AVX-470, an Orally Delivered Anti-TNF Antibody for Treatment of Acute Ulcerative Colitis: Results of a First-in- Human Trial
|
|
|
- Stephen Goodwin
- 6 years ago
- Views:
Transcription
1 AVX-470, an Orally Delivered Anti-TNF Antibody for Treatment of Acute Ulcerative Colitis: Results of a First-in- Human Trial M Scott Harris 1, Deborah Hartman 1, Sharon Spence 1, Sally Kennedy 1, Theodore Ptak 2, Ronald Pruitt 3, Severine Vermeire 4, Barbara Fox 1 1 Avaxia Biologics, Lexington, United States; 2 Toronto Digestive Disease Associates, Toronto, Canada; 3 Nashville Medical Research Institute, Nashville, United States; 4 University Hospital Gasthuisberg, Leuven, Belgium
2 Disclosures I am currently consult for Avaxia Biologics, the sponsor of this study. I also disclose that I have consulted for or have had financial interests in the following companies or organizations in the past two years: Ardelyx Bill and Melinda Gates Foundation Biomedical Systems CIPAC CymaBay Drais Pharmaceuticals Kala Pharmaceuticals Lyric Pharmaceuticals Neurogastrx Ocera Therapeutics Orchid Pharmaceuticals PATH-One World Health Rhythm Pharmaceuticals Symbiomix Theravance White Sands Pharmaceuticals ZS Pharma
3 Disadvantages of Parenteral Anti-TNF Antibodies PARENTERAL ANTI-TNF Abs Parenterally administered Infusion reactions common Distribute throughout the body Cause systemic immunosuppression May develop anti-drug antibodies Drugs lose effectiveness
4 Advantages of Oral Gut-Targeted Anti-TNF Antibodies PARENTERAL ANTI-TNF Abs ORAL Anti-TNF Abs Parenterally administered Infusion reactions common Orally administered Distribute throughout the body Locally active Works at site of disease Cause systemic immunosuppression May develop anti-drug antibodies Drugs lose effectiveness No systemic immunosuppression Overcomes major drawback of anti-tnfs Anti-drug antibodies not induced Potential for better longterm efficacy
5 AVX-470 Milk-based polyclonal anti-tnf antibodies for oral delivery Pregnant dairy cows immunized with recombinant human TNF Antibody is purified from colostrum (early milk) Approximately % by weight is TNF-specific Antibody retains activity in GI tract Bovine milk antibodies are inherently stable to intestinal digestion Enteric coating designed to release drug at ph 6.0 Long history of safe oral exposure to bovine immunoglobulin Dairy products, meat, whey-based food additives Lactose free, no risk of BSE in dairy products
6 First-in-Human Trial of AVX-470 Study Endpoints and Objectives Endpoint Objective Primary Safety Assess safety in adult UC patients Assess systemic exposure Secondary Exploratory Pharmacokinetics Immunogenicity Pharmacodynamics Clinical efficacy Confirm stability of AVX-470 in human GI tract Assess ability of AVX-470 to penetrate colonic mucosa of UC patients Assess induction of anti-drug antibodies Measure TNF levels in tissues Assess systemic markers of disease activity (CRP, IL-6) Assess clinical and endoscopic remission and response
7 Overview Screening Double-blind treatment F/U Week Colonoscopy-Bx Biomarkers Colonoscopy-Bx Biomarkers 37 patients with active UC 13 centers in US, Canada, Belgium and Hungary Placebo-controlled, double-blind 4 weeks treatment, 3 ascending-dose cohorts 0.2 g/d, 1.6 g/d, and 3.5 g/d in divided dose 2x or 3x daily Within each cohort, patient randomized 3:1 to AVX-470 or placebo Colonoscopy and biopsy by central reading at baseline and Week 4
8 Inclusion and Exclusion Criteria Men and women ages Total Mayo score 5-12, inclusive Mayo endoscopic subscore 2 at or above 15 cm from anus Permitted medications: Concomitant use of 5-ASA, corticosteroids (up to prednisone 20mg equivalent), immunosuppressives Prior use of TNF agents (secondary failure only)
9 Definitions Clinical Response Reduction of 3 points on the total Mayo score and an overall decrease of at least 30%, plus a decrease in the rectal bleeding subscore of at least 1-point or an absolute rectal bleeding score of 1 or less Clinical Remission Total Mayo score of 2 or lower and no subscores higher than 1 Endoscopic Response 1-point decrease in Mayo endoscopic subscore Endoscopic Remission Mayo endoscopic subscore of 0-1
10 Colonoscopy UCEIS (Ulcerative Colitis Index of Severity) scored in 3 segments Proximal colon (ascending and transverse colons) Descending colon and sigmoid (above 15 cm) Rectum (below 15 cm)` Biopsies obtained from worst area of inflammation in each segment. Mucosal bovine Ig and TNF assessed by immunohistochemistry
11 Study Enrollment and Disposition Parameter Placebo 0.2 g/d 1.6 g/d 3.5 g/d Pooled Active Overall Enrolled Treated Completed study Reason for Early Termination (ET): Withdrew consent 1 a 1 a 1 b 2 3 Investigator opinion 1 c 1 1 A uncontrolled UC activity during Week 1 of treatment; B patient withdrew before first dosing; C recurrence of nausea and dysphagia for medications, established pre-study, precluded continued study participation
12 Demographics Parameter Overall (n = 36) Months since diagnosis, mean (SD) 87.5 (86.0) Site of disease Rectum 2.8% Left colon 50.0% Entire colon 47.2% Prior and Concomitant Meds 5-ASA 77.8% Corticosteroids AZA/ 6-MP 55.6% A 41.7% B Prior anti-tnf use (secondary failures only) 33.3% Total Mayo score, mean (SD) 8.1 (1.2) A concurrent use 36.1%; B concurrent use 30.6%
13 Treatment-Emergent Adverse Events (AEs) Parameter Placebo (n = 9) 0.2 g/d (n = 8) 1.6 g/d (n = 12) 3.5 g/d (n = 7) Pooled Active (n = 27) Overall (n = 36) All, n (%) A 7 (77.8) 3 (37.5) 6 (50.0) 5 (71.4) 14 (51.9) 26 (72.2) AEs (Total) B 13 (100.0) 8 (100.0) 13 (100.0) 13 (100.0) 34 (100.0) 47 (100.0) Mild 9 (69.2) 7 (87.5) 10 (77.0) 9 (69.2) 26 (76.5) 35 (74.5) Moderate 4 (30.8) 1 (12.5) 2 (15.9) 4 (30.8) 7 (20.6) 12 (23.4) Severe 0 (0.0) 0 (0.0) 1 (7.7) 0 (0.0) 1 (2.9) 1 (2.1) SAEs, n (%) A 0 (0.0) 0 (0.0) 1 (8.3) 0 (0.0) 1 (3.7) 1 (2.8) AEs of Special Interest C 0 (0.0) 0 (0.0) 0 (0.0) 0 (0.0) 0 (0.0) 0 (0.0) A no. of patients experiencing AEs, n (%); B no. of AEs, n (%); C allergic reaction or opportunistic infection Worsening UC on Day 3 of study drug;
14 Pharmacokinetics Minimal Levels of Bovine Ig in Serum Treatment Group # positive (%) Pre-dose (Visit 3) Post-dose (Visit 7A) Bovine Ig Bovine Ig Anti-TNF range (µg/ml) # positive (%) range (µg/ml) range (µg/ml) Placebo 0/8 (0) - 1/8 (13%) g/d 2/8 (25%) /8 (13%) g/d 0/11 (0) - 2/11 (18%) LLOQ = µg/ml 3.5 g/d 1/6 (17%) /6 (67%) Bovine Ig detectable before dosing, presumably of dietary origin Concentrations not significantly different from baseline levels and remained 1000x lower than levels associated with clinical activity
15 Bovine Ig in Colon Tissue Bovine Ig Detected by IHC in Colon Lamina Propria Placebo (105004) Pre-Dosing (502003) Post-Dosing (502003) Bovine Ig staining observed in baseline samples and after 4 weeks dosing All colon regions Every patient With or without AVX-470 dosing With or without endoscopic disease activity Submucosa stained when present
16 Bovine Ig in Stool Patients with Bovine Ig in stool Levels of Bovine Ig in stool (ng/g) % of patients Placebo AVX AVX AVX g/d 1.6 g/d 3.5 g/d Bovine Ig level, ng/g (average, SEM) Placebo AVX g/d g AVX g/d g AVX g/d g 16 All pre-dose samples were negative. Post-dosing stool samples with highest levels of bovine Ig contained anti-tnf activity.
17 Immunogenicity HABA Concentrations by Treatment Arm 40.0 Mean Percent Inhibition Pre- and Post Treatment First dose Last dose Inhibition (%) g/d 0.1 g AVX-470 BID 1.6 g/d 0.8 g AVX-470 BID 3.5 g/d 1.2 g AVX-470 TID Placebo Placebo Visit
18 Clinical and Endoscopic Remission Parameter Placebo (n = 9) 0.2 g/d (n = 8) 1.6 g/d (n = 12) 3.5 g/d (n = 7) Pooled Active (n = 27) Clinical Response 1/9 (11.1) 3/8 (37.5) 2/12 (16.7) 2/7 (28.6) 7/25 (25.9) Clinical Remission /7 (14.3) 1/27 (3.7) Endoscopic Response 0 0 1/12 (8.3) 1/7(14.3) 2/27 (7.4) Endoscopic Remission 0 0 1/12 (8.3) 1/7 (14.3) 2/27 (7.4) expressed as n/n (%)
19 UC Endoscopic Index of Severity (UCEIS) (0-8) Parameter Placebo (n = 9) 0.2 g/d (n = 8) 1.6 g/d (n = 12) 3.5 g/d (n = 7) Pooled Active (n = 27) Proximal Colon Baseline Week from Baseline A * +0.3 * p = 0.14, student t-test Descending Colon and Sigmoid (above 15 cm) Baseline Week from Baseline A A mean of individual patient changes
20 Changes in Serum CRP Across Treatment Arms and Treatment Duration Change over 4 weeks Time course of change (3.5 g/d) Change in Serum CRP (µg/ml) p = =0.052 Change in Serum CRP (µg/ml) p p = =0.058 W eek 4 W eek 5 W eek 6 W eek g/day vs placebo, paired analysis Week 4 vs baseline CRP,paired analysis
21 Changes in Serum IL-6 Across Treatment Arms 15 IL-6 Change in Serum IL-6 pg/ml p = Placebo 0.2 g/day 1.6 g/day 3.5 g/day 3.5 g/day vs placebo, paired analysis
22 Changes in Tissue TNF Across Treatment Arms Fold Change (IHC staining index) p = Placebo 0.2 g/day 1.6 g/day 3.5 g/day 3.5 g/day vs placebo, un-paired group analysis of all colon segments
23 Conclusions 1. AVX-470 was well-tolerated with no drug-related SAEs, opportunistic infections or allergic reactions. 2. AVX-470 was stable in passage through the GI tract and was not associated with significant systemic exposures. No immunogenicity was observed. 3. Prior dietary exposures interfered with detection of changes in tissue levels. However, bovine Ig was shown to penetrate the colonic mucosa, even in areas of normal endoscopic activity. 4. Efficacy trends were observed across multiple parameters (clinical, endoscopic, biomarker) of disease activity, most favoring the 3.5g/d dose group, with a proximal to distal gradient of response. 5. Future studies will examine the effects of higher doses and longer dose treatment duration.
The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only.
 The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only. Please note that the results reported in any single trial may not reflect the overall
The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only. Please note that the results reported in any single trial may not reflect the overall
Synopsis (C0168T37 ACT 1)
 () Module 5.3 Protocol: CR004777 EudraCT No.: Not Applicable Title of the study: A Randomized, Placebo-controlled, Double-blind Trial to Evaluate the Safety and Efficacy of Infliximab in Patients with
() Module 5.3 Protocol: CR004777 EudraCT No.: Not Applicable Title of the study: A Randomized, Placebo-controlled, Double-blind Trial to Evaluate the Safety and Efficacy of Infliximab in Patients with
Mucosal Healing in Crohn s Disease. Geert D Haens MD, PhD University Hospital Gasthuisberg University of Leuven Leuven, Belgium
 Mucosal Healing in Crohn s Disease Geert D Haens MD, PhD University Hospital Gasthuisberg University of Leuven Leuven, Belgium Mucosal Lesions in CD: General Features CD can affect the entire GI tract
Mucosal Healing in Crohn s Disease Geert D Haens MD, PhD University Hospital Gasthuisberg University of Leuven Leuven, Belgium Mucosal Lesions in CD: General Features CD can affect the entire GI tract
Efficacy and Safety of Treatment for Pediatric IBD
 Efficacy and Safety of Treatment for Pediatric IBD Andrew B. Grossman MD Co-Director, Center for Pediatric Inflammatory Bowel Disease Associate Professor of Clinical Pediatrics Division of Gastroenterology,
Efficacy and Safety of Treatment for Pediatric IBD Andrew B. Grossman MD Co-Director, Center for Pediatric Inflammatory Bowel Disease Associate Professor of Clinical Pediatrics Division of Gastroenterology,
Disclosures. What Do I Do When Anti-TNF Therapy Is Not Working Anymore? Fadi Hamid, M.D. Saint Luke s GI Specialists
 What Do I Do When Anti-TNF Therapy Is Not Working Anymore? Fadi Hamid, M.D. Saint Luke s GI Specialists Disclosures No financial relationships to disclose. 1 Learning Objectives Case 24M with ileocolonic
What Do I Do When Anti-TNF Therapy Is Not Working Anymore? Fadi Hamid, M.D. Saint Luke s GI Specialists Disclosures No financial relationships to disclose. 1 Learning Objectives Case 24M with ileocolonic
Synopsis. Clinical Report Synopsis for Protocol Name of Company: Otsuka Pharmaceutical Development & Commercialization, Inc.
 Synopsis Clinical Report Synopsis for Protocol 197-02-220 Name of Company: Otsuka Pharmaceutical Development & Commercialization, Inc. Name of Product: Tetomilast (OPC-6535) Study Title: A Phase 3, Multicenter,
Synopsis Clinical Report Synopsis for Protocol 197-02-220 Name of Company: Otsuka Pharmaceutical Development & Commercialization, Inc. Name of Product: Tetomilast (OPC-6535) Study Title: A Phase 3, Multicenter,
Update on Biologics in Ulcerative Colitis. Scott Plevy, MD University of North Carolina Chapel Hill, NC
 Update on Biologics in Ulcerative Colitis Scott Plevy, MD University of North Carolina Chapel Hill, NC Objectives Discuss the latest advances in the pharmacologic management of ulcerative colitis Describe
Update on Biologics in Ulcerative Colitis Scott Plevy, MD University of North Carolina Chapel Hill, NC Objectives Discuss the latest advances in the pharmacologic management of ulcerative colitis Describe
Ulcerative Colitis. ulcerative colitis usually only affects the colon.
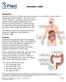 Ulcerative Colitis Introduction Ulcerative colitis is an inflammatory bowel disease. It is one of the 2 most common inflammatory bowel diseases. The other one is Crohn s disease. Ulcerative colitis and
Ulcerative Colitis Introduction Ulcerative colitis is an inflammatory bowel disease. It is one of the 2 most common inflammatory bowel diseases. The other one is Crohn s disease. Ulcerative colitis and
Efficacy and Safety of Treatment for Pediatric IBD
 Efficacy and Safety of Treatment for Pediatric IBD Andrew B. Grossman MD Co-Director, Center for Pediatric Inflammatory Bowel Disease Assistant Professor of Clinical Pediatrics Division of Gastroenterology,
Efficacy and Safety of Treatment for Pediatric IBD Andrew B. Grossman MD Co-Director, Center for Pediatric Inflammatory Bowel Disease Assistant Professor of Clinical Pediatrics Division of Gastroenterology,
Title: Author: Journal:
 IMPORTANT COPYRIGHT NOTICE: This electronic article is provided to you by courtesy of Ferring Pharmaceuticals. The document is provided for personal usage only. Further reproduction and/or distribution
IMPORTANT COPYRIGHT NOTICE: This electronic article is provided to you by courtesy of Ferring Pharmaceuticals. The document is provided for personal usage only. Further reproduction and/or distribution
Endpoints for Stopping Treatment in UC
 Endpoints for Stopping Treatment in UC Jana G. Hashash, MD Assistant Professor of Medicine Inflammatory Bowel Disease Center Division of Gastroenterology, Hepatology, and Nutrition University of Pittsburgh
Endpoints for Stopping Treatment in UC Jana G. Hashash, MD Assistant Professor of Medicine Inflammatory Bowel Disease Center Division of Gastroenterology, Hepatology, and Nutrition University of Pittsburgh
Selective leucocyte trafficking inhibitors for treatment of IBD
 Selective leucocyte trafficking inhibitors for treatment of IBD Séverine Vermeire MD, PhD Department of Gastroenterology University Hospitals Leuven Belgium Migration of Leucocytes plays a key role in
Selective leucocyte trafficking inhibitors for treatment of IBD Séverine Vermeire MD, PhD Department of Gastroenterology University Hospitals Leuven Belgium Migration of Leucocytes plays a key role in
Gionata Fiorino VEDOLIZUMAB E IBD. Un nuovo target terapeutico
 Gionata Fiorino VEDOLIZUMAB E IBD Un nuovo target terapeutico Anti cell adhesion molecules Danese S, NEJM 2011 6 Steps leukocyte recruitment Fiorino G. et al. 2010 Vedolizumab Blocks Fewer Biological Pathways
Gionata Fiorino VEDOLIZUMAB E IBD Un nuovo target terapeutico Anti cell adhesion molecules Danese S, NEJM 2011 6 Steps leukocyte recruitment Fiorino G. et al. 2010 Vedolizumab Blocks Fewer Biological Pathways
Implementation of disease and safety predictors during disease management in UC
 Implementation of disease and safety predictors during disease management in UC DR ARIELLA SHITRIT DIGESTIVE DISEASES INSTITUTE SHAARE ZEDEK MEDICAL CENTER JERUSALEM Case presentation A 52 year old male
Implementation of disease and safety predictors during disease management in UC DR ARIELLA SHITRIT DIGESTIVE DISEASES INSTITUTE SHAARE ZEDEK MEDICAL CENTER JERUSALEM Case presentation A 52 year old male
CCFA. Crohns Disease vs UC: What is the best treatment for me? November
 CCFA Crohns Disease vs UC: What is the best treatment for me? November 8 2009 Ellen J. Scherl,, MD, FACP,AGAF Roberts Inflammatory Bowel Disease Center Weill Medical College Cornell University New York
CCFA Crohns Disease vs UC: What is the best treatment for me? November 8 2009 Ellen J. Scherl,, MD, FACP,AGAF Roberts Inflammatory Bowel Disease Center Weill Medical College Cornell University New York
SER-287 Phase 1b topline study results in patients with mild-to-moderate Ulcerative Colitis October 2, 2017
 SER-287 Phase 1b topline study results in patients with mild-to-moderate Ulcerative Colitis October 2, 2017 Leading the Microbiome Revolution Forward Looking Statements Some of the statements in this presentation
SER-287 Phase 1b topline study results in patients with mild-to-moderate Ulcerative Colitis October 2, 2017 Leading the Microbiome Revolution Forward Looking Statements Some of the statements in this presentation
Treatment Goals. Current Therapeutic Pyramids Crohn s Disease Ulcerative Colitis 11/14/10
 Current Management of IBD: From Conventional Agents to Biologics Stephen B. Hanauer, M.D. University of Chicago Treatment Goals Induce and maintain response/ remission Prevent complications Improve quality
Current Management of IBD: From Conventional Agents to Biologics Stephen B. Hanauer, M.D. University of Chicago Treatment Goals Induce and maintain response/ remission Prevent complications Improve quality
Moderately to severely active ulcerative colitis
 Adalimumab in the Treatment of Moderate-to-Severe Ulcerative Colitis: ULTRA 2 Trial Results Sandborn WJ, van Assche G, Reinisch W, et al. Adalimumab induces and maintains clinical remission in patients
Adalimumab in the Treatment of Moderate-to-Severe Ulcerative Colitis: ULTRA 2 Trial Results Sandborn WJ, van Assche G, Reinisch W, et al. Adalimumab induces and maintains clinical remission in patients
SYNOPSIS. Approved Date: 9 November 2015 Prepared by: Status: Janssen Research & Development, LLC
 SYNOPSIS Name of Sponsor/Company Janssen Research & Development* Name of Investigational Product STELARA (ustekinumab) * Janssen Research & Development is a global organization that operates through different
SYNOPSIS Name of Sponsor/Company Janssen Research & Development* Name of Investigational Product STELARA (ustekinumab) * Janssen Research & Development is a global organization that operates through different
Treatment of Inflammatory Bowel Disease. Michael Weiss MD, FACG
 Treatment of Inflammatory Bowel Disease Michael Weiss MD, FACG What is IBD? IBD is an immune-mediated chronic intestinal disorder, characterized by chronic or relapsing inflammation within the GI tract.
Treatment of Inflammatory Bowel Disease Michael Weiss MD, FACG What is IBD? IBD is an immune-mediated chronic intestinal disorder, characterized by chronic or relapsing inflammation within the GI tract.
SYNOPSIS. Issue Date: 25 Oct 2011
 SYNOPSIS Issue Date: 25 Oct 2011 Name of Sponsor/Company Name of Finished Product Name of Active Ingredient(s) Janssen Research & Development STELARA Ustekinumab Protocol No.: Title of Study: Study Name:
SYNOPSIS Issue Date: 25 Oct 2011 Name of Sponsor/Company Name of Finished Product Name of Active Ingredient(s) Janssen Research & Development STELARA Ustekinumab Protocol No.: Title of Study: Study Name:
Positioning Biologics in Ulcerative Colitis
 Positioning Biologics in Ulcerative Colitis Bruce E. Sands, MD, MS Acting Chief, Gastrointestinal Unit Massachusetts General Hospital Associate Professor of Medicine Harvard Medical School Sequential Therapies
Positioning Biologics in Ulcerative Colitis Bruce E. Sands, MD, MS Acting Chief, Gastrointestinal Unit Massachusetts General Hospital Associate Professor of Medicine Harvard Medical School Sequential Therapies
Definitions. Clinical remission: Resolution of symptoms (stool frequency 3/day, no bleeding and no urgency)
 CROHN S DISEASE Definitions Clinical remission: Resolution of symptoms (stool frequency 3/day, no bleeding and no urgency) Recurrence: The reappearance of lesions after surgical resection Endoscopic remission:
CROHN S DISEASE Definitions Clinical remission: Resolution of symptoms (stool frequency 3/day, no bleeding and no urgency) Recurrence: The reappearance of lesions after surgical resection Endoscopic remission:
Azathioprine for Induction and Maintenance of Remission in Crohn s Disease
 Azathioprine for Induction and Maintenance of Remission in Crohn s Disease William J. Sandborn, MD Chief, Division of Gastroenterology Director, UCSD IBD Center Objectives Azathioprine as induction and
Azathioprine for Induction and Maintenance of Remission in Crohn s Disease William J. Sandborn, MD Chief, Division of Gastroenterology Director, UCSD IBD Center Objectives Azathioprine as induction and
Medical Therapy for Pediatric IBD: Efficacy and Safety
 Medical Therapy for Pediatric IBD: Efficacy and Safety Betsy Maxwell, MD Assistant Professor of Clinical Pediatrics Division of Gastroenterology, Hepatology, and Nutrition Pediatric IBD: Defining Remission
Medical Therapy for Pediatric IBD: Efficacy and Safety Betsy Maxwell, MD Assistant Professor of Clinical Pediatrics Division of Gastroenterology, Hepatology, and Nutrition Pediatric IBD: Defining Remission
AAPS NBC 2016 IBD Symposium
 AAPS NBC 2016 IBD Symposium Steven W Martin, PhD Group,, Pfizer, Cambridge, MA MAdCAM plays a central role in lymphocyte homing to the gut Adapted from Cheroutre and Madakamutil 2004 Anti-MAdCAM Mechanistic
AAPS NBC 2016 IBD Symposium Steven W Martin, PhD Group,, Pfizer, Cambridge, MA MAdCAM plays a central role in lymphocyte homing to the gut Adapted from Cheroutre and Madakamutil 2004 Anti-MAdCAM Mechanistic
IBD Understanding Your Medications. Thomas V. Aguirre, MD Santa Barbara GI Consultants
 IBD Understanding Your Medications Thomas V. Aguirre, MD Santa Barbara GI Consultants IBD Understanding Your Medications (& Your Doctor) Thomas V. Aguirre, MD Santa Barbara GI Consultants Disclosure I
IBD Understanding Your Medications Thomas V. Aguirre, MD Santa Barbara GI Consultants IBD Understanding Your Medications (& Your Doctor) Thomas V. Aguirre, MD Santa Barbara GI Consultants Disclosure I
Literature Review: CORE I & II: COlonic RElease Budesonide for the Induction of Remission for Mild- Moderate Ulcerative Colitis
 VOLUME 13, ISSUE 1, YEAR 2014 Literature Review: CORE I & II: COlonic RElease Budesonide for the Induction of Remission for Mild- Moderate Ulcerative Colitis Peter R. McNally, DO, MACG, MSRF Center for
VOLUME 13, ISSUE 1, YEAR 2014 Literature Review: CORE I & II: COlonic RElease Budesonide for the Induction of Remission for Mild- Moderate Ulcerative Colitis Peter R. McNally, DO, MACG, MSRF Center for
Predicting the natural history of IBD. Séverine Vermeire, MD, PhD Department of Gastroenterology University Hospital Leuven Belgium
 Predicting the natural history of IBD Séverine Vermeire, MD, PhD Department of Gastroenterology University Hospital Leuven Belgium Patient 1 Patient 2 Age 22 Frequent cramps and diarrhea for 6 months Weight
Predicting the natural history of IBD Séverine Vermeire, MD, PhD Department of Gastroenterology University Hospital Leuven Belgium Patient 1 Patient 2 Age 22 Frequent cramps and diarrhea for 6 months Weight
STELARA (ustekinumab) Clinical Study Report CNTO1275CRD3001
 SYNOPSIS Name of Sponsor/Company Janssen Research & Development* Name of Investigational Product STELARA (ustekinumab) * Janssen Research & Development is a global organization that operates through different
SYNOPSIS Name of Sponsor/Company Janssen Research & Development* Name of Investigational Product STELARA (ustekinumab) * Janssen Research & Development is a global organization that operates through different
Inflammatory bowel disease (IBD) Overview of the Paediatric investigation plans. Presented by: Richard Veselý. An agency of the European Union
 Inflammatory bowel disease (IBD) Overview of the Paediatric investigation plans Presented by: Richard Veselý An agency of the European Union Adalimumab - Crohn s disease Indication: Treatment of severe,
Inflammatory bowel disease (IBD) Overview of the Paediatric investigation plans Presented by: Richard Veselý An agency of the European Union Adalimumab - Crohn s disease Indication: Treatment of severe,
National Institute for Health and Care Excellence
 National Institute for Health and Care Excellence 4-year surveillance (2017) Ulcerative colitis: management (2013) NICE guideline CG166 Appendix A.2: Summary of new evidence from surveillance Patient information
National Institute for Health and Care Excellence 4-year surveillance (2017) Ulcerative colitis: management (2013) NICE guideline CG166 Appendix A.2: Summary of new evidence from surveillance Patient information
2. SYNOPSIS. Clinical Study Report CD-LAQ-201. November 2012
 2. SYNOPSIS Protocol No.: Study Title A Phase IIA, Multicenter, Randomized, Double-Blind, Placebo-Controlled, Sequential Cohorts, Dose Range Finding Study to Evaluate the Safety, Tolerability and Clinical
2. SYNOPSIS Protocol No.: Study Title A Phase IIA, Multicenter, Randomized, Double-Blind, Placebo-Controlled, Sequential Cohorts, Dose Range Finding Study to Evaluate the Safety, Tolerability and Clinical
U of Cape Town, South Africa, 10 U of Washington, Seattle, WA,USA, 11 CHRU de Lille, Hôpital Claude Huriez, Lille, France, 12
 A Multicenter, Double-blind, Placebo-controlled Phase 3 Study of Ustekinumab, a Human IL-12/23p40 Monoclonal Antibody, in Moderate-severe Crohn s Disease Refractory to Anti-TNFα: UNITI-1 WJ Sandborn 1,
A Multicenter, Double-blind, Placebo-controlled Phase 3 Study of Ustekinumab, a Human IL-12/23p40 Monoclonal Antibody, in Moderate-severe Crohn s Disease Refractory to Anti-TNFα: UNITI-1 WJ Sandborn 1,
Anne Griffiths MD, FRCPC. SickKids Hospital, University of Toronto. Buenos Aires, August 16, 2014
 Management and Medical Therapies for Crohn disease: strategies to enhance mucosal healing Anne Griffiths MD, FRCPC SickKids Hospital, University of Toronto Buenos Aires, August 16, 2014 New onset Crohn
Management and Medical Therapies for Crohn disease: strategies to enhance mucosal healing Anne Griffiths MD, FRCPC SickKids Hospital, University of Toronto Buenos Aires, August 16, 2014 New onset Crohn
The Best of IBD at UEGW (Crohn s)
 The Best of IBD at UEGW (Crohn s) Iyad Issa MD Head of Gastroenterology, Rafik Hariri Univ Hosp Adjunct Faculty, School of Medicine, Leb Univ Founding Faculty, School Of Medicine, Leb Am Univ 1 The Best
The Best of IBD at UEGW (Crohn s) Iyad Issa MD Head of Gastroenterology, Rafik Hariri Univ Hosp Adjunct Faculty, School of Medicine, Leb Univ Founding Faculty, School Of Medicine, Leb Am Univ 1 The Best
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION. 20 October 2010
 The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 20 October 2010 MEZAVANT LP 1200 mg, prolonged-release gastro-resistant tablets B/60 (CIP code: 378 689-2) Applicant
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 20 October 2010 MEZAVANT LP 1200 mg, prolonged-release gastro-resistant tablets B/60 (CIP code: 378 689-2) Applicant
Does Fecal Microbiota Transplantation Cause Clinical Remission in Patients with Ulcerative Colitis?
 Philadelphia College of Osteopathic Medicine DigitalCommons@PCOM PCOM Physician Assistant Studies Student Scholarship Student Dissertations, Theses and Papers 12-2017 Does Fecal Microbiota Transplantation
Philadelphia College of Osteopathic Medicine DigitalCommons@PCOM PCOM Physician Assistant Studies Student Scholarship Student Dissertations, Theses and Papers 12-2017 Does Fecal Microbiota Transplantation
Ulcerative colitis (UC) is a chronic inflammatory
 Induction and Maintenance Therapy with Vedolizumab, a Novel Biologic Therapy for Ulcerative Colitis Feagan BG, Rutgeerts P, Sands BE, et al; GEMINI 1 Study Group. Vedolizumab as induction and maintenance
Induction and Maintenance Therapy with Vedolizumab, a Novel Biologic Therapy for Ulcerative Colitis Feagan BG, Rutgeerts P, Sands BE, et al; GEMINI 1 Study Group. Vedolizumab as induction and maintenance
Biologic Therapy for Inflammatory. Is Top-Down Too Top-Heavy? S. Devi Rampertab, MD, FACG, AGAF Associate Professor of Medicine University of Florida
 Biologic Therapy for Inflammatory Bowel Disease: Is Top-Down Too Top-Heavy? S. Devi Rampertab, MD, FACG, AGAF Associate Professor of Medicine University of Florida Learning Objectives Evaluate evidence
Biologic Therapy for Inflammatory Bowel Disease: Is Top-Down Too Top-Heavy? S. Devi Rampertab, MD, FACG, AGAF Associate Professor of Medicine University of Florida Learning Objectives Evaluate evidence
How to use infliximab?
 How to use infliximab? Séverine Vermeire, MD, PhD Division of Gastroenterology University Hospital Gasthuisberg Leuven The how to use infliximab rules Before starting IFX: try optimizing chances for response!
How to use infliximab? Séverine Vermeire, MD, PhD Division of Gastroenterology University Hospital Gasthuisberg Leuven The how to use infliximab rules Before starting IFX: try optimizing chances for response!
Crohn's Disease. What causes Crohn s disease? What are the symptoms?
 Crohn's Disease Crohn s disease is an ongoing disorder that causes inflammation of the digestive tract, also referred to as the gastrointestinal (GI) tract. Crohn s disease can affect any area of the GI
Crohn's Disease Crohn s disease is an ongoing disorder that causes inflammation of the digestive tract, also referred to as the gastrointestinal (GI) tract. Crohn s disease can affect any area of the GI
Ulcerative Colitis Therapy. Faculty Disclosure. Acknowledgements 28/11/2013. Amy Morse November 30/13
 Ulcerative Colitis Therapy Amy Morse November 30/13 GI for GP s Jasper AB Faculty Disclosure Faculty: Amy Morse Relationships with commercial interests: Grants/Research Support: Therapeutic Fellowship
Ulcerative Colitis Therapy Amy Morse November 30/13 GI for GP s Jasper AB Faculty Disclosure Faculty: Amy Morse Relationships with commercial interests: Grants/Research Support: Therapeutic Fellowship
vedolizumab (Entyvio )
 vedolizumab (Entyvio ) Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ),
vedolizumab (Entyvio ) Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ),
Clinical Trials in IBD. Bruce Yacyshyn MD Professor of Medicine Division of Digestive Diseases
 Clinical Trials in IBD Bruce Yacyshyn MD Professor of Medicine Division of Digestive Diseases Objectives Today s discussion will address the following topics: Similarities and differences between Crohn
Clinical Trials in IBD Bruce Yacyshyn MD Professor of Medicine Division of Digestive Diseases Objectives Today s discussion will address the following topics: Similarities and differences between Crohn
5-ASA Therapy, Steroids and Antibiotics in Inflammatory Bowel Disease
 5-ASA Therapy, Steroids and Antibiotics in Inflammatory Bowel Disease David T. Rubin, MD Associate Professor of Medicine Co-Director, Inflammatory Bowel Disease Center University it of Chicago Medical
5-ASA Therapy, Steroids and Antibiotics in Inflammatory Bowel Disease David T. Rubin, MD Associate Professor of Medicine Co-Director, Inflammatory Bowel Disease Center University it of Chicago Medical
New treatment options in UC. Rob Bryant IBD Consultant Royal Adelaide Hospital
 New treatment options in UC Rob Bryant IBD Consultant Royal Adelaide Hospital Talk Outline 1. Raising expectations 2. Optimising UC therapy 3. Clinical trials 4. What s new on the PBS? 5. Questions 1.
New treatment options in UC Rob Bryant IBD Consultant Royal Adelaide Hospital Talk Outline 1. Raising expectations 2. Optimising UC therapy 3. Clinical trials 4. What s new on the PBS? 5. Questions 1.
Predicting response to anti - integrin therapy: long term efficacy and roles for optimisation with vedolizumab.
 Predicting response to anti - integrin therapy: long term efficacy and roles for optimisation with vedolizumab. Dr Peter Irving Guy s and St Thomas Hospital, London King s College London Response to vedolizumab
Predicting response to anti - integrin therapy: long term efficacy and roles for optimisation with vedolizumab. Dr Peter Irving Guy s and St Thomas Hospital, London King s College London Response to vedolizumab
David Y. Graham, MD Baylor College of Medicine, Houston TX, USA
 Phase III Randomized, Double Blind, Placebo- Controlled, Multicenter, Parallel Group Study to Assess the Efficacy and Safety of Add-On Fixed-Dose Anti- Mycobacterial Therapy (RHB-104) in Moderately to
Phase III Randomized, Double Blind, Placebo- Controlled, Multicenter, Parallel Group Study to Assess the Efficacy and Safety of Add-On Fixed-Dose Anti- Mycobacterial Therapy (RHB-104) in Moderately to
8. The polyp in the illustration can be described as (circle all that apply) a. Exophytic b. Pedunculated c. Sessile d. Frank
 Quiz 1 Overview 1. Beginning with the cecum, which is the correct sequence of colon subsites? a. Cecum, ascending, splenic flexure, transverse, hepatic flexure, descending, sigmoid. b. Cecum, ascending,
Quiz 1 Overview 1. Beginning with the cecum, which is the correct sequence of colon subsites? a. Cecum, ascending, splenic flexure, transverse, hepatic flexure, descending, sigmoid. b. Cecum, ascending,
Biologics in IBD. Brian P. Bosworth, MD, NYSGEF Associate Professor of Medicine Weill Cornell Medical College
 Biologics in IBD Brian P. Bosworth, MD, NYSGEF Associate Professor of Medicine Weill Cornell Medical College Case 30 year old man diagnosed with ulcerative proctitis diagnosed in 2003 Had been maintained
Biologics in IBD Brian P. Bosworth, MD, NYSGEF Associate Professor of Medicine Weill Cornell Medical College Case 30 year old man diagnosed with ulcerative proctitis diagnosed in 2003 Had been maintained
Anti tumor necrosis factor (TNF) agents have
 Achieving Clinical Response and Remission in Moderate-to-Severe Ulcerative Colitis With Golimumab Sandborn WJ, Feagan BG, Marano C, et al; PURSUIT-SC Study Group. Subcutaneous golimumab induces clinical
Achieving Clinical Response and Remission in Moderate-to-Severe Ulcerative Colitis With Golimumab Sandborn WJ, Feagan BG, Marano C, et al; PURSUIT-SC Study Group. Subcutaneous golimumab induces clinical
John F. Valentine, MD Inflammatory Bowel Disease Program University of Utah
 John F. Valentine, MD Inflammatory Bowel Disease Program University of Utah Hawaii 1/20/2017 DISCLOSURES Research Support: NIH, Pfizer, Celgene, AbbVie, Roche/Genentech, Takeda, CCFA OBJECTIVES Review
John F. Valentine, MD Inflammatory Bowel Disease Program University of Utah Hawaii 1/20/2017 DISCLOSURES Research Support: NIH, Pfizer, Celgene, AbbVie, Roche/Genentech, Takeda, CCFA OBJECTIVES Review
Positioning New Therapies
 Positioning New Therapies Stephen Hanauer, MD Professor of Medicine Medical Director, Digestive Disease Center Northwestern Medicine Chicago, Illinois Speaker Disclosure Stephen Hanauer, MD has disclosed
Positioning New Therapies Stephen Hanauer, MD Professor of Medicine Medical Director, Digestive Disease Center Northwestern Medicine Chicago, Illinois Speaker Disclosure Stephen Hanauer, MD has disclosed
Indications for use of Infliximab
 Indications for use of Infliximab Moscow, June 10 th 2006 Prof. Dr. Dr. Gerhard Rogler Klinik und Poliklinik für Innere Medizin I Universität Regensburg Case report 1989: Diagnosis of Crohn s disease of
Indications for use of Infliximab Moscow, June 10 th 2006 Prof. Dr. Dr. Gerhard Rogler Klinik und Poliklinik für Innere Medizin I Universität Regensburg Case report 1989: Diagnosis of Crohn s disease of
New and Future Adhesion Molecule Based Therapies in IBD
 New and Future Adhesion Molecule Based Therapies in IBD Brian G. Feagan Professor of Medicine, Epidemiology and Biostatistics University of Western Ontario Robarts Clinical Trials London, Ontario, Canada
New and Future Adhesion Molecule Based Therapies in IBD Brian G. Feagan Professor of Medicine, Epidemiology and Biostatistics University of Western Ontario Robarts Clinical Trials London, Ontario, Canada
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION. 3 October 2012
 The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 3 October 2012 REMICADE 100 mg, powder for concentrate for solution for infusion B/1 vial (CIP code: 562 070-1) Applicant:
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 3 October 2012 REMICADE 100 mg, powder for concentrate for solution for infusion B/1 vial (CIP code: 562 070-1) Applicant:
FERRING PHARMACEUTICALS. Enjoy The simple COR/939/2014/CH3
 Enjoy The simple pleasures of life COR/939/2014/CH3 Ulcerative colitis disrupts the simple pleasures in life Patients with ulcerative colitis may live with a considerable symptom burden despite medical
Enjoy The simple pleasures of life COR/939/2014/CH3 Ulcerative colitis disrupts the simple pleasures in life Patients with ulcerative colitis may live with a considerable symptom burden despite medical
A Case of Crohn s Disease with Mesalazine Allergy that was Difficult to Differentiate from Comorbid Ulcerative Colitis
 doi: 10.2169/internalmedicine.1607-18 http://internmed.jp CASE REPORT A Case of Crohn s Disease with Mesalazine Allergy that was Difficult to Differentiate from Comorbid Ulcerative Colitis Rumiko Tsuboi,
doi: 10.2169/internalmedicine.1607-18 http://internmed.jp CASE REPORT A Case of Crohn s Disease with Mesalazine Allergy that was Difficult to Differentiate from Comorbid Ulcerative Colitis Rumiko Tsuboi,
AESOP Overview and Inclusion/Exclusion Criteria Richard Pencek, PhD
 747-207 AESOP Overview and Inclusion/ Criteria Richard Pencek, PhD Sr Director, Clinical Research, Intercept Pharmaceuticals, Inc. 2 PSC Forum 2 AESOP: A Phase 2 Randomized, Placebo-Controlled Trial, Dose-Finding
747-207 AESOP Overview and Inclusion/ Criteria Richard Pencek, PhD Sr Director, Clinical Research, Intercept Pharmaceuticals, Inc. 2 PSC Forum 2 AESOP: A Phase 2 Randomized, Placebo-Controlled Trial, Dose-Finding
Referring to Part of Dossier: Volume: Page:
 Synopsis Abbott Laboratories Name of Study Drug: Adalimumab Name of Active Ingredient: Adalimumab Title of Study: Individual Study Table Referring to Part of Dossier: Volume: Page: (For National Authority
Synopsis Abbott Laboratories Name of Study Drug: Adalimumab Name of Active Ingredient: Adalimumab Title of Study: Individual Study Table Referring to Part of Dossier: Volume: Page: (For National Authority
Temporary Compliance Waiver Notice
 Temporary Compliance Waiver Notice At the time of initial posting on August 24th, 2012, the attached PDF document may not be fully accessible to readers using assistive technology. A fully accessible version
Temporary Compliance Waiver Notice At the time of initial posting on August 24th, 2012, the attached PDF document may not be fully accessible to readers using assistive technology. A fully accessible version
How do I choose amongst medicines for inflammatory bowel disease. Maria T. Abreu, MD
 How do I choose amongst medicines for inflammatory bowel disease Maria T. Abreu, MD Overview of IBD Pathogenesis Bacterial Products Moderately Acutely Inflamed Chronic Inflammation = IBD Normal Gut Mildly
How do I choose amongst medicines for inflammatory bowel disease Maria T. Abreu, MD Overview of IBD Pathogenesis Bacterial Products Moderately Acutely Inflamed Chronic Inflammation = IBD Normal Gut Mildly
Beyond Anti TNFs: positioning of other biologics for Crohn s disease. Christina Ha, MD Cedars Sinai Inflammatory Bowel Disease Center
 Beyond Anti TNFs: positioning of other biologics for Crohn s disease Christina Ha, MD Cedars Sinai Inflammatory Bowel Disease Center Objectives: To define high and low risk patient and disease features
Beyond Anti TNFs: positioning of other biologics for Crohn s disease Christina Ha, MD Cedars Sinai Inflammatory Bowel Disease Center Objectives: To define high and low risk patient and disease features
Biologic Therapy for Ulcerative Colitis in 2015
 5/6/215 Biologic Therapy for Ulcerative Colitis in 215 John K. Marshall MD MSc FRCPC AGAF Division of Gastroenterology McMaster University Bressler B, Marshall JK, et al. Gastroenterology 215;148: 135-58
5/6/215 Biologic Therapy for Ulcerative Colitis in 215 John K. Marshall MD MSc FRCPC AGAF Division of Gastroenterology McMaster University Bressler B, Marshall JK, et al. Gastroenterology 215;148: 135-58
INTERNATIONAL COURSE ON THE PATHOLOGY OF THE DIGESTIVE SYSTEM VICTOR BABES NATIONAL INSTITUTE OF PATHOLOGY BUCHAREST 7-8 BUCHAREST 2014
 INTERNATIONAL COURSE ON THE PATHOLOGY OF THE DIGESTIVE SYSTEM VICTOR BABES NATIONAL INSTITUTE OF PATHOLOGY BUCHAREST 7-8 BUCHAREST 2014 Endoscopic biopsy samples of naïve colitides patients: Role of basal
INTERNATIONAL COURSE ON THE PATHOLOGY OF THE DIGESTIVE SYSTEM VICTOR BABES NATIONAL INSTITUTE OF PATHOLOGY BUCHAREST 7-8 BUCHAREST 2014 Endoscopic biopsy samples of naïve colitides patients: Role of basal
Selection and use of the non-anti- TNF biological therapies: Who? When? How?
 Selection and use of the non-anti- TNF biological therapies: Who? When? How? Asher Kornbluth, MD Clinical Professor of Medicine The Henry D. Janowitz Division of Gastroenterology The Icahn School of Medicine
Selection and use of the non-anti- TNF biological therapies: Who? When? How? Asher Kornbluth, MD Clinical Professor of Medicine The Henry D. Janowitz Division of Gastroenterology The Icahn School of Medicine
Phase 2b/3 Topline Trial Results
 Phase 2b/3 Topline Trial Results RP-G28 For the Treatment of Lactose Intolerance March 2017 Forward - Looking Statement To the extent that statements contained in this presentation are not descriptions
Phase 2b/3 Topline Trial Results RP-G28 For the Treatment of Lactose Intolerance March 2017 Forward - Looking Statement To the extent that statements contained in this presentation are not descriptions
Case presentation. Eran Zittan. MD Mount Sinai Hospital, Toronto, Canada. Emek Medical Center, Afula, Israel. March, 2016
 Case presentation Eran Zittan. MD Mount Sinai Hospital, Toronto, Canada. Emek Medical Center, Afula, Israel. March, 2016 60 y/o man with long standing UC+PSC. Last 10 years on clinical and endoscopic remission.
Case presentation Eran Zittan. MD Mount Sinai Hospital, Toronto, Canada. Emek Medical Center, Afula, Israel. March, 2016 60 y/o man with long standing UC+PSC. Last 10 years on clinical and endoscopic remission.
Billing Guideline. Subject: Colorectal Cancer Screening Exams (Invasive Procedures) Effective Date: 1/1/14 Last revision effective 4/16
 Billing Guideline Subject: Colorectal Cancer Screening Exams (Invasive Procedures) Effective Date: 1/1/14 Last revision effective 4/16 Florida Hospital Care Advantage plans include full coverage of in-network
Billing Guideline Subject: Colorectal Cancer Screening Exams (Invasive Procedures) Effective Date: 1/1/14 Last revision effective 4/16 Florida Hospital Care Advantage plans include full coverage of in-network
Position of Biologics in IBD Circa 2006: Top Down vs. Step Up Therapy
 Position of Biologics in IBD Circa 2006: Top Down vs. Step Up Therapy Stephen B. Hanauer, MD University of Chicago Potential Conflicts: Centocor/Schering, Abbott, UCB, Elan, Berlex, PDL Goals of Treatment
Position of Biologics in IBD Circa 2006: Top Down vs. Step Up Therapy Stephen B. Hanauer, MD University of Chicago Potential Conflicts: Centocor/Schering, Abbott, UCB, Elan, Berlex, PDL Goals of Treatment
To help protect your privacy, PowerPoint prevented this external picture from being automatically downloaded. To download and display this picture,
 To help protect your privacy, PowerPoint prevented this external picture from being automatically downloaded. To download and display this picture, click Options in the Message Bar, and then click Enable
To help protect your privacy, PowerPoint prevented this external picture from being automatically downloaded. To download and display this picture, click Options in the Message Bar, and then click Enable
Treating to Achieve a Target and Disease Monitoring in 2015: State of the Art
 Treating to Achieve a Target and Disease Monitoring in 2015: State of the Art David T. Rubin, MD The Joseph B. Kirsner Professor of Medicine Chief, Section of Gastroenterology, Hepatology and Nutrition
Treating to Achieve a Target and Disease Monitoring in 2015: State of the Art David T. Rubin, MD The Joseph B. Kirsner Professor of Medicine Chief, Section of Gastroenterology, Hepatology and Nutrition
IBD Updates. Themes in IBD IBD management journey. New tools for therapeutic monitoring. First-line treatment in IBD
 IBD Updates Maria T. Abreu, MD University of Miami Miller School of Medicine Miami, Florida Themes in IBD 213 First-line treatment in IBD New tools for therapeutic monitoring Biologic therapy for CD and
IBD Updates Maria T. Abreu, MD University of Miami Miller School of Medicine Miami, Florida Themes in IBD 213 First-line treatment in IBD New tools for therapeutic monitoring Biologic therapy for CD and
Crohn s Disease: Should We Treat Based on Symptoms or Based on Objective Markers of Inflammation?
 Crohn s Disease: Should We Treat Based on Symptoms or Based on Objective Markers of Inflammation? Edward V. Loftus, Jr., M.D. Professor of Medicine Division of Gastroenterology and Hepatology Mayo Clinic
Crohn s Disease: Should We Treat Based on Symptoms or Based on Objective Markers of Inflammation? Edward V. Loftus, Jr., M.D. Professor of Medicine Division of Gastroenterology and Hepatology Mayo Clinic
Review article: induction therapy for patients with active ulcerative colitis
 Alimentary Pharmacology & Therapeutics Review article: induction therapy for patients with active ulcerative colitis S. P. L. TRAVIS John Radcliffe Hospital and Linacre College, Oxford, UK Correspondence
Alimentary Pharmacology & Therapeutics Review article: induction therapy for patients with active ulcerative colitis S. P. L. TRAVIS John Radcliffe Hospital and Linacre College, Oxford, UK Correspondence
An Update on the Biologic Treatment for Patients with Inflammatory Bowel Disease. David A. Schwartz, MD
 An Update on the Biologic Treatment for Patients with Inflammatory Bowel Disease David A. Schwartz, MD Director, Inflammatory Bowel Disease Center Associate Professor of Medicine Vanderbilt University
An Update on the Biologic Treatment for Patients with Inflammatory Bowel Disease David A. Schwartz, MD Director, Inflammatory Bowel Disease Center Associate Professor of Medicine Vanderbilt University
September 12, 2015 Millie D. Long MD, MPH, FACG
 Update on Biologic Therapy in 2015 September 12, 2015 Millie D. Long MD, MPH, FACG Assistant Professor of Medicine Inflammatory Bowel Disease Center University of North Carolina-Chapel Hill Outline Crohn
Update on Biologic Therapy in 2015 September 12, 2015 Millie D. Long MD, MPH, FACG Assistant Professor of Medicine Inflammatory Bowel Disease Center University of North Carolina-Chapel Hill Outline Crohn
Mild-moderate Ulcerative Colitis Sequential & Combined treatments need to be tested. Philippe Marteau, Paris, France
 Mild-moderate Ulcerative Colitis Sequential & Combined treatments need to be tested Philippe Marteau, Paris, France Sequential vs combined treatments When should one switch? Sequential vs combined treatments
Mild-moderate Ulcerative Colitis Sequential & Combined treatments need to be tested Philippe Marteau, Paris, France Sequential vs combined treatments When should one switch? Sequential vs combined treatments
Nutritional Requirements for Inflammatory Bowel Disease. Dale Lee, MD, MSCE Assistant Professor of Pediatrics University of Washington
 Nutritional Requirements for Inflammatory Bowel Disease Dale Lee, MD, MSCE Assistant Professor of Pediatrics University of Washington No disclosures Objectives 1) Review the biological mechanisms disturbed
Nutritional Requirements for Inflammatory Bowel Disease Dale Lee, MD, MSCE Assistant Professor of Pediatrics University of Washington No disclosures Objectives 1) Review the biological mechanisms disturbed
Once Daily Dosing for Induction and Maintenance of Remission in Ulcerative Colitis
 Once Daily Dosing for Induction and Maintenance of Remission in Ulcerative Colitis John K. Marshall MD MSc FRCPC AGAF Division of Gastroenterology McMaster University JKM 2014 Svartz N. Acta Med Scand
Once Daily Dosing for Induction and Maintenance of Remission in Ulcerative Colitis John K. Marshall MD MSc FRCPC AGAF Division of Gastroenterology McMaster University JKM 2014 Svartz N. Acta Med Scand
Ali Keshavarzian MD Rush University Medical Center
 Treatment: Step Up or Top Down? Ali Keshavarzian MD Rush University Medical Center Questions What medication should IBD be treated with? Can we predict which patients with IBD are high risk? Is starting
Treatment: Step Up or Top Down? Ali Keshavarzian MD Rush University Medical Center Questions What medication should IBD be treated with? Can we predict which patients with IBD are high risk? Is starting
Emerging g therapies for IBD: A practical approach to positioning. Sequential Therapies for IBD
 Emerging g therapies for IBD: A practical approach to positioning Stephen B. Hanauer, MD Sequential Therapies for IBD Disease Severity at Presentation Severe Anti-TNF +/IS Cyclosporine (UC) Colectomy (UC)
Emerging g therapies for IBD: A practical approach to positioning Stephen B. Hanauer, MD Sequential Therapies for IBD Disease Severity at Presentation Severe Anti-TNF +/IS Cyclosporine (UC) Colectomy (UC)
Use of extrapolation in small clinical trials:
 Use of extrapolation in small clinical trials: Infliximab for pediatric ulcerative colitis Jessica J. Lee, MD, MMSc Medical Officer Division of Gastroenterology and Inborn Errors Products CDER/ FDA 1 Learning
Use of extrapolation in small clinical trials: Infliximab for pediatric ulcerative colitis Jessica J. Lee, MD, MMSc Medical Officer Division of Gastroenterology and Inborn Errors Products CDER/ FDA 1 Learning
Aminosalicylates. Giovanni Barbara. Department of Medical and Surgical Sciences Alma Mater Studiorum, University of Bologna, Italy ( )
 Aminosalicylates Giovanni Barbara Department of Medical and Surgical Sciences Alma Mater Studiorum, University of Bologna, Italy (1088 2016) Colonic Diverticula, a Painless Ageing Change? 20% develop symptoms
Aminosalicylates Giovanni Barbara Department of Medical and Surgical Sciences Alma Mater Studiorum, University of Bologna, Italy (1088 2016) Colonic Diverticula, a Painless Ageing Change? 20% develop symptoms
Dr. Elmer Schabel, MD. Bundesinstitut für Arzneimittel und Medizinprodukte, Bonn, Germany (No conflicts of interest)
 EMA workshop on the development of new medicinal products for the treatment of ulcerative colitis and Crohn s disease Overview of authorised medicines for IBD in Europe - previous regulatory positions
EMA workshop on the development of new medicinal products for the treatment of ulcerative colitis and Crohn s disease Overview of authorised medicines for IBD in Europe - previous regulatory positions
TRANSPARENCY COMMITTEE
 The legally binding text is the original French version TRANSPARENCY COMMITTEE Opinion 19 February 2014 SIMPONI 50 mg, solution for injection in pre-filled pen Pre-filled B/1 pen - 0.5 ml (CIP: 34009 397
The legally binding text is the original French version TRANSPARENCY COMMITTEE Opinion 19 February 2014 SIMPONI 50 mg, solution for injection in pre-filled pen Pre-filled B/1 pen - 0.5 ml (CIP: 34009 397
How to Interpret a Clinical Trial Result
 How to Interpret a Clinical Trial Result Edward V. Loftus, Jr., M.D. Professor of Medicine Division of Gastroenterology and Hepatology Mayo Clinic College of Medicine Rochester MN CP123456-1 Are results
How to Interpret a Clinical Trial Result Edward V. Loftus, Jr., M.D. Professor of Medicine Division of Gastroenterology and Hepatology Mayo Clinic College of Medicine Rochester MN CP123456-1 Are results
How to differentiate Segmental Colitis Associated with Diverticulosis and Inflammatory Bowel Diseases?
 How to differentiate Segmental Colitis Associated with Diverticulosis and Inflammatory Bowel Diseases? Alessandro Armuzzi Lead IBD Unit Complesso Integrato Columbus Fondazione Policlinico Gemelli Università
How to differentiate Segmental Colitis Associated with Diverticulosis and Inflammatory Bowel Diseases? Alessandro Armuzzi Lead IBD Unit Complesso Integrato Columbus Fondazione Policlinico Gemelli Università
Public Assessment Report Scientific discussion. Mesalazin ESPL (mesalazine) SE/H/1654/01/DC
 Public Assessment Report Scientific discussion Mesalazin ESPL (mesalazine) SE/H/1654/01/DC This module reflects the scientific discussion for the approval of Mesalazin ESPL. The procedure was finalised
Public Assessment Report Scientific discussion Mesalazin ESPL (mesalazine) SE/H/1654/01/DC This module reflects the scientific discussion for the approval of Mesalazin ESPL. The procedure was finalised
5-ASA for the treatment of Crohn s disease DR. STEPHEN HANAUER FEINBERG SCHOOL OF MEDICINE, NORTHWESTERN UNIVERSITY, CHICAGO, IL, USA
 5-ASA for the treatment of Crohn s disease DR. STEPHEN HANAUER FEINBERG SCHOOL OF MEDICINE, NORTHWESTERN UNIVERSITY, CHICAGO, IL, USA Background RCTs investigating the efficacy of aminosalicylates for
5-ASA for the treatment of Crohn s disease DR. STEPHEN HANAUER FEINBERG SCHOOL OF MEDICINE, NORTHWESTERN UNIVERSITY, CHICAGO, IL, USA Background RCTs investigating the efficacy of aminosalicylates for
X-Plain Sigmoidoscopy Reference Summary
 X-Plain Sigmoidoscopy Reference Summary Introduction Colon diseases are common. A sigmoidoscopy is a test that can help detect colon diseases. If your doctor recommends that you have a sigmoidoscopy, the
X-Plain Sigmoidoscopy Reference Summary Introduction Colon diseases are common. A sigmoidoscopy is a test that can help detect colon diseases. If your doctor recommends that you have a sigmoidoscopy, the
Mucosal healing: does it really matter?
 Oxford Inflammatory Bowel Disease MasterClass Mucosal healing: does it really matter? Professor Jean-Frédéric Colombel, New York, USA Oxford Inflammatory Bowel Disease MasterClass Mucosal healing: does
Oxford Inflammatory Bowel Disease MasterClass Mucosal healing: does it really matter? Professor Jean-Frédéric Colombel, New York, USA Oxford Inflammatory Bowel Disease MasterClass Mucosal healing: does
SYNOPSIS (PROTOCOL WX17796)
 TITLE OF THE STUDY A prospective, randomized, double-blind, placebo-controlled, parallel group, multicenter, 52-week trial to assess the efficacy and safety of adjunct MMF to achieve remission with reduced
TITLE OF THE STUDY A prospective, randomized, double-blind, placebo-controlled, parallel group, multicenter, 52-week trial to assess the efficacy and safety of adjunct MMF to achieve remission with reduced
Ulcerative colitis (UC) is a. The Patient with Newly Diagnosed Ulcerative Colitis: Anticipating the Questions and Individualizing the Answers
 The Patient with Newly Diagnosed Ulcerative Colitis: Anticipating the Questions and Individualizing the Answers James Gregor, MD, Division of Gastroenterology, The University of Western Ontario, London,
The Patient with Newly Diagnosed Ulcerative Colitis: Anticipating the Questions and Individualizing the Answers James Gregor, MD, Division of Gastroenterology, The University of Western Ontario, London,
This clinical study synopsis is provided in line with Boehringer Ingelheim s Policy on Transparency and Publication of Clinical Study Data.
 abcd Clinical Study for Public Disclosure This clinical study synopsis is provided in line with s Policy on Transparency and Publication of Clinical Study Data. The synopsis which is part of the clinical
abcd Clinical Study for Public Disclosure This clinical study synopsis is provided in line with s Policy on Transparency and Publication of Clinical Study Data. The synopsis which is part of the clinical
Crohn s Disease: A New Approach to an Old Problem
 Management of Postoperative Crohn s Disease: A New Approach to an Old Problem Miguel Regueiro, M.D. Associate Professor of Medicine Associate Chief for Education Clinical Head and Co-Director, IBD Center
Management of Postoperative Crohn s Disease: A New Approach to an Old Problem Miguel Regueiro, M.D. Associate Professor of Medicine Associate Chief for Education Clinical Head and Co-Director, IBD Center
Eosinophilic Esophagitis. Kristine J. Krueger M.D. June 2014
 Eosinophilic Esophagitis Kristine J. Krueger M.D. June 2014 A Most Interesting Patient 36 year old self employed tree surgeon with long standing history of intermittent dysphagia and atypical GERD, NOT
Eosinophilic Esophagitis Kristine J. Krueger M.D. June 2014 A Most Interesting Patient 36 year old self employed tree surgeon with long standing history of intermittent dysphagia and atypical GERD, NOT
Colon Cancer , The Patient Education Institute, Inc. oc Last reviewed: 05/17/2017 1
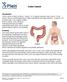 Colon Cancer Introduction Colon cancer is fairly common. About 1 in 15 people develop colon cancer. Colon cancer can be a life threatening condition that affects the large intestine. However, if it is
Colon Cancer Introduction Colon cancer is fairly common. About 1 in 15 people develop colon cancer. Colon cancer can be a life threatening condition that affects the large intestine. However, if it is
