Impact of a Pharmacist Implemented Protocol on Overall Use of Alvimopan (Entereg ) and Length of Stay in Laparoscopic Colorectal Surgeries
|
|
|
- Marshall Jenkins
- 6 years ago
- Views:
Transcription
1 Journal of Pharmacy and Pharmacology 4 (2016) doi: / / D DAVID PUBLISHING Impact of a Pharmacist Implemented Protocol on Overall Use of Alvimopan (Entereg ) and Length of Stay in Laparoscopic Colorectal Surgeries Halle M. Orlinski, Rachana R. Patel, Bradley J. Champagne, Joseph A. Trunzo and Karen L. Kier University Hospitals St. John Medical Center, Westlake, Ohio 44145, USA Abstract: The primary objective of this study is to assess the impact of a pharmacist-implemented protocol on number of post-operative alvimopan doses. The secondary objective of this study is to assess LOS (length of stay), in days, before and after protocol implementation. A retrospective chart review was conducted from October 2015 through March 2016 for all laparoscopic colorectal surgeries. Number of post-operative alvimopan doses received and LOS was recorded for each patient that received at least one dose of alvimopan. Comparative data, before protocol implementation, from November 2014 through June 2015 were analyzed against the study data. Number of post-operative alvimopan doses and LOS were recorded. The mean number of doses was 6.41 in the comparator group and 4.25 in the study group (probability size P < 0.001), which did meet statistical significance. Although the secondary objective was not statistically significant, LOS slightly decreased as the mean LOS was 5.01 days in the comparator group versus 4.49 days in the study group (P = 0.256). At the current price of $120 per capsule, close to $30,000 was saved during the study period, projecting an annual cost savings of approximately $68,000. Results from this study show that pharmacists can play a vital role in cost savings and ensuring appropriate use of certain high-risk medications, like alvimopan, without increasing overall length of stay. Key words: Alvimopan, pharmacist-implemented, colorectal, cost savings. 1. Introduction POI (post-operative ileus) is defined as the transient cessation of bowel motility after GI (gastrointestinal) or other surgery. POI is a common cause of morbidity and prolonged patient recovery, leading to increased hospital costs, utilization of healthcare resources and higher readmission rates. POI is thought to be a multifactorial phenomenon brought on by inflammation from surgical manipulation, opioid use and endogenous hormones that all work to slow down GI motility [1]. Characteristics of POI can include abdominal pain, nausea and vomiting, and diet intolerance. Recent studies show that 20% of patients undergoing laparoscopic GI surgery will develop POI, increasing their length of stay by approximately five days. It is estimated that approximately $1,500,000,000 is spent annually to manage the total hospital costs that Corresponding author: Halle M. Orlinski, Pharm.D., research fields: critical care, trauma and infectious diseases. go along with POI [2, 3]. It was once thought that restricting oral feedings would help prevent nausea and vomiting, as well as protect the anastomosis from the stress of digesting food, allowing it time to heal. However, several studies have shown that earlier oral feedings have led to quicker GI recovery times, fewer post-operative complications, and consequently shorter hospital stays. Proper nutrition is necessary for wound healing and therefore a vital piece in the recovery process [4]. Alvimopan (Entereg ) is a peripherally acting, mu-opioid, receptor antagonist indicated to accelerate the time to upper and lower GI recovery following surgeries that include partial bowel resection with primary anastomosis. Acting peripherally, alvimopan has shown to accelerate the time to GI recovery by antagonizing mu-opioid receptors in the GI tract. Due to its inability to penetrate the CNS (central nervous system) and its large molecular size, alvimopan still
2 522 Impact of a Pharmacist Implemented Protocol on Overall Use of Alvimopan (Entereg ) allows analgesia to take place due to its inability to act on the mu-opioid receptors found in the central nervous system. In-vitro binding also shows that alvimopan binds with a high affinity to the mu-opioid receptor, but it has little to no activity towards the delta-opioid or kappa-opioid receptors [5]. The most common adverse reaction seen in patients taking alvimopan was dyspepsia (incidence 1.5%). Alvimopan 12 mg should be administered orally 30 min to 5 h prior to surgery, followed by 12 mg orally twice daily, beginning on post-operative Day 1, for a maximum of seven days, or 15 total doses. According to the FDA (Food and Drug Administration), there are no dosage adjustments for elderly patients, those with mild-moderate hepatic impairment or mild-severe renal impairment. It is recommended that these patients be monitored for adverse reactions. Alvimopan is not recommended in patients with severe hepatic impairment or those with end-stage renal disease. Contraindications to alvimopan therapy include patients who have taken therapeutic doses of opioids for more than seven consecutive days prior to taking alvimopan. In addition, patients recently exposed to opioids are expected to be more sensitive to the effects of alvimopan and may experience abdominal pain, nausea, vomiting and diarrhea. Alvimopan therapy is also not recommended in patients with a complete GI obstruction or in patients undergoing surgery for complete bowel obstruction. All patients receiving alvimopan therapy should be monitored for adverse reactions. Alvimopan is restricted to a total of 15 in-hospital doses and is associated with a REMS (Risk Evaluation and Mitigation Strategy) program. This program, E.A.S.E. (Entereg Access Support and Education), is to ensure that only certified hospital pharmacies are dispensing alvimopan, and appropriate staff and physicians are educated on the medication. The E.A.S.E program was brought about to limit the cumulative number of doses which a patient may receive due to the increased risk of myocardial infarction with prolonged use [6]. A Phase-3, double-blind, placebo-controlled study, conducted over 12 months, using alvimopan 0.5 mg vs. placebo in patients with chronic non-cancer pain, showed an increased number of myocardial infarctions in the alvimopan group compared to placebo, 1.3% vs. 0%, respectively [7]. In this study, the majority of myocardial infarctions occurred in the first four months of therapy. A causal relationship has not been established [6]. In 2014, SJMC (St. John Medical Center) Pharmacy dispensed 1,102 doses of alvimopan, costing approximately $126,000. Current usage of alvimopan at SJMC for 2015 was approaching the annual usage of 2014, in just the first six months of the year. Increased use of alvimopan at SJMC was attributed to growth of the colorectal surgery service, as well as a less stringent discontinuation process. A pharmacist-initiated protocol, which will be discussed below, was put into place to ensure proper use of alvimopan and limit any unnecessary spending that results from unwarranted doses. As previously mentioned, alvimopan use at SJMC had been increasing due to an increased number of colorectal surgeries and lack of regimented discontinuation by prescribers. Formerly, alvimopan doses could be held at nursing discretion if a patient was tolerating their GI soft diet and having bowel movements, through a nursing notification to pharmacy, but this seldom occurred. Typically, alvimopan was discontinued from the medication profile at the physician s discretion. At a majority of the time, a patient would be tolerating a GI soft diet and having bowel movements, yet he would still be receiving alvimopan inappropriately, leading to increased exposure to this high-risk medication and increased usage of a high-cost medication. A pharmacist-driven initiative, with physician input, was put into place ensuring alvimopan therapy would be discontinued at the appropriate time. As mentioned previously, early oral feedings have shown
3 Impact of a Pharmacist Implemented Protocol on Overall Use of Alvimopan (Entereg ) 523 to accelerate the time to GI recovery; Therefore, accelerated diet orders were made available for the colorectal surgeons to advance feedings for patients they deemed appropriate. Order sets were also updated to ensure that every patient without contraindications received a pre-operative dose of alvimopan, as well as post-operative doses, twice daily, until bowel function was restored, or a maximum of 15 doses, whichever happened first. Furthermore, physicians agreed to hold the alvimopan dose just prior to the administration of a soft diet order to help cut unnecessary doses. Therefore, if the soft diet was tolerated, an unnecessary dose of alvimopan was not given in the morning prior to the patient receiving their breakfast tray, cutting back on at least one dose of alvimopan, if not more. In addition, pharmacists also began reviewing contraindications for every patient on alvimopan therapy on post-operative Day 1. The colorectal surgeons reviewed contraindications to alvimopan therapy for their respective patients pre-operatively, while the pharmacist was able to serve as a double-check in the post-operative period. 2. Methods The research protocol received exempt status from St. Vincent Medical Center IRB (Investigational Review Board) in November Retrospective data were collected from October 2015 to March The data included number of post-operative doses of alvimopan and length of stay, in days. Patient identifiers were not collected. All data were stored in a web-based database. Informed consent was not required because no changes were made to current practice. During the study period, patients received alvimopan 12 mg orally twice daily post-operatively until toleration of a GI soft diet or bowel movements were presented. Nurses, under physician orders, agreed to hold alvimopan doses prior to initiation of a GI soft diet in order to cut down on unnecessary alvimopan exposure. Pharmacists already reviewed contraindications to alvimopan therapy on post-operative day one for all patients receiving therapy. Colorectal surgeons reviewed contraindications to alvimopan therapy pre-operatively for their respective patients. The primary objective of this study was to assess the impact of a pharmacist-implemented protocol on number of post-operative alvimopan doses, before and after protocol implementation. The secondary objective of this study was to assess LOS (length of stay), in days, before and after protocol implementation. Statistical analysis was done for both the primary and secondary endpoints. A chi-square analysis was used to evaluate demographic data to ensure both sample groups (pre- and post-protocol implementation) were similar prior to running the primary variable. A Student s t-test was used to analyze total post-operative doses of alvimopan, as well as compare length of stay post-operatively. Power was set at 80% (assumed extent α = 0.05) to detect a difference of two doses, or one day of therapy, between the comparator group and the study group. The minimum sample size needed was 64 patients per group, to detect a reduction of two doses of alvimopan post-operatively post-protocol implementation. 3. Results A total of 225 patients met inclusion criteria, 114 patients in the study group (post-protocol implementation) and 111 patients in the comparator group (pre-protocol implementation). No differences were noted between the two sample groups. The mean number of doses was 6.41 in the comparator group and 4.25 in the study group (probability size P < 0.001, 95% CI (confidence interval): 1.441, 2.890), which did meet statistical significance. Although the secondary objective was not statistically significant, LOS slightly decreased post-protocol implementation, as the mean LOS was 5.01 days in the comparator group versus 4.49 days in the study group (P = 0.256, 95% CI: 0.379, 2.894) (Table 1).
4 524 Impact of a Pharmacist Implemented Protocol on Overall Use of Alvimopan (Entereg ) Table 1 Primary and secondary endpoint results. Items Study group Comparator group 95% CI (confidence P value (n = 111) (n = 114) interval) Post-operative alvimopan doses < , Length of stay (days) , Discussion This study showed that pharmacists can make meaningful interventions in the post-operative setting that can impact patient care in a positive way. Previous studies have shown the efficacy that alvimopan can have in accelerating time to GI recovery in colorectal surgery. In contrast, studies have also found increased risk of myocardial infarction with prolonged use. Therefore, use of alvimopan must be for the appropriate indication and the shortest amount of time possible to avoid overexposure. This study had many positive implications. Power was far exceeded, with 115 patients in the study group and 111 in the comparator group. The primary endpoint was statistically significant, showing a reduction in alvimopan usage of 2.16 doses per patient post-operatively, without leading to an increase in length of stay. By reducing the amount of alvimopan usage post-operatively, a cost-savings of approximately $30,000 resulted during the study period, projecting an annual cost savings of $68,000. This was only due to drug savings alone, not taking into account the shorter length of hospital stay in the post-protocol implementation group, or any adverse effects from prolonged exposure to alvimopan that could have led to escalated care and increased utilization of resources. Nonetheless, there are several limitations to this study. Only patients who underwent laparoscopic colorectal surgeries were included, therefore excluding open colorectal procedures. Also, patients who went to intensive care unit floors post-operatively were excluded. These two factors alone excluded a high-risk subset of patients that could have benefited from inclusion into the study protocol. Furthermore, additional screening by a pharmacist for contraindications to alvimopan therapy was limited to the post-operative phase. Therefore patients had the potential to receive alvimopan pre-operatively in the setting of a contraindication. Future studies should include open colorectal surgeries and patients transferred to intensive care units post-operatively, as both of these patient populations are considered high-risk and could benefit from inclusion in the pharmacist-implemented protocol. 5. Conclusions Based on the study result, pharmacists can have an impact in many areas regarding high-risk, high-cost medications in the post-operative setting. Unnecessary doses can be eliminated without increasing length of stay in post-operative laparoscopic colorectal surgery patients. In addition, this study demonstrated an area of cost-savings. Future studies should investigate the inclusion of high-risk patients, such as those who underwent open colorectal procedures, as well as patients who were transferred to intensive care units post-operatively. These patients are a unique subset of patients who could potentially benefit from the inclusion into the pharmacist-implemented protocol. References [1] Lubawski, J., and Saclarides, T Postoperative Ileus: Strategies for Reduction. Therapeutics and Clinical Risk Management 4 (5): [2] Nguyen, D. L., Maithel, S., Nguyen, E. T., and Bechtold, M. L Does Alvimopan Return Bowel Function in Laparoscopic Gastrointestinal Surgery? A Meta-analysis. Annals of Gastroenterology 28: 1-7. [3] Wang, S., Shah, N., Philip, J., Caraccio, T., Feuerman, M., and Malone, B Role of Alvimopan (Entereg ) in Gastrointestinal Recovery and Hospital Length of Stay after Bowel Resection. P&T 37 (9): [4] Zhuang, C. L., Ye, X. Z., Zhang, C. J., Dong, Q. T., Chen, B. C., and Yu, Z Early Versus Traditional
5 Impact of a Pharmacist Implemented Protocol on Overall Use of Alvimopan (Entereg ) 525 Postoperative Oral Feeding in Patients Undergoing Elective Colorectal Surgery: A Meta-analysis of Randomized Clinical Trials. Dig. Surg. 30: [5] Kraft, M., MacLaren, R., Du, W., and Owens, G Alvimopan (Entereg) for the Management of Postoperative Ileus in Patients Undergoing Bowel Resection. P&T 35 (1): [6] Whitehouse Station Entereg (Alvimopan) Package Insert. New Jersey: Merck. [7] GlaxoSmithKline and Adolor Corporation Press Release. GSK and Adolor Announce Preliminary Results from Phase 3 Safety Study of Alvimopan (Alvimopan/Entereg ). GlaxoSmithKline and Adolor Corporation Press. Accessed September 2, rol-newsarticle&t=regular&id=982960&.
Role of Alvimopan (Entereg) in Gastrointestinal Recovery And Hospital Length of Stay After Bowel Resection
 Role of Alvimopan (Entereg) in Gastrointestinal Recovery And Hospital Length of Stay After Bowel Resection Shan Wang, PharmD, RPh; Neal Shah, PharmD Candidate; Jessin Philip, PharmD, RPh; Tom Caraccio,
Role of Alvimopan (Entereg) in Gastrointestinal Recovery And Hospital Length of Stay After Bowel Resection Shan Wang, PharmD, RPh; Neal Shah, PharmD Candidate; Jessin Philip, PharmD, RPh; Tom Caraccio,
Postoperative Ileus. UCSF Postgraduate Course in General Surgery Maui, HI March 20, 2011
 Postoperative Ileus UCSF Postgraduate Course in General Surgery Maui, HI March 20, 2011 Hobart W. Harris, MD, MPH Introduction Pathophysiology Clinical Research Management Summary Postoperative Ileus:
Postoperative Ileus UCSF Postgraduate Course in General Surgery Maui, HI March 20, 2011 Hobart W. Harris, MD, MPH Introduction Pathophysiology Clinical Research Management Summary Postoperative Ileus:
ENTEREG (alvimopan) Capsules Initial U.S. Approval: 2008
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use ENTEREG safely and effectively. See full prescribing information for ENTEREG. ENTEREG (alvimopan)
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use ENTEREG safely and effectively. See full prescribing information for ENTEREG. ENTEREG (alvimopan)
Multimodal Approach for Managing Postoperative Ileus: Role of Health- System Pharmacists (ACPE program H01P)
 1. In the normal gastrointestinal tract, what percent of nutrient absorption occurs in the jejunum? a. 20%. b. 40%. c. 70%. d. 90%. 2. According to Dr. Erstad, the four components of gastrointestinal control
1. In the normal gastrointestinal tract, what percent of nutrient absorption occurs in the jejunum? a. 20%. b. 40%. c. 70%. d. 90%. 2. According to Dr. Erstad, the four components of gastrointestinal control
ALVIMOPAN 0.0 OVERVIEW
 ALVIMOPAN 0.0 OVERVIEW A. Alvimopan is a peripherally restricted mu-opioid receptor antagonist. B. DOSING INFORMATION : For the treatment of opioid bowel dysfunction, oral alvimopan doses between 0.5 milligrams
ALVIMOPAN 0.0 OVERVIEW A. Alvimopan is a peripherally restricted mu-opioid receptor antagonist. B. DOSING INFORMATION : For the treatment of opioid bowel dysfunction, oral alvimopan doses between 0.5 milligrams
Fast Track Surgery and Surgical Carepath in Optimising Colorectal Surgery. R Sim Centre for Advanced Laparoscopic Surgery, TTSH
 Fast Track Surgery and Surgical Carepath in Optimising Colorectal Surgery R Sim Centre for Advanced Laparoscopic Surgery, TTSH Conventional Surgery Postop care Nasogastric tube Enteral feeds when ileus
Fast Track Surgery and Surgical Carepath in Optimising Colorectal Surgery R Sim Centre for Advanced Laparoscopic Surgery, TTSH Conventional Surgery Postop care Nasogastric tube Enteral feeds when ileus
Enhanced Recovery after Surgery - A Colorectal Perspective. R Sim Centre for Advanced Laparoscopic Surgery, TTSH
 Enhanced Recovery after Surgery - A Colorectal Perspective R Sim Centre for Advanced Laparoscopic Surgery, TTSH Conventional Surgery Postop care Nasogastric tube Enteral feeds when ileus resolves Opioid
Enhanced Recovery after Surgery - A Colorectal Perspective R Sim Centre for Advanced Laparoscopic Surgery, TTSH Conventional Surgery Postop care Nasogastric tube Enteral feeds when ileus resolves Opioid
Opioid-Induced Constipation
 Objectives Opioid-Induced Constipation Brianna Jansma, PharmD Alex Smith, PharmD Megan Robinson, PharmD Summarize epidemiology of opioid-induced constipation (OIC) Understand opiates effects on the gastrointestinal
Objectives Opioid-Induced Constipation Brianna Jansma, PharmD Alex Smith, PharmD Megan Robinson, PharmD Summarize epidemiology of opioid-induced constipation (OIC) Understand opiates effects on the gastrointestinal
R Sim, D Cheong, KS Wong, B Lee, QY Liew Tan Tock Seng Hospital Singapore
 Prospective randomized, double-blind, placebo-controlled study of pre- and postoperative administration of a COX-2- specific inhibitor as opioid-sparing analgesia in major colorectal resections R Sim,
Prospective randomized, double-blind, placebo-controlled study of pre- and postoperative administration of a COX-2- specific inhibitor as opioid-sparing analgesia in major colorectal resections R Sim,
Lehman Brothers Eighth Annual Global Healthcare Conference March 30, 2005 Bruce A. Peacock President and Chief Executive Officer
 Lehman Brothers Eighth Annual Global Healthcare Conference March 30, 2005 Bruce A. Peacock President and Chief Executive Officer 2005 Adolor Corporation. All rights reserved. Safe Harbor Statement This
Lehman Brothers Eighth Annual Global Healthcare Conference March 30, 2005 Bruce A. Peacock President and Chief Executive Officer 2005 Adolor Corporation. All rights reserved. Safe Harbor Statement This
ERAS. Presented by Timothy L. Beard MD, FACS, CPI Bend Memorial Clinic
 ERAS Presented by Timothy L. Beard MD, FACS, CPI Bend Memorial Clinic Outline Definition Justification Ileus Pain Outline Specifics Data BMC Data Worldwide Data Implementation What is ERAS? AKA Fast-track
ERAS Presented by Timothy L. Beard MD, FACS, CPI Bend Memorial Clinic Outline Definition Justification Ileus Pain Outline Specifics Data BMC Data Worldwide Data Implementation What is ERAS? AKA Fast-track
Clinical and Economic Outcomes of Prolonged Postoperative Ileus in Patients Undergoing Hysterectomy and Hemicolectomy
 Clinical and Economic Outcomes of Prolonged Postoperative Ileus in Patients Undergoing Hysterectomy and Hemicolectomy Christopher G. Salvador, PharmD, Mirko Sikirica, PharmD, Adam Evans, BS, Laura Pizzi,
Clinical and Economic Outcomes of Prolonged Postoperative Ileus in Patients Undergoing Hysterectomy and Hemicolectomy Christopher G. Salvador, PharmD, Mirko Sikirica, PharmD, Adam Evans, BS, Laura Pizzi,
Dsuvia (sufentanil) NEW PRODUCT SLIDESHOW
 Dsuvia (sufentanil) NEW PRODUCT SLIDESHOW Introduction Brand name: Dsuvia Generic name: Sufentanil Pharmacological class: Opioid agonist Strength and Formulation: 30mcg; sublingual tabs (housed in a disposable,
Dsuvia (sufentanil) NEW PRODUCT SLIDESHOW Introduction Brand name: Dsuvia Generic name: Sufentanil Pharmacological class: Opioid agonist Strength and Formulation: 30mcg; sublingual tabs (housed in a disposable,
Understanding the Benefits and Risks
 LOTRONEX and its authorized generic alosetron hydrochloride: Understanding the Benefits and Risks The LOTRONEX REMS Program Prescriber Education Slide Deck LOTRONEX is a registered trademark of Prometheus
LOTRONEX and its authorized generic alosetron hydrochloride: Understanding the Benefits and Risks The LOTRONEX REMS Program Prescriber Education Slide Deck LOTRONEX is a registered trademark of Prometheus
44 P&T January 2010 Vol. 35 No. 1. Various strategies have been implemented in attempts to reduce the duration of POI, including: 9 11
 (Entereg) for the Management Of Postoperative Ileus in Patients Undergoing Bowel Resection Michael Kraft, PharmD, BCNSP; Robert MacLaren, PharmD; Wei Du, PhD; and Gay Owens, PharmD ABSTRACT Postoperative
(Entereg) for the Management Of Postoperative Ileus in Patients Undergoing Bowel Resection Michael Kraft, PharmD, BCNSP; Robert MacLaren, PharmD; Wei Du, PhD; and Gay Owens, PharmD ABSTRACT Postoperative
Movantik (naloxegol), Relistor (methylnaltrexone bromide)
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.50.06 Subject: Opioid Antagonist Drug Class Page: 1 of 5 Last Review Date: December 2, 2016 Opioid Antagonist
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.50.06 Subject: Opioid Antagonist Drug Class Page: 1 of 5 Last Review Date: December 2, 2016 Opioid Antagonist
A Retrospective Study of Patients Undergoing Radical Cystectomy and Receiving Peri-Operative Naloxegol or Alvimopan: Comparison of Length of Stay
 Journal of Surgery 2018; 6(5): 129-134 http://www.sciencepublishinggroup.com/j/js doi: 10.11648/j.js.20180605.14 ISSN: 2330-0914 (Print); ISSN: 2330-0930 (Online) A Retrospective Study of Patients Undergoing
Journal of Surgery 2018; 6(5): 129-134 http://www.sciencepublishinggroup.com/j/js doi: 10.11648/j.js.20180605.14 ISSN: 2330-0914 (Print); ISSN: 2330-0930 (Online) A Retrospective Study of Patients Undergoing
Movantik (naloxegol), Relistor (methylnaltrexone bromide), Symproic (naldemedine)
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.50.06 Subject: Opioid Antagonist Drug Class Page: 1 of 7 Last Review Date: November 30, 2018 Opioid Antagonist
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.50.06 Subject: Opioid Antagonist Drug Class Page: 1 of 7 Last Review Date: November 30, 2018 Opioid Antagonist
Movantik (naloxegol), Relistor (methylnaltrexone bromide), Symproic (naldemedine)
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.50.06 Subject: Opioid Antagonist Drug Class Page: 1 of 5 Last Review Date: June 22, 2017 Opioid Antagonist
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.50.06 Subject: Opioid Antagonist Drug Class Page: 1 of 5 Last Review Date: June 22, 2017 Opioid Antagonist
Gastrointestinal Feedings Post Op: What s the deal on beginning oral feedings?
 Gastrointestinal Feedings Post Op: What s the deal on beginning oral feedings? Kate Willcutts, DCN, RD, CNSC University of Virginia Health System Charlottesville, VA kfw3w@virginia.edu Objectives 1. Discuss
Gastrointestinal Feedings Post Op: What s the deal on beginning oral feedings? Kate Willcutts, DCN, RD, CNSC University of Virginia Health System Charlottesville, VA kfw3w@virginia.edu Objectives 1. Discuss
Horizon Scanning Technology Summary. Methylnaltrexone for opioid induced constipation in advanced illness and palliative care
 Horizon Scanning Technology Summary National Horizon Scanning Centre Methylnaltrexone for opioid induced constipation in advanced illness and palliative care April 2007 This technology summary is based
Horizon Scanning Technology Summary National Horizon Scanning Centre Methylnaltrexone for opioid induced constipation in advanced illness and palliative care April 2007 This technology summary is based
SYMPROIC (naldemedine tosylate) oral capsule
 SYMPROIC (naldemedine tosylate) oral capsule Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This
SYMPROIC (naldemedine tosylate) oral capsule Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This
LOTRONEX and its authorized generic alosetron hydrochloride:
 LOTRONEX and its authorized generic alosetron hydrochloride: Understanding the Benefits and Risks The Prescribing Program for LOTRONEX TM Prescriber Education Slide Deck PROMETHEUS and the Link Design
LOTRONEX and its authorized generic alosetron hydrochloride: Understanding the Benefits and Risks The Prescribing Program for LOTRONEX TM Prescriber Education Slide Deck PROMETHEUS and the Link Design
TITLE: Alvimopan for Surgical Patients: A Review of the Clinical and Cost-Effectiveness
 TITLE: Alvimopan for Surgical Patients: A Review of the Clinical and Cost-Effectiveness DATE: 06 February 2009 CONTEXT AND POLICY ISSUES: Post-operative ileus (POI) is a condition that affects all patients
TITLE: Alvimopan for Surgical Patients: A Review of the Clinical and Cost-Effectiveness DATE: 06 February 2009 CONTEXT AND POLICY ISSUES: Post-operative ileus (POI) is a condition that affects all patients
Daniel Canafax, PharmD VP, Clinical Research Theravance, Inc.
 Demonstrates Improvement in Bowel Movement Frequency and Bristol Stool Scores in a Phase 2b Study of Patients with Opioid-Induced Constipation (OIC) Ross Vickery, PhD, 1 Yu-Ping Li, PhD, 1 Ullrich Schwertschlag,
Demonstrates Improvement in Bowel Movement Frequency and Bristol Stool Scores in a Phase 2b Study of Patients with Opioid-Induced Constipation (OIC) Ross Vickery, PhD, 1 Yu-Ping Li, PhD, 1 Ullrich Schwertschlag,
Operational Efficiency in Colon Surgery Enhanced Recovery Pathways: 23 hour laparoscopic colectomy
 Enhanced Recovery Pathways: 23 hour laparoscopic colectomy Conor P. Delaney MD MCh PhD Chairman, Digestive Disease Institute Professor of Surgery, Cleveland, Ohio Disclosure Slide Conor Delaney MD PhD
Enhanced Recovery Pathways: 23 hour laparoscopic colectomy Conor P. Delaney MD MCh PhD Chairman, Digestive Disease Institute Professor of Surgery, Cleveland, Ohio Disclosure Slide Conor Delaney MD PhD
D DAVID PUBLISHING. 1. Introduction. Kathryn Koliha 1, Julie Falk 1, Rachana Patel 1 and Karen Kier 2
 Journal of Pharmacy and Pharmacology 5 (2017) 607-615 doi: 10.17265/2328-2150/2017.09.001 D DAVID PUBLISHING Comparative Evaluation of Pharmacist-Managed Vancomycin Dosing in a Community Hospital Following
Journal of Pharmacy and Pharmacology 5 (2017) 607-615 doi: 10.17265/2328-2150/2017.09.001 D DAVID PUBLISHING Comparative Evaluation of Pharmacist-Managed Vancomycin Dosing in a Community Hospital Following
Theravance Announces Positive Results from Phase 1 and Phase 2 Clinical Studies with TD-1211 in Development for Opioid-Induced Constipation
 Theravance Announces Positive Results from Phase 1 and Phase 2 Clinical Studies with TD-1211 in Development for Opioid-Induced Constipation TD-1211 Achieves Primary and Secondary Endpoints SOUTH SAN FRANCISCO,
Theravance Announces Positive Results from Phase 1 and Phase 2 Clinical Studies with TD-1211 in Development for Opioid-Induced Constipation TD-1211 Achieves Primary and Secondary Endpoints SOUTH SAN FRANCISCO,
Prevent gastric distention and vomiting after surgery
 Remove toxic and unwanted substances from the stomach Administration of enteral nutrition, drugs and so on It favors lung expansion in mechanically unconscious and ventilated subjects Aspiration gastric
Remove toxic and unwanted substances from the stomach Administration of enteral nutrition, drugs and so on It favors lung expansion in mechanically unconscious and ventilated subjects Aspiration gastric
The ideal drug therapy for postoperative
 Emerging pharmacologic options for treating postoperative ileus The ideal drug therapy for postoperative ileus (POI), a common surgical complication, would have several characteristics. It would selectively
Emerging pharmacologic options for treating postoperative ileus The ideal drug therapy for postoperative ileus (POI), a common surgical complication, would have several characteristics. It would selectively
Horizon Scanning Technology Briefing. Alvimopan (Entrareg ) for opioid-induced bowel disfunction. National Horizon Scanning Centre.
 Horizon Scanning Technology Briefing National Horizon Scanning Centre Alvimopan (Entrareg ) for opioid-induced bowel disfunction August 2006 This technology briefing is based on information available at
Horizon Scanning Technology Briefing National Horizon Scanning Centre Alvimopan (Entrareg ) for opioid-induced bowel disfunction August 2006 This technology briefing is based on information available at
Alvimopan. Dennis J. Cada, PharmD, FASHP, FASCP (Editor)*; Terri L. Levien, PharmD ; and Danial E. Baker, PharmD, FASHP, FASCP
 Hospital Pharmacy Volume 43, Number 10, pp 819 829 2008 Wolters Kluwer Health, Inc. FORMULARY DRUG REVIEWS Alvimopan Dennis J. Cada, PharmD, FASHP, FASCP (Editor)*; Terri L. Levien, PharmD ; and Danial
Hospital Pharmacy Volume 43, Number 10, pp 819 829 2008 Wolters Kluwer Health, Inc. FORMULARY DRUG REVIEWS Alvimopan Dennis J. Cada, PharmD, FASHP, FASCP (Editor)*; Terri L. Levien, PharmD ; and Danial
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Lotronex) Reference Number: CP.PMN.153 Effective Date: 11.16.16 Last Review Date: 11.18 Line of Business: Commercial, Medicaid Revision Log See Important Reminder at the end of this policy
Clinical Policy: (Lotronex) Reference Number: CP.PMN.153 Effective Date: 11.16.16 Last Review Date: 11.18 Line of Business: Commercial, Medicaid Revision Log See Important Reminder at the end of this policy
Alvimopan (Entereg), a Peripherally Acting mu-opioid Receptor Antagonist For Postoperative Ileus Goldina Ikezuagu Erowele, PharmD
 Alvimopan (Entereg), a Peripherally Acting mu-opioid Receptor Antagonist For Postoperative Ileus Goldina Ikezuagu Erowele, PharmD INTRODUCTION In 2007, Goldstein et al. sought to determine the economic
Alvimopan (Entereg), a Peripherally Acting mu-opioid Receptor Antagonist For Postoperative Ileus Goldina Ikezuagu Erowele, PharmD INTRODUCTION In 2007, Goldstein et al. sought to determine the economic
Clarification of Drug Allergy Information Using a Standardized Drug Allergy Questionnaire and Interview
 Clarification of Drug Allergy Information Using a Standardized Drug Allergy Questionnaire and Interview Amy Harig, PharmD, BCPS; Amy Rybarczyk, PharmD, BCPS; Amanda Benedetti, PharmD; and Jacob Zimmerman,
Clarification of Drug Allergy Information Using a Standardized Drug Allergy Questionnaire and Interview Amy Harig, PharmD, BCPS; Amy Rybarczyk, PharmD, BCPS; Amanda Benedetti, PharmD; and Jacob Zimmerman,
Full details and resource documents available:
 Clinical & Regulatory News by Pharmerica Urinary Tract Infection (UTI) Second Most Common Cause of Hospital Readmission within 30 days UTIs are prevalent and account for up to 22% of infections in LTC,
Clinical & Regulatory News by Pharmerica Urinary Tract Infection (UTI) Second Most Common Cause of Hospital Readmission within 30 days UTIs are prevalent and account for up to 22% of infections in LTC,
The effect of laxative use in length of hospital stay and complication rate in patients undergoing elective colorectal surgery within an ERAS setting.
 The effect of laxative use in length of hospital stay and complication rate in patients undergoing elective colorectal surgery within an ERAS setting. { Thalia Petropoulou, Clinical Fellow Paul Hainsworth,Colorectal
The effect of laxative use in length of hospital stay and complication rate in patients undergoing elective colorectal surgery within an ERAS setting. { Thalia Petropoulou, Clinical Fellow Paul Hainsworth,Colorectal
ENHANCED RECOVERY AFTER SURGERY (ERAS) PATHWAYS PARESH C. SHAH MD FACS VICE CHAIR OF SURGERY DIRECTOR OF GENERAL SURGERY
 Department of Surgery Divison of General Surgery ENHANCED RECOVERY AFTER SURGERY (ERAS) PATHWAYS PARESH C. SHAH MD FACS VICE CHAIR OF SURGERY DIRECTOR OF GENERAL SURGERY December 2016 Disclosure Paresh
Department of Surgery Divison of General Surgery ENHANCED RECOVERY AFTER SURGERY (ERAS) PATHWAYS PARESH C. SHAH MD FACS VICE CHAIR OF SURGERY DIRECTOR OF GENERAL SURGERY December 2016 Disclosure Paresh
In systems that try to minimize hospital stay after abdominal surgery, one of. Pharmacological management of postoperative ileus
 FORMATION MÉDICALE CONTINUE SURGICAL BIOLOGY FOR THE CLINICIAN Pharmacological management of postoperative ileus Farhad Zeinali, MD Jonah J. Stulberg, MPH Conor P. Delaney, MD, MCh, PhD Department of Surgery,
FORMATION MÉDICALE CONTINUE SURGICAL BIOLOGY FOR THE CLINICIAN Pharmacological management of postoperative ileus Farhad Zeinali, MD Jonah J. Stulberg, MPH Conor P. Delaney, MD, MCh, PhD Department of Surgery,
Laparoscopic Colorectal Surgery
 Laparoscopic Colorectal Surgery 20 th November 2015 Dr Adam Cichowitz General Surgeon Laparoscopic Colorectal Surgery Introduced in early 1990s Uptake slow Steep learning curve Requirement for equipment
Laparoscopic Colorectal Surgery 20 th November 2015 Dr Adam Cichowitz General Surgeon Laparoscopic Colorectal Surgery Introduced in early 1990s Uptake slow Steep learning curve Requirement for equipment
LONG TERM OUTCOME OF ELECTIVE SURGERY
 LONG TERM OUTCOME OF ELECTIVE SURGERY Roberto Persiani Associate Professor Mini-invasive Oncological Surgery Unit Institute of Surgical Pathology (Dir. prof. D. D Ugo) Dis Colon Rectum, March 2000 Dis
LONG TERM OUTCOME OF ELECTIVE SURGERY Roberto Persiani Associate Professor Mini-invasive Oncological Surgery Unit Institute of Surgical Pathology (Dir. prof. D. D Ugo) Dis Colon Rectum, March 2000 Dis
The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only.
 The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only. Please note that the results reported in any single trial may not reflect the overall
The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only. Please note that the results reported in any single trial may not reflect the overall
Dispensing and administration of emergency opioid antagonist without a
 68-7-23. Dispensing and administration of emergency opioid antagonist without a prescription. (a) A pharmacist may dispense an FDA-approved emergency opioid antagonist and the necessary medical supplies
68-7-23. Dispensing and administration of emergency opioid antagonist without a prescription. (a) A pharmacist may dispense an FDA-approved emergency opioid antagonist and the necessary medical supplies
Thoracic epidural versus patient-controlled analgesia in elective bowel resections Paulsen E K, Porter M G, Helmer S D, Linhardt P W, Kliewer M L
 Thoracic epidural versus patient-controlled analgesia in elective bowel resections Paulsen E K, Porter M G, Helmer S D, Linhardt P W, Kliewer M L Record Status This is a critical abstract of an economic
Thoracic epidural versus patient-controlled analgesia in elective bowel resections Paulsen E K, Porter M G, Helmer S D, Linhardt P W, Kliewer M L Record Status This is a critical abstract of an economic
5.2 Strategies to Optimize Delivery and Minimize Risks of EN: Motility Agents May 2015
 5.2 Strategies to Optimize Delivery and Minimize Risks of EN: Motility Agents May 2015 There were no new randomized controlled trials since the 2009 and 2013 updates and hence there are no changes to the
5.2 Strategies to Optimize Delivery and Minimize Risks of EN: Motility Agents May 2015 There were no new randomized controlled trials since the 2009 and 2013 updates and hence there are no changes to the
National Horizon Scanning Centre. Methylnaltrexone (MOA-728) for postoperative ileus. April 2008
 (MOA-728) for postoperative ileus April 2008 This technology summary is based on information available at the time of research and a limited literature search. It is not intended to be a definitive statement
(MOA-728) for postoperative ileus April 2008 This technology summary is based on information available at the time of research and a limited literature search. It is not intended to be a definitive statement
Interprofessional Trauma Conference September 28th 2018 Montreal
 Interprofessional Trauma Conference September 28th 2018 Montreal Marc Perreault & Marc Alexandre Duceppe ICU Pharmacists MGH & RVH-CUSM Faculté de Pharmacie Université de Montréal I have no potential conflict
Interprofessional Trauma Conference September 28th 2018 Montreal Marc Perreault & Marc Alexandre Duceppe ICU Pharmacists MGH & RVH-CUSM Faculté de Pharmacie Université de Montréal I have no potential conflict
Fluid Balance in an Enhanced Recovery Pathway. Edwin Itenberg, DO, FACS, FASCRS St. Joseph Mercy Oakland MSQC/ASPIRE Meeting April 28, 2017
 Fluid Balance in an Enhanced Recovery Pathway Edwin Itenberg, DO, FACS, FASCRS St. Joseph Mercy Oakland MSQC/ASPIRE Meeting April 28, 2017 No Disclosures 2 Introduction The optimal intravenous fluid regimen
Fluid Balance in an Enhanced Recovery Pathway Edwin Itenberg, DO, FACS, FASCRS St. Joseph Mercy Oakland MSQC/ASPIRE Meeting April 28, 2017 No Disclosures 2 Introduction The optimal intravenous fluid regimen
Current evidence in acute pain management. Jeremy Cashman
 Current evidence in acute pain management Jeremy Cashman Optimal analgesia Best possible pain relief Lowest incidence of side effects Optimal analgesia Best possible pain relief Lowest incidence of side
Current evidence in acute pain management Jeremy Cashman Optimal analgesia Best possible pain relief Lowest incidence of side effects Optimal analgesia Best possible pain relief Lowest incidence of side
Objectives 9/7/2012. Optimizing Analgesia to Enhance the Recovery After Surgery CME FACULTY DISCLOSURE
 Optimizing Analgesia to Enhance the Recovery After Surgery Francesco Carli, M.D.. McGill University, Montreal, QC, Canada. ASPMN, Baltimore, 2012 CME FACULTY DISCLOSURE Francesco Carli has no affiliation
Optimizing Analgesia to Enhance the Recovery After Surgery Francesco Carli, M.D.. McGill University, Montreal, QC, Canada. ASPMN, Baltimore, 2012 CME FACULTY DISCLOSURE Francesco Carli has no affiliation
Digital RIC. Rhode Island College. Linda M. Green Rhode Island College
 Rhode Island College Digital Commons @ RIC Master's Theses, Dissertations, Graduate Research and Major Papers Overview Master's Theses, Dissertations, Graduate Research and Major Papers 1-1-2013 The Relationship
Rhode Island College Digital Commons @ RIC Master's Theses, Dissertations, Graduate Research and Major Papers Overview Master's Theses, Dissertations, Graduate Research and Major Papers 1-1-2013 The Relationship
Setting Department of Gynecology and Obstetrics, Cleveland Clinic Foundation (tertiary care academic centre), USA.
 Prospective randomized clinical trial of laparoscopically assisted vaginal hysterectomy versus total abdominal hysterectomy Falcone T, Paraiso M F, Mascha E Record Status This is a critical abstract of
Prospective randomized clinical trial of laparoscopically assisted vaginal hysterectomy versus total abdominal hysterectomy Falcone T, Paraiso M F, Mascha E Record Status This is a critical abstract of
Welcome Charles Kennedy
 Welcome Charles Kennedy Comoderators Girish P. Joshi, MBBS, MD, FFARCI Professor of Anesthesiology and Pain Management University of Texas Southwestern Medical Center Dallas, Texas David E. Beck, MD, FACS
Welcome Charles Kennedy Comoderators Girish P. Joshi, MBBS, MD, FFARCI Professor of Anesthesiology and Pain Management University of Texas Southwestern Medical Center Dallas, Texas David E. Beck, MD, FACS
Clinical Policy: Lofexidine (Lucemyra) Reference Number: ERX.NPA.88 Effective Date:
 Clinical Policy: (Lucemyra) Reference Number: ERX.NPA.88 Effective Date: 07.31.18 Last Review Date: 08.18 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
Clinical Policy: (Lucemyra) Reference Number: ERX.NPA.88 Effective Date: 07.31.18 Last Review Date: 08.18 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
ABSTRACT. KEY WORDS antibiotics; prophylaxis; hysterectomy
 Infectious Diseases in Obstetrics and Gynecology 8:230-234 (2000) (C) 2000 Wiley-Liss, Inc. Wound Infection in Gynecologic Surgery Aparna A. Kamat,* Leo Brancazio, and Mark Gibson Department of Obstetrics
Infectious Diseases in Obstetrics and Gynecology 8:230-234 (2000) (C) 2000 Wiley-Liss, Inc. Wound Infection in Gynecologic Surgery Aparna A. Kamat,* Leo Brancazio, and Mark Gibson Department of Obstetrics
Viberzi. Viberzi (eluxadoline) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subsection: Gastrointestinal Agents Original Policy Date: July 24, 2015 Subject: Viberzi Page: 1 of 5 Last
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subsection: Gastrointestinal Agents Original Policy Date: July 24, 2015 Subject: Viberzi Page: 1 of 5 Last
TIVORBEX Now Available in U.S. Pharmacies for the Treatment of Acute Pain
 TIVORBEX Now Available in U.S. Pharmacies for the Treatment of Acute Pain Second Low-Dose SoluMatrix NSAID from Iroko Now Available by Prescription PHILADELPHIA, June 29, 2015 Iroko Pharmaceuticals, LLC,
TIVORBEX Now Available in U.S. Pharmacies for the Treatment of Acute Pain Second Low-Dose SoluMatrix NSAID from Iroko Now Available by Prescription PHILADELPHIA, June 29, 2015 Iroko Pharmaceuticals, LLC,
Quality Outcomes and Financial Benefits of Nutrition Intervention. Tracy R. Smith, PhD, RD, LD Senior Clinical Manager, Abbott Nutrition
 Quality Outcomes and Financial Benefits of Nutrition Intervention Tracy R. Smith, PhD, RD, LD Senior Clinical Manager, Abbott Nutrition January 28, 2016 SHIFTING MARKET DYNAMICS PROVIDE AN OPPORTUNITY
Quality Outcomes and Financial Benefits of Nutrition Intervention Tracy R. Smith, PhD, RD, LD Senior Clinical Manager, Abbott Nutrition January 28, 2016 SHIFTING MARKET DYNAMICS PROVIDE AN OPPORTUNITY
Probuphine. (buprenorphine implant) New Product Slideshow
 Probuphine (buprenorphine implant) New Product Slideshow Introduction Brand name: Probuphine Generic name: Buprenorphine Pharmacological class: Opioid (partial agonist-antagonist) Strength and Formulation:
Probuphine (buprenorphine implant) New Product Slideshow Introduction Brand name: Probuphine Generic name: Buprenorphine Pharmacological class: Opioid (partial agonist-antagonist) Strength and Formulation:
HOPE. Considerations. Considerations ISING. Safe Opioid Prescribing Guidelines for ACUTE Non-Malignant Pain
 Due to the high level of prescription drug use and abuse in Lake County, these guidelines have been developed to standardize prescribing habits and limit risk of unintended harm when prescribing opioid
Due to the high level of prescription drug use and abuse in Lake County, these guidelines have been developed to standardize prescribing habits and limit risk of unintended harm when prescribing opioid
Alvimopan in an Enhanced Recovery Program Following Radical Cystectomy
 Bladder Cancer 1 (2015) 137 142 DOI 10.3233/BLC-150017 IOS Press Research Report 137 Alvimopan in an Enhanced Recovery Program Following Radical Cystectomy Zach Hamilton a, Will Parker a, Josh Griffin
Bladder Cancer 1 (2015) 137 142 DOI 10.3233/BLC-150017 IOS Press Research Report 137 Alvimopan in an Enhanced Recovery Program Following Radical Cystectomy Zach Hamilton a, Will Parker a, Josh Griffin
Regulatory Status FDA-approved indications: Emend is a substance P/neurokinin 1 (NK1) receptor antagonist, indicated: (1-2)
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.50.05 Section: Prescription Drugs Effective Date: April 1, 2017 Subject: Emend Page: 1 of 6 Last Review
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.50.05 Section: Prescription Drugs Effective Date: April 1, 2017 Subject: Emend Page: 1 of 6 Last Review
Labeled Uses: Treatment of Clostiridum Difficile associated diarrhea (CDAD)
 Brand Name: Dificid Generic Name: fidaxomicin Manufacturer 1,2,3,4,5 : Optimer Pharmaceuticals, Inc. Drug Class 1,2,3,4,5 : Macrolide Antibiotic Uses 1,2,3,4,5 : Labeled Uses: Treatment of Clostiridum
Brand Name: Dificid Generic Name: fidaxomicin Manufacturer 1,2,3,4,5 : Optimer Pharmaceuticals, Inc. Drug Class 1,2,3,4,5 : Macrolide Antibiotic Uses 1,2,3,4,5 : Labeled Uses: Treatment of Clostiridum
Viberzi. Viberzi (eluxadoline) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subsection: Gastrointestinal Agents Original Policy Date: July 24, 2015 Subject: Viberzi Page: 1 of 5 Last
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subsection: Gastrointestinal Agents Original Policy Date: July 24, 2015 Subject: Viberzi Page: 1 of 5 Last
STARTING PATIENTS ON. AA-CLOZAPINE is the only Canadian-made treatment indicated for the symptoms of treatment-resistant schizophrenia.
 STARTING PATIENTS ON AA-CLOZAPINE is the only Canadian-made treatment indicated for the symptoms of treatment-resistant schizophrenia. Indication: AA-CLOZAPINE (clozapine) is indicated in the management
STARTING PATIENTS ON AA-CLOZAPINE is the only Canadian-made treatment indicated for the symptoms of treatment-resistant schizophrenia. Indication: AA-CLOZAPINE (clozapine) is indicated in the management
TAP blocks vs wound infiltration in laparoscopic colectomies Results of a Randomised Controlled Clinical Trial
 TAP blocks vs wound infiltration in laparoscopic colectomies Results of a Randomised Controlled Clinical Trial Kim Gorissen Frederic Ris Martijn Gosselink Ian Lindsey Dept of Colorectal Surgery Dept of
TAP blocks vs wound infiltration in laparoscopic colectomies Results of a Randomised Controlled Clinical Trial Kim Gorissen Frederic Ris Martijn Gosselink Ian Lindsey Dept of Colorectal Surgery Dept of
Technivie. Technivie (ombitasvir, paritaprevir, ritonavir) and Ribavirin. Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.01.37 Subject: Technivie Page: 1 of 6 Last Review Date: December 8, 2017 Technivie Description Technivie
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.01.37 Subject: Technivie Page: 1 of 6 Last Review Date: December 8, 2017 Technivie Description Technivie
Surgery while taking Protonix? 1,368 conversations on the web about experiences with taking Protonix before or after having a Surgery.
 Surgery while taking Protonix? 1,368 conversations on the web about experiences with taking Protonix before or after having a Surgery. Everything you ever wanted to know about gastric sleeve surgery. What
Surgery while taking Protonix? 1,368 conversations on the web about experiences with taking Protonix before or after having a Surgery. Everything you ever wanted to know about gastric sleeve surgery. What
Complete the Qsymia Healthcare Provider Training Program in 2 easy steps:
 Overview FDA has required a Risk Evaluation and Mitigation Strategy (REMS) for Qsymia so that healthcare providers can be informed about the increased risk of teratogenicity associated with Qsymia therapy.
Overview FDA has required a Risk Evaluation and Mitigation Strategy (REMS) for Qsymia so that healthcare providers can be informed about the increased risk of teratogenicity associated with Qsymia therapy.
Nutritional Support in the Perioperative Period
 Nutritional Support in the Perioperative Period Topic 17 Module 17.3 Nutritional Support in the Perioperative Period Ken Fearon Learning Objectives Understand the principles behind nutritional care for
Nutritional Support in the Perioperative Period Topic 17 Module 17.3 Nutritional Support in the Perioperative Period Ken Fearon Learning Objectives Understand the principles behind nutritional care for
Long-Term Care Updates
 Long-Term Care Updates April 2017 Bezlotoxumab to Prevent Recurrent Infection By Amy Wilson, PharmD and Zara Risoldi Cochrane, PharmD, MS, FASCP Introduction The Gram-positive bacteria is a common cause
Long-Term Care Updates April 2017 Bezlotoxumab to Prevent Recurrent Infection By Amy Wilson, PharmD and Zara Risoldi Cochrane, PharmD, MS, FASCP Introduction The Gram-positive bacteria is a common cause
Original article Postoperative ileus in colorectal surgery: is there any difference between laparoscopic and open surgery?
 Gastroenterology Report 1 (2013) 138 143, doi:10.1093/gastro/got008 Advance access publication 4 April 2013 Original article Postoperative ileus in colorectal surgery: is there any difference between laparoscopic
Gastroenterology Report 1 (2013) 138 143, doi:10.1093/gastro/got008 Advance access publication 4 April 2013 Original article Postoperative ileus in colorectal surgery: is there any difference between laparoscopic
Annex III. Amendments to relevant sections of the Product Information
 Annex III Amendments to relevant sections of the Product Information Note: These amendments to the relevant sections of the product information are the outcome of the referral procedure. The product information
Annex III Amendments to relevant sections of the Product Information Note: These amendments to the relevant sections of the product information are the outcome of the referral procedure. The product information
Outcomes of Patients with Preoperative Weight Loss following Colorectal Surgery
 Outcomes of Patients with Preoperative Weight Loss following Colorectal Surgery Zhobin Moghadamyeghaneh MD 1, Michael J. Stamos MD 1 1 Department of Surgery, University of California, Irvine Nothing to
Outcomes of Patients with Preoperative Weight Loss following Colorectal Surgery Zhobin Moghadamyeghaneh MD 1, Michael J. Stamos MD 1 1 Department of Surgery, University of California, Irvine Nothing to
EFFECT OF AN ENHANCED RECOVERY AFTER SURGERY PROGRAM ON OPIOID USE AND PATIENT-REPORTED OUTCOMES
 EFFECT OF AN ENHANCED RECOVERY AFTER SURGERY PROGRAM ON OPIOID USE AND PATIENT-REPORTED OUTCOMES Obstetrics & Gynecology Vol. 132, No. 2, August 2018 KUSMW OBGYN Journal Club Thomas Greaves, MD, PGY4 August
EFFECT OF AN ENHANCED RECOVERY AFTER SURGERY PROGRAM ON OPIOID USE AND PATIENT-REPORTED OUTCOMES Obstetrics & Gynecology Vol. 132, No. 2, August 2018 KUSMW OBGYN Journal Club Thomas Greaves, MD, PGY4 August
7/31/2015. Enhanced Recovery After Surgery: Change Your Mind, Change Your Practice. Objectives. Enhanced Recovery Society
 Enhanced Recovery After Surgery: Change Your Mind, Change Your Practice Margaret Odhner MS, ANP-BC, COCN Kim Meacham, MSN FNP-C, CWON Objectives 1. Describe the Enhanced Recover After Surgery (ERAS) pathway.
Enhanced Recovery After Surgery: Change Your Mind, Change Your Practice Margaret Odhner MS, ANP-BC, COCN Kim Meacham, MSN FNP-C, CWON Objectives 1. Describe the Enhanced Recover After Surgery (ERAS) pathway.
Postoperative Ultrasound Evaluation of Gastric Distension; A Pilot study
 Postoperative Ultrasound Evaluation of Gastric Distension; A Pilot study M Jaronczyk MD, W Boyan Jr. MD, M Goldfarb MD. FACS. MMC Dept of Surgery Nausea and vomiting are common complaints of postoperative
Postoperative Ultrasound Evaluation of Gastric Distension; A Pilot study M Jaronczyk MD, W Boyan Jr. MD, M Goldfarb MD. FACS. MMC Dept of Surgery Nausea and vomiting are common complaints of postoperative
4/18/2018. Disclosure. Learning Objectives. St. Vincent Healthcare. Supportive Literature. Background
 Implementation of a risk-score-dependent antiemetic protocol to reduce post-operative nausea and vomiting (PONV) in inpatient surgical patients Eugene P. Eldridge, PharmD, RPh PGY-1 Pharmacy Resident St.
Implementation of a risk-score-dependent antiemetic protocol to reduce post-operative nausea and vomiting (PONV) in inpatient surgical patients Eugene P. Eldridge, PharmD, RPh PGY-1 Pharmacy Resident St.
PACKAGE LEAFLET: Information for the user. METFORMINE Film-coated tablets 500 mg, 850 mg or 1000 mg (Metformin hydrochloride)
 PACKAGE LEAFLET: Information for the user METFORMINE Film-coated tablets 500 mg, 850 mg or 1000 mg (Metformin hydrochloride) Read this leaflet carefully before you start taking this medicine. - Keep this
PACKAGE LEAFLET: Information for the user METFORMINE Film-coated tablets 500 mg, 850 mg or 1000 mg (Metformin hydrochloride) Read this leaflet carefully before you start taking this medicine. - Keep this
VELTASSA (patiromer) oral suspension
 VELTASSA (patiromer) oral suspension Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Pharmacy
VELTASSA (patiromer) oral suspension Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Pharmacy
Let s Talk about Nutrition: Communication Skills for Health Care Professionals Topic 39
 Let s Talk about Nutrition: Communication Skills for Health Care Professionals Topic 39 Module 39.2. Nutrition Facts: Cost-effectiveness of Nutrition Support Prof. Maurizio Muscaritoli, MD Full Professor
Let s Talk about Nutrition: Communication Skills for Health Care Professionals Topic 39 Module 39.2. Nutrition Facts: Cost-effectiveness of Nutrition Support Prof. Maurizio Muscaritoli, MD Full Professor
Iroko Pharmaceuticals Gains Additional Patents for ZORVOLEX and TIVORBEX TM
 Iroko Pharmaceuticals Gains Additional Patents for ZORVOLEX and TIVORBEX TM PHILADELPHIA, April 8, 2015 Iroko Pharmaceuticals, LLC, a global specialty pharmaceutical company dedicated to advancing the
Iroko Pharmaceuticals Gains Additional Patents for ZORVOLEX and TIVORBEX TM PHILADELPHIA, April 8, 2015 Iroko Pharmaceuticals, LLC, a global specialty pharmaceutical company dedicated to advancing the
Cigna Drug and Biologic Coverage Policy
 Cigna Drug and Biologic Coverage Policy Subject Antiemetic Therapy Table of Contents Coverage Policy... 1 General Background... 6 Coding/Billing Information... 8 References... 8 Effective Date... 1/1/2018
Cigna Drug and Biologic Coverage Policy Subject Antiemetic Therapy Table of Contents Coverage Policy... 1 General Background... 6 Coding/Billing Information... 8 References... 8 Effective Date... 1/1/2018
PFIZER INC. THERAPEUTIC AREA AND FDA APPROVED INDICATIONS: See United States Package Insert (USPI)
 PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
Influenza Outbreaks. An Overview for Pharmacists Prescribing Antiviral Medications
 Influenza Outbreaks An Overview for Pharmacists Prescribing Antiviral Medications Under the Collaborative Drug Therapy Agreement for Influenza Antiviral Medications Learning Objectives 1. Understand the
Influenza Outbreaks An Overview for Pharmacists Prescribing Antiviral Medications Under the Collaborative Drug Therapy Agreement for Influenza Antiviral Medications Learning Objectives 1. Understand the
Immodium / loprarmide
 Immodium / loprarmide IMODIUM (loperamide hydrochloride) is indicated for the control and symptomatic relief of acute nonspecific diarrhea and of chronic diarrhea associated with inflammatory bowel disease.
Immodium / loprarmide IMODIUM (loperamide hydrochloride) is indicated for the control and symptomatic relief of acute nonspecific diarrhea and of chronic diarrhea associated with inflammatory bowel disease.
Study population The study population comprised patients who had undergone major abdominal surgery in routine care.
 Evaluation of costs and effects of epidural analgesia and patient-controlled intravenous analgesia after major abdominal surgery. Bartha E, Carlsson P, Kalman S Record Status This is a critical abstract
Evaluation of costs and effects of epidural analgesia and patient-controlled intravenous analgesia after major abdominal surgery. Bartha E, Carlsson P, Kalman S Record Status This is a critical abstract
Motility Disorders. Pelvic Floor. Colorectal Center for Functional Bowel Disorders (N = 701) January 2010 November 2011
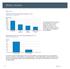 Motility Disorders Pelvic Floor Colorectal Center for Functional Bowel Disorders (N = 71) January 21 November 211 New Patients 35 3 25 2 15 1 5 Constipation Fecal Incontinence Rectal Prolapse Digestive-Genital
Motility Disorders Pelvic Floor Colorectal Center for Functional Bowel Disorders (N = 71) January 21 November 211 New Patients 35 3 25 2 15 1 5 Constipation Fecal Incontinence Rectal Prolapse Digestive-Genital
Improving documentation and coding of malnutrition a five year journey
 Improving documentation and coding of malnutrition a five year journey Natalie Simmance, Chief Dietitian Clara Newsome Sally Bell Sonia Grundy St Vincent's Hospital Melbourne Patient Malnutrition common
Improving documentation and coding of malnutrition a five year journey Natalie Simmance, Chief Dietitian Clara Newsome Sally Bell Sonia Grundy St Vincent's Hospital Melbourne Patient Malnutrition common
Postoperative ileus is a temporary
 clinical review Postoperative ileus is a temporary impairment in gastrointestinal (GI) motility. Though considered an inevitable complication of abdominal and pelvic surgeries, postoperative ileus may
clinical review Postoperative ileus is a temporary impairment in gastrointestinal (GI) motility. Though considered an inevitable complication of abdominal and pelvic surgeries, postoperative ileus may
LOKELMA (sodium zirconium cyclosilicate) oral suspension
 LOKELMA (sodium zirconium cyclosilicate) oral suspension Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit
LOKELMA (sodium zirconium cyclosilicate) oral suspension Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit
Balanced Analgesia With NSAIDS and Coxibs. Raymond S. Sinatra MD, Ph.D
 Balanced Analgesia With NSAIDS and Coxibs Raymond S. Sinatra MD, Ph.D Prostaglandins and Pain The primary noxious mediator released from damaged tissue is prostaglandin (PG) PG is responsible for nociceptor
Balanced Analgesia With NSAIDS and Coxibs Raymond S. Sinatra MD, Ph.D Prostaglandins and Pain The primary noxious mediator released from damaged tissue is prostaglandin (PG) PG is responsible for nociceptor
PFIZER INC. THERAPEUTIC AREA AND FDA APPROVED INDICATIONS: See USPI.
 PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
Clinical Trial Results Database Page 1
 Clinical Trial Results Database Page 1 Sponsor Novartis Pharmaceuticals Corporation Generic Drug Name Therapeutic Area of Trial Major Depressive Disorder (MDD) Approved Indication Treatment of major depressive
Clinical Trial Results Database Page 1 Sponsor Novartis Pharmaceuticals Corporation Generic Drug Name Therapeutic Area of Trial Major Depressive Disorder (MDD) Approved Indication Treatment of major depressive
Lokelma (sodium zirconium cyclosilicate), Veltassa (patiromer)
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.30.42 Subject: Potassium Binders Page: 1 of 5 Last Review Date: November 30, 2018 Potassium Binders Description
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.30.42 Subject: Potassium Binders Page: 1 of 5 Last Review Date: November 30, 2018 Potassium Binders Description
Laparoscopic (keyhole) colorectal (bowel) resection
 Laparoscopic (keyhole) colorectal (bowel) resection Your operation explained Information for patients Colorectal Surgery Introduction This leaflet should be read together with a booklet which explains
Laparoscopic (keyhole) colorectal (bowel) resection Your operation explained Information for patients Colorectal Surgery Introduction This leaflet should be read together with a booklet which explains
Glucophage XR is contra-indicated during breast-feeding.
 Name GLUCOPHAGE XR 1000 mg Prolonged release tablets Active ingredient Metformin hydrochloride Composition Each Glucophage XR 1000 mg prolonged release tablet contains as active ingredient 1000 mg metformin
Name GLUCOPHAGE XR 1000 mg Prolonged release tablets Active ingredient Metformin hydrochloride Composition Each Glucophage XR 1000 mg prolonged release tablet contains as active ingredient 1000 mg metformin
Impavido. (miltefosine) New Product Slideshow
 Impavido (miltefosine) New Product Slideshow Introduction Brand name: Impavido Generic name: Miltefosine Pharmacological class: Antileishmanial agent Strength and Formulation: 50mg; hard gel capsules Manufacturer:
Impavido (miltefosine) New Product Slideshow Introduction Brand name: Impavido Generic name: Miltefosine Pharmacological class: Antileishmanial agent Strength and Formulation: 50mg; hard gel capsules Manufacturer:
Nutritional Support in the Perioperative Period
 Nutritional Support in the Perioperative Period Topic 17 Module 17.6 Facilitating Oral or Enteral Nutrition in the Postoperative Period Mattias Soop Learning Objectives To review the causes of postoperative
Nutritional Support in the Perioperative Period Topic 17 Module 17.6 Facilitating Oral or Enteral Nutrition in the Postoperative Period Mattias Soop Learning Objectives To review the causes of postoperative
