August 2007 Check you have the most recent fact sheet by visiting
|
|
|
- Brittany Pearson
- 6 years ago
- Views:
Transcription
1 HEALTH RISK ASSESSMENT GUIDANCE FOR METALS HERAG MUTAGENICITY FACT SHEET 05 August 2007 Check you have the most recent fact sheet by visiting
2 HERAG FACT SHEET Page 2 of 27 Table of contents 1. Introduction Definition of Test Systems Commonly Applied in the Study of Metals and Metal Compounds Mechanisms of of Metals and Metal Compounds Response Profiles for Metal Genotoxicity Recommendations for a Testing Strategy for Metals and Metal Compounds References and abbreviations Annex I: HERAG /Genotoxicity and Carcinogenicity Workshop Annex II: Current EU Genotoxicity Testing Guidelines... 23
3 HERAG FACT SHEET Page 3 of Introduction The mutagenic and/or genotoxic properties of a substance are an important property upon which hazard classification is based within the context of the Existing Substances Regulation (ESR). Mutagenic properties will also be of fundamental importance with the adoption of REACH provisions within the pending Chemical s Policy, serving as a potential mandatory trigger for REACH Authorisation provisions. Although classification criteria may evolve with the expected adoption of the Global Harmonised System (GHS) for classification, criteria for mutagenicity classification should be similar to those presently in place within the EU. Mutagenic potential can also be of importance within Risk Assessments and affect the fashion in which dose response relationships are evaluated for other health endpoints. For example, genotoxic carcinogens can be presumed to exert effects without a threshold and to exhibit presume low dose linearity for the induction of effects. Similarly, reproductive impacts mediated by mutagenic effects upon germ cells may be assumed to lack thresholds and to also exhibit linear dose response at low exposure levels. This is in contrast to the presumed thresholds and No Observable Effect Levels that characterise reproductive or carcinogenic impacts mediated by non-genotoxic mechanisms. The mutagenic effects of metals and their compounds have been evaluated within the context of several EU Risk Assessments and were further the subject of a workshop convened in Hannover, Germany on November 2005 (see annex 1). The workshop assembled external independent experts from government and academia to address key issues associated with classification and risk assessment for both metal genotoxicity and carcinogenicity. In the presence of a small number of industry experts and observers, the external experts addressed a series of charge questions focussed upon testing strategies, classification criteria and risk assessment principles that might be appropriate for metals. The detailed deliberations of the external experts are described in workshop minutes, which may be obtained upon request from the authors of this fact sheet. As will be discussed, the experts concluded that testing strategies developed for the classification of organic substances (see annex 2) were not appropriate for metals and that alternate decision tree approaches for in vitro and in vivo genotoxicity testing were appropriate. The means by which test data could be interpreted and further work conducted for purposes of risk assessment were also reviewed. This analysis was undertaken with recognition that much of the experimental work on the mutagenic potential of metals and metal compounds has been conducted using soluble metal compounds. Issues of bioavailability, and their potential ramifications for read across in hazard identification and for quantitative risk assessment were recognised as important but beyond the scope of the current document. Rather, this testing strategy document seeks to identify test systems and procedures optimal for evaluating the mutagenic potential of substances that may induce effects through a set of indirect mechanisms atypical for mutagenic organic substances. 2. Definition of As defined by the Technical Guidance Document and Annex VI to Directive 67/548/EEC, a mutagenic effect is a permanent change in the amount or structure of the genetic material in an organism, resulting in a change of the phenotypic characteristics of the organism. Alterations may involve a single gene, a block of genes or a whole chromosome. A mutation in the germ cells of sexually reproducing organisms is of paramount concern since it may be transmitted to the offspring. A mutagen is thus an agent that gives rise to an increased frequency of mutations. The term genotoxicity is inclusive of mutagenicity but further encompasses a wider range of events indicative that a given substance has induced changes that may lead to mutations. Thus, genotoxicity assays may include endpoints such as the induction of DNA damage, DNA repair and/or recombination events such as those which give rise to sister chromatid exchanges. The frequency with which genotoxic changes result in mutagenic events is variable as a function of factors such as the efficiency and efficacy of the repair processes that reverse DNA damage. The distinction between genotoxicity and mutagenicity test results can be most important in weight of evidence evaluations with higher priority (all other factors being equal) being assigned to the results of mutagenicity tests.
4 HERAG FACT SHEET Page 4 of 27 Mutagens are classified under the directive into one of three categories and decisions leading to inclusion into these categories can be based upon the demonstration of genotoxic changes and mutagenic changes. Category 1 mutagens are substances known to be mutagenic to man. To place a substance in category 1, positive evidence from human epidemiology studies documenting mutagenic effects is needed. To date, no substance has been classified as a Category 1 mutagen and it is recognised that the collection of reliable data on the incidence of mutations in human populations, or on possible increases in mutation frequencies, Is extremely difficult. Category 2 mutagens are substances that should be regarded as if they are mutagenic to man. Substances are placed into this category based upon evidence that supports a strong presumption that human exposure to the substance may result in the development of heritable genetic damage. This determination is generally made from appropriate animal studies and other relevant (e.g. mechanistic) information. Supporting evidence from in vitro assays or positive results from host-mediated assays, can contribute to this classification. In vivo germ cell mutagenesis assays can be applied to demonstrate the appearance of affected progeny or a defect in a developing embryo. Examples of tests that provide such a demonstration include the specific locus mutation test, the heritable translocation test and the dominant lethal mutation test. Alternatively, in vivo assays may show relevant interactions with germ cell DNA. Assays for chromosomal abnormalities, including aneuploidy, provide evidence of mutagenic effects in germ cells. Other assays detect the ability of substances to interact with DNA, this interaction being an indirect indication of the possibility that mutations may be induced. Assays of this latter category include genotoxicity tests for sister chromatid exchange (SCE s), unscheduled DNA synthesis (UDS), covalent binding of a substance to DNA, or assaying other types of DNA damage (e.g. DNA strand breaks). Under most circumstances it would be anticipated that effects upon germ cell tissue would be accompanied by similar effects in the somatic tissue cells of mammals. Finally, in vivo assays may demonstrate mutagenic effects in the somatic cells of animals. Combined with supporting toxicokinetic data, or other information that demonstrates that a compound or its reactive metabolites reaches the germ cells, a presumption may be made that germ cells may be affected. Activity exerted in somatic cells may, in and of itself, be of relevance to carcinogenesis and is generally reviewed within that context. Category 3 mutagens are substances that cause concern for man owing to possible mutagenic effects. Evidence will be available from mutagenicity studies, but is insufficient for placement in category 2. Such substances are identified based upon positive results from in vivo assays that demonstrate either mutagenic effects in somatic cells or other cellular interactions relevant to mutagenicity in mammals. In vivo somatic cell mutagenicity assays include the bone marrow micronucleus test, metaphase analysis of peripheral lymphocytes, or the mouse coat colour spot test. The determination of whether a substance is capable of interacting with the DNA of somatic cells is provided by genotoxicity tests for SCE s, UDS, covalent binding to DNA, or other forms of DNA damage. Supporting data from in vitro mutagenicity assays assist in assigning this classification. It should be noted that a proportion of the assay methods mentioned in the directive are no longer in routine use. For reasons related to cost, animal welfare issues and the limited number of laboratories with the requisite level of expertise required to perform some assays, tests such as the mouse coat colour spot test or the heritable translocation test are seldom performed Test Systems Commonly Applied in the Study of Metals and Metal Compounds The majority of test data for many metals is actually for metal salts with high water solubility and thus more convenient to use in the conduct of testing. These salts need not be substances of commercial importance and extrapolation can be required if inferences are to be drawn regarding the properties of poorly soluble substances or the metal itself. Issues of bioavailability, and their potential ramifications for read across in hazard identification and when applied in quantitative risk assessment are important but beyond the scope of the current document. Assessments of the mutagenic properties of metals and their compounds are typically first based upon in vitro testing, with follow-up work using in vivo testing as appropriate. The following
5 HERAG FACT SHEET Page 5 of 27 summary describes the most commonly applied test systems in greater detail, along with some of the criteria that can be used to judge aspects of study quality. As noted above, in vitro and animal studies of metal genotoxicity have most often been conducted upon soluble compounds. Such studies tended to assume that counter ion effects are minimal, although this assumption was not always valid. For example, chromate salts of metals may elicit responses due to activity of the chromate anion (ATSDR 2000). Thus, whereas most compounds of lead are not bacterial mutagens, lead chromate is mutagenic for Salmonella typhimurium (Nestmann et al., 1979). Similarly, calcium chromate will produced response in the mouse lymphoma mammalian cell mutagenesis assay (Caspary et al., 1988). Halogenated metal salts are typically highly soluble and, for this reason, used in mutagenesis assays. However, halogens may, at sufficiently high concentrations, elicit gene and chromosome mutations (Seeberg et al., 1988; Wangenheim and Bolcsfoldi, 1988). In instances where such counter ion effects are suspected, the overall response profile must be examined to determine if other soluble metal salts also produce a similar profile of positive responses. In general, in vitro studies have usually been conducted in aqueous media buffered with components capable of complexing metals (e.g. carbonate formation from bicarbonate buffers). The speciation of the soluble metal salts at the time of addition to in vitro tests should thus be regarded as indicative of initial speciation only and may not represent the compound actually present in a given test system. The nature, type and biological relevance of the test system used for the study of the activity of inorganic compounds must be considered when assessing the relative weight of the evidence for or against mutagenic activity. A variety of endpoints can be assessed in vivo or in vitro, but are not equivalent with respect to assessment of mutagenic potential. The sensitivity and specificity of individual assays varies with different classes of compounds and should be considered when evaluating the significance of data from each assay type. High sensitivity assays will respond to a broad range of genotoxic substances and thus yield few false negative responses. Assay specificity is the converse of sensitivity and refers to the tendency of the assay to yield false positive results. A highly specific assay will yield few false positives. The ideal assay has both high specificity and sensitivity but this ideal is seldom achieved. High sensitivity is often achieved with a loss of specificity. The sensitivity and specificity of many in vitro test systems, and their ability to successfully predict carcinogenic potential for the rat carcinogens has recently been questioned by Kirkland et al (2005) who have observed that, at least with respect to rodent carcinogens and noncarcinogens, some assays for genotoxicity may have inadequate specificity and yield significant false positive responses for noncarcinogens. Sensitivity and specificity are not addressed further in this section except to note specific issues of relevance to the interpretation of assay results for metals and their compounds. Assays Mutagenic effects can be detected through the observation of induced gene and/or chromosome changes. Gene mutation assays detect changes in the primary sequence of DNA that result in altered protein production and/or function. The detection of mutagenic effects in somatic cells is an important factor in the assessment of cancer hazard. Germ cell alterations indicate risk of adverse heritable genetic changes. Although some mutagenicity assays can be conducted in vivo, in vitro mutagenicity tests are more commonly applied in the initial assessment of potential mutagenic properties. The most common test systems study cultured bacteria or mammalian cells. Properly conducted assays employ both positive and negative controls, include metabolic activation preparations, evaluate the impact of multiple concentrations of the test substance, and seek to define dose-dependent increases in mutation frequency in the absence of high levels of cytotoxity. Cell survival is most commonly monitored in mammalian cell test systems via colony formation assays. Dose dependent induction of mammalian cell mutations in the absence of high levels of cytotoxicity is regarded to provide the most relevant indication of mutagenic potential that can be obtained from in vitro studies. The acceptable level of cytotoxicity associated with mutagenic response varies with the assay employed but is typically on the order of 50% or less. Different cell types or gene loci vary in their sensitivity to genotoxic agents for mechanistic reasons. For example, some test systems are primarily sensitive to specific nucleotide base change alterations - only a limited range of mutagenic changes may thus be detected. This limitation is particularly relevant to metals and reverse mutation assays in bacteria (e.g. the Ames test with Salmonella typhimurium). This assay assesses the ability of a substance to reverse the impacts of a prior known mutation that has inactivated a gene. Different strains of S. typhimurium, each with a specific reverse mutation requirement, must thus be used in the study of a given substance to ensure sensitivity to a spectrum of genetic changes. Only a few
6 HERAG FACT SHEET Page 6 of 27 metal compounds are suspected to produce direct DNA damage, but a number are suspected to express mutagenic potential via indirect pathways. For example, the most commonly used strains of Salmonella have poor sensitivity to genetic changes associated with the action of oxygen radicals which are in turn suspected to be the indirect mediators of genotoxicity for some metals. False negative responses will thus result if the reverse mutation assay has limited sensitivity to the spectrum of genetic changes that occur as a result of exposure to metal compounds. Interpretation of bacterial mutagenesis data for metals can also be difficult if it is not known if the metal under study is taken up by bacteria a number of metals are actively excluded from uptake. As such, the sensitivity of bacterial systems to metal genotoxicity appears to be low. studies with mammalian cells are usually forward mutation assays that assess the ability of a substance to inactivate a cell enzyme such as thymidine kinase (TK) or hypoxanthine phosphoribosyl transferase (HPRT). Mutations occur that inactivate the target enzyme render the mutant cells resistant to specific substances that are transformed to toxic metabolites by the functional form of the enzyme. Forward mutation assays can be sensitive to a broad array of genetic changes, including point mutations, small deletions and large deletions. The sensitivity of forward mutation assays to very large DNA deletions is suspected to be limited by the presence of essential genes near the target gene evaluated in many mutagenesis assays. Large deletions that inactivate the target gene may thus also result in damage to genes that are essential to cell survival. If cell viability is lost, the induction of large deletions may not be detected. Of the preceding mutation assays mentioned, loss of activity at the TK locus may be able to tolerate larger deletion events without eliminating cell viability than loss of activity at the HPRT locus. Genetically modified test systems are sometimes used to assess the mutagenic potential of substances suspected to primarily cause large deletions in DNA. The cells used in such test systems contain a bacterial gene (e.g. guanine-hypoxanthine phosphoribosyl transferase or gpt) inserted in a chromosome region not adjacent to essential genes. Mutagenic events affecting large regions of DNA that also eliminate activity in the bacterial gene can thus be detected with greater ease. While genetically modified cells may have sensitivity to a broader subset of genetic alterations, assays using such cells have been only recently developed and have not been as extensively validated as traditional forward mutation assays and assay specificity, sensitivity remains to be determined. Moreover, it would appear that, at least in some instances, loss of gene activity may result from events that affect gene expression (e.g. methylation) as opposed to changes in the primary sequence of DNA (Klein, 1994). However, given these limitations, such assays have been suggested to be sensitive to some of the indirect mechanisms of mutagenicity that may be associated with some metal compounds (Ariza and Williams, 1996). Each mutation assay has it s own specific recommended protocols covering treatment conditions, mutation expression time, mutant selection and acceptable rates of background mutation frequency. Detailed specification of the technical nuances associated with each type of assay will not be provided here. Chromosome Changes Alterations in DNA sequence also result from changes in the structure of chromosomes and alterations in the amount of DNA from changes in the number of chromosomes. Using cells exposed in vitro, or cells (e.g. bone marrow from animals or peripheral blood lymphocytes from humans) harvested and placed in cell culture after in vivo exposure, metaphase chromosomes can be routinely screened for numerical and structural chromosome aberrations (CA). The frequency of CA in peripheral blood lymphocytes was the only genotoxic biomarker recently suggested to correlate with future cancer risk in humans. An approximate twofold elevated cancer risk has been estimated among subjects with high frequencies of CA (Hagmar et al. 2004a). CA are divided into two types: 1) chromatid-type aberrations, which affect only one chromatid; and 2) chromosome-type aberrations, which affect the same locus on two sister chromatids. Agents that produce double strand breaks, such as ionising radiation, produce mainly chromosome aberrations and these are independent of DNA replication. Chromosome-type aberrations represent unrepaired or incompletely repaired double strand breaks. Agents that produce other DNA lesions (adducts, crosslinks, pyrimidine dimers, single strand breaks) produce chromatid-type aberrations and these are DNA replication-dependant and form in vitro during S-phase. Lesions can also result from cellular events such as topoisomerase action or repair of endogenous lesions. Although they are induced by different mechanisms, both types are reported to be associated with increased cancer risk although the correlation with chromosome-type lesions may be stronger (Hagmar et al., 2004b). The strength of the correlation between different classes of
7 HERAG FACT SHEET Page 7 of 27 chromosomal changes and endpoints such as cancer is likely to vary as a function of the class of substance being tested and the mechanism(s) responsible for the induction of CAs at present the strength of the correlation for metals is not known. Chromosome-type aberration types include: chromosome breaks, ring chromosomes, marker chromosomes, and dicentrics. Chromatid-type aberrations include chromatid breaks and chromatid exchanges (these are not the same as sister chromatid exchanges). The chromosome gaps that were sometimes reported in earlier cytogenetic studies are now thought to be artefacts (often the result of the staining procedures used) and are not in and of themselves regarded as reliable indicators of mutagenic effects. In the 1990 s attention was focused on CA s produced under extreme conditions such as high osmolarity, high ionic strength and low ph. Such conditions are obviously germane to testing conducted with high concentrations of metal salts. These conditions, as well as some non-genotoxic chemicals (e.g. aphidicolin, an inhibitor of DNA polymerase) caused non-specific interference with cellular functions that could lead to CA (Galloway et al., 1998). It was found that compounds that induced CA at <50% cytotoxicity were more likely to be genotoxic in other assays than compounds that only caused CA at >50% toxicity. It is now recommended that chromosome aberration assays should incorporate some measure of cytotoxicity to aid in the assessment of the relevance of study results, although this is often not done adequately (Komissarova et al., 2004). Finally, there is often a dose-related delay in appearance of aberrations, due to cell cycle delay, such that dose-response experiments assayed at one time point may not yield a dose-related effect (Kirkland 1998). Some agents thus require assessment of CA s after variable post-treatment recovery times. Conversely, decreased cell cycle times may be associated with an increase in aberrations and should be considered in the evaluation of metals known to be mitogens. Cells harvested from the bone marrow or other in vivo sites tend to accumulate spontaneous aberrations during successive cell divisions in vitro. Accordingly, increases in aberrations associated with increased mitotic indices or decreased cell cycle times must be interpreted with caution. The in vitro micronucleus (MN) assay was developed as a short-term screening assay for animal and human genotoxic exposures and cytogenetic effects. In comparison with CA, the scoring of micronuclei (MNs) is simpler, less expensive and less time consuming. In principle, the MN assay can be expected to be more sensitive than the CA assay because of the increased statistical power when large numbers of cells are analysed (1000 per culture is recommended) compared with a hundred or a few hundred cells usually scored for CA. The in vivo bone marrow MN test is now accepted as being equivalent to the bone marrow CA test in identifying clastogens, and the in vitro MN assay shows good potential to replace the in vitro CA test (Kirsch- Volders et al., 2003b). MN are defined as small, membrane bound DNA-containing cytoplasmic bodies formed during cell division by loss of either acentric chromatin fragments or whole chromosomes. The two basic phenomena leading to the formation of MN are chromosome breakage (double strand breaks associated with clastogenesis) and dysfunction of the mitotic apparatus that impairs chromosome segregation during mitosis/meiosis to result in the gain or loss of entire chromosomes (aneugenesis). Aneuploidy is defined as a change in chromosome number from the normal diploid or haploid number other than an exact multiple (polyploidy). MN produced as a result of clastogenesis contain acentric chromosome or chromatid fragments and MN associated with aneuploidy contain whole chromosomes that lag behind in anaphase and are left outside the daughter nuclei in telophase. Because cell division is necessary for the generation of MN, the cytokinesis block micronucleus (CBMN) assay is recommended for use with human lymphocytes and is optional for use with cell lines (Kirsch-Volders et al., 2003b). In this assay, cultures are treated with cytochalasin B, an inhibitor of actin polymerisation. Cytochalasin B prevents cytokinesis but allows nuclear division, resulting in cells with multiple nuclei. It is thus possible to identify cells that have divided once, because they contain two nuclei. By restricting scoring of micronuclei only to cells with two nuclei, problems caused by treatment-related variations in cell cycle times are reduced. Furthermore, the scoring of cells with two nuclei helps ensure that only events in viable cells are scored and that apoptosis has not occurred. When applied to cell cultures, cytochalasin B blocks also help ensure that the MN scored were induced after the treatment of cells with the test material. Attempts have been made to distinguish between numerical (aneugenic) and structural (clastogenic) changes that can occur following treatment with a test compound. Currently, the most widespread and reliable assays identify whole chromosomes in MN by labelling their kinetochores or centromeres. Kinetochore proteins can be identified by immunofluorescence with CREST anti-kinetochore antibody
8 HERAG FACT SHEET Page 8 of 27 reactive with conserved human centromere proteins (labelled MN are termed K+). Alternatively, centromeric DNA sequences can be identified by fluorescent in situ hybridization (FISH) using repetitive DNA sequence probes (labelled MN are termed C+). When these techniques are used, the in vitro MN assay is considered a suitable alternative to in vitro chromosome aberrations tests for detection of clastogenic and aneugenic agents. As a generalization, since metals have a strong tendency to bind to cellular proteins, and could alter antibody binding to proteins, FISH may be preferable to CREST staining in the study of metal-treated cells. It is recommended that this assay should be performed under conditions of high survival (>50% or more viable cells). It is also recommended that markers for apoptosis and necrosis be included, particularly if cells are being evaluated in vitro after treatment in vivo (Kirsch-Volders et al., 2003b). At least 2000 cells should be scored per concentration (1000 per culture, in duplicate). Genotoxicity tests providing indications of potential mutagenicity A number of test systems do not actually assess mutagenic effects and are best regarded as indicator tests for genotoxic potential. These test systems monitor cellular changes thought to occur in association with mutagenic change, but the changes measured are not in and of themselves proof that mutagenic change has occurred. In a number of instances, positive responses in indicator tests can occur via mechanisms that are not the same as those for mutagenic change. As such, results from such indicator test systems are often assigned secondary priority in weight of evidence genotoxicity assessments. A variety of indicator tests exist only those that have been applied most frequently in the study of metals are discussed here. Sister Chromatid Exchange Sister Chromatid Exchange (SCE) frequency has been commonly used as an index of chromosomal stability after treatment with mutagens. SCE s are induced during DNA replication, most likely as a result of a highly conserved homologous recombination (HR) mechanism that uses the nascent sister chromatid to repair potentially lethal DNA lesions during replication (Sonoda et al., 1999). SCE induction is stimulated by DNA damage, but also by replication fork arrest (Matsuoka et al., 2004). Thus, one cannot conclude from SCE data alone that an agent causes DNA damage, particularly if the SCE response is only seen in the toxic dose range where replication fork arrest might be occurring. The toxicity of metal concentrations inducing SCE s, and whether DNA replication is inhibited at those doses, must be ascertained in order to interpret SCE induction data properly. The most relevant responses will be those that: 1) occur at concentrations with minimal toxicity; 2) exhibit dose dependence; and 3) result in a greater than two-fold increase in SCE frequency above baseline. Single Cell Electrophoresis (COMET assay) In the Comet assay, individual cells are electrophoresed, stained and visualized microscopically for evidence of treatment induced damage to DNA (Collins, 2004). The alkaline version of this assay (conducted at ph>13) is accepted as the most sensitive protocol since it will detect both DNA strand breaks and alkali labile DNA adducts (Collins, 2004). When there is DNA damage, stained cells appear as a nucleus (the head ) with a tail like that of a comet. This tail consists of broken DNA strands that are released by the cell lysis and DNA unwinding stages of the procedure. Undamaged DNA does not migrate and remains in the head. The parameters measured in the Comet assay are tail length, % DNA in tail or tail moment (% DNA in tail x Tail length). Tail moment is regarded as the best index of effect. The Comet assay detects both single and double strand DNA breaks as well as alkali-labile sites. As a consequence of sensitivity to DNA stand breaks, Comet assay results often correlate with those of the chromosome aberration assay. The assay protocol can be readily applied to cells treated in vivo and, as a result, has been used to study multiple tissues from treated animals and human cells that can be harvested by relatively non-invasive procedures. A number of human biomonitoring studies have thus been conducted, but the results of such studies have been difficult to interpret. The effect of tobacco smoking on the Comet assay has produced mixed outcomes, with 9 reports finding increased damage in smokers and 16 reports not finding such an effect (Faust et al., 2004). Although it can be argued that tobacco smoke represents low level chronic exposure to mutagens and that consistent positive responses would not be expected, the assay
9 HERAG FACT SHEET Page 9 of 27 has demonstrated responsiveness to other substances (e.g. pesticides) that are either weakly mutagenic or non-mutagenic in other test systems (Faust et al., 2004). Interpretation of Comet assay results with respect to mutagenic or carcinogenic risk in humans is further complicated by the impact of age, stress, nutrition and exercise upon assay results and the extreme degree of inter-individual variability that generally exceeds the differential between the exposed and control subjects in most studies (Collins, 2004). The mixed performance of the Comet assay in human biomonitoring validation studies is likely, at least in part, to be due to the mixed exposures and uncontrolled confounding characteristic of any human biomonitoring study. Assay responses may also reflect sensitivity to biological effects not necessarily relevant to genotoxicity. Single strand breaks are quickly repaired and are not regarded as significant promutagenic lesions. Alkali-labile sites can result from some DNA adducts which spontaneously depurinate (or depyrimidinate) leaving AP sites which are cleaved by alkali. However, excision repair of adducts can create AP sites and breaks as intermediates. In order to avoid false positive responses, Henderson et al. (1998) suggest that the concentration of test substance should produce >75% viability since necrotic cells also display DNA damage (Fairbairn et al., 1996). For studies conducted using cells derived from tissues exposed in vivo, histopathological examination of the treated tissues is generally regarded as the optimal means of ensuring that excessive toxicity and necrosis has not been produced (Burlinson et al., 2007). Positive Comet assay results can also be produced by the induction of apoptosis, programmed cell death that results in DNA fragmentation into segments of 180 base pairs, and apoptosis can in turn be induced by non-genotoxic agents. Extremes of ph, ionic strength and osmolarity, and fast ligand binding may induce apoptosis (Henderson et al. 1998; Choucroun et al., 2001) and a number of metals have been found to modulate apoptosis induction. Thus, it is advisable to evaluate apoptosis in the population of cells being used in the Comet assay although this is not usually done. Comet assay results reflective of apoptosis can be detected by evaluating the distribution of DNA damage amongst cells (e.g. by calculating the tail moment in each cell). If apoptosis is occurring, a bimodal distribution will be seen with a population of cells that show no damage and a population of cells that show extensive damage. This occurs with compounds like dexamethasone and camptothecin (Lee et al., 2003). Alternatively, or in addition, apoptosis induction can be monitored using an Annexin V affinity assay or caspase-3 activation assay to detect apoptosis induction within the target cell population (Roser et al., 2001; Komissarova et al., 2004). When the Comet assay is performed in combination with controls for apoptosis, it has been reported to have a higher specificity than the Comet assay alone (Lee et al., 2003). Application of the Comet assay under controlled laboratory conditions has produced more reproducible and consistent results than those produced by human biomonitoring studies (Sasaki et al., 2000). In a test of 208 chemicals, 110 out of 117 rodent genotoxic (Ames + ) carcinogens gave positive Comet assay results compared to 6 of30 tested noncarcinogens. For Ames (-) carcinogens, less than 50% were positive in the Comet assay, suggesting that the assay will have understandable limitations in the detection of nongenotoxic carcinogens. Moreover, while responses were observed in vivo in tissues that were known to be the target tissues for carcinogenesis, greater amounts of DNA damage are at times observed in non-target tissues Mechanisms of of Metals and Metal Compounds Compounds of a number of metals (e.g. Pb, Zn, Cu, Cd) have been evaluated for mutagenicity and/or genotoxicity in vitro and in vivo. Observation of mutagenicity in vitro has been common, but for a number of metals responses appear to be elicited in the absence of direct metal interaction with DNA. This has stimulated research on indirect pathways by which metals could produce an effect. A variety of hypotheses have been put forward to explain mutagenic and genotoxic responses induced by metals and their compounds and are summarized here. These hypotheses are not mutually exclusive; different mechanisms may underlie the activity of different metals and/or a single metal may have several potential mechanisms by which effects might be induced. A number of these mechanisms are, however, predicated upon the assumption that some metals may be capable of substituting for essential metals involved in key physiological processes. As such, this mechanistic aspect of metal genotoxicity is unlike that responsible for the activity of most genotoxic organic substances. The importance of oxidative damage to DNA in mutagenesis and carcinogenesis has been extensively studied with respect to several metals. Oxygen is present as 20% of the atmosphere and is the terminal oxidant for aerobic organisms. All forms of aerobic life are subjected to oxidant pressure from molecular
10 HERAG FACT SHEET Page 10 of 27 oxygen and reactive oxygen species (ROS). Most of the potentially harmful effects of oxygen are due to the formation and activation of ROS. Oxidative stress can be defined most simply as the imbalance between the production of ROS capable of causing cellular damage and the body s antioxidant defences. ROS include singlet oxygen, hydrogen peroxide (H 2 O 2 ), superoxide radical (O 2- ), nitric oxide (NO), organic oxyradicals, and organic peroxyradicals. Many of these are free radicals (i.e. molecules with one or more unpaired electrons and therefore unstable and highly reactive). Seeking stability, radicals attack nearby molecules to obtain another electron, causing damage to the molecule. If free radicals are not inactivated, their chemical reactivity can damage all cellular macromolecules, including proteins, carbohydrates, lipids and nucleic acids. Upon reaction with DNA, ROS have been documented to produce more than 30 different base adducts, DNA strand breaks and crosslinks as well as various amino acid, protein and lipid addition products (Marnett and Burcham, 1993). Although 8-OHdG is only one of a plethora of oxidized bases, it has been useful as a marker for oxidative DNA damage. Hydroxyl radical attack on the deoxyribose in DNA induces strand breaks that might be expected to contribute to the deletions within DNA and structural chromosome changes (Joenje, 1989). Free radicals and other reactive oxygen species can be derived from normal metabolism in the human body as well as from external sources, such as exposure to radiation, ozone, cigarette smoking, certain drugs, pesticides, air pollutants and industrial chemicals. Mitochondria represent the greatest source of cellular oxidants because their electron transport system consumes approximately 85% of the oxygen utilized by the cell (Shigenaga et al., 1994). 1-2% of oxygen molecules are converted to superoxide by autooxidation [one electron transfer to O 2 (Sohal and Dubey, 1994)]. Through dismutation, this is the major source of H 2 O 2 in cells, although smaller amounts of ROS are also contributed by other oxidant-producing systems (cytochrome p450, peroxisomes, NO synthetase, and cytoplasmic oxidases). Levels of oxidative damage to mitochondrial DNA are at least 10-fold higher than to nuclear DNA in the same cell, a fact consistent with the higher evolutionary mutation rate of mitochondrial DNA compared with nuclear DNA (Shigenaga et al., 1994). H 2 O 2 itself does not react with DNA, but requires a metal ion that can produce the DNA-damaging hydroxyl radical via Fenton reactions with H 2 O 2 (Joenje, 1989). Since iron and copper ions are powerful Fenton agents generating hydroxyl radicals, a complex system of storage and transport proteins exist to sequester these ions. Iron bound to ferritin, its storage form, or transferrin, its transporter, will not take part in Fenton reactions (Halliwell and Gutteridge, 1989). However, if superoxide escapes dismutation, it too can damage DNA. A favourable condition for uncontrolled oxidative reactions exists in cell membranes because of their unsaturated lipids. Mutagens such as malondialdehyde and unsaturated aldehydes (e.g. acrolein) can be formed endogenously as a result of lipid peroxidation. These compounds modify DNA bases with propano and etheno adducts (Marnett, 1994). Among these adducts so far found as endogenous DNA lesions, the acrolein- and the crotonaldehyde-derived 1,N2-propano deoxyguanosines are most ubiquitously detected at relatively high levels in tissue DNA (Nath and Chung, 1994). It is suggested that these adducts could represent the main DNA damage resulting from lipid peroxidation. It stands to reason that agents increasing lipid peroxidation in cells should increase the abundance of malondialdehyde, acrolein, and their DNA adducts. In many cases, the conclusion that free radical production is part of a genotoxic mechanism for metals follows the observation of increased amounts of free radical damage products, particularly markers of lipid peroxidation. It is important to consider that lipid peroxidation inevitably accompanies cell death from any cause. Metals may cause oxidative stress as a result of cell death, or as a result of Fenton-type reactions with H 2 O 2. Mitochondria are also centers of heme production and use, have naturally high metal concentrations and as such may be sensitive targets for metal toxicity. More research is needed to understand the chemical and biochemical determinants of metal interactions with cellular oxidants. For example, does a specific metal ion cause a Fenton reaction or does it displace iron from some molecules, resulting in iron-induced Fenton reaction? Do some metals affect mitochondrial function to produce more ROS and, if so, does this occur at metal concentrations that are not lethal to the cell? Several authors have conducted studies demonstrating that mutagenic and genotoxic effects of metal compounds could result from indirect oxidative mechanisms in vitro (Ariza and Williams, 1998; Yang et al., 1999; Vaglenov et al. 1998; Roy and Rossman, 1992) and in vivo (Huang, 1988; Fracasso et al., 2002; Valverde et al. 2001) although the specific mechanism speculated to underly the effects of different metals varies.
11 HERAG FACT SHEET Page 11 of 27 Other indirect mechanisms may be important in the consideration of metals. Hartwig et al (1994) postulated that some metals may interfere with the fidelity of DNA repair and through this mechanism increase the effects of other genotoxic substances. Subsequent studies have confirmed that metals such as cobalt, arsenic, nickel and cadmium interfere with base and nucleotide excision repair, even though they may affect different steps of the respective repair systems and act by different, not yet completely understood mechanisms. The activity of some metals could be mediated by their unique ability to substitute for essential metals that are involved in DNA repair or replication. Potential target molecules for some metal ions are socalled zinc finger structures in DNA repair proteins (Hartwig, 2001). Such proteins have structural motifs containing zinc that might be replaced by other metals with a resulting alteration in DNA repair efficacy. However, different zinc finger proteins exhibit sensitivity towards different toxic metal ions and the resulting complexity of potential effects has proved difficult to dissect (Hartwig and Schwerdtle 2002; Hartwig et al., 2003). Although the precise mechanism of action may not be understood, metal ion induced effects upon DNA repair can be seen at low, non-cytotoxic concentrations and can greatly reduce the repair of DNA adducts induced by substances such as benzo[a]pyrene. Finally, nickel, arsenic, and cobalt ion have been observed to interfere with cell cycle progression and cell cycle control following ultraviolet C radiation, a presumed indication of interference with excision repair of UV-induced DNA damage (Hartwig et al., 2002). Of potential particular significance are specific inhibitory effects of some metal ions at low concentrations with plausible physiological relevance. For example, arsenic induced effects have been observed in the range of 10 nm arsenic concentration. While an impact of metals upon mutagenesis through oxidative stress and/or decreased repair fidelity cannot be precluded, the lesions thus generated (adducts or base pair changes) would most likely elicit responses in assays for point mutations. Evidence for point mutations induction in response to treatment with many metal compounds is often quite limited, but few studies have evaluated the combined impact of treatment with metal compounds and direct-acting genotoxic agents. The relative importance of either pathway in producing mutagenic effects in vitro or in vivo is thus difficult to judge. Moreover, it is unclear if either pathway is operative in inducing chromosomal changes frequently observed after treatment with metal compounds. Novel mechanisms for clastogenic activity have recently been proposed. Thier et al (2003) examined micronucleus induction by lead and mercury salts in cultured V79 hamster cells. Dose dependent induction of micronuclei was observed and additional studies suggested that the induced micronuclei were entire chromosomes as opposed to chromosome fragments. Interference with function of the mitotic apparatus, most likely mediated by metal specific binding to mitotic spindle proteins, thus represents yet another indirect pathway through which metals might act. Moreover, it would suggest that some metals may preferentially induce aneuploidy, as opposed to chromosome fragmentation, to yield micronuclei Response Profiles for Metal Genotoxicity The genotoxicity of numerous metal compounds has been examined. For many metals and their compounds, testing has been extensive, but contradictory reports have appeared in the scientific literature. The response profile for metals that are the subject of ongoing or recently completed EU risk assessments is presented in the following table in an attempt to summarize the overall weight of evidence judgements of these risk assessments. The table indicates the metal ion tested, along with a summary evaluation of whether or not the responses elicited within in vitro test systems required physiologically relevant concentrations of metal ions. Most studies have been conducted using soluble metal salts (e.g. acetates, chlorides, nitrates), but the broad range of compounds actually tested is not presented in the interests of simplicity and due to the presumption that the metal ion is the moiety responsible for any activity observed. As a generalisation, all metals appear to elicit genotoxicity in one or more in vitro test systems. Summary evaluations are offered for in vitro testing for point mutations in mammalian cells and cytogenetic alterations (chromosome aberrations or micronucleus induction). testing in bacteria is negative for most of the metal compounds tested and is thus not included in this summary evaluation. However, several metal compounds, such as tetravalent platinum compounds, rhodium compounds and hexavalent chromium salts, are reported in the scientific literature to be bacterial mutagens (Lantzsh and Gebel, 1997; Bunger et al., 1996). Results obtained in the Comet assay and in vivo cytogenetic assays are also listed. Finally, summary evaluations are offered of the carcinogenic potential for both animals and humans.
12 HERAG FACT SHEET Page 12 of 27 For most metals, the overall profile that emerges is complex and lacks consistency. A portion of this inconsistency is likely derived from older studies that are poorly reported and/or of probable low quality. As a generalisation, tests for point mutations in mammalian cells are usually negative, but this finding must be interpreted with caution since some indirect mechanisms of action (e.g. through reactive oxygen species) could induce large deletions not efficiently detected by most point mutation assays. Assays for in vitro cytogenetic changes are usually positive, with some indications that aneuploidy induction may be common. The Comet assay provides either positive or equivocal results for most metals, although many of the studies conducted have not included adequate controls for cytotoxicity or apoptosis.. In vivo cytogenetics assay results are generally less definitive than in vitro studies, with a tendency for positive findings to result from routes of administration such as i.p. injection and negative studies to result from studies conducted via oral administration or inhalation exposure. Weight of evidence evaluations are thus needed to ascertain the physiological relevance of i.p. studies and the adequacy of oral or inhalation dosing regimens for achieving adequate dose to target tissues. The nature of such weight of evidence evaluations was extensively discussed during the external experts workshop (see annex 1) and will be reviewed in the next section on recommendations for a genotoxicity testing strategy for metals. The carcinogenicity evaluations listed are generally the findings of recent risk assessments and/or IARC although it is inappropriate to assume an obligatory linkage between carcinogenicity and genotoxicity for all metals. Summary Result of Genotoxicity Testing and Carcinogenicity Evaluations for Selected Metals 1 Metal Mutagenesis in vitro Cytogenetics COMET in vivo Cytogenetics Cancer Animal/Hum Cd (phys) +/- +? -? +/+ Co (phys)? /- Cu (not phys) +/- +? - -/- Ni (phys) soluble insoluble Pb (not phys) +/- +/- +/- +/- +/-? Zn (phys) +/- + NT +/- -/-? 1 Test results are defined as follows. (-) indicates that most tests are negative, (+/-) indicates that responses are inconsistent with both positive and negative results, (?) indicates that test result are equivocal, (-?) Indicates that results are generally negative with some indications of a positive response. NT indicates that the endpoint has not been tested. Finally, a summary appraisal if offered of whether the responses observed were obtained using test concentrations with probable physiological relevance (phys) or concentrations unlikely to be achieved via normal routes of exposure (not phys.) Note that the preceding matrix does not include biomonitoring studies of occupationally - or environmentallyexposed humans. The results of such studies will be discussed in the companion HERAG fact sheet on Carcinogenicity NT -/+ +/+
13 HERAG FACT SHEET Page 13 of Recommendations for a Testing Strategy for Metals and Metal Compounds Current base set strategies for genotoxicity testing (see annex 2) start with assays for bacterial mutagenesis. Bacterial mutagenicity tests appear to have little utility for the testing of metals (test results are almost always negative) and should be replaced with mammalian cell test systems. However, the indirect mechanisms suspected to mediate metal genotoxicity may induce large deletions for which the sensitivity of gene mutation assays such forward mutation at the HPRT locus may be limited. Large scale deletions, while inactivating the HPRT gene, may also affect neighbouring genes essential for cell survival and thus go undetected. Considerations should therefore be given to use of forward mutation assays with demonstrated sensitivity to both point mutations and large deletions. The mouse lymphoma test system, assaying mutations at the thymidine kinase gene, has been suggested to be sensitive to both types of lesions. Small point mutations result in rapidly growing mutant colonies. Mutations that inactivate the thymidine kinase gene via large deletions result in mutants with impaired growth capacity that form slow growing small colonies. Comparison of small vs large mutant colonies can thus provide an indication of the size of the genetic lesions responsible for mutagenesis. Alternatively, genetically engineered cell lines with bacterial gene inserts (such as the gpt gene) appear capable of detecting both large and small scale mutagenic events that inactivate the gpt gene. These newer test systems are not, however, yet validated or extensively characterised with respect to assay sensitivity or specificity. Epigenetic events (e.g. DNA methylation) may also result in loss of gpt activity (Klein, (1994) and at least one metal (nickel) has been found to produce positive assay responses via reversible alterations in transgene methylation as opposed to the induction of actual mutations (Kargacin et al., 1993). Verification that DNA sequence changes have occurred may thus be required for definitive interpretation of positive results from some of these newer assays. Tests for chromosome aberrations or micronucleus induction are currently recommended as part of base set testing and appear to be sensitive to the effects of metals. Retention of testing for these endpoints is thus indicated in the in vitro base set. Assays for micronucleus induction may be the simpler and more rapid assay for assessing cytogenetic damage and if coupled with kinetochore staining can further serve to assess aneuploidy induction in addition to chromosome damage. Although not yet ready for routine use, it is expected that assays for effects upon DNA repair might be both possible and useful in the near term. However, for the time being, such assays remain highly specialized and are not yet amenable for routine screening purposes. A tiered testing strategy is depicted in Figure 1 overleaf and can be applied for purposes of both hazard identification (i.e. classification) and risk assessment. The proposed scheme would conduct in vitro testing for gene mutations and chromosomal changes/micronucleus induction. However, based upon both the high prevalence of positive in vitro data for metals, and the theoretical premise that metal ions will bind to multiple cellular targets and induce genetic changes via indirect mechanisms, many metals compounds would test positive in the recommended in vitro base set. An in vivo follow-up strategy is thus also defined which can, on a case by case basis, be used to assess the relevance of mutagenic potential that may be suggested by in vitro testing. In vivo follow-up strategies would be guided by the endpoints suggested to be relevant by in vitro testing. Thus, observation of cytogenetic changes in vitro should be followed up by testing for cytogenetic effects in vivo etc. The conduct of gene mutation assays has historically been a challenge in that the range of tissues that could be routinely examined for was limited. However, with the growing sophistication and acceptance of mutagenesis assays in transgenic animals, in vivo follow-up for gene mutation endpoints will be increasingly feasible and accepted. Consideration may also be given to the prioritization of follow-up in accordance with the relevance of the concentrations required to produce effects in vitro to the concentrations of metal that can be tolerated by in vivo systems. For example, if µm concentrations of a metal are the maximum that can be tolerated in vivo then priority might be assigned to evaluated effects induced by µm (as opposed to mm) concentrations in vitro.
14 HERAG FACT SHEET Page 14 of 27 Start in vitro tests Gene Mutation Assay sensitive to point and large mutations (e.g. mouse lymphoma assay) Mammalian cell (preferably human) micronucleus test All tests (-) Yes No further testing One or more (+) results COMET assay if target tissue(s) need to be determined Toxicokinetic evaluation of relevant tissues and exposure routes for subsequent in vivo tests Hazard Classification Mutagenesis test in relevant tissue of transgenic animals if in vitro point mutation test was (+) Classification in vivo tests In vivo MN test in relevant tissue if in vitro MN test was (+). Distinguish between structural and numerical chromosome effects. Mechanistic Studies Dose response for micronucleus or gene mutation induction in relevant tissues If in vivo tests (+), further studies with relevant exposure routes and endpoints are required In vivo dose response for ROS metabolism effects Dose response for DNA repair effects: 1. in vitro 2. in vivo Dose response comparison between mutagenic effects and putative mechanisms FIGURE 1: PROPOSED MUTAGENICITY TESTING STRATEGY FOR METALS AND METAL COMPOUNDS
15 HERAG FACT SHEET Page 15 of 27 Understanding of the target organ specificity and toxicokinetics for a specific metal would facilitate the identification of in vivo tissues that should be selected for study. Preferred tissues for testing would be those known to experience high concentrations of metal ions and/or to be the targets of carcinogenesis. For example, assays for micronucleus induction in bone marrow cells would be relevant if the metal to be tested is known to affect bone marrow or to attain concentrations adequate for producing a meaningful test result. Knowledge of metal toxicokinetics and target organ specificity would also assist in assessing the routes of exposure that might be relevant for testing. Inhalation exposure would be appropriate for pulmonary toxicants, oral exposure for agents impacting the gastrointestinal tract etc. Whereas routes of administration such as intraperitoneal or intravenous injection may be appropriate so as to maximize the sensitivity of assays for producing genotoxicity responses in internal organ tissues, due consideration should also be given to the fashion in which routes of exposure may alter metal toxicokinetics. For metals that demonstrate high affinity binding to carrier proteins, non-physiological routes of administration may bypass normal control mechanisms to yield patterns of tissue specificity and effect that may not be relevant to oral or inhalation exposure. There may further be concern that injection will yield systemic free metal ion concentrations that could not be achieved without causing severe local or systemic toxicity. For metals with strong toxicokinetic dependence upon protein carrier systems, observation of genotoxicity at physiological exposure concentrations, obtained via physiologically relevant exposure routes, would be of greatest concern. In situations where studies employing physiological routes of exposure are negative and those employing non-physiological routes are positive, expert judgement and weight of evidence evaluations will be required for appropriate interpretation of results. For metals with poor uptake rates, short-term oral or inhalation treatment regimens might not be adequate to produce systemic metal ion concentrations required for a relevant test result. Longer treatment regimens could be indicated in such cases. In the absence of adequate information regarding metal toxicokinetics and target organ specificity, indicator assays such as the Comet assay could be applied to identify tissues that may be targets for mutagenesis. However, some caution should be exercised in that organ-specificity for Comet assay results is not always fully concordant with tissue-specificity for carcinogenic responses induced by mutagenic carcinogens (Sasaki et al., 2000). Noticeably absent from the proposed in vivo testing strategy is reliance upon the dominant lethal test. Although long-standing validated OECD testing protocols exist for this test, refinements have since been proposed that more carefully adopt treatment protocols to the timing of different stages of male germ cell development and the action to mutagenic substances (Adler, 2000). Moreover, while a number of metals have been evaluated in the dominant lethal test, much of the data is in the older literature and of questionable quality. As animal welfare issues assume greater importance, risk of genotoxicity to germ cells is more likely to be determined by careful weight of evidence evaluations that determine 1) whether a metal is genotoxic in somatic cells in vivo; and 2) whether the substance reaches the germ cells in significant quantity. This recommendation is made with recognition that there is at present uncertainty with respect to the criteria that are to be applied for definition of whether a substance reaches germ cells and that use of transgenic animals may soon make it possible to assess mutagenic effects in germ cells. Conduct of dominant lethal tests, with appropriately modified and updated protocols (Adler, 2000), remains an option that can be pursued should there emerge to be a legitimate need to quantify risk. The results of the in vivo testing strategy described here would be used for final classification decisions using a weight of evidence approach that carefully evaluates the relevance of the endpoints examined, routes of administration employed and the target tissues studied. This weight of evidence evaluation, particularly if guided by studies employing physiological routes of exposure, would further point the way for the conduct of dose response and mechanistic studies that would be critical for risk assessment. Such strategies are still in early stages of development but might consist of studies that quantify the dose response for both genotoxic effects of concern and the mechanisms that are believed to be responsible for those effects in the target tissues of greatest concern. For example, dose response studies could be conducted to determine dose responses for: 1. Generation of DNA adducts and DNA strand breakage 2. Mouse erythrocyte MN induction. 3. Modulation of ROS induction or metabolism
16 HERAG FACT SHEET Page 16 of 27 Determination of the shape of the dose response curves for each of the above could provide information regarding effects at low exposure levels and subsequent estimates of risk. Given that indirect mechanisms are suspected to mediate potential genotoxicity for many metals, nonlinear or quasi threshold dose response curves may be observed. This will be particularly true if, as is suspected for numerous metals, genotoxicity results from the substitution of exogenous metals for essential metals that play a role in DNA replication and repair or the metabolism of oxygen radicals. Such substitution mechanisms will exhibit kinetics reflective of the relative competitive binding affinity of different metals for target molecules and would be expected to be exhibit distinctly nonlinear dose responses. Concordance between the dose response for biological effects and mechanisms believed to mediate effects would increase the relative degree of confidence placed upon levels of risk assumed at different levels of exposure. However, there is no standard approach or general agreement on data needs and evaluation strategy established at the moment.
17 HERAG FACT SHEET Page 17 of References and abbreviations References Adler, I.D. (2000). Spermatogenesis and mutagenicity of environmental hazards: extrapolation of genetic risk from mouse to man. Andrologia 32: ATSDR (2000). Tocicological Profile for Chromium. U.S. Dept. Health and Human Services. Ariza, M.E. and Williams, M.V. (1996). Mutagenesis of AS52 Cells by Low Concentrations of Lead(II) and Mercury(II). Environ Molecular Mutagen 27: Ariza, M.E., Bijur, G.N. and Williams, M.V. (1998). Lead and Mercury Mutagenesis: Role of H 2 O 2, Superoxide Dismutase, and Xanthine Oxidase. Environ Molecul Mutagen 31: Bunger, J., Stork, J. and Stalder, K. (1996). Cyto- and genotoxic effects of coordination complexes of platinum, palladium and rhodium in vitro. Int Arch Occup Environ Health. 69: Burlinson, B., Tice, R.R., Speit, G., Agurell, E., Brendler-Schwab, S.Y., Collins, A.R., Excobar, P., Kumaravel, T.S., Nkijima, M., Sasaki, Y.F., Thybaud, V., Uno, Y., Vasquez, M. and Hartmann, A. (2007). Fourth International Workgroup on Genotoxicity Testing: Results of the in vivo Comet assay workgroup. Mutat. Res. 627: Caspary, W.J., Daston, D.S., Myhr, B.C., Mitchell, A.D., Rudd, C. and Lee, P.S. (1988). Evaluation of the L5178Y mouse lymphoma cell mutagenesis assay: Interlaboratory reproducibility and assessment. Environ. Mol. Mutagen. 12 (suppl. 13): Choucroun, P., Gillet, D., Dorange, G., Sawicki, B. and Dewitte, J.D. (2001). Comet Assay and Early Apoptosis. Mutat Res 478: Collins, A.R. (2004). The Comet assay for DNA damage and repair. Molec. Biotech. 26: Fairbairn, D.W., Walburger, D.K., Fairbairn, J.J. and O Neill, KL. (1996) Key Morphologic Changes and DNA Strand Breaks in Human Lymphoid Cells: Discriminating Apoptosis from Necrosis. Scanning 18: Faust, F., Kassie, F., Knasmüller, S., Kevekordes, S. and Mersch-Sundermann, V. (2004) Use of Primary Blood Cells for the Assessment of Exposure to Occupational Genotoxicants in Human Biomonitoring Studies. Toxicol 198: Fracasso, M.E., Perbellini, L., Solda, S., Talamini, G. and Franceschetti, P. (2002). Lead Induced DNA Strand Breaks in Lymphocytes of Exposed Workers: Role of Reactive Oxygen Species and Protein Kinase C. Mutat Res 515: Galloway, S.M., Miller, J.E., Armstrong, M.J., Bean, C.L., Skopek, T.R. and Nichols, W.W. (1998). DNA Synthesis Inhibition as an Indirect Mechanism of Chromosome Aberrations: Comparison of DNA-Reactive and non-dna-reactive Clastogens. Mutat Res 400: Hagmar, L., Strömberg, U., Tinnerberg, H. and Mikoczy, Z. (2004a). Epidemiological Evaluation of Cytogenetic Biomarkers as Potential Surrogate End-Points for Cancer. IARC Sci Publ 157: Hagmar, L., Strömberg, U., Bonassi, S., Hansteen, I.-L., Knudsen, L.E., Lindholm, C. and Narpa, H. (2004b). Impact of Types of Lymphocyte Chromosome Aberrations on Human Cancer Risk: Results from Nordic and Italian Cohorts. Cancer Res 64: Halliwell, B. and Gutteridge, J.M.C. (1989). Free Radicals in Biology and Medicine, 2nd Ed. Oxford, Clarendon, Press.
18 HERAG FACT SHEET Page 18 of 27 Hartwig, A. (1994). Role of DNA Repair Inhibition in Lead- and Cadmium-Induced Genotoxicity: A Review. Environ Health Perspect 102: Hartwig A. (2001). Zinc finger proteins as potential targets for toxic metal ions: differential effects on structure and function. Antioxid Redox Signal. 20: Hartwig A., Asmuss M., Ehleben I., Herzer U., Kostelac D., Pelzer A., Schwerdtle T. and Burkle A. (2002). Interference by toxic metal ions with DNA repair processes and cell cycle control: molecular mechanisms. Environ Health Perspect. 110 Suppl 5: Hartwig A., Blessing H., Schwerdtle T., and Walter, I. (2003). Modulation of DNA repair processes by arsenic and selenium compounds. Toxicology 193: Hartwig A. and Schwerdtle T. (2002) Interactions by carcinogenic metal compounds with DNA repair processes: toxicological implications. Toxicol Lett. 127: Henderson, L., Wolfreys, A., Fedyk, J., Bourner, C. and Windebank, S. (1998). The Ability of the Comet Assay to Discriminate between Genotoxins and Cytotoxins. Mutagenesis 13: Huang, X., Feng, Z., Zhai, W. and Xu, J. (1988). Chromosomal Aberrations and Sister Chromatid Exchanges in Workers Exposed to Lead. Biomed Environ Sci 1: Joenje, H. (1989). Genetic Toxicology of Oxygen. Mutat Res 219: Kargacin, B., Klein, C.B. and Costa, M. (1993). Mutagenic responses of nickel oxides and nickel sulphides in Chinese hamster V79 cell lines at the xanthine-guanine phosphoribosyl transferase locus. Mutat. Res. 300: Kirkland, D. (1998). Chromosome Aberration Testing in Genetic Toxicology-Past, Present, and Future. Mutat Res 404: Kirkland, D., Aardema, Henderson, L., and Muller, L. (2005). Evaluation of the Ability of a Battery of Three In Vitro Genotoxicity Tests to Discriminate Rodent Carcinogens and non-carcinogens. I. Sensitivity, Specificity and Relative Predictivity. Mutat. Res. 584: Kirkland, D., Hayashi, M., MacGregor, J.T., Müller, L., Schechtman, L.M. and Sofuni, T. (2003). Summary of Major Conclusions. Mutat Res 540: Kirsch-Volders, M., Vanhauwaert, A., Eichenlaub, U. and Decordier, I. (2003a). Indirect Mechanisms of Genotoxicity. Toxicol Lett : Kirsch-Volders, M., Sfuni, T., Aardema, M., Albertini, S., Eastmond, D., Fenech, M., Ishidate, M. Jr, Kirchner, S., Lorge, E., Morita, T., Norppa, H., Surralles, J., Vanhauwaert, A. and Wakata, A. (2003b). Report from the In Vitro Micronucleus Assay Working Group. Mutat Res 540: Klein, C.B. (1994). Epigenetic control of GPT expression in G12 cells. Environ. Mol. Mutagen. 23:31. Komissarova, E.V., Saha, S.K. and Rossman, T.G. (2004). Dead or Dying: The Importance of Time in Cytotoxicity Assays using Arsenite as an Example. Toxicol Appl Pharmacol (in press). Lantzsch, H. and Gebel T. (1997). Genotoxicity of selected metal compounds in the SOS chromotest. Mutat Res. 389: Lee, M., Kwon, J. and Chung M-K (2003). Enhanced Prediction of Potential Rodent Carcinogenicity by Utilizing Comet Assay and Apoptotic Assay in Combination. Mutat Res 541:9-19. Marnett, L.J. and Burcham, P.C. (1993). Endogenous DNA Adducts: Potential and Paradox. Chem Res Toxicol 6:
19 HERAG FACT SHEET Page 19 of 27 Marnett, L.J. (1994). DNA Adducts of Poly-unsaturated Aldehydes and Dicarbonyl Compounds. In DNA Adducts: Identification and Biological Significance: IARC Scientific Publication No. 125, eds. Hemminki, K., Dipple, A., Shuker, D. E. G., Kadlubar, F. F., Segerbäck, D. & Bartsch, H. (International Agency for Research on Cancer, Lyon), IARC Sci Pub 125, pp Matsuoka, A, Lundin, C, Johansson, F, Sahlin, M, Fukuhara, K, Sjöberg B-M, Jenssen D, and Önfelt A. (2004). Correlation of Sister Chromatid Exchange Formation through Homologous Recombination with Robonucleotide Reductase Inhibition. Mutat Res 547: Nath, R.G. and Chung, F.-L. (1994). Detection of Exocyclic 1,N 2 -Propano-Deoxyguanosine Adducts as Common DNA Lesions in Rodents and Humans. Proc. Natl. Acad. Sci. USA 91: Nestmann, E. R., Matula, T.I., Douglas, G.R. et al (1979). Detection of the mutagenic activity of lead chromate using a battery of microbial tests. Mutat. Res. 66: Roy, N.K. and Rossman, T.G. (1992). Mutagenesis and Comutagenesis by Lead Compounds. Mutat Res 298: Roser S, Pool-Zobel B.L. and Rechkemmer G. (2001). Contribution of Apoptosis to Responses in the Comet Assay. Mutat Res 497: Sasaki YF, Sekihashi K, Izumiyama F, Nishidate E, Saga A, Ishida K, Tsuda S. (2000). The Comet assay with multiple mouse organs: Comparison of Comet assay results and carcinogenicity with 208 chemicals selected from the IARC Monographs and U.S. NTP carcinogenicity database. Crit. Rev. Toxicol. 30: Seeberg, A.H., Mosesso, P. and Forster, R. (1988). High-dose-level effects in mutagenicity assays utilizing mammalian cells in culture. Mutagenesis 3: Shigenaga, M.K., Hagen, T.M. and Ames, B.N. (1994). Oxidative Damage and Mitochondrial Decay in Aging. Proc Natl Acad Sci USA 91: Sonoda E, Sasaki MS, Morrison C, Yamaguchi-Iwai Y, Takata M, and Takada S. (1999). Sister Chromatid Exchanges are Mediated by Homologous Recombination in Vertebrate Cells. Mol Cell Biol 19: Their, R., Bonacker, D., Stoiber, T., Hohm, K.J., Wang, M., Unger, E., Bolt, H.M. and Degen, G. (2003). Interaction of Metal salts with Cytoskeletal Motor Protein Systems. Toxicol Lett : Vaglenov, A., Carbonell, E. and Marcos, R. (1998). Biomonitoring of Workers Exposed to Lead. Genotoxic Effects, Its Modulation by Polyvitamin Treatment and Evaluation of the Induced Radioresistance. Mutat Res 418: Valverde, M., Trejo, C. and Rojas, E. (2001). Is the Capacity of Lead Acetate and Cadmium Chloride to Induce Genotoxic Damage Due to Direct DNA-Metal Interaction? Mutagen 16: Wangenheim, J. and Bolcsfoldi, G. (1988). Mouse lymphoma L5178Y thymidine kinase locus assay of 50 compounds. Mutagenesis 3: Yang, J.L., Wang, L.C., Chang, C.Y. and Liu, T.Y. (1999). Singlet Oxygen is the Major Species Participating in the Induction of DNA Single Strand Breakage and 8-hydroxydeoxyguanosine Adduct by Lead Acetate. Environ Mol Mutagen 33:
20 HERAG FACT SHEET Page 20 of 27 Abbreviations ATSDR Agency for Toxic Substances and Disease Registry HPRT Hypoxanthine PhosphoRibosyl Transferase CA Chromosome Aberrations HR Homologous Recombination CBMN Cytokinesis Block Micronucleus Assay IARC International Agency for Research on Cancer ECB European Chemical Bureau MN Micronucleus / Micronuclei ESR Existing Substances Regulation ROS Reactive Oxygen Species EU European Union SCE Sister Chromatid Exchange FISH Fuorescent In Situ Hybridization TK Thymidine Kinase GHS Global Harmonised System UDS Unscheduled DNA Synthesis GPT Guanine-hypoxanthine PhosphoribosylTransferase
21 HERAG FACT SHEET Page 21 of 27 Annex I: HERAG /Genotoxicity and Carcinogenicity Workshop Held November 2005 in Hannover Germany Workshop Participants: Ilse-Dore Adler (retired, Institute of Experimental Genetics, GSF - National Research Center for Environment and Health, Neuherberg, Germany) Stefano Bonassi (National Cancer Research Institute, Unit of Molecular Epidemiology, Genoa, Italy) Tom Gebel (Federal Institute for Occupational Safety and Health, Dortmund, Germany) Andrea Hartwig (Institute of Food Technology and Food Chemistry, Technical University of Berlin, Germany) Micheline Kirsch-Volders (Laboratory of Cell Genetics, Vrije Universiteit Brussel, Belgium) Tom Sorahan (Institute of Occupational Health, Birmingham University, United Kingdom) Apologies: Awni Sarrif (ECETOC, Belgium) and James Parry (Centre for Molecular Genetics and Toxicology, University of Wales, United Kingdom) Workshop Organizers and Observers: Rodger Battersby (EBRC Consulting GmbH Craig Boreiko (International Lead Zinc Research Organization) James Deyo (Eastman Chemical Company) Kevin Klipsch (EBRC Consulting GmbH) Pat Koundakjian (Eurofer) Adriana Öller (NiPERA) Violaine Verougstraete (Eurometaux) Agenda including charge questions Day 1 9:00 9:15 I. Welcome and Introductions 9:15 10:00 II. Overview a. Goals and objectives of HERAG b. Goals and objectives of the Workshop 10:00 10:30 III. EU Testing Genotoxicity Testing Guidelines a. Present and Future guidelines b. Metal response profiles in vitro 10:30 11:00 Coffee Break 11:00 13:00 IV. Discussion In Vitro Genotoxicity Testing: a. What mechanisms appear to underlie metal responses? b. Are existing/pending guidelines appropriate for metals/ c. To what extent do indirect mechanisms explain metal responses? d. What are the ramifications of indirect mechanisms for testing strategy or interpretation? e. What are the ramifications of indirect mechanisms for assay sensitivity and specificity? f. Are alternate testing strategies appropriate for some groups of metals? If so, can these groups be identified? 13:00 14:00 Lunch 14:00 15:30 V. In Vivo Genotoxicity Testing in Animals a. How should in vitro data be followed-up in animals? b. Do indirect mechanisms modify testing strategy? c. Do metals present testing challenges in terms of target tissues or relevance of route of administration?
22 HERAG FACT SHEET Page 22 of 27 d. What are the appropriate controls for (delayed) cytotoxity and/or apoptosis in assay responses? 15:30 16:30 VI. Presentation and Discussion: Case Study of Metal Genotoxicity Risk Assessment (Kirsh-Volders) 16:30 18:00 VII. Animal Carcinogenicity Response Profiles a. Responses elicited by different metals b. Concordance between carcinogenicity and genotoxicity c. Conditions under which linkages can/cannot be assumed Day 2 9:00 10:00 VIII. Metal Exposures, Epidemiology and Human Cancer a. Metals carcinogenic in humans b. Concordance between animal and human cancer data 10:00 10:45 IX. Discussion: Interpretation of Epidemiology Data a. Importance of exposure assessment/co-exposures b. Which genotoxicity markers would be expected to correspond with carcinogenicity? c. Given probable mechanisms of action, under what conditions can animal and human cancer be inferred? 10:45 11:15 Coffee Break 11:15 13:00 Discussion (continued) d. What lifestyle determinants of cancer are likely to confound metal carcinogenicity/genotoxicity in humans? e. Under what conditions can causal inferences be made between in vitro genotoxicity, in vivo genotoxicity, animal carcinogenicity and human carcinogenicity data? f. Can a decision-tree approach be proposed for testing and data interpretation for studies of metals? 13:00 14:00 Lunch 14:00 16:00 X. Next Steps a. Do we know enough? What data gaps are there that should be filled? b. Research needs? c. Disposition of workshop deliberations XI. Final reflections and adjournment The detailed deliberations of the external experts are described in workshop minutes, which may be obtained upon request from the authors of this fact sheet: ILZRO International Lead Zinc Research Organization Mr. Craig Boreiko 2525 Meridian Parkway P.O. Box Research Triangle Park NC USA cboreiko@ilzro.org or EBRC Consulting GmbH Mr. R. V. Battersby Zeppelinstr Hannover GERMANY rvb@ebrc.de
23 HERAG FACT SHEET Page 23 of 27 Annex II: Current EU Genotoxicity Testing Guidelines Testing guidelines & strategy (in the EU): Preliminary considerations: 1. Annex VII A to Directive 67/548/EEC specifies that minimal data requirements are that genotoxicity data should be available from at least two tests: o bacterial gene mutation test o chromosomal aberration test, which in the absence of contra-indications should be conducted in vitro 2. Before testing, consider the available data from human & animal studies, others and assess their quality/reliability: the quality of the different assays and the weight of evidence may allow a conclusion about hazard based on expert judgement Evaluation of the available data: consider e.g.: - exposure of the target organ - test substance: dose, concentration, volatility, metabolism - quality of the data/studies : reproducibility and biological plausibility of the findings, study design, compliance with GLP - human data: adequacy of exposure information, confounding factors, sources of bias, statistical power, - presence/absence of a dose-response relationship 3. Default assumption: mutagenicity does not have a threshold, unless the opposite is demonstrated by e.g. investigation of the mechanism of action - stochastic process - any level of exposure carries a risk - possible to get information on the relationship between effects (e.g. incidence and severity) and the dose or concentration of the test substance (lowest effective dose) - 2 cases where mutagenicity may have a demonstrable threshold: a) interaction with non- DNA cellular targets (proteins or enzymes) which indirectly affect DNA (incl. aneugens); b) mutagenicity only at high doses, e.g. due to saturation of the metabolic pathway 4. Changes in the new TGD (2003 vs. 1996): - importance of toxicokinetic/toxicodynamic data - importance of aneuploidy - new assay introduced - changes in the testing strategy
24 HERAG FACT SHEET Page 24 of 27 Testing strategy: Objectives: a) to allow appropriate hazard Classification and Labelling and adequate risk characterisation at all levels of supply; b) to meet notification/registration requirements; c) be flexible; d) minimise animal testing & costs; and e)be based on scientific evidence can be applied to existing substances by using the results of previous genotoxicity tests AND human exposure patterns (route, level, duration, involving consumers ) as primary influences, rather than tonnages a comprehensive coverage of the potential mutagenicity requires to have information on gene mutation, structural chromosome aberration (clastogenicity) and numerical chromosomal aberration (aneugenicity) GENOTOXICANTS OTHERS Direct (DNA) Indirect (non DNA) Lesions -Adducts -Breaks -Damage Repair enzymes Mitotic spindle Cell cycle methylation mitogens cytotoxicity apoptosis.. Mutations -Gene -Chromosome -Genome Cell proliferation CARCINOGENESIS (adapted from M. Kirsch-Volders et al.) tests need not be performed if it is not technically possible to do so, or if they are not considered necessary in the light of current scientific knowledge. preferably use test methods of Annex V to Directive 67/548 but other methods may be used when necessary provided that they are scientifically justified regulatory guidelines are still to be established for some of the in vivo tests (consultation on the protocol with an appropriate regulatory authority is recommended)
25 HERAG FACT SHEET Page 25 of 27 Stepwise testing (see decision tree below): 1. Base level: Normally: 2 in vitro tests: gene mutations in bacteria mammalian cell test capable of detecting chromosome aberrations: o in vitro chromosome aberration test (cytogenetic assay for structural chromosome aberrations using metaphase analysis o mouse lymphoma assay (L5178Y cells, TK locus). Not sufficient to detect aneugens. o in vitro MN test NB for suspected aneugens in vitro MN is to be preferred Results: If base level is negative: generally the substance is considered as non-genotoxic but consider extent of human exposure and intended uses of the substance!! Further testing possible: the preferred test will depend on which tests were conducted at base level, e.g. modified test protocol (metabolic activation conditions: alternative to rat liver S9-mix), another test from the base set panel, rat primary hepatocyte UDS test or exceptionally an in vivo test If base level positive: exceptionally no further testing necessary (e.g. only the bacterial test is positive, low tonnage & very low exposure potential. In general, further testing necessary: o Only the bacterial test is positive: conduct another in vitro mammalian cell test. When the latter is negative, decide if further testing (in vitro or in vivo) is needed on a case-by-case basis o Positive results in somatic cells: conduct 1 or 2 in vivo somatic cell test(s) 2. Beyond base level: Before undertaking any in vivo testing, a review of the in vitro test results and all available information on the toxicokinetic and toxicodynamic profile of the test substance is needed. A particular in vivo test should be conducted only when it can be reasonably expected from all the properties of the test substance and the proposed test protocol that the specific target tissue will be adequately exposed to the test substance and/or its metabolites. Recommended in vivo tests are (test should be decided using expert judgement on a case-by-case basis, consider endpoint specificity): A rodent bone marrow or mouse peripheral blood MN test or a rodent bone marrow clastogenicity study. Potential species-specific effects may influence the choice of species and test method used (if in vitro chromosome aberration/mn positive) A rat liver unscheduled DNA synthesis test (if e.g. in vitro gene mutations positive) When it may be more appropriate to evaluate genotoxicity in systemic tissues other than the bone marrow or liver, alternative tests can be selected: in vivo modification of the Comet assay, gene mutation tests with transgenic animals and in vivo DNA adducts studies If the first in vivo test is negative, the need for a further in vivo test should be considered (depending on quality and relevance of the available data, the adequacy of target tissue exposure and the potential for human exposure
26 HERAG FACT SHEET Page 26 of 27 If in vivo test positive: Examine potential for mutagenic effects to be transmitted to the next generation If sufficient information (expert judgement) is available to conclude that the substance may pose a hazard to germ cells, it can be concluded that the substance may cause heritable genetic damage and no further testing is justified: o Assess toxicokinetic/toxicodynamic profile and apply expert judgement o Undertake toxicokinetic study If this information is lacking or inconclusive, germ cell tests are necessary (select the most appropriate on the bas of expert judgement) : o Internationally recognised guidelines are available for investigating clastogenicity in rodent spermatogonial cells and for the dominant lethal test o Alternatively: comet assay, gene mutation tests with transgenic animals, DNA adducts analysis) o Avoid heritable translocation or specific locus test Substances that have given positive results only in cytogenetic tests in vitro and in somatic cells in vivo can be studied further, to differentiate between a clastogenic or aneugenic mode of action. References: Technical Guidance Document, nd edition J. Van Benthem The revised EU Technical Guidance Document for mutagenicity testing, presentation June 2004 M.Kirsch-Volders, M. De Boeck, D. Lison Biomonitoring of gentoxic effects in workers exposed to mutagens/carcinogens
27 HERAG FACT SHEET Page 27 of 27 B a s e s e t + Chromosome aberration test or mouse lymphoma assay or MN test in vitro Gene mutation test bacteria + - Chromosome aberration test or mouse lymphoma assay or MN test in vitro - - Expert judgement: not genotoxic or further testing Select adequate in vivo test Not genotoxic Unscheduled DNA synthesis test (gene mutation test in vivo) - + Chromosome aberration test in vivo or micronucleus test in vivo (Comet assay) - Expert judgement: not genotoxic or further testing considered genotoxic provisional classification and labelling eventual germ cell mutagenicity tests Expert judgement: not genotoxic or further testing
Lecture -2- Environmental Biotechnology
 Lecture -2-1-General Bioassay in pollution Monitoring 1 1 Genotoxicity test At the early testing stages, the genotoxicity assays for predicting potential heritable germ cell damage are the same as used
Lecture -2-1-General Bioassay in pollution Monitoring 1 1 Genotoxicity test At the early testing stages, the genotoxicity assays for predicting potential heritable germ cell damage are the same as used
VICH GL23: Studies to evaluate the safety of residues of veterinary drugs in human food: genotoxicity testing
 6 November 2014 EMA/CVMP/VICH/526/2000 Committee for Medicinal Products for Veterinary Use (CVMP) VICH GL23: Studies to evaluate the safety of residues of veterinary drugs in human food: genotoxicity testing
6 November 2014 EMA/CVMP/VICH/526/2000 Committee for Medicinal Products for Veterinary Use (CVMP) VICH GL23: Studies to evaluate the safety of residues of veterinary drugs in human food: genotoxicity testing
Genotoxicity Testing Strategies: application of the EFSA SC opinion to different legal frameworks in the food and feed area
 Genotoxicity Testing Strategies: application of the EFSA SC opinion to different legal frameworks in the food and feed area Juan Manuel Parra Morte. Pesticides Unit. EFSA. 19th Annual Conference of the
Genotoxicity Testing Strategies: application of the EFSA SC opinion to different legal frameworks in the food and feed area Juan Manuel Parra Morte. Pesticides Unit. EFSA. 19th Annual Conference of the
Genotoxicity of formaldehyde in vitro and in vivo. Günter Speit
 Genotoxicity of formaldehyde in vitro and in vivo Günter Speit Universität Ulm Institut für Humangenetik D-89069 Ulm (Germany) guenter.speit@uni-ulm.de CEFIC-2012 Günter Speit 1 Genotoxicity of formaldehyde
Genotoxicity of formaldehyde in vitro and in vivo Günter Speit Universität Ulm Institut für Humangenetik D-89069 Ulm (Germany) guenter.speit@uni-ulm.de CEFIC-2012 Günter Speit 1 Genotoxicity of formaldehyde
Breaking Up is Hard to Do (At Least in Eukaryotes) Mitosis
 Breaking Up is Hard to Do (At Least in Eukaryotes) Mitosis Chromosomes Chromosomes were first observed by the German embryologist Walther Fleming in 1882. Chromosome number varies among organisms most
Breaking Up is Hard to Do (At Least in Eukaryotes) Mitosis Chromosomes Chromosomes were first observed by the German embryologist Walther Fleming in 1882. Chromosome number varies among organisms most
The effect of REACH implementation on genotoxicity and carcinogenicity testing Jan van Benthem
 The effect of REACH implementation on genotoxicity and carcinogenicity testing Jan van Benthem National Institute for Public Health and the Environment Laboratory for Health Protection Research Bilthoven
The effect of REACH implementation on genotoxicity and carcinogenicity testing Jan van Benthem National Institute for Public Health and the Environment Laboratory for Health Protection Research Bilthoven
6.3 DNA Mutations. SBI4U Ms. Ho-Lau
 6.3 DNA Mutations SBI4U Ms. Ho-Lau DNA Mutations Gene expression can be affected by errors that occur during DNA replication. Some errors are repaired, but others can become mutations (changes in the nucleotide
6.3 DNA Mutations SBI4U Ms. Ho-Lau DNA Mutations Gene expression can be affected by errors that occur during DNA replication. Some errors are repaired, but others can become mutations (changes in the nucleotide
Comments CLH proposal Cadmium hydroxide
 1 Comments CLH proposal Cadmium hydroxide GENERAL COMMENTS: The International Cadmium association (ICdA) welcomes the opportunity to provide its contribution to the public consultation on the proposed
1 Comments CLH proposal Cadmium hydroxide GENERAL COMMENTS: The International Cadmium association (ICdA) welcomes the opportunity to provide its contribution to the public consultation on the proposed
Breaking Up is Hard to Do (At Least in Eukaryotes) Mitosis
 Breaking Up is Hard to Do (At Least in Eukaryotes) Mitosis Prokaryotes Have a Simpler Cell Cycle Cell division in prokaryotes takes place in two stages, which together make up a simple cell cycle 1. Copy
Breaking Up is Hard to Do (At Least in Eukaryotes) Mitosis Prokaryotes Have a Simpler Cell Cycle Cell division in prokaryotes takes place in two stages, which together make up a simple cell cycle 1. Copy
The questions below refer to the following terms. Each term may be used once, more than once, or not at all.
 The questions below refer to the following terms. Each term may be used once, more than once, or not at all. a) telophase b) anaphase c) prometaphase d) metaphase e) prophase 1) DNA begins to coil and
The questions below refer to the following terms. Each term may be used once, more than once, or not at all. a) telophase b) anaphase c) prometaphase d) metaphase e) prophase 1) DNA begins to coil and
5/25/2015. Replication fork. Replication fork. Replication fork. Replication fork
 Mutations Chapter 5 Cellular Functions Lecture 3: and Cell Division Most DNA mutations alter the protein product May Make it function better (rarely) Change its function Reduce its function Make it non-functional
Mutations Chapter 5 Cellular Functions Lecture 3: and Cell Division Most DNA mutations alter the protein product May Make it function better (rarely) Change its function Reduce its function Make it non-functional
cells divide? Growth Development Repair Asexual reproduction Formation of gametes
 mitosis and meiosis cells divide? Growth Development Repair Asexual reproduction Formation of gametes How does a cell know when to divide? the cell cycle A repeating process of cell growth and division
mitosis and meiosis cells divide? Growth Development Repair Asexual reproduction Formation of gametes How does a cell know when to divide? the cell cycle A repeating process of cell growth and division
Ploidy and Human Cell Types. Cell Cycle and Mitosis. DNA and Chromosomes. Where It All Began 11/19/2014. Chapter 12 Pg
 Ploidy and Human Cell Types Cell Cycle and Mitosis Chapter 12 Pg. 228 245 Cell Types Somatic cells (body cells) have 46 chromosomes, which is the diploid chromosome number. A diploid cell is a cell with
Ploidy and Human Cell Types Cell Cycle and Mitosis Chapter 12 Pg. 228 245 Cell Types Somatic cells (body cells) have 46 chromosomes, which is the diploid chromosome number. A diploid cell is a cell with
Chemical and Biochemical Mechanism Of Cell Injury.
 Chemical and Biochemical Mechanism Of Cell Injury. Professor Dr. M. Tariq Javed Dept. of Pathology Faculty of Vet. Science The University Of Agriculture Faisalabad Cell Injury When the cell is exposed
Chemical and Biochemical Mechanism Of Cell Injury. Professor Dr. M. Tariq Javed Dept. of Pathology Faculty of Vet. Science The University Of Agriculture Faisalabad Cell Injury When the cell is exposed
A. Incorrect! All the cells have the same set of genes. (D)Because different types of cells have different types of transcriptional factors.
 Genetics - Problem Drill 21: Cytogenetics and Chromosomal Mutation No. 1 of 10 1. Why do some cells express one set of genes while other cells express a different set of genes during development? (A) Because
Genetics - Problem Drill 21: Cytogenetics and Chromosomal Mutation No. 1 of 10 1. Why do some cells express one set of genes while other cells express a different set of genes during development? (A) Because
Mitosis & Meiosis. Diploid cells- (2n)- a cell that has 2 of each chromosome - 1 from mom, 1 from dad = 1 pair
 Mitosis & Meiosis Diploid cells- (2n)- a cell that has 2 of each chromosome - 1 from mom, 1 from dad = 1 pair The pair is called homologous chromosomes The homologous chromosomes contain the same gene
Mitosis & Meiosis Diploid cells- (2n)- a cell that has 2 of each chromosome - 1 from mom, 1 from dad = 1 pair The pair is called homologous chromosomes The homologous chromosomes contain the same gene
Genetic Toxicology. Toxicology for Industrial and Regulatory Scientists
 Toxicology for Industrial and Regulatory Scientists Genetic Toxicology Mark W. Powley, Ph.D. Pharmacology/Toxicology Reviewer Division of Antiviral Products FDA/CDER/OND April 29, 2015 1 Outline Background
Toxicology for Industrial and Regulatory Scientists Genetic Toxicology Mark W. Powley, Ph.D. Pharmacology/Toxicology Reviewer Division of Antiviral Products FDA/CDER/OND April 29, 2015 1 Outline Background
Cell Division. Chromosome structure. Made of chromatin (mix of DNA and protein) Only visible during cell division
 Chromosome structure Made of chromatin (mix of DNA and protein) Only visible during cell division Chromosome structure The DNA in a cell is packed into an elaborate, multilevel system of coiling and folding.
Chromosome structure Made of chromatin (mix of DNA and protein) Only visible during cell division Chromosome structure The DNA in a cell is packed into an elaborate, multilevel system of coiling and folding.
Mitosis and the Cell Cycle
 Mitosis and the Cell Cycle Chapter 12 The Cell Cycle: Cell Growth & Cell Division Where it all began You started as a cell smaller than a period at the end of a sentence Getting from there to here Cell
Mitosis and the Cell Cycle Chapter 12 The Cell Cycle: Cell Growth & Cell Division Where it all began You started as a cell smaller than a period at the end of a sentence Getting from there to here Cell
DOSE SELECTION FOR CARCINOGENICITY STUDIES OF PHARMACEUTICALS *)
 DOSE SELECTION FOR CARCINOGENICITY STUDIES OF PHARMACEUTICALS *) Guideline Title Dose Selection for Carcinogenicity Studies of Pharmaceuticals *) Legislative basis Directive 75/318/EEC as amended Date
DOSE SELECTION FOR CARCINOGENICITY STUDIES OF PHARMACEUTICALS *) Guideline Title Dose Selection for Carcinogenicity Studies of Pharmaceuticals *) Legislative basis Directive 75/318/EEC as amended Date
nuclear science and technology
 EUROPEAN COMMISSION nuclear science and technology Radiation-specific DNA non-double strand break lesions: repair mechanisms and biological effects (Non-DSB Lesions) Contract N o FIGH-CT2002-00207 Final
EUROPEAN COMMISSION nuclear science and technology Radiation-specific DNA non-double strand break lesions: repair mechanisms and biological effects (Non-DSB Lesions) Contract N o FIGH-CT2002-00207 Final
Chapter 2. Mitosis and Meiosis
 Chapter 2. Mitosis and Meiosis Chromosome Theory of Heredity What structures within cells correspond to genes? The development of genetics took a major step forward by accepting the notion that the genes
Chapter 2. Mitosis and Meiosis Chromosome Theory of Heredity What structures within cells correspond to genes? The development of genetics took a major step forward by accepting the notion that the genes
Genotoxic and acute toxic properties of selected synthetic cannabinoids
 Genotoxic and acute toxic properties of selected synthetic cannabinoids Franziska Ferk 1, Verena Koller 1, Halh Al-Serori 1, Volker Auwärter 2, Siegfried Knasmüller 1 1 Medical University of Vienna, Department
Genotoxic and acute toxic properties of selected synthetic cannabinoids Franziska Ferk 1, Verena Koller 1, Halh Al-Serori 1, Volker Auwärter 2, Siegfried Knasmüller 1 1 Medical University of Vienna, Department
Cellular Reproduction, Part 2: Meiosis Lecture 10 Fall 2008
 Mitosis & 1 Cellular Reproduction, Part 2: Lecture 10 Fall 2008 Mitosis Form of cell division that leads to identical daughter cells with the full complement of DNA Occurs in somatic cells Cells of body
Mitosis & 1 Cellular Reproduction, Part 2: Lecture 10 Fall 2008 Mitosis Form of cell division that leads to identical daughter cells with the full complement of DNA Occurs in somatic cells Cells of body
Example: Distance in M.U. % Crossing Over Why? Double crossovers
 Example: Distance in M.U. % Crossing Over 1 5 10 15 50 80 100 107 Why? Double crossovers 232 .. A B. a b. 1. A fully heterozygous gray-bodied (b+), normal winged (vg+) female F 1 fruit fly crossed with
Example: Distance in M.U. % Crossing Over 1 5 10 15 50 80 100 107 Why? Double crossovers 232 .. A B. a b. 1. A fully heterozygous gray-bodied (b+), normal winged (vg+) female F 1 fruit fly crossed with
Part II The Cell Cell Division, Chapter 2 Outline of class notes
 Part II The Cell Cell Division, Chapter 2 Outline of class notes 1 Cellular Division Overview Types of Cell Division Chromosomal Number The Cell Cycle Mitoses Cancer Cells In Vitro Fertilization Infertility
Part II The Cell Cell Division, Chapter 2 Outline of class notes 1 Cellular Division Overview Types of Cell Division Chromosomal Number The Cell Cycle Mitoses Cancer Cells In Vitro Fertilization Infertility
nuclear science and technology
 EUROPEAN COMMISSION nuclear science and technology The role of intercellular communication and DNA double-strand breaks in the induction of bystander effects (INTERSTANDER) Contract N o FIGH-CT2002-00218
EUROPEAN COMMISSION nuclear science and technology The role of intercellular communication and DNA double-strand breaks in the induction of bystander effects (INTERSTANDER) Contract N o FIGH-CT2002-00218
Chapter 6. Cell Reproduction
 Chapter 6 Cell Reproduction Chromosomes The growth and development of your body are controlled by instructions contained in your DNA DNA decides what you will be.. DNA & Chromosomes Very long molecules
Chapter 6 Cell Reproduction Chromosomes The growth and development of your body are controlled by instructions contained in your DNA DNA decides what you will be.. DNA & Chromosomes Very long molecules
Omnis cellula e cellula
 Chapter 12 The Cell Cycle Omnis cellula e cellula 1855- Rudolf Virchow German scientist all cells arise from a previous cell Every cell from a cell In order for this to be true, cells must have the ability
Chapter 12 The Cell Cycle Omnis cellula e cellula 1855- Rudolf Virchow German scientist all cells arise from a previous cell Every cell from a cell In order for this to be true, cells must have the ability
Reproduction is a fundamental property of life. Cells are the fundamental unit of life. Reproduction occurs at the cellular level with one mother
 Cell Division ision Reproduction is a fundamental property of life. Cells are the fundamental unit of life. Reproduction occurs at the cellular level with one mother cell giving rise to two daughter cells.
Cell Division ision Reproduction is a fundamental property of life. Cells are the fundamental unit of life. Reproduction occurs at the cellular level with one mother cell giving rise to two daughter cells.
The Comet and TUNEL Assays
 Sperm DNA: organization, protection and vulnerability from basic science to clinical application Methods to assess the status of the sperm DNA: The Comet and TUNEL Assays Lars Björndahl Centre for Andrology
Sperm DNA: organization, protection and vulnerability from basic science to clinical application Methods to assess the status of the sperm DNA: The Comet and TUNEL Assays Lars Björndahl Centre for Andrology
Cell cycle and apoptosis
 Cell cycle and apoptosis Cell cycle Definition Stages and steps Cell cycle Interphase (G1/G0, S, and G2) Mitosis (prophase, metaphase, anaphase, telophase, karyokinesis, cytokinesis) Control checkpoints
Cell cycle and apoptosis Cell cycle Definition Stages and steps Cell cycle Interphase (G1/G0, S, and G2) Mitosis (prophase, metaphase, anaphase, telophase, karyokinesis, cytokinesis) Control checkpoints
Cell Cycle Notes --PreAP
 Cell Cycle Notes --PreAP I. DNA Deoxyribonucleic acid; located in nucleus A. Long and thread-like DNA in a non-dividing cell B. Thick, short, coiled doubled DNA in a dividing cell chromosome 1. chromosome
Cell Cycle Notes --PreAP I. DNA Deoxyribonucleic acid; located in nucleus A. Long and thread-like DNA in a non-dividing cell B. Thick, short, coiled doubled DNA in a dividing cell chromosome 1. chromosome
Cell Division and Inheritance
 Cell Division and Inheritance Continuing life relies on reproduction Individual organism replacing dead or damaged cells Species making more of same species Reproduction Cells divide, grow, divide again
Cell Division and Inheritance Continuing life relies on reproduction Individual organism replacing dead or damaged cells Species making more of same species Reproduction Cells divide, grow, divide again
Chapter 8: Cellular Reproduction
 Chapter 8: Cellular Reproduction 1. The Cell Cycle 2. Mitosis 3. Meiosis 2 Types of Cell Division 2n 1n Mitosis: occurs in somatic cells (almost all cells of the body) generates cells identical to original
Chapter 8: Cellular Reproduction 1. The Cell Cycle 2. Mitosis 3. Meiosis 2 Types of Cell Division 2n 1n Mitosis: occurs in somatic cells (almost all cells of the body) generates cells identical to original
The Cell Cycle CHAPTER 12
 The Cell Cycle CHAPTER 12 The Key Roles of Cell Division cell division = reproduction of cells All cells come from pre-exisiting cells Omnis cellula e cellula Unicellular organisms division of 1 cell reproduces
The Cell Cycle CHAPTER 12 The Key Roles of Cell Division cell division = reproduction of cells All cells come from pre-exisiting cells Omnis cellula e cellula Unicellular organisms division of 1 cell reproduces
Mugimane Manjanatha, Ph.D
 Genotoxicity of Doxorubicin in F344 Rats by Combining the Comet Assay, Peripheral Blood Micronucleus Test, and Pathway-Focused Gene Expression Profiling Mugimane Manjanatha, Ph.D FDA/NCTR/DGMT DISCLAIMER
Genotoxicity of Doxorubicin in F344 Rats by Combining the Comet Assay, Peripheral Blood Micronucleus Test, and Pathway-Focused Gene Expression Profiling Mugimane Manjanatha, Ph.D FDA/NCTR/DGMT DISCLAIMER
LESSON 3.2 WORKBOOK. How do normal cells become cancer cells? Workbook Lesson 3.2
 For a complete list of defined terms, see the Glossary. Transformation the process by which a cell acquires characteristics of a tumor cell. LESSON 3.2 WORKBOOK How do normal cells become cancer cells?
For a complete list of defined terms, see the Glossary. Transformation the process by which a cell acquires characteristics of a tumor cell. LESSON 3.2 WORKBOOK How do normal cells become cancer cells?
Mitosis and Cellular Division. EQ: How do the cells in our body divide?
 Mitosis and Cellular Division EQ: How do the cells in our body divide? Cell division is the process by which cellular material is divided between two new daughter cells. 1 Mother Cell 2 Daughter cells.
Mitosis and Cellular Division EQ: How do the cells in our body divide? Cell division is the process by which cellular material is divided between two new daughter cells. 1 Mother Cell 2 Daughter cells.
MIE KE2 AO Mutations per LacZ mutant. [van Delft and Baan 1995] [O'Brien et al. 2014]
![MIE KE2 AO Mutations per LacZ mutant. [van Delft and Baan 1995] [O'Brien et al. 2014] MIE KE2 AO Mutations per LacZ mutant. [van Delft and Baan 1995] [O'Brien et al. 2014]](/thumbs/88/115815301.jpg) Table I. Empirical data for male rodents exposed to germ cells for ENU. Dose (mg/kg) a Alkyl Adducts per nucleotide in male germ cells b MIE KE2 AO Mutations per LacZ mutant nucleotide in Time of frequency
Table I. Empirical data for male rodents exposed to germ cells for ENU. Dose (mg/kg) a Alkyl Adducts per nucleotide in male germ cells b MIE KE2 AO Mutations per LacZ mutant nucleotide in Time of frequency
meiosis asexual reproduction CHAPTER 9 & 10 The Cell Cycle, Meiosis & Sexual Life Cycles Sexual reproduction mitosis
 meiosis asexual reproduction CHAPTER 9 & 10 The Cell Cycle, Meiosis & Sexual Sexual reproduction Life Cycles mitosis Chromosomes Consists of a long DNA molecule (represents thousands of genes) Also consists
meiosis asexual reproduction CHAPTER 9 & 10 The Cell Cycle, Meiosis & Sexual Sexual reproduction Life Cycles mitosis Chromosomes Consists of a long DNA molecule (represents thousands of genes) Also consists
MUTATIONS, MUTAGENESIS, AND CARCINOGENESIS
 MUTATIONS, MUTAGENESIS, AND CARCINOGENESIS How do different alleles arise? ( allele : form of a gene; specific base sequence at a site on DNA) Mutations: heritable changes in genes Mutations occur in DNA
MUTATIONS, MUTAGENESIS, AND CARCINOGENESIS How do different alleles arise? ( allele : form of a gene; specific base sequence at a site on DNA) Mutations: heritable changes in genes Mutations occur in DNA
DISCUSSION GROUP 3. Mechanism of carcinogenicity. EFSA Scientific Colloquium on Acrylamide carcinogenicity, 22/23 May
 DISCUSSION GROUP 3 Mechanism of carcinogenicity EFSA Scientific Colloquium on Acrylamide carcinogenicity, 22/23 May 2008 1 1. Review the recent evidence for the mutagenicity and genotoxicity of acrylamide
DISCUSSION GROUP 3 Mechanism of carcinogenicity EFSA Scientific Colloquium on Acrylamide carcinogenicity, 22/23 May 2008 1 1. Review the recent evidence for the mutagenicity and genotoxicity of acrylamide
Chapter 10 Chromosomes and Cell Reproduction
 Chapter 10 Chromosomes and Cell Reproduction Chromosomes Organisms grow by dividing of cells Binary Fission form of asexual reproduction that produces identical offspring (Bacteria) Eukaryotes have two
Chapter 10 Chromosomes and Cell Reproduction Chromosomes Organisms grow by dividing of cells Binary Fission form of asexual reproduction that produces identical offspring (Bacteria) Eukaryotes have two
Genomic Instability Induced by Ionizing Radiation
 Genomic Instability Induced by Ionizing Radiation Christian Streffer Universitätsklinikum Essen, 45122 Essen, Germany INTRODUCTION In contrast to general assumptions it has frequently been shown that DNA
Genomic Instability Induced by Ionizing Radiation Christian Streffer Universitätsklinikum Essen, 45122 Essen, Germany INTRODUCTION In contrast to general assumptions it has frequently been shown that DNA
The Cell Cycle and How Cells Divide
 The Cell Cycle and How Cells Divide 1 Phases of the Cell Cycle The cell cycle consists of Interphase normal cell activity The mitotic phase cell divsion INTERPHASE Growth G 1 (DNA synthesis) Growth G 2
The Cell Cycle and How Cells Divide 1 Phases of the Cell Cycle The cell cycle consists of Interphase normal cell activity The mitotic phase cell divsion INTERPHASE Growth G 1 (DNA synthesis) Growth G 2
NOTES- CHAPTER 6 CHROMOSOMES AND CELL REPRODUCTION
 NOTES- CHAPTER 6 CHROMOSOMES AND CELL REPRODUCTION Section I Chromosomes Formation of New Cells by Cell Division New cells are formed when old cells divide. 1. Cell division is the same as cell reproduction.
NOTES- CHAPTER 6 CHROMOSOMES AND CELL REPRODUCTION Section I Chromosomes Formation of New Cells by Cell Division New cells are formed when old cells divide. 1. Cell division is the same as cell reproduction.
Creating Identical Body Cells
 Creating Identical Body Cells 5.A Students will describe the stages of the cell cycle, including DNA replication and mitosis, and the importance of the cell cycle to the growth of organisms 5.D Students
Creating Identical Body Cells 5.A Students will describe the stages of the cell cycle, including DNA replication and mitosis, and the importance of the cell cycle to the growth of organisms 5.D Students
5.1. KEY CONCEPT Cells have distinct phases of growth, reproduction, and normal functions. 68 Reinforcement Unit 2 Resource Book
 5.1 THE CELL CYCLE KEY CONCEPT Cells have distinct phases of growth, reproduction, and normal functions. Cells have a regular pattern of growth, DNA duplication, and division that is called the cell cycle.
5.1 THE CELL CYCLE KEY CONCEPT Cells have distinct phases of growth, reproduction, and normal functions. Cells have a regular pattern of growth, DNA duplication, and division that is called the cell cycle.
Cell Growth and Division *
 OpenStax-CNX module: m46034 1 Cell Growth and Division * OpenStax This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License 3.0 By the end of this section, you will
OpenStax-CNX module: m46034 1 Cell Growth and Division * OpenStax This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License 3.0 By the end of this section, you will
ENVIRONMENTAL REGULATIONS AND STANDARD SETTING Health Based Standards: Oncology - Luc Hens
 HEALTH BASED STANDARDS: ONCOLOGY Luc Hens Human Ecology Department, Free University of Brussels (VUB), Belgium. Keywords: carcinogen, carcinogenesis, genotoxin, dose-response relationship, mutagen threshold,
HEALTH BASED STANDARDS: ONCOLOGY Luc Hens Human Ecology Department, Free University of Brussels (VUB), Belgium. Keywords: carcinogen, carcinogenesis, genotoxin, dose-response relationship, mutagen threshold,
Cellular Reproduction, Part 1: Mitosis Lecture 10 Fall 2008
 Cell Theory 1 Cellular Reproduction, Part 1: Mitosis Lecture 10 Fall 2008 Cell theory: All organisms are made of cells All cells arise from preexisting cells How do new cells arise? Cell division the reproduction
Cell Theory 1 Cellular Reproduction, Part 1: Mitosis Lecture 10 Fall 2008 Cell theory: All organisms are made of cells All cells arise from preexisting cells How do new cells arise? Cell division the reproduction
Cell Cycle. Interphase, Mitosis, Cytokinesis, and Cancer
 Cell Cycle Interphase, Mitosis, Cytokinesis, and Cancer Cell Division One cell divides into 2 new identical daughter cells. Chromosomes carry the genetic information (traits) of the cell How many Chromosomes
Cell Cycle Interphase, Mitosis, Cytokinesis, and Cancer Cell Division One cell divides into 2 new identical daughter cells. Chromosomes carry the genetic information (traits) of the cell How many Chromosomes
Unit 2: Reproduction and Development. The Cell Cycle
 PAGE : 1 The Cell Cycle Cell Cycle: A continuous series of cell growth and division for a cell. All cells go through a cell cycle of some sort. The cell cycle consists of two stages. a. Growth Phase Diagram
PAGE : 1 The Cell Cycle Cell Cycle: A continuous series of cell growth and division for a cell. All cells go through a cell cycle of some sort. The cell cycle consists of two stages. a. Growth Phase Diagram
CYTOGENETICS Dr. Mary Ann Perle
 CYTOGENETICS Dr. Mary Ann Perle I) Mitosis and metaphase chromosomes A) Chromosomes are most fully condensed and clearly distinguishable during mitosis. B) Mitosis (M phase) takes 1 to 2 hrs and is divided
CYTOGENETICS Dr. Mary Ann Perle I) Mitosis and metaphase chromosomes A) Chromosomes are most fully condensed and clearly distinguishable during mitosis. B) Mitosis (M phase) takes 1 to 2 hrs and is divided
Chemistry 107 Exam 4 Study Guide
 Chemistry 107 Exam 4 Study Guide Chapter 10 10.1 Recognize that enzyme catalyze reactions by lowering activation energies. Know the definition of a catalyst. Differentiate between absolute, relative and
Chemistry 107 Exam 4 Study Guide Chapter 10 10.1 Recognize that enzyme catalyze reactions by lowering activation energies. Know the definition of a catalyst. Differentiate between absolute, relative and
Unit 4: Cell Division Guided Notes
 Unit 4: Cell Division Guided Notes 1 Chromosomes are structures that contain material When Eukaryotes are not dividing, DNA and Proteins are in a mass called: When the cell divides, it condenses and becomes
Unit 4: Cell Division Guided Notes 1 Chromosomes are structures that contain material When Eukaryotes are not dividing, DNA and Proteins are in a mass called: When the cell divides, it condenses and becomes
The Cell Cycle. Chapter 10
 The Cell Cycle Chapter 10 Why Do Cells Divide? Unicellular 1. Reproduction Multicellular 1. Grow 2. Repair 3. Development/reproduction Types of Division Prokaryotic cells Binary fission = asexual reproduction
The Cell Cycle Chapter 10 Why Do Cells Divide? Unicellular 1. Reproduction Multicellular 1. Grow 2. Repair 3. Development/reproduction Types of Division Prokaryotic cells Binary fission = asexual reproduction
Chapter 9. Cells Grow and Reproduce
 Chapter 9 Cells Grow and Reproduce DNA Replication DNA polymerase Addition of a nucleotide to the 3 end of a growing strand Use dntps as substrate Release of pyrophosphate Initiation of Replication Replication
Chapter 9 Cells Grow and Reproduce DNA Replication DNA polymerase Addition of a nucleotide to the 3 end of a growing strand Use dntps as substrate Release of pyrophosphate Initiation of Replication Replication
Outline Interphase Mitotic Stage Cell Cycle Control Apoptosis Mitosis Mitosis in Animal Cells Cytokinesis Cancer Prokaryotic Cell Division
 The Cell Cycle and Cellular Reproduction Chapter 9 Outline Interphase Mitotic Stage Cell Cycle Control Apoptosis Mitosis Mitosis in Animal Cells Cytokinesis Cancer Prokaryotic Cell Division 1 2 Interphase
The Cell Cycle and Cellular Reproduction Chapter 9 Outline Interphase Mitotic Stage Cell Cycle Control Apoptosis Mitosis Mitosis in Animal Cells Cytokinesis Cancer Prokaryotic Cell Division 1 2 Interphase
Cancer. Questions about cancer. What is cancer? What causes unregulated cell growth? What regulates cell growth? What causes DNA damage?
 Questions about cancer What is cancer? Cancer Gil McVean, Department of Statistics, Oxford What causes unregulated cell growth? What regulates cell growth? What causes DNA damage? What are the steps in
Questions about cancer What is cancer? Cancer Gil McVean, Department of Statistics, Oxford What causes unregulated cell growth? What regulates cell growth? What causes DNA damage? What are the steps in
Biology is the only subject in which multiplication is the same thing as division
 Biology is the only subject in which multiplication is the same thing as division 2007-2008 The Cell Cycle: Cell Growth, Cell Division 2007-2008 Where it all began You started as a cell smaller than a
Biology is the only subject in which multiplication is the same thing as division 2007-2008 The Cell Cycle: Cell Growth, Cell Division 2007-2008 Where it all began You started as a cell smaller than a
Biology is the only subject in which multiplication is the same thing as division
 The Cell Cycle Biology is the only subject in which multiplication is the same thing as division Why do cells divide? For reproduction asexual reproduction For growth one-celled organisms from fertilized
The Cell Cycle Biology is the only subject in which multiplication is the same thing as division Why do cells divide? For reproduction asexual reproduction For growth one-celled organisms from fertilized
Campbell Biology in Focus (Urry) Chapter 9 The Cell Cycle. 9.1 Multiple-Choice Questions
 Campbell Biology in Focus (Urry) Chapter 9 The Cell Cycle 9.1 Multiple-Choice Questions 1) Starting with a fertilized egg (zygote), a series of five cell divisions would produce an early embryo with how
Campbell Biology in Focus (Urry) Chapter 9 The Cell Cycle 9.1 Multiple-Choice Questions 1) Starting with a fertilized egg (zygote), a series of five cell divisions would produce an early embryo with how
Scientific opinion on genotoxicity testing strategies applicable to food and feed safety assessment 1
 DRAFT opinion on genotoxicity testing strategies 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 28 29 30 ENDORSED FOR PUBLIC CONSULTATION DRAFT SCIENTIFIC OPINION Scientific opinion
DRAFT opinion on genotoxicity testing strategies 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 28 29 30 ENDORSED FOR PUBLIC CONSULTATION DRAFT SCIENTIFIC OPINION Scientific opinion
BCHM3972 Human Molecular Cell Biology (Advanced) 2013 Course University of Sydney
 BCHM3972 Human Molecular Cell Biology (Advanced) 2013 Course University of Sydney Page 2: Immune Mechanisms & Molecular Biology of Host Defence (Prof Campbell) Page 45: Infection and Implications for Cell
BCHM3972 Human Molecular Cell Biology (Advanced) 2013 Course University of Sydney Page 2: Immune Mechanisms & Molecular Biology of Host Defence (Prof Campbell) Page 45: Infection and Implications for Cell
Biology is the only subject in which multiplication is the same thing as division. AP Biology
 Biology is the only subject in which multiplication is the same thing as division Chapter 12. The Cell Cycle: Cell Growth, Cell Division Where it all began You started as a cell smaller than a period at
Biology is the only subject in which multiplication is the same thing as division Chapter 12. The Cell Cycle: Cell Growth, Cell Division Where it all began You started as a cell smaller than a period at
Chapter 12. living /non-living? growth repair renew. Reproduction. Reproduction. living /non-living. fertilized egg (zygote) next chapter
 Chapter 12 How cells divide Reproduction living /non-living? growth repair renew based on cell division first mitosis - distributes identical sets of chromosomes cell cycle (life) Cell Division in Bacteria
Chapter 12 How cells divide Reproduction living /non-living? growth repair renew based on cell division first mitosis - distributes identical sets of chromosomes cell cycle (life) Cell Division in Bacteria
Biology is the only subject in which multiplication is the same thing as division
 Biology is the only subject in which multiplication is the same thing as division The Cell Cycle: Cell Growth, Cell Division 2007-2008 2007-2008 Getting from there to here Going from egg to baby. the original
Biology is the only subject in which multiplication is the same thing as division The Cell Cycle: Cell Growth, Cell Division 2007-2008 2007-2008 Getting from there to here Going from egg to baby. the original
Fig. 1. Simple columnar epithelial cells lining the small intestine.
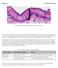 Mitosis Prelab Reading Fig. 1. Simple columnar epithelial cells lining the small intestine. The tall cells pictured in Fig. 1 form the lining of the small intestine in humans and other animals. These cells
Mitosis Prelab Reading Fig. 1. Simple columnar epithelial cells lining the small intestine. The tall cells pictured in Fig. 1 form the lining of the small intestine in humans and other animals. These cells
BIOLOGY. Cell Cycle - Mitosis. Outline. Overview: The Key Roles of Cell Division. identical daughter cells. I. Overview II.
 2 Cell Cycle - Mitosis CAMPBELL BIOLOGY TENTH EDITION Reece Urry Cain Wasserman Minorsky Jackson Outline I. Overview II. Mitotic Phase I. Prophase II. III. Telophase IV. Cytokinesis III. Binary fission
2 Cell Cycle - Mitosis CAMPBELL BIOLOGY TENTH EDITION Reece Urry Cain Wasserman Minorsky Jackson Outline I. Overview II. Mitotic Phase I. Prophase II. III. Telophase IV. Cytokinesis III. Binary fission
Chromosome Abnormalities
 Chromosome Abnormalities Chromosomal abnormalities vs. molecular mutations Simply a matter of size Chromosomal abnormalities are big errors Two types of abnormalities 1. Constitutional problem present
Chromosome Abnormalities Chromosomal abnormalities vs. molecular mutations Simply a matter of size Chromosomal abnormalities are big errors Two types of abnormalities 1. Constitutional problem present
The bases on complementary strands of DNA bond with each other in a specific way A-T and G-C
 1 Bio 1101 Lecture 6 (Guided Notes) Ch. 8: Cellular Basis of Reproduction 2 3 4 5 6 Cellular Basis of Reproduction & Inheritance In order for an organism to replace dead cells or to grow and produce new
1 Bio 1101 Lecture 6 (Guided Notes) Ch. 8: Cellular Basis of Reproduction 2 3 4 5 6 Cellular Basis of Reproduction & Inheritance In order for an organism to replace dead cells or to grow and produce new
B.5ABCD Cell Differentiation
 B.5ABCD Cell Differentiation Picture Vocabulary gene A segment of DNA nucleotides that codes for a specific protein to be produced and leads to the expression of a hereditary trait or characteristic cell
B.5ABCD Cell Differentiation Picture Vocabulary gene A segment of DNA nucleotides that codes for a specific protein to be produced and leads to the expression of a hereditary trait or characteristic cell
Cell Division. The Process of Cell Division Section Section 10.2: The Process of Cell Division 12/8/2010
 The Process of Cell Division Section 10.2 Biology B Section 10.2: The Process of Cell Division The student will investigate and understand common mechanisms of inheritance and protein synthesis. Key concepts
The Process of Cell Division Section 10.2 Biology B Section 10.2: The Process of Cell Division The student will investigate and understand common mechanisms of inheritance and protein synthesis. Key concepts
APGRU4L1 Chap 12 Extra Reading Cell Cycle and Mitosis
 APGRU4L1 Chap 12 Extra Reading Cell Cycle and Mitosis Dr. Ramesh Biology is the only subject in which multiplication is the same thing as division 2007-2008 The Cell Cycle: Cell Growth, Cell Division 2007-2008
APGRU4L1 Chap 12 Extra Reading Cell Cycle and Mitosis Dr. Ramesh Biology is the only subject in which multiplication is the same thing as division 2007-2008 The Cell Cycle: Cell Growth, Cell Division 2007-2008
Scientific opinion on genotoxicity testing strategies applicable to food and feed safety assessment 1
 EFSA Journal 2011;9(9):2379 SCIENTIFIC OPINION Scientific opinion on genotoxicity testing strategies applicable to food and feed safety assessment 1 ABSTRACT EFSA Scientific Committee 2, 3 European Food
EFSA Journal 2011;9(9):2379 SCIENTIFIC OPINION Scientific opinion on genotoxicity testing strategies applicable to food and feed safety assessment 1 ABSTRACT EFSA Scientific Committee 2, 3 European Food
Mutagenesis. David M. DeMarini, Ph.D. University of North Carolina at Chapel Hill.
 Mutagenesis David M. DeMarini, Ph.D. University of North Carolina at Chapel Hill ddemarini@nc.rr.com 1. DNA as the Target Mutagenesis Overview 2. DNA Damage (The Mutagenesis Paradigm) A. Types of Damage
Mutagenesis David M. DeMarini, Ph.D. University of North Carolina at Chapel Hill ddemarini@nc.rr.com 1. DNA as the Target Mutagenesis Overview 2. DNA Damage (The Mutagenesis Paradigm) A. Types of Damage
Biology is the only subject in which multiplication is the same thing as division
 Biology is the only subject in which multiplication is the same thing as division 2007-2008 The Cell Cycle: Cell Growth, Cell Division 2007-2008 Getting from there to here Going from egg to baby. the original
Biology is the only subject in which multiplication is the same thing as division 2007-2008 The Cell Cycle: Cell Growth, Cell Division 2007-2008 Getting from there to here Going from egg to baby. the original
Name Date Class. Interphase. (1) The. grows. DNA is duplicated.
 Concept Mapping The Cell Cycle Complete the cycle map about the cell cycle. These terms may be used more than once: cell, cytoplasm, metaphase, nuclear membrane, nucleoli, poles. (1) The Interphase grows.
Concept Mapping The Cell Cycle Complete the cycle map about the cell cycle. These terms may be used more than once: cell, cytoplasm, metaphase, nuclear membrane, nucleoli, poles. (1) The Interphase grows.
BIOLOGY 4/6/2015. Cell Cycle - Mitosis. Outline. Overview: The Key Roles of Cell Division. identical daughter cells. I. Overview II.
 2 Cell Cycle - Mitosis CAMPBELL BIOLOGY TENTH EDITION Reece Urry Cain Wasserman Minorsky Jackson Outline I. Overview II. Mitotic Phase I. Prophase II. III. Telophase IV. Cytokinesis III. Binary fission
2 Cell Cycle - Mitosis CAMPBELL BIOLOGY TENTH EDITION Reece Urry Cain Wasserman Minorsky Jackson Outline I. Overview II. Mitotic Phase I. Prophase II. III. Telophase IV. Cytokinesis III. Binary fission
Mitosis/Meiosis Simulation Activities
 Mitosis/Meiosis Simulation Activities In this simulation, you will demonstrate an understanding of mitosis, meiosis, segregation, independent assortment, and crossing over, all processes involved with
Mitosis/Meiosis Simulation Activities In this simulation, you will demonstrate an understanding of mitosis, meiosis, segregation, independent assortment, and crossing over, all processes involved with
KEY CONCEPT Cells have distinct phases of growth, reproduction, and normal functions.
 5.1 The Cell Cycle KEY CONCEPT Cells have distinct phases of growth, reproduction, and normal functions. Objective: Cells have distinct phases of growth, reproduction and normal functions. APK: Why do
5.1 The Cell Cycle KEY CONCEPT Cells have distinct phases of growth, reproduction, and normal functions. Objective: Cells have distinct phases of growth, reproduction and normal functions. APK: Why do
Chapter 12 The Cell Cycle
 Chapter 12 The Cell Cycle Objectives Describe how cell reproduction contributes to repair and growth. Compare and contrast prokaryotic and eukaryotic cell division. Compare and contrast asexual and sexual
Chapter 12 The Cell Cycle Objectives Describe how cell reproduction contributes to repair and growth. Compare and contrast prokaryotic and eukaryotic cell division. Compare and contrast asexual and sexual
The bases on complementary strands of DNA bond with each other in a specific way A-T and G-C
 1 Bio 1101 Lecture 6 Ch. 8: Cellular Basis of Reproduction 2 3 4 5 6 Cellular Basis of Reproduction & Inheritance In order for an organism to replace dead cells or to grow and produce new cells, existing
1 Bio 1101 Lecture 6 Ch. 8: Cellular Basis of Reproduction 2 3 4 5 6 Cellular Basis of Reproduction & Inheritance In order for an organism to replace dead cells or to grow and produce new cells, existing
Science 9 Chapter 5 Section 1
 Science 9 Chapter 5 Section 1 The Cell Cycle and Mitosis (pp. 136-151) The Importance of Cell Division Cells, like all living things, eventually wear out and cannot sustain life anymore. They need to be
Science 9 Chapter 5 Section 1 The Cell Cycle and Mitosis (pp. 136-151) The Importance of Cell Division Cells, like all living things, eventually wear out and cannot sustain life anymore. They need to be
Regulators of Cell Cycle Progression
 Regulators of Cell Cycle Progression Studies of Cdk s and cyclins in genetically modified mice reveal a high level of plasticity, allowing different cyclins and Cdk s to compensate for the loss of one
Regulators of Cell Cycle Progression Studies of Cdk s and cyclins in genetically modified mice reveal a high level of plasticity, allowing different cyclins and Cdk s to compensate for the loss of one
Part I: The Cell Cycle
 Cellular Differentiation Part I: The Cell Cycle During your lifetime, trillions of your cells will undergo the cell cycle. This process allows you to grow, heal, and maintain your vital tissues and organs.
Cellular Differentiation Part I: The Cell Cycle During your lifetime, trillions of your cells will undergo the cell cycle. This process allows you to grow, heal, and maintain your vital tissues and organs.
Unit 9: The Cell Cycle
 Unit 9: The Cell Cycle Name: Period: Test Date: 1 Table of Contents Title of Page Page Number Teacher Stamp Unit 9 Warm-Ups 3-4 Cell Cycle/Interphase Notes 5 DNA Replication Video 6 Cancer Notes 15-16
Unit 9: The Cell Cycle Name: Period: Test Date: 1 Table of Contents Title of Page Page Number Teacher Stamp Unit 9 Warm-Ups 3-4 Cell Cycle/Interphase Notes 5 DNA Replication Video 6 Cancer Notes 15-16
Mitosis. AND Cell DiVISION
 Mitosis AND Cell DiVISION Cell Division Characteristic of living things: ability to reproduce their own kind. Cell division purpose: When unicellular organisms such as amoeba divide to form offspring reproduction
Mitosis AND Cell DiVISION Cell Division Characteristic of living things: ability to reproduce their own kind. Cell division purpose: When unicellular organisms such as amoeba divide to form offspring reproduction
Molecular Radiobiology Module 4 Part #3
 Molecular Radiobiology Module 4 Part #3 Bushong - Chapter 31 10-526-197 - Rhodes Interaction & damage is a matter of chance Energy deposited rapidly 10-17 seconds Interactions are non-selective in tissue
Molecular Radiobiology Module 4 Part #3 Bushong - Chapter 31 10-526-197 - Rhodes Interaction & damage is a matter of chance Energy deposited rapidly 10-17 seconds Interactions are non-selective in tissue
Histones- protein molecules that are used to fold and package DNA into chromosomes.
 Chromosome- a portion of the DNA in a cell, a chromosome is created when the DNA segment coils around histones then twists further to create a long twisted mass. Histones- protein molecules that are used
Chromosome- a portion of the DNA in a cell, a chromosome is created when the DNA segment coils around histones then twists further to create a long twisted mass. Histones- protein molecules that are used
Introduction. Biochemistry: It is the chemistry of living things (matters).
 Introduction Biochemistry: It is the chemistry of living things (matters). Biochemistry provides fundamental understanding of the molecular basis for the function and malfunction of living things. Biochemistry
Introduction Biochemistry: It is the chemistry of living things (matters). Biochemistry provides fundamental understanding of the molecular basis for the function and malfunction of living things. Biochemistry
Biology is the only subject in which multiplication is the same thing as division
 Biology is the only subject in which multiplication is the same thing as division 2007-2008 The Cell Cycle: Cell Growth, Cell Division 2007-2008 Where it all began You started as a cell smaller than a
Biology is the only subject in which multiplication is the same thing as division 2007-2008 The Cell Cycle: Cell Growth, Cell Division 2007-2008 Where it all began You started as a cell smaller than a
Mutations. A2 Biology For WJEC
 12. Mutation is a change in the amount, arrangement or structure in the DNA of an organism. 13. There are two types of mutations, chromosome mutations and gene mutations. Mutations A2 Biology For WJEC
12. Mutation is a change in the amount, arrangement or structure in the DNA of an organism. 13. There are two types of mutations, chromosome mutations and gene mutations. Mutations A2 Biology For WJEC
The Cell Cycle. Chapter 12. Biology. Edited by Shawn Lester. Eighth Edition Neil Campbell and Jane Reece. PowerPoint Lecture Presentations for
 Chapter 12 The Cell Cycle Edited by Shawn Lester PowerPoint Lecture Presentations for Biology Eighth Edition Neil Campbell and Jane Reece Lectures by Chris Romero, updated by Erin Barley with contributions
Chapter 12 The Cell Cycle Edited by Shawn Lester PowerPoint Lecture Presentations for Biology Eighth Edition Neil Campbell and Jane Reece Lectures by Chris Romero, updated by Erin Barley with contributions
WHEN DO MUTATIONS OCCUR?
 WHEN DO MUTATIONS OCCUR? While most DNA replicates with fairly high accuracy, mistakes do happen. DNA polymerase sometimes inserts the wrong nucleotide or too many or too few nucleotides into a sequence.
WHEN DO MUTATIONS OCCUR? While most DNA replicates with fairly high accuracy, mistakes do happen. DNA polymerase sometimes inserts the wrong nucleotide or too many or too few nucleotides into a sequence.
MUTATIONS, MUTAGENESIS, AND CARCINOGENESIS. (Start your clickers)
 MUTATIONS, MUTAGENESIS, AND CARCINOGENESIS (Start your clickers) How do mutations arise? And how do they affect a cell and its organism? Mutations: heritable changes in genes Mutations occur in DNA But
MUTATIONS, MUTAGENESIS, AND CARCINOGENESIS (Start your clickers) How do mutations arise? And how do they affect a cell and its organism? Mutations: heritable changes in genes Mutations occur in DNA But
Sexual Reproduction and Genetics. Section 1. Meiosis
 Chromosomes and Chromosome Number! Human body cells have 46 chromosomes! Each parent contributes 23 chromosomes! Homologous chromosomes one of two paired chromosomes, one from each parent Chromosomes and
Chromosomes and Chromosome Number! Human body cells have 46 chromosomes! Each parent contributes 23 chromosomes! Homologous chromosomes one of two paired chromosomes, one from each parent Chromosomes and
Unit 5 Part B Cell Growth, Division and Reproduction
 Unit 5 Part B Cell Growth, Division and Reproduction Cell Size Are whale cells the same size as sea stars cells? Yes! Cell Size Limitations Cells that are too big will have difficulty diffusing materials
Unit 5 Part B Cell Growth, Division and Reproduction Cell Size Are whale cells the same size as sea stars cells? Yes! Cell Size Limitations Cells that are too big will have difficulty diffusing materials
