So vaccines have been licensed in a few countries. The first country to license is China, and China has also been the first country to have reported
|
|
|
- Kelley Flynn
- 5 years ago
- Views:
Transcription
1 Transcript of virtual press conference with Gregory Hartl, Spokesperson for H1N1, and Dr Marie-Paule Kieny, WHO Director of the Initiative for Vaccine Research, World Health Organization 24 September 2009 Gregory Hartl: Welcome to the WHO's virtual press briefings, today Thursday 24 September 17:00 o'clock Geneva time. My name is Gregory Hartl. The speaker today is Dr Marie-Paule Kieny, who is the Director of the Initiative for Vaccine Research, at WHO Headquarters, and she will in just a second or two she update you about what is happening with H1N1 Pandemic vaccine. One or two house-keeping matters before we go to Dr Kieny, to let you know that tomorrow, Friday, on the WHO website, you can expect to see the weekly update on the epidemiology of the pandemic and secondly, there will be an update tomorrow on antiviral and antiviral resistance. Today is for pandemic vaccine and the transcript will be up tonight on the WHO website. Welcome. I will turn over now to Dr Kieny. Dr Marie-Paule Kieny: Thank you Gregory. Good morning and good afternoon. It is a pleasure to have this opportunity, to give you the briefing on pandemic vaccines. I will try to address 3 points successively, and will try to discuss when the vaccine will be available, the second point is how much of it will it be, and the last point is for whom. So in terms of when, vaccine development is on track. In May, we announced that it will take about five months from the time of the identification of the pandemic virus to the time the first vaccine will become available for use, and not for experimental trials. So here we are and the first vaccines have indeed been licensed and plenty of results have come out of clinical trials, in China, in the USA, as well as some other places in the world. These trials have actually given very good news, and have demonstrated to immunize a protective level of immunity with a single dose of vaccine, at least in the general population. Special populations, eventually children, people with immuno-deficiencies may need to have more than one dose, but in the vast majority of people, one dose will be sufficient. The big impact of this is that with the same dose of vaccine, we will be able to immunize as many as twice the population. The second good news is that, a formulation which has been shown to work with the normal seasonal vaccination, seemed to work very well with this pandemic virus. So is this surprising? Yes, and no. Some people predicted that may be it will be possible, but many people thought of as an example of the effort they had to develop H5N1 potentially pandemic vaccines, and for H5N1, it had been very difficult to make vaccines, in the senses that either you need to have very high doses or to add adjuvant in order to reduce the dose, or/and of course, to use two doses. So this is very good news, and it shows that all manufacturers who are producing usually seasonal vaccines, will be also to make pandemic vaccines and that there will be no problem of access or intellectual property rights associated with/to adjuvant, of which most of them are proprietary. 1
2 So vaccines have been licensed in a few countries. The first country to license is China, and China has also been the first country to have reported the first clinical results. This has been followed by other countries. The US FDA has licensed vaccines as well as Hungary and many countries will come now and follow suite. Why is then that not all countries have licensed vaccines? Well not all vaccines are equal. Vaccines which have been licensed right now are vaccines which are similar to traditional seasonal vaccines, and in these, they are not new vaccines, and are registered by the USFDA, for example, a strain variation variation of seasonal vaccines. For some newer vaccines in a number of countries, they will need to have a bit more time to look at the dossier with more scrutiny, I may say, because this will be considered as new and therefore certain vaccines may take a little bit longer to be licensed and this due to the consideration. The overarching consideration that national regulatory authorities are given to safety. Now these vaccines are here, today. They will not be all. All the doses will not be available on the first day. This is quite obvious. They had started now to be deployed. The first country to deploy was China, and the Chinese have reported that 34,000 people have been immunized in the area of Beijing. Other countries are following suit. Hungary has indicated their intentions to start a roll-out vaccination campaign, on the 29 th of September, so this is next week, and other countries will follow suit. Safety? Well, there has been a report of 14 of these adverse events in China. Out of 44,000 vaccinated all these adverse events have been reported were mild. Many of them may not even be associated with the vaccine. And this is about the range that you will expect for any seasonal vaccine or any vaccine as a matter of fact. There is dizziness, headache, something which is quite common, when we administer vaccines. So is there a chance that there will be more adverse events, yes of course. There could be and these volunteers will need to be followed a lit bit longer than what we know from the outcome of the vaccination right now. And it may be when millions of population are vaccinated, there would be some incidents of severe adverse events. So these adverse events will either be investigated, all of them, and then the investigators will find that either yes, they are associated with the vaccines, and immediately, this will prompt more research into seeing if other people receiving the same type of vaccines also have had adverse events. But the vast majority will be unrelated. Because when you vaccinate thousands and millions of people, of course, some of them were going to have heart attacks, some women were going to have miscarriages, and these may - because they have just been vaccinated be associated in the mind of people with vaccinations. So there will need to be some careful analysis, to try to see which adverse event, which is really a worry, is associated with vaccination, and which are only co-incidental. Now, we have tried to address the when, so how much? At the beginning of the pandemic in May, WHO had conducted a survey, with all manufacturers that we thought will be able to make potential pandemic vaccine and we asked 36 manufacturers to give us what will be the prospect for capacity to make H1N1 vaccines. We had a 100% response and 26 manufacturers indicated their willingness to make pandemic vaccines. We asked them also to indicate how much they would be able to produce, which gave us a total of a little bit less than a 100 million doses at the best, in the best case scenario per week, which, saying that a year had 52 weeks, by just calculating and extrapolation of the weeks per production capacity to a year, gave us a total production capacity of a little bit less than 5 billion per 12 months, 4.9 to be more precise, but of course, this was a theoretical number. Where are we now? And are the assumptions that were underlying figures still thought as being realistic? Not all of them. 2
3 The first assumption was that, the yields for the quantity of vaccines that the manufacturers could produce would be similar to what they routinely have seasonal vaccines. You have seen that we have discussed numerous times that the yields were not good. We are talking recently about a third to a half of what they would expect. Now there has been good news and we know that the yields that are obtained with the latest strain which have been given to the manufacturers, are closer to what they obtained usually with seasonal vaccines, but not completely quite there yet. So the yields are a little bit lower. The second assumption is that they will be able to produce, the formulation which could give the most dose-sparing effect that they had and this is still the type of assumption that we think is correct right now. And the third assumption was that they will be producing only pandemic vaccines in their facility where they can make flu vaccines. So this we knew was unrealistic because as you know there is need to have seasonal influenza vaccines for the southern and northern hemisphere, and because of downtime that the manufacturers will have to implement in their production plan, we know that this figure of 5 billion doses, per 12 months, will not be reached. How do we think that the figure will be, right now? So our latest assumption is 3 billion doses per 12 months, into the pandemic, and we will launch another survey with the manufacturers in October, to try to refine this prediction, and to base the new figures that we will able to give on actual data and responses directly from the manufactures. Third point, for whom. Who will get the vaccine? Well, of course the first countries to receive the vaccines will be two categories of countries. First are the rich countries, with a high income. These are the ones which have already in the beginning even before the pandemic started, purchase agreements with manufacturers. They will be served first, because they have binding contracts with the manufacturers and some of them have already received vaccines in their countries and are ready to start vaccination campaigns. The other type of countries to be served very early with the vaccines is the countries that they do not have to be rich, but who have domestic production. Among those is China, as being demonstrated by the fact that China has already started to vaccinate. Another country, maybe another producing country although we do not have the precise data, may be Russia, or others. So other countries will follow suite and we will see in which order they start to vaccinate. Which population should be the first ones to be vaccinated? The WHO SAGE, which is the Strategic Advisory Group of Experts, the highest level of advisors to the Director-General on immunization matters, has recommended that all countries should vaccinate their health workers. These would represent about 2 percent of the world's population, and WHO is working currently with donors, with manufacturers, through donations, and when we have, or when we do not have enough donations, we have a purchase agreement with the manufacturers to obtain vaccines at reduced prices for the developing countries. We are trying, the Director-General has announced that she will try to obtain enough vaccines to be able to provide to countries in need, which are the countries that will not have access without WHO, to cover up to 10% of their population. So this will cover - about 2% of the population that will be health workers, and other populations to be decided among the priority groups by the national governments. Indeed the choice of a population to be vaccinated is a national prerogative and each country will have to take this decision in view of their own epidemiological and national characteristics. So we hope that the vaccines will be made available to most people in need, in all countries as quickly as possible. WHO expects to receive the first doses of vaccines to be distributed to the poor countries somewhere by end of October; we may be able to start the first delivery during November, or this is still exactly to be defined. We need to discuss with the governments of the receiving countries to see when they will be ready to receive the vaccines.
4 Now there have been many questions about the health workers. Does it mean that the fact that WHO has recommended that health workers should be vaccinated. Does it mean that there will be an obligation for all health workers to be vaccinated? Well, WHO does not recommend that vaccinating is mandatory. It is up to a person to decide. It is a voluntary choice to be vaccinated. Nevertheless, there are plenty of reasons for health workers to be willing to be vaccinated. One is to protect themselves because they may be more exposed than other people because they will be caring for sick patients, with the H1N1 pandemic flu. The second is also to help maintain a functional health system, because if health workers are sick and have to stay at home, then there is nobody to take care of the patients from influenza or other patients. And the third reason, which calls really upon the social responsibility of the health workers, is that health workers who would be sick with pandemic flu, are or will be putting their patients at risk of getting infected themselves. So for all these reasons, we think that they are good reasons for the health workers to seek vaccinations against pandemic flu. So where are we now with WHO in terms of working on access for pandemic influenza vaccine? As we have said before numerous times, we have been offered 150 million doses from two manufacturers. This is still the same. We are discussing with more manufacturers who have expressed interest to give us vaccines. There has been also announced very recently in the press and we are discussing with governments that some nine governments right now have made an official offer to WHO to give up to 10% of the supplies that they have bought for themselves. And we really look forward to discussing with those and other governments and other stakeholders, to help to get more equity in the distribution of this very important vaccine. Indeed as you know again, vaccines are the most important tool to combat infections and pandemic influenza vaccine, and pandemic influenza in particular. So we count on the generosity of the governments of the manufacturers to help WHO and the international stakeholders to make more equity in this world. Thanks for your attention. Gregory Hartl: Dr Kieny, thank you very much. Now before we open it up for questions, just a couple of reminders, that the transcripts and the audio file for this briefing will be available on the WHO website, shortly after the briefing is over, and to all journalists listening if you would like to ask a question, dial 01 on your keypad to enter the question cue. So the first question is from: Frank Jordan, Associated Press: Hi Dr Kieny. It seems obvious now that not all of the world's population will be getting the vaccine in the first year. In fact, you said there will only be 3 billion doses available. So the question is how are you going to prioritize and what percentage of the population definitely needs the vaccine? I will appreciate if you could talk a little bit about that, and also mention which populations you think will need two doses. Dr Marie-Paule Kieny: In terms of the countries, we think that, as already said, all countries will need access to vaccines so we are working with the Regional Offices to identify those countries which are the most in need. Our estimate is that we will have to cover countries accounting for about three billion of the population so a little bit less than half of the world and as I said we intend to provide these countries up to 10% of the population-worth of vaccines and will use the 150 million doses/donations that we have from manufacturers as well as donations from governments and we will also purchase vaccines if needed. So who would be in this population? As we said, the health workers would be the first priority and then it would be up to the decision of the national governments, they would consider, of course, people with underlying health conditions such as people with asthma, with a cardiac condition, people with immunodeficiency, people with other health problems of all ages, pregnant women and other minorities for some countries - so it will really be each country's government to make a detailed analysis of which population at the national level will most benefit from pandemic immunization. 4
5 So of course we are lucky that the pandemic is moderate in severity that most people experience a mild illness and recover spontaneously, therefore although one would want to protect everybody. Of course one always wants to give the maximum protection to everybody, it is also clear that most people will do well without vaccines. In terms of the number of doses it is clear that in clinical studies so far (we are not at the end of it) - we don t know yet for example what the duration of protection induced by a single dose of vaccine - the earliest data that we have indicates that at least in adults and in adolescents, one dose is sufficient, so in young children in special groups it is probable that two doses would be needed. Not all vaccines have been tested but generally it is likely to be one dose. Gregory Hartl: Thank you very much so the next question will be from Elizabeth Sukkar. Go ahead please. Sukkar: Hello. It is to do with the regulatory process to approve the vaccines, the US versus the EU, do they use similar application processes and secondly recently in Australia the CSL vaccines were approved there for an age group of ten years and over strangely the US FDA approved the same vaccines for eighteen years and upwards, so I was just wondering if you can comment on those two new questions? Kieny: So now all countries have slightly different regulatory pathways as you know. So the vaccines which have been licensed so far by the US FDA have been licensed on the basis of a strain change, which is that they were considered as seasonal vaccines with a different strain which is a pandemic one. Therefore in the US the vaccines have obtained the licensing indications so for the population for which these vaccines are licensed for seasonal flu in the US. So the vaccines where the corresponding vaccine is licensed six months to no limit of age, this is the indication that the pandemic vaccine will get into the US, because it is a strain change. In Europe where there is not this procedure of strain change, there are two procedures in place, one is the mock-up dossier which is to register vaccines which are similar and which are made out of a model which was the H5N1 vaccine prototype, and other vaccines will take a little bit longer because they will have to undergo a full regulatory approval. In Australia they have their own way of licensing vaccines; the producer could come with clinical trial results in the population covering the indication that was given. So yes it is the case that all the products are different and also that all the pathways or most pathways where the regulators are different and therefore this is why the vaccines get different indications. Hartl: Thank you the next question is from Mr Gold of Deutsche Presseagentur in Geneva, go ahead please. Gold: I just wanted to ask about this issue of donations to the developing world and if you could be a bit more specific about what exactly, what amounts you will be expecting the developing world to receive and secondly you said that it would be up to the governments to decide how to distribute that, do you have any concerns that certain governments might not distribute the vaccines to the most vulnerable populations and might do it on a favouritism basis? Kieny: Well we hope not! The governments are usually very responsible for that, one of the strict recommendations that we have is that it is used for health workers so that is really an important recommendation that we give to these governments. One other important factor before we deliver the vaccine: we need to have an assessment of the readiness of a country to distribute the vaccine and the readiness will include, of course, having a plan and having identified the target groups and we will review all this with the government before we send the vaccines, because nobody wants the vaccines to be sent and to stay in a warehouse for months or go wasted, even worse! So therefore one of the important factors that will help us define at what time point a particular county receives vaccines is the readiness and as I said we need to identify the priority groups because if we need to target health workers or pregnant women or children these are completely different strategies that
6 we need to put in place to be sure that we reach the maximum number of this type of category of people. So we are really confident that the vaccine will be put to good use. I didn t respond to the beginning of your question, sorry. The number of doses, as I said we have currently access to 150 million doses through the manufacturers. With the calculation of what we know of what the countries who are willing to give us 10% of their own supply have purchased on their own, we know that from this we are likely to receive 50 more million doses, so that this would get us to 200 million doses. We hear and we are in contact with other governments who are willing to do something also to support this effort so this quantity is very likely to increase and we will try our best with funds raised by sister UN Organizations to be able to purchase what will be missing to allow us to deliver up to 10% in each of the countries. Hartl: Thank you very much, so just a reminder to those listening if you would like to ask a question dial 01 on your key pad to enter the queue. The next question is from Helen Branswell of Canadian Press, go ahead please. Branswell: You said that at this point you think that the estimate is more likely to be three billion doses over twelve months. Does that number hold if manufactures resume making seasonal flu vaccines for the Southern and Northern Hemisphere? Kieny: Well this is what we factored in in decreasing from five billion to three billion. We don t know for the time being exactly how many vaccines will need to be produced for the seasonal immunization and there haven't been any WHO recommendations on the formulation of the seasonal vaccine. So you may have seen that the Melbourne expert meeting this week has made a recommendation of the composition, not on the formulation, of seasonal vaccination of the Southern Hemisphere and that the pandemic strain is among the strain identified. So this and whether the pandemic strain is introduced in the seasonal vaccine or is kept as a stand-alone will have an impact on the number of doses so we will - this is an estimate for the time being and we will try our best in October to refine it as I said based on real data. Hartl: Thank you very much, the next question is from Mr Mackmillen, go ahead please. Mackmillen: I comment that it would be ideal to vaccinate the entire population at the same time but that would not be possible, what could possibly be the drawback in vaccinating half the world and let's say the other half not being vaccinated? I am referring to the chances of the virus becoming resistant sooner than we expected because there will be this divide between those who have the vaccination and those who do not have it. Kieny: Well we hope that the whole world will have some access to vaccinations, this is certainly the commitment of the Director-General and the Secretary General of the UN, not to let a country to be without a vaccine so of course, you are right, in some countries it will be possible to vaccinate the whole population, in some other countries it will be only 10%. So will this have a role to play in the apparition of variants of drift variants, this is not sure. You know the influenza virus is unstable by nature so we know it will evolve. For the time being it hasn t changed and the viruses circulating are still like the ones which have been isolated at the beginning. So what will happen if a virus drifts, changes, well if it doesn t change too much the vaccine that we have now will still be efficient. If a virus drifts a lot then a new formulation of vaccine will have to be developed and produced. So we hope that the best use will be made of about three billion doses that the world will be able to produce. Hartl: Thank you very much, the next question from Ms Favaro, CTV. Go ahead please. Favaro: Two questions. Number one, would it have been better for the world to go with just one vaccine so that one didn t have this difference between countries and its readiness and its testing and number two I am wondering if you are aware of this unpublished study 6
7 from Canada that is linking prior seasonal flu vaccinations to an increased risk of H1N1 infection and if you have seen it and what the implications might be. Kieny: So in terms of having one vaccine, of course it would make logistics easier, but there are individual manufacturers and they all make their own vaccines. There are historical reasons for this - there are all intellectual property rights reasons from time to time and regulatory reasons why they might be different. So it is like as if you have different cooks in different countries all making a choucroute, this may be different or roast beef or something and it may taste a little bit different - this is why these vaccines are also different. In terms of the Canadian study which is still unpublished of course, we have heard about it and we are of course linking with the investigators. This data for those of you who are not aware of it yet seems to indicate that having received a seasonal vaccine puts you more at risk of contacting H1N1 pandemic influenza or be infected. So we are trying to discuss with other experts and the Canadians themselves are trying to put a panel of experts together who will allow them to review their data. These investigators are well known, they are very capable but all studies can have biases - we are in contact also with investigators in other countries knowing these findings, [they] have tried they have looked in their own data in their own country whether they could find a similar observation and none of the other countries have been able to find anything like this. So the reason why this may be different in Canada and in this particular study as in other places in the world is not yet identified. It may be that it may be a study bias, it may be that something is real but certainly the WHO as well as the regulators in all these countries are looking forward to be able to see the data, to study the data and to come with a better understanding of whether this has any chance of putting people at risk, the fact that they have received the seasonal vaccination. I must say also that years and years of seasonal vaccination has not indicated that a previous vaccination has an adverse impact but again more data, much more investigation of data already collected and investigation of new data will be needed to see if this is really real. Hartl: Thank you very much, next questions from Jennifer Tryon, go ahead please. Tryon: You have indicated nine countries are willing to donate up to 10% of their countries supply, is Canada one of those countries and can you list the other countries. Kieny: I will have a hard time to name the countries, they have been put in a press release so I don t want to miss any one of them but I will try. The US started the movement, there is the UK, France, Norway, Brazil, Italy, New Zealand???, Switzerland and not Canada yet. We are discussing with Canada and we will see. Canada and other countries may be coming forward, maybe not on the initial list but we hope that as many as possible will come forward and will help with providing another vaccine. Hartl: Thank you very much, next question is from Ms Udi from Japan. Udi: I have two questions, one is concerning the early data of the clinical trial, you said for healthy adults one dose is enough, I would like to know why only one dose is enough, does that mean that some adults have some immunity for the pandemic influenza or not, and the second question is concerning the safety of the vaccine with a new type of adjuvant and produced by cell culture. Kieny: Why one dose, it is true that it is a little bit striking because if everybody is naive which we would expect with a new virus and a new pandemic virus you would expect that one would need two doses - it may well be that there is enough similarity between this H1N1 virus and other H1N1 viruses which have been circulated before to make it that people are already primed, they are not immune but they have already seen something which resembles this virus and therefore they respond immediately after the first dose. It is interesting in the sense that numerous experiments have shown that immunization against the seasonal flu of last year for example did not induce any immune response that was
8 neutralizing the pandemic virus so it seems to function in one direction and maybe not in the other one. In terms of safety, the safety of seasonal vaccine without adjuvants has been demonstrated in millions and millions of people in all types of populations from 6 months to old people, pregnant women. In fact it is recommended for pregnant women. So this is one thing, the other vaccines which use adjuvants are newer there is no doubt that the safety data base is not as large, nevertheless the safety data base with one of the adjuvant vaccines, the one from Novartis includes 40 million individuals, usually elderly people but still it is quite a large safety date base. The safety data base with the other adjuvanted vaccines - the one from GSK - also includes tens of thousands of people - is not that high, it hasn t been tested or evaluated in special groups of the population. We have currently no reason to think that they will not be safe. We had an expert consultation on this topic in June, and the experts opinion was that, in all likelihood, these vaccines will be very safe. David Brown, The Washington Post: Can you give an estimate of how many countries are going to be dependent on WHO for their supply of vaccines and what 10% of that total population, the summed population of those countries is? Dr Marie-Paule Kieny: We estimate that the population that will be dependent on WHO is about 3 billion people, and this is a little bit more than 90 countries. Therefore, 10% of these 3 billion would be 300 million doses and this is our target in terms of getting access to vaccines right now. Melda Albano: I would like to get your spot on the fact that developing countries like the Philippines cannot totally complete their vaccination in the purchase of the H1N1 vaccines. Prices are expected to go up because of the big demand. Dr Marie-Paule Kieny: We are aware of the fact that many of the developing countries have two hurdles to get access to vaccines. The first one at the very beginning was the price of the vaccines. Of course, vaccine is expensive and some countries don t have the financial means to buy pandemic vaccine. The other one is also supply because of the limited number of doses of pandemic vaccine which can be produced in a year this is what we discussed a bit earlier, and therefore because a lot of countries have already placed their orders, there is no offer any more, or very little. This is exactly for this reason that WHO is looking forward with the help of the other stakeholders to provide direct access through donations to these countries of pandemic vaccine. 8
Transcript of virtual press conference with Dr Marie-Paule Kieny, Director, Initiative for Vaccine Research World Health Organization 19 November 2009
 Transcript of virtual press conference with Dr Marie-Paule Kieny, Director, Initiative for Vaccine Research World Health Organization 19 November 2009 Nyka Alexander, WHO communications officer: Good afternoon.
Transcript of virtual press conference with Dr Marie-Paule Kieny, Director, Initiative for Vaccine Research World Health Organization 19 November 2009 Nyka Alexander, WHO communications officer: Good afternoon.
Transcript of virtual press conference with Dr Keiji Fukuda, Special Adviser to the Director-General on Pandemic Influenza
 Transcript of virtual press conference with Dr Keiji Fukuda, Special Adviser to the Director-General on Pandemic Influenza 18 February 2010 Gregory Hartl: Good afternoon. It is just gone one o'clock in
Transcript of virtual press conference with Dr Keiji Fukuda, Special Adviser to the Director-General on Pandemic Influenza 18 February 2010 Gregory Hartl: Good afternoon. It is just gone one o'clock in
Transcript of virtual press conference with Dr Keiji Fukuda, Special Adviser to the Director-General on Pandemic Influenza, World Health Organization
 Transcript of virtual press conference with Dr Keiji Fukuda, Special Adviser to the Director-General on Pandemic Influenza, World Health Organization 17 December 2009 Nyka Alexander: Good afternoon. We
Transcript of virtual press conference with Dr Keiji Fukuda, Special Adviser to the Director-General on Pandemic Influenza, World Health Organization 17 December 2009 Nyka Alexander: Good afternoon. We
12 November NB: there are some passages in this transcript which have not been transcribed because of poor audio quality.
 Transcript of virtual press conference with Gregory Hartl, Spokesperson for H1N1, and Dr Nikki Shindo, Medical Officer, Global Influenza Programme, World Health Organization 12 November 2009 NB: there
Transcript of virtual press conference with Gregory Hartl, Spokesperson for H1N1, and Dr Nikki Shindo, Medical Officer, Global Influenza Programme, World Health Organization 12 November 2009 NB: there
identified, both by the country and by the company and it was quickly acted upon. The information was gotten out and the appropriate steps were
 Transcript of virtual press conference with Kristen Kelleher, Communications Officer for pandemic (H1N1) 2009, and Dr Keiji Fukuda, Special Adviser to the Director-General on Pandemic Influenza, World
Transcript of virtual press conference with Kristen Kelleher, Communications Officer for pandemic (H1N1) 2009, and Dr Keiji Fukuda, Special Adviser to the Director-General on Pandemic Influenza, World
and these are more regular parts of influenza as a general disease but there are also some features which are clearly different.
 Transcript of virtual press conference with Gregory Hartl, spokesperson for pandemic (H1N1) 2009, and Dr Keiji Fukuda, Special Adviser to the Director-General on Pandemic Influenza, World Health Organization
Transcript of virtual press conference with Gregory Hartl, spokesperson for pandemic (H1N1) 2009, and Dr Keiji Fukuda, Special Adviser to the Director-General on Pandemic Influenza, World Health Organization
Update on A(H1N1) pandemic and seasonal vaccine availability. July 7, 2009
 Update on A(H1N1) pandemic and seasonal vaccine availability July 7, 2009 Presentation objectives and approach Presentation Objectives Review production status for 2009-2010 Northern Hemisphere vaccine
Update on A(H1N1) pandemic and seasonal vaccine availability July 7, 2009 Presentation objectives and approach Presentation Objectives Review production status for 2009-2010 Northern Hemisphere vaccine
or seen with seasonal influenza. So we will continue to follow this and see how the picture of clinical symptoms evolves.
 Transcript of Virtual Press conference with Dr Keiji Fukuda, Assistant Director-General ad. Interim for Health Security and Environment World Health Organization 29 April 2009 Dr Fukuda: Good afternoon
Transcript of Virtual Press conference with Dr Keiji Fukuda, Assistant Director-General ad. Interim for Health Security and Environment World Health Organization 29 April 2009 Dr Fukuda: Good afternoon
Transcript of press briefing at the Palais des Nations, Geneva Dr Keiji Fukuda, Special Adviser to the Director-General on Pandemic Influenza
 Transcript of press briefing at the Palais des Nations, Geneva Dr Keiji Fukuda, Special Adviser to the Director-General on Pandemic Influenza 29 March 2010 Gregory Hartl: without further ado, we will ask
Transcript of press briefing at the Palais des Nations, Geneva Dr Keiji Fukuda, Special Adviser to the Director-General on Pandemic Influenza 29 March 2010 Gregory Hartl: without further ado, we will ask
An Update on BioMarin Clinical Research and Studies in the PKU Community
 An Update on BioMarin Clinical Research and Studies in the PKU Community Barbara Burton, MD, Professor of Pediatrics, Northwestern University Feinberg School of Medicine, Director of PKU Clinic, Children
An Update on BioMarin Clinical Research and Studies in the PKU Community Barbara Burton, MD, Professor of Pediatrics, Northwestern University Feinberg School of Medicine, Director of PKU Clinic, Children
H1N1 Vaccine Based on CDCs ACIP Meeting, July 29, 2009
 August 6, 2009 H1N1 Vaccine Based on CDCs ACIP Meeting, July 29, 2009 CDC s Advisory Committee on Immunization Practices (ACIP), a panel made up of medical and public health experts, met July 29, 2009,
August 6, 2009 H1N1 Vaccine Based on CDCs ACIP Meeting, July 29, 2009 CDC s Advisory Committee on Immunization Practices (ACIP), a panel made up of medical and public health experts, met July 29, 2009,
other chronic conditions, which can be illnesses such as diabetes or lung disease or some form of immunodeficiency and so on. This is in-keeping with
 Transcript of virtual press conference with Gregory Hartl, WHO Spokesperson for Epidemic and Pandemic Diseases, and Dr Keiji Fukuda, Assistant Director-General ad Interim for Health Security and Environment,
Transcript of virtual press conference with Gregory Hartl, WHO Spokesperson for Epidemic and Pandemic Diseases, and Dr Keiji Fukuda, Assistant Director-General ad Interim for Health Security and Environment,
FINAL RECOMMENDATIONS ON PANDEMIC INFLUENZA
 Comprehensive Family Immunization Family and Community Health Area FINAL RECOMMENDATIONS ON PANDEMIC INFLUENZA TECHNICAL ADVISORY GROUP ON VACCINE-PREVENTABLE DISEASES 24-26 AUGUST 2009, COSTA RICA Epidemiological
Comprehensive Family Immunization Family and Community Health Area FINAL RECOMMENDATIONS ON PANDEMIC INFLUENZA TECHNICAL ADVISORY GROUP ON VACCINE-PREVENTABLE DISEASES 24-26 AUGUST 2009, COSTA RICA Epidemiological
Working Papers Project on the Public and Biological Security Harvard School of Public Health 17.
 Working Papers Project on the Public and Biological Security Harvard School of Public Health 17. FLU VACCINE SURVEY Robert J. Blendon, Harvard School of Public Health, Project Director John M. Benson,
Working Papers Project on the Public and Biological Security Harvard School of Public Health 17. FLU VACCINE SURVEY Robert J. Blendon, Harvard School of Public Health, Project Director John M. Benson,
Flu Vaccines: Questions and Answers
 Flu Vaccines: Questions and s Question 1 Does the flu shot give me the flu? Does the flu shot give me the flu? I heard people get sick after the shot. Well, I m really glad you asked that question about
Flu Vaccines: Questions and s Question 1 Does the flu shot give me the flu? Does the flu shot give me the flu? I heard people get sick after the shot. Well, I m really glad you asked that question about
COMMISSION OF THE EUROPEAN COMMUNITIES COMMISSION STAFF WORKING DOCUMENT. Vaccination strategies against pandemic (H1N1) 2009.
 COMMISSION OF THE EUROPEAN COMMUNITIES Brussels, 15.9.2009 SEC(2009) 1189 final COMMISSION STAFF WORKING DOCUMENT Vaccination strategies against pandemic (H1N1) 2009 accompanying the COMMUNICATION FROM
COMMISSION OF THE EUROPEAN COMMUNITIES Brussels, 15.9.2009 SEC(2009) 1189 final COMMISSION STAFF WORKING DOCUMENT Vaccination strategies against pandemic (H1N1) 2009 accompanying the COMMUNICATION FROM
National Inspection of services that support looked after children and care leavers
 National Inspection of services that support looked after children and care leavers Introduction Children and young people that are looked after and those leaving care need the best support possible. Support
National Inspection of services that support looked after children and care leavers Introduction Children and young people that are looked after and those leaving care need the best support possible. Support
Questions and Answers Regarding Federal Contracts for State-Purchased Antiviral Drugs (Relenza & Tamiflu ) (07/26/06)
 Questions and Answers Regarding Federal Contracts for State-Purchased Antiviral Drugs (Relenza & Tamiflu ) (07/26/06) These questions were received in response to the recently executed contracts with Roche
Questions and Answers Regarding Federal Contracts for State-Purchased Antiviral Drugs (Relenza & Tamiflu ) (07/26/06) These questions were received in response to the recently executed contracts with Roche
Influenza Pandemic (H1N1) 2009
 Influenza Pandemic (H1N1) 2009 7 th Congress on Communicable Diseases Lisboa 29 January 2010 World Health Organization Overview Since April 2009, first influenza pandemic of 21 st century underway Most
Influenza Pandemic (H1N1) 2009 7 th Congress on Communicable Diseases Lisboa 29 January 2010 World Health Organization Overview Since April 2009, first influenza pandemic of 21 st century underway Most
The HPV Data Is In What Do the Newest Updates in Screening Mean For Your Patients?
 Transcript Details This is a transcript of a continuing medical education (CME) activity accessible on the ReachMD network. Additional media formats for the activity and full activity details (including
Transcript Details This is a transcript of a continuing medical education (CME) activity accessible on the ReachMD network. Additional media formats for the activity and full activity details (including
Why Tobacco Cessation?
 Tobacco Cessation in Community Settings Introduction Hello and welcome to the Learning and Action Network event, Reaching Those in Need of Tobacco Cessation in Community Settings: Research, Recommendations
Tobacco Cessation in Community Settings Introduction Hello and welcome to the Learning and Action Network event, Reaching Those in Need of Tobacco Cessation in Community Settings: Research, Recommendations
Should i get the H1N1 Vaccine?
 Should i get the H1N1 Vaccine? 2 comments and 10 questions.. 12 slides Dr. Mike Evans Associate Professor, Family & Community Medicine, University of Toronto, Staff Physician, St. Michael s Hospital Director,
Should i get the H1N1 Vaccine? 2 comments and 10 questions.. 12 slides Dr. Mike Evans Associate Professor, Family & Community Medicine, University of Toronto, Staff Physician, St. Michael s Hospital Director,
Prepare Your Practice To Fight Flu: Make a Strong Influenza Vaccine Recommendation and Improve Your Influenza Vaccination Rates This Season
 Prepare Your Practice To Fight Flu: Make a Strong Influenza Vaccine Recommendation and Improve Your Influenza Vaccination Rates This Season The thing that motivates me to FIGHT FLU is the ability to prevent
Prepare Your Practice To Fight Flu: Make a Strong Influenza Vaccine Recommendation and Improve Your Influenza Vaccination Rates This Season The thing that motivates me to FIGHT FLU is the ability to prevent
Genetic Counselor: Hi Lisa. Hi Steve. Thanks for coming in today. The BART results came back and they are positive.
 Hi, I m Kaylene Ready, a genetic counselor who specializes in the education and counseling of individuals at high-risk for hereditary breast and ovarian cancer syndrome. Women with an inherited BRCA 1
Hi, I m Kaylene Ready, a genetic counselor who specializes in the education and counseling of individuals at high-risk for hereditary breast and ovarian cancer syndrome. Women with an inherited BRCA 1
Press conference with Dr Keiji Fukuda, Assistant Director-General for Health Security and the Environment on MERS-Coronavirus, 25 September 2013
 Press conference with Dr Keiji Fukuda, Assistant Director-General for Health Security and the Environment on MERS-Coronavirus, 25 September 2013 Speaker key KF GH WN HB EK MK FD SB BR BD AK Keiji Fukuda
Press conference with Dr Keiji Fukuda, Assistant Director-General for Health Security and the Environment on MERS-Coronavirus, 25 September 2013 Speaker key KF GH WN HB EK MK FD SB BR BD AK Keiji Fukuda
Recording Transcript Wendy Down Shift #9 Practice Time August 2018
 Recording Transcript Wendy Down Shift #9 Practice Time August 2018 Hi there. This is Wendy Down and this recording is Shift #9 in our 6 month coaching program. [Excuse that I referred to this in the recording
Recording Transcript Wendy Down Shift #9 Practice Time August 2018 Hi there. This is Wendy Down and this recording is Shift #9 in our 6 month coaching program. [Excuse that I referred to this in the recording
Individual Packet. Instructions
 Individual Packet Instructions Step : Introductions and Instructions ( minutes). Start by having each person introduce themselves including their name and what they found most interesting about the introductory
Individual Packet Instructions Step : Introductions and Instructions ( minutes). Start by having each person introduce themselves including their name and what they found most interesting about the introductory
Nova Scotia s Response to H1N1. Summary Report
 Nova Scotia s Response to H1N1 Summary Report December 2010 H1N1 Summary Report l 1 Introduction In April 2009, an outbreak of a new virus called H1N1 influenza was identified in Veracruz, Mexico. As the
Nova Scotia s Response to H1N1 Summary Report December 2010 H1N1 Summary Report l 1 Introduction In April 2009, an outbreak of a new virus called H1N1 influenza was identified in Veracruz, Mexico. As the
PCSK9 Antibodies for Dyslipidemia: Efficacy, Safety, and Non-Lipid Effects
 Transcript Details This is a transcript of an educational program accessible on the ReachMD network. Details about the program and additional media formats for the program are accessible by visiting: https://reachmd.com/programs/lipid-luminations/pcsk9-antibodies-dyslipidemia-efficacy-safety-and-nonlipid-effects/8335/
Transcript Details This is a transcript of an educational program accessible on the ReachMD network. Details about the program and additional media formats for the program are accessible by visiting: https://reachmd.com/programs/lipid-luminations/pcsk9-antibodies-dyslipidemia-efficacy-safety-and-nonlipid-effects/8335/
How you can help support the Beat Flu campaign
 How you can help support the Beat Flu campaign Toolkit www.beatflu.org f T Beat Flu 1 How can you help Beat Flu? Template news story You may wish to use the following news story as a template, which can
How you can help support the Beat Flu campaign Toolkit www.beatflu.org f T Beat Flu 1 How can you help Beat Flu? Template news story You may wish to use the following news story as a template, which can
Stockpiling of H5N1 vaccines
 Stockpiling of H5N1 vaccines Norbert W. Hehme EVM Influenza Pandemic Working Group EU-Commission Workshop on Stockpiling of Medicinal Products February 21/22, 2008 European Vaccine Manufacturers 1 (EVM)
Stockpiling of H5N1 vaccines Norbert W. Hehme EVM Influenza Pandemic Working Group EU-Commission Workshop on Stockpiling of Medicinal Products February 21/22, 2008 European Vaccine Manufacturers 1 (EVM)
Addressing Information Gaps in Advanced Prenatal Screening: What Your Expecting Patients Need to Know
 Transcript Details This is a transcript of an educational program accessible on the ReachMD network. Details about the program and additional media formats for the program are accessible by visiting: https://reachmd.com/programs/medical-industry-feature/addressing-information-gaps-advancedprenatal-screening-what-your-expecting-patients-need-know/7651/
Transcript Details This is a transcript of an educational program accessible on the ReachMD network. Details about the program and additional media formats for the program are accessible by visiting: https://reachmd.com/programs/medical-industry-feature/addressing-information-gaps-advancedprenatal-screening-what-your-expecting-patients-need-know/7651/
Gregory Hartl: Steve Connor, The Independent: Gregory Hartl: Saki Ouchi, Japanese broadcast: Dr Ben Embarek:
 Transcript of virtual press conference with Gregory Hartl, WHO Spokesperson for Epidemic and Pandemic Diseases, and Dr Peter Ben Embarek, WHO Food Safety Scientist, World Health Organization 3 May 2009
Transcript of virtual press conference with Gregory Hartl, WHO Spokesperson for Epidemic and Pandemic Diseases, and Dr Peter Ben Embarek, WHO Food Safety Scientist, World Health Organization 3 May 2009
BBC Learning English 6 Minute English 2 October 2014 Sleeping on the job
 BBC Learning English 6 Minute English 2 October 2014 Sleeping on the job NB: This is not a word for word transcript Hello I'm. Welcome to 6 Minute English. I'm joined today by. Hello..? Hello? Oh sorry,
BBC Learning English 6 Minute English 2 October 2014 Sleeping on the job NB: This is not a word for word transcript Hello I'm. Welcome to 6 Minute English. I'm joined today by. Hello..? Hello? Oh sorry,
PART I. Answer Questions 1-10 with reference to TEXT 1 that follows. Only one option is correct for each question.
 PART I Answer Questions 1-10 with reference to TEXT 1 that follows. Only one option is correct for each question. 1. The title of the article: A. suggests that influenza is no longer a major public health
PART I Answer Questions 1-10 with reference to TEXT 1 that follows. Only one option is correct for each question. 1. The title of the article: A. suggests that influenza is no longer a major public health
Cross-Border Cooperation in the Global Fight against Communicable Diseases
 "Cross-Border Cooperation in the Global Fight against Communicable Diseases," East Asian Regional Cooperation in the Fight Against HIV/AIDS, Tuberculosis, and Malaria; (Beijing Conference, 2006), Tokyo:
"Cross-Border Cooperation in the Global Fight against Communicable Diseases," East Asian Regional Cooperation in the Fight Against HIV/AIDS, Tuberculosis, and Malaria; (Beijing Conference, 2006), Tokyo:
UNDERSTANDING CAPACITY & DECISION-MAKING VIDEO TRANSCRIPT
 I m Paul Bourque, President and CEO of the Investment Funds Institute of Canada. IFIC is preparing materials to assist advisors and firms in managing effective and productive relationships with their aging
I m Paul Bourque, President and CEO of the Investment Funds Institute of Canada. IFIC is preparing materials to assist advisors and firms in managing effective and productive relationships with their aging
Dr. Coakley, so virtual colonoscopy, what is it? Is it a CT exam exactly?
 Virtual Colonoscopy Webcast January 26, 2009 Fergus Coakley, M.D. Please remember the opinions expressed on Patient Power are not necessarily the views of UCSF Medical Center, its medical staff or Patient
Virtual Colonoscopy Webcast January 26, 2009 Fergus Coakley, M.D. Please remember the opinions expressed on Patient Power are not necessarily the views of UCSF Medical Center, its medical staff or Patient
Scouter Support Training Participant Workbook
 Scouter Support Training Participant Workbook Version 2.1 June 15, 2011 Minor changes November 5, 2012 For use with the Scouter Support Playbook Scouter Support Training: Trainer s Manual 2 Participant
Scouter Support Training Participant Workbook Version 2.1 June 15, 2011 Minor changes November 5, 2012 For use with the Scouter Support Playbook Scouter Support Training: Trainer s Manual 2 Participant
UCT Conference Incoming President s Speech Delivered by UCT President Chris Phelan July 4, 2018
 UCT Conference 2018 - Incoming President s Speech Delivered by UCT President Chris Phelan July 4, 2018 Change is the law of life. Those who look only to the past and present are certain to miss the future.
UCT Conference 2018 - Incoming President s Speech Delivered by UCT President Chris Phelan July 4, 2018 Change is the law of life. Those who look only to the past and present are certain to miss the future.
Global Perspectives on Organ Donation
 Transcript Details This is a transcript of an educational program accessible on the ReachMD network. Details about the program and additional media formats for the program are accessible by visiting: https://reachmd.com/programs/focus-on-global-medicine/global-perspectives-on-organ-donation/3990/
Transcript Details This is a transcript of an educational program accessible on the ReachMD network. Details about the program and additional media formats for the program are accessible by visiting: https://reachmd.com/programs/focus-on-global-medicine/global-perspectives-on-organ-donation/3990/
Sam: Annette, can we just start out with a brief simple explanation of what neuro-immune diseases are, including ME and CFS?
 Sam: Annette, can we just start out with a brief simple explanation of what neuro-immune diseases are, including ME and CFS? Annette: They are systemic diseases that impact the nervous system and immune
Sam: Annette, can we just start out with a brief simple explanation of what neuro-immune diseases are, including ME and CFS? Annette: They are systemic diseases that impact the nervous system and immune
9 INSTRUCTOR GUIDELINES
 STAGE: Ready to Quit You are a clinician in a family practice group and are seeing 16-yearold Nicole Green, one of your existing patients. She has asthma and has come to the office today for her yearly
STAGE: Ready to Quit You are a clinician in a family practice group and are seeing 16-yearold Nicole Green, one of your existing patients. She has asthma and has come to the office today for her yearly
Vaccine Financing and Delivery: Room for Improvement
 Transcript Details This is a transcript of an educational program accessible on the ReachMD network. Details about the program and additional media formats for the program are accessible by visiting: https://reachmd.com/programs/focus-on-public-health-policy/vaccine-financing-and-delivery-room-forimprovement/3738/
Transcript Details This is a transcript of an educational program accessible on the ReachMD network. Details about the program and additional media formats for the program are accessible by visiting: https://reachmd.com/programs/focus-on-public-health-policy/vaccine-financing-and-delivery-room-forimprovement/3738/
In some states, prisoners beat out priority groups
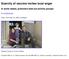 Scarcity of vaccine incites local anger In some states, prisoners beat out priority groups By Jeff McDonald Friday, November 20, 2009 at midnight Eduardo Contreras / Union-Tribune Coralynn McCoy, 4, with
Scarcity of vaccine incites local anger In some states, prisoners beat out priority groups By Jeff McDonald Friday, November 20, 2009 at midnight Eduardo Contreras / Union-Tribune Coralynn McCoy, 4, with
How to Work with the Patterns That Sustain Depression
 How to Work with the Patterns That Sustain Depression Module 5.2 - Transcript - pg. 1 How to Work with the Patterns That Sustain Depression How the Grieving Mind Fights Depression with Marsha Linehan,
How to Work with the Patterns That Sustain Depression Module 5.2 - Transcript - pg. 1 How to Work with the Patterns That Sustain Depression How the Grieving Mind Fights Depression with Marsha Linehan,
B.C. s Response to the H1N1 Pandemic. A Summary Report
 B.C. s Response to the H1N1 Pandemic A Summary Report June 2010 ii A Summary Report by British Columbia s Provincial Health Officer June 2010 B.C. s Response to the H1N1 Pandemic 1 Table of Contents INTRODUCTION...........................................................................
B.C. s Response to the H1N1 Pandemic A Summary Report June 2010 ii A Summary Report by British Columbia s Provincial Health Officer June 2010 B.C. s Response to the H1N1 Pandemic 1 Table of Contents INTRODUCTION...........................................................................
Pandemic lessons learnt
 Pandemic lessons learnt PCWP-HCPWG Joint meeting 28 February 2012 Presented by: Ragini Shivji, Monika Benstetter EMA An agency of the European Union Overview of presentation Background & Objective European
Pandemic lessons learnt PCWP-HCPWG Joint meeting 28 February 2012 Presented by: Ragini Shivji, Monika Benstetter EMA An agency of the European Union Overview of presentation Background & Objective European
abcdefghijklmnopqrstu
 abcdefghijklmnopqrstu Swine Flu UK Planning Assumptions Issued 3 September 2009 Planning Assumptions for the current A(H1N1) Influenza Pandemic 3 September 2009 Purpose These planning assumptions relate
abcdefghijklmnopqrstu Swine Flu UK Planning Assumptions Issued 3 September 2009 Planning Assumptions for the current A(H1N1) Influenza Pandemic 3 September 2009 Purpose These planning assumptions relate
Subliminal Programming
 Subliminal Programming Directions for Use Common Questions Background Information Session Overview These sessions are a highly advanced blend of several mind development technologies. Your mind will be
Subliminal Programming Directions for Use Common Questions Background Information Session Overview These sessions are a highly advanced blend of several mind development technologies. Your mind will be
Interviewing, or MI. Bear in mind that this is an introductory training. As
 Motivational Interviewing Module 2 Slide Transcript Slide 1 In this module, you will be introduced to the basics of Motivational Interviewing, or MI. Bear in mind that this is an introductory training.
Motivational Interviewing Module 2 Slide Transcript Slide 1 In this module, you will be introduced to the basics of Motivational Interviewing, or MI. Bear in mind that this is an introductory training.
Industry Perspective: Strategies and costs associated with increasing seasonal influenza vaccine. since 2006
 Industry Perspective: Strategies and costs associated with increasing seasonal influenza vaccine use from high to low income countries since 2006 Dr. Michael Watson, sanofi-pasteur on behalf of WHO GAP
Industry Perspective: Strategies and costs associated with increasing seasonal influenza vaccine use from high to low income countries since 2006 Dr. Michael Watson, sanofi-pasteur on behalf of WHO GAP
Investigating Plinabulin for Prevention of Chemotherapy-Induced Neutropenia
 Transcript Details This is a transcript of an educational program accessible on the ReachMD network. Details about the program and additional media formats for the program are accessible by visiting: https://reachmd.com/programs/project-oncology/origins-and-impacts-severe-neutropenia-newpharmacotherapy/9650/
Transcript Details This is a transcript of an educational program accessible on the ReachMD network. Details about the program and additional media formats for the program are accessible by visiting: https://reachmd.com/programs/project-oncology/origins-and-impacts-severe-neutropenia-newpharmacotherapy/9650/
Flu is a more severe form of what people generally associate with as Cough, Cold and Fever and symptoms are usually incapacitating.
 SEASONAL HUMAN INFLUENZA (THE FLU) What is Seasonal Human Influenza? Seasonal Influenza is a viral infection that affects millions of people worldwide. It is transmitted from person to person through direct
SEASONAL HUMAN INFLUENZA (THE FLU) What is Seasonal Human Influenza? Seasonal Influenza is a viral infection that affects millions of people worldwide. It is transmitted from person to person through direct
A Hopeful Beginning for Malaria Vaccines
 Transcript Details This is a transcript of an educational program accessible on the ReachMD network. Details about the program and additional media formats for the program are accessible by visiting: https://reachmd.com/programs/focus-on-global-medicine/a-hopeful-beginning-for-malariavaccines/4043/
Transcript Details This is a transcript of an educational program accessible on the ReachMD network. Details about the program and additional media formats for the program are accessible by visiting: https://reachmd.com/programs/focus-on-global-medicine/a-hopeful-beginning-for-malariavaccines/4043/
Occupational Infections: Risks for the Anesthesiologist
 Occupational Infections: Risks for the Anesthesiologist 2017 {Music} Dr. Alan Jay Schwartz: Hello. This is Alan Jay Schwartz, Editor-in-Chief of the American Society of Anesthesiologists 2017 Refresher
Occupational Infections: Risks for the Anesthesiologist 2017 {Music} Dr. Alan Jay Schwartz: Hello. This is Alan Jay Schwartz, Editor-in-Chief of the American Society of Anesthesiologists 2017 Refresher
Section 4 Decision-making
 Decision-making : Decision-making Summary Conversations about treatments Participants were asked to describe the conversation that they had with the clinician about treatment at diagnosis. The most common
Decision-making : Decision-making Summary Conversations about treatments Participants were asked to describe the conversation that they had with the clinician about treatment at diagnosis. The most common
Hello and welcome to Patient Power sponsored by UCSF Medical Center. I m Andrew Schorr.
 The Integrated Approach to Treating Cancer Symptoms Webcast March 1, 2012 Michael Rabow, M.D. Please remember the opinions expressed on Patient Power are not necessarily the views of UCSF Medical Center,
The Integrated Approach to Treating Cancer Symptoms Webcast March 1, 2012 Michael Rabow, M.D. Please remember the opinions expressed on Patient Power are not necessarily the views of UCSF Medical Center,
Advances in Viral Immunity Stemming from the 1918 Flu Pandemic
 Transcript Details This is a transcript of an educational program accessible on the ReachMD network. Details about the program and additional media formats for the program are accessible by visiting: https://reachmd.com/programs/clinicians-roundtable/advances-in-viral-immunity-stemming-from-the-
Transcript Details This is a transcript of an educational program accessible on the ReachMD network. Details about the program and additional media formats for the program are accessible by visiting: https://reachmd.com/programs/clinicians-roundtable/advances-in-viral-immunity-stemming-from-the-
The Top 10 Things You Should Know Before Choosing Your
 A d v a n c e d H e a r i n g C e n t e r The Top 10 Things You Should Know Before Choosing Your 516.484.0811 www.ny.com Welcome Letter from the Advanced Hearing Center Team Dear Friend, If you re reading
A d v a n c e d H e a r i n g C e n t e r The Top 10 Things You Should Know Before Choosing Your 516.484.0811 www.ny.com Welcome Letter from the Advanced Hearing Center Team Dear Friend, If you re reading
BDO Dunwoody Weekly CEO/Business Leader Poll By COMPAS in Canadian Business For Publication October 5, 2009
 vs. Avian Flu (2005) and SARS (2003): Both Employers and Provincial Ministries of Health Much Better Prepared; Health Ministries Earn Good Performance Scores on Pandemic Preparations for First Time BDO
vs. Avian Flu (2005) and SARS (2003): Both Employers and Provincial Ministries of Health Much Better Prepared; Health Ministries Earn Good Performance Scores on Pandemic Preparations for First Time BDO
TRANSCRIPT Opening Remarks
 TRANSCRIPT Opening Remarks Amy Gutmann, Ph.D. Commission Chair James Wagner, Ph.D. Commission Vice-Chair Meeting 5, Day 1 Opening Remarks May 18, 2011 New York, New York DR. GUTMANN: Good morning, everybody.
TRANSCRIPT Opening Remarks Amy Gutmann, Ph.D. Commission Chair James Wagner, Ph.D. Commission Vice-Chair Meeting 5, Day 1 Opening Remarks May 18, 2011 New York, New York DR. GUTMANN: Good morning, everybody.
USING ASSERTIVENESS TO COMMUNICATE ABOUT SEX
 Chapter 5: Sexual Health Exercise 1 USING ASSERTIVENESS TO COMMUNICATE ABOUT SEX Aggressive Passive Manipulative/manipulation Assertive Balance of power Sex Sexual coercion 1. To build learners communication
Chapter 5: Sexual Health Exercise 1 USING ASSERTIVENESS TO COMMUNICATE ABOUT SEX Aggressive Passive Manipulative/manipulation Assertive Balance of power Sex Sexual coercion 1. To build learners communication
Situation Update Pandemic (H1N1) August 2009
 Situation Update Pandemic (H1N1) 2009 31 August 2009 Timeline pandemic (H1N1) 2009 April 12: an outbreak of influenza-like illness in Veracruz, Mexico reported to WHO April 15-17: two cases of the new
Situation Update Pandemic (H1N1) 2009 31 August 2009 Timeline pandemic (H1N1) 2009 April 12: an outbreak of influenza-like illness in Veracruz, Mexico reported to WHO April 15-17: two cases of the new
Working Group on H5N1 Influenza Vaccines
 Report of the Strategic Advisory Group of Experts on immunization Working Group on H5N1 Influenza Vaccines 27 September 2010 Introduction The Chair summarized the objectives of the Strategic Advisory Group
Report of the Strategic Advisory Group of Experts on immunization Working Group on H5N1 Influenza Vaccines 27 September 2010 Introduction The Chair summarized the objectives of the Strategic Advisory Group
Acquisition of Novartis Influenza Vaccines Business. 27 th October 2014
 1 Acquisition of Novartis Influenza Vaccines Business 27 th October 2014 2 Legal Notice Forward looking statements The materials in this presentation speak only as of the date of these materials, and include
1 Acquisition of Novartis Influenza Vaccines Business 27 th October 2014 2 Legal Notice Forward looking statements The materials in this presentation speak only as of the date of these materials, and include
Options in HIV Prevention A Participant-Centered Counseling Approach
 Options in HIV Prevention A Participant-Centered Counseling Approach Options Counseling Flipchart, Version 3.0, 10 Oct 2017 Enrollment Visit Welcome and thank you! 3 HOPE Adherence Counseling CHOICE: Helping
Options in HIV Prevention A Participant-Centered Counseling Approach Options Counseling Flipchart, Version 3.0, 10 Oct 2017 Enrollment Visit Welcome and thank you! 3 HOPE Adherence Counseling CHOICE: Helping
Combining Individualized Treatment Options with Patient-Clinician Dialogue
 Transcript Details This is a transcript of a continuing medical education (CME) activity accessible on the ReachMD network. Additional media formats for the activity and full activity details (including
Transcript Details This is a transcript of a continuing medical education (CME) activity accessible on the ReachMD network. Additional media formats for the activity and full activity details (including
Breast Cancer Imaging Webcast October 21, 2009 Peter Eby, M.D. Introduction
 Breast Cancer Imaging Webcast October 21, 2009 Peter Eby, M.D. Please remember the opinions expressed on Patient Power are not necessarily the views of Seattle Cancer Care Alliance, its medical staff or
Breast Cancer Imaging Webcast October 21, 2009 Peter Eby, M.D. Please remember the opinions expressed on Patient Power are not necessarily the views of Seattle Cancer Care Alliance, its medical staff or
WHO GLOBAL ACTION PLAN FOR INFLUENZA VACCINES
 WHO GLOBAL ACTION PLAN FOR INFLUENZA VACCINES SURVEY QUESTIONNAIRE A.1. Introduction The Global Action Plan for Influenza Vaccines (GAP) was launched in 2006 as a ten year initiative to address the anticipated
WHO GLOBAL ACTION PLAN FOR INFLUENZA VACCINES SURVEY QUESTIONNAIRE A.1. Introduction The Global Action Plan for Influenza Vaccines (GAP) was launched in 2006 as a ten year initiative to address the anticipated
Interviews with Volunteers from Immigrant Communities Regarding Volunteering for a City. Process. Insights Learned from Volunteers
 Interviews with Volunteers from Immigrant Communities Regarding Volunteering for a City Cities across Minnesota are taking a new look at involving volunteers to assist the city. One of the opportunities
Interviews with Volunteers from Immigrant Communities Regarding Volunteering for a City Cities across Minnesota are taking a new look at involving volunteers to assist the city. One of the opportunities
Welcome to Progress in Community Health Partnerships latest episode our Beyond the Manuscript podcast. In each
 Beyond the Manuscript 91 Podcast Interview Transcript Erin Kobetz, Joan Bloom, Irma Robbins, Kim Engelman Welcome to Progress in Community Health Partnerships latest episode our Beyond the Manuscript podcast.
Beyond the Manuscript 91 Podcast Interview Transcript Erin Kobetz, Joan Bloom, Irma Robbins, Kim Engelman Welcome to Progress in Community Health Partnerships latest episode our Beyond the Manuscript podcast.
2009 / 2010 H1N1 FAQs
 The information contained within this document was compiled from sources that include the Center for Disease Control and Prevention (CDC), U.S. Department of Health and Human Services, and the Oregon Department
The information contained within this document was compiled from sources that include the Center for Disease Control and Prevention (CDC), U.S. Department of Health and Human Services, and the Oregon Department
Pandemic Influenza Preparedness. Vaccine Sharing. Robert Gatter Center for Health Law Studies
 Pandemic Influenza Preparedness and the Global Stalemate over Vaccine Sharing Robert Gatter Center for Health Law Studies Saint Louis University GISN and Indonesia sprotests WHO member states share flu
Pandemic Influenza Preparedness and the Global Stalemate over Vaccine Sharing Robert Gatter Center for Health Law Studies Saint Louis University GISN and Indonesia sprotests WHO member states share flu
U.S. Readiness for Pandemics
 0 U.S. Readiness for Pandemics 5 th National Emergency Management Summit Brooklyn, NY September 15, 2011 Robin Robinson, Ph.D. Deputy Assistant Secretary for Preparedness & Response (HHS/ASPR) Director
0 U.S. Readiness for Pandemics 5 th National Emergency Management Summit Brooklyn, NY September 15, 2011 Robin Robinson, Ph.D. Deputy Assistant Secretary for Preparedness & Response (HHS/ASPR) Director
Aspirin Resistance and Its Implications in Clinical Practice
 Transcript Details This is a transcript of an educational program accessible on the ReachMD network. Details about the program and additional media formats for the program are accessible by visiting: https://reachmd.com/programs/clinicians-roundtable/aspirin-resistance-and-its-implications-in-clinicalpractice/3819/
Transcript Details This is a transcript of an educational program accessible on the ReachMD network. Details about the program and additional media formats for the program are accessible by visiting: https://reachmd.com/programs/clinicians-roundtable/aspirin-resistance-and-its-implications-in-clinicalpractice/3819/
Sickle Cell Disease: How Should YOU Reassess Management & Treatment?
 Transcript Details This is a transcript of an educational program accessible on the ReachMD network. Details about the program and additional media formats for the program are accessible by visiting: https://reachmd.com/programs/changing-conversation-sickle-cell-disease/sickle-cell-disease-howshould-you-reassess-management-treatment/10184/
Transcript Details This is a transcript of an educational program accessible on the ReachMD network. Details about the program and additional media formats for the program are accessible by visiting: https://reachmd.com/programs/changing-conversation-sickle-cell-disease/sickle-cell-disease-howshould-you-reassess-management-treatment/10184/
Human H5N1 and Pandemic Vaccines Practicalities of Production: A perspective from Industry
 Human H5N1 and Pandemic Vaccines Practicalities of Production: A perspective from Industry Luc Hessel M.D. Pandemic Influenza Working Group European Vaccine Manufacturers 4th Joint EC/ECDC/WHO Workshop
Human H5N1 and Pandemic Vaccines Practicalities of Production: A perspective from Industry Luc Hessel M.D. Pandemic Influenza Working Group European Vaccine Manufacturers 4th Joint EC/ECDC/WHO Workshop
Five Features of Fighting the Flu
 Five Features of Fighting the Flu Public Health Emergency Preparedness Pandemic Influenza Prevention Curriculum Grades 9-12 1 Day One Understand the Flu Virus 2 Five Features of Flu Fighting Code 1: Understand
Five Features of Fighting the Flu Public Health Emergency Preparedness Pandemic Influenza Prevention Curriculum Grades 9-12 1 Day One Understand the Flu Virus 2 Five Features of Flu Fighting Code 1: Understand
PANDEMIC INFLUENZA PREPAREDNESS: STATE CHALLENGES
 PANDEMIC INFLUENZA PREPAREDNESS: STATE CHALLENGES A Presentation to the Council of State Governments Health Policy Forum by Jeffrey Levi, Ph.D., Executive Director Trust for America s Health June 23, 2006
PANDEMIC INFLUENZA PREPAREDNESS: STATE CHALLENGES A Presentation to the Council of State Governments Health Policy Forum by Jeffrey Levi, Ph.D., Executive Director Trust for America s Health June 23, 2006
20. The purpose of this document is to examine the pre-pandemic efforts and the response to the new influenza A (H1N1) virus since April 2009.
 B. UPDATE ON THE PANDEMIC (H1N1) 2009 CD50/INF/6 (Eng.) - 7 - Annex B Background 20. The purpose of this document is to examine the pre-pandemic efforts and the response to the new influenza A (H1N1) virus
B. UPDATE ON THE PANDEMIC (H1N1) 2009 CD50/INF/6 (Eng.) - 7 - Annex B Background 20. The purpose of this document is to examine the pre-pandemic efforts and the response to the new influenza A (H1N1) virus
Importance and Benefits of Being Prepared
 Importance and Benefits of Being Prepared It is difficult to predict when the next influenza pandemic will occur or how severe it will be. The effects of a pandemic can be lessened if preparations are
Importance and Benefits of Being Prepared It is difficult to predict when the next influenza pandemic will occur or how severe it will be. The effects of a pandemic can be lessened if preparations are
FREQUENTLY ASKED QUESTIONS SWINE FLU
 FREQUENTLY ASKED QUESTIONS SWINE FLU Updated 5/6/09 ER FAQ What is swine flu? Swine flu is common disease of pigs and is caused by the same category of influenza virus (influenza A) that causes flu in
FREQUENTLY ASKED QUESTIONS SWINE FLU Updated 5/6/09 ER FAQ What is swine flu? Swine flu is common disease of pigs and is caused by the same category of influenza virus (influenza A) that causes flu in
Communicating in Changing and Difficult Communication Environments: Some Things I ve Learned Regarding Influenza Immunization
 Communicating in Changing and Difficult Communication Environments: Some Things I ve Learned Regarding Influenza Immunization Glen Nowak, Ph.D. Acting Director of Media Relations Centers for Disease Control
Communicating in Changing and Difficult Communication Environments: Some Things I ve Learned Regarding Influenza Immunization Glen Nowak, Ph.D. Acting Director of Media Relations Centers for Disease Control
Novel H1N1 Influenza. It s the flu after all! William Muth M.D. Samaritan Health Services 9 November 2009
 Novel H1N1 Influenza It s the flu after all! William Muth M.D. Samaritan Health Services 9 November 2009 Influenza A Primer.. What is the flu? How do you get it? What s a virus anyhow? Can the flu be prevented,
Novel H1N1 Influenza It s the flu after all! William Muth M.D. Samaritan Health Services 9 November 2009 Influenza A Primer.. What is the flu? How do you get it? What s a virus anyhow? Can the flu be prevented,
The Expanding Value of Biomarkers in NSCLC Treatment
 Transcript Details This is a transcript of an educational program accessible on the ReachMD network. Details about the program and additional media formats for the program are accessible by visiting: https://reachmd.com/programs/closing-gaps-nsclc/the-expanding-value-of-biomarkers-in-nsclctreatment/10283/
Transcript Details This is a transcript of an educational program accessible on the ReachMD network. Details about the program and additional media formats for the program are accessible by visiting: https://reachmd.com/programs/closing-gaps-nsclc/the-expanding-value-of-biomarkers-in-nsclctreatment/10283/
Flex case study. Pádraig MacGinty Owner, North West Hearing Clinic Donegal, Ireland
 Flex case study Pádraig MacGinty Owner, North West Hearing Clinic Donegal, Ireland Pádraig MacGinty has been in business for 15 years, owning two clinics in North West Ireland. His experience with Flex:trial
Flex case study Pádraig MacGinty Owner, North West Hearing Clinic Donegal, Ireland Pádraig MacGinty has been in business for 15 years, owning two clinics in North West Ireland. His experience with Flex:trial
B - Planning for a Turkey Flu Pandemic
 RA 2014-15 Pandemic risk: B - Planning for a Turkey Flu Pandemic 8 B - Planning for a Turkey Flu Pandemic You are employed by the government OR service and your team has been asked to provide OR support
RA 2014-15 Pandemic risk: B - Planning for a Turkey Flu Pandemic 8 B - Planning for a Turkey Flu Pandemic You are employed by the government OR service and your team has been asked to provide OR support
Sample Observation Form
 Sample Observation Form (NOTE: This is just an example. You will need to modify it based on the type of exercise. Also, you might want to break up the checklist items by observer. For example, you could
Sample Observation Form (NOTE: This is just an example. You will need to modify it based on the type of exercise. Also, you might want to break up the checklist items by observer. For example, you could
Minnesota Cancer Alliance SUMMARY OF MEMBER INTERVIEWS REGARDING EVALUATION
 Minnesota Cancer Alliance SUMMARY OF MEMBER INTERVIEWS REGARDING EVALUATION Minnesota Cancer Alliance Comprehensive Cancer Control Program P.O. Box 64882 St. Paul, MN 55164 651-201-3661 lisa.gemlo@state.mn.us
Minnesota Cancer Alliance SUMMARY OF MEMBER INTERVIEWS REGARDING EVALUATION Minnesota Cancer Alliance Comprehensive Cancer Control Program P.O. Box 64882 St. Paul, MN 55164 651-201-3661 lisa.gemlo@state.mn.us
Attitude toward Fundraising - Positive Attitude toward fundraising refers to how fundraising is valued and integrated within an organization
 Attitude toward Fundraising - Positive Attitude toward fundraising refers to how fundraising is valued and integrated within an organization We believe fundraising is an opportunity to talk personally
Attitude toward Fundraising - Positive Attitude toward fundraising refers to how fundraising is valued and integrated within an organization We believe fundraising is an opportunity to talk personally
Congressional Record December 9, 2009 S12786-S12787
 Congressional Record December 9, 2009 S12786-S12787 Mr. MENENDEZ. I thank the distinguished chairman of the Senate Finance Committee for yielding me the time. Madam President, I rise in strong opposition
Congressional Record December 9, 2009 S12786-S12787 Mr. MENENDEZ. I thank the distinguished chairman of the Senate Finance Committee for yielding me the time. Madam President, I rise in strong opposition
Module 4: Emergencies: Prevention, Preparedness, Response and Recovery
 Module 4: Emergencies: Prevention, Preparedness, Response and Recovery Part 1: Introduction and Prevention Tamar: Hello, my name is Tamar Klaiman. I'm an Assistant Professor at the University of the Sciences
Module 4: Emergencies: Prevention, Preparedness, Response and Recovery Part 1: Introduction and Prevention Tamar: Hello, my name is Tamar Klaiman. I'm an Assistant Professor at the University of the Sciences
SANOFI PASTEUR, LEADING PROVIDER OF SEASONAL INFLUENZA VACCINES
 FACT SHEET SANOFI PASTEUR, LEADING PROVIDER OF SEASONAL INFLUENZA VACCINES WORLD S LEADING INFLUENZA VACCINE MANUFACTURER Annual production capacity of Sanofi Pasteur seasonal influenza vaccines have doubled
FACT SHEET SANOFI PASTEUR, LEADING PROVIDER OF SEASONAL INFLUENZA VACCINES WORLD S LEADING INFLUENZA VACCINE MANUFACTURER Annual production capacity of Sanofi Pasteur seasonal influenza vaccines have doubled
Unraveling Recent Cervical Cancer Screening Updates and the Impact on Your Practice
 Transcript Details This is a transcript of a continuing medical education (CME) activity accessible on the ReachMD network. Additional media formats for the activity and full activity details (including
Transcript Details This is a transcript of a continuing medical education (CME) activity accessible on the ReachMD network. Additional media formats for the activity and full activity details (including
What Antivirals Can Be Used for 2009 H1N1 Influenza?
 www.upmc-biosecurity.org www.upmc-cbn.org May 13, 2009 The Use of Antivirals for 2009 H1N1 Influenza Virus Infection By Ann Norwood, MD, Brooke Courtney, JD, MPH, Eric Toner, MD, and Amesh Adalja, MD Use
www.upmc-biosecurity.org www.upmc-cbn.org May 13, 2009 The Use of Antivirals for 2009 H1N1 Influenza Virus Infection By Ann Norwood, MD, Brooke Courtney, JD, MPH, Eric Toner, MD, and Amesh Adalja, MD Use
How you can help support the Beat Flu campaign
 How you can help support the Beat Flu campaign Toolkit www.beatflu.org f T Beat Flu 1 How can you help Beat Flu? There are a number of ways your organisation can get involved in raising awareness of the
How you can help support the Beat Flu campaign Toolkit www.beatflu.org f T Beat Flu 1 How can you help Beat Flu? There are a number of ways your organisation can get involved in raising awareness of the
ENGLESKI JEZIK. 02. Every time Jim came to see Jill, her some flowers. a) he d bring b) he d brought c) he ll bring
 12.00 h I GRUPA ENGLESKI JEZIK 01. I ll inform you as soon as we the results. a) will be hearing b) shall hear c) have heard 02. Every time Jim came to see Jill, her some flowers. a) he d bring b) he d
12.00 h I GRUPA ENGLESKI JEZIK 01. I ll inform you as soon as we the results. a) will be hearing b) shall hear c) have heard 02. Every time Jim came to see Jill, her some flowers. a) he d bring b) he d
Making Your Treatment Work Long-Term
 Making Your Treatment Work Long-Term How to keep your treatment working... and why you don t want it to fail Regardless of the particular drugs you re taking, your drugs will only work when you take them.
Making Your Treatment Work Long-Term How to keep your treatment working... and why you don t want it to fail Regardless of the particular drugs you re taking, your drugs will only work when you take them.
Living My Best Life. Today, after more than 30 years of struggling just to survive, Lynn is in a very different space.
 Living My Best Life Lynn Allen-Johnson s world turned upside down when she was 16. That s when her father and best friend died of Hodgkin s disease leaving behind her mom and six kids. Lynn s family was
Living My Best Life Lynn Allen-Johnson s world turned upside down when she was 16. That s when her father and best friend died of Hodgkin s disease leaving behind her mom and six kids. Lynn s family was
