Breast Reconstruction Surgery after Mastectomy or Lumpectomy
|
|
|
- Hector Moore
- 6 years ago
- Views:
Transcription
1 Breast Reconstruction Surgery after Mastectomy or Lumpectomy Date of Origin: 11/1998 Last Review Date: 11/25/2017 Effective Date: 11/25/2017 Dates Reviewed: 08/2000, 09/2001, 11/2003, 11/2004, 12/2005, 12/2006, 10/2007, 10/2008, 07/2010, 07/2011, 05/2012, 03/2013, 04/2014, 04/2015, 12/2015, 11/2016, 11/2017 Developed By: Medical Necessity Criteria Committee I. Description II. Reconstructive breast surgery is performed following a mastectomy, lumpectomy or prophylactic mastectomy for high-risk patients to re-establish symmetry between the two breasts. It includes reconstruction of the mastectomy site, creation of a new breast mound and creation of a new areolar complex following removal of a breast. It also includes reconstruction of a non-diseased breast to achieve symmetry. III. Criteria: CWQI HCS-0011 A. Moda Health covers breast reconstruction for ALL of the following conditions: a. In accordance with the Women s Health and Cancer Rights Act of 1998 Moda Health covers reconstruction surgery following a mastectomy for the diagnosis of cancer or cancer prophylaxis as medically necessary including 1 or more of the following conditions: i. The original reconstruction of the breast following mastectomy, including the areolar complex, on which the mastectomy was performed for 1 or more of the following: 1. Reconstruction can be performed at any phase or timeframe post mastectomy for commercial and Medicare members 2. Reconstruction must occur within 5 years of mastectomy for Medicaid members (See Oregon Guideline Note 79). ii. The surgery and reconstruction of the opposite breast to achieve a symmetrical appearance iii. Prostheses and physical complications at all stages following the original mastectomy including all lymphedemas iv. Inpatient care related to the mastectomy and post-mastectomy services v. Reconstruction is covered for both females and males following a mastectomy or lumpectomy for breast cancer. b. Surgery for breast reconstruction/augmentation for which medical necessity cannot be established is considered cosmetic and is not covered by the plan (refer to member handbook language for cosmetic exclusions). c. If the breast reconstruction is requested related to gender reassignment, refer to Moda Health Gender Reassignment criteria CWQI: HCS-0145 and Member s Plan Handbook. Moda Health Medical Necessity Criteria Breast Reconstruction after Mastectomy or Lumpectomy Page 1/8
2 IV. Information Submitted with the Prior Authorization Request: 1. Clinical records from the treating physician to include: a. Original diagnosis and date of mastectomy b. Phases of reconstructive surgery and completion dates c. Documentation of pain, contractures, complications or reconstruction, etc. V. CPT or HCPC codes covered: Codes Description Tattooing, intradermal introduction of insoluble opaque pigments to correct color defects of skin, including micropigmentation; 6.0 sq cm or less Tattooing, intradermal introduction of insoluble opaque pigments to correct color defects of skin, including micropigmentation; 6.1 to 20.0 sq cm Replacement of tissue expander with permanent prosthesis Removal of tissue expander(s) without insertion of prosthesis Repair, complex, trunk; 1.1 cm to 2.5 cm Repair, complex, trunk; 2.6 cm to 7.5 cm Repair, complex, trunk; each additional 5 cm or less (List separately in addition to code for primary procedure) Acellular dermal allograft, trunk, arms, legs; first 100 sq cm or less, or 1% of body area of infants and children Acellular dermal allograft, trunk, arms, legs; each additional 100 sq cm, or each additional 1% of body area of infants and children, or part thereof (List separately in addition to code for primary procedure) Acellular xenograft implant; first 100 sq cm or less, or 1% of body area of infants and children Acellular xenograft implant; each additional 100 sq cm, or each additional 1% of body area of infants and children, or part thereof (List separately in addition to code for primary procedure) Implantation of biologic implant (e.g. acellular dermal matrix) for soft tissue reinforcement Mastopexy Reduction mammaplasty Mammaplasty, augmentation; without prosthetic implant Mammaplasty, augmentation; with prosthetic implant Removal of intact mammary implant Removal of mammary implant material Immediate insertion of breast prosthesis following mastopexy, mastectomy or in reconstruction Delayed insertion of breast prosthesis following mastopexy, mastectomy or in reconstruction Nipple/areola reconstruction Breast reconstruction, immediate or delayed, with tissue expander, including subsequent expansion Moda Health Medical Necessity Criteria Breast Reconstruction after Mastectomy or Lumpectomy Page 2/8
3 19361 Breast reconstruction with latissimus dorsi flap, without prosthetic implant Breast reconstruction with free flap Breast reconstruction with other technique Breast reconstruction with transverse rectus abdominis myocutaneous flap (TRAM), single pedicle, including closure of donor site Breast reconstruction with transverse rectus abdominis myocutaneous flap (TRAM), single pedicle, including closure of donor site; with microvascular anastomosis (supercharging) Breast reconstruction with transverse rectus abdominis myocutaneous flap (TRAM), double pedicle, including closure of donor site Open periprosthetic capsulotomy, breast Periprosthetic capsulectomy, breast Revision of reconstructed breast C1789 Prosthesis, breast (implantable) L8600 Implantable breast prosthesis, silicone or equal Q4100 Skin substitute, not otherwise specified Q4116 Skin substitute, alloderm, per square centimeter (Effective 07/01/2009) S2066 Breast reconstruction with gluteal artery perforator (GAP) flap, including harvesting of the flap, microvascular transfer, closure of donor site and shaping the flap into a breast, unilateral S2067 Breast reconstruction of a single breast with "stacked" deep inferior epigastric perforator (DIEP) flap(s) and/or gluteal artery perforator (GAP) flap(s), including harvesting of the flap(s), microvascular transfer, closure of donor site(s) and shaping the flap into a breast, unilateral S2068 Breast reconstruction with deep inferior epigastric perforator (DIEP) flap or superficial inferior epigastric artery (SIEA) flap, including harvesting of the flap, microvascular transfer, closure of donor site and shaping the flap into a breast, unilateral V. Annual Review History Review Date Revisions Effective Date 03/2013 Annual Review: Added table with review date, revisions, 04/03/2013 and effective date. 04/2014 Annual Review: No changes 04/03/ /2015 Annual Review: Added ICD-10 codes, updated references 04/01/ /2016 Annual Review:- removed criteria for additional 11/30/2016 reconstruction 10/2017 Annual Review: Updated to new template, no changes 10/25/2017 Moda Health Medical Necessity Criteria Breast Reconstruction after Mastectomy or Lumpectomy Page 3/8
4 VI. References 1. Alderman AK, Kuzon WM Jr, Wilkins EG. A two-year prospective analysis of trunk function in TRAM breast reconstructions. Plast Reconstr Surg Jun;117(7): American Cancer Society (ACS): Breast reconstruction after mastectomy. Updated September 1, Accessed July 21, 2011 at: tectomy/breast-reconstruction-after-mastectomy-toc 3. American Society of Plastic Surgeons (ASPS). Guiding principals: Fat Transfer/Fat Graft and Fat Injection. Accessed July 21, 2011 at: ples/asps-fat-transfer-graft-guiding-principles.pdf 4. Bajaj AK, Chevray PM, Chang DW. Comparison of donor-site complications and functional outcomes in free muscle-sparing TRAM flap and free DIEP flap breast reconstruction. Plast Reconstr Surg Mar;117(3):737 46; discussion Becker S, Saint-Cyr M, Wong C, Dauwe P, Nagarkar P, Thornton JF, Peng Y. AlloDerm versus DermaMatrix in immediate expander-based breast reconstruction: a preliminary comparison of complication profiles and material compliance. Plast Reconstr Surg Jan;123(1):1-6; discussion Centers for Medicare & Medicaid Services (CMS). The Women s Health and Cancer Rights Act. Accessed July 21, 2011 at: 7. Chan CW, McCulley SJ, Macmillan RD. Autologous fat transfer--a review of the literature with a focus on breast cancer surgery. J Plast Reconstr Aesthet Surg Dec;61(12): Epub 2008 Oct 9. Review. 8. Chevray PM. Breast reconstruction with superficial inferior epigastric artery flaps: a prospective comparison with TRAM and DIEP flaps. Plast Reconstr Surg Oct;114(5): ; discussion Deutsch MF, Robb GL, Talavera F, Shenaq SM, Slenkovich N, Aly A. Breast Reconstruction, Other Free Flaps. Updated October 16, Accessed July 22, Available at URL address: Deutsch MF, Robb GL, Talavera F, Shenaq SM, Slenkovich N, Aly A. Breast Reconstruction, Refinements & Finishing Touches. Updated October 16, Accessed July 21, 2011 at: Diana M, Robb GL, Talavera F, Shenaq SM, Slenkovich N. Breast Reconstruction, Nipple-Areola Reconstruction. Updated August 20, Accessed July 21, 2011 at: Edlich RF, Winters KL, Faulkner BC, et al. Advances in breast reconstruction after mastectomy. J Long Term Eff Med Implants. 2005;15(2): ERISA, 1974: Title IX Women s Health and Cancer Rights Act, Onmibus Appropriations 14. Fischbacher C. Immediate versus delayed breast reconstruction. STEER: Succint and Timely Evaluated Evidence Reviews. Bazian, Ltd., eds. London, UK: Wessex Institute for Health Research and Development, University of Southampton; 2002; 2(17): Hu E, Alderman AK. Breast reconstruction. Surg Clin North Am Apr;87(2):453-67, x. 16. Hyakusoku H, Ogawa R, Ono S, et al. Complications after autologous fat injection to the breast. Plast Reconstr Surg. 2009;123(1): ; discussion Moda Health Medical Necessity Criteria Breast Reconstruction after Mastectomy or Lumpectomy Page 4/8
5 17. Javaid M, Song F, Leinster S, et al. Radiation effects on the cosmetic outcomes of immediate and delayed autologous breast reconstruction: An argument about timing. J Plast Reconstr Aesthet Surg. 2006;59(1): Losken A, Carolson GW, Bostwick J III, Jones GE, Culbertson JH, Schoemann M. Trends in unilateral breast reconstruction and management of the contralateral breast: the Emory experience. Plast Reconstr Surg Jul;110(1): Losken A, Hamdi M. Partial breast reconstruction: Current perspectives. Plast Reconstr Surg. 2009;124(3): Losken A, Hamdi M. Partial breast reconstruction: Current perspectives. Plast Reconstr Surg. 2009;124(3): Mizuno H, Hyakusoku H. Fat grafting to the breast and adipose-derived stem cells: Recent scientific consensus and controversy. Aesthet Surg J. 2010;30(3): National Comprehensive Cancer Network (NCCN). NCCN practice guidelines in oncology. Breast Cancer. V Accessed July 212, 2011 at: National Library of Medicine. Breast Reconstruction. Reviewed April 7, Accessed July 22, Available at URL address: ORA: JL E 59E, Department of Health & Human Services, Clarification of the Women s Health and Cancer Right Act per Richard Brummel, Deputy Regional Administrator 25. U.S. Department of Labor (DOL). Your Rights After A Mastectomy. Women's Health & Cancer Rights Act of Accessed July 21, 2011 at: U.S. Food and Drug Administration (FDA). Breast implants. Updated 2005 Dec. Accessed July 21, 2011 at: Physician Advisors Appendix 1 Applicable Diagnosis Codes: Codes Description C Malignant neoplasm of nipple and areola, right female breast C Malignant neoplasm of nipple and areola, left female breast C Malignant neoplasm of nipple and areola, unspecified female breast C Malignant neoplasm of central portion of right female breast C Malignant neoplasm of central portion of left female breast C Malignant neoplasm of central portion of unspecified female breast C Malignant neoplasm of upper-inner quadrant of right female breast C Malignant neoplasm of upper-inner quadrant of left female breast C Malignant neoplasm of upper-inner quadrant of unspecified female breast C Malignant neoplasm of lower-inner quadrant of right female breast C Malignant neoplasm of lower-inner quadrant of left female breast C Malignant neoplasm of lower-inner quadrant of unspecified female breast Moda Health Medical Necessity Criteria Breast Reconstruction after Mastectomy or Lumpectomy Page 5/8
6 C Malignant neoplasm of upper-outer quadrant of right female breast C Malignant neoplasm of upper-outer quadrant of left female breast C Malignant neoplasm of upper-outer quadrant of unspecified female breast C Malignant neoplasm of lower-outer quadrant of right female breast C Malignant neoplasm of lower-outer quadrant of left female breast C Malignant neoplasm of lower-outer quadrant of unspecified female breast C Malignant neoplasm of axillary tail of right female breast C Malignant neoplasm of axillary tail of left female breast C Malignant neoplasm of axillary tail of unspecified female breast C Malignant neoplasm of overlapping sites of right female breast C Malignant neoplasm of overlapping sites of left female breast C Malignant neoplasm of overlapping sites of unspecified female breast C Malignant neoplasm of unspecified site of right female breast C Malignant neoplasm of unspecified site of left female breast C Malignant neoplasm of unspecified site of unspecified female breast C Malignant neoplasm of nipple and areola, right male breast C Malignant neoplasm of nipple and areola, left male breast C Malignant neoplasm of nipple and areola, unspecified male breast C Malignant neoplasm of central portion of right male breast C Malignant neoplasm of central portion of left male breast C Malignant neoplasm of central portion of unspecified male breast C Malignant neoplasm of upper-inner quadrant of right male breast C Malignant neoplasm of upper-inner quadrant of left male breast C Malignant neoplasm of upper-inner quadrant of unspecified male breast C Malignant neoplasm of lower-inner quadrant of right male breast C Malignant neoplasm of lower-inner quadrant of left male breast C Malignant neoplasm of lower-inner quadrant of unspecified male breast C Malignant neoplasm of upper-outer quadrant of right male breast C Malignant neoplasm of upper-outer quadrant of left male breast C Malignant neoplasm of upper-outer quadrant of unspecified male breast C Malignant neoplasm of lower-outer quadrant of right male breast C Malignant neoplasm of lower-outer quadrant of left male breast C Malignant neoplasm of lower-outer quadrant of unspecified male breast C Malignant neoplasm of axillary tail of right male breast C Malignant neoplasm of axillary tail of left male breast C Malignant neoplasm of axillary tail of unspecified male breast C Malignant neoplasm of overlapping sites of right male breast C Malignant neoplasm of overlapping sites of left male breast C Malignant neoplasm of overlapping sites of unspecified male breast C Malignant neoplasm of unspecified site of right male breast C Malignant neoplasm of unspecified site of left male breast C Malignant neoplasm of unspecified site of unspecified male breast C79.81 Secondary malignant neoplasm of breast D05.00 Lobular carcinoma in situ of unspecified breast Moda Health Medical Necessity Criteria Breast Reconstruction after Mastectomy or Lumpectomy Page 6/8
7 D05.01 Lobular carcinoma in situ of right breast D05.02 Lobular carcinoma in situ of left breast D05.10 Intraductal carcinoma in situ of unspecified breast D05.11 Intraductal carcinoma in situ of right breast D05.12 Intraductal carcinoma in situ of left breast D05.80 Other specified type of carcinoma in situ of unspecified breast D05.82 Other specified type of carcinoma in situ of left breast D05.90 Unspecified type of carcinoma in situ of unspecified breast D05.91 Unspecified type of carcinoma in situ of right breast D05.92 Unspecified type of carcinoma in situ of left breast N60.11 Diffuse cystic mastopathy of right breast N60.12 Diffuse cystic mastopathy of left breast N60.19 Diffuse cystic mastopathy of unspecified breast N64.81 Ptosis of breast N64.82 Hypoplasia of breast N64.89 Other specified disorders of breast M95.4 Acquired deformity of chest and rib M99.82 Other biomechanical lesions of thoracic region M99.88 Other biomechanical lesions of rib cage Q67.6 Pectus excavatum Q76.6 Other congenital malformations of ribs Q76.7 Congenital malformation of sternum Q76.8 Other congenital malformations of bony thorax Q76.9 Congenital malformation of bony thorax, unspecified Q79.8 Other congenital malformations of musculoskeletal system Q83.0 Congenital absence of breast with absent nipple Q83.1 Accessory breast Q83.2 Absent nipple Q83.3 Accessory nipple Q83.8 Other congenital malformations of breast Q83.9 Congenital malformation of breast, unspecified Z90.10 Acquired absence of unspecified breast and nipple Z90.11 Acquired absence of right breast and nipple Z90.12 Acquired absence of left breast and nipple Z90.13 Acquired absence of bilateral breasts and nipples Z80.3 Family history of malignant neoplasm of breast Z85.3 Personal history of malignant neoplasm of breast Moda Health Medical Necessity Criteria Breast Reconstruction after Mastectomy or Lumpectomy Page 7/8
8 Appendix 2 Centers for Medicare and Medicaid Services (CMS) Medicare coverage for outpatient (Part B) drugs is outlined in the Medicare Benefit Policy Manual (Pub ), Chapter 15, 50 Drugs and Biologicals. In addition, National Coverage Determination (NCD) and Local Coverage Determinations (LCDs) may exist and compliance with these policies is required where applicable. They can be found at: Additional indications may be covered at the discretion of the health plan. Medicare Part B Covered Diagnosis Codes (applicable to existing NCD/LCD): Jurisdiction(s): 5, 8 NCD/LCD Document (s): NCD/LCD Document (s): Medicare Part B Administrative Contractor (MAC) Jurisdictions Jurisdiction Applicable State/US Territory Contractor F (2 & 3) AK, WA, OR, ID, ND, SD, MT, WY, UT, AZ Noridian Healthcare Solutions, LLC Moda Health Medical Necessity Criteria Breast Reconstruction after Mastectomy or Lumpectomy Page 8/8
Medical Policy Original Effective Date: Revised Date: Page 1 of 8
 Page 1 of 8 Disclaimer Description Coverage Determination Refer to the member s specific benefit plan and Schedule of Benefits to determine coverage. This may not be a benefit on all plans, or the plan
Page 1 of 8 Disclaimer Description Coverage Determination Refer to the member s specific benefit plan and Schedule of Benefits to determine coverage. This may not be a benefit on all plans, or the plan
Medical Policy An independent licensee of the Blue Cross Blue Shield Association
 Breast Reconstructive Surgery After Mastectomy Page 1 of 8 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Breast Reconstructive Surgery After Mastectomy PRE-DETERMINATION
Breast Reconstructive Surgery After Mastectomy Page 1 of 8 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Breast Reconstructive Surgery After Mastectomy PRE-DETERMINATION
BREAST RECONSTRUCTION POST MASTECTOMY
 UnitedHealthcare Commercial Coverage Determination Guideline BREAST RECONSTRUCTION POST MASTECTOMY Guideline Number: SUR057 Effective Date: February 1, 2018 Table of Contents Page INSTRUCTIONS FOR USE...
UnitedHealthcare Commercial Coverage Determination Guideline BREAST RECONSTRUCTION POST MASTECTOMY Guideline Number: SUR057 Effective Date: February 1, 2018 Table of Contents Page INSTRUCTIONS FOR USE...
BREAST RECONSTRUCTION POST MASTECTOMY
 UnitedHealthcare Commercial Coverage Determination Guideline BREAST RECONSTRUCTION POST MASTECTOMY Guideline Number: SUR057 Effective Date: January 1, 2019 Table of Contents Page INSTRUCTIONS FOR USE...
UnitedHealthcare Commercial Coverage Determination Guideline BREAST RECONSTRUCTION POST MASTECTOMY Guideline Number: SUR057 Effective Date: January 1, 2019 Table of Contents Page INSTRUCTIONS FOR USE...
Breast Reconstruction Following Mastectomy or Lumpectomy
 Breast Reconstruction Following Mastectomy or Lumpectomy [For the list of services and procedures that need preauthorization, please refer to www.mcs.com.pr go to Comunicados a Proveedores, and click Cartas
Breast Reconstruction Following Mastectomy or Lumpectomy [For the list of services and procedures that need preauthorization, please refer to www.mcs.com.pr go to Comunicados a Proveedores, and click Cartas
Breast Reconstruction Surgery
 Breast Reconstruction Surgery I. Policy University Health Alliance (UHA) will reimburse for Breast Reconstruction Surgery when it is determined to be medically necessary and when it meets the medical criteria
Breast Reconstruction Surgery I. Policy University Health Alliance (UHA) will reimburse for Breast Reconstruction Surgery when it is determined to be medically necessary and when it meets the medical criteria
CLINICAL MEDICAL POLICY
 CLINICAL MEDICAL POLICY Policy Name: Reconstructive Breast Surgery Policy Number: MP-052-MD-DE Approved By: Medical Management Medical Policy Provider Notice Date: 08/01/2017 Original Effective Date: 09/01/2017
CLINICAL MEDICAL POLICY Policy Name: Reconstructive Breast Surgery Policy Number: MP-052-MD-DE Approved By: Medical Management Medical Policy Provider Notice Date: 08/01/2017 Original Effective Date: 09/01/2017
Breast Surgery Corporate Medical Policy
 File name: Breast Surgery File code: UM.SURG.17 Origination: 2016 Last Review: 11/2018 (PA List Review) Next Review: 11/2019 Effective Date: 04/01/2018 Breast Surgery Corporate Medical Policy Description/Summary
File name: Breast Surgery File code: UM.SURG.17 Origination: 2016 Last Review: 11/2018 (PA List Review) Next Review: 11/2019 Effective Date: 04/01/2018 Breast Surgery Corporate Medical Policy Description/Summary
Medical Policy Original Effective Date: Revised Date: 09/26/2018 Page 1 of 26
 Contents: This includes the following items: 1. Breast Reconstruction Following Mastectomy: Page 1 of 26 2. Breast Implant Removal and/or Replacement and Capsulectomy: 3. Breast Reduction Mammaplasty for
Contents: This includes the following items: 1. Breast Reconstruction Following Mastectomy: Page 1 of 26 2. Breast Implant Removal and/or Replacement and Capsulectomy: 3. Breast Reduction Mammaplasty for
Reconstructive Breast Surgery and Management of Breast Implants
 Reconstructive Breast Surgery and Management of Breast Implants Policy Number: 7.01.22 Last Review: 1/2018 Origination: 3/1993 Next Review: 1/2019 Policy Blue Cross and Blue Shield of Kansas City (Blue
Reconstructive Breast Surgery and Management of Breast Implants Policy Number: 7.01.22 Last Review: 1/2018 Origination: 3/1993 Next Review: 1/2019 Policy Blue Cross and Blue Shield of Kansas City (Blue
Medical Review Criteria Breast Surgeries
 Medical Review Criteria Breast Surgeries Subject: Breast Surgeries Authorization: Prior authorization is required for the following procedures requested for members enrolled in HPHC commercial (HMO, POS,
Medical Review Criteria Breast Surgeries Subject: Breast Surgeries Authorization: Prior authorization is required for the following procedures requested for members enrolled in HPHC commercial (HMO, POS,
Medical Review Criteria Breast Surgeries
 Medical Review Criteria Breast Surgeries Effective Date: November 8, 2016 Subject: Breast Surgeries Policy: HPHC covers medically necessary breast surgeries including mastectomy, breast reconstruction,
Medical Review Criteria Breast Surgeries Effective Date: November 8, 2016 Subject: Breast Surgeries Policy: HPHC covers medically necessary breast surgeries including mastectomy, breast reconstruction,
BREAST RECONSTRUCTION/REMOVAL AND REPLACEMENT OF IMPLANTS
 Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services, procedures, medical devices and drugs are dependent upon
Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services, procedures, medical devices and drugs are dependent upon
Breast Reconstruction: Current Strategies and Future Opportunities
 Breast Reconstruction: Current Strategies and Future Opportunities Hani Sbitany, MD Assistant Professor of Surgery University of California, San Francisco Division of Plastic and Reconstructive Surgery
Breast Reconstruction: Current Strategies and Future Opportunities Hani Sbitany, MD Assistant Professor of Surgery University of California, San Francisco Division of Plastic and Reconstructive Surgery
Current Strategies in Breast Reconstruction
 Current Strategies in Breast Reconstruction Hani Sbitany, MD Assistant Professor of Surgery University of California, San Francisco Division of Plastic and Reconstructive Surgery 12 th Annual School of
Current Strategies in Breast Reconstruction Hani Sbitany, MD Assistant Professor of Surgery University of California, San Francisco Division of Plastic and Reconstructive Surgery 12 th Annual School of
Reconstruction of the Breast after Cancer An Overview of Procedures and Options by Karen M. Horton, MD, MSc, FRCSC
 Downloaded from Reconstruction of the Breast after Cancer An Overview of Procedures and Options by Karen M. Horton, MD, MSc, FRCSC What is Breast Reconstruction? Reconstruction of the breast involves recreating
Downloaded from Reconstruction of the Breast after Cancer An Overview of Procedures and Options by Karen M. Horton, MD, MSc, FRCSC What is Breast Reconstruction? Reconstruction of the breast involves recreating
Reduction Mammoplasty
 Reduction Mammoplasty Date of Origin: 02/1999 Last Review Date: 06/28/2017 Effective Date: 06/28/2017 Dates Reviewed: 05/1999, 10/2000, 09/2001, 03/2002, 05/2002, 08/2002, 10/2003, 10/2004, 09/2005, 11/2005,
Reduction Mammoplasty Date of Origin: 02/1999 Last Review Date: 06/28/2017 Effective Date: 06/28/2017 Dates Reviewed: 05/1999, 10/2000, 09/2001, 03/2002, 05/2002, 08/2002, 10/2003, 10/2004, 09/2005, 11/2005,
Breast Reconstruction Options
 Breast Reconstruction Options Natural reconstruction using your ABDOMINAL tissue: TRAM Flap (Transverse Rectus Abdominis Myocutaneous) There are various forms of TRAM flap reconstruction that are commonly
Breast Reconstruction Options Natural reconstruction using your ABDOMINAL tissue: TRAM Flap (Transverse Rectus Abdominis Myocutaneous) There are various forms of TRAM flap reconstruction that are commonly
Dates Reviewed: 12/2012, 3/2013, 6/2013, 9/2013, 11/2013, 12/2013, 3/2014, 6/2014, 9/2014, 12/2014,
 Perjeta (pertuzumab) Last Review Date: 5/30/2017 Date of Origin: 11/01/2012 Document Number: IC-0096 Dates Reviewed: 12/2012, 3/2013, 6/2013, 9/2013, 11/2013, 12/2013, 3/2014, 6/2014, 9/2014, 12/2014,
Perjeta (pertuzumab) Last Review Date: 5/30/2017 Date of Origin: 11/01/2012 Document Number: IC-0096 Dates Reviewed: 12/2012, 3/2013, 6/2013, 9/2013, 11/2013, 12/2013, 3/2014, 6/2014, 9/2014, 12/2014,
Intrathecal Opioid Therapy for Management of Chronic Pain
 Intrathecal Opioid Therapy for Management of Chronic Pain Date of Origin: 01/2000 Last Review Date: 09/27/2017 Effective Date: 09/27/2017 Dates Reviewed: 11/2002, 12/2003, 12/2004, 12/2005, 12/2006, 12/2007,
Intrathecal Opioid Therapy for Management of Chronic Pain Date of Origin: 01/2000 Last Review Date: 09/27/2017 Effective Date: 09/27/2017 Dates Reviewed: 11/2002, 12/2003, 12/2004, 12/2005, 12/2006, 12/2007,
Breast Reconstruction
 Steven E. Copit, M.D. Chief- Division of Plastic Surgery Thomas Jefferson University Hospital Philadelphia, PA analysis of The Defect Skin Breast Volume Nipple Areola Complex analysis of The Defect the
Steven E. Copit, M.D. Chief- Division of Plastic Surgery Thomas Jefferson University Hospital Philadelphia, PA analysis of The Defect Skin Breast Volume Nipple Areola Complex analysis of The Defect the
CMS Limitations Guide Mammography & Bone Mass Measurement
 Starting October 1, 2015, CMS will update their existing medical necessity limitations on tests and procedures to correspond to ICD-10 codes. This limitations guide provides you with the latest changes.
Starting October 1, 2015, CMS will update their existing medical necessity limitations on tests and procedures to correspond to ICD-10 codes. This limitations guide provides you with the latest changes.
Herniated Disc Treatment Non-covered Procedures
 Herniated Disc Treatment Non-covered Procedures Date of Origin: 03/2004 Last Review Date: 03/22/2017 Effective Date: 03/22/2017 Dates Reviewed: 02/2005, 02/2006, 02/2007, 03/2008, 01/2009, 04/2009, 02/2011,
Herniated Disc Treatment Non-covered Procedures Date of Origin: 03/2004 Last Review Date: 03/22/2017 Effective Date: 03/22/2017 Dates Reviewed: 02/2005, 02/2006, 02/2007, 03/2008, 01/2009, 04/2009, 02/2011,
Continuous Glucose Monitoring (CGM)
 Continuous Glucose Monitoring (CGM) Date of Origin: 02/2001 Last Review Date: 07/26/2017 Effective Date: 07/26/2017 Dates Reviewed: 04/2004, 04/2005, 03/2006, 11/2006, 12/2007, 03/2008, 09/2008, 04/2009,
Continuous Glucose Monitoring (CGM) Date of Origin: 02/2001 Last Review Date: 07/26/2017 Effective Date: 07/26/2017 Dates Reviewed: 04/2004, 04/2005, 03/2006, 11/2006, 12/2007, 03/2008, 09/2008, 04/2009,
POLICY PRODUCT VARIATIONS DESCRIPTION/BACKGROUND RATIONALE DEFINITIONS BENEFIT VARIATIONS DISCLAIMER CODING INFORMATION REFERENCES POLICY HISTORY
 Original Issue Date (Created): July 26, 2011 Most Recent Review Date (Revised): March 25, 2014 Effective Date: June 1, 2014 POLICY PRODUCT VARIATIONS DESCRIPTION/BACKGROUND RATIONALE DEFINITIONS BENEFIT
Original Issue Date (Created): July 26, 2011 Most Recent Review Date (Revised): March 25, 2014 Effective Date: June 1, 2014 POLICY PRODUCT VARIATIONS DESCRIPTION/BACKGROUND RATIONALE DEFINITIONS BENEFIT
National Medical Policy
 National Medical Policy Subject: Policy Number: Breast Reconstructive Surgery NMP492 Effective Date*: February 2013 Updated: April 2016 This National Medical Policy is subject to the terms in the IMPORTANT
National Medical Policy Subject: Policy Number: Breast Reconstructive Surgery NMP492 Effective Date*: February 2013 Updated: April 2016 This National Medical Policy is subject to the terms in the IMPORTANT
Breast cancer reconstruction surgery (immediate and delayed) across Ontario: Patient indications and appropriate surgical options
 A Quality Initiative of the Program in Evidence-Based Care (PEBC), Cancer Care Ontario (CCO) Breast cancer reconstruction surgery (immediate and delayed) across Ontario: Patient indications and appropriate
A Quality Initiative of the Program in Evidence-Based Care (PEBC), Cancer Care Ontario (CCO) Breast cancer reconstruction surgery (immediate and delayed) across Ontario: Patient indications and appropriate
Imfinzi (durvalumab) (Intravenous)
 Imfinzi (durvalumab) (Intravenous) Last Review Date: 09/05/2018 Date of Origin: 05/30/2017 Dates Reviewed: 05/2017, 08/2017, 11/2017, 02/2018, 05/2018, 09/2018 Document Number: IC-0301 I. Length of Authorization
Imfinzi (durvalumab) (Intravenous) Last Review Date: 09/05/2018 Date of Origin: 05/30/2017 Dates Reviewed: 05/2017, 08/2017, 11/2017, 02/2018, 05/2018, 09/2018 Document Number: IC-0301 I. Length of Authorization
Pulmonary Rehabilitation
 Pulmonary Rehabilitation Date of Origin: 06/2005 Last Review Date: 10/25/2017 Effective Date: 10/25/2017 Dates Reviewed: 05/2006, 05/2007, 05/2008, 11/2009, 02/2011, 01/2012, 08/2013, 07/2014, 09/2015,
Pulmonary Rehabilitation Date of Origin: 06/2005 Last Review Date: 10/25/2017 Effective Date: 10/25/2017 Dates Reviewed: 05/2006, 05/2007, 05/2008, 11/2009, 02/2011, 01/2012, 08/2013, 07/2014, 09/2015,
Breast Restoration Surgery After a mastectomy
 UW MEDICINE PATIENT EDUCATION Breast Restoration Surgery After a mastectomy This handout explains the most common procedures that are used at University of Washington Medical Center (UWMC) to restore a
UW MEDICINE PATIENT EDUCATION Breast Restoration Surgery After a mastectomy This handout explains the most common procedures that are used at University of Washington Medical Center (UWMC) to restore a
Tecentriq (atezolizumab) (Intravenous)
 Tecentriq (atezolizumab) (Intravenous) Last Review Date: 06/01/2018 Date of Origin: 06/28/2016 Document Number: IC-0278 Dates Reviewed: 06/2016, 08/2016, 10/2016, 02/2017, 04/2017, 08/2017, 11/2017, 02/2018,
Tecentriq (atezolizumab) (Intravenous) Last Review Date: 06/01/2018 Date of Origin: 06/28/2016 Document Number: IC-0278 Dates Reviewed: 06/2016, 08/2016, 10/2016, 02/2017, 04/2017, 08/2017, 11/2017, 02/2018,
Diagnosis and Treatment of Patients with Primary and Metastatic Breast Cancer. Oncoplastic and Reconstructive Surgery
 Diagnosis and Treatment of Patients with Primary and Metastatic Breast Cancer Oncoplastic and Reconstructive Surgery Plastic-reconstructive aspects after mastectomy Versions 2002 2017: Audretsch / Bauerfeind
Diagnosis and Treatment of Patients with Primary and Metastatic Breast Cancer Oncoplastic and Reconstructive Surgery Plastic-reconstructive aspects after mastectomy Versions 2002 2017: Audretsch / Bauerfeind
How To Make a Good Mastectomy for Reconstruction Based on the Anatomy. Zhang Jin, Ph.D MD
 How To Make a Good Mastectomy for Reconstruction Based on the Anatomy Zhang Jin, Ph.D MD Deputy Director and Professor Tianjin Medical University Cancer Institute and Hospital People s Republic of China
How To Make a Good Mastectomy for Reconstruction Based on the Anatomy Zhang Jin, Ph.D MD Deputy Director and Professor Tianjin Medical University Cancer Institute and Hospital People s Republic of China
Prophylactic Mastectomy & Reconstructive Implications
 Prophylactic Mastectomy & Reconstructive Implications Minas T Chrysopoulo, MD PRMA Center For Advanced Breast Reconstruction Prophylactic Mastectomy Surgical removal of one or both breasts to reduce the
Prophylactic Mastectomy & Reconstructive Implications Minas T Chrysopoulo, MD PRMA Center For Advanced Breast Reconstruction Prophylactic Mastectomy Surgical removal of one or both breasts to reduce the
(https://www.aetna.com/) Number: Policy *Please see amendment for Pennsylvania Medicaid at the end
 (https://www.aetna.com/) Number: 0185 Policy *Please see amendment for Pennsylvania Medicaid at the end of this CPB. Aetna considers reconstructive breast surgery medically necessary after a medically
(https://www.aetna.com/) Number: 0185 Policy *Please see amendment for Pennsylvania Medicaid at the end of this CPB. Aetna considers reconstructive breast surgery medically necessary after a medically
Cyramza (ramucirumab) (Intravenous)
 Cyramza (ramucirumab) (Intravenous) Document Number: IC 0199 Last Review Date: 5/1/2018 Date of Origin: 06/24/2014 Dates Reviewed: 09/2014, 01/2015, 05/2015, 11/2015, 04/2016, 08/2016, 11/2016, 05/2017,
Cyramza (ramucirumab) (Intravenous) Document Number: IC 0199 Last Review Date: 5/1/2018 Date of Origin: 06/24/2014 Dates Reviewed: 09/2014, 01/2015, 05/2015, 11/2015, 04/2016, 08/2016, 11/2016, 05/2017,
Breast Reconstruction. Westmead Breast Cancer Institute
 Breast Reconstruction Westmead Breast Cancer Institute What is breast reconstruction? Breast reconstruction is a surgical procedure that creates a shape on the chest wall following a mastectomy. Occasionally,
Breast Reconstruction Westmead Breast Cancer Institute What is breast reconstruction? Breast reconstruction is a surgical procedure that creates a shape on the chest wall following a mastectomy. Occasionally,
Anesthesia for Routine Gastrointestinal Endoscopic Procedures (Additional description)
 Anesthesia for Routine Gastrointestinal Endoscopic Procedures (Additional description) Date of Origin: 05/2012 Last Review Date: 12/06/2017 Effective Date: 01/01/2018 Dates Reviewed: 12/2013, 11/2014,
Anesthesia for Routine Gastrointestinal Endoscopic Procedures (Additional description) Date of Origin: 05/2012 Last Review Date: 12/06/2017 Effective Date: 01/01/2018 Dates Reviewed: 12/2013, 11/2014,
Protocol. Reconstructive Breast Surgery/Management of Breast Implants
 Protocol Reconstructive Breast Surgery/Management of Breast Implants Medical Benefit Effective Date: 04/01/14 Next Review Date: 11/18 Preauthorization Yes Review Dates: 02/07, 02/08, 01/09, 01/10, 01/11,
Protocol Reconstructive Breast Surgery/Management of Breast Implants Medical Benefit Effective Date: 04/01/14 Next Review Date: 11/18 Preauthorization Yes Review Dates: 02/07, 02/08, 01/09, 01/10, 01/11,
Neil J. Zemmel, MD, FACS Steven J. Montante, MD Megan J. Russell, PA-C. Your Guide To BREAST RECONSTRUCTION
 Neil J. Zemmel, MD, FACS Steven J. Montante, MD Megan J. Russell, PA-C Your Guide To BREAST RECONSTRUCTION Introduction The diagnosis of breast cancer begins a journey of making many informed decisions
Neil J. Zemmel, MD, FACS Steven J. Montante, MD Megan J. Russell, PA-C Your Guide To BREAST RECONSTRUCTION Introduction The diagnosis of breast cancer begins a journey of making many informed decisions
Prior Authorization Review Panel MCO Policy Submission
 Prior Authorization Review Panel MCO Policy Submission A separate copy of this form must accompany each policy submitted for review. Policies submitted without this form will not be considered for review.
Prior Authorization Review Panel MCO Policy Submission A separate copy of this form must accompany each policy submitted for review. Policies submitted without this form will not be considered for review.
Plastic surgery of the breast includes; augmentation, reduction, Plastic Surgery of the Breast. Abstract. Continuing Education Column
 Plastic Surgery of the Breast Keuk Shun Shin, M.D. Keuk SHUN SHIN s Asthetic Plastic Surgery E mail: drsks@drsks.co.kr Abstract Plastic surgery of the breast includes; augmentation, reduction, reconstruction
Plastic Surgery of the Breast Keuk Shun Shin, M.D. Keuk SHUN SHIN s Asthetic Plastic Surgery E mail: drsks@drsks.co.kr Abstract Plastic surgery of the breast includes; augmentation, reduction, reconstruction
Treatment or Removal of Benign Skin Lesions
 Treatment or Removal of Benign Skin Lesions Date of Origin: 10/26/2016 Last Review Date: 12/15/2017 Effective Date: 10/25/2017 Dates Reviewed: 10/2016, 10/2017, 12/15/2017 Developed By: Medical Necessity
Treatment or Removal of Benign Skin Lesions Date of Origin: 10/26/2016 Last Review Date: 12/15/2017 Effective Date: 10/25/2017 Dates Reviewed: 10/2016, 10/2017, 12/15/2017 Developed By: Medical Necessity
Eylea (aflibercept) Document Number: IC-0026
 Eylea (aflibercept) Document Number: IC-0026 Last Review Date: 3/1/2018 Date of Origin: 02/07/2013 Dates Reviewed: 03/07/2013, 06/2013, 09/2013, 12/2013, 03/2014, 06/2014, 09/2014, 12/2014, 03/2015, 04/2015,
Eylea (aflibercept) Document Number: IC-0026 Last Review Date: 3/1/2018 Date of Origin: 02/07/2013 Dates Reviewed: 03/07/2013, 06/2013, 09/2013, 12/2013, 03/2014, 06/2014, 09/2014, 12/2014, 03/2015, 04/2015,
Breast Cancer Reconstruction
 Breast Cancer Jerome H. Liu, MD Tom S. Liu, MD Jerome H. Liu, MD Undergraduate: Brown University Medical School: University of California, Los Angeles Residency: UCLA Medical Center Fellowship:UCLA Medical
Breast Cancer Jerome H. Liu, MD Tom S. Liu, MD Jerome H. Liu, MD Undergraduate: Brown University Medical School: University of California, Los Angeles Residency: UCLA Medical Center Fellowship:UCLA Medical
Cardiac Disease Screening Lipid Profile
 Cardiac Disease Screening Lipid Profile Date of Origin: 04/2003 Last Review Date: 08/23/2017 Effective Date: 08/23/2017 Dates Reviewed: 01/2004, 04/2005, 12/2005, 06/2006, 06/2007, 07/2008, 6/2011, 05/2012,
Cardiac Disease Screening Lipid Profile Date of Origin: 04/2003 Last Review Date: 08/23/2017 Effective Date: 08/23/2017 Dates Reviewed: 01/2004, 04/2005, 12/2005, 06/2006, 06/2007, 07/2008, 6/2011, 05/2012,
Wound & Burn. Reimbursement & Coding Guide
 Wound & Burn Reimbursement & Coding Guide Wound & Burn Reimbursement and Coding Guide MicroMatrix and Cytal devices facilitate the remodeling of functional, site-appropriate tissue. Comprised of ACell
Wound & Burn Reimbursement & Coding Guide Wound & Burn Reimbursement and Coding Guide MicroMatrix and Cytal devices facilitate the remodeling of functional, site-appropriate tissue. Comprised of ACell
High Frequency Chest Wall Oscillating Devices (HFCWO) (Airway Clearance Systems)
 High Frequency Chest Wall Oscillating Devices (HFCWO) (Airway Clearance Systems) Date of Origin: 05/2015 Last Review Date: 07/26/2017 Effective Date: 07/26/2017 Dates Reviewed: 07/2016 Developed By: Medical
High Frequency Chest Wall Oscillating Devices (HFCWO) (Airway Clearance Systems) Date of Origin: 05/2015 Last Review Date: 07/26/2017 Effective Date: 07/26/2017 Dates Reviewed: 07/2016 Developed By: Medical
The Case FOR Oncoplastic Surgery in Small Breasts. Barbara L. Smith, MD, PhD Massachusetts General Hospital Harvard Medical School Boston, MA USA
 The Case FOR Oncoplastic Surgery in Small Breasts Barbara L. Smith, MD, PhD Massachusetts General Hospital Harvard Medical School Boston, MA USA Changing issues in breast cancer management Early detection
The Case FOR Oncoplastic Surgery in Small Breasts Barbara L. Smith, MD, PhD Massachusetts General Hospital Harvard Medical School Boston, MA USA Changing issues in breast cancer management Early detection
Continuous Glucose Monitoring (CGM)
 Continuous Glucose Monitoring (CGM) Date of Origin: 02/2001 Last Review Date: 08/22/2018 Effective Date: 08/22/2018 Dates Reviewed: 04/2004, 04/2005, 03/2006, 11/2006, 12/2007, 03/2008, 09/2008, 04/2009,
Continuous Glucose Monitoring (CGM) Date of Origin: 02/2001 Last Review Date: 08/22/2018 Effective Date: 08/22/2018 Dates Reviewed: 04/2004, 04/2005, 03/2006, 11/2006, 12/2007, 03/2008, 09/2008, 04/2009,
In a second stage or a second operation that tissue expander is removed through the same incision and the implant is placed within the chest pocket.
 Hello, I m Summer Hanson. I m an assistant professor in the Department of Plastics & Reconstructive Surgery at The University of Texas MD Anderson Cancer Center and today I m going to talk about the role
Hello, I m Summer Hanson. I m an assistant professor in the Department of Plastics & Reconstructive Surgery at The University of Texas MD Anderson Cancer Center and today I m going to talk about the role
Transcranial Magnetic Stimulation
 Transcranial Magnetic Stimulation Date of Origin: 7/24/2018 Last Review Date: 7/24/2018 Effective Date: 08/01/18 Dates Reviewed: 7/24/2018 Developed By: Medical Necessity Criteria Committee I. Description
Transcranial Magnetic Stimulation Date of Origin: 7/24/2018 Last Review Date: 7/24/2018 Effective Date: 08/01/18 Dates Reviewed: 7/24/2018 Developed By: Medical Necessity Criteria Committee I. Description
AHM Breast Reconstruction
 AHM Breast Reconstruction Clinical Indications Breast Reconstruction o TIP: State and Federal regulations concerning breast reconstruction after a medically necessary mastectomy impact plans variously,
AHM Breast Reconstruction Clinical Indications Breast Reconstruction o TIP: State and Federal regulations concerning breast reconstruction after a medically necessary mastectomy impact plans variously,
Contralateral Prophylactic Mastectomy with Immediate Reconstruction: Added Benefits, Added Risks
 Contralateral Prophylactic Mastectomy with Immediate Reconstruction: Added Benefits, Added Risks Grant W. Carlson Wadley R. Glenn Professor of Surgery Divisions of Plastic Surgery & Surgical Oncology Emory
Contralateral Prophylactic Mastectomy with Immediate Reconstruction: Added Benefits, Added Risks Grant W. Carlson Wadley R. Glenn Professor of Surgery Divisions of Plastic Surgery & Surgical Oncology Emory
Advances in Breast Surgery. Catherine Campo, D.O. Breast Surgeon Meridian Health System April 17, 2015
 Advances in Breast Surgery Catherine Campo, D.O. Breast Surgeon Meridian Health System April 17, 2015 Objectives Understand the surgical treatment of breast cancer Be able to determine when a lumpectomy
Advances in Breast Surgery Catherine Campo, D.O. Breast Surgeon Meridian Health System April 17, 2015 Objectives Understand the surgical treatment of breast cancer Be able to determine when a lumpectomy
2017 PHYSICIAN CODING GUIDE
 2017 PHYSICIAN CODING GUIDE Breast Repair and/or Reconstruction Surgeon s Procedure Codes 11970 Replacement of tissue expander with permanent prosthesis 8.01 $628.05 +15777 Implantation of biologic implant
2017 PHYSICIAN CODING GUIDE Breast Repair and/or Reconstruction Surgeon s Procedure Codes 11970 Replacement of tissue expander with permanent prosthesis 8.01 $628.05 +15777 Implantation of biologic implant
Is Unilateral Implant or Autologous Breast Reconstruction Better in Obtaining Breast Symmetry?
 ORIGINAL ARTICLE Is Unilateral Implant or Autologous Breast Reconstruction Better in Obtaining Breast Symmetry? Oriana Cohen, MD, Kevin Small, MD, Christina Lee, BA, Oriana Petruolo, MD, Nolan Karp, MD,
ORIGINAL ARTICLE Is Unilateral Implant or Autologous Breast Reconstruction Better in Obtaining Breast Symmetry? Oriana Cohen, MD, Kevin Small, MD, Christina Lee, BA, Oriana Petruolo, MD, Nolan Karp, MD,
Updates in Breast Care. Truth or Hype. History of Breast Cancer Surgery. Dr Karen Barbosa 5/3/2017 4/20/2017
 Updates in Breast Care Dr Karen Barbosa 4/20/2017 Truth or Hype Princess Bust Developer Sears, Roebuck and Co. 1897 Promised to make the breast round, firm and beautiful History of Breast Cancer Surgery
Updates in Breast Care Dr Karen Barbosa 4/20/2017 Truth or Hype Princess Bust Developer Sears, Roebuck and Co. 1897 Promised to make the breast round, firm and beautiful History of Breast Cancer Surgery
Krystexxa (pegloticase) Document Number: IC-0158
 Krystexxa (pegloticase) Document Number: IC-0158 Last Review Date: 06/27/2017 Date of Origin: 02/07/20103 Dates Reviewed: 11/2013, 08/2014, 07/2015, 07/2016, 09/2016, 12/2016, 03/2017, 06/2017 I. Length
Krystexxa (pegloticase) Document Number: IC-0158 Last Review Date: 06/27/2017 Date of Origin: 02/07/20103 Dates Reviewed: 11/2013, 08/2014, 07/2015, 07/2016, 09/2016, 12/2016, 03/2017, 06/2017 I. Length
Chapter 11 Worksheet Code It
 Class: Date: Chapter 11 Worksheet 3 2 1 Code It True/False Indicate whether the statement is true or false. 1. Surgical destruction is considered part of the surgical procedure description. 2. Prepping
Class: Date: Chapter 11 Worksheet 3 2 1 Code It True/False Indicate whether the statement is true or false. 1. Surgical destruction is considered part of the surgical procedure description. 2. Prepping
Clinical Policy Title: Breast reconstruction following breast cancer surgery
 Clinical Policy Title: Breast reconstruction following breast cancer surgery Clinical Policy Number: 13.03.04 Effective Date: April 1, 2017 Initial Review Date: November 16, 2016 Most Recent Review Date:
Clinical Policy Title: Breast reconstruction following breast cancer surgery Clinical Policy Number: 13.03.04 Effective Date: April 1, 2017 Initial Review Date: November 16, 2016 Most Recent Review Date:
The decision to repair a partial mastectomy CME. State of the Art and Science in Postmastectomy Breast Reconstruction.
 CME State of the Art and Science in Postmastectomy Breast Reconstruction Steven J. Kronowitz, M.D. Houston, Texas Learning Objectives: After reading this article, the participant should be able to: 1.
CME State of the Art and Science in Postmastectomy Breast Reconstruction Steven J. Kronowitz, M.D. Houston, Texas Learning Objectives: After reading this article, the participant should be able to: 1.
Cardiac Disease Screening Lipid Profile
 Cardiac Disease Screening Lipid Profile Date of Origin: 04/2003 Last Review Date: 02/27/2019 Effective Date: 03/01/2019 Dates Reviewed: 01/2004, 04/2005, 12/2005, 06/2006, 06/2007, 07/2008, 6/2011, 05/2012,
Cardiac Disease Screening Lipid Profile Date of Origin: 04/2003 Last Review Date: 02/27/2019 Effective Date: 03/01/2019 Dates Reviewed: 01/2004, 04/2005, 12/2005, 06/2006, 06/2007, 07/2008, 6/2011, 05/2012,
JPRAS Open 3 (2015) 1e5. Contents lists available at ScienceDirect. JPRAS Open. journal homepage:
 JPRAS Open 3 (2015) 1e5 Contents lists available at ScienceDirect JPRAS Open journal homepage: http://www.journals.elsevier.com/ jpras-open Case report The pedicled transverse partial latissimus dorsi
JPRAS Open 3 (2015) 1e5 Contents lists available at ScienceDirect JPRAS Open journal homepage: http://www.journals.elsevier.com/ jpras-open Case report The pedicled transverse partial latissimus dorsi
Velcade (bortezomib) Document Number: IC-0137
 Velcade (bortezomib) Document Number: IC-0137 Last Review Date: 11/21/2017 Date of Origin: 11/28/2011 Dates Reviewed: 12/2011, 03/2012, 06/2012, 09/2012, 12/2012, 03/2013, 06/2013, 09/2013, 12/2013, 03/2014,
Velcade (bortezomib) Document Number: IC-0137 Last Review Date: 11/21/2017 Date of Origin: 11/28/2011 Dates Reviewed: 12/2011, 03/2012, 06/2012, 09/2012, 12/2012, 03/2013, 06/2013, 09/2013, 12/2013, 03/2014,
Breast Reconstruction with Superficial Inferior Epigastric Artery Flaps: A Prospective Comparison with TRAM and DIEP Flaps
 Breast Reconstruction with Superficial Inferior Epigastric Artery Flaps: A Prospective Comparison with TRAM and DIEP Flaps Pierre M. Chevray, M.D., Ph.D. Houston, Texas Breast reconstruction using the
Breast Reconstruction with Superficial Inferior Epigastric Artery Flaps: A Prospective Comparison with TRAM and DIEP Flaps Pierre M. Chevray, M.D., Ph.D. Houston, Texas Breast reconstruction using the
Breast reconstruction has an important role BREAST. A Two-Year Prospective Analysis of Trunk Function in TRAM Breast Reconstructions
 BREAST A Two-Year Prospective Analysis of Trunk Function in TRAM Breast Reconstructions Amy K. Alderman, M.D. William M. Kuzon, Jr., M.D., Ph.D. Edwin G. Wilkins, M.D. Ann Arbor, Mich. Background: Functional
BREAST A Two-Year Prospective Analysis of Trunk Function in TRAM Breast Reconstructions Amy K. Alderman, M.D. William M. Kuzon, Jr., M.D., Ph.D. Edwin G. Wilkins, M.D. Ann Arbor, Mich. Background: Functional
Yervoy (ipilmumab) Last Review Date: 03/25/2014 Date of Origin: 11/28/2011. Prior Auth Available: Post-Service Edit:
 Yervoy (ipilmumab) Date of Origin: 11/28/2011 Prior Auth Available: Post-Service Edit: Dates Reviewed: 12/13/2011, 03/2012, 06/19/2012, 09/06/2012, 12/06/2012, 05/16/2013, 06/06/2013, 09/05/2013, 12/05/2013,
Yervoy (ipilmumab) Date of Origin: 11/28/2011 Prior Auth Available: Post-Service Edit: Dates Reviewed: 12/13/2011, 03/2012, 06/19/2012, 09/06/2012, 12/06/2012, 05/16/2013, 06/06/2013, 09/05/2013, 12/05/2013,
Medical Policy An independent licensee of the Blue Cross Blue Shield Association
 Surgical Treatment of Gynecomastia Page 1 of 6 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Surgical Treatment of Gynecomastia Professional Institutional Original
Surgical Treatment of Gynecomastia Page 1 of 6 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Surgical Treatment of Gynecomastia Professional Institutional Original
Reconstructive and Cosmetic Services
 Reconstructive and Cosmetic Services Policy Number: 10.01.09 Last Review: 4/2018 Origination: 2/2006 Next Review: 4/2019 Policy Determination of whether a proposed therapy would be considered reconstructive
Reconstructive and Cosmetic Services Policy Number: 10.01.09 Last Review: 4/2018 Origination: 2/2006 Next Review: 4/2019 Policy Determination of whether a proposed therapy would be considered reconstructive
Figure 1. Anatomy of the breast
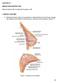 CHAPTER 12 BREAST RECONSTRUCTION Mihaela Rapolti, MD and Michelle Roughton, MD I. BREAST ANATOMY A. Mastering breast anatomy is essential for understanding how the breast changes with aging and principles
CHAPTER 12 BREAST RECONSTRUCTION Mihaela Rapolti, MD and Michelle Roughton, MD I. BREAST ANATOMY A. Mastering breast anatomy is essential for understanding how the breast changes with aging and principles
Intravitreal Avastin (Bevacizumab)
 Intravitreal Avastin (Bevacizumab) Date of Origin: 10/18/2018 Last Review Date: 10/18/2018 Effective Date: 10/18/2018 Dates Reviewed: 10/2018 Developed By: Medical Criteria Committee I. Length of Authorization
Intravitreal Avastin (Bevacizumab) Date of Origin: 10/18/2018 Last Review Date: 10/18/2018 Effective Date: 10/18/2018 Dates Reviewed: 10/2018 Developed By: Medical Criteria Committee I. Length of Authorization
Reconstructive surgery following mastectomy
 Reconstructive surgery following mastectomy Kseniya Roudakova, MD A CASE FROM SUNY DOWNSTATE 60F who presented for right mastectomy with immediate TRAM flap reconstruction for recurrent breast cancer Oncologic
Reconstructive surgery following mastectomy Kseniya Roudakova, MD A CASE FROM SUNY DOWNSTATE 60F who presented for right mastectomy with immediate TRAM flap reconstruction for recurrent breast cancer Oncologic
PROS AND CONS OF IMMEDIATE PROSTHETIC IMPLANTS VS USE OF EXPANDER FOR POST MASTECTOMY BREAST RECONSTRUCTIONS
 PROS AND CONS OF IMMEDIATE PROSTHETIC IMPLANTS VS USE OF EXPANDER FOR POST MASTECTOMY BREAST Dr Tienie van Rooyen Mediclinic Kloof Hospital Pretoria IMMEDIATE Since 1990 s Skin sparing mastectomies proven
PROS AND CONS OF IMMEDIATE PROSTHETIC IMPLANTS VS USE OF EXPANDER FOR POST MASTECTOMY BREAST Dr Tienie van Rooyen Mediclinic Kloof Hospital Pretoria IMMEDIATE Since 1990 s Skin sparing mastectomies proven
Central Breast Excision With Immediate Autologous Reconstruction for Recurrent Periductal Sepsis: An Application of Oncoplastic Surgical Techniques
 Central Breast Excision With Immediate Autologous Reconstruction for Recurrent Periductal Sepsis: An Application of Oncoplastic Surgical Techniques Sinclair M. Gore, FRCS(Plast), a Gordon C. Wishart, FRCS,
Central Breast Excision With Immediate Autologous Reconstruction for Recurrent Periductal Sepsis: An Application of Oncoplastic Surgical Techniques Sinclair M. Gore, FRCS(Plast), a Gordon C. Wishart, FRCS,
Rituxan Hycela (rituximab and hyaluronidase human) (Subcutaneous)
 Rituxan Hycela (rituximab and hyaluronidase human) (Subcutaneous) Document Number: IC-0322 Last Review Date: 02/06/2018 Date of Origin: 7/20/2010 Dates Reviewed: 09/2010, 12/2010, 02/2011, 03/2011, 05/2011,
Rituxan Hycela (rituximab and hyaluronidase human) (Subcutaneous) Document Number: IC-0322 Last Review Date: 02/06/2018 Date of Origin: 7/20/2010 Dates Reviewed: 09/2010, 12/2010, 02/2011, 03/2011, 05/2011,
The Use of Vertical Scar Techniques in Reconstructive Surgery
 The Use of Vertical Scar Techniques in Reconstructive Surgery 12 Moustapha Hamdi, Phillip Blondeel, Koenraad Van Landuyt, Stan Monstrey H e who does not possess a thing cannot give it. Folk tradition Introduction
The Use of Vertical Scar Techniques in Reconstructive Surgery 12 Moustapha Hamdi, Phillip Blondeel, Koenraad Van Landuyt, Stan Monstrey H e who does not possess a thing cannot give it. Folk tradition Introduction
Nipple-Areolar Complex Reconstruction: A Review of the Literature and Introduction of the Rectangle-to-Cube Nipple Flap
 Nipple-Areolar Complex Reconstruction: A Review of the Literature and Introduction of the Rectangle-to-Cube Nipple Flap Joshua T. Henderson, BA, a ThomasJ.Lee,MD, b Andrew M. Swiergosz, BS, a Andrea R.
Nipple-Areolar Complex Reconstruction: A Review of the Literature and Introduction of the Rectangle-to-Cube Nipple Flap Joshua T. Henderson, BA, a ThomasJ.Lee,MD, b Andrew M. Swiergosz, BS, a Andrea R.
Breast Reconstruction: Patient Information Document
 breastreconstructioncanada.ca Breast Reconstruction: Patient Information Document By Dr. Nicolas Guay Dr. Haemi Lee STANDARDIZED BREAST RECONSTRUCTION PATIENT INFORMATION TABLE OF CONTENTS Glossary...
breastreconstructioncanada.ca Breast Reconstruction: Patient Information Document By Dr. Nicolas Guay Dr. Haemi Lee STANDARDIZED BREAST RECONSTRUCTION PATIENT INFORMATION TABLE OF CONTENTS Glossary...
Post-mastectomy breast reconstruction
 Follow the link from the online version of this article to obtain certified continuing medical education credits Post-mastectomy breast reconstruction Paul T R Thiruchelvam, 1 Fiona McNeill, 2 Navid Jallali,
Follow the link from the online version of this article to obtain certified continuing medical education credits Post-mastectomy breast reconstruction Paul T R Thiruchelvam, 1 Fiona McNeill, 2 Navid Jallali,
Clinical Policy Title: Cosmetic, plastic, and scar revision surgery
 Clinical Policy Title: Cosmetic, plastic, and scar revision surgery Clinical Policy Number: CCP.1184 Effective Date: October 1, 2015 Initial Review Date: August 19, 2015 Most Recent Review Date: September
Clinical Policy Title: Cosmetic, plastic, and scar revision surgery Clinical Policy Number: CCP.1184 Effective Date: October 1, 2015 Initial Review Date: August 19, 2015 Most Recent Review Date: September
Supplementary Online Content
 Supplementary Online Content Abt NB, Flores JM, Baltodano PA, et al. Neoadjuvant chemotherapy and short-term in patients undergoing mastectomy with and without breast reconstruction. JAMA Surg. Published
Supplementary Online Content Abt NB, Flores JM, Baltodano PA, et al. Neoadjuvant chemotherapy and short-term in patients undergoing mastectomy with and without breast reconstruction. JAMA Surg. Published
Four-flap Breast Reconstruction: Bilateral Stacked DIEP and PAP Flaps
 Original Article Breast Four-flap Breast Reconstruction: Bilateral Stacked DIEP and PAP Flaps James L. Mayo, MD Robert J. Allen, MD, FACS Alireza Sadeghi, MD, FACS Background: In cases of bilateral breast
Original Article Breast Four-flap Breast Reconstruction: Bilateral Stacked DIEP and PAP Flaps James L. Mayo, MD Robert J. Allen, MD, FACS Alireza Sadeghi, MD, FACS Background: In cases of bilateral breast
Breast Reconstruction Postmastectomy. Using DermaMatrix Acellular Dermis in breast reconstruction with tissue expander.
 Breast Reconstruction Postmastectomy. Using DermaMatrix Acellular Dermis in breast reconstruction with tissue expander. Strong and flexible Bacterially inactivated Provides implant support Breast Reconstruction
Breast Reconstruction Postmastectomy. Using DermaMatrix Acellular Dermis in breast reconstruction with tissue expander. Strong and flexible Bacterially inactivated Provides implant support Breast Reconstruction
2017 FlexHD Abdominal Wall Reconstruction Reimbursement Coding Reference
 2017 FlexHD Abdominal Wall Reconstruction Reimbursement Coding Reference Most Commonly Reported ICD-10-CM Procedure Codes and Descriptors ICD-10-CM Description 0WUF0KZ Supplement Abdominal Wall with Nonautologous
2017 FlexHD Abdominal Wall Reconstruction Reimbursement Coding Reference Most Commonly Reported ICD-10-CM Procedure Codes and Descriptors ICD-10-CM Description 0WUF0KZ Supplement Abdominal Wall with Nonautologous
SIMPOSIO Ricostruzione mammaria ed implicazioni radioterapiche Indicazioni
 SIMPOSIO Ricostruzione mammaria ed implicazioni radioterapiche Indicazioni Icro Meattini, MD Radiation Oncology Department - University of Florence Azienda Ospedaliero Universitaria Careggi Firenze Breast
SIMPOSIO Ricostruzione mammaria ed implicazioni radioterapiche Indicazioni Icro Meattini, MD Radiation Oncology Department - University of Florence Azienda Ospedaliero Universitaria Careggi Firenze Breast
Outcomes Evaluation Following Bilateral Breast Reconstruction Using Latissimus Dorsi Myocutaneous Flaps
 BREAST SURGERY Outcomes Evaluation Following Bilateral Breast Reconstruction Using Latissimus Dorsi Myocutaneous Flaps Albert Losken, MD, FACS, Claire S. Nicholas, MD, Ximena A. inell, MD, and Grant W.
BREAST SURGERY Outcomes Evaluation Following Bilateral Breast Reconstruction Using Latissimus Dorsi Myocutaneous Flaps Albert Losken, MD, FACS, Claire S. Nicholas, MD, Ximena A. inell, MD, and Grant W.
Citation for published version (APA): Benditte-Klepetko, H. C. (2014). Breast surgery: A problem of beauty or health?
 UvA-DARE (Digital Academic Repository) Breast surgery: A problem of beauty or health? Benditte-Klepetko, H.C. Link to publication Citation for published version (APA): Benditte-Klepetko, H. C. (2014).
UvA-DARE (Digital Academic Repository) Breast surgery: A problem of beauty or health? Benditte-Klepetko, H.C. Link to publication Citation for published version (APA): Benditte-Klepetko, H. C. (2014).
Vagus Nerve Stimulation (VNS)
 Vagus Nerve Stimulation (VNS) Date of Origin: 12/2008 Last Review Date: 06/27/2018 Effective Date: 07/01/2018 Dates Reviewed: 07/2010, 07/2011, 07/2012, 05/2013, 07/2014, 01/2016, 01/2017, 06/2018 Developed
Vagus Nerve Stimulation (VNS) Date of Origin: 12/2008 Last Review Date: 06/27/2018 Effective Date: 07/01/2018 Dates Reviewed: 07/2010, 07/2011, 07/2012, 05/2013, 07/2014, 01/2016, 01/2017, 06/2018 Developed
Infectious Complications Leading to Explantation in Implant-Based Breast Reconstruction With AlloDerm
 Infectious Complications Leading to Explantation in Implant-Based Breast Reconstruction With AlloDerm Minh-Doan Nguyen, MD, PhD, a Chen Chen, MS, b Salih Colakoğlu, MD, b Donald J. Morris, MD, b Adam M.
Infectious Complications Leading to Explantation in Implant-Based Breast Reconstruction With AlloDerm Minh-Doan Nguyen, MD, PhD, a Chen Chen, MS, b Salih Colakoğlu, MD, b Donald J. Morris, MD, b Adam M.
Author's response to reviews
 Author's response to reviews Title: Classifying breast cancer surgery: a novel, complexity-based system for oncological, oncoplastic and reconstructive procedures, and proof of principle by analysis of
Author's response to reviews Title: Classifying breast cancer surgery: a novel, complexity-based system for oncological, oncoplastic and reconstructive procedures, and proof of principle by analysis of
AESTHETIC SURGERY OF THE BREAST: MASTOPEXY, AUGMENTATION & REDUCTION
 CHAPTER 18 AESTHETIC SURGERY OF THE BREAST: MASTOPEXY, AUGMENTATION & REDUCTION Ali A. Qureshi, MD and Smita R. Ramanadham, MD Aesthetic surgery of the breast aims to either correct ptosis with a mastopexy,
CHAPTER 18 AESTHETIC SURGERY OF THE BREAST: MASTOPEXY, AUGMENTATION & REDUCTION Ali A. Qureshi, MD and Smita R. Ramanadham, MD Aesthetic surgery of the breast aims to either correct ptosis with a mastopexy,
Document Number: IC I. Length of Authorization. Dosing Limits
 Hyaluronic Acid Derivatives: Durolane, Euflexxa, Gel-One, GelSyn-3, GenVisc 850, Hyalgan, Hymovis, Monovisc, Orthovisc, Supartz/Supartz FX, Synojoynt, Synvisc, & Synvisc-One, TriVisc, Visco-3 (Intra-articular)
Hyaluronic Acid Derivatives: Durolane, Euflexxa, Gel-One, GelSyn-3, GenVisc 850, Hyalgan, Hymovis, Monovisc, Orthovisc, Supartz/Supartz FX, Synojoynt, Synvisc, & Synvisc-One, TriVisc, Visco-3 (Intra-articular)
Selected Operative Procedure Categories for KNHSS SSI Surveillance
 Selected Operative Procedure Categories for KNHSS SSI Surveillance Breast Surgery Excision of lesion or tissue of breast including radical, modified, or quadrant resection, lumpectomy, incisional biopsy,
Selected Operative Procedure Categories for KNHSS SSI Surveillance Breast Surgery Excision of lesion or tissue of breast including radical, modified, or quadrant resection, lumpectomy, incisional biopsy,
NIPPLE SPARING PRE-PECTORAL BREAST RECONSTRUCTION
 NIPPLE SPARING PRE-PECTORAL BREAST RECONSTRUCTION 42 yo female healthy athlete Right breast mass. Past medical history: none Family history: aunt with Breast cancer Candidates for nipple-sparing mastectomy
NIPPLE SPARING PRE-PECTORAL BREAST RECONSTRUCTION 42 yo female healthy athlete Right breast mass. Past medical history: none Family history: aunt with Breast cancer Candidates for nipple-sparing mastectomy
Advances and Innovations in Breast Reconstruction and Brest Surgery Presented by PCMC plastic surgeons
 Advances and Innovations in Breast Reconstruction and Brest Surgery Presented by PCMC plastic surgeons Options for reconstruction after mastectomy Implants Autologous tissue = from your own body: skin
Advances and Innovations in Breast Reconstruction and Brest Surgery Presented by PCMC plastic surgeons Options for reconstruction after mastectomy Implants Autologous tissue = from your own body: skin
Corporate Medical Policy
 Corporate Medical Policy Breast Surgeries File Name: Origination: Last CAP Review: Next CAP Review: Last Review: breast_surgeries 1/2000 8/2017 8/2018 8/2017 Description of Procedure or Service Mastectomy
Corporate Medical Policy Breast Surgeries File Name: Origination: Last CAP Review: Next CAP Review: Last Review: breast_surgeries 1/2000 8/2017 8/2018 8/2017 Description of Procedure or Service Mastectomy
Clinical Policy Title: Breast reconstruction following breast cancer surgery
 Clinical Policy Title: Breast reconstruction following breast cancer surgery Clinical Policy Number: 13.03.04 Effective Date: April 1, 2017 Initial Review Date: November 16, 2016 Most Recent Review Date:
Clinical Policy Title: Breast reconstruction following breast cancer surgery Clinical Policy Number: 13.03.04 Effective Date: April 1, 2017 Initial Review Date: November 16, 2016 Most Recent Review Date:
Advances in Localized Breast Cancer
 Advances in Localized Breast Cancer Melissa Camp, MD, MPH and Fariba Asrari, MD June 18, 2018 Moderated by Elissa Bantug 1 Advances in Surgery for Breast Cancer Melissa Camp, MD June 18, 2018 2 Historical
Advances in Localized Breast Cancer Melissa Camp, MD, MPH and Fariba Asrari, MD June 18, 2018 Moderated by Elissa Bantug 1 Advances in Surgery for Breast Cancer Melissa Camp, MD June 18, 2018 2 Historical
