Effect of innovative double layer treatment on tooth color change and nitrate penetration
|
|
|
- Harry Robertson
- 5 years ago
- Views:
Transcription
1 University of Iowa Iowa Research Online Theses and Dissertations Spring 2016 Effect of innovative double layer treatment on tooth color change and nitrate penetration Abdullah AlShehri University of Iowa Copyright 2016 Abdullah Mohammed Alshehri This thesis is available at Iowa Research Online: Recommended Citation AlShehri, Abdullah. "Effect of innovative double layer treatment on tooth color change and nitrate penetration." MS (Master of Science) thesis, University of Iowa, Follow this and additional works at: Part of the Other Dentistry Commons
2 Effect of Innovative Double Layer Treatment on Tooth Color Change and Nitrate Penetration by Abdullah AlShehri A thesis submitted in partial fulfillment of the requirements for the Master of Science degree in Oral Science in the Graduate College of The University of Iowa May 2016 Thesis Supervisors: Associate professor SoRan Kwon Professor Philip Wertz
3 Copyright by Abdullah AlShehri 2016 All Rights Reserved
4 Graduate College The University of Iowa Iowa City, Iowa CERTIFICATE OF APPROVAL This is to certify that the Master s thesis of MASTER S THESIS ABDULLAH ALSHEHRI has been approved by the Examining Committee for the thesis requirement for the Master of Science degree in Oral Science at the May 2016 graduation. Thesis Committee: So Ran Kwon, Thesis supervisor Philip Wertz, Thesis supervisor Deborah Dawson Marcos Vargos
5 To my dear parents, for their unconditional love and patience. To all my family and friends, who supported me through it all. To my mentors for their enlightenment and guidance. ii
6 ACKNOWLEDGEMENTS First and foremost praised be God, who, whenever he is thanked for one of his blessings, provides another blessing, which in turn obliges one to thank him again because to give thanks for blessings is itself a blessing, one can never cease to give thanks. I would like to express my deepest gratitude to my mentor, Dr. SoRan Kwon. The knowledge and wisdom you have imparted upon me has been a great help and support throughout my research. I truly appreciate and value everything I have learned from you. It will forever remain a major contributor behind my success and achievements. I would like to thank my Co chair mentor, Dr. Philip Wertz, for his unconditional support throughout the course of my thesis work. A special thanks goes to, Dr. Deborah Dawson, for providing an excellent and detailed statistical analysis. Thanks for the significant guidance on how to report the results in my thesis and suggestions on my presentation. To Dr. Marcos Vargas, for his encouragement and support. He has been an influential advisor throughout my thesis work, never ceased to share his knowledge and priceless ideas, which I am really thankful for. I would like to acknowledge the manufacturer, Philips Oral Healthcare for kindly providing the materials. Finally, I give thanks to my parents. Their love was my driving forces every step of the way throughout this journey. Everything I am and will become I owe to them. So, thank you. iii
7 ABSTRACT Objectives: Evaluate and assess the effect of an innovative double layer, single application desensitizing/whitening technique on Nitrate penetration and total color change. Methods: Specimens were prepared from extracted caries free human molars (n=160). Teeth were randomly assigned into four groups: 100μl 25% hydrogen peroxide Philips Zoom chairside as control group (CTRL), double layer treatment of 20μl 5% potassium nitrate Relief ACP, Philips oral care and 100μl 25% hydrogen peroxide (DL20), double layer treatment of 40μl 5% potassium nitrate and 100μl 25% hydrogen peroxide (DL40), and one layer treatment of 40 μl 5% potassium nitrate (PN40). Spectrophotometric color measurements (Vita EasyShade) were done at base line (T0), one day (T1), and one month (T2) following the treatment. Nitrate penetration was measured using a nitrate/nitrite assay kit. Group comparisons of tooth color difference measurements, and nitrate penetration readings were made using the Kruskal-Wallis test. Adjustment was made for pairwise treatment comparisons using the Tukey method in conjunction with an overall 0.05 level of significance. Results: 160 teeth were used. Color difference (ΔE) results at (T1) and (T2) showed no significant difference among the CTRL, DL20, DL40 groups. But there was a statistical significant difference between those groups and the single layer (PN40) group (p<0.001, p<0.001 respectively). Data provided strong evidence of differences in nitrate penetration among the four groups (p<0.0001). Groups DL20, DL40, PN40 significantly differed from CTRL, but could not be said to differ from each other. Conclusion: Double layer technique showed no effect on color difference at one day and one month. The application of hydrogen peroxide didn t affect the penetration of potassium nitrate. iv
8 PUBLIC ABSTRACT For dentists, tooth whitening is a great aid for reaching patient expectations of a flawless smile. However, a major drawback of tooth whitening is tooth sensitivity. Potassium nitrate has been recommended to lessen tooth sensitivity in which it is applied in a tray prior to the tooth whitening treatment. This technique is time consuming and inconvenient, so companies incorporated potassium nitrate into the tooth whitening gel to make it a more time efficient technique. Due to the similar penetration rate of both potassium nitrate and tooth whitening gel and the lower molecular weight of tooth whitening gel, there is the assumption of the tooth whitening gel penetrating first through the tooth and onto the pulp in which it will leave it in an excitation state, by the time potassium nitrate reaches the pulp it will be too late to have the desired effect. So the purpose of this study is to innovate a one step technique that is less time consuming and will allow for potassium nitrate to penetrate first followed by hydrogen peroxide to interact with the stain molecules. An in vitro study to assess the effectiveness of 25% hydrogen peroxide tooth whitening agent, in terms of tooth color change when preceded by different amounts of potassium nitrate gel as a desensitizing agent was conducted. This technique is Time efficient technique, convenient technique for both the patient and the clinician, and it will pave the road for future approaches to manage tooth sensitivity. v
9 TABLE OF CONTENTS LIST OF TABLES... viii LIST OF FIGURES... x CHAPTER 1 INTRODUCTION Gap Purpose of Study... 4 CHAPTER 2 LITERATURE REVIEW Tooth Development Tooth Color Color Order Systems Color Matching and Reproduction Color Measuring Devices Tooth Discoloration History of Tooth Whitening Mechanism of Tooth Whitening Tooth Whitening Techniques Tooth Whitening Materials Factors Affecting Whitening Efficacy Time and Frequency Concentration of Whitening Agent Light Activation Heat Activation Sealed Environment Side Effect of Tooth Whitening Surface Change Effect on Oral Soft Tissues Tooth Sensitivity Desensitizing Agents Strontium Ions Sodium Fluoride Stannous Fluoride Sodium Mono Fluorophosphate Potassium Nitrate CHAPTER 3 MATERIALS AND METHODS Introduction Research Questions Hypotheses vi
10 3.4. Operational Definitions Study Design Sample Selection and Preparation Random Assignment of Teeth and Whitening treatment Determination of Color Change Nitrate and Nitrite Assay kit Calculation Data Entry and Storage Data Analysis Preliminary Studies Sample Size Estimation CHAPTER 4 RESULTS Baseline Comparisons Color Change within Tooth Whitening Group Group Comparison of Color Change Overall color change (ΔE*) Change in Lightness (ΔL*) Change in The Red-Green Color Dimension (Δa*) Change in The Yellow-Blue Color Dimension (Δb*) Potassium Nitrate Penetration CHAPTER 5 DISCUSSION Baseline Comparisons Sealed Tooth Whitening Technique Group Comparison of Overall color change Effect of Double Layer Technique on Overall Color Efficacy Potassium Nitrate Penetration Plugging of The Dentinal Tubules Cytotoxicity of Hydrogen peroxide, Fluoride Limitations of The Study Future Directions CHAPTER 6 CONCLUSION REFERENCES vii
11 List of Tables Table 1: Descriptive statistics based upon raw data from pilot study: Control: 80μL Hydrogen Peroxide Table 2: Descriptive statistics based upon raw data from pilot study: 20μL Potassium Nitrate + 80μL Hydrogen Peroxide Table 3: Descriptive statistics based upon raw data from pilot study: 60μL Potassium Nitrate + 80μL Hydrogen Peroxide Table 4: Descriptive statistics based upon raw data from final study: One-way ANOVA showed no statistical difference among groups at baseline Table 5: Descriptive statistics based upon raw data from final study: Control: 100μL Hydrogen Peroxide (CTRL) Table 6: Descriptive statistics based upon raw data from final study: Control: 100μL Hydrogen Peroxide + 20μL Potassium nitrate (DL20) Table 7: Descriptive statistics based upon raw data from final study: 100μL Hydrogen Peroxide + 40μL Potassium nitrate (DL40) Table 8: Descriptive statistics based upon raw data from final study: 40μL Potassium nitrate (PN40) Table 9: Tooth color difference (ΔL*, Δa*, Δb*, ΔE*) at one-day post tooth whitening Table 10: Tooth color difference (ΔL*, Δa*, Δb*, ΔE*) at one-month post tooth whitening Table 11: Descriptive statistics based upon raw data from final study: illustrating the overall color change (ΔE) between Time 1 and Time Table 12: Descriptive statistics based upon raw data from final study: illustrating the overall color change (ΔE) between Time 2 and Time Table 13: Descriptive statistics based upon raw data from final study: illustrating the overall color change (ΔE) between Time 2 and Time Table 14: Descriptive statistics based upon raw data from final study: illustrating the Change in lightness (ΔL*) between Time 1 and Time viii
12 Table 15: Descriptive statistics based upon raw data from final study: illustrating the Change in lightness (ΔL*) between Time 2 and Time Table 16: Descriptive statistics based upon raw data from final study: illustrating the Change in lightness (ΔL*) between Time 2 and Time Table 17: Descriptive statistics based upon raw data from final study: illustrating the Change in (Δa*) between Time 1 and Time Table 18: Descriptive statistics based upon raw data from final study: illustrating the Change in (Δa*) between Time 2 and Time Table 19: Descriptive statistics based upon raw data from final study: illustrating the Change in (Δa*) between Time 2 and Time Table 20: Descriptive statistics based upon raw data from final study: illustrating the Change in (Δb*) between Time 1 and Time Table 21: Descriptive statistics based upon raw data from final study: illustrating the Change in (Δb*) between Time 2 and Time Table 22: Descriptive statistics based upon raw data from final study: illustrating the Change in (Δb*) between Time 2 and Time Table 23: Descriptive statistics based upon raw data from final study: Nitrate penetration levels for the four treatment groups ix
13 List of Figures Figures 1. Box plots illustrating baseline L* by treatment group Figures 2. Box plots illustrating baseline a* by treatment group Figures 3. Box plots illustrating baseline b* by treatment group Figure 4. Longitudinal course of changes in ΔE* Figures 5. Box plots illustrating the overall color change (ΔE) between Time 1 and Time Figures 6. Box plots illustrating the overall color change (ΔE) between Time 2 and Time Figures 7. Box plots illustrating the overall color change (ΔE) between Time 2 and Time Figure 8. Longitudinal course of changes in ΔL* Figures 9. Box plots illustrating the overall color change (ΔL) between Time 1 and Time Figures 10. Box plots illustrating the overall color change (ΔL) between Time 2 and Time Figures 11. Box plots illustrating the overall color change (ΔL) between Time 2 and Time Figure 12. Longitudinal course of changes in Δa* Figures 13. Box plots illustrating the overall color change (Δa*) between Time 1 and Time Figures 14. Box plots illustrating the overall color change (Δa*) between Time 2 and Time Figures 15. Box plots illustrating the overall color change (Δa*) between Time 2 and Time Figure 16. Longitudinal course of changes in Δb* Figures 17. Box plots illustrating the overall color change (Δb*) between Time 1 and Time x
14 Figures 18. Box plots illustrating the overall color change (Δb*) between Time 2 and Time Figures 19. Box plots illustrating the overall color change (Δb*) between Time 2 and Time Figures 20. Box plots illustrating the nitrate penetration levels for the four treatments groups xi
15 Chapter 1 Introduction The growth of esthetic-based dentistry remains persistent as the media continues to emphasize beauty and health. Tooth whitening is the most common request for esthetic dentistry because it is the most conservative and economical solutions to lighten discolored teeth. It is also considered to be a safe and effective procedure when performed under the supervision of the dentist. This supervision includes proper diagnosis of the discoloration, treatment planning, and monitoring of the tooth color change. An attractive smile is accomplished with restorative procedures that modify the shape, position or shade of teeth [1]. However, when the primary goal is to solely enhance tooth color, non-restorative procedures such as enamel micro-abrasion and tooth whitening are the most effective and popular alternatives to restorative treatment [2]. Tooth whitening efficacy has been proven by many studies; Heymann et al. [3] reported a mean change of 7 units on the Vita shade guide after whitening with 10% carbamide peroxide (CP) over 7 days. However, there was a wide range of responses, ranging from 3 to 13 units, which highlights the unpredictable nature of whitening teeth. A mean change of 8 Vita units was reported with the use of 15% hydrogen peroxide (HP) [4]. Varying the concentrations of carbamide peroxide or hydrogen peroxide produced similar results, but the time taken to reach the end point was quicker with higher concentrations of whitening agents [5]. Long-term results of whitening efficacy were reported by Leonard [6], with at least 43% shade retention without retreatment at 10 years. Another study showed shade retention of up to 17 years [7]. 1
16 Despite the fact that tooth whitening is safe and effective, it is still accompanied by side effects that adversely affect patient compliance and satisfaction with their whitening experience. Tooth sensitivity is the most commonly reported side effect, with an incidence ranging from 0% to 75% [8]. The degree can vary from mild to intolerable, leading some patients to discontinue the treatment. Tooth sensitivity prevalence has been reported to be between 11% and 93%. This variation could be due to the subjective nature of sensitivity or different concentration of whitening agents [9]. Tooth sensitivity can be better described based on the occurrence or severity. Many studies have evaluated tooth sensitivity based on the time of onset and duration. Subjects were enrolled in a 5 day study which evaluated the effect of 10% carbamide peroxide reported tooth sensitivity after 4.8 days of treatment [10]. To test the widely held belief that higher concentrations of whitening agents produce a greater prevalence of tooth sensitivity, studies were conducted that used 30% HP and showed that 63% of the participants in their clinical study reported tooth sensitivity [11], while higher tooth sensitivity rates were reported such as 70% [12] and 80%[13]. Jorgenson et al. [14] evaluated the severity of tooth sensitivity caused by 10% CP. They found that 54% of subjects reported mild sensitivity, 10% reported moderate sensitivity, and 4% reported severe sensitivity. Sensitivity decreased with time; by the second week, no severe sensitivity was reported, and by the fourth week, no moderate sensitivity was reported. Although exposed dentin, root caries or exposed root surface could induce sensitivity, whitening-induced tooth sensitivity is believed to be related to the passage of hydrogen molecules through dentinal tubules and to the coronal part of the pulp [15]. Whitening-induced tooth sensitivity commonly presents itself as generalized sensitivity to cold stimuli, but can also manifest as spontaneous sharp shooting pain in a few teeth. While some authors have regarded the teeth as pressure-sensitive over a very high range of pressures, the most common sensation experienced is pain. Therefore, efforts to 2
17 elucidate the etiology and develop strategies for the prevention and treatment of whitening-induced tooth sensitivity continue to be a central issue to be addressed in tooth whitening procedures. According to Brännström s hydrodynamic theory, in the absence of a smear layer, fluids within the dentinal tubules can be subjected to various thermal, mechanical or osmotic stimuli that cause the outward flow of fluid in the dentinal tubules. Rapid fluid flow causes pressure changes across the dentin, which stimulate nerve receptors and result in the perception of pain [16]. Furthermore, Croll suggested that oxygen molecules released from carbamide peroxide or hydrogen peroxide diffuse into enamel and dentin. If sufficient oxygen molecules occupy the intracoronal space, pressure could affect pain receptors in the dentinal tubules and the pulp. The fact that higher concentrations of whitening agents are known to cause more severe sensitivity supports this theory [17]. Following Bra nnstro m s hydrodynamic theory, reducing the permeability of the dentinal tubules can alleviate tooth sensitivity; this can be achieved by the use of agents such as amorphous calcium salts or fluoride to aid in reducing the fluid flow within the dentinal tubules. Decreasing nerve excitations and blocking the transmission of pain to the central nervous system is another treatment modality that can be used to treat tooth sensitivity. Potassium nitrate is a salt that can act as a desensitizing agent which reduces the nerve excitations by diffusing the potassium salts through enamel and dentin; those salts can reach nerve termination, affect nerve impulse and eventually result in pain reduction or elimination [18]. Several studies had shown that the penetration time of hydrogen peroxide to the pulp chamber could vary from 5 to 15 minutes depending on the concentration used, and whether heat and light are used[19]. Recently Kwon et al [20], evaluated the penetration time of potassium nitrate and found that potassium nitrate could be detected in the pulp chamber as early as 5 minutes. 3
18 There are two common techniques for the delivery of potassium nitrate desensitizing agent to patients. The first technique is the application of potassium nitrate in a tray for thirty minutes prior to the whitening treatment [21]; the second treatment is incorporation of the potassium nitrate within the whitening gel [22]. However, there are significant drawbacks to such techniques. With the first technique, the added application of potassium nitrate in a separate tray makes whitening a time-consuming two-step technique. The second technique eliminates the drawback of the first technique by combining the potassium nitrate with the whitening gel. However, because studies have shown that potassium nitrate penetration into the pulp chamber can take up to 30 minutes [20], and 5-10 minutes for the hydrogen peroxide [19], is that there would be an interchangeable depolarization excitation action resulting from simultaneous penetration of the potassium nitrate and hydrogen peroxide Gap A double-layer treatment single application of potassium nitrate covered by hydrogen peroxide as the active whitening agent has not been investigated in the literature before Purpose of Study Appraise and assess an innovative double layer single application technique in which a layer of potassium nitrate is encrusted with another layer of hydrogen peroxide, with the presumption that this will allow for potassium nitrate penetration and temporarily delay the penetration of hydrogen peroxide without significantly affecting total color change during tooth whitening. 4
19 Chapter 2 Literature Review 2.1. Tooth Development Tooth formation begins at the 37 th day of development in which a continuous thickened band of epithelium is formed in the upper and lower jaws. This primary epithelial band will give rise to both the dental lamina and vestibular lamina. Dental lamina will appear as a thickening of the oral epithelium adjacent to the condensation of the ectomesenchyme. After that, 20 areas of enlargement will appear to form the tooth buds of the 20 primary teeth. The dental lamina begins to function at the 6th prenatal week and continues to the 15th year after birth, depending on which tooth it will form, since some teeth are predecessors to others. Even though tooth formation is a continuous process, it can be divided into three stages: Bud stage, Cap stage, and Bell stage. Bud stage is characterized by rounded, localized growth of epithelium surrounded by proliferating mesenchymal cells, which are packed closely beneath and around the epithelial buds. In the Cap stage, the ectomesenchyme will condense immediately subjacent to the tooth bud because of the lack of extracellular matrix secretion by the cells. Histodifferentiation begins at the end of this stage. Enamel will form through an epithelial outgrowth called the enamel organ. Moreover, the dentin and pulp will form through the dental papilla, which is a ball of condensed ectomesenchymal cells. The support structures of the tooth (cementum and periodontal ligament) will form through the dental follicle which is a condensed ectomesenchymal tissue surrounding the enamel organ and dental papilla. In the Bell stage, there is a continuation of histodifferentiation including the definition of 5
20 ameloblasts and odontoblasts, and the beginning of morpho-differentiation in which the tooth crown assumes its final shape [23] Tooth Color A smile is considered one of the most interactive communication skills of a person [24]. As a result, the ultimate objective of esthetic dentistry is to create a beautiful smile with teeth that exhibit the correct form and shape, reflect the correct color, and to be in harmony with the surrounding gingiva, lips and face. These characteristics should be considered when teeth are being restored [25]. Color change is determined by the combined effects of intrinsic and extrinsic colorations. Intrinsic change is determined by the absorbance and reflectance properties of enamel and dentin [26], while extrinsic changes result from the absorption of materials (tea, coffee) into the outer surface of enamel [27] Color Order System Color is a psychophysical response to the physical interaction of the light energy with an object and the subjective experience of the observer. That being said, three factors affect the perception of color: the light source, the object, and the subjectivity of the observer. Color can be described according to a visual system called Munsell color space in terms of hue, value and chroma. Hue is the attribute of a color that enables one to distinguish between different families of color, for example, reds, blues and greens. Value indicates the lightness of a color ranging from pure black to pure white. Chroma is the degree of color saturation and describes the intensity of a color [28]. The Commission Internationale d el E ćlairage (CIE) is another color space instrumental system that gained its popularity by adapting the theory of the three colors receptors in the eye (red, green, blue). In this three-dimensional color space, the three axes are L*, b*, a*. The L* axis corresponds to the lightness of an object, with a value of 6
21 zero presenting a black object and a value of a 100 presenting a perfectly reflecting object. The b* axis is the yellow to blue scale with the yellow representing a (positive b) and the blueness representing a (negative b). The a* value is the red to green scale in which a (negative a) corresponds to greenness and a (positive a) to redness [29]. The CIE color system is well suited for instrumental color analyses and research applications; it measures the reflectance of the object at each wavelength, and transforms the data into useful color descriptors in an appropriate color space [29]. A formula is used in this system to calculate color difference. ΔEab= [(L1-L2) 2 +(a1-a2) 2 +(b1-b2) 2 ] 1/ Color Matching and Reproduction Throughout history, advances in technologies and techniques to improve colormatching skills have been pushed even further by the stress on having the ideal restoration. Since the 1970s, companies have developed several shade guides with the ultimate goal of having one that encapsulates all existing natural tooth shades. The Vita Lumin classical shade guide offers ease of use and accuracy through its logical system of shade grouping. This guide consists of 16 hue tabs divided into four shade groups A, B, C, and D. Hues differ as groups change, wherein group A is reddish-brown, group B is reddish-yellow, group C is grey, and group D is reddish-grey. Within each group, lightness decreases and chroma increases as the number increases. The Vita classic is the most commonly used shade guide by dental practitioners. However, a logical and adequate distribution in the color space as defined by the CIELab* was lacking. Therefore, a more comprehensive shade guide was needed to compensate of the shortcomings of the Vita classical. As a result, the Vitapan 3D master shade guide surfaced. This shade guide system provides a systemic organization of all existing natural tooth color as assumed by the manufacturer based on spectrophotometric 7
22 measurements of natural teeth. The shade guide consists of 26 tabs and is arranged in a way that it covers natural tooth color space in a logical order; the tabs are organized in five value levels. Within each level are tabs that represent different chromas and hues. Tabs are arranged according to chroma vertically and hue horizontally. The sequence of shade selection is value, then chroma, followed by hue [30-32]. The use of the Vitapan 3D master was challenging when it comes to measure teeth undergoing tooth whitening. Thus, the vita bleach guide was designed to provide and simplify the visual evaluation of tooth whitening efficacy as compared to the other shade tabs. This 15-tab shade guide was re-designed to allow for a linear shade guide that is familiar to dental practitioners, along with decreasing the gap of L among groups [33] Color Measuring Devices Tooth color matching has always been a stressful decision making process undertaken by dental practitioners, due to the subjectivity, lack of control and lack of an ideal environment while attempting tooth shade matching. The introduction of new technologies in tooth color matching with the sole purpose of analyzing and verifying tooth color has advanced the dental profession [31]. Shade measuring devices have eliminated the difficulties associated with the subjective shade matching technique, such as metamerism, suboptimal color matching conditions, tools and method as well as the receiver s age fatigue, mood and medication [34]. Shade measuring devices can be divided into three categories; spectrophotometers, colorimeters and digital imaging. Spectrophotometers are one of the most popular, accurate and easy to use shademeasuring devices for overall color matching. They measure the amount of light energy reflected from an object at 1-25nm along the visible spectrum [35]. They contain a source of optical radiation, a means of dispersing light, an optical system for measuring, a detector and a means of converting light obtained to a signal that can be analyzed [35]. 8
23 Data obtained from the devices are translated into more simple information resembling shade tabs, which are more readily perceived by dental practitioners and lab technicians. A huge advantage of this system is the virtual shade tab in which the shade taken by the device can be superimposed over a clinical tooth image to aid the dental practitioners in a more precise shade selection. In fact, these devices were proved to be 33% more accurate than subjective shade matching [35]. Different models of spectrophotometers are available; EasyShade (VITA Easyshade ) ShadeX (X Rite, Grand Rapids, Michigan, and SpectroShade Micro (MHT, Verona, Italy) are the most common. EasyShade is a costefficient, cordless, tooth contact device that aids in the analysis and verification of tooth shade color. Different modes are available, such as tooth single mode, tooth area mode, restoration color verification, and shade tab mode. ShadeX closely resembles EasyShade because it s cordless and easy to use, but differs in the interface because it has two databases in which it can detect the color of the dentin and incisal color. SpectroShade Micro is an imaging spectrophotometer that uses a digital camera-led spectrophotometer combination [31] Tooth Discoloration Tooth color is dependent on the reflectance and absorbance properties of the tooth, which are influenced by the structures of the tooth (enamel, dentin, pulp). Any changes that effect these structures before or after eruption will result in tooth color change and hence tooth discoloration [36]. Tooth discoloration can be divided into two main groups: intrinsic stains and extrinsic stains. Intrinsic stains could be due to metabolic, inherited, iatrogenic, traumatic, idiopathic, or ageing causes. Metabolic disorders such as alkaptonuria and congenital erthropoietic porphyria may cause tooth discoloration. Alkaptonuria is an inborn error of metabolism that results in a brown discoloration of the permanent dentition [37], whereas congenital erthropoietic porphyria gives a red purple brown discoloration of teeth [38]. 9
24 Amelogenesis imperfecta and Dentinogensis imperfecta are examples of inherited causes of tooth discoloration. Amelogenesis imperfecta is presented in different forms; teeth in the mild form are yellowish to brown with thin, hypoplastic enamel. The darker the tooth, the more severe the hypomineralization of the enamel. Dentinogensis imperfecta is characterized by opalescent primary teeth; this condition affects primary teeth more than permanent teeth. Teeth are grey to purple blue in color or opalescence [39]. Iatrogenic causes of tooth-discoloration include tetracycline drugs that are administered systematically either by taking the drug or through mothers breastfeeding their children. The severity of the stain is dependent on the age of the person, amount of drug intake, and exposure time. Teeth generally look worse immediately after eruption, and discoloration tends to lessen after that due to photo-oxidation (exposure to light). These teeth exhibit a yellow or brown grey discoloration [40]. Another cause of discoloration is fluorosis, which may be due to the natural occurrence of fluoride in water, toothpaste, or tablets. Teeth are characterized by a chalky, white enamel or dark brown color, depending on the severity[41]. One of the most common causes of intrinsic tooth discoloration is trauma, which is believed to be due to accumulation of hemoglobin molecules in the traumatized tooth. Enamel hypoplasia is another cause of intrinsic staining that is believed to be due to disturbance to the tooth germ following trauma [42]. Molar incisor hypomineralization is a condition of unknown etiology that affects 1 st molars and incisors. Teeth are characterized by enamel loss and discolorations that may vary from white, yellow or brown areas [43]. Color changes associated with aging could be due to thinning of enamel and thickening of the deposition of dentin as teeth age and become darker and more yellow [44]. Extrinsic tooth discoloration is another major category of tooth discoloration. This type is more manageable than intrinsic tooth discoloration and commonly occurs 10
25 with age, where it is more prevalent in men than women [45]. Dietary components, beverages, tobacco and mouth rinses causes non-metallic stains, which are adsorbed onto the tooth surface deposits such as plaque. In children with poor oral hygiene, primary teeth may present with green and yellowish-orange stains mainly caused by bacteria in bacterial plaque. These types of stains are usually found on the buccal surface of teeth. Other stains due to bacteria are the dark brown to black discolorations found on the cervical portion of primary teeth; in this case, patients exhibit a low caries rate with fair oral hygiene[46]. Metallic stains most commonly occur with exposure to metallic salts [47], and may be manifested as a black stain caused by iron supplements. Green stain is caused by mouth rinses containing copper [48]. Other metals have associated colors such as potassium permanganate producing a violet to black color when used in mouth rinses; silver nitrate salt used in dentistry causes a grey color [49]. Moreover, stannous fluoride causes golden brown discoloration. It is believed that that the mechanism of the metallic stain process is due to the production of sulphide salt of the specific metal involved. In fact, the extrinsic stain coincides with the color of the sulphide of the metal concerned. However, strong evidence for the chemical process necessary to produce the metal sulphide is lacking [50]. Brown stains are a very common type of stain that is bacteria free; these stains are usually due to a thin pigmented pellicle which is found most commonly on the buccal surface of maxillary molars and the lingual surfaces of lower anterior teeth. This type of stain is believed to be caused by the deposition of tannin found in chromatic beverages and is generally associated with poor oral hygiene and improper brushing [51]. Tobacco stains usually mask the gingival portion of the tooth and mainly occupy enamel defects. They manifest as a dark brown/ black discoloration, mainly caused by deposition of the coal tar with the possibility of enamel penetration [52]. This stain does not entirely depend on the amount of tobacco consumed, but rather the amount of 11
26 preexisting coating and roughened enamel that will eventually allow tobacco products to adhere [53]. Green stains frequently occur in children, affecting boys more than girls, and appears as a thick band on the buccal surface of maxillary anterior teeth in the gingival third. Some believe it is a remnant of the primary enamel cuticle. Others suggest it could be due to fluorescent bacteria and fungi. Photo-activation is essential for this type of bacteria which explains their presence on maxillary anterior teeth [51]. Orange stains are the least common type of stain. They occur on the labial surface of the upper and lower anterior teeth, mainly caused by bacteria such as Serratia marcescens and Flavobacterium lutescens and are usually associated with poor oral hygiene [52] History of Tooth Whitening Tooth whitening is the most conservative treatment for discolored teeth when compared to composites veneers and crowns. Tooth whitening is very effective in changing tooth color [54]. In the late 1800s, esthetic dentistry was popular, and some of the techniques that were used back then are still utilized, including recontouring, tooth whitening, gold onlays and ceramic inlays to save as much of tooth structure as possible [55]. During this period, recontouring in which the dentist used a type of ink to mask the portion of the tooth to be contoured for patient approval was an emerging trend [56]. From the mid-1800s to early 1900s, reputable dental journals were filled with tooth whitening articles. Truman was the first who described non-vital tooth whitening, which used chlorine from a solution of calcium hydrochloride and acetic acid [15]. Different agents were also introduced in conjunction with non-vital whitening, for example aluminum chloride [57], oxalic acid [55] pyrozone (ether-peroxide) [58] hydrogen dioxide (hydrogen peroxide or perhydrol)/sodium peroxide [59], sulphorus acid [60], sodium hypophosphate, chloride of lime [61], and cyanide of potassium [62]. 12
27 Regardless of which agent was used, they were all considered either direct or indirect oxidizers that acted on the organic portion of the tooth [60]. Whitening agents were categorized depending on the stains that they removed. For example, iron stains were removed with oxalic acid [63], silver and copper stains with chlorine [64], and iodine stains with ammonia [65]. The stains of metallic salts from metallic restorations such as amalgam were considered the most resistant to whitening. Even though cyanide of potassium would remove such metallic stains, its use was not recommended because of its being a very active poison [66]. In the 1800s up to the 1900s, dental literature was more focused on non-vital whitening. However, vital whitening was attempted in 1886 with the use of oxalic acid [63]. Later, hydrogen peroxide or Pyrozone were introduced. Moreover, by 1910 these vital whitening techniques generally included the use of hydrogen peroxide with a heating instrument or a light source [67]. In 1893, 3% of Pyrozone (ether-peroxide), a soluble form of hydrogen dioxide, was used as a mouthwash by both children and adults. In children with pitted teeth, it had the beneficial side effect of reducing caries and whitening the teeth. In addition, the 5% solution was used to bleach teeth; however, the 25% solution, which is the most effective whitening agent, was recommended to be used with caution, because of its unwanted adverse effects such as gingival burns [68]. In the 1960s Klusmier developed a technique in which he used 10% carbamide peroxide delivered in a custom fitting tray. His intention was to treat gingival inflammation, but he found that teeth became lighter in color. Unfortunately, this technique was not recognized until Haywood popularized it in 1989 [69]. 13
28 2.5. Mechanism of Tooth Whitening Hydrogen peroxide is the active whitening ingredient by acting as a strong oxidizing agent that forms free radicals. These free radicals reduce the amount of pigmented molecules by one of two methods: break down these pigmented molecules to smaller particles that diffuses through the tooth, or transform them into molecules that absorb less light, and, as a result, appear lighter [70]. Hydrogen peroxide forms a loose reaction with urea to form carbamide peroxide, and the newly formed material breaks down to free radicals that will penetrate through the enamel and into the dentin to produce the desired whitening effect. Furthermore, this material could decompose into ammonia and carbon dioxide, which is believed to elevate the ph in the tooth to improve the performance of the whitening agent [71]. There are three theories for the mechanism of tooth whitening: diffusion, interaction and surface color change[72]. In diffusion, the tooth whitening gel penetrates tooth layers and interacts with organic chromophores. Dental hard tissues are highly permeable to fluids with the interprismatic spaces being the most permeable [73, 74]. Thus, it s theorized that enamel and dentin will act as semipermeable membranes. They will thus allow hydrogen peroxide to move according to Fick s second law of diffusion, which states that the diffusion of a molecule is proportional to the surface area, diffusion coefficient, and concentration, and that it is inversely proportional to the diffusion distance [75]. Many factors affect tooth whitening gel penetration. Such factors include: concentration of the whitening gel, prolonged application, large dentinal tubules diameter, and increase in temperature [19, 76-78]. However, no clinical relevance was found since these factors do not replicate a clinical situation with the pulp producing an outward pulp pressure Interaction, also known as the chromophore theory, refers to an interaction between the hydrogen peroxide and the organic chromophore. These organic chromophores are colored molecules that consist of either conjugated pi systems, such as 14
29 aromatic compounds that have electron-rich areas, or bioinorganic metallic complexes, such as chelates [79]. Reactive oxygen interacts with the stain molecules to convert them to less complex molecules. Because these less complex molecules will be lower in molecular weight and more polar, this process will enable such molecules to be washed out in an aqueous environment [72]. Temperature and concentration of the peroxide and ph play a very crucial rule in the interaction and formation of oxygen [72, 80]. The third theory for the mechanism of tooth whitening is surface change and color. In this process, tooth color is the result of interaction of the light energy with an object, and the subjective experience of the observer. Three factors would affect the perception of color; the light source, the object, and the subjectivity of the observer [28]. Considering these factors, along with inclusion of optical properties such as opacity, translucency and opalescence [81], selection of tooth shade could be a difficult task to achieve. Tooth color is mainly derived from the body of tooth, i.e. dentin, with the translucency of enamel playing a minor contributing role to the overall color [82]. This was suggested by Bosch et al who evaluated color difference between extracted teeth in which they measure tooth color before and after removal of enamel, and concluded that tooth color is mainly derived from dentin and enamel plays insignificant role in the equation [26]. Moreover, dentin plays an important role in the tooth whitening mechanism, as it believed that the whitening gel interacts with dentin to produce the desired overall color, Several studies that evaluated the separate contributions of enamel and dentin to the overall change in tooth color during whitening concluded that the change was mainly due to changes in the subsurface of dentin [83, 84]. In contrast, enamel has been shown to play an important role in the overall color change as it was proven that, due to the decrease in enamel translucency, hence masking the dentin, production of a lighter shade was achieved [85, 86]. This decrease in translucency is accounted for by the micro-morphological alterations of the most superficial enamel through deproteinization, demineralization, and oxidation [87, 88]. Another important 15
30 factor in affecting the overall color is the effect of tooth whitening on the surface characteristics. The coarser the surface, the more diffusion in reflection, resulting in an opaque surface which will result in a brighter object [86, 88, 89] Tooth Whitening Techniques Tooth whitening techniques can be divided into at-home and in-office. At-home techniques, which Haywood and Heymann introduced in 1989, have been widely used, under dentist supervision. Although different whitening agent concentrations have been introduced, the ADA recommendation is 10 % carbamide peroxide. At-home whitening usually has different application times (1, 3, and 8 hours) depending on treatment needs and agent concentration. Sensitivity and gingival irritation are common side effects in this type of treatment. Nevertheless, at-home whitening has been proven by numerous studies to be effective and safe [90]. At-home whitening has been extensively used in United States for several decades. However, one of its drawbacks is that it is time consuming. For people who demand rapid results, in-office whitening is the treatment of choice. In-office whitening uses high concentrations of hydrogen peroxide with less exposure time. Studies have shown that in-office whitening may cause an adverse effect on tooth structure due to the use of high concentration of hydrogen peroxide [91]. Numerous studies were conducted to assess the efficacy of different tooth whitening techniques. Dawson et. al. [9] assessed the efficacy of color change after home whitening alone vs. two different regimens of combined whitening. 36 subjects were randomly assigned to three groups: at-home whitening for two weeks with 16% carbamide peroxide, at-home whitening for two weeks with 16% carbamide peroxide supplemented with in-office whitening with 9% hydrogen peroxide, and at-home whitening for two weeks with 16% carbamide peroxide supplemented with in-office whitening with 27% hydrogen peroxide. To evaluate the color change, one examiner and the examiner s 16
31 assistant assessed tooth shades independently with reference to a value ordered shade guide. They found that there was a statistically significant difference between tooth shade before and after treatment, as shown in Table 1. However, no significant difference was noted among groups in terms of color change after treatment. Table 1. Shade changes at start and end of home whitening phase.[9] Group Mean shade at Mean shade at Mean p value baseline 1week follow up difference A <0.05 B <0.05 C <0.05 A study by Giachetti et. al. [91] compared whitening results of in-home whitening with those of in-office and the longevity of the effects of each at nine months after whitening was measured. 17 participants had their maxillary first premolars treated with either 10% carbamide peroxide at-home whitening with an application time for 6 to 8 hours/day (14 days) or 38% hydrogen peroxide in-office whitening (one visit) at the end of at-home whitening treatment (14 th day). A split mouth design was followed. Color parameters, lightness (L*), redness (a*) and yellowness (b*) were measured using a spectrophotometer at baseline, at one week, one month and nine months after whitening. After measurements, whiteness index was calculated. They found that at the one week assessment, there were no statistically significant reduction in a*(p=0.85), L*(p=0.663) or W (p=0.5) values for both techniques. In-office whitening resulted in b* values that were statistically significantly higher than for the athome technique (p<0.03). At one month and nine months, no statistically significant 17
32 difference was found between the two groups in any of the color parameters. Both techniques attained higher W values at one week after whitening than at baseline and then remained elevated at the follow up appointments Tooth Whitening Materials Tooth whitening materials have been increased in popularity, with carbamide peroxide and hydrogen peroxide being the most popular. These materials can be delivered in a custom fitting tray or a standard size tray. More viscous whitening materials have replaced glycerin-based materials due to issues with tooth dehydration. However, such restorations leak easily, causing gingival irritation. In addition, hydrogen peroxide has been used with polyethylene strips as over-the-counter whitening agents, in which individuals apply the strip on their front teeth and overlap it to the lingual surface. Disposable trays can be used to bleach the teeth with 9% hydrogen peroxide [92]. Hydrogen peroxide is a chemical compound with the formula H 2 O 2. Colorless in its pure form, this compound is slightly more viscous than water, but it s used as an aqueous solution for precautionary reasons. It is the simplest of all peroxides, used as a strong oxidizer agent and as a disinfectant. Concentrated H 2 O 2 is highly reactive and used as propellant in rocketry. This compound could cause burns when in contact with skin. Although chemically, it could be mistaken for water with an extra oxygen atom, this compound is dominated by the nature of its unstable peroxide bond [93]. Hydrogen peroxide is used in dentistry as an oxidizing agent. Because of its low molecular weight, hydrogen peroxide can penetrate dentin and release oxygen that breaks the double bonds of the organic and inorganic compounds inside the dentinal tubules. Hydrogen peroxide is used as a tooth whitening material at different concentrations from 5% to 35%. In order to improve its ease of use in whitening teeth, it also comes in different delivery systems, most commonly in the form of gel. It acts as a strong oxidizing agent through the formation of reactive oxygen species and hydrogen peroxide anions [94, 95]. 18
33 Hydrogen peroxide breakdown is highly dependent on the temperature, type and concentration of the peroxide, as well as the ph [96]. There are two types of hydrogen peroxide reactions, hemolytic and heterolytic cleavage. In the former, free radicals are formed presence of light and heat as shared electrons split apart, leaving an unshared electron thus forming free radicals. In heterolytic cleavage, upon the increases in ph upon deprotonation leave the electron pair and generates hydroxyl anions. Interestingly, a combination of these two techniques will result in the formation of both an anion and a free radical [97]. The active oxygen is attracted to the electron-rich areas of stain molecules and, by cleaving the double bond, contribute to the decrease of color or the removal of the compound [98]. Carbamide peroxide is a solid compound containing an equal amount of hydrogen peroxide and urea; it is a white crystalline compound that dissolves in water to produce free hydrogen peroxide and is often used in dentistry in tooth whitening procedures. It used in a concentration ranging from 10% to 35%. A 10% carbamide peroxide solution breaks down into 3.35% hydrogen peroxide and 6.65% urea [99]. Urea further breaks into ammonia and water, raising the ph of the solution [100]. Carbamide peroxide contains a carbopol base in which it slows down the release of hydrogen peroxide, making it more effective over long period of time [101]. Sodium perborate is an odorless, water soluble chemical compound. This compound is available in the form of a powder and is used as an oxidizer and as an intracoronal whitening agent for necrotic teeth. It reacts with acid, warm air and water in which it breaks into sodium metaborate, hydrogen peroxide and nascent oxygen [94]. Sodium perborate can be present in the form of monohydrate, trihydrate, and tetrahydrate, which differ in oxygen content and thus their bleaching efficacy [102]. 19
TOOTH DISCOLORATION. Multimedia Health Education. Disclaimer
 Disclaimer This movie is an educational resource only and should not be used to manage dental health. All decisions about the management of tooth discoloration must be made in conjunction with your dentist
Disclaimer This movie is an educational resource only and should not be used to manage dental health. All decisions about the management of tooth discoloration must be made in conjunction with your dentist
5Recommended Shade-Matching Protocol
 5Recommended Shade-Matching Protocol In this chapter: Seven steps to a successful shade match 5 Recommended Shade-Matching Protocol Figs 5-1 and 5-2 Conventional methods of shade selection, when used alone,
5Recommended Shade-Matching Protocol In this chapter: Seven steps to a successful shade match 5 Recommended Shade-Matching Protocol Figs 5-1 and 5-2 Conventional methods of shade selection, when used alone,
Learning and applying the Natural Layering Concept
 I industry report _ natural layering concept Learning and applying the Natural Layering Concept Author_ Prof Didier Dietschi, Switzerland Right_Dentine samples of the Miris 2 system, developed according
I industry report _ natural layering concept Learning and applying the Natural Layering Concept Author_ Prof Didier Dietschi, Switzerland Right_Dentine samples of the Miris 2 system, developed according
Randomized, Prospective Evaluation of The Effectiveness of the Zoom2 Dental Whitening Lamp and Light-Catalyzed Peroxide Gel
 Randomized, Prospective Evaluation of The Effectiveness of the Zoom2 Dental Whitening Lamp and Light-Catalyzed Peroxide Gel Steven L. Ziémba, M.S. 1 Heather Felix, M.S. 1 Martin Giniger, DDS 2 Marilyn
Randomized, Prospective Evaluation of The Effectiveness of the Zoom2 Dental Whitening Lamp and Light-Catalyzed Peroxide Gel Steven L. Ziémba, M.S. 1 Heather Felix, M.S. 1 Martin Giniger, DDS 2 Marilyn
Peninsula Dental Social Enterprise (PDSE)
 Peninsula Dental Social Enterprise (PDSE) Bleaching (Whitening) policy Version 1.0 Date approved: October 2016 Approved by: The Board Review due: October 2019 Policy will be updated as required in response
Peninsula Dental Social Enterprise (PDSE) Bleaching (Whitening) policy Version 1.0 Date approved: October 2016 Approved by: The Board Review due: October 2019 Policy will be updated as required in response
Whitening. Give your patients pure with Philips Zoom! Whitening. +44(0)
 Whitening Give your patients pure with Philips Zoom! Whitening. www.bfmulholland.com/philips +44(0) 28 94 452 668 philipsorders@bfmulholland.com Philips Zoom! WhiteSpeed Light-activated technology, up
Whitening Give your patients pure with Philips Zoom! Whitening. www.bfmulholland.com/philips +44(0) 28 94 452 668 philipsorders@bfmulholland.com Philips Zoom! WhiteSpeed Light-activated technology, up
Chapter 14 Outline. Chapter 14: Hygiene-Related Oral Disorders. Dental Caries. Dental Caries. Prevention. Hygiene-Related Oral Disorders
 Chapter 14 Outline Chapter 14: Hygiene-Related Oral Disorders Hygiene-Related Oral Disorders Dental caries Prevention Gingivitis Prevention Tooth hypersensitivity Pathophysiology Treatment 2 Hygiene-Related
Chapter 14 Outline Chapter 14: Hygiene-Related Oral Disorders Hygiene-Related Oral Disorders Dental caries Prevention Gingivitis Prevention Tooth hypersensitivity Pathophysiology Treatment 2 Hygiene-Related
Effect of Small Starfruit (Averrhoa bilimbi L.) Extract Gel On Tooth Enamel Color Changes
 Journal of Physics: Conference Series PAPER OPEN ACCESS Effect of Small Starfruit (Averrhoa bilimbi L.) Extract Gel On Tooth Enamel Color Changes To cite this article: S I M Musnadi et al 2018 J. Phys.:
Journal of Physics: Conference Series PAPER OPEN ACCESS Effect of Small Starfruit (Averrhoa bilimbi L.) Extract Gel On Tooth Enamel Color Changes To cite this article: S I M Musnadi et al 2018 J. Phys.:
Development of teeth. 5.DM - Pedo
 Development of teeth 5.DM - Pedo Tooth development process of continuous changes in predetermined order starts from dental lamina A band of ectodermal cells growing from the epithelium of the embryonic
Development of teeth 5.DM - Pedo Tooth development process of continuous changes in predetermined order starts from dental lamina A band of ectodermal cells growing from the epithelium of the embryonic
In Vivo Study of Two Carbamide Peroxide Gels with Different Desensitizing Agents
 ! Operative Dentistry, 2007, 32-6, 549-555 In Vivo Study of Two Carbamide Peroxide Gels with Different Desensitizing Agents!"#$%&'(#)#$"#*+,-.%/# 01#2,34.&#)#15#$%&'(# # # "#$%!&'!($)*+##+,-!./($)01+2$!3$-1+#1)'4!5-67!!8)0-#*+##+,-!,)!)$(),%961+,-!,:!
! Operative Dentistry, 2007, 32-6, 549-555 In Vivo Study of Two Carbamide Peroxide Gels with Different Desensitizing Agents!"#$%&'(#)#$"#*+,-.%/# 01#2,34.&#)#15#$%&'(# # # "#$%!&'!($)*+##+,-!./($)01+2$!3$-1+#1)'4!5-67!!8)0-#*+##+,-!,)!)$(),%961+,-!,:!
DENTAL STAINING AMONG PATIENTS VISITING THE ORAL DIAGNOSIS CLINIC IN THE UNIVERSITY OF NAIROBI DENTAL HOSPITAL
 DENTAL STAINING AMONG PATIENTS VISITING THE ORAL DIAGNOSIS CLINIC IN THE UNIVERSITY OF NAIROBI DENTAL HOSPITAL A COMMUNlTY DENTISTRY PROJECT SUBMITTED FOR PARTIAL FULFllLMENT OF BACHELOR OF DENTAL SURGERY
DENTAL STAINING AMONG PATIENTS VISITING THE ORAL DIAGNOSIS CLINIC IN THE UNIVERSITY OF NAIROBI DENTAL HOSPITAL A COMMUNlTY DENTISTRY PROJECT SUBMITTED FOR PARTIAL FULFllLMENT OF BACHELOR OF DENTAL SURGERY
Applying color theory in clinical practice to improve patient treatment
 Applying color theory in clinical practice to improve patient treatment Asbjørn Jokstad Science Manager, FDI World Dental Federation Professor, University of Oslo, Norway 1 Learning objectives Be familiar
Applying color theory in clinical practice to improve patient treatment Asbjørn Jokstad Science Manager, FDI World Dental Federation Professor, University of Oslo, Norway 1 Learning objectives Be familiar
Teeth Whitening 101: Everything You Need to Know About Teeth Whitening. By Penn Dental Family Practice
 Teeth Whitening 101: Everything You Need to Know About Teeth Whitening By Penn Dental Family Practice What is Teeth Whitening? Teeth whitening is a cosmetic dental treatment aimed at making your teeth
Teeth Whitening 101: Everything You Need to Know About Teeth Whitening By Penn Dental Family Practice What is Teeth Whitening? Teeth whitening is a cosmetic dental treatment aimed at making your teeth
QuickPro. Instructions for use
 QuickPro Instructions for use Step 1 Step 2 Step 3 Step 4 Step 5 2 Step 6 Step 7 Step 8 Step 9 Step 10 3 Symbols used in labeling Irritant Irritant Consult instruction manual for use Batch code REF Catalog
QuickPro Instructions for use Step 1 Step 2 Step 3 Step 4 Step 5 2 Step 6 Step 7 Step 8 Step 9 Step 10 3 Symbols used in labeling Irritant Irritant Consult instruction manual for use Batch code REF Catalog
TOOTH WHITENING. Copyright 2017 Dr Krystyna
 TOOTH WHITENING What is tooth whitening? Tooth whitening can be a very effective way of lightening the natural colour of your teeth without removing any of the tooth surface. It cannot make a complete
TOOTH WHITENING What is tooth whitening? Tooth whitening can be a very effective way of lightening the natural colour of your teeth without removing any of the tooth surface. It cannot make a complete
Evidence Based Tooth Whitening Dr. Bruce A. Matis
 Evidence Based Tooth Whitening Dr. Bruce A. Matis www.bamatis.com The objective of this presentation is to increase your understanding of tooth whitening. We will do this by reviewing the clinical studies
Evidence Based Tooth Whitening Dr. Bruce A. Matis www.bamatis.com The objective of this presentation is to increase your understanding of tooth whitening. We will do this by reviewing the clinical studies
EVALUATION OF COLOR CHANGE IN WHITE SPOT LESIONS OF ENAMEL FLUOROSIS USING A RESIN INFILTRATE
 EVALUATION OF COLOR CHANGE IN WHITE SPOT LESIONS OF ENAMEL FLUOROSIS USING A RESIN INFILTRATE Monisha Singhal PG STUDENT DEPARTMENT OF PEDODONTICS & PREVENTIVE DENTISTRY CHANDRA DENTAL COLLEGE & HOSPITAL
EVALUATION OF COLOR CHANGE IN WHITE SPOT LESIONS OF ENAMEL FLUOROSIS USING A RESIN INFILTRATE Monisha Singhal PG STUDENT DEPARTMENT OF PEDODONTICS & PREVENTIVE DENTISTRY CHANDRA DENTAL COLLEGE & HOSPITAL
Tooth Whitening. General Information pack The Smile Clinic
 Tooth Whitening General Information pack The Smile Clinic This pack contains general information on tooth whitening You need to discuss your requirements with your dentist who will tailor the treatment
Tooth Whitening General Information pack The Smile Clinic This pack contains general information on tooth whitening You need to discuss your requirements with your dentist who will tailor the treatment
Management of Inadequate Margins and Gingival Recession Presenting as Tooth Sensitivity
 Management of Inadequate Margins and Gingival Recession Presenting as Tooth Sensitivity Nicolas Elian, DDS Private Practice Englewood Cliffs, New Jersey David Geon U Kim, DDS, MS Faculty and Research Coordinator
Management of Inadequate Margins and Gingival Recession Presenting as Tooth Sensitivity Nicolas Elian, DDS Private Practice Englewood Cliffs, New Jersey David Geon U Kim, DDS, MS Faculty and Research Coordinator
The Dental Board of California - Dental Materials Fact Sheet Adopted by the Board on October 17, 2001
 The Dental Board of California - Dental Materials Fact Sheet Adopted by the Board on October 17, 2001 As required by Chapter 801, Statutes of 1992, the Dental Board of California has prepared this fact
The Dental Board of California - Dental Materials Fact Sheet Adopted by the Board on October 17, 2001 As required by Chapter 801, Statutes of 1992, the Dental Board of California has prepared this fact
POWER BLEACHING A SOLUTION FOR DISCOLOURED TEETH
 doi:10.5368/aedj.2011.3.1.3.5 POWER BLEACHING A SOLUTION FOR DISCOLOURED TEETH 1 Chandrasekhar M 2 Jaya prakash D patil 3 Ramesh T 1 Director, Professor and Head, Department of Conservative Dentistry 2
doi:10.5368/aedj.2011.3.1.3.5 POWER BLEACHING A SOLUTION FOR DISCOLOURED TEETH 1 Chandrasekhar M 2 Jaya prakash D patil 3 Ramesh T 1 Director, Professor and Head, Department of Conservative Dentistry 2
TOOTH WHITENING. Why would you want your teeth whitened?
 COSMETIC DENTISTRY TOOTH WHITENING COMPOSITE FILLINGS CROWNS BRIDGES RESTORATIONS - INLAY AND ONLAY VENEERS IMPLANTS FIXED ORTHODONTIC BRACES REMOVABLE APPLIANCES TOOTH WHITENING Why would you want your
COSMETIC DENTISTRY TOOTH WHITENING COMPOSITE FILLINGS CROWNS BRIDGES RESTORATIONS - INLAY AND ONLAY VENEERS IMPLANTS FIXED ORTHODONTIC BRACES REMOVABLE APPLIANCES TOOTH WHITENING Why would you want your
Fundamental & Preventive Curvatures of Teeth and Tooth Development. Lecture Three Chapter 15 Continued; Chapter 6 (parts) Dr. Margaret L.
 Fundamental & Preventive Curvatures of Teeth and Tooth Development Lecture Three Chapter 15 Continued; Chapter 6 (parts) Dr. Margaret L. Dennis Proximal contact areas Contact areas are on the mesial and
Fundamental & Preventive Curvatures of Teeth and Tooth Development Lecture Three Chapter 15 Continued; Chapter 6 (parts) Dr. Margaret L. Dennis Proximal contact areas Contact areas are on the mesial and
riva helping you help your patients
 riva helping you help your patients what is a glass ionomer? how will a dentist benefit from using glass ionomers? how will a patient benefit from their glass ionomer? Glass ionomer is the generic name
riva helping you help your patients what is a glass ionomer? how will a dentist benefit from using glass ionomers? how will a patient benefit from their glass ionomer? Glass ionomer is the generic name
A conservative restorative smile makeover
 C L I N I C A L A conservative restorative smile makeover Aneta Grzesinska 1 Introduction The patient was a 37-year-old female who presented to the practice requesting six porcelain veneers for her upper
C L I N I C A L A conservative restorative smile makeover Aneta Grzesinska 1 Introduction The patient was a 37-year-old female who presented to the practice requesting six porcelain veneers for her upper
Medical College of Georgia. School of Dentistry. Augusta, Georgia
 In Practice TOOTH WHITENING IN YOUR PRACTICE: : TREAT REATMENT TIME AND FEE SCHEDULES One area about which I often receive questions is the relationship between whitening time and fee schedules. Answering
In Practice TOOTH WHITENING IN YOUR PRACTICE: : TREAT REATMENT TIME AND FEE SCHEDULES One area about which I often receive questions is the relationship between whitening time and fee schedules. Answering
PREVALENCE AND FACTORS ASSOCIATED WITH TOOTH DISCOLORATION AMONG UAE UNIVERSITY STUDENTS
 GMJ GULF MEDICAL JOURNAL ORAL PROCEEDINGS PREVALENCE AND FACTORS ASSOCIATED WITH TOOTH Omar Barakat 1 *, Ardeshir Amirhosseini 1, Mohammad Ahmad 1, Sara Al-Fallahi 1, Talal Atassi 1, Walid El-Sayed 2 1Final
GMJ GULF MEDICAL JOURNAL ORAL PROCEEDINGS PREVALENCE AND FACTORS ASSOCIATED WITH TOOTH Omar Barakat 1 *, Ardeshir Amirhosseini 1, Mohammad Ahmad 1, Sara Al-Fallahi 1, Talal Atassi 1, Walid El-Sayed 2 1Final
Your Smile Wish. Find Answers to Your Smile Wish. Kathryn Alderman, DDS
 Your Smile Wish Find Answers to Your Smile Wish., Kathryn Alderman, DDS Smile Wish: Whiter, Brighter Smiles As you age, you may begin to notice that your teeth are not quite the same shade of white as
Your Smile Wish Find Answers to Your Smile Wish., Kathryn Alderman, DDS Smile Wish: Whiter, Brighter Smiles As you age, you may begin to notice that your teeth are not quite the same shade of white as
Draft 11/14/08. by Luke S. Kahng, CDT. Un d e r s ta n d i n g Zirconia Ba c k g r o u n d s. In t r o d u c t i o n. Naperville, IL
 Un d e r s ta n d i n g Zirconia Ba c k g r o u n d s for Custom Shade Matching by Luke S. Kahng, CDT Naperville, IL www.lsk121.com In t r o d u c t i o n Before beginning a case requiring the fabrication
Un d e r s ta n d i n g Zirconia Ba c k g r o u n d s for Custom Shade Matching by Luke S. Kahng, CDT Naperville, IL www.lsk121.com In t r o d u c t i o n Before beginning a case requiring the fabrication
Informed Consent for In-House Teeth Whitening Treatment
 Informed Consent for In-House Teeth Whitening Treatment INTRODUCTION This information has been given to me so that I can make an informed decision about having my teeth whitened. I may take as much time
Informed Consent for In-House Teeth Whitening Treatment INTRODUCTION This information has been given to me so that I can make an informed decision about having my teeth whitened. I may take as much time
SECRETS TO A LIFETIME OF WHITE TEETH. Pasha Hakimzadeh, DDS
 SECRETS TO A LIFETIME OF WHITE TEETH Pasha Hakimzadeh, DDS MEDICAL INFORMATION DISCLAIMER: This book is not intended as a substitute for the medical advice of physicians. The reader should regularly consult
SECRETS TO A LIFETIME OF WHITE TEETH Pasha Hakimzadeh, DDS MEDICAL INFORMATION DISCLAIMER: This book is not intended as a substitute for the medical advice of physicians. The reader should regularly consult
IPS Empress System Information for the. Laboratory. Confidence. Reliability. Esthetics. Empress IPS. System. The ultimate esthetic restorative system
 IPS Empress System Information for the Laboratory Confidence Reliability Esthetics IPS Empress The ultimate esthetic restorative system System A time-tested system offers new possibilities For nearly 20
IPS Empress System Information for the Laboratory Confidence Reliability Esthetics IPS Empress The ultimate esthetic restorative system System A time-tested system offers new possibilities For nearly 20
Take-Home Whitening. in vitro study. Benefits of ACP TAKE-HOME WHITENING
 Fluoride and Potassium Nitrate-Fluoride Whitening Agents: in vitro Caries Study Fluoride and Potassium Nitrate-Fluoride Whitening Agents: in vitro Caries Study in vitro study Hicks J and Flaitz C. Effects
Fluoride and Potassium Nitrate-Fluoride Whitening Agents: in vitro Caries Study Fluoride and Potassium Nitrate-Fluoride Whitening Agents: in vitro Caries Study in vitro study Hicks J and Flaitz C. Effects
DH220 Dental Materials
 DH220 Dental Materials Lecture #5 Prof. Lamanna RDH, MS Restorative Dentistry: Glass Ionomer Bird & Robinson p.740-741 I. Use Liner Base Luting agent Restorative material: Class III, V, & eroded/abraded
DH220 Dental Materials Lecture #5 Prof. Lamanna RDH, MS Restorative Dentistry: Glass Ionomer Bird & Robinson p.740-741 I. Use Liner Base Luting agent Restorative material: Class III, V, & eroded/abraded
International Symposium on Tooth Whitening: Evidence & Clinical Based Dr. Bruce A. Matis Introduction
 International Symposium on Tooth Whitening: Evidence & Clinical Based Dr. Bruce A. Matis www.bamatis.com 9-2017 The objective of this presentation is to increase your understanding of tooth whitening.
International Symposium on Tooth Whitening: Evidence & Clinical Based Dr. Bruce A. Matis www.bamatis.com 9-2017 The objective of this presentation is to increase your understanding of tooth whitening.
Clinical Studies - Tooth Whitening
 Clinical Studies - Tooth Whitening ADA Seal of Acceptance Certification Study for Discus Dental, Inc. Efficacy of Nite White Whitening Gel: A Clinical Trial Final Report, March 22, 1997 Investigation:
Clinical Studies - Tooth Whitening ADA Seal of Acceptance Certification Study for Discus Dental, Inc. Efficacy of Nite White Whitening Gel: A Clinical Trial Final Report, March 22, 1997 Investigation:
Applying color theory in clinical practice
 Problem solving in Dentistry: Applying color theory in clinical practice Asbjørn Jokstad Institute of Clinical Dentistry, University of Oslo, NORWAY Topics to cover 1. Discolored teeth - etiology 2. Materials
Problem solving in Dentistry: Applying color theory in clinical practice Asbjørn Jokstad Institute of Clinical Dentistry, University of Oslo, NORWAY Topics to cover 1. Discolored teeth - etiology 2. Materials
Therapeutic aesthetics
 C L I N I C A L Therapeutic aesthetics Linda Greenwall 1 With the success of tooth whitening treatments, several health benefits have emerged that can improve patients oral health (Li and Greenwall, 2013).
C L I N I C A L Therapeutic aesthetics Linda Greenwall 1 With the success of tooth whitening treatments, several health benefits have emerged that can improve patients oral health (Li and Greenwall, 2013).
COURSE CURRICULUM FOR AESTHETIC DENTISTRY
 COURSE CURRICULUM FOR AESTHETIC DENTISTRY Esthetic Dentistry is actually the fourth dimension in clinical dentistry. In addition to biologic, Physiologic, and mechanical factors, all of which must be understood
COURSE CURRICULUM FOR AESTHETIC DENTISTRY Esthetic Dentistry is actually the fourth dimension in clinical dentistry. In addition to biologic, Physiologic, and mechanical factors, all of which must be understood
Case Report. ISSN (Print)
 Scholars Journal of Dental Sciences (SJDS) Sch. J. Dent. Sci., 2016; 3(2):58-62 Scholars Academic and Scientific Publisher (An International Publisher for Academic and Scientific Resources) www.saspublisher.com
Scholars Journal of Dental Sciences (SJDS) Sch. J. Dent. Sci., 2016; 3(2):58-62 Scholars Academic and Scientific Publisher (An International Publisher for Academic and Scientific Resources) www.saspublisher.com
The Facts About Fillings
 Dental Board of California 2005 Evergreen Street, Suite 1550 Sacramento CA 95815 www.dbc.ca.gov Dental Materials Fact Sheet What About the Safety of Filling Materials? Patient health and the safety of
Dental Board of California 2005 Evergreen Street, Suite 1550 Sacramento CA 95815 www.dbc.ca.gov Dental Materials Fact Sheet What About the Safety of Filling Materials? Patient health and the safety of
Call the UK s leading Mailorder dental supplier
 Save s on WHITENING WHITENING Buy 10 Get 6 FREE BUY 2 GET 1 FREE Professional Whitening Strips NEW Call the UK s leading Mailorder dental supplier 01634 878787 www.kentexpress.co.uk sales@kentexpress.co.uk
Save s on WHITENING WHITENING Buy 10 Get 6 FREE BUY 2 GET 1 FREE Professional Whitening Strips NEW Call the UK s leading Mailorder dental supplier 01634 878787 www.kentexpress.co.uk sales@kentexpress.co.uk
A Clinical Evaluation of Bleaching Using Whitening Wraps and Strips
 Operative Dentistry, 2005, 30-5, 588-592 A Clinical Evaluation of Bleaching Using Whitening Wraps and Strips!!"#$%&'(#)#$"#*+,-.%/#)#0#1%/2# $#3.%/,+#)#04#5,67.&#)#84#*%.9+&&'#)#*#!.:%/# # "#$%!&'!($)*+##+,-!./($)01+2$!3$-1+#1)'4!5-67!!8)0-#*+##+,-!,)!)$(),%961+,-!,:!
Operative Dentistry, 2005, 30-5, 588-592 A Clinical Evaluation of Bleaching Using Whitening Wraps and Strips!!"#$%&'(#)#$"#*+,-.%/#)#0#1%/2# $#3.%/,+#)#04#5,67.&#)#84#*%.9+&&'#)#*#!.:%/# # "#$%!&'!($)*+##+,-!./($)01+2$!3$-1+#1)'4!5-67!!8)0-#*+##+,-!,)!)$(),%961+,-!,:!
Continually Fluoride Releasing Aesthetic Dental Restorative Material
 Continually Fluoride Releasing Aesthetic Dental Restorative Material Research is our best product Image provided by Dr. Sushil Koirala BEAUTIFIL II More than just filling BEAUTIFIL II stands out for its
Continually Fluoride Releasing Aesthetic Dental Restorative Material Research is our best product Image provided by Dr. Sushil Koirala BEAUTIFIL II More than just filling BEAUTIFIL II stands out for its
NIH Public Access Author Manuscript J Evid Based Dent Pract. Author manuscript; available in PMC 2015 June 01.
 NIH Public Access Author Manuscript Published in final edited form as: J Evid Based Dent Pract. 2014 June ; 14 Suppl: 70 76. doi:10.1016/j.jebdp.2014.02.006. Tooth Whitening: What We Now Know Clifton M.
NIH Public Access Author Manuscript Published in final edited form as: J Evid Based Dent Pract. 2014 June ; 14 Suppl: 70 76. doi:10.1016/j.jebdp.2014.02.006. Tooth Whitening: What We Now Know Clifton M.
Smile design with composites: A case study
 Clinical Smile design with composites: A case study Eduardo Mahn 1 and Christian Coachman 2 Adhesive dentistry has improved dramatically over the last decade. Not only are adhesives more reliable and easier
Clinical Smile design with composites: A case study Eduardo Mahn 1 and Christian Coachman 2 Adhesive dentistry has improved dramatically over the last decade. Not only are adhesives more reliable and easier
OUR EXPERIENCE WITH GRADIA DIRECT IN THE RESTORATION OF ANTERIOR TEETH
 ISSN: 1312-773X (Online) Journal of IMAB - Annual Proceeding (Scientific Papers) 2006, vol. 12, issue 2 OUR EXPERIENCE WITH GRADIA DIRECT IN THE RESTORATION OF ANTERIOR TEETH Snezhanka Topalova-Pirinska,
ISSN: 1312-773X (Online) Journal of IMAB - Annual Proceeding (Scientific Papers) 2006, vol. 12, issue 2 OUR EXPERIENCE WITH GRADIA DIRECT IN THE RESTORATION OF ANTERIOR TEETH Snezhanka Topalova-Pirinska,
SMILE DESIGNING. In a wide smile do your teeth show visible difference in their colour?
 SMILE DESIGNING What is done is smile designing? From minor changes to major repairs, we have myriad techniques to improve your smile. There are vast variety of options available to treat teeth that are
SMILE DESIGNING What is done is smile designing? From minor changes to major repairs, we have myriad techniques to improve your smile. There are vast variety of options available to treat teeth that are
Bleach n Smile. Chairside Bleaching
 Bleach n Smile Chairside Bleaching only 1 appointment up to 10 shades only 20 minutes* Shiny white teeth with the Bleach'n Smile Automix Set Intended for chairside use only Provide specific and efficient
Bleach n Smile Chairside Bleaching only 1 appointment up to 10 shades only 20 minutes* Shiny white teeth with the Bleach'n Smile Automix Set Intended for chairside use only Provide specific and efficient
Clinical UM Guideline
 Clinical UM Guideline Subject: Clinical Policy on Dental Prophylaxis Guideline #: 01-101 Current Effective Date: 03/24/2017 Status: New Last Review Date: 02/08/2017 Description This document addresses
Clinical UM Guideline Subject: Clinical Policy on Dental Prophylaxis Guideline #: 01-101 Current Effective Date: 03/24/2017 Status: New Last Review Date: 02/08/2017 Description This document addresses
Are your yellow teeth keeping you from smiling more?
 Are your yellow teeth keeping you from smiling more? In Just Over an Hour, You ll Wonder Why You Didn t Whiten Your Teeth Sooner With Zoom 2 Whitening. Give Me Less Than 20 Minutes of Your Time to Learn
Are your yellow teeth keeping you from smiling more? In Just Over an Hour, You ll Wonder Why You Didn t Whiten Your Teeth Sooner With Zoom 2 Whitening. Give Me Less Than 20 Minutes of Your Time to Learn
Influence of Bioactive Materials on Whitened Human Enamel Surface in vitro study
 Influence of Bioactive Materials on Whitened Human Enamel Surface Influence of Bioactive Materials on Whitened Human Enamel Surface in vitro study Pinheiro HB, Cardoso PEC, Universidade de São Paulo, São
Influence of Bioactive Materials on Whitened Human Enamel Surface Influence of Bioactive Materials on Whitened Human Enamel Surface in vitro study Pinheiro HB, Cardoso PEC, Universidade de São Paulo, São
ADHESIVE RECONSTRUCTION IN HELP OF THE ORTHODONTIC TREATMENT
 ISSN: 1312-773X (Online) Journal of IMAB - Annual Proceeding (Scientific Papers) 2006, vol. 12, issue 2 ADHESIVE RECONSTRUCTION IN HELP OF THE ORTHODONTIC TREATMENT Snezhanka Topalova-Pirinska 1, R. Pirinska
ISSN: 1312-773X (Online) Journal of IMAB - Annual Proceeding (Scientific Papers) 2006, vol. 12, issue 2 ADHESIVE RECONSTRUCTION IN HELP OF THE ORTHODONTIC TREATMENT Snezhanka Topalova-Pirinska 1, R. Pirinska
The Dental Board of California Dental Materials Fact Sheet Adopted by the Board on October 17, 2001
 The following document is the Dental Board of California s Dental Materials Fact Sheet. The Department of Consumer Affairs has no position with respect to the language of the Dental Materials Fact Sheet;
The following document is the Dental Board of California s Dental Materials Fact Sheet. The Department of Consumer Affairs has no position with respect to the language of the Dental Materials Fact Sheet;
Research Article A Clinical Study of the Effectiveness of Two Different 10% Carbamide Peroxide Bleaching Products: A 6-Month Followup
 International Dentistry Volume 211, Article ID 167525, 5 pages doi:1.1155/211/167525 Research Article A Clinical Study of the Effectiveness of Two Different 1% Carbamide Peroxide Bleaching Products: A
International Dentistry Volume 211, Article ID 167525, 5 pages doi:1.1155/211/167525 Research Article A Clinical Study of the Effectiveness of Two Different 1% Carbamide Peroxide Bleaching Products: A
The Facts About Fillings
 The Facts About Fillings DENTAL BOARD OF CALIFORNIA 1432 Howe Avenue Sacramento, California 95825 www.dbc.ca.gov Dental Materials Fact Sheet What About the Safety of Filling Materials? Patient health and
The Facts About Fillings DENTAL BOARD OF CALIFORNIA 1432 Howe Avenue Sacramento, California 95825 www.dbc.ca.gov Dental Materials Fact Sheet What About the Safety of Filling Materials? Patient health and
The tooth shade system that makes shade matching simple
 The tooth shade system that makes shade matching simple TESTIMONIALS TESTIMONIALS TESTIMONIALS TESTIMONIALS TESTIMONIALS TESTIMONIALS TESTIMONIALS Te s t i m o n i a l s The Vita 3D-Master Shade Guide
The tooth shade system that makes shade matching simple TESTIMONIALS TESTIMONIALS TESTIMONIALS TESTIMONIALS TESTIMONIALS TESTIMONIALS TESTIMONIALS Te s t i m o n i a l s The Vita 3D-Master Shade Guide
The Facts About Fillings
 Page 1 of 8 The Facts About Fillings DENTAL BOARD OF CALIFORNIA 1428 Howe Avenue Sacramento, California 95825 www.dbc.ca.gov Page 2 of 8 Dental Materials Fact Sheet What About the Safety of Filling Materials?
Page 1 of 8 The Facts About Fillings DENTAL BOARD OF CALIFORNIA 1428 Howe Avenue Sacramento, California 95825 www.dbc.ca.gov Page 2 of 8 Dental Materials Fact Sheet What About the Safety of Filling Materials?
Layering Guide for Labs. Lava Ceram. Overlay Porcelain for Lava Zirconia. Esthetic Restorations that are Truly Masterpieces
 Layering Guide for Labs Lava Ceram Overlay Porcelain for Lava Zirconia Esthetic Restorations that are Truly Masterpieces The product is the essence, based on experience. An excellent match for your high
Layering Guide for Labs Lava Ceram Overlay Porcelain for Lava Zirconia Esthetic Restorations that are Truly Masterpieces The product is the essence, based on experience. An excellent match for your high
Evidence Based Tooth Whitening Dr. Bruce A. Matis Introduction
 Evidence Based Tooth Whitening Dr. Bruce A. Matis www.bamatis.com The objective of this presentation is to increase your understanding of tooth whitening. We will do this by reviewing mostly clinical studies
Evidence Based Tooth Whitening Dr. Bruce A. Matis www.bamatis.com The objective of this presentation is to increase your understanding of tooth whitening. We will do this by reviewing mostly clinical studies
Direct composite restorations for large posterior cavities extended range of applications for high-performance materials
 Direct composite restorations for large posterior cavities extended range of applications for high-performance materials A case study by Ann-Christin Meier, Dr. med. dent., Stapelfeld, Germany When large
Direct composite restorations for large posterior cavities extended range of applications for high-performance materials A case study by Ann-Christin Meier, Dr. med. dent., Stapelfeld, Germany When large
Danville Family Dentist Dental Practice of Shailaja Singh DDS
 Danville Family Dentist Dental Practice of Shailaja Singh DDS The Facts About Dental Materials DENTAL BOARD OF CALIFORNIA 1432 Howe Avenue Sacramento, California 95825 www.dbc.ca.gov Dental Materials Fact
Danville Family Dentist Dental Practice of Shailaja Singh DDS The Facts About Dental Materials DENTAL BOARD OF CALIFORNIA 1432 Howe Avenue Sacramento, California 95825 www.dbc.ca.gov Dental Materials Fact
SPACE MAINTAINER. Multimedia Health Education. Disclaimer
 Disclaimer This movie is an educational resource only and should not be used to manage your health. All decisions about the management of premature loss of primary teeth and use of space maintainers must
Disclaimer This movie is an educational resource only and should not be used to manage your health. All decisions about the management of premature loss of primary teeth and use of space maintainers must
Structure of an Incisor
 MAMMALIAN TEETH Mammals have different types and shapes of teeth and they are thus termed Heterodonts. Those which have teeth of the same size and shapes are termed as Homodonts. In mammals teeth consist
MAMMALIAN TEETH Mammals have different types and shapes of teeth and they are thus termed Heterodonts. Those which have teeth of the same size and shapes are termed as Homodonts. In mammals teeth consist
Thomas E. Pillar, DDS
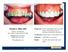 Thomas E. Pillar, DDS 5200 S. Cliff Avenue Sioux Falls, South Dakota 57108 Phone: 605.334.2607 Fax: 605.334.5402 TPillar@home.com Diagnosis: M & D restorations with recurrent decay; discoloration due to
Thomas E. Pillar, DDS 5200 S. Cliff Avenue Sioux Falls, South Dakota 57108 Phone: 605.334.2607 Fax: 605.334.5402 TPillar@home.com Diagnosis: M & D restorations with recurrent decay; discoloration due to
From the office of: Nahidh D. Andrews, DMD 3332 Portage Ave South Bend, IN (574) Are Your Teeth a Sensitive Subject?
 From the office of: Nahidh D. Andrews, DMD 3332 Portage Ave South Bend, IN 46628-3656 (574) 273-3900 Are Your Teeth a Sensitive Subject? A patient s guide to sensitive teeth and better oral care ARE YOUR
From the office of: Nahidh D. Andrews, DMD 3332 Portage Ave South Bend, IN 46628-3656 (574) 273-3900 Are Your Teeth a Sensitive Subject? A patient s guide to sensitive teeth and better oral care ARE YOUR
New Tooth Bleaching Research* Dr. Bruce A. Matis
 New Tooth Bleaching Research* Dr. Bruce A. Matis www.bamatis.com 11-8-07 Part 1 Need for Bleaching and In-office Bleaching Introduction Goal is to remove stain -Extrinsic Stain, which is deposited on the
New Tooth Bleaching Research* Dr. Bruce A. Matis www.bamatis.com 11-8-07 Part 1 Need for Bleaching and In-office Bleaching Introduction Goal is to remove stain -Extrinsic Stain, which is deposited on the
Applications in Dermatology, Dentistry and LASIK Eye Surgery using LASERs
 Applications in Dermatology, Dentistry and LASIK Eye Surgery using LASERs http://www.medispainstitute.com/menu_laser_tattoo.html http://www.life123.com/bm.pix/bigstockphoto_close_up_of_eye_surgery_catar_2264267.s600x600.jpg
Applications in Dermatology, Dentistry and LASIK Eye Surgery using LASERs http://www.medispainstitute.com/menu_laser_tattoo.html http://www.life123.com/bm.pix/bigstockphoto_close_up_of_eye_surgery_catar_2264267.s600x600.jpg
With and Without Tray Bleaching
 Operative Dentistry, 2009, 34-2, 142-149 A Clinical Evaluation of Two In-office Bleaching Regimens With and Without Tray Bleaching BA Matis MA Cochran G Wang GJ Eckert Clinical Relevance In-office tooth
Operative Dentistry, 2009, 34-2, 142-149 A Clinical Evaluation of Two In-office Bleaching Regimens With and Without Tray Bleaching BA Matis MA Cochran G Wang GJ Eckert Clinical Relevance In-office tooth
Near-UV light detection
 C L I N I C A L Near-UV light detection Javier Tapia Guadix 1 Near-UV light induced fluorescence has already proven to be very useful as an alternative to classic caries-detector dyes. However its potential
C L I N I C A L Near-UV light detection Javier Tapia Guadix 1 Near-UV light induced fluorescence has already proven to be very useful as an alternative to classic caries-detector dyes. However its potential
Dental Policy. Subject: Prophylaxis Guideline #: Publish Date: 03/15/2018 Status: Revised Last Review Date: 02/06/2018
 Dental Policy Subject: Prophylaxis Guideline #: 01-101 Publish Date: 03/15/2018 Status: Revised Last Review Date: 02/06/2018 Description This document addresses the procedure of dental prophylaxis for
Dental Policy Subject: Prophylaxis Guideline #: 01-101 Publish Date: 03/15/2018 Status: Revised Last Review Date: 02/06/2018 Description This document addresses the procedure of dental prophylaxis for
FIVE THINGS YOU NEED TO KNOW ABOUT GLASS IONOMERS
 FIVE THINGS YOU NEED TO KNOW ABOUT GLASS IONOMERS FIVE THINGS YOU NEED TO KNOW ABOUT GLASS IONOMERS Glass Ionomers Solve Clinicians Quandaries Amalgam fillings have been around for almost two centuries,
FIVE THINGS YOU NEED TO KNOW ABOUT GLASS IONOMERS FIVE THINGS YOU NEED TO KNOW ABOUT GLASS IONOMERS Glass Ionomers Solve Clinicians Quandaries Amalgam fillings have been around for almost two centuries,
RelyX Unicem Self-Adhesive Universal Resin Cement Frequently Asked Questions
 RelyX Unicem Self-Adhesive Universal Resin Cement Frequently Asked Questions Q1. What about the clinical history of The first restorations were cemented with RelyX Unicem cement in 2001. The excellent
RelyX Unicem Self-Adhesive Universal Resin Cement Frequently Asked Questions Q1. What about the clinical history of The first restorations were cemented with RelyX Unicem cement in 2001. The excellent
Introduction to Layering with Filtek Supreme Plus Universal Restorative. Filtek. Supreme Plus Universal Restorative
 Introduction to Layering with Filtek Supreme Plus Universal Restorative Filtek Supreme Plus Universal Restorative Introduction to Layering with Filtek Supreme Plus Universal Restorative TM. Multishade
Introduction to Layering with Filtek Supreme Plus Universal Restorative Filtek Supreme Plus Universal Restorative Introduction to Layering with Filtek Supreme Plus Universal Restorative TM. Multishade
It s more than a denture, It s a smile. IVOCLAR VIVADENT REMOVABLE. More than dentures. Tooth Line Portfolio
 It s more than a denture, It s a smile. IVOCLAR VIVADENT REMOVABLE More than dentures. Tooth Line Portfolio 9458.indd 1 12/8/16 10:28 AM Ivoclar Vivadent Removable Master Class Collection The ultimate
It s more than a denture, It s a smile. IVOCLAR VIVADENT REMOVABLE More than dentures. Tooth Line Portfolio 9458.indd 1 12/8/16 10:28 AM Ivoclar Vivadent Removable Master Class Collection The ultimate
THE CLASSIC COMPOSITE FOR EXQUISITE ESTHETICS
 THE CLASSIC COMPOSITE FOR EXQUISITE ESTHETICS Final Layer Option For a single layer, place approximately 0.5mm of Pearl Frost (PF) or Pearl Neutral (PN) to enhance the restoration and allow the underlying
THE CLASSIC COMPOSITE FOR EXQUISITE ESTHETICS Final Layer Option For a single layer, place approximately 0.5mm of Pearl Frost (PF) or Pearl Neutral (PN) to enhance the restoration and allow the underlying
P r o f e s s i o n a l T e e t h W h i t e n i n g Manual
 P r o f e s s i o n a l T e e t h W h i t e n i n g Manual Legislation Hygiene & Safety After Whitening Care Insurance What Causes Teeth Staining How Does Whitening Work How White Can You Go Risks & Sensitivities
P r o f e s s i o n a l T e e t h W h i t e n i n g Manual Legislation Hygiene & Safety After Whitening Care Insurance What Causes Teeth Staining How Does Whitening Work How White Can You Go Risks & Sensitivities
Pulpal Protection: bases, liners, sealers, caries control Module A: Basic Concepts
 Readings: Fundamentals of Operative Dentistry, 3 nd Edition; Summitt, et al Chapters 5, 6 and 8 Pulpal Protection: bases, liners, sealers, caries control Module A: Basic Concepts REST 528A Operative #3A
Readings: Fundamentals of Operative Dentistry, 3 nd Edition; Summitt, et al Chapters 5, 6 and 8 Pulpal Protection: bases, liners, sealers, caries control Module A: Basic Concepts REST 528A Operative #3A
OliNano Seal Professional prophylaxis for long-term protection
 Professional prophylaxis for long-term protection NEW The patented formula of silicone polymer NANO Technology General information Dental health is one of the main factors to maintain overall health, and
Professional prophylaxis for long-term protection NEW The patented formula of silicone polymer NANO Technology General information Dental health is one of the main factors to maintain overall health, and
Art of Smile. Cosmetic Dentistry. Youngman Dental Clinic Dr. Terry Youngman 3400 E. McDowell Rd. Phoenix, AZ (602)
 Art of Smile Cosmetic Dentistry Youngman Dental Clinic Dr. Terry Youngman 3400 E. McDowell Rd. Phoenix, AZ 85008 (602) 225-9090 Transform your life when you transform your smile! New techniques and products
Art of Smile Cosmetic Dentistry Youngman Dental Clinic Dr. Terry Youngman 3400 E. McDowell Rd. Phoenix, AZ 85008 (602) 225-9090 Transform your life when you transform your smile! New techniques and products
Essentials of. Dental Assisting. Edition 6. Debbie S. Robinson Doni L. Bird
 Essentials of Dental Assisting Edition 6 Debbie S. Robinson Doni L. Bird CHAPTER21 Restorative Procedures http://evolve.elsevier.com/robinson/essentials/ LEARNING OBJECTIVES KEY TERMS 1. Pronounce, define,
Essentials of Dental Assisting Edition 6 Debbie S. Robinson Doni L. Bird CHAPTER21 Restorative Procedures http://evolve.elsevier.com/robinson/essentials/ LEARNING OBJECTIVES KEY TERMS 1. Pronounce, define,
SHOFU BLOCK & DISK CAD/CAM CERAMIC-BASED RESTORATIVE. Visit or call mm x 12mm x 16mm. 98mm x 14mm. 12mm x 14mm x 18mm
 SHOFU BLOCK & DISK CAD/CAM CERAMIC-BASED RESTORATIVE 1mm x 12mm x 16mm 12mm x 14mm x 18mm 98mm x 14mm Visit www.shofu.com or call 8.827.4638 Created through a rigorous manufacturing process, Shofu Blocks
SHOFU BLOCK & DISK CAD/CAM CERAMIC-BASED RESTORATIVE 1mm x 12mm x 16mm 12mm x 14mm x 18mm 98mm x 14mm Visit www.shofu.com or call 8.827.4638 Created through a rigorous manufacturing process, Shofu Blocks
Informed Consent. (Initials )
 Informed Consent 1. EXAMINATIONS AND X-RAYS I understand that the initial visit may require radiographs in order to complete the examination, diagnosis and treatment plan. I understand I am to have work
Informed Consent 1. EXAMINATIONS AND X-RAYS I understand that the initial visit may require radiographs in order to complete the examination, diagnosis and treatment plan. I understand I am to have work
VivaStyle. Serious Whitening. A bright smile connects
 VivaStyle Serious Whitening A bright smile connects VivaStyle A comprehensive system for esthetic dentistry Naturally white teeth a desire frequently voiced by patients. In order to meet the expectations
VivaStyle Serious Whitening A bright smile connects VivaStyle A comprehensive system for esthetic dentistry Naturally white teeth a desire frequently voiced by patients. In order to meet the expectations
Evidence Based Tooth Whitening Dr. Bruce A. Matis Introduction
 Evidence Based Tooth Whitening Dr. Bruce A. Matis www.bamatis.com The objective of this presentation is to increase your understanding of tooth whitening. We will do this by reviewing the clinical studies
Evidence Based Tooth Whitening Dr. Bruce A. Matis www.bamatis.com The objective of this presentation is to increase your understanding of tooth whitening. We will do this by reviewing the clinical studies
Amaris. You want outstanding esthetic restorations with the WOW-EFFECT
 The Composite For Simplified Esthetics The Simplified Esthetic Concept You want outstanding esthetic restorations with the WOW-EFFECT You do not have the time to work on a patient for hours You do not
The Composite For Simplified Esthetics The Simplified Esthetic Concept You want outstanding esthetic restorations with the WOW-EFFECT You do not have the time to work on a patient for hours You do not
COLOURS ON CERAMIC PRODUCTS FOR DENTAL USE
 COLOURS ON CERAMIC PRODUCTS FOR DENTAL USE BTEETH : COLOUR LINE FOR DENTAL CERAMICS ZIRCONIA The technological evolution of dental laboratories will be increasingly based on the use of innovative materials
COLOURS ON CERAMIC PRODUCTS FOR DENTAL USE BTEETH : COLOUR LINE FOR DENTAL CERAMICS ZIRCONIA The technological evolution of dental laboratories will be increasingly based on the use of innovative materials
aura INSTRUCTIONS FOR USE ULTRA UNIVERSAL RESTORATIVE MATERIAL ULTRA UNIVERSAL RESTAURATIONS MATERIAL MATÉRIEL DE RESTAURATION ULTRA UNIVERSEL
 INSTRUCTIONS FOR USE aura ULTRA UNIVERSAL RESTORATIVE MATERIAL ULTRA UNIVERSAL RESTAURATIONS MATERIAL MATÉRIEL DE RESTAURATION ULTRA UNIVERSEL MATERIALE DA RICOSTRUZIONE ULTRA UNIVERSALE MATERIAL RESTAURADOR
INSTRUCTIONS FOR USE aura ULTRA UNIVERSAL RESTORATIVE MATERIAL ULTRA UNIVERSAL RESTAURATIONS MATERIAL MATÉRIEL DE RESTAURATION ULTRA UNIVERSEL MATERIALE DA RICOSTRUZIONE ULTRA UNIVERSALE MATERIAL RESTAURADOR
BLOCK RANGE CAD. IPS e.max
 BLOCK RANGE 682185 / en / 2018-01-24 www.ivoclarvivadent.com Ivoclar Vivadent AG Bendererstr. 2 9494 Schaan Liechtenstein Tel.: +423 235 35 35 Fax: +423 235 33 60 FOR A COMPREHENSIVE RANGE OF INDICATIONS
BLOCK RANGE 682185 / en / 2018-01-24 www.ivoclarvivadent.com Ivoclar Vivadent AG Bendererstr. 2 9494 Schaan Liechtenstein Tel.: +423 235 35 35 Fax: +423 235 33 60 FOR A COMPREHENSIVE RANGE OF INDICATIONS
Philips Zoom! at home whitening
 Want whiter teeth? Philips Zoom! at home whitening A visibly whiter smile after 3 days 1 Ask your dentist or hygienist about Philips Zoom! whitening Why consider dental whitening? Your smile is the way
Want whiter teeth? Philips Zoom! at home whitening A visibly whiter smile after 3 days 1 Ask your dentist or hygienist about Philips Zoom! whitening Why consider dental whitening? Your smile is the way
SCD Case Study. Background
 SCD Case Study Background A female aged over 70 presented with an unremarkable medical history seeking a comprehensive examination as the last dental examination was over four years ago. The patient is
SCD Case Study Background A female aged over 70 presented with an unremarkable medical history seeking a comprehensive examination as the last dental examination was over four years ago. The patient is
Tooth Whitening: Comprehensive Review and Clinical Guidelines. Updated credit hours (2 CEs)
 The Academy of Dental Learning & OSHA Training 1101 Sibley Memorial Hwy Ste. 211 St. Paul, MN 55118 (800) 522-1207 CESupport@DentalLearning.org Tooth Whitening: Comprehensive Review and Clinical Guidelines
The Academy of Dental Learning & OSHA Training 1101 Sibley Memorial Hwy Ste. 211 St. Paul, MN 55118 (800) 522-1207 CESupport@DentalLearning.org Tooth Whitening: Comprehensive Review and Clinical Guidelines
Colour Communication.
 Colour Communication. Understanding and expressing colour to your lab to achieve the best results. I by no means claim to be an expert on colour or even on communication, as my technicians will tell you.
Colour Communication. Understanding and expressing colour to your lab to achieve the best results. I by no means claim to be an expert on colour or even on communication, as my technicians will tell you.
Clinical Studies - Tooth Whitening
 Discus Dental Clinical Data and Safety Clinical Studies Tooth Whitening Evaluation of Tray Fabrication Design and Effects on Vital Tooth Bleaching Investigation: M. Bosma J. Bowman W. Dorfman K. Soll Hill
Discus Dental Clinical Data and Safety Clinical Studies Tooth Whitening Evaluation of Tray Fabrication Design and Effects on Vital Tooth Bleaching Investigation: M. Bosma J. Bowman W. Dorfman K. Soll Hill
Digital Smile Design using the M Proportions and GPS 2D to 3D Digital Facebow: Clinical Case 1
 Digital Smile Design using the M Proportions and GPS 2D to 3D Digital Facebow: Clinical Case 1 Dr Alain Méthot Dr Marco Delcorso A young female patient previously suffering from gastric reflux came to
Digital Smile Design using the M Proportions and GPS 2D to 3D Digital Facebow: Clinical Case 1 Dr Alain Méthot Dr Marco Delcorso A young female patient previously suffering from gastric reflux came to
Patient demand for esthetic dentistry
 PROFILE Masters of Esthetic Dentistry Keys to Success in Creating Esthetic Class IV Restorations Robert C. Margeas, DDS Current occupation Private Practice Adjunct Professor, Department of Operative Dentistry,
PROFILE Masters of Esthetic Dentistry Keys to Success in Creating Esthetic Class IV Restorations Robert C. Margeas, DDS Current occupation Private Practice Adjunct Professor, Department of Operative Dentistry,
dental fillings facts About the brochure:
 dental fillings facts About the brochure: Your dentist is dedicated to protecting and improving oral health while providing safe dental treatment. This fact sheet provides information you need to discuss
dental fillings facts About the brochure: Your dentist is dedicated to protecting and improving oral health while providing safe dental treatment. This fact sheet provides information you need to discuss
Celtra, Cercon, Celtra Ceram, Universal Stain and Glaze. All-ceramic solutions for every need. Brochure for the dental laboratory
 Celtra, Cercon, Celtra Ceram, Universal Stain and Glaze All-ceramic solutions for every need Brochure for the dental laboratory Content Celtra Ceram High esthetic veneering 4 Celtra Press Monolithic stained
Celtra, Cercon, Celtra Ceram, Universal Stain and Glaze All-ceramic solutions for every need Brochure for the dental laboratory Content Celtra Ceram High esthetic veneering 4 Celtra Press Monolithic stained
For the Perfect Class V and All Cervical Area Gingival Margins when Placing Direct Composites, Create an Injection Molding Matrix
 Cronicon OPEN ACCESS EC DENTAL SCIENCE Case Report For the Perfect Class V and All Cervical Area Gingival Margins when Placing Direct Composites, Create an Injection Molding Paul C Belvedere* Adjunct Professor,
Cronicon OPEN ACCESS EC DENTAL SCIENCE Case Report For the Perfect Class V and All Cervical Area Gingival Margins when Placing Direct Composites, Create an Injection Molding Paul C Belvedere* Adjunct Professor,
BLOCK RANGE CAD. IPS e.max
 BLOCK RANGE 682185 / en / 2015-11-16 www.ivoclarvivadent.com Ivoclar Vivadent AG Bendererstr. 2 9494 Schaan Liechtenstein Tel.: +423 235 35 35 Fax: +423 235 33 60 FOR A COMPREHENSIVE RANGE OF INDICATIONS
BLOCK RANGE 682185 / en / 2015-11-16 www.ivoclarvivadent.com Ivoclar Vivadent AG Bendererstr. 2 9494 Schaan Liechtenstein Tel.: +423 235 35 35 Fax: +423 235 33 60 FOR A COMPREHENSIVE RANGE OF INDICATIONS
