Treatment of Constipation in Adults
|
|
|
- Kelly McGee
- 6 years ago
- Views:
Transcription
1 This Clinical Resource gives subscribers additional insight related to the Recommendations published in April 2017 ~ Resource # Treatment of Constipation in Adults Use this document as a stepwise guide to managing in nonpregnant adults. For information regarding the treatment of in children and pregnant women, see our charts, Treatment of Constipation in Children and GI Med Use in Pregnancy and Lactation. We also have a chart, Treatments for Irritable Bowel Syndrome, which includes meds useful for -predominant IBS (IBS-C). Step 1: Assess for symptoms of 1,2 Difficulty passing stool Incomplete passage of stool Reduced frequency of bowel movements (e.g., <3x/week) Step 2: If constipated patients have alarm symptoms, recommend colon cancer screening 1,3,4 Change in bowel habits after 50 years of age Blood in stool Weight loss of 5 kg (10 lbs) or more in past six months Anemia Family history of colon cancer Refractory Step 3: Assess for underlying causes of 1,3-6 Evaluate for medications that can cause 1. Is the patient taking any of the following? Antacids with aluminum or calcium, anticholinergics (e.g., tricyclic antidepressants, antihistamines, antipsychotics), antidiarrheals, beta-blockers, calcium channel blockers, calcium supplements, nonpotassium sparing diuretics, NSAIDs, oral iron supplements, opioids, 5-HT3 receptor antagonists (e.g., ondansetron). 2. If yes, consider reducing the dose or switching to another medication if risks of the med outweigh its benefits. Evaluate for disease states or metabolic abnormalities that can cause 1. Does the patient have any of the following? Anxiety, autonomic neuropathy, chronic kidney disease, colorectal cancer, dementia, depression, diabetes, diverticulitis, hemorrhoids, hypercalcemia, hypokalemia, hypothyroidism, irritable bowel syndrome, multiple sclerosis, Parkinson s disease, rectal prolapse, stroke, systemic sclerosis (scleroderma). 2. If yes, treat the underlying cause if possible.
2 (Clinical Resource #330407: Page 2 of 6) Step 4: Recommend first-line treatment options Assess for special considerations Patients who have difficulty swallowing, are on fluid restriction, or are immobile: avoid bulk laxatives. 2 Patients with intestinal obstruction: avoid oral laxatives, use enemas or suppositories for patients with fecal impaction. 2,7 Patients taking opioids: use a bowel regimen of an osmotic laxative (PEG 3350, etc) or a stimulant laxative while patients are taking opioids (i.e., initiate bowel regimen at start of opioid therapy). 2 (Note that adding a stool softener [docusate] to a stimulant laxative is widely recommended, although small studies show that adding docusate is not beneficial.) 11 Patients who should avoid straining after surgery, heart attack, etc: use an osmotic laxative (PEG 3350, etc). 13 Patients at risk for electrolyte abnormalities, such as the elderly, those with renal failure or heart failure, and those who take diuretics: avoid osmotic saline laxatives (e.g., magnesium-containing, oral sodium phosphate). 1,2 Implement appropriate lifestyle changes (may take days to weeks for results) Increase dietary fiber to a total of 20 to 25 g per day, via fiber-rich foods such as whole grains, wheat bran, or vegetables. Encourage a target fluid intake of 1.5 to 2 L per day. Start slowly and titrate up over one to two weeks to improve tolerance. 2,4,8 Consider increased physical activity if possible. 8 Counsel on toileting habits such as not holding it, not rushing on the toilet, sitting on the toilet about 30 minutes after breakfast, and sitting on the toilet in a position where the knees are at least as high as the hips. 1,2 Recommend OTC meds if necessary, titrating dose to soft stools 1. Consider a two- to four-week trial of a laxative the patient previously found to be effective Consider a two- to four-week trial of fiber (e.g., psyllium, methylcellulose) or an osmotic laxative, such as PEG 3350 (preferred because of good evidence and good tolerability), sorbitol, lactulose (Rx in U.S.), or magnesium hydroxide. 1,9 3. Consider switching to or adding a stimulant laxative such as bisacodyl or senna if additional effects are needed. 4,9 4. Consider a stimulant laxative as a rescue method with other laxatives if the patient has not had a bowel movement for two days or longer. 3 Step 5: If lifestyle changes and OTC options are not effective after a trial period, consider diagnostic tests (e.g., anorectal manometry, rectal balloon expulsion, etc) to determine cause of 9 Continue to the next section for general information about laxatives and prescription medications for
3 (Clinical Resource #330407: Page 3 of 6) Step 6: Consider therapies targeted to specific diagnoses, including Rx therapies as indicated Rx Therapy Cost* Indication Therapeutic Considerations (Adults Only) Linaclotide 14,15 (Linzess [U.S.]; Constella [Canada]) ~$12/day (U.S.) Chronic idiopathic Do not crush or chew; can sprinkle contents on applesauce or water (can give via nasogastric tube) Give on an empty stomach, 30 minutes Lubiprostone 17 (Amitiza-U.S. only) Methylnaltrexone (Relistor) 18,19 ~$4 to $6/day (Canada) ~$12/day ~$55/day (oral; U.S.) $109/ 12 mg (injectable, U.S.) ~$40/ 12 mg (injectable, Canada; oral not available in Canada) IBS with Chronic idiopathic IBS (women) with Opioid-induced in pain Opioid-induced in pain (U.S.) or with advanced illness, receiving palliative care before first meal of the day Minimal absorption, interactions unlikely Expect improvement in week 1 for bowel symptoms; longer onset for abdominal symptoms. Keep this in mind if treatment delayed (e.g., during transitions of care). No adjustments needed for renal or Protect from moisture; keep in original container with supplied desiccant Swallow whole, do not break or chew No known drug interactions Reduce dose for moderate and severe May not be effective in patients taking methadone. In vitro and preliminary data 26 suggest methadone may interfere with lubiprostone s activation of GI chloride channels. No known drug interactions Reduce dose for moderate and severe Reduce oral (U.S.) and injectable dose for renal impairment Adjust dose for under- or overweight Stop other laxatives; can restart if needed after 3 days (U.S.) May see less response in those on opioids for less than 4 weeks Monitor for opioid withdrawal Oral Give on an empty stomach, at least 30 minutes prior to first meal Subcutaneous Injection Works within 4 hours of injection in up to 50% of patients (median 24 minutes); advise patients to stay close to the toilet Give injection while seated or lying down Protect from light Consider stopping if no response after 4 doses (Canada)
4 Rx Therapy Cost* Indication (Adults Only) Naldemedine 25 ~$11/day Opioid-induced (Symproic-U.S. in only) pain (Clinical Resource #330407: Page 4 of 6) Therapeutic Considerations Take with or without food Avoid with strong CYP3A inducers and other opioid antagonists; monitor with moderate and strong CYP3A4 inhibitors and P-gp inhibitors Avoid with severe May see less response in those on opioids for less than 4 weeks Monitor for opioid withdrawal Naloxegol 20,21 (Movantik) ~$11/day (U.S.) ~$7/day (Canada) Opioid-induced in pain Can crush tablet; mix powder with water for oral or nasogastric tube use Give on an empty stomach, 1 hour prior to first meal or 2 hours after Contraindicated with strong CYP3A4 inhibitors; avoid with moderate inhibitors (if possible) and grapefruit juice Reduce dose for moderate to severe renal impairment, with weak CYP3A4 inhibitors (Canada), with moderate CYP3A4 inhibitors (if use can t be avoided), or if not tolerated Avoid with severe May see less response in those on opioids for less than 4 weeks Stop other laxative therapy; may restart in 3 days if needed Monitor for opioid withdrawal Oxycodone/ naloxone 16 (Targin-Canada only) Plecanatide 22 (Trulance-U.S. only) ~$2 to $6/day ~$12/day Oxycodone for treatment of severe pain (similar pharmacokinetics to OxyContin) Naloxone for relief of opioid-induced Chronic idiopathic Controlled release; swallow whole High first pass metabolism of naloxone results in almost no systemic action Contraindicated in severe renal impairment and moderate to severe Remnants of capsule may appear in stool Holding oxycodone (e.g., during transitions of care) could result in withdrawal symptoms Swallow whole; can crush in applesauce or water (can give via nasogastric tube) Give with or without food Negligible absorption; no expected drug interactions Protect from moisture; keep in original bottle with desiccant * Medication pricing by Elsevier, accessed November 2017 (for U.S. pricing). Cost listed is wholesale acquisition cost (WAC) in U.S. and Canada.
5 Mechanisms of Action of Laxatives:* (Clinical Resource #330407: Page 5 of 6) Osmotic agents (e.g., lactulose [Rx in U.S.], PEG 3350 [Miralax-U.S., RestoraLAX-Canada, others], sorbitol, and saline laxatives [see below]) promote secretion of water into the lumen of the colon and stimulate movement of the bowel. 1,2 The main side effect is diarrhea. Onset is typically from 12 to 96 hours. 2 Glycerin suppository onset is usually within 15 to 60 minutes. 23 Fiber/Bulk agents (e.g., methylcellulose-u.s. only [Citrucel, etc], calcium polycarbophil [FiberCon-U.S., Prodiem Fibre Therapy-Canada, etc], psyllium [Metamucil, etc]) hold water in stool, increase stool weight, increase colonic distension, and improve frequency of bowel movements. 1 The main side effects are bloating and cramping. Onset is typically from 12 to 72 hours. 2 Stimulant laxatives (e.g., bisacodyl [Dulcolax, etc], sennoside [Senokot, etc]) increase intestinal motility and colonic secretions. 1,4 The usual onset with oral formulations is six to ten hours, possibly up to 24 hours. The onset with rectal suppositories is 15 to 60 minutes. The main side effect is cramping. 2 Stool softeners (e.g., docusate [Colace, etc]) improve the interaction of water and solid stool. 1 See our commentary, Docusate: Is It Effective?, for more about the role of docusate. Saline laxatives, (a type of osmotic laxative; e.g., magnesium hydroxide, magnesium citrate, oral sodium phosphate liquid) draw water into intestines and colon by osmosis to increase motility. 2,3 Major side effects are cramping, dehydration, and electrolyte disturbances. 2,12 Onset is 30 minutes to six hours (magnesium hydroxide) and 30 minutes to three hours (magnesium citrate, oral sodium phosphate). 10 The onset of sodium phosphate enema (Fleet, etc) is usually within one to five minutes. 24 *OTC unless otherwise indicated. Be aware of the possibility for confusion due to brand name extensions, and generally recommend that patients choose a product based on generic rather than brand name. Users of this resource are cautioned to use their own professional judgment and consult any other necessary or appropriate sources prior to making clinical judgments based on the content of this document. Our editors have researched the information with input from experts, government agencies, and national organizations. Information and internet links in this article were current as of the date of publication. Project Leaders in preparation of this clinical resource (330407): Stacy A. Hester, R.Ph., BCPS, Associate Editor (Original); Annette Murray, BScPharm (November 2017 update) References 1. Gandell D, Straus SE, Bundookwala M, et al. Treatment of in older people. CMAJ 2013;185: Weitzel KW, Goode JR. Constipation. In: Krinsky DL, Ferreri SP, Hemstreet B, et al, Eds. Handbook of Nonprescription Drugs. 18 th ed. Washington, DC: American Pharmaceutical Association, Bharucha AE, Pemberton JH, Locke GR 3rd. American Gastroenterological Association technical review on. Gastroenterology 2013;144: Lembo A, Camilleri M. Chronic. N Engl J Med 2003;349: Gras-Miralles B, Cremonini F. A critical appraisal of lubiprostone in the treatment of chronic in the elderly. Clin Interv Aging 2013;8: Meek PD, Evang SD, Tadrous M, et al. Overactive bladder drugs and : a meta-analysis of randomized, placebo-controlled trials. Dig Dis Sci 2011;56: Ford AC, Talley NJ. Laxatives for chronic in adults. BMJ 2012;345:e Ternent CA, Bastawrous AL, Morin NA, et al. Practice parameters for the evaluation and management of. Dis Colon Rectum 2007;50: Bharucha AE, Dorn SD, Lembo A, Pressman A. American Gastroenterological Association medical position statement on. Gastroenterology 2013;144: Clinical Resource, Medications for Constipation. Pharmacist s Letter/Prescriber s Letter. May Tarumi Y, Wilson MP, Szafran O, Spooner GR. Randomized, double-blind, placebo-controlled trial of oral docusate in the management of in hospice patients. J Pain Symptom Manage 2013;45: FDA. Drug safety communication: FDA warns of possible harm from exceeding recommended dose of over-the-counter sodium phosphate products to treat. Updated January 15, tm?source=govdelivery&utm_medium= &utm_ source=govdelivery. (Accessed November 3, 2017). 13. McGraw T. Polyethylene glycol 3350 in occasional : a one-week, randomized, placebocontrolled, double-blind trial. World J Gastrointest Pharmacol Ther 2016;7:
6 (Clinical Resource #330407: Page 6 of 6) 14. Product information for Linzess. Allergan USA, Inc. Irvine, CA March Product monograph for Constella. Forest Laboratories Canada, Inc. Markham, ON L6G 0B5. July Product monograph for Targin. Purdue Pharma. Pickering, ON L1W 3W8. May Product information for Amitiza. Takeda Pharmaceuticals America, Inc. Deerfield, IL August Product information for Relistor. Salix Pharmaceuticals, Inc. Bridgewater, NJ August Product monograph for Relistor. Salix Pharmaceuticals, Inc. Raleigh, NC February Product information for Movantik. AstraZeneca Pharmaceuticals LP. Wilmington, DE August Product monograph for Movantik. Knight therapeutics, Inc. Montreal, QC H3Z 3B8. July Product information for Trulance. Synergy Pharmaceuticals, Inc. New York, NY February Product information for glycerin suppository. G & W Labs. South Plainfield, NJ January Product information for enema saline laxative. AmerisourceBergen. Chesterbrook, PA June Product information for Symproic. Purdue Pharma LP. Stamford, CT August Spierings EL, Drossman DA, Cryer B, et al. Efficacy and safety of lubiprostone in opioid-induced : phase 3 study results and pooled analysis of the effect of concomitant methadone use on clinical outcomes. Pain Med 2017 Jul 17. doi: /pm/pnx156. Cite this document as follows: Clinical Resource, Treatment of Constipation in Adults. Pharmacist s Letter/Prescriber s Letter. April Evidence and Recommendations You Can Trust 3120 West March Lane, Stockton, CA ~ TEL (209) ~ FAX (209) Copyright 2017 by Therapeutic Research Center Subscribers to the Letter can get clinical resources, like this one, on any topic covered in any issue by going to PharmacistsLetter.com, PrescribersLetter.com, PharmacyTechniciansLetter.com, or NursesLetter.com
Constipation an Old Friend. Presented by Dr. Keith Harris
 Constipation an Old Friend Presented by Dr. Keith Harris Irregularity and the Tricks of the Trade." CONSTIPATION What is constipation? INFREQUENT BOWEL MOVEMENTS DIFFICULTY DURING DEFECATION SENSATION
Constipation an Old Friend Presented by Dr. Keith Harris Irregularity and the Tricks of the Trade." CONSTIPATION What is constipation? INFREQUENT BOWEL MOVEMENTS DIFFICULTY DURING DEFECATION SENSATION
What Is Constipation?
 CONSTIPATION What Is Constipation? Constipation is when you have infrequent or hard-to-pass bowel movements (meaning they are painful or you have to strain), have hard stools or feel like your bowel movements
CONSTIPATION What Is Constipation? Constipation is when you have infrequent or hard-to-pass bowel movements (meaning they are painful or you have to strain), have hard stools or feel like your bowel movements
CENTENE PHARMACY AND THERAPEUTICS DRUG REVIEW 3Q17 July August
 BRAND NAME Symproic GENERIC NAME Naldemedine MANUFACTURER Shionogi Inc. DATE OF APPROVAL March 23, 2017 PRODUCT LAUNCH DATE Anticipated to launch mid-summer 2017 REVIEW TYPE Review type 1 (RT1): New Drug
BRAND NAME Symproic GENERIC NAME Naldemedine MANUFACTURER Shionogi Inc. DATE OF APPROVAL March 23, 2017 PRODUCT LAUNCH DATE Anticipated to launch mid-summer 2017 REVIEW TYPE Review type 1 (RT1): New Drug
Amitiza. Amitiza (lubiprostone) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.50.22 Subject: Amitiza Page: 1 of 5 Last Review Date: March 16, 2018 Amitiza Description Amitiza (lubiprostone)
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.50.22 Subject: Amitiza Page: 1 of 5 Last Review Date: March 16, 2018 Amitiza Description Amitiza (lubiprostone)
Elderly Man With Chronic Constipation
 Elderly Man With Chronic Constipation Linda Nguyen, MD Director, Neurogastroenterology and Motility Clinical Assistant Professor Stanford University Overview Normal bowel function Defining Constipation:
Elderly Man With Chronic Constipation Linda Nguyen, MD Director, Neurogastroenterology and Motility Clinical Assistant Professor Stanford University Overview Normal bowel function Defining Constipation:
CENTENE PHARMACY AND THERAPEUTICS DRUG REVIEW 2Q17 April May
 BRAND NAME Trulance GENERIC NAME Plecanatide MANUFACTURER Synergy Pharmaceuticals, Inc. DATE OF APPROVAL January 19, 2017 PRODUCT LAUNCH DATE Anticipated in 1Q2017 REVIEW TYPE Review type 1 (RT1): New
BRAND NAME Trulance GENERIC NAME Plecanatide MANUFACTURER Synergy Pharmaceuticals, Inc. DATE OF APPROVAL January 19, 2017 PRODUCT LAUNCH DATE Anticipated in 1Q2017 REVIEW TYPE Review type 1 (RT1): New
daily; available as 10- mg g PO
 Overview of the PRN: The Pain and Palliative Care PRN of ACCP is an organization of pharmacy practitioners, clinical scientists, pharmacy educators, and others. Its mission is to advance pain and palliative
Overview of the PRN: The Pain and Palliative Care PRN of ACCP is an organization of pharmacy practitioners, clinical scientists, pharmacy educators, and others. Its mission is to advance pain and palliative
Primary Care Constipation Guidelines. Version 1 November 2016
 Primary Care Constipation Guidelines Version 1 November 2016 VERSION CONTROL Version Date Amendments made Version 1 November 2016 New guideline Contents 1. Management of constipation in adults: acute and
Primary Care Constipation Guidelines Version 1 November 2016 VERSION CONTROL Version Date Amendments made Version 1 November 2016 New guideline Contents 1. Management of constipation in adults: acute and
Antidiarrheals Antidiarrheal
 Antidiarrheals Major factors in diarrhea Increased motility of the GI tract. Decreased absorption of fluid. Antidiarrheal drugs include: Antimotility agents. Adsorbents. Drugs that modify fluid and electrolyte
Antidiarrheals Major factors in diarrhea Increased motility of the GI tract. Decreased absorption of fluid. Antidiarrheal drugs include: Antimotility agents. Adsorbents. Drugs that modify fluid and electrolyte
Pharmacy Benefit Determination Policy
 Policy Subject: Opioid Induced Constipation Policy Number: SHS PBD11 Category: GI Agents Policy Type: Medical Pharmacy Department: Pharmacy Product (check all that apply): Group HMO/POS Individual HMO/POS
Policy Subject: Opioid Induced Constipation Policy Number: SHS PBD11 Category: GI Agents Policy Type: Medical Pharmacy Department: Pharmacy Product (check all that apply): Group HMO/POS Individual HMO/POS
Constipation An Overview. Definition Physiology of GI tract Etiology Assessment Treatment
 CONSTIPATION Constipation An Overview Definition Physiology of GI tract Etiology Assessment Treatment Definition Constipation = the infrequent passage of hard feces Definition of Infrequent The meaning
CONSTIPATION Constipation An Overview Definition Physiology of GI tract Etiology Assessment Treatment Definition Constipation = the infrequent passage of hard feces Definition of Infrequent The meaning
Drugs Affecting the Gastrointestinal System. Antidiarrheal and Laxatives
 Drugs Affecting the Gastrointestinal System Antidiarrheal and Laxatives Diarrhea Abnormal frequent passage of loose stools or Abnormal passage of stools with increased frequency, fluidity, and weight,
Drugs Affecting the Gastrointestinal System Antidiarrheal and Laxatives Diarrhea Abnormal frequent passage of loose stools or Abnormal passage of stools with increased frequency, fluidity, and weight,
Movantik (naloxegol), Relistor (methylnaltrexone bromide), Symproic (naldemedine)
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.50.06 Subject: Opioid Antagonist Drug Class Page: 1 of 7 Last Review Date: November 30, 2018 Opioid Antagonist
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.50.06 Subject: Opioid Antagonist Drug Class Page: 1 of 7 Last Review Date: November 30, 2018 Opioid Antagonist
Understanding & Alleviating Constipation. Living (Well!) with Gastroparesis Program Warm-Up Class
 Understanding & Alleviating Constipation Living (Well!) with Gastroparesis Program Warm-Up Class Please Remember The information presented is for educational purposes only and is in no way intended as
Understanding & Alleviating Constipation Living (Well!) with Gastroparesis Program Warm-Up Class Please Remember The information presented is for educational purposes only and is in no way intended as
FOOT OFF THE BRAKES. Kerri Novak MD MSc FRCPC. Chronic Constipation: Taking the Foot off the Brakes Dr. Kerri Novak
 CHRONIC CONSTIPATION: TAKING THE FOOT OFF THE BRAKES Kerri Novak MD MSc FRCPC www.seacourses.com 1 OUTLINE Epidemiology i Quality of life Approach Therapies www.seacourses.com 2 DEFINING CHRONIC CONSTIPATION
CHRONIC CONSTIPATION: TAKING THE FOOT OFF THE BRAKES Kerri Novak MD MSc FRCPC www.seacourses.com 1 OUTLINE Epidemiology i Quality of life Approach Therapies www.seacourses.com 2 DEFINING CHRONIC CONSTIPATION
Constipation. What is constipation? What is the criteria for having constipation? What are the different types of constipation?
 What is constipation? is defined as having a bowel movement less than 3 times per week. It is usually associated with hard stools or difficulty passing stools. You may have pain while passing stools or
What is constipation? is defined as having a bowel movement less than 3 times per week. It is usually associated with hard stools or difficulty passing stools. You may have pain while passing stools or
Efficacy and Safety of Lubiprostone. Laura Wozniak February 23, 2010 K30 Monthly Journal Club
 Efficacy and Safety of Lubiprostone Laura Wozniak February 23, 2010 K30 Monthly Journal Club Objectives Brief overview of constipation Review of article Discussion Constipation in Children 3-5% of all
Efficacy and Safety of Lubiprostone Laura Wozniak February 23, 2010 K30 Monthly Journal Club Objectives Brief overview of constipation Review of article Discussion Constipation in Children 3-5% of all
Chronic constipation in the elderly
 Chronic constipation in the elderly 1 Dec,2011 R 2 Natta Asanaleykha Epidemiology Definition Scope The impact of chronic constipation in the elderly Pathophysiology Evaluation the elderly patient with
Chronic constipation in the elderly 1 Dec,2011 R 2 Natta Asanaleykha Epidemiology Definition Scope The impact of chronic constipation in the elderly Pathophysiology Evaluation the elderly patient with
SYMPROIC (naldemedine tosylate) oral capsule
 SYMPROIC (naldemedine tosylate) oral capsule Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This
SYMPROIC (naldemedine tosylate) oral capsule Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This
Opioid constipation treatment dulcolax
 P ford residence southampton, ny Opioid constipation treatment dulcolax Posts about how to relieve constipation instantly written by gbrese1958. Official Web site for MOVANTIK (naloxegol) tablets, for
P ford residence southampton, ny Opioid constipation treatment dulcolax Posts about how to relieve constipation instantly written by gbrese1958. Official Web site for MOVANTIK (naloxegol) tablets, for
3.4 gram/7 gram powder -- $5.63
 MEDICATION COVERAGE POLICY PHARMACY AND THERAPEUTICS ADVISORY COMMITTEE POLICY: Bowel Movements P&T DATE: 9/11/2018 CLASS: Gastrointestinal Disorders REVIEW HISTORY: 12/16, 9/15, 9/12, 5/08 LOB: MCL (MONTH/YEAR)
MEDICATION COVERAGE POLICY PHARMACY AND THERAPEUTICS ADVISORY COMMITTEE POLICY: Bowel Movements P&T DATE: 9/11/2018 CLASS: Gastrointestinal Disorders REVIEW HISTORY: 12/16, 9/15, 9/12, 5/08 LOB: MCL (MONTH/YEAR)
Class Review: Drugs for Constipation
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
OPIOID-INDUCED CONSTIPATION DR ANDREW DAVIES
 OPIOID-INDUCED CONSTIPATION DR ANDREW DAVIES Introduction Introduction Mean faecal weight 128 g / cap / day Mean range 51-796 g Absolute range 15-1505 g Main factors affecting mass are caloric intake,
OPIOID-INDUCED CONSTIPATION DR ANDREW DAVIES Introduction Introduction Mean faecal weight 128 g / cap / day Mean range 51-796 g Absolute range 15-1505 g Main factors affecting mass are caloric intake,
Identify electrolytes that should be monitored whenever prolonged or severe diarrhea is present
 Chapter 35 Drugs Used to Treat Constipation and Diarrhea Learning Objectives State the underlying causes of constipation Explain the meaning of normal bowel habits Cite nine causes of diarrhea Describe
Chapter 35 Drugs Used to Treat Constipation and Diarrhea Learning Objectives State the underlying causes of constipation Explain the meaning of normal bowel habits Cite nine causes of diarrhea Describe
Patient information: Constipation in adults
 Page 1 of 9 Official reprint from UpToDate www.uptodate.com 2011 UpToDate Patient information: Constipation in adults Author Arnold Wald, MD Section Editor J Thomas LaMont, MD Deputy Editor Anne C Travis,
Page 1 of 9 Official reprint from UpToDate www.uptodate.com 2011 UpToDate Patient information: Constipation in adults Author Arnold Wald, MD Section Editor J Thomas LaMont, MD Deputy Editor Anne C Travis,
Protectives and Adsorbents. Inorganic chemistry Course 1 Third year Assist. Lecturer Ahlam A. Shafeeq MSc. Pharmaceutical chemistry
 Protectives and Adsorbents Inorganic chemistry Course 1 Third year 2016-2017 Assist. Lecturer Ahlam A. Shafeeq MSc. Pharmaceutical chemistry Protectives and Adsorbents This group of gastrointestinal agents
Protectives and Adsorbents Inorganic chemistry Course 1 Third year 2016-2017 Assist. Lecturer Ahlam A. Shafeeq MSc. Pharmaceutical chemistry Protectives and Adsorbents This group of gastrointestinal agents
Constipation and Other Gastrointestinal Problems in PD
 Living with PD Constipation and Other Gastrointestinal Problems in PD As you know, Parkinson s disease (PD) affects many body systems, not just movement. This includes the autonomic nervous system that
Living with PD Constipation and Other Gastrointestinal Problems in PD As you know, Parkinson s disease (PD) affects many body systems, not just movement. This includes the autonomic nervous system that
Opioid-Induced Constipation
 Objectives Opioid-Induced Constipation Brianna Jansma, PharmD Alex Smith, PharmD Megan Robinson, PharmD Summarize epidemiology of opioid-induced constipation (OIC) Understand opiates effects on the gastrointestinal
Objectives Opioid-Induced Constipation Brianna Jansma, PharmD Alex Smith, PharmD Megan Robinson, PharmD Summarize epidemiology of opioid-induced constipation (OIC) Understand opiates effects on the gastrointestinal
Opioid-induced constipation a preventable problem
 www.veteransmates.net.au Opioid-induced a preventable problem One of the most common adverse effects of chronic opioid therapy is. 1-4 Up to 95% of patients prescribed an opioid report as a side effect,
www.veteransmates.net.au Opioid-induced a preventable problem One of the most common adverse effects of chronic opioid therapy is. 1-4 Up to 95% of patients prescribed an opioid report as a side effect,
Advancing gastroenterology, improving patient care
 American College of Gastroenterology Advancing gastroenterology, improving patient care Note to Visitors: A fully updated ACG Systematic Review on the Management of Chronic Idiopathic Constipation and
American College of Gastroenterology Advancing gastroenterology, improving patient care Note to Visitors: A fully updated ACG Systematic Review on the Management of Chronic Idiopathic Constipation and
Movantik (naloxegol), Relistor (methylnaltrexone bromide), Symproic (naldemedine)
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.50.06 Subject: Opioid Antagonist Drug Class Page: 1 of 5 Last Review Date: June 22, 2017 Opioid Antagonist
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.50.06 Subject: Opioid Antagonist Drug Class Page: 1 of 5 Last Review Date: June 22, 2017 Opioid Antagonist
Understanding the Prevalence and Impact of Constipation in Canada. A Special Report from the Canadian Digestive Health Foundation
 Understanding the Prevalence and Impact of Constipation in Canada A Special Report from the Canadian Digestive Health Foundation February 2014 Understanding the Prevalence and Impact of Constipation in
Understanding the Prevalence and Impact of Constipation in Canada A Special Report from the Canadian Digestive Health Foundation February 2014 Understanding the Prevalence and Impact of Constipation in
Xifaxan, Lotronex and Viberzi Prior Authorization and Quantity Limit Program Summary
 Xifaxan, Lotronex and Viberzi Prior Authorization and Quantity Limit Program Summary FDA APPROVED INDICATIONS DOSAGE 1,2 Lotronex (alosetron) a Indication For women with severe diarrheapredominant irritable
Xifaxan, Lotronex and Viberzi Prior Authorization and Quantity Limit Program Summary FDA APPROVED INDICATIONS DOSAGE 1,2 Lotronex (alosetron) a Indication For women with severe diarrheapredominant irritable
Is one of the most common chronic disorders. causing patients to seek medical treatment.
 ILOs After this lecture you should be able to : Define IBS Identify causes and risk factors of IBS Determine the appropriate therapeutic options for IBS Is one of the most common chronic disorders causing
ILOs After this lecture you should be able to : Define IBS Identify causes and risk factors of IBS Determine the appropriate therapeutic options for IBS Is one of the most common chronic disorders causing
Revisionary Capsulorrhaphy Surgery Aftercare Guidelines Following Day Date:
 Surgery Sleep in a reclined positon Medications- take as directed Soft Diet and liquids Stool softener/high Fiber foods Leave on all dressing Surgical Stockings First Night Revisionary Capsulorrhaphy Surgery
Surgery Sleep in a reclined positon Medications- take as directed Soft Diet and liquids Stool softener/high Fiber foods Leave on all dressing Surgical Stockings First Night Revisionary Capsulorrhaphy Surgery
Breast Augmentation Saline Overfilled Surgery Aftercare Guidelines
 Surgery Breast Augmentation Saline Overfilled Surgery Aftercare Guidelines First Night Following Day 3-5 Days 1 st Post op Visit 7-10 Days 2 nd Post op Visit 2 Weeks Post op 4-6 Weeks Post op 8-12Weeks
Surgery Breast Augmentation Saline Overfilled Surgery Aftercare Guidelines First Night Following Day 3-5 Days 1 st Post op Visit 7-10 Days 2 nd Post op Visit 2 Weeks Post op 4-6 Weeks Post op 8-12Weeks
ABC s of Bowel Management
 ABC s of Bowel Management Spina Bifida Education Day Conference SBA of Northeastern New York Albany, New York April 14, 2018 Eric Levey, M.D. Pediatrics & Neurodevelopmental Disabilities Chief Medical
ABC s of Bowel Management Spina Bifida Education Day Conference SBA of Northeastern New York Albany, New York April 14, 2018 Eric Levey, M.D. Pediatrics & Neurodevelopmental Disabilities Chief Medical
OCTOBER 7-10 PHILADELPHIA, PENNSYLVANIA
 OMED 17 OCTOBER 7-10 PHILADELPHIA, PENNSYLVANIA 29.5 Category 1-A CME credits anticipated ACOFP / AOA s 122 nd Annual Osteopathic Medical Conference & Exposition Joint Session with ACOFP and Cleveland
OMED 17 OCTOBER 7-10 PHILADELPHIA, PENNSYLVANIA 29.5 Category 1-A CME credits anticipated ACOFP / AOA s 122 nd Annual Osteopathic Medical Conference & Exposition Joint Session with ACOFP and Cleveland
A patient s guide to the. management of constipation following surgery
 A patient s guide to the management of constipation following surgery This leaflet has been designed to give you more information about using laxatives after your surgery. What is constipation? After your
A patient s guide to the management of constipation following surgery This leaflet has been designed to give you more information about using laxatives after your surgery. What is constipation? After your
Opioid Use in Palliative Care
 Opioid Use in Palliative Care Relief of pain is one of the core components of palliative care 1,2 Up to 69% of patients with advanced cancer experience pain 3 ~65% of patients dying from nonmalignant disease
Opioid Use in Palliative Care Relief of pain is one of the core components of palliative care 1,2 Up to 69% of patients with advanced cancer experience pain 3 ~65% of patients dying from nonmalignant disease
The Road to Opioid-Induced Constipation: Pathophysiology and Impact Kenneth C. Jackson, II, PharmD, CPE
 Disclaimer This slide deck in its original and unaltered format is for educational purposes and is current as of March 2015. The content and views presented in this educational activity are those of the
Disclaimer This slide deck in its original and unaltered format is for educational purposes and is current as of March 2015. The content and views presented in this educational activity are those of the
MANAGING CONSTIPATION
 MANAGING CONSTIPATION INFORMATION FOR OLDER ADULTS, FAMILIES, AND CAREGIVERS READ THIS PAMPHLET TO LEARN: What Constipation is. The Symptoms of Constipation. How to Help Manage Constipation. WHAT IS CONSTIPATION?
MANAGING CONSTIPATION INFORMATION FOR OLDER ADULTS, FAMILIES, AND CAREGIVERS READ THIS PAMPHLET TO LEARN: What Constipation is. The Symptoms of Constipation. How to Help Manage Constipation. WHAT IS CONSTIPATION?
Protocol to support reducing the use of and the effective use of laxatives. HaRD CCG employed Pharmacists and Medicines Optimisation Technicians.
 NY&AWC Medicines Management Team Protocol to support reducing the use of and the effective use of laxatives Applies to HaRD CCG employed Pharmacists and Medicines Optimisation Technicians. These protocols
NY&AWC Medicines Management Team Protocol to support reducing the use of and the effective use of laxatives Applies to HaRD CCG employed Pharmacists and Medicines Optimisation Technicians. These protocols
Movantik (naloxegol), Relistor (methylnaltrexone bromide)
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.50.06 Subject: Opioid Antagonist Drug Class Page: 1 of 5 Last Review Date: December 2, 2016 Opioid Antagonist
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.50.06 Subject: Opioid Antagonist Drug Class Page: 1 of 5 Last Review Date: December 2, 2016 Opioid Antagonist
Primary Management of Irritable Bowel Syndrome
 Primary Management of Irritable Bowel Syndrome Jasmine Zia, MD Acting Instructor, Division of Gastroenterology Current Concepts in Drug Therapy CME Course April 23, 2015 Irritable Bowel Syndrome (IBS)
Primary Management of Irritable Bowel Syndrome Jasmine Zia, MD Acting Instructor, Division of Gastroenterology Current Concepts in Drug Therapy CME Course April 23, 2015 Irritable Bowel Syndrome (IBS)
Constipation. Self-study course
 Constipation Self-study course 2 Course objectives: At the end of this course you will be able to: 1. Define the term constipation 2. Explain three reasons why older adults are at greater risk for constipation
Constipation Self-study course 2 Course objectives: At the end of this course you will be able to: 1. Define the term constipation 2. Explain three reasons why older adults are at greater risk for constipation
Purchase (over the counter):
 . COLONOSCOPY PREPARATION LOCATION: SOUTHDALE MEDICAL CENTER Appointment Date: Time: 6545 FRANCE AVE S. SUITE 290 EDINA, MN 55435 You are scheduled for a colonoscopy, a procedure in which a doctor examines
. COLONOSCOPY PREPARATION LOCATION: SOUTHDALE MEDICAL CENTER Appointment Date: Time: 6545 FRANCE AVE S. SUITE 290 EDINA, MN 55435 You are scheduled for a colonoscopy, a procedure in which a doctor examines
10/10/16. Disclosures. Educational Objectives
 Nimish Vakil, MD, FACP, FACG, AGAF, FASGE Clinical Adjunct Professor University of Wisconsin Madison, Wisconsin Disclosures All faculty, course directors, planning committee, content reviewers and others
Nimish Vakil, MD, FACP, FACG, AGAF, FASGE Clinical Adjunct Professor University of Wisconsin Madison, Wisconsin Disclosures All faculty, course directors, planning committee, content reviewers and others
National Naval Medical Center Bethesda, MD
 Brooks Cash. MD National Naval Medical Center Bethesda, MD Multicomponen nt Approach to Functional GI Disorders Establish therapeutic relations ship Assess patient s medical h istory, personality, and
Brooks Cash. MD National Naval Medical Center Bethesda, MD Multicomponen nt Approach to Functional GI Disorders Establish therapeutic relations ship Assess patient s medical h istory, personality, and
Constipation a symptom NOT a disease! Bowels/Constipation Documentation The A-B-C-D. Documentation: Bowels/Constipation
 Documentation The A-B-C-D Terry Broda, RN(EC), BScN, NPHC,CDDN Elizabeth Kacew, RN(EC), MScN, NP-PHC October 30, 2014 Constipation a symptom NOT a disease! 1 How serious a problem? 50-85% of older people
Documentation The A-B-C-D Terry Broda, RN(EC), BScN, NPHC,CDDN Elizabeth Kacew, RN(EC), MScN, NP-PHC October 30, 2014 Constipation a symptom NOT a disease! 1 How serious a problem? 50-85% of older people
Opioids: Safe Use and Side Effects
 Opioids: Safe Use and Side Effects Your doctor has prescribed an opioid (OHpee-oyd). This drug can control moderate to severe pain without increased risk of bleeding and kidney problems. Always take the
Opioids: Safe Use and Side Effects Your doctor has prescribed an opioid (OHpee-oyd). This drug can control moderate to severe pain without increased risk of bleeding and kidney problems. Always take the
Constipation in Children. Amani Al Hajeri, MD, CABFM, IBFM, MSc MG*
 1 Bahrain Medical Bulletin, Vol. 33, No. 4, December 2011 Education-Family Physician Corner Constipation in Children Amani Al Hajeri, MD, CABFM, IBFM, MSc MG* Constipation in pediatrics age group is frequently
1 Bahrain Medical Bulletin, Vol. 33, No. 4, December 2011 Education-Family Physician Corner Constipation in Children Amani Al Hajeri, MD, CABFM, IBFM, MSc MG* Constipation in pediatrics age group is frequently
Scoop on the poop: Constipation in the Elderly
 Scoop on the poop: Constipation in the Elderly Laurie Archbald-Pannone, MD, MPH Assistant Professor Division of General Medicine, Geriatrics, & Palliative Care Division of Infectious Diseases and International
Scoop on the poop: Constipation in the Elderly Laurie Archbald-Pannone, MD, MPH Assistant Professor Division of General Medicine, Geriatrics, & Palliative Care Division of Infectious Diseases and International
Constipation. Myeloma Infosheet Series. Symptoms and complications. Infoline:
 Constipation This Infosheet explains what constipation is, what causes it in myeloma patients, what the symptoms of it are, how it is treated and some tips for self-management. What is constipation? Constipation
Constipation This Infosheet explains what constipation is, what causes it in myeloma patients, what the symptoms of it are, how it is treated and some tips for self-management. What is constipation? Constipation
Constipation: Assessment and Management
 Constipation: Assessment and Management Page 1 of 8 Table of Contents Introduction for the Learner (Resident/Oncology Fellow).............................................. 2 Introduction for Clinical Faculty
Constipation: Assessment and Management Page 1 of 8 Table of Contents Introduction for the Learner (Resident/Oncology Fellow).............................................. 2 Introduction for Clinical Faculty
Constipation. National Digestive Diseases Information Clearinghouse
 Constipation National Digestive Diseases Information Clearinghouse National Institute of Diabetes and Digestive and Kidney Diseases NATIONAL INSTITUTES OF HEALTH Constipation is defined as having a bowel
Constipation National Digestive Diseases Information Clearinghouse National Institute of Diabetes and Digestive and Kidney Diseases NATIONAL INSTITUTES OF HEALTH Constipation is defined as having a bowel
Slide #43. Functional Disorders - An Update 11/8/ MA ACP Annual Scientific Meeting. Functional Disorders: An Update
 Functional Disorders: An Update Anthony Lembo, M.D. Associate Professor of Medicine Beth Israel Deaconess Medical Center Harvard Medical School Boston, MA Disclosure of Financial Relationships Anthony
Functional Disorders: An Update Anthony Lembo, M.D. Associate Professor of Medicine Beth Israel Deaconess Medical Center Harvard Medical School Boston, MA Disclosure of Financial Relationships Anthony
I ve had it with you and your emotional constipation.
 I ve had it with you and your emotional constipation. Washington Irving If Mother had to be told not to put the entire brick of ivory up Junior s hindquarters, constipation is the least of his problems.
I ve had it with you and your emotional constipation. Washington Irving If Mother had to be told not to put the entire brick of ivory up Junior s hindquarters, constipation is the least of his problems.
Constipation. Disease Review
 bowel. 1 Constipation is a symptom, not a disease. Almost everyone experiences Disease Review Constipation Introduction: Constipation is exceedingly common and is rarely associated with mortality in developed
bowel. 1 Constipation is a symptom, not a disease. Almost everyone experiences Disease Review Constipation Introduction: Constipation is exceedingly common and is rarely associated with mortality in developed
Authors and Disclosures
 Role of Carbon Dioxide-Releasing Suppositories in the Treatment of Chronic Functional Constipation A Double-Blind, Randomised, Placebo-Controlled Trial M. Lazzaroni; V. Casini; G. Bianchi Porro Authors
Role of Carbon Dioxide-Releasing Suppositories in the Treatment of Chronic Functional Constipation A Double-Blind, Randomised, Placebo-Controlled Trial M. Lazzaroni; V. Casini; G. Bianchi Porro Authors
David Leff, DO. April 13, Disclosure. I have the following financial relationships to disclosure:
 David Leff, DO AOMA 94 th Annual Convention April 13, 2016 Disclosure I have the following financial relationships to disclosure: Speaker s Bureau: Allergan Labs, Takeda Pharmaceutical, Valeant Pharmaceutical
David Leff, DO AOMA 94 th Annual Convention April 13, 2016 Disclosure I have the following financial relationships to disclosure: Speaker s Bureau: Allergan Labs, Takeda Pharmaceutical, Valeant Pharmaceutical
Irritable Bowel Disease. Dr. Alexandra Ilnyckyj MD
 Irritable Bowel Disease Dr. Alexandra Ilnyckyj MD Exactly what is IBS? Common condition affecting mostly women Symptoms are variable but they reflect altered gut movement (motility) and sensation Commonly
Irritable Bowel Disease Dr. Alexandra Ilnyckyj MD Exactly what is IBS? Common condition affecting mostly women Symptoms are variable but they reflect altered gut movement (motility) and sensation Commonly
Constipation. What is constipation? What causes constipation? How common is constipation?
 Constipation What is constipation? Constipation is the word we use to describe any difficulty having a bowel movement. This does not just mean infrequent bowel movements, (two or fewer bowel movements
Constipation What is constipation? Constipation is the word we use to describe any difficulty having a bowel movement. This does not just mean infrequent bowel movements, (two or fewer bowel movements
Current Pharmacological Treatment Options in Chronic Constipation and IBS with Constipation
 Current Pharmacological Treatment Options in Chronic Constipation and IBS with Constipation Anthony Lembo, M.D. Associate Professor of Medicine Harvard Medical School Director, GI Motility Center Beth
Current Pharmacological Treatment Options in Chronic Constipation and IBS with Constipation Anthony Lembo, M.D. Associate Professor of Medicine Harvard Medical School Director, GI Motility Center Beth
management of this patient within the Enhanced Primary Care Pathway is appropriate, without need for specialist consultation at this time.
 TRIAGE CATEGORY: Enhanced Primary Care Pathway CONSTIPATION Dear Colleague, The clinical and diagnostic information you have provided for the above-named patient is consistent with a diagnosis of chronic
TRIAGE CATEGORY: Enhanced Primary Care Pathway CONSTIPATION Dear Colleague, The clinical and diagnostic information you have provided for the above-named patient is consistent with a diagnosis of chronic
Pharmacology. Drugs that Affect the Gastrointestinal System
 Pharmacology Drugs that Affect the Gastrointestinal System Topics Peptic Ulcer Disease Constipation Diarrhea Emesis Digestion Peptic Ulcer Disease Factors that Increase Acidity Factors that Protect Against
Pharmacology Drugs that Affect the Gastrointestinal System Topics Peptic Ulcer Disease Constipation Diarrhea Emesis Digestion Peptic Ulcer Disease Factors that Increase Acidity Factors that Protect Against
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Relistor) Reference Number: CP.CPA.274 Effective Date: 11.16.16 Last Review Date: 11.17 Line of Business: Commercial Revision Log See Important Reminder at the end of this policy for
Clinical Policy: (Relistor) Reference Number: CP.CPA.274 Effective Date: 11.16.16 Last Review Date: 11.17 Line of Business: Commercial Revision Log See Important Reminder at the end of this policy for
Management of Neurogenic Bowel Dysfunction. Fiona Paul, DNP, RN, CPNP Center for Motility and Functional Gastrointestinal Disorders
 Management of Neurogenic Bowel Dysfunction Fiona Paul, DNP, RN, CPNP Center for Motility and Functional Gastrointestinal Disorders DEFECATION Delivery of colon contents to the rectum Rectal compliance
Management of Neurogenic Bowel Dysfunction Fiona Paul, DNP, RN, CPNP Center for Motility and Functional Gastrointestinal Disorders DEFECATION Delivery of colon contents to the rectum Rectal compliance
Colonoscopy Preparation. Daniel Sussman, MD 19 February 2010 SGNA Course
 Colonoscopy Preparation Daniel Sussman, MD 19 February 2010 SGNA Course Objectives Importance of preparation Types of preps Comparative evidence behind prep choice Efficacy Tolerability Safety profiles
Colonoscopy Preparation Daniel Sussman, MD 19 February 2010 SGNA Course Objectives Importance of preparation Types of preps Comparative evidence behind prep choice Efficacy Tolerability Safety profiles
Constipation. H. David Vargas, MD. Overview
 Constipation H. David Vargas, MD Overview Constipation is a very common complaint affecting upwards of 15% of all Americans. Fortunately, constipation usually is simple to avoid and easy to treat when
Constipation H. David Vargas, MD Overview Constipation is a very common complaint affecting upwards of 15% of all Americans. Fortunately, constipation usually is simple to avoid and easy to treat when
Aging Persons with Intellectual Developmental Disorders (IDD): Constipation KEYPOINTS OVERVIEW
 Aging Persons with Intellectual Developmental Disorders (IDD): Constipation KEYPOINTS A major medical conditions that commonly is seen among persons with IDD and may lead to serious complications is constipation.
Aging Persons with Intellectual Developmental Disorders (IDD): Constipation KEYPOINTS A major medical conditions that commonly is seen among persons with IDD and may lead to serious complications is constipation.
Compounding KI for Radiation Exposure
 PL Detail-Document #270323 This PL Detail-Document gives subscribers additional insight related to the Recommendations published in PHARMACIST S LETTER / PRESCRIBER S LETTER March 2011 Compounding KI for
PL Detail-Document #270323 This PL Detail-Document gives subscribers additional insight related to the Recommendations published in PHARMACIST S LETTER / PRESCRIBER S LETTER March 2011 Compounding KI for
Using Evidence Based Medicine to Ethically Provide End of Life Symptom Control
 Using Evidence Based Medicine to Ethically Provide End of Life Symptom Control Erin Zimny, MD Emergency Medicine Hospice and Palliative Medicine Henry Ford Hospital Disclosures I do NOT have any financial
Using Evidence Based Medicine to Ethically Provide End of Life Symptom Control Erin Zimny, MD Emergency Medicine Hospice and Palliative Medicine Henry Ford Hospital Disclosures I do NOT have any financial
Comparison of strategies and goals for treatment of chronic constipation among gastroenterologists and general practitioners
 ORIGINAL ARTICLE Annals of Gastroenterology (18) 31, 1-6 Comparison of strategies and goals for treatment of chronic constipation among gastroenterologists and general practitioners Dan Carter a,c, Eytan
ORIGINAL ARTICLE Annals of Gastroenterology (18) 31, 1-6 Comparison of strategies and goals for treatment of chronic constipation among gastroenterologists and general practitioners Dan Carter a,c, Eytan
Inhaled Corticosteroid Dose Comparison in Asthma
 This Clinical Resource gives subscribers additional insight related to the Recommendations published in April 2017 ~ Resource #330402 Inhaled Corticosteroid Dose Comparison in Asthma The chart below provides
This Clinical Resource gives subscribers additional insight related to the Recommendations published in April 2017 ~ Resource #330402 Inhaled Corticosteroid Dose Comparison in Asthma The chart below provides
Irritable Bowel Syndrome Now. George M. Logan, MD Friday, May 5, :35 4:05 PM
 Irritable Bowel Syndrome Now George M. Logan, MD Friday, May 5, 2017 3:35 4:05 PM Dr. Logan indicated no potential conflict of interest to this presentation. He does not intend to discuss any unapproved/investigative
Irritable Bowel Syndrome Now George M. Logan, MD Friday, May 5, 2017 3:35 4:05 PM Dr. Logan indicated no potential conflict of interest to this presentation. He does not intend to discuss any unapproved/investigative
Common Gastrointestinal Problems in the Elderly
 Common Gastrointestinal Problems in the Elderly Brian Viviano, D.O. Objectives Understand the pathophysiology, clinical manifestations, diagnosis and management of GI diseases of the elderly. Differentiate
Common Gastrointestinal Problems in the Elderly Brian Viviano, D.O. Objectives Understand the pathophysiology, clinical manifestations, diagnosis and management of GI diseases of the elderly. Differentiate
Active ingredients per ml: Docusate sodium 1 mg/sorbitol solution (70%) (crystallising) 357 mg Structural formula: Docusate.
 NAME OF THE MEDICINE KLYX Enema Active ingredients per ml: Docusate sodium 1 mg/sorbitol solution (70%) (crystallising) 357 mg Structural formula: Docusate Sorbitol C20H37NaO7S MW: 444.56 CAS no: 577-11-7
NAME OF THE MEDICINE KLYX Enema Active ingredients per ml: Docusate sodium 1 mg/sorbitol solution (70%) (crystallising) 357 mg Structural formula: Docusate Sorbitol C20H37NaO7S MW: 444.56 CAS no: 577-11-7
LESSON ASSIGNMENT. After completing this lesson, you should be able to:
 LESSON ASSIGNMENT LESSON 4 Cathartics. LESSON ASSIGNMENT Paragraphs 4-1 through 4-10. LESSON OBJECTIVES After completing this lesson, you should be able to: 4-1. Given a group of statements, select the
LESSON ASSIGNMENT LESSON 4 Cathartics. LESSON ASSIGNMENT Paragraphs 4-1 through 4-10. LESSON OBJECTIVES After completing this lesson, you should be able to: 4-1. Given a group of statements, select the
Opioid-Induced Constipation: Update on Prevention and Management EDUCATIONALPROGRAM
 EDUCATIONALPROGRAM Recognizing i the Growing Burden of OIC Bill H. McCarberg, MD Founder, Chronic Pain Management Program Kaiser Permanente San Diego Adjunct Assistant Clinical Professor University of
EDUCATIONALPROGRAM Recognizing i the Growing Burden of OIC Bill H. McCarberg, MD Founder, Chronic Pain Management Program Kaiser Permanente San Diego Adjunct Assistant Clinical Professor University of
Objectives Brand name Generic name Primary indication, drug classes Dosage forms Patient counseling information Important side effects and contraindic
 laxatives Hawler medical university Collage of pharmacy/ fourth stage /pharmacy practice Sham A. Talat Shareef (B.Sc. Msc. clinical pharmacy) 2017-2018 Sham_talat@yahoo.com Head of Department Of Clinical
laxatives Hawler medical university Collage of pharmacy/ fourth stage /pharmacy practice Sham A. Talat Shareef (B.Sc. Msc. clinical pharmacy) 2017-2018 Sham_talat@yahoo.com Head of Department Of Clinical
BNF CHAPTER 1: GASTRO-INTESTINAL SYSTEM
 BNF CHAPTER 1: GASTRO-INTESTINAL SYSTEM 1 ANTACIDS Maalox and Peptac are effective antacids that can be bought Over the Counter for self-limiting symptoms. Choice Product Cost Effective Options &Comments
BNF CHAPTER 1: GASTRO-INTESTINAL SYSTEM 1 ANTACIDS Maalox and Peptac are effective antacids that can be bought Over the Counter for self-limiting symptoms. Choice Product Cost Effective Options &Comments
Disclosures. Objectives. Pre-Test Question 1. Pre-Test Question 2. Pre-Test Question 3 9/23/2016
 Disclosures Beating the Bowel Blues: An Update on the Treatment of Irritable Bowel Syndrome Matthew Nelson, PharmDBCPS, Roosevelt University College of Pharmacy Matthew Nelson declares no conflicts of
Disclosures Beating the Bowel Blues: An Update on the Treatment of Irritable Bowel Syndrome Matthew Nelson, PharmDBCPS, Roosevelt University College of Pharmacy Matthew Nelson declares no conflicts of
Bowel Health in Adults
 Bowel Health in Adults Patient Information Contact Details: Bladder and Bowel Specialist Nurse Service First Floor New Alderley House Macclesfield District General Hospital Victoria Road Macclesfield SK10
Bowel Health in Adults Patient Information Contact Details: Bladder and Bowel Specialist Nurse Service First Floor New Alderley House Macclesfield District General Hospital Victoria Road Macclesfield SK10
CONSTIPATION OR STYPSIS. Prof. G. Zuliani
 CONSTIPATION OR STYPSIS Prof. G. Zuliani How Do We Define Constipation? The American College of Gastroenterology (ACG) definition of constipation: Unsatisfactory defecation characterized by infrequent
CONSTIPATION OR STYPSIS Prof. G. Zuliani How Do We Define Constipation? The American College of Gastroenterology (ACG) definition of constipation: Unsatisfactory defecation characterized by infrequent
ACTIVITY DESCRIPTION FACULTY. Opioid Use in Palliative Care. Opioid Analgesic Use in Chronic, Non-cancer Pain
 ACTIVITY DESCRIPTION Target Audience This continuing pharmacy education activity is planned to meet the needs of pharmacists in a variety of practice settings, including large and small healthcare systems,
ACTIVITY DESCRIPTION Target Audience This continuing pharmacy education activity is planned to meet the needs of pharmacists in a variety of practice settings, including large and small healthcare systems,
MOVICOL Junior Chocolate Flavour Powder for Solution (macrogol 3350)
 MOVICOL Junior Chocolate Flavour Powder for Solution (macrogol 3350) Product Name: MOVICOL Junior Chocolate Flavour Product Description: Each sachet of MOVICOL Junior Chocolate contains: Macrogol 3350
MOVICOL Junior Chocolate Flavour Powder for Solution (macrogol 3350) Product Name: MOVICOL Junior Chocolate Flavour Product Description: Each sachet of MOVICOL Junior Chocolate contains: Macrogol 3350
Efficacy and Safety of Traditional Medical Therapies for Chronic Constipation: Systematic Review
 American Journal of Gastroenterology ISSN 0002-9270 C 2005 by Am. Coll. of Gastroenterology doi: 10.1111/j.1572-0241.2005.40925.x Published by Blackwell Publishing Efficacy and Safety of Traditional Medical
American Journal of Gastroenterology ISSN 0002-9270 C 2005 by Am. Coll. of Gastroenterology doi: 10.1111/j.1572-0241.2005.40925.x Published by Blackwell Publishing Efficacy and Safety of Traditional Medical
Exclusion Criteria. Required Medical Documentation. Specified reasons for denial (if any), other than not meeting criteria requirements.
 Partnership HealthPlan of California: New and Revised, 3nd Quarter P & T 2016 Effective 10/1/2016 Generic substitution required when there is an FDA approved generic equivalent; additional criteria apply
Partnership HealthPlan of California: New and Revised, 3nd Quarter P & T 2016 Effective 10/1/2016 Generic substitution required when there is an FDA approved generic equivalent; additional criteria apply
MOVICOL Liquid Orange Flavour Concentrate for Oral Solution (macrogol 3350)
 MOVICOL Liquid Orange Flavour Concentrate for Oral Solution (macrogol 3350) NAME OF THE MEDICINE: MOVICOL Liquid Orange Flavour, Concentrate for Oral Solution. DESCRIPTION: A clear colourless solution.
MOVICOL Liquid Orange Flavour Concentrate for Oral Solution (macrogol 3350) NAME OF THE MEDICINE: MOVICOL Liquid Orange Flavour, Concentrate for Oral Solution. DESCRIPTION: A clear colourless solution.
Medications and ostomies. Sarah Drost, BSc Pharm, RPh, ACPR
 Medications and ostomies Sarah Drost, BSc Pharm, RPh, ACPR Overview 1. Medication absorption 2. Medication forms 1. Oral medications 2. Non-oral medication routes 3. Monitoring medications 4. Specific
Medications and ostomies Sarah Drost, BSc Pharm, RPh, ACPR Overview 1. Medication absorption 2. Medication forms 1. Oral medications 2. Non-oral medication routes 3. Monitoring medications 4. Specific
Don t let. Irritable irregularity 2.0
 Don t let constipation Over 2 million Americans visit their health care providers each year for constipationrelated problems, yet constipation is often overlooked until it becomes serious. We ll help you
Don t let constipation Over 2 million Americans visit their health care providers each year for constipationrelated problems, yet constipation is often overlooked until it becomes serious. We ll help you
Constipation Information Leaflet THE DIGESTIVE SYSTEM. gutscharity.org.uk
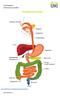 THE DIGESTIVE SYSTEM http://healthfavo.com/digestive-system-for-kids.html This factsheet is about Constipation Constipation is a symptom that can mean different things to different people but the usual
THE DIGESTIVE SYSTEM http://healthfavo.com/digestive-system-for-kids.html This factsheet is about Constipation Constipation is a symptom that can mean different things to different people but the usual
Constipation and bowel obstruction
 Constipation and bowel obstruction Constipation Infrequent or difficult defecation with reduced number of bowel movements, which may or may not be abnormally hard with increased difficulty or discomfort
Constipation and bowel obstruction Constipation Infrequent or difficult defecation with reduced number of bowel movements, which may or may not be abnormally hard with increased difficulty or discomfort
IBS Irritable Bowel syndrome Therapeutics II PHCL 430
 Salman Bin AbdulAziz University College Of Pharmacy IBS Irritable Bowel syndrome Therapeutics II PHCL 430 Email:- ahmedadel.pharmd@gmail.com Ahmed A AlAmer PharmD R.S is 32-year-old woman experiences intermittent
Salman Bin AbdulAziz University College Of Pharmacy IBS Irritable Bowel syndrome Therapeutics II PHCL 430 Email:- ahmedadel.pharmd@gmail.com Ahmed A AlAmer PharmD R.S is 32-year-old woman experiences intermittent
Evidence About the Pharmacological Management of CONSTIPATION PART 2: IMPLICATIONS FOR PALLIATIVE CARE
 Evidence About the Pharmacological Management of CONSTIPATION PART 2: IMPLICATIONS FOR PALLIATIVE CARE Constipation remains a challenging problem for patients and caregivers in home healthcare. Part 1
Evidence About the Pharmacological Management of CONSTIPATION PART 2: IMPLICATIONS FOR PALLIATIVE CARE Constipation remains a challenging problem for patients and caregivers in home healthcare. Part 1
LiquaFiber Clinical Trial
 LiquaFiber Clinical Trial Effects of Dietary Fiber LiquaFiber - on Replacing Bowel Medications and Psyllium Fiber use in Long Term Care Residents This clinical trial was performed at Monroe Community Hospital
LiquaFiber Clinical Trial Effects of Dietary Fiber LiquaFiber - on Replacing Bowel Medications and Psyllium Fiber use in Long Term Care Residents This clinical trial was performed at Monroe Community Hospital
Stool softeners are medicines like (ducolox - pericolace - senokot). You want the stool to remain soft so it is easier to empty the bowel.
 "Bowel Management" How the Body Works The food you eat and drink provides your body with many nutrients. These nutrients give you energy and help you stay healthy. As food moves through your body it breaks
"Bowel Management" How the Body Works The food you eat and drink provides your body with many nutrients. These nutrients give you energy and help you stay healthy. As food moves through your body it breaks
SUMMARY OF PRODUCT CHARACTERISTICS
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Omnilax 10 g powder for oral solution, sachet 2. QUALITATIVE AND QUANTITATIVE COMPOSITION One sachet contains 10 g of macrogol 4000.
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Omnilax 10 g powder for oral solution, sachet 2. QUALITATIVE AND QUANTITATIVE COMPOSITION One sachet contains 10 g of macrogol 4000.
Constipation. What are the signs and symptoms of constipation? Less than three bowel movements per week. Pain or discomfort when opening your bowels
 Infosheet Constipation What is constipation? Constipation is usually diagnosed when your bowels are moving less than three times per week. When you are constipated your bowel movements may be dry, hard
Infosheet Constipation What is constipation? Constipation is usually diagnosed when your bowels are moving less than three times per week. When you are constipated your bowel movements may be dry, hard
