Evaluating the acceptability of vaccine and vaccination programmes: an individual and public health perspective
|
|
|
- Gerard Ray
- 6 years ago
- Views:
Transcription
1 The regulatory process Evaluating the acceptability of vaccine and vaccination programmes: an individual and public health perspective Annecy, Les Pensières July 7-9, 2008 Dr. Nora Dellepiane, Scientist WHO/IVB/QSS July 2008
2 Outline Vaccines of assured quality Recommended regulatory functions and source of vaccines. WHO support to strengthen regulatory functions New vaccines: New challenges WHO innovative approaches to support strengthening of regulatory capacity
3 Vaccines of assured quality
4 Contribution to World Health Organization Goal Ensure that 100% of vaccines used in all national immunization programmes are of assured quality Definition iti of Assured d quality vaccines National Regulatory Authority (NRA) independently controls the quality of vaccines in accordance with the six specified functions defined by WHO No unresolved confirmed reports of quality related problems Guided by Expert Committee on Standardization of Biologicals i l (ECBS) recommendations on safety, efficacy and quality issued in WHO Technical Report Series (TRS)
5 Recommended regulatory functions and source of vaccines. WHO support to strengthen regulatory functions
6 Regulatory functions depending on vaccine source Regulatory functions Regulation System Licensing AEFI monitoring UN agency Vaccine source Direct Procurement Lot release Functions Access to laboratory assured by NRA of Regulatory inspections producing Fuctions assured country and by NRA of Authorization of producing WHO PQ system clinical trials country Production
7 Regulatory guidance WHO norms and standards Global written standards Global measurement standards Support for the evidence base for standards
8 WHO Global written standards Technical specifications that help define safe and efficacious vaccines. These are intended to be scientific and advisory in nature Guidance for national regulatory authorities and manufacturers on international regulatory expectations for the production and quality control of vaccines, non-clinical and clinical evaluation of vaccines Facilitate international harmonisation of vaccine licensure Living documents revised in response to scientific advances
9 Use of WHO guidance and standards The WHO Recommendations on production and quality control of vaccines are used as: -thebasis of national regulations in many countries - the technical specifications against which compliance is assessed for the purposes of pre-qualification of vaccine supply by UN agencies
10 Process to strengthen NRAs The five step capacity building programme 1)Benchmarking 2)NRA assessment 3)Planning to address gaps (IDP) Planning to address gaps NRA Network of regulatory experts 4)Implementation pe e tato of pa plan, including training courses (GTN), technical inputs, in country workshops 5)Monitoring and evaluation NRA Training i i Assessment Institutional needs using joint development assessment plan p to to tools address gaps Technical (Drug (Drug& vaccine) support 5 days assessment GTN placement within 1-3 months Follow up up visits months (6-8 months in needs much improvement)
11 Country Status: country assessment of vaccine regulatory system conducted, September 1997 NRA assessments conducted & planned NRA assessments completed Not yet conducted T
12 Country Status: 86 country assessment of vaccine regulatory system conducted, Sept.1997 Dec 2007 NRA assessments conducted & planned NRA assessments completed Not yet conducted New assessments to be conducted in 2008
13 New vaccines: New challenges
14 The Vaccine Pipeline & PQ vaccines HIV/AIDS Future Malaria TB Underutilized Vaccines Cholera Typhoid Hib (conj) Dengue Mening (conj) HPV Rotavirus Pneumo (conj) YF Influenza JE Rubella HepB Measles Traditional EPI Tetanus Polio Pertussis Diphtheria // // Vaccine Development Pipeline
15 Key messages The vaccine development pipeline is especially buoyant; some complex products are under development; specialist regulatory oversight is needed WHO is committed to support countries to ensure and sustain that 100% of vaccines used in all national immunization programmes are of assured quality NRA strengthening activities and other innovative approaches are being applied to attain this goal
16 New challenges Regulation of new vaccines : Responsibility now falls more on Developing Countries using these vaccines and less on Industrialized Countries where they are produced Countries have insufficient i expertise and experience to assess data and dossiers (Manufacturing, preclinical and clinical data, etc.) NRAs must acquire new skills Clinical trials for new vaccines Are being run in ANY country, no matter the expertise/strength t th of their National Regulatory Authority Quality of the trials must be guaranteed
17 Responding to challenges 1. Development of new support mechanisms for regulatory authorities 2. Development of new regulatory pathways
18 New Support Mechanisms for National Regulatory Authorities Establishment of Developing Countries Vaccines Regulators Network (DCVRN) in 2004 Establishment of African Vaccine Regulatory Forum (AVAREF) in 2006 Development of new Global Training Network Courses and country workshops on a needs basis Sentinel Network to monitor safety during introduction of novel vaccines and DTP based combinations
19 New Regulatory Support Mechanisms 1. Establishment of the Developing Countries Vaccine Regulators Network (Global) Brazil, Cuba, China, India, Indonesia Korea, Russia, South Africa and Thailand
20 Objectives 1. Developing Countries Vaccine Regulators Network Promote and support the strengthening of the regulatory capacity of NRAs for the evaluation of clinical i l trial proposals and clinical i l trial data through expertise and exchange of relevant information. Modus Operandi Meetings twice a year Discuss critical aspects to be considered for the review of clinical data for registration of novel vaccines Develop procedures, forms and other relevant documents to harmonize regulation of clinical trials and evaluation of clinical trial data Exchange of information and mutual support Provide support to other countries in their region of influence (i.e. SA and Indonesia
21 1. African Vaccine Regulators Forum (AVAREF) established in 2006 Objectives To provide information to regulators of countries that are target for clinical trials To promote communication/collaboration between NRAs and Ethics committees and among regulators in the Region and others To provide a resource of expert advise to regulators To identify needs for expert support to NRAs Modus Operandi One plenary meeting per year (2 to date) plus support activities between meetings; joint WHO/HQ/AFRO activity 19 countries involved; NRA and National Ethics Committee representatives ti
22 AVAREF achievements An historical moment : For the 1st time in Africa, regulators and ethics committee members from Burkina Faso, The Gambia, Ghana, Ethiopia and Mali conducted an inspection of Good Clinical Practice (GCP) inspection of phase II observer- blind, randomized, active controlled clinical trial of meningococcal a conjugate vaccine at the Centre for Vaccine Development (CVD), Bamako, Mali; January, 2007
23 Training Planning workshops for NRA strengthening g Twenty six countries The Gambia, Ghana, Nigeria, Ethiopia, Uganda and Kenya (Addis January 2005) Senegal, Mali, Niger, Benin, Togo, RCA, Cameroon, Burkina Faso and Guinea (Ouagadougou May 2005) Angola, Botswana, DRC, Malawi, Mozambique, Namibia, Rwanda, South Africa, Tanzania, Zambia and Zimbabwe (Gaborone Dec 2005)
24 Training Workshops and courses Evaluation of clinical trials, Pretoria, March 2005 Regulatory Procedures for Evaluation of Vaccines (Addis Sept. 2005) Regulatory forum on clinical evaluation of rotavirus vaccines (Botswana Dec. 2005) Joint review of CTA of Conjugate Meningitis A vaccine (Banjul Jun 2006) Vaccine Regulation (in French), M bour, Senegal, March 2006 Surveillance of AEFIs (in French) in Senegal (2006), Tunis (2007) and (in English) in Cape Town (RSA), 2006 Authorization & Inspection of Clinical Trials in French in Ouidah in December 2006, and in English in Harare in July 2007 Good Clinical Practices in English in Harare in July 2007 Joint Inspection of Men. A Clinical Trials, Bamako, Mali, Jan. 2007** African trainees sent in 2006 to GTN courses on lot relase in Lyon, France, and on GMP in Seoul, Korea
25 New GTN courses GMP inspections. Training Center: KFDA, Korea Testing of Hib conjugate vaccines. Training Center: NVI, The Netherlands Authorization of Clinical Trials (Itinerant) GCP inspection course (Itinerant) Evaluation of Clinical data (Updated 2008) (Itinerant) Lot Release (Updated 2007). Training Centers: AFSSAPS (in French) CDL Kasauli (in English) Product Evaluation (under development)
26 2. New Regulatory Pathways Collaboration with EMEA for the establishment of Scientific Opinion procedure (Art. 58) Procedure for expedited review of imported prequalified vaccines for use in immunization programmes Considering i feasibility of establishing mutual support mechanism between NRAs in Asia for the regulation of JE vaccines
27 Procedure for expedited review of PQd vaccines It is intended for countries that source their vaccines through UN agencies, or who use the WHO prequalification as a basis for selection of vaccines for use in their national immunization programmes, importing them through direct procurement. It provides guidance on how NRAs of such countries can expedite the regulatory review for such products. Not intended to affect in any way post-approval activities in place in these countries Countries intending to use the procedure should ensure that their national regulations contain provisions to allow to shorten the normal regulatory approval process.
28 Criteria for use Scenario 1 Scenario 2 For an expedited approval vaccines WHO-prequalified that are sourced through UN procurement agency. For an expedited approval of WHO-prequalified vaccines that are procured directly.
29 Requirements for licensing vaccines from PQ sources Scenario 1 Scenario 2 1. Check prequalification status 1. Same as in scenario 1 2. Submit product samples, product 2. Same as in scenario 1 inserts, NRA lot release certificates 3. Same as in scenario 1 from the country of origin, a list of 4. Same as in scenario 1in addition to countries where the product is licensed and marketed, and summary lot protocols of three final lots. 3. Visual inspection on samples 4. Review protocols (check specifications), labels, boxes and inserts against WHO model. Ensure presence of VVM 5. Prepare report of compliance (noncompliance) 6. If compliant, issue Certificate of Approval 7. Inform manufacturer and WHO 8. If novel vaccine with limited clinical data, review of clinical data may be needed ensure consistency with national tender specifications if different 5. Same as in scenario 1 6. Same as in scenario 1 7. Same as in scenario 1 8. Same as in scenario 1
30 Outcome and timeframes for expedited review procedure Scenario 1 Scenario 2 1. Waiver of fees from countries is requested 2. Total timeframe for evaluation should not exceed 30 days unless clinical i l data need to be reviewed, in which case timeframe is extended to 120 days 3. If info submitted by manufacturer is not complete, clock is halted awaiting. Completion. 4. Inform WHO that the procedure is being adopted. WHO will keep NRA informed of updates regarding PQ status. 5. Procedure applies to vaccines used in NIP 1. Same as in scenario 1 2. Same as in scenario 1. If country has testing capabilities and vaccine samples will be tested as part of the registration process, 90 days instead of 30 will be the timeframe for completion of procedure, except when clinical data need to be reviewed (120 days) 3. Same as in scenario 1 4. Same as in scenario 1; 5. Same as in scenario 1 6. Same as in scenario 1 6. Countries should not stop use of PQ vaccines not yet registered in the country
31
Ensuring quality, safety and efficacy of vaccines
 EDCTP Third Annual Forum 2006 Partnership and African Leadership: Challenges & Opportunities Ensuring quality, safety and efficacy of vaccines Vaccine regulatory issues in African countries Lahouari Belgharbi,
EDCTP Third Annual Forum 2006 Partnership and African Leadership: Challenges & Opportunities Ensuring quality, safety and efficacy of vaccines Vaccine regulatory issues in African countries Lahouari Belgharbi,
Procedure for Expedited Review of imported pre-qualified vaccines for use in national immunization programmes
 Procedure for Expedited Review of imported pre-qualified vaccines for use in national immunization programmes Dr Nora Dellepiane/Dr Anil Kumar Chawla WHO/HQ-Geneva, Switzerland 1 Expedited review procedure
Procedure for Expedited Review of imported pre-qualified vaccines for use in national immunization programmes Dr Nora Dellepiane/Dr Anil Kumar Chawla WHO/HQ-Geneva, Switzerland 1 Expedited review procedure
Procedure for the expedited review of imported prequalified vaccines with view to granting a marketing authorization
 Procedure for the expedited review of imported prequalified vaccines with view to granting a marketing authorization Prequalification Stakeholders Meeting Geneva, CICG 4 and 5 April 2011 Dr. Nora Dellepiane
Procedure for the expedited review of imported prequalified vaccines with view to granting a marketing authorization Prequalification Stakeholders Meeting Geneva, CICG 4 and 5 April 2011 Dr. Nora Dellepiane
Quality assurance for essential medicines and health products: moving towards an harmonized approach
 Quality assurance for essential medicines and health products: moving towards an harmonized approach Prequalification of vaccines David Wood QSS/EMP/WHO 12 December 2012 A growing portion of vaccines procured
Quality assurance for essential medicines and health products: moving towards an harmonized approach Prequalification of vaccines David Wood QSS/EMP/WHO 12 December 2012 A growing portion of vaccines procured
Regulatory Capacity Building and Developing Countries: CBER Perspective
 Regulatory Capacity Building and Developing Countries: CBER Perspective Karen Midthun, M.D. Director Center for Biologics Evaluation and Research US Food and Drug Administration IOM Meeting Strengthening
Regulatory Capacity Building and Developing Countries: CBER Perspective Karen Midthun, M.D. Director Center for Biologics Evaluation and Research US Food and Drug Administration IOM Meeting Strengthening
Collaborative procedure for licensing Prequalified vaccines
 Collaborative procedure for licensing Prequalified vaccines DCVMN meeting Shanghai, China March 2015 World Health Organization, EMP/RHT/PQT rodriguezhernandezc@who.int meekd@who.int Strategic priorities
Collaborative procedure for licensing Prequalified vaccines DCVMN meeting Shanghai, China March 2015 World Health Organization, EMP/RHT/PQT rodriguezhernandezc@who.int meekd@who.int Strategic priorities
Vaccine assessment for prequalification
 Vaccine assessment for prequalification Briefing on Vaccine Prequalification for manufacturers DCVMN meeting, Kunming, China, 13 March 2016 Olivier Lapujade World Health Organization, EMP/RHT/PQT lapujadeo@who.int
Vaccine assessment for prequalification Briefing on Vaccine Prequalification for manufacturers DCVMN meeting, Kunming, China, 13 March 2016 Olivier Lapujade World Health Organization, EMP/RHT/PQT lapujadeo@who.int
Programme update. Prequalification of Vaccines
 Programme update Prequalification of Vaccines UN Prequalification of Medicines, Diagnostics and Vaccines 6th Consultative Stakeholder Meeting 4 April 2011 David Wood, WHO/IVB/QSS Outline Good news - A
Programme update Prequalification of Vaccines UN Prequalification of Medicines, Diagnostics and Vaccines 6th Consultative Stakeholder Meeting 4 April 2011 David Wood, WHO/IVB/QSS Outline Good news - A
From development to delivery: Decision-making for the introduction of a new vaccine
 From development to delivery: Decision-making for the introduction of a new vaccine Prince Mahidol Award Conference Bangkok, Thailand 1-2 February 2007 Dr. J.M. Okwo-Bele Department of Immunization, Vaccines
From development to delivery: Decision-making for the introduction of a new vaccine Prince Mahidol Award Conference Bangkok, Thailand 1-2 February 2007 Dr. J.M. Okwo-Bele Department of Immunization, Vaccines
Overall presentation of IVR Strategy
 Annex 3 Overall presentation of IVR Strategy Presentation to the IVR Advisory committee (IVAC) April 2007 Outline Why does WHO invest in Research & Development? What is the Initiative for Vaccine Research
Annex 3 Overall presentation of IVR Strategy Presentation to the IVR Advisory committee (IVAC) April 2007 Outline Why does WHO invest in Research & Development? What is the Initiative for Vaccine Research
Vaccine Development in the Developing World; past, present and future: SEAR Perspective
 Speech by Dr Samlee Plianbangchang Regional Director, WHO South-East Asia Vaccine Development in the Developing World; past, present and future: SEAR Perspective 15 June 2010 Bangkok, Thailand Vaccine
Speech by Dr Samlee Plianbangchang Regional Director, WHO South-East Asia Vaccine Development in the Developing World; past, present and future: SEAR Perspective 15 June 2010 Bangkok, Thailand Vaccine
Update on Meningococcal A Vaccine Development and Introduction
 Update on Meningococcal A Vaccine Development and Introduction WHO Product Development for Vaccines Advisory Committee Meeting (PD-VAC) @ Geneva, 7-9 September 2015 Dr Marie-Pierre Preziosi, WHO Initiative
Update on Meningococcal A Vaccine Development and Introduction WHO Product Development for Vaccines Advisory Committee Meeting (PD-VAC) @ Geneva, 7-9 September 2015 Dr Marie-Pierre Preziosi, WHO Initiative
WHO/PQT Updates on WHO Prequalification Priorities and Regulatory Systems Strengthening work
 WHO/PQT Updates on WHO Prequalification Priorities and Regulatory Systems Strengthening work Dr Drew Meek Prequalification Team Regulation of Medicines and other Health Technologies Essential Medicines
WHO/PQT Updates on WHO Prequalification Priorities and Regulatory Systems Strengthening work Dr Drew Meek Prequalification Team Regulation of Medicines and other Health Technologies Essential Medicines
IMMUNIZATION VACCINE DEVELOPMENT
 IMMUNIZATION VACCINE DEVELOPMENT MONTHLY IMMUNIZATION UPDATE IN THE AFRICAN REGION July-August 2016 (Vol 4, issue N 5) Special issue on 2015 WHO/UNICEF Estimates of National Immunization Coverage (WUENIC)
IMMUNIZATION VACCINE DEVELOPMENT MONTHLY IMMUNIZATION UPDATE IN THE AFRICAN REGION July-August 2016 (Vol 4, issue N 5) Special issue on 2015 WHO/UNICEF Estimates of National Immunization Coverage (WUENIC)
Update from GAVI Aurelia Nguyen
 Update from GAVI Aurelia Nguyen (Copenhagen, Denmark, 27 June 2012) GAVI vaccine support Currently supported vaccines: pentavalent, pneumococcal, rotavirus, meningitis A, human papillomavirus (HPV), rubella,
Update from GAVI Aurelia Nguyen (Copenhagen, Denmark, 27 June 2012) GAVI vaccine support Currently supported vaccines: pentavalent, pneumococcal, rotavirus, meningitis A, human papillomavirus (HPV), rubella,
Vaccine Production and Regulation
 Vaccine Production and Regulation Dr Houda LANGAR Essential Vaccines and Biologicals Policy Department of Health Systems & Services Development Capacity Development Workshop on Health System Development
Vaccine Production and Regulation Dr Houda LANGAR Essential Vaccines and Biologicals Policy Department of Health Systems & Services Development Capacity Development Workshop on Health System Development
TT Procured by UNICEF
 TT TT Procured by UNICEF 2001-08 250,000,000 200,000,000 150,000,000 100,000,000 50,000,000 0 2001 2002 2003 2004 2005 2006 2007 2008 Routine SIA TT historical demand and forecast overview Upcoming Tender
TT TT Procured by UNICEF 2001-08 250,000,000 200,000,000 150,000,000 100,000,000 50,000,000 0 2001 2002 2003 2004 2005 2006 2007 2008 Routine SIA TT historical demand and forecast overview Upcoming Tender
IMMUNIZATION VACCINES & EMERGENCIES
 No report No data No data IMMUNIZATION VACCINES & EMERGENCIES ROUTINE IMMUNIZATION PERFORMANCE IN THE AFRICAN REGION October 2013 issue Number of children vaccinated with the 3 rd dose of DTPcontaining
No report No data No data IMMUNIZATION VACCINES & EMERGENCIES ROUTINE IMMUNIZATION PERFORMANCE IN THE AFRICAN REGION October 2013 issue Number of children vaccinated with the 3 rd dose of DTPcontaining
Update on Implementation of NUV. Carsten Mantel WHO/FCH/IVB/EPI
 Update on Implementation of NUV Carsten Mantel WHO/FCH/IVB/EPI Introduction of New Vaccines The continuum of activity REGULATORY NORMS & STANDARDS VACCINE SUPPLY PROCUREMENT PRICING FINANCING SURVEILLANCE
Update on Implementation of NUV Carsten Mantel WHO/FCH/IVB/EPI Introduction of New Vaccines The continuum of activity REGULATORY NORMS & STANDARDS VACCINE SUPPLY PROCUREMENT PRICING FINANCING SURVEILLANCE
History, implementation and impact of MenA conjugate on disease burden in Africa
 History, implementation and impact of MenA conjugate on disease burden in Africa First Regional Meningococcal Symposium, 19-20 March 2012 Buenos Aires, Argentina Co-hosted by the Sabin Vaccine Institute
History, implementation and impact of MenA conjugate on disease burden in Africa First Regional Meningococcal Symposium, 19-20 March 2012 Buenos Aires, Argentina Co-hosted by the Sabin Vaccine Institute
GAVI ALLIANCE: UPDATE AND FUTURE DIRECTIONS FOR GLOBAL VACCINES AND IMMUNISATIONS
 GAVI ALLIANCE: UPDATE AND FUTURE DIRECTIONS FOR GLOBAL VACCINES AND IMMUNISATIONS Ranjana Kumar International Rotavirus Symposium Istanbul, 3 4 June 2008 The GAVI Alliance Public-private partnership bringing
GAVI ALLIANCE: UPDATE AND FUTURE DIRECTIONS FOR GLOBAL VACCINES AND IMMUNISATIONS Ranjana Kumar International Rotavirus Symposium Istanbul, 3 4 June 2008 The GAVI Alliance Public-private partnership bringing
Quality Assurance Policy. for the. Procurement of HIV Point-of-Care Technology. under the UNITAID Grant
 Quality Assurance Policy for the Procurement of HIV Point-of-Care Technology under the UNITAID Grant UNICEF Supply Division Quality Assurance Centre and Health Technology Centre Revisions Version Date
Quality Assurance Policy for the Procurement of HIV Point-of-Care Technology under the UNITAID Grant UNICEF Supply Division Quality Assurance Centre and Health Technology Centre Revisions Version Date
The MenAfrivac Experience A Successful Approach. Dr Bernard FRITZELL BFL conseils France
 The MenAfrivac Experience A Successful Approach Dr Bernard FRITZELL BFL conseils France 1 2 1996 Ministers of Health and Interior from 16 African countries recognized epidemic meningitis as a high priority
The MenAfrivac Experience A Successful Approach Dr Bernard FRITZELL BFL conseils France 1 2 1996 Ministers of Health and Interior from 16 African countries recognized epidemic meningitis as a high priority
IMMUNIZATION VACCINE DEVELOPMENT
 IMMUNIZATION VACCINE DEVELOPMENT MONTHLY IMMUNIZATION AND POLIO UPDATE IN THE AFRICAN REGION January February 2017 (Vol 5 issue N 1) District data completeness and coverage of DTP3 containing vaccine per
IMMUNIZATION VACCINE DEVELOPMENT MONTHLY IMMUNIZATION AND POLIO UPDATE IN THE AFRICAN REGION January February 2017 (Vol 5 issue N 1) District data completeness and coverage of DTP3 containing vaccine per
IMMUNIZATION VACCINE DEVELOPMENT
 IMMUNIZATION VACCINE DEVELOPMENT MONTHLY IMMUNIZATION UPDATE IN THE AFRICAN REGION November December 2016 (Vol 4, issue N 7) District data completeness and coverage of DTP3 containing vaccine per country
IMMUNIZATION VACCINE DEVELOPMENT MONTHLY IMMUNIZATION UPDATE IN THE AFRICAN REGION November December 2016 (Vol 4, issue N 7) District data completeness and coverage of DTP3 containing vaccine per country
Aboubacar Kampo Chief of Health UNICEF Nigeria
 Aboubacar Kampo Chief of Health UNICEF Nigeria Many thanks to UNICEF colleagues in Supply Division-Copenhagen and NY for contributing to this presentation Thirty-five countries are responsible for 98%
Aboubacar Kampo Chief of Health UNICEF Nigeria Many thanks to UNICEF colleagues in Supply Division-Copenhagen and NY for contributing to this presentation Thirty-five countries are responsible for 98%
Update from the GAVI Alliance Seth Berkley, MD Chief Executive Officer
 Update from the GAVI Alliance Seth Berkley, MD Chief Executive Officer SAGE meeting Geneva, 8-10 November 2011 Topics to cover Update on new vaccine introduction Introductions New approvals Co-financing
Update from the GAVI Alliance Seth Berkley, MD Chief Executive Officer SAGE meeting Geneva, 8-10 November 2011 Topics to cover Update on new vaccine introduction Introductions New approvals Co-financing
REGIONAL ALLIANCE FOR NATIONAL REGULATORY AUTHORITIES FOR VACCINES IN THE WESTERN PACIFIC. second edition
 REGIONAL ALLIANCE FOR NATIONAL REGULATORY AUTHORITIES FOR VACCINES IN THE WESTERN PACIFIC second edition World Health Organization 2014 The designations employed and the presentation of the material in
REGIONAL ALLIANCE FOR NATIONAL REGULATORY AUTHORITIES FOR VACCINES IN THE WESTERN PACIFIC second edition World Health Organization 2014 The designations employed and the presentation of the material in
Global Update of the 2010 GIVS Goals
 Global Update of the 2010 GIVS Goals Global Immunization Meeting New York, 17-19 Feb 2009 JM Okwo-Bele, WHO P. Salama, UNICEF Projected Changes in Under-5 yr mortality due to VPDs M illio n s 3.0 2.5 2.0
Global Update of the 2010 GIVS Goals Global Immunization Meeting New York, 17-19 Feb 2009 JM Okwo-Bele, WHO P. Salama, UNICEF Projected Changes in Under-5 yr mortality due to VPDs M illio n s 3.0 2.5 2.0
The Financial Sustainability of New Vaccine Introduction in the Poorest Countries :
 The Financial Sustainability of New Vaccine Introduction in the Poorest Countries : Evidence from the First Phase of GAVI (Global Alliance for Vaccines and Immunization) Patrick Lydon lydonp@who.int Session:
The Financial Sustainability of New Vaccine Introduction in the Poorest Countries : Evidence from the First Phase of GAVI (Global Alliance for Vaccines and Immunization) Patrick Lydon lydonp@who.int Session:
POLIOMYELITIS ERADICATION: PROGRESS REPORT. Information Document CONTENTS BACKGROUND PROGRESS MADE NEXT STEPS... 12
 5 August 9 REGIONAL COMMITTEE FOR AFRICA ORIGINAL: ENGLISH Fifty-ninth session Kigali, Republic of Rwanda, August 4 September 9 POLIOMYELITIS ERADICATION: PROGRESS REPORT Information Document CONTENTS
5 August 9 REGIONAL COMMITTEE FOR AFRICA ORIGINAL: ENGLISH Fifty-ninth session Kigali, Republic of Rwanda, August 4 September 9 POLIOMYELITIS ERADICATION: PROGRESS REPORT Information Document CONTENTS
CONTENTS. Paragraphs I. BACKGROUND II. PROGRESS REPORT ON THE AFRICAN REGIONAL IMMUNIZATION STRATEGIC PLAN
 23 September 2013 REGIONAL COMMITTEE FOR AFRICA ORIGINAL: ENGLISH Sixty-third session Brazzaville, Republic of Congo, 2 6 September, 2013 Agenda item 14 IMMUNIZATION IN THE AFRICAN REGION: PROGRESS REPORT
23 September 2013 REGIONAL COMMITTEE FOR AFRICA ORIGINAL: ENGLISH Sixty-third session Brazzaville, Republic of Congo, 2 6 September, 2013 Agenda item 14 IMMUNIZATION IN THE AFRICAN REGION: PROGRESS REPORT
What is this document and who is it for?
 Measles and Rubella Initiative s Standard Operating Procedures for Accessing Support for Measles and Rubella Supplementary Immunization Activities During 2016 In the context of measles and rubella elimination
Measles and Rubella Initiative s Standard Operating Procedures for Accessing Support for Measles and Rubella Supplementary Immunization Activities During 2016 In the context of measles and rubella elimination
AIDS in Africa. An Update. Basil Reekie
 AIDS in Africa An Update Basil Reekie Contents General Statistics The trend of HIV in Africa Ugandan experience UNAIDS 2006 Latest African Statistics by Country HIV Intervention Light at the end of the
AIDS in Africa An Update Basil Reekie Contents General Statistics The trend of HIV in Africa Ugandan experience UNAIDS 2006 Latest African Statistics by Country HIV Intervention Light at the end of the
Regulatory harmonization and alliances to foster vaccination
 Regulatory harmonization and alliances to foster vaccination Developing Country Vaccine Manufacturers Network 14th AGM 7-9 October 2013, Hanoi Drs David Wood and Ivana Knezevic WHO/HIS/EMP/TSN Outline
Regulatory harmonization and alliances to foster vaccination Developing Country Vaccine Manufacturers Network 14th AGM 7-9 October 2013, Hanoi Drs David Wood and Ivana Knezevic WHO/HIS/EMP/TSN Outline
1) SO1: We would like to suggest that the indicator used to measure vaccine hesitancy be DTP 1 to measles first dose dropout.
 To SAGE Secretariat, WHO Dear Professor Helen Rees, Dear Dr. Jean Marie Okwo-Bele, On behalf of the Civil Society Constituency of the GAVI Alliance, we would like to thank SAGE and its members for the
To SAGE Secretariat, WHO Dear Professor Helen Rees, Dear Dr. Jean Marie Okwo-Bele, On behalf of the Civil Society Constituency of the GAVI Alliance, we would like to thank SAGE and its members for the
Yellow fever Vaccine investment strategy
 Yellow fever Vaccine investment strategy Background document #5 November 2013 Executive summary Since 2001, GAVI has spent ~$250M on yellow fever control $102M on routine vaccination in 17 countries ~$160M
Yellow fever Vaccine investment strategy Background document #5 November 2013 Executive summary Since 2001, GAVI has spent ~$250M on yellow fever control $102M on routine vaccination in 17 countries ~$160M
Malaria Funding. Richard W. Steketee MACEPA, PATH. April World Malaria Day 2010, Seattle WA
 Malaria Funding Richard W. Steketee MACEPA, PATH April World Malaria Day 2010, Seattle WA Malaria Funding Is there a plan? Is there money? Where does the money come from? Is the money moving efficiently?
Malaria Funding Richard W. Steketee MACEPA, PATH April World Malaria Day 2010, Seattle WA Malaria Funding Is there a plan? Is there money? Where does the money come from? Is the money moving efficiently?
WHO VACCINE SUPPLY & QUALITY SUPPORT FOR NATIONAL IMMUNIZATION PROGRAMMES
 WHO VACCINE SUPPLY & QUALITY SUPPORT FOR NATIONAL IMMUNIZATION PROGRAMMES Immunization and vaccine development in the last decade Since the Millennium Summit in 2000, immunization substantial international
WHO VACCINE SUPPLY & QUALITY SUPPORT FOR NATIONAL IMMUNIZATION PROGRAMMES Immunization and vaccine development in the last decade Since the Millennium Summit in 2000, immunization substantial international
PROGRESS REPORT ON THE ROAD MAP FOR ACCELERATING THE ATTAINMENT OF THE MILLENNIUM DEVELOPMENT GOALS RELATED TO MATERNAL AND NEWBORN HEALTH IN AFRICA
 5 July 2011 REGIONAL COMMITTEE FOR AFRICA ORIGINAL: ENGLISH Sixty-first session Yamoussoukro, Côte d Ivoire, 29 August 2 September 2011 Provisional agenda item 17.1 PROGRESS REPORT ON THE ROAD MAP FOR
5 July 2011 REGIONAL COMMITTEE FOR AFRICA ORIGINAL: ENGLISH Sixty-first session Yamoussoukro, Côte d Ivoire, 29 August 2 September 2011 Provisional agenda item 17.1 PROGRESS REPORT ON THE ROAD MAP FOR
TFI Proceedings, Recommendations and implications for 2005
 January 2005 N 053 TFI 2004 - Proceedings, Recommendations and implications for 2005 The 12th meeting of the Task Force on Immunization (TFI) in Africa and the 11th meeting of the Africa Regional Inter-
January 2005 N 053 TFI 2004 - Proceedings, Recommendations and implications for 2005 The 12th meeting of the Task Force on Immunization (TFI) in Africa and the 11th meeting of the Africa Regional Inter-
IMMUNIZATION & VACCINE PREVENTABLE DISEASES
 IMMUNIZATION & VACCINE PREVENTABLE DISEASES MONTHLY IMMUNIZATION UPDATE IN THE AFRICAN REGION July-August 2015 (Vol 3, issue N 6 ) Special issue on WHO/UNICEF Estimates of National Immunization Coverage
IMMUNIZATION & VACCINE PREVENTABLE DISEASES MONTHLY IMMUNIZATION UPDATE IN THE AFRICAN REGION July-August 2015 (Vol 3, issue N 6 ) Special issue on WHO/UNICEF Estimates of National Immunization Coverage
Annex 2 A. Regional profile: West Africa
 Annex 2 A. Regional profile: West Africa 355 million people at risk for malaria in 215 297 million at high risk A. Parasite prevalence, 215 Funding for malaria increased from US$ 233 million to US$ 262
Annex 2 A. Regional profile: West Africa 355 million people at risk for malaria in 215 297 million at high risk A. Parasite prevalence, 215 Funding for malaria increased from US$ 233 million to US$ 262
Overview of the PAHO strategy and activities to strengthen National Regulatory Authorities in the Americas
 Overview of the PAHO strategy and activities to strengthen National Regulatory Authorities in the Americas Ma. de los Angeles Cortes Regional Advisor on Vaccines and Biologicals HSS/MT Annecy, France March
Overview of the PAHO strategy and activities to strengthen National Regulatory Authorities in the Americas Ma. de los Angeles Cortes Regional Advisor on Vaccines and Biologicals HSS/MT Annecy, France March
( A JICA-IRRI-PhilRice Initiative) Presented by Noel Magor, Head Unit Impact Acceleration and Training Center, IRRI
 ( A JICA-IRRI-PhilRice Initiative) Presented by Noel Magor, Head Unit Impact Acceleration and Training Center, IRRI Long partnership between; IRRI & PhilRice IRRI & JICA PhilRice & JICA The Season Long
( A JICA-IRRI-PhilRice Initiative) Presented by Noel Magor, Head Unit Impact Acceleration and Training Center, IRRI Long partnership between; IRRI & PhilRice IRRI & JICA PhilRice & JICA The Season Long
Sourcing of ARVs & HIV diagnostics. Procurement for Impact P4i
 Sourcing of ARVs & HIV diagnostics Procurement for Impact P4i AMDS ANNUAL STAKEHOLDERS AND PARTNERS MEETING 29 September 2014 Global Fund: Procurement for Impact: P4i Our objective was straightforward:
Sourcing of ARVs & HIV diagnostics Procurement for Impact P4i AMDS ANNUAL STAKEHOLDERS AND PARTNERS MEETING 29 September 2014 Global Fund: Procurement for Impact: P4i Our objective was straightforward:
Overview of WHO/UNICEF Immunization Coverage Estimates
 Overview of WHO/UNICEF Immunization Coverage Estimates Marta Gacic-Dobo, Anthony Burton, David Durrheim Meeting of the Strategic Advisory Group of Experts on Immunization (SAGE) 8-10 November 2011 CCV/CICG,
Overview of WHO/UNICEF Immunization Coverage Estimates Marta Gacic-Dobo, Anthony Burton, David Durrheim Meeting of the Strategic Advisory Group of Experts on Immunization (SAGE) 8-10 November 2011 CCV/CICG,
Vaccine standardization in the context of PQ. Dr Ivana Knezevic Quality, Safety and Standards WHO, Geneva
 Vaccine standardization in the context of PQ Dr Ivana Knezevic Quality, Safety and Standards WHO, Geneva Outline WHO standards for vaccines Vaccines of assured quality Written and measurement standards
Vaccine standardization in the context of PQ Dr Ivana Knezevic Quality, Safety and Standards WHO, Geneva Outline WHO standards for vaccines Vaccines of assured quality Written and measurement standards
511,000 (57% new cases) ~50,000 ~30,000
 Latest global TB estimates - 2007 (Updated Mar 2009) All forms of TB Greatest number of cases in Asia; greatest rates per capita in Africa Multidrug-resistant TB (MDR-TB) Estimated number of cases 9.27
Latest global TB estimates - 2007 (Updated Mar 2009) All forms of TB Greatest number of cases in Asia; greatest rates per capita in Africa Multidrug-resistant TB (MDR-TB) Estimated number of cases 9.27
PROGRESS REPORT ON DECADE OF TRADITIONAL MEDICINE IN THE AFRICAN REGION. Progress Report. CONTENTS Paragraphs BACKGROUND PROGRESS MADE...
 5 July 2011 REGIONAL COMMITTEE FOR AFRICA ORIGINAL: ENGLISH Sixty-first session Yamoussoukro, Côte d Ivoire, 29 August 2 September 2011 Provisional agenda item 17.2 PROGRESS REPORT ON DECADE OF TRADITIONAL
5 July 2011 REGIONAL COMMITTEE FOR AFRICA ORIGINAL: ENGLISH Sixty-first session Yamoussoukro, Côte d Ivoire, 29 August 2 September 2011 Provisional agenda item 17.2 PROGRESS REPORT ON DECADE OF TRADITIONAL
POLIO ERADICATION IN THE AFRICAN REGION: PROGRESS REPORT. Information document EXECUTIVE SUMMARY
 7 July 2006 REGIONAL COMMITTEE FOR AFRICA ORIGINAL: ENGLISH Fifty-sixth session Addis Ababa, Ethiopia, 28 August 1 September 2006 Provisional agenda item 10.1 POLIO ERADICATION IN THE AFRICAN REGION: PROGRESS
7 July 2006 REGIONAL COMMITTEE FOR AFRICA ORIGINAL: ENGLISH Fifty-sixth session Addis Ababa, Ethiopia, 28 August 1 September 2006 Provisional agenda item 10.1 POLIO ERADICATION IN THE AFRICAN REGION: PROGRESS
ASLM Building laboratory capacity in Africa in a sustainable way
 ASLM Building laboratory capacity in Africa in a sustainable way Tsehaynesh Messele, PhD ASLM, Chief Executive Officer Annual AMDS meeting September 29-30, 2014 Geneva Laboratory capacity gaps in Africa
ASLM Building laboratory capacity in Africa in a sustainable way Tsehaynesh Messele, PhD ASLM, Chief Executive Officer Annual AMDS meeting September 29-30, 2014 Geneva Laboratory capacity gaps in Africa
Heat and Freeze Sensitivity of Vaccines
 Heat and Freeze Sensitivity of Vaccines DCVMN Workshop "Vaccine quality management systems for manufacturing excellence" Michael Rush, MBA Executive Director - Global Health Policy October 24-25, 2013
Heat and Freeze Sensitivity of Vaccines DCVMN Workshop "Vaccine quality management systems for manufacturing excellence" Michael Rush, MBA Executive Director - Global Health Policy October 24-25, 2013
HPV Vaccine Lessons Learned & New Ways Forward
 HPV Vaccine Lessons Learned & New Ways Forward The Gavi Alliance June 2016, Geneva www.gavi.org Overview 1 Background 2 Lessons learned 3 New Way Forward 2 1 Background 3 HPV Background HPV is responsible
HPV Vaccine Lessons Learned & New Ways Forward The Gavi Alliance June 2016, Geneva www.gavi.org Overview 1 Background 2 Lessons learned 3 New Way Forward 2 1 Background 3 HPV Background HPV is responsible
TB Disease Prevalence Survey - Progress Report
 TB Disease Prevalence Survey - Progress Report 30 Oct 2011, Lille Ikushi Onozaki MD, MPH Team Leader, TB Prevalence Survey WHO Global Task Force on TB Impact Measurement Stop TB Department, WHO onozakii@who.int
TB Disease Prevalence Survey - Progress Report 30 Oct 2011, Lille Ikushi Onozaki MD, MPH Team Leader, TB Prevalence Survey WHO Global Task Force on TB Impact Measurement Stop TB Department, WHO onozakii@who.int
O c t o b e r 1 0,
 STUDENT VOICES FROM THE FIELD: WORLD HEALTH ORGANIZATION GENEVA, SWITZERLAND O c t o b e r 1 0, 2 0 1 6 M o l l y P e z z u l o University at Albany School of Public Health MPH Epidemiology Candidate 17
STUDENT VOICES FROM THE FIELD: WORLD HEALTH ORGANIZATION GENEVA, SWITZERLAND O c t o b e r 1 0, 2 0 1 6 M o l l y P e z z u l o University at Albany School of Public Health MPH Epidemiology Candidate 17
Expert Group Meeting on Strategies for Creating Urban Youth Employment: Solutions for Urban Youth in Africa
 Expert Group Meeting on Strategies for Creating Urban Youth Employment: Solutions for Urban Youth in Africa Measurement/indicators of youth employment Gora Mboup Global Urban Observatory (GUO) UN-HABITAT
Expert Group Meeting on Strategies for Creating Urban Youth Employment: Solutions for Urban Youth in Africa Measurement/indicators of youth employment Gora Mboup Global Urban Observatory (GUO) UN-HABITAT
Medicines Regulation, Regulatory Harmonization, Global Initiatives. Tonya Villafana, PhD, MPH Senior Health Specialist, World Bank
 Medicines Regulation, Regulatory Harmonization, Global Initiatives Tonya Villafana, PhD, MPH Senior Health Specialist, World Bank September, 13, 2012 Presentation Outline The importance of regulation Attributes
Medicines Regulation, Regulatory Harmonization, Global Initiatives Tonya Villafana, PhD, MPH Senior Health Specialist, World Bank September, 13, 2012 Presentation Outline The importance of regulation Attributes
Pneumococcal Conjugate Vaccine: Current Supply & Demand Outlook. UNICEF Supply Division
 Pneumococcal Conjugate Vaccine: Current Supply & Demand Outlook UNICEF Supply Division Update: October 2013 0 Pneumococcal Conjugate Vaccine (PCV) Supply & Demand Outlook October 2013 Update Key updates
Pneumococcal Conjugate Vaccine: Current Supply & Demand Outlook UNICEF Supply Division Update: October 2013 0 Pneumococcal Conjugate Vaccine (PCV) Supply & Demand Outlook October 2013 Update Key updates
Vaccines: (inter)national regulation and quality in resource limited settings
 Vaccines: (inter)national regulation and quality in resource limited settings Raffaella Ravinetto QUAMED Research & Networking Institute of Tropical Medicine Antwerp 12th July 2017 Based on a presentation
Vaccines: (inter)national regulation and quality in resource limited settings Raffaella Ravinetto QUAMED Research & Networking Institute of Tropical Medicine Antwerp 12th July 2017 Based on a presentation
Health systems and HIV: advocacy. Interagency Coalition on AIDS and Development
 Health systems and HIV: Priorities for civil society advocacy Michelle Munro Interagency Coalition on AIDS and Development 1 Overview GTAG, civil society and health systems advocacy Health systems and
Health systems and HIV: Priorities for civil society advocacy Michelle Munro Interagency Coalition on AIDS and Development 1 Overview GTAG, civil society and health systems advocacy Health systems and
IMMUNIZATION VACCINE DEVELOPMENT
 IMMUNIZATION VACCINE DEVELOPMENT IMMUNIZATION AND POLIO UPDATE IN THE AFRICAN REGION July August 2017 (Vol 5 issue N 4) Special issue featuring WHO/UNICEF Estimates of National Immunization Coverage (WUENIC
IMMUNIZATION VACCINE DEVELOPMENT IMMUNIZATION AND POLIO UPDATE IN THE AFRICAN REGION July August 2017 (Vol 5 issue N 4) Special issue featuring WHO/UNICEF Estimates of National Immunization Coverage (WUENIC
JOINT TB AND HIV PROGRAMMING
 JOINT TB AND HIV PROGRAMMING Haileyesus Getahun, WHO. On behalf of the Global Fund Interagency TB and HIV Working Group (Global Fund, PEPFAR, Stop TB Partnership, UNAIDS, WHO) I was admitted in a hospital
JOINT TB AND HIV PROGRAMMING Haileyesus Getahun, WHO. On behalf of the Global Fund Interagency TB and HIV Working Group (Global Fund, PEPFAR, Stop TB Partnership, UNAIDS, WHO) I was admitted in a hospital
STRATEGIC PLAN
 DRAFT: 24 March 2010 STRATEGIC PLAN 2010-15 DEPARTMENT OF IMMUNIZATION, VACCINES AND BIOLOGICALS 1 IVB Strategic Plan 2010-15 Contents Foreword 1. Introduction 1.1 Context of this Strategic Plan 1.2 Analysis
DRAFT: 24 March 2010 STRATEGIC PLAN 2010-15 DEPARTMENT OF IMMUNIZATION, VACCINES AND BIOLOGICALS 1 IVB Strategic Plan 2010-15 Contents Foreword 1. Introduction 1.1 Context of this Strategic Plan 1.2 Analysis
Dr. Valentina Picot Research Advisor. Fondation Mérieux
 Dr. Valentina Picot Research Advisor Fondation Mérieux Mission Strengthen local capacities in developing countries to reduce the impact of infectious diseases on vulnerable populations Official public
Dr. Valentina Picot Research Advisor Fondation Mérieux Mission Strengthen local capacities in developing countries to reduce the impact of infectious diseases on vulnerable populations Official public
Ouagadougou Declaration
 Ouagadougou Declaration on Primary Health Care and Health Systems in Africa: Achieving Better Health for Africa in the New Millennium A declaration by the Members States of the WHO African Region 30 April
Ouagadougou Declaration on Primary Health Care and Health Systems in Africa: Achieving Better Health for Africa in the New Millennium A declaration by the Members States of the WHO African Region 30 April
Status Report on WSS MDG Roadmaps and Country Status Overviews WSP Africa
 Status Report on WSS MDG Roadmaps and Country Status Overviews WSP Africa Ede Ijjasz Global Manager Water and Sanitation Program World Water Forum, Mexico, March 18, 2006 Outline 1. The Water Supply and
Status Report on WSS MDG Roadmaps and Country Status Overviews WSP Africa Ede Ijjasz Global Manager Water and Sanitation Program World Water Forum, Mexico, March 18, 2006 Outline 1. The Water Supply and
Expert Group Meeting on the Regional Report for the African Gender and Development Index
 Expert Group Meeting on the Regional Report for the African Gender and Development Index 9-10 October 2017 United Nations Conference Centre, Addis Ababa, Ethiopia Aide Memoire July 2017 I. Background and
Expert Group Meeting on the Regional Report for the African Gender and Development Index 9-10 October 2017 United Nations Conference Centre, Addis Ababa, Ethiopia Aide Memoire July 2017 I. Background and
PROGRESS REPORT ON CHILD SURVIVAL: A STRATEGY FOR THE AFRICAN REGION. Information Document CONTENTS
 29 June 2009 REGIONAL COMMITTEE FOR AFRICA ORIGINAL: ENGLISH Fifty-ninth session Kigali, Republic of Rwanda, 31 August 4 September 2009 Provisional agenda item 9.2 PROGRESS REPORT ON CHILD SURVIVAL: A
29 June 2009 REGIONAL COMMITTEE FOR AFRICA ORIGINAL: ENGLISH Fifty-ninth session Kigali, Republic of Rwanda, 31 August 4 September 2009 Provisional agenda item 9.2 PROGRESS REPORT ON CHILD SURVIVAL: A
CANCER OF THE CERVIX IN THE AFRICAN REGION: CURRENT SITUATION AND WAY FORWARD
 23 June 2010 REGIONAL COMMITTEE FOR AFRICA ORIGINAL: FRENCH Sixtieth session Malabo, Equatorial Guinea, 30 August 3 September 2010 Provisional agenda item 7.4 CANCER OF THE CERVIX IN THE AFRICAN REGION:
23 June 2010 REGIONAL COMMITTEE FOR AFRICA ORIGINAL: FRENCH Sixtieth session Malabo, Equatorial Guinea, 30 August 3 September 2010 Provisional agenda item 7.4 CANCER OF THE CERVIX IN THE AFRICAN REGION:
Lessons learned from the IeDEA West Africa Collaboration
 Lessons learned from the IeDEA West Africa Collaboration François DABIS with the contribution of Didier Koumavi EKOUEVI INSERM U-897, Bordeaux, France, Programme PACCI, ANRS site, Abidjan, Côte d Ivoire
Lessons learned from the IeDEA West Africa Collaboration François DABIS with the contribution of Didier Koumavi EKOUEVI INSERM U-897, Bordeaux, France, Programme PACCI, ANRS site, Abidjan, Côte d Ivoire
A vaccine s journey: the many steps to saving lives
 A vaccine s journey: the many steps to saving lives GW-USAID Mini-University 7 March 2014 Endale Beyene, MA, MPH Rebecca Fields, MPH Angela Shen, ScD, MPH Outline I. What is the problem and where are we
A vaccine s journey: the many steps to saving lives GW-USAID Mini-University 7 March 2014 Endale Beyene, MA, MPH Rebecca Fields, MPH Angela Shen, ScD, MPH Outline I. What is the problem and where are we
Prospective Models of Vaccine Security Collaborations in Research and Development
 Prospective Models of Vaccine Security Collaborations in Research and Development Georges Thiry, PhD IVI (International Vaccine Institute) Phuket, Oct 01, 2014 Content R&D Examples of collaborations from
Prospective Models of Vaccine Security Collaborations in Research and Development Georges Thiry, PhD IVI (International Vaccine Institute) Phuket, Oct 01, 2014 Content R&D Examples of collaborations from
Global Immunization Vision and Strategies (GIVS) Vaccine Tender , Pretender Meeting, Copenhagen December 2008
 Global Immunization Vision and Strategies (GIVS) 2006-2015 Vaccine Tender 2010-2012, Pretender Meeting, Copenhagen 10-11 December 2008 Dr Ahmed Magan & Dr Osman David Mansoor Programme Division UNICEF
Global Immunization Vision and Strategies (GIVS) 2006-2015 Vaccine Tender 2010-2012, Pretender Meeting, Copenhagen 10-11 December 2008 Dr Ahmed Magan & Dr Osman David Mansoor Programme Division UNICEF
Funding for AIDS: The World Bank s Role. Yolanda Tayler, WB Bi-regional Workshop for the Procurement of ARVs Phnom Penh, Cambodia
 Funding for AIDS: The World Bank s Role Yolanda Tayler, WB Bi-regional Workshop for the Procurement of ARVs Phnom Penh, Cambodia Outline New resources needs estimates Bridging the gap Global overview of
Funding for AIDS: The World Bank s Role Yolanda Tayler, WB Bi-regional Workshop for the Procurement of ARVs Phnom Penh, Cambodia Outline New resources needs estimates Bridging the gap Global overview of
Standardization for Preclinical Evaluation of Influenza Vaccines in Animal Models - WHO activities
 Standardization for Preclinical Evaluation of Influenza Vaccines in Animal Models - WHO activities 2 nd WHO Integrated Meeting on development and clinical trials of Influenza vaccines that induce broadly
Standardization for Preclinical Evaluation of Influenza Vaccines in Animal Models - WHO activities 2 nd WHO Integrated Meeting on development and clinical trials of Influenza vaccines that induce broadly
What Really Works for Raising African Women Leaders in Global Health
 What Really Works for Raising African Women Leaders in Global Health DR. AMA POKUAA FENNY 2018 EXTREME AFFORDABILITY CONFERENCE JULY 9, 2018 ENSIGN COLLEGE OF PUBLIC HEALTH, KPONG, GHANA Outline Goals
What Really Works for Raising African Women Leaders in Global Health DR. AMA POKUAA FENNY 2018 EXTREME AFFORDABILITY CONFERENCE JULY 9, 2018 ENSIGN COLLEGE OF PUBLIC HEALTH, KPONG, GHANA Outline Goals
Immunization Update & focus on meningococcal vaccine PART 1
 Immunization Update & focus on meningococcal vaccine PART 1 Gregory Hussey Vaccines for Africa Initiative Institute of Infectious Diseases University of Cape Town www.vacfa.uct.ac.za Disclosures Received
Immunization Update & focus on meningococcal vaccine PART 1 Gregory Hussey Vaccines for Africa Initiative Institute of Infectious Diseases University of Cape Town www.vacfa.uct.ac.za Disclosures Received
Gavi Unicef Vaccine Industry Consultation. Dominic Hein Market Shaping / Gavi. October Reach every child
 Gavi Update @ Unicef Vaccine Industry Consultation Dominic Hein Market Shaping / Gavi October 2018 Reach every child www.gavi.org Gavi commitments to countries US$ 13.67 billion committed since 2001 New
Gavi Update @ Unicef Vaccine Industry Consultation Dominic Hein Market Shaping / Gavi October 2018 Reach every child www.gavi.org Gavi commitments to countries US$ 13.67 billion committed since 2001 New
HIV/AIDS Country Publications
 1 de 5 01/07/2008 18:18 Publications POLICY at Barcelona The following are abstracts of papers and posters accepted for presentation at the International AIDS Conference 2002: Cost savings and affordability
1 de 5 01/07/2008 18:18 Publications POLICY at Barcelona The following are abstracts of papers and posters accepted for presentation at the International AIDS Conference 2002: Cost savings and affordability
Development of Vaccine Security at the Regional Level
 Workshop among ASEAN countries on Opportunities for Regional Vaccine Security September 30 to October 3, 2014, Cape Panwa, Phuket, Thailand Development of Vaccine Security at the Regional Level Stephane
Workshop among ASEAN countries on Opportunities for Regional Vaccine Security September 30 to October 3, 2014, Cape Panwa, Phuket, Thailand Development of Vaccine Security at the Regional Level Stephane
Presentation by Dr Philippe Douste-Blazy. Chair of UNITAID Special Representative of the UN SG for Innovative Financing for Development
 Presentation by Dr Philippe Douste-Blazy Chair of UNITAID Special Representative of the UN SG for Innovative Financing for Development The Millennium Goals : Hope for Humanity 2000 UN Millennium Summit
Presentation by Dr Philippe Douste-Blazy Chair of UNITAID Special Representative of the UN SG for Innovative Financing for Development The Millennium Goals : Hope for Humanity 2000 UN Millennium Summit
Regional Consultation on Nutrition and HIV/AIDS in French Speaking Countries in Africa Region
 Regional Consultation on Nutrition and HIV/AIDS in French Speaking Countries in Africa Region Evidence, lessons and recommendations for action Ouagadougou, Burkina Faso 17-20 November 2008 CONCEPT PAPER
Regional Consultation on Nutrition and HIV/AIDS in French Speaking Countries in Africa Region Evidence, lessons and recommendations for action Ouagadougou, Burkina Faso 17-20 November 2008 CONCEPT PAPER
Wild Poliovirus*, 03 Aug 2004 to 02 Aug 2005
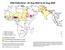 Wild Poliovirus*, 03 Aug 2004 to 02 Aug 2005 Wild virus type 1 Wild virus type 3 Wild virus type 1 & 3 Endemic countries Re-established transmission countries Case or outbreak following importation *Excludes
Wild Poliovirus*, 03 Aug 2004 to 02 Aug 2005 Wild virus type 1 Wild virus type 3 Wild virus type 1 & 3 Endemic countries Re-established transmission countries Case or outbreak following importation *Excludes
WHO Global Task Force on TB Impact Measurement An overview
 WHO Global Task Force on TB Impact Measurement An overview Katherine Floyd (WHO/GTB/TME) 3 rd meeting of the TB estimates subgroup Glion-sur-Montreux, 31March 2 April 2015 GLOBAL TB PROGRAMME 1. Broad
WHO Global Task Force on TB Impact Measurement An overview Katherine Floyd (WHO/GTB/TME) 3 rd meeting of the TB estimates subgroup Glion-sur-Montreux, 31March 2 April 2015 GLOBAL TB PROGRAMME 1. Broad
Prioritizing Emergency Polio Eradication Activities
 Prioritizing Emergency Polio Eradication Activities Managing the Financing Gap the other half of the Emergency Hamid Jafari GPEI Financing 2012-13: Budget = $2.23 b - Confirmed contributions = $1.14 b
Prioritizing Emergency Polio Eradication Activities Managing the Financing Gap the other half of the Emergency Hamid Jafari GPEI Financing 2012-13: Budget = $2.23 b - Confirmed contributions = $1.14 b
40 years of childhood vaccination programmes in Africa. Mind the gap
 40 years of childhood vaccination programmes in Africa Mind the gap Charles Shey Umaru Wiysonge MD, MPhil, PhD, MASSAf Centre for Evidence-based Health Care, Stellenbosch University, South Africa Email
40 years of childhood vaccination programmes in Africa Mind the gap Charles Shey Umaru Wiysonge MD, MPhil, PhD, MASSAf Centre for Evidence-based Health Care, Stellenbosch University, South Africa Email
Global Health Policy: Vaccines
 Global Health Policy: Vaccines Edwin J. Asturias Senior Investigator Colorado School of Public Health Department of Pediatrics Children s Hospital Colorado UNIVERSITY OF COLORADO COLORADO STATE UNIVERSITY
Global Health Policy: Vaccines Edwin J. Asturias Senior Investigator Colorado School of Public Health Department of Pediatrics Children s Hospital Colorado UNIVERSITY OF COLORADO COLORADO STATE UNIVERSITY
Why is Medicine Quality Important?
 Why is Medicine Quality Important? March 3, 2015 Boston University School of Public Health Pharmaceutical Track Spring Symposium Laura Krech, MPH Consultant, based in Boston Promoting the Quality of Medicines
Why is Medicine Quality Important? March 3, 2015 Boston University School of Public Health Pharmaceutical Track Spring Symposium Laura Krech, MPH Consultant, based in Boston Promoting the Quality of Medicines
Yellow Fever Vaccine: Current Outlook. UNICEF Supply Division
 Yellow Fever Vaccine: Current Outlook UNICEF Supply Division March 2015 0 Yellow Fever Vaccine - Current Outlook March 2015 This update provides revised information on the continued constrained 2014-2017
Yellow Fever Vaccine: Current Outlook UNICEF Supply Division March 2015 0 Yellow Fever Vaccine - Current Outlook March 2015 This update provides revised information on the continued constrained 2014-2017
Optimizing Quality of Life through Palliative Care for Adults and Children affected by Cancer
 Optimizing Quality of Life through Palliative Care for Adults and Children affected by Cancer The Public Health need for Palliative Care for Adults and Children with Cancer Dr Emmanuel Luyirika 105M-T3
Optimizing Quality of Life through Palliative Care for Adults and Children affected by Cancer The Public Health need for Palliative Care for Adults and Children with Cancer Dr Emmanuel Luyirika 105M-T3
Diagnostic Procurement The Clinton Foundation HIV/AIDS Initiative
 Diagnostic Procurement The Clinton Foundation HIV/AIDS Initiative 28 October 2008 Geneva 1 CHAI Work in Diagnostics - Context Diagnostic Test Agreements - In 2002 when CHAI was formed, the price of many
Diagnostic Procurement The Clinton Foundation HIV/AIDS Initiative 28 October 2008 Geneva 1 CHAI Work in Diagnostics - Context Diagnostic Test Agreements - In 2002 when CHAI was formed, the price of many
THE CARE WE PROMISE FACTS AND FIGURES 2017
 THE CARE WE PROMISE FACTS AND FIGURES 2017 2 SOS CHILDREN S VILLAGES INTERNATIONAL WHERE WE WORK Facts and Figures 2017 205 58 79 families and transit 31 Foster homes 162 8 3 173 214 2 115 159 136 148
THE CARE WE PROMISE FACTS AND FIGURES 2017 2 SOS CHILDREN S VILLAGES INTERNATIONAL WHERE WE WORK Facts and Figures 2017 205 58 79 families and transit 31 Foster homes 162 8 3 173 214 2 115 159 136 148
CEO Board report Seth Berkley MD CEO
 CEO Board report Seth Berkley MD CEO Dhaka, Bangladesh, 16 17 November 2011 Bangladesh, November 2011 Meningitis Incidence Children < 12 months Dhaka Shishu / Shishu Shahsta Catchment Area 2008 2011 1800
CEO Board report Seth Berkley MD CEO Dhaka, Bangladesh, 16 17 November 2011 Bangladesh, November 2011 Meningitis Incidence Children < 12 months Dhaka Shishu / Shishu Shahsta Catchment Area 2008 2011 1800
BRIEFING ON RTS,S/AS01 MALARIA VACCINE FOR THE SEPTEMBER 2012 MEETING OF MPAC
 BRIEFING ON RTS,S/AS01 MALARIA VACCINE FOR THE SEPTEMBER 2012 MEETING OF MPAC Introduction Date: 12 August 2012. Author: WHO Secretariat with input from JTEG Chair The most advanced vaccine candidate against
BRIEFING ON RTS,S/AS01 MALARIA VACCINE FOR THE SEPTEMBER 2012 MEETING OF MPAC Introduction Date: 12 August 2012. Author: WHO Secretariat with input from JTEG Chair The most advanced vaccine candidate against
Introduction to PAHWP-EAC-LSHTM Workshop on Assessment of Clinical Performance of in-vitro Diagnostics
 Introduction to PAHWP-EAC-LSHTM Workshop on Assessment of Clinical Performance of in-vitro Diagnostics Rosanna W Peeling Professor and Chair, Diagnostic Research Director, International Diagnostics Centre
Introduction to PAHWP-EAC-LSHTM Workshop on Assessment of Clinical Performance of in-vitro Diagnostics Rosanna W Peeling Professor and Chair, Diagnostic Research Director, International Diagnostics Centre
Gender, Poverty, and Health in Sub-Saharan Africa: A Framework for Analysis
 Gender, Poverty, and Health in Sub-Saharan Africa: A Framework for Analysis Pathways to Improved Health Outcomes Health outcomes Households/ Communities Household behaviors & risk factors Community factors
Gender, Poverty, and Health in Sub-Saharan Africa: A Framework for Analysis Pathways to Improved Health Outcomes Health outcomes Households/ Communities Household behaviors & risk factors Community factors
Comparative Analyses of Adolescent Nutrition Indicators
 Comparative Analyses of Adolescent Nutrition Indicators Rukundo K. Benedict, PhD The DHS Program Stakeholders Consultation on Adolescent Girls Nutrition: Evidence, Guidance, and Gaps October 30 31, 2017
Comparative Analyses of Adolescent Nutrition Indicators Rukundo K. Benedict, PhD The DHS Program Stakeholders Consultation on Adolescent Girls Nutrition: Evidence, Guidance, and Gaps October 30 31, 2017
Targeted Diseases and Immunization. Strategic plan
 Targeted Diseases and Immunization Strategic plan 2008-2013 Communicable Diseases Unit WHO Regional Office for Europe Mission To provide technical support to WHO European Region Member States to ensure
Targeted Diseases and Immunization Strategic plan 2008-2013 Communicable Diseases Unit WHO Regional Office for Europe Mission To provide technical support to WHO European Region Member States to ensure
Objectives of Rotavirus Surveillance
 Objectives of Rotavirus Surveillance Contribute data to estimate the burden of rotavirus diarrhea in children < 5 children Document circulating rotavirus strains in the AFR region Support awareness and
Objectives of Rotavirus Surveillance Contribute data to estimate the burden of rotavirus diarrhea in children < 5 children Document circulating rotavirus strains in the AFR region Support awareness and
