Anesthesiology, V 92, No 1, Jan 2000
|
|
|
- Rosamund Underwood
- 6 years ago
- Views:
Transcription
1 75 Anesthesiology 2000; 92: American Society of Anesthesiologists, Inc. Lippincott Williams & Wilkins, Inc. Concentration Effect Relationship of Intravenous Lidocaine on the Allodynia of Complex Regional Pain Syndrome Types I and II Mark S. Wallace, M.D.,* Beri M. Ridgeway, B.S., Albert Y. Leung, M.D., Afshin Gerayli, M.D., Tony L. Yaksh, Ph.D. * Associate Professor. Clinical Research Associate. Research Fellow. Anesthesiology Resident. Professor. Received from the Department of Anesthesiology, University of California San Diego, San Diego, California. Submitted for publication January 19, Accepted for publication August 2, Supported by the International Anesthesia Research Society Clinical Research Scholar Grant, Cleveland, Ohio. Address reprint requests to Dr. Wallace: University of California, San Diego, 9500 Gilman Drive # 0924, La Jolla, California Address electronic mail to: mswallace@ucsd.edu Background: Several lines of evidence suggest that neuropathic pain (including Complex Regional Pain Syndrome [CRPS] I and CRPS II) is mediated in part by an increase in the density of voltage-sensitive sodium channels in injured axons and the dorsal root ganglion of injured axons. This study sought to characterize the effects of intravenous lidocaine (a sodium channel blocker) on acute sensory thresholds within the painful area and the size of the painful area in patients suffering from CRPS I and II. Methods: This study used a randomized, double-blind, placebocontrolled design in 16 subjects suffering from CRPS I and II with a prominent allodynia. Each subject received an intravenous infusion of lidocaine and diphenhydramine separated by 1 week. A computer-controlled infusion pump targeted stair-step increases in plasma levels of lidocaine of 1, 2, and 3 g/ml. At baseline and at each plasma level, spontaneous and evoked pain scores and neurosensory testing within the painful area were measured. The neurosensory testing consisted of thermal thresholds, tactile thresholds and the area of allodynia to punctate, and stroking and thermal stimuli. Results: Intravenous lidocaine and diphenhydramine had no significant effect on the cool, warm, or cold pain thresholds. However, lidocaine caused a significant elevation of the hot pain thresholds in the painful area. Intravenous lidocaine caused a significantly decreased response to stroking and cool stimuli in the allodynic area. There was also a significant decrease in pain scores to cool stimuli at all plasma levels and the spontaneous pain at the highest plasma level. Conclusions: This study demonstrates that intravenous lidocaine affects pain in response to cool stimuli more than mechanical pain in subjects with neuropathic pain. There is a lesser effect on spontaneous pain and pain induced by stroking stimuli and no effect on the pain induced by punctate stimuli. (Key words: Anesthetic; local; neuropathic.) COMPLEX Regional Pain Syndrome (CRPS) is a term used to describe a variety of painful conditions resulting from injury. Spontaneous pain or allodynia/hyperalgesia is the cardinal symptom of this syndrome. CRPS I develops after an initiating noxious event, and the pain is not limited to the territory of a single peripheral nerve. In contrast, CRPS II develops after a nerve injury with pain referred to a peripheral body region that includes, but typically extends beyond, the dermatome of the injured nerve. This pain may display several characteristics: (1) an ongoing sensation described as unpleasant or electrical shock like (dysesthesia); (2) an exaggerated pain response to a given noxious stimulus (hyperalgesia); or (3) a report of pain secondary to a nonpainful stimulus (allodynia, both thermal and mechanical). Thus, patients with CRPS I and II often report pain to cool temperatures as high as 26 C 1 and tactile stimuli that only activate low-threshold mechanoreceptive afferents. 1 5 Mechanisms underlying these anomalous sensory states are not resolved. However, as in humans, animal models of nerve injury demonstrate spontaneous pain behavior, allodynia (pain behavior secondary to light touch), or hyperalgesia (e.g., a reduced thermal escape latency). 6,7 Several lines of evidence developed in such models suggest that both the spontaneous and evoked pain is mediated in part by an increase in the density of voltage-sensitive sodium channels in the neuroma and dorsal root ganglion of the injured axon. 8 Evidence of the importance of this altered sodium channel expression to neuropathic conditions is that systemic delivery of use-dependent sodium channel blockers has no effect on acute nociceptive thresholds, but attenuates in a dose-dependent manner experimental neuropathic states.
2 76 WALLACE ET AL. Table 1. Patient Characteristics Patient No. Sex Age Diagnosis Pain Location Pain Duration (months) Cause of Pain Injured Nerve 1 F 32 RSD LLE 31 Bone fracture 2 M 23 RSD RLE 31 Bone fracture 3 M 51 RSD RLE 50 Soft tissue injury 4 F 26 RSD LUE 28 Soft tissue injury 5 F 58 RSD LUE 21 Soft tissue injury 6 F 63 RSD RUE 121 Postmastectomy 7 F 43 RSD RLE 27 Soft tissue injury 8 F 62 RSD LUE 40 Postmastectomy 9 M 38 RSD LLE 20 Bone fracture 10 M 43 Causalgia RLE 28 Surgery Posterior Tibial 11 M 50 Causalgia RUE 28 Surgery Median 12 F 46 Causalgia LLE 46 Surgery L5 nerve root 13 M 40 Causalgia RUE 128 Accident Brachial plexus 14 M 25 Causalgia LLE 7 Surgery L5 nerve root 15 M 32 Causalgia LLE 45 Surgery Ilioinguinal 16 M 74 Causalgia RLE 40 Hip fracture Sciatic Mean RSD reflex sympathetic dystrophy; LLE left lower extremity; RLE right lower extremity; LUE left upper extremity; RUE right upper extremity. Importantly, these effects occur at plasma concentrations that do not produce an afferent conduction block Comparable results have been noted in humans. Systematic studies in humans using experimental models of threshold detection have demonstrated that lidocaine has no effect on acute thermal and mechanical thresholds in normal volunteers. 12,13 However, systemic lidocaine has significant attenuating effects on the regional hyperalgesia otherwise evoked by intradermal capsaicin. 12 When examined in patients reporting significant pain secondary to a variety of neuropathic states, subanesthetic doses of systemic lidocaine produce clinically relevant relief in diabetes, 13,14 nerve injury pain states, 15,16 and late-stage cancer. 9,16,17 These results suggest that in humans, as in animals, sodium channels mediate a substantive facilitation of afferent processing after nerve injury. There are several reports in the literature on the effects of sympathetic blockade on quantitative neurosensory testing in CRPS I and II. All of these studies have demonstrated a significant reduction in pain and hyperalgesia after sympathetic blockade However, there are no studies using quantitative neurosensory testing for evaluating the effects of systemic lidocaine on this interesting syndrome. In the present study, we sought to examine the effects of lidocaine on the decreased pain threshold to cold and tactile stimuli and the size of the receptive field to which these decreased thresholds are referred in patients with CRPS I and II. An important question relates to whether these several components are similarly dependent on sodium channel activity. Accordingly, we must define the plasma concentration dependency of these measures under steady-state drug conditions. To accomplish that, we have validated a lidocaine infusion paradigm that uses pseudo steadystate kinetics to permit the patient to be exposed over a reasonable period to several plasma lidocaine concentrations. 21 Methods Subjects All work was conducted according to protocol approved by the institutional review board of the University of California, San Diego. With informed consent, 16 subjects (7 women and 9 men) suffering from CRPS I (9 patients) and CRPS II (7 patients) were recruited for the study. The mean subject age ( SD) was yr (range, yr), the mean weight was kg (range, kg), and the mean duration of pain was months (range, months). Table 1 summarizes the demographics of the cohort. After the protocol was explained and informed consent obtained, they were entered into the following experimental trials. Clinical Methodology Infusion. This study used a randomized, double-blind, placebo-controlled design. Each subject received in separate sessions an intravenous infusion of lidocaine (Astra,
3 77 EFFECT OF SYSTEMIC LIDOCAINE ON ALLODYNIA Westboro, MA) or diphenhydramine (Parke-Davis, Morris Plains, NJ). The order of the study sessions was randomized and separated by 1 week. Because intravenous lidocaine results in significant side effects that may lead to a placebo response, diphenhydramine was used as the placebo control. Previous studies have demonstrated that diphenhydramine produces side effects similar to lidocaine but does not affect sensory thresholds. 17 During each study session, two 20-gauge intravenous cannulae were inserted into the right arm: one into the antecubital vein and one into a hand or distal forearm vein. Procaine was used to anesthetize the skin to avoid interference with the lidocaine assay. The antecubital cannula was connected to a computer-controlled infusion pump (CCIP) and the other was capped with a saline flush and used to collect venous blood samples. The CCIP was programmed with the pharmacokinetic data obtained from a previous pharmacokinetic study of intravenous lidocaine in healthy volunteers. 21 In brief, parameters were estimated in those studies from the pooled analysis of arterial concentration-versus-time profiles of those volunteers for the purpose of optimizing CCIP performance. Based on those parameters, plasma lidocaine concentration steps of 1 g/ml, 2 g/ml, and 3 g/ml were targeted and maintained for 20 min. This resulted in a pseudosteady state of lidocaine, so named because we cannot assume that the lidocaine has equilibrated in all body compartments. After a baseline neurosensory test of the painful area (described in the following section), heart rate, and blood pressure, the CCIP was initiated to achieve a plasma lidocaine level of 1 g/ml and allowed to reach a pseudosteady state for 20 min. At 20 min the following procedures were performed in the order indicated: (1) blood pressure and heart rate were recorded; (2) a venous blood sample (2 ml) was collected; (3) side effects were assessed (described in the following section); (4) spontaneous and evoked pain scores were measured; and (5) neurosensory testing of the painful area was performed. After completing these tests, the process was repeated at the targeted 2- and 3- g/ml plasma lidocaine level. The infusion was stopped if the following side effects occurred (which usually occur at plasma lidocaine levels 5 g/ml) 22 : arrhythmias, nausea, tinnitus, visual hallucination, and muscle twitching. Light headedness, sedation, perioral numbness, metallic taste, and dry mouth were allowed, and the infusion was continued if these effects were reported because they usually occur at plasma lidocaine levels 5 mg/ml. 22 Three-lead electrocardiogram, heart rate, and blood pressure were monitored throughout the study. Testing. Determination of Area of Allodynia to Frey s Hair and Stroking. The region of allodynia was determined before initiating the study. The edge of the region of allodynia was established with a 5.18 Frey s hair and a cotton wisp gently stroked on the skin. These stimuli were started away from the painful area of skin and were repeated tangentially to the allodynic area at a progressively closer radius until the subject reported pain or tenderness. That site was marked on the skin with a felt-tip pen, and a new series was started from the periphery at a different angle until at least eight determinations of the borders of allodynia were outlined on the skin. These two borders were outlined onto a homunculus for area determination (in squared centimeters). The process was repeated at the completion of the infusion at the highest targeted plasma level. Neurosensory testing. Once the area of allodynia was established, three neurosensory thresholds were established in the central portion of the allodynic area: (1) warm and cool sensation; (2) hot and cold pain; and (3) touch. The same order of the stimuli was used in all subjects: cool, warm, cold pain, and hot pain. This order was chosen because it goes from the lowest stimulus (cool) to the highest stimulus (hot pain). Warm and cool sensation were measured using a Thermal Sensory Analyzer (Medoc Advanced Medical Systems, Minneapolis, MN). This device consists of a thermode measuring mm. The temperature of the thermode can either increase or decrease (at a rate of 1.0 C/s) depending on the direction of current flow through the device. The patient holds a switch that is pressed at the first sensation of warmth or cold; pressing the switch reverses the temperature change, returning to a neutral temperature of 32 C. Warm and cold pain measurements also use the Thermal Sensory Analyzer, but the end point is pain instead of temperature change sensation. It also uses a temperature change rate of 1.5 C/s. Touch was measured using Frey s hairs. Calibrated Frey s hairs are filaments of varying size. The filaments are selected at random, and three successive stimuli are applied for 2 s at 5-s intervals per filament, applied in an ascending pattern. The patient is instructed to report if the stimulus is felt. Thresholds are expressed in millinewtons and measured as positive if the patient felt any of the three successive stimuli.
4 78 WALLACE ET AL. Determination of the Area of Thermal Allodynia. Patients who reported pain at temperatures 40 C or 20 C were classified as having thermal allodynia. The area of the allodynia was established using the same method previously described for the Frey s hair and cotton wisp. The temperature used was halfway between the sensory threshold temperature and the temperature that produced pain. This border was outlined onto a homunculus for area determination (in squared centimeters). The process was repeated at the completion of the infusion at the highest targeted plasma level. Pain Measurements. Both spontaneous pain scores and evoked pain scores were measured. Pain scores were measured using a visual analog scale that consisted of a 100-mm line with no pain written at one end and the worst imaginable pain written at the other end. The subject was asked to place a mark along the line that corresponded with their pain. The distance (in millimeters) from the no-pain end to the location of the mark gives a measurement of the pain. Evoked pain was established with a 5.18 Frey s hair applied for 2 s, cotton wisp gently stroked on the skin for 2 s, and a 2 2-cm probe heated to a predetermined temperature applied for 2 s (see previous section). Side Effects. Side effects were measured by the subject using a visual analog scale that consisted of a 100-mm line with no side effect written at one end and the worst imaginable side effect written at the other end. The patient was asked to place a mark along the line that corresponded with the following side effects: sedation, nausea, light headedness, muscle twitching, tinnitus, blurred vision, perioral numbness, metallic taste, or dry mouth. The distance (in millimeters) from the noside-effect end to the location of the mark gives a measurement of the side effect. Lidocaine Assay Lidocaine was extracted from the frozen serum samples after thawing by solid-phase extraction chromatography and quantified by capillary gas chromatography with nitrogen phosphorous detection. 23 Total run time was 5 min, and lidocaine and bupivacaine eluted at 2.4 and 4.0 min, respectively. The limit of detection for lidocaine by this method was 0.05 ng/ml plasma. The interassay precision (C.V) was 7.5% and 3.5% for lidocaine levels of 0.1 ng/ml and 1.0 ng/ml in serum, respectively. Accuracy in the range of ng/ml was 99%. Table 2. Baseline Experimental Measurement Thresholds Experimental Measure Week 1 Week 2 Cool threshold ( C) Warm threshold ( C) Cold pain ( C) Hot pain ( C) Frey s detectable threshold (mn pressure) Values are mean SD. Data Analysis Data are expressed as mean SD. Baseline sensory thresholds and pain scores before lidocaine and placebo infusion were compared using a one-factor repeatedmeasure analysis of variance to ensure that no differences existed before onset of treatment. Subsequently, data for each sensory threshold measure and pain score were compared using a two-factor repeated-measures analysis of variance with both drug treatment (lidocaine vs. placebo) and titrated drug level as within-subjects factors. Allodynic areas (preadministration, postadministration) were analyzed by a paired Student t test, and, as appropriate, follow-up comparisons of individual means consisted of paired t tests, with significance held at a constant level of P 0.05 through the correction method of Bonferroni. Results Lidocaine Infusion and Plasma Lidocaine Levels The mean dose of lidocaine ( SD) infused was mg (range, mg). Based on the CCIP parameters used, measured venous lidocaine levels were considered to be close to targeted levels. Mean measured plasma levels for each targeted level (1, 2, and 3 g/ml, respectively) were 1.3 g/ml (range, g/ml), 2.4 g/ml (range, g/ml), and 3.4 g/ml (range, g/ml). Thus, the mean percent difference (concentration error [(Real Targeted)/Targeted] 100)( SD) between targeted and measured concentration at targeted plasma concentrations of 1, 2, and 3 g/ml received by each patient was 29% 46%, 21% 46%, and 16% 26%, respectively. Effect of Lidocaine on Neurosensory Thresholds Table 2 summarizes the baseline neurosensory thresholds. Thresholds were not significantly different over the 7-day interval between observations. In addition, there was no significant difference in baseline neurosensory
5 79 EFFECT OF SYSTEMIC LIDOCAINE ON ALLODYNIA Fig. 1. The effect of intravenous lidocaine at three different plasma concentrations (0 baseline; 1, 2, 3 the targeted plasma concentration in g/ml) on cool, warm, cold pain, and hot pain thermal thresholds of the painful area in patients with neuropathic pain and allodynia. *P < thresholds between the CRPS I and II subgroups. Continuous infusion of lidocaine or diphenhydramine had no statistically significant effect on cool, warm, or cold pain thresholds (fig. 1). At the highest plasma level, intravenous lidocaine caused a significant elevation of hot pain thresholds (from a mean baseline of 44.7 C to a mean of 47.9 C; fig. 1). Effect of Lidocaine on Allodynic Area and Pain Scores All of the subjects had allodynia to Frey s hair and stroking stimulation. Eight of 16 (50%) reported pain to cold stimuli, and in 5 of these patients (62.5%), it was possible to map the distribution of the cold-evoked allodynia. No patients had allodynia to heat. Mapping of the allodynic area showed that it was greatest for the Frey s hair stimulus ( cm 2 ), followed by stroking ( cm 2 ) and cold stimuli ( cm 2 ). Intravenous lidocaine resulted in a significant decrease in response to stroking and cold in the area of allodynia. This effect was greatest for cold allodynia (fig. 2). Intravenous lidocaine also resulted in a significant decrease in pain in response to cooling stimuli and in spontaneous pain (fig. 3). There was no effect of intravenous lidocaine on pain scores evoked by Frey s hair and stroking stimulation (fig. 3). Side Effects of Lidocaine Lidocaine produced significantly more light headedness than diphenhydramine. Sedation and dry mouth were similar in both groups (fig. 4). All other side effects measured were negligible in both groups. Fig. 2. The effect of intravenous lidocaine (3 g/ml plasma concentration) on the area of allodynia (in squared centimeters) to Frey s hairs, stroking, and cool stimuli in patients with neuropathic pain and allodynia. *P < Effect of Lidocaine on Blood Pressure and Heart Rate Intravenous lidocaine produced a significant increase in systolic blood pressure at the high plasma level only (from a mean baseline of mmhg to a mean of mmhg). There was no significant effect on heart rate. Fig. 3. The effect of intravenous lidocaine at three different plasma concentrations (0 baseline; 1, 2, 3 the targeted plasma concentration in g/ml) on the spontaneous pain and pain scores to Frey s hair, stroking, and cool stimuli. Pain scores were measured on a visual analog scale from 0 (no pain) to 100 (worst imaginable pain). *P < 0.05.
6 80 WALLACE ET AL. nerve fibers are being stimulated with these techniques, 30,31 the basic theories are the premises that established the model used to study the effects of intravenous lidocaine on neurosensory processing. All patients in this cohort were suffering from allodynia. Neurosensory testing in this area (compared with norms in healthy volunteers 12 ) showed grossly elevated warm, cool, and Frey s hair thresholds, modest elevation in hot pain thresholds, and normal cold pain thresholds. Because we used a small electrode for the thermal thresholds, the warm and cool thresholds may have been overestimated because these measurements are more dependent on spatial summation. 32,33 However, even taking this into account, it can still be concluded that these thresholds were elevated. Fig. 4. The side effects produced by intravenous lidocaine (3 g/ml plasma concentration) vs. diphenhydramine (70 80 mg). Side effects were scaled on a visual analog scale from 0 (no side effect) to 100 (worst imaginable side effect). *P < Discussion Description of the Study Cohort In humans, skin temperatures 33 C or 30 C will evoke an initial report of warmth or coolness, respectively. At further extremes of temperature, the subject reports the stimulus as painful, with the magnitude of the pain state being proportional to the stimulus intensity. 1 A low-intensity mechanical stimulus yields a sensation of touch, whereas higher intensities leading to physical distortion/injury will yield a report of pain, with the magnitude of the pain state being proportional to the stimulus intensity. 24 The correlation between sensation and nerve fiber activity has been extensively studied, and no definite conclusions can be made as to what nerve fibers correlate with certain sensations. Fascicular recording and compression ischemia block have shown that low-threshold tactile sensations are subserved by large myelinated fibers (A ), cool sensation by small myelinated fibers (A ), and warmth and pain by small unmyelinated fibers (C fibers). 2,4,25 29 Large myelinated fiber function can be assessed with millinewtons of pressure applied to the skin, 27 small myelinated fiber function can be assessed with quantitative thermal sensory testing, and small unmyelinated fiber function can be assessed with quantitative thermal sensory testing and mechanical pain (pressure/pinch algometer). 1 Although we cannot make a definite conclusion on what Plasma Lidocaine Levels Previous studies of intravenous lidocaine on pain states in humans have used bolus dosing of intravenous lidocaine, which results in a continuously changing plasma concentration. 10,34 To define the relationship between drug concentration and the observed effect, a technique is required that achieves and maintains a plasma concentration rapidly with minimal overshoot. The technique described in this study seems to achieve this goal. Effect of Lidocaine on Neurosensory Thresholds in Humans Although direct application of lidocaine to a nerve results in axonal conduction block, systemic delivery can exert potent effects on sensory processing at doses that do not produce conduction block. At the concentrations used in our study, we observed no prominent effects on acute thermal or mechanical thresholds. This is similar to a study in human volunteers in which it was demonstrated that there is no effect of intravenous lidocaine, in the same plasma concentrations used in the present study, on acute thermal or mechanical thresholds. 12 These findings are consistent with those of a previous report by Bach et al. 13 in a small group of normal human volunteers. Their study concluded that intravenous lidocaine, at plasma concentrations that have been shown to decrease neuropathic pain, 10,17 do not effect acute neurosensory processing in the unaltered system. Therefore, it can be concluded that systemic lidocaine exerts similar effects on acute neurosensory processing on the altered sensory system versus the unaltered sensory system. This lack of effect of intravenous lidocaine on acute thermal and mechanical stimuli seems different from several previous observations. First,
7 81 EFFECT OF SYSTEMIC LIDOCAINE ON ALLODYNIA the lack of a robust effect of intravenous lidocaine on acute neurosensory thresholds is somewhat inconsistent with preclinical findings showing a depressed conduction velocity in C fibers, and to a lesser extent A fibers, after intravenous lidocaine, although the difference suggests that these modest changes may not be relevant to detection thresholds In addition, this selective effect of lidocaine in unmyelinated fibers is supported by the observation that lidocaine decreases the flare response induced by intradermal capsaicin. We previously demonstrated a reduction in this flare response without demonstrating an effect on warm or hot pain thresholds. 12 These different C-fiber mediated events may be served by different voltage-sensitive channels in the same nociceptor membrane. 38 Therefore, the different sensitivities of these channels to systemic lidocaine may explain the differential psychophysical responses. It could also be argued that the explanation for the decrease in the flare response has nothing to do with voltage-sensitive channels, but rather is the result of vasoconstriction produced by lidocaine. In this study, we demonstrated a significant elevation of systolic blood pressure, a finding consistent with previous studies. 12 This elevation of blood pressure could be interpreted as the result of vasoconstriction from the low plasma level of lidocaine, thus resulting in a decrease in the flare response. However, recent studies using laser Doppler imaging have demonstrated no change in basal perfusion in the flare after intradermal capsaicin in the presence of systemic lidocaine (M. Schmelz, personal communication, April 1999). Another explanation is that lidocaine stabilizes the membranes of other cells (i.e., mast cells) that release peptides or vasodilator agents. Effect of Systemic Lidocaine on Mechanical and Cool Allodynia and Pain Scores The mechanism of hyperalgesia and allodynia after peripheral nerve injury is not completely understood. There are lines of evidence that static allodynia is mediated by unmyelinated fibers and dynamic allodynia is mediated by myelinated fibers. 25,38 The exact mechanism of cool allodynia is yet to be determined, because not every patient with mechanical allodynia has cool allodynia. All of the patients in this study had both static and dynamic allodynia, and only eight had cool allodynia (with only five having mappable allodynia to cool stimuli). Although there were not enough subjects with mappable allodynia to cool stimuli to make firm conclusions, the large decrease in the area of allodynia to cool stimuli coincided with a significant decrease in pain scores induced by cool stimuli. There was no consistency between the area of allodynia to mechanical stimuli and pain induced by mechanical stimuli, which suggests two different mechanism of mechanical and cool allodynia. These two mechanisms seem to have different sensitivities to intravenous lidocaine. The observation that intravenous lidocaine has a significant effect on cool and stroking allodynia and not Frey s allodynia is interesting. If both cool and mechanical allodynia are mediated by the same population of nerve fibers, one would expect systemic lidocaine to decrease the area of both. However, we demonstrated a consistent effect on cool allodynia only (both area of allodynia and evoked pain). There are several possible explanations for this observation. First, as previously mentioned, thermal and mechanical allodynia may be served by different voltage-sensitive channels in the same nociceptor membrane (on C fibers) that have different sensitivities to lidocaine. Second, a focal punctate stimulus is a higher-intensity stimulus than a diffuse nonnoxious cool stimulus, and these two stimuli may activate a different population of nociceptors with different sensitivities to systemic lidocaine. It has been demonstrated that all C-polymodal nociceptors respond to mechanical and heat stimuli, and a subgroup of these nociceptors respond to noxious low temperatures. 39 Patients with cool allodynia report pain to cool stimuli at temperatures that do not result in pain. Whether this represents a sensitization of primary C-polymodal nociceptors to low temperatures or release of an inhibitory action of myelinated fibers on second-order nociceptor neurons in the spinal cord is unclear. Third, the discrepancies in Frey s hair testing of allodynia and the area of mapping to stroking and cooling may be a result of spatial summation effects. The Frey s test may not have the sensitivity to detect minor changes. On the other hand, the thermal testing is performed with a bigger probe that may be more sensitive in detecting changes. Likewise, stroking also stimulates a larger surface area. Fourth, previous reports have shown an absence of correlation between mechanical allodynia and cool allodynia in neuropathic patients. 38,40 Our study also demonstrated this and suggests different mechanisms for mechanical and cool allodynia. The effect of systemic lidocaine on the pain scores is consistent with the aforementioned observations because systemic lidocaine had no effect on spontaneous or mechanically induced pain scores but had a significant effect on cool-induced pain scores. The lack of effect of systemic lidocaine on spontaneous pain scores and mechanical allodynia is inconsistent
8 82 WALLACE ET AL. with previous reports of lidocaine on neuropathic pain However, this can be explained by the fact that this study involved intense neurosensory testing that may have resulted in some wind-up (as demonstrated by the increase in pain scores to cooling in the placebo group; fig. 3), which masked the effects of lidocaine. In addition, most studies have shown a greater effect of systemic lidocaine in the area of allodynia than on pain scores. 12 Physiologic Effects of Lidocaine When studying the analgesic efficacy of a drug, it is important to determine if the analgesic effect is occurring below, at, or above the plasma concentration that results in known physiologic effects. To determine this, we monitored side effects, heart rate, and blood pressure. To rule out the possibility of a placebo effect of lidocaine, we used diphenhydramine as a placebo control. We chose this drug because of the prominent sedation (which is a prominent side effect of intravenous lidocaine) that usually results from its delivery. The most prominent side effects in both groups were light headedness, sedation, and dry mouth. All other side effects measured were negligible in both groups. Light headedness was the only side effect that was significantly higher in the lidocaine group than in the placebo group. Therefore, we conclude that diphenhydramine is a suitable agent for placebo-controlled studies on systemic lidocaine. These results are consistent with our previous reports. 12 Intravenous lidocaine resulted in a dose-dependent significant increase in systolic blood pressure and a modest increase in heart rate. Because lidocaine exerts an arterial vasoconstriction at plasma levels between 10 and 103 ng/ml, we assumed that an elevation in blood pressure should occur in our study. 41 In addition, plasma concentrations of local anesthetics that produce central nervous system toxicity will result in an increase in heart rate. Clinical Relevance This study demonstrates that drugs may have a specificity for certain components of the pain syndrome. By characterizing the pain, one can begin to sort out which drugs to select for a given pain characteristic. It may be possible to characterize the pain through neurosensory testing and select one or a combination of drugs to alleviate the pain. Lidocaine is an example of a drug that may be the choice for pain that has a strong cool-evoked component. Until further studies are completed with different classes of agents, we can make no further conclusions on how to select the drugs. References 1. Verdugo R, Ochoa JL: Quantitative somatosensory thermotest: A key method for functional evaluation of small calibre afferent channels. Brain 1992; 115: Yarnitsky D, Ochoa JL: Warm and cold specific somatosensory systems: Psychophysical thresholds, reaction times and peripheral conduction velocities. Brain 1991a; 114: Culp WJ, Ochoa J, Cline M, Dotson R: Heat and mechanical hyperalgesia induced by capsaicin: Cross modality threshold modulation in human C nociceptors. Brain 1989; 112: Torebjork HE, Vallbo AB, Ochoa JL: Intraneural microstimulation in man: Its relation to specificity of tactile sensations. Brain 1987; 110: Campbell JN, Raja SN, Meyer RA, Machinnon SE: Myelinated afferents signal the hyperalgesia associated with nerve injury. Pain 1988; 32: Kim SH, Chung JM: An experimental model for peripheral neuropathy produced by segmental spinal nerve ligation in the rat. Pain 1992; 50: Bennett GJ, Xie YK: A peripheral mononeuropathy in rat that produces disorders of pain sensation like those seen in man. Pain 1988; 33: Chaplan SR, Bach FW, Shafer SL, Yaksh TL: Prolonged alleviation of tactile allodynia by intravenous lidocaine in neuropathic rats. ANES- THESIOLOGY 1995; 83: Tanelian DL, Brose WG: Neuropathic pain can be relieved by drugs that are use-dependent sodium channel blockers: Lidocaine, carbamazepine, and mexiletine. ANESTHESIOLOGY 1991; 74: Boas RA, Covino BG, Shahnarian A: Analgesic responses to I.V. Lignocaine. Br J Anaesth 1982; 54: Devor M, Wall PD, Catalan N: Systemic lidocaine silences ectopic neuroma and DRG discharge without blocking nerve conduction. Pain 1992; 48: Wallace MS, Laitin S, Licht D, Yaksh TL: Concentration-effect relations for intravenous lidocaine infusions in huma volunteers. ANES- THESIOLOGY 1997; 86: Bach FW, Jensen TS, Kastrup J, Stigsby B, Dejgard A: The effect of intravenous lidocaine on nociceptive processing in diabetic neuropathy. Pain 1990; 40: Kastrup J, Petersen P, Dejgard A, Angeo HR, Hilsted J: Intravenous lidocaine infusion: A new treatment of chronic painful diabetic neuropathy? Pain 1987; 28: Marchettini P, Lacerenza M, Marangoni C, Pellegata G, Sotgui ML, Smirne S: Lidocaine test in neuralgia. Pain 1992; 48: Rowbotham MC, Reisner-Keller LA, Fields HL: Both intravenous lidocaine and morphine reduce the pain of postherpetic neuralgia. Neurology 1991; 41: Wallace MS, Dyck JB, Rossi SS, Yaksh TL: Computer controlled lidocaine infusion for the evaluation of neuropathic pain after peripheral nerve injury. Pain 1996; 66: Price DD, Bennett GJ, Rafii A: Psychophysical observations on patients with neuropathic pain relieved by a sympathetic block. Pain 1989; 36: Treede RD, Davis KD, Campbell JN, Raja SN: The plasticity of
9 83 EFFECT OF SYSTEMIC LIDOCAINE ON ALLODYNIA cutaneous hyperalgesia during sympathetic ganglion blockade in patients with neuropathic pain. Brain 1992; 115: Wahren LK, Torebjork E: Quantitative sensory tests in patients with neuralgia 11 to 25 years after injury. Pain 1992; 48: Dyck JB, Wallace MS, Lu JQ, Rossi SS, Yaksh TL: Pharmacokinetics of lidocaine administered by computer-controlled infusion pump in humans. Eur J Pain 1997; 1: Covino BG: Clinical pharmacology of local anesthetic agents, Neural Blockade in Clinical Anesthesia and Management of Pain, 2nd Edition. Edited by Cousins MJ, Bridenbaugh PO. Philadelphia, Lippincott, 1988, pp Chen Y, Pueetr JM, Ravenscroft PS: A quick, sensitive high performance liquid chromatography assay for monethyl-glycin-exylidide and lignocaine in serum plasma using solid-phase extraction. Ther Drug Monit 1992; 14: Raja SN, Meyer RA, Campbell JN: Peripheral mechanisms of somatic pain. ANESTHESIOLOGY 1988; 68: Yarnitsky D, Ochoa JL: Release of cold-induced burning pain by block of cold-specific afferent input. Brain 1990; 113: Yarnitsky D, Ochoa JL: Differential effect of compression-ischemia block on warm sensation and heat-induced pain. Brain 1991b; 114: Gruener G, Dyck PJ: Quantitative sensory testing: Methodology, applications, and future directions. J Clin Neurophys 1994; 11: Ochoa JL, Torebjork E: Sensations evoked by intraneural microstimulation of C nociceptor fibres in human skin nerves. J Physiol 1989; 415: Ochoa JL, Torebjork E: Sensations evoked by intraneural microstimulation of single mechanoreceptor units innervating the human hand. J Physiol 1983; 342: Konietzney F: Peripheral neural correlates of temperature sensation in man. Hum Neurobiol 1984; 3: Nordin N: Low-threshold mechanoreceptive and nociceptive units with unmyelinated C fibers in the human supraorbital nerve. J Physiol 1990; 426: Defrin R, Urca G: Spatial summation of heat pain: A reassessment. Pain 1996; 66: Dyck PJ, O Brien PC, Kosanke JL, Gillen DA, Karnes JL: A 4, 2, and 1 stepping algorithm for quick and accurate estimation of cutaneous sensation threshold. Neurology 1993; 43: Rowlingson JC, DiFazio CA, Foster J, Carron H: Lidocaine as an analgesic for experimental pain. ANESTHESIOLOGY 1980; 52: Woolf CJ: Somatic pain-pathogenesis and prevention. Br J Anaesth 1995; 75: Tanelian DL, MacIver MB: Analgesic concentrations of lidocaine suppress tonic A-delta and C fiber discharges produced by acute injury. ANESTHESIOLOGY 1991; 74: DeJong RH, Nace RA: Nerve impulse conduction during intravenous lidocaine injection. ANESTHESIOLOGY 1968; 29: Ochoa JL, Yarnitsky D: Mechanical hyperalgesias in neuropathic pain patients: Dynamic and static subtypes. Ann Neurol 1993; 33: Campero M, Serra J, Ochoa JL: C-polymodal nociceptors activated by noxious low temperature in human skin. J Physiol 1996; 497: Lindblom U, Verrillo RT: Sensory functions in chronic neuralgia. J Neurol Neurosurg Psychiatry 1979; 42: Blair MR: Cardiovascular pharmacology of local anesthetics. Br J Anaesth 1975; 47:247 52
Differential effects of systemically administered ketamine and lidocaine on dynamic and static hyperalgesia induced by intradermal capsaicin in humans
 British Journal of Anaesthesia 84 (2): 155 62 (2000) Differential effects of systemically administered ketamine and lidocaine on dynamic and static hyperalgesia induced by intradermal capsaicin in humans
British Journal of Anaesthesia 84 (2): 155 62 (2000) Differential effects of systemically administered ketamine and lidocaine on dynamic and static hyperalgesia induced by intradermal capsaicin in humans
Sensory Assessment of Regional Analgesia in Humans
 REVIEW ARTICLE Dennis M. Fisher, M.D., Editor-in-Chief Anesthesiology 2000; 93:1517 30 2000 American Society of Anesthesiologists, Inc. Lippincott Williams & Wilkins, Inc. Sensory Assessment of Regional
REVIEW ARTICLE Dennis M. Fisher, M.D., Editor-in-Chief Anesthesiology 2000; 93:1517 30 2000 American Society of Anesthesiologists, Inc. Lippincott Williams & Wilkins, Inc. Sensory Assessment of Regional
Lidocaine inhibits neurite growth in mouse dorsal root ganglion cells in culture
 Lidocaine inhibits neurite growth in mouse dorsal root ganglion cells in culture 3 Hiromi ~iruma', Hiroshi ~ aru~ama~, Zyun'ici B. Simada, Takashi Katakural, Sumio ~ oka~, Toshifumi ~akenaka~ and Tadashi
Lidocaine inhibits neurite growth in mouse dorsal root ganglion cells in culture 3 Hiromi ~iruma', Hiroshi ~ aru~ama~, Zyun'ici B. Simada, Takashi Katakural, Sumio ~ oka~, Toshifumi ~akenaka~ and Tadashi
A Review of Neuropathic Pain: From Diagnostic Tests to Mechanisms
 DOI 10.1007/s40122-017-0085-2 REVIEW A Review of Neuropathic Pain: From Diagnostic Tests to Mechanisms Andrea Truini Received: September 19, 2017 Ó The Author(s) 2017. This article is an open access publication
DOI 10.1007/s40122-017-0085-2 REVIEW A Review of Neuropathic Pain: From Diagnostic Tests to Mechanisms Andrea Truini Received: September 19, 2017 Ó The Author(s) 2017. This article is an open access publication
Spinal Cord Injury Pain. Michael Massey, DO CentraCare Health St Cloud, MN 11/07/2018
 Spinal Cord Injury Pain Michael Massey, DO CentraCare Health St Cloud, MN 11/07/2018 Objectives At the conclusion of this session, participants should be able to: 1. Understand the difference between nociceptive
Spinal Cord Injury Pain Michael Massey, DO CentraCare Health St Cloud, MN 11/07/2018 Objectives At the conclusion of this session, participants should be able to: 1. Understand the difference between nociceptive
Somatic Sensation (MCB160 Lecture by Mu-ming Poo, Friday March 9, 2007)
 Somatic Sensation (MCB160 Lecture by Mu-ming Poo, Friday March 9, 2007) Introduction Adrian s work on sensory coding Spinal cord and dorsal root ganglia Four somatic sense modalities Touch Mechanoreceptors
Somatic Sensation (MCB160 Lecture by Mu-ming Poo, Friday March 9, 2007) Introduction Adrian s work on sensory coding Spinal cord and dorsal root ganglia Four somatic sense modalities Touch Mechanoreceptors
Corporate Medical Policy
 Corporate Medical Policy Intravenous Anesthetics for the Treatment of Chronic Pain File Name: Origination: Last CAP Review: Next CAP Review: Last Review: intravenous_anesthetics_for_the_treatment_of_chronic_pain
Corporate Medical Policy Intravenous Anesthetics for the Treatment of Chronic Pain File Name: Origination: Last CAP Review: Next CAP Review: Last Review: intravenous_anesthetics_for_the_treatment_of_chronic_pain
Coding of Sensory Information
 Coding of Sensory Information 22 November, 2016 Touqeer Ahmed PhD Atta-ur-Rahman School of Applied Biosciences National University of Sciences and Technology Sensory Systems Mediate Four Attributes of
Coding of Sensory Information 22 November, 2016 Touqeer Ahmed PhD Atta-ur-Rahman School of Applied Biosciences National University of Sciences and Technology Sensory Systems Mediate Four Attributes of
Intravenous Lidocaine for Neuropathic Pain: Diagnostic Utility and Therapeutic Efficacy
 Intravenous Lidocaine for Neuropathic Pain: Diagnostic Utility and Therapeutic Efficacy Ian Carroll, MD, MS Corresponding author Ian Carroll, MD, MS Stanford University School of Medicine, 780 Welch Road,
Intravenous Lidocaine for Neuropathic Pain: Diagnostic Utility and Therapeutic Efficacy Ian Carroll, MD, MS Corresponding author Ian Carroll, MD, MS Stanford University School of Medicine, 780 Welch Road,
211MDS Pain theories
 211MDS Pain theories Definition In 1986, the International Association for the Study of Pain (IASP) defined pain as a sensory and emotional experience associated with real or potential injuries, or described
211MDS Pain theories Definition In 1986, the International Association for the Study of Pain (IASP) defined pain as a sensory and emotional experience associated with real or potential injuries, or described
Somatosensory modalities!
 Somatosensory modalities! The somatosensory system codes five major sensory modalities:! 1. Discriminative touch! 2. Proprioception (body position and motion)! 3. Nociception (pain and itch)! 4. Temperature!
Somatosensory modalities! The somatosensory system codes five major sensory modalities:! 1. Discriminative touch! 2. Proprioception (body position and motion)! 3. Nociception (pain and itch)! 4. Temperature!
Sensory coding and somatosensory system
 Sensory coding and somatosensory system Sensation and perception Perception is the internal construction of sensation. Perception depends on the individual experience. Three common steps in all senses
Sensory coding and somatosensory system Sensation and perception Perception is the internal construction of sensation. Perception depends on the individual experience. Three common steps in all senses
Anesthesiology, V 92, No 3, Mar 2000
 691 Anesthesiology 2000; 92:691 8 2000 American Society of Anesthesiologists, Inc. Lippincott Williams & Wilkins, Inc. Heterogenous Patterns of Sensory Dysfunction in Postherpetic Neuralgia Suggest Multiple
691 Anesthesiology 2000; 92:691 8 2000 American Society of Anesthesiologists, Inc. Lippincott Williams & Wilkins, Inc. Heterogenous Patterns of Sensory Dysfunction in Postherpetic Neuralgia Suggest Multiple
A role for uninjured afferents in neuropathic pain
 Acta Physiologica Sinica, October 25, 2008, 60 (5): 605-609 http://www.actaps.com.cn 605 Review A role for uninjured afferents in neuropathic pain Richard A. Meyer 1,2,3,*, Matthias Ringkamp 1 Departments
Acta Physiologica Sinica, October 25, 2008, 60 (5): 605-609 http://www.actaps.com.cn 605 Review A role for uninjured afferents in neuropathic pain Richard A. Meyer 1,2,3,*, Matthias Ringkamp 1 Departments
Bi/CNS/NB 150: Neuroscience. November 11, 2015 SOMATOSENSORY SYSTEM. Ralph Adolphs
 Bi/CNS/NB 150: Neuroscience November 11, 2015 SOMATOSENSORY SYSTEM Ralph Adolphs 1 Menu for today Touch -peripheral -central -plasticity Pain 2 Sherrington (1948): senses classified as --teloreceptive
Bi/CNS/NB 150: Neuroscience November 11, 2015 SOMATOSENSORY SYSTEM Ralph Adolphs 1 Menu for today Touch -peripheral -central -plasticity Pain 2 Sherrington (1948): senses classified as --teloreceptive
Somatosensation. Recording somatosensory responses. Receptive field response to pressure
 Somatosensation Mechanoreceptors that respond to touch/pressure on the surface of the body. Sensory nerve responds propotional to pressure 4 types of mechanoreceptors: Meissner corpuscles & Merkel discs
Somatosensation Mechanoreceptors that respond to touch/pressure on the surface of the body. Sensory nerve responds propotional to pressure 4 types of mechanoreceptors: Meissner corpuscles & Merkel discs
Update on the Neurophysiology of Pain Transmission and Modulation: Focus on the NMDA-Receptor
 S2 Journal of Pain and Symptom Management Vol. 19 No. 1(Suppl.) January 2000 Proceedings Supplement NMDA-Receptor Antagonists: Evolving Role in Analgesia Update on the Neurophysiology of Pain Transmission
S2 Journal of Pain and Symptom Management Vol. 19 No. 1(Suppl.) January 2000 Proceedings Supplement NMDA-Receptor Antagonists: Evolving Role in Analgesia Update on the Neurophysiology of Pain Transmission
Pain teaching. Muhammad Laklouk
 Pain teaching Muhammad Laklouk Definition Pain An unpleasant sensory and emotional experience associated with actual or potential tissue damage or described in terms of such damage. Sensory (discriminatiory)
Pain teaching Muhammad Laklouk Definition Pain An unpleasant sensory and emotional experience associated with actual or potential tissue damage or described in terms of such damage. Sensory (discriminatiory)
CHAPTER 10 THE SOMATOSENSORY SYSTEM
 CHAPTER 10 THE SOMATOSENSORY SYSTEM 10.1. SOMATOSENSORY MODALITIES "Somatosensory" is really a catch-all term to designate senses other than vision, hearing, balance, taste and smell. Receptors that could
CHAPTER 10 THE SOMATOSENSORY SYSTEM 10.1. SOMATOSENSORY MODALITIES "Somatosensory" is really a catch-all term to designate senses other than vision, hearing, balance, taste and smell. Receptors that could
Intravenous Lidocaine for Neuropathic Pain: A Retrospective Analysis of Tolerability and Efficacy
 Pain Medicine 2014; 2015; : 16: 531 536 Wiley Periodicals, Inc. NEUROPATHIC Intravenous Lidocaine PAIN SECTION for Neuropathic Pain: A Retrospective Analysis of Tolerability Brief and Efficacy Research
Pain Medicine 2014; 2015; : 16: 531 536 Wiley Periodicals, Inc. NEUROPATHIC Intravenous Lidocaine PAIN SECTION for Neuropathic Pain: A Retrospective Analysis of Tolerability Brief and Efficacy Research
San Francisco Chronicle, June 2001
 PAIN San Francisco Chronicle, June 2001 CONGENITAL INSENSITIVITY TO PAIN PAIN IS A SUBJECTIVE EXPERIENCE: It is not a stimulus MAJOR FEATURES OF THE PAIN EXPERIENCE: Sensory discriminative Affective (emotional)
PAIN San Francisco Chronicle, June 2001 CONGENITAL INSENSITIVITY TO PAIN PAIN IS A SUBJECTIVE EXPERIENCE: It is not a stimulus MAJOR FEATURES OF THE PAIN EXPERIENCE: Sensory discriminative Affective (emotional)
Prof Wayne Derman MBChB,BSc (Med)(Hons) PhD, FFIMS. Pain Management in the Elite Athlete: The 2017 IOC Consensus Statement
 Prof Wayne Derman MBChB,BSc (Med)(Hons) PhD, FFIMS Pain Management in the Elite Athlete: The 2017 IOC Consensus Statement 2 as 20 Experts published and leaders in their respective field 12 month lead in
Prof Wayne Derman MBChB,BSc (Med)(Hons) PhD, FFIMS Pain Management in the Elite Athlete: The 2017 IOC Consensus Statement 2 as 20 Experts published and leaders in their respective field 12 month lead in
PAIN IS A SUBJECTIVE EXPERIENCE: It is not a stimulus. MAJOR FEATURES OF THE PAIN EXPERIENCE: Sensory discriminative Affective (emotional) Cognitive
 PAIN PAIN IS A SUBJECTIVE EXPERIENCE: It is not a stimulus MAJOR FEATURES OF THE PAIN EXPERIENCE: Sensory discriminative Affective (emotional) Cognitive MEASUREMENT OF PAIN: A BIG PROBLEM Worst pain ever
PAIN PAIN IS A SUBJECTIVE EXPERIENCE: It is not a stimulus MAJOR FEATURES OF THE PAIN EXPERIENCE: Sensory discriminative Affective (emotional) Cognitive MEASUREMENT OF PAIN: A BIG PROBLEM Worst pain ever
How strong is it? What is it? Where is it? What must sensory systems encode? 9/8/2010. Spatial Coding: Receptive Fields and Tactile Discrimination
 Spatial Coding: Receptive Fields and Tactile Discrimination What must sensory systems encode? How strong is it? What is it? Where is it? When the brain wants to keep certain types of information distinct,
Spatial Coding: Receptive Fields and Tactile Discrimination What must sensory systems encode? How strong is it? What is it? Where is it? When the brain wants to keep certain types of information distinct,
Spatial Coding: Receptive Fields and Tactile Discrimination
 Spatial Coding: Receptive Fields and Tactile Discrimination What must sensory systems encode? How strong is it? What is it? Where is it? When the brain wants to keep certain types of information distinct,
Spatial Coding: Receptive Fields and Tactile Discrimination What must sensory systems encode? How strong is it? What is it? Where is it? When the brain wants to keep certain types of information distinct,
ANAT2010. Concepts of Neuroanatomy (II) S2 2018
 ANAT2010 Concepts of Neuroanatomy (II) S2 2018 Table of Contents Lecture 13: Pain and perception... 3 Lecture 14: Sensory systems and visual pathways... 11 Lecture 15: Techniques in Neuroanatomy I in vivo
ANAT2010 Concepts of Neuroanatomy (II) S2 2018 Table of Contents Lecture 13: Pain and perception... 3 Lecture 14: Sensory systems and visual pathways... 11 Lecture 15: Techniques in Neuroanatomy I in vivo
MANAGEMENT OF CRPS. Brachial Neuralgia n = 81 patients. CRPS type II n = 126 patients CONTENTS. Neuropathic Pain. Introduction.
 MANAGEMENT OF CRPS Paradoxical Hypoaesthetic Skin, Painful to Touch: A Target to relieve Neuropathic Pain Spicher, C.J. et al. (2010) Paris: Sauramps medical Paradoxical Hypoaesthetic Skin, Painful to
MANAGEMENT OF CRPS Paradoxical Hypoaesthetic Skin, Painful to Touch: A Target to relieve Neuropathic Pain Spicher, C.J. et al. (2010) Paris: Sauramps medical Paradoxical Hypoaesthetic Skin, Painful to
Somatic Sensory System I. Background
 Somatic Sensory System I. Background A. Differences between somatic senses and other senses 1. Receptors are distributed throughout the body as opposed to being concentrated at small, specialized locations
Somatic Sensory System I. Background A. Differences between somatic senses and other senses 1. Receptors are distributed throughout the body as opposed to being concentrated at small, specialized locations
SOMATOSENSORY SYSTEMS
 SOMATOSENSORY SYSTEMS Schematic diagram illustrating the neural pathways that convey somatosensory information to the cortex and, subsequently, to the motor system. Double arrows show reciprocal connections.
SOMATOSENSORY SYSTEMS Schematic diagram illustrating the neural pathways that convey somatosensory information to the cortex and, subsequently, to the motor system. Double arrows show reciprocal connections.
TRANSCUTANEOUS ELECTRICAL STIMULATION
 TRANSCUTANEOUS ELECTRICAL STIMULATION Transcutaneous electrical stimulation (TENS) Transcutaneous electrical stimulation ; An electronic device that produces electrical signals used to stimulate nerve
TRANSCUTANEOUS ELECTRICAL STIMULATION Transcutaneous electrical stimulation (TENS) Transcutaneous electrical stimulation ; An electronic device that produces electrical signals used to stimulate nerve
Pain Pathways. Dr Sameer Gupta Consultant in Anaesthesia and Pain Management, NGH
 Pain Pathways Dr Sameer Gupta Consultant in Anaesthesia and Pain Management, NGH Objective To give you a simplistic and basic concepts of pain pathways to help understand the complex issue of pain Pain
Pain Pathways Dr Sameer Gupta Consultant in Anaesthesia and Pain Management, NGH Objective To give you a simplistic and basic concepts of pain pathways to help understand the complex issue of pain Pain
Dose Response of Intrathecal Adenosine in Experimental Pain and Allodynia James C. Eisenach, M.D.,* Regina Curry, R.N., David D. Hood, M.D.
 Anesthesiology 2002; 97:938 42 2002 American Society of Anesthesiologists, Inc. Lippincott Williams & Wilkins, Inc. Dose Response of Intrathecal Adenosine in Experimental Pain and Allodynia James C. Eisenach,
Anesthesiology 2002; 97:938 42 2002 American Society of Anesthesiologists, Inc. Lippincott Williams & Wilkins, Inc. Dose Response of Intrathecal Adenosine in Experimental Pain and Allodynia James C. Eisenach,
Mechanism of Pain Production
 Mechanism of Pain Production Pain conducting nerve fibers are small myelinated (A-delta) or unmyelinated nerve fibers (C-fibers). Cell bodies are in the dorsal root ganglia (DRG) or sensory ganglia of
Mechanism of Pain Production Pain conducting nerve fibers are small myelinated (A-delta) or unmyelinated nerve fibers (C-fibers). Cell bodies are in the dorsal root ganglia (DRG) or sensory ganglia of
Neuronal Circuits and Neuronal Pools
 Neuronal Circuits and Neuronal Pools Anatomical Orientation Clinical Orientation Atlas Fig. 6-24 The Reality of the Clinical Environment Atlas Fig. 6-24A One Cardinal Plane to Learn AND Understand
Neuronal Circuits and Neuronal Pools Anatomical Orientation Clinical Orientation Atlas Fig. 6-24 The Reality of the Clinical Environment Atlas Fig. 6-24A One Cardinal Plane to Learn AND Understand
Pain. Types of Pain. Types of Pain 8/21/2013
 Pain 1 Types of Pain Acute Pain Complex combination of sensory, perceptual, & emotional experiences as a result of a noxious stimulus Mediated by rapidly conducting nerve pathways & associated with increased
Pain 1 Types of Pain Acute Pain Complex combination of sensory, perceptual, & emotional experiences as a result of a noxious stimulus Mediated by rapidly conducting nerve pathways & associated with increased
Seizure: the clinical manifestation of an abnormal and excessive excitation and synchronization of a population of cortical
 Are There Sharing Mechanisms of Epilepsy, Migraine and Neuropathic Pain? Chin-Wei Huang, MD, PhD Department of Neurology, NCKUH Basic mechanisms underlying seizures and epilepsy Seizure: the clinical manifestation
Are There Sharing Mechanisms of Epilepsy, Migraine and Neuropathic Pain? Chin-Wei Huang, MD, PhD Department of Neurology, NCKUH Basic mechanisms underlying seizures and epilepsy Seizure: the clinical manifestation
Brian Kahan, D.O. FAAPMR, DABPM, DAOCRM, FIPP Center for Pain Medicine and Physiatric Rehabilitation 2002 Medical Parkway Suite 150 Annapolis, MD
 Brian Kahan, D.O. FAAPMR, DABPM, DAOCRM, FIPP Center for Pain Medicine and Physiatric Rehabilitation 2002 Medical Parkway Suite 150 Annapolis, MD 1630 Main Street Suite 215 Chester, MD 410-571-9000 www.4-no-pain.com
Brian Kahan, D.O. FAAPMR, DABPM, DAOCRM, FIPP Center for Pain Medicine and Physiatric Rehabilitation 2002 Medical Parkway Suite 150 Annapolis, MD 1630 Main Street Suite 215 Chester, MD 410-571-9000 www.4-no-pain.com
The Sympathetic Nervous System Contributes to Capsaicin-Evoked Mechanical Allodynia But Not Pinprick Hyperalgesia in Humans
 The Journal of Neuroscience, November 15, 1996, 16(22):7331 7335 The Sympathetic Nervous System Contributes to Capsaicin-Evoked Mechanical Allodynia But Not Pinprick Hyperalgesia in Humans Maywin Liu,
The Journal of Neuroscience, November 15, 1996, 16(22):7331 7335 The Sympathetic Nervous System Contributes to Capsaicin-Evoked Mechanical Allodynia But Not Pinprick Hyperalgesia in Humans Maywin Liu,
Somatosensory System. Steven McLoon Department of Neuroscience University of Minnesota
 Somatosensory System Steven McLoon Department of Neuroscience University of Minnesota 1 Course News Dr. Riedl s review session this week: Tuesday (Oct 10) 4-5pm in MCB 3-146B 2 Sensory Systems Sensory
Somatosensory System Steven McLoon Department of Neuroscience University of Minnesota 1 Course News Dr. Riedl s review session this week: Tuesday (Oct 10) 4-5pm in MCB 3-146B 2 Sensory Systems Sensory
Is Nociceptor Activation by Alpha-1 Adrenoreceptors the Culprit in Sympathetically Maintained Pain?
 Focus Is Nociceptor Activation by Alpha-1 Adrenoreceptors the Culprit in Sympathetically Maintained Pain? James N. Campbell, *t Richard A. Meyer, *t and Srinivasa N. Raja_~ In certain patients, pain depends
Focus Is Nociceptor Activation by Alpha-1 Adrenoreceptors the Culprit in Sympathetically Maintained Pain? James N. Campbell, *t Richard A. Meyer, *t and Srinivasa N. Raja_~ In certain patients, pain depends
The biochemical origin of pain: The origin of all pain is inflammation and the inflammatory response: Inflammatory profile of pain syndromes
 The biochemical origin of pain: The origin of all pain is inflammation and the inflammatory response: Inflammatory profile of pain syndromes 1 Medical Hypothesis 2007, Vol. 69, pp. 1169 1178 Sota Omoigui
The biochemical origin of pain: The origin of all pain is inflammation and the inflammatory response: Inflammatory profile of pain syndromes 1 Medical Hypothesis 2007, Vol. 69, pp. 1169 1178 Sota Omoigui
Pain and Touch. Academic Press. Edited by Lawrence Kruger. Department of Neurobiology University of California, Los Angeles Los Angeles, California
 Pain and Touch Edited by Lawrence Kruger Department of Neurobiology University of California, Los Angeles Los Angeles, California San Diego New York Sydney Academic Press London Boston Tokyo Toronto Contributors
Pain and Touch Edited by Lawrence Kruger Department of Neurobiology University of California, Los Angeles Los Angeles, California San Diego New York Sydney Academic Press London Boston Tokyo Toronto Contributors
ISPUB.COM. Lumbar Sympathectomy by Laser Technique. S Kantha, B Kantha METHODS AND MATERIALS
 ISPUB.COM The Internet Journal of Minimally Invasive Spinal Technology Volume 1 Number 2 Lumbar Sympathectomy by Laser Technique S Kantha, B Kantha Citation S Kantha, B Kantha. Lumbar Sympathectomy by
ISPUB.COM The Internet Journal of Minimally Invasive Spinal Technology Volume 1 Number 2 Lumbar Sympathectomy by Laser Technique S Kantha, B Kantha Citation S Kantha, B Kantha. Lumbar Sympathectomy by
Neural Integration I: Sensory Pathways and the Somatic Nervous System
 15 Neural Integration I: Sensory Pathways and the Somatic Nervous System PowerPoint Lecture Presentations prepared by Jason LaPres Lone Star College North Harris An Introduction to Sensory Pathways and
15 Neural Integration I: Sensory Pathways and the Somatic Nervous System PowerPoint Lecture Presentations prepared by Jason LaPres Lone Star College North Harris An Introduction to Sensory Pathways and
NORLAND AVENUE PHARMACY PRESCRIPTION COMPOUNDING FOR PAIN MANAGEMENT
 NOVEMBER 2011 NORLAND AVENUE PHARMACY PRESCRIPTION COMPOUNDING N ORLANDA VENUEP HARMACY. COM We customize individual prescriptions for the specific needs of our patients. INSIDE THIS ISSUE: Sciatic Pain
NOVEMBER 2011 NORLAND AVENUE PHARMACY PRESCRIPTION COMPOUNDING N ORLANDA VENUEP HARMACY. COM We customize individual prescriptions for the specific needs of our patients. INSIDE THIS ISSUE: Sciatic Pain
By the end of this lecture the students will be able to:
 UNIT VII: PAIN Objectives: By the end of this lecture the students will be able to: Review the concept of somatosensory pathway. Describe the function of Nociceptors in response to pain information. Describe
UNIT VII: PAIN Objectives: By the end of this lecture the students will be able to: Review the concept of somatosensory pathway. Describe the function of Nociceptors in response to pain information. Describe
Somatosensory Physiology (Pain And Temperature) Richard M. Costanzo, Ph.D.
 Somatosensory Physiology (Pain And Temperature) Richard M. Costanzo, Ph.D. OBJECTIVES After studying the material of this lecture the student should be familiar with: 1. The relationship between nociception
Somatosensory Physiology (Pain And Temperature) Richard M. Costanzo, Ph.D. OBJECTIVES After studying the material of this lecture the student should be familiar with: 1. The relationship between nociception
What is pain?: An unpleasant sensation. What is an unpleasant sensation?: Pain. - Aristotle.
 What is pain?: An unpleasant sensation. What is an unpleasant sensation?: Pain. - Aristotle. Nociception The detection of tissue damage or impending tissue damage, but There can be tissue damage without
What is pain?: An unpleasant sensation. What is an unpleasant sensation?: Pain. - Aristotle. Nociception The detection of tissue damage or impending tissue damage, but There can be tissue damage without
Effect of Intravenous Lidocaine on the Neuropathic Pain of Failed Back Surgery Syndrome
 Original Article Korean J Pain 2012 April; Vol. 25, No. 2: 94-98 pissn 2005-9159 eissn 2093-0569 http://dx.doi.org/10.3344/kjp.2012.25.2.94 Effect of Intravenous Lidocaine on the Neuropathic Pain of Failed
Original Article Korean J Pain 2012 April; Vol. 25, No. 2: 94-98 pissn 2005-9159 eissn 2093-0569 http://dx.doi.org/10.3344/kjp.2012.25.2.94 Effect of Intravenous Lidocaine on the Neuropathic Pain of Failed
Interventional Pain. Judith Dunipace MD Board certified in Anesthesiology, Pain Management and Hospice and Palliative Care
 Interventional Pain Judith Dunipace MD Board certified in Anesthesiology, Pain Management and Hospice and Palliative Care IASP Definition of Pain Pain is an unpleasant sensory or emotional experience associated
Interventional Pain Judith Dunipace MD Board certified in Anesthesiology, Pain Management and Hospice and Palliative Care IASP Definition of Pain Pain is an unpleasant sensory or emotional experience associated
ANAT2010. Concepts of Neuroanatomy (II) S2 2018
 ANAT2010 Concepts of Neuroanatomy (II) S2 2018 Table of Contents Lecture 13: Pain and perception... 3 Lecture 14: Sensory systems and visual pathways... 11 Lecture 15: Techniques in Neuroanatomy I in vivo
ANAT2010 Concepts of Neuroanatomy (II) S2 2018 Table of Contents Lecture 13: Pain and perception... 3 Lecture 14: Sensory systems and visual pathways... 11 Lecture 15: Techniques in Neuroanatomy I in vivo
PAIN MANAGEMENT in the CANINE PATIENT
 PAIN MANAGEMENT in the CANINE PATIENT Laurie Edge-Hughes, BScPT, MAnimSt (Animal Physio), CAFCI, CCRT Part 1: Laurie Edge-Hughes, BScPT, MAnimSt (Animal Physio), CAFCI, CCRT 1 Pain is the most common reason
PAIN MANAGEMENT in the CANINE PATIENT Laurie Edge-Hughes, BScPT, MAnimSt (Animal Physio), CAFCI, CCRT Part 1: Laurie Edge-Hughes, BScPT, MAnimSt (Animal Physio), CAFCI, CCRT 1 Pain is the most common reason
Materials and Methods
 Anesthesiology 2003; 98:203 8 2003 American Society of Anesthesiologists, Inc. Lippincott Williams & Wilkins, Inc. Intrathecal Lidocaine Reverses Tactile Allodynia Caused by Nerve Injuries and Potentiates
Anesthesiology 2003; 98:203 8 2003 American Society of Anesthesiologists, Inc. Lippincott Williams & Wilkins, Inc. Intrathecal Lidocaine Reverses Tactile Allodynia Caused by Nerve Injuries and Potentiates
Mechanical sensitization of cutaneous sensory fibers in the spared nerve injury mouse model
 Smith et al. Molecular Pain 2013, 9:61 MOLECULAR PAIN SHORT REPORT Open Access Mechanical sensitization of cutaneous sensory fibers in the spared nerve injury mouse model Amanda K Smith, Crystal L O Hara
Smith et al. Molecular Pain 2013, 9:61 MOLECULAR PAIN SHORT REPORT Open Access Mechanical sensitization of cutaneous sensory fibers in the spared nerve injury mouse model Amanda K Smith, Crystal L O Hara
NMDA-Receptor Antagonists and Opioid Receptor Interactions as Related to Analgesia and Tolerance
 Vol. 19 No. 1(Suppl.) January 2000 Journal of Pain and Symptom Management S7 Proceedings Supplement NDMA-Receptor Antagonists: Evolving Role in Analgesia NMDA-Receptor Antagonists and Opioid Receptor Interactions
Vol. 19 No. 1(Suppl.) January 2000 Journal of Pain and Symptom Management S7 Proceedings Supplement NDMA-Receptor Antagonists: Evolving Role in Analgesia NMDA-Receptor Antagonists and Opioid Receptor Interactions
Management of Neuropathic pain
 Management of Neuropathic pain Ravi Parekodi Consultant in Anaesthetics and Pain Management 08/04/2014 Ref: BJA July2013, Map of Medicine2013, Pain Physician 2007, IASP 2012, Nice guideline 2013 Aims Highlight
Management of Neuropathic pain Ravi Parekodi Consultant in Anaesthetics and Pain Management 08/04/2014 Ref: BJA July2013, Map of Medicine2013, Pain Physician 2007, IASP 2012, Nice guideline 2013 Aims Highlight
Sensory information processing, somato-sensory systems
 mm? Sensory information processing, somato-sensory systems Recommended literature 1. Kandel ER, Schwartz JH, Jessel TM (2000) Principles of Neural Science, McGraw-Hill, Ch. xx. 2. Berne EM, Levy MN, Koeppen
mm? Sensory information processing, somato-sensory systems Recommended literature 1. Kandel ER, Schwartz JH, Jessel TM (2000) Principles of Neural Science, McGraw-Hill, Ch. xx. 2. Berne EM, Levy MN, Koeppen
Enhanced formalin nociceptive responses following L5 nerve ligation in the rat reveals neuropathy-induced inflammatory hyperalgesia
 University of Kentucky From the SelectedWorks of Renee R. Donahue 2001 Enhanced formalin nociceptive responses following L5 nerve ligation in the rat reveals neuropathy-induced inflammatory hyperalgesia
University of Kentucky From the SelectedWorks of Renee R. Donahue 2001 Enhanced formalin nociceptive responses following L5 nerve ligation in the rat reveals neuropathy-induced inflammatory hyperalgesia
A Patient s Guide to Pain Management: Complex Regional Pain Syndrome
 A Patient s Guide to Pain Management: Complex Regional Pain Syndrome 950 Breckinridge Lane Suite 220 Louisville, KY 40223 Phone: 502.708.2940 DISCLAIMER: The information in this booklet is compiled from
A Patient s Guide to Pain Management: Complex Regional Pain Syndrome 950 Breckinridge Lane Suite 220 Louisville, KY 40223 Phone: 502.708.2940 DISCLAIMER: The information in this booklet is compiled from
MYOFASCIAL PAIN. Dr. Janet Travell ( ) credited with bringing MTrPs to the attention of healthcare providers.
 Myofascial Trigger Points background info Laurie Edge-Hughes BScPT, MAnimSt (Animal Physio), CAFCI, CCRT History lesson Dr. Janet Travell (1901 1997) credited with bringing MTrPs to the attention of healthcare
Myofascial Trigger Points background info Laurie Edge-Hughes BScPT, MAnimSt (Animal Physio), CAFCI, CCRT History lesson Dr. Janet Travell (1901 1997) credited with bringing MTrPs to the attention of healthcare
MEDICAL POLICY SUBJECT: KETAMINE INFUSION THERAPY FOR THE TREATMENT OF CHRONIC PAIN SYNDROMES POLICY NUMBER: CATEGORY: Technology Assessment
 Clinical criteria used to make utilization review decisions are based on credible scientific evidence published in peer reviewed medical literature generally recognized by the medical community. Guidelines
Clinical criteria used to make utilization review decisions are based on credible scientific evidence published in peer reviewed medical literature generally recognized by the medical community. Guidelines
GENERAL PAIN DEFINITIONS
 I. OVERVIEW GENERAL PAIN DEFINITIONS Charles E. Argoff, MD CHAPTER 1 1. What is pain? Some dictionaries define pain as An unpleasant sensation, occurring in varying degrees of severity as a consequence
I. OVERVIEW GENERAL PAIN DEFINITIONS Charles E. Argoff, MD CHAPTER 1 1. What is pain? Some dictionaries define pain as An unpleasant sensation, occurring in varying degrees of severity as a consequence
A Patient s Guide to Pain Management: Complex Regional Pain Syndrome
 A Patient s Guide to Pain Management: Complex Regional Pain Syndrome Suite 11-13/14/15 Mount Elizabeth Medical Center 3 Mount Elizabeth Singapore, 228510 Phone: (65) 6738 2628 Fax: (65) 6738 2629 DISCLAIMER:
A Patient s Guide to Pain Management: Complex Regional Pain Syndrome Suite 11-13/14/15 Mount Elizabeth Medical Center 3 Mount Elizabeth Singapore, 228510 Phone: (65) 6738 2628 Fax: (65) 6738 2629 DISCLAIMER:
Various Types of Pain Defined
 Various Types of Pain Defined Pain: The International Association for the Study of Pain describes pain as, An unpleasant sensory and emotional experience associated with actual or potential tissue damage,
Various Types of Pain Defined Pain: The International Association for the Study of Pain describes pain as, An unpleasant sensory and emotional experience associated with actual or potential tissue damage,
Chapter 14: The Cutaneous Senses
 Chapter 14: The Cutaneous Senses Somatosensory System There are three parts Cutaneous senses - perception of touch and pain from stimulation of the skin Proprioception - ability to sense position of the
Chapter 14: The Cutaneous Senses Somatosensory System There are three parts Cutaneous senses - perception of touch and pain from stimulation of the skin Proprioception - ability to sense position of the
Anatomical Substrates of Somatic Sensation
 Anatomical Substrates of Somatic Sensation John H. Martin, Ph.D. Center for Neurobiology & Behavior Columbia University CPS The 2 principal somatic sensory systems: 1) Dorsal column-medial lemniscal system
Anatomical Substrates of Somatic Sensation John H. Martin, Ph.D. Center for Neurobiology & Behavior Columbia University CPS The 2 principal somatic sensory systems: 1) Dorsal column-medial lemniscal system
Long-term neuropathic pain
 Long-term neuropathic pain Per Hansson, Professor, MD, DMSci, DDS, specialist in neurology (and pain management) -Dept of Pain Management and Research, Oslo University Hospital, Oslo, Norway - Norwegian
Long-term neuropathic pain Per Hansson, Professor, MD, DMSci, DDS, specialist in neurology (and pain management) -Dept of Pain Management and Research, Oslo University Hospital, Oslo, Norway - Norwegian
COMPOUNDING PHARMACY SOLUTIONS PRESCRIPTION COMPOUNDING FOR PAIN MANAGEMENT
 JUNE 2012 COMPOUNDING PHARMACY SOLUTIONS PRESCRIPTION COMPOUNDING WWW.CPSRXS. COM We customize individual prescriptions for the specific needs of our patients. INSIDE THIS ISSUE: Acute Pain 2 Neuropathic
JUNE 2012 COMPOUNDING PHARMACY SOLUTIONS PRESCRIPTION COMPOUNDING WWW.CPSRXS. COM We customize individual prescriptions for the specific needs of our patients. INSIDE THIS ISSUE: Acute Pain 2 Neuropathic
Neuropathic Pain. Scott Magnuson, MD Pain Management of North Idaho, PLLC
 Neuropathic Pain Scott Magnuson, MD Pain Management of North Idaho, PLLC Pain is our friend "An unpleasant sensory and emotional experience associated with actual or potential tissue damage, or described
Neuropathic Pain Scott Magnuson, MD Pain Management of North Idaho, PLLC Pain is our friend "An unpleasant sensory and emotional experience associated with actual or potential tissue damage, or described
The activation of n-methyl-d-aspartate (NMDA) receptors is
 ORIGINAL ARTICLE Topically Administered Ketamine Reduces Capsaicin-Evoked Mechanical Hyperalgesia Reino Pöyhiä, MD, PhD* and Anneli Vainio, MD, PhD Background: The n-methyl-d-aspartate receptor antagonists
ORIGINAL ARTICLE Topically Administered Ketamine Reduces Capsaicin-Evoked Mechanical Hyperalgesia Reino Pöyhiä, MD, PhD* and Anneli Vainio, MD, PhD Background: The n-methyl-d-aspartate receptor antagonists
Biomechanics of Pain: Dynamics of the Neuromatrix
 Biomechanics of Pain: Dynamics of the Neuromatrix Partap S. Khalsa, D.C., Ph.D. Department of Biomedical Engineering The Neuromatrix From: Melzack R (1999) Pain Suppl 6:S121-6. NIOSH STAR Symposium May
Biomechanics of Pain: Dynamics of the Neuromatrix Partap S. Khalsa, D.C., Ph.D. Department of Biomedical Engineering The Neuromatrix From: Melzack R (1999) Pain Suppl 6:S121-6. NIOSH STAR Symposium May
The Somatosensory System
 The Somatosensory System Reading: BCP Chapter 12 cerebrovortex.com Divisions of the Somatosensory System Somatosensory System Exteroceptive External stimuli Proprioceptive Body position Interoceptive Body
The Somatosensory System Reading: BCP Chapter 12 cerebrovortex.com Divisions of the Somatosensory System Somatosensory System Exteroceptive External stimuli Proprioceptive Body position Interoceptive Body
Chapter 16. Sense of Pain
 Chapter 16 Sense of Pain Pain Discomfort caused by tissue injury or noxious stimulation, and typically leading to evasive action important /// helps to protect us lost of pain in diabetes mellitus = diabetic
Chapter 16 Sense of Pain Pain Discomfort caused by tissue injury or noxious stimulation, and typically leading to evasive action important /// helps to protect us lost of pain in diabetes mellitus = diabetic
Psychophysical laws. Legge di Fechner: I=K*log(S/S 0 )
 Psychophysical laws Legge di Weber: ΔS=K*S Legge di Fechner: I=K*log(S/S 0 ) Sensory receptors Vision Smell Taste Touch Thermal senses Pain Hearing Balance Proprioception Sensory receptors Table 21-1 Classification
Psychophysical laws Legge di Weber: ΔS=K*S Legge di Fechner: I=K*log(S/S 0 ) Sensory receptors Vision Smell Taste Touch Thermal senses Pain Hearing Balance Proprioception Sensory receptors Table 21-1 Classification
The Egyptian Journal of Hospital Medicine (January 2018) Vol. 70 (12), Page
 The Egyptian Journal of Hospital Medicine (January 2018) Vol. 70 (12), Page 2172-2177 Blockage of HCN Channels with ZD7288 Attenuates Mechanical Hypersensitivity in Rats Model of Diabetic Neuropathy Hussain
The Egyptian Journal of Hospital Medicine (January 2018) Vol. 70 (12), Page 2172-2177 Blockage of HCN Channels with ZD7288 Attenuates Mechanical Hypersensitivity in Rats Model of Diabetic Neuropathy Hussain
Nleurophysiology, Academic Hospital, Uppsala, Sweden
 Journal of Physiology (1989), 415, pp. 583-599 583 W'ith 3 text-figures Printed in Great Britain SENSATIONS EVOKED BY INTRANEURAL MICROSTIMULATION OF C NOCICEPTOR FIBRES IN HUMAN SKIN NERVES By JOSE OCHOA
Journal of Physiology (1989), 415, pp. 583-599 583 W'ith 3 text-figures Printed in Great Britain SENSATIONS EVOKED BY INTRANEURAL MICROSTIMULATION OF C NOCICEPTOR FIBRES IN HUMAN SKIN NERVES By JOSE OCHOA
Brian J. Snyder, M.D. Director - Functional and Restorative Neurosurgery NYU Winthrop Hospital Neurosurgery for Movement Disorders, Pain, Epilepsy,
 Brian J. Snyder, M.D. Director - Functional and Restorative Neurosurgery NYU Winthrop Hospital Neurosurgery for Movement Disorders, Pain, Epilepsy, and Psychiatric Illness WHAT IS COMPLEX REGIONAL PAIN
Brian J. Snyder, M.D. Director - Functional and Restorative Neurosurgery NYU Winthrop Hospital Neurosurgery for Movement Disorders, Pain, Epilepsy, and Psychiatric Illness WHAT IS COMPLEX REGIONAL PAIN
Overview of Questions
 Overview of Questions What are the sensors in the skin, what do they respond to and how is this transmitted to the brain? How does the brain represent touch information? What is the system for sensing
Overview of Questions What are the sensors in the skin, what do they respond to and how is this transmitted to the brain? How does the brain represent touch information? What is the system for sensing
Pain classifications slow and fast
 Pain classifications slow and fast Fast Pain Slow Pain Sharp, pricking (Aδ) fiber Short latency Well localized Short duration Dull, burning (C) fiber Slower onset Diffuse Long duration Less emotional Emotional,
Pain classifications slow and fast Fast Pain Slow Pain Sharp, pricking (Aδ) fiber Short latency Well localized Short duration Dull, burning (C) fiber Slower onset Diffuse Long duration Less emotional Emotional,
III: To define the mechanoreceptive and thermoreceptive sensations.
 Somatic Sensations I: To explain the proper way of sensory testing. II: To describe the somatic sensations. III: To define the mechanoreceptive and thermoreceptive sensations. SOMATIC SENSATIONS Somatic
Somatic Sensations I: To explain the proper way of sensory testing. II: To describe the somatic sensations. III: To define the mechanoreceptive and thermoreceptive sensations. SOMATIC SENSATIONS Somatic
SYLLABUS SPRING 2011 COURSE: NSC NEUROBIOLOGY OF PAIN
 SYLLABUS NSC 4358 NEUROBIOLOGY OF PAIN SPRING 2011 1 SYLLABUS SPRING 2011 COURSE: NSC 4358 001 NEUROBIOLOGY OF PAIN Instructor: Aage R. Møller PhD E-mail: AMOLLER@UTDALLAS.EDU Class schedule: Main Campus:
SYLLABUS NSC 4358 NEUROBIOLOGY OF PAIN SPRING 2011 1 SYLLABUS SPRING 2011 COURSE: NSC 4358 001 NEUROBIOLOGY OF PAIN Instructor: Aage R. Møller PhD E-mail: AMOLLER@UTDALLAS.EDU Class schedule: Main Campus:
Neuropathic pain, pain matrix dysfunction, and pain syndromes
 Neuropathic pain, pain matrix dysfunction, and pain syndromes MSTN121 - Neurophysiology Session 3 Department of Myotherapy Session objectives Describe the mechanism of nociceptive chronic pain. Define
Neuropathic pain, pain matrix dysfunction, and pain syndromes MSTN121 - Neurophysiology Session 3 Department of Myotherapy Session objectives Describe the mechanism of nociceptive chronic pain. Define
Pathophysiology of Pain. Ramon Go MD Assistant Professor Anesthesiology and Pain medicine NYP-CUMC
 Pathophysiology of Pain Ramon Go MD Assistant Professor Anesthesiology and Pain medicine NYP-CUMC Learning Objectives Anatomic pathway of nociception Discuss the multiple target sites of pharmacological
Pathophysiology of Pain Ramon Go MD Assistant Professor Anesthesiology and Pain medicine NYP-CUMC Learning Objectives Anatomic pathway of nociception Discuss the multiple target sites of pharmacological
Assay Sensitivity.
 Assay Sensitivity Michael C. Rowbotham, MD Professor of Neurology UCSF-Mount Zion Pain Management Center Senior Scientist and IRB Chair, CPMC Research Institute Michael.Rowbotham@ucsf.edu Outline What
Assay Sensitivity Michael C. Rowbotham, MD Professor of Neurology UCSF-Mount Zion Pain Management Center Senior Scientist and IRB Chair, CPMC Research Institute Michael.Rowbotham@ucsf.edu Outline What
SUPPLEMENTARY INFORMATION
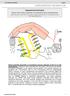 doi: 1.138/nature588 SUPPLEMENTARY INFORMATION Supplemental Information Sensory neuron sodium channel Na v 1.8 is essential for pain at cold temperatures Katharina Zimmermann*, Andreas Leffler*, Alexandru
doi: 1.138/nature588 SUPPLEMENTARY INFORMATION Supplemental Information Sensory neuron sodium channel Na v 1.8 is essential for pain at cold temperatures Katharina Zimmermann*, Andreas Leffler*, Alexandru
SENSORY FUNCTIONS OF THE SKIN OF HUMANS
 SENSORY FUNCTIONS OF THE SKIN OF HUMANS SENSORY FUNCTIONS OF THE SKIN OF HUMANS EDITED BY DAN R. KENSHALO Florida State University Tallahassee, Florida PLENUM PRESS NEW YORK AND LONDON Library of Congress
SENSORY FUNCTIONS OF THE SKIN OF HUMANS SENSORY FUNCTIONS OF THE SKIN OF HUMANS EDITED BY DAN R. KENSHALO Florida State University Tallahassee, Florida PLENUM PRESS NEW YORK AND LONDON Library of Congress
Proceedings of the World Small Animal Veterinary Association Sydney, Australia 2007
 Proceedings of the World Small Animal Sydney, Australia 2007 Hosted by: Next WSAVA Congress REDUCING THE PAIN FACTOR AN UPDATE ON PERI-OPERATIVE ANALGESIA Sandra Forysth, BVSc DipACVA Institute of Veterinary,
Proceedings of the World Small Animal Sydney, Australia 2007 Hosted by: Next WSAVA Congress REDUCING THE PAIN FACTOR AN UPDATE ON PERI-OPERATIVE ANALGESIA Sandra Forysth, BVSc DipACVA Institute of Veterinary,
PAIN MODULATION. numerical value. adjectives. DR SYED SHAHID HABIB Professor & Consultant Dept. of Physiology College of Medicine & KKUH
 PAIN MODULATION numerical value adjectives DR SYED SHAHID HABIB Professor & Consultant Dept. of Physiology College of Medicine & KKUH OBJECTIVES At the end of this lecture you should be able to describe:
PAIN MODULATION numerical value adjectives DR SYED SHAHID HABIB Professor & Consultant Dept. of Physiology College of Medicine & KKUH OBJECTIVES At the end of this lecture you should be able to describe:
Ch. 47 Somatic Sensations: Tactile and Position Senses (Reading Homework) - Somatic senses: three types (1) Mechanoreceptive somatic senses: tactile
 Ch. 47 Somatic Sensations: Tactile and Position Senses (Reading Homework) - Somatic senses: three types (1) Mechanoreceptive somatic senses: tactile and position sensations (2) Thermoreceptive senses:
Ch. 47 Somatic Sensations: Tactile and Position Senses (Reading Homework) - Somatic senses: three types (1) Mechanoreceptive somatic senses: tactile and position sensations (2) Thermoreceptive senses:
David S. Warner, M.D., Editor. Testing the Link between Sympathetic Efferent and Sensory Afferent Fibers in Neuropathic Pain
 David S. Warner, M.D., Editor Testing the Link between Sympathetic Efferent and Sensory Afferent Fibers in Neuropathic Pain Srinivasa N. Raja, M.D.,* Rolf-Detlef Treede, M.D., Ph.D. Systemic -adrenergic
David S. Warner, M.D., Editor Testing the Link between Sympathetic Efferent and Sensory Afferent Fibers in Neuropathic Pain Srinivasa N. Raja, M.D.,* Rolf-Detlef Treede, M.D., Ph.D. Systemic -adrenergic
EE 791 Lecture 2 Jan 19, 2015
 EE 791 Lecture 2 Jan 19, 2015 Action Potential Conduction And Neural Organization EE 791-Lecture 2 1 Core-conductor model: In the core-conductor model we approximate an axon or a segment of a dendrite
EE 791 Lecture 2 Jan 19, 2015 Action Potential Conduction And Neural Organization EE 791-Lecture 2 1 Core-conductor model: In the core-conductor model we approximate an axon or a segment of a dendrite
Testing the gate-control theory of pain in man
 Journal of Neurology, Neurosurgery, and Psychiatry, 1974, 37, 1366-1372 Testing the gate-control theory of pain in man P. W. NATHAN' AND P. RUDGE From the National Hospital, Queen Square, London SYNOPSIS
Journal of Neurology, Neurosurgery, and Psychiatry, 1974, 37, 1366-1372 Testing the gate-control theory of pain in man P. W. NATHAN' AND P. RUDGE From the National Hospital, Queen Square, London SYNOPSIS
Pathophysiology of Pain
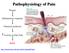 Pathophysiology of Pain Wound Inflammatory response Chemical mediators Activity in Pain Path PAIN http://neuroscience.uth.tmc.edu/s2/chapter08.html Chris Cohan, Ph.D. Dept. of Pathology/Anat Sci University
Pathophysiology of Pain Wound Inflammatory response Chemical mediators Activity in Pain Path PAIN http://neuroscience.uth.tmc.edu/s2/chapter08.html Chris Cohan, Ph.D. Dept. of Pathology/Anat Sci University
Human cutaneous C fibres activated by cooling, heating and menthol
 J Physiol 587.23 (2009) pp 5633 5652 5633 Human cutaneous C fibres activated by cooling, heating and menthol M. Campero 1, T. K. Baumann 2,H.Bostock 3 and J. L. Ochoa 4 1 Facultad de Medicina, Clínica
J Physiol 587.23 (2009) pp 5633 5652 5633 Human cutaneous C fibres activated by cooling, heating and menthol M. Campero 1, T. K. Baumann 2,H.Bostock 3 and J. L. Ochoa 4 1 Facultad de Medicina, Clínica
Standardized Evaluation of Pain (StEP) Neuropathic Pain
 Standardized Evaluation of Pain (StEP) Neuropathic Pain Patient s name: Date of birth: / / Patient ID Examiner s name: Date of the examination: / / Introduction (please read to the patient) To get a better
Standardized Evaluation of Pain (StEP) Neuropathic Pain Patient s name: Date of birth: / / Patient ID Examiner s name: Date of the examination: / / Introduction (please read to the patient) To get a better
Pain. Pain. Pain: One definition. Pain: One definition. Pain: One definition. Pain: One definition. Psyc 2906: Sensation--Introduction 9/27/2006
 Pain Pain Pain: One Definition Classic Paths A new Theory Pain and Drugs According to the international Association for the Study (Merskey & Bogduk, 1994), Pain is an unpleasant sensory and emotional experience
Pain Pain Pain: One Definition Classic Paths A new Theory Pain and Drugs According to the international Association for the Study (Merskey & Bogduk, 1994), Pain is an unpleasant sensory and emotional experience
Cardiovascular Responses to Exercise
 CARDIOVASCULAR PHYSIOLOGY 69 Case 13 Cardiovascular Responses to Exercise Cassandra Farias is a 34-year-old dietician at an academic medical center. She believes in the importance of a healthy lifestyle
CARDIOVASCULAR PHYSIOLOGY 69 Case 13 Cardiovascular Responses to Exercise Cassandra Farias is a 34-year-old dietician at an academic medical center. She believes in the importance of a healthy lifestyle
Peripheral Nervous System
 Peripheral Nervous System 1 Sensory Receptors Sensory Receptors and Sensation Respond to changes (stimuli) in the environment Generate graded potentials that can trigger an action potential that is carried
Peripheral Nervous System 1 Sensory Receptors Sensory Receptors and Sensation Respond to changes (stimuli) in the environment Generate graded potentials that can trigger an action potential that is carried
