TGL clinical User Guide
|
|
|
- Bartholomew Bruce
- 6 years ago
- Views:
Transcription
1 TGL clinical User Guide The Institute of Cancer Research Brookes Lawley Building 15 Cotswold Road Sutton, Surrey SM2 5NG Page 1 of 7
2 TGL clinical This document describes the services provided by TGL clinical to internal and external users. The laboratory is situated in the Brookes Lawley Building, on the Sutton campus of the Institute of Cancer Research (ICR). TGL clinical provide a molecular diagnostic service for germline mutation analysis of genes that confer a predisposition to cancer. Uniquely TGL clinical provides an integrated laboratory and clinical interpretation service utilising expertise based at ICR and RMH to test for and interpret germline variation in cancer predisposition genes to aid and inform clinical management decisions. Staff list: Laboratory Director and Consultant Clinical Geneticist: Consultant Oncogeneticists: Clinical and Research Scientists: Scientific Officer: Database Manager: Statistician / Bioinformaticians: Operational Manager: Professor Nazneen Rahman Dr. Angela George Dr. Zoe Kemp Sheila Seal Dr. Shazia Mahamdallie Anthony Renwick Silvana Powell Emma Ramsay Bertha Adjei Anna Elliott Dr. Elise Ruark Dr. Shawn Yost Ann Strydom Page 2 of 7
3 Contact details: Laboratory contact: Postal address: Phone: TGLclinical laboratory Brookes Lawley Building The Institute of Cancer Research 15 Cotswold Road Sutton, Surrey SM2 5NG Website Hours of service: 9.00am to 5.00pm, Monday to Friday (excluding bank holidays). Out of hour s service: No out of hours service is provided. Samples received after 5.00pm will be processed the following working day. Instructions for completion of the request form: Please fill ALL the fields provided with the relevant demographic/clinical information. For patient details, stickers with the patient s information can be used. Similarly, stickers can be used for the Consultant and referring hospital information. If some information is unknown or not applicable please fill the field in with UNKNOWN and N/A, respectively, rather that leaving it blank. Samples with attached request forms that do not contain all the relevant information can be rejected by the lab or put on hold until a new and completely filled request form has been received. Clinical details, required investigations and family history should be filled in where possible. If in doubt please contact us on the laboratory or numbers given above. Consent: Upon receipt of samples, the laboratory assumes that appropriate consent has been obtained by the referring clinician. Page 3 of 7
4 Laboratory repertoire Test Genes included Cost A Cost B Turn-around time Panel tests Effective 1 st April 2017 BRCA panel BRCA1, BRCA % < 3 weeks Breast / Ovarian panel BRCA1, BRCA2, TP53, PALB2, PTEN, MLH1, MSH2, MSH6, STK11, % < 4 weeks BRIP1, RAD51C, RAD51D, ATM, CHEK2 Lynch panel MLH1, MSH2, MSH6, PMS2, EPCAM % < 4 weeks Polyposis panel APC, MUTYH % < 4 weeks Renal panel BAP1, FH, FLCN, MET, PTEN, SDHB, VHL % < 4 weeks Pheo / Para panel SDHB, SDHC, SDHD, SDHAF2, MAX, TMEM127, NF1, RET, VHL % < 4 weeks Melanoma panel CDKN2A, CDK % < 4 weeks Single gene tests Available for any gene included in a panel or one of the following genes: BUB1B, CDC73, CDH1, DICER1, MEN1, NF2, SMARCB1, SUFU, WT % < 3 weeks Specific mutation (predictive) tests Available for any gene listed above in a panel test or single test % < 2 weeks The full coding sequence and intron-exon boundaries are analysed for small variants and exon deletions and duplications, except PMS2 (exons 1-11 only), EPCAM (exon 9 CNV only) POLE, POLD1 (select mutations only). For further details see Cost A: UK NHS, UK self-funding, Middle and Low income countries (World bank classification) Cost B: Privately insured / funded, High income countries (World bank classification) cles/ world-bank-country-and-lending-groups Page 4 of 7
5 Cancer predisposition gene tests not performed by TGL clinical : When requests are received for cancer predisposition gene tests not offered by TGL clinical we will arrange testing by external providers. These providers will be selected based on their accreditation and known expertise for specific tests. When the results are received by TGL clinical, they are forwarded to the referring clinician with a clear indication as to the accreditation status of the external laboratory. Reporting times and accreditation status of the external labs are monitored by TGL clinical. DNA storage: We can store DNA from individuals for which no test is currently indicated, but for which it is considered possible that testing may be appropriate in the future. We do not store blood samples and thus if blood samples are received for storage the DNA is extracted and stored. Turnaround time standards TGL clinical strives to achieve the turnaround times outlined in the Laboratory repertoire table above which are all within the UK national target standards. Reporting of service and standards Where TGL clinical undertakes more than 10 tests per month for any individual institution a monthly report will be provided within the first seven working days of the following month, which will include information on the number of tests, the test type, the referring department and/or clinician, and the average turnaround times per test type Specimens required: Please send 2 x 9 ml of blood in PLASTIC EDTA tubes. Smaller volumes, for example from children, can be extracted if necessary, but we require a minimum of 2 ml. Samples should be sent on the day of collection and at ambient temperature. DNA samples can also be received. We strongly prefer to receive blood samples but if this is not possible, please contact the lab to discuss sending a saliva sample. Unsuitable samples: Clotted samples are occasionally unsuitable for molecular analysis, where possible the referring clinician will be informed and a new sample requested. Samples should be labelled with the patient first name and surname, D.O.B., hospital number and the date and time the sample was taken. The details on the sample tube must correspond to those on the request form. At least two unique identifiers are required. Page 5 of 7
6 Unidentifiable/Unlabelled/Broken samples will NOT be accepted. Where possible the referring clinician will be informed and a new sample requested. High risk blood samples (HIV, Hep B or C) received by TGL clinical must be indicated as such both on the blood tubes and the request form. These samples cannot be extracted by TGL clinical and so will be sent to an external provider for testing. This may in-turn affect the turnaround time for that specific sample. Please note that all DNA samples are stored indefinitely unless otherwise requested. Transportation of samples: At Royal Marsden Hospital, samples will be sent together with the request form from the phlebotomy unit to the Royal Marsden Hospital Central Pathology Department at the Sutton site, or can be delivered to TGL clinical pigeon hole at BLB reception. Samples should be dispatched as soon as possible after taking the sample. In the case of samples sent from the Chelsea site, they will be transported in the pathology containers on the RMH inter-site coach, to be delivered to the RMH Central Pathology. Samples will be then collected from the RMH Central Pathology and the BLB pigeon hole by laboratory staff and transported to the lab contained inside an opaque sample transportation bag. See the HSE guidance on biological agents Blood and DNA samples from external users should be sent by first class mail, please address specimens to the TGL clinical postal address given above. Any diagnostic sample sent to the laboratory should be sent in accordance with the UN3373 regulations and the P650 Packaging instruction ( Failure to abide by these regulations will result in the rejection of that sample by TGL clinical. Data protection: TGL clinical ensures secure and safeguarded storage of patient information and compliance with the Data Protection Act. Charges for tests: TGL clinical will provide notice of any charge in advance of any service being undertaken. Receipt of a test request by TGL clinical will be considered an agreement of that charge. Any changes in charges will be notified 3 months in advance of the change being effected. Quality control: All TGL clinical laboratory examination procedures that generate patient results include appropriate quality control measures. The results of all quality control samples are evaluated for every patient sample by an authorised member of staff prior to the release of the relevant patient results. Page 6 of 7
7 If during an individual examination process a quality control result is deemed to have failed and to invalidate patient results, then these patient results are repeated and are not released without successful accompanying quality control results. Reporting of Results: Reports are produced electronically and can be provided in either electronic or paper format according to the user s requirements. Reports can be printed to produce paper copies when requested but are stored within TGL clinical and routinely dispatched as electronic copies. Clinical interpretations: TGL clinical examination reports contain a clear explanation of the result and its clinical implications for the patient and their relatives. To obtain further clinical advice, contact the lab directly by to tgl@icr.ac.uk. Please note that TGL clinical does not offer any interpretation of results obtained from external providers. Queries and Complaints procedure: Any questions about test methodology, interpretation of the results or complaints should be notified by to tgl@icr.ac.uk, phone or by post to: TGLclinical laboratory Brookes Lawley Building The Institute of Cancer Research 15 Cotswold Road Sutton, Surrey SM2 5NG Queries will be responded to as soon as possible. Complaints will be acknowledged as being received within two working days. Feedback will be provided within seven working days and will include a timeline and process for more detailed consideration of the complaint if necessary. Page 7 of 7
CentoCancer STRIVE FOR THE MOST COMPLETE INFORMATION
 CentoCancer STRIVE FOR THE MOST COMPLETE INFORMATION CentoCancer our most comprehensive oncogenetics panel for hereditary mutations Hereditary pathogenic variants confer an increased risk of developing
CentoCancer STRIVE FOR THE MOST COMPLETE INFORMATION CentoCancer our most comprehensive oncogenetics panel for hereditary mutations Hereditary pathogenic variants confer an increased risk of developing
patient guide CancerNext-Expanded genetic testing for hereditary cancer Because knowing your risk can mean early detection and prevention
 patient guide CancerNext-Expanded genetic testing for hereditary cancer Because knowing your risk can mean early detection and prevention Know the Basics Cancer occurs in about 1 in 3 adults in their lifetime
patient guide CancerNext-Expanded genetic testing for hereditary cancer Because knowing your risk can mean early detection and prevention Know the Basics Cancer occurs in about 1 in 3 adults in their lifetime
Germline Multigene Panel Testing in Oncology: Genetic Counseling Perspective
 Germline Multigene Panel Testing in Oncology: Genetic Counseling Perspective Sarah L. Campian, MS CGC Certified Genetic Counselor Nancy & James Grosfeld Cancer Genetics Center Objectives Identify patients/families
Germline Multigene Panel Testing in Oncology: Genetic Counseling Perspective Sarah L. Campian, MS CGC Certified Genetic Counselor Nancy & James Grosfeld Cancer Genetics Center Objectives Identify patients/families
The benefit of. knowing. Genetic testing for hereditary cancer. A patient support guide
 The benefit of knowing Genetic testing for hereditary cancer A patient support guide Does cancer run in your family? Cancer is more common in some families. Sometimes cancer is caused by a change in a
The benefit of knowing Genetic testing for hereditary cancer A patient support guide Does cancer run in your family? Cancer is more common in some families. Sometimes cancer is caused by a change in a
The Next Generation of Hereditary Cancer Testing
 The Next Generation of Hereditary Cancer Testing Why Genetic Testing? Cancers can appear to run in families. Often this is due to shared environmental or lifestyle patterns, such as tobacco use. However,
The Next Generation of Hereditary Cancer Testing Why Genetic Testing? Cancers can appear to run in families. Often this is due to shared environmental or lifestyle patterns, such as tobacco use. However,
WHAT IS A GENE? CHROMOSOME DNA PROTEIN. A gene is made up of DNA. It carries instructions to make proteins.
 WHAT IS A GENE? CHROMOSOME GENE DNA A gene is made up of DNA. It carries instructions to make proteins. The proteins have specific jobs that help your body work normally. PROTEIN 1 WHAT HAPPENS WHEN THERE
WHAT IS A GENE? CHROMOSOME GENE DNA A gene is made up of DNA. It carries instructions to make proteins. The proteins have specific jobs that help your body work normally. PROTEIN 1 WHAT HAPPENS WHEN THERE
WHAT IS A GENE? CHROMOSOME DNA PROTEIN. A gene is made up of DNA. It carries instructions to make proteins.
 WHAT IS A GENE? CHROMOSOME E GEN DNA A gene is made up of DNA. It carries instructions to make proteins. The proteins have specific jobs that help your body work normally. PROTEIN 1 WHAT HAPPENS WHEN THERE
WHAT IS A GENE? CHROMOSOME E GEN DNA A gene is made up of DNA. It carries instructions to make proteins. The proteins have specific jobs that help your body work normally. PROTEIN 1 WHAT HAPPENS WHEN THERE
Learn your genetic risk for the most common hereditary cancers.
 Learn your genetic risk for the most common hereditary cancers. color.com Color analyzes 30 genes including BRCA1 and BRCA2 to help women and men understand their risk for the most common hereditary cancers,
Learn your genetic risk for the most common hereditary cancers. color.com Color analyzes 30 genes including BRCA1 and BRCA2 to help women and men understand their risk for the most common hereditary cancers,
Genetic Risk Assessment for Cancer
 Genetic Risk Assessment for Cancer Jennifer Siettmann, MS CGC Certified Genetic Counselor Banner MD Anderson Cancer Center Objectives Describe the role of genetic counseling and genetic testing in patient
Genetic Risk Assessment for Cancer Jennifer Siettmann, MS CGC Certified Genetic Counselor Banner MD Anderson Cancer Center Objectives Describe the role of genetic counseling and genetic testing in patient
GYNplus. genetic testing for hereditary ovarian and/or uterine cancer
 GYNplus genetic testing for hereditary ovarian and/or uterine cancer What Are the Causes of Hereditary Ovarian and Uterine Cancer? uterine cancer ovarian cancer sporadic 70-80% hereditary 5% Lynch syndrome
GYNplus genetic testing for hereditary ovarian and/or uterine cancer What Are the Causes of Hereditary Ovarian and Uterine Cancer? uterine cancer ovarian cancer sporadic 70-80% hereditary 5% Lynch syndrome
GYNplus: A Genetic Test for Hereditary Ovarian and/or Uterine Cancer
 GYNplus: A Genetic Test for Hereditary Ovarian and/or Uterine Cancer Causes of Hereditary Ovarian and Uterine Cancer uterine cancer ovarian cancer Sporadic 75-90% Sporadic 70-80% Hereditary, 5% Lynch syndrome
GYNplus: A Genetic Test for Hereditary Ovarian and/or Uterine Cancer Causes of Hereditary Ovarian and Uterine Cancer uterine cancer ovarian cancer Sporadic 75-90% Sporadic 70-80% Hereditary, 5% Lynch syndrome
Are you at risk of Hereditary Cancer? Your Guide to the Answers
 Are you at risk of Hereditary Cancer? Your Guide to the Answers What is Hereditary Cancer? The genes we are born with may contribute to our risk of developing certain types of cancer, including breast,
Are you at risk of Hereditary Cancer? Your Guide to the Answers What is Hereditary Cancer? The genes we are born with may contribute to our risk of developing certain types of cancer, including breast,
Genetic testing for hereditary cancer
 Genetic testing for hereditary cancer THE GENETICS OF HEREDITARY CANCER About half of all men and one-third of all women in the US will develop cancer during their lifetimes. Approximately 5% to 10% of
Genetic testing for hereditary cancer THE GENETICS OF HEREDITARY CANCER About half of all men and one-third of all women in the US will develop cancer during their lifetimes. Approximately 5% to 10% of
Versus. Cancer genomics Competitive Analysis
 Cancer genomics Competitive Analysis Versus AGH Competitor #1 MYRIAD GENETICS, INC. Headquarters: UT, USA & Switzerland Publicly Traded Company - MYGN Founded: 1992 Company Size: 1001-5000 employees (source:
Cancer genomics Competitive Analysis Versus AGH Competitor #1 MYRIAD GENETICS, INC. Headquarters: UT, USA & Switzerland Publicly Traded Company - MYGN Founded: 1992 Company Size: 1001-5000 employees (source:
patient guide CancerNext genetic testing for hereditary cancer Because knowing your risk can mean early detection and prevention
 patient guide CancerNext genetic testing for hereditary cancer Because knowing your risk can mean early detection and prevention Know the Basics Cancer occurs in about 1 in 3 adults in their lifetime types
patient guide CancerNext genetic testing for hereditary cancer Because knowing your risk can mean early detection and prevention Know the Basics Cancer occurs in about 1 in 3 adults in their lifetime types
A guide to genetic testing for hereditary cancers
 Cancer Testing Solutions A guide to genetic testing for hereditary cancers The benefit of knowing TM Hereditary cancer genetic testing can play a critical role in managing health Cancer touches millions
Cancer Testing Solutions A guide to genetic testing for hereditary cancers The benefit of knowing TM Hereditary cancer genetic testing can play a critical role in managing health Cancer touches millions
DNA Genetic Cancer Risk Test. Test Report
 Test Report CONTENTS SECTION 1 1-1. Customer Information 1-2. Test Report Summary 1-3. Test Report Details 1-4. List of Cancers / Tumors Tested SECTION 2 2-1. About the Test 2-2. References 1 CL0040-1_B
Test Report CONTENTS SECTION 1 1-1. Customer Information 1-2. Test Report Summary 1-3. Test Report Details 1-4. List of Cancers / Tumors Tested SECTION 2 2-1. About the Test 2-2. References 1 CL0040-1_B
Genetic Risk Assessment for Cancer
 Genetic Risk Assessment for Cancer Jennifer Siettmann, MS CGC Certified Genetic Counselor/Cancer Risk Counselor Banner Good Samaritan Cancer Screening & Prevention Program Objectives Describe the role
Genetic Risk Assessment for Cancer Jennifer Siettmann, MS CGC Certified Genetic Counselor/Cancer Risk Counselor Banner Good Samaritan Cancer Screening & Prevention Program Objectives Describe the role
Royal Marsden Breast Unit. BRCA testing learning resources pack (MCG LRP1)
 Royal Marsden Breast Unit BRCA testing learning resources pack (MCG LRP1) Please read all the information in this pack. You may also find it useful to return to this information when you are BRCA testing.
Royal Marsden Breast Unit BRCA testing learning resources pack (MCG LRP1) Please read all the information in this pack. You may also find it useful to return to this information when you are BRCA testing.
Be Ready Pack Learn more about how Myriad myrisk is revolutionizing hereditary cancer testing.
 Be Ready Pack Learn more about how Myriad myrisk is revolutionizing hereditary cancer testing. Hereditary cancer and you Approximately 5% to 10% of all cancers develop because a person inherited a genetic
Be Ready Pack Learn more about how Myriad myrisk is revolutionizing hereditary cancer testing. Hereditary cancer and you Approximately 5% to 10% of all cancers develop because a person inherited a genetic
OvaNext. patient guide. genetic testing for hereditary breast, ovarian, and uterine cancer
 patient guide OvaNext genetic testing for hereditary breast, ovarian, and uterine cancer Because knowing your risk can mean early detection and prevention About half of the women diagnosed with uterine
patient guide OvaNext genetic testing for hereditary breast, ovarian, and uterine cancer Because knowing your risk can mean early detection and prevention About half of the women diagnosed with uterine
Assessment and Management of Genetic Predisposition to Breast Cancer. Dr Munaza Ahmed Consultant Clinical Geneticist 2/7/18
 Assessment and Management of Genetic Predisposition to Breast Cancer Dr Munaza Ahmed Consultant Clinical Geneticist 2/7/18 Overview The role of the Cancer Genetics team NICE guidelines for Familial Breast
Assessment and Management of Genetic Predisposition to Breast Cancer Dr Munaza Ahmed Consultant Clinical Geneticist 2/7/18 Overview The role of the Cancer Genetics team NICE guidelines for Familial Breast
Medical Policy An independent licensee of the Blue Cross Blue Shield Association
 Genetic Cancer Susceptibility Panels Using Next Generation Sequencing Page 1 of 35 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Genetic Cancer Susceptibility
Genetic Cancer Susceptibility Panels Using Next Generation Sequencing Page 1 of 35 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Genetic Cancer Susceptibility
Why Test for Hereditary Cancer in Preventive Care?
 Why Test for Hereditary Cancer in Preventive Care? Millions of people are sidelined by cancer. Wouldn't it be worth it for your patients to know their risk? background HEREDITARY (5-10%) More than 1 in
Why Test for Hereditary Cancer in Preventive Care? Millions of people are sidelined by cancer. Wouldn't it be worth it for your patients to know their risk? background HEREDITARY (5-10%) More than 1 in
MOLECULAR SERVICES. mlabs.umich.edu
 MOLECULAR SERVICES mlabs.umich.edu 800.862.7284 PICTURED ON LEFT IS MARWAN TAYEH, PH.D, CLINICAL ASSISTANT PROFESSOR, PEDIATRICS - GENETICS, ON RIGHT IS TODD ACKLEY, LABORATORY MANAGER MLabs is a full-service
MOLECULAR SERVICES mlabs.umich.edu 800.862.7284 PICTURED ON LEFT IS MARWAN TAYEH, PH.D, CLINICAL ASSISTANT PROFESSOR, PEDIATRICS - GENETICS, ON RIGHT IS TODD ACKLEY, LABORATORY MANAGER MLabs is a full-service
Genetic Cancer Susceptibility Panels Using Next Generation Sequencing
 Genetic Cancer Susceptibility Panels Using Next Generation Sequencing Policy Number: 2.04.93 Last Review: 7/2018 Origination: 7/2013 Next Review: 7/2019 Policy Blue Cross and Blue Shield of Kansas City
Genetic Cancer Susceptibility Panels Using Next Generation Sequencing Policy Number: 2.04.93 Last Review: 7/2018 Origination: 7/2013 Next Review: 7/2019 Policy Blue Cross and Blue Shield of Kansas City
A Patient s Guide to Hereditary Cancer. Is Hereditary Cancer Testing Right for You?
 A Patient s Guide to Hereditary Cancer Is Hereditary Cancer Testing Right for You? What is Hereditary Cancer? Most cancers occur in people who do not have a strong family history of that cancer. This is
A Patient s Guide to Hereditary Cancer Is Hereditary Cancer Testing Right for You? What is Hereditary Cancer? Most cancers occur in people who do not have a strong family history of that cancer. This is
Cancer Survivorship Symposium Cancer and Heredity January 16, Jeanne P. Homer, MS Licensed Certified Genetic Counselor
 Cancer Survivorship Symposium Cancer and Heredity January 16, 2017 Jeanne P. Homer, MS Licensed Certified Genetic Counselor Outline Cancer and Heredity Hereditary Cancer Risk Assessment & Genetic testing
Cancer Survivorship Symposium Cancer and Heredity January 16, 2017 Jeanne P. Homer, MS Licensed Certified Genetic Counselor Outline Cancer and Heredity Hereditary Cancer Risk Assessment & Genetic testing
Hereditary Cancer. Genetic Testing Services. The GeneDx Experience
 Hereditary Cancer Genetic Testing Services The GeneDx Experience GeneDx: Where Rare is Common In medical school there is a saying, When you hear hoofbeats, think horses not zebras. Think of common causes
Hereditary Cancer Genetic Testing Services The GeneDx Experience GeneDx: Where Rare is Common In medical school there is a saying, When you hear hoofbeats, think horses not zebras. Think of common causes
Manchester Cytology Centre Synovial Fluid Analysis Service User Manual 2017/2018
 Page 1 of 12 Author: Sarah Ferris Directorate of Laboratory Medicine Manchester Cytology Centre Synovial Fluid Analysis Service User Manual 2017/2018 Page 2 of 12 Author: Sarah Ferris Table of contents
Page 1 of 12 Author: Sarah Ferris Directorate of Laboratory Medicine Manchester Cytology Centre Synovial Fluid Analysis Service User Manual 2017/2018 Page 2 of 12 Author: Sarah Ferris Table of contents
Germline Testing for Hereditary Cancer with Multigene Panel
 Germline Testing for Hereditary Cancer with Multigene Panel Po-Han Lin, MD Department of Medical Genetics National Taiwan University Hospital 2017-04-20 Disclosure No relevant financial relationships with
Germline Testing for Hereditary Cancer with Multigene Panel Po-Han Lin, MD Department of Medical Genetics National Taiwan University Hospital 2017-04-20 Disclosure No relevant financial relationships with
Myriad Financial Assistance Program (MFAP)
 Myriad Financial Assistance Program (MFAP) MEDICAL CRITERIA Hereditary Cancer Products The Myriad Financial Assistance Program offers aid to patients who meet specific financial and medical requirements.
Myriad Financial Assistance Program (MFAP) MEDICAL CRITERIA Hereditary Cancer Products The Myriad Financial Assistance Program offers aid to patients who meet specific financial and medical requirements.
OncoGeneDx Test Requisition Form
 OncoGeneDx Test Requisition Form Patient Information First name Last name Gender Male Female of birth (mm/dd/yy) Ancestry Caucasian Eastern European Northern European Western European Native American Middle
OncoGeneDx Test Requisition Form Patient Information First name Last name Gender Male Female of birth (mm/dd/yy) Ancestry Caucasian Eastern European Northern European Western European Native American Middle
Genetic testing and pancreatic disease
 Genetic testing and pancreatic disease February 2 d, 2018 Yale Pancreas Symposium 2018: Multidisciplinary Management of Pancreatic Cancer Xavier Llor, M.D., PhD. Associate Professor of Medicine Co-Director,
Genetic testing and pancreatic disease February 2 d, 2018 Yale Pancreas Symposium 2018: Multidisciplinary Management of Pancreatic Cancer Xavier Llor, M.D., PhD. Associate Professor of Medicine Co-Director,
MSI positive MSI negative
 Pritchard et al. 2014 Supplementary Figure 1 MSI positive MSI negative Hypermutated Median: 673 Average: 659.2 Non-Hypermutated Median: 37.5 Average: 43.6 Supplementary Figure 1: Somatic Mutation Burden
Pritchard et al. 2014 Supplementary Figure 1 MSI positive MSI negative Hypermutated Median: 673 Average: 659.2 Non-Hypermutated Median: 37.5 Average: 43.6 Supplementary Figure 1: Somatic Mutation Burden
OncoGeneDx Test Requisition Form
 OncoGeneDx Test Requisition Form Patient Information First name Last name Gender Male Female Date of birth (mm/dd/yy) Ancestry Caucasian Eastern European Northern European Western European Native American
OncoGeneDx Test Requisition Form Patient Information First name Last name Gender Male Female Date of birth (mm/dd/yy) Ancestry Caucasian Eastern European Northern European Western European Native American
The Role of genetic Testing for Inherited Prostate Cancer Risk
 FOIU July 2018 The Role of genetic Testing for Inherited Prostate Cancer Risk Leonard G. Gomella, MD Chairman, Department of Urology Sidney Kimmel Cancer Center Thomas Jefferson University Philadelphia,
FOIU July 2018 The Role of genetic Testing for Inherited Prostate Cancer Risk Leonard G. Gomella, MD Chairman, Department of Urology Sidney Kimmel Cancer Center Thomas Jefferson University Philadelphia,
Corporate Medical Policy
 Corporate Medical Policy Genetic Cancer Susceptibility Panels Using Next Generation File Name: Origination: Last CAP Review: Next CAP Review: Last Review: genetic_cancer_susceptibility_panels_using_next_generation_sequencing
Corporate Medical Policy Genetic Cancer Susceptibility Panels Using Next Generation File Name: Origination: Last CAP Review: Next CAP Review: Last Review: genetic_cancer_susceptibility_panels_using_next_generation_sequencing
Expert Interview: Inherited Susceptibility to Cancer with Dr. Nicoleta Voian
 Expert Interview: Inherited Susceptibility to Cancer with Dr. Nicoleta Voian ANNOUNCER OPEN: Welcome to CME on ReachMD. This segment, entitled Inherited Susceptibility to Cancer: What Do Primary Care Providers
Expert Interview: Inherited Susceptibility to Cancer with Dr. Nicoleta Voian ANNOUNCER OPEN: Welcome to CME on ReachMD. This segment, entitled Inherited Susceptibility to Cancer: What Do Primary Care Providers
Use of panel tests in place of single gene tests in the cancer genetics clinic
 Clin Genet 2015: 88: 278 282 Printed in Singapore. All rights reserved CLINICAL GENETICS doi: 10.1111/cge.12488 Short Report se of panel tests in place of single gene tests in the cancer genetics clinic
Clin Genet 2015: 88: 278 282 Printed in Singapore. All rights reserved CLINICAL GENETICS doi: 10.1111/cge.12488 Short Report se of panel tests in place of single gene tests in the cancer genetics clinic
Scottish E. coli O157/Shiga toxin-producing E. coli Reference Laboratory
 Scottish E. coli O157/Shiga toxin-producing E. coli Reference Laboratory 1 Authors: SERL68:Version L. Allison 6 Issue date & M. 27/09/2017 Hanson This is an electronic document Authors: L. Allison which
Scottish E. coli O157/Shiga toxin-producing E. coli Reference Laboratory 1 Authors: SERL68:Version L. Allison 6 Issue date & M. 27/09/2017 Hanson This is an electronic document Authors: L. Allison which
BRCAplus. genetic testing for hereditary breast cancer
 BRCAplus genetic testing for hereditary breast cancer Developed in collaboration with Fox Chase Cancer Center and the Arcadia University Genetic Counseling Program. Causes of Hereditary Breast Cancer familial
BRCAplus genetic testing for hereditary breast cancer Developed in collaboration with Fox Chase Cancer Center and the Arcadia University Genetic Counseling Program. Causes of Hereditary Breast Cancer familial
Genetic Cancer Susceptibility Panels Using Next Generation Sequencing
 Genetic Cancer Susceptibility Panels Using Next Generation Sequencing Policy Number: 2.04.93 Last Review: 7/2017 Origination: 7/2013 Next Review: 7/2018 Policy Blue Cross and Blue Shield of Kansas City
Genetic Cancer Susceptibility Panels Using Next Generation Sequencing Policy Number: 2.04.93 Last Review: 7/2017 Origination: 7/2013 Next Review: 7/2018 Policy Blue Cross and Blue Shield of Kansas City
Understanding Your Positive Result. A guide to understanding your risk and taking action
 Understanding Your Positive Result A guide to understanding your risk and taking action 2017 Myriad Genetics, Inc. 320 Wakara Way, Salt Lake City, Utah 84108 PH: 1-800-469-7423 FX: 801-584-3615 1 PART
Understanding Your Positive Result A guide to understanding your risk and taking action 2017 Myriad Genetics, Inc. 320 Wakara Way, Salt Lake City, Utah 84108 PH: 1-800-469-7423 FX: 801-584-3615 1 PART
patient guide RenalNext genetic testing for hereditary kidney cancer Because knowing your risk can mean early detection and prevention
 patient guide RenalNext genetic testing for hereditary kidney cancer Because knowing your risk can mean early detection and prevention Know the Basics Risk factors for developing kidney cancer include
patient guide RenalNext genetic testing for hereditary kidney cancer Because knowing your risk can mean early detection and prevention Know the Basics Risk factors for developing kidney cancer include
Hereditary Cancer Products
 Hereditary Products Integrated BRACAnalysis (BRCA1 and BRCA2 sequencing and large rearrangement testing (BART)), covered when: PERSONAL HISTORY of Breast (must meet at least 1) Diagnosed
Hereditary Products Integrated BRACAnalysis (BRCA1 and BRCA2 sequencing and large rearrangement testing (BART)), covered when: PERSONAL HISTORY of Breast (must meet at least 1) Diagnosed
Populations Interventions Comparators Outcomes Individuals: With a personal and/or family history suggesting an inherited cancer syndrome
 Genetic Cancer Susceptibility Panels Using Next- Generation (20493) Medical Benefit Effective Date: 01/01/18 Next Review Date: 09/18 Preauthorization No Review Dates: 09/13, 09/14, 09/15, 09/16, 09/17
Genetic Cancer Susceptibility Panels Using Next- Generation (20493) Medical Benefit Effective Date: 01/01/18 Next Review Date: 09/18 Preauthorization No Review Dates: 09/13, 09/14, 09/15, 09/16, 09/17
A pathogenic mutation was identified in the BRCA1 gene.
 Hereditary Cancer Risk Test ORDERING PHYSICIAN Dr. Jenny Jones Sample Medical Group 123 Main St. Sample, CA SPECIMEN Type: Saliva Barcode: 223 234234 2343 Collected: Apr 13, 2016 Received: Apr 14, 2016
Hereditary Cancer Risk Test ORDERING PHYSICIAN Dr. Jenny Jones Sample Medical Group 123 Main St. Sample, CA SPECIMEN Type: Saliva Barcode: 223 234234 2343 Collected: Apr 13, 2016 Received: Apr 14, 2016
Division of Genomic Diagnostics CANCER GENOMIC TEST REQUISITION Page 1 of 6
 Page 1 of 6 SHIP TO: Children s Hospital of Philadelphia, Genomic Diagnostic Laboratory, 3615 Civic Center Blvd., Abramson Research Center, 714, Philadelphia, PA 19104-4302 - Phone: (215) 590-4452 Patient
Page 1 of 6 SHIP TO: Children s Hospital of Philadelphia, Genomic Diagnostic Laboratory, 3615 Civic Center Blvd., Abramson Research Center, 714, Philadelphia, PA 19104-4302 - Phone: (215) 590-4452 Patient
Clinical Cancer Genetics
 Clinical Cancer Genetics Lisen Axell, MS, CGC University of Colorado Cancer Center Individuals with cancer Making surgical decisions (lump vs. mast) Making treatment decisions (XRT) Concerns for additional
Clinical Cancer Genetics Lisen Axell, MS, CGC University of Colorado Cancer Center Individuals with cancer Making surgical decisions (lump vs. mast) Making treatment decisions (XRT) Concerns for additional
Proactive Patient Paves the Way for Genetic Testing of Eight Family Members
 CASE STUDY Proactive Patient Paves the Way for Genetic Testing of Eight Family Members Quick Summary Samar Mohite * was diagnosed with colon adenocarcinoma at the age of 49 years. Genetic counselling was
CASE STUDY Proactive Patient Paves the Way for Genetic Testing of Eight Family Members Quick Summary Samar Mohite * was diagnosed with colon adenocarcinoma at the age of 49 years. Genetic counselling was
Supplementary Information
 Supplementary Information Implementing rapid, robust, cost-effective, patient-centred, routine genetic testing in ovarian cancer patients Angela George 1,2,3, Daniel Riddell 1, Sheila Seal 1,4, Sabrina
Supplementary Information Implementing rapid, robust, cost-effective, patient-centred, routine genetic testing in ovarian cancer patients Angela George 1,2,3, Daniel Riddell 1, Sheila Seal 1,4, Sabrina
PROVIDER POLICIES & PROCEDURES
 PROVIDER POLICIES & PROCEDURES GENETIC CANCER SUSCEPTIBILITY PANELS USING NEXT GENERATION SEQUENCING The purpose of this document is to assist providers enrolled in the Connecticut Medical Assistance Program
PROVIDER POLICIES & PROCEDURES GENETIC CANCER SUSCEPTIBILITY PANELS USING NEXT GENERATION SEQUENCING The purpose of this document is to assist providers enrolled in the Connecticut Medical Assistance Program
Scottish Bacterial Sexually Transmitted Infections Reference Laboratory (SBSTIRL) User Manual Jan 2018
 Scottish Bacterial Sexually Transmitted Infections Reference Laboratory (SBSTIRL) User Manual Jan 2018 Page 1 Contents 1 Introduction Page 3 2 Contact details Page 4 3 Opening hours Page 4 4 Services provided
Scottish Bacterial Sexually Transmitted Infections Reference Laboratory (SBSTIRL) User Manual Jan 2018 Page 1 Contents 1 Introduction Page 3 2 Contact details Page 4 3 Opening hours Page 4 4 Services provided
Manchester Cytology Centre Synovial Fluid Analysis Service User Manual 2016
 Page 1 of 13 Directorate of Laboratory Medicine Manchester Cytology Centre Synovial Fluid Analysis Service User Manual 2016 Page 2 of 13 Table of contents Table of contents... 2 1. About us... 3 1.1 Opening
Page 1 of 13 Directorate of Laboratory Medicine Manchester Cytology Centre Synovial Fluid Analysis Service User Manual 2016 Page 2 of 13 Table of contents Table of contents... 2 1. About us... 3 1.1 Opening
Medical Policy Update
 Medical Policy Update Summer 2017 Highlights of recent medical policy revisions as well as any new medical policies approved by Prevea360 Health Plan s Medical Policy Committee are shown below. The Medical
Medical Policy Update Summer 2017 Highlights of recent medical policy revisions as well as any new medical policies approved by Prevea360 Health Plan s Medical Policy Committee are shown below. The Medical
Mainstreaming Cancer Genetics (MCG) Information Pack
 Mainstreaming Cancer Genetics (MCG) Key Points Information Pack Genetic information is important for people with cancer and their relatives. New gene testing methods can be harnessed so that more genes
Mainstreaming Cancer Genetics (MCG) Key Points Information Pack Genetic information is important for people with cancer and their relatives. New gene testing methods can be harnessed so that more genes
TumorNext-HRD with OvaNext: Paired Germline and Tumor Analyses of Genes Involved in
 SAMPLE REPORT Ordered By Contact ID:1251298 Example, Doctor, MD MOCKORG44 (10829) 123 Somewhere LaneSuite 4 Heaven NV 78872 US Ph:123-123-1234 Fx:123-123-1223 Org ID:8141 Normal Specimen Accession #: 00-086947
SAMPLE REPORT Ordered By Contact ID:1251298 Example, Doctor, MD MOCKORG44 (10829) 123 Somewhere LaneSuite 4 Heaven NV 78872 US Ph:123-123-1234 Fx:123-123-1223 Org ID:8141 Normal Specimen Accession #: 00-086947
Schedule of Accreditation issued by United Kingdom Accreditation Service 2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK
 2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK NW Thames Regional Genetics Laboratory Northwick Park Hospital Watford Road Harrow HA1 3UJ United Kingdom Contact: Caroline Sullivan Tel:
2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK NW Thames Regional Genetics Laboratory Northwick Park Hospital Watford Road Harrow HA1 3UJ United Kingdom Contact: Caroline Sullivan Tel:
patient guide BreastNext genetic testing for hereditary breast cancer Because knowing your risk can mean early detection and prevention
 patient guide BreastNext genetic testing for hereditary breast cancer Because knowing your risk can mean early detection and prevention Know the Basics Breast cancer is the most common cancer in women
patient guide BreastNext genetic testing for hereditary breast cancer Because knowing your risk can mean early detection and prevention Know the Basics Breast cancer is the most common cancer in women
Enabling Personalized
 Molecular Enabling Personalized Diagnostics Medicine- Targeted Sequencing: NGS-based solutions Silvia Dorn Roel Reinders- Andreas Diplas Friday, 19.06.2015 Company Overview Founded in April 2011 Development
Molecular Enabling Personalized Diagnostics Medicine- Targeted Sequencing: NGS-based solutions Silvia Dorn Roel Reinders- Andreas Diplas Friday, 19.06.2015 Company Overview Founded in April 2011 Development
6/8/17. Genetics 101. Professor, College of Medicine. President & Chief Medical Officer. Hereditary Breast and Ovarian Cancer 2017
 Genetics 101 Hereditary Breast and Ovarian Cancer 2017 Rebecca Sutphen, MD, FACMG Professor, College of Medicine President & Chief Medical Officer INVASIVE CANCER GENETICALLY ALTERED CELL HYPERPLASIA DYSPLASIA
Genetics 101 Hereditary Breast and Ovarian Cancer 2017 Rebecca Sutphen, MD, FACMG Professor, College of Medicine President & Chief Medical Officer INVASIVE CANCER GENETICALLY ALTERED CELL HYPERPLASIA DYSPLASIA
Genetic Testing for BRCA1 and BRCA2 Genes
 Genetic Testing for BRCA1 and BRCA2 Genes MP9478 Covered Service: Prior Authorization Required: Additional Information: Yes when meets criteria below Yes as shown below Pre and post-test genetic counseling
Genetic Testing for BRCA1 and BRCA2 Genes MP9478 Covered Service: Prior Authorization Required: Additional Information: Yes when meets criteria below Yes as shown below Pre and post-test genetic counseling
CASE STUDY. Mutation-Specific Testing: Eligibility for PARP Inhibitor Therapy Established. Introduction. Patient Profile.
 CASE STUDY Mutation-Specific Testing: Eligibility for PARP Introduction Genetic testing is an emergent diagnostic technique that can provide specific and actionable inputs to the management of cancer therapy.
CASE STUDY Mutation-Specific Testing: Eligibility for PARP Introduction Genetic testing is an emergent diagnostic technique that can provide specific and actionable inputs to the management of cancer therapy.
CASE STUDY. Germline Cancer Testing in A Proband: Extension of Benefits to Unaffected Family Members. Introduction. Patient Profile.
 CASE STUDY Germline Cancer Testing in A Proband: Extension of Benefits to Unaffected Family Members Introduction Most cancers are caused by genetic damage resulting from random mutations and exposure to
CASE STUDY Germline Cancer Testing in A Proband: Extension of Benefits to Unaffected Family Members Introduction Most cancers are caused by genetic damage resulting from random mutations and exposure to
A CANCER GENETICIST S (2 ND ) BEST FRIEND IS THEIR PATHOLOGIST. Dr Alan Donaldson Consultant in Clinical Genetics St Michaels Hospital Bristol
 A CANCER GENETICIST S (2 ND ) BEST FRIEND IS THEIR PATHOLOGIST Dr Alan Donaldson Consultant in Clinical Genetics St Michaels Hospital Bristol Topics to be covered How pathology contributes to genetic diagnosis
A CANCER GENETICIST S (2 ND ) BEST FRIEND IS THEIR PATHOLOGIST Dr Alan Donaldson Consultant in Clinical Genetics St Michaels Hospital Bristol Topics to be covered How pathology contributes to genetic diagnosis
Genetic Testing for Familial Gastrointestinal Cancer Syndromes. C. Richard Boland, MD La Jolla, CA January 21, 2017
 Genetic Testing for Familial Gastrointestinal Cancer Syndromes C. Richard Boland, MD La Jolla, CA January 21, 2017 Disclosure Information C. Richard Boland, MD I have no financial relationships to disclose.
Genetic Testing for Familial Gastrointestinal Cancer Syndromes C. Richard Boland, MD La Jolla, CA January 21, 2017 Disclosure Information C. Richard Boland, MD I have no financial relationships to disclose.
Parkinson s Specialist Practitioner
 Parkinson s Specialist Practitioner Delivered by The Royal Marsden NHS Foundation Trust 2 The Parkinson s Specialist is experienced in caring for people affected by Parkinson s. The Specialist will work
Parkinson s Specialist Practitioner Delivered by The Royal Marsden NHS Foundation Trust 2 The Parkinson s Specialist is experienced in caring for people affected by Parkinson s. The Specialist will work
Plasma-Seq conducted with blood from male individuals without cancer.
 Supplementary Figures Supplementary Figure 1 Plasma-Seq conducted with blood from male individuals without cancer. Copy number patterns established from plasma samples of male individuals without cancer
Supplementary Figures Supplementary Figure 1 Plasma-Seq conducted with blood from male individuals without cancer. Copy number patterns established from plasma samples of male individuals without cancer
Informed Consent Columbia Whole Genome or Whole Exome Sequencing
 Informed Consent Columbia Whole Genome or Whole Exome Sequencing Please read the following form carefully and discuss with your ordering physician before signing consent. This consent is intended for the
Informed Consent Columbia Whole Genome or Whole Exome Sequencing Please read the following form carefully and discuss with your ordering physician before signing consent. This consent is intended for the
patient guide PancNext genetic testing for hereditary pancreatic c a ncer Because knowing your risk can mean early detection and prevention
 patient guide PancNext genetic testing for hereditary pancreatic c a ncer Because knowing your risk can mean early detection and prevention Know the Basics Risk factors for developing pancreatic cancer
patient guide PancNext genetic testing for hereditary pancreatic c a ncer Because knowing your risk can mean early detection and prevention Know the Basics Risk factors for developing pancreatic cancer
NGS for Cancer Predisposition
 NGS for Cancer Predisposition Colin Pritchard MD, PhD University of Washington Dept. of Lab Medicine AMP Companion Society Meeting USCAP Boston March 22, 2015 Disclosures I am an employee of the University
NGS for Cancer Predisposition Colin Pritchard MD, PhD University of Washington Dept. of Lab Medicine AMP Companion Society Meeting USCAP Boston March 22, 2015 Disclosures I am an employee of the University
Genetic Testing for BRCA1 and BRCA2 Genes
 Genetic Testing f BRCA1 and BRCA2 Genes MP9478 Covered Service: Pri Authization Required: Additional Infmation: Yes when meets criteria below Yes--as shown below Pre and post-test genetic counseling is
Genetic Testing f BRCA1 and BRCA2 Genes MP9478 Covered Service: Pri Authization Required: Additional Infmation: Yes when meets criteria below Yes--as shown below Pre and post-test genetic counseling is
Diagnostic and Treatment of Patients with Primary and Metastatic Breast Cancer
 Diagnostic and Treatment of Patients with Primary and Metastatic Breast Cancer Breast Cancer Risk and Prevention Breast Cancer Risk and Prevention Versions 2003 2017: Schmutzler / Albert / Blohmer / Fasching
Diagnostic and Treatment of Patients with Primary and Metastatic Breast Cancer Breast Cancer Risk and Prevention Breast Cancer Risk and Prevention Versions 2003 2017: Schmutzler / Albert / Blohmer / Fasching
Genetic causes 90% Other causes 10% No variants are found in known genes associated with ADPKD
 CLINICIAN PRODUCT SHEET Genome.One Polycystic Kidney Disease Test Genome.One offers a diagnostic genetic test for patients with polycystic kidney disease (PKD), with a focus on the most common form, autosomal
CLINICIAN PRODUCT SHEET Genome.One Polycystic Kidney Disease Test Genome.One offers a diagnostic genetic test for patients with polycystic kidney disease (PKD), with a focus on the most common form, autosomal
BowelGene. How do I know if I am at risk? Families with hereditary bowel cancer generally show one or more of the following clues:
 BowelGene BowelGene What is hereditary bowel cancer? Bowel cancer (also known as colorectal cancer) is the fourth most common cancer in the UK. Unfortunately 1 in 19 women and 1 in 14 men will develop
BowelGene BowelGene What is hereditary bowel cancer? Bowel cancer (also known as colorectal cancer) is the fourth most common cancer in the UK. Unfortunately 1 in 19 women and 1 in 14 men will develop
Family Assessment. Objectives. Comprehensive Family History Important Inexpensive Underutilized genetic tool
 Besides the BRCA genes, what else to consider in hereditary breast and ovarian cancer? Laurie M. Connors DNP, APNG, FNP-BC, AGN-BC Objectives Evaluate personal & family history to assess risk for hereditary
Besides the BRCA genes, what else to consider in hereditary breast and ovarian cancer? Laurie M. Connors DNP, APNG, FNP-BC, AGN-BC Objectives Evaluate personal & family history to assess risk for hereditary
GEN ETICS AN D GEN OM ICS IN CANCER PREVENTION AN D TREATM EN T. Robert Nathan Slotnick MD PhD Director, Medical Genetics and Genomics
 GEN ETICS AN D GEN OM ICS IN CANCER PREVENTION AN D TREATM EN T Robert Nathan Slotnick MD PhD Director, Medical Genetics and Genomics The Medical/Surgical/Radiation Oncologist s View of Genetics Cancer
GEN ETICS AN D GEN OM ICS IN CANCER PREVENTION AN D TREATM EN T Robert Nathan Slotnick MD PhD Director, Medical Genetics and Genomics The Medical/Surgical/Radiation Oncologist s View of Genetics Cancer
MEDICAL POLICY Genetic Testing for Breast and Ovarian Cancers
 POLICY: PG0067 ORIGINAL EFFECTIVE: 07/30/02 LAST REVIEW: 01/25/18 MEDICAL POLICY Genetic Testing for Breast and Ovarian Cancers GUIDELINES This policy does not certify benefits or authorization of benefits,
POLICY: PG0067 ORIGINAL EFFECTIVE: 07/30/02 LAST REVIEW: 01/25/18 MEDICAL POLICY Genetic Testing for Breast and Ovarian Cancers GUIDELINES This policy does not certify benefits or authorization of benefits,
Genetics and Cancer Care. Cynthia Forster-Gibson, MD, PhD and Loren Mackay- Loder, MSc Genetics Program, THP
 Genetics and Cancer Care Cynthia Forster-Gibson, MD, PhD and Loren Mackay- Loder, MSc Genetics Program, THP Faculty/Presenter Disclosure Faculty: Cynthia Forster-Gibson Relationships with commercial interests:
Genetics and Cancer Care Cynthia Forster-Gibson, MD, PhD and Loren Mackay- Loder, MSc Genetics Program, THP Faculty/Presenter Disclosure Faculty: Cynthia Forster-Gibson Relationships with commercial interests:
NEWSLETTER. Summer Test All Baby Boomers for Hepatitis C, Experts Say
 NEWSLETTER Summer 2016 Test All Baby Boomers for Hepatitis C, Experts Say This generation has the highest rate of infection, likely contracted decades ago WebMD News from HealthDay By Steven Reinberg,
NEWSLETTER Summer 2016 Test All Baby Boomers for Hepatitis C, Experts Say This generation has the highest rate of infection, likely contracted decades ago WebMD News from HealthDay By Steven Reinberg,
Genetics & Precision Medicine Cancer Care
 Genetics & Precision Medicine Cancer Care Johnathan M. Lancaster, MD PhD Chief Medical Officer Myriad Genetics Copyright 2015 Myriad Genetics, Inc., all rights reserved. www.myriad.com. Precision Medicine
Genetics & Precision Medicine Cancer Care Johnathan M. Lancaster, MD PhD Chief Medical Officer Myriad Genetics Copyright 2015 Myriad Genetics, Inc., all rights reserved. www.myriad.com. Precision Medicine
PREPARED FOR: U.S. Army Medical Research and Materiel Command Fort Detrick, Maryland
 AWARD NUMBER: W81XWH-13-1-0421 TITLE: The Fanconi Anemia BRCA Pathway as a Predictor of Benefit from Bevacizumab in a Large Phase III Clinical Trial in Ovarian Cancer PRINCIPAL INVESTIGATOR: Elizabeth
AWARD NUMBER: W81XWH-13-1-0421 TITLE: The Fanconi Anemia BRCA Pathway as a Predictor of Benefit from Bevacizumab in a Large Phase III Clinical Trial in Ovarian Cancer PRINCIPAL INVESTIGATOR: Elizabeth
The Center for PERSONALIZED DIAGNOSTICS
 The Center for PERSONALIZED DIAGNOSTICS Precision Diagnostics for Personalized Medicine A joint initiative between The Department of Pathology and Laboratory Medicine & The Abramson Cancer Center The (CPD)
The Center for PERSONALIZED DIAGNOSTICS Precision Diagnostics for Personalized Medicine A joint initiative between The Department of Pathology and Laboratory Medicine & The Abramson Cancer Center The (CPD)
Multigene Panel Testing for Hereditary Cancer Risk
 Multigene Panel Testing for Hereditary Cancer Risk Dana Zakalik, M.D. Director, Nancy and James Grosfeld Cancer Genetics Center Professor, OUWB Medical School MCC Annual Meeting November 4, 2015 Outline
Multigene Panel Testing for Hereditary Cancer Risk Dana Zakalik, M.D. Director, Nancy and James Grosfeld Cancer Genetics Center Professor, OUWB Medical School MCC Annual Meeting November 4, 2015 Outline
Hereditary Cancer Risk Assessment for Gynecological Cancers. FarrNezhatMD.com
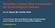 Hereditary Cancer Risk Assessment for Gynecological Cancers FarrNezhatMD.com Image credit: PLOS blogs 5-10% hereditary 10-20% 70-80% sporadic Genetic Changes and Cancer Cancer begins with a genetic
Hereditary Cancer Risk Assessment for Gynecological Cancers FarrNezhatMD.com Image credit: PLOS blogs 5-10% hereditary 10-20% 70-80% sporadic Genetic Changes and Cancer Cancer begins with a genetic
Monitoring the Health of Transplanted Organs DONOR GENOTYPING MANUAL
 Monitoring the Health of Transplanted Organs DONOR GENOTYPING MANUAL 10101 Innovation Drive. Suite 600. Wauwatosa, WI 53226 1-888-214-3151 MKT-008 v3.0 MKT-008 v3.0 TABLE OF CONTENTS I Intended Use...
Monitoring the Health of Transplanted Organs DONOR GENOTYPING MANUAL 10101 Innovation Drive. Suite 600. Wauwatosa, WI 53226 1-888-214-3151 MKT-008 v3.0 MKT-008 v3.0 TABLE OF CONTENTS I Intended Use...
Multiple gene sequencing for risk assessment in patients with early-onset or familial breast cancer
 www.impactjournals.com/oncotarget/ Oncotarget, Supplementary Materials 2016 Multiple gene sequencing for risk assessment in patients with early-onset or familial breast cancer Supplementary Materials Supplementary
www.impactjournals.com/oncotarget/ Oncotarget, Supplementary Materials 2016 Multiple gene sequencing for risk assessment in patients with early-onset or familial breast cancer Supplementary Materials Supplementary
patient guide ProstateNext genetic testing for hereditary prostate cancer Because knowing your risk can mean early detection and prevention
 patient guide ProstateNext genetic testing for hereditary prostate cancer Because knowing your risk can mean early detection and prevention Know the Basics Prostate cancer is the most common cancer in
patient guide ProstateNext genetic testing for hereditary prostate cancer Because knowing your risk can mean early detection and prevention Know the Basics Prostate cancer is the most common cancer in
Hereditary Cancer Update: What do GPOs need to know?
 Hereditary Cancer Update: What do GPOs need to know? Mary McCullum, RN, MSN, CON(C) Nurse Educator, Hereditary Cancer Program BC Cancer Agency October 1, 2016 Conflict of Interest Disclosure Nothing to
Hereditary Cancer Update: What do GPOs need to know? Mary McCullum, RN, MSN, CON(C) Nurse Educator, Hereditary Cancer Program BC Cancer Agency October 1, 2016 Conflict of Interest Disclosure Nothing to
Genetic Cancer Susceptibility Panels Using Next Generation Sequencing. Policy Specific Section: September 27, 2013 January 30, 2015
 Medical Policy Genetic Cancer Susceptibility Panels Using Next Generation Sequencing Type: Investigational / Experimental Policy Specific Section: Laboratory/Pathology Original Policy Date: Effective Date:
Medical Policy Genetic Cancer Susceptibility Panels Using Next Generation Sequencing Type: Investigational / Experimental Policy Specific Section: Laboratory/Pathology Original Policy Date: Effective Date:
COLON CANCER & GENETICS VERMONT COLORECTAL CANCER SUMMIT NOVEMBER 15, 2014
 COLON CANCER & GENETICS VERMONT COLORECTAL CANCER SUMMIT NOVEMBER 15, 2014 WENDY MCKINNON, MS, CGC CERTIFIED GENETIC COUNSELOR FAMILIAL CANCER PROGRAM UNIVERSIT Y OF VERMONT MEDICAL CENTER 1 CHARACTERISTICS
COLON CANCER & GENETICS VERMONT COLORECTAL CANCER SUMMIT NOVEMBER 15, 2014 WENDY MCKINNON, MS, CGC CERTIFIED GENETIC COUNSELOR FAMILIAL CANCER PROGRAM UNIVERSIT Y OF VERMONT MEDICAL CENTER 1 CHARACTERISTICS
ProstateGene What is hereditary prostate cancer? What are genes?
 ProstateGene ProstateGene What is hereditary prostate cancer? Prostate cancer is the most common cancer among men in the UK. Unfortunately, 1 in 8 men will be diagnosed with prostate cancer, with 75% being
ProstateGene ProstateGene What is hereditary prostate cancer? Prostate cancer is the most common cancer among men in the UK. Unfortunately, 1 in 8 men will be diagnosed with prostate cancer, with 75% being
NeoTYPE Cancer Profiles
 NeoTYPE Cancer Profiles 30+ Multimethod Assays for Hematologic Diseases and Solid Tumors Molecular FISH Anatomic Pathology The next generation of diagnostic, prognostic, and therapeutic assessment What
NeoTYPE Cancer Profiles 30+ Multimethod Assays for Hematologic Diseases and Solid Tumors Molecular FISH Anatomic Pathology The next generation of diagnostic, prognostic, and therapeutic assessment What
Information for You and Your Family
 Information for You and Your Family What is Prevention? Cancer prevention is action taken to lower the chance of getting cancer. In 2017, more than 1.6 million people will be diagnosed with cancer in the
Information for You and Your Family What is Prevention? Cancer prevention is action taken to lower the chance of getting cancer. In 2017, more than 1.6 million people will be diagnosed with cancer in the
MEDICAL GENOMICS LABORATORY. Next-Gen Sequencing and Deletion/Duplication Analysis of NF1 Only (NF1-NG)
 Next-Gen Sequencing and Deletion/Duplication Analysis of NF1 Only (NF1-NG) Ordering Information Acceptable specimen types: Fresh blood sample (3-6 ml EDTA; no time limitations associated with receipt)
Next-Gen Sequencing and Deletion/Duplication Analysis of NF1 Only (NF1-NG) Ordering Information Acceptable specimen types: Fresh blood sample (3-6 ml EDTA; no time limitations associated with receipt)
Yes when meets criteria below. Dean Health Plan covers when Medicare also covers the benefit.
 Genetic Testing for Lynch Syndrome MP9487 Covered Service: Prior Authorization Required: Additional Information: Yes when meets criteria below Yes-as shown below Pre and post test genetic counseling is
Genetic Testing for Lynch Syndrome MP9487 Covered Service: Prior Authorization Required: Additional Information: Yes when meets criteria below Yes-as shown below Pre and post test genetic counseling is
