GOVERNMENT OF INDIA MINISTRY OF FINANCE (DEPARTMENT OF REVENUE) Notification. No. 48/ Customs (N.T.)
|
|
|
- Marian Morton
- 6 years ago
- Views:
Transcription
1 [TO BE PUBLISHED IN THE GAZETTE OF INDIA, EXTRAORDINARY, PART-II, SECTION 3, SUB-SECTION (i)] GOVERNMENT OF INDIA MINISTRY OF FINANCE (DEPARTMENT OF REVENUE) Notification New Delhi, the 16 th May, 2017 No. 48/ Customs (N.T.) G.S.R. (E).- In exercise of the powers conferred by sub-section (1) of section 5 of the Customs Tariff Act, 1975 ( 51 of 1975), the Central Government, being satisfied that it is necessary in the public interest so to do, hereby makes the following rules to amend the Rules of Determination of Origin of Goods under the Preferential Trading Agreement between the Republic of India and the Republic of Chile Rules, 2007 (hereinafter referred to as the said rules), notified by the Government of India in the Ministry of Finance (Department of Revenue), Notification No. 84/2007-Customs (N.T.), dated the 17 th August, 2007, published in the Gazette of India, Extraordinary, Part II, Section 3, Sub-section (ii), vide number S.O (E), dated the 17 th August, 2007, namely:- 1. (1) These rules may be called the Determination of Origin of Goods under the Preferential Trading Agreement between the Republic of India and the Republic of Chile (Amendment) Rules, (2) They shall come into force on the date of their publication in the Official Gazette. 2. In the said rules, in rule 6, after sub-rule (5), the following sub-rule shall be inserted, namely:- (6) Notwithstanding the provisions of the sub-rule (1) and sub-rule (2), a product shall be considered as originating if it fulfils the conditions laid down in the Annex to these rules and final process of manufacturer is performed within the territory of the exporting Party.. 3. In the said rules, in Section III, under the heading PROOF OF ORIGIN, for rule 15 and rule 16, the following rules shall be substituted, namely:- Rule 15. Certificate of Origin (1) The certificate of origin is the document that certifies that goods fulfil the origin requirements as set out in these rules so that they can benefit from the 1
2 preferential tariff treatment as foreseen in this Agreement and it is valid for only one importing operation concerning one or more goods. (2) For the purpose of claiming the preferential tariff treatment for an originating goods of the exporting Party, the certificate of origin shall be submitted in original to the customs authority of the importing Party. (3) The certificate of origin shall be submitted within its validity period. (4) The certificate of origin submitted to the customs authority of importing Party after the expiration of the validity of the certificate of origin shall be accepted for the purpose of claiming preferential tariff when the failure to observe the time limit results from force majeure or other valid reasons beyond the control of the exporter: Provided that, in all cases, the customs authority of importing Party shall accept such certificate of origin, if the products have been imported before the expiry of the validity period of the said certificate of origin. (5) The customs authority of importing Party may request an importer for information or documents relating to the origin of imported product in accordance with the law for the time being in force in the territory of the importing Party. (6) Notwithstanding the provisions of the sub-rule (5), if the importer does not possess the information or if the information provided is not sufficient to determine whether the goods are originating in the exporting Party, the customs authority of the importing Party shall request information from the issuing authority in accordance with section IV of these rules. (7) The issue of certificates of origin and its control, shall be under the responsibility of a Government office in each Party and the certificates of origin shall be directly issued by those authorities or through delegation as referred to in sub-rule (8) and shall be in English. (8) The certificate of origin shall be signed and issued by Government offices to be indicated by the Parties who may delegate the signing and issuing of certificate of origin to other Government offices or representative corporate body. (9) Each Party shall inform the competent authorities of other Party of the names and addresses of the authorised officials of its respective issuing authority of certificate of origin and also provide sets of their specimen signatures and specimen of official seals. (10) Each Party shall intimate the name, designation and contact details (address, phone number, fax number, ) of its competent authorities for the purposes of these rules- 2
3 (i) to whom the specimen seals and signatures of the issuing authorities of the other Party should be communicated:- India: The Director (International Customs), Central Board of Excise and Customs, Department of Revenue, Ministry of Finance, Government of India, Room No. 49, North Block, New Delhi , INDIA. Telephone: Fax Chile: Certification and Verification Department, General Directorate of International Economic Affairs, Ministry of Foreign Affairs, Teatinos 180 Santiago Chile; Telephone: ; (ii) to whom the references of verification of Certificate of Origin issued by the Party, should be addressed:- India: The Director, RMTR Division, Department of Commerce, Ministry of Commerce & Industry, Government of India, Room No. 224C, Udyog Bhawan, New Delhi. Tel. No d2rmtr-doc@nic.in Chile: Certification and Verification Department, General Directorate of International Economic Affairs, Ministry of Foreign Affairs, Teatinos 180, Santiago. 3
4 Telephone: ; (iii) from whom the specimen seals and signatures of the Issuing Authorities of the other Party would be received: - India: The Director, RMTR Division, Department of Commerce, Ministry of Commerce & Industry, Government of India, Room No. 224C, Udyog Bhawan, New Delhi. Tel. No d1rmtr-doc@nic.in, d2rmtr-doc@nic.in Chile: Certification and Verification Department, General Directorate of International Economic Affairs, Ministry of Foreign Affairs, Teatinos 180, Santiago. Telephone: certificacionorigen@direcon.gob.cl ; (iv) from whom references would emanate for verification of Certificate of Origin issued by the other Party:- India: The Director (International Customs), Central Board of Excise and Customs, Department of Revenue, Ministry of Finance, Government of India, Room No. 49, North Block, New Delhi , INDIA. Telephone: Fax diricd-cbec@nic.in Chile: Head of Origin Sub-Department Customs Technique Sub-Directorate 4
5 National Customs Service Sotomayor 60, 1 st Floor, Valparaiso, Chile Telephone: (56-32) (56-32) (11) Any change in the competent authorities or the names, designations, addresses, specimen signatures or officials seals shall be promptly informed to the other Party. (12) The certificate of origin shall be issued in the format in Appendix to these rules and upon submission of a sworn declaration by the final producer of the goods, referred to in sub-rule (1) of rule 16, along with the respective commercial invoice. (13) In all cases, the number and date of the commercial invoice shall be indicated in the box reserved for this purpose in the certificate of origin. (14) When the goods to be traded is invoiced by a non-party operator, the producer or exporter of the originating Party shall inform, in the field titled observations of the respective certificate of origin, that the goods subject to declaration shall be invoiced from that non-party operator, reproducing the following data from the commercial invoice issued by this operator, namely:- name, address, country, number and date: Provided that value addition, where applicable, carried out only in the territory of the exporting Party shall be taken into account for calculation of local value addition. Rule 16. Issue of Certificates of Origin (1) For the issue of a certificate of origin, the final producer or exporter of the goods shall present the corresponding commercial invoice and a request containing a sworn declaration by the final producer certifying that the goods fulfil the origin criteria of these rules, as well as the necessary documents supporting such a declaration and the said sworn declaration shall contain at least the following data, namely:- (a) (b) (c) (d) individual's name or company name; address; description of the goods to be exported and its tariff classification; FOB value of the goods to be exported; and 5
6 (e) information relating to the goods to be exported, which must indicate: (i) (ii) materials, components and/or parts originating from the exporting Party and the Customs tariff heading, wherever possible, materials, components and/or parts originating from the other Party indicating: - origin, - tariff classification (at least 6 digit level), - CIF value, in United States of America dollars, and - percentage on the total value of the final product. (iii) non-originating materials, components and/or parts indicating: - exporting Country, - tariff classification (at least 6 digit level), - CIF value, in United States of America dollars, and - percentage on the total value of the final product; and (iv) description of the manufacturing process. (2) The description of the goods in the sworn declaration, which certifies the fulfilment of the origin requirements set out in these rules, shall correspond to the respective tariff classification, as well as with the description of the goods in the commercial invoice and in the certificate of origin. (3) If the goods are regularly exported and their manufacturing process, as well as their materials are not modified, the sworn declaration of the producer may be valid for a period of up to one year counted from the date of the issue of the certificate of origin. (4) The certificate of origin shall be issued not later than five working days after the presentation of request and it shall be valid for a period of one year from the date of its issue. (5) The certificates of origin shall not be issued before the date of the issue of the commercial invoice relating to the consignment, but may be issued on the same date 6
7 or within the following sixty days. (6) In the event of theft, loss or destruction of a certificate of origin, the exporter may apply in writing to the issuing authority which issued it for a certified true copy of the original to be made on the basis of the export documents in their possession bearing the endorsement of the words CERTIFIED TRUE COPY, (in lieu of the original certificate) in box 4 of the certificate of origin and this copy shall bear the date of the original certificate of origin. (7) Minor discrepancies between the certificate of origin and the documents submitted to the customs authority of the importing Party for the purpose of carrying out the formalities for importing the products shall not ipso facto invalidate the certificate of origin if it corresponds to the products under importation. (8) The requesting Party and the certifying offices or authorised institutions shall keep the documents supporting the certificate of origin for a period no less than five years, from the date of its issue and the issuing authority shall enumerate the certificates issued by them in sequential order. (9) The issuing authority shall keep a permanent record of all issued certificate of origin, which shall contain at least the certificate number, the requesting entity s name and the date of its issue.. 4. In the said rules, in Section IV, under the heading Control and Verification of Certificates of Origin, for rule 17 to rule 36 the following shall be substituted; namely:- Rule Regardless of the presentation of a certificate of origin in accordance with the these rules, the customs authorities of importing Party in the cases of reasonable doubt, as to the authenticity of a certificate of origin, as well as the veracity of the information contained therein, may initiate an verification in accordance with the procedures established in rule 18 and this shall not preclude the application of the respective national legislation including customs law. 2. The competent authorities of importing Party shall put forward the reasons for the doubts concerning the authenticity of the certificate of origin or the veracity of its data in a clear and concrete way to the issuing authority of the exporting Party. Rule 18 During the verification proceedings, the customs authority of the importing Party may request the issuing authority of the exporting Party, in writing, for a verification of origin in accordance with the following procedure, namely:- (a) the request for information, as well as any copy of the documentation in 7
8 possession of the issuing authority which issued the certificate of origin under verification, which may be deemed necessary for verifying the authenticity of the said certificate of origin and the veracity of the information contained therein and in such a request, the number and the date of the issue of the certificate of origin under verification shall be indicated; (b) for the purposes of verification of the contents of the local or regional added value, the producer or exporter shall facilitate the access to any information or documentation necessary for establishing the CIF value of the non-originating goods used in the production of the goods under verification; (c) for the purposes of verification of the characteristics of certain production processes, the exporter or producer shall facilitate the access to any information and documentation that allow the confirmation of such processes; (d) the customs authority of the importing Party may send a questionnaire to the issuing authority of the exporting Party which shall be passed on to the exporter or producer, indicating the certificate of origin under verification; (e) when the customs authority of the importing Party is not satisfied with the results of the verification of origin pursuant to clauses (a) to (d), it shall deliver a written request to the issuing authority of the exporting Party to facilitate visits to the premises of the producer, with a view to examining the production processes, as well as the equipment and tools utilised in the manufacture of the product under verification; (f) the issuing authority of the exporting Party will provide the requisitioned documents, statement and cost sheet of materials to the customs authority of the importing Party at least fifteen days before the date of visit; (g) the issuing authority of the exporting Party shall accompany the authorities of the importing Party in their above-mentioned visit, which may include the participation of specialists who shall act as observers: Provided that, each Party may designate specialists, who shall be neutral and have no interest whatsoever in the verification: Provided further that, each Party may deny the participation of such specialists whenever the latter represent the interests of the companies involved in the verification; (h) once the visit is concluded, the participants shall subscribe the minutes of it, in which it shall be indicated that it was carried out according to the conditions established in these rules and the said minutes shall contain, in addition, the 8
9 following information, namely:- (i) (ii) date and place of the carrying out of the visit; identification of the certificates of origin which led to the verification; (iii) identification of the goods under verification; (iv) identification of the participants, including indications of the organs and institutions to which they belong; and (v) a visit report; (i) the exporting Party may request the postponement of a verification visit for a period not more than thirty days; and (j) the Parties may carry out other actions as mutually agreed upon between them, relating to the case under verification. Rule 19 The issuing authorities of the exporting Party shall provide the information and documentation requested according to rule 18, within,- (i) (ii) thirty days of the date of receipt of the request, if the request pertains to the authenticity of seal and signatures of the issuing authority of the beneficiary country; thirty days of the date of receipt of the request, if the request is to seek a copy of the application made by the exporter or producer; (iii) sixty days from the date of receipt of such request, if the request is on the grounds of suspicion of the accuracy of the information regarding the origin of the product and such period can be extended through mutual consultation for a period no more than thirty days upon request of the exporting Party. Rule 20 (1) The customs authorities of the importing Party shall not suspend the importation operations of the goods or of identical goods from the same exporter or producer. (2) The customs authorities of the importing Party may deny preferential tariff treatment and request a guarantee in any of its modalities or may take any action 9
10 necessary in order to preserve fiscal interests, as a pre-condition for the completion of the importation operations subject to and in accordance with the laws and procedures of the importing Party: Provided that, when a guarantee is required, its amount shall not be higher than the value of the applicable custom duties concerning the importation of the product from third countries, according to the legislation of the importing country. Rule 21 The information obtained under the conditions of these rules shall be confidential in character, in accordance with its law, and each Party shall protect such information from disclosure that could prejudice the competitive position of the persons providing the information and it shall be utilised with a view to clarifying the matter under verification by the competent authorities of the importing Party as well as during the verification and legal proceedings. Rule 22 The customs authorities of the importing Party shall immediately notify the importer and the competent authorities of the exporting Party upon initiation of the verification envisaged under rule 17, in accordance with the procedures established in rule 18. Rule 23 The competent authorities of the importing Party shall engage to conclude the verification in a period preferably within sixty days but not more than ninety days, from the date of the receipt of all the information requested in accordance with rule 18: Provided that where new investigative actions or the presentation of more information are necessary, the competent authorities of the importing Party shall communicate the fact to the competent authorities of the exporting Party and the term for the execution of such new actions or for the presentation of additional information shall be not more than sixty days, from the date of the receipt of all the additional information, according to rule 18. Rule 24 Except as otherwise provided in these rules, the competent authorities of the importing Party may consider that the goods under verification do not fulfil the origin requirements and may deny preferential tariff treatment to the goods mentioned in the certificate of origin under verification according to rule 17, when,- 10
11 (a) (b) (c) (d) (e) (f) the goods do not meet the requirements of these rules; the exporter, producer or importer of the goods fail to demonstrate compliance with the requirements which are applicable under these rules; the exporter or producer of the goods deny access to the relevant records or documentation; the competent authorities of the exporting Party fail to provide the information in pursuance to a written request for verification within stipulated time; the consent to a request for verification visit is not received from the competent authorities of the exporting Party or the exporter or producer; or the information provided by the issuing authorities of the exporting Party or exporter or producer is not sufficient to prove that the goods qualifies as an originating goods of the exporting Party. Rule 25 (1) In cases where the certificate of origin is rejected by the competent authorities of the importing Party, the original certificate of origin shall be returned to the competent authorities of the exporting Party within a reasonable period but not exceeding two months from the date of such rejection and the competent authorities of the importing Party shall communicate the grounds for denial of preferential tariff treatment to the importer and the competent authorities of the exporting Party. (2) The customs authority of the importing Party shall grant the competent authority of the exporting Party, access to the verification files, in accordance with its domestic legislation. Rule 26 During the investigation process, occasional modifications in the manufacturing conditions made by the companies under verification shall be taken into account for future shipments. Rule 27 Once the verification for the qualification of the origin concludes with a determination in favour of the importer, the importer shall be released from the guarantees requested in rule 20, within not more than thirty days and shall be promptly refunded the duties paid in excess in accordance with the domestic legislation of the Parties. 11
12 Rule 28 (1) Once the verification establishes the non-qualification of the origin criterion of the goods contained in the certificate of origin, the duties shall be levied in accordance with the domestic legislation in force in the importing Party. (2) In such a case, the competent authorities of the importing Party may deny preferential tariff treatment to new imports relating to identical goods from the same producer, until it is clearly demonstrated that the manufacturing conditions were modified so as to fulfil the origin requirements of the rules of origin of these rules. (3) Once the competent authorities of the exporting Party has sent the information demonstrating that the manufacturing conditions were modified and goods fulfil the origin criterion, the competent authorities of the importing Party shall have fortyfive day time, from the date of the receipt of the said information, to communicate its decision there upon, or to request for a verification visit to the producer's premises, according to clause (e) of rule 18, if deemed necessary. (4) If the competent authorities of the importing and the exporting Parties fail to agree on the demonstration of the modification of the manufacturing conditions, they may make use of the Dispute Settlement Procedure established as per Article XVIII of this Agreement. Rule 29 (1) A Party may request to the other Party to investigate the origin of a goods imported by the latter from other Party, whenever there are well-founded reasons for suspecting that its products undergo competition from imported products with preferential tariff treatment which do not fulfil the requirements of these rules. (3) For such purposes, the competent authorities of the Party requesting the verification shall bring to the notice of the authorities of the importing Party the relevant information within sixty days, from the date of the request and once this information is received, the importing Party may initiate the proceedings established in these rules, giving notice of this to the Party that requested the initiation of the verification. Rule 30 The proceedings of verification and control of origin as foreseen in these rules may also apply to the goods already cleared for home consumption. Rule 31 Within sixty days, from the receipt of the communication as provided in rule 25 or in 12
13 the sub-rule (3) of rule 28, in case the measure is inconsistent, the exporting Party may request for consultation to the Joint Administration Committee referred to in Article XVII of this Agreement, stating the technical and legal reasons that would indicate that the measure adopted by the competent authorities of the importing Party are not consistent with these rules; and/or request a technical advice with the aim of establishing whether the goods under verification fulfil the requirements of these rules. Rule 32 The time periods set in these rules shall be calculated on a consecutive day basis as from the day following the fact or event which they refer to. Rule 33 Each Party shall adopt or maintain measures that provide for the imposition of civil, administrative, and, where appropriate, criminal sanctions for violations of its customs laws and regulations, including those governing tariff classification, customs valuation, rules of origin, and the entitlement to preferential tariff treatment under this Agreement.. 5. In the said rules, after rule 33, the following Annex shall be inserted, namely:- Annex [see sub-rule (6) of rule 6] Product Specific Rules PART 1 General Notes For the purposes of the product specific rules set out in this Annex,- (a) (b) (c) the product specific rule, or specific set of rules, that applies to a particular chapter, heading or subheading is set out immediately adjacent to the chapter, heading or subheading; where the specific set of rules provides for more than one rule to be selectively applied, the order of the description of the rules does not indicate priority of application; the following definitions apply: (i) (ii) the term chapter means a chapter of the Harmonized System; the term heading means the first four digits in the tariff classification number under the Harmonized System; and 13
14 (iii) the term subheading means the first six digits in the tariff classification number under the Harmonized System; and (d) this Annex is based on the Harmonized System as amended on January 1, PART 2 PRODUCT SPECIFIC RULES No. HS Code Product Specific Rule 1 Chapter 1 A change to Chapter 1 from any other chapter 2 Chapter 2 A change to Chapter 2 from any other chapter 3 Chapter 3 A change to Chapter 3 from any other chapter 4 Chapter 4 A change to Chapter 4 from any other chapter 5 Chapter 5 A change to Chapter 5 from any other chapter 6 Chapter 6 A change to Chapter 6 from any other chapter 7 Chapter 7 A change to Chapter 7 from any other chapter 8 Chapter 8 A change to Chapter 8 from any other chapter 9 Chapter 9 A change to Chapter 9 from any other chapter 10 Chapter 10 A change to Chapter 10 from any other chapter 11 Chapter 11 A change to Chapter 11 from any other chapter 12 Chapter 12 A change to Chapter 12 from any other chapter for Oil seeds and oleaginous fruits; miscellaneous grains, seeds and fruit; industrial or medicinal plants; straw and fodder except for Ginseng roots and other seeds chilled and frozen 13 Chapter 13 A change to Chapter 13 from any other chapter 14 Chapter 14 A change to Chapter 14 from any other chapter 15 Chapter 15 A change to Chapter 15 from any other chapter 16 Chapter 16 A change to Chapter 16 from any other chapter A change to sub-headings through from any other heading A change to sub-heading from any other heading A change to sub-heading from any other heading A change to sub-heading from any other heading A change to sub-heading from any other heading A change to sub-heading from any other heading A change to sub-heading from any other heading A change to sub-heading through from any other heading A change to sub-heading from any other heading A change to sub-heading from any other heading A change to sub-heading from any other heading A change to sub-heading from any other heading 14
15 No. HS Code Product Specific Rule A change to sub-heading from any other heading A change to sub-heading from any other heading A change to sub-heading from any other heading A change to sub-heading from any other heading A change to sub-heading from any other heading A change to sub-heading from any other heading A change to sub-heading from any other heading A change to sub-heading from any other heading A change to sub-heading from any other heading A change to sub-heading from any other heading or the A change to sub-heading from any other heading or the A change to sub-heading from any other chapter A change to sub-heading from any other chapter A change to sub-heading from any other chapter A change to sub-heading from any other chapter A change to sub-heading from any other chapter A change to sub-heading from any other chapter A change to sub-heading from any other chapter A change to sub-heading from any other chapter A change to sub-heading from any other chapter A change to sub-heading from any other heading A change to sub-heading from any other heading A change to sub-heading from any other heading A change to sub-heading from any other heading or the A change to sub-heading through from any other heading A change to sub-heading through from any other heading A change to sub-heading from any other heading A change to sub-heading from any other heading A change to sub-heading from any other heading A change to sub-heading from any other heading A change to sub-heading from any other heading 15
16 No. HS Code Product Specific Rule A change to sub-heading through from any other heading A change to sub-heading through from any other heading A change to sub-heading from any other heading A change to sub-heading from any other heading A change to sub-heading from any other heading A change to sub-heading from any other heading A change to sub-heading from any other heading A change to sub-heading from any other heading A change to sub-heading from any other heading A change to sub-heading from any other heading A change to sub-heading from any other heading A change to sub-heading from any other heading A change to sub-heading from any other heading or the A change to sub-heading from any other heading or the A change to sub-heading from any other heading or the A change to sub-heading from any other heading or the A change to sub-heading from any other heading or the A change to sub-heading from any other heading or the A change to sub-heading from any other heading or the A change to sub-heading from any other heading or the A change to sub-heading from any other heading or the 16
17 No. HS Code Product Specific Rule A change to sub-heading from any other heading or the A change to sub-heading from any other heading or the A change to sub-heading from any other heading or the A change to sub-heading from any other heading or the A change to sub-heading through from any other A change to sub-heading through from any other A change to sub-heading through from any other A change to sub-heading through from any other A change to sub-heading from any other heading or the A change to sub-heading from any other heading or the A change to sub-heading through from any other A change to sub-heading from any other heading or the 17
18 No. HS Code Product Specific Rule A change to sub-heading from any other heading or the A change to sub-heading from any other heading or the A change to sub-heading from any other heading or the A change to sub-heading from any other heading or the A change to sub-heading from any other heading or the A change to sub-heading through from any other A change to sub-heading through from any other A change to sub-heading through from any other A change to sub-heading through from any other A change to sub-heading from any other heading or the A change to sub-heading through from any other A change to sub-heading through from any other 18
19 No. HS Code Product Specific Rule A change to sub-heading from any other heading or the A change to sub-heading from any other heading or the A change to sub-heading through from any other A change to sub-heading through from any other A change to sub-heading through from any other A change to sub-heading through from any other A change to sub-heading through from any other A change to sub-heading from any other heading or the A change to sub-heading from any other heading or the A change to sub-heading from any other heading or the A change to sub-heading through from any other A change to sub-heading from any other heading or the 19
20 No. HS Code Product Specific Rule A change to sub-heading through from any other A change to sub-heading through from any other A change to sub-heading through from any other A change to sub-heading through from any other A change to sub-heading from any other heading or the A change to sub-heading from any other heading or the A change to sub-heading through from any other A change to sub-heading through from any other A change to sub-heading through from any other A change to sub-heading from any other heading or the A change to sub-heading through from any other 20
21 No. HS Code Product Specific Rule A change to sub-heading through from any other A change to sub-heading through from any other A change to sub-heading through from any other A change to sub-heading from any other heading or the A change to sub-heading through from any other A change to sub-heading from any other heading or the A change to sub-heading from any other heading or the A change to sub-heading from any other heading or the A change to sub-heading from any other heading or the A change to sub-heading through from any other A change to sub-heading through from any other A change to sub-heading from any other heading or the 21
22 No. HS Code Product Specific Rule A change to sub-heading from any other heading or the A change to sub-heading from any other heading or the A change to sub-heading through from any other A change to sub-heading from any other heading or the A change to sub-heading from any other heading or the A change to subheading from any other heading excepting 8431 or the total value of all non-originating materials or of of the product for Parts of machinery of heading 8428 except of lifts, skip hoists or escalators, which are Monopods, bipods, tripods and similar articles.. [F. No.528/15003/2009-Cus/ICD] (Satyajit Mohanty) Director, Government of India Note: The notification No.84/2007- Customs (N.T.), dated 17 th August, 2007, was published in the Gazette of India, Extraordinary, vide number S.O (E), dated the 17 th August,
AGREEMENT ON COMMON PRINCIPLES AND RULES OF CIRCULATION OF MEDICINAL PRODUCTS WITHIN THE EURASIAN ECONOMIC UNION. (Moscow, 23 December 2014)
 AGREEMENT ON COMMON PRINCIPLES AND RULES OF CIRCULATION OF MEDICINAL PRODUCTS WITHIN THE EURASIAN ECONOMIC UNION (Moscow, 23 December 2014) Member States of the Eurasian Economic Union, hereinafter referred
AGREEMENT ON COMMON PRINCIPLES AND RULES OF CIRCULATION OF MEDICINAL PRODUCTS WITHIN THE EURASIAN ECONOMIC UNION (Moscow, 23 December 2014) Member States of the Eurasian Economic Union, hereinafter referred
Employment Contract. This sample employment contract is from Self-Employment vs. Employment Status, CDHA (no date available)
 Employment Contract This sample employment contract is from Self-Employment vs. Employment Status, CDHA (no date available (NOTE: This is only one example of an employment contract. This example is meant
Employment Contract This sample employment contract is from Self-Employment vs. Employment Status, CDHA (no date available (NOTE: This is only one example of an employment contract. This example is meant
REGULATION (EC) No.141/2000
 REGULATION (EC) No.141/2000 Community legislation in force Document 300R0141 Directory chapters where this document can be found: [15.30 Health promotion] Regulation (EC) No.141/2000 of the European Parliament
REGULATION (EC) No.141/2000 Community legislation in force Document 300R0141 Directory chapters where this document can be found: [15.30 Health promotion] Regulation (EC) No.141/2000 of the European Parliament
Purpose: Policy: The Fair Hearing Plan is not applicable to mid-level providers. Grounds for a Hearing
 Subject: Fair Hearing Plan Policy #: CR-16 Department: Credentialing Approvals: Credentialing Committee QM Committee Original Effective Date: 5/00 Revised Effective Date: 1/03, 2/04, 1/05, 11/06, 12/06,
Subject: Fair Hearing Plan Policy #: CR-16 Department: Credentialing Approvals: Credentialing Committee QM Committee Original Effective Date: 5/00 Revised Effective Date: 1/03, 2/04, 1/05, 11/06, 12/06,
PHYSIOTHERAPY ACT AUTHORIZATION REGULATIONS
 c t PHYSIOTHERAPY ACT AUTHORIZATION REGULATIONS PLEASE NOTE This document, prepared by the Legislative Counsel Office, is an office consolidation of this regulation, current to July 11, 2009. It is intended
c t PHYSIOTHERAPY ACT AUTHORIZATION REGULATIONS PLEASE NOTE This document, prepared by the Legislative Counsel Office, is an office consolidation of this regulation, current to July 11, 2009. It is intended
GENERAL TERMS AND CONDITIONS WCO PUBLICATIONS
 Version 1.0 GENERAL TERMS AND CONDITIONS WCO PUBLICATIONS These General Terms and Conditions govern all sales of Publications, on paper and/or in digital format whether directly at the Sales Counter or
Version 1.0 GENERAL TERMS AND CONDITIONS WCO PUBLICATIONS These General Terms and Conditions govern all sales of Publications, on paper and/or in digital format whether directly at the Sales Counter or
THE GYMNASIUMS AND FITNESS CENTRES (REGULATION) BILL, 2016
 1 AS INTRODUCED IN LOK SABHA Bill No. 78 of 2016 5 THE GYMNASIUMS AND FITNESS CENTRES (REGULATION) BILL, 2016 By SHRI A.T. NANA PATIL, M.P. A BILL to provide for regulation of gymnasiums and fitness centres
1 AS INTRODUCED IN LOK SABHA Bill No. 78 of 2016 5 THE GYMNASIUMS AND FITNESS CENTRES (REGULATION) BILL, 2016 By SHRI A.T. NANA PATIL, M.P. A BILL to provide for regulation of gymnasiums and fitness centres
OREGON MEDICAL MARIJUANA ACT
 OREGON MEDICAL MARIJUANA ACT 475.300 Findings. The people of the state of Oregon hereby find that: (1) Patients and doctors have found marijuana to be an effective treatment for suffering caused by debilitating
OREGON MEDICAL MARIJUANA ACT 475.300 Findings. The people of the state of Oregon hereby find that: (1) Patients and doctors have found marijuana to be an effective treatment for suffering caused by debilitating
Details of Authorised Personnel
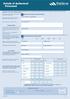 Details of Authorised Personnel This form is to be used to register authorised personnel to act on behalf of your entity via the FishServe website and through the use of paper applications, documents and
Details of Authorised Personnel This form is to be used to register authorised personnel to act on behalf of your entity via the FishServe website and through the use of paper applications, documents and
LAW ON PREVENTION AND SUPPRESSION OF THE ABUSE OF NARCOTIC DRUGS
 LAW ON PREVENTION AND SUPPRESSION OF THE ABUSE OF NARCOTIC DRUGS CHAPTER I. GENERAL PROVISIONS Article 1 (Purpose of this Law) This Law is enacted in the implementation of the Single Convention on Narcotic
LAW ON PREVENTION AND SUPPRESSION OF THE ABUSE OF NARCOTIC DRUGS CHAPTER I. GENERAL PROVISIONS Article 1 (Purpose of this Law) This Law is enacted in the implementation of the Single Convention on Narcotic
PCN-ISO GEN Requirements for the Qualification of personnel for Limited Applications of Non-destructive Testing
 Certification Services Division The Newton Building St. George s Avenue Northampton NN2 6JB Tel: +44(0)1604-893-811. Fax: +44(0)1604-893-868. E-mail: pcn@bindt.org PCN-ISO 20807-GEN Requirements for the
Certification Services Division The Newton Building St. George s Avenue Northampton NN2 6JB Tel: +44(0)1604-893-811. Fax: +44(0)1604-893-868. E-mail: pcn@bindt.org PCN-ISO 20807-GEN Requirements for the
These Rules of Membership apply in respect of all Products purchased by a Member from Sigma (and any Program Partner) on or after 1 February 2017.
 Rules of Membership 1. Introduction These Rules of Membership apply in respect of all Products purchased by a Member from Sigma (and any Program Partner) on or after 1 February 2017. The previously published
Rules of Membership 1. Introduction These Rules of Membership apply in respect of all Products purchased by a Member from Sigma (and any Program Partner) on or after 1 February 2017. The previously published
COUNCIL OF THE EUROPEAN UNION. Brussels, 7 September 2009 (OR. en) 11261/09 Interinstitutional File: 2008/0002 (COD) DENLEG 51 CODEC 893
 COUNCIL OF THE EUROPEAN UNION Brussels, 7 September 2009 (OR. en) 11261/09 Interinstitutional File: 2008/0002 (COD) DLEG 51 CODEC 893 LEGISLATIVE ACTS AND OTHER INSTRUMTS Subject: Common Position with
COUNCIL OF THE EUROPEAN UNION Brussels, 7 September 2009 (OR. en) 11261/09 Interinstitutional File: 2008/0002 (COD) DLEG 51 CODEC 893 LEGISLATIVE ACTS AND OTHER INSTRUMTS Subject: Common Position with
131. Public school enrollees' immunization program; exemptions
 TITLE 14. EDUCATION PART I. FREE PUBLIC SCHOOLS CHAPTER 1. DEPARTMENT OF EDUCATION SUBCHAPTER II. POWERS AND DUTIES 14 Del. C. 131 (2007) 131. Public school enrollees' immunization program; exemptions
TITLE 14. EDUCATION PART I. FREE PUBLIC SCHOOLS CHAPTER 1. DEPARTMENT OF EDUCATION SUBCHAPTER II. POWERS AND DUTIES 14 Del. C. 131 (2007) 131. Public school enrollees' immunization program; exemptions
The proposal affects Texas Occupations Code, Title 3, Subtitle D and Texas Administrative Code, Title 22, Part 5.
 Page 1 of 22 TITLE 22.EXAMINING BOARDS Part 5. STATE BOARD OF DENTAL EXAMINERS Chapter 101. DENTAL LICENSURE 22 TAC 101.1-101.7, 101.9 The State Board of Dental Examiners (Board) proposes amendments to
Page 1 of 22 TITLE 22.EXAMINING BOARDS Part 5. STATE BOARD OF DENTAL EXAMINERS Chapter 101. DENTAL LICENSURE 22 TAC 101.1-101.7, 101.9 The State Board of Dental Examiners (Board) proposes amendments to
Medical Devices Act 1
 Issuer: Riigikogu Type: act In force from: 01.01.2016 In force until: 31.05.2016 Translation published: 01.02.2016 Medical Devices Act 1 Amended by the following acts Passed 13.10.2004 RT I 2004, 75, 520
Issuer: Riigikogu Type: act In force from: 01.01.2016 In force until: 31.05.2016 Translation published: 01.02.2016 Medical Devices Act 1 Amended by the following acts Passed 13.10.2004 RT I 2004, 75, 520
(Acts whose publication is obligatory) REGULATION (EC) No 726/2004 OF THE EUROPEAN PARLIAMENT AND OF THE COUNCIL. of 31 March 2004
 30.4.2004 Official Journal of the European Union L 136/1 I (Acts whose publication is obligatory) REGULATION (EC) No 726/2004 OF THE EUROPEAN PARLIAMENT AND OF THE COUNCIL of 31 March 2004 laying down
30.4.2004 Official Journal of the European Union L 136/1 I (Acts whose publication is obligatory) REGULATION (EC) No 726/2004 OF THE EUROPEAN PARLIAMENT AND OF THE COUNCIL of 31 March 2004 laying down
INGHAM COUNTY. Effective January 1, 2016 as amended November 10, 2015
 INGHAM COUNTY REGULATION TO REQUIRE A LICENSE FOR THE RETAIL SALE OF ELECTROINC SMOKING DEVICES, PROHIBIT SALE OF ELECTROINC SMOKING DEVICESTO MINORS, AND TO RESTRICT LOCATION OF ELECTROINC SMOKING DEVICES
INGHAM COUNTY REGULATION TO REQUIRE A LICENSE FOR THE RETAIL SALE OF ELECTROINC SMOKING DEVICES, PROHIBIT SALE OF ELECTROINC SMOKING DEVICESTO MINORS, AND TO RESTRICT LOCATION OF ELECTROINC SMOKING DEVICES
This document is meant purely as a documentation tool and the institutions do not assume any liability for its contents
 1997R0258 EN 07.08.2009 004.001 1 This document is meant purely as a documentation tool and the institutions do not assume any liability for its contents B REGULATION (EC) No 258/97 OF THE EUROPEAN PARLIAMENT
1997R0258 EN 07.08.2009 004.001 1 This document is meant purely as a documentation tool and the institutions do not assume any liability for its contents B REGULATION (EC) No 258/97 OF THE EUROPEAN PARLIAMENT
COVENANT ON POSTGRADUATE TRAINEESHIP NECESSARY FOR ADMISSION TO THE STATE EXAMINATION FOR THE QUALIFICATION TO THE PSYCHOLOGICAL PROFESSION
 SCHOOL OF PSYCHOLOGY COVENANT ON POSTGRADUATE TRAINEESHIP NECESSARY FOR ADMISSION TO THE STATE EXAMINATION FOR THE QUALIFICATION TO THE PSYCHOLOGICAL PROFESSION (Section A of the Psychological Register
SCHOOL OF PSYCHOLOGY COVENANT ON POSTGRADUATE TRAINEESHIP NECESSARY FOR ADMISSION TO THE STATE EXAMINATION FOR THE QUALIFICATION TO THE PSYCHOLOGICAL PROFESSION (Section A of the Psychological Register
Z E N I T H M E D I C A L P R O V I D E R N E T W O R K P O L I C Y Title: Provider Appeal of Network Exclusion Policy
 TheZenith's Z E N I T H M E D I C A L P R O V I D E R N E T W O R K P O L I C Y Title: Provider Appeal of Network Exclusion Policy Application: Zenith Insurance Company and Wholly Owned Subsidiaries Policy
TheZenith's Z E N I T H M E D I C A L P R O V I D E R N E T W O R K P O L I C Y Title: Provider Appeal of Network Exclusion Policy Application: Zenith Insurance Company and Wholly Owned Subsidiaries Policy
Some of the benefits to your entity as a member of the Local Government Testing Consortium (LGTC), are as follows:
 TO: FROM: SUBJECT: Interested Parties Deborah Miner Director of Administration & Finance LGTC Program Administrator Federal DOT & Non-DOT Drug/Alcohol Testing Rules In 1993, the Oklahoma legislature passed
TO: FROM: SUBJECT: Interested Parties Deborah Miner Director of Administration & Finance LGTC Program Administrator Federal DOT & Non-DOT Drug/Alcohol Testing Rules In 1993, the Oklahoma legislature passed
RFQ:-HPCSA 03/2017 REQUEST FOR QUOTATION FOR OCCUPATIONAL HEALTH SERVICE ON BEHALF OF THE HEALTH PROFESSIONS COUNCIL OF SOUTH AFRICA
 RFQ:-HPCSA 03/2017 REQUEST FOR QUOTATION FOR OCCUPATIONAL HEALTH SERVICE ON BEHALF OF THE HEALTH PROFESSIONS COUNCIL OF SOUTH AFRICA Situated at: 553 Madiba Street, Arcadia, Pretoria Deadline for submission:
RFQ:-HPCSA 03/2017 REQUEST FOR QUOTATION FOR OCCUPATIONAL HEALTH SERVICE ON BEHALF OF THE HEALTH PROFESSIONS COUNCIL OF SOUTH AFRICA Situated at: 553 Madiba Street, Arcadia, Pretoria Deadline for submission:
Division of Registrations Gregory Ferland Interim Division Director. Corrected Notice of Proposed Rulemaking and Rulemaking Hearing
 Division of Registrations Gregory Ferland Interim Division Director State Physical Therapy Board Deann Conroy Program Director John W. H1ckenlooper Governor Barbara J. Kelley Executive Director Corrected
Division of Registrations Gregory Ferland Interim Division Director State Physical Therapy Board Deann Conroy Program Director John W. H1ckenlooper Governor Barbara J. Kelley Executive Director Corrected
PGE Energy Imbalance Market (PGE EIM BP)
 Business Practice: PGE Energy Imbalance Market (PGE EIM BP) Posted: September 30, 2017 Effective: October 1, 2017 Revision No.: 0 Table of Contents 1. Introduction... 3 2. Communication with the PGE EIM
Business Practice: PGE Energy Imbalance Market (PGE EIM BP) Posted: September 30, 2017 Effective: October 1, 2017 Revision No.: 0 Table of Contents 1. Introduction... 3 2. Communication with the PGE EIM
CHAPTER 40 PROFESSIONAL LICENSING AND FACILITY REGULATION
 216-RICR-40-05-33 TITLE 216 - DEPARTMENT OF HEALTH CHAPTER 40 PROFESSIONAL LICENSING AND FACILITY REGULATION SUBCHAPTER 05 PROFESSIONAL LICENSING PART 33 - Speech Pathologists and Audiologists 33.1 Authority
216-RICR-40-05-33 TITLE 216 - DEPARTMENT OF HEALTH CHAPTER 40 PROFESSIONAL LICENSING AND FACILITY REGULATION SUBCHAPTER 05 PROFESSIONAL LICENSING PART 33 - Speech Pathologists and Audiologists 33.1 Authority
(Legislative acts) REGULATIONS
 31.12.2010 Official Journal of the European Union L 348/1 I (Legislative acts) REGULATIONS REGULATION (EU) No 1235/2010 OF THE EUROPEAN PARLIAMENT AND OF THE COUNCIL of 15 December 2010 amending, as regards
31.12.2010 Official Journal of the European Union L 348/1 I (Legislative acts) REGULATIONS REGULATION (EU) No 1235/2010 OF THE EUROPEAN PARLIAMENT AND OF THE COUNCIL of 15 December 2010 amending, as regards
METROLINX ADMINISTRATIVE FEE DISPUTE RESOLUTION PROCESS RULES OF PRACTICE
 METROLINX ADMINISTRATIVE FEE DISPUTE RESOLUTION PROCESS RULES OF PRACTICE Overview The Metrolinx Act, 2006, gives Metrolinx ( Metrolinx ) the authority to establish a system of administrative fees to ensure
METROLINX ADMINISTRATIVE FEE DISPUTE RESOLUTION PROCESS RULES OF PRACTICE Overview The Metrolinx Act, 2006, gives Metrolinx ( Metrolinx ) the authority to establish a system of administrative fees to ensure
ORDER of the Minister of Health No. 269/2017 of 14 March 2017 on mandatory provision of medicinal product adequate and continuous supplies
 ORDER of the Minister of Health No. 269/2017 of 14 March 2017 on mandatory provision of medicinal product adequate and continuous supplies ISSUING BODY: The Ministry of Health published in: the Official
ORDER of the Minister of Health No. 269/2017 of 14 March 2017 on mandatory provision of medicinal product adequate and continuous supplies ISSUING BODY: The Ministry of Health published in: the Official
Attachment 5 2. SCOPE OF DOCUMENT
 Attachment 5 CITY MANAGER S POLICIES AND PROCEDURES FOR ADMINISTRATIVE HEARINGS CONDUCTED UNDER CHAPTER ONE OF THE SAN DIEGO MUNICIPAL CODE July 19, 2010 1. PURPOSE The purpose of this document is to establish
Attachment 5 CITY MANAGER S POLICIES AND PROCEDURES FOR ADMINISTRATIVE HEARINGS CONDUCTED UNDER CHAPTER ONE OF THE SAN DIEGO MUNICIPAL CODE July 19, 2010 1. PURPOSE The purpose of this document is to establish
e-cigarette Regulation
 e-cigarette Regulation The Act prohibits the sale of electronic smoking devices and alternative nicotine products to minors, and requires child-resistant packaging for liquid nicotine containers. The Act
e-cigarette Regulation The Act prohibits the sale of electronic smoking devices and alternative nicotine products to minors, and requires child-resistant packaging for liquid nicotine containers. The Act
IPC Athletics Classification Rules and Regulations
 IPC Athletics Classification Rules and Regulations February 2013 International Paralympic Committee Adenauerallee 212-214 Tel. +49 228 2097-200 www.paralympic.org 53113 Bonn, Germany Fax +49 228 2097-209
IPC Athletics Classification Rules and Regulations February 2013 International Paralympic Committee Adenauerallee 212-214 Tel. +49 228 2097-200 www.paralympic.org 53113 Bonn, Germany Fax +49 228 2097-209
NAPPO Executive Committee Decision Sheet
 D No. 6 Issue: NAPPO Executive Committee Decision Sheet A Dispute Settlement Mechanism for NAPPO countries Background: In order to resolve phytosanitary disputes among NAPPO member countries, a mechanism
D No. 6 Issue: NAPPO Executive Committee Decision Sheet A Dispute Settlement Mechanism for NAPPO countries Background: In order to resolve phytosanitary disputes among NAPPO member countries, a mechanism
This license is required for any businesses offering tobacco products for sale.
 Guidelines for City of Moorhead 500 Center Avenue, PO Box 779 Moorhead, MN 56560-0799 Phone: 218.299.5304 Fax: 218.299.5306 cityclerk@ci.moorhead.mn.us Moorhead City Code, 2-5A OVERVIEW This license is
Guidelines for City of Moorhead 500 Center Avenue, PO Box 779 Moorhead, MN 56560-0799 Phone: 218.299.5304 Fax: 218.299.5306 cityclerk@ci.moorhead.mn.us Moorhead City Code, 2-5A OVERVIEW This license is
Strategic Environmental Assessment Implementation Step by Step!
 Strategic Environmental Assessment Implementation Step by Step! 2 Brochure Strategic Environmental Assessment Implementation Step by Step! Dear readers, We have designed this Brochure in order to introduce
Strategic Environmental Assessment Implementation Step by Step! 2 Brochure Strategic Environmental Assessment Implementation Step by Step! Dear readers, We have designed this Brochure in order to introduce
(4) Be as detailed as necessary to provide history of work performed; and:
 www.omarfigueroa.com Page 66 of 278 (4) Be as detailed as necessary to provide history of work performed; and: (A) Include information adequate to identify any associated manufacturing facility (e.g.,
www.omarfigueroa.com Page 66 of 278 (4) Be as detailed as necessary to provide history of work performed; and: (A) Include information adequate to identify any associated manufacturing facility (e.g.,
Appendix C Resolution of a Complaint against an Employee
 Appendix C Resolution of a Complaint against an Employee Appendix C: Resolution of a Complaint Against an Employee As outlined in the Union College Sexual Misconduct Policy, an individual who wishes to
Appendix C Resolution of a Complaint against an Employee Appendix C: Resolution of a Complaint Against an Employee As outlined in the Union College Sexual Misconduct Policy, an individual who wishes to
EUR-Lex L EN. Council Directive 93/74/EEC of 13 September 1993 on feedingstuffs intended for particular nutritional purposes
 Page 1 of 5 Avis juridique important 31993L0074 Council Directive 93/74/EEC of 13 September 1993 on feedingstuffs intended for particular nutritional purposes Official Journal L 237, 22/09/1993 P. 0023-0027
Page 1 of 5 Avis juridique important 31993L0074 Council Directive 93/74/EEC of 13 September 1993 on feedingstuffs intended for particular nutritional purposes Official Journal L 237, 22/09/1993 P. 0023-0027
Chapter TOBACCO RETAILER'S PERMIT
 Sections: 8.60.010 - Definitions. 8.60.020 - Requirements for Tobacco Retailer's Permit. 8.60.030 - Application procedure. 8.60.040 - Issuance of permit. 8.60.050 - Display of permit. 8.60.060 - Fees for
Sections: 8.60.010 - Definitions. 8.60.020 - Requirements for Tobacco Retailer's Permit. 8.60.030 - Application procedure. 8.60.040 - Issuance of permit. 8.60.050 - Display of permit. 8.60.060 - Fees for
POL HR CDL DRUG AND ALCOHOL TESTING PLAN Page 1 of 8 POLICY. See Also: POL-0409-HR; PRO HR; PRO HR Res
 POL -0410-HR CDL DRUG AND ALCOHOL TESTING PLAN Page 1 of 8 Effective Date: 12/7/09 POLICY Cancels: # 069526 Approved by: See Also: POL-0409-HR; PRO-0410-1-HR; PRO-0410-2-HR Res. 070124 POL -0410-HR Document
POL -0410-HR CDL DRUG AND ALCOHOL TESTING PLAN Page 1 of 8 Effective Date: 12/7/09 POLICY Cancels: # 069526 Approved by: See Also: POL-0409-HR; PRO-0410-1-HR; PRO-0410-2-HR Res. 070124 POL -0410-HR Document
SOUTH DAKOTA BOARD OF REGENTS. Policy Manual
 SUBJECT: Drug Free Environment NUMBER: 4:27 SOUTH DAKOTA BOARD OF REGENTS Policy Manual Drug Free Workplace Policy The South Dakota Board of Regents is committed to providing a drug free workplace. Additional
SUBJECT: Drug Free Environment NUMBER: 4:27 SOUTH DAKOTA BOARD OF REGENTS Policy Manual Drug Free Workplace Policy The South Dakota Board of Regents is committed to providing a drug free workplace. Additional
Minister of Social Affairs. Passed No. 73 RTL 2005, 57, 807 Entry into force
 Issuer: Minister of Social Affairs Type: regulation In force from: 12.02.2011 In force until: Did not enter into force Translation published: 08.10.2014 Conditions and Procedure for Handling of Narcotic
Issuer: Minister of Social Affairs Type: regulation In force from: 12.02.2011 In force until: Did not enter into force Translation published: 08.10.2014 Conditions and Procedure for Handling of Narcotic
THE GENERAL ASSEMBLY OF PENNSYLVANIA SENATE BILL AN ACT
 PRINTER'S NO. THE GENERAL ASSEMBLY OF PENNSYLVANIA SENATE BILL No. Session of INTRODUCED BY LEACH AND FARNESE, FEBRUARY, REFERRED TO LAW AND JUSTICE, FEBRUARY, AN ACT 1 1 1 Providing for personal use of
PRINTER'S NO. THE GENERAL ASSEMBLY OF PENNSYLVANIA SENATE BILL No. Session of INTRODUCED BY LEACH AND FARNESE, FEBRUARY, REFERRED TO LAW AND JUSTICE, FEBRUARY, AN ACT 1 1 1 Providing for personal use of
AN ACT. relating to the medical use of low-thc cannabis and the regulation. of related organizations and individuals; requiring a dispensing
 Chapter 0 S.B. No. AN ACT relating to the medical use of low-thc cannabis and the regulation of related organizations and individuals; requiring a dispensing organization to obtain a license to dispense
Chapter 0 S.B. No. AN ACT relating to the medical use of low-thc cannabis and the regulation of related organizations and individuals; requiring a dispensing organization to obtain a license to dispense
Act 443 of 2009 House Bill 1379
 Act 443 of 2009 House Bill 1379 AN ACT TO PROVIDE FOR THE LICENSURE OF ALCOHOLISM AND DRUG ABUSE COUNSELORS; TO PROVIDE FOR THE REGISTRATION OF CLINICAL ALCOHOLISM AND DRUG ABUSE COUNSELOR SUPERVISORS;
Act 443 of 2009 House Bill 1379 AN ACT TO PROVIDE FOR THE LICENSURE OF ALCOHOLISM AND DRUG ABUSE COUNSELORS; TO PROVIDE FOR THE REGISTRATION OF CLINICAL ALCOHOLISM AND DRUG ABUSE COUNSELOR SUPERVISORS;
77th OREGON LEGISLATIVE ASSEMBLY Regular Session. Enrolled. House Bill 3460
 77th OREGON LEGISLATIVE ASSEMBLY--2013 Regular Session Enrolled House Bill 3460 Sponsored by Representative BUCKLEY, Senator PROZANSKI; Representative FREDERICK, Senator DINGFELDER CHAPTER... AN ACT Relating
77th OREGON LEGISLATIVE ASSEMBLY--2013 Regular Session Enrolled House Bill 3460 Sponsored by Representative BUCKLEY, Senator PROZANSKI; Representative FREDERICK, Senator DINGFELDER CHAPTER... AN ACT Relating
78th OREGON LEGISLATIVE ASSEMBLY Regular Session. Senate Bill 964 SUMMARY
 Sponsored by Senators BURDICK, KRUSE th OREGON LEGISLATIVE ASSEMBLY-- Regular Session Senate Bill SUMMARY The following summary is not prepared by the sponsors of the measure and is not a part of the body
Sponsored by Senators BURDICK, KRUSE th OREGON LEGISLATIVE ASSEMBLY-- Regular Session Senate Bill SUMMARY The following summary is not prepared by the sponsors of the measure and is not a part of the body
SECTION PRESCRIPTIONS
 SECTION.1800 - PRESCRIPTIONS 21 NCAC 46.1801 EXERCISE OF PROFESSIONAL JUDGMENT IN FILLING PRESCRIPTIONS (a) A pharmacist or device and medical equipment dispenser shall have a right to refuse to fill or
SECTION.1800 - PRESCRIPTIONS 21 NCAC 46.1801 EXERCISE OF PROFESSIONAL JUDGMENT IN FILLING PRESCRIPTIONS (a) A pharmacist or device and medical equipment dispenser shall have a right to refuse to fill or
IC ARTICLE 13. DENTAL HYGIENISTS. IC Chapter 1. Regulation of Dental Hygienists by State Board of Dentistry
 IC 25-13 ARTICLE 13. DENTAL HYGIENISTS IC 25-13-1 Chapter 1. Regulation of Dental Hygienists by State Board of Dentistry IC 25-13-1-1 Short title Sec. 1. This chapter may be known and cited as The Dental
IC 25-13 ARTICLE 13. DENTAL HYGIENISTS IC 25-13-1 Chapter 1. Regulation of Dental Hygienists by State Board of Dentistry IC 25-13-1-1 Short title Sec. 1. This chapter may be known and cited as The Dental
Rules of Procedure for Screening and Hearing Meetings
 Page: 1 of 15 SYNOPSIS: The purpose of this document is to provide rules of procedure for Screening and Hearing meetings conducted pursuant to the City s Parking Administrative Monetary Penalties By-law
Page: 1 of 15 SYNOPSIS: The purpose of this document is to provide rules of procedure for Screening and Hearing meetings conducted pursuant to the City s Parking Administrative Monetary Penalties By-law
(City, State, Zip Code)
 This Partner Agency Agreement, dated this day of, 2015, is between COMMUNITY FOOD SHARE, INC. (CFS), whose address is 650 South Taylor Avenue, Louisville, CO 80027, and (Partner Agency) whose address is
This Partner Agency Agreement, dated this day of, 2015, is between COMMUNITY FOOD SHARE, INC. (CFS), whose address is 650 South Taylor Avenue, Louisville, CO 80027, and (Partner Agency) whose address is
Medicinal Plants Act. Chapter One GENERAL PROVISIONS
 Medicinal Plants Act Promulgated, State Gazette No. 29/7.04.2000, amended, SG No. 23/1.03.2002, SG No. 91/25.09.2002, effective 1.01.2003, SG No. 30/11.04.2006, effective 12.07.2006, SG No. 65/11.08.2006,
Medicinal Plants Act Promulgated, State Gazette No. 29/7.04.2000, amended, SG No. 23/1.03.2002, SG No. 91/25.09.2002, effective 1.01.2003, SG No. 30/11.04.2006, effective 12.07.2006, SG No. 65/11.08.2006,
ANNEXURE A SUPPLIER DECLARATION FORM P a g e
 ANNEXURE A SUPPLIER DECLARATION FORM - 1 - P a g e Health Professions Council of South Africa This form must be completed and submitted with TENDER: Health Professions Council of South Africa P O Box 205
ANNEXURE A SUPPLIER DECLARATION FORM - 1 - P a g e Health Professions Council of South Africa This form must be completed and submitted with TENDER: Health Professions Council of South Africa P O Box 205
Drug and Alcohol Policy
 CDL Program Drug and Alcohol Policy Effective as of [10/08/2018] Adopted by: Date Adopted: [10/08/2018] Last Revised: [10/08/2018] Table of Contents 1. Purpose of Policy... 3 2. Covered Employees... 3
CDL Program Drug and Alcohol Policy Effective as of [10/08/2018] Adopted by: Date Adopted: [10/08/2018] Last Revised: [10/08/2018] Table of Contents 1. Purpose of Policy... 3 2. Covered Employees... 3
DELTA DENTAL PREMIER
 DELTA DENTAL PREMIER PARTICIPATING DENTIST AGREEMENT THIS AGREEMENT made and entered into this day of, 20 by and between Colorado Dental Service, Inc. d/b/a Delta Dental of Colorado, as first party, hereinafter
DELTA DENTAL PREMIER PARTICIPATING DENTIST AGREEMENT THIS AGREEMENT made and entered into this day of, 20 by and between Colorado Dental Service, Inc. d/b/a Delta Dental of Colorado, as first party, hereinafter
IMPORTANT DISCLAIMER. Note
 yn EUROPEAN COMMISSION HEALTH AND CONSUMERS DIRECTORATE-GENERAL June 2012 DRAFT GUIDANCE DOCUMENT FOR COMPETENT AUTHORITIES FOR THE CONTROL OF COMPLIANCE WITH EU LEGISLATION ON: Regulation (EU) No 1169/2011
yn EUROPEAN COMMISSION HEALTH AND CONSUMERS DIRECTORATE-GENERAL June 2012 DRAFT GUIDANCE DOCUMENT FOR COMPETENT AUTHORITIES FOR THE CONTROL OF COMPLIANCE WITH EU LEGISLATION ON: Regulation (EU) No 1169/2011
XOSERVE LIMITED SERVICES SCHEDULE FOR THE PROVISION OF NON-CODE USER PAYS SERVICES (REFERENCE NUMBER XNCUP(SS)06) DATED 20 INTRODUCTION
 XOSERVE LIMITED SERVICES SCHEDULE FOR THE PROVISION OF NON-CODE USER PAYS SERVICES (REFERENCE NUMBER XNCUP(SS)06) DATED 20 INTRODUCTION 1 This services schedule forms part of the framework contract for
XOSERVE LIMITED SERVICES SCHEDULE FOR THE PROVISION OF NON-CODE USER PAYS SERVICES (REFERENCE NUMBER XNCUP(SS)06) DATED 20 INTRODUCTION 1 This services schedule forms part of the framework contract for
Lao People s Democratic Republic
 Authentic in Lao Only Lao People s Democratic Republic Peace Independence Democracy Unity Prosperity Ministry of Mines and Energy Ref. 1116/MEM Vientiane Capital, dated 21.09.2010 Regulation on Sending
Authentic in Lao Only Lao People s Democratic Republic Peace Independence Democracy Unity Prosperity Ministry of Mines and Energy Ref. 1116/MEM Vientiane Capital, dated 21.09.2010 Regulation on Sending
Policies, Procedures and Guidelines
 Policies, Procedures and Guidelines Complete Policy Title: Faculty Grievance Review Panel Guidelines for Hearing Committees Approved by: Faculty Grievance Review Panel Date of Original Approval(s): Policy
Policies, Procedures and Guidelines Complete Policy Title: Faculty Grievance Review Panel Guidelines for Hearing Committees Approved by: Faculty Grievance Review Panel Date of Original Approval(s): Policy
Annex A TITLE 49. PROFESSIONAL AND VOCATIONAL STANDARDS PART I. DEPARTMENT OF STATE. Subpart A. PROFESSIONAL AND OCCUPATIONAL AFFAIRS
 Annex A 1 TITLE 49. PROFESSIONAL AND VOCATIONAL STANDARDS PART I. DEPARTMENT OF STATE Subpart A. PROFESSIONAL AND OCCUPATIONAL AFFAIRS CHAPTER 16. STATE BOARD OF MEDICINE GENERAL PROVISIONS 16.1. Definitions.
Annex A 1 TITLE 49. PROFESSIONAL AND VOCATIONAL STANDARDS PART I. DEPARTMENT OF STATE Subpart A. PROFESSIONAL AND OCCUPATIONAL AFFAIRS CHAPTER 16. STATE BOARD OF MEDICINE GENERAL PROVISIONS 16.1. Definitions.
TERMS AND CONDITIONS NATIONAL GOOD LABORATORY PRACTICE (GLP) COMPLIANCE MONITORING AUTHORITY
 TERMS AND CONDITIONS OF NATIONAL GOOD LABORATORY PRACTICE (GLP) COMPLIANCE MONITORING AUTHORITY FOR OBTAINING AND MAINTAINING ITS GLP CERTIFICATION BY A TEST FACILITY Document No.GLP-101 Version/Issue
TERMS AND CONDITIONS OF NATIONAL GOOD LABORATORY PRACTICE (GLP) COMPLIANCE MONITORING AUTHORITY FOR OBTAINING AND MAINTAINING ITS GLP CERTIFICATION BY A TEST FACILITY Document No.GLP-101 Version/Issue
WHEREAS, the Tennessee General Assembly finds that thousands of Tennesseans are
 AN ACT to license sign language interpreters WHEREAS, the Tennessee General Assembly finds that thousands of Tennesseans are individuals who are Deaf, Deaf-Blind, or Hard of Hearing; and WHEREAS, the General
AN ACT to license sign language interpreters WHEREAS, the Tennessee General Assembly finds that thousands of Tennesseans are individuals who are Deaf, Deaf-Blind, or Hard of Hearing; and WHEREAS, the General
GENERAL INFORMATION AND INSTRUCTIONS
 NON-PARTICIPATING MANUFACTURER CERTIFICATION FOR LISTING ON OREGON DIRECTORY GENERAL INFORMATION AND INSTRUCTIONS Who is required to file this Certification? Any tobacco product manufacturer who is a non-participating
NON-PARTICIPATING MANUFACTURER CERTIFICATION FOR LISTING ON OREGON DIRECTORY GENERAL INFORMATION AND INSTRUCTIONS Who is required to file this Certification? Any tobacco product manufacturer who is a non-participating
Regulation of the Chancellor
 Regulation of the Chancellor Category: STUDENTS Issued: 6/22/09 Number: A-450 Subject: INVOLUNTARY TRANSFER PROCEDURES Page: 1 of 1 SUMMARY OF CHANGES This regulation supersedes Chancellor s Regulation
Regulation of the Chancellor Category: STUDENTS Issued: 6/22/09 Number: A-450 Subject: INVOLUNTARY TRANSFER PROCEDURES Page: 1 of 1 SUMMARY OF CHANGES This regulation supersedes Chancellor s Regulation
Appendix C NEWBORN HEARING SCREENING PROJECT
 Appendix C NEWBORN HEARING SCREENING PROJECT I. WEST VIRGINIA STATE LAW All newborns born in the State of West Virginia must be screened for hearing impairment as required in WV Code 16-22A and 16-1-7,
Appendix C NEWBORN HEARING SCREENING PROJECT I. WEST VIRGINIA STATE LAW All newborns born in the State of West Virginia must be screened for hearing impairment as required in WV Code 16-22A and 16-1-7,
Certification in Lower Extremity Geriatric Medicine Handbook
 Certification in Lower Extremity Geriatric Medicine Handbook 555 8 th Ave, Ste 1902, New York, NY 10018 888 852 1442 1 Mission Statement We exist to protect and improve the podiatric health and welfare
Certification in Lower Extremity Geriatric Medicine Handbook 555 8 th Ave, Ste 1902, New York, NY 10018 888 852 1442 1 Mission Statement We exist to protect and improve the podiatric health and welfare
Regulations. On Proper Conduct in Research TEL AVIV UNIVERSITY
 Regulations On Proper Conduct in Research TEL AVIV UNIVERSITY 1. Preamble: Tel Aviv University aspires to excellence in research. Excellence is not gauged solely according to research results. It depends
Regulations On Proper Conduct in Research TEL AVIV UNIVERSITY 1. Preamble: Tel Aviv University aspires to excellence in research. Excellence is not gauged solely according to research results. It depends
EDICT ON PROMULGATION OF THE TOBACCO LAW TOBACCO LAW
 "Official Gazette of the Republic of Montenegro", Nos. 80/2004, 5/2005 By virtue of Article 88 item 2 of the Constitution of the Republic of Montenegro, I herewith enact this EDICT ON PROMULGATION OF THE
"Official Gazette of the Republic of Montenegro", Nos. 80/2004, 5/2005 By virtue of Article 88 item 2 of the Constitution of the Republic of Montenegro, I herewith enact this EDICT ON PROMULGATION OF THE
PRINCE GEORGE S COUNTY PUBLIC SCHOOLS Board of Education Upper Marlboro, Maryland Policy No. BOARD OF EDUCATION POLICY
 PRINCE GEORGE S COUNTY PUBLIC SCHOOLS Board of Education Upper Marlboro, Maryland BOARD OF EDUCATION POLICY 4200 Policy No. PERSONNEL Employee and 4-205 Appeals Before the Board of Education I. Purpose
PRINCE GEORGE S COUNTY PUBLIC SCHOOLS Board of Education Upper Marlboro, Maryland BOARD OF EDUCATION POLICY 4200 Policy No. PERSONNEL Employee and 4-205 Appeals Before the Board of Education I. Purpose
The Cannabis Control (Saskatchewan) Regulations
 CANNABIS CONTROL (SASKATCHEWAN) C-2.111 REG 1 1 The Cannabis Control (Saskatchewan) Regulations being Chapter C-2.111 Reg 1 (effective October 17, 2018). NOTE: This consolidation is not official. Amendments
CANNABIS CONTROL (SASKATCHEWAN) C-2.111 REG 1 1 The Cannabis Control (Saskatchewan) Regulations being Chapter C-2.111 Reg 1 (effective October 17, 2018). NOTE: This consolidation is not official. Amendments
Law of the Republic of Azerbaijan on the Control of Illicit Trafficking in Narcotic Drugs, Psychotropic Substances and Precursors
 Law of the Republic of Azerbaijan on the Control of Illicit Trafficking in Narcotic Drugs, Psychotropic Substances and Precursors This Law, which stresses the danger of offences associated with illegal
Law of the Republic of Azerbaijan on the Control of Illicit Trafficking in Narcotic Drugs, Psychotropic Substances and Precursors This Law, which stresses the danger of offences associated with illegal
City of Carson 701 E. Carson St., Carson, CA Telephone: (310) ; ci.carson.ca.us
 OFFICE USE ONLY Case No. City of Carson 701 E. Carson St., Carson, CA 90745 Telephone: (310) 830-7600; ci.carson.ca.us Application Submittal Date Fee Accepted By SUPPLEMENTAL APPLICATION FOR COMMERCIAL
OFFICE USE ONLY Case No. City of Carson 701 E. Carson St., Carson, CA 90745 Telephone: (310) 830-7600; ci.carson.ca.us Application Submittal Date Fee Accepted By SUPPLEMENTAL APPLICATION FOR COMMERCIAL
Adopted by the State Duma on July 8, 2005 Approved by the Federation Council on July 13, 2005
 FEDERAL LAW NO. 102-FZ OF JULY 21, 2005 ON AMENDING THE FEDERAL LAW ON THE STATE REGULATION OF PRODUCTION AND CIRCULATION OF ETHYL ALCOHOL, ALCOHOLIC PRODUCTS AND ALCOHOL-CONTAINING PRODUCTS AND ON DECLARING
FEDERAL LAW NO. 102-FZ OF JULY 21, 2005 ON AMENDING THE FEDERAL LAW ON THE STATE REGULATION OF PRODUCTION AND CIRCULATION OF ETHYL ALCOHOL, ALCOHOLIC PRODUCTS AND ALCOHOL-CONTAINING PRODUCTS AND ON DECLARING
This is an unofficial English translation of the Royal Decree 1344/2007 revised by the Spanish Agency for Medicines and Medical Devices
 agencia española de medicamentos y productos sanitarios This is an unofficial English translation of the Royal Decree 1344/2007 revised by the Spanish Agency for Medicines and Medical Devices ROYAL DECREE
agencia española de medicamentos y productos sanitarios This is an unofficial English translation of the Royal Decree 1344/2007 revised by the Spanish Agency for Medicines and Medical Devices ROYAL DECREE
HILLSBOROUGH COUNTY AVIATION AUTHORITY AIRPORT BOARD OF ADJUSTMENT RULES OF PROCEDURE
 HILLSBOROUGH COUNTY AVIATION AUTHORITY AIRPORT BOARD OF ADJUSTMENT RULES OF PROCEDURE PURPOSE AND AUTHORITY Adopted May 6, 2010 Revised June 2, 2016 The Hillsborough County Aviation Authority Airport Board
HILLSBOROUGH COUNTY AVIATION AUTHORITY AIRPORT BOARD OF ADJUSTMENT RULES OF PROCEDURE PURPOSE AND AUTHORITY Adopted May 6, 2010 Revised June 2, 2016 The Hillsborough County Aviation Authority Airport Board
Article XIV: MINIMUM CONTINUING EDUCATION FOR DENTISTS AND DENTAL HYGIENISTS
 AMENDMENT MARKUP Article XIV: MINIMUM CONTINUING EDUCATION FOR DENTISTS AND DENTAL HYGIENISTS A. Purpose: The Rules in this chapter set forth the requirements and guidelines for minimum continuing education
AMENDMENT MARKUP Article XIV: MINIMUM CONTINUING EDUCATION FOR DENTISTS AND DENTAL HYGIENISTS A. Purpose: The Rules in this chapter set forth the requirements and guidelines for minimum continuing education
CUSTOMS, EXCISE TARIFF, ETC. (CONSOLIDATION) (AMENDMENT) ACT NO
 213 EDITION CUSTOMS, EXCISE TARIFF, ETC. (CONSOLIDATION) (AMENDMENT) ACT NO. 16 1997 THE Federal Government here decrees as follows:- Amendment of 1995 No. 4. 1996 No. 13. 1. The Customs, Excise Tariff,
213 EDITION CUSTOMS, EXCISE TARIFF, ETC. (CONSOLIDATION) (AMENDMENT) ACT NO. 16 1997 THE Federal Government here decrees as follows:- Amendment of 1995 No. 4. 1996 No. 13. 1. The Customs, Excise Tariff,
Please find enclosed Dosimetry Service Licence No which replaces Dosimetry Service Licence No
 Directorate of Nuclear Substance Regulation Telephone: 1 (888) 229-2672 April 12, 2017 Licence Number Christopher Passmore Landauer Inc. 2 Science Road Glenwood, IL 60425-1586 Subject: Dosimetry Service
Directorate of Nuclear Substance Regulation Telephone: 1 (888) 229-2672 April 12, 2017 Licence Number Christopher Passmore Landauer Inc. 2 Science Road Glenwood, IL 60425-1586 Subject: Dosimetry Service
DIRECTIVE 2004/24/EC OF THE EUROPEAN PARLIAMENT AND OF THE COUNCIL. of 31 March 2004
 30.4.2004 Official Journal of the European Union L 136/85 DIRECTIVE 2004/24/EC OF THE EUROPEAN PARLIAMENT AND OF THE COUNCIL of 31 March 2004 amending, as regards traditional herbal medicinal products,
30.4.2004 Official Journal of the European Union L 136/85 DIRECTIVE 2004/24/EC OF THE EUROPEAN PARLIAMENT AND OF THE COUNCIL of 31 March 2004 amending, as regards traditional herbal medicinal products,
Guidance for Municipalities Regarding Marijuana for Adult Use January 2018
 Guidance for Municipalities Regarding Marijuana for Adult Use January 2018 The following information is provided to assist municipalities by addressing questions related to the regulation of marijuana
Guidance for Municipalities Regarding Marijuana for Adult Use January 2018 The following information is provided to assist municipalities by addressing questions related to the regulation of marijuana
Recall Guidelines. for Chinese Medicine Products
 Recall Guidelines for Chinese Medicine Products April 2018 Recall Guidelines for Chinese Medicine Products Chinese Medicines Board Chinese Medicine Council of Hong Kong Compiled in September 2005 1 st
Recall Guidelines for Chinese Medicine Products April 2018 Recall Guidelines for Chinese Medicine Products Chinese Medicines Board Chinese Medicine Council of Hong Kong Compiled in September 2005 1 st
Title 32: PROFESSIONS AND OCCUPATIONS
 Title 32: PROFESSIONS AND OCCUPATIONS Chapter 45-A: PHYSICAL THERAPIST PRACTICE ACT Table of Contents Section 3111. DEFINITIONS... 3 Section 3111-A. SCOPE OF PRACTICE... 3 Section 3112. BOARD CREATED;
Title 32: PROFESSIONS AND OCCUPATIONS Chapter 45-A: PHYSICAL THERAPIST PRACTICE ACT Table of Contents Section 3111. DEFINITIONS... 3 Section 3111-A. SCOPE OF PRACTICE... 3 Section 3112. BOARD CREATED;
PROPOSED REGULATION OF THE BOARD OF HEARING AID SPECIALISTS. LCB File No. R July 6, 2001
 PROPOSED REGULATION OF THE BOARD OF HEARING AID SPECIALISTS LCB File No. R062-01 July 6, 2001 EXPLANATION Matter in italics is new; matter in brackets [omitted material] is material to be omitted. AUTHORITY:
PROPOSED REGULATION OF THE BOARD OF HEARING AID SPECIALISTS LCB File No. R062-01 July 6, 2001 EXPLANATION Matter in italics is new; matter in brackets [omitted material] is material to be omitted. AUTHORITY:
CLIENT PROCEDURE FOR ANNUAL APPROVAL OF SHIP REPAIR COMPANIES
 CLIENT PROCEDURE FOR ANNUAL APPROVAL OF SHIP REPAIR COMPANIES 1.0 PURPOSE Safe work environment is essential for performing all kinds of ship repair operations. In this regard and to enable safe and smooth
CLIENT PROCEDURE FOR ANNUAL APPROVAL OF SHIP REPAIR COMPANIES 1.0 PURPOSE Safe work environment is essential for performing all kinds of ship repair operations. In this regard and to enable safe and smooth
Volume of Colorado Commission for the Deaf and Hard of Hearing (12 CCR )
 Disclaimer: On-line versions of these regulations are the most current versions available; however, these are not official publication. For official publication of these and all State of Colorado regulations,
Disclaimer: On-line versions of these regulations are the most current versions available; however, these are not official publication. For official publication of these and all State of Colorado regulations,
(Tentative Translation)
 (Tentative Translation) Public Notice of the Ministry of Education, Culture, Sports, Science and Technology (MEXT) and the Ministry of Health, Labour and Welfare (MHLW) No. 2 of December 17, 2010 Table
(Tentative Translation) Public Notice of the Ministry of Education, Culture, Sports, Science and Technology (MEXT) and the Ministry of Health, Labour and Welfare (MHLW) No. 2 of December 17, 2010 Table
DIRECTIVE January 13, 2010 D Revision DP&P Narcotics Medical Marijuana
 Seattle Police Department DIRECTIVE An Accredited Law Enforcement Agency January 13, 2010 D 10-001 Revision DP&P 15.150-Narcotics Medical Marijuana DP&P section 15.150 (Preliminary Investigations-Narcotics)
Seattle Police Department DIRECTIVE An Accredited Law Enforcement Agency January 13, 2010 D 10-001 Revision DP&P 15.150-Narcotics Medical Marijuana DP&P section 15.150 (Preliminary Investigations-Narcotics)
Alberta - US Comparator: Standard-Making and Enforcement Functions
 Alberta - US Comparator: Standard-Making and Enforcement Functions Current Reliability Standards Below is a link to current reliability standards in Alberta: http://www.aeso.ca/rulesprocedures/17006.html
Alberta - US Comparator: Standard-Making and Enforcement Functions Current Reliability Standards Below is a link to current reliability standards in Alberta: http://www.aeso.ca/rulesprocedures/17006.html
Section 32: BIMM Institute Student Disciplinary Procedure
 Section 32: BIMM Institute Student Disciplinary Procedure Introduction Academic Development & Quality Assurance Manual This Student Disciplinary Procedure provides a framework for the regulation of BIMM
Section 32: BIMM Institute Student Disciplinary Procedure Introduction Academic Development & Quality Assurance Manual This Student Disciplinary Procedure provides a framework for the regulation of BIMM
on the advertising of medicinal products for human use
 30. 4. 92 Official Journal of the European Communities No L 113 / 13 COUNCIL DIRECTIVE 92/28/EEC of 31 March 1992 on the advertising of medicinal products for human use THE COUNCIL OF THE EUROPEAN COMMUNITIES,
30. 4. 92 Official Journal of the European Communities No L 113 / 13 COUNCIL DIRECTIVE 92/28/EEC of 31 March 1992 on the advertising of medicinal products for human use THE COUNCIL OF THE EUROPEAN COMMUNITIES,
SPECIAL DISCLAIMER FOR INTERPRETING SERVICES INVOLVING CALLS TO EMERGENCY SERVICE PROVIDERS (911/E911), OR LEGAL, MEDICAL OR MENTAL HEALTH ISSUES
 SPECIAL DISCLAIMER FOR INTERPRETING SERVICES INVOLVING CALLS TO EMERGENCY SERVICE PROVIDERS (911/E911), OR LEGAL, MEDICAL OR MENTAL HEALTH ISSUES Governing Language. The English language version of this
SPECIAL DISCLAIMER FOR INTERPRETING SERVICES INVOLVING CALLS TO EMERGENCY SERVICE PROVIDERS (911/E911), OR LEGAL, MEDICAL OR MENTAL HEALTH ISSUES Governing Language. The English language version of this
Criteria and Application for Men
 Criteria and Application for Men Return completed form via fax or email to LIVESTRONG Foundation attn LIVESTRONG Fertility Fax 512.309.5515 email Cancer.Navigation@LIVESTRONG.org Made possible by participating
Criteria and Application for Men Return completed form via fax or email to LIVESTRONG Foundation attn LIVESTRONG Fertility Fax 512.309.5515 email Cancer.Navigation@LIVESTRONG.org Made possible by participating
DENTAL HYGIENE LICENSURE BY CREDENTIALS
 LOUISIANA STATE BOARD OF DENTISTRY 365 CANAL PLACE, SUITE 2680 NEW ORLEANS, LOUISIANA 70130 PHONE: 504-568-8574 ~ FAX: 504-568-8598 www.lsbd.org DENTAL HYGIENE LICENSURE BY CREDENTIALS Applying for a license
LOUISIANA STATE BOARD OF DENTISTRY 365 CANAL PLACE, SUITE 2680 NEW ORLEANS, LOUISIANA 70130 PHONE: 504-568-8574 ~ FAX: 504-568-8598 www.lsbd.org DENTAL HYGIENE LICENSURE BY CREDENTIALS Applying for a license
BERMUDA DENTAL TECHNICIANS REGULATIONS 1962 BX 6 / 1962
 QUO FA T A F U E R N T BERMUDA DENTAL TECHNICIANS REGULATIONS 1962 BX 6 / 1962 TABLE OF CONTENTS 1 2 3 4 5 6 7 8 9 10 11 12 15 Citation Interpretation Unqualified persons; offences Register of dental technicians
QUO FA T A F U E R N T BERMUDA DENTAL TECHNICIANS REGULATIONS 1962 BX 6 / 1962 TABLE OF CONTENTS 1 2 3 4 5 6 7 8 9 10 11 12 15 Citation Interpretation Unqualified persons; offences Register of dental technicians
(No. 349) (Approved September 2, 2000) AN ACT. To create and establish the Bill of Rights for Carriers of the HIV/AIDS Virus in Puerto Rico.
 (H.B. 2980) (Conference) (No. 349) (Approved September 2, 2000) AN ACT To create and establish the Bill of Rights for Carriers of the HIV/AIDS Virus in Puerto Rico. STATEMENT OF MOTIVES The HIV virus has
(H.B. 2980) (Conference) (No. 349) (Approved September 2, 2000) AN ACT To create and establish the Bill of Rights for Carriers of the HIV/AIDS Virus in Puerto Rico. STATEMENT OF MOTIVES The HIV virus has
(A) results from that individual's participation in or training for sports, fitness training, or other athletic competition; or
 VT AT Act 12/04 Title 26: Professions and Occupations Chapter 83: ATHLETIC TRAINERS 4151. Definitions As used in this chapter: (1) "Athlete" means any individual participating in fitness training and conditioning,
VT AT Act 12/04 Title 26: Professions and Occupations Chapter 83: ATHLETIC TRAINERS 4151. Definitions As used in this chapter: (1) "Athlete" means any individual participating in fitness training and conditioning,
Home Sleep Test (HST) Instructions
 Home Sleep Test (HST) Instructions 1. Your physician has ordered an unattended home sleep test (HST) to diagnose or rule out sleep apnea. This test cannot diagnose any other sleep disorders. 2. This device
Home Sleep Test (HST) Instructions 1. Your physician has ordered an unattended home sleep test (HST) to diagnose or rule out sleep apnea. This test cannot diagnose any other sleep disorders. 2. This device
New York Certified Peer Specialist
 New York Certified Peer Specialist PROVISIONAL Application New York Peer Specialist Certification Board 11 North Pearl Street, Suite 801 Albany New York 12207 Phone: 518.426.0945 Fax: 518.426.1046 www.nypeerspecialist.org
New York Certified Peer Specialist PROVISIONAL Application New York Peer Specialist Certification Board 11 North Pearl Street, Suite 801 Albany New York 12207 Phone: 518.426.0945 Fax: 518.426.1046 www.nypeerspecialist.org
AFFILIATION PROGRAM AGREEMENT
 AFFILIATION PROGRAM AGREEMENT This AFFILIATION PROGRAM AGREEMENT (this Agreement ) is made and entered into by and between FACULTY PHYSICIANS & SURGEONS OF LLUSM dba LOMA LINDA UNIVERSITY FACULTY MEDICAL
AFFILIATION PROGRAM AGREEMENT This AFFILIATION PROGRAM AGREEMENT (this Agreement ) is made and entered into by and between FACULTY PHYSICIANS & SURGEONS OF LLUSM dba LOMA LINDA UNIVERSITY FACULTY MEDICAL
CHAPTER 3. Union Referral Procedures MAY 2014
 EUROPEAN COMMISSION HEALTH AND CONSUMERS DIRECTORATE-GENERAL Health systems and products Medicinal products Brussels, Revision May 2014 NOTICE TO APPLICANTS VOLUME 2A Procedures for marketing authorisation
EUROPEAN COMMISSION HEALTH AND CONSUMERS DIRECTORATE-GENERAL Health systems and products Medicinal products Brussels, Revision May 2014 NOTICE TO APPLICANTS VOLUME 2A Procedures for marketing authorisation
