Haemolytic Uraemic Syndrome and The ECUSTEC Trial
|
|
|
- Amos Austin
- 5 years ago
- Views:
Transcription
1 Haemolytic Uraemic Syndrome and The ECUSTEC Trial Dr Sally Johnson Consultant Paediatric Nephrologist Great North Children s Hospital 14 th October 2016
2 Haemolytic Uraemic Syndrome Commonest cause of intrinsic acute kidney injury in children Classical triad Microangiopathic haemolytic anaemia Thrombocytopenia Acute kidney injury cases per year in UK 95% cases follow infection with Shiga-toxin producing E. coli (STEC)
3 STEC infection STEC causes around 1000 cases of gastro-enteritis in England p.a. Diarrhoea and vomiting, abdominal pain Bloody diarrhoea (red flag symptom) Natural reservoir of STEC - intestines of ruminants Infection follows Ingestion of contaminated foods/water Direct or indirect contact with animals (e.g. cattle, sheep) Person-to-person spread Commonest serotype is O157 Also O55, O26, O111 and others Key attribute is the secretion of Shiga-toxin Diagnosis Stool culture specialised culture Stool PCR for Shiga-toxin Serology
4 STEC (typical) HUS Occurs after 10-15% of STEC infections 3-10 days following colitis onset 50% require Renal Replacement Therapy (RRT), mean duration 10 days 1-3% acute mortality Extra renal complications 20-25% CNS involvement Severe gut disease Pancreatitis Management entirely supportive Aim for euvolaemia - saline RRT and blood transfusion Anticipation and management of complications Outcome 12% risk end-stage renal failure or death 25% risk chronic kidney disease Long term follow-up advocated
5 STEC HUS Intestine Blood Target organ Thrombotic microangiopathy Gb3 receptor Endothelium Inflammatory mediators MAHA Thromobycytopenia Acute kidney injury
6 Normal glomerulus Thrombotic microangiopathy
7
8
9 Adapted from Besbas Kidney Int 2006 Causes of HUS 5% 95% Infection-induced (a) Shiga toxin-producing bacteria (STEC, Shigella) (b) Streptococcus pneumoniae Disorders of complement regulation (a) Genetic (b) Acquired Miscellaneous ADAMTS13 deficiency Defective cobalamin metabolism Quinine-induced Malignancy, cancer chemotherapy, ionizing radiation Calcineurin inhibitors and transplantation SLE, anti-phospholipid antibody syndrome HIV infection Pregnancy, HELLP syndrome, contraceptive pill
10 Alternative complement pathway C3 Factor D C3bBb C3 convertase C3bB C5 Factor B C3bBbC3b C5 convertase C3b CFH, MCP, CR-1, DAF CD59 C5b-9 MAC C5a C3a CFH, MCP or CR-1 with CFI ic3b Lysis / sublytic effects Chemotaxis Opsonisation
11 Atypical HUS underlying abnormalities Gene or abnormality Complement Factor H 20-30% Membrane Cofactor Protein 5-15% Complement Factor I 4-10% Complement Factor B 1-4% C3 2-10% Thrombomodulin 0-3% DGKe 3-5% Anti-CFH antibodies 6% Frequency Relapsing course Progressive 50% end stage renal failure or death at 5 years from diagnosis Traditional therapy plasma therapy Unexplained 30-40%
12 Creatinine (mg per dl) Platelet Count LDH (U per L) Case report of chronic eculizumab 8,000 Plasma Therapy therapy Eculizumab 6,000 4,000 2,000 (x10 3 per µl) Days of Treatment Days of Treatment Gruppo RA, et al. N Engl J Med. 2009;360(5):544-6.
13 Eculizumab is a safe and effective treatment in pediatric patients with atypical hemolytic uremic syndrome Figure 2. Improvement in platelet count over 27 weeks of eculizumab treatment. N values <5 were not included. Bars represent SEM. Arrows denote administration of eculizumab. Larry A. Greenbaum, Marc Fila, Gianluigi Ardissino, Samhar I. Al-Akash, Jonathan Evans, Paul Henning, Kenneth V. Lieberman, Silvio Maringhini, Lars Pape, Lesley Rees, Nicole C.A.J. van de Kar, Johan Vande Walle, Masayo Ogawa, Camille L. Bedrosian, Christoph Licht Kidney International, Volume 89, Issue 3, 2016,
14 Lapeyraque NEJM 2011
15 German outbreak May-July STEC cases 845 (22%) developed HUS 54 deaths 88% of those with HUS were adults, mostly women Causative serotype E coli O104:H4 Combination of characteristics from EAEC and EHEC Frank NEJM 2011; Wadl Euro Surveill. 2011
16 German outbreak data Ad hoc use of eculizumab Compassionate use Often combined with plasma exchange Outcome data difficult to interpret Industry sponsored trial 198 patients 7 doses ( 84,000) Data presented to ASN 2012, no publication Off-label use increasing globally Kielstein NDT 2012 Menne BMJ 2012
17 Complement in STEC HUS Low plasma C3 levels in acute STEC-HUS are associated with leucocytosis and severe disease Levels of C3a, Bb and sc5b-9 are transiently elevated in children with STEC-HUS mean plasma concentration of Bb (A) and SC5b-9 (B) in patients with D+HUS plasma concentration of Bb (A) and SC5b-9 (B) over the course of the D+HUS episode Robson 1992 Thurman 2009 Stahl 2011 Morigi 2001
18 Shiga toxin reduces podocyte VEGF which in turn reduces endothelial complement regulator expression Podocyte GBM Endothelium Stx interacts with CFH in fluid phase Stx activates complement on endothelium via ACP (P-selectin binds C3) Activated complement components are detected on plateletleucocyte complexes Orth J Immunol 2009, Sakuma Art Thromb Vasc Biol 2010, Mason J Biol Chem 2004 Eremina NEJM 2008, Keir Ped Nephrol 2014, Morigi J Immunol 2011 Complement mediates proliferative response to vascular injury (C3a/C5a)
19 ECUSTEC A randomised, double-blind placebo controlled trial of ECUlizumab in STEC Haemolytic Uraemic Syndrome NIHR Efficacy and Mechanism Evaluation funded 2015
20 The ECUSTEC team Co-applicant Role Institution Sally Johnson Consultant Paediatric Nephrologist Newcastle Nick Webb Consultant Paediatric Nephrologist Manchester Moin Saleem Consultant Paediatric Nephrologist Bristol Rodney Gilbert Consultant Paediatric Nephrologist Southampton Aoife Waters Consultant Paediatric Nephrologist GOSH Natalie Ives Senior Statistician Birmingham Clinical Trials Unit Elizabeth Brettell Renal Trials Manager Birmingham Clinical Trials Unit Hugh McCloud Health Economist Birmingham Clinical Trials Unit Munir Ahmed General Paediatrician (SPIN) Worcestershire Steve Nash Co-founder HUSH (E coli support group) Claire Jenkins* Head of Gastrointestinal Reference Laboratory Public Health England * collaborator
21 Research objectives In children aged 6 months to <19 years inclusive, we intend: To determine whether the severity of STEC HUS is less in those given Ecu compared with those given placebo To assess the safety of Ecu in STEC HUS To determine whether the incidence of CKD following STEC HUS is less in those receiving Ecu compared with those receiving placebo To evaluate the cost-effectiveness of administration of Ecu in STEC HUS from the perspective of the NHS
22 Trial overview 134 participants
23 Inclusion criteria Age 6 months to <19 years Weight 5kg Diagnosis of HUS Micro-angiopathic haemolytic anaemia (indicated by fragmented red cells on blood film OR plasma lactate dehydrogenase above local centre reference range) AND Thrombocytopenia (platelets <150x10 9 /l) AND Acute Kidney Injury (AKI): injury or failure category of prifle criteria 27 (Table 1) despite correction of hypovolaemia EITHER Reported diarrhoea within 14 days prior to diagnosis of HUS (defined according to World Health Organisation as the passage of three or more loose or liquid stools per day - or more frequent passage than is normal for the individual ) OR Stool culture or shiga toxin polymerase chain reaction (PCR) or STEC serology result indicating STEC in the patient or household contact within 14 days prior to diagnosis of HUS Patient intended to be able to receive trial drug within 36 hours of arrival at renal unit, or within 24 hours of eligibility if already at renal unit Sexually active male or female patients must agree to be practicing an effective, reliable and medically approved contraceptive regimen for 6 months after enrolment. Written informed consent obtained from the participant s parents/guardians and written assent obtained from participant (where age appropriate). Participants aged 16 years and above will provide their own written consent.
24 Patient Identification Centre (PIC) NHS site Can identify and refer potential patients to another site for consideration of entry into clinical trial Simple set-up Can give patient information Activity is acknowledged
25 Sequence District general hospital (may or may not be a PIC) STEC HUS is suspected (all inclusion criteria do not need to be met) Clinical stabilisation Contact the regional paediatric nephrologist on-call Renal unit If diagnosis highly likely to be STEC HUS, will try to accept the referral and arrange transfer If child does not require transfer for clinical purposes, the paediatric nephrologist will explain that transfer will be accepted earlier than usual so that participation in the trial can be offered and explain that there will be no obligation to enter the trial Information regarding ECUSTEC will be offered pre-transfer if the referring hospital is a PIC
26 After arrival at renal unit Clinical stabilisation and hypovolaemia correction Information about study given Creatinine repeated If remains eligible, seek informed consent Randomisation Completion of baseline assessments Meningococcal prophylaxis/vaccination Administration of study drug within 36 hours of arrival Blinded pharmacy staff Eculizumab or saline Second dose 7 days later
27 Diagnosis Screening Enrolment Immunisation Randomisation Patient identification centre Transfer Randomisation minimised prifle stage Volume of saline received Penicillin prophylaxis Supportive care ECUSTEC Clinical severity score Formal GFR** Placebo Eculizumab Exclude hypovolaemia Supportive care Penicillin prophylaxis Daily assessments until discharge or D14 ECUSTEC Clinical severity score Formal GFR**
28 Primary outcome measure: ECUSTEC Clinical Severity Score 80% power to detect a 5 point reduction in CSS Pilot data: Mean score SD score 9.66
29 Secondary outcome measures Survival Duration of thrombocytopenia (number of days until platelet count >150x10 9 /l) Duration of haemolysis (number of days until LDH within normal reference range) Number of packed red blood cell transfusions required and volume (ml/kg) Markers of inflammation (number of days until normal white cell count and CRP) CKD at 1 year - a composite endpoint of the presence of Hypertension Average of 3 readings by manual method using centiles for age/sex/height Above 95 th centile for age/sex/height Albuminuria Early morning urine albumin-creatinine ratio >2.5mg/mmol egfr<90ml/min/1.73m 2 at 1 year Persistent neurological defect at 60 days
30 Additional outcomes Safety Mechanistic studies Health economic evaluation Cost-effectiveness of eculizumab vs. placebo will be measured cost per ECUSTEC CSS point cost per QALY Health-related QoL CHU9D (>5y) and PEDsQL (<5y)
31 Recruitment projections
32 ECULISHU Inclusion criteria egfr <75 Exclusion criteria STEC-HUS patient with severe multiorgan involvement at diagnosis: Neurological involvement (seizures, coma, focal deficit) with signs of microangiopathy on cerebral Magnetic Resonance Imaging. Cardiac involvement (cardiac failure, ischemic myocarditis, conduction or rhythm troubles) Digestive involvement (severe pancreatitis defined by lipasemia>500ui/l, severe hepatitis defined by transaminase >x10uln and/or prothrombin time<60%, hemorrhagic colitis, bowel perforation, rectal prolapsus) If develops severe disease in placebo arm Eculizumab
33 Challenges Patient identification Your help is vital! Majority of cases discussed with regional nephrology unit Nominated Patient Identification Centres Please let your colleagues know! Transfer Earlier than usual if possible Rapid enrolment after transfer PIC can give information prior to transfer Patient and carer involvement in production of patient information and pathways Rapid administration of study drug Will require site-specific solutions
34
35 Exclusion criteria Family history of ahus Previous episode of HUS Known pre-existing egfr <90ml/min/1.73m2 Known or suspected pneumococcal infection Patient taking a drug known to be associated with HUS, e.g. calcineurin inhibitors, chemotherapy, quinine, oral contraceptive pill Pregnancy Refusal of consent, including consent for meningococcal vaccination Currently participating in another CTIMP
36 Standardised non-trial management Neurological features Pilot data (5 centres) - 14/100 developed CNS features PEx in 8/14 dependent on centre and not clinical features Agreement to avoid PEx in trial participants Agreement to avoid eculizumab in control arm NO PLASMA EXCHANGE (8) PLASMA EXCHANGE (6) Presentation Presentation No of PEX 1 Seizure, focal neurology <24 hrs* Tonic Seizure- diffuse slow EEG? Hallucinations Seizure- over 3 days 3 3x seizure- 2 vacant and 1 focal Slurred Speech <24 hours 5 1x Tonic Clonic seizure 1x Tonic Clonic seizure 6 Generalised weakness Rt>Lt. >24hrs Confusion, hemiparesis <24hrs 6 Encephalopathy then Seizures 1x Tonic Clonic seizure 3 Encephalopathy <24hours Tonic clonic seizure**
37 prifle criteria Schwartz 2009 (k=41.3) Height required Creatinine real world
Supplementary Appendix
 Supplementary Appendix This appendix has been provided by the authors to give readers additional information about their work. Supplement to: Lapeyraque A-L, Malina M, Fremeaux-Bacchi V, et al. Eculizumab
Supplementary Appendix This appendix has been provided by the authors to give readers additional information about their work. Supplement to: Lapeyraque A-L, Malina M, Fremeaux-Bacchi V, et al. Eculizumab
Updates on Paediatric clinical research activities
 Updates on Paediatric clinical research activities Shivaram Hegde Consultant paediatric nephrologist Children Kidney centre UHW, Cardiff Aim to Brief introduction Research updates Improve contribution
Updates on Paediatric clinical research activities Shivaram Hegde Consultant paediatric nephrologist Children Kidney centre UHW, Cardiff Aim to Brief introduction Research updates Improve contribution
Hemolytic uremic syndrome: Investigations and management
 Hemolytic uremic syndrome: Investigations and management SAWAI Toshihiro M.D., Ph.D. Department of Pediatrics, Shiga University of Medical Science Otsu, JAPAN AGENDA TMA; Thrombotic micro angiopathy STEC-HUS;
Hemolytic uremic syndrome: Investigations and management SAWAI Toshihiro M.D., Ph.D. Department of Pediatrics, Shiga University of Medical Science Otsu, JAPAN AGENDA TMA; Thrombotic micro angiopathy STEC-HUS;
DRUG NAME: Eculizumab Brand(s): Soliris DOSAGE FORM/ STRENGTH: 10 mg/ml (300 mg per vial)
 Preamble: A confirmed diagnosis of atypical hemolytic uremic syndrome (ahus) is required for eculizumab funding. The information below is to provide clinicians with context for how a diagnosis of ahus
Preamble: A confirmed diagnosis of atypical hemolytic uremic syndrome (ahus) is required for eculizumab funding. The information below is to provide clinicians with context for how a diagnosis of ahus
R. Coward has documented that he has received cooperative grants from Takeda and Novo Nordisk
 R. Coward has documented that he has received cooperative grants from Takeda and Novo Nordisk Advances in our understanding of the pathogenesis of glomerular thrombotic microangiopathy Lindsay Keir Richard
R. Coward has documented that he has received cooperative grants from Takeda and Novo Nordisk Advances in our understanding of the pathogenesis of glomerular thrombotic microangiopathy Lindsay Keir Richard
Safety and Efficacy of Eculizumab in Pediatric Patients With ahus, With or Without Baseline Dialysis
 SA-PO546 Safety and Efficacy of Eculizumab in Pediatric Patients With ahus, With or Without Baseline Johan Vande Walle, 1 Larry A. Greenbaum, 2 Camille L. Bedrosian, 3 Masayo Ogawa, 3 John F. Kincaid,
SA-PO546 Safety and Efficacy of Eculizumab in Pediatric Patients With ahus, With or Without Baseline Johan Vande Walle, 1 Larry A. Greenbaum, 2 Camille L. Bedrosian, 3 Masayo Ogawa, 3 John F. Kincaid,
What is meant by Thrombotic Microangiopathy (TMA)?
 What is meant by Thrombotic Microangiopathy (TMA)? Thrombotic Microangiopathy (TMA) is a group of disorders characterized by injured endothelial cells, microangiopathic hemolytic anemia (MAHA), with its
What is meant by Thrombotic Microangiopathy (TMA)? Thrombotic Microangiopathy (TMA) is a group of disorders characterized by injured endothelial cells, microangiopathic hemolytic anemia (MAHA), with its
A Case of Escherichia coli Hemolytic Uremic Syndrome in a 10-Year-Old Male With Severe Neurologic Involvement Successfully Treated With Eculizumab
 741144HICXXX10.1177/2324709617741144Journal of Investigative Medicine High Impact Case ReportsRasa et al research-article20172017 Case Report A Case of Escherichia coli Hemolytic Uremic Syndrome in a 10-Year-Old
741144HICXXX10.1177/2324709617741144Journal of Investigative Medicine High Impact Case ReportsRasa et al research-article20172017 Case Report A Case of Escherichia coli Hemolytic Uremic Syndrome in a 10-Year-Old
Hemolytic uremic syndrome
 Hemolytic uremic syndrome Doyeun Oh Department of Internal Medicine CHA University School of Medicine Disclosures for Doyeun Oh Research Support/P.I. Employee Consultant Major Stockholder Speakers Bureau
Hemolytic uremic syndrome Doyeun Oh Department of Internal Medicine CHA University School of Medicine Disclosures for Doyeun Oh Research Support/P.I. Employee Consultant Major Stockholder Speakers Bureau
Safety and Efficacy of Eculizumab in Pediatric Patients With ahus, With or Without Baseline Dialysis
 SP281 Safety and Efficacy of Eculizumab in Pediatric Patients With ahus, With or Without Baseline Johan Vande Walle, 1 Larry A. Greenbaum, 2 Camille L. Bedrosian, 3 Masayo Ogawa, 3 John F. Kincaid, 3 Chantal
SP281 Safety and Efficacy of Eculizumab in Pediatric Patients With ahus, With or Without Baseline Johan Vande Walle, 1 Larry A. Greenbaum, 2 Camille L. Bedrosian, 3 Masayo Ogawa, 3 John F. Kincaid, 3 Chantal
Recent advances in pathogenesis & treatment of ahus
 Recent advances in pathogenesis & treatment of ahus Miquel Blasco Pelicano Nephrology and Kidney Transplant Unit Hospital Clínic, Barcelona Atypical Hemolytic Uremic Syndrome (ahus) Ultra-rare disease:
Recent advances in pathogenesis & treatment of ahus Miquel Blasco Pelicano Nephrology and Kidney Transplant Unit Hospital Clínic, Barcelona Atypical Hemolytic Uremic Syndrome (ahus) Ultra-rare disease:
Risk factors of chronic renal failure after atypical Hemolytic Uremic Syndrome under plasmatherapy
 Risk factors of chronic renal failure after atypical Hemolytic Uremic Syndrome under plasmatherapy Professeur Eric Rondeau Urgences néphrologiques et Transplantation rénale Hôpital Tenon, Paris WWA SFH
Risk factors of chronic renal failure after atypical Hemolytic Uremic Syndrome under plasmatherapy Professeur Eric Rondeau Urgences néphrologiques et Transplantation rénale Hôpital Tenon, Paris WWA SFH
Specialised Services Policy: CP98 Eculizumab for Atypical Haemolytic Uraemic Syndrome (ahus)
 Specialised Services Policy: CP98 Eculizumab for Atypical Haemolytic Uraemic Syndrome (ahus) Document Author: Assistant Director for Evidence, Evaluation and Effectiveness Executive Lead: Medical Director
Specialised Services Policy: CP98 Eculizumab for Atypical Haemolytic Uraemic Syndrome (ahus) Document Author: Assistant Director for Evidence, Evaluation and Effectiveness Executive Lead: Medical Director
Beyond Plasma Exchange: Targeted Therapy for Thrombotic Thrombocytopenic Purpura
 Beyond Plasma Exchange: Targeted Therapy for Thrombotic Thrombocytopenic Purpura Kristen Knoph, PharmD, BCPS PGY2 Pharmacotherapy Resident Pharmacy Grand Rounds April 25, 2017 2016 MFMER slide-1 Objectives
Beyond Plasma Exchange: Targeted Therapy for Thrombotic Thrombocytopenic Purpura Kristen Knoph, PharmD, BCPS PGY2 Pharmacotherapy Resident Pharmacy Grand Rounds April 25, 2017 2016 MFMER slide-1 Objectives
1. INSTRUCTIONS 2. DEFINITION OF HUS
 CQ_IBK_aHUS_01 / version 25/11/09 European Paediatric Research Group for HUS and related disorders Case questionnaire for diarrhoea negative/vtec (STEC) negative cases acute phase 1. INSTRUCTIONS Please
CQ_IBK_aHUS_01 / version 25/11/09 European Paediatric Research Group for HUS and related disorders Case questionnaire for diarrhoea negative/vtec (STEC) negative cases acute phase 1. INSTRUCTIONS Please
Hemolytic Uremic Syndrome
 Hemolytic Uremic Syndrome Francesco Emma Division of Nephrology and Dialysis Bambino Gesù Children s Hospital, IRCCS Rome, Italy Hemolytic Uremic Syndrome (HUS) microangiopathic hemolytic anemia thrombocytopenia
Hemolytic Uremic Syndrome Francesco Emma Division of Nephrology and Dialysis Bambino Gesù Children s Hospital, IRCCS Rome, Italy Hemolytic Uremic Syndrome (HUS) microangiopathic hemolytic anemia thrombocytopenia
Soliris Medical Policy Prior Authorization Program Summary
 Soliris Medical Policy Prior Authorization Program Summary Precertification/Prior Authorization may be required under certain plans. Please verify each member s benefits. OBJECTIVE The intent of the Soliris
Soliris Medical Policy Prior Authorization Program Summary Precertification/Prior Authorization may be required under certain plans. Please verify each member s benefits. OBJECTIVE The intent of the Soliris
M.Weitz has documented that he has no relevant financial relationships to disclose or conflict of interest to resolve.
 M.Weitz has documented that he has no relevant financial relationships to disclose or conflict of interest to resolve. Prophylactic eculizumab prior to kidney transplantation for atypical hemolytic uremic
M.Weitz has documented that he has no relevant financial relationships to disclose or conflict of interest to resolve. Prophylactic eculizumab prior to kidney transplantation for atypical hemolytic uremic
Soliris (eculizumab) Inhibits TMA and Improves Renal Function in Pediatric and Adult Patients with atypical Hemolytic Uremic Syndrome (ahus)
 Soliris (eculizumab) Inhibits TMA and Improves Renal Function in Pediatric and Adult Patients with atypical Hemolytic Uremic Syndrome (ahus) New Data from the Largest Prospective Trial of Adult Patients
Soliris (eculizumab) Inhibits TMA and Improves Renal Function in Pediatric and Adult Patients with atypical Hemolytic Uremic Syndrome (ahus) New Data from the Largest Prospective Trial of Adult Patients
Atypical Hemolytic Uremic Syndrome: When the Environment and Mutations Affect Organ Systems. A Case Report with Review of Literature
 Atypical Hemolytic Uremic Syndrome: When the Environment and Mutations Affect Organ Systems. A Case Report with Review of Literature Mouhanna Abu Ghanimeh 1, Omar Abughanimeh 1, Ayman Qasrawi 1, Abdulraheem
Atypical Hemolytic Uremic Syndrome: When the Environment and Mutations Affect Organ Systems. A Case Report with Review of Literature Mouhanna Abu Ghanimeh 1, Omar Abughanimeh 1, Ayman Qasrawi 1, Abdulraheem
Renal failure and thrombocytopaenia? Don t forget TTP/HUS. Jonathan Wala Nephrologist
 Renal failure and thrombocytopaenia? Don t forget TTP/HUS Jonathan Wala Nephrologist Thrombotic microangiopathies Disorders characterized by: thrombocytopaenia microangiopathic haemolytic anaemia (MAHA)
Renal failure and thrombocytopaenia? Don t forget TTP/HUS Jonathan Wala Nephrologist Thrombotic microangiopathies Disorders characterized by: thrombocytopaenia microangiopathic haemolytic anaemia (MAHA)
Dr. E.SUDHA (Fellow in Pediatric Nephrology) DEPT OF PEDIATRIC NEPHROLOGY & DIALYSIS Dr.MEHTA CHILDRENS HOSPITAL
 Dr. E.SUDHA (Fellow in Pediatric Nephrology) DEPT OF PEDIATRIC NEPHROLOGY & DIALYSIS Dr.MEHTA CHILDRENS HOSPITAL CASE HISTORY 4 yrs old previously well boy Born to 2 nd degree consanguinity Fever x 5 days
Dr. E.SUDHA (Fellow in Pediatric Nephrology) DEPT OF PEDIATRIC NEPHROLOGY & DIALYSIS Dr.MEHTA CHILDRENS HOSPITAL CASE HISTORY 4 yrs old previously well boy Born to 2 nd degree consanguinity Fever x 5 days
Clinical Study Eculizumab Therapy Leads to Rapid Resolution of Thrombocytopenia in Atypical Hemolytic Uremic Syndrome
 Hindawi Publishing Corporation Advances in Hematology Volume 214, Article ID 295323, 7 pages http://dx.doi.org/1.1155/214/295323 Clinical Study Therapy Leads to Rapid Resolution of Thrombocytopenia in
Hindawi Publishing Corporation Advances in Hematology Volume 214, Article ID 295323, 7 pages http://dx.doi.org/1.1155/214/295323 Clinical Study Therapy Leads to Rapid Resolution of Thrombocytopenia in
TMA in HUS and TTP: new insights
 TMA in HUS and TTP: new insights Daan Dierickx University Hospitals Leuven, Department of Hematology, Belgium 20th Annual Meeting Belgian Society on Thrombosis and Haemostatis Antwerpen, 22 th November
TMA in HUS and TTP: new insights Daan Dierickx University Hospitals Leuven, Department of Hematology, Belgium 20th Annual Meeting Belgian Society on Thrombosis and Haemostatis Antwerpen, 22 th November
Soliris. Soliris (eculizumab) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.85.11 Subject: Soliris Page: 1 of 5 Last Review Date: September 20, 2018 Soliris Description Soliris
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.85.11 Subject: Soliris Page: 1 of 5 Last Review Date: September 20, 2018 Soliris Description Soliris
Primary causes: Complement dysregulation (50% of non-shiga toxin-producing E. coli ) Secondary causes:
 General department INTRODUCTION The hemolytic uremic syndrome (HUS): microangiopathic hemolytic anemia, thrombocytopenia, and acute kidney injury One of the main causes of acute kidney injury in children
General department INTRODUCTION The hemolytic uremic syndrome (HUS): microangiopathic hemolytic anemia, thrombocytopenia, and acute kidney injury One of the main causes of acute kidney injury in children
New insights in thrombotic microangiopathies : TTP and ahus
 New insights in thrombotic microangiopathies : TTP and ahus Dr Catherine LAMBERT Hematology Cliniques universitaires Saint-Luc Catherine.lambert@uclouvain.be New insights in thrombotic microangiopathies
New insights in thrombotic microangiopathies : TTP and ahus Dr Catherine LAMBERT Hematology Cliniques universitaires Saint-Luc Catherine.lambert@uclouvain.be New insights in thrombotic microangiopathies
Haemolytic uraemic syndrome the story of a whodunit
 Haemolytic uraemic syndrome the story of a whodunit Paul Warwicker Lancashire Teaching Hospitals NHS Trust RCP Kidney for the General Physician Conference Nov 17 Renal thrombotic microangiopathy (TMA)
Haemolytic uraemic syndrome the story of a whodunit Paul Warwicker Lancashire Teaching Hospitals NHS Trust RCP Kidney for the General Physician Conference Nov 17 Renal thrombotic microangiopathy (TMA)
Introduction to pathogenesis and treatment of thrombotic microangiopathies (TMA)
 Introduction to pathogenesis and treatment of thrombotic microangiopathies (TMA) JM.Campistol, Nephrology and Renal Transplant Department, Hospital Clinic, University of Barcelona, Barcelona, Spain. jmcampis@clinic.cat
Introduction to pathogenesis and treatment of thrombotic microangiopathies (TMA) JM.Campistol, Nephrology and Renal Transplant Department, Hospital Clinic, University of Barcelona, Barcelona, Spain. jmcampis@clinic.cat
Atypical hemolytic uremic syndrome. Diana Karpman Department of Pediatrics Lund University
 Atypical hemolytic uremic syndrome Diana Karpman Department of Pediatrics Lund University Image courtesy of Dr. Sabine Leh, Haukeland University Hospital, Bergen Norway Hemolytic Uremic Syndrome Non-immune
Atypical hemolytic uremic syndrome Diana Karpman Department of Pediatrics Lund University Image courtesy of Dr. Sabine Leh, Haukeland University Hospital, Bergen Norway Hemolytic Uremic Syndrome Non-immune
Foodborne Outbreak of E. coli Infections and Hemolytic Uremic Syndrome in Germany, 2011
 Foodborne Outbreak of E. coli Infections and Hemolytic Uremic Syndrome in Germany, 2011 Kirk Smith, DVM, MS, PhD Supervisor Foodborne, Vectorborne and Zoonotic Diseases Unit Minnesota Department of Health
Foodborne Outbreak of E. coli Infections and Hemolytic Uremic Syndrome in Germany, 2011 Kirk Smith, DVM, MS, PhD Supervisor Foodborne, Vectorborne and Zoonotic Diseases Unit Minnesota Department of Health
ahus A PATIENT S GUIDE To learn more about ahus, visit Copyright 2011, Alexion Pharmaceuticals, Inc. All rights reserved.
 To learn more about ahus, visit www.ahussource.com ahus A PATIENT S GUIDE Copyright 2011, Alexion Pharmaceuticals, Inc. All rights reserved. SOL 1169 BECOME EMPOWERED By learning more and taking control
To learn more about ahus, visit www.ahussource.com ahus A PATIENT S GUIDE Copyright 2011, Alexion Pharmaceuticals, Inc. All rights reserved. SOL 1169 BECOME EMPOWERED By learning more and taking control
Trial Name: Eculizumab in Shiga-Toxin producing E. Coli Haemolytic Syndrome (ECUSTEC): A Randomised, Double-Blind, Placebo- Controlled Trial
 Trial Name: Eculizumab in Shiga-Toxin producing E. Coli Haemolytic Syndrome (ECUSTEC): A Randomised, Double-Blind, Placebo- Controlled Trial ECUSTEC Trial Number:..... Patient name:.... You are participating
Trial Name: Eculizumab in Shiga-Toxin producing E. Coli Haemolytic Syndrome (ECUSTEC): A Randomised, Double-Blind, Placebo- Controlled Trial ECUSTEC Trial Number:..... Patient name:.... You are participating
ahus: Facts, Controversies & Treatment Updates Patrick D. Brophy MD Pediatric Nephrology, Dialysis & Transplant University of Iowa
 ahus: Facts, Controversies & Treatment Updates Patrick D. Brophy MD Pediatric Nephrology, Dialysis & Transplant University of Iowa DISCLOSURE STATEMENT I, Patrick Brophy disclose the following relationships.
ahus: Facts, Controversies & Treatment Updates Patrick D. Brophy MD Pediatric Nephrology, Dialysis & Transplant University of Iowa DISCLOSURE STATEMENT I, Patrick Brophy disclose the following relationships.
THROMBOTIC MICROANGIOPATHY. Jun-Ki Park 7/19/11
 THROMBOTIC MICROANGIOPATHY Jun-Ki Park 7/19/11 TMAs are microvascular occlusive disorders characterized by systemic or intrarenal aggregation of platelets, thrombocytopenia, and mechanical injury to erythrocytes.
THROMBOTIC MICROANGIOPATHY Jun-Ki Park 7/19/11 TMAs are microvascular occlusive disorders characterized by systemic or intrarenal aggregation of platelets, thrombocytopenia, and mechanical injury to erythrocytes.
Atypical hemolytic uremic syndrome (ahus)
 Sunday, July 12 10:00-18:30 Registration open I. Atypical hemolytic uremic syndrome (ahus) Chairs: Kronenberg, Speth 13:00 L1 ahus Role of complemet, overview Meri, FI 13:30 L2 ahus Role of lectin pathway
Sunday, July 12 10:00-18:30 Registration open I. Atypical hemolytic uremic syndrome (ahus) Chairs: Kronenberg, Speth 13:00 L1 ahus Role of complemet, overview Meri, FI 13:30 L2 ahus Role of lectin pathway
Initial management of TMA syndromes
 Initial management of TMA syndromes Elie Azoulay, Saint-Louis Hospital, Medical Intensive Care Unit Paris Diderot Sorbonne University Groupe de Recherche Respiratoire en Réanimation Onco-Hématologique
Initial management of TMA syndromes Elie Azoulay, Saint-Louis Hospital, Medical Intensive Care Unit Paris Diderot Sorbonne University Groupe de Recherche Respiratoire en Réanimation Onco-Hématologique
ahus: recent insights and management
 ahus: recent insights and management Steven Van Laecke MD, PhD Renal Division, Ghent University Hospital, Belgium 13 th BANTAO Sarajevo October 8 2017 The complement is everywhere I could benefit from
ahus: recent insights and management Steven Van Laecke MD, PhD Renal Division, Ghent University Hospital, Belgium 13 th BANTAO Sarajevo October 8 2017 The complement is everywhere I could benefit from
A 60 year old woman with altered mental status and thrombotic microangiopathy. Josh Veatch
 A 60 year old woman with altered mental status and thrombotic microangiopathy Josh Veatch Previously healthy 60 year old woman 2 3 months of fatigue following a URI, transient episodes being out of it
A 60 year old woman with altered mental status and thrombotic microangiopathy Josh Veatch Previously healthy 60 year old woman 2 3 months of fatigue following a URI, transient episodes being out of it
Clinical trials in childhood steroid sensitive nephrotic syndrome: the PREDNOS studies
 Clinical Research Facility Central Manchester University Hospitals NHS Foundation Trust Clinical trials in childhood steroid sensitive nephrotic syndrome: the PREDNOS studies Professor Nick Webb DM FRCP
Clinical Research Facility Central Manchester University Hospitals NHS Foundation Trust Clinical trials in childhood steroid sensitive nephrotic syndrome: the PREDNOS studies Professor Nick Webb DM FRCP
THE MULTIPLE FACETS OF THROMBOTIC MICROANGIOPATHIES
 THE MULTIPLE FACETS OF THROMBOTIC MICROANGIOPATHIES Summary of Presentations from the Alexion-Sponsored Symposium, held at the 19 th EHA Congress, Milan, Italy, on 12 th June 2014 Chairperson Pier Mannuccio
THE MULTIPLE FACETS OF THROMBOTIC MICROANGIOPATHIES Summary of Presentations from the Alexion-Sponsored Symposium, held at the 19 th EHA Congress, Milan, Italy, on 12 th June 2014 Chairperson Pier Mannuccio
TMA CASE STUDY. Pamela Harmon, RN & Keturah Tomlin, RN Toronto General Hospital Apheresis Unit
 TMA CASE STUDY Pamela Harmon, RN & Keturah Tomlin, RN Toronto General Hospital Apheresis Unit Cumulative fraction of patients free of events ahus is a catastrophic disease that can result in sudden & progressive
TMA CASE STUDY Pamela Harmon, RN & Keturah Tomlin, RN Toronto General Hospital Apheresis Unit Cumulative fraction of patients free of events ahus is a catastrophic disease that can result in sudden & progressive
HUS and TTP Testing. Kenneth D. Friedman, M.D. Director, Hemostasis Reference Lab BloodCenter of Wisconsin, Milwaukee WI
 HUS and TTP Testing Kenneth D. Friedman, M.D. Director, Hemostasis Reference Lab BloodCenter of Wisconsin, Milwaukee WI Disclosures Relevant Financial Relationships Consultant: Ablynx, Bayer, CSL Behring,
HUS and TTP Testing Kenneth D. Friedman, M.D. Director, Hemostasis Reference Lab BloodCenter of Wisconsin, Milwaukee WI Disclosures Relevant Financial Relationships Consultant: Ablynx, Bayer, CSL Behring,
COMPLEMENT INHIBITORS (SOLIRIS & ULTOMIRIS )
 COMPLEMENT INHIBITORS (SOLIRIS & ULTOMIRIS ) Protocol: PHA029 Effective Date: March 1, 2019 Table of Contents Page COVERAGE RATIONALE... 1 US FOOD AND DRUG ADMINISTRATION (FDA)... 4 BACKGROUND... 4 APPLICABLE
COMPLEMENT INHIBITORS (SOLIRIS & ULTOMIRIS ) Protocol: PHA029 Effective Date: March 1, 2019 Table of Contents Page COVERAGE RATIONALE... 1 US FOOD AND DRUG ADMINISTRATION (FDA)... 4 BACKGROUND... 4 APPLICABLE
Soliris (eculizumab) DRUG.00050
 Market DC Soliris (eculizumab) DRUG.00050 Override(s) Prior Authorization Approval Duration 1 year Medications Soliris (eculizumab) APPROVAL CRITERIA Paroxysmal Nocturnal Hemoglobinuria I. Initiation of
Market DC Soliris (eculizumab) DRUG.00050 Override(s) Prior Authorization Approval Duration 1 year Medications Soliris (eculizumab) APPROVAL CRITERIA Paroxysmal Nocturnal Hemoglobinuria I. Initiation of
Medical Policy An independent licensee of the Blue Cross Blue Shield Association
 Soliris (eculizumab) Page 1 of 11 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Soliris (eculizumab) Prime Therapeutics will review Prior Authorization requests
Soliris (eculizumab) Page 1 of 11 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Soliris (eculizumab) Prime Therapeutics will review Prior Authorization requests
DR V PHILIP CLINICAL HAEMATOLOGY UNIT CHRIS HANI BARAGWANATH ACADEMIC HOSPITAL
 DR V PHILIP CLINICAL HAEMATOLOGY UNIT CHRIS HANI BARAGWANATH ACADEMIC HOSPITAL Rare but fatal disease if unrecognized and untreated Incidence about 1: 1 million in the USA Female preponderance of 2:1 Part
DR V PHILIP CLINICAL HAEMATOLOGY UNIT CHRIS HANI BARAGWANATH ACADEMIC HOSPITAL Rare but fatal disease if unrecognized and untreated Incidence about 1: 1 million in the USA Female preponderance of 2:1 Part
w ahus pathology is linked to dysregulation of the alternative complement pathway.
 ahus - Pathogenesis, Etiology, and Clinical Advances Craig B. Langman MD The Isaac A Abt MD Professor of Kidney Diseases Feinberg School of Medicine, Northwestern University Head, Kidney Diseases The Ann
ahus - Pathogenesis, Etiology, and Clinical Advances Craig B. Langman MD The Isaac A Abt MD Professor of Kidney Diseases Feinberg School of Medicine, Northwestern University Head, Kidney Diseases The Ann
Thrombotic Microangiopathies (TMA) / TTP/HUS/αHUS Pathology & Molecular. Genetics
 Thrombotic Microangiopathies (TMA) / TTP/HUS/αHUS Pathology & Molecular Genetics Helen Liapis, M.D. Senior Consultant Arkana Labs Professor of Pathology & Immunology. retired Washington University School
Thrombotic Microangiopathies (TMA) / TTP/HUS/αHUS Pathology & Molecular Genetics Helen Liapis, M.D. Senior Consultant Arkana Labs Professor of Pathology & Immunology. retired Washington University School
Pathogenetic mechanisms in atypical HUS
 Pathogenetic mechanisms in atypical HUS Seppo Meri University & University Hospital Helsinki, Finland Antalya, Turkey Photo: Markku Kallio 21.10.2017 HUS after eating Kebab - 22 yo female - watery diarrhea
Pathogenetic mechanisms in atypical HUS Seppo Meri University & University Hospital Helsinki, Finland Antalya, Turkey Photo: Markku Kallio 21.10.2017 HUS after eating Kebab - 22 yo female - watery diarrhea
A 23 year old Caucasian male presented with shortness of breath, hypertension, bloody sputum, and a history of drug abuse (confirmed by urinalysis).
 A 23 year old Caucasian male presented with shortness of breath, hypertension, bloody sputum, and a history of drug abuse (confirmed by urinalysis). He was found to have severe kidney injury requiring
A 23 year old Caucasian male presented with shortness of breath, hypertension, bloody sputum, and a history of drug abuse (confirmed by urinalysis). He was found to have severe kidney injury requiring
Microangiopatia trombotica (MAT) e Sindrome emolitico-uremica atipica (SEUa): Basi patogenetiche, inquadramento diagnostico e principi del
 Microangiopatia trombotica (MAT) e Sindrome emolitico-uremica atipica (SEUa): Basi patogenetiche, inquadramento diagnostico e principi del trattamento Vincenzo Montinaro U.O. Nefrologia Azienda Ospedaliera
Microangiopatia trombotica (MAT) e Sindrome emolitico-uremica atipica (SEUa): Basi patogenetiche, inquadramento diagnostico e principi del trattamento Vincenzo Montinaro U.O. Nefrologia Azienda Ospedaliera
RaDaR Inclusion and Exclusion Criteria. Diagnosis Inclusion Criteria Exclusion Criteria. Alport Syndrome definite or probable
 Alport Syndrome and Type IV collagenopathies APRT Deficiency Alport Syndrome definite or probable Alport carrier definite or probable Thin basement membrane nephropathy APRT Deficiency confirmed Abolished
Alport Syndrome and Type IV collagenopathies APRT Deficiency Alport Syndrome definite or probable Alport carrier definite or probable Thin basement membrane nephropathy APRT Deficiency confirmed Abolished
DIAGNOSTIC CHALLENGES IN THROMBOTIC MICROANGIOPATHIES
 DIAGNOSTIC CHALLENGES IN THROMBOTIC MICROANGIOPATHIES Summary of Presentations from the Alexion-Sponsored Symposium, held at the 51 st ERA EDTA Congress, Amsterdam, the Netherlands, on 1 st June 2014 Chairperson
DIAGNOSTIC CHALLENGES IN THROMBOTIC MICROANGIOPATHIES Summary of Presentations from the Alexion-Sponsored Symposium, held at the 51 st ERA EDTA Congress, Amsterdam, the Netherlands, on 1 st June 2014 Chairperson
Spectrum of complement-mediated thrombotic microangiopathies after kidney transplantation
 Spectrum of complement-mediated thrombotic microangiopathies after kidney transplantation Marius Miglinas Vilnius university hospital: Nephrology center, Center of Rare Kidney Diseases Vilnius university
Spectrum of complement-mediated thrombotic microangiopathies after kidney transplantation Marius Miglinas Vilnius university hospital: Nephrology center, Center of Rare Kidney Diseases Vilnius university
Thrombotic Microangiopathy (TMA) The Clinical Facets of TMA
 International Consensus on Management Atypical Hemolytic Uremic Syndrome in Children Loirat C. et al. Pediatr Nephrol 31: 15-39, 2016 Ruth A. McDonald, MD Professor and Vice Chair Clinical Affairs Department
International Consensus on Management Atypical Hemolytic Uremic Syndrome in Children Loirat C. et al. Pediatr Nephrol 31: 15-39, 2016 Ruth A. McDonald, MD Professor and Vice Chair Clinical Affairs Department
Soliris and You. Your Guide To Living With ahus. INDICATION & IMPORTANT SAFETY INFORMATION FOR SOLIRIS (eculizumab)
 INDICATION & IMPORTANT SAFETY INFORMATION FOR SOLIRIS (eculizumab) INDICATION What is SOLIRIS? SOLIRIS is a prescription medicine called a monoclonal antibody. SOLIRIS is used to treat: adults and children
INDICATION & IMPORTANT SAFETY INFORMATION FOR SOLIRIS (eculizumab) INDICATION What is SOLIRIS? SOLIRIS is a prescription medicine called a monoclonal antibody. SOLIRIS is used to treat: adults and children
PRODUCT INFORMATION. SOLIRIS Concentrated Solution for Intravenous Infusion
 PRODUCT INFORMATION SOLIRIS Concentrated Solution for Intravenous Infusion WARNING: SERIOUS MENINGOCOCCAL INFECTION SOLIRIS increases the risk of meningococcal infections Vaccinate patients with a meningococcal
PRODUCT INFORMATION SOLIRIS Concentrated Solution for Intravenous Infusion WARNING: SERIOUS MENINGOCOCCAL INFECTION SOLIRIS increases the risk of meningococcal infections Vaccinate patients with a meningococcal
Five papers that influenced my practice. Stephen Marks Consultant Paediatric Nephrologist
 Five papers that influenced my practice Stephen Marks Consultant Paediatric Nephrologist Great Ormond Street Hospital for Children and UCL Institute of Child Health, London, UK. Nephrology for the General
Five papers that influenced my practice Stephen Marks Consultant Paediatric Nephrologist Great Ormond Street Hospital for Children and UCL Institute of Child Health, London, UK. Nephrology for the General
Thrombotic microangiopathy and associated renal disorders*
 Nephrol Dial Transplant (2012) 27: 2673 2685 doi: 10.1093/ndt/gfs279 NDT Perspectives Thrombotic microangiopathy and associated renal disorders* Thomas Barbour 1, Sally Johnson 2, Solomon Cohney 3 and
Nephrol Dial Transplant (2012) 27: 2673 2685 doi: 10.1093/ndt/gfs279 NDT Perspectives Thrombotic microangiopathy and associated renal disorders* Thomas Barbour 1, Sally Johnson 2, Solomon Cohney 3 and
Things to never miss in the office. Brett Houston MD FRCPC (PYG-5, hematology) Leonard Minuk MD FRCPC
 Things to never miss in the office Brett Houston MD FRCPC (PYG-5, hematology) Leonard Minuk MD FRCPC Presenter Disclosure Faculty / Speaker s name: Brett Houston / Leonard Minuk Relationships with commercial
Things to never miss in the office Brett Houston MD FRCPC (PYG-5, hematology) Leonard Minuk MD FRCPC Presenter Disclosure Faculty / Speaker s name: Brett Houston / Leonard Minuk Relationships with commercial
ISTITUTO DI RICERCHE FARMACOLOGICHE MARIO NEGRI CLINICAL RESEARCH CENTER ALDO E FOR CELE RARE DACCO DISEASES ALDO E CELE DACCO
 ISTITUTO DI RICERCHE FARMACOLOGICHE MARIO NEGRI CENTRO MARIO DI NEGRI RICERCHE INSTITUTE CLINICHE FOR PHARMACOLOGICAL PER LE MALATTIE RESEARCH RARE CLINICAL RESEARCH CENTER ALDO E FOR CELE RARE DACCO DISEASES
ISTITUTO DI RICERCHE FARMACOLOGICHE MARIO NEGRI CENTRO MARIO DI NEGRI RICERCHE INSTITUTE CLINICHE FOR PHARMACOLOGICAL PER LE MALATTIE RESEARCH RARE CLINICAL RESEARCH CENTER ALDO E FOR CELE RARE DACCO DISEASES
Non-immune acquired haemolytic anaemias. Dr.Maysem
 Non-immune acquired haemolytic anaemias Dr.Maysem Causes of Non-immune acquired haemolytic anaemias. Infections Infections can cause haemolysis in a variety of ways: -They may precipitate an acute haemolytic
Non-immune acquired haemolytic anaemias Dr.Maysem Causes of Non-immune acquired haemolytic anaemias. Infections Infections can cause haemolysis in a variety of ways: -They may precipitate an acute haemolytic
Medication Prior Authorization Form
 Policy Number: 1054 Policy History Approve Date: 06/01/2018 Effective Date: 06/01/2018 Preauthorization All Plans Benefit plans vary in coverage and some plans may not provide coverage for certain service(s)
Policy Number: 1054 Policy History Approve Date: 06/01/2018 Effective Date: 06/01/2018 Preauthorization All Plans Benefit plans vary in coverage and some plans may not provide coverage for certain service(s)
Guideline. Abstract. Key words
 bs_bs_banner Pediatrics International (2014) 56, 1 5 doi: 10.1111/ped.12274 Guideline Diagnostic criteria for atypical hemolytic uremic syndrome proposed by the Joint Committee of the Japanese Society
bs_bs_banner Pediatrics International (2014) 56, 1 5 doi: 10.1111/ped.12274 Guideline Diagnostic criteria for atypical hemolytic uremic syndrome proposed by the Joint Committee of the Japanese Society
Soliris (eculizumab, rmc) Section 100 Restriction Criteria
 Soliris (eculizumab, rmc) Section 100 Restriction Criteria A guide to initiation and ongoing access to (Section 100) funded Soliris for the treatment of patients with atypical Haemolytic Uraemic Syndrome
Soliris (eculizumab, rmc) Section 100 Restriction Criteria A guide to initiation and ongoing access to (Section 100) funded Soliris for the treatment of patients with atypical Haemolytic Uraemic Syndrome
Eculizumab in ahus: where do we stand in Prof. Fadi Fakhouri Dept. of nephrology and immunology, CHU de Nantes.
 Eculizumab in ahus: where do we stand in 2016 Prof. Fadi Fakhouri Dept. of nephrology and immunology, CHU de Nantes. INSERM UMR S-1064 Thrombotic microangiopathy: evolving concepts ST-HUS Coagulation mediated-tma
Eculizumab in ahus: where do we stand in 2016 Prof. Fadi Fakhouri Dept. of nephrology and immunology, CHU de Nantes. INSERM UMR S-1064 Thrombotic microangiopathy: evolving concepts ST-HUS Coagulation mediated-tma
Extrarenal involvement in pediatric ahus Kosan C et al
 Case Report J Ped. Nephrology 2014;2(4):154-157 http://journals.sbmu.ac.ir/jpn Atypical Hemolytic Uremic Syndrome with Severe Extrarenal Manifestations: a Case Report How to Cite This Article: KOŞAN C,
Case Report J Ped. Nephrology 2014;2(4):154-157 http://journals.sbmu.ac.ir/jpn Atypical Hemolytic Uremic Syndrome with Severe Extrarenal Manifestations: a Case Report How to Cite This Article: KOŞAN C,
Les microangiopathies thrombotiques Quoi de neuf?
 Les microangiopathies thrombotiques Quoi de neuf? Société des Sciences Vasculaires du Québec 16 Septembre 2016 Anne-Laure Lapeyraque CHU Sainte Justine, Montréal anne.laure.lapeyraque@umontreal.ca Objectifs
Les microangiopathies thrombotiques Quoi de neuf? Société des Sciences Vasculaires du Québec 16 Septembre 2016 Anne-Laure Lapeyraque CHU Sainte Justine, Montréal anne.laure.lapeyraque@umontreal.ca Objectifs
National Audit of CKD in Primary Care
 National Audit of CKD in Primary Care David C Wheeler Royal Free Campus University College London d.wheeler@ucl.ac.uk Kidney for General Physicians RCP London 24 th November 2017 Who looks after CKD patients
National Audit of CKD in Primary Care David C Wheeler Royal Free Campus University College London d.wheeler@ucl.ac.uk Kidney for General Physicians RCP London 24 th November 2017 Who looks after CKD patients
Novel aspects of atypical haemolytic uraemic syndrome and the role of eculizumab
 Nephrol Dial Transplant (2014) 29: iv131 iv141 doi: 10.1093/ndt/gfu235 Full Review Novel aspects of atypical haemolytic uraemic syndrome and the role of eculizumab Jacobien C. Verhave 1, Jack F.M. Wetzels
Nephrol Dial Transplant (2014) 29: iv131 iv141 doi: 10.1093/ndt/gfu235 Full Review Novel aspects of atypical haemolytic uraemic syndrome and the role of eculizumab Jacobien C. Verhave 1, Jack F.M. Wetzels
HUS-MPGN-TTP. & related disorders
 ES-PCR 4 th International Conference HUS-MPGN-TTP & related disorders Current diagnosis and therapy of hemolytic uremic syndrome (HUS), membranoproliferative glomerulonephritis (MPGN), thrombotic thrombocytopenic
ES-PCR 4 th International Conference HUS-MPGN-TTP & related disorders Current diagnosis and therapy of hemolytic uremic syndrome (HUS), membranoproliferative glomerulonephritis (MPGN), thrombotic thrombocytopenic
Thrombotic thrombocytopenic purpura: a look at the future
 Thrombotic thrombocytopenic purpura: a look at the future Andrea Artoni, MD Ph.D. Angelo Bianchi Bonomi Hemophilia and Thrombosis Center IRCCS Ca Granda Ospedale Maggiore Policlinico Milan, Italy andrea.artoni@policlinico.mi.it
Thrombotic thrombocytopenic purpura: a look at the future Andrea Artoni, MD Ph.D. Angelo Bianchi Bonomi Hemophilia and Thrombosis Center IRCCS Ca Granda Ospedale Maggiore Policlinico Milan, Italy andrea.artoni@policlinico.mi.it
A 36 year old previously healthy female develops fever and ruising
 Renal vascular diseases CPC G.A. Appel MD M.B. Stokes MD Case 1 A 36 year old previously healthy female develops fever and bruising. She goes to her LMD and CBC shows plats 15 K, Hct 28%, normal PT and
Renal vascular diseases CPC G.A. Appel MD M.B. Stokes MD Case 1 A 36 year old previously healthy female develops fever and bruising. She goes to her LMD and CBC shows plats 15 K, Hct 28%, normal PT and
Chronic Kidney Disease. Paul Cockwell Queen Elizabeth Hospital Birmingham
 Chronic Kidney Disease Paul Cockwell Queen Elizabeth Hospital Birmingham Paradigms for chronic disease 1. Acute and chronic disease is closely linked 2. Stratify risk and tailor interventions around failure
Chronic Kidney Disease Paul Cockwell Queen Elizabeth Hospital Birmingham Paradigms for chronic disease 1. Acute and chronic disease is closely linked 2. Stratify risk and tailor interventions around failure
Haemolytic uraemic syndrome
 Review Haemolytic uraemic syndrome doi: 10.1111/joim.12546 Diana Karpman, Sebastian Loos, Ramesh Tati & Ida Arvidsson From the Department of Pediatrics, Clinical Sciences Lund, Lund University, Lund, Sweden
Review Haemolytic uraemic syndrome doi: 10.1111/joim.12546 Diana Karpman, Sebastian Loos, Ramesh Tati & Ida Arvidsson From the Department of Pediatrics, Clinical Sciences Lund, Lund University, Lund, Sweden
Thrombotic microangiopathies and antineoplastic agents
 Thrombotic microangiopathies and antineoplastic agents Paul Coppo paul.coppo@aphp.fr Service d Hématologie - Hôpital Saint-Antoine AP-HP et Université Pierre & Marie Curie Centre de Référence des Microangiopathies
Thrombotic microangiopathies and antineoplastic agents Paul Coppo paul.coppo@aphp.fr Service d Hématologie - Hôpital Saint-Antoine AP-HP et Université Pierre & Marie Curie Centre de Référence des Microangiopathies
Medical Policy. MP Eculizumab (Soliris) Related Policies None. Last Review: 01/24/2019 Effective Date: 04/25/2019 Section: Prescription Drug
 Medical Policy Last Review: 01/24/2019 Effective Date: 04/25/2019 Section: Prescription Drug Related Policies None DISCLAIMER Our medical policies are designed for informational purposes only and are not
Medical Policy Last Review: 01/24/2019 Effective Date: 04/25/2019 Section: Prescription Drug Related Policies None DISCLAIMER Our medical policies are designed for informational purposes only and are not
Year 2004 Paper one: Questions supplied by Megan
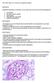 QUESTION 53 Endothelial cell pathology on renal biopsy is most characteristic of which one of the following diagnoses? A. Pre-eclampsia B. Haemolytic uraemic syndrome C. Lupus nephritis D. Immunoglobulin
QUESTION 53 Endothelial cell pathology on renal biopsy is most characteristic of which one of the following diagnoses? A. Pre-eclampsia B. Haemolytic uraemic syndrome C. Lupus nephritis D. Immunoglobulin
Clinical Commissioning Policy Statement: Eculizumab for atypical haemolytic uraemic syndrome. September 2013 Reference: E03/PS(HSS)/a.
 Clinical Commissioning Policy Statement: Eculizumab for atypical haemolytic uraemic syndrome September 2013 Reference: E03/PS(HSS)/a England 1 NHS England INFORMATION READER BOX Directorate Medical Operations
Clinical Commissioning Policy Statement: Eculizumab for atypical haemolytic uraemic syndrome September 2013 Reference: E03/PS(HSS)/a England 1 NHS England INFORMATION READER BOX Directorate Medical Operations
PNH ahus. Dosing and Administration. For Paroxysmal Nocturnal Hemoglobinuria (PNH) and atypical Hemolytic Uremic Syndrome (ahus) patients
 For Paroxysmal Nocturnal Hemoglobinuria (PNH) and atypical Hemolytic Uremic Syndrome (ahus) patients PNH ahus Dosing and Administration Soliris is indicated for the treatment of patients with paroxysmal
For Paroxysmal Nocturnal Hemoglobinuria (PNH) and atypical Hemolytic Uremic Syndrome (ahus) patients PNH ahus Dosing and Administration Soliris is indicated for the treatment of patients with paroxysmal
Launch Meeting 3 rd April 2014, Lucas House, Birmingham
 Angiotensin Converting Enzyme inhibitor (ACEi) / Angiotensin Receptor Blocker (ARB) To STOP OR Not in Advanced Renal Disease Launch Meeting 3 rd April 2014, Lucas House, Birmingham Prof Sunil Bhandari
Angiotensin Converting Enzyme inhibitor (ACEi) / Angiotensin Receptor Blocker (ARB) To STOP OR Not in Advanced Renal Disease Launch Meeting 3 rd April 2014, Lucas House, Birmingham Prof Sunil Bhandari
Some renal vascular disorders
 Some renal vascular disorders Introduction Nearly all diseases of the kidney involve the renal blood vessels secondarily We will discuss: -Hypertension (arterionephrosclerosis in benign HTN & hyperplastic
Some renal vascular disorders Introduction Nearly all diseases of the kidney involve the renal blood vessels secondarily We will discuss: -Hypertension (arterionephrosclerosis in benign HTN & hyperplastic
Therapeutic Complement Intervention Michael Kirschfink, Institute of Immunology, University of Heidelberg, Germany
 Therapeutic Complement Intervention Michael Kirschfink, Institute of Immunology, University of Heidelberg, Germany Complement-mediated diseases Therapeutic intervention The complement system is involved
Therapeutic Complement Intervention Michael Kirschfink, Institute of Immunology, University of Heidelberg, Germany Complement-mediated diseases Therapeutic intervention The complement system is involved
Not So Benign Hematology Aplastic anemia, Paroxysmal Nocturnal Hemoglobinuria, atypical Hemolytic Uremic Syndrome, Thrombotic Thrombocytopenic Purpura
 Not So Benign Hematology Aplastic anemia, Paroxysmal Nocturnal Hemoglobinuria, atypical Hemolytic Uremic Syndrome, Thrombotic Thrombocytopenic Purpura Robert A. Brodsky, MD Johns Hopkins Family Professor
Not So Benign Hematology Aplastic anemia, Paroxysmal Nocturnal Hemoglobinuria, atypical Hemolytic Uremic Syndrome, Thrombotic Thrombocytopenic Purpura Robert A. Brodsky, MD Johns Hopkins Family Professor
A Rational Approach to Evaluation of Thrombotic Microangiopathy
 A Rational Approach to Evaluation of Thrombotic Microangiopathy An Algorithmic Approach C. Christopher Hook, MD for the Complement Alternative Pathway Thrombotic Micro- Angiopathy (CAP-TMA) Disease-Oriented
A Rational Approach to Evaluation of Thrombotic Microangiopathy An Algorithmic Approach C. Christopher Hook, MD for the Complement Alternative Pathway Thrombotic Micro- Angiopathy (CAP-TMA) Disease-Oriented
eculizumab, 300mg concentrate for solution for infusion (Soliris ) SMC No. (436/07) Alexion Pharma UK Ltd
 eculizumab, 300mg concentrate for solution for infusion (Soliris ) SMC No. (436/07) Alexion Pharma UK Ltd 8 October 2010 (Amended 11 July 2011) The Scottish Medicines Consortium (SMC) has completed its
eculizumab, 300mg concentrate for solution for infusion (Soliris ) SMC No. (436/07) Alexion Pharma UK Ltd 8 October 2010 (Amended 11 July 2011) The Scottish Medicines Consortium (SMC) has completed its
Guideline on the clinical management of Henoch Schonlein Purpura (HSP)
 Guideline on the clinical management of Henoch Schonlein Purpura (HSP) Purpose To ensure a standardised approach in the management of children with HSP in southern Derbyshire. Scope The scope of this guideline
Guideline on the clinical management of Henoch Schonlein Purpura (HSP) Purpose To ensure a standardised approach in the management of children with HSP in southern Derbyshire. Scope The scope of this guideline
CDX1135: A Drug Trial for DDD
 CDX1135: A Drug Trial for DDD In late 2012, the Food and Drug Administration (FDA) approved a drug trial to test CDX-1135 in pediatric and adult patients with Dense Deposit Disease (DDD). This is the FIRST-EVER
CDX1135: A Drug Trial for DDD In late 2012, the Food and Drug Administration (FDA) approved a drug trial to test CDX-1135 in pediatric and adult patients with Dense Deposit Disease (DDD). This is the FIRST-EVER
PDF hosted at the Radboud Repository of the Radboud University Nijmegen
 PDF hosted at the Radboud Repository of the Radboud University Nijmegen The following full text is a publisher's version. For additional information about this publication click this link. http://hdl.handle.net/2066/140173
PDF hosted at the Radboud Repository of the Radboud University Nijmegen The following full text is a publisher's version. For additional information about this publication click this link. http://hdl.handle.net/2066/140173
Management of Nephrotic Syndrome
 Management of Nephrotic Syndrome 1. Introduction Incidence 2-4/100,000. Boys > girls 3:2; age of onset 2-6 years 80% of cases in children is due to minimal change (MCD) of which 80% will respond to steroid
Management of Nephrotic Syndrome 1. Introduction Incidence 2-4/100,000. Boys > girls 3:2; age of onset 2-6 years 80% of cases in children is due to minimal change (MCD) of which 80% will respond to steroid
PNH ahus gmg. Dosing and Administration Guide
 Injection for Intravenous Use PNH ahus gmg For Paroxysmal Nocturnal Hemoglobinuria (PNH) and atypical Hemolytic Uremic Syndrome (ahus), and generalized Myasthenia Gravis (gmg) patients Dosing and Administration
Injection for Intravenous Use PNH ahus gmg For Paroxysmal Nocturnal Hemoglobinuria (PNH) and atypical Hemolytic Uremic Syndrome (ahus), and generalized Myasthenia Gravis (gmg) patients Dosing and Administration
* Renal insufficiencies
 Thrombotic Thrombocytopenic Purpura Behzad Poopak, DCLS PhD. Tehran medical Branch Islamic Azad university bpoopak@yahoo.com Case Summary Ms. X, a 35-year year-old woman Complained of weakness, low grade
Thrombotic Thrombocytopenic Purpura Behzad Poopak, DCLS PhD. Tehran medical Branch Islamic Azad university bpoopak@yahoo.com Case Summary Ms. X, a 35-year year-old woman Complained of weakness, low grade
PREGNANCY ASSOCIATED THROMBOTIC THROMBOCYTOPENIC PURPURA AND ACUTE KIDNEY INJURY
 VII, 2013, 2 33 A, PREGNANCY ASSOCIATED THROMBOTIC THROMBOCYTOPENIC PURPURA AND ACUTE KIDNEY INJURY M. Lubomirova Clinic of Nephrology, University Hospital Aleksandrovska So a : ( ), /HELLP, (AFLP) (TTP)
VII, 2013, 2 33 A, PREGNANCY ASSOCIATED THROMBOTIC THROMBOCYTOPENIC PURPURA AND ACUTE KIDNEY INJURY M. Lubomirova Clinic of Nephrology, University Hospital Aleksandrovska So a : ( ), /HELLP, (AFLP) (TTP)
Atypical hemolytic uremic syndrome (ahus) is a progressive
 www.kidney-international.org Eculizumab is a safe and effective treatment in pediatric patients with atypical hemolytic uremic syndrome OPEN Larry A. Greenbaum 1, Marc Fila 2, Gianluigi Ardissino 3, Samhar
www.kidney-international.org Eculizumab is a safe and effective treatment in pediatric patients with atypical hemolytic uremic syndrome OPEN Larry A. Greenbaum 1, Marc Fila 2, Gianluigi Ardissino 3, Samhar
Soliris (eculizumab): Physician s guide to prescribing for patients with paroxysmal nocturnal haemoglobinuria (PNH)
 Soliris (eculizumab): Physician s guide to prescribing for patients with paroxysmal nocturnal haemoglobinuria (PNH) CONTENTS PAGE WHAT IS SOLIRIS? SOLIRIS INDICATIONS IMPORTANT SAFETY INFORMATION SOLIRIS
Soliris (eculizumab): Physician s guide to prescribing for patients with paroxysmal nocturnal haemoglobinuria (PNH) CONTENTS PAGE WHAT IS SOLIRIS? SOLIRIS INDICATIONS IMPORTANT SAFETY INFORMATION SOLIRIS
3/6/2017. Prevention of Complement Activation and Antibody Development: Results from the Duet Trial
 Prevention of Complement Activation and Antibody Development: Results from the Duet Trial Jignesh Patel MD PhD FACC FRCP Medical Director, Heart Transplant Cedars-Sinai Heart Institute Disclosures Name:
Prevention of Complement Activation and Antibody Development: Results from the Duet Trial Jignesh Patel MD PhD FACC FRCP Medical Director, Heart Transplant Cedars-Sinai Heart Institute Disclosures Name:
Henoch Schonlein Purpura
 CHILDREN S SERVICES Henoch Schonlein Purpura Definition A vasculitic syndrome of small vessels classically characterised by a purpuric rash, abdominal pain, arthritis, and nephritis. Platelet count and
CHILDREN S SERVICES Henoch Schonlein Purpura Definition A vasculitic syndrome of small vessels classically characterised by a purpuric rash, abdominal pain, arthritis, and nephritis. Platelet count and
An international consensus approach to the management of atypical hemolytic uremic syndrome in children
 DOI 10.1007/s00467-015-3076-8 REVIEW An international consensus approach to the management of atypical hemolytic uremic syndrome in children Chantal Loirat & Fadi Fakhouri & Gema Ariceta & Nesrin Besbas
DOI 10.1007/s00467-015-3076-8 REVIEW An international consensus approach to the management of atypical hemolytic uremic syndrome in children Chantal Loirat & Fadi Fakhouri & Gema Ariceta & Nesrin Besbas
