5/19/2017. State of Total Disc Replacement: Past, Present, and Future. Disclosures. Introduction. Richard D. Guyer, M.D.
|
|
|
- Juniper Horn
- 6 years ago
- Views:
Transcription
1 State of Total Disc Replacement: Past, Present, and Future Richard D. Guyer, M.D. Disclosures Guyer (a) Alphatec; (b) Spinal Kinetics, Spinal Ventures, Mimedix; (c) DePuy Synthes Spine, K2M, Flexuspine, Mimedix, Aescalup; Safe Orthopedics (d) DePuy Synthes Spine, K2M, Paradigm Spine Key: (a) royalties; (b) stock/options; (c) consulting/sab; (d) Speaker/ faculty; (e) Research; (f) Fellowship and related research; (g) other Introduction Lumbar and cervical TDR Have been the most studied spinal conditions with more than a dozen IDEs Long history in US dating to 2000 Strong outcomes Lower than expected adoption for Lumbar but excellent for Cervical Promising future 1
2 TDR Market Overview Global market for non-fusion treatments to expand from $521M in 2014 to $749M in 2021 Growth driven by cervical TDR due to: Increasing availability of clinical efficacy data Improving reimbursement environment Rising prevalence of degenerative spine diseases Industry s aggressive efforts in physician training and DTC marketing Market underpenetrated at only 20% of procedures globally, less in the US despite regulatory clearances of devices Global Data Healthcare. Dec Spinal Non Fusion, Global Analysis and Market Forecasts; Markets and Markets report. The Spine Group/LinkedIn TDR Market Overview Europe has been dominant market for C-TDR until ~2013 due to less stringent regulatory approval process US is now, and will remain, dominant market for C-TDR through 2020 with increasing number of approved devices, improved reimbursement 1. MedMarket Diligence. The Spine Market Group/LinkedIn Looking first at Lumbar TDR: Lumbar TDR has been used OUS since 1980s We know a lot about the outcomes Multiple studies, from multiple countries show good outcomes at 2, 5, >10 yr follow-up Multiple FDA-regulated trials found TDR to be at least as effective as fusion and superior on some measures 2
3 US IDE Studies (Not a Comprehensive List) Charité approved 2004, removed from US market 9/2013 by DePuy due to poor sales and purchase of Synthes (ProDisc), not disasters!!! ProDisc-L approved 2006 activl approved 6/2015 Maverick completed, patent issues FlexiCore withdrawn by Stryker Kineflex-L withdrawn by Spinal Motion Freedom 3 rd gen, polymer, not submitted L TDR lateral insertion, aborted Triumph posterior insertion aborted TDR Design Types Ball and Socket Semiconstrained Unconstrained Viscoelastic Elastomeric polymer FDA Trial Data Published Charite vs. ALIF - 5 yr data ProDisc-L vs yr data Maverick vs. ALIF - 5 yr data Flexicore vs yr data Kineflex-L vs. Charité - 5 yr data activl vs. Charité or ProDisc-L - 2 yr data 5 yr coming very soon 3
4 What Is Needed for Widespread Use of an Implant? Must establish: Safety Efficacy Defined indications Address cost Equally Important Reimbursement Surgeon perception Safety Multiple studies found a significantly lower re-op rate for TDR vs. fusion Others found rate lower or similar for TDR vs. fusion No randomized studies found a higher re-op rate for TDR TBI Re operation Experience Consecutive series beginning with 1 st TDR case in 2000 from IDE studies Included all TDR pts at least 2 yrs (longest 134 months) post-op and all pts who were fusion controls in randomized FDA IDE TDR trials 1,058 TDR 112 hybrid 67 fusion ISSLS,
5 Re op Rates by Surgery Type 25% 20% 15% 20.9% 10% 10.4% 11.6% 5% 0% TDR Hybrid Fusion TDR significantly lower than fusion (p<0.02); trend for hybrid< fusion (0.05<p<0.08) TBI, ISSLS, 2013 TDR: Effectiveness Lumbar FDA IDE trials: 4 with 5 yr follow-up 2 additional with 2 yr follow-up Multiple studies from Europe with >10 yr follow-up Multiple studies from Asia with 2-5 yr follow-up TDR: Effectiveness All these studies have consistently reported maintained good outcomes throughout longest term follow-up FDA IDE trials found significant improvement by 6 wk post-op and maintained through 2 and 5 year follow-up with NO DEGRADATION Following are new additional lumbar TDR studies 5
6 DDD group (n=39): Axial LBP from DDD DDD+other group (n=15): DDD + >1 of the following: previously fused adjacent level, spondy, retrolisthesis, facet joint arthritis, lateral recess stenosis Mean follow-up mo Mean 10 yr Clinical Follow up Significantly better outcomes in DDD vs. DDD+other dx group: Success: 76.9% vs. 40.0% Satisfaction: 87.2% vs. 60.0% 5 pts (9.3%) had revision fusion surgeries, all were DDD+other group Segmental ROM was well maintained in DDD group, but not in the other group Park et al, Spine, pts receiving M6 lumbar TDR (49 1-level, 43 multi-level) Prospective, post-market, multicenter registry study Significant improvement in VAS and ODI scores (p<0.05; >50% improvement) No device revisions or removals (no comment on other re-ops) 6
7 Data from 48 pts with viscoelastic TDR (Freedom Disc) from SWISSspine registry Compared with 131 ALIF pts from Spine Tango registry Covariates included in the models: age, gender, number of levels treated, level(s) treated, disc herniation as additional dx, pre-op back and leg pain scores, and follow-up interval TDR associated with significantly greater back and leg pain relief (p<0.01) compared with ALIF What s New for Lumbar TDR and Updates Orthopedics, 2016 Used CT and finite element modeling to evaluate interaction of pt anatomy, implant geometry, and implant positioning with goal of maximizing ROM and outcomes Device positioning Pre op ht Optimal ht 7
8 activl Preliminary 5 Year Outcome Data ODI Back Pain VAS Mean ODI Score activl Artificial Disc ProDisc L Charité Mean VAS Score activl Artificial Disc ProDisc L Charité Years Years Preliminary Minimum 10 Year Data for ProDisc L vs. 360 Fusion FDA Study at TBI 10 year VAS 10 year ODI TDR (n=49) Fusion (n=5) TDR (n=49) Fusion (n=5) Pre op 6 wk 24 mo 10 yr 0 Pre op 6wk 24 mo 10 yr TDR had statistically significantly greater improvement in back pain, ODI scores and pt satisfaction Conclusions: differences in clinical improvement were not beyond generally accepted boundaries for clinical relevance 8
9 Meta analysis of RCTs Purpose: Compare effectiveness and safety of TDR vs. fusion Searched Medline, Embase, Clinical, Ovid, BIOSIS and Cochrane registry of controlled clinical trials 7 relevant RCTs with total of 1,584 pts, 2 yr follow-up TDR more effective in ODI, VAS, hospital stay duration, greater willingness to choose the same operation again Differences not significant for op time, blood loss, complications, re-op rate or RTW rates Rao, Arch Orthop Trauma Surg, 2014 Other Clinical Outcome One of the major potential clinical benefits of lumbar TDR is reducing the occurrence of adjacent segment degeneration Those of us involved in TDR from the beginning have been searching for this evidence as it is what has motivated our research But ASD is like old Natural history vs Increased stress of fusion or Genetics vs Environment arguments ASD: ProDisc TDR vs. Fusion The Holy Grail of lessened ASD finally!!! Δ ASD observed at 5 years in: 28.6% of fusion patients 9.2% of TDR patients > 3 : 1 Difference P<0.01 Zigler et al, J Neurosurg Spine,
10 ASD and ROM of TDR Level: 5 yr Follow up from Activ L 12% % of patients with ASD 10% 8% 6% 4% 2% 0% >0 o >1 o >2 o >3 o >4 o >5 o >6 o >7 o >8 o ROM at TDR level For each additional degree of motion at the TDR level, there was a decline in the rate of ASD (data from activl FDA trial) Guyer, ISASS, 2017 ASD and Age at 5 yr Follow up % of patients with ASD 20% 15% 10% 5% 0% 5.0% 19.6% <40 yr old >40 yrs old ASD significantly greater in older patient subset, p<0.01 (data from activl FDA trial data) Guyer, ISASS, 2017 Adjacent Segment Reviewed 27 Class I and II fusion and TDR studies 314/926 fusion patients developed AgDD = 31% 31/313 TDR patients developed AgDD = 3% p<
11 What About Cost? Assumption: New technology is always more expensive But, look at the data! Lumbar TDR Costs Studies Several studies compared TDR to fusion Methods included economic modeling (Guyer et al, 2007), randomly selected patients and averaged database charges/costs (Patel et al, 2008), IDE trial patients (Leven et al. 2007), and national data registries with unmatched comparison groups (Kurtz et al, 2010) Lumbar TDR Costs Studies Although the methods used in the studies varied greatly they all found TDR was less expensive than fusion with the one exception of a cost model for ALIF with autograft only which is rarely used today Cost advantage vs fusion with various implants, various approaches, various graft materials with little uniformity from surgeon to surgeon 11
12 Cervical TDR Best data, so briefly FDA Approved in US Approved Prestige ST (2007) ProDisc C (2007) Bryan (2009) PCM (2012) SeCure C (2012) Mobi C (1 and 2 level; 2013) Prestige LP (1 level 2014, 2 level 2016) Current FDA Status in US Trial completed / withdrawn Kineflex C Trials ongoing M6 Simplify (1 and 2 level studies) Trials ended Discover Neodisc Cervicore 12
13 FDA IDE Trials All trials found TDR to be non-inferior to ACF Superior on some measures Good results maintained for >5 yrs in studies with long-term follow-up Mean NDI Scores for TDR and ACF in Various FDA IDE Trials showing similar results Prestige TDR Bryan TDR ProDisc C TDR Kineflex C TDR Mobi C TDR Prestige ACF Bryan ACF ProDisc C ACF Kineflex C ACF Mobi C ACF 0 Pre op 6 wk 3 mo 6 mo 12 mo 24 mo What Happens 7 10 Years Post TDR? 13
14 Int J Spine Surg, pts, 20 sites, 1 level TDR 265 historical control ACDF pts From original Prestige study, same indications and assessments TDR maintained significantly improved clinical outcomes and segmental motion TDR at least non-inferior to ADF Prestige LP: 7 yr Follow up TDR ACDF Follow up rate 75.9% 70.0% Overall success 74.9% 63.2% NDI success 86.1% 80.1% % satisfied 90.9% 85.6% Re op (index level) 6.4% 10.9% Gornet, IJSS 2016 Mobi C: 7 yr Follow up TDR ACDF Follow up rate 80.1% 73.5% Overall success 55.2% 50.0% NDI success 76.5% 80.8% % satisfied 90.9% 80.8% ASD inferior level 43.8% 63.0% ASD superior level 40.4% 66.7% Re op 3.0% 12.3% Hisey et al, NASS,
15 10 yr follow-up of 1 center s FDA IDE pts 19 TDR, 23 ACDF (follow-up rates 84.6% and 92.0%) Re-op rates: TDR 9% ACDF 32% TDR ROM In addition to pain reduction, a primary goal of TDR is to allow motion at the operated segment Does TDR achieve this goal? Range of Motion Maintained: Data from FDA IDE Trials Device Pre op 2 yr 4 5 yr Bryan 6.5 o 8.1 o 8.5 o Prestige 7.5 o 7.6 o NA ProDisc C 8.5 o 9.4 o 8.1 o Kineflex C 8.2 o 9.8 o NA Secure C 8.5 o 10.2 o NA PCM 7.9 o 5.7 o NA Mobi C 8.2 o 10.6 o NA Sasso et al, JBJS 2011; Mummaneni et al, J Neurosurg Spine, 2007; Zigler et al, Spine, 2013; Secure C FDA SSED; Coric et al, J Neurosurg Spine, 2011; Hisey, ISASS 2011; PCM FDA SSED 15
16 Adjacent Segment Degeneration One potential benefit of cervical TDR vs. ACF is reducing the acceleration of ASD Literature mixed on whether this benefit has been recognized Kineflex C Adjacent Segment Disc Degeneration 100% 80% 60% 40% 20% Severe Moderate Mild None 0% TDR ACF TDR ACF Pre-op 24 mo post-op Pre-op: no difference; Post-op: significant difference (p<0.01), indicating more patients in ACF group had greater grades of adjacent segment degeneration Coric et al, J Neurosurg Spine, 2011 Adjacent Level Subsequent Surgery Rates through 60 Months 12% 10% 11.1% 11.4% 8% 6% 4% 2% 2.2% 3.4% 0% CDA ACF 1 Level 2 Level 1 Level 2 Level Mobi C FDA IDE trial data 16
17 Imaging One potential disadvantage of TDRs with metal components is artifact seen on MRI MRI technology continually improving to reduce artifact Materials used for TDRs is also changing that will facilitate imaging Simplify Disc Currently enrolling 1 and 2 level IDE study PEEK superior endplate Ceramic core PEEK inferior endplate Titanium plasma spray (porous) Semi-constrained design Post TDR Artifact on MRI PEEK on Ceramic Metal on metal 17
18 Simplify discs lower heights match cervical anatomy 180 Average Disc Height From Previous IDE Study (N=525) 160 Simplify Disc Coverage Most cervical discs designed for these heights 0 <= to to to to to to 6.0 > 6.0 Elastomeric Discs Motion occurs within polymer, Provides shock absorption, Mimics properties of natural disc (Axis of Rotation, etc) Elastomeric Disc All Elastomer Metal Backed CADisc Mobile Bumper Attached Core Polymer-Bone abrasion Addition of HA to enhance Nucleus Replacement interface problems Prosthetic-endplate Bryan M6 C ESP K2M Rhine Freedom Free motion between metal and polymer Core free to move Polymer-Metal Sliding Wear Need for Surrounding Envelope More complex Viscoelastic component adhered to titanium endplates, one-piece deformable but cohesive interbody spacer 6 degrees of freedom about 3 axes including shock absorption Allows limited rotation and translation with resistance to motion (elastic return property) Rotation center can vary freely during motion 18
19 CP ESP Gaining wide use OUS Viscoelastic polyurethane Recently approved in Australia No study data US cervical IDE never initiated and lumbar IDE never completed Freedom Disc Rhine Disc K2M CE Mark Jan st implant Jan, 2016 Germany, Belgium, Spin, Italy, UK, South Africa 300 implanted so far 60% one level 40% 2 level 19
20 Rhine Disc K2M RHINE TDA C5 C6 ROM Regardless of Anterior or Lateral Positioning ROM not affected Advantage of Viscoelastic C5-C6 Segmental ROM (deg) 1.5Nm Flexion Nm Extension Intact Anterior placement at C5-C6 Optimal placement at C5-C6 Optimal placement at C6-C7 Lateral placement at C5-C6 C5-C6 ROM N Follower Load 150N Follower Load M6 30 pts, TDR at 1 or 2 levels Significant improvements throughout 24 mo in: NDI: 67.8 to 20.8 VAS neck pain: 7.7 to 2.4; right arm pain: 6.0 to 2.1; left 6.3 to
21 M6 FDA IDE 12mo, 2 Centers, 39 patients Mean VAS Neck Pain Pre op 6 wk 3 mo 6 mo 12 mo Mean NDI Scores Pre op 6 wk 3 mo 6 mo 12 mo Mean VAS Arm Pain Pre op 6 wk 3 mo 6 mo 12 mo Mean VAS neck, VAS arm pain, and NDI scores improved significantly by 6 wk postop and remained improved through 12 mo follow up Summary Strong data to support lumbar and cervical TDR Appropriate patient selection is key Indications continue to be refined What we are not seeing: publications, even case reports, reporting high numbers of failed TDRs, catastrophic failures, deaths from revisions, etc. Ongoing development of surface technology, materials, viscoelastic devices Patient specific design like TKA? Summary Currently, greatest barrier to TDR adoption is insurance reimbursement Is slowly improving Cervical better than lumbar with 85% coverage vs 45% coverage For lumbar, some surgeons may be slow to adopt due to need for access surgeon and/or not familiar with anterior approach Also, the lumbar disc companies are not likely to jeopardize their much larger and profitable fusion business with the exception of Aesculap 21
22 Do Lumbar and Cervical TDR Have a Future? The most studied devices in the history of spine with Level 1 IDE data Strong data for lumbar and cervical TDR from variety of countries and study formats yr follow up Similar or superior to fusion with less ASD in longer term f/u Favorable cost profile No rash of reports on device failures Thank You Center of rotation (COR) with simplify disc Lateral Bending Pre-Op Lateral Bending Post-Op (6 Mos) 22
23 Viscoelastic Discs Company Cervical Devices Lumbar Devices Comments Freedom Cervical Freedom Lumbar Features viscoelastic polymer core to mimic normal human disc characteristics AxioMed Spinal Kinetics M6 C M6 L Features viscoelastic polymer core to simulate native disc and UHMWPE fibers to emulate native annulus K2M Physio L Acquired Physio L PCU disc technology from NexGen Spine Confidential Appropriately Selected Patients LBP +/- leg pain of lesser severity Failed >6 mo non-op care (PT, meds, education, activity modification, etc.) MRI and disco correlative with symptoms and physical exam DDD at 1-2 levels No other spine pathology No significant psychological problems Good bone quality Adjacent Segment Disease, Natural History or Biomechanical?? Literature Review Biomechanically fusions increase intradiscal pressure, facet loading, and hypermobility above and beyond natural history Radiographic 5.2%- 100% at mo f/u Symptomatic % at mo f/u (higher with pedicle screws, %) Risk factors = pedicle screws, facet joint violation, sagittal malalignment, and fusion length Park et al., Spine
24 Mean NDI Scores for TDR and ACF in Various FDA IDE Trials As seen in the graph, data is extremely reproducible across multiple devices All represent multicenter trials Most conducted at different centers Supports generalizability of outcomes Charité Year Follow up Old evaluation methods, but excellent follow up of >90% with 90% exe-gd CP ESP Titanium endplates and hydroxyapatite coating Inner and outer core (polycarbonate urethane) 6 degrees of freedom restores natural mobility 24
25 Inferior and Superior ASD at 60 mos in 2 level Study (Kellgren Lawrence Scale) 80% 70% Mobi-C ACDF % of Randomized Patients 60% 50% 40% 30% 20% 10% 0% Inferior Adj. Superior Adj. Level Level 24M Inferior Adj. Superior Adj. Inferior Adj. Superior Adj. Inferior Adj. Superior Adj. Level Level Level Level Level Level 36M 48M 60M Bryan: 10 yr Follow up Mean NDI Scores TDR ACDF Pre op Post op Mean VAS Scores TDR ACDF 0 pre op Post op pre op Post op Neck pain Arm pain Sasso et al, Spine, 2017 Quality of Motion Study Experimental protocol: Intact C5-C6 TDA Anterior placement C5-C6 TDA Optimal placement C6-C7 TDA Optimal placement C5-C6 TDA Lateral placement Cervical spine set-up showing intact specimen. 25
26 Elastomeric Designs The human disc provides Shock absorption Stability Controlled motion Ball & Socket Design Elastomeric Polymers Motion occurs within polymer Provides shock absorption Mimics properties of natural disc Attractive & intuitive solution to surgeons Elastomeric Design Inherently UNSTABLE Inherently STABLE From Other Countries Often relatively small numbers of pts Good results for cervical TDR, no indication of less favorable outcomes compared with ACF Europ Spine J 22(8): , cases; 64% follow-up at 5 yrs Significantly improved scores for: neck pain, arm pain, EQ5-D Significantly reduced analgesic use 4.5% revision surgery 26
27 Meta analysis: TDR vs. ACF No significant differences in NDI, SF-36, or pain scores TDR had significantly: Lower re-op rate Greater neurological success rate Lower re-op rate for adjacent-level when analyzed using fixed effects model, but not significant using random effects model Upadhyaya et al, J Neurosurg Spine, 2012 What about the Cost of Cervical TDR? Cost Used outcome literature, NIS, and Medicare reimbursement data to create decision tree model for cost-effectiveness analysis Cost model designed for period of 20 yrs TDR was more beneficial and less expensive than ACF 27
28 Cost Only construct less expensive than TDR was Brantigan cage w no graft not standard of care Other cost studies also favor TDR TDR less expensive than ACF Insurance industry data generally unavailable Blue Health spun off as a for profit venture by the Blues, allowing access to payment database Allowed authors to work backwards from payments to clinical events (post op, peri op, and pre op) by CPT and ICD 9 codes Total Costs: TDR ~ 12% Lower TDR ACDF Index event $20,722 $22,379 Index event + 90 day global post op $22,761 $25,029 period Discharge to 6 wks $791 $1,236 6 wks to 3 mos $1,216 $1,497 3 to 6 mos $2,147 $2,631 6 to 12 mos $4,127 $4, to 18 mos $3,106 $3, to 24 mos $2,862 $3, to 36 mos $3,753 $4, to 48 mos $1,040 $1,526 TOTAL $34,979 $39,820 Radcliff et al., Spine
29 Center of rotation (COR) with simplify disc Flexion/Extension Pre-Op Flexion/Extension Post-Op (6 Mos) Single level IDE study demographics Number of Subjects Enrollment Male Female Age and BMI (mean, min/max) 10 0 Total to date Approved 0 Age (yrs) BMI (kg/m2) Single level IDE study ndi NDI Pre Op 6 weeks 3 months 6 months 29
30 Single level IDE study VAS (Neck and Arm) 90 VAS (neck, arm) Pre Op Post Op 6 weeks 3 months 6 months Mobi C: Adjacent Segment Degeneration (Kellgren Lawrence Scale) 30% 25% 20% 15% 10% * * TDR ACDF 5% 0% 12 Months 24 Months 12 Months 24 Months Superior Adjacent Inferior Adjacent * Adjacent segment degeneration significant (p<0.05) Hisey et al Mobi C: 7 yr Follow up Improvement in NDI, VAS neck pain, and SF-12 scores similar between groups TDR group maintained segmental ROM at every time point through 84 mo No significant difference in rates of AEs or major complications Hisey et al, NASS,
5/19/2017. Disclosures. Introduction. How Much Kyphosis is Allowable for Cervical Total Disc Replacement? And Other Considerations
 How Much Kyphosis is Allowable for Cervical Total Disc Replacement? And Other Considerations Richard D. Guyer, M.D. Disclosures Guyer (a) Alphatec; (b) Spinal Kinetics, Spinal Ventures, Mimedix; (c) DePuy
How Much Kyphosis is Allowable for Cervical Total Disc Replacement? And Other Considerations Richard D. Guyer, M.D. Disclosures Guyer (a) Alphatec; (b) Spinal Kinetics, Spinal Ventures, Mimedix; (c) DePuy
Stability of Clinical Outcome Measures Following Anterior Cervical Spine Surgery
 Stability of Clinical Outcome Measures Following Anterior Cervical Spine Surgery Donna D. Ohnmeiss, Dr.Med., Richard D. Guyer, M.D., Jack E. Zigler, M.D., Scott L. Blumenthal, M.D. Disclosures: Ohnmeiss
Stability of Clinical Outcome Measures Following Anterior Cervical Spine Surgery Donna D. Ohnmeiss, Dr.Med., Richard D. Guyer, M.D., Jack E. Zigler, M.D., Scott L. Blumenthal, M.D. Disclosures: Ohnmeiss
The Artificial Cervical Disc: 2016 update
 The Artificial Cervical Disc: 2016 update Anthony K. Frempong-Boadu, MD Associate Professor Co-Director NYU Langone Spine Center Director - Division of Spinal Surgery Department of Neurosurgery New York
The Artificial Cervical Disc: 2016 update Anthony K. Frempong-Boadu, MD Associate Professor Co-Director NYU Langone Spine Center Director - Division of Spinal Surgery Department of Neurosurgery New York
ProDisc-L Total Disc Replacement. IDE Clinical Study.
 ProDisc-L Total Disc Replacement. IDE Clinical Study. A multi-center, prospective, randomized clinical trial. Instruments and implants approved by the AO Foundation Table of Contents Indications, Contraindications
ProDisc-L Total Disc Replacement. IDE Clinical Study. A multi-center, prospective, randomized clinical trial. Instruments and implants approved by the AO Foundation Table of Contents Indications, Contraindications
Freedom Lumbar Disc Clinical Outcomes Benchmarked Against All TDRs in the SWISSspine Registry
 Freedom Lumbar Disc Clinical Outcomes Benchmarked Against All TDRs in the SWISSspine Registry CAUTION: Investigational device. Limited by Federal law to investigational use. 1 White Paper Freedom Lumbar
Freedom Lumbar Disc Clinical Outcomes Benchmarked Against All TDRs in the SWISSspine Registry CAUTION: Investigational device. Limited by Federal law to investigational use. 1 White Paper Freedom Lumbar
TOTAL ARTIFICIAL DISC REPLACEMENT FOR THE SPINE
 TOTAL ARTIFICIAL DISC REPLACEMENT FOR THE SPINE Protocol: OTH034 Effective Date: July 1, 2017 Table of Contents Page COMMERCIAL COVERAGE RATIONALE... 1 MEDICARE & MEDICAID COVERAGE RATIONALE... 2 DESCRIPTION
TOTAL ARTIFICIAL DISC REPLACEMENT FOR THE SPINE Protocol: OTH034 Effective Date: July 1, 2017 Table of Contents Page COMMERCIAL COVERAGE RATIONALE... 1 MEDICARE & MEDICAID COVERAGE RATIONALE... 2 DESCRIPTION
Artificial Intervertebral Disc
 Medical Policy Manual Surgery, Policy No. 127 Artificial Intervertebral Disc Next Review: February 2019 Last Review: September 2018 Effective: October 1, 2018 IMPORTANT REMINDER Medical Policies are developed
Medical Policy Manual Surgery, Policy No. 127 Artificial Intervertebral Disc Next Review: February 2019 Last Review: September 2018 Effective: October 1, 2018 IMPORTANT REMINDER Medical Policies are developed
Corporate Medical Policy
 Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: artificial_intervertebral_disc 4/2004 10/2017 10/2018 10/2017 Description of Procedure or Service During
Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: artificial_intervertebral_disc 4/2004 10/2017 10/2018 10/2017 Description of Procedure or Service During
Matthew Colman, MD Assistant Professor, Spine Surgery and Musculoskeletal Oncology Rush University Medical Center ACDF
 is the most reliable option for twolevel anterior cervical surgery Matthew Colman, MD Assistant Professor, Spine Surgery and Musculoskeletal Oncology Rush University Medical Center Disclosures Medicrea:
is the most reliable option for twolevel anterior cervical surgery Matthew Colman, MD Assistant Professor, Spine Surgery and Musculoskeletal Oncology Rush University Medical Center Disclosures Medicrea:
TOTAL ARTIFICIAL DISC REPLACEMENT FOR THE SPINE
 UnitedHealthcare Commercial Medical Policy TOTAL ARTIFICIAL DISC REPLACEMENT FOR THE SPINE Policy Number: 2017T0437R Effective Date: May 1, 2017 Table of Contents Page INSTRUCTIONS FOR USE... 1 BENEFIT
UnitedHealthcare Commercial Medical Policy TOTAL ARTIFICIAL DISC REPLACEMENT FOR THE SPINE Policy Number: 2017T0437R Effective Date: May 1, 2017 Table of Contents Page INSTRUCTIONS FOR USE... 1 BENEFIT
Systematic review Cervical artificial disc replacement versus fusion in the cervical spine: a systematic review (...)
 Systematic review Cervical artificial disc replacement versus fusion in the cervical spine: a systematic review (...) 59 59 66 Cervical artificial disc replacement versus fusion in the cervical spine:
Systematic review Cervical artificial disc replacement versus fusion in the cervical spine: a systematic review (...) 59 59 66 Cervical artificial disc replacement versus fusion in the cervical spine:
Two-Level Cervical Total Disc Replacement versus ACDF: Results of a Prospective, Randomized, Clinical Trial with 48 Months Follow-up
 Two-Level Cervical Total Disc Replacement versus : Results of a Prospective, Randomized, Clinical Trial with 48 Follow-up Prof Jean Paul Steib 1, MD; Reginald Davis, MD 2 1 Spine Surgery Dept, University
Two-Level Cervical Total Disc Replacement versus : Results of a Prospective, Randomized, Clinical Trial with 48 Follow-up Prof Jean Paul Steib 1, MD; Reginald Davis, MD 2 1 Spine Surgery Dept, University
GECO 2010 LES-ARCS FRANCE LP-ESP II Elastic Spine PAD2 FH-ORTHOPEDICS
 GECO 2010 LES-ARCS FRANCE LP-ESP II Elastic Spine PAD2 FH-ORTHOPEDICS First results with 100 implanted LP-ESP after one year Klinikum Leer Departement of Spinal Surgery Germany Dr. med. Hamid Afshar Klinikum
GECO 2010 LES-ARCS FRANCE LP-ESP II Elastic Spine PAD2 FH-ORTHOPEDICS First results with 100 implanted LP-ESP after one year Klinikum Leer Departement of Spinal Surgery Germany Dr. med. Hamid Afshar Klinikum
ProDisc-L Total Disc Replacement. IDE Clinical Study
 Total Disc Replacement IDE Clinical Study Study Design TDR vs. circumferential fusion: Multi-center, prospective, randomized trial 17 centers, 292 patients 162 patients 80 fusion patients 50 non-randomized
Total Disc Replacement IDE Clinical Study Study Design TDR vs. circumferential fusion: Multi-center, prospective, randomized trial 17 centers, 292 patients 162 patients 80 fusion patients 50 non-randomized
Quality of Life. Quality of Motion.
 Quality of Life. Quality of Motion. Lateral Bend Vertical Translation Flexion Extension Lateral Translation Axial Rotation Anterior Posterior Translation Motion in all Directions Kinematics is the study
Quality of Life. Quality of Motion. Lateral Bend Vertical Translation Flexion Extension Lateral Translation Axial Rotation Anterior Posterior Translation Motion in all Directions Kinematics is the study
Single-Level Radiculopathy. Artificial Disc: It Works BETTER than ACDF
 Single-Level Radiculopathy Artificial Disc: It Works BETTER than ACDF Pierce D. Nunley MD Director, Spine Institute of Louisiana Assistant Professor, Louisiana State University Disclosures Stock ownership:
Single-Level Radiculopathy Artificial Disc: It Works BETTER than ACDF Pierce D. Nunley MD Director, Spine Institute of Louisiana Assistant Professor, Louisiana State University Disclosures Stock ownership:
MEDICAL POLICY SUBJECT: ARTIFICIAL CERVICAL INTERVERTEBRAL DISC
 MEDICAL POLICY SUBJECT: ARTIFICIAL CERVICAL PAGE: 1 OF: 13 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product, including
MEDICAL POLICY SUBJECT: ARTIFICIAL CERVICAL PAGE: 1 OF: 13 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product, including
Artificial Intervertebral Disc: Cervical Spine
 Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
Lumbar Facet Joint Replacement
 Rome Spine 2011 THE SPINE TODAY International Congress Rome 6-7th December 2011 Lumbar Facet Joint Replacement Prof. Dr. Karin Büttner-Janz Past President International Society for the Advancement of Spine
Rome Spine 2011 THE SPINE TODAY International Congress Rome 6-7th December 2011 Lumbar Facet Joint Replacement Prof. Dr. Karin Büttner-Janz Past President International Society for the Advancement of Spine
POLICY PRODUCT VARIATIONS DESCRIPTION/BACKGROUND RATIONALE DEFINITIONS BENEFIT VARIATIONS DISCLAIMER CODING INFORMATION REFERENCES POLICY HISTORY
 Original Issue Date (Created): June 14, 2004 Most Recent Review Date (Revised): July 22, 2014 Effective Date: October 1, 2014 POLICY PRODUCT VARIATIONS DESCRIPTION/BACKGROUND RATIONALE DEFINITIONS BENEFIT
Original Issue Date (Created): June 14, 2004 Most Recent Review Date (Revised): July 22, 2014 Effective Date: October 1, 2014 POLICY PRODUCT VARIATIONS DESCRIPTION/BACKGROUND RATIONALE DEFINITIONS BENEFIT
TOTAL ARTIFICIAL DISC REPLACEMENT FOR THE SPINE
 UnitedHealthcare Commercial Medical Policy TOTAL ARTIFICIAL DISC REPLACEMENT FOR THE SPINE Policy Number: 2018T0437T Effective Date: November 1, 2018 Instructions for Use Table of Contents Page COVERAGE
UnitedHealthcare Commercial Medical Policy TOTAL ARTIFICIAL DISC REPLACEMENT FOR THE SPINE Policy Number: 2018T0437T Effective Date: November 1, 2018 Instructions for Use Table of Contents Page COVERAGE
Artificial Intervertebral Disc: Cervical Spine
 Protocol Artificial Intervertebral Disc: Cervical Spine (701108) Medical Benefit Effective Date: 01/01/08 Next Review Date: 03/13 Preauthorization* No Review Dates: 06/07, 07/08, 05/09, 05/10, 03/11, 03/12
Protocol Artificial Intervertebral Disc: Cervical Spine (701108) Medical Benefit Effective Date: 01/01/08 Next Review Date: 03/13 Preauthorization* No Review Dates: 06/07, 07/08, 05/09, 05/10, 03/11, 03/12
Quality of Life. Quality of Motion. A Patient s Guide to. Artificial Lumbar Disc Replacement
 Quality of Life. Quality of Motion. A Patient s Guide to Artificial Lumbar Disc Replacement Each year, hundreds of thousands of adults are diagnosed with Lumbar Disc Degeneration, a lower spine condition
Quality of Life. Quality of Motion. A Patient s Guide to Artificial Lumbar Disc Replacement Each year, hundreds of thousands of adults are diagnosed with Lumbar Disc Degeneration, a lower spine condition
Artificial Intervertebral Disc: Cervical Spine
 Artificial Intervertebral Disc: Cervical Spine Policy Number: 7.01.108 Last Review: 6/2014 Origination: 12/2007 Next Review: 12/2014 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will not
Artificial Intervertebral Disc: Cervical Spine Policy Number: 7.01.108 Last Review: 6/2014 Origination: 12/2007 Next Review: 12/2014 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will not
Cervical Motion Preservation
 Spinal Disorders D. Pelinkovic, M. D. M&M Orthopaedics 1259 Rickert Drive Naperville, IL 1900 Ogden Ave Aurora, IL Cervical Motion Preservation Neck Pain Symptoms Trapezius myalgia ( Phosphates Bengston
Spinal Disorders D. Pelinkovic, M. D. M&M Orthopaedics 1259 Rickert Drive Naperville, IL 1900 Ogden Ave Aurora, IL Cervical Motion Preservation Neck Pain Symptoms Trapezius myalgia ( Phosphates Bengston
Medical Policy An independent licensee of the Blue Cross Blue Shield Association
 Artificial Intervertebral Disc: Cervical Spine Page 1 of 23 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: See also: Artificial Intervertebral Disc: Cervical Spine
Artificial Intervertebral Disc: Cervical Spine Page 1 of 23 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: See also: Artificial Intervertebral Disc: Cervical Spine
Artificial Intervertebral Disc: Cervical Spine
 Artificial Intervertebral Disc: Cervical Spine Policy Number: 7.01.108 Last Review: 5/2018 Origination: 12/2007 Next Review: 11/2018 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will provide
Artificial Intervertebral Disc: Cervical Spine Policy Number: 7.01.108 Last Review: 5/2018 Origination: 12/2007 Next Review: 11/2018 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will provide
Artificial Intervertebral Disc: Cervical Spine. Description
 Subject: Artificial Intervertebral Disc: Cervical Spine Page: 1 of 22 Last Review Status/Date: September 2015 Artificial Intervertebral Disc: Cervical Spine Description Several prosthetic devices are currently
Subject: Artificial Intervertebral Disc: Cervical Spine Page: 1 of 22 Last Review Status/Date: September 2015 Artificial Intervertebral Disc: Cervical Spine Description Several prosthetic devices are currently
Adjacent segment disease and C-ADR: promises fulfilled?
 Systematic review Adjacent segment disease and C-ADR: promises fulfilled? 39 39 46 Adjacent segment disease and C-ADR: promises fulfilled? Authors K Daniel Riew 1, Jeannette M Schenk-Kisser 2, Andrea C
Systematic review Adjacent segment disease and C-ADR: promises fulfilled? 39 39 46 Adjacent segment disease and C-ADR: promises fulfilled? Authors K Daniel Riew 1, Jeannette M Schenk-Kisser 2, Andrea C
Artificial Intervertebral Disc: Cervical Spine
 7.01.108 Artificial Intervertebral Disc: Cervical Spine Section 7.0 Surgery Subsection Effective Date December 15, 2014 Original Policy Date December 15, 2014 Next Review Date December 2015 Medical Policy
7.01.108 Artificial Intervertebral Disc: Cervical Spine Section 7.0 Surgery Subsection Effective Date December 15, 2014 Original Policy Date December 15, 2014 Next Review Date December 2015 Medical Policy
Innovative Techniques in Minimally Invasive Cervical Spine Surgery. Bruce McCormack, MD San Francisco California
 Innovative Techniques in Minimally Invasive Cervical Spine Surgery Bruce McCormack, MD San Francisco California PCF Posterior Cervical Fusion PCF not currently an ambulatory care procedure Pearl diver
Innovative Techniques in Minimally Invasive Cervical Spine Surgery Bruce McCormack, MD San Francisco California PCF Posterior Cervical Fusion PCF not currently an ambulatory care procedure Pearl diver
MEDICAL POLICY SUBJECT: ARTIFICIAL LUMBAR INTERVERTEBRAL DISC
 MEDICAL POLICY SUBJECT: ARTIFICIAL LUMBAR PAGE: 1 OF: 7 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product, including an
MEDICAL POLICY SUBJECT: ARTIFICIAL LUMBAR PAGE: 1 OF: 7 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product, including an
Intervertebral Disc Prostheses
 (https://www.aetna.com/) Intervertebral Disc Prostheses Number: 0591 Policy *Please see amendment for Pennsylvania Medicaid at the end of this CPB. Last Review 06/23/2016 Effective: 03/08/2002 Next Review:
(https://www.aetna.com/) Intervertebral Disc Prostheses Number: 0591 Policy *Please see amendment for Pennsylvania Medicaid at the end of this CPB. Last Review 06/23/2016 Effective: 03/08/2002 Next Review:
Artificial Disc Replacement, Cervical
 Artificial Disc Replacement, Cervical Policy Number: Original Effective Date: MM.06.001 02/01/2010 Line(s) of Business: Current Effective Date: HMO; PPO 11/01/2011 Section: Surgery Place(s) of Service:
Artificial Disc Replacement, Cervical Policy Number: Original Effective Date: MM.06.001 02/01/2010 Line(s) of Business: Current Effective Date: HMO; PPO 11/01/2011 Section: Surgery Place(s) of Service:
Freedom. Lumbar Disc Polymer-Metal Bond Integrity. CAUTION: Investigational device. Limited by Federal law to investigational use.
 Freedom Lumbar Disc Polymer-Metal Bond Integrity CAUTION: Investigational device. Limited by Federal law to investigational use. 1 White Paper Freedom Lumbar Disc Polymer-Metal Bond Integrity Abstract
Freedom Lumbar Disc Polymer-Metal Bond Integrity CAUTION: Investigational device. Limited by Federal law to investigational use. 1 White Paper Freedom Lumbar Disc Polymer-Metal Bond Integrity Abstract
Lumbar Disc Replacement FAQs
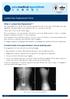 ISO 9001:2015 FS 550968 What is Lumbar Disc Replacement? The intervertebral discs are the shock absorbers between the bones of the spine. Unfortunately, they often degenerate tearing, bursting, or just
ISO 9001:2015 FS 550968 What is Lumbar Disc Replacement? The intervertebral discs are the shock absorbers between the bones of the spine. Unfortunately, they often degenerate tearing, bursting, or just
The Changing Landscape of ASCs: Cash Cow or Troubled Venture for Surgeons?
 The Changing Landscape of ASCs: Cash Cow or Troubled Venture for Surgeons? Richard D. Guyer, M.D. Chairman, Texas Back Institute RF Associate Clinical Professor UT Southwestern Medical School Disclosures
The Changing Landscape of ASCs: Cash Cow or Troubled Venture for Surgeons? Richard D. Guyer, M.D. Chairman, Texas Back Institute RF Associate Clinical Professor UT Southwestern Medical School Disclosures
Mobi-C CERVICAL DISC TWO-LEVEL APPLICATIONS
 Mobi-C CERVICAL DISC Product Brochure Features TWO-LEVEL APPLICATIONS INDICATIONS mobi-c Cervical disc bone sparing two-level indications Ease of insertion mobile bearing Over 17,000 Mobi-C discs have
Mobi-C CERVICAL DISC Product Brochure Features TWO-LEVEL APPLICATIONS INDICATIONS mobi-c Cervical disc bone sparing two-level indications Ease of insertion mobile bearing Over 17,000 Mobi-C discs have
MEDICAL POLICY MEDICAL POLICY DETAILS POLICY STATEMENT. Page: 1 of 7
 Page: 1 of 7 MEDICAL POLICY MEDICAL POLICY DETAILS Medical Policy Title ARTIFICIAL LUMBAR INTERVERTEBRAL DISC Policy Number 7.01.63 Category Technology Assessment Effective Date 03/18/04 Revised Date 03/17/05,
Page: 1 of 7 MEDICAL POLICY MEDICAL POLICY DETAILS Medical Policy Title ARTIFICIAL LUMBAR INTERVERTEBRAL DISC Policy Number 7.01.63 Category Technology Assessment Effective Date 03/18/04 Revised Date 03/17/05,
Interspinous Fusion Devices. Midterm results. ROME SPINE 2012, 7th International Meeting Rome, 6-7 December 2012
 Interspinous Fusion Devices. Midterm results. ROME SPINE 2012, 7th International Meeting Rome, 6-7 December 2012 Posterior distraction and decompression Secure Fixation and Stabilization Integrated Bone
Interspinous Fusion Devices. Midterm results. ROME SPINE 2012, 7th International Meeting Rome, 6-7 December 2012 Posterior distraction and decompression Secure Fixation and Stabilization Integrated Bone
Artificial Intervertebral Disc Replacement - Cervical
 Artificial Intervertebral Disc Replacement - Cervical Policy Number: Original Effective Date: MM.06.001 02/01/2010 Line(s) of Business: Current Effective Date: HMO; PPO; QUEST Integration 11/01/2018 Section:
Artificial Intervertebral Disc Replacement - Cervical Policy Number: Original Effective Date: MM.06.001 02/01/2010 Line(s) of Business: Current Effective Date: HMO; PPO; QUEST Integration 11/01/2018 Section:
5/19/2017. Interspinous Process Fixation with the Minuteman G3. What is the Minuteman G3. How Does it Work?
 Interspinous Process Fixation with the Minuteman G3 LLOYDINE J. JACOBS, MD CASTELLVI SPINE MEETING MAY 13, 2017 What is the Minuteman G3 The world s first spinous process plating system that is: Minimally
Interspinous Process Fixation with the Minuteman G3 LLOYDINE J. JACOBS, MD CASTELLVI SPINE MEETING MAY 13, 2017 What is the Minuteman G3 The world s first spinous process plating system that is: Minimally
3D titanium interbody fusion cages sharx. White Paper
 3D titanium interbody fusion cages sharx (SLM selective laser melted) Goal of the study: Does the sharx intervertebral cage due to innovative material, new design, and lordotic shape solve some problems
3D titanium interbody fusion cages sharx (SLM selective laser melted) Goal of the study: Does the sharx intervertebral cage due to innovative material, new design, and lordotic shape solve some problems
Outpatient Spine Surgery: The Next Five Years. Richard Wohns, M.D., JD, MBA Neospine, Puget Sound Region, Washington
 Outpatient Spine Surgery: The Next Five Years Richard Wohns, M.D., JD, MBA Neospine, Puget Sound Region, Washington Disclosures Aqueduct Neurosciences, Board of Directors angelmd, Chief Medical Officer
Outpatient Spine Surgery: The Next Five Years Richard Wohns, M.D., JD, MBA Neospine, Puget Sound Region, Washington Disclosures Aqueduct Neurosciences, Board of Directors angelmd, Chief Medical Officer
Artificial Disc Replacement, Cervical
 Artificial Disc Replacement, Cervical Policy Number: Original Effective Date: MM.06.001 02/01/2010 Line(s) of Business: Current Effective Date: HMO; PPO; QUEST 01/01/2014 Section: Surgery Place(s) of Service:
Artificial Disc Replacement, Cervical Policy Number: Original Effective Date: MM.06.001 02/01/2010 Line(s) of Business: Current Effective Date: HMO; PPO; QUEST 01/01/2014 Section: Surgery Place(s) of Service:
TOTAL ARTIFICIAL DISC REPLACEMENT FOR THE SPINE
 UnitedHealthcare Oxford Clinical Policy TOTAL ARTIFICIAL DISC REPLACEMENT FOR THE SPINE Policy Number: DME 021.27 T2 Effective Date: September 1, 2018 Table of Contents Page INSTRUCTIONS FOR USE... 1 CONDITIONS
UnitedHealthcare Oxford Clinical Policy TOTAL ARTIFICIAL DISC REPLACEMENT FOR THE SPINE Policy Number: DME 021.27 T2 Effective Date: September 1, 2018 Table of Contents Page INSTRUCTIONS FOR USE... 1 CONDITIONS
Cervical Disc Arthroplasty Reimbursement Guide
 Cervical Disc Arthroplasty 2015 Reimbursement Guide 63075 63064 63057 63045 2285163055 22553 22556 63048 20930 22612 22851 20937 3047 22614 20936 63057 63090 22612 63005 20840 61783 22551 22595 63055 63042
Cervical Disc Arthroplasty 2015 Reimbursement Guide 63075 63064 63057 63045 2285163055 22553 22556 63048 20930 22612 22851 20937 3047 22614 20936 63057 63090 22612 63005 20840 61783 22551 22595 63055 63042
DISCLOSURES. Goal of Fusion. Expandable Cages: Do they play a role in lumbar MIS surgery? CON 2/15/2017
 Expandable Cages: Do they play a role in lumbar MIS surgery? CON Jean-Jacques Abitbol, M.D., FRCSC San Diego, California DISCLOSURES SAB; K2M, Osprey, Nanovis, Vertera, St Theresa Royalties; Osprey, K2M,
Expandable Cages: Do they play a role in lumbar MIS surgery? CON Jean-Jacques Abitbol, M.D., FRCSC San Diego, California DISCLOSURES SAB; K2M, Osprey, Nanovis, Vertera, St Theresa Royalties; Osprey, K2M,
Agreement to Acquire Spinal Kinetics, Inc. March 15, 2018
 Agreement to Acquire Spinal Kinetics, Inc. 1 March 15, 2018 2 SAFE HARBOR STATEMENT AND USE OF NON GAAP MEASURES WHY SPINAL KINETICS? The Spinal Kinetics M6 artificial discs further round out Orthofix
Agreement to Acquire Spinal Kinetics, Inc. 1 March 15, 2018 2 SAFE HARBOR STATEMENT AND USE OF NON GAAP MEASURES WHY SPINAL KINETICS? The Spinal Kinetics M6 artificial discs further round out Orthofix
Cervical artificial disc replacement versus fusion in the cervical spine: a systematic review comparing multilevel versus single-level surgery
 Systematic review Cervical artificial disc replacement versus fusion in the cervical spine: a systematic review ( ) 19 19 30 Cervical artificial disc replacement versus fusion in the cervical spine: a
Systematic review Cervical artificial disc replacement versus fusion in the cervical spine: a systematic review ( ) 19 19 30 Cervical artificial disc replacement versus fusion in the cervical spine: a
Original Effective Date:6/14/06 Revision Date(s): 1/28/09, 12/14/11, 4/14
 Subject: Artificial Intervertebral Disc Replacement (ADR) Surgery (Lumbar and Cervical) Guidance Number: MCG-011 Medical Coverage Guidance Approval Date: 4/2/14 PREFACE This Medical Guidance is intended
Subject: Artificial Intervertebral Disc Replacement (ADR) Surgery (Lumbar and Cervical) Guidance Number: MCG-011 Medical Coverage Guidance Approval Date: 4/2/14 PREFACE This Medical Guidance is intended
Intervertebral Disc (IVD) Prostheses
 Medical Coverage Policy Intervertebral Disc (IVD) Prostheses Effective Date... 1/15/2018 Next Review Date... 1/15/2019 Coverage Policy Number... 0104 Table of Contents Coverage Policy... 1 Overview...
Medical Coverage Policy Intervertebral Disc (IVD) Prostheses Effective Date... 1/15/2018 Next Review Date... 1/15/2019 Coverage Policy Number... 0104 Table of Contents Coverage Policy... 1 Overview...
Am I eligible for the TOPS study? Possibly, if you suffer from one or more of the following conditions:
 Am I eligible for the TOPS study? Possibly, if you suffer from one or more of the following conditions: Radiating leg pain Greater leg / buttock pain than back pain Severe pain sets in when walking as
Am I eligible for the TOPS study? Possibly, if you suffer from one or more of the following conditions: Radiating leg pain Greater leg / buttock pain than back pain Severe pain sets in when walking as
porous titanium cervical implants.
 Preliminary results of quantitative functional X-ray analysis of CABO meeting 2017 stand-alone interbody 3D printed porous titanium cervical implants. Mark Arts, MD PhD Jasper Wolfs, MD Neurosurgery Department
Preliminary results of quantitative functional X-ray analysis of CABO meeting 2017 stand-alone interbody 3D printed porous titanium cervical implants. Mark Arts, MD PhD Jasper Wolfs, MD Neurosurgery Department
Cigna Medical Coverage Policy
 Cigna Medical Coverage Policy Subject Intervertebral Disc (IVD) Prostheses Table of Contents Coverage Policy... 1 General Background... 3 Coding/Billing Information... 25 References... 26 Effective Date...
Cigna Medical Coverage Policy Subject Intervertebral Disc (IVD) Prostheses Table of Contents Coverage Policy... 1 General Background... 3 Coding/Billing Information... 25 References... 26 Effective Date...
FEP Medical Policy Manual
 FEP Medical Policy Manual Last Review: December 2016 Effective Date: January 15, 2017 Related Policies: 7.01.87 Artificial Intervertebral Disc: Lumbar Spine Artificial Intervertebral Disc: Cervical Spine
FEP Medical Policy Manual Last Review: December 2016 Effective Date: January 15, 2017 Related Policies: 7.01.87 Artificial Intervertebral Disc: Lumbar Spine Artificial Intervertebral Disc: Cervical Spine
EFSPINE CERVICAL COMBINED SET DISC PROTHESIS ORGANIZER BOX
 EFSPINE CERVICAL COMBINED SET INSTRUMENTS CERVICAL CAGE & DISC PROTHESIS ORGANIZER BOX Cervical Thoracic Thoraco - Lumbar Sacral EFSPINE CERVICAL COMBINED SET CERVICAL IMPLANTS INTRODUCTION Cervical Disc
EFSPINE CERVICAL COMBINED SET INSTRUMENTS CERVICAL CAGE & DISC PROTHESIS ORGANIZER BOX Cervical Thoracic Thoraco - Lumbar Sacral EFSPINE CERVICAL COMBINED SET CERVICAL IMPLANTS INTRODUCTION Cervical Disc
Original Policy Date
 MP 7.01.88 Artificial Intervertebral Disc: Cervical Spine Medical Policy Section Surgery Issue 12/2013 Original Policy Date 12/2013 Last Review Status/Date Local policy created/12/2013 Return to Medical
MP 7.01.88 Artificial Intervertebral Disc: Cervical Spine Medical Policy Section Surgery Issue 12/2013 Original Policy Date 12/2013 Last Review Status/Date Local policy created/12/2013 Return to Medical
Mobi-C Cervical Disc Bibliography as of September 2015
 Mobi-C Cervical Disc Bibliography as of September 2015 PUBLISHED PAPERS Bae H, Kim KD, Nunley PD, Jackson RJ, Hisey MS, Davis RJ, Hoffman GA, Gaede SE, Danielson GO, Peterson DL, Stokes JM, Araghi A: Comparison
Mobi-C Cervical Disc Bibliography as of September 2015 PUBLISHED PAPERS Bae H, Kim KD, Nunley PD, Jackson RJ, Hisey MS, Davis RJ, Hoffman GA, Gaede SE, Danielson GO, Peterson DL, Stokes JM, Araghi A: Comparison
Patient Information MIS LLIF. Lateral Lumbar Interbody Fusion Using Minimally Invasive Surgical Techniques
 Patient Information MIS LLIF Lateral Lumbar Interbody Fusion Using Minimally Invasive Surgical Techniques Table of Contents Anatomy of Spine...2 General Conditions of the Spine....4 What is Spondylolisthesis....5
Patient Information MIS LLIF Lateral Lumbar Interbody Fusion Using Minimally Invasive Surgical Techniques Table of Contents Anatomy of Spine...2 General Conditions of the Spine....4 What is Spondylolisthesis....5
Comparison of Clinical Outcomes Following Minimally Invasive Lateral Interbody Fusion Stratified by Preoperative Diagnosis
 Comparison of Clinical Outcomes Following Minimally Invasive Lateral Interbody Fusion Stratified by Preoperative Diagnosis Kaveh Khajavi, MD, FACS Alessandria Y. Shen, MSPH Anthony Hutchison, MSN Disclosures
Comparison of Clinical Outcomes Following Minimally Invasive Lateral Interbody Fusion Stratified by Preoperative Diagnosis Kaveh Khajavi, MD, FACS Alessandria Y. Shen, MSPH Anthony Hutchison, MSN Disclosures
ANTERIOR CERVICAL FUSION CERVICAL TOTAL DISC ARTHROPLASTY
 ANTERIOR CERVICAL FUSION CERVICAL TOTAL DISC ARTHROPLASTY LEFLOT JEAN-LOUIS M.D. UH BRUGMANN BRUSSELS WHAT INTERESTS US? ANATOMY (R.LOUIS) ANATOMY PATHOLOGICAL ANATOMY PATHOLOGICAL ANATOMY SAGITTAL DISBALANCE
ANTERIOR CERVICAL FUSION CERVICAL TOTAL DISC ARTHROPLASTY LEFLOT JEAN-LOUIS M.D. UH BRUGMANN BRUSSELS WHAT INTERESTS US? ANATOMY (R.LOUIS) ANATOMY PATHOLOGICAL ANATOMY PATHOLOGICAL ANATOMY SAGITTAL DISBALANCE
6/13/2014. Cervical Disc Arthroplasty (CDA) preserves motion
 6/13/2014 Joseph R. O Brien, MD, MPH Associate Director of Spine Surgery, GWUH Associate Professor of Orthopaedic Surgery and Neurosurgery Department of Orthopaedic Surgery The George Washington University
6/13/2014 Joseph R. O Brien, MD, MPH Associate Director of Spine Surgery, GWUH Associate Professor of Orthopaedic Surgery and Neurosurgery Department of Orthopaedic Surgery The George Washington University
Patient Information MIS LLIF. Lateral Lumbar Interbody Fusion Using Minimally Invasive Surgical Techniques
 Patient Information MIS LLIF Lateral Lumbar Interbody Fusion Using Minimally Invasive Surgical Techniques Table of Contents Anatomy of Spine....2 General Conditions of the Spine....4 What is Spondylolisthesis....5
Patient Information MIS LLIF Lateral Lumbar Interbody Fusion Using Minimally Invasive Surgical Techniques Table of Contents Anatomy of Spine....2 General Conditions of the Spine....4 What is Spondylolisthesis....5
MAS TLIF MAXIMUM ACCESS SURGERY TRANSFORAMINAL LUMBAR INTERBODY FUSION AN INTRODUCTION TO
 AN INTRODUCTION TO MAS TLIF MAXIMUM ACCESS SURGERY TRANSFORAMINAL LUMBAR INTERBODY FUSION This booklet is designed to inform you about the Maximum Access Surgery (MAS ) Transforaminal Lumbar Interbody
AN INTRODUCTION TO MAS TLIF MAXIMUM ACCESS SURGERY TRANSFORAMINAL LUMBAR INTERBODY FUSION This booklet is designed to inform you about the Maximum Access Surgery (MAS ) Transforaminal Lumbar Interbody
The Role of Surgery in the Treatment of Low Back Pain and Radiculopathy. Christian Etter, MD, Spine Surgeon Zürich, Switzerland
 The Role of Surgery in the Treatment of Low Back Pain and Radiculopathy Christian Etter, MD, Spine Surgeon Zürich, Switzerland WW Fusion Volume by Disorder 2004E % Tumor/Trauma 11% Deformity 15% Degeneration
The Role of Surgery in the Treatment of Low Back Pain and Radiculopathy Christian Etter, MD, Spine Surgeon Zürich, Switzerland WW Fusion Volume by Disorder 2004E % Tumor/Trauma 11% Deformity 15% Degeneration
The Coflex vs. Fusion U.S. I.D.E. Trial An in vivo Biomechanical Study of Adjacent Segment Motion following Fusion
 The Coflex vs. Fusion U.S. I.D.E. Trial An in vivo Biomechanical Study of Adjacent Segment Motion following Fusion W.R. Sears 1, R.J. Davis 2, J.D. Auerbach 3 1 Wentworth Spine Clinic, Sydney, Australia,
The Coflex vs. Fusion U.S. I.D.E. Trial An in vivo Biomechanical Study of Adjacent Segment Motion following Fusion W.R. Sears 1, R.J. Davis 2, J.D. Auerbach 3 1 Wentworth Spine Clinic, Sydney, Australia,
5/27/2016. Stand-Alone Lumbar Lateral Interbody Fusion (LLIF) vs. Supplemental Fixation. Disclosures. LLIF Approach
 Stand-Alone Lumbar Lateral Interbody Fusion (LLIF) vs. Supplemental Fixation Joseph M. Zavatsky, M.D. Spine & Scoliosis Specialists Tampa, FL Disclosures Consultant - Zimmer / Biomet, DePuy Synthes Spine,
Stand-Alone Lumbar Lateral Interbody Fusion (LLIF) vs. Supplemental Fixation Joseph M. Zavatsky, M.D. Spine & Scoliosis Specialists Tampa, FL Disclosures Consultant - Zimmer / Biomet, DePuy Synthes Spine,
Anterior cervical diskectomy icd 10 procedure code
 Home Anterior cervical diskectomy icd 10 procedure code Access to discounts at hundreds of restaurants, travel destinations, retail stores, and service providers. AAPC members also have opportunities to
Home Anterior cervical diskectomy icd 10 procedure code Access to discounts at hundreds of restaurants, travel destinations, retail stores, and service providers. AAPC members also have opportunities to
Disc herniation caused by a viscoelastic nucleus after total lumbar disc replacement a case report
 Case Report Disc herniation caused by a viscoelastic nucleus after total lumbar disc replacement a case report Lukas Grassner 1,2,3,4, Andreas Grillhösl 5, Michael Bierschneider 1, Martin Strowitzki 1
Case Report Disc herniation caused by a viscoelastic nucleus after total lumbar disc replacement a case report Lukas Grassner 1,2,3,4, Andreas Grillhösl 5, Michael Bierschneider 1, Martin Strowitzki 1
The Fusion of Power and Performance.
 The Fusion of Power and Performance. Leading the Way in Lumbar Interbody Fusion with 1 Years of Clinical Results. The JAGUAR Lumbar I/F CAGE System, approved in the U.S. in February 1999, has revolutionized
The Fusion of Power and Performance. Leading the Way in Lumbar Interbody Fusion with 1 Years of Clinical Results. The JAGUAR Lumbar I/F CAGE System, approved in the U.S. in February 1999, has revolutionized
Comparison of Clinical Outcomes Following Minimally Invasive Lateral Interbody Fusion Stratified by Preoperative Diagnosis
 Comparison of Clinical Outcomes Following Minimally Invasive Lateral Interbody Fusion Stratified by Preoperative Diagnosis Kaveh Khajavi, MD, FACS Alessandria Y. Shen, MSPH Anthony Hutchison, MSN The following
Comparison of Clinical Outcomes Following Minimally Invasive Lateral Interbody Fusion Stratified by Preoperative Diagnosis Kaveh Khajavi, MD, FACS Alessandria Y. Shen, MSPH Anthony Hutchison, MSN The following
The Fusion of. Strength, Design. Performance. and
 The Fusion of Strength, Design and Performance Interbody Fusion Platform Interbody Fusion Platform The DePuy Spine Interbody Fusion platform is designed to provide an open architecture implant in a load-sharing
The Fusion of Strength, Design and Performance Interbody Fusion Platform Interbody Fusion Platform The DePuy Spine Interbody Fusion platform is designed to provide an open architecture implant in a load-sharing
Anew artificial spinal disc for patients with lower back pain is generating a
 Technology trends New artificial spinal disc has a learning curve, payment issues Anew artificial spinal disc for patients with lower back pain is generating a buzz. In October, the Food and Drug Administration
Technology trends New artificial spinal disc has a learning curve, payment issues Anew artificial spinal disc for patients with lower back pain is generating a buzz. In October, the Food and Drug Administration
Coflex TM for Lumbar Stenosis with
 Coflex TM for Lumbar Stenosis with Segmental Instability : 1 yr outcomes Eun-Sang Kim, M.D., Ph.D. Clinical Professor Dept of Neurosurgery Samsung Medical Center Seoul, Korea Surgery for Spinal Stenosis
Coflex TM for Lumbar Stenosis with Segmental Instability : 1 yr outcomes Eun-Sang Kim, M.D., Ph.D. Clinical Professor Dept of Neurosurgery Samsung Medical Center Seoul, Korea Surgery for Spinal Stenosis
U.S. MARKET FOR MINIMALLY INVASIVE SPINAL IMPLANTS
 U.S. MARKET FOR MINIMALLY INVASIVE SPINAL IMPLANTS idata_usmis15_rpt Published in December 2014 By idata Research Inc., 2014 idata Research Inc. Suite 308 4211 Kingsway Burnaby, British Columbia, Canada,
U.S. MARKET FOR MINIMALLY INVASIVE SPINAL IMPLANTS idata_usmis15_rpt Published in December 2014 By idata Research Inc., 2014 idata Research Inc. Suite 308 4211 Kingsway Burnaby, British Columbia, Canada,
MEDICAL POLICY SUBJECT: ARTIFICIAL CERVICAL INTERVERTEBRAL DISC
 MEDICAL POLICY SUBJECT: ARTIFICIAL CERVICAL PAGE: 1 OF: 14 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product (including
MEDICAL POLICY SUBJECT: ARTIFICIAL CERVICAL PAGE: 1 OF: 14 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product (including
Patient Information MIS TLIF. Transforaminal Lumbar Interbody Fusion Using Minimally Invasive Surgical Techniques
 Patient Information MIS TLIF Transforaminal Lumbar Interbody Fusion Using Minimally Invasive Surgical Techniques MIS TLIF Table of Contents Anatomy of Spine..............................................
Patient Information MIS TLIF Transforaminal Lumbar Interbody Fusion Using Minimally Invasive Surgical Techniques MIS TLIF Table of Contents Anatomy of Spine..............................................
Chapter 20 Advances in Biomaterials that Led to the Artificial Disc
 Chapter 20 Advances in Biomaterials that Led to the Artificial Disc Jeremy C. Wang, M.D. and Regis W. Haid, Jr., M.D. INTRODUCTION It is undeniable that advances in biomaterial technology were indispensable
Chapter 20 Advances in Biomaterials that Led to the Artificial Disc Jeremy C. Wang, M.D. and Regis W. Haid, Jr., M.D. INTRODUCTION It is undeniable that advances in biomaterial technology were indispensable
A Patient s Guide to Artificial Cervical Disc Replacement
 A Patient s Guide to Artificial Cervical Disc Replacement Each year, hundreds of thousands of adults are diagnosed with Cervical Disc Degeneration, an upper spine condition that can cause pain and numbness
A Patient s Guide to Artificial Cervical Disc Replacement Each year, hundreds of thousands of adults are diagnosed with Cervical Disc Degeneration, an upper spine condition that can cause pain and numbness
Technique Guide. NFlex. Semi-rigid rods for posterior lumbar stabilization.
 Technique Guide NFlex. Semi-rigid rods for posterior lumbar stabilization. Table of Contents Introduction NFlex 2 Indications and Contraindications 4 NFlex Principles 6 Surgical Technique Preoperative
Technique Guide NFlex. Semi-rigid rods for posterior lumbar stabilization. Table of Contents Introduction NFlex 2 Indications and Contraindications 4 NFlex Principles 6 Surgical Technique Preoperative
Patient Information MIS TLIF. Transforaminal Lumbar Interbody Fusion Using Minimally Invasive Surgical Techniques
 Patient Information MIS TLIF Transforaminal Lumbar Interbody Fusion Using Minimally Invasive Surgical Techniques MIS TLIF Table of Contents Anatomy of Spine...2 General Conditions of the Spine...4 6 MIS-TLIF
Patient Information MIS TLIF Transforaminal Lumbar Interbody Fusion Using Minimally Invasive Surgical Techniques MIS TLIF Table of Contents Anatomy of Spine...2 General Conditions of the Spine...4 6 MIS-TLIF
Solutions For The Aging Spine
 Solutions For The Aging Spine NASDAQ: ATEC February 2009 www.alphatecspine.com Slide 0 5818 El Camino Real, Carlsbad, CA 92008 www.alphatecspine.com Forward Looking Statements This presentation and our
Solutions For The Aging Spine NASDAQ: ATEC February 2009 www.alphatecspine.com Slide 0 5818 El Camino Real, Carlsbad, CA 92008 www.alphatecspine.com Forward Looking Statements This presentation and our
Original Date: October 2015 LUMBAR SPINAL FUSION FOR
 National Imaging Associates, Inc. Clinical guidelines Original Date: October 2015 LUMBAR SPINAL FUSION FOR Page 1 of 9 INSTABILITY AND DEGENERATIVE DISC CONDITIONS FOR CMS (MEDICARE) MEMBERS ONLY CPT4
National Imaging Associates, Inc. Clinical guidelines Original Date: October 2015 LUMBAR SPINAL FUSION FOR Page 1 of 9 INSTABILITY AND DEGENERATIVE DISC CONDITIONS FOR CMS (MEDICARE) MEMBERS ONLY CPT4
Artificial Intervertebral Disc: Lumbar Spine
 7.01.87 Artificial Intervertebral Disc: Lumbar Spine Section 7.0 Surgery Subsection Effective Date December 15, 2014 Original Policy Date March 5, 2012 Next Review Date December 2015 Medical Policy Description
7.01.87 Artificial Intervertebral Disc: Lumbar Spine Section 7.0 Surgery Subsection Effective Date December 15, 2014 Original Policy Date March 5, 2012 Next Review Date December 2015 Medical Policy Description
Clinical Policy Title: Cervical artificial total disc replacement
 Clinical Policy Title: Cervical artificial total disc replacement Clinical Policy Number: 03.03.09 Effective Date: October 1, 2014 Initial Review Date: June 18, 2014 Most Recent Review Date: June 22, 2017
Clinical Policy Title: Cervical artificial total disc replacement Clinical Policy Number: 03.03.09 Effective Date: October 1, 2014 Initial Review Date: June 18, 2014 Most Recent Review Date: June 22, 2017
Cigna Medical Coverage Policy
 Cigna Medical Coverage Policy Subject Intervertebral Disc (IVD) Prostheses Table of Contents Coverage Policy... 1 General Background... 3 Coding/Billing Information... 31 References... 33 Policy History...
Cigna Medical Coverage Policy Subject Intervertebral Disc (IVD) Prostheses Table of Contents Coverage Policy... 1 General Background... 3 Coding/Billing Information... 31 References... 33 Policy History...
OPERATIVE TECHNIQUE. CONSTRUX Mini PTC. Mini PTC Spacer System
 OPERATIVE TECHNIQUE CONSTRUX Mini PTC Mini PTC Spacer System TABLE OF CONTENTS Introduction 1 Operative Technique 2 Part Numbers 6 Indications For Use 7 INTRODUCTION 1 INTRODUCTION The CONSTRUX Mini PTC
OPERATIVE TECHNIQUE CONSTRUX Mini PTC Mini PTC Spacer System TABLE OF CONTENTS Introduction 1 Operative Technique 2 Part Numbers 6 Indications For Use 7 INTRODUCTION 1 INTRODUCTION The CONSTRUX Mini PTC
EXACTECH SPINE. Operative Technique. Cervical Spacer System. Surgeon focused. Patient driven. TM
 EXACTECH SPINE Operative Technique Cervical Spacer System Surgeon focused. Patient driven. TM ACAPELLA ONE Acapella One Cervical Spacer System is an anterior cervical discectomy and fusion device with
EXACTECH SPINE Operative Technique Cervical Spacer System Surgeon focused. Patient driven. TM ACAPELLA ONE Acapella One Cervical Spacer System is an anterior cervical discectomy and fusion device with
nvp Posterior Lumbar Interbody Fusion System
 nvp Posterior Lumbar Interbody Fusion System 1 IMPORTANT INFORMATION FOR PHYSICIANS, SURGEONS, AND/OR STAFF The nv a, nv p, and nv t are an intervertebral body fusion device used in the lumbar spine following
nvp Posterior Lumbar Interbody Fusion System 1 IMPORTANT INFORMATION FOR PHYSICIANS, SURGEONS, AND/OR STAFF The nv a, nv p, and nv t are an intervertebral body fusion device used in the lumbar spine following
nvt Transforaminal Lumbar Interbody Fusion System
 nvt Transforaminal Lumbar Interbody Fusion System 1 IMPORTANT INFORMATION FOR PHYSICIANS, SURGEONS, AND/OR STAFF The nv a, nv p, and nv t are an intervertebral body fusion device used in the lumbar spine
nvt Transforaminal Lumbar Interbody Fusion System 1 IMPORTANT INFORMATION FOR PHYSICIANS, SURGEONS, AND/OR STAFF The nv a, nv p, and nv t are an intervertebral body fusion device used in the lumbar spine
Lumbar Dynamic. Reinhold Knebel Product Manager
 Lumbar Dynamic Reinhold Knebel Product Manager The DSS system provides options for dynamic and rigid stabilization to treat hypermobile segments and segments that require fusion. Allows pedicle displacement
Lumbar Dynamic Reinhold Knebel Product Manager The DSS system provides options for dynamic and rigid stabilization to treat hypermobile segments and segments that require fusion. Allows pedicle displacement
Surgical treatment of symptomatic cervical radiculopathy
 SPINE Volume 40, Number 11, pp 759-766 2015, Wolters Kluwer Health, Inc. All rights reserved. Comparison of Clinical Outcomes of 1- and 2-Level Total Disc Replacement Four-Year Results From a Prospective,
SPINE Volume 40, Number 11, pp 759-766 2015, Wolters Kluwer Health, Inc. All rights reserved. Comparison of Clinical Outcomes of 1- and 2-Level Total Disc Replacement Four-Year Results From a Prospective,
Facet Arthroplasty. Policy Number: Last Review: 9/2018 Origination: 9/2009 Next Review: 3/2019
 Facet Arthroplasty Policy Number: 7.01.120 Last Review: 9/2018 Origination: 9/2009 Next Review: 3/2019 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will not provide coverage for total facet
Facet Arthroplasty Policy Number: 7.01.120 Last Review: 9/2018 Origination: 9/2009 Next Review: 3/2019 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will not provide coverage for total facet
ASJ. Outcome of Salvage Lumbar Fusion after Lumbar Arthroplasty. Asian Spine Journal. Introduction. Hussein Alahmadi, Harel Deutsch
 Asian Spine Journal Asian Spine Clinical Journal Study Asian Spine J 2014;8(1):13-18 Lumbar fusion http://dx.doi.org/10.4184/asj.2014.8.1.13 after lumbar arthroplasty 13 Outcome of Salvage Lumbar Fusion
Asian Spine Journal Asian Spine Clinical Journal Study Asian Spine J 2014;8(1):13-18 Lumbar fusion http://dx.doi.org/10.4184/asj.2014.8.1.13 after lumbar arthroplasty 13 Outcome of Salvage Lumbar Fusion
L owbackpainisanexceedinglycommoncauseofdisability
 PRIMARY RESEARCH Downloaded from https://journals.na.lww.com/jspinaldisorders by e8uh+klvnesopbmb8bes3ozqdxp/xhrsun8u2ygapsa0s4erxxgp8ncguoprx1y8hjlhwnesybvbzroippap4sa3e8uqxkdo3nhdm9y+ceycfqdxoebfvbevleari7wgjcqszcdxnbzzzxk0mjs+dwc55usiskslocjphk0n76fszxmdezkiig==
PRIMARY RESEARCH Downloaded from https://journals.na.lww.com/jspinaldisorders by e8uh+klvnesopbmb8bes3ozqdxp/xhrsun8u2ygapsa0s4erxxgp8ncguoprx1y8hjlhwnesybvbzroippap4sa3e8uqxkdo3nhdm9y+ceycfqdxoebfvbevleari7wgjcqszcdxnbzzzxk0mjs+dwc55usiskslocjphk0n76fszxmdezkiig==
Clinical Policy Title: Cervical artificial total disc replacement
 Clinical Policy Title: Cervical artificial total disc replacement Clinical Policy Number: 03.03.09 Effective Date: October 1, 2014 Initial Review Date: June 18, 2014 Most Recent Review Date: May 1, 2018
Clinical Policy Title: Cervical artificial total disc replacement Clinical Policy Number: 03.03.09 Effective Date: October 1, 2014 Initial Review Date: June 18, 2014 Most Recent Review Date: May 1, 2018
Introduction. Castellvi Spine Are We Closer to Finding the True Lumbar Pain Generator? DISCLOSURE 5/27/2016. Dom Coric, M.D.
 Castellvi Spine Are We Closer to Finding the True Lumbar Pain Generator? Dom Coric, M.D. Carolina Neurosurgery and Spine Associates Chief, Department of Neurosurgery, CMC Charlotte, NC 5/20/16 DISCLOSURE
Castellvi Spine Are We Closer to Finding the True Lumbar Pain Generator? Dom Coric, M.D. Carolina Neurosurgery and Spine Associates Chief, Department of Neurosurgery, CMC Charlotte, NC 5/20/16 DISCLOSURE
MEDICAL POLICY MEDICAL POLICY DETAILS POLICY STATEMENT. Page: 1 of 17
 Page: 1 of 17 MEDICAL POLICY MEDICAL POLICY DETAILS Medical Policy Title ARTIFICIAL CERVICAL INTERVERTEBRAL DISC Policy Number 7.01.80 Category Technology Assessment Effective Date 03/20/08 Revised Date
Page: 1 of 17 MEDICAL POLICY MEDICAL POLICY DETAILS Medical Policy Title ARTIFICIAL CERVICAL INTERVERTEBRAL DISC Policy Number 7.01.80 Category Technology Assessment Effective Date 03/20/08 Revised Date
