COMBINATION VACCINE STANDING ORDER
|
|
|
- Brian Gordon
- 5 years ago
- Views:
Transcription
1 VAN BUREN/CASS DSTRCT HEALTH DEPARTMENT COMBNATON VACCNE STANDNG ORDER t is the policy of the Van Buren/Cass District Health Department to follow all the recommendations of the Advisory Committee on mmunization Practices (ACP) when administering vaccines. The Michigan Department of Community Health (MDCH) has developed "Quick Look" guidelines incorporating ACP recommendations to assist in the use of various vaccines (including combination vaccines). Combination vaccines may be used when each of the single products are due to be administered. The antigen requiring the longest minimum interval will determine the spacing between doses. Refer to the appropriate "Quick Look" sheet for the combination vaccine. Note that there may be limitations as to the number of doses that may be given, minimum and maximum ages and schedule variations. Refer to the specific antigen standing orders for contraindications and precautions when administering combination vaccines. The Multiple Vaccines Vaccine nformation Statement (VS) may be used, if appropriate, or the separate VSs for each antigen must be given as required by US Code 300aa and CDC. REFERENCES 1. then search for "quick looks" DSTRBUTON mmunization Manual Frederick A. Joha Medical Director e Date :\Admin\R JOHANSEN\OPPM\ VBCHD\StandingOrders\Current\CombinationVaccine.doc Pagel of 1
2 -.~ -.-~ -.-~ -- :1~ ~~.!! Quick Reference to Combination and Reconstituted Vaccines: Childhood Hiberix, GSK Hib 12 months thru 59 months - 12 months and MMR l, Merck_ MMR older 12 months and Varivax, Merck Var older, (Highlight Vaccines in Your Refrigerator and Post) COMBNATON VACCNES Brand Name What it contains Use for Ages: Use for Dose: Administration Tips Draw up the DTaP/lPV liquid (diluent) Pentacel, SP DTaP, PV, Hib 6 weeks thru 1,2,3 or4 of DTaP, PVor Add diluent to the Hib vial; shake well 4 years Hib Administer within 30 minutes; give 1M 6 weeks thru 1,2, or 3 of PV or DTaP; Premixed Pediarix, GSK DTaP, PV, Hep B 6 years any dose of hep B Shake well before administering; give 1M 5th dose of DTaP', Premixed Kinrix, GSK DTaP,PV 4 to 6lears 4th dose of PV Shake well before administering; give 1M weeks thru Premixed Comvax, Merck Hep B, Hib 4 years Any dose of hep B or Hib Shake well before administering; give 1M MMRV Draw up "diluent for Merck vaccines" ProQuad, Merck (Measles, Mumps, 12 months thru 1 or 2 of MMR and Var Add diluent to MMRV vial; shake well..._._- J3@ella,Varicella) ~?years ~Aclminister vvlthin 30 minutes; give SC -.. -~ RECONSTTUTED VACCNES Brand name What it contains Use for Ages: Use for Dose: Administration Tips 6 weeks thru Draw up diluent packaged with Hib vial ActHB, SP Hib 4 years Any dose of Hib Add diluent to Hib vial; shake well; give 1M Add diluent from packaged pre-filled Only the booster (final) syringe to Hib vial; leave needle inserted dose of Hib series Shake well; redraw into syringe; give 1M Draw up "diluent for Merck vaccines" 1 or 2 of MMR Add diluent to MMR vial;, shake well; give SC 1 or 2 of Var Draw up "diluent for Merck vaccines" Add diluent to Varicella vial; shake well. Administer within 30 minutes; give SC Rotarix, GSK Rotavirus (RV1) 6weeks thru 7 months Any dose of RV Use diluent in pre-filled oral applicator Add to RV1 vial; shake; withdraw; give orally --. Avoid medication errors! Use only the diluent that is packaged or sent with each specific vaccine-don't use any other liquid December 18, 2009
3 A Quick Look at Using DTaP/PV (KNRXTM) ndications for Use and Schedule Approved for. Routine schedule of one dose at 4-6 years Second booster dose of DTaP (5 th dose} and PV (4th dose) Ages 4 years through 6years (6 years, 364 days) Do not use: For dose 1, 2, 3, or 4of DTaP or dose 1, 2. or 3 of PV n children 7years of age and older Make sure minimum age and minimum intervals are met: Minimum age for the 5 th dose ofdtap is 4 years Minimum interval between dose 4 &5of DTaP is 6months [.ntramuscular (1M) injection in the deltoid of the arm or anterolateral thigh 1inch needle; gauge Professional judgment is appropriate when selecting needle length and site Can be given with other vaccines. at the same visit (use separate sites; space at least 1inch apart) Store in the refrigerator between F (2..a C) Do NOT freeze Keep in the original box Shake well before using KNRX CONTRANDCA,.ONS An anaphylactic reaction to a prior dose of DTaPlPV (KNRX n,,). DTaP or PV An anaphylactic reaction to acomponent of DTaP/lPV (KNRXTM) including neomycin or polymyxin B Encephalopathy not due to another cause occurring within 7days after vaccination with a pertussis-containing vaccine Progressive neurologic disorder (such as infantile spasms, uncontrolled epilepsy or progressive encephalopathy) PRECAUTONS Moderate to severe acute illness Guillain-Barre syndrome within 6weeks of aprior vaccine containing tetanus toxoid Precautions specific to DTaP vaccine. Temperature greater than or equal to 105 Q Fwithin 48 hours of vaccination with no other identifiable cause Collapse or shock-like state within 48 hours of vaccination Persistent inconsolable crying lasting more than 3 hours within 48 hours of vaccination Convulsions with or without afever within 3 days of vaccination FURTHER PONTS. The second booster dose of DTaP (5 th dose) is not necessary if the 4th dose of DTaP is administered on or after the ~~~. DTaPPV (KNRXTM) does not contain a preservative. After removal of the dose, any remaining vaccine should be discarded. The combined DTaPlPV vaccine may be used when any component of the vaccine is indicated, and if the other components are not contraindicated There is not aseparate Vaccine nformation Statement (VS) for DTaPPV (KNRXTM). Use the current VSs for DTaP and PV. or the Multi-vaccine VS that include information about the Michigan Care mprovement Registry (MCR). VSs with MCR information are available at or at your local health department. Document as "DTaP/PV' in MCR, on the vaccine administration record &immunization record card. Publicly purchased DTapnpV {KNRXTM} can be administered to eligible children 4through 6years of age through the Vaccines for Children "(VFC) Program in private providers' offices. Eligible children include those who are uninsured, underinsured, Medicaid eligible, Native American or Alaskan Natives. Contact your local health department for more information For additional information, refer to YFC Progl1llll Resdu!ioos 011 "Vaccines 10 Prevent Diphtheria, Tetanus and Pertussis" and "Vaccines to Prevent Poliomyelitis" and the ACP Recommendations 011 the use of DTaP and PV vaccines,located at hltptlwww.cdc.gcn/vaccineslrecs January
4 A Quick Look at Using DTaP/PV/Hep B (Pediarix ) ndications for Use and Schedule Approved for: Routine schedule 2, 4, 6 months of age Dose 1, 2, 3of DTaP; dose 1, 2, 3of PV; any dose of hep B Ages 6 weeks through 6yearS (6 years, 364 days) Do not use: For t~e 4th or 5 th dose of DTaP or the 4th dose of PV For children 7 years of age or older.ntramuscular (1M) injection in the deltoid of the arm or anterolateral thigh 1 inch needle; gauge Professional judgment is appropriate when selecting needle length and site Can be given with other vaccines, at the same visit (use separate sites; space at least 1 inch apart) Make sure minimum g)ge and minimum intervals are met: Minimum age for dose 1 is 6weeks Minimum age for dose 3 is 6 months Minimum intervals: - 4 weeks between dose 1 & 2-8weeks between dose 2& 3 Store in the refrigerator between 35 4(;0 F(2 o a C) Do NOT freeze Keep in the original box Shake well before using Pediarb CONTRANDCATONS An anaphylactic reaction to a prior dose of Pediarix, DTaP, PV or Hep Bvaccines An anaphylactic reaction to a component of DTaP/lPVlHep B(Pediarix ) including yeast, neomycin or polymyxin B. Encephalopathy not due to another cause occurring within 7 days after vaccination with a pertussis-containing vaccine Progressive neurologic disorder (such as infantile spasms, uncontrolled epilepsy or progressive encephalopathy). PRECAUTONS Moderate to severe acute illness Guillain-Barre syndrome within 6 weeks of a prior vaccine containing tetanus toxoid Temp~rature greater than or equal to 105 F within 48 hours of vaccination with no other identifiable cause Collapse or shock-like state within 48 hours of vaccination Persistent inconsolable crying lasting more than 3 hours within 48 hours of vaccination Convulsions with or without a fever within 3days of vaccination FURTHER PONTS Continue to give a birth dose of single-antigen hep Bvaccine soon after birth and before hospital discharge. You may follow with 3 doses of Pediarix, spaced appropriately, to children without contraindications. This 4-dose heps series is considered acceptable as long as the last dose of heps vaccine is given auafter age 6 months. Pediarix does not contain a preservative. After removal of the dose, any remaining vaccine should be discarded. The combined DTaP-PV-Hep Bvaccine may be used when any component of the vaccine is indicated, and if the other components are not contraindicated. There is not a separate Vaccine nformation Statement (VS) for Pediarix. Use the current VSs for DTaP, PV, and hep Bor the Multi-vaccine VS that include information about the Michigan Care mproveinentregistry (MCR). VSs with MCR information are available at or at your local health department. Document as "DTaP/lPVlHep B" in MCR, on the vaccine administration record & immunization record card. Publicly purchased DTaP/lPVHep B(Pediarix ) can be administered to eligible children 6weeks through 6years of age through the Vaccines for Children (VFC) Program in private providers' offices. Eligible children include those who are uninsured, underinsured, Medicaid eligible, Native American or Alaskan Natives. Contact your local health department for more information. For additional information, refer to VFC Program Resolutions on 'Vaccines to Prevent Diphtheria, Tetanus and Pertussis,' 'Vaccines to Prevent Poliomyelitis', 'Vaccines to Prevent Hepatitis S' and the ACP Recommendations on the use of DTaP, PV and HepS vaccines, located at January 26, 2009
5 ndications for Use and Schedule Approved for: Routine schedule of 2, 4, 6, and 15~18 months of age Ages 6 weeks through 4 years (4 years, 364 days} For doses 1,2,3 or 4 of DTaP, PV or Hib Do not use: For any person 5 years of age and older For the 5111 dose of DTaP or, if needed, a 5111 dose of PV ntramuscular (1M) injection in the deltoid of the arm or anterolateral thigh 1inch needle; gauge Professional judgment is appropriate when selecting needle length and site Can be given with other vaccines, at the same visit.see "Further Points" for reconstitution instructions Make sure minimum age and minimum intervals are met: Minimum age for dose 1 is 6 weeks Minimum age for dose 4 is 12 months Minimum intervals: 4 weeks between dose 1& 2 4 weeks between dose 2 & 3. 6 months between dose 3 &4 Store the DTaP-PV vial and the Hib vial in the original box' and in the refrigerator at 3SO 46 F (2' SOC) Shake well before using Administer within 30 min of reconstituting Pentacel CONTRANDCATONS An anaphylactic reaction to a prior dose of Pentacel', DTaP, PVor Hib vaccine An anaphylactic reaction to a component of DTaPPV/Hib (Pentacel) including neomycin or polymyxin B Encephalopathy not due to another cause occurring within 7 days after vaccination with a pertussis-containing vaccine Progressive neurologic disorder (such as infantile spasms. uncontrolled epilepsy or progressive encephalop.athy) PRECAUTONS. Moderate to severe acute illness Guillain-Barre syndrome within 6 weeks of a prior tetanus toxoid-containing vaccine.. Temperature greater than or equal to 105 F within 48 hours of vaccination with no other identifiable cause Collapse or shock-like state within 48 hours of vaccination Persistent inconsolable crying lasting more than 3 hours within 48 hours of vaccination Convulsions with or without a fever within 3days of vaccination FURTHER PONTS.Be sure to reconstitute the Hlb vial with DlaP PV vial before administration. Use only this Clap PV as the diluent. CDC recommends that the lot numbers from the DTaP-PVand the Hib vials be recorded on the vaccine administration record. Currently, MCR will only allow for one lot number to be entered; use the lot number from the Hib vial (matches outer box #)..Pentacel does not contain a preservative. After removal of the dose. any remaining vaccine should be discarded. The combined DTaP-PV-Hib vaccine may be used when any component of the vaccine is indicated. and if the other components are not contraindicated There is not aseparate Vaccine nformation Statement (VS) for Pentacel. Use the current VSs for DTaP, PV, and Hib or the MUlti-vaccine VS that include information about the Michigan Care mprovement Registry (MCR). VSs with MCR information are available at or your local health department Document as "DTaPPVlHib' in MCR, on the vaccine administration record &immunization record card Publicly purchased DTaP/lPVlHib (Pentacel) can be administered to eligible children 6weeks through 4years of age,utilizing the Vaccines for Children (VFC) Program, in private providers' offices. Eligible children include those who are uninsured, underinsured, Medicaid eligible,. Native American or Alaskan Natives. Contact your local health department for more information For additional information, refer to VFC Program Resolutions on 'Vaccines to Prevent Diphtheria, Tetanus and Pertussis', 'Vaccines to Prevent Poliomyelitis', and 'Vaccines to Prevent Haemophilus influenzae type B' and the ACP Recommendations on the use of DTaP, PVand Hib vaccines, located at hhp:llwml.cdc.goyjyaccines/recs January 15, 2010
6 ,!.~:, b~a Quick Look at Using Hep A/Hep B (Twinrix@) ndications for Use and Schedule Approved for: Rouone schedule of 3doses; 0, 1,6 months.persons with indications for both hepatitis A and hepatitis Bvaccines 'Alternate schedule of 4 doses; 0, 7, days and a booster dose 12 months after the first dose.ntramuscular (1M) injection in the deltoid of the arm '1-1.5 inch needle; gauge Professionaljudg'ment is appropriate when selecting needle length Can be given with other vaccines, at the same visit (use separate sites; space at least 1 inch apart) Each dose of Twinrix contains: One adult dose of hepatitis Bvaccine One pediatric dose of hepatitis A vaccine Make sure minimum age and minimum intervals are met: Minimum age for any dose is 18 years Minimum intervals for 3-dose schedule: - 4 weeks between dose 1& 2-5 months between dose 2&3 Store in the refrigerator between 3S D.4&' F(2 S0C) Do NOT freeze.keep in the original box Shake well before using, Twinrix CONTRANDCA1'ONS An anaphylactic reaction to a prior dose of Twinrix, hepatitis A or hepatitis 8 vaccine An anaphylactic reaction to a component of Twinrix (hep Nhep 8) including yeast and neomycin PRECAUTONS Moderate to severe acute illness FURTHER PONTS Because the hepatitis Bcomponent of Twinrix is equivalent to astandard adult dose of hep 8 vaccine, the schedule is the same Whether Twinrix or singe-antigen hep Svaccine is used Because the hepatitis A component of Twinrix is equivalent to a pediatric dose of hep Avaccine, persons 19 years and older who receive only 1or 2doses of Twinrix will need additional adult doses of single-antigen hep A vaccine Completing hepatitis A and hepatitis B series with single antigen hep A. hep B andlor Twinrix An'j combination of 3 doses of adult hepatitis Sor 3doses of Twinrix = a com~ete series of hep_autis B 1dose of Twinrix + 2 doses of adult hepatitis A =a complete series of hepatitis A 2doses of Twinrix + 1 dose of adult hepatitis A = a complete series of hepatitis A There is not aseparate Vaccine nformation Statement (VS) for Twinrix. Use the current VSs for hep A and hep Bthat include information about the Michigan Care mprovement Registry (MCR). V1Ss with MCR information are available at or at your local health department. Document as "Hep NHep S' in MCR, on the vaccine administration record &immunization record card Publicly purchased hep AJhep B(Twinrix ) and single-antigen hep Aand hep Bvaccines are available for persons at high risk for hepatitis A or hepatitis Bvirus infection when served at local health department or select sites. Eligible adults 19 years and older include those who are uninsured or underinsured. Adults who are Medicaid-eligible and meet high risk criteria for hep Aor hep Bmay receive privately purchased single-antigen vaccines or Twinrix; bill Medicaid for the vaccine &administration fee. Medicare part B&Dwill cover privately purchased hep A or hep Bunder certain circumstances-see policies. For persons 18 years and younger,- publicly purchased vaccines (excluding Twinrix ) are available in private provider offices under the Vaccines for Children (VFC) program. Eligible children are those with Medi~" underinsured, uninsured, or Native American or Alaskan Natives. Contact your local health department for more information on these programs. For additional information, refer to the ACP Recommendations on the use of Hap Aand Hep Bvaccines, located at ht!d:/www,cdc.govtvaccinesirecs, January 6, 2009
Vaccine Label Examples
 Vaccine Label Examples With the large amount of vaccine carried in most clinics, staff can easily become confused about vaccines within the storage unit. Labeling the area where vaccines are stored can
Vaccine Label Examples With the large amount of vaccine carried in most clinics, staff can easily become confused about vaccines within the storage unit. Labeling the area where vaccines are stored can
Vaccine Label Examples
 Vaccine Label Examples With the large amount of vaccine carried in most clinics, staff can easily become confused about vaccines within the storage unit. Labeling the area where vaccines are stored can
Vaccine Label Examples With the large amount of vaccine carried in most clinics, staff can easily become confused about vaccines within the storage unit. Labeling the area where vaccines are stored can
Immunization Guidelines for the Use of State Supplied Vaccine April 18, 2013
 DTaP / DT DTaP/IPV/Hep B Combination (Pediarix ) Children from 6 weeks of age up to the 7 th birthday Children from 2 months of age up to the 7th birthday: Indicated for the primary doses of DTaP, IPV,
DTaP / DT DTaP/IPV/Hep B Combination (Pediarix ) Children from 6 weeks of age up to the 7 th birthday Children from 2 months of age up to the 7th birthday: Indicated for the primary doses of DTaP, IPV,
From: New York State Department of Health, Bureau of Communicable Disease Control, Immunization Program
 November 3, 2008 To: Hospitals, Providers, Local Health Departments From: New York State Department of Health, Bureau of Communicable Disease Control, Immunization Program HEALTH ADVISORY: USE OF PENTACEL
November 3, 2008 To: Hospitals, Providers, Local Health Departments From: New York State Department of Health, Bureau of Communicable Disease Control, Immunization Program HEALTH ADVISORY: USE OF PENTACEL
New Jersey Department of Health Vaccine Preventable Disease Program Childhood and Adolescent Recommended Vaccines
 New Jersey Department of Health Vaccine Preventable Disease Program Childhood and Adolescent Recommended Vaccines Antigens Vaccine Approved Age Daptacel Diphtheria, Tetanus, and acellular Pertussis (DTaP)
New Jersey Department of Health Vaccine Preventable Disease Program Childhood and Adolescent Recommended Vaccines Antigens Vaccine Approved Age Daptacel Diphtheria, Tetanus, and acellular Pertussis (DTaP)
Immunization Guidelines For the Use of State Supplied Vaccine July 1, 2011
 DTaP / DT DTaP/IPV/Hep B Combination (Pediarix ) Children from 6 weeks of age up to the 7 th birthday Children from 2 months of age up to the 7th birthday: Indicated for the primary doses of DTaP, IPV,
DTaP / DT DTaP/IPV/Hep B Combination (Pediarix ) Children from 6 weeks of age up to the 7 th birthday Children from 2 months of age up to the 7th birthday: Indicated for the primary doses of DTaP, IPV,
Deployment of Combination Vaccines and STI vaccines
 Deployment of Combination Vaccines and STI vaccines Advancing Prevention Technologies for Sexual and Reproductive Health Symposium Berkeley, Ca March 24, 2009 Eileen Yamada, MD, MPH California Department
Deployment of Combination Vaccines and STI vaccines Advancing Prevention Technologies for Sexual and Reproductive Health Symposium Berkeley, Ca March 24, 2009 Eileen Yamada, MD, MPH California Department
Immunization Guidelines for the Use of State Supplied Vaccine May 17, 2015
 DTaP / DT DTaP/IPV/Hep B Combination (Pediarix ) Children from 6 weeks of age up to the 7 th birthday Children from 2 months of age up to the 7th birthday: Indicated for the primary doses of DTaP, IPV,
DTaP / DT DTaP/IPV/Hep B Combination (Pediarix ) Children from 6 weeks of age up to the 7 th birthday Children from 2 months of age up to the 7th birthday: Indicated for the primary doses of DTaP, IPV,
Vaccination Decision Making: What Providers Need to Know
 Objectives Vaccination Decision Making: What Providers Need to Know Kelli Smith, RN, BSN Iowa Department of Public Health Immunization Program Catch-up Schedule: how to most efficiently bring persons up-to-date
Objectives Vaccination Decision Making: What Providers Need to Know Kelli Smith, RN, BSN Iowa Department of Public Health Immunization Program Catch-up Schedule: how to most efficiently bring persons up-to-date
The Use of Combination Vaccines in the United States
 The Use of Combination Vaccines in the United States John W Ward, MD Senior Scientist, CDC Director, Program for Viral Hepatitis Elimination Task Force for Global Health Revised 2018 Combination Vaccine
The Use of Combination Vaccines in the United States John W Ward, MD Senior Scientist, CDC Director, Program for Viral Hepatitis Elimination Task Force for Global Health Revised 2018 Combination Vaccine
Appendix An Assessment Tool to Determine the Validity of Vaccine Doses
 Appendix 4.4 - An Assessment Tool to Determine the Validity of Vaccine Doses Note: Refer to the Canadian Immunization Guide and New Brunswick (NB) immunization program directives for recommendations for
Appendix 4.4 - An Assessment Tool to Determine the Validity of Vaccine Doses Note: Refer to the Canadian Immunization Guide and New Brunswick (NB) immunization program directives for recommendations for
C o l o r a d o I m m u n i z a t i o n M a n u a l S E C T I O N 8 Vaccines
 S E C T I O N 8 Vaccines 8. Vaccines I S S U E D : 9 / 1 / 9 8 R E V I S E D : 5 / 1 / 1 1 S E C T I O N - P A G E : 8-1 S U B J E C T : C O N T E N T S CONTENTS S E C T I O N 8 Vaccines DTaP...............................................
S E C T I O N 8 Vaccines 8. Vaccines I S S U E D : 9 / 1 / 9 8 R E V I S E D : 5 / 1 / 1 1 S E C T I O N - P A G E : 8-1 S U B J E C T : C O N T E N T S CONTENTS S E C T I O N 8 Vaccines DTaP...............................................
Use of Infanrix -IPV+Hib in the infant primary immunisation schedule
 Use of Infanrix -IPV+Hib in the infant primary immunisation schedule An update for registered healthcare practitioners July 2014 Quality Education for a Healthier Scotland 1 Acknowledgments Many thanks
Use of Infanrix -IPV+Hib in the infant primary immunisation schedule An update for registered healthcare practitioners July 2014 Quality Education for a Healthier Scotland 1 Acknowledgments Many thanks
Agenda. Richard Moriarty, MD, FAAP, Co-Director, MCAAP Immunization Initiative. Pentacel Vaccine Shortage Update MMRV Vaccine (Proquad ) Update
 Massachusetts Department of Public Health Division of Epidemiology and Immunization Agenda Introduction Richard Moriarty, MD, FAAP, Co-Director, MCAAP Immunization Initiative Pentacel Vaccine Shortage
Massachusetts Department of Public Health Division of Epidemiology and Immunization Agenda Introduction Richard Moriarty, MD, FAAP, Co-Director, MCAAP Immunization Initiative Pentacel Vaccine Shortage
~~-o.., :...; C. (=~~-o--"'~...q J
 STATE OF ALABAMA DEPARTMENT OF PUBLIC HEALTH Donald E. Williamson, MD State Health Officer September 30, 2005 Dear Vaccines for Children (VFC) Provider: SUBJECT: Annual VFC Benchmark Data Is Due New VFC
STATE OF ALABAMA DEPARTMENT OF PUBLIC HEALTH Donald E. Williamson, MD State Health Officer September 30, 2005 Dear Vaccines for Children (VFC) Provider: SUBJECT: Annual VFC Benchmark Data Is Due New VFC
PREVENTIVE IMMUNIZATIONS. PREVENTIVE IMMUNIZATIONS These codes do not have a diagnosis code requirement for preventive benefits to apply.
 An immunization that does not fall under one of the exclusions in the Certificate of Coverage is considered covered after the following conditions are satisfied: (1) FDA approval; (2) explicit ACIP recommendation
An immunization that does not fall under one of the exclusions in the Certificate of Coverage is considered covered after the following conditions are satisfied: (1) FDA approval; (2) explicit ACIP recommendation
Diphtheria, Tetanus, and Pertussis. DTaP/DT and Tdap/Td Vaccines
 Centers for Disease Control and Prevention National Center for Immunization and Respiratory Diseases Diphtheria, Tetanus, and Pertussis DTaP/DT and Tdap/Td Vaccines Jean C. Smith, MD, MPH Medical Officer
Centers for Disease Control and Prevention National Center for Immunization and Respiratory Diseases Diphtheria, Tetanus, and Pertussis DTaP/DT and Tdap/Td Vaccines Jean C. Smith, MD, MPH Medical Officer
PREVENTIVE IMMUNIZATIONS. PREVENTIVE IMMUNIZATIONS These codes do not have a diagnosis code requirement for preventive benefits to apply.
 An immunization that does not fall under one of the exclusions in the Certificate of Coverage is considered covered after the following conditions are satisfied: (1) FDA approval; (2) explicit ACIP recommendation
An immunization that does not fall under one of the exclusions in the Certificate of Coverage is considered covered after the following conditions are satisfied: (1) FDA approval; (2) explicit ACIP recommendation
GENERAL IMMUNIZATION GUIDE FOR CHILDCARE PROVIDERS August 2018 **CHILD VACCINES** DIPHTHERIA, TETANUS, PERTUSSIS VACCINES
 GENERAL IMMUNIZATION GUIDE FOR CHILDCARE PROVIDERS August 2018 **CHILD VACCINES** DIPHTHERIA, TETANUS, PERTUSSIS VACCINES DTaP: Diphtheria, Tetanus, acellular Pertussis Vaccine Infanrix Licensed in 1997
GENERAL IMMUNIZATION GUIDE FOR CHILDCARE PROVIDERS August 2018 **CHILD VACCINES** DIPHTHERIA, TETANUS, PERTUSSIS VACCINES DTaP: Diphtheria, Tetanus, acellular Pertussis Vaccine Infanrix Licensed in 1997
Legal Aspects of Children s Health Services A Guide to Public Health Services for Children. Part 4: Immunizations
 Legal Aspects of Children s Health Services A Guide to Public Health Services for Children Part 4: Immunizations Developed in 2014 by in partnership with www.nwcphp.org Updated June 24, 2014 Authors: Jane
Legal Aspects of Children s Health Services A Guide to Public Health Services for Children Part 4: Immunizations Developed in 2014 by in partnership with www.nwcphp.org Updated June 24, 2014 Authors: Jane
What DO the childhood immunization footnotes reveal? Questions and answers
 What DO the childhood immunization footnotes reveal? Questions and answers Stanley E. Grogg, DO, FACOP, FAAP he Advisory Committee on Immunization Practices (ACIP) recommends the childhood vaccination
What DO the childhood immunization footnotes reveal? Questions and answers Stanley E. Grogg, DO, FACOP, FAAP he Advisory Committee on Immunization Practices (ACIP) recommends the childhood vaccination
Immunization Protocol with Procedures and Standing Orders for Nurses
 New Mexico Department of Health Immunization Protocol with Procedures and Standing Orders for Nurses Updated September 1, 2016 Minor Revision October 7, 2016 HPV Revision December 27, 2016 Immunization
New Mexico Department of Health Immunization Protocol with Procedures and Standing Orders for Nurses Updated September 1, 2016 Minor Revision October 7, 2016 HPV Revision December 27, 2016 Immunization
DATE: & 5, 2015 SUBJECT:
 TO: Immunization Program Directors of Licensed Child Day Care Centers & Group Day Care Homes FROM: Mick Bolduc-Epidemiologist, Immunization Program DATE: January 5, 2015 SUBJECT: Annual Immunization Survey
TO: Immunization Program Directors of Licensed Child Day Care Centers & Group Day Care Homes FROM: Mick Bolduc-Epidemiologist, Immunization Program DATE: January 5, 2015 SUBJECT: Annual Immunization Survey
Adult Immunizations: Ensuring Family Planning Clients are Protected
 Objectives Adult Immunization: Identify the recommended vaccines for adults Identify three functions of the Michigan Care Improvement Registry (MCIR) Discuss vaccine procurement options for adults Barbara
Objectives Adult Immunization: Identify the recommended vaccines for adults Identify three functions of the Michigan Care Improvement Registry (MCIR) Discuss vaccine procurement options for adults Barbara
Diphtheria-Tetanus-Acellular Pertussis-Polio- Haemophilus influenzae type b Conjugate Combined Vaccine Biological Page (DTaP-IPV-Hib)
 Diphtheria-Tetanus-Acellular Pertussis-Polio- Haemophilus influenzae type b Conjugate Combined Vaccine Biological Page (DTaP-IPV-Hib) Section 7: Biological Product Information Standard #: 07.211 Created
Diphtheria-Tetanus-Acellular Pertussis-Polio- Haemophilus influenzae type b Conjugate Combined Vaccine Biological Page (DTaP-IPV-Hib) Section 7: Biological Product Information Standard #: 07.211 Created
Objectives. Immunity. Childhood Immunization Risk of Non-Vaccinated Children 12/22/2015
 Childhood Immunization Risk of Non-Vaccinated Children Bertha P. Rojas, Pharm.D. PGY-1 Pharmacy Resident South Miami Hospital Objectives Understand the definition of herd immunity Identify vaccine-preventable
Childhood Immunization Risk of Non-Vaccinated Children Bertha P. Rojas, Pharm.D. PGY-1 Pharmacy Resident South Miami Hospital Objectives Understand the definition of herd immunity Identify vaccine-preventable
Advisory Committee on Immunization Practices VACCINE ACRONYMS
 Vaccine Acronyms Page 1 of 5 Advisy Committee on Immunization Practices VACCINE ACRONYMS Vaccines Included in the Immunization Schedules f Children, Adolescents, and Adults Following is a table of standardized
Vaccine Acronyms Page 1 of 5 Advisy Committee on Immunization Practices VACCINE ACRONYMS Vaccines Included in the Immunization Schedules f Children, Adolescents, and Adults Following is a table of standardized
ADMINISTERING VACCINATIONS AT OFFSITE LOCATIONS
 ADMINISTERING VACCINATIONS AT OFFSITE LOCATIONS Purpose: To reduce morbidity and mortality for all vaccine-preventable diseases by administering routinely recommended vaccines to children and adults who
ADMINISTERING VACCINATIONS AT OFFSITE LOCATIONS Purpose: To reduce morbidity and mortality for all vaccine-preventable diseases by administering routinely recommended vaccines to children and adults who
WVSIIS Vaccine Type Cheat Sheet Updated June 2010
 DTaP / DTP / DT / Td / Tdap Combo Vaccines Comvax HepB / Hib Merck (MSD) 1996 - Present 90748 Kinrix DTaP / IPV GlaxoSmithKline (SKB) 2008 - Present 90696 Pediarix DTaP / HepB / IPV GlaxoSmithKline (SKB)
DTaP / DTP / DT / Td / Tdap Combo Vaccines Comvax HepB / Hib Merck (MSD) 1996 - Present 90748 Kinrix DTaP / IPV GlaxoSmithKline (SKB) 2008 - Present 90696 Pediarix DTaP / HepB / IPV GlaxoSmithKline (SKB)
TRICARE Retail Vaccination Program Vaccine List - September 2018*
 Diphtheria, tetanus and pertussis Diphtheria and tetanus toxoids adsorbed acellular pertussis adsorbed DT DTaP Diphtheria and tetanus toxoids adsorbed Daptacel, Infanrix Tetanus and diphtheria toxoids
Diphtheria, tetanus and pertussis Diphtheria and tetanus toxoids adsorbed acellular pertussis adsorbed DT DTaP Diphtheria and tetanus toxoids adsorbed Daptacel, Infanrix Tetanus and diphtheria toxoids
Varicella (Chickenpox) and Varicella Vaccines
 Centers for Disease Control and Prevention National Center for Immunization and Respiratory Diseases Varicella (Chickenpox) and Varicella Vaccines September 2018 Photographs and images included in this
Centers for Disease Control and Prevention National Center for Immunization and Respiratory Diseases Varicella (Chickenpox) and Varicella Vaccines September 2018 Photographs and images included in this
7.0 Nunavut Childhood and Adult Immunization Schedules and Catch-up Aids
 7.0 Nunavut Childhood and Adult Immunization Schedules and Catch-up Aids Contents Introduction Nunavut Recommended Childhood Immunization Schedule Nunavut Routine Adult Immunization Schedule Nunavut Immunization
7.0 Nunavut Childhood and Adult Immunization Schedules and Catch-up Aids Contents Introduction Nunavut Recommended Childhood Immunization Schedule Nunavut Routine Adult Immunization Schedule Nunavut Immunization
Return those vaccines to the storage unit and repeat for all VFC vaccines before completing the rest of this worksheet.
 Instructions: Complete the worksheet that matches your provider category (e.g., low-, medium-, or high-volume) before submitting routine vaccine orders. Refer to the VFC Program Provider Operations Manual
Instructions: Complete the worksheet that matches your provider category (e.g., low-, medium-, or high-volume) before submitting routine vaccine orders. Refer to the VFC Program Provider Operations Manual
Coverage of Vaccines Medicaid and Child Health Plus Members
 Coverage of Vaccines Medicaid and Child Health Plus Members For children between the ages 0-18, routine recommended vaccinations are covered through Vaccines for Children program [VFC]. Fidelis Care will
Coverage of Vaccines Medicaid and Child Health Plus Members For children between the ages 0-18, routine recommended vaccinations are covered through Vaccines for Children program [VFC]. Fidelis Care will
This document was published by: The National Department of Health. Editorial team: Provincial EPI Team National EPI Team Dr NJ Ngcobo
 REVISED : OCTOBER 2010 This document was published by: The National Department of Health Editorial team: Provincial EPI Team National EPI Team Dr NJ Ngcobo Copies may be obtained from: The National Department
REVISED : OCTOBER 2010 This document was published by: The National Department of Health Editorial team: Provincial EPI Team National EPI Team Dr NJ Ngcobo Copies may be obtained from: The National Department
STANWOOD-CAMANO SCHOOL DISTRICT REQUEST FOR PART-TIME ATTENDANCE OR ANCILLARY SERVICES FROM PRIVATE SCHOOL PUPIL
 STANWOOD-CAMANO SCHOOL DISTRICT REQUEST FOR PART-TIME ATTENDANCE OR ANCILLARY SERVICES FROM PRIVATE SCHOOL PUPIL 114 F2 Students Please complete ALL information: Name of Pupil Grade Address of Pupil City
STANWOOD-CAMANO SCHOOL DISTRICT REQUEST FOR PART-TIME ATTENDANCE OR ANCILLARY SERVICES FROM PRIVATE SCHOOL PUPIL 114 F2 Students Please complete ALL information: Name of Pupil Grade Address of Pupil City
9/11/2018. Tdap/Td Vaccines. Why Adolescents and Adults Need Pertussis Vaccine. Pertussis Complications Among Adolescents and Adults
 Centers for Disease Control and Prevention National Center for Immunization and Respiratory Diseases Tdap/Td Vaccines September 2018 Chapters 7, 21 and 16 Photographs and images included in this presentation
Centers for Disease Control and Prevention National Center for Immunization and Respiratory Diseases Tdap/Td Vaccines September 2018 Chapters 7, 21 and 16 Photographs and images included in this presentation
VACCINE MANAGEMENT. Recommendations for Handling and Storage of Selected Biologicals. January 2001 DEPARTMENT OF HEALTH AND HUMAN SERVICES
 VACCINE MANAGEMENT Recommendations for Handling and Storage of Selected Biologicals January 2001 DEPARTMENT OF HEALTH AND HUMAN SERVICES DTaP: Diphtheria Toxoid, Tetanus Toxoid, Acellular Pertussis Vaccine
VACCINE MANAGEMENT Recommendations for Handling and Storage of Selected Biologicals January 2001 DEPARTMENT OF HEALTH AND HUMAN SERVICES DTaP: Diphtheria Toxoid, Tetanus Toxoid, Acellular Pertussis Vaccine
Immunizations are among the most cost effective and widely used public health interventions.
 Focused Issue of This Month Recommended by the Korean Pediatric Society, 2008 Hoan Jong Lee, MD Department of Pediatrics, Seoul National University College of Medicine E mail : hoanlee@snu.ac.kr J Korean
Focused Issue of This Month Recommended by the Korean Pediatric Society, 2008 Hoan Jong Lee, MD Department of Pediatrics, Seoul National University College of Medicine E mail : hoanlee@snu.ac.kr J Korean
SHOTS ADDING VFC VACCINES
 Florida SHOTS ADDING VFC VACCINES Contact Information www.flshots.com Free help desk: 877-888-SHOT (7468) Monday Friday, 8 A.M. to 5 P.M. Eastern Quick Content Finder ADDING A VFC VACCINATION RECORD 1
Florida SHOTS ADDING VFC VACCINES Contact Information www.flshots.com Free help desk: 877-888-SHOT (7468) Monday Friday, 8 A.M. to 5 P.M. Eastern Quick Content Finder ADDING A VFC VACCINATION RECORD 1
RECOMMENDED IMMUNIZATIONS
 Recommended Immunization Schedule for Persons Aged 0 Through 6 Years United States 2010 1 2 4 6 12 15 18 19 23 2 3 4 6 Vaccine Age Birth month months months months months months months months years years
Recommended Immunization Schedule for Persons Aged 0 Through 6 Years United States 2010 1 2 4 6 12 15 18 19 23 2 3 4 6 Vaccine Age Birth month months months months months months months months years years
Childhood Immunization Status
 emeasure Title emeasure Identifier (Measure Authoring Tool) Childhood Immunization Status 117 emeasure Version number 5.1.000 NQF Number 0038 GUID b2802b7a-3580-4be8-9458- 921aea62b78c Measurement Period
emeasure Title emeasure Identifier (Measure Authoring Tool) Childhood Immunization Status 117 emeasure Version number 5.1.000 NQF Number 0038 GUID b2802b7a-3580-4be8-9458- 921aea62b78c Measurement Period
SUMMARY OF PRODUCT CHARACTERISTICS
 1. NAME OF THE MEDICINAL PRODUCT Diphtheria, Tetanus and Pertussis Vaccine (Adsorbed) I.P. Injectable, Suspension for injection. 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each dose of 0.5 ml contains:
1. NAME OF THE MEDICINAL PRODUCT Diphtheria, Tetanus and Pertussis Vaccine (Adsorbed) I.P. Injectable, Suspension for injection. 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each dose of 0.5 ml contains:
Table of Contents. 1.0 INFANRIX hexa VACCINE...3
 Table of Contents 1.0 INFANRIX hexa VACCINE...3 1.1 What is INFANRIX hexa vaccine?...3 1.2 When will INFANRIX hexa be introduced?...3 1.3 What is the difference between INFANRIX hexa and Pediacel?...3
Table of Contents 1.0 INFANRIX hexa VACCINE...3 1.1 What is INFANRIX hexa vaccine?...3 1.2 When will INFANRIX hexa be introduced?...3 1.3 What is the difference between INFANRIX hexa and Pediacel?...3
Vaccinations Outside Recommended Ages 2014; Six Immunization Information System Sentinel Sites
 National Center for Immunization & Respiratory Diseases Vaccinations Outside Recommended Ages 2014; Six Immunization Information System Sentinel Sites Loren Rodgers, Ph.D. CDC Immunization Information
National Center for Immunization & Respiratory Diseases Vaccinations Outside Recommended Ages 2014; Six Immunization Information System Sentinel Sites Loren Rodgers, Ph.D. CDC Immunization Information
Pentabio Vaccine (DTP-HB-Hib)
 SUMMARY OF PRODUCT CHARACTERISTICS Product Name Pharmaceutical Form Strength Presentation : Pentabio : Suspension for injection : 1, 5 and 10 doses : Box of 10 vials @ 0.5 ml Box of 10 vials @ 2.5 ml Box
SUMMARY OF PRODUCT CHARACTERISTICS Product Name Pharmaceutical Form Strength Presentation : Pentabio : Suspension for injection : 1, 5 and 10 doses : Box of 10 vials @ 0.5 ml Box of 10 vials @ 2.5 ml Box
Advisory Committee on Immunization Practices VACCINE ACRONYMS
 May 1, 2015 Vaccine Acronyms Page 1 of 5 Advisy Committee on Immunization Practices VACCINE ACRONYMS Vaccines Included in the Immunization Schedules f Children, Adolescents, and Adults Following is a table
May 1, 2015 Vaccine Acronyms Page 1 of 5 Advisy Committee on Immunization Practices VACCINE ACRONYMS Vaccines Included in the Immunization Schedules f Children, Adolescents, and Adults Following is a table
Recommended Childhood Immunization Schedu...ates, January - December 2000, NP Central
 Recommended Childhood Immunization Schedule United States, January - December 2000 Vaccines 1 are listed under routinely recommended ages. Solid-colored bars indicate range of recommended ages for immunization.
Recommended Childhood Immunization Schedule United States, January - December 2000 Vaccines 1 are listed under routinely recommended ages. Solid-colored bars indicate range of recommended ages for immunization.
CHILDHOOD IMMUNIZATIONS
 CHILDHOOD IMMUNIZATIONS APPLICATIONS OBJECTIVE Purpose of Measure: ELIGIBLE POPULATION Which members are included? STANDARD OF CARE What immunizations should be administered? NCQA ACCEPTED CODES DOCUMENTATION
CHILDHOOD IMMUNIZATIONS APPLICATIONS OBJECTIVE Purpose of Measure: ELIGIBLE POPULATION Which members are included? STANDARD OF CARE What immunizations should be administered? NCQA ACCEPTED CODES DOCUMENTATION
Immunization Program. Directors of Licensed Child Day Care Centers & Group Day Care Homes. FROM: Mick Bolduc-Epidemiologist Debra L.
 Immunization Program TO: Directors of Licensed Child Day Care Centers & Group Day Care Homes FROM: Mick Bolduc-Epidemiologist Debra L. Johnson-Chief Immunization Program Community Based Regulation Section
Immunization Program TO: Directors of Licensed Child Day Care Centers & Group Day Care Homes FROM: Mick Bolduc-Epidemiologist Debra L. Johnson-Chief Immunization Program Community Based Regulation Section
HIBERIX PRODUCT INFORMATION
 HIBERIX PRODUCT INFORMATION NAME OF THE MEDICINE HIBERIX Haemophilus influenzae type b (Hib) vaccine DESCRIPTION Powder and diluent for solution for injection. After reconstitution, 1 dose (0.5 ml) contains:
HIBERIX PRODUCT INFORMATION NAME OF THE MEDICINE HIBERIX Haemophilus influenzae type b (Hib) vaccine DESCRIPTION Powder and diluent for solution for injection. After reconstitution, 1 dose (0.5 ml) contains:
Healthy People 2020 objectives were released in 2010, with a 10-year horizon to achieve the goals by 2020.
 Appendix 1: Healthy People 2020 Immunization-related Objectives Healthy People provides science-based, 10-year national objectives for improving the health of all Americans. For three decades, Healthy
Appendix 1: Healthy People 2020 Immunization-related Objectives Healthy People provides science-based, 10-year national objectives for improving the health of all Americans. For three decades, Healthy
Creating an Immunization Niche in the Community Pharmacy. Module 4: Communication and Counseling The Patient s Immunization Experience
 Creating an Immunization Niche in the Community Pharmacy Module 4: Communication and Counseling The Patient s Immunization Experience Jonathan G. Marquess, PharmD, CDE, CDM Learning Objectives Discuss
Creating an Immunization Niche in the Community Pharmacy Module 4: Communication and Counseling The Patient s Immunization Experience Jonathan G. Marquess, PharmD, CDE, CDM Learning Objectives Discuss
OVERVIEW 2018 VACCINE UPDATE 3/19/ VACCINE SCHEDULE
 2018 VACCINE UPDATE Mick Bolduc Epidemiologist/Vaccine Coordinator Connecticut Immunization Program March 22, 2018 1 OVERVIEW - Immunization Schedule - New ACIP Recommendations - Religious & Medical Exemptions
2018 VACCINE UPDATE Mick Bolduc Epidemiologist/Vaccine Coordinator Connecticut Immunization Program March 22, 2018 1 OVERVIEW - Immunization Schedule - New ACIP Recommendations - Religious & Medical Exemptions
Diphtheria-Tetanus-Acellular Pertussis-Polio- Combined Vaccine Biological Page (DTaP-IPV)
 Diphtheria-Tetanus-Acellular Pertussis-Polio- Combined Vaccine Biological Page (DTaP-IPV) Section 7: Biological Product Information Standard #: 07.212 Created by: Province-wide Immunization Program Standards
Diphtheria-Tetanus-Acellular Pertussis-Polio- Combined Vaccine Biological Page (DTaP-IPV) Section 7: Biological Product Information Standard #: 07.212 Created by: Province-wide Immunization Program Standards
1. QUALITATIVE AND QUANTITATIVE COMPOSITION
 INFANRIX 1. QUALITATIVE AND QUANTITATIVE COMPOSITION contains diphtheria toxoid, tetanus toxoid, and three purified pertussis antigens [pertussis toxoid (PT), filamentous haemagglutinin (FHA) and 69 kilodalton
INFANRIX 1. QUALITATIVE AND QUANTITATIVE COMPOSITION contains diphtheria toxoid, tetanus toxoid, and three purified pertussis antigens [pertussis toxoid (PT), filamentous haemagglutinin (FHA) and 69 kilodalton
Vaccines for Children
 Vaccines for Children 0-9 old Our goal is to offer your family the best care possible, which includes making sure your child is up to date on all vaccines. DTaP (Diptheria, Tetanus, Pertussis) Vaccine
Vaccines for Children 0-9 old Our goal is to offer your family the best care possible, which includes making sure your child is up to date on all vaccines. DTaP (Diptheria, Tetanus, Pertussis) Vaccine
3 rd dose. 3 rd or 4 th dose, see footnote 5. see footnote 13. for certain high-risk groups
 Figure 1. Recommended immunization schedule for persons aged 0 through 18 years 2013. (FOR THOSE WHO FALL BEHIND OR START LATE, SEE THE CATCH-UP SCHEDULE [FIGURE 2]). These recommendations must be read
Figure 1. Recommended immunization schedule for persons aged 0 through 18 years 2013. (FOR THOSE WHO FALL BEHIND OR START LATE, SEE THE CATCH-UP SCHEDULE [FIGURE 2]). These recommendations must be read
EARLY CHILDHOOD DEVELOPMENT & EDUCATION
 A division of Pawsitively Bassett, Inc. EARLY CHILDHOOD DEVELOPMENT & EDUCATION 08-09 67 N. Monroe St., Spokane WA 9908 www.nlcpreschool.com (509) 8-00 Dear Parents, Thank you, for your interest in the
A division of Pawsitively Bassett, Inc. EARLY CHILDHOOD DEVELOPMENT & EDUCATION 08-09 67 N. Monroe St., Spokane WA 9908 www.nlcpreschool.com (509) 8-00 Dear Parents, Thank you, for your interest in the
Schools. Kindergarten
 1 of 5 7/29/2014 4:14 PM Schools North Carolina law requires all children in the state to receive certain immunizations. Records are checked when children enter school or child care. Kindergarten Vaccination
1 of 5 7/29/2014 4:14 PM Schools North Carolina law requires all children in the state to receive certain immunizations. Records are checked when children enter school or child care. Kindergarten Vaccination
Package leaflet: information for the user
 Package leaflet: information for the user Pentavac, powder and suspension for injection Diphtheria, tetanus, pertussis (acellular, component), poliomyelitis (inactivated) and Haemophilus influenzae type
Package leaflet: information for the user Pentavac, powder and suspension for injection Diphtheria, tetanus, pertussis (acellular, component), poliomyelitis (inactivated) and Haemophilus influenzae type
All Kindergarteners and 4-6 year old transfer students. 4 doses DTP or DTaP 1 dose must be at or after 4 years of age. None
 Parents Vaccines Required for School Entry in Michigan Whenever children are brought into group settings, there is a chance for diseases to spread. Children must follow state vaccine laws in order to attend
Parents Vaccines Required for School Entry in Michigan Whenever children are brought into group settings, there is a chance for diseases to spread. Children must follow state vaccine laws in order to attend
Immunizations Across the Lifespan: Updates and Standards for Immunization
 Immunizations Across the Lifespan: Updates and Standards for Immunization April 12, 2011 Southwestern PA Immunization Coalition Conference Donna L. Weaver, RN, MN Nurse Educator Education, Information,
Immunizations Across the Lifespan: Updates and Standards for Immunization April 12, 2011 Southwestern PA Immunization Coalition Conference Donna L. Weaver, RN, MN Nurse Educator Education, Information,
Routine Immunization Products
 Newfoundland and Labrador Immunization Manual Section 3 Routine Immunization Products Immunization with Diphtheria, Pertussis, Tetanus, Polio and Hib Vaccine (DTap-IPV- Hib 3.1 Immunization with Diphtheria,
Newfoundland and Labrador Immunization Manual Section 3 Routine Immunization Products Immunization with Diphtheria, Pertussis, Tetanus, Polio and Hib Vaccine (DTap-IPV- Hib 3.1 Immunization with Diphtheria,
Molina Healthcare of CA Medi-Cal Wellness Services Bonus. MHC Quality Dept. Revised 12/15/17
 Molina Healthcare of CA Medi-Cal Wellness Services Bonus MHC Quality Dept. Revised 12/15/17 Changes to the Wellness Service Bonus As of January 1, 2018 DHCS is sun-setting the Information Only PM160 form
Molina Healthcare of CA Medi-Cal Wellness Services Bonus MHC Quality Dept. Revised 12/15/17 Changes to the Wellness Service Bonus As of January 1, 2018 DHCS is sun-setting the Information Only PM160 form
ADVISORY COMMITTEE ON IMMUNIZATION PRACTICES VACCINES FOR CHILDREN PROGRAM VACCINES TO PREVENT ROTAVIRUS GASTROENTERITIS
 Resolution No. 2/06-2 ADVISORY COMMITTEE ON IMMUNIZATION PRACTICES VACCINES FOR CHILDREN PROGRAM VACCINES TO PREVENT ROTAVIRUS GASTROENTERITIS The purpose of this resolution is to add rotavirus vaccine
Resolution No. 2/06-2 ADVISORY COMMITTEE ON IMMUNIZATION PRACTICES VACCINES FOR CHILDREN PROGRAM VACCINES TO PREVENT ROTAVIRUS GASTROENTERITIS The purpose of this resolution is to add rotavirus vaccine
Proof of residency in East Orange is mandatory (see Residency Requirements)
 Pre-K Registration Requirements Child must be at least 3 or 4 years old by October 1st of the current school year Immunization (shot records) are mandatory Age appropriate vaccinations for children entering
Pre-K Registration Requirements Child must be at least 3 or 4 years old by October 1st of the current school year Immunization (shot records) are mandatory Age appropriate vaccinations for children entering
Infanrix TM Diphtheria (D), tetanus (T), pertussis (acellular, component) (Pa) vaccine
 Infanrix TM Diphtheria (D), tetanus (T), pertussis (acellular, component) (Pa) vaccine QUALITATIVE AND QUANTITATIVE COMPOSITION Infanrix TM contains diphtheria toxoid, tetanus toxoid and three purified
Infanrix TM Diphtheria (D), tetanus (T), pertussis (acellular, component) (Pa) vaccine QUALITATIVE AND QUANTITATIVE COMPOSITION Infanrix TM contains diphtheria toxoid, tetanus toxoid and three purified
EXPANDED PROGRAM OF IMMUNIZATION (EPI) Definition Program adopted by WHO since l974, it includes child immunization & vaccination of pregnant women.
 EXPANDED PROGRAM OF IMMUNIZATION (EPI) Definition Program adopted by WHO since l974, it includes child immunization & vaccination of pregnant women. Strategy A. Child immunization Egyptian immunization
EXPANDED PROGRAM OF IMMUNIZATION (EPI) Definition Program adopted by WHO since l974, it includes child immunization & vaccination of pregnant women. Strategy A. Child immunization Egyptian immunization
301 W. Alder, Missoula, MT or
 301 W. Alder, Missoula, MT 59802 406-258-4745 or 406-258-3363 Routine Immunizations are available on a walk-in basis: Mondays, Tuesdays, Thursdays, and Fridays from 9:00am to 4:30pm Wednesdays, 10:00am-4:30pm
301 W. Alder, Missoula, MT 59802 406-258-4745 or 406-258-3363 Routine Immunizations are available on a walk-in basis: Mondays, Tuesdays, Thursdays, and Fridays from 9:00am to 4:30pm Wednesdays, 10:00am-4:30pm
Vaccine Finance. Overview of stakeholder input and NVAC working group draft white paper. Walt Orenstein, MD
 Vaccine Finance Overview of stakeholder input and NVAC working group draft white paper Walt Orenstein, MD Consultant to the National Vaccine Program Office July 24, 2008 Number of Vaccines in the Routine
Vaccine Finance Overview of stakeholder input and NVAC working group draft white paper Walt Orenstein, MD Consultant to the National Vaccine Program Office July 24, 2008 Number of Vaccines in the Routine
46825 (260) $UPONT
 Be wise. Immunize. Keeping track of the shots your children receive can be confusing. This is an important responsibility that is shared by you and your immunization providers. This booklet contains the
Be wise. Immunize. Keeping track of the shots your children receive can be confusing. This is an important responsibility that is shared by you and your immunization providers. This booklet contains the
Immunizations (Guideline Intervals Using The Rule of Six for Vaccines Birth to Six Years)
 Immunizations (Guideline Intervals Using The Rule of Six for Vaccines Birth to Six Years) Guideline developed by Shelly Baldwin, MD, in collaboration with the ANGELS Team. Last reviewed by Shelly Baldwin,
Immunizations (Guideline Intervals Using The Rule of Six for Vaccines Birth to Six Years) Guideline developed by Shelly Baldwin, MD, in collaboration with the ANGELS Team. Last reviewed by Shelly Baldwin,
NOTE: The above recommendations must be read along with the footnotes of this schedule.
 Figure 1. Recommended immunization schedule for persons aged 0 through 18 years United States, 2014. (FOR THOSE WHO FALL BEHIND OR START LATE, SEE THE CATCH-UP SCHEDULE [FIGURE 2]). These recommendations
Figure 1. Recommended immunization schedule for persons aged 0 through 18 years United States, 2014. (FOR THOSE WHO FALL BEHIND OR START LATE, SEE THE CATCH-UP SCHEDULE [FIGURE 2]). These recommendations
More Changes! VFC Program Recommendations and Requirements
 More Changes! 2014-2015 VFC Program Maribel Chavez-Torres, MPH Immunization Program Director City of Chicago Mayor Rahm Emanuel Chicago Department of Public Health Commissioner Bechara Choucair, M.D. VFC
More Changes! 2014-2015 VFC Program Maribel Chavez-Torres, MPH Immunization Program Director City of Chicago Mayor Rahm Emanuel Chicago Department of Public Health Commissioner Bechara Choucair, M.D. VFC
NEW ZEALAND DATA SHEET
 1. PRODUCT NAME NEW ZEALAND DATA SHEET HIBERIX Haemophilus influenzae type b (Hib) powder and diluent for solution for injection 2. QUALITATIVE AND QUANTITATIVE COMPOSITION After reconstitution, 1 dose
1. PRODUCT NAME NEW ZEALAND DATA SHEET HIBERIX Haemophilus influenzae type b (Hib) powder and diluent for solution for injection 2. QUALITATIVE AND QUANTITATIVE COMPOSITION After reconstitution, 1 dose
See 17 for PATIENT COUNSELING INFORMATION. Revised: XX/2010
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use HIBERIX safely and effectively. See full prescribing information for HIBERIX. HIBERIX [Haemophilus
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use HIBERIX safely and effectively. See full prescribing information for HIBERIX. HIBERIX [Haemophilus
Vaccine Innovation: Challenges and Opportunities to Protect Health. Julie Louise Gerberding, M.D., M.P.H President, Merck Vaccines
 Vaccine Innovation: Challenges and Opportunities to Protect Health Julie Louise Gerberding, M.D., M.P.H President, Merck Vaccines Protecting Health with Vaccines HEALTH IMPROVEMENT Population Impact Guidelines
Vaccine Innovation: Challenges and Opportunities to Protect Health Julie Louise Gerberding, M.D., M.P.H President, Merck Vaccines Protecting Health with Vaccines HEALTH IMPROVEMENT Population Impact Guidelines
I certify that the information provided on this form is correct and verifiable. Month Day Year
 Certificate of Immunization Status (CIS) DOH 48-0 January 05 Please print. See back for instructions on how to fill out this form or get it printed from the Immunization Information System. Child s Last
Certificate of Immunization Status (CIS) DOH 48-0 January 05 Please print. See back for instructions on how to fill out this form or get it printed from the Immunization Information System. Child s Last
Package leaflet: Information for the user
 Package leaflet: Information for the user Tetravac, suspension for injection Diphtheria, tetanus, pertussis (acellular, component) and poliomyelitis (inactivated) vaccine, adsorbed. Read all of this leaflet
Package leaflet: Information for the user Tetravac, suspension for injection Diphtheria, tetanus, pertussis (acellular, component) and poliomyelitis (inactivated) vaccine, adsorbed. Read all of this leaflet
Annotated Bibliography:
 Annotated Bibliography: Montana Code Annotated 2009. Montana Legislative Services. September 10, 2010 http://data.opi.mt.gov/bills/mca/20/5/20-5-403.htm. A school may not allow a student to attend unless
Annotated Bibliography: Montana Code Annotated 2009. Montana Legislative Services. September 10, 2010 http://data.opi.mt.gov/bills/mca/20/5/20-5-403.htm. A school may not allow a student to attend unless
Part 1: Vaccine-Preventable Diseases and Childhood Vaccines
 Part One Vaccine-Preventable Diseases and Childhood Vaccines Part 1: Vaccine-Preventable Diseases and Childhood Vaccines Diseases Here are the 14 diseases that can be prevented with routine childhood vaccination,
Part One Vaccine-Preventable Diseases and Childhood Vaccines Part 1: Vaccine-Preventable Diseases and Childhood Vaccines Diseases Here are the 14 diseases that can be prevented with routine childhood vaccination,
Pertussis. (Whole-cell pertussis vaccines) must not be frozen, but stored at 2 8 C. All wp vaccines have an expiry date of months.
 Program Management 61_5 The wp vaccines have a considerably lower price than ap vaccines and, where resources are limited and the vaccine is well accepted by the local population, wp vaccine remains the
Program Management 61_5 The wp vaccines have a considerably lower price than ap vaccines and, where resources are limited and the vaccine is well accepted by the local population, wp vaccine remains the
MONTHLY INVENTORY REPORT
 Reporting Period: Facility Name: Facility PIN: VACCINE Expiration Date A B C D E F Added (Orders Received/ Expired/ Administered Transferred Out Transferred In) Wasted Beginning of Month Inventory End
Reporting Period: Facility Name: Facility PIN: VACCINE Expiration Date A B C D E F Added (Orders Received/ Expired/ Administered Transferred Out Transferred In) Wasted Beginning of Month Inventory End
The Childhood Immunization Schedule and the National Immunization Survey
 The Childhood Immunization Schedule and the National Immunization Survey Melinda Wharton, MD, MPH Deputy Director, National Center for Immunization & Respiratory Diseases Institute of Medicine 9 February
The Childhood Immunization Schedule and the National Immunization Survey Melinda Wharton, MD, MPH Deputy Director, National Center for Immunization & Respiratory Diseases Institute of Medicine 9 February
Immunization Update Texas Immunization Conference
 Immunization Update 2017 Texas Immunization Conference Agenda NIS Child and Teen Coverage Levels Adult Program Updates TVFC Influenza Updates TVFC Program Updates 2 1 CDC National Immunization Survey Childhood
Immunization Update 2017 Texas Immunization Conference Agenda NIS Child and Teen Coverage Levels Adult Program Updates TVFC Influenza Updates TVFC Program Updates 2 1 CDC National Immunization Survey Childhood
SUMMARY OF PRODUCT CHARACTERISTICS
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Tetravac, suspension for injection Diphtheria, tetanus, pertussis (acellular, component) and poliomyelitis (inactivated) vaccine, adsorbed.
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Tetravac, suspension for injection Diphtheria, tetanus, pertussis (acellular, component) and poliomyelitis (inactivated) vaccine, adsorbed.
Enhanced immunisation schedule Victoria
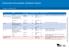 Children from March 2016 Age Disease Vaccine brand Birth Hepatitis B H-B-Vax-II Paediatric 2 months - from 6 weeks Diphtheria-tetanus-pertussis, poliomyelitis-hepatitis B- Reconstitute Site given Route
Children from March 2016 Age Disease Vaccine brand Birth Hepatitis B H-B-Vax-II Paediatric 2 months - from 6 weeks Diphtheria-tetanus-pertussis, poliomyelitis-hepatitis B- Reconstitute Site given Route
Utah s Immunization Rule Individual Vaccine Requirements
 Utah s Immunization Rule Individual Vaccine Requirements Which vaccines are required for school entry in Utah? Grades K-6: 5 doses DTaP (4 doses if the 4 th dose was given after the 4 th birthday) 4 doses
Utah s Immunization Rule Individual Vaccine Requirements Which vaccines are required for school entry in Utah? Grades K-6: 5 doses DTaP (4 doses if the 4 th dose was given after the 4 th birthday) 4 doses
Centers for Disease Control and Prevention. Final Revised Vaccine Information Materials for MMR
 This document is scheduled to be published in the Federal Register on 03/15/2018 and available online at https://federalregister.gov/d/2018-05299, and on FDsys.gov BILLING CODE: 4163-18-P DEPARTMENT OF
This document is scheduled to be published in the Federal Register on 03/15/2018 and available online at https://federalregister.gov/d/2018-05299, and on FDsys.gov BILLING CODE: 4163-18-P DEPARTMENT OF
Routine Immunization Products
 Newfoundland and Labrador Immunization Manual Section 3 Routine Immunization Products Immunization with Diphtheria, Pertussis, Tetanus, Polio and Hib Vaccine (DTap-IPV- Hib 3.1 Immunization with Diphtheria,
Newfoundland and Labrador Immunization Manual Section 3 Routine Immunization Products Immunization with Diphtheria, Pertussis, Tetanus, Polio and Hib Vaccine (DTap-IPV- Hib 3.1 Immunization with Diphtheria,
9/11/2018. Polio and Polio Vaccine. Poliomyelitis Disease. Poliovirus. First outbreak described in the U.S. in 1843
 Centers for Disease Control and Prevention National Center for Immunization and Respiratory Diseases Polio and Polio Vaccine Chapter 18 September 2018 Photographs and images included in this presentation
Centers for Disease Control and Prevention National Center for Immunization and Respiratory Diseases Polio and Polio Vaccine Chapter 18 September 2018 Photographs and images included in this presentation
ISMP National Medication Error Reporting Program and Vaccine Error Reporting Program Operated by the Institute for Safe Medication Practices
 ISMP National Medication Error Reporting Program and Vaccine Error Reporting Program Operated by the Institute for Safe Medication Practices www.ismp.org ISMP is a federally certified patient safety organization
ISMP National Medication Error Reporting Program and Vaccine Error Reporting Program Operated by the Institute for Safe Medication Practices www.ismp.org ISMP is a federally certified patient safety organization
Vaccine Information Statement: PEDIATRIC VACCINES (Multiple)
 Vaccine Information Statement: PEDIATRIC VACCINES (Multiple) 1. Your Baby s First Vaccines: What You Need to Know Babies get six vaccines between birth and 6 months of age. These vaccines protect your
Vaccine Information Statement: PEDIATRIC VACCINES (Multiple) 1. Your Baby s First Vaccines: What You Need to Know Babies get six vaccines between birth and 6 months of age. These vaccines protect your
Centers for Disease Control and Prevention. Proposed Revised Vaccine Information Materials for MMR
 This document is scheduled to be published in the Federal Register on 10/18/2016 and available online at https://federalregister.gov/d/2016-25144, and on FDsys.gov BILLING CODE: 4163-18-P DEPARTMENT OF
This document is scheduled to be published in the Federal Register on 10/18/2016 and available online at https://federalregister.gov/d/2016-25144, and on FDsys.gov BILLING CODE: 4163-18-P DEPARTMENT OF
Polio Vaccine Biological Page
 Polio Vaccine Biological Page Section 7: Biological Product Information Standard #: 07.300 Created by: Approved by: Province-wide Immunization Program Standards and Quality Province-wide Immunization Program,
Polio Vaccine Biological Page Section 7: Biological Product Information Standard #: 07.300 Created by: Approved by: Province-wide Immunization Program Standards and Quality Province-wide Immunization Program,
Routine Immunization Products
 Newfoundland and Labrador Immunization Manual Section 3 Routine Immunization Products Immunization with Diphtheria, Pertussis, Tetanus, Polio and Hib Vaccine (DTap-IPV- Hib 3.1 Immunization with Diphtheria,
Newfoundland and Labrador Immunization Manual Section 3 Routine Immunization Products Immunization with Diphtheria, Pertussis, Tetanus, Polio and Hib Vaccine (DTap-IPV- Hib 3.1 Immunization with Diphtheria,
Public Statement: Medical Policy. Effective Date: 01/01/2012 Revision Date: 03/24/2014 Code(s): Many. Document: ARB0454:04.
 ARBenefits Approval: 01/01/2012 Effective Date: 01/01/2012 Revision Date: 03/24/2014 Code(s): Many Medical Policy Title: Immunization Coverage Document: ARB0454:04 Administered by: Public Statement: 1.
ARBenefits Approval: 01/01/2012 Effective Date: 01/01/2012 Revision Date: 03/24/2014 Code(s): Many Medical Policy Title: Immunization Coverage Document: ARB0454:04 Administered by: Public Statement: 1.
