Infant of the Diabetic Mother
|
|
|
- Dale Valentine Short
- 5 years ago
- Views:
Transcription
1 Infant of the Diabetic Mother Tanya Hatfield, MSN, RNC-NIC Neonatal Outreach Educator Objectives Discuss the effects of maternal diabetes on the fetus Identify potential neonatal complications from maternal hyperglycemia Review morbidities associated with IDM Describe glucose metabolism in the newborn and current management in the hypoglycemic infant 1
2 Definitions GDM: carbohydrate intolerance of variable severity with onset or first recognition during pregnancy IDM: Any offspring of a gestational or insulin dependent diabetic woman Classification of diabetes in pregnancy Pregestational: Type 1 Diabetes Mellitus Type 2 Diabetes Mellitus Gestational: GDM A1 Impaired glucose tolerance: diet controlled GDM A2 medication tx 2
3 IDM: The Numbers Infants of Diabetic Mothers Are Twice as likely to suffer serious birth injury 3 times as likely to be born by cesarean section 4 times as likely to be admitted to a neonatal intensive care unit 5 times more likely to be stillborn GDM Incidence by Race/Ethnicity in CA 2005 California 5.88 GDM per 100 deliveries Asian/Pacific Islander 8.73% Hispanic* 8.06% White 4.67% African American 5.43% * Hispanic refers to all of Hispanic ethnicity regardless of race (Bardenheier et al., 2012) 3
4 Morbidities in the IDM: A to Z Asphyxia Birth injury Cardiomyopathy Caudal regression Congenital anomalies Double outlet right ventricle Heart failure Hyperbilirubinemia Morbidities in the IDM: A to Z Hypocalcemia Hypoglycemia Hypomagnesemia Increased blood volume Macrosomia Neurologic issues Organomegaly Polycythemia and hyperviscosity 4
5 Morbidities in the IDM: A to Z Renal vein thrombosis Respiratory distress RDS Septal hypertrophy Shoulder dystocia Small left colon syndrome Transient hematuria Truncus arteriosus Teratogenesis Process by which congenital malformations are produced in an embryo or fetus Congenital malformations account for approx. 50% of perinatal deaths in IDMs Mechanism not well understood Abnormal metabolic environment is teratogenic 5
6 Teratogenesis 1 Hyperglycemic state may damage mitochondria, increase free radicals and oxidative stress in cells Regulating genes altered Alters cellular mitosis Exaggerated apoptosis Leads to spontaneous abortions and malformations 6
7 Slide 11 1 Figure this out Tanya Hatfield, 7/20/2018
8 Diabetic Embryopathy Caused by maternal hyperglycemia in the 1 st trimester Primarily in pregnancies with pregestational diabetes Results in major birth defects and spontaneous abortions Risk of major malformations is 5 to 6%, with a higher prevalence rate of 10 to 12% when mothers require insulin therapy Diabetic Fetopathy Occurs in 2 nd and 3 rd trimesters Results in fetal hyperglycemia, hyperinsulinemia, and macrosomia 7
9 Maternal Hyperglycemia Maternal hyperglycemia results in fetal hyperglycemia glucose readily crosses through the placenta Insulin does not Before 20 weeks gestation the fetal islet cells are not capable of responsive insulin secretion After 20 weeks gestation the fetus has a functioning pancreas and is responsible for its own glucose homeostasis Fetal Response to Maternal Hyperglycemia 2 Fetal hyperglycemia results in fetal hyperinsulinemia fetal hyperinsulinism related to suppressed surfactant production Islet cell hyperplasia Increased growth Excess accumulation of glycogen in liver Inadequate utilization of fatty acids in adipose tissue 8
10 Slide 16 2 figure it out Tanya Hatfield, 6/25/2018
11 Fetal Response to Maternal Hyperglycemia Increases metabolic rate and O2 consumption leading to hypoxemia Fetal hypoxemia contributes to increased mortality, metabolic acidosis, alteration in fetal iron distribution and increased erythropoiesis Increased production of erythropoietin leads to polycythemia promotes catecholamine production, which can results in hypertension and cardiac hypertrophy 9
12 Effects of Maternal Hyperglycemia on the Fetus Barnes -Powell, 2007 Barnes -Powell, 2007 Fetal Hyperinsulinemic State 10
13 Uncontrolled hyperglycemic mother Fetal hyperglycemia In first trimester leads to congenital anomalies Fetal hyperinsulinemia Anabolic effect glucogenesis lipogenesis protein synthesis Neonatal hypoglycemia Hypocalcemia Hypomagnesemia Increased metabolic rate hypoxemia Increased erythropoeisis Anticortisol effect Suppress alveoli Type II enlarged viscera macrosomia Asymptomatic, jittery, seizures, lethargy, apnea Polycythemia, increased viscosity Hyperbilirubinemia Vascular sludging Decrease surfactant synthesis Increase risk of RDS obstructed labor and birth trauma Stroke, jaundice, PPHN Barnes -Powell,
14 Potential neonatal complications 1. Growth/Metabolic Imprinting 2. Glucose Metabolism/Hypoglycemia 3. Electrolyte imbalances 4. Respiratory Issues 5. Cardiovascular Issues 6. Hematologic Changes 7. Birth Trauma 8. Neurologic Issues 9. GI/GU Issues 1. Growth Poor maternal glycemic control predicts neonatal macrosomia > 95%-tile considered abnormal IDM generally have increased fat mass so will have higher weight than length and head circumference percentiles 12
15 Growth-Macrosomia Infants with BW 4000gms or >90%tile, with excess fat accumulation in abdominal and scapular regions, along with visceromegaly Risk increases when: Mean maternal glucose chronically exceeds 130 mg/dl When rises in maternal glucose levels are episodically elevated with surges increasing fetal insulin production Growth-Macrosomia Occurs in >25% of diabetic pregnancies and puts babies at risk for birth injuries Disproportionate growth: higher chest-to-head and shoulder-tohead ratios More likely to have hyperbilirubinemia, hypoglycemia, acidosis, respiratory distress, shoulder dystocia, brachial plexus injury 13
16 Impaired Fetal Growth SGA BW <10 th percentile May occur in up to 20% of diabetic pregnancies Associated with pregnant mothers with renal, retinal or cardiac diseases More likely to deliver SGA, premature, poor fetal outcome, fetal distress, or fetal death Growth-Metabolic Imprinting Macrosomic IDMs have tendency to regain adiposity in late childhood/adolescence National Collaborative Perinatal Project ( ) compared GDM to non GDM offspring 61% higher chance of being overweight at age 7 than non GDM Adjusted for maternal BMI, income, race and BW 14
17 Growth-Metabolic Imprinting Pima Indian Studies Compared siblings exposed to diabetes in utero with those born before mom diagnosed Exposed siblings had higher BMI No difference in BMI after father s diagnosis of Type 2 Data suggests exposure to GDM influences growth and body size independent of genetic predisposition (Bergeon & Dabelea 2009) Growth-Metabolic Imprinting Pima Indian Studies 45% of offspring of mothers with GDM develop Type 2 diabetes btwn 20 and 24 years of age (1988) In follow-up study more than 2/3 of offspring of mothers with GDM developed Type 2 diabetes by 34 years of age (2000) Thought to stem from exposure to hyperglycemia and increased insulin secretion leading to abnormal glucose uptake and utilization in peripheral tissue, especially muscle 15
18 2. Glucose Metabolism The fetus glucose concentration is proportional to maternal concentrations and measures about 70-80% of maternal levels Almost all of this comes from facilitated diffusion across the placenta Glycogen stored in liver, heart, lung and skeletal muscle Glycogen content slowly increases during first two trimesters Majority made and stored in the liver during the third trimester Glucose Metabolism IDM infant undergoes sudden interruption of glucose delivery, which when accompanied by high neonatal insulin levels results in neonatal hypoglycemia Up to 50% of IDMs experience significant hypoglycemia after birth Nadir in blood glucose usually occurs between 1 and 3 hours of life Hyperinsulinemia may persists up to 72 hours and may last up to 1 week 16
19 Insulin and the Fetus Major stimulus for fetal growth Present in the fetal pancreas by 8-10 weeks of gestation Increased production approaching term Insulin inhibits fasting metabolic systems at birth therefore infant can not mobilize glycogen stores 3. Electrolyte Imbalances: Hypocalcemia Serum Ca <7 mg/dl or ionized Ca < 1mmol/L Occurs in up to 30% of IDMs Usually occurs in first hours of life Caused by poor late-trimester transfer of calcium across the placenta, delay in normal postnatal parathyroid hormone elevation, and poor fetal and neonatal bone mineralization (ie, poor calcium stores). Not routinely screened unless symptomatic S/Sx: asymptomatic and self-resolves or jitteriness, tachypnea, seizures/tetany, lethargy and apnea 17
20 Electrolyte Imbalances: Hypomagnesemia Serum magnesium <1.5 mg/dl Occurs in up to 40% IDMs within first 72h of life Most likely due to maternal hypomagnesemia caused by increased urinary losses Usually not treated, but may need treatment if infant is hypocalcemic as well S/Sx: usually transient and asymptomatic, sweating, tachypnea, irritability, seizures, jitteriness 4.Respiratory Issues: RDS Respiratory Distress Syndrome six times more frequent in IDMs until 38 weeks* RDS more common for moms with unstable Type I Possible causes: premature delivery maternal hyperglycemia delays surfactant synthesis Fetal hyperinsulinemia interferes with the induction of lung maturation by glucocorticoids 18
21 Respiratory System: TTN and PPHN Transient tachypnea of the newborn occurs 2-3x more in IDMs related to reduced fluid clearance in the diabetic lung and cesarean delivery Persistent pulmonary hypertension of the newborn Possibly related to vascular sludging L/S ratio >3 for IDMs associated with decreased incidence of PPHN 5. Cardiovascular Issues Hypertrophic cardiomyopathy caused by fat and glycogen deposition into myocardial cells causing thickening of intraventricular septum with reduction in the size of ventricular chambers results in potential obstructed left ventricular outflow infants often asymptomatic but 5-10% have respiratory distress or signs of poor cardiac output usually transient and resolves as plasma insulin levels normalize Symptomatic infants recover after 2-3 weeks of supportive care and ECHO findings resolve within 6 to 12 months 19
22 5. Cardiovascular Issues Hypertrophic cardiomyopathy Cardiovascular Other defects associated with IDM: TGA VSD, ASD Coarctation of the aorta Single ventricle, hypoplastic left ventricle Pulmonic stenosis Pulmonary valve atresia DORV Truncus Arteriosus Tricuspid Atresia PDA 20
23 2458 infants of mothers with type 1 diabetes w/glycated haemoglobin measurement +/- 3 months conception 1,159,865 infants of mothers without diabetes
24 6. Hematologic Changes Hyperglycemia decreases fetal oxygen tension Chronic fetal hypoxemia stimulates production of erythropoietin RBC produc on leads to polycythemia hyperbilirubinemia Intervention required when central Hct >65 with symptoms or > 70 when asymptomatic Hematologic Changes Hyperviscosity risk of vascular sludging, ischemia, seizures, necrotizing enterocolitis, renal vein thrombosis, infarction of vital organs Pulmonary vascular bed sludging can lead to PPHN Low iron stores 22
25 7. Birth Trauma Shoulder dystocia occurs in 1/3 of IDMs that are macrosomic Cephalhematoma Brachial plexus injury Erb s palsy Facial nerve palsy 23
26 8. Neurologic Issues CNS damage from: Asphyxia Birth trauma Hypoglycemia Hyperviscosity Electrolyte abnormalities 24
27 Neurologic Malformations CNS malformation incidence in IDMs vs non- IDMs Anencephaly 13x Spina bifida 20x Caudal regression 200 x Caudal Regression Syndrome Incomplete development of the lumbar and sacral vertebrae Spectrum of structural defects possible Associated with neurologic impairment due to involvement of distal spine 25
28 Neurodevelopmental outcome Outcome of infants of well-controlled diabetic mothers appear similar to that of normal infants Poorly controlled may result in developmental abnormalities Head circumference at 3 years of age negative correlated with HbA1c levels during pregnancy Smaller H.C. associated with poorer intellectual performance 9. GI/GU Issues GI Anomalies Situs inversus atresias (duodenal, anorectal) small left colon syndrome Imperforate anus GU Anomalies: Ureteral duplication Renal agenesis Hydronephrosis 26
29 Glucose Utilization Glucose Homeostasis Balance between hepatic glucose output and peripheral glucose utilization Peripheral utilization varies with demands on the neonate Hypoxia/Hypoxemia Hyperinsulinemia Cold stress Hepatic output depends on glycogenolysis and gluconeogenesis 27
30 Glucose Homeostasis Glucose concentration at which physiologic disturbances occur differ between infants The healthy breastfed term infant has significant levels of circulating ketone bodies The infant with hyperinsulinemia cannot produce ketones or release stored glycogen from the liver Glucose Physiology in the Neonate Glycogenolysis is the mobilization of glucose from hepatic glycogen during the first 6-12 hours of life for cellular use The hormones epinephrine and glucagon stimulate glycogenolysis 28
31 Glucose Physiology in the Neonate Lipolysis, fatty acid oxidation and ketogenesis: occurs hours into the fasting and spares muscle breakdown Making of free fatty acids (a fuel that can be used by the muscles but not the brain) from adipose tissue and fatty acid-derived ketones (fuels that the brain can use) from the liver Normal Glucose Physiology at Delivery Adaptation response Increase in plasma glucagon Decrease in plasma insulin Mobilization of glucose and fatty acids from glycogen and triglyceride deposits 29
32 Glucose Physiology after the Delivery Maternal supply of glucose is interrupted Fetal glycogen storage is temporarily inactivated and blood glucose falls Glycogen phosphorylase breaks down hepatic glycogen stores to supply glucose Gluconeogenesis increases High catecholamine levels stimulate the release of substrates in the form of free fatty acids and free amino acids What is Hypoglycemia? NO UNIVERSAL DEFINITION Operational threshold is defined as that concentration of plasma or whole blood glucose at which clinicians should consider intervention, based on the evidence currently available in literature This threshold is currently believed to be a blood glucose value of less than 40 mg/dl (plasma glucose less than 45 mg/dl). 30
33 Committee on Fetus and Newborn. Postnatal Glucose Homeostasis in Late-Preterm and Term Infants Pediatrics 2011; 127(3); Blood glucose concentrations as low as 30 mg/dl are common in healthy neonates by 1-2 hrs after birth Low concentrations are usually transient, asymptomatic and considered to be part of normal adaptation to postnatal life Clinical Hypoglycemia Newborns < 24 hours of age: blood glucose levels should be consistently > 40mg/dl Newborns > 24 hours of age: blood glucose levels should be consistently > 50mg/dl Preterm infants should have blood glucose consistently >
34 Incidence of Hypoglycemia 1 5 per 1000 births 5-15% in healthy infants 8 % in LGA infants 15 % in SGA infants Up to 40-45% of IDMs (half are asymptomatic) Etiology of Hypoglycemia Decreased hepatic glucose production Immature liver Impaired gluconeogenesis Impaired glycogenolysis Decrease substrate availability due to oral or parental intake issues 32
35 Etiology of Hypoglycemia Sustained hypoxic stress Anaerobic metabolism Increased neurologic needs from a proportionally large brain 13 % of total body mass to 2 % for an adult The increase needs by the brain places increase risks of potential neurologic side effects during hypoglycemia Glycogenolysis and Gluconeogenesis Glycogenolysis Maintains plasma glucose through breakdown of hepatic glycogen Gluconeogenesis Plasma glucose maintained through synthesis of glucose from lactate, glycerol and amino acids Lipolysis Breakdown of fat What happens if first feed is delayed? 66 33
36 Delayed first feeding However, if the first feeding is delayed for three to six hours after birth, approximately 10 percent of normal term newborns cannot maintain a plasma glucose concentration above 30 mg/dl (1.7 mmol/l) 67 Normal low neonatal blood glucose levels Normal levels in utero about 70% of mothers blood glucose Loss of transplacental glucose supply Nadir around 2 hours (<40 mg/dl) Stabilize by 4-6 hours of age to late gestation fetal glucose levels (40-80 mg/dl) Consistent with adult levels by 3-4 days of life 68 34
37 Suckling Ketogenesis Ketones are an alternative fuel for the brain during a fast or periods of hypoglycemia Produced from breakdown of fatty acids in the colostrum (high fat content), peaks on 3rd day Response is blunted with formula May protect newborn brain for injurious affects of hypoglycemia (Hawdon, Ward Platt, & Aynsley-Green, 1992) 69 Inadequate/diminished glucose supply Inadequate glycogen stores Prematurity Fetal growth restriction (FGR/IUGR) Impaired glucose production Glycogenolysis Gluconeogenesis Inborn errors of metabolism Endocrine disorders Other causes (maternal causes, hypothermic infants, hepatic dysfunction) 70 35
38 Increased Glucose Utilization With Hyperinsulinism Infants of diabetic mothers Fetal growth restricted (FGR) infants Beckwith-Wiedemann syndrome (BWS) Perinatal asphyxia or stress 71 Increased Glucose Utilization Without hyperinsulinism Asymmetric FGR Anaerobic glycolysis Decreased tissue perfusion, poor oxygenation, interference with aerobic glucose metabolism Polycythemia Heart failure Perinatal asphyxia Sepsis Drug withdrawal Cold stress 72 36
39 Anaerobic glycolysis 73 Etiology of Hypoglycemia Cold Stress Anaerobic metabolism metabolism glucose Cold stress Hypoxemia O2 consumption respiratory rate 37
40 Perinatal Asphyxia What group of babies are at higher risk of perinatal asphyxia? Shoulder dystocia? IDM babies!! IDM infant prone to hypoglycemia and now, to make matters more complicated
41 Hypoglycemia is Associated with Increased Risk for Brain Injury and Adverse Neurodevelopmental Outcome in Neonates at Risk for Encephalopathy Hypoglycemia (glucose <46) seen in the first 24 hours after birth in 16% of the cohort. After adjusting for potential confounders of early perinatal distress and need for resuscitation Neonatal hypoglycemia was associated with: 3.72-fold increased odds of corticospinal tract injury (P =.047) 4.82-fold increased odds of worsened neuromotor score (P =.038) Lower cognitive and language score on the Bayley Scales of Infant Development (P =.015). Neonatal hypoglycemia is associated with additional risks in the setting of neonatal encephalopathy with increased corticospinal tract injury and adverse motor and cognitive outcomes. 77 Nursing Care of the IDM Comprehensive physical assessment Macrosomic? IUGR? Increased or decreased subcutaneous tissue? Plethoric? Jaundiced? Resp distress? 39
42 Neonatal Management Prior to delivery: Assess need for neonatal resuscitation based on GA, anticipated BW, presence of a congenital anomaly or labor complications/mode of delivery After delivery: Routine care: warm/dry/stim, clearing airway, rapid assessment and examination to identify congenital anomalies Glucose monitoring per unit policy Routine newborn care Neonatal Management - Further evaluation: Cyanosis/resp distress: assess for cardiac and respiratory disease CXR Pre/post ductal oxygen saturations 4 limb BP EKG ECHO Bilirubin, hematocrit checks Electrolytes in symptomatic infants 40
43 Physical Assessment 41
44 The Macrosomic Infant FEED Feed Me! ME!! Dude! I m Will you Hungry be my bodyguard? TOO!!! Erb s Palsy 42
45 Facial nerve palsy Plethoric Baby 43
46 This Baby is Jittery What is the history? When is the onset? Hypoglycemia? Hypomagnesemia? Hypocalcemia? Hyperviscosity? Neurogenic (autonomic) symptoms Jitteriness/tremors Sweating Irritability Tachypnea Pallor 88 44
47 Neuroglycopenic symptoms Poor suck or poor feeding Weak or high-pitched cry Change in level of consciousness Seizures Hypotonia 89 Additional hypoglycemia symptoms Apnea Bradycardia Cyanosis Hypothermia NO SYMPTOMS AT ALL 90 45
48 Jittery Infant Seizing Infant 46
49 Who should be evaluated? Blood glucose concentrations should not be measured in healthy asymptomatic term infants born after an uncomplicated pregnancy and delivery Blood glucose concentration should be measured in infants at risk for hypoglycemia and in infants who exhibit signs or symptoms consistent with hypoglycemia 93 What babies are at risk? Preterm infants including late preterm infants Large or small for gestational age infants Infants with fetal growth restriction (FGR) Infants of diabetic mothers Infants who have experienced perinatal stress: Birth ashphxia/ischemia, Maternal preeclampsia/eclampsia, Meconium aspiration syndrome, Hydrops, Polycythemia Postmature infants Infants requiring intensive care 94 47
50 Timing and frequency of glucose screening Schedule for glucose screening is dependent on the clinical setting: Whenever infant is symptomatic At risk infants One hour after birth Before feeds q3-6 hours for the first hours of life Neonates with identified low blood glucose Continue monitoring until concentrations in a normal range 95 Glucose testing Point of care testing istat Plasma glucose concentration Treatment should be started immediately after the blood sample is obtained and before confirmatory results are available 96 48
51 Diagnosis What makes diagnosing hypoglycemia difficult? 97 Insert chart this report does not identify any specific value or range of plasma glucose concentrations that potentially could result in brain injury. Instead, it is a pragmatic approach to a controversial issue for which evidence is lacking but 98 guidance is needed. 49
52 Management of Hypoglycemia Important to have unit policy and procedure Enteral breastfeeding donor breast milk dextrose water formula dextrose gel Parenteral D10W bolus 2ml/kg Start D10W at 80ml/kg/day Management of Hypoglycemia Recheck glucose within 30 minutes after intervention If glucose does not improve, may need to bolus again and increase IV rate and/or dextrose concentration May need to consider central access if requires concentration exceeds D12.5W Do not use D25W, D50W or large volume boluses as this creates rebound hypoglycemia 50
53 Management of Hypoglycemia Weaning IV infusions: when BG levels have been stable for hours, Begin decreasing IV infusion by 1-2 ml/hr q 3-4 hours if BG > 50-60mg/dL. Further testing should be done in infants who continue to require glucose infusions at rates exceeding 8-10mg/kg/min to maintain normal glucose levels beyond 1 st week of life Adamkin and Committee on Fetus and Newborn,
54 AAP clinical report- when to treat Symptomatic patients requiring treatment Jitteriness/tremors, hypotonia, changes in LOC, apnea/bradycardia, cyanosis, tachypnea, poor suck/feeding, hypothermia, seizures Less than 48 hours of life with plasma glucose levels <50 mg/dl (2.8 mmol/l) Greater than 48 hours of life with plasma glucose levels <60 mg/dl (3.3 mmol/l) 103 AAP clinical report- when to treat Asymptomatic patients at risk for hypoglycemia, or patients in whom low glucose was identified as an incidental laboratory finding Less than 4 hours of life with plasma glucose levels <25 mg/dl (1.4 mmol/l) Between 4 and 24 hours of life with plasma glucose <35 mg/dl (1.9 mmol/l) Between 24 and 48 hours of life with plasma glucose levels <50 mg/dl (2.8 mmol/l) Greater than 48 hours of life with plasma glucose levels <60 mg/dl (3.3 mmol/l)
55 UCSF NC 2 (Northern CA Neonatology Consortium) Representatives from multiple northern California hospitals Meet quarterly to recommend consensus statements on neonatal conditions Consensus statements available on UCSF website Statements include: Hypoglycemia Hyperbilirubinemia Early onset sepsis Soon Apnea of prematurity 105 UCSF NC 2 Asymptomatic Infants/at-risk
56 UCSF NC 2 Asymptomatic Infants-at risk 107 UCSF NC 2 Symptomatic Infants
57 UCSF Newborn Nursery Policy Glucose testing done at 1, 3, 6, 9, 12 hours of life All blood sugars except 1 hour to be done prior to feeds (AC) LGA, IDM, asymptomatic stable infants can d/c testing after 3 comnsecutive BS >45 SGA or LPI also need BS at 18, 24 and 36 hours of life (also completed AC) 109 UCSF Newborn Nursery Policy If glucose >45 and infant asymptomatic, test at next specified time If glucose <45 repeat with different strip and/or meter if possible If test strip is 30-45, place infant skin-to-skin, allow breastfeeding, recheck 1 hour from initiation of feed If the infant is not interested in breastfeeding or mother is not available Oral feed infant D5W or formula at 7 ml/kg and recheck blood sugar 1 hour after initiation of feeding
58 UCSF Newborn Nursery Policy If recheck glucose is > 45 mg/dl, continue with glucose check at next specified time per protocol If recheck, glucose is still mg/dl, and baby is asymptomatic follow procedural steps above If upon recheck glucose is still mg/dl and baby is symptomatic: Notify nursery provider. Oral feed infant D5W or formula at 7mL/kg. If baby has 3 values notify provider 111 UCSF Newborn Nursery Policy If the glucose test is mg/dl repeat the glucose test with a different strip and/or meter if possible Notify provider STAT Oral feed infant D5W or formula at 7ml/kg and recheck on hour from initiation of feeding If recheck glucose test strip is still mg/dl: Draw serum glucose STAT and send to lab. Notify provider STAT. Oral feed infant D5W or formula at 7 ml/kg. Further instructions per provider. Provider may consider IV glucose infusion
59 UCSF Newborn Nursery Policy If glucose test is < 20mg/dL repeat the glucose test with a different test strip and/or meter if possible. Draw serum glucose STAT and send to lab. Notify provider STAT Oral feed infant D5W or formula at 7 mg/kg if baby willing Further instructions per provider. Provider may consider IV glucose infusion. NICU and Special Care / Transitional Care Nursery Treatment thresholds and treatment methods are different for infants in these higher level of care settings; consult neonatology for recommendations 113 UCSF Neonatal Hypoglycemia in the Critical and Transitional Care Setting The picture can't be displayed
60 What if the blood sugar is still low?? Treatment for Refractory Hypoglycemia Hydrocortisone if GIR >12-15 mg/kg/min If GIR >15, consider Endocrine consult Glucagon (wont work if neonate has decreased stores) Diazoxide Octreotide Calcium channel blockers Surgery 115 IV Therapy for Hypoglycemia D10W 2 ml/kg IV bolus, followed by: 80 ml/kg/day (3.5 ml/kg/hr Glucose infusion rate (GIR) 5.5 mg/kg/min Increase D10W in increments of 20 ml/kg/day (0.8 ml/kg/hr) if needed for persistant hypoglycemia If fluid administration exceeds 150 ml/kg/day consider increasing Dextrose concentration to avoid fluid overload Continue therapy until glucose levels are stable >50, then gradually wean IV fluids
61 Neonatal Glycemia and Neurodevelopmental Outcomes at 2 Years (McKinlay et al., 2015) Neonatal hypoglycemia was not associated with adverse neurologic outcomes when infants were treated with the aim of maintaining a blood glucose concentration of at least 47 mg per deciliter The possibility that blood glucose concentrations at the high end of the normal range or unstable blood glucose concentrations and rapid correction of hypoglycemia may be harmful requires further investigation. 117 Neonatal Glycemia and Neurodevelopmental Outcomes at 2 Years (McKinlay et al., 2015) New evidence showing an association of neurodevelopmental impairment with rapid correction of hypoglycemia has led to a suggested approach of bypassing the 2 ml/kg bolus of glucose in favor of simply starting a continuous glucose infusion of 4 to 6mg/kg/min in asymptomatic patients with a gestational age 35 weeks, and at risk for hypoglycemia
62 Results of Interstitial Glucose Monitoring in Children with and Those without Neurosensory Disability at 2 Years. McKinlay CJ et al. N Engl J Med 2015;373: So, You Want Some Sugar?? 60
63 Harris, D. L., Weston, P. J., & Harding, J. E. (2012). Incidence of neonatal hypoglycemia in babies identified as at risk. The Journal of Pediatrics The Sugar Babies Study designed to test efficacy of dextrose gel for treatment of hypoglycemia 514 infants identified as at risk for hypoglycemia (SGA, LGA, IDM, LPI) Screened 1 hr then ac q 4 x 24 hrs then q 8 x 24 hrs Measured incidence of blood glucose (BG) 47 mg/dl and 36 mg/dl The Sugar Babies Study 51% of all pts had BG 47 mg/dl 19% had BG 36 mg/dl 19% had more than one episode Most babies (79%) who became hypoglycemic showed no clinical signs Supports AAP recommendations for screening these groups 61
64 Management of Study Participants Skin to skin contact and early feeding encouraged for all patients in study Infants fed on demand but no more than 3 hrs between feeds Hypoglycemic infants received glucose gel or placebo and encouraged to feed (or were cup or tube fed if unable to suck) If after 30 mins, if the baby was still hypoglycemic, the same process was repeated The Sugar Babies Study-Measured outcomes Primary outcome Treatment failure: (BG < 47) after 2 treatment attempts Secondary Outcomes Admission to the NICU Feeding frequency Volume of expressed breast milk Need for further dextrose (IV or oral) Rebound hypoglycemia Duration of hypoglycemia (up to 48hrs after birth) 62
65 The Sugar Babies Study-Results Primary outcome data showed fewer babies in the dextrose gel group (14%) failed treatment compared to the placebo group (24%) p value 0.04 Babies in the dextrose gel group were less likely to need extra doses of dextrose However those who did receive IV dextrose had similar amounts Admission rates to the NICU were similar, but the dextrose gel group was less likely to be admitted for hypoglycemia The Sugar Babies Study-Results Giving dextrose gel had no adverse effects on breastfeeding Babies in the dextrose gel group: Breastfed more frequently Received less expressed breastmilk At 2 weeks of age, fewer babies were formula feeding Fewer episodes of recurrent hypoglycemia 63
66 The Sugar Babies Study-Results No adverse effects noted from the dextrose gel Treatment with 40% dextrose gel is more effective than feeding alone for reversal of neonatal hypoglycemia in at-risk late preterm and term babies in the first 48 hours after birth The Sugar Babies Study-Impact on Practice Dextrose gel provides a viable first-line/intermediate treatment for neonatal hypoglycemia Easy to administer Keeps mother and baby together Extremely inexpensive Less invasive High availability 64
67 Implementing a Protocol Using Glucose Gel to Treat Neonatal Hypoglycemia Bennett, C., Fagan, E., Chaharbakhshi, E., Zamfirova, I., & Flicker, J. (2016). Implementing a Protocol Using Glucose Gel to Treat Neonatal Hypoglycemia. Nursing For Women's Health, 20(1), Neonates are placed skin to skin and breastfed for the first hour of life A BG level is obtained 30 minutes after this feed is completed If the BG level is <35, the nurse administers a weight based dose of 40% glucose gel by syringe to the neonate s buccal cavity and then places with the mother to feed A BG level is repeated 1 hour after gel administration Implementing a Protocol Using Glucose Gel to Treat Neonatal Hypoglycemia Bennett, C., Fagan, E., Chaharbakhshi, E., Zamfirova, I., & Flicker, J. (2016). Implementing a Protocol Using Glucose Gel to Treat Neonatal Hypoglycemia. Nursing For Women's Health, 20(1), If this BG level is >35 mg/dl, the neonate s BG levels are assessed before feedings until two consecutive readings are >45 mg/dl. If the neonate s BG level is <35, a second dose of gel is given, and again placed with mother to feed In the event that a second dose is needed, a BG level is obtained 1 hour after gel administration. If hypoglycemia is not reversed after the second dose of 40% glucose, the physician is contacted for further orders. 65
68 66
69 Glucose Infusion Rates Glucose production rate in healthy fasting term newborns 4-6mg/kg/minute D10W at 80ml/kg/day = 5.5mg/kg/min Advance GIR to maintain euglycemia Further testing should be done in infants who continue to require glucose infusions at rates exceeding 8-10mg/kg/min to maintain normal glucose levels beyond 1 st week of life 67
70 Oral Sucrose Is not treatment for hypoglycemia, and its usage is independent of needed therapy Legal Implications In one review of neonatal litigation cases from 2 years, 37 % of the cases dealt with neonatal hypoglycemia Lack of screening of high risk neonates, failure to confirm a low glucose concentration and poor documentation of prompt intervention with resolution of clinical symptoms led to litigation 68
71 Long Term Sequelae Motor cognitive delay Impaired glucose tolerance Metabolic syndrome in adulthood 50% of macrosomic IDMs weigh more than the heaviest nondiabetic children at 5-8 years of age (Touger et al.,2005) 69
72 Long term sequelae Adolescent offspring of mothers with GDM had a higher median BMI, greater risk of being overweight, larger median waist circumference and higher fasting insulin levels (2009) Adolescent offspring of mothers with pregestational Type 1 or Type 2 diabetes had a greater BMI compared to control adolescents born to nondiabetic mothers (2010) Macrosomia often present at birth, resolved by 1 year of age but obesity recurrent in childhood Impaired glucose tolerance occurred in 36% Long term sequelae Infants of diabetic mothers are at increased risk for future insulin resistance. May lead to a significant rise in GDM, throwing future generations to a cycle of obesity, insulin resistance, diabetes and various metabolic complications 70
73 Neonatal Hypoglycemia: Answers but More Questions (Rozance & Hay 2012) What are the advantages of lower neonatal glucose concentrations for first 3-4 days of life? Stimulation of glucose production in neonate? Stimulation of appetite? Adaptation of fast/feed cycles? What do our interventions do to interrupt nature? Case Study I 37 6/7 week female infant born to a G 2, P1-2 mother with poorly controlled GDM. Maternal history significant for BMI 38; GBS+; GDM 2001 no interval follow up; hemoglobin A1c 10.6 at first prenatal visit Subsequent Hgb A1C s 10.4, 10.6 (just prior to delivery) IOL delayed for immature lungs 71
74 Case study C/S for intolerance of labor: Birthweight 4065 gm Infant profoundly cyanotic after birth. Echocardiogram reveals transposition of the great arteries (TGA). Questions to ask: Why was she induced? How significant is A1C in 3 rd trimester? Why were the lungs immature? What caused the TGA? Case Study II 72
75 Case Study Male 5kg infant born to a 27 year old G3P1 woman with GDM Pushed x 1 hr when OB attempted vacuum x2 but could tell baby would not come down so moved to C/S under general Thick mec and nuchal cord PPV and chest compressions initially Apgars 1 1, 6 5, 8 10 Case Study CUA: 6.92/82/-18.5 Initial BG 92 HR 160 RR 66 HS blood gases attempted but clotted 7.05/40/-19 via 3 HOL BG 3 HOL 2 ml/kg D10W bolus followed by infusion of 80 ml/kg/d 73
76 Case Study Initially in oxygen via NC at 40% to keep sats 95% Developed increased WOB and chest xray revealed patchy infiltrates consistent with meconium aspiration Also developed upper extremity jerking and eye deviation Transport call placed at 8 hours of life Case Study Review with UCSF neonatologist revealed significant metabolic acidosis at birth and findings consistent with hypoxic ischemic encephalopathy Passive cooling initiated Transport arrived at 9 hours of life, core temperature 36.5 degrees (97.7 F) Target temperature of 33.5 degrees achieved at 10 hours of life (92.3 F) 74
77 Brain Care What issues could have predisposed this baby to have brain injury? What is the criteria for passive cooling? What concerning signs were present that might have indicated this baby was at risk for brain injury? Legal Implications Unfortunately, untoward long-term outcomes in infants with one or two low blood glucose levels have become the grounds for litigation and for alleged malpractice, even though the causative relationship between the two is tenuous at best The definition of clinically significant hypoglycemia remains one of the most confused and contentious issues in contemporary neonatology. Cornblath et al.,
78 Legal Implications In one review of neonatal litigation cases from 2 years, 37 % of the cases dealt with neonatal hypoglycemia. Lack of screening of high risk neonates, failure to confirm a low glucose concentration and poor documentation of prompt intervention with resolution of clinical symptoms led to litigation. 151 In summary Neonatal hypoglycemia remains a controversial topic, as no literature defines what blood glucose is too low, for how long, and at what level or time does irreversible neurodevelopmental injury occur Clinical consensus and guidelines recommend levels that symptomatic and at-risk infants should be treated Dextrose/glucose gels are potentially providing a new management approach for hypoglycemia, while promoting exclusive breastfeeding
79 References Ashwal, E. Hod, M. Gestational diabetes mellitus: Where are we now?, Clin Chim Acta (2015), Barker, D. J., Osmond, C., Forsen, T. J., Kajantie, E., & Eriksson, J. G. (2005). Trajectories of growth among children who have coronary events as adults. The New England Journal of Medicine, 353(17), Barnes-Powell, L. L. (2007). Infants of diabetic mothers: The effects of hyperglycemia on the fetus and neonate. Neonatal Network : NN, 26(5), Bardenheier, B., Elixhauser, A., Imperatore, G., Devlin, H., Kuklina, E., Geiss, L., & Correa, A. (2012). Variation in Prevalence of Gestational Diabetes Mellitus Among Hospital Discharges for Obstetric Delivery Across 23 States in the United States. Diabetes Care, 36(5), doi: /dc Cornblath, M., Hawdon, J. M., Williams, A. F., Aynsley-Green, A., Ward-Platt, M. P., Schwartz, R., et al. (2000). Controversies regarding definition of neonatal hypoglycemia: Suggested operational thresholds. Pediatrics, 105(5), Cowett, R. M., & Loughead, J. L. (2002). Neonatal glucose metabolism: Differential diagnoses, evaluation, and treatment of hypoglycemia. Neonatal Network : NN, 21(4), Esakoff, T. F., Cheng, Y. W., Sparks, T. N., & Caughey, A. B. (2009). The association between birthweight 4000 g or greater and perinatal outcomes in patients with and without gestational diabetes mellitus. American Journal of Obstetrics and Gynecology, References Garg, M., & Devaskar, S. U. (2006). Glucose metabolism in the late preterm infant. Clinics in Perinatology, 33(4), ; abstract ix-x. Harris, D. L., Weston, P. J., & Harding, J. E. (2012). Incidence of neonatal hypoglycemia in babies identified as at risk. The Journal of Pediatrics, Hatfield, L., Schwoebel, A., & Lynyak, C. (2011). Caring for the infant of a diabetic mother. MCN.the American Journal of Maternal Child Nursing, 36(1), Hay, W. W.,Jr. (2012). Care of the infant of the diabetic mother. Current Diabetes Reports, 12(1), Hay, W. W.,Jr, McGowan, J. E., Kalhan, S., Adamkin, D., Hawdon, J. M., Williams, A. F., et al. (2008). How low can I go? the impact of hypoglycemia on the immature brain. Pediatrics, 122(6), ; author reply Hay, W. W.,Jr, Raju, T. N., Higgins, R. D., Kalhan, S. C., & Devaskar, S. U. (2009). Knowledge gaps and research needs for understanding and treating neonatal hypoglycemia: Workshop report from eunice kennedy shriver national institute of child health and human development. The Journal of Pediatrics, 155(5), Kaaja, R. (2009). Vascular complications in diabetic pregnancy. Thrombosis Research, 123 Suppl 2, S
80 References Laptook, A., & Jackson, G. L. (2006). Cold stress and hypoglycemia in the late preterm ("near-term") infant: Impact on nursery of admission. Seminars in Perinatology, 30(1), Maayan-Metzger, A., Lubin, D., & Kuint, J. (2009). Hypoglycemia rates in the first days of life among term infants born to diabetic mothers. Neonatology, 96(2), Milcic, T. L. (2008). Neonatal glucose homeostasis. Neonatal Network : NN, 27(3), Nold, J., & Georgieff, M.K. (2004). Infants of diabetic mothers. Pediatric Clinics of North America, 51, Riskin, A. Infant of a diabetic mother, Post, TW (Ed), UpToDate, Waltham, MA, Rozance, P. J., & Hay, W. W. (2006). Hypoglycemia in newborn infants: Features associated with adverse outcomes. Biology of the Neonate, 90(2), Rozance, P. J., & Hay, W. W.,Jr. (2012). Neonatal hypoglycemia-answers, but more questions. The Journal of Pediatrics Questions? Thank you!! Tanya.Hatfield@ucsf.edu
Neonatal Hypoglycemia. Presented By : Kamlah Olaimat 25\7\2010
 Neonatal Hypoglycemia Presented By : Kamlah Olaimat 25\7\2010 Definition The S.T.A.B.L.E. Program defines hypoglycemia as: Glucose delivery or availability is inadequate to meet glucose demand (Karlsen,
Neonatal Hypoglycemia Presented By : Kamlah Olaimat 25\7\2010 Definition The S.T.A.B.L.E. Program defines hypoglycemia as: Glucose delivery or availability is inadequate to meet glucose demand (Karlsen,
Evidence-Based Update: Using Glucose Gel to Treat Neonatal Hypoglycemia
 Neonatal Nursing Education Brief: Evidence-Based Update: Using Glucose Gel to Treat Neonatal Hypoglycemia http://www.seattlechildrens.org/healthcare-professionals/education/continuing-medicalnursing-education/neonatal-nursing-education-briefs/
Neonatal Nursing Education Brief: Evidence-Based Update: Using Glucose Gel to Treat Neonatal Hypoglycemia http://www.seattlechildrens.org/healthcare-professionals/education/continuing-medicalnursing-education/neonatal-nursing-education-briefs/
Learning Objectives. At the conclusion of this module, participants should be better able to:
 Learning Objectives At the conclusion of this module, participants should be better able to: Treat asymptomatic neonatal hypoglycemia with buccal dextrose gel Develop patient-specific approaches to intravenous
Learning Objectives At the conclusion of this module, participants should be better able to: Treat asymptomatic neonatal hypoglycemia with buccal dextrose gel Develop patient-specific approaches to intravenous
Hypoglycemia. Objectives. Glucose Metabolism
 Hypoglycemia Instructor: Janet Mendis, MSN, RNC-NIC, CNS Outline: Janet Mendis, MSN, RNC-NIC, CNS Summer Morgan, MSN, RNC-NIC, CPNP UC San Diego Health System Objectives State the blood glucose level at
Hypoglycemia Instructor: Janet Mendis, MSN, RNC-NIC, CNS Outline: Janet Mendis, MSN, RNC-NIC, CNS Summer Morgan, MSN, RNC-NIC, CPNP UC San Diego Health System Objectives State the blood glucose level at
Hypoglycaemia of the neonate. Dr. L.G. Lloyd Dept. Paediatrics
 Hypoglycaemia of the neonate Dr. L.G. Lloyd Dept. Paediatrics Why is glucose important? It provides 60-70% of energy needs Utilization obligatory by red blood cells, brain and kidney as major source of
Hypoglycaemia of the neonate Dr. L.G. Lloyd Dept. Paediatrics Why is glucose important? It provides 60-70% of energy needs Utilization obligatory by red blood cells, brain and kidney as major source of
Neonatal Hypoglycemia
 PedsCases Podcast Scripts This is a text version of a podcast from Pedscases.com on Neonatal Hypoglycemia. These podcasts are designed to give medical students an overview of key topics in pediatrics.
PedsCases Podcast Scripts This is a text version of a podcast from Pedscases.com on Neonatal Hypoglycemia. These podcasts are designed to give medical students an overview of key topics in pediatrics.
Disclosures. Objectives. Clinical Case. Risk Factors. Not So Sweet: Evaluation and Management of Infants of Diabetic Mothers
 Not So Sweet: Evaluation and Management of Infants of Diabetic Mothers Disclosures None But. I am a neonatologist NOT an endocrinologist Clinical Advances in Pediatrics September 27, 2017 Jessica Brunkhorst,
Not So Sweet: Evaluation and Management of Infants of Diabetic Mothers Disclosures None But. I am a neonatologist NOT an endocrinologist Clinical Advances in Pediatrics September 27, 2017 Jessica Brunkhorst,
PRACTICE GUIDELINES WOMEN S HEALTH PROGRAM
 C Title: NEWBORN: HYPOGLYCEMIA IN NEONATES BORN AT 35+0 WEEKS GESTATION AND GREATER: DIAGNOSIS AND MANAGEMENT IN THE FIRST 72 HOURS Authorization Section Head, Neonatology, Program Director, Women s Health
C Title: NEWBORN: HYPOGLYCEMIA IN NEONATES BORN AT 35+0 WEEKS GESTATION AND GREATER: DIAGNOSIS AND MANAGEMENT IN THE FIRST 72 HOURS Authorization Section Head, Neonatology, Program Director, Women s Health
7/11/2018. Oral Dextrose Gel Treatment for Newborns with Hypoglycemia Reduces NICU Admissions DISCLOSURE. Objectives
 Gaps in Knowledge, Competence, Performance, or Patient Outcomes DISCLOSURE The content of this presentation does not relate to any product of a commercial entity; therefore, I have no relationships to
Gaps in Knowledge, Competence, Performance, or Patient Outcomes DISCLOSURE The content of this presentation does not relate to any product of a commercial entity; therefore, I have no relationships to
Neurodevelopmental Risk?
 Normal Newborn During transitional hypoglycemia normal newborns have an enhanced ketogenic response to fasting. Newborn brains have enhanced capability to use ketone bodies for fuel Allows newborns to
Normal Newborn During transitional hypoglycemia normal newborns have an enhanced ketogenic response to fasting. Newborn brains have enhanced capability to use ketone bodies for fuel Allows newborns to
W1A- Cases I Learned From
 W1A- Cases I Learned From David H Adamkin, MD Professor of Pediatrics Director, Division of Neonatology Director of Nutritional Research Rounsavall Chair of Neonatal Medicine Co-Director of Neonatal Fellowship
W1A- Cases I Learned From David H Adamkin, MD Professor of Pediatrics Director, Division of Neonatology Director of Nutritional Research Rounsavall Chair of Neonatal Medicine Co-Director of Neonatal Fellowship
Management of Pregestational and Gestational Diabetes Mellitus
 Background and Prevalence Management of Pregestational and Gestational Diabetes Mellitus Pregestational Diabetes - 8 million women in the US are affected, complicating 1% of all pregnancies. Type II is
Background and Prevalence Management of Pregestational and Gestational Diabetes Mellitus Pregestational Diabetes - 8 million women in the US are affected, complicating 1% of all pregnancies. Type II is
Controversies in Neonatal Hypoglycemia PAC / LAC CONFERENCE, JUNE 1 ST 2017
 Controversies in Neonatal Hypoglycemia PRIYA JEGATHEESAN, MD PAC / LAC CONFERENCE, JUNE 1 ST 2017 Disclosure I have no conflicts of interest to disclose Objectives Review Recommendations from different
Controversies in Neonatal Hypoglycemia PRIYA JEGATHEESAN, MD PAC / LAC CONFERENCE, JUNE 1 ST 2017 Disclosure I have no conflicts of interest to disclose Objectives Review Recommendations from different
Newborn Hypoglycemia
 Newborn Hypoglycemia Self Learning Module Developed by the Interprofessional Education and Research Committee of the Champlain Maternal Newborn Regional Program (CMNRP) 2013 TABLE OF CONTENTS Definition....3
Newborn Hypoglycemia Self Learning Module Developed by the Interprofessional Education and Research Committee of the Champlain Maternal Newborn Regional Program (CMNRP) 2013 TABLE OF CONTENTS Definition....3
Infant Of Diabetic Mother(IDM)
 Infant Of Diabetic Mother(IDM) Sangram Satish Magar 1, Sanskriti Mirashi 2 1. M.D. Sch.(Kaumarbhrutya-Balrog) 2.Guide (Kaumarbhrutya-Balrog), L.R.P.Medical college,islampur,tal- Walwa, dist- Sangli, Maharashtra,
Infant Of Diabetic Mother(IDM) Sangram Satish Magar 1, Sanskriti Mirashi 2 1. M.D. Sch.(Kaumarbhrutya-Balrog) 2.Guide (Kaumarbhrutya-Balrog), L.R.P.Medical college,islampur,tal- Walwa, dist- Sangli, Maharashtra,
NEONATAL HYPOGLYCEMIA HEATHER MCKNIGHT-MENCI, MSN, CRNP CHILDREN S HOSPITAL OF PHILADELPHIA
 NEONATAL HYPOGLYCEMIA HEATHER MCKNIGHT-MENCI, MSN, CRNP CHILDREN S HOSPITAL OF PHILADELPHIA WHAT IS NEONATAL HYPOGLYCEMIA? Glucose concentration low enough to cause signs and symptoms of impaired brain
NEONATAL HYPOGLYCEMIA HEATHER MCKNIGHT-MENCI, MSN, CRNP CHILDREN S HOSPITAL OF PHILADELPHIA WHAT IS NEONATAL HYPOGLYCEMIA? Glucose concentration low enough to cause signs and symptoms of impaired brain
Diabetes in Pregnancy
 Disclosure Diabetes in Pregnancy I have no conflicts of interest to disclose Jennifer Krupp, MD Maternal Fetal Medicine St. Marys Hospital/SSM Health Madison, WI Objectives Classification of Diabetes Classifications
Disclosure Diabetes in Pregnancy I have no conflicts of interest to disclose Jennifer Krupp, MD Maternal Fetal Medicine St. Marys Hospital/SSM Health Madison, WI Objectives Classification of Diabetes Classifications
Diabetes: The Effects of Maternal Diabetes on Fetal Development and Outcomes Sherrie McElvy, MD May 18, 2016
 Diabetes: The Effects of Maternal Diabetes on Fetal Development and Outcomes Sherrie McElvy, MD May 18, 2016 Medical Director Sweet Success Sutter Medical Center Sacramento Perinatal Associates of Sacramento
Diabetes: The Effects of Maternal Diabetes on Fetal Development and Outcomes Sherrie McElvy, MD May 18, 2016 Medical Director Sweet Success Sutter Medical Center Sacramento Perinatal Associates of Sacramento
Neonatal Hypoglycaemia Guidelines
 N.B. Staff should be discouraged from printing this document. This is to avoid the risk of out of date printed versions of the document. The Intranet should be referred to for the current version of the
N.B. Staff should be discouraged from printing this document. This is to avoid the risk of out of date printed versions of the document. The Intranet should be referred to for the current version of the
Diabetes in Pregnancy. L.Sekhavat MD
 Diabetes in Pregnancy L.Sekhavat MD Diabetes in Pregnancy Gestational Diabetes Pre-gestational diabetes (overt) Insulin dependent (type1) Non-insulin dependent (type 2) Definition Gestational diabetes
Diabetes in Pregnancy L.Sekhavat MD Diabetes in Pregnancy Gestational Diabetes Pre-gestational diabetes (overt) Insulin dependent (type1) Non-insulin dependent (type 2) Definition Gestational diabetes
Vishwanath Pattan Endocrinology Wyoming Medical Center
 Vishwanath Pattan Endocrinology Wyoming Medical Center Disclosure Holdings in Tandem Non for this Training Introduction In the United States, 5 to 6 percent of pregnancies almost 250,000 women are affected
Vishwanath Pattan Endocrinology Wyoming Medical Center Disclosure Holdings in Tandem Non for this Training Introduction In the United States, 5 to 6 percent of pregnancies almost 250,000 women are affected
بنام خدا هیپوگلیسمی درنوزادان و گاالکتوزمی دکتر انتظاری
 بنام خدا هیپوگلیسمی درنوزادان و گاالکتوزمی دکتر انتظاری Serum glucose< 35 mg/dl 1-3 hr of life < 40 mg/dl 3-24 hr < 45 mg/dl after 24 hr 10% NL newborns can t maintain BS>30 if delayed feeding >3-6 hrs
بنام خدا هیپوگلیسمی درنوزادان و گاالکتوزمی دکتر انتظاری Serum glucose< 35 mg/dl 1-3 hr of life < 40 mg/dl 3-24 hr < 45 mg/dl after 24 hr 10% NL newborns can t maintain BS>30 if delayed feeding >3-6 hrs
Case 1. Managing Neonatal Hypoglycemia: Can formula supplementation be avoided? Faculty Financial Disclosures 3/8/2018
 Managing Neonatal Hypoglycemia: Can formula supplementation be avoided? Shawnte R. James, MD, FAAP Assistant Professor Newborn Medicine Hospitalist Emory University School of Medicine Department of Pediatrics
Managing Neonatal Hypoglycemia: Can formula supplementation be avoided? Shawnte R. James, MD, FAAP Assistant Professor Newborn Medicine Hospitalist Emory University School of Medicine Department of Pediatrics
What is symptomatic? Neonatal hypoglycemia: how low can you go? Hypoglycemia and MRI. Conflicts. What s the problem? Hypoglycemia and MRI
 Neonatal hypoglycemia: how low can you go? Kristi Watterberg, MD Professor of Pediatrics, UNM What is symptomatic? Jitteriness Cyanosis Poor feeding Weak, high-pitched cry Seizures Apnea Lethargy, low
Neonatal hypoglycemia: how low can you go? Kristi Watterberg, MD Professor of Pediatrics, UNM What is symptomatic? Jitteriness Cyanosis Poor feeding Weak, high-pitched cry Seizures Apnea Lethargy, low
The New GDM Screening Guidelines. Jennifer Klinke MD, FRCPC Endocrinologist and Co director RCH Diabetes in Pregnancy Program
 The New GDM Screening Guidelines Jennifer Klinke MD, FRCPC Endocrinologist and Co director RCH Diabetes in Pregnancy Program Disclosures Current participant (RCH site) for MiTy study Metformin in women
The New GDM Screening Guidelines Jennifer Klinke MD, FRCPC Endocrinologist and Co director RCH Diabetes in Pregnancy Program Disclosures Current participant (RCH site) for MiTy study Metformin in women
Neonatal Hypoglycaemia
 Neonatal Hypoglycaemia Dr Shubha Srinivasan Paediatric Endocrinologist The Children s Hospital at Westmead Hypoglycaemia and the Brain CSF glucose is 2/3 that of plasma Intracerebral glucose 1/3 that of
Neonatal Hypoglycaemia Dr Shubha Srinivasan Paediatric Endocrinologist The Children s Hospital at Westmead Hypoglycaemia and the Brain CSF glucose is 2/3 that of plasma Intracerebral glucose 1/3 that of
NEONATAL CLINICAL PRACTICE GUIDELINE
 NEONATAL CLINICAL PRACTICE GUIDELINE 1.0 PURPOSE AND INTENT Title: Approval Date: March 2016 Approved by: Neonatal Patient Care Teams, HSC & SBH Women s Health Maternal Newborn Committee Child Health Standards
NEONATAL CLINICAL PRACTICE GUIDELINE 1.0 PURPOSE AND INTENT Title: Approval Date: March 2016 Approved by: Neonatal Patient Care Teams, HSC & SBH Women s Health Maternal Newborn Committee Child Health Standards
Prevention and Management of Hypoglycaemia of the Breastfed Newborn Reference Number:
 This is an official Northern Trust policy and should not be edited in any way Prevention and Management of Hypoglycaemia of the Breastfed Newborn Reference Number: NHSCT/10/293 Target audience: Midwifery,
This is an official Northern Trust policy and should not be edited in any way Prevention and Management of Hypoglycaemia of the Breastfed Newborn Reference Number: NHSCT/10/293 Target audience: Midwifery,
Screening for Critical Congenital Heart Disease
 Screening for Critical Congenital Heart Disease Caroline K. Lee, MD Pediatric Cardiology Disclosures I have no relevant financial relationships or conflicts of interest 1 Most Common Birth Defect Most
Screening for Critical Congenital Heart Disease Caroline K. Lee, MD Pediatric Cardiology Disclosures I have no relevant financial relationships or conflicts of interest 1 Most Common Birth Defect Most
Guideline for the Prevention and Management of Neonatal Hypoglycaemia GL359
 Guideline for the Prevention and Management of Neonatal Hypoglycaemia GL359 Approval and Authorisation Approved by Job Title Date Paediatric Clinical Chair of Paediatric Clinical Governance Governance
Guideline for the Prevention and Management of Neonatal Hypoglycaemia GL359 Approval and Authorisation Approved by Job Title Date Paediatric Clinical Chair of Paediatric Clinical Governance Governance
Summary of Changes: References/content updated to reflect most current standards of practice.
 Alaska Native Medical Center: Mother Baby Unit Guideline: Neonatal Hypoglycemia Subject: Neonatal Hypoglycemia REVISION DATE: Jan 2015,12/2011, 02/2009, 11, 2007, 07/2007,04/2001, 04/1999 REPLACES: NSY:
Alaska Native Medical Center: Mother Baby Unit Guideline: Neonatal Hypoglycemia Subject: Neonatal Hypoglycemia REVISION DATE: Jan 2015,12/2011, 02/2009, 11, 2007, 07/2007,04/2001, 04/1999 REPLACES: NSY:
Objectives. Birth Depression Management. Birth Depression Terms
 Objectives Birth Depression Management Regional Perinatal Outreach Program 2016 Understand the terms and the clinical characteristics of birth depression. Be familiar with the evidence behind therapeutic
Objectives Birth Depression Management Regional Perinatal Outreach Program 2016 Understand the terms and the clinical characteristics of birth depression. Be familiar with the evidence behind therapeutic
Pregestational and Gestational Diabetes
 Pregestational and Gestational Diabetes Francis S. Nuthalapaty, MD Greenville Health System University of South Carolina School of Medicine - Greenville Case History 30 year old black female presents to
Pregestational and Gestational Diabetes Francis S. Nuthalapaty, MD Greenville Health System University of South Carolina School of Medicine - Greenville Case History 30 year old black female presents to
Infection. Risk factor for infection ACoRN alerting sign with * Clinical deterioration. Problem List. Respiratory. Cardiovascular
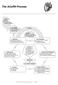 The ACoRN Process Baby at risk Unwell Risk factors Post-resuscitation requiring stabilization Resuscitation Ineffective breathing Heart rate < 100 bpm Central cyanosis Support Infection Risk factor for
The ACoRN Process Baby at risk Unwell Risk factors Post-resuscitation requiring stabilization Resuscitation Ineffective breathing Heart rate < 100 bpm Central cyanosis Support Infection Risk factor for
Guidelines for the Prevention and Management of Hypoglycaemia
 ASHFORD & ST PETER S HOSPITALS NHS TRUST CHILDREN S SERVICES Guidelines for the Prevention and Management of Hypoglycaemia 1. The Care Plan for Prevention of Hypoglycaemia on Labour Ward and the Postnatal
ASHFORD & ST PETER S HOSPITALS NHS TRUST CHILDREN S SERVICES Guidelines for the Prevention and Management of Hypoglycaemia 1. The Care Plan for Prevention of Hypoglycaemia on Labour Ward and the Postnatal
The high risk neonate
 The high risk neonate Infant classification by gestational (postmenstrual) age Preterm. Less than 37 completed weeks (259 days). Term. Thirty-seven to 416/7 weeks (260-294 days). Post-term. Forty-two weeks
The high risk neonate Infant classification by gestational (postmenstrual) age Preterm. Less than 37 completed weeks (259 days). Term. Thirty-seven to 416/7 weeks (260-294 days). Post-term. Forty-two weeks
2/13/2018. Update on Gestational Diabetes. Disclosure. Objectives. I have no financial conflicts of interest.
 Update on Gestational Diabetes Lorie M. Harper, MD, MSCI Department of Obstetrics & Gynecology Division of Maternal-Fetal Medicine 2/18/2018 Disclosure I have no financial conflicts of interest. Objectives
Update on Gestational Diabetes Lorie M. Harper, MD, MSCI Department of Obstetrics & Gynecology Division of Maternal-Fetal Medicine 2/18/2018 Disclosure I have no financial conflicts of interest. Objectives
Newborn Glucose Management Clinical Decision Support System
 Newborn Glucose Management Clinical Decision Support System Marie Kozel, MBA, BSN, RNC-BC 6 th Annual Nursing Quality Conference 2012 Thursday, January 26, 2012 2:45 PM Objectives 1. Describe a newborn
Newborn Glucose Management Clinical Decision Support System Marie Kozel, MBA, BSN, RNC-BC 6 th Annual Nursing Quality Conference 2012 Thursday, January 26, 2012 2:45 PM Objectives 1. Describe a newborn
Gestational Diabetes Mellitus Dr. Fawaz Amin Saad
 Gestational Diabetes Mellitus Dr. Fawaz Amin Saad Senior Consultant OB/GYN, Al-Hayat Medical Center, Doha, Qatar DISCLOSURE OF CONFLICT OF INTEREST I am a full-time Employee at Al-Hayat Medical Center.
Gestational Diabetes Mellitus Dr. Fawaz Amin Saad Senior Consultant OB/GYN, Al-Hayat Medical Center, Doha, Qatar DISCLOSURE OF CONFLICT OF INTEREST I am a full-time Employee at Al-Hayat Medical Center.
Intrapartum and Postpartum Management of the Diabetic Mother and Infant
 Intrapartum and Postpartum Management of the Diabetic Mother and Infant Intrapartum Management Women with gestational diabetes who maintain normal glucose levels during pregnancy on diet and exercise therapy
Intrapartum and Postpartum Management of the Diabetic Mother and Infant Intrapartum Management Women with gestational diabetes who maintain normal glucose levels during pregnancy on diet and exercise therapy
Hypoglycemia in congenital hyperinsulinism
 How a normal body works: Our body is constantly at work. Our cells need a source of energy, and this source of energy is called glucose. The process is quite simple; think of it like an assembly line.
How a normal body works: Our body is constantly at work. Our cells need a source of energy, and this source of energy is called glucose. The process is quite simple; think of it like an assembly line.
Hypothermia in Neonates with HIE TARA JENDZIO, DNP(C), RN, RNC-NIC
 Hypothermia in Neonates with HIE TARA JENDZIO, DNP(C), RN, RNC-NIC Objectives 1. Define Hypoxic-Ischemic Encephalopathy (HIE) 2. Identify the criteria used to determine if an infant qualifies for therapeutic
Hypothermia in Neonates with HIE TARA JENDZIO, DNP(C), RN, RNC-NIC Objectives 1. Define Hypoxic-Ischemic Encephalopathy (HIE) 2. Identify the criteria used to determine if an infant qualifies for therapeutic
Diabetes in Pregnancy
 Diabetes in Pregnancy Resident School November 5 2014 Goals Be able to screen for gestational and preexisting diabetes Be able to counsel women on the diagnosis of gestational diabetes Understand glucose
Diabetes in Pregnancy Resident School November 5 2014 Goals Be able to screen for gestational and preexisting diabetes Be able to counsel women on the diagnosis of gestational diabetes Understand glucose
CARE OF THE NEWBORN HYPOGLYCAEMIA
 CARE OF THE NEWBORN HYPOGLYCAEMIA Background Definition A true blood glucose of less than 2.6 mmol/l venous sample. Neonatal hypoglycaemia is commonly defined as a true blood glucose of less than 2.6 mmol/l
CARE OF THE NEWBORN HYPOGLYCAEMIA Background Definition A true blood glucose of less than 2.6 mmol/l venous sample. Neonatal hypoglycaemia is commonly defined as a true blood glucose of less than 2.6 mmol/l
Gestational Diabetes. Gestational Diabetes:
 Gestational Diabetes Detection and Management Steven Gabbe, MD The Ohio State University Medical Center Gestational Diabetes: Detection and Management Learning Objectives: At the conclusion of this presentation,
Gestational Diabetes Detection and Management Steven Gabbe, MD The Ohio State University Medical Center Gestational Diabetes: Detection and Management Learning Objectives: At the conclusion of this presentation,
PES Recommendations for Evaluation and Management of Hypoglycemia in Neonates, Infants, and Children Paul S. Thornton On behalf of the Team
 Cook Children s 1 PES Recommendations for Evaluation and Management of Hypoglycemia in Neonates, Infants, and Children Paul S. Thornton On behalf of the Team Cook Children s 2 Co-Chair: Charles Stanley
Cook Children s 1 PES Recommendations for Evaluation and Management of Hypoglycemia in Neonates, Infants, and Children Paul S. Thornton On behalf of the Team Cook Children s 2 Co-Chair: Charles Stanley
Gestational Diabetes. Benjamin Byers, D.O., FACOG Center for Maternal and Fetal Care Bryan Physician Network
 Gestational Diabetes Benjamin Byers, D.O., FACOG Center for Maternal and Fetal Care Bryan Physician Network Outline Definition Prevalence Risk factors complications Diagnosis Management Nonpharmacologic
Gestational Diabetes Benjamin Byers, D.O., FACOG Center for Maternal and Fetal Care Bryan Physician Network Outline Definition Prevalence Risk factors complications Diagnosis Management Nonpharmacologic
The Ever-Changing Approaches to Diabetes in Pregnancy
 The Ever-Changing Approaches to Diabetes in Pregnancy Kirsten E. Salmeen, MD Assistant Professor Obstetrics, Gynecology & Reproductive Sciences Maternal-Fetal Medicine I have nothing to disclose. Approaches
The Ever-Changing Approaches to Diabetes in Pregnancy Kirsten E. Salmeen, MD Assistant Professor Obstetrics, Gynecology & Reproductive Sciences Maternal-Fetal Medicine I have nothing to disclose. Approaches
Lectures 4 Early fetal assessment, screening, ultrasound and treatment modalities during pregnancy. II. Asphyxia and Resuscitation (3 lectures)...
 Outline of a 2 year Neonatology educational course (80 lectures) PLUS 2 graduate level courses (GENETICS and BIOSTATISTICS & EPIDEMIOLOGY Approximate Percent in Examination I. Maternal-Fetal Medicine (6
Outline of a 2 year Neonatology educational course (80 lectures) PLUS 2 graduate level courses (GENETICS and BIOSTATISTICS & EPIDEMIOLOGY Approximate Percent in Examination I. Maternal-Fetal Medicine (6
NEONATOLOGY Healthy newborn. Neonatal sequelaes
 NEONATOLOGY Healthy newborn. Neonatal sequelaes Ágnes Harmath M.D. Ph.D. senior lecturer 11. November 2016. Tasks of the neonatologist Prenatal diagnosed condition Inform parents, preparation of necessary
NEONATOLOGY Healthy newborn. Neonatal sequelaes Ágnes Harmath M.D. Ph.D. senior lecturer 11. November 2016. Tasks of the neonatologist Prenatal diagnosed condition Inform parents, preparation of necessary
Effect of Various Degrees of Maternal Hyperglycemia on Fetal Outcome
 ORIGINAL ARTICLE Effect of Various Degrees of Maternal Hyperglycemia on Fetal Outcome ABSTRACT Shagufta Tahir, Shaheen Zafar, Savita Thontia Objective Study design Place & Duration of study Methodology
ORIGINAL ARTICLE Effect of Various Degrees of Maternal Hyperglycemia on Fetal Outcome ABSTRACT Shagufta Tahir, Shaheen Zafar, Savita Thontia Objective Study design Place & Duration of study Methodology
A S Y N T H E S I Z E D H A N D B O O K ON G E S T A T I O N A L D I A B E T E S
 A S Y N T H E S I Z E D H A N D B O O K ON G E S T A T I O N A L D I A B E T E S P R E F A C E Dear reader, This is a synthesized handbook conceived to serve as a tool to health personnel in the screening,
A S Y N T H E S I Z E D H A N D B O O K ON G E S T A T I O N A L D I A B E T E S P R E F A C E Dear reader, This is a synthesized handbook conceived to serve as a tool to health personnel in the screening,
Admission/Discharge Form for Infants Born in Please DO NOT mail or fax this form to the CPQCC Data Center. This form is for internal use ONLY.
 Selection Criteria Admission/Discharge Form for Infants Born in 2016 To be eligible, you MUST answer YES to at least one of the possible criteria (A-C) A. 401 1500 grams o Yes B. GA range 22 0/7 31 6/7
Selection Criteria Admission/Discharge Form for Infants Born in 2016 To be eligible, you MUST answer YES to at least one of the possible criteria (A-C) A. 401 1500 grams o Yes B. GA range 22 0/7 31 6/7
Gestational Diabetes: An Update on Testing. Kimberlee A McKay, M.D. Avera Medical Group Ob/GYN
 Gestational Diabetes: An Update on Testing Kimberlee A McKay, M.D. Avera Medical Group Ob/GYN Gestational Diabetes Increased risks of: Still Birth Hydramnios Should Dystocia Prolonged Labor Preeclampsia
Gestational Diabetes: An Update on Testing Kimberlee A McKay, M.D. Avera Medical Group Ob/GYN Gestational Diabetes Increased risks of: Still Birth Hydramnios Should Dystocia Prolonged Labor Preeclampsia
How to Recognize a Suspected Cardiac Defect in the Neonate
 Neonatal Nursing Education Brief: How to Recognize a Suspected Cardiac Defect in the Neonate https://www.seattlechildrens.org/healthcareprofessionals/education/continuing-medical-nursing-education/neonatalnursing-education-briefs/
Neonatal Nursing Education Brief: How to Recognize a Suspected Cardiac Defect in the Neonate https://www.seattlechildrens.org/healthcareprofessionals/education/continuing-medical-nursing-education/neonatalnursing-education-briefs/
A Study of Prevalence and Risk Factors of Polycythemia in Neonatal Nursery in Duhok
 ORIGINAL ARTICLE A Study of Prevalence and Risk Factors of Polycythemia in Neonatal Nursery in Duhok Akrem Mohammad Mostefa 1 ABSTRACT OBJECTIVE: To find the prevalence of neonatal polycythemia among neonates
ORIGINAL ARTICLE A Study of Prevalence and Risk Factors of Polycythemia in Neonatal Nursery in Duhok Akrem Mohammad Mostefa 1 ABSTRACT OBJECTIVE: To find the prevalence of neonatal polycythemia among neonates
OB Well Baby Nursery Admission (Term) [ ] For specialty focused order sets for your patient, refer to: General
![OB Well Baby Nursery Admission (Term) [ ] For specialty focused order sets for your patient, refer to: General OB Well Baby Nursery Admission (Term) [ ] For specialty focused order sets for your patient, refer to: General](/thumbs/85/91909911.jpg) OB Well Baby Nursery Admission (Term) [3040000234] For specialty focused order sets for your patient, refer to: 3040000424 Neonatal Circumcision Order Set 3040000522 Neonatal Herpes Viral Order Set 3040000524
OB Well Baby Nursery Admission (Term) [3040000234] For specialty focused order sets for your patient, refer to: 3040000424 Neonatal Circumcision Order Set 3040000522 Neonatal Herpes Viral Order Set 3040000524
Pregnancies complicated by diabetes. Marina Mickleson Nurse Practitioner Midwife CDE
 Pregnancies complicated by diabetes Marina Mickleson Nurse Practitioner Midwife CDE Two types Pre gestational Gestational diabetes Both types are on the increase Pre conception work up is imperative for
Pregnancies complicated by diabetes Marina Mickleson Nurse Practitioner Midwife CDE Two types Pre gestational Gestational diabetes Both types are on the increase Pre conception work up is imperative for
Nottingham Neonatal Service Guideline
 Title: Screening and Management of Neonatal Hypoglycaemia Version: 10 Ratification Date: Review Date: Jan 2023 Approval: Nottingham Neonatal Service Clinical Guideline Meeting Author: Shalini Ojha 1, Lara
Title: Screening and Management of Neonatal Hypoglycaemia Version: 10 Ratification Date: Review Date: Jan 2023 Approval: Nottingham Neonatal Service Clinical Guideline Meeting Author: Shalini Ojha 1, Lara
Data from birth certificates in the United
 Chapter 36 Pregnancy in Preexisting Diabetes Thomas A. Buchanan, M.D. SUMMARY Data from birth certificates in the United States indicate that maternal diabetes complicates 2%-3% of all pregnancies, but
Chapter 36 Pregnancy in Preexisting Diabetes Thomas A. Buchanan, M.D. SUMMARY Data from birth certificates in the United States indicate that maternal diabetes complicates 2%-3% of all pregnancies, but
How Un It is: The Conundrum of What to Do for Transient Neonatal Hypoglycemia
 How Un It is: The Conundrum of What to Do for Transient Neonatal Hypoglycemia Jeffrey R. Kaiser, MD, MA Kenneth V. and Eleanor M. Hatt Professor of Neonatal Medicine Division of Neonatal-Perinatal Medicine
How Un It is: The Conundrum of What to Do for Transient Neonatal Hypoglycemia Jeffrey R. Kaiser, MD, MA Kenneth V. and Eleanor M. Hatt Professor of Neonatal Medicine Division of Neonatal-Perinatal Medicine
Objectives. Diabetes and Obesity in Pregnancy. In Diabetes. Diabetes in Pregnancy
 Objectives Diabetes and Obesity in Pregnancy. Health Impact for the mother and child Bresta Miranda, MD Assistant Professor of Clinical Medicine University of Miami, Miller School of Medicine Review physiologic
Objectives Diabetes and Obesity in Pregnancy. Health Impact for the mother and child Bresta Miranda, MD Assistant Professor of Clinical Medicine University of Miami, Miller School of Medicine Review physiologic
Diabetes in Pregnancy
 Diabetes in Pregnancy Ebony Boyce Carter, MD, MPH Division of Maternal Fetal Medicine Washington University School of Medicine Disclosures I have no financial disclosures to report. Objectives Review the
Diabetes in Pregnancy Ebony Boyce Carter, MD, MPH Division of Maternal Fetal Medicine Washington University School of Medicine Disclosures I have no financial disclosures to report. Objectives Review the
Review of Neonatal Respiratory Problems
 Review of Neonatal Respiratory Problems Respiratory Distress Occurs in about 7% of infants Clinical presentation includes: Apnea Cyanosis Grunting Inspiratory stridor Nasal flaring Poor feeding Tachypnea
Review of Neonatal Respiratory Problems Respiratory Distress Occurs in about 7% of infants Clinical presentation includes: Apnea Cyanosis Grunting Inspiratory stridor Nasal flaring Poor feeding Tachypnea
Diabetes in obstetric patients
 Diabetes in obstetric patients Swedish Society of Obstetric Anaesthesia & Intensive Care Anita Banerjee Obstetric Physician Diabetes & Endocrinology Consultant Outline Scope of the problem Diabetes and
Diabetes in obstetric patients Swedish Society of Obstetric Anaesthesia & Intensive Care Anita Banerjee Obstetric Physician Diabetes & Endocrinology Consultant Outline Scope of the problem Diabetes and
Metformin Hydrochloride
 Metformin Hydrochloride 500 mg, 850 mg, 500 mg LA and 750 mg LA Tablet Description Informet is a preparation of metformin hydrochloride that belongs to a biguanide class of oral antidiabetic drugs. Metformin
Metformin Hydrochloride 500 mg, 850 mg, 500 mg LA and 750 mg LA Tablet Description Informet is a preparation of metformin hydrochloride that belongs to a biguanide class of oral antidiabetic drugs. Metformin
CCHD Screening with Pulse Oximetry: A Success Story!
 CCHD Screening with Pulse Oximetry: A Success Story! Nicole Spillane, MD Associate Director of Neonatology, HackensackUMC Hackensack Meridian Health Objective Recognize the contribution of pulse oximetry
CCHD Screening with Pulse Oximetry: A Success Story! Nicole Spillane, MD Associate Director of Neonatology, HackensackUMC Hackensack Meridian Health Objective Recognize the contribution of pulse oximetry
The Blue Baby. Network Stabilisation of the Term Infant Study Day 15 th March 2017 Joanna Behrsin
 The Blue Baby Network Stabilisation of the Term Infant Study Day 15 th March 2017 Joanna Behrsin Session Structure Definitions and assessment of cyanosis Causes of blue baby Structured approach to assessing
The Blue Baby Network Stabilisation of the Term Infant Study Day 15 th March 2017 Joanna Behrsin Session Structure Definitions and assessment of cyanosis Causes of blue baby Structured approach to assessing
Running head: ORAL DEXTROSE GEL IN THE TREATMENT OF NH 1. Oral Dextrose Gel in the Treatment of Neonatal Hypoglycemia:
 Running head: ORAL DEXTROSE GEL IN THE TREATMENT OF NH 1 Oral Dextrose Gel in the Treatment of Neonatal Hypoglycemia: An Evidence Based Practice Change Sylvia Cruz Arizona State University NEONATAL HYPOGLYCEMIA
Running head: ORAL DEXTROSE GEL IN THE TREATMENT OF NH 1 Oral Dextrose Gel in the Treatment of Neonatal Hypoglycemia: An Evidence Based Practice Change Sylvia Cruz Arizona State University NEONATAL HYPOGLYCEMIA
HYPOGLYCAEMIA OF THE NEWBORN ON BIRTHING SUITE AND POSTNATAL WARD
 HYPOGLYCAEMIA OF THE NEWBORN ON BIRTHING SUITE AND POSTNATAL WARD INTRODUCTION Healthy term infants are able to mobilise energy stores through a process known as counter regulation and are unlikely to
HYPOGLYCAEMIA OF THE NEWBORN ON BIRTHING SUITE AND POSTNATAL WARD INTRODUCTION Healthy term infants are able to mobilise energy stores through a process known as counter regulation and are unlikely to
Maximizing the Role of WIC Nutritionists in Prevention of DM2 among High Risk Clients ESTHER G. SCHUSTER, MS,RD,CDE
 Maximizing the Role of WIC Nutritionists in Prevention of DM2 among High Risk Clients ESTHER G. SCHUSTER, MS,RD,CDE Heavy Numbers Surgeon General report: 68% of adults in U. S. are overweight or obese
Maximizing the Role of WIC Nutritionists in Prevention of DM2 among High Risk Clients ESTHER G. SCHUSTER, MS,RD,CDE Heavy Numbers Surgeon General report: 68% of adults in U. S. are overweight or obese
Diabetes in Pregnancy
 Diabetes in Pregnancy Susan Drummond RN MSN C-EFM Objectives 1. Describe types of diabetes and diagnosis of gestational diabetes 2. Identify a management plan for diabetes during pregnancy 3. Describe
Diabetes in Pregnancy Susan Drummond RN MSN C-EFM Objectives 1. Describe types of diabetes and diagnosis of gestational diabetes 2. Identify a management plan for diabetes during pregnancy 3. Describe
NEONATAL CLINICAL PRACTICE GUIDELINE
 NEONATAL CLINICAL PRACTICE GUIDELINE 1.0 PURPOSE AND INTENT Title: Approval Date: March 2016, Last Revised July 2018 Approved by: Neonatal Patient Care Teams, HSC & SBH Women s Health Maternal Newborn
NEONATAL CLINICAL PRACTICE GUIDELINE 1.0 PURPOSE AND INTENT Title: Approval Date: March 2016, Last Revised July 2018 Approved by: Neonatal Patient Care Teams, HSC & SBH Women s Health Maternal Newborn
DIABETES WITH PREGNANCY
 DIABETES WITH PREGNANCY Prof. Aasem Saif MD,MRCP(UK),FRCP (Edinburgh) Maternal and Fetal Risks Diabetes in pregnancy is associated with risks to the woman and to the developing fetus. Maternal and Fetal
DIABETES WITH PREGNANCY Prof. Aasem Saif MD,MRCP(UK),FRCP (Edinburgh) Maternal and Fetal Risks Diabetes in pregnancy is associated with risks to the woman and to the developing fetus. Maternal and Fetal
UNIVERSITY OF PNG SCHOOL OF MEDICINE AND HEALTH SCIENCES DIVISION OF BASIC MEDICAL SCIENCES DISCIPLINE OF BIOCHEMISTRY AND MOLECULAR BIOLOGY
 1 UNIVERSITY OF PNG SCHOOL OF MEDICINE AND HEALTH SCIENCES DIVISION OF BASIC MEDICAL SCIENCES DISCIPLINE OF BIOCHEMISTRY AND MOLECULAR BIOLOGY GLUCOSE HOMEOSTASIS An Overview WHAT IS HOMEOSTASIS? Homeostasis
1 UNIVERSITY OF PNG SCHOOL OF MEDICINE AND HEALTH SCIENCES DIVISION OF BASIC MEDICAL SCIENCES DISCIPLINE OF BIOCHEMISTRY AND MOLECULAR BIOLOGY GLUCOSE HOMEOSTASIS An Overview WHAT IS HOMEOSTASIS? Homeostasis
Standard of Newborn Care in the Age of Birth Plans. Stephanie Deal, MD Tiffany McKee-Garrett, MD
 Standard of Newborn Care in the Age of Birth Plans Stephanie Deal, MD Tiffany McKee-Garrett, MD Disclosure We have no relevant financial relationships with the manufacturers(s) of any commercial products(s)
Standard of Newborn Care in the Age of Birth Plans Stephanie Deal, MD Tiffany McKee-Garrett, MD Disclosure We have no relevant financial relationships with the manufacturers(s) of any commercial products(s)
Hormonal Regulations Of Glucose Metabolism & DM
 Hormonal Regulations Of Glucose Metabolism & DM What Hormones Regulate Metabolism? What Hormones Regulate Metabolism? Insulin Glucagon Thyroid hormones Cortisol Epinephrine Most regulation occurs in order
Hormonal Regulations Of Glucose Metabolism & DM What Hormones Regulate Metabolism? What Hormones Regulate Metabolism? Insulin Glucagon Thyroid hormones Cortisol Epinephrine Most regulation occurs in order
during pregnancy. any degree of impaired glucose intolerance 11/19/2012 Prevalence & Diagnosis of Gestational Diabetes
 Prevalence & Diagnosis of Gestational Diabetes A.Ziaee, MD Endocrinologist Gestational diabetes mellitus (GDM); any degree of impaired glucose intolerance with onset or first recognition during pregnancy.
Prevalence & Diagnosis of Gestational Diabetes A.Ziaee, MD Endocrinologist Gestational diabetes mellitus (GDM); any degree of impaired glucose intolerance with onset or first recognition during pregnancy.
GESTATIONAL DIABETES for GP Obstetric Shared Care Accreditation Seminar. Simon Kane March 2016
 GESTATIONAL DIABETES for GP Obstetric Shared Care Accreditation Seminar Simon Kane March 2016 Objectives History and definitions Definition and Australian data Pathophysiology and prevalence Rationale
GESTATIONAL DIABETES for GP Obstetric Shared Care Accreditation Seminar Simon Kane March 2016 Objectives History and definitions Definition and Australian data Pathophysiology and prevalence Rationale
Early life influences on adult chronic
 Early life influences on adult chronic disease among aboriginal people Sandra Eades, Lina Gubhaju, Bridgette McNamara, Ibrahima Diouf, Catherine Chamberlain, Fiona Stanley University of Sydney October
Early life influences on adult chronic disease among aboriginal people Sandra Eades, Lina Gubhaju, Bridgette McNamara, Ibrahima Diouf, Catherine Chamberlain, Fiona Stanley University of Sydney October
Gestational Diabetes: Long Term Metabolic Consequences. Outline 5/27/2014
 Gestational Diabetes: Long Term Metabolic Consequences Gladys (Sandy) Ramos, MD Associate Clinical Professor Maternal Fetal Medicine Outline Population rates of obesity and T2DM Obesity and metabolic syndrome
Gestational Diabetes: Long Term Metabolic Consequences Gladys (Sandy) Ramos, MD Associate Clinical Professor Maternal Fetal Medicine Outline Population rates of obesity and T2DM Obesity and metabolic syndrome
NEONATAL HYPOXIC-ISCHAEMIC ENCEPHALOPATHY (HIE) & COOLING THERAPY
 Background NEONATAL HYPOXIC-ISCHAEMIC ENCEPHALOPATHY (HIE) & COOLING THERAPY A perinatal hypoxic-ischaemic insult may present with varying degrees of neonatal encephalopathy, neurological disorder and
Background NEONATAL HYPOXIC-ISCHAEMIC ENCEPHALOPATHY (HIE) & COOLING THERAPY A perinatal hypoxic-ischaemic insult may present with varying degrees of neonatal encephalopathy, neurological disorder and
Objectives. Diabetes in Pregnancy: A Growing Dilemma. Diabetes in the US 10/6/2015. Disclosure. The presenter has no conflicts to disclose
 Diabetes in Pregnancy: A Growing Dilemma Kathy O Connell, MN RN Perinatal Clinical Nurse Specialist University of Washington Medical Center Seattle, WA koconnll@uw.edu Objectives Describe pathophysiologic
Diabetes in Pregnancy: A Growing Dilemma Kathy O Connell, MN RN Perinatal Clinical Nurse Specialist University of Washington Medical Center Seattle, WA koconnll@uw.edu Objectives Describe pathophysiologic
8/20/12. Discuss the importance of thermoregulation in the neonate.
 Sharon Rush MSN NNP-BC Discuss the importance of thermoregulation in the neonate. To maintain correct body temperature range in order to: Reduce oxygen consumption Reduce calorie expenditure Maximize metabolic
Sharon Rush MSN NNP-BC Discuss the importance of thermoregulation in the neonate. To maintain correct body temperature range in order to: Reduce oxygen consumption Reduce calorie expenditure Maximize metabolic
Clinical evaluation Jaundice skin and mucous membranes
 JAUNDICE Framework The definition of Neonatal Jaundice Billirubin Metabolism Special characteristic in neonates Dangerous of the Hyperbillirubinemia The diseases in relation with Neonatal Jaundice Objectives:
JAUNDICE Framework The definition of Neonatal Jaundice Billirubin Metabolism Special characteristic in neonates Dangerous of the Hyperbillirubinemia The diseases in relation with Neonatal Jaundice Objectives:
Beyond the Naked Eye: A Case Presentation on a Rare Form of Congenital Hyperinsulinism (HI) Patient Demographics 5/12/2016
 Beyond the Naked Eye: A Case Presentation on a Rare Form of Congenital Hyperinsulinism (HI) Pediatric Endocrine Nursing Society May 14, 2016 Enyo Dzata, MSN, CRNP Congenital Hyperinsulinism Center Division
Beyond the Naked Eye: A Case Presentation on a Rare Form of Congenital Hyperinsulinism (HI) Pediatric Endocrine Nursing Society May 14, 2016 Enyo Dzata, MSN, CRNP Congenital Hyperinsulinism Center Division
What systems are involved in homeostatic regulation (give an example)?
 1 UNIVERSITY OF PNG SCHOOL OF MEDICINE AND HEALTH SCIENCES DIVISION OF BASIC MEDICAL SCIENCES DISCIPLINE OF BIOCHEMISTRY AND MOLECULAR BIOLOGY GLUCOSE HOMEOSTASIS (Diabetes Mellitus Part 1): An Overview
1 UNIVERSITY OF PNG SCHOOL OF MEDICINE AND HEALTH SCIENCES DIVISION OF BASIC MEDICAL SCIENCES DISCIPLINE OF BIOCHEMISTRY AND MOLECULAR BIOLOGY GLUCOSE HOMEOSTASIS (Diabetes Mellitus Part 1): An Overview
Managing Diabetes Before, During, and After Pregnancy
 Managing Diabetes Before, During, and After Pregnancy This program is supported by an educational grant from Novo Nordisk Inc. It has been accredited by AADE for pharmacists, nurses, and dietitians. This
Managing Diabetes Before, During, and After Pregnancy This program is supported by an educational grant from Novo Nordisk Inc. It has been accredited by AADE for pharmacists, nurses, and dietitians. This
Metabolic Programming. Mary ET Boyle, Ph. D. Department of Cognitive Science UCSD
 Metabolic Programming Mary ET Boyle, Ph. D. Department of Cognitive Science UCSD nutritional stress/stimuli organogenesis of target tissues early period critical window consequence of stress/stimuli are
Metabolic Programming Mary ET Boyle, Ph. D. Department of Cognitive Science UCSD nutritional stress/stimuli organogenesis of target tissues early period critical window consequence of stress/stimuli are
ETIOLOGY AND PATHOGENESIS OF HYPOXIC-ISCHEMIC ENCEPHALOPATHY
 ETIOLOGY AND PATHOGENESIS OF HYPOXIC-ISCHEMIC ENCEPHALOPATHY HYPOXIC-ISCHEMIC ENCEPHALOPATHY Hypoxic-İschemic Encephalopathy Encephalopathy due to hypoxic-ischemic injury [Hypoxic-ischemic encephalopathy
ETIOLOGY AND PATHOGENESIS OF HYPOXIC-ISCHEMIC ENCEPHALOPATHY HYPOXIC-ISCHEMIC ENCEPHALOPATHY Hypoxic-İschemic Encephalopathy Encephalopathy due to hypoxic-ischemic injury [Hypoxic-ischemic encephalopathy
When is Risky to Apply Oxygen for Congenital Heart Disease 부천세종병원 소아청소년과최은영
 When is Risky to Apply Oxygen for Congenital Heart Disease 부천세종병원 소아청소년과최은영 The Korean Society of Cardiology COI Disclosure Eun-Young Choi The author have no financial conflicts of interest to disclose
When is Risky to Apply Oxygen for Congenital Heart Disease 부천세종병원 소아청소년과최은영 The Korean Society of Cardiology COI Disclosure Eun-Young Choi The author have no financial conflicts of interest to disclose
ADMISSION/DISCHARGE FORM FOR INFANTS BORN IN 2019 DO NOT mail or fax this form to the CPQCC Data Center. This form is for internal use ONLY.
 1 Any eligible inborn infant who dies in the delivery room or at any other location in your hospital within 12 hours after birth and prior to admission to the NICU is defined as a "Delivery Room Death."
1 Any eligible inborn infant who dies in the delivery room or at any other location in your hospital within 12 hours after birth and prior to admission to the NICU is defined as a "Delivery Room Death."
11/8/12. KERNICTERUS: The reason we have to care about bilirubin. MANAGING JAUNDICE IN THE BREASTFEEDING INFANT AKA: Lack of Breastfeeding Jaundice
 MANAGING JAUNDICE IN THE BREASTFEEDING INFANT AKA: Lack of Breastfeeding Jaundice November 16, 2012 Orange County Lawrence M. Gartner, M.D. University of Chicago and Valley Center, California KERNICTERUS:
MANAGING JAUNDICE IN THE BREASTFEEDING INFANT AKA: Lack of Breastfeeding Jaundice November 16, 2012 Orange County Lawrence M. Gartner, M.D. University of Chicago and Valley Center, California KERNICTERUS:
INTRAVENOUS FLUIDS PRINCIPLES
 INTRAVENOUS FLUIDS PRINCIPLES Postnatal physiological weight loss is approximately 5-10% Postnatal diuresis is delayed in Respiratory Distress Syndrome (RDS) Preterm babies have limited capacity to excrete
INTRAVENOUS FLUIDS PRINCIPLES Postnatal physiological weight loss is approximately 5-10% Postnatal diuresis is delayed in Respiratory Distress Syndrome (RDS) Preterm babies have limited capacity to excrete
Prevention and Management of Hypoglycaemia
 Prevention and Management of Hypoglycaemia Use for ALL babies on the Labour Ward, Postnatal Ward & Transitional Care. This guideline is based on the BAPM Hypoglycaemia Guideline 2017. This guideline should
Prevention and Management of Hypoglycaemia Use for ALL babies on the Labour Ward, Postnatal Ward & Transitional Care. This guideline is based on the BAPM Hypoglycaemia Guideline 2017. This guideline should
Guslihan Dasa Tjipta Division of Perinatology Department of Child Health Medical School University of Sumatera Utara
 Guslihan Dasa Tjipta Division of Perinatology Department of Child Health Medical School University of Sumatera Utara 1 Definition Perinatal asphyxia is a fetus/newborn, due to: is an insult to the Lack
Guslihan Dasa Tjipta Division of Perinatology Department of Child Health Medical School University of Sumatera Utara 1 Definition Perinatal asphyxia is a fetus/newborn, due to: is an insult to the Lack
Pediatric Toxic Hypoglycemia. Sara Kazim, MD, FRCP (EM) Clinical Pharmacology and Medical Toxicology Fellowship IEMC May Antalya
 Pediatric Toxic Hypoglycemia Sara Kazim, MD, FRCP (EM) Clinical Pharmacology and Medical Toxicology Fellowship IEMC May 2016 - Antalya Conflicts of Interests... None Learning Needs... By the end of this
Pediatric Toxic Hypoglycemia Sara Kazim, MD, FRCP (EM) Clinical Pharmacology and Medical Toxicology Fellowship IEMC May 2016 - Antalya Conflicts of Interests... None Learning Needs... By the end of this
Neonatal Seizure. Dr.Nawar Yahya. Presented by: Sarah Khalil Zeina Shamil Zainab Waleed Zainab Qahtan. Supervised by:
 Neonatal Seizure Supervised by: Dr.Nawar Yahya Presented by: Sarah Khalil Zeina Shamil Zainab Waleed Zainab Qahtan Objectives: What is neonatal seizure Etiology Clinical presentation Differential diagnosis
Neonatal Seizure Supervised by: Dr.Nawar Yahya Presented by: Sarah Khalil Zeina Shamil Zainab Waleed Zainab Qahtan Objectives: What is neonatal seizure Etiology Clinical presentation Differential diagnosis
DIABETES AND PREGNANCY. CDE Exam Preparation March 22 & 27, 2018 Presented by Wendy Graham RD CDE Mentor
 DIABETES AND PREGNANCY CDE Exam Preparation March 22 & 27, 2018 Presented by Wendy Graham RD CDE Mentor OBJECTIVES Describe targets for blood glucose in pregnancy Discuss the risks to baby if blood glucose
DIABETES AND PREGNANCY CDE Exam Preparation March 22 & 27, 2018 Presented by Wendy Graham RD CDE Mentor OBJECTIVES Describe targets for blood glucose in pregnancy Discuss the risks to baby if blood glucose
EU RISK MANAGEMENT PLAN (EU RMP) Nutriflex Omega peri emulsion for infusion , version 1.1
 EU RISK MANAGEMENT PLAN (EU RMP) Nutriflex Omega peri emulsion for infusion 13.7.2015, version 1.1 III.1. Elements for a Public Summary III.1.1. Overview of disease epidemiology Patients may need parenteral
EU RISK MANAGEMENT PLAN (EU RMP) Nutriflex Omega peri emulsion for infusion 13.7.2015, version 1.1 III.1. Elements for a Public Summary III.1.1. Overview of disease epidemiology Patients may need parenteral
