The Involvement of Autophagy Pathway in Exaggerated Ischemic Brain Damage in Diabetic Mice
|
|
|
- Emerald Dawson
- 5 years ago
- Views:
Transcription
1 ORIGINAL ARTICLE The Involvement of Autophagy Pathway in Exaggerated Ischemic Brain Damage in Diabetic Mice Ning Wei, 1,2 Shan-Ping Yu, 2 Xiao-Huan Gu, 2 Dong-Dong Chen, 2 Matthew K. Whalin, 2 Ge-Lin Xu, 1 Xin-Feng Liu 1 & Ling Wei 2,3 1 Department of Neurology, Nanjing University School of Medicine, Jinling Hospital, Nanjing, China 2 Department of Anesthesiology, Emory University School of Medicine, Atlanta, GA, USA 3 Department of Neurology, Emory University School of Medicine, Atlanta, GA, USA Keywords Autophagy; Blood brain barrier; Cell death; Diabetes; Ischemic stroke. Correspondence Ling Wei, M.D., Department of Anesthesiology, Emory University School of Medicine, Atlanta, 101 Woodruff Circle, Suite 630, GA 30322, USA. Tel.: ; Fax: ; lwei7@emory.edu or Xinfeng Liu, M.D., Ph.D., Department of Neurology, Nanjing University School of Medicine, Jinling Hospital, 305 East Zhongshan Road, Nanjing, , Jiangsu Province, China Tel.: ; Fax: ; xfliu2@gmail.com Received 11 January 2013; revision 3 April 2013; accepted 7 April 2013 SUMMARY Background: Patients with Diabetes are at greater risk for ischemic stroke and usually suffer more severe ischemic brain damage than nondiabetic patients. However, the underlying mechanism of the exaggerated injury is not well defined. Aims: Macroautophagy (hereafter called autophagy in this report) plays a key role in cellular homeostasis and may contribute to cell death as well. Our aim was to determine whether autophagy was involved in the enhanced susceptibility of diabetic brain cells to ischemic injury and explore it as a possible target for the treatment of stroke in a diabetic condition. Results: A type II diabetic mouse model generated by combined administration of streptozotocin and nicotinamide showed enlarged infarct volume, increased cell death and excessive blood brain barrier (BBB) disruption compared with nondiabetic stroke mice. After ischemic stroke, both diabetic and nondiabetic mice showed enhanced autophagosome formation and autophagic flux as demonstrated by increased expression of autophagy signals Beclin 1, microtubule-associated protein light-chain II (LC3-II), and decreased autophagy-specific substrate p62. The increased autophagic activity was significantly higher in diabetic stroke mice than that in nondiabetic stroke mice. The autophagy inhibitor 3-methyladenine (3-MA) attenuated the exaggerated brain injury and improved functional recovery. Conclusions: These data suggest that autophagy contributes to exacerbated brain injury in diabetic condition, and autophagy-mediated cell death may be a therapeutic target in diabetic stroke. doi: /cns Introduction Focal cerebral ischemia is one of the leading causes of death and disability worldwide, and more than 30% of stroke patients are known to be diabetic [1,2]. More than 90% of the 200 million patients with diabetes across the world are type 2 diabetes, which is characterized by high blood glucose in the context of insulin resistance and relative insulin deficiency. It is well known that both acute hyperglycemia and chronic diabetes exaggerate ischemic brain damage in experimental and clinical stroke, but the mechanisms remain elusive [3 5]. Brain lactic acid accumulation and tissue acidosis due to hyperglycemia have been most commonly correlated with more extensive tissue damage in diabetic stroke [6]. Nevertheless, rapid correction of the hyperglycemia after acute ischemia with reduced brain lactic acid level has not improved outcomes in clinical trials [6,7]. In addition to the vascular and hyperglycemic alterations, other mechanisms might contribute to the enhanced susceptibility of the diabetic brain to ischemic injury [1,8]. Autophagy is a physiological process long known to maintain cellular homeostasis through lysosomal degradation, recycling long-lived proteins and eliminating damaged organelles such as mitochondria and endoplasmic reticulum. Autophagy is thus known primarily as a prosurvival mechanism for cells facing nutrient deprivation or other stress conditions. However, accumulating evidence indicates that autophagy can contribute to cell death processes under pathological conditions [9 12]. The double-edged effect of autophagy on pancreatic b-cell loss in diabetes ª 2013 John Wiley & Sons Ltd CNS Neuroscience & Therapeutics 19 (2013)
2 Autophagic Cell Death in Diabetic Stroke N. Wei et al. and in cerebral ischemia has been implicated [12 14]. The functional role of autophagy in the pathogenesis of diabetic complications such as atherosclerosis, cardiomyopathy, nephropathy, and neuropathy is currently under intensive investigation [15 17]. In studies of nondiabetic animals, autophagic activities increase following cerebral ischemia [10,18 20], and some evidence indicates that the autophagy pathway could contribute to ischemiamediated neuronal cell death [18,21 23]. Information about autophagy in diabetic stroke animals has been very limited. Two recent reports showed that 23 days after transient common carotid artery (CCA) occlusion seemingly mimicking a global ischemic insult, increased neuronal loss, astrocytes activation, Ab generation, and autophagy activity were observed in the hippocampus of diabetic mice [17,24]. On the other hand, to our knowledge, there has been no study to investigate the involvement of autophagic pathway in the pathogenesis of exaggerating acute brain damage after focal ischemic stroke in diabetic animals. The present investigation examined the autophagic activity in a murine type 2 diabetic stroke model. We further examined a possible treatment of diabetic stroke using an autophagy inhibitor 3- methyladenine (3-MA) and determine the role of autophagy in the pathogenesis of ischemic brain damage in diabetic mice. Materials and Methods Diabetic Mouse Model All animal experiments and surgical procedures were approved by the Institutional Animal Care and Use Committee at Emory University and met NIH standards. Adult male C57BL/6 mice (National Cancer Institute, NCI, Bethesda, MD, USA), weeks old and approximately 25 g in weight, were used in this study. Diabetes was induced by i.p. injection of nicotinamide (210 mg/kg; Sigma, St. Louis, MO, USA) dissolved in saline 15 min before an i.p. administration of streptozotocin (180 mg/kg; Sigma) dissolved in citrate buffer (ph 4.5; Sigma) prepared immediately before use [25,26]. Controls received the vehicles of both substances. After treatment with STZ or vehicle, animals were housed for 2 months and given food and water ad libitum. Blood glucose levels following a four-hour fast were monitored twice per month using samples from the tail vein with the FreeStyle â glucose meter (Abbott Diabetes Care, Alameda, CA, USA). Two months after the induction of diabetes, blood was collected from orbit. Twenty-five microlitres of plasma were assayed for insulin by ELISA using a Mercodia Ultrasensitive Insulin ELISA Kit (Mercodia AB Uppsala, Sweden). Focal Cerebral Ischemic Stroke Model The focal cerebral ischemic stroke in adult male mice was induced as previously described [27]. Briefly, animals were anesthetized with i.p. injection of 4% chloral hydrate. Focal ischemia restricted to the right barrel/sensorimotor cortex was induced by permanent occlusion of 2 3 distal branches of the MCA. This was accompanied by 7-min bilateral CCA ligation. Sham animals were done with the same surgery procedure without MCA or CCA occlusion. During surgery and recovery periods, body temperature was monitored using a rectal probe and maintained at C using a heating pad and a temperature-controlled, ventilated incubator. 3-Methyladenine Administration 3-Methyladenine (3-MA; Sigma), a widely used pharmacological inhibitor of autophagy, was prepared to a final concentration of 30 mg/ml in 2 ll saline and was administered by intracerebroventricular (i.c.v.) injection immediately after ischemia onset into the ipsilateral ventricle. Saline alone injection served as a negative control. Infarct Volume Assessment At 24 h after stroke, the brain was removed and sliced into 1-mm coronal sections using a mouse brain matrix and incubated in 2% 2, 3-5-triphenyl-tetrazolium chloride solution at 37 C for 5 min. Brain sections were scanned, and the unstained versus stained area was determined using NIH Image J on the ventral side of six brain slides per animal. The infarct area (mm 2 ) of staining in each slice was multiplied by the slice thickness (1 mm) to get the infarct volume (mm 3 ). The indirect infarct volume was calculated by the difference between the volume of contralateral cortex and the volume of the TTC-stained portion (nonischemic) of ipsilateral cortex of each mouse, following the calculation: contralateral volume (ipsilateral volume infarct volume). This method helps to correct edema in the ipsilateral hemisphere in order to achieve accurate assessments of infarct volume. The brain infarct volume was the summation of six individual section volumes. Terminal Deoxynucleotidyl Transferase BiotindUPT Nick End Labeling Ten lm brain sections were prepared every 90 lm across the region of interest. Cell death was assessed using the Terminal deoxynucleotidyl transferase biotin-dupt nick end labeling (TUNEL) staining kit according to the manufacturer s instructions (Dead- End Fluorometric TUNEL system; Promega, Madison, WI, USA). The slides were counterstained with Hoechst (1:20,000; Life Technologies, Grand Island, NY, USA) for 5 min to reveal the nucleus of all cells before mounting with ProLong Antifade mounting medium (Life Technologies). The result was presented as ratio of TUNEL-positive cells among all nuclei within 4009 magnification fields. Immunohistochemical Staining Brain cryosections (10 lm thickness) were fixed with 4% paraformaldehyde for 15 min, permeabilized with 0.2% Triton X-100 for 5 min, and blocked with 1% fish gelatin (Sigma) for 1 h at room temperature. Specimens were then incubated with the following primary antibodies overnight at 4 C: goat anticollagen type IV (1:400; Millipore, Billerica, MA, USA), rabbit anti-lc3 (microtubule-associated protein 1A light-chain 3; 1:400; Cell Signaling, Danvers, MA, USA), goat anticathepsin D (1:200; Santa Cruz Biotechnology, Santa Cruz, CA, USA) and anti-beclin 1 (1:5000; Abcam, Cambridge, MA, USA). For the staining of occludin, brain cryosections were fixed with ethanol at 4 C for 30 min followed by ice-cold acetone for 3 min, washed, blocked, and incubated with mouse antioccludin (1:100; Life Technologies). After rinsing with PBS, brain sections were then treated for 1 h at room tem- 754 CNS Neuroscience & Therapeutics 19 (2013) ª 2013 John Wiley & Sons Ltd
3 N. Wei et al. Autophagic Cell Death in Diabetic Stroke perature with the relevant secondary antibodies: Cy5-conjugated donkey anti-goat IgG, Cy3-conjugated donkey anti-rabbit IgG (Jackson ImmunoResearch, West Grove, PA, USA) or Alexa Fluor 488 anti-rabbit IgG and anti-mouse IgG (1:200; Life Technologies). Slides were mounted with ProLong Antifade mounting medium (Life Technologies) and analyzed under a fluorescent microscope (BX51; Olympus, Tokyo, Japan). Cell Counting For systematic random sampling in design-based stereological cell counting, every ninth brain section (90 lm apart) across the entire region of interest was counted. For multistage random sampling, six fields per brain section were randomly chosen in the penumbra region [27] under 4009 magnification of a fluorescent microscope. This was repeated in six separate sections per brain. Western Blotting Twenty-four hours after ischemia, 3 5 animals for each group were used to collect ipsilateral penumbra tissue samples. The brain tissues were lysed using modified radioimmunoprecipitation assay buffer (50 mmol/l HEPES, ph 7.3, 1% sodium deoxycholate, 1% Triton X-100, 0.1% sodium dodecylsulfate, 150 mmol/l NaCl, 1 mmol/l ethylenediaminetetraacetic acid, 1 mmol/l Na 3 VO 4, 1 mmol/l NaF), and protease inhibitor cocktail (Sigma) for 30 min, followed by centrifugation at 17,000 g for 15 min. Protein concentration of each sample was determined using the bicinchoninic acid assay (Sigma). Samples of 20 lg proteins were electrophoresed on a 6% to 20% sodium dodecyl sulfate polyacrylamide gradient gel in a Hoefer Mini-Gel system (Amersham Biosciences, Piscataway, NJ, USA) and transferred in the Hoefer Transfer Tank (Amersham Biosciences) to a polyvinylidene difluoride membrane (BioRad, Hercules, CA). Membranes were blocked with buffer (Tris-buffered saline containing 0.1% Tween-20, ph 7.6%, 5% bovine serum albumin) and then incubated with one of the following antibodies overnight at 4 C: rabbit anti-lc3 (1:1000; Novus, Littleton, CO, USA), anti-sqstm1/a170/p62, rabbit (1:500; Wako Inc., Osaka, Japan), rabbit anti-bcl-2 (1:1000; Cell Signaling), rabbit anticleaved caspase-3 (1:500; Cell Signaling) and mouse antioccludin (1:500; Life Technologies). Mouse b- actin (Sigma) was used as the protein loading control. The blots were washed in 0.5% Tris-buffered saline containing 0.1% Tween-20 (TBST) and incubated with alkaline phosphatase-conjugated anti-rabbit or anti-mouse IgG (Promega) for 2 hrs at room temperature. Finally, membranes were washed with TBST followed by three washes with Tris-buffered saline. Signal was detected by the addition of 5-bromo-4-chloro-3-indolyl-phosphate/nitroblue tetrazolium (BCIP/NBT) solution (Sigma). Data were quantified and analyzed using the NIH image J program (NIH, Bethesda, MD, USA). The expression level of each protein was corrected against the loading control of b-actin. Evaluation of Neurological Deficits The adhesive-removal test is a sensitive test for assessing sensorimotor deficits after focal cerebral ischemia affecting the sensorimotor cortex [18,28]. This was used to evaluate the neurological dysfunction after stroke, as previously described [18]. Briefly, a training session (for 3 days, 1 2 trialsperday)wasperformeduntilthemice could take off the sticky dots on their paws within 12 seconds before surgical procedures. Animals were tested before and 23 h after ischemia by an investigator who was blinded to the experimental groups. The shortest time (tested from 4 to 5 trials) required to sense (time to detect) and remove (time to remove) the sticky dot from the left paw was recorded. All testing trials were conducted during the day time. Statistical Analysis Two-tailed Student s t-test was used for comparison of two experimental groups. Multiple comparisons were performed using oneway analysis of variance followed by Tukey s test for multiple comparisons. For pairwise comparisons, one-way analysis followed by Bonferroni selective comparisons were performed. Changes were identified as significant if P was less than Data were expressed as mean SEM. Results Pathophysiological Assessments of the Experimental Diabetic Mouse A diabetic animal model was created in adult male mice via injection of streptozotocin and nicotinamide (STZ/NA). Within 2 months after the induction, animals exhibited significantly lower body weight and elevated levels of blood glucose ( mg/dl) with respect to age-matched vehicle control mice (Table 1). This hyperglycemia was stable during the 2-month period before ischemic surgery. Plasma insulin levels, measured 2 months after STZ/NA treatment, were decreased by 68% in diabetic mice as compared with controls. Treatment with the autophagy inhibitor 3-MA (30 mg/ml in 2 ll saline, i.c.v. injection) had no effect on these variables in diabetic mice before and during 24-hr period after ischemia. The mortality rate after stroke surgery in diabetes group was seemingly higher, yet not significant different from nondiabetic stroke animals. Increased Autophagosome Formation in Diabetic Stroke Mice To study autophagy after ischemic stroke, diabetic and nondiabetic animals were subjected to a focal ischemic insult targeting the right barrel/sensorimotor cortex [27]. Beclin 1 is an essential autophagy protein known to activate the lipid kinase Vps34 and induce autophagy [29]. Immunostaining showed that the Table 1 General features of the experimental diabetic mouse models Measurements Nondiabetic Diabetic Diabetic + 3-MA Body weight (g) * * Blood glucose (mg/dl) * * Serum insulin (lg/l) * * N Values are the mean SEM. 3-MA, 3-Methyladenine. *P < 0.05 vs. nondiabetes controls. ª 2013 John Wiley & Sons Ltd CNS Neuroscience & Therapeutics 19 (2013)
4 Autophagic Cell Death in Diabetic Stroke N. Wei et al. increase in brain beclin 1 expression in diabetic stroke mice was significantly greater than that in nondiabetic stroke animals at 12 and 24 h after stroke (Figure 1). A cytosolic form of microtubule-associated protein light-chain I (LC3-I) is lipidated during autophagy to form LC3-II, a widely used marker for autophagosome [21,30,31]. To evaluate whether the autophagy pathway is involved in the diabetic ischemic brain, immunostaining of LC3 was visualized by fluorescence microscopy as punctuate structures representing autophagosomes 24 hrs after ischemic insult (Figure 2A C and Figure 3A C). On the other hand, diffuse staining of LC3 could be intermittently detected in the sham control brain (Figure 2A). The LC3-positive cells were primarily located in the penumbra region of the brain after focal ischemia. The percentage of cells with bright LC3 punctae and cells colabeled with TUNEL staining was significantly elevated in diabetic stroke mice compared with nondiabetic stroke controls 24 h after ischemia (Figure 2B D). The conversion from LC3-I to LC3-II occurs in autophagy, while the LC3-II level is regarded more reliable in autophagy measurement [32,33]. LC3-II was thus detected using Western blotting in penumbra tissues of the ischemic brain. No difference was seen in the basal level of LC3-II between diabetic and nondiabetic mice (data not shown). In stroke animals, LC3-II levels significantly increased 24 h after ischemia in both groups (Figure 2E). Even more LC3 conversion was detected in diabetic stroke mice compared with nondiabetic ischemic controls (Figure 2F). As expected, the augmented LC3-II conversion could be largely prevented by the autophagy inhibitor 3-MA (Figure 2F). (A) (B) Increased Autophagosome Flux in Diabetic Stroke Mice (C) (E) (D) As authophagy is a dynamic process, the increased number of autophagosomes could result from either increased autophagic activity (autophagic flux) or decreased degradation of autophagosomes [21]. To distinguish these possibilities, levels of the lysosomal protease cathepsin D and the autophagy-specific substrate p62 were measured in the postischemic brain [21,34]. At 24 h postischemia, immunostaining revealed cells with cathepsin D- positive dots in the peri-infarct region of both diabetic and nondiabetic stroke brain sections (Figure 3B,C). At high magnification, double staining showed strong autophagosomal LC3 labeling and lysosomal cathepsin D labeling in the same cells (Figure 3A C). In Western blot analysis, focal cerebral ischemia induced significant decreases in the p62 level. In diabetic stroke mice, the reduction was much greater compared with nondiabetic stroke animals (Figure 3D). Thus, increased LC3 signaling and decreased p62 expression support an upregulation of autophagic flux in diabetic stroke mice. Increased Ischemic Brain Damage and Neuroprotective Effect of Autophagy Inhibition in Diabetic Stroke Mice Figure 1 Increased autophagosome after cerebral ischemia in diabetic mice. Immunohistochemical staining of brain sections from stroke animals, focusing on the peri-infarct region. (A D) Representative images of beclin signal (red) and TUNEL-positive cells (green) at 24 h after ischemia in nondiabetic stroke mice (A and B) and diabetic stroke mice (C, D). B and D are higher magnification of the framed region in A and C, respectively. Arrowheads point to beclin-positive cells. E. Quantified data showed significantly increased beclin signals in the penumbra region at 12 and 24 h postischemia in diabetic animals compared with regular controls. N = 5 per group at each time point. *P < 0.05 vs. nondiabetic stroke controls. Diabetic stroke mice showed significantly larger brain infarct volume: the infarct volume measured using TTC staining was 28% larger than that in age-matched nondiabetic stroke animals (Figure 4A,B). Diabetic stroke mice showed an increased percentage of TUNEL-positive cells in the penumbra region ( vs , in diabetic and nondiabetic mice, respectively, n = 5 and 15 for nondiabetic and diabetic mice, respectively, P < 0.05) 24 h after stroke (Figure 4C F). As we observed augmented autophagic activity in the diabetic stroke brain, we tested the hypothesis that the increased autophagy contributed to the more severe brain damage. Intraventricular injection of 3-MA, at a dosage that could block LC3-II conversion (Figure 2F), was applied to diabetic stroke animals. 3- MA reduced infarct volume both in regular and diabetic mice measured using TTC staining (Figure 4B). In a closer examination at the cellular level, a significant reduction of TUNEL-positive cells was seen with 3-MA 24 h after stroke in nondiabetic stroke mice and diabetic stroke mice (Figure 4G). 756 CNS Neuroscience & Therapeutics 19 (2013) ª 2013 John Wiley & Sons Ltd
5 N. Wei et al. Autophagic Cell Death in Diabetic Stroke (A) (B) (C) (D) (E) (F) Figure 2 Increased autophagic cell death in the diabetic stroke brain. Immunohistochemical staining and Western blotting were applied to specifically detect autophagy-associated signals in sham controls and after ischemic stroke. (A D) The LC3 staining in sham controls are diffuse red signals as shown in image A. The bright red punctuate structures around nuclei (DAPI: blue) in images B and C are the LC3 signal representing autophagosomes 24 h after ischemic insult. The arrows indicate the cells with increased autophagosome; dashed arrows showed cells that are positive for both LC3 and TUNEL staining [54]. (D) Quantification data show significant increases in LC3-positive cells as well as LC3 and TUNEL double-positive cells in the penumbra region 24 h after ischemia in diabetic mice. N = 5 per group. *P < 0.05 vs. nondiabetic stroke controls. (E) LC3-II conversion assessed using Western blotting in sham control and the postischemic brain. Upper panel is representative immunoblot of LC3 in extracts of the penumbra region. Lower panel shows quantification of the LC3-II band intensity ratio compared with loading controls. At 24 h after ischemia, the level of LC3-II in both nondiabetic control and diabetic animals was elevated, but the increase was much greater in the diabetic brain. N = 2 for sham control, n = 5 for nondiabetic stroke and diabetic stroke mice, respectively. (F) In diabetic stroke mice, the LC3-II expression was about doubled from the sham control level. 3-MA treatment significantly brought down the LC3-II conversation. N = 3 for sham group, n = 4 for diabetes stroke saline or 3-MA group. One-way ANOVA followed by Bonferroni selective comparisons was performed for analyzing the level of LC3 between nondiabetic stroke and sham animals. *P < 0.05 vs. sham control, # P < 0.05 vs. nondiabetic stroke in E or saline control in F. The tight junction protein occludin is a marker of interendothelial clefts and an important indicator of the integrity and paracellular permeability of the blood brain barrier (BBB) [35,36]. Diabetes had no effect on occludin expression in sham controls (data not shown). Compared with sham controls, occludin levels were significantly decreased 24 hrs after ischemia in both diabetic and normoglycemic groups. The decrease, however, was significantly greater in diabetic stroke animals (Figure 5A E). 3-MA treatment soon after stroke led to noticeable preservation of occludin levels in nondiabetic stroke mice; the effect on occludin expression in diabetic stroke mice, however, was not significant in immunostaining experiments (Figure 5E). As the immunohistochemical method ª 2013 John Wiley & Sons Ltd CNS Neuroscience & Therapeutics 19 (2013)
6 Autophagic Cell Death in Diabetic Stroke N. Wei et al. (A) (B) (C) (D) surveys a limited region of the ischemic brain, we then assessed the occludin expression in the peri-infarct cortex using Western blot analysis and identified that 3-MA treatment significantly increased the occludin protein level in diabetic stroke mice comparing with saline-treated diabetic mice (Figure 5F). Effects of 3-MA Treatment on Apoptotic Cell Death Pathways The interaction between apoptotic and autophagic pathways has been proven both in vitro and in nondiabetic cerebral ischemic animals [10,37]. We tested whether this cross-talk exists in a diabetic condition. In Western blotting assays using the antibody against cleavaged caspase-3, both nondiabetic and diabetic stroke mice showed significant caspase-3 activation in peri-infarct region (Figure 6A). 3-MA treatment significantly suppressed the caspase-3 activation by about 50% (Figure 6A). In diabetic stroke mice, the cerebral ischemia did not change the level of antiapoptotic protein Bcl-2 24 h postischemia, while 3-MA treatment significantly doubled the expression of Bcl-2 in the diabetic stroke brain (Figure 6B). These experiments focused on the diabetic condition support the idea that apoptotic cell death contributed significantly to ischemic brain injury. Sensorimotor Functional Deficit after Ischemic Stroke and Effect of 3-MA In neurological functional assays, worse deficits were seen with diabetic stroke mice compared with nondiabetic stroke mice. In adhesive-removal test, both nondiabetic and diabetic stroke mice took significantly more time to detect and remove sticky dots attached to their ischemia-affected left paws (Figure 7A,B). Stroke mice in both groups received the 3-MA treatment (30 ng, i.c.v. soon after ischemia) showed improvements in the functional assessment. 3-MA-treated diabetic stroke mice was significantly faster in detecting the sticky dot (shorter latency), although the reduction in removal time was not significant (Figure 7A,B). The 3-MA treatment in nondiabetic stroke animals did not reach to statistical significance in these experiments (Figure 7A,B). Discussion Figure 3 Increased autophagic flux in diabetic stroke mice. Immunohistochemical staining or Western blotting of autophagic flux signals in sham control and stroke animals. (A C) Representative images showing colocalization of autophagosomal marker (LC3, red) and lysosomal marker (cathepsin D, green) within the same cells (DAPI: blue) in the penumbral cortex of diabetes mice. Arrows show cells double-labeled with LC3 and cathepsin D. Dashed arrow shows a cell which is negative for both signals. (D) Upper panel: Representative immunoblot of p62 in extracts of ischemic penumbra cortex at 24 h postischemia. Lower panel: Quantification of the p62 band intensity expressed as a percentage of sham p62. There was significantly decreased expression level of p62 in the penumbra cortex of both nondiabetic and diabetic stroke animals at 24 h postischemia. N = 2 for sham, n = 5 for nondiabetic and diabetic stroke mice, respectively. One-way ANOVA followed by Bonferroni selective comparisons was carried out for the analysis. *P < 0.05 vs. sham control, # P < 0.05 vs. nondiabetic stroke. The present investigation demonstrates that ischemic brain damage is exacerbated in STZ/NA-induced diabetic mice. The exaggerated brain damage occurs concurrently with upregulated autophagic activity in the ischemic brain. We provide novel evidence that not only autophagosome formation but also autophagic flux is augmented in diabetic stroke mice. Blocking the postischemic autophagy using the autophagy inhibitor 3-MA prevents the excessive ischemic brain damage in diabetic mice as well as shows improvements in functional recovery after ischemic stroke. In the STZ/NA-induced diabetic mouse model, we confirmed that these mice developed nonobese diabetic symptoms characterized by stable hyperglycemia ( mg/dl), hypoinsulinemia (68% reduction), and growth impairment after STZ treatment [26,38]. Due to the partial protection of NA against the b-cell cytotoxic effect of STZ, these diabetic mice showed better overall well-being and lower mortality rates when subjected to the cere- 758 CNS Neuroscience & Therapeutics 19 (2013) ª 2013 John Wiley & Sons Ltd
7 N. Wei et al. Autophagic Cell Death in Diabetic Stroke (A) (B) (C) (D) (E) (F) (G) Figure 4 Protective effect of autophagy inhibition with 3-MA in the diabetic ischemic brain. Cerebra ischemia induced brain infarct formation and cell death were measured 24 h after the onset of MCA occlusion using TTC and TUNEL staining, respectively. Autophagy was blocked using i.c.v. injection of 3-MA immediately after ischemia. (A) TTC staining images of infarct area (white) in nondiabetic stroke control, diabetic stroke mice received saline vehicle treatment and 3-MA treatment. (B) Quantified TTC staining measurement of infarct volume calculated using the indirect method. Diabetic mice showed significantly larger infarct volume compared with nondiabetic stroke mice (n = 8 and 11, respectively). 3-MA treatment showed a trend of reducing the infarct volume (n = 10, P = 0.14). The 3-MA effect become significant when direct measurement of infarct volume was applied (data not shown), indicating that 3-MA had an inhibitory action on brain edema. (C F) Immunofluorescent images of all nuclei (Hoechst, blue) and TUNEL-positive (green) cells in the penumbra region of the nondiabetic and diabetic stroke brain with and without 3-MA. (G) Cell counting results from the immunostaining assay. Diabetes significantly increased the percentage of TUNEL-positive cells (n = 5 and 15 for nondiabetic and diabetic mice, respectively). 3-MA treatment soon after ischemia (n = 12) significantly attenuated the percentage of TUNEL-positive cells. *P < 0.05 versus saline controls, # P < 0.05 versus nondiabetes saline group. bral ischemic insult compared with animals rendered diabetic by STZ alone. This type of diabetic model is closer to type 2 diabetes with respect to b-cell dysfunction, diminished insulin-to-glucose ratio, and calorie-controlled high-fat diet leading to insulin resistance [38]. Upon focal cerebral ischemia after 2 months of hyperglycemia, these diabetic animals exhibited larger infarct volume, enhanced cell death ratio in the penumbral region, more severe BBB disruptions and worse functional deficits compared with normoglycemic controls. As it is well known that ischemic injury is exaggerated with hyperglycemia [39], we assume the increased damage is closely related to the hyperglycemia-associated oxidative stress, vascular inflammation and tissue acidosis as well as impaired cellular metabolism [40 42]. Autophagy occurs at basal levels in most tissues as part of homeostatic functions and is involved in development, differentiation, and tissue remodeling [43,44]. Autophagy plays a doubleedged role in certain diseases, including cancer, liver disease, muscular disorders, b-cell mass loss in diabetes, and neurodegenerative diseases. Despite the physiological role of cellular protection, autophagic activities in these disease states may contribute to cell damage depending on the cellular milieu [9,13,43]. Previous evidence indicates that autophagic cell death is a potential contributor to ischemia-induced neuronal cell death in nondiabetic stroke animals [10,18 23]. Consistent with the general idea that autophagy is an insult to neuronal cells, autophagy has been shown to generate a preconditioning effect against ischemic damage [45,46]. Our present investigation suggests that increased autophagy flux plays a role in exaggerated cell death in the diabetic ischemic brain. This is in line with a previous report that autophagy increases after a global ischemic insult in diabetic mice [17]. ª 2013 John Wiley & Sons Ltd CNS Neuroscience & Therapeutics 19 (2013)
8 Autophagic Cell Death in Diabetic Stroke N. Wei et al. (A) (B) (E) (C) (D) (F) Figure 5 Effect of inhibiting autophagy on tight junction disruption. Immunohistochemical staining and Western blotting were used to detect the expression level of the tight junction marker occludin in the penumbra of normal and stroke animals. (A D) Immunostaining images of occludin (red) and collagen IV (blue) in sham control animals (A), nondiabetic stroke mice (B), diabetic stroke mice (C), and diabetic stroke animals with 3-MA treatment. (E) Image data were quantified by the area percentage based on fluorescence density. Stroke markedly decreased the occludin expression level in the saline groups of both nondiabetic and diabetic mice. 3-MA treatment significantly attenuated the occludin decrease. N = 4 5in each group; *P < 0.05 vs. sham control, # P < 0.05 vs. nondiabetic stroke control. (F) Western blot analysis of occludin protein levels in the brain from diabetic stroke animals. Upper panel: representative immunoblots. Lower panel: Quantification of occludin band intensities expressed as ratio compared with b-actin loading controls. 3-MA treatment preserved occluding levels relative to stroke saline group of diabetic mice. N = 5 in each group, *P < 0.05 vs. diabetic stroke plus saline. During autophagy process, the membrane-bound structure autophagosome is formed to sequester cytoplasm. The autophagosome fuses with a lysosome, where it is degraded and the macromolecules recycled [43]. We showed that there is an upregulation of LC3-II conjugate, a marker for autophagosomes, assessed by both Western blotting and immunostaining in diabetic stroke mice comparing with age-matched nondiabetic stroke mice. Double labeling showed that the increased number of autophagosomes colocalized with the upregulated lysosomal marker cathepsin D in the same cells. Further evidence for upregulated autophagic flux came from the assay on the level of p62, a specific substrate that is preferentially degraded by autophagy. This assay showed greater decreases of p62 in diabetic stroke animals compared with nondiabetic controls, which is consistent with augmented autophagic activities. These results provide evidence that there is an increase in autophagic flux, not just an increase in autophagosomes formation in diabetic stroke mice. To further determine the role of the upregulated autophagic flux in the pathology of diabetic stroke, we tested the effects of the autophagy inhibitor 3-MA. Our results showed that 3-MA prevented exaggerated brain damage in diabetic mice and lowered the injury to the level of nondiabetic stroke mice. 3-MA also showed functional benefits in diabetic stroke animals. These data indicate that the increased autophagic flux contributes to the exacerbated ischemic brain damage in diabetic mice. There are limitations that can be identified in the experiment using 3-MA. 3-MA is a PI3-kinase inhibitor (both class I and class III) and has been widely used as a pharmacological inhibitor in autophagy studies. However, 3-MA is not autophagy specific [21,47]. 3-MA acts on other kinases and affects other cellular process such as lysosomal acidification and the mitochondrial permeability transition [21]. In the present investigation, we did not solely rely on the pharmacological tool for the demonstration of the involvement of autophagy. The increased expression level of LC3 and decreased level of P62 in diabetic stoke mice and the colocalization of LC3 and cathepsin D in the same cells agree that autophagic flux indeed increases in the diabetic mice. Knocking down or deleting specific autophagy genes such as Atg7 and combined therapy with autophagy and apoptosis inhibitors may provide more specific evidence in further experiments. It has been shown that autophagy can either directly execute cell death in Bax/Bak double knockout mice or alter the dynamics of death through interactions with apoptosis [43,48,49]. Apoptosis and autophagy pathways may act synergistically or counter each other by sharing many of the same molecular regulators, such as Bcl-2 family proteins and sphingo-lipid metabolic pathways 760 CNS Neuroscience & Therapeutics 19 (2013) ª 2013 John Wiley & Sons Ltd
9 N. Wei et al. Autophagic Cell Death in Diabetic Stroke (A) (B) Figure 6 Effects of 3-MA on caspase-3 activation and the expression level of Bcl-2 in the diabetic stroke brain. Western blot was performed 24 hr after the onset of MCA occlusion. (A) Expression of cleaved caspase-3 in the penumbra region. Upper panel: representative immunoblots. Lower panel: Quantification of cleaved caspase-3 band intensities expressed as a fold change from the sham level of cleaved caspase-3. Caspase-3 activity was significantly increased both non-diabetic and diabetic stroke mice penumbra. 3-MA treatment significantly suppressed the caspase-3 activation by about 50% in diabetic stroke mice. (B) Upper panel: representative immunoblots of Bcl-2 expression level in the extracts of ischemic penumbra cortex. Lower panel: Quantification of Bcl-2 band intensities normalized to actin loading controls. In diabetic stroke mice, the cerebral ischemia did not change the Bcl-2 level, while 3-MA treatment significantly doubled the expression of Bcl-2 in the diabetic stroke brain. N = 5 in each group, *P < 0.05 vs. sham controls, # P < 0.05 vs. diabetic stroke plus saline. (A) (B) Figure 7 Functional benefits of 3-MA treatment in diabetic stroke mice. Sensorimotor functional activity was assessed using the removal test performed 24 h after stroke. (A) Measurement of time to detect the attached dot on the affected paw corresponding to the ischemic sensorimotor cortex. There were no differences among groups before stroke. Functional deficit of prolonged detection time was seen 24 h after stroke in both nondiabetic and diabetic animals. 3-MA showed a strong trend of shortening the detecting time (P = 0.09) in nondiabetic stroke mice. The shortening in 3-MA-treated mice was statistically significant. (B) Measurement of time to remove the attached dot. All animals showed similar removal times before stroke, stroke caused significant delay in the removal time. Between nondiabetic and diabetic groups, diabetic stroke mice took much longer time to remove the sticky dot from their pawns. This functional deficit was largely corrected by 3-MA treatment. The correction by 3-MA in nondiabetic mice was not significant. N = 7 in control groups, n = 11 for nondiabetic stroke groups, n = 8 and 6 for diabetic with and without 3-MA treatments. *P < 0.05 vs. nonstroke controls (before stroke), # P < 0.05 vs. diabetic stroke control. [9,37,50]. The cross-talk between these two pathways has been proven both in vitro and in nondiabetic cerebral ischemic animals [10,37]. It remains to be verified whether the postischemia autophagic activity damages the cells directly or through interactions with the apoptotic cell death pathway. Our data suggested that autophagy and apoptosis seemed to be linked in diabetic cerebral ischemic animals, because inhibiting autophagy with 3-MA markedly attenuates caspase-3 activation and augments the antiapoptotic protein Bcl-2 expression. The latter is the founding member of the Bcl-2 family of apoptosis regulators [51] and is reported to be participate in the inhibition of autophagy through the interaction of Bcl-2/Bcl-XL with Beclin 1 [52]. This observation further suggests possible crosstalk between autophagy and apoptosis after focal cerebral ischemia in diabetic mice. The markedly increased antiapoptotic gene Bcl-2 by 3-MA is an interesting observation, which may contribute partially to the neuroprotective effect of 3- MA in the ischemic brain at the time when PI3K kinases are inhibited. In our study, over 80% of TUNEL-positive cells in the penumbra region showed positive staining with autophagic markers such ª 2013 John Wiley & Sons Ltd CNS Neuroscience & Therapeutics 19 (2013)
10 Autophagic Cell Death in Diabetic Stroke N. Wei et al. as LC3. Given the fact that cell death is often accompanied by features of autophagy, we cannot conclude that this majority of TUN- EL/LC3 double-positive cells undergo cell death through a pure autophagy pathway [53]. In previous investigations, we showed that ischemia-induced neuronal damage is a mixed form of cell death, including concurrent necrotic and apoptotic features in the same cells [7]. This hybrid or mixed cell death can be demonstrated in vitro under conditions such as oxygen glucose deprivation and Na +,K + -ATPase failure [28]. More evidence implicates that the hybrid cell death is a common form of cell death after ischemia in vivo when the insult simultaneously triggers multiple cell death pathways [7,37]. The present investigation provides further evidence that autophagy is one of the contributors to the hybrid cell death mechanism in the ischemic brain. We recognize that identification of autophagy in different cell populations will provide additional information for the cell death mechanism in different cells such as glial and endothelial cells. In our cell counting assay on double staining with LC3-II and NeuN, over 90% LC3-II-positive cells were NeuN positive. On the other hand, ischemia-induced BBB disruption is worse in diabetic mice than in nondiabetic mice, suggesting exacerbated injury to vascular endothelial cells. As 3-MA attenuates the BBB damage, autophagy may occur in endothelial cells. A further investigation will be needed to focus on the role of autophagy in endothelial cells, neurovascular unit and BBB damage in nondiabetic and diabetic mice. In conclusion, we here report novel evidence for the involvement of upregulated autophagic activity in mediating exaggerated brain damage in diabetic mice after focal cerebral ischemia. The excessive autophagy in diabetic stroke mice could result from diabetes-related factors like hyperglycemia, insulin deficiency, and other related metabolic defects. Further studies will be needed to explore the underlying mechanism. Collectively, these results suggest that autophagy pathway is associated with worsened tissue damage in diabetic stroke. The involvement of autophagy-mediated neuronal and vascular cell injury merits further attention in the cell death mechanism of diabetic stroke and provides a potential new target for the treatment of ischemic stroke in patients with diabetes. Acknowledgments This work was supported by NIH grants NS (LW), NS (LW), NS (LW), NS (SPY), and an AHA Established Investigator Award (LW). This work was also supported by the NIH grant C06 RR from the Extramural Research Facilities Program of the National Center for Research Resources. Conflict of Interest The authors declare no conflict of interest. References 1. Moskowitz MA, Lo EH, Iadecola C. The science of stroke: mechanisms in search of treatments. Neuron 2010;67: Lawes CM, Parag V, Bennett DA, et al. Blood glucose and risk of cardiovascular disease in the Asia Pacific region. Diabetes Care 2004;27: Gisselsson L, Smith ML, Siesjo BK. Hyperglycemia and focal brain ischemia. J Cereb Blood Flow Metab 1999;19: Muranyi M, Fujioka M, He Q, et al. Diabetes activates cell death pathway after transient focal cerebral ischemia. Diabetes 2003;52: Arboix A, Rivas A, Garcia-Eroles L, et al. Cerebral infarction in diabetes: clinical pattern, stroke subtypes, and predictors of in-hospital mortality. BMC Neurol 2005;5:9. 6. Bruno A, Liebeskind D, Hao Q, et al. Diabetes mellitus, acute hyperglycemia, and ischemic stroke. Curr Treat Options Neurol 2010;12: Wei L, Ying DJ, Cui L, et al. Necrosis, apoptosis and hybrid death in the cortex and thalamus after barrel cortex ischemia in rats. Brain Res 2004;1022: Biessels GJ, van der Heide LP, Kamal A, et al. Ageing and diabetes: implications for brain function. Eur J Pharmacol 2002;441: Eisenberg-Lerner A, Bialik S, Simon HU, et al. Life and death partners: apoptosis, autophagy and the cross-talk between them. Cell Death Differ 2009;16: Puyal J, Vaslin A, Mottier V, et al. Postischemic treatment of neonatal cerebral ischemia should target autophagy. Ann Neurol 2009;66: Wang P, Miao CY. Autophagy in the disorders of central nervous system: vital and/or fatal? CNS Neurosci Ther 2012;18: Wei K, Wang P, Miao CY. A double-edged sword with therapeutic potential: an updated role of autophagy in ischemic cerebral injury. CNS Neurosci Ther 2012;18: Chen ZF, Li YB, Han JY, et al. The double-edged effect of autophagy in pancreatic beta cells and diabetes. Autophagy 2011;7: Kaniuk NA, Kiraly M, Bates H, et al. Ubiquitinatedprotein aggregates form in pancreatic beta-cells during diabetes-induced oxidative stress and are regulated by autophagy. Diabetes 2007;56: Tanaka Y, Kume S, Kitada M, et al. Autophagy as a therapeutic target in diabetic nephropathy. Exp Diabetes Res 2012;2012: Gonzalez CD, Lee MS, Marchetti P, et al. The emerging role of autophagy in the pathophysiology of diabetes mellitus. Autophagy 2011;7: Zhang T, Yan W, Li Q, et al. 3-n-Butylphthalide (NBP) attenuated neuronal autophagy and amyloid-beta expression in diabetic mice subjected to brain ischemia. Neurol Res 2011;33: Bouet V, Boulouard M, Toutain J, et al. The adhesive removal test: a sensitive method to assess sensorimotor deficits in mice. Nat Protoc 2009;4: He S, Wang C, Dong H, et al. Immune-related GTPase M (IRGM1) regulates neuronal autophagy in a mouse model of stroke. Autophagy 2012;8: Liu C, Gao Y, Barrett J, et al. Autophagy and protein aggregation after brain ischemia. J Neurochem 2010;115: Mizushima N, Yoshimori T, Levine B. Methods in mammalian autophagy research. Cell 2010;140: Wen YD, Sheng R, Zhang LS, et al. Neuronal injury in rat model of permanent focal cerebral ischemia is associated with activation of autophagic and lysosomal pathways. Autophagy 2008;4: Grishchuk Y, Ginet V, Truttmann AC, et al. Beclin 1- independent autophagy contributes to apoptosis in cortical neurons. Autophagy 2011;7: Zhang T, Liu X, Li Q, et al. Exacerbation of ischemiainduced amyloid-beta generation by diabetes is associated with autophagy activation in mice brain. Neurosci Lett 2010;479: Masiello P, Broca C, Gross R, et al. Experimental NIDDM: development of a new model in adult rats administered streptozotocin and nicotinamide. Diabetes 1998;47: Novelli M, D Aleo V, Lupi R, et al. Reduction of oxidative stress by a new low-molecular-weight antioxidant improves metabolic alterations in a nonobese mouse diabetes model. Pancreas 2007;35:e10 e Whitaker VR, Cui L, Miller S, et al. Whisker stimulation enhances angiogenesis in the barrel cortex following focal ischemia in mice. J Cereb Blood Flow Metab 2007;27: Xiao AY, Wei L, Xia S, et al. Ionic mechanism of ouabaininduced concurrent apoptosis and necrosis in individual cultured cortical neurons. J Neurosci 2002;22: Kihara A, Kabeya Y, Ohsumi Y, et al. Beclinphosphatidylinositol 3-kinase complex functions at the trans-golgi network. EMBO Rep 2001;2: Rangaraju S, Verrier JD, Madorsky I, et al. Rapamycin activates autophagy and improves myelination in explant cultures from neuropathic mice. J Neurosci 2010;30: Kabeya Y, Mizushima N, Ueno T, et al. LC3, a mammalian homologue of yeast Apg8p, is localized in autophagosome membranes after processing. EMBO J 2000;19: Mizushima N, Yoshimori T. How to interpret LC3 immunoblotting. Autophagy 2007;3: Tanida I, Ueno T, Kominami E. LC3 and Autophagy. Methods Mol Biol 2008;445: CNS Neuroscience & Therapeutics 19 (2013) ª 2013 John Wiley & Sons Ltd
11 N. Wei et al. Autophagic Cell Death in Diabetic Stroke 34. Bjorkoy G, Lamark T, Brech A, et al. p62/sqstm1 forms protein aggregates degraded by autophagy and has a protective effect on huntingtin-induced cell death. J Cell Biol 2005;171: Liu W, Hendren J, Qin XJ, et al. Normobaric hyperoxia attenuates early blood-brain barrier disruption by inhibiting MMP-9-mediated occludin degradation in focal cerebral ischemia. J Neurochem 2009;108: Tavelin S, Hashimoto K, Malkinson J, et al. A new principle for tight junction modulation based on occludin peptides. Mol Pharmacol 2003;64: Wei L, Han BH, Li Y, et al. Cell death mechanism and protective effect of erythropoietin after focal ischemia in the whisker-barrel cortex of neonatal rats. J Pharmacol Exp Ther 2006;317: Nakamura T, Terajima T, Ogata T, et al. Establishment and pathophysiological characterization of type 2 diabetic mouse model produced by streptozotocin and nicotinamide. Biol Pharm Bull 2006;29: Harada S, Fujita-Hamabe W, Tokuyama S. Ischemic stroke and glucose intolerance: a review of the evidence and exploration of novel therapeutic targets. J Pharmacol Sci 2012;118: Folbergrova J, Memezawa H, Smith ML, et al. Focal and perifocal changes in tissue energy state during middle cerebral artery occlusion in normo- and hyperglycemic rats. J Cereb Blood Flow Metab 1992;12: Widmer H, Abiko H, Faden AI, et al. Effects of hyperglycemia on the time course of changes in energy metabolism and ph during global cerebral ischemia and reperfusion in rats: correlation of 1H and 31P NMR spectroscopy with fatty acid and excitatory amino acid levels. J Cereb Blood Flow Metab 1992;12: Yan J, Feng Z, Liu J, et al. Enhanced autophagy plays a cardinal role in mitochondrial dysfunction in type 2 diabetic Goto-Kakizaki (GK) rats: ameliorating effects of (-)-epigallocatechin-3-gallate. J Nutr Biochem 2012;23: Shintani T, Klionsky DJ. Autophagy in health and disease: a double-edged sword. Science 2004;306: Levine B, Klionsky DJ. Development by self-digestion: molecular mechanisms and biological functions of autophagy. Devel Cell 2004;6: Wang P, Guan YF, Du H, et al. Induction of autophagy contributes to the neuroprotection of nicotinamide phosphoribosyltransferase in cerebral ischemia. Autophagy 2012;8: Sheng R, Liu XQ, Zhang LS, et al. Autophagy regulates endoplasmic reticulum stress in ischemic preconditioning. Autophagy 2012;8: Petiot A, Ogier-Denis E, Blommaart EF, et al. Distinct classes of phosphatidylinositol 3 -kinases are involved in signaling pathways that control macroautophagy in HT-29 cells. J Biol Chem 2000;275: Yu L, Alva A, Su H, et al. Regulation of an ATG7-beclin 1 program of autophagic cell death by caspase-8. Science 2004;304: Shimizu S, Kanaseki T, Mizushima N, et al. Role of Bcl-2 family proteins in a non-apoptotic programmed cell death dependent on autophagy genes. Nat Cell Biol 2004;6: Pattingre S, Tassa A, Qu X, et al. Bcl-2 antiapoptotic proteins inhibit Beclin 1-dependent autophagy. Cell 2005;122: Zhang T, Jia W. Sun X 3-n-Butylphthalide (NBP) reduces apoptosis and enhances vascular endothelial growth factor (VEGF) up-regulation in diabetic rats. Neurol Res 2010;32: Levine B, Sinha S, Kroemer G. Bcl-2 family members: dual regulators of apoptosis and autophagy. Autophagy 2008;4: Denton D, Nicolson S, Kumar S. Cell death by autophagy: facts and apparent artefacts. Cell Death Differ 2012;19: Galluzzi L, Vitale I, Abrams JM, et al. Molecular definitions of cell death subroutines: recommendations of the Nomenclature Committee on Cell Death Cell Death Differ 2012;19: ª 2013 John Wiley & Sons Ltd CNS Neuroscience & Therapeutics 19 (2013)
Sestrin2 and BNIP3 (Bcl-2/adenovirus E1B 19kDa-interacting. protein3) regulate autophagy and mitophagy in renal tubular cells in. acute kidney injury
 Sestrin2 and BNIP3 (Bcl-2/adenovirus E1B 19kDa-interacting protein3) regulate autophagy and mitophagy in renal tubular cells in acute kidney injury by Masayuki Ishihara 1, Madoka Urushido 2, Kazu Hamada
Sestrin2 and BNIP3 (Bcl-2/adenovirus E1B 19kDa-interacting protein3) regulate autophagy and mitophagy in renal tubular cells in acute kidney injury by Masayuki Ishihara 1, Madoka Urushido 2, Kazu Hamada
Impact factor: Reporter:4A1H0019 Chen Zi Hao 4A1H0023 Huang Wan ting 4A1H0039 Sue Yi Zhu 4A1H0070 Lin Guan cheng 4A1H0077 Chen Bo xuan
 Curcumin Protects Neonatal Rat Cardiomyocytes against High Glucose-Induced Apoptosis via PI3K/Akt Signalling Pathway Wei Yu,1,2 Wenliang Zha,1 Zhiqiang Ke,1 Qing Min,2 Cairong Li,1 Huirong Sun,3 and Chao
Curcumin Protects Neonatal Rat Cardiomyocytes against High Glucose-Induced Apoptosis via PI3K/Akt Signalling Pathway Wei Yu,1,2 Wenliang Zha,1 Zhiqiang Ke,1 Qing Min,2 Cairong Li,1 Huirong Sun,3 and Chao
Bone marrow-derived mesenchymal stem cells improve diabetes-induced cognitive impairment by
 Nakano et al. Supplementary information 1. Supplementary Figure 2. Methods 3. References Bone marrow-derived mesenchymal stem cells improve diabetes-induced cognitive impairment by exosome transfer into
Nakano et al. Supplementary information 1. Supplementary Figure 2. Methods 3. References Bone marrow-derived mesenchymal stem cells improve diabetes-induced cognitive impairment by exosome transfer into
Supplementary Figure 1
 Supplementary Figure 1 The average sigmoid parametric curves of capillary dilation time courses and average time to 50% peak capillary diameter dilation computed from individual capillary responses averaged
Supplementary Figure 1 The average sigmoid parametric curves of capillary dilation time courses and average time to 50% peak capillary diameter dilation computed from individual capillary responses averaged
Islet viability assay and Glucose Stimulated Insulin Secretion assay RT-PCR and Western Blot
 Islet viability assay and Glucose Stimulated Insulin Secretion assay Islet cell viability was determined by colorimetric (3-(4,5-dimethylthiazol-2-yl)-2,5- diphenyltetrazolium bromide assay using CellTiter
Islet viability assay and Glucose Stimulated Insulin Secretion assay Islet cell viability was determined by colorimetric (3-(4,5-dimethylthiazol-2-yl)-2,5- diphenyltetrazolium bromide assay using CellTiter
Chemical Chaperones Mitigate Experimental Asthma By Attenuating Endoplasmic
 Chemical Chaperones Mitigate Experimental Asthma By Attenuating Endoplasmic Reticulum Stress Lokesh Makhija, BE, Veda Krishnan, MSc, Rakhshinda Rehman, MTech, Samarpana Chakraborty, MSc, Shuvadeep Maity,
Chemical Chaperones Mitigate Experimental Asthma By Attenuating Endoplasmic Reticulum Stress Lokesh Makhija, BE, Veda Krishnan, MSc, Rakhshinda Rehman, MTech, Samarpana Chakraborty, MSc, Shuvadeep Maity,
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION Supplementary Figure 1. Long-term protection studies. 45 minutes of ischemia was induced in wild type (S1pr2 +/+ ) and S1pr2 -/- by MCAO. A) 5 days later brains were harvested
SUPPLEMENTARY INFORMATION Supplementary Figure 1. Long-term protection studies. 45 minutes of ischemia was induced in wild type (S1pr2 +/+ ) and S1pr2 -/- by MCAO. A) 5 days later brains were harvested
SUPPLEMENTARY INFORMATION
 Supplementary Figures Supplementary Figure S1. Binding of full-length OGT and deletion mutants to PIP strips (Echelon Biosciences). Supplementary Figure S2. Binding of the OGT (919-1036) fragments with
Supplementary Figures Supplementary Figure S1. Binding of full-length OGT and deletion mutants to PIP strips (Echelon Biosciences). Supplementary Figure S2. Binding of the OGT (919-1036) fragments with
SUPPLEMENTARY INFORMATION In format provided by JAATTELA (NOVEMBER 2005)
 Box S1: Methods for studying lysosomal function and integrity Volume and distribution of the acidic compartment. Acridine orange is a metachromatic fluorochrome and a weak base that accumulates in the
Box S1: Methods for studying lysosomal function and integrity Volume and distribution of the acidic compartment. Acridine orange is a metachromatic fluorochrome and a weak base that accumulates in the
Li et al. Journal of Experimental & Clinical Cancer Research (2018) 37:108
 Li et al. Journal of Experimental & Clinical Cancer Research (2018) 37:108 https://doi.org/10.1186/s13046-018-0774-7 CORRECTION Correction to: Novel smac mimetic APG- 1387 elicits ovarian cancer cell killing
Li et al. Journal of Experimental & Clinical Cancer Research (2018) 37:108 https://doi.org/10.1186/s13046-018-0774-7 CORRECTION Correction to: Novel smac mimetic APG- 1387 elicits ovarian cancer cell killing
Programmed necrosis, not apoptosis, is a key mediator of cell loss and DAMP-mediated inflammation in dsrna-induced retinal degeneration
 Programmed necrosis, not apoptosis, is a key mediator of cell loss and DAMP-mediated inflammation in dsrna-induced retinal degeneration The Harvard community has made this article openly available. Please
Programmed necrosis, not apoptosis, is a key mediator of cell loss and DAMP-mediated inflammation in dsrna-induced retinal degeneration The Harvard community has made this article openly available. Please
Downregulation of angiotensin type 1 receptor and nuclear factor-κb. by sirtuin 1 contributes to renoprotection in unilateral ureteral
 Supplementary Information Downregulation of angiotensin type 1 receptor and nuclear factor-κb by sirtuin 1 contributes to renoprotection in unilateral ureteral obstruction Shao-Yu Yang 1,2, Shuei-Liong
Supplementary Information Downregulation of angiotensin type 1 receptor and nuclear factor-κb by sirtuin 1 contributes to renoprotection in unilateral ureteral obstruction Shao-Yu Yang 1,2, Shuei-Liong
Name Animal source Vendor Cat # Dilutions
 Supplementary data Table S1. Primary and Secondary antibody sources Devi et al, TXNIP in mitophagy A. Primary Antibodies Name Animal source Vendor Cat # Dilutions 1. TXNIP mouse MBL KO205-2 1:2000 (WB)
Supplementary data Table S1. Primary and Secondary antibody sources Devi et al, TXNIP in mitophagy A. Primary Antibodies Name Animal source Vendor Cat # Dilutions 1. TXNIP mouse MBL KO205-2 1:2000 (WB)
hexahistidine tagged GRP78 devoid of the KDEL motif (GRP78-His) on SDS-PAGE. This
 SUPPLEMENTAL FIGURE LEGEND Fig. S1. Generation and characterization of. (A) Coomassie staining of soluble hexahistidine tagged GRP78 devoid of the KDEL motif (GRP78-His) on SDS-PAGE. This protein was expressed
SUPPLEMENTAL FIGURE LEGEND Fig. S1. Generation and characterization of. (A) Coomassie staining of soluble hexahistidine tagged GRP78 devoid of the KDEL motif (GRP78-His) on SDS-PAGE. This protein was expressed
THE ROLE OF ALTERED CALCIUM AND mtor SIGNALING IN THE PATHOGENESIS OF CYSTINOSIS
 Research Foundation, 18 month progress report THE ROLE OF ALTERED CALCIUM AND mtor SIGNALING IN THE PATHOGENESIS OF CYSTINOSIS Ekaterina Ivanova, doctoral student Elena Levtchenko, MD, PhD, PI Antonella
Research Foundation, 18 month progress report THE ROLE OF ALTERED CALCIUM AND mtor SIGNALING IN THE PATHOGENESIS OF CYSTINOSIS Ekaterina Ivanova, doctoral student Elena Levtchenko, MD, PhD, PI Antonella
Receptor-interacting Protein Kinases Mediate Necroptosis In Neural Tissue Damage After Spinal Cord Injury
 Receptor-interacting Protein Kinases Mediate Necroptosis In Neural Tissue Damage After Spinal Cord Injury Haruo Kanno, M.D., Ph.D., Hiroshi Ozawa, M.D., Ph.D., Satoshi Tateda, M.D., Kenichiro Yahata, M.D.,
Receptor-interacting Protein Kinases Mediate Necroptosis In Neural Tissue Damage After Spinal Cord Injury Haruo Kanno, M.D., Ph.D., Hiroshi Ozawa, M.D., Ph.D., Satoshi Tateda, M.D., Kenichiro Yahata, M.D.,
Supplemental Information. Autophagy in Oncogenic K-Ras. Promotes Basal Extrusion. of Epithelial Cells by Degrading S1P. Current Biology, Volume 24
 Current Biology, Volume 24 Supplemental Information Autophagy in Oncogenic K-Ras Promotes Basal Extrusion of Epithelial Cells by Degrading S1P Gloria Slattum, Yapeng Gu, Roger Sabbadini, and Jody Rosenblatt
Current Biology, Volume 24 Supplemental Information Autophagy in Oncogenic K-Ras Promotes Basal Extrusion of Epithelial Cells by Degrading S1P Gloria Slattum, Yapeng Gu, Roger Sabbadini, and Jody Rosenblatt
Imaging ischemic strokes: Correlating radiological findings with the pathophysiological evolution of an infarct
 Imaging ischemic strokes: Correlating radiological findings with the pathophysiological evolution of an infarct Jay Chyung,, PhD, HMS III Patient A: history 91 y.o. woman Acute onset R sided weakness and
Imaging ischemic strokes: Correlating radiological findings with the pathophysiological evolution of an infarct Jay Chyung,, PhD, HMS III Patient A: history 91 y.o. woman Acute onset R sided weakness and
6. TNF-α regulates oxidative stress, mitochondrial function and autophagy in neuronal cells
 6. TNF-α regulates oxidative stress, mitochondrial function and autophagy in neuronal cells 6.1 TNF-α induces mitochondrial oxidative stress in SH-SY5Y cells. The dysregulation of mitochondria and oxidative
6. TNF-α regulates oxidative stress, mitochondrial function and autophagy in neuronal cells 6.1 TNF-α induces mitochondrial oxidative stress in SH-SY5Y cells. The dysregulation of mitochondria and oxidative
Fluorescence Microscopy
 Fluorescence Microscopy Imaging Organelles Mitochondria Lysosomes Nuclei Endoplasmic Reticulum Plasma Membrane F-Actin AAT Bioquest Introduction: Organelle-Selective Stains Organelles are tiny, specialized
Fluorescence Microscopy Imaging Organelles Mitochondria Lysosomes Nuclei Endoplasmic Reticulum Plasma Membrane F-Actin AAT Bioquest Introduction: Organelle-Selective Stains Organelles are tiny, specialized
Supplementary Figure 1
 Supplementary Figure 1 AAV-GFP injection in the MEC of the mouse brain C57Bl/6 mice at 4 months of age were injected with AAV-GFP into the MEC and sacrificed at 7 days post injection (dpi). (a) Brains
Supplementary Figure 1 AAV-GFP injection in the MEC of the mouse brain C57Bl/6 mice at 4 months of age were injected with AAV-GFP into the MEC and sacrificed at 7 days post injection (dpi). (a) Brains
TFEB-mediated increase in peripheral lysosomes regulates. Store Operated Calcium Entry
 TFEB-mediated increase in peripheral lysosomes regulates Store Operated Calcium Entry Luigi Sbano, Massimo Bonora, Saverio Marchi, Federica Baldassari, Diego L. Medina, Andrea Ballabio, Carlotta Giorgi
TFEB-mediated increase in peripheral lysosomes regulates Store Operated Calcium Entry Luigi Sbano, Massimo Bonora, Saverio Marchi, Federica Baldassari, Diego L. Medina, Andrea Ballabio, Carlotta Giorgi
Supporting Information
 Copyright WILEY-VCH Verlag GmbH & Co. KGaA, 69469 Weinheim, Germany, 212. Supporting Information for Adv. Funct. Mater., DOI:.2/adfm.2122233 MnO Nanocrystals: A Platform for Integration of MRI and Genuine
Copyright WILEY-VCH Verlag GmbH & Co. KGaA, 69469 Weinheim, Germany, 212. Supporting Information for Adv. Funct. Mater., DOI:.2/adfm.2122233 MnO Nanocrystals: A Platform for Integration of MRI and Genuine
Supplementary Figure 1: Hsp60 / IEC mice are embryonically lethal (A) Light microscopic pictures show mouse embryos at developmental stage E12.
 Supplementary Figure 1: Hsp60 / IEC mice are embryonically lethal (A) Light microscopic pictures show mouse embryos at developmental stage E12.5 and E13.5 prepared from uteri of dams and subsequently genotyped.
Supplementary Figure 1: Hsp60 / IEC mice are embryonically lethal (A) Light microscopic pictures show mouse embryos at developmental stage E12.5 and E13.5 prepared from uteri of dams and subsequently genotyped.
Effect of low temperatures on BAX and BCL2 proteins in rats with spinal cord ischemia reperfusion injury
 Effect of low temperatures on BAX and BCL2 proteins in rats with spinal cord ischemia reperfusion injury P. Zhu 1 *, M.Y. Zhao 1,2 *, X.H. Li 1 *, Q. Fu 1, Z.F. Zhou 1, C.F. Huang 1, X.S. Zhang 1, H.L.
Effect of low temperatures on BAX and BCL2 proteins in rats with spinal cord ischemia reperfusion injury P. Zhu 1 *, M.Y. Zhao 1,2 *, X.H. Li 1 *, Q. Fu 1, Z.F. Zhou 1, C.F. Huang 1, X.S. Zhang 1, H.L.
Mechanisms of Cell Injury
 Causes of Cell Injury 1- oxygen deprivation (anoxia) 2- physical agents 3- chemical agents 4- infections agents 5- immunologic reactions 6- genetic defects 7- nutritional imbalances Mechanisms of Cell
Causes of Cell Injury 1- oxygen deprivation (anoxia) 2- physical agents 3- chemical agents 4- infections agents 5- immunologic reactions 6- genetic defects 7- nutritional imbalances Mechanisms of Cell
c Ischemia (30 min) Reperfusion (8 w) Supplementary Figure bp 300 bp Ischemia (30 min) Reperfusion (4 h) Dox 20 mg/kg i.p.
 a Marker Ripk3 +/ 5 bp 3 bp b Ischemia (3 min) Reperfusion (4 h) d 2 mg/kg i.p. 1 w 5 w Sacrifice for IF size A subset for echocardiography and morphological analysis c Ischemia (3 min) Reperfusion (8
a Marker Ripk3 +/ 5 bp 3 bp b Ischemia (3 min) Reperfusion (4 h) d 2 mg/kg i.p. 1 w 5 w Sacrifice for IF size A subset for echocardiography and morphological analysis c Ischemia (3 min) Reperfusion (8
Supplemental Figures:
 Supplemental Figures: Figure 1: Intracellular distribution of VWF by electron microscopy in human endothelial cells. a) Immunogold labeling of LC3 demonstrating an LC3-positive autophagosome (white arrow)
Supplemental Figures: Figure 1: Intracellular distribution of VWF by electron microscopy in human endothelial cells. a) Immunogold labeling of LC3 demonstrating an LC3-positive autophagosome (white arrow)
Pretargeting and Bioorthogonal Click Chemistry-Mediated Endogenous Stem Cell Homing for Heart Repair
 Pretargeting and Bioorthogonal Click Chemistry-Mediated Endogenous Stem Cell Homing for Heart Repair Mouse Model of Myocardial Infarction (MI) All animal work was compliant with the Institutional Animal
Pretargeting and Bioorthogonal Click Chemistry-Mediated Endogenous Stem Cell Homing for Heart Repair Mouse Model of Myocardial Infarction (MI) All animal work was compliant with the Institutional Animal
Supplementary Figure 1.
 Supplementary Figure 1. Increased β cell mass and islet diameter in βtsc2 -/- mice up to 35 weeks A: Reconstruction of multiple anti-insulin immunofluorescence images showing differences in β cell mass
Supplementary Figure 1. Increased β cell mass and islet diameter in βtsc2 -/- mice up to 35 weeks A: Reconstruction of multiple anti-insulin immunofluorescence images showing differences in β cell mass
Comparison of Young and Old Cardiac Telocytes Using Atomic Force Microscopy
 Comparison of Young and Old Cardiac Telocytes Using Atomic Force Microscopy Jiali Luo 1, 2, 3, 4, a, Shanshan Feng 1, 2, 3, 4, b 1Key Laboratory of Regenerative Medicine, Ministry of Education, Jinan University,
Comparison of Young and Old Cardiac Telocytes Using Atomic Force Microscopy Jiali Luo 1, 2, 3, 4, a, Shanshan Feng 1, 2, 3, 4, b 1Key Laboratory of Regenerative Medicine, Ministry of Education, Jinan University,
Apoptosis Chapter 9. Neelu Yadav PhD
 Apoptosis Chapter 9 Neelu Yadav PhD Neelu.Yadav@Roswellpark.org 1 Apoptosis: Lecture outline Apoptosis a programmed cell death pathway in normal homeostasis Core Apoptosis cascade is conserved Compare
Apoptosis Chapter 9 Neelu Yadav PhD Neelu.Yadav@Roswellpark.org 1 Apoptosis: Lecture outline Apoptosis a programmed cell death pathway in normal homeostasis Core Apoptosis cascade is conserved Compare
Supplemental Information. Otic Mesenchyme Cells Regulate. Spiral Ganglion Axon Fasciculation. through a Pou3f4/EphA4 Signaling Pathway
 Neuron, Volume 73 Supplemental Information Otic Mesenchyme Cells Regulate Spiral Ganglion Axon Fasciculation through a Pou3f4/EphA4 Signaling Pathway Thomas M. Coate, Steven Raft, Xiumei Zhao, Aimee K.
Neuron, Volume 73 Supplemental Information Otic Mesenchyme Cells Regulate Spiral Ganglion Axon Fasciculation through a Pou3f4/EphA4 Signaling Pathway Thomas M. Coate, Steven Raft, Xiumei Zhao, Aimee K.
SUPPLEMENTAL MATERIAL. Supplementary Methods
 SUPPLEMENTAL MATERIAL Supplementary Methods Culture of cardiomyocytes, fibroblasts and cardiac microvascular endothelial cells The isolation and culturing of neonatal rat ventricular cardiomyocytes was
SUPPLEMENTAL MATERIAL Supplementary Methods Culture of cardiomyocytes, fibroblasts and cardiac microvascular endothelial cells The isolation and culturing of neonatal rat ventricular cardiomyocytes was
ab SREBP-2 Translocation Assay Kit (Cell-Based)
 ab133114 SREBP-2 Translocation Assay Kit (Cell-Based) Instructions for Use For analysis of translocation of SREBP-2 into nuclei. This product is for research use only and is not intended for diagnostic
ab133114 SREBP-2 Translocation Assay Kit (Cell-Based) Instructions for Use For analysis of translocation of SREBP-2 into nuclei. This product is for research use only and is not intended for diagnostic
SUPPLEMENTARY INFORMATION
 Figure S1 Induction of non-apoptotic death of SV40-transformed and primary DKO MEFs, and DKO thymocytes. (A-F) STS-induced non-apoptotic death of DKO MEF. (A, B) Reduced viability of DKO MEFs after exposure
Figure S1 Induction of non-apoptotic death of SV40-transformed and primary DKO MEFs, and DKO thymocytes. (A-F) STS-induced non-apoptotic death of DKO MEF. (A, B) Reduced viability of DKO MEFs after exposure
Supplemental Information. Menin Deficiency Leads to Depressive-like. Behaviors in Mice by Modulating. Astrocyte-Mediated Neuroinflammation
 Neuron, Volume 100 Supplemental Information Menin Deficiency Leads to Depressive-like Behaviors in Mice by Modulating Astrocyte-Mediated Neuroinflammation Lige Leng, Kai Zhuang, Zeyue Liu, Changquan Huang,
Neuron, Volume 100 Supplemental Information Menin Deficiency Leads to Depressive-like Behaviors in Mice by Modulating Astrocyte-Mediated Neuroinflammation Lige Leng, Kai Zhuang, Zeyue Liu, Changquan Huang,
Cell Quality Control. Peter Takizawa Department of Cell Biology
 Cell Quality Control Peter Takizawa Department of Cell Biology Cellular quality control reduces production of defective proteins. Cells have many quality control systems to ensure that cell does not build
Cell Quality Control Peter Takizawa Department of Cell Biology Cellular quality control reduces production of defective proteins. Cells have many quality control systems to ensure that cell does not build
(A) PCR primers (arrows) designed to distinguish wild type (P1+P2), targeted (P1+P2) and excised (P1+P3)14-
 1 Supplemental Figure Legends Figure S1. Mammary tumors of ErbB2 KI mice with 14-3-3σ ablation have elevated ErbB2 transcript levels and cell proliferation (A) PCR primers (arrows) designed to distinguish
1 Supplemental Figure Legends Figure S1. Mammary tumors of ErbB2 KI mice with 14-3-3σ ablation have elevated ErbB2 transcript levels and cell proliferation (A) PCR primers (arrows) designed to distinguish
mm Distance (mm)
 b a Magnet Illumination Coverslips MPs Objective 2575 µm 1875 µm 1575 µm 1075 µm 875 µm 545 µm 20µm 2 3 0.5 0.3mm 1 1000 100 10 1 0.1 1000 100 10 1 0.1 Field Induction (Gauss) 1.5 0 5 10 15 20 Distance
b a Magnet Illumination Coverslips MPs Objective 2575 µm 1875 µm 1575 µm 1075 µm 875 µm 545 µm 20µm 2 3 0.5 0.3mm 1 1000 100 10 1 0.1 1000 100 10 1 0.1 Field Induction (Gauss) 1.5 0 5 10 15 20 Distance
#19 Apoptosis Chapter 9. Neelu Yadav PhD
 #19 Apoptosis Chapter 9 Neelu Yadav PhD Neelu.Yadav@Roswellpark.org Why cells decide to die? - Stress, harmful, not needed - Completed its life span Death stimulation or Stress Cell Survival Death Functions
#19 Apoptosis Chapter 9 Neelu Yadav PhD Neelu.Yadav@Roswellpark.org Why cells decide to die? - Stress, harmful, not needed - Completed its life span Death stimulation or Stress Cell Survival Death Functions
1. Materials and Methods 1.1 Animals experiments process The experiments were approved by the Institution Animal Ethics Committee of Jilin University
 1. Materials and Methods 1.1 Animals experiments process The experiments were approved by the Institution Animal Ethics Committee of Jilin University (Reference NO. 2015-003). 96 Kunming (KM) mice (8 weeks;
1. Materials and Methods 1.1 Animals experiments process The experiments were approved by the Institution Animal Ethics Committee of Jilin University (Reference NO. 2015-003). 96 Kunming (KM) mice (8 weeks;
Diabetes Mellitus and Dementia. Andrea Shelton & Adena Zadourian
 Diabetes Mellitus and Dementia Andrea Shelton & Adena Zadourian Abstract Diabetes mellitus increases the risk for developing dementia...but there is inconsistency with the subtypes of dementia Diabetes
Diabetes Mellitus and Dementia Andrea Shelton & Adena Zadourian Abstract Diabetes mellitus increases the risk for developing dementia...but there is inconsistency with the subtypes of dementia Diabetes
(a) Significant biological processes (upper panel) and disease biomarkers (lower panel)
 Supplementary Figure 1. Functional enrichment analyses of secretomic proteins. (a) Significant biological processes (upper panel) and disease biomarkers (lower panel) 2 involved by hrab37-mediated secretory
Supplementary Figure 1. Functional enrichment analyses of secretomic proteins. (a) Significant biological processes (upper panel) and disease biomarkers (lower panel) 2 involved by hrab37-mediated secretory
A549 and A549-fLuc cells were maintained in high glucose Dulbecco modified
 Cell culture and animal model A549 and A549-fLuc cells were maintained in high glucose Dulbecco modified Eagle medium supplemented with 10% fetal bovine serum at 37 C in humidified atmosphere containing
Cell culture and animal model A549 and A549-fLuc cells were maintained in high glucose Dulbecco modified Eagle medium supplemented with 10% fetal bovine serum at 37 C in humidified atmosphere containing
Effect of Taurine on Acinar Cell Apoptosis and Pancreatic Fibrosis in Dibutyltin Dichloride-induced Chronic Pancreatitis
 212 66 4 329334 Effect of Taurine on Acinar Cell Apoptosis and Pancreatic Fibrosis in Dibutyltin Dichloride-induced Chronic Pancreatitis a,c a* b b a a b a a b c 33 66 4 ʼ 6 6 6 28 6 5 5 5 28 45 ʼ ʼ ʼ
212 66 4 329334 Effect of Taurine on Acinar Cell Apoptosis and Pancreatic Fibrosis in Dibutyltin Dichloride-induced Chronic Pancreatitis a,c a* b b a a b a a b c 33 66 4 ʼ 6 6 6 28 6 5 5 5 28 45 ʼ ʼ ʼ
Proteomic profiling of small-molecule inhibitors reveals dispensability of MTH1 for cancer cell survival
 Supplementary Information for Proteomic profiling of small-molecule inhibitors reveals dispensability of MTH1 for cancer cell survival Tatsuro Kawamura 1, Makoto Kawatani 1, Makoto Muroi, Yasumitsu Kondoh,
Supplementary Information for Proteomic profiling of small-molecule inhibitors reveals dispensability of MTH1 for cancer cell survival Tatsuro Kawamura 1, Makoto Kawatani 1, Makoto Muroi, Yasumitsu Kondoh,
Pathophysiology and treatment of focal cerebral ischemia
 J Neurosurg 77: 169-184, 1992 Review Article Pathophysiology and treatment of focal cerebral ischemia Part I: Pathophysiology Bo K. SIESJO, M.D. Laborutory for Experimental Bruin Reseurch, Experrmc~ntul
J Neurosurg 77: 169-184, 1992 Review Article Pathophysiology and treatment of focal cerebral ischemia Part I: Pathophysiology Bo K. SIESJO, M.D. Laborutory for Experimental Bruin Reseurch, Experrmc~ntul
A. Generation and characterization of Ras-expressing autophagycompetent
 Supplemental Material Supplemental Figure Legends Fig. S1 A. Generation and characterization of Ras-expressing autophagycompetent and -deficient cell lines. HA-tagged H-ras V12 was stably expressed in
Supplemental Material Supplemental Figure Legends Fig. S1 A. Generation and characterization of Ras-expressing autophagycompetent and -deficient cell lines. HA-tagged H-ras V12 was stably expressed in
Evaluation of directed and random motility in microslides Assessment of leukocyte adhesion in flow chambers
 Evaluation of directed and random motility in microslides Motility experiments in IBIDI microslides, image acquisition and processing were performed as described. PMN, which ended up in an angle < 180
Evaluation of directed and random motility in microslides Motility experiments in IBIDI microslides, image acquisition and processing were performed as described. PMN, which ended up in an angle < 180
The subcortical maternal complex controls symmetric division of mouse zygotes by
 The subcortical maternal complex controls symmetric division of mouse zygotes by regulating F-actin dynamics Xing-Jiang Yu 1,2, Zhaohong Yi 1, Zheng Gao 1,2, Dan-dan Qin 1,2, Yanhua Zhai 1, Xue Chen 1,
The subcortical maternal complex controls symmetric division of mouse zygotes by regulating F-actin dynamics Xing-Jiang Yu 1,2, Zhaohong Yi 1, Zheng Gao 1,2, Dan-dan Qin 1,2, Yanhua Zhai 1, Xue Chen 1,
SCIRF Award #2016 I-03 PI: Azizul Haque, PhD Grant Title: Neuron-specific Enolase and SCI
 SCIRF Award #2016 I-03 PI: Azizul Haque, PhD Grant Title: Neuron-specific Enolase and SCI 10-month Technical Progress Report Enolase is a multifunctional glycolytic enzyme involved in growth control, hypoxia,
SCIRF Award #2016 I-03 PI: Azizul Haque, PhD Grant Title: Neuron-specific Enolase and SCI 10-month Technical Progress Report Enolase is a multifunctional glycolytic enzyme involved in growth control, hypoxia,
Disrupting GluA2-GAPDH Interaction Affects Axon and Dendrite Development
 Disrupting GluA2-GAPDH Interaction Affects Axon and Dendrite Development 1 Frankie Hang Fung Lee, 1 Ping Su, 1 Yu Feng Xie, 1 Kyle Ethan Wang, 2 Qi Wan and 1,3 Fang Liu 1 Campbell Research Institute, Centre
Disrupting GluA2-GAPDH Interaction Affects Axon and Dendrite Development 1 Frankie Hang Fung Lee, 1 Ping Su, 1 Yu Feng Xie, 1 Kyle Ethan Wang, 2 Qi Wan and 1,3 Fang Liu 1 Campbell Research Institute, Centre
ab LDL Uptake Assay Kit (Cell-Based)
 ab133127 LDL Uptake Assay Kit (Cell-Based) Instructions for Use For the detection of LDL uptake into cultured cells. This product is for research use only and is not intended for diagnostic use. Version
ab133127 LDL Uptake Assay Kit (Cell-Based) Instructions for Use For the detection of LDL uptake into cultured cells. This product is for research use only and is not intended for diagnostic use. Version
Supplementary Figure 1. Characterization of human carotid plaques. (a) Flash-frozen human plaques were separated into vulnerable (V) and stable (S),
 Supplementary Figure 1. Characterization of human carotid plaques. (a) Flash-frozen human plaques were separated into vulnerable (V) and stable (S), regions which were then quantified for mean fluorescence
Supplementary Figure 1. Characterization of human carotid plaques. (a) Flash-frozen human plaques were separated into vulnerable (V) and stable (S), regions which were then quantified for mean fluorescence
Dissected tissues were minced and lysed in lysis buffer (1x Tris buffered saline (TBS), 1% NP-40,
 Data Supplement for Dincheva et al., Effect of Early-Life Fluoxetine on Anxiety-Like Behaviors in BDNF Val66Met Mice. Am J Psychiatry (doi: 10.1176/appi.ajp.2017.15121592) Contents Supplemental Methods
Data Supplement for Dincheva et al., Effect of Early-Life Fluoxetine on Anxiety-Like Behaviors in BDNF Val66Met Mice. Am J Psychiatry (doi: 10.1176/appi.ajp.2017.15121592) Contents Supplemental Methods
Supplementary Table 1. Antibodies and dilutions used in the immunohistochemical study
 Supplementary Table 1. Antibodies and dilutions used in the immunohistochemical study Antigen Species Clone Source Dilution Insulin Guinea pig - Dako, Carpinteria, 1:200 Glucagon Rabbit - Dako, Carpinteria,
Supplementary Table 1. Antibodies and dilutions used in the immunohistochemical study Antigen Species Clone Source Dilution Insulin Guinea pig - Dako, Carpinteria, 1:200 Glucagon Rabbit - Dako, Carpinteria,
Effect of starvation-induced autophagy on cell cycle of tumor cells
 [Chinese Journal of Cancer 27:8, 102-108; August 2008]; 2008 Sun Yat-Sen University Cancer Center Basic Research Paper Effect of starvation-induced autophagy on cell cycle of tumor cells Jun-Na Ge, 1 Dan
[Chinese Journal of Cancer 27:8, 102-108; August 2008]; 2008 Sun Yat-Sen University Cancer Center Basic Research Paper Effect of starvation-induced autophagy on cell cycle of tumor cells Jun-Na Ge, 1 Dan
Antibodies for Unfolded Protein Response
 Novus-lu-2945 Antibodies for Unfolded rotein Response Unfolded roteins ER lumen GR78 IRE-1 GR78 ERK Cytosol GR78 TRAF2 ASK1 JNK Activator Intron RIDD elf2α Degraded mrna XB1 mrna Translation XB1-S (p50)
Novus-lu-2945 Antibodies for Unfolded rotein Response Unfolded roteins ER lumen GR78 IRE-1 GR78 ERK Cytosol GR78 TRAF2 ASK1 JNK Activator Intron RIDD elf2α Degraded mrna XB1 mrna Translation XB1-S (p50)
About OMICS Group Conferences
 About OMICS Group OMICS Group International is an amalgamation of Open Access publications and worldwide international science conferences and events. Established in the year 2007 with the sole aim of
About OMICS Group OMICS Group International is an amalgamation of Open Access publications and worldwide international science conferences and events. Established in the year 2007 with the sole aim of
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION FOR Liver X Receptor α mediates hepatic triglyceride accumulation through upregulation of G0/G1 Switch Gene 2 (G0S2) expression I: SUPPLEMENTARY METHODS II: SUPPLEMENTARY FIGURES
SUPPLEMENTARY INFORMATION FOR Liver X Receptor α mediates hepatic triglyceride accumulation through upregulation of G0/G1 Switch Gene 2 (G0S2) expression I: SUPPLEMENTARY METHODS II: SUPPLEMENTARY FIGURES
A particular set of insults induces apoptosis (part 1), which, if inhibited, can switch to autophagy. At least in some cellular settings, autophagy se
 A particular set of insults induces apoptosis (part 1), which, if inhibited, can switch to autophagy. At least in some cellular settings, autophagy serves as a defence mechanism that prevents or retards
A particular set of insults induces apoptosis (part 1), which, if inhibited, can switch to autophagy. At least in some cellular settings, autophagy serves as a defence mechanism that prevents or retards
Essential Medium, containing 10% fetal bovine serum, 100 U/ml penicillin and 100 µg/ml streptomycin. Huvec were cultured in
 Supplemental data Methods Cell culture media formulations A-431 and U-87 MG cells were maintained in Dulbecco s Modified Eagle s Medium. FaDu cells were cultured in Eagle's Minimum Essential Medium, containing
Supplemental data Methods Cell culture media formulations A-431 and U-87 MG cells were maintained in Dulbecco s Modified Eagle s Medium. FaDu cells were cultured in Eagle's Minimum Essential Medium, containing
marker. DAPI labels nuclei. Flies were 20 days old. Scale bar is 5 µm. Ctrl is
 Supplementary Figure 1. (a) Nos is detected in glial cells in both control and GFAP R79H transgenic flies (arrows), but not in deletion mutant Nos Δ15 animals. Repo is a glial cell marker. DAPI labels
Supplementary Figure 1. (a) Nos is detected in glial cells in both control and GFAP R79H transgenic flies (arrows), but not in deletion mutant Nos Δ15 animals. Repo is a glial cell marker. DAPI labels
Supporting Information
 Supporting Information Pang et al. 10.1073/pnas.1322009111 SI Materials and Methods ELISAs. These assays were performed as previously described (1). ELISA plates (MaxiSorp Nunc; Thermo Fisher Scientific)
Supporting Information Pang et al. 10.1073/pnas.1322009111 SI Materials and Methods ELISAs. These assays were performed as previously described (1). ELISA plates (MaxiSorp Nunc; Thermo Fisher Scientific)
ab Membrane Fractionation Kit Instructions for Use For the rapid and simple separation of membrane, cytosolic and nuclear cellular fractions.
 ab139409 Membrane Fractionation Kit Instructions for Use For the rapid and simple separation of membrane, cytosolic and nuclear cellular fractions. This product is for research use only and is not intended
ab139409 Membrane Fractionation Kit Instructions for Use For the rapid and simple separation of membrane, cytosolic and nuclear cellular fractions. This product is for research use only and is not intended
Supplementary Figure 1. Confocal immunofluorescence showing mitochondrial translocation of Drp1. Cardiomyocytes treated with H 2 O 2 were prestained
 Supplementary Figure 1. Confocal immunofluorescence showing mitochondrial translocation of Drp1. Cardiomyocytes treated with H 2 O 2 were prestained with MitoTracker (red), then were immunostained with
Supplementary Figure 1. Confocal immunofluorescence showing mitochondrial translocation of Drp1. Cardiomyocytes treated with H 2 O 2 were prestained with MitoTracker (red), then were immunostained with
McWilliams et al., http :// /cgi /content /full /jcb /DC1
 Supplemental material JCB McWilliams et al., http ://www.jcb.org /cgi /content /full /jcb.201603039 /DC1 THE JOURNAL OF CELL BIOLOGY Figure S1. In vitro characterization of mito-qc. (A and B) Analysis
Supplemental material JCB McWilliams et al., http ://www.jcb.org /cgi /content /full /jcb.201603039 /DC1 THE JOURNAL OF CELL BIOLOGY Figure S1. In vitro characterization of mito-qc. (A and B) Analysis
Cell Injury MECHANISMS OF CELL INJURY
 Cell Injury MECHANISMS OF CELL INJURY The cellular response to injurious stimuli depends on the following factors: Type of injury, Its duration, and Its severity. Thus, low doses of toxins or a brief duration
Cell Injury MECHANISMS OF CELL INJURY The cellular response to injurious stimuli depends on the following factors: Type of injury, Its duration, and Its severity. Thus, low doses of toxins or a brief duration
Implication of aquaporins in ischemic stroke. New target?
 Implication of aquaporins in ischemic stroke. New target? Balseanu Tudor-Adrian, MD, PhD EXPERIMENTAL RESEARCH CENTER OF NORMAL AND PATHOLOGICAL AGING University of Medicine and Pharmacy of Craiova, Romania
Implication of aquaporins in ischemic stroke. New target? Balseanu Tudor-Adrian, MD, PhD EXPERIMENTAL RESEARCH CENTER OF NORMAL AND PATHOLOGICAL AGING University of Medicine and Pharmacy of Craiova, Romania
HCC1937 is the HCC1937-pcDNA3 cell line, which was derived from a breast cancer with a mutation
 SUPPLEMENTARY INFORMATION Materials and Methods Human cell lines and culture conditions HCC1937 is the HCC1937-pcDNA3 cell line, which was derived from a breast cancer with a mutation in exon 20 of BRCA1
SUPPLEMENTARY INFORMATION Materials and Methods Human cell lines and culture conditions HCC1937 is the HCC1937-pcDNA3 cell line, which was derived from a breast cancer with a mutation in exon 20 of BRCA1
A novel role for vitamin D: modulation of expression and function of the local renin angiotensin system in mouse pancreatic islets
 Diabetologia () 5:77 DOI.7/s5--- SHORT COMMUNICATION A novel role for vitamin D: modulation of expression and function of the local renin angiotensin system in mouse pancreatic islets Q. Cheng & Y. C.
Diabetologia () 5:77 DOI.7/s5--- SHORT COMMUNICATION A novel role for vitamin D: modulation of expression and function of the local renin angiotensin system in mouse pancreatic islets Q. Cheng & Y. C.
VEGFR2-Mediated Vascular Dilation as a Mechanism of VEGF-Induced Anemia and Bone Marrow Cell Mobilization
 Cell Reports, Volume 9 Supplemental Information VEGFR2-Mediated Vascular Dilation as a Mechanism of VEGF-Induced Anemia and Bone Marrow Cell Mobilization Sharon Lim, Yin Zhang, Danfang Zhang, Fang Chen,
Cell Reports, Volume 9 Supplemental Information VEGFR2-Mediated Vascular Dilation as a Mechanism of VEGF-Induced Anemia and Bone Marrow Cell Mobilization Sharon Lim, Yin Zhang, Danfang Zhang, Fang Chen,
Fang et al. NMuMG. PyVmT unstained Anti-CCR2-PE MDA-MB MCF MCF10A
 A NMuMG PyVmT 16.5+.5 47.+7.2 Fang et al. unstained Anti-CCR2-PE 4T1 Control 37.6+6.3 56.1+.65 MCF1A 16.1+3. MCF-7 3.1+5.4 MDA-M-231 42.1+5.5 unstained Secondary antibody only Anti-CCR2 SUPPLEMENTAL FIGURE
A NMuMG PyVmT 16.5+.5 47.+7.2 Fang et al. unstained Anti-CCR2-PE 4T1 Control 37.6+6.3 56.1+.65 MCF1A 16.1+3. MCF-7 3.1+5.4 MDA-M-231 42.1+5.5 unstained Secondary antibody only Anti-CCR2 SUPPLEMENTAL FIGURE
Supplementary Information POLO-LIKE KINASE 1 FACILITATES LOSS OF PTEN-INDUCED PROSTATE CANCER FORMATION
 Supplementary Information POLO-LIKE KINASE 1 FACILITATES LOSS OF PTEN-INDUCED PROSTATE CANCER FORMATION X. Shawn Liu 1, 3, Bing Song 2, 3, Bennett D. Elzey 3, 4, Timothy L. Ratliff 3, 4, Stephen F. Konieczny
Supplementary Information POLO-LIKE KINASE 1 FACILITATES LOSS OF PTEN-INDUCED PROSTATE CANCER FORMATION X. Shawn Liu 1, 3, Bing Song 2, 3, Bennett D. Elzey 3, 4, Timothy L. Ratliff 3, 4, Stephen F. Konieczny
Biodegradable Zwitterionic Nanogels with Long. Circulation for Antitumor Drug Delivery
 Supporting Information Biodegradable Zwitterionic Nanogels with Long Circulation for Antitumor Drug Delivery Yongzhi Men, Shaojun Peng, Peng Yang, Qin Jiang, Yanhui Zhang, Bin Shen, Pin Dong, *, Zhiqing
Supporting Information Biodegradable Zwitterionic Nanogels with Long Circulation for Antitumor Drug Delivery Yongzhi Men, Shaojun Peng, Peng Yang, Qin Jiang, Yanhui Zhang, Bin Shen, Pin Dong, *, Zhiqing
Instructions for Use. APO-AB Annexin V-Biotin Apoptosis Detection Kit 100 tests
 3URGXFW,QIRUPDWLRQ Sigma TACS Annexin V Apoptosis Detection Kits Instructions for Use APO-AB Annexin V-Biotin Apoptosis Detection Kit 100 tests For Research Use Only. Not for use in diagnostic procedures.
3URGXFW,QIRUPDWLRQ Sigma TACS Annexin V Apoptosis Detection Kits Instructions for Use APO-AB Annexin V-Biotin Apoptosis Detection Kit 100 tests For Research Use Only. Not for use in diagnostic procedures.
Defective Hepatic Autophagy in Obesity Promotes ER Stress and Causes Insulin Resistance
 Cell Metabolism, Volume 11 Supplemental Information Defective Hepatic Autophagy in Obesity Promotes ER Stress and Causes Insulin Resistance Ling Yang, Ping Li, Suneng Fu, Ediz S. Calay, and Gökhan S. Hotamisligil
Cell Metabolism, Volume 11 Supplemental Information Defective Hepatic Autophagy in Obesity Promotes ER Stress and Causes Insulin Resistance Ling Yang, Ping Li, Suneng Fu, Ediz S. Calay, and Gökhan S. Hotamisligil
Supplementary Materials. for Garmy-Susini, et al, Integrin 4 1 signaling is required for lymphangiogenesis and tumor metastasis
 Supplementary Materials for Garmy-Susini, et al, Integrin 4 1 signaling is required for lymphangiogenesis and tumor metastasis 1 Supplementary Figure Legends Supplementary Figure 1: Integrin expression
Supplementary Materials for Garmy-Susini, et al, Integrin 4 1 signaling is required for lymphangiogenesis and tumor metastasis 1 Supplementary Figure Legends Supplementary Figure 1: Integrin expression
Neuroprotective properties of epoetin alfa
 Nephrol Dial Transplant (2002) 17 [Suppl 1]: 8 12 Neuroprotective properties of epoetin alfa Anthony Cerami 1, Michael Brines 1, Pietro Ghezzi 1,2, Carla Cerami 1 and Loretta M. Itri 3 1 The Kenneth S.
Nephrol Dial Transplant (2002) 17 [Suppl 1]: 8 12 Neuroprotective properties of epoetin alfa Anthony Cerami 1, Michael Brines 1, Pietro Ghezzi 1,2, Carla Cerami 1 and Loretta M. Itri 3 1 The Kenneth S.
Muse Assays for Cell Analysis
 Muse Assays for Cell Analysis Multiple Assay Outputs for Cell Analysis Cell Health Cell Signalling Immunology Muse Count & Viability Kit Muse Cell Cycle Kit Muse Annexin V & Dead Cell Kit Muse Caspase
Muse Assays for Cell Analysis Multiple Assay Outputs for Cell Analysis Cell Health Cell Signalling Immunology Muse Count & Viability Kit Muse Cell Cycle Kit Muse Annexin V & Dead Cell Kit Muse Caspase
Pathophysiology and treatment of focal cerebral ischemia
 J Neurosurg 77:337-354, 1992 Review Article Pathophysiology and treatment of focal cerebral ischemia Part 11: Mechanisms of damage and treatment Bo K. SIESJO, M.D. Laboratory for Experimental Brain Research,
J Neurosurg 77:337-354, 1992 Review Article Pathophysiology and treatment of focal cerebral ischemia Part 11: Mechanisms of damage and treatment Bo K. SIESJO, M.D. Laboratory for Experimental Brain Research,
Nature Neuroscience: doi: /nn Supplementary Figure 1. PICALM expression in brain capillary endothelium in human brain and in mouse brain.
 Supplementary Figure 1 PICALM expression in brain capillary endothelium in human brain and in mouse brain. a, Double immunostaining for PICALM (red, left) and lectin positive endothelial profiles (blue,
Supplementary Figure 1 PICALM expression in brain capillary endothelium in human brain and in mouse brain. a, Double immunostaining for PICALM (red, left) and lectin positive endothelial profiles (blue,
death if the deficit is prolonged. Children and adults exposed to hypoglycaemia can develop long-term impairment of cognitive function (Blattner,
 Introduction Glucose is the primary source of fuel for the cells of the brain. Brain is dependent on a continuous supply of glucose diffusing from the blood into the interstitial tissue within the central
Introduction Glucose is the primary source of fuel for the cells of the brain. Brain is dependent on a continuous supply of glucose diffusing from the blood into the interstitial tissue within the central
Effect of anesthetic post-treatment on blood-brain barrier integrity and cerebral edema after transient cerebral ischemia in rats
 Effect of anesthetic post-treatment on blood-brain barrier integrity and cerebral edema after transient cerebral ischemia in rats Jae Hoon Lee Department of Medicine The Graduate School, Yonsei University
Effect of anesthetic post-treatment on blood-brain barrier integrity and cerebral edema after transient cerebral ischemia in rats Jae Hoon Lee Department of Medicine The Graduate School, Yonsei University
Protective effect of ginsenoside Rg1 against MPTP-induced apoptosis in mouse substantia nigra neurons 1
 Chen XC et al / Acta Pharmacol Sin 2002 Sep; 23 (9): 829-834 829 2002, Acta Pharmacologica Sinica ISSN 1671-4083 Shanghai Institute of Materia Medica Chinese Academy of Sciences http://www.chinaphar.com
Chen XC et al / Acta Pharmacol Sin 2002 Sep; 23 (9): 829-834 829 2002, Acta Pharmacologica Sinica ISSN 1671-4083 Shanghai Institute of Materia Medica Chinese Academy of Sciences http://www.chinaphar.com
Elucidation of Viral Replication Mechanisms in an Animal Model for Multiple Sclerosis
 Elucidation of Viral Replication Mechanisms in an Animal Model for Multiple Sclerosis Introduction It is often difficult to research human diseases due to obvious ethical implications. However, when an
Elucidation of Viral Replication Mechanisms in an Animal Model for Multiple Sclerosis Introduction It is often difficult to research human diseases due to obvious ethical implications. However, when an
Detection of Apoptosis in Primary Cells by Annexin V Binding Using the Agilent 2100 Bioanalyzer. Application Note
 Detection of Apoptosis in Primary Cells by Annexin V Binding Using the Agilent 2100 Bioanalyzer Application Note Samuel D. H. Chan Marc Valer and Tobias Preckel, Introduction The Agilent 2100 bioanalyzer
Detection of Apoptosis in Primary Cells by Annexin V Binding Using the Agilent 2100 Bioanalyzer Application Note Samuel D. H. Chan Marc Valer and Tobias Preckel, Introduction The Agilent 2100 bioanalyzer
TSH Receptor Monoclonal Antibody (49) Catalog Number MA3-218 Product data sheet
 Website: thermofisher.com Customer Service (US): 1 800 955 6288 ext. 1 Technical Support (US): 1 800 955 6288 ext. 441 TSH Receptor Monoclonal Antibody (49) Catalog Number MA3-218 Product data sheet Details
Website: thermofisher.com Customer Service (US): 1 800 955 6288 ext. 1 Technical Support (US): 1 800 955 6288 ext. 441 TSH Receptor Monoclonal Antibody (49) Catalog Number MA3-218 Product data sheet Details
Supplementary Figure 1.TRIM33 binds β-catenin in the nucleus. a & b, Co-IP of endogenous TRIM33 with β-catenin in HT-29 cells (a) and HEK 293T cells
 Supplementary Figure 1.TRIM33 binds β-catenin in the nucleus. a & b, Co-IP of endogenous TRIM33 with β-catenin in HT-29 cells (a) and HEK 293T cells (b). TRIM33 was immunoprecipitated, and the amount of
Supplementary Figure 1.TRIM33 binds β-catenin in the nucleus. a & b, Co-IP of endogenous TRIM33 with β-catenin in HT-29 cells (a) and HEK 293T cells (b). TRIM33 was immunoprecipitated, and the amount of
Reperfusion Injury: How Can We Reduce It?
 MI/CAD: Practical Question in Management of AMI Patients Reperfusion Injury: How Can We Reduce It? Hyun-Jai Cho, M.D., Ph.D Cardiovascular Center & Department of Internal Medicine Seoul National University
MI/CAD: Practical Question in Management of AMI Patients Reperfusion Injury: How Can We Reduce It? Hyun-Jai Cho, M.D., Ph.D Cardiovascular Center & Department of Internal Medicine Seoul National University
Experimental Assessment of Infarct Lesion Growth in Mice using Time-Resolved T2* MR Image Sequences
 Experimental Assessment of Infarct Lesion Growth in Mice using Time-Resolved T2* MR Image Sequences Nils Daniel Forkert 1, Dennis Säring 1, Andrea Eisenbeis 2, Frank Leypoldt 3, Jens Fiehler 2, Heinz Handels
Experimental Assessment of Infarct Lesion Growth in Mice using Time-Resolved T2* MR Image Sequences Nils Daniel Forkert 1, Dennis Säring 1, Andrea Eisenbeis 2, Frank Leypoldt 3, Jens Fiehler 2, Heinz Handels
Product Datasheet. CD133 Antibody NB Unit Size: 0.1 mg
 Product Datasheet CD133 Antibody NB120-16518 Unit Size: 0.1 mg Store at 4C short term. Aliquot and store at -20C long term. Avoid freeze-thaw cycles. Publications: 8 Protocols, Publications, Related Products,
Product Datasheet CD133 Antibody NB120-16518 Unit Size: 0.1 mg Store at 4C short term. Aliquot and store at -20C long term. Avoid freeze-thaw cycles. Publications: 8 Protocols, Publications, Related Products,
Boucher et al NCOMMS B
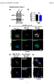 1 Supplementary Figure 1 (linked to Figure 1). mvegfr1 constitutively internalizes in endothelial cells. (a) Immunoblot of mflt1 from undifferentiated mouse embryonic stem (ES) cells with indicated genotypes;
1 Supplementary Figure 1 (linked to Figure 1). mvegfr1 constitutively internalizes in endothelial cells. (a) Immunoblot of mflt1 from undifferentiated mouse embryonic stem (ES) cells with indicated genotypes;
Nature Neuroscience: doi: /nn Supplementary Figure 1. Diverse anorexigenic signals induce c-fos expression in CEl PKC-δ + neurons
 Supplementary Figure 1 Diverse anorexigenic signals induce c-fos expression in CEl PKC-δ + neurons a-c. Quantification of CEl c-fos expression in mice intraperitoneal injected with anorexigenic drugs (a),
Supplementary Figure 1 Diverse anorexigenic signals induce c-fos expression in CEl PKC-δ + neurons a-c. Quantification of CEl c-fos expression in mice intraperitoneal injected with anorexigenic drugs (a),
(a) Schematic diagram of the FS mutation of UVRAG in exon 8 containing the highly instable
 Supplementary Figure 1. Frameshift (FS) mutation in UVRAG. (a) Schematic diagram of the FS mutation of UVRAG in exon 8 containing the highly instable A 10 DNA repeat, generating a premature stop codon
Supplementary Figure 1. Frameshift (FS) mutation in UVRAG. (a) Schematic diagram of the FS mutation of UVRAG in exon 8 containing the highly instable A 10 DNA repeat, generating a premature stop codon
Hypothalamic Autophagy and Regulation of Energy Balance
 Hypothalamic Autophagy and Regulation of Energy Balance Rajat Singh, MD Albert Einstein College of Medicine New York NuGOweek 211 Sept 6-9, 211 Autophagy Evolutionarily conserved cellular recycling program
Hypothalamic Autophagy and Regulation of Energy Balance Rajat Singh, MD Albert Einstein College of Medicine New York NuGOweek 211 Sept 6-9, 211 Autophagy Evolutionarily conserved cellular recycling program
SUPPLEMENTAL INFORMATION
 SUPPLEMENTAL INFORMATION EXPERIMENTAL PROCEDURES Tryptic digestion protection experiments - PCSK9 with Ab-3D5 (1:1 molar ratio) in 50 mm Tris, ph 8.0, 150 mm NaCl was incubated overnight at 4 o C. The
SUPPLEMENTAL INFORMATION EXPERIMENTAL PROCEDURES Tryptic digestion protection experiments - PCSK9 with Ab-3D5 (1:1 molar ratio) in 50 mm Tris, ph 8.0, 150 mm NaCl was incubated overnight at 4 o C. The
Supplementary Information
 Supplementary Information Title Degeneration and impaired regeneration of gray matter oligodendrocytes in amyotrophic lateral sclerosis Authors Shin H. Kang, Ying Li, Masahiro Fukaya, Ileana Lorenzini,
Supplementary Information Title Degeneration and impaired regeneration of gray matter oligodendrocytes in amyotrophic lateral sclerosis Authors Shin H. Kang, Ying Li, Masahiro Fukaya, Ileana Lorenzini,
Expression of acid base transporters in the kidney collecting duct in Slc2a7 -/-
 Supplemental Material Results. Expression of acid base transporters in the kidney collecting duct in Slc2a7 -/- and Slc2a7 -/- mice. The expression of AE1 in the kidney was examined in Slc26a7 KO mice.
Supplemental Material Results. Expression of acid base transporters in the kidney collecting duct in Slc2a7 -/- and Slc2a7 -/- mice. The expression of AE1 in the kidney was examined in Slc26a7 KO mice.
