The Spine Institute for Quality Conservative Care QCDR, Powered by Premier, Inc.
|
|
|
- Randolph Wright
- 5 years ago
- Views:
Transcription
1 The Spine Institute for Quality Conservative Care QCDR, Powered by Premier, Inc non-mips Measure Specifications Last updated March 7, 2018
2 SPINEIQ5: Change in Functional Outcomes- National Quality Strategy Domain: Person and Caregiver- Centered Experience Outcomes MEASURE TYPE: Patient Reported Outcome DESCRIPTION: Average percentage decrease in PROMIS Pain Interference Score for all episodes of care for back or neck pain. NOTE: Negative decreases (or increases in pain inference) for a provider's overall score are reported as a 0% reduction. DENOMINAT: All patients 18 years of age and older with an episode of care for back or neck pain and at least two functional outcomes assessments (one baseline and at least one follow-up) using the PROMIS Pain Interference assessment during the episode of care. Denominator Criteria (Eligible Cases): Diagnosis of back or neck pain: G57.00, G57.01, G57. 02, M40.05, M40.15, M40.205, M40.209, M40.295, M40.299, M40.30, M40.35, M40.36, M40.37, M40.50, M40.55, M40.56, M40.57, M41.04, M41.05, M41.06, M41.07, M41.08, M41.24, M41.25, M41.26, M41.27, M41.34, M41.35, M41.44, M41.45, M41.46, M41.47, M41.84, M41.85, M41.86, M41.87, M41.9, M43.04, M43.05, M43.06, M43.07, M43.08, M43.09, M43.14, M43.15, M43.16, M43.17, M43.18, M43.19, M43.24, M43.25, M43.26, M43.27, M43.28, M43.8X4, M43.8X5, M43.8X6, M43.8X7, M43.8X8, M43.8X9, M43.9, M46.44, M46.45, M46.46, M46.47, M46.48, M47.14, M47.15, M47.16, M47.811, M47.812, M47.813, M47.814, M47.815, M47.816, M47.817, M47.894, M47.895, M47.896, M47.897, M47.898, M48.04, M48.05, M48.061, M48.062, M48.07, M48.08, M48.14, M48.15, M48.16, M48.17, M48.18, M48.24, M48.25, M48.26, M48.27, M50.20, M50.21, M50.221, M50.222, M50.223, M50.23, M50.30, M50.31, M50.321, M50.322, M50.323, M50.33, M51.37, M51.44, M51.45, M51.46, M51.47, M51.84, M51.85, M51.86, M51.87, M53.2X4, M53.2X5, M53.2X6, M53.2x7, M53.2x8, M53.3, M53.80, M53.84, M53.85, M53.86, M53.87, M53.88, M54.00, M54.12, M54.13, M54.16, M54.17, M54.30, M54.31, M54.32, M54.40, M54.41, M54.42, M54.5, M54.6, M54.89, M54.9, M79.1, M99.00, M99.01, M99.02, M99.03, M99.04, M99.22, M99.23, M99.32, M99.33, M99.42, M99.43, M99.52, M99.53, M99.62, M99.63, M99.72, M99.73, M99.82, M99.83, M99.84, Q76.414, Q76.415, Q76.425, Q76.426, Q76.427, Q76.49, S13.4xxA, S13.4xxD, S13.4xxS, S13.8xxA, S13.8xxD, S13.8xxS, S16.1xxA, S16.1xxD, S16.1xxS, S23.100A, S23.100D, S23.100S, S23.110A, S23.110D, S23.110S, S23.120A, S23.120D, S23.120S, S23.122A, S23.122D, S23.122S, S23.130A, S23.130D, S23.130S, S23.132A, S23.132D, S23.132S, S23.140A, S23.140D, S23.140S, S23.142A, S23.142D, S23.142S, S23.150A, S23.150D, S23.150S, S23.152A, S23.152D, S23.152S, S23.160A, S23.160D, S23.160S, S23.162A, S23.162D, S23.162S, S23.170A, S23.170D, S23.170S, S23.3XXA, S23.3XXD, S23.3XXS, S23.41XA, S23.41XD, S23.41XS, S23.8XXA, S23.8XXD, S23.8XXS, S23.9XXA, S23.9XXD, S23.9XXS, S29.001A, S29.001D, S29.001S, S29.002A, S29.002D, S29.002S, S29.009A, S29.009D, S29.009S, S29.011A, S29.011D, S29.011S, S29.012A, S29.012D, S29.012S, S29.019A, S29.019D, S29.019S, S29.091A, S29.091D, S29.091S, S29.092A, S29.092D, S29.092S, S29.099A, S29.099D, S29.099S, S29.8XXA, S29.8XXD, S29.8XXS, S29.9XXA, S29.9XXD, S29.9XXS, S33.100A, S33.100D, S33.100S, S33.110A, S33.110D, S33.110S, S33.120A, S33.120D, S33.120S, S33.130A, S33.130D, S33.130S, S33.140A, S33.140D, S33.140S, S33.5XXA, S33.5XXD, S33.5XXS, S33.6XXA, S33.6XXD, S33.6XXS, S33.8XXA, S33.8XXD, S33.8XXS, S33.9XXA, S39.002A, S39.002D, S39.002S, S39.011A, S39.011D, S39.011S, S39.012A, S39.012D, S39.012S, S39.013A, S39.013D, S39.013S, S39.092A, S39.092D, S39.092S, S39.82XA, S39.82XD, S39.82XS, S39.92XA, S39.92XD, S39.92XS Encounter for back or neck pain: 27096, 62320, 62321, 62322, 62323, 64479, 64480, 64483, 64484, 64461, 64462, 72125, 72128, 72131, 72141, 72146, 72148, 97124, 97140, 97810, 97811, 97813, 97814, 98940, 98941, 98942, 98943, 99201, 99202, 99203, 99204, 99205, 99211, 99212, 99213, 99214, 99215, 97161, 97162, 97163, 97164, 98925, 98926, 98927, 98928, Baseline functional outcome assessment using the PROMIS Pain Interference assessment during the episode of care: (SpineIQ_1.NUMER.1.YES) Follow-up functional outcome assessment using the PROMIS Pain Interference assessment during the episode of care: (SpineIQ_1.NUMER.1.YES) NUMERAT: Definitions:
3 Table 2: Pain Interference 4a: How much did pain interfere with your day to day activities? How much did pain interfere with work around the home? How much did pain interfere with your ability to participate in social activities? How much did pain interfere with your household chores? 1 = Not at all 2 = A little bit 3 = Somewhat 4 = Quite a bit 5 = Very much 1 = Not at all 2 = A little bit 3 = Somewhat 4 = Quite a bit 5 = Very much 1 = Not at all 2 = A little bit 3 = Somewhat 4 = Quite a bit 5 = Very much 1 = Not at all 2 = A little bit 3 = Somewhat 4 = Quite a bit 5 = Very much Percentage change in PROMIS Pain Interference Score for each eligible episode of care for back or neck pain. Percentage change in each episode of care is calculated as follows: [(Baseline Assessment - Final Follow-Up Assessment) / Baseline Assessment] * 100 Numerator Options: Performance Met: Performance Not Met: The patient had a 30% point or greater decrease in PROMIS Pain Interference Score from initial assessment to final assesment during an episode of care for a spine-related disorder. (SpineIQ_3.NUMER.1.YES) The patient did not have a 30% point or greater decrease in PROMIS Pain Interference Score from initial assessment to final assesment during an episode of care for a spine-related disorder. (SpineIQ_3.NUMER.3.NO)
4 SPINEIQ6: Change in Pain Intensity- National Quality Strategy Domain: Person and Caregiver-Centered Experience Outcomes MEASURE TYPE: Patient Reported Outcome DESCRIPTION: Average percentage decrease in PROMIS Pain Intensity Score for all episodes of care for back or neck pain. NOTE: Negative decreases (or increases in pain inference) for a provider's overall score are reported as a 0% reduction. DENOMINAT: All patients 18 years of age and older with an episode of care for back or neck pain and at least two functional outcomes assessments (first non-zero score will be used as the baseline assessment and at least one subsequent follow-up) using the PROMIS Pain Intensity assessment during the episode of care. Denominator Criteria (Eligible Cases): Diagnosis of back or neck pain: G57.00, G57.01, G57. 02, M40.05, M40.15, M40.205, M40.209, M40.295, M40.299, M40.30, M40.35, M40.36, M40.37, M40.50, M40.55, M40.56, M40.57, M41.04, M41.05, M41.06, M41.07, M41.08, M41.24, M41.25, M41.26, M41.27, M41.34, M41.35, M41.44, M41.45, M41.46, M41.47, M41.84, M41.85, M41.86, M41.87, M41.9, M43.04, M43.05, M43.06, M43.07, M43.08, M43.09, M43.14, M43.15, M43.16, M43.17, M43.18, M43.19, M43.24, M43.25, M43.26, M43.27, M43.28, M43.8X4, M43.8X5, M43.8X6, M43.8X7, M43.8X8, M43.8X9, M43.9, M46.44, M46.45, M46.46, M46.47, M46.48, M47.14, M47.15, M47.16, M47.811, M47.812, M47.813, M47.814, M47.815, M47.816, M47.817, M47.894, M47.895, M47.896, M47.897, M47.898, M48.04, M48.05, M48.061, M48.062, M48.07, M48.08, M48.14, M48.15, M48.16, M48.17, M48.18, M48.24, M48.25, M48.26, M48.27, M50.20, M50.21, M50.221, M50.222, M50.223, M50.23, M50.30, M50.31, M50.321, M50.322, M50.323, M50.33, M51.37, M51.44, M51.45, M51.46, M51.47, M51.84, M51.85, M51.86, M51.87, M53.2X4, M53.2X5, M53.2X6, M53.2x7, M53.2x8, M53.3, M53.80, M53.84, M53.85, M53.86, M53.87, M53.88, M54.00, M54.12, M54.13, M54.16, M54.17, M54.30, M54.31, M54.32, M54.40, M54.41, M54.42, M54.5, M54.6, M54.89, M54.9, M79.1, M99.00, M99.01, M99.02, M99.03, M99.04, M99.22, M99.23, M99.32, M99.33, M99.42, M99.43, M99.52, M99.53, M99.62, M99.63, M99.72, M99.73, M99.82, M99.83, M99.84, Q76.414, Q76.415, Q76.425, Q76.426, Q76.427, Q76.49, S13.4xxA, S13.4xxD, S13.4xxS, S13.8xxA, S13.8xxD, S13.8xxS, S16.1xxA, S16.1xxD, S16.1xxS, S23.100A, S23.100D, S23.100S, S23.110A, S23.110D, S23.110S, S23.120A, S23.120D, S23.120S, S23.122A, S23.122D, S23.122S, S23.130A, S23.130D, S23.130S, S23.132A, S23.132D, S23.132S, S23.140A, S23.140D, S23.140S, S23.142A, S23.142D, S23.142S, S23.150A, S23.150D, S23.150S, S23.152A, S23.152D, S23.152S, S23.160A, S23.160D, S23.160S, S23.162A, S23.162D, S23.162S, S23.170A, S23.170D, S23.170S, S23.3XXA, S23.3XXD, S23.3XXS, S23.41XA, S23.41XD, S23.41XS, S23.8XXA, S23.8XXD, S23.8XXS, S23.9XXA, S23.9XXD, S23.9XXS, S29.001A, S29.001D, S29.001S, S29.002A, S29.002D, S29.002S, S29.009A, S29.009D, S29.009S, S29.011A, S29.011D, S29.011S, S29.012A, S29.012D, S29.012S, S29.019A, S29.019D, S29.019S, S29.091A, S29.091D, S29.091S, S29.092A, S29.092D, S29.092S, S29.099A, S29.099D, S29.099S, S29.8XXA, S29.8XXD, S29.8XXS, S29.9XXA, S29.9XXD, S29.9XXS, S33.100A, S33.100D, S33.100S, S33.110A, S33.110D, S33.110S, S33.120A, S33.120D, S33.120S, S33.130A, S33.130D, S33.130S, S33.140A, S33.140D, S33.140S, S33.5XXA, S33.5XXD, S33.5XXS, S33.6XXA, S33.6XXD, S33.6XXS, S33.8XXA, S33.8XXD, S33.8XXS, S33.9XXA, S39.002A, S39.002D, S39.002S, S39.011A, S39.011D, S39.011S, S39.012A, S39.012D, S39.012S, S39.013A, S39.013D, S39.013S, S39.092A, S39.092D, S39.092S, S39.82XA, S39.82XD, S39.82XS, S39.92XA, S39.92XD, S39.92XS Encounter for back or neck pain: 27096, 62320, 62321, 62322, 62323, 64479, 64480, 64483, 64484, 64461, 64462, 72125, 72128, 72131, 72141, 72146, 72148, 97124, 97140, 97810, 97811, 97813, 97814, 98940, 98941, 98942, 98943, 99201, 99202, 99203, 99204, 99205, 99211, 99212, 99213, 99214, 99215, 97161, 97162, 97163, 97164, 98925, 98926, 98927, 98928, Baseline functional outcome assessment using the PROMIS Pain Interference assessment during the episode of care: (SpineIQ_1.NUMER.1.YES) Follow-up functional outcome assessment using the PROMIS Pain Interference assessment during the episode of care: (SpineIQ_1.NUMER.1.YES)
5 NUMERAT: Definitions: PROMIS Pain Intensity NRS How would you rate your pain on average? [scale] 0 = No Pain 10 = Worst Imaginable Pain Percentage change in PROMIS Pain Intensity Score for each eligible episode of care for back or neck pain. Percentage change in each episode of care is calculated as follows: [(Baseline Assessment - Final Follow-Up Assessment) / Baseline Assessment] * 100 Numerator Options: Performance Met: Performance Not Met: The patient had a 30% point or greater decrease in PROMIS Pain Intensity Score from initial assessment to final assesment during an episode of care for a spine-related disorder. (SpineIQ_3.NUMER.1.YES) The patient did not have a 30% point or greater decrease in PROMIS Pain Intensity Score from initial assessment to final assesment during an episode of care for a spine-related disorder. (SpineIQ_3.NUMER.3.NO)
6 SPINEIQ3: Repeated X-ray Imaging- National Quality Strategy Domain: Efficiency and Cost Reduction Use MEASURE TYPE: Process HIGH PRIITY: Yes DESCRIPTION: Percentage of patients with spine-related disorders who received two or more of the same x-ray imaging studies for the purpose of monitoring the patient s condition, either performed or ordered by an eligible clinician within the measurement period. DENOMINAT: All patients 18 years of age and older with a diagnosis of spine-related disorders on an eligible encounter during the measurement period in which an x-ray study is ordered or performed for the purpose of monitoring the patient s condition by an eligible clinician. Denominator Criteria (Eligible Cases): Diagnosis of back pain or neck pain: G57.00, G57.01, G57. 02, M40.05, M40.15, M40.205, M40.209, M40.295, M40.299, M40.30, M40.35, M40.36, M40.37, M40.50, M40.55, M40.56, M40.57, M41.04, M41.05, M41.06, M41.07, M41.08, M41.24, M41.25, M41.26, M41.27, M41.34, M41.35, M41.44, M41.45, M41.46, M41.47, M41.84, M41.85, M41.86, M41.87, M41.9, M43.04, M43.05, M43.06, M43.07, M43.08, M43.09, M43.14, M43.15, M43.16, M43.17, M43.18, M43.19, M43.24, M43.25, M43.26, M43.27, M43.28, M43.8X4, M43.8X5, M43.8X6, M43.8X7, M43.8X8, M43.8X9, M43.9, M46.44, M46.45, M46.46, M46.47, M46.48, M47.14, M47.15, M47.16, M47.811, M47.812, M47.813, M47.814, M47.815, M47.816, M47.817, M47.894, M47.895, M47.896, M47.897, M47.898, M48.04, M48.05, M48.061, M48.062, M48.07, M48.08, M48.14, M48.15, M48.16, M48.17, M48.18, M48.24, M48.25, M48.26, M48.27, M50.20, M50.21, M50.221, M50.222, M50.223, M50.23, M50.30, M50.31, M50.321, M50.322, M50.323, M50.33, M51.37, M51.44, M51.45, M51.46, M51.47, M51.84, M51.85, M51.86, M51.87, M53.2X4, M53.2X5, M53.2X6, M53.2x7, M53.2x8, M53.3, M53.80, M53.84, M53.85, M53.86, M53.87, M53.88, M54.00, M54.12, M54.13, M54.16, M54.17, M54.30, M54.31, M54.32, M54.40, M54.41, M54.42, M54.5, M54.6, M54.89, M54.9, M79.1, M99.00, M99.01, M99.02, M99.03, M99.04, M99.22, M99.23, M99.32, M99.33, M99.42, M99.43, M99.52, M99.53, M99.62, M99.63, M99.72, M99.73, M99.82, M99.83, M99.84, Q76.414, Q76.415, Q76.425, Q76.426, Q76.427, Q76.49, S13.4xxA, S13.4xxD, S13.4xxS, S13.8xxA, S13.8xxD, S13.8xxS, S16.1xxA, S16.1xxD, S16.1xxS, S23.100A, S23.100D, S23.100S, S23.110A, S23.110D, S23.110S, S23.120A, S23.120D, S23.120S, S23.122A, S23.122D, S23.122S, S23.130A, S23.130D, S23.130S, S23.132A, S23.132D, S23.132S, S23.140A, S23.140D, S23.140S, S23.142A, S23.142D, S23.142S, S23.150A, S23.150D, S23.150S, S23.152A, S23.152D, S23.152S, S23.160A, S23.160D, S23.160S, S23.162A, S23.162D, S23.162S, S23.170A, S23.170D, S23.170S, S23.3XXA, S23.3XXD, S23.3XXS, S23.41XA, S23.41XD, S23.41XS, S23.8XXA, S23.8XXD, S23.8XXS, S23.9XXA, S23.9XXD, S23.9XXS, S29.001A, S29.001D, S29.001S, S29.002A, S29.002D, S29.002S, S29.009A, S29.009D, S29.009S, S29.011A, S29.011D, S29.011S, S29.012A, S29.012D, S29.012S, S29.019A, S29.019D, S29.019S, S29.091A, S29.091D, S29.091S, S29.092A, S29.092D, S29.092S, S29.099A, S29.099D, S29.099S, S29.8XXA, S29.8XXD, S29.8XXS, S29.9XXA, S29.9XXD, S29.9XXS, S33.100A, S33.100D, S33.100S, S33.110A, S33.110D, S33.110S, S33.120A, S33.120D, S33.120S, S33.130A, S33.130D, S33.130S, S33.140A, S33.140D, S33.140S, S33.5XXA, S33.5XXD, S33.5XXS, S33.6XXA, S33.6XXD, S33.6XXS, S33.8XXA, S33.8XXD, S33.8XXS, S33.9XXA, S39.002A, S39.002D, S39.002S, S39.011A, S39.011D, S39.011S, S39.012A, S39.012D, S39.012S, S39.013A, S39.013D, S39.013S, S39.092A, S39.092D, S39.092S, S39.82XA, S39.82XD, S39.82XS, S39.92XA, S39.92XD, S39.92XS Eligible encounter during the measurement period: 97140, 98940, 98941, 98942, 98943, 99201, 99202, 99203, 99204, 99205, 99211, 99212, 99213, 99214, X-Ray study performed during the encounter: 72020, 72040, 72050, 72052, 72070, 72072, 72074, 72080, 72081, 72082, 72083, 72084, 72100, 72110, 72114, 72170, 72200, NUMERAT: Patients with two or more of the same x-ray imaging studies within the one year measurement period.
7 Numerator Instructions: INVERSE MEASURE Numerator Options: Performance Met: Denominator Exception: Performance Not Met: The patient had two or more of the same X-Ray imaging study within the one year measurement period (SpineIQ_3.NUMER.1.YES) The patient did have two or more of the same X-Ray imaging study within the one year measurement period, but medical reasons documented (e.g., patient with a new presenting complaint, documentation of worsening symptoms, documentation of signs of progressive disease (SpineIQ_3.NUMER.2.YES) The patient did not have two or more of the same X-Ray imaging study within the one year measurement period. SpineIQ_3.NUMER.3.NO)
8 SPINEIQ4: MRI of the Lumbar Spine Without Prior Conservative Care- National Quality Strategy Domain: Communication and Care Coordination MEASURE TYPE: Process HIGH PRIITY: Yes DESCRIPTION: Percentage of patients with diagnosis of low back pain receiving MRI in the absence of prior conservative care DENOMINAT: All patients 18 years of age and older with low back pain receiving lumbar spine MRI study Denominator Criteria (Eligible Cases): Diagnosis of low back pain: G57.00, G57.01, G57. 02, M40.05, M40.15, M40.205, M40.295, M40.35, M40.36, M40.37, M40.55, M40.56, M40.57, M43.05, M43.06, M43.07, M43.15, M43.16, M43.17, M43.25, M43.26, M43.27, M43.8X5, M43.8X6, M43.8X7, M46.45, M46.46, M46.47, M47.15, M47.16, M47.815, M47.816, M47.817, M47.895, M47.896, M47.897, M48.05, M48.061, M48.062, M48.07, M48.15, M48.16, M48.17, M48.25, M48.26, M48.27, M51.37, M51.45, M51.46, M51.47, M51.85, M51.86, M51.87, M53.2X5, M53.2X6, M53.2x7, M53.85, M53.86, M53.87, M54.16, M54.17, M54.30, M54.31, M54.32, M54.40, M54.41, M54.42, M54.5, M54.89, M54.9, M79.1, M99.03, M99.23, M99.33, M99.43, M99.53, M99.63, M99.73, M99.83, Q76.415, Q76.426, Q76.427, Q76.49, S33.100A, S33.100D, S33.100S, S33.110A, S33.110D, S33.110S, S33.120A, S33.120D, S33.120S, S33.130A, S33.130D, S33.130S, S33.140A, S33.140D, S33.140S, S33.5XXA, S33.5XXD, S33.5XXS, S33.8XXA, S33.8XXD, S33.8XXS, S33.9XXA, S39.002A, S39.002D, S39.002S, S39.012A, S39.012D, S39.012S, S39.092A, S39.092D, S39.092S, S39.82XA, S39.82XD, S39.82XS, S39.92XA, S39.92XD, S39.92XS MRI study of lumbar spine: 72148, 72149, NUMERAT: All patients 18 years of age and older without conservative care for low back pain. Indications for conservative low back care include: Evaluation and management services and an order/prescription for conservative care. Conservative care includes chiropractic manipulative therapy, osteopathic manipulative therapy, acupuncture, physical therapy, massage therapy. Numerator Instructions: INVERSE MEASURE Numerator Options: Performance Met: Denominator Exception: Performance Not Met: The patient did not have conservative care for low back pain prior to receiving an MRI, no reason or other reason(s) specified. (SpineIQ_4.NUMER.1.YES) The patient did not have conservative care for low back pain due to one or more of the following reasons: Neoplasia, spine surgery 90 days prior to MRI, Congenital and spinal cord malformation, Inflammatory and autoimmune disorders within 5 years, /spinal vascular malformations, infection, infarction, abnormalities associated with scoliosis, trauma (within 45 days), neurological impairment, IV drug use, HIV (within one year, Intraspinal abcess (SpineIQ_4.NUMER.2.YES) The patient had conservative care prior to receiving a MRI for low back pain (SpineIQ_4.NUMER.3.NO)
OP-8: MRI LUMBAR SPINE FOR LOW BACK PAIN
 Description of Measure OP-8: MRI LUMBAR SPINE FOR LOW BACK PAIN This measure calculates the percentage of MRI of the Lumbar Spine studies with a diagnosis of low back pain on the imaging claim and for
Description of Measure OP-8: MRI LUMBAR SPINE FOR LOW BACK PAIN This measure calculates the percentage of MRI of the Lumbar Spine studies with a diagnosis of low back pain on the imaging claim and for
Use the Physician Extender modifier for non-physician services. Additional acupuncture information is available later in this chapter.
 Chapter 18 Chiropractic Services Definition Chiropractic services are medically necessary therapies that employ manipulation and specific adjustment of body structures, such as the spinal column, provided
Chapter 18 Chiropractic Services Definition Chiropractic services are medically necessary therapies that employ manipulation and specific adjustment of body structures, such as the spinal column, provided
Tri-County Opioid Safety Coalition Data Brief December 2017 Clackamas, Multnomah, and Washington Counties
 Medicaid-Funded Alternative Treatment for Back Pain in the Tri-County Region Key Findings The percentage of members with a back pain diagnosis who received an alternative treatment increased from 29% in
Medicaid-Funded Alternative Treatment for Back Pain in the Tri-County Region Key Findings The percentage of members with a back pain diagnosis who received an alternative treatment increased from 29% in
2018 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY. MEASURE TYPE: Outcome
 Quality ID #345 (NQF 1543): Rate of Asymptomatic Patients Undergoing Carotid Artery Stenting (CAS) Who Are Stroke Free or Discharged Alive National Quality Strategy Domain: Effective Clinical Care 2018
Quality ID #345 (NQF 1543): Rate of Asymptomatic Patients Undergoing Carotid Artery Stenting (CAS) Who Are Stroke Free or Discharged Alive National Quality Strategy Domain: Effective Clinical Care 2018
2019 COLLECTION TYPE: MIPS CLINICAL QUALITY MEASURES (CQMS) MEASURE TYPE: Patient Reported Outcome High Priority
 Quality ID #460: Average Change in Back Pain Following Lumbar Fusion National Quality Strategy Domain: Person and Caregiver-Centered Experience and Outcomes Meaningful Measure Area: Patient Reported Functional
Quality ID #460: Average Change in Back Pain Following Lumbar Fusion National Quality Strategy Domain: Person and Caregiver-Centered Experience and Outcomes Meaningful Measure Area: Patient Reported Functional
Kathryn Goodwin, Senior Project Manager, and Karen Johnson, Senior Director
 TO: FR: RE: Consensus Standards Approval Committee (CSAC) Kathryn Goodwin, Senior Project Manager, and Karen Johnson, Senior Director Musculoskeletal Off-Cycle Measure Review DA: April 26, 2017 The CSAC
TO: FR: RE: Consensus Standards Approval Committee (CSAC) Kathryn Goodwin, Senior Project Manager, and Karen Johnson, Senior Director Musculoskeletal Off-Cycle Measure Review DA: April 26, 2017 The CSAC
DENOMINATOR: Patients aged 18 and older with infrarenal non-ruptured endovascular AAA repairs
 Measure #347 (NQF 1534): Rate of Endovascular Aneurysm Repair (EVAR) of Small or Moderate Non-Ruptured Abdominal Aortic Aneurysms (AAA) Who Die While in Hospital National Quality Strategy Domain: Patient
Measure #347 (NQF 1534): Rate of Endovascular Aneurysm Repair (EVAR) of Small or Moderate Non-Ruptured Abdominal Aortic Aneurysms (AAA) Who Die While in Hospital National Quality Strategy Domain: Patient
2019 COLLECTION TYPE: MIPS CLINICAL QUALITY MEASURES (CQMS) MEASURE TYPE: Patient Reported Outcome High Priority
 Quality ID #223 (NQF 0428): Functional Status Change for Patients with General Orthopedic Impairments National Quality Strategy Domain: Communication and Care Coordination Meaningful Measure Area: Patient
Quality ID #223 (NQF 0428): Functional Status Change for Patients with General Orthopedic Impairments National Quality Strategy Domain: Communication and Care Coordination Meaningful Measure Area: Patient
2019 COLLECTION TYPE: MIPS CLINICAL QUALITY MEASURES (CQMS) MEASURE TYPE: Patient Reported Outcome High Priority
 Quality ID #220 (NQF 0425): Functional Status Change for Patients with Low Back Impairments National Quality Strategy Domain: Communication and Care Coordination Meaningful Measure Area: Patient Reported
Quality ID #220 (NQF 0425): Functional Status Change for Patients with Low Back Impairments National Quality Strategy Domain: Communication and Care Coordination Meaningful Measure Area: Patient Reported
2016 PQRS OPTIONS FOR INDIVIDUAL MEASURES: CLAIMS, REGISTRY
 Measure #416: Emergency Medicine: Emergency Department Utilization of CT for Minor Blunt Head Trauma for Patients Aged 2 through 17 Years - National Quality Strategy Domain: Efficiency and Cost Reduction
Measure #416: Emergency Medicine: Emergency Department Utilization of CT for Minor Blunt Head Trauma for Patients Aged 2 through 17 Years - National Quality Strategy Domain: Efficiency and Cost Reduction
2017 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY. MEASURE TYPE: Efficiency
 Measure #416: Emergency Medicine: Emergency Department Utilization of CT for Minor Blunt Head Trauma for Patients Aged 2 Through 17 Years - National Quality Strategy Domain: Efficiency and Cost Reduction
Measure #416: Emergency Medicine: Emergency Department Utilization of CT for Minor Blunt Head Trauma for Patients Aged 2 Through 17 Years - National Quality Strategy Domain: Efficiency and Cost Reduction
2017 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY. MEASURE TYPE: Outcome
 Measure #445 (NQF 0119): Risk-Adjusted Operative Mortality for Coronary Artery Bypass Graft (CABG) National Quality Strategy Domain: Effective Clinical Care 2017 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY
Measure #445 (NQF 0119): Risk-Adjusted Operative Mortality for Coronary Artery Bypass Graft (CABG) National Quality Strategy Domain: Effective Clinical Care 2017 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY
JAW. R68.84 Jaw pain M26.60 Temporomandibular joint disorder, unspecified
 HEAD, JAW AND NECK HEADACHE M53.0 Cervicocranial Syndrome R51 Headache (excludes migraine and other headache syndromes) G44.209 Tension-type headache, unspecified, not intractable G44.309 Post-traumatic
HEAD, JAW AND NECK HEADACHE M53.0 Cervicocranial Syndrome R51 Headache (excludes migraine and other headache syndromes) G44.209 Tension-type headache, unspecified, not intractable G44.309 Post-traumatic
2017 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY. MEASURE TYPE: Outcome
 Measure #221 (NQF 0426): Functional Status Change for Patients with Shoulder Impairments National Quality Strategy Domain: Communication and Care Coordination 2017 OPTIONS F INDIVIDUAL MEASURES: REGISTRY
Measure #221 (NQF 0426): Functional Status Change for Patients with Shoulder Impairments National Quality Strategy Domain: Communication and Care Coordination 2017 OPTIONS F INDIVIDUAL MEASURES: REGISTRY
ACO #44 Use of Imaging Studies for Low Back Pain
 Measure Information Form (MIF) DATA SOURCE Medicare Claims Medicare beneficiary enrollment data MEASURE SET ID ACO #44 VERSION NUMBER AND EFFECTIVE DATE Version 1, effective 01/01/18 CMS APPROVAL DATE
Measure Information Form (MIF) DATA SOURCE Medicare Claims Medicare beneficiary enrollment data MEASURE SET ID ACO #44 VERSION NUMBER AND EFFECTIVE DATE Version 1, effective 01/01/18 CMS APPROVAL DATE
2019 COLLECTION TYPE: MIPS CLINICAL QUALITY MEASURES (CQMS) MEASURE TYPE: Process High Priority
 Quality ID #261: Referral for Otologic Evaluation for Patients with Acute or Chronic Dizziness National Quality Strategy Domain: Communication and Care Coordination Meaningful Measure Area: Transfer of
Quality ID #261: Referral for Otologic Evaluation for Patients with Acute or Chronic Dizziness National Quality Strategy Domain: Communication and Care Coordination Meaningful Measure Area: Transfer of
DENOMINATOR: All patients aged birth and older presenting with acute or chronic dizziness
 Quality ID #261: Referral for Otologic Evaluation for Patients with Acute or Chronic Dizziness National Quality Strategy Domain: Communication and Care Coordination 2018 OPTIONS FOR INDIVIDUAL MEASURES:
Quality ID #261: Referral for Otologic Evaluation for Patients with Acute or Chronic Dizziness National Quality Strategy Domain: Communication and Care Coordination 2018 OPTIONS FOR INDIVIDUAL MEASURES:
Spine Pain Management Program
 Spine Pain Management Program Please complete the following information: Patient Name: Patient ID Number: Patient DOB: The procedure being requested is: Please check the indication (reason) for this procedure
Spine Pain Management Program Please complete the following information: Patient Name: Patient ID Number: Patient DOB: The procedure being requested is: Please check the indication (reason) for this procedure
Quality ID #6 (NQF 0067): Coronary Artery Disease (CAD): Antiplatelet Therapy National Quality Strategy Domain: Effective Clinical Care
 Quality ID #6 (NQF 0067): Coronary Artery Disease (CAD): Antiplatelet Therapy National Quality Strategy Domain: Effective Clinical Care 2018 OPTIONS F INDIVIDUAL MEASURES: REGISTRY ONLY MEASURE TYPE: Process
Quality ID #6 (NQF 0067): Coronary Artery Disease (CAD): Antiplatelet Therapy National Quality Strategy Domain: Effective Clinical Care 2018 OPTIONS F INDIVIDUAL MEASURES: REGISTRY ONLY MEASURE TYPE: Process
PATIENT REGISTRATION FORM
 PATIENT REGISTRATION FORM NAME: D.O.B AGE: SEX: STREET: CITY: STATE: ZIP: SS #: ETHNICITY: RACE: LANGUAGE: PHONE # TO LEAVE A PERSONAL MESSAGE: HOME PHONE #: WORK #: CELL #: E MAIL ADDRESS: EMERGENCY CONTACT:
PATIENT REGISTRATION FORM NAME: D.O.B AGE: SEX: STREET: CITY: STATE: ZIP: SS #: ETHNICITY: RACE: LANGUAGE: PHONE # TO LEAVE A PERSONAL MESSAGE: HOME PHONE #: WORK #: CELL #: E MAIL ADDRESS: EMERGENCY CONTACT:
Quality ID #224 (NQF 0562): Melanoma: Overutilization of Imaging Studies in Melanoma National Quality Strategy Domain: Efficiency and Cost Reduction
 Quality ID #224 (NQF 0562): Melanoma: Overutilization of Imaging Studies in Melanoma National Quality Strategy Domain: Efficiency and Cost Reduction 2018 OPTIONS F INDIVIDUAL MEASURES: REGISTRY ONLY MEASURE
Quality ID #224 (NQF 0562): Melanoma: Overutilization of Imaging Studies in Melanoma National Quality Strategy Domain: Efficiency and Cost Reduction 2018 OPTIONS F INDIVIDUAL MEASURES: REGISTRY ONLY MEASURE
2018 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY. MEASURE TYPE: Outcome
 Quality ID #217 (NQF 0422): Functional Status Change for Patients with Knee Impairments National Quality Strategy Domain: Communication and Care Coordination 2018 OPTIONS F INDIVIDUAL MEASURES: REGISTRY
Quality ID #217 (NQF 0422): Functional Status Change for Patients with Knee Impairments National Quality Strategy Domain: Communication and Care Coordination 2018 OPTIONS F INDIVIDUAL MEASURES: REGISTRY
INDIANA HEALTH COVERAGE PROGRAMS
 INDIANA HEALTH COVERAGE PROGRAMS PROVIDER CODE TABLES Note: Due to possible changes in Indiana Health Coverage Programs (IHCP) policy or national coding updates, inclusion of a code on the code tables
INDIANA HEALTH COVERAGE PROGRAMS PROVIDER CODE TABLES Note: Due to possible changes in Indiana Health Coverage Programs (IHCP) policy or national coding updates, inclusion of a code on the code tables
Current ICD-10 Codes
 Current ICD-10 Codes Description (Facet Syndromes) M53.81 Other specified dorsopathies, occipito-atlanto-axial region M53.82 Other specified dorsopathies, cervical region M53.83 Other specified dorsopathies,
Current ICD-10 Codes Description (Facet Syndromes) M53.81 Other specified dorsopathies, occipito-atlanto-axial region M53.82 Other specified dorsopathies, cervical region M53.83 Other specified dorsopathies,
Measure #412: Documentation of Signed Opioid Treatment Agreement National Quality Strategy Domain: Effective Clinical Care
 Measure #412: Documentation of Signed Opioid Treatment Agreement National Quality Strategy Domain: Effective Clinical Care 2016 PQRS OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY DESCRIPTION: All patients
Measure #412: Documentation of Signed Opioid Treatment Agreement National Quality Strategy Domain: Effective Clinical Care 2016 PQRS OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY DESCRIPTION: All patients
Measure #131 (NQF 0420): Pain Assessment and Follow-Up National Quality Strategy Domain: Communication and Care Coordination
 Measure #131 (NQF 0420): Pain Assessment and Follow-Up National Quality Strategy Domain: Communication and Care Coordination 2017 OPTIONS F INDIVIDUAL MEASURE: REGISTRY ONLY MEASURE TYPE: Process DESCRIPTION:
Measure #131 (NQF 0420): Pain Assessment and Follow-Up National Quality Strategy Domain: Communication and Care Coordination 2017 OPTIONS F INDIVIDUAL MEASURE: REGISTRY ONLY MEASURE TYPE: Process DESCRIPTION:
Measure #286: Dementia: Counseling Regarding Safety Concerns National Quality Strategy Domain: Patient Safety
 Measure #286: Dementia: Counseling Regarding Safety Concerns National Quality Strategy Domain: Patient Safety 2017 OPTIONS F INDIVIDUAL MEASURES: REGISTRY ONLY MEASURE TYPE: Process DESCRIPTION: Percentage
Measure #286: Dementia: Counseling Regarding Safety Concerns National Quality Strategy Domain: Patient Safety 2017 OPTIONS F INDIVIDUAL MEASURES: REGISTRY ONLY MEASURE TYPE: Process DESCRIPTION: Percentage
Quality ID #117 (NQF 0055): Diabetes: Eye Exam National Quality Strategy Domain: Effective Clinical Care
 Quality ID #117 (NQF 0055): Diabetes: Eye Exam National Quality Strategy Domain: Effective Clinical Care 2018 OPTIONS F INDIVIDUAL MEASURES: REGISTRY ONLY MEASURE TYPE: Process DESCRIPTION: Percentage
Quality ID #117 (NQF 0055): Diabetes: Eye Exam National Quality Strategy Domain: Effective Clinical Care 2018 OPTIONS F INDIVIDUAL MEASURES: REGISTRY ONLY MEASURE TYPE: Process DESCRIPTION: Percentage
Measure #117 (NQF 0055): Diabetes: Eye Exam National Quality Strategy Domain: Effective Clinical Care
 Measure #117 (NQF 0055): Diabetes: Eye Exam National Quality Strategy Domain: Effective Clinical Care 2017 OPTIONS F INDIVIDUAL MEASURES: CLAIMS ONLY MEASURE TYPE: Process DESCRIPTION: Percentage of patients
Measure #117 (NQF 0055): Diabetes: Eye Exam National Quality Strategy Domain: Effective Clinical Care 2017 OPTIONS F INDIVIDUAL MEASURES: CLAIMS ONLY MEASURE TYPE: Process DESCRIPTION: Percentage of patients
Quality ID #342: Pain Brought Under Control Within 48 Hours National Quality Strategy Domain: Person and Caregiver-Centered Experience and Outcomes
 Quality ID #342: Pain Brought Under Control Within 48 Hours National Quality Strategy Domain: Person and Caregiver-Centered Experience and Outcomes 2018 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY MEASURE
Quality ID #342: Pain Brought Under Control Within 48 Hours National Quality Strategy Domain: Person and Caregiver-Centered Experience and Outcomes 2018 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY MEASURE
Measure #412: Documentation of Signed Opioid Treatment Agreement National Quality Strategy Domain: Effective Clinical Care
 Measure #412: Documentation of Signed Opioid Treatment Agreement National Quality Strategy Domain: Effective Clinical Care 2017 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY MEASURE TYPE: Process DESCRIPTION:
Measure #412: Documentation of Signed Opioid Treatment Agreement National Quality Strategy Domain: Effective Clinical Care 2017 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY MEASURE TYPE: Process DESCRIPTION:
Renal Physicians Association Kidney Quality Improvement Registry, Powered by Premier, Inc non-mips Measure Specifications
 Renal Physicians Association Kidney Quality Improvement Registry, Powered by Premier, Inc. 2018 non-mips Measure Specifications Last updated January 2, 2018 RPAQIR1: Angiotensin Converting Enzyme (ACE)
Renal Physicians Association Kidney Quality Improvement Registry, Powered by Premier, Inc. 2018 non-mips Measure Specifications Last updated January 2, 2018 RPAQIR1: Angiotensin Converting Enzyme (ACE)
Numbness: o o o o o. Grade your overall pain. Pain Rating Scale Mosby. Worst Possible Pain. No Pain HURTS LITTLE MORE HURTS EVEN MORE
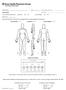 Patient Name: Original Referring Physician: Current PCP: PAIN DIAGRAM Is your condition the result of a: Work injury? YES NO Auto accident? YES NO Date of Injury: / / Please mark the areas of discomfort
Patient Name: Original Referring Physician: Current PCP: PAIN DIAGRAM Is your condition the result of a: Work injury? YES NO Auto accident? YES NO Date of Injury: / / Please mark the areas of discomfort
2017 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY. MEASURE TYPE: Outcome
 Measure #347 (NQF 1534): Rate of Endovascular Aneurysm Repair (EVAR) of Small or Moderate Non- Ruptured Infrarenal Abdominal Aortic Aneurysms (AAA) Who Die While in Hospital National Quality Strategy Domain:
Measure #347 (NQF 1534): Rate of Endovascular Aneurysm Repair (EVAR) of Small or Moderate Non- Ruptured Infrarenal Abdominal Aortic Aneurysms (AAA) Who Die While in Hospital National Quality Strategy Domain:
ICD-10-CM (GEM) ICD10Data.com CMS.gov/icd10 ICD10CODEsearch.com
 ICD-10-CM (GEM) General Equivalency Mapping (GEM): Bidirectional conversions between ICD-9 vs. ICD-10 Not intended to be crosswalks. A tool to help map/code accurately Clinical judgement & decision making
ICD-10-CM (GEM) General Equivalency Mapping (GEM): Bidirectional conversions between ICD-9 vs. ICD-10 Not intended to be crosswalks. A tool to help map/code accurately Clinical judgement & decision making
DENOMINATOR: All patients aged 18 years and older with a diagnosis of coronary artery disease seen within a 12 month period
 Quality ID #6 (NQF 0067): Coronary Artery Disease (CAD): Antiplatelet Therapy National Quality Strategy Domain: Effective Clinical Care Meaningful Measure Area: Management of Chronic Conditions 2019 COLLECTION
Quality ID #6 (NQF 0067): Coronary Artery Disease (CAD): Antiplatelet Therapy National Quality Strategy Domain: Effective Clinical Care Meaningful Measure Area: Management of Chronic Conditions 2019 COLLECTION
DESCRIPTION: Percent of asymptomatic patients undergoing CEA who are discharged to home no later than post-operative day #2
 Measure #260: Rate of Carotid Endarterectomy (CEA) for Asymptomatic Patients, without Major Complications (Discharged to Home by Post-Operative Day #2) National Quality Strategy Domain: Patient Safety
Measure #260: Rate of Carotid Endarterectomy (CEA) for Asymptomatic Patients, without Major Complications (Discharged to Home by Post-Operative Day #2) National Quality Strategy Domain: Patient Safety
2) Patients who are 18 years and older with a diagnosis of CAD or history of cardiac surgery who have a prior myocardial infarction
 Measure #7 (NQF 0070): Coronary Artery Disease (CAD): Beta-Blocker Therapy Prior Myocardial Infarction (MI) or Left Ventricular Systolic Dysfunction (LVEF < 40%) National Quality Strategy Domain: Effective
Measure #7 (NQF 0070): Coronary Artery Disease (CAD): Beta-Blocker Therapy Prior Myocardial Infarction (MI) or Left Ventricular Systolic Dysfunction (LVEF < 40%) National Quality Strategy Domain: Effective
GET BACK TO YOUR FUTURE WITH SPECIALIZED SPINE CARE. A Guide for Patients
 GET BACK TO YOUR FUTURE WITH SPECIALIZED SPINE CARE A Guide for Patients Your Spine Deserves Special Care Your spine is at the center of a delicately balanced system that controls all of your body s movements.
GET BACK TO YOUR FUTURE WITH SPECIALIZED SPINE CARE A Guide for Patients Your Spine Deserves Special Care Your spine is at the center of a delicately balanced system that controls all of your body s movements.
Musculoskeletal: Off- Cycle: Measure Review 2017
 Musculoskeletal: Off- Cycle: Measure Review 2017 DRAFT REPORT April 26, 2017 This report is funded by the Department of Health and Human Services under contract HHSM-500-2012-00009I Task Order HHSM-500-T0000
Musculoskeletal: Off- Cycle: Measure Review 2017 DRAFT REPORT April 26, 2017 This report is funded by the Department of Health and Human Services under contract HHSM-500-2012-00009I Task Order HHSM-500-T0000
2016 PQRS OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY
 Measure #260: Rate of Carotid Endarterectomy (CEA) for Asymptomatic Patients, without Major Complications (Discharged to Home by Post-Operative Day #2) National Quality Strategy Domain: Patient Safety
Measure #260: Rate of Carotid Endarterectomy (CEA) for Asymptomatic Patients, without Major Complications (Discharged to Home by Post-Operative Day #2) National Quality Strategy Domain: Patient Safety
2014 PQRS OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY
 Measure #7 (NQF 0070): Coronary Artery Disease (CAD): Beta-Blocker Therapy Prior Myocardial Infarction (MI) or Left Ventricular Systolic Dysfunction (LVEF < 40%) 2014 PQRS OPTIONS F INDIVIDUAL MEASURES:
Measure #7 (NQF 0070): Coronary Artery Disease (CAD): Beta-Blocker Therapy Prior Myocardial Infarction (MI) or Left Ventricular Systolic Dysfunction (LVEF < 40%) 2014 PQRS OPTIONS F INDIVIDUAL MEASURES:
Measure #114: Preventive Care and Screening: Inquiry Regarding Tobacco Use
 Measure #114: Preventive Care and Screening: Inquiry Regarding Tobacco Use 2010 PQRI REPTING OPTIONS F INDIVIDUAL MEASURES: CLAIMS, REGISTRY DESCRIPTION: Percentage of patients aged 18 years or older who
Measure #114: Preventive Care and Screening: Inquiry Regarding Tobacco Use 2010 PQRI REPTING OPTIONS F INDIVIDUAL MEASURES: CLAIMS, REGISTRY DESCRIPTION: Percentage of patients aged 18 years or older who
2018 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY. MEASURE TYPE: Process
 Quality ID #7 (NQF 0070): Coronary Artery Disease (CAD): Beta-Blocker Therapy Prior Myocardial Infarction (MI) or Left Ventricular Systolic Dysfunction (LVEF < 40%) National Quality Strategy Domain: Effective
Quality ID #7 (NQF 0070): Coronary Artery Disease (CAD): Beta-Blocker Therapy Prior Myocardial Infarction (MI) or Left Ventricular Systolic Dysfunction (LVEF < 40%) National Quality Strategy Domain: Effective
DENOMINATOR: All patients 18 and older prescribed opiates for longer than six weeks duration
 Quality ID #412: Documentation of Signed Opioid Treatment Agreement National Quality Strategy Domain: Effective Clinical Care Meaningful Measure Area: Prevention and Treatment of Opioid and Substance Use
Quality ID #412: Documentation of Signed Opioid Treatment Agreement National Quality Strategy Domain: Effective Clinical Care Meaningful Measure Area: Prevention and Treatment of Opioid and Substance Use
New Patient Pain Evaluation
 New Patient Pain Evaluation Name: Date: Using the following symbols, mark the areas of the body diagrams which are affected by your pain: \\ = Stabbing * = Electrical X = Aching N = Numbness 0 = Dull S
New Patient Pain Evaluation Name: Date: Using the following symbols, mark the areas of the body diagrams which are affected by your pain: \\ = Stabbing * = Electrical X = Aching N = Numbness 0 = Dull S
Tobacco Use: Screening & Cessation Intervention
 Tobacco Use: Screening and Cessation Intervention MSSP ACO Measure Tobacco Use: Screening & Cessation Intervention Domain: Preventive Care and Screening ACO 17 PREV- 10 PQRS - 226 NQF 0028 Measure Steward:
Tobacco Use: Screening and Cessation Intervention MSSP ACO Measure Tobacco Use: Screening & Cessation Intervention Domain: Preventive Care and Screening ACO 17 PREV- 10 PQRS - 226 NQF 0028 Measure Steward:
An Interview with a Chiropractor
 An Interview with a Chiropractor Doctor Scott Warner took the time out of his busy schedule to talk to us about chiropractic medicine what it is, what it isn t, and why he chose it as a profession. What
An Interview with a Chiropractor Doctor Scott Warner took the time out of his busy schedule to talk to us about chiropractic medicine what it is, what it isn t, and why he chose it as a profession. What
2018 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY. MEASURE TYPE: Process
 Quality ID #358: Patient-Centered Surgical Risk Assessment and Communication National Quality Strategy Domain: Person and Caregiver-Centered Experience and Outcomes 2018 OPTIONS FOR INDIVIDUAL MEASURES:
Quality ID #358: Patient-Centered Surgical Risk Assessment and Communication National Quality Strategy Domain: Person and Caregiver-Centered Experience and Outcomes 2018 OPTIONS FOR INDIVIDUAL MEASURES:
MedStar Health, Inc. POLICY AND PROCEDURE MANUAL
 MedStar Health, Inc. POLICY AND PROCEDURE MANUAL MP.049.MH Visually Evoked Response Test This policy applies to the following lines of business: MedStar Employee (Select) MedStar MA DSNP CSNP MedStar CareFirst
MedStar Health, Inc. POLICY AND PROCEDURE MANUAL MP.049.MH Visually Evoked Response Test This policy applies to the following lines of business: MedStar Employee (Select) MedStar MA DSNP CSNP MedStar CareFirst
DENOMINATOR: All patients, regardless of age, with a diagnosis of inflammatory bowel disease who initiated an anti-tnf agent
 Quality ID #275: Inflammatory Bowel Disease (IBD): Assessment of Hepatitis B Virus (HBV) Status Before Initiating Anti-TNF (Tumor Necrosis Factor) Therapy National Quality Strategy Domain: Effective Clinical
Quality ID #275: Inflammatory Bowel Disease (IBD): Assessment of Hepatitis B Virus (HBV) Status Before Initiating Anti-TNF (Tumor Necrosis Factor) Therapy National Quality Strategy Domain: Effective Clinical
Quality ID #236 (NQF 0018): Controlling High Blood Pressure National Quality Strategy Domain: Effective Clinical Care
 Quality ID #236 (NQF 0018): Controlling High Blood Pressure National Quality Strategy Domain: Effective Clinical Care 2018 OPTIONS F INDIVIDUAL MEASURES: REGISTRY ONLY MEASURE TYPE: Intermediate Outcome
Quality ID #236 (NQF 0018): Controlling High Blood Pressure National Quality Strategy Domain: Effective Clinical Care 2018 OPTIONS F INDIVIDUAL MEASURES: REGISTRY ONLY MEASURE TYPE: Intermediate Outcome
Quality ID #406: Appropriate Follow-up Imaging for Incidental Thyroid Nodules in Patients National Quality Strategy Domain: Effective Clinical Care
 Quality ID #406: Appropriate Follow-up Imaging for Incidental Thyroid Nodules in Patients National Quality Strategy Domain: Effective Clinical Care 2018 OPTIONS FOR INDIVIDUAL MEASURES: CLAIMS ONLY MEASURE
Quality ID #406: Appropriate Follow-up Imaging for Incidental Thyroid Nodules in Patients National Quality Strategy Domain: Effective Clinical Care 2018 OPTIONS FOR INDIVIDUAL MEASURES: CLAIMS ONLY MEASURE
Measure #383 (NQF 1879): Adherence to Antipsychotic Medications For Individuals with Schizophrenia National Quality Strategy Domain: Patient Safety
 Measure #383 (NQF 1879): Adherence to Antipsychotic Medications For Individuals with Schizophrenia National Quality Strategy Domain: Patient Safety 2017 OPTIONS F INDIVIDUAL MEASURES: REGISTRY ONLY MEASURE
Measure #383 (NQF 1879): Adherence to Antipsychotic Medications For Individuals with Schizophrenia National Quality Strategy Domain: Patient Safety 2017 OPTIONS F INDIVIDUAL MEASURES: REGISTRY ONLY MEASURE
NUMERATOR: Female patients or caregivers counseled at least once a year about how epilepsy and its treatment may affect contraception OR pregnancy
 Quality ID #268 (NQF 1814): Epilepsy: Counseling for Women of Childbearing Potential with Epilepsy National Quality Strategy Domain: Effective Clinical Care 2018 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY
Quality ID #268 (NQF 1814): Epilepsy: Counseling for Women of Childbearing Potential with Epilepsy National Quality Strategy Domain: Effective Clinical Care 2018 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY
DENOMINATOR: All patients aged 18 years and older seen for at least two visits or at least one preventive visit during the measurement period
 Quality ID #431 (NQF 2152): Preventive Care and Screening: Unhealthy Alcohol Use: Screening & Brief Counseling - National Quality Strategy Domain: Community / Population Health 2018 OPTIONS F INDIVIDUAL
Quality ID #431 (NQF 2152): Preventive Care and Screening: Unhealthy Alcohol Use: Screening & Brief Counseling - National Quality Strategy Domain: Community / Population Health 2018 OPTIONS F INDIVIDUAL
Measure #167 (NQF 0114): Coronary Artery Bypass Graft (CABG): Postoperative Renal Failure National Quality Strategy Domain: Effective Clinical Care
 Measure #167 (NQF 0114): Coronary Artery Bypass Graft (CABG): Postoperative Renal Failure National Quality Strategy Domain: Effective Clinical Care 2017 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY MEASURE
Measure #167 (NQF 0114): Coronary Artery Bypass Graft (CABG): Postoperative Renal Failure National Quality Strategy Domain: Effective Clinical Care 2017 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY MEASURE
Medicare Regulations for Chiropractors. Presented by Clinic Pro Software Inc. Marilyn K. Gard. CEO, MBA
 Medicare Regulations for Chiropractors Presented by Clinic Pro Software Inc. Marilyn K. Gard. CEO, MBA Use AT modifier which means active treatment. Claims submitted for Chiropractic manipulative treatment
Medicare Regulations for Chiropractors Presented by Clinic Pro Software Inc. Marilyn K. Gard. CEO, MBA Use AT modifier which means active treatment. Claims submitted for Chiropractic manipulative treatment
1) Scoliosis: Biomechanics and Rationale for Manipulative Treatment. Danbert, RJ. JMPT 1989;
 Peer Reviewed Journals: 1) Scoliosis: Biomechanics and Rationale for Manipulative Treatment. Danbert, RJ. JMPT 1989; 12(1) 38-45. Scoliosis is a biomechanical problem deserving a biomechanical treatment,
Peer Reviewed Journals: 1) Scoliosis: Biomechanics and Rationale for Manipulative Treatment. Danbert, RJ. JMPT 1989; 12(1) 38-45. Scoliosis is a biomechanical problem deserving a biomechanical treatment,
Spine Pain Management Program
 Spine Pain Management Program Please complete the following information: Patient Name: Patient ID Number: Patient DOB: The procedure being requested: Facet Injection Please check the indication (reason)
Spine Pain Management Program Please complete the following information: Patient Name: Patient ID Number: Patient DOB: The procedure being requested: Facet Injection Please check the indication (reason)
OP-14: SIMULTANEOUS USE OF BRAIN COMPUTED TOMOGRAPHY (CT) AND SINUS COMPUTED TOMOGRAPHY (CT)
 OP-14: SIMULTANEOUS USE OF BRAIN COMPUTED TOMOGRAPHY (CT) AND SINUS COMPUTED TOMOGRAPHY (CT) Description of Measure This measure calculates the percentage of Brain CT studies with a simultaneous Sinus
OP-14: SIMULTANEOUS USE OF BRAIN COMPUTED TOMOGRAPHY (CT) AND SINUS COMPUTED TOMOGRAPHY (CT) Description of Measure This measure calculates the percentage of Brain CT studies with a simultaneous Sinus
Measure #131 (NQF 0420): Pain Assessment and Follow-Up National Quality Strategy Domain: Community/Population Health*
 Measure #131 (NQF 0420): Pain Assessment and Follow-Up National Quality Strategy Domain: Community/Population Health* *Please note that PQRS 131 is incorrectly listed under the Communication and Care Coordination
Measure #131 (NQF 0420): Pain Assessment and Follow-Up National Quality Strategy Domain: Community/Population Health* *Please note that PQRS 131 is incorrectly listed under the Communication and Care Coordination
National Imaging Associates, Inc. Clinical guidelines CHIROPRACTIC SERVICES. Original Date: Page 1 of FOR CMS (MEDICARE) MEMBERS ONLY
 National Imaging Associates, Inc. Clinical guidelines CHIROPRACTIC SERVICES CPT4 Codes: Please refer to pages 2-10 LCD ID Number: L35424 Novitas: J-H: DC, DE, MD, NJ, PA J-L: AR, CO, LA, MS, NM, OK, TX
National Imaging Associates, Inc. Clinical guidelines CHIROPRACTIC SERVICES CPT4 Codes: Please refer to pages 2-10 LCD ID Number: L35424 Novitas: J-H: DC, DE, MD, NJ, PA J-L: AR, CO, LA, MS, NM, OK, TX
2) Percentage of adult patients (aged 18 years or older) with a diagnosis of major depression or dysthymia and an
 Quality ID #370 (NQF 0710): Depression Remission at Twelve Months National Quality Strategy Domain: Effective Clinical Care Meaningful Measure Area: Prevention, Treatment, and Management of Mental Health
Quality ID #370 (NQF 0710): Depression Remission at Twelve Months National Quality Strategy Domain: Effective Clinical Care Meaningful Measure Area: Prevention, Treatment, and Management of Mental Health
2018 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY. MEASURE TYPE: Process
 Quality ID #290: Parkinson s Disease: Psychiatric Symptoms Assessment for Patients with Parkinson s Disease National Quality Strategy Domain: Effective Clinical Care 2018 OPTIONS FOR INDIVIDUAL MEASURES:
Quality ID #290: Parkinson s Disease: Psychiatric Symptoms Assessment for Patients with Parkinson s Disease National Quality Strategy Domain: Effective Clinical Care 2018 OPTIONS FOR INDIVIDUAL MEASURES:
Measure #72 (NQF 0385): Colon Cancer: Chemotherapy for AJCC Stage III Colon Cancer Patients National Quality Strategy Domain: Effective Clinical Care
 Measure #72 (NQF 0385): Colon Cancer: Chemotherapy for AJCC Stage III Colon Cancer Patients National Quality Strategy Domain: Effective Clinical Care 2016 PQRS OPTIONS F INDIVIDUAL MEASURES: CLAIMS, REGISTRY
Measure #72 (NQF 0385): Colon Cancer: Chemotherapy for AJCC Stage III Colon Cancer Patients National Quality Strategy Domain: Effective Clinical Care 2016 PQRS OPTIONS F INDIVIDUAL MEASURES: CLAIMS, REGISTRY
Quality ID# 406: Appropriate Follow-up Imaging for Incidental Thyroid Nodules in Patients National Quality Strategy Domain: Effective Clinical Care
 Quality ID# 406: Appropriate Follow-up Imaging for Incidental Thyroid Nodules in Patients National Quality Strategy Domain: Effective Clinical Care 2018 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY MEASURE
Quality ID# 406: Appropriate Follow-up Imaging for Incidental Thyroid Nodules in Patients National Quality Strategy Domain: Effective Clinical Care 2018 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY MEASURE
Measure #164 (NQF 0129): Coronary Artery Bypass Graft (CABG): Prolonged Intubation National Quality Strategy Domain: Effective Clinical Care
 Measure #164 (NQF 0129): Coronary Artery Bypass Graft (CABG): Prolonged Intubation National Quality Strategy Domain: Effective Clinical Care 2016 PQRS OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY DESCRIPTION:
Measure #164 (NQF 0129): Coronary Artery Bypass Graft (CABG): Prolonged Intubation National Quality Strategy Domain: Effective Clinical Care 2016 PQRS OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY DESCRIPTION:
Physical Evidence Chiropractic 7035 Beracasa Way, Suite 103 Boca Raton Florida, Phone# (561) Fax# (561)
 7035 Beracasa Way, Suite 103 Boca Raton Florida, 33433 Phone# (561)674-1217 Fax# (561)361-4999 Date File # PERSONAL HISTORY Last Name First Name middle Address City State Zip Date of Birth Age Social Security
7035 Beracasa Way, Suite 103 Boca Raton Florida, 33433 Phone# (561)674-1217 Fax# (561)361-4999 Date File # PERSONAL HISTORY Last Name First Name middle Address City State Zip Date of Birth Age Social Security
NUMERATOR: All patients with a diagnosis of Parkinson s Disease who were assessed for cognitive impairment or dysfunction in the past 12 months
 Quality ID #291: Parkinson s Disease: Cognitive Impairment or Dysfunction Assessment for Patients with Parkinson s Disease National Quality Strategy Domain: Effective Clinical Care Meaningful Measure Area:
Quality ID #291: Parkinson s Disease: Cognitive Impairment or Dysfunction Assessment for Patients with Parkinson s Disease National Quality Strategy Domain: Effective Clinical Care Meaningful Measure Area:
Measure #117 (NQF 0055): Diabetes: Eye Exam National Quality Strategy Domain: Effective Clinical Care
 Measure #117 (NQF 0055): Diabetes: Eye Exam National Quality Strategy Domain: Effective Clinical Care 2016 PQRS OPTIONS F INDIVIDUAL MEASURES: CLAIMS, REGISTRY DESCRIPTION: Percentage of patients 18-75
Measure #117 (NQF 0055): Diabetes: Eye Exam National Quality Strategy Domain: Effective Clinical Care 2016 PQRS OPTIONS F INDIVIDUAL MEASURES: CLAIMS, REGISTRY DESCRIPTION: Percentage of patients 18-75
Spine Pain Management Program
 Spine Pain Management Program Please complete the following information: Patient Name: Patient ID Number: Patient DOB: The procedure being requested: Epidural Adhesiolysis Please check the indication (reason)
Spine Pain Management Program Please complete the following information: Patient Name: Patient ID Number: Patient DOB: The procedure being requested: Epidural Adhesiolysis Please check the indication (reason)
DESCRIPTION: Percentage of patients aged 18 years and older undergoing isolated CABG surgery who received an IMA graft
 Measure #43 (NQF 0134): Coronary Artery Bypass Graft (CABG): Use of Internal Mammary Artery (IMA) in Patients with Isolated CABG Surgery National Quality Strategy Domain: Effective Clinical Care 2017 OPTIONS
Measure #43 (NQF 0134): Coronary Artery Bypass Graft (CABG): Use of Internal Mammary Artery (IMA) in Patients with Isolated CABG Surgery National Quality Strategy Domain: Effective Clinical Care 2017 OPTIONS
2018 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY. MEASURE TYPE: Process
 Quality ID #325: Adult Major Depressive Disorder (MDD): Coordination of Care of Patients with Specific Comorbid Conditions National Quality Strategy Domain: Communication and Care Coordination 2018 OPTIONS
Quality ID #325: Adult Major Depressive Disorder (MDD): Coordination of Care of Patients with Specific Comorbid Conditions National Quality Strategy Domain: Communication and Care Coordination 2018 OPTIONS
2019 COLLECTION TYPE: MIPS CLINICAL QUALITY MEASURES (CQMS) MEASURE TYPE: Process
 Quality ID #317: Preventive Care and Screening: Screening for High Blood Pressure and Follow-Up Documented National Quality Strategy Domain: Community/Population Health Meaningful Measure Area: Preventive
Quality ID #317: Preventive Care and Screening: Screening for High Blood Pressure and Follow-Up Documented National Quality Strategy Domain: Community/Population Health Meaningful Measure Area: Preventive
SEEDS OF HOPE FAMILY CHIROPRACTIC HEALTH HISTORY
 SEEDS OF HOPE FAMILY CHIROPRACTIC HEALTH HISTORY Welcome! PLEASE PRINT CLEARLY PERSONAL DATA Today s Date First name MI: Last name: Nickname Gender M F Age Date of Birth SS# (optional) Current address
SEEDS OF HOPE FAMILY CHIROPRACTIC HEALTH HISTORY Welcome! PLEASE PRINT CLEARLY PERSONAL DATA Today s Date First name MI: Last name: Nickname Gender M F Age Date of Birth SS# (optional) Current address
Measure #408: Opioid Therapy Follow-up Evaluation National Quality Strategy Domain: Effective Clinical Care
 Measure #408: Opioid Therapy Follow-up Evaluation National Quality Strategy Domain: Effective Clinical Care 2016 PQRS OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY DESCRIPTION: All patients 18 and older
Measure #408: Opioid Therapy Follow-up Evaluation National Quality Strategy Domain: Effective Clinical Care 2016 PQRS OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY DESCRIPTION: All patients 18 and older
CLINICAL MEDICAL POLICY
 Policy Name: Policy Number: Responsible Department(s): CLINICAL MEDICAL POLICY Hyperbaric Oxygen Therapy (HBOT) MP-007-MD-WV Medical Management Provider Notice Date: 04/01/2018; 10/03/2016 Issue Date:
Policy Name: Policy Number: Responsible Department(s): CLINICAL MEDICAL POLICY Hyperbaric Oxygen Therapy (HBOT) MP-007-MD-WV Medical Management Provider Notice Date: 04/01/2018; 10/03/2016 Issue Date:
ChiroCredit.com / OnlineCE.com presents Documentation 101 Part 4 of 10 Instructor: Paul Sherman, DC
 Online Continuing Education Courses www.onlinece.com www.chirocredit.com ChiroCredit.com / OnlineCE.com presents Documentation 101 Part 4 of 10 Instructor: Paul Sherman, DC Important Notice: This download
Online Continuing Education Courses www.onlinece.com www.chirocredit.com ChiroCredit.com / OnlineCE.com presents Documentation 101 Part 4 of 10 Instructor: Paul Sherman, DC Important Notice: This download
2017 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY. MEASURE TYPE: Process
 Measure #143 (NQF 0384): Oncology: Medical and Radiation Pain Intensity Quantified National Quality Strategy Domain: Person and Caregiver-Centered Experience and Outcomes 2017 OPTIONS FOR INDIVIDUAL MEASURES:
Measure #143 (NQF 0384): Oncology: Medical and Radiation Pain Intensity Quantified National Quality Strategy Domain: Person and Caregiver-Centered Experience and Outcomes 2017 OPTIONS FOR INDIVIDUAL MEASURES:
DESCRIPTION: The percentage of adolescents 13 years of age who had the recommended immunizations by their 13th birthday
 Quality ID #394 (NQF 1407): Immunizations for Adolescents National Quality Strategy Domain: Community/Population Health Meaningful Measure Area: Preventive Care 2019 COLLECTION TYPE: MIPS CLINICAL QUALITY
Quality ID #394 (NQF 1407): Immunizations for Adolescents National Quality Strategy Domain: Community/Population Health Meaningful Measure Area: Preventive Care 2019 COLLECTION TYPE: MIPS CLINICAL QUALITY
Spine Pain Management Program
 Spine Pain Management Program Please complete the following information: Patient Name: Patient ID Number: Patient DOB: The procedure being requested: Epidural Injection Please check the indication (reason)
Spine Pain Management Program Please complete the following information: Patient Name: Patient ID Number: Patient DOB: The procedure being requested: Epidural Injection Please check the indication (reason)
2017 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY. MEASURE TYPE: Process
 Measure #127 (NQF 0416): Diabetes Mellitus: Diabetic Foot and Ankle Care, Ulcer Prevention Evaluation of Footwear National Quality Strategy Domain: Effective Clinical Care 2017 OPTIONS FOR INDIVIDUAL MEASURES:
Measure #127 (NQF 0416): Diabetes Mellitus: Diabetic Foot and Ankle Care, Ulcer Prevention Evaluation of Footwear National Quality Strategy Domain: Effective Clinical Care 2017 OPTIONS FOR INDIVIDUAL MEASURES:
Denominator Criteria (Eligible Cases): Patient encounter during the performance period (CPT): 78300, 78305, 78306, 78315, 78320
 Quality ID #147: Nuclear Medicine: Correlation with Existing Imaging Studies for All Patients Undergoing Bone Scintigraphy National Quality Strategy Domain: Communication and Care Coordination 2018 OPTIONS
Quality ID #147: Nuclear Medicine: Correlation with Existing Imaging Studies for All Patients Undergoing Bone Scintigraphy National Quality Strategy Domain: Communication and Care Coordination 2018 OPTIONS
2019 COLLECTION TYPE: MIPS CLINICAL QUALITY MEASURES (CQMS) MEASURE TYPE: Process
 Quality ID #119 (NQF 0062): Diabetes: Medical Attention for Nephropathy National Quality Strategy Domain: Effective Clinical Care Meaningful Measure Area: Management of Chronic Conditions 2019 COLLECTION
Quality ID #119 (NQF 0062): Diabetes: Medical Attention for Nephropathy National Quality Strategy Domain: Effective Clinical Care Meaningful Measure Area: Management of Chronic Conditions 2019 COLLECTION
Measure Reporting via Registry: CPT only copyright 2015 American Medical Association. All rights reserved. 11/17/2015 Page 1 of 9
 Measure #223 (NQF 0428): Functional Deficit: Change in Risk-Adjusted Functional Status for Patients with Neck, Cranium, Mandible, Thoracic Spine, Ribs, or Other General Orthopedic Impairments National
Measure #223 (NQF 0428): Functional Deficit: Change in Risk-Adjusted Functional Status for Patients with Neck, Cranium, Mandible, Thoracic Spine, Ribs, or Other General Orthopedic Impairments National
High Priority Measure? Type NQS Domain None None N/A Yes Outcome Outcome Communication. None None N/A Yes Patient Safety
 Name IDs Title Description Denominator Numerator Denominator Exclusions NPA11 Unplanned Readmission Percentage of patients aged 1 QOD Patients Number of patients aged 1 Denominator Exclusions Following
Name IDs Title Description Denominator Numerator Denominator Exclusions NPA11 Unplanned Readmission Percentage of patients aged 1 QOD Patients Number of patients aged 1 Denominator Exclusions Following
RAINIER VALLEY CHIROPRACTIC P.S th Avenue S. Seattle, WA 98118
 Patient Health History Full Name Date Street Address City & State Zip Phone Number Gender Date of Birth Age SSN How did you hear about our office? Marital Status # of Children? Currently Pregnant? / How
Patient Health History Full Name Date Street Address City & State Zip Phone Number Gender Date of Birth Age SSN How did you hear about our office? Marital Status # of Children? Currently Pregnant? / How
Medical Affairs Policy
 Medical Affairs Policy Service: Back Pain Procedures-Epidural Injection (Caudal Epidural, Selective Nerve Root Block, Interlaminar, Transforaminal, Translaminar Epidural Injection) PUM 250-0015-1706 Medical
Medical Affairs Policy Service: Back Pain Procedures-Epidural Injection (Caudal Epidural, Selective Nerve Root Block, Interlaminar, Transforaminal, Translaminar Epidural Injection) PUM 250-0015-1706 Medical
Quality ID #291: Parkinson s Disease: Cognitive Impairment or Dysfunction Assessment National Quality Strategy Domain: Effective Clinical Care
 Quality ID #291: Parkinson s Disease: Cognitive Impairment or Dysfunction Assessment National Quality Strategy Domain: Effective Clinical Care 2018 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY MEASURE
Quality ID #291: Parkinson s Disease: Cognitive Impairment or Dysfunction Assessment National Quality Strategy Domain: Effective Clinical Care 2018 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY MEASURE
PATIENT HEALTH HISTORY
 Southern Oregon Physical Therapy Associates, Inc. 924 S. Riverside Ave. Medford OR 97501 541.773.7678 Fax 541.773.5517 Email: sopta@integra.net website: southernoregonphysicaltherapy.com Health History-1
Southern Oregon Physical Therapy Associates, Inc. 924 S. Riverside Ave. Medford OR 97501 541.773.7678 Fax 541.773.5517 Email: sopta@integra.net website: southernoregonphysicaltherapy.com Health History-1
For more information call , or visit
 Target Coding ICD-10 List for Chiropractic HEADACHE G43: Migraine G43.0: Migraine without aura (common migraine) G43.009: Migraine without aura, not intractable, without status migrainosus (migraine without
Target Coding ICD-10 List for Chiropractic HEADACHE G43: Migraine G43.0: Migraine without aura (common migraine) G43.009: Migraine without aura, not intractable, without status migrainosus (migraine without
2017 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY. MEASURE TYPE: Process
 Measure #294: Parkinson s Disease: Parkinson s Disease Medical and Surgical Treatment Options Reviewed National Quality Strategy Domain: Communication and Care Coordination 2017 OPTIONS FOR INDIVIDUAL
Measure #294: Parkinson s Disease: Parkinson s Disease Medical and Surgical Treatment Options Reviewed National Quality Strategy Domain: Communication and Care Coordination 2017 OPTIONS FOR INDIVIDUAL
2016 PQRS OPTIONS FOR INDIVIDUAL MEASURES: CLAIMS, REGISTRY
 Measure #22 (NQF 0271): Perioperative Care: Discontinuation of Prophylactic Parenteral Antibiotics (Non-Cardiac Procedures) National Quality Strategy Domain: Patient Safety 2016 PQRS OPTIONS FOR INDIVIDUAL
Measure #22 (NQF 0271): Perioperative Care: Discontinuation of Prophylactic Parenteral Antibiotics (Non-Cardiac Procedures) National Quality Strategy Domain: Patient Safety 2016 PQRS OPTIONS FOR INDIVIDUAL
2018 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY. MEASURE TYPE: Process
 Quality ID #127 (NQF 0416): Diabetes Mellitus: Diabetic Foot and Ankle Care, Ulcer Prevention Evaluation of Footwear National Quality Strategy Domain: Effective Clinical Care 2018 OPTIONS FOR INDIVIDUAL
Quality ID #127 (NQF 0416): Diabetes Mellitus: Diabetic Foot and Ankle Care, Ulcer Prevention Evaluation of Footwear National Quality Strategy Domain: Effective Clinical Care 2018 OPTIONS FOR INDIVIDUAL
Quality ID #249 (NQF 1854): Barrett s Esophagus National Quality Strategy Domain: Effective Clinical Care
 Quality ID #249 (NQF 1854): Barrett s Esophagus National Quality Strategy Domain: Effective Clinical Care 2018 OPTIONS FOR INDIVIDUAL MEASURES: CLAIMS ONLY MEASURE TYPE: Process DESCRIPTION: Percentage
Quality ID #249 (NQF 1854): Barrett s Esophagus National Quality Strategy Domain: Effective Clinical Care 2018 OPTIONS FOR INDIVIDUAL MEASURES: CLAIMS ONLY MEASURE TYPE: Process DESCRIPTION: Percentage
Quality ID #187: Stroke and Stroke Rehabilitation: Thrombolytic Therapy National Quality Strategy Domain: Effective Clinical Care
 Quality ID #187: Stroke and Stroke Rehabilitation: Thrombolytic Therapy National Quality Strategy Domain: Effective Clinical Care 2018 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY MEASURE TYPE: Process
Quality ID #187: Stroke and Stroke Rehabilitation: Thrombolytic Therapy National Quality Strategy Domain: Effective Clinical Care 2018 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY MEASURE TYPE: Process
Measure #394 (NQF 1407): Immunizations for Adolescents National Quality Strategy Domain: Community/Population Health
 Measure #394 (NQF 1407): Immunizations for Adolescents National Quality Strategy Domain: Community/Population Health 2017 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY MEASURE TYPE: Process DESCRIPTION:
Measure #394 (NQF 1407): Immunizations for Adolescents National Quality Strategy Domain: Community/Population Health 2017 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY MEASURE TYPE: Process DESCRIPTION:
