THE LARGE MAJORITY of women gain weight and fat
|
|
|
- Martha Lawrence
- 5 years ago
- Views:
Transcription
1 /08/$15.00/0 Endocrinology 149(5): Printed in U.S.A. Copyright 2008 by The Endocrine Society doi: /en Adverse Adipose Phenotype and Hyperinsulinemia in Gravid Mice Deficient in Placental Growth Factor Bianca Hemmeryckx, Rita van Bree, Berthe Van Hoef, Lisbeth Vercruysse, H. Roger Lijnen, and Johan Verhaeghe Department of Obstetrics and Gynecology (B.H., R.v.B., L.V., J.V.) and the Center for Molecular and Vascular Biology (B.V.H., H.R.L.), Health Campus Gasthuisberg, Katholieke Universiteit Leuven, 3000 Leuven, Belgium THE LARGE MAJORITY of women gain weight and fat mass during pregnancy. Seventy-six percent of the gestational fat is stored in sc depots and 68% in the trunk (1). Signals from an expanded adipose organ are believed to explain in part the physiological insulin resistance and hyperinsulinemia of pregnancy (2, 3). Adipose tissue (AT) expansion is the result of adipocyte hyperplasia and/or hypertrophy. Neoadipogenesis is preceded or accompanied by an angiogenic response (endothelial cell proliferation, vessel sprouting) (4 7). It is unknown whether pregnancy induces angiogenesis and adipogenesis. However, adipocyte hypertrophy has been documented in gravid rats (8, 9) and raised circulating leptin and TNF- suggest a comparable response in human pregnancy (2). Placental growth factor (PlGF) is an important angiogenic factor. PlGF shares homology with vascular endothelial growth factor (VEGF) and binds with high affinity First Published Online February 7, 2008 Abbreviations: Ang-2, Angiopoetin II; 3 -AR, 3 -adrenergic receptor; AT, adipose tissue; Ct, cycle threshold; E, embryonic day; FFA, free fatty acid; HOMA-IR, homeostasis model assessment of insulin resistance; ing, inguinal; gon, perigonadal; HIF, hypoxia-inducible factor; mes, mesenteric; PGC, PPAR coactivator; PlGF, placental growth factor; PPAR, peroxisome proliferator-activated receptor; RT, room temperature; UCP, uncoupling protein; VEGF, vascular endothelial growth factor; VEGFR, VEGF receptor; wt, wild type. Endocrinology is published monthly by The Endocrine Society ( the foremost professional society serving the endocrine community Pregnancy-induced metabolic changes are regulated by signals from an expanded adipose organ. Placental growth factor (PlGF), acting through vascular endothelial growth factor receptor-1, may be among those signals. There is a steep rise in circulating PlGF during normal pregnancy, which is repressed in gravidas who develop preeclampsia. PlGF-deficiency in mice impairs adipose vascularization and development. Here we studied young-adult PlGF-deficient (PlGF / ) and wild-type mice on a high-fat diet in the nongravid state and at embryonic day (E) 13.5 or E18.5 of gestation. Litter size and weight were normal, but E18.5 placentas were smaller in PlGF / pregnancies. PlGF / mice showed altered intraadipose dynamics, with the following: 1) less blood vessels and fewer brown, uncoupling protein (UCP)-1-positive, adipocytes in white sc and perigonadal fat compartments and 2) white adipocyte hypertrophy. The mrna expression of 3 - adrenergic receptors, peroxisome proliferator-activated receptor- coactivator-1, and UCP-1 was decreased accordingly. Moreover, PlGF / mice showed hyperinsulinemia. Pregnancy-associated changes were largely comparable in PlGF / and wild-type dams. They included expanded sc fat compartments and adipocyte hypertrophy, whereas adipose expression of key angiogenesis/adipogenesis (vascular endothelial growth factor receptor-1, peroxisome proliferatoractivated receptor- 2 ) and thermogenesis ( 3 -adrenergic receptors, peroxisome proliferator-activated receptor- coactivator-1, and UCP-1) genes was down-regulated; circulating insulin levels gradually increased during pregnancy. In conclusion, reduced adipose vascularization in PlGF / mice impairs adaptive thermogenesis in favor of energy storage, thereby promoting insulin resistance and hyperinsulinemia. Pregnancy adds to these changes by PlGF-independent mechanisms. Disturbed intraadipose dynamics is a novel mechanism to explain metabolic changes in late pregnancy in general and preeclamptic pregnancy in particular. (Endocrinology 149: , 2008) to VEGF receptor-1 (VEGFR-1/Flt-1) (10). There are at least three isoforms, but PlGF-2 is the only isoform expressed in the mouse. Its name is derived from the strong expression in trophoblast and the putative role in placental development (11, 12), yet PlGF is expressed in numerous other cell types including endothelial cells (13) and adipocytes (6). Circulating PlGF increases steeply during pregnancy, with peak levels attained in the early third trimester; this increment is dampened in gravidas who develop preeclampsia, a disease characterized by placental and endothelial cell dysfunction (14). Whereas PlGF is not necessary for embryonic angiogenesis, it is required for the neovascularization that occurs with wound healing, fracture repair, ischemia, and cancer (15, 16). PlGF is also involved in AT neovascularization because PlGF-deficient (PlGF / ) mice showed reduced AT vascularization and were partially resistant for AT expansion when fed an obesogenic diet (17). Also, PlGF expression is up-regulated in sc fat depots of obese mice (6). These observations led us to hypothesize that PlGF is one of the proangiogenic factors that regulate AT vascularization and expansion during pregnancy. In the current study, we assessed the gestational changes in AT mass and physiology in normal and PlGF / mice fed a high-fat diet. Our a priori hypothesis was that PlGF deficiency would impair fat mass expansion through its effect on vascularization.
2 Hemmeryckx et al. PlGF and Adipose Tissue in Pregnancy Endocrinology, May 2008, 149(5): Materials and Methods Animals and procedures The experiments were approved and supervised by the Ethical Committee for Animal Experimentation at the Katholieke Universiteit Leuven. Breeding couples of wild-type (wt) and PlGF / mice in a 50% 129Sv 50% Swiss background were obtained from P. Carmeliet (Center for Transgene Technology and Gene Therapy, VIB-3, Leuven, Belgium) and maintained as described (15). The animals were housed individually in temperature-, humidity-, and light-controlled conditions with ad libitum access to water and a standard diet with a caloric value of 10.9 kj/g, 13% fat-derived (KM-04-k12, Muracon; Carfil, Oud-Turnhout, Belgium). At the age of 8 wk, wt and PlGF / females were mated overnight with wt and PlGF / males, respectively; the presence of a copulatory plug the next morning indicated successful mating [i.e. embryonic day (E) 0.5 of gestation]. Both successfully (gravid) and nonsuccessfully (nongravid controls) mated females were weighed at this time and switched to a Western-style high-fat diet with a caloric value of 20.1 kj/g, 42% of which is derived from fat (TD 88137; Harlan Teklad, Madison, WI). This diet was introduced to promote AT angiogenesis and fat deposition (6, 17). Food intake was recorded daily by weighing the pellets to the nearest 0.01 g, and an average was calculated at the end of the experiment for each mouse; an average was then calculated per group. Food spillage was minimal and was not taken into account. We studied gravid mice at gestational ages E13.5 or E18.5; all groups were weighed 7 d after diet switching, and the E18.5 group and the nongravid controls at the 14- and 17-d time point as well. At E13.5 or E18.5, the mice were fasted from 0900 to 1300 h and weighed. After very brief exposure to diethylether, blood was sampled from the retroorbital sinus by an uncoated hematocrit capillary tube (Hirschmann, Eberstadt, Germany) and collected into a heparin-li/ NaF-coated Eppendorf tube (Analis, Ghent, Belgium). Blood glucose was measured on the spot using Glucocard strips and a glucometer (Memory 2; Menarini, Florence, Italy). The remainder was rapidly centrifuged, aliquoted, and stored at 20 C until analysis. In the gravid mice, a median laparotomy was performed subsequently. To ensure effective anesthesia for both dam and fetuses during the procedure, the animals were given pentobarbital (60 mg/kg) ip (Nembutal; Santé Animale, Brussels, Belgium) 5 min before surgery. Blood was sampled from the axilla of each fetus by a Pasteur pipette and pooled into one heparinized Eppendorf tube per litter; the plasma samples were stored at 20 C. Individual placentas and fetuses were weighed. All animals were euthanized by gently elongating the spinal cord. Subcutaneous inguinal (ing) and trunk fat were exposed and removed after a vertical midline skin incision from the pelvic bone to the sternum and two 45 oblique incisions from the lower sternum to the hindlimbs. We defined ing fat as sc fat below the hindlimb level, and trunk fat as sc fat below the sternum but above the hindlimbs extending into the dorsal wall of the trunk. Perigonadal (gon), and mesenteric (mes) fat were removed from the abdominal cavity. All AT samples were weighed and processed for histology or snap frozen into liquid nitrogen and stored at 80 C for quantitative real-time PCR. A small piece of the tail was preserved to confirm the genotype by PCR, as described (16). Cold exposure Nine nongravid PlGF / mice (8 wk, high fat diet since 18 d) were caged individually in a cold room (5 C) for the last 7 d. Rectal temperature was recorded before and daily during cold exposure, using a Vaseline-coated digital thermometer. Fat depots were obtained as described. Plasma assays Insulin and leptin concentrations were determined using mousespecific ELISA kits (Mercodia, Uppsala, Sweden) and Linco Research (Millipore, Brussels, Belgium), respectively. Free (nonesterified) fatty acids (FFA) were measured enzymatically using the nonesterified fatty acid C assay adapted to 96-well microtiter plates (Wako, Neuss, Germany). In the fetuses, glucose was measured enzymatically using the YSI 2300 STAT PLUS glucose analyzer (Ankersmid, Wilrijk, Belgium) and insulin with the ELISA. Histological and histochemical analyses AT biopsies were fixated in Shandon Zinc Formal-Fixx (Anatomical Pathology International, Chester, UK) for 20 h, washed thrice (10 min each) in PBS, transferred to 70% ethanol at room temperature (RT), and embedded in paraffin. For the measurement of adipocyte area (size), 10- m sections were processed onto Superfrost Plus slides (Menzel-Glaser, Braunschweig, Germany) and hematoxylin-eosin stained. At least seven fields per animal were analyzed using the KS400 image analyzer (Zeiss, Jena, Germany) at magnification 25, thus assessing (mean sem) adipocytes per animal. The estimated adipocyte density was calculated as the ratio of the number of adipocytes to the total section area. The results of all histological analyses were averaged per animal and per group. For the measurement of AT vascularization, 10- m sections were deparaffinized in xylene (VWR, Leuven, Belgium) and dehydrated in a downgraded series of alcohol solutions. Endogenous peroxidase signals were quenched by a 3% hydrogen peroxide block (Merck KGaA, Darmstadt, Germany) 10 min at RT. Sections were blocked (30 min, RT) with TNB [0.1 m Tris-HCl (ph 7.5), 0.15 NaCl, 0.5% blocking reagent (TSA Cyanine 3 System, PerkinElmer, Boston, MA)] and then stained with the biotinylated version of Bandeiraea (Griffonia) Simplicifolia BSI lectin (1:50 dilution; Sigma-Aldrich, Bornem, Belgium) to visualize endothelial cells in blood vessels. The Cy3 tyramide reagent (TSA Cyanine 3) was applied to amplify the signal. Photographs were taken from eight fields under fluorescence microscopy at magnification 20 using the Axio Vision software release 4.5 and 4.6 (Zeiss) and analyzed using the KS400 image analyzer (Zeiss). The total section area was delineated, and the number of individual blood vessels present in that area was indicated. The blood vessel density was calculated as the ratio of blood vessel number to total section area (6). We noticed, as described by others (18, 19), the variable presence of brown adipocytes in these white fat depots, characterized by their multilocular appearance and brown-reddish color owing to a high vascularization pattern. To quantitate the area consisting of brown adipocytes, we delineated the total section area and subtracted the areas consisting of identifiable blood vessels or mammary glandular tissue (for the sc fat depots, particularly in gravid mice) in the hematoxylin-eosin stained sections. We then delineated the areas (islets) consisting of brown adipocytes and calculated the brown fat fraction (brown adipocyte area/ total fat area, as a percent). To confirm the presence of brown adipocytes, ing fat sections were processed histochemically for uncoupling protein (UCP)-1, a brown adipocyte-specific marker (20). Ten-micrometer sections were deparaffinized in tolyol (Labonord, Rekkem, Belgium) and dehydrated in 100% ethanol; endogenous peroxidase signals were blocked as described. The samples were then washed and the epitopes of the antigen were exposed by digesting the tissues with trypsin (VWR) for 20 min at RT. To reduce aspecific background, a blocking solution containing 2% BSA (Sigma-Aldrich), 1% milk powder (Nestlé, Brussels, Belgium), 0.1% Tween 80 (Merck KGaA), and 0.1% sodium azide (Sigma- Aldrich) in 1% Tris-buffered saline, supplemented with normal goat serum (1:30; Dako Cytomation, Heverlee, Belgium), was applied for 30 min at RT. UCP-1 expression was visualized by incubating the sections overnight at 4 C with a polyclonal rabbit antimouse UCP-1 antiserum (1:500; Sigma-Aldrich), followed by a horseradish peroxidase-coupled goat antirabbit second antibody (Dako Cytomation) incubated for 30 min at RT. To obtain negative controls, the primary antibody was omitted. Color development was initiated by incubating sections with 3,3- diaminobenzidine tetrahydrochloride (Sigma-Aldrich) in the dark and in a moist environment for 10 min at RT. Finally, sections were quickly counterstained with Mayer hematoxylin and dehydrated in isopropanol, followed by xylene baths, and mounted with DEPEX (VWR). The UCP- 1-positive area was measured by image analysis as described for the brown fat fraction. Quantitative RT-PCR All procedures were carried out as described previously (21). Total RNA was extracted from mg homogenized fat using TriPure Reagent (Roche, Mannheim, Germany), and its concentration was determined spectrophotometrically (ND-1000; Nanodrop Technologies, Wilmington, DE). The reverse transcription reaction was initiated from
3 2178 Endocrinology, May 2008, 149(5): Hemmeryckx et al. PlGF and Adipose Tissue in Pregnancy 0.1 g RNA in a volume of 20 l containing TaqMan reverse transcription reagents (these and all subsequent RT-PCR materials were purchased from Applied Biosystems, Lennik, Belgium) and random hexamers. The samples were incubated for 10 min at 25 C, 30 min at 48 C, and 5 min at 95 C. We measured the expression of the following genes by RT-PCR using the ABI 7000 sequence detector (Applied Biosystems): 1) angiogenesis markers PlGF (TaqMan predeveloped assay Mm m1) and its receptor VEGFR-1 (Mm _m1) (6) and angiopoetin-2 (Ang-2, Mm m1); 2) peroxisome proliferator-activated receptor (PPAR)- 2, which is involved in preadipocyte differentiation and AT angiogenesis (22) (forward primer, 5 -CGCTGATGCACTGCCTATGA- 3, reverse primer, 5 -AATGGCATCTCTGTGTCAACCA-3, and probe, 5 -(FAM)-CACTTCACAAGAAATTAC-3 ); 3) the adipokines leptin (Mm m1), apelin (Mm m1), and TNF- (Mm m1), which are believed to reflect adipocyte size (21, 23, 24); 4) markers of adaptive thermogenesis in mitochondria, 3 -adrenergic receptors ( 3 -AR) (Mm m1), PPAR coactivator (PGC)-1 (Mm m1), and UCP-1 (Mm m1) (20, 25); and 5) hypoxia-inducible factor (HIF)-1 (Mm m1), a regulator of the adaptive response to tissue hypoxia (26). 18s rrna primers and Vic-Tamra probe were used as endogenous reference; a validation experiment was completed for each marker to confirm equal PCR efficiency for target gene and reference. We also confirmed a comparable expression of 18s rrna in gon and ing fat from the four groups. The RT-PCR consisted of a 1:8 dilution of cdna, 2 Taqman universal PCR master mix and 10 primers (unlabeled)/probe (Taqman minor groove binder probe, FAM labeled) sets for each target gene and endogenous control in a final volume of 10 l. Thermal cycling conditions were: 50 C for 2 min, 95 C for 10 min, followed by 50 cycles (for PGC-1 60 cycles) of 15 sec at 95 C and 1 min at 60 C. Duplicate samples were run and the increase in fluorescence was monitored using the Sequence Detector 1.1 (Applied Biosystems, Lennik, Belgium) software. The data were obtained as cycle threshold (Ct) values and normalized to the reference ( Ct Ct target Ct 18s rrna). We then calculated the expression of the samples relative to that of the calibrator (i.e. wt nongravid) group as Ct Ct sample Ct calibrator. Finally, the mrna expression levels of the target genes were expressed as 2 Ct ( sem), with the numerical values indicating the n-fold change in expression of the target mrna relative to the calibrator and the sem calculated as [2 Ct ln2sd( Ct)/(number of samples) 1/2 ]. Data analysis We used the NCSS 2004 software (Kaysville, UT). Two indirect indices of insulin resistance were calculated: 1) the homeostasis model assessment of insulin resistance (HOMA-IR) as fasting insulin (microunits per milliliter 1 ) fasting glucose (millimoles)/22.5 and 2) the product of insulin (nanograms per milliliter 1 ) and FFA (millimoles) (27). All results are presented as means sem. Differences between two groups (e.g. fetal insulin concentrations on E18.5) were analyzed using two-sample t tests and differences between more than two groups by ANOVA. Because the study contained six groups of mice with a different genetic background (wt or PlGF / ) and gestational status (nongravid, E13.5, or E18.5), we first assessed the main effects of background and gestational status, as well as their possible interaction, by general linear model (two-factor) ANOVA. If an interaction was detected at P 0.05, pointing to a heterogeneity effect, we proceeded to one-way ANOVA, examining the six groups individually. If one-way ANOVA confirmed a P 0.05, we examined the intergroup differences (PlGF / vs. wt of same gestational status, E13.5 and E18.5 gravid vs. nongravid of same background) by Fisher s least significant differences test. Results Hyperinsulinemia despite lower placental weight in PlGF / mice PlGF deficiency did not affect body weight and had no consistent effects on food intake, blood glucose, or plasma FFA and leptin concentrations (Table 1). There was, however, a slight increase in weight gain since diet switching in the PlGF / animals. Moreover, PlGF deficiency clearly increased plasma insulin concentrations, the insulin FFA product (Fig. 1), and the HOMA-IR index (data not shown). As expected, gravid mice consumed more food and gained more weight than did nongravid mice. Gestationinduced differences included a rise in circulating insulin and leptin in both wt and PlGF / dams. FFA and insulin FFA were raised in E18.5 dams. Comparison of wt and PlGF / dams showed no differences on E18.5; on E13.5, we detected a difference in glucose (decreased, P 0.05) and leptin (increased, P 0.005) concentrations between PlGF / and wt dams. Regarding the reproductive outcome (Table 2), we found a small decrement in individual fetal weight in PlGF / vs. wt animals but no difference in total litter weight. Placental weight, however, was 13% lower (P 0.01) on E18.5 in PlGF / mice. Fetal plasma glucose and insulin were comparable in E18.5 PlGF / and wt fetuses. Altered intraadipose dynamics in PlGF / mice PlGF deficiency had no unequivocal effect on overall adiposity (Table 3) but was clearly associated with a decrease in AT blood vessel density and the fraction of AT consisting of brown adipocytes or UCP-1-positive cells (see also Fig. 2). PlGF / mice showed larger white adipocytes in gon and ing fat, with a concomitant decrease in estimated adipocyte density. Corroborating these histomorphometric data, the mrna expression of several mitochondrial markers ( 3 -AR, PGC- 1, and UCP-1) was down-regulated in all examined fat pads of PlGF / animals (Table 4). This down-regulation was most marked for UCP-1 (29 51% of control levels). The ex- TABLE 1. General parameters of nongravid and gravid (gestational d 13.5 and 18.5) wt and PlGF / mice Nongravid wt wt E13.5 (n 7) wt E18.5 Nongravid PlGF / (n 12) PlGF / E13.5 (n 8) PlGF / E18.5 Background Gestational status Interaction Weight (g) (9) 0.72 < Weight gain (g) (7) < Food intake (g/d) a a b a a 0.20 < Blood glucose (mm) (7) a,b a Plasma FFA (mm) 1, , (7) 1, < Plasma leptin (ng/ml) (9) a a,b a 0.26 < Statistical analysis was performed as explained in Materials and Methods. Briefly, two-factor ANOVA was used to examine the data. If an interaction was noted at the P 0.05 level, we reassessed the data of the six groups by one-way ANOVA and (if P 0.05) used Fisher s least significant differences multiple-comparison test to examine intergroup differences. The number of animals is in parentheses. a Significant differences in gravid (E13.5 or E18.5) vs. the nongravid mice of the same genetic background. b Significant differences in PlGF / vs. wt mice of the same gestational status.
4 Hemmeryckx et al. PlGF and Adipose Tissue in Pregnancy Endocrinology, May 2008, 149(5): FIG. 1. Plasma insulin concentrations (A) and the insulin resistance index [(insulin) (FFA)] (B) in nongravid and gravid PlGF / and wt mice. Differences according to genetic background and gestational status were assessed by two-factor ANOVA, and Fisher s least significant differences multiple-comparison test was used to identify statistically significant between backgrounds of the same gestational status [ : P (A) and : P (B)] and differences between animals with a different gestational status of the same background [f: P 0.02 (A) and x: P (B)]. No interaction between background and gestational status was found (P 0.34 for both A and B). pression of HIF-1 was up-regulated in ing fat of PlGF / mice, but the expression of Ang-2 remained within the normal range (data not shown). The expression of the apelin and TNF- (not shown) was normal, whereas the expression of leptin was up-regulated in nongravid and E13.5 animals. Cold exposure in PlGF / mice lowered their body temperature from C to C after 24 h (P 0.001) and C after 7 d (P 0.05). Compared with mice at room temperature, the percent of brown adipocytes in fat was slightly increased in ing fat ( %; P 0.06) and gon fat ( %; P 0.07) but not trunk fat ( %; P 0.78). Additive effect of pregnancy (Tables 3 and 4 and Fig. 2) Gravidity was accompanied by expanded sc trunk and ing fat compartments; this expansion was already evident on TABLE 2. Reproductive outcome of wt and PlGF / mice E13.5 in PlGF / dams but only on E18.5 in wt dams. By contrast, there was no consistent expansion of gon fat or an expanded mes fat compartment (data not shown). In sc fat, the adipocytes were larger during gestation with a concomitant drop in estimated adipocyte density. AT blood vessel density did not change appreciably in the course of gestation, but blood vessel size was lower among gravid animals; for ing fat, the post hoc test indicated a significant decrease between nongravid and E13.5 mice. The brown adipocyte fraction was decreased in sc trunk fat of gravid animals. Thus, the additive effects of PlGF-deficiency and gestation (E18.5) resulted in a 67% decrement in the brown adipocyte fraction in this fat depot. The histomorphometric data were corroborated by gene expression analysis. During pregnancy, the expression levels of the adipogenesis marker PPAR- 2 were markedly down-regulated in sc trunk and ing fat (levels on E18.5 were 8 31%, compared with nongravid levels). By contrast, apelin expression was dramatically up-regulated (10- to 18-fold) in sc fat and 1.4-fold in gon fat. However, there was no up-regulation of leptin expression in sc fat, and leptin mrna levels were actually suppressed during gestation in PlGF-null mice. TNF- mrna levels did not change in any fat depot during the course of gestation (data not shown). The expression of PlGF and VEGFR-1 was not increased during pregnancy; in fact, the expression of PlGF was 23% lower in gon fat of E18.5 wt dams, compared with their nongravid counterparts, and the VEGF-R1 expression was robustly repressed in sc fat. Gestation did not influence Ang-2 mrna levels (not shown). The expression of genes regulating mitochondrial biogenesis and function ( 3 -AR, PGC-1, and UCP-1) in sc fat were down-regulated during gestation. Again, the effects of PlGF deficiency and gestation on the mitochondrial markers were additive so that the expression level of 3 -AR and PGC-1 in sc fat of E18.5 PlGF / dams was less than 20% of that measured in nongravid wt mice and the expression of UCP-1 less than 1%. Associations between adipose changes and hyperinsulinemia (Table 5) Whereas insulin was not correlated with FFA concentrations (r 0.12, P 0.39), insulin, the insulin FFA product, and the HOMA-IR index were positively correlated with several measures of adiposity (body weight, weight gain, weight of all sampled fat pads, circulating leptin), adipocyte size, and apelin gene expression in sc fat. In addition, insulin and the derived indices were inversely correlated with blood wt E13.5 (n 7) wt E18.5 PlGF / E13.5 (n 8) PlGF / E18.5 Background Gestational time Interaction Litter size Individual fetal weight (mg) (49) (51) (53) (60) 0.01 < Individual placental weight (mg) 65 4 (56) (51) 73 4 (53) 98 4 a (60) 0.34 < Litter weight (mg) < Fetal plasma glucose (mm) ND (8) ND (8) 0.77 b Fetal plasma insulin ( g/liter) ND ND b Statistical analysis was as explained in Materials and Methods and Table 1. The number of animals is in parentheses. ND, Not determined. a and b See Table 1 legend.
5 2180 Endocrinology, May 2008, 149(5): Hemmeryckx et al. PlGF and Adipose Tissue in Pregnancy TABLE 3. Weight and histology of adipose tissue in nongravid and gravid wt and PlGF / mice Nongravid wt wt E13.5 (n 7) wt E18.5 vessel density, the brown adipocyte fraction, and the expression of 3 -AR and UCP-1 in sc fat. Blood vessel density was correlated with the brown adipocyte fraction (r 0.62 in ing and gon fat, P 0.001) and the expression of UCP-1 (r 0.30 in ing fat, P 0.03). Discussion PlGF deficiency and AT physiology We documented that PlGF deficiency alters the adipometabolic phenotype of postpubertal mice fed a high-fat diet for d. PlGF / mice showed: 1) a lower fraction of brown adipocytes, which was only slightly increased by cold exposure, and a reduced AT expression of thermogenic genes; and 2) larger white adipocytes. These AT changes were accompanied by hyperinsulinemia. We submit that the altered adipometabolic phenotype is caused by a reduction in microvessel density. Nongravid PlGF / (n 12) PlGF / E13.5 (n 8) PlGF / E18.5 Background Gestational status Interaction Weight of individual fat compartment Ing a a,b b 0.02 < (mg) Trunk a a,b b 0.95 <0.001 <0.001 Gon a,b Total fat weight (mg) a a,b <0.001 <0.001 <0.001 Blood vessel density ( 10 6 m 2 1 ) Ing < Gon < Blood vessel size ( m 2 ) Ing (9) (11) < Gon a a (11) < Brown fat fraction (%) Ing (11) < Trunk (9) Gon (7) UCP-1-positive fraction (%) Ing (11) (9) < Adipocyte size ( m 2 ) Ing < Trunk Gon < Adipocyte density 10 6 ( m 2 ) 1 Ing a (11) < Trunk Gon Total fat weight is the combined weight of the sc inguinal and trunk (defined in Materials and Methods) and the intraabdominal gon and mes (data not shown) fat depots. Statistical analysis was performed as explained in Materials and Methods and Table 1. a and b See Table 1 legend. Brown adipocytes are more abundant than previously recognized. Nuclear imaging has identified several active brown fat depots in adults (28). In rodents, brown adipocytes are present not only in a separate interscapular depot but also to a variable degree in white fat depots. The latter depends on the genetic strain and age of the animals, the presence of 3 -AR in the fat depot, and the dietary fat content (18, 25, 29, 30). In our young-adult wt mice fed a high-fat diet, 16 29% of AT sections consisted of brown adipocytes with the largest proportion in sc trunk fat. The role of brown adipocytes is thermogenesis, i.e. the release of heat through oxidation of fatty acids, made possible by the abundant mitochondria and UCP-1 expression (20, 25). Their intense substrate and oxygen requirement and the release of heat necessitate an intricate microvascular network (7, 31). The thermogenic capacity of brown adipocytes within white fat depots likely constitutes a physiological mechanism to curb fat accumulation (32). FIG. 2. Both PlGF deficiency and gestation decreased the UCP-1-positive fraction in ing fat. Arrows point to brown adipocyte islets in hematoxylin and eosin-stained (A C) and UCP-1-immunostained sections (D F) of nongravid wt fat sample (A and D), nongravid PlGF / fat sample (B and E), and gravid E18.5 wt fat sample (C and F). Bar, 100 m.
6 Hemmeryckx et al. PlGF and Adipose Tissue in Pregnancy Endocrinology, May 2008, 149(5): TABLE 4. Adipose gene expression markers in nongravid and gravid wt and PlGF / mice Nongravid wt wt E13.5 (n 7) wt E18.5 Norepinephrine and 3 -AR activate the thermogenic process (25). Upon adrenergic binding, PGC-1 expression is induced, which stimulates brown adipocyte differentiation through coactivation of PPAR- on the UCP-1 promoter (33, 34). Dietary-induced or genetic obesity in mice represses ing PGC-1 mrna levels, whereas PPAR- agonists augment PGC-1 and UCP-1 expression (20). However, mice with a disruption of the adipocyte-specific PPAR- 2 gene showed no (35) or mild (36) changes in brown AT appearance and gene expression. In PlGF-null mice, the AT expression of 3 -AR, PGC-1, and UCP-1 was down-regulated, consistent with impaired thermogenesis, but the expression of the Nongravid PlGF / (n 12) PlGF / E13.5 (n 8) PlGF / E18.5 Background Gestational status Interaction Angiogenesis PlGF Ing ND ND ND 0.94 c Trunk (6) ND ND ND 0.82 c Gon a ND ND ND 0.03 c VEGF-R1 Ing < Trunk < Gon (9) Adipogenesis/angiogenesis PPAR 2 Ing (9) 0.62 < Trunk a a 16 6 a 0.92 < Gon Adipokines Apelin Ing , (9) 0.09 < Trunk < Gon (7) Leptin Ing a,b a <0.001 < Trunk a a 0.02 < Gon b a,b <0.001 Mitochondrial markers 3 -AR Ing (6) (8) (8) 0.40 < Trunk a a a 11 4 a,b < Gon (7) PGC-1 Ing (9) (7) Trunk (7) 14 4 (9) < Gon UCP-1 Ing (7) <0.001 < Trunk (9) (7) (9) <0.001 < Gon (9) Hypoxia HIF-1 Ing ND ND Trunk ND ND Gon ND ND Gene expression data were transformed from Ct values to n-fold differences (decrease or increase) in expression of the target mrna to the calibrator sample (nongravid wt sample, expression level set at 1.0). The results were then multiplied by 100 to present the data as percentages SEM. Statistical analysis was performed as explained in Materials and Methods and Table 1. ND, Not determined. a and b See Table 1 legend. c P value obtained by one-way ANOVA (wt mice only). PPAR- 2 isoform was normal. Although the mes fat depot is thought to be metabolically important, we found no effects of PlGF deficiency on mes fat, pointing to a depot-specific response (37). VEGF is the dominant angiogenic factor in white AT (5) and probably also in brown AT (31, 38). But there was no consistent up-regulation of VEGF-A expression in PlGF / mice (17), and the expression of Ang-2, another angiogenic factor, was unchanged in this study. White adipocyte hypertrophy results in adipokine dysregulation, mediated in part by impaired AT perfusion and hypoxia (39, 40). HIF-1 expression was indeed moderately TABLE 5. Spearman rank correlation coefficients between insulin or insulin resistance indexes (HOMA-IR, insulin FFA) and adiposity parameters (n 52 56) Insulin HOMA-IR Insulin FFA Correlation coefficient P value Correlation coefficient P value Correlation coefficient P value Body weight ( ) ( ) ( ) Weight gain ( ) ( ) ( ) Total fat weight ( ) ( ) ( ) Plasma leptin ( ) ( ) ( ) Adipocyte size Ing ( ) ( ) ( ) Trunk ( ) ( ) ( ) Blood vessel density Ing ( ) ( ) ( ) Brown fat fraction Ing ( ) ( ) ( ) Trunk ( ) ( ) ( ) Apelin (mrna) Ing ( ) ( ) ( ) Trunk ( ) ( ) ( ) AR (mrna) Ing ( ) ( ) ( ) Trunk ( ) ( ) ( ) UCP-1 (mrna) Ing ( ) ( ) ( ) Trunk ( ) ( ) ( )
7 2182 Endocrinology, May 2008, 149(5): Hemmeryckx et al. PlGF and Adipose Tissue in Pregnancy up-regulated in ing fat of PlGF / animals. A correlation between adipocyte size and leptin mrna levels within fat depots is well established (23). Here we documented elevated leptin mrna levels in nongravid and E13.5 PlGF / vs. wt mice and higher plasma leptin in E13.5 PlGF / dams. Impaired thermogenesis and white-adipocyte hypertrophy are expected to result in higher weight gain and fat accumulation. A slightly higher weight gain was indeed observed in PlGF / mice, and E13.5 PlGF / dams had a larger total fat mass than wt dams. The PlGF / female mice in the current study were also hyperinsulinemic, whereas a previous study in PlGF / male mice on standard chow did not reveal differences in glucose or insulin tolerance (41). It appears from these data that the metabolic effects of PlGF deficiency depend on animal characteristics (sex, age) and the duration of high-fat feeding (17, 24, 42). Hypoinsulinemia was observed in mice treated with the angiogenesis inhibitor TNP-470, which suppressed white adipogenesis (27). Thus, different angiogenic factors appear to interfere differently with adipogenesis and insulin metabolism. Pregnancy-induced changes We found that pregnancy resulted in an expansion of sc fat compartments and adipocyte hypertrophy, whereas the expression of genes regulating angiogenesis, adipogenesis, and thermogenesis was down-regulated. These changes were associated with gradual hyperinsulinemia. Comparable changes occurred in wt and PlGF / dams, indicating that the pregnancy-induced adipometabolic changes are largely if not completely PlGF independent. The adipocyte hypertrophy was accompanied by a gradual, robust up-regulation of apelin expression in sc fat. Apelin is oversecreted in obese and hyperinsulinemic states (40, 43). We documented that apelin mrna levels correlated with adipocyte size in leptin-resistant mice (21). In rats, the adipocyte hypertrophy of pregnancy (8, 9) was associated with reduced adiponectin expression (3). The observation that adipose vascularization failed to increase during gestation was contrary to our a priori hypothesis. Blood vessel size was decreased in midgestation; the AT expression of PlGF and Ang-2 was normal or decreased, whereas VEGFR-1 expression was down-regulated robustly in sc fat. Adipogenesis appeared to be repressed accordingly, as suggested by the strong down-regulation of PPAR- 2 mrna levels in sc and gon fat. The mechanism driving the inhibition of the angiogenesis-adipogenesis sequence during pregnancy warrants further investigation. In rats, pregnancy is accompanied by atrophy of the interscapular brown fat depot (25). Here we confirmed a reduced fraction of brown adipocytes, as well as strongly down-regulated gene expression of 3 -AR, PGC-1, and UCP-1, in sc fat of gravid mice. It has been proposed that brown fat atrophy reflects the increased energy use imposed by the growing fetus (25). However, an impaired neoangiogenic response might also have implications for brown adipogenesis. The effects of pregnancy and PlGF deficiency on insulin parameters were additive. E18.5 dams showed increased concentrations of both insulin and FFA concentrations, confirming data in rats (8). Overall, we found powerful correlations between white adipocyte size or brown fat fraction in sc trunk fat and insulin parameters (Table 5), extending previous observations in humans and animal models. Indeed, the size of adipocytes in sc trunk fat was an independent predictor of type 2 diabetes in diabetesprone Pima Indians (44). Similarly, studies in mice treated with PPAR- modulators showed concordant effects on adipocyte size and insulin resistance (45). Regarding brown fat, there is preliminary evidence in man that less brown fat confers insulin resistance (28); in mice, the evidence that brown fat ablation produces insulin resistance is quite strong (46, 47). The present study focused on AT. Yet it is clear that we need to delineate further the metabolic phenotype of gravid PlGF / mice using insulin tolerance tests, glucose tolerance tests, and perhaps hyperinsulinemic clamps. Also, we need to study insulin secretion dynamics. Human preeclamptic pregnancy is characterized by reduced concentrations of proangiogenic factors (PlGF) and augmented concentrations of antiangiogenic factors (soluble Flt-1, endoglin) (14). Women with preeclampsia are also more likely to be obese and hyperinsulinemic (48). Although preeclamptic pregnancies would be expected to be less insulin resistant than normal pregnancies owing to smaller placentas (2), there is no evidence that such scenario occurs. In fact, there is some evidence pointing toward an adverse adipose phenotype (e.g. increased TNF- and leptin to adiponectin ratio) related to the overabundance of antiangiogenic factors (49); this may have implications for insulin metabolism. Similarly, in PlGF / pregnancies, placental weight was lower at term, whereas the adipose phenotype was adversely affected. Conclusions PlGF-deficient mice fed a high-fat diet showed disturbed intraadipose dynamics with reduced brown adipocyte activity but white adipocyte hypertrophy and hyperinsulinemia. These effects were accentuated during pregnancy through PlGF-independent mechanisms. Our results may be relevant to explain the hyperinsulinemia of pregnancy and preeclamptic pregnancy in particular. Acknowledgments We thank Sven Terclaevers and Annemie De Wolf for excellent technical assistance, Nele Geusens and Robert Pijnenborg for valuable discussions, and Suzan Lambin for assistance with the RT-PCR. Received September 14, Accepted January 28, Address all correspondence and requests for reprints to: J. Verhaeghe, M.D. Ph.D., Department of Obstetrics and Gynecology, U.Z. Gasthuisberg, Herestraat 49, 3000 Leuven, Belgium. johan.verhaeghe@uz.kuleuven.be. This work was supported by Grants G and G from the Fonds voor Wetenschappelijk Onderzoek-Vlaanderen (Belgium). The Center for Molecular and Vascular Biology is supported by the Excellentie financiering Katholieke Universiteit Leuven Leuven (EF/05/13) and the Leducq Foundation International Network Against Thrombosis (LINAT, Paris, France) and IUAP-P6/30. Disclosure Statement: The authors have nothing to disclose. References 1. Sohlström A, Forsum E 1995 Changes in adipose tissue volume and distribution during reproduction in Swedish women as assessed by magnetic resonance imaging. Am J Clin Nutr 61: Barbour LA, McCurdy CE, Hernandez TL, Kirwan JP, Catalano PM, Fried-
8 Hemmeryckx et al. PlGF and Adipose Tissue in Pregnancy Endocrinology, May 2008, 149(5): man JE 2007 Cellular mechanisms for insulin resistance in normal pregnancy and gestational diabetes. Diabetes Care 30(Suppl 2):S112 S Sevillano J, de Castro J, Bocos C, Herrera E, Ramos MP 2007 Role of insulin receptor substrate-1 serine 307 phosphorylation and adiponectin in adipose tissue insulin resistance in late pregnancy. Endocrinology 148: Crandall DL, Hausman GJ, Kral JG 1997 A review of the microcirculation of adipose tissue: anatomic, metabolic, and angiogenic perspectives. Microcirculation 4: Hausman GJ, Richardson RL 2004 Adipose tissue angiogenesis. J Anim Sci 82: Voros G, Maquoi E, Demeulemeester D, Clerx N, Collen D, Lijnen HR 2005 Modulation of angiogenesis during adipose tissue development in murine models of obesity. Endocrinology 146: Cao Y 2007 Angiogenesis modulates adipogenesis and obesity. J Clin Invest 117: Ramos MP, Crespo-Solans MD, del Campo S, Cacho J, Herrera E 2003 Fat accumulation in the rat during early pregnancy is modulated by enhanced insulin responsiveness. Am J Physiol Endocrinol Metab 285:E318 E Pujol E, Proenza A, Llado I, Roca P 2005 Pregnancy effects on rat adipose tissue lipolytic capacity are dependent on anatomical location. Cell Physiol Biochem 16: Auterio M, Waltenberger J, Communi D, Kranz A, Moons L, Lambrechts D, Kroll J, Plaisance S, De Mol M, Bono F, Kliche S, Fellbrich G, Ballmer-Hofer K, Maglione D, Mayr-Beyrle U, Dewerchin M, Dombrowski S, Stanimirovic D, Van Hummelen P, Dehio C, Hicklin DJ, Persico G, Herbert J-M, Communi D, Shibuya M, Collen D, Conway EM, Carmeliet P 2003 Role of PlGF in the intra- and intermolecular cross talk between the VEGF receptors Flt1 and Flk1. Nat Med 9: Achen MG, Gad JM, Stacker SA, Wilks AF 1997 Placenta growth factor and vascular endothelial growth factor are co-expressed during early embryonic development. Growth Factors 15: Vuorela P, Hatva E, Lymboussaki A, Kaipainen A, Joukov V, Persico MG, Alitalo K, Halmesmäki E 1997 Expression of vascular endothelial growth factor and placenta growth factor in human placenta. Biol Reprod 56: Yonekura H, Sakurai S, Liu X, Migita H, Wang H, Yamagishi S, Nomura M, Abedin MJ, Unoki H, Yamamoto Y, Yamamoto H 1999 Placenta growth factor and vascular endothelial growth factor B and C expression in microvascular endothelial cells and pericytes. J Biol Chem 274: Levine RJ, Maynard SE, Qian C, Lim K-H, England LJ, Yu KF, Schisterman EF, Thadhani R, Sachs BP, Epstein FH, Sibai BM, Sukhatme VP, Karumanchi SA 2004 Circulating angiogenic factors and the risk of preeclampsia. N Engl J Med 350: Carmeliet P, Moons L, Luttun A, Vincenti V, Compernolle V, De Mol M, Wy Y, Bono F, Devy L, Beck H, Scholz D, Acker T, DiPalma T, Dewerchin M, Noel A, Stalmans I, Barra A, Blacher S, Vandendriessche T, Ponten A, Eriksson U, Plate KH, Foidart J-M, Schaper W, Charnock-Jones DS, Hicklin DJ, Herbert J-M, Collen D, Persico MG 2001 Synergism between vascular endothelial growth factor and placental growth factor contributes to angiogenesis and plasma extravasation in pathological conditions. Nat Med 7: Maes C, Coenegrachts L, Stockmans I, Daci E, Luttun A, Petryk A, Gopalakrishnan R, Moermans K, Smets N, Verfaillie CM, Carmeliet P, Bouillon R, Carmeliet G 2006 Placental growth factor mediates mesenchymal cell development, cartilage turnover, and bone remodeling during fracture repair. J Clin Invest 116: Lijnen HR, Christiaens V, Scroyen I, Voros G, Tjwa M, Carmeliet P, Collen D 2006 Impaired adipose tissue development in mice with inactivation of placental growth factor function. Diabetes 55: Cinti S 2005 The adipose organ. Prostaglandins Leukot Essent Fatty Acids 73: Rong JX, Qiu Y, Hansen MK, Zhu L, Zhang V, Xie M, Okamoto Y, Mattie MD, Higashiyama H, Asano S, Strum JC, Ryan TE 2007 Adipose mitochondrial biogenesis is suppressed in db/db and high-fat diet-fed mice and improved by rosiglitazone. Diabetes 56: Krauss S, Zhang CY, Lowell BB 2005 The mitochondrial uncoupling-protein homologues. Nat Rev Mol Cell Biol 6: Lambin S, van Bree R, Caluwaerts S, Vercruysse L, Vergote I, Verhaeghe J 2007 Adipose tissue in offspring of Lepr db/ mice: early-life environment vs. genotype. Am J Physiol Endocrinol Metab 292:E262 E Fukumura D, Ushiyama A, Duda DG, Xu L, Tam J, Chatterjee KK, Garkavtsev I, Jain RK 2003 Paracrine regulation of angiogenesis and adipocyte differentiation during in vivo adipogenesis. Circ Res 93:e88 e Guo K-Y, Halo P, Leibel RL, Zhang Y 2004 Effects of obesity on the relationship of leptin mrna expression and adipocyte size in anatomically distinct fat depots in mice. Am J Physiol Regul Integr Comp Physiol 287:R112 R Strissel KJ, Stancheva Z, Miyoshi H, Perfield JW, DeFuria J, Jick Z, Greenberg AS, Obin MS 2007 Adipocyte death, adipose tissue remodeling, and obesity complications. Diabetes 56: Cannon B, Nedergaard J 2004 Brown adipose tissue: function and physiological significance. Physiol Rev 84: Semenza GL 2000 Surviving ischemia: adaptive responses mediated by hypoxia-inducible factor 1. J Clin Invest 106: Bråkenhielm E, Cao R, Gao B, Angelin B, Cannon B, Parini P, Cao Y 2004 Angiogenesis inhibitor, TNP-470, prevents diet-induced and genetic obesity in mice. Circ Res 94: Nedergaard J, Bengtsson T, Cannon B 2007 Unexpected evidence for active brown adipose tissue in adult humans. Am J Physiol Endocrinol Metab 293: E444 E Guerra C, Koza RA, Yamashita H, Walsh K, Kozak LP 1998 Emergence of brown adipocytes in white fat in mice is under genetic control. J Clin Invest 102: Jimenez M, Barbatelli G, Allevi R, Cinti S, Seydoux J, Giacobino J-P, Muzzin P, Preitner F Adrenoceptor knockout in C57BL/6J mice depresses the occurrence of brown adipocytes in white fat. Eur J Biochem 270: Fredriksson JM, Lindquist JM, Bronnikov GE, Nedergaard J 2000 Norepinephrine induces vascular endothelial growth factor gene expression in brown adipocytes through a -adrenoreceptor/camp/protein kinase A pathway involving Src but independently of Erk1/2. J Biol Chem 275: Tiraby C, Tavernier G, Lefort C, Larrouy D, Bouillaud F, Ricquier D, Langin D 2002 Acquirement of brown fat cell features by human white adipocytes. J Biol Chem 278: Puigserver P, Wu Z, Park CW, Graves R, Wright M, Spiegelman BM 1998 A cold-inducible coactivator of nuclear receptors linked to adaptive thermogenesis. Cell 92: Liang H, Ward WF 2006 PGC-1 : a key regulator of energy metabolism. Adv Physiol Educ 30: Medina-Gomez G, Virtue S, Lelliott C, Boiani R, Campbell M, Christodoulides C, Perrin C, Jimenez-Linan M, Blount M, Dixon J, Zahn D, Thresher RR, Aparicio S, Carlton M, Colledge WH, Kettunen MI, Seppänen-Laakso T, Sethi JK, O Rahilly S, Brindle K, Cinti S, Orešič M, Burcelin R, Vidal-Puig A 2005 The link between nutritional status and insulin sensitivity is dependent on the adipocyte-specific peroxisome proliferator-activated receptor- 2 isoform. Diabetes 54: Zhang J, Fu M, Cui T, Xiong C, Xu K, Zhong W, Xiao Y, Floyd D, Liang J, Li E, Song Q, Chen YE 2004 Selective disruption of PPAR 2 impairs the development of adipose tissue and insulin sensitivity. Proc Natl Acad Sci USA 101: Laplante M, Sell H, MacNaul KL, Richard D, Berger JP, Deshaies Y 2003 PPAR- activation mediates adipose depot-specific effects on gene expression and lipoprotein lipase activity: mechanisms for modulation of postprandial lipemia and differential adipose accretion. Diabetes 52: Asano A, Morimatsu M, Nikami H, Yoshida T, Saito M 1997 Adrenergic activation of vascular endothelial growth factor mrna expression in rat brown adipose tissue: implication in cold-induced angiogenesis. Biochem J 328: Hosogai N, Fukuhara A, Oshima K, Miyata Y, Tanaka S, Segawa K, Furukawa S, Tochino Y, Komuro R, Matsuda M, Shimomura I 2007 Adipose tissue hypoxia in obesity and its impact on adipocytokine dysregulation. Diabetes 56: Glassford AJ, Yue P, Sheikh AY, Chun HJ, Zarafshar S, Chan DA, Reaven GM, Quertermous T, Tsao PS 2007 HIF-1 regulates hypoxia- and insulin-induced expression of apelin in adipocytes. Am J Physiol Endocrinol Metab 293:E1590 E Voros G, Lijnen HR 2006 Role of the placental growth factor in glucose metabolism. J Thromb Haemost 4: Christiaens V, Voros G, Scroyen I, Lijnen HR 2007 On the role of placental growth factor in murine adipogenesis. Thromb Res 120: Boucher J, Masri B, Daviaud D, Gesta S, Guigne C, Mazzucotelli A, Castas- Laurell I, Tack I, Kaibichler B, Carpene C, Audigiu Y, Saulnier-Blache JS, Valet P 2005 Apelin, a newly identified adipokine up-regulated by insulin and obesity. Endocrinology 146: Weyer C, Foley JE, Bogardus C, Tataranni PA, Pratley RE 2000 Enlarged subcutaneous abdominal adipocyte size, but not obesity itself, predicts type II diabetes independent of insulin resistance. Diabetologia 43: Rieusset J, Touri F, Michalik L, Escher P, Desvergne B, Niesor E, Wahli W 2002 A new selective peroxisome proliferator-activated receptor antagonist with antiobesity and antidiabetic activity. Mol Endocrinol 16: Lowell BB, Susulic VS, Hamann A, Lawitts JA, Himms-Hagen J, Boyer BB 1993 Development of obesity in transgenic mice after ablation of brown adipose tissue. Nature (London) 366: Mantzoros C, Frederich RC, Ou D, Lowell BB, Maratos-Flier E, Flier JS 1998 Severe leptin resistance in brown fat-deficient uncoupling protein promotordriven diphtheria toxin A mice despite suppression of hypothalamic neuropeptide Y and circulating corticosterone concentrations. Diabetes 47: Seely EW, Solomon CG 2003 Insulin resistance and its potential role in pregnancy-induced hypertension. J Clin Endocrinol Metab 88: Masuyama H, Nakatsukasa H, Takamoto N, Hiramatsu Y 2007 Correlation between soluble endoglin, vascular endothelial growth factor receptor-1, and adipocytokines in preeclampsia. J Clin Endocrinol Metab 92: Endocrinology is published monthly by The Endocrine Society ( the foremost professional society serving the endocrine community.
General Laboratory methods Plasma analysis: Gene Expression Analysis: Immunoblot analysis: Immunohistochemistry:
 General Laboratory methods Plasma analysis: Plasma insulin (Mercodia, Sweden), leptin (duoset, R&D Systems Europe, Abingdon, United Kingdom), IL-6, TNFα and adiponectin levels (Quantikine kits, R&D Systems
General Laboratory methods Plasma analysis: Plasma insulin (Mercodia, Sweden), leptin (duoset, R&D Systems Europe, Abingdon, United Kingdom), IL-6, TNFα and adiponectin levels (Quantikine kits, R&D Systems
Males- Western Diet WT KO Age (wks) Females- Western Diet WT KO Age (wks)
 Relative Arv1 mrna Adrenal 33.48 +/- 6.2 Skeletal Muscle 22.4 +/- 4.93 Liver 6.41 +/- 1.48 Heart 5.1 +/- 2.3 Brain 4.98 +/- 2.11 Ovary 4.68 +/- 2.21 Kidney 3.98 +/-.39 Lung 2.15 +/-.6 Inguinal Subcutaneous
Relative Arv1 mrna Adrenal 33.48 +/- 6.2 Skeletal Muscle 22.4 +/- 4.93 Liver 6.41 +/- 1.48 Heart 5.1 +/- 2.3 Brain 4.98 +/- 2.11 Ovary 4.68 +/- 2.21 Kidney 3.98 +/-.39 Lung 2.15 +/-.6 Inguinal Subcutaneous
GPR120 *** * * Liver BAT iwat ewat mwat Ileum Colon. UCP1 mrna ***
 a GPR120 GPR120 mrna/ppia mrna Arbitrary Units 150 100 50 Liver BAT iwat ewat mwat Ileum Colon b UCP1 mrna Fold induction 20 15 10 5 - camp camp SB202190 - - - H89 - - - - - GW7647 Supplementary Figure
a GPR120 GPR120 mrna/ppia mrna Arbitrary Units 150 100 50 Liver BAT iwat ewat mwat Ileum Colon b UCP1 mrna Fold induction 20 15 10 5 - camp camp SB202190 - - - H89 - - - - - GW7647 Supplementary Figure
Yiying Zhang, PhD Research Scientist. Research Summary:
 Yiying Zhang, PhD Research Scientist Research Summary: Address: Naomi Berrie Diabetes Center at Columbia University Medical Center Russ Berrie Medical Science Pavilion 1150 St. Nicholas Avenue New York,
Yiying Zhang, PhD Research Scientist Research Summary: Address: Naomi Berrie Diabetes Center at Columbia University Medical Center Russ Berrie Medical Science Pavilion 1150 St. Nicholas Avenue New York,
For pair feeding, mice were fed 2.7g of HFD containing tofogliflozin
 Materials and Methods Pair Feeding Experiment For pair feeding, mice were fed 2.7g of HFD containing tofogliflozin (0.005%), which is average daily food intake of mice fed control HFD ad libitum at week
Materials and Methods Pair Feeding Experiment For pair feeding, mice were fed 2.7g of HFD containing tofogliflozin (0.005%), which is average daily food intake of mice fed control HFD ad libitum at week
Method of leptin dosing, strain, and group housing influence leptin sensitivity in high-fat-fed weanling mice
 Am J Physiol Regul Integr Comp Physiol 284: R87 R100, 2003; 10.1152/ajpregu.00431.2002. Method of leptin dosing, strain, and group housing influence leptin sensitivity in high-fat-fed weanling mice HEATHER
Am J Physiol Regul Integr Comp Physiol 284: R87 R100, 2003; 10.1152/ajpregu.00431.2002. Method of leptin dosing, strain, and group housing influence leptin sensitivity in high-fat-fed weanling mice HEATHER
Human Leptin ELISA Kit
 Product Manual Human Leptin ELISA Kit Catalog Numbers MET-5057 MET-5057-5 96 assays 5 x 96 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Leptin is a polypeptide hormone
Product Manual Human Leptin ELISA Kit Catalog Numbers MET-5057 MET-5057-5 96 assays 5 x 96 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Leptin is a polypeptide hormone
Rat Primary Pre-adipocytes Culture Kit
 Primary Cell Co., Ltd Rat Primary Pre-adipocytes Culture Kit Primary Cells from rat mesenteric, epididymal, and subcutaneous adipose tissues. Catalog # PMC-VAC01-COS, PMC-EAC01-COS, PMC-SAC01-COS Notice
Primary Cell Co., Ltd Rat Primary Pre-adipocytes Culture Kit Primary Cells from rat mesenteric, epididymal, and subcutaneous adipose tissues. Catalog # PMC-VAC01-COS, PMC-EAC01-COS, PMC-SAC01-COS Notice
Islet viability assay and Glucose Stimulated Insulin Secretion assay RT-PCR and Western Blot
 Islet viability assay and Glucose Stimulated Insulin Secretion assay Islet cell viability was determined by colorimetric (3-(4,5-dimethylthiazol-2-yl)-2,5- diphenyltetrazolium bromide assay using CellTiter
Islet viability assay and Glucose Stimulated Insulin Secretion assay Islet cell viability was determined by colorimetric (3-(4,5-dimethylthiazol-2-yl)-2,5- diphenyltetrazolium bromide assay using CellTiter
Implications of mitochondrial skeletal muscle metabolism on diabetes and obesity before and after weight loss
 GG2 Implications of mitochondrial skeletal muscle metabolism on diabetes and obesity before and after weight loss Dr Giacomo Gastaldi CHRU Montpellier Folie 1 GG2 19.10.2009 GG_PC; 12.10.2009 Plan Introduction
GG2 Implications of mitochondrial skeletal muscle metabolism on diabetes and obesity before and after weight loss Dr Giacomo Gastaldi CHRU Montpellier Folie 1 GG2 19.10.2009 GG_PC; 12.10.2009 Plan Introduction
VEGFR2-Mediated Vascular Dilation as a Mechanism of VEGF-Induced Anemia and Bone Marrow Cell Mobilization
 Cell Reports, Volume 9 Supplemental Information VEGFR2-Mediated Vascular Dilation as a Mechanism of VEGF-Induced Anemia and Bone Marrow Cell Mobilization Sharon Lim, Yin Zhang, Danfang Zhang, Fang Chen,
Cell Reports, Volume 9 Supplemental Information VEGFR2-Mediated Vascular Dilation as a Mechanism of VEGF-Induced Anemia and Bone Marrow Cell Mobilization Sharon Lim, Yin Zhang, Danfang Zhang, Fang Chen,
BEIGE AND BROWN FAT: BASIC BIOLOGY AND NOVEL THERAPEUTICS Dr. Carl Ascoli
 BEIGE AND BROWN FAT: BASIC BIOLOGY AND NOVEL THERAPEUTICS Dr. Carl Ascoli Symposium Co-Chairs: Bruce M. Spiegelman (Harvard/Dana Farber) and Sven Enerbäck (U.Gothenburg) April 17-23, 2015 Snowbird Resort,
BEIGE AND BROWN FAT: BASIC BIOLOGY AND NOVEL THERAPEUTICS Dr. Carl Ascoli Symposium Co-Chairs: Bruce M. Spiegelman (Harvard/Dana Farber) and Sven Enerbäck (U.Gothenburg) April 17-23, 2015 Snowbird Resort,
SUPPLEMENTARY INFORMATION
 doi: 1.138/nature7221 Brown fat selective genes 12 1 Control Q-RT-PCR (% of Control) 8 6 4 2 Ntrk3 Cox7a1 Cox8b Cox5b ATPase b2 ATPase f1a1 Sirt3 ERRα Elovl3/Cig3 PPARα Zic1 Supplementary Figure S1. stimulates
doi: 1.138/nature7221 Brown fat selective genes 12 1 Control Q-RT-PCR (% of Control) 8 6 4 2 Ntrk3 Cox7a1 Cox8b Cox5b ATPase b2 ATPase f1a1 Sirt3 ERRα Elovl3/Cig3 PPARα Zic1 Supplementary Figure S1. stimulates
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION FOR Liver X Receptor α mediates hepatic triglyceride accumulation through upregulation of G0/G1 Switch Gene 2 (G0S2) expression I: SUPPLEMENTARY METHODS II: SUPPLEMENTARY FIGURES
SUPPLEMENTARY INFORMATION FOR Liver X Receptor α mediates hepatic triglyceride accumulation through upregulation of G0/G1 Switch Gene 2 (G0S2) expression I: SUPPLEMENTARY METHODS II: SUPPLEMENTARY FIGURES
( ) Downloaded from ijem.sbmu.ac.ir at 20: on Friday March 22nd 2019 VEFG. HIIT. MICT. :.
 ( ) VEFG 2 1 1 ( ) (2 (1 : ( e-mail: Mostafa.rahimi20@gmail.com (MICT) (HIIT) :. (VEGF) ( ± ) :. (HIIT) (MICT) MICT. HIIT.. :. Real-time RT-PCR (P / ) VEGF.(P / ) :. : 96/5/15 : 96/4/20 96//6 : 1 2014
( ) VEFG 2 1 1 ( ) (2 (1 : ( e-mail: Mostafa.rahimi20@gmail.com (MICT) (HIIT) :. (VEGF) ( ± ) :. (HIIT) (MICT) MICT. HIIT.. :. Real-time RT-PCR (P / ) VEGF.(P / ) :. : 96/5/15 : 96/4/20 96//6 : 1 2014
YK052 Mouse Leptin ELISA
 YK052 Mouse Leptin ELISA FOR LABORATORY USE ONLY YANAIHARA INSTITUTE INC. 2480-1 AWAKURA, FUJINOMIYA-SHI SHIZUOKA, JAPAN 418-0011 Contents Ⅰ. Introduction 2 Ⅱ. Characteristics 3 Ⅲ. Composition 4 Ⅳ. Method
YK052 Mouse Leptin ELISA FOR LABORATORY USE ONLY YANAIHARA INSTITUTE INC. 2480-1 AWAKURA, FUJINOMIYA-SHI SHIZUOKA, JAPAN 418-0011 Contents Ⅰ. Introduction 2 Ⅱ. Characteristics 3 Ⅲ. Composition 4 Ⅳ. Method
Supplemental Information Supplementary Table 1. Tph1+/+ Tph1 / Analyte Supplementary Table 2. Tissue Vehicle LP value
 Supplemental Information Supplementary Table. Urinary and adipose tissue catecholamines in Tph +/+ and Tph / mice fed a high fat diet for weeks. Tph +/+ Tph / Analyte ewat ibat ewat ibat Urine (ng/ml)
Supplemental Information Supplementary Table. Urinary and adipose tissue catecholamines in Tph +/+ and Tph / mice fed a high fat diet for weeks. Tph +/+ Tph / Analyte ewat ibat ewat ibat Urine (ng/ml)
Supporting Information
 Supporting Information Pang et al. 10.1073/pnas.1322009111 SI Materials and Methods ELISAs. These assays were performed as previously described (1). ELISA plates (MaxiSorp Nunc; Thermo Fisher Scientific)
Supporting Information Pang et al. 10.1073/pnas.1322009111 SI Materials and Methods ELISAs. These assays were performed as previously described (1). ELISA plates (MaxiSorp Nunc; Thermo Fisher Scientific)
Naohito AOKI, Erina ARAKAWA and Miyuki ITO. Department of Life Science, Graduate School of Bioresources, Mie University Tsu ABSTRACT
 Naohito AOKI, Erina ARAKAWA and Miyuki ITO Department of Life Science, Graduate School of Bioresources, Mie University Tsu 514-857 ABSTRACT C57BL/6J mice (male, 4wk old) were fed low fat diet (LF), high
Naohito AOKI, Erina ARAKAWA and Miyuki ITO Department of Life Science, Graduate School of Bioresources, Mie University Tsu 514-857 ABSTRACT C57BL/6J mice (male, 4wk old) were fed low fat diet (LF), high
Rat Leptin-HS ELISA FOR LABORATORY USE ONLY YANAIHARA INSTITUTE INC AWAKURA, FUJINOMIYA - SHI SHIZUOKA, JAPAN
 YK051 Rat Leptin-HS ELISA FOR LABORATORY USE ONLY YANAIHARA INSTITUTE INC. 2480-1 AWAKURA, FUJINOMIYA - SHI SHIZUOKA, JAPAN 418 0011 Contents Introduction 2 Characteristics 3 Composition 4 Method 5-6 Notes
YK051 Rat Leptin-HS ELISA FOR LABORATORY USE ONLY YANAIHARA INSTITUTE INC. 2480-1 AWAKURA, FUJINOMIYA - SHI SHIZUOKA, JAPAN 418 0011 Contents Introduction 2 Characteristics 3 Composition 4 Method 5-6 Notes
Rat Leptin ELISA FOR LABORATORY USE ONLY YANAIHARA INSTITUTE INC AWAKURA, FUJINOMIYA - SHI SHIZUOKA, JAPAN
 YK050 Rat Leptin ELISA FOR LABORATORY USE ONLY YANAIHARA INSTITUTE INC. 2480-1 AWAKURA, FUJINOMIYA - SHI SHIZUOKA, JAPAN 418-0011 Contents Introduction 2 Characteristics 3 Composition 4 Method 5-6 Notes
YK050 Rat Leptin ELISA FOR LABORATORY USE ONLY YANAIHARA INSTITUTE INC. 2480-1 AWAKURA, FUJINOMIYA - SHI SHIZUOKA, JAPAN 418-0011 Contents Introduction 2 Characteristics 3 Composition 4 Method 5-6 Notes
 This article was published in an Elsevier journal. The attached copy is furnished to the author for non-commercial research and education use, including for instruction at the author s institution, sharing
This article was published in an Elsevier journal. The attached copy is furnished to the author for non-commercial research and education use, including for instruction at the author s institution, sharing
Supplementary Figure 1. DNA methylation of the adiponectin promoter R1, Pparg2, and Tnfa promoter in adipocytes is not affected by obesity.
 Supplementary Figure 1. DNA methylation of the adiponectin promoter R1, Pparg2, and Tnfa promoter in adipocytes is not affected by obesity. (a) Relative amounts of adiponectin, Ppar 2, C/ebp, and Tnf mrna
Supplementary Figure 1. DNA methylation of the adiponectin promoter R1, Pparg2, and Tnfa promoter in adipocytes is not affected by obesity. (a) Relative amounts of adiponectin, Ppar 2, C/ebp, and Tnf mrna
The Epigenetics of Obesity: Individual, Social, and Environmental Influences. K. J. Claycombe, Ph.D.
 The Epigenetics of Obesity: Individual, Social, and Environmental Influences K. J. Claycombe, Ph.D. What can happen to our gene(s) that would cause obesity? Modification via Epigenetic alterations C
The Epigenetics of Obesity: Individual, Social, and Environmental Influences K. J. Claycombe, Ph.D. What can happen to our gene(s) that would cause obesity? Modification via Epigenetic alterations C
Taylor Yohe. Project Advisor: Dr. Martha A. Belury. Department of Human Nutrition at the Ohio State University
 Atherosclerosis Development and the Inflammatory Response of Hepatocytes to Sesame Oil Supplementation Taylor Yohe Project Advisor: Dr. Martha A. Belury Department of Human Nutrition at the Ohio State
Atherosclerosis Development and the Inflammatory Response of Hepatocytes to Sesame Oil Supplementation Taylor Yohe Project Advisor: Dr. Martha A. Belury Department of Human Nutrition at the Ohio State
Metabolic responses to leptin in obese db/db mice are strain dependent
 Am J Physiol Regulatory Integrative Comp Physiol 281: R115 R132, 2001. Metabolic responses to leptin in obese db/db mice are strain dependent RUTH B. S. HARRIS, TIFFANY D. MITCHELL, XIAOLANG YAN, JACOB
Am J Physiol Regulatory Integrative Comp Physiol 281: R115 R132, 2001. Metabolic responses to leptin in obese db/db mice are strain dependent RUTH B. S. HARRIS, TIFFANY D. MITCHELL, XIAOLANG YAN, JACOB
Role of fatty acids in the development of insulin resistance and type 2 diabetes mellitus
 Emerging Science Role of fatty acids in the development of insulin resistance and type 2 diabetes mellitus George Wolf Insulin resistance is defined as the reduced responsiveness to normal circulating
Emerging Science Role of fatty acids in the development of insulin resistance and type 2 diabetes mellitus George Wolf Insulin resistance is defined as the reduced responsiveness to normal circulating
(A) PCR primers (arrows) designed to distinguish wild type (P1+P2), targeted (P1+P2) and excised (P1+P3)14-
 1 Supplemental Figure Legends Figure S1. Mammary tumors of ErbB2 KI mice with 14-3-3σ ablation have elevated ErbB2 transcript levels and cell proliferation (A) PCR primers (arrows) designed to distinguish
1 Supplemental Figure Legends Figure S1. Mammary tumors of ErbB2 KI mice with 14-3-3σ ablation have elevated ErbB2 transcript levels and cell proliferation (A) PCR primers (arrows) designed to distinguish
Effects of growth hormone secretagogue receptor agonist and antagonist in nonobese type 2 diabetic MKR mice
 Effects of growth hormone secretagogue receptor agonist and antagonist in nonobese type 2 diabetic MKR mice Rasha Mosa (MBCHC, M.D, PhD candidate) School of Biomedical Sciences University of Queensland
Effects of growth hormone secretagogue receptor agonist and antagonist in nonobese type 2 diabetic MKR mice Rasha Mosa (MBCHC, M.D, PhD candidate) School of Biomedical Sciences University of Queensland
SUPPLEMENTARY INFORMATION
 Supplementary Figures Supplementary Figure S1. Binding of full-length OGT and deletion mutants to PIP strips (Echelon Biosciences). Supplementary Figure S2. Binding of the OGT (919-1036) fragments with
Supplementary Figures Supplementary Figure S1. Binding of full-length OGT and deletion mutants to PIP strips (Echelon Biosciences). Supplementary Figure S2. Binding of the OGT (919-1036) fragments with
Altered food consumption in mice lacking lysophosphatidic acid receptor-1
 J Physiol Biochem, 65 (4), 000-000, 2009 Altered food consumption in mice lacking lysophosphatidic acid receptor-1 R. Dusaulcy, D. Daviaud, J-P. Pradère, S. Grès, Ph. Valet and J.S. Saulnier-Blache Institut
J Physiol Biochem, 65 (4), 000-000, 2009 Altered food consumption in mice lacking lysophosphatidic acid receptor-1 R. Dusaulcy, D. Daviaud, J-P. Pradère, S. Grès, Ph. Valet and J.S. Saulnier-Blache Institut
Free Fatty Acid Assay Kit (Fluorometric)
 Product Manual Free Fatty Acid Assay Kit (Fluorometric) Catalog Number STA-619 100 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Triglycerides (TAG) are a type of lipid
Product Manual Free Fatty Acid Assay Kit (Fluorometric) Catalog Number STA-619 100 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Triglycerides (TAG) are a type of lipid
SUPPLEMENTARY DATA. Supplementary Table 1. Primers used in qpcr
 Supplementary Table 1. Primers used in qpcr Gene forward primer (5'-3') reverse primer (5'-3') β-actin AGAGGGAAATCGTGCGTGAC CAATAGTGATGACCTGGCCGT Hif-p4h-2 CTGGGCAACTACAGGATAAAC GCGTCCCAGTCTTTATTTAGATA
Supplementary Table 1. Primers used in qpcr Gene forward primer (5'-3') reverse primer (5'-3') β-actin AGAGGGAAATCGTGCGTGAC CAATAGTGATGACCTGGCCGT Hif-p4h-2 CTGGGCAACTACAGGATAAAC GCGTCCCAGTCTTTATTTAGATA
Expression of acid base transporters in the kidney collecting duct in Slc2a7 -/-
 Supplemental Material Results. Expression of acid base transporters in the kidney collecting duct in Slc2a7 -/- and Slc2a7 -/- mice. The expression of AE1 in the kidney was examined in Slc26a7 KO mice.
Supplemental Material Results. Expression of acid base transporters in the kidney collecting duct in Slc2a7 -/- and Slc2a7 -/- mice. The expression of AE1 in the kidney was examined in Slc26a7 KO mice.
control kda ATGL ATGLi HSL 82 GAPDH * ** *** WT/cTg WT/cTg ATGLi AKO/cTg AKO/cTg ATGLi WT/cTg WT/cTg ATGLi AKO/cTg AKO/cTg ATGLi iwat gwat ibat
 body weight (g) tissue weights (mg) ATGL protein expression (relative to GAPDH) HSL protein expression (relative to GAPDH) ### # # kda ATGL 55 HSL 82 GAPDH 37 2.5 2. 1.5 1..5 2. 1.5 1..5.. Supplementary
body weight (g) tissue weights (mg) ATGL protein expression (relative to GAPDH) HSL protein expression (relative to GAPDH) ### # # kda ATGL 55 HSL 82 GAPDH 37 2.5 2. 1.5 1..5 2. 1.5 1..5.. Supplementary
Serum Amyloid A3 Gene Expression in Adipocytes is an Indicator. of the Interaction with Macrophages
 Serum Amyloid A3 Gene Expression in Adipocytes is an Indicator of the Interaction with Macrophages Yohei Sanada, Takafumi Yamamoto, Rika Satake, Akiko Yamashita, Sumire Kanai, Norihisa Kato, Fons AJ van
Serum Amyloid A3 Gene Expression in Adipocytes is an Indicator of the Interaction with Macrophages Yohei Sanada, Takafumi Yamamoto, Rika Satake, Akiko Yamashita, Sumire Kanai, Norihisa Kato, Fons AJ van
IN EARLY STAGE development of adipose tissue, adipogenesis
 0013-7227/05/$15.00/0 Endocrinology 146(10):4545 4554 Printed in U.S.A. Copyright 2005 by The Endocrine Society doi: 10.1210/en.2005-0532 Modulation of Angiogenesis during Adipose Tissue Development in
0013-7227/05/$15.00/0 Endocrinology 146(10):4545 4554 Printed in U.S.A. Copyright 2005 by The Endocrine Society doi: 10.1210/en.2005-0532 Modulation of Angiogenesis during Adipose Tissue Development in
ENHANCEMENT OF BROWN ADIPOSE TISSUE DEVELOPMENT IN VIVO BY A NOVEL INSULIN SENSITIZER
 ENHANCEMENT F BRWN ADIPSE TISSUE DEVELPMENT IN VIV BY A NVEL INSULIN SENSITIZER William G. McDonald 1, Serena L. Cole 1, Brian N. Finck 2, Danielle D. Holewa 1, Angela S. Brightwell-Conrad 1, Charles Mackenzie
ENHANCEMENT F BRWN ADIPSE TISSUE DEVELPMENT IN VIV BY A NVEL INSULIN SENSITIZER William G. McDonald 1, Serena L. Cole 1, Brian N. Finck 2, Danielle D. Holewa 1, Angela S. Brightwell-Conrad 1, Charles Mackenzie
Ruth B. S. Harris, Tiffany D. Mitchell, Xiaolang Yan, Jacob S. Simpson and Stephen M. Redmann, Jr.
 Metabolic responses to leptin in obesedb/db mice are strain dependent Ruth B. S. Harris, Tiffany D. Mitchell, Xiaolang Yan, Jacob S. Simpson and Stephen M. Redmann, Jr. Am J Physiol Regul Integr Comp Physiol
Metabolic responses to leptin in obesedb/db mice are strain dependent Ruth B. S. Harris, Tiffany D. Mitchell, Xiaolang Yan, Jacob S. Simpson and Stephen M. Redmann, Jr. Am J Physiol Regul Integr Comp Physiol
Endothelial PGC 1 - α 1 mediates vascular dysfunction in diabetes
 Endothelial PGC-1α mediates vascular dysfunction in diabetes Reporter: Yaqi Zhou Date: 04/14/2014 Outline I. Introduction II. Research route & Results III. Summary Diabetes the Epidemic of the 21st Century
Endothelial PGC-1α mediates vascular dysfunction in diabetes Reporter: Yaqi Zhou Date: 04/14/2014 Outline I. Introduction II. Research route & Results III. Summary Diabetes the Epidemic of the 21st Century
In The Name Of God. In The Name Of. EMRI Modeling Group
 In The Name Of God In The Name Of God EMRI Modeling Group Cells work together in functionally related groups called tissues Types of tissues: Epithelial lining and covering Connective support Muscle movement
In The Name Of God In The Name Of God EMRI Modeling Group Cells work together in functionally related groups called tissues Types of tissues: Epithelial lining and covering Connective support Muscle movement
I. Introduction. II. Characteristics
 YK050 Rat Leptin ELISA I. Introduction Leptin, which is a product of ob gene, is a protein consisting of 167 amino acids and it is secreted from white adipose tissue. It is known that leptin acts on hypothalamus
YK050 Rat Leptin ELISA I. Introduction Leptin, which is a product of ob gene, is a protein consisting of 167 amino acids and it is secreted from white adipose tissue. It is known that leptin acts on hypothalamus
Supplementary Information Titles Journal: Nature Medicine
 Supplementary Information Titles Journal: Nature Medicine Article Title: Corresponding Author: Supplementary Item & Number Supplementary Fig.1 Fig.2 Fig.3 Fig.4 Fig.5 Fig.6 Fig.7 Fig.8 Fig.9 Fig. Fig.11
Supplementary Information Titles Journal: Nature Medicine Article Title: Corresponding Author: Supplementary Item & Number Supplementary Fig.1 Fig.2 Fig.3 Fig.4 Fig.5 Fig.6 Fig.7 Fig.8 Fig.9 Fig. Fig.11
RNA extraction, RT-PCR and real-time PCR. Total RNA were extracted using
 Supplementary Information Materials and Methods RNA extraction, RT-PCR and real-time PCR. Total RNA were extracted using Trizol reagent (Invitrogen,Carlsbad, CA) according to the manufacturer's instructions.
Supplementary Information Materials and Methods RNA extraction, RT-PCR and real-time PCR. Total RNA were extracted using Trizol reagent (Invitrogen,Carlsbad, CA) according to the manufacturer's instructions.
Mouse C-peptide EIA. Cat. No. YII-YK013-EX FOR LABORATORY USE ONLY
 Mouse C-peptide EIA Cat. No. YII-YK013-EX FOR LABORATORY USE ONLY TOYO 2CHOME, KOTO-KU, TOKYO, 135-0016, JAPAN http://www.cosmobio.co.jp e-mail : export@cosmobio.co.jp Phone : +81-3-5632-9617 FAX : +81-3-5632-9618
Mouse C-peptide EIA Cat. No. YII-YK013-EX FOR LABORATORY USE ONLY TOYO 2CHOME, KOTO-KU, TOKYO, 135-0016, JAPAN http://www.cosmobio.co.jp e-mail : export@cosmobio.co.jp Phone : +81-3-5632-9617 FAX : +81-3-5632-9618
SUPPLEMENTARY INFORMATION
 doi:10.1038/nature12652 Supplementary Figure 1. PRDM16 interacts with endogenous EHMT1 in brown adipocytes. Immunoprecipitation of PRDM16 complex by flag antibody (M2) followed by Western blot analysis
doi:10.1038/nature12652 Supplementary Figure 1. PRDM16 interacts with endogenous EHMT1 in brown adipocytes. Immunoprecipitation of PRDM16 complex by flag antibody (M2) followed by Western blot analysis
A novel role for vitamin D: modulation of expression and function of the local renin angiotensin system in mouse pancreatic islets
 Diabetologia () 5:77 DOI.7/s5--- SHORT COMMUNICATION A novel role for vitamin D: modulation of expression and function of the local renin angiotensin system in mouse pancreatic islets Q. Cheng & Y. C.
Diabetologia () 5:77 DOI.7/s5--- SHORT COMMUNICATION A novel role for vitamin D: modulation of expression and function of the local renin angiotensin system in mouse pancreatic islets Q. Cheng & Y. C.
Metabolic Solutions Development Company, Kalamazoo, USA.
 New Insulin Sensitizers Produce Differentiation of Brown-like Adipose Cells from a Subcutaneous Fat Depot and Increase Secretion of Adiponectin in vitro William G. McDonald, Serena L. Cole, Danielle D.
New Insulin Sensitizers Produce Differentiation of Brown-like Adipose Cells from a Subcutaneous Fat Depot and Increase Secretion of Adiponectin in vitro William G. McDonald, Serena L. Cole, Danielle D.
Supplementary Figure 1. DJ-1 modulates ROS concentration in mouse skeletal muscle.
 Supplementary Figure 1. DJ-1 modulates ROS concentration in mouse skeletal muscle. (a) mrna levels of Dj1 measured by quantitative RT-PCR in soleus, gastrocnemius (Gastroc.) and extensor digitorum longus
Supplementary Figure 1. DJ-1 modulates ROS concentration in mouse skeletal muscle. (a) mrna levels of Dj1 measured by quantitative RT-PCR in soleus, gastrocnemius (Gastroc.) and extensor digitorum longus
Supplemental Data. Wnt/β-Catenin Signaling in Mesenchymal Progenitors. Controls Osteoblast and Chondrocyte
 Supplemental Data Wnt/β-Catenin Signaling in Mesenchymal Progenitors Controls Osteoblast and Chondrocyte Differentiation during Vertebrate Skeletogenesis Timothy F. Day, Xizhi Guo, Lisa Garrett-Beal, and
Supplemental Data Wnt/β-Catenin Signaling in Mesenchymal Progenitors Controls Osteoblast and Chondrocyte Differentiation during Vertebrate Skeletogenesis Timothy F. Day, Xizhi Guo, Lisa Garrett-Beal, and
Supplemental Information. Increased 4E-BP1 Expression Protects. against Diet-Induced Obesity and Insulin. Resistance in Male Mice
 Cell Reports, Volume 16 Supplemental Information Increased 4E-BP1 Expression Protects against Diet-Induced Obesity and Insulin Resistance in Male Mice Shih-Yin Tsai, Ariana A. Rodriguez, Somasish G. Dastidar,
Cell Reports, Volume 16 Supplemental Information Increased 4E-BP1 Expression Protects against Diet-Induced Obesity and Insulin Resistance in Male Mice Shih-Yin Tsai, Ariana A. Rodriguez, Somasish G. Dastidar,
AMPK Phosphorylation Assay Kit
 AMPK Phosphorylation Assay Kit Catalog Number KA3789 100 assays Version: 02 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
AMPK Phosphorylation Assay Kit Catalog Number KA3789 100 assays Version: 02 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
Supporting Information
 Supporting Information Charalambous et al. 10.1073/pnas.1406119111 SI Experimental Procedures Serum and Tissue Biochemistry. Enzymatic assay kits were used for determination of plasma FFAs (Roche), TAGs
Supporting Information Charalambous et al. 10.1073/pnas.1406119111 SI Experimental Procedures Serum and Tissue Biochemistry. Enzymatic assay kits were used for determination of plasma FFAs (Roche), TAGs
Cell Culture. The human thyroid follicular carcinoma cell lines FTC-238, FTC-236 and FTC-
 Supplemental material and methods Reagents. Hydralazine was purchased from Sigma-Aldrich. Cell Culture. The human thyroid follicular carcinoma cell lines FTC-238, FTC-236 and FTC- 133, human thyroid medullary
Supplemental material and methods Reagents. Hydralazine was purchased from Sigma-Aldrich. Cell Culture. The human thyroid follicular carcinoma cell lines FTC-238, FTC-236 and FTC- 133, human thyroid medullary
Mouse Leptin ELISA Kit (mleptin-elisa)
 Mouse Leptin ELISA Kit (mleptin-elisa) Cat. No. EK0438 96 Tests in 8 x 12 divisible strips Background Leptin (or obese, OB) is a circulating hormone that is expressed abundantly and specifically in the
Mouse Leptin ELISA Kit (mleptin-elisa) Cat. No. EK0438 96 Tests in 8 x 12 divisible strips Background Leptin (or obese, OB) is a circulating hormone that is expressed abundantly and specifically in the
Offspring from mothers fed a junk food diet in pregnancy and lactation exhibit exacerbated adiposity that is more pronounced in females
 J Physiol 586.13 (2008) pp 3219 3230 3219 Offspring from mothers fed a junk food diet in pregnancy and lactation exhibit exacerbated adiposity that is more pronounced in females S. A. Bayol, B. H. Simbi,
J Physiol 586.13 (2008) pp 3219 3230 3219 Offspring from mothers fed a junk food diet in pregnancy and lactation exhibit exacerbated adiposity that is more pronounced in females S. A. Bayol, B. H. Simbi,
Mouse Adiponectin Immunoassay Kit
 Antibody and Immunoassay Services Li Ka Shing Faculty of Medicine, The University of Hong Kong Mouse Adiponectin Immunoassay Kit Catalogue Number: 32010 For the quantitative determination of mouse adiponectin
Antibody and Immunoassay Services Li Ka Shing Faculty of Medicine, The University of Hong Kong Mouse Adiponectin Immunoassay Kit Catalogue Number: 32010 For the quantitative determination of mouse adiponectin
Adipose Tissue Dysfunction and Diabetic Cardiovascular Disease
 Adipose Tissue Dysfunction and Diabetic Cardiovascular Disease Xin-Liang Ma, MD, PhD Thomas Jefferson University Philadelphia, PA USA CVD As The #1 Causes of Death In USA Male Female Heart Disease and
Adipose Tissue Dysfunction and Diabetic Cardiovascular Disease Xin-Liang Ma, MD, PhD Thomas Jefferson University Philadelphia, PA USA CVD As The #1 Causes of Death In USA Male Female Heart Disease and
ab65336 Triglyceride Quantification Assay Kit (Colorimetric/ Fluorometric)
 Version 10 Last updated 19 December 2017 ab65336 Triglyceride Quantification Assay Kit (Colorimetric/ Fluorometric) For the measurement of triglycerides in various samples. This product is for research
Version 10 Last updated 19 December 2017 ab65336 Triglyceride Quantification Assay Kit (Colorimetric/ Fluorometric) For the measurement of triglycerides in various samples. This product is for research
HRP cytochemistry. Division of Radiooncology, Deutsches Krebsforschungszentrum, Heidelberg, Germany
 HRP cytochemistry WOLF D. KUHLMANN, M.D. Division of Radiooncology, Deutsches Krebsforschungszentrum, 69120 Heidelberg, Germany A range of substrates is available for the cytochemical staining of peroxidase
HRP cytochemistry WOLF D. KUHLMANN, M.D. Division of Radiooncology, Deutsches Krebsforschungszentrum, 69120 Heidelberg, Germany A range of substrates is available for the cytochemical staining of peroxidase
The Function of the Nuclear Receptor Peroxisome Proliferator activated Receptor Delta in Energy Homeostasis
 22. Castellot JJ Jr, Karnovsky MJ, Spiegelman BM. Potent stimulation of vascular endothelial cell growth by differentiated 3T3 adipocytes. Proc Natl Acad Sci U S A.1980;77:6007 6011. 23. Castellot JJ Jr,
22. Castellot JJ Jr, Karnovsky MJ, Spiegelman BM. Potent stimulation of vascular endothelial cell growth by differentiated 3T3 adipocytes. Proc Natl Acad Sci U S A.1980;77:6007 6011. 23. Castellot JJ Jr,
Adipose Tissue as an Endocrine Organ. Abdel Moniem Ibrahim, MD Professor of Physiology Cairo University
 Adipose Tissue as an Endocrine Organ Abdel Moniem Ibrahim, MD Professor of Physiology Cairo University Functions of Adipose Tissue Adipose tissue expresses and secretes a variety of bioactive peptides,
Adipose Tissue as an Endocrine Organ Abdel Moniem Ibrahim, MD Professor of Physiology Cairo University Functions of Adipose Tissue Adipose tissue expresses and secretes a variety of bioactive peptides,
Supplementary Materials for
 www.sciencesignaling.org/cgi/content/full/8/407/ra127/dc1 Supplementary Materials for Loss of FTO in adipose tissue decreases Angptl4 translation and alters triglyceride metabolism Chao-Yung Wang,* Shian-Sen
www.sciencesignaling.org/cgi/content/full/8/407/ra127/dc1 Supplementary Materials for Loss of FTO in adipose tissue decreases Angptl4 translation and alters triglyceride metabolism Chao-Yung Wang,* Shian-Sen
Effec<ve Use of PI3K and MEK Inhibitors to Treat Mutant K Ras G12D and PIK3CA H1047R Murine Lung Cancers
 Effec
Effec
Supplementary Table 1. Primer sequences for conventional RT-PCR on mouse islets
 Supplementary Table 1. Primer sequences for conventional RT-PCR on mouse islets Gene 5 Forward 3 5 Reverse 3.T. Product (bp) ( C) mnox1 GTTCTTGGGCTGCCTTGG GCTGGGGCGGCGG 60 300 mnoxa1 GCTTTGCCGCGTGC GGTTCGGGTCCTTTGTGC
Supplementary Table 1. Primer sequences for conventional RT-PCR on mouse islets Gene 5 Forward 3 5 Reverse 3.T. Product (bp) ( C) mnox1 GTTCTTGGGCTGCCTTGG GCTGGGGCGGCGG 60 300 mnoxa1 GCTTTGCCGCGTGC GGTTCGGGTCCTTTGTGC
Lecithin Cholesterol Acyltransferase (LCAT) ELISA Kit
 Product Manual Lecithin Cholesterol Acyltransferase (LCAT) ELISA Kit Catalog Number STA-616 96 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Cholesterol is a lipid sterol
Product Manual Lecithin Cholesterol Acyltransferase (LCAT) ELISA Kit Catalog Number STA-616 96 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Cholesterol is a lipid sterol
Data Sheet. PCSK9[Biotinylated]-LDLR Binding Assay Kit Catalog # 72002
![Data Sheet. PCSK9[Biotinylated]-LDLR Binding Assay Kit Catalog # 72002 Data Sheet. PCSK9[Biotinylated]-LDLR Binding Assay Kit Catalog # 72002](/thumbs/79/80181612.jpg) Data Sheet PCSK9[Biotinylated]-LDLR Binding Assay Kit Catalog # 72002 DESCRIPTION: The PCSK9[Biotinylated]-LDLR Binding Assay Kit is designed for screening and profiling purposes. PCSK9 is known to function
Data Sheet PCSK9[Biotinylated]-LDLR Binding Assay Kit Catalog # 72002 DESCRIPTION: The PCSK9[Biotinylated]-LDLR Binding Assay Kit is designed for screening and profiling purposes. PCSK9 is known to function
Free Fatty Acid Assay Kit
 Free Fatty Acid Assay Kit Catalog Number KA1667 100 assays Version: 05 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 General Information...
Free Fatty Acid Assay Kit Catalog Number KA1667 100 assays Version: 05 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 General Information...
Human Obestatin ELISA
 K-ASSAY Human Obestatin ELISA For the quantitative determination of obestatin in human serum and plasma Cat. No. KT-495 For Research Use Only. 1 Rev. 081309 K-ASSAY PRODUCT INFORMATION Human Obestatin
K-ASSAY Human Obestatin ELISA For the quantitative determination of obestatin in human serum and plasma Cat. No. KT-495 For Research Use Only. 1 Rev. 081309 K-ASSAY PRODUCT INFORMATION Human Obestatin
Introduction. Leptin secretion after a high-fat meal in normal-weight rats: strong predictor of long-term body fat accrual on a high-fat diet
 Leptin secretion after a high-fat meal in normal-weight rats: strong predictor of long-term body fat accrual on a high-fat diet - Diet - Activity - Genetics etc. Introduction Obesity - Cardiovascular disease
Leptin secretion after a high-fat meal in normal-weight rats: strong predictor of long-term body fat accrual on a high-fat diet - Diet - Activity - Genetics etc. Introduction Obesity - Cardiovascular disease
Defective Hepatic Autophagy in Obesity Promotes ER Stress and Causes Insulin Resistance
 Cell Metabolism, Volume 11 Supplemental Information Defective Hepatic Autophagy in Obesity Promotes ER Stress and Causes Insulin Resistance Ling Yang, Ping Li, Suneng Fu, Ediz S. Calay, and Gökhan S. Hotamisligil
Cell Metabolism, Volume 11 Supplemental Information Defective Hepatic Autophagy in Obesity Promotes ER Stress and Causes Insulin Resistance Ling Yang, Ping Li, Suneng Fu, Ediz S. Calay, and Gökhan S. Hotamisligil
For in vitro Veterinary Diagnostics only. Kylt Rotavirus A. Real-Time RT-PCR Detection.
 For in vitro Veterinary Diagnostics only. Kylt Rotavirus A Real-Time RT-PCR Detection www.kylt.eu DIRECTION FOR USE Kylt Rotavirus A Real-Time RT-PCR Detection A. General Kylt Rotavirus A products are
For in vitro Veterinary Diagnostics only. Kylt Rotavirus A Real-Time RT-PCR Detection www.kylt.eu DIRECTION FOR USE Kylt Rotavirus A Real-Time RT-PCR Detection A. General Kylt Rotavirus A products are
Biodegradable Zwitterionic Nanogels with Long. Circulation for Antitumor Drug Delivery
 Supporting Information Biodegradable Zwitterionic Nanogels with Long Circulation for Antitumor Drug Delivery Yongzhi Men, Shaojun Peng, Peng Yang, Qin Jiang, Yanhui Zhang, Bin Shen, Pin Dong, *, Zhiqing
Supporting Information Biodegradable Zwitterionic Nanogels with Long Circulation for Antitumor Drug Delivery Yongzhi Men, Shaojun Peng, Peng Yang, Qin Jiang, Yanhui Zhang, Bin Shen, Pin Dong, *, Zhiqing
BMP6 treatment compensates for the molecular defect and ameliorates hemochromatosis in Hfe knockout mice
 SUPPLEMENTARY MATERIALS BMP6 treatment compensates for the molecular defect and ameliorates hemochromatosis in Hfe knockout mice Elena Corradini, Paul J. Schmidt, Delphine Meynard, Cinzia Garuti, Giuliana
SUPPLEMENTARY MATERIALS BMP6 treatment compensates for the molecular defect and ameliorates hemochromatosis in Hfe knockout mice Elena Corradini, Paul J. Schmidt, Delphine Meynard, Cinzia Garuti, Giuliana
Free Glycerol Assay Kit (Colorimetric)
 Product Manual Free Glycerol Assay Kit (Colorimetric) Catalog Number STA-398 100 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Glycerol is the backbone of Triglycerides
Product Manual Free Glycerol Assay Kit (Colorimetric) Catalog Number STA-398 100 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Glycerol is the backbone of Triglycerides
Supplemental Information. Intermittent Fasting Promotes. White Adipose Browning and Decreases Obesity. by Shaping the Gut Microbiota
 Cell Metabolism, Volume 26 Supplemental Information Intermittent Fasting Promotes White Adipose Browning and Decreases Obesity by Shaping the Gut Microbiota Guolin Li, Cen Xie, Siyu Lu, Robert G. Nichols,
Cell Metabolism, Volume 26 Supplemental Information Intermittent Fasting Promotes White Adipose Browning and Decreases Obesity by Shaping the Gut Microbiota Guolin Li, Cen Xie, Siyu Lu, Robert G. Nichols,
SUPPLEMENTAL MATERIAL. Supplementary Methods
 SUPPLEMENTAL MATERIAL Supplementary Methods Culture of cardiomyocytes, fibroblasts and cardiac microvascular endothelial cells The isolation and culturing of neonatal rat ventricular cardiomyocytes was
SUPPLEMENTAL MATERIAL Supplementary Methods Culture of cardiomyocytes, fibroblasts and cardiac microvascular endothelial cells The isolation and culturing of neonatal rat ventricular cardiomyocytes was
Supporting Information
 Supporting Information Franco et al. 10.1073/pnas.1015557108 SI Materials and Methods Drug Administration. PD352901 was dissolved in 0.5% (wt/vol) hydroxyl-propyl-methylcellulose, 0.2% (vol/vol) Tween
Supporting Information Franco et al. 10.1073/pnas.1015557108 SI Materials and Methods Drug Administration. PD352901 was dissolved in 0.5% (wt/vol) hydroxyl-propyl-methylcellulose, 0.2% (vol/vol) Tween
Requires Signaling though Akt2 Independent of the. Transcription Factors FoxA2, FoxO1, and SREBP1c
 Cell Metabolism, Volume 14 Supplemental Information Postprandial Hepatic Lipid Metabolism Requires Signaling though Akt2 Independent of the Transcription Factors FoxA2, FoxO1, and SREBP1c Min Wan, Karla
Cell Metabolism, Volume 14 Supplemental Information Postprandial Hepatic Lipid Metabolism Requires Signaling though Akt2 Independent of the Transcription Factors FoxA2, FoxO1, and SREBP1c Min Wan, Karla
SHORT-TERM AND LONG-TERM EFFECT OF OXYTOCIN ON THE MORPHOLOGY AND FUNCTIONAL ACTIVITY OF FEMALE RAT SUBCUTANEOUS ADIPOSE TISSUE. P.
 PROCEEDINGS OF THE BALKAN SCIENTIFIC CONFERENCE OF BIOLOGY IN PLOVDIV (BULGARIA) FROM 19 TH TILL 21 ST OF MAY 2005 (EDS B. GRUEV, M. NIKOLOVA AND A. DONEV), 2005 (P. 739 743) SHORT-TERM AND LONG-TERM EFFECT
PROCEEDINGS OF THE BALKAN SCIENTIFIC CONFERENCE OF BIOLOGY IN PLOVDIV (BULGARIA) FROM 19 TH TILL 21 ST OF MAY 2005 (EDS B. GRUEV, M. NIKOLOVA AND A. DONEV), 2005 (P. 739 743) SHORT-TERM AND LONG-TERM EFFECT
Obesity in aging: Hormonal contribution
 Obesity in aging: Hormonal contribution Hormonal issues in obesity and aging Hormonal role in regulation of energy balance Genetic component in hormonal regulation Life style contribution to hormonal changes
Obesity in aging: Hormonal contribution Hormonal issues in obesity and aging Hormonal role in regulation of energy balance Genetic component in hormonal regulation Life style contribution to hormonal changes
SUPPLEMENTARY INFORMATION
 DOI: 10.1038/ncb2211 a! mir-143! b! mir-103/107! let-7a! mir-144! mir-122a! mir-126-3p! mir-194! mir-27a! mir-30c! Figure S1 Northern blot analysis of mir-143 expression dependent on feeding conditions.
DOI: 10.1038/ncb2211 a! mir-143! b! mir-103/107! let-7a! mir-144! mir-122a! mir-126-3p! mir-194! mir-27a! mir-30c! Figure S1 Northern blot analysis of mir-143 expression dependent on feeding conditions.
Nicolucci C. (1), Rossi S. (2), Catapane M. (1), Introduction:
 Bisphenol A and Nicolucci C. (1), Rossi S. (2), Catapane M. (1), (1) Dept. Experimental Medicine, Second University of (2) Institute of Genetic and Biophysics, CNR, Naples (3) Dept. of Pediatrics 'F. Fede',
Bisphenol A and Nicolucci C. (1), Rossi S. (2), Catapane M. (1), (1) Dept. Experimental Medicine, Second University of (2) Institute of Genetic and Biophysics, CNR, Naples (3) Dept. of Pediatrics 'F. Fede',
TECHNICAL BULLETIN. Ghrelin EIA Kit for serum, plasma, culture supernatant, and cell lysates. Catalog Number RAB0207 Storage Temperature 20 C
 Ghrelin EIA Kit for serum, plasma, culture supernatant, and cell lysates Catalog Number RAB0207 Storage Temperature 20 C TECHNICAL BULLETIN Product Description The Ghrelin EIA (Enzyme Immunoassay) Kit
Ghrelin EIA Kit for serum, plasma, culture supernatant, and cell lysates Catalog Number RAB0207 Storage Temperature 20 C TECHNICAL BULLETIN Product Description The Ghrelin EIA (Enzyme Immunoassay) Kit
ab Adipogenesis Assay Kit (Cell-Based)
 ab133102 Adipogenesis Assay Kit (Cell-Based) Instructions for Use For the study of induction and inhibition of adipogenesis in adherent cells. This product is for research use only and is not intended
ab133102 Adipogenesis Assay Kit (Cell-Based) Instructions for Use For the study of induction and inhibition of adipogenesis in adherent cells. This product is for research use only and is not intended
exposed operative area for presence of small islands (±1-2 cm) of adipose tissue from
 Supplemental Material Details for the biopsy procedure During surgery, both the surgeon (N.B.) and the researcher (G.V.) visually inspected the exposed operative area for presence of small islands (±1-2
Supplemental Material Details for the biopsy procedure During surgery, both the surgeon (N.B.) and the researcher (G.V.) visually inspected the exposed operative area for presence of small islands (±1-2
Procine sphingomyelin ELISA Kit
 Procine sphingomyelin ELISA Kit For the quantitative in vitro determination of Procine sphingomyelin concentrations in serum - plasma - celiac fluid - tissue homogenate - body fluid FOR LABORATORY RESEARCH
Procine sphingomyelin ELISA Kit For the quantitative in vitro determination of Procine sphingomyelin concentrations in serum - plasma - celiac fluid - tissue homogenate - body fluid FOR LABORATORY RESEARCH
SUPPLEMENTARY MATERIAL. Sample preparation for light microscopy
 SUPPLEMENTARY MATERIAL Sample preparation for light microscopy To characterize the granulocytes and melanomacrophage centers, cross sections were prepared for light microscopy, as described in Material
SUPPLEMENTARY MATERIAL Sample preparation for light microscopy To characterize the granulocytes and melanomacrophage centers, cross sections were prepared for light microscopy, as described in Material
Lipoprotein Lipase Activity Assay Kit (Fluorometric)
 Lipoprotein Lipase Activity Assay Kit (Fluorometric) Catalog Number KA4538 100 assays Version: 02 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Background... 3 General
Lipoprotein Lipase Activity Assay Kit (Fluorometric) Catalog Number KA4538 100 assays Version: 02 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Background... 3 General
Crosstalk between Adiponectin and IGF-IR in breast cancer. Prof. Young Jin Suh Department of Surgery The Catholic University of Korea
 Crosstalk between Adiponectin and IGF-IR in breast cancer Prof. Young Jin Suh Department of Surgery The Catholic University of Korea Obesity Chronic, multifactorial disorder Hypertrophy and hyperplasia
Crosstalk between Adiponectin and IGF-IR in breast cancer Prof. Young Jin Suh Department of Surgery The Catholic University of Korea Obesity Chronic, multifactorial disorder Hypertrophy and hyperplasia
Control 7 d cold 7 d CL
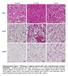 Control 7 d cold 7 d ibat iwat gwat Supplementary Figure 1. Histology of adipose tissues after cold or 3-adrenergic receptor stimulation. C57BL/6J wild-type mice were housed at 4 C or injected daily with
Control 7 d cold 7 d ibat iwat gwat Supplementary Figure 1. Histology of adipose tissues after cold or 3-adrenergic receptor stimulation. C57BL/6J wild-type mice were housed at 4 C or injected daily with
PROGRESS TOWARDS OBJECTIVES, SIGNIFICANT RESULTS, DEVIATIONS, REASONS FOR FAILING, CORRECTIVE ACTIONS
 FINAL REPORT Grant Agreement PIEF-GA-2011-303197 Obesity and Light PROGRESS TOWARDS OBJECTIVES, SIGNIFICANT RESULTS, DEVIATIONS, REASONS FOR FAILING, CORRECTIVE ACTIONS For the total period (2 years) three
FINAL REPORT Grant Agreement PIEF-GA-2011-303197 Obesity and Light PROGRESS TOWARDS OBJECTIVES, SIGNIFICANT RESULTS, DEVIATIONS, REASONS FOR FAILING, CORRECTIVE ACTIONS For the total period (2 years) three
Human Immunodeficiency Virus type 1 (HIV-1) p24 / Capsid Protein p24 ELISA Pair Set
 Human Immunodeficiency Virus type 1 (HIV-1) p24 / Capsid Protein p24 ELISA Pair Set Catalog Number : SEK11695 To achieve the best assay results, this manual must be read carefully before using this product
Human Immunodeficiency Virus type 1 (HIV-1) p24 / Capsid Protein p24 ELISA Pair Set Catalog Number : SEK11695 To achieve the best assay results, this manual must be read carefully before using this product
Expanded View Figures
 Expanded View Figures A B C D E F G H I J K L Figure EV1. The dysregulated lipid metabolic phenotype of mouse models of metabolic dysfunction is most pronounced in the fasted state. A L Male 12-weeks-old
Expanded View Figures A B C D E F G H I J K L Figure EV1. The dysregulated lipid metabolic phenotype of mouse models of metabolic dysfunction is most pronounced in the fasted state. A L Male 12-weeks-old
GLP-2 (Rat) ELISA. For the quantitative determination of glucagon-like peptide 2 (GLP-2) in rat serum and plasma
 GLP-2 (Rat) ELISA For the quantitative determination of glucagon-like peptide 2 (GLP-2) in rat serum and plasma For Research Use Only. Not For Use In Diagnostic Procedures. Catalog Number: 48-GP2RT-E01
GLP-2 (Rat) ELISA For the quantitative determination of glucagon-like peptide 2 (GLP-2) in rat serum and plasma For Research Use Only. Not For Use In Diagnostic Procedures. Catalog Number: 48-GP2RT-E01
SensoLyte pnpp Alkaline Phosphatase Assay Kit *Colorimetric*
 SensoLyte pnpp Alkaline Phosphatase Assay Kit *Colorimetric* Catalog # 72146 Kit Size 500 Assays (96-well plate) Optimized Performance: This kit is optimized to detect alkaline phosphatase activity Enhanced
SensoLyte pnpp Alkaline Phosphatase Assay Kit *Colorimetric* Catalog # 72146 Kit Size 500 Assays (96-well plate) Optimized Performance: This kit is optimized to detect alkaline phosphatase activity Enhanced
Human Apolipoprotein A1 EIA Kit
 A helping hand for your research Product Manual Human Apolipoprotein A1 EIA Kit Catalog Number: 83901 96 assays 1 Table of Content Product Description 3 Assay Principle 3 Kit Components 3 Storage 4 Reagent
A helping hand for your research Product Manual Human Apolipoprotein A1 EIA Kit Catalog Number: 83901 96 assays 1 Table of Content Product Description 3 Assay Principle 3 Kit Components 3 Storage 4 Reagent
IL METABOLISMO EPATICO DEI CARBOIDRATI IN FISIOLOGIA E PATOLOGIA
 UNIGASTRO Il fegato come centrale metabolica e i fattori di danno oltre ai virus epatitici IL METABOLISMO EPATICO DEI CARBOIDRATI IN FISIOLOGIA E PATOLOGIA Dr Elisabetta Bugianesi Divisione di Gastro-Epatologia
UNIGASTRO Il fegato come centrale metabolica e i fattori di danno oltre ai virus epatitici IL METABOLISMO EPATICO DEI CARBOIDRATI IN FISIOLOGIA E PATOLOGIA Dr Elisabetta Bugianesi Divisione di Gastro-Epatologia
