Frequently Asked Questions: Iodinated Contrast Agents 1
|
|
|
- Buck McKenzie
- 5 years ago
- Views:
Transcription
1 CURRENT PRACTICE ISSUES Frequently Asked Questions: Iodinated Contrast Agents 1 Michael A. Bettmann, MD Although iodinated contrast agents are safe and widely used, adverse events occur and questions remain about their use, safety, and interactions. Some questions are easily answered and others still require extensive investigation. For one frequent question is informed consent necessary before all contrast media injections the simple answer is no. Another question concerns use of contrast media in patients with prior reactions or allergies. Contrast agents can be safely used in such patients, but special care must be taken to be aware of what the previous reaction was and to be ready to treat any reaction. The protective role of pre-treatment with steroids is well established for minor reactions, but they may not prevent major reactions. It is important to realize that even life-threatening, anaphylactoid reactions are not the result of a true allergy to contrast media. Many questions arise about contrast agent induced nephropathy. Baseline serum creatinine values should be obtained in patients who are at risk, not all patients. The incidence and natural history of contrast agent induced nephropathy remain unclear. It occurs only in patients with compromised renal function before contrast agent injection, but even patients with normal serum creatinine levels can have renal dysfunction. Calculated creatinine clearance is a better way to determine risk and to follow this complication. The outcome in almost all patients is benign, with progression to end-stage renal disease being rare. The major risk factors, in addition to renal dysfunction, are long-standing diabetes mellitus, dehydration, and use of other nephrotoxic medications. Recent work in preventing and ameliorating contrast agent induced nephropathy with N-acetyl cysteine, substitution of an isosmolal nonionic contrast agent, and various hydration regimens has been promising. Another common concern is use of iodinated contrast agents in pregnant or breast-feeding women. In both cases, there is no evidence of harm to the fetus or infant, but it is prudent to weigh the theoretical risks and benefits and avoid contrast agent administration unless it is truly necessary. RSNA, 2004 S3 Abbreviation: n-ac N-acetyl cysteine Index terms: Contrast media Radiology and radiologists, socioeconomic issues 2004; 24:S3 S10 Published online /rg.24si Content Codes: 1 From the Department of Radiology, New York Methodist Hospital, Brooklyn, NY. Received June 3, 2004; revision requested June 23 and received July 23; accepted July 26. The author has received grant support from GE Healthcare Group and Guerbet, and has been a consultant for Bracco Diagnostics and Berlex Imaging. Address correspondence to the author, 453 Sixth St, Brooklyn, NY ( mab9099@nyp.org). RSNA, 2004
2 S4 October 2004 RG f Volume 24 Special Issue Introduction Every radiologist is intimately familiar with contrast agents, and, with the increasing use of computed tomography (CT), the number of patients receiving these medications continues to increase. This widespread use has helped engender improved understanding of contrast agents, but many questions and misunderstandings remain. The intent of this article is to pragmatically address some of the main concerns that arise with the day-to-day use of iodinated contrast agents. These include the need for informed consent and for measurement of renal function, definition of risk factors that may predict adverse events, how to deal with patients who have had a prior reaction, and how to assess for and deal with contrast nephrotoxicity. Do I Have to Obtain Consent before Contrast Material Injection? The simple answer is no, but that is probably too simple. If there were no risks associated with injection of contrast agents, or if the risks were vanishingly small, there would be no need for consent. Conversely, if there were high risk, perhaps risk of death in one of 100 or even one of 1,000 exposures, informed consent would be mandatory. The actual risk of death, however, is very low, less than one in 130,000 at most (1,2). The risk of other severe adverse events, such as renal failure or stroke, is also very low. An additional major consideration is that it is becoming almost physically impossible for a physician to obtain consent for every contrast agent injection, given the increasing number of CT examinations being performed. If a lawyer were asked the above question, the answer would almost invariably be yes. The definition of malpractice, however, is based on usual and customary practice, and clearly many radiologists, probably a majority, do not obtain formal informed consent. As with many areas in the practice of medicine, there are no absolute guidelines that pertain. The compromise that is often used is an information sheet (3). It is important to remember that informed consent is a process, one that is intended to provide information to both the patient and the physician. To this end, most such information sheets ask the patient to answer certain questions that help define a patient s risk, such as diabetic and renal function status, prior reaction to contrast agents (if any had been administered previously), and history of allergies. The questions are followed by a short paragraph that explains the risks that may be associated with contrast agent injection, including the very small risk of death or cerebrovascular accident. The patient is also informed that if he or she has any questions related to contrast agent use, a radiologist will be made available to provide further information. In summary, the injection of an iodinated contrast agent is very safe, particularly in comparison to other situations in which consent is usually obtained, such as surgery or angiography. Usual and customary practice the practice of at least a reasonable minority of responsible radiologists is to not obtain informed consent. The purpose of consent is to gather and impart relevant information to allow the procedure to be as safe and comfortable as possible. A straightforward, one-page information sheet is a practical, reasonable, and widely used approach. How Should I Deal with a Patient Who Is Allergic to Contrast Agents? First, although typical adverse reactions encountered after the administration of an iodinated contrast agent resemble allergic reactions, with signs ranging from urticaria to an anaphylactoid process, such reactions are not allergies. Contrast agent molecules are small, with a molecular weight of about 850 kd, and are probably too small to be able to act as antigens. Second, despite extensive investigations, no antibodies to contrast agents have been reliably identified in patients who have had systemic, allergylike reactions. Finally, there is extensive experience indicating that a patient who had an allergylike adverse event after contrast agent injection is unlikely to experience a similar or more severe reaction if contrast agent were injected again (1). In fact, although a prior reaction remains the best predictor of a future adverse event, the likelihood of a recurrent reaction is in the range of 8% 25% (1). If the previous reaction were caused by a true allergy, the risk would approach 100%. If the Patient Had a Prior Reaction to a Contrast Agent, Should I Ignore It, Begin a Pre- Treatment Regimen, or Avoid Any Further Contrast Agent Injection? There is no simple answer. Many different approaches have been suggested. It is not necessary to avoid any further contrast agent injection in most cases. A very small number of patients, anecdotally, have recurrent reactions, which seem to occur after various types of contrast agents and which are not preventable with a pre-treatment regimen. In most cases, these adverse effects are
3 RG f Volume 24 Special Issue Bettmann S5 delayed cutaneous reactions (discussed later) that may be severe but are not life threatening. Very rarely, recurrent life-threatening reactions are reported to occur, but these cases are both rare and poorly documented. Use of a different contrast agent is a reasonable approach, although it is often impossible to define either the specifics of a prior reaction or the specific contrast agent used. Obtaining a careful patient history is helpful in this regard. If the patient s reaction occurred in the United States before 1985, a nonionic, low-osmolality agent could not have been used, so one could be used now. If the reaction occurred before 1997, iodixanol, the nonionic isotonic dimer, was not available, so it could be substituted. Regardless, the two crucial factors are the awareness of a prior reaction and the ready availability of equipment and expertise to treat any reaction that may occur. The use of a pre-treatment regimen, including administration of corticosteroids with or without antihistamines or other medications, has been proved safe (4,5). It has also been proved effective, but with the major caveat that it is effective in preventing only minor adverse events. Prospective studies of pre-treatment have been too small to be able to determine whether pre-treatment with steroids prevents the life-threatening reactions (4,6). There is evidence from a large prospective registry, however, that they do not (1). If pre-treatment is to be used, the crucial points to remember are that it almost certainly won t hurt, that steroid use must begin about 12 hours before contrast agent injection to prevent even minor adverse events (4), and that the attending radiologist and other personnel must still be ready to treat a reaction. In summary, unless the patient has a clearly documented history of multiple significant prior reactions and this is very rare there is no need to refrain from injecting iodinated contrast media in a patient who has had a prior reaction. Pretreatment with steroids almost certainly won t harm the patient, but it will delay the procedure at least 12 hours, and it is unlikely to prevent a severe reaction. Use of a contrast agent that the patient has not been exposed to previously may help, but being ready and able to treat a lifethreatening reaction is the crucial element (7). If a Patient Has Other Allergies or Asthma, Does It Predict an Increased Risk of an Adverse Event? It does, but minimally. The presence of a history of multiple severe allergies does increase the risk of a reaction following contrast agent injection, but only by a very small percent (1). Further, most such reactions will be minor, as are most reactions in general. A strong history of allergies should increase awareness of the possible risks of a reaction, but there is no need to avoid injection of contrast media. A history of asthma has also been thought to be a good predictor of increased risk. Empirically, asthma appears to increase the risk only of bronchospasm, not other adverse events. The risk, again empirically, seems to be particularly high in patients with active asthma. Fortunately, such patients are generally familiar with the use of -agonist inhalers and have one with them. Nevertheless, such inhalers should always be available where contrast media are being administered. In the past, many thought that an allergy to shellfish was a predictor of an increased risk of a reaction to a contrast agent, presumably because both shellfish and contrast agents contain iodine. Such a relationship is clearly not true. Organic iodide as found in shellfish is an essential element, so individuals cannot be allergic to it. Should I Obtain Serum Urea Nitrogen and Creatinine Values for Every Patient before I Inject Contrast Media? Not every patient is at risk of developing contrast agent induced nephropathy, so recent values of serum urea nitrogen (ie, BUN) and creatinine are not needed in every patient. As will be discussed, the risk is confined to patients with underlying compromise of normal renal function (8). If a patient has known renal dysfunction, it is important to have recent measurements of serum urea nitrogen and creatinine (eg, within the preceding month) to make sure that the patient s renal function is stable. For all other patients, it is important to find out if they have risk factors for renal problems. These factors include diabetes mellitus; history of recurrent renal stones or recurrent urinary tract infections; history of bladder outlet obstruction (eg, benign prostatic hypertrophy or bladder prolapse); recent history of an event known to cause a risk of renal damage, such as major surgery, dehydration, or cardiogenic shock; and use of nephrotoxic medications such as gentamycin, high-dose nonsteroidal anti-inflammatory drugs, or certain chemotherapeutic drugs. It is not clear whether the risk related to diabetes mellitus is different for types 1 and 2. The risk is probably more a function of the duration of the diabetes and the ongoing control of blood sugar level. In general, it is better to err on the side of caution and obtain a serum urea nitrogen value (as a rough guide to the level of hydration) and a creatinine value (as an indicator of whether there
4 S6 October 2004 RG f Volume 24 Special Issue is true renal dysfunction). The frequency with which contrast agent enhanced examinations must be delayed because this information is missing clearly depends on the patient population, the usual approach of the referring doctors, and the protocols of the radiology department. If patients are well screened and if referring physicians are properly informed, contrast-enhanced imaging studies most likely will need to be delayed infrequently. What about Patients Who Take Metformin? Patients who take metformin or metformin-containing medications present a different challenge for radiologists. If a patient s blood levels of metformin are high, the patient has a risk of developing lactic acidosis, which has been reported to be fatal in about 50% of cases (9). Metformin is excreted unchanged by the kidney; therefore, people with compromised renal function will have higher than expected serum levels and are at risk of developing lactic acidosis. In addition, individuals with very poor cardiac function or acute hepatic compromise have increased lactate production or impaired lactate breakdown and also are at risk of developing lactic acidosis. For all such patients, the use of metformin is contraindicated. Because contrast agents are associated with nephrotoxicity, it was postulated that the concomitant use of metformin and an iodinated contrast agent could lead to an increased risk of lactic acidosis. This hypothesis has been disproved empirically, and the package insert approved by the U.S. Food and Drug Administration has been altered accordingly. Although the package insert is perhaps overly cautious, it states that metformin use should be stopped at the time an iodinated contrast agent is administered and that the patient should wait 48 hours before resuming use of metformin. There is no mandate to measure serum creatinine levels at that time. Rather, the patient should be reevaluated clinically, and a creatinine value should be obtained if there are any other reasons that the patient s renal function may have been compromised, such as major surgery or cardiogenic shock (7). The major point to remember is that metformin is contraindicated in patients with any compromise in renal function. It is surprising how often patients who take metformin and who have elevated serum creatinine levels are referred for a contrast agent study. In these patients, the imaging study should be delayed and the referring physician should be notified. How Should I Deal with a Woman Who Is Pregnant or Breast-Feeding? These concerns have been addressed by the American College of Radiology in its recently released fifth edition of the Manual on Contrast Media (10). Because contrast media are small molecules that are rapidly distributed throughout the extracellular space, they must be assumed to readily cross the placental barrier. Further, contrast media molecules are most likely present in breast milk shortly after parenteral administration to a breast-feeding patient. There is no evidence suggesting that iodinated contrast agents are teratogenic in humans. On the other hand, the experience is too limited to conclude that they are safe. It is, therefore, wise to avoid administering them in women who are pregnant (particularly in the first trimester) when possible. It is appropriate, however, to use contrast agents when the procedure requiring them is thought to be essential. That is, the risks, as always, must be balanced against the possible benefits. Overall, the theoretical risks of radiation exposure are most likely greater than those incurred from contrast agents. In breast-feeding women as well, the risks to the patient and to her infant must be weighed against any possible benefits. If an iodinated contrast agent is administered, the infant is likely to receive a small amount orally and then to absorb a small amount. There is no real evidence of contrast agent induced toxicity in newborns, but on the other hand there is little concrete information confirming the safety of these medications in infants. A further concern regarding the use of iodinated contrast agents in both pregnant and breast-feeding women is the theoretical effect of these agents on the fetal or neonatal thyroid. Although in theory all of the iodine in contrast agents is organically bound rather than free, it is not clear that no free iodide is present or to what extent there is thyroid uptake in the fetus or newborn and what the effect on thyroid function might be. The consensus recommendation is that a breast-feeding mother should use a pump to remove breast milk before contrast agent administration, and then afterward, she should use the pump and discard breast milk for hours before resuming normal breast-feeding. Since the biologic half-life of iodinated contrast agents is less that 60 minutes, the amount remaining in the mother (assuming her renal function is normal) after 12 hours is essentially undetectable. What Is Contrast Agent induced Nephropathy? How Is It Defined and How Often Does It Occur? In the most general terms, contrast agent induced nephropathy is the occurrence of renal failure, as indicated by an increase in serum creati-
5 RG f Volume 24 Special Issue Bettmann S7 nine level or a fall in calculated creatinine clearance, after the administration of an iodinated contrast agent. For several reasons, there is no widely accepted definition of contrast agent induced nephropathy. First, such nephropathy occurs essentially only in those patients who have abnormal renal function before contrast agent injection. Second, the parameters used clinically are imprecise. That is, serum creatinine levels vary with age, muscle mass, and gender. A serum creatinine value of 1.2 mg/dl in a healthy 20- year-old man or woman is normal and indicates that the glomerular filtration rate is in the normal range of ml/min, most likely close to or above 100 ml/min. The same serum creatinine value in a 50-kg, 80-year-old woman, however, likely correlates with a significantly decreased glomerular filtration rate, perhaps even less than 40 ml/min. This difference is because glomerular filtration rate decreases with increasing age, and creatinine production, a function of muscle mass, also decreases. In short, the serum creatinine value is a reasonable screening parameter, since it is inexpensive and readily available, but it is not particularly accurate. It is not practical to measure true creatinine clearance, but it is helpful to use a formula, such as that of Cockcroft and Gault (11) or Levey et al (12), to calculate creatinine clearance. In fact, some institutional laboratories routinely report calculated creatinine clearance with serum creatinine levels. Most studies of contrast agent induced nephropathy have used serum creatinine values, at baseline and after contrast agent administration. Because of the limitations of serum creatinine, these studies have shown widely varying results. Contrast agents have been cited as one of the most frequent causes of in-hospital renal failure (13,14), which is probably a valid impression, but many other factors must be considered. These factors include not only real glomerular filtration rate, but also concomitant risk factors such as dehydration, surgery, and use of additional nephrotoxins (eg, gentamycin, nonsteroidal anti-inflammatory drugs, certain chemotherapeutic drugs). At this time, it is impossible to define the true incidence of contrast agent induced nephropathy. What Are the Risk Factors for Contrast Agent induced Nephropathy? As previously implied, the major risk factor is underlying renal dysfunction. Contrast agent induced nephropathy essentially never occurs if a patient s renal function is truly normal. The presence of diabetes, particularly if long standing, is probably not an independent risk factor, but it is a major contributing factor in the presence of renal dysfunction (8,15). Additional concerns, as noted, are dehydration, poor renal perfusion (as occurs with severe congestive heart failure), and the presence of other factors that may be nephrotoxic, such as certain medications or major surgery. The volume of contrast agent administered is another dependent risk factor: If renal function is truly normal (the 20-year-old healthy patient with a serum creatinine level of 1.2 mg/dl), then a high volume of contrast agent will not lead to nephropathy. If the patient has underlying renal dysfunction, however, increasing the volume of contrast agent injected will increase the likelihood of worsening renal function (16). What Is the Natural History of Contrast Agent induced Nephropathy? In the vast majority of cases, contrast agent induced nephropathy will be manifest by a transient increase in serum creatinine level, which usually peaks at 4 7 days and gradually returns to baseline. A persistent elevation of serum creatinine level is unusual, as is progression to end-stage renal disease (8,17). The risk of either of these endpoints increases as a direct function of the severity of a patient s underlying renal failure; that is, most patients who require dialysis after contrast agent injection had very poor renal function at baseline. In one study of over 600 patients with elevated serum creatinine levels who underwent cardiac catheterization, only seven patients required dialysis, and permanent dialysis was necessary in only three of these (8). How Can Contrast Agent induced Nephropathy Be Prevented? First, nephropathy is not really a worry in patients with truly normal renal function, regardless of the volume of contrast agent injected. For patients at risk, the first step in preventing nephropathy is adequate hydration (18). In practical terms, adequate hydration is usually achieved by instructing patients to drink several liters of fluid over hours before contrast agent injection. When such hydration isn t possible because an imaging study is urgent, because the patient cannot take oral fluids, or because sedation or anesthesia will be needed and thus oral fluids are contraindicated, intravenous hydration can be given and may actually be more effective. Intravenous hydration should consist of at least 1 ml of normal saline per kilogram of body weight per hour, beginning 12 hours before contrast agent injection and continuing for 12 hours afterward. It is also important to remember that, invariably, patients do not adequately hydrate themselves; thus,
6 S8 October 2004 RG f Volume 24 Special Issue for patients at risk of nephropathy and if it is feasible, intravenous hydration should be used, beginning at least 12 hours before contrast agent injection (19). There is evidence that normal saline is more effective than is half-normal saline (20). Recently, the use of sodium bicarbonate for hydration, as compared with sodium chloride (normal saline), has been reported as being effective in decreasing the incidence of contrast agent induced nephropathy (21). This approach is appealing because of its low cost and relative ease of use, but further proof of efficacy is necessary. One concern with the Merten et al study (21) is that hydration consisted of a 7-hour infusion begun 1 hour before contrast agent injection. Other studies have shown that hydration with an infusion of normal saline that begins at least 12 hours before contrast agent injection is more effective than either short infusions or random oral hydration (19). Many other approaches for preventing contrast agent induced nephropathy have been investigated. It is logical to think that administration of either a diuretic such as furosemide or dialysis during or immediately after contrast agent injection would help. Furosemide has been shown to be not helpful (18). Results of studies with dialysis do not agree, but most suggest that dialysis also is not helpful (22,23). Use of fenoldapam mesylate, a selective dopamine 1 -receptor agonist (thus a post glomerular renal vasodilator), has been advocated, but the bulk of the evidence suggests it does not have any protective or ameliorating effect (24), and its use requires close monitoring. Various other vasoactive agents have been investigated, mainly in small clinical studies, without any real proof of efficacy. Two agents that have been considered promising, although neither has been conclusively proved to be effective, are N-acetyl cysteine (n-ac) and iodixanol (Visipaque; GE Healthcare Group, Princeton, NJ), the only available nonionic, isosmolal, iodinated contrast agent. N-AC acts as both a vasodilator and as a free radical scavenger. Contrast agent nephrotoxicity was believed to be caused by a prolonged decrease in glomerular flow, so enhancing flow (as with n-ac or fenoldapam) might be protective. These hemodynamic changes have been demonstrated repeatedly in animal studies, but in humans, injection of contrast agent does not lead to a significant decrease in renal blood flow. It is well known, however, that the renal medulla is poorly oxygenated and is close to hypoxic under normal conditions. If hypoxia occurs, reactive oxygen species that are toxic to cells are produced. In theory, then, n-ac may be effective because it is a free radical scavenger (25). The studies performed to date and analyses of them reach conflicting conclusions (26 28). Most but not all studies that used either oral administration with four doses twice daily starting on the day before contrast agent injection or intravenous administration starting 30 minutes before injection have shown a protective effect. In addition, n-ac is safe, readily available, and inexpensive. Iodixanol has been shown to have a protective effect in two relatively small studies (29,30) and in other empiric reports. The reason why iodixanol should be effective is not entirely clear. To step back a step, or a generation of contrast agents, the bulk of the evidence suggests that there is a difference, probably minor, in the incidence of contrast agent induced nephropathy when high-osmolality versus low-osmolality contrast agents are used, but no difference between them in the need for dialysis (8). It may be that the same holds true for iodixanol: The likelihood of a rise in serum creatinine level may be decreased, but the outcome for patients may not be different. Thus, iodixanol is promising, but further studies are needed to prove that it is effective in preventing or limiting contrast agent induced nephropathy. Many additional approaches have been used to prevent or at least to minimize the incidence and severity of nephropathy, and other larger studies are necessary to answer the persistent and clinically important questions that remain. Is There a Level of Serum Creatinine above Which a Patient Should Not Receive an Iodinated Contrast Agent? The short answer is no. In all patients, regardless of whether renal function is normal, the potential risks must be balanced against the possible benefits. A serum creatinine level of 2.0 mg/dl, or 3.0 or 1.8, does not by itself indicate what the glomerular filtration rate is, although it is clearly reduced, nor does it accurately predict the risk of increased morbidity or mortality after injection of iodinated contrast media. From a practical point of view, the radiologist should first calculate the creatinine clearance and then determine whether the relevant diagnostic question can be as adequately answered with another examination that does not require injection of an iodinated contrast agent. For example, if a patient is strongly suspected of having pulmonary embolism and has a mildly elevated creatinine level, but cannot undergo magnetic resonance angiography because of a pacemaker, what is the risk of performing CT angiography, which is likely to yield definitive results, versus a ventila-
7 RG f Volume 24 Special Issue Bettmann S9 tion perfusion lung scan? If the patient is stable and at low risk for anticoagulation, it is reasonable to do a ventilation perfusion scan, at least initially. If, however, a rapid and definitive answer is needed (eg, if the patient is hemodynamically compromised), CT pulmonary angiography should be performed. It is likely to cause no more than a transient increase in creatinine level while providing a definitive answer. In such a situation, it is probably better to start hydration (with normal saline or perhaps with sodium bicarbonate), start intravenous n-ac, and perform CT angiography. Remember, the risks related to contrast agent induced nephropathy primarily depend on the baseline renal function and concomitant factors that place the patient at risk, such as dehydration, congestive heart failure, and recent major surgery. In most cases in which the patient has an elevated serum creatinine level (even if it is fairly markedly elevated), his or her renal function, as reflected in serum creatinine values, will likely worsen but then return to baseline. What about Delayed Reactions to Contrast Agents? Delayed reactions have been reported, with a low frequency, for many years (31,32). The reports have increased with the use of nonionic dimeric contrast media (33). Such reactions are almost invariably cutaneous, with a maculopapular, erythematous eruption occurring hours after contrast agent administration. Although the rash is usually self-limited and only mildly symptomatic, it can be very uncomfortable and can spread over the entire body over the course of a few days. If the rash is localized and mildly pruritic, a topical steroid can be used. If it continues to spread and is more bothersome, the patient should be referred to a dermatologist. Such reactions have been reported to recur, even when a different contrast agent is used (34), but usually they do not. They also do not appear to presage other contrast agent reactions. Summary Contrast agents continue to cause concern among patients, referring physicians, and radiologists because of their widespread use and the rare but potentially important adverse events associated with them. The questions that arise concerning contrast agents have changed over time, with changes in the formulations and consequent improvement in safety. Mortality still occurs after injection of contrast agents of all types, but it is very rare. The major questions associated with use of contrast media currently concern practical matters, such as how to appropriately inform patients about the risks and benefits of contrast agent use, how to deal with patients who have particular risk factors for an adverse event (eg, prior reaction, strong history of allergies, compromised renal function, diabetes mellitus), and how to deal with concerns of nephrotoxicity. Obtaining formal informed consent is both impractical and unnecessary, but it is imperative that radiologists get information from patients before contrast agent injection and supply information to them regarding the nature of the contrast media. Anaphylactoid reactions are not true allergies and are fortunately rare, but unfortunately can occur unpredictably. The crucial factor in dealing with them is to be prepared to treat them whenever and wherever they occur. Such preparation requires the presence of appropriate resuscitation equipment and medications and the availability of fully trained personnel. Contrast agent nephrotoxicity is a topic of interest currently because of the aging population, the associated increase in the number of patients with compromised renal function, and the increasing use of contrast-enhanced examinations. It is encouraging that there are several promising approaches to decreasing the incidence and severity of this problem, although neither the efficacy of these treatments nor the natural history of contrast agent induced nephropathy is yet clear. The most important measure currently is ensuring that hydration is adequate. At this time, the approach that is best documented as effective is intravenous normal saline infused at 1 ml/kg/hr for 12 hours before and 12 hours after contrast agent injection. A 7-hour infusion of sodium bicarbonate beginning 1 hour before injection is promising, but the method has not yet been conclusively proved to be effective. It is encouraging that contrast agents have become safer and more thoroughly understood over the past few years, but many questions and concerns remain. References 1. Bettmann MA, Heeren T, Greenfield A, Goudey C. Adverse events with radiographic contrast agents: results of the SCVIR Contrast Agent Registry. Radiology 1997; 203: Katayama H, Yamaguchi K, Kozuka T, Takashima T, Seez P, Matsuura K. Adverse reactions to ionic and nonionic contrast media: a report from the Japanese Committee on the Safety of Contrast Media. Radiology 1990; 175: Bush WH. Informed consent for contrast media (opinion). AJR Am J Roentgenol 1989; 152: Lasser EC, Berry CC, Talner LB, et al. Pre-treatment with corticosteroids to alleviate reactions to intravenous contrast material. N Engl J Med 1987; 317:
8 S10 October 2004 RG f Volume 24 Special Issue 5. Greenberger PA, Patterson R. The prevention of immediate generalized reactions to radiocontrast media in high risk patients. J Allergy Clin Immunol 1991; 87: Lasser EC, Berry CC, Mishkin MM, Williamson B, Zheutlin N, Silverman JM. Pretreatment with corticosteroids to prevent adverse reactions to nonionic contrast media. AJR Am J Roentgenol 1994; 162: American College of Radiology. American College of Radiology manual on contrast media. 4th ed. Reston, Va: American College of Radiology, Rudnick MR, Goldfarb S, Wexler L, et al. Nephrotoxicity of ionic and nonionic contrast media in 1196 patients: a randomized trial. Kidney Int 1995; 47: Bailey CJ, Turner RC. Metformin. N Engl J Med 1996; 334: American College of Radiology Committee on Drugs and Contrast Media. Manual on contrast media. 5th ed. Reston, Va: American College of Radiology, Cockcroft DW, Gault MH. Prediction of creatinine clearance from serum creatinine. Nephron 1976; 16: Levey AS, Bosch JP, Lewis JB, Greene T, Rogers N, Roth T. A more accurate method to estimate glomerular infiltration rate from serum creatinine: a new prediction equation. Ann Intern Med 1999; 130: Shusterman N, Strom BL, Murray TG, Morrison G, West SL, Maislin G. Risk factors and outcome of hospital-acquired acute renal failure: clinical epidemiologic study. Am J Med 1987; 83: Levy EM, Viscoli CM, Horwitz RI. The effect of acute renal failure on mortality: a cohort analysis. JAMA 1996; 275: Parfrey PS, Griffiths SM, Barrett BJ, et al. Contrast material-induced renal failure in patients with diabetes mellitus, renal insufficiency or both: a prospective controlled study. N Engl J Med 1989; 320: Cigarroa RG, Lange RA, Williams RH, Hillis LD. Dosing of contrast material to prevent contrast nephropathy in patients with renal disease. Am J Med 1989; 86: McCullough P, Wolyn R, Rocher L, Levin R, O Neill W. Acute renal failure after coronary intervention: incidence, risk factors, and relationship to mortality. Am J Med 1997; 103: Solomon R, Werner C, Mann D, D Elia J, Silva P. Effects of saline, mannitol and furosemide on acute decreases in renal function induced by contrast agents. N Engl J Med 1994; 331: Trivedi HS, Moore H, Nasr S, et al. A randomized prospective trial to assess the role of saline hydration on the development of contrast nephrotoxicity. Nephron Clin Pract 2003; 93:C29 C Mueller C, Buerkle G, Buettner HJ, et al. Prevention of contrast media-associated nephropathy: randomized comparison of 2 hydration regimens in 1620 patients undergoing coronary angioplasty. Arch Intern Med 2002; 162: Merten GJ, Burgess WP, Gray LV, et al. Prevention of contrast-induced nephropathy with sodium bicarbonate: a randomized controlled trial. JAMA 2004; 291: Marenzi G, Marana I, Lauri G, et al. The prevention of radiocontrast-agent-induced nephropathy by hemofiltration. N Engl J Med 2003; 349: Frank H, Werner D, Lorusso V, et al. Simultaneous hemodialysis during coronary angiography fails to prevent radiocontrast-induced nephropathy in chronic renal failure. Clin Nephrol 2003; 60: Stone GW, McCoullough PA, Tumlin JA, et al. Fenoldapam mesylate for the prevention of contrast-induced nephropathy: a randomized controlled trial. JAMA 2003; 290: Tepel M, van der Giet M, Schwartzfeld C, Laufer U, Liermann D, Zidek W. Prevention of radiographic-contrast-agent-induced reductions in renal function by acetylcysteine. N Engl J Med 2000; 343: Pannu N, Manns B, Lee H, Tonelli M. Systematic review of the impact of n-acetylcysteine on contrast nephropathy. Kidney Int 2004; 65: Kshirsagar AV, Poole C, Mottl A, et al. N-acetylcysteine for the prevention of radiocontrast induced nephropathy: a meta-analysis of prospective controlled trials. J Am Soc Nephrol 2004; 15: Kay J, Chow WH, Chan TM, et al. Acetylcysteine for prevention of acute deterioration of renal function following elective coronary angiography and intervention: a randomized controlled trial. JAMA 2003; 289: Chalmers N, Jackson RW. Comparison of iodixanol and iohexol in renal impairment. Br J Radiol 1999; 72: Aspelin P, Aubry P, Fransson SG, et al. Nephrotoxic effects in high-risk patients undergoing angiography. N Engl J Med 2003; 348: McCullough M, Davies P, Richardson R. A large trial of intravenous Conray 325 and Niopam 300 to assess immediate and delayed reactions. Br J Radiol 1989; 62: Yoshikawa H. Late adverse reactions to nonionic contrast media. Radiology 1992; 183: Davidson CJ, Laskey WK, Hermiller JB, et al. Randomized controlled trial of contrast media utilization in high-risk PTCA. Circulation 2000; 101: Romano A, Artesani MC, Andriolo M, Violo M, Pettinato R, Veccholi-Scaldazza A. Effective prophylactic protocol in delayed hypersensitivity to contrast media: report of a case involving lymphocyte transformation studies with different compounds. Radiology 2002; 225:
N-acetylcysteine in the prevention of contrast agent-induced nephrotoxicity in patients undergoing computed tomography studies
 RESEARCH REPORT N-acetylcysteine in the prevention of contrast agent-induced nephrotoxicity in patients undergoing computed tomography studies Hossein Khalili, Simin Dashti-Khavidaki, Hamed Tabifar, Nasrin
RESEARCH REPORT N-acetylcysteine in the prevention of contrast agent-induced nephrotoxicity in patients undergoing computed tomography studies Hossein Khalili, Simin Dashti-Khavidaki, Hamed Tabifar, Nasrin
Prevention of Contrast induced Nephropathy
 55 th Annual Scientific Meeting of The Korean Society of Cardiology 11:50 12:10 Message from Nephrologists Dec 03, 2011 Prevention of Contrast induced Nephropathy Soo Wan Kim, MD, PhD Department of Internal
55 th Annual Scientific Meeting of The Korean Society of Cardiology 11:50 12:10 Message from Nephrologists Dec 03, 2011 Prevention of Contrast induced Nephropathy Soo Wan Kim, MD, PhD Department of Internal
Contrast Induced Nephropathy
 Contrast Induced Nephropathy O CIAKI refers to an abrupt deterioration in renal function associated with the administration of iodinated contrast media O CIAKI is characterized by an acute (within 48 hours)
Contrast Induced Nephropathy O CIAKI refers to an abrupt deterioration in renal function associated with the administration of iodinated contrast media O CIAKI is characterized by an acute (within 48 hours)
Evaluating the Efficacy of Single Daily Dose of 1200mg of N-Acetyl-Cysteine in Preventing Contrast Agent-Associated Nephrotoxicity
 ISPUB.COM The Internet Journal of Internal Medicine Volume 6 Number 1 Evaluating the Efficacy of Single Daily Dose of 1200mg of N-Acetyl-Cysteine in Preventing Contrast Agent-Associated Nephrotoxicity
ISPUB.COM The Internet Journal of Internal Medicine Volume 6 Number 1 Evaluating the Efficacy of Single Daily Dose of 1200mg of N-Acetyl-Cysteine in Preventing Contrast Agent-Associated Nephrotoxicity
Preventing Nephropathy Induced by Contrast Medium
 T h e n e w e ng l a nd j o u r na l of m e dic i n e Preventing Nephropathy Induced by Contrast Medium Brendan J. Barrett, M.B., and Patrick S. Parfrey, M.D. This Journal feature begins with a case vignette
T h e n e w e ng l a nd j o u r na l of m e dic i n e Preventing Nephropathy Induced by Contrast Medium Brendan J. Barrett, M.B., and Patrick S. Parfrey, M.D. This Journal feature begins with a case vignette
A Rapid Protocol for the Prevention of Contrast- Induced Renal Dysfunction: The RAPPID Study
 Journal of the American College of Cardiology Vol. 41, No. 12, 2003 2003 by the American College of Cardiology Foundation ISSN 0735-1097/03/$30.00 Published by Elsevier Inc. doi:10.1016/s0735-1097(03)00487-x
Journal of the American College of Cardiology Vol. 41, No. 12, 2003 2003 by the American College of Cardiology Foundation ISSN 0735-1097/03/$30.00 Published by Elsevier Inc. doi:10.1016/s0735-1097(03)00487-x
CATH LAB SYMPOSIUM 2010
 CATH LAB SYMPOSIUM 2010 Low resistance system High Pressure in Capillaries to filter plasma RBF: 1.2-1.3 L/min (25% of C.O.) Low AV difference ( shunt ) Kidney: 14 ml O2/L blood Brain: 62 ml O2/L blood
CATH LAB SYMPOSIUM 2010 Low resistance system High Pressure in Capillaries to filter plasma RBF: 1.2-1.3 L/min (25% of C.O.) Low AV difference ( shunt ) Kidney: 14 ml O2/L blood Brain: 62 ml O2/L blood
CONTRAST-INDUCED NEPHROPATHY Y HỌC CHỨNG CỨ VÀ BIỆN PHÁP DỰ PHÒNG
 CONTRAST-INDUCED NEPHROPATHY Y HỌC CHỨNG CỨ VÀ BIỆN PHÁP DỰ PHÒNG DEFINITION Reversible form of acute kidney injury that occurs soon after the administration of radiocontrast media. PATHOGENESIS RISK FACTORS
CONTRAST-INDUCED NEPHROPATHY Y HỌC CHỨNG CỨ VÀ BIỆN PHÁP DỰ PHÒNG DEFINITION Reversible form of acute kidney injury that occurs soon after the administration of radiocontrast media. PATHOGENESIS RISK FACTORS
Steroid premedication in patients with increased risk of contrast reaction: are you familiar with the latest local and international guidelines?
 Steroid premedication in patients with increased risk of contrast reaction: are you familiar with the latest local and international guidelines? Poster No.: C-1369 Congress: ECR 2014 Type: Authors: Keywords:
Steroid premedication in patients with increased risk of contrast reaction: are you familiar with the latest local and international guidelines? Poster No.: C-1369 Congress: ECR 2014 Type: Authors: Keywords:
SAFETY IN THE CATH LAB How to Minimise Contrast Toxicity
 SAFETY IN THE CATH LAB How to Minimise Contrast Toxicity Dr. Vijay Kunadian MBBS, MD, MRCP Senior Lecturer and Consultant Interventional Cardiologist Institute of Cellular Medicine, Faculty of Medical
SAFETY IN THE CATH LAB How to Minimise Contrast Toxicity Dr. Vijay Kunadian MBBS, MD, MRCP Senior Lecturer and Consultant Interventional Cardiologist Institute of Cellular Medicine, Faculty of Medical
The New England Journal of Medicine PREVENTION OF RADIOGRAPHIC-CONTRAST-AGENT INDUCED REDUCTIONS IN RENAL FUNCTION BY ACETYLCYSTEINE.
 PREVENTION OF RADIOGRAPHIC-CONTRAST-AGENT INDUCED REDUCTIONS IN RENAL FUNCTION BY ACETYLCYSTEINE MARTIN TEPEL, M.D., MARCUS VAN DER GIET, M.D., CAROLA SCHWARZFELD, ULF LAUFER, M.D., DIETER LIERMANN, M.D.,
PREVENTION OF RADIOGRAPHIC-CONTRAST-AGENT INDUCED REDUCTIONS IN RENAL FUNCTION BY ACETYLCYSTEINE MARTIN TEPEL, M.D., MARCUS VAN DER GIET, M.D., CAROLA SCHWARZFELD, ULF LAUFER, M.D., DIETER LIERMANN, M.D.,
Protocol for iv. iodine and gadolinium contrast studies
 Protocol for iv. iodine and gadolinium contrast studies Royal College of Radiologists Standard The individual administering the contrast agent must ensure that the patient understands that it is to be
Protocol for iv. iodine and gadolinium contrast studies Royal College of Radiologists Standard The individual administering the contrast agent must ensure that the patient understands that it is to be
Dr.Nahid Osman Ahmed 1
 1 ILOS By the end of the lecture you should be able to Identify : Functions of the kidney and nephrons Signs and symptoms of AKI Risk factors to AKI Treatment alternatives 2 Acute kidney injury (AKI),
1 ILOS By the end of the lecture you should be able to Identify : Functions of the kidney and nephrons Signs and symptoms of AKI Risk factors to AKI Treatment alternatives 2 Acute kidney injury (AKI),
Introduction. Hyuck-Jun Yoon, MD, and Seung-Ho Hur, MD
 ORIGINAL ARTICLE DOI 10.4070/kcj.2011.41.5.265 Print ISSN 1738-5520 / On-line ISSN 1738-5555 Copyright 2011 The Korean Society of Cardiology Open Access Determination of Safe Contrast Media Dosage to Estimated
ORIGINAL ARTICLE DOI 10.4070/kcj.2011.41.5.265 Print ISSN 1738-5520 / On-line ISSN 1738-5555 Copyright 2011 The Korean Society of Cardiology Open Access Determination of Safe Contrast Media Dosage to Estimated
Follow-up of patients with contrast-induced nephropathy
 http://www.kidney-international.org & 2006 International Society of Nephrology Follow-up of patients with contrast-induced nephropathy R Solomon 1 and B Barrett 2 1 Fletcher Allen Health Care, University
http://www.kidney-international.org & 2006 International Society of Nephrology Follow-up of patients with contrast-induced nephropathy R Solomon 1 and B Barrett 2 1 Fletcher Allen Health Care, University
JACC: CARDIOVASCULAR INTERVENTIONS VOL. 2, NO. 5, BY THE AMERICAN COLLEGE OF CARDIOLOGY FOUNDATION ISSN /09/$36.00 PUBLISHED BY ELS
 JACC: CARDIOVASCULAR INTERVENTIONS VOL. 2, NO. 5, 2009 2009 BY THE AMERICAN COLLEGE OF CARDIOLOGY FOUNDATION ISSN 1936-8798/09/$36.00 PUBLISHED BY ELSEVIER INC. DOI: 10.1016/j.jcin.2009.03.007 Ionic Low-Osmolar
JACC: CARDIOVASCULAR INTERVENTIONS VOL. 2, NO. 5, 2009 2009 BY THE AMERICAN COLLEGE OF CARDIOLOGY FOUNDATION ISSN 1936-8798/09/$36.00 PUBLISHED BY ELSEVIER INC. DOI: 10.1016/j.jcin.2009.03.007 Ionic Low-Osmolar
16/05/2018 NEFROPATIA DA MEZZO DI CONTRASTO: ANCORA UNA VECCHIA NEMICA?
 16/05/2018 NEFROPATIA DA MEZZO DI CONTRASTO: ANCORA UNA VECCHIA NEMICA? Dott. Andrea Boccatonda Università degli Studi G. d Annunzio Chieti Chi di voi non ha mai discusso con un radiologo per eseguire
16/05/2018 NEFROPATIA DA MEZZO DI CONTRASTO: ANCORA UNA VECCHIA NEMICA? Dott. Andrea Boccatonda Università degli Studi G. d Annunzio Chieti Chi di voi non ha mai discusso con un radiologo per eseguire
Metformin Hydrochloride
 Metformin Hydrochloride 500 mg, 850 mg, 500 mg LA and 750 mg LA Tablet Description Informet is a preparation of metformin hydrochloride that belongs to a biguanide class of oral antidiabetic drugs. Metformin
Metformin Hydrochloride 500 mg, 850 mg, 500 mg LA and 750 mg LA Tablet Description Informet is a preparation of metformin hydrochloride that belongs to a biguanide class of oral antidiabetic drugs. Metformin
Clodronate BE/H/PSUR/001/001 October 2011 Agreed CSP
 Clodronate BE/H/PSUR/001/001 October 2011 Agreed CSP 4. CLINICAL PARTICULARS 4.1 Therapeutic indications Intravenous use Treatment of hypercalcemia due to malignancy. Oral use Treatment of hypercalcemia
Clodronate BE/H/PSUR/001/001 October 2011 Agreed CSP 4. CLINICAL PARTICULARS 4.1 Therapeutic indications Intravenous use Treatment of hypercalcemia due to malignancy. Oral use Treatment of hypercalcemia
The Incidence Of Contrast-Induced Nephropathy Or Radiocontrast Nephropathy
 ISPUB.COM The Internet Journal of Radiology Volume 18 Number 1 The Incidence Of Contrast-Induced Nephropathy Or Radiocontrast Nephropathy K O Kragha Citation K O Kragha. The Incidence Of Contrast-Induced
ISPUB.COM The Internet Journal of Radiology Volume 18 Number 1 The Incidence Of Contrast-Induced Nephropathy Or Radiocontrast Nephropathy K O Kragha Citation K O Kragha. The Incidence Of Contrast-Induced
Contrast-Induced Nephropathy: Evidenced Based Prevention
 Contrast-Induced Nephropathy: Evidenced Based Prevention Michael J Cowley, MD, FSCAI Nothing to disclose Contrast-Induced Nephropathy (CIN) Definitions New onset or worsening of renal function after contrast
Contrast-Induced Nephropathy: Evidenced Based Prevention Michael J Cowley, MD, FSCAI Nothing to disclose Contrast-Induced Nephropathy (CIN) Definitions New onset or worsening of renal function after contrast
of developing contrast -induced
 Original Article Singapore Med 1 2009, 50 (3) : 250 Diabetic patients with normal baseline renal function are at increased risk of developing contrast -induced nephropathy post-percutaneous coronary intervention
Original Article Singapore Med 1 2009, 50 (3) : 250 Diabetic patients with normal baseline renal function are at increased risk of developing contrast -induced nephropathy post-percutaneous coronary intervention
The Prevention of Radiocontrast-Agent Induced Nephropathy by Hemofiltration
 The new england journal of medicine original article The Prevention of Radiocontrast-Agent Induced Nephropathy by Hemofiltration Giancarlo Marenzi, M.D., Ivana Marana, M.D., Gianfranco Lauri, M.D., Emilio
The new england journal of medicine original article The Prevention of Radiocontrast-Agent Induced Nephropathy by Hemofiltration Giancarlo Marenzi, M.D., Ivana Marana, M.D., Gianfranco Lauri, M.D., Emilio
5-FU & Cisplatin + Cetuximab
 5-FU & Cisplatin + Cetuximab Available for Routine Use in Not routinely commissioned, each case requires prior documented approval before offering & commencing therapy from either: a) the relevant PCT
5-FU & Cisplatin + Cetuximab Available for Routine Use in Not routinely commissioned, each case requires prior documented approval before offering & commencing therapy from either: a) the relevant PCT
ASDIN 10th Annual Scientific Meeting Final. COI Disclosure Statement. Intravenous Contrast Media: Basics
 COI Disclosure Statement There are no financial relationships or conflicts of interest to disclose with this presentation Melissa Hicks, BA, RT(R)(VI) Vascular Interventional Technologist University of
COI Disclosure Statement There are no financial relationships or conflicts of interest to disclose with this presentation Melissa Hicks, BA, RT(R)(VI) Vascular Interventional Technologist University of
Contrast Materials Patient Safety: What are contrast materials and how do they work?
 Contrast Materials Patient Safety: What are contrast materials and how do they work? Which imaging exams use contrast materials? How safe are contrast materials? How should I prepare for my imaging procedure
Contrast Materials Patient Safety: What are contrast materials and how do they work? Which imaging exams use contrast materials? How safe are contrast materials? How should I prepare for my imaging procedure
doi: /j.jemermed THE RELATIONSHIP OF RADIOCONTRAST, IODINE, AND SEAFOOD ALLERGIES: A MEDICAL MYTH EXPOSED
 doi:10.1016/j.jemermed.2009.10.014 The Journal of Emergency Medicine, Vol. 39, No. 5, pp. 701 707, 2010 Copyright 2010 Elsevier Inc. Printed in the USA. All rights reserved 0736-4679/$ see front matter
doi:10.1016/j.jemermed.2009.10.014 The Journal of Emergency Medicine, Vol. 39, No. 5, pp. 701 707, 2010 Copyright 2010 Elsevier Inc. Printed in the USA. All rights reserved 0736-4679/$ see front matter
Doreen P. Foley MS RN ANP-C Doctor of Nursing Practice Program Chamberlain College of Nursing
 Doreen P. Foley MS RN ANP-C Doctor of Nursing Practice Program Chamberlain College of Nursing This program has been developed solely for the purposes of describing the level of nurse practitioner (NP)
Doreen P. Foley MS RN ANP-C Doctor of Nursing Practice Program Chamberlain College of Nursing This program has been developed solely for the purposes of describing the level of nurse practitioner (NP)
Surviving Sepsis Campaign: International Guidelines for Management of Sepsis and Septic Shock: 2016
 Surviving Sepsis Campaign: International Guidelines for Management of Sepsis and Septic Shock: 2016 Mitchell M. Levy MD, MCCM Professor of Medicine Chief, Division of Pulmonary, Sleep, and Critical Care
Surviving Sepsis Campaign: International Guidelines for Management of Sepsis and Septic Shock: 2016 Mitchell M. Levy MD, MCCM Professor of Medicine Chief, Division of Pulmonary, Sleep, and Critical Care
Sofia Fayngold CRC Rotation IRB proposal 9/1/2011
 Sofia Fayngold CRC Rotation IRB proposal 9/1/2011 Sodium Bicarbonate Hydration for the Prevention of Contrast Induced Nephropathy following Computed Tomography Angiography of the Chest for Pulmonary Emoblism
Sofia Fayngold CRC Rotation IRB proposal 9/1/2011 Sodium Bicarbonate Hydration for the Prevention of Contrast Induced Nephropathy following Computed Tomography Angiography of the Chest for Pulmonary Emoblism
of Contrast Induced Nephropathy
 Consensus Guidelines for the Prevention of Contrast Induced Nephropathy Benko A, Fraser-Hill M, Magner P, Capusten B, Barrett B, Myers A, Owen RJ. Correspondence to Canadian Association of Radiologists,
Consensus Guidelines for the Prevention of Contrast Induced Nephropathy Benko A, Fraser-Hill M, Magner P, Capusten B, Barrett B, Myers A, Owen RJ. Correspondence to Canadian Association of Radiologists,
With increased use of contrast media (CM), interest in
 ORIGINAL RESEARCH S. Langner S. Stumpe M. Kirsch M. Petrik N. Hosten No Increased Risk for Contrast-Induced Nephropathy after Multiple CT Perfusion Studies of the Brain with a Nonionic, Dimeric, Iso-Osmolal
ORIGINAL RESEARCH S. Langner S. Stumpe M. Kirsch M. Petrik N. Hosten No Increased Risk for Contrast-Induced Nephropathy after Multiple CT Perfusion Studies of the Brain with a Nonionic, Dimeric, Iso-Osmolal
DOSING GUIDE. Indications. Important Safety Information. Enable the immune system. RECOGNIZE. RESPOND.
 DOSING GUIDE For patients with unresectable Stage III NSCLC following concurrent CRT For patients with locally advanced or metastatic UC previously treated with platinum-based therapy Enable the immune
DOSING GUIDE For patients with unresectable Stage III NSCLC following concurrent CRT For patients with locally advanced or metastatic UC previously treated with platinum-based therapy Enable the immune
Canadian Association of Radiologists: Guidelines for the Prevention of Contrast Induced Nephropathy
 Canadian Association of Radiologists: Guidelines for the Prevention of Contrast Induced Nephropathy Canadian Association of Radiologists: Guidelines for the Prevention of Contrast Induced Nephropathy Authors:
Canadian Association of Radiologists: Guidelines for the Prevention of Contrast Induced Nephropathy Canadian Association of Radiologists: Guidelines for the Prevention of Contrast Induced Nephropathy Authors:
EU RISK MANAGEMENT PLAN (EU RMP) Nutriflex Omega peri emulsion for infusion , version 1.1
 EU RISK MANAGEMENT PLAN (EU RMP) Nutriflex Omega peri emulsion for infusion 13.7.2015, version 1.1 III.1. Elements for a Public Summary III.1.1. Overview of disease epidemiology Patients may need parenteral
EU RISK MANAGEMENT PLAN (EU RMP) Nutriflex Omega peri emulsion for infusion 13.7.2015, version 1.1 III.1. Elements for a Public Summary III.1.1. Overview of disease epidemiology Patients may need parenteral
The PRESERVE trial: does it guide prevention strategies for post angiography acute kidney injury?
 Editorial Page 1 of 7 The PRESERVE trial: does it guide prevention strategies for post angiography acute kidney injury? Richard Solomon Larner College of Medicine, University of Vermont, Burlington, VT,
Editorial Page 1 of 7 The PRESERVE trial: does it guide prevention strategies for post angiography acute kidney injury? Richard Solomon Larner College of Medicine, University of Vermont, Burlington, VT,
Non-protein nitrogenous substances (NPN)
 Non-protein nitrogenous substances (NPN) A simple, inexpensive screening test a routine urinalysis is often the first test conducted if kidney problems are suspected. A small, randomly collected urine
Non-protein nitrogenous substances (NPN) A simple, inexpensive screening test a routine urinalysis is often the first test conducted if kidney problems are suspected. A small, randomly collected urine
Contrast media Purpose of using contrast Contrast reaction Nephrotoxicity from contrast Nephrogenic systemic fibrosis When should contrast be used
 Contrast vs Non-Contrast When to Order Stephen McManus, M.D. Contrast media Purpose of using contrast Contrast reaction Nephrotoxicity from contrast Nephrogenic systemic fibrosis When should contrast be
Contrast vs Non-Contrast When to Order Stephen McManus, M.D. Contrast media Purpose of using contrast Contrast reaction Nephrotoxicity from contrast Nephrogenic systemic fibrosis When should contrast be
Contrast-induced nephropathy
 Acute kidney injury with iodinated contrast Peter A. McCullough, MD, MPH, FACC, FACP, FAHA, FCCP Diagnostic and interventional radiographic procedures in critically ill patients commonly depend on iodinated
Acute kidney injury with iodinated contrast Peter A. McCullough, MD, MPH, FACC, FACP, FAHA, FCCP Diagnostic and interventional radiographic procedures in critically ill patients commonly depend on iodinated
PHARMACEUTICAL INFORMATION AZILSARTAN
 AZEARLY Tablets Each Tablet Contains Azilsartan 20/40/80 mg PHARMACEUTICAL INFORMATION AZILSARTAN Generic name: Azilsartan Chemical name: 2-Ethoxy-1-{[2'-(5-oxo-2,5-dihydro-1,2,4-oxadiazol-3-yl)-4-biphenylyl]methyl}-
AZEARLY Tablets Each Tablet Contains Azilsartan 20/40/80 mg PHARMACEUTICAL INFORMATION AZILSARTAN Generic name: Azilsartan Chemical name: 2-Ethoxy-1-{[2'-(5-oxo-2,5-dihydro-1,2,4-oxadiazol-3-yl)-4-biphenylyl]methyl}-
(telavancin) Healthcare Professional s Guide. Version 2, 4 November 2014
 VIBATIV (telavancin) Healthcare Professional s Guide Version 2, 4 November 2014 1 Table of Contents Introduction... 3 About Vibativ / Therapeutic indications... 3 Antimicrobial spectrum of activity for
VIBATIV (telavancin) Healthcare Professional s Guide Version 2, 4 November 2014 1 Table of Contents Introduction... 3 About Vibativ / Therapeutic indications... 3 Antimicrobial spectrum of activity for
Mortality Associated With Nephropathy After Radiographic Contrast Exposure
 ORIGINAL ARTICLE MORTALITY ASSOCIATED WITH NEPHROPATHY AFTER RADIOGRAPHIC CONTRAST EXPOSURE Mortality Associated With Nephropathy After Radiographic Contrast Exposure AARON M. FROM, MD; BRIAN J. BARTHOLMAI,
ORIGINAL ARTICLE MORTALITY ASSOCIATED WITH NEPHROPATHY AFTER RADIOGRAPHIC CONTRAST EXPOSURE Mortality Associated With Nephropathy After Radiographic Contrast Exposure AARON M. FROM, MD; BRIAN J. BARTHOLMAI,
The Incidence of Contrast Induced Nephropathy in Trauma Patients.
 The Incidence of Contrast Induced Nephropathy in Trauma Patients. Item Type Thesis Authors Cordeiro, Samuel Publisher The University of Arizona. Rights Copyright is held by the author. Digital access to
The Incidence of Contrast Induced Nephropathy in Trauma Patients. Item Type Thesis Authors Cordeiro, Samuel Publisher The University of Arizona. Rights Copyright is held by the author. Digital access to
Lab Values Explained. working at full strength. Other possible causes of an elevated BUN include dehydration and heart failure.
 Patient Education Lab Values Explained Common Tests to Help Diagnose Kidney Disease Lab work, urine samples and other tests may be given as you undergo diagnosis and treatment for renal failure. The test
Patient Education Lab Values Explained Common Tests to Help Diagnose Kidney Disease Lab work, urine samples and other tests may be given as you undergo diagnosis and treatment for renal failure. The test
Who Should Be Premediciated for Contrast-Enhanced Exams?
 Who Should Be Premediciated for Contrast-Enhanced Exams? Jeffrey C. Weinreb, MD,FACR Yale University School of Medicine jeffrey.weinreb@yale.edu Types of Intravenous Contrast Media Iodinated Contrast Agents
Who Should Be Premediciated for Contrast-Enhanced Exams? Jeffrey C. Weinreb, MD,FACR Yale University School of Medicine jeffrey.weinreb@yale.edu Types of Intravenous Contrast Media Iodinated Contrast Agents
Standard vs double dose of N-acetylcysteine to prevent contrast agent associated nephrotoxicity
 European Heart Journal (2004) 25, 206 211 Clinical research Standard vs double dose of N-acetylcysteine to prevent contrast agent associated nephrotoxicity Carlo Briguori a,b *, Antonio Colombo b, Anna
European Heart Journal (2004) 25, 206 211 Clinical research Standard vs double dose of N-acetylcysteine to prevent contrast agent associated nephrotoxicity Carlo Briguori a,b *, Antonio Colombo b, Anna
Information for patients having an isotope kidney (renal) scan (also known as a DMSA scan)
 Information for patients having an isotope kidney (renal) scan (also known as a DMSA scan) The leaflet tells you about having an isotope kidney (renal) scan. It is also known as a DMSA scan. It explains
Information for patients having an isotope kidney (renal) scan (also known as a DMSA scan) The leaflet tells you about having an isotope kidney (renal) scan. It is also known as a DMSA scan. It explains
Glucophage XR is contra-indicated during breast-feeding.
 Name GLUCOPHAGE XR 1000 mg Prolonged release tablets Active ingredient Metformin hydrochloride Composition Each Glucophage XR 1000 mg prolonged release tablet contains as active ingredient 1000 mg metformin
Name GLUCOPHAGE XR 1000 mg Prolonged release tablets Active ingredient Metformin hydrochloride Composition Each Glucophage XR 1000 mg prolonged release tablet contains as active ingredient 1000 mg metformin
Clinical guidelines to reduce the risk of contrast induced nephropathy
 Nephrology Subject: Objective: Prepared by: Consultation: Clinical guidelines to reduce the risk of contrast induced nephropathy To implement all possible measures to reduce the risk of nephropathy from
Nephrology Subject: Objective: Prepared by: Consultation: Clinical guidelines to reduce the risk of contrast induced nephropathy To implement all possible measures to reduce the risk of nephropathy from
Summary of the risk management plan (RMP) for Ebymect (dapagliflozin / metformin)
 EMA/672415/2015 Summary of the risk management plan (RMP) for Ebymect (dapagliflozin / metformin) This is a summary of the risk management plan (RMP) for Ebymect, which details the measures to be taken
EMA/672415/2015 Summary of the risk management plan (RMP) for Ebymect (dapagliflozin / metformin) This is a summary of the risk management plan (RMP) for Ebymect, which details the measures to be taken
azilsartan medoxomil
 azilsartan medoxomil edarbi 40mg Tablet 80mg Tablet ANTIHYPERTENSIVE Angiotensin II Receptor Antagonist FORMULATION: Each tablet contains 40mg Azilsartan medoxomil (as potassium) Each tablet contains 80mg
azilsartan medoxomil edarbi 40mg Tablet 80mg Tablet ANTIHYPERTENSIVE Angiotensin II Receptor Antagonist FORMULATION: Each tablet contains 40mg Azilsartan medoxomil (as potassium) Each tablet contains 80mg
Prevention of Contrast Induced Acute Kidney Injury (CI-AKI) In Adult Patients. on behalf of
 Prevention of Contrast Induced Acute Kidney Injury (CI-AKI) In Adult Patients on behalf of The Renal Association, British Cardiovascular Intervention Society and The Royal College of Radiologists Dr Andrew
Prevention of Contrast Induced Acute Kidney Injury (CI-AKI) In Adult Patients on behalf of The Renal Association, British Cardiovascular Intervention Society and The Royal College of Radiologists Dr Andrew
Farmadol. Paracetamol 10 mg/ml INFUSION SOLUTION
 Farmadol Paracetamol 10 mg/ml INFUSION SOLUTION Composition Each ml contains: Paracetamol 10 mg Pharmacology Pharmacodynamic properties The precise mechanism of the analgesic and antipyretic properties
Farmadol Paracetamol 10 mg/ml INFUSION SOLUTION Composition Each ml contains: Paracetamol 10 mg Pharmacology Pharmacodynamic properties The precise mechanism of the analgesic and antipyretic properties
Prevention of Acute Renal Failure Role of vasoactive drugs and diuretic agents
 of Acute Renal Failure Role of vasoactive drugs and diuretic agents Armand R.J. Girbes Prof.dr. A.R.J. Girbes Chairman department of Intensive Care VU University Medical Center Netherlands (Failure of)
of Acute Renal Failure Role of vasoactive drugs and diuretic agents Armand R.J. Girbes Prof.dr. A.R.J. Girbes Chairman department of Intensive Care VU University Medical Center Netherlands (Failure of)
Minimizing the Renal Toxicity of Iodinated Contrast
 Minimizing the Renal Toxicity of Iodinated Contrast Peter A. McCullough, MD, MPH, FACC, FACP, FAHA, FCCP Chief Academic and Scientific Officer St. John Providence Health System Detroit, MI USA Outline
Minimizing the Renal Toxicity of Iodinated Contrast Peter A. McCullough, MD, MPH, FACC, FACP, FAHA, FCCP Chief Academic and Scientific Officer St. John Providence Health System Detroit, MI USA Outline
Posology. Posology and Dosage Regimen: Factors Affecting Drug Dosage: 08/09/ ) Age: 1. Young s Rule, based on age:
 Posology and Dosage Regimen: Posology Posology: (Derived from the Greek posos- how much, and logos- science) is the branch of medicine/pharmacy dealing with doses. Posology is a branch of medical science
Posology and Dosage Regimen: Posology Posology: (Derived from the Greek posos- how much, and logos- science) is the branch of medicine/pharmacy dealing with doses. Posology is a branch of medical science
Special Challenges and Co-Morbidities
 Special Challenges and Co-Morbidities Renal Disease/ Hypertension/ Diabetes in African-Americans M. Keith Rawlings, MD Medical Director Peabody Health Center AIDS Arms, Inc Dallas, TX Chair, Internal Medicine
Special Challenges and Co-Morbidities Renal Disease/ Hypertension/ Diabetes in African-Americans M. Keith Rawlings, MD Medical Director Peabody Health Center AIDS Arms, Inc Dallas, TX Chair, Internal Medicine
M0BCore Safety Profile
 M0BCore Safety Profile Active substance: Aciclovir Pharmaceutical form(s)/strength: Tablets 200, 400 or 800 mg Dispersible tablets 200, 400 or 800 mg Oral suspensions 200 mg or 400 mg per 5 ml. Freeze
M0BCore Safety Profile Active substance: Aciclovir Pharmaceutical form(s)/strength: Tablets 200, 400 or 800 mg Dispersible tablets 200, 400 or 800 mg Oral suspensions 200 mg or 400 mg per 5 ml. Freeze
Prevention of Contrast Induced Nephropathy (CIN) Guidelines
 Prevention of Contrast Induced Nephropathy (CIN) Guidelines This procedural document supersedes: PAT/T 48 v.1 - Guidelines for Prevention of Contrast Induced Nephropathy (CIN) Did you print this document
Prevention of Contrast Induced Nephropathy (CIN) Guidelines This procedural document supersedes: PAT/T 48 v.1 - Guidelines for Prevention of Contrast Induced Nephropathy (CIN) Did you print this document
Acute Kidney Injury in the ED
 + Acute Kidney Injury in the ED + Dr Eric Clark, MD FRCPC University of Ottawa Canada Canadian Association of Emergency Physicians + Outline 1. Diagnostic challenges 2. ED treatment 3. Contrast induced
+ Acute Kidney Injury in the ED + Dr Eric Clark, MD FRCPC University of Ottawa Canada Canadian Association of Emergency Physicians + Outline 1. Diagnostic challenges 2. ED treatment 3. Contrast induced
GALLIUM CITRATE Ga 67 INJECTION
 511945-0903 September 2003 USA Bristol-Myers Squibb Medical Imaging 331 Treble Cove Road N. Billerica, MA 01862 USA GALLIUM CITRATE Ga 67 INJECTION FOR DIAGNOSTIC USE DESCRIPTION: Gallium Citrate Ga 67
511945-0903 September 2003 USA Bristol-Myers Squibb Medical Imaging 331 Treble Cove Road N. Billerica, MA 01862 USA GALLIUM CITRATE Ga 67 INJECTION FOR DIAGNOSTIC USE DESCRIPTION: Gallium Citrate Ga 67
Management of Acute Kidney Injury in the Neonate. Carolyn Abitbol, M.D. University of Miami Miller School of Medicine / Holtz Children s Hospital
 Management of Acute Kidney Injury in the Neonate Carolyn Abitbol, M.D. University of Miami Miller School of Medicine / Holtz Children s Hospital Objectives Summarize the dilemmas in diagnosing & recognizing
Management of Acute Kidney Injury in the Neonate Carolyn Abitbol, M.D. University of Miami Miller School of Medicine / Holtz Children s Hospital Objectives Summarize the dilemmas in diagnosing & recognizing
Research Article Changes in Renal Function in Elderly Patients Following Intravenous Iodinated Contrast Administration: A Retrospective Study
 Radiology Research and Practice, Article ID 459583, 4 pages http://dx.doi.org/10.1155/2014/459583 Research Article Changes in Renal Function in Elderly Patients Following Intravenous Iodinated Contrast
Radiology Research and Practice, Article ID 459583, 4 pages http://dx.doi.org/10.1155/2014/459583 Research Article Changes in Renal Function in Elderly Patients Following Intravenous Iodinated Contrast
CRRT Fundamentals Pre- and Post- Test. AKI & CRRT Conference 2018
 CRRT Fundamentals Pre- and Post- Test AKI & CRRT Conference 2018 Question 1 Which ONE of the following statements regarding solute clearance in CRRT is MOST correct? A. Convective and diffusive solute
CRRT Fundamentals Pre- and Post- Test AKI & CRRT Conference 2018 Question 1 Which ONE of the following statements regarding solute clearance in CRRT is MOST correct? A. Convective and diffusive solute
Acute Coronary Syndrome (ACS) Patients with Chronic Kidney Disease being considered for Cardiac Catheterization. PROVINCIAL PROTOCOL March 2015
 Acute Coronary Syndrome (ACS) Patients with Chronic Kidney Disease being considered for Cardiac Catheterization PROVINCIAL PROTOCOL March 2015 Contents Introduction.......................1 Assessing kidney
Acute Coronary Syndrome (ACS) Patients with Chronic Kidney Disease being considered for Cardiac Catheterization PROVINCIAL PROTOCOL March 2015 Contents Introduction.......................1 Assessing kidney
PRODUCT CIRCULAR. Tablets COZAAR (losartan potassium) I. THERAPEUTIC CLASS II. INDICATIONS III. DOSAGE AND ADMINISTRATION PAK-CZR-T
 PRODUCT CIRCULAR Tablets I. THERAPEUTIC CLASS, the first of a new class of agents for the treatment of hypertension, is an angiotensin II receptor (type AT 1 ) antagonist. also provides a reduction in
PRODUCT CIRCULAR Tablets I. THERAPEUTIC CLASS, the first of a new class of agents for the treatment of hypertension, is an angiotensin II receptor (type AT 1 ) antagonist. also provides a reduction in
Objectives. Pre-dialysis CKD: The Problem. Pre-dialysis CKD: The Problem. Objectives
 The Role of the Primary Physician and the Nephrologist in the Management of Chronic Kidney Disease () By Brian Young, M.D. Assistant Clinical Professor of Medicine David Geffen School of Medicine at UCLA
The Role of the Primary Physician and the Nephrologist in the Management of Chronic Kidney Disease () By Brian Young, M.D. Assistant Clinical Professor of Medicine David Geffen School of Medicine at UCLA
3% Sorbitol Urologic Irrigating Solution in UROMATIC Plastic Container
 3% Sorbitol Urologic Irrigating Solution in UROMATIC Plastic Container Description 3% Sorbitol Urologic Irrigating Solution is a sterile, nonpyrogenic, nonhemolytic, electrically nonconductive solution
3% Sorbitol Urologic Irrigating Solution in UROMATIC Plastic Container Description 3% Sorbitol Urologic Irrigating Solution is a sterile, nonpyrogenic, nonhemolytic, electrically nonconductive solution
Risk factors for adverse reactions from contrast agents for computed tomography
 Kobayashi et al. BMC Medical Informatics and Decision Making 2013, 13:18 RESEARCH ARTICLE Open Access Risk factors for adverse reactions from contrast agents for computed tomography Daiki Kobayashi 1*,
Kobayashi et al. BMC Medical Informatics and Decision Making 2013, 13:18 RESEARCH ARTICLE Open Access Risk factors for adverse reactions from contrast agents for computed tomography Daiki Kobayashi 1*,
PACKAGE INSERT TEMPLATE FOR SALBUTAMOL TABLET & SALBUTAMOL SYRUP
 PACKAGE INSERT TEMPLATE FOR SALBUTAMOL TABLET & SALBUTAMOL SYRUP Brand or Product Name [Product name] Tablet 2mg [Product name] Tablet 4mg [Product name] Syrup 2mg/5ml Name and Strength of Active Substance(s)
PACKAGE INSERT TEMPLATE FOR SALBUTAMOL TABLET & SALBUTAMOL SYRUP Brand or Product Name [Product name] Tablet 2mg [Product name] Tablet 4mg [Product name] Syrup 2mg/5ml Name and Strength of Active Substance(s)
P-RMS: LT/H/PSUR/0004/001
 Core Safety Profile Active substance: Dalteparine Pharmaceutical form(s)/strength: Solution for injection, 2500 I.U./0.2ml, 2500 I.U./ml, 5000 I.U./0.2ml, 7500 I.U./0.3ml, 7500 I.U./0.75ml, 10000 I.U./0.4ml,
Core Safety Profile Active substance: Dalteparine Pharmaceutical form(s)/strength: Solution for injection, 2500 I.U./0.2ml, 2500 I.U./ml, 5000 I.U./0.2ml, 7500 I.U./0.3ml, 7500 I.U./0.75ml, 10000 I.U./0.4ml,
After i.v injection 45% of the amount of desmopressin is found in the urine within 24 hours.
 MINIRIN Tablets 0.1 and 0.2 mg Declaration Tablets 0.1 mg. Each tablet contains desmopressin acetate 0.1 mg and excipients q.s Tablets 0.2 mg. Each tablet contains desmopressin acetate 0.2 mg and excipients
MINIRIN Tablets 0.1 and 0.2 mg Declaration Tablets 0.1 mg. Each tablet contains desmopressin acetate 0.1 mg and excipients q.s Tablets 0.2 mg. Each tablet contains desmopressin acetate 0.2 mg and excipients
CEDIAMATE Metformin Tablets USP 500 mg
 CEDIAMATE Metformin Tablets USP 500 mg COMPOSITION: Cediamate Each un-coated tablet contains: Metformin Hydrochloride USP Excipients 500 mg Q.S PHARMACOLOGY: Pharmacotherapeutic group: Blood Glucose lowering
CEDIAMATE Metformin Tablets USP 500 mg COMPOSITION: Cediamate Each un-coated tablet contains: Metformin Hydrochloride USP Excipients 500 mg Q.S PHARMACOLOGY: Pharmacotherapeutic group: Blood Glucose lowering
Metformin Associated Lactic Acidosis. Jun-Ki Park 9/6/11
 Metformin Associated Lactic Acidosis Jun-Ki Park 9/6/11 Probably the most common mechanism by which metformin elevates blood lactate is by inducing catecholamine release in those who regulate or prescribe
Metformin Associated Lactic Acidosis Jun-Ki Park 9/6/11 Probably the most common mechanism by which metformin elevates blood lactate is by inducing catecholamine release in those who regulate or prescribe
CKD FOR INTERNISTS. Dr Ahmed Hossain Associate professor Medicine Sir Salimullah Medical College
 CKD FOR INTERNISTS Dr Ahmed Hossain Associate professor Medicine Sir Salimullah Medical College INTRODUCTION In 2002, the National Kidney Foundation s Kidney Disease Outcomes Quality Initiative(KDOQI)
CKD FOR INTERNISTS Dr Ahmed Hossain Associate professor Medicine Sir Salimullah Medical College INTRODUCTION In 2002, the National Kidney Foundation s Kidney Disease Outcomes Quality Initiative(KDOQI)
NEW YORK CITY DEPARTMENT OF HEALTH AND MENTAL HYGIENE Thomas R. Frieden, MD, MPH Commissioner
 NEW YORK CITY DEPARTMENT OF HEALTH AND MENTAL HYGIENE Thomas R. Frieden, MD, MPH Commissioner Interim Guidance on Dosage, Precautions, and Adverse Effects of Antiviral Medications used to Treat or Prevent
NEW YORK CITY DEPARTMENT OF HEALTH AND MENTAL HYGIENE Thomas R. Frieden, MD, MPH Commissioner Interim Guidance on Dosage, Precautions, and Adverse Effects of Antiviral Medications used to Treat or Prevent
CEDR 2018 QCDR Measures for CMS 2018 MIPS Performance Year Reporting
 ACEP19 Emergency Department Utilization of CT for Minor Blunt Head Trauma for Aged 18 Years and Older Percentage of visits for aged 18 years and older who presented with a minor blunt head trauma who had
ACEP19 Emergency Department Utilization of CT for Minor Blunt Head Trauma for Aged 18 Years and Older Percentage of visits for aged 18 years and older who presented with a minor blunt head trauma who had
Each tablet contains:
 Composition: Each tablet contains: Tolvaptan 15/30mg Pharmacokinetic properties: In healthy subjects the pharmacokinetics of tolvaptan after single doses of up to 480 mg and multiple doses up to 300 mg
Composition: Each tablet contains: Tolvaptan 15/30mg Pharmacokinetic properties: In healthy subjects the pharmacokinetics of tolvaptan after single doses of up to 480 mg and multiple doses up to 300 mg
Salapin: Salbutamol BP 2mg as sulphate in each 5mL of a raspberry cola flavoured, sugar free syrup.
 Salapin Salbutamol Syrup 2mg/5mL Qualitative and quantitative composition Salapin: Salbutamol BP 2mg as sulphate in each 5mL of a raspberry cola flavoured, sugar free syrup. Clinical particulars Therapeutic
Salapin Salbutamol Syrup 2mg/5mL Qualitative and quantitative composition Salapin: Salbutamol BP 2mg as sulphate in each 5mL of a raspberry cola flavoured, sugar free syrup. Clinical particulars Therapeutic
ACETYLCYSTEINE INJECTION
 ACETYLCYSTEINE INJECTION 1. Name of the medicinal product Acetylcysteine 200 mg/ml Injection 2. Qualitative and quantitative composition Acetylcysteine 200mg per ml (as N-acetylcysteine) Each 10ml ampoule
ACETYLCYSTEINE INJECTION 1. Name of the medicinal product Acetylcysteine 200 mg/ml Injection 2. Qualitative and quantitative composition Acetylcysteine 200mg per ml (as N-acetylcysteine) Each 10ml ampoule
Journal of the American College of Cardiology Vol. 48, No. 5, by the American College of Cardiology Foundation ISSN /06/$32.
 Journal of the American College of Cardiology Vol. 48, No. 5, 2006 2006 by the American College of Cardiology Foundation ISSN 0735-1097/06/$32.00 Published by Elsevier Inc. doi:10.1016/j.jacc.2006.06.047
Journal of the American College of Cardiology Vol. 48, No. 5, 2006 2006 by the American College of Cardiology Foundation ISSN 0735-1097/06/$32.00 Published by Elsevier Inc. doi:10.1016/j.jacc.2006.06.047
Environmental Variability
 1 Environmental Variability Body Size, Body Composition, Maturation and Organ Function Nick Holford Dept Pharmacology & Clinical Pharmacology University of Auckland 2 Objectives Understand the major sources
1 Environmental Variability Body Size, Body Composition, Maturation and Organ Function Nick Holford Dept Pharmacology & Clinical Pharmacology University of Auckland 2 Objectives Understand the major sources
Hydronephrosis. What is hydronephrosis?
 What is hydronephrosis? Hydronephrosis Hydronephrosis describes the situation where the urine collecting system of the kidney is dilated. This may be a normal variant or it may be due to an underlying
What is hydronephrosis? Hydronephrosis Hydronephrosis describes the situation where the urine collecting system of the kidney is dilated. This may be a normal variant or it may be due to an underlying
Ureteral Stenting and Nephrostomy
 Scan for mobile link. Ureteral Stenting and Nephrostomy Ureteral stenting and nephrostomy help restore urine flow through blocked ureters and return the kidney to normal function. Ureters are long, narrow
Scan for mobile link. Ureteral Stenting and Nephrostomy Ureteral stenting and nephrostomy help restore urine flow through blocked ureters and return the kidney to normal function. Ureters are long, narrow
BIOL 2402 Renal Function
 BIOL 2402 Renal Function Dr. Chris Doumen Collin County Community College 1 Renal Clearance and GFR Refers to the volume of blood plasma from which a component is completely removed in one minute by all
BIOL 2402 Renal Function Dr. Chris Doumen Collin County Community College 1 Renal Clearance and GFR Refers to the volume of blood plasma from which a component is completely removed in one minute by all
Bevacizumab + Paclitaxel + Cisplatin
 Bevacizumab + Paclitaxel + Cisplatin Available for Routine Use in Not routinely commissioned, each case requires prior documented approval before offering & commencing therapy from the Cancer Drugs Fund
Bevacizumab + Paclitaxel + Cisplatin Available for Routine Use in Not routinely commissioned, each case requires prior documented approval before offering & commencing therapy from the Cancer Drugs Fund
Use of abatacept in rheumatoid arthritis - patient information
 Practical management of patients receiving abatacept 1 Use of abatacept in rheumatoid arthritis - patient information Evidence-based Medicine Official Recommendations Expert opinion Key points 1. Abatacept
Practical management of patients receiving abatacept 1 Use of abatacept in rheumatoid arthritis - patient information Evidence-based Medicine Official Recommendations Expert opinion Key points 1. Abatacept
Section 3: Prevention and Treatment of AKI
 http://www.kidney-international.org & 2012 KDIGO Summary of ommendation Statements Kidney International Supplements (2012) 2, 8 12; doi:10.1038/kisup.2012.7 Section 2: AKI Definition 2.1.1: AKI is defined
http://www.kidney-international.org & 2012 KDIGO Summary of ommendation Statements Kidney International Supplements (2012) 2, 8 12; doi:10.1038/kisup.2012.7 Section 2: AKI Definition 2.1.1: AKI is defined
Beyond NSF: Acute GBCA adverse reactions
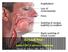 Source images Beyond NSF: Acute GBCA adverse reactions Martin R. Prince, MD, PhD, FACR Disclosures Patent Agreements: GE, Siemens, Philips, Hitachi, Toshiba, Bayer, Bracco, Mallinckrodt, Medrad, Nemoto,
Source images Beyond NSF: Acute GBCA adverse reactions Martin R. Prince, MD, PhD, FACR Disclosures Patent Agreements: GE, Siemens, Philips, Hitachi, Toshiba, Bayer, Bracco, Mallinckrodt, Medrad, Nemoto,
Contrast nephropathy: can we see the whole picture
 Contrast nephropathy: can we see the whole picture Raff M, MSc(Wits), MBChB(Pret), FFA(SA) Correspondence to: Dr Milton Raff, e-mail: raffs@iafrica.com Introduction Renal protection has remained a highly
Contrast nephropathy: can we see the whole picture Raff M, MSc(Wits), MBChB(Pret), FFA(SA) Correspondence to: Dr Milton Raff, e-mail: raffs@iafrica.com Introduction Renal protection has remained a highly
Renal Disease and PK/PD. Anjay Rastogi MD PhD Division of Nephrology
 Renal Disease and PK/PD Anjay Rastogi MD PhD Division of Nephrology Drugs and Kidneys Kidney is one of the major organ of drug elimination from the human body Renal disease and dialysis alters the pharmacokinetics
Renal Disease and PK/PD Anjay Rastogi MD PhD Division of Nephrology Drugs and Kidneys Kidney is one of the major organ of drug elimination from the human body Renal disease and dialysis alters the pharmacokinetics
GUIDELINES FOR WEIGHT-BASED DOSING AND INFUSION
 GUIDELINES FOR WEIGHT-BASED DOSING AND INFUSION Includes Example dose calculation wheel Preparation and administration information for healthcare professionals Please see enclosed full Prescribing Information,
GUIDELINES FOR WEIGHT-BASED DOSING AND INFUSION Includes Example dose calculation wheel Preparation and administration information for healthcare professionals Please see enclosed full Prescribing Information,
GLUCOPHAGE 500 mg Merck Serono
 GLUCOPHAGE 500 mg Film-coated tablets Composition Film-coated tablets containing 500 mg of metformin (INN) hydrochloride (equivalent to 390 mg of metformin Excipients Polyvidone K 30, magnesium stearate,
GLUCOPHAGE 500 mg Film-coated tablets Composition Film-coated tablets containing 500 mg of metformin (INN) hydrochloride (equivalent to 390 mg of metformin Excipients Polyvidone K 30, magnesium stearate,
The CARI Guidelines Caring for Australians with Renal Impairment. Cardiovascular Risk Factors
 Cardiovascular Risk Factors ROB WALKER (Dunedin, New Zealand) Lipid-lowering therapy in patients with chronic kidney disease Date written: January 2005 Final submission: August 2005 Author: Rob Walker
Cardiovascular Risk Factors ROB WALKER (Dunedin, New Zealand) Lipid-lowering therapy in patients with chronic kidney disease Date written: January 2005 Final submission: August 2005 Author: Rob Walker
Contrast-induced nephropathy prevention and risk reduction
 Nephrol Dial Transplant (2006) 21 [Suppl 1]: i11 i23 doi:10.1093/ndt/gfl215 Contrast-induced nephropathy prevention and risk reduction Norbert H. Lameire Renal Division, Department of Internal Medicine,
Nephrol Dial Transplant (2006) 21 [Suppl 1]: i11 i23 doi:10.1093/ndt/gfl215 Contrast-induced nephropathy prevention and risk reduction Norbert H. Lameire Renal Division, Department of Internal Medicine,
Prophylactic Use of Potassium Iodide (KI) in Radiological Emergencies*: Information for Physicians
 Prophylactic Use of Potassium Iodide (KI) in Radiological Emergencies*: Information for Physicians * Based on FDA's guidance document on use of KI as a thyroid blocking agent in radiation emergencies 1.
Prophylactic Use of Potassium Iodide (KI) in Radiological Emergencies*: Information for Physicians * Based on FDA's guidance document on use of KI as a thyroid blocking agent in radiation emergencies 1.
45779C/Revised: April 2008 MANNITOL INJECTION, USP
 45779C/Revised: April 2008 MANNITOL INJECTION, USP 25% For Intravenous Use and Urologic Irrigation DESCRIPTION: Mannitol is a 6-carbon sugar alcohol and has the following structure: C 6 H 14 O 6 182.17
45779C/Revised: April 2008 MANNITOL INJECTION, USP 25% For Intravenous Use and Urologic Irrigation DESCRIPTION: Mannitol is a 6-carbon sugar alcohol and has the following structure: C 6 H 14 O 6 182.17
Guidelines for Contrast Media from the European Society of Urogenital Radiology
 Guidelines for Contrast Media from the European Society of Urogenital Radiology T his perspective will present all guidelines produced to date by the Contrast Media Safety Committee of the European Society
Guidelines for Contrast Media from the European Society of Urogenital Radiology T his perspective will present all guidelines produced to date by the Contrast Media Safety Committee of the European Society
Prevention of Contrast-Induced Nephropathy in the Emergency Department
 EVIDENCE-BASED EMERGENCY MEDIE REVIEW Prevention of Contrast-Induced Nephropathy in the Emergency Department Richard Sinert, DO Christopher I. Doty, MD From the Department of Emergency Medicine, State
EVIDENCE-BASED EMERGENCY MEDIE REVIEW Prevention of Contrast-Induced Nephropathy in the Emergency Department Richard Sinert, DO Christopher I. Doty, MD From the Department of Emergency Medicine, State
Update on Cardiorenal Syndrome: A Clinical Conundrum
 Advances in Peritoneal Dialysis, Vol. 27, 2011 Eric J. Chan, 1 Kevin C. Dellsperger 1 3 Update on Cardiorenal Syndrome: A Clinical Conundrum Our understanding of the cardiorenal syndrome continues to progress.
Advances in Peritoneal Dialysis, Vol. 27, 2011 Eric J. Chan, 1 Kevin C. Dellsperger 1 3 Update on Cardiorenal Syndrome: A Clinical Conundrum Our understanding of the cardiorenal syndrome continues to progress.
