Phototherapy, Photochemotherapy and Photodynamic Therapy for Dermatologic Conditions
|
|
|
- Joshua Briggs
- 5 years ago
- Views:
Transcription
1 Last Review Date: October 12, 2018 Number: MG.MM.ME.27j Medical Guideline Disclaimer Property of EmblemHealth. All rights reserved. The treating physician or primary care provider must submit to EmblemHealth the clinical evidence that the patient meets the criteria for the treatment or surgical procedure. Without this documentation and information, EmblemHealth will not be able to properly review the request for prior authorization. The clinical review criteria expressed below reflects how EmblemHealth determines whether certain services or supplies are medically necessary. EmblemHealth established the clinical review criteria based upon a review of currently available clinical information (including clinical outcome studies in the peer-reviewed published medical literature, regulatory status of the technology, evidence-based guidelines of public health and health research agencies, evidence-based guidelines and positions of leading national health professional organizations, views of physicians practicing in relevant clinical areas, and other relevant factors). EmblemHealth expressly reserves the right to revise these conclusions as clinical information changes, and welcomes further relevant information. Each benefit program defines which services are covered. The conclusion that a particular service or supply is medically necessary does not constitute a representation or warranty that this service or supply is covered and/or paid for by EmblemHealth, as some programs exclude coverage for services or supplies that EmblemHealth considers medically necessary. If there is a discrepancy between this guideline and a member's benefits program, the benefits program will govern. In addition, coverage may be mandated by applicable legal requirements of a state, the Federal Government or the Centers for Medicare & Medicaid Services (CMS) for Medicare and Medicaid members. All coding and web site links are accurate at time of publication. EmblemHealth Services Company LLC, ( EmblemHealth ) has adopted the herein policy in providing management, administrative and other services to HIP Health Plan of New York, HIP Insurance Company of New York, Group Health Incorporated and GHI HMO Select, related to health benefit plans offered by these entities. All of the aforementioned entities are affiliated companies under common control of EmblemHealth Inc. Definitions Phototherapy Photochemotherapy Psoralen and ultraviolet A (PUVA) The application of ultraviolet light, or actinotherapy consists of exposure to nonionizing radiation. The treatment may involve exposure to any: Ultraviolet B (UVB) Ultraviolet A (UVA) Combined UVB and UVA delivered using a broad or narrow-beamed laser PUVA utilizes UVA radiation in combination with a photosensitizing chemical that increases the skin s sensitivity to the UVA. Photodynamic Therapy (PDT) PDT is a multi-step (typically 2-day) process that consists of the application of a topical photosensitizer cream followed by a laser light source (e.g., methyl aminolevulinate hydrochloride [MAL] that is accompanied by a red light; 5-aminolevulinic acid [5-ALA] or by a blue). Guideline Phototherapy, PUVA and PDT are considered medically necessary for certain dermatologic conditions refractory to topical or systemic drug therapies when any of the applicable criteria sets in Tables 1 4 are met. (Note: For case-by-case consideration of vitiligo treatment; see Table 4)
2 Page 2 of 7 Table 1 Phototherapy 1. Allergic contact dermatitis 2. Atopic dermatitis (moderate severe) 3. Chronic urticaria 4. Dermatologic manifestations of graft vs host disease 5. Eczema 6. Granuloma annulare 7. Lichen planus 8. Mycosis fungoides (cutaneous T-cell lymphoma) 9. Nummular dermatitis 10. Photodermatosis 11. Pityriasis lichenoides 12. Pityriasis rosea 13. Pruritic eruptions of HIV infection 14. Pruritus 15. Parapsoriasis 16. Psoriasis Home Phototherapy (UVB) Units (DME benefit required) Coverage for members with moderate to severe persistent psoriasis covering at least 20% of the body surface may be provided for the purchase of a home UVB Phototherapy unit. All of the following criteria must be met: 1. Documentation of effective psoriasis suppression as a result of at least 6 months of UVB treatment, whereby the continuation of home-uvb would be construed as a reasonable means to deter exacerbations. 2. Physician documentation of medical necessity, which includes: Severity description, e.g., if there is involvement of the palms, soles, or intertriginous areas, the percent of the affected area involved, and the associated disability should be part of the record. A prescription describing the UVB exposure protocol. A follow-up plan to determine treatment effectiveness, i.e., office visit frequency. 3. Demonstration of patient proficiency in the use of UVB with the understanding of the necessity of physician communication with the occurrence of any unexpected side effects. 4. History of ineffective (or intolerance to) treatments with multiple topical agents or systemic therapy.
3 Page 3 of 7 Table 2 PUVA 1. Acute/chronic pityriasis lichenoides 2. Atopic dermatitis (moderate severe) 3. Chronic urticaria 4. Dermatologic manifestations of graft-versus-host disease 5. Eczema (severe) 6. Granuloma annulare 7. Lichen planus 8. Morphea and localized skin lesions associated with scleroderma 9. Mycosis fungoides (cutaneous T-cell lymphoma) 10. Parapsoriasis (severe) 11. Psoriasis (severe) Table 3 PDT Presence of either of the following lesions that have failed to adequately respond to 3 weeks of topical 5-fluorouracil, imiquimod, Diclofenac or cryosurgery: 1. Non-hyperkeratotic actinic keratoses lesions on the face or scalp. 2. Actinic cheilitis, also known as solar cheilitis, sailor s lip or farmer s lip. Note: A 2 nd treatment post 8 weeks of the initial therapy may be necessary for any lesions that fail to respond to therapy. Table 4 Treatment of Vitiligo On a case-by-case basis, coverage consideration will be given for excimer laser, PUVA, UVB light (alone or in combination with other treatment modalities) when treatment is confined to areas of the face, neck or hands only. Prior to Medical Director consideration, substantiating documentation must first be submitted for review; these include: 1. Progress notes indicative of the following: a. Baseline skin color. b. Treatment history; documented failure of adherent 3-month trial of both: i. high-potency (Class II steroids) ii. Protopic. c. Extent and distribution of vitiligo to the face, neck and or hands. 2. Photographic evidence. Limitations/Exclusions 1. Phototherapy, PUVA or PDT is not considered medically necessary for any indications other than those listed above. 2. More than 2 courses of PDT treatments per year are not considered medically necessary, as effectiveness beyond this timeframe has not been established. 3. Requests for coverage of more than 30 units of Phototherapy, PUVA or PDT per course of treatment must be accompanied by documentation that substantiates medical necessity. 4. Grenz ray therapy is not considered medically necessary for any indications, as it is considered investigational. 5. Prior authorization is required for all procedures listed in this policy.
4 Page 4 of 7 Revision History Oct. 12, 2018 Sept. 21, 2017 Feb. 10, 2017 Added allergic contact dermatitis and nummular dermatitis to phototherapy table. Clarified that medical documentation substantiating medical necessity must be submitted for coverage consideration of > 30 units per course of treatment for phototherapy/photochemotherapy. Added PUVA and UVB light to vitiligo table for case-by-case consideration. Applicable Procedure Codes Photodynamic therapy by external application of light to destroy premalignant and/or malignant lesions of the skin and adjacent mucosa (eg, lip) by activation of photosensitive drug(s), each phototherapy exposure session Photodynamic therapy by external application of light to destroy premalignant lesions of the skin and adjacent mucosa with application and illumination/activation of photosensitizing drug(s) provided by a physician or other qualified health care professional, per day (Eff. 01/01/2018) Photochemotherapy; tar and ultraviolet B (Goeckerman treatment) or petrolatum and ultraviolet B Photochemotherapy; psoralens and ultraviolet A (PUVA) Photochemotherapy (Goeckerman and/or PUVA) for severe photoresponsive dermatoses requiring at least four to eight hours of care under direct supervision of the physician (includes application of medication and dressings) Laser treatment for inflammatory skin disease (psoriasis); total area less than 250 sq cm (The appropriate ICD-10 codes to use are: L40.0, L40.1, L40.2, L40.4, L40.8, L40.9, L41.3, L41.4, L41.5, L41.8 and L41.9) Laser treatment for inflammatory skin disease (psoriasis); 250 sq cm to 500 sq cm (The appropriate ICD-10 codes to use are: L40.0, L40.1, L40.2, L40.4, L40.8, L40.9, L41.3, L41.4, L41.5, L41.8 and L41.9) Laser treatment for inflammatory skin disease (psoriasis); over 500 sq cm (The appropriate ICD-10 codes to use are: L40.0, L40.1, L40.2, L40.4, L40.8, L40.9, L41.3, L41.4, L41.5, L41.8 and L41.9) Unlisted special dermatological service or procedure E0691 E0692 E0693 E0694 A4633 J7308 J7309 Ultraviolet light therapy system, includes bulbs/lamps, timer and eye protection; treatment area 2 sq ft or less Ultraviolet light therapy system panel, includes bulbs/lamps, timer, and eye protection, 4 ft. panel Ultraviolet light therapy system panel, includes bulbs/lamps, timer, and eye protection, 6 ft. panel Ultraviolet multidirectional light therapy system in 6 ft. cabinet, includes bulbs/lamps, timer, and eye protection Replacement bulb/lamp for ultraviolet light therapy system, each Aminolevulinic acid HCl for topical administration, 20%, single unit dosage form (354 mg) Methyl aminolevulinate (MAL) for topical administration, 16.8%, 1 g J7345 Aminolevulinic acid hcl for topical administration, 10% gel, 10 mg (Eff. 01/01/2018)
5 Page 5 of 7 Applicable Diagnosis Codes C84.00 Mycosis fungoides, unspecified site C84.01 Mycosis fungoides, lymph nodes of head, face, and neck L20.81 Atopic neurodermatitis L20.82 Flexural eczema L20.84 Intrinsic (allergic) eczema L20.89 Other atopic dermatitis L20.9 Atopic dermatitis, unspecified L23.1 Allergic contact dermatitis due to adhesives L23.3 Allergic contact dermatitis due to drugs in contact with skin L23.5 Allergic contact dermatitis due to other chemical products L24.4 Irritant contact dermatitis due to drugs in contact with skin L24.5 Irritant contact dermatitis due to other chemical products L25.1 Unspecified contact dermatitis due to drugs in contact with skin L25.3 Unspecified contact dermatitis due to other chemical products L29.8 Other pruritus L29.9 Pruritus, unspecified L30.0 Nummular dermatitis L30.4 Erythema intertrigo L30.5 Pityriasis alba L40.0 Psoriasis vulgaris L40.1 Generalized pustular psoriasis L40.2 Acrodermatitis continua L40.4 Guttate psoriasis L40.8 Other psoriasis L40.9 Psoriasis, unspecified L41.0 Pityriasis lichenoides et varioliformis acuta L41.1 Pityriasis lichenoides chronica L41.3 Small plaque parapsoriasis L41.4 Large plaque parapsoriasis L41.5 Retiform parapsoriasis L41.8 Other parapsoriasis L41.9 Parapsoriasis, unspecified L42 Pityriasis rosea
6 Page 6 of 7 L43.0 Hypertrophic lichen planus L43.1 Bullous lichen planus L43.3 Subacute (active) lichen planus L43.8 Other lichen planus L43.9 Lichen planus, unspecified L50.6 Contact urticaria L50.8 Other urticaria L56.2 Photocontact dermatitis [berloque dermatitis] L56.3 Solar urticaria L56.5 Disseminated superficial actinic porokeratosis (DSAP) L56.8 Other specified acute skin changes due to ultraviolet radiation L56.9 Acute skin change due to ultraviolet radiation, unspecified L57.0 Actinic keratosis L66.1 Lichen planopilaris L80 Vitiligo L90.0 Lichen sclerosus et atrophicus L92.0 Granuloma annulare L94.0 Localized scleroderma [morphea] References Alabdulkareem AS, Abahussein AA, Okoro A. Minimal benefit from photochemotherapy for alopecia areata. Int J Dermatol. 1996;35: Cather J, Menter A. Novel therapies for psoriasis. Am J Clin Dermatol. 2002;3(3): Davis MD, McEvoy MT, el-azhary RA. Topical psoralen-ultraviolet A therapy for palmoplantar dermatoses: experience with 35 consecutive patients. Mayo Clin Proc. 1998;73: Dutz J. Treatment options for localized scleroderma. Skin Therapy Lett. 2000;5(2):3-5. Gordon PM, Diffey BL, Matthews JN, Farr PM. A randomized comparison of narrow-band TL-01 phototherapy and PUVA photochemotherapy for psoriasis. J Am Acad Dermatol. 1999;41(5 Pt 1): Griffiths CE, Clark CM, Chalmers RJ, Li Wan Po A, Williams HC. A systematic review of treatments for severe psoriasis. Health Technol Assess. 2000;4(40): Guidelines of care for phototherapy and photochemotherapy. American Academy of Dermatology Committee on Guidelines of Care. J Am Acad Dermatol. 1994;31: Hawk A, English JC 3rd. Localized and systemic scleroderma. Semin Cutan Med Surg. March 2001;20: Hayes, Inc. Laser Therapy for Psoriasis. Lansdale, Pa: Winifred S. Hayes, Inc.; July Search updated February 28, Honig B, Morison WL, Karp D. Photochemotherapy beyond psoriasis. J Am Acad Dermatol. 1994;31(5 Pt 1): Millard TP, Hawk JL. Photosensitivity disorders: cause, effect and management. Am J Clin Dermatol. 2002;3: Momtaz K, Fitzpatrick TB. The benefits and risks of long-term PUVA photochemotherapy. Dermatol Clin. 1998;16:
7 Page 7 of 7 Sapadin AN, Fleischmajer R. Treatment of scleroderma. Arch Dermatol. January 2002;138: Saricaoglu H, Karadogan SK, Baskan EB, Tunali S. Narrowband UVB therapy in the treatment of lichen planus. Photodermatol Photoimmunol Photomed. October 2003;19: Specialty-matched clinical peer review. Storbeck K, Holzle E, Schurer N, Lehmann P, Plewig G. Narrow-band UVB (311 nm) versus conventional broad-band UVB with and without dithranol in phototherapy for psoriasis. J Am Acad Dermatol. 1993;28(2 Pt 1): Taylor CR, Hawk JL. PUVA treatment of alopecia areata partialis, totalis and universalis: audit of 10 years' experience at St. John's Institute of Dermatology. Br J Dermatol. 1995;133: Vogelsang GB, Wolff D, Altomonte V, et al. Treatment of chronic graft-versus-host disease with ultraviolet irradiation and psoralen (PUVA). Bone Marrow Transplant. 1996;17: Wolff K. Treatment of cutaneous mastocytosis. Int Arch Allergy Immunol. Feburary 2002;127: Zanolli MD. Psoriasis and Reiter's syndrome. In: Sams WM Jr, Lynch PJ, eds. Principles and Practice of Dermatology. 2nd ed. New York, NY: Churchill Livingstone Inc. 1996:
Phototherapy, Photochemotherapy and Photodynamic Therapy for Dermatologic Conditions
 Last Review Date: September 21, 2017 Number: MG.MM.ME.27iv2 Medical Guideline Disclaimer Property of EmblemHealth. All rights reserved. The treating physician or primary care provider must submit to EmblemHealth
Last Review Date: September 21, 2017 Number: MG.MM.ME.27iv2 Medical Guideline Disclaimer Property of EmblemHealth. All rights reserved. The treating physician or primary care provider must submit to EmblemHealth
Photochemotherapy MM /09/2004. HMO; PPO; QUEST Integration June 1, 2016 Section: Medicine Place(s) of Service: Home; Office
 Photochemotherapy Policy Number: Original Effective Date: MM.02.015 11/09/2004 Line(s) of Business: Current Effective Date: HMO; PPO; QUEST Integration June 1, 2016 Section: Medicine Place(s) of Service:
Photochemotherapy Policy Number: Original Effective Date: MM.02.015 11/09/2004 Line(s) of Business: Current Effective Date: HMO; PPO; QUEST Integration June 1, 2016 Section: Medicine Place(s) of Service:
Photochemotherapy MM /09/2004. HMO; PPO; QUEST Integration 08/25/2017 Section: Medicine Place(s) of Service: Home; Office
 Photochemotherapy Policy Number: Original Effective Date: MM.02.015 11/09/2004 Line(s) of Business: Current Effective Date: HMO; PPO; QUEST Integration 08/25/2017 Section: Medicine Place(s) of Service:
Photochemotherapy Policy Number: Original Effective Date: MM.02.015 11/09/2004 Line(s) of Business: Current Effective Date: HMO; PPO; QUEST Integration 08/25/2017 Section: Medicine Place(s) of Service:
Clinical Policy: Phototherapy and Photochemotherapy for Dermatological Conditions Reference Number: CP.MP. 441
 Clinical Policy: Phototherapy and Photochemotherapy for Dermatological Conditions Reference Number: CP.MP. 441 Effective Date: November 2008 Last Review Date: January 2017 See Important Reminder at the
Clinical Policy: Phototherapy and Photochemotherapy for Dermatological Conditions Reference Number: CP.MP. 441 Effective Date: November 2008 Last Review Date: January 2017 See Important Reminder at the
Phototherapy and Photochemotherapy Treatment (Ultraviolet A [PUVA] and B [UBV])
![Phototherapy and Photochemotherapy Treatment (Ultraviolet A [PUVA] and B [UBV]) Phototherapy and Photochemotherapy Treatment (Ultraviolet A [PUVA] and B [UBV])](/thumbs/89/99382632.jpg) Origination: 09/27/07 Revised: 08/2/17 Annual Review: 11/2/17 Purpose: To provide Phototherapy and Photochemotherapy Treatment (PUVA and UBV) guidelines for the Medical Department staff to reference when
Origination: 09/27/07 Revised: 08/2/17 Annual Review: 11/2/17 Purpose: To provide Phototherapy and Photochemotherapy Treatment (PUVA and UBV) guidelines for the Medical Department staff to reference when
Corporate Medical Policy
 Corporate Medical Policy Ultraviolet Light Therapy in the Home Setting(UVB) File Name: Origination: Last CAP Review: Next CAP Review: Last Review: ultraviolet_light_therapy_in_the_home 3/1996 11/2017 11/2018
Corporate Medical Policy Ultraviolet Light Therapy in the Home Setting(UVB) File Name: Origination: Last CAP Review: Next CAP Review: Last Review: ultraviolet_light_therapy_in_the_home 3/1996 11/2017 11/2018
ICD 10 Codes. L82.1 Seborrheic Keratosis L82.0 Irritated Seborrheic Keratosis
 Leon H. Kircik M.D. Clinical Associate Professor of Dermatology Indiana University School of Medicine Mount Sinai Medical Center, New York, NY Physicians Skin Care, PLLC Louisville, KY 1 ICD 10 Codes L82.1
Leon H. Kircik M.D. Clinical Associate Professor of Dermatology Indiana University School of Medicine Mount Sinai Medical Center, New York, NY Physicians Skin Care, PLLC Louisville, KY 1 ICD 10 Codes L82.1
MEDICAL POLICY SUBJECT: LIGHT AND LASER THERAPIES FOR DERMATOLOGIC CONDITIONS
 MEDICAL POLICY SUBJECT: LIGHT AND LASER THERAPIES FOR PAGE: 1 OF: 9 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product (including
MEDICAL POLICY SUBJECT: LIGHT AND LASER THERAPIES FOR PAGE: 1 OF: 9 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product (including
Yescarta (axicabtagene ciloleucel)
 Last Review Date: March 30, 2018 Effective Date: April 1, 2018 Number: MG.MM.PH.42 Medical Guideline Disclaimer Property of EmblemHealth. All rights reserved. The treating physician or primary care provider
Last Review Date: March 30, 2018 Effective Date: April 1, 2018 Number: MG.MM.PH.42 Medical Guideline Disclaimer Property of EmblemHealth. All rights reserved. The treating physician or primary care provider
Comparison of the narrow band UVB versus systemic corticosteroids in the treatment of lichen planus: A randomized clinical trial
 Received: 10.7.2011 Accepted: 5.12.2011 Original Article Comparison of the narrow band UVB versus systemic corticosteroids in the treatment of lichen planus: A randomized clinical trial Fariba Iraji, 1
Received: 10.7.2011 Accepted: 5.12.2011 Original Article Comparison of the narrow band UVB versus systemic corticosteroids in the treatment of lichen planus: A randomized clinical trial Fariba Iraji, 1
A Retrospective Study on the Risk of Non-Melanoma Skin Cancer in PUVA and Narrowband UVB Treated Patients
 Volume 1, Issue 3 Research Article A Retrospective Study on the Risk of Non-Melanoma Skin Cancer in PUVA and Narrowband UVB Treated Patients Darukarnphut P, Rattanakaemakorn P *, Rajatanavin N Division
Volume 1, Issue 3 Research Article A Retrospective Study on the Risk of Non-Melanoma Skin Cancer in PUVA and Narrowband UVB Treated Patients Darukarnphut P, Rattanakaemakorn P *, Rajatanavin N Division
Light Therapy for Psoriasis and Eczema
 Light Therapy for Psoriasis and Eczema Policy Number: 2.01.47 Last Review: 5/2018 Origination: 5/2006 Next Review: 5/2019 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will provide coverage
Light Therapy for Psoriasis and Eczema Policy Number: 2.01.47 Last Review: 5/2018 Origination: 5/2006 Next Review: 5/2019 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will provide coverage
Home Pulse Oximetry for Infants and Children
 Last Review Date: April 21, 2017 Number: MG.MM.DM.12aC2v2 Medical Guideline Disclaimer Property of EmblemHealth. All rights reserved. The treating physician or primary care provider must submit to EmblemHealth
Last Review Date: April 21, 2017 Number: MG.MM.DM.12aC2v2 Medical Guideline Disclaimer Property of EmblemHealth. All rights reserved. The treating physician or primary care provider must submit to EmblemHealth
Topical Oxygen Wound Therapy (MEDICAID)
 Topical Oxygen Wound Therapy (MEDICAID) Last Review Date: September 8, 2017 Number: MG.MM.DM.15C8v2 Medical Guideline Disclaimer Property of EmblemHealth. All rights reserved. The treating physician or
Topical Oxygen Wound Therapy (MEDICAID) Last Review Date: September 8, 2017 Number: MG.MM.DM.15C8v2 Medical Guideline Disclaimer Property of EmblemHealth. All rights reserved. The treating physician or
Clinical Policy Title: Phototherapy and photochemotherapy (PUVA) for skin conditions
 Clinical Policy Title: Phototherapy and photochemotherapy (PUVA) for skin conditions Clinical Policy Number: 16.02.04 Effective Date: October 1, 2015 Initial Review Date: May 20, 2015 Most Recent Review
Clinical Policy Title: Phototherapy and photochemotherapy (PUVA) for skin conditions Clinical Policy Number: 16.02.04 Effective Date: October 1, 2015 Initial Review Date: May 20, 2015 Most Recent Review
Baxdela (delafloxacin) for Injection
 Last Review Date: July 25th, 2018 Number: MG.MM.PH.53 Medical Guideline Disclaimer Property of EmblemHealth. All rights reserved. The treating physician or primary care provider must submit to EmblemHealth
Last Review Date: July 25th, 2018 Number: MG.MM.PH.53 Medical Guideline Disclaimer Property of EmblemHealth. All rights reserved. The treating physician or primary care provider must submit to EmblemHealth
Automatic External Defibrillators
 Last Review Date: April 21, 2017 Number: MG.MM.DM.10dC3v4 Medical Guideline Disclaimer Property of EmblemHealth. All rights reserved. The treating physician or primary care provider must submit to EmblemHealth
Last Review Date: April 21, 2017 Number: MG.MM.DM.10dC3v4 Medical Guideline Disclaimer Property of EmblemHealth. All rights reserved. The treating physician or primary care provider must submit to EmblemHealth
Intrathecal Baclofen for CNS Spasticity
 Intrathecal Baclofen for CNS Spasticity Last Review Date: October 13, 2017 Number: MG.MM.ME.31bC7 Medical Guideline Disclaimer Property of EmblemHealth. All rights reserved. The treating physician or primary
Intrathecal Baclofen for CNS Spasticity Last Review Date: October 13, 2017 Number: MG.MM.ME.31bC7 Medical Guideline Disclaimer Property of EmblemHealth. All rights reserved. The treating physician or primary
Clinical Policy Title: Phototherapy and photochemotherapy (PUVA) for skin conditions
 Clinical Policy Title: Phototherapy and photochemotherapy (PUVA) for skin conditions Clinical Policy Number: 16.02.04 Effective Date: October 1, 2015 Initial Review Date: May 20, 2015 Most Recent Review
Clinical Policy Title: Phototherapy and photochemotherapy (PUVA) for skin conditions Clinical Policy Number: 16.02.04 Effective Date: October 1, 2015 Initial Review Date: May 20, 2015 Most Recent Review
Clinical Policy: Laser Therapy for Skin Conditions Reference Number: CP.MP.123 Last Review Date: 08/17
 Clinical Policy: Laser Therapy for Skin Conditions Reference Number: CP.MP.123 Last Review Date: 08/17 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory
Clinical Policy: Laser Therapy for Skin Conditions Reference Number: CP.MP.123 Last Review Date: 08/17 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory
Original article Comparative study of psoralen-uvb vs. UVB-alone therapy in the treatment of psoriasis
 Original article Comparative study of psoralen-uvb vs. UVB-alone therapy in the treatment of psoriasis Syed Shamsuddin, *Tahir Saeed Haroon Department of Dermatology, Bolan Medical Complex, Quetta * Department
Original article Comparative study of psoralen-uvb vs. UVB-alone therapy in the treatment of psoriasis Syed Shamsuddin, *Tahir Saeed Haroon Department of Dermatology, Bolan Medical Complex, Quetta * Department
Outpatient Cardiac Rehabilitation
 Last Review Date: May 12, 2017 Number: MG.MM.ME.26bC3v2 Medical Guideline Disclaimer Property of EmblemHealth. All rights reserved. The treating physician or primary care provider must submit to EmblemHealth
Last Review Date: May 12, 2017 Number: MG.MM.ME.26bC3v2 Medical Guideline Disclaimer Property of EmblemHealth. All rights reserved. The treating physician or primary care provider must submit to EmblemHealth
Corporate Medical Policy
 Corporate Medical Policy Light Therapy for Dermatologic Conditions File Name: Origination: Last CAP Review: Next CAP Review: Last Review: light_therapy_for_dermatologic_conditions 5/2012 11/2017 11/2018
Corporate Medical Policy Light Therapy for Dermatologic Conditions File Name: Origination: Last CAP Review: Next CAP Review: Last Review: light_therapy_for_dermatologic_conditions 5/2012 11/2017 11/2018
The utilization of phototherapy in the department of dermatology, Hospital Kuala Lumpur: A 5-year audit
 ORIGINAL ARTICLE The utilization of phototherapy in the department of dermatology, Hospital Kuala Lumpur: A 5-year audit Vaani Valerie Visuvanathan, AdvMDerm 1, Min Moon Tang, AdvMDerm 2, Li Lian Tan,
ORIGINAL ARTICLE The utilization of phototherapy in the department of dermatology, Hospital Kuala Lumpur: A 5-year audit Vaani Valerie Visuvanathan, AdvMDerm 1, Min Moon Tang, AdvMDerm 2, Li Lian Tan,
Clinical Policy Title: Phototherapy and photochemotherapy for skin conditions
 Clinical Policy Title: Phototherapy and photochemotherapy for skin conditions Clinical Policy Number: 16.02.04 Effective Date: October 1, 2015 Initial Review Date: May 20, 2015 Most Recent Review Date:
Clinical Policy Title: Phototherapy and photochemotherapy for skin conditions Clinical Policy Number: 16.02.04 Effective Date: October 1, 2015 Initial Review Date: May 20, 2015 Most Recent Review Date:
POLICY PRODUCT VARIATIONS DESCRIPTION/BACKGROUND RATIONALE DEFINITIONS BENEFIT VARIATIONS DISCLAIMER CODING INFORMATION REFERENCES POLICY HISTORY
 Original Issue Date (Created): July 1, 2002 Most Recent Review Date (Revised): March 25, 2014 Effective Date: June 1, 2014 POLICY PRODUCT VARIATIONS DESCRIPTION/BACKGROUND RATIONALE DEFINITIONS BENEFIT
Original Issue Date (Created): July 1, 2002 Most Recent Review Date (Revised): March 25, 2014 Effective Date: June 1, 2014 POLICY PRODUCT VARIATIONS DESCRIPTION/BACKGROUND RATIONALE DEFINITIONS BENEFIT
Original Policy Date
 MP 2.01.07 Psoralens with Ultraviolet A (PUVA) Medical Policy Section Medicine Issue 12:2013 Original Policy Date 12:2013 Last Review Status/Date Reviewed by consensus/12:2013 Return to Medical Policy
MP 2.01.07 Psoralens with Ultraviolet A (PUVA) Medical Policy Section Medicine Issue 12:2013 Original Policy Date 12:2013 Last Review Status/Date Reviewed by consensus/12:2013 Return to Medical Policy
Table of Contents: Part 1 Medical Dermatology. Chapter 1 Acneiform Disorders. Acne. Acne Vulgaris. Pomade Acne. Steroid Acne
 Table of Contents: Part 1 Medical Dermatology Chapter 1 Acneiform Disorders Acne Acne Vulgaris Pomade Acne Steroid Acne Infantile Acne Pediatric Perspectives Neonatal Acne (Acne Neonatorum) Pediatric Perspectives
Table of Contents: Part 1 Medical Dermatology Chapter 1 Acneiform Disorders Acne Acne Vulgaris Pomade Acne Steroid Acne Infantile Acne Pediatric Perspectives Neonatal Acne (Acne Neonatorum) Pediatric Perspectives
Narrow-band UVB PHOTOTHERAPY for Skin Diseases
 Narrow-band UVB PHOTOTHERAPY for Skin Diseases By Dr. Manal Bosseila Cairo University, Egypt HISTORICAL ASPECT In 1978: Irradiation cabin with broad band UVB tubes was introduced for psoriasis & uremic
Narrow-band UVB PHOTOTHERAPY for Skin Diseases By Dr. Manal Bosseila Cairo University, Egypt HISTORICAL ASPECT In 1978: Irradiation cabin with broad band UVB tubes was introduced for psoriasis & uremic
Name of Policy: Phototherapy for the Treatment of Skin Disorders
 Name of Policy: Phototherapy for the Treatment of Skin Disorders Policy #: 301 Latest Review Date: April 2014 Category: Medical/DME Policy Grade: B Background: As a general rule, benefits are payable under
Name of Policy: Phototherapy for the Treatment of Skin Disorders Policy #: 301 Latest Review Date: April 2014 Category: Medical/DME Policy Grade: B Background: As a general rule, benefits are payable under
In former years, patients were treated with broad-band
 ORIGINAL ARTICLE Efficacy of UVA1 phototherapy in 23 patients with various skin diseases S. Rombold, K. Lobisch, K. Katzer, T. C. Grazziotin, J. Ring & B. Eberlein Department of Dermatology and Allergy
ORIGINAL ARTICLE Efficacy of UVA1 phototherapy in 23 patients with various skin diseases S. Rombold, K. Lobisch, K. Katzer, T. C. Grazziotin, J. Ring & B. Eberlein Department of Dermatology and Allergy
Chelation Therapy Last Review Date: February 10, 2017 Number: MG.MM.ME.48C3 Medical Guideline Disclaimer Definition Guideline Limitations/Exclusions
 Last Review Date: February 10, 2017 Number: MG.MM.ME.48C3 Medical Guideline Disclaimer Property of EmblemHealth. All rights reserved. The treating physician or primary care provider must submit to EmblemHealth
Last Review Date: February 10, 2017 Number: MG.MM.ME.48C3 Medical Guideline Disclaimer Property of EmblemHealth. All rights reserved. The treating physician or primary care provider must submit to EmblemHealth
Photodynamic Therapy for the Treatment of Actinic Keratoses and Other Skin Lesions
 Photodynamic Therapy for the Treatment of Actinic Keratoses and Other Skin Lesions Policy Number: Original Effective Date: MM.02.016 04/01/2008 Line(s) of Business: Current Effective Date: HMO; PPO; QUEST
Photodynamic Therapy for the Treatment of Actinic Keratoses and Other Skin Lesions Policy Number: Original Effective Date: MM.02.016 04/01/2008 Line(s) of Business: Current Effective Date: HMO; PPO; QUEST
Clinical Policy Title: Vitiligo dermatology treatment
 Clinical Policy Title: Vitiligo dermatology treatment Clinical Policy Number: 16.02.08 Effective Date: June 1, 2017 Initial Review Date: April 19, 2017 Most Recent Review Date: April 10, 2018 Next Review
Clinical Policy Title: Vitiligo dermatology treatment Clinical Policy Number: 16.02.08 Effective Date: June 1, 2017 Initial Review Date: April 19, 2017 Most Recent Review Date: April 10, 2018 Next Review
Clinical Policy Title: Psoriasis dermatology treatment
 Clinical Policy Title: Psoriasis dermatology treatment Clinical Policy Number: 16.02.06 Effective Date: October 1, 2016 Initial Review Date: September 17, 2016 Most Recent Review Date: September 21, 2017
Clinical Policy Title: Psoriasis dermatology treatment Clinical Policy Number: 16.02.06 Effective Date: October 1, 2016 Initial Review Date: September 17, 2016 Most Recent Review Date: September 21, 2017
Summary. DOI /j x
 PHOTOBIOLOGY DOI 10.1111/j.1365-2133.2005.06533.x Comparison of the 308-nm excimer laser and a 308-nm excimer lamp with 311-nm narrowband ultraviolet B in the treatment of psoriasis K. Köllner, M.B. Wimmershoff,
PHOTOBIOLOGY DOI 10.1111/j.1365-2133.2005.06533.x Comparison of the 308-nm excimer laser and a 308-nm excimer lamp with 311-nm narrowband ultraviolet B in the treatment of psoriasis K. Köllner, M.B. Wimmershoff,
Therapeutic Shoes for Diabetics
 Last Review Date: August 11, 2017 Number: MG.MM.DM.03bC8v2 Medical Guideline Disclaimer Property of EmblemHealth. All rights reserved. The treating physician or primary care provider must submit to EmblemHealth
Last Review Date: August 11, 2017 Number: MG.MM.DM.03bC8v2 Medical Guideline Disclaimer Property of EmblemHealth. All rights reserved. The treating physician or primary care provider must submit to EmblemHealth
They are updated regularly as new NICE guidance is published. To view the latest version of this NICE Pathway see:
 bring together everything NICE says on a topic in an interactive flowchart. are interactive and designed to be used online. They are updated regularly as new NICE guidance is published. To view the latest
bring together everything NICE says on a topic in an interactive flowchart. are interactive and designed to be used online. They are updated regularly as new NICE guidance is published. To view the latest
Topical Immunomodulator Step Therapy Program
 Topical Immunomodulator Step Therapy Program Policy Number: 5.01.557 Last Review: 8/2017 Origination: 7/2013 Next Review: 8/2018 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) BCBSKC will provide
Topical Immunomodulator Step Therapy Program Policy Number: 5.01.557 Last Review: 8/2017 Origination: 7/2013 Next Review: 8/2018 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) BCBSKC will provide
Appendix D: Authorization Guidelines for Dermatology Services
 Appendix D: Authorization Guidelines for Dermatology Services Revised June 2011 1 Appendix D: Authorization Guidelines for Dermatology Dermatologists are limited to the CPT codes referenced in this Section.
Appendix D: Authorization Guidelines for Dermatology Services Revised June 2011 1 Appendix D: Authorization Guidelines for Dermatology Dermatologists are limited to the CPT codes referenced in this Section.
Efficacy of Concomitant Use of PUVA and Methotrexate in Disease Clearance Time in Plaque Type Psoriasis
 Efficacy of Concomitant Use of PUVA and Methotrexate in Disease Clearance Time in Plaque Type Psoriasis T. Shehzad ( Departments of Dermatology Naval Hospital PNS Shifa, Karachi. ) N. R. Dar ( Departments
Efficacy of Concomitant Use of PUVA and Methotrexate in Disease Clearance Time in Plaque Type Psoriasis T. Shehzad ( Departments of Dermatology Naval Hospital PNS Shifa, Karachi. ) N. R. Dar ( Departments
Narrow band UVB (311 nm), psoralen UVB (311 nm) and PUVA therapy in the treatment of early-stage mycosis fungoides: a right left comparative study
 Photodermatol Photoimmunol Photomed 2005; 21: 281 286 Blackwell Munksgaard Copyright r Blackwell Munksgaard 2005 Narrow band UVB (311 nm), psoralen UVB (311 nm) and therapy in the treatment of early-stage
Photodermatol Photoimmunol Photomed 2005; 21: 281 286 Blackwell Munksgaard Copyright r Blackwell Munksgaard 2005 Narrow band UVB (311 nm), psoralen UVB (311 nm) and therapy in the treatment of early-stage
SCIENTIFIC PAPER ABSTRACT
 SCIENTIFIC PAPER ABSTRACT Vitiligo Treatment with Monochromatic Excimer Light and Tacrolimus: Results of an Open Randomized Controlled Study Nistico` S., Chiricozzi A., M.D., Rosita Saraceno R., Schipani
SCIENTIFIC PAPER ABSTRACT Vitiligo Treatment with Monochromatic Excimer Light and Tacrolimus: Results of an Open Randomized Controlled Study Nistico` S., Chiricozzi A., M.D., Rosita Saraceno R., Schipani
Light Therapy for Psoriasis Protocol Medical Benefit Effective Date Next Review Date Preauthorization Review Dates Preauthorization is required.
 Protocol Light Therapy for Psoriasis (20147) Medical Benefit Effective Date: 07/01/16 Next Review Date: 03/18 Preauthorization Yes Review Dates: 03/16, 03/17 Preauthorization is required. The following
Protocol Light Therapy for Psoriasis (20147) Medical Benefit Effective Date: 07/01/16 Next Review Date: 03/18 Preauthorization Yes Review Dates: 03/16, 03/17 Preauthorization is required. The following
Name of Policy: Phototherapy for the Treatment of Skin Disorders
 Name of Policy: Phototherapy for the Treatment of Skin Disorders Policy #: 301 Latest Review Date: December 2017 Category: Medical/DME Policy Grade: B Background: As a general rule, benefits are payable
Name of Policy: Phototherapy for the Treatment of Skin Disorders Policy #: 301 Latest Review Date: December 2017 Category: Medical/DME Policy Grade: B Background: As a general rule, benefits are payable
Blepharoplasty. Definitions
 Last Review Date: June 9, 2017 Number: MG.MM.SU.10eC5 Medical Guideline Disclaimer Property of EmblemHealth. All rights reserved. The treating physician or primary care provider must submit to EmblemHealth
Last Review Date: June 9, 2017 Number: MG.MM.SU.10eC5 Medical Guideline Disclaimer Property of EmblemHealth. All rights reserved. The treating physician or primary care provider must submit to EmblemHealth
DERMATOLOGIC APPLICATIONS OF PHOTODYNAMIC THERAPY
 DERMATOLOGIC APPLICATIONS OF PHOTODYNAMIC THERAPY Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services, procedures,
DERMATOLOGIC APPLICATIONS OF PHOTODYNAMIC THERAPY Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services, procedures,
Topical Psoralen and Sunlight in Treatment of Palmoplantar Psoriasis in Iraqi Patients
 Topical Psoralen and Sunlight in Treatment of Palmoplantar Psoriasis (M.B.CH.B FIBMS) Abstract Back ground: Topical psoralens plus sunlight is a common practice among Dermatologists in the world for treatment
Topical Psoralen and Sunlight in Treatment of Palmoplantar Psoriasis (M.B.CH.B FIBMS) Abstract Back ground: Topical psoralens plus sunlight is a common practice among Dermatologists in the world for treatment
Medical Policy An independent licensee of the Blue Cross Blue Shield Association
 Ultraviolet Light Therapy for Skin Conditions Page 1 of 17 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Ultraviolet Light Therapy for Skin Conditions Professional
Ultraviolet Light Therapy for Skin Conditions Page 1 of 17 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Ultraviolet Light Therapy for Skin Conditions Professional
Laser, Light Therapy, and Cryotherapy for Acne Vulgaris Non-Pharmacologic Treatment of Rosacea
 2.01.47 Light Therapy for Psoriasis Section 2.0 Medicine Subsection Effective Date October 31, 2014 Original Policy Date June 13, 2001 Next Review Date October 2015 Description Plaque psoriasis, also called
2.01.47 Light Therapy for Psoriasis Section 2.0 Medicine Subsection Effective Date October 31, 2014 Original Policy Date June 13, 2001 Next Review Date October 2015 Description Plaque psoriasis, also called
Vitiligo is an acquired cutaneous disorder of
 Narrow-band ultraviolet B is a useful and well-tolerated treatment for vitiligo Lubomira Scherschun, MD, Jane J. Kim, MD, and Henry W. Lim, MD Detroit, Michigan Background: The treatment of vitiligo remains
Narrow-band ultraviolet B is a useful and well-tolerated treatment for vitiligo Lubomira Scherschun, MD, Jane J. Kim, MD, and Henry W. Lim, MD Detroit, Michigan Background: The treatment of vitiligo remains
Original Policy Date
 MP 2.01.58 Light Therapy for Vitiligo Medical Policy Section Medicine Issue 12:2013 Original Policy Date 12:2013 Last Review Status/Date Created with literature search/12:2013 Return to Medical Policy
MP 2.01.58 Light Therapy for Vitiligo Medical Policy Section Medicine Issue 12:2013 Original Policy Date 12:2013 Last Review Status/Date Created with literature search/12:2013 Return to Medical Policy
Corporate Medical Policy
 Corporate Medical Policy Dermatologic Applications of Photodynamic Therapy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: dermatologic_applications_of_photodynamic_therapy 10/2003
Corporate Medical Policy Dermatologic Applications of Photodynamic Therapy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: dermatologic_applications_of_photodynamic_therapy 10/2003
Medical Policy An independent licensee of the Blue Cross Blue Shield Association
 Topical Doxepin Page 1 of 6 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Topical Doxepin (For Individuals Who Purchased BlueCare/KS Solutions/EPO Products) Prime
Topical Doxepin Page 1 of 6 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Topical Doxepin (For Individuals Who Purchased BlueCare/KS Solutions/EPO Products) Prime
A systematic review of treatments for severe psoriasis Griffiths C E, Clark C M, Chalmers R J, Li Wan Po A, Williams H C
 A systematic review of treatments for severe psoriasis Griffiths C E, Clark C M, Chalmers R J, Li Wan Po A, Williams H C Authors' objectives To compare the effectiveness of currently available treatments
A systematic review of treatments for severe psoriasis Griffiths C E, Clark C M, Chalmers R J, Li Wan Po A, Williams H C Authors' objectives To compare the effectiveness of currently available treatments
Phototherapy For Dermatologic Conditions
 Phototherapy For Dermatologic Conditions Policy Number: 2016M0050B Effective Date: November 01, 2016 Table of Contents: Page: Cross Reference Policy: POLICY DESCRIPTION 2 Port Wine Stains Treatment, 2016M0042A
Phototherapy For Dermatologic Conditions Policy Number: 2016M0050B Effective Date: November 01, 2016 Table of Contents: Page: Cross Reference Policy: POLICY DESCRIPTION 2 Port Wine Stains Treatment, 2016M0042A
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Cosentyx, Cosentyx Sensoready) Reference Number: HIM.PA.SP29 Effective Date: 05/17 Last Review Date: Line of Business: Health Insurance Marketplace Coding Implications Revision Log See
Clinical Policy: (Cosentyx, Cosentyx Sensoready) Reference Number: HIM.PA.SP29 Effective Date: 05/17 Last Review Date: Line of Business: Health Insurance Marketplace Coding Implications Revision Log See
1. Skin care: Cleansing, lubrication, debriding and administration of antimicrobial therapy.
 Lymphedema Treatment Last Review Date: October 13, 2017 Number: MG.MM.ME.05aC9 Medical Guideline Disclaimer Property of EmblemHealth. All rights reserved. The treating physician or primary care provider
Lymphedema Treatment Last Review Date: October 13, 2017 Number: MG.MM.ME.05aC9 Medical Guideline Disclaimer Property of EmblemHealth. All rights reserved. The treating physician or primary care provider
Policy. Number: *Pleasesee amendment forpennsylvaniamedicaidattheend ofthis CPB.
 1 of 49 Number: 0205 Policy *Pleasesee amendment forpennsylvaniamedicaidattheend ofthis CPB. PUVA Aetna considers psoralens and ultraviolet A light (PUVA) treatments medically necessary for the following
1 of 49 Number: 0205 Policy *Pleasesee amendment forpennsylvaniamedicaidattheend ofthis CPB. PUVA Aetna considers psoralens and ultraviolet A light (PUVA) treatments medically necessary for the following
Clinical Policy: Benign Skin Lesion Removal Reference Number: CP.MP.HN150
 Clinical Policy: Reference Number: CP.MP.HN150 Effective Date: 6/04 Last Review Date: 8/17 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory and
Clinical Policy: Reference Number: CP.MP.HN150 Effective Date: 6/04 Last Review Date: 8/17 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory and
Index. derm.theclinics.com. Note: Page numbers of article titles are in boldface type.
 Note: Page numbers of article titles are in boldface type. A Abatacept for DLE, 493 for SLE, 497 Ablative therapies, localized, for cutaneous T-cell lymphoma, 502 506. See also Cutaneous T-cell lymphoma,
Note: Page numbers of article titles are in boldface type. A Abatacept for DLE, 493 for SLE, 497 Ablative therapies, localized, for cutaneous T-cell lymphoma, 502 506. See also Cutaneous T-cell lymphoma,
Cigna Drug and Biologic Coverage Policy
 Cigna Drug and Biologic Coverage Policy Subject Apremilast Table of Contents Coverage Policy... 1 General Background... 2 Coding/Billing Information... 4 References... 4 Effective Date... 1/1/2018 Next
Cigna Drug and Biologic Coverage Policy Subject Apremilast Table of Contents Coverage Policy... 1 General Background... 2 Coding/Billing Information... 4 References... 4 Effective Date... 1/1/2018 Next
Phototherapy, Photochemotherapy, and Excimer Laser Therapy for Dermatologic Conditions
 Medical Coverage Policy Effective Date... 5/15/2018 Next Review Date... 5/15/2019 Coverage Policy Number... 0031 Phototherapy, Photochemotherapy, and Excimer Laser Therapy for Dermatologic Conditions Table
Medical Coverage Policy Effective Date... 5/15/2018 Next Review Date... 5/15/2019 Coverage Policy Number... 0031 Phototherapy, Photochemotherapy, and Excimer Laser Therapy for Dermatologic Conditions Table
Medical Policy An independent licensee of the Blue Cross Blue Shield Association
 Topical Doxepin Page 1 of 5 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Topical Doxepin Prime Therapeutics will review Prior Authorization requests Prior Authorization
Topical Doxepin Page 1 of 5 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Topical Doxepin Prime Therapeutics will review Prior Authorization requests Prior Authorization
Rash Decisions Approach to the patient with a skin condition
 National Conference for Nurse Practitioners April 25, 2014 Rash Decisions Approach to the patient with a skin condition Margaret A. Bobonich, DNP, FNP C, DCNP, FAANP Assistant Professor, Case Western Reserve
National Conference for Nurse Practitioners April 25, 2014 Rash Decisions Approach to the patient with a skin condition Margaret A. Bobonich, DNP, FNP C, DCNP, FAANP Assistant Professor, Case Western Reserve
BRAF Mutation Analysis
 Last Review Date: October 13, 2017 Number: MG.MM.LA.38aC Medical Guideline Disclaimer Property of EmblemHealth. All rights reserved. The treating physician or primary care provider must submit to EmblemHealth
Last Review Date: October 13, 2017 Number: MG.MM.LA.38aC Medical Guideline Disclaimer Property of EmblemHealth. All rights reserved. The treating physician or primary care provider must submit to EmblemHealth
Comparison of PUVA and UVB therapy in moderate plaque psoriasis. Arfan ul Bari, Nadia Iftikhar*, Simeen ber Rahman*
 Comparison of PUVA and UVB therapy in moderate plaque psoriasis Arfan ul Bari et al. Arfan ul Bari, Nadia Iftikhar*, Simeen ber Rahman* Department of Dermatology, PAF Hospital, Sargodha. * Department of
Comparison of PUVA and UVB therapy in moderate plaque psoriasis Arfan ul Bari et al. Arfan ul Bari, Nadia Iftikhar*, Simeen ber Rahman* Department of Dermatology, PAF Hospital, Sargodha. * Department of
It is estimated that about 26,000 new cases of
 Focus on CME at Dalhousie University Set On Soothing Psoriasis A. H. Murray, MD, FRCP(C) Presented at the 76th Annual Dalhousie Refresher Course It is estimated that about 26,000 new cases of psoriasis
Focus on CME at Dalhousie University Set On Soothing Psoriasis A. H. Murray, MD, FRCP(C) Presented at the 76th Annual Dalhousie Refresher Course It is estimated that about 26,000 new cases of psoriasis
Index. Angiosarcoma diagnosis, 47 lymphedema-related vs. non-lymphedemarelated, 48
 A Acneiform rash biopsy, 134 cetuximab, EGFR, 132 133 diagnosis, 131 patient history, 131 134 treatment, 134 135 Acne vulgaris, 109 AGA. See Androgenetic alopecia Alopecia areata, 148 American Joint Committee
A Acneiform rash biopsy, 134 cetuximab, EGFR, 132 133 diagnosis, 131 patient history, 131 134 treatment, 134 135 Acne vulgaris, 109 AGA. See Androgenetic alopecia Alopecia areata, 148 American Joint Committee
An otherwise healthy 12-year-old
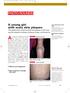 1105 Photo Rounds.finalREV 10/19/05 2:05 PM Page 947 A young girl with scaly skin plaques The patient had numerous thick red plaques on her back and the extensor surfaces of elbows, knees, and forearms
1105 Photo Rounds.finalREV 10/19/05 2:05 PM Page 947 A young girl with scaly skin plaques The patient had numerous thick red plaques on her back and the extensor surfaces of elbows, knees, and forearms
See Important Reminder at the end of this policy for important regulatory and legal information.
 Policy: Atopic Dermatitis and Topical Antipsoriatics Reference Number: TCHP.PHAR.18004 Effective Date: 01.01.18 Last Review Date: 10.12.18 Line of Business: Oregon Health Plan Revision Log See Important
Policy: Atopic Dermatitis and Topical Antipsoriatics Reference Number: TCHP.PHAR.18004 Effective Date: 01.01.18 Last Review Date: 10.12.18 Line of Business: Oregon Health Plan Revision Log See Important
4. Pityriasis lichenoides
 Go Back to the Top To Order, Visit the Purchasing Page for Details usually more than 5 cm in diameter and accompanied by poikiloderma. Some but not all patients may develop mycosis fungoides (Fig. 22.35).
Go Back to the Top To Order, Visit the Purchasing Page for Details usually more than 5 cm in diameter and accompanied by poikiloderma. Some but not all patients may develop mycosis fungoides (Fig. 22.35).
Combination Topical PUVAsol with Methotrexate Versus Methotrexate in the Treatment of Palmoplantar Psoriasis Karn D, KC S
 Combination Topical PUVAsol with Methotrexate Versus Methotrexate in the Treatment of Palmoplantar Psoriasis Karn D, KC S ABSTRACT Background Department of Dermatology Dhulikhel Hospital, Kathmandu University
Combination Topical PUVAsol with Methotrexate Versus Methotrexate in the Treatment of Palmoplantar Psoriasis Karn D, KC S ABSTRACT Background Department of Dermatology Dhulikhel Hospital, Kathmandu University
UVB phototherapy and skin cancer risk: a review of the literature
 Oxford, IJD International 0011-9059 Blackwell 45 UK Publishing Journal Ltd. Ltd, of Dermatology 2003 Review Lee, Koo, phototherapy and Berger and skin cancer risk UVB phototherapy and skin cancer risk:
Oxford, IJD International 0011-9059 Blackwell 45 UK Publishing Journal Ltd. Ltd, of Dermatology 2003 Review Lee, Koo, phototherapy and Berger and skin cancer risk UVB phototherapy and skin cancer risk:
Follow this and additional works at: Part of the Skin and Connective Tissue Diseases Commons
 Philadelphia College of Osteopathic Medicine DigitalCommons@PCOM PCOM Physician Assistant Studies Student Scholarship Student Dissertations, Theses and Papers 2016 Is Narrowband UVB Phototherapy in Combination
Philadelphia College of Osteopathic Medicine DigitalCommons@PCOM PCOM Physician Assistant Studies Student Scholarship Student Dissertations, Theses and Papers 2016 Is Narrowband UVB Phototherapy in Combination
Atopic dermatitis (AD) is a common chronic skin. Phototherapy in the management of atopic dermatitis: a systematic review.
 Photodermatol Photoimmunol Photomed 2007; 23: 106 112 Blackwell Munksgaard r 2007 The Authors Journal compilation r 2007 Blackwell Munksgaard Review article Phototherapy in the management of atopic dermatitis:
Photodermatol Photoimmunol Photomed 2007; 23: 106 112 Blackwell Munksgaard r 2007 The Authors Journal compilation r 2007 Blackwell Munksgaard Review article Phototherapy in the management of atopic dermatitis:
Site and distribution: symmetrical, asymmetrical. Surface characteristics: smooth, scaly, warty
 B I O T E R R O R I S M PRINCIPLES AND PRACTICE OF DERMATOLOGY Dr Matthew Ng Joo Ming INTRODUCTION Medical schools and textbooks teach us dermatology by subjects such as eczema and psoriasis. This is useful
B I O T E R R O R I S M PRINCIPLES AND PRACTICE OF DERMATOLOGY Dr Matthew Ng Joo Ming INTRODUCTION Medical schools and textbooks teach us dermatology by subjects such as eczema and psoriasis. This is useful
Medical Policy. MP Light Therapy for Psoriasis
 Medical Policy MP 2.01.47 BCBSA Ref. Policy: 2.01.47 Last Review: 12/27/2017 Effective Date: 12/27/2017 Section: Medicine Related Policies 2.01.44 Dermatologic Applications of Photodynamic Therapy 2.01.86
Medical Policy MP 2.01.47 BCBSA Ref. Policy: 2.01.47 Last Review: 12/27/2017 Effective Date: 12/27/2017 Section: Medicine Related Policies 2.01.44 Dermatologic Applications of Photodynamic Therapy 2.01.86
Clinical Policy: Ustekinumab (Stelara) Reference Number: CP.PHAR.264
 Clinical Policy: (Stelara) Reference Number: CP.PHAR.264 Effective Date: 08/16 Last Review Date: 05/16 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory
Clinical Policy: (Stelara) Reference Number: CP.PHAR.264 Effective Date: 08/16 Last Review Date: 05/16 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory
CONDITIONS OF THE SKIN
 CONDITIONS OF THE SKIN UCSF/SFGH Family & Community Medicine Residency Program Educational Objectives I. Knowledge The resident will be able to discuss the definition, diagnosis, and initial management
CONDITIONS OF THE SKIN UCSF/SFGH Family & Community Medicine Residency Program Educational Objectives I. Knowledge The resident will be able to discuss the definition, diagnosis, and initial management
Clinical Policy: Apremilast (Otezla) Reference Number: CP.PHAR.245 Effective Date: 08/16 Last Review Date 08/17
 Clinical Policy: (Otezla) Reference Number: CP.PHAR.245 Effective Date: 08/16 Last Review Date 08/17 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
Clinical Policy: (Otezla) Reference Number: CP.PHAR.245 Effective Date: 08/16 Last Review Date 08/17 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
Comparison of the efficacy of PUVA versus BBUVB in the treatment of psoriasis vulgaris
 IJMAMR 5 (2017) 1-6 ISSN 2053-1834 Comparison of the efficacy of PUVA versus BBUVB in the treatment of psoriasis vulgaris Tran Hau Khang* and Le Huu Doanh National Hospital of Dermatology and Venereology,
IJMAMR 5 (2017) 1-6 ISSN 2053-1834 Comparison of the efficacy of PUVA versus BBUVB in the treatment of psoriasis vulgaris Tran Hau Khang* and Le Huu Doanh National Hospital of Dermatology and Venereology,
Clinical Policy Title: Vitiligo dermatology treatment
 Clinical Policy Title: Vitiligo dermatology treatment Clinical Policy Number: 16.02.08 Effective Date: June 1, 2017 Initial Review Date: April 19, 2017 Most Recent Review Date: May 19, 2017 Next Review
Clinical Policy Title: Vitiligo dermatology treatment Clinical Policy Number: 16.02.08 Effective Date: June 1, 2017 Initial Review Date: April 19, 2017 Most Recent Review Date: May 19, 2017 Next Review
Review Article. Narrow band UVB phototherapy in dermatology
 Review Article Narrow band UVB phototherapy in dermatology Sunil Dogra, Amrinder Jit Kanwar Department of Dermatology, Venereology and Leprology, Postgraduate Institute of Medical Education & Research,
Review Article Narrow band UVB phototherapy in dermatology Sunil Dogra, Amrinder Jit Kanwar Department of Dermatology, Venereology and Leprology, Postgraduate Institute of Medical Education & Research,
Clinical Policy: Secukinumab (Cosentyx) Reference Number: CP.PHAR.261 Effective Date: 08/16 Last Review Date: 08/17
 Clinical Policy: (Cosentyx) Reference Number: CP.PHAR.261 Effective Date: 08/16 Last Review Date: 08/17 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
Clinical Policy: (Cosentyx) Reference Number: CP.PHAR.261 Effective Date: 08/16 Last Review Date: 08/17 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
Field vs Lesional Therapies for AKs 3/2/2019, 9:00-12 AM
 Dilemmas and Challenges in Skin Cancer Therapies and Management Field vs Lesional Therapies for AKs 3/2/2019, 9:00-12 AM Roger I. Ceilley, M.D. Clinical Professor of Dermatology The University of Iowa
Dilemmas and Challenges in Skin Cancer Therapies and Management Field vs Lesional Therapies for AKs 3/2/2019, 9:00-12 AM Roger I. Ceilley, M.D. Clinical Professor of Dermatology The University of Iowa
Acne Related Procedures ACNE RELATED PROCEDURES HS-258. Policy Number: HS-258. Original Effective Date: 9/4/2014. Revised Date(s): 6/8/2015
 Easy Choice Health Plan, Inc. Harmony Health Plan of Illinois, Inc. Missouri Care, Inc. Ohana Health Plan, a plan offered by WellCare Health Insurance of Arizona, Inc. WellCare Health Insurance of Illinois,
Easy Choice Health Plan, Inc. Harmony Health Plan of Illinois, Inc. Missouri Care, Inc. Ohana Health Plan, a plan offered by WellCare Health Insurance of Arizona, Inc. WellCare Health Insurance of Illinois,
Clinical Policy: Brodalumab (Siliq) Reference Number: CP.PHAR.375 Effective Date: Last Review Date: 05.18
 Clinical Policy: (Siliq) Reference Number: CP.PHAR.375 Effective Date: 06.01.18 Last Review Date: 05.18 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
Clinical Policy: (Siliq) Reference Number: CP.PHAR.375 Effective Date: 06.01.18 Last Review Date: 05.18 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
Title: Automatic External Defibrillators Division: Medical Management Department: Utilization Management
 Retired Date: Page 1 of 7 1. POLICY DESCRIPTION: Automatic External Defibrillators 2. RESPONSIBLE PARTIES: Medical Management Administration, Utilization Management, Integrated Care Management, Pharmacy,
Retired Date: Page 1 of 7 1. POLICY DESCRIPTION: Automatic External Defibrillators 2. RESPONSIBLE PARTIES: Medical Management Administration, Utilization Management, Integrated Care Management, Pharmacy,
Scottish Medicines Consortium
 Scottish Medicines Consortium imiquimod 5% cream (Aldara) No. (385/07) Meda Pharmaceuticals Ltd 04 April 2008 The Scottish Medicines Consortium has completed its assessment of the above product and advises
Scottish Medicines Consortium imiquimod 5% cream (Aldara) No. (385/07) Meda Pharmaceuticals Ltd 04 April 2008 The Scottish Medicines Consortium has completed its assessment of the above product and advises
Clinical Policy: Ixekizumab (Taltz) Reference Number: CP.PHAR.257 Effective Date: Last Review Date: 05.18
 Clinical Policy: (Taltz) Reference Number: CP.PHAR.257 Effective Date: 08.01.16 Last Review Date: 05.18 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
Clinical Policy: (Taltz) Reference Number: CP.PHAR.257 Effective Date: 08.01.16 Last Review Date: 05.18 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
Subspecialty Rotation: Dermatology
 Subspecialty Rotation: Dermatology Faculty: Wesley Galen, M.D. GOAL: Prevention, Counseling and Screening (Dermatology). Understand the pediatrician's role in preventing illness and dysfunction related
Subspecialty Rotation: Dermatology Faculty: Wesley Galen, M.D. GOAL: Prevention, Counseling and Screening (Dermatology). Understand the pediatrician's role in preventing illness and dysfunction related
Coding Wars: The Coding and Documentation Weapons to Win the Battle
 Coding Wars: The Coding and Documentation Weapons to Win the Battle Howard W. Rogers M.D., Ph.D. Advanced Dermatology Norwich, CT rogershoward@sbcglobal.net Do We Still Need to Learn Coding? Low Complexity
Coding Wars: The Coding and Documentation Weapons to Win the Battle Howard W. Rogers M.D., Ph.D. Advanced Dermatology Norwich, CT rogershoward@sbcglobal.net Do We Still Need to Learn Coding? Low Complexity
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE SCOPE
 NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE 1 Guideline title SCOPE Psoriasis: the management of psoriasis 1.1 Short title Psoriasis 2 The remit The Department of Health has asked NICE: 'to produce
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE 1 Guideline title SCOPE Psoriasis: the management of psoriasis 1.1 Short title Psoriasis 2 The remit The Department of Health has asked NICE: 'to produce
Series in Dermatological Treatment
 Series in Dermatological Treatment Series editors Steven R Feldman and Peter van de Kerkhof Published in association with the Journal of Dermatological Treatment 1. Robert Baran, Roderick Hay, Eckhart
Series in Dermatological Treatment Series editors Steven R Feldman and Peter van de Kerkhof Published in association with the Journal of Dermatological Treatment 1. Robert Baran, Roderick Hay, Eckhart
Project manager. Dr. Nicola Zerbinati. Therapeutic protocols of monochromatic source 355 nm λ
 Project manager Therapeutic protocols of monochromatic source 355 nm λ INTRODUCTION Artificial ultraviolet rays (UV) such as sunbeds, lamps, solar panels, are used both in the beauty and medical field,
Project manager Therapeutic protocols of monochromatic source 355 nm λ INTRODUCTION Artificial ultraviolet rays (UV) such as sunbeds, lamps, solar panels, are used both in the beauty and medical field,
