ORGANIZATION OF ANTIBIOTIC AMPHOTERICIN B IN MODEL LIPID MEMBRANES. A MINI REVIEW
|
|
|
- Julius George
- 6 years ago
- Views:
Transcription
1 CELLULAR & MOLECULAR BIOLOGY LETTERS Volume 8, (2003) pp Received 30 September 2002 Accepted 13 February 2003 ORGANIZATION OF ANTIBIOTIC AMPHOTERICIN B IN MODEL LIPID MEMBRANES. A MINI REVIEW WIESŁAW I. GRUSZECKI 1 *, MARIUSZ GAGOŚ 2, MONIKA HEREĆ 1 and PETER KERNEN 3 1 Department of Biophysics, Institute of Physics, Maria Curie-Skłodowska University, Lublin, Poland, 2 Department of Physics, Agricultural University, Lublin, Poland, 3 Zyomyx, Inc., Research Road, Hayward, CA 94545, USA Abstract: Amphotericin B (AmB) is a polyene antibiotic frequently applied in the treatment of fungal infections. According to the general understanding, the mode of action of AmB is directly related to the molecular organization of the drug in the lipid environment, in particular to the formation of pore-like molecular aggregates. Electronic absorption and fluorescence techniques were applied to investigate formation of molecular aggregates of AmB in the lipid environment of liposomes and monomolecular layers formed at the argon-water interface. It appears that AmB dimers, stabilized by van der Waals interactions, are present in the membrane environment along with the aggregates formed by a greater number of molecules. Linear dichroism measurements reveal that AmB is distributed between two fractions of molecules, differently oriented with respect to the bilayer. Molecules in one fraction remain parallel to the plane of the membrane and molecules in the other one are perpendicular. Scanning Force Microscopy imaging of the surface topography of the monolayers formed with AmB in the presence of lipids reveals formation of pore-like structures characterized by the external diameter close to 17 Å and the internal diameter close to 6 Å. All the findings are discussed in terms of importance of the molecular organization of AmB in the pharmacological action, as well as of the toxic side effects of the drug. Key Words: Amphotericin B, Molecular Aggregates, Lipid Membranes *Corresponding author, Fax + (4881) , wieslaw@tytan.umcs.lublin.pl Abbreviations used: AmB - amphotericin B; SFM - scanning force microscopy; DPPC - dipalmitoylphosphatidylcholine
2 162 CELL. MOL. BIOL. LETT. Vol. 8. No INTRODUCTION Fig. 1. Chemical structure of amphotericin B Amphotericin B (AmB) is a polyene antibiotic frequently used in medical treatment of systemic fungal infections. According to the general understanding, the pharmacological action of the drug is directly associated with the formation by its molecules of pore structures across biomembranes, which considerably affects physiological ion transport [1, 2]. The ability of AmB to form hydrophilic pores results directly from the amphiphilic molecular structure of the drug (see Fig. 1). Selectivity of AmB toward lipid membranes of fungi is most probably related to the presence of ergosterol (instead of cholesterol), the sterol supposed to participate in the formation of membrane pores [1, 2]. On the other hand, the ionic channel activity has also been observed in the lipid membrane system containing AmB without any sterols [3]. Aggregation of AmB and pore formation in the ergosterol-free membranes is most probably a direct cause of the toxic side effects associated with medical treatment with the application of preparations based on AmB [4]. It has been proposed that not only formation of porous molecular structures, but also affection of the physical properties of the lipid bilayers brings about increased membrane permeability to ions [5]. This effect is expected to be particularly pronounced at low concentrations of AmB, promoting monomeric organization of the drug within the lipid phase. In this work we present our recent research, carried out with the application of electronic spectroscopic techniques, aimed to elucidate the molecular organization of AmB in the lipid membrane system. Understanding of molecular mechanisms that govern the organization of AmB in the lipid environment seems to be important not only for the understanding of various biological effects, but also for minimizing severe side effects of the drug. SPECTROSCOPY OF AMPHOTERICIN B IN LIPID MEMBRANES The electronic absorption spectrum of AmB present as a monomer in the 2- propanol:water mixture and embedded in lipid membranes is shown in Figure 2. A very distinct hypsochromic shift of the spectrum is indicative of the formation of molecular aggregates, the so-called card pack or H-type structures. Such a spectral effect may be interpreted as a manifestation of the formation of
3 CELLULAR & MOLECULAR BIOLOGY LETTERS propanol / H 2 O 4/6 5 mol % AmB in DPPC 0.4 Absorbance Wavelength [nm] Fig. 2. Absorption spectra of AmB: in monomeric form in solution of 2-propanol:water (4:6, v:v) - solid line, in aggregate form in suspension of DPPC liposomes containing 5 mol% AmB - dashed line. molecular pores: hydrophobic [6] or hydrophilic [7], depending on the polarity of the environment. On the other hand, a very similar spectral shift can be reproduced in the monomolecular layers formed with AmB [8]. The molecular organization of AmB in this system gave rise to the hypsochromic spectral shift, but an analysis of the sample topography, carried out with the application of Scanning Force Microscopy (SFM), revealed semi homogeneous distribution of molecules [8]. Interestingly, the presence of lipids in the monolayer system, at a concentration as low as 10 mol%, has been found to promote formation of pore structures by AmB molecules, characterized by the internal diameter of 6 Å and the external diameter of 17 Å [8]. The SFM image of such a structure is presented in Figure 3. The distance between the chromophores of the molecules involved in the formation of the hydrophilic pore (a possible model presented in Fig. 4) is comparable with the distance between the chromophores in the linear aggregate, and therefore both molecular structures, in principle, may give rise to very similar spectroscopic effects. The spectral shift accompanying the aggregation can be described by the following formula [9]: mπ ν m = ν mon + 2β cos (1) N + 1 where ν m is the position of the m-th excitation state in the spectra of the aggregated form, ν mon is the position of the electronic transition in the spectrum
4 164 CELL. MOL. BIOL. LETT. Vol. 8. No Fig. 3. Scanning Force Microscopy image of a two-component AmB-DPPC monolayer (9:1) deposited to mica. The 3D-view is shown with scan sizes of 2nm x 2nm and is plotted with z-range of 0.1 nm. A pore-like structure marked with a white circle. cylindrical aggregate polar groups chromophore AmB Fig. 4. Schematic representation of the cylindrical aggregate structure of AmB
5 CELLULAR & MOLECULAR BIOLOGY LETTERS mol% AmB in DPPC Fig. 5. Temperature dependences of absorption spectra of DPPC liposomes containing 1 mol% AmB of monomeric chromophores, N is the number of all exciton states, and m is the number of the exciton state considered. In the case of a maximum hypsochromic shift m=1. β is a dipole-dipole coupling matrix element [9] and is expressed as: µ β = 4πε η 2 mon 2 ( cosθ 3cos α) (2) 2 3 o R In the above formula µ mon is the dipole transition moment of a monomer, ε o is the dielectric permittivity of free space, η is the refractive index of the medium, and R is the distance between the centers of the transition dipoles of the nearest neighbors in the aggregate. The angle between monomeric transition moment vectors θ, and the angle between the transition moment vector and the axis connecting the centers of the transition moment vectors of neighboring molecules α, were assumed to be 0 deg. and 90 deg. in the case of H-aggregates of AmB. Figure 5 presents temperature dependencies of the electronic absorption spectra of AmB embedded in dipalmitoylphosphatidylcholine (DPPC) liposomes at a concentration of 1 mol%. The two spectral forms, corresponding to monomeric and aggregated AmB, can be distinguished in the temperature range both below and above the main phase transition (P β L α ). The short-wavelength absorption bands of AmB, indicative of aggregated molecular structures, which are particularly pronounced in the ordered phase of DPPC, decrease in the intensity at the temperatures above the phase transition.
6 166 CELL. MOL. BIOL. LETT. Vol. 8. No As may be seen, the short-wavelength absorption maximum shifts also from ca. 350 nm to 332 nm in this temperature range. Analyzing the formation of molecular structures manifested by the hypsochromic shift from 408 nm (monomeric AmB) to 332 nm one arrives, for example, at a model of a linear aggregate formed with 6 molecules or at a model of a cylindrical aggregate formed with 8 AmB molecules, in which polar groups are located inside a structure (see Fig. 4). In the first case the neighboring chromophores are spaced by R=4.9 Å and in the second by R=4.7 Å. A value of 11.3 Debye for the transition dipole of monomeric AmB was taken for calculations, as evaluated based on the integration of the absorption spectra in 2-propanol:water (see Fig. 2), and η was taken as 1.40, corresponding to the hydrophobic core of the DPPC membrane [7]. Other combinations of the number of molecules (between 6 and 10) and the distance of chromophores are also possible to reproduce the spectroscopic effects observed in the AmB-containing DPPC liposomes [7]. Interestingly, linear dichroism measurements carried out in the AmB-containing oriented multibilayers formed with DPPC reveal that the average dipole transition moment of monomeric AmB forms an angle close to 40 deg. with respect to the axis normal to the plane of the lipid membrane. Such a result can be understood as an expression of the presence of AmB in two, orthogonal pools of AmB, one oriented in the plane of the membrane (ca. 38 % molecules) and one perpendicular to it (ca. 62 % molecules). Figure 6A presents the electronic absorption spectra of AmB embedded in oriented DPPC multibilayers, recorded with the probing light beam polarized in the plane of incidence, and with a tilt angle of 0 deg. and 45 deg. The difference between these spectra (Fig. 6B) shows a distinct band in the short-wavelength region, thus indicating that aggregated molecular forms are primarily present in the fraction oriented in the plane of the membrane. Very likely, this fraction represents linear aggregates, not involved in the formation of pores across the membrane. On the other hand, one should not exclude the effects of horizontally oriented AmB molecules on the physical properties of lipid membranes. The chromophores of those molecules are also located in the hydrocarbon zone of the membrane (most probably in the interface region), but polar groups are very likely to form hydrogen bonds with head groups of lipids. Currently we are working on a method to distinguish the effects of these different pools of AmB on physical properties of lipid membranes, in particular directly related to passive ion transport. Despite the exact model of the aggregated forms of AmB that give rise to the hypsochromic shift, discussed above, this spectral effect may be applied to follow aggregation level of the drug in the lipid membrane environment. Recent studies carried out in our laboratory revealed that also dimeric forms of AmB have a specific spectroscopic signature [10]. A relatively high fluorescence quantum yield of the dimers of AmB (Q=4, excitation at 352 nm), as compared to the fluorescence quantum yield of the monomers (Q=0.06, excitation at 408 nm), enables discrimination of these two spectral forms.
7 CELLULAR & MOLECULAR BIOLOGY LETTERS A 0 deg 45 deg Absorbance B 45deg - 0deg Wavelength [nm] Fig. 6. Electronic absorption spectra of an oriented multilayer formed with 200 bilayers of DPPC containing 2 mol% AmB, recorded with polarized light in which the electric vector was oriented parallel with respect to the plane of incidence and the axis normal to the plane of the sample was tilted by 0 and 45 deg. with respect to the direction of the measuring beam (panel A) and the difference of these spectra (panel B). Fluorescence Intensity [a.u.] mol % AmB in DPPC Ex352(Em 471) Ex408(Em 606) Monomer/Dimer Ex 408 / Ex 352 (Em 606)(Em 471) Temperature [ o C] Fig. 7. Temperature dependences of the fluorescence signal corresponding to the dimeric (Ex 352 nm, Em 471 nm) and monomeric (Ex 408 nm, Em 606 nm) form of AmB embedded in DPPC liposomes at 5 mol% concentration and the ratio of fluorescence intensities, representing changes of monomers and dimers. Lines were fit in the temperature range below 35 o C and above 45 o C. The line cross-section points at the temperature of the main phase transition of lipid membranes formed with DPPC (T m ). T m
8 168 CELL. MOL. BIOL. LETT. Vol. 8. No Monomer/Aggregate Absorption measurements 1mol% AmB in DPPC Monomer/Dimer Fluorescence measurements Ex408/Ex352 (Em606)(Em471) 0.02 T m Temperature [ o C] Fig. 8. Temperature dependences of the ratio of Gaussian components representing the 0-0 transition of the monomeric structure (centered at 408 nm) and the aggregated structure (centered at 334 nm) produced on the basis of the Gaussian analysis of absorption spectra of DPPC liposomes containing 1 mol% AmB (upper panel) and temperature dependences of the ratio of fluorescence intensities, representing changes of proportion of monomers (Ex 408 nm, Em 606 nm) and dimers (Ex 352 nm, Em 471) of AmB embedded in DPPC liposomes at 1 mol%. Figure 7 presents the temperature dependencies of fluorescence signals corresponding to the dimeric and monomeric forms of AmB embedded in DPPC liposomes. The ratio of these traces displays a distinct transition that corresponds to the main phase transition of DPPC. As may be seen, the rise in the temperature of liposomes results in a relative increase in the population of monomers. A very similar effect was observed in the case of AmB aggregated structures, as demonstrated by means of the Gaussian analysis of the electronic absorption spectra of AmB-containing DPPC liposomes (see Fig. 8). Figure 9 presents concentration profiles of the dimer to monomer and aggregate to monomer ratios, based on fluorescence and absorption measurements, respectively. Apparently, the maximum that appears at 1 mol% AmB in DPPC in the dimer concentration, corresponds to the minimum in the concentration of the aggregated structures. Such an apparent paradox may be understood in terms of composition of molecular structures of AmB at the expense of dimers. The
9 CELLULAR & MOLECULAR BIOLOGY LETTERS A I D /I M B 1.2 I A /I M 0.8 Fig. 9. Concentration profile of the AmB dimerization (panel A) and aggregation (panel B) level expressed as the I D /I M and I A /I M ratio, respectively, corresponding to 35 o C. The level of dimers and aggregates relative to monomers was determined on the basis of fluorescence and absorption measurements, as explained in the caption to Fig. 8. appearance of the minimum in the aggregation level corresponding to a certain concentration of AmB, itself, seems to be particularly interesting and may suggest different (opposite?) biological effects of the drug, depending on its actual local concentration in the lipid phase. Acknowledgements. This work was supported by the Polish Committee for Scientific Research (KBN) Grant No. 3P04A REFERENCES mol % AmB 1. De Kruijff, B. and Demel, R.A. Polyene antibiotic-sterol interaction in membranes of Acholeplasma Laidlawii cells and lecithin liposomes; III Molecular structure of the polyene antibiotic-cholesterol complex. Biochim. Biophys. Acta. 339 (1974)
10 170 CELL. MOL. BIOL. LETT. Vol. 8. No Baginski, M., Resat, H. and McCammon, J.A. Molecular properties of amphotericin B membrane channel: a molecular dynamics simulation. Mol. Pharmacol. 52 (1997) Cotero, B.V., Rebolledo-Antunez, S. and Ortega-Blake, I. On the role of sterol in the formation of the amphotericin B channel. Biochim. Biophys. Acta 1375 (1998) Hartsel, S. and Bolard, J. Amphotericin B: new life for an old drug. TiPS 17 (1996) Wójtowicz, K., Gruszecki, W. I., Walicka, M. and Barwicz, J. Effect of amphotericin B on dipalmitoylphosphatidylcholine membranes: calorimetry, ultrasound absorption and monolayer technique studies. Biochim. Biophys. Acta 1373 (1998) Barwicz, J., Gruszecki, W.I. and Gruda I. Spontaneous Organization of Amphotericin B in Aqueous Medium. J. Colloid Interf. Sci. 158 (1993) Gagoś, M., Koper, R. and Gruszecki, W.I. Spectrophotometric analysis of organisation of dipalmitoylphosphatidylcholine bilayers containing the polyene antibiotic amphotericin B. Biochim. Biophys. Acta 1511 (2001) Gruszecki, W.I., Gagoś, M. and Kernen, P. Polyene Antibiotic Amphotericin B in Monomolecular Layers: Spectrophotometric and Scanning Force Microscopic Analysis. FEBS Lett. 524 (2002) Parkash, J., Robblee, J. H., Agnew, J., Gibbs, E., Collings, P., Pasternack, R. F. and de Paula, J. C. Depolarized Resonance Light Scattering by Porphyrin and Chlorophyll a Aggregates. Biophys. J. 74 (1998) Gruszecki, W.I., Gagoś, M. and Hereć, M. Dimers of polyene antibiotic amphotericin B detected by means of fluorescence spectroscopy: molecular organization in solution and in lipid membranes. J. Photochem. Photobiol. B: Biol. 69 (2003)
Effect of antibiotic amphotericin B on structural and dynamic properties of lipid membranes formed with egg yolk phosphatidylcholine
 Chemistry and Physics of Lipids 147 (2007) 78 86 Effect of antibiotic amphotericin B on structural and dynamic properties of lipid membranes formed with egg yolk phosphatidylcholine Monika Hereć a,b, Akhmed
Chemistry and Physics of Lipids 147 (2007) 78 86 Effect of antibiotic amphotericin B on structural and dynamic properties of lipid membranes formed with egg yolk phosphatidylcholine Monika Hereć a,b, Akhmed
Molecular organization of antifungal antibiotic amphotericin B in lipid monolayers studied by means of Fluorescence Lifetime Imaging Microscopy
 Molecular organization of antifungal antibiotic amphotericin B in lipid monolayers studied by means of Fluorescence Lifetime Imaging Microscopy Wieslaw I. Gruszecki, Rafal Luchowski, Mariusz Gagoś, Marta
Molecular organization of antifungal antibiotic amphotericin B in lipid monolayers studied by means of Fluorescence Lifetime Imaging Microscopy Wieslaw I. Gruszecki, Rafal Luchowski, Mariusz Gagoś, Marta
Polarization and Circular Dichroism (Notes 17)
 Polarization and Circular Dichroism - 2014 (Notes 17) Since is vector, if fix molec. orient., E-field interact (absorb) with molecule differently when change E-orientation (polarization) Transitions can
Polarization and Circular Dichroism - 2014 (Notes 17) Since is vector, if fix molec. orient., E-field interact (absorb) with molecule differently when change E-orientation (polarization) Transitions can
Liquid crystals; biological and artificial membranes
 Liquid crystals; biological and artificial membranes Dr. István Voszka Liquid crystals: Intermediate state between liquids and crystalline solids anisotropic liquids. (anisotropy = the physical properties
Liquid crystals; biological and artificial membranes Dr. István Voszka Liquid crystals: Intermediate state between liquids and crystalline solids anisotropic liquids. (anisotropy = the physical properties
Lecture 15. Membrane Proteins I
 Lecture 15 Membrane Proteins I Introduction What are membrane proteins and where do they exist? Proteins consist of three main classes which are classified as globular, fibrous and membrane proteins. A
Lecture 15 Membrane Proteins I Introduction What are membrane proteins and where do they exist? Proteins consist of three main classes which are classified as globular, fibrous and membrane proteins. A
Chemical Surface Transformation 1
 Chemical Surface Transformation 1 Chemical reactions at Si H surfaces (inorganic and organic) can generate very thin films (sub nm thickness up to µm): inorganic layer formation by: thermal conversion:
Chemical Surface Transformation 1 Chemical reactions at Si H surfaces (inorganic and organic) can generate very thin films (sub nm thickness up to µm): inorganic layer formation by: thermal conversion:
Model for measurement of water layer thickness under lipid bilayers by surface plasmon resonance
 Model for measurement of water layer thickness under lipid bilayers by surface plasmon resonance Koyo Watanabe Unit of Measurement Technology, CEMIS-OULU, University of Oulu, PO Box 51, 87101 Kajaani,
Model for measurement of water layer thickness under lipid bilayers by surface plasmon resonance Koyo Watanabe Unit of Measurement Technology, CEMIS-OULU, University of Oulu, PO Box 51, 87101 Kajaani,
Polyoxometalate Macroion Induced Phase and Morphology
 Polyoxometalate Macroion Induced Phase and Morphology Instability of Lipid Membrane Benxin Jing a, Marie Hutin c, Erin Connor a, Leroy Cronin c,* and Yingxi Zhu a,b,* a Department of Chemical and Biomolecular
Polyoxometalate Macroion Induced Phase and Morphology Instability of Lipid Membrane Benxin Jing a, Marie Hutin c, Erin Connor a, Leroy Cronin c,* and Yingxi Zhu a,b,* a Department of Chemical and Biomolecular
0.5 nm nm acyl tail region (hydrophobic) 1.5 nm. Hydrophobic repulsion organizes amphiphilic molecules: These scales are 5 10xk B T:
 Lecture 31: Biomembranes: The hydrophobic energy scale and membrane behavior 31.1 Reading for Lectures 30-32: PKT Chapter 11 (skip Ch. 10) Upshot of last lecture: Generic membrane lipid: Can be cylindrical
Lecture 31: Biomembranes: The hydrophobic energy scale and membrane behavior 31.1 Reading for Lectures 30-32: PKT Chapter 11 (skip Ch. 10) Upshot of last lecture: Generic membrane lipid: Can be cylindrical
Photochemical Applications to the Study of Complexity Phospholipid Bilayer Environments
 Virginia Commonwealth University VCU Scholars Compass Theses and Dissertations Graduate School 2006 Photochemical Applications to the Study of Complexity Phospholipid Bilayer Environments Christopher John
Virginia Commonwealth University VCU Scholars Compass Theses and Dissertations Graduate School 2006 Photochemical Applications to the Study of Complexity Phospholipid Bilayer Environments Christopher John
Chapter 12: Membranes. Voet & Voet: Pages
 Chapter 12: Membranes Voet & Voet: Pages 390-415 Slide 1 Membranes Essential components of all living cells (define boundry of cells) exclude toxic ions and compounds; accumulation of nutrients energy
Chapter 12: Membranes Voet & Voet: Pages 390-415 Slide 1 Membranes Essential components of all living cells (define boundry of cells) exclude toxic ions and compounds; accumulation of nutrients energy
Supporting Information
 Supporting Information Size Tunable ZnO Nanoparticles to Enhance Electron Injection in Solution Processed QLEDs Jiangyong Pan,, Jing Chen,, Qianqian Huang, Qasim Khan, Xiang Liu, Zhi Tao, Zichen Zhang,
Supporting Information Size Tunable ZnO Nanoparticles to Enhance Electron Injection in Solution Processed QLEDs Jiangyong Pan,, Jing Chen,, Qianqian Huang, Qasim Khan, Xiang Liu, Zhi Tao, Zichen Zhang,
Evidence of intrinsic ferromagnetism in individual dilute magnetic semiconducting nanostructures O-K. (a) Zn-L Zn-L 2,3
 SUPPLEMENTARY INFORMATION Evidence of intrinsic ferromagnetism in individual dilute magnetic semiconducting nanostructures O-K (a) O-K Fe-L Co-L 2,3 2,3 Zn-L Zn-L 2,3 2,3 (b) Intensity (a. u.) 500 750
SUPPLEMENTARY INFORMATION Evidence of intrinsic ferromagnetism in individual dilute magnetic semiconducting nanostructures O-K (a) O-K Fe-L Co-L 2,3 2,3 Zn-L Zn-L 2,3 2,3 (b) Intensity (a. u.) 500 750
Membrane lipid peroxidation by ultrasound: Mechanism and implications
 J. Biosci., Vol. 15, Number 3, September 1990, pp. 211 215. Printed in India. Membrane lipid peroxidation by ultrasound: Mechanism and implications A. K. JANA, S. AGARWAL and S. N. CHATTERJEE* Biophysics
J. Biosci., Vol. 15, Number 3, September 1990, pp. 211 215. Printed in India. Membrane lipid peroxidation by ultrasound: Mechanism and implications A. K. JANA, S. AGARWAL and S. N. CHATTERJEE* Biophysics
Structure and Phase Behaviour of Binary Mixtures of Cholesterol with DPPC and DMPC
 Chapter 3 Structure and Phase Behaviour of Binary Mixtures of Cholesterol with DPPC and DMPC 3.1 Introduction As discussed in chapter 1, phospholipids and cholesterol are important constituents of plasma
Chapter 3 Structure and Phase Behaviour of Binary Mixtures of Cholesterol with DPPC and DMPC 3.1 Introduction As discussed in chapter 1, phospholipids and cholesterol are important constituents of plasma
8 Influence of permeation modulators on the behaviour of a SC lipid model mixture
 8 Influence of permeation modulators on the behaviour of a SC lipid model mixture 8.1 Introduction In the foregoing parts of this thesis, a model membrane system of SC lipids has been developed and characterized.
8 Influence of permeation modulators on the behaviour of a SC lipid model mixture 8.1 Introduction In the foregoing parts of this thesis, a model membrane system of SC lipids has been developed and characterized.
Life Sciences 1a. Practice Problems 4
 Life Sciences 1a Practice Problems 4 1. KcsA, a channel that allows K + ions to pass through the membrane, is a protein with four identical subunits that form a channel through the center of the tetramer.
Life Sciences 1a Practice Problems 4 1. KcsA, a channel that allows K + ions to pass through the membrane, is a protein with four identical subunits that form a channel through the center of the tetramer.
3.1 Background. Preformulation Studies
 Preformulation Studies 3.1 Background Delivery of any drug requires a suitable dosage form to get optimum therapeutic effects. The development of such dosage forms fundamental properties of the drug molecule
Preformulation Studies 3.1 Background Delivery of any drug requires a suitable dosage form to get optimum therapeutic effects. The development of such dosage forms fundamental properties of the drug molecule
Microtubule Teardrop Patterns
 Supporting Information Microtubule Teardrop Patterns Kosuke Okeyoshi 1, Ryuzo Kawamura 1, Ryo Yoshida 2, and Yoshihito Osada 1 * 1 RIKEN Advanced Science Institute, 2-1 Hirosawa, Wako-shi, Saitama 351-0198,
Supporting Information Microtubule Teardrop Patterns Kosuke Okeyoshi 1, Ryuzo Kawamura 1, Ryo Yoshida 2, and Yoshihito Osada 1 * 1 RIKEN Advanced Science Institute, 2-1 Hirosawa, Wako-shi, Saitama 351-0198,
SDS-Assisted Protein Transport Through Solid-State Nanopores
 Supplementary Information for: SDS-Assisted Protein Transport Through Solid-State Nanopores Laura Restrepo-Pérez 1, Shalini John 2, Aleksei Aksimentiev 2 *, Chirlmin Joo 1 *, Cees Dekker 1 * 1 Department
Supplementary Information for: SDS-Assisted Protein Transport Through Solid-State Nanopores Laura Restrepo-Pérez 1, Shalini John 2, Aleksei Aksimentiev 2 *, Chirlmin Joo 1 *, Cees Dekker 1 * 1 Department
Supporting material. Membrane permeation induced by aggregates of human islet amyloid polypeptides
 Supporting material Membrane permeation induced by aggregates of human islet amyloid polypeptides Chetan Poojari Forschungszentrum Jülich GmbH, Institute of Complex Systems: Structural Biochemistry (ICS-6),
Supporting material Membrane permeation induced by aggregates of human islet amyloid polypeptides Chetan Poojari Forschungszentrum Jülich GmbH, Institute of Complex Systems: Structural Biochemistry (ICS-6),
Interactions of Polyethylenimines with Zwitterionic and. Anionic Lipid Membranes
 Interactions of Polyethylenimines with Zwitterionic and Anionic Lipid Membranes Urszula Kwolek, Dorota Jamróz, Małgorzata Janiczek, Maria Nowakowska, Paweł Wydro, Mariusz Kepczynski Faculty of Chemistry,
Interactions of Polyethylenimines with Zwitterionic and Anionic Lipid Membranes Urszula Kwolek, Dorota Jamróz, Małgorzata Janiczek, Maria Nowakowska, Paweł Wydro, Mariusz Kepczynski Faculty of Chemistry,
PROCEEDINGS OF THE YEREVAN STATE UNIVERSITY
 PROCEEDINGS OF THE YEREVAN STATE UNIVERSITY Physical and Mathematical Sciences 2018, 52(3), p. 217 221 P h y s i c s STUDY OF THE SWELLING OF THE PHOSPHOLIPID BILAYER, DEPENDING ON THE ANGLE BETWEEN THE
PROCEEDINGS OF THE YEREVAN STATE UNIVERSITY Physical and Mathematical Sciences 2018, 52(3), p. 217 221 P h y s i c s STUDY OF THE SWELLING OF THE PHOSPHOLIPID BILAYER, DEPENDING ON THE ANGLE BETWEEN THE
Pressure Modulation of the Enzymatic Activity of. Phospholipase A2, a Putative Membraneassociated
 SUPPORTING INFORMATION Pressure Modulation of the Enzymatic Activity of Phospholipase A2, a Putative Membraneassociated Pressure Sensor Saba Suladze, Suleyman Cinar, Benjamin Sperlich, and Roland Winter*
SUPPORTING INFORMATION Pressure Modulation of the Enzymatic Activity of Phospholipase A2, a Putative Membraneassociated Pressure Sensor Saba Suladze, Suleyman Cinar, Benjamin Sperlich, and Roland Winter*
The Transport and Organization of Cholesterol in Planar Solid-Supported Lipid Bilayer Depend on the Phospholipid Flip-Flop Rate
 Supporting Information The Transport and Organization of Cholesterol in Planar Solid-Supported Lipid Bilayer Depend on the Phospholipid Flip-Flop Rate Ting Yu, 1,2 Guangnan Zhou, 1 Xia Hu, 1,2 Shuji Ye
Supporting Information The Transport and Organization of Cholesterol in Planar Solid-Supported Lipid Bilayer Depend on the Phospholipid Flip-Flop Rate Ting Yu, 1,2 Guangnan Zhou, 1 Xia Hu, 1,2 Shuji Ye
N-Methyl-N-D-fructosyl amphotericin B methyl ester (MF-AME), a novel antifungal agent of low toxicity: Monomer/micelle control over selective toxicity
 Vol. 47 No. 1/2000 121 131 QUARTERLY N-Methyl-N-D-fructosyl amphotericin B methyl ester (MF-AME), a novel antifungal agent of low toxicity: Monomer/micelle control over selective toxicity Barbara Cybulska
Vol. 47 No. 1/2000 121 131 QUARTERLY N-Methyl-N-D-fructosyl amphotericin B methyl ester (MF-AME), a novel antifungal agent of low toxicity: Monomer/micelle control over selective toxicity Barbara Cybulska
Food protein powders classification and discrimination by FTIR spectroscopy and principal component analysis
 APPLICATION NOTE AN53037 Food protein powders classification and discrimination by FTIR spectroscopy and principal component analysis Author Ron Rubinovitz, Ph.D. Thermo Fisher Scientific Key Words FTIR,
APPLICATION NOTE AN53037 Food protein powders classification and discrimination by FTIR spectroscopy and principal component analysis Author Ron Rubinovitz, Ph.D. Thermo Fisher Scientific Key Words FTIR,
1.4 Page 1 Cell Membranes S. Preston 1
 AS Unit 1: Basic Biochemistry and Cell Organisation Name: Date: Topic 1.3 Cell Membranes and Transport Page 1 1.3 Cell Membranes and Transport from your syllabus l. Cell Membrane Structure 1. Read and
AS Unit 1: Basic Biochemistry and Cell Organisation Name: Date: Topic 1.3 Cell Membranes and Transport Page 1 1.3 Cell Membranes and Transport from your syllabus l. Cell Membrane Structure 1. Read and
SAXS on lipid structures
 Practical Course in Biophysics, Experiment R2b SAXS on lipid structures Summer term 2015 Room: Advisor: X-ray lab at LS Rädler, NU111 Stefan Fischer Tel: +49-(0)89-2180-1459 Email: stefan.f.fischer@physik.lmu.de
Practical Course in Biophysics, Experiment R2b SAXS on lipid structures Summer term 2015 Room: Advisor: X-ray lab at LS Rädler, NU111 Stefan Fischer Tel: +49-(0)89-2180-1459 Email: stefan.f.fischer@physik.lmu.de
bio-mof-1 DMASM Wavenumber (cm -1 ) Supplementary Figure S1 FTIR spectra of bio-mof-1, DMASMI, and bio-mof-1 DMASM.
 bio-mof-1 Transmittance bio-mof-1 DMASM DMASMI 2000 1500 1000 500 Wavenumber (cm -1 ) Supplementary Figure S1 FTIR spectra of bio-mof-1, DMASMI, and bio-mof-1 DMASM. Intensity (a.u.) bio-mof-1 DMASM as
bio-mof-1 Transmittance bio-mof-1 DMASM DMASMI 2000 1500 1000 500 Wavenumber (cm -1 ) Supplementary Figure S1 FTIR spectra of bio-mof-1, DMASMI, and bio-mof-1 DMASM. Intensity (a.u.) bio-mof-1 DMASM as
Antibiotic assisted molecular ion transport across a membrane in real time
 Antibiotic assisted molecular ion transport across a membrane in real time Jian Liu, Xiaoming Shang, Rebecca Pompano and Kenneth B. Eisenthal* Department of Chemistry, Columbia University, New York, NY
Antibiotic assisted molecular ion transport across a membrane in real time Jian Liu, Xiaoming Shang, Rebecca Pompano and Kenneth B. Eisenthal* Department of Chemistry, Columbia University, New York, NY
Chapter 1 Membrane Structure and Function
 Chapter 1 Membrane Structure and Function Architecture of Membranes Subcellular fractionation techniques can partially separate and purify several important biological membranes, including the plasma and
Chapter 1 Membrane Structure and Function Architecture of Membranes Subcellular fractionation techniques can partially separate and purify several important biological membranes, including the plasma and
Organisation of xanthophyll pigments lutein and zeaxanthin in lipid membranes formed with dipalmitoylphosphatidylcholine
 Biochimica et Biophysica Acta 1509 (2000) 255^263 www.elsevier.com/locate/bba Organisation of xanthophyll pigments lutein and zeaxanthin in lipid membranes formed with dipalmitoylphosphatidylcholine Agnieszka
Biochimica et Biophysica Acta 1509 (2000) 255^263 www.elsevier.com/locate/bba Organisation of xanthophyll pigments lutein and zeaxanthin in lipid membranes formed with dipalmitoylphosphatidylcholine Agnieszka
Quiz 8 Introduction to Polymers (Chemistry)
 051117 Quiz 8 Introduction to Polymers (Chemistry) (Figures from Heimenz Colloid Sci.) 1) Surfactants are amphiphilic molecules (molecules having one end hydrophobic and the other hydrophilic) and are
051117 Quiz 8 Introduction to Polymers (Chemistry) (Figures from Heimenz Colloid Sci.) 1) Surfactants are amphiphilic molecules (molecules having one end hydrophobic and the other hydrophilic) and are
Structure of Dipalmitoylphosphatidylcholine/Cholesterol Bilayer at Low and High Cholesterol Concentrations: Molecular Dynamics Simulation
 Biophysical Journal Volume 77 October 1999 2075 2089 2075 Structure of Dipalmitoylphosphatidylcholine/Cholesterol Bilayer at Low and High Cholesterol Concentrations: Molecular Dynamics Simulation Alexander
Biophysical Journal Volume 77 October 1999 2075 2089 2075 Structure of Dipalmitoylphosphatidylcholine/Cholesterol Bilayer at Low and High Cholesterol Concentrations: Molecular Dynamics Simulation Alexander
The Cell Membrane (Ch. 7)
 The Cell Membrane (Ch. 7) Phospholipids Phosphate head hydrophilic Fatty acid tails hydrophobic Arranged as a bilayer Phosphate attracted to water Fatty acid repelled by water Aaaah, one of those structure
The Cell Membrane (Ch. 7) Phospholipids Phosphate head hydrophilic Fatty acid tails hydrophobic Arranged as a bilayer Phosphate attracted to water Fatty acid repelled by water Aaaah, one of those structure
Boundary Lipid bilayer Selectively Permeable Fluid mosaic of lipids and proteins Contains embedded proteins
 1 Boundary Lipid bilayer Selectively Permeable Fluid mosaic of lipids and proteins Contains embedded proteins 2 Phosphate head hydrophilic Fatty acid tails hydrophobic Amphipathic Phosphate attracted to
1 Boundary Lipid bilayer Selectively Permeable Fluid mosaic of lipids and proteins Contains embedded proteins 2 Phosphate head hydrophilic Fatty acid tails hydrophobic Amphipathic Phosphate attracted to
Phosphatidylcholines are a class of glycerophospholipids which along with other phospholipids
 Phosphatidylcholine Phosphatidylcholines are a class of glycerophospholipids which along with other phospholipids account for more than half of the lipids in most membranes. Phosphatidylcholines can further
Phosphatidylcholine Phosphatidylcholines are a class of glycerophospholipids which along with other phospholipids account for more than half of the lipids in most membranes. Phosphatidylcholines can further
Membrane Structure and Function
 Membrane Structure and Function Chapter 7 Objectives Define the following terms: amphipathic molecules, aquaporins, diffusion Distinguish between the following pairs or sets of terms: peripheral and integral
Membrane Structure and Function Chapter 7 Objectives Define the following terms: amphipathic molecules, aquaporins, diffusion Distinguish between the following pairs or sets of terms: peripheral and integral
Plasmon resonance spectroscopy: probing molecular interactions at surfaces and interfaces
 Spectroscopy 15 (2001) 161 175 161 IOS Press Plasmon resonance spectroscopy: probing molecular interactions at surfaces and interfaces Zdzislaw Salamon and Gordon Tollin Department of Biochemistry and
Spectroscopy 15 (2001) 161 175 161 IOS Press Plasmon resonance spectroscopy: probing molecular interactions at surfaces and interfaces Zdzislaw Salamon and Gordon Tollin Department of Biochemistry and
Changes in Vesicular Membrane ESR Spin Label Parameters Upon Isotope Solvent Substitution
 Gen. Physiol. Biophys. (1987). 6, 297 302 297 Short communication Changes in Vesicular Membrane ESR Spin Label Parameters Upon Isotope Solvent Substitution V. I. LOBYSHEV 1, T. HIANIK 2, M. MASAROVA 1
Gen. Physiol. Biophys. (1987). 6, 297 302 297 Short communication Changes in Vesicular Membrane ESR Spin Label Parameters Upon Isotope Solvent Substitution V. I. LOBYSHEV 1, T. HIANIK 2, M. MASAROVA 1
Physical Pharmacy. Interfacial phenomena. Khalid T Maaroof MSc. Pharmaceutical sciences School of pharmacy Pharmaceutics department
 Physical Pharmacy Interfacial phenomena Khalid T Maaroof MSc. Pharmaceutical sciences School of pharmacy Pharmaceutics department 1 Introduction The boundary between two phases is generally described as
Physical Pharmacy Interfacial phenomena Khalid T Maaroof MSc. Pharmaceutical sciences School of pharmacy Pharmaceutics department 1 Introduction The boundary between two phases is generally described as
Interactions between Bisphosphate. Geminis and Sodium Lauryl Ether
 Chapter 5 Interactions between Bisphosphate Geminis and Sodium Lauryl Ether Sulphate 110 5.1 Introduction The physiochemical and surface active properties of mixed surfactants are of more interest and
Chapter 5 Interactions between Bisphosphate Geminis and Sodium Lauryl Ether Sulphate 110 5.1 Introduction The physiochemical and surface active properties of mixed surfactants are of more interest and
The Interaction between Lipid Bilayers and Biological Membranes. Chapter 18
 The Interaction between Lipid Bilayers and Biological Membranes Chapter 18 Introduction Membrane & Phospholipid Bilayer Structure Membrane Lipid bilayer 2 Introduction Forces Acting between Surfaces in
The Interaction between Lipid Bilayers and Biological Membranes Chapter 18 Introduction Membrane & Phospholipid Bilayer Structure Membrane Lipid bilayer 2 Introduction Forces Acting between Surfaces in
Supplementary Figures
 Supplementary Figures Absorption 4 3 2 1 Intensity Energy U(R) relaxation ~~~ ~~~~~~ 2 3 4 1 S S 1 2 3 4 1 Fluoescence 4 3 2 1 Intensity H-aggregation ~~~~ J-aggregation Absorption Emission Vibrational
Supplementary Figures Absorption 4 3 2 1 Intensity Energy U(R) relaxation ~~~ ~~~~~~ 2 3 4 1 S S 1 2 3 4 1 Fluoescence 4 3 2 1 Intensity H-aggregation ~~~~ J-aggregation Absorption Emission Vibrational
Electrical Properties of Lipid Membrane Role of Bathing Solution
 Internet Electronic Journal of Molecular Design 2003, 2, 000 000 Electrical Properties of Lipid Membrane Role of Bathing Solution D. Ghosh, 1, * S. Manna, 1 S. De, 2 R. Basu, 3 and P. Nandy 1 1 Department
Internet Electronic Journal of Molecular Design 2003, 2, 000 000 Electrical Properties of Lipid Membrane Role of Bathing Solution D. Ghosh, 1, * S. Manna, 1 S. De, 2 R. Basu, 3 and P. Nandy 1 1 Department
BIOPHYSICS II. By Prof. Xiang Yang Liu Department of Physics,
 BIOPHYSICS II By Prof. Xiang Yang Liu Department of Physics, NUS 1 Hydrogen bond and the stability of macromolecular structure Membrane Model Amphiphilic molecule self-assembly at the surface and din the
BIOPHYSICS II By Prof. Xiang Yang Liu Department of Physics, NUS 1 Hydrogen bond and the stability of macromolecular structure Membrane Model Amphiphilic molecule self-assembly at the surface and din the
Supplementary information for Effects of Stretching Speed on. Mechanical Rupture of Phospholipid/Cholesterol Bilayers: Molecular
 Supplementary information for Effects of Stretching Speed on Mechanical Rupture of Phospholipid/Cholesterol Bilayers: Molecular Dynamics Simulation Taiki Shigematsu, Kenichiro Koshiyama*, and Shigeo Wada
Supplementary information for Effects of Stretching Speed on Mechanical Rupture of Phospholipid/Cholesterol Bilayers: Molecular Dynamics Simulation Taiki Shigematsu, Kenichiro Koshiyama*, and Shigeo Wada
Roles of Sterol Derivatives in Regulating the Properties of Phospholipid Bilayer Systems
 SUPPORTING INFORMATION Roles of Sterol Derivatives in Regulating the Properties of Phospholipid Bilayer Systems Tham Thi Bui, Keishi Suga, Hiroshi Umakoshi* Division of Chemical Engineering, Graduate School
SUPPORTING INFORMATION Roles of Sterol Derivatives in Regulating the Properties of Phospholipid Bilayer Systems Tham Thi Bui, Keishi Suga, Hiroshi Umakoshi* Division of Chemical Engineering, Graduate School
Drugs, Bugs & Neutrons where are we and where do we go from here?
 Drugs, Bugs & Neutrons where are we and where do we go from here? Dave Barlow, Pharmacy Department, King s College London The Future and Next Generation Capabilities of Accelerator-driven Neutron and Muon
Drugs, Bugs & Neutrons where are we and where do we go from here? Dave Barlow, Pharmacy Department, King s College London The Future and Next Generation Capabilities of Accelerator-driven Neutron and Muon
AFM In Liquid: A High Sensitivity Study On Biological Membranes
 University of Wollongong Research Online Faculty of Science - Papers (Archive) Faculty of Science, Medicine and Health 2006 AFM In Liquid: A High Sensitivity Study On Biological Membranes Michael J. Higgins
University of Wollongong Research Online Faculty of Science - Papers (Archive) Faculty of Science, Medicine and Health 2006 AFM In Liquid: A High Sensitivity Study On Biological Membranes Michael J. Higgins
Internal Electrostatic Potentials in Bilayers: Measuring and Controlling Dipole Potentials in Lipid Vesicles
 Biophysical Journal Volume 65 July 1993 289-299 Internal Electrostatic Potentials in Bilayers: Measuring and Controlling Dipole Potentials in Lipid Vesicles 289 J. Craig Franklin and David S. Cafiso From
Biophysical Journal Volume 65 July 1993 289-299 Internal Electrostatic Potentials in Bilayers: Measuring and Controlling Dipole Potentials in Lipid Vesicles 289 J. Craig Franklin and David S. Cafiso From
Transport. Slide 1 of 47. Copyright Pearson Prentice Hall
 & Transport 1 of 47 Learning Targets TN Standard CLE 3216.1.3 Explain how materials move into and out of cells. CLE 3216.1.5 Investigate how proteins regulate the internal environment of a cell through
& Transport 1 of 47 Learning Targets TN Standard CLE 3216.1.3 Explain how materials move into and out of cells. CLE 3216.1.5 Investigate how proteins regulate the internal environment of a cell through
Supporting Information
 Supporting Information High Efficiency Colloidal Quantum Dot Photovoltaics via Robust Self- Assembled Monolayers Gi-Hwan Kim,,, F. Pelayo García de Arquer,, Yung Jin Yoon, Xinzheng Lan, Men gxia Liu, Oleksandr
Supporting Information High Efficiency Colloidal Quantum Dot Photovoltaics via Robust Self- Assembled Monolayers Gi-Hwan Kim,,, F. Pelayo García de Arquer,, Yung Jin Yoon, Xinzheng Lan, Men gxia Liu, Oleksandr
Membrane Structure. Membrane Structure. Membrane Structure. Membranes
 Membrane Structure Membranes Chapter 5 The fluid mosaic model of membrane structure contends that membranes consist of: -phospholipids arranged in a bilayer -globular proteins inserted in the lipid bilayer
Membrane Structure Membranes Chapter 5 The fluid mosaic model of membrane structure contends that membranes consist of: -phospholipids arranged in a bilayer -globular proteins inserted in the lipid bilayer
Ionic channels in Langmuir-Blodgett films imaged by a scanning tunneling microscope
 onic channels in Langmuir-Blodgett films imaged by a scanning tunneling microscope Oleg V. Kolomytkin,* Alexander 0. Golubok,* Dmitri N. Davydov,t Vladimir A. Timofeev,$ Svetlana A. Vinogradova,t and Serge
onic channels in Langmuir-Blodgett films imaged by a scanning tunneling microscope Oleg V. Kolomytkin,* Alexander 0. Golubok,* Dmitri N. Davydov,t Vladimir A. Timofeev,$ Svetlana A. Vinogradova,t and Serge
LIPOSOME STABILITY VERIFICATION BY ATOMIC FORCE MICROSCOPY
 34 Rev.Adv.Mater.Sci. 5 (2003) 34-40 E. F. de Souza and O. Teschke LIPOSOME STABILITY VERIFICATION BY ATOMIC FORCE MICROSCOPY E. F. de Souza 1 and O. Teschke 2 1 Faculdade de Química, CEATEC/PUC-Campinas,
34 Rev.Adv.Mater.Sci. 5 (2003) 34-40 E. F. de Souza and O. Teschke LIPOSOME STABILITY VERIFICATION BY ATOMIC FORCE MICROSCOPY E. F. de Souza 1 and O. Teschke 2 1 Faculdade de Química, CEATEC/PUC-Campinas,
Reading for lecture 6
 Reading for lecture 6 1. Lipids and Lipid Bilayers 2. Membrane Proteins Voet and Voet, Chapter 11 Alberts et al Chapter 6 Jones, R.A.L, Soft Condensed Matter 195pp Oxford University Press, ISBN 0-19-850590-6
Reading for lecture 6 1. Lipids and Lipid Bilayers 2. Membrane Proteins Voet and Voet, Chapter 11 Alberts et al Chapter 6 Jones, R.A.L, Soft Condensed Matter 195pp Oxford University Press, ISBN 0-19-850590-6
Diffusion across cell membrane
 The Cell Membrane and Cellular Transport Diffusion across cell membrane Cell membrane is the boundary between inside & outside separates cell from its environment Can it be an impenetrable boundary? NO!
The Cell Membrane and Cellular Transport Diffusion across cell membrane Cell membrane is the boundary between inside & outside separates cell from its environment Can it be an impenetrable boundary? NO!
CALCIUM CASEINATE. What Is Casein?
 CALCIUM CASEINATE A high quality milk protein that is Calcium Rich, manufactured from fresh pasteurized skimmed milk through precipitation of casein followed by neutralization and natural drying, which
CALCIUM CASEINATE A high quality milk protein that is Calcium Rich, manufactured from fresh pasteurized skimmed milk through precipitation of casein followed by neutralization and natural drying, which
Binding of aromatic anion amphiphile to phospholipid vesicles: a fluorescence study
 Colloids and Surfaces A: Physicochemical and Engineering Aspects 204 (2002) 125 129 www.elsevier.com/locate/colsurfa Binding of aromatic anion amphiphile to phospholipid vesicles: a fluorescence study
Colloids and Surfaces A: Physicochemical and Engineering Aspects 204 (2002) 125 129 www.elsevier.com/locate/colsurfa Binding of aromatic anion amphiphile to phospholipid vesicles: a fluorescence study
The Role of Group Structure in the Action of Some Morpholinium Chloride Derivatives on Model Systems
 Gen. Physiol Biophys (1987), 6, 149 153 149 The Role of Group Structure in the Action of Some Morpholinium Chloride Derivatives on Model Systems A. HENDRICH 1, J. SARAPUK' and S. WITEK. 2 1 Department
Gen. Physiol Biophys (1987), 6, 149 153 149 The Role of Group Structure in the Action of Some Morpholinium Chloride Derivatives on Model Systems A. HENDRICH 1, J. SARAPUK' and S. WITEK. 2 1 Department
RETINOID-PHOSPHOLIPID INTERACTIONS AS STUDIED BY MAGNETIC RESONANCE. Stephen R. Wassail* and William Stillwellt
 Vol.''% No. 3 85 RETINOID-PHOSPHOLIPID INTERACTIONS AS STUDIED BY MAGNETIC RESONANCE Stephen R. Wassail* and William Stillwellt Departments of Physics* and Biology+ Indiana University-Purdue University
Vol.''% No. 3 85 RETINOID-PHOSPHOLIPID INTERACTIONS AS STUDIED BY MAGNETIC RESONANCE Stephen R. Wassail* and William Stillwellt Departments of Physics* and Biology+ Indiana University-Purdue University
The Cell Membrane & Movement of Materials In & Out of Cells PACKET #11
 1 February 26, The Cell Membrane & Movement of Materials In & Out of Cells PACKET #11 Introduction I 2 Biological membranes are phospholipid bilayers with associated proteins. Current data support a fluid
1 February 26, The Cell Membrane & Movement of Materials In & Out of Cells PACKET #11 Introduction I 2 Biological membranes are phospholipid bilayers with associated proteins. Current data support a fluid
Lipids and Membranes
 Lipids and Membranes Presented by Dr. Mohammad Saadeh The requirements for the Pharmaceutical Biochemistry I Philadelphia University Faculty of pharmacy Biological membranes are composed of lipid bilayers
Lipids and Membranes Presented by Dr. Mohammad Saadeh The requirements for the Pharmaceutical Biochemistry I Philadelphia University Faculty of pharmacy Biological membranes are composed of lipid bilayers
Interaction of Functionalized C 60 Nanoparticles with Lipid Membranes
 Interaction of Functionalized C 60 Nanoparticles with Lipid Membranes Kevin Gasperich Advisor: Dmitry Kopelevich ABSTRACT The recent rapid development of applications for fullerenes has led to an increase
Interaction of Functionalized C 60 Nanoparticles with Lipid Membranes Kevin Gasperich Advisor: Dmitry Kopelevich ABSTRACT The recent rapid development of applications for fullerenes has led to an increase
Effects of Epicholesterol on the Phosphatidylcholine Bilayer: A Molecular Simulation Study
 1818 Biophysical Journal Volume 84 March 2003 1818 1826 Effects of Epicholesterol on the Phosphatidylcholine Bilayer: A Molecular Simulation Study Tomasz Róg and Marta Pasenkiewicz-Gierula Department of
1818 Biophysical Journal Volume 84 March 2003 1818 1826 Effects of Epicholesterol on the Phosphatidylcholine Bilayer: A Molecular Simulation Study Tomasz Róg and Marta Pasenkiewicz-Gierula Department of
Supplementary Figures
 Supplementary Figures Supplementary Figure 1. (a) Uncropped version of Fig. 2a. RM indicates that the translation was done in the absence of rough mcirosomes. (b) LepB construct containing the GGPG-L6RL6-
Supplementary Figures Supplementary Figure 1. (a) Uncropped version of Fig. 2a. RM indicates that the translation was done in the absence of rough mcirosomes. (b) LepB construct containing the GGPG-L6RL6-
Supplementary Materials for
 advances.sciencemag.org/cgi/content/full/2/12/e1601838/dc1 Supplementary Materials for General and programmable synthesis of hybrid liposome/metal nanoparticles Jin-Ho Lee, Yonghee Shin, Wooju Lee, Keumrai
advances.sciencemag.org/cgi/content/full/2/12/e1601838/dc1 Supplementary Materials for General and programmable synthesis of hybrid liposome/metal nanoparticles Jin-Ho Lee, Yonghee Shin, Wooju Lee, Keumrai
Lecture Series 4 Cellular Membranes
 Lecture Series 4 Cellular Membranes Reading Assignments Read Chapter 11 Membrane Structure Review Chapter 21 pages 709-717 717 (Animal( Cell Adhesion) Review Chapter 12 Membrane Transport Review Chapter
Lecture Series 4 Cellular Membranes Reading Assignments Read Chapter 11 Membrane Structure Review Chapter 21 pages 709-717 717 (Animal( Cell Adhesion) Review Chapter 12 Membrane Transport Review Chapter
Supplementary Figure 1. EL spectral characteristics. (a)
 Supplementary Figure 1. EL spectral characteristics. (a) Angular dependence of normalized light intensity at 540 nm according to ZnO layers; Lambertian emission pattern (black square), ZnO-F (red circle),
Supplementary Figure 1. EL spectral characteristics. (a) Angular dependence of normalized light intensity at 540 nm according to ZnO layers; Lambertian emission pattern (black square), ZnO-F (red circle),
Cell Membrane Structure (1.3) IB Diploma Biology
 Cell Membrane Structure (1.3) IB Diploma Biology Essential idea: The structure of biological membranes makes them fluid and dynamic http://www.flickr.com/photos/edsweeney/6346198056/ 1.3.1 Phospholipids
Cell Membrane Structure (1.3) IB Diploma Biology Essential idea: The structure of biological membranes makes them fluid and dynamic http://www.flickr.com/photos/edsweeney/6346198056/ 1.3.1 Phospholipids
OXIDATIVE STRESS STUDIES ON LIPID MODEL MEMBRANES
 OXIDATIVE STRESS STUDIES ON LIPID MODEL MEMBRANES MARCELA ELISABETA BARBINTA-PATRASCU *, LAURA TUGULEA * * Faculty of Physics, University of Bucharest, Romania Received December 21, 2004 The liposomes
OXIDATIVE STRESS STUDIES ON LIPID MODEL MEMBRANES MARCELA ELISABETA BARBINTA-PATRASCU *, LAURA TUGULEA * * Faculty of Physics, University of Bucharest, Romania Received December 21, 2004 The liposomes
THE RESPONSE OF THE NEURONAL MEMBRANE TO ACETALDEHYDE TREATMENT
 CELLULAR & MOLECULAR BIOLOGY LETTERS Volume 6, (2001) pp 265 Ł 269 Received 13 May 2001 Accepted 28 May 2001 Short Communication THE RESPONSE OF THE NEURONAL MEMBRANE TO ACETALDEHYDE TREATMENT ANTON LIOPO
CELLULAR & MOLECULAR BIOLOGY LETTERS Volume 6, (2001) pp 265 Ł 269 Received 13 May 2001 Accepted 28 May 2001 Short Communication THE RESPONSE OF THE NEURONAL MEMBRANE TO ACETALDEHYDE TREATMENT ANTON LIOPO
Small angle neutron scattering study of mixed micelles of oppositely charged surfactants
 PRAMANA c Indian Academy of Sciences Vol. 71, No. 5 journal of November 2008 physics pp. 1039 1043 Small angle neutron scattering study of mixed micelles of oppositely charged surfactants J V JOSHI 1,,
PRAMANA c Indian Academy of Sciences Vol. 71, No. 5 journal of November 2008 physics pp. 1039 1043 Small angle neutron scattering study of mixed micelles of oppositely charged surfactants J V JOSHI 1,,
Bilayer Deformation, Pores & Micellation Induced by Oxidized Lipids
 Supporting Information Bilayer Deformation, Pores & Micellation Induced by Oxidized Lipids Phansiri Boonnoy 1, Viwan Jarerattanachat 1,2, Mikko Karttunen 3*, and Jirasak Wongekkabut 1* 1 Department of
Supporting Information Bilayer Deformation, Pores & Micellation Induced by Oxidized Lipids Phansiri Boonnoy 1, Viwan Jarerattanachat 1,2, Mikko Karttunen 3*, and Jirasak Wongekkabut 1* 1 Department of
Membranes. Chapter 5. Membrane Structure
 Membranes Chapter 5 Membrane Structure Lipid Bilayer model: - double phospholipid layer - Gorter & Grendel: 1925 Fluid Mosaic model: consist of -phospholipids arranged in a bilayer -globular proteins inserted
Membranes Chapter 5 Membrane Structure Lipid Bilayer model: - double phospholipid layer - Gorter & Grendel: 1925 Fluid Mosaic model: consist of -phospholipids arranged in a bilayer -globular proteins inserted
Singlet Oxygen Production Photosensitized by Fluorescein in Reversed Micellar Solutions. Norio Miyoshi and Giiti Tomita*
 Singlet Oxygen Production Photosensitized by Fluorescein in Reversed Micellar Solutions Norio Miyoshi and Giiti Tomita* Institute of Biophysics, Faculty of Agriculture, Kyushu University, Fukuoka 812,
Singlet Oxygen Production Photosensitized by Fluorescein in Reversed Micellar Solutions Norio Miyoshi and Giiti Tomita* Institute of Biophysics, Faculty of Agriculture, Kyushu University, Fukuoka 812,
Raman spectroscopy methods for detecting and imaging supported lipid bilayers
 Spectroscopy 24 (2010) 113 117 113 DOI 10.3233/SPE-2010-0426 IOS Press Raman spectroscopy methods for detecting and imaging supported lipid bilayers Claire S. Sweetenham and Ioan Notingher Nanoscience
Spectroscopy 24 (2010) 113 117 113 DOI 10.3233/SPE-2010-0426 IOS Press Raman spectroscopy methods for detecting and imaging supported lipid bilayers Claire S. Sweetenham and Ioan Notingher Nanoscience
The Cell Membrane & Movement of Materials In & Out of Cells PACKET #11
 1 The Cell Membrane & Movement of Materials In & Out of Cells PACKET #11 Introduction I 2 Biological membranes are phospholipid bilayers with associated proteins. Current data support a fluid mosaic model
1 The Cell Membrane & Movement of Materials In & Out of Cells PACKET #11 Introduction I 2 Biological membranes are phospholipid bilayers with associated proteins. Current data support a fluid mosaic model
Structure-Transport Relationship in Organized Soft Matter Systems by Diffusion NMR. Sergey Vasenkov
 Structure-Transport Relationship in Organized Soft Matter Systems by Diffusion NMR Sergey Vasenkov Outline Combining advantages of high field and high gradients in diffusion NMR Relationship between diffusivities
Structure-Transport Relationship in Organized Soft Matter Systems by Diffusion NMR Sergey Vasenkov Outline Combining advantages of high field and high gradients in diffusion NMR Relationship between diffusivities
Biological Membranes. Lipid Membranes. Bilayer Permeability. Common Features of Biological Membranes. A highly selective permeability barrier
 Biological Membranes Structure Function Composition Physicochemical properties Self-assembly Molecular models Lipid Membranes Receptors, detecting the signals from outside: Light Odorant Taste Chemicals
Biological Membranes Structure Function Composition Physicochemical properties Self-assembly Molecular models Lipid Membranes Receptors, detecting the signals from outside: Light Odorant Taste Chemicals
Membranes. Chapter 5
 Membranes Chapter 5 Membrane Structure The fluid mosaic model of membrane structure contends that membranes consist of: -phospholipids arranged in a bilayer -globular proteins inserted in the lipid bilayer
Membranes Chapter 5 Membrane Structure The fluid mosaic model of membrane structure contends that membranes consist of: -phospholipids arranged in a bilayer -globular proteins inserted in the lipid bilayer
Paper 4. Biomolecules and their interactions Module 22: Aggregates of lipids: micelles, liposomes and their applications OBJECTIVE
 Paper 4. Biomolecules and their interactions Module 22: Aggregates of lipids: micelles, liposomes and their applications OBJECTIVE The main aim of this module is to introduce the students to the types
Paper 4. Biomolecules and their interactions Module 22: Aggregates of lipids: micelles, liposomes and their applications OBJECTIVE The main aim of this module is to introduce the students to the types
Cellular membranes are fluid mosaics of lipids and proteins.
 Study Guide e Plasma Membrane You should be able to write out the definitions to each of the following terms in your own words: plasma membrane fluid mosaic integral proteins peripheral proteins receptor
Study Guide e Plasma Membrane You should be able to write out the definitions to each of the following terms in your own words: plasma membrane fluid mosaic integral proteins peripheral proteins receptor
Raman spectroscopy of chocolate bloom
 Raman spectroscopy of chocolate bloom Siyu He, and Dmitri V. Voronine,3 Texas A&M University, College Station, TX 77843, USA Xi an Jiaotong University, Xi an 70049, China 3 Baylor University, Waco, TX
Raman spectroscopy of chocolate bloom Siyu He, and Dmitri V. Voronine,3 Texas A&M University, College Station, TX 77843, USA Xi an Jiaotong University, Xi an 70049, China 3 Baylor University, Waco, TX
EFFECT OF SOLVENTS ON PARTICLE STRUCTURE, MORPHOLOGY AND OPTICAL PROPERTIES OF ZINC OXIDE NANOPARTICLES
 EFFECT OF SOLVENTS ON PARTICLE STRUCTURE, MORPHOLOGY AND OPTICAL PROPERTIES OF ZINC OXIDE NANOPARTICLES A.Vanaja 1 and K.Srinivasa Rao 2 1 Department of Physics, Lingayya s University, Old Faridabad, Haryana,
EFFECT OF SOLVENTS ON PARTICLE STRUCTURE, MORPHOLOGY AND OPTICAL PROPERTIES OF ZINC OXIDE NANOPARTICLES A.Vanaja 1 and K.Srinivasa Rao 2 1 Department of Physics, Lingayya s University, Old Faridabad, Haryana,
Custom-Made Products / Scientific Research Instruments
 Synchrotron Radiation Instruments Double Crystal Monochromator A double crystal monochromator is an instrument that extracts light of a specific wavelength using crystal diffraction. Light of various wavelengths
Synchrotron Radiation Instruments Double Crystal Monochromator A double crystal monochromator is an instrument that extracts light of a specific wavelength using crystal diffraction. Light of various wavelengths
Fall Name Student ID
 Name Student ID PART 1: Matching. Match the organelle to its function (11 points) 1.Proton motive force 2. Fluid Mosiac 3. Oxidative Phosphorylation 4. Pyruvate dehydrogenase 5. Electrochemical Force 6.
Name Student ID PART 1: Matching. Match the organelle to its function (11 points) 1.Proton motive force 2. Fluid Mosiac 3. Oxidative Phosphorylation 4. Pyruvate dehydrogenase 5. Electrochemical Force 6.
Lecture Series 5 Cellular Membranes
 Lecture Series 5 Cellular Membranes Cellular Membranes A. Membrane Composition and Structure B. Animal Cell Adhesion C. Passive Processes of Membrane Transport D. Active Transport E. Endocytosis and Exocytosis
Lecture Series 5 Cellular Membranes Cellular Membranes A. Membrane Composition and Structure B. Animal Cell Adhesion C. Passive Processes of Membrane Transport D. Active Transport E. Endocytosis and Exocytosis
Will s Pre-Test. (4) A collection of cells that work together to perform a function is termed a(n): a) Organelle b) Organ c) Cell d) Tissue e) Prison
 Will s Pre-Test This is a representative of Exam I that you will take Tuesday September 18, 2007. The actual exam will be 50 multiple choice questions. (1) The basic structural and functional unit of the
Will s Pre-Test This is a representative of Exam I that you will take Tuesday September 18, 2007. The actual exam will be 50 multiple choice questions. (1) The basic structural and functional unit of the
A. Membrane Composition and Structure. B. Animal Cell Adhesion. C. Passive Processes of Membrane Transport. D. Active Transport
 Cellular Membranes A. Membrane Composition and Structure Lecture Series 5 Cellular Membranes B. Animal Cell Adhesion E. Endocytosis and Exocytosis A. Membrane Composition and Structure The Fluid Mosaic
Cellular Membranes A. Membrane Composition and Structure Lecture Series 5 Cellular Membranes B. Animal Cell Adhesion E. Endocytosis and Exocytosis A. Membrane Composition and Structure The Fluid Mosaic
Membranes 9/15/2016. Phospholipids. Phospholipid bilayer
 Membranes Phospholipids Type of complex lipid that forms biological membranes. Have a polar hydrophilic head and two nonpolar hydrophobic tails. Amphipathic. This causes the tails to cluster together in
Membranes Phospholipids Type of complex lipid that forms biological membranes. Have a polar hydrophilic head and two nonpolar hydrophobic tails. Amphipathic. This causes the tails to cluster together in
Chapter CHAPTER 7. ELECTRICAL PROPERTIES OF ZnO DOPED MAGESIUM ALUMIUM SILICATE GLASS-CERAMICS
 Chapter 7 102 CHAPTER 7 ELECTRICAL PROPERTIES OF ZnO DOPED MAGESIUM ALUMIUM SILICATE GLASS-CERAMICS Chapter 7 103 CHAPTER 7 ELECTRICAL PROPERTIES OF ZnO DOPED MAGNESIUM ALUMINUM SILICATE GLASS-CERAMICS
Chapter 7 102 CHAPTER 7 ELECTRICAL PROPERTIES OF ZnO DOPED MAGESIUM ALUMIUM SILICATE GLASS-CERAMICS Chapter 7 103 CHAPTER 7 ELECTRICAL PROPERTIES OF ZnO DOPED MAGNESIUM ALUMINUM SILICATE GLASS-CERAMICS
Cellular Neurophysiology I Membranes and Ion Channels
 Cellular Neurophysiology I Membranes and Ion Channels Reading: BCP Chapter 3 www.bioelectriclab All living cells maintain an electrical potential (voltage) across their membranes (V m ). Resting Potential
Cellular Neurophysiology I Membranes and Ion Channels Reading: BCP Chapter 3 www.bioelectriclab All living cells maintain an electrical potential (voltage) across their membranes (V m ). Resting Potential
The main biological functions of the many varied types of lipids include: energy storage protection insulation regulation of physiological processes
 Big Idea In the biological sciences, a dehydration synthesis (condensation reaction) is typically defined as a chemical reaction that involves the loss of water from the reacting molecules. This reaction
Big Idea In the biological sciences, a dehydration synthesis (condensation reaction) is typically defined as a chemical reaction that involves the loss of water from the reacting molecules. This reaction
Biology 5357: Membranes
 s 5357 Biology 5357: s Assembly and Thermodynamics of Soft Matter Paul H. MD, PhD Department of Cell Biology and Physiology pschlesinger@.wustl.edu 362-2223 Characteristics s 5357 s are polymorphic s 5357
s 5357 Biology 5357: s Assembly and Thermodynamics of Soft Matter Paul H. MD, PhD Department of Cell Biology and Physiology pschlesinger@.wustl.edu 362-2223 Characteristics s 5357 s are polymorphic s 5357
ก Chirality Control on Lipid Nanotubule Morphology Investigated by Circular Dichroism 1, 1, 2, 2,,
 ก Chirality Control on Lipid Nanotubule Morphology Investigated by Circular Dichroism 1, 1, 2, 2,, ก 3, 1 Yuwathida Jantippana 1, Weerawat Intaratat 1, Wisit Singhsomroje 2, Sujint Wangsuya 2, Piboon Pantu
ก Chirality Control on Lipid Nanotubule Morphology Investigated by Circular Dichroism 1, 1, 2, 2,, ก 3, 1 Yuwathida Jantippana 1, Weerawat Intaratat 1, Wisit Singhsomroje 2, Sujint Wangsuya 2, Piboon Pantu
Cell Membrane (Transport) Notes
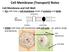 Cell Membrane (Transport) Notes Cell Membrane and Cell Wall: ALL cells have a cell membrane made of proteins and lipids protein channel Cell Membrane Layer 1 Layer 2 lipid bilayer protein pump SOME cells
Cell Membrane (Transport) Notes Cell Membrane and Cell Wall: ALL cells have a cell membrane made of proteins and lipids protein channel Cell Membrane Layer 1 Layer 2 lipid bilayer protein pump SOME cells
