Permeation of organic molecules in water and ethanol-water solutions by reverse osmosis
|
|
|
- Samson Johnson
- 6 years ago
- Views:
Transcription
1 Proceedings of European Congress of Chemical Engineering (ECCE-6) Copenhagen, September 2007 Permeation of organic molecules in water and ethanol-water solutions by reverse osmosis J. Labanda, J. Llorens * Department of Chemical Engineering, University of Barcelona, Martí i Franquès 1, Barcelona, Spain jllorensl@ub.edu Abstract The permeation of different organic molecules in water solvent and in 12% ethanolwater solution by hydrophilic reverse osmosis membrane was studied. The membrane was conditioned to the ethanol-water solution before the experiments. Initially, the membrane was characterized by water and ethanol-water solutions. The permeation of four solutes (tyramine, proline, glucose and tartaric acid) showed that the solute rejections increased with permeate flux and they were than 90% in water solvent, and they were decreased significantly in presence of ethanol. Keywords: reverse osmosis, ethanol-water mixtures, solute rejection, organic molecules, Spiegler-Kendem model. 1. Introduction Most of the established reverse osmosis and nanofiltration membrane processes are related to the treatment of aqueous systems, or single organic solvent solution. The use of these process for systems of solvent mixture especially mixture of water and organic solvent is considerably sparse. Furthermore, most of the research is being done with relatively low concentration, and a few results have been reported of solution with high concentration of organic solute in the mixture of organic solvent and water. Koops et al. (2001) and Yang et al. (2001) showed that rejections of the same solute are significantly different for a given membrane when measured in solvents with different polarities. The implication of this finding is that the molecular weight cut-off (MWCO) or pore size data provided by membrane manufacturers (which are usually measured in aqueous environments) may not be valid in organic solvents. There is little agreement in the literature on the mechanisms of solute solvent membrane interactions in organic solvents.
2 J. Labanda et al. There is usually a difference in solute rejection and flux in organic solvents when compared with performance in aqueous solutions (Machado et al., 1999). In most cases, rejection of the same molecule in an organic solvent is significantly lower than in aqueous solution for the same membrane and operating conditions (Su et al., 2005). Membrane manufacturers usually supply membranes semi-dry or wet with water. It is important to condition the membranes before to use with organic solvents. Conditioning involves rinsing the membrane and permeating water or the solvent under the appropriate pressure to ensure removal of preservatives and humectants on the membrane surface and from within the pores. The membrane should then be exposed to solvents with gradually changing polarities in sequence, ending with a solvent of a polarity matching that of the membrane. This solvent exchange process was first described by van Oss (1970). Membrane conditioning ensures complete wetting of membrane by solvent which facilitates solvent permeation and improves its performance. The method of conditioning has a strong effect on flux, membrane integrity and pressure rating of polymeric membranes (Shukla et al., 2002). Swelling and deformation of the membranes when exposed to organic solvents is common and is dealt with by conditioning the membranes through gradual solvent change (Shukla et al., 2003). Once conditioned, the membrane s performance may be significantly different from that in an aqueous system and may have to be described for each individual solutesolvent-membrane system. The permeability is influenced by these factors as well as molecular size and hydrophobicity. The behavior of the membranes in organic solvents has been described using several different models with varying degrees of success, taking into account characteristics of the solvent such as viscosity, molar volume, and surface tension as well as characteristics of the membranes and solutes (Bhanushali et al., 2001; Van der Bruggen et al., 1999). In this study, the influence of feed pressure, feed concentration on the permeate flux and rejection of organic molecules have been analyzed in water solvent and in 12% (v/v) ethanol in ethanol-water mixture through low pressure reverse osmosis membrane. 2. Theoretical background Transport of a binary solution through reverse osmosis membranes can be described by the phenomenological model of Kedem and Katchalsky, which is based on nonequilibrium thermodynamics: (1) The solvent flux through a membrane, J v, as a function of differences in applied pressure between both sides of membrane, P, is given by the following expression: ( σ π ) Jv = Lp P (1) where L p is the solvent permeability, π is the difference in osmotic pressure between both sizes of membrane and σ is the reflection coefficient, which is a measure of the 2
3 degree of the solute permeation through the membrane and corresponds to the maximum solute rejection. (2) The steady state transport of a solute, J s, in a pressure-driven process is described by diffusive and convective transport as: J s ( 1 ) J C = w π + σ (2) v where w is the solute permeability, and C represents the average of the solute concentration between both sides of the membrane and usually is calculated as a logarithm mean. Following this equation, the solute permeation is a function of diffusion and convection flow. As applied pressure increases, the contribution of convection increases and the solute rejection approaches to the reflection coefficient. In contrast, the solute permeability corresponds to purely diffusive transport when convection flux is negligible (solvent flux tends to zero). Spiegler-Kendem modified the model of Kendem and Katchalsky by a differential form of Equation (2). The Spiegler-Kendem model takes into account the concentration profile inside the membrane. (3) The solute rejection, which is defined by the permeate and retention concentrations (C p and C r, respectively) as R = 1 C p Cr, for an ideal solution was deduced by Spiegler-Kendem by integration the differential form of solute transport equation and was expressed as a function of reflection coefficient with the following equations: ( 1 F) σ R = (3) 1 σ F with F = exp ( 1 σ ) J R T w v (4) Then, the separation process in reverse osmosis is performed by the determination of L p, w and σ. 3. Experimental The permeation of four organic solutes was analyzed in water and ethanol-water solutions: tyramine, proline, glucose and tartaric acid. The ethanol concentration in the ethanol-water solutions was fixed to 12%. The experimental set-up for membrane testing consisted of a thermostatic (25 ± 0.5 ºC) 2 L feed vessel, a positive displacement pump equipped with a closed-pipe pressure dampener to prevent pressure oscillations, supplied by CAT-PUMPS (Kontich, Belgium), a membrane cell for a flat sheet polymeric membrane (Sepa CF membrane cell), supplied by GE Infrastructure Water & Process Technology, pressure gauges to measure the inlet and outlet pressures in the membrane cells and flow meters for retentate and permeate flux rate measurements. 3
4 J. Labanda et al. The reverse osmosis membrane was supplied by Toray (ref. UTC70). This flat sheet (155 cm 2 ) polysulfone-polyamide nanofiltration membrane has salt rejection of 97.2%. The membrane system was operated in steady state mode, in which both retentate and permeate streams are recycled to the feed vessel. The time needed to reach the steady state was 60 minutes. The performance of the process was determined by measuring the solute concentrations in the retentate and permeate streams, the transmembrane pressure and the resulting permeate flux rate. The membrane was conditioned to ethanol solvent. The membrane was initially soaked in 12% (v/) aqueous ethanol overnight at room temperature and atmospheric pressure. The membrane was then placed in the cell and the flux of 12% ethanol measured as a function of pressure. This procedure was repeated until the flux was constant. 4. Results and discussions The solvent permeate flux depends on not only operational conditions (i.e., transmembrane pressure, temperature) and membrane properties, but also interaction between solution and membrane, which are determined by solution and membrane properties. Figure 1 shows the water and ethanol-water permeate fluxes as a function of transmembrane pressure and ethanol volume fraction. Linear relationships with transmembrane pressure were obtained for both solutions. The ethanol-water fluxes were much lower than those of the water. The experimental data was well correlated by Equation (1), and the fitted and calculated values of Splieger-Kendem model are shown in Table I. L p and σ π depended on ethanol volume fraction, while σ π increased, L p decreased with ethanol volume fraction. As water has the highest affinity for hydrophilic membranes, the addition of ethanol has a stronger effect on the solvent permeability. Therefore, ethanol-water mixtures have lower permeabilities than water ones. This fact could be due to the polarity differences and surface phenomena (i.e., swelling), since the presence of a relative low alcohol concentrations decreases the mixture polarity, leading to higher resistance at the membrane-solvent interphase. As a result, ethanol has lower affinity for hydrophilic membranes. Jv [ L h -1 m -2 ] % ethanol 12% ethanol P [ bar ] Figure 1. Evolution of solvent permeate flux (open symbols) and ethanol rejection (filled symbols) as a function of transmembrane pressure for the two solvent solutions. 100% 90% 80% 70% 60% 50% 40% 30% 20% 10% 0% Ethanol rejection 4
5 Table I. Solvent permeability and σ π as a function of ethanol concentration. Ethanol concentration L p (L h -1 m -2 bar -1 ) σ π r 2 0% % In general, the retention of organic species in water solvent can be considered very high for reverse osmosis. Figure 2 shows the solute rejections as a function of permeate flux for both solvents. The solute rejection increased with permeate flux tending to the reflection coefficient of the solute, σ. The Spiegler-Kendem model was well correlated with the experimental data for the best values of solute permeability, w, and reflection coefficient, σ, following Equations (3) and (4). The best fitting curves are illustrated in Figure 2 as solid lines and the results are shown in Table II. The solute rejections were higher than 90% in water solvent, and they were decreased significantly in presence of ethanol. The higher rejection in water solvent than in ethanol-water mixture could suggest that the complexation of water molecules with the solute molecules makes their effective size larger in water solvent than in ethanolwater mixture. Rejections were approximately the same for all solutes because of the comparable molecular size, if molecular size is taken as a factor determining the separation, without taking any effect of the solvent into account. Solute permeability, showed in Table II, depended on solute type and increased in presence of ethanol. This fact could be due to the reduction of solvent polarity. Therefore, the decrease in solute rejection with ethanol-water mixture is due to the reduction of solvent flux although solvent permeability increased. Mechanisms and mathematical models for retention of organic solutes by reverse osmosis have been well developed for water solvent, while transport and retention mechanisms in presence of organic solvents need further investigation for the determination of the solute-solvent-membrane interactions. Solute rejection 100% 80% 60% 40% 20% Tyramine Proline Glucose Tartaric acid Solute rejection 100% 80% 60% 40% 20% Tyramine Proline Glucose Tartaric acid (a) 0% Jv ( L m -2 h -1 ) 0% Jv ( L m -2 h -1 ) Figure 2. Solute rejections as a function of solvent fluxes: (a) water solvent and (b) 12% ethanol in ethanol-water mixture. (b) 5
6 J. Labanda et al. Table II. Reflection coefficient and solute permeability in different solvent. Solvent Solute σ (-) w (mol h -1 m -2 bar -1 ) Tyramine Proline Water Glucose Tartaric acid Tyramine Proline Glucose Ethanol-water (12% ethanol) Tartaric acid Conclusions In this study, it has been studied the permeation of four organic solutes in water solvent and in ethanol-water mixture with an ethanol volume fraction of 12%. The permeate flux of the ethanol-water mixture was lower than that of the pure water for the hydrophilic membrane and the result was theoretically analyzed. The solute rejections increased with permeate flux and they were than 90% in water solvent, and they were decreased significantly in presence of ethanol. The experimental data was well correlated by the Spiegler-Kendem model. Acknowledgements The authors are grateful to the Spanish Ministerio de Ciencia y Tecnología (project CTQ C02-01/PPQ) for funds received to carry out this study. References Bhanushali, D., Kloos, S., Kurth, C., and Bhattacharyya, D. (2001) Journal of Membrane Science, 189, Koops, G.H., Yamada, S. and Nakao, S.I., (2001) Journal of Membrane Science, 189, Machado, D.R., Hasson, D. and Semita, R., (1999) Journal of Membrane Science, 163, Shukla, R. and Cheryan, M., (2002) Journal of Membrane Science, 198,
7 Van der Bruggen, B., Schaep, J., Wilms, D., and Vandecasteele, C. (1999) Journal Membrane Science, 156, Shukla, R. and Cheryan, M., (2003) Separation Science and Technology, 38, Su, B., Wang Z., Wang, J. and Wang, S., (2005) Journal of Membrane Science, 251, van Oss, C.J., Progress in Separation and Purification, vol. 3, Wiley/ Interscience, New York (1970). Yang, X.J., Livingston, A.G. and Freitas dos Santos, L., (2001) Journal of Membrane Science, 190,
Control of irreversible fouling by application of dynamic membranes
 Desalination 192 (2006) 63 67 Control of irreversible fouling by application of dynamic membranes M.T. Pessoa de Amorim*, Ilia Rosa Afonso Ramos Department of Textile Engineering, University of Minho,
Desalination 192 (2006) 63 67 Control of irreversible fouling by application of dynamic membranes M.T. Pessoa de Amorim*, Ilia Rosa Afonso Ramos Department of Textile Engineering, University of Minho,
Introduction to Membrane
 Heinrich Strathmann Introduction to Membrane Science and Technology WILEY- VCH WILEY-VCH Verlag GmbH & Co. KG aa V Preface Symbols XIII XV 1 Introduction 2 1.1 Overview of Membrane Science and Technology
Heinrich Strathmann Introduction to Membrane Science and Technology WILEY- VCH WILEY-VCH Verlag GmbH & Co. KG aa V Preface Symbols XIII XV 1 Introduction 2 1.1 Overview of Membrane Science and Technology
Passive Transport. Does not expend cellular energy for the movement to take place. Ex-rolling down a hill
 Passive Transport Fluid Mosaic Model Passive Transport Does not expend cellular energy for the movement to take place Ex-rolling down a hill Parts of a Solution Solute: what gets dissolved Solvent: What
Passive Transport Fluid Mosaic Model Passive Transport Does not expend cellular energy for the movement to take place Ex-rolling down a hill Parts of a Solution Solute: what gets dissolved Solvent: What
POLY(DIMETHYL SILOXANE) BASED COMPOSITE NANOFILTRATION MEMBRANES FOR NON-AQUEOUS APPLICATIONS
 POLY(DIMETHYL SILOXANE) BASED COMPOSITE NANOFILTRATION MEMBRANES FOR NON-AQUEOUS APPLICATIONS DISSERTATION To obtain The doctor s degree at the University of Twente, On the authority of the rector magnificus,
POLY(DIMETHYL SILOXANE) BASED COMPOSITE NANOFILTRATION MEMBRANES FOR NON-AQUEOUS APPLICATIONS DISSERTATION To obtain The doctor s degree at the University of Twente, On the authority of the rector magnificus,
EXPERIMENTAL INVESTIGATION ON REJECTION OF SODIUM SULFATE BY REVERSE OSMOSIS MEMBRANES
 EXPERIMENTAL INVESTIGATION ON REJECTION OF SODIUM SULFATE BY REVERSE OSMOSIS MEMBRANES By DIAN TANUWIDJAJA UNIVERSITY OF CALIFORNIA, LOS ANGELES 2002 EXPERIMENTAL INVESTIGATION ON REJECTION OF SODIUM SULFATE
EXPERIMENTAL INVESTIGATION ON REJECTION OF SODIUM SULFATE BY REVERSE OSMOSIS MEMBRANES By DIAN TANUWIDJAJA UNIVERSITY OF CALIFORNIA, LOS ANGELES 2002 EXPERIMENTAL INVESTIGATION ON REJECTION OF SODIUM SULFATE
Mass transfer of ionic species in direct and reverse osmosis processes
 University of South Florida Scholar Commons Graduate Theses and Dissertations Graduate School 2003 Mass transfer of ionic species in direct and reverse osmosis processes Silvana Melania Stefania Ghiu University
University of South Florida Scholar Commons Graduate Theses and Dissertations Graduate School 2003 Mass transfer of ionic species in direct and reverse osmosis processes Silvana Melania Stefania Ghiu University
Effect of Rejection Performance on Hollow Fiber Composite Reverse Osmosis Membrane by Alcohols Additives
 Effect of Rejection Performance on Hollow Fiber Composite Reverse Osmosis Membrane by Alcohols Additives Xinghua Wang & Wei Wang Key Laboratory of Hollow Fiber Membrane Material and Membrane Process Tianjin
Effect of Rejection Performance on Hollow Fiber Composite Reverse Osmosis Membrane by Alcohols Additives Xinghua Wang & Wei Wang Key Laboratory of Hollow Fiber Membrane Material and Membrane Process Tianjin
MMS AG Membrane Systems Milk Fractionation
 MMS AG Milk Fractionation George Bou-Habib MMS - Company background Founded in June 1995 Based in Zürich, Switzerland. 30 Employees In-house process development, engineering and construction. Industrial
MMS AG Milk Fractionation George Bou-Habib MMS - Company background Founded in June 1995 Based in Zürich, Switzerland. 30 Employees In-house process development, engineering and construction. Industrial
μ i = chemical potential of species i C i = concentration of species I
 BIOE 459/559: Cell Engineering Membrane Permeability eferences: Water Movement Through ipid Bilayers, Pores and Plasma Membranes. Theory and eality, Alan Finkelstein, 1987 Membrane Permeability, 100 Years
BIOE 459/559: Cell Engineering Membrane Permeability eferences: Water Movement Through ipid Bilayers, Pores and Plasma Membranes. Theory and eality, Alan Finkelstein, 1987 Membrane Permeability, 100 Years
Membrane filtration in water recycling: removal of natural hormones
 Membrane filtration in water recycling: removal of natural hormones L.D. Nghiem,.I. Schäfer and T.D.Waite Centre for Water and Waste Technology, School of Civil and Environmental Engineering, The University
Membrane filtration in water recycling: removal of natural hormones L.D. Nghiem,.I. Schäfer and T.D.Waite Centre for Water and Waste Technology, School of Civil and Environmental Engineering, The University
FOULING RESISTANT REVERSE OSMOSIS MEMBRANES. Ja-young Koo, Sung Pyo Hong, Hoon Hyung, Young Hun Kim, Sungro Yoon, Soon Sik Kim
 FOULING RESISTANT REVERSE OSMOSIS MEMBRANES Ja-young Koo, Sung Pyo Hong, Hoon Hyung, Young Hun Kim, Sungro Yoon, Soon Sik Kim Filter Business Division Saehan Industries, Inc, 1 Jungsandong, Kyungsan city,
FOULING RESISTANT REVERSE OSMOSIS MEMBRANES Ja-young Koo, Sung Pyo Hong, Hoon Hyung, Young Hun Kim, Sungro Yoon, Soon Sik Kim Filter Business Division Saehan Industries, Inc, 1 Jungsandong, Kyungsan city,
Separation of d-limonene from supercritical CO 2 by means of membranes
 J. of Supercritical Fluids 34 (2005) 143 147 Separation of d-limonene from supercritical CO 2 by means of membranes Luiz Henrique Castelan Carlson, Ariovaldo Bolzan, Ricardo Antônio Francisco Machado Universidade
J. of Supercritical Fluids 34 (2005) 143 147 Separation of d-limonene from supercritical CO 2 by means of membranes Luiz Henrique Castelan Carlson, Ariovaldo Bolzan, Ricardo Antônio Francisco Machado Universidade
The Study of Dynamic Milk Ultrafiltration Performance Influenced by Membrane Molecular Weight Cut off
 Iran. J. Chem. Chem. Eng. Vol. 26, No.1, 27 The Study of Dynamic Milk Ultrafiltration Performance Influenced by Membrane Molecular Weight Cut off azavi, Sayed Mohammad Ali* + Department of Food Science
Iran. J. Chem. Chem. Eng. Vol. 26, No.1, 27 The Study of Dynamic Milk Ultrafiltration Performance Influenced by Membrane Molecular Weight Cut off azavi, Sayed Mohammad Ali* + Department of Food Science
Biology. Membranes.
 1 Biology Membranes 2015 10 28 www.njctl.org 2 Vocabulary active transport carrier protein channel protein concentration gradient diffusion enzymatic activity facilitated diffusion fluid mosaic hypertonic
1 Biology Membranes 2015 10 28 www.njctl.org 2 Vocabulary active transport carrier protein channel protein concentration gradient diffusion enzymatic activity facilitated diffusion fluid mosaic hypertonic
WEDNESDAY 10/18/17. Why is the cell/plasma membrane important? What is the cell/plasma membrane made of? Label the cell membrane on your notes.
 WEDNESDAY 10/18/17 Why is the cell/plasma membrane important? What is the cell/plasma membrane made of? Label the cell membrane on your notes. THE PLASMA MEMBRANE - 2 Gateway to Cell HOMEOSTASIS Balanced
WEDNESDAY 10/18/17 Why is the cell/plasma membrane important? What is the cell/plasma membrane made of? Label the cell membrane on your notes. THE PLASMA MEMBRANE - 2 Gateway to Cell HOMEOSTASIS Balanced
109 IWA Publishing 2002 Journal of Water Supply: Research and Technology AQUA
 109 IWA Publishing 2002 Journal of Water Supply: Research and Technology AQUA 51.2 2002 Effects of molecular weight cutoff, f/k ratio (a hydrodynamic condition), and hydrophobic interactions on natural
109 IWA Publishing 2002 Journal of Water Supply: Research and Technology AQUA 51.2 2002 Effects of molecular weight cutoff, f/k ratio (a hydrodynamic condition), and hydrophobic interactions on natural
Journal of Membrane Science & Research
 Journal of Membrane Science and Research 3 (2017) 102-108 Journal of Membrane Science & Research journal homepage: www.msrjournal.com Research Paper Glycerin Removal from Ultrafiltration Flat Sheet Membranes
Journal of Membrane Science and Research 3 (2017) 102-108 Journal of Membrane Science & Research journal homepage: www.msrjournal.com Research Paper Glycerin Removal from Ultrafiltration Flat Sheet Membranes
REACTIONS IN SUPERCRITICAL CARBON DIOXIDE EFFICIENT PRODUCT FRACTIONATION FOLLOWING ENZYMATIC AROMA SYNTHESIS
 REACTIONS IN SUPERCRITICAL CARBON DIOXIDE EFFICIENT PRODUCT FRACTIONATION FOLLOWING ENZYMATIC AROMA SYNTHESIS T.Gamse *, G.Kracker-Semler, R.Marr Institute of Thermal Process and Environmental Engineering
REACTIONS IN SUPERCRITICAL CARBON DIOXIDE EFFICIENT PRODUCT FRACTIONATION FOLLOWING ENZYMATIC AROMA SYNTHESIS T.Gamse *, G.Kracker-Semler, R.Marr Institute of Thermal Process and Environmental Engineering
TABLE OF CONTENT CHAPTER CONTENT PAGE
 vii TABLE OF CONTENT CHAPTER CONTENT PAGE TITLE PAGE DECLARATION DEDICATION ACKNOWLEDGEMENT ABSTRACT ABSTRAK TABLE OF CONTENT LIST OF TABLES LIST OF FIGURES NOMENCLATURE LIST OF APPENDICES i ii iii iv
vii TABLE OF CONTENT CHAPTER CONTENT PAGE TITLE PAGE DECLARATION DEDICATION ACKNOWLEDGEMENT ABSTRACT ABSTRAK TABLE OF CONTENT LIST OF TABLES LIST OF FIGURES NOMENCLATURE LIST OF APPENDICES i ii iii iv
Separating xylose from glucose using spiral wound nanofiltration membrane: Effect of cross-flow parameters on sugar rejection
 IOP Conference Series: Materials Science and Engineering PAPER OPEN ACCESS Separating xylose from glucose using spiral wound nanofiltration membrane: Effect of cross-flow parameters on sugar rejection
IOP Conference Series: Materials Science and Engineering PAPER OPEN ACCESS Separating xylose from glucose using spiral wound nanofiltration membrane: Effect of cross-flow parameters on sugar rejection
TRANSPORT OF PHARMACEUTICAL AND NOM
 TRANSPORT OF PHARMACEUTICAL AND NOM IN NF AND TIGHT UF MEMBRANES Gun-Young Park *, Jaeweon Cho * Department of environmental science and engineering GIST, 1 Oryong-dong, Buk-gu, Gwangju 500-712, Korea
TRANSPORT OF PHARMACEUTICAL AND NOM IN NF AND TIGHT UF MEMBRANES Gun-Young Park *, Jaeweon Cho * Department of environmental science and engineering GIST, 1 Oryong-dong, Buk-gu, Gwangju 500-712, Korea
Introduction of Tangential Flow Filtration (TFF) Karen Chan 16 May 2017
 Introduction of Tangential Flow Filtration (TFF) Karen Chan 16 May 2017 What you will learn TFF principles and applications in mammalian cell processes TFF vocabulary definitions and key process parameters
Introduction of Tangential Flow Filtration (TFF) Karen Chan 16 May 2017 What you will learn TFF principles and applications in mammalian cell processes TFF vocabulary definitions and key process parameters
AGENDA for 01/09/14 AGENDA: HOMEWORK: Due end of period OBJECTIVES:
 AGENDA for 01/09/14 AGENDA: 1. 2.3.2: Diabetic Emergency! Blood Glucose Effects on Simulated Cellular Models Egg Demo Day 3 OBJECTIVES: 1. Design an experiment to simulate osmosis in body cells 2. Relate
AGENDA for 01/09/14 AGENDA: 1. 2.3.2: Diabetic Emergency! Blood Glucose Effects on Simulated Cellular Models Egg Demo Day 3 OBJECTIVES: 1. Design an experiment to simulate osmosis in body cells 2. Relate
Material balance calculations involved with dilution and mixing
 Material balance calculations involved with dilution and mixing 1 Dilution of NaCl Water, (W) 15 kg of NaCl NaCl, 20% Dilution NaCl sol., (S) 2 Volume changes on mixing When two liquids are mixed, the
Material balance calculations involved with dilution and mixing 1 Dilution of NaCl Water, (W) 15 kg of NaCl NaCl, 20% Dilution NaCl sol., (S) 2 Volume changes on mixing When two liquids are mixed, the
LAB 04 Diffusion and Osmosis
 LAB 04 Diffusion and Osmosis Objectives: Describe the physical mechanisms of diffusion and osmosis. Understand the relationship between surface area and rate of diffusion. Describe how molar concentration
LAB 04 Diffusion and Osmosis Objectives: Describe the physical mechanisms of diffusion and osmosis. Understand the relationship between surface area and rate of diffusion. Describe how molar concentration
Explain how the structure of the plasma membrane allows material to move through. Explain the processes of Passive Transport and Active Transport.
 Objectives: Explain how the structure of the plasma membrane allows material to move through. Explain the processes of Passive Transport and Active Transport. Predict whether active or passive transport
Objectives: Explain how the structure of the plasma membrane allows material to move through. Explain the processes of Passive Transport and Active Transport. Predict whether active or passive transport
membrane from 3,5-diaminobenzoic acid
 ORIGINAL ARTICLE Preparation and characterization of interfacial polymerised membrane from 3,5-diaminobenzoic acid Abdul Latif Ahmad 1, Boon Seng Ooi 2, and Jyoti Prosad Choudhury 3 Abstract Ahmad, A.L.,
ORIGINAL ARTICLE Preparation and characterization of interfacial polymerised membrane from 3,5-diaminobenzoic acid Abdul Latif Ahmad 1, Boon Seng Ooi 2, and Jyoti Prosad Choudhury 3 Abstract Ahmad, A.L.,
Cell Membrane (Transport) Notes
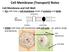 Cell Membrane (Transport) Notes Cell Membrane and Cell Wall: ALL cells have a cell membrane made of proteins and lipids protein channel Cell Membrane Layer 1 Layer 2 lipid bilayer protein pump SOME cells
Cell Membrane (Transport) Notes Cell Membrane and Cell Wall: ALL cells have a cell membrane made of proteins and lipids protein channel Cell Membrane Layer 1 Layer 2 lipid bilayer protein pump SOME cells
Ch. 5 Homeostasis & Cell Transport
 Ch. 5 Homeostasis & Cell Transport 5.1 Homeostasis & Permeability Homeostasis ability of cell to maintain balance needed for life To maintain balance: cells must transport needed materials into cells &
Ch. 5 Homeostasis & Cell Transport 5.1 Homeostasis & Permeability Homeostasis ability of cell to maintain balance needed for life To maintain balance: cells must transport needed materials into cells &
SOLVENT RECOVERY FROM SOYBEAN OIL/N-HEXANE MIXTURES USING HOLLOW FIBER MEMBRANE
 Brazilian Journal of Chemical Engineering ISSN 14-6632 Printed in Brazil www.abeq.org.br/bjche Vol. 29, No. 3, pp. 577-584, July - September, 212 SOLVENT RECOVERY FROM SOYBEAN OIL/N-HEXANE MIXTURES USING
Brazilian Journal of Chemical Engineering ISSN 14-6632 Printed in Brazil www.abeq.org.br/bjche Vol. 29, No. 3, pp. 577-584, July - September, 212 SOLVENT RECOVERY FROM SOYBEAN OIL/N-HEXANE MIXTURES USING
TRANSPORT ACROSS MEMBRANES
 Unit 2: Cells, Membranes and Signaling TRANSPORT ACROSS MEMBRANES Chapter 5 Hillis Textbook TYPES OF TRANSPORT ACROSS THE CELL (PLASMA) MEMBRANE: What do you remember? Complete the chart with what you
Unit 2: Cells, Membranes and Signaling TRANSPORT ACROSS MEMBRANES Chapter 5 Hillis Textbook TYPES OF TRANSPORT ACROSS THE CELL (PLASMA) MEMBRANE: What do you remember? Complete the chart with what you
Membranes. Chapter 5
 Membranes Chapter 5 Membrane Structure The fluid mosaic model of membrane structure contends that membranes consist of: -phospholipids arranged in a bilayer -globular proteins inserted in the lipid bilayer
Membranes Chapter 5 Membrane Structure The fluid mosaic model of membrane structure contends that membranes consist of: -phospholipids arranged in a bilayer -globular proteins inserted in the lipid bilayer
Membrane Structure. Membrane Structure. Membrane Structure. Membranes
 Membrane Structure Membranes Chapter 5 The fluid mosaic model of membrane structure contends that membranes consist of: -phospholipids arranged in a bilayer -globular proteins inserted in the lipid bilayer
Membrane Structure Membranes Chapter 5 The fluid mosaic model of membrane structure contends that membranes consist of: -phospholipids arranged in a bilayer -globular proteins inserted in the lipid bilayer
Transport Movement across the Cell Membrane
 Transport Movement across the Cell Membrane Lipids of cell membrane Membrane consists primarily of phosphos phospho bilayer inside cell phosphate hydrophilic outside cell hydrophobic S1 The Fluidity of
Transport Movement across the Cell Membrane Lipids of cell membrane Membrane consists primarily of phosphos phospho bilayer inside cell phosphate hydrophilic outside cell hydrophobic S1 The Fluidity of
There are mainly two types of transport :
 There are mainly two types of transport : # Type one: Passive diffusion 1- which does not require additional energy and occurs down the concentration gradient (high low concentration) " Down Hill" (^_^
There are mainly two types of transport : # Type one: Passive diffusion 1- which does not require additional energy and occurs down the concentration gradient (high low concentration) " Down Hill" (^_^
Membranes 9/15/2016. Phospholipids. Phospholipid bilayer
 Membranes Phospholipids Type of complex lipid that forms biological membranes. Have a polar hydrophilic head and two nonpolar hydrophobic tails. Amphipathic. This causes the tails to cluster together in
Membranes Phospholipids Type of complex lipid that forms biological membranes. Have a polar hydrophilic head and two nonpolar hydrophobic tails. Amphipathic. This causes the tails to cluster together in
Modified cellulosic nanofiltration membrane with improved characteristics for desalination and concentration of reactive dyest
 Indian Journal of Chemical Technology Vol.3,May 1996,pp. 131-135 Modified cellulosic nanofiltration membrane with improved characteristics for desalination and concentration of reactive dyest G Venkidachalam
Indian Journal of Chemical Technology Vol.3,May 1996,pp. 131-135 Modified cellulosic nanofiltration membrane with improved characteristics for desalination and concentration of reactive dyest G Venkidachalam
Chapter 3b Cells Membrane transport - Student Notes
 Chapter 3b Cells Membrane transport - Student Notes 1 Transport are permeable Some molecules the membrane; others do 2 Types of Membrane Transport processes No cellular required Substance its processes
Chapter 3b Cells Membrane transport - Student Notes 1 Transport are permeable Some molecules the membrane; others do 2 Types of Membrane Transport processes No cellular required Substance its processes
Chapter 5Membrane Structure and. Function
 Chapter 5Membrane Structure and Function Cell (plasma) membrane Cells need an inside & an outside separate cell from its environment ability to discriminate chemical exchanges Phospholipid Bilayer A membrane
Chapter 5Membrane Structure and Function Cell (plasma) membrane Cells need an inside & an outside separate cell from its environment ability to discriminate chemical exchanges Phospholipid Bilayer A membrane
Biology. Slide 1 / 74. Slide 2 / 74. Slide 3 / 74. Membranes. Vocabulary
 Slide 1 / 74 Slide 2 / 74 iology Membranes 2015-10-28 www.njctl.org Vocabulary Slide 3 / 74 active transport carrier protein channel protein concentration gradient diffusion enzymatic activity facilitated
Slide 1 / 74 Slide 2 / 74 iology Membranes 2015-10-28 www.njctl.org Vocabulary Slide 3 / 74 active transport carrier protein channel protein concentration gradient diffusion enzymatic activity facilitated
Cellular Neurophysiology I Membranes and Ion Channels
 Cellular Neurophysiology I Membranes and Ion Channels Reading: BCP Chapter 3 www.bioelectriclab All living cells maintain an electrical potential (voltage) across their membranes (V m ). Resting Potential
Cellular Neurophysiology I Membranes and Ion Channels Reading: BCP Chapter 3 www.bioelectriclab All living cells maintain an electrical potential (voltage) across their membranes (V m ). Resting Potential
Chapter 7 Cell Structure and Function. Chapter 7, Section 3 Cell Boundaries and Transport
 Chapter 7 Cell Structure and Function Chapter 7, Section 3 Cell Boundaries and Transport 1 7.3 A. Cell Membrane Is Described Three Ways: 1. Selectively Permeable Membrane: Limits what enters and exits
Chapter 7 Cell Structure and Function Chapter 7, Section 3 Cell Boundaries and Transport 1 7.3 A. Cell Membrane Is Described Three Ways: 1. Selectively Permeable Membrane: Limits what enters and exits
PROGRESS OF OF NANOFILTRATION MEMBRANES. 1 st Korea-US Nano Forum Kew-Ho Lee
 PROGRESS OF OF NANOFILTRATION MEMBRANES 1 st Korea-US Nano Forum 2003. 10. 14 Kew-Ho Lee Membrane and Separation Research Center Korea Research Institute of Chemical Technology TEL) 82-42-860-7240 FAX)
PROGRESS OF OF NANOFILTRATION MEMBRANES 1 st Korea-US Nano Forum 2003. 10. 14 Kew-Ho Lee Membrane and Separation Research Center Korea Research Institute of Chemical Technology TEL) 82-42-860-7240 FAX)
Cellular Physiology. Body Fluids: 1) Water: (universal solvent) Body water varies based on of age, sex, mass, and body composition
 Membrane Physiology Body Fluids: 1) Water: (universal solvent) Body water varies based on of age, sex, mass, and body composition H 2 O ~ 73% body weight Low body fat; Low bone mass H 2 O ( ) ~ 60% body
Membrane Physiology Body Fluids: 1) Water: (universal solvent) Body water varies based on of age, sex, mass, and body composition H 2 O ~ 73% body weight Low body fat; Low bone mass H 2 O ( ) ~ 60% body
Membranes. Chapter 5. Membrane Structure
 Membranes Chapter 5 Membrane Structure Lipid Bilayer model: - double phospholipid layer - Gorter & Grendel: 1925 Fluid Mosaic model: consist of -phospholipids arranged in a bilayer -globular proteins inserted
Membranes Chapter 5 Membrane Structure Lipid Bilayer model: - double phospholipid layer - Gorter & Grendel: 1925 Fluid Mosaic model: consist of -phospholipids arranged in a bilayer -globular proteins inserted
The Cell Membrane. Usman Sumo Friend Tambunan Arli Aditya Parikesit. Bioinformatics Group Faculty of Mathematics and Science University of Indonesia
 The Cell Membrane Usman Sumo Friend Tambunan Arli Aditya Parikesit Bioinformatics Group Faculty of Mathematics and Science University of Indonesia Overview Cell membrane separates living cell from nonliving
The Cell Membrane Usman Sumo Friend Tambunan Arli Aditya Parikesit Bioinformatics Group Faculty of Mathematics and Science University of Indonesia Overview Cell membrane separates living cell from nonliving
Phospholipids. Extracellular fluid. Polar hydrophilic heads. Nonpolar hydrophobic tails. Polar hydrophilic heads. Intracellular fluid (cytosol)
 Module 2C Membranes and Cell Transport All cells are surrounded by a plasma membrane. Eukaryotic cells also contain internal membranes and membrane- bound organelles. In this module, we will examine the
Module 2C Membranes and Cell Transport All cells are surrounded by a plasma membrane. Eukaryotic cells also contain internal membranes and membrane- bound organelles. In this module, we will examine the
International Journal of Mechanical Engineering and Technology (IJMET) IAEME Scopus
 International Journal of Mechanical Engineering and Technology (IJMET) Volume 5, Issue 2, February 2014, pp. 194 206, Article ID: IJMET_05_02_022 Available online at http://www.iaeme.com/ijmet/issues.asp?jtype=ijmet&vtype=5&itype=2
International Journal of Mechanical Engineering and Technology (IJMET) Volume 5, Issue 2, February 2014, pp. 194 206, Article ID: IJMET_05_02_022 Available online at http://www.iaeme.com/ijmet/issues.asp?jtype=ijmet&vtype=5&itype=2
Cell Structure and Function Exam Study Guide Part I
 Cell Structure and Function Exam Study Guide Part I 1. Which image best depicts the hot water, which the cold? 2. What is the relationship between temperature and the speed of molecular motion? 3. If a
Cell Structure and Function Exam Study Guide Part I 1. Which image best depicts the hot water, which the cold? 2. What is the relationship between temperature and the speed of molecular motion? 3. If a
THE ART TO CLEAR SOLUTIONS
 THE ART TO CLEAR SOLUTIONS FOOD AND BEVERAGE BEVERAGES Proteins and more... ln recent years membrane filtration as a modern and innovative technology has undergone significant developments and meanwhile
THE ART TO CLEAR SOLUTIONS FOOD AND BEVERAGE BEVERAGES Proteins and more... ln recent years membrane filtration as a modern and innovative technology has undergone significant developments and meanwhile
= only some molecules can get in or out of the cell. allow substances (other than lipids) in and out
 Name: Cell Membrane and Cell Transport Notes I. Cell Membrane (cells need an inside and outside) a. separate cell from its environment b. cell membrane is the boundary c. cell membrane controls what gets
Name: Cell Membrane and Cell Transport Notes I. Cell Membrane (cells need an inside and outside) a. separate cell from its environment b. cell membrane is the boundary c. cell membrane controls what gets
Membrane Transport II (Osmosis) Linda S. Costanzo, Ph.D.
 Membrane Transport II (Osmosis) Linda S. Costanzo, Ph.D. OBJECTIVES: 1. Be able to define and calculate osmolarity 2. Describe osmosis across a semipermeable membrane and the volume changes that will occur
Membrane Transport II (Osmosis) Linda S. Costanzo, Ph.D. OBJECTIVES: 1. Be able to define and calculate osmolarity 2. Describe osmosis across a semipermeable membrane and the volume changes that will occur
Paper 12: Membrane Biophysics Module 15: Principles of membrane transport, Passive Transport, Diffusion, Fick s law
 Paper 12: Membrane Biophysics Module 15: Principles of membrane transport, Passive Transport, Diffusion, Fick s law LEARNING OBJECTIVES OF MODULE: We would begin this module by considering some general
Paper 12: Membrane Biophysics Module 15: Principles of membrane transport, Passive Transport, Diffusion, Fick s law LEARNING OBJECTIVES OF MODULE: We would begin this module by considering some general
Influence of External Coagulant Water Types on the Performances of PES Ultrafiltration Membranes
 30 Journal of Membrane and Separation Technology, 2012, 1, 30-34 Influence of External Coagulant Water Types on the Performances of PES Ultrafiltration Membranes Jing He, Lingyun Ji and Baoli Shi * Polymer
30 Journal of Membrane and Separation Technology, 2012, 1, 30-34 Influence of External Coagulant Water Types on the Performances of PES Ultrafiltration Membranes Jing He, Lingyun Ji and Baoli Shi * Polymer
Transport through membranes
 Transport through membranes Membrane transport refers to solute and solvent transfer across both cell membranes, epithelial and capillary membranes. Biological membranes are composed of phospholipids stabilised
Transport through membranes Membrane transport refers to solute and solvent transfer across both cell membranes, epithelial and capillary membranes. Biological membranes are composed of phospholipids stabilised
Chapter 2 Transport Systems
 Chapter 2 Transport Systems The plasma membrane is a selectively permeable barrier between the cell and the extracellular environment. It permeability properties ensure that essential molecules such as
Chapter 2 Transport Systems The plasma membrane is a selectively permeable barrier between the cell and the extracellular environment. It permeability properties ensure that essential molecules such as
Ultrafiltration as a method of separation of natural organic matter from water
 Materials Science-Poland, Vol. 26, No. 2, 2008 Ultrafiltration as a method of separation of natural organic matter from water M. KABSCH-KORBUTOWICZ * Wrocław University of Technology, Institute of Environment
Materials Science-Poland, Vol. 26, No. 2, 2008 Ultrafiltration as a method of separation of natural organic matter from water M. KABSCH-KORBUTOWICZ * Wrocław University of Technology, Institute of Environment
Diffusion and Osmosis
 Diffusion and Osmosis The Cell Membrane The cell membrane is: Selectively permeable Permeable = Pass through (Latin) Cell membrane = Gate-Keeper that determines what can and can t enter the cell. The
Diffusion and Osmosis The Cell Membrane The cell membrane is: Selectively permeable Permeable = Pass through (Latin) Cell membrane = Gate-Keeper that determines what can and can t enter the cell. The
INVESTIGATION : Determining Osmolarity of Plant Tissue
 INVESTIGATION : Determining Osmolarity of Plant Tissue AP Biology This lab investigation has two main components. In the first component, you will learn about the osmolarity of plant tissues and the property
INVESTIGATION : Determining Osmolarity of Plant Tissue AP Biology This lab investigation has two main components. In the first component, you will learn about the osmolarity of plant tissues and the property
Environment Protection Engineering ULTRAFILTRATION OF DYE SOLUTIONS IN THE PRESENCE OF CATIONIC AND ANIONIC SURFACTANTS
 Environment Protection Engineering Vol. 35 9 No. KATARZYNA MAJEWSKA-NOWAK* ULTRAFILTRATION OF DYE SOLUTIONS IN THE PRESENCE OF CATIONIC AND ANIONIC SURFACTANTS The objective of the study was to investigate
Environment Protection Engineering Vol. 35 9 No. KATARZYNA MAJEWSKA-NOWAK* ULTRAFILTRATION OF DYE SOLUTIONS IN THE PRESENCE OF CATIONIC AND ANIONIC SURFACTANTS The objective of the study was to investigate
Movement Through the Cell Membrane
 Movement Through the Cell Membrane Cellular Movement All living organisms rely on diffusion Get oxygen for respiration Removing waste products Transpiration in plants Cellular Movement The cell membrane
Movement Through the Cell Membrane Cellular Movement All living organisms rely on diffusion Get oxygen for respiration Removing waste products Transpiration in plants Cellular Movement The cell membrane
Membrane Transport. Anatomy 36 Unit 1
 Membrane Transport Anatomy 36 Unit 1 Membrane Transport Cell membranes are selectively permeable Some solutes can freely diffuse across the membrane Some solutes have to be selectively moved across the
Membrane Transport Anatomy 36 Unit 1 Membrane Transport Cell membranes are selectively permeable Some solutes can freely diffuse across the membrane Some solutes have to be selectively moved across the
The use of dead-end and cross-flow nanofiltration to purify prebiotic oligosaccharides from reaction mixtures
 ORIGINAL ARTICLE to purify prebiotic oligosaccharides from reaction mixtures. Alistair S. Grandison 1, Athanasios K. Goulas 2, and Robert A. Rastall 3 Abstract Grandison, A.S., Goulas, A.K., and Rastall,
ORIGINAL ARTICLE to purify prebiotic oligosaccharides from reaction mixtures. Alistair S. Grandison 1, Athanasios K. Goulas 2, and Robert A. Rastall 3 Abstract Grandison, A.S., Goulas, A.K., and Rastall,
What is superhydrophobicity? How it is defined? What is oleophobicity?
 Avijit Baidya 12.11.2016 What is superhydrophobicity? How it is defined? What is oleophobicity? Introduction : Hydrophobic and amphiphobic surfaces have been studied in great depth over the past couple
Avijit Baidya 12.11.2016 What is superhydrophobicity? How it is defined? What is oleophobicity? Introduction : Hydrophobic and amphiphobic surfaces have been studied in great depth over the past couple
3D Modeling of Plaque Progression in the Human Coronary Artery
 Proceedings 3D Modeling of Plaque Progression in the Human Coronary Artery Igor Saveljic 1,2, *, Dalibor Nikolic 1,2, Zarko Milosevic 1,2, Velibor Isailovic 1,2, Milica Nikolic 1,2, Oberdan Parodi 3 and
Proceedings 3D Modeling of Plaque Progression in the Human Coronary Artery Igor Saveljic 1,2, *, Dalibor Nikolic 1,2, Zarko Milosevic 1,2, Velibor Isailovic 1,2, Milica Nikolic 1,2, Oberdan Parodi 3 and
Biochemical Techniques 06 Salt Fractionation of Proteins. Biochemistry
 . 1 Description of Module Subject Name Paper Name 12 Module Name/Title 2 1. Objectives Understanding the concept of protein fractionation Understanding protein fractionation with salt 2. Concept Map 3.
. 1 Description of Module Subject Name Paper Name 12 Module Name/Title 2 1. Objectives Understanding the concept of protein fractionation Understanding protein fractionation with salt 2. Concept Map 3.
The Cell Membrane. Also known as the Plasma Membrane
 Student Objectives Know the different parts of the cell membrane Understand the role of the cell membrane in cellular transport Understand diffusion and osmosis Determine what will happen to plant and
Student Objectives Know the different parts of the cell membrane Understand the role of the cell membrane in cellular transport Understand diffusion and osmosis Determine what will happen to plant and
Osmosis. Computer OBJECTIVES
 Osmosis Computer 22 In order to survive, all organisms need to move molecules in and out of their cells. Molecules such as gases (e.g., O 2, CO 2 ), water, food, and wastes pass across the cell membrane.
Osmosis Computer 22 In order to survive, all organisms need to move molecules in and out of their cells. Molecules such as gases (e.g., O 2, CO 2 ), water, food, and wastes pass across the cell membrane.
Plasma Membrane Structure and Function
 Plasma Membrane Structure and Function The plasma membrane separates the internal environment of the cell from its surroundings. The plasma membrane is a phospholipid bilayer with embedded proteins. The
Plasma Membrane Structure and Function The plasma membrane separates the internal environment of the cell from its surroundings. The plasma membrane is a phospholipid bilayer with embedded proteins. The
Cellular Transport Worksheet
 Cellular Transport Worksheet Name Section A: Cell Membrane Structure 1. Label the cell membrane diagram. You ll need to draw lines to some of the structures. **Draw cholesterol molecules in the membrane.**
Cellular Transport Worksheet Name Section A: Cell Membrane Structure 1. Label the cell membrane diagram. You ll need to draw lines to some of the structures. **Draw cholesterol molecules in the membrane.**
Development of an Organic-Inorganic PVDF/ anoclay Ultrafiltration Composite Membrane
 Development of an Organic-Inorganic PVDF/ anoclay Ultrafiltration Composite Membrane A.C.D. Morihama 1 and J.C. Mierwza 1 1 Escola Politécnica da Universidade de São Paulo, São Paulo, São Paulo, 05508-900,
Development of an Organic-Inorganic PVDF/ anoclay Ultrafiltration Composite Membrane A.C.D. Morihama 1 and J.C. Mierwza 1 1 Escola Politécnica da Universidade de São Paulo, São Paulo, São Paulo, 05508-900,
Transport Across the Cell Membrane 11/5/07
 11/5/07 "The difference between the internal and external chemical composition of a cell represents a degree of order, that can be maintained only by a barrier to free movement into and out of the cell.
11/5/07 "The difference between the internal and external chemical composition of a cell represents a degree of order, that can be maintained only by a barrier to free movement into and out of the cell.
Chromatography on Immobilized Artificial Membrane
 Chromatography on Immobilized Artificial Membrane Principles of measuring compounds affinity to phospholipids using immobilized artificial membrane chromatography Chromatography on Immobilized Artificial
Chromatography on Immobilized Artificial Membrane Principles of measuring compounds affinity to phospholipids using immobilized artificial membrane chromatography Chromatography on Immobilized Artificial
3. Valorization of olive oil waste streams by the development of thin film composite membranes for selective removal of polyphenols BGU, Dr. C.
 3. Valorization of olive oil waste streams by the development of thin film composite membranes for selective removal of polyphenols BGU, Dr. C. Linder Development Objectives Cost effective Extraction of
3. Valorization of olive oil waste streams by the development of thin film composite membranes for selective removal of polyphenols BGU, Dr. C. Linder Development Objectives Cost effective Extraction of
Chapter MEMBRANE TRANSPORT
 Chapter 3 I MEMBRANE TRANSPORT The cell membrane, or plasma membrane, is the outermost layer of the cell. It completely surrounds the protoplasm or living portion of the cell, separating the cell s interior
Chapter 3 I MEMBRANE TRANSPORT The cell membrane, or plasma membrane, is the outermost layer of the cell. It completely surrounds the protoplasm or living portion of the cell, separating the cell s interior
Outline. Membrane Structure and Function. Membrane Models Fluid-Mosaic. Chapter 5
 Membrane Structure and Function Chapter 5 Membrane Models Fluid-Mosaic Outline Plasma Membrane Structure and Function Protein Functions Plasma Membrane Permeability! Diffusion! Osmosis! Transport Via Carrier
Membrane Structure and Function Chapter 5 Membrane Models Fluid-Mosaic Outline Plasma Membrane Structure and Function Protein Functions Plasma Membrane Permeability! Diffusion! Osmosis! Transport Via Carrier
A molecule that can pass though the cell membrane.
 Cell Membrane All cells and most organelles are surrounded by a protective barrier known as the cell membrane, aka the plasma membrane. Section of a cell membrane It is mostly made of phospholipids (fats).
Cell Membrane All cells and most organelles are surrounded by a protective barrier known as the cell membrane, aka the plasma membrane. Section of a cell membrane It is mostly made of phospholipids (fats).
Study on the retentivity of the volatile components of simulated guava juice using ultrafiltration
 ORIGINAL ARTICLE Study on the retentivity of the volatile components of simulated guava juice using ultrafiltration Saad Hashim Khalil 1, Mohamed Kheireddine Aroua 2, and Nik Meriam Sulaiman 3 Abstract
ORIGINAL ARTICLE Study on the retentivity of the volatile components of simulated guava juice using ultrafiltration Saad Hashim Khalil 1, Mohamed Kheireddine Aroua 2, and Nik Meriam Sulaiman 3 Abstract
NANO 243/CENG 207 Course Use Only
 L9. Drug Permeation Through Biological Barriers May 3, 2018 Lipids Lipid Self-Assemblies 1. Lipid and Lipid Membrane Phospholipid: an amphiphilic molecule with a hydrophilic head and 1~2 hydrophobic tails.
L9. Drug Permeation Through Biological Barriers May 3, 2018 Lipids Lipid Self-Assemblies 1. Lipid and Lipid Membrane Phospholipid: an amphiphilic molecule with a hydrophilic head and 1~2 hydrophobic tails.
Cell Membrane-Structure and Function
 Cell Membrane-Structure and Function BIO 250 Living things are composed of cells and cell products (extracellular) Cells are the basic unit of structure They are the basic unit of function They vary in
Cell Membrane-Structure and Function BIO 250 Living things are composed of cells and cell products (extracellular) Cells are the basic unit of structure They are the basic unit of function They vary in
Plasma Membrane Function
 Plasma Membrane Function Cells have to maintain homeostasis, they do this by controlling what moves across their membranes Structure Double Layer of phospholipids Head (polar) hydrophiliclikes water -
Plasma Membrane Function Cells have to maintain homeostasis, they do this by controlling what moves across their membranes Structure Double Layer of phospholipids Head (polar) hydrophiliclikes water -
The Cell Membrane & Movement of Materials In & Out of Cells PACKET #11
 1 February 26, The Cell Membrane & Movement of Materials In & Out of Cells PACKET #11 Introduction I 2 Biological membranes are phospholipid bilayers with associated proteins. Current data support a fluid
1 February 26, The Cell Membrane & Movement of Materials In & Out of Cells PACKET #11 Introduction I 2 Biological membranes are phospholipid bilayers with associated proteins. Current data support a fluid
Hybrid silica nanofiltration membranes with low MCWO values
 Hybrid silica nanofiltration membranes with low MCWO values H.M. van Veen M.M.A. van Tuel J.P. Overbeek mw. M.D.A. Rietkerk H.J. Marsman J.F. Vente December 2014 ECN-M--14-070 1 Introduction In industrial
Hybrid silica nanofiltration membranes with low MCWO values H.M. van Veen M.M.A. van Tuel J.P. Overbeek mw. M.D.A. Rietkerk H.J. Marsman J.F. Vente December 2014 ECN-M--14-070 1 Introduction In industrial
Studies on the PVA/P(AA-Co-AN/SiO 2 )/ PVA Composite Membranes for Pervaporation Dehydration of Methanol
 Studies on the PVA/P(AA-Co-AN/SiO 2 )/ PVA Composite Membranes for Pervaporation Dehydration of Methanol Xiangchuan Pang School of Materials Science and Engineering, Tianjin Polytechnic University No.
Studies on the PVA/P(AA-Co-AN/SiO 2 )/ PVA Composite Membranes for Pervaporation Dehydration of Methanol Xiangchuan Pang School of Materials Science and Engineering, Tianjin Polytechnic University No.
The Cell Membrane. Lecture 3a. Overview: Membranes. What is a membrane? Structure of the cell membrane. Fluid Mosaic Model. Membranes and Transport
 Lecture 3a. The Cell Membrane Membranes and Transport Overview: Membranes Structure of cell membranes Functions of cell membranes How things get in and out of cells What is a membrane? Basically, a covering
Lecture 3a. The Cell Membrane Membranes and Transport Overview: Membranes Structure of cell membranes Functions of cell membranes How things get in and out of cells What is a membrane? Basically, a covering
BIOLOGY 12 - Cell Membrane and Cell Wall Function: Chapter Notes
 BIOLOGY 12 - Cell Membrane and Cell Wall Function: Chapter Notes The cell membrane is the gateway into the cell, and must allow needed things such as nutrients into the cell without letting them escape.
BIOLOGY 12 - Cell Membrane and Cell Wall Function: Chapter Notes The cell membrane is the gateway into the cell, and must allow needed things such as nutrients into the cell without letting them escape.
The Cell Membrane & Movement of Materials In & Out of Cells PACKET #11
 1 The Cell Membrane & Movement of Materials In & Out of Cells PACKET #11 Introduction I 2 Biological membranes are phospholipid bilayers with associated proteins. Current data support a fluid mosaic model
1 The Cell Membrane & Movement of Materials In & Out of Cells PACKET #11 Introduction I 2 Biological membranes are phospholipid bilayers with associated proteins. Current data support a fluid mosaic model
Movement across the Cell Membrane (Ch. 7.3)
 Movement across the Cell Membrane (Ch. 7.3) 2007-2008 Diffusion 2nd Law of Thermodynamics governs biological systems universe tends towards disorder (entropy) Diffusion movement from HIGH LOW concentration
Movement across the Cell Membrane (Ch. 7.3) 2007-2008 Diffusion 2nd Law of Thermodynamics governs biological systems universe tends towards disorder (entropy) Diffusion movement from HIGH LOW concentration
Phospholipids. Phosphate head. Fatty acid tails. Arranged as a bilayer. hydrophilic. hydrophobic. Phosphate. Fatty acid. attracted to water
 The Cell Membrane Phospholipids Phosphate head hydrophilic Fatty acid tails hydrophobic Arranged as a bilayer Phosphate attracted to water Fatty acid repelled by water I want you to remember: Structure
The Cell Membrane Phospholipids Phosphate head hydrophilic Fatty acid tails hydrophobic Arranged as a bilayer Phosphate attracted to water Fatty acid repelled by water I want you to remember: Structure
Equilibrium when two areas have the same concentration or are filled evenly
 Aim: How does the cell membrane function to maintain homeostasis? Do Now: Describe what homeostasis is. Homework: Vocab: Homeostasis, equilibrium, concentration gradient, diffusion, carrier protein, osmosis,
Aim: How does the cell membrane function to maintain homeostasis? Do Now: Describe what homeostasis is. Homework: Vocab: Homeostasis, equilibrium, concentration gradient, diffusion, carrier protein, osmosis,
Diffusion and Osmosis
 Diffusion and Osmosis OBJECTIVES: 1. To explore how different molecules move by diffusion and osmosis through semi-permeable membranes. 2. To understand how concentration affects the movement of substances
Diffusion and Osmosis OBJECTIVES: 1. To explore how different molecules move by diffusion and osmosis through semi-permeable membranes. 2. To understand how concentration affects the movement of substances
The Cell Membrane AP Biology
 The Cell Membrane 2007-2008 Warm Up What would happen if you gave a patient an IV of pure water? a. Their blood cells would shrink. b. Their blood cells would burst. c. The patient would slowly become
The Cell Membrane 2007-2008 Warm Up What would happen if you gave a patient an IV of pure water? a. Their blood cells would shrink. b. Their blood cells would burst. c. The patient would slowly become
Chapter 3.4 & 3.5 Cell Transport (Osmosis and Diffusion) = only some molecules can get in or out of the cell
 Chapter 3.4 & 3.5 Cell Transport (Osmosis and Diffusion) I. Cell Membrane (cells need an inside and outside) a. separate cell from its environment b. cell membrane is the boundary c. cell membrane controls
Chapter 3.4 & 3.5 Cell Transport (Osmosis and Diffusion) I. Cell Membrane (cells need an inside and outside) a. separate cell from its environment b. cell membrane is the boundary c. cell membrane controls
Unit 3 : Homeostasis and Cell Transport
 Unit 3 : Homeostasis and Cell Transport A. Maintaining Homeostasis 1. Homeostasis process of regulating and maintaining a constant internal env. 2. Need to maintain homeostasis of water levels, glucose
Unit 3 : Homeostasis and Cell Transport A. Maintaining Homeostasis 1. Homeostasis process of regulating and maintaining a constant internal env. 2. Need to maintain homeostasis of water levels, glucose
FIGURE A. The phosphate end of the molecule is polar (charged) and hydrophilic (attracted to water).
 PLASMA MEMBRANE 1. The plasma membrane is the outermost part of a cell. 2. The main component of the plasma membrane is phospholipids. FIGURE 2.18 A. The phosphate end of the molecule is polar (charged)
PLASMA MEMBRANE 1. The plasma membrane is the outermost part of a cell. 2. The main component of the plasma membrane is phospholipids. FIGURE 2.18 A. The phosphate end of the molecule is polar (charged)
Rapid Gradient and Elevated Temperature UHPLC of Flavonoids in Citrus Fruit
 Rapid Gradient and Elevated Temperature UHPLC of Flavonoids in Citrus Fruit Application Note General Chromatography, Food Industry Authors John W. Henderson Jr., Judy Berry, Anne Mack, William Long Agilent
Rapid Gradient and Elevated Temperature UHPLC of Flavonoids in Citrus Fruit Application Note General Chromatography, Food Industry Authors John W. Henderson Jr., Judy Berry, Anne Mack, William Long Agilent
E. Sassaroli and B. E. O Neill. The Methodist Hospital Research Institute Houston TX
 Lowering of the Interstitial Fluid Pressure as a Result of Tissue Compliance Changes during High Intensity Focused Ultrasound Exposure: Insights from a Numerical Model E. Sassaroli and B. E. O Neill The
Lowering of the Interstitial Fluid Pressure as a Result of Tissue Compliance Changes during High Intensity Focused Ultrasound Exposure: Insights from a Numerical Model E. Sassaroli and B. E. O Neill The
Process intensification in the esterification of rosin and glycerol
 Process intensification in the esterification of rosin and glycerol Proceedings of European Congress of Chemical Engineering (ECCE-6) Copenhagen, 16-0 September 007 Process intensification in the esterification
Process intensification in the esterification of rosin and glycerol Proceedings of European Congress of Chemical Engineering (ECCE-6) Copenhagen, 16-0 September 007 Process intensification in the esterification
Cell Transport. Movement of molecules
 Cell Transport Movement of molecules TEKS Students will investigate and explain cellular processes, including homeostasis and transport of molecules Homeostasis The maintaining of a stable body system
Cell Transport Movement of molecules TEKS Students will investigate and explain cellular processes, including homeostasis and transport of molecules Homeostasis The maintaining of a stable body system
Anti-(bio)fouling composite membranes by Polyacrylic acid/poly(vinyl alcohol) electrospun layer
 Anti-(bio)fouling composite membranes by Polyacrylic acid/poly(vinyl alcohol) electrospun layer Berta Díez Odriozola Chemical Engineering Department Universidad de Alcalá Madrid Spain Introduction NEW
Anti-(bio)fouling composite membranes by Polyacrylic acid/poly(vinyl alcohol) electrospun layer Berta Díez Odriozola Chemical Engineering Department Universidad de Alcalá Madrid Spain Introduction NEW
