1/3/2011. Chapter 17 Carboxylic Acids and Their Derivatives. Nucleophilic Addition- Elimination at the Acyl Carbon
|
|
|
- Phyllis Gallagher
- 5 years ago
- Views:
Transcription
1 Introduction The carboxyl group (-CO 2 H) is the parent group of a family of compounds called acyl compounds or carboxylic acid derivatives Chapter 17 Carboxylic Acids and Their Derivatives. Nucleophilic Addition- Elimination at the Acyl Carbon O R OH Carboxylic acids - Substitution of hydroxyl group will produce 2 Nomenclature and Physical Properties In IUPAC nomenclature, the name of a carboxylic acid is obtained by changing the -e of the corresponding parent alkane to -oic acid The carboxyl carbon is assigned position 1 and need not be explicitly numbered Carboxylic acids Physical Properties The common names for many carboxylic acids remain in use Methanoic and ethanoic acid are usually referred to as formic and acetic acid Carboxylic acids can form strong hydrogen bonds with each other and with water Carboxylic acids with up to 4 carbons are miscible with water in all proportions 3 4 1
2 Acidity of Carboxylic Acids The carboxyl proton of most carboxylic acids has a pk a = 4-5 Carboxylic acids are readily deprotonated by sodium hydroxide or sodium bicarbonate to form carboxylate salts Carboxylate salts are more water soluble than the corresponding carboxylic acid Dicarboxylic Acids Dicarboxylic acids are named as alkanedioic acids in the IUPAC system Common names are often used for simple dicarboxylic acids Electron-withdrawing groups near the carboxyl group increase the carboxylic acid s acidity They stabilize the carboxylate anion by inductive delocalization of charge 5 6 Nomenclature Esters Name the R as an alkyl group. This becomes the first part of the name. Name the acyl group by changing the ic acid ending of the parent carboxylic acid to the suffix ate. Nomenclature Anhydrides Symmetrical anhydrides are named by changing the acid ending of the carboxylic acid to the word anhydride. Mixed anhydrides, which are derived from two different carboxylic acids, are named by alphabetizing the names for both acids and replacing the word acid with the word anhydride
3 Nomenclature Acid Chlorides For acyclic acid chlorides: change the suffix ic acid of the parent carboxylic acid to the suffix yl chloride; or When the COCl group is bonded to a ring: change the suffix carboxylic acid to carbonyl chloride. Nomenclature Amides All 1 0 amides are named by replacing the -ic acid, -oic acid, or -ylic acid ending with the suffix amide. For 2 o and 3 0 amides, use the prefix N- preceding the name of each alkyl group to show that it is bonded to a nitrogen atom. If the two alkyl groups are different, alphabetize their names. Use additional prefix di-. One N- is needed for each alkyl group, even if both R groups are identical. Chapter Nomenclature Nitriles In naming a nitrile, the CN carbon is carbon atom ONE of the longest chain. Number the chain to put CN at C1, and omit this number from the name. So, CH 3 CH 2 CN is propanenitrile, not ethanenitrile. Common names of nitriles are derived from the names of the carboxylic acid having the same number of carbon atoms by replacing the ic acid ending of the carboxylic acid by the suffix onitrile. When the CN is named as a substituent it is called a cyano group. Spectral Properties of Acyl Compounds: IR spectra is important in identifying carboxylic acids, because C=O stretching bond occurs at different frequencies for acid, ester & amides ( cm -1 )
4 13 C NMR Spectral Properties of Acyl Componds: 13 C NMR spectra is also important in identifying carboxylic acids derivatives. The carbonyl carbon of acyl derivatives occurs downfield in the region. Please note that they resonates not as far as ketones and aldehydes which have their peaks at The nitrile carbon absorbs in the region. Preparation of Carboxylic Acids By Oxidation of Alkanes By Oxidation of Aldehydes and Primary Alcohols By Oxidation of Alkylbenzenes By Oxidation of Methyl Ketones (The Haloform Reaction) By Carbonation of Grignard Reagents Experiment which you did in Org. Chem lab. By Hydrolysis of Cyanohydrins and Other Nitriles Hydrolysis of a cyanohydrin yields an -hydroxy acid Primary alkyl halides can react with cyanide to form nitriles and these can be hydrolyzed to carboxylic acids
5 Nucleophilic Acyl Substitution Critical Concept. This is the characteristic reaction of carboxylic acid derivatives. This reaction occurs with both negatively charged nucleophiles and neutral nucleophiles. Nucleophilic Acyl Substitution - MECHANISM Acyl Substitution occurs by a Nucleophilic Addition Elimination mechanism (Important new concept!) Chapter [1] Find the sp 2 hybridized carbon with the leaving group. [2] Identify the nucleophile. [3] Substitute the nucleophile for the leaving group. With a neutral nucleophile, 18 the proton must be lost to obtain a neutral substitution product. Acyl substitution requires a leaving group (LG) at the carbonyl carbon Review the relative strength of acids (pk a ) (See table 3.1 on page REMEMBER: 111) The stronger the acid (lower pk a ) OR the weaker the conjugated base - the better the leaving group) To undergo nucleophilic addition-elimination the acyl compound must have a good leaving group or a group that can be converted into a good leaving group Acid chlorides react with loss of chloride ion Anhydrides react with loss of a carboxylate ion Esters, carboxylic acids and amides generally react with loss of the leaving groups alcohol, water and amine, respectively These leaving groups are generated by protonation of the acyl compound BUT Aldehydes and ketones cannot react by this mechanism because they lack a good leaving group
6 Relative Reactivity of Acyl Compounds The relative reactivity of carboxylic acids and their derivatives is as follows: Acid Chlorides Synthesis of Acid Chlorides Acid chlorides are made from carboxylic acids by reaction with thionyl chloride, phosphorus trichloride or phosphorus pentachloride These reagents work because they turn the hydroxyl group of the carboxylic acid into an excellent leaving group In general, reactivity can be related to the ability of the leaving group (L) to depart Leaving group ability is inversely related to basicity Chloride is the weakest base and the best leaving group Amines are the strongest bases and the worst leaving groups As a general rule, less reactive acyl compounds can be synthesized from more reactive ones (CRITICAL: help in avoiding memorization) Synthesis of more reactive acyl derivatives from less reactive ones is difficult and requires special reagents (if at all possible) A mechanism for the reaction of thionyl chloride with carboxylic acids. Reactions of Acyl Chlorides Acyl chlorides are the most reactive acyl compounds and can be used to make any of the other derivatives (Why? Because chloride ion is the best LG. Why is the best LG?) Since acyl chlorides are easily made from carboxylic acids they provide a way to synthesize any acyl compound from a carboxylic acid Acyl chlorides react readily with water, but this is not a synthetically useful reaction
7 Carboxylic Acid Anhydrides Synthesis of Carboxylic Acid Anhydrides Acid chlorides react with carboxylic acids to form mixed or symmetrical anhydrides It is necessary to use a base such as pyridine Reactions of Carboxylic Acid Anhydrides Carboxylic acid anhydrides are very reactive and can be used to synthesize esters and amides Hydrolysis of an anhydride yields the corresponding carboxylic acids Sodium carboxylates react readily with acid chlorides to form anhydrides Cyclic anhydrides with 5- and 6-membered rings can be synthesized by heating diacid Esters Synthesis of Esters: Esterification Acid catalyzed reaction of alcohols and carboxylic acids to form esters is called Fischer esterification Fischer esterification is an equilibrium process Ester formation is favored by use of a large excess of either the alcohol or carboxylic acid Ester formation is also favored by removal of water Mechanism of the Fischer Esterification To remember: reaction is catalyzed by acid and is reversible The reverse reaction is called: ACID CATALYZED ESTER HYDROLYSIS
8 Esters from Acid Chlorides Acid chlorides react readily with alcohols in the presence of a base (e.g. pyridine) to form esters Esters from Carboxylic Acid Anhydrides Alcohols react readily with anhydrides to form esters 29 What is the mechanism? Nucleophilic Addition Elimination: Hint: Find carbonyl carbon and nucleophile 30 Base-Promoted Hydrolysis of Esters: Saponification Reaction of an ester with sodium hydroxide results in the formation of a sodium carboxylate and an alcohol Lactones - or -Hydroxyacids undergo acid catalyzed reaction to give cyclic esters known as - or -lactones, respectively This mechanism is IRREVERSIBLE because the salt of the carboxylic acid is formed The last step draws the overall equilibrium toward completion of the hydrolysis Please workout problem 17.5 (p. 802) at home
9 Lactones can be hydrolyzed with aqueous base Since reaction is reversible acidification of the carboxylate product can lead back to the original lactone if too much acid is added. Exactly one equivalent of acid produced hydroxycaid Amides Synthesis of Amides Amides From Acyl Chlorides (widely used) Ammonia, primary or secondary amines react with acid chlorides to form amides An excess of amine is added to neutralize the HCl formed in the reaction Carboxylic acids can be converted to amides via the corresponding acid chloride Lactones in nature Amides can be also prepared from Esters 33 Please note chemoselectivity of the Nucleophilic Addition Elimination vs. Nucleophilic Substitution 34 Amides from Carboxylic Anhydrides Anhydrides (carbonyl substrate) react with 2 equivalents of amine (nucleophile) to produce an amide and an ammonium carboxylate Amides from Carboxylic Acids and Ammonium Carboxylates Direct reaction of carboxylic acids and ammonia yields ammonium salts Reaction of a cyclic anhydride with an amine, followed by acidification yields a product containing both amide and carboxylic acid functional groups Heating this product results in the formation of a cyclic imide Some ammonium salts of carboxylic acids can be dehydrated to the amide at high temperatures This is generally a poor method of amide synthesis THE BEST way to synthesize an amide is to convert a carboxylic acid to an acid chloride and to then to react the acid chloride with ammonia or an amine 35 Note: Amides are the linkages that join individual amino acids together to form proteins. 36 Formation of amide bonds is therefore of great importance in biochemistry. Please see useful dicyclohexylcarbodiimide(dcc)-promoted amide synthesis on 9
10 Hydrolysis of Amides Heating an amide in concentrated aqueous acid or base causes hydrolysis Hydrolysis of an amide is slower than hydrolysis of an ester Acid hydrolysis of an Amide Acid hydrolysis Basic hydrolysis Basic hydrolysis of an Amide Lactams: Cyclic Amides The size of the rings designed by greek letters Recall: lactones (cyclic esters) The penicillin antibiotic is -lactam: It acts by interfering with the synthesis of bacterial cell walls by reaction with an amino group of the essential enzymes of the cell wall biosynthetic pathway
11 Nitriles: Preparation and Chemical Properties Nitriles by the Dehydration of Amides A nitrile can be formed by reaction of an amide with phosphorous pentoxide or boiling acetic anhydride Acidic Hydrolysis of Nitriles Hydrolysis of Nitrilesil A nitrile is the synthetic equivalent of a carboxylic acid because it can be converted to a carboxylic acid by hydrolysis Basic Hydrolysis of Nitriles Decarboxylation of Carboxylic Acids -Keto carboxylic acids decarboxylate (release of CO 2 ) readily when heated. Some even decarboxylate slowly at room temperature The mechanism of -keto acid decarboxylation proceeds through a 6- membered ring transition state Malonic acids (which are too -keto acids) also decarboxylate readily (malonic ester synthesis: more in Chapter 19) 43 Here dicarboxylic acid is converted to mono carboxylic acid 44 11
Chapter 18 Carboxylic Acids and Their Derivatives. Nucleophilic Addition- Elimination at the Acyl Carbon
 Chapter 18 Carboxylic Acids and Their Derivatives. Nucleophilic Addition- Elimination at the Acyl Carbon Introduction The carboxyl group (-CO 2 H) is the parent group of a family of compounds called acyl
Chapter 18 Carboxylic Acids and Their Derivatives. Nucleophilic Addition- Elimination at the Acyl Carbon Introduction The carboxyl group (-CO 2 H) is the parent group of a family of compounds called acyl
Chapter 18. Carboxylic Acids and Their Derivatives. Nucleophilic Addition-Elimination at the Acyl Carbon
 Chapter 18 Carboxylic Acids and Their Derivatives. Nucleophilic Addition-Elimination at the Acyl Carbon Carboxylic Acids Organic compounds characterized by their acidity Contains COOH group (must be at
Chapter 18 Carboxylic Acids and Their Derivatives. Nucleophilic Addition-Elimination at the Acyl Carbon Carboxylic Acids Organic compounds characterized by their acidity Contains COOH group (must be at
CARBOXYLIC ACIDS AND THEIR DERIVATIVES: NUCLEOPHILIC ADDITION-ELIMINATION AT THE ACYL CARBON
 CARBOXYLIC ACIDS AND THEIR DERIVATIVES: NUCLEOPHILIC ADDITION-ELIMINATION AT THE ACYL CARBON RED ANT WAS SOURCE OF FORMIC ACID (RCOOH) Lecture 8 ORGANIC CHEMISTRY 2 Introduction The carboxyl group (-CO
CARBOXYLIC ACIDS AND THEIR DERIVATIVES: NUCLEOPHILIC ADDITION-ELIMINATION AT THE ACYL CARBON RED ANT WAS SOURCE OF FORMIC ACID (RCOOH) Lecture 8 ORGANIC CHEMISTRY 2 Introduction The carboxyl group (-CO
Fundamentals of Organic Chemistry CHEM 109 For Students of Health Colleges Credit hrs.: (2+1)
 Fundamentals of Organic Chemistry CHEM 109 For Students of Health Colleges Credit hrs.: (2+1) King Saud University College of Science, Chemistry Department CHEM 109 CHAPTER 7. CARBOXYLIC ACIDS AND THEIR
Fundamentals of Organic Chemistry CHEM 109 For Students of Health Colleges Credit hrs.: (2+1) King Saud University College of Science, Chemistry Department CHEM 109 CHAPTER 7. CARBOXYLIC ACIDS AND THEIR
10. CARBOXYLIC ACIDS AND THEIR DERIVATIVES 10.1 Nomenclature of Carboxylic Acids 10.2 Physical Properties of Carboxylic Acids 10.
 BOOKS 1) Organic Chemistry Structure and Function, K. Peter C. Vollhardt, Neil Schore, 6th Edition 2) Organic Chemistry, T. W. Graham Solomons, Craig B. Fryhle 3) Organic Chemistry: A Short Course, H.
BOOKS 1) Organic Chemistry Structure and Function, K. Peter C. Vollhardt, Neil Schore, 6th Edition 2) Organic Chemistry, T. W. Graham Solomons, Craig B. Fryhle 3) Organic Chemistry: A Short Course, H.
Esters of Carboxylic Acids These are derivatives of carboxylic acids where the hydroxyl group is replaced by an alkoxy group.
 Carboxylic acid Derivatives Carboxylic acid derivatives are described as compounds that can be converted to carboxylic acids via simple acidic or basic hydrolysis. The most important acid derivatives are
Carboxylic acid Derivatives Carboxylic acid derivatives are described as compounds that can be converted to carboxylic acids via simple acidic or basic hydrolysis. The most important acid derivatives are
Chapter 10. Carboxylic Acids and Derivatives. Naming Carboxylic Acids and Derivatives. Carboxylic Acids: RCOOH (RCO 2 H)
 Chapter 10 Carboxylic Acids and Derivatives Naming Carboxylic Acids and Derivatives Carboxylic Acids: RCH (RC 2 H) The functional group of a carboxylic acid is a carboxyl group (carbonyl & hydroxyl group)
Chapter 10 Carboxylic Acids and Derivatives Naming Carboxylic Acids and Derivatives Carboxylic Acids: RCH (RC 2 H) The functional group of a carboxylic acid is a carboxyl group (carbonyl & hydroxyl group)
Carboxylic Acids. The Importance of Carboxylic Acids (RCO 2 H)
 Carboxylic Acids The Importance of Carboxylic Acids (RCO 2 H) Starting materials for acyl derivatives (esters, amides, and acid chlorides) Abundant in nature from oxidation of aldehydes and alcohols in
Carboxylic Acids The Importance of Carboxylic Acids (RCO 2 H) Starting materials for acyl derivatives (esters, amides, and acid chlorides) Abundant in nature from oxidation of aldehydes and alcohols in
Carboxylic Acid Derivatives Reading Study Problems Key Concepts and Skills Lecture Topics: Structures and reactivity of carboxylic acid derivatives
 Carboxylic Acid Derivatives Reading: Wade chapter 21, sections 21-1- 21-16 Study Problems: 21-45, 21-46, 21-48, 21-49, 21-50, 21-53, 21-56, 21-58, 21-63 Key Concepts and Skills: Interpret the spectra of
Carboxylic Acid Derivatives Reading: Wade chapter 21, sections 21-1- 21-16 Study Problems: 21-45, 21-46, 21-48, 21-49, 21-50, 21-53, 21-56, 21-58, 21-63 Key Concepts and Skills: Interpret the spectra of
Carboxylic Acids and their Derivatives I
 2302272 Org Chem II Part I Lecture 5 Carboxylic Acids and their Derivatives I Instructor: Dr. Tanatorn Khotavivattana E-mail: tanatorn.k@chula.ac.th Recommended Textbook: Chapter 20 in Organic Chemistry,
2302272 Org Chem II Part I Lecture 5 Carboxylic Acids and their Derivatives I Instructor: Dr. Tanatorn Khotavivattana E-mail: tanatorn.k@chula.ac.th Recommended Textbook: Chapter 20 in Organic Chemistry,
Chapter 20: Carboxylic Acid Derivatives: Nucleophilic Acyl Substitution
 hapter 20: arboxylic Acid Derivatives: ucleophilic Acyl Substitution 20.1: omenclature of arboxylic Acid Derivatives (please read) carboxylic acid -oic acid ' ester -oate ' lactone cyclic ester l acid
hapter 20: arboxylic Acid Derivatives: ucleophilic Acyl Substitution 20.1: omenclature of arboxylic Acid Derivatives (please read) carboxylic acid -oic acid ' ester -oate ' lactone cyclic ester l acid
Carboxylic Acids and Their Derivatives. Chapter 17. Carboxylic Acids and Their Derivatives
 Chapter 17 Carboxylic Acids and Their Derivatives Chapter 17 suggested problems: 36, 38, 40, 42, 44, 52, 54, 56, 62, 64, 66, 70 Class Notes I. Carboxylic acids (organic acids) and their derivatives A.
Chapter 17 Carboxylic Acids and Their Derivatives Chapter 17 suggested problems: 36, 38, 40, 42, 44, 52, 54, 56, 62, 64, 66, 70 Class Notes I. Carboxylic acids (organic acids) and their derivatives A.
Carboxylic Acid Derivatives: Nucleophilic Acyl Substitution
 Carboxylic Acid Derivatives: Nucleophilic Acyl Substitution Carboxylic Acid Derivatives Carboxylic acid derivatives. Acyl chloride Acid anhydride Ester Amide Nucleophilic acyl substitution 19.1 Nomenclature
Carboxylic Acid Derivatives: Nucleophilic Acyl Substitution Carboxylic Acid Derivatives Carboxylic acid derivatives. Acyl chloride Acid anhydride Ester Amide Nucleophilic acyl substitution 19.1 Nomenclature
R O R' Acid anhydride. Acid halide. Carboxylic acid. Ester O O O O. Nitrile Acyl phosphate Thioester. Amide
 Chapter 10. Carboxylic Acids and Derivatives Carboxylic acid X Acid halide ' Acid anhydride Ester ' P N 2 C N S' Amide Nitrile Acyl phosphate Thioester The common structural feature of all these compounds
Chapter 10. Carboxylic Acids and Derivatives Carboxylic acid X Acid halide ' Acid anhydride Ester ' P N 2 C N S' Amide Nitrile Acyl phosphate Thioester The common structural feature of all these compounds
Chapter 19: Carboxylic Acid Derivatives: Nucleophilic Acyl Substitution 19.1: Nomenclature of Carboxylic Acid Derivatives (please read)
 problem 18.33b - = 128.7 123.9 179.7 146.8 147.4 45.3 18.0 161 hapter 19: arboxylic Acid Derivatives: ucleophilic Acyl Substitution 19.1: omenclature of arboxylic Acid Derivatives (please read) carboxylic
problem 18.33b - = 128.7 123.9 179.7 146.8 147.4 45.3 18.0 161 hapter 19: arboxylic Acid Derivatives: ucleophilic Acyl Substitution 19.1: omenclature of arboxylic Acid Derivatives (please read) carboxylic
Chapter 20: Carboxylic Acids and Nitriles شیمی آلی 2
 Chapter 20: Carboxylic Acids and Nitriles شیمی آلی 2 Dr M. Mehrdad University of Guilan, Department of Chemistry, Rasht, Iran m-mehrdad@guilan.ac.ir Based on McMurry s Organic Chemistry, 7 th edition The
Chapter 20: Carboxylic Acids and Nitriles شیمی آلی 2 Dr M. Mehrdad University of Guilan, Department of Chemistry, Rasht, Iran m-mehrdad@guilan.ac.ir Based on McMurry s Organic Chemistry, 7 th edition The
Alehydes, Ketones and Carboxylic Acid
 Alehydes, Ketones and Carboxylic Acid Aldehydes and Ketones: Introduction Aldedydes and ketones are organic compounds that contain carbon-oxygen doule bonds. The general formula for aldehydes is O C R
Alehydes, Ketones and Carboxylic Acid Aldehydes and Ketones: Introduction Aldedydes and ketones are organic compounds that contain carbon-oxygen doule bonds. The general formula for aldehydes is O C R
Carboxylic Acids and Nitriles. Chapters 20, 21 Organic Chemistry, 8th Edition John McMurry
 Carboxylic Acids and Nitriles Chapters 20, 21 Organic Chemistry, 8th Edition John McMurry 1 Carboxylic Acid Derivatives 2 Carboxylic Acid Derivatives nitrile R = CH 3 acetonitrile 3 Structure and Bonding
Carboxylic Acids and Nitriles Chapters 20, 21 Organic Chemistry, 8th Edition John McMurry 1 Carboxylic Acid Derivatives 2 Carboxylic Acid Derivatives nitrile R = CH 3 acetonitrile 3 Structure and Bonding
Loudon Chapter 21 Review: Carboxylic Acid Derivatives Jacquie Richardson, CU Boulder Last updated 3/20/2018
 Loudon Chapter 21 eview: Carboxylic Acid Derivatives Jacquie ichardson, CU Boulder Last updated 3/20/2018 We learned how to make a lot of carboxylic acid derivatives from acids in Ch. 20, but now we ll
Loudon Chapter 21 eview: Carboxylic Acid Derivatives Jacquie ichardson, CU Boulder Last updated 3/20/2018 We learned how to make a lot of carboxylic acid derivatives from acids in Ch. 20, but now we ll
Chapter 20 Carboxylic Acids. Introduction
 hapter 20 arboxylic Acids Introduction arbonyl (-=) and hydroxyl (-H) on the same carbon is carboxyl group. arboxyl group is usually written -H or 2 H. Aliphatic acids have an alkyl group bonded to -H.
hapter 20 arboxylic Acids Introduction arbonyl (-=) and hydroxyl (-H) on the same carbon is carboxyl group. arboxyl group is usually written -H or 2 H. Aliphatic acids have an alkyl group bonded to -H.
Ch 21 Carboxylic Acid Derivatives and Nu Acyl Subst n
 Ch 21 Carboxylic Acid Derivatives and Nu Acyl Subst n Acid Derivatives and their Names - Acid Halides have a Cl or Br instead of OH. Replace ic acid with yl halide, such as propionyl chloride (a common
Ch 21 Carboxylic Acid Derivatives and Nu Acyl Subst n Acid Derivatives and their Names - Acid Halides have a Cl or Br instead of OH. Replace ic acid with yl halide, such as propionyl chloride (a common
Chapter 17 Carboxylic Acids, Esters, and Amides Prepared by Andrea D. Leonard University of Louisiana at Lafayette
 Chapter 17 Carboxylic Acids, Esters, and Amides Prepared by Andrea D. Leonard University of Louisiana at Lafayette Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display.
Chapter 17 Carboxylic Acids, Esters, and Amides Prepared by Andrea D. Leonard University of Louisiana at Lafayette Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display.
Lecture 19. Nucleophilic Acyl Substitution Y - + X - Y X R C X. April 2, Chemistry 328N
 Lecture 19 Nucleophilic Acyl Substitution X Y - - Y X X - Y April 2, 2019 hemistry 328N Acid-catalyzed Esterification (also called Fischer esterification) H H 3 H H H 2 H 3 Please study the mechanism hemistry
Lecture 19 Nucleophilic Acyl Substitution X Y - - Y X X - Y April 2, 2019 hemistry 328N Acid-catalyzed Esterification (also called Fischer esterification) H H 3 H H H 2 H 3 Please study the mechanism hemistry
EXPERIMENT 8 (Organic Chemistry II) Carboxylic Acids Reactions and Derivatives
 EXPERIMENT 8 (rganic Chemistry II) Carboxylic Acids Reactions and Derivatives Pahlavan/Cherif Materials Medium test tubes (6) Test tube rack Beakers (50, 150, 400 ml) Ice Hot plate Graduated cylinders
EXPERIMENT 8 (rganic Chemistry II) Carboxylic Acids Reactions and Derivatives Pahlavan/Cherif Materials Medium test tubes (6) Test tube rack Beakers (50, 150, 400 ml) Ice Hot plate Graduated cylinders
Carboxylic Acid Derivatives: Nucleophilic Acyl Substitution Reactions
 Carboxylic Acid Derivatives: Nucleophilic Acyl Substitution Reactions Dr. Ayad Kareem Department of Pharmaceutical Chemistry, Collage of Pharmacy Al-Mustansiriyah University (2017-2018). Closely related
Carboxylic Acid Derivatives: Nucleophilic Acyl Substitution Reactions Dr. Ayad Kareem Department of Pharmaceutical Chemistry, Collage of Pharmacy Al-Mustansiriyah University (2017-2018). Closely related
Lecture 20. Herman Emil Fischer Nobel Prize 1902 Sugars, Esters and Purines. April 4, Chemistry 328N
 Lecture 20 April 4, 2019 Herman Emil Fischer 1852-1919 Nobel Prize 1902 Sugars, Esters and Purines Acid-catalyzed Esterification (also called Fischer esterification) CH CH 3 H H H 2 CCH 3 Please study
Lecture 20 April 4, 2019 Herman Emil Fischer 1852-1919 Nobel Prize 1902 Sugars, Esters and Purines Acid-catalyzed Esterification (also called Fischer esterification) CH CH 3 H H H 2 CCH 3 Please study
Carboxylic Acids and Carboxylic Acid Deriva3ves. Nucleophilic Acyl Subs0tu0on (Addi0on- Elimina0on)
 Carboxylic Acids and Carboxylic Acid Deriva3ves Nucleophilic Acyl Subs0tu0on (Addi0on- Elimina0on) 1 Carboxylic Compounds Acyl group bonded to X, an electronega3ve atom or leaving group Includes: X = halide
Carboxylic Acids and Carboxylic Acid Deriva3ves Nucleophilic Acyl Subs0tu0on (Addi0on- Elimina0on) 1 Carboxylic Compounds Acyl group bonded to X, an electronega3ve atom or leaving group Includes: X = halide
Prelab 6: Carboxylic Acids
 The Structure of Carboxylic Acids Prelab 6: Carboxylic Acids Carboxylic acids contain a carboxyl functional group attached to a hydrocarbon (alkyl group) part. Carboxyl groups contain both a carbonyl group,
The Structure of Carboxylic Acids Prelab 6: Carboxylic Acids Carboxylic acids contain a carboxyl functional group attached to a hydrocarbon (alkyl group) part. Carboxyl groups contain both a carbonyl group,
13. Carboxylic Acids (text )
 2009, Department of Chemistry, The University of Western ntario 13.1 13. Carboxylic Acids (text 14.1 14.9) A. Structure and Nomenclature The carboxylic acid functional group results from the connection
2009, Department of Chemistry, The University of Western ntario 13.1 13. Carboxylic Acids (text 14.1 14.9) A. Structure and Nomenclature The carboxylic acid functional group results from the connection
Chapters 13/14: Carboxylic Acids and Carboxylic Acid Derivatives
 CHM 201 (Elements of Organic Chemistry) Dr. Virgil Lee Cal Poly Pomona Chapters 13/14: Carboxylic Acids and Carboxylic Acid Derivatives resonance stabilized OH group donates electron density to carbonyl
CHM 201 (Elements of Organic Chemistry) Dr. Virgil Lee Cal Poly Pomona Chapters 13/14: Carboxylic Acids and Carboxylic Acid Derivatives resonance stabilized OH group donates electron density to carbonyl
Physical properties: C L = L. Cl, NH 2, OCH 3, OH, OCR O O O NH 2 CH 3 N(CH 3 ) 2. Sol. in H 2 O
 Lecture Notes hem 51 S. King hapter 22 arboxylic Acids and their Derivatives: Nucleophilic Acyl Substitution I. Structure and Physical Properties: Type 2 carbonyl compounds (carboxylic acids and derivatives)
Lecture Notes hem 51 S. King hapter 22 arboxylic Acids and their Derivatives: Nucleophilic Acyl Substitution I. Structure and Physical Properties: Type 2 carbonyl compounds (carboxylic acids and derivatives)
Chapter 21 The Chemistry of Carboxylic Acid Deriva7ves
 Organic Chemistry, 5th ed. Marc Loudon Chapter 21 The Chemistry of Carboxylic Acid Deriva7ves Eric J. Kantorowski California Polytechnic State University San Luis Obispo, CA Chapter 21 Overview 21.1 Nomenclature
Organic Chemistry, 5th ed. Marc Loudon Chapter 21 The Chemistry of Carboxylic Acid Deriva7ves Eric J. Kantorowski California Polytechnic State University San Luis Obispo, CA Chapter 21 Overview 21.1 Nomenclature
Chem 263 Nov 26, 2013 O R' alkyl. acid. ethyl. acetic acid. ethyl acetate ethyl ethanoate
 hem 263 ov 26, 2013 arboxylic Acids and Derivatives omenclature Esters Systematic names for esters are derived by first giving the name of the alkyl group attached to the oxygen, and then identifying the
hem 263 ov 26, 2013 arboxylic Acids and Derivatives omenclature Esters Systematic names for esters are derived by first giving the name of the alkyl group attached to the oxygen, and then identifying the
DERIVATIVES OF CARBOXYLIC ACIDS
 13 Rl RH RNH 2 RR RR DERIVATIVES F ARBXYLI AIDS HAPTER SUMMARY 13.1 Structure and Nomenclature of arboxylic Acid Derivatives A. Structure arboxylic acids and their derivatives can be expressed as variations
13 Rl RH RNH 2 RR RR DERIVATIVES F ARBXYLI AIDS HAPTER SUMMARY 13.1 Structure and Nomenclature of arboxylic Acid Derivatives A. Structure arboxylic acids and their derivatives can be expressed as variations
Carboxylic acid derivatives
 Carboxylic acid derivatives Nucleophilic acyl substitution reaction Among the most important reactions of carboxylic acids are those that convert the carboxyl group into other acid derivatives by a nucleophilic
Carboxylic acid derivatives Nucleophilic acyl substitution reaction Among the most important reactions of carboxylic acids are those that convert the carboxyl group into other acid derivatives by a nucleophilic
Functional Derivatives of Carboxylic Acids
 Functional Derivatives of Carboxylic Acids Derivatives of Carboxylic Acids are compounds in which the OH of a carboxyl group has been replaced by CI, OOCR, NH2, or OR'to convert acid chlorides,anhydrides,
Functional Derivatives of Carboxylic Acids Derivatives of Carboxylic Acids are compounds in which the OH of a carboxyl group has been replaced by CI, OOCR, NH2, or OR'to convert acid chlorides,anhydrides,
Carbonyl Chemistry VI + C O C. 1pm In Geology Room 112. Exam is Monday 11am-1pm. Chemistry /06/02
 arbonyl hemistry VI Ō - + hemistry 391 11/06/02 Exam is Monday 11am-1pm 1pm In Geology Room 112 The Dibasic Acids h - My - Such - hemistry 391 11/06/02 Good- Apple- Pie- Fischer Esterification Esters can
arbonyl hemistry VI Ō - + hemistry 391 11/06/02 Exam is Monday 11am-1pm 1pm In Geology Room 112 The Dibasic Acids h - My - Such - hemistry 391 11/06/02 Good- Apple- Pie- Fischer Esterification Esters can
CH 3 C H 3 O. anhydride acid. ester amide. O acid O. amide. acid. amide. acid. nitriles
 C 21: Carboxylic Acid Derivatives Topics: aming Interconversion of Acid Derivatives eactions of each functional group Connections: anhydride acid ester amide acid ester amide acid amide 2 acid nitriles
C 21: Carboxylic Acid Derivatives Topics: aming Interconversion of Acid Derivatives eactions of each functional group Connections: anhydride acid ester amide acid ester amide acid amide 2 acid nitriles
Carboxylic Acid Derivatives
 arboxylic Acid Derivatives The most important derivatives of carboxylic acids are l " ' ' acid halide acid anhydride an ester an amide Although not direct derivatives, nitriles, -, are related to carboxylic
arboxylic Acid Derivatives The most important derivatives of carboxylic acids are l " ' ' acid halide acid anhydride an ester an amide Although not direct derivatives, nitriles, -, are related to carboxylic
Carboxylic Acids, Esters and Acyl Chlorides
 R hemistry A 432 arboxylic Acids, Esters and Acyl hlorides arboxylic Acids, Esters and Acyl hlorides arboxylic acids contain the functional group, attached to an alkyl stem. They are widely found in nature,
R hemistry A 432 arboxylic Acids, Esters and Acyl hlorides arboxylic Acids, Esters and Acyl hlorides arboxylic acids contain the functional group, attached to an alkyl stem. They are widely found in nature,
ORGANIC SYNTHESIS VIA ENOLATES
 1 ORGANIC SYNTHESIS VIA ENOLATES Aldehydes and ketones undergo nucleophilic addition reaction at the carbonyl group. Further, α-hydrogen containing compounds are acidic in nature. In addition to carbonyl
1 ORGANIC SYNTHESIS VIA ENOLATES Aldehydes and ketones undergo nucleophilic addition reaction at the carbonyl group. Further, α-hydrogen containing compounds are acidic in nature. In addition to carbonyl
REACTIONS OF CARBOXYLIC ACID DERIVATIVES WITH NUCLEOPHILES A. Reactions of Acid Chlorides with Nucleophiles
 1016 CHAPTER 1 THE CHEMITRY F CARBXYLIC ACID DERIVATIVE 1.8 REACTI F CARBXYLIC ACID DERIVATIVE WITH UCLEPHILE ection 1.7 showed that all carboxylic acid derivatives hydrolyze to carboxylic acids. Water
1016 CHAPTER 1 THE CHEMITRY F CARBXYLIC ACID DERIVATIVE 1.8 REACTI F CARBXYLIC ACID DERIVATIVE WITH UCLEPHILE ection 1.7 showed that all carboxylic acid derivatives hydrolyze to carboxylic acids. Water
Chemistry Chapter 19
 hemistry 2100 hapter 19 arboxyl Derivatives In this chapter, we study three classes of compounds derived from carboxylic acids; anhydrides, esters, and amides. Each is related to a carboxyl group by loss
hemistry 2100 hapter 19 arboxyl Derivatives In this chapter, we study three classes of compounds derived from carboxylic acids; anhydrides, esters, and amides. Each is related to a carboxyl group by loss
Lecture Notes Chemistry Mukund P. Sibi Lecture 31 Reactions at the Alpha-Carbon of Carbonyl Compounds
 Lecture Notes hemistry 342-2008 Mukund P. Sibi eactions at the Alpha-arbon of arbonyl ompounds Enolates are nucleophilic and undergo reaction with electrophiles. For example, one can do halogenation under
Lecture Notes hemistry 342-2008 Mukund P. Sibi eactions at the Alpha-arbon of arbonyl ompounds Enolates are nucleophilic and undergo reaction with electrophiles. For example, one can do halogenation under
Carboxylic Acids and Their Derivatives
 arboxylic Acids and Their Derivatives Families ontaining the arbonyl Group Family Y Z Y Z aldehyde or ketone carboxylic acid or -- ester or -- acid halide or -F,-l,-Br,-I acid anhydride or amide or -N
arboxylic Acids and Their Derivatives Families ontaining the arbonyl Group Family Y Z Y Z aldehyde or ketone carboxylic acid or -- ester or -- acid halide or -F,-l,-Br,-I acid anhydride or amide or -N
Ch. 21: CARBOXYLIC ACID DERIVATIVES AND NUCLEOPHILIC ACYL SUBSTITUTION REACTIONS Nomenclature of Carboxylic Acid Derivatives:
 h. 21: ABXYLI AID DEIVATIVES AND NULEPILI AYL SUBSTITUTIN EATINS Nomenclature of arboxylic Acid Derivatives: arboxylic acids "-oic acid" Examples: 3 2 Propanoic acid yclohexanecarboxylic acid 1 arboxylate
h. 21: ABXYLI AID DEIVATIVES AND NULEPILI AYL SUBSTITUTIN EATINS Nomenclature of arboxylic Acid Derivatives: arboxylic acids "-oic acid" Examples: 3 2 Propanoic acid yclohexanecarboxylic acid 1 arboxylate
(iii). Decarboxylation (iv)kolbe Electrolysis
 ALKANOIC ACID/CARBOXYLIC ACID Contains carboxyl functional group COOH Two functional groups are contained in carboxyl- carbonyl (C=O)and hydroxyl (-OH) Saturated aliphatic alkanoic acids have general formula
ALKANOIC ACID/CARBOXYLIC ACID Contains carboxyl functional group COOH Two functional groups are contained in carboxyl- carbonyl (C=O)and hydroxyl (-OH) Saturated aliphatic alkanoic acids have general formula
Organic Chemistry. Chapter 23. Hill, Petrucci, McCreary & Perry 4 th. Ed. Alkane to Substituent Group methane CH 4 methyl CH 3
 hapter 23 rganic hemistry ill, Petrucci, Mcreary & Perry 4 th Ed. Alkane to Substituent Group methane 4 methyl 3 ethane 3 3 ethyl 3 2 propane 3 2 3 propyl 3 2 2 isopropyl ( 3 ) 2 or 3 3 butyl 3 2 2 2 butane
hapter 23 rganic hemistry ill, Petrucci, Mcreary & Perry 4 th Ed. Alkane to Substituent Group methane 4 methyl 3 ethane 3 3 ethyl 3 2 propane 3 2 3 propyl 3 2 2 isopropyl ( 3 ) 2 or 3 3 butyl 3 2 2 2 butane
Chapter 16 and GHW#6 Questions. Carboxylic Acids, Esters, and Other Acid Derivatives
 Chapter 16 and GHW#6 Questions Carboxylic Acids, Esters, and Other Acid Derivatives Bonding Characteristics of Carboxylic Acids A carboxylic acid has functional a carboxyl group. A carboxyl group is a
Chapter 16 and GHW#6 Questions Carboxylic Acids, Esters, and Other Acid Derivatives Bonding Characteristics of Carboxylic Acids A carboxylic acid has functional a carboxyl group. A carboxyl group is a
Chem 263 Nov 21, 2013
 hem 263 Nov 21, 2013 arbohydrates- emiacetal Formation You know from previous lectures that carbonyl compounds react with all kinds of nucleophiles. ydration and hemiacetal formation are typical examples.
hem 263 Nov 21, 2013 arbohydrates- emiacetal Formation You know from previous lectures that carbonyl compounds react with all kinds of nucleophiles. ydration and hemiacetal formation are typical examples.
A carboxylic acid is an organic compound that contains a carboxyl group, COOH
 1.6 Carboxylic Acids, Esters and Fats Carboxylic Acids A carboxylic acid is an organic compound that contains a carboxyl group, COOH These compounds are weak acids. Citrus fruits, crabapples, rhubarb,
1.6 Carboxylic Acids, Esters and Fats Carboxylic Acids A carboxylic acid is an organic compound that contains a carboxyl group, COOH These compounds are weak acids. Citrus fruits, crabapples, rhubarb,
Chemistry 1120 Exam 1 Study Guide
 Chemistry 1120 Exam 1 Study Guide Chapter 3 3.1 a) Know that alcohols contain a hydroxy (-OH) group. Determine the IUPAC name for a given structure by determining the longest chain. b) Determine the number
Chemistry 1120 Exam 1 Study Guide Chapter 3 3.1 a) Know that alcohols contain a hydroxy (-OH) group. Determine the IUPAC name for a given structure by determining the longest chain. b) Determine the number
where R doesn t have to equal R or R
 hem 263 Nov 24, 2016 arboxylic Acids and Derivatives arboxylic acids are very important compounds in nature and serve as building blocks for preparing related derivatives such as esters and amides. The
hem 263 Nov 24, 2016 arboxylic Acids and Derivatives arboxylic acids are very important compounds in nature and serve as building blocks for preparing related derivatives such as esters and amides. The
Paper 9: ORGANIC CHEMISTRY-III (Reaction Mechanism-2) Module17: Reduction by Metal hydrides Part-II CHEMISTRY
 Subject Chemistry Paper No and Title Module No and Title Module Tag 9: ORGANIC -III (Reaction Mechanism-2) 17: Reduction by Metal hydrides Part-1I CHE_P9_M17 Table of Contents 1. Learning Outcomes 2. Introduction
Subject Chemistry Paper No and Title Module No and Title Module Tag 9: ORGANIC -III (Reaction Mechanism-2) 17: Reduction by Metal hydrides Part-1I CHE_P9_M17 Table of Contents 1. Learning Outcomes 2. Introduction
Chemistry 212 Fall Semester 1996 Examination #2
 Chemistry 212 Fall Semester 1996 Examination #2 University of Missouri Columbia Prof. Rainer Glaser Wednesday, October 16, 1996 103 Schlundt Hall, 8:40-9:30 featuring Carboxylic Acids and Carboxylic Acid
Chemistry 212 Fall Semester 1996 Examination #2 University of Missouri Columbia Prof. Rainer Glaser Wednesday, October 16, 1996 103 Schlundt Hall, 8:40-9:30 featuring Carboxylic Acids and Carboxylic Acid
Carboxylic Acids and Esters
 24 Carboxylic Acids and Esters The sour tang in fruit juice comes from carboxylic acids. Introduction to General, Organic, and Biochemistry, 10e John Wiley & Sons, Inc Morris Hein, Scott Pattison, and
24 Carboxylic Acids and Esters The sour tang in fruit juice comes from carboxylic acids. Introduction to General, Organic, and Biochemistry, 10e John Wiley & Sons, Inc Morris Hein, Scott Pattison, and
6/9/2015. Unit 15: Organic Chemistry Lesson 15.2: Substituted Hydrocarbons & Functional Groups
 1-chloropropane 2-methylpropane 1-iodobutane Ethanoic Acid Unit 15: Organic Chemistry Lesson 15.2: Substituted Hydrocarbons & Functional Groups 43 It Ain t Just Hydrocarbons There are all sorts of organic
1-chloropropane 2-methylpropane 1-iodobutane Ethanoic Acid Unit 15: Organic Chemistry Lesson 15.2: Substituted Hydrocarbons & Functional Groups 43 It Ain t Just Hydrocarbons There are all sorts of organic
REVIEW IN CARBOXYLIC ACIDS AND ITS DERIVATIVES
 IASET: International Journal of Agricultural & Bio-Chemical Science (IASET: IJABS) ISSN(P): Applied; ISSN(E): Applied Vol. 1, Issue 1, Jan - Jun 2017, 49-66 IASET REVIEW IN CARBXYLIC ACIDS AND ITS DERIVATIVES
IASET: International Journal of Agricultural & Bio-Chemical Science (IASET: IJABS) ISSN(P): Applied; ISSN(E): Applied Vol. 1, Issue 1, Jan - Jun 2017, 49-66 IASET REVIEW IN CARBXYLIC ACIDS AND ITS DERIVATIVES
Fundamentals of Organic Chemistry CHEM 109 For Students of Health Colleges
 Fundamentals of Organic Chemistry CHEM 109 For Students of Health Colleges Credit hrs.: (2+1) King Saud University College of Science, Chemistry Department CHEM 109 CHAPTER 9. AMINO ACIDS, PEPTIDES AND
Fundamentals of Organic Chemistry CHEM 109 For Students of Health Colleges Credit hrs.: (2+1) King Saud University College of Science, Chemistry Department CHEM 109 CHAPTER 9. AMINO ACIDS, PEPTIDES AND
Carbon s unique bonding pattern arises from the hybridization of the electrons.
 Unit 8 Neptune, the 8 th planet of our solar system Organic Chemistry Organic: compound containing carbon, excluding oxides and carbonates Carbon is an allotrope, meaning it has different bonding patterns.
Unit 8 Neptune, the 8 th planet of our solar system Organic Chemistry Organic: compound containing carbon, excluding oxides and carbonates Carbon is an allotrope, meaning it has different bonding patterns.
H 3 C OCH 3 3 C N(CH 3 ) 2 H 3 C H H 3 C CH 3. ketone. pk a = 9 H H. 1,3-keto ester pk a = 11
 hapter 21: Ester Enolates 21.1: Ester α ydrogens and Their pk a s. The α-protons of s are less acidic that ketones and aldehydes. Typical pk a s of carbonyl compounds (α-protons): aldehydes 17 ketones
hapter 21: Ester Enolates 21.1: Ester α ydrogens and Their pk a s. The α-protons of s are less acidic that ketones and aldehydes. Typical pk a s of carbonyl compounds (α-protons): aldehydes 17 ketones
Carboxylic Acids. Carboxylic acid groups are always terminal groups with a carbonyl carbon also bound to a hydroxyl For example:
 Carboxylic Acids The functional group of carboxylic acids consists of a C=O with -OH bonded to the same carbon. Structure of Carboxyl Carbon is sp 2 hybridized. Bond angles are close to 120. O-H eclipsed
Carboxylic Acids The functional group of carboxylic acids consists of a C=O with -OH bonded to the same carbon. Structure of Carboxyl Carbon is sp 2 hybridized. Bond angles are close to 120. O-H eclipsed
H O. rapidly reduces. They dissolve. because they can hydrogen bond to the water molecules.
 3.9 arboxylic Acids and Derivatives Naming arboxylic acids These have the ending oic acid but no number is necessary for the acid group as it must always be at the end of the chain. The numbering always
3.9 arboxylic Acids and Derivatives Naming arboxylic acids These have the ending oic acid but no number is necessary for the acid group as it must always be at the end of the chain. The numbering always
Chapter 21. Carboxylic Acid Derivatives: Nucleophilic Acyl Substitution Reactions شیمی آلی 2
 Chapter 21. Carboxylic Acid Derivatives: Nucleophilic Acyl Substitution Reactions شیمی آلی 2 Dr M. Mehrdad University of Guilan, Department of Chemistry, Rasht, Iran m-mehrdad@guilan.ac.ir Based on McMurry
Chapter 21. Carboxylic Acid Derivatives: Nucleophilic Acyl Substitution Reactions شیمی آلی 2 Dr M. Mehrdad University of Guilan, Department of Chemistry, Rasht, Iran m-mehrdad@guilan.ac.ir Based on McMurry
A. Carboxylic acid functional groups contain the carboxyl structural feature. 1. Features of the carboxyl group
 Chapter 17 Carboxylic Acids and Their Derivatives Chem 306 Roper I. Overview A. Carboxylic acid functional groups contain the carboxyl structural feature. 1. Features of the carboxyl group 2. The reactivity
Chapter 17 Carboxylic Acids and Their Derivatives Chem 306 Roper I. Overview A. Carboxylic acid functional groups contain the carboxyl structural feature. 1. Features of the carboxyl group 2. The reactivity
Alcohol aldehydes cetones and carboxylic acids
 Alcohol aldehydes cetones and carboxylic acids 1 Classes of organic compounds 2 Alcohols Alcohols are organic compounds containing hydroxyl (-OH) group attached to C atom. In an alcohol, -OH group replaces
Alcohol aldehydes cetones and carboxylic acids 1 Classes of organic compounds 2 Alcohols Alcohols are organic compounds containing hydroxyl (-OH) group attached to C atom. In an alcohol, -OH group replaces
Chap 7: Alcohols, Phenols, & Thiols
 Chap 7: Alcohols, Phenols, & Thiols Objectives: Chap 7: Alcohols, Phenols, & Thiols (Chapter 7 and pages 283-285 & 296-297, A-1 & A-2 in lab manual) 1. Identify molecules as an alcohol, phenol, glycol,
Chap 7: Alcohols, Phenols, & Thiols Objectives: Chap 7: Alcohols, Phenols, & Thiols (Chapter 7 and pages 283-285 & 296-297, A-1 & A-2 in lab manual) 1. Identify molecules as an alcohol, phenol, glycol,
Chemistry B11 Chapters 13 Esters, amides and carbohydrates
 Chapters 13 Esters, amides and carbohydrates Esters: esters are derived from carboxylic acids (the hydrogen atom in the carboxyl group of carboxylic acid is replaced by an alkyl group). The functional
Chapters 13 Esters, amides and carbohydrates Esters: esters are derived from carboxylic acids (the hydrogen atom in the carboxyl group of carboxylic acid is replaced by an alkyl group). The functional
Identifying Functional Groups. (Chapter 2 in the Klein text)
 Identifying Functional Groups (Chapter 2 in the Klein text) Basic Ideas A functional group is a substructure within a molecule that will have the potential to undergo chemical change, i.e. the group has
Identifying Functional Groups (Chapter 2 in the Klein text) Basic Ideas A functional group is a substructure within a molecule that will have the potential to undergo chemical change, i.e. the group has
Ch14. Carboxylic Acids. Combining the hydroxyl and carbonyl functional groups. To make more powerful functional groups. version 1.
 Ch14 Carboxylic Acids Combining the hydroxyl and carbonyl functional groups. To make more powerful functional groups. version 1.0 Nick DeMello, PhD. 2007-2015 Ch14 Carboxylic Acids & Esters Carboxylic
Ch14 Carboxylic Acids Combining the hydroxyl and carbonyl functional groups. To make more powerful functional groups. version 1.0 Nick DeMello, PhD. 2007-2015 Ch14 Carboxylic Acids & Esters Carboxylic
ANSWERS BIOCHEMISTRY CARBOHYDRATES
 1. Fructose ANSWERS BIOCEMISTRY CARBOYDRATES 2. Sucrose or maltose 3. (C 6 10 O 5 ) n 4. Monosaccharides are the simplest carbohydrates which cannot be further hydrolysed. 5. D(-) fructofuranose structure:
1. Fructose ANSWERS BIOCEMISTRY CARBOYDRATES 2. Sucrose or maltose 3. (C 6 10 O 5 ) n 4. Monosaccharides are the simplest carbohydrates which cannot be further hydrolysed. 5. D(-) fructofuranose structure:
ORGANIC AND BIOORGANIC CHEMISTRY
 P. GERGELY Department os Medical Chemistry Medical and Health Science Center University of Debrecen ORGANIC AND BIOORGANIC CHEMISTRY FÓR MEDICAL STUDENTS THIRD EDITION University of Debrecen Medical and
P. GERGELY Department os Medical Chemistry Medical and Health Science Center University of Debrecen ORGANIC AND BIOORGANIC CHEMISTRY FÓR MEDICAL STUDENTS THIRD EDITION University of Debrecen Medical and
Chemistry B11 Chapters 14 Amines, aldehydes, ketones and carboxylic acids
 Chapters 4 Amines, aldehydes, ketones and carboxylic acids Amines: are derivatives from ammonia ( 3 ). Aliphatic amines: an amine in which nitrogen is bonded only to alkyl group or hydrogens. Aromatic
Chapters 4 Amines, aldehydes, ketones and carboxylic acids Amines: are derivatives from ammonia ( 3 ). Aliphatic amines: an amine in which nitrogen is bonded only to alkyl group or hydrogens. Aromatic
Chapter 21. Carboxylic Acid Derivatives. and Nucleophilic Acyl Substitution. Reactions. - many carboxylic acid derivatives are known:
 hapter 21 arboxylic Acid Derivatives and ucleophilic Acyl Substitution eactions - many carboxylic acid derivatives are known: X ' carboxylic acid acid halide (X = F, l, Br, I) acid anhydride ' 2 ester
hapter 21 arboxylic Acid Derivatives and ucleophilic Acyl Substitution eactions - many carboxylic acid derivatives are known: X ' carboxylic acid acid halide (X = F, l, Br, I) acid anhydride ' 2 ester
Chapter 15. Alcohols, Diols, and Thiols. B. Sources: there are two principal sources of simple aliphatic alcohols
 Chapter 15 Alcohols, Diols, and Thiols Chapter 15 suggested problems: 17, 19, 24, 28, 37, 39 I. Introduction A. The relevance of alcohols (from M&B: 497): "If an organic chemist were allowed to choose
Chapter 15 Alcohols, Diols, and Thiols Chapter 15 suggested problems: 17, 19, 24, 28, 37, 39 I. Introduction A. The relevance of alcohols (from M&B: 497): "If an organic chemist were allowed to choose
Chapter 9 Lecture Notes: Carboxylic Acids, Amines, and Amides
 Educational Goals Chapter 9 Lecture Notes: Carboxylic Acids, Amines, and Amides 1. Given the structure of a carboxylic acid, carboxylate ion, ester, amide, or amine molecule, be able to give the systematic
Educational Goals Chapter 9 Lecture Notes: Carboxylic Acids, Amines, and Amides 1. Given the structure of a carboxylic acid, carboxylate ion, ester, amide, or amine molecule, be able to give the systematic
Ch07. Carboxylic Acids. Combining the hydroxyl and carbonyl functional groups. To make organic acids. version 1.0
 Ch07 Carboxylic Acids Combining the hydroxyl and carbonyl functional groups. To make organic acids. version 1.0 Nick DeMello, PhD. 2007-2015 Important Dates This Wednesday: - Lab Checkout (you must check
Ch07 Carboxylic Acids Combining the hydroxyl and carbonyl functional groups. To make organic acids. version 1.0 Nick DeMello, PhD. 2007-2015 Important Dates This Wednesday: - Lab Checkout (you must check
Amines. Learning Check. Subclasses of Amines. IUPAC Naming for Amines. Common Naming for Amines. Chapter 16 Amines and Amides
 Amines Chapter 16 Amines and Amides Derivatives of ammonia, NH 3, where one or more hydrogen atoms have been replaced by an aromatic or alkyl group (R) Classified on the basis of molecular structure Primary
Amines Chapter 16 Amines and Amides Derivatives of ammonia, NH 3, where one or more hydrogen atoms have been replaced by an aromatic or alkyl group (R) Classified on the basis of molecular structure Primary
Carboxylic Acids. Seminar_7
 Seminar_7 1. Nomenclature of Carboxylic Acids 2. Preparation of Carboxylic Acids 3. Reactivity 4. Soaps and Detergents 5. Fats and Oils TEST Carboxylic acides, esters and fats. Give the names Carboxylic
Seminar_7 1. Nomenclature of Carboxylic Acids 2. Preparation of Carboxylic Acids 3. Reactivity 4. Soaps and Detergents 5. Fats and Oils TEST Carboxylic acides, esters and fats. Give the names Carboxylic
Chem 263 B6 Notes March 30, 2006 Demo-In-Class: O
 hem 263 B6 otes March 30, 2006 Demo-In-lass: + 2 carbon dioxide carbonic acid arbon dioxide ( 2 ) is a solid at -78. It is dry ice. When it is added to water, we made carbonated water (as in soda pop).
hem 263 B6 otes March 30, 2006 Demo-In-lass: + 2 carbon dioxide carbonic acid arbon dioxide ( 2 ) is a solid at -78. It is dry ice. When it is added to water, we made carbonated water (as in soda pop).
ORGANIC AND BIOLOGICAL CHEMISTRY SYSTEMATIC NOMENCLATURE
 ORGANIC AND BIOLOGICAL CHEMISTRY SYSTEMATIC NOMENCLATURE Organic compounds are carbon containing compounds. Carbon has the following unique bonding properties: 1) it has a covalence of four 2) carbon atoms
ORGANIC AND BIOLOGICAL CHEMISTRY SYSTEMATIC NOMENCLATURE Organic compounds are carbon containing compounds. Carbon has the following unique bonding properties: 1) it has a covalence of four 2) carbon atoms
Chapter 9 Educational Goals
 Chapter 9 Educational Goals 1. Given the structure of a carboxylic acid, carboxylate ion, ester, amide, or amine molecule, be able to give the systematic names and vice versa. 2. Know and understand the
Chapter 9 Educational Goals 1. Given the structure of a carboxylic acid, carboxylate ion, ester, amide, or amine molecule, be able to give the systematic names and vice versa. 2. Know and understand the
IR Spectroscopy Part II
 IR Spectroscopy Part II Carbonyl - compounds For simple aldehydes and ketones, the stretching vibration of the carbonyl group is a strong infrared absorption beetwen 1710 and 1740 cm -1. Alkyl substituents
IR Spectroscopy Part II Carbonyl - compounds For simple aldehydes and ketones, the stretching vibration of the carbonyl group is a strong infrared absorption beetwen 1710 and 1740 cm -1. Alkyl substituents
Carboxylic Acids and Esters
 arboxylic Acids and Esters N Goalby hemrevise.org - absorption IR Spectrum for arboxylic acids Butanoic acid 1 Solubility in Water The smaller carboxylic (up to 4) acids dissolve in water in all proportions
arboxylic Acids and Esters N Goalby hemrevise.org - absorption IR Spectrum for arboxylic acids Butanoic acid 1 Solubility in Water The smaller carboxylic (up to 4) acids dissolve in water in all proportions
ORGANIC CHEMISTRY Serkan SAYINER, DVM PhD. Assist. Prof.
 ORGANIC CHEMISTRY Serkan SAYINER, DVM PhD. Assist. Prof. Near East University, Faculty of Veterinary Medicine, Department of Biochemistry serkan.sayiner@neu.edu.tr Compounds Containing a C=O (Carbonyl)
ORGANIC CHEMISTRY Serkan SAYINER, DVM PhD. Assist. Prof. Near East University, Faculty of Veterinary Medicine, Department of Biochemistry serkan.sayiner@neu.edu.tr Compounds Containing a C=O (Carbonyl)
CELLULAR METABOLISM. Metabolic pathways can be linear, branched, cyclic or spiral
 CHM333 LECTURE 24 & 25: 3/27 29/13 SPRING 2013 Professor Christine Hrycyna CELLULAR METABOLISM What is metabolism? - How cells acquire, transform, store and use energy - Study reactions in a cell and how
CHM333 LECTURE 24 & 25: 3/27 29/13 SPRING 2013 Professor Christine Hrycyna CELLULAR METABOLISM What is metabolism? - How cells acquire, transform, store and use energy - Study reactions in a cell and how
Infrared Spectroscopy
 Carbonyl Compounds Cl H H N 2 1810 cm -1 (band 1) 1800 cm -1 1760 cm -1 both present (band 2) 1735 cm -1 1725 cm -1 1715 cm -1 1710 cm -1 1690 cm -1 Inductive Effects esonance Effects stronger bond W W
Carbonyl Compounds Cl H H N 2 1810 cm -1 (band 1) 1800 cm -1 1760 cm -1 both present (band 2) 1735 cm -1 1725 cm -1 1715 cm -1 1710 cm -1 1690 cm -1 Inductive Effects esonance Effects stronger bond W W
Organic Chemistry - Problem Drill 23: Amino Acids, Peptides, and Proteins
 rganic Chemistry - Problem Drill 23: Amino Acids, Peptides, and Proteins No. 1 of 10 1. Which amino acid does not contain a chiral center? (A) Serine (B) Proline (C) Alanine (D) Phenylalanine (E) Glycine
rganic Chemistry - Problem Drill 23: Amino Acids, Peptides, and Proteins No. 1 of 10 1. Which amino acid does not contain a chiral center? (A) Serine (B) Proline (C) Alanine (D) Phenylalanine (E) Glycine
Esters An Introduction To Organic Chemistry Reactions
 We have made it easy for you to find a PDF Ebooks without any digging. And by having access to our ebooks online or by storing it on your computer, you have convenient answers with esters an introduction
We have made it easy for you to find a PDF Ebooks without any digging. And by having access to our ebooks online or by storing it on your computer, you have convenient answers with esters an introduction
ESTERS AND RELATED CARBOXYLIC ACID DERIVATIVES. Jack DeRuiter
 ESTES AD ELATED ABYLI AID DEIVATIVES I. Structure and Preparation Jack Deuiter Esters are derivatives of carboxylic acids that arise via replacement of the hydroxyl () portion of the acid function with
ESTES AD ELATED ABYLI AID DEIVATIVES I. Structure and Preparation Jack Deuiter Esters are derivatives of carboxylic acids that arise via replacement of the hydroxyl () portion of the acid function with
Nu: - Addition or Nu: - Acyl Substitution?
 12. Apply nucleophilic addition and elimination concepts to nucleophilic acyl substution reactions of acids and derivatives (focus on esters and amides) In Class problems: 1. The reactive site of aldehydes,
12. Apply nucleophilic addition and elimination concepts to nucleophilic acyl substution reactions of acids and derivatives (focus on esters and amides) In Class problems: 1. The reactive site of aldehydes,
Structure of Alkenes In ethene (ethylene) each carbon is bonded to 3 other atoms, with zero nonbonding electrons => sp 2 hybridization.
 Structure and Synthesis of Alkenes Alkenes (olefins) are hydrocarbons which have carbon carbon double bonds. A double bond is a bond and a bond. Double bond B.D.E. bond B.D.E. = 146 kcal/mol = 83 kcal/mol
Structure and Synthesis of Alkenes Alkenes (olefins) are hydrocarbons which have carbon carbon double bonds. A double bond is a bond and a bond. Double bond B.D.E. bond B.D.E. = 146 kcal/mol = 83 kcal/mol
Organic Chemistry Diversity of Carbon Compounds
 Organic Chemistry Diversity of Carbon Compounds Hydrocarbons The Alkanes The Alkenes The Alkynes Naming Hydrocarbons Cyclic Hydrocarbons Alkyl Groups Aromatic Hydrocarbons Naming Complex Hydrocarbons Chemical
Organic Chemistry Diversity of Carbon Compounds Hydrocarbons The Alkanes The Alkenes The Alkynes Naming Hydrocarbons Cyclic Hydrocarbons Alkyl Groups Aromatic Hydrocarbons Naming Complex Hydrocarbons Chemical
AA s are the building blocks of proteins
 Chamras Chemistry 106 Lecture otes Chapter 24: Amino Acids, Peptides, and Proteins General Formula: () n (') α-amino Acids: (n = 1) Example: Amino Acids and Proteins: Glycine Alanine Valine AA s are the
Chamras Chemistry 106 Lecture otes Chapter 24: Amino Acids, Peptides, and Proteins General Formula: () n (') α-amino Acids: (n = 1) Example: Amino Acids and Proteins: Glycine Alanine Valine AA s are the
Chapter 22 Carbohydrates
 Chapter 22 Carbohydrates Introduction Classification of Carbohydrates Carbohydrates have the general formula C x (H 2 O) y Carbohydrates are defined as polyhydroxy aldehydes or ketones or substances that
Chapter 22 Carbohydrates Introduction Classification of Carbohydrates Carbohydrates have the general formula C x (H 2 O) y Carbohydrates are defined as polyhydroxy aldehydes or ketones or substances that
Carboxylic Acids and Derivatives. Decarboxylation R H + CO 2. R OH Reaction type: Elimination. H H Malonic acid. Mechanism:
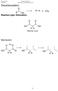 rganic hemistry arboxylic Acids and Derivatives Decarboxylation eaction type: Elimination 2 Malonic acid Mechanism: 235 rganic hemistry arboxylic Acids and Derivatives ucleophilic Acyl Substitution u Two
rganic hemistry arboxylic Acids and Derivatives Decarboxylation eaction type: Elimination 2 Malonic acid Mechanism: 235 rganic hemistry arboxylic Acids and Derivatives ucleophilic Acyl Substitution u Two
PAPER No. : 16 Bioorganic and biophysical chemistry MODULE No. : 25 Coenzyme-I Coenzyme A, TPP, B12 and biotin
 Subject Paper No and Title Module No and Title Module Tag 16, Bio organic and Bio physical chemistry 25, Coenzyme-I : Coenzyme A, TPP, B12 and CHE_P16_M25 TABLE OF CONTENTS 1. Learning Outcomes 2. Introduction
Subject Paper No and Title Module No and Title Module Tag 16, Bio organic and Bio physical chemistry 25, Coenzyme-I : Coenzyme A, TPP, B12 and CHE_P16_M25 TABLE OF CONTENTS 1. Learning Outcomes 2. Introduction
Chemistry 1120 Exam 2 Study Guide
 Chemistry 1120 Exam 2 Study Guide Chapter 6 6.1 Know amines are derivatives of ammonia, which is not an amine. Classify amines as primary, secondary or tertiary. Master Tutor Section 6.1 Review Section
Chemistry 1120 Exam 2 Study Guide Chapter 6 6.1 Know amines are derivatives of ammonia, which is not an amine. Classify amines as primary, secondary or tertiary. Master Tutor Section 6.1 Review Section
Ex17. Analgesics, TLC Analysis. Analgesics. The Experiment. Part A. Carboxylic Acids. Part B. Willow Bark Esters & Esterification
 Ex17 Analgesics, TLC Analysis Analgesics Carboxylic Acids Structure Properties Willow Bark Esters & Esterification The Experiment Part A Use TLC to Separate Compounds Part B Determine Elution Solvents
Ex17 Analgesics, TLC Analysis Analgesics Carboxylic Acids Structure Properties Willow Bark Esters & Esterification The Experiment Part A Use TLC to Separate Compounds Part B Determine Elution Solvents
