulin] bleach embryos let hatch overnight transfer to RNAi plates; incubate 5 days at 15 C RNAi food L1 worms adult worms
Supplementary Figure S1
 Supplementary Figure S1 Supplementary Figure S1. PARP localization patterns using GFP-PARP and PARP-specific antibody libraries GFP-PARP localization in non-fixed (A) and formaldehyde fixed (B) GFP-PARPx
Supplementary Figure S1 Supplementary Figure S1. PARP localization patterns using GFP-PARP and PARP-specific antibody libraries GFP-PARP localization in non-fixed (A) and formaldehyde fixed (B) GFP-PARPx
Supplemental Materials. STK16 regulates actin dynamics to control Golgi organization and cell cycle
 Supplemental Materials STK16 regulates actin dynamics to control Golgi organization and cell cycle Juanjuan Liu 1,2,3, Xingxing Yang 1,3, Binhua Li 1, Junjun Wang 1,2, Wenchao Wang 1, Jing Liu 1, Qingsong
Supplemental Materials STK16 regulates actin dynamics to control Golgi organization and cell cycle Juanjuan Liu 1,2,3, Xingxing Yang 1,3, Binhua Li 1, Junjun Wang 1,2, Wenchao Wang 1, Jing Liu 1, Qingsong
Outline Interphase Mitotic Stage Cell Cycle Control Apoptosis Mitosis Mitosis in Animal Cells Cytokinesis Cancer Prokaryotic Cell Division
 The Cell Cycle and Cellular Reproduction Chapter 9 Outline Interphase Mitotic Stage Cell Cycle Control Apoptosis Mitosis Mitosis in Animal Cells Cytokinesis Cancer Prokaryotic Cell Division 1 2 Interphase
The Cell Cycle and Cellular Reproduction Chapter 9 Outline Interphase Mitotic Stage Cell Cycle Control Apoptosis Mitosis Mitosis in Animal Cells Cytokinesis Cancer Prokaryotic Cell Division 1 2 Interphase
Supplementary information. The Light Intermediate Chain 2 Subpopulation of Dynein Regulates Mitotic. Spindle Orientation
 Supplementary information The Light Intermediate Chain 2 Subpopulation of Dynein Regulates Mitotic Spindle Orientation Running title: Dynein LICs distribute mitotic functions. Sagar Mahale a, d, *, Megha
Supplementary information The Light Intermediate Chain 2 Subpopulation of Dynein Regulates Mitotic Spindle Orientation Running title: Dynein LICs distribute mitotic functions. Sagar Mahale a, d, *, Megha
T H E J O U R N A L O F C E L L B I O L O G Y
 T H E J O U R N A L O F C E L L B I O L O G Y Supplemental material Dunsch et al., http://www.jcb.org/cgi/content/full/jcb.201202112/dc1 Figure S1. Characterization of HMMR and CHICA antibodies. (A) HeLa
T H E J O U R N A L O F C E L L B I O L O G Y Supplemental material Dunsch et al., http://www.jcb.org/cgi/content/full/jcb.201202112/dc1 Figure S1. Characterization of HMMR and CHICA antibodies. (A) HeLa
Nature Structural and Molecular Biology: doi: /nsmb Supplementary Figure 1
 Supplementary Figure 1 Mutational analysis of the SA2-Scc1 interaction in vitro and in human cells. (a) Autoradiograph (top) and Coomassie stained gel (bottom) of 35 S-labeled Myc-SA2 proteins (input)
Supplementary Figure 1 Mutational analysis of the SA2-Scc1 interaction in vitro and in human cells. (a) Autoradiograph (top) and Coomassie stained gel (bottom) of 35 S-labeled Myc-SA2 proteins (input)
T H E J O U R N A L O F C E L L B I O L O G Y
 T H E J O U R N A L O F C E L L B I O L O G Y Supplemental material Lu et al., http://www.jcb.org/cgi/content/full/jcb.201012063/dc1 Figure S1. Kinetics of nuclear envelope assembly, recruitment of Nup133
T H E J O U R N A L O F C E L L B I O L O G Y Supplemental material Lu et al., http://www.jcb.org/cgi/content/full/jcb.201012063/dc1 Figure S1. Kinetics of nuclear envelope assembly, recruitment of Nup133
Lecture 10. G1/S Regulation and Cell Cycle Checkpoints. G1/S regulation and growth control G2 repair checkpoint Spindle assembly or mitotic checkpoint
 Lecture 10 G1/S Regulation and Cell Cycle Checkpoints Outline: G1/S regulation and growth control G2 repair checkpoint Spindle assembly or mitotic checkpoint Paper: The roles of Fzy/Cdc20 and Fzr/Cdh1
Lecture 10 G1/S Regulation and Cell Cycle Checkpoints Outline: G1/S regulation and growth control G2 repair checkpoint Spindle assembly or mitotic checkpoint Paper: The roles of Fzy/Cdc20 and Fzr/Cdh1
The Cell Cycle. Dr. SARRAY Sameh, Ph.D
 The Cell Cycle Dr. SARRAY Sameh, Ph.D Overview When an organism requires additional cells (either for growth or replacement of lost cells), new cells are produced by cell division (mitosis) Somatic cells
The Cell Cycle Dr. SARRAY Sameh, Ph.D Overview When an organism requires additional cells (either for growth or replacement of lost cells), new cells are produced by cell division (mitosis) Somatic cells
Supplementary figures
 Supplementary figures Supplementary Figure 1. B cells stimulated with pokeweed mitogen display normal mitotic figures but not cells infected with B95-8. The figures show cells stimulated with pokeweed
Supplementary figures Supplementary Figure 1. B cells stimulated with pokeweed mitogen display normal mitotic figures but not cells infected with B95-8. The figures show cells stimulated with pokeweed
SUPPLEMENTARY INFORMATION
 DOI: 1.138/ncb222 / b. WB anti- WB anti- ulin Mitotic index (%) 14 1 6 2 T (h) 32 48-1 1 2 3 4 6-1 4 16 22 28 3 33 e. 6 4 2 Time (min) 1-6- 11-1 > 1 % cells Figure S1 depletion leads to mitotic defects
DOI: 1.138/ncb222 / b. WB anti- WB anti- ulin Mitotic index (%) 14 1 6 2 T (h) 32 48-1 1 2 3 4 6-1 4 16 22 28 3 33 e. 6 4 2 Time (min) 1-6- 11-1 > 1 % cells Figure S1 depletion leads to mitotic defects
Biology is the only subject in which multiplication is the same thing as division
 Biology is the only subject in which multiplication is the same thing as division 2007-2008 The Cell Cycle: Cell Growth, Cell Division Ch. 10 Where it all began You started as a cell smaller than a period
Biology is the only subject in which multiplication is the same thing as division 2007-2008 The Cell Cycle: Cell Growth, Cell Division Ch. 10 Where it all began You started as a cell smaller than a period
Cell division functions in 1. reproduction, 2. growth, and 3. repair
 Cell division functions in 1. reproduction, 2. growth, and 3. repair What do you think you are looking at here??? Can something like you or I do this??? Fig. 12.1 How did you start out? How did you grow?
Cell division functions in 1. reproduction, 2. growth, and 3. repair What do you think you are looking at here??? Can something like you or I do this??? Fig. 12.1 How did you start out? How did you grow?
Biology is the only subject in which multiplication is the same thing as division
 Biology is the only subject in which multiplication is the same thing as division The Cell Cycle: Cell Growth, Cell Division 2007-2008 2007-2008 Getting from there to here Going from egg to baby. the original
Biology is the only subject in which multiplication is the same thing as division The Cell Cycle: Cell Growth, Cell Division 2007-2008 2007-2008 Getting from there to here Going from egg to baby. the original
WDR62 is associated with the spindle pole and mutated in human microcephaly
 WDR62 is associated with the spindle pole and mutated in human microcephaly Adeline K. Nicholas, Maryam Khurshid, Julie Désir, Ofélia P. Carvalho, James J. Cox, Gemma Thornton, Rizwana Kausar, Muhammad
WDR62 is associated with the spindle pole and mutated in human microcephaly Adeline K. Nicholas, Maryam Khurshid, Julie Désir, Ofélia P. Carvalho, James J. Cox, Gemma Thornton, Rizwana Kausar, Muhammad
Cell Cycle, Mitosis, and Microtubules. LS1A Final Exam Review Friday 1/12/07. Processes occurring during cell cycle
 Cell Cycle, Mitosis, and Microtubules LS1A Final Exam Review Friday 1/12/07 Processes occurring during cell cycle Replicate chromosomes Segregate chromosomes Cell divides Cell grows Cell Growth 1 The standard
Cell Cycle, Mitosis, and Microtubules LS1A Final Exam Review Friday 1/12/07 Processes occurring during cell cycle Replicate chromosomes Segregate chromosomes Cell divides Cell grows Cell Growth 1 The standard
The Cell Cycle. Packet #9. Thursday, August 20, 2015
 1 The Cell Cycle Packet #9 2 Introduction Cell Cycle An ordered sequence of events in the life of a dividing eukaryotic cell and is a cellular asexual reproduction. The contents of the parent s cell nucleus
1 The Cell Cycle Packet #9 2 Introduction Cell Cycle An ordered sequence of events in the life of a dividing eukaryotic cell and is a cellular asexual reproduction. The contents of the parent s cell nucleus
BIOLOGY - CLUTCH CH.12 - CELL DIVISION.
 !! www.clutchprep.com CONCEPT: CELL DIVISION Cell division is the process by which one cell splits into two or more daughter cells. Cell division generally requires that cells produce enough materials,
!! www.clutchprep.com CONCEPT: CELL DIVISION Cell division is the process by which one cell splits into two or more daughter cells. Cell division generally requires that cells produce enough materials,
Unduplicated. Chromosomes. Telophase
 10-2 Cell Division The Cell Cycle Interphase Mitosis Prophase Cytokinesis G 1 S G 2 Chromatin in Parent Nucleus & Daughter Cells Chromatin Daughter Nuclei Telophase Mitotic Anaphase Metaphase Use what
10-2 Cell Division The Cell Cycle Interphase Mitosis Prophase Cytokinesis G 1 S G 2 Chromatin in Parent Nucleus & Daughter Cells Chromatin Daughter Nuclei Telophase Mitotic Anaphase Metaphase Use what
CELL CYCLE INTRODUCTION PART I ANIMAL CELL CYCLE INTERPHASE
 CELL CYCLE INTRODUCTION The nuclei in cells of eukaryotic organisms contain chromosomes with clusters of genes, discrete units of hereditary information consisting of double-stranded DNA. Structural proteins
CELL CYCLE INTRODUCTION The nuclei in cells of eukaryotic organisms contain chromosomes with clusters of genes, discrete units of hereditary information consisting of double-stranded DNA. Structural proteins
Regulators of Cell Cycle Progression
 Regulators of Cell Cycle Progression Studies of Cdk s and cyclins in genetically modified mice reveal a high level of plasticity, allowing different cyclins and Cdk s to compensate for the loss of one
Regulators of Cell Cycle Progression Studies of Cdk s and cyclins in genetically modified mice reveal a high level of plasticity, allowing different cyclins and Cdk s to compensate for the loss of one
T H E J O U R N A L O F C E L L B I O L O G Y
 T H E J O U R N A L O F C E L L B I O L O G Y Supplemental material Courtheoux et al., http://www.jcb.org/cgi/content/full/jcb.200902093/dc1 S1 Figure S2. Visualization of multiple merotelic attachments.
T H E J O U R N A L O F C E L L B I O L O G Y Supplemental material Courtheoux et al., http://www.jcb.org/cgi/content/full/jcb.200902093/dc1 S1 Figure S2. Visualization of multiple merotelic attachments.
APGRU4L1 Chap 12 Extra Reading Cell Cycle and Mitosis
 APGRU4L1 Chap 12 Extra Reading Cell Cycle and Mitosis Dr. Ramesh Biology is the only subject in which multiplication is the same thing as division 2007-2008 The Cell Cycle: Cell Growth, Cell Division 2007-2008
APGRU4L1 Chap 12 Extra Reading Cell Cycle and Mitosis Dr. Ramesh Biology is the only subject in which multiplication is the same thing as division 2007-2008 The Cell Cycle: Cell Growth, Cell Division 2007-2008
Campbell Biology in Focus (Urry) Chapter 9 The Cell Cycle. 9.1 Multiple-Choice Questions
 Campbell Biology in Focus (Urry) Chapter 9 The Cell Cycle 9.1 Multiple-Choice Questions 1) Starting with a fertilized egg (zygote), a series of five cell divisions would produce an early embryo with how
Campbell Biology in Focus (Urry) Chapter 9 The Cell Cycle 9.1 Multiple-Choice Questions 1) Starting with a fertilized egg (zygote), a series of five cell divisions would produce an early embryo with how
基醫所. The Cell Cycle. Chi-Wu Chiang, Ph.D. IMM, NCKU
 基醫所 The Cell Cycle Chi-Wu Chiang, Ph.D. IMM, NCKU 1 1 Introduction to cell cycle and cell cycle checkpoints 2 2 Cell cycle A cell reproduces by performing an orderly sequence of events in which it duplicates
基醫所 The Cell Cycle Chi-Wu Chiang, Ph.D. IMM, NCKU 1 1 Introduction to cell cycle and cell cycle checkpoints 2 2 Cell cycle A cell reproduces by performing an orderly sequence of events in which it duplicates
Biology is the only subject in which multiplication is the same thing as division
 Biology is the only subject in which multiplication is the same thing as division 2007-2008 The Cell Cycle: Cell Growth, Cell Division 2007-2008 Where it all began You started as a cell smaller than a
Biology is the only subject in which multiplication is the same thing as division 2007-2008 The Cell Cycle: Cell Growth, Cell Division 2007-2008 Where it all began You started as a cell smaller than a
Biology is the only subject in which multiplication is the same thing as division
 Biology is the only subject in which multiplication is the same thing as division 2007-2008 The Cell Cycle: Cell Growth, Cell Division 2007-2008 Getting from there to here Going from egg to baby. the original
Biology is the only subject in which multiplication is the same thing as division 2007-2008 The Cell Cycle: Cell Growth, Cell Division 2007-2008 Getting from there to here Going from egg to baby. the original
SUPPLEMENTARY INFORMATION
 DOI: 10.1038/ncb3076 Supplementary Figure 1 btrcp targets Cep68 for degradation during mitosis. a) Cep68 immunofluorescence in interphase and metaphase. U-2OS cells were transfected with control sirna
DOI: 10.1038/ncb3076 Supplementary Figure 1 btrcp targets Cep68 for degradation during mitosis. a) Cep68 immunofluorescence in interphase and metaphase. U-2OS cells were transfected with control sirna
Mitosis and the Cell Cycle
 Mitosis and the Cell Cycle Chapter 12 The Cell Cycle: Cell Growth & Cell Division Where it all began You started as a cell smaller than a period at the end of a sentence Getting from there to here Cell
Mitosis and the Cell Cycle Chapter 12 The Cell Cycle: Cell Growth & Cell Division Where it all began You started as a cell smaller than a period at the end of a sentence Getting from there to here Cell
Origin of replication. Septum
 Bacterial cell Bacterial chromosome: Double-stranded DNA Origin of replication Septum 1 2 3 Chromosome Rosettes of Chromatin Loops Chromatin Loop Solenoid Scaffold protein Scaffold protein Chromatin loop
Bacterial cell Bacterial chromosome: Double-stranded DNA Origin of replication Septum 1 2 3 Chromosome Rosettes of Chromatin Loops Chromatin Loop Solenoid Scaffold protein Scaffold protein Chromatin loop
Bacterial cell. Origin of replication. Septum
 Bacterial cell Bacterial chromosome: Double-stranded DNA Origin of replication Septum 1 2 3 Chromosome Rosettes of Chromatin Loops Scaffold protein Chromatin Loop Solenoid Scaffold protein Chromatin loop
Bacterial cell Bacterial chromosome: Double-stranded DNA Origin of replication Septum 1 2 3 Chromosome Rosettes of Chromatin Loops Scaffold protein Chromatin Loop Solenoid Scaffold protein Chromatin loop
TITLE: The Role of Oncogene/Tumor Suppressor Interaction with the Centrosome Protein Pericentrin in Prostate Tumorigenesis
 AD Award Number: W81XWH-6-1-14 TITLE: The Role of Oncogene/Tumor Suppressor Interaction with the Centrosome Protein Pericentrin in Prostate Tumorigenesis PRINCIPAL INVESTIGATOR: Chun-Ting Chen, Ph.D. CONTRACTING
AD Award Number: W81XWH-6-1-14 TITLE: The Role of Oncogene/Tumor Suppressor Interaction with the Centrosome Protein Pericentrin in Prostate Tumorigenesis PRINCIPAL INVESTIGATOR: Chun-Ting Chen, Ph.D. CONTRACTING
Chapter 12 The Cell Cycle: Cell Growth, Cell Division
 Chapter 12 The Cell Cycle: Cell Growth, Cell Division 2007-2008 Where it all began You started as a cell smaller than a period at the end of a sentence And now look at you How did you get from there to
Chapter 12 The Cell Cycle: Cell Growth, Cell Division 2007-2008 Where it all began You started as a cell smaller than a period at the end of a sentence And now look at you How did you get from there to
Biology is the only subject in which multiplication is the same thing as division
 Biology is the only subject in which multiplication is the same thing as division 2007-2008 The Cell Cycle: Cell Growth, Cell Division 2007-2008 Where it all began You started as a cell smaller than a
Biology is the only subject in which multiplication is the same thing as division 2007-2008 The Cell Cycle: Cell Growth, Cell Division 2007-2008 Where it all began You started as a cell smaller than a
The Cell Cycle and How Cells Divide
 The Cell Cycle and How Cells Divide 1 Phases of the Cell Cycle The cell cycle consists of Interphase normal cell activity The mitotic phase cell divsion INTERPHASE Growth G 1 (DNA synthesis) Growth G 2
The Cell Cycle and How Cells Divide 1 Phases of the Cell Cycle The cell cycle consists of Interphase normal cell activity The mitotic phase cell divsion INTERPHASE Growth G 1 (DNA synthesis) Growth G 2
BIOLOGY LTF DIAGNOSTIC TEST CELL CYCLE & MITOSIS
 Biology Multiple Choice 016044 BIOLOGY LTF DIAGNOSTIC TEST CELL CYCLE & MITOSIS TEST CODE: 016044 Directions: Each of the questions or incomplete statements below is followed by five suggested answers
Biology Multiple Choice 016044 BIOLOGY LTF DIAGNOSTIC TEST CELL CYCLE & MITOSIS TEST CODE: 016044 Directions: Each of the questions or incomplete statements below is followed by five suggested answers
Chapter 8: Cellular Reproduction
 Chapter 8: Cellular Reproduction 1. The Cell Cycle 2. Mitosis 3. Meiosis 2 Types of Cell Division 2n 1n Mitosis: occurs in somatic cells (almost all cells of the body) generates cells identical to original
Chapter 8: Cellular Reproduction 1. The Cell Cycle 2. Mitosis 3. Meiosis 2 Types of Cell Division 2n 1n Mitosis: occurs in somatic cells (almost all cells of the body) generates cells identical to original
Breaking Up is Hard to Do (At Least in Eukaryotes) Mitosis
 Breaking Up is Hard to Do (At Least in Eukaryotes) Mitosis Chromosomes Chromosomes were first observed by the German embryologist Walther Fleming in 1882. Chromosome number varies among organisms most
Breaking Up is Hard to Do (At Least in Eukaryotes) Mitosis Chromosomes Chromosomes were first observed by the German embryologist Walther Fleming in 1882. Chromosome number varies among organisms most
The subcortical maternal complex controls symmetric division of mouse zygotes by
 The subcortical maternal complex controls symmetric division of mouse zygotes by regulating F-actin dynamics Xing-Jiang Yu 1,2, Zhaohong Yi 1, Zheng Gao 1,2, Dan-dan Qin 1,2, Yanhua Zhai 1, Xue Chen 1,
The subcortical maternal complex controls symmetric division of mouse zygotes by regulating F-actin dynamics Xing-Jiang Yu 1,2, Zhaohong Yi 1, Zheng Gao 1,2, Dan-dan Qin 1,2, Yanhua Zhai 1, Xue Chen 1,
Molecular Cell Biology - Problem Drill 22: The Mechanics of Cell Division
 Molecular Cell Biology - Problem Drill 22: The Mechanics of Cell Division Question No. 1 of 10 1. Which of the following statements about mitosis is correct? Question #1 (A) Mitosis involves the dividing
Molecular Cell Biology - Problem Drill 22: The Mechanics of Cell Division Question No. 1 of 10 1. Which of the following statements about mitosis is correct? Question #1 (A) Mitosis involves the dividing
Breaking Up is Hard to Do (At Least in Eukaryotes) Mitosis
 Breaking Up is Hard to Do (At Least in Eukaryotes) Mitosis Prokaryotes Have a Simpler Cell Cycle Cell division in prokaryotes takes place in two stages, which together make up a simple cell cycle 1. Copy
Breaking Up is Hard to Do (At Least in Eukaryotes) Mitosis Prokaryotes Have a Simpler Cell Cycle Cell division in prokaryotes takes place in two stages, which together make up a simple cell cycle 1. Copy
Cellular Reproduction, Part 1: Mitosis Lecture 10 Fall 2008
 Cell Theory 1 Cellular Reproduction, Part 1: Mitosis Lecture 10 Fall 2008 Cell theory: All organisms are made of cells All cells arise from preexisting cells How do new cells arise? Cell division the reproduction
Cell Theory 1 Cellular Reproduction, Part 1: Mitosis Lecture 10 Fall 2008 Cell theory: All organisms are made of cells All cells arise from preexisting cells How do new cells arise? Cell division the reproduction
Supplementary Figure 1. Mother centrioles can reduplicate while in the close association
 C1-GFP distance (nm) C1-GFP distance (nm) a arrested HeLa cell expressing C1-GFP and Plk1TD-RFP -3 s 1 2 3 4 5 6 7 8 9 11 12 13 14 16 17 18 19 2 21 22 23 24 26 27 28 29 3 b 9 8 7 6 5 4 3 2 arrested HeLa
C1-GFP distance (nm) C1-GFP distance (nm) a arrested HeLa cell expressing C1-GFP and Plk1TD-RFP -3 s 1 2 3 4 5 6 7 8 9 11 12 13 14 16 17 18 19 2 21 22 23 24 26 27 28 29 3 b 9 8 7 6 5 4 3 2 arrested HeLa
Microtubule-based Cancer Therapeutics BIOS-010 September 12, 2018
 Microtubule-based Cancer Therapeutics 1 BIOS-010 September 12, 2018 Jessica C. Leung PhD Candidate Cassimeris Lab What are microtubules? Why target them in cancer therapy? How do microtubule-based cancer
Microtubule-based Cancer Therapeutics 1 BIOS-010 September 12, 2018 Jessica C. Leung PhD Candidate Cassimeris Lab What are microtubules? Why target them in cancer therapy? How do microtubule-based cancer
meiosis asexual reproduction CHAPTER 9 & 10 The Cell Cycle, Meiosis & Sexual Life Cycles Sexual reproduction mitosis
 meiosis asexual reproduction CHAPTER 9 & 10 The Cell Cycle, Meiosis & Sexual Sexual reproduction Life Cycles mitosis Chromosomes Consists of a long DNA molecule (represents thousands of genes) Also consists
meiosis asexual reproduction CHAPTER 9 & 10 The Cell Cycle, Meiosis & Sexual Sexual reproduction Life Cycles mitosis Chromosomes Consists of a long DNA molecule (represents thousands of genes) Also consists
10.2 The Cell Cycle *
 OpenStax-CNX module: m52672 1 10.2 The Cell Cycle * Shannon McDermott Based on The Cell Cycle by OpenStax This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License
OpenStax-CNX module: m52672 1 10.2 The Cell Cycle * Shannon McDermott Based on The Cell Cycle by OpenStax This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License
Mitosis. AND Cell DiVISION
 Mitosis AND Cell DiVISION Cell Division Characteristic of living things: ability to reproduce their own kind. Cell division purpose: When unicellular organisms such as amoeba divide to form offspring reproduction
Mitosis AND Cell DiVISION Cell Division Characteristic of living things: ability to reproduce their own kind. Cell division purpose: When unicellular organisms such as amoeba divide to form offspring reproduction
supplementary information
 DOI: 10.1038/ncb2133 Figure S1 Actomyosin organisation in human squamous cell carcinoma. (a) Three examples of actomyosin organisation around the edges of squamous cell carcinoma biopsies are shown. Myosin
DOI: 10.1038/ncb2133 Figure S1 Actomyosin organisation in human squamous cell carcinoma. (a) Three examples of actomyosin organisation around the edges of squamous cell carcinoma biopsies are shown. Myosin
Biology is the only subject in which multiplication is the same thing as division. AP Biology
 Biology is the only subject in which multiplication is the same thing as division Chapter 12. The Cell Cycle: Cell Growth, Cell Division Where it all began You started as a cell smaller than a period at
Biology is the only subject in which multiplication is the same thing as division Chapter 12. The Cell Cycle: Cell Growth, Cell Division Where it all began You started as a cell smaller than a period at
Chapter 12. The Cell Cycle
 Chapter 12 The Cell Cycle The Key Roles of Cell Division The ability of organisms to produce more of their own kind is the one characteristic that best distinguishes living things from nonliving things.
Chapter 12 The Cell Cycle The Key Roles of Cell Division The ability of organisms to produce more of their own kind is the one characteristic that best distinguishes living things from nonliving things.
SUPPLEMENTARY INFORMATION
 DOI: 0.038/ncb33 a b c 0 min 6 min 7 min (fixed) DIC -GFP, CenpF 3 µm Nocodazole Single optical plane -GFP, CenpF Max. intensity projection d µm -GFP, CenpF, -GFP CenpF 3-D rendering e f 0 min 4 min 0
DOI: 0.038/ncb33 a b c 0 min 6 min 7 min (fixed) DIC -GFP, CenpF 3 µm Nocodazole Single optical plane -GFP, CenpF Max. intensity projection d µm -GFP, CenpF, -GFP CenpF 3-D rendering e f 0 min 4 min 0
The Cell Cycle CHAPTER 12
 The Cell Cycle CHAPTER 12 The Key Roles of Cell Division cell division = reproduction of cells All cells come from pre-exisiting cells Omnis cellula e cellula Unicellular organisms division of 1 cell reproduces
The Cell Cycle CHAPTER 12 The Key Roles of Cell Division cell division = reproduction of cells All cells come from pre-exisiting cells Omnis cellula e cellula Unicellular organisms division of 1 cell reproduces
Mitosis vs. microtubule
 Mitosis vs. microtubule Anaphase-promoting complex/cyclosome (APC/C) Duplicated centrosomes align and begin separating in prophase Relation of centrosome duplication to the cell cycle. Parent centrioles
Mitosis vs. microtubule Anaphase-promoting complex/cyclosome (APC/C) Duplicated centrosomes align and begin separating in prophase Relation of centrosome duplication to the cell cycle. Parent centrioles
 a Control IgG Intestine c Testis b Thymus 1 3 2 S S 2 1 3 4 4 Figure S1 The wild-type mouse (C57BL/6J) organs (intestine, thymus and testis) were frozen in liquid nitrogen and sectioned at 5 µm on a cryostat.
a Control IgG Intestine c Testis b Thymus 1 3 2 S S 2 1 3 4 4 Figure S1 The wild-type mouse (C57BL/6J) organs (intestine, thymus and testis) were frozen in liquid nitrogen and sectioned at 5 µm on a cryostat.
University of Bristol - Explore Bristol Research
 Shandilya, J., Medler, K., & Roberts, S. G. E. (2016). Regulation of AURORA B function by mitotic checkpoint protein MAD2. Cell Cycle, 15(16), 2196-2201. https://doi.org/10.1080/15384101.2016.1200773 Peer
Shandilya, J., Medler, K., & Roberts, S. G. E. (2016). Regulation of AURORA B function by mitotic checkpoint protein MAD2. Cell Cycle, 15(16), 2196-2201. https://doi.org/10.1080/15384101.2016.1200773 Peer
10-2 Cell Division. Chromosomes
 Cell Division In eukaryotes, cell division occurs in two major stages. The first stage, division of the cell nucleus, is called mitosis. The second stage, division of the cell cytoplasm, is called cytokinesis.
Cell Division In eukaryotes, cell division occurs in two major stages. The first stage, division of the cell nucleus, is called mitosis. The second stage, division of the cell cytoplasm, is called cytokinesis.
Chapter 12. living /non-living? growth repair renew. Reproduction. Reproduction. living /non-living. fertilized egg (zygote) next chapter
 Chapter 12 How cells divide Reproduction living /non-living? growth repair renew based on cell division first mitosis - distributes identical sets of chromosomes cell cycle (life) Cell Division in Bacteria
Chapter 12 How cells divide Reproduction living /non-living? growth repair renew based on cell division first mitosis - distributes identical sets of chromosomes cell cycle (life) Cell Division in Bacteria
Cell Cycle and Mitosis
 Cell Cycle and Mitosis Name Period A# THE CELL CYCLE The cell cycle, or cell-division cycle, is the series of events that take place in a eukaryotic cell between its formation and the moment it replicates
Cell Cycle and Mitosis Name Period A# THE CELL CYCLE The cell cycle, or cell-division cycle, is the series of events that take place in a eukaryotic cell between its formation and the moment it replicates
CELL CYCLE INTRODUCTION PART I ANIMAL CELL CYCLE INTERPHASE EVOLUTION/HEREDITY UNIT. Activity #3
 AP BIOLOGY EVOLUTION/HEREDITY UNIT Unit 1 Part 3 Chapter 12 Activity #3 INTRODUCTION CELL CYCLE NAME DATE PERIOD The nuclei in cells of eukaryotic organisms contain chromosomes with clusters of genes,
AP BIOLOGY EVOLUTION/HEREDITY UNIT Unit 1 Part 3 Chapter 12 Activity #3 INTRODUCTION CELL CYCLE NAME DATE PERIOD The nuclei in cells of eukaryotic organisms contain chromosomes with clusters of genes,
Why do cells divide? Cells divide in order to make more cells they multiply in order to create a larger surface to volume ratio!!!
 Why do cells divide? Cells divide in order to make more cells they multiply in order to create a larger surface to volume ratio!!! Chromosomes Are made of chromatin: a mass of genetic material composed
Why do cells divide? Cells divide in order to make more cells they multiply in order to create a larger surface to volume ratio!!! Chromosomes Are made of chromatin: a mass of genetic material composed
Cell Division. Learning Objectives: Introduction. Revised Fall 2018
 Revised Fall 2018 Cell Division Learning Objectives: 1. Define cell cycle and the ordered sequence of events in the cell cycle (Interphase and The divisional phase or M phase) 2. Explain the stages in
Revised Fall 2018 Cell Division Learning Objectives: 1. Define cell cycle and the ordered sequence of events in the cell cycle (Interphase and The divisional phase or M phase) 2. Explain the stages in
MITOSIS IN ONION ROOTLET CELLS
 Lesson 6: CELL CYCLE, MITOSIS Name: Group: MITOSIS IN ONION ROOTLET CELLS Permanent slide: onion rootlet stained with acetorcein The particular mitotic phases are visible in the onion rootlet cells. Chromosomes
Lesson 6: CELL CYCLE, MITOSIS Name: Group: MITOSIS IN ONION ROOTLET CELLS Permanent slide: onion rootlet stained with acetorcein The particular mitotic phases are visible in the onion rootlet cells. Chromosomes
(a) Reproduction. (b) Growth and development. (c) Tissue renewal
 100 µm 200 µm 20 µm (a) Reproduction (b) Growth and development (c) Tissue renewal 1 20 µm 2 0.5 µm Chromosomes DNA molecules Chromosome arm Centromere Chromosome duplication (including DNA synthesis)
100 µm 200 µm 20 µm (a) Reproduction (b) Growth and development (c) Tissue renewal 1 20 µm 2 0.5 µm Chromosomes DNA molecules Chromosome arm Centromere Chromosome duplication (including DNA synthesis)
DAPI ASY1 DAPI/ASY1 DAPI RAD51 DAPI/RAD51. Supplementary Figure 1. Additional information on meiosis in R. pubera. a) The
 a % 10 Number of crossover per bivalent b 0 1 c DAPI/telomere 80 1 60 40 1 2 20 d 0 0 1 2 >=3 DAPI ASY1 DAPI/ASY1 e DAPI RAD51 DAPI/RAD51 Supplementary Figure 1. Additional information on meiosis in R.
a % 10 Number of crossover per bivalent b 0 1 c DAPI/telomere 80 1 60 40 1 2 20 d 0 0 1 2 >=3 DAPI ASY1 DAPI/ASY1 e DAPI RAD51 DAPI/RAD51 Supplementary Figure 1. Additional information on meiosis in R.
Chapter 10. Cell Growth and Division
 Chapter 10 Cell Growth and Division Cell Growth A. Limits to Cell Growth 1. Two main reasons why cells divide: a. Demands on DNA as the cell get too large Cell Growth b. Moving nutrients and waste across
Chapter 10 Cell Growth and Division Cell Growth A. Limits to Cell Growth 1. Two main reasons why cells divide: a. Demands on DNA as the cell get too large Cell Growth b. Moving nutrients and waste across
Supplementary table 1
 Supplementary table 1 S. pombe strain list Fig. 1A JX38 h + ade6-m216 nda3-km311 PX476 PW775 PX545 PX546 h- ade6-m216 sgo2::ura4 + nda3-km311 h 9 mad2::ura4 + nda3-km311 h + ade6-m21 nda3-km311 rad21 +
Supplementary table 1 S. pombe strain list Fig. 1A JX38 h + ade6-m216 nda3-km311 PX476 PW775 PX545 PX546 h- ade6-m216 sgo2::ura4 + nda3-km311 h 9 mad2::ura4 + nda3-km311 h + ade6-m21 nda3-km311 rad21 +
SLX4 + MUS81 SLX4 + GEN1 SLX4 CONTROL SLX4
 GEN MUS8 GEN MUS8 GEN MUS8 GEN MUS8 GEN C LM MUS8 XPF (loading control) D H2AX Frequency of -positive bridges (% of anaphase cells) 6 4 2 p =.8 x -4 GM855 p =.27 PSNF5 E H2AX Figure S. Analysis of anaphase
GEN MUS8 GEN MUS8 GEN MUS8 GEN MUS8 GEN C LM MUS8 XPF (loading control) D H2AX Frequency of -positive bridges (% of anaphase cells) 6 4 2 p =.8 x -4 GM855 p =.27 PSNF5 E H2AX Figure S. Analysis of anaphase
Figure S1. HP1α localizes to centromeres in mitosis and interacts with INCENP. (A&B) HeLa
 SUPPLEMENTARY FIGURES Figure S1. HP1α localizes to centromeres in mitosis and interacts with INCENP. (A&B) HeLa tet-on cells that stably express HP1α-CFP, HP1β-CFP, or HP1γ-CFP were monitored with livecell
SUPPLEMENTARY FIGURES Figure S1. HP1α localizes to centromeres in mitosis and interacts with INCENP. (A&B) HeLa tet-on cells that stably express HP1α-CFP, HP1β-CFP, or HP1γ-CFP were monitored with livecell
Mitosis THE CELL CYCLE. In unicellular organisms, division of one cell reproduces the entire organism Multicellular organisms use cell division for..
 Mitosis THE CELL CYCLE In unicellular organisms, division of one cell reproduces the entire organism Multicellular organisms use cell division for.. Development from a fertilized cell Growth Repair Cell
Mitosis THE CELL CYCLE In unicellular organisms, division of one cell reproduces the entire organism Multicellular organisms use cell division for.. Development from a fertilized cell Growth Repair Cell
General Embryology. School of Medicine Department of Anatomy and Histology School of medicine The University of Jordan
 General Embryology 2019 School of Medicine Department of Anatomy and Histology School of medicine The University of Jordan https://www.facebook.com/dramjad-shatarat What is embryology? Is the science that
General Embryology 2019 School of Medicine Department of Anatomy and Histology School of medicine The University of Jordan https://www.facebook.com/dramjad-shatarat What is embryology? Is the science that
Chapter 2. Mitosis and Meiosis
 Chapter 2. Mitosis and Meiosis Chromosome Theory of Heredity What structures within cells correspond to genes? The development of genetics took a major step forward by accepting the notion that the genes
Chapter 2. Mitosis and Meiosis Chromosome Theory of Heredity What structures within cells correspond to genes? The development of genetics took a major step forward by accepting the notion that the genes
T H E J O U R N A L O F C E L L B I O L O G Y
 Supplemental material Edens and Levy, http://www.jcb.org/cgi/content/full/jcb.201406004/dc1 T H E J O U R N A L O F C E L L B I O L O G Y Figure S1. Nuclear shrinking does not depend on the cytoskeleton
Supplemental material Edens and Levy, http://www.jcb.org/cgi/content/full/jcb.201406004/dc1 T H E J O U R N A L O F C E L L B I O L O G Y Figure S1. Nuclear shrinking does not depend on the cytoskeleton
9 The Cell Cycle CAMPBELL BIOLOGY IN FOCUS. Urry Cain Wasserman Minorsky Jackson Reece
 CAMPBELL BIOLOGY IN FOCUS Urry Cain Wasserman Minorsky Jackson Reece 9 The Cell Cycle Lecture Presentations by Kathleen Fitzpatrick and Nicole Tunbridge 2014 Pearson Education, Inc. Cell division plays
CAMPBELL BIOLOGY IN FOCUS Urry Cain Wasserman Minorsky Jackson Reece 9 The Cell Cycle Lecture Presentations by Kathleen Fitzpatrick and Nicole Tunbridge 2014 Pearson Education, Inc. Cell division plays
MITOSIS AND THE CELL CYCLE PowerPoint Notes
 1 Name: Date: MITOSIS AND THE CELL CYCLE PowerPoint Notes THE FUNCTIONS OF CELL DIVISION 1. Cell division is vital for all. living organisms This is the only process that can create. new cells 2. Cell
1 Name: Date: MITOSIS AND THE CELL CYCLE PowerPoint Notes THE FUNCTIONS OF CELL DIVISION 1. Cell division is vital for all. living organisms This is the only process that can create. new cells 2. Cell
Karen L.P. McNally, Amy S. Fabritius, Marina L. Ellefson, Jonathan R. Flynn, Jennifer A. Milan, and Francis J. McNally
 Developmental Cell, Volume 22 Supplemental Information Kinesin-1 Prevents Capture of the Oocyte Meiotic Spindle by the Sperm Aster Karen L.P. McNally, Amy S. Fabritius, Marina L. Ellefson, Jonathan R.
Developmental Cell, Volume 22 Supplemental Information Kinesin-1 Prevents Capture of the Oocyte Meiotic Spindle by the Sperm Aster Karen L.P. McNally, Amy S. Fabritius, Marina L. Ellefson, Jonathan R.
T H E J O U R N A L O F C E L L B I O L O G Y
 T H E J O U R N A L O F C E L L B I O L O G Y Supplemental material Posch et al., http://www.jcb.org/cgi/content/full/jcb.200912046/dc1 Figure S1. Biochemical characterization of the interaction between
T H E J O U R N A L O F C E L L B I O L O G Y Supplemental material Posch et al., http://www.jcb.org/cgi/content/full/jcb.200912046/dc1 Figure S1. Biochemical characterization of the interaction between
2014 Pearson Education, Inc.
 2 The Cell Cycle CAMPBELL BIOLOGY TENTH EDITION Reece Urry Cain Wasserman Minorsky Jackson The Key Roles of Cell Division The ability of organisms to produce more of their own kind best distinguishes living
2 The Cell Cycle CAMPBELL BIOLOGY TENTH EDITION Reece Urry Cain Wasserman Minorsky Jackson The Key Roles of Cell Division The ability of organisms to produce more of their own kind best distinguishes living
scientificreport The PITSLRE/CDK11 p58 protein kinase promotes centrosome maturation and bipolar spindle formation scientific report
 scientificreport The PITSLRE/ p58 protein kinase promotes centrosome maturation and bipolar spindle formation Clotilde Petretti 1, Matthew Savoian 2, Emilie Montembault 1,DavidM.Glover 2,ClaudePrigent
scientificreport The PITSLRE/ p58 protein kinase promotes centrosome maturation and bipolar spindle formation Clotilde Petretti 1, Matthew Savoian 2, Emilie Montembault 1,DavidM.Glover 2,ClaudePrigent
The questions below refer to the following terms. Each term may be used once, more than once, or not at all.
 The questions below refer to the following terms. Each term may be used once, more than once, or not at all. a) telophase b) anaphase c) prometaphase d) metaphase e) prophase 1) DNA begins to coil and
The questions below refer to the following terms. Each term may be used once, more than once, or not at all. a) telophase b) anaphase c) prometaphase d) metaphase e) prophase 1) DNA begins to coil and
10-2 Cell Division mitosis. cytokinesis. Chromosomes chromosomes Slide 1 of 38
 In eukaryotes, cell division occurs in two major stages. The first stage, division of the cell nucleus, is called mitosis. The second stage, division of the cell cytoplasm, is called cytokinesis. Chromosomes
In eukaryotes, cell division occurs in two major stages. The first stage, division of the cell nucleus, is called mitosis. The second stage, division of the cell cytoplasm, is called cytokinesis. Chromosomes
How Cells Divide. Chapter 10
 How Cells Divide Chapter 10 Bacterial Cell Division Bacteria divide by binary fission. -the single, circular bacterial chromosome is replicated -replication begins at the origin of replication and proceeds
How Cells Divide Chapter 10 Bacterial Cell Division Bacteria divide by binary fission. -the single, circular bacterial chromosome is replicated -replication begins at the origin of replication and proceeds
Biology is the only subject in which multiplication is the same thing as division
 The Cell Cycle Biology is the only subject in which multiplication is the same thing as division Why do cells divide? For reproduction asexual reproduction For growth one-celled organisms from fertilized
The Cell Cycle Biology is the only subject in which multiplication is the same thing as division Why do cells divide? For reproduction asexual reproduction For growth one-celled organisms from fertilized
The TACC proteins: TACC-ling microtubule dynamics and centrosome
 Review The TACC proteins: TACC-ling microtubule dynamics and centrosome function Isabel Peset 1 and Isabelle Vernos 1,2 1 Cell and Developmental Biology Program, Centre for Genomic Regulation (CRG), University
Review The TACC proteins: TACC-ling microtubule dynamics and centrosome function Isabel Peset 1 and Isabelle Vernos 1,2 1 Cell and Developmental Biology Program, Centre for Genomic Regulation (CRG), University
General Biology. Overview: The Key Roles of Cell Division. Unicellular organisms
 General Biology Course No: BNG2003 Credits: 3.00 8. The Cell Cycle Prof. Dr. Klaus Heese Overview: The Key Roles of Cell Division The continuity of life is based upon the reproduction of cells, or cell
General Biology Course No: BNG2003 Credits: 3.00 8. The Cell Cycle Prof. Dr. Klaus Heese Overview: The Key Roles of Cell Division The continuity of life is based upon the reproduction of cells, or cell
General Biology. Overview: The Key Roles of Cell Division The continuity of life is based upon the reproduction of cells, or cell division
 General Biology Course No: BNG2003" Credits: 3.00 " " " 8. The Cell Cycle Prof. Dr. Klaus Heese Overview: The Key Roles of Cell Division The continuity of life is based upon the reproduction of cells,
General Biology Course No: BNG2003" Credits: 3.00 " " " 8. The Cell Cycle Prof. Dr. Klaus Heese Overview: The Key Roles of Cell Division The continuity of life is based upon the reproduction of cells,
5.1. KEY CONCEPT Cells have distinct phases of growth, reproduction, and normal functions. 68 Reinforcement Unit 2 Resource Book
 5.1 THE CELL CYCLE KEY CONCEPT Cells have distinct phases of growth, reproduction, and normal functions. Cells have a regular pattern of growth, DNA duplication, and division that is called the cell cycle.
5.1 THE CELL CYCLE KEY CONCEPT Cells have distinct phases of growth, reproduction, and normal functions. Cells have a regular pattern of growth, DNA duplication, and division that is called the cell cycle.
BIOLOGY 4/6/2015. Cell Cycle - Mitosis. Outline. Overview: The Key Roles of Cell Division. identical daughter cells. I. Overview II.
 2 Cell Cycle - Mitosis CAMPBELL BIOLOGY TENTH EDITION Reece Urry Cain Wasserman Minorsky Jackson Outline I. Overview II. Mitotic Phase I. Prophase II. III. Telophase IV. Cytokinesis III. Binary fission
2 Cell Cycle - Mitosis CAMPBELL BIOLOGY TENTH EDITION Reece Urry Cain Wasserman Minorsky Jackson Outline I. Overview II. Mitotic Phase I. Prophase II. III. Telophase IV. Cytokinesis III. Binary fission
CH 9: The Cell Cycle Overview. Cellular Organization of the Genetic Material. Distribution of Chromosomes During Eukaryotic Cell Division
 CH 9: The Cell Cycle Overview The ability of organisms to produce more of their own kind best distinguishes living things from nonliving matter The continuity of life is based on the reproduction of cells,
CH 9: The Cell Cycle Overview The ability of organisms to produce more of their own kind best distinguishes living things from nonliving matter The continuity of life is based on the reproduction of cells,
Mitosis/Meiosis Simulation Activities
 Mitosis/Meiosis Simulation Activities In this simulation, you will demonstrate an understanding of mitosis, meiosis, segregation, independent assortment, and crossing over, all processes involved with
Mitosis/Meiosis Simulation Activities In this simulation, you will demonstrate an understanding of mitosis, meiosis, segregation, independent assortment, and crossing over, all processes involved with
Cell Division Questions. Mitosis and Meiosis
 Cell Division Questions Mitosis and Meiosis 1 10 Do not write outside the box 5 Figure 3 shows a pair of chromosomes at the start of meiosis. The letters represent alleles. Figure 3 E E e e F F f f 5 (a)
Cell Division Questions Mitosis and Meiosis 1 10 Do not write outside the box 5 Figure 3 shows a pair of chromosomes at the start of meiosis. The letters represent alleles. Figure 3 E E e e F F f f 5 (a)
Chromosomes Can Congress To The Metaphase Plate Prior. To Bi-Orientation
 Chromosomes Can Congress To The Metaphase Plate Prior To Bi-Orientation Tarun M. Kapoor 1,2, Michael Lampson 1, Polla Hergert 3, Lisa Cameron 2,4, Daniela Cimini 4, E.D. Salmon 2,4, Bruce F. McEwen 3,
Chromosomes Can Congress To The Metaphase Plate Prior To Bi-Orientation Tarun M. Kapoor 1,2, Michael Lampson 1, Polla Hergert 3, Lisa Cameron 2,4, Daniela Cimini 4, E.D. Salmon 2,4, Bruce F. McEwen 3,
Ploidy and Human Cell Types. Cell Cycle and Mitosis. DNA and Chromosomes. Where It All Began 11/19/2014. Chapter 12 Pg
 Ploidy and Human Cell Types Cell Cycle and Mitosis Chapter 12 Pg. 228 245 Cell Types Somatic cells (body cells) have 46 chromosomes, which is the diploid chromosome number. A diploid cell is a cell with
Ploidy and Human Cell Types Cell Cycle and Mitosis Chapter 12 Pg. 228 245 Cell Types Somatic cells (body cells) have 46 chromosomes, which is the diploid chromosome number. A diploid cell is a cell with
Prentice Hall Biology Slide 1 of 38
 Prentice Hall Biology 1 of 38 2 of 38 In eukaryotes, cell division occurs in two major stages. The first stage, division of the cell nucleus, is called mitosis. The second stage, division of the cell cytoplasm,
Prentice Hall Biology 1 of 38 2 of 38 In eukaryotes, cell division occurs in two major stages. The first stage, division of the cell nucleus, is called mitosis. The second stage, division of the cell cytoplasm,
Why do cells divide? The Cell Cycle: Cell Growth, Cell Division. Making new cells. Getting the right stuff. Overview of mitosis 1/5/2015
 Why do cells divide? The Cell Cycle: Cell Growth, Cell Division For reproduction asexual reproduction one-celled organisms For growth from fertilized egg to multi-celled organism For repair & renewal replace
Why do cells divide? The Cell Cycle: Cell Growth, Cell Division For reproduction asexual reproduction one-celled organisms For growth from fertilized egg to multi-celled organism For repair & renewal replace
Bis2A 15.0 The Cell Cycle *
 OpenStax-CNX module: m56084 1 Bis2A 15.0 The Cell Cycle * Mitch Singer Based on The Cell Cycle by OpenStax This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License
OpenStax-CNX module: m56084 1 Bis2A 15.0 The Cell Cycle * Mitch Singer Based on The Cell Cycle by OpenStax This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License
Cell Division. During interphase, a cell s DNA is in a loose form called. It condenses into tightly coiled structures called chromosomes during.
 Cell Division The is a cell s total DNA. Prokaryotes DNA is found mostly in a single called the and also in small circles called. Eukaryotes have several DNA double helices packaged into. During interphase,
Cell Division The is a cell s total DNA. Prokaryotes DNA is found mostly in a single called the and also in small circles called. Eukaryotes have several DNA double helices packaged into. During interphase,
10-2 Cell Division. Slide 1 of 38. End Show. Copyright Pearson Prentice Hall
 1 of 38 Cell Division In eukaryotes, cell division occurs in two major stages. The first stage, division of the cell nucleus, is called mitosis. The second stage, division of the cell cytoplasm, is called
1 of 38 Cell Division In eukaryotes, cell division occurs in two major stages. The first stage, division of the cell nucleus, is called mitosis. The second stage, division of the cell cytoplasm, is called
Supporting Information
 Supporting Information ou et al..73/pnas.08791112 dd Thymidine Release & transfection dd Thymidine Release dd MG132 Fix and IF -14 h 0 h 8 h 24 h 34 h 36 h siontrol simps1-1 simps1-1 simps1-1 simps1-2
Supporting Information ou et al..73/pnas.08791112 dd Thymidine Release & transfection dd Thymidine Release dd MG132 Fix and IF -14 h 0 h 8 h 24 h 34 h 36 h siontrol simps1-1 simps1-1 simps1-1 simps1-2
Cell Division Mitosis Notes
 Cell Division Mitosis Notes Cell Division process by which a cell divides into 2 new cells Why do cells need to divide? 1.Living things grow by producing more cells, NOT because each cell increases in
Cell Division Mitosis Notes Cell Division process by which a cell divides into 2 new cells Why do cells need to divide? 1.Living things grow by producing more cells, NOT because each cell increases in
