Clostridium difficile infection occurs as a complication CASE REPORT
|
|
|
- Barry Mills
- 5 years ago
- Views:
Transcription
1 GASTROENTEROLOGY 2005;128: CASE REPORT Clostridium difficile Toxoid Vaccine in Recurrent C. difficile Associated Diarrhea STAVROS SOUGIOULTZIS,* LORRAINE KYNE, DENISE DRUDY,* SARAH KEATES,* SEEMA MAROO,* CHARALABOS POTHOULAKIS,* PAUL J. GIANNASCA, CYNTHIA K. LEE, MICHEL WARNY, THOMAS P. MONATH, and CIARÁN P. KELLY* *Gastroenterology Division and Gerontology Division, Beth Israel Deaconess Medical Center, Harvard Medical School, Boston, Massachusetts and Acambis, Inc., Cambridge, Massachusetts Background & Aims: Recurrent C difficile-associated diarrhea (CDAD) is associated with a lack of protective immunity to C difficile toxins. A parenteral C difficile vaccine containing toxoid A and toxoid B was reported previously to be safe and immunogenic in healthy volunteers. Our aim was to examine whether the vaccine is also well tolerated and immunogenic in patients with recurrent CDAD. Methods: Subjects received 4, 50- g intramuscular inoculations of the C difficile vaccine over an 8-week period. Serum antitoxin antibodies were measured by ELISA, and toxin neutralizing activity was evaluated using the tissue culture cytotoxin assay. Results: Three patients with multiple episodes of recurrent CDAD were vaccinated. Two of the 3 showed an increase in serum IgG antitoxin A antibodies (3-fold and 4-fold increases, respectively) and in serum IgG antitoxin B antibodies (52-fold and 20-fold, respectively). Both also developed cytotoxin neutralizing activity against toxin A and toxin B. Prior to vaccination, the subjects had required nearly continuous treatment with oral vancomycin for 7, 9, and 22 months, respectively, to treat recurrent episodes of CDAD. After vaccination, all 3 subjects discontinued treatment with oral vancomycin without any further recurrence. Conclusions: A C difficile toxoid vaccine induced immune responses to toxins A and B in patients with CDAD and was associated with resolution of recurrent diarrhea. The results of this study support the feasibility of active vaccination against C difficile and its toxins in high-risk individuals but must be validated in larger, randomized, controlled trials. Clostridium difficile infection occurs as a complication of antibiotic use and is now the most commonly identified cause of nosocomial infectious diarrhea in the developed world. 1 3 C difficile associated diarrhea (CDAD) and pseudomembranous colitis are believed to result from the actions of 2 bacterial exotoxins, toxin A and toxin B, that are produced by pathogenic strains of the bacterium. 4 6 Both toxins have cytotoxic and proinflammatory properties Toxin A causes intestinal injury and secretion in both animals and humans. 9,11,12 Toxin B has little effect on the intact intestine in animal models but appears to be a potent toxin for human colon Management of CDAD involves discontinuation of the inciting antimicrobial agent if possible, as well as supportive care. 1 3,18 If symptoms are prolonged or severe, therapy with oral metrondiazole or vancomycin is indicated. Although these agents are usually effective in treating initial episodes of CDAD, approximately 20% of patients treated with metronidazole or vancomycin will have recurrent diarrhea after treatment is discontinued, and some will have multiple recurrences. 19 Patients who suffer 2 or more episodes of CDAD have a greater than 50% risk for further relapses, and this recurrent form of CDAD continues to be a substantial management problem Factors associated with an increased risk for recurrence include advanced age ( 65 years), severity of the patient s comorbid medical problems, and prolonged antibiotic use. 23 Furthermore, several studies indicate that the host s immune response to C difficile toxins influences the clinical course of the infection as well as the risk of recurrence We reported recently that asymptomatic carriers of C difficile have serum IgG antitoxin A antibody levels that are approximately 3-fold higher than patients with CDAD. 27 In another study, we found that patients with high serum IgG antitoxin A antibody levels during a first episode of CDAD were 48-fold less likely to develop recurrent diarrhea compared with patients with lower serum antitoxin antibody levels. 23 These and other Abbreviations used in this paper: CDAD, C difficile-associated diarrhea by the American Gastroenterological Association /05/$30.00 doi: /j.gastro
2 March 2005 CLOSTRIDIUM DIFFICILE VACCINE 765 studies indicate that a host immune response to C difficile toxins may be protective and reduce the risk for recurrent CDAD. An anti-c difficile toxoid vaccine has been developed containing formalin-inactivated partially purified toxins A and B. The parenterally administered vaccine also protected hamsters from diarrhea and death following oral challenge with toxigenic C difficile. 28,29 Kotloff et al have reported their experience using the C difficile toxoid vaccine administered to healthy adult volunteers. 30 The candidate vaccine was well tolerated and induced robust immune responses to both toxin A and toxin B. In a subsequent study, we also found that the serum antitoxin A antibody concentrations that were achieved during vaccination far exceeded the concentrations that were associated with protection in our clinical studies of CDAD. 31 The aims of this study were to examine whether the C difficile vaccine would be safe and immunogenic in patients with multiple episodes of recurrent CDAD. Materials and Methods Entry Criteria An open-label study was performed in subjects with recurrent CDAD defined as a history of a change in bowel habit with 3 or more unformed bowel movements per day for at least 2 days, associated with a positive stool toxin test for C difficile (either tissue culture cytotoxin assay or toxin A or B enzyme immunoassay) that occurred within 30 days of discontinuation of therapy with metronidazole or oral vancomycin that had been administered for treatment of a prior episode of CDAD. Study inclusion criteria required that subjects be 18 to 85 years of age and in stable health and that clinical laboratory tests (complete blood count, blood chemistries, urinalysis) did not show any unexpected clinically significant abnormalities. Serologic testing for HIV, HBsAg, and HCV were negative. Patients did not have any history of anaphylaxis, wheezing, urticaria, or angioedema or of previous severe allergic reactions to a drug or vaccine. They were not immunosuppressed and did not have a history of chronic or recurrent immune-mediated or inflammatory disease, including collagen vascular diseases or rheumatoid arthritis. The study protocol was approved by the Institutional Review Board at Beth Israel Deaconess Medical Center, Harvard Medical School, Boston, MA, and written informed consent was obtained from all study subjects. C difficile Toxoid Vaccine The C difficile toxoid vaccine was produced as previously described. 28,30,31 Briefly, toxins A and B were partially purified from the culture filtrate of C difficile strain ATCC by ammonium sulfate precipitation. Toxins were further purified by an S300 Sephacryl size exclusion column and were then inactivated with formaldehyde. The total protein concentration of the vaccine was 0.52 mg/ml of which toxins A and B composed approximately 44% at a 1.5:1 toxin A to toxin B ratio. Finally, the vaccine was diluted to contain 50 g protein per 0.4 ml, which was the dose delivered in each inoculation. Vaccination Regimen and Safety Assessments C difficile toxoid vaccine was injected intramuscularly at the deltoid region on 4 occasions, on days 0, 7, 28, and 56. Patients were monitored at the study site for 30 minutes after vaccination to detect immediate adverse reactions. The 33- year-old female subject was advised to practice contraception throughout the course of the study, and a urine pregnancy test was obtained at the screening visit and before each inoculation. Participants also completed a daily symptom diary throughout the treatment period to record all adverse events, including pain, redness, and itching at the injection site as well as general symptoms such as abdominal pain, headache, allergic reactions, and change in bowel habit. Participants were also contacted by telephone 2 consecutive days after each inoculation to monitor for any change in their symptoms. Blood and urine samples were obtained at study entry, prior to each vaccination and 2 weeks after the last vaccination (day 70) to monitor the complete blood count and differential white blood cell count, prothrombin and partial thromboplastin time, serum electrolytes, glucose, liver and renal function tests, amylase, and urinalysis. Serum samples were also collected at baseline, before each of the 4 vaccinations, and 2 weeks after the last vaccination and stored at 80 C for subsequent antitoxin antibody measurements and cytotoxin neutralization assays. All patients continued on vancomycin therapy at doses ranging from 125 mg twice daily to 250 mg 4 times daily until their fourth inoculation on day 56. Antitoxin ELISAs and Cytotoxin Neutralization Assays Serum IgA, IgG, and IgM antitoxin A and antitoxin B antibody levels were measured by ELISA as previously described. 23,25 27,32,33 Toxin A and toxin B were purified from culture supernatant of C difficile VPI strain (ATCC 43255). Microplates (PolySorp, Nunc, Fisher Scientific, Pittsburgh, PA) were coated overnight with toxin A or toxin B (0.05 g in100 L carbonate-bicarbonate buffer, ph 9.6) at 4 C. After washing with PBS supplemented with 0.05% Tween 20 (PBST), plates were saturated with 1% BSA/PBST (200 L/well) for 1 hour at room temperature. After washing, serum samples diluted 100-fold in 0.1% BSA/PBST (100 L/well) were incubated for 1 hour in duplicate. Bound antibodies were detected using horseradish peroxidase-conjugated anti-immunoglobulin G, A, or M (Dako Corporation, Carpinteria, CA) diluted 1000-fold in PBST/0.1%BSA (100 L/ well). After washing with PBST, TMB peroxidase substrate and peroxidase solution (Kirkegaard and Perrry Laboratories, Gaithersburg, MD) was added (100 L/well). The reaction was stopped after 3 minutes by the addition of 1 mol/l phosphoric
3 766 SOUGIOULTZIS ET AL GASTROENTEROLOGY Vol. 128, No. 3 acid (50 L) and optical density (OD) was measured at 450 nm with a reference OD at 630 nm. Human pooled plasma containing high anti-c difficile toxin A and toxin B antibody activity was used in all assays as the standard; the undiluted standard was assigned an arbitrary level of 10,000 ELISA units (EU). Cytotoxicity was determined by rounding of IMR-90 fibroblasts in tissue culture after exposure to purified C difficile toxin A or B. 30,33 38 Neutralization of cytotoxicity by subjects sera was quantified by adding an equal volume of serial 2-fold dilutions of the serum sample (diluted in EMEM culture medium) to 8 times the minimum 50% cytotoxic dose of the toxin preparation (in EMEM) to achieve a final toxin concentration of 4 times the minimum 50% cytotoxic dose. The minimum 50% cytotoxic dose for each toxin preparation was defined as the minimum dose resulting in 50% cell rounding at 24 hours (in these studies, 10 ng/ml for toxin A, and 0.04 ng/ml for toxin B). After incubation at room temperature for 60 minutes, the serum/toxin mixture was added to the IMR-90 monolayers (100 L/well). The neutralizing activity of the serum sample was reported as the highest dilution of serum that prevented rounding of 50% of the IMR-90 cells after overnight culture at 37 C. Results Case 1 A 51-year-old man presented to his primary care physician because of increased frequency of micturation and nocturia. He was found to have an elevated level of serum prostate-specific antigen and was treated with ciprofloxacin 500 mg, twice daily for 21 days for suspected chronic prostatitis. Further studies revealed a localized prostate lesion, and, in February 2001, he underwent transrectal prostate biopsy after enema preparation and an additional single prophylactic dose of ciprofloxacin 500 mg. Five days later, he developed diarrhea, and a stool sample was positive for C difficile toxin A. Metronidazole, 250 mg 4 times daily, was prescribed. After 3 days of therapy, there was no improvement in his diarrhea. Metronidazole was discontinued and replaced by oral vancomycin, 125 mg 3 times daily. His diarrhea resolved, and vancomycin therapy was discontinued after 21 days of treatment. Ten days later, he developed recurrent diarrhea, and stool testing was again positive for C difficile toxin. Vancomycin was restarted, and the diarrhea resolved but recurred again when the vancomycin was discontinued. He again resumed treatment with oral vancomycin, and, in June 2001, he underwent a radical prostatectomy for early stage prostatic carcinoma. After surgery, the dosage of vancomycin was gradually tapered followed by pulse dosing. 22 In August, he discontinued treatment with vancomycin completely but, within 1 week, developed another episode of CDAD. He enrolled in the vaccine trial in September 2001 having been on treatment with oral vancomycin almost continuously for 7 months. Case 2 In August 2000, a 71-year-old woman developed diarrhea after taking cefuroxime 250 mg twice daily for 7 days to treat a urinary tract infection. A stool test was positive for C difficile toxin A. The diarrhea responded well to treatment with metronidazole, 500 mg twice daily for 10 days. However, the diarrhea recurred 10 days after stopping metronidazole treatment. Stool testing was again positive for C difficile toxin, and vancomycin 125 mg was administered 4 times daily. The diarrhea resolved but recurred when vancomycin was discontinued. During the following months, the patient attempted to discontinue treatment with oral vancomycin on at least 10 occasions, but, at each attempt, diarrhea recurred within 3 to 10 days of stopping the antibiotic therapy. On most occasions, the vancomycin dose was gradually tapered. During this 18-month period, she also received courses of therapy with cholestyramine, Pepto- Bismol, VSL3 (a cocktail of probiotics containing strains of Lactobacillus, Bifidobacterium, and Streptococcus salivarius) or pooled normal intravenous immunoglobulin, but none of these different approaches was effective in preventing subsequent recurrence of CDAD. 3,19 She enrolled in the vaccine trial in June 2002 having been on treatment with oral vancomycin almost continuously for 22 months. Case 3 In September 2001, a 33-year-old woman developed diarrhea while taking cefuroxime 250 mg twice daily for 4 days to treat a urinary tract infection. A stool test was positive for C difficile toxin. She took metronidazole 250 mg 4 times daily for 2 weeks with resolution of the diarrhea. Three days after stopping metronidazole, the diarrhea recurred, and she was again treated with oral metronidazole for 2 additional weeks. When the metronidazole therapy was stopped, she suffered a recurrence of diarrhea, and a stool test was again positive for C difficile toxin. She was subsequently treated with a variety of regimens, including a gradually tapering dose of vancomycin, bacitracin, rifampicin, Saccharomyces boulardii, and Lactobacillus GG. On each occasion, diarrhea recurred within 2 to 9 days of stopping antibiotic therapy. She enrolled in the vaccine trial in June 2002 having been on treatment with metronidazole or vancomycin almost continuously for 9 months.
4 March 2005 CLOSTRIDIUM DIFFICILE VACCINE 767 Figure 1. Serum IgG antitoxin A antibody values in 3 subjects with recurrent C difficile associated diarrhea. The subjects received intramuscular inoculations of a C difficile toxin vaccine on days 0, 7, 28, and 56 (arrows). Serum IgG antitoxin A antibody was measured by ELISA. The highest dilutions of serum that neutralized the cytotoxicity of purified C difficile toxin A are shown in parentheses for all serum samples that had detectable toxin neutralizing activity. Clinical Outcome and Safety Data All 3 subjects discontinued treatment with oral vancomycin after their fourth and final inoculation with the C difficile toxoid vaccine. Follow-up was available for all subjects for at least 6 months after they completed vaccination and discontinued vancomycin treatment, and none developed recurrent CDAD. The vaccine was well tolerated, and there were no adverse events that were definitely or probably related to vaccination. Adverse events that were possibly related to vaccination based on temporal association included a mild headache in subject 1 and mild abdominal pain and a change in bowel habit in subject 2. Subject 2 also reported transient polyarthralgia after the fourth inoculation. She later developed atypical polyarthritis with a normal erythrocyte sedimentation rate, a negative rheumatoid factor test, and slightly elevated C-reactive protein. Approximately 2 months after completion of the study, a clinical diagnosis of polymyalgia rheumatica was made by a rheumatologist. She was treated with a course of oral corticosteroids with good effect. Immune Response to C difficile Toxins Two of the 3 subjects showed an increase in both antitoxin A and antitoxin B serum IgG antibody levels compared with their baseline values (Figures 1 and 2). Subject 2 showed 3-fold (1.9 to 5.7 EU) and 52-fold (1.2 to 63.0 EU) increases in serum IgG antibodies against toxin A and toxin B, respectively. Subject 3 showed a 4-fold (8.2 to 34.6 EU) increase in serum IgG antitoxin A and a 20-fold (4.6 to 93.9 EU) increase in serum IgG antitoxin B antibody values. Antibody increases were seen after the fourth vaccination for toxin A and after the third for toxin B (Figures 1 and 2). Subject 1 did not demonstrate any substantial increase in antibody levels against either toxin (1.3-fold for both toxin A and toxin B) (Figures 1 and 2). There was no significant increase in serum IgA or IgM antitoxin A or B antibodies in any subject. The ability of serum to neutralize the cytotoxic activity of C difficile toxin A and toxin B generally coincided with increases in antitoxin antibody levels as measured by ELISA (Figures 1 and 2). Subject 1 did not develop serum toxin neutralizing activity at any time point. Subject 2 developed neutralizing activity against toxin A after the fourth vaccination and against toxin B after the third. Subject 3 developed neutralizing activity against toxin A after the third vaccination and against toxin B after the fourth. Discussion Recurrent C difficile associated diarrhea is a common clinical problem and affects as many as 35% of all patients treated for CDAD in our institution. 23,39 The management of patients with multiple recurrences is especially challenging. 19 The subjects in this study are typical of such patients in that they had good control of symptoms for prolonged periods while taking vancomycin but repeatedly suffered recurrent diarrhea within a few days of attempting to discontinue this antibiotic. They had also previously used a variety of treatment regimens in an attempt to prevent further recurrences. Figure 2. Serum IgG antitoxin B antibody values in 3 subjects with recurrent C difficile associated diarrhea. The subjects received intramuscular inoculations of a C difficile toxin vaccine on days 0, 7, 28, and 56 (arrows). Serum IgG antitoxin B antibody was measured by ELISA. The highest dilutions of serum that neutralized the cytotoxicity of purified C difficile toxin B are shown in parentheses for all serum samples that had detectable toxin neutralizing activity.
5 768 SOUGIOULTZIS ET AL GASTROENTEROLOGY Vol. 128, No. 3 These included tapering and pulsed doses of oral vancomycin, combination therapy with other antibiotics (rifampicin, bacitracin), cholestyramine, probiotic therapy (Saccharomyces boulardii, Lactobacillus GG, and VSL 3), and passive immunotherapy using intravenous immunoglobulin. The reason why some patients are predisposed to prolonged and recurrent CDAD is not entirely clear. The failure to cure CDAD is seldom associated with microbial resistance to metronidazole or vancomycin. It may result from persistence of C difficile or its spores in the intestine, despite antibiotic therapy. 22 Alternatively, recurrent diarrhea and colitis may follow reinfection of susceptible individuals with the same or a different strain of C difficile from their environment. 40 There is now substantial evidence that an inadequate humoral immune response to C difficile toxins is associated with an increased risk for recurrent CDAD. The earliest published report was that high titers of serum IgG antitoxin B antibodies were associated with recovery from CDAD without subsequent relapse. 24 Warny et al reported that serum IgG and fecal IgA antitoxin antibodies were significantly higher in patients with a single, brief episode of CDAD compared to those with recurrent CDAD. 26 In another study, children with relapsing CDAD had significantly lower serum IgG antitoxin A antibodies compared with controls. 25 Furthermore, intravenous infusion of pooled normal immunoglobulin containing IgG antitoxin A increased serum IgG antitoxin A levels and appeared to be effective in preventing further relapses. In a recent study, patients with symptomless C difficile infection had a high serum IgG antitoxin A antibody, but there was no evident association between protection and either serum or secretory IgA antitoxin. 27 In another prospective cohort study of 63 patients, those who had only a single episode of diarrhea showed a significant increase in their serum IgG antitoxin A antibodies during their first episode of CDAD, whereas those who later had recurrent CDAD did not. 23 Taken together, these studies indicate that recurrent CDAD is associated with a failure to develop a protective immune response to C difficile toxins. 41 Vaccination using inactivated C difficile toxins protects animals against C difficile-associated enterocolitis and death. In a previous study, a C difficile toxoid vaccine similar to that used in this study was administered to 30 healthy volunteers. 30 Over 90% of the subjects showed substantial increases in their serum antitoxin A and antitoxin B antibodies and developed serum toxin neutralizing activity. However, as discussed above, the ability of patients with recurrent CDAD to mount an immune response to C difficile toxins seems to be impaired. In a previous study of healthy volunteers, the C difficile toxoid vaccine induced substantially greater immune responses, in the form of elevations in serum IgG antitoxin A, than those observed in patients with C difficile infection. 31 The presentation of C difficile toxin antigens in the context of multiple intramuscular inoculations of purified toxoids is very different from that occurring during colonic luminal infection. These differences in antigen type and in the route and nature of antigen presentation may account for the ability of the vaccine to induce immune responses that are different from those observed during natural infection. However, it may be especially difficult to induce protective immunity through vaccination in those individuals who fail to develop immunity during natural challenge with C difficile. 42 This issue was a major factor in our decision to perform this pilot study in patients with recurrent CDAD. If active vaccination is ineffective in this population, alternative approaches such as passive immunotherapy using hyperimmune globulin may be required. 23,25,33,42 This study examines vaccination against C difficile in at-risk subjects. Two of our 3 subjects with recurrent CDAD showed an increase in serum IgG antitoxin antibodies. However, perhaps predictably, the fold increases in antitoxin antibodies were relatively modest when compared with those observed previously in healthy volunteers. 30,31 This was especially the case for serum IgG antitoxin A of which 3-fold and 4-fold increases above baseline were observed, whereas, for serum IgG antitoxin B, there were 52-fold and 20-fold increases, respectively, in the same 2 subjects. This finding agrees with our previous studies, demonstrating a specific decrease in humoral immune responses to toxin A in symptomatic and recurrent CDAD. 23,25 27 Both subjects who had increases in antitoxin antibodies by ELISA showed concomitant toxin neutralizing activity in their sera. This is arguably an important indicator of protective immunity reflecting the inhibition of a key biologic function of the toxins, ie, monoglucosylation of rho proteins leading to disaggregation of filamentous actin and cell rounding. 43,44 However, a correlation between serum toxin neutralizing activity as determined using the tissue culture cytotoxin assay and inhibition of enterotoxicity or protection against C difficile associated diarrhea and colitis has not been demonstrated in humans. After vaccination, all 3 subjects, including one who showed no substantial increase in serum antitoxin antibodies or serum toxin neutralizing activity, were able to discontinue vancomycin without further recurrence of CDAD. There are a number of possible explanations for our finding that 1 of the 3 subjects did not mount an evident antitoxin antibody response during vaccination yet remained disease free. One possibility is that his
6 March 2005 CLOSTRIDIUM DIFFICILE VACCINE 769 recovery from recurrent CDAD was independent of the vaccination regime and occurred despite his continued lack of a protective immune response. A second possibility is that vaccination was protective but that the parameters used to monitor the immune response (ie, serum antitoxin antibody concentrations by ELISA and cytotoxicity neutralizing activity) do not accurately measure the true effectors of protective immunity to CDAD. It is not clear whether the positive outcome in all 3 subjects indicates that the C difficile vaccine is clinically effective because the study was small and uncontrolled. However, our findings do support the feasibility of active vaccination against C difficile and its toxins in high-risk individuals. Suitable study population may include those with chronic diseases that are likely to require frequent antibiotic therapy and admission to health care facilities at which CDAD is endemic. 41,42,45 These populations are similar to those in whom immunization against influenza and pneumococcus are currently performed. This factor could facilitate the administration of a C difficile vaccine if it can be combined with other recommended immunizations. In institutions such as hospitals or nursing homes, herd immunity can be very important in limiting the spread of infectious diseases. 46 It is not clear that a toxoid-based vaccine can prevent intestinal colonization by C difficile. 29 Nonetheless, vaccination may indirectly reduce cross infection if infected individuals are protected against CDAD because patients with diarrhea are a major source for person-to-person spread and for environmental contamination by C difficile and its spores Based on the encouraging results obtained in this pilot study, we believe that the efficacy of vaccination against C difficile should be tested by randomized controlled trials in populations at risk for C difficile toxin-induced diarrhea and pseudomembranous colitis. References 1. Bartlett JG. Clinical practice. Antibiotic-associated diarrhea. N Engl J Med 2002;346: Kelly CP, Pothoulakis C, LaMont JT. Clostridium difficile colitis. N Engl J Med 1994;330: Kyne L, Farrell RJ, Kelly CP. Clostridium difficile. Gastroenterol Clin North Am 2001;30: Bartlett JG, Chang TW, Gurwith M, Gorbach SL, Onderdonk AB. Antibiotic-associated pseudomembranous colitis due to toxinproducing clostridia. N Engl J Med 1978;298: Borriello SP. 12th C.L. Oakley lecture. Pathogenesis of Clostridium difficile infection of the gut. J Med Microbiol 1990;33: Pothoulakis C. Pathogenesis of Clostridium difficile-associated diarrhoea. Eur J Gastroenterol Hepatol 1996;8: Linevsky JK, Pothoulakis C, Keates S, Warny M, Keates AC, LaMont JT, Kelly CP. IL-8 release and neutrophil activation by Clostridium difficile toxin-exposed human monocytes. Am J Physiol 1997;273:G1333 G Sullivan NM, Pellett S, Wilkins TD. Purification and characterization of toxins A and B of Clostridium difficile. Infect Immun 1982;35: Triadafilopoulos G, Pothoulakis C, O Brien MJ, LaMont JT. Differential effects of Clostridium difficile toxins A and B on rabbit ileum. Gastroenterology 1987;93: Warny M, Keates AC, Keates S, Castagliuolo I, Zacks JK, Aboudola S, Qamar A, Pothoulakis C, LaMont JT, Kelly CP. p38 MAP kinase activation by Clostridium difficile toxin A mediates monocyte necrosis, IL-8 production, and enteritis. J Clin Invest 2000; 105: Lyerly DM, Saum KE, MacDonald DK, Wilkins TD. Effects of Clostridium difficile toxins given intragastrically to animals. Infect Immun 1985;47: Mitchell TJ, Ketley JM, Haslam SC, Stephen J, Burdon DW, Candy DC, Daniel R. Effect of toxin A and B of Clostridium difficile on rabbit ileum and colon. Gut 1986;27: Johnson S, Kent SA, O Leary KJ, Merrigan MM, Sambol SP, Peterson LR, Gerding DN. Fatal pseudomembranous colitis associated with a variant Clostridium difficile strain not detected by toxin A immunoassay. Ann Intern Med 2001;135: Limaye AP, Turgeon DK, Cookson BT, Fritsche TR. Pseudomembranous colitis caused by a toxin A( ) B( ) strain of Clostridium difficile. J Clin Microbiol 2000;38: Lyerly DM, Barroso LA, Wilkins TD, Depitre C, Corthier G. Characterization of a toxin A-negative, toxin B-positive strain of Clostridium difficile. Infect Immun 1992;60: Riegler M, Sedivy R, Pothoulakis C, Hamilton G, Zacherl J, Bischof G, Cosentini E, Feil W, Schiessel R, LaMont JT. Clostridium difficile toxin B is more potent than toxin A in damaging human colonic epithelium in vitro. J Clin Invest 1995;95: Sambol SP, Merrigan MM, Lyerly D, Gerding DN, Johnson S. Toxin gene analysis of a variant strain of Clostridium difficile that causes human clinical disease. Infect Immun 2000;68: Fekety R. Guidelines for the diagnosis and management of Clostridium difficile-associated diarrhea and colitis. American College of Gastroenterology, Practice Parameters Committee. Am J Gastroenterol 1997;92: Kyne L, Kelly CP. Recurrent Clostridium difficile diarrhoea. Gut 2001;49: Fekety R, Shah AB. Diagnosis and treatment of Clostridium difficile colitis. JAMA 1993;269: McFarland LV, Surawicz CM, Greenberg RN, Fekety R, Elmer GW, Moyer KA, Melcher SA, Bowen KE, Cox JL, Noorani Z. A randomized placebo-controlled trial of Saccharomyces boulardii in combination with standard antibiotics for Clostridium difficile disease [published erratum appears in JAMA 1994;272:518]. JAMA 1994;271: Tedesco FJ, Gordon D, Fortson WC. Approach to patients with multiple relapses of antibiotic-associated pseudomembranous colitis. Am J Gastroenterol 1985;80: Kyne L, Warny M, Qamar A, Kelly CP. Association between antibody response to toxin A and protection against recurrent Clostridium difficile diarrhoea. Lancet 2001;357: Aronsson B, Granstrom M, Mollby R, Nord CE. Serum antibody response to Clostridium difficile toxins in patients with Clostridium difficile diarrhoea. Infection 1985;13: Leung DY, Kelly CP, Boguniewicz M, Pothoulakis C, LaMont JT, Flores A. Treatment with intravenously administered gamma globulin of chronic relapsing colitis induced by Clostridium difficile toxin. J Pediatr 1991;118: Warny M, Vaerman JP, Avesani V, Delmee M. Human antibody response to Clostridium difficile toxin A in relation to clinical course of infection. Infect Immun 1994;62:
7 770 SOUGIOULTZIS ET AL GASTROENTEROLOGY Vol. 128, No Kyne L, Warny M, Qamar A, Kelly CP. Asymptomatic carriage of Clostridium difficile and serum levels of IgG antibody against toxin A. N Engl J Med 2000;342: Giannasca PJ, Zhang ZX, Lei WD, Boden JA, Giel MA, Monath TP, Thomas WD Jr. Serum antitoxin antibodies mediate systemic and mucosal protection from Clostridium difficile disease in hamsters. Infect Immun 1999;67: Torres JF, Lyerly DM, Hill JE, Monath TP. Evaluation of formalininactivated Clostridium difficile vaccines administered by parenteral and mucosal routes of immunization in hamsters. Infect Immun 1995;63: Kotloff KL, Wasserman SS, Losonsky GA, Thomas W Jr, Nichols R, Edelman R, Bridwell M, Monath TP. Safety and immunogenicity of increasing doses of a Clostridium difficile toxoid vaccine administered to healthy adults. Infect Immun 2001;69: Aboudola S, Kotloff KL, Kyne L, Warny M, Kelly EC, Sougioultzis S, Giannasca PJ, Monath TP, Kelly CP. Clostridium difficile vaccine and serum immunoglobulin G antibody response to toxin A. Infect Immun 2003;71: Kelly CP, Pothoulakis C, Orellana J, LaMont JT. Human colonic aspirates containing immunoglobulin A antibody to Clostridium difficile toxin A inhibit toxin A-receptor binding. Gastroenterology 1992;102: Salcedo J, Keates S, Pothoulakis C, Warny M, Castagliuolo I, LaMont JT, Kelly CP. Intravenous immunoglobulin therapy for severe Clostridium difficile colitis. Gut 1997;41: Kelly CP, Pothoulakis C, Vavva F, Castagliuolo I, Bostwick EF, O Keane JC, Keates S, LaMont JT. Anti-Clostridium difficile bovine immunoglobulin concentrate inhibits cytotoxicity and enterotoxicity of C difficile toxins. Antimicrob Agents Chemother 1996; 40: Kelly CP, Chetham S, Keates S, Bostwick EF, Roush AM, Castagliuolo I, LaMont JT, Pothoulakis C. Survival of anti-clostridium difficile bovine immunoglobulin concentrate in the human gastrointestinal tract. Antimicrob Agents Chemother 1997;41: Kim K, Pickering LK, DuPont HL, Sullivan N, Wilkins T. In vitro and in vivo neutralizing activity of human colostrum and milk against purified toxins A and B of Clostridium difficile. J Infect Dis 1984; 150: Wada N, Nishida N, Iwaki S, Ohi H, Miyawaki T, Taniguchi N, Migita S. Neutralizing activity against Clostridium difficile toxin in the supernatants of cultured colostral cells. Infect Immun 1980; 29: Warny M, Fatimi A, Bostwick EF, Laine DC, Lebel F, LaMont JT, Pothoulakis C, Kelly CP. Bovine immunoglobulin concentrateclostridium difficile retains C difficile toxin neutralising activity after passage through the human stomach and small intestine. Gut 1999;44: Fekety R, McFarland LV, Surawicz CM, Greenberg RN, Elmer GW, Mulligan ME. Recurrent Clostridium difficile diarrhea: characteristics of and risk factors for patients enrolled in a prospective, randomized, double-blinded trial. Clin Infect Dis 1997; 24: Wilcox MH, Fawley WN, Settle CD, Davidson A. Recurrence of symptoms in Clostridium difficile infection relapse or reinfection? J Hosp Infect 1998;38: Kelly CP. Immune response to Clostridium difficile infection. Eur J Gastroenterol Hepatol 1996;8: Wilcox M, Minton J. Role of antibody response in outcome of antibiotic-associated diarrhoea. Lancet 2001;357: Just I, Wilm M, Selzer J, Rex G, Eichel-Streiber C, Mann M, Aktories K. The enterotoxin from Clostridium difficile (ToxA) monoglucosylates the Rho proteins. J Biol Chem 1995;270: Just I, Selzer J, Wilm M, Eichel-Streiber C, Mann M, Aktories K. Glucosylation of Rho proteins by Clostridium difficile toxin B. Nature 1995;375: Kyne L, Kelly CP. Prospects for a vaccine for Clostridium difficile. BioDrugs 1998;10: Starr JM, Rogers TR, Impallomeni M. Hospital-acquired Clostridium difficile diarrhoea and herd immunity. Lancet 1997;349: Gerding DN, Johnson S, Peterson LR, Mulligan ME, Silva J Jr. Clostridium difficile-associated diarrhea and colitis. Infect Control Hosp Epidemiol 1995;16: Nolan NP, Kelly CP, Humphreys JF, Cooney C, O Connor R, Walsh TN, Weir DG, O Briain DS. An epidemic of pseudomembranous colitis: importance of person to person spread. Gut 1987;28: Samore MH. Epidemiology of nosocomial clostridium difficile diarrhoea. J Hosp Infect 1999;43(Suppl): Received June 27, Accepted October 27, Address requests for reprints to: Ciarán P. Kelly, M.D., Dana 601, Gastroenterology, Beth Israel Deaconess Medical Center, Boston, Massachusetts ckelly2@bidmc.harvard.edu; fax: (617) Drs. Kellly and Pothoulakis have acted as consultants or scientific advisors to companies, Bicodex Inc., GelTex Pharmaceuticals Inc., SynSorb Biotech Inc., and ActivBiotics Inc., interested in developing therapeutic intervention for C difficile diarrhea. Dr. Kyne acted as a consultant for GelTex Pharmaceuticals Inc. Drs. Giannasca, Warny, Lee, and Monath are employees and shareholders of Acamis Inc. During this study, L. Kyne held a Paul Beeson Faculty Scholars in Aging Research career development award. Supported by grants AG00971 (to L.K.), AI (to C.P.K.), and RR01032 (to Beth Israel Deaconess Medical Center, General Clinical Research Center) from the National Institutes of Health and by Acambis, Inc. The authors thank Dr. Scott Kitchener and Vincent Knobel for their assistance.
Prospective Derivation and Validation of a Clinical Prediction Rule for Recurrent Clostridium difficile Infection
 GASTROENTEROLOGY 2009;136:1206 1214 Prospective Derivation and Validation of a Clinical Prediction Rule for Recurrent Clostridium difficile Infection MARY Y. HU,* KIANOOSH KATCHAR,* LORRAINE KYNE, SEEMA
GASTROENTEROLOGY 2009;136:1206 1214 Prospective Derivation and Validation of a Clinical Prediction Rule for Recurrent Clostridium difficile Infection MARY Y. HU,* KIANOOSH KATCHAR,* LORRAINE KYNE, SEEMA
The New England Journal of Medicine. Study Protocol
 ASYMPTOMATIC CARRIAGE OF CLOSTRIDIUM DIFFICILE AND SERUM LEVELS OF IgG ANTIBODY AGAINST TOXIN A LORRAINE KYNE, M.B., M.P.H., MICHEL WARNY, M.D., AMIR QAMAR, M.D., AND CIARÁN P. KELLY, M.D. ABSTRACT Background
ASYMPTOMATIC CARRIAGE OF CLOSTRIDIUM DIFFICILE AND SERUM LEVELS OF IgG ANTIBODY AGAINST TOXIN A LORRAINE KYNE, M.B., M.P.H., MICHEL WARNY, M.D., AMIR QAMAR, M.D., AND CIARÁN P. KELLY, M.D. ABSTRACT Background
Intravenous immunoglobulin therapy for severe Clostridium diycile colitis
 366 Section of Gastroenterology, Boston University School of Medicine, Boston, Massachusetts, USA J Salcedo Gastroenterology Division, Beth Israel Deaconess Medical Center, Harvard Medical School, Boston,
366 Section of Gastroenterology, Boston University School of Medicine, Boston, Massachusetts, USA J Salcedo Gastroenterology Division, Beth Israel Deaconess Medical Center, Harvard Medical School, Boston,
ENGLISH FOR PROFESSIONAL PURPOSES UNIT 3 HOW TO DEAL WITH CLOSTRIDIUM DIFFICILE
 ENGLISH FOR PROFESSIONAL PURPOSES UNIT 3 HOW TO DEAL WITH CLOSTRIDIUM DIFFICILE The diagnosis of CDI should be based on a combination of clinical and laboratory findings. A case definition for the usual
ENGLISH FOR PROFESSIONAL PURPOSES UNIT 3 HOW TO DEAL WITH CLOSTRIDIUM DIFFICILE The diagnosis of CDI should be based on a combination of clinical and laboratory findings. A case definition for the usual
Division of GIM Lecture Series Case Presentation David A. Erickson, M.D October 9th, 2013
 Division of GIM Lecture Series Case Presentation David A. Erickson, M.D October 9th, 2013 Financial Disclosures No financial disclosures Objectives Review a case of recurrent Clostridium difficile infection
Division of GIM Lecture Series Case Presentation David A. Erickson, M.D October 9th, 2013 Financial Disclosures No financial disclosures Objectives Review a case of recurrent Clostridium difficile infection
EDUCATIONAL COMMENTARY CLOSTRIDIUM DIFFICILE UPDATE
 EDUCATIONAL COMMENTARY CLOSTRIDIUM DIFFICILE UPDATE Educational commentary is provided through our affiliation with the American Society for Clinical Pathology (ASCP). To obtain FREE CME/CMLE credits click
EDUCATIONAL COMMENTARY CLOSTRIDIUM DIFFICILE UPDATE Educational commentary is provided through our affiliation with the American Society for Clinical Pathology (ASCP). To obtain FREE CME/CMLE credits click
ABSTRACT PURPOSE METHODS
 ABSTRACT PURPOSE The purpose of this study was to characterize the CDI population at this institution according to known risk factors and to examine the effect of appropriate evidence-based treatment selection
ABSTRACT PURPOSE The purpose of this study was to characterize the CDI population at this institution according to known risk factors and to examine the effect of appropriate evidence-based treatment selection
C lostridium difficile is an anaerobic Gram positive bacillus
 673 GASTROINTESTINAL INFECTION Clostridium difficile associated diarrhoea in hospitalised patients: onset in the community and hospital and role of S S Johal, J Hammond, K Solomon, P D James, Y R Mahida...
673 GASTROINTESTINAL INFECTION Clostridium difficile associated diarrhoea in hospitalised patients: onset in the community and hospital and role of S S Johal, J Hammond, K Solomon, P D James, Y R Mahida...
Antibiotic treatment comparison in patients with diarrhea
 Original Research Article Antibiotic treatment comparison in patients with diarrhea Deva Lal Kast * Senior Consultant Physician, Department of General Medicine, Krishna Hospital, Ex senior Specialist and
Original Research Article Antibiotic treatment comparison in patients with diarrhea Deva Lal Kast * Senior Consultant Physician, Department of General Medicine, Krishna Hospital, Ex senior Specialist and
Predictors of Mortality and Morbidity in Clostridium Difficile Infection
 Predictors of Mortality and Morbidity in Clostridium Difficile Infection Jill Dixon, Brian F. Menezes Corresponding author: dippers82@hotmail.com Pages 23-26 ISSN 1840-4529 http://www.iomcworld.com/ijcrimph
Predictors of Mortality and Morbidity in Clostridium Difficile Infection Jill Dixon, Brian F. Menezes Corresponding author: dippers82@hotmail.com Pages 23-26 ISSN 1840-4529 http://www.iomcworld.com/ijcrimph
International Journal of Food and Allied Sciences
 International Journal of Food and Allied Sciences ISSN: 2415-0290 (Print) ISSN: 2413-2543 (Online) DOI:10.21620/ijfaas.2017120-26 Research Article History The Role of Saccharomyces boulardii in the Treatment
International Journal of Food and Allied Sciences ISSN: 2415-0290 (Print) ISSN: 2413-2543 (Online) DOI:10.21620/ijfaas.2017120-26 Research Article History The Role of Saccharomyces boulardii in the Treatment
Labeled Uses: Treatment of Clostiridum Difficile associated diarrhea (CDAD)
 Brand Name: Dificid Generic Name: fidaxomicin Manufacturer 1,2,3,4,5 : Optimer Pharmaceuticals, Inc. Drug Class 1,2,3,4,5 : Macrolide Antibiotic Uses 1,2,3,4,5 : Labeled Uses: Treatment of Clostiridum
Brand Name: Dificid Generic Name: fidaxomicin Manufacturer 1,2,3,4,5 : Optimer Pharmaceuticals, Inc. Drug Class 1,2,3,4,5 : Macrolide Antibiotic Uses 1,2,3,4,5 : Labeled Uses: Treatment of Clostiridum
Health Care Costs and Mortality Associated with Nosocomial Diarrhea Due to Clostridium difficile
 MAJOR ARTICLE Health Care Costs and Mortality Associated with Nosocomial Diarrhea Due to Clostridium difficile Lorraine Kyne, 1 Mary Beth Hamel, 2 Rajashekhar Polavaram, 3 and Ciarán P. Kelly 3 Divisions
MAJOR ARTICLE Health Care Costs and Mortality Associated with Nosocomial Diarrhea Due to Clostridium difficile Lorraine Kyne, 1 Mary Beth Hamel, 2 Rajashekhar Polavaram, 3 and Ciarán P. Kelly 3 Divisions
Clostridium difficile Infection: Diagnosis and Management
 Clostridium difficile Infection: Diagnosis and Management Brian Viviano D.O. Case study 42 year old female with history of essential hypertension and COPD presents to ED complaining of 24 hours of intractable,
Clostridium difficile Infection: Diagnosis and Management Brian Viviano D.O. Case study 42 year old female with history of essential hypertension and COPD presents to ED complaining of 24 hours of intractable,
The Epidemiology of Clostridium difficile DANIEL SAMAN, DRPH, MPH RESEARCH SCIENTIST ESSENTIA INSTITUTE OF RURAL HEALTH
 The Epidemiology of Clostridium difficile DANIEL SAMAN, DRPH, MPH RESEARCH SCIENTIST ESSENTIA INSTITUTE OF RURAL HEALTH Some history first Clostridium difficile, a spore-forming gram-positive (i.e., thick
The Epidemiology of Clostridium difficile DANIEL SAMAN, DRPH, MPH RESEARCH SCIENTIST ESSENTIA INSTITUTE OF RURAL HEALTH Some history first Clostridium difficile, a spore-forming gram-positive (i.e., thick
Clinical Infectious Diseases Advance Access published December 7, 2012
 Clinical Infectious Diseases Advance Access published December 7, 2012 1 Physician Attitudes Towards the Use of Fecal Transplantation for Recurrent Clostridium Difficile Infection in a Large Metropolitan
Clinical Infectious Diseases Advance Access published December 7, 2012 1 Physician Attitudes Towards the Use of Fecal Transplantation for Recurrent Clostridium Difficile Infection in a Large Metropolitan
Diagnosis, Management, and Prevention of Clostridium difficile infection in Long-Term Care Facilities: A Review
 Diagnosis, Management, and Prevention of Clostridium difficile infection in Long-Term Care Facilities: A Review October 18, 2010 James Kahn and Carolyn Kenney, MSIV Overview Burden of disease associated
Diagnosis, Management, and Prevention of Clostridium difficile infection in Long-Term Care Facilities: A Review October 18, 2010 James Kahn and Carolyn Kenney, MSIV Overview Burden of disease associated
Efficacy of a synbiotic chewable tablet in the prevention of antibiotic-associated diarrhea
 Vol.3, No.2, 110-115 (2011) doi:10.4236/health.2011.32020 Health Efficacy of a synbiotic chewable tablet in the prevention of antibiotic-associated diarrhea Charles Spielholz Nutraceutical Medical Research,
Vol.3, No.2, 110-115 (2011) doi:10.4236/health.2011.32020 Health Efficacy of a synbiotic chewable tablet in the prevention of antibiotic-associated diarrhea Charles Spielholz Nutraceutical Medical Research,
Le infezioni da Clostridium difficile, gravi, ricorrenti e complicate Nicola Petrosillo
 Le infezioni da Clostridium difficile, gravi, ricorrenti e complicate Nicola Petrosillo Istituto Nazionale per le Malattie Infettive «lazzaro Spallanzani», IRCCS-Roma The infectious cycle of transmission
Le infezioni da Clostridium difficile, gravi, ricorrenti e complicate Nicola Petrosillo Istituto Nazionale per le Malattie Infettive «lazzaro Spallanzani», IRCCS-Roma The infectious cycle of transmission
Terapia dell infezione da Clostridium difficile. Massimo Coen I Div Mal Inf AO L Sacco
 Terapia dell infezione da Clostridium difficile Massimo Coen I Div Mal Inf AO L Sacco Disease Severity Mild CDI 3 5 BM/day WBC 15,000/mm 3 Defining CDI Disease Severity Mild abdominal pain due to CDI Moderate
Terapia dell infezione da Clostridium difficile Massimo Coen I Div Mal Inf AO L Sacco Disease Severity Mild CDI 3 5 BM/day WBC 15,000/mm 3 Defining CDI Disease Severity Mild abdominal pain due to CDI Moderate
Long-Term Care Updates
 Long-Term Care Updates April 2018 By Austin Smith, PharmD Candidate and Lindsay Slowiczek, PharmD is the most common healthcare-acquired infection (HAI) in the United States. 1,2 A 2014 prevalence survey
Long-Term Care Updates April 2018 By Austin Smith, PharmD Candidate and Lindsay Slowiczek, PharmD is the most common healthcare-acquired infection (HAI) in the United States. 1,2 A 2014 prevalence survey
Human monoclonal antibodies against Clostridium difficile toxin A and B inhibit inflammatory
 AAC Accepts, published online ahead of print on 29 April 213 Antimicrob. Agents Chemother. doi:1.1128/aac.2633-12 Copyright 213, American Society for Microbiology. All Rights Reserved. Anti-inflammatory
AAC Accepts, published online ahead of print on 29 April 213 Antimicrob. Agents Chemother. doi:1.1128/aac.2633-12 Copyright 213, American Society for Microbiology. All Rights Reserved. Anti-inflammatory
complicating inflammatory bowel disease
 Gut, 1982, 23, 410-414 Clostridium difficile toxin in acute diarrhoea complicating inflammatory bowel disease M R B KEIGHLEY*, DENISE YOUNGS, MARGARET JOHNSON, R N ALLAN, and D W BURDON From The General
Gut, 1982, 23, 410-414 Clostridium difficile toxin in acute diarrhoea complicating inflammatory bowel disease M R B KEIGHLEY*, DENISE YOUNGS, MARGARET JOHNSON, R N ALLAN, and D W BURDON From The General
Probiotics for Primary Prevention of Clostridium difficile Infection
 Probiotics for Primary Prevention of Clostridium difficile Infection Objectives Review risk factors for Clostridium difficile infection (CDI) Describe guideline recommendations for CDI prevention Discuss
Probiotics for Primary Prevention of Clostridium difficile Infection Objectives Review risk factors for Clostridium difficile infection (CDI) Describe guideline recommendations for CDI prevention Discuss
Updates to pharmacological management in the prevention of recurrent Clostridium difficile
 Updates to pharmacological management in the prevention of recurrent Clostridium difficile Julia Shlensky, PharmD PGY2 Internal Medicine Resident September 12, 2017 2017 MFMER slide-1 Clinical Impact Increasing
Updates to pharmacological management in the prevention of recurrent Clostridium difficile Julia Shlensky, PharmD PGY2 Internal Medicine Resident September 12, 2017 2017 MFMER slide-1 Clinical Impact Increasing
Clostridium difficile causes to cases of diarrhea and colitis in the United
 Clostridium difficile Associated Diarrhea A Review REVIEW ARTICLE Eleftherios Mylonakis, MD; Edward T. Ryan, MD; Stephen B. Calderwood, MD Clostridium difficile causes 300000 to 3000000 cases of diarrhea
Clostridium difficile Associated Diarrhea A Review REVIEW ARTICLE Eleftherios Mylonakis, MD; Edward T. Ryan, MD; Stephen B. Calderwood, MD Clostridium difficile causes 300000 to 3000000 cases of diarrhea
In 1935, Hall and O Toole first isolated a gram-positive, cytotoxinproducing
 The new england journal of medicine review article Current Concepts Clostridium difficile More Difficult Than Ever Ciarán P. Kelly, M.D., and J. Thomas LaMont, M.D. From the Gastroenterology Division,
The new england journal of medicine review article Current Concepts Clostridium difficile More Difficult Than Ever Ciarán P. Kelly, M.D., and J. Thomas LaMont, M.D. From the Gastroenterology Division,
ACG Clinical Guideline: Diagnosis, Treatment, and Prevention of Clostridium difficile Infections
 ACG Clinical Guideline: Diagnosis, Treatment, and Prevention of Clostridium difficile Infections Christina M. Surawicz, MD 1, Lawrence J. Brandt, MD 2, David G. Binion, MD 3, Ashwin N. Ananthakrishnan,
ACG Clinical Guideline: Diagnosis, Treatment, and Prevention of Clostridium difficile Infections Christina M. Surawicz, MD 1, Lawrence J. Brandt, MD 2, David G. Binion, MD 3, Ashwin N. Ananthakrishnan,
Fecal Microbiota Transplantation in C. diff. colitis Benefits and Limitations
 January 27th 2017, 8th Gastro Foundation Weekend for Fellows; Spier Hotel & Conference Centre, Stellenbosch Fecal Microbiota Transplantation in C. diff. colitis Benefits and Limitations Gerhard Rogler,
January 27th 2017, 8th Gastro Foundation Weekend for Fellows; Spier Hotel & Conference Centre, Stellenbosch Fecal Microbiota Transplantation in C. diff. colitis Benefits and Limitations Gerhard Rogler,
March 3, To: Hospitals, Long Term Care Facilities, and Local Health Departments
 March 3, 2010 To: Hospitals, Long Term Care Facilities, and Local Health Departments From: NYSDOH Bureau of Healthcare Associated Infections HEALTH ADVISORY: GUIDANCE FOR PREVENTION AND CONTROL OF HEALTHCARE
March 3, 2010 To: Hospitals, Long Term Care Facilities, and Local Health Departments From: NYSDOH Bureau of Healthcare Associated Infections HEALTH ADVISORY: GUIDANCE FOR PREVENTION AND CONTROL OF HEALTHCARE
CLOSTRIDIUM DIFICILE. Negin N Blattman Infectious Diseases Phoenix VA Healthcare System
 CLOSTRIDIUM DIFICILE Negin N Blattman Infectious Diseases Phoenix VA Healthcare System ANTIBIOTIC ASSOCIATED DIARRHEA 1978: C diff first identified 1989-1992: Four large outbreaks in the US caused by J
CLOSTRIDIUM DIFICILE Negin N Blattman Infectious Diseases Phoenix VA Healthcare System ANTIBIOTIC ASSOCIATED DIARRHEA 1978: C diff first identified 1989-1992: Four large outbreaks in the US caused by J
Long-Term Care Updates
 Long-Term Care Updates April 2017 Bezlotoxumab to Prevent Recurrent Infection By Amy Wilson, PharmD and Zara Risoldi Cochrane, PharmD, MS, FASCP Introduction The Gram-positive bacteria is a common cause
Long-Term Care Updates April 2017 Bezlotoxumab to Prevent Recurrent Infection By Amy Wilson, PharmD and Zara Risoldi Cochrane, PharmD, MS, FASCP Introduction The Gram-positive bacteria is a common cause
Stony Brook Adult Clostridium difficile Management Guidelines. Discontinue all unnecessary antibiotics
 Stony Brook Adult Clostridium difficile Management Guidelines Summary: Use of the C Diff Infection (CDI) PowerPlan (Adult) Required Patient with clinical findings suggestive of Clostridium difficile infection
Stony Brook Adult Clostridium difficile Management Guidelines Summary: Use of the C Diff Infection (CDI) PowerPlan (Adult) Required Patient with clinical findings suggestive of Clostridium difficile infection
Atypical Presentation of Clostridium Difficille Infection (CDI).
 Article ID: WMC004648 ISSN 2046-1690 Atypical Presentation of Clostridium Difficille Infection (CDI). Peer review status: No Corresponding Author: Dr. Syed A Gardezi, CT1, Medicine,NevillHall Hospital
Article ID: WMC004648 ISSN 2046-1690 Atypical Presentation of Clostridium Difficille Infection (CDI). Peer review status: No Corresponding Author: Dr. Syed A Gardezi, CT1, Medicine,NevillHall Hospital
Zinplava. (bezlotoxumab) New Product Slideshow
 Zinplava (bezlotoxumab) New Product Slideshow Introduction Brand name: Zinplava Generic name: Bezlotoxumab Pharmacological class: Human IgG1 monoclonal antibody Strength and Formulation: 25mg/mL; solution
Zinplava (bezlotoxumab) New Product Slideshow Introduction Brand name: Zinplava Generic name: Bezlotoxumab Pharmacological class: Human IgG1 monoclonal antibody Strength and Formulation: 25mg/mL; solution
Update on Clostridium difficile infection.
 Update on Clostridium difficile infection. K. Honein Gastroenterologist, HDF Associate Professor Head of Medicine Department St Joseph University-Beirut. Introduction Gram+anaerobic bacillus responsible
Update on Clostridium difficile infection. K. Honein Gastroenterologist, HDF Associate Professor Head of Medicine Department St Joseph University-Beirut. Introduction Gram+anaerobic bacillus responsible
Clostridium Difficile Infection: Applying New Treatment Guidelines and Strategies to Reduce Recurrence Rate
 Clostridium Difficile Infection: Applying New Treatment Guidelines and Strategies to Reduce Recurrence Rate Objectives Summarize the changing epidemiology and demographics of patients at risk for Clostridium
Clostridium Difficile Infection: Applying New Treatment Guidelines and Strategies to Reduce Recurrence Rate Objectives Summarize the changing epidemiology and demographics of patients at risk for Clostridium
Modern approach to Clostridium Difficile Infection
 Modern approach to Clostridium Difficile Infection Pseudomembranous Colitis: Principles for diagnosis and treatment Aggelos Stefos Internist, Infectious diseases Specialist Department of Medicine and Research
Modern approach to Clostridium Difficile Infection Pseudomembranous Colitis: Principles for diagnosis and treatment Aggelos Stefos Internist, Infectious diseases Specialist Department of Medicine and Research
Homeopathic Products. Evidence??
 Homeopathic Products Principle of analogy or Law of Similars Small or infinitesimal doses (3X-30C) Avogadro s number=6x10 23 = ~23X Succussion and potentization (see http://www.boiron.com/en/htm/02_medi_hom
Homeopathic Products Principle of analogy or Law of Similars Small or infinitesimal doses (3X-30C) Avogadro s number=6x10 23 = ~23X Succussion and potentization (see http://www.boiron.com/en/htm/02_medi_hom
! Macrolide antibacterial. Fidaxomicin (Dificid ) package labeling. Optimer Pharmaceuticals, Inc. May 2011.
 Disclosure! I have no conflicts of interest related to this presentation Nina Naeger Murphy, Pharm.D., BCPS Clinical Pharmacy Specialist Infectious Diseases MetroHealth Medical Center Learning Objectives!
Disclosure! I have no conflicts of interest related to this presentation Nina Naeger Murphy, Pharm.D., BCPS Clinical Pharmacy Specialist Infectious Diseases MetroHealth Medical Center Learning Objectives!
Updated Clostridium difficile Treatment Guidelines
 Updated Clostridium difficile Treatment Guidelines Arielle Arnold, PharmD, BCPS Clinical Pharmacist Saint Alphonsus Regional Medical Center September 29 th, 2018 Disclosures Nothing to disclose Learning
Updated Clostridium difficile Treatment Guidelines Arielle Arnold, PharmD, BCPS Clinical Pharmacist Saint Alphonsus Regional Medical Center September 29 th, 2018 Disclosures Nothing to disclose Learning
Factors associated with prolonged symptoms and severe disease due to Clostridium difficile
 Age and Ageing 1999; 28: 107 113 Factors associated with prolonged symptoms and severe disease due to Clostridium difficile LORRAINE KYNE, CONCEPTA MERRY 2,BRIAN O CONNELL 2,ALAN KELLY 3,CONOR KEANE 2,
Age and Ageing 1999; 28: 107 113 Factors associated with prolonged symptoms and severe disease due to Clostridium difficile LORRAINE KYNE, CONCEPTA MERRY 2,BRIAN O CONNELL 2,ALAN KELLY 3,CONOR KEANE 2,
Probiotics: Their Role in Medicine Today. Objectives. Probiotics: What Are They? 11/3/2017
 Probiotics: Their Role in Medicine Today Viki Barr Pharm.D., BCPS AQ ID Assistant Professor, Pharmacy Practice Rosalind Franklin University of Medicine and Science Clinical Pharmacist, Infectious Diseases
Probiotics: Their Role in Medicine Today Viki Barr Pharm.D., BCPS AQ ID Assistant Professor, Pharmacy Practice Rosalind Franklin University of Medicine and Science Clinical Pharmacist, Infectious Diseases
Clostridium Difficile Infection in Adults Treatment and Prevention
 Clostridium Difficile Infection in Adults Treatment and Prevention Definition: Clostridium Difficile colonizes the human intestinal tract after the normal gut flora has been altered by antibiotic therapy
Clostridium Difficile Infection in Adults Treatment and Prevention Definition: Clostridium Difficile colonizes the human intestinal tract after the normal gut flora has been altered by antibiotic therapy
SMT19969: A Selective Therapy for C. difficile Infection
 SMT19969: A Selective Therapy for C. difficile Infection One Bug, One Drug 25 th September 2012 SMT19969: A Selective Therapy for CDI SMT19969 is a novel antibiotic for the specific treatment of Clostridium
SMT19969: A Selective Therapy for C. difficile Infection One Bug, One Drug 25 th September 2012 SMT19969: A Selective Therapy for CDI SMT19969 is a novel antibiotic for the specific treatment of Clostridium
Clostridium Difficile Associated Disease. Edmund Krasinski, Jr., D.O., F.A.C.G. Southwest Conference on Medicine 2011
 Clostridium Difficile Associated Disease Edmund Krasinski, Jr., D.O., F.A.C.G. Southwest Conference on Medicine 2011 Introduction Which of the following is more common in community hospitals in the Southeast
Clostridium Difficile Associated Disease Edmund Krasinski, Jr., D.O., F.A.C.G. Southwest Conference on Medicine 2011 Introduction Which of the following is more common in community hospitals in the Southeast
Antibiotic Associated Diarrhea (AAD) and Clostridium Difficile Infection (CDI)
 Antibiotic Associated Diarrhea (AAD) and Clostridium Difficile Infection (CDI) Dario Conte, M.D. Gastroenterology and Endoscopy Unit Postgraduate School of Gastroenterology Fondazione IRCCS Ca Granda Ospedale
Antibiotic Associated Diarrhea (AAD) and Clostridium Difficile Infection (CDI) Dario Conte, M.D. Gastroenterology and Endoscopy Unit Postgraduate School of Gastroenterology Fondazione IRCCS Ca Granda Ospedale
T he bacterium Clostridium difficile is the leading infectious
 973 ORIGINAL ARTICLE Colonic IgA producing cells and macrophages are reduced in recurrent and non-recurrent Clostridium difficile associated diarrhoea S S Johal, C P Lambert, J Hammond, P D James, S P
973 ORIGINAL ARTICLE Colonic IgA producing cells and macrophages are reduced in recurrent and non-recurrent Clostridium difficile associated diarrhoea S S Johal, C P Lambert, J Hammond, P D James, S P
Clostridium difficile Infection (CDI) Management Guideline
 Clostridium difficile Infection (CDI) Management Guideline Do not test all patients with loose or watery stools for CDI o CDI is responsible for
Clostridium difficile Infection (CDI) Management Guideline Do not test all patients with loose or watery stools for CDI o CDI is responsible for
Bezlotoxumab (Zinplava) as Adjunct Treatment for Clostridium difficile. Janel Liane Cala, RPh Medical Center Hospital
 Bezlotoxumab (Zinplava) as Adjunct Treatment for Clostridium difficile Janel Liane Cala, RPh Medical Center Hospital Objectives Review pathophysiology, risk factors, prevention, and treatment options of
Bezlotoxumab (Zinplava) as Adjunct Treatment for Clostridium difficile Janel Liane Cala, RPh Medical Center Hospital Objectives Review pathophysiology, risk factors, prevention, and treatment options of
Learning Goals. Clostridium difficile. Historical Context. Historical Context 6/27/2012
 Learning Goals Clostridium difficile Justin L. Sewell, MD, MPH Assistant Clinical Professor of Medicine University of California San Francisco San Francisco General Hospital Understand the epidemiology,
Learning Goals Clostridium difficile Justin L. Sewell, MD, MPH Assistant Clinical Professor of Medicine University of California San Francisco San Francisco General Hospital Understand the epidemiology,
DETECTION OF TOXIGENIC CLOSTRIDIUM DIFFICILE
 CLINICAL GUIDELINES For use with the UnitedHealthcare Laboratory Benefit Management Program, administered by BeaconLBS DETECTION OF TOXIGENIC CLOSTRIDIUM DIFFICILE Policy Number: PDS 021 Effective Date:
CLINICAL GUIDELINES For use with the UnitedHealthcare Laboratory Benefit Management Program, administered by BeaconLBS DETECTION OF TOXIGENIC CLOSTRIDIUM DIFFICILE Policy Number: PDS 021 Effective Date:
Los Angeles County Department of Public Health: Your Partner in CDI Prevention
 Los Angeles County Department of Public Health: Your Partner in CDI Prevention Dawn Terashita, MD, MPH Acute Communicable Disease Control Los Angeles County Department of Public Health dterashita@ph.lacounty.gov
Los Angeles County Department of Public Health: Your Partner in CDI Prevention Dawn Terashita, MD, MPH Acute Communicable Disease Control Los Angeles County Department of Public Health dterashita@ph.lacounty.gov
Clostridium difficile toxin A
 9969 JClin Pathol 1997;50:996-1 000 Department of Pathology, Brigham and Women's Hospital, Harvard Medical School, 75 Francis Street, Boston, MA 02115, USA S 0 Vargas D Horensky A B Onderdonk Correspondence
9969 JClin Pathol 1997;50:996-1 000 Department of Pathology, Brigham and Women's Hospital, Harvard Medical School, 75 Francis Street, Boston, MA 02115, USA S 0 Vargas D Horensky A B Onderdonk Correspondence
Study on Incidence of Antibiotic Associated Diarrhoea in General Paediatric Ward
 HK J Paediatr (new series) 2002;7:33-38 Study on Incidence of Antibiotic Associated Diarrhoea in General Paediatric Ward CM HUI, K TSE Abstract Objective: To estimate the incidence rate and risk factors
HK J Paediatr (new series) 2002;7:33-38 Study on Incidence of Antibiotic Associated Diarrhoea in General Paediatric Ward CM HUI, K TSE Abstract Objective: To estimate the incidence rate and risk factors
Title: Fecal microbiota transplantation in recurrent Clostridium difficile infection in a patient with concomitant inflammatory bowel disease
 Title: Fecal microbiota transplantation in recurrent Clostridium difficile infection in a patient with concomitant inflammatory bowel disease Authors: Marta Gravito-Soares, Elisa Gravito-Soares, Francisco
Title: Fecal microbiota transplantation in recurrent Clostridium difficile infection in a patient with concomitant inflammatory bowel disease Authors: Marta Gravito-Soares, Elisa Gravito-Soares, Francisco
-2002: Rectal blood loss, UC? (no definite diagnosis) rectal mesalazine. -June 2008: Recurrence of rectal blood loss and urgency
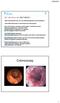 SD, male 40 yrs. old. (680718M467.) -2002: Rectal blood loss, UC? (no definite diagnosis) rectal mesalazine -June 2008: Recurrence of rectal blood loss and urgency Total colonoscopy: ulcerative rectitis,
SD, male 40 yrs. old. (680718M467.) -2002: Rectal blood loss, UC? (no definite diagnosis) rectal mesalazine -June 2008: Recurrence of rectal blood loss and urgency Total colonoscopy: ulcerative rectitis,
OCTOBER 7-10 PHILADELPHIA, PENNSYLVANIA
 OMED 17 OCTOBER 7-10 PHILADELPHIA, PENNSYLVANIA 29.5 Category 1-A CME credits anticipated ACOFP / AOA s 122 nd Annual Osteopathic Medical Conference & Exposition Joint Session with ACOFP and Cleveland
OMED 17 OCTOBER 7-10 PHILADELPHIA, PENNSYLVANIA 29.5 Category 1-A CME credits anticipated ACOFP / AOA s 122 nd Annual Osteopathic Medical Conference & Exposition Joint Session with ACOFP and Cleveland
Overview of Clostridium difficile infection: implications for China
 Overview of Clostridium difficile infection: implications for China The Harvard community has made this article openly available. Please share how this access benefits you. Your story matters. Citation
Overview of Clostridium difficile infection: implications for China The Harvard community has made this article openly available. Please share how this access benefits you. Your story matters. Citation
L. Clifford McDonald, MD. Senior Advisor for Science and Integrity September 16, 2015
 Controversies and Current Issues in Diagnosis, Surveillance, and Treatment of Clostridium difficile infeciton L. Clifford McDonald, MD Senior Advisor for Science and Integrity September 16, 2015 Division
Controversies and Current Issues in Diagnosis, Surveillance, and Treatment of Clostridium difficile infeciton L. Clifford McDonald, MD Senior Advisor for Science and Integrity September 16, 2015 Division
Long-Term Clinical Outcome of Clostridium difficile Infection in Hospitalized Patients: A Single Center Study
 ORIGINAL ARTICLE ISSN 1598-9100(Print) ISSN 2288-1956(Online) http://dx.doi.org/10.5217/ir.2014.12.4.299 Intest Res 2014;12(4):299-305 Long-Term Clinical Outcome of Clostridium difficile Infection in Hospitalized
ORIGINAL ARTICLE ISSN 1598-9100(Print) ISSN 2288-1956(Online) http://dx.doi.org/10.5217/ir.2014.12.4.299 Intest Res 2014;12(4):299-305 Long-Term Clinical Outcome of Clostridium difficile Infection in Hospitalized
Gastroenteritis and viral infections
 Gastroenteritis and viral infections A Large number of viruses are found in the human gut; these include some that are associated with gastroenteritis Rotaviruses Adenoviruses 40/41 Caliciviruses Norwalk-like
Gastroenteritis and viral infections A Large number of viruses are found in the human gut; these include some that are associated with gastroenteritis Rotaviruses Adenoviruses 40/41 Caliciviruses Norwalk-like
L infezione da Clostridium difficile (CDI) Quadri clinici e nuovi approcci terapeutici
 L infezione da Clostridium difficile (CDI) Quadri clinici e nuovi approcci terapeutici Roberto Luzzati SC Malattie Infettive, AOU Trieste Presidente :Prof. Enzo Raise Clinical presentation of infection
L infezione da Clostridium difficile (CDI) Quadri clinici e nuovi approcci terapeutici Roberto Luzzati SC Malattie Infettive, AOU Trieste Presidente :Prof. Enzo Raise Clinical presentation of infection
John G. Bartlett: Contributions to the Discovery of Clostridium difficile Antibiotic-Associated Diarrhea
 SUPPLEMENT ARTICLE John G. Bartlett: Contributions to the Discovery of Clostridium difficile Antibiotic-Associated Diarrhea Sherwood L. Gorbach Cubist Pharmaceuticals, Inc, and Department of Public Health
SUPPLEMENT ARTICLE John G. Bartlett: Contributions to the Discovery of Clostridium difficile Antibiotic-Associated Diarrhea Sherwood L. Gorbach Cubist Pharmaceuticals, Inc, and Department of Public Health
Introduction ORIGINAL ARTICLE 1
 ORIGINAL ARTICLE 1 Laboratory diagnosis of Clostridium difficile infection. An evaluation of tests for faecal toxin, glutamate dehydrogenase, lactoferrin and toxigenic culture in the diagnostic laboratory
ORIGINAL ARTICLE 1 Laboratory diagnosis of Clostridium difficile infection. An evaluation of tests for faecal toxin, glutamate dehydrogenase, lactoferrin and toxigenic culture in the diagnostic laboratory
AN OUTBREAK OF TOXIN A NEGATIVE, TOXIN B POSITIVE CLOSTRIDIUM DIFFICILE -ASSOCIATED DIARRHEA IN A CANADIAN TERTIARY-CARE HOSPITAL
 Vol. 25-7 Date of publication: 1 April 1999 Contained in this FAX issue: (No. of pages: 6) Official page numbers: AN OUTBREAK OF TOXIN A NEGATIVE, TOXIN B POSITIVE CLOSTRIDIUM DIFFICILE-ASSOCIATED DIARRHEA
Vol. 25-7 Date of publication: 1 April 1999 Contained in this FAX issue: (No. of pages: 6) Official page numbers: AN OUTBREAK OF TOXIN A NEGATIVE, TOXIN B POSITIVE CLOSTRIDIUM DIFFICILE-ASSOCIATED DIARRHEA
Hospital-acquired diarrhoea in elderly patients: epidemiology and staff awareness
 Age and Ageing 1998; 27: 339-343 Hospital-acquired diarrhoea in elderly patients: epidemiology and staff awareness LORRAINE KYNE, AIDEEN MORAN, CONOR KEANE 1, DESMOND O'NEILL Age-Related Health Cane, Meath
Age and Ageing 1998; 27: 339-343 Hospital-acquired diarrhoea in elderly patients: epidemiology and staff awareness LORRAINE KYNE, AIDEEN MORAN, CONOR KEANE 1, DESMOND O'NEILL Age-Related Health Cane, Meath
more intense treatments are needed to get rid of the infection.
 What Is Clostridium Difficile (C. Diff)? Clostridium difficile, or C. diff for short, is an infection from a bacterium that can grow in your intestines and cause bad GI symptoms. The main risk of getting
What Is Clostridium Difficile (C. Diff)? Clostridium difficile, or C. diff for short, is an infection from a bacterium that can grow in your intestines and cause bad GI symptoms. The main risk of getting
ESCMID Online Lecture Library. by author
 ECDIS-NET: Update on Clostridium difficile epidemiology in Europe 1 E d J. K u i j p e r, S o f i e v a n D o r p a n d D a a n N o t e r m a n s. D e p a r t m e n t o f M e d i c a l M i c r o b i o
ECDIS-NET: Update on Clostridium difficile epidemiology in Europe 1 E d J. K u i j p e r, S o f i e v a n D o r p a n d D a a n N o t e r m a n s. D e p a r t m e n t o f M e d i c a l M i c r o b i o
! MQ is a 44 year old woman that I first saw in Sept ! In MVA in Jan 2003 requiring spinal surgery
 Case MQ is a 44 year old woman that I first saw in Sept 2006 UPDATE ON CLOSTRIDIUM DIFFICILE DISEASE Richard A. Jacobs, M.D.,PhD In MVA in Jan 2003 requiring spinal surgery Subsequently developed fecal
Case MQ is a 44 year old woman that I first saw in Sept 2006 UPDATE ON CLOSTRIDIUM DIFFICILE DISEASE Richard A. Jacobs, M.D.,PhD In MVA in Jan 2003 requiring spinal surgery Subsequently developed fecal
Study summaries L. casei 431
 This binder provides you with summaries of selected publications on Lactobacillus paracasei subsp. paracasei L. casei 431. The publications are clinical studies performed in humans documenting the effects
This binder provides you with summaries of selected publications on Lactobacillus paracasei subsp. paracasei L. casei 431. The publications are clinical studies performed in humans documenting the effects
Review. Treatment of Clostridium difficile-associated disease: old therapies and new strategies. Introduction
 Treatment of Clostridium difficile-associated disease: old therapies and new strategies Saima Aslam, Richard J Hamill, Daniel M Musher Clostridium difficile-associated disease (CDAD) causes substantial
Treatment of Clostridium difficile-associated disease: old therapies and new strategies Saima Aslam, Richard J Hamill, Daniel M Musher Clostridium difficile-associated disease (CDAD) causes substantial
Medical Virology Immunology. Dr. Sameer Naji, MB, BCh, PhD (UK) Head of Basic Medical Sciences Dept. Faculty of Medicine The Hashemite University
 Medical Virology Immunology Dr. Sameer Naji, MB, BCh, PhD (UK) Head of Basic Medical Sciences Dept. Faculty of Medicine The Hashemite University Human blood cells Phases of immune responses Microbe Naïve
Medical Virology Immunology Dr. Sameer Naji, MB, BCh, PhD (UK) Head of Basic Medical Sciences Dept. Faculty of Medicine The Hashemite University Human blood cells Phases of immune responses Microbe Naïve
The incubation period is unknown. However; the onset of clinical disease is typically 5-10 days after initiation of antimicrobial treatment.
 C. DIFFICILE Case definition CONFIRMED CASE A patient is defined as a case if they are one year of age or older AND have one of the following requirements: A laboratory confirmation of a positive toxin
C. DIFFICILE Case definition CONFIRMED CASE A patient is defined as a case if they are one year of age or older AND have one of the following requirements: A laboratory confirmation of a positive toxin
DISCLOSURE Relevant relationships with commercial entities Wyeth (received advisory board & speaker honoraria) Potential for conflicts of interest wit
 GASTROENTERITIS DISCLOSURE Relevant relationships with commercial entities Wyeth (received advisory board & speaker honoraria) Potential for conflicts of interest within this presentation fidaxomicin (which
GASTROENTERITIS DISCLOSURE Relevant relationships with commercial entities Wyeth (received advisory board & speaker honoraria) Potential for conflicts of interest within this presentation fidaxomicin (which
Clostridium difficile infection (CDI) Week 52 (Ending 30/12/2017)
 Clostridium difficile infection (CDI) Week 52 (Ending 30/12/2017) What is Clostridium difficile? Clostridium difficile is a Gram-positive anaerobic spore forming bacillus. It is ubiquitous in nature and
Clostridium difficile infection (CDI) Week 52 (Ending 30/12/2017) What is Clostridium difficile? Clostridium difficile is a Gram-positive anaerobic spore forming bacillus. It is ubiquitous in nature and
E. Histolytica IgG (Amebiasis)
 DIAGNOSTIC AUTOMATION, INC. 21250 Califa Street, Suite 102 and 116, Woodland hills, CA 91367 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com
DIAGNOSTIC AUTOMATION, INC. 21250 Califa Street, Suite 102 and 116, Woodland hills, CA 91367 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com
Clostridium difficile Not Just a Hospital Problem Anymore. Objectives. The C. difficile Carrier State. Conflicts of Interest Disclosure
 Conflicts of Interest Disclosure Daniel Leffler, MD, MS I disclose the following financial relationships with commercial entities that produce health carerelated products or services relevant to the content
Conflicts of Interest Disclosure Daniel Leffler, MD, MS I disclose the following financial relationships with commercial entities that produce health carerelated products or services relevant to the content
Patient presentation
 Update: Clostridium difficile Colitis David H. Kerman, MD Assistant Professor of Clinical Medicine Director, Fellowship Program Division of Gastroenterology University of Miami Miller School of Medicine
Update: Clostridium difficile Colitis David H. Kerman, MD Assistant Professor of Clinical Medicine Director, Fellowship Program Division of Gastroenterology University of Miami Miller School of Medicine
Objectives Clostridium difficile Infections, So Many Tests, Which One to Choose?
 Objectives Clostridium difficile Infections, So Many Tests, Which One to Choose? March 9, 0 http://www.slh.wisc.edu/outreach-data/event-detail.php?id=03 Raymond P. Podzorski, Ph.D., D(ABMM) Clinical Microbiologist
Objectives Clostridium difficile Infections, So Many Tests, Which One to Choose? March 9, 0 http://www.slh.wisc.edu/outreach-data/event-detail.php?id=03 Raymond P. Podzorski, Ph.D., D(ABMM) Clinical Microbiologist
Star Articles in Review
 Star Articles in Review CDDW/CASL Meeting Toronto, February 10, 2014 Christina M. Surawicz, MD MACG Professor of Medicine Division of Gastroenterology Department of Medicine University of Washington Disclosure
Star Articles in Review CDDW/CASL Meeting Toronto, February 10, 2014 Christina M. Surawicz, MD MACG Professor of Medicine Division of Gastroenterology Department of Medicine University of Washington Disclosure
Xifaxan. Xifaxan (rifaximin) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.01.34 Subject: Xifaxan Page: 1 of 6 Last Review Date: December 8, 2017 Xifaxan Description Xifaxan (rifaximin)
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.01.34 Subject: Xifaxan Page: 1 of 6 Last Review Date: December 8, 2017 Xifaxan Description Xifaxan (rifaximin)
Antibiotic use, gastroenteritis and respiratory illness in South Australian children
 Epidemiol. Infect. (2002), 129, 507 513. f 2002 Cambridge University Press DOI: 10.1017/S0950268802007628 Printed in the United Kingdom Antibiotic use, gastroenteritis and respiratory illness in South
Epidemiol. Infect. (2002), 129, 507 513. f 2002 Cambridge University Press DOI: 10.1017/S0950268802007628 Printed in the United Kingdom Antibiotic use, gastroenteritis and respiratory illness in South
12/14/2009. Diagnostic considerations: C. difficile in IBD
 Diagnosis and treatment of Gastrointestinal Tract Infections in IBD Case Studies Leonard Baidoo,MD David Binion,MD University of Pittsburgh Medical Center Pittsburgh, Pennsylvania Case 1 46yr old man,1
Diagnosis and treatment of Gastrointestinal Tract Infections in IBD Case Studies Leonard Baidoo,MD David Binion,MD University of Pittsburgh Medical Center Pittsburgh, Pennsylvania Case 1 46yr old man,1
9/18/2018. Clostridium Difficile: Updates on Diagnosis and Treatment. Clostridium difficile Infection (CDI) Clostridium difficile Infection (CDI)
 Clostridium Difficile: Updates on Diagnosis and Treatment Elizabeth Hudson, DO, MPH 9/25/18 Antibiotic-associated diarrhea and colitis were well established soon after widespread use of antibiotics In
Clostridium Difficile: Updates on Diagnosis and Treatment Elizabeth Hudson, DO, MPH 9/25/18 Antibiotic-associated diarrhea and colitis were well established soon after widespread use of antibiotics In
C. difficile Infection: How it all comes out
 C. difficile Infection: How it all comes out Larry Danziger, Pharm.D. Professor of Pharmacy and Medicine College of Pharmacy University of Illinois at Chicago The speaker has no conflicts to disclose.
C. difficile Infection: How it all comes out Larry Danziger, Pharm.D. Professor of Pharmacy and Medicine College of Pharmacy University of Illinois at Chicago The speaker has no conflicts to disclose.
Fecal microbiota transplantation: Breaking the chain of recurrent C. difficile infection
 Fecal microbiota transplantation: Breaking the chain of recurrent C. difficile infection Issue Date: June 2013 Vol. 8 No. 6 Author: Amy Marinski, MSN, RN, CCRN, CNL More than 3 million new cases of Clostridium
Fecal microbiota transplantation: Breaking the chain of recurrent C. difficile infection Issue Date: June 2013 Vol. 8 No. 6 Author: Amy Marinski, MSN, RN, CCRN, CNL More than 3 million new cases of Clostridium
Sherwood L. Gorbach, MD Professor of Public Health, Medicine, and Microbiology Tufts University School of Medicine
 Sherwood L. Gorbach, MD Professor of Public Health, Medicine, and Microbiology Tufts University School of Medicine Chief Scientific Officer, Optimer Pharmaceuticals, Inc. Conflicts: Chief Scientific Officer,
Sherwood L. Gorbach, MD Professor of Public Health, Medicine, and Microbiology Tufts University School of Medicine Chief Scientific Officer, Optimer Pharmaceuticals, Inc. Conflicts: Chief Scientific Officer,
Clostridium difficile Essential information
 Clostridium difficile Essential information Clostridium difficile Origins Clostridium difficile (C. diff) is a Gram positive, spore forming, anaerobic bacterium with a rod structure. It was first identified
Clostridium difficile Essential information Clostridium difficile Origins Clostridium difficile (C. diff) is a Gram positive, spore forming, anaerobic bacterium with a rod structure. It was first identified
GUIDELINE FOR THE MANAGEMENT OF ANTIBIOTIC- ASSOCIATED DIARRHOEA IN ADULTS
 GUIDELINE FOR THE MANAGEMENT OF ANTIBIOTIC- ASSOCIATED DIARRHOEA IN ADULTS Version 3.0 Date ratified May 2008 Review date May 2010 Ratified by NUH Antibiotic Guidelines Committee NUH Drugs and Therapeutics
GUIDELINE FOR THE MANAGEMENT OF ANTIBIOTIC- ASSOCIATED DIARRHOEA IN ADULTS Version 3.0 Date ratified May 2008 Review date May 2010 Ratified by NUH Antibiotic Guidelines Committee NUH Drugs and Therapeutics
Clostridium difficile
 Clostridium difficile Alex Aspinall MD, PhD, FRCPC Clinical Assistant Professor, University of Calgary Division of Gastroenterology and Hepatology, South Health Campus www.seacourses.com 1 Learning Points
Clostridium difficile Alex Aspinall MD, PhD, FRCPC Clinical Assistant Professor, University of Calgary Division of Gastroenterology and Hepatology, South Health Campus www.seacourses.com 1 Learning Points
Safety of Saccharomyces boulardii (Florastor) in Solid Organ Transplant Patients
 Butler University Digital Commons @ Butler University Undergraduate Honors Thesis Collection Undergraduate Scholarship 5-12-2012 Safety of Saccharomyces boulardii (Florastor) in Solid Organ Transplant
Butler University Digital Commons @ Butler University Undergraduate Honors Thesis Collection Undergraduate Scholarship 5-12-2012 Safety of Saccharomyces boulardii (Florastor) in Solid Organ Transplant
Does Extending Clostridium Difficile Treatment In Patients Who Are Receiving Concomitant Antibiotics Reduce The Rate Of Relapse?
 ISPUB.COM The Internet Journal of Infectious Diseases Volume 15 Number 1 Does Extending Clostridium Difficile Treatment In Patients Who Are Receiving Concomitant Antibiotics Reduce The Rate Of Relapse?
ISPUB.COM The Internet Journal of Infectious Diseases Volume 15 Number 1 Does Extending Clostridium Difficile Treatment In Patients Who Are Receiving Concomitant Antibiotics Reduce The Rate Of Relapse?
were obtained from the American Type Culture Collection obtained from the National Collection of Type Cultures,
 JOURNAL OF CLINICAL MICROBIOLOGY, Mar. 1985, p. 323-327 0095-1137/85/030323-05$02.00/0 Copyright ( 1985, American Society for Microbiology Vol. 21, No. 3 Serogrouping of Clostridium difficile Strains by
JOURNAL OF CLINICAL MICROBIOLOGY, Mar. 1985, p. 323-327 0095-1137/85/030323-05$02.00/0 Copyright ( 1985, American Society for Microbiology Vol. 21, No. 3 Serogrouping of Clostridium difficile Strains by
Managing Clostridium Difficile: An Old Bug With
 932 The Red Section see related editorial on page x Managing Clostridium Difficile: An Old Bug With New Tricks Stephen M. Vindigni, MD, MPH 1,2 and Christina M. Surawicz, MD 1 Am J Gastroenterol (2018)
932 The Red Section see related editorial on page x Managing Clostridium Difficile: An Old Bug With New Tricks Stephen M. Vindigni, MD, MPH 1,2 and Christina M. Surawicz, MD 1 Am J Gastroenterol (2018)
Fecal transplantation as a treatment option for recurrent Clostridium difficile infection
 Fecal transplantation as a treatment option for recurrent Clostridium difficile infection Josbert Keller Department of Gastroenterology Haga Teaching Hospital, The Hague Case: 81 yrs, CVA, recurrent UTI,
Fecal transplantation as a treatment option for recurrent Clostridium difficile infection Josbert Keller Department of Gastroenterology Haga Teaching Hospital, The Hague Case: 81 yrs, CVA, recurrent UTI,
Patient Safety Summit 2014
 Patient Safety Summit 2014 The War on C Diff Mark Mellow, MD + C Diff The Organism Gram + bacillus Anaerobic Spore forming Intestinal flora (up to 35% hospitalized patients, 3% of healthy adults) Leading
Patient Safety Summit 2014 The War on C Diff Mark Mellow, MD + C Diff The Organism Gram + bacillus Anaerobic Spore forming Intestinal flora (up to 35% hospitalized patients, 3% of healthy adults) Leading
CLOSTRIDIUM DIFFICILE: IMPROVING DIAGNOSIS AND TREATMENT. Joshua T. Watson, M.D. Lowcountry Gastroenterology Associates
 CLOSTRIDIUM DIFFICILE: IMPROVING DIAGNOSIS AND TREATMENT Joshua T. Watson, M.D. Lowcountry Gastroenterology Associates Learning Objectives Recognize patients who are highest risk for C. diff infections
CLOSTRIDIUM DIFFICILE: IMPROVING DIAGNOSIS AND TREATMENT Joshua T. Watson, M.D. Lowcountry Gastroenterology Associates Learning Objectives Recognize patients who are highest risk for C. diff infections
GI Bacterial Infections (part-2)
 GI Bacterial Infections (part-2) Mohammed Abdulla Mehdi FIBMS (internal medicine), FIBMS (G&H) Clostridium difficile infection C. difficile is the most commonly diagnosed cause of antibioticassociated
GI Bacterial Infections (part-2) Mohammed Abdulla Mehdi FIBMS (internal medicine), FIBMS (G&H) Clostridium difficile infection C. difficile is the most commonly diagnosed cause of antibioticassociated
Confirmed (Laboratory Tests) Serum positive for IgM anti-hbc or, hepatitis B surface antigen (HbsAg).
 Hepatitis B Hepatitis B is a liver disease that results from infection with the Hepatitis B virus. It can range in severity from a mild illness lasting a few weeks to a serious, lifelong illness. Hepatitis
Hepatitis B Hepatitis B is a liver disease that results from infection with the Hepatitis B virus. It can range in severity from a mild illness lasting a few weeks to a serious, lifelong illness. Hepatitis
