OPIOID-INDUCED CONSTIPATION DR ANDREW DAVIES
|
|
|
- Betty Barker
- 6 years ago
- Views:
Transcription
1 OPIOID-INDUCED CONSTIPATION DR ANDREW DAVIES
2 Introduction
3 Introduction Mean faecal weight 128 g / cap / day Mean range g Absolute range g Main factors affecting mass are caloric intake, type of diet, body weight (Caloric intake accounts for ~ 28% variability)
4 Introduction 75% water 25% solids Organic (84-93% solids) - bacteria 25-54% - carbohydrate 25% - fat 2-15% - protein 2-25% Inorganic - calcium phosphate - iron phosphate - cells - secretions
5 Introduction
6 Introduction Opioids Constipation Functional constipation Other causes
7 Introduction Opioid-induced constipation Functional constipation
8 Introduction Mechanisms of OIC: Decreased small bowel motility Decreased water & electrolyte secretion small bowel Increased tone ileocaecal valve Decreased large bowel motility Increased water & electrolyte absorption large bowel Increased tone anal sphincter Reduced anorectal sensitivity (to distension)
9 Epidemiology
10 Non-malignant disease
11 Non-malignant disease Adverse event Opioid group Placebo group Relative risk (95% CI) Constipation 41% 11% 3.6 ( ) Nausea 32% 12% 2.7 ( ) Somnolence / sedation 29% 10% 3.3 ( ) Vomiting 15% 3% 6.1 (3.3-11) Dizziness 20% 7% 2.8 ( ) Itching 15% 7% 2.2 ( ) Dry mouth 13% 9% 1.5 ( )
12 Non-malignant disease
13 Malignant disease
14 Malignant disease Group Total number patients reporting no constipation Number patients reporting no constipation, and taking no laxatives Number of patients (n = 270) 77 (28.5%) 41 (15.2%) Number patients reporting no constipation, and taking laxatives 36 (13.3%)
15 Opioid-induced constipation
16 Opioid-induced constipation
17 Diagnosis
18 Opioid-induced constipation Rome IV criteria: New or worsening symptoms of constipation when initiating, changing, or increasing opioid therapy that must include 2 or more of the following: a) straining during > ¼ (25%) of defaecations b) lumpy or hard stools (BSFS 1-2) > ¼ (25%) of defaecations c) sensation of incomplete evacuation > ¼ (25%) of defaecations d) sensation of anorectal obstruction/blockage > ¼ (25%) of defaecations
19 Opioid-induced constipation Rome IV criteria: e) manual manoeuvres to facilitate > ¼ (25%) of defaecations f) fewer then 3 spontaneous bowel movements per week Loose stools are rarely present without the use of laxatives
20 Opioid-induced constipation A change, when initiating opioid therapy, from baseline bowel habits and defecation pattern that is characterized by any of the following: a) reduced bowel frequency; b) development or worsening of straining; c) a sense of incomplete evacuation; or d) patients perception of distress related to bowel habits Camilleri et al, 2014
21 Opioid-induced constipation The BFI is a simple assessment tool with a validated threshold of clinically significant constipation. Prescription treatments for opioidinduced constipation should be considered for patients who have a BFI score of 30 points and an inadequate response to first-line interventions. Argoff et al, 2015
22 Opioid-induced constipation
23 Clinical features
24 Opioid-induced constipation Abdominal pain Anorexia Early satiety Nausea Vomiting Abdominal distension Diarrhoea ( overflow )
25 Opioid-induced constipation Flatulence Halitosis Heartburn Intestinal obstruction Intestinal perforation Anal fissure Haemorrhoids
26 Opioid-induced constipation General malaise Confusion Headache Pulmonary embolism Urinary retention
27 Opioid-induced constipation Psychological problems Social problems [Health economic burden]
28 Opioid-induced constipation It s another stress. I thought you would die, blow up inside, a couple of crazy things. Or [I thought I would] get infected or something would go wrong [with] one of your intestines. Dhingra L et al, 2012
29 Opioid-induced constipation When I am faced with taking a pain med, I will always think about the constipation that might result, and so I will try to take the smallest dose possible or do without Dhingra L et al, 2012
30 Opioid-induced constipation
31 Management
32 Opioid-induced constipation Lifestyle measures Conventional laxatives Rectal interventions Lubiprostone Prucalopride Methylnaltrexone Naloxegol Other PAMORAs Complementary therapies Opioid switching
33 Correct position
34 Lubiprostone Oral chloride channel activator (CIC-2) Increases GI secretions... is indicated for the treatment of chronic idiopathic constipation and associated symptoms in adults, when response to diet and other non-pharmacological measures (e.g., educational measures, physical activity) are inappropriate Efficacy in non-malignant OIC (? Efficacy in methadone induced constipation)
35 Lubiprostone Activation of CIC-2 channel leads to active secretion of Cl - into GI lumen, which leads to passive para-cellular movement of Na + (electro- chemical gradient) and water into GI lumen. Increased intraluminal volume may stimulate stretch receptors, which may stimulate smooth muscle contraction (which may also have a therapeutic effect).
36 Lubiprostone 24 mcg twice a day Median time SBM hr Improvement in SBM (versus placebo) Improvements in other symptoms straining, stool consistency GI adverse effects nausea ( %), diarrhoea ( %) No effect on opioids Effective in functional constipation Approved OIC in USA
37 Prucalopride Oral 5-HT4 receptor agonist Increases GI motility...is indicated for symptomatic treatment of chronic constipation in women in whom laxatives fail to provide adequate relief Efficacy in non-malignant OIC
38 Prucalopride Stimulation of 5HT4 receptors increases GI motility via a number of different mechanisms. Stimulation of epithelial 5HT4 receptors increases GI secretions (which may also have a therapeutic effect)
39 Prucalopride 2 mg once a day Improvement in SBM (versus placebo) Improvements in other symptoms stool consistency GI adverse effects abdominal pain (12.1%), nausea (~ 11.0%) No effect on opioids Effective in functional constipation
40 Methylnaltrexone
41 Methylnaltrexone Relistor SPC: Methylnaltrexone is indicated for the treatment of opioid-induced constipation when response to laxative therapy has not been sufficient in adult patients, aged 18 years and older.
42 Methylnaltrexone Relistor SPC: Use of methylnaltrexone bromide in patients with known or suspected mechanical gastrointestinal obstruction, patients at increased risk for recurrent obstruction or in patients with acute surgical abdomen is contraindicated due to the potential for gastrointestinal perforation.
43 Methylnaltrexone Relistor SPC: Opioid - induced constipation in adult patients with chronic pain (except palliative care patients with advanced illness) The recommended dose of methylnaltrexone bromide is 12 mg (0.6 ml of solution) subcutaneously, as needed, given as at least 4 doses weekly, up to once daily (7 doses weekly). In these patients, the treatment with usual laxatives should be stopped when commencing treatment with Relistor.
44 Methylnaltrexone Relistor SPC: Opioid - induced constipation in adult patients with advanced illness (palliative care patients) The recommended dose of methylnaltrexone bromide is 8 mg (0.4 ml of solution) (for patients weighing kg) or 12 mg (0.6 ml of solution) (for patients weighing kg). The usual administration schedule is one single dose every other day. Doses may also be given with longer intervals, as per clinical need.
45 Methylnaltrexone Relistor SPC: Opioid - induced constipation in adult patients with chronic pain (except palliative care patients with advanced illness) Patients may receive two consecutive doses 24 hours apart, only when there has been no response (bowel movement) to the dose on the preceding day. In palliative care patients, Relistor is added to usual laxative treatment.
46 Methylnaltrexone
47 Naloxegol
48 Naloxegol Moventig SPC: Naloxegol is indicated for the treatment of opioid-induced constipation (OIC) in adult patients who have had an inadequate response to laxative(s). To qualify as LIR, in the two weeks prior to first study visit patients had to have reported concurrent OIC symptoms of at least moderate severity while taking at least one laxative class for a minimum of four days during the pre-study period.
49 Naloxegol Moventig SPC: Patients with known or suspected gastrointestinal (GI) obstruction or in patients at increased risk of recurrent obstruction, due to the potential for gastrointestinal perforation. Patients with underlying cancer who are at heightened risk of GI perforation, such as those with: underlying malignancies of gastrointestinal tract or peritoneum; recurrent or advanced ovarian cancer; vascular endothelial growth factor (VEGF) inhibitor treatment.
50 Naloxegol
51 Naloxegol Moventig SPC: When naloxegol therapy is initiated, it is recommended that all currently used maintenance laxative therapy should be halted, until clinical effect of naloxegol is determined.
52 Naloxegol NICE technology appraisal guidance: Naloxegol is recommended, within its marketing authorisation, as an option for treating opioid induced constipation in adults whose constipation has not adequately responded to laxatives.
53 Naloxegol NICE technology appraisal guidance: An inadequate response is defined as opioid-induced constipation symptoms of at least moderate severity in at least 1 of the 4 stool symptom domains (that is, incomplete bowel movement, hard stools, straining or false alarms) while taking at least 1 laxative class for at least 4 days during the prior 2 weeks.
54 Naloxegol Naloxegol (vs. methylnaltrexone): NICE approval Oral formulation (Daily treatment) Slower onset of action? Less acute effects? Less risk GI perforation Lower cost / 30 days vs / 30 days (3 doses/week)
55 Other PAMORAs Naloxone (modified release formulation) Methylnaltrexone (oral formulation) Naldemedine cancer data [Oxycodone / naloxone combination]
56 Complementary therapies
57 Petroleum jelly
58 Conclusion
59 Conclusion
60 Conclusion...I m already sick [with cancer]. I m getting sicker [because of constipation]....it s a contributor...i feel terrible, because it is contributing to my sickness. Dhingra L et al. A qualitative study to explore psychological distress and illness burden associated with opioid-induced constipation in cancer patients with advanced disease. Palliat Med 2012; 27:
CENTENE PHARMACY AND THERAPEUTICS DRUG REVIEW 3Q17 July August
 BRAND NAME Symproic GENERIC NAME Naldemedine MANUFACTURER Shionogi Inc. DATE OF APPROVAL March 23, 2017 PRODUCT LAUNCH DATE Anticipated to launch mid-summer 2017 REVIEW TYPE Review type 1 (RT1): New Drug
BRAND NAME Symproic GENERIC NAME Naldemedine MANUFACTURER Shionogi Inc. DATE OF APPROVAL March 23, 2017 PRODUCT LAUNCH DATE Anticipated to launch mid-summer 2017 REVIEW TYPE Review type 1 (RT1): New Drug
Opioid-Induced Constipation
 Objectives Opioid-Induced Constipation Brianna Jansma, PharmD Alex Smith, PharmD Megan Robinson, PharmD Summarize epidemiology of opioid-induced constipation (OIC) Understand opiates effects on the gastrointestinal
Objectives Opioid-Induced Constipation Brianna Jansma, PharmD Alex Smith, PharmD Megan Robinson, PharmD Summarize epidemiology of opioid-induced constipation (OIC) Understand opiates effects on the gastrointestinal
daily; available as 10- mg g PO
 Overview of the PRN: The Pain and Palliative Care PRN of ACCP is an organization of pharmacy practitioners, clinical scientists, pharmacy educators, and others. Its mission is to advance pain and palliative
Overview of the PRN: The Pain and Palliative Care PRN of ACCP is an organization of pharmacy practitioners, clinical scientists, pharmacy educators, and others. Its mission is to advance pain and palliative
SUPPLEMENTARY INFORMATION Associated with
 Table1: Rome III and Rome IV diagnostic criteria for IBS, functional constipation and functional dyspepsia. Rome III diagnostic criteria 1,2 Rome IV diagnostic criteria 3,4 Diagnostic criteria for IBS
Table1: Rome III and Rome IV diagnostic criteria for IBS, functional constipation and functional dyspepsia. Rome III diagnostic criteria 1,2 Rome IV diagnostic criteria 3,4 Diagnostic criteria for IBS
Movantik (naloxegol), Relistor (methylnaltrexone bromide), Symproic (naldemedine)
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.50.06 Subject: Opioid Antagonist Drug Class Page: 1 of 7 Last Review Date: November 30, 2018 Opioid Antagonist
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.50.06 Subject: Opioid Antagonist Drug Class Page: 1 of 7 Last Review Date: November 30, 2018 Opioid Antagonist
Movantik (naloxegol), Relistor (methylnaltrexone bromide), Symproic (naldemedine)
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.50.06 Subject: Opioid Antagonist Drug Class Page: 1 of 5 Last Review Date: June 22, 2017 Opioid Antagonist
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.50.06 Subject: Opioid Antagonist Drug Class Page: 1 of 5 Last Review Date: June 22, 2017 Opioid Antagonist
Irritable Bowel Syndrome. Mustafa Giaffer March 2017
 Irritable Bowel Syndrome Mustafa Giaffer March 2017 Introduction First described in 1771. 50% of patients present
Irritable Bowel Syndrome Mustafa Giaffer March 2017 Introduction First described in 1771. 50% of patients present
Movantik (naloxegol), Relistor (methylnaltrexone bromide)
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.50.06 Subject: Opioid Antagonist Drug Class Page: 1 of 5 Last Review Date: December 2, 2016 Opioid Antagonist
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.50.06 Subject: Opioid Antagonist Drug Class Page: 1 of 5 Last Review Date: December 2, 2016 Opioid Antagonist
Amitiza. Amitiza (lubiprostone) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.50.22 Subject: Amitiza Page: 1 of 5 Last Review Date: March 16, 2018 Amitiza Description Amitiza (lubiprostone)
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.50.22 Subject: Amitiza Page: 1 of 5 Last Review Date: March 16, 2018 Amitiza Description Amitiza (lubiprostone)
Primary Care Constipation Guidelines. Version 1 November 2016
 Primary Care Constipation Guidelines Version 1 November 2016 VERSION CONTROL Version Date Amendments made Version 1 November 2016 New guideline Contents 1. Management of constipation in adults: acute and
Primary Care Constipation Guidelines Version 1 November 2016 VERSION CONTROL Version Date Amendments made Version 1 November 2016 New guideline Contents 1. Management of constipation in adults: acute and
Technology appraisal guidance Published: 22 July 2015 nice.org.uk/guidance/ta345
 Naloxegol for treating opioid-induced constipation Technology appraisal guidance Published: 22 July 2015 nice.org.uk/guidance/ta345 NICE 2017. All rights reserved. Subject to Notice of rights (https://www.nice.org.uk/terms-and-conditions#notice-ofrights).
Naloxegol for treating opioid-induced constipation Technology appraisal guidance Published: 22 July 2015 nice.org.uk/guidance/ta345 NICE 2017. All rights reserved. Subject to Notice of rights (https://www.nice.org.uk/terms-and-conditions#notice-ofrights).
Efficacy and Safety of Lubiprostone. Laura Wozniak February 23, 2010 K30 Monthly Journal Club
 Efficacy and Safety of Lubiprostone Laura Wozniak February 23, 2010 K30 Monthly Journal Club Objectives Brief overview of constipation Review of article Discussion Constipation in Children 3-5% of all
Efficacy and Safety of Lubiprostone Laura Wozniak February 23, 2010 K30 Monthly Journal Club Objectives Brief overview of constipation Review of article Discussion Constipation in Children 3-5% of all
SYMPROIC (naldemedine tosylate) oral capsule
 SYMPROIC (naldemedine tosylate) oral capsule Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This
SYMPROIC (naldemedine tosylate) oral capsule Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This
The Road to Opioid-Induced Constipation: Pathophysiology and Impact Kenneth C. Jackson, II, PharmD, CPE
 Disclaimer This slide deck in its original and unaltered format is for educational purposes and is current as of March 2015. The content and views presented in this educational activity are those of the
Disclaimer This slide deck in its original and unaltered format is for educational purposes and is current as of March 2015. The content and views presented in this educational activity are those of the
Pharmacy Benefit Determination Policy
 Policy Subject: Opioid Induced Constipation Policy Number: SHS PBD11 Category: GI Agents Policy Type: Medical Pharmacy Department: Pharmacy Product (check all that apply): Group HMO/POS Individual HMO/POS
Policy Subject: Opioid Induced Constipation Policy Number: SHS PBD11 Category: GI Agents Policy Type: Medical Pharmacy Department: Pharmacy Product (check all that apply): Group HMO/POS Individual HMO/POS
Prof. Dr. Mufti Munsurar Rahman Dept. of Medicine Enam Medical College Hospital
 Prof. Dr. Mufti Munsurar Rahman Dept. of Medicine Enam Medical College Hospital Functional constipation : prevalence and life style factors in a district of Bangladesh Perveen I, Rahman MM, Saha M, Parvin
Prof. Dr. Mufti Munsurar Rahman Dept. of Medicine Enam Medical College Hospital Functional constipation : prevalence and life style factors in a district of Bangladesh Perveen I, Rahman MM, Saha M, Parvin
Summary of the risk management plan (RMP) for Moventig (naloxegol)
 EMA/611606/2014 Summary of the risk management plan (RMP) for Moventig (naloxegol) This is a summary of the risk management plan (RMP) for Moventig, which details the measures to be taken in order to ensure
EMA/611606/2014 Summary of the risk management plan (RMP) for Moventig (naloxegol) This is a summary of the risk management plan (RMP) for Moventig, which details the measures to be taken in order to ensure
Constipation An Overview. Definition Physiology of GI tract Etiology Assessment Treatment
 CONSTIPATION Constipation An Overview Definition Physiology of GI tract Etiology Assessment Treatment Definition Constipation = the infrequent passage of hard feces Definition of Infrequent The meaning
CONSTIPATION Constipation An Overview Definition Physiology of GI tract Etiology Assessment Treatment Definition Constipation = the infrequent passage of hard feces Definition of Infrequent The meaning
Elderly Man With Chronic Constipation
 Elderly Man With Chronic Constipation Linda Nguyen, MD Director, Neurogastroenterology and Motility Clinical Assistant Professor Stanford University Overview Normal bowel function Defining Constipation:
Elderly Man With Chronic Constipation Linda Nguyen, MD Director, Neurogastroenterology and Motility Clinical Assistant Professor Stanford University Overview Normal bowel function Defining Constipation:
Oral Methylnaltrexone for the. Constipation in Patients with Chronic Non-cancer Pain
 Oral Methylnaltrexone for the Treatment of Opioid-induced Constipation in Patients with Chronic Non-cancer Pain Richard L. Rauck, 1 John F. Peppin, 2 Robert J.Israel, 3 Jennifer Carpenito, 3 Jeffrey Cohn,
Oral Methylnaltrexone for the Treatment of Opioid-induced Constipation in Patients with Chronic Non-cancer Pain Richard L. Rauck, 1 John F. Peppin, 2 Robert J.Israel, 3 Jennifer Carpenito, 3 Jeffrey Cohn,
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE Premeeting briefing Prucalopride for the treatment of chronic constipation in women
 NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE Premeeting briefing Prucalopride for the treatment of chronic constipation in women This briefing presents the key issues arising from the manufacturer
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE Premeeting briefing Prucalopride for the treatment of chronic constipation in women This briefing presents the key issues arising from the manufacturer
The Digestive System or tract extends from the mouth to the anus.
 The Digestive System or tract extends from the mouth to the anus. FUNCTION The Digestive System breaks down and absorbs food materials e.g. amino acids, glucose DEFINITIONS: Ingestion: Ingestion is the
The Digestive System or tract extends from the mouth to the anus. FUNCTION The Digestive System breaks down and absorbs food materials e.g. amino acids, glucose DEFINITIONS: Ingestion: Ingestion is the
Angie Jefferson Registered Dietitian & Consultant Nutritionist
 Angie Jefferson Registered Dietitian & Consultant Nutritionist Constipation & Haemorrhoids are two of the most common problems experienced by pregnant women Incidence increases dramatically 40% women reportedto
Angie Jefferson Registered Dietitian & Consultant Nutritionist Constipation & Haemorrhoids are two of the most common problems experienced by pregnant women Incidence increases dramatically 40% women reportedto
Understanding & Alleviating Constipation. Living (Well!) with Gastroparesis Program Warm-Up Class
 Understanding & Alleviating Constipation Living (Well!) with Gastroparesis Program Warm-Up Class Please Remember The information presented is for educational purposes only and is in no way intended as
Understanding & Alleviating Constipation Living (Well!) with Gastroparesis Program Warm-Up Class Please Remember The information presented is for educational purposes only and is in no way intended as
Is one of the most common chronic disorders. causing patients to seek medical treatment.
 ILOs After this lecture you should be able to : Define IBS Identify causes and risk factors of IBS Determine the appropriate therapeutic options for IBS Is one of the most common chronic disorders causing
ILOs After this lecture you should be able to : Define IBS Identify causes and risk factors of IBS Determine the appropriate therapeutic options for IBS Is one of the most common chronic disorders causing
Technology appraisal guidance Published: 15 December 2010 nice.org.uk/guidance/ta211
 Prucalopride for the treatment of chronic constipation in women Technology appraisal guidance Published: 15 December 2010 nice.org.uk/guidance/ta211 NICE 2018. All rights reserved. Subject to Notice of
Prucalopride for the treatment of chronic constipation in women Technology appraisal guidance Published: 15 December 2010 nice.org.uk/guidance/ta211 NICE 2018. All rights reserved. Subject to Notice of
Constipation and bowel obstruction
 Constipation and bowel obstruction Constipation Infrequent or difficult defecation with reduced number of bowel movements, which may or may not be abnormally hard with increased difficulty or discomfort
Constipation and bowel obstruction Constipation Infrequent or difficult defecation with reduced number of bowel movements, which may or may not be abnormally hard with increased difficulty or discomfort
TRANSPARENCY COMMITTEE OPINION. 10 December 2008
 The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 10 December 2008 RELISTOR 12 mg/0.6 ml solution for injection 1 vial (CIP: 387 365-1) 2 vials + 2 sterile syringes
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 10 December 2008 RELISTOR 12 mg/0.6 ml solution for injection 1 vial (CIP: 387 365-1) 2 vials + 2 sterile syringes
15. Prevention of UTI and lifestyle modifications
 15. Prevention of UTI and lifestyle modifications Key questions: Does improving poor voiding habits help prevent UTI recurrence? Does improving constipation help prevent UTI recurrence? Does increasing
15. Prevention of UTI and lifestyle modifications Key questions: Does improving poor voiding habits help prevent UTI recurrence? Does improving constipation help prevent UTI recurrence? Does increasing
FOOT OFF THE BRAKES. Kerri Novak MD MSc FRCPC. Chronic Constipation: Taking the Foot off the Brakes Dr. Kerri Novak
 CHRONIC CONSTIPATION: TAKING THE FOOT OFF THE BRAKES Kerri Novak MD MSc FRCPC www.seacourses.com 1 OUTLINE Epidemiology i Quality of life Approach Therapies www.seacourses.com 2 DEFINING CHRONIC CONSTIPATION
CHRONIC CONSTIPATION: TAKING THE FOOT OFF THE BRAKES Kerri Novak MD MSc FRCPC www.seacourses.com 1 OUTLINE Epidemiology i Quality of life Approach Therapies www.seacourses.com 2 DEFINING CHRONIC CONSTIPATION
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE SCOPE
 NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE 1 Guideline title SCOPE Constipation: management of idiopathic constipation in children in primary and secondary care 1.1 Short title Constipation
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE 1 Guideline title SCOPE Constipation: management of idiopathic constipation in children in primary and secondary care 1.1 Short title Constipation
Gastrointestinal obstruction Dr Iain Lawrie
 Gastrointestinal obstruction Dr Iain Lawrie Consultant and Honorary Clinical Senior Lecturer in Palliative Medicine The Pennine Acute Hospitals NHS Trust / The University of Manchester iain.lawrie@pat.nhs.uk
Gastrointestinal obstruction Dr Iain Lawrie Consultant and Honorary Clinical Senior Lecturer in Palliative Medicine The Pennine Acute Hospitals NHS Trust / The University of Manchester iain.lawrie@pat.nhs.uk
Emerging Treatments for IBS-C and Clinical Trial Endpoints
 Emerging Treatments for IBS-C and Clinical Trial Endpoints Lin Chang, M.D. Oppenheimer Family Center for Neurobiology of Stress David Geffen School of Medicine at UCLA Learning Objectives Describe current
Emerging Treatments for IBS-C and Clinical Trial Endpoints Lin Chang, M.D. Oppenheimer Family Center for Neurobiology of Stress David Geffen School of Medicine at UCLA Learning Objectives Describe current
Constipation Information Leaflet THE DIGESTIVE SYSTEM. gutscharity.org.uk
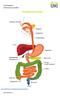 THE DIGESTIVE SYSTEM http://healthfavo.com/digestive-system-for-kids.html This factsheet is about Constipation Constipation is a symptom that can mean different things to different people but the usual
THE DIGESTIVE SYSTEM http://healthfavo.com/digestive-system-for-kids.html This factsheet is about Constipation Constipation is a symptom that can mean different things to different people but the usual
What Is Constipation?
 CONSTIPATION What Is Constipation? Constipation is when you have infrequent or hard-to-pass bowel movements (meaning they are painful or you have to strain), have hard stools or feel like your bowel movements
CONSTIPATION What Is Constipation? Constipation is when you have infrequent or hard-to-pass bowel movements (meaning they are painful or you have to strain), have hard stools or feel like your bowel movements
Technology appraisal guidance Published: 23 July 2014 nice.org.uk/guidance/ta318
 Lubiprostone for treating chronic idiopathic constipation Technology appraisal guidance Published: 23 July 2014 nice.org.uk/guidance/ta318 NICE 2017. All rights reserved. Subject to Notice of rights (https://www.nice.org.uk/terms-and-conditions#notice-ofrights).
Lubiprostone for treating chronic idiopathic constipation Technology appraisal guidance Published: 23 July 2014 nice.org.uk/guidance/ta318 NICE 2017. All rights reserved. Subject to Notice of rights (https://www.nice.org.uk/terms-and-conditions#notice-ofrights).
Drug Evaluation. Use of lubiprostone in constipating disorders and its potential for opioid-induced bowel dysfunction
 Use of lubiprostone in constipating disorders and its potential for opioid-induced bowel dysfunction Lubiprostone is a novel medication, approved by the US FDA for the treatment of chronic idiopathic constipation
Use of lubiprostone in constipating disorders and its potential for opioid-induced bowel dysfunction Lubiprostone is a novel medication, approved by the US FDA for the treatment of chronic idiopathic constipation
Constipation. What is constipation? What is the criteria for having constipation? What are the different types of constipation?
 What is constipation? is defined as having a bowel movement less than 3 times per week. It is usually associated with hard stools or difficulty passing stools. You may have pain while passing stools or
What is constipation? is defined as having a bowel movement less than 3 times per week. It is usually associated with hard stools or difficulty passing stools. You may have pain while passing stools or
DOCTOR DISCUSSION GUIDE
 What your doctor needs to know To make your appointment more productive, share this completed guide with your doctor. That way, he or she can recommend an appropriate treatment plan for you. Check the
What your doctor needs to know To make your appointment more productive, share this completed guide with your doctor. That way, he or she can recommend an appropriate treatment plan for you. Check the
2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each sachet of Macrovic Junior powder for oral solution contains the following active ingredients:
 SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Macrovic Junior powder for oral solution 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each sachet of Macrovic Junior powder for oral solution
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Macrovic Junior powder for oral solution 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each sachet of Macrovic Junior powder for oral solution
Clinical problems related to GI involvement in SSc
 Clinical problems related to GI involvement in SSc Incontinence Abdominal pain/distension Gastro-oesophageal Diarrhoea Weight loss/al Issues Constipation Management of incontinence Establish diagnosis
Clinical problems related to GI involvement in SSc Incontinence Abdominal pain/distension Gastro-oesophageal Diarrhoea Weight loss/al Issues Constipation Management of incontinence Establish diagnosis
MOVICOL HALF PI December MOVICOL-Half. Powder for Solution (macrogol 3350) Potassium 5.4 mmol/l. Bicarbonate 17 mmol/l
 MOVICOL -Half Powder for Solution (macrogol 3350) Product Name: Product Description: MOVICOL-Half Each sachet of MOVICOL-Half contains: Macrogol 3350 6.563 g Sodium chloride 175.4 mg Sodium bicarbonate
MOVICOL -Half Powder for Solution (macrogol 3350) Product Name: Product Description: MOVICOL-Half Each sachet of MOVICOL-Half contains: Macrogol 3350 6.563 g Sodium chloride 175.4 mg Sodium bicarbonate
RELISTOR. Methylnaltrexone bromide, Subcutaneous solution for injection. Consumer Medicine Information
 RELISTOR Methylnaltrexone bromide, Subcutaneous solution for injection Consumer Medicine Information What is in this leaflet? This leaflet answers some common questions about RELISTOR. It does not contain
RELISTOR Methylnaltrexone bromide, Subcutaneous solution for injection Consumer Medicine Information What is in this leaflet? This leaflet answers some common questions about RELISTOR. It does not contain
SUMMARY OF PRODUCT CHARACTERISTICS
 SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Lacrofarm Junior, powder for oral solution, sachet 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each sachet of contains the following active
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Lacrofarm Junior, powder for oral solution, sachet 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each sachet of contains the following active
IRONWOOD AND FOREST ANNOUNCE POSITIVE LINACLOTIDE RESULTS FROM PHASE 3 TRIAL IN PATIENTS WITH IRRITABLE BOWEL SYNDROME WITH CONSTIPATION
 FOR IMMEDIATE RELEASE Ironwood Contact: Forest Contact: Susan Brady Frank J. Murdolo Corporate Communications Vice President, Investor Relations 617.621.8304 212.224.6714 sbrady@ironwoodpharma.com frank.murdolo@frx.com
FOR IMMEDIATE RELEASE Ironwood Contact: Forest Contact: Susan Brady Frank J. Murdolo Corporate Communications Vice President, Investor Relations 617.621.8304 212.224.6714 sbrady@ironwoodpharma.com frank.murdolo@frx.com
MOVICOL Junior Powder for Solution (macrogol 3350)
 MOVICOL Junior Powder for Solution (macrogol 3350) Product Name: MOVICOL Junior Product Description: Each sachet of MOVICOL Junior contains: Macrogol 3350 Sodium chloride Sodium bicarbonate Potassium chloride
MOVICOL Junior Powder for Solution (macrogol 3350) Product Name: MOVICOL Junior Product Description: Each sachet of MOVICOL Junior contains: Macrogol 3350 Sodium chloride Sodium bicarbonate Potassium chloride
Childhood constipation, a real problem..? Marc Benninga, Emma Children s Hospital, AMC, Amsterdam, the Netherlands
 Childhood constipation, a real problem..? Marc Benninga, Emma Children s Hospital, AMC, Amsterdam, the Netherlands Constipation 0-10% >10-20% >20-30% >30-40% Mugie SM, et al. Best Pract & Res Clin Gastroenterol
Childhood constipation, a real problem..? Marc Benninga, Emma Children s Hospital, AMC, Amsterdam, the Netherlands Constipation 0-10% >10-20% >20-30% >30-40% Mugie SM, et al. Best Pract & Res Clin Gastroenterol
Chronic constipation in the elderly
 Chronic constipation in the elderly 1 Dec,2011 R 2 Natta Asanaleykha Epidemiology Definition Scope The impact of chronic constipation in the elderly Pathophysiology Evaluation the elderly patient with
Chronic constipation in the elderly 1 Dec,2011 R 2 Natta Asanaleykha Epidemiology Definition Scope The impact of chronic constipation in the elderly Pathophysiology Evaluation the elderly patient with
OCTOBER 7-10 PHILADELPHIA, PENNSYLVANIA
 OMED 17 OCTOBER 7-10 PHILADELPHIA, PENNSYLVANIA 29.5 Category 1-A CME credits anticipated ACOFP / AOA s 122 nd Annual Osteopathic Medical Conference & Exposition Joint Session with ACOFP and Cleveland
OMED 17 OCTOBER 7-10 PHILADELPHIA, PENNSYLVANIA 29.5 Category 1-A CME credits anticipated ACOFP / AOA s 122 nd Annual Osteopathic Medical Conference & Exposition Joint Session with ACOFP and Cleveland
AMITIZA (lubiprostone) capsules, for oral use Initial U.S. Approval: 2006
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use AMITIZA safely and effectively. See full prescribing information for AMITIZA. AMITIZA (lubiprostone)
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use AMITIZA safely and effectively. See full prescribing information for AMITIZA. AMITIZA (lubiprostone)
IBS - Definition. Chronic functional disorder of GI generally characterized by:
 IBS - Definition Chronic functional disorder of GI generally characterized by: 3500 3000 No. of Publications 2500 2000 1500 1000 Irritable Bowel syndrome Irritable Bowel Syndrome 500 0 1968-1977 1978-1987
IBS - Definition Chronic functional disorder of GI generally characterized by: 3500 3000 No. of Publications 2500 2000 1500 1000 Irritable Bowel syndrome Irritable Bowel Syndrome 500 0 1968-1977 1978-1987
MOVICOL Junior Chocolate Flavour Powder for Solution (macrogol 3350)
 MOVICOL Junior Chocolate Flavour Powder for Solution (macrogol 3350) Product Name: MOVICOL Junior Chocolate Flavour Product Description: Each sachet of MOVICOL Junior Chocolate contains: Macrogol 3350
MOVICOL Junior Chocolate Flavour Powder for Solution (macrogol 3350) Product Name: MOVICOL Junior Chocolate Flavour Product Description: Each sachet of MOVICOL Junior Chocolate contains: Macrogol 3350
IBS Irritable Bowel syndrome Therapeutics II PHCL 430
 Salman Bin AbdulAziz University College Of Pharmacy IBS Irritable Bowel syndrome Therapeutics II PHCL 430 Email:- ahmedadel.pharmd@gmail.com Ahmed A AlAmer PharmD R.S is 32-year-old woman experiences intermittent
Salman Bin AbdulAziz University College Of Pharmacy IBS Irritable Bowel syndrome Therapeutics II PHCL 430 Email:- ahmedadel.pharmd@gmail.com Ahmed A AlAmer PharmD R.S is 32-year-old woman experiences intermittent
SUMMARY OF PRODUCT CHARACTERISTICS. 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each sachet contains the following active ingredients:
 SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Lacrofarm, powder for oral solution, sachet 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each sachet contains the following active ingredients:
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Lacrofarm, powder for oral solution, sachet 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each sachet contains the following active ingredients:
MOVICOL Lemon-Lime Flavour Powder for Solution (macrogol 3350)
 MOVICOL Lemon-Lime Flavour Powder for Solution (macrogol 3350) Product Name: MOVICOL Lemon-Lime Flavour Product Description: Each sachet of MOVICOL Lemon-Lime contains: Macrogol 3350 Sodium chloride Sodium
MOVICOL Lemon-Lime Flavour Powder for Solution (macrogol 3350) Product Name: MOVICOL Lemon-Lime Flavour Product Description: Each sachet of MOVICOL Lemon-Lime contains: Macrogol 3350 Sodium chloride Sodium
MOVICOL Liquid Orange Flavour Concentrate for Oral Solution (macrogol 3350)
 MOVICOL Liquid Orange Flavour Concentrate for Oral Solution (macrogol 3350) NAME OF THE MEDICINE: MOVICOL Liquid Orange Flavour, Concentrate for Oral Solution. DESCRIPTION: A clear colourless solution.
MOVICOL Liquid Orange Flavour Concentrate for Oral Solution (macrogol 3350) NAME OF THE MEDICINE: MOVICOL Liquid Orange Flavour, Concentrate for Oral Solution. DESCRIPTION: A clear colourless solution.
WORKING PARTY ON HERBAL MEDICINAL PRODUCTS FINAL PROPOSAL FOR A CORE DATA FOR ISPAGHULA HUSK
 The European Agency for the Evaluation of Medicinal Products Evaluation of Medicines for Human Use 27 March 2003 EMEA/HMPWP/15/00 WORKING PARTY ON HERBAL MEDICINAL PRODUCTS FINAL PROPOSAL FOR A CORE DATA
The European Agency for the Evaluation of Medicinal Products Evaluation of Medicines for Human Use 27 March 2003 EMEA/HMPWP/15/00 WORKING PARTY ON HERBAL MEDICINAL PRODUCTS FINAL PROPOSAL FOR A CORE DATA
Horizon Scanning Technology Summary. Methylnaltrexone for opioid induced constipation in advanced illness and palliative care
 Horizon Scanning Technology Summary National Horizon Scanning Centre Methylnaltrexone for opioid induced constipation in advanced illness and palliative care April 2007 This technology summary is based
Horizon Scanning Technology Summary National Horizon Scanning Centre Methylnaltrexone for opioid induced constipation in advanced illness and palliative care April 2007 This technology summary is based
Bowel cancer risk in the under 50s. Greg Rubin Professor of General Practice and Primary Care
 Bowel cancer risk in the under 50s Greg Rubin Professor of General Practice and Primary Care Prevalence of GI problems in the consulting population Thompson et al, Gut 2000 Number of patients % of patients
Bowel cancer risk in the under 50s Greg Rubin Professor of General Practice and Primary Care Prevalence of GI problems in the consulting population Thompson et al, Gut 2000 Number of patients % of patients
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE SCOPE
 NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE 1 Guideline title SCOPE Constipation: the diagnosis and management of idiopathic childhood constipation in primary and secondary care 1.1 Short title
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE 1 Guideline title SCOPE Constipation: the diagnosis and management of idiopathic childhood constipation in primary and secondary care 1.1 Short title
2 QUALITATIVE AND QUANTITATIVE COMPOSITION
 SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Compound Macrogol 13.72 g powder for oral solution 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each sachet of Compound Macrogol 13.72 g
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Compound Macrogol 13.72 g powder for oral solution 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each sachet of Compound Macrogol 13.72 g
Gastrointestinal Involvement in Adult Mitochondrial Disease
 Newcastle Mitochondrial Disease Guidelines At a glance guidelines: Gastrointestinal Involvement in Adult Mitochondrial Disease For full guideline visit: http://www.newcastle-mitochondria.com/service/patient-care-guidelines/
Newcastle Mitochondrial Disease Guidelines At a glance guidelines: Gastrointestinal Involvement in Adult Mitochondrial Disease For full guideline visit: http://www.newcastle-mitochondria.com/service/patient-care-guidelines/
Improving the Diagnosis and Management of Opioid-induced Constipation to Optimize Outcomes of Patients with Chronic Pain
 Improving the Diagnosis and Management of Opioid-induced Constipation to Optimize Outcomes of Patients with Chronic Pain Sponsored by Integrity Continuing Education, Inc. Supported by an educational grant
Improving the Diagnosis and Management of Opioid-induced Constipation to Optimize Outcomes of Patients with Chronic Pain Sponsored by Integrity Continuing Education, Inc. Supported by an educational grant
Multimodal Approach for Managing Postoperative Ileus: Role of Health- System Pharmacists (ACPE program H01P)
 1. In the normal gastrointestinal tract, what percent of nutrient absorption occurs in the jejunum? a. 20%. b. 40%. c. 70%. d. 90%. 2. According to Dr. Erstad, the four components of gastrointestinal control
1. In the normal gastrointestinal tract, what percent of nutrient absorption occurs in the jejunum? a. 20%. b. 40%. c. 70%. d. 90%. 2. According to Dr. Erstad, the four components of gastrointestinal control
Irritable Bowel Syndrome Now. George M. Logan, MD Friday, May 5, :35 4:05 PM
 Irritable Bowel Syndrome Now George M. Logan, MD Friday, May 5, 2017 3:35 4:05 PM Dr. Logan indicated no potential conflict of interest to this presentation. He does not intend to discuss any unapproved/investigative
Irritable Bowel Syndrome Now George M. Logan, MD Friday, May 5, 2017 3:35 4:05 PM Dr. Logan indicated no potential conflict of interest to this presentation. He does not intend to discuss any unapproved/investigative
Opioid constipation treatment dulcolax
 P ford residence southampton, ny Opioid constipation treatment dulcolax Posts about how to relieve constipation instantly written by gbrese1958. Official Web site for MOVANTIK (naloxegol) tablets, for
P ford residence southampton, ny Opioid constipation treatment dulcolax Posts about how to relieve constipation instantly written by gbrese1958. Official Web site for MOVANTIK (naloxegol) tablets, for
Authors and Disclosures
 Role of Carbon Dioxide-Releasing Suppositories in the Treatment of Chronic Functional Constipation A Double-Blind, Randomised, Placebo-Controlled Trial M. Lazzaroni; V. Casini; G. Bianchi Porro Authors
Role of Carbon Dioxide-Releasing Suppositories in the Treatment of Chronic Functional Constipation A Double-Blind, Randomised, Placebo-Controlled Trial M. Lazzaroni; V. Casini; G. Bianchi Porro Authors
Fact Sheet. Zohydro ER (hydrocodone bitartrate) Extended-Release Capsules, CII
 Zohydro ER (hydrocodone bitartrate) Extended-Release Capsules, CII Fact Sheet Zohydro ER (hydrocodone bitartrate) Extended-Release Capsule, CII, is a long-acting (extendedrelease) type of pain medication
Zohydro ER (hydrocodone bitartrate) Extended-Release Capsules, CII Fact Sheet Zohydro ER (hydrocodone bitartrate) Extended-Release Capsule, CII, is a long-acting (extendedrelease) type of pain medication
FULL PRESCRIBING INFORMATION: CONTENTS*
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use AMITIZA safely and effectively. See full prescribing information for AMITIZA. AMITIZA (lubiprostone)
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use AMITIZA safely and effectively. See full prescribing information for AMITIZA. AMITIZA (lubiprostone)
Opioids and the Gastroenterologist: A Painful Issue
 Opioids and the Gastroenterologist: A Painful Issue Wendell K. Clarkston, MD Professor of Medicine GI Program Director UMKC School of Medicine Disclosures: No financial relationships to disclose 1 Outline
Opioids and the Gastroenterologist: A Painful Issue Wendell K. Clarkston, MD Professor of Medicine GI Program Director UMKC School of Medicine Disclosures: No financial relationships to disclose 1 Outline
Human Anatomy rectum
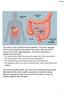 rectum The colon is also called the large intestine. The ileum (last part of the small intestine) connects to the cecum (first part of the colon) in the lower right abdomen. The rest of the colon is divided
rectum The colon is also called the large intestine. The ileum (last part of the small intestine) connects to the cecum (first part of the colon) in the lower right abdomen. The rest of the colon is divided
Package leaflet: Information for the patient. Moventig 12.5 mg film-coated tablets Moventig 25 mg film-coated tablets. naloxegol
 Package leaflet: Information for the patient Moventig 12.5 mg film-coated tablets Moventig 25 mg film-coated tablets naloxegol This medicine is subject to additional monitoring. This will allow quick identification
Package leaflet: Information for the patient Moventig 12.5 mg film-coated tablets Moventig 25 mg film-coated tablets naloxegol This medicine is subject to additional monitoring. This will allow quick identification
Drugs in Development for Opioid-Induced Constipation
 7 Drugs in Development for Opioid-Induced Constipation Kelly S. Sprawls, Egilius L.H. Spierings and Dustin Tran MedVadis Research Corporation USA 1. Introduction Opioid-induced bowel dysfunction (OIBD)
7 Drugs in Development for Opioid-Induced Constipation Kelly S. Sprawls, Egilius L.H. Spierings and Dustin Tran MedVadis Research Corporation USA 1. Introduction Opioid-induced bowel dysfunction (OIBD)
National Institute for Health and Clinical Excellence. Health Technology Appraisal. Prucalopride for the treatment of chronic constipation in women
 Health Technology Appraisal Summary form Prucalopride for the treatment of chronic constipation in women Comment 1: the draft remit Appropriateness Movetis Movetis entirely welcomes the opportunity to
Health Technology Appraisal Summary form Prucalopride for the treatment of chronic constipation in women Comment 1: the draft remit Appropriateness Movetis Movetis entirely welcomes the opportunity to
EXENATIDE (BYETTA ) PROTOCOL, 5mcg and 10mcg SC injection pre-filled pens
 EXENATIDE (BYETTA ) PROTOCOL, 5mcg and 10mcg SC injection pre-filled pens This document should be read in conjunction with the current Summary of Product Characteristics http://www.medicines.org.uk 1.
EXENATIDE (BYETTA ) PROTOCOL, 5mcg and 10mcg SC injection pre-filled pens This document should be read in conjunction with the current Summary of Product Characteristics http://www.medicines.org.uk 1.
SUMMARY OF THE PRODUCT CHARACTERISTICS. The content of electrolyte ions per sachet when made up to 125 ml of solution.
 SUMMARY OF THE PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Molaxole powder for oral solution 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each sachet contains following active substances Macrogol
SUMMARY OF THE PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Molaxole powder for oral solution 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each sachet contains following active substances Macrogol
Constipation. Information for adults. GI Motility Clinic (UMCCC University Medical Clinics of Campbelltown and Camden) Page 1
 Constipation Information for adults GI Motility Clinic (UMCCC University Medical Clinics of Campbelltown and Camden) Page 1 Contents Role of the large intestine..3 Mass movements in the large intestine..4
Constipation Information for adults GI Motility Clinic (UMCCC University Medical Clinics of Campbelltown and Camden) Page 1 Contents Role of the large intestine..3 Mass movements in the large intestine..4
RELISTOR (methylnaltrexone bromide) INJECTION FOR SUBCUTANEOUS USE
 RELISTOR (methylnaltrexone bromide) INJECTION FOR SUBCUTANEOUS USE Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for
RELISTOR (methylnaltrexone bromide) INJECTION FOR SUBCUTANEOUS USE Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for
Scandinavian Journal of Pain
 Scandinavian Journal of Pain 11 (2016) 111 122 Contents lists available at ScienceDirect Scandinavian Journal of Pain journal homepage: www.scandinavianjournalpain.com Topical review Definition, diagnosis
Scandinavian Journal of Pain 11 (2016) 111 122 Contents lists available at ScienceDirect Scandinavian Journal of Pain journal homepage: www.scandinavianjournalpain.com Topical review Definition, diagnosis
Treatment Conversation Guide
 Created For: Doctor Appointment Date: Treatment Conversation Guide What your healthcare provider needs to know How does your condition affect your life? To make your appointment more productive, fill out
Created For: Doctor Appointment Date: Treatment Conversation Guide What your healthcare provider needs to know How does your condition affect your life? To make your appointment more productive, fill out
Antidiarrheals Antidiarrheal
 Antidiarrheals Major factors in diarrhea Increased motility of the GI tract. Decreased absorption of fluid. Antidiarrheal drugs include: Antimotility agents. Adsorbents. Drugs that modify fluid and electrolyte
Antidiarrheals Major factors in diarrhea Increased motility of the GI tract. Decreased absorption of fluid. Antidiarrheal drugs include: Antimotility agents. Adsorbents. Drugs that modify fluid and electrolyte
Opioid Induced Constipation Lisa Clince Acute Pain CNS Palmerston North Hospital
 Opioid Induced Constipation Lisa Clince Acute Pain CNS Palmerston North Hospital 21-04-2016 MidCentral District Health Board 1 Overview Patient Experience Opioid Collaborative Constipation defined Opioid
Opioid Induced Constipation Lisa Clince Acute Pain CNS Palmerston North Hospital 21-04-2016 MidCentral District Health Board 1 Overview Patient Experience Opioid Collaborative Constipation defined Opioid
A guide to Anoplasty (anal surgery)
 Saint Mary s Hospital Newborn Intensive Care Unit Information for Parents A guide to Anoplasty (anal surgery) Introduction This information leaflet is designed to help parents and families to care for
Saint Mary s Hospital Newborn Intensive Care Unit Information for Parents A guide to Anoplasty (anal surgery) Introduction This information leaflet is designed to help parents and families to care for
Opioid Use in Palliative Care
 Opioid Use in Palliative Care Relief of pain is one of the core components of palliative care 1,2 Up to 69% of patients with advanced cancer experience pain 3 ~65% of patients dying from nonmalignant disease
Opioid Use in Palliative Care Relief of pain is one of the core components of palliative care 1,2 Up to 69% of patients with advanced cancer experience pain 3 ~65% of patients dying from nonmalignant disease
OIC, opioid-induced constipation.
 Disclosures Charles E. Argoff, MD Speakers Bureau for Allergan, Inc., AstraZeneca plc, Depomed, Inc., Iroko Pharmaceuticals LLC, Janssen Pharmaceuticals, Inc., Millenium Laboratories, and Xenoport Inc.
Disclosures Charles E. Argoff, MD Speakers Bureau for Allergan, Inc., AstraZeneca plc, Depomed, Inc., Iroko Pharmaceuticals LLC, Janssen Pharmaceuticals, Inc., Millenium Laboratories, and Xenoport Inc.
MANAGEMENT OF CHRONIC CONSTIPATION BEYOND LAXATIVES
 Enrique Rey Professor of Medicine Head. Department of Digestive Diseases Hospital Clínico San Carlos Complutense University Madrid, Spain MANAGEMENT OF CHRONIC CONSTIPATION BEYOND LAXATIVES CONSTIPATION:
Enrique Rey Professor of Medicine Head. Department of Digestive Diseases Hospital Clínico San Carlos Complutense University Madrid, Spain MANAGEMENT OF CHRONIC CONSTIPATION BEYOND LAXATIVES CONSTIPATION:
ARDELYX REPORTS POSITIVE T3MPO-2 PHASE 3 TRIAL RESULTS IN IBS-C
 ARDELYX REPORTS POSITIVE T3MPO-2 PHASE 3 TRIAL RESULTS IN IBS-C OCTOBER 11, 2017 NASDAQ: ARDX FORWARD-LOOKING STATEMENTS To the extent that statements contained in this presentation are not descriptions
ARDELYX REPORTS POSITIVE T3MPO-2 PHASE 3 TRIAL RESULTS IN IBS-C OCTOBER 11, 2017 NASDAQ: ARDX FORWARD-LOOKING STATEMENTS To the extent that statements contained in this presentation are not descriptions
COMMITTEE ON HERBAL MEDICINAL PRODUCTS (HMPC) DRAFT COMMUNITY HERBAL MONOGRAPH ON PSYLLIUM SEED (PLANTAGO AFRA ET PLANTAGO INDICA, SEMEN)
 European Medicines Agency Evaluation of Medicines for Human Use London, 24 October 2005 Doc. Ref. EMEA/HMPC/340865/2005 COMMITTEE ON HERBAL MEDICINAL PRODUCTS (HMPC) DRAFT COMMUNITY HERBAL MONOGRAPH ON
European Medicines Agency Evaluation of Medicines for Human Use London, 24 October 2005 Doc. Ref. EMEA/HMPC/340865/2005 COMMITTEE ON HERBAL MEDICINAL PRODUCTS (HMPC) DRAFT COMMUNITY HERBAL MONOGRAPH ON
PALLIATIVE TREATMENT BY DR. KHRONGKAMOL SIHABAN MEDICAL ONCOLOGIST
 PALLIATIVE TREATMENT BY DR. KHRONGKAMOL SIHABAN MEDICAL ONCOLOGIST TREATMENT IN ONCOLOGY Main treatment : surgery Neoadjuvant treatment : RT, CMT Adjuvant treatment : Tx micrometastatic disease -CMT,Targeted
PALLIATIVE TREATMENT BY DR. KHRONGKAMOL SIHABAN MEDICAL ONCOLOGIST TREATMENT IN ONCOLOGY Main treatment : surgery Neoadjuvant treatment : RT, CMT Adjuvant treatment : Tx micrometastatic disease -CMT,Targeted
UBS Global Healthcare Conference May 19, 2014
 UBS Global Healthcare Conference May 19, 2014 Safe Harbor Statement This presentation may contain forward-looking statements within the meaning of Section 27A of the Securities Act of 1933 and Section
UBS Global Healthcare Conference May 19, 2014 Safe Harbor Statement This presentation may contain forward-looking statements within the meaning of Section 27A of the Securities Act of 1933 and Section
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS
 ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 This medicinal product is subject to additional monitoring. This will allow quick identification of new safety information. Healthcare professionals are asked
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 This medicinal product is subject to additional monitoring. This will allow quick identification of new safety information. Healthcare professionals are asked
GI Pharmacology -4 Irritable Bowel Syndrome and Antiemetics. Dr. Alia Shatanawi
 GI Pharmacology -4 Irritable Bowel Syndrome and Antiemetics Dr. Alia Shatanawi 11-04-2018 Drugs used in Irritable Bowel Syndrome Idiopathic, chronic, relapsing disorder characterized by abdominal discomfort
GI Pharmacology -4 Irritable Bowel Syndrome and Antiemetics Dr. Alia Shatanawi 11-04-2018 Drugs used in Irritable Bowel Syndrome Idiopathic, chronic, relapsing disorder characterized by abdominal discomfort
Daniel Canafax, PharmD VP, Clinical Research Theravance, Inc.
 Demonstrates Improvement in Bowel Movement Frequency and Bristol Stool Scores in a Phase 2b Study of Patients with Opioid-Induced Constipation (OIC) Ross Vickery, PhD, 1 Yu-Ping Li, PhD, 1 Ullrich Schwertschlag,
Demonstrates Improvement in Bowel Movement Frequency and Bristol Stool Scores in a Phase 2b Study of Patients with Opioid-Induced Constipation (OIC) Ross Vickery, PhD, 1 Yu-Ping Li, PhD, 1 Ullrich Schwertschlag,
Costing statement: chronic idiopathic constipation - Lubiprostone
 Putting NICE guidance into practice Costing statement: chronic idiopathic constipation - Lubiprostone Implementing the NICE guidance on Lubiprostone for treating chronic idiopathic constipation (TA318)
Putting NICE guidance into practice Costing statement: chronic idiopathic constipation - Lubiprostone Implementing the NICE guidance on Lubiprostone for treating chronic idiopathic constipation (TA318)
CHAPTER 11 Functional Gastrointestinal Disorders (FGID) Mr. Ashok Kumar Dept of Pharmacy Practice SRM College of Pharmacy SRM University
 CHAPTER 11 Functional Gastrointestinal Disorders (FGID) Mr. Ashok Kumar Dept of Pharmacy Practice SRM College of Pharmacy SRM University 1 Definition of FGID Chronic and recurrent symptoms of the gastrointestinal
CHAPTER 11 Functional Gastrointestinal Disorders (FGID) Mr. Ashok Kumar Dept of Pharmacy Practice SRM College of Pharmacy SRM University 1 Definition of FGID Chronic and recurrent symptoms of the gastrointestinal
Applying Quality to Postsurgical Opioid-Induced Constipation
 Applying Quality to Postsurgical Opioid-Induced Constipation Applying Quality to Postsurgical Opioid-Induced Constipation Ernest J. Dole, PharmD, PhC, FASHP, BCPS Clinical Pharmacist University of New
Applying Quality to Postsurgical Opioid-Induced Constipation Applying Quality to Postsurgical Opioid-Induced Constipation Ernest J. Dole, PharmD, PhC, FASHP, BCPS Clinical Pharmacist University of New
PRUCAPLA Tablets (Prucalopride)
 Published on: 2 May 2018 PRUCAPLA Tablets (Prucalopride) Composition PRUCAPLA 1 mg Each film coated tablet contains: Prucalopride succinate equivalent to Prucalopride 1 mg Excipients. q. s. Color: Titanium
Published on: 2 May 2018 PRUCAPLA Tablets (Prucalopride) Composition PRUCAPLA 1 mg Each film coated tablet contains: Prucalopride succinate equivalent to Prucalopride 1 mg Excipients. q. s. Color: Titanium
Guidelines NICE, not NICE and the Daily Mail. Dr Andy Poullis Consultant Gastroenterologist
 Guidelines NICE, not NICE and the Daily Mail 2018 Dr Andy Poullis Consultant Gastroenterologist Coeliac IBS Gall bladder polyps PEI PPI Who to test for Coeliac persistent unexplained abdominal or gastrointestinal
Guidelines NICE, not NICE and the Daily Mail 2018 Dr Andy Poullis Consultant Gastroenterologist Coeliac IBS Gall bladder polyps PEI PPI Who to test for Coeliac persistent unexplained abdominal or gastrointestinal
European Medicines Agency Evaluation of Medicines for Human Use. Superseded COMMITTEE ON HERBAL MEDICINAL PRODUCTS (HMPC) FINAL
 European Medicines Agency Evaluation of Medicines for Human Use COMMITTEE ON HERBAL MEDICINAL PRODUCTS (HMPC) FINAL COMMUNITY HERBAL MONOGRAPH ON LINUM USITATISSIMUM L., SEMEN DISCUSSION IN THE DRAFTING
European Medicines Agency Evaluation of Medicines for Human Use COMMITTEE ON HERBAL MEDICINAL PRODUCTS (HMPC) FINAL COMMUNITY HERBAL MONOGRAPH ON LINUM USITATISSIMUM L., SEMEN DISCUSSION IN THE DRAFTING
COMMITTEE ON HERBAL MEDICINAL PRODUCTS (HMPC) FINAL COMMUNITY HERBAL MONOGRAPH ON LINUM USITATISSIMUM L., SEMEN
 European Medicines Agency Evaluation of Medicines for Human Use London, 26 October 2006 Doc. Ref: EMEA/HMPC/340849/2005 COMMITTEE ON HERBAL MEDICINAL PRODUCTS (HMPC) FINAL COMMUNITY HERBAL MONOGRAPH ON
European Medicines Agency Evaluation of Medicines for Human Use London, 26 October 2006 Doc. Ref: EMEA/HMPC/340849/2005 COMMITTEE ON HERBAL MEDICINAL PRODUCTS (HMPC) FINAL COMMUNITY HERBAL MONOGRAPH ON
